CN114097783A - Peracetic acid disinfectant and preparation method thereof - Google Patents
Peracetic acid disinfectant and preparation method thereof Download PDFInfo
- Publication number
- CN114097783A CN114097783A CN202210092725.2A CN202210092725A CN114097783A CN 114097783 A CN114097783 A CN 114097783A CN 202210092725 A CN202210092725 A CN 202210092725A CN 114097783 A CN114097783 A CN 114097783A
- Authority
- CN
- China
- Prior art keywords
- acid
- disinfectant
- peroxyacetic
- liquid surfactant
- peroxyacetic acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- KFSLWBXXFJQRDL-UHFFFAOYSA-N Peracetic acid Chemical compound CC(=O)OO KFSLWBXXFJQRDL-UHFFFAOYSA-N 0.000 title claims abstract description 114
- 239000000645 desinfectant Substances 0.000 title claims abstract description 33
- 238000002360 preparation method Methods 0.000 title claims abstract description 13
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims abstract description 55
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 claims abstract description 24
- 239000004094 surface-active agent Substances 0.000 claims abstract description 24
- 239000002608 ionic liquid Substances 0.000 claims abstract description 21
- 239000003381 stabilizer Substances 0.000 claims abstract description 18
- -1 carbon chain organic acid Chemical class 0.000 claims abstract description 12
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 10
- 239000002781 deodorant agent Substances 0.000 claims abstract description 7
- 239000002994 raw material Substances 0.000 claims abstract description 4
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 claims description 8
- 238000003756 stirring Methods 0.000 claims description 7
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 claims description 6
- 238000002156 mixing Methods 0.000 claims description 5
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 claims description 4
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical compound N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 claims description 4
- 239000003795 chemical substances by application Substances 0.000 claims description 4
- FBUKVWPVBMHYJY-UHFFFAOYSA-N nonanoic acid Chemical compound CCCCCCCCC(O)=O FBUKVWPVBMHYJY-UHFFFAOYSA-N 0.000 claims description 4
- WWZKQHOCKIZLMA-UHFFFAOYSA-N octanoic acid Chemical compound CCCCCCCC(O)=O WWZKQHOCKIZLMA-UHFFFAOYSA-N 0.000 claims description 4
- JDWPDZLCSRBZPY-SQGDDOFFSA-N C(=O)(O)C(C(=O)O)N[C@@H](C)C(=O)O.[Na].[Na].[Na] Chemical compound C(=O)(O)C(C(=O)O)N[C@@H](C)C(=O)O.[Na].[Na].[Na] JDWPDZLCSRBZPY-SQGDDOFFSA-N 0.000 claims description 3
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 claims description 3
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 claims description 3
- 125000002091 cationic group Chemical group 0.000 claims description 3
- 239000007788 liquid Substances 0.000 claims description 3
- GYSCBCSGKXNZRH-UHFFFAOYSA-N 1-benzothiophene-2-carboxamide Chemical compound C1=CC=C2SC(C(=O)N)=CC2=C1 GYSCBCSGKXNZRH-UHFFFAOYSA-N 0.000 claims description 2
- AWQSAIIDOMEEOD-UHFFFAOYSA-N 5,5-Dimethyl-4-(3-oxobutyl)dihydro-2(3H)-furanone Chemical compound CC(=O)CCC1CC(=O)OC1(C)C AWQSAIIDOMEEOD-UHFFFAOYSA-N 0.000 claims description 2
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 claims description 2
- GHVNFZFCNZKVNT-UHFFFAOYSA-N Decanoic acid Natural products CCCCCCCCCC(O)=O GHVNFZFCNZKVNT-UHFFFAOYSA-N 0.000 claims description 2
- OBETXYAYXDNJHR-UHFFFAOYSA-N alpha-ethylcaproic acid Natural products CCCCC(CC)C(O)=O OBETXYAYXDNJHR-UHFFFAOYSA-N 0.000 claims description 2
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 claims description 2
- YKYONYBAUNKHLG-UHFFFAOYSA-N n-Propyl acetate Natural products CCCOC(C)=O YKYONYBAUNKHLG-UHFFFAOYSA-N 0.000 claims description 2
- 229940090181 propyl acetate Drugs 0.000 claims description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 claims description 2
- 230000001502 supplementing effect Effects 0.000 claims description 2
- 229940061605 tetrasodium glutamate diacetate Drugs 0.000 claims description 2
- UZVUJVFQFNHRSY-OUTKXMMCSA-J tetrasodium;(2s)-2-[bis(carboxylatomethyl)amino]pentanedioate Chemical compound [Na+].[Na+].[Na+].[Na+].[O-]C(=O)CC[C@@H](C([O-])=O)N(CC([O-])=O)CC([O-])=O UZVUJVFQFNHRSY-OUTKXMMCSA-J 0.000 claims description 2
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 claims description 2
- MLDWGLQSBBCMMO-UHFFFAOYSA-N [Na].[Na].[Na].CNCC(=O)O Chemical compound [Na].[Na].[Na].CNCC(=O)O MLDWGLQSBBCMMO-UHFFFAOYSA-N 0.000 claims 1
- 238000004519 manufacturing process Methods 0.000 claims 1
- 239000002184 metal Substances 0.000 abstract description 19
- 229910052751 metal Inorganic materials 0.000 abstract description 19
- 230000000694 effects Effects 0.000 abstract description 14
- 238000004659 sterilization and disinfection Methods 0.000 abstract description 10
- 230000001954 sterilising effect Effects 0.000 abstract description 9
- 150000002739 metals Chemical class 0.000 abstract description 3
- 229960000583 acetic acid Drugs 0.000 description 15
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 9
- 244000005700 microbiome Species 0.000 description 8
- SCKXCAADGDQQCS-UHFFFAOYSA-N Performic acid Chemical compound OOC=O SCKXCAADGDQQCS-UHFFFAOYSA-N 0.000 description 6
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 6
- 210000000170 cell membrane Anatomy 0.000 description 6
- 230000002147 killing effect Effects 0.000 description 5
- 239000000047 product Substances 0.000 description 5
- 230000000052 comparative effect Effects 0.000 description 4
- 238000005260 corrosion Methods 0.000 description 4
- 230000007797 corrosion Effects 0.000 description 4
- 125000004433 nitrogen atom Chemical group N* 0.000 description 4
- 230000035699 permeability Effects 0.000 description 4
- 241000894006 Bacteria Species 0.000 description 3
- 125000005907 alkyl ester group Chemical group 0.000 description 3
- 239000003054 catalyst Substances 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 150000002191 fatty alcohols Chemical class 0.000 description 3
- 230000002209 hydrophobic effect Effects 0.000 description 3
- 230000002195 synergetic effect Effects 0.000 description 3
- 230000009471 action Effects 0.000 description 2
- 230000000844 anti-bacterial effect Effects 0.000 description 2
- 230000001580 bacterial effect Effects 0.000 description 2
- 239000012362 glacial acetic acid Substances 0.000 description 2
- MNWFXJYAOYHMED-UHFFFAOYSA-N heptanoic acid Chemical compound CCCCCCC(O)=O MNWFXJYAOYHMED-UHFFFAOYSA-N 0.000 description 2
- 125000000623 heterocyclic group Chemical group 0.000 description 2
- 229910052739 hydrogen Inorganic materials 0.000 description 2
- 239000001257 hydrogen Substances 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 239000000203 mixture Substances 0.000 description 2
- 150000007523 nucleic acids Chemical group 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- BAERPNBPLZWCES-UHFFFAOYSA-N (2-hydroxy-1-phosphonoethyl)phosphonic acid Chemical compound OCC(P(O)(O)=O)P(O)(O)=O BAERPNBPLZWCES-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 244000063299 Bacillus subtilis Species 0.000 description 1
- 235000014469 Bacillus subtilis Nutrition 0.000 description 1
- 208000035143 Bacterial infection Diseases 0.000 description 1
- 239000007848 Bronsted acid Substances 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 1
- 241000191967 Staphylococcus aureus Species 0.000 description 1
- PDIZYYQQWUOPPK-UHFFFAOYSA-N acetic acid;2-(methylamino)acetic acid Chemical compound CC(O)=O.CC(O)=O.CNCC(O)=O PDIZYYQQWUOPPK-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000000845 anti-microbial effect Effects 0.000 description 1
- 230000002421 anti-septic effect Effects 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 229940064004 antiseptic throat preparations Drugs 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 208000022362 bacterial infectious disease Diseases 0.000 description 1
- 239000003899 bactericide agent Substances 0.000 description 1
- 235000013361 beverage Nutrition 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 210000002421 cell wall Anatomy 0.000 description 1
- 239000013522 chelant Substances 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 239000003205 fragrance Substances 0.000 description 1
- 230000002070 germicidal effect Effects 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 230000007124 immune defense Effects 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 239000007769 metal material Substances 0.000 description 1
- 238000000034 method Methods 0.000 description 1
- 239000000693 micelle Substances 0.000 description 1
- 230000000813 microbial effect Effects 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 238000011056 performance test Methods 0.000 description 1
- 150000002978 peroxides Chemical group 0.000 description 1
- 150000003904 phospholipids Chemical class 0.000 description 1
- 230000035479 physiological effects, processes and functions Effects 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- SOBHUZYZLFQYFK-UHFFFAOYSA-K trisodium;hydroxy-[[phosphonatomethyl(phosphonomethyl)amino]methyl]phosphinate Chemical compound [Na+].[Na+].[Na+].OP(O)(=O)CN(CP(O)([O-])=O)CP([O-])([O-])=O SOBHUZYZLFQYFK-UHFFFAOYSA-K 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/16—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing the group; Thio analogues thereof
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N25/00—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests
- A01N25/22—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests containing ingredients stabilising the active ingredients
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N25/00—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests
- A01N25/30—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests characterised by the surfactants
Landscapes
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- Agronomy & Crop Science (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Toxicology (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
The invention discloses a peroxyacetic acid disinfectant, which comprises the following preparation raw materials in percentage by weight: 10% -30% of hydrogen peroxide; 10% -30% of acetic acid; 1% -5% of medium-long carbon chain organic acid; 1% -5% of ionic liquid surfactant; 0.1% -2% of a stabilizer; 1% -5% of a deodorant; the balance being water. The peroxyacetic acid disinfectant disclosed by the invention has better stability and good sterilization effect, and reduces the corrosivity and pungent smell of metals. The invention also provides a preparation method of the peroxyacetic acid disinfectant.
Description
Technical Field
The invention relates to the technical field of disinfectants, and particularly relates to a peroxyacetic acid disinfectant and a preparation method thereof.
Background
The peroxyacetic acid disinfectant is a high-efficiency disinfectant with strong oxidizing property, has a killing effect on various microorganisms, and is widely applied to the microbial control in the industries of medical treatment, health care and food and beverage processing. At present, the synthesis method of peracetic acid is commonly used, wherein glacial acetic acid is mixed with hydrogen peroxide, and catalysts such as sulfuric acid and the like are added to synthesize the peracetic acid. The peroxyacetic acid products available commercially, whether in single-pack or binary-pack products, are prepared by reacting hydrogen peroxide with glacial acetic acid. Peroxyacetic acid is used as a broad-spectrum bactericide, and has the disadvantages of strong corrosivity to metals, strong pungent smell and the like, so that the use of the peroxyacetic acid is limited to a certain extent. In patent CN103732063A, fatty alcohol is added to cover the pungent smell of acetic acid by reacting with acetic acid in the system to generate alkyl ester with fragrance. However, fatty alcohols and acetic acid are difficult to form alkyl esters under normal conditions, and if one considers the gradual reaction to form alkyl esters during storage, the added fatty alcohol affects the peroxycarboxylic acid stability.
Therefore, it is necessary to develop a peroxyacetic acid disinfectant and a preparation method thereof to solve the above technical drawbacks.
Disclosure of Invention
The invention aims to provide a peracetic acid disinfectant with good sterilization effect and good stability and capable of reducing metal corrosivity and pungent smell and a preparation method thereof.
In order to achieve the purpose, the invention discloses a peroxyacetic acid disinfectant, which comprises the following preparation raw materials in percentage by weight: 10% -30% of hydrogen peroxide; 10% -30% of acetic acid; 1% -5% of medium-long carbon chain organic acid; 1% -5% of ionic liquid surfactant; 0.1% -2% of a stabilizer; 1% -5% of a deodorant; the balance being water.
Preferably, the weight ratio of hydrogen peroxide to acetic acid is 1.2-2: 1.
preferably, the medium-long carbon chain organic acid is selected from one or more of heptanoic acid, octanoic acid, nonanoic acid and decanoic acid.
Preferably, the ionic liquid surfactant is a cationic liquid surfactant, and the cationic liquid surfactant is selected from one or more of imidazole, pyridine and quinoline.
Preferably, the stabilizer is selected from at least one of trisodium dicarboxymethylalanine, tetrasodium glutamate diacetate, and trisodium methylglycinediacetate.
Preferably, the smell removing agent is selected from one or more of methyl acetate, ethyl acetate, propyl acetate and butyl acetate.
The invention also discloses a preparation method of the peroxyacetic acid disinfectant, which comprises the following steps:
(1) mixing and stirring a stabilizer and hydrogen peroxide according to a proportion;
(2) adding medium-long carbon chain organic acid, deodorant and ionic liquid surfactant, mixing and stirring;
(3) adding acetic acid and supplementing water.
Compared with the prior art, the invention has the following advantages:
(1) the ionic liquid surfactant is used as a catalyst, plays a role of Bronsted acid in chemical reaction, catalyzes hydrogen peroxide and acetic acid to react to generate peroxyacetic acid, and improves the content of a peroxyacetic acid product in a system; meanwhile, the ionic liquid surfactant can catalyze the medium-long carbon chain carboxylic acid to be converted into medium-long carbon chain peroxycarboxylic acid; the use of a sulfuric acid catalyst is avoided, thereby avoiding safety problems caused by the release of a large amount of heat due to the addition of sulfuric acid.
(2) Biofilms are one of the leading causes of bacterial disease that is difficult to eradicate. Bacteria in the form of biofilms, unlike planktonic bacteria, are highly resistant to antiseptics such as antibiotics, harsh environments, and host immune defense mechanisms, and have unique properties in terms of physiology, metabolism, degradation or utilization of substrates, and resistance to the environment. The ionic liquid surfactant has good effect of removing the biological membrane, increases the permeability of the bacterial cell membrane, and weakens the growth, adhesion and formation of the biological membrane.
(3) In the process of killing microorganisms, the ionic liquid surfactant attacks the cell plasma membrane of bacteria or the cell membrane of yeast and interacts with phospholipid components in the cell membrane to lose the permeability of the cell membrane, so that the effect of killing the microorganisms is achieved. Meanwhile, the ionic liquid surfactant has a surface activity effect, so that the interfacial tension can be reduced, the contact area of the peroxyacetic acid product and the surface of the harmful microorganism is increased, and the permeability to cell walls and cell membranes of the harmful microorganism is increased, so that the resistance of the peroxyacetic acid entering the interior of the microorganism is reduced, and the killing effect of the peroxyacetic acid on the microorganism is promoted.
(4) The reaction product in the system is a mixture of peroxyacetic acid and medium-long carbon chain peroxycarboxylic acid, and the sterilizing effect of the mixture is better than that of single peroxyacetic acid. The short carbon chain peroxyacetic acid has too short carbon chain length and poor lipid permeability, while the carbon chain structure of the medium and long carbon chain peroxycarboxylic acid can enable the selectivity of the combination of the short carbon chain peroxyacetic acid and the-SH group of the bacterial intracellular enzyme to be stronger, so that factors of protein and nucleic acid functional groups can be changed or the activity of the protein and the nucleic acid functional groups can be inhibited more easily, hydrophobic medium and long carbon chain peroxycarboxylic acid and hydrophilic peroxyacetic acid formed in a system generate an antimicrobial synergistic effect, and the sterilization effect of the short carbon chain peroxyacetic acid is better than that of a single peroxyacetic acid disinfectant.
(5) N atoms on the heterocycle of the ionic liquid surfactant have a pair of lone-pair electrons, so that N is changed into an active group, in an acidic environment medium, the metal surface has positive charges, the N atoms on the heterocycle of the ionic liquid surfactant and the metal surface are adsorbed on the metal surface through the action of electrostatic attraction, alkyl groups adsorbed through the electrostatic attraction are nonpolar groups, and the nonpolar groups are arranged in a direction on the metal surface to form a hydrophobic adsorption layer, are attached to the metal surface, and separate a corrosive medium from the metal surface, so that the corrosion of the metal is slowed down.
(6) The stabilizer can chelate metal ions in the system, inhibit the decomposition of peroxide and stabilize the system. Meanwhile, N atoms in the molecules of the stabilizer have lone-pair electrons, the metal surface has positive charges in an acid environment medium, the N atoms in the molecules of the stabilizer and the metal surface extrude water molecules on the metal surface under the action of electrostatic attraction and are directly attached to the surface of the metal material, and meanwhile, hydrogen ions are prevented from approaching the metal surface, so that hydrogen evolution is difficult to carry out, and a corrosion inhibition effect is realized on the metal.
(7) The acetate has aromatic odor, and can mask the pungent odor of acetic acid and reduce the adverse effect of odor during application. After the ionic liquid surfactant is dissolved in water, the hydrophilic group faces inwards, the hydrophobic agent faces outwards, and a film is formed on the surface of the solution to inhibit acetic acid from diffusing to the outside; meanwhile, micelle groups formed in the solution have an attraction effect on the acetic acid, and the volatilization of the acetic acid is inhibited, so that the pungent smell of the product is reduced.
Detailed Description
The technical solutions in the embodiments of the present invention will be clearly and completely described below with reference to the embodiments of the present invention, and it is obvious that the described embodiments are only a part of the embodiments of the present invention, and not all of the embodiments. All other embodiments, which can be derived by a person skilled in the art from the embodiments given herein without making any creative effort, shall fall within the protection scope of the present invention.
Firstly, the components and contents of the peracetic acid disinfectant in example 1 and the preparation method of the peracetic acid disinfectant are as follows.
A peroxyacetic acid disinfectant is prepared from the following raw materials: 12% of hydrogen peroxide; 10% of acetic acid; 5% of medium-long carbon chain organic acid; 5% of ionic liquid surfactant; 0.1% of a stabilizer; 1% of deodorant; the balance being water.
In this example, the medium-long carbon chain organic acid is heptanoic acid; the ionic liquid surfactant is imidazole; the stabilizer is dicarboxymethylalanine trisodium; the smell removing agent is methyl acetate.
The preparation method of the peroxyacetic acid disinfectant comprises the following steps:
(1) mixing, stirring and dissolving a stabilizer and hydrogen peroxide in proportion uniformly;
(2) adding medium-long carbon chain organic acid, deodorant and ionic liquid surfactant, stirring and dissolving uniformly;
(3) adding acetic acid, stirring, and adding water.
Secondly, the components and contents of the peroxyacetic acid disinfectant in examples 1 to 6 and comparative examples 1 to 6 and the preparation method of the peroxyacetic acid disinfectant are as follows.
TABLE 1 ingredients and contents of Peroxyacetic acid disinfectant solutions in examples 1-6 and comparative examples 1-6
The balance of water in examples 1 to 6 and comparative examples 1 to 6 was prepared in the same manner as in example 1, and is not specifically described herein.
And thirdly, carrying out performance test on the disinfectants in the examples 1-6 and the comparative examples 1-6.
3.1 Peroxyacetic acid stability
Peroxyacetic acid stability was performed according to the Disinfection Specification (2002 edition).
The prepared sample is stored for 90 days at 37 ℃, and the peroxyacetic acid content is respectively measured before and after the storage, and the test results are as follows.
The experimental result shows that the used stabilizer can effectively stabilize the content of the peroxyacetic acid in the system, and the stabilizer effect of the stabilizer on the peroxyacetic acid used in the patent is superior to that of the commonly used hydroxyethylidene diphosphonic acid; the reduction rate of the peroxyacetic acid content in the system without adding the stabilizer is large.
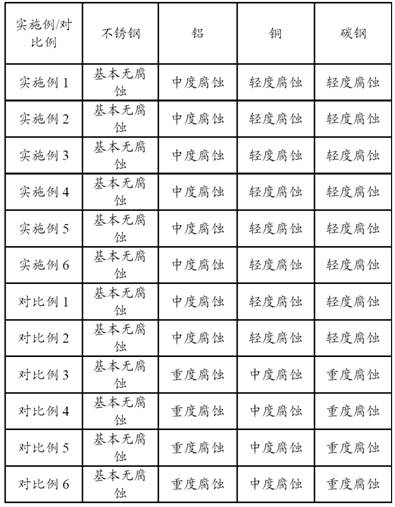
3.2 corrosive Properties of metals
Peroxyacetic acid metal corrosivity was performed according to the Disinfection Specification (2002 edition).
The results of the metal corrosion experiments are as follows.
The experimental result shows that after the ionic liquid surfactant and the stabilizing agent are added, the corrosion of the peroxyacetic acid in the system to metal is reduced.
3.3 germicidal Effect
The sterilizing effect of the peracetic acid disinfectant is carried out according to the disinfection technical specification (2002 edition).
The results of the peracetic acid disinfectant on the sterilization of escherichia coli are as follows.
The peroxyacetic acid disinfectant has the following sterilization result on staphylococcus aureus.
The results of the peroxyacetic acid disinfectant on the sterilization of the bacillus subtilis var niger spores are as follows.
The peroxyacetic acid has a microorganism killing effect, and the ionic liquid surfactant has a synergistic promotion effect on the peroxyacetic acid sterilization effect; the ionic liquid surfactant can catalyze the reaction of medium-long chain carboxylic acid to generate medium-long chain peroxycarboxylic acid, and has a synergistic effect on the bactericidal effect of peroxyacetic acid.
The above disclosure is only for the purpose of illustrating the preferred embodiments of the present invention and is not to be construed as limiting the scope of the present invention, therefore, the present invention is not limited by the appended claims.
Claims (7)
1. The peroxyacetic acid disinfectant is characterized by comprising the following preparation raw materials in percentage by weight: 10% -30% of hydrogen peroxide; 10% -30% of acetic acid; 1% -5% of medium-long carbon chain organic acid; 1% -5% of ionic liquid surfactant; 0.1% -2% of a stabilizer; 1% -5% of a deodorant; the balance being water.
2. The peracetic acid disinfectant as set forth in claim 1, wherein the weight ratio of hydrogen peroxide to acetic acid is from 1.2 to 2: 1.
3. the peroxyacetic acid disinfectant of claim 1 wherein the medium-long carbon chain organic acid is selected from one or more of heptanoic acid, octanoic acid, nonanoic acid and decanoic acid.
4. The peroxyacetic acid disinfectant of claim 1 wherein the ionic liquid surfactant is a cationic liquid surfactant selected from the group consisting of one or more of imidazole, pyridine, and quinoline.
5. The peroxyacetic acid disinfectant of claim 1 wherein the stabilizing agent is selected from at least one of trisodium dicarboxymethylalanine, tetrasodium glutamate diacetate, and trisodium methylglycine diacetate.
6. The peracetic acid disinfectant of claim 1, wherein the odor eliminating agent is one or more selected from the group consisting of methyl acetate, ethyl acetate, propyl acetate and butyl acetate.
7. A process for preparing a peroxyacetic acid disinfectant of any one of claims 1 to 6 comprising the steps of:
(1) mixing and stirring a stabilizer and hydrogen peroxide according to a proportion;
(2) adding medium-long carbon chain organic acid, deodorant and ionic liquid surfactant, mixing and stirring;
(3) adding acetic acid and supplementing water.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202210092725.2A CN114097783A (en) | 2022-01-26 | 2022-01-26 | Peracetic acid disinfectant and preparation method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202210092725.2A CN114097783A (en) | 2022-01-26 | 2022-01-26 | Peracetic acid disinfectant and preparation method thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN114097783A true CN114097783A (en) | 2022-03-01 |
Family
ID=80361622
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202210092725.2A Pending CN114097783A (en) | 2022-01-26 | 2022-01-26 | Peracetic acid disinfectant and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN114097783A (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115669662A (en) * | 2022-11-21 | 2023-02-03 | 成都科宏达科技有限公司 | Low-irritation stable monoperoxyacetic acid disinfectant and preparation method thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106614584A (en) * | 2016-12-01 | 2017-05-10 | 广东环凯微生物科技有限公司 | Compound peroxycarboxylic acid disinfection solution and preparation method thereof |
| US20170347645A1 (en) * | 2014-11-28 | 2017-12-07 | Laurence Geret | Two components disinfecting composition containing peracetic acid and chelating agent |
-
2022
- 2022-01-26 CN CN202210092725.2A patent/CN114097783A/en active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20170347645A1 (en) * | 2014-11-28 | 2017-12-07 | Laurence Geret | Two components disinfecting composition containing peracetic acid and chelating agent |
| CN106614584A (en) * | 2016-12-01 | 2017-05-10 | 广东环凯微生物科技有限公司 | Compound peroxycarboxylic acid disinfection solution and preparation method thereof |
Non-Patent Citations (4)
| Title |
|---|
| 李孟 等: "《新型水处理材料的理论与应用研究》", 31 August 2005, 武汉理工大学出版社,第1版 * |
| 王增品 等: "《腐蚀与防护工程》", 30 April 1991, 高等教育出版社,第1版 * |
| 王萍 等: "离子液体表面活性剂的合成与应用( Ⅸ)——杀菌性能", 《日用化学工业》 * |
| 范云场: "《酶抑制技术在评估离子液体毒性中的应用》", 30 September 2017, 中国矿业大学出版社,第1版 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115669662A (en) * | 2022-11-21 | 2023-02-03 | 成都科宏达科技有限公司 | Low-irritation stable monoperoxyacetic acid disinfectant and preparation method thereof |
| CN115669662B (en) * | 2022-11-21 | 2024-04-30 | 成都科宏达科技有限公司 | Low-irritation-stability unitary peroxyacetic acid disinfectant and preparation method thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1076164C (en) | Compositions and uses thereof | |
| US5674538A (en) | Process for inhibition of microbial growth in aqueous food transport or process streams | |
| US8940792B2 (en) | Antimicrobial composition and methods for using same | |
| US20190045789A1 (en) | Antimicrobial composition | |
| WO2005014057A1 (en) | Disinfecting compositions and methods of making and using same | |
| JP2008543850A (en) | Methods and compositions for inhibiting microbial growth on aqueous systems and objects | |
| EP4087557A1 (en) | Solid state antimicrobial compositions and methods for producing and using same | |
| CN111296476A (en) | Long-acting disinfectant containing potassium hydrogen persulfate and preparation method thereof | |
| CA2611796A1 (en) | Improved antimicrobial peroxidase compositions | |
| WO2021240315A1 (en) | Probiotic bacillus sanitiser | |
| CN114097783A (en) | Peracetic acid disinfectant and preparation method thereof | |
| CN112755050B (en) | Alcohol-free iodine-containing disinfectant with high component stability and preparation method thereof | |
| CN115669662B (en) | Low-irritation-stability unitary peroxyacetic acid disinfectant and preparation method thereof | |
| KR100837194B1 (en) | Liquid antimicrobial composition | |
| WO2021142148A1 (en) | Solid state antimicrobial compositions and methods for producing and using same | |
| EP1393629A1 (en) | Aqueous disinfecting compositions based on quaternary ammonium monomers | |
| KR102787089B1 (en) | Solid Peracetic Acid Disinfectant | |
| JP7475424B2 (en) | Ready-to-use disinfectant | |
| CN113303330A (en) | Composite disinfectant and preparation process thereof | |
| US20230030675A1 (en) | Solid state antimicrobial compositions and methods for producing and using same | |
| HK40091974A (en) | Solid composition for producing antibacterial, antiviral, antifungal and disinfectant solutions | |
| RU2745120C2 (en) | Disinfectant agent | |
| CN121511980A (en) | A weakly alkaline, low-corrosion, and highly efficient solid sterilization composition and its preparation method | |
| CN115474610A (en) | Hydrogen peroxide disinfectant and preparation method thereof | |
| EA049230B1 (en) | KIT FOR PREPARING ANTIBACTERIAL, ANTIVIRAL, ANTIFUNGAL OR DISINFECTANT SOLUTION |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20220301 |
|
| RJ01 | Rejection of invention patent application after publication |