CN113433249B - Method for detecting minocycline intermediate related substances by high performance liquid chromatography - Google Patents
Method for detecting minocycline intermediate related substances by high performance liquid chromatography Download PDFInfo
- Publication number
- CN113433249B CN113433249B CN202110825303.7A CN202110825303A CN113433249B CN 113433249 B CN113433249 B CN 113433249B CN 202110825303 A CN202110825303 A CN 202110825303A CN 113433249 B CN113433249 B CN 113433249B
- Authority
- CN
- China
- Prior art keywords
- solution
- impurity
- minocycline
- minocycline intermediate
- following
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 229960004023 minocycline Drugs 0.000 title claims abstract description 35
- 238000000034 method Methods 0.000 title claims abstract description 23
- 239000000126 substance Substances 0.000 title claims abstract description 15
- 238000004128 high performance liquid chromatography Methods 0.000 title claims abstract description 11
- DYKFCLLONBREIL-KVUCHLLUSA-N minocycline Chemical compound C([C@H]1C2)C3=C(N(C)C)C=CC(O)=C3C(=O)C1=C(O)[C@@]1(O)[C@@H]2[C@H](N(C)C)C(O)=C(C(N)=O)C1=O DYKFCLLONBREIL-KVUCHLLUSA-N 0.000 title claims abstract 10
- 239000012535 impurity Substances 0.000 claims abstract description 57
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 claims abstract description 18
- 238000002360 preparation method Methods 0.000 claims abstract description 12
- 229950000614 sancycline Drugs 0.000 claims abstract description 6
- 238000010829 isocratic elution Methods 0.000 claims abstract description 4
- XDVCLKFLRAWGIT-ADOAZJKMSA-N sancycline Chemical compound C([C@H]1C2)C3=CC=CC(O)=C3C(=O)C1=C(O)[C@@]1(O)[C@@H]2[C@H](N(C)C)C(O)=C(C(N)=O)C1=O XDVCLKFLRAWGIT-ADOAZJKMSA-N 0.000 claims abstract 2
- 239000000243 solution Substances 0.000 claims description 32
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 31
- 239000000523 sample Substances 0.000 claims description 22
- 238000007865 diluting Methods 0.000 claims description 11
- 239000012085 test solution Substances 0.000 claims description 7
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 claims description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 6
- 238000001914 filtration Methods 0.000 claims description 6
- 239000011259 mixed solution Substances 0.000 claims description 6
- XUKUURHRXDUEBC-SXOMAYOGSA-N (3s,5r)-7-[2-(4-fluorophenyl)-3-phenyl-4-(phenylcarbamoyl)-5-propan-2-ylpyrrol-1-yl]-3,5-dihydroxyheptanoic acid Chemical compound C=1C=CC=CC=1C1=C(C=2C=CC(F)=CC=2)N(CC[C@@H](O)C[C@H](O)CC(O)=O)C(C(C)C)=C1C(=O)NC1=CC=CC=C1 XUKUURHRXDUEBC-SXOMAYOGSA-N 0.000 claims description 5
- AAEQXEDPVFIFDK-UHFFFAOYSA-N 3-(4-fluorobenzoyl)-2-(2-methylpropanoyl)-n,3-diphenyloxirane-2-carboxamide Chemical compound C=1C=CC=CC=1NC(=O)C1(C(=O)C(C)C)OC1(C=1C=CC=CC=1)C(=O)C1=CC=C(F)C=C1 AAEQXEDPVFIFDK-UHFFFAOYSA-N 0.000 claims description 5
- 238000002347 injection Methods 0.000 claims description 4
- 239000007924 injection Substances 0.000 claims description 4
- 239000012488 sample solution Substances 0.000 claims description 4
- 239000012982 microporous membrane Substances 0.000 claims description 3
- 229910000403 monosodium phosphate Inorganic materials 0.000 claims description 2
- 235000019799 monosodium phosphate Nutrition 0.000 claims description 2
- AJPJDKMHJJGVTQ-UHFFFAOYSA-M sodium dihydrogen phosphate Chemical compound [Na+].OP(O)([O-])=O AJPJDKMHJJGVTQ-UHFFFAOYSA-M 0.000 claims description 2
- 238000001514 detection method Methods 0.000 abstract description 11
- 239000007788 liquid Substances 0.000 abstract description 5
- 230000015572 biosynthetic process Effects 0.000 abstract description 3
- 238000000926 separation method Methods 0.000 abstract description 3
- 238000003786 synthesis reaction Methods 0.000 abstract description 3
- 239000003112 inhibitor Substances 0.000 abstract description 2
- 238000012544 monitoring process Methods 0.000 abstract description 2
- 239000007787 solid Substances 0.000 abstract description 2
- FFTVPQUHLQBXQZ-KVUCHLLUSA-N (4s,4as,5ar,12ar)-4,7-bis(dimethylamino)-1,10,11,12a-tetrahydroxy-3,12-dioxo-4a,5,5a,6-tetrahydro-4h-tetracene-2-carboxamide Chemical compound C1C2=C(N(C)C)C=CC(O)=C2C(O)=C2[C@@H]1C[C@H]1[C@H](N(C)C)C(=O)C(C(N)=O)=C(O)[C@@]1(O)C2=O FFTVPQUHLQBXQZ-KVUCHLLUSA-N 0.000 description 25
- 238000012360 testing method Methods 0.000 description 15
- 238000011084 recovery Methods 0.000 description 11
- 238000005303 weighing Methods 0.000 description 6
- 239000000047 product Substances 0.000 description 5
- MTCQOMXDZUULRV-ADOAZJKMSA-N (4s,4as,5ar,12ar)-4-(dimethylamino)-1,10,11,12a-tetrahydroxy-3,12-dioxo-4a,5,5a,6-tetrahydro-4h-tetracene-2-carboxamide Chemical compound C1C2=CC=CC(O)=C2C(O)=C2[C@@H]1C[C@H]1[C@H](N(C)C)C(=O)C(C(N)=O)=C(O)[C@@]1(O)C2=O MTCQOMXDZUULRV-ADOAZJKMSA-N 0.000 description 4
- 239000011550 stock solution Substances 0.000 description 4
- 238000004458 analytical method Methods 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000012528 membrane Substances 0.000 description 3
- 239000013558 reference substance Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 230000000844 anti-bacterial effect Effects 0.000 description 2
- 238000011835 investigation Methods 0.000 description 2
- 238000002372 labelling Methods 0.000 description 2
- 238000012417 linear regression Methods 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 241000588724 Escherichia coli Species 0.000 description 1
- 229940126524 Semi-synthetic tetracycline antibiotic Drugs 0.000 description 1
- 241000191967 Staphylococcus aureus Species 0.000 description 1
- 241000194017 Streptococcus Species 0.000 description 1
- 239000004098 Tetracycline Substances 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000012467 final product Substances 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000008267 milk Substances 0.000 description 1
- 210000004080 milk Anatomy 0.000 description 1
- 235000013336 milk Nutrition 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 239000012086 standard solution Substances 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 238000011003 system suitability test Methods 0.000 description 1
- 229960002180 tetracycline Drugs 0.000 description 1
- 229930101283 tetracycline Natural products 0.000 description 1
- 235000019364 tetracycline Nutrition 0.000 description 1
- 150000003522 tetracyclines Chemical class 0.000 description 1
- 238000012795 verification Methods 0.000 description 1
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N30/00—Investigating or analysing materials by separation into components using adsorption, absorption or similar phenomena or using ion-exchange, e.g. chromatography or field flow fractionation
- G01N30/02—Column chromatography
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N30/00—Investigating or analysing materials by separation into components using adsorption, absorption or similar phenomena or using ion-exchange, e.g. chromatography or field flow fractionation
- G01N30/02—Column chromatography
- G01N30/04—Preparation or injection of sample to be analysed
- G01N30/06—Preparation
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N30/00—Investigating or analysing materials by separation into components using adsorption, absorption or similar phenomena or using ion-exchange, e.g. chromatography or field flow fractionation
- G01N30/02—Column chromatography
- G01N30/50—Conditioning of the sorbent material or stationary liquid
- G01N30/52—Physical parameters
- G01N30/54—Temperature
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N30/00—Investigating or analysing materials by separation into components using adsorption, absorption or similar phenomena or using ion-exchange, e.g. chromatography or field flow fractionation
- G01N30/02—Column chromatography
- G01N30/62—Detectors specially adapted therefor
- G01N30/74—Optical detectors
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Landscapes
- Physics & Mathematics (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Analytical Chemistry (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Immunology (AREA)
- Pathology (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Investigating Or Analysing Biological Materials (AREA)
Abstract
The invention provides a method for detecting minocycline intermediate related substances by high performance liquid chromatography, which adopts the chromatographic conditions as follows: a chromatographic column: c185.0 μm 4.6 x 250mm; a detector: an ultraviolet detector; column temperature: 30 ℃; wavelength: 355nm; sample introduction amount: 10 mu L of the solution; flow rate: 0.6mL/min. The method adopts high performance liquid chromatography, the detector is an ultraviolet detector or a diode array detector, isocratic elution is adopted, the instrument requirement is low, the preparation of the mobile phase is simple, and the detection is rapid; triethylamine is a tailing inhibitor, so that the separation can be effectively improved, the main peak symmetry is good, and the triethylamine and impurities are effectively separated; the chromatographic condition can effectively separate minocycline intermediate, sancycline, from various related substances, and can be used for quality monitoring of liquid and solid samples in the synthesis process.
Description
Technical Field
The invention relates to the technical field of drug analysis, in particular to a method for detecting minocycline intermediate related substances by high performance liquid chromatography.
Background
Minocycline, also known as minocycline and minocycline, is a semi-synthetic tetracycline antibiotic with broad-spectrum antibacterial effect, has the strongest antibacterial effect in the genus, is characterized by high efficiency, quick acting and long acting, can overcome staphylococcus aureus, streptococcus and escherichia coli of milk tetracycline, and has quick oral administration absorption and no food influence. The sancycline (demethoxytetracycline) is an important intermediate of minocycline, is an important semi-synthetic intermediate raw material, generates byproducts, and is easy to epimerize in the production and storage processes to cause the quality change of products, so that the quality of the sancycline not only influences the overall reaction yield, but also indirectly influences the quality of final products. The analysis and detection of the method have important effects on condition exploration and reaction control of the previous synthesis steps and subsequent reaction yield improvement, so that the establishment of an efficient, rapid, simple, stable and effective analysis and detection method is very necessary for analyzing and detecting the minocycline intermediate.
Disclosure of Invention
The invention aims to solve the defects in the prior art and provides a method for detecting minocycline intermediate related substances by high performance liquid chromatography, which has strong specificity, good linearity and good accuracy.
The technical scheme adopted by the invention is as follows: a method for detecting minocycline intermediate related substances by high performance liquid chromatography, which adopts the chromatographic conditions as follows:
a chromatographic column: c18,5.0 μm 4.6 x 250mm or C18,3.0 μm 4.6 x 150mm;
a detector: an ultraviolet detector or a diode array detector;
column temperature: 30 ℃;
wavelength: 355nm;
sample introduction amount: 10 mu L of the solution;
flow rate: 0.6mL/min;
mobile phase: solution A: methanol: acetonitrile =600:240, 160;
isocratic elution was used.
Further, the preparation method of the solution A comprises the following steps: 0.16M sodium dihydrogen phosphate is adjusted to pH 6 + -0.2 by triethylamine and shaken up for standby.
Further, the method also includes the preparation of a test solution, a control solution and a system suitability solution.
Further, the preparation method of the test solution comprises the following steps: taking a minocycline intermediate sample, dissolving or diluting the minocycline intermediate sample with 0.1M hydrochloric acid, diluting to a constant volume of 1mg/mL, and filtering with a microporous filter membrane before sample injection.
Further, the preparation method of the control solution comprises the following steps: precisely measuring 1mL of sample solution, diluting with 0.1M hydrochloric acid in a 100mL volumetric flask, diluting to constant volume to scale, shaking, and filtering with microporous membrane before sample introduction.
Further, the preparation method of the system applicability solution comprises the following steps: preparing a mixed solution of 7 impurities and a minocycline intermediate control solution to be used as a system applicability solution; the concentration of 7 impurities in the mixed solution is as follows: the concentration of the impurity A, the impurity D and the impurity F is respectively 10ug/mL, the concentration of the impurity B, the impurity C, the impurity E and the impurity G is 2ug/mL, and the concentration of the minocycline intermediate is 1mg/mL.
The beneficial effects obtained by the invention are as follows: the method adopts high performance liquid chromatography, the detector is an ultraviolet detector or a diode array detector, isocratic elution is adopted, the instrument requirement is low, the preparation of the mobile phase is simple, and the detection is rapid; triethylamine is a tailing inhibitor, so that the separation can be effectively improved, the main peak symmetry is good, and the triethylamine and impurities are effectively separated; the chromatographic condition can effectively separate minocycline intermediate, sancycline, from various related substances, and can be used for quality monitoring of liquid and solid samples in the synthesis process; the detection method is accurate and feasible through methodology verification, simple and convenient to operate and good in linearity, and can be used for detecting related substances of the minocycline intermediate.
Drawings
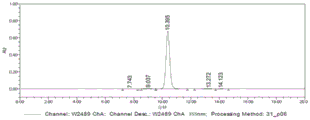
FIG. 1 is a chromatogram of a system of the present invention;
FIG. 2 is a blank solvent chromatogram of the present invention;
FIG. 3 is a chromatogram of crude product of the present invention;
FIG. 4 is a chromatogram of a finished product of the present invention.
Detailed Description
The technical solutions in the embodiments of the present invention will be clearly and completely described below with reference to the drawings in the embodiments of the present invention, and it is obvious that the described embodiments are only a part of the embodiments of the present invention, and not all of the embodiments.
Example 1: experiment of system applicability
Preparing a system applicability solution: the 7 known impurities in the table above and the minocycline intermediate, the sancycline control, are configured into a mixed solution to be used as a system applicability solution, the concentrations of the 7 related impurities in the mixed solution are 10ug/mL respectively of impurity A, impurity D and impurity F, the concentrations of impurity B, impurity C, impurity E and impurity G are 2ug/mL, and the concentration of the minocycline intermediate is 1mg/mL.
And precisely measuring 10uL of the system applicability solution, and injecting the solution into a high performance liquid chromatograph.
The system suitability chromatogram is shown in FIG. 1, and the blank solvent chromatogram is shown in FIG. 2. The peak time and system suitability test results for each impurity are shown in table 1:
TABLE 1 System suitability study
And (4) conclusion: under the wavelength of 355nm, the blank solvent does not generate peaks at the main peak and each impurity peak, the separation degrees among the impurities and between the impurities and the main peak meet the requirements, and the theoretical plate number and the symmetry factor meet the measurement requirements of related substances.
Example 2: linear range test
Precisely weighing appropriate amounts of 7 impurities and minocycline intermediate reference substances, respectively dissolving and diluting with 0.1M hydrochloric acid to obtain a series of linear sample solutions, and shaking up.
And precisely measuring 10uL of the system applicability solution, injecting the solution into a high performance liquid chromatograph, recording peak area, and drawing a linear curve and an equation. The results are shown in Table 2:
TABLE 2 Linear investigation of minocycline intermediate and impurity A, B, C, D, E, F, G
And (4) conclusion: the linear regression equation and R ^2 of the 7 impurities and the minocycline intermediate meet the requirements, and the linear regression is remarkable.
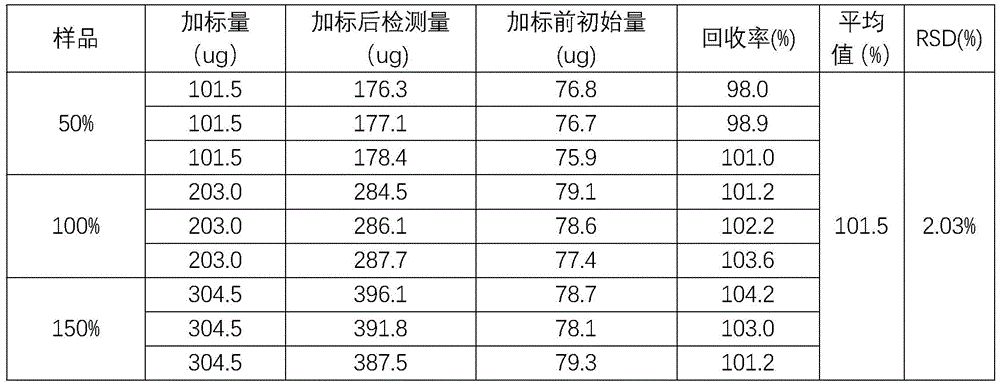
Example 3: recovery test
Preparing an unlabeled sample solution: 20mg of the sample is precisely weighed, dissolved in a 20mL volumetric flask with 0.1M hydrochloric acid, diluted to a constant volume and shaken up.
Impurity control stock solution: taking appropriate amount of reference substances of impurities A, B, D, C, D, E, F and G, dissolving with 0.1M hydrochloric acid to obtain a mixed reference substance solution containing 100ug/mL of impurities A, D and F and 20ug/mL of impurities B, C, E and G.
Preparing a standard solution:
50% spiked solution: precisely weighing 20mg of a test article, dissolving the test article in a 20mL volumetric flask by using 0.1M hydrochloric acid, adding 1mL of impurity reference article stock solution, fixing the volume by using 0.1M hydrochloric acid, and shaking up; triplicate samples were prepared.
50% spiked solution: precisely weighing 20mg of a test article, dissolving the test article in a 20mL volumetric flask by using 0.1M hydrochloric acid, adding 2mL of impurity reference article stock solution, fixing the volume by using 0.1M hydrochloric acid, and shaking up; triplicate samples were prepared.
50% spiked solution: precisely weighing 20mg of a test article, dissolving the test article in a 20mL volumetric flask by using 0.1M hydrochloric acid, adding 3mL of impurity reference article stock solution, performing constant volume by using 0.1M hydrochloric acid, and shaking up; triplicate samples were prepared.
Each of the above samples was diluted 100-fold with 0.1M hydrochloric acid to prepare a corresponding control solution.
10uL of each of the above solutions was injected into a liquid chromatograph, and the content of each impurity was measured.
The results of the sample testing before labeling are shown in Table 3:
TABLE 3 sample test results before labeling
| Name (R) | Content% |
| Impurity A | 0.80 |
| Impurity B | Not detected out |
| Impurity C | Not detected out |
| Impurity D | 0.41 |
| Impurity E | Undetected |
| Impurity F | 0.24 |
| Impurity G | Not detected out |
The recovery of the impurity A, B, C, D, E, F, G was calculated separately and the results are shown in tables 4-10:
TABLE 4 impurity A recovery Rate investigation
TABLE 5 impurity B recovery considerations
TABLE 6 impurity C recovery test
TABLE 7 impurity D recovery considerations
TABLE 8 impurity E recovery test
TABLE 9 impurity F recovery test
TABLE 10 impurity G recovery test
And (4) conclusion: according to the results of the recovery rates of the impurities, the recovery rate of the impurities is between 90% and 110%, the RSD (remote subscriber identification) is not more than 5%, the measurement requirement of the impurities is met, and the method is good in accuracy.
Example 4: actual sample detection
Preparing a test solution:
crude test solution: precisely weighing 25mg of a minocycline intermediate sample, dissolving the minocycline intermediate sample with 0.1M hydrochloric acid, diluting to a constant volume of 25mL, and filtering with a microporous filter membrane before sample injection.
Finished product test solution: precisely weighing 25mg of a minocycline intermediate sample, dissolving the minocycline intermediate sample with 0.1M hydrochloric acid, diluting to a constant volume of 25mL, and filtering with a microporous filter membrane before sample injection.
10uL of the sample was precisely measured, and the results were measured and calculated according to the above chromatographic conditions.
The crude chromatogram is shown in FIG. 3, and the detection results are shown in Table 11:
TABLE 11 crude detection results
The chromatogram of the final product is shown in FIG. 4, and the detection results are shown in Table 12:
TABLE 12 test results of the finished products
And (4) conclusion: the detection result of the actual sample shows that the method can detect 7 impurities in the intermediate and can realize the detection of related substances of the minocycline intermediate sample.
The relevant known impurities in the present invention are, respectively, as shown in table 13:
TABLE 13 structural formula of related impurities
The above description is only for the preferred embodiment of the present invention, but the scope of the present invention is not limited thereto, and any person skilled in the art should be considered to be within the technical scope of the present invention, and the technical solutions and the inventive concepts thereof according to the present invention should be equivalent or changed within the scope of the present invention.
Claims (3)
1. A method for detecting minocycline intermediate related substances by high performance liquid chromatography is characterized by comprising the following steps: the method adopts the chromatographic conditions as follows:
a chromatographic column: c18,5.0 μm 4.6 x 250mm or C18,3.0 μm 4.6 x 150mm;
a detector: an ultraviolet detector or a diode array detector;
column temperature: 30 ℃;
wavelength: 355nm;
sample introduction amount: 10 mu L of the solution;
flow rate: 0.6mL/min;
mobile phase: solution A: methanol: acetonitrile = 600;
isocratic elution is adopted;
the preparation method of the solution A comprises the following steps: adjusting pH of 0.16M sodium dihydrogen phosphate to 6 + -0.2 with triethylamine, and shaking up for use;
the method also comprises the preparation of a test solution, a control solution and a system applicability solution;
the preparation method of the test solution comprises the following steps: dissolving or diluting minocycline intermediate sample with 0.1M hydrochloric acid, diluting to a constant volume of 1mg/mL, and filtering with microporous membrane before sample injection;
the minocycline intermediate related substances comprise sancycline and related impurities, wherein the related impurities have the following structural formula:
2. the method for detecting minocycline intermediate related substances by high performance liquid chromatography according to claim 1, wherein the method comprises the following steps: the preparation method of the control solution comprises the following steps: precisely measuring 1mL of sample solution, diluting with 0.1M hydrochloric acid in a 100mL volumetric flask, diluting to constant volume to scale, shaking, and filtering with microporous membrane before sample introduction.
3. The method for detecting minocycline intermediate related substances by high performance liquid chromatography according to claim 1, wherein the method comprises the following steps: the preparation method of the system applicability solution comprises the following steps: preparing a mixed solution of 7 impurities and a minocycline intermediate control solution to be used as a system applicability solution; the concentration of 7 impurities in the mixed solution is as follows: the concentration of the impurity A, the impurity D and the impurity F is respectively 10ug/mL, the concentration of the impurity B, the impurity C, the impurity E and the impurity G is 2ug/mL, and the concentration of the minocycline intermediate is 1mg/mL.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110825303.7A CN113433249B (en) | 2021-07-21 | 2021-07-21 | Method for detecting minocycline intermediate related substances by high performance liquid chromatography |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110825303.7A CN113433249B (en) | 2021-07-21 | 2021-07-21 | Method for detecting minocycline intermediate related substances by high performance liquid chromatography |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN113433249A CN113433249A (en) | 2021-09-24 |
| CN113433249B true CN113433249B (en) | 2023-03-24 |
Family
ID=77761334
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110825303.7A Active CN113433249B (en) | 2021-07-21 | 2021-07-21 | Method for detecting minocycline intermediate related substances by high performance liquid chromatography |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113433249B (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN117310012B (en) * | 2023-08-21 | 2025-08-08 | 浙江省食品药品检验研究院(《中国现代应用药学》杂志社) | HPLC Detection Method for Minocycline Hydrochloride and Its Impurities |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103387512A (en) * | 2012-05-08 | 2013-11-13 | 成都睿智化学研究有限公司 | Preparation method and intermediate of minocycline |
| CN108324722A (en) * | 2008-05-19 | 2018-07-27 | 帕拉特克药品公司 | The salt and polymorph of tetracycline compound |
| WO2020135508A1 (en) * | 2018-12-26 | 2020-07-02 | 浙江昌海制药有限公司 | Minocycline(ii) p-chlorobenzene sulfonate crystal form, preparation method therefor and use thereof |
| CN111855857A (en) * | 2020-07-30 | 2020-10-30 | 武汉药品医疗器械检验所 | High performance liquid chromatography detection method for minocycline hydrochloride and impurities thereof |
-
2021
- 2021-07-21 CN CN202110825303.7A patent/CN113433249B/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108324722A (en) * | 2008-05-19 | 2018-07-27 | 帕拉特克药品公司 | The salt and polymorph of tetracycline compound |
| CN103387512A (en) * | 2012-05-08 | 2013-11-13 | 成都睿智化学研究有限公司 | Preparation method and intermediate of minocycline |
| WO2020135508A1 (en) * | 2018-12-26 | 2020-07-02 | 浙江昌海制药有限公司 | Minocycline(ii) p-chlorobenzene sulfonate crystal form, preparation method therefor and use thereof |
| CN111855857A (en) * | 2020-07-30 | 2020-10-30 | 武汉药品医疗器械检验所 | High performance liquid chromatography detection method for minocycline hydrochloride and impurities thereof |
Non-Patent Citations (1)
| Title |
|---|
| 高效液相色谱法测定盐酸米诺环素的有关物质;王珑等;《海峡药学》;20061231;第18卷(第01期);85-86 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN113433249A (en) | 2021-09-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN115097023A (en) | High performance liquid chromatography detection method for zolmitamine related substances | |
| CN114646701B (en) | HPLC test method for related substances in L-prolylamide | |
| CN113433249B (en) | Method for detecting minocycline intermediate related substances by high performance liquid chromatography | |
| CN113049687B (en) | Method for detecting ambroxol hydrochloride raw material and injection related substances | |
| CN117486751A (en) | Method for separating omacycline isomers using high performance liquid chromatography | |
| CN115656360B (en) | Quality Control Method for Vonoprazan Fumarate Intermediates | |
| CN107941970A (en) | The method for separating and detecting of D D-pHPGs and its enantiomter | |
| CN113866337B (en) | Mass analysis method for separating and measuring oseltamivir phosphate isomer | |
| CN111579704A (en) | Method for detecting content of N, N' -diacetyl-L-cystine in compound amino acid injection | |
| CN111983055B (en) | Method for separating and measuring rivaroxaban intermediate related substances by using HPLC (high performance liquid chromatography) | |
| CN112986449B (en) | Method for detecting 2-amino-5-nitrothiazole genotoxic impurities in nitazoxanide | |
| CN114755320B (en) | Detection method of 3-amino-6-methoxypyridazine related substances | |
| CN114216988B (en) | Method for detecting related substances of 4- (methylamino) -1- (3-pyridyl) -1-butanone hydrochloride | |
| CN113866318B (en) | Detection method of (6-aminopyridine-2-yl) (1-methylpiperidine-4-yl) methanone dihydrochloride | |
| CN111521693B (en) | Method for detecting isosorbide mononitrate | |
| CN109001342B (en) | High performance liquid chromatography method for detecting N-2, 3-dimethoxybenzyl piperonylethylamine and salt content thereof | |
| CN109239214B (en) | Method for detecting Shakubiqu isomers in Shakubiqu sodium | |
| CN107991404B (en) | A kind of detection method of (2S,5R)-benzyloxyamino piperidine-2-carboxamide | |
| CN1227528C (en) | High performance liquid chromatography analysis method of fenfenzuril and its impurities | |
| CN112394112A (en) | Method for detecting content of hydroxychloroquine nitrogen oxide impurities in hydroxychloroquine sulfate | |
| CN116223650B (en) | Detection Method of Imidazole Chloride by High Performance Liquid Chromatography | |
| CN114324637B (en) | Method for detecting three potential genotoxic impurities in meropenem | |
| CN116008435B (en) | Method for measuring content of 2, 4-dibromo-5-chlorobenzoic acid by high performance liquid chromatography | |
| CN105372343A (en) | Method for separating and measuring substances related to memantine hydrochloride intermediate through gas chromatography | |
| CN110646540B (en) | HPLC-ELSD detection method for Retamolin starting material and intermediate |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |