CN113368132A - Liquid dressing for treating psoriasis - Google Patents
Liquid dressing for treating psoriasis Download PDFInfo
- Publication number
- CN113368132A CN113368132A CN202110662447.5A CN202110662447A CN113368132A CN 113368132 A CN113368132 A CN 113368132A CN 202110662447 A CN202110662447 A CN 202110662447A CN 113368132 A CN113368132 A CN 113368132A
- Authority
- CN
- China
- Prior art keywords
- parts
- zinc
- film
- liquid dressing
- skin
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 239000007788 liquid Substances 0.000 title claims abstract description 56
- 201000004681 Psoriasis Diseases 0.000 title claims abstract description 40
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 40
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims abstract description 32
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 claims abstract description 32
- 229940043810 zinc pyrithione Drugs 0.000 claims abstract description 24
- PICXIOQBANWBIZ-UHFFFAOYSA-N zinc;1-oxidopyridine-2-thione Chemical compound [Zn+2].[O-]N1C=CC=CC1=S.[O-]N1C=CC=CC1=S PICXIOQBANWBIZ-UHFFFAOYSA-N 0.000 claims abstract description 24
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical compound [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 claims abstract description 22
- 239000011707 mineral Substances 0.000 claims abstract description 22
- 229910052711 selenium Inorganic materials 0.000 claims abstract description 22
- 239000011669 selenium Substances 0.000 claims abstract description 22
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 claims abstract description 21
- 229910052500 inorganic mineral Inorganic materials 0.000 claims abstract description 21
- 239000011701 zinc Substances 0.000 claims abstract description 21
- 229910052725 zinc Inorganic materials 0.000 claims abstract description 21
- 239000008213 purified water Substances 0.000 claims abstract description 20
- NOOLISFMXDJSKH-UTLUCORTSA-N (+)-Neomenthol Chemical compound CC(C)[C@@H]1CC[C@@H](C)C[C@@H]1O NOOLISFMXDJSKH-UTLUCORTSA-N 0.000 claims abstract description 16
- NOOLISFMXDJSKH-UHFFFAOYSA-N DL-menthol Natural products CC(C)C1CCC(C)CC1O NOOLISFMXDJSKH-UHFFFAOYSA-N 0.000 claims abstract description 16
- 229940041616 menthol Drugs 0.000 claims abstract description 16
- 239000011787 zinc oxide Substances 0.000 claims abstract description 16
- DTGKSKDOIYIVQL-WEDXCCLWSA-N (+)-borneol Chemical compound C1C[C@@]2(C)[C@@H](O)C[C@@H]1C2(C)C DTGKSKDOIYIVQL-WEDXCCLWSA-N 0.000 claims abstract description 13
- REPVLJRCJUVQFA-UHFFFAOYSA-N (-)-isopinocampheol Natural products C1C(O)C(C)C2C(C)(C)C1C2 REPVLJRCJUVQFA-UHFFFAOYSA-N 0.000 claims abstract description 13
- OSDLLIBGSJNGJE-UHFFFAOYSA-N 4-chloro-3,5-dimethylphenol Chemical compound CC1=CC(O)=CC(C)=C1Cl OSDLLIBGSJNGJE-UHFFFAOYSA-N 0.000 claims abstract description 13
- 229940116229 borneol Drugs 0.000 claims abstract description 13
- CKDOCTFBFTVPSN-UHFFFAOYSA-N borneol Natural products C1CC2(C)C(C)CC1C2(C)C CKDOCTFBFTVPSN-UHFFFAOYSA-N 0.000 claims abstract description 13
- DTGKSKDOIYIVQL-UHFFFAOYSA-N dl-isoborneol Natural products C1CC2(C)C(O)CC1C2(C)C DTGKSKDOIYIVQL-UHFFFAOYSA-N 0.000 claims abstract description 13
- 239000002994 raw material Substances 0.000 claims abstract description 13
- 239000000126 substance Substances 0.000 claims abstract description 12
- 239000003795 chemical substances by application Substances 0.000 claims abstract description 7
- 229960004756 ethanol Drugs 0.000 claims abstract description 6
- 235000010755 mineral Nutrition 0.000 claims description 21
- 229920000036 polyvinylpyrrolidone Polymers 0.000 claims description 21
- 239000001267 polyvinylpyrrolidone Substances 0.000 claims description 21
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 claims description 21
- 238000010438 heat treatment Methods 0.000 claims description 15
- 238000003756 stirring Methods 0.000 claims description 15
- 239000004372 Polyvinyl alcohol Substances 0.000 claims description 11
- 229920002451 polyvinyl alcohol Polymers 0.000 claims description 11
- DHFUFHYLYSCIJY-WSGIOKLISA-N CCCCCCCCCCCCCCCCCCCCO[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O Chemical compound CCCCCCCCCCCCCCCCCCCCO[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O DHFUFHYLYSCIJY-WSGIOKLISA-N 0.000 claims description 6
- 238000011049 filling Methods 0.000 claims description 6
- 238000002156 mixing Methods 0.000 claims description 5
- 230000035807 sensation Effects 0.000 claims description 5
- 238000005303 weighing Methods 0.000 claims description 5
- JIAARYAFYJHUJI-UHFFFAOYSA-L zinc dichloride Chemical compound [Cl-].[Cl-].[Zn+2] JIAARYAFYJHUJI-UHFFFAOYSA-L 0.000 claims description 4
- FMRLDPWIRHBCCC-UHFFFAOYSA-L Zinc carbonate Chemical compound [Zn+2].[O-]C([O-])=O FMRLDPWIRHBCCC-UHFFFAOYSA-L 0.000 claims description 2
- 239000011667 zinc carbonate Substances 0.000 claims description 2
- 235000004416 zinc carbonate Nutrition 0.000 claims description 2
- 229910000010 zinc carbonate Inorganic materials 0.000 claims description 2
- 239000011592 zinc chloride Substances 0.000 claims description 2
- 235000005074 zinc chloride Nutrition 0.000 claims description 2
- NWONKYPBYAMBJT-UHFFFAOYSA-L zinc sulfate Chemical compound [Zn+2].[O-]S([O-])(=O)=O NWONKYPBYAMBJT-UHFFFAOYSA-L 0.000 claims description 2
- 229960001763 zinc sulfate Drugs 0.000 claims description 2
- 229910000368 zinc sulfate Inorganic materials 0.000 claims description 2
- 238000000034 method Methods 0.000 claims 2
- 239000012530 fluid Substances 0.000 claims 1
- 230000000694 effects Effects 0.000 abstract description 18
- 208000017520 skin disease Diseases 0.000 abstract description 14
- 208000003251 Pruritus Diseases 0.000 abstract description 11
- 239000003814 drug Substances 0.000 abstract description 9
- 230000012010 growth Effects 0.000 abstract description 7
- 230000007794 irritation Effects 0.000 abstract description 6
- 230000007803 itching Effects 0.000 abstract description 6
- 230000002195 synergetic effect Effects 0.000 abstract description 6
- 208000024891 symptom Diseases 0.000 abstract description 5
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 abstract description 4
- 244000052616 bacterial pathogen Species 0.000 abstract description 4
- 230000009286 beneficial effect Effects 0.000 abstract description 4
- 229940079593 drug Drugs 0.000 abstract description 4
- 229910052760 oxygen Inorganic materials 0.000 abstract description 4
- 239000001301 oxygen Substances 0.000 abstract description 4
- 230000001681 protective effect Effects 0.000 abstract description 4
- 230000037311 normal skin Effects 0.000 abstract description 3
- 230000008439 repair process Effects 0.000 abstract description 3
- 230000002500 effect on skin Effects 0.000 abstract description 2
- 239000012752 auxiliary agent Substances 0.000 abstract 1
- 241000699670 Mus sp. Species 0.000 description 16
- 210000003491 skin Anatomy 0.000 description 16
- 230000000052 comparative effect Effects 0.000 description 15
- 239000000243 solution Substances 0.000 description 12
- 238000002360 preparation method Methods 0.000 description 10
- 229930182478 glucoside Natural products 0.000 description 9
- 238000012360 testing method Methods 0.000 description 8
- 150000008131 glucosides Chemical class 0.000 description 7
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- 206010015150 Erythema Diseases 0.000 description 5
- 206010052428 Wound Diseases 0.000 description 5
- 208000027418 Wounds and injury Diseases 0.000 description 5
- 206010040882 skin lesion Diseases 0.000 description 5
- 231100000444 skin lesion Toxicity 0.000 description 5
- 230000015572 biosynthetic process Effects 0.000 description 4
- 201000010099 disease Diseases 0.000 description 4
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 4
- 231100000321 erythema Toxicity 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- 230000007774 longterm Effects 0.000 description 4
- 239000000203 mixture Substances 0.000 description 4
- -1 polyoxyethylene Polymers 0.000 description 4
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- 241000699666 Mus <mouse, genus> Species 0.000 description 3
- 208000010668 atopic eczema Diseases 0.000 description 3
- 238000011156 evaluation Methods 0.000 description 3
- 229960002751 imiquimod Drugs 0.000 description 3
- DOUYETYNHWVLEO-UHFFFAOYSA-N imiquimod Chemical compound C1=CC=CC2=C3N(CC(C)C)C=NC3=C(N)N=C21 DOUYETYNHWVLEO-UHFFFAOYSA-N 0.000 description 3
- 230000037380 skin damage Effects 0.000 description 3
- 238000003860 storage Methods 0.000 description 3
- 206010067484 Adverse reaction Diseases 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 201000004624 Dermatitis Diseases 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 230000006838 adverse reaction Effects 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 230000002354 daily effect Effects 0.000 description 2
- 230000006378 damage Effects 0.000 description 2
- 210000004207 dermis Anatomy 0.000 description 2
- 238000001704 evaporation Methods 0.000 description 2
- 230000008020 evaporation Effects 0.000 description 2
- 230000003203 everyday effect Effects 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 239000003862 glucocorticoid Substances 0.000 description 2
- 208000015181 infectious disease Diseases 0.000 description 2
- 230000010354 integration Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 239000013641 positive control Substances 0.000 description 2
- 230000001185 psoriatic effect Effects 0.000 description 2
- 238000001223 reverse osmosis Methods 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 2
- 238000013112 stability test Methods 0.000 description 2
- 230000008961 swelling Effects 0.000 description 2
- 229940037128 systemic glucocorticoids Drugs 0.000 description 2
- 230000008719 thickening Effects 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- AZUYLZMQTIKGSC-UHFFFAOYSA-N 1-[6-[4-(5-chloro-6-methyl-1H-indazol-4-yl)-5-methyl-3-(1-methylindazol-5-yl)pyrazol-1-yl]-2-azaspiro[3.3]heptan-2-yl]prop-2-en-1-one Chemical compound ClC=1C(=C2C=NNC2=CC=1C)C=1C(=NN(C=1C)C1CC2(CN(C2)C(C=C)=O)C1)C=1C=C2C=NN(C2=CC=1)C AZUYLZMQTIKGSC-UHFFFAOYSA-N 0.000 description 1
- OCKGFTQIICXDQW-ZEQRLZLVSA-N 5-[(1r)-1-hydroxy-2-[4-[(2r)-2-hydroxy-2-(4-methyl-1-oxo-3h-2-benzofuran-5-yl)ethyl]piperazin-1-yl]ethyl]-4-methyl-3h-2-benzofuran-1-one Chemical compound C1=C2C(=O)OCC2=C(C)C([C@@H](O)CN2CCN(CC2)C[C@H](O)C2=CC=C3C(=O)OCC3=C2C)=C1 OCKGFTQIICXDQW-ZEQRLZLVSA-N 0.000 description 1
- 241000205585 Aquilegia canadensis Species 0.000 description 1
- 241001061264 Astragalus Species 0.000 description 1
- 235000007866 Chamaemelum nobile Nutrition 0.000 description 1
- 235000000604 Chrysanthemum parthenium Nutrition 0.000 description 1
- 241000931705 Cicada Species 0.000 description 1
- 208000014311 Cushing syndrome Diseases 0.000 description 1
- 206010012434 Dermatitis allergic Diseases 0.000 description 1
- 206010012504 Dermatophytosis Diseases 0.000 description 1
- 206010059866 Drug resistance Diseases 0.000 description 1
- 241000233866 Fungi Species 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- 235000000177 Indigofera tinctoria Nutrition 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical group C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 1
- 240000007890 Leonurus cardiaca Species 0.000 description 1
- 235000000802 Leonurus cardiaca ssp. villosus Nutrition 0.000 description 1
- 244000042664 Matricaria chamomilla Species 0.000 description 1
- 235000007232 Matricaria chamomilla Nutrition 0.000 description 1
- 241001460074 Microsporum distortum Species 0.000 description 1
- 241001530038 Pantherophis obsoletus Species 0.000 description 1
- 239000004264 Petrolatum Substances 0.000 description 1
- 241000972672 Phellodendron Species 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 241000101040 Pityriasis Species 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 240000001341 Reynoutria japonica Species 0.000 description 1
- 235000018167 Reynoutria japonica Nutrition 0.000 description 1
- 244000299790 Rheum rhabarbarum Species 0.000 description 1
- 235000009411 Rheum rhabarbarum Nutrition 0.000 description 1
- 206010039793 Seborrhoeic dermatitis Diseases 0.000 description 1
- 206010040867 Skin hypertrophy Diseases 0.000 description 1
- 240000009022 Smilax rotundifolia Species 0.000 description 1
- 235000003205 Smilax rotundifolia Nutrition 0.000 description 1
- 208000002474 Tinea Diseases 0.000 description 1
- 206010043866 Tinea capitis Diseases 0.000 description 1
- 208000025865 Ulcer Diseases 0.000 description 1
- 208000024780 Urticaria Diseases 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 230000006978 adaptation Effects 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- SHGAZHPCJJPHSC-YCNIQYBTSA-N all-trans-retinoic acid Chemical compound OC(=O)\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-YCNIQYBTSA-N 0.000 description 1
- 208000026935 allergic disease Diseases 0.000 description 1
- 230000000172 allergic effect Effects 0.000 description 1
- 230000007815 allergy Effects 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000000843 anti-fungal effect Effects 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 229940121375 antifungal agent Drugs 0.000 description 1
- 235000006533 astragalus Nutrition 0.000 description 1
- 201000008937 atopic dermatitis Diseases 0.000 description 1
- 230000003385 bacteriostatic effect Effects 0.000 description 1
- 230000003115 biocidal effect Effects 0.000 description 1
- 239000010495 camellia oil Substances 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 210000002249 digestive system Anatomy 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 235000006694 eating habits Nutrition 0.000 description 1
- 210000000750 endocrine system Anatomy 0.000 description 1
- 230000001815 facial effect Effects 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 229960005150 glycerol Drugs 0.000 description 1
- LPLVUJXQOOQHMX-QWBHMCJMSA-N glycyrrhizinic acid Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@H](O[C@@H]1O[C@@H]1C([C@H]2[C@]([C@@H]3[C@@]([C@@]4(CC[C@@]5(C)CC[C@@](C)(C[C@H]5C4=CC3=O)C(O)=O)C)(C)CC2)(C)CC1)(C)C)C(O)=O)[C@@H]1O[C@H](C(O)=O)[C@@H](O)[C@H](O)[C@H]1O LPLVUJXQOOQHMX-QWBHMCJMSA-N 0.000 description 1
- 230000035876 healing Effects 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 238000005286 illumination Methods 0.000 description 1
- 229940097275 indigo Drugs 0.000 description 1
- COHYTHOBJLSHDF-UHFFFAOYSA-N indigo powder Natural products N1C2=CC=CC=C2C(=O)C1=C1C(=O)C2=CC=CC=C2N1 COHYTHOBJLSHDF-UHFFFAOYSA-N 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 230000009545 invasion Effects 0.000 description 1
- 238000005342 ion exchange Methods 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 239000002085 irritant Substances 0.000 description 1
- 231100000021 irritant Toxicity 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 238000005461 lubrication Methods 0.000 description 1
- 235000013372 meat Nutrition 0.000 description 1
- 229960000485 methotrexate Drugs 0.000 description 1
- 229940049016 methotrexate 2.5 mg Drugs 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 210000004400 mucous membrane Anatomy 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- 230000003071 parasitic effect Effects 0.000 description 1
- 235000019271 petrolatum Nutrition 0.000 description 1
- 229940066842 petrolatum Drugs 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 229940068918 polyethylene glycol 400 Drugs 0.000 description 1
- 238000004321 preservation Methods 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 201000009395 primary hyperaldosteronism Diseases 0.000 description 1
- 230000035755 proliferation Effects 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004405 propyl p-hydroxybenzoate Substances 0.000 description 1
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 229960004889 salicylic acid Drugs 0.000 description 1
- 208000008742 seborrheic dermatitis Diseases 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 210000004927 skin cell Anatomy 0.000 description 1
- 230000007480 spreading Effects 0.000 description 1
- 238000003892 spreading Methods 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 210000004233 talus Anatomy 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 229960001727 tretinoin Drugs 0.000 description 1
- 230000036269 ulceration Effects 0.000 description 1
- 238000012795 verification Methods 0.000 description 1
- 239000000341 volatile oil Substances 0.000 description 1
- 230000002747 voluntary effect Effects 0.000 description 1
- 230000029663 wound healing Effects 0.000 description 1
- 229910001656 zinc mineral Inorganic materials 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4425—Pyridinium derivatives, e.g. pralidoxime, pyridostigmine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/045—Hydroxy compounds, e.g. alcohols; Salts thereof, e.g. alcoholates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/045—Hydroxy compounds, e.g. alcohols; Salts thereof, e.g. alcoholates
- A61K31/05—Phenols
- A61K31/055—Phenols the aromatic ring being substituted by halogen
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K33/00—Medicinal preparations containing inorganic active ingredients
- A61K33/24—Heavy metals; Compounds thereof
- A61K33/30—Zinc; Compounds thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/32—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. carbomers, poly(meth)acrylates, or polyvinyl pyrrolidone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/70—Web, sheet or filament bases ; Films; Fibres of the matrix type containing drug
- A61K9/7007—Drug-containing films, membranes or sheets
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L26/00—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form
- A61L26/0004—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form containing inorganic materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L26/00—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form
- A61L26/0009—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form containing macromolecular materials
- A61L26/0014—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form containing macromolecular materials obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L26/00—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form
- A61L26/0061—Use of materials characterised by their function or physical properties
- A61L26/0066—Medicaments; Biocides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/04—Antipruritics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/10—Antimycotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/10—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices containing or releasing inorganic materials
- A61L2300/102—Metals or metal compounds, e.g. salts such as bicarbonates, carbonates, oxides, zeolites, silicates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/20—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices containing or releasing organic materials
- A61L2300/204—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices containing or releasing organic materials with nitrogen-containing functional groups, e.g. aminoxides, nitriles, guanidines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/20—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices containing or releasing organic materials
- A61L2300/216—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices containing or releasing organic materials with other specific functional groups, e.g. aldehydes, ketones, phenols, quaternary phosphonium groups
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/40—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices characterised by a specific therapeutic activity or mode of action
- A61L2300/402—Anaestetics, analgesics, e.g. lidocaine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/40—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices characterised by a specific therapeutic activity or mode of action
- A61L2300/404—Biocides, antimicrobial agents, antiseptic agents
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Materials Engineering (AREA)
- Dermatology (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Pain & Pain Management (AREA)
- Rheumatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
The invention belongs to the technical field of medicines, and particularly discloses a liquid dressing for treating psoriasis, which comprises zinc pyrithione, zinc oxide, zinc-containing mineral substances, menthol, ethanol, borneol, parachlorometaxylenol, a film-forming agent, a film-forming auxiliary agent and purified water containing selenium. The synergistic effect of the components can obviously enhance the treatment effect on skin diseases such as psoriasis and the like and reduce the irritation to the skin, has the effects of relieving itching and easing pain, can effectively relieve the symptoms of patients, simultaneously enhances the stability of a raw material system in a film forming mode, can isolate oxygen in the air, stops the growth and reproduction of pathogenic bacteria, forms a grid shape on the surface of the skin of the protective film, and is beneficial to normal skin growth and repair.
Description
Technical Field
The invention belongs to the technical field of medicines, and particularly relates to a liquid dressing for treating psoriasis.
Background
Today, some intractable skin diseases are still difficult to be eradicated completely in the case of the rapid development of modern medicine. The skin diseases are caused by a plurality of reasons, and the skin diseases are caused by daily bad living habits except family inheritance. Most patients belong to allergic constitution, and abnormal secretion of endocrine system and gastrointestinal digestive system of the patients may cause skin diseases with the increase of age. The disease probability can be increased in living environment with strong sunlight, and high humidity or dryness. Bad living habits can also cause skin diseases, such as frequent use of irritant skin care products, excessive use of facial cleanser and the like, which can damage skin cell tissues on the surface of a human body, cause the fragility of an epidermal tissue system and cannot resist the invasion of external pathogenic bacteria, thus causing a large amount of flora to be parasitic on the surface of the skin, and further inducing the generation of the skin diseases. The clinical symptoms of the disease are manifested by pruritus, patients can not endure the pruritus and buckle the skin, the cuticle of the skin is thinned, the ulceration infection of the focus area is easily caused, the focus area is further expanded, and the recurrence rate is very high.
At present, antifungal medicines, such as antibiotics, glucocorticoids, tretinoin and the like, are adopted to treat skin diseases such as tinea capitis, seborrheic dermatitis, pityriasis capitis and the like, but long-term use of the antibiotic medicines can easily cause drug resistance of human bodies, long-term use of the glucocorticoids can cause great harm to the human bodies due to Cushing's syndrome, and although a plurality of medical apparatus products for treating the skin diseases exist in the market, the treatment effects are different, the better treatment effect is difficult to achieve, and the problem of easy relapse exists after the skin diseases are cured, so that the invention of a treatment product with good treatment effect on the skin diseases and capable of effectively reducing the relapse rate is urgently needed.
Patent document CN105709273A discloses a liquid medical adjuvant containing polyethylene glycol, which aims to provide a multifunctional medical liquid dressing for promoting wound healing, and is technically characterized by containing polyethylene glycol 400, zinc pyrithione, glycerol, menthol and the balance of propylene glycol. The zinc pyrithione can effectively inhibit excessive proliferation of epidermoid epigastric cells, has a bacteriostatic action, can relieve inflammatory reaction at skin lesions, relieves pruritus and pain at the skin lesions, has a synergistic effect when being combined with polyethylene glycol and menthol, enhances the anti-inflammatory, itching relieving and pain relieving effects of the menthol, is used for superficial wounds (superficial dermis and wounds above the superficial dermis) of human body surfaces, can form a protective film on the wounds, protects the wounds, provides a healing environment for the wounds, has the effects of bacteriostasis, lubrication, moisture preservation, dry crack prevention, cooling, itching relieving and infection inhibition, but has certain irritation on skin and the problem of easy relapse.
Patent document CN105412731A discloses an ointment for treating cutaneous pruritus, which is prepared from the following raw materials: the traditional Chinese medicine composition is prepared from phellodendron, glabrous greenbrier rhizome, black snake meat, manyleaf paris rhizome, cicada slough, raw fleece-flower root, raw rhubarb, rhizoma atractylodis, natural indigo, honeysuckle, cortex dictamni, motherwort herb, astragalus, camellia oil, chamomile essential oil, propyl p-hydroxybenzoate, polyoxyethylene fatty acid ester, zinc pyrithione, ammonium glycyrrhizinate, diethanolamide and salicylic acid, can better treat skin pruritus caused by allergic dermatitis, eczema, urticaria, dermatophytosis and the like, has obvious curative effect and quick response, can treat both symptoms and root causes, has no toxic or side effect through clinical verification, is simple and easy to operate in preparation process, easily available in raw materials, does not have adverse reaction, and has the problem of incomplete treatment and easy recurrence.
Through search, the composition containing zinc pyrithione, zinc oxide and mineral substances and the application of the composition in medical liquid dressing for treating psoriasis are not discovered at present.
Disclosure of Invention
The invention aims to provide a liquid dressing for treating psoriasis, which can effectively treat the psoriasis, has better stability, has no irritation to skin and can effectively reduce the recurrence rate of skin diseases such as the psoriasis and the like.
In order to achieve the purpose, the invention adopts the following technical scheme: a liquid dressing for treating psoriasis comprises the following raw materials in parts by weight: 1.0-5.0 parts of zinc pyrithione, 1.0-3.0 parts of zinc oxide, 1.0-6.0 parts of zinc-containing mineral substance, 0.1-1.0 part of menthol, 1.0-3.0 parts of ethanol, 0.1-1.0 part of borneol, 0.5-2.0 parts of parachlorometaxylenol, 10.0-20.0 parts of film-forming agent, 1.0-5.0 parts of film-forming assistant and 54.0-84.3 parts of purified water containing selenium
Preferably, the feed comprises the following raw materials in parts by weight: 3.0 parts of zinc pyrithione, 6.0 parts of zinc oxide, 1.5 parts of zinc-containing mineral substances, 0.5 part of menthol, 2.0 parts of ethanol, 0.5 part of borneol, 1.0 part of parachlorometaxylenol, 15.0 parts of film-forming agent, 3.0 parts of film-forming assistant and 67.0 parts of purified water containing selenium.
Preferably, the zinc-containing minerals include zinc carbonate, zinc sulfate, zinc oxide, and zinc chloride.
Preferably, the film former is prepared from polyvinyl alcohol and polyvinylpyrrolidone in a ratio of 1: 2 in mass ratio.
Preferably, the coalescing agent is arachidyl glucoside.
Preferably, the weight part ratio of the zinc pyrithione to the zinc-containing mineral is 1 (2-4).
In addition, a preparation method of the liquid dressing for treating psoriasis is also disclosed, and comprises the following steps:
s1, weighing polyvinylpyrrolidone with a formula amount and selenium-containing water with a formula amount of 1/5, purifying, mixing, putting into a water bath heating pot, heating in a water bath, and fully dissolving until no granular sensation exists, thus obtaining polyvinylpyrrolidone solution;
s2, placing the rest selenium-containing purified water in a stirring pot, heating, adding zinc pyrithione, zinc oxide, zinc-containing mineral substances, polyvinyl alcohol, arachidyl glucoside, menthol, ethanol, borneol and parachlorometaxylenol in the formula amount, and stirring fully for later use;
s3, transferring the polyvinylpyrrolidone solution prepared in the step S1 to the stirrer in the step S2, fully stirring, and filling the finally obtained solution to obtain the required liquid dressing.
Preferably, the selenium-containing purified water in the step S2 is heated to 40-60 ℃.
Compared with the prior art, the invention has the following beneficial effects:
(1) through the synergistic effect of the zinc pyrithione and the traditional Chinese medicine composition, the invention can obviously enhance the effects of inhibiting the growth of fungi, relieving itching and easing pain of the product, effectively reduce the irritation of the raw material components to the skin and simultaneously effectively reduce the recurrence rate of skin diseases such as psoriasis and the like.
(2) The invention enhances the stability of the raw material system in the form of film formation, can isolate oxygen in the air, can stop the oxygen for the growth and the propagation of germs, and can form a grid shape on the surface of the skin by the protective film, thereby being beneficial to the growth and the repair of normal skin.
Detailed Description
The present invention will be further described below by way of specific embodiments, but the present invention is not limited to only the following examples. It will be apparent to those skilled in the art that the invention can be modified and replaced with other components having the same effects without departing from the spirit and scope of the invention, and all such modifications and substitutions are deemed to be within the scope of the invention.
The raw materials used in the invention are all conventional commercial products.
In the embodiment of the invention, the preparation method of the selenium-containing purified water comprises the following steps:
taking raw water suitable for spring warm water, firstly pressurizing the raw water by a raw water booster pump, then sequentially filtering the raw water by a multi-medium filter and an activated carbon filter for multiple times, carrying out ion exchange on the filtered purified water by an ion exchanger, then filtering, carrying out reverse osmosis and distillation by a security filter, a multi-stage high-pressure pump and an RO reverse osmosis system to complete further purification of the purified water, and finally sterilizing and filling the purified water to obtain the selenium-containing purified water required by the invention.
Example 1A liquid dressing for the treatment of psoriasis
The formula is as follows: 100g of zinc pyrithione, 100g of zinc oxide, 300g of zinc-containing mineral, 10g of menthol, 100g of ethanol, 10g of borneol, 50g of parachlorometaxylenol, 800g of polyvinylpyrrolidone, 400g of polyvinyl alcohol, 100g of arachidonol glucoside and 8030g of purified water containing selenium.
The preparation method comprises the following steps:
s1, weighing polyvinylpyrrolidone with a formula amount and selenium-containing water with a formula amount of 1/5, purifying, mixing, putting into a water bath heating pot, heating in a water bath, and fully dissolving until no granular sensation exists, thus obtaining polyvinylpyrrolidone solution;
s2, placing the residual selenium-containing purified water in a stirring pot, heating to 40 ℃, adding zinc pyrithione, zinc oxide, zinc-containing mineral substances, polyvinyl alcohol, arachidonol glucoside, menthol, ethanol, borneol and parachlorometaxylenol in the formula, and fully stirring for later use;
s3, transferring the polyvinylpyrrolidone solution prepared in the step S1 to the stirrer in the step S2, fully stirring, and filling the finally obtained solution to obtain the required liquid dressing.
Example 2A liquid dressing for the treatment of psoriasis
The formula is as follows: 300g of zinc pyrithione, 200g of zinc oxide, 600g of zinc-containing mineral, 50g of menthol, 200g of ethanol, 50g of borneol, 100g of parachlorometaxylenol, 1000g of polyvinylpyrrolidone, 500g of polyvinyl alcohol, 300g of arachidonol glucoside and 6700g of purified water containing selenium.
The preparation method comprises the following steps:
s1, weighing polyvinylpyrrolidone with a formula amount and selenium-containing water with a formula amount of 1/5, purifying, mixing, putting into a water bath heating pot, heating in a water bath, and fully dissolving until no granular sensation exists, thus obtaining polyvinylpyrrolidone solution;
s2, placing the rest selenium-containing purified water in a stirring pot, heating to 50 ℃, adding zinc pyrithione, zinc oxide, zinc-containing mineral, polyvinyl alcohol, arachidonol glucoside, menthol, ethanol, borneol and parachlorometaxylenol in the formula, and fully stirring for later use;
s3, transferring the polyvinylpyrrolidone solution prepared in the step S1 to the stirrer in the step S2, fully stirring, and filling the finally obtained solution to obtain the required liquid dressing.
Example 3A liquid dressing for the treatment of psoriasis
The formula is as follows: 300g of zinc pyrithione, 300g of zinc oxide, 600g of zinc-containing mineral, 100g of menthol, 300g of ethanol, 100g of borneol, 200g of parachlorometaxylenol, 1200g of polyvinylpyrrolidone, 600g of polyvinyl alcohol, 500g of arachidonol glucoside and 5800g of purified water containing selenium.
The preparation method comprises the following steps:
s1, weighing polyvinylpyrrolidone with a formula amount and selenium-containing water with a formula amount of 1/5, purifying, mixing, putting into a water bath heating pot, heating in a water bath, and fully dissolving until no granular sensation exists, thus obtaining polyvinylpyrrolidone solution;
s2, placing the residual selenium-containing purified water in a stirring pot, heating to 60 ℃, adding zinc pyrithione, zinc oxide, zinc-containing mineral substances, polyvinyl alcohol, arachidonol glucoside, menthol, ethanol, borneol and parachlorometaxylenol in the formula, and fully stirring for later use;
s3, transferring the polyvinylpyrrolidone solution prepared in the step S1 to the stirrer in the step S2, fully stirring, and filling the finally obtained solution to obtain the required liquid dressing.
Comparative example 1a liquid dressing for the treatment of psoriasis
The comparative example differs from example 2 only in that: no zinc pyrithione is contained.
The preparation method refers to example 2.
Comparative example 2 a liquid dressing for the treatment of psoriasis
The comparative example differs from example 2 only in that: contains no zinc-containing minerals.
The preparation method refers to example 2.
Comparative example 3a liquid dressing for the treatment of psoriasis
The comparative example differs from example 2 only in that: contains no arachidyl glucoside.
The preparation method refers to example 2.
Comparative example 4 a liquid dressing for the treatment of psoriasis
The comparative example differs from example 2 only in that: propylene glycol was used in place of arachidonol glucoside.
The preparation method refers to example 2.
Test example one, psoriasis treatment effect test of liquid dressing
Firstly, experimental samples: liquid dressings were prepared in examples 1 to 3 and comparative examples 1 to 2.
Second, Experimental methods
2.1 establishment of psoriasis model in mice
The mice are raised in a clean environment, the temperature is 22 +/-2 ℃, the relative humidity is 40-60%, and the illumination time is controlled to be 14 h. The experiment was started after 1 week of adaptation. 104 mice were randomly divided into a normal control group, a model control group, and a methotrexate group (positive control group), examples 1 to 3, and comparative examples 1 to 2, and 13 mice were each group. The back hair of each group of mice is removed by 4cm2The normal control group was topically applied with petrolatum only, and the remaining groups of mice were applied with imiquimod cream 10mg each time, 2 times daily for 14 days continuously. At the same time of molding, the experimental group is respectively smeared with the liquid dressings prepared in examples 1-3 and comparative examples 1-2, 1.0ml of the liquid dressings is used each time, methotrexate 2.5mg/kg is given to a positive control group, and the mice of a normal control group and a model control group are smeared with physiological saline with the same volume for 1 time every day and are continuously administrated for 14 days.
2.2 evaluation of mouse skin lesion Change on psoriasis-like skin lesion area and disease severity (PASI) score
The skin damage condition of the mice is observed every day and recorded by a camera, and the erythema, phosphorus chips and infiltration thickening degree of the skin damage of the mice are given 0-4 points according to the PASI scoring standard, wherein the standard is as follows. 0: none; 1: mild; 2, moderate; 3: (ii) severe; 4: the mouse is extremely severe, the integration of the three is added to obtain a total score, and the integration of each group of mice is averaged.
Third, experimental results
The experimental results are shown in tables 1 to 4.
After the imiquimod is smeared for 7 days, obvious erythema appears on the skin of each group of mice, the skin is continuously thickened and resembles psoriasis-like skin lesions, and the psoriasis symptoms are aggravated after the imiquimod is smeared for 14 days.
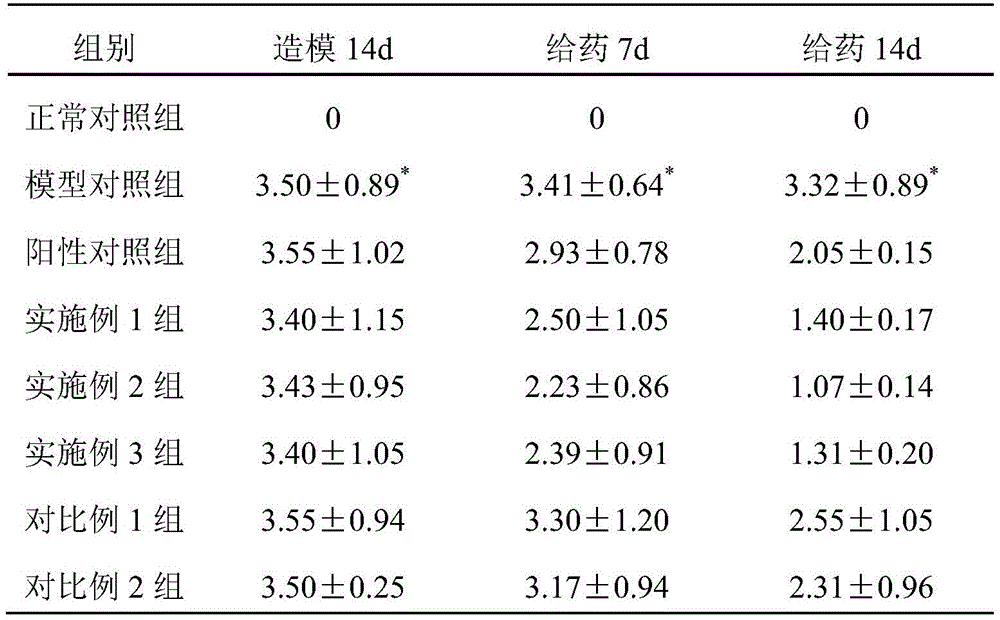
TABLE 1 Effect of liquid dressings on skin hypertrophy Scoring of psoriasis mice
Note that P <0.01 in comparison with normal control group
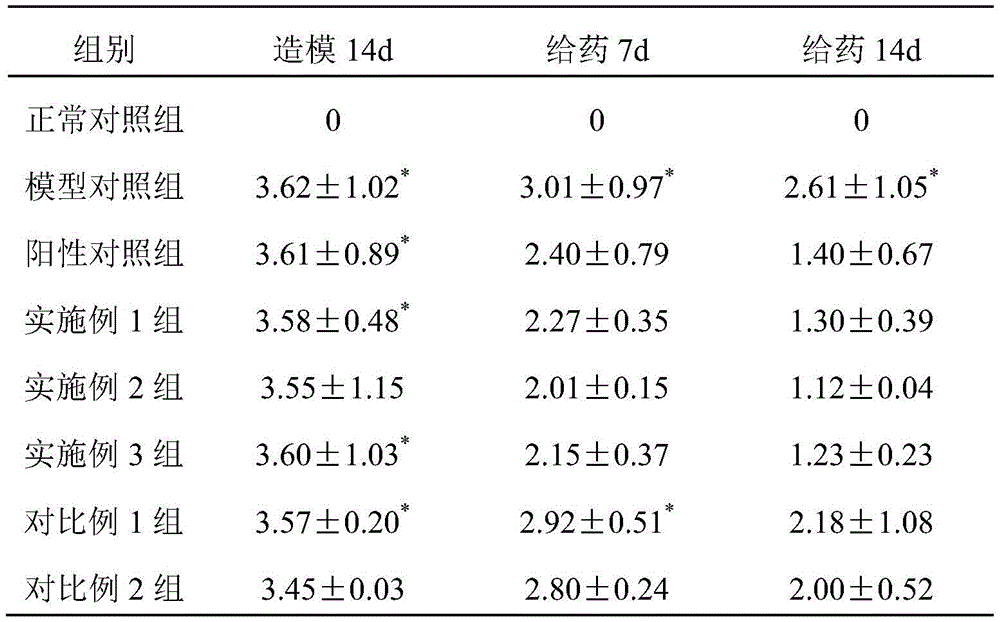
TABLE 2 Effect of liquid dressings on skin Scale Scoring in psoriatic mice
Note that P <0.01 in comparison with normal control group
TABLE 3 Effect of liquid dressings on skin erythema score in psoriasis mice
Note that P <0.01 in comparison with normal control group
TABLE 4 Total score results for area of psoriatic lesions and disease severity in the backs of mice
Note that P <0.0 in comparison with the normal control group
From the data in tables 1 to 4, it can be known that the mice in the groups of examples 1 to 3 of the present invention have lighter erythema, less scales, and slight thickening and swelling degree; the data of the embodiments 1-3 groups and the model group can be used for obtaining that the liquid dressing prepared by the invention can remarkably relieve the corresponding symptoms of the psoriasis mouse; the data of the groups of examples 1-3 and the groups of comparative examples 1-2 show that the synergistic effect between the zinc pyrithione and the zinc-containing mineral matter can effectively treat psoriasis, and can reduce the irritation of the raw material components such as the zinc pyrithione to the skin.
Test example II Long-term storage stability test
Firstly, experimental samples: the liquid dressings prepared in examples 1 to 3 and comparative examples 3 to 4.
Second, Experimental methods
The liquid dressings prepared in examples 1-3 and comparative examples 3-4 were stored at room temperature of 25 ℃. + -. 5 ℃ for 12 months, and were taken out at 0, 3, 6, 9, and 12 months, and the spreading condition of the sample and the filming condition of the sample after 12 months of storage were observed.
The scoring criteria are shown in table 5.
TABLE 5 liquid dressing spreadability Scoring standards
Third, experimental results
The experimental results are shown in tables 6 to 7.
TABLE 6 Long-term storage stability test results
TABLE 7 results of film formation at month 12 of the samples
As can be seen from table 6, the liquid dressings prepared in examples 1 to 3 of the present invention are easy to apply when used, and still have good applicability after being left for 3 months, 6 months, 9 months, and 12 months, and the liquid dressings prepared in examples 1 to 3 have gradually deteriorated applicability as the leaving time is prolonged, thereby proving that the addition of arachidonoyl glucoside of the present invention can significantly improve the applicability of the liquid dressings, and enhance the stability of the liquid dressings.
As can be seen from table 7, the liquid dressings prepared in examples 1 to 3 of the present invention still had a rapid film formation after moisture evaporation and maintained good stability after being left for 12 months, while the liquid dressings prepared in comparative examples 3 to 4 lacked arachidonoyl glucoside, and therefore, after being left for 12 months, the samples agglomerated and accumulated after moisture evaporation, and a film was hardly formed.
The experimental results show that the liquid dressing prepared in the embodiments 1 to 3 of the present invention can be uniformly applied, and the faster film forming property is probably because the hydroxyl groups in the structures of the polyvinyl alcohol and the arachidyl glucoside can form hydrogen bonds with the carbonyl group of the polyvinylpyrrolidone, so that the stability of the formed reticular film structure is enhanced, and the treatment effect of the liquid dressing on psoriasis is further ensured.
Test example three, evaluation of therapeutic effect of liquid dressing and evaluation of psoriasis recurrence suppressing effect
Firstly, experimental samples: liquid dressing of examples 1 to 3
Second, the tested population
Skin condition: the psoriasis patients are healthy, only have obvious erythema, have no history of skin allergy, and meet the voluntary selection standard of the subjects.
Grouping: divided into 3 groups of 50, 23 women and 27 men.
The test method comprises the following steps: the medicine is uniformly coated on the skin damage area 1 time per day, the phenomenon that eyes and other mucous membrane parts are contacted with the medicine is strictly avoided, the trial is carried out for 1 month, and the testee keeps good living and rest time and eating habits during the trial period. The feedback condition of the subjects was counted after 1 month, and the subjects were followed up half a year after the experiment, and the psoriasis recurrence condition of the subjects of examples 1-3 was counted.
Third, experimental results
The experimental results are shown in tables 8 to 10.
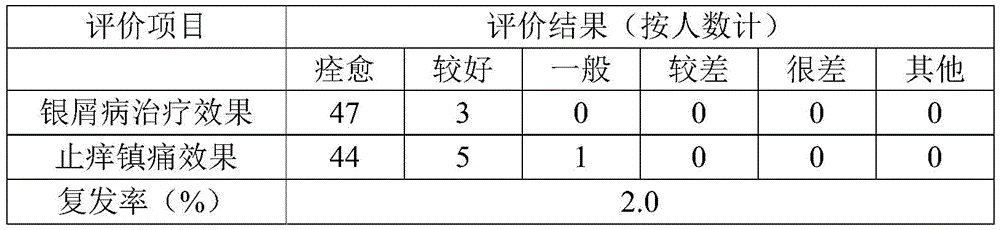
Table 8 test results for the liquid dressing of example 1
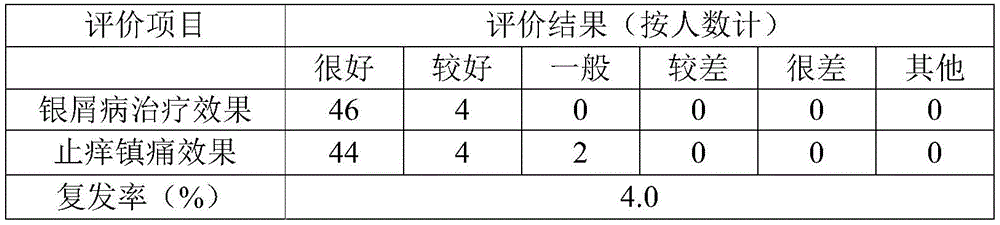
Table 9 test results for liquid dressing of example 2
Table 10 test results for example 3 liquid dressing
From tables 8 to 10, the liquid dressings of examples 1 to 3 of the present invention were approved by most subjects, had a good effect of treating psoriasis, and were effective in relieving itching and pain, and among them, the liquid dressing of example 1 had the highest approval.
From the data of psoriasis recurrence in the subject after the follow-up of the table for half a year, it can be known that the liquid dressing of the present invention has a low psoriasis recurrence rate, i.e. the present invention can effectively inhibit the recurrence after the psoriasis is cured. All subjects do not have adverse reactions such as allergy, red swelling and the like in the test of various projects.
The synergistic effect of the zinc pyrithione and the zinc-containing mineral substance can obviously enhance the treatment effect on skin diseases such as psoriasis and the like, has the effect of relieving itching and easing pain and can effectively relieve the pain of patients, and simultaneously, the zinc-containing mineral substance can effectively reduce the irritation of other raw material components to the skin. In addition, the synergistic effect between the film forming assistant and the film forming agent enhances the formation of the film, enhances the stability of a raw material system, can isolate oxygen in the air, stops the growth and reproduction of pathogenic bacteria, forms a grid shape on the surface of the skin of the protective film, and is beneficial to normal skin growth and repair.
The foregoing embodiments are merely illustrative of the principles and utilities of the present invention and are not intended to limit the invention. Any person skilled in the art can modify or change the above-mentioned embodiments without departing from the spirit and scope of the present invention. Accordingly, it is intended that all equivalent modifications or changes which can be made by those skilled in the art without departing from the spirit and technical spirit of the present invention be covered by the claims of the present invention.
Claims (8)
1. The liquid dressing for treating psoriasis is characterized by comprising the following raw materials in parts by weight: 1.0-5.0 parts of zinc pyrithione, 1.0-3.0 parts of zinc oxide, 1.0-6.0 parts of zinc-containing mineral substances, 0.1-1.0 part of menthol, 1.0-3.0 parts of ethanol, 0.1-1.0 part of borneol, 0.5-2.0 parts of parachlorometaxylenol, 10.0-20.0 parts of film-forming agent, 1.0-5.0 parts of film-forming assistant and 54.0-84.3 parts of purified water containing selenium.
2. The liquid dressing of claim 1, comprising the following raw materials in parts by weight: 3.0 parts of zinc pyrithione, 2.0 parts of zinc oxide, 6.0 parts of zinc-containing mineral substances, 0.5 part of menthol, 2.0 parts of ethanol, 0.5 part of borneol, 1.0 part of parachlorometaxylenol, 15.0 parts of film-forming agent, 3.0 parts of film-forming assistant and 67.0 parts of purified water containing selenium.
3. The fluid dressing of claim 1, wherein said zinc-containing mineral comprises zinc carbonate, zinc sulfate, zinc oxide, and zinc chloride.
4. The liquid dressing of claim 1, wherein said film former is formed from polyvinyl alcohol and polyvinyl pyrrolidone in a ratio of 1: 2 in mass ratio.
5. The liquid dressing of claim 1, wherein said film-forming aid is arachidyl glucoside.
6. The liquid dressing of claim 1, wherein the weight part ratio of the zinc pyrithione to the zinc-containing mineral is 1 (2-4).
7. A method of preparing a liquid dressing for use in the treatment of psoriasis as claimed in any of claims 1 to 6 comprising the steps of:
s1, weighing polyvinylpyrrolidone with a formula amount and selenium-containing water with a formula amount of 1/5, purifying, mixing, putting into a water bath heating pot, heating in a water bath, and fully dissolving until no granular sensation exists, thus obtaining polyvinylpyrrolidone solution;
s2, placing the rest selenium-containing purified water in a stirring pot, heating, adding zinc pyrithione, zinc oxide, zinc-containing mineral substances, polyvinyl alcohol, arachidyl glucoside, menthol, ethanol, borneol and parachlorometaxylenol in the formula amount, and stirring fully for later use;
s3, transferring the polyvinylpyrrolidone solution prepared in the step S1 to the stirrer in the step S2, fully stirring, and filling the finally obtained solution to obtain the required liquid dressing.
8. The method of preparing a liquid dressing for treating psoriasis as claimed in claim 7 wherein the purified water containing selenium of step S2 is heated to 40-60 ℃.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110662447.5A CN113368132A (en) | 2021-06-15 | 2021-06-15 | Liquid dressing for treating psoriasis |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110662447.5A CN113368132A (en) | 2021-06-15 | 2021-06-15 | Liquid dressing for treating psoriasis |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN113368132A true CN113368132A (en) | 2021-09-10 |
Family
ID=77574462
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110662447.5A Withdrawn CN113368132A (en) | 2021-06-15 | 2021-06-15 | Liquid dressing for treating psoriasis |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113368132A (en) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4210633A (en) * | 1978-10-20 | 1980-07-01 | Eli Lilly And Company | Flurandrenolide film formulation |
| US5747064A (en) * | 1993-07-28 | 1998-05-05 | Pfizer Inc. | Psoriasis treatment |
| US6248370B1 (en) * | 1997-07-24 | 2001-06-19 | Leroy Harris | Skin treatment and methods |
| US20180147213A1 (en) * | 2015-05-06 | 2018-05-31 | The Procter & Gamble Company | Methods of cosmetically treating skin conditions with a cosmetic personal cleansing composition |
-
2021
- 2021-06-15 CN CN202110662447.5A patent/CN113368132A/en not_active Withdrawn
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4210633A (en) * | 1978-10-20 | 1980-07-01 | Eli Lilly And Company | Flurandrenolide film formulation |
| US5747064A (en) * | 1993-07-28 | 1998-05-05 | Pfizer Inc. | Psoriasis treatment |
| US6248370B1 (en) * | 1997-07-24 | 2001-06-19 | Leroy Harris | Skin treatment and methods |
| US20180147213A1 (en) * | 2015-05-06 | 2018-05-31 | The Procter & Gamble Company | Methods of cosmetically treating skin conditions with a cosmetic personal cleansing composition |
Non-Patent Citations (3)
| Title |
|---|
| CHARLES E. CRUTCHFIELD III,等: "The highly effective use of topical zinc pyrithione in the treatment of psoriasis: a case report", 《DERMATOLOGY ONLINE JOURNAL》 * |
| 杨志波: "《中成药临床应用指南 皮肤病分册》", 31 January 2017, 中国中医药出版社 * |
| 钟静芬,等: "《表面活性剂在药学中的应用》", 29 February 1996, 人民卫生出版社 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN109044915B (en) | Infant hip-protecting cream and preparation process thereof | |
| CN104906203A (en) | Tinea manus and pedis treatment painless nano emulsifiable paste | |
| WO2002030467A2 (en) | Pharmaceutical composition containing honey for the treatment of wounds | |
| WO2017080048A1 (en) | Film spraying agent for superfical burns and preparation method therefor | |
| CN110638727B (en) | Infant hip-protecting composition and preparation method and application thereof | |
| CN110859762B (en) | Gel containing Camellia japonica extract and having after-sun repair effect and preparation method thereof | |
| KR100894439B1 (en) | Cosmetic composition having a prophylactic effect of atopic dermatitis and method for producing same | |
| CN105997785B (en) | A kind of stern protection cream for baby and preparation method thereof | |
| CN106619823A (en) | Ointment for therapy of eczema and preparation method of same | |
| CN113995775B (en) | A kind of probiotic foot mask with foot protection effect and preparation method thereof | |
| CN102283899A (en) | Traditional Chinese medicine external preparation and application thereof | |
| CN105168361A (en) | Compound plant oil liniment for treating burns and scalds and preparation method of compound plant oil liniment | |
| CN113368132A (en) | Liquid dressing for treating psoriasis | |
| CN100455311C (en) | Composition of Chinese traditional medicine in use for treating burn and scald, and product for resolving poxes, and preparation method | |
| HK1045813A1 (en) | Non-solid composition for local application comprising glycerol and alchemilla vulgaris extract | |
| CN101332286B (en) | Traditional Chinese medicine composition for treating skin disease and preparation method thereof | |
| CN114984132B (en) | Wound spray capable of promoting coagulation and repair and preparation method thereof | |
| CN111631991A (en) | Private nursing lotion | |
| CN105012992B (en) | A kind of medical bio vagina hydrogel functional dressing and preparation method thereof | |
| CN116549489A (en) | Antibacterial repair debridement gel and preparation method thereof | |
| CN109091596A (en) | A kind of compound nursing gel for women and preparation method thereof | |
| CN107158051A (en) | Tannic acid ointment for treating eczema and preparation method thereof | |
| CN117045717B (en) | External compound essential oil composition for treating eczema and preparation method thereof | |
| CN116211781B (en) | Composition for preventing and treating chapped skin, and preparation method and application thereof | |
| US20240415767A1 (en) | Tattoo Aftercare Sealant Gel |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| WW01 | Invention patent application withdrawn after publication |
Application publication date: 20210910 |
|
| WW01 | Invention patent application withdrawn after publication |