CN113350277A - Mesalazine gargle for treating chemotherapy oral mucositis and preparation method thereof - Google Patents
Mesalazine gargle for treating chemotherapy oral mucositis and preparation method thereof Download PDFInfo
- Publication number
- CN113350277A CN113350277A CN202110731248.5A CN202110731248A CN113350277A CN 113350277 A CN113350277 A CN 113350277A CN 202110731248 A CN202110731248 A CN 202110731248A CN 113350277 A CN113350277 A CN 113350277A
- Authority
- CN
- China
- Prior art keywords
- mesalazine
- oral mucositis
- gargle
- treatment
- mouthrinse
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- KBOPZPXVLCULAV-UHFFFAOYSA-N mesalamine Chemical compound NC1=CC=C(O)C(C(O)=O)=C1 KBOPZPXVLCULAV-UHFFFAOYSA-N 0.000 title claims abstract description 88
- 229960004963 mesalazine Drugs 0.000 title claims abstract description 88
- 208000003265 stomatitis Diseases 0.000 title claims abstract description 32
- 238000002512 chemotherapy Methods 0.000 title claims abstract description 31
- 238000002360 preparation method Methods 0.000 title claims abstract description 18
- 239000004094 surface-active agent Substances 0.000 claims abstract description 15
- 239000000080 wetting agent Substances 0.000 claims abstract description 15
- 239000000796 flavoring agent Substances 0.000 claims abstract description 8
- 235000013355 food flavoring agent Nutrition 0.000 claims abstract description 8
- 239000007864 aqueous solution Substances 0.000 claims abstract description 5
- 239000002245 particle Substances 0.000 claims description 25
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 claims description 16
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 14
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 12
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 claims description 9
- 239000002324 mouth wash Substances 0.000 claims description 9
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 8
- 150000001875 compounds Chemical class 0.000 claims description 7
- 238000003756 stirring Methods 0.000 claims description 7
- 235000011187 glycerol Nutrition 0.000 claims description 6
- 238000000034 method Methods 0.000 claims description 6
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 claims description 6
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 claims description 6
- 229940068968 polysorbate 80 Drugs 0.000 claims description 6
- 229920000053 polysorbate 80 Polymers 0.000 claims description 6
- 239000008213 purified water Substances 0.000 claims description 6
- 238000004090 dissolution Methods 0.000 claims description 4
- FTLYMKDSHNWQKD-UHFFFAOYSA-N (2,4,5-trichlorophenyl)boronic acid Chemical compound OB(O)C1=CC(Cl)=C(Cl)C=C1Cl FTLYMKDSHNWQKD-UHFFFAOYSA-N 0.000 claims description 3
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 claims description 3
- UDIPTWFVPPPURJ-UHFFFAOYSA-M Cyclamate Chemical compound [Na+].[O-]S(=O)(=O)NC1CCCCC1 UDIPTWFVPPPURJ-UHFFFAOYSA-M 0.000 claims description 3
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 claims description 3
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 claims description 3
- 229920003171 Poly (ethylene oxide) Polymers 0.000 claims description 3
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 claims description 3
- UEDUENGHJMELGK-HYDKPPNVSA-N Stevioside Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(=O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O UEDUENGHJMELGK-HYDKPPNVSA-N 0.000 claims description 3
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 claims description 3
- 229930006000 Sucrose Natural products 0.000 claims description 3
- 239000004359 castor oil Substances 0.000 claims description 3
- 235000019438 castor oil Nutrition 0.000 claims description 3
- 239000000625 cyclamic acid and its Na and Ca salt Substances 0.000 claims description 3
- 238000001914 filtration Methods 0.000 claims description 3
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 claims description 3
- 239000000787 lecithin Substances 0.000 claims description 3
- 235000010445 lecithin Nutrition 0.000 claims description 3
- 229940067606 lecithin Drugs 0.000 claims description 3
- 239000012528 membrane Substances 0.000 claims description 3
- 229910000403 monosodium phosphate Inorganic materials 0.000 claims description 3
- 235000019799 monosodium phosphate Nutrition 0.000 claims description 3
- NOOLISFMXDJSKH-UHFFFAOYSA-N p-menthan-3-ol Chemical compound CC(C)C1CCC(C)CC1O NOOLISFMXDJSKH-UHFFFAOYSA-N 0.000 claims description 3
- 229940068918 polyethylene glycol 400 Drugs 0.000 claims description 3
- -1 polyoxyethylene Polymers 0.000 claims description 3
- 229940085605 saccharin sodium Drugs 0.000 claims description 3
- 239000001509 sodium citrate Substances 0.000 claims description 3
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 claims description 3
- 235000011083 sodium citrates Nutrition 0.000 claims description 3
- 229960001462 sodium cyclamate Drugs 0.000 claims description 3
- AJPJDKMHJJGVTQ-UHFFFAOYSA-M sodium dihydrogen phosphate Chemical compound [Na+].OP(O)([O-])=O AJPJDKMHJJGVTQ-UHFFFAOYSA-M 0.000 claims description 3
- 235000011121 sodium hydroxide Nutrition 0.000 claims description 3
- 239000000600 sorbitol Substances 0.000 claims description 3
- 229940013618 stevioside Drugs 0.000 claims description 3
- OHHNJQXIOPOJSC-UHFFFAOYSA-N stevioside Natural products CC1(CCCC2(C)C3(C)CCC4(CC3(CCC12C)CC4=C)OC5OC(CO)C(O)C(O)C5OC6OC(CO)C(O)C(O)C6O)C(=O)OC7OC(CO)C(O)C(O)C7O OHHNJQXIOPOJSC-UHFFFAOYSA-N 0.000 claims description 3
- 235000019202 steviosides Nutrition 0.000 claims description 3
- 239000005720 sucrose Substances 0.000 claims description 3
- 229960004793 sucrose Drugs 0.000 claims description 3
- 238000013329 compounding Methods 0.000 claims description 2
- 238000007781 pre-processing Methods 0.000 claims description 2
- 239000003814 drug Substances 0.000 abstract description 24
- 230000000694 effects Effects 0.000 abstract description 15
- 229940079593 drug Drugs 0.000 abstract description 13
- 238000012360 testing method Methods 0.000 description 24
- 239000007788 liquid Substances 0.000 description 7
- 102000003815 Interleukin-11 Human genes 0.000 description 5
- 108090000177 Interleukin-11 Proteins 0.000 description 5
- 229940074383 interleukin-11 Drugs 0.000 description 5
- 239000002994 raw material Substances 0.000 description 5
- 239000011715 vitamin B12 Substances 0.000 description 5
- 229930003779 Vitamin B12 Natural products 0.000 description 4
- FDJOLVPMNUYSCM-WZHZPDAFSA-L cobalt(3+);[(2r,3s,4r,5s)-5-(5,6-dimethylbenzimidazol-1-yl)-4-hydroxy-2-(hydroxymethyl)oxolan-3-yl] [(2r)-1-[3-[(1r,2r,3r,4z,7s,9z,12s,13s,14z,17s,18s,19r)-2,13,18-tris(2-amino-2-oxoethyl)-7,12,17-tris(3-amino-3-oxopropyl)-3,5,8,8,13,15,18,19-octamethyl-2 Chemical compound [Co+3].N#[C-].N([C@@H]([C@]1(C)[N-]\C([C@H]([C@@]1(CC(N)=O)C)CCC(N)=O)=C(\C)/C1=N/C([C@H]([C@@]1(CC(N)=O)C)CCC(N)=O)=C\C1=N\C([C@H](C1(C)C)CCC(N)=O)=C/1C)[C@@H]2CC(N)=O)=C\1[C@]2(C)CCC(=O)NC[C@@H](C)OP([O-])(=O)O[C@H]1[C@@H](O)[C@@H](N2C3=CC(C)=C(C)C=C3N=C2)O[C@@H]1CO FDJOLVPMNUYSCM-WZHZPDAFSA-L 0.000 description 4
- 210000000214 mouth Anatomy 0.000 description 4
- 239000000047 product Substances 0.000 description 4
- 235000019163 vitamin B12 Nutrition 0.000 description 4
- 238000001514 detection method Methods 0.000 description 3
- 238000011068 loading method Methods 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 210000004400 mucous membrane Anatomy 0.000 description 3
- 238000005070 sampling Methods 0.000 description 3
- 238000013112 stability test Methods 0.000 description 3
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- 208000025865 Ulcer Diseases 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 239000012467 final product Substances 0.000 description 2
- 238000011010 flushing procedure Methods 0.000 description 2
- 238000005286 illumination Methods 0.000 description 2
- 208000015181 infectious disease Diseases 0.000 description 2
- 230000002757 inflammatory effect Effects 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 239000000203 mixture Substances 0.000 description 2
- 238000004806 packaging method and process Methods 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 238000001959 radiotherapy Methods 0.000 description 2
- 230000001954 sterilising effect Effects 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 230000000699 topical effect Effects 0.000 description 2
- NOOLISFMXDJSKH-UTLUCORTSA-N (+)-Neomenthol Chemical compound CC(C)[C@@H]1CC[C@@H](C)C[C@@H]1O NOOLISFMXDJSKH-UTLUCORTSA-N 0.000 description 1
- 208000003322 Coinfection Diseases 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 241000167880 Hirundinidae Species 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 206010028116 Mucosal inflammation Diseases 0.000 description 1
- 201000010927 Mucositis Diseases 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 206010030973 Oral discomfort Diseases 0.000 description 1
- 206010033078 Otitis media Diseases 0.000 description 1
- 241000736199 Paeonia Species 0.000 description 1
- 235000006484 Paeonia officinalis Nutrition 0.000 description 1
- 206010034038 Parotitis Diseases 0.000 description 1
- 102000019197 Superoxide Dismutase Human genes 0.000 description 1
- 108010012715 Superoxide dismutase Proteins 0.000 description 1
- 230000001133 acceleration Effects 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 230000002924 anti-infective effect Effects 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 230000007012 clinical effect Effects 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 238000001647 drug administration Methods 0.000 description 1
- 210000002919 epithelial cell Anatomy 0.000 description 1
- 239000010408 film Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 230000035876 healing Effects 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 239000005550 inflammation mediator Substances 0.000 description 1
- 230000006749 inflammatory damage Effects 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 238000010902 jet-milling Methods 0.000 description 1
- VNYSSYRCGWBHLG-AMOLWHMGSA-M leukotriene B4(1-) Chemical compound CCCCC\C=C/C[C@@H](O)\C=C\C=C\C=C/[C@@H](O)CCCC([O-])=O VNYSSYRCGWBHLG-AMOLWHMGSA-M 0.000 description 1
- GWNVDXQDILPJIG-NXOLIXFESA-N leukotriene C4 Chemical compound CCCCC\C=C/C\C=C/C=C/C=C/[C@H]([C@@H](O)CCCC(O)=O)SC[C@@H](C(=O)NCC(O)=O)NC(=O)CC[C@H](N)C(O)=O GWNVDXQDILPJIG-NXOLIXFESA-N 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 239000007937 lozenge Substances 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 235000012054 meals Nutrition 0.000 description 1
- 229940041616 menthol Drugs 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 210000002200 mouth mucosa Anatomy 0.000 description 1
- 230000000474 nursing effect Effects 0.000 description 1
- 235000006286 nutrient intake Nutrition 0.000 description 1
- 230000009965 odorless effect Effects 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 230000008058 pain sensation Effects 0.000 description 1
- 230000001717 pathogenic effect Effects 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 238000003359 percent control normalization Methods 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 1
- 150000003180 prostaglandins Chemical class 0.000 description 1
- 238000010298 pulverizing process Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 238000007619 statistical method Methods 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 230000009967 tasteless effect Effects 0.000 description 1
- 230000002277 temperature effect Effects 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 238000005303 weighing Methods 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/60—Salicylic acid; Derivatives thereof
- A61K31/606—Salicylic acid; Derivatives thereof having amino groups
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/10—Alcohols; Phenols; Salts thereof, e.g. glycerol; Polyethylene glycols [PEG]; Poloxamers; PEG/POE alkyl ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/32—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. carbomers, poly(meth)acrylates, or polyvinyl pyrrolidone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
- A61K9/006—Oral mucosa, e.g. mucoadhesive forms, sublingual droplets; Buccal patches or films; Buccal sprays
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/02—Stomatological preparations, e.g. drugs for caries, aphtae, periodontitis
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Nutrition Science (AREA)
- Physiology (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
The invention provides a mesalazine gargle for treating chemotherapy oral mucositis and a preparation method thereof, wherein the mesalazine gargle is an aqueous solution and contains the following components in percentage concentration by mass (w/v%): 0.5-1.0% of mesalazine, 0.5-3.0% of wetting agent, 1.0-5.0% of surfactant, 0.1-0.5% of flavoring agent and 0.1-0.3% of pH regulator. The mesalazine gargle for treating chemotherapy oral mucositis is convenient for clinical medication, can effectively reduce the concentration of OFRs, and has good treatment effect on oral mucositis.
Description
Technical Field
The invention relates to the field of gargle, in particular to mesalazine gargle for treating chemotherapy oral mucositis and a preparation method thereof.
Background
Mesalazine (Mesalazine) is a common name of 5-aminosalicylic acid (5-aminosalicylic acid, 5-ASA), is off-white crystalline powder, is odorless and tasteless, and gradually turns dark when being exposed to light. Mesalazine is very slightly soluble in water, insoluble in ethanol, chloroform or propanol, and soluble in dilute hydrochloric acid and dilute sodium hydroxide solution. The therapeutic action of mesalazine mainly depends on the local contact condition of the medicine and the pathological part of the mucous membrane, and in-vitro experimental results show that mesalazine has an inhibiting effect on biosynthesis and release of certain inflammation mediators (prostaglandin, leukotriene B4 and C4) and can remove Oxygen Free Radicals (OFRs) so as to improve the inflammation of the mucous membrane.
Oral Mucositis (Oral Mucositis) refers to inflammatory and ulcerative reactions of the Oral cavity, the incidence of chemotherapy patients is high, and the continuous pain of the Oral Mucositis not only seriously affects the nutrient intake and the quality of life of the patients, but also can induce complications such as parotitis, otitis media or systemic severe infection. A great deal of research at present considers that the OFRs are an important pathogenic link causing oral mucositis, the activity of superoxide dismutase for removing the OFRs is reduced due to radiotherapy and chemotherapy, the concentration of the OFRs is increased, and the OFRs can attack some important enzymes in epithelial cells to cause inflammatory damage to oral mucosal tissues.
The existing topical medicines for treating chemotherapy oral mucositis comprise anti-infective medicines, cytokines, healing promoting medicines, pain relieving medicines and the like, the medicines are prepared into different dosage forms (films, lozenges, powder, gargle and the like) to act on affected parts, the topical application is convenient to operate, the effect is obvious, and the toxic and side effects of the whole body are small. However, the drugs only have the effects of relieving oral discomfort or preventing secondary infection, are temporary and permanent, and cannot effectively reduce the concentration of OFRs, so that oral mucosa inflammation is easy to relapse.
Disclosure of Invention
In order to make up the defects in the prior art, the invention provides the mesalazine gargle for treating chemotherapy oral mucositis, which is convenient for clinical medication, can effectively reduce the concentration of OFRs and has good treatment effect on oral mucositis.
In order to achieve the purpose, the technical scheme adopted by the invention is as follows:
the mesalazine gargle for treating chemotherapy oral mucositis is an aqueous solution and comprises the following components in percentage by mass and volume (w/v%): 0.5-1.0% of mesalazine, 0.5-3.0% of wetting agent, 1.0-5.0% of surfactant, 0.1-0.5% of flavoring agent and 0.1-0.3% of pH regulator.
Further, the particle size of the mesalazine is 1-10 μm.
Further, the mass ratio of the mesalazine to the wetting agent is 1:2, and the mass ratio of the mesalazine to the surfactant is 1: 3.
Further, the wetting agent is a compound of any two of ethanol, propylene glycol, polyethylene glycol 400, glycerol and sorbitol.
Further, the wetting agent is prepared by compounding ethanol and glycerol according to the proportion of 1: 2.
Further, the surfactant is a compound of any two of polysorbate-80, lecithin, polyvinylpyrrolidone-15, polyvinylpyrrolidone-17, polyvinylpyrrolidone-30 and polyoxyethylene 40 hydrogenated castor oil.
Furthermore, the surfactant is compounded by polysorbate-80 and polyvinylpyrrolidone-15 according to the proportion of 1: 1.5.
Further, the flavoring agent is one or more of menthol, sodium cyclamate, saccharin sodium, sucrose and stevioside.
Further, the pH regulator is one or a mixture of sodium citrate, sodium bicarbonate, sodium dihydrogen phosphate and sodium hydroxide.
The invention also provides a preparation method of the mesalazine gargle for treating chemotherapy oral mucositis, which comprises the following steps:
s1, preprocessing: crushing mesalazine into particles with the particle size of 1-10 mu m by airflow;
s2, preparation: adding purified water accounting for 70% of the total preparation amount into a preparation tank, sequentially adding a wetting agent, a surfactant and a flavoring agent into the preparation tank according to a proportion, uniformly stirring for 20 minutes at 30-35 ℃, adding pretreated mesalazine according to a proportion, stirring for 30-40 minutes at 35 ℃, adding the purified water to a specified amount after complete dissolution, adjusting the pH value to 6.5-7.0 by using a pH regulator, and finally filtering by using a filter membrane with the aperture of 0.45 mu m. Sterilizing the obtained mesalazine gargle, and packaging to obtain the final product.
Compared with the prior art, the invention has the following beneficial technical effects:
1. the mesalazine gargle is a mucosal inflammation treatment drug, has strong specificity, and has obvious treatment effect on oral mucositis (inflammatory and ulcerative reactions) caused by a chemotherapy process, so that the smooth chemotherapy can be ensured, and the survival quality of tumor patients is improved.
2. The mesalazine gargle disclosed by the invention adopts local administration, is convenient for clinical medication, has small stimulation to the affected part of the oral cavity by liquid medicine, is economical and practical, and can be used as a long-term oral cavity nursing medicine when a patient starts chemotherapy.
3. The invention solves the problem of low drug-loading rate of mesalazine in aqueous solution by controlling the particle size of the mesalazine raw material, and simultaneously, the mesalazine raw material is scientifically screened and combined with other auxiliary materials, so that the bioavailability of the drug is high.
Detailed Description
In the following description, numerous specific details are set forth in order to provide a thorough understanding of the present invention. This invention may, however, be embodied in many different forms and should not be construed as limited to the embodiments set forth herein.
The mesalazine gargle is an aqueous solution and contains the following components in percentage by mass and volume (w/v%):
| mesalazine | Wetting agent | Surface active agent | Flavouring agent | pH regulator | |
| Example 1 | 0.25 | 0.5 | 0.75 | 0.1 | 0.1 |
| Example 2 | 0.5 | 1.0 | 0.15 | 0.2 | 0.2 |
| Example 3 | 1.0 | 2.0 | 3.0 | 0.4 | 0.3 |
| Example 4 | 1.5 | 3.0 | 4.5 | 0.6 | 0.4 |
In the raw materials, the particle size of the mesalazine is controlled to be 1-10 mu m, so that the drug loading of the mesalazine in the gargle can be obviously improved. The mass ratio of mesalazine to the wetting agent is 1:2, and the mass ratio of mesalazine to the surfactant is 1: 3.
The wetting agent is a compound of any two of ethanol, propylene glycol, polyethylene glycol 400, glycerin and sorbitol, and preferably the ethanol and the glycerin are compounded according to the proportion of 1: 2. The surfactant is a compound of any two of polysorbate-80, lecithin, polyvinylpyrrolidone-15, polyvinylpyrrolidone-17, polyvinylpyrrolidone-30 and polyoxyethylene 40 hydrogenated castor oil, preferably the compound of polysorbate-80 and polyvinylpyrrolidone-15 according to the proportion of 1: 1.5.
The correctant is one or more of Mentholum, sodium cyclamate, saccharin sodium, sucrose, and stevioside. The pH regulator is one or more of sodium citrate, sodium bicarbonate, sodium dihydrogen phosphate and sodium hydroxide.
A preparation method of mesalazine gargle for treating chemotherapy oral mucositis comprises the following steps:
1. pretreatment: setting the pressure of an air compressor of a QYF-2600 type jet mill to be 0.4MPa, the crushing pressure of a main air valve to be 0.8MPa, the pressure of a back-flushing valve to be 0.4-0.6 MPa and the crushing amount to be 5-10 g/s, and then carrying out jet milling on mesalazine to obtain the particles with the particle size of 1-10 microns by controlling the rotating speed of a grading wheel.
2. Preparation: adding purified water accounting for 70% of the total preparation amount into a preparation tank, sequentially adding a wetting agent, a surfactant and a flavoring agent into the preparation tank according to a proportion, uniformly stirring for 20 minutes at 30-35 ℃, adding pretreated mesalazine according to a proportion, stirring for 30-40 minutes at 35 ℃, adding the purified water to a specified amount after complete dissolution, adjusting the pH value to 6.5-7.0 by using a pH regulator, and finally filtering by using a filter membrane with the aperture of 0.45 mu m. Sterilizing the obtained mesalazine gargle, and packaging to obtain the final product.
Mesalazine solubility test
Materials and apparatus
Crushed sample sources: mesalazine (peony river Hengyun pharmaceutical Co., Ltd., lot number: 2-20200315);
airflow crushing equipment: model QYF-2600 jet mill (kunshan miller mechanical manufacturing limited).
Pulverizing mesalazine
Firstly setting the pressure of an air compressor of a QYF-2600 type jet mill to be 0.4MPa, the crushing pressure of a main air valve to be 0.8MPa, the pressure of a back-flushing valve to be 0.4-0.6 MPa and the crushing amount to be 5-10 g/s, and then controlling the rotating speed of a grading wheel to crush mesalazine into raw materials with different particle size ranges (1-10 microns, 10-20 microns, 20-30 microns and 30-50 microns).
Solubility test
Respectively weighing 5.0g of mesalazine with different particle sizes, placing the mesalazine in 100ml of blank gargle (pH value of 6.5-7.0) at 25 +/-2 ℃, and stirring for 15 minutes at room temperature. And observing the dissolution of mesalazine with different particle sizes in the gargle. Taking the liquid medicine, and measuring the content of mesalazine in the gargle by using a high performance liquid chromatography according to a measuring method under the item of mesalazine content measurement of national drug administration standard YBH 17982006.
Test results
The solubility of mesalazine with different particle sizes in a blank gargle (pH value of 6.5-7.0) is shown in table 1, the solubility difference of the mesalazine with different particle sizes obtained by crushing in the gargle is obvious, the smaller the particle size is, the larger the solubility of the mesalazine in the gargle is, and the solubility of the mesalazine with the particle size of 1-10 mu m can reach 20.21mg/ml, which shows that the drug-loading rate of the mesalazine in the gargle can be obviously improved by controlling the particle size of the mesalazine raw material.
TABLE 1 solubility chart of mesalazine with different particle sizes in blank gargle (pH 6.5-7.0)
| Sample particle size | Solubility of sample |
| Particle size of unpulverized mesalazine: 100-200 mu m | 0.34mg/ml |
| Particle size: 30-50 mu m | 9.91mg/ml |
| Particle size: 20-30 mu m | 15.12mg/ml |
| Particle size: 10-20 mu m | 18.75mg/ml |
| Particle size: 1-10 mu m | 20.21mg/ml |
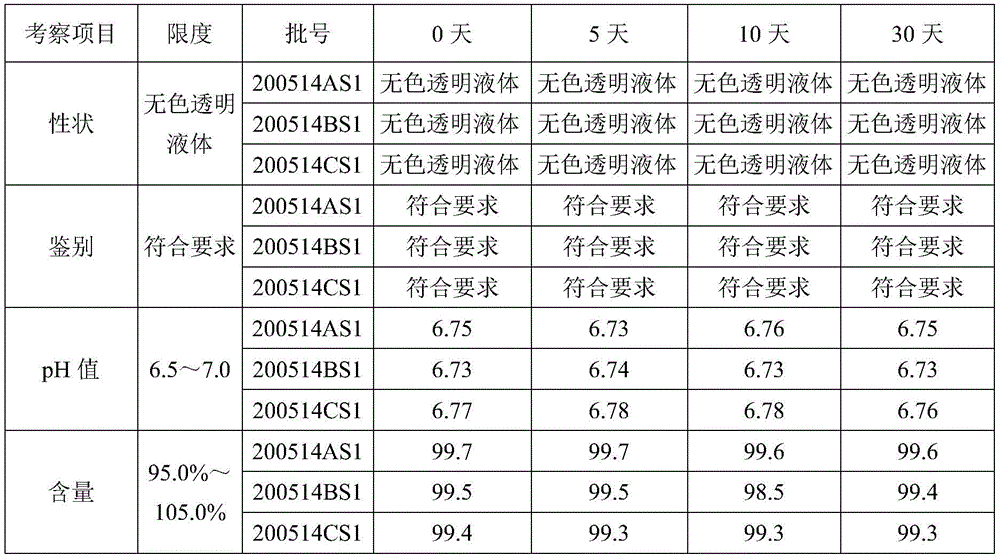
Mesalazine mouthrinse stability test
The stability test of the 1.0% mesalazine gargle prepared in example 3 was carried out, and the 3 batches of products (batch numbers: 200514AS1, 200514BS1 and 200514CS1) were subjected to the light irradiation and high temperature effect test and the 6-month acceleration test respectively according to the guidelines of stability test in the' Chinese pharmacopoeia 2020 edition, and the specific test method and structure are AS follows:
high-temperature test: placing the test sample at the high temperature of 60 +/-2 ℃, and sampling and detecting on 5 days, 10 days and 30 days; detection indexes are as follows: properties, identification, pH value and content.
TABLE 21.0% Mesalazine gargle high temperature test (60 + -2 deg.C) results table
And (3) illumination test: placing the test sample under the condition of 4500 +/-500 lx light, and sampling and detecting on 5 days, 10 days and 30 days; detection indexes are as follows: properties, identification, pH value and content.
TABLE 31.0% Mesalazine collutory illumination test (4500 + -500 lx) test results Table
And (3) accelerated test: placing the test sample at 40 + -2 deg.C and RH less than or equal to 25%, and sampling and detecting at month 1, month 3 and month 6; detection indexes are as follows: properties, identification, pH value and content.
TABLE 41.0% Mesalazine gargle accelerated test (40 + -2 deg.C, 25% RH or less) test results Table
From the above test results table, the 1.0% mesalazine gargle product of example 3 of the present invention has good stability and the formulation process is reasonable and feasible.
Mesalazine gargle clinical effect test
A comparison test is carried out on the 1.0% mesalazine gargle product in the embodiment 3 of the invention, 0.15% interleukin 11 gargle of a common medicament for treating chemotherapy oral mucositis and 0.75% vitamin B12 gargle to confirm the clinical treatment effect of the product in treating the chemotherapy oral mucositis.
Test grouping
The test randomly divides the chemotherapy patients into 1.0% mesalazine gargle group, 0.15% interleukin 11 gargle group, 0.75% vitamin B12 gargle group and a control group, and the comparative differences of the sex, the age, the oral mucositis degree and the like of 60 patients and 4 patients with chemotherapy in each group have no statistical significance (P is more than 0.05).
Method of administration
The patients in the 1.0% mesalazine gargle group, the 0.15% interleukin 11 gargle group and the 0.75% vitamin B12 gargle group use 60ml of liquid medicine per treatment day under the guidance of doctors, and the liquid medicine is divided into 4 equal parts, 15ml of each part. The patient first cleans the oral cavity with water, then gargles the liquid medicine for 3min and swallows the liquid medicine, gargles the liquid medicine for 4 times a day. The gargle is used after a meal to ensure that the mucosal contact time is as long as possible. The gargle is used until the last 1 day of the radiotherapy treatment course; the control group was intervened with the routine care of the department.
Criteria for efficacy assessment
The clinical treatment effect was classified into 4 grades according to the change record of pain sensation before and after treatment of patients: clinical control, obvious effect, effectiveness and ineffectiveness.
And (3) clinical control: pain disappeared, none (score 0);
the effect is shown: pain ranges from heavy (7-10 points) to light (less than 3 points);
the method has the following advantages: pain varies from heavy (7-10 min) to light (4-6 min);
and (4) invalidation: the pain was not improved.
Statistical method
SPSS16.0 software statistical analysis is performed on the data, a Fisher accurate test is used for analyzing a frequency table, repeated measurement variance analysis is used for analyzing repeated measurement statistical data, and the difference is statistically significant when P is less than 0.05.
Test results
The comparative test of the treatment effect of the 1.0% mesalazine gargle and other medicaments is shown in table 5, the total effective rate of the 1.0% mesalazine gargle for treating chemotherapy oral mucositis reaches 100%, the clinical control rate reaches 63.33%, the significance is higher than 0.15% of interleukin 11 gargle and 0.75% of vitamin B12 gargle, and the result shows that the 1.0% mesalazine gargle has good clinical treatment effect on chemotherapy oral mucositis.
TABLE 51.0 comparison of the therapeutic effects of mesalazine gargle with other drugs
| Group of | Number of examples | Clinical control | Show effect | Is effective | Invalidation | Clinical control rate | Total effective rate |
| 1.0% mesalazine gargle | 60 | 38 | 18 | 4 | 0 | 63.33% | 100.00% |
| 0.15% interleukin 11 gargle | 60 | 15 | 21 | 16 | 8 | 25.00% | 86.67% |
| 0.75% vitamin B12Gargle | 60 | 8 | 12 | 21 | 19 | 13.33% | 68.33% |
| Control group | 60 | 0 | 0 | 5 | 55 | 0 | 8.30% |
Although the present invention has been described in detail with reference to the embodiments, it will be apparent to those skilled in the art that modifications may be made to the embodiments or portions thereof without departing from the spirit and scope of the invention.
Claims (10)
1. The mesalazine gargle for treating chemotherapy oral mucositis is characterized by being an aqueous solution and containing the following components in percentage by mass (w/v%): 0.5-1.0% of mesalazine, 0.5-3.0% of wetting agent, 1.0-5.0% of surfactant, 0.1-0.5% of flavoring agent and 0.1-0.3% of pH regulator.
2. The mesalazine mouthrinse for the treatment of chemotherapy oral mucositis according to claim 1, characterized in that: the particle size of the mesalazine is 1-10 mu m.
3. The mesalazine mouthrinse for the treatment of chemotherapy oral mucositis according to claim 2, characterized in that: the mass ratio of the mesalazine to the wetting agent is 1:2, and the mass ratio of the mesalazine to the surfactant is 1: 3.
4. The mesalazine mouthrinse for the treatment of chemotherapy oral mucositis according to claim 3, characterized in that: the wetting agent is a compound of any two of ethanol, propylene glycol, polyethylene glycol 400, glycerol and sorbitol.
5. The mesalazine mouthrinse for the treatment of chemotherapy oral mucositis according to claim 4, characterized in that: the wetting agent is prepared by compounding ethanol and glycerol according to the proportion of 1: 2.
6. The mesalazine mouthrinse for the treatment of chemotherapy oral mucositis according to claim 5, characterized in that: the surfactant is a compound of any two of polysorbate-80, lecithin, polyvinylpyrrolidone-15, polyvinylpyrrolidone-17, polyvinylpyrrolidone-30 and polyoxyethylene 40 hydrogenated castor oil.
7. The mesalazine mouthrinse for the treatment of chemotherapy oral mucositis according to claim 6, characterized in that: the surfactant is compounded by polysorbate-80 and polyvinylpyrrolidone-15 according to the proportion of 1: 1.5.
8. The mesalazine mouthrinse for the treatment of chemotherapy oral mucositis according to claim 7, characterized in that: the correctant is one or more of Mentholum, sodium cyclamate, saccharin sodium, sucrose, and stevioside.
9. The mesalazine mouthrinse for the treatment of chemotherapy oral mucositis according to claim 8, characterized in that: the pH regulator is one or more of sodium citrate, sodium bicarbonate, sodium dihydrogen phosphate and sodium hydroxide.
10. The method for preparing mesalazine gargle for treating chemotherapy oral mucositis according to any of claims 1 to 9, comprising the following steps:
s1, preprocessing: crushing mesalazine into particles with the particle size of 1-10 mu m by airflow;
s2, preparation: adding purified water accounting for 70% of the total preparation amount into a preparation tank, sequentially adding a wetting agent, a surfactant and a flavoring agent into the preparation tank according to a proportion, uniformly stirring for 20 minutes at 30-35 ℃, adding pretreated mesalazine according to a proportion, stirring for 30-40 minutes at 35 ℃, adding the purified water to a specified amount after complete dissolution, adjusting the pH value to 6.5-7.0 by using a pH regulator, and finally filtering by using a filter membrane with the aperture of 0.45 mu m.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110731248.5A CN113350277A (en) | 2021-06-30 | 2021-06-30 | Mesalazine gargle for treating chemotherapy oral mucositis and preparation method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110731248.5A CN113350277A (en) | 2021-06-30 | 2021-06-30 | Mesalazine gargle for treating chemotherapy oral mucositis and preparation method thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN113350277A true CN113350277A (en) | 2021-09-07 |
Family
ID=77537169
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110731248.5A Pending CN113350277A (en) | 2021-06-30 | 2021-06-30 | Mesalazine gargle for treating chemotherapy oral mucositis and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113350277A (en) |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5010069A (en) * | 1989-05-15 | 1991-04-23 | Marion Laboratories, Inc. | Stable liquid form of 5-aminosalicylic acid |
| US6217897B1 (en) * | 1996-08-13 | 2001-04-17 | Tillotts Pharma Ag | Oral mucosal composition comprising 5-aminosalicylic acid |
| US20030185761A1 (en) * | 1997-10-01 | 2003-10-02 | Dugger Harry A. | Buccal, polar and non-polar spray or capsule containing drugs for treating pain |
| CN104603100A (en) * | 2012-05-23 | 2015-05-06 | 塞利克斯比奥私人有限公司 | Compositions and methods for treating inflammatory bowel disease |
| CN104706582A (en) * | 2013-12-12 | 2015-06-17 | 四川健能制药有限公司 | Mesalazine-containing drug composition |
| CN111686186A (en) * | 2020-06-24 | 2020-09-22 | 杭州巴洛特生物科技有限公司 | Production process of mesalazine oral liquid |
-
2021
- 2021-06-30 CN CN202110731248.5A patent/CN113350277A/en active Pending
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5010069A (en) * | 1989-05-15 | 1991-04-23 | Marion Laboratories, Inc. | Stable liquid form of 5-aminosalicylic acid |
| US6217897B1 (en) * | 1996-08-13 | 2001-04-17 | Tillotts Pharma Ag | Oral mucosal composition comprising 5-aminosalicylic acid |
| US20030185761A1 (en) * | 1997-10-01 | 2003-10-02 | Dugger Harry A. | Buccal, polar and non-polar spray or capsule containing drugs for treating pain |
| CN104603100A (en) * | 2012-05-23 | 2015-05-06 | 塞利克斯比奥私人有限公司 | Compositions and methods for treating inflammatory bowel disease |
| CN104706582A (en) * | 2013-12-12 | 2015-06-17 | 四川健能制药有限公司 | Mesalazine-containing drug composition |
| CN111686186A (en) * | 2020-06-24 | 2020-09-22 | 杭州巴洛特生物科技有限公司 | Production process of mesalazine oral liquid |
Non-Patent Citations (1)
| Title |
|---|
| 傅超美、刘文: "《中药药剂学》", 31 August 2018, 中国医药科技出版社 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN109288816A (en) | A kind of chloroquine gel and preparation method and application thereof | |
| CN106361784A (en) | Use of mangosteen fruit shell extract for treating skin diseases | |
| CN106138190B (en) | External gel preparation, preparation method and application thereof | |
| CN103446294B (en) | Murrayae folium ET Cacumen extract preparation method, Murrayae folium ET Cacumen extract obtained thereby and application thereof | |
| WO2024027673A1 (en) | Use of nocardia rubra cell wall skeleton in treatment of chronic cervicitis | |
| CN113350277A (en) | Mesalazine gargle for treating chemotherapy oral mucositis and preparation method thereof | |
| CN115998761A (en) | Antiallergic rhinitis composition and preparation method and application thereof | |
| CN111683670B (en) | Use of nocardia rubra cell wall scaffold in the treatment of recurrent aphtha | |
| CN114886945A (en) | Supramolecular system for regulating purine metabolism and application thereof | |
| CN110448527B (en) | Colchicine oral solution and prescription composition thereof | |
| CN101961321B (en) | Zinc gluconate sugar tablet preparation and preparation method thereof | |
| CN104800322A (en) | Traditional Chinese medicine composition for treating chronic pharyngitis and preparation method of traditional Chinese medicine composition | |
| CN113563493B (en) | Hydrophobic polysaccharide and preparation method and application thereof | |
| EP3708153A1 (en) | Solution preparation for aerosol inhalation of carbocisteine, and preparation method therefor | |
| CN117338717B (en) | A baicalein external preparation and its application in treating atopic dermatitis | |
| CN107174577A (en) | A kind of gel containing EGCG and preparation method thereof | |
| CN115443144B (en) | Application of Nocardia rubrum cell wall skeleton in the treatment of radiation sickness | |
| CN114159414A (en) | Favipiravir solution for atomization inhalation and preparation method thereof | |
| CN116942721B (en) | Nasal cavity nursing liquid based on garlicin | |
| CN111419883B (en) | Preparation method of nasal spray for treating allergic rhinitis | |
| CN114948914B (en) | Moxifloxacin hydrochloride solution for aerosol inhalation and preparation method thereof | |
| CN115300576B (en) | Compound traditional Chinese medicine nanogel for treating beriberi and tinea pedis and preparation method thereof | |
| CN111529491B (en) | Quick-acting safe ticagrelor oral spray and preparation method thereof | |
| CN119818628A (en) | Spray film agent for local bacteriostasis and preparation method thereof | |
| HK40076960A (en) | Use of nocardia rubra cell wall skeleton in treatment of radiation sickness |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20210907 |