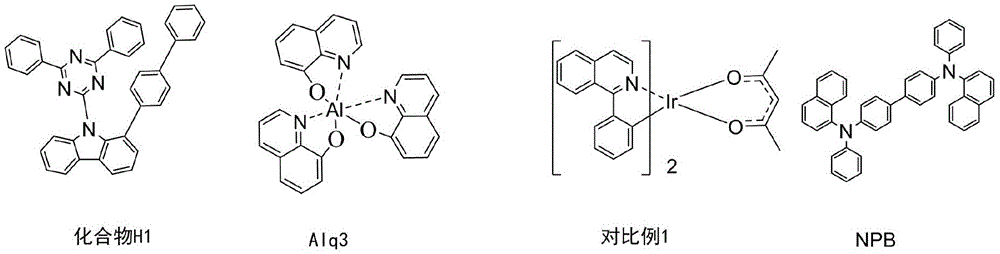
CN113563386A - A kind of iridium metal complex and its application - Google Patents
A kind of iridium metal complex and its application Download PDFInfo
- Publication number
- CN113563386A CN113563386A CN202110857295.4A CN202110857295A CN113563386A CN 113563386 A CN113563386 A CN 113563386A CN 202110857295 A CN202110857295 A CN 202110857295A CN 113563386 A CN113563386 A CN 113563386A
- Authority
- CN
- China
- Prior art keywords
- sub
- metal complex
- substituted
- iridium metal
- alkyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F15/00—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table
- C07F15/0006—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table compounds of the platinum group
- C07F15/0033—Iridium compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/341—Transition metal complexes, e.g. Ru(II)polypyridine complexes
- H10K85/342—Transition metal complexes, e.g. Ru(II)polypyridine complexes comprising iridium
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/18—Metal complexes
- C09K2211/185—Metal complexes of the platinum group, i.e. Os, Ir, Pt, Ru, Rh or Pd
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/549—Organic PV cells
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Crystallography & Structural Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
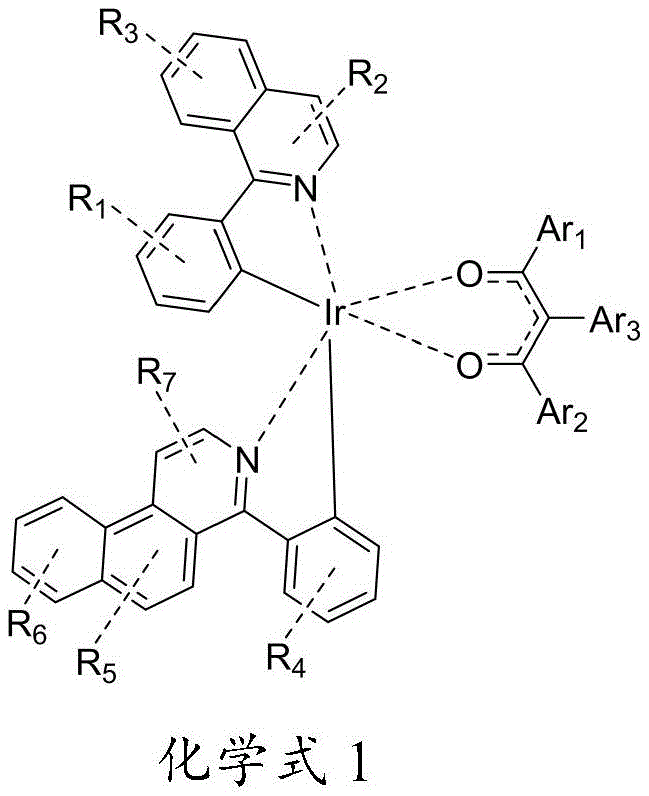
本发明公开了一种铱金属配合物及其应用,属于有机发光材料技术领域,其结构式如化学式1所示,其中所述R1‑R7和Ar1‑Ar3各自独立地选自氢、氘、卤素、氰基、取代或非取代的C1‑C8烷基、取代或非取代的C6‑C10芳基、取代或非取代的4‑8元芳族杂环基。本发明提供的新型结构的铱金属配合物,应用于有机电致发光器件后,能够使得器件的发光效率以及寿命提高。此外,本发明提供的铱金属配合物的制备方法,制备步骤简单,产物纯度高。 The invention discloses an iridium metal complex and an application thereof, belonging to the technical field of organic light - emitting materials. Deuterium, halogen, cyano, substituted or unsubstituted C 1 -C 8 alkyl, substituted or unsubstituted C 6 -C 10 aryl, substituted or unsubstituted 4-8 membered aromatic heterocyclic group. After the iridium metal complex with the novel structure provided by the invention is applied to an organic electroluminescence device, the luminous efficiency and the service life of the device can be improved. In addition, the preparation method of the iridium metal complex provided by the present invention has simple preparation steps and high product purity.
Description
技术领域technical field
本发明涉及有机光电材料技术领域,更具体的说是涉及一种铱金属配合物及其应用。The invention relates to the technical field of organic optoelectronic materials, and more particularly to an iridium metal complex and its application.
背景技术Background technique
有机电致发光技术是最新一代平板显示技术,可用于平板显示器和照明光源,目前商品化的平板显示器已投入市场。电致发光器件具有全固态结构,有机电致发光材料是构成该器件的核心和基础,新材料的开发是推动电致发光技术不断进步的源动力。在有机发光中,磷光发光现象自从发现以来,一直受到大家的追崇,因为磷光材料的发光效率明显高于荧光发光效率,从理论上能达到100%的发光效率,所以很多科研机构都在加大磷光材料的研发力度,试图通过磷光材料来加快产业化发展。Organic electroluminescence technology is the latest generation of flat-panel display technology, which can be used for flat-panel displays and lighting sources, and commercialized flat-panel displays have been put into the market. Electroluminescent devices have an all-solid-state structure, and organic electroluminescent materials are the core and foundation of the device. The development of new materials is the driving force behind the continuous progress of electroluminescent technology. In organic light-emitting, the phenomenon of phosphorescent luminescence has been admired by everyone since its discovery, because the luminous efficiency of phosphorescent materials is significantly higher than that of fluorescent materials, and theoretically it can reach 100% luminous efficiency, so many scientific research institutions are adding The research and development of large phosphorescent materials is trying to speed up the industrialization development through phosphorescent materials.
但是,现有技术中,部分磷光材料应用于有机发光器件中有效率低寿命短的现象。因此,如何提供一种寿命长效率高的有机电致发光材料是本领域技术人员亟需解决的问题。However, in the prior art, some phosphorescent materials are used in organic light-emitting devices with low efficiency and short lifetime. Therefore, how to provide an organic electroluminescent material with long life and high efficiency is an urgent problem for those skilled in the art.
发明内容SUMMARY OF THE INVENTION
有鉴于此,本发明提供了一种铱金属配合物及其应用,本发明提供的铱金属配合物,通过选择特定的杂环的配体结合,调节化合物的波长,得到的有机金属化合物在用于有机电致发光器件后,使得器件的发光效率提高、寿命增加及起动电压降低。In view of this, the present invention provides an iridium metal complex and its application. The iridium metal complex provided by the present invention can adjust the wavelength of the compound by selecting the ligand binding of a specific heterocyclic ring, and the obtained organometallic compound is used in After the organic electroluminescence device is used, the luminous efficiency of the device is increased, the lifespan is increased, and the starting voltage is decreased.
为了实现上述目的,本发明采用如下技术方案:In order to achieve the above object, the present invention adopts the following technical solutions:
一种铱金属配合物,其结构式如化学式1所示:A kind of iridium metal complex, its structural formula is shown in chemical formula 1:
其中,in,
所述R1-R7和Ar1-Ar3各自独立地选自氢、氘、卤素、氰基、取代或非取代的C1-C8烷基、取代或非取代的C6-C10芳基、取代或非取代的4-8元芳族杂环基。Said R 1 -R 7 and Ar 1 -Ar 3 are each independently selected from hydrogen, deuterium, halogen, cyano, substituted or unsubstituted C 1 -C 8 alkyl, substituted or unsubstituted C 6 -C 10 Aryl, substituted or unsubstituted 4-8 membered aromatic heterocyclic group.
优选的,所述R1-R7至少包括一个氟取代基。Preferably, the R 1 -R 7 include at least one fluorine substituent.
优选的,所述R1-R7取代位置为所在环的任意位置;Preferably, the substitution position of R 1 -R 7 is any position in the ring;
所述R2、R5和R7的取代基的数量为0-2;The number of substituents of the R 2 , R 5 and R 7 is 0-2;
所述R1、R3、R4、R6取代基的数量为0-4。The number of the R 1 , R 3 , R 4 , and R 6 substituents is 0-4.
优选的,所述R1数量为2,且为未取代或1个取代基取代的C1烷基或C3烷基;Preferably, the number of R 1 is 2, and it is an unsubstituted or 1-substituted C 1 alkyl group or a C 3 alkyl group;
所述R2为H;The R 2 is H;
所述R3为未取代或1个取代基取代的C3烷基;The R 3 is unsubstituted or 1 substituent substituted C 3 alkyl;
所述R4数量为2-3,且为氟、未取代或1个取代基取代的C1烷基和/或C3烷基;The number of said R 4 is 2-3, and it is fluorine, unsubstituted or 1 substituent substituted C 1 alkyl and/or C 3 alkyl;
所述R5为H或C3烷基;The R 5 is H or C 3 alkyl;
所述R6数量为1-2,且为氟和/或C3烷基;The R 6 number is 1-2, and is fluorine and/or C 3 alkyl;
所述R7为H。The R7 is H.
优选的,所述烷基包括直链烷基、支链烷基、环烷基、至少1个取代基取代的直链烷基、至少1个取代基取代的支链烷基或至少1个取代基取代的环烷基中的一种。Preferably, the alkyl group includes straight-chain alkyl, branched-chain alkyl, cycloalkyl, straight-chain alkyl substituted with at least one substituent, branched alkyl substituted with at least one substituent or at least one substituted One of the cycloalkyl substituted groups.
优选的,所述芳族杂环基优选为未取代的杂芳基或至少1个取代基取代的芳族杂环基。Preferably, the aromatic heterocyclic group is preferably an unsubstituted heteroaryl group or an aromatic heterocyclic group substituted with at least one substituent.
优选的,所述杂芳基中的杂原子为氮、硫或氧。Preferably, the heteroatom in the heteroaryl group is nitrogen, sulfur or oxygen.
优选的,所述取代基独立的选自卤素、氘或氰基中的一种或几种。Preferably, the substituents are independently selected from one or more of halogen, deuterium or cyano.
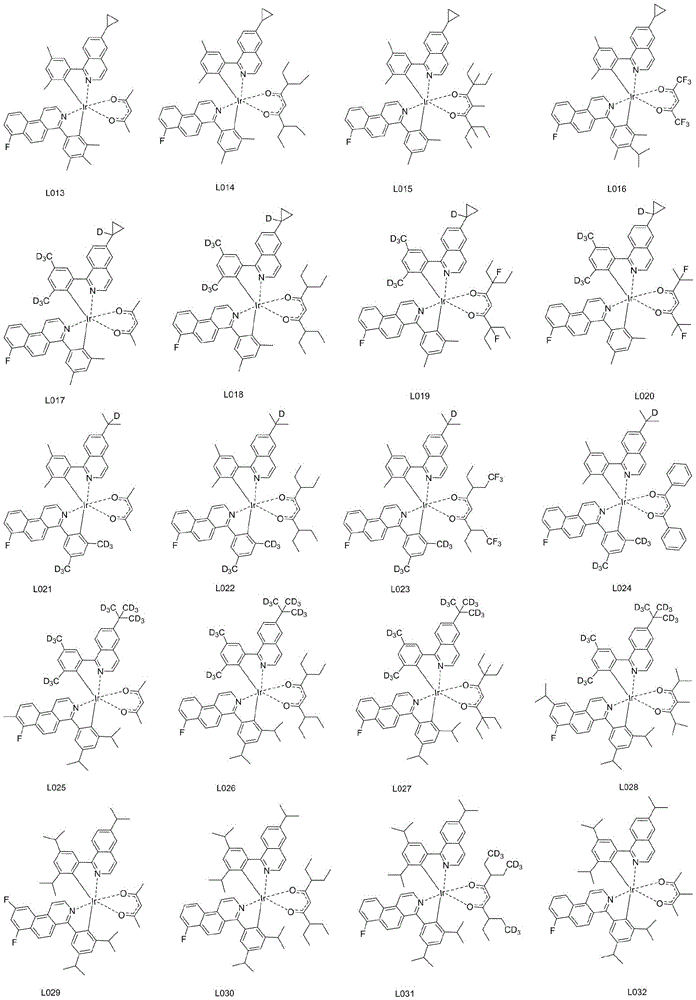
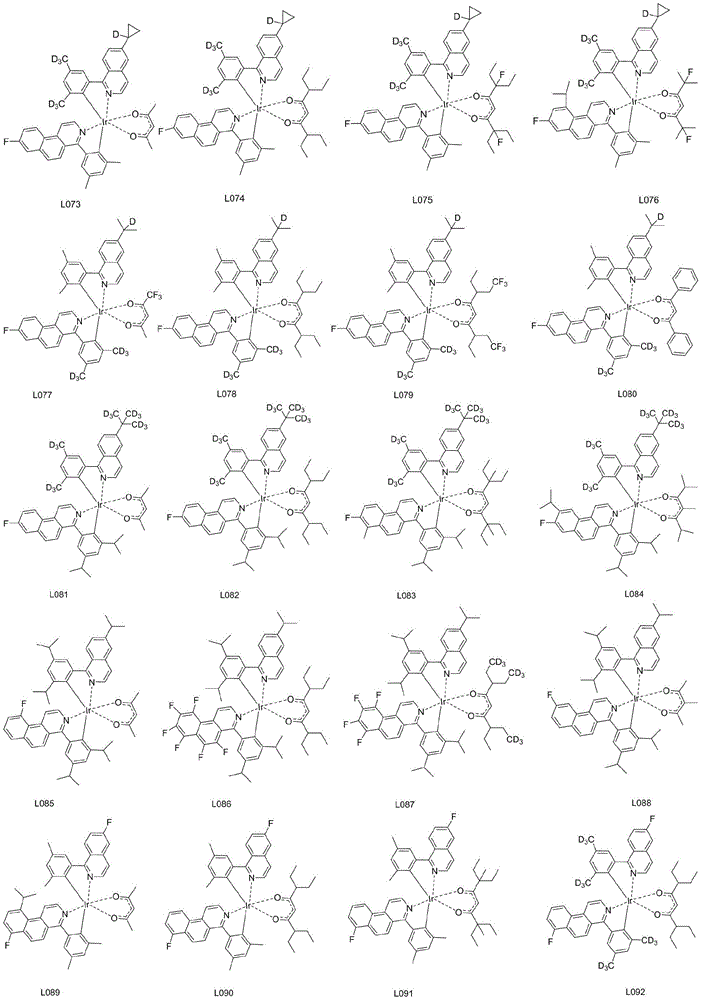
优选的,所述铱金属配合物选自以下结构中的任意一种:Preferably, the iridium metal complex is selected from any one of the following structures:
所述铱金属配合物的制备方法,步骤如下:The preparation method of described iridium metal complex, the steps are as follows:
1)分别提供如下结构式表示的化合物A、化合物B和化合物C: 1) Provide compound A, compound B and compound C represented by the following structural formulas:
2)将原料A和原料B与三氯化铱充分反应,制得桥联配体中间体C;2) fully react raw material A and raw material B with iridium trichloride to obtain bridging ligand intermediate C;
3)将中间体C与中间体D充分反应,制得化学式1所述的铱金属配合物。作为本发明再进一步的方案:桥联配体中间体C的结构为:3) fully react the intermediate C and the intermediate D to obtain the iridium metal complex described in the chemical formula 1. As a further scheme of the present invention: the structure of the bridging ligand intermediate C is:
其合成路线如下:Its synthetic route is as follows:
其中:R1、R2、R3、R4、R5、R6、R7、Ar1、Ar2、Ar3及其各自的取代基数量与化学式1中限定的范围一致。Wherein: R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , Ar 1 , Ar 2 , Ar 3 and the number of their respective substituents are consistent with the range defined in Chemical Formula 1.
一种铱金属配合物在有机电致发光器件中的应用,所述有机电致发光器件中包括所述的铱金属配合物,还包括第一电极、第二电极和置于所述两电极之间的有机物层,其中,所述有机物层中包含有本发明化学式1所示的铱金属配合物;本发明化学式1所示的铱金属配合物可以是单一形态或与其它物质混合存在于有机物层中。An application of an iridium metal complex in an organic electroluminescence device, the organic electroluminescence device comprises the iridium metal complex, and further comprises a first electrode, a second electrode and a device disposed between the two electrodes. The organic layer between the two, wherein the organic layer contains the iridium metal complex shown in the chemical formula 1 of the present invention; the iridium metal complex shown in the chemical formula 1 of the present invention can exist in a single form or mixed with other substances in the organic layer middle.
优选的,所述有机物层包括发光层,所述发光层含有本发明化学式1所示的铱金属配合物。优选所述有机电致发光器件的发光层包括主体材料和掺杂材料,所述掺杂材料为本发明化学式1所示的铱金属配合物。Preferably, the organic layer includes a light-emitting layer, and the light-emitting layer contains the iridium metal complex represented by Chemical Formula 1 of the present invention. Preferably, the light-emitting layer of the organic electroluminescent device includes a host material and a dopant material, and the dopant material is an iridium metal complex represented by Chemical Formula 1 of the present invention.
优选的,所述发光层主体材料与掺杂材料的混合比例为90:(10-99.5):0.5。Preferably, the mixing ratio of the host material of the light-emitting layer and the dopant material is 90:(10-99.5):0.5.
优选的,所述有机物层还包括空穴注入层、空穴传输层、既具备空穴注入又具备空穴传输层、电子阻挡层、空穴阻挡层、电子传输层、电子注入层和既具备电子传输又具备电子注入层中的一种或几种。Preferably, the organic layer further includes a hole injection layer, a hole transport layer, a hole injection layer and a hole transport layer, an electron blocking layer, a hole blocking layer, an electron transport layer, an electron injection layer, and both a hole injection layer and a hole transport layer. The electron transport also has one or more of the electron injection layers.
其中至少一层功能层含有本发明化学式1所示的铱金属配合物。At least one functional layer contains the iridium metal complex represented by Chemical Formula 1 of the present invention.
一种有机电致发光器件在有机发光器件、有机太阳电池、电子纸、有机感光体或有机薄膜晶体管中的应用。Application of an organic electroluminescent device in organic light-emitting device, organic solar cell, electronic paper, organic photoreceptor or organic thin film transistor.
经由上述的技术方案可知,与现有技术相比,本发明公开提供了一种铱金属配合物,本发明提供的新型结构的铱金属配合物,通过选择特定的杂环的配体结合,调节化合物的波长,得到的有机金属化合物在用于有机电致发光器件后,使得器件的发光效率以及寿命提高。此外,本发明提供的铱金属配合物的制备方法,制备步骤简单,产物纯度高。It can be seen from the above technical solutions that, compared with the prior art, the present disclosure provides an iridium metal complex, and the iridium metal complex of the novel structure provided by the present invention can be adjusted by selecting a specific heterocyclic ligand for binding. The wavelength of the compound, after the obtained organometallic compound is used in an organic electroluminescent device, improves the luminous efficiency and life of the device. In addition, the preparation method of the iridium metal complex provided by the present invention has simple preparation steps and high product purity.
具体实施方式Detailed ways
下面将结合本发明实施例中的技术方案进行清楚、完整地描述,显然,所描述的实施例仅仅是本发明一部分实施例,而不是全部的实施例。基于本发明中的实施例,本领域普通技术人员在没有做出创造性劳动前提下所获得的所有其他实施例,都属于本发明保护的范围。The following will clearly and completely describe the technical solutions in the embodiments of the present invention. Obviously, the described embodiments are only a part of the embodiments of the present invention, rather than all the embodiments. Based on the embodiments of the present invention, all other embodiments obtained by those of ordinary skill in the art without creative efforts shall fall within the protection scope of the present invention.
实施例1Example 1
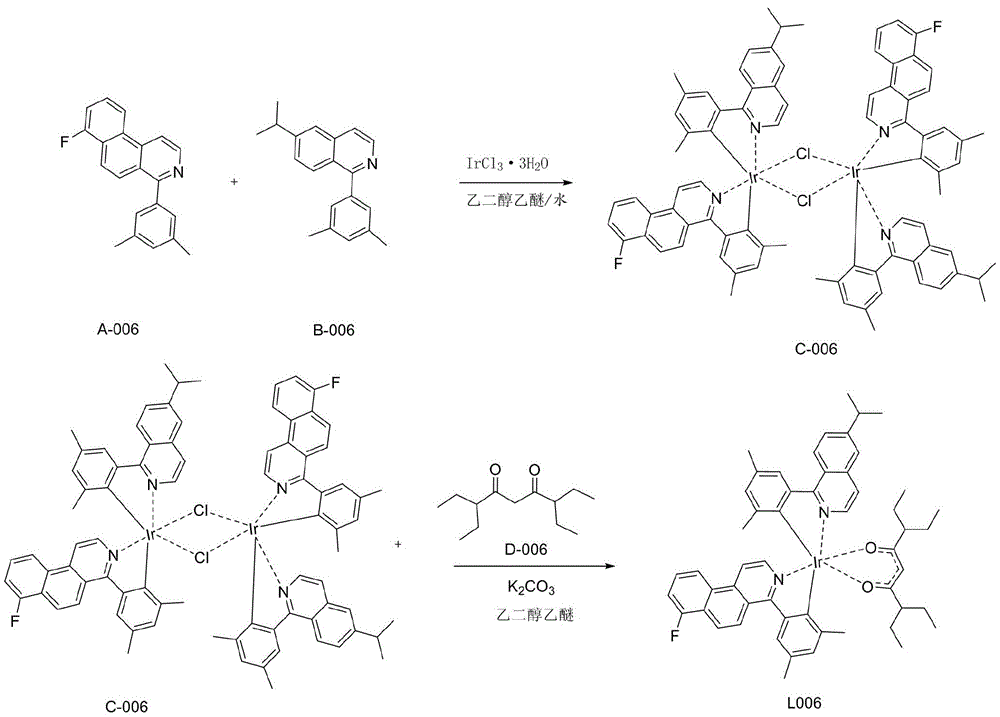
一种铱金属配合物L006,即编号为L006的化合物,具体合成步骤如下:A kind of iridium metal complex L006, namely the compound of numbering L006, concrete synthesis steps are as follows:
1)在氮气保护体系下,称取A-006(28.36mmol,8.55g)和B-006(28.36mmol,7.81g)IrCl3·3H2O(28.36mmol,10g)加入反应体系中,加入500mL乙二醇乙醚和167mL纯净水的混合溶液,氮气保护下120℃回流24小时,然后冷却到室温,有沉淀析出,将沉淀减压过滤,用水、无水乙醇、石油醚依次冲洗烘干,得到红色粉末的桥联配体C-006(11.38g,产率为50%);1) Under nitrogen protection system, weigh A-006 (28.36mmol, 8.55g) and B-006 (28.36mmol, 7.81g) IrCl 3 ·3H 2 O (28.36mmol, 10g) into the reaction system, add 500mL The mixed solution of ethylene glycol ether and 167 mL of pure water was refluxed at 120°C for 24 hours under nitrogen protection, and then cooled to room temperature, and a precipitate was precipitated. The precipitate was filtered under reduced pressure, washed with water, absolute ethanol, and petroleum ether in turn to obtain Bridging ligand C-006 as red powder (11.38 g, 50% yield);
2)称取中间体C-006(7.09mmol,11.38g),加入配体D-006(21.27mmol,4.5g),再向体系中加乙二醇乙醚300mL和碳酸钾(7.5g),氮气保护下,120℃搅拌24小时,减压过滤,醇洗,烘干,用二氯甲烷做溶剂,用硅胶柱层析,滤液浓缩固体析出,得到最终红色的化合物L006(4.16g,产率30%);2) Weigh intermediate C-006 (7.09mmol, 11.38g), add ligand D-006 (21.27mmol, 4.5g), then add ethylene glycol ether 300mL and potassium carbonate (7.5g) to the system, nitrogen Under protection, stirred at 120 ° C for 24 hours, filtered under reduced pressure, washed with alcohol, dried, used dichloromethane as a solvent, used silica gel column chromatography, the filtrate was concentrated and the solid was precipitated to obtain the final red compound L006 (4.16g, yield 30 %);
HPLC纯度:大于99.5%;HPLC purity: greater than 99.5%;
质谱:计算值为978.29;测试值为978.30。Mass Spec: Calculated 978.29; Tested 978.30.
元素分析:计算值C:66.30%;H:5.98%;F:1.94%;Ir:19.65%;N:2.86;O:3.27%;测试值为:C:66.30%;H:6.00%;F:1.95%;Ir:19.66%;N:2.86;O:3.28%;Elemental analysis: Calculated value C: 66.30%; H: 5.98%; F: 1.94%; Ir: 19.65%; N: 2.86; O: 3.27%; Test value: C: 66.30%; H: 6.00%; F: 1.95%; Ir: 19.66%; N: 2.86; O: 3.28%;
具体的,步骤1)至2)的反应式如下:Specifically, the reaction formula of steps 1) to 2) is as follows:
本实施例中,所述铱金属配合物在制备有机电致发光器件产品中的应用。In this embodiment, the iridium metal complex is used in the preparation of organic electroluminescent device products.
实施例2Example 2
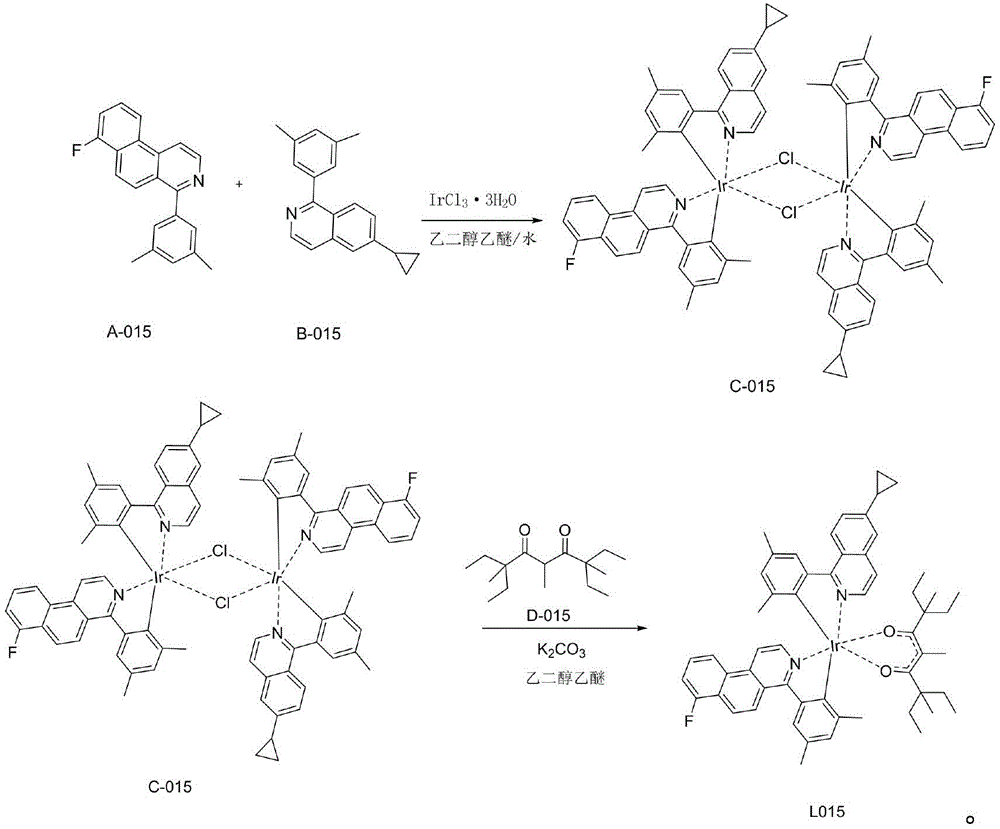
一种铱金属配合物L015,即编号为L015的化合物,具体合成步骤如下:1)在氮气保护体系下,称取A-015(28.36mmol,8.55g)和B-015(28.36mmol,7.75g)IrCl3·3H2O(28.36mmol,10g)加入反应体系中,加入500mL乙二醇乙醚和167mL纯净水的混合溶液,氮气保护下120℃回流24小时,然后冷却到室温,有沉淀析出,将沉淀减压过滤,用水、无水乙醇、石油醚依次冲洗烘干,得到红色粉末的桥联配体C-015(11.84g,产率为52%);A kind of iridium metal complex L015, namely the compound numbered L015, the specific synthesis steps are as follows: 1) under nitrogen protection system, weigh A-015 (28.36mmol, 8.55g) and B-015 (28.36mmol, 7.75g) ) IrCl 3 ·3H 2 O (28.36 mmol, 10 g) was added to the reaction system, a mixed solution of 500 mL of ethylene glycol ether and 167 mL of purified water was added, refluxed at 120° C. for 24 hours under nitrogen protection, and then cooled to room temperature, there was precipitation, The precipitate was filtered under reduced pressure, washed with water, anhydrous ethanol, and petroleum ether in turn and dried to obtain a red powder bridging ligand C-015 (11.84 g, yield 52%);
2)称取中间体C-015(7.4mmol,11.84g),加入配体D-015(22.2mmol,5.65g),再向体系中加乙二醇乙醚300mL和碳酸钾(7.8g),氮气保护下,120℃搅拌24小时,减压过滤,醇洗,烘干,用二氯甲烷做溶剂,用硅胶柱层析,滤液浓缩固体析出,得到最终红色的化合物L015(4.22g,产率28%);2) Weigh intermediate C-015 (7.4mmol, 11.84g), add ligand D-015 (22.2mmol, 5.65g), then add ethylene glycol ether 300mL and potassium carbonate (7.8g) to the system, nitrogen Under the protection, stirring at 120 ° C for 24 hours, filtering under reduced pressure, washing with alcohol, drying, using dichloromethane as solvent, using silica gel column chromatography, the filtrate was concentrated and the solid was precipitated to obtain the final red compound L015 (4.22 g, yield 28 %);
HPLC纯度:大于99.5%;HPLC purity: greater than 99.5%;
质谱:计算值为1018.35;测试值为1018.30。Mass Spec: Calculated 1018.35; Tested 1018.30.
元素分析:计算值C:67.23%;H:6.14%;F:1.87%;Ir:18.88%;N:2.75;O:3.14%;测试值为:C:67.20%;H:6.15%;F:1.88%;Ir:18.90%;N:2.75;O:3.15%;Elemental Analysis: Calculated C: 67.23%; H: 6.14%; F: 1.87%; Ir: 18.88%; N: 2.75; O: 3.14%; Tested: C: 67.20%; H: 6.15%; F: 1.88%; Ir: 18.90%; N: 2.75; O: 3.15%;
具体的,步骤1)至2)的反应式如下:Specifically, the reaction formula of steps 1) to 2) is as follows:
本实施例中,所述铱金属配合物在制备有机电致发光器件产品中的应用。In this embodiment, the iridium metal complex is used in the preparation of organic electroluminescent device products.
实施例3Example 3
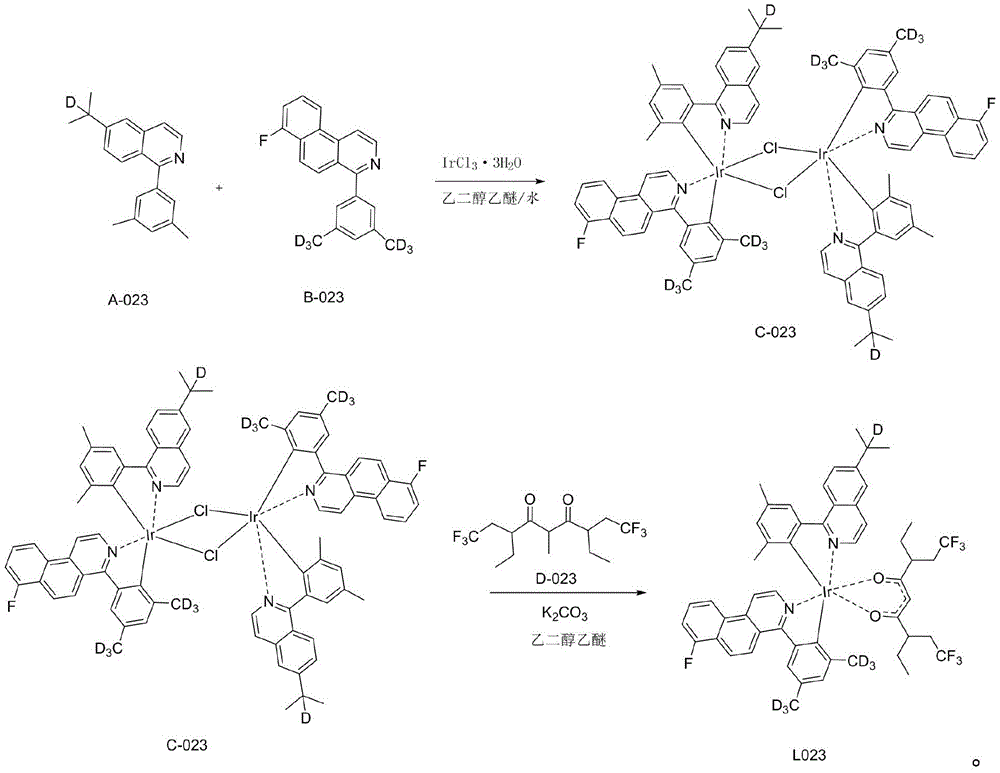
一种铱金属配合物L023,即编号为L023的化合物,具体合成步骤如下:A kind of iridium metal complex L023, namely the compound of numbering L023, concrete synthesis steps are as follows:
1)在氮气保护体系下,称取A-023(28.36mmol,7.84g)和B-006(28.36mmol,8.7g)IrCl3·3H2O(28.36mmol,10g)加入反应体系中,加入500mL乙二醇乙醚和167mL纯净水的混合溶液,氮气保护下120℃回流24小时,然后冷却到室温,有沉淀析出,将沉淀减压过滤,用水、无水乙醇、石油醚依次冲洗烘干,得到红色粉末的桥联配体C-023(11.25g,产率为49%);1) Under nitrogen protection system, weigh A-023 (28.36mmol, 7.84g) and B-006 (28.36mmol, 8.7g) IrCl 3 ·3H 2 O (28.36mmol, 10g) into the reaction system, add 500mL The mixed solution of ethylene glycol ether and 167 mL of pure water was refluxed at 120°C for 24 hours under nitrogen protection, and then cooled to room temperature, and a precipitate was precipitated. The precipitate was filtered under reduced pressure, washed with water, absolute ethanol, and petroleum ether in turn to obtain Bridging ligand C-023 as red powder (11.25 g, 49% yield);
2)称取中间体C-023(6.95mmol,11.25g),加入配体D-023(20.85mmol,6.97g),再向体系中加乙二醇乙醚300mL和碳酸钾(7.4g),氮气保护下,120℃搅拌24小时,减压过滤,醇洗,烘干,用二氯甲烷做溶剂,用硅胶柱层析,滤液浓缩固体析出,得到最终红色的化合物L023(4.1g,产率27%);2) Weigh intermediate C-023 (6.95mmol, 11.25g), add ligand D-023 (20.85mmol, 6.97g), add ethylene glycol ether 300mL and potassium carbonate (7.4g) to the system, nitrogen Under the protection, stirred at 120 ° C for 24 hours, filtered under reduced pressure, washed with alcohol, dried, used dichloromethane as solvent, used silica gel column chromatography, the filtrate was concentrated and the solid was precipitated to obtain the final red compound L023 (4.1 g, yield 27 %);
HPLC纯度:大于99.5%;HPLC purity: greater than 99.5%;
质谱:计算值为1093.27;测试值为1093.30。Mass Spec: Calculated 1093.27; Tested 1093.30.
元素分析:计算值C:59.33%;H:5.44%;F:12.16%;Ir:17.58%;N:2.56;O:2.93%;测试值为:C:59.30%;H:5.45%;F:12.15%;Ir:17.60%;N:2.856;O:2.90%;Elemental Analysis: Calculated C: 59.33%; H: 5.44%; F: 12.16%; Ir: 17.58%; N: 2.56; O: 2.93%; Tested: C: 59.30%; H: 5.45%; F: 12.15%; Ir: 17.60%; N: 2.856; O: 2.90%;
具体的,步骤1)至2)的反应式如下:Specifically, the reaction formula of steps 1) to 2) is as follows:
本实施例中,所述铱金属配合物在制备有机电致发光器件产品中的应用。In this embodiment, the iridium metal complex is used in the preparation of organic electroluminescent device products.
实施例4Example 4
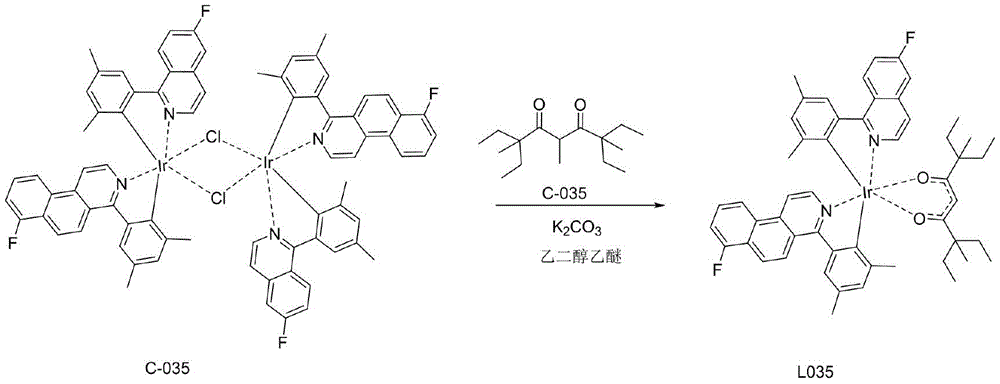
一种铱金属配合物L035,即编号为L035的化合物,具体合成步骤如下:1)在氮气保护体系下,称取A-035(28.36mmol,8.55g)和B-035(28.36mmol,7.13g)IrCl3·3H2O(28.36mmol,10g)加入反应体系中,加入500mL乙二醇乙醚和167mL纯净水的混合溶液,氮气保护下120℃回流24小时,然后冷却到室温,有沉淀析出,将沉淀减压过滤,用水、无水乙醇、石油醚依次冲洗烘干,得到红色粉末的桥联配体C-035(11.70g,产率为53%);A kind of iridium metal complex L035, namely the compound numbered L035, the specific synthesis steps are as follows: 1) under nitrogen protection system, weigh A-035 (28.36mmol, 8.55g) and B-035 (28.36mmol, 7.13g) ) IrCl 3 ·3H 2 O (28.36 mmol, 10 g) was added to the reaction system, a mixed solution of 500 mL of ethylene glycol ether and 167 mL of purified water was added, refluxed at 120° C. for 24 hours under nitrogen protection, and then cooled to room temperature, there was precipitation, The precipitate was filtered under reduced pressure, washed with water, anhydrous ethanol, and petroleum ether in turn and dried to obtain the bridged ligand C-035 (11.70 g, 53% yield) as a red powder;
2)称取中间体C-035(7.5mmol,11.70g),加入配体D-035(22.50mmol,5.72g),再向体系中加乙二醇乙醚300mL和碳酸钾(8.0g),氮气保护下,120℃搅拌24小时,减压过滤,醇洗,烘干,用二氯甲烷做溶剂,用硅胶柱层析,滤液浓缩固体析出,得到最终红色的化合物L035(4.05g,产率27.5%);2) Weigh intermediate C-035 (7.5mmol, 11.70g), add ligand D-035 (22.50mmol, 5.72g), then add ethylene glycol ether 300mL and potassium carbonate (8.0g) to the system, nitrogen Under the protection, stirred at 120 ° C for 24 hours, filtered under reduced pressure, washed with alcohol, dried, used dichloromethane as solvent, used silica gel column chromatography, the filtrate was concentrated and the solid was precipitated to obtain the final red compound L035 (4.05 g, yield 27.5 %);
HPLC纯度:大于99.5%;HPLC purity: greater than 99.5%;
质谱:计算值为982.25;测试值为982.26。Mass spectrum: Calculated value is 982.25; Test value is 982.26.
元素分析:计算值C:64.81%;H:5.64%;F:3.87%;Ir:19.57%;N:2.85;O:3.26%;测试值为:C:64.80%;H:5.60%;F:3.84%;Ir:19.60%;N:2.86;O:3.25%;Elemental Analysis: Calculated C: 64.81%; H: 5.64%; F: 3.87%; Ir: 19.57%; N: 2.85; O: 3.26%; Tested: C: 64.80%; H: 5.60%; F: 3.84%; Ir: 19.60%; N: 2.86; O: 3.25%;
具体的,步骤1)至2)的反应式如下:Specifically, the reaction formula of steps 1) to 2) is as follows:
本实施例中,所述铱金属配合物在制备有机电致发光器件产品中的应用。In this embodiment, the iridium metal complex is used in the preparation of organic electroluminescent device products.
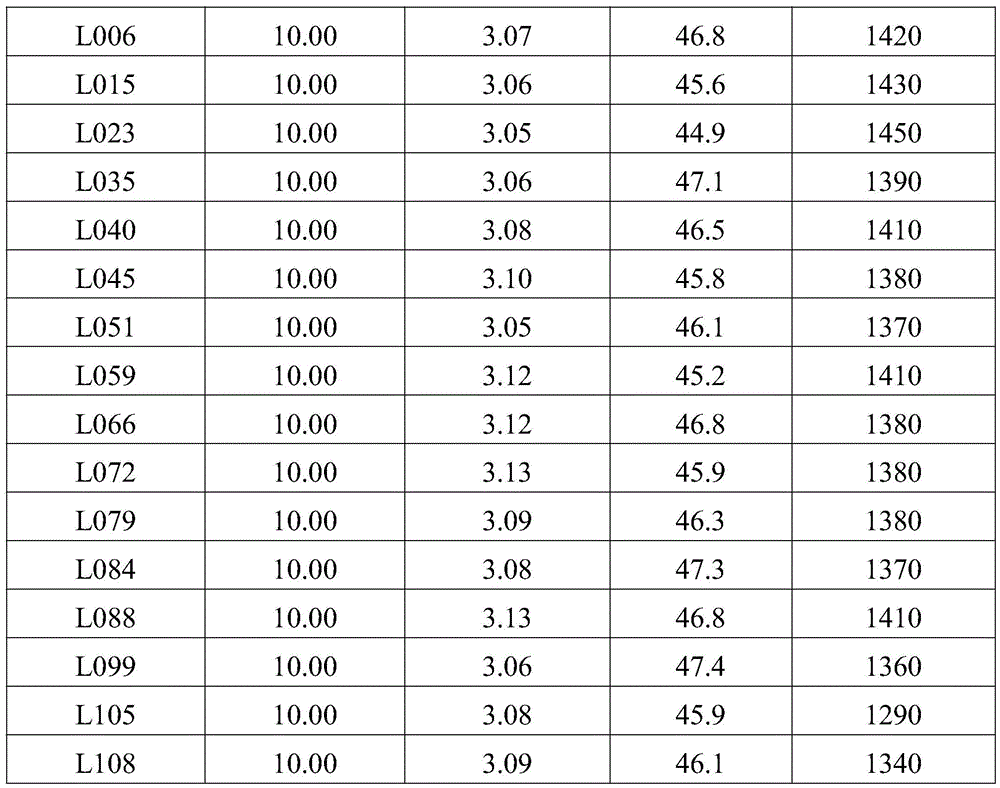
本实施例中,所述铱金属配合物在制备有机电致发光器件产品中的应用。其他化合物的合成方法与上述相同,在此不一一赘述,其他合成实施例质谱或分子式如下表所示:In this embodiment, the iridium metal complex is used in the preparation of organic electroluminescent device products. The synthetic methods of other compounds are the same as above, and will not be repeated here. The mass spectra or molecular formulas of other synthetic examples are shown in the following table:
本发明还提供了一种有机电致发光器件,所述有机电致发光器件由所述的铱金属配合物制成,更具体的为由化学式1所示的铱金属配合物制成。The present invention also provides an organic electroluminescence device, wherein the organic electroluminescence device is made of the iridium metal complex, more specifically, the iridium metal complex shown in Chemical Formula 1.
实施例5Example 5
使用实施例1制备的化合物L006制备有机电致发光器件,其更具体的:An organic electroluminescent device was prepared using the compound L006 prepared in Example 1, more specifically:
将涂层厚度为的ITO玻璃基板放在蒸馏水中清洗2次,超声波洗涤30分钟,用蒸馏水反复清洗2次,超声波洗涤10分钟,蒸馏水清洗结束后,异丙醇、丙酮、甲醇等溶剂按顺序超声波洗涤以后干燥,转移到等离子体清洗机里,将上述基板洗涤5分钟,送到蒸镀机里。首先ITO(阳极)上面蒸镀紧接着蒸镀主体物质化合物H1和掺杂物质化合物L00695:5重量比混合蒸镀蒸镀电子传输层蒸镀电子注入层蒸镀阴极形式制备得到有机电致发光器件。对得到的器件的性能发光特性测试,测量采用KEITHLEY2400型源测量单元,CS-2000分光辐射亮度计,以评价驱动电压,发光亮度,发光效率。The coating thickness is The ITO glass substrate was washed twice in distilled water, ultrasonically washed for 30 minutes, repeatedly washed with distilled water for 2 times, and ultrasonically washed for 10 minutes. It was transferred to a plasma cleaner, the substrate was washed for 5 minutes, and then sent to a vapor deposition machine. First, ITO (anode) is evaporated on top followed by evaporation Host compound H1 and dopant compound L00695:5 weight ratio mixed vapor deposition Evaporated Electron Transport Layer Evaporated electron injection layer Evaporated cathode The organic electroluminescent device was prepared in the form of . To test the performance luminescence characteristics of the obtained device, KEITHLEY2400 source measurement unit and CS-2000 spectroradiometer were used to evaluate the driving voltage, luminous brightness and luminous efficiency.
参照上述方法,将化合物L006分别替换为L015、L023、L035、L040、L045、L051、L059、L066、L072、L079、L084、L088、L099、L105、L108,制备得到相应化合物的有机电致发光器件。Referring to the above method, compound L006 was replaced with L015, L023, L035, L040, L045, L051, L059, L066, L072, L079, L084, L088, L099, L105, L108, respectively, to prepare organic electroluminescent devices of corresponding compounds. .
比较例1Comparative Example 1
按照实施例5相同的方法制备有机电致发光器件,器件结构化合物结构如下:The organic electroluminescent device was prepared according to the same method as in Example 5, and the structure of the device structure compound was as follows:
对制备的有机电致发光器件进行与实施例5相同的检测,结果见表1。The prepared organic electroluminescent device was tested in the same manner as in Example 5, and the results are shown in Table 1.
表1实施例5以及对比例1中有机电致发光器件检测结果Table 1 Test results of organic electroluminescent devices in Example 5 and Comparative Example 1
表1可以看出,使用本发明提供铱金属配合物作为发光层掺杂材料所制备的有机电致发光器件与使用比较化合物对比例1作为发光层掺杂材料所制备的有机电致发光器件相比,驱动电压明显降低,电流效率和寿命得到显著提高。As can be seen from Table 1, the organic electroluminescent device prepared by using the iridium metal complex provided by the present invention as the doping material for the light-emitting layer is similar to the organic electroluminescent device prepared by using the comparative compound Comparative Example 1 as the doping material for the light-emitting layer. ratio, the driving voltage is significantly reduced, and the current efficiency and life are significantly improved.
本说明书中各个实施例采用递进的方式描述,每个实施例重点说明的都是与其他实施例的不同之处,各个实施例之间相同相似部分互相参见即可。The various embodiments in this specification are described in a progressive manner, and each embodiment focuses on the differences from other embodiments, and the same and similar parts between the various embodiments can be referred to each other.
对所公开的实施例的上述说明,使本领域专业技术人员能够实现或使用本发明。对这些实施例的多种修改对本领域的专业技术人员来说将是显而易见的,本文中所定义的一般原理可以在不脱离本发明的精神或范围的情况下,在其它实施例中实现。因此,本发明将不会被限制于本文所示的这些实施例,而是要符合与本文所公开的原理和新颖特点相一致的最宽的范围。The above description of the disclosed embodiments enables any person skilled in the art to make or use the present invention. Various modifications to these embodiments will be readily apparent to those skilled in the art, and the generic principles defined herein may be implemented in other embodiments without departing from the spirit or scope of the invention. Thus, the present invention is not intended to be limited to the embodiments shown herein, but is to be accorded the widest scope consistent with the principles and novel features disclosed herein.
Claims (10)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110857295.4A CN113563386A (en) | 2021-07-28 | 2021-07-28 | A kind of iridium metal complex and its application |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110857295.4A CN113563386A (en) | 2021-07-28 | 2021-07-28 | A kind of iridium metal complex and its application |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN113563386A true CN113563386A (en) | 2021-10-29 |
Family
ID=78168451
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110857295.4A Pending CN113563386A (en) | 2021-07-28 | 2021-07-28 | A kind of iridium metal complex and its application |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113563386A (en) |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20200313097A1 (en) * | 2019-03-29 | 2020-10-01 | Samsung Electronics Co., Ltd. | Organometallic compound, organic light-emitting device including the same, and electronic apparatus including the organic light-emitting device |
-
2021
- 2021-07-28 CN CN202110857295.4A patent/CN113563386A/en active Pending
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20200313097A1 (en) * | 2019-03-29 | 2020-10-01 | Samsung Electronics Co., Ltd. | Organometallic compound, organic light-emitting device including the same, and electronic apparatus including the organic light-emitting device |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN107021926B (en) | Compound containing azaspirofluorene and nitrogen-containing six-membered heterocycle and application of compound in OLED | |
| CN111253442B (en) | Organic iridium metal complex, preparation method thereof and organic electroluminescent device | |
| CN110642893A (en) | A kind of iridium metal complex and its preparation method and device | |
| CN107586261B (en) | Organic compound containing spiro dibenzosuberene fluorene and application thereof | |
| CN110330531A (en) | A kind of iridium metal complex and its preparation method and application | |
| CN110669077A (en) | Organic phosphorus luminescent material, preparation method thereof and organic electroluminescent device | |
| CN103204880B (en) | A kind of organophosphorus luminescent material, its preparation method and the organic electroluminescence device be made up of it | |
| CN110642892B (en) | Novel iridium metal complex and its preparation method and organic electroluminescent device | |
| CN110922432A (en) | Iridium metal complex, preparation method thereof and organic electroluminescent device | |
| CN111471452A (en) | Organic light-emitting compound, preparation method thereof and organic electroluminescent device | |
| CN111690016A (en) | Iridium coordination compound, preparation method thereof and photoelectric device | |
| CN110498816A (en) | Organic phosphorescent material, preparation method thereof, and organic electroluminescent device | |
| CN110330530A (en) | Iridium metal complex and preparation method thereof and organic electroluminescence device containing the compound | |
| CN111484532A (en) | Phosphor luminescent material, preparation method thereof and organic electroluminescent device | |
| CN111333684A (en) | Organic iridium metal complex and preparation method and application thereof | |
| CN110294735B (en) | Compound with anthracene and phenanthrene as core and application of compound in organic electroluminescent device | |
| CN111153890A (en) | Organic light-emitting material, preparation method thereof and organic electroluminescent device | |
| CN110698517B (en) | Iridium metal complex and its preparation method and organic electroluminescent device | |
| CN112479901B (en) | Organic compound with indeno-anthracene derivative as core and application thereof | |
| CN108947925B (en) | 1,3,4-oxadiazole derivatives, materials and organic electroluminescent devices | |
| CN111747962A (en) | A kind of organic electroluminescent compound and its preparation method and organic electroluminescent device | |
| CN119462771A (en) | An organic metal iridium complex, preparation method and organic electroluminescent device containing the same | |
| CN106699731A (en) | Maleic hydrazine derivative and preparation method and application thereof | |
| CN111484839A (en) | Organic light-emitting compound, preparation method thereof and organic electroluminescent device | |
| CN112479905A (en) | Organic compound containing benzoanthracene fluorene and diarylamine and application thereof in organic electroluminescent device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination |