CN113186275A - Application of serum amyloid A1 in preparation of biomarker for diagnosing polycystic ovarian syndrome - Google Patents
Application of serum amyloid A1 in preparation of biomarker for diagnosing polycystic ovarian syndrome Download PDFInfo
- Publication number
- CN113186275A CN113186275A CN202110665056.9A CN202110665056A CN113186275A CN 113186275 A CN113186275 A CN 113186275A CN 202110665056 A CN202110665056 A CN 202110665056A CN 113186275 A CN113186275 A CN 113186275A
- Authority
- CN
- China
- Prior art keywords
- saa1
- serum amyloid
- biomarker
- pcos
- serum
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
- C12Q1/6883—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/158—Expression markers
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Wood Science & Technology (AREA)
- Analytical Chemistry (AREA)
- Zoology (AREA)
- Genetics & Genomics (AREA)
- Engineering & Computer Science (AREA)
- Pathology (AREA)
- Immunology (AREA)
- Microbiology (AREA)
- Molecular Biology (AREA)
- Biotechnology (AREA)
- Biophysics (AREA)
- Physics & Mathematics (AREA)
- Biochemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Abstract
The invention provides application of serum amyloid A1 in preparation of a biomarker for diagnosing polycystic ovary syndrome. The invention also provides application of a reagent for detecting serum amyloid A1 in preparing a diagnostic composition for in vitro diagnosis of a biomarker of polycystic ovarian syndrome, wherein the upstream primer sequence for detecting serum amyloid A1 is shown as SEQ ID NO: 2, the sequence of the downstream primer for detecting serum amyloid A1 is shown as SEQ ID NO: 3, the diagnosis comprises: detecting the level of serum amyloid a1 biomarker from the patient. The experimental research result of the invention indicates that SAA1 plays an important role in the occurrence and development of PCOS disease, and SAA1 can be used as a biomarker for evaluating the PCOS disease state.
Description
Technical Field
The invention belongs to the field of biological medicines, and relates to serum amyloid A1, in particular to application of serum amyloid A1 as a biomarker in diagnosis of polycystic ovarian syndrome.
Background
Polycystic ovary syndrome (PCOS) is the most common disorder of endocrine reproduction and metabolism, and the incidence rate of women in the reproductive age is 5% -10%, accounting for about 75% of anovulatory infertility. PCOS is characterized primarily by rare or no ovulation, hyperandrogenism and or clinical hyperandrogenism, with ovarian polycystic manifestations as typified by infertility, obesity, impaired glucose tolerance and insulin resistance. To date, the etiology and pathogenesis of PCOS remains unclear.
Serum Amyloid A (SAA) is a reactive protein in the acute phase of inflammation or injury, is secreted by the liver in a large amount in a short time, is 1000 times higher than the normal value, and is one of the most sensitive inflammatory reaction markers at present. SAA is also expressed in extrahepatic tissues such as macrophages and adipocytes, and is also used as a marker reflecting the degree of chronic inflammatory response in the body. SAA is mainly divided into four subtypes of SAA1, SAA2, SAA3 and SAA4, wherein SAA1 and SAA2 are acute phase reaction proteins, SAA3 is mainly expressed in mice but not in human bodies, and SAA4 is a constitutive gene and is not regulated by inflammation and stress. PCOS is a chronic inflammatory disease and the role of SAA in the development and progression of PCOS is not clear.
The amino acid sequence of SAA1 is shown in SEQ ID NO. 5.
Disclosure of Invention
In view of the above technical problems in the prior art, the present invention provides the use of serum amyloid a1 in the preparation of a biomarker for diagnosing polycystic ovarian syndrome, and the use is to solve the technical problem that no good method for diagnosing polycystic ovarian syndrome exists in the prior art.
The invention provides application of serum amyloid A1 in preparation of a biomarker for diagnosing polycystic ovary syndrome.
The invention also provides application of a reagent for detecting serum amyloid A1 in preparing a diagnostic composition for in vitro diagnosis of a biomarker of polycystic ovarian syndrome, wherein the upstream primer sequence for detecting serum amyloid A1 is shown as SEQ ID NO: 1, and the sequence of the downstream primer for detecting serum amyloid A1 is shown as SEQ ID NO: 2, the diagnosis comprising: detecting the level of serum amyloid a1 biomarker from the patient.
The invention discovers that SAA1 (the most important acute phase reaction protein of SAA) is remarkably increased in the serum and follicular fluid of a patient with PCOS, and the serum SAA1 level is closely related to the clinical characteristics of PCOS, such as high LH blood disease, high androgen blood disease, insulin resistance and the like. Meanwhile, the ovarian local SAA1 level is positively correlated with the serum SAA1 level, but the ovarian local SAA1 level is obviously higher than the serum SAA1 level, which indicates that the synthesis of the ovarian local SAA1 and the serum-derived SAA1 are both reasons for the increase of the PCOS ovarian local SAA1, and the ovarian local increased SAA1 can cause the insulin resistance of granulosa cells, thereby causing the dysfunction of the granulosa cells and influencing the follicular development.
Compared with the prior art, the invention has remarkable technical progress. The experimental research result of the invention indicates that SAA1 plays an important role in the occurrence and development of PCOS disease, and SAA1 can be used as a biomarker for evaluating the PCOS disease state.
Drawings
Figure 1 shows that serum SAA1 is significantly elevated in the PCOS population; the figure is as follows: p < 0.001.
Fig. 2 shows that follicular fluid SAA1 is significantly elevated locally in the PCOS ovary; the figure is as follows: p < 0.001.
Figure 3 shows the correlation analysis of follicular fluid with serum SAA 1.
FIG. 4 shows SAA1 expression in ovarian granulosa cells; the figure is as follows: a: SAA1 PCR gel map; b: SAA1 immunofluorescence represents the graph.
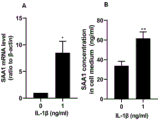
FIG. 5 shows that granulosa cells have the ability to synthesize SAA1 de novo; the figure is as follows: a: IL-1 β promotes granulosa cell SAA1mRNA expression,/vs 0, P < 0.05; b IL-1 β promotes synthesis and secretion of granulocyte SAA1 protein,. v s0, P < 0.01.
Figure 6 shows that SAA1 causes granulocytic insulin resistance; the figure is as follows: a: PTEN WB represents diagram, B: vs 0P < 0.01; #, vs 0.01, P <0.05. C: PAKT represents a graph; d: p <0.01, P < 0.001.
Detailed Description
Example 1
[ Experimental techniques ]:
experimental method and materials
1. Collecting human serum and follicular fluid: PCOS and control patient sera and follicular fluid were collected.
The above cases were obtained from the reproductive medicine center of the affiliated renji hospital of Shanghai university of transportation medical school, and were reviewed by the ethical Committee of the renji hospital.
The PCOS diagnosis standard is the LUTAN standard, and the control is the patient with single oviduct factor infertility or male factor infertility. Ovulation is promoted by a conventional method, and blood and follicular fluid are collected on the day of ovum collection. Serum is obtained after blood centrifugation and is frozen, dominant follicular fluid in the first tube is frozen after centrifugation, and granulosa cells are obtained after density gradient centrifugation and enzyme digestion of the residual follicular fluid and are used for freezing and RNA extraction.
2. Granulosa cells SAA1 expression: non-PCOS ovary granular cells are collected, total RNA is extracted, after reverse transcription is carried out, SAA1 is amplified by adopting a common PCR method, an amplification product is subjected to agarose electrophoresis gel electrophoresis, and a solar imaging system is adopted for photographing after the electrophoresis is finished. Performing SAA1 protein expression detection by immunofluorescence, culturing granular cells in vitro for 1 day, adding 1% paraformaldehyde for fixation, performing membrane permeation by 0.1% PBST, performing blocking by 5% goat serum for 2 hours, incubating SAA1 antibody (1:100) overnight, washing with PBS for 3 times, incubating with Alexa Fluor 488 (green) labeled secondary antibody for 2 hours, washing with PBS for 3 times in the dark, and taking pictures by a Zeiss microscope.
3. Human granulosa cell in vitro culture: collecting follicular fluid obtained after ovum retrieval of normal non-PCOS, centrifuging, adding hyaluronidase for digestion, inoculating the isolated granulosa cells into 6-well, 12-well or 24-well plate coated with rat tail collagen, and placing in CO with volume percentage concentration of 5%2After three days of incubation in an incubator at 37 ℃, the next step was carried out by changing to DMEM/F12 medium (GIBCO, Saimer fly) without serum and phenol red.
4. Human granular cell in-vitro culture treatment: after the granulosa cells obtained in section 1) above were treated with IL-1. beta. (1ng/ml) for 24 hours, cell culture supernatants and RNAs were collected, and the RNA and protein expression levels of SAA1 were measured. After 24 hours of treatment with SAA1(0, 0.01, 0.1, 1. mu.g/ml), the medium was replaced with serum-free medium and treated with 100nM insulin for 15 minutes to detect the expression of PTEN and Akt phosphorylation.
5. Total RNA extraction and reverse transcription: total RNA was isolated from endometrial tissue and cells using TRIzol reagent (Invitrogen) according to the instructions. RNA concentration and quality were determined by a Nanodrop ND-2000 spectrophotometer (Thermo Fisher scientific). cDNA was synthesized using PrimeScript RT kit (Takara, Dalian, China) according to the instructions, taking 500ng of total RNA.
6. Detecting the expression level of SAA1 by real-time fluorescent quantitative PCR: real-time fluorescent quantitative PCR was performed using SYBR Green Master Mix (Takara, Dalian, China) and ABI Prism System (Applied Biosystems, Thermo Fisher Scientific)
Sequence detection of SAA 1:
forward 5'-tttctgctccttggtcctgg-3'; shown as SEQ ID NO. 1.
reverse 5'-ctctggcatcgctgatcact-3'; shown as SEQ ID NO. 2.
Sequence detection for ACTB:
forward 5'-gggaaatcgtgcgtgacattaag-3'; shown as SEQ ID NO. 3.
reverse 5'-tgtgttggcgtacaggtctttg-3'; shown as SEQ ID NO. 4.
All quantitative RT-PCR reactions were performed in triplicate. Relative mRNA levels were calculated using ACTB as the internal control and using a comparative cycle threshold method.
7. Detection of serum and follicular fluid SAA 1: serum and follicular fluid of PCOS and control population were collected and assayed for SAA1 protein levels (R & D) by enzyme-linked immunosorbent assay (ELISA).
8. The statistical method comprises the following steps: adopting single-factor variance analysis to test whether the mean difference of each group has statistical significance; the checking level is p <0.05. SPSS 22.0 statistical software is used for data statistics and analysis.
Second, experimental results
1. Serum SAA1 was significantly elevated in the PCOS population and correlated with the clinical profile of PCOS
We tested serum SAA1 in PCOS and control populations using ELISA and found that serum SAA1 levels were significantly elevated in PCOS populations (P <0.001, N ═ 32/group), as shown in figure 1. Correlation analysis is carried out with clinical indexes, and the serum SAA1 level is found to be in significant positive correlation with BMI, basic LH, LH/FSH, T and AMH levels, which indicates that the serum SAA1 level is closely related with the clinical characteristics of PCOS (Table 1).
BMI, baseline LH, LH/FSH, T and AMH level indicators are important clinical indicators of PCOS in order to show that SAA1 is closely related to the clinical features of PCOS.
TABLE 1 correlation of serum SAA1 with clinical indicators of PCOS
2. The follicular fluid SAA1 is significantly increased in ovary local part of PCOS patient
When the SAA1 level in follicular fluid is detected by ELISA method by collecting follicular fluid of PCOS and control, the SAA1 level of follicular fluid of PCOS patient is remarkably increased (P)<0.001, N ═ 32/group), see fig. 2. Further correlation analysis, we found that the level of SAA1 in follicular fluid was positively correlated with the level of SAA1 in serum (R)2=0.18,P<0.001), see fig. 3.
3. Ovary granular cell SAA1 expression and synthesis
We found that ovarian granulosa cells express both SAA1mRNA and SAA1 protein using immunofluorescence and PCR techniques, as shown in FIG. 4. We collected cell culture supernatant by using a granulosa cell in vitro culture model, and found that the granulosa cells can secrete SAA1 protein, the basal expression level of the protein is about 30ng/ml, and the addition of inflammatory factor interleukin-1 beta can significantly increase the ability of the granulosa cells to secrete SAA1, as shown in FIG. 5. This suggests that granulosa cells have the ability to de novo synthesize SAA 1.
SAA1 causing insulin resistance in ovarian granulosa cells
We used an in vitro granular cell culture model, and added SAA1 in vitro, and found that SAA1 can promote PTEN expression with a concentration gradient. But significantly down-regulated insulin-stimulated Akt phosphorylation, leading to granular cell insulin resistance, see figure 6. This suggests that too much SAA1 may lead to granulocytic insulin resistance.
The results show that the SAA1 is remarkably increased in the serum and the ovary of the PCOS patient, and the serum SAA1 level is closely related to the clinical characteristics of PCOS high LH blood disease, high androgen blood disease and the like. At the same time, locally increased SAA1 of the ovary can cause the insulin resistance of the granulosa cell to cause the dysfunction of the granulosa cell, and further influences the development of the PCOS follicle. These studies suggest that SAA1 plays an important role in the development of PCOS, and SAA1 can be used as a new biomarker to indicate the occurrence and progression of PCOS diseases.
Sequence listing
<110> Shanghai university of traffic medical college affiliated renji hospital
Application of <120> serum amyloid A1 in preparation of biomarker for diagnosing polycystic ovarian syndrome
<160> 5
<170> SIPOSequenceListing 1.0
<210> 1
<211> 20
<212> DNA
<213> Artificial Sequence (Artificial Sequence)
<400> 1
<210> 2
<211> 20
<212> DNA
<213> Artificial Sequence (Artificial Sequence)
<400> 2
ctctggcatc gctgatcact 20
<210> 3
<211> 23
<212> DNA
<213> Artificial Sequence (Artificial Sequence)
<400> 3
gggaaatcgt gcgtgacatt aag 23
<210> 4
<211> 22
<212> DNA
<213> Artificial Sequence (Artificial Sequence)
<400> 4
tgtgttggcg tacaggtctt tg 22
<210> 5
<211> 122
<212> PRT
<213> Homo sapiens
<400> 5
Met Lys Leu Leu Thr Gly Leu Val Phe Cys Ser Leu Val Leu Gly Val
1 5 10 15
Ser Ser Arg Ser Phe Phe Ser Phe Leu Gly Glu Ala Phe Asp Gly Ala
20 25 30
Arg Asp Met Trp Arg Ala Tyr Ser Asp Met Arg Glu Ala Asn Tyr Ile
35 40 45
Gly Ser Asp Lys Tyr Phe His Ala Arg Gly Asn Tyr Asp Ala Ala Lys
50 55 60
Arg Gly Pro Gly Gly Ala Trp Ala Ala Glu Val Ile Thr Asp Ala Arg
65 70 75 80
Glu Asn Ile Gln Arg Phe Phe Gly His Gly Ala Glu Asp Ser Leu Ala
85 90 95
Asp Gln Ala Ala Asn Glu Trp Gly Arg Ser Gly Lys Asp Pro Asn His
100 105 110
Phe Arg Pro Ala Gly Leu Pro Glu Lys Tyr
115 120
Claims (2)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110665056.9A CN113186275A (en) | 2021-06-16 | 2021-06-16 | Application of serum amyloid A1 in preparation of biomarker for diagnosing polycystic ovarian syndrome |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110665056.9A CN113186275A (en) | 2021-06-16 | 2021-06-16 | Application of serum amyloid A1 in preparation of biomarker for diagnosing polycystic ovarian syndrome |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN113186275A true CN113186275A (en) | 2021-07-30 |
Family
ID=76976538
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110665056.9A Pending CN113186275A (en) | 2021-06-16 | 2021-06-16 | Application of serum amyloid A1 in preparation of biomarker for diagnosing polycystic ovarian syndrome |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113186275A (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115407067A (en) * | 2022-06-22 | 2022-11-29 | 郑州大学第一附属医院 | Sepsis diagnostic marker |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1993136A (en) * | 2003-12-17 | 2007-07-04 | 爱尔康公司 | Use of serum amyloid a gene in diagnosis and treatment of glaucoma and identification of anti-glaucoma agents |
| GB0821839D0 (en) * | 2008-12-01 | 2009-01-07 | Tan Bee K | The use of metformin and compounds containing metformin in the treatment of diseases or conditions in patients with raised serum amyloid A |
| CN106084050A (en) * | 2009-09-25 | 2016-11-09 | 佐马美国有限公司 | New regulator |
| CN112662752A (en) * | 2021-01-18 | 2021-04-16 | 中国农业大学 | Application of biomarker for diagnosis |
-
2021
- 2021-06-16 CN CN202110665056.9A patent/CN113186275A/en active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1993136A (en) * | 2003-12-17 | 2007-07-04 | 爱尔康公司 | Use of serum amyloid a gene in diagnosis and treatment of glaucoma and identification of anti-glaucoma agents |
| GB0821839D0 (en) * | 2008-12-01 | 2009-01-07 | Tan Bee K | The use of metformin and compounds containing metformin in the treatment of diseases or conditions in patients with raised serum amyloid A |
| CN106084050A (en) * | 2009-09-25 | 2016-11-09 | 佐马美国有限公司 | New regulator |
| CN112662752A (en) * | 2021-01-18 | 2021-04-16 | 中国农业大学 | Application of biomarker for diagnosis |
Non-Patent Citations (1)
| Title |
|---|
| PRAVEENKUMAR DEVARBHAVI等: "Identification of key pathways and genes in polycystic ovary syndrome via integrated bioinformatics analysis and prediction of small therapeutic molecules", 《REPRODUCTIVE BIOLOGY AND ENDOCRINOLOGY》 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115407067A (en) * | 2022-06-22 | 2022-11-29 | 郑州大学第一附属医院 | Sepsis diagnostic marker |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Caballero-Campo et al. | Hormonal and embryonic regulation of chemokines IL-8, MCP-1 and RANTES in the human endometrium during the window of implantation | |
| Nandula et al. | Role of Canagliflozin on function of CD34+ ve endothelial progenitor cells (EPC) in patients with type 2 diabetes | |
| Maurice et al. | Expression of the thioredoxin–thioredoxin reductase system in the inflamed joints of patients with rheumatoid arthritis | |
| Dos Santos et al. | Adiponectin and leptin systems in human endometrium during window of implantation | |
| Morgan et al. | Visfatin expression is elevated in normal human pregnancy | |
| Ranieri et al. | Urinary IL-6/EGF ratio: a useful prognostic marker for the progression of renal damage in IgA nephropathy | |
| Kleiblova et al. | Expression of adipokines and estrogen receptors in adipose tissue and placenta of patients with gestational diabetes mellitus | |
| Nemeth et al. | Fetal membrane distention: II. Differentially expressed genes regulated by acute distention in vitro | |
| KR102118631B1 (en) | Composition for diagnosing arthritis and kit comprising the same | |
| CN109666739B (en) | A serum/plasma LncRNA marker combination related to the auxiliary diagnosis of intrahepatic cholestasis of pregnancy and its application | |
| Choi et al. | Transcriptional profiling of human femoral mesenchymal stem cells in osteoporosis and its association with adipogenesis | |
| KR100752986B1 (en) | A method of identifying an eventual modification of at least one biological parameter implementing young and aged living cells | |
| Gan et al. | LncRNA MAAMT facilitates macrophage recruitment and proinflammatory activation and exacerbates autoimmune myocarditis through the SRSF1/NF-κB axis | |
| CN113186275A (en) | Application of serum amyloid A1 in preparation of biomarker for diagnosing polycystic ovarian syndrome | |
| CN109868313A (en) | Application of the HMGA2 gene in Stein-Leventhal syndrome disease | |
| Zhang et al. | Decreased CD56+ CD16‐CD94+ uNK cells in the mid‐luteal phase in women with recurrent implantation failure are associated with IL‐15 deficiency | |
| CN118308475B (en) | Application of HTR1B as a diagnostic marker and therapeutic target for embryonic developmental arrest | |
| Golden et al. | Extracellular Vesicles Alter Trophoblast Function in Pregnancies Complicated by COVID‐19 | |
| CN117683883B (en) | Application of miR-126-5p in the diagnosis and treatment of unexplained recurrent spontaneous abortion | |
| Kha et al. | Injured proximal tubular epithelial cells lose hepatocyte nuclear factor 4α expression crucial for brush border formation and transport | |
| Byford et al. | Endothelial‐to‐mesenchymal transition in the fetoplacental macrovasculature and microvasculature in pregnancies complicated by gestational diabetes | |
| CN109097358B (en) | Application of lncRNA in prevention or treatment of hypertension | |
| Quezada et al. | Proenkephalin A and the γ-aminobutyric acid A receptor π subunit: expression, localization, and dynamic changes in human secretory endometrium | |
| CN110564846B (en) | TYW3 for diagnosing male osteoporosis | |
| CN101517096A (en) | Application of DKK-1 protein in cancer diagnosis |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20210730 |
|
| RJ01 | Rejection of invention patent application after publication |