CN113995751B - Application of epalrestat in the preparation of medicine for preventing and treating ischemic stroke - Google Patents
Application of epalrestat in the preparation of medicine for preventing and treating ischemic stroke Download PDFInfo
- Publication number
- CN113995751B CN113995751B CN202111346876.8A CN202111346876A CN113995751B CN 113995751 B CN113995751 B CN 113995751B CN 202111346876 A CN202111346876 A CN 202111346876A CN 113995751 B CN113995751 B CN 113995751B
- Authority
- CN
- China
- Prior art keywords
- epalrestat
- ischemic stroke
- medicament
- group
- ischemic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/425—Thiazoles
- A61K31/426—1,3-Thiazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/02—Antithrombotic agents; Anticoagulants; Platelet aggregation inhibitors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
Landscapes
- Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Neurology (AREA)
- Diabetes (AREA)
- Biomedical Technology (AREA)
- Hematology (AREA)
- Neurosurgery (AREA)
- Epidemiology (AREA)
- Urology & Nephrology (AREA)
- Vascular Medicine (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
技术领域technical field
本发明属于医药技术领域,尤其涉及依帕司他在制备防治缺血性脑卒中的药物中的应用。The invention belongs to the technical field of medicine, and in particular relates to the application of epalrestat in the preparation of medicines for preventing and treating ischemic stroke.
背景技术Background technique
脑卒中又称中风,是一种急性脑血管疾病,是由于脑部血管突然破裂或因血管阻塞导致血流不能流入大脑而引起脑组织损伤的疾病,包括缺血性和出血性卒中,其中缺血性卒中的发病率高于出血性卒中,占脑卒中总数的60~70%。缺血性脑卒中是一个复杂的多系统参与的过程,缺血后会导致神经元损伤,进而血脑屏障通透性增加,造成脑水肿,致使颅内压升高,形成脑疝,导致疾病恶化。Stroke, also known as apoplexy, is an acute cerebrovascular disease. It is a disease that causes brain tissue damage due to sudden rupture of blood vessels in the brain or blockage of blood vessels that prevents blood from flowing into the brain, including ischemic and hemorrhagic strokes. The incidence of hemorrhagic stroke is higher than that of hemorrhagic stroke, accounting for 60-70% of the total number of strokes. Ischemic stroke is a complex process involving multiple systems. After ischemia, neurons will be damaged, and the permeability of the blood-brain barrier will increase, causing cerebral edema, resulting in increased intracranial pressure, forming brain herniation, and leading to disease. deterioration.
我国治疗缺血性脑卒中的用药以三类药物为主:(1)改善脑循环药物:溶栓药物(t-PA、尿激酶)、抗血小板药物(阿司匹林)、抗凝药物(肝素)和其他类药物(丁基苯酞等);(2)神经保护药物:神经保护剂(依达拉奉、胞二磷胆碱、神经节苷脂等);(3)其他:丹参、川穹等中药。在血管闭塞的情况下,静脉内使用t-PA等溶栓药物早期溶栓治疗是当前针对缺血性脑卒中的标准疗法,及时恢复卒中患者的血流可以有效减轻神经功能缺失。对于符合国家和国际资格指南的患者,在脑缺血发作4.5小时内给予t-PA可以在3~6个月内改善预后。In my country, there are mainly three types of drugs for the treatment of ischemic stroke: (1) drugs for improving cerebral circulation: thrombolytic drugs (t-PA, urokinase), antiplatelet drugs (aspirin), anticoagulant drugs (heparin) and Other drugs (butylphthalide, etc.); (2) Neuroprotective drugs: neuroprotective agents (edaravone, citicoline, gangliosides, etc.); (3) Others: Danshen, Chuanqiong, etc. traditional Chinese medicine. In the case of vascular occlusion, early thrombolytic therapy with intravenous thrombolytic drugs such as t-PA is currently the standard therapy for ischemic stroke, and timely restoration of blood flow in stroke patients can effectively alleviate neurological deficits. For patients meeting national and international eligibility guidelines, administration of t-PA within 4.5 hours of ischemic onset improves outcomes within 3 to 6 months.
但溶栓治疗存在较大的限制,治疗时间窗狭窄,且溶栓后血流再通时会导致再灌注损伤,加重缺血性脑损伤。此外,缺血后脑组织损伤是一个复杂过程,该过程不但包括缺血缺氧对神经细胞的直接损伤,还包括在直接损伤的基础上诱发的多种炎症细胞和炎症介质对脑组织的间接损伤。越来越多研究证实,免疫参与在缺血性脑卒中发病过程中起到重要的作用。目前,针对已经错过治疗时间窗、不能使用溶栓治疗的缺血性脑卒中患者的缺血性脑损伤仍缺少有效的防治药物。However, thrombolytic therapy has major limitations, such as a narrow treatment time window, and reperfusion injury after thrombolytic recanalization, which can aggravate ischemic brain injury. In addition, brain tissue damage after ischemia is a complex process, which includes not only the direct damage to nerve cells caused by ischemia and hypoxia, but also the indirect damage to brain tissue induced by various inflammatory cells and inflammatory mediators on the basis of direct damage. . More and more studies have confirmed that immune involvement plays an important role in the pathogenesis of ischemic stroke. At present, there is still a lack of effective prevention and treatment drugs for ischemic brain injury in patients with ischemic stroke who have missed the treatment time window and cannot use thrombolytic therapy.
发明内容Contents of the invention
本发明针对缺血性脑卒中发病早期及已经错过治疗时间窗、不能使用溶栓治疗的缺血性脑卒中患者,提供了依帕司他在制备防治缺血性脑卒中的药物中的应用。The present invention provides the application of epalrestat in the preparation of medicines for the prevention and treatment of ischemic stroke, aiming at ischemic stroke patients in the early stage of the onset of ischemic stroke and who have missed the treatment time window and cannot be treated with thrombolysis.
本发明的技术方案:Technical scheme of the present invention:
依帕司他Epalrestat是一种醛糖还原酶抑制药,以可逆地抑制与糖尿病性并发症的发病机制相关的多元醇代谢中葡萄糖转化为山梨醇的醛糖还原酶而发挥作用,用于预防、改善和治疗糖尿病并发的末梢神经障碍。Epalrestat is an aldose reductase inhibitor drug that reversibly inhibits the aldose reductase that converts glucose into sorbitol in polyol metabolism related to the pathogenesis of diabetic complications, and is used to prevent , Improve and treat peripheral nerve disorders complicated by diabetes.
本发明提供了一种依帕司他的药物新用途,即依帕司他在制备防治缺血性脑卒中的药物中的应用。The invention provides a new drug application of epalrestat, that is, the application of epalrestat in the preparation of drugs for preventing and treating ischemic stroke.
进一步的,所述缺血性脑卒中的临床类型为脑血栓形成、腔隙性梗死或脑栓塞。Further, the clinical type of ischemic stroke is cerebral thrombosis, lacunar infarction or cerebral embolism.
进一步的,所述防治缺血性脑卒中的药物中依帕司他为唯一活性成分。Further, epalrestat is the only active ingredient in the drug for preventing and treating ischemic stroke.
进一步的,所述防治缺血性脑卒中的药物还包括药学上可接受的辅料和/或载体。Further, the medicament for preventing and treating ischemic stroke also includes pharmaceutically acceptable adjuvants and/or carriers.
进一步的,所述防治缺血性脑卒中的药物中依帕司他的含量为0.1~99wt%。Further, the content of epalrestat in the drug for preventing and treating ischemic stroke is 0.1-99wt%.
进一步的,所述防治缺血性脑卒中的药物的剂型包括口服剂型及非经胃肠道给药剂型。Further, the dosage form of the drug for preventing and treating ischemic stroke includes oral dosage form and parenteral dosage form.
进一步的,所述防治缺血性脑卒中的药物的剂型为非经胃肠道给药剂型,具体为注射给药剂型、呼吸道给药剂型、皮肤给药剂型、黏膜给药剂型或腔道给药剂型。Further, the dosage form of the drug for preventing and treating ischemic stroke is a parenteral administration dosage form, specifically an injection dosage form, a respiratory dosage dosage form, a skin dosage dosage form, a mucosal dosage dosage form or an oral cavity dosage dosage form. drug form.
进一步的,所述防治缺血性脑卒中的药物的剂型为口服剂型,具体为颗粒剂、片剂、胶囊剂、丸剂、滴丸剂或口服液体制剂。Further, the dosage form of the drug for preventing and treating ischemic stroke is an oral dosage form, specifically granules, tablets, capsules, pills, drop pills or oral liquid preparations.
本发明的有益效果:Beneficial effects of the present invention:
本发明提供了一种依帕司他的药物新用途,即依帕司他在制备防治缺血性脑卒中的药物中的应用。在缺血性脑卒中后使用依帕司他进行治疗,能够通过抑制脑血管内皮细胞醛糖还原酶的表达,进而上调脑微血管内皮细胞的紧密连接蛋白表达,减轻内皮细胞凋亡,改善缺血性脑卒中后的神经功能障碍,维持缺血性脑卒中后血脑屏障的完整性;此外,依帕司他能够有效降低外周炎症细胞的浸润,减轻缺血脑组织中炎症反应。本发明为缺血性脑卒中发病早期及已经错过治疗时间窗、不能使用溶栓治疗的缺血性脑卒中患者提供了一条新的有效治疗途径,为依帕司他治疗临床缺血性脑卒中提供了理论依据。The invention provides a new drug application of epalrestat, that is, the application of epalrestat in the preparation of drugs for preventing and treating ischemic stroke. The use of epalrestat after ischemic stroke can inhibit the expression of aldose reductase in cerebral vascular endothelial cells, thereby up-regulating the expression of tight junction protein in cerebral microvascular endothelial cells, reducing endothelial cell apoptosis, and improving ischemia In addition, epalrestat can effectively reduce the infiltration of peripheral inflammatory cells and reduce the inflammatory response in ischemic brain tissue. The present invention provides a new and effective treatment method for patients with ischemic stroke who are in the early stage of onset of ischemic stroke and who have missed the treatment time window and cannot be treated with thrombolysis, and provide epalrestat for the treatment of clinical ischemic stroke A theoretical basis is provided.
附图说明Description of drawings
图1、图2依次为实施例1缺血脑组织TTC染色图和坏死组织体积对比图;Figure 1 and Figure 2 are sequentially the TTC staining diagram of the ischemic brain tissue and the volume comparison diagram of the necrotic tissue in Example 1;
图3为实施例2脑缺血过程中小鼠神经行为学评分结果图;Fig. 3 is the mouse neurobehavioral score result figure in the cerebral ischemia process of
图4、图5依次为实施例3中缺血脑组织EB染色图和缺血脑组织中EB含量对比图;Figure 4 and Figure 5 are sequentially the EB staining diagram of the ischemic brain tissue and the comparison diagram of the EB content in the ischemic brain tissue in Example 3;
图6、图7依次为实施例4脑微血管内皮细胞(CD31)周围巨噬细胞(F4/80)的荧光染色照片和巨噬细胞平均荧光强度对比图;Fig. 6 and Fig. 7 are the photographs of the fluorescent staining of the macrophages (F4/80) around the brain microvascular endothelial cells (CD31) in Example 4 and the comparison chart of the average fluorescence intensity of the macrophages;
图8为实施例4流式细胞术测定缺血脑组织中浸润中性粒细胞结果图;Fig. 8 is a diagram showing the results of measuring infiltrating neutrophils in ischemic brain tissue by flow cytometry in Example 4;
图9为实施例4缺血脑组织中浸润的中性粒细胞数百分比对比图;Figure 9 is a comparison chart of the percentage of infiltrated neutrophils in ischemic brain tissue in Example 4;
图10、图11和图12依次为实施例5Western Blot实验测定缺血脑组织中TJs蛋白图、Occludin蛋白的表达量对比图和ZO-1蛋白的表达量对比图;Figure 10, Figure 11 and Figure 12 are the TJs protein map, the Occludin protein expression comparison chart and the ZO-1 protein expression comparison chart in the ischemic brain tissue measured by Western Blot experiment in Example 5;
图13、图14依次为实施例6缺血脑组织中内皮细胞(CD31)和Occludin共染照片和内皮细胞(CD31)和Occludin共染平均荧光强度对比图;Figure 13 and Figure 14 are sequentially the co-staining photos of endothelial cells (CD31) and Occludin in the ischemic brain tissue of Example 6 and the comparison chart of the average fluorescence intensity of endothelial cells (CD31) and Occludin co-staining;
图15、图16依次为实施例6缺血脑组织中内皮细胞(GFAP)和ZO-1共染照片和内皮细胞(CD31)和ZO-1共染平均荧光强度对比图;Figure 15 and Figure 16 are sequentially the co-staining photos of endothelial cells (GFAP) and ZO-1 in the ischemic brain tissue of Example 6 and the comparison chart of the average fluorescence intensity of endothelial cells (CD31) and ZO-1 co-staining;
图17、图18依次为实施例6缺血脑组织中星形胶质细胞(GFAP)周围Occludin的共染照片和星形胶质细胞(GFAP)周围Occludin的平均荧光强度对比图;Figure 17 and Figure 18 are sequentially the co-stained photos of Occludin around astrocytes (GFAP) in the ischemic brain tissue of Example 6 and the comparison chart of the average fluorescence intensity of Occludin around astrocytes (GFAP);
图19、图20依次为实施例6缺血脑组织中星形胶质细胞(GFAP)周围ZO-1的共染照片和星形胶质细胞(GFAP)周围ZO-1的平均荧光强度对比图;Figure 19 and Figure 20 are the co-stained photos of ZO-1 around astrocytes (GFAP) and the comparison of the average fluorescence intensity of ZO-1 around astrocytes (GFAP) in the ischemic brain tissue of Example 6 ;
图21、图22和图23依次为实施例7Western Blot实验测定缺血脑组织中Bax、Bcl2和Cleaved-casp3凋亡相关蛋白图、Cleaved-casp3蛋白的表达量对比图和Bcl2与Bax蛋白的比值对比图;Figure 21, Figure 22 and Figure 23 are the diagrams of Bax, Bcl2 and Cleaved-casp3 apoptosis-related proteins in ischemic brain tissue measured by Western Blot experiment in Example 7, the comparison chart of Cleaved-casp3 protein expression, and the ratio of Bcl2 to Bax protein comparison chart;
图24、图25和图26依次为实施例8Western Blot实验测定OGD作用下内皮细胞中TJs蛋白图、ZO-1蛋白的表达量对比图和Occludin蛋白的表达量对比图;Figure 24, Figure 25 and Figure 26 are the TJs protein map, the expression comparison chart of ZO-1 protein and the comparison chart of Occludin protein expression in the endothelial cells under the action of OGD determined by Western Blot experiment in Example 8;
图27为实施例8OGD作用下体外血脑屏障的通透性统计结果对比图;Figure 27 is a comparison chart of the statistical results of the permeability of the blood-brain barrier in vitro under the action of Example 8 OGD;
图28、图29和图30依次为实施例9Western Blot实验测定OGD作用下内皮细胞中Bax、Bcl2和Cleaved-Casp3的蛋白图、Cleaved-casp3蛋白的表达量对比图和Bcl2与Bax蛋白的比值对比图;Figure 28, Figure 29 and Figure 30 are the protein maps of Bax, Bcl2 and Cleaved-Casp3 in endothelial cells under the action of OGD measured by Western Blot in Example 9, the comparison chart of the expression of Cleaved-casp3 protein, and the comparison of the ratio of Bcl2 and Bax proteins picture;
图31、图32依次为实施例9流式细胞术测定内皮细胞PI和AnnexinV的表达结果图和各组内皮细胞PI和AnnexinV的表达量对比图;Fig. 31 and Fig. 32 are the results of flow cytometry determination of the expression of PI and AnnexinV in endothelial cells in Example 9 and the comparison of the expression levels of PI and AnnexinV in endothelial cells in each group;
图33、图34依次为实施例10内皮细胞(CD31)和LC3B蛋白的共染照片和内皮细胞(CD31)和LC3B共染的平均荧光强度对比图;Figure 33 and Figure 34 are the photos of the co-staining of endothelial cells (CD31) and LC3B protein in Example 10 and the comparison chart of the average fluorescence intensity of the co-staining of endothelial cells (CD31) and LC3B;
图35、图36依次为实施例10内皮细胞(CD31)和Beclin1蛋白的共染照片和内皮细胞(CD31)和Beclin1共染的平均荧光强度对比图;Figure 35 and Figure 36 are the co-staining photos of endothelial cells (CD31) and Beclin1 protein in Example 10 and the comparison chart of the average fluorescence intensity of endothelial cells (CD31) and Beclin1 co-staining;
图37、图38依次为实施例11Western Blot实验测定bEnd.3细胞中LC3B-I和LC3B-II自噬相关蛋白图和LC3B-I与LC3B-II的比值对比图;Figure 37 and Figure 38 are the autophagy-related protein maps of LC3B-I and LC3B-II in bEnd.3 cells measured by Western Blot experiment in Example 11 and the comparison chart of the ratio between LC3B-I and LC3B-II;
图39为实施例11内皮细胞(CD31)和Beclin1共染照片;Figure 39 is a photo of co-staining of endothelial cells (CD31) and Beclin1 in Example 11;
图40为实施例11内皮细胞(CD31)和Beclin1共染平均荧光强度对比图;Figure 40 is a comparison chart of average fluorescence intensity co-stained with endothelial cells (CD31) and Beclin1 in Example 11;
图41为实施例11内皮细胞(CD31)和LAMP-1共染照片;Figure 41 is a photo of co-staining of endothelial cells (CD31) and LAMP-1 in Example 11;
图42为实施例11内皮细胞(CD31)和LAMP-1共染平均荧光强度对比图;Figure 42 is a comparison chart of average fluorescence intensity co-stained with endothelial cells (CD31) and LAMP-1 in Example 11;
图43为实施例11内皮细胞(CD31)和P62共染照片;Fig. 43 is the photo of co-staining of endothelial cells (CD31) and P62 in embodiment 11;
图44为实施例11内皮细胞(CD31)和P62共染平均荧光强度对比图;Figure 44 is a comparison chart of average fluorescence intensity co-stained with endothelial cells (CD31) and P62 in Example 11;
图45、图46依次为实施例12Western Blot实验测定缺血后脑组织中AR蛋白图和AR蛋白的表达量对比图;Figure 45 and Figure 46 are the comparison charts of the AR protein map and the expression level of AR protein in the brain tissue after ischemia measured by Western Blot experiment in Example 12;
图47为实施例12缺血后脑组织中内皮细胞(CD31)和AR蛋白的共染照片;Figure 47 is a co-staining photo of endothelial cells (CD31) and AR protein in ischemic brain tissue in Example 12;
图48为实施例12缺血后脑组织中内皮细胞(CD31)和AR共染的平均荧光强度对比图;Fig. 48 is a comparison diagram of average fluorescence intensity co-stained with endothelial cells (CD31) and AR in ischemic brain tissue in Example 12;
图49、图50依次为实施例12OGD不同时间内皮细胞中AR蛋白图和AR蛋白的表达量对比图;Figure 49 and Figure 50 are the graphs of AR protein in different endothelial cells of the embodiment 12 OGD and the comparison charts of the expression of AR protein in sequence;
图51、图52和图53依次为实施例13依帕司他改变内皮细胞中p-AKT和p-mTOR蛋白表达的对比图、p-AKT蛋白表达量的对比图和p-mTOR蛋白表达量的对比图;Fig. 51, Fig. 52 and Fig. 53 are the comparison diagram of epalrestat changing the protein expression of p-AKT and p-mTOR in endothelial cells, the comparison diagram of p-AKT protein expression and the expression of p-mTOR protein in Example 13 comparison chart;
图54、图55和图56依次为实施例13依帕司他、AKTi和mTORi改变内皮细胞中p-AKT和p-mTOR蛋白表达的对比图、p-AKT蛋白表达量对比图和p-mTOR蛋白表达量对比图;Figure 54, Figure 55 and Figure 56 are the comparison chart of p-AKT and p-mTOR protein expression in endothelial cells changed by epalrestat, AKTi and mTORi in Example 13, the comparison chart of p-AKT protein expression and p-mTOR Protein expression comparison chart;
图57、图58和图59依次为实施例13依帕司他、AKTi和mTORi改变内皮细胞中ZO-1和Occludind的蛋白表达的对比图、ZO-1蛋白表达量对比图和Occludind蛋白表达量对比图;Fig. 57, Fig. 58 and Fig. 59 are the comparison diagram of the protein expression of ZO-1 and Occludind in the endothelial cells changed by epalrestat, AKTi and mTORi in Example 13, the comparison diagram of the protein expression of ZO-1 and the expression of Occludind protein comparison chart;
图60、图61和图62依次为实施例14依帕司他、AKTi和mTORi改变内皮细胞中Bax、Bcl2和Cleaved-Casp3的蛋白表达的对比图、Cleaved-Casp3的蛋白表达量对比图和Bcl2/Bax的比值对比图;Figure 60, Figure 61 and Figure 62 are the comparison charts of the protein expression of Bax, Bcl2 and Cleaved-Casp3 in endothelial cells changed by epalrestat, AKTi and mTORi in Example 14, and the comparison chart of the protein expression of Cleaved-Casp3 and Bcl2 /Bax ratio comparison chart;
图63、图64和图65依次为实施例14依帕司他、AKTi和mTORi改变内皮细胞中LC3B-I、LC3B-II和Beclin-1的蛋白表达的对比图、Beclin-1的蛋白表达量对比图和Beclin-1的比值对比图。Figure 63, Figure 64 and Figure 65 are the comparison charts of the protein expression of LC3B-I, LC3B-II and Beclin-1 in endothelial cells changed by epalrestat, AKTi and mTORi in Example 14, and the protein expression of Beclin-1 Comparison chart and ratio comparison chart of Beclin-1.
具体实施方式Detailed ways
下面结合实施例对本发明的技术方案做进一步的说明,但并不局限于此,凡是对本发明技术方案进行修改或者等同替换,而不脱离本发明技术方案的精神和范围,均应涵盖在本发明的保护范围中。下列实施例中未具体注明的工艺设备或装置均采用本领域内的常规设备或装置,若未特别指明,本发明实施例中所用的原料等均可市售获得;若未具体指明,本发明实施例中所用的技术手段均为本领域技术人员所熟知的常规手段。The technical solution of the present invention will be further described below in conjunction with the examples, but it is not limited thereto. Any modification or equivalent replacement of the technical solution of the present invention without departing from the spirit and scope of the technical solution of the present invention should be covered by the present invention within the scope of protection. The process equipment or devices not specifically indicated in the following examples all adopt conventional equipment or devices in the art. If not specified, the raw materials used in the examples of the present invention, etc. can be commercially available; if not specified, this The technical means used in the embodiments of the invention are conventional means well known to those skilled in the art.
以下实施例将通过体内实验和体外实验分别验证依帕司他对于缺血性脑卒中的防治效果。The following examples will verify the prevention and treatment effect of epalrestat on ischemic stroke through in vivo experiments and in vitro experiments.
实施例1Example 1
本实施例考察了脑缺血过程中,依帕司他对脑缺血损伤的影响。This example investigates the effect of epalrestat on cerebral ischemic injury in the process of cerebral ischemia.
本发明使用的实验动物均为纯种C57BL/6雄性小鼠,体重在19~21g之间,购买于辽宁长生生物技术股份有限公司。The experimental animals used in the present invention are all purebred C57BL/6 male mice with a body weight of 19-21 g, purchased from Liaoning Changsheng Biotechnology Co., Ltd.
建模、分组、给药方法:分别建立小鼠永久性大脑中动脉阻塞模型(pMCAL)和假手术组sham,将pMCAL模型和假手术(sham)组分别分为正常(缺血)组-标记为Normal组、依帕司他治疗组-Eps组和sham组,在建立模型后立即灌胃给药,Eps组给药剂量为100μg/g,Normal组灌胃等量生理盐水。Modeling, grouping, and administration methods: establish the mouse permanent middle cerebral artery occlusion model (pMCAL) and the sham operation group sham, respectively, and divide the pMCAL model and the sham operation (sham) group into normal (ischemic) groups - labeling The normal group, the epalrestat treatment group-Eps group and the sham group were intragastrically administered immediately after the establishment of the model. The dosage of the Eps group was 100 μg/g, and the Normal group was intragastrically administered with the same amount of normal saline.
在缺血时间分别为12h、1d、3d和5d的时间点取脑组织切片,按照本领域常规TTC染色方法对脑组织切片进行TTC染色(TTC全称为2,3,5-氯化三苯基四氮唑)。Brain tissue sections were taken at the time points of ischemia time of 12h, 1d, 3d and 5d respectively, and TTC staining was performed on the brain tissue sections according to the conventional TTC staining method in this field (TTC is called 2,3,5-triphenyl chloride Tetrazolium).
TTC染色实验结果如图1所示,小鼠脑组织均分5片,厚度2mm/片,红色区域为正常脑组织,白色区域为脑缺血坏死组织。统计各组小鼠脑坏死组织体积如图2显示,与正常组相比较,脑缺血1d,3d和5d依帕司他治疗组脑坏死组织体积有所减小,具有统计学差异(*P<0.05,**P<0.01),由此证明依帕司他治疗能够减轻脑缺血损伤。The results of the TTC staining experiment are shown in Figure 1. The mouse brain tissue was divided into 5 slices with a thickness of 2 mm per slice. The red area is normal brain tissue, and the white area is cerebral ischemic necrosis tissue. The volume of brain necrosis tissue in each group of mice was counted as shown in Figure 2. Compared with the normal group, the volume of brain necrosis tissue in the
实施例2Example 2
本实施例考察了脑缺血过程中,依帕司他对小鼠神经行为学功能的影响。This example investigates the effect of epalrestat on the neurobehavioral function of mice during cerebral ischemia.
本实施例观察脑缺血后小鼠的状态和神经行为学变化,反应小鼠脑缺血严重程度情况,按照实验计划对小鼠进行pMCAL模型,建模、分组、给药方法与实施例1相同。正常小鼠神经评分为0分,与缺血相关的症状越严重评分越大,神经行为评分标准如表1所示:This example observes the state and neurobehavioral changes of mice after cerebral ischemia, and reflects the severity of cerebral ischemia in mice. According to the experimental plan, mice are modeled with pMCAL, and the modeling, grouping, and administration methods are the same as in Example 1. same. The neurological score of normal mice is 0, and the more severe the symptoms related to ischemia, the greater the score. The neurobehavioral scoring criteria are shown in Table 1:
表1Table 1
通过比较图3所示各组小鼠缺血后神经行为学评分结果发现,缺血组小鼠神经行为学表现明显差于假手术(Sham)组;脑缺血3d,正常组缺血小鼠神经行为学评分高于依帕司他治疗组,具有统计差异(*P<0.05),这说明依帕司他治疗能够恢复小鼠部分神经功能,改善缺血损伤引起的神经功能障碍。By comparing the neurobehavioral scores of each group of mice after ischemia shown in Figure 3, it was found that the neurobehavioral performance of the mice in the ischemia group was significantly worse than that of the sham operation (Sham) group; The neurobehavioral score was higher than that of the epalrestat treatment group, with a statistical difference (*P<0.05), which indicated that epalrestat treatment could restore part of the neurological function of the mice and improve the neurological dysfunction caused by ischemic injury.
实施例3Example 3
本实施例考察了脑缺血过程中,依帕司他对血脑屏障功能的影响。This example investigates the effect of epalrestat on the blood-brain barrier function during cerebral ischemia.
本实施例建模、分组、给药方法均与实施例1相同,在缺血时间分别为12h、1d、3d和5d的时间点对小鼠进行尾静脉注射Evans Blue(EB)染料,2h后灌流取出脑组织观察脑组织蓝色染料渗透情况。结果如图4所示,蓝色为血脑屏障破坏区域,白色为正常脑组织;脑缺血损伤导致血脑屏障完整性破坏,EB染料能够通过破损的血脑屏障进入脑实质将缺血区域染成蓝色。The modeling, grouping, and administration methods of this embodiment are all the same as in Example 1, and the mice are injected with Evans Blue (EB) dye into the tail vein at the time point when the ischemia time is 12h, 1d, 3d, and 5d, and after 2h The brain tissue was removed by perfusion to observe the penetration of the blue dye in the brain tissue. The results are shown in Figure 4. The blue is the damaged area of the blood-brain barrier, and the white is the normal brain tissue; cerebral ischemia damage leads to the destruction of the integrity of the blood-brain barrier, and the EB dye can enter the brain parenchyma through the damaged blood-brain barrier and destroy the ischemic area. dyed blue.
通过三氯乙酸萃取,紫外分光光度计检测各组脑组织中EB的含量,统计分析数据如图5显示,正常组脑内EB的含量高于Sham组;与正常组相比较,脑缺血3d和5d的依帕司他治疗组缺血脑组织中EB的含量显著降低,具有统计差异(*P<0.05),这说明依帕司他治疗能够减轻脑缺血损伤过程中血脑屏障的损伤程度。Through the extraction of trichloroacetic acid, the content of EB in the brain tissue of each group was detected by ultraviolet spectrophotometer, and the statistical analysis data are shown in Figure 5. The content of EB in the brain of the normal group was higher than that of the sham group; compared with the normal group, the
实施例4Example 4
本实施例考察了脑缺血过程中,依帕司他对血管周围炎症细胞浸润的影响。This example investigates the effect of epalrestat on the infiltration of perivascular inflammatory cells during cerebral ischemia.
本实施例建模、分组、给药方法均与实施例1相同,取缺血时间3d的sham组、Normal-3d组和Eps-3d组的小鼠,通过免疫荧光染色检测脑内巨噬细胞的浸润情况,免疫荧光染色标记脑微血管内皮细胞周围巨噬细胞的结果图6显示,CD31标记血管内皮细胞呈绿色,F4/80标记巨噬细胞呈红色,DAPI标记细胞核呈蓝色;统计结果分析如图7显示,依帕司他治疗组缺血脑组织血管周围巨噬细胞的数量少于正常组,具有统计差异(**P<0.01)。In this example, the modeling, grouping, and administration methods are the same as in Example 1. The mice in the sham group, the Normal-3d group, and the Eps-3d group with an ischemia time of 3 days were taken, and the macrophages in the brain were detected by immunofluorescence staining. Figure 6 shows that the macrophages around the brain microvascular endothelial cells are marked by immunofluorescence staining, CD31-labeled vascular endothelial cells are green, F4/80-labeled macrophages are red, and DAPI-labeled nuclei are blue; statistical analysis As shown in Figure 7, the number of perivascular macrophages in the ischemic brain tissue of the epalrestat treatment group was less than that of the normal group, with a statistical difference (**P<0.01).
使用流式细胞术检测缺血脑组织内中性粒细胞的浸润情况,结果如图8显示,CD11b+CD45highLy6G+标记脑内浸润的中性粒细胞;统计各组中性粒细胞百分比如图9所示,与正常组相比较,依帕司他治疗组缺血脑组织内中性粒细胞数量(CD11b+CD45highLy6G+)百分比较低,具有统计差异(**P<0.01)。Flow cytometry was used to detect the infiltration of neutrophils in ischemic brain tissue, and the results are shown in Figure 8, CD11b + CD45 high Ly6G + labeled neutrophils infiltrated in the brain; the percentage of neutrophils in each group was counted as As shown in Figure 9, compared with the normal group, the number of neutrophils (CD11b + CD45 high Ly6G + ) in the ischemic brain tissue of the epalrestat treatment group was lower, with a statistical difference (**P<0.01).
以上结果说明,依帕司他治疗能够维持缺血损伤后血脑屏障的完整性,有效降低外周炎症细胞的浸润,减轻缺血脑组织中炎症反应。The above results show that epalrestat treatment can maintain the integrity of the blood-brain barrier after ischemic injury, effectively reduce the infiltration of peripheral inflammatory cells, and reduce the inflammatory response in ischemic brain tissue.
实施例5Example 5
血脑屏障主要由脑微血管内皮细胞(CD31)、星形胶质细胞(GFAP)和周细胞构成。脑微血管内皮细胞表达紧密连接蛋白TJs是维持血脑屏障选择通透性的结构基础。本实施例考察了脑缺血过程中,依帕司他对缺血脑半球中紧密连接蛋白TJs:Occludin和ZO-1表达的影响。The blood-brain barrier is mainly composed of brain microvascular endothelial cells (CD31), astrocytes (GFAP) and pericytes. The expression of tight junction proteins TJs in brain microvascular endothelial cells is the structural basis for maintaining the selective permeability of the blood-brain barrier. This example investigates the effect of epalrestat on the expression of tight junction proteins TJs: Occludin and ZO-1 in the ischemic cerebral hemisphere during cerebral ischemia.
本实施例建模、分组、给药方法均与实施例1相同,缺血不同时间灌流取脑组织提蛋白,通过Western Blot实验检测依帕司他对缺血脑组织中TJs表达的影响。结果如图10显示,缺血脑组织中TJs(Occludin和ZO-1)表达情况;统计分析如图11和图12所示,与Sham组相比较,正常组脑内ZO-1和Occludin的表达降低;脑缺血1d和3d依帕司他治疗组小鼠缺血脑组织中Occludin的表达量高于正常组,具有统计差异(*P<0.05,**P<0.01);脑缺血12h、1d和3d依帕司他治疗组小鼠缺血脑组织中ZO-1的表达量高于正常组,具有统计差异(*P<0.05,**P<0.01)。结果说明脑缺血过程中,依帕司他治疗能够上调缺血脑组织中TJs的表达量。The modeling, grouping, and administration methods of this example were the same as in Example 1. The protein was extracted from the brain tissue by perfusion at different times of ischemia, and the effect of epalrestat on the expression of TJs in the ischemic brain tissue was detected by Western Blot experiment. The results are shown in Figure 10, the expression of TJs (Occludin and ZO-1) in the ischemic brain tissue; the statistical analysis is shown in Figure 11 and Figure 12, compared with the Sham group, the expression of ZO-1 and Occludin in the brain of the normal group decreased; the expression of Occludin in ischemic brain tissue of mice treated with epalrestat for 1d and 3d of cerebral ischemia was higher than that of normal group, with statistical difference (*P<0.05, **P<0.01);
实施例6Example 6
本实施例考察了脑缺血过程中,依帕司他对内皮细胞(CD31)上和星形胶质细胞(GFAP)周围紧密连接蛋白TJS:Occludin和ZO-1表达的影响。This example investigates the effect of epalrestat on the expression of tight junction proteins TJS: Occludin and ZO-1 on endothelial cells (CD31) and around astrocytes (GFAP) during cerebral ischemia.
本实施例建模、分组、给药方法均与实施例1相同,使用免疫荧光分别检测内皮细胞上与星形胶质细胞周围紧密连接蛋白表达情况,各组脑缺血3d后取脑组织制备冰冻切片,Occludin和ZO-1分别与CD31和GFAP双染。The modeling, grouping, and administration methods of this example are the same as those in Example 1. Immunofluorescence is used to detect the expression of tight junction proteins on endothelial cells and around astrocytes, and the brain tissue of each group is prepared after 3 days of cerebral ischemia. Frozen sections were double-stained with Occludin and ZO-1, CD31 and GFAP, respectively.
通过免疫荧光染色检测缺血脑半球微血管内皮细胞表达Occludin的情况,结果如图13所示,CD31标记脑微血管内皮细胞呈绿色,Occludin染色呈红色,CD31和Occludin共表达呈黄色。统计数据分析结果如图14所示,依帕司他治疗组缺血脑组织中CD31和Occludin共表达的平均荧光强度明显高于正常组,具有统计差异(**P<0.01)。The expression of Occludin in ischemic brain microvascular endothelial cells was detected by immunofluorescence staining. The results are shown in Figure 13. CD31-labeled brain microvascular endothelial cells were green, Occludin staining was red, and co-expression of CD31 and Occludin was yellow. Statistical data analysis results are shown in Figure 14, the average fluorescence intensity of CD31 and Occludin co-expression in the ischemic brain tissue of the epalrestat treatment group was significantly higher than that of the normal group, with a statistical difference (**P<0.01).
通过免疫荧光染色检测缺血脑半球微血管内皮细胞表达ZO-1的情况,结果如图15所示,CD31标记脑微血管内皮细胞呈绿色,ZO-1染色呈红色,CD31和ZO-1共表达呈黄色。统计数据分析结果如图16所示,依帕司他治疗组缺血脑组织中CD31和ZO-1共表达的平均荧光强度明显高于正常组,具有统计差异(**P<0.01)。The expression of ZO-1 in ischemic cerebral hemisphere microvascular endothelial cells was detected by immunofluorescence staining. The results are shown in Figure 15. CD31-labeled cerebral microvascular endothelial cells were green, ZO-1 staining was red, and the co-expression of CD31 and ZO-1 was yellow. Statistical data analysis results are shown in Figure 16, the average fluorescence intensity of CD31 and ZO-1 co-expression in the ischemic brain tissue of the epalrestat treatment group was significantly higher than that of the normal group, with a statistical difference (**P<0.01).
以上结果说明依帕司他能够增加缺血脑组织中微血管内皮细胞上TJs:Occludin和ZO-1的表达水平。The above results indicate that epalrestat can increase the expression levels of TJs: Occludin and ZO-1 on microvascular endothelial cells in ischemic brain tissue.
通过免疫荧光染色检测缺血脑半球星形胶质细胞周围Occludin的情况,结果如图17所示,GFAP标记星形胶质细胞呈绿色,Occludin染色呈红色,细胞核呈蓝色。统计数据分析结果如图18所示,依帕司他治疗组缺血脑组织中GFAP周围Occludin表达的平均荧光强度明显高于正常组,具有统计差异(***P<0.001)。The Occludin around the astrocytes in the ischemic brain hemisphere was detected by immunofluorescence staining, and the results were shown in Figure 17. The GFAP-labeled astrocytes were green, the Occludin staining was red, and the nuclei were blue. Statistical data analysis results are shown in Figure 18, the average fluorescence intensity of Occludin expression around GFAP in the ischemic brain tissue of the epalrestat treatment group was significantly higher than that of the normal group, with a statistical difference (***P<0.001).
通过免疫荧光染色检测缺血脑半球星形胶质细胞周围ZO-1的情况,结果如图19所示,GFAP标记星形胶质细胞呈绿色,ZO-1染色呈红色,细胞核呈蓝色。统计数据分析结果如图20所示,依帕司他治疗组缺血脑组织中GFAP周围ZO-1表达的平均荧光强度明显高于正常组,具有统计差异(**P<0.01)。The situation of ZO-1 around the astrocytes in the ischemic brain hemisphere was detected by immunofluorescence staining, and the results were shown in Figure 19, GFAP-labeled astrocytes were green, ZO-1 staining was red, and the nuclei were blue. Statistical data analysis results are shown in Figure 20, the average fluorescence intensity of ZO-1 expression around GFAP in the ischemic brain tissue of the epalrestat treatment group was significantly higher than that of the normal group, with a statistical difference (**P<0.01).
以上结果说明依帕司他能够增加缺血脑组织中星形胶质细胞周围TJs:Occludin和ZO-1的表达水平,减少脑缺血后内皮细胞TJs缺失。The above results show that epalrestat can increase the expression levels of TJs around astrocytes in ischemic brain tissue: Occludin and ZO-1, and reduce the loss of TJs in endothelial cells after cerebral ischemia.
实施例7Example 7
本实施例考察了脑缺血过程中,依帕司他对缺血脑组织中凋亡相关蛋白的影响。This example investigates the effect of epalrestat on apoptosis-related proteins in ischemic brain tissue during cerebral ischemia.
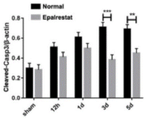
本实施例建模、分组、给药方法均与实施例1相同,脑缺血不同时间取各组脑组织蛋白,通过Western Blot实验检测缺血脑组织中凋亡相关蛋白的表达情况。结果如图21所示,随着脑缺血时间的延长,缺血脑组织中促凋亡蛋白cleaved-casp3和Bax的表达逐渐增加,而抑制凋亡蛋白Bcl2的表达逐渐减少。The modeling, grouping, and administration methods of this example were the same as those in Example 1. The brain tissue proteins of each group were collected at different times of cerebral ischemia, and the expression of apoptosis-related proteins in the ischemic brain tissue was detected by Western Blot experiment. The results are shown in Figure 21. With the prolongation of cerebral ischemia time, the expressions of pro-apoptotic proteins cleaved-casp3 and Bax in ischemic brain tissue gradually increased, while the expression of anti-apoptotic protein Bcl2 gradually decreased.
统计分析如图22显示,与正常组相比较,依帕司他治疗组缺血脑组织中cleaved-casp3的表达量减少,且在脑缺血3d和5d时具有统计差异(**P<0.01,***P<0.001)。统计分析Bcl2和Bax的比值如图23显示,脑缺血3d依帕司他治疗组缺血脑组织中Bcl2/Bax的值明显高于正常组,具有统计差异(*P<0.05)。结果说明依帕司他治疗能够有效降低脑缺血后缺血脑组织中凋亡蛋白的产生。Statistical analysis As shown in Figure 22, compared with the normal group, the expression of cleaved-casp3 in the ischemic brain tissue of the epalrestat treatment group was reduced, and there was a statistical difference between 3d and 5d of cerebral ischemia (**P<0.01 , ***P<0.001). Statistical analysis of the ratio of Bcl2 and Bax as shown in Figure 23, the value of Bcl2/Bax in the ischemic brain tissue of the
实施例8Example 8
本实施例考察了OGD条件下,依帕司他对体外血脑屏障完整性及内皮细胞表达TJs蛋白的影响。This example investigated the effect of epalrestat on the integrity of the blood-brain barrier in vitro and the expression of TJs protein in endothelial cells under OGD conditions.
建立体外氧糖剥夺(OGD)模型模拟脑内缺血条件,将bEnd.3(小鼠脑微血管内皮细胞系)细胞在OGD(Control)和Eps条件下培养不同时间:0h、2h、4h、6h和8h,利用WesternBlot实验检测bEnd.3细胞表达紧密蛋白TJs:Occludin和ZO-1的情况。结果如图24所示,bEnd.3细胞紧密蛋白表达情况;统计分析如图25和图26所示,OGD条件能够减少bEnd.3细胞上ZO-1和Occludin蛋白的表达;OGD作用4h和6h后,依帕司他组中内皮细胞ZO-1和Occludin的蛋白表达量明显高于Control组,具有统计差异(*P<0.05)。An in vitro oxygen-glucose deprivation (OGD) model was established to simulate cerebral ischemia conditions, and bEnd.3 (mouse brain microvascular endothelial cell line) cells were cultured under OGD (Control) and Eps conditions for different time: 0h, 2h, 4h, 6h And 8h, the expression of compact protein TJs: Occludin and ZO-1 in bEnd.3 cells was detected by Western Blot experiment. The results are shown in Figure 24, the expression of compact protein in bEnd.3 cells; statistical analysis is shown in Figure 25 and Figure 26, OGD conditions can reduce the expression of ZO-1 and Occludin proteins on bEnd.3 cells; OGD acts for 4h and 6h After treatment, the protein expressions of ZO-1 and Occludin in endothelial cells in the epalrestat group were significantly higher than those in the Control group, with statistical differences (*P<0.05).
使用bEnd.3细胞建立体外血脑屏障模型,在OGD 4h后检测血脑屏障的通透情况。统计结果如图27显示,与非OGD组相比较,OGD4h后BSA-FITC扩散率升高;使用依帕司他能够有效降低BSA-FITC扩散率,与OGD组比较具有统计学差异(*P<0.05)。The in vitro blood-brain barrier model was established using bEnd.3 cells, and the permeability of the blood-brain barrier was detected after OGD for 4 hours. The statistical results are shown in Figure 27. Compared with the non-OGD group, the BSA-FITC diffusion rate increased after
以上结果说明依帕司他能够上调OGD后bEnd.3细胞上ZO-1和Occludin的表达和降低体外血脑屏障模型的通透率。The above results indicate that epalrestat can up-regulate the expression of ZO-1 and Occludin on bEnd.3 cells after OGD and reduce the permeability of the blood-brain barrier model in vitro.
实施例9Example 9
本实施例考察了OGD条件下,依帕司他对bEnd.3细胞凋亡的影响。This example examines the effect of epalrestat on the apoptosis of bEnd.3 cells under OGD conditions.
建立体外氧糖剥夺OGD模型模拟脑内缺血条件,将bEnd.3(小鼠脑微血管内皮细胞系)细胞在OGD和Eps条件下培养不同时间:0h、2h、4h、6h和8h,利用Western Blot实验检测bEnd.3细胞表达凋亡相关蛋白的变化情况。结果如图28所示,bEnd.3细胞凋亡相关蛋白表达情况;统计分析如图29和30所示,OGD作用下内皮细胞中促凋亡蛋白cleaved-casp3和Bax表达上调,而抑制凋亡蛋白Bcl2表达下调;与OGD(Control)组相比较,OGD+Eps组内皮细胞中cleaved-casp3蛋白的表达减少,且在4h和6h的条件下具有统计学差异(*P<0.05)。同时,统计分析Bcl2/Bax的比值显示,与OGD4h组相比较,OGD+Eps组内皮细胞中Bcl2/Bax的比值显著升高,具有统计学差异(*P<0.05)。An in vitro oxygen-glucose deprivation OGD model was established to simulate cerebral ischemia conditions, and bEnd.3 (mouse brain microvascular endothelial cell line) cells were cultured under OGD and Eps conditions for different time: 0h, 2h, 4h, 6h and 8h, using Western Blot assay was used to detect the changes of expression of apoptosis-related proteins in bEnd.3 cells. The results are shown in Figure 28, the expression of apoptosis-related proteins in bEnd.3 cells; statistical analysis is shown in Figures 29 and 30, the expression of pro-apoptotic proteins cleaved-casp3 and Bax in endothelial cells under the action of OGD is up-regulated, and apoptosis is inhibited The expression of protein Bcl2 was down-regulated; compared with the OGD (Control) group, the expression of cleaved-casp3 protein in the endothelial cells of the OGD+Eps group was reduced, and there was a statistical difference between 4h and 6h (*P<0.05). At the same time, statistical analysis of the Bcl2/Bax ratio showed that compared with the OGD4h group, the Bcl2/Bax ratio in the endothelial cells of the OGD+Eps group was significantly increased, with a statistical difference (*P<0.05).
利用流式细胞术检测OGD 4h后内皮细胞的凋亡情况。结果如图31显示,各组内皮细胞表达PI-PE和AnnexinV-FITC阳性情况;统计分析如图32所示,与OGD组相比较,OGD+Eps组中(AnnexinV-FITC)+/PI-的内皮细胞占比显著降低(*P<0.05)。Flow cytometry was used to detect the apoptosis of endothelial cells after
结果说明OGD条件下,使用依帕司他能够下调bEnd.3细胞中促凋亡蛋白的表达,上调抑制凋亡蛋白的表达,从而减少OGD作用后内皮细胞的凋亡情况。The results showed that under OGD conditions, epalrestat could down-regulate the expression of pro-apoptotic proteins and up-regulate the expression of anti-apoptotic proteins in bEnd.3 cells, thereby reducing the apoptosis of endothelial cells after OGD.
实施例10Example 10
本实施例考察了脑缺血损伤后,依帕司他对脑微血管内皮细胞自噬情况的影响。This example investigates the effect of epalrestat on the autophagy of cerebral microvascular endothelial cells after cerebral ischemic injury.
本实施例建模、分组、给药方法均与实施例1相同,利用免疫荧光标记技术检测缺血脑组织中脑微血管内皮细胞表达自噬相关蛋白:LC3B和Beclin1的情况。各组脑缺血3d后取脑组织制备冰冻切片,LC3B和Beclin1与CD31双染。The modeling, grouping, and administration methods of this example were the same as those in Example 1, and the expression of autophagy-related proteins: LC3B and Beclin1 in cerebral microvascular endothelial cells in ischemic brain tissue was detected by immunofluorescence labeling technology. After 3 days of cerebral ischemia in each group, brain tissue was taken to prepare frozen sections, and LC3B, Beclin1 and CD31 were double-stained.
使用免疫荧光染色检测依帕司他对脑微血管内皮细胞CD31和LC3B的共定位情况,结果如图33显示,CD31标记脑微血管内皮细胞呈绿色,LC3B染色呈红色,CD31和LC3B共表达呈黄色。统计分析结果如图34所示,与Sham组相比较,脑缺血3d缺血脑组织中CD31和LC3B的共表达平均荧光强度升高(*P<0.05,**P<0.01);依帕司他治疗组缺血脑组织中CD31和LC3B的共表达平均荧光强度高于正常组(*P<0.05)。Immunofluorescence staining was used to detect the co-localization of CD31 and LC3B on brain microvascular endothelial cells by epalrestat. As shown in Figure 33, CD31-labeled brain microvascular endothelial cells were green, LC3B staining was red, and co-expression of CD31 and LC3B was yellow. The results of statistical analysis are shown in Figure 34. Compared with the Sham group, the co-expression mean fluorescence intensity of CD31 and LC3B in ischemic brain tissue increased after 3 days of cerebral ischemia (*P<0.05, **P<0.01); The mean fluorescence intensity of co-expression of CD31 and LC3B in the ischemic brain tissue of the sestat treatment group was higher than that of the normal group (*P<0.05).
使用免疫荧光染色检测依帕司他对脑微血管内皮细胞和Beclin1的共定位情况,结果如图35显示,CD31标记脑微血管内皮细胞呈绿色,Beclin1染色呈红色,CD31和Beclin1共表达呈黄色。统计分析结果如图36所示,与Sham组相比较,脑缺血3d缺血脑组织中CD31和Beclin1的共表达平均荧光强度升高(*P<0.05,**P<0.01);依帕司他治疗组缺血脑组织中CD31和Beclin1的共表达平均荧光强度高于正常组(*P<0.05)。结果说明脑缺血3d后,依帕司他能够促进缺血脑组织中内皮细胞自噬。Immunofluorescence staining was used to detect the co-localization of epalrestat on brain microvascular endothelial cells and Beclin1. As shown in Figure 35, CD31-labeled brain microvascular endothelial cells were green, Beclin1 staining was red, and CD31 and Beclin1 co-expression was yellow. The results of statistical analysis are shown in Figure 36. Compared with the Sham group, the co-expression mean fluorescence intensity of CD31 and Beclin1 in
实施例11Example 11
本实施例考察了OGD条件下,依帕司他对bEnd.3细胞自噬的影响。This example investigates the effect of epalrestat on autophagy of bEnd.3 cells under OGD conditions.
建立体外氧糖剥夺OGD模型模拟脑内缺血条件,OGD条件下作用bEnd.3细胞4h后,提取Control组、Eps组、OGD组和OGD+Eps组细胞蛋白,利用Western Blot实验检测各组细胞表达自噬相关蛋白LC3B-I和LC3B-II的情况。An in vitro oxygen-glucose deprivation OGD model was established to simulate cerebral ischemia conditions. After the bEnd.3 cells were exposed to OGD conditions for 4 hours, the cell proteins in Control group, Eps group, OGD group and OGD+Eps group were extracted, and Western Blot was used to detect the cells in each group Expression of autophagy-related proteins LC3B-I and LC3B-II.
结果如图37和图38显示,在OGD条件下内皮细胞中LC3B-II和LC3B-I的比值与非OGD组相比较显著升高(*P<0.05,**P<0.01);与OGD组相比较,依帕司他在OGD条件下能够显著上调LC3B-II和LC3B-I的比值(*P<0.05)。The results are shown in Figure 37 and Figure 38, the ratio of LC3B-II and LC3B-I in endothelial cells under OGD conditions was significantly increased compared with the non-OGD group (*P<0.05, **P<0.01); compared with the OGD group In comparison, epalrestat could significantly up-regulate the ratio of LC3B-II and LC3B-I under OGD conditions (*P<0.05).
使用免疫荧光技术检测各组内皮细胞表达促进自噬相关蛋白:Beclin1和LAMP1的情况,结果如图39所示,CD31标记脑微血管内皮细胞呈红色,Beclin1染色呈绿色,统计数据分析结果如图40所示;如图41所示,CD31标记脑微血管内皮细胞呈红色,LAMP1染色呈绿色,统计数据分析结果如图42所示;由图40和图42所示,与OGD组相比较,依帕司他在OGD条件下能够促进内皮细胞中Beclin1、LAMP-1的表达(*P<0.05,**P<0.01)。Immunofluorescence technology was used to detect the expression of autophagy-related proteins: Beclin1 and LAMP1 in each group of endothelial cells. The results are shown in Figure 39. CD31-labeled brain microvascular endothelial cells were red, and Beclin1 staining was green. The statistical data analysis results were shown in Figure 40 As shown in Figure 41, the CD31-labeled brain microvascular endothelial cells were red, and the LAMP1 staining was green, and the statistical data analysis results were shown in Figure 42; as shown in Figure 40 and Figure 42, compared with the OGD group, Epa Sestat can promote the expression of Beclin1 and LAMP-1 in endothelial cells under OGD conditions (*P<0.05, **P<0.01).
各组内皮细胞中P62蛋白的表达如图43所示,CD31标记脑微血管内皮细胞呈红色,P62染色呈绿色,统计数据分析结果如图44所示,与OGD组相比较,依帕司他在OGD条件下抑制了P62的表达(*P<0.05)。The expression of P62 protein in the endothelial cells of each group is shown in Figure 43. CD31-labeled brain microvascular endothelial cells are red, and P62 staining is green. The results of statistical data analysis are shown in Figure 44. Compared with the OGD group, epalrestat in OGD inhibited the expression of P62 (*P<0.05).
以上结果说明OGD作用4h后,依帕司他能够上调内皮细胞自噬蛋白的表达,促进自噬现象的发生。The above results indicated that after 4 hours of OGD treatment, epalrestat could up-regulate the expression of autophagy proteins in endothelial cells and promote the occurrence of autophagy.
实施例12Example 12
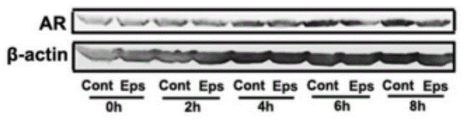
AR即醛糖还原酶,属于醛酮还原酶中的一种,在糖尿病疾病中,血糖升高,糖代谢增加,AR被过度激活,在AR的催化下,多余的葡萄糖通过多元醇途径转化为山梨醇。山梨醇在神经细胞中大量积累,改变渗透压,导致细胞水肿并破裂,引起能量代谢障碍。本实施例考察了依帕司他对缺血后内皮细胞表达AR蛋白的影响。AR is aldose reductase, which belongs to a type of aldehyde and ketone reductase. In diabetes, blood sugar rises, glucose metabolism increases, and AR is overactivated. Under the catalysis of AR, excess glucose is converted into sorbitol. Sorbitol accumulates in large quantities in nerve cells, changes osmotic pressure, causes cell edema and rupture, and causes energy metabolism disorders. This example examines the effect of epalrestat on the expression of AR protein in endothelial cells after ischemia.
本实施例建模、分组、给药方法均与实施例1相同,脑缺血不同时间取各组脑组织,通过Western Blot实验检测缺血脑组织中AR蛋白的表达情况。结果如图45和图46显示,脑缺血3d后依帕司他治疗组缺血脑组织内AR蛋白的表达低于正常组,具有统计学差异(**P<0.01)。The modeling, grouping, and administration methods of this example were the same as those in Example 1. Brain tissues of each group were collected at different times of cerebral ischemia, and the expression of AR protein in the ischemic brain tissues was detected by Western Blot experiment. The results are shown in Figure 45 and Figure 46. After 3 days of cerebral ischemia, the expression of AR protein in the ischemic brain tissue of the epalrestat treatment group was lower than that of the normal group, with a statistical difference (**P<0.01).
利用免疫荧光技术检测脑缺血3d各组内皮细胞与AR的共定位情况,结果如图47显示,CD31标记脑微血管内皮细胞呈绿色,AR染色呈红色,CD31和AR共染呈黄色。缺血脑组织中内皮细胞CD31表达AR;统计分析CD31和AR共染的平均荧光强度如图48显示,在脑缺血3d后,依帕司他治疗组缺血脑组织中CD31和AR共染的平均荧光强度要低于正常组,具有统计学差异(**P<0.01)。The co-localization of endothelial cells and AR in each group of
建立体外氧糖剥夺OGD模型模拟脑内缺血条件,OGD不同时间:0h、2h、4h、6h和8h培养bEnd.3细胞,使用Western Blot实验检测内皮细胞中AR蛋白的变化情况。结果如图49和图50显示,随着OGD作用时间延长内皮细胞中AR蛋白的表达升高;在OGD作用4h和6h时,依帕司他的干预能够显著降低AR在内皮细胞中的表达量,具有统计学差异(*P<0.05,**P<0.01)。An in vitro oxygen-glucose deprivation OGD model was established to simulate cerebral ischemia conditions, and bEnd.3 cells were cultured at different times of OGD: 0h, 2h, 4h, 6h and 8h, and Western Blot was used to detect the changes of AR protein in endothelial cells. The results shown in Figure 49 and Figure 50 show that the expression of AR protein in endothelial cells increases with the prolongation of OGD action time; when OGD is applied for 4h and 6h, the intervention of epalrestat can significantly reduce the expression of AR in endothelial cells , with statistical difference (*P<0.05, **P<0.01).
结果说明脑缺血损伤后,依帕司他能够有效降低内皮细胞上AR蛋白的表达。The results indicated that after cerebral ischemia injury, epalrestat could effectively reduce the expression of AR protein on endothelial cells.
实施例13Example 13
本实施例考察了OGD条件下,依帕司他通过调控AKT/mTOR/AR信号通路对内皮细胞表达TJs蛋白的影响。This example investigated the effect of epalrestat on the expression of TJs protein in endothelial cells by regulating the AKT/mTOR/AR signaling pathway under OGD conditions.
建立体外氧糖剥夺OGD模型模拟脑内缺血条件,OGD条件下作用bEnd.3细胞4h后,提取各组Control组、Eps组、OGD组和OGD+Eps组细胞蛋白,利用Western Blot实验检测各组细胞表达AKT、p-AKT、mTOR和p-mTOR的情况。结果如图51、图52和图53显示,OGD作用下内皮细胞中AKT和mTOR的磷酸化蛋白表达升高;统计分析数据发现与OGD组相比较,Eps+OGD组内皮细胞中p-AKT和p-mTOR蛋白的表达量显著降低,具有统计学差异(*P<0.05,***P<0.001)。An in vitro oxygen-glucose deprivation OGD model was established to simulate cerebral ischemia conditions. After the bEnd.3 cells were exposed to OGD conditions for 4 hours, the cell proteins of Control group, Eps group, OGD group and OGD+Eps group were extracted, and Western Blot was used to detect each group. Group cells express AKT, p-AKT, mTOR and p-mTOR. The results are shown in Figure 51, Figure 52 and Figure 53, the expression of phosphorylated proteins of AKT and mTOR in endothelial cells increased under the action of OGD; statistical analysis data found that compared with the OGD group, p-AKT and mTOR in the endothelial cells of the Eps+OGD group The expression level of p-mTOR protein was significantly decreased with statistical difference (*P<0.05, ***P<0.001).
使用AKT抑制剂比卡鲁胺15μm/L和mTOR抑制剂雷帕霉素10nm/L作为对照比较依帕司他抑制AKT/mTOR信号通路情况。OGD作用4h后提取各组Control组、OGD组、OGD+Eps组、OGD+AKTi组和OGD+mTOR组内皮细胞蛋白,利用Western Blot实验检测各组细胞中AKT、p-AKT、mTOR、p-mTOR和TJs蛋白的表达。结果如图54、图55和图56显示,在OGD条件下依帕司他、比卡鲁胺和雷帕霉素都能降低AKT/mTOR信号通路活化程度;统计分析数据发现与OGD组相比较,Eps+OGD组和AKTi+OGD组内皮细胞中p-AKT和p-mTOR蛋白的表达量降低,具有统计学差异(**P<0.01,***P<0.001);mTORi+OGD组内皮细胞中p-mTOR蛋白的表达量与OGD组相比显著降低(***P<0.001),而p-AKT蛋白的表达量差异不显著(P>0.05)。AKT inhibitor bicalutamide 15μm/L and mTOR inhibitor rapamycin 10nm/L were used as controls to compare epalrestat inhibition of AKT/mTOR signaling pathway. After 4 hours of OGD treatment, endothelial cell proteins were extracted from Control group, OGD group, OGD+Eps group, OGD+AKTi group and OGD+mTOR group, and Western Blot was used to detect AKT, p-AKT, mTOR, p- Expression of mTOR and TJs proteins. The results are shown in Figure 54, Figure 55 and Figure 56. Under OGD conditions, epalrestat, bicalutamide and rapamycin can all reduce the activation degree of the AKT/mTOR signaling pathway; statistical analysis data found that compared with the OGD group , the expressions of p-AKT and p-mTOR proteins in endothelial cells of Eps+OGD group and AKTi+OGD group decreased, with statistical difference (**P<0.01, ***P<0.001); mTORi+OGD group endothelial cells The expression of p-mTOR protein in the cells was significantly lower than that of the OGD group (***P<0.001), while the expression of p-AKT protein was not significantly different (P>0.05).
对各组细胞中ZO-1和Occludin蛋白表达量分析如图57、图58和图59显示,与OGD组相比较,Eps+OGD组、AKTi+OGD组和mTORi+OGD组中内皮细胞中ZO-1和Occludin蛋白表达量增加,具有统计学差异(*P<0.05)。The analysis of ZO-1 and Occludin protein expression in the cells of each group is shown in Figure 57, Figure 58 and Figure 59. Compared with the OGD group, ZO in endothelial cells in the Eps+OGD group, AKTi+OGD group and mTORi+OGD group -1 and Occludin protein expression increased, with statistical difference (*P<0.05).
以上结果说明OGD条件下依帕司他与比卡鲁胺的作用相似,能够通过下调AKT蛋白磷酸化程度,抑制AKT/mTOR信号通路的激活,进而促进内皮细胞中TJs蛋白的表达。The above results indicate that epalrestat and bicalutamide have similar effects under OGD conditions, and can inhibit the activation of AKT/mTOR signaling pathway by down-regulating the phosphorylation of AKT protein, thereby promoting the expression of TJs protein in endothelial cells.
实施例14Example 14
本实施例考察了OGD条件下,依帕司他通过抑制AKT/mTOR/AR信号通路对内皮细胞凋亡和自噬的影响。This example investigated the effect of epalrestat on endothelial cell apoptosis and autophagy by inhibiting the AKT/mTOR/AR signaling pathway under OGD conditions.
建立体外氧糖剥夺OGD模型模拟脑内缺血条件,使用AKT抑制剂比卡鲁胺15μm/L和mTOR抑制剂雷帕霉素10nm/L作为对照比较依帕司他抑制AKT/mTOR信号通路情况。OGD作用4h后提取各组Control组、OGD组、OGD+Eps组、OGD+AKTi组和OGD+mTORi组内皮细胞蛋白,利用Western Blot实验检测细胞中凋亡和自噬相关蛋白的表达情况。结果如图60显示,Western Blot测定了各组细胞中Bax、Bcl2和cleaved-casp3凋亡相关的蛋白。统计分析如图61和图62所示,OGD条件下内皮细胞中cleaved-casp3蛋白表达量与control组相比较显著升高,具有统计学差异(**P<0.01,***P<0.001);与OGD组相比较,OGD+Eps组、OGD+AKTi组和OGD+mTORi组中cleaved-casp3蛋白的表达量降低,具有统计学差异(**P<0.01);而与OGD组相比较,Control组和抑制剂三组中Bcl2和Bax蛋白量的比值显著升高,具有统计学差异(**P<0.01,***P<0.001)。结果说明OGD条件下,依帕司他调控ATK/mTOR/AR信号抑制内皮细胞凋亡的作用与ATK抑制剂和mTOR抑制剂相同。An in vitro oxygen-glucose deprivation OGD model was established to simulate cerebral ischemia conditions, and the AKT inhibitor bicalutamide 15 μm/L and the mTOR inhibitor rapamycin 10 nm/L were used as controls to compare the inhibition of epalrestat on the AKT/mTOR signaling pathway . After 4 hours of OGD treatment, endothelial cell proteins were extracted from Control group, OGD group, OGD+Eps group, OGD+AKTi group and OGD+mTORi group, and Western Blot was used to detect the expression of apoptosis and autophagy-related proteins in cells. The results are shown in Figure 60. Western Blot was used to detect Bax, Bcl2 and cleaved-casp3 apoptosis-related proteins in the cells of each group. Statistical analysis As shown in Figure 61 and Figure 62, the expression of cleaved-casp3 protein in endothelial cells under OGD conditions was significantly higher than that in the control group, with a statistical difference (**P<0.01, ***P<0.001) ;Compared with OGD group, the expression of cleaved-casp3 protein in OGD+Eps group, OGD+AKTi group and OGD+mTORi group decreased, with statistical difference (**P<0.01); and compared with OGD group, The ratio of Bcl2 and Bax protein in the control group and the three inhibitor groups was significantly increased, with statistical difference (**P<0.01, ***P<0.001). The results indicated that under the condition of OGD, epalrestat regulates the ATK/mTOR/AR signal to inhibit the apoptosis of endothelial cells, which is the same as ATK inhibitors and mTOR inhibitors.
通过Western Blot测定各组细胞中LC3BI、LC3BII和Beclin-1自噬相关的蛋白,结果如图63所示,统计分析结果如图64和图65显示,OGD条件下内皮细胞中LC3BII/LC3BI和Beclin-1蛋白表达量与control组相比较显著升高,具有统计学差异(**P<0.01,***P<0.001);与OGD组相比较,OGD+Eps组、OGD+AKTi组和OGD+mTORi组中LC3BII/LC3BI和Beclin-1蛋白的表达量增加,具有统计学差异(***P<0.001);结果说明OGD条件下,依帕司他调控ATK/mTOR/AR信号通过促进内皮细胞自噬的作用与ATK抑制剂和mTOR抑制剂相同。The autophagy-related proteins of LC3BI, LC3BII and Beclin-1 in each group of cells were measured by Western Blot, the results are shown in Figure 63, and the statistical analysis results are shown in Figure 64 and Figure 65, LC3BII/LC3BI and Beclin-1 in endothelial cells under OGD conditions Compared with the control group, the expression of -1 protein was significantly increased, with a statistical difference (**P<0.01, ***P<0.001); compared with the OGD group, OGD+Eps group, OGD+AKTi group and OGD The expressions of LC3BII/LC3BI and Beclin-1 proteins in the +mTORi group were significantly increased (***P<0.001); the results indicated that under OGD conditions, epalrestat regulates ATK/mTOR/AR signaling by promoting endothelial The role of autophagy is the same as that of ATK inhibitors and mTOR inhibitors.
Claims (8)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111346876.8A CN113995751B (en) | 2021-11-15 | 2021-11-15 | Application of epalrestat in the preparation of medicine for preventing and treating ischemic stroke |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111346876.8A CN113995751B (en) | 2021-11-15 | 2021-11-15 | Application of epalrestat in the preparation of medicine for preventing and treating ischemic stroke |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN113995751A CN113995751A (en) | 2022-02-01 |
| CN113995751B true CN113995751B (en) | 2022-11-01 |
Family
ID=79929066
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202111346876.8A Active CN113995751B (en) | 2021-11-15 | 2021-11-15 | Application of epalrestat in the preparation of medicine for preventing and treating ischemic stroke |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113995751B (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN116763771B (en) * | 2023-07-12 | 2025-06-03 | 中国药科大学 | Application of bicalutamide in the preparation of drugs for preventing and treating cerebrovascular diseases |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107837271A (en) * | 2017-11-07 | 2018-03-27 | 中国药科大学 | Epalrestat is preparing the application in treating medicine for treating diabetic nephropathy |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9132191B2 (en) * | 2011-06-08 | 2015-09-15 | Yale University | Compositions and methods of preventing or ameliorating abnormal thrombus formation and cardiovascular disease |
-
2021
- 2021-11-15 CN CN202111346876.8A patent/CN113995751B/en active Active
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107837271A (en) * | 2017-11-07 | 2018-03-27 | 中国药科大学 | Epalrestat is preparing the application in treating medicine for treating diabetic nephropathy |
Non-Patent Citations (1)
| Title |
|---|
| Excess salt intake promotes M1 microglia polarization via a p38/MAPK/ARdependent pathway after cerebral ischemia in mice;Tongshuai Zhang等;《International Immunopharmacology》;20200430;第81卷;106176 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN113995751A (en) | 2022-02-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN109939102B (en) | A kind of pharmaceutical composition comprising butylphthalide and borneol and application thereof | |
| CN113995751B (en) | Application of epalrestat in the preparation of medicine for preventing and treating ischemic stroke | |
| US7470666B2 (en) | Use of Ulinastatin and its pharmaceutical composition for treating severe acute respiratory syndrome | |
| CN113827587A (en) | Application of salvianolic acid A in the preparation of drugs for preventing thrombotic cerebral ischemia | |
| Gordon et al. | Induction of Labor After Intrauterine Fetal Death A Comparison Between Prostaglandin E2 and Oxytocin | |
| CN113185595B (en) | Protein with effects of inhibiting angiogenesis and inflammatory reaction activity and preparation method thereof | |
| CN116514719A (en) | A compound SD82-170 for treating cerebrovascular diseases | |
| CN108295082A (en) | Application of the trifloroside in preventing cerebral ischemia re-pouring injured drug | |
| CN101375847A (en) | Medicament composition with dehydration and diuresis functions | |
| WO2022165794A2 (en) | Method for inhibiting tumor stem cells, and method for regulating tumor blood vessel normalization | |
| Wedeen et al. | Angiotensin II in the treatment of shock | |
| CN109091667B (en) | Application of human urinary kallidinogenase in preparing medicine for treating migraine and composition thereof | |
| Wang et al. | Xanthohumol promotes neuronal and behavioral recovery by suppressing inflammatory response and apoptosis in a rat model of intracerebral hemorrhage | |
| CN118846067B (en) | Medicament for preventing or treating scar pregnancy | |
| CN113694066B (en) | Application of trigonin C in preparation of medicine for treating ischemic stroke | |
| CN112209920B (en) | A kind of oxygen-containing heterocyclic compound for treating cerebral apoplexy | |
| CN120000671A (en) | Application of Poliumoside in the treatment of stroke | |
| CN110585210B (en) | Application of sparganium stoloniferum A in preparation of anti-ischemic cerebral apoplexy medicines | |
| CN100457106C (en) | Use of 1-deoxynojirimycin for preparing diabete and disney disease drug | |
| CN102784133B (en) | Application of salvianic acid A in prevention and/or treatment of brain microvascular thromboembolic diseases | |
| CN117562901A (en) | Traditional Chinese medicine monomer for improving heart ischemia by promoting angiogenesis and application thereof | |
| Grotenhuis et al. | The treatment of cerebral arterial spasm with nimodipine | |
| CN101164558A (en) | Clot-dispersing pain-relieving medicinal composition and preparation method and application thereof | |
| CN111228284A (en) | Application of hesperidin in drugs for preventing liver ischemia-reperfusion injury | |
| JP2024536531A (en) | Use of tetramethylpyrazine nitrone derivatives in the treatment of ischemic stroke - Patent Application 2007023333 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |