CN114146021A - Cosmetic composition containing epidermal growth factor and preparation method thereof - Google Patents
Cosmetic composition containing epidermal growth factor and preparation method thereof Download PDFInfo
- Publication number
- CN114146021A CN114146021A CN202111678259.8A CN202111678259A CN114146021A CN 114146021 A CN114146021 A CN 114146021A CN 202111678259 A CN202111678259 A CN 202111678259A CN 114146021 A CN114146021 A CN 114146021A
- Authority
- CN
- China
- Prior art keywords
- cosmetic composition
- growth factor
- epidermal growth
- polyoxyethylene
- tween
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 239000002537 cosmetic Substances 0.000 title claims abstract description 47
- 239000000203 mixture Substances 0.000 title claims abstract description 36
- 102000009024 Epidermal Growth Factor Human genes 0.000 title claims abstract description 25
- 101800003838 Epidermal growth factor Proteins 0.000 title claims abstract description 23
- 229940116977 epidermal growth factor Drugs 0.000 title claims abstract description 23
- VBEQCZHXXJYVRD-GACYYNSASA-N uroanthelone Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CS)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C(C)C)[C@@H](C)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C1=CC=C(O)C=C1 VBEQCZHXXJYVRD-GACYYNSASA-N 0.000 title claims abstract description 23
- 238000002360 preparation method Methods 0.000 title abstract description 9
- 229920000747 poly(lactic acid) Polymers 0.000 claims abstract description 11
- 239000004626 polylactic acid Substances 0.000 claims abstract description 11
- 239000002736 nonionic surfactant Substances 0.000 claims abstract description 5
- 239000011159 matrix material Substances 0.000 claims abstract description 4
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 claims description 29
- PRAKJMSDJKAYCZ-UHFFFAOYSA-N squalane Chemical compound CC(C)CCCC(C)CCCC(C)CCCCC(C)CCCC(C)CCCC(C)C PRAKJMSDJKAYCZ-UHFFFAOYSA-N 0.000 claims description 18
- -1 polyoxyethylene Polymers 0.000 claims description 17
- 238000002156 mixing Methods 0.000 claims description 15
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 14
- 229920002385 Sodium hyaluronate Polymers 0.000 claims description 10
- 235000011187 glycerol Nutrition 0.000 claims description 10
- GVUGOAYIVIDWIO-UFWWTJHBSA-N nepidermin Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)NC(=O)CNC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H](CS)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CS)NC(=O)[C@H](C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C(C)C)C(C)C)C1=CC=C(O)C=C1 GVUGOAYIVIDWIO-UFWWTJHBSA-N 0.000 claims description 10
- 229940010747 sodium hyaluronate Drugs 0.000 claims description 10
- YWIVKILSMZOHHF-QJZPQSOGSA-N sodium;(2s,3s,4s,5r,6r)-6-[(2s,3r,4r,5s,6r)-3-acetamido-2-[(2s,3s,4r,5r,6r)-6-[(2r,3r,4r,5s,6r)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2- Chemical group [Na+].CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 YWIVKILSMZOHHF-QJZPQSOGSA-N 0.000 claims description 10
- 101500025419 Homo sapiens Epidermal growth factor Proteins 0.000 claims description 9
- 229920001213 Polysorbate 20 Polymers 0.000 claims description 9
- 229940116978 human epidermal growth factor Drugs 0.000 claims description 9
- JXTPJDDICSTXJX-UHFFFAOYSA-N n-Triacontane Natural products CCCCCCCCCCCCCCCCCCCCCCCCCCCCCC JXTPJDDICSTXJX-UHFFFAOYSA-N 0.000 claims description 9
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 claims description 9
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 claims description 9
- 229940032094 squalane Drugs 0.000 claims description 9
- 239000003906 humectant Substances 0.000 claims description 7
- 239000000843 powder Substances 0.000 claims description 7
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 6
- 230000002421 anti-septic effect Effects 0.000 claims description 6
- 238000001816 cooling Methods 0.000 claims description 6
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 claims description 6
- 238000010438 heat treatment Methods 0.000 claims description 6
- 238000009849 vacuum degassing Methods 0.000 claims description 6
- 229920003171 Poly (ethylene oxide) Polymers 0.000 claims description 5
- 239000002245 particle Substances 0.000 claims description 5
- 229920005862 polyol Polymers 0.000 claims description 5
- 150000003077 polyols Chemical class 0.000 claims description 5
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 claims description 4
- 239000003921 oil Substances 0.000 claims description 4
- 238000000034 method Methods 0.000 claims description 3
- FFJCNSLCJOQHKM-CLFAGFIQSA-N (z)-1-[(z)-octadec-9-enoxy]octadec-9-ene Chemical compound CCCCCCCC\C=C/CCCCCCCCOCCCCCCCC\C=C/CCCCCCCC FFJCNSLCJOQHKM-CLFAGFIQSA-N 0.000 claims description 2
- RFVNOJDQRGSOEL-UHFFFAOYSA-N 2-hydroxyethyl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCCO RFVNOJDQRGSOEL-UHFFFAOYSA-N 0.000 claims description 2
- GWFGDXZQZYMSMJ-UHFFFAOYSA-N Octadecansaeure-heptadecylester Natural products CCCCCCCCCCCCCCCCCOC(=O)CCCCCCCCCCCCCCCCC GWFGDXZQZYMSMJ-UHFFFAOYSA-N 0.000 claims description 2
- 239000002202 Polyethylene glycol Substances 0.000 claims description 2
- 229920001214 Polysorbate 60 Polymers 0.000 claims description 2
- 235000019437 butane-1,3-diol Nutrition 0.000 claims description 2
- 239000004359 castor oil Substances 0.000 claims description 2
- 235000019438 castor oil Nutrition 0.000 claims description 2
- 229940081733 cetearyl alcohol Drugs 0.000 claims description 2
- 239000004205 dimethyl polysiloxane Substances 0.000 claims description 2
- SZXQTJUDPRGNJN-UHFFFAOYSA-N dipropylene glycol Chemical compound OCCCOCCCO SZXQTJUDPRGNJN-UHFFFAOYSA-N 0.000 claims description 2
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 claims description 2
- 239000004519 grease Substances 0.000 claims description 2
- GLDOVTGHNKAZLK-UHFFFAOYSA-N octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 claims description 2
- NKBWPOSQERPBFI-UHFFFAOYSA-N octadecyl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCCOC(=O)CCCCCCCCCCCCCCCCC NKBWPOSQERPBFI-UHFFFAOYSA-N 0.000 claims description 2
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 claims description 2
- 229920001223 polyethylene glycol Polymers 0.000 claims description 2
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 claims description 2
- 229920001451 polypropylene glycol Polymers 0.000 claims description 2
- 229920000136 polysorbate Polymers 0.000 claims description 2
- 229920000053 polysorbate 80 Polymers 0.000 claims description 2
- 229940058015 1,3-butylene glycol Drugs 0.000 claims 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims 1
- 239000004166 Lanolin Substances 0.000 claims 1
- PHYFQTYBJUILEZ-UHFFFAOYSA-N Trioleoylglycerol Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC(OC(=O)CCCCCCCC=CCCCCCCCC)COC(=O)CCCCCCCC=CCCCCCCCC PHYFQTYBJUILEZ-UHFFFAOYSA-N 0.000 claims 1
- 229940039717 lanolin Drugs 0.000 claims 1
- 235000019388 lanolin Nutrition 0.000 claims 1
- 150000005846 sugar alcohols Polymers 0.000 claims 1
- PHYFQTYBJUILEZ-IUPFWZBJSA-N triolein Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC(OC(=O)CCCCCCC\C=C/CCCCCCCC)COC(=O)CCCCCCC\C=C/CCCCCCCC PHYFQTYBJUILEZ-IUPFWZBJSA-N 0.000 claims 1
- 229940117972 triolein Drugs 0.000 claims 1
- 230000032683 aging Effects 0.000 abstract description 6
- 230000002500 effect on skin Effects 0.000 abstract description 6
- 230000000694 effects Effects 0.000 abstract description 6
- 210000002950 fibroblast Anatomy 0.000 abstract description 6
- 230000007774 longterm Effects 0.000 abstract description 3
- 239000003755 preservative agent Substances 0.000 abstract description 2
- 230000002335 preservative effect Effects 0.000 abstract description 2
- 230000035755 proliferation Effects 0.000 abstract description 2
- 230000002035 prolonged effect Effects 0.000 abstract description 2
- 230000001737 promoting effect Effects 0.000 abstract 1
- 210000003491 skin Anatomy 0.000 description 25
- 239000000047 product Substances 0.000 description 12
- 239000008367 deionised water Substances 0.000 description 8
- 229910021641 deionized water Inorganic materials 0.000 description 8
- 210000004027 cell Anatomy 0.000 description 7
- 210000002615 epidermis Anatomy 0.000 description 7
- 102000008186 Collagen Human genes 0.000 description 6
- 108010035532 Collagen Proteins 0.000 description 6
- 230000010261 cell growth Effects 0.000 description 6
- 229920001436 collagen Polymers 0.000 description 6
- 210000004207 dermis Anatomy 0.000 description 6
- 239000000835 fiber Substances 0.000 description 5
- 239000003102 growth factor Substances 0.000 description 5
- 210000004304 subcutaneous tissue Anatomy 0.000 description 5
- 230000000052 comparative effect Effects 0.000 description 4
- 206010033675 panniculitis Diseases 0.000 description 4
- 230000009759 skin aging Effects 0.000 description 4
- 210000001519 tissue Anatomy 0.000 description 4
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- 230000001153 anti-wrinkle effect Effects 0.000 description 3
- 238000004140 cleaning Methods 0.000 description 3
- 241000512259 Ascophyllum nodosum Species 0.000 description 2
- XUMBMVFBXHLACL-UHFFFAOYSA-N Melanin Chemical compound O=C1C(=O)C(C2=CNC3=C(C(C(=O)C4=C32)=O)C)=C2C4=CNC2=C1C XUMBMVFBXHLACL-UHFFFAOYSA-N 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 101710098940 Pro-epidermal growth factor Proteins 0.000 description 2
- 238000002835 absorbance Methods 0.000 description 2
- 235000011399 aloe vera Nutrition 0.000 description 2
- 239000002299 complementary DNA Substances 0.000 description 2
- 210000002808 connective tissue Anatomy 0.000 description 2
- 210000004177 elastic tissue Anatomy 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 239000000284 extract Substances 0.000 description 2
- 230000012010 growth Effects 0.000 description 2
- 230000003394 haemopoietic effect Effects 0.000 description 2
- BJRNKVDFDLYUGJ-RMPHRYRLSA-N hydroquinone O-beta-D-glucopyranoside Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1OC1=CC=C(O)C=C1 BJRNKVDFDLYUGJ-RMPHRYRLSA-N 0.000 description 2
- 239000006210 lotion Substances 0.000 description 2
- LXCFILQKKLGQFO-UHFFFAOYSA-N methylparaben Chemical compound COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 2
- 239000003226 mitogen Substances 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 235000019198 oils Nutrition 0.000 description 2
- 239000013612 plasmid Substances 0.000 description 2
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 2
- 210000001732 sebaceous gland Anatomy 0.000 description 2
- 230000004936 stimulating effect Effects 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 210000000106 sweat gland Anatomy 0.000 description 2
- 230000002087 whitening effect Effects 0.000 description 2
- 230000037303 wrinkles Effects 0.000 description 2
- DIGQNXIGRZPYDK-WKSCXVIASA-N (2R)-6-amino-2-[[2-[[(2S)-2-[[2-[[(2R)-2-[[(2S)-2-[[(2R,3S)-2-[[2-[[(2S)-2-[[2-[[(2S)-2-[[(2S)-2-[[(2R)-2-[[(2S,3S)-2-[[(2R)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[2-[[(2S)-2-[[(2R)-2-[[2-[[2-[[2-[(2-amino-1-hydroxyethylidene)amino]-3-carboxy-1-hydroxypropylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1-hydroxyethylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxyethylidene]amino]-1-hydroxypropylidene]amino]-1,3-dihydroxypropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxybutylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1-hydroxypropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxyethylidene]amino]-1,5-dihydroxy-5-iminopentylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxybutylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxyethylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1-hydroxyethylidene]amino]hexanoic acid Chemical compound C[C@@H]([C@@H](C(=N[C@@H](CS)C(=N[C@@H](C)C(=N[C@@H](CO)C(=NCC(=N[C@@H](CCC(=N)O)C(=NC(CS)C(=N[C@H]([C@H](C)O)C(=N[C@H](CS)C(=N[C@H](CO)C(=NCC(=N[C@H](CS)C(=NCC(=N[C@H](CCCCN)C(=O)O)O)O)O)O)O)O)O)O)O)O)O)O)O)N=C([C@H](CS)N=C([C@H](CO)N=C([C@H](CO)N=C([C@H](C)N=C(CN=C([C@H](CO)N=C([C@H](CS)N=C(CN=C(C(CS)N=C(C(CC(=O)O)N=C(CN)O)O)O)O)O)O)O)O)O)O)O)O DIGQNXIGRZPYDK-WKSCXVIASA-N 0.000 description 1
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 description 1
- FYGDTMLNYKFZSV-URKRLVJHSA-N (2s,3r,4s,5s,6r)-2-[(2r,4r,5r,6s)-4,5-dihydroxy-2-(hydroxymethyl)-6-[(2r,4r,5r,6s)-4,5,6-trihydroxy-2-(hydroxymethyl)oxan-3-yl]oxyoxan-3-yl]oxy-6-(hydroxymethyl)oxane-3,4,5-triol Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1OC1[C@@H](CO)O[C@@H](OC2[C@H](O[C@H](O)[C@H](O)[C@H]2O)CO)[C@H](O)[C@H]1O FYGDTMLNYKFZSV-URKRLVJHSA-N 0.000 description 1
- 229940015975 1,2-hexanediol Drugs 0.000 description 1
- 229940031723 1,2-octanediol Drugs 0.000 description 1
- 241001116389 Aloe Species 0.000 description 1
- 244000144927 Aloe barbadensis Species 0.000 description 1
- 235000002961 Aloe barbadensis Nutrition 0.000 description 1
- 206010003694 Atrophy Diseases 0.000 description 1
- 229920002498 Beta-glucan Polymers 0.000 description 1
- 102000012410 DNA Ligases Human genes 0.000 description 1
- 108010061982 DNA Ligases Proteins 0.000 description 1
- 101150084418 EGF gene Proteins 0.000 description 1
- 208000002197 Ehlers-Danlos syndrome Diseases 0.000 description 1
- 241000672609 Escherichia coli BL21 Species 0.000 description 1
- 241000620209 Escherichia coli DH5[alpha] Species 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 101000757236 Homo sapiens Angiogenin Proteins 0.000 description 1
- 101000851176 Homo sapiens Pro-epidermal growth factor Proteins 0.000 description 1
- 102000011782 Keratins Human genes 0.000 description 1
- 108010076876 Keratins Proteins 0.000 description 1
- 241000186660 Lactobacillus Species 0.000 description 1
- 102000003792 Metallothionein Human genes 0.000 description 1
- 108090000157 Metallothionein Proteins 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 1
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 1
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 1
- 206010072170 Skin wound Diseases 0.000 description 1
- 102000019197 Superoxide Dismutase Human genes 0.000 description 1
- 108010012715 Superoxide dismutase Proteins 0.000 description 1
- 102400001320 Transforming growth factor alpha Human genes 0.000 description 1
- 101800004564 Transforming growth factor alpha Proteins 0.000 description 1
- 240000004922 Vigna radiata Species 0.000 description 1
- 235000010721 Vigna radiata var radiata Nutrition 0.000 description 1
- 235000011469 Vigna radiata var sublobata Nutrition 0.000 description 1
- MIJPAVRNWPDMOR-UHFFFAOYSA-N [2-(1,2-dihydroxyethyl)-3-hydroxy-5-oxo-2h-furan-4-yl] dihydrogen phosphate Chemical compound OCC(O)C1OC(=O)C(OP(O)(O)=O)=C1O MIJPAVRNWPDMOR-UHFFFAOYSA-N 0.000 description 1
- 210000000577 adipose tissue Anatomy 0.000 description 1
- 125000000539 amino acid group Chemical group 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 229960000271 arbutin Drugs 0.000 description 1
- 230000037444 atrophy Effects 0.000 description 1
- 210000002469 basement membrane Anatomy 0.000 description 1
- 230000003796 beauty Effects 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000017531 blood circulation Effects 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 210000001185 bone marrow Anatomy 0.000 description 1
- OWBTYPJTUOEWEK-UHFFFAOYSA-N butane-2,3-diol Chemical compound CC(O)C(C)O OWBTYPJTUOEWEK-UHFFFAOYSA-N 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000007850 degeneration Effects 0.000 description 1
- 210000004443 dendritic cell Anatomy 0.000 description 1
- 230000029087 digestion Effects 0.000 description 1
- 210000003981 ectoderm Anatomy 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 230000001804 emulsifying effect Effects 0.000 description 1
- 210000000981 epithelium Anatomy 0.000 description 1
- 235000020774 essential nutrients Nutrition 0.000 description 1
- 229960001617 ethyl hydroxybenzoate Drugs 0.000 description 1
- 235000010228 ethyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004403 ethyl p-hydroxybenzoate Substances 0.000 description 1
- NUVBSKCKDOMJSU-UHFFFAOYSA-N ethylparaben Chemical compound CCOC(=O)C1=CC=C(O)C=C1 NUVBSKCKDOMJSU-UHFFFAOYSA-N 0.000 description 1
- 210000000744 eyelid Anatomy 0.000 description 1
- 239000003925 fat Substances 0.000 description 1
- 238000000855 fermentation Methods 0.000 description 1
- 230000004151 fermentation Effects 0.000 description 1
- 235000021323 fish oil Nutrition 0.000 description 1
- 210000003953 foreskin Anatomy 0.000 description 1
- 230000027119 gastric acid secretion Effects 0.000 description 1
- 238000010353 genetic engineering Methods 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- ZFGMDIBRIDKWMY-PASTXAENSA-N heparin Chemical compound CC(O)=N[C@@H]1[C@@H](O)[C@H](O)[C@@H](COS(O)(=O)=O)O[C@@H]1O[C@@H]1[C@@H](C(O)=O)O[C@@H](O[C@H]2[C@@H]([C@@H](OS(O)(=O)=O)[C@@H](O[C@@H]3[C@@H](OC(O)[C@H](OS(O)(=O)=O)[C@H]3O)C(O)=O)O[C@@H]2O)CS(O)(=O)=O)[C@H](O)[C@H]1O ZFGMDIBRIDKWMY-PASTXAENSA-N 0.000 description 1
- FHKSXSQHXQEMOK-UHFFFAOYSA-N hexane-1,2-diol Chemical compound CCCCC(O)CO FHKSXSQHXQEMOK-UHFFFAOYSA-N 0.000 description 1
- 210000005260 human cell Anatomy 0.000 description 1
- 229920002674 hyaluronan Polymers 0.000 description 1
- 229960003160 hyaluronic acid Drugs 0.000 description 1
- GOMNOOKGLZYEJT-UHFFFAOYSA-N isoflavone Chemical compound C=1OC2=CC=CC=C2C(=O)C=1C1=CC=CC=C1 GOMNOOKGLZYEJT-UHFFFAOYSA-N 0.000 description 1
- CJWQYWQDLBZGPD-UHFFFAOYSA-N isoflavone Natural products C1=C(OC)C(OC)=CC(OC)=C1C1=COC2=C(C=CC(C)(C)O3)C3=C(OC)C=C2C1=O CJWQYWQDLBZGPD-UHFFFAOYSA-N 0.000 description 1
- 235000008696 isoflavones Nutrition 0.000 description 1
- 229940039696 lactobacillus Drugs 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 239000003055 low molecular weight heparin Substances 0.000 description 1
- 229940127215 low-molecular weight heparin Drugs 0.000 description 1
- 239000002932 luster Substances 0.000 description 1
- 210000002751 lymph Anatomy 0.000 description 1
- 210000001161 mammalian embryo Anatomy 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 210000004379 membrane Anatomy 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 210000002901 mesenchymal stem cell Anatomy 0.000 description 1
- 210000003716 mesoderm Anatomy 0.000 description 1
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004292 methyl p-hydroxybenzoate Substances 0.000 description 1
- 229960002216 methylparaben Drugs 0.000 description 1
- 230000003020 moisturizing effect Effects 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 231100000957 no side effect Toxicity 0.000 description 1
- AEIJTFQOBWATKX-UHFFFAOYSA-N octane-1,2-diol Chemical compound CCCCCCC(O)CO AEIJTFQOBWATKX-UHFFFAOYSA-N 0.000 description 1
- BJRNKVDFDLYUGJ-UHFFFAOYSA-N p-hydroxyphenyl beta-D-alloside Natural products OC1C(O)C(O)C(CO)OC1OC1=CC=C(O)C=C1 BJRNKVDFDLYUGJ-UHFFFAOYSA-N 0.000 description 1
- 230000008832 photodamage Effects 0.000 description 1
- 230000035790 physiological processes and functions Effects 0.000 description 1
- 239000000419 plant extract Substances 0.000 description 1
- 229920001184 polypeptide Polymers 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 102000004196 processed proteins & peptides Human genes 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- 230000002062 proliferating effect Effects 0.000 description 1
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004405 propyl p-hydroxybenzoate Substances 0.000 description 1
- 229960003415 propylparaben Drugs 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 230000008439 repair process Effects 0.000 description 1
- 238000010839 reverse transcription Methods 0.000 description 1
- 238000007665 sagging Methods 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 210000004761 scalp Anatomy 0.000 description 1
- 210000002374 sebum Anatomy 0.000 description 1
- 230000036548 skin texture Effects 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 210000000434 stratum corneum Anatomy 0.000 description 1
- 210000004003 subcutaneous fat Anatomy 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 210000004243 sweat Anatomy 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- XPFJYKARVSSRHE-UHFFFAOYSA-K trisodium;2-hydroxypropane-1,2,3-tricarboxylate;2-hydroxypropane-1,2,3-tricarboxylic acid Chemical compound [Na+].[Na+].[Na+].OC(=O)CC(O)(C(O)=O)CC(O)=O.[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O XPFJYKARVSSRHE-UHFFFAOYSA-K 0.000 description 1
- 230000005740 tumor formation Effects 0.000 description 1
- 210000003954 umbilical cord Anatomy 0.000 description 1
- 238000011144 upstream manufacturing Methods 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 230000029663 wound healing Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/84—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions otherwise than those involving only carbon-carbon unsaturated bonds
- A61K8/85—Polyesters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/64—Proteins; Peptides; Derivatives or degradation products thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/08—Anti-ageing preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/41—Particular ingredients further characterized by their size
- A61K2800/412—Microsized, i.e. having sizes between 0.1 and 100 microns
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Birds (AREA)
- Epidemiology (AREA)
- Gerontology & Geriatric Medicine (AREA)
- Dermatology (AREA)
- Cosmetics (AREA)
Abstract
The invention relates to a cosmetic composition containing epidermal growth factor and a preparation method thereof. The cosmetic composition of the present invention comprises epidermal growth factor, nonionic surfactant, polylactic acid, pH regulator, preservative, matrix, etc. According to the cosmetic composition of the present invention, the epidermal growth factor is uniformly distributed in the composition, the activity is not reduced during long-term storage, the biological efficacy of the epidermal growth factor is prolonged, and the composition has improved proliferation promoting ability for human dermal fibroblasts, thereby improving aging and achieving a cosmetic effect.
Description
Technical Field
The invention belongs to the field of daily chemical products, and particularly relates to a cosmetic composition containing an epidermal growth factor and a preparation method thereof.
Background
The skin is subdivided into superficial, thin epidermis, more fibrous dermis and subcutaneous tissue. The epidermis is differentiated from ectoderm covering the surface of the embryo, and is composed of multiple layers of squamous epithelia, and the thicknesses of the layers vary from thick scalp to thinnest eyelid skin throughout the body. The epidermis can be subdivided into keratin cells, dendritic cells and Michael cells; the dermis, developed primarily from the mesoderm, underlies the epidermis and is separated from the epidermal quality by a basement membrane. The dermis is composed primarily of connective tissue, including blood vessels, nerves, muscles, lymph, sweat glands and sebaceous glands, and is called the papillary layer, superficial, thin, and the reticular layer, deep and thick. The main component of the dermis is collagen, the content of collagen fibers in the papillary layer is small, the collagen fibers are arranged irregularly, but the collagen fibers are arranged in a bundle shape and parallel to the surface in the network layer; the subcutaneous tissue, or lipid membrane, is the superficial fascia of the body surface, which is composed of loose connective tissue and adipose tissue. The dermis extends a plurality of collagen fiber bundles with different sizes into the subcutaneous tissue, so that the skin is firmly connected with the subcutaneous tissue, and no obvious limit exists between the two.
Skin aging is a phenomenon of skin aging caused by natural factors or non-natural factors. After birth, the skin tissue of a person is developed increasingly, the function of the person is active gradually, and the person starts to degenerate when the person reaches a certain age, and the degeneration is gradually performed unconsciously. The growth period of skin tissues generally ends at about 25 years old, which is called as 'skin bend angle' by some people, since the growth and aging of the skin tissues are carried out simultaneously, the skin elastic fibers become thicker gradually, and the skin aging is slowly and obviously in the early aging period of 40-50 years, but the aging degree varies from person to person.
The skin aging phenomenon is mainly manifested in two aspects:
1. the skin tissue deteriorates. The thickness of the skin changes significantly with age. Human epidermis is thickest at age of 20 years, and gradually thins later, and by the age, the granular layer can shrink to disappear, and the survival time of spine cells is shortened. The division of the epidermis nucleus is increased, so the melanin is also increased, so the color of the skin of the old is brownish black. The aged cells adhere to the stratum corneum of the epidermis, which hardens the skin surface and loses its luster. The dermis is thickest at age 30, and becomes thinner and more complicated with atrophy later. The subcutaneous fat is reduced and the elasticity and tension of the skin are gradually lost due to the changes of the elastic fiber and the collagen fiber, further resulting in the skin relaxation and the generation of wrinkles.
2. The physiological function is low. Sebaceous glands and sweat glands are in failure, sweat and sebum are eliminated less, and skin gradually loses sunglow and becomes dry. The blood circulation function is degraded to supplement essential nutrients of the skin, and thus the skin wound of the elderly is difficult to heal.
The cell growth factor is an early hematopoietic stimulating factor which can play a role in the bone marrow hematopoietic microenvironment and has various biological activities. Including epidermal growth factor, and small molecule mitogen containing 3 disulfide bonds. And is involved in the processes of normal cell growth, tumor formation, wound healing and the like. The sequence and function of which are homologous to TGF-alpha. Polypeptides capable of stimulating epidermal and epithelial tissue proliferation are mitogens. And can inhibit gastric acid secretion. Is composed of 53 amino acid residues, and the mass of the precursor molecule thereof is more than 100 kDa.
The prior art discloses cosmetic compositions containing epidermal growth factor. CN101816620A discloses a whitening cosmetic and a preparation method and application thereof. The cosmetic comprises the following components in percentage by mass: 0.00001-0.001% of recombinant human angiogenin, 0.00001-0.001% of metallothionein, 0.00001-0.001% of human epidermal growth factor, 0.00001-0.001% of IL-1RA, 0.1-0.5% of superoxide dismutase, 0.1-0.5% of low molecular weight heparin sodium, 1-5% of arbutin, 5-25% of stabilizer, 0.02-0.3% of hyaluronic acid, 0.5-5% of methyl propylene glycol, 0.5-3.5% of lactobacillus/mung bean fermentation liquor, 0.5-5% of dissolving protease, 0.5-8% of vitamin C phosphate sodium, 0.3-0.7% of 1, 2 hexanediol or 1, 2 octanediol, 1-5% of oat beta glucan, 1-5% of soybean isoflavone and the balance of water. The whitening cosmetic can be stored for a long time, has no side effect on skin, and is suitable for long-term use.
CN113813192A discloses a cosmetic for assisting skin repair and a preparation method thereof, wherein the cosmetic comprises the following components in percentage by mass: 0.06-0.12% of recombinant human epidermal growth factor, 2.5-5.0% of aloe vera gel, 0.1-0.2% of low molecular weight sodium hyaluronate with an average molecular weight of 3-10 kDa, 0.2-0.4% of kelp extract and 94.44-96.98% of base material; the recombinant human epidermal growth factor, the aloe gel and the kelp extract have synergistic effect on skin moisturizing and repairing, trace recombinant human epidermal growth factor can be utilized to the maximum extent, and the product can efficiently help skin repairing.
CN109674682A discloses a cosmetic preparation rich in cell growth factors, which comprises the following components: 0.01-0.2% of STP, 0.01-0.1% of cell growth factor, 1-10% of fish oil, 1-10% of surfactant, 1-15% of humectant, 0.1-1% of antioxidant, 0.1-3% of pH regulator, 3-5% of emulsifier, 0.1-5% of natural plant extract and deionized water; the patent proposes a cosmetic preparation rich in cell growth factors, which can continuously replace aged cells with new cells, thus generating anti-wrinkle and anti-wrinkle effects through the renewal of the skin; can prevent skin wrinkles, dryness, sagging, aging, photodamage, or improve skin texture and smoothness; can enhance the emulsifying effect of cosmetic and improve the stability of cosmetic.
Disclosure of Invention
The invention aims to overcome the defects of the prior art and provide a cosmetic composition containing an epidermal growth factor and a preparation method thereof.
The technical scheme of the invention is as follows:
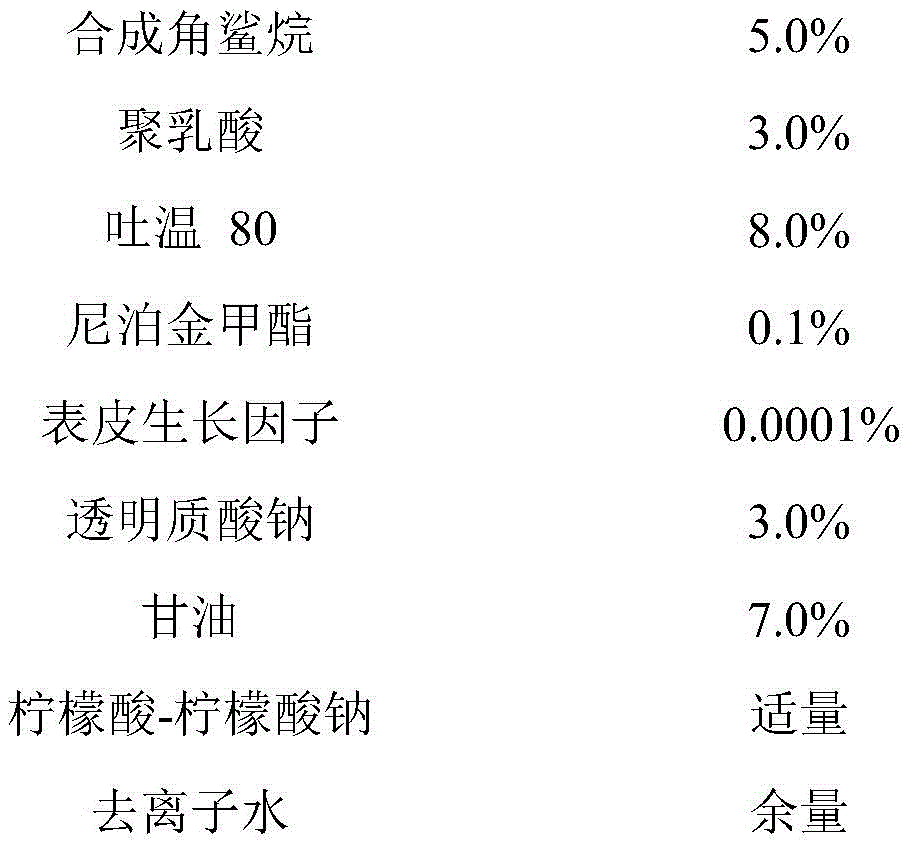
a cosmetic composition containing human epidermal growth factor comprises the following components by mass percent:
the wt% is based on the total weight of the cosmetic composition.
In a preferred embodiment, the cosmetic composition comprises:
in another preferred embodiment, the cosmetic composition comprises:
the purity of the epidermal growth factor can be 85-99%. The epidermal growth factor can be a recombinant human epidermal growth factor prepared by using a genetic engineering method or a commercially available human epidermal growth factor product.
The nonionic surfactant may be selected from one or more of polyoxyethylene oleyl ether, polyoxyethylene stearyl ether, tween 20, tween 40, tween 60, tween 80, polyoxyethylene monostearate, polyoxyethylene glycerol triisostearate, polyoxyethylene hydrogenated castor oil, stearyl stearate, polyoxyethylene lauryl ether stearate, ethylene glycol monostearate, polyoxyethylene triolein, and the like.
The particle size of the polylactic acid is preferably 0.05-1 micron, and more preferably 0.1-0.4 micron. Commercially available polylactic acid products for pharmaceutical or cosmetic use can be used.
The humectant is preferably sodium hyaluronate.
The oil is one or more of squalane, lanonol, cetearyl alcohol and polydimethylsiloxane.
The pH regulator may be citric acid-sodium citrate.
The preservative can be one or more of methyl paraben, ethyl paraben and propyl paraben.
The matrix may comprise water and/or a polyol, the mass ratio of water to polyol may be any ratio, such as 1:0.1-10, further such as 1:0.5-5, depending on the specific product, the polyol may be selected from one or more of ethylene glycol, diethylene glycol, dipropylene glycol, polyethylene glycol, polypropylene glycol, 1, 3-butanediol, glycerol. Preferably, glycerol and/or water are used.
Preferably, at least one of a grease and a humectant is present.
The cosmetics can comprise skin care products, anti-wrinkle products, cleaning cosmetics, functional cosmetics and beauty cosmetics; the cleaning cosmetics comprise cleaning cream, face washing lotion, bath lotion and shaving products.
The present invention further provides a method for preparing the above cosmetic composition, which comprises:
(1) mixing nonionic surfactant, oil and fat, humectant, and matrix under heating (such as at 60-100 deg.C, preferably 65-85 deg.C, preferably 70-80 deg.C);
(2) cooling to 37-50 deg.C, preferably 37-45 deg.C, adding polylactic acid powder and epidermal growth factor or previously mixed polylactic acid powder and epidermal growth factor, pH regulator and antiseptic, mixing, and vacuum degassing.
THE ADVANTAGES OF THE PRESENT INVENTION
According to the cosmetic composition of the present invention, the epidermal growth factor is uniformly distributed in the composition, the activity is not reduced during long-term storage, the biological efficacy of the epidermal growth factor is prolonged, and the composition has improved proliferation-promoting ability to human dermal fibroblasts, thereby improving aging and achieving an improved cosmetic effect.
Drawings
FIG. 1 is a graph showing the effect of epidermal growth factor on human dermal fibroblasts.
Detailed Description
The invention is further illustrated by the following specific examples. It is further noted that, in the present application,% by weight is unless otherwise specified.
Example 1
Human epidermal growth factor acquisition
Extracting total RNA from human umbilical cord mesenchymal stem cells, and performing reverse transcription to obtain cDNA. And designing a PCR primer according to the full-length gene sequence of the human EGF. EGF upstream primer sequence: 5'-CAAGAGTCCAGCTTAATGCAGGCAG-3', downstream primer sequence: 5'-GCTAGAAGGGGAACTCGCAGACGTA-3' are provided. The EGF gene was obtained by PCR using the cDNA obtained above as a template. EGF and pET-28a plasmids are subjected to double digestion by Nde and BamH1, T4 DNA ligase is used for connection, the connection object is transformed into competent Escherichia coli DH5 alpha, and the recombinant plasmid pET-28a is transformed into Escherichia coli BL21 for expression after being successfully constructed. The expressed recombinant human cell growth factor is purified by a nickel column to obtain the target recombinant protein with the purity of about 95 percent.
Example 2
A skin care product, comprising:
heating synthetic squalane, Tween 20, sodium hyaluronate, glycerol and deionized water to 70 deg.C, mixing thoroughly, cooling to about 40 deg.C, adding epidermal growth factor into polylactic acid powder (particle size 0.05-1 micrometer), mixing, adding into the mixture of synthetic squalane, Tween 20, sodium hyaluronate, glycerol and deionized water together with pH regulator and antiseptic, mixing well, and vacuum degassing.
Example 3
A skin care product, comprising:
heating synthetic squalane, Tween 20, sodium hyaluronate, glycerol and deionized water to 70 deg.C, mixing thoroughly, cooling to about 40 deg.C, adding epidermal growth factor into polylactic acid powder (particle size 0.05-1 micrometer), mixing, adding into the mixture of synthetic squalane, Tween 20, sodium hyaluronate, glycerol and deionized water together with pH regulator and antiseptic, mixing well, and vacuum degassing.
Example 4
A skin care product, comprising:
heating synthetic squalane, Tween 20, sodium hyaluronate, glycerol and deionized water to 70 deg.C, mixing thoroughly, cooling to about 40 deg.C, adding epidermal growth factor into polylactic acid powder (particle size 0.05-1 micrometer), mixing, adding into the mixture of synthetic squalane, Tween 20, sodium hyaluronate, glycerol and deionized water together with pH regulator and antiseptic, mixing well, and vacuum degassing.
Comparative example 1
A skin care product, comprising:
heating synthetic squalane, Tween 20, sodium hyaluronate, glycerol and deionized water to 70 deg.C, mixing, cooling to about 40 deg.C, adding epidermal growth factor, pH regulator and antiseptic, mixing, and vacuum degassing.
Effect of cosmetic on human dermal fibroblasts were tested, isolated from human foreskin cells in age 16, to 80-90% confluence, digested, and 5 x 104The cells were densely plated in 96-well plates at a concentration of 100. mu.l/well, and after 12 hours, 10. mu.l of the cosmetic composition was added to separate the experiment into example 2, example 4 and comparative example 1. Each group is provided with 4 holes. 37 ℃ and 5% CO2And incubating in a saturated humidity incubator for 48 hours, adding MTT, continuously incubating for 4 hours, adding DMSO, measuring the absorbance value at the wavelength of 570nm, and repeating the experiment for three times.
The results showed that the absorbance values of the example 2 group and the example 4 group were significantly increased and significantly different from those of the comparative example 1 group, as shown in fig. 1. The results in fig. 1 show that, in a certain concentration range, the examples of the present invention have significantly improved proliferation-promoting ability to human dermal fibroblasts, compared to the comparative example 1 group.
Examples 2 and 4 of the present invention the proliferative capacity of the cosmetic composition to fibroblasts remained substantially unchanged after half a year of storage.
The above embodiments are preferred embodiments of the present invention, but the present invention is not limited to the above embodiments, and any other changes, modifications, substitutions, combinations, and simplifications which do not depart from the spirit and principle of the present invention should be construed as equivalents thereof, and all such changes, modifications, substitutions, combinations, and simplifications are intended to be included in the scope of the present invention.
Claims (10)
4. the cosmetic composition according to any one of claims 1 to 3, wherein the nonionic surfactant is selected from one or more of polyoxyethylene oleyl ether, polyoxyethylene stearyl ether, Tween 20, Tween 40, Tween 60, Tween 80, polyoxyethylene monostearate, polyoxyethylene glycerol triisostearate, polyoxyethylene hydrogenated castor oil, stearyl stearate, polyoxyethylene lauryl ether stearate, ethylene glycol monostearate, and polyoxyethylene glyceryl trioleate.
5. Cosmetic composition according to any one of claims 1 to 3, characterized in that the particle size of the polylactic acid is between 0.05 and 1 micron, preferably between 0.1 and 0.4 micron.
6. The cosmetic composition of any one of claims 1 to 3, wherein the humectant is sodium hyaluronate.
7. The cosmetic composition according to any one of claims 1 to 3, wherein the oil or fat is one or more selected from squalane, lanolin alcohol, cetearyl alcohol, and polydimethylsiloxane.
8. Cosmetic composition according to any one of claims 1 to 3, characterized in that the base comprises water and/or a polyol, the mass ratio of water to polyol being any ratio, such as 1:0.1 to 10, further such as 1:0.5 to 5,
preferably, the polyhydric alcohol is one or more selected from ethylene glycol, diethylene glycol, dipropylene glycol, polyethylene glycol, polypropylene glycol, 1, 3-butylene glycol, and glycerin.
9. The cosmetic composition according to any one of claims 1 to 3, wherein at least one of an oil and a humectant is present.
10. A method of preparing a cosmetic composition according to any one of claims 1 to 9, comprising:
(1) heating and mixing the nonionic surfactant, the grease, the humectant and the matrix;
(2) cooling to 37-50 deg.C, preferably 37-45 deg.C, adding polylactic acid powder and epidermal growth factor or previously mixed polylactic acid powder and epidermal growth factor, pH regulator and antiseptic, mixing, and vacuum degassing.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111678259.8A CN114146021A (en) | 2021-12-31 | 2021-12-31 | Cosmetic composition containing epidermal growth factor and preparation method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111678259.8A CN114146021A (en) | 2021-12-31 | 2021-12-31 | Cosmetic composition containing epidermal growth factor and preparation method thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN114146021A true CN114146021A (en) | 2022-03-08 |
Family
ID=80449864
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202111678259.8A Pending CN114146021A (en) | 2021-12-31 | 2021-12-31 | Cosmetic composition containing epidermal growth factor and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN114146021A (en) |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1720989A (en) * | 2004-07-15 | 2006-01-18 | 深圳市清华源兴生物医药科技有限公司 | Sustained release microsphere of epidermal growth factor, its preparation method and application |
| US20070104654A1 (en) * | 2005-11-08 | 2007-05-10 | Industrial Technology Research Institute | Amphiphilic block copolymers and nano particles comprising the same |
| CN101400327A (en) * | 2006-03-13 | 2009-04-01 | 凯塞拉生物药品公司 | Fluidic tissue augmentation compositions and methods |
| US20090232855A1 (en) * | 2001-04-13 | 2009-09-17 | Sun Sang Kwon | Percutaneous controlled releasing material using nano-sized polymer particles and external application agent containing the same |
| CN104784065A (en) * | 2015-03-20 | 2015-07-22 | 金正容 | Beautifying method |
| CN108524323A (en) * | 2018-04-18 | 2018-09-14 | 广州赛莱拉干细胞科技股份有限公司 | Toner containing epithelical cell growth factor |
| CN110141533A (en) * | 2019-06-25 | 2019-08-20 | 吉林省华恩生物科技有限公司 | One kind going wrinkle Firm liquid and preparation method |
-
2021
- 2021-12-31 CN CN202111678259.8A patent/CN114146021A/en active Pending
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20090232855A1 (en) * | 2001-04-13 | 2009-09-17 | Sun Sang Kwon | Percutaneous controlled releasing material using nano-sized polymer particles and external application agent containing the same |
| CN1720989A (en) * | 2004-07-15 | 2006-01-18 | 深圳市清华源兴生物医药科技有限公司 | Sustained release microsphere of epidermal growth factor, its preparation method and application |
| US20070104654A1 (en) * | 2005-11-08 | 2007-05-10 | Industrial Technology Research Institute | Amphiphilic block copolymers and nano particles comprising the same |
| CN101400327A (en) * | 2006-03-13 | 2009-04-01 | 凯塞拉生物药品公司 | Fluidic tissue augmentation compositions and methods |
| CN104784065A (en) * | 2015-03-20 | 2015-07-22 | 金正容 | Beautifying method |
| CN108524323A (en) * | 2018-04-18 | 2018-09-14 | 广州赛莱拉干细胞科技股份有限公司 | Toner containing epithelical cell growth factor |
| CN110141533A (en) * | 2019-06-25 | 2019-08-20 | 吉林省华恩生物科技有限公司 | One kind going wrinkle Firm liquid and preparation method |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102265730B1 (en) | cosmetic composition for skin wrinkle improvement and whitening facial skin | |
| CN115137665B (en) | A composition with long-lasting moisturizing and soothing effects and its application | |
| JP2011519353A (en) | Active ingredients that stimulate fibroblast proliferation and / or activity | |
| JP2005534691A (en) | Cosmetic or dermopharmaceutical composition comprising kombucha | |
| CN113397998A (en) | Application of recombinant humanized collagen in preparation of anti-skin-aging products | |
| CN113663051A (en) | Composition of skipjack elastin peptide and collagen oligopeptide and preparation method thereof | |
| JP7329588B2 (en) | A color cosmetic composition containing a color base reacted with a low-temperature, high-pressure enzyme and an extract | |
| CN113786379A (en) | Lactic acid bacteria whitening and repairing essence and preparation method thereof | |
| KR20190010795A (en) | Moisturizing, anti-wrinkling, and whitening cosmetic composition comprising novel Hyalpol Matrix Mixture and preparation method of the same | |
| CN108030721A (en) | A kind of Essence and preparation method thereof | |
| CN114948803A (en) | Anti-aging essence and preparation method thereof | |
| CN110882177A (en) | Skin repair oligopeptide composition and preparation method thereof | |
| CN114557909A (en) | Multiple peptide anti-wrinkle essence and preparation method thereof | |
| CN111588650B (en) | Repairing composition, repairing mask liquid and repairing mask | |
| CN114146021A (en) | Cosmetic composition containing epidermal growth factor and preparation method thereof | |
| CN112675063A (en) | Skin-tendering anti-wrinkle composition containing PDRN and application thereof | |
| CN114272184A (en) | Polypeptide composition, anti-aging repair eye cream containing polypeptide composition and preparation method of anti-aging repair eye cream | |
| CN113730289B (en) | An eye and lip care composition with multiple repair effects and its application | |
| KR20070022170A (en) | Skin cosmetic composition containing enzyme and amino acid | |
| CN114129490A (en) | Sea-buckthorn collagen anti-aging moisturizing mask and preparation method thereof | |
| KR100847299B1 (en) | Silk peptide fraction with excellent moisturizing ability and use thereof | |
| CN109662938B (en) | Plant polysaccharide moisturizing factor and preparation method and application thereof | |
| CN107854344A (en) | A kind of Essence and preparation method thereof | |
| CN105213294A (en) | There is the skin care compositions of skin effect of compacting | |
| CN112451469A (en) | Stem cell double-layer skin care essence and preparation method thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination |