CN113980143B - Chimeric antigen receptor, chimeric antigen receptor T cell targeting CD276, preparation method and pharmaceutical application - Google Patents
Chimeric antigen receptor, chimeric antigen receptor T cell targeting CD276, preparation method and pharmaceutical application Download PDFInfo
- Publication number
- CN113980143B CN113980143B CN202111290880.7A CN202111290880A CN113980143B CN 113980143 B CN113980143 B CN 113980143B CN 202111290880 A CN202111290880 A CN 202111290880A CN 113980143 B CN113980143 B CN 113980143B
- Authority
- CN
- China
- Prior art keywords
- antigen receptor
- chimeric antigen
- targeting
- seq
- gly
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/2827—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against B7 molecules, e.g. CD80, CD86
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/0005—Vertebrate antigens
- A61K39/0011—Cancer antigens
- A61K39/001102—Receptors, cell surface antigens or cell surface determinants
- A61K39/001111—Immunoglobulin superfamily
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/70503—Immunoglobulin superfamily
- C07K14/7051—T-cell receptor (TcR)-CD3 complex
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/70578—NGF-receptor/TNF-receptor superfamily, e.g. CD27, CD30, CD40, CD95
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/79—Vectors or expression systems specially adapted for eukaryotic hosts
- C12N15/85—Vectors or expression systems specially adapted for eukaryotic hosts for animal cells
- C12N15/86—Viral vectors
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0634—Cells from the blood or the immune system
- C12N5/0636—T lymphocytes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/51—Medicinal preparations containing antigens or antibodies comprising whole cells, viruses or DNA/RNA
- A61K2039/515—Animal cells
- A61K2039/5158—Antigen-pulsed cells, e.g. T-cells
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/60—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments
- C07K2317/62—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments comprising only variable region components
- C07K2317/622—Single chain antibody (scFv)
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/01—Fusion polypeptide containing a localisation/targetting motif
- C07K2319/02—Fusion polypeptide containing a localisation/targetting motif containing a signal sequence
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/01—Fusion polypeptide containing a localisation/targetting motif
- C07K2319/03—Fusion polypeptide containing a localisation/targetting motif containing a transmembrane segment
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/33—Fusion polypeptide fusions for targeting to specific cell types, e.g. tissue specific targeting, targeting of a bacterial subspecies
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2510/00—Genetically modified cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2740/00—Reverse transcribing RNA viruses
- C12N2740/00011—Details
- C12N2740/10011—Retroviridae
- C12N2740/15011—Lentivirus, not HIV, e.g. FIV, SIV
- C12N2740/15041—Use of virus, viral particle or viral elements as a vector
- C12N2740/15043—Use of virus, viral particle or viral elements as a vector viral genome or elements thereof as genetic vector
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2800/00—Nucleic acids vectors
- C12N2800/10—Plasmid DNA
- C12N2800/106—Plasmid DNA for vertebrates
- C12N2800/107—Plasmid DNA for vertebrates for mammalian
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Organic Chemistry (AREA)
- Genetics & Genomics (AREA)
- General Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Zoology (AREA)
- Biochemistry (AREA)
- Biomedical Technology (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Wood Science & Technology (AREA)
- Cell Biology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biotechnology (AREA)
- Biophysics (AREA)
- General Engineering & Computer Science (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Microbiology (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Gastroenterology & Hepatology (AREA)
- Public Health (AREA)
- Toxicology (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Mycology (AREA)
- Oncology (AREA)
- Plant Pathology (AREA)
- Hematology (AREA)
- Virology (AREA)
- Physics & Mathematics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Peptides Or Proteins (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
本发明公开了一种靶向CD276的嵌合抗原受体、嵌合抗原受体T细胞及制备方法和制药应用,属于生物医药技术领域。本发明公开的靶向CD276的嵌合抗原受体T细胞可以靶向表达CD276的肿瘤细胞,激活T细胞发挥细胞免疫作用,高效且特异性地杀伤CD276阳性肿瘤细胞,更好地维持嵌合抗原受体T细胞的活力和杀伤力,是有潜力的根治肝细胞癌的免疫细胞疗法。本发明经过体外功能测试,靶向CD276的嵌合抗原受体T细胞对CD276阳性肝癌细胞系有杀伤活性。
The invention discloses a chimeric antigen receptor targeting CD276, a chimeric antigen receptor T cell, a preparation method and a pharmaceutical application, and belongs to the technical field of biomedicine. The chimeric antigen receptor T cells targeting CD276 disclosed in the present invention can target tumor cells expressing CD276, activate T cells to exert cellular immunity, efficiently and specifically kill CD276-positive tumor cells, and better maintain chimeric antigens The vitality and lethality of recipient T cells are potential immune cell therapy for hepatocellular carcinoma. The present invention has been tested in vitro, and the CD276-targeted chimeric antigen receptor T cells have killing activity on CD276-positive liver cancer cell lines.
Description
技术领域technical field
本发明属于生物医药技术领域,涉及一种靶向CD276的嵌合抗原受体、嵌合抗原受体T细胞及制备方法和制药应用。The invention belongs to the technical field of biomedicine, and relates to a CD276-targeted chimeric antigen receptor, chimeric antigen receptor T cells, a preparation method and a pharmaceutical application.
背景技术Background technique
肝细胞癌是严重威胁人类健康的恶性肿瘤,占所有原发性肝癌的90%,是全球第二大致死性癌症。目前针对肝细胞癌的治疗方法比如化疗、放疗、肝切除术、消融疗法、靶向疗法如索拉菲尼、抗体疗法如免疫检查点抑制剂、血管内皮生长因子受体抑制剂,然而这些疗法的疗效都是不够的(Lee,Y.H.,et al.,Combinational Immunotherapy forHepatocellular Carcinoma:Radiotherapy,Immune Checkpoint Blockade andBeyond.Frontiers in Immunology,2020.11.),对病人和病人的家庭造成了难以挽回的伤害。因此迫切需要的新的治疗肝细胞癌的药物。Hepatocellular carcinoma is a malignant tumor that seriously threatens human health, accounting for 90% of all primary liver cancers, and is the second most deadly cancer in the world. Current treatments for hepatocellular carcinoma such as chemotherapy, radiotherapy, hepatectomy, ablation therapy, targeted therapy such as sorafenib, antibody therapy such as immune checkpoint inhibitors, vascular endothelial growth factor receptor inhibitors, however, these therapies The curative effect is not enough (Lee, Y.H., et al., Combinational Immunotherapy for Hepatocellular Carcinoma: Radiotherapy, Immune Checkpoint Blockade and Beyond. Frontiers in Immunology, 2020.11.), causing irreparable damage to the patient and the patient's family. Therefore, new drugs for the treatment of HCC are urgently needed.
嵌合抗原受体T细胞(CAR-T)疗法是新型的免疫细胞治疗方法,具有特异性且高效的杀伤表达相应抗原的肿瘤细胞的功能,它通过基因工程手段使嵌合抗原受体表达在T细胞表面,通过在体外制备和扩增嵌合抗原受体T细胞,然后回输至人体,激活自身免疫系统,发挥对肿瘤细胞的高效杀伤(1.Rafiq,S.,C.S.Hackett,and R.J.Brentjens,Engineeringstrategies to overcome the current roadblocks in CAR T cell therapy.NatureReviews Clinical Oncology,2020.17(3):p.147-167.;2.Benmebarek,M.R.,et al.,Killing Mechanisms of Chimeric Antigen Receptor(CAR)T Cells.InternationalJournal of Molecular Sciences,2019.20(6).)嵌合抗原受体T细胞技术被认为是最有可能攻克癌症的疗法,并且靶向CD19的CAR-T细胞在血液肿瘤中产生了良好的效果。目前全球已有6款CAR-T药物获批上市,展现了CAR-T细胞治疗在治疗癌症中的巨大应用前景。Chimeric antigen receptor T cell (CAR-T) therapy is a new type of immune cell therapy, which has the function of specifically and efficiently killing tumor cells expressing corresponding antigens. On the surface of T cells, chimeric antigen receptor T cells are prepared and expanded in vitro, and then reinfused into the human body to activate the autoimmune system and effectively kill tumor cells (1. Rafiq, S., C.S. Hackett, and R.J. Brentjens, Engineering strategies to overcome the current roadblocks in CAR T cell therapy. Nature Reviews Clinical Oncology, 2020.17(3): p.147-167.; 2. Benmebarek, M.R., et al., Killing Mechanisms of Chimeric Antigen Receptor(CAR)T Cells.International Journal of Molecular Sciences, 2019.20(6).) Chimeric antigen receptor T cell technology is considered to be the most likely therapy to overcome cancer, and CAR-T cells targeting CD19 have produced good results in blood tumors . At present, 6 CAR-T drugs have been approved for marketing in the world, showing the great application prospects of CAR-T cell therapy in the treatment of cancer.
CD276又称为B7-H3,是属于B7家族的一类跨膜蛋白。CD276在多种癌症中高表达,包括肺腺癌、脑胶质瘤、神经母细胞瘤、胰腺癌和卵巢癌等,在正常组织中几乎不表达。靶向CD276的单克隆抗体在多种肿瘤模型中介导肿瘤的有效清除。CD276在肝细胞癌高表达并且与肝癌的进展和肿瘤病人的不良预后相关。这提示,CD276可以作为一个治疗肝细胞癌的有潜力靶点。CD276, also known as B7-H3, is a type of transmembrane protein belonging to the B7 family. CD276 is highly expressed in a variety of cancers, including lung adenocarcinoma, glioma, neuroblastoma, pancreatic cancer, and ovarian cancer, and is almost not expressed in normal tissues. Monoclonal antibodies targeting CD276 mediate efficient tumor clearance in various tumor models. CD276 is highly expressed in HCC and is associated with the progression of HCC and the poor prognosis of tumor patients. This suggests that CD276 can be used as a potential target for the treatment of HCC.
目前,靶向CD276的CAR-T细胞对肝细胞癌和其它高表达CD276的恶性肿瘤的杀伤活性研究有限。Currently, studies on the killing activity of CAR-T cells targeting CD276 on hepatocellular carcinoma and other malignant tumors with high expression of CD276 are limited.
发明内容Contents of the invention
为了克服上述现有技术的缺点,本发明的目的在于提供一种靶向CD276的嵌合抗原受体、嵌合抗原受体T细胞及制备方法和制药应用。In order to overcome the above-mentioned shortcomings of the prior art, the object of the present invention is to provide a CD276-targeting chimeric antigen receptor, chimeric antigen receptor T cells, preparation method and pharmaceutical application.
为了达到上述目的,本发明采用以下技术方案予以实现:In order to achieve the above object, the present invention adopts the following technical solutions to achieve:
本发明的内容之一在于,提供一种靶向CD276的嵌合抗原受体,所述靶向CD276的嵌合抗原受体包括如SEQ ID NO:1所示的氨基酸序列。One of the contents of the present invention is to provide a CD276-targeting chimeric antigen receptor, said CD276-targeting chimeric antigen receptor comprising the amino acid sequence shown in SEQ ID NO:1.
优选地,所述靶向CD276的嵌合抗原受体的编码基因包括如SEQ ID NO:2所示的核苷酸序列。Preferably, the coding gene of the chimeric antigen receptor targeting CD276 includes the nucleotide sequence shown in SEQ ID NO:2.
优选地,所述靶向CD276的嵌合抗原受体包括从氨基端到羧基端依次为靶向CD276的单链抗体、胞外铰链区、跨膜区和胞内结构区的氨基酸序列。Preferably, the CD276-targeting chimeric antigen receptor comprises the amino acid sequence of a CD276-targeting single-chain antibody, an extracellular hinge region, a transmembrane region and an intracellular structural region from the amino terminal to the carboxyl terminal.
进一步优选地,所述靶向CD276的单链抗体的氨基酸序列包括如SEQ ID NO:3所示的氨基酸序列。Further preferably, the amino acid sequence of the single-chain antibody targeting CD276 includes the amino acid sequence shown in SEQ ID NO:3.
更进一步优选地,所述靶向CD276的单链抗体的编码基因包括如SEQ ID NO:4所示的核苷酸序列。Still further preferably, the gene encoding the single-chain antibody targeting CD276 includes the nucleotide sequence shown in SEQ ID NO:4.
进一步优选地,所述胞外铰链区为CD8α铰链区。Further preferably, the extracellular hinge region is a CD8α hinge region.
更进一步优选地,所述CD8α铰链区的氨基酸序列包括如SEQ ID NO:5所示的氨基酸序列。Still further preferably, the amino acid sequence of the CD8α hinge region includes the amino acid sequence shown in SEQ ID NO:5.
更进一步优选地,所述CD8α铰链区的编码基因包括如SEQ ID NO:6所示的核苷酸序列。Still further preferably, the gene encoding the hinge region of CD8α includes the nucleotide sequence shown in SEQ ID NO:6.
当然,胞外铰链区还可以使用CD28铰链区、IgG1铰链区或IgG4铰链区。Of course, the extracellular hinge region can also use the CD28 hinge region, IgG1 hinge region or IgG4 hinge region.
进一步优选地,所述跨膜区为CD8α跨膜区。Further preferably, the transmembrane region is CD8α transmembrane region.
更进一步优选地,所述CD8α跨膜区的氨基酸序列包括如SEQ ID NO:7所示的氨基酸序列。Still further preferably, the amino acid sequence of the CD8α transmembrane region includes the amino acid sequence shown in SEQ ID NO:7.
更进一步优选地,所述CD8α跨膜区的编码基因包括如SEQ ID NO:8所示的核苷酸序列。Still further preferably, the gene encoding the CD8α transmembrane region includes a nucleotide sequence as shown in SEQ ID NO:8.
当然,跨膜区还可以使用CD4跨膜区、CD28跨膜区、CD3ζ跨膜区或ICOS的跨膜区。Of course, the transmembrane region can also use CD4 transmembrane region, CD28 transmembrane region, CD3ζ transmembrane region or ICOS transmembrane region.
进一步优选地,所述胞内结构区由4-1BB共刺激结构区和CD3ζ信号结构区依次串联构成。Further preferably, the intracellular structural region is composed of a 4-1BB co-stimulatory structural region and a CD3ζ signaling structural region sequentially in series.
更进一步优选地,所述4-1BB共刺激结构区的氨基酸序列包括如SEQ ID NO:9所示的氨基酸序列。Still further preferably, the amino acid sequence of the 4-1BB co-stimulatory structural region includes the amino acid sequence shown in SEQ ID NO:9.
更进一步优选地,所述4-1BB共刺激结构区的编码基因包括如SEQ ID NO:10所示的核苷酸序列。Still further preferably, the coding gene of the 4-1BB co-stimulatory structural region includes the nucleotide sequence shown in SEQ ID NO:10.
进一步优选地,所述CD3ζ信号结构区的氨基酸序列包括如SEQ ID NO:11所示的氨基酸序列。Further preferably, the amino acid sequence of the CD3ζ signal domain includes the amino acid sequence shown in SEQ ID NO: 11.
更进一步优选地,CD3ζ信号结构区的编码基因包括如SEQ ID NO:12所示的核苷酸序列。Still further preferably, the coding gene of the CD3ζ signal structural region includes the nucleotide sequence shown in SEQ ID NO: 12.
当然,共刺激结构区还可以使用CD28、OX40、ICOS或CD27的胞内区。Of course, the co-stimulatory domain can also use the intracellular domain of CD28, OX40, ICOS or CD27.
本发明的内容之二在于提供一种靶向CD276的嵌合抗原受体T细胞,包括上述的靶向CD276的嵌合抗原受体;该靶向CD276的嵌合抗原受体的氨基酸序列包括如SEQ ID NO:1所示的氨基酸序列。The second content of the present invention is to provide a chimeric antigen receptor T cell targeting CD276, including the above-mentioned chimeric antigen receptor targeting CD276; the amino acid sequence of the chimeric antigen receptor targeting CD276 includes: The amino acid sequence shown in SEQ ID NO:1.
进一步地,所述靶向CD276的嵌合抗原受体CAR-CD276的编码基因包括如SEQ IDNO:2所示的核苷酸序列。Further, the coding gene of the chimeric antigen receptor CAR-CD276 targeting CD276 includes the nucleotide sequence shown in SEQ ID NO:2.
本发明的内容之三在于提供一种靶向CD276的嵌合抗原受体T细胞的制备方法,包括以下步骤:The third content of the present invention is to provide a method for preparing chimeric antigen receptor T cells targeting CD276, comprising the following steps:
1)提供靶向CD276的嵌合抗原受体CAR-CD276的编码基因,所述的靶向CD276的嵌合抗原体CAR-CD276的编码基因如SEQ ID NO:2所示的核苷酸序列。1) Provide the coding gene of the chimeric antigen receptor CAR-CD276 targeting CD276, the coding gene of the chimeric antigen receptor CAR-CD276 targeting CD276 has the nucleotide sequence shown in SEQ ID NO:2.
2)将所述CAR-CD276的编码基因插入到pWPXLd载体中,得到pWPXLd-CAR-CD276重组质粒;2) inserting the gene encoding CAR-CD276 into the pWPXLd vector to obtain the pWPXLd-CAR-CD276 recombinant plasmid;
3)将所述pWPXLd-CAR-CD276重组质粒与包膜质粒、包装质粒共转染宿主细胞,得到重组慢病毒;3) co-transfecting the host cell with the pWPXLd-CAR-CD276 recombinant plasmid, the envelope plasmid, and the packaging plasmid to obtain a recombinant lentivirus;
4)将所述重组慢病毒转染CD3阳性T淋巴细胞,经分离获得靶向CD276的嵌合抗原受体T细胞。4) Transfect CD3-positive T lymphocytes with the recombinant lentivirus, and obtain chimeric antigen receptor T cells targeting CD276 through separation.
进一步地,所述包膜质粒为PMD2G,所述包装质粒为psPAX2,所述宿主细胞为HEK293T细胞。Further, the envelope plasmid is PMD2G, the packaging plasmid is psPAX2, and the host cell is HEK293T cells.
进一步地,步骤4)中,所述CD3阳性T淋巴细胞是从人外周血单个核细胞中分离获得。Further, in step 4), the CD3-positive T lymphocytes are isolated from human peripheral blood mononuclear cells.
更进一步地,所述人源外周血单个核细胞来源于自体静脉血、自体骨髓、脐带血和胎盘血等。Furthermore, the human-derived peripheral blood mononuclear cells are derived from autologous venous blood, autologous bone marrow, umbilical cord blood, placental blood, and the like.
本发明的内容之四在于提供一种重组病毒载体,所述重组病毒载体包括如上述的靶向CD276的嵌合抗原受体CAR-CD276的编码基因。The fourth content of the present invention is to provide a recombinant virus vector, which includes the coding gene of the CD276-targeting chimeric antigen receptor CAR-CD276 as described above.
该重组病毒载体包含靶向CD276的嵌合抗原受体CAR-CD276的编码基因,该编码基因的核苷酸序列如SEQ ID NO:2或SEQ ID NO:13所示;The recombinant virus vector comprises the coding gene of the chimeric antigen receptor CAR-CD276 targeting CD276, the nucleotide sequence of the coding gene is shown in SEQ ID NO: 2 or SEQ ID NO: 13;
如SEQ ID NO:13所示的核苷酸序列与如SEQ ID NO:2所示的核苷酸序列相比,多了信号肽的编码基因。所述信号肽的编码基因可以较好地指导所述嵌合抗原受体CAR-CD276表达到细胞表面。Compared with the nucleotide sequence shown in SEQ ID NO: 2, the nucleotide sequence shown in SEQ ID NO: 13 has more coding genes for signal peptides. The gene encoding the signal peptide can better guide the expression of the chimeric antigen receptor CAR-CD276 to the cell surface.
优选地,所述靶向CD276的嵌合抗原受体CAR-CD276的编码基因,包括从5’端到3’端依次连接的CD8α信号肽的编码基因、靶向CD276的单链抗体的编码基因、CD8α胞外铰链区的编码基因、CD8α跨膜区的编码基因和由4-1BB共刺激结构区和CD3ζ信号结构区依次串联构成的胞内信号区的编码基因。Preferably, the coding gene of the chimeric antigen receptor CAR-CD276 targeting CD276 includes the coding gene of the CD8α signal peptide and the coding gene of the CD276-targeting single-chain antibody sequentially connected from the 5' end to the 3' end , the coding gene of the CD8α extracellular hinge region, the coding gene of the CD8α transmembrane region and the coding gene of the intracellular signal region composed of the 4-1BB co-stimulatory structure region and the CD3ζ signal structure region in sequence.
进一步优选地,所述CAR-CD276的氨基酸序列包括如SEQ ID NO:14所示的氨基酸序列。Further preferably, the amino acid sequence of the CAR-CD276 includes the amino acid sequence shown in SEQ ID NO: 14.
进一步优选地,所述CD8α信号肽的编码基因包括如SEQ ID NO:15所示的核苷酸序列。Further preferably, the gene encoding the CD8α signal peptide includes the nucleotide sequence shown in SEQ ID NO:15.
进一步优选地,所述CD8α信号肽的氨基酸序列包括如SEQ ID NO:16所示的氨基酸序列。Further preferably, the amino acid sequence of the CD8α signal peptide includes the amino acid sequence shown in SEQ ID NO: 16.
进一步优选地,所述靶向CD276的单链抗体的编码基因包括如SEQ ID NO:4所示的核苷酸序列、所述CD8α胞外铰链区的编码基因包括如SEQ ID NO:6所示的核苷酸序列、所述CD8α跨膜区的编码基因包括如SEQ ID NO:8所示的核苷酸序列、所述4-1BB共刺激结构区的编码基因包括如SEQ ID NO:10所示的核苷酸序列、所述CD3ζ信号结构区的编码基因包括如SEQ ID NO:12所示的核苷酸序列。Further preferably, the coding gene of the single-chain antibody targeting CD276 includes the nucleotide sequence shown in SEQ ID NO: 4, and the coding gene of the CD8α extracellular hinge region includes the nucleotide sequence shown in SEQ ID NO: 6. The nucleotide sequence of the nucleotide sequence, the coding gene of the CD8α transmembrane region includes the nucleotide sequence shown in SEQ ID NO: 8, the coding gene of the 4-1BB co-stimulatory structural region includes the coding gene shown in SEQ ID NO: 10 The nucleotide sequence shown, the coding gene of the CD3ζ signal structure region include the nucleotide sequence shown in SEQ ID NO:12.
所述病毒载体为pWPXLd慢病毒载体。The viral vector is pWPXLd lentiviral vector.
本发明的内容之五在于提供上述的靶向CD276的嵌合抗原受体、靶向CD276的嵌合抗原受体T细胞或重组病毒载体在制备治疗肝癌的药物中的应用。The fifth content of the present invention is to provide the above CD276-targeting chimeric antigen receptor, CD276-targeting chimeric antigen receptor T cell or recombinant virus vector in the preparation of drugs for treating liver cancer.
优选地,该药物对表达CD276的阳性肿瘤细胞杀伤活性强。Preferably, the drug has strong killing activity on positive tumor cells expressing CD276.
本发明的目的之六在于提供一种治疗肝细胞癌的药物,该药物包括上述的靶向CD276的嵌合抗原受体、靶向CD276的嵌合抗原受体T细胞或重组病毒载体。The sixth object of the present invention is to provide a drug for treating hepatocellular carcinoma, which includes the above CD276-targeted chimeric antigen receptor, CD276-targeted chimeric antigen receptor T cell or recombinant virus vector.
当然,所述靶向CD276的嵌合抗原受体T细胞也适用于用于其它高表达CD276的恶性肿瘤的治疗。Of course, the chimeric antigen receptor T cells targeting CD276 are also applicable to the treatment of other malignant tumors that highly express CD276.
与现有技术相比,本发明具有以下有益效果:Compared with the prior art, the present invention has the following beneficial effects:
本发明提供了一种靶向CD276的嵌合抗原受体,以及包括靶向CD276的嵌合抗原受体T细胞。靶向CD276的嵌合抗原受体T细胞可以靶向表达CD276的肿瘤细胞,激活T细胞发挥细胞免疫作用,高效且特异性地杀伤CD276阳性肿瘤细胞,更好地维持嵌合抗原受体T细胞的活力和杀伤力,是有潜力的根治肝细胞癌的免疫细胞疗法。The present invention provides a chimeric antigen receptor targeting CD276 and T cells including the chimeric antigen receptor targeting CD276. Chimeric antigen receptor T cells targeting CD276 can target tumor cells expressing CD276, activate T cells to play a role in cellular immunity, efficiently and specifically kill CD276-positive tumor cells, and better maintain chimeric antigen receptor T cells Its vitality and lethality are potential immune cell therapy for hepatocellular carcinoma.
本发明公开的靶向CD276的嵌合抗原受体T细胞以4-1BB为共刺激结构区,使得靶向CD276的嵌合抗原受体T细胞在体内具有更长的持久性,有利于记忆表型T细胞的产生,对表达CD276的阳性肿瘤细胞系有强的杀伤活性。本发明经过体外功能测试,靶向CD276的嵌合抗原受体T细胞对CD276阳性肝癌细胞系有杀伤活性。The chimeric antigen receptor T cells targeting CD276 disclosed in the present invention use 4-1BB as a co-stimulatory structural region, so that the chimeric antigen receptor T cells targeting CD276 have longer persistence in the body, which is beneficial to the memory table The production of type T cells has strong killing activity against positive tumor cell lines expressing CD276. The present invention has been tested in vitro, and the CD276-targeted chimeric antigen receptor T cells have killing activity on CD276-positive liver cancer cell lines.
附图说明Description of drawings
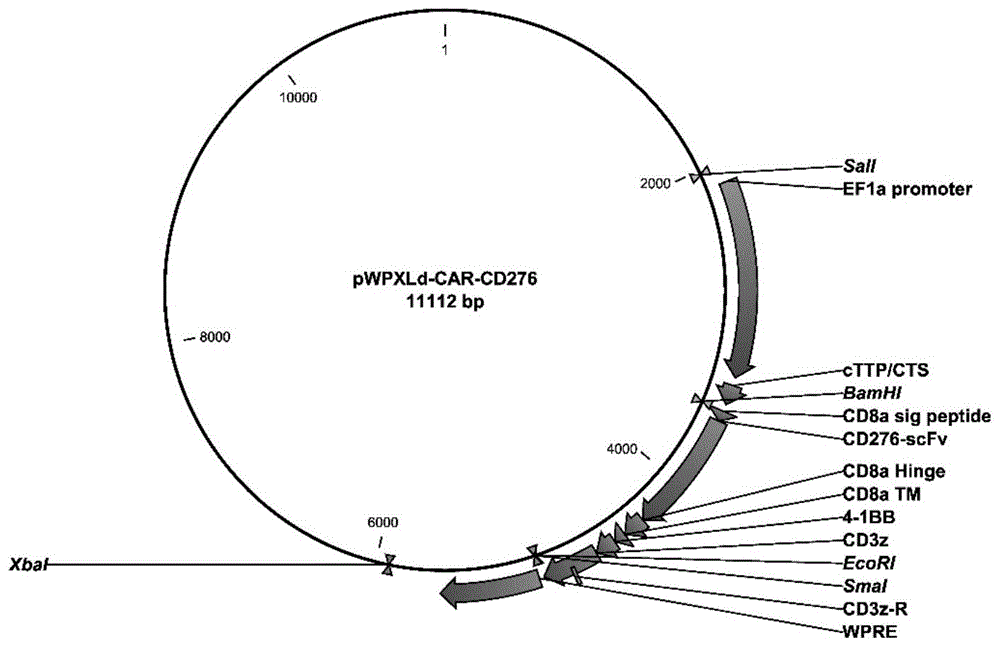
图1为pWPXLd-CAR-CD276重组质粒的质粒图谱;Figure 1 is a plasmid map of the pWPXLd-CAR-CD276 recombinant plasmid;
图2为靶向CD276的嵌合抗原受体在T细胞表面表达结果图;其中,(a)为UTD非转导病毒的T细胞;(b)为CAR-CD276转导病毒的T细胞;Figure 2 is a diagram showing the expression results of chimeric antigen receptors targeting CD276 on the surface of T cells; wherein, (a) is T cells with UTD non-transduced virus; (b) is T cells transduced with CAR-CD276 virus;
图3为靶向CD276的嵌合抗原受体T细胞对肝细胞癌细胞系Huh7的杀伤结果图。Figure 3 is a graph showing the killing results of the hepatocellular carcinoma cell line Huh7 by chimeric antigen receptor T cells targeting CD276.
具体实施方式Detailed ways
为了使本技术领域的人员更好地理解本发明方案,下面将结合本发明实施例中的附图,对本发明实施例中的技术方案进行清楚、完整地描述,显然,所描述的实施例仅仅是本发明一部分的实施例,而不是全部的实施例。基于本发明中的实施例,本领域普通技术人员在没有做出创造性劳动前提下所获得的所有其他实施例,都应当属于本发明保护的范围。In order to enable those skilled in the art to better understand the solutions of the present invention, the following will clearly and completely describe the technical solutions in the embodiments of the present invention in conjunction with the drawings in the embodiments of the present invention. Obviously, the described embodiments are only It is an embodiment of a part of the present invention, but not all embodiments. Based on the embodiments of the present invention, all other embodiments obtained by persons of ordinary skill in the art without making creative efforts shall fall within the protection scope of the present invention.
需要说明的是,本发明的说明书和权利要求书及上述附图中的术语“第一”、“第二”等是用于区别类似的对象,而不必用于描述特定的顺序或先后次序。应该理解这样使用的数据在适当情况下可以互换,以便这里描述的本发明的实施例能够以除了在这里图示或描述的那些以外的顺序实施。此外,术语“包括”和“具有”以及他们的任何变形,意图在于覆盖不排他的包含,例如,包含了一系列步骤或单元的过程、方法、系统、产品或设备不必限于清楚地列出的那些步骤或单元,而是可包括没有清楚地列出的或对于这些过程、方法、产品或设备固有的其它步骤或单元。It should be noted that the terms "first" and "second" in the description and claims of the present invention and the above drawings are used to distinguish similar objects, but not necessarily used to describe a specific sequence or sequence. It is to be understood that the data so used are interchangeable under appropriate circumstances such that the embodiments of the invention described herein can be practiced in sequences other than those illustrated or described herein. Furthermore, the terms "comprising" and "having", as well as any variations thereof, are intended to cover a non-exclusive inclusion, for example, a process, method, system, product or device comprising a sequence of steps or elements is not necessarily limited to the expressly listed instead, may include other steps or elements not explicitly listed or inherent to the process, method, product or apparatus.
下面结合附图对本发明做进一步详细描述:The present invention is described in further detail below in conjunction with accompanying drawing:
一、一种靶向CD276的嵌合抗原受体T细胞的制备方法,包括以下步骤:One, a kind of preparation method of the chimeric antigen receptor T cell targeting CD276, comprises the following steps:
(1)制备靶向CD276的嵌合抗原受体CAR-CD276的基因序列(1) Prepare the gene sequence of chimeric antigen receptor CAR-CD276 targeting CD276
靶向CD276的嵌合抗原受体CAR-CD276结构包括CD8α信号肽、靶向CD276的单链抗体(CD276-scFv)、CD8α铰链区、CD8α跨膜区、4-1BB共刺激结构区和CD3ζ信号结构区的编码基因,所述信号肽的编码基因如SEQ ID NO:15所示,所述靶向CD276的单链抗体的编码基因包括如SEQ ID NO:4所示的核苷酸序列、所述CD8α胞外铰链区的编码基因包括如SEQ IDNO:6所示的核苷酸序列、所述CD8α跨膜区的编码基因包括如SEQ ID NO:8所示的核苷酸序列、所述4-1BB共刺激结构区的编码基因包括如SEQ ID NO:10所示的核苷酸序列、所述CD3ζ信号结构区的编码基因包括如SEQ ID NO:12所示的核苷酸序列。The CD276-targeting chimeric antigen receptor CAR-CD276 structure includes CD8α signal peptide, CD276-targeting single-chain antibody (CD276-scFv), CD8α hinge region, CD8α transmembrane region, 4-1BB co-stimulatory domain and CD3ζ signal The coding gene of the structural region, the coding gene of the signal peptide is shown in SEQ ID NO: 15, and the coding gene of the single-chain antibody targeting CD276 includes the nucleotide sequence shown in SEQ ID NO: 4, the The coding gene of the CD8α extracellular hinge region includes the nucleotide sequence shown in SEQ ID NO:6, the coding gene of the CD8α transmembrane region includes the nucleotide sequence shown in SEQ ID NO:8, the 4 - The coding gene of the costimulatory structural region of 1BB includes the nucleotide sequence shown in SEQ ID NO:10, and the coding gene of the CD3ζ signal structural region includes the nucleotide sequence shown in SEQ ID NO:12.
所述靶向CD276的嵌合抗原受体CAR-CD276结构由江苏金唯智生物技术有限公司进行基因合成,得到靶向CD276的嵌合抗原受体CAR-CD276的编码基因,所述CAR-CD276的编码基因包括如SEQ ID NO:13所示的核苷酸序列。The chimeric antigen receptor CAR-CD276 structure targeting CD276 was gene-synthesized by Jiangsu Jinweizhi Biotechnology Co., Ltd., and the coding gene of the chimeric antigen receptor CAR-CD276 targeting CD276 was obtained. The CAR-CD276 The coding gene includes the nucleotide sequence shown in SEQ ID NO:13.
(2)构建pWPXLd-CAR-CD276重组质粒(2) Construction of pWPXLd-CAR-CD276 recombinant plasmid
将合成的CAR-CD276的编码基因插入到pWPXLd载体的BamH1和EcoR1酶切位点之间,然后转入大肠杆菌感受态细胞DH5α,进行阳性克隆PCR鉴定和测序鉴定。如图1所示为pWPXLd-CAR-CD276重组质粒图谱。The synthetic CAR-CD276 coding gene was inserted between the BamH1 and EcoR1 restriction sites of the pWPXLd vector, and then transformed into Escherichia coli competent cells DH5α for positive clone PCR identification and sequencing identification. Figure 1 shows the pWPXLd-CAR-CD276 recombinant plasmid map.
(3)重组慢病毒构建(3) Construction of recombinant lentivirus
将pWPXLd-CAR-CD276重组质粒、包装质粒psPAX2、包膜质粒pMD2G使用lipofectamine3000转染试剂共转染入培养好的HEK293T细胞。第48h收集含病毒的上清,首先2000rpm室温离心5分钟,取上层上清,然后经0.45μm滤膜过滤,得到的重组慢病毒上清用于T细胞感染。The pWPXLd-CAR-CD276 recombinant plasmid, packaging plasmid psPAX2, and envelope plasmid pMD2G were co-transfected into cultured HEK293T cells using lipofectamine3000 transfection reagent. The virus-containing supernatant was collected at 48 hours, first centrifuged at 2000 rpm for 5 minutes at room temperature, the supernatant was taken, and then filtered through a 0.45 μm filter membrane, and the obtained recombinant lentivirus supernatant was used for T cell infection.
(4)靶向CD276的嵌合抗原受体T细胞的制备(4) Preparation of chimeric antigen receptor T cells targeting CD276
a)PBMC(外周血单个核细胞)的分离a) Isolation of PBMC (peripheral blood mononuclear cells)
PBMC来源于健康志愿者的自体静脉血。PBMC were derived from autologous venous blood of healthy volunteers.
抽取所述健康志愿者血液,使用Ficoll收集外周血单个核细胞,离心分离后取中间层细胞;经PBS洗涤计数后得到PBMC。The blood of the healthy volunteers was extracted, the peripheral blood mononuclear cells were collected by Ficoll, and the middle layer cells were collected after centrifugation; PBMC were obtained after washing and counting with PBS.
b)免疫磁珠法分离抗原特异性T淋巴细胞b) Isolation of antigen-specific T lymphocytes by immunomagnetic bead method
取上述PBMC,加入KBM581培养基,配成细胞悬液;按磁珠与细胞的比例为1:1,加入CD3/CD28免疫磁珠,室温下于摇床进行孵育;采用磁铁对孵育磁珠的细胞进行筛选;去除未吸附的细胞悬液后,加入上述KBM581培养基重悬磁珠-细胞混合物得到CD3阳性T淋巴细胞,继续培养24小时后用于慢病毒感染。Take the above PBMCs, add KBM581 medium to make a cell suspension; add CD3/CD28 immunomagnetic beads according to the ratio of magnetic beads to cells at 1:1, and incubate on a shaker at room temperature; The cells were screened; after removing the unadsorbed cell suspension, the above-mentioned KBM581 medium was added to resuspend the magnetic bead-cell mixture to obtain CD3-positive T lymphocytes, which were then used for lentivirus infection after continuing to culture for 24 hours.
c)病毒转染法制备抗原特异性T淋巴细胞c) Preparation of antigen-specific T lymphocytes by virus transfection
取上述经过免疫磁珠分离法得到的CD3阳性T淋巴细胞,加入与CD3阳性细胞数相应的所述重组慢病毒进行培养。The above-mentioned CD3 positive T lymphocytes obtained by the immunomagnetic bead separation method were added to the recombinant lentivirus corresponding to the number of CD3 positive cells for cultivation.
培养的第3天,收集合适数量的感染慢病毒的T细胞,流式细胞术分析表面CAR的表达,结果如图2所示,图2中(a)为UTD非转导病毒的T细胞,(b)为CAR-CD276转导病毒的T细胞。使用生物素标记的anti-fab一抗进行染色,然后使用链霉亲和素偶联的APC二抗进行流式分析。图2结果显示,减去UTD背景值,CAR-CD276有26%的表面表达,表明靶向CD276的嵌合抗原受体T细胞制备成功。继续培养48小时,收集靶向CD276的嵌合抗原受体T细胞用于杀伤实验分析或保存在细胞冻存液中,放置于程序降温盒中-80℃保存24小时,然后转移至液氮灌长期保存。On the third day of culture, collect an appropriate number of T cells infected with lentivirus, and analyze the expression of CAR on the surface by flow cytometry. (b) T cells transduced with CAR-CD276 virus. Biotinylated anti-fab primary antibody was used for staining, followed by streptavidin-conjugated APC secondary antibody for flow cytometric analysis. The results in Figure 2 show that CAR-CD276 has 26% surface expression after subtracting the UTD background value, indicating that chimeric antigen receptor T cells targeting CD276 were successfully prepared. Continue to culture for 48 hours, collect chimeric antigen receptor T cells targeting CD276 for killing assay analysis or store in cell cryopreservation solution, place in a programmed cooling box at -80°C for 24 hours, and then transfer to liquid nitrogen irrigation Long-term preservation.
二、体外功能测试2. In vitro function test
本发明经过体外功能测试,靶向CD276的嵌合抗原受体T细胞对CD276阳性肝癌细胞系有杀伤活性。具体方法和结果如下:The present invention has been tested in vitro, and the CD276-targeted chimeric antigen receptor T cells have killing activity on CD276-positive liver cancer cell lines. The specific methods and results are as follows:
(1)使用RTCA系统分析靶向CD276嵌合抗原受体T细胞对CD276阳性肿瘤细胞系杀伤。(1) RTCA system was used to analyze the killing of CD276-positive tumor cell lines by T cells targeting CD276 chimeric antigen receptor.
首先用50ul DMEM培养基进行RTCA单板的平衡,然后收集培养好的靶细胞Huh7,细胞计数,在每个孔中加入含有5000个Huh7细胞的50ul细胞悬液,37℃培养箱中放置15分钟,然后放置在RTCA电阻系统中,24小时后,收集靶向CD276的嵌合抗原受体T细胞,细胞计数,按不同的效应细胞与靶细胞的比例(E:T)10:1,5:1,2.5:1把相应细胞数目的嵌合抗原受体T细胞悬液100ul加入到Huh7细胞中,设置对照组(control,加培养基组),UTD组(加未转导病毒的T细胞),CAR-T组(加靶向CD276嵌合抗原受体T细胞组),每组设置3个复孔,放入RTCA电阻系统中,24小时后进行杀伤活性分析。First use 50ul DMEM medium to balance the RTCA single plate, then collect the cultured target cells Huh7, count the cells, add 50ul cell suspension containing 5000 Huh7 cells to each well, and place in a 37°C incubator for 15 minutes , and then placed in the RTCA resistance system, after 24 hours, collect the chimeric antigen receptor T cells targeting CD276, and count the cells according to the ratio of different effector cells to target cells (E:T) 10:1, 5: 1, 2.5:1 Add 100ul of chimeric antigen receptor T cell suspension corresponding to the number of cells into Huh7 cells, set up control group (control, plus culture medium group), UTD group (plus non-transduced virus T cells) , CAR-T group (plus chimeric antigen receptor T cell group targeting CD276), each group was set up with 3 duplicate wells, put into RTCA resistance system, and the killing activity was analyzed after 24 hours.
(2)结果分析(2) Analysis of results
使用指定时间点的细胞指数(cell index)进行UTD或CAR-T杀伤率分析。The UTD or CAR-T killing rate analysis was performed using the cell index at the specified time point.
所述UTD杀伤率计算公式为:The formula for calculating the UTD kill rate is:
UTD杀伤率=(对照组细胞指数值-UTD组细胞指数值)/对照组细胞指数值×100。UTD killing rate=(cell index value of control group-cell index value of UTD group)/cell index value of control group×100.
所述CAR-T杀伤率计算公式为:The formula for calculating the CAR-T killing rate is:
CAR-T杀伤率=(对照组细胞指数值-CAR-T组细胞指数值)/对照组细胞指数值×100。CAR-T killing rate=(cell index value of control group-cell index value of CAR-T group)/cell index value of control group×100.
结果如图3所示显示,CAR-T与UTD组相比,对Huh7细胞有更强的杀伤活性(图3)。表明构建的靶向CD276的嵌合抗原受体T细胞对表达CD276的阳性肿瘤细胞系有较强的杀伤活性,预示着所述靶向CD276的嵌合抗原受体T细胞在肝细胞癌的治疗中有较大的应用前景。The results are shown in Figure 3. Compared with the UTD group, CAR-T has a stronger killing activity on Huh7 cells (Figure 3). It shows that the constructed CD276-targeting chimeric antigen receptor T cells have strong killing activity on positive tumor cell lines expressing CD276, which indicates that the CD276-targeting chimeric antigen receptor T cells are effective in the treatment of hepatocellular carcinoma. has a great application prospect.
以上内容仅为说明本发明的技术思想,不能以此限定本发明的保护范围,凡是按照本发明提出的技术思想,在技术方案基础上所做的任何改动,均落入本发明权利要求书的保护范围之内。The above content is only to illustrate the technical ideas of the present invention, and cannot limit the protection scope of the present invention. Any changes made on the basis of the technical solutions according to the technical ideas proposed in the present invention shall fall within the scope of the claims of the present invention. within the scope of protection.
序列表sequence listing
<110> 中国科学院深圳先进技术研究院<110> Shenzhen Institute of Advanced Technology, Chinese Academy of Sciences
<120> 靶向CD276的嵌合抗原受体、嵌合抗原受体T细胞及制备方法和制药应用<120> Chimeric antigen receptor targeting CD276, chimeric antigen receptor T cell, preparation method and pharmaceutical application
<160> 16<160> 16
<170> SIPOSequenceListing 1.0<170> SIPOSequenceListing 1.0
<210> 1<210> 1
<211> 467<211> 467
<212> PRT<212> PRT
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 1<400> 1
Glu Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly GlyGlu Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly
1 5 10 151 5 10 15
Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Ser Ser PheSer Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Ser Ser Phe
20 25 3020 25 30
Gly Met His Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp ValGly Met His Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp Val
35 40 4535 40 45
Ala Tyr Ile Ser Ser Asp Ser Ser Ala Ile Tyr Tyr Ala Asp Thr ValAla Tyr Ile Ser Ser Ser Asp Ser Ser Ala Ile Tyr Tyr Ala Asp Thr Val
50 55 6050 55 60
Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys Asn Ser Leu TyrLys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys Asn Ser Leu Tyr
65 70 75 8065 70 75 80
Leu Gln Met Asn Ser Leu Arg Asp Glu Asp Thr Ala Val Tyr Tyr CysLeu Gln Met Asn Ser Leu Arg Asp Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 9585 90 95
Gly Arg Gly Arg Glu Asn Ile Tyr Tyr Gly Ser Arg Leu Asp Tyr TrpGly Arg Gly Arg Glu Asn Ile Tyr Tyr Gly Ser Arg Leu Asp Tyr Trp
100 105 110100 105 110
Gly Gln Gly Thr Thr Val Thr Val Ser Ser Gly Gly Gly Gly Ser GlyGly Gln Gly Thr Thr Val Thr Val Ser Ser Gly Gly Gly Gly Ser Gly
115 120 125115 120 125
Gly Gly Gly Ser Gly Gly Gly Gly Ser Asp Ile Gln Leu Thr Gln SerGly Gly Gly Ser Gly Gly Gly Gly Ser Asp Ile Gln Leu Thr Gln Ser
130 135 140130 135 140
Pro Ser Phe Leu Ser Ala Ser Val Gly Asp Arg Val Thr Ile Thr CysPro Ser Phe Leu Ser Ala Ser Val Gly Asp Arg Val Thr Ile Thr Cys
145 150 155 160145 150 155 160
Lys Ala Ser Gln Asn Val Asp Thr Asn Val Ala Trp Tyr Gln Gln LysLys Ala Ser Gln Asn Val Asp Thr Asn Val Ala Trp Tyr Gln Gln Lys
165 170 175165 170 175
Pro Gly Lys Ala Pro Lys Ala Leu Ile Tyr Ser Ala Ser Tyr Arg TyrPro Gly Lys Ala Pro Lys Ala Leu Ile Tyr Ser Ala Ser Tyr Arg Tyr
180 185 190180 185 190
Ser Gly Val Pro Ser Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp PheSer Gly Val Pro Ser Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp Phe
195 200 205195 200 205
Thr Leu Thr Ile Ser Ser Leu Gln Pro Glu Asp Phe Ala Thr Tyr TyrThr Leu Thr Ile Ser Ser Leu Gln Pro Glu Asp Phe Ala Thr Tyr Tyr
210 215 220210 215 220
Cys Gln Gln Tyr Asn Asn Tyr Pro Phe Thr Phe Gly Gln Gly Thr LysCys Gln Gln Tyr Asn Asn Tyr Pro Phe Thr Phe Gly Gln Gly Thr Lys
225 230 235 240225 230 235 240
Leu Glu Ile Lys Thr Thr Thr Pro Ala Pro Arg Pro Pro Thr Pro AlaLeu Glu Ile Lys Thr Thr Thr Pro Ala Pro Arg Pro Pro Thr Pro Ala
245 250 255245 250 255
Pro Thr Ile Ala Ser Gln Pro Leu Ser Leu Arg Pro Glu Ala Cys ArgPro Thr Ile Ala Ser Gln Pro Leu Ser Leu Arg Pro Glu Ala Cys Arg
260 265 270260 265 270
Pro Ala Ala Gly Gly Ala Val His Thr Arg Gly Leu Asp Phe Ala CysPro Ala Ala Gly Gly Ala Val His Thr Arg Gly Leu Asp Phe Ala Cys
275 280 285275 280 285
Asp Ile Tyr Ile Trp Ala Pro Leu Ala Gly Thr Cys Gly Val Leu LeuAsp Ile Tyr Ile Trp Ala Pro Leu Ala Gly Thr Cys Gly Val Leu Leu
290 295 300290 295 300
Leu Ser Leu Val Ile Thr Leu Tyr Cys Lys Arg Gly Arg Lys Lys LeuLeu Ser Leu Val Ile Thr Leu Tyr Cys Lys Arg Gly Arg Lys Lys Leu
305 310 315 320305 310 315 320
Leu Tyr Ile Phe Lys Gln Pro Phe Met Arg Pro Val Gln Thr Thr GlnLeu Tyr Ile Phe Lys Gln Pro Phe Met Arg Pro Val Gln Thr Thr Gln
325 330 335325 330 335
Glu Glu Asp Gly Cys Ser Cys Arg Phe Pro Glu Glu Glu Glu Gly GlyGlu Glu Asp Gly Cys Ser Cys Arg Phe Pro Glu Glu Glu Glu Gly Gly
340 345 350340 345 350
Cys Glu Leu Arg Val Lys Phe Ser Arg Ser Ala Asp Ala Pro Ala TyrCys Glu Leu Arg Val Lys Phe Ser Arg Ser Ala Asp Ala Pro Ala Tyr
355 360 365355 360 365
Lys Gln Gly Gln Asn Gln Leu Tyr Asn Glu Leu Asn Leu Gly Arg ArgLys Gln Gly Gln Asn Gln Leu Tyr Asn Glu Leu Asn Leu Gly Arg Arg
370 375 380370 375 380
Glu Glu Tyr Asp Val Leu Asp Lys Arg Arg Gly Arg Asp Pro Glu MetGlu Glu Tyr Asp Val Leu Asp Lys Arg Arg Gly Arg Asp Pro Glu Met
385 390 395 400385 390 395 400
Gly Gly Lys Pro Arg Arg Lys Asn Pro Gln Glu Gly Leu Tyr Asn GluGly Gly Lys Pro Arg Arg Lys Asn Pro Gln Glu Gly Leu Tyr Asn Glu
405 410 415405 410 415
Leu Gln Lys Asp Lys Met Ala Glu Ala Tyr Ser Glu Ile Gly Met LysLeu Gln Lys Asp Lys Met Ala Glu Ala Tyr Ser Ser Glu Ile Gly Met Lys
420 425 430420 425 430
Gly Glu Arg Arg Arg Gly Lys Gly His Asp Gly Leu Tyr Gln Gly LeuGly Glu Arg Arg Arg Gly Lys Gly His Asp Gly Leu Tyr Gln Gly Leu
435 440 445435 440 445
Ser Thr Ala Thr Lys Asp Thr Tyr Asp Ala Leu His Met Gln Ala LeuSer Thr Ala Thr Lys Asp Thr Tyr Asp Ala Leu His Met Gln Ala Leu
450 455 460450 455 460
Pro Pro ArgPro Pro Arg
465465
<210> 2<210> 2
<211> 1401<211> 1401
<212> DNA<212>DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 2<400> 2
gaggtacaac tggtggagtc tggggggggc ctggttcagc ctgggggctc tctgagactg 60gaggtacaac tggtggagtc tggggggggc ctggttcagc ctgggggctc tctgagactg 60
agctgtgctg cctctggctt caccttcagc agctttggca tgcactgggt gagacaagcc 120agctgtgctg cctctggctt caccttcagc agctttggca tgcactgggt gagacaagcc 120
cctggcaagg gcctggagtg ggtggcctac atcagctctg acagctctgc catctactat 180cctggcaagg gcctggagtg ggtggcctac atcagctctg acagctctgc catctactat 180
gctgacacag tgaagggcag attcaccatc agcagagaca atgccaagaa cagcctgtac 240gctgacacag tgaagggcag attcaccatc agcagagaca atgccaagaa cagcctgtac 240
ctgcagatga acagcctgag agatgaggac acagctgtgt actactgtgg cagaggcaga 300ctgcagatga acagcctgag agatgaggac acagctgtgt actactgtgg cagaggcaga 300
gagaacatct actatggcag cagactggac tattggggcc aaggcacaac agtgacagtc 360gagaacatct actatggcag cagactggac tattggggcc aaggcacaac agtgacagtc 360
agctctgggg gtggaggatc tggaggtggg ggctctgggg gtgggggatc tgacattcag 420agctctgggg gtggaggatc tggaggtggg ggctctgggg gtgggggatc tgacattcag 420
ctgacacaga gccctagctt cctgtctgcc tctgtggggg acagagtgac catcacctgc 480ctgacacaga gccctagctt cctgtctgcc tctgtggggg acagagtgac catcacctgc 480
aaggcttctc agaatgtgga caccaatgtg gcctggtatc agcagaagcc tggcaaggcc 540aaggcttctc agaatgtgga caccaatgtg gcctggtatc agcagaagcc tggcaaggcc 540
cccaaggccc tgatctactc tgctagctac agatactctg gggtgcctag cagattctct 600cccaaggccc tgatctactc tgctagctac agatactctg gggtgcctag cagattctct 600
ggctctggct ctggcacaga cttcaccctg accatcagca gcctgcagcc tgaggacttt 660ggctctggct ctggcacaga cttcaccctg accatcagca gcctgcagcc tgaggacttt 660
gccacctact actgtcagca gtacaacaac taccccttca cctttggcca aggcactaag 720gccacctact actgtcagca gtacaacaac taccccttca cctttggcca aggcactaag 720
ctggaaatca agaccacgac gccagcgccg cgaccaccaa caccggcgcc caccatcgcg 780ctggaaatca agaccacgac gccagcgccg cgaccaccaa caccggcgcc caccatcgcg 780
tcgcagcccc tgtccctgcg cccagaggcg tgccggccag cggcgggggg cgcagtgcac 840tcgcagcccc tgtccctgcg cccagaggcg tgccggccag cggcgggggg cgcagtgcac 840
acgagggggc tggacttcgc ctgtgatatc tacatctggg cgcccttggc cgggacttgt 900acgagggggc tggacttcgc ctgtgatatc tacatctggg cgcccttggc cgggacttgt 900
ggggtccttc tcctgtcact ggttatcacc ctttactgca aacggggcag aaagaaactc 960ggggtccttc tcctgtcact ggttatcacc ctttatactgca aacggggcag aaagaaactc 960
ctgtatatat tcaaacaacc atttatgaga ccagtacaaa ctactcaaga ggaagatggc 1020ctgtatatat tcaaacaacc atttatgaga ccagtacaaa ctactcaaga ggaagatggc 1020
tgtagctgcc gatttccaga agaagaagaa ggaggatgtg aactgagagt gaagttcagc 1080tgtagctgcc gatttccaga agaagaagaa ggaggatgtg aactgagagt gaagttcagc 1080
aggagcgcag acgcccccgc gtacaagcag ggccagaacc agctctataa cgagctcaat 1140aggagcgcag acgcccccgc gtacaagcag ggccagaacc agctctataa cgagctcaat 1140
ctaggacgaa gagaggagta cgatgttttg gacaagagac gtggccggga ccctgagatg 1200ctaggacgaa gagaggagta cgatgttttg gacaagagac gtggccggga ccctgagatg 1200
gggggaaagc cgagaaggaa gaaccctcag gaaggcctgt acaatgaact gcagaaagat 1260gggggaaagc cgagaaggaa gaaccctcag gaaggcctgt acaatgaact gcagaaagat 1260
aagatggcgg aggcctacag tgagattggg atgaaaggcg agcgccggag gggcaagggg 1320aagatggcgg aggcctacag tgagattggg atgaaaggcg agcgccggag gggcaagggg 1320
cacgatggcc tttaccaggg tctcagtaca gccaccaagg acacctacga cgcccttcac 1380cacgatggcc tttaccaggg tctcagtaca gccaccaagg aacctacga cgcccttcac 1380
atgcaggccc tgccccctcg c 1401atgcaggccc tgccccctcg c 1401
<210> 3<210> 3
<211> 244<211> 244
<212> PRT<212> PRT
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 3<400> 3
Glu Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly GlyGlu Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly
1 5 10 151 5 10 15
Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Ser Ser PheSer Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe Thr Phe Ser Ser Phe
20 25 3020 25 30
Gly Met His Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp ValGly Met His Trp Val Arg Gln Ala Pro Gly Lys Gly Leu Glu Trp Val
35 40 4535 40 45
Ala Tyr Ile Ser Ser Asp Ser Ser Ala Ile Tyr Tyr Ala Asp Thr ValAla Tyr Ile Ser Ser Ser Asp Ser Ser Ala Ile Tyr Tyr Ala Asp Thr Val
50 55 6050 55 60
Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys Asn Ser Leu TyrLys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys Asn Ser Leu Tyr
65 70 75 8065 70 75 80
Leu Gln Met Asn Ser Leu Arg Asp Glu Asp Thr Ala Val Tyr Tyr CysLeu Gln Met Asn Ser Leu Arg Asp Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 9585 90 95
Gly Arg Gly Arg Glu Asn Ile Tyr Tyr Gly Ser Arg Leu Asp Tyr TrpGly Arg Gly Arg Glu Asn Ile Tyr Tyr Gly Ser Arg Leu Asp Tyr Trp
100 105 110100 105 110
Gly Gln Gly Thr Thr Val Thr Val Ser Ser Gly Gly Gly Gly Ser GlyGly Gln Gly Thr Thr Val Thr Val Ser Ser Gly Gly Gly Gly Ser Gly
115 120 125115 120 125
Gly Gly Gly Ser Gly Gly Gly Gly Ser Asp Ile Gln Leu Thr Gln SerGly Gly Gly Ser Gly Gly Gly Gly Ser Asp Ile Gln Leu Thr Gln Ser
130 135 140130 135 140
Pro Ser Phe Leu Ser Ala Ser Val Gly Asp Arg Val Thr Ile Thr CysPro Ser Phe Leu Ser Ala Ser Val Gly Asp Arg Val Thr Ile Thr Cys
145 150 155 160145 150 155 160
Lys Ala Ser Gln Asn Val Asp Thr Asn Val Ala Trp Tyr Gln Gln LysLys Ala Ser Gln Asn Val Asp Thr Asn Val Ala Trp Tyr Gln Gln Lys
165 170 175165 170 175
Pro Gly Lys Ala Pro Lys Ala Leu Ile Tyr Ser Ala Ser Tyr Arg TyrPro Gly Lys Ala Pro Lys Ala Leu Ile Tyr Ser Ala Ser Tyr Arg Tyr
180 185 190180 185 190
Ser Gly Val Pro Ser Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp PheSer Gly Val Pro Ser Arg Phe Ser Gly Ser Gly Ser Gly Thr Asp Phe
195 200 205195 200 205
Thr Leu Thr Ile Ser Ser Leu Gln Pro Glu Asp Phe Ala Thr Tyr TyrThr Leu Thr Ile Ser Ser Leu Gln Pro Glu Asp Phe Ala Thr Tyr Tyr
210 215 220210 215 220
Cys Gln Gln Tyr Asn Asn Tyr Pro Phe Thr Phe Gly Gln Gly Thr LysCys Gln Gln Tyr Asn Asn Tyr Pro Phe Thr Phe Gly Gln Gly Thr Lys
225 230 235 240225 230 235 240
Leu Glu Ile LysLeu Glu Ile Lys
<210> 4<210> 4
<211> 732<211> 732
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 4<400> 4
gaggtacaac tggtggagtc tggggggggc ctggttcagc ctgggggctc tctgagactg 60gaggtacaac tggtggagtc tggggggggc ctggttcagc ctgggggctc tctgagactg 60
agctgtgctg cctctggctt caccttcagc agctttggca tgcactgggt gagacaagcc 120agctgtgctg cctctggctt caccttcagc agctttggca tgcactgggt gagacaagcc 120
cctggcaagg gcctggagtg ggtggcctac atcagctctg acagctctgc catctactat 180cctggcaagg gcctggagtg ggtggcctac atcagctctg acagctctgc catctactat 180
gctgacacag tgaagggcag attcaccatc agcagagaca atgccaagaa cagcctgtac 240gctgacacag tgaagggcag attcaccatc agcagagaca atgccaagaa cagcctgtac 240
ctgcagatga acagcctgag agatgaggac acagctgtgt actactgtgg cagaggcaga 300ctgcagatga acagcctgag agatgaggac acagctgtgt actactgtgg cagaggcaga 300
gagaacatct actatggcag cagactggac tattggggcc aaggcacaac agtgacagtc 360gagaacatct actatggcag cagactggac tattggggcc aaggcacaac agtgacagtc 360
agctctgggg gtggaggatc tggaggtggg ggctctgggg gtgggggatc tgacattcag 420agctctgggg gtggaggatc tggaggtggg ggctctgggg gtgggggatc tgacattcag 420
ctgacacaga gccctagctt cctgtctgcc tctgtggggg acagagtgac catcacctgc 480ctgacacaga gccctagctt cctgtctgcc tctgtggggg acagagtgac catcacctgc 480
aaggcttctc agaatgtgga caccaatgtg gcctggtatc agcagaagcc tggcaaggcc 540aaggcttctc agaatgtgga caccaatgtg gcctggtatc agcagaagcc tggcaaggcc 540
cccaaggccc tgatctactc tgctagctac agatactctg gggtgcctag cagattctct 600cccaaggccc tgatctactc tgctagctac agatactctg gggtgcctag cagattctct 600
ggctctggct ctggcacaga cttcaccctg accatcagca gcctgcagcc tgaggacttt 660ggctctggct ctggcacaga cttcaccctg accatcagca gcctgcagcc tgaggacttt 660
gccacctact actgtcagca gtacaacaac taccccttca cctttggcca aggcactaag 720gccacctact actgtcagca gtacaacaac taccccttca cctttggcca aggcactaag 720
ctggaaatca ag 732ctggaaatca ag 732
<210> 5<210> 5
<211> 45<211> 45
<212> PRT<212> PRT
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 5<400> 5
Thr Thr Thr Pro Ala Pro Arg Pro Pro Thr Pro Ala Pro Thr Ile AlaThr Thr Thr Pro Ala Pro Arg Pro Pro Thr Pro Ala Pro Thr Ile Ala
1 5 10 151 5 10 15
Ser Gln Pro Leu Ser Leu Arg Pro Glu Ala Cys Arg Pro Ala Ala GlySer Gln Pro Leu Ser Leu Arg Pro Glu Ala Cys Arg Pro Ala Ala Gly
20 25 3020 25 30
Gly Ala Val His Thr Arg Gly Leu Asp Phe Ala Cys AspGly Ala Val His Thr Arg Gly Leu Asp Phe Ala Cys Asp
35 40 4535 40 45
<210> 6<210> 6
<211> 135<211> 135
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 6<400> 6
accacgacgc cagcgccgcg accaccaaca ccggcgccca ccatcgcgtc gcagcccctg 60accacgacgc cagcgccgcg accaccaaca ccggcgccca ccatcgcgtc gcagcccctg 60
tccctgcgcc cagaggcgtg ccggccagcg gcggggggcg cagtgcacac gagggggctg 120tccctgcgcc cagaggcgtg ccggccagcg gcggggggcg cagtgcacac gagggggctg 120
gacttcgcct gtgat 135gacttcgcct gtgat 135
<210> 7<210> 7
<211> 24<211> 24
<212> PRT<212> PRT
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 7<400> 7
Ile Tyr Ile Trp Ala Pro Leu Ala Gly Thr Cys Gly Val Leu Leu LeuIle Tyr Ile Trp Ala Pro Leu Ala Gly Thr Cys Gly Val Leu Leu Leu
1 5 10 151 5 10 15
Ser Leu Val Ile Thr Leu Tyr CysSer Leu Val Ile Thr Leu Tyr Cys
2020
<210> 8<210> 8
<211> 72<211> 72
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 8<400> 8
atctacatct gggcgccctt ggccgggact tgtggggtcc ttctcctgtc actggttatc 60atctacatct gggcgccctt ggccgggact tgtggggtcc ttctcctgtc actggttatc 60
accctttact gc 72accctttact gc 72
<210> 9<210> 9
<211> 42<211> 42
<212> PRT<212> PRT
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 9<400> 9
Lys Arg Gly Arg Lys Lys Leu Leu Tyr Ile Phe Lys Gln Pro Phe MetLys Arg Gly Arg Lys Lys Leu Leu Tyr Ile Phe Lys Gln Pro Phe Met
1 5 10 151 5 10 15
Arg Pro Val Gln Thr Thr Gln Glu Glu Asp Gly Cys Ser Cys Arg PheArg Pro Val Gln Thr Thr Gln Glu Glu Asp Gly Cys Ser Cys Arg Phe
20 25 3020 25 30
Pro Glu Glu Glu Glu Gly Gly Cys Glu LeuPro Glu Glu Glu Glu Gly Gly Cys Glu Leu
35 4035 40
<210> 10<210> 10
<211> 126<211> 126
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 10<400> 10
aaacggggca gaaagaaact cctgtatata ttcaaacaac catttatgag accagtacaa 60aaacggggca gaaagaaact cctgtatata ttcaaacaac catttatgag accagtacaa 60
actactcaag aggaagatgg ctgtagctgc cgatttccag aagaagaaga aggaggatgt 120actactcaag aggaagatgg ctgtagctgc cgatttccag aagaagaaga aggagatgt 120
gaactg 126gaactg 126
<210> 11<210> 11
<211> 112<211> 112
<212> PRT<212> PRT
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 11<400> 11
Arg Val Lys Phe Ser Arg Ser Ala Asp Ala Pro Ala Tyr Lys Gln GlyArg Val Lys Phe Ser Arg Ser Ala Asp Ala Pro Ala Tyr Lys Gln Gly
1 5 10 151 5 10 15
Gln Asn Gln Leu Tyr Asn Glu Leu Asn Leu Gly Arg Arg Glu Glu TyrGln Asn Gln Leu Tyr Asn Glu Leu Asn Leu Gly Arg Arg Glu Glu Tyr
20 25 3020 25 30
Asp Val Leu Asp Lys Arg Arg Gly Arg Asp Pro Glu Met Gly Gly LysAsp Val Leu Asp Lys Arg Arg Gly Arg Asp Pro Glu Met Gly Gly Lys
35 40 4535 40 45
Pro Arg Arg Lys Asn Pro Gln Glu Gly Leu Tyr Asn Glu Leu Gln LysPro Arg Arg Lys Asn Pro Gln Glu Gly Leu Tyr Asn Glu Leu Gln Lys
50 55 6050 55 60
Asp Lys Met Ala Glu Ala Tyr Ser Glu Ile Gly Met Lys Gly Glu ArgAsp Lys Met Ala Glu Ala Tyr Ser Glu Ile Gly Met Lys Gly Glu Arg
65 70 75 8065 70 75 80
Arg Arg Gly Lys Gly His Asp Gly Leu Tyr Gln Gly Leu Ser Thr AlaArg Arg Gly Lys Gly His Asp Gly Leu Tyr Gln Gly Leu Ser Thr Ala
85 90 9585 90 95
Thr Lys Asp Thr Tyr Asp Ala Leu His Met Gln Ala Leu Pro Pro ArgThr Lys Asp Thr Tyr Asp Ala Leu His Met Gln Ala Leu Pro Pro Arg
100 105 110100 105 110
<210> 12<210> 12
<211> 339<211> 339
<212> DNA<212>DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 12<400> 12
agagtgaagt tcagcaggag cgcagacgcc cccgcgtaca agcagggcca gaaccagctc 60agagtgaagt tcagcaggag cgcagacgcc cccgcgtaca agcagggcca gaaccagctc 60
tataacgagc tcaatctagg acgaagagag gagtacgatg ttttggacaa gagacgtggc 120tataacgagc tcaatctagg acgaagagag gagtacgatg ttttggacaa gagacgtggc 120
cgggaccctg agatgggggg aaagccgaga aggaagaacc ctcaggaagg cctgtacaat 180cgggaccctg agatgggggg aaagccgaga aggaagaacc ctcaggaagg cctgtacaat 180
gaactgcaga aagataagat ggcggaggcc tacagtgaga ttgggatgaa aggcgagcgc 240gaactgcaga aagataagat ggcggaggcc tacagtgaga ttgggatgaa aggcgagcgc 240
cggaggggca aggggcacga tggcctttac cagggtctca gtacagccac caaggacacc 300cggaggggca aggggcacga tggcctttac cagggtctca gtacagccac caaggacacc 300
tacgacgccc ttcacatgca ggccctgccc cctcgctga 339tacgacgccc ttcacatgca ggccctgccc cctcgctga 339
<210> 13<210> 13
<211> 1467<211> 1467
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 13<400> 13
atggccctgc ctgtgacagc cctgttactt cccctggccc tgttactgca tgctgctaga 60atggccctgc ctgtgacagc cctgttactt cccctggccc tgttactgca tgctgctaga 60
cctgaggtac aactggtgga gtctgggggg ggcctggttc agcctggggg ctctctgaga 120cctgaggtac aactggtgga gtctgggggg ggcctggttc agcctggggg ctctctgaga 120
ctgagctgtg ctgcctctgg cttcaccttc agcagctttg gcatgcactg ggtgagacaa 180ctgagctgtg ctgcctctgg cttcaccttc agcagctttg gcatgcactg ggtgagacaa 180
gcccctggca agggcctgga gtgggtggcc tacatcagct ctgacagctc tgccatctac 240gcccctggca agggcctgga gtgggtggcc tacatcagct ctgacagctc tgccatctac 240
tatgctgaca cagtgaaggg cagattcacc atcagcagag acaatgccaa gaacagcctg 300tatgctgaca cagtgaaggg cagattcacc atcagcagag acaatgccaa gaacagcctg 300
tacctgcaga tgaacagcct gagagatgag gacacagctg tgtactactg tggcagaggc 360tacctgcaga tgaacagcct gagagatgag gacacagctg tgtactactg tggcagaggc 360
agagagaaca tctactatgg cagcagactg gactattggg gccaaggcac aacagtgaca 420agagagaaca tctactatgg cagcagactg gactattggg gccaaggcac aacagtgaca 420
gtcagctctg ggggtggagg atctggaggt gggggctctg ggggtggggg atctgacatt 480gtcagctctg gggtggagg atctggaggt gggggctctg gggtggggg atctgacatt 480
cagctgacac agagccctag cttcctgtct gcctctgtgg gggacagagt gaccatcacc 540cagctgacac agagccctag cttcctgtct gcctctgtgg gggacagagt gaccatcacc 540
tgcaaggctt ctcagaatgt ggacaccaat gtggcctggt atcagcagaa gcctggcaag 600tgcaaggctt ctcagaatgt ggacaccaat gtggcctggt atcagcagaa gcctggcaag 600
gcccccaagg ccctgatcta ctctgctagc tacagatact ctggggtgcc tagcagattc 660gcccccaagg ccctgatcta ctctgctagc tacagatact ctggggtgcc tagcagattc 660
tctggctctg gctctggcac agacttcacc ctgaccatca gcagcctgca gcctgaggac 720tctggctctg gctctggcac agacttcacc ctgaccatca gcagcctgca gcctgaggac 720
tttgccacct actactgtca gcagtacaac aactacccct tcacctttgg ccaaggcact 780tttgccacct actactgtca gcagtacaac aactacccct tcacctttgg ccaaggcact 780
aagctggaaa tcaagaccac gacgccagcg ccgcgaccac caacaccggc gcccaccatc 840aagctggaaa tcaagaccac gacgccagcg ccgcgaccac caacaccggc gcccaccatc 840
gcgtcgcagc ccctgtccct gcgcccagag gcgtgccggc cagcggcggg gggcgcagtg 900gcgtcgcagc ccctgtccct gcgccccagag gcgtgccggc cagcggcggg gggcgcagtg 900
cacacgaggg ggctggactt cgcctgtgat atctacatct gggcgccctt ggccgggact 960cacacgaggg ggctggactt cgcctgtgat atctacatct gggcgccctt ggccgggact 960
tgtggggtcc ttctcctgtc actggttatc accctttact gcaaacgggg cagaaagaaa 1020tgtggggtcc ttctcctgtc actggttatc accctttact gcaaacgggg cagaaagaaa 1020
ctcctgtata tattcaaaca accatttatg agaccagtac aaactactca agaggaagat 1080ctcctgtata tattcaaaca accatttatg agaccagtac aaactactca agaggaagat 1080
ggctgtagct gccgatttcc agaagaagaa gaaggaggat gtgaactgag agtgaagttc 1140ggctgtagct gccgatttcc agaagaagaa gaaggaggat gtgaactgag agtgaagttc 1140
agcaggagcg cagacgcccc cgcgtacaag cagggccaga accagctcta taacgagctc 1200agcaggagcg cagacgcccc cgcgtacaag cagggccaga accagctcta taacgagctc 1200
aatctaggac gaagagagga gtacgatgtt ttggacaaga gacgtggccg ggaccctgag 1260aatctaggac gaagagagga gtacgatgtt ttggacaaga gacgtggccg ggaccctgag 1260
atggggggaa agccgagaag gaagaaccct caggaaggcc tgtacaatga actgcagaaa 1320atggggggaa agccgagaag gaagaaccct caggaaggcc tgtacaatga actgcagaaa 1320
gataagatgg cggaggccta cagtgagatt gggatgaaag gcgagcgccg gaggggcaag 1380gataagatgg cggaggccta cagtgagatt gggatgaaag gcgagcgccg gaggggcaag 1380
gggcacgatg gcctttacca gggtctcagt acagccacca aggacaccta cgacgccctt 1440gggcacgatg gcctttacca gggtctcagt acagccacca aggacccta cgacgccctt 1440
cacatgcagg ccctgccccc tcgctga 1467cacatgcagg ccctgccccc tcgctga 1467
<210> 14<210> 14
<211> 488<211> 488
<212> PRT<212> PRT
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 14<400> 14
Met Ala Leu Pro Val Thr Ala Leu Leu Leu Pro Leu Ala Leu Leu LeuMet Ala Leu Pro Val Thr Ala Leu Leu Leu Pro Leu Ala Leu Leu Leu
1 5 10 151 5 10 15
His Ala Ala Arg Pro Glu Val Gln Leu Val Glu Ser Gly Gly Gly LeuHis Ala Ala Arg Pro Glu Val Gln Leu Val Glu Ser Gly Gly Gly Leu
20 25 3020 25 30
Val Gln Pro Gly Gly Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly PheVal Gln Pro Gly Gly Ser Leu Arg Leu Ser Cys Ala Ala Ser Gly Phe
35 40 4535 40 45
Thr Phe Ser Ser Phe Gly Met His Trp Val Arg Gln Ala Pro Gly LysThr Phe Ser Ser Phe Gly Met His Trp Val Arg Gln Ala Pro Gly Lys
50 55 6050 55 60
Gly Leu Glu Trp Val Ala Tyr Ile Ser Ser Asp Ser Ser Ala Ile TyrGly Leu Glu Trp Val Ala Tyr Ile Ser Ser Asp Ser Ser Ala Ile Tyr
65 70 75 8065 70 75 80
Tyr Ala Asp Thr Val Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn AlaTyr Ala Asp Thr Val Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala
85 90 9585 90 95
Lys Asn Ser Leu Tyr Leu Gln Met Asn Ser Leu Arg Asp Glu Asp ThrLys Asn Ser Leu Tyr Leu Gln Met Asn Ser Leu Arg Asp Glu Asp Thr
100 105 110100 105 110
Ala Val Tyr Tyr Cys Gly Arg Gly Arg Glu Asn Ile Tyr Tyr Gly SerAla Val Tyr Tyr Cys Gly Arg Gly Arg Glu Asn Ile Tyr Tyr Gly Ser
115 120 125115 120 125
Arg Leu Asp Tyr Trp Gly Gln Gly Thr Thr Val Thr Val Ser Ser GlyArg Leu Asp Tyr Trp Gly Gln Gly Thr Thr Val Thr Val Ser Ser Gly
130 135 140130 135 140
Gly Gly Gly Ser Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser Asp IleGly Gly Gly Ser Gly Gly Gly Gly Ser Gly Gly Gly Gly Ser Asp Ile
145 150 155 160145 150 155 160
Gln Leu Thr Gln Ser Pro Ser Phe Leu Ser Ala Ser Val Gly Asp ArgGln Leu Thr Gln Ser Pro Ser Phe Leu Ser Ala Ser Val Gly Asp Arg
165 170 175165 170 175
Val Thr Ile Thr Cys Lys Ala Ser Gln Asn Val Asp Thr Asn Val AlaVal Thr Ile Thr Cys Lys Ala Ser Gln Asn Val Asp Thr Asn Val Ala
180 185 190180 185 190
Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Ala Leu Ile Tyr SerTrp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Ala Leu Ile Tyr Ser
195 200 205195 200 205
Ala Ser Tyr Arg Tyr Ser Gly Val Pro Ser Arg Phe Ser Gly Ser GlyAla Ser Tyr Arg Tyr Ser Gly Val Pro Ser Arg Phe Ser Gly Ser Gly
210 215 220210 215 220
Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro Glu AspSer Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro Glu Asp
225 230 235 240225 230 235 240
Phe Ala Thr Tyr Tyr Cys Gln Gln Tyr Asn Asn Tyr Pro Phe Thr PhePhe Ala Thr Tyr Tyr Cys Gln Gln Tyr Asn Asn Tyr Pro Phe Thr Phe
245 250 255245 250 255
Gly Gln Gly Thr Lys Leu Glu Ile Lys Thr Thr Thr Pro Ala Pro ArgGly Gln Gly Thr Lys Leu Glu Ile Lys Thr Thr Thr Pro Ala Pro Arg
260 265 270260 265 270
Pro Pro Thr Pro Ala Pro Thr Ile Ala Ser Gln Pro Leu Ser Leu ArgPro Pro Thr Pro Ala Pro Thr Ile Ala Ser Gln Pro Leu Ser Leu Arg
275 280 285275 280 285
Pro Glu Ala Cys Arg Pro Ala Ala Gly Gly Ala Val His Thr Arg GlyPro Glu Ala Cys Arg Pro Ala Ala Gly Gly Ala Val His Thr Arg Gly
290 295 300290 295 300
Leu Asp Phe Ala Cys Asp Ile Tyr Ile Trp Ala Pro Leu Ala Gly ThrLeu Asp Phe Ala Cys Asp Ile Tyr Ile Trp Ala Pro Leu Ala Gly Thr
305 310 315 320305 310 315 320
Cys Gly Val Leu Leu Leu Ser Leu Val Ile Thr Leu Tyr Cys Lys ArgCys Gly Val Leu Leu Leu Ser Leu Val Ile Thr Leu Tyr Cys Lys Arg
325 330 335325 330 335
Gly Arg Lys Lys Leu Leu Tyr Ile Phe Lys Gln Pro Phe Met Arg ProGly Arg Lys Lys Leu Leu Tyr Ile Phe Lys Gln Pro Phe Met Arg Pro
340 345 350340 345 350
Val Gln Thr Thr Gln Glu Glu Asp Gly Cys Ser Cys Arg Phe Pro GluVal Gln Thr Thr Gln Glu Glu Asp Gly Cys Ser Cys Arg Phe Pro Glu
355 360 365355 360 365
Glu Glu Glu Gly Gly Cys Glu Leu Arg Val Lys Phe Ser Arg Ser AlaGlu Glu Glu Gly Gly Cys Glu Leu Arg Val Lys Phe Ser Arg Ser Ala
370 375 380370 375 380
Asp Ala Pro Ala Tyr Lys Gln Gly Gln Asn Gln Leu Tyr Asn Glu LeuAsp Ala Pro Ala Tyr Lys Gln Gly Gln Asn Gln Leu Tyr Asn Glu Leu
385 390 395 400385 390 395 400
Asn Leu Gly Arg Arg Glu Glu Tyr Asp Val Leu Asp Lys Arg Arg GlyAsn Leu Gly Arg Arg Glu Glu Tyr Asp Val Leu Asp Lys Arg Arg Gly
405 410 415405 410 415
Arg Asp Pro Glu Met Gly Gly Lys Pro Arg Arg Lys Asn Pro Gln GluArg Asp Pro Glu Met Gly Gly Lys Pro Arg Arg Lys Asn Pro Gln Glu
420 425 430420 425 430
Gly Leu Tyr Asn Glu Leu Gln Lys Asp Lys Met Ala Glu Ala Tyr SerGly Leu Tyr Asn Glu Leu Gln Lys Asp Lys Met Ala Glu Ala Tyr Ser
435 440 445435 440 445
Glu Ile Gly Met Lys Gly Glu Arg Arg Arg Gly Lys Gly His Asp GlyGlu Ile Gly Met Lys Gly Glu Arg Arg Arg Gly Lys Gly His Asp Gly
450 455 460450 455 460
Leu Tyr Gln Gly Leu Ser Thr Ala Thr Lys Asp Thr Tyr Asp Ala LeuLeu Tyr Gln Gly Leu Ser Thr Ala Thr Lys Asp Thr Tyr Asp Ala Leu
465 470 475 480465 470 475 480
His Met Gln Ala Leu Pro Pro ArgHis Met Gln Ala Leu Pro Pro Arg
485485
<210> 15<210> 15
<211> 63<211> 63
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 15<400> 15
atggccctgc ctgtgacagc cctgttactt cccctggccc tgttactgca tgctgctaga 60atggccctgc ctgtgacagc cctgttactt cccctggccc tgttactgca tgctgctaga 60
cct 63cct 63
<210> 16<210> 16
<211> 21<211> 21
<212> PRT<212> PRT
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence (Artificial Sequence)
<400> 16<400> 16
Met Ala Leu Pro Val Thr Ala Leu Leu Leu Pro Leu Ala Leu Leu LeuMet Ala Leu Pro Val Thr Ala Leu Leu Leu Pro Leu Ala Leu Leu Leu
1 5 10 151 5 10 15
His Ala Ala Arg ProHis Ala Ala Arg Pro
2020
Claims (10)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111290880.7A CN113980143B (en) | 2021-11-02 | 2021-11-02 | Chimeric antigen receptor, chimeric antigen receptor T cell targeting CD276, preparation method and pharmaceutical application |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111290880.7A CN113980143B (en) | 2021-11-02 | 2021-11-02 | Chimeric antigen receptor, chimeric antigen receptor T cell targeting CD276, preparation method and pharmaceutical application |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN113980143A CN113980143A (en) | 2022-01-28 |
| CN113980143B true CN113980143B (en) | 2023-04-25 |
Family
ID=79745927
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202111290880.7A Active CN113980143B (en) | 2021-11-02 | 2021-11-02 | Chimeric antigen receptor, chimeric antigen receptor T cell targeting CD276, preparation method and pharmaceutical application |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113980143B (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115678856A (en) * | 2022-06-09 | 2023-02-03 | 郑州大学第一附属医院 | A kind of tumor killer cell and its application |
| CN116239699B (en) * | 2022-11-10 | 2023-11-17 | 汕头普罗凯融生物医药科技有限公司 | Chimeric antigen receptor targeting CD276 and application thereof |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11472889B2 (en) * | 2017-10-14 | 2022-10-18 | Cytomx Therapeutics, Inc. | Antibodies, activatable antibodies, bispecific antibodies, and bispecific activatable antibodies and methods of use thereof |
| JP2021514648A (en) * | 2018-03-01 | 2021-06-17 | エフ・ホフマン−ラ・ロシュ・アクチェンゲゼルシャフト | Specificity assay for novel target antigen binding moieties |
| JP2022512684A (en) * | 2018-10-11 | 2022-02-07 | インヒブルクス インコーポレイテッド | B7H3 single domain antibody and therapeutic composition thereof |
| US20240082399A1 (en) * | 2019-10-11 | 2024-03-14 | The Board Of Trustees Of The Leland Stanford Junior University | Recombinant polypeptides for regulatable cellular localization |
-
2021
- 2021-11-02 CN CN202111290880.7A patent/CN113980143B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN113980143A (en) | 2022-01-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN113248616B (en) | Chimeric antigen receptor targeting GPC3 and uses thereof | |
| CN109111525B (en) | HLA-G chimeric antigen receptor, coding sequence, expression vector and application | |
| CN110144326A (en) | A kind of antitumor T cell of targeting and its preparation method and application | |
| CN109266667A (en) | Target the Chimeric antigen receptor and its application of CD5 | |
| CN114163537B (en) | Chimeric antigen receptor T cell secreting bispecific antibody, and preparation method and application thereof | |
| CN110144328A (en) | A kind of antitumor T cell of targeting and its preparation method and application | |
| WO2025242064A1 (en) | Nanoantibody targeting phosphatidylinositol proteoglycan 3 and use thereof | |
| CN113980143B (en) | Chimeric antigen receptor, chimeric antigen receptor T cell targeting CD276, preparation method and pharmaceutical application | |
| CN114957484A (en) | CAR vector targeting solid tumor cell B7-H3 protein, CAR-T cell and construction method and application thereof | |
| CN114163538B (en) | Chimeric antigen receptors, chimeric antigen receptor T cells targeting GPC3 and CD276 simultaneously, and preparation methods and applications thereof | |
| CN111378624B (en) | Targeting anti-tumor T cell and preparation method and application thereof | |
| CN113999320B (en) | CD 276-targeted chimeric antigen receptor taking CD28 and 4-1BB as co-stimulation domains and application thereof | |
| CN105343874A (en) | Prostate cancer nucleic acid vaccine | |
| CN110526981A (en) | A kind of AntiCD3 McAb and EpCAM bispecific antibody and its application in treatment lung cancer | |
| CN113045665B (en) | CAIX-CAR-T cell driven by HVEM (high-voltage alternating current) co-stimulation signal as well as preparation method and application of CAIX-CAR-T cell | |
| CN107557341B (en) | anti-WT1 enhanced chimeric antigen receptor modified immune cell and application thereof | |
| CN118001381A (en) | Pharmaceutical composition and application thereof | |
| CN111378623B (en) | Targeting anti-tumor T cell and preparation method and application thereof | |
| CN114075548B (en) | AXL-targeted CAR-T cell, and preparation method and application thereof | |
| CN116903756A (en) | A bispecific chimeric antigen receptor targeting PSMA antigen and its application | |
| CN108864288A (en) | The double target spot CAR-T therapy vectors and its construction method of breast cancer and application | |
| CN110526974A (en) | It is a kind of to target the single-chain antibody of MUC16, Chimeric antigen receptor T cell and its preparation method and application | |
| CN110144325A (en) | A kind of targeting T lymphocyte and its preparation method and application | |
| CN110526990A (en) | Target Chimeric antigen receptor, the Chimeric antigen receptor T cell and its preparation method and application of CD30 | |
| CN118005794A (en) | A heavy chain antibody against CLDN18.2, related products and uses |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CP03 | Change of name, title or address |
Address after: 1068 Xueyuan Avenue Xili University City Shenzhen Guangdong Province Patentee after: SHENZHEN INSTITUTES OF ADVANCED TECHNOLOGY Country or region after: China Patentee after: Shenzhen University of Technology (preparatory) Address before: No.1068 Xueyuan Avenue, Xili University Town, Shenzhen, Guangdong 518055 Patentee before: SHENZHEN INSTITUTES OF ADVANCED TECHNOLOGY Country or region before: China Patentee before: Shenzhen University of technology Chinese Academy of Sciences (Preparatory) |
|
| CP03 | Change of name, title or address | ||
| CP03 | Change of name, title or address |
Address after: No.1068 Xueyuan Avenue, Xili University Town, Shenzhen, Guangdong 518055 Patentee after: SHENZHEN INSTITUTES OF ADVANCED TECHNOLOGY Country or region after: China Patentee after: Shenzhen University of Technology Address before: No.1068 Xueyuan Avenue, Xili University Town, Shenzhen, Guangdong 518055 Patentee before: SHENZHEN INSTITUTES OF ADVANCED TECHNOLOGY Country or region before: China Patentee before: Shenzhen University of Technology (preparatory) |
|
| CP03 | Change of name, title or address |