CN113144273A - Drive response composite material and preparation method and application thereof - Google Patents
Drive response composite material and preparation method and application thereof Download PDFInfo
- Publication number
- CN113144273A CN113144273A CN202110338133.XA CN202110338133A CN113144273A CN 113144273 A CN113144273 A CN 113144273A CN 202110338133 A CN202110338133 A CN 202110338133A CN 113144273 A CN113144273 A CN 113144273A
- Authority
- CN
- China
- Prior art keywords
- molecular polymer
- gel
- high molecular
- layer
- nanofiber
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000002131 composite material Substances 0.000 title claims abstract description 60
- 230000004044 response Effects 0.000 title claims abstract description 37
- 238000002360 preparation method Methods 0.000 title abstract description 17
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims abstract description 82
- 239000002121 nanofiber Substances 0.000 claims abstract description 78
- 239000000126 substance Substances 0.000 claims abstract description 45
- 229920000642 polymer Polymers 0.000 claims abstract description 40
- 238000009987 spinning Methods 0.000 claims abstract description 39
- 239000000243 solution Substances 0.000 claims abstract description 32
- 238000000034 method Methods 0.000 claims abstract description 28
- 238000010041 electrostatic spinning Methods 0.000 claims abstract description 26
- 230000008569 process Effects 0.000 claims abstract description 20
- 239000011248 coating agent Substances 0.000 claims abstract description 19
- 238000000576 coating method Methods 0.000 claims abstract description 19
- 239000007864 aqueous solution Substances 0.000 claims abstract description 15
- 230000002209 hydrophobic effect Effects 0.000 claims abstract description 14
- 239000000463 material Substances 0.000 claims abstract description 12
- 239000002994 raw material Substances 0.000 claims abstract description 9
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 30
- 239000003431 cross linking reagent Substances 0.000 claims description 25
- 239000012528 membrane Substances 0.000 claims description 18
- 229920001606 poly(lactic acid-co-glycolic acid) Polymers 0.000 claims description 15
- 108010022355 Fibroins Proteins 0.000 claims description 14
- 238000004132 cross linking Methods 0.000 claims description 13
- 238000001035 drying Methods 0.000 claims description 13
- 239000003102 growth factor Substances 0.000 claims description 11
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 claims description 10
- 229920002674 hyaluronan Polymers 0.000 claims description 10
- 229960003160 hyaluronic acid Drugs 0.000 claims description 10
- -1 alginic acid polysaccharide Chemical class 0.000 claims description 9
- 238000006243 chemical reaction Methods 0.000 claims description 9
- BYEAHWXPCBROCE-UHFFFAOYSA-N 1,1,1,3,3,3-hexafluoropropan-2-ol Chemical compound FC(F)(F)C(O)C(F)(F)F BYEAHWXPCBROCE-UHFFFAOYSA-N 0.000 claims description 8
- 229920000615 alginic acid Polymers 0.000 claims description 8
- 239000000783 alginic acid Substances 0.000 claims description 8
- 235000010443 alginic acid Nutrition 0.000 claims description 8
- 229960001126 alginic acid Drugs 0.000 claims description 8
- 229920001282 polysaccharide Polymers 0.000 claims description 8
- 239000005017 polysaccharide Substances 0.000 claims description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 8
- 108010035532 Collagen Proteins 0.000 claims description 7
- 102000008186 Collagen Human genes 0.000 claims description 7
- 229920001436 collagen Polymers 0.000 claims description 7
- 239000008367 deionised water Substances 0.000 claims description 7
- 229910021641 deionized water Inorganic materials 0.000 claims description 7
- 238000003756 stirring Methods 0.000 claims description 6
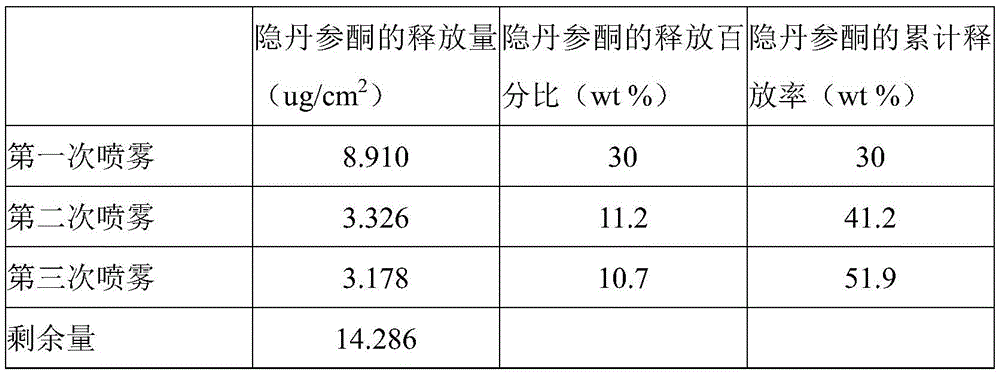
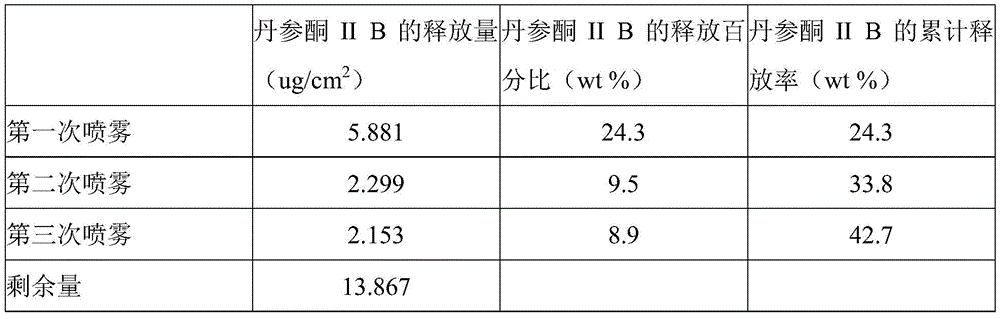
- 241000304195 Salvia miltiorrhiza Species 0.000 claims description 4
- 235000011135 Salvia miltiorrhiza Nutrition 0.000 claims description 4
- IDGUHHHQCWSQLU-UHFFFAOYSA-N ethanol;hydrate Chemical compound O.CCO IDGUHHHQCWSQLU-UHFFFAOYSA-N 0.000 claims description 4
- 238000011068 loading method Methods 0.000 claims description 4
- 229940124350 antibacterial drug Drugs 0.000 claims description 2
- 239000001110 calcium chloride Substances 0.000 claims description 2
- 229910001628 calcium chloride Inorganic materials 0.000 claims description 2
- 239000003398 denaturant Substances 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims description 2
- 239000003607 modifier Substances 0.000 claims description 2
- 239000000178 monomer Substances 0.000 claims description 2
- 239000002904 solvent Substances 0.000 claims description 2
- 229920001600 hydrophobic polymer Polymers 0.000 claims 1
- 206010052428 Wound Diseases 0.000 abstract description 46
- 208000027418 Wounds and injury Diseases 0.000 abstract description 46
- 239000012466 permeate Substances 0.000 abstract description 5
- 230000000844 anti-bacterial effect Effects 0.000 abstract description 4
- 230000003110 anti-inflammatory effect Effects 0.000 abstract description 4
- 230000029663 wound healing Effects 0.000 abstract description 3
- 230000001737 promoting effect Effects 0.000 abstract description 2
- 239000000499 gel Substances 0.000 description 65
- 238000005507 spraying Methods 0.000 description 37
- 238000012360 testing method Methods 0.000 description 35
- GVKKJJOMQCNPGB-JTQLQIEISA-N Cryptotanshinone Chemical compound O=C1C(=O)C2=C3CCCC(C)(C)C3=CC=C2C2=C1[C@@H](C)CO2 GVKKJJOMQCNPGB-JTQLQIEISA-N 0.000 description 29
- GVKKJJOMQCNPGB-UHFFFAOYSA-N Cryptotanshinone Natural products O=C1C(=O)C2=C3CCCC(C)(C)C3=CC=C2C2=C1C(C)CO2 GVKKJJOMQCNPGB-UHFFFAOYSA-N 0.000 description 29
- 239000000835 fiber Substances 0.000 description 27
- 239000003814 drug Substances 0.000 description 16
- 229940079593 drug Drugs 0.000 description 11
- 102000005789 Vascular Endothelial Growth Factors Human genes 0.000 description 8
- 108010019530 Vascular Endothelial Growth Factors Proteins 0.000 description 8
- XDUXBBDRILEIEZ-UHFFFAOYSA-N 6-(hydroxymethyl)-1,6-dimethyl-8,9-dihydro-7h-naphtho[1,2-g][1]benzofuran-10,11-dione Chemical compound O=C1C(=O)C2=C3CCCC(C)(CO)C3=CC=C2C2=C1C(C)=CO2 XDUXBBDRILEIEZ-UHFFFAOYSA-N 0.000 description 7
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 7
- 230000009471 action Effects 0.000 description 6
- 229920000747 poly(lactic acid) Polymers 0.000 description 5
- 239000004626 polylactic acid Substances 0.000 description 5
- 238000009423 ventilation Methods 0.000 description 5
- 108010010803 Gelatin Proteins 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 229920000159 gelatin Polymers 0.000 description 4
- 239000008273 gelatin Substances 0.000 description 4
- 235000019322 gelatine Nutrition 0.000 description 4
- 235000011852 gelatine desserts Nutrition 0.000 description 4
- 239000000017 hydrogel Substances 0.000 description 4
- 229920000954 Polyglycolide Polymers 0.000 description 3
- 229920001610 polycaprolactone Polymers 0.000 description 3
- HYXITZLLTYIPOF-UHFFFAOYSA-N 1,6,6-trimethyl-8,9-dihydro-7H-naphtho[1,2-g]benzofuran-10,11-dione Chemical compound O=C1C(=O)C2=C3CCCC(C)(C)C3=CC=C2C2=C1C(C)=CO2 HYXITZLLTYIPOF-UHFFFAOYSA-N 0.000 description 2
- 230000006378 damage Effects 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- JVTAAEKCZFNVCJ-REOHCLBHSA-N L-lactic acid Chemical compound C[C@H](O)C(O)=O JVTAAEKCZFNVCJ-REOHCLBHSA-N 0.000 description 1
- 208000034530 PLAA-associated neurodevelopmental disease Diseases 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000003115 biocidal effect Effects 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 210000000416 exudates and transudate Anatomy 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- JEGUKCSWCFPDGT-UHFFFAOYSA-N h2o hydrate Chemical compound O.O JEGUKCSWCFPDGT-UHFFFAOYSA-N 0.000 description 1
- 230000035876 healing Effects 0.000 description 1
- 229920001477 hydrophilic polymer Polymers 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 230000028709 inflammatory response Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 230000003020 moisturizing effect Effects 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- 229920001432 poly(L-lactide) Polymers 0.000 description 1
- 239000004632 polycaprolactone Substances 0.000 description 1
- 239000004633 polyglycolic acid Substances 0.000 description 1
- 229920005594 polymer fiber Polymers 0.000 description 1
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 1
- 239000004810 polytetrafluoroethylene Substances 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000004321 preservation Methods 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 238000002791 soaking Methods 0.000 description 1
- 230000017423 tissue regeneration Effects 0.000 description 1
- 238000001291 vacuum drying Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/40—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons containing ingredients of undetermined constitution or reaction products thereof, e.g. plant or animal extracts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/22—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons containing macromolecular materials
- A61L15/26—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/22—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons containing macromolecular materials
- A61L15/28—Polysaccharides or their derivatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/22—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons containing macromolecular materials
- A61L15/32—Proteins, polypeptides; Degradation products or derivatives thereof, e.g. albumin, collagen, fibrin, gelatin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/42—Use of materials characterised by their function or physical properties
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/42—Use of materials characterised by their function or physical properties
- A61L15/44—Medicaments
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/42—Use of materials characterised by their function or physical properties
- A61L15/46—Deodorants or malodour counteractants, e.g. to inhibit the formation of ammonia or bacteria
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/42—Use of materials characterised by their function or physical properties
- A61L15/62—Compostable, hydrosoluble or hydrodegradable materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/20—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices containing or releasing organic materials
- A61L2300/30—Compounds of undetermined constitution extracted from natural sources, e.g. Aloe Vera
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/40—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices characterised by a specific therapeutic activity or mode of action
- A61L2300/404—Biocides, antimicrobial agents, antiseptic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/40—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices characterised by a specific therapeutic activity or mode of action
- A61L2300/41—Anti-inflammatory agents, e.g. NSAIDs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/40—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices characterised by a specific therapeutic activity or mode of action
- A61L2300/412—Tissue-regenerating or healing or proliferative agents
- A61L2300/414—Growth factors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2400/00—Materials characterised by their function or physical properties
- A61L2400/12—Nanosized materials, e.g. nanofibres, nanoparticles, nanowires, nanotubes; Nanostructured surfaces
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Epidemiology (AREA)
- Hematology (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Zoology (AREA)
- Botany (AREA)
- Materials For Medical Uses (AREA)
Abstract
Description
Claims (10)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110338133.XA CN113144273B (en) | 2021-03-30 | 2021-03-30 | Drive response composite material and preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110338133.XA CN113144273B (en) | 2021-03-30 | 2021-03-30 | Drive response composite material and preparation method and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN113144273A true CN113144273A (en) | 2021-07-23 |
| CN113144273B CN113144273B (en) | 2022-08-26 |
Family
ID=76885245
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110338133.XA Active CN113144273B (en) | 2021-03-30 | 2021-03-30 | Drive response composite material and preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113144273B (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114028603A (en) * | 2021-11-02 | 2022-02-11 | 太原理工大学 | Double-layer asymmetric dressing for promoting skin wound repair and preparation method thereof |
| CN116876108A (en) * | 2023-07-07 | 2023-10-13 | 江西省纳米技术研究院 | Multifunctional coaxial composite fiber, its preparation method and application |
Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102600018A (en) * | 2012-03-31 | 2012-07-25 | 华南理工大学 | Medical dressing with cooling function and wound healing prompting function and preparation method of medical dressing |
| US20130150763A1 (en) * | 2011-12-07 | 2013-06-13 | Esmaeil Mirzaei | Electro spun nanofibrous wound dressing and a method of synthesizing the same |
| CN103751851A (en) * | 2014-01-17 | 2014-04-30 | 东华大学 | Preparation method of inorganic/organic multi-drug controlled release composite nano fiber scaffold |
| WO2014182536A1 (en) * | 2013-05-10 | 2014-11-13 | Biovation Ii, Llc | Biopolymer multi-layer multi-functional medical dressing and method of making same |
| CN104491914A (en) * | 2014-12-25 | 2015-04-08 | 中国人民解放军第四军医大学 | Porous complex gel-nanofiber oxygen permeation dressing and preparation method thereof |
| KR20150084519A (en) * | 2014-01-14 | 2015-07-22 | 연세대학교 산학협력단 | Multi-layered electrospun fiber incorporated hydrogel |
| CN106676753A (en) * | 2016-12-28 | 2017-05-17 | 嘉兴学院 | Antibacterial nano fiber and preparation method thereof |
| CN107519524A (en) * | 2017-09-21 | 2017-12-29 | 滨州医学院 | A kind of polycaprolactone/collagen/quaternary ammonium salt electrospun composite fibers film and preparation method thereof |
| CN108042838A (en) * | 2017-12-20 | 2018-05-18 | 华中科技大学同济医学院附属协和医院 | A kind of preparation method of anti-oxidative nanofiber electrospinning film medical dressing |
| CN109529123A (en) * | 2018-11-08 | 2019-03-29 | 中国人民解放军第四军医大学 | The vascularization holostrome organization engineering skin and preparation method thereof that hydrogel, nano fiber scaffold and Skin Cell assemble layer by layer |
| KR20190060326A (en) * | 2017-11-24 | 2019-06-03 | 연세대학교 산학협력단 | Multifunctional nanofiber scaffolds and preparation method thereof |
| CN110876711A (en) * | 2019-12-11 | 2020-03-13 | 北京化工大学 | A kind of method for preparing multilayer polymer microneedle |
| CN111001028A (en) * | 2019-12-29 | 2020-04-14 | 北京工商大学 | Methylene blue antioxidant repair polymer nanofiber membrane and preparation method thereof |
-
2021
- 2021-03-30 CN CN202110338133.XA patent/CN113144273B/en active Active
Patent Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20130150763A1 (en) * | 2011-12-07 | 2013-06-13 | Esmaeil Mirzaei | Electro spun nanofibrous wound dressing and a method of synthesizing the same |
| CN102600018A (en) * | 2012-03-31 | 2012-07-25 | 华南理工大学 | Medical dressing with cooling function and wound healing prompting function and preparation method of medical dressing |
| WO2014182536A1 (en) * | 2013-05-10 | 2014-11-13 | Biovation Ii, Llc | Biopolymer multi-layer multi-functional medical dressing and method of making same |
| KR20150084519A (en) * | 2014-01-14 | 2015-07-22 | 연세대학교 산학협력단 | Multi-layered electrospun fiber incorporated hydrogel |
| CN103751851A (en) * | 2014-01-17 | 2014-04-30 | 东华大学 | Preparation method of inorganic/organic multi-drug controlled release composite nano fiber scaffold |
| CN104491914A (en) * | 2014-12-25 | 2015-04-08 | 中国人民解放军第四军医大学 | Porous complex gel-nanofiber oxygen permeation dressing and preparation method thereof |
| CN106676753A (en) * | 2016-12-28 | 2017-05-17 | 嘉兴学院 | Antibacterial nano fiber and preparation method thereof |
| CN107519524A (en) * | 2017-09-21 | 2017-12-29 | 滨州医学院 | A kind of polycaprolactone/collagen/quaternary ammonium salt electrospun composite fibers film and preparation method thereof |
| KR20190060326A (en) * | 2017-11-24 | 2019-06-03 | 연세대학교 산학협력단 | Multifunctional nanofiber scaffolds and preparation method thereof |
| CN108042838A (en) * | 2017-12-20 | 2018-05-18 | 华中科技大学同济医学院附属协和医院 | A kind of preparation method of anti-oxidative nanofiber electrospinning film medical dressing |
| CN109529123A (en) * | 2018-11-08 | 2019-03-29 | 中国人民解放军第四军医大学 | The vascularization holostrome organization engineering skin and preparation method thereof that hydrogel, nano fiber scaffold and Skin Cell assemble layer by layer |
| CN110876711A (en) * | 2019-12-11 | 2020-03-13 | 北京化工大学 | A kind of method for preparing multilayer polymer microneedle |
| CN111001028A (en) * | 2019-12-29 | 2020-04-14 | 北京工商大学 | Methylene blue antioxidant repair polymer nanofiber membrane and preparation method thereof |
Non-Patent Citations (4)
| Title |
|---|
| ASGHAR ESKANDARINIA等: "A novel Bilayer Wound Dressing composed of a Dense polyurethane/propolis Membrane and a Biodegradable polycaprolactone/Gelatin Nanofbrous Scafold", 《SCIENTIFIC REPORTS》 * |
| CHENG-HUNG LEE等: "Nanofibrous rhPDGF-eluting PLGA–collagen hybrid scaffolds enhance healing of diabetic wounds", 《RSC ADV.》 * |
| YUAN LIAN等: "Fabrication and characterization of curcumin-loaded silk fibroin/P(LLA-CL) nanofibrous scaffold", 《FRONT. MATER. SCI.》 * |
| 石锐等: "PCL基电纺纤维膜的抑菌、相容及降解性能研究", 《生物骨科材料与临床研究》 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114028603A (en) * | 2021-11-02 | 2022-02-11 | 太原理工大学 | Double-layer asymmetric dressing for promoting skin wound repair and preparation method thereof |
| CN116876108A (en) * | 2023-07-07 | 2023-10-13 | 江西省纳米技术研究院 | Multifunctional coaxial composite fiber, its preparation method and application |
Also Published As
| Publication number | Publication date |
|---|---|
| CN113144273B (en) | 2022-08-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Heydari et al. | Preparation and evaluation of poly glycerol sebacate/poly hydroxy butyrate core‐shell electrospun nanofibers with sequentially release of ciprofloxacin and simvastatin in wound dressings | |
| CN102068339B (en) | Preparation method of biodegradable nanofiber medical dressing loaded with medicine | |
| CN100577720C (en) | Biodegradable and absorbable polymer nanofiber membrane material and its preparation method and use | |
| CN103394114B (en) | A kind of preparation method of medical dressing chitosan-based superfine fiber carrier material | |
| CN111588900B (en) | Waterproof, breathable, high-elasticity, self-repairing double-layer nanofiber membrane for skin dressing and its preparation method | |
| CN1961974B (en) | Biodegradable and absorbable polymer nanofiber membrane material and its preparation method and use | |
| Nur et al. | Recent advances in bioactive wound dressings | |
| CN103611182A (en) | Preparation method of core-shell structure superfine fiber carrier material for medical dressing | |
| Zhang et al. | Recent progress of electrospun nanofibers as burning dressings | |
| CN102266582A (en) | Preparation method for drug-loaded nanometer fiber medical dressing | |
| EP3658197B1 (en) | Electrospun nanofibers and membrane | |
| KR20140090704A (en) | Artificial dura mater and manufacturing method thereof | |
| CN103933602A (en) | Preparation method of chitosan-based drug-loading composite antibacterial superfine fiber membrane | |
| Coelho et al. | Electrospinning technology: designing nanofibers toward wound healing application | |
| WO2020186715A1 (en) | Drug-loaded nanofiber film and preparation method and application thereof | |
| Abdelhakeem et al. | State-of-the-art review of advanced electrospun nanofiber composites for enhanced wound healing | |
| CN113144273A (en) | Drive response composite material and preparation method and application thereof | |
| Tamilarasi et al. | Electrospun scaffold-based antibiotic therapeutics for chronic wound recovery | |
| Wu et al. | Electrospun nanofiber scaffold for skin tissue engineering: a review | |
| CN115058892A (en) | A kind of lactic acid porous fiber membrane and its application | |
| Plascencia Martinez et al. | Comparative study of single and coaxial electrospun antimicrobial cross-linked scaffolds enriched with Aloe Vera: Characterization, antimicrobial activity, drug delivery, cytotoxicity, and cell proliferation on adipose stem cells and human skin fibroblast | |
| Rajchal et al. | Insights into electrospun polymeric nanofiber mats: an innovative dressing for wound healing applications | |
| CN117860945A (en) | Sandwich-like structure drug-carrying micro-nano fiber composite film medical dressing and preparation method thereof | |
| CN103263381B (en) | Fiber type controllable drug sustained release system and preparation method thereof | |
| CN112353780A (en) | Drug sustained and controlled release platform system with double nano composite structures |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| CB03 | Change of inventor or designer information |
Inventor after: Yin Anlin Inventor after: Sun Wenling Inventor after: Zhang Kuihua Inventor after: Li Zhe Inventor after: Li Yunhuan Inventor after: Fan Yongyong Inventor before: Yin Anlin Inventor before: Zhang Kuihua Inventor before: Li Zhe Inventor before: Li Yunhuan Inventor before: Fan Yongyong |
|
| CB03 | Change of inventor or designer information | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CP03 | Change of name, title or address |
Address after: 314000 No. 899, guangqiong Road, Nanhu District, Jiaxing City, Zhejiang Province Patentee after: Jiaxing University Country or region after: China Address before: No. 899 Guangqiong Road, Nanhu District, Jiaxing City, Zhejiang Province Patentee before: JIAXING University Country or region before: China |
|
| CP03 | Change of name, title or address |