CN113583467B - A nucleic acid fluorescent dye and its preparation and application - Google Patents
A nucleic acid fluorescent dye and its preparation and application Download PDFInfo
- Publication number
- CN113583467B CN113583467B CN202110958363.6A CN202110958363A CN113583467B CN 113583467 B CN113583467 B CN 113583467B CN 202110958363 A CN202110958363 A CN 202110958363A CN 113583467 B CN113583467 B CN 113583467B
- Authority
- CN
- China
- Prior art keywords
- compound
- formula
- fluorescent dye
- sybr green
- compound shown
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B23/00—Methine or polymethine dyes, e.g. cyanine dyes
- C09B23/02—Methine or polymethine dyes, e.g. cyanine dyes the polymethine chain containing an odd number of >CH- or >C[alkyl]- groups
- C09B23/04—Methine or polymethine dyes, e.g. cyanine dyes the polymethine chain containing an odd number of >CH- or >C[alkyl]- groups one >CH- group, e.g. cyanines, isocyanines, pseudocyanines
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6844—Nucleic acid amplification reactions
- C12Q1/6851—Quantitative amplification
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N27/00—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means
- G01N27/26—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means by investigating electrochemical variables; by using electrolysis or electrophoresis
- G01N27/416—Systems
- G01N27/447—Systems using electrophoresis
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
- C09K2211/1037—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom with sulfur
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Engineering & Computer Science (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Zoology (AREA)
- Analytical Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Immunology (AREA)
- Biochemistry (AREA)
- Physics & Mathematics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Wood Science & Technology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Microbiology (AREA)
- Biophysics (AREA)
- General Engineering & Computer Science (AREA)
- Biotechnology (AREA)
- Genetics & Genomics (AREA)
- Materials Engineering (AREA)
- Electrochemistry (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
- Investigating Or Analysing Biological Materials (AREA)
Abstract
Description
技术领域technical field
本发明涉及荧光染料领域,特别是涉及一种新型的核酸荧光染料。The present invention relates to the field of fluorescent dyes, in particular to a novel nucleic acid fluorescent dye.
背景技术Background technique
荧光染料,指吸收某一波长的激发光后可以发射出特定波长的荧光物质。将荧光染料与不发射荧光的物质以某种方式结合,可以有选择性地将该物质的化学信号转变 为易被分析仪器测量到的荧光信号,从而实现对特定目标的荧光检测。该检测方法具 有灵敏性高,选择性好,可实现可视化以及生物体内实时、非破坏性在线监测等优势, 因此在生物分析领域得到广泛的应用。Fluorescent dyes refer to fluorescent substances that can emit specific wavelengths after absorbing excitation light of a certain wavelength. Combining a fluorescent dye with a substance that does not emit fluorescence can selectively convert the chemical signal of the substance into a fluorescent signal that can be easily measured by analytical instruments, thereby realizing the fluorescence detection of a specific target. The detection method has the advantages of high sensitivity, good selectivity, visualization, and real-time, non-destructive online monitoring in vivo, so it has been widely used in the field of biological analysis.
核酸(如DNA及RNA)是生命体遗传信息的载体,由于其携带信息量大,并且 在细胞内含量相对稳定经常被作为目标分析物。目前,荧光检测是实现核酸分析的主 流方法。除通过共价偶联标记荧光染料外,非共价结合的“发光探针”(即核酸荧光染 料)是核酸定性定量分析最有利的手段。荧光染料可与核酸分子非共价结合,通过结 合前后自身荧光性质的巨大差异实现对靶标核酸分子的检测。这类荧光染料被广泛用 于各种状态下,如溶液、凝胶电泳、细胞内及环境中等的核酸分析。目前,市售的核 酸分析荧光染料较多,其中由美国Invitrogen公司销售的SYBR系列染料性能优异, 占据了国际上大部分的核酸染料市场。Nucleic acids (such as DNA and RNA) are carriers of genetic information of living organisms, and are often used as target analytes due to their large amount of information and relatively stable content in cells. Currently, fluorescence detection is the mainstream method for nucleic acid analysis. In addition to labeling fluorescent dyes by covalent coupling, non-covalently bound "luminescent probes" (i.e. nucleic acid fluorescent dyes) are the most advantageous means for qualitative and quantitative analysis of nucleic acids. Fluorescent dyes can be non-covalently bound to nucleic acid molecules, and the detection of target nucleic acid molecules can be achieved through the huge difference in autofluorescence properties before and after binding. These fluorescent dyes are widely used for nucleic acid analysis in a variety of states, such as solution, gel electrophoresis, intracellular and environmental. At present, there are many fluorescent dyes for nucleic acid analysis on the market, among which the SYBR series dyes sold by Invitrogen Corporation of the United States have excellent performance and occupy most of the nucleic acid dyes market in the world.
在SYBR系列的染料中,应用最广的是SYBR Green I,因其具有低背景、低毒性 及超高的灵敏度已被广泛应用于DNA常规分析、DNA凝胶染色以及作为实时定量 PCR(qPCR)常规检测专用的核酸染料。然而,SYBR Green I仍有如下不足:Among the SYBR series of dyes, the most widely used is SYBR Green I, which has been widely used in routine DNA analysis, DNA gel staining and real-time quantitative PCR (qPCR) due to its low background, low toxicity and ultra-high sensitivity. Nucleic acid dyes for routine detection. However, SYBR Green I still has the following shortcomings:
(1)首先,应用于qPCR的染料在PCR(105℃-55℃)和保存期间(-20℃-4℃) 应具有优异的化学稳定性。这就要求染料及其与核酸分子的复合物应具备一定的热稳 定性。此外,qPCR多使用Tris作为缓冲液,低温为碱性高温为微酸性。因此,要求 染料也具备较好的酸碱稳定性。然而,在上述条件下SYBR Green I并不具备良好的 稳定性;(1) First, the dye applied to qPCR should have excellent chemical stability during PCR (105°C-55°C) and storage period (-20°C-4°C). This requires dyes and their complexes with nucleic acid molecules to have certain thermal stability. In addition, qPCR mostly uses Tris as a buffer, and the low temperature is alkaline and the high temperature is slightly acidic. Therefore, the dyes are also required to have good acid-base stability. However, SYBR Green I does not have good stability under the above conditions;
(2)SYBR Green I对PCR过程具有浓度依赖的抑制效果,高浓度的染料使用会 抑制PCR过程;(2) SYBR Green I has a concentration-dependent inhibitory effect on the PCR process, and the use of high-concentration dyes can inhibit the PCR process;
(3)在核酸的常规分析中(包括qPCR以及凝胶电泳成像),染料结合DNA的 序列应偏好低或没有。但据文献报道,SYBR Green I存在较明显的A-T序列偏好(参 见Zipper H,Brunner H,Bernhagen J,Vitzthum F(2004)Nucleic Acids Res 32:103e);(3) In routine analysis of nucleic acids (including qPCR and gel electrophoresis imaging), the sequence of dye-bound DNA should have low or no preference. But according to literature reports, SYBR Green I has obvious A-T sequence preference (see Zipper H, Brunner H, Bernhagen J, Vitzthum F (2004) Nucleic Acids Res 32:103e);
(4)根据Invitrogen公司提供的使用说明,SYBR Green I在DMSO中可保持良 好的稳定性,在水溶液中稳定性差;(4) according to the instructions for use provided by Invitrogen company, SYBR Green I can keep good stability in DMSO, and has poor stability in aqueous solution;
(5)另外,虽然SYBR Green I荧光性能优异,但根据逆合成分析,其合成极其 困难,并且收率极低(小于2%)。此外,由于其原料的价格较高致使SYBR Green I 价格昂贵(规格10000x,1mL的储备液官方售价高达1万元左右),在实际应用中 成本居高不下。(5) In addition, although SYBR Green I has excellent fluorescence properties, its synthesis is extremely difficult and the yield is extremely low (less than 2%) according to retrosynthetic analysis. In addition, due to the high price of its raw materials, SYBR Green I is expensive (the specification is 10,000x, and the official price of 1 mL of the stock solution is as high as about 10,000 yuan), and the cost remains high in practical applications.
尽管Invitrogen公司还售有另一种染料SYBR Safe,价格较低且诱变性低,可以作为SYBR Green I的替代品,但是这种替代的染料水溶性和灵敏度不够理想。Although Invitrogen also sells another dye, SYBR Safe, which is less expensive and less mutagenic and can be used as an alternative to SYBR Green I, this alternative dye is not water-soluble and sensitive enough.
发明内容SUMMARY OF THE INVENTION
针对现有技术中高效的核酸荧光染料(如SYBR Green I)不稳定、高浓度影响 PCR过程、合成困难等缺点,本发明对染料进行结构改造,提供另一种灵敏度高、稳 定性好、原料易得、合成简单、序列偏好低、对PCR的抑制性小的化合物。Aiming at the shortcomings of the high-efficiency nucleic acid fluorescent dyes (such as SYBR Green I) in the prior art, such as instability, high concentration affecting the PCR process, and difficulty in synthesis, the present invention carries out structural modification of the dyes to provide another kind of high sensitivity, good stability, and raw materials. Compounds that are readily available, simple to synthesize, have low sequence preference, and are less inhibitory to PCR.
本发明的技术方案:Technical scheme of the present invention:
本发明要解决的第一个技术问题是提供一种荧光染料,所述荧光染料的结构式如式I所示:The first technical problem to be solved by the present invention is to provide a fluorescent dye, and the structural formula of the fluorescent dye is shown in formula I:
其中,A-为氯离子、溴离子或碘离子。Wherein, A - is chloride ion, bromide ion or iodide ion.
本发明要解决的第二个技术问题是提供上述荧光染料的制备方法,所述制备方法为:将式II所示的化合物与单体M室温下在溶剂中搅拌反应6~24h得混合物,然后 将混合物纯化处理得式I所示的荧光染料;其中,所述单体M为烷胺基取代的丙胺。The second technical problem to be solved by the present invention is to provide a preparation method of the above-mentioned fluorescent dye. The preparation method is as follows: the compound represented by the formula II and the monomer M are stirred and reacted in a solvent at room temperature for 6 to 24 hours to obtain a mixture, and then a mixture is obtained. The mixture is purified to obtain the fluorescent dye shown in formula I; wherein, the monomer M is propylamine substituted with an alkylamino group.
进一步,上述制备方法中,式II所示的化合物与单体M的当量比为1:1~1:5。Further, in the above preparation method, the equivalent ratio of the compound represented by formula II to the monomer M is 1:1 to 1:5.
进一步,上述制备方法中,单体M选自:N,N-二甲基-1,3-二氨基丙烷或3-二乙胺基丙胺。Further, in the above preparation method, the monomer M is selected from: N,N-dimethyl-1,3-diaminopropane or 3-diethylaminopropylamine.
进一步,上述制备方法中,所述溶剂为乙醇或二氯甲烷。Further, in the above preparation method, the solvent is ethanol or dichloromethane.
进一步,上述反应中,搅拌时间为6~12小时。Further, in the above reaction, the stirring time is 6 to 12 hours.
进一步,所述式II所示的化合物采用下述方法制得:Further, the compound shown in the formula II is prepared by the following method:
先制得式III所示的化合物 First prepare the compound represented by formula III
再制得式Ⅳ所示的化合物 The compound represented by formula IV is then prepared
将式Ⅳ所示的化合物与三氯氧磷在溶剂1中于65℃~85℃回流反应得式Ⅴ所示的化合 物其中,所述溶剂1选自乙腈、二氯乙烷或二甲基甲酰胺中的至少一种;The compound represented by formula IV is reacted with phosphorus oxychloride in
式Ⅴ所示的化合物与式III所示的化合物在溶剂2中,以当量比为1:1~1:3于室温下混 合得式II所示的化合物;所述溶剂2为乙醇或二氯甲烷。The compound represented by formula V and the compound represented by formula III are mixed in
进一步,式III所示的化合物采用下述方法制得:用甲基化试剂(如碘甲烷)将2-甲巯基苯并噻唑(含有或不含有取代基)通过甲基化反应即得式III所示的化合物。Further, the compound shown in formula III is prepared by the following method: the 2-methylmercaptobenzothiazole (with or without a substituent) is subjected to a methylation reaction with a methylating reagent (such as methyl iodide) to obtain formula III compounds shown.
进一步,式Ⅳ所示的化合物采用下述方法制得:将2-羟基-4-甲基喹啉与碘苯或苯硼酸 在催化剂的作用下制得式Ⅳ所示的化合物;其中,所述催化剂为醋酸铜、碳酸钾和铜粉。Further, the compound shown in formula IV is prepared by the following method: the compound shown in formula IV is prepared by 2-hydroxy-4-methylquinoline and iodobenzene or phenylboronic acid under the action of a catalyst; The catalysts are copper acetate, potassium carbonate and copper powder.
本发明要解决的第三个技术问题式提供上述荧光染料的用途,其可用在qPCR分析或凝胶电泳成像。The third technical problem to be solved by the present invention is to provide the use of the above-mentioned fluorescent dyes, which can be used in qPCR analysis or gel electrophoresis imaging.
本发明首次发现,当使用式II所示的化合物与N,N-二甲基-1,3-二氨基丙烷等单体 为原料时,仅在室温下搅拌反应6~24h就能够得到一类新型荧光染料(式I),并且 所得荧光染料较SYBR Green I具有更高的核酸分子响应灵敏度,与核酸结合之后具有 更高的热稳定性,同时还能够提高核酸分子的熔解温度。It is found for the first time in the present invention that when the compound represented by formula II and monomers such as N,N-dimethyl-1,3-diaminopropane are used as raw materials, a class of The novel fluorescent dye (Formula I), and the obtained fluorescent dye has higher nucleic acid molecule response sensitivity than SYBR Green I, has higher thermal stability after being combined with nucleic acid, and can also improve the melting temperature of nucleic acid molecules.
另外,本发明荧光染料中的苯并噻唑和喹啉环通过甲川键连接,其中的单键可自由转动,导致该染料分子在自由状态下存在强烈的分子内扭转电荷转移(TICT)。并 且,由于喹啉环上苯基取代基和烷胺基取代基的位阻作用,降低了染料分子聚集堆积 的紧密性,进而导致染料单体本身几乎没有荧光。当该荧光染料与核酸分子结合后, 其中连接苯并噻唑和喹啉环的甲川键受到核酸分子结构的限制而固定,便于共轭结构 的形成,从而可以获得增强的荧光信号。In addition, the benzothiazole and quinoline rings in the fluorescent dye of the present invention are connected by a methine bond, and the single bond therein can rotate freely, resulting in strong intramolecular torsional charge transfer (TICT) in the free state of the dye molecule. Moreover, due to the steric hindrance of the phenyl substituents and alkylamino substituents on the quinoline ring, the compactness of the aggregation and stacking of the dye molecules is reduced, and the dye monomer itself has almost no fluorescence. When the fluorescent dye is combined with a nucleic acid molecule, the methine bond connecting the benzothiazole and the quinoline ring is fixed by the restriction of the nucleic acid molecule structure, which facilitates the formation of a conjugated structure, thereby obtaining an enhanced fluorescent signal.
本发明的有益效果:Beneficial effects of the present invention:
1)本发明提供了一种新型核酸荧光染料的设计,制备该类染料的原料易得、购买渠道众多、成本低廉,同时合成条件简单、容易合成(室温下短时间即可完成反应), 并且无需对中间产物进行额外纯化。1) The present invention provides the design of a novel nucleic acid fluorescent dye, the raw materials for preparing such dyes are easily available, the purchase channels are numerous, the cost is low, and the synthesis conditions are simple and easy to synthesize (the reaction can be completed in a short time at room temperature), and No additional purification of the intermediate product is required.
2)本发明所得荧光染料具有优异的荧光性能和良好的稳定性,可以直接用于普通的PCR缓冲液(甚至纯水中),不需要添加保护试剂;可特异性识别核酸分子的二级 结构;该染料在与核酸分子作用后具有良好热稳定性,可以有效提高核酸分子的熔解 温度。2) The fluorescent dye obtained in the present invention has excellent fluorescence performance and good stability, and can be directly used in common PCR buffers (even pure water) without adding protective reagents; it can specifically identify the secondary structure of nucleic acid molecules ; The dye has good thermal stability after interacting with nucleic acid molecules, and can effectively increase the melting temperature of nucleic acid molecules.
3)本发明所得荧光染料对核酸分子具有低的序列偏好性,并且具有高灵敏度。通过探测染料与核酸分子混合体系的荧光信号可实现对溶液、凝胶、细胞及环境中核酸 分子的高灵敏检测。3) The fluorescent dye obtained in the present invention has low sequence preference for nucleic acid molecules and high sensitivity. The highly sensitive detection of nucleic acid molecules in solution, gel, cell and environment can be achieved by detecting the fluorescent signal of the mixed system of dye and nucleic acid molecule.
附图说明:Description of drawings:
图1为本发明实施例1的化合物c的吸收光谱及荧光光谱。FIG. 1 is the absorption spectrum and fluorescence spectrum of compound c of Example 1 of the present invention.
图2为本发明化合物c(-■-),SYBR Green I(-●-)及SYBR Green I类似物(-▲-)结合不同浓度双链DNA后的荧光强度与相应的双链DNA浓度关系的曲线图。Figure 2 shows the relationship between the fluorescence intensity of the compound c (-■-), SYBR Green I (-●-) and SYBR Green I analogs (-▲-) of the present invention combined with different concentrations of double-stranded DNA and the corresponding double-stranded DNA concentration curve graph.
图3A为本发明化合物c(实线)与SYBR Green I(虚线)结合双链DNA样品dsDNA-2后的荧光强度对温度关系的曲线图,图3B为相应的dsDNA-2的熔解曲线。Figure 3A is a graph showing the relationship between the fluorescence intensity of the compound c (solid line) of the present invention and SYBR Green I (dotted line) combined with a double-stranded DNA sample dsDNA-2 against temperature, and Figure 3B is the corresponding melting curve of dsDNA-2.
图4A为本发明化合物c(实线)与SYBR Green I(虚线)结合双链DNA样品Harpin-1荧光强度对温度关系的曲线图,图4B为相应的Harpin-1的熔解曲线。Figure 4A is a graph showing the relationship between the fluorescence intensity of the compound c (solid line) of the present invention and SYBR Green I (dotted line) binding to double-stranded DNA sample Harpin-1 versus temperature, and Figure 4B is the corresponding melting curve of Harpin-1.
图5A为本发明化合物c(实线)与SYBR Green I(虚线)结合双链DNA样品dsDNA-3荧光强度对温度关系的曲线图,图5B为相应的dsDNA-3的熔解曲线。Figure 5A is a graph showing the relationship between the fluorescence intensity of compound c (solid line) of the present invention and SYBR Green I (dotted line) binding to double-stranded DNA sample dsDNA-3 versus temperature, and Figure 5B is the corresponding melting curve of dsDNA-3.
图6为本发明化合物c与SYBR Green I对不同d(A-T)含量的双链DNA荧光响应变化的柱状图。Figure 6 is a bar graph showing the change of the fluorescence response of compound c and SYBR Green I to double-stranded DNA with different d(A-T) contents.
图7左图为本发明化合物c在0.5、1、2、4μmol/L浓度下的qPCR荧光扩增曲线,图 7右图为SYBR Green I在0.5(0.55x)、1(1.1x)、2(2.2x)、4(4.4x)μmol/L浓度下 的qPCR荧光扩增曲线。The left picture of Fig. 7 is the qPCR fluorescence amplification curve of the compound c of the present invention at the concentration of 0.5, 1, 2, and 4 μmol/L, and the right picture of Fig. 7 is the SYBR Green I at 0.5(0.55x), 1(1.1x), 2 qPCR fluorescence amplification curves at (2.2x) and 4 (4.4x) μmol/L concentrations.
图8上图为1μmol/L本发明化合物c在引物浓度为50、100、200、300nmol/L浓度下的qPCR扩增曲线,图8下图为1μmol/L(1.1x)SYBR Green I在相同条件下的qPCR扩 增曲线。The upper picture of Fig. 8 is the qPCR amplification curve of 1 μmol/L compound c of the present invention at the primer concentrations of 50, 100, 200, and 300 nmol/L, and the lower picture of Fig. 8 is that 1 μmol/L (1.1x) SYBR Green I at the same qPCR amplification curve under conditions.
图9上图为1μmol/L本发明化合物c在引物浓度为50、100、200、300nmol/L浓度下的扩增产物熔解曲线,图8下图为1μmol/L(1.1x)SYBR Green I在相同条件下的扩增产 物熔解曲线。The upper picture of Fig. 9 is the melting curve of the amplification product of 1 μmol/L compound c of the present invention at primer concentrations of 50, 100, 200 and 300 nmol/L, and the lower picture of Fig. 8 is that 1 μmol/L (1.1x) SYBR Green I Melting curves of amplification products under the same conditions.
图10为标准分子量DNA在前染方式下的凝胶成像效果对比图,左图为1μmol/L(1.1x)SYBR Green I以及右图为1μmol/L本发明化合物c。Figure 10 is a comparison chart of the gel imaging effect of standard molecular weight DNA under the pre-staining mode, the left picture is 1 μmol/L (1.1×) SYBR Green I and the right picture is 1 μmol/L of the compound c of the present invention.
图11为标准分子量DNA在后染方式下的凝胶成像效果对比图,左图为1μmol/L(1.1x)SYBR Green I以及右图为1μmol/L本发明化合物c。Figure 11 is a comparison chart of the gel imaging effect of standard molecular weight DNA under post-staining mode, the left picture is 1 μmol/L (1.1×) SYBR Green I and the right picture is 1 μmol/L of compound c of the present invention.
图12左图为1μmol/L(1.1x)SYBR Green I以及1μmol/L本发明化合物c对PCR扩 增产物在前染方式下的凝胶成像效果对比图,图12右图为后染方式下的凝胶成像效果对 比图。The left picture of Fig. 12 is a comparison chart of the gel imaging effect of 1 μmol/L (1.1x) SYBR Green I and 1 μmol/L compound c of the present invention on PCR amplification products in the pre-staining mode, and the right picture of Fig. 12 is the post-staining mode. Comparison of gel imaging effects.
图13为本发明化合物c在55℃检测的荧光强度与经历热循环次数的关系图。FIG. 13 is a graph showing the relationship between the fluorescence intensity detected at 55° C. of the compound c of the present invention and the number of thermal cycles experienced.
图14A为本发明化合物c(-●-)与SYBR Green I(-■-)在纯水中吸光度与时间变化 的关系图,图14B为本发明化合物c(-●-)与SYBR Green I(-■-)在Tris-HCl缓冲溶液中吸光度与时间变化的关系图。Figure 14A is a graph showing the relationship between the absorbance of the present compound c (-•-) and SYBR Green I (-•-) in pure water versus time, and Figure 14B is the present compound c (-•-) and SYBR Green I ( -■-) Graph of absorbance versus time in Tris-HCl buffer solution.
图15化合物c与SYBR Green I的合成路线、成本对比图。Fig. 15 The synthetic route and cost comparison diagram of compound c and SYBR Green I.
具体实施方式Detailed ways
本发明的荧光染料可采用下述制备方式制得,制备方法包括以下步骤:The fluorescent dye of the present invention can be prepared by the following preparation method, and the preparation method comprises the following steps:
1)先用甲基化试剂(如碘甲烷)将2-甲巯基苯并噻唑(含有或不含有取代基)进行甲基化反应,得到化合物a 1) First use methylating reagent (such as methyl iodide) to carry out methylation reaction on 2-methylmercaptobenzothiazole (with or without substituent) to obtain compound a
2)将2-羟基-4-甲基喹啉与苯硼酸在醋酸铜的催化下得到化合物b 2) 2-hydroxy-4-methylquinoline and phenylboronic acid are obtained under the catalysis of copper acetate to obtain compound b
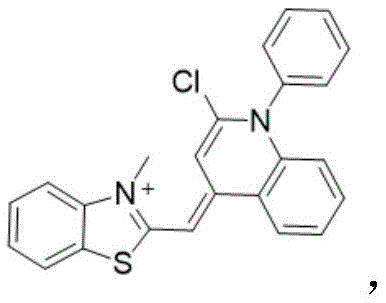
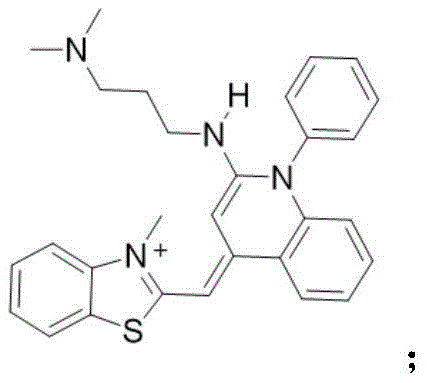
3)将与三氯氧磷混合,得到化合物随后将其与化合物混合得到化合物其与N,N-二甲基-1,3-二氨基丙烷反应,可得到最终的染料结构 3) will Mix with phosphorus oxychloride to give compound Then combine it with the compound compound to mix It reacts with N,N-dimethyl-1,3-diaminopropane to give the final dye structure
具体的反应式如下:The specific reaction formula is as follows:
下面结合实施例对本发明的具体实施方式做进一步的描述,并不因此将本发明限制在所述的实例范围之中。The specific embodiments of the present invention will be further described below with reference to the examples, but the present invention is not limited to the scope of the described examples.
实施例1:本发明核酸荧光染料的制备Example 1: Preparation of the nucleic acid fluorescent dye of the present invention
制备方法如下:The preparation method is as follows:
1)将原料2-甲巯基苯并噻唑(1.5g,8.3mmol,1eq)溶于30mL的干燥乙醇中,加 入碘甲烷(1.4g,10mmol,1.2eq),60℃下反应5小时。反应完成后,移除溶剂,使用 石油醚多次清洗,利用抽滤的方法收集米白色固体产物a:1) The raw material 2-Methylmercaptobenzothiazole (1.5g, 8.3mmol, 1eq) was dissolved in 30mL of dry ethanol, iodomethane (1.4g, 10mmol, 1.2eq) was added, and the reaction was carried out at 60°C for 5 hours. After the reaction is completed, remove the solvent, use petroleum ether to wash repeatedly, utilize the method of suction filtration to collect off-white solid product a:
2)将原料2-羟基-4-甲基喹啉(3.0g,18.8mmol,1eq)及苯硼酸(2.3g,18.8mmol,1eq)和醋酸铜(3.75g,18.8mmol,1eq)溶于150-250mL的二氯甲烷中,常温搅拌反应48-96h后,减压抽滤。将过滤液用水萃取三次,留取二氯甲烷相萃取液,加入无水氯化钙 干燥1-2h。浓缩萃取液,通过SiO2柱纯化后得到白色固体b:2) The raw materials 2-hydroxy-4-methylquinoline (3.0g, 18.8mmol, 1eq), phenylboronic acid (2.3g, 18.8mmol, 1eq) and copper acetate (3.75g, 18.8mmol, 1eq) were dissolved in 150 -250mL of dichloromethane, after stirring at room temperature for 48-96h, suction filtration under reduced pressure. The filtrate was extracted three times with water, and the dichloromethane phase extract was taken, and dried by adding anhydrous calcium chloride for 1-2 hours. The extract was concentrated and purified by SiO column to give a white solid b:
3)将产物b(100mg,0.4mmol,1eq)溶于30mL乙腈中,加入三氯氧磷(184mg,1.2mmol,3eq),在氮气氛围下60-85℃回流12-24h。反应完成后,冷却至室温,移除溶 剂,与化合物a一起溶于20-30mL二氯甲烷,室温下边搅拌边滴加三乙胺至无明显白烟冒 出。室温下搅拌5h后滴加原料N,N-二甲基-1,3-二氨基丙烷(122mg,1.2mmol,3eq), 室温下继续搅拌6-24h,浓缩反应混合物,通过SiO2柱纯化分离,在二氯甲烷和石油醚溶 剂中重结晶得到纯的化合物c。3) Dissolve the product b (100 mg, 0.4 mmol, 1 eq) in 30 mL of acetonitrile, add phosphorus oxychloride (184 mg, 1.2 mmol, 3 eq), and reflux at 60-85 °C for 12-24 h under nitrogen atmosphere. After the reaction is completed, cool to room temperature, remove the solvent, dissolve with compound a in 20-30 mL of dichloromethane, and add triethylamine dropwise with stirring at room temperature until no obvious white smoke emerges. After stirring at room temperature for 5 h, the raw material N,N-dimethyl-1,3-diaminopropane (122 mg, 1.2 mmol, 3 eq) was added dropwise, stirring was continued at room temperature for 6-24 h, the reaction mixture was concentrated, and purified and separated through a SiO 2 column. , and recrystallized in dichloromethane and petroleum ether solvent to obtain pure compound c.
实施例2:化合物c的吸收光谱及荧光光谱Example 2: Absorption spectrum and fluorescence spectrum of compound c
DNA样品购买自生工生物工程(上海)股份有限公司,为两条互补的寡聚核苷酸链,如表1所示。DNA样品经HPLC纯化,按照说明书将DNA适量溶于TE缓冲(pH 7.4, 10mmol/LTris,1mmol/L EDTA)中配置相应浓度的储备液,4℃储存。使用时,分别加 入相同浓度的上述两条单链(Pro-20与Tar-20)即可形成双链DNA(dsDNA-1)。The DNA samples were purchased from Sangon Bioengineering (Shanghai) Co., Ltd., and were two complementary oligonucleotide chains, as shown in Table 1. DNA samples were purified by HPLC, and appropriate amount of DNA was dissolved in TE buffer (pH 7.4, 10 mmol/L Tris, 1 mmol/L EDTA) according to the instructions to prepare a stock solution of corresponding concentration, and stored at 4°C. During use, the two single strands (Pro-20 and Tar-20) of the same concentration were added to form double-stranded DNA (dsDNA-1).
表1Table 1
在TE缓冲中检测化合物c(1μmol/L)的吸收光谱及与dsDNA-1(20bp,100nmol/L)结合后的荧光发射光谱(图1所示)。化合物c的吸收峰最大值为467nm(蓝光),结合 DNA后的荧光最大发射峰位于505nm(绿光)。化合物c可被400nm-500nm区域内的 光源激发,与目前市面上主流的qPCR以及化学发光成像仪的光学通道匹配,这表明该染 料可良好的适配于现有仪器。The absorption spectrum of compound c (1 μmol/L) and the fluorescence emission spectrum after binding with dsDNA-1 (20 bp, 100 nmol/L) were detected in TE buffer (shown in FIG. 1 ). The maximum absorption peak of compound c is 467 nm (blue light), and the fluorescence maximum emission peak after binding to DNA is located at 505 nm (green light). Compound c can be excited by a light source in the range of 400nm-500nm, which matches the optical channel of the mainstream qPCR and chemiluminescence imagers on the market, which shows that the dye can be well adapted to the existing instruments.
实施例3:化合物c用于常规核酸分子定量Example 3: Compound c for routine nucleic acid molecule quantification
DNA样品同实施例2。DNA samples were the same as in Example 2.
对比了化合物c与化合物c的N-H键被正丙基封闭后的结构SYBR Green I以及被甲基封闭后的SYBR Green I类似物对不同 浓度核酸分子的荧光响应灵敏度。在体积为200μL,TE缓冲液中,以5nmol/L为浓度梯 度,分别配置含有0-75nmol/L终浓度的dsDNA-1与终浓度为0.5μmol/L的化合物c、SYBR Green I(0.55x)及SYBR Green I类似物的混合液。使用荧光光谱仪Fluorolog-3(HORIBA, 美国)检测了上述混合物的荧光,如图2所示,以探测到的荧光强度对dsDNA-1浓度作图。 首先可以看出,dsDNA-1浓度为0时,几乎没有检测到荧光信号响应。随着dsDNA浓度 的增加,在一定范围内荧光对DNA浓度变化呈线性;而DNA浓度较高时,响应变为非线 性,信号逐渐趋于饱和。线性变化范围内的上升区域斜率代表染料分子对DNA浓度变化 的响应灵敏度。结果显示,化合物c对5nmol/L的dsDNA-1的响应灵敏度明显高于SYBR Green I及SYBR Green I的类似物。其次,对比相同浓度的染料与dsDNA-1结合饱和后的 荧光强度,化合物c的饱和荧光强度要明显高于SYBR Green I和SYBR Green I类似物。 以上结果表明:相比于SYBRGreen I以及SYBR Green I类似物,化合物c的结构具有独 特的优势,对DNA分子具有更好的结合能力和更高的检测灵敏度。Comparing the structure of compound c and compound c after the NH bond of compound c is blocked by n-propyl group SYBR Green I and SYBR Green I analogs blocked by methyl Fluorescence response sensitivity to different concentrations of nucleic acid molecules. In TE buffer with a volume of 200 μL and a concentration gradient of 5 nmol/L, dsDNA-1 containing a final concentration of 0-75 nmol/L and compound c and SYBR Green I (0.55x ) and a mixture of SYBR Green I analogs. The fluorescence of the above mixture was detected using a fluorescence spectrometer Fluorolog-3 (HORIBA, USA), as shown in Figure 2, and the detected fluorescence intensity was plotted against dsDNA-1 concentration. First, it can be seen that at 0 dsDNA-1 concentration, almost no fluorescence signal response was detected. With the increase of the dsDNA concentration, the fluorescence changes linearly with the DNA concentration in a certain range; however, when the DNA concentration is higher, the response becomes nonlinear and the signal gradually tends to be saturated. The slope of the rising region within the linear variation range represents the sensitivity of the response of the dye molecule to changes in DNA concentration. The results showed that the response sensitivity of compound c to 5nmol/L dsDNA-1 was significantly higher than that of SYBR Green I and SYBR Green I analogs. Secondly, comparing the fluorescence intensity of the same concentration of dye after binding to dsDNA-1, the saturated fluorescence intensity of compound c was significantly higher than that of SYBR Green I and SYBR Green I analogs. The above results show that compared with SYBRGreen I and SYBR Green I analogs, the structure of compound c has unique advantages, and has better binding ability and higher detection sensitivity to DNA molecules.
实施例4:化合物c与核酸分子结合后的热稳定性Example 4: Thermal stability of compound c combined with nucleic acid molecules
DNA样品制备同实施例2,DNA使用序列如表2所示。The preparation of DNA samples is the same as that in Example 2, and the DNA sequence used is shown in Table 2.
表2Table 2
热稳定性测定方案:将含有1μmol/L化合物c和100nmol/L DNA样品,与体积为20 μL,pH=8.3的Tris-HCl缓冲液(含Mg2+)在95℃保持15s随后退火;在45℃保温15min 后,使用StepOnePlusTMqPCR仪(ABI,美国)每隔+0.5℃采集一次荧光信号(包括45℃); 升温至95℃,保温30s(SYBR Green I的热稳定性测试条件与化合物c保持一致)。绘制 化合物c及SYBRGreen I与上述不同DNA样品形成复合物的荧光强度对温度的曲线(图 3A、图4A及图5A),并对每一点的斜率取负倒数后绘制不同DNA样品的熔解曲线(图 3B、图4B及图5B)。测试结果表明:一方面本发明实施例1制备的化合物c与DNA结合 后在升温过程中荧光强度的下降速度(在荧光强度变化发生陡降之前较为平缓的变化区 域)明显慢于SYBR Green I,这表明化合物c与DNA形成的复合物热稳定性较SYBR Green I有显著提升。另一方面对比不同DNA样品熔解曲线的峰值处对应的横坐标(即熔解温度 值,Tm值),化合物c探测到的Tm值也要大于SYBR Green I(如表3所示),进一步证实 证明化合物c与DNA的复合物具有良好的结合能力和热稳定性。Thermal stability assay protocol: The samples containing 1 μmol/L of compound c and 100 nmol/L of DNA were kept at 95 °C for 15 s with a volume of 20 μL, pH=8.3 Tris-HCl buffer (containing Mg 2+ ) and then annealed; After incubating at 45°C for 15min, use the StepOnePlus ™ qPCR instrument (ABI, USA) to collect fluorescence signals at every +0.5°C (including 45°C); raise the temperature to 95°C and hold for 30s (Thermal stability test conditions of SYBR Green I and compounds c remains the same). Plot the fluorescence intensity of compound c and SYBRGreen I and the above-mentioned different DNA samples to form complexes with the curve of temperature (Figure 3A, Figure 4A and Figure 5A), and draw the melting curve of different DNA samples after taking the negative reciprocal of the slope of each point ( 3B, 4B and 5B). The test results show that: on the one hand, after the compound c prepared in Example 1 of the present invention is combined with DNA, the rate of decrease of the fluorescence intensity during the heating process (a relatively gentle change region before the change of the fluorescence intensity changes sharply) is significantly slower than that of SYBR Green I, This indicated that the thermal stability of the complex formed by compound c and DNA was significantly improved compared with SYBR Green I. On the other hand, comparing the abscissas (that is, melting temperature value, Tm value) corresponding to the peaks of the melting curves of different DNA samples, the Tm value detected by compound c is also greater than that of SYBR Green I (as shown in Table 3), and further It was confirmed that the complex of compound c and DNA has good binding ability and thermal stability.
表3table 3
实施例5:化合物c的序列选择性Example 5: Sequence selectivity of compound c
DNA样品制备同实施例2,DNA使用序列如表4所示。The preparation of DNA samples is the same as that in Example 2, and the DNA sequence used is shown in Table 4.
表4Table 4
在体积为1mL,pH=7.4TE缓冲液中分别配置浓度为100nmol/L,含10%腺嘌呤(A)-胸腺嘧啶(T)的DNA样品(AT-1)和含100%腺嘌呤(A)-胸腺嘧啶(T)的DNA样 品(AT-2),并在其中加入终浓度为1μmol/L的化合物c。孵育10min后使用荧光光谱仪 Fluorolog-3(HORIBA,美国)测定了化合物c与两种不同DNA样品形成复合物的荧光强 度,用于对比的SYBR Green I的序列选择性测试条件与之保持一致,结果如图6所示。化 合物c对A-T序列偏好的信号响应差异仅为15%,明显低于SYBR Green I(40%),表明 化合物c对核酸分子的序列偏好性很低,适用于核酸分子的常规检测。The DNA sample (AT-1) containing 10% adenine (A)-thymine (T) and the DNA sample (AT-1) containing 100% adenine (A) were prepared in a volume of 1 mL, pH=7.4 TE buffer with a concentration of 100 nmol/L. )-thymine (T) DNA sample (AT-2), to which compound c was added at a final concentration of 1 μmol/L. After incubation for 10 min, the fluorescence intensity of the complex formed by compound c and two different DNA samples was measured using a fluorescence spectrometer Fluorolog-3 (HORIBA, USA). As shown in Figure 6. The signal response difference of compound c to A-T sequence preference is only 15%, which is significantly lower than that of SYBR Green I (40%), indicating that compound c has very low sequence preference to nucleic acid molecules and is suitable for routine detection of nucleic acid molecules.
实施例6:化合物c在qPCR扩增中的应用Example 6: Application of compound c in qPCR amplification
以实施例1中制备的化合物c为荧光染料,使用StepOnePlusTMqPCR仪(ABI,美国),对沙门氏菌中的invA基因进行扩增。qPCR扩增的引物序列如下:Using Compound c prepared in Example 1 as a fluorescent dye, StepOnePlus ™ qPCR instrument (ABI, USA) was used to amplify the invA gene in Salmonella. The primer sequences amplified by qPCR are as follows:
上游引物:5’-TCCCTTTCCAGTACGCTTCG-3’Upstream primer: 5'-TCCCTTTCCAGTACGCTTCG-3'
下游引物:5’-TCTGGATGGTATGCCCGGTA-3’Downstream primer: 5'-TCTGGATGGTATGCCCGGTA-3'
模板基因获取:使用细菌DNA提取试剂盒(TIANamp Bacteria DNA Kit,货号:DP302, TIANGEN,北京),按照说明书步骤提取沙门氏菌DNA。Template gene acquisition: use a bacterial DNA extraction kit (TIANamp Bacteria DNA Kit, item number: DP302, TIANGEN, Beijing) to extract Salmonella DNA according to the instructions.
qPCR条件:95℃保持20s后55℃保温30s,并在55℃时设定测试荧光强度,接着在72℃延伸30s,共30-40个循环。qPCR conditions: hold at 95°C for 20s, incubate at 55°C for 30s, set the test fluorescence intensity at 55°C, and then extend at 72°C for 30s, for a total of 30-40 cycles.
选用PCR试剂盒(TaKaRa TaqTM HS Low DNA,货号:R090A,TaKaRa,日本)按 照使用说明书配制体积为20μL的反应体系,包括TaqTM HS Low DNA(2x),10μL;上 游引物及下游引物各0.4μL(分别含终浓度为50nmol/L、100nmol/L、200nmol/L及300 nmol/L,DNA);化合物c或用于对比的SYBR Green I,2μL(分别含终浓度为0.5μmol/L、 1μmol/L、2μmol/L及4μmol/L);模板基因,2μL;加入无菌水补齐至终体积20μL。A PCR kit (TaKaRa Taq TM HS Low DNA, Cat. No.: R090A, TaKaRa, Japan) was used to prepare a reaction system with a volume of 20 μL according to the instruction manual, including Taq TM HS Low DNA (2x), 10 μL; upstream primer and downstream primer 0.4 μL each μL (with final concentrations of 50 nmol/L, 100 nmol/L, 200 nmol/L and 300 nmol/L, respectively, DNA); Compound c or SYBR Green I for comparison, 2 μL (with final concentrations of 0.5 μmol/L, 1 μmol/L, 2 μmol/L and 4 μmol/L); template gene, 2 μL; add sterile water to make up to a final volume of 20 μL.
1)比较qPCR中化合物c与SYBR Green I的信号强度1) Compare the signal intensity of compound c and SYBR Green I in qPCR
保持其他条件一致,仅改变化合物c浓度的qPCR荧光扩增曲线(荧光强度对循环个数) 如图7左图所示,用于对比的不同浓度的SYBR Green I如图7右图所示,结果表明不同浓 度的化合物c所监测到的荧光信号变化幅度和强度都要高于相应浓度下的SYBR GreenI。 另外,在较高浓度时,SYBR Green I出现了较明显的浓度抑制效应,相比于相同浓度下的 化合物c,出现CT值滞后(约1个循环)。实验证实了相比于SYBR Green I,化合物c在 较大的浓度范围内均能用于核酸的实时扩增检测,并能够实现更加灵敏的检测。Keeping other conditions the same, the qPCR fluorescence amplification curve (fluorescence intensity versus cycle number) of only changing the concentration of compound c is shown in the left panel of Figure 7, and the different concentrations of SYBR Green I used for comparison are shown in the right panel of Figure 7. The results showed that the amplitude and intensity of the fluorescence signal changes monitored by different concentrations of compound c were higher than those of SYBR GreenI at the corresponding concentrations. In addition, at higher concentrations, SYBR Green I showed a more obvious concentration inhibition effect, and compared with compound c at the same concentration, the CT value lag (about 1 cycle). The experiment confirmed that compared with SYBR Green I, compound c can be used for real-time amplification detection of nucleic acid in a larger concentration range, and can achieve more sensitive detection.
2)比较qPCR中化合物c与SYBR Green I的扩增曲线2) Compare the amplification curves of compound c and SYBR Green I in qPCR
保持其他条件一致(化合物c与SYBR Green I的浓度均为1μmol/L),仅改变引物浓度的qPCR扩增曲线(纵坐标ΔRn对横坐标循环个数)如图8上图所示,用于对比的不同 浓度的SYBR Green I如图8下图所示。结果表明:本发明所合成的化合物c具有较低的扩 增背景,并且在低引物浓度(50nmol/L)时,相比于SYBR Green I也没有明显的浓度依 赖性。Keep other conditions the same (the concentrations of compound c and SYBR Green I are both 1 μmol/L), and only change the qPCR amplification curve of the primer concentration (the ordinate ΔR n versus the number of cycles on the abscissa) as shown in the upper figure of Figure 8, using Different concentrations of SYBR Green I for comparison are shown in the lower panel of Figure 8. The results show that the compound c synthesized by the present invention has a lower amplification background, and also has no obvious concentration dependence compared with SYBR Green I at a low primer concentration (50 nmol/L).
3)化合物c监测PCR扩增子熔解温度3) Compound c monitors the melting temperature of PCR amplicons
据报道,SYBR Green I是监测qPCR扩增完成后扩增产物(即扩增子的熔解温度)的有利工具。测定扩增子的熔解温度能提供是否存在非特异性扩增、扩增子是否单一等有价值的信息。据此,同时测定了改变引物浓度,以本发明中化合物c监测扩增子的熔解曲线(图9上图),以测试以化合物c监测扩增子熔解峰的效果,并与SYBR Green I(图9下 图)进行对比。首先,二者扩增子熔解温度测试结果具有良好的相关性,测定结果一致。 其次,相较于SYBR Green I,化合物c的测试基线更低,表明本发明中实施例1提供的化 合物c能够实现对于扩增子熔解温度的检测。SYBR Green I has been reported to be a useful tool for monitoring the amplification product (ie, the melting temperature of the amplicon) after qPCR amplification is complete. Determining the melting temperature of the amplicon can provide valuable information such as whether there is non-specific amplification and whether the amplicon is single. Accordingly, the concentration of the primers was changed at the same time, and the melting curve of the amplicon was monitored with compound c in the present invention (the upper figure in FIG. 9 ), to test the effect of monitoring the melting peak of the amplicon with compound c, and with SYBR Green I ( Figure 9 bottom) for comparison. First of all, there is a good correlation between the two amplicon melting temperature test results, and the measurement results are consistent. Secondly, compared with SYBR Green I, the test baseline of compound c is lower, indicating that compound c provided in Example 1 of the present invention can detect the melting temperature of amplicon.
实施例7:凝胶染色效果对比实验Example 7: Comparative experiment of gel dyeing effect
据报道,凝胶染色目前主要通过两种方式实现:(1)进行分离前,将染料预先加入DNA 混合液中进行电泳分离后进行荧光成像,即前染法;(2)DNA混合液进行电泳分离后,将凝胶至于荧光染料的水溶液或缓冲液中浸泡一段时间后进行荧光成像,即后染法;两种使用方式应用都非常普遍。将实施例1制备的化合物c(终浓度为1μmol/L)与SYBR Green I(终浓度为1μmol/L,1.1x)分别对前染法(图10)与后染法(图11)效果进行对比, 使用的DNA样品为25-500bp DNA分子量标准Marker(货号:B600303,BBI,美国)按 照使用说明书控制上样量依次为:10ng、5ng、2ng、1ng及0.5ng。According to reports, gel staining is currently mainly achieved in two ways: (1) before separation, the dye is pre-added to the DNA mixture for electrophoretic separation and then fluorescent imaging is performed, that is, the pre-staining method; (2) the DNA mixture is electrophoresed After separation, the gel is immersed in an aqueous solution or buffer of fluorescent dye for a period of time for fluorescence imaging, that is, post-staining; both methods are very common. Compound c (final concentration: 1 μmol/L) and SYBR Green I (final concentration: 1 μmol/L, 1.1×) prepared in Example 1 were used to effect the pre-staining method (Fig. 10) and the post-staining method (Fig. 11) respectively. In contrast, the DNA sample used was a 25-500bp DNA molecular weight standard Marker (Cat. No. B600303, BBI, USA), and the loading amount was controlled to be 10ng, 5ng, 2ng, 1ng and 0.5ng according to the instruction manual.
从前染法(图10)和后染法的效果对比(图11)可以看出,本发明实施例1制备的化合物c两种染色方式都与SYBR Green I效果相当,并且在前染法中,与SYBR Green I对 比,化合物c的染色结果没有出现明显的拖尾现象,条带更干净。另外,也对比了化合物 c(终浓度为1μmol/L)与SYBR Green I(终浓度为1μmol/L,1.1x)对实施例6中产生的 扩增子样品进行前染法与后染法的效果对比(图12),结果表明在实际应用的过程中,化 合物c与SYBRGreen I相比无论前染还是后染,效果相当。并且更明显地,相比于SYBR Green I化合物c前染实际样品没有的拖尾现象,条带更干净清晰,对不同DNA片段的区 分更加明显。It can be seen from the comparison of the effects of the pre-dyeing method (Fig. 10) and the post-dyeing method (Fig. 11) that the two dyeing methods of compound c prepared in Example 1 of the present invention are comparable to SYBR Green I, and in the pre-dyeing method, Compared with SYBR Green I, there is no obvious tailing phenomenon in the staining result of compound c, and the band is cleaner. In addition, compound c (final concentration of 1 μmol/L) and SYBR Green I (final concentration of 1 μmol/L, 1.1×) were also compared for pre-staining and post-staining of the amplicon samples generated in Example 6. Comparing the effects (Fig. 12), the results show that in the process of practical application, compound c has the same effect as SYBRGreen I regardless of pre-dyeing or post-dyeing. And more obviously, compared with SYBR Green I compound c pre-stained with no tailing phenomenon in the actual sample, the band is cleaner and clearer, and the distinction between different DNA fragments is more obvious.
实施例8:化合物c的本征稳定性Example 8: Intrinsic stability of compound c
一般情况qPCR的温度范围为55℃-105℃,qPCR试剂储存要在-20℃-4℃。另外,核酸分子需要在在纯水溶液或者缓冲溶液的环境。这就要求染料分子在水中或缓冲液中稳定,并且同样在温度范围为55℃-105℃及-20℃-4℃是稳定的。Generally, the temperature range of qPCR is 55℃-105℃, and the qPCR reagents should be stored at -20℃-4℃. In addition, nucleic acid molecules need to be in a pure aqueous or buffered solution environment. This requires the dye molecule to be stable in water or buffer, and also stable in the temperature range of 55°C-105°C and -20°C-4°C.
1)热稳定性1) Thermal stability
在体积为20μL,含有终浓度为10μmol/L的化合物c,pH=8.3的Tris-HCl缓冲液(含Mg2+)的体系中,化合物c在经历95℃保持20s、55℃保温30s、72℃保温30s的40次 热循环后,如图13所示,依旧保持稳定(55℃时设定测试荧光强度),信号无明显降低, 证实化合物c在qPCR中的耐用性。In a system with a volume of 20 μL, containing compound c with a final concentration of 10 μmol/L, and Tris-HCl buffer (containing Mg 2+ ) at pH=8.3, compound c was kept at 95°C for 20s, 55°C for 30s, 72°C After 40 thermal cycles of 30 s incubation at °C, as shown in Figure 13, it remained stable (the test fluorescence intensity was set at 55 °C), and the signal did not decrease significantly, confirming the durability of compound c in qPCR.
2)溶液中的稳定性2) Stability in solution
在体积为3mL,含有终浓度为1μmol/L的化合物c或SYBR Green I(1.1x)的pH=8.3的Tris-HCl缓冲液(含Mg2+)或纯水的体系中,每隔12-24h监测化合物c及SYBR Green I在各自最大吸收波长处的信号变化,纯水中为图14A,缓冲液中为图14B。结果表明:纯 水中SYBRGreen I相比于缓冲中极不稳定,这与已报道的现象是一致的,在经历72h已 有近一半的分子分解;而经历相同时间化合物c无论在纯水中还是缓冲液中都要较SYBR Green I稳定。更为重要的是,化合物c在纯水中非常稳定,这表明化合物c的存储环境不 如SYBR Green I苛刻,在凝胶染色采用后染法时,可以直接使用纯水配置无需额外再配置 缓冲液,使用更为方便。In a system with a volume of 3 mL, a pH=8.3 Tris-HCl buffer (containing Mg 2+ ) or pure water containing compound c or SYBR Green I (1.1×) at a final concentration of 1 μmol/L, every 12- The signal changes of compound c and SYBR Green I at their respective maximum absorption wavelengths were monitored for 24 hours, as shown in Figure 14A in pure water and Figure 14B in buffer solution. The results show that: SYBRGreen I in pure water is extremely unstable compared with buffer, which is consistent with the reported phenomenon, nearly half of the molecules are decomposed after 72 hours; All buffers are more stable than SYBR Green I. More importantly, compound c is very stable in pure water, which indicates that the storage environment of compound c is not as harsh as that of SYBR Green I. When the post-staining method is used for gel staining, it can be directly prepared with pure water without additional buffer configuration. , it is more convenient to use.
实施例9:比较化合物c与SYBR Green I合成成本Example 9: Comparison of synthetic cost of compound c and SYBR Green I
目前,SYBR Green I是目前市场上应用最为广泛、且相对性价比较高的染料。然而根据对SYBR Green I合成路径分析(图15),化合物c反应条件要求低,原料购买渠道众 多,无需自行合成,且价格便宜(试剂价格查询网站:https://www.bidepharm.com; https://www.chemicalbook.com/ProductIndex.aspx);仅需三步反应,无需中间产物纯化,关 键步骤反应无需加热,且产率更高,并且上述实验已表明化合物c的性能要优于SYBR Green I。At present, SYBR Green I is the most widely used and relatively cost-effective dye on the market. However, according to the analysis of the synthetic route of SYBR Green I (Fig. 15), the reaction conditions of compound c are low, there are many purchasing channels for raw materials, there is no need for self-synthesis, and the price is cheap (reagent price query website: https://www.bidepharm.com; https://www.bidepharm.com; https://www.bidepharm.com; https://www.bidepharm.com ://www.chemicalbook.com/ProductIndex.aspx); only three steps are required, no intermediate product purification is required, no heating is required for the key step reaction, and the yield is higher, and the above experiments have shown that the performance of compound c is better than SYBR Green I.
更为重要的是,在实际的核酸分子检测中,染料成本是影响实验总成本的最重要一环。 虽然SYBR Green I是高灵敏的核酸染料,但是其价格昂贵,官方售价约1万元1mL(10000x),其试剂本身成本过高,特别是投放于大规模核酸检测和凝胶电泳成像中。如 实施例7的凝胶染色实验中已证实了化合物c不仅染色效果与SYBR Green I相当,甚至效 果更优,但化合物c合成成本远远低于SYBR Green I。这表明了化合物c能够在保持高灵 敏的条件下,能够极大地降低与之匹配的生产成本。More importantly, in the actual nucleic acid molecule detection, the cost of dye is the most important part that affects the total cost of the experiment. Although SYBR Green I is a highly sensitive nucleic acid dye, it is expensive. The official price is about 10,000 yuan per 1mL (10,000x). The cost of the reagent itself is too high, especially for large-scale nucleic acid detection and gel electrophoresis imaging. As in the gel staining experiment of Example 7, it has been confirmed that compound c not only has the same staining effect as SYBR Green I, but even better effect, but the synthesis cost of compound c is far lower than that of SYBR Green I. This shows that compound c can greatly reduce the matching production cost under the condition of maintaining high sensitivity.
SEQUENCE LISTINGSEQUENCE LISTING
<110> 四川大学<110> Sichuan University
<120> 一种核酸荧光染料及其制备和应用<120> A nucleic acid fluorescent dye and its preparation and application
<130> 2021.8.2<130> 2021.8.2
<160> 13<160> 13
<170> PatentIn version 3.5<170> PatentIn version 3.5
<210> 1<210> 1
<211> 20<211> 20
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence
<400> 1<400> 1
acagacacct acagacacct 20
<210> 2<210> 2
<211> 20<211> 20
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence
<400> 2<400> 2
aggtgtctgt aggtgtctgt 20
<210> 3<210> 3
<211> 20<211> 20
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence
<400> 3<400> 3
ctctcagtct gcatctcact 20
<210> 4<210> 4
<211> 20<211> 20
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence
<400> 4<400> 4
agtgagatgc agactgagag 20
<210> 5<210> 5
<211> 60<211> 60
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence
<400> 5<400> 5
acagacacct acagacacct acagacacct acagacacct acagacacct acagacacct 60acagacacct acagacacct acagacacct acagacacct acagacacct acagacacct 60
<210> 6<210> 6
<211> 60<211> 60
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence
<400> 6<400> 6
aggtgtctgt aggtgtctgt aggtgtctgt aggtgtctgt aggtgtctgt aggtgtctgt 60aggtgtctgt aggtgtctgt aggtgtctgt aggtgtctgt aggtgtctgt aggtgtctgt 60
<210> 7<210> 7
<211> 48<211> 48
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence
<400> 7<400> 7
gcagacacct acagacacct aaaaaaatag gtgtctgtag gtgtctgc 48gcagacacct acagacacct aaaaaaatag gtgtctgtag gtgtctgc 48
<210> 8<210> 8
<211> 20<211> 20
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence
<400> 8<400> 8
tccgcgcgcc cgggccgcca 20
<210> 9<210> 9
<211> 20<211> 20
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence
<400> 9<400> 9
tggcggcccg ggcgcgcgga 20
<210> 10<210> 10
<211> 20<211> 20
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence
<400> 10<400> 10
aaaaaaaaaa aaaaaaaaaa 20
<210> 11<210> 11
<211> 20<211> 20
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence
<400> 11<400> 11
tttttttttt tttttttttt 20
<210> 12<210> 12
<211> 20<211> 20
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence
<400> 12<400> 12
tccctttcca gtacgcttcg 20
<210> 13<210> 13
<211> 20<211> 20
<212> DNA<212> DNA
<213> 人工序列(Artificial Sequence)<213> Artificial Sequence
<400> 13<400> 13
tctggatggt atgcccggta 20
Claims (10)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110958363.6A CN113583467B (en) | 2021-08-20 | 2021-08-20 | A nucleic acid fluorescent dye and its preparation and application |
| PCT/CN2022/086524 WO2023019983A1 (en) | 2021-08-20 | 2022-04-13 | Nucleic acid fluorescent dye and preparation and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110958363.6A CN113583467B (en) | 2021-08-20 | 2021-08-20 | A nucleic acid fluorescent dye and its preparation and application |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN113583467A CN113583467A (en) | 2021-11-02 |
| CN113583467B true CN113583467B (en) | 2022-04-26 |
Family
ID=78238630
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110958363.6A Active CN113583467B (en) | 2021-08-20 | 2021-08-20 | A nucleic acid fluorescent dye and its preparation and application |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN113583467B (en) |
| WO (1) | WO2023019983A1 (en) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN113583467B (en) * | 2021-08-20 | 2022-04-26 | 四川大学 | A nucleic acid fluorescent dye and its preparation and application |
| CN115724835B (en) * | 2022-09-21 | 2025-03-28 | 成都阅微澜生物科技有限公司 | Nucleic acid dye with low concentration effect and high fluorescence efficiency and its preparation and application |
| CN117164577B (en) * | 2023-08-10 | 2024-11-15 | 郑州玛特瑞斯生物科技有限公司 | A fluorescent compound, kit and preparation method for dsDNA quantification |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9133343B2 (en) * | 2009-11-30 | 2015-09-15 | Enzo Biochem, Inc. | Dyes and compositions, and processes for using same in analysis of protein aggregation and other applications |
| CN102115610B (en) * | 2010-01-05 | 2014-07-02 | 大连理工大学 | Fluorescent dye, preparation method and application thereof |
| US9682970B2 (en) * | 2012-06-29 | 2017-06-20 | Biotium, Inc. | Fluorescent compounds and uses thereof |
| CN104650609B (en) * | 2013-11-22 | 2017-04-26 | 沈阳药科大学 | Thiazol orange derivative, manufacturing method and application of thiazol orange derivative used as double helix nucleic acid fluorescence molecular probe |
| CN106147753B (en) * | 2015-04-24 | 2020-03-10 | 广东工业大学 | Thiazole orange styrene compound as G-quadruplex nucleic acid fluorescent probe |
| CN114341627A (en) * | 2019-11-13 | 2022-04-12 | 李峰 | Nucleic acid detection method and device |
| CN113201583B (en) * | 2021-04-29 | 2022-02-08 | 国科宁波生命与健康产业研究院 | Method for synthesizing nucleic acid under constant temperature condition, kit and application |
| CN113583467B (en) * | 2021-08-20 | 2022-04-26 | 四川大学 | A nucleic acid fluorescent dye and its preparation and application |
-
2021
- 2021-08-20 CN CN202110958363.6A patent/CN113583467B/en active Active
-
2022
- 2022-04-13 WO PCT/CN2022/086524 patent/WO2023019983A1/en not_active Ceased
Also Published As
| Publication number | Publication date |
|---|---|
| CN113583467A (en) | 2021-11-02 |
| WO2023019983A1 (en) | 2023-02-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN113583467B (en) | A nucleic acid fluorescent dye and its preparation and application | |
| JP4602956B2 (en) | Probes labeled with energy transfer linked dyes | |
| US8071388B2 (en) | Compositions and methods for biodetection by nucleic acid-templated chemistry | |
| CN101076537B (en) | Oligonucleotides labeled with a plurality of fluorophores | |
| CN106147753B (en) | Thiazole orange styrene compound as G-quadruplex nucleic acid fluorescent probe | |
| Zhang et al. | Hybridization induced fluorescence turn-on of AIEgen–oligonucleotide conjugates for specific DNA detection | |
| EP1110088B1 (en) | Energy transfer dyes | |
| JP7046082B2 (en) | Its use for detection of new compounds and target molecules in samples | |
| Yotapan et al. | Pyrrolidinyl peptide nucleic acid terminally labeled with fluorophore and end-stacking quencher as a probe for highly specific DNA sequence discrimination | |
| CN106471131B (en) | Fluorescently labeled single-stranded nucleic acid and its use | |
| JP4265134B2 (en) | Novel fluorescent dye and method for measuring nucleic acid | |
| Holzhauser et al. | Synthesis and optical properties of cyanine dyes as fluorescent DNA base substitutions for live cell imaging | |
| WO2013095137A2 (en) | Methods, means and kits for detecting scarce analytes | |
| US9353405B2 (en) | Optimized real time nucleic acid detection processes | |
| EP1223226B1 (en) | Novel fluorescent dye and method of measuring nucleic acid | |
| Bai et al. | A new phosphoramidite enables orthogonal double labelling to form combination oligonucleotide probes | |
| CN115724835B (en) | Nucleic acid dye with low concentration effect and high fluorescence efficiency and its preparation and application | |
| Watanabe et al. | Studies on Luciferase from Photobacterium phosphoreum XI. Interaction of 8-Substituted FMNH 2 with Luciferase | |
| US6967250B1 (en) | Energy transfer dyes | |
| JP4454218B2 (en) | Polynucleotide derivatives and uses thereof | |
| CN110295217B (en) | Method for applying quantum dot doped ratiometric fluorescent probe to microRNA in-vitro detection | |
| Eustáquio et al. | Affordable small molecules as promising fluorescent labels for biomolecules: application in RNA-FISH probes | |
| CN117164577A (en) | Fluorescent compound for dsDNA quantification, kit and preparation method | |
| CN114656394A (en) | Fluorescent compound, preparation method thereof, fluorescence modified nucleotide and kit | |
| CN110642806A (en) | Double-stranded DNA (deoxyribonucleic acid) combined fluorescent dye as well as preparation and application thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| TR01 | Transfer of patent right | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20230818 Address after: No. 577, 5th Floor, Building 1, No. 1388, Middle Section of Tianfu Avenue, Chengdu High tech Zone, China (Sichuan) Pilot Free Trade Zone, Chengdu City, Sichuan Province, 610000 Patentee after: Chengdu Yueweilan Biotechnology Co.,Ltd. Address before: 610065, No. 24, south section of Ring Road, Sichuan, Chengdu Patentee before: SICHUAN University |