CN113813245B - Slow-release sleep-aiding snuff - Google Patents
Slow-release sleep-aiding snuff Download PDFInfo
- Publication number
- CN113813245B CN113813245B CN202111180413.9A CN202111180413A CN113813245B CN 113813245 B CN113813245 B CN 113813245B CN 202111180413 A CN202111180413 A CN 202111180413A CN 113813245 B CN113813245 B CN 113813245B
- Authority
- CN
- China
- Prior art keywords
- sleep
- essential oil
- release
- aiding
- slow
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000000341 volatile oil Substances 0.000 claims abstract description 73
- 150000001875 compounds Chemical class 0.000 claims abstract description 47
- 239000008188 pellet Substances 0.000 claims abstract description 43
- 239000003094 microcapsule Substances 0.000 claims abstract description 41
- 238000013268 sustained release Methods 0.000 claims abstract description 30
- 239000012730 sustained-release form Substances 0.000 claims abstract description 30
- 238000002360 preparation method Methods 0.000 claims abstract description 27
- 239000000843 powder Substances 0.000 claims abstract description 24
- 239000002775 capsule Substances 0.000 claims abstract description 20
- 239000000463 material Substances 0.000 claims abstract description 19
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 19
- 239000000080 wetting agent Substances 0.000 claims abstract description 18
- 239000007779 soft material Substances 0.000 claims abstract description 16
- 239000007787 solid Substances 0.000 claims abstract description 14
- 239000000654 additive Substances 0.000 claims abstract description 13
- 239000000853 adhesive Substances 0.000 claims abstract description 10
- 230000001070 adhesive effect Effects 0.000 claims abstract description 10
- 239000006187 pill Substances 0.000 claims abstract 4
- 238000002156 mixing Methods 0.000 claims abstract 2
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims description 23
- 239000000243 solution Substances 0.000 claims description 18
- 239000007864 aqueous solution Substances 0.000 claims description 17
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 claims description 15
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 claims description 15
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 claims description 14
- 239000000661 sodium alginate Substances 0.000 claims description 14
- 235000010413 sodium alginate Nutrition 0.000 claims description 14
- 229940005550 sodium alginate Drugs 0.000 claims description 14
- RVGRUAULSDPKGF-UHFFFAOYSA-N Poloxamer Chemical group C1CO1.CC1CO1 RVGRUAULSDPKGF-UHFFFAOYSA-N 0.000 claims description 13
- 229940044476 poloxamer 407 Drugs 0.000 claims description 13
- 229920001992 poloxamer 407 Polymers 0.000 claims description 13
- HVUMOYIDDBPOLL-XWVZOOPGSA-N Sorbitan monostearate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O HVUMOYIDDBPOLL-XWVZOOPGSA-N 0.000 claims description 10
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 claims description 10
- 229920000053 polysorbate 80 Polymers 0.000 claims description 10
- 238000003756 stirring Methods 0.000 claims description 10
- 229920001661 Chitosan Polymers 0.000 claims description 9
- 244000014047 Polianthes tuberosa Species 0.000 claims description 9
- 235000016067 Polianthes tuberosa Nutrition 0.000 claims description 9
- 244000197975 Solidago virgaurea Species 0.000 claims description 9
- 235000000914 Solidago virgaurea Nutrition 0.000 claims description 9
- 230000000996 additive effect Effects 0.000 claims description 9
- 229910052751 metal Inorganic materials 0.000 claims description 9
- 239000002861 polymer material Substances 0.000 claims description 9
- 229920002261 Corn starch Polymers 0.000 claims description 8
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 claims description 8
- 229930195725 Mannitol Natural products 0.000 claims description 8
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 claims description 8
- 239000001768 carboxy methyl cellulose Substances 0.000 claims description 8
- 239000008120 corn starch Substances 0.000 claims description 8
- 239000000594 mannitol Substances 0.000 claims description 8
- 235000010355 mannitol Nutrition 0.000 claims description 8
- 239000007769 metal material Substances 0.000 claims description 8
- 238000000034 method Methods 0.000 claims description 8
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 claims description 8
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 claims description 8
- 229920000168 Microcrystalline cellulose Polymers 0.000 claims description 7
- 238000005354 coacervation Methods 0.000 claims description 7
- 239000000839 emulsion Substances 0.000 claims description 7
- 239000008108 microcrystalline cellulose Substances 0.000 claims description 7
- 229940016286 microcrystalline cellulose Drugs 0.000 claims description 7
- 235000019813 microcrystalline cellulose Nutrition 0.000 claims description 7
- 239000003921 oil Substances 0.000 claims description 7
- 240000002505 Pogostemon cablin Species 0.000 claims description 5
- 235000011751 Pogostemon cablin Nutrition 0.000 claims description 5
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 claims description 4
- 241000675108 Citrus tangerina Species 0.000 claims 2
- 239000011148 porous material Substances 0.000 claims 2
- 238000001035 drying Methods 0.000 claims 1
- 238000004108 freeze drying Methods 0.000 claims 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical group OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 claims 1
- 239000002184 metal Substances 0.000 claims 1
- 238000012216 screening Methods 0.000 claims 1
- 238000007789 sealing Methods 0.000 claims 1
- 230000000694 effects Effects 0.000 abstract description 14
- 239000011230 binding agent Substances 0.000 abstract description 8
- 210000003928 nasal cavity Anatomy 0.000 abstract description 2
- 238000005453 pelletization Methods 0.000 description 10
- 208000013738 Sleep Initiation and Maintenance disease Diseases 0.000 description 8
- 206010022437 insomnia Diseases 0.000 description 8
- 239000007788 liquid Substances 0.000 description 6
- 239000008367 deionised water Substances 0.000 description 5
- 229910021641 deionized water Inorganic materials 0.000 description 5
- 238000009826 distribution Methods 0.000 description 5
- 229940079593 drug Drugs 0.000 description 5
- 239000003814 drug Substances 0.000 description 5
- 239000004615 ingredient Substances 0.000 description 5
- 239000000203 mixture Substances 0.000 description 5
- 239000002904 solvent Substances 0.000 description 5
- 239000002562 thickening agent Substances 0.000 description 5
- 238000011156 evaluation Methods 0.000 description 4
- 239000012467 final product Substances 0.000 description 4
- 239000000796 flavoring agent Substances 0.000 description 4
- 235000019634 flavors Nutrition 0.000 description 4
- 239000003205 fragrance Substances 0.000 description 4
- 239000001738 pogostemon cablin oil Substances 0.000 description 4
- 239000002994 raw material Substances 0.000 description 4
- 238000000222 aromatherapy Methods 0.000 description 3
- 230000000052 comparative effect Effects 0.000 description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 3
- 229960003943 hypromellose Drugs 0.000 description 3
- 230000003860 sleep quality Effects 0.000 description 3
- 241000196324 Embryophyta Species 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 239000012153 distilled water Substances 0.000 description 2
- 230000000147 hypnotic effect Effects 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 239000000932 sedative agent Substances 0.000 description 2
- 230000001624 sedative effect Effects 0.000 description 2
- 239000013589 supplement Substances 0.000 description 2
- 238000011282 treatment Methods 0.000 description 2
- NIXOWILDQLNWCW-UHFFFAOYSA-N Acrylic acid Chemical compound OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- 102220487426 Actin-related protein 2/3 complex subunit 3_K15M_mutation Human genes 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 229920000881 Modified starch Polymers 0.000 description 1
- 206010062519 Poor quality sleep Diseases 0.000 description 1
- 229920002125 Sokalan® Polymers 0.000 description 1
- 230000001133 acceleration Effects 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 229940023476 agar Drugs 0.000 description 1
- 239000000935 antidepressant agent Substances 0.000 description 1
- 229940005513 antidepressants Drugs 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000001914 calming effect Effects 0.000 description 1
- 229960001631 carbomer Drugs 0.000 description 1
- 229940031663 carbomer-974p Drugs 0.000 description 1
- 235000010980 cellulose Nutrition 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 239000003576 central nervous system agent Substances 0.000 description 1
- 238000013329 compounding Methods 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 210000002249 digestive system Anatomy 0.000 description 1
- 208000035475 disorder Diseases 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 206010013663 drug dependence Diseases 0.000 description 1
- 230000005802 health problem Effects 0.000 description 1
- 210000000777 hematopoietic system Anatomy 0.000 description 1
- 239000010977 jade Substances 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 210000000653 nervous system Anatomy 0.000 description 1
- 229940093429 polyethylene glycol 6000 Drugs 0.000 description 1
- 230000029058 respiratory gaseous exchange Effects 0.000 description 1
- 230000033764 rhythmic process Effects 0.000 description 1
- 201000002859 sleep apnea Diseases 0.000 description 1
- 208000019116 sleep disease Diseases 0.000 description 1
- 208000011117 substance-related disease Diseases 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000002485 urinary effect Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/88—Liliopsida (monocotyledons)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/28—Asteraceae or Compositae (Aster or Sunflower family), e.g. chamomile, feverfew, yarrow or echinacea
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/53—Lamiaceae or Labiatae (Mint family), e.g. thyme, rosemary or lavender
- A61K36/532—Agastache, e.g. giant hyssop
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/75—Rutaceae (Rue family)
- A61K36/752—Citrus, e.g. lime, orange or lemon
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0043—Nose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/5005—Wall or coating material
- A61K9/5021—Organic macromolecular compounds
- A61K9/5036—Polysaccharides, e.g. gums, alginate; Cyclodextrin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/5005—Wall or coating material
- A61K9/5021—Organic macromolecular compounds
- A61K9/5036—Polysaccharides, e.g. gums, alginate; Cyclodextrin
- A61K9/5042—Cellulose; Cellulose derivatives, e.g. phthalate or acetate succinate esters of hydroxypropyl methylcellulose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/5089—Processes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M21/00—Other devices or methods to cause a change in the state of consciousness; Devices for producing or ending sleep by mechanical, optical, or acoustical means, e.g. for hypnosis
- A61M21/02—Other devices or methods to cause a change in the state of consciousness; Devices for producing or ending sleep by mechanical, optical, or acoustical means, e.g. for hypnosis for inducing sleep or relaxation, e.g. by direct nerve stimulation, hypnosis, analgesia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M31/00—Devices for introducing or retaining media, e.g. remedies, in cavities of the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/20—Hypnotics; Sedatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2210/00—Anatomical parts of the body
- A61M2210/06—Head
- A61M2210/0618—Nose
Landscapes
- Health & Medical Sciences (AREA)
- Natural Medicines & Medicinal Plants (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Medical Informatics (AREA)
- Alternative & Traditional Medicine (AREA)
- Biotechnology (AREA)
- Botany (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Anesthesiology (AREA)
- Biomedical Technology (AREA)
- Hematology (AREA)
- Heart & Thoracic Surgery (AREA)
- Psychology (AREA)
- Acoustics & Sound (AREA)
- Physics & Mathematics (AREA)
- Pain & Pain Management (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Otolaryngology (AREA)
- Medicinal Preparation (AREA)
Abstract
本发明公开了一种缓释助眠鼻吸。本发明的缓释助眠鼻吸包括助眠缓释微丸和固体架。所述的助眠缓释微丸包括以质量百分比计的复方精油微胶囊粉末5‑15%,粘合剂0.1‑20%,制丸辅料10‑40%,添加剂0.01‑8%及余量的水。其中,助眠缓释微丸的制备方法为:将粘合剂、添加剂溶解于纯水中,所得溶液作为润湿剂;将润湿剂与复方精油微胶囊、制丸辅料混合均匀,制备成软材;采用制丸机将软材制备成微丸,干燥,筛分,即可;所述固体架包括弹性带、链接带、微孔囊,用于放置助眠缓释微丸并将其固定于鼻腔中。本发明所述缓释助眠鼻吸,助眠效果极佳,缓释时间长达12小时以上。
The invention discloses a nasal suction for slow release and sleep aid. The slow-release sleep-aid nasal suction of the present invention comprises sleep-aid slow-release pellets and a solid frame. The sleep-aiding slow-release pellets include 5-15% of compound essential oil microcapsule powder in mass percentage, 0.1-20% of binder, 10-40% of pill-making auxiliary materials, 0.01-8% of additives and the balance of water. Among them, the preparation method of sleep-aiding sustained-release pellets is: dissolving adhesives and additives in pure water, and using the obtained solution as a wetting agent; mixing the wetting agent with compound essential oil microcapsules and pelleting auxiliary materials evenly, and preparing Soft material; the soft material is prepared into pellets by a pill making machine, dried and sieved; the solid frame includes elastic belts, link belts, and microporous capsules, which are used to place sleep-promoting slow-release pellets and fixed in the nasal cavity. The slow-release sleep-aiding nasal suction of the present invention has excellent sleep-aiding effect, and the slow-release time is as long as more than 12 hours.
Description
技术领域technical field
本发明涉及一种缓释助眠鼻吸,属于日用化学技术领域。The invention relates to a slow-release nasal suction for sleep aid, which belongs to the technical field of daily chemistry.
背景技术Background technique
随着现代生活节奏的加快、生活工作压力的加重,现今失眠患者日益增多,失眠已成为人体健康的一种潜在威胁。根据国外学者流行病学调查显示10%-30%的人群遭受失眠的影响。失眠作为世界范围内的常见疾病,对人们日常生活和工作造成了极大困扰,愈发成为当前社会亟需解决的健康问题。With the acceleration of the pace of modern life and the aggravation of the pressure of life and work, there are more and more patients with insomnia, and insomnia has become a potential threat to human health. According to the epidemiological survey of foreign scholars, 10%-30% of the population suffers from insomnia. As a common disease in the world, insomnia has caused great troubles to people's daily life and work, and has become an urgent health problem in the current society.
据统计,慢性失眠的患者约有20%选择使用镇静安眠药物、抗抑郁药来解决失眠问题,长期使用会出现药物依赖、睡眠呼吸暂停、睡眠节奏紊乱、过敏反应,其毒性可影响到神经系统、消化系统、造血系统及泌尿系统等诸多副作用。因此,有不少研究者意识到非药物治疗睡眠障碍的重要意义。关于失眠的非药物治疗方法多种多样,芳香疗法用于镇定平气,安神助眠的使用历史已经非常悠久。随着现代技术进步芳香疗法的应用拓展了精油和香薰等多种剂型,使用更加安全便携。According to statistics, about 20% of patients with chronic insomnia choose to use sedative and hypnotic drugs and antidepressants to solve the insomnia problem. Long-term use will lead to drug dependence, sleep apnea, sleep rhythm disorder, and allergic reactions. The toxicity can affect the nervous system. , digestive system, hematopoietic system and urinary system and many other side effects. Therefore, many researchers are aware of the importance of non-drug treatment of sleep disorders. There are a variety of non-drug treatments for insomnia. Aromatherapy is used to calm the mind and calm the mind. It has a long history of use to help sleep. With the advancement of modern technology, the application of aromatherapy has expanded various dosage forms such as essential oils and aromatherapy, and the use is safer and more portable.
发明内容Contents of the invention
本发明解决的技术问题是:如何规避药物干预助眠的副作用和效果不确定性,从而针对失眠人群开发一种安神助眠的鼻吸产品的技术问题。The technical problem solved by the present invention is: how to avoid the side effects and uncertainty of the effect of drug intervention to help sleep, so as to develop a nasal suction product for calming the nerves and helping sleep for people with insomnia.
为了解决上述技术问题,本发明提供了一种缓释助眠鼻吸,包括助眠缓释微丸和固体架,所述助眠缓释微丸包括以质量分数计的复方精油微胶囊粉末5-15%、粘合剂0.1-20%、制丸辅料10-40%、添加剂0.01-8%及余量的水;所述复方精油微胶囊粉末的成分包括复方精油,所述复方精油包括晚香玉精油、广藿香精油、一枝黄花精油和橘叶油精油。In order to solve the above-mentioned technical problems, the present invention provides a kind of sustained-release sleep-aid nasal suction, comprising sleep-aid sustained-release pellets and a solid frame, the sleep-aid slow-release pellets include compound essential oil microcapsule powder 5- 15%, binder 0.1-20%, pelleting auxiliary material 10-40%, additive 0.01-8% and the rest of water; the composition of the compound essential oil microcapsule powder includes compound essential oil, and the compound essential oil includes tuberose Jade essential oil, patchouli essential oil, goldenrod essential oil and orange leaf essential oil.
优选地,所述复方精油微胶囊粉末中复方精油的含量为0.5-5%;所述复方精油中晚香玉精油、广藿香精油、一枝黄花精油和橘叶油精油的质量比为5-10:1-4:1-4:0.5-3。Preferably, the content of compound essential oil in the compound essential oil microcapsule powder is 0.5-5%; the mass ratio of tuberose essential oil, patchouli essential oil, goldenrod essential oil and orange leaf oil in the compound essential oil is 5- 10:1-4:1-4:0.5-3.
优选地,所述的粘合剂为羟丙甲纤维素K4M、羟丙甲纤维素K15M或羟丙甲纤维素K100M中的一种或几种的复配;所述的制丸辅料为微晶纤维素、乳糖、预胶化淀粉、玉米淀粉、羧甲基纤维素钠或甘露醇中的一种或几种的复配;所述的添加剂为琼脂、泊洛沙姆407、卡波姆974P NF、卡波姆971P NF或聚乙二醇6000中的一种或几种的复配。Preferably, the binder is a combination of one or more of hypromellose K4M, hypromellose K15M or hypromellose K100M; the pelletizing auxiliary material is microcrystalline A compound of one or more of cellulose, lactose, pregelatinized starch, corn starch, sodium carboxymethylcellulose or mannitol; the additives are agar, poloxamer 407, carbomer 974P One or more of NF, Carbomer 971P NF or polyethylene glycol 6000.
优选地,所述助眠缓释微丸的制备方法包括:Preferably, the preparation method of the sleep-aiding sustained-release pellets comprises:
将粘合剂、添加剂溶解于纯水中,所得溶液作为润湿剂;将润湿剂与复方精油微胶囊粉末、制丸辅料混合均匀,制备成软材;采用制丸机将软材制备成微丸,干燥,筛分,即可;所述润湿剂的浓度0.05-0.1%。The adhesive and additives are dissolved in pure water, and the resulting solution is used as a wetting agent; the wetting agent is mixed with the compound essential oil microcapsule powder and pelletizing auxiliary materials evenly to prepare a soft material; the soft material is prepared by using a pellet machine The pellets can be dried and sieved; the concentration of the wetting agent is 0.05-0.1%.
优选地,所述复方精油微胶囊粉末的制备方法包括:将复方精油、吐温80和司盘60加入到海藻酸钠的水溶液,搅拌均匀后得到浑浊液,将壳聚糖醋酸溶液滴加到上述浑浊液中,通过复凝聚法制得复方油微胶囊乳液,将其冷冻干燥得复方精油微胶囊粉末。Preferably, the preparation method of the compound essential oil microcapsule powder comprises: adding the compound essential oil, Tween 80 and Span 60 to the aqueous solution of sodium alginate, stirring evenly to obtain a turbid liquid, adding the chitosan acetic acid solution dropwise to The compound oil microcapsule emulsion is prepared by complex coacervation method in the above turbid liquid, which is freeze-dried to obtain the compound essential oil microcapsule powder.
优选地,所述的复方精油、吐温80和司盘60的质量比为5:15:2,所述海藻酸钠的水溶液中海藻酸钠的质量浓度为1-2%;所述壳聚糖醋酸溶液中壳聚糖的质量浓度为2-3%;所述复方精油、海藻酸钠的水溶液和壳聚糖醋酸溶液的比例为5g:2L:1L。Preferably, the mass ratio of the compound essential oil, Tween 80 and Span 60 is 5:15:2, and the mass concentration of sodium alginate in the aqueous solution of sodium alginate is 1-2%; the chitosan The mass concentration of chitosan in the sugar-acetic acid solution is 2-3%; the ratio of the compound essential oil, the aqueous solution of sodium alginate and the chitosan-acetic acid solution is 5g:2L:1L.
优选地,所述固体架由弹性带、链接带和微孔囊组成;所述助眠缓释微丸设于具有密封结构的微孔囊中。Preferably, the solid frame is composed of elastic belts, link belts and microporous capsules; the sleep-aiding sustained-release pellets are arranged in the microporous capsules with a sealed structure.
优选地,所述的弹性带是由金属或高分子材料中的至少一种材料制成,长度为2-5毫米;所述的链接带是由金属或高分子材料中的至少一种材料制成,长度为3-6毫米。Preferably, the elastic belt is made of at least one material of metal or polymer material, and has a length of 2-5 mm; the link belt is made of at least one material of metal or polymer material into, the length is 3-6 mm.
优选地,所述的微孔囊是由金属或高分子材料中的至少一种材料制成,所述的微孔囊为直径3-5毫米的球形,所述微孔囊的表面靠近链接带的一侧设有均匀分布的微孔。Preferably, the microporous capsule is made of at least one material in metal or polymer material, the microporous capsule is spherical with a diameter of 3-5 mm, and the surface of the microporous capsule is close to the linking band One side is provided with evenly distributed micropores.
优选地,所述微孔的直径设为0.5毫米。Preferably, the diameter of the micropore is set to 0.5 mm.
本发明与现有技术相比,具有如下有益效果:Compared with the prior art, the present invention has the following beneficial effects:
1.本发明的一种缓释助眠鼻吸,直接置于鼻腔,通过正常呼吸即可作用于人体,植物精油的生物利用率较高,且具有助眠效果极佳、使用方便舒适、便于携带等优点,适合各年龄段人群使用;1. A slow-release sleep-aiding nasal suction of the present invention is directly placed in the nasal cavity and can act on the human body through normal breathing. The bioavailability of plant essential oils is high, and it has excellent sleep-aiding effect, convenient and comfortable to use, and easy to use. Portable and other advantages, suitable for people of all ages to use;
2.本发明的一种缓释助眠鼻吸,以天然植物精油配方为主要原料,安全高效,避免了镇静催眠类药物的副作用,具有极高的推广应用价值;2. A slow-release sleep aid nasal suction of the present invention, which uses natural plant essential oil formula as the main raw material, is safe and efficient, avoids the side effects of sedative and hypnotic drugs, and has extremely high promotion and application value;
3.本发明的缓释助眠鼻吸的制备方法简单,易操作,成本低。3. The preparation method of the slow-release sleep aid nasal suction of the present invention is simple, easy to operate, and low in cost.
附图说明Description of drawings
图1为本发明的一种缓释助眠鼻吸的结构示意图。Fig. 1 is a structural schematic diagram of a slow-release sleep-aid nasal suction of the present invention.
具体实施方式detailed description
为使本发明更明显易懂,兹以优选实施例,并配合附图作详细说明如下。以下实施例中所用的各种原料或试剂的规格及生产厂家的信息如表1所示In order to make the present invention more comprehensible, preferred embodiments are described in detail below with accompanying drawings. The specifications of various raw materials or reagents used in the following examples and the information of the manufacturer are shown in Table 1
表1原料试剂规格及生产厂家Table 1 Specifications and manufacturers of raw material reagents
以下实施例中所用的仪器设备的型号及生产厂家的信息如表2所示:The model of instrument equipment used in the following examples and the information of manufacturer are as shown in table 2:
表2仪器型号及生产厂家Table 2 Instrument model and manufacturer
实施例1Example 1
(1)复方精油微胶囊的制备(1) Preparation of Compound Essential Oil Microcapsules
将晚香玉、广藿香油、一枝黄花、橘叶油按照质量份量比为5:2:2:1混合均匀,取混合后的复方精油5g、15g吐温80、2g司盘60加入到2L 1%海藻酸钠水溶液中,搅拌均匀后,将1L 1%壳聚糖-醋酸水溶液滴加到上述浑浊液中,通过复凝聚法制得复方精油微胶囊乳液,再经冷冻干燥得精油微胶囊粉末。(其中1%海藻酸钠水溶液配置:精密称取2g海藻酸钠的固体粉末,加入100ml蒸馏水中,常温使用搅拌器搅拌1~2h,溶解后,再继续加入100ml的蒸馏水,持续性搅拌,呈均匀粘稠溶液);(1%壳聚糖-醋酸水溶液制备:精密称取1g壳聚糖置烧杯中,加入1%醋酸水溶液100ml于磁力搅拌器下搅拌,30℃加热,直到溶解完全,备用)Mix tuberose, patchouli oil, goldenrod, and orange leaf oil according to the mass-to-volume ratio of 5:2:2:1, and add 5g of compound essential oil, 15g of Tween 80, and 2g of Span 60 to 2L 1% sodium alginate aqueous solution, after stirring evenly, add 1L of 1% chitosan-acetic acid aqueous solution dropwise to the above cloudy liquid, and prepare compound essential oil microcapsule emulsion by complex coacervation method, and then freeze-dry to obtain essential oil microcapsule powder . (The configuration of 1% sodium alginate aqueous solution: accurately weigh 2g of sodium alginate solid powder, add 100ml of distilled water, stir with a stirrer at room temperature for 1 to 2 hours, after dissolving, continue to add 100ml of distilled water, continue stirring, Uniform viscous solution); (preparation of 1% chitosan-acetic acid aqueous solution: accurately weigh 1g of chitosan and put it in a beaker, add 100ml of 1% acetic acid aqueous solution and stir under a magnetic stirrer, heat at 30°C until completely dissolved, set aside )
(2)助眠缓释微丸的制备(2) Preparation of Sleep-Aiding Sustained Release Pellets
助眠缓释微丸的制备的各组分的配比如表3所示。The proportioning ratio of each component of the preparation of sleep-aiding sustained-release pellets is shown in Table 3.
表3助眠缓释微丸各组分配比表Table 3 The distribution ratio of each component of Zhumian Sustained Release Pellets
将粘合剂(HPMC K4M)和添加剂(泊洛沙姆407)溶解于纯水中,所得溶液作为润湿剂;再将润湿剂与精油微胶囊和制丸辅料混合均匀,制备成软材;采用制丸机将所述软材制备成微丸,干燥,筛分,即得。Dissolve the binder (HPMC K4M) and additive (poloxamer 407) in pure water, and the resulting solution is used as a wetting agent; then the wetting agent is mixed with essential oil microcapsules and pelleting auxiliary materials to prepare a soft material ; The soft material is prepared into pellets by using a pellet machine, dried and sieved to obtain the product.
(3)缓释助眠鼻吸的制备(3) Preparation of slow-release nasal suction for sleep aid
将制备好的助眠缓释微丸放置于固体架的微孔囊中,将鼻吸密封包装即可。缓释助眠鼻吸的具体结构如图1所示,其包括助眠缓释微丸和固体架;固体架由弹性带、链接带和微孔囊组成;助眠缓释微丸设于具有密封结构的微孔囊中。The prepared sleep-aiding sustained-release pellets are placed in the microporous capsules of the solid frame, and the nasal suction is sealed and packaged. The specific structure of the slow-release sleep-aid nasal suction is shown in Figure 1, which includes sleep-aid slow-release pellets and a solid frame; the solid frame is composed of elastic bands, link belts and microporous capsules; the sleep-aid slow-release pellets are located In the microporous capsule of the structure.
其中,弹性带是由金属或高分子材料中的至少一种材料制成,长度为2-5毫米;链接带是由金属或高分子材料中的至少一种材料制成,长度为3-6毫米;微孔囊是由金属或高分子材料中的至少一种材料制成,微孔囊为直径3-5毫米的球形,微孔囊的表面靠近链接带的一侧设有均匀分布的微孔;微孔的直径设为0.5毫米。Among them, the elastic belt is made of at least one material of metal or polymer material, and the length is 2-5 mm; the link belt is made of at least one material of metal or polymer material, and the length is 3-6 mm. mm; the microporous capsule is made of at least one material in metal or polymer material, the microporous capsule is spherical with a diameter of 3-5 mm, and the surface of the microporous capsule is provided with evenly distributed microporous hole; the diameter of the microhole is set to 0.5 mm.
对比实施例1Comparative Example 1
(1)微胶囊粉末的制备(1) Preparation of microcapsule powder
将15g吐温80、2g司盘60加入到2L 1%海藻酸钠水溶液中,搅拌均匀后,将1L 1%壳聚糖-醋酸水溶液滴加到上述浑浊液中,通过复凝聚法制得微胶囊乳液,再经冷冻干燥得微胶囊粉末。Add 15g of Tween 80 and 2g of Span 60 into 2L of 1% sodium alginate aqueous solution, stir evenly, then drop 1L of 1% chitosan-acetic acid aqueous solution into the above cloudy solution, and prepare microcapsules by complex coacervation method emulsion, and then freeze-dried to obtain microcapsule powder.
(2)助眠缓释微丸的制备(2) Preparation of Sleep-Aiding Sustained Release Pellets
助眠缓释微丸的制备的各组分的配比如表4所示。The proportioning ratio of each component of the preparation of sleep-aiding sustained-release pellets is shown in Table 4.
表4助眠缓释微丸各组分配比表Table 4 The distribution ratio of each component of Zhumian Sustained Release Pellets
将粘合剂(HPMC K4M)和添加剂(泊洛沙姆407)溶解于纯水中,所得溶液作为润湿剂;再将润湿剂与微胶囊空壳和制丸辅料混合均匀,制备成软材;采用制丸机将所述软材制备成微丸,干燥,筛分,即得。The binder (HPMC K4M) and the additive (poloxamer 407) are dissolved in pure water, and the resulting solution is used as a wetting agent; then the wetting agent is mixed with the microcapsule empty shell and pelletizing auxiliary materials to prepare a soft material; the soft material is prepared into pellets by using a pelletizing machine, dried and sieved to obtain final product.
(3)缓释助眠鼻吸的制备(3) Preparation of slow-release nasal suction for sleep aid
将制备好的助眠缓释微丸放置于固体架的微孔囊中,将鼻吸密封包装即可。The prepared sleep-aiding sustained-release pellets are placed in the microporous capsules of the solid frame, and the nasal suction is sealed and packaged.
实施例2Example 2
(1)复方精油微胶囊粉末的制备(1) Preparation of compound essential oil microcapsule powder
将晚香玉、广藿香油、一枝黄花、橘叶油按照质量份量比为5:3.5:1:0.5混合均匀,取混合后的复方精油5g、15g吐温80、2g司盘60加入到2L 1%海藻酸钠水溶液中,搅拌均匀后,将1L 1%壳聚糖-醋酸水溶液滴加到上述浑浊液中,通过复凝聚法制得复方精油微胶囊乳液,再经冷冻干燥得精油微胶囊粉末。Mix tuberose, patchouli oil, goldenrod, and orange leaf oil according to the mass-to-volume ratio of 5:3.5:1:0.5. Take 5g of the mixed compound essential oil, 15g of Tween 80, and 2g of Span 60 and add it to 2L 1% sodium alginate aqueous solution, after stirring evenly, add 1L of 1% chitosan-acetic acid aqueous solution dropwise to the above cloudy liquid, and prepare compound essential oil microcapsule emulsion by complex coacervation method, and then freeze-dry to obtain essential oil microcapsule powder .
(2)助眠缓释微丸的制备(2) Preparation of Sleep-Aiding Sustained Release Pellets
助眠缓释微丸的制备的各组分的配比如表5所示。The proportioning ratio of each component of the preparation of sleep-aiding sustained-release pellets is shown in Table 5.
表5助眠缓释微丸各组分配比表Table 5 The distribution ratio of each component of Zhumian Sustained Release Pellets
将粘合剂(HPMC K4M)和添加剂(泊洛沙姆407)溶解于纯水中,所得溶液作为润湿剂;再将润湿剂与微胶囊空壳和制丸辅料混合均匀,制备成软材;采用制丸机将所述软材制备成微丸,干燥,筛分,即得。The binder (HPMC K4M) and the additive (poloxamer 407) are dissolved in pure water, and the resulting solution is used as a wetting agent; then the wetting agent is mixed with the microcapsule empty shell and pelletizing auxiliary materials to prepare a soft material; the soft material is prepared into pellets by using a pelletizing machine, dried and sieved to obtain final product.
(3)缓释助眠鼻吸的制备(3) Preparation of slow-release nasal suction for sleep aid
将制备好的助眠缓释微丸放置于固体架的微孔囊中,将鼻吸密封包装即可。The prepared sleep-aiding sustained-release pellets are placed in the microporous capsules of the solid frame, and the nasal suction is sealed and packaged.
实施例3Example 3
(1)复方精油微胶囊粉末的制备(1) Preparation of compound essential oil microcapsule powder
将晚香玉、广藿香油、一枝黄花、橘叶油按照质量份量比为3:3:3:1混合均匀,取混合后的复方精油5g、15g吐温80、2g司盘60加入到2L 1%海藻酸钠水溶液中,搅拌均匀后,将1L 1%壳聚糖-醋酸水溶液滴加到上述浑浊液中,通过复凝聚法制得复方精油微胶囊乳液,再经冷冻干燥得精油微胶囊粉末。Mix tuberose, patchouli oil, goldenrod, and orange leaf oil according to the mass-to-volume ratio of 3:3:3:1, and add 5g of compound essential oil, 15g of Tween 80, and 2g of Span 60 to 2L 1% sodium alginate aqueous solution, after stirring evenly, add 1L of 1% chitosan-acetic acid aqueous solution dropwise to the above cloudy liquid, and prepare compound essential oil microcapsule emulsion by complex coacervation method, and then freeze-dry to obtain essential oil microcapsule powder .
(2)助眠缓释微丸的制备(2) Preparation of Sleep-Aiding Sustained Release Pellets
助眠缓释微丸的制备的各组分的配比如表6所示。The proportioning ratio of each component of the preparation of sleep-aiding sustained-release pellets is shown in Table 6.
表6助眠缓释微丸各组分配比表Table 6 The distribution ratio table of each component of Zhumian Sustained Release Pellets
将粘合剂(HPMC K4M)和添加剂(泊洛沙姆407)溶解于纯水中,所得溶液作为润湿剂;再将润湿剂与微胶囊空壳和制丸辅料混合均匀,制备成软材;采用制丸机将所述软材制备成微丸,干燥,筛分,即得。The binder (HPMC K4M) and the additive (poloxamer 407) are dissolved in pure water, and the resulting solution is used as a wetting agent; then the wetting agent is mixed with the microcapsule empty shell and pelletizing auxiliary materials to prepare a soft material; the soft material is prepared into pellets by using a pelletizing machine, dried and sieved to obtain final product.
(3)缓释助眠鼻吸的制备(3) Preparation of slow-release nasal suction for sleep aid
将制备好的助眠缓释微丸放置于固体架的微孔囊中,将鼻吸密封包装即可。The prepared sleep-aiding sustained-release pellets are placed in the microporous capsules of the solid frame, and the nasal suction is sealed and packaged.
实施例4Example 4
(1)复方精油微胶囊粉末的制备(1) Preparation of compound essential oil microcapsule powder
将晚香玉、广藿香油、一枝黄花、橘叶油按照质量份量比为2.5:2.5:2.5:2.5混合均匀,取混合后的复方精油5g、15g吐温80、2g司盘60加入到2L 1%海藻酸钠水溶液中,搅拌均匀后,将1L 1%壳聚糖-醋酸水溶液滴加到上述浑浊液中,通过复凝聚法制得复方精油微胶囊乳液,再经冷冻干燥得精油微胶囊粉末。Mix tuberose, patchouli oil, goldenrod, and orange leaf oil according to the mass-to-volume ratio of 2.5:2.5:2.5:2.5. Take 5g of the mixed compound essential oil, 15g of Tween 80, and 2g of Span 60 and add it to 2L 1% sodium alginate aqueous solution, after stirring evenly, add 1L of 1% chitosan-acetic acid aqueous solution dropwise to the above cloudy liquid, and prepare compound essential oil microcapsule emulsion by complex coacervation method, and then freeze-dry to obtain essential oil microcapsule powder .
(2)助眠缓释微丸的制备(2) Preparation of Sleep-Aiding Sustained Release Pellets
助眠缓释微丸的制备的各组分的配比如表7所示。The proportioning ratio of each component for the preparation of sleep-aiding sustained-release pellets is shown in Table 7.
表7助眠缓释微丸各组分配比表Table 7 The distribution ratio of each component of Zhumian Sustained Release Pellets
将粘合剂(HPMC K4M)和添加剂(泊洛沙姆407)溶解于纯水中,所得溶液作为润湿剂;再将润湿剂与微胶囊空壳和制丸辅料混合均匀,制备成软材;采用制丸机将所述软材制备成微丸,干燥,筛分,即得。The binder (HPMC K4M) and the additive (poloxamer 407) are dissolved in pure water, and the resulting solution is used as a wetting agent; then the wetting agent is mixed with the microcapsule empty shell and pelletizing auxiliary materials to prepare a soft material; the soft material is prepared into pellets by using a pelletizing machine, dried and sieved to obtain final product.
(3)缓释助眠鼻吸的制备(3) Preparation of slow-release nasal suction for sleep aid
将制备好的助眠缓释微丸放置于固体架的微孔囊中,将鼻吸密封包装即可。The prepared sleep-aiding sustained-release pellets are placed in the microporous capsules of the solid frame, and the nasal suction is sealed and packaged.
实施例5Example 5
缓释助眠鼻吸的功效评价:Efficacy evaluation of slow-release nasal suction for sleep aid:
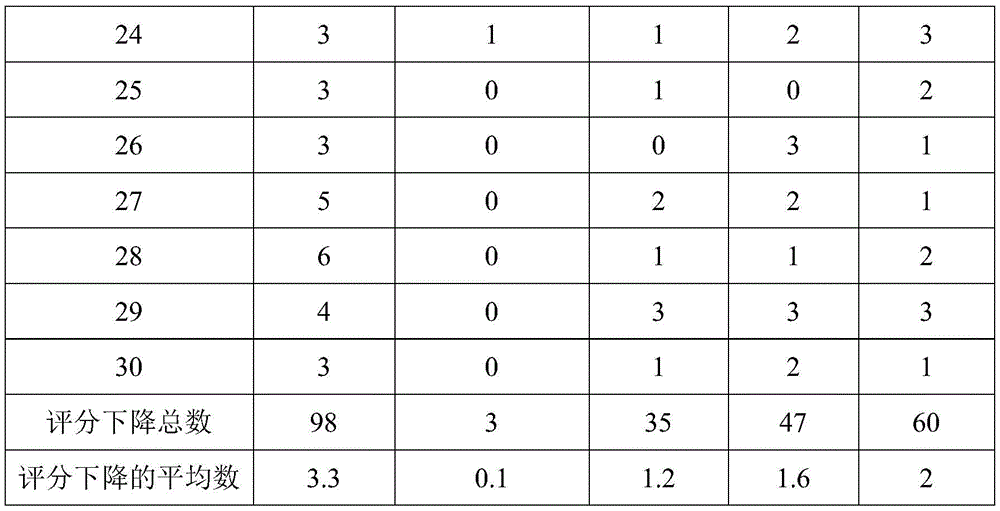
利用匹茨堡睡眠指数量表(PSQI)主观评价受试者的睡眠质量。PSQI是1989年由美国匹兹堡大学Buysse博士等人编制,是权威的国际上常用睡眠实验评估量表。当此表的评分下降≥3分,判定为主观睡眠质量改善。The sleep quality of the subjects was subjectively evaluated by the Pittsburgh Sleep Index (PSQI). PSQI was compiled by Dr. Buysse and others from the University of Pittsburgh in 1989. It is an authoritative and commonly used sleep test evaluation scale in the world. When the score of this table drops ≥ 3 points, it is judged that the subjective sleep quality has improved.
根据匹兹堡睡眠指数量表选择得分在11≤X≤15分之间的睡眠质量较差人群(该人群过去一周未曾使用过中枢神经系统药物并且身体无其它疾病),最终筛选出每组30例受试者。受试者每天晚上睡觉时将睡眠鼻吸佩戴在鼻孔,连续使用7天,在根据匹兹堡睡眠指数量表做出主观评价。评价结果如表8所示。According to the Pittsburgh Sleep Index Scale, select the poor sleep quality group with scores between 11≤X≤15 points (this group has not used central nervous system drugs in the past week and has no other diseases in the body), and finally screened out 30 patients in each group. tester. The subjects wore the sleep suction in their nostrils every night when they went to bed, used it for 7 consecutive days, and made a subjective evaluation according to the Pittsburgh Sleep Index Scale. The evaluation results are shown in Table 8.
表8受试者使用睡眠鼻吸7天前后PSQI评分下降情况Table 8 The decline of PSQI scores before and after 7 days of using sleep snorting
从表8的实施例1和对比实施例1的实验结果可以看出,含精油的助眠鼻吸表现出的好的助眠效果,不含精油的鼻吸不具有助眠效果。从表8的实施例1和对比实施例1、实施例2、实施例3、实施例4的实验结果可以看出,精油复配比例也会助眠效果产生较大影响,建议最佳的使用比例为5:2:2:1。From the experimental results of Example 1 and Comparative Example 1 in Table 8, it can be seen that the sleep-promoting nasal suction containing essential oils has a good sleep-promoting effect, and the nasal suction without essential oil has no sleep-promoting effect. From the experimental results of Example 1 and Comparative Example 1, Example 2, Example 3, and Example 4 in Table 8, it can be seen that the compounding ratio of essential oils will also have a greater impact on the sleep-promoting effect, and the best use is recommended The ratio is 5:2:2:1.
以上所述,仅为本发明的较佳实施例,并非对本发明任何形式上和实质上的限制,应当指出,对于本技术领域的普通技术人员,在不脱离本发明的前提下,还将可以做出若干改进和补充,这些改进和补充也应视为本发明的保护范围。The foregoing is only a preferred embodiment of the present invention, and is not intended to limit the present invention in any form and in essence. Several improvements and supplements are made, and these improvements and supplements should also be regarded as the protection scope of the present invention.
Claims (8)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111180413.9A CN113813245B (en) | 2021-10-11 | 2021-10-11 | Slow-release sleep-aiding snuff |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111180413.9A CN113813245B (en) | 2021-10-11 | 2021-10-11 | Slow-release sleep-aiding snuff |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN113813245A CN113813245A (en) | 2021-12-21 |
| CN113813245B true CN113813245B (en) | 2022-12-09 |
Family
ID=78920211
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202111180413.9A Active CN113813245B (en) | 2021-10-11 | 2021-10-11 | Slow-release sleep-aiding snuff |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113813245B (en) |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007094000A2 (en) * | 2006-02-15 | 2007-08-23 | Botanocap Ltd. | Applications of microencapsulated essential oils |
| WO2016141754A1 (en) * | 2015-03-12 | 2016-09-15 | 施敬东 | Nerve-soothing and sleep-aiding patch and preparation method therefor |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6550474B1 (en) * | 1997-01-29 | 2003-04-22 | Cns, Inc. | Microencapsulated fragrances and methods of coating microcapsules |
| CN2757844Y (en) * | 2004-12-20 | 2006-02-15 | 健康元药业集团股份有限公司 | Miniature nasal suction device |
| CN203677786U (en) * | 2013-11-14 | 2014-07-02 | 吴胜群 | Nasal aspirator |
| TWM484432U (en) * | 2014-02-13 | 2014-08-21 | Sundex Corp | Sleep aid patch |
| GB2577747A (en) * | 2018-10-05 | 2020-04-08 | This Works Products Ltd | Fragrance Compositions |
-
2021
- 2021-10-11 CN CN202111180413.9A patent/CN113813245B/en active Active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007094000A2 (en) * | 2006-02-15 | 2007-08-23 | Botanocap Ltd. | Applications of microencapsulated essential oils |
| WO2016141754A1 (en) * | 2015-03-12 | 2016-09-15 | 施敬东 | Nerve-soothing and sleep-aiding patch and preparation method therefor |
Also Published As
| Publication number | Publication date |
|---|---|
| CN113813245A (en) | 2021-12-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| DE3780222T2 (en) | PHARMACEUTICAL COMPOSITION WITH CONTROLLED ACTIVE SUBSTANCE RELEASE. | |
| CA1296633C (en) | Controlled release bases for pharmaceuticals | |
| US4814179A (en) | Floating sustained release therapeutic compositions | |
| CN1257711C (en) | Butyl benzene phthalein soft capsule and its preparation method | |
| RS50104B (en) | NEW ORAL FORMULATION FOR 5-TH4 AGONISTS OR ANTAGONISTS | |
| CN113813245B (en) | Slow-release sleep-aiding snuff | |
| CN102138911A (en) | Divalproex sodium sustained release tablets and preparation method thereof | |
| CN116421573B (en) | Topiroxostat sustained release preparation and preparation method thereof | |
| CN101536989B (en) | Metformin hydrochloride intragastric floating sustained-release tablet and preparation method thereof | |
| CN111557923A (en) | Controllable timing zero-order drug release system and preparation method thereof | |
| CA2948852C (en) | Orally administrable formulations for the controlled release of a pharmacologically active agent | |
| CN101496790A (en) | Acarbose chewable tablet | |
| CN113274364A (en) | Ramelteon sustained-release preparation and preparation method thereof | |
| CN106619481B (en) | Long-acting 5-HT1A receptor agonist and preparation method thereof | |
| JP2023504409A (en) | Tandospirone pharmaceutical composition and its production method and use | |
| CN115444831B (en) | Epalrestat gastric floating tablet and preparation method thereof | |
| CN103800290B (en) | Aminoglucose hydrochloride pellet preparations and preparation method thereof | |
| EP2087893A1 (en) | Compositions comprising alprazolam for treating primary insomnia and insomnia associated with anxiety states and processes for preparing them | |
| EP4313014B1 (en) | A method for the production of gastroretentive compact matrices for the controlled release of active substances and compact matrices thus obtained | |
| CN113730381B (en) | Lidocaine gel plaster and preparation method thereof | |
| CN116036232A (en) | Nerve-soothing and sleep-aiding medicament containing 5-aminolevulinic acid or derivative thereof, and preparation method and application thereof | |
| CN117462516A (en) | Novel sustained-release pellet, preparation method thereof and pellet tablet thereof | |
| KR20130047262A (en) | Composition containing ascorbic acid for oral administration with controlled release property and method of analyzing the same | |
| CN102266284A (en) | Secnidazole pessary and preparation process thereof | |
| CN115154435B (en) | Micro-bubble type gastric retention sustained and controlled release tablet and preparation method thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |