CN112898286A - Benzothiophene compound or pharmaceutically acceptable salt and isomer thereof, and preparation method, pharmaceutical composition and application thereof - Google Patents
Benzothiophene compound or pharmaceutically acceptable salt and isomer thereof, and preparation method, pharmaceutical composition and application thereof Download PDFInfo
- Publication number
- CN112898286A CN112898286A CN202110117453.2A CN202110117453A CN112898286A CN 112898286 A CN112898286 A CN 112898286A CN 202110117453 A CN202110117453 A CN 202110117453A CN 112898286 A CN112898286 A CN 112898286A
- Authority
- CN
- China
- Prior art keywords
- alkyl
- substituted
- unsubstituted
- hydrogen
- group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- -1 Benzothiophene compound Chemical class 0.000 title claims abstract description 119
- 150000003839 salts Chemical class 0.000 title claims abstract description 53
- 239000008194 pharmaceutical composition Substances 0.000 title claims abstract description 25
- FCEHBMOGCRZNNI-UHFFFAOYSA-N thianaphthalene Natural products C1=CC=C2SC=CC2=C1 FCEHBMOGCRZNNI-UHFFFAOYSA-N 0.000 title claims abstract description 25
- 238000002360 preparation method Methods 0.000 title claims abstract description 13
- 108010050904 Interferons Proteins 0.000 claims abstract description 33
- 206010028980 Neoplasm Diseases 0.000 claims abstract description 16
- 239000003814 drug Substances 0.000 claims abstract description 15
- 208000035473 Communicable disease Diseases 0.000 claims abstract description 7
- 230000004936 stimulating effect Effects 0.000 claims abstract 4
- 125000000217 alkyl group Chemical group 0.000 claims description 273
- 150000001875 compounds Chemical class 0.000 claims description 232
- 229910052739 hydrogen Inorganic materials 0.000 claims description 190
- 239000001257 hydrogen Substances 0.000 claims description 190
- 125000005842 heteroatom Chemical group 0.000 claims description 175
- 229910052736 halogen Inorganic materials 0.000 claims description 130
- 150000002367 halogens Chemical class 0.000 claims description 130
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 123
- 230000015572 biosynthetic process Effects 0.000 claims description 116
- 238000003786 synthesis reaction Methods 0.000 claims description 116
- 229910052760 oxygen Inorganic materials 0.000 claims description 88
- 229910052717 sulfur Inorganic materials 0.000 claims description 86
- 125000003545 alkoxy group Chemical group 0.000 claims description 77
- 150000002431 hydrogen Chemical class 0.000 claims description 60
- 125000006376 (C3-C10) cycloalkyl group Chemical group 0.000 claims description 53
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 45
- 239000002207 metabolite Substances 0.000 claims description 42
- 125000001313 C5-C10 heteroaryl group Chemical group 0.000 claims description 41
- 125000006719 (C6-C10) aryl (C1-C6) alkyl group Chemical group 0.000 claims description 40
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 39
- 229940002612 prodrug Drugs 0.000 claims description 39
- 239000000651 prodrug Substances 0.000 claims description 39
- 239000012453 solvate Substances 0.000 claims description 34
- 125000000592 heterocycloalkyl group Chemical group 0.000 claims description 31
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 claims description 30
- 125000001072 heteroaryl group Chemical group 0.000 claims description 27
- 239000002904 solvent Substances 0.000 claims description 25
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 24
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 24
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 24
- 125000003118 aryl group Chemical group 0.000 claims description 22
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 22
- 239000000460 chlorine Substances 0.000 claims description 19
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 claims description 18
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 claims description 18
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 claims description 18
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 claims description 18
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 claims description 18
- 229910052794 bromium Inorganic materials 0.000 claims description 18
- 229910052801 chlorine Inorganic materials 0.000 claims description 18
- 239000011737 fluorine Substances 0.000 claims description 18
- 229910052731 fluorine Inorganic materials 0.000 claims description 18
- 239000011630 iodine Substances 0.000 claims description 18
- 229910052740 iodine Inorganic materials 0.000 claims description 18
- 125000004432 carbon atom Chemical group C* 0.000 claims description 16
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 claims description 16
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 16
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 16
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 16
- 125000004737 (C1-C6) haloalkoxy group Chemical group 0.000 claims description 14
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 14
- 238000000034 method Methods 0.000 claims description 14
- 125000005553 heteroaryloxy group Chemical group 0.000 claims description 13
- 125000003342 alkenyl group Chemical group 0.000 claims description 11
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 claims description 10
- 229910052805 deuterium Inorganic materials 0.000 claims description 10
- 125000004767 (C1-C4) haloalkoxy group Chemical group 0.000 claims description 8
- 125000004765 (C1-C4) haloalkyl group Chemical group 0.000 claims description 8
- 125000000171 (C1-C6) haloalkyl group Chemical group 0.000 claims description 8
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 claims description 8
- 229940076376 protein agonist Drugs 0.000 claims description 8
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 claims description 6
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims description 6
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 claims description 6
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 claims description 6
- 150000004677 hydrates Chemical class 0.000 claims description 6
- 229910052799 carbon Inorganic materials 0.000 claims description 5
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 4
- 125000004850 cyclobutylmethyl group Chemical group C1(CCC1)C* 0.000 claims description 4
- 125000004210 cyclohexylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H] 0.000 claims description 4
- 125000004851 cyclopentylmethyl group Chemical group C1(CCCC1)C* 0.000 claims description 4
- 125000004186 cyclopropylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C1([H])[H] 0.000 claims description 4
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 4
- 125000004213 tert-butoxy group Chemical group [H]C([H])([H])C(O*)(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 4
- 125000005301 thienylmethyl group Chemical group [H]C1=C([H])C([H])=C(S1)C([H])([H])* 0.000 claims description 4
- 229940124931 vaccine adjuvant Drugs 0.000 claims description 4
- 239000012646 vaccine adjuvant Substances 0.000 claims description 4
- 125000006570 (C5-C6) heteroaryl group Chemical group 0.000 claims description 2
- DQXKOHDUMJLXKH-PHEQNACWSA-N (e)-n-[2-[2-[[(e)-oct-2-enoyl]amino]ethyldisulfanyl]ethyl]oct-2-enamide Chemical compound CCCCC\C=C\C(=O)NCCSSCCNC(=O)\C=C\CCCCC DQXKOHDUMJLXKH-PHEQNACWSA-N 0.000 claims description 2
- 125000004504 1,2,4-oxadiazolyl group Chemical group 0.000 claims description 2
- 125000004514 1,2,4-thiadiazolyl group Chemical group 0.000 claims description 2
- 125000001781 1,3,4-oxadiazolyl group Chemical group 0.000 claims description 2
- 125000004520 1,3,4-thiadiazolyl group Chemical group 0.000 claims description 2
- XQQZRZQVBFHBHL-UHFFFAOYSA-N 12-crown-4 Chemical compound C1COCCOCCOCCO1 XQQZRZQVBFHBHL-UHFFFAOYSA-N 0.000 claims description 2
- VFTFKUDGYRBSAL-UHFFFAOYSA-N 15-crown-5 Chemical compound C1COCCOCCOCCOCCO1 VFTFKUDGYRBSAL-UHFFFAOYSA-N 0.000 claims description 2
- XEZNGIUYQVAUSS-UHFFFAOYSA-N 18-crown-6 Chemical compound C1COCCOCCOCCOCCOCCO1 XEZNGIUYQVAUSS-UHFFFAOYSA-N 0.000 claims description 2
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 claims description 2
- FGUUSXIOTUKUDN-IBGZPJMESA-N C1(=CC=CC=C1)N1C2=C(NC([C@H](C1)NC=1OC(=NN=1)C1=CC=CC=C1)=O)C=CC=C2 Chemical compound C1(=CC=CC=C1)N1C2=C(NC([C@H](C1)NC=1OC(=NN=1)C1=CC=CC=C1)=O)C=CC=C2 FGUUSXIOTUKUDN-IBGZPJMESA-N 0.000 claims description 2
- 125000003601 C2-C6 alkynyl group Chemical group 0.000 claims description 2
- 239000002671 adjuvant Substances 0.000 claims description 2
- 125000000258 cyclohexylethoxy group Chemical group [H]C([H])(O*)C([H])([H])C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H] 0.000 claims description 2
- 125000002933 cyclohexyloxy group Chemical group C1(CCCCC1)O* 0.000 claims description 2
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 claims description 2
- 125000003253 isopropoxy group Chemical group [H]C([H])([H])C([H])(O*)C([H])([H])[H] 0.000 claims description 2
- 125000006203 morpholinoethyl group Chemical group [H]C([H])(*)C([H])([H])N1C([H])([H])C([H])([H])OC([H])([H])C1([H])[H] 0.000 claims description 2
- 125000001064 morpholinomethyl group Chemical group [H]C([H])(*)N1C([H])([H])C([H])([H])OC([H])([H])C1([H])[H] 0.000 claims description 2
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 claims description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 2
- LFGREXWGYUGZLY-UHFFFAOYSA-N phosphoryl Chemical group [P]=O LFGREXWGYUGZLY-UHFFFAOYSA-N 0.000 claims description 2
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 2
- 125000005554 pyridyloxy group Chemical group 0.000 claims description 2
- 125000001424 substituent group Chemical group 0.000 claims description 2
- 125000000876 trifluoromethoxy group Chemical group FC(F)(F)O* 0.000 claims description 2
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 2
- 125000000753 cycloalkyl group Chemical group 0.000 claims 9
- 150000003254 radicals Chemical class 0.000 claims 4
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 claims 2
- 125000004438 haloalkoxy group Chemical group 0.000 claims 2
- 230000002265 prevention Effects 0.000 claims 2
- 125000005344 pyridylmethyl group Chemical group [H]C1=C([H])C([H])=C([H])C(=N1)C([H])([H])* 0.000 claims 2
- 125000005302 thiazolylmethyl group Chemical group [H]C1=C([H])N=C(S1)C([H])([H])* 0.000 claims 2
- WNXJIVFYUVYPPR-UHFFFAOYSA-N 1,3-dioxolane Chemical compound C1COCO1 WNXJIVFYUVYPPR-UHFFFAOYSA-N 0.000 claims 1
- FCSKOFQQCWLGMV-UHFFFAOYSA-N 5-{5-[2-chloro-4-(4,5-dihydro-1,3-oxazol-2-yl)phenoxy]pentyl}-3-methylisoxazole Chemical compound O1N=C(C)C=C1CCCCCOC1=CC=C(C=2OCCN=2)C=C1Cl FCSKOFQQCWLGMV-UHFFFAOYSA-N 0.000 claims 1
- FKLJPTJMIBLJAV-UHFFFAOYSA-N Compound IV Chemical compound O1N=C(C)C=C1CCCCCCCOC1=CC=C(C=2OCCN=2)C=C1 FKLJPTJMIBLJAV-UHFFFAOYSA-N 0.000 claims 1
- 125000005343 heterocyclic alkyl group Chemical group 0.000 claims 1
- 108090000623 proteins and genes Proteins 0.000 abstract description 25
- 102000004169 proteins and genes Human genes 0.000 abstract description 24
- 230000000694 effects Effects 0.000 abstract description 18
- 230000007547 defect Effects 0.000 abstract description 4
- 229940079593 drug Drugs 0.000 abstract description 3
- 239000002773 nucleotide Substances 0.000 abstract description 3
- 125000003729 nucleotide group Chemical group 0.000 abstract description 3
- 238000005481 NMR spectroscopy Methods 0.000 description 98
- 238000006243 chemical reaction Methods 0.000 description 68
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 60
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 38
- 239000000243 solution Substances 0.000 description 38
- 102000014150 Interferons Human genes 0.000 description 27
- 229940079322 interferon Drugs 0.000 description 25
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 21
- 238000004440 column chromatography Methods 0.000 description 20
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 18
- 239000012074 organic phase Substances 0.000 description 18
- 229940076838 Immune checkpoint inhibitor Drugs 0.000 description 15
- 102000037984 Inhibitory immune checkpoint proteins Human genes 0.000 description 15
- 108091008026 Inhibitory immune checkpoint proteins Proteins 0.000 description 15
- 239000012274 immune-checkpoint protein inhibitor Substances 0.000 description 15
- 239000000203 mixture Substances 0.000 description 15
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 15
- 239000000126 substance Substances 0.000 description 14
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 12
- 125000006721 (C5-C10) heteroaryl (C1-C6) alkyl group Chemical group 0.000 description 12
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 12
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 12
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 12
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 10
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 10
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 10
- 230000028993 immune response Effects 0.000 description 10
- 239000002994 raw material Substances 0.000 description 10
- SJRJJKPEHAURKC-UHFFFAOYSA-N N-Methylmorpholine Chemical compound CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 description 9
- 210000001744 T-lymphocyte Anatomy 0.000 description 9
- 239000002253 acid Substances 0.000 description 9
- 108020004414 DNA Proteins 0.000 description 8
- 230000002708 enhancing effect Effects 0.000 description 8
- 239000000523 sample Substances 0.000 description 8
- UNILWMWFPHPYOR-KXEYIPSPSA-M 1-[6-[2-[3-[3-[3-[2-[2-[3-[[2-[2-[[(2r)-1-[[2-[[(2r)-1-[3-[2-[2-[3-[[2-(2-amino-2-oxoethoxy)acetyl]amino]propoxy]ethoxy]ethoxy]propylamino]-3-hydroxy-1-oxopropan-2-yl]amino]-2-oxoethyl]amino]-3-[(2r)-2,3-di(hexadecanoyloxy)propyl]sulfanyl-1-oxopropan-2-yl Chemical compound O=C1C(SCCC(=O)NCCCOCCOCCOCCCNC(=O)COCC(=O)N[C@@H](CSC[C@@H](COC(=O)CCCCCCCCCCCCCCC)OC(=O)CCCCCCCCCCCCCCC)C(=O)NCC(=O)N[C@H](CO)C(=O)NCCCOCCOCCOCCCNC(=O)COCC(N)=O)CC(=O)N1CCNC(=O)CCCCCN\1C2=CC=C(S([O-])(=O)=O)C=C2CC/1=C/C=C/C=C/C1=[N+](CC)C2=CC=C(S([O-])(=O)=O)C=C2C1 UNILWMWFPHPYOR-KXEYIPSPSA-M 0.000 description 7
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 7
- 239000012065 filter cake Substances 0.000 description 7
- 244000052769 pathogen Species 0.000 description 7
- 102000005962 receptors Human genes 0.000 description 7
- 108020003175 receptors Proteins 0.000 description 7
- 238000001308 synthesis method Methods 0.000 description 7
- 239000012623 DNA damaging agent Substances 0.000 description 6
- WTDHULULXKLSOZ-UHFFFAOYSA-N Hydroxylamine hydrochloride Chemical compound Cl.ON WTDHULULXKLSOZ-UHFFFAOYSA-N 0.000 description 6
- 230000027455 binding Effects 0.000 description 6
- 210000004027 cell Anatomy 0.000 description 6
- 238000009472 formulation Methods 0.000 description 6
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 6
- 238000002347 injection Methods 0.000 description 6
- 239000007924 injection Substances 0.000 description 6
- 239000000047 product Substances 0.000 description 6
- 238000010791 quenching Methods 0.000 description 6
- 238000010992 reflux Methods 0.000 description 6
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 description 5
- 239000007821 HATU Substances 0.000 description 5
- 239000003795 chemical substances by application Substances 0.000 description 5
- 238000001727 in vivo Methods 0.000 description 5
- 239000003112 inhibitor Substances 0.000 description 5
- 230000015788 innate immune response Effects 0.000 description 5
- 229910052757 nitrogen Inorganic materials 0.000 description 5
- 239000000546 pharmaceutical excipient Substances 0.000 description 5
- SZUVGFMDDVSKSI-WIFOCOSTSA-N (1s,2s,3s,5r)-1-(carboxymethyl)-3,5-bis[(4-phenoxyphenyl)methyl-propylcarbamoyl]cyclopentane-1,2-dicarboxylic acid Chemical compound O=C([C@@H]1[C@@H]([C@](CC(O)=O)([C@H](C(=O)N(CCC)CC=2C=CC(OC=3C=CC=CC=3)=CC=2)C1)C(O)=O)C(O)=O)N(CCC)CC(C=C1)=CC=C1OC1=CC=CC=C1 SZUVGFMDDVSKSI-WIFOCOSTSA-N 0.000 description 4
- QFLWZFQWSBQYPS-AWRAUJHKSA-N (3S)-3-[[(2S)-2-[[(2S)-2-[5-[(3aS,6aR)-2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]pentanoylamino]-3-methylbutanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-4-[1-bis(4-chlorophenoxy)phosphorylbutylamino]-4-oxobutanoic acid Chemical compound CCCC(NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@@H](NC(=O)CCCCC1SC[C@@H]2NC(=O)N[C@H]12)C(C)C)P(=O)(Oc1ccc(Cl)cc1)Oc1ccc(Cl)cc1 QFLWZFQWSBQYPS-AWRAUJHKSA-N 0.000 description 4
- 229940045513 CTLA4 antagonist Drugs 0.000 description 4
- 102100031256 Cyclic GMP-AMP synthase Human genes 0.000 description 4
- 102000004190 Enzymes Human genes 0.000 description 4
- 108090000790 Enzymes Proteins 0.000 description 4
- 239000003153 chemical reaction reagent Substances 0.000 description 4
- 229940126543 compound 14 Drugs 0.000 description 4
- 230000007062 hydrolysis Effects 0.000 description 4
- 238000006460 hydrolysis reaction Methods 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 239000012528 membrane Substances 0.000 description 4
- 239000013642 negative control Substances 0.000 description 4
- 230000001717 pathogenic effect Effects 0.000 description 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 4
- 238000000746 purification Methods 0.000 description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N pyridine Substances C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- 229940124597 therapeutic agent Drugs 0.000 description 4
- 230000001225 therapeutic effect Effects 0.000 description 4
- GHYOCDFICYLMRF-UTIIJYGPSA-N (2S,3R)-N-[(2S)-3-(cyclopenten-1-yl)-1-[(2R)-2-methyloxiran-2-yl]-1-oxopropan-2-yl]-3-hydroxy-3-(4-methoxyphenyl)-2-[[(2S)-2-[(2-morpholin-4-ylacetyl)amino]propanoyl]amino]propanamide Chemical compound C1(=CCCC1)C[C@@H](C(=O)[C@@]1(OC1)C)NC([C@H]([C@@H](C1=CC=C(C=C1)OC)O)NC([C@H](C)NC(CN1CCOCC1)=O)=O)=O GHYOCDFICYLMRF-UTIIJYGPSA-N 0.000 description 3
- XRILCFTWUCUKJR-INFSMZHSSA-N 2'-3'-cGAMP Chemical compound C([C@H]([C@H]1O)O2)OP(O)(=O)O[C@H]3[C@@H](O)[C@H](N4C5=NC=NC(N)=C5N=C4)O[C@@H]3COP(O)(=O)O[C@H]1[C@@H]2N1C=NC2=C1NC(N)=NC2=O XRILCFTWUCUKJR-INFSMZHSSA-N 0.000 description 3
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- OJRUSAPKCPIVBY-KQYNXXCUSA-N C1=NC2=C(N=C(N=C2N1[C@H]3[C@@H]([C@@H]([C@H](O3)COP(=O)(CP(=O)(O)O)O)O)O)I)N Chemical compound C1=NC2=C(N=C(N=C2N1[C@H]3[C@@H]([C@@H]([C@H](O3)COP(=O)(CP(=O)(O)O)O)O)O)I)N OJRUSAPKCPIVBY-KQYNXXCUSA-N 0.000 description 3
- 239000012275 CTLA-4 inhibitor Substances 0.000 description 3
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- 102100034458 Hepatitis A virus cellular receptor 2 Human genes 0.000 description 3
- 101001068133 Homo sapiens Hepatitis A virus cellular receptor 2 Proteins 0.000 description 3
- 101000666896 Homo sapiens V-type immunoglobulin domain-containing suppressor of T-cell activation Proteins 0.000 description 3
- 229940043367 IDO1 inhibitor Drugs 0.000 description 3
- 102100026720 Interferon beta Human genes 0.000 description 3
- 108090000467 Interferon-beta Proteins 0.000 description 3
- 239000012269 PD-1/PD-L1 inhibitor Substances 0.000 description 3
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 3
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 3
- 102100038282 V-type immunoglobulin domain-containing suppressor of T-cell activation Human genes 0.000 description 3
- 239000004480 active ingredient Substances 0.000 description 3
- 239000000556 agonist Substances 0.000 description 3
- 239000007864 aqueous solution Substances 0.000 description 3
- 125000004429 atom Chemical group 0.000 description 3
- RFCBNSCSPXMEBK-INFSMZHSSA-N c-GMP-AMP Chemical compound C([C@H]1O2)OP(O)(=O)O[C@H]3[C@@H](O)[C@H](N4C5=NC=NC(N)=C5N=C4)O[C@@H]3COP(O)(=O)O[C@H]1[C@@H](O)[C@@H]2N1C(N=C(NC2=O)N)=C2N=C1 RFCBNSCSPXMEBK-INFSMZHSSA-N 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 229940125797 compound 12 Drugs 0.000 description 3
- 229940125758 compound 15 Drugs 0.000 description 3
- 229940125782 compound 2 Drugs 0.000 description 3
- 150000001924 cycloalkanes Chemical class 0.000 description 3
- 210000000805 cytoplasm Anatomy 0.000 description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- IJJVMEJXYNJXOJ-UHFFFAOYSA-N fluquinconazole Chemical compound C=1C=C(Cl)C=C(Cl)C=1N1C(=O)C2=CC(F)=CC=C2N=C1N1C=NC=N1 IJJVMEJXYNJXOJ-UHFFFAOYSA-N 0.000 description 3
- 125000000524 functional group Chemical group 0.000 description 3
- 238000011534 incubation Methods 0.000 description 3
- 208000015181 infectious disease Diseases 0.000 description 3
- 229940047124 interferons Drugs 0.000 description 3
- 239000003446 ligand Substances 0.000 description 3
- 238000007911 parenteral administration Methods 0.000 description 3
- 108010089193 pattern recognition receptors Proteins 0.000 description 3
- 102000007863 pattern recognition receptors Human genes 0.000 description 3
- 229940121653 pd-1/pd-l1 inhibitor Drugs 0.000 description 3
- 229940124531 pharmaceutical excipient Drugs 0.000 description 3
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 3
- 230000001105 regulatory effect Effects 0.000 description 3
- 150000003384 small molecules Chemical class 0.000 description 3
- 241000894007 species Species 0.000 description 3
- 239000000725 suspension Substances 0.000 description 3
- 210000004881 tumor cell Anatomy 0.000 description 3
- KZPYGQFFRCFCPP-UHFFFAOYSA-N 1,1'-bis(diphenylphosphino)ferrocene Chemical compound [Fe+2].C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1 KZPYGQFFRCFCPP-UHFFFAOYSA-N 0.000 description 2
- YYROPELSRYBVMQ-UHFFFAOYSA-N 4-toluenesulfonyl chloride Chemical compound CC1=CC=C(S(Cl)(=O)=O)C=C1 YYROPELSRYBVMQ-UHFFFAOYSA-N 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical class [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 2
- 102000003930 C-Type Lectins Human genes 0.000 description 2
- 108090000342 C-Type Lectins Proteins 0.000 description 2
- 101710118064 Cyclic GMP-AMP synthase Proteins 0.000 description 2
- 108030002637 Cyclic GMP-AMP synthases Proteins 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- 238000002965 ELISA Methods 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- 101001011382 Homo sapiens Interferon regulatory factor 3 Proteins 0.000 description 2
- 101001034842 Homo sapiens Interferon-induced transmembrane protein 2 Proteins 0.000 description 2
- 101000665442 Homo sapiens Serine/threonine-protein kinase TBK1 Proteins 0.000 description 2
- 101000643024 Homo sapiens Stimulator of interferon genes protein Proteins 0.000 description 2
- 102000002227 Interferon Type I Human genes 0.000 description 2
- 108010014726 Interferon Type I Proteins 0.000 description 2
- 102100029843 Interferon regulatory factor 3 Human genes 0.000 description 2
- 102100040020 Interferon-induced transmembrane protein 2 Human genes 0.000 description 2
- 102100024064 Interferon-inducible protein AIM2 Human genes 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- 229940125563 LAG3 inhibitor Drugs 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- PCNDJXKNXGMECE-UHFFFAOYSA-N Phenazine Natural products C1=CC=CC2=NC3=CC=CC=C3N=C21 PCNDJXKNXGMECE-UHFFFAOYSA-N 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- KYQCOXFCLRTKLS-UHFFFAOYSA-N Pyrazine Natural products C1=CN=CC=N1 KYQCOXFCLRTKLS-UHFFFAOYSA-N 0.000 description 2
- 102100038192 Serine/threonine-protein kinase TBK1 Human genes 0.000 description 2
- 102100035533 Stimulator of interferon genes protein Human genes 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 229940125555 TIGIT inhibitor Drugs 0.000 description 2
- 102000002689 Toll-like receptor Human genes 0.000 description 2
- 108020000411 Toll-like receptor Proteins 0.000 description 2
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 2
- 238000002835 absorbance Methods 0.000 description 2
- 230000002378 acidificating effect Effects 0.000 description 2
- 230000004913 activation Effects 0.000 description 2
- 230000033289 adaptive immune response Effects 0.000 description 2
- 230000001270 agonistic effect Effects 0.000 description 2
- 230000000259 anti-tumor effect Effects 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 201000011510 cancer Diseases 0.000 description 2
- 229940125773 compound 10 Drugs 0.000 description 2
- 229940125898 compound 5 Drugs 0.000 description 2
- 239000006071 cream Substances 0.000 description 2
- 230000001086 cytosolic effect Effects 0.000 description 2
- 210000004443 dendritic cell Anatomy 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 210000002472 endoplasmic reticulum Anatomy 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 239000000945 filler Substances 0.000 description 2
- 239000000706 filtrate Substances 0.000 description 2
- XZBIXDPGRMLSTC-UHFFFAOYSA-N formohydrazide Chemical compound NNC=O XZBIXDPGRMLSTC-UHFFFAOYSA-N 0.000 description 2
- GNOIPBMMFNIUFM-UHFFFAOYSA-N hexamethylphosphoric triamide Chemical compound CN(C)P(=O)(N(C)C)N(C)C GNOIPBMMFNIUFM-UHFFFAOYSA-N 0.000 description 2
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 2
- 230000003308 immunostimulating effect Effects 0.000 description 2
- 238000009169 immunotherapy Methods 0.000 description 2
- 239000012442 inert solvent Substances 0.000 description 2
- 230000002757 inflammatory effect Effects 0.000 description 2
- 230000000977 initiatory effect Effects 0.000 description 2
- 230000002601 intratumoral effect Effects 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- KQNPFQTWMSNSAP-UHFFFAOYSA-N isobutyric acid Chemical compound CC(C)C(O)=O KQNPFQTWMSNSAP-UHFFFAOYSA-N 0.000 description 2
- ZLVXBBHTMQJRSX-VMGNSXQWSA-N jdtic Chemical compound C1([C@]2(C)CCN(C[C@@H]2C)C[C@H](C(C)C)NC(=O)[C@@H]2NCC3=CC(O)=CC=C3C2)=CC=CC(O)=C1 ZLVXBBHTMQJRSX-VMGNSXQWSA-N 0.000 description 2
- 238000002372 labelling Methods 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 239000012669 liquid formulation Substances 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 230000001404 mediated effect Effects 0.000 description 2
- 201000001441 melanoma Diseases 0.000 description 2
- 230000004060 metabolic process Effects 0.000 description 2
- HNQIVZYLYMDVSB-UHFFFAOYSA-N methanesulfonimidic acid Chemical compound CS(N)(=O)=O HNQIVZYLYMDVSB-UHFFFAOYSA-N 0.000 description 2
- XNGIFLGASWRNHJ-UHFFFAOYSA-N phthalic acid Chemical compound OC(=O)C1=CC=CC=C1C(O)=O XNGIFLGASWRNHJ-UHFFFAOYSA-N 0.000 description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 description 2
- 238000012827 research and development Methods 0.000 description 2
- 238000007789 sealing Methods 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 239000012089 stop solution Substances 0.000 description 2
- TYFQFVWCELRYAO-UHFFFAOYSA-N suberic acid Chemical compound OC(=O)CCCCCCC(O)=O TYFQFVWCELRYAO-UHFFFAOYSA-N 0.000 description 2
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- 230000009466 transformation Effects 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- QBYIENPQHBMVBV-HFEGYEGKSA-N (2R)-2-hydroxy-2-phenylacetic acid Chemical compound O[C@@H](C(O)=O)c1ccccc1.O[C@@H](C(O)=O)c1ccccc1 QBYIENPQHBMVBV-HFEGYEGKSA-N 0.000 description 1
- UVNPEUJXKZFWSJ-LMTQTHQJSA-N (R)-N-[(4S)-8-[6-amino-5-[(3,3-difluoro-2-oxo-1H-pyrrolo[2,3-b]pyridin-4-yl)sulfanyl]pyrazin-2-yl]-2-oxa-8-azaspiro[4.5]decan-4-yl]-2-methylpropane-2-sulfinamide Chemical compound CC(C)(C)[S@@](=O)N[C@@H]1COCC11CCN(CC1)c1cnc(Sc2ccnc3NC(=O)C(F)(F)c23)c(N)n1 UVNPEUJXKZFWSJ-LMTQTHQJSA-N 0.000 description 1
- LMDZBCPBFSXMTL-UHFFFAOYSA-N 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide Substances CCN=C=NCCCN(C)C LMDZBCPBFSXMTL-UHFFFAOYSA-N 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- KAESVJOAVNADME-UHFFFAOYSA-N 1H-pyrrole Natural products C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 1
- VOZKAJLKRJDJLL-UHFFFAOYSA-N 2,4-diaminotoluene Chemical compound CC1=CC=C(N)C=C1N VOZKAJLKRJDJLL-UHFFFAOYSA-N 0.000 description 1
- OBTZDIRUQWFRFZ-UHFFFAOYSA-N 2-(5-methylfuran-2-yl)-n-(4-methylphenyl)quinoline-4-carboxamide Chemical compound O1C(C)=CC=C1C1=CC(C(=O)NC=2C=CC(C)=CC=2)=C(C=CC=C2)C2=N1 OBTZDIRUQWFRFZ-UHFFFAOYSA-N 0.000 description 1
- FALRKNHUBBKYCC-UHFFFAOYSA-N 2-(chloromethyl)pyridine-3-carbonitrile Chemical compound ClCC1=NC=CC=C1C#N FALRKNHUBBKYCC-UHFFFAOYSA-N 0.000 description 1
- REXUYBKPWIPONM-UHFFFAOYSA-N 2-bromoacetonitrile Chemical compound BrCC#N REXUYBKPWIPONM-UHFFFAOYSA-N 0.000 description 1
- FPQQSJJWHUJYPU-UHFFFAOYSA-N 3-(dimethylamino)propyliminomethylidene-ethylazanium;chloride Chemical compound Cl.CCN=C=NCCCN(C)C FPQQSJJWHUJYPU-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- WDBQJSCPCGTAFG-QHCPKHFHSA-N 4,4-difluoro-N-[(1S)-3-[4-(3-methyl-5-propan-2-yl-1,2,4-triazol-4-yl)piperidin-1-yl]-1-pyridin-3-ylpropyl]cyclohexane-1-carboxamide Chemical compound FC1(CCC(CC1)C(=O)N[C@@H](CCN1CCC(CC1)N1C(=NN=C1C)C(C)C)C=1C=NC=CC=1)F WDBQJSCPCGTAFG-QHCPKHFHSA-N 0.000 description 1
- 108060000255 AIM2 Proteins 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 108010074708 B7-H1 Antigen Proteins 0.000 description 1
- 102000008096 B7-H1 Antigen Human genes 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 208000026310 Breast neoplasm Diseases 0.000 description 1
- 102000008203 CTLA-4 Antigen Human genes 0.000 description 1
- 108010021064 CTLA-4 Antigen Proteins 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 241000700198 Cavia Species 0.000 description 1
- 241000282693 Cercopithecidae Species 0.000 description 1
- 206010008342 Cervix carcinoma Diseases 0.000 description 1
- 102000019034 Chemokines Human genes 0.000 description 1
- 108010012236 Chemokines Proteins 0.000 description 1
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 1
- 206010009944 Colon cancer Diseases 0.000 description 1
- AEMOLEFTQBMNLQ-AQKNRBDQSA-N D-glucopyranuronic acid Chemical compound OC1O[C@H](C(O)=O)[C@@H](O)[C@H](O)[C@H]1O AEMOLEFTQBMNLQ-AQKNRBDQSA-N 0.000 description 1
- 102000053602 DNA Human genes 0.000 description 1
- 230000005778 DNA damage Effects 0.000 description 1
- 231100000277 DNA damage Toxicity 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Divinylene sulfide Natural products C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 1
- IAJILQKETJEXLJ-UHFFFAOYSA-N Galacturonsaeure Natural products O=CC(O)C(O)C(O)C(O)C(O)=O IAJILQKETJEXLJ-UHFFFAOYSA-N 0.000 description 1
- 102100039928 Gamma-interferon-inducible protein 16 Human genes 0.000 description 1
- 208000032612 Glial tumor Diseases 0.000 description 1
- 206010018338 Glioma Diseases 0.000 description 1
- SXRSQZLOMIGNAQ-UHFFFAOYSA-N Glutaraldehyde Chemical compound O=CCCCC=O SXRSQZLOMIGNAQ-UHFFFAOYSA-N 0.000 description 1
- 241000700721 Hepatitis B virus Species 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101000960209 Homo sapiens Gamma-interferon-inducible protein 16 Proteins 0.000 description 1
- 101001037256 Homo sapiens Indoleamine 2,3-dioxygenase 1 Proteins 0.000 description 1
- 101000833614 Homo sapiens Interferon-inducible protein AIM2 Proteins 0.000 description 1
- 101000831007 Homo sapiens T-cell immunoreceptor with Ig and ITIM domains Proteins 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 102100040061 Indoleamine 2,3-dioxygenase 1 Human genes 0.000 description 1
- 241000712431 Influenza A virus Species 0.000 description 1
- 208000008839 Kidney Neoplasms Diseases 0.000 description 1
- 102000017578 LAG3 Human genes 0.000 description 1
- 101150030213 Lag3 gene Proteins 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 208000034578 Multiple myelomas Diseases 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- LFZAGIJXANFPFN-UHFFFAOYSA-N N-[3-[4-(3-methyl-5-propan-2-yl-1,2,4-triazol-4-yl)piperidin-1-yl]-1-thiophen-2-ylpropyl]acetamide Chemical compound C(C)(C)C1=NN=C(N1C1CCN(CC1)CCC(C=1SC=CC=1)NC(C)=O)C LFZAGIJXANFPFN-UHFFFAOYSA-N 0.000 description 1
- FIWILGQIZHDAQG-UHFFFAOYSA-N NC1=C(C(=O)NCC2=CC=C(C=C2)OCC(F)(F)F)C=C(C(=N1)N)N1N=C(N=C1)C1(CC1)C(F)(F)F Chemical compound NC1=C(C(=O)NCC2=CC=C(C=C2)OCC(F)(F)F)C=C(C(=N1)N)N1N=C(N=C1)C1(CC1)C(F)(F)F FIWILGQIZHDAQG-UHFFFAOYSA-N 0.000 description 1
- 102000012064 NLR Proteins Human genes 0.000 description 1
- 108091005686 NOD-like receptors Proteins 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 206010033128 Ovarian cancer Diseases 0.000 description 1
- 206010061535 Ovarian neoplasm Diseases 0.000 description 1
- 239000012661 PARP inhibitor Substances 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 206010061902 Pancreatic neoplasm Diseases 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 229940121906 Poly ADP ribose polymerase inhibitor Drugs 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-N R-2-phenyl-2-hydroxyacetic acid Natural products OC(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-N 0.000 description 1
- 108091005685 RIG-I-like receptors Proteins 0.000 description 1
- 102000014450 RNA Polymerase III Human genes 0.000 description 1
- 108010078067 RNA Polymerase III Proteins 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 206010038389 Renal cancer Diseases 0.000 description 1
- 241000580858 Simian-Human immunodeficiency virus Species 0.000 description 1
- 241000700584 Simplexvirus Species 0.000 description 1
- 208000005718 Stomach Neoplasms Diseases 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 102100024834 T-cell immunoreceptor with Ig and ITIM domains Human genes 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- YZCKVEUIGOORGS-NJFSPNSNSA-N Tritium Chemical compound [3H] YZCKVEUIGOORGS-NJFSPNSNSA-N 0.000 description 1
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 description 1
- 208000036142 Viral infection Diseases 0.000 description 1
- PNDPGZBMCMUPRI-XXSWNUTMSA-N [125I][125I] Chemical compound [125I][125I] PNDPGZBMCMUPRI-XXSWNUTMSA-N 0.000 description 1
- 239000003070 absorption delaying agent Substances 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 239000003463 adsorbent Substances 0.000 description 1
- 239000000443 aerosol Substances 0.000 description 1
- 150000001335 aliphatic alkanes Chemical group 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 230000009435 amidation Effects 0.000 description 1
- 238000007112 amidation reaction Methods 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- BIVUUOPIAYRCAP-UHFFFAOYSA-N aminoazanium;chloride Chemical compound Cl.NN BIVUUOPIAYRCAP-UHFFFAOYSA-N 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 230000002924 anti-infective effect Effects 0.000 description 1
- 239000000427 antigen Substances 0.000 description 1
- 230000014102 antigen processing and presentation of exogenous peptide antigen via MHC class I Effects 0.000 description 1
- 102000036639 antigens Human genes 0.000 description 1
- 108091007433 antigens Proteins 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-N benzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-N 0.000 description 1
- 229940092714 benzenesulfonic acid Drugs 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 239000012472 biological sample Substances 0.000 description 1
- 229940088623 biologically active substance Drugs 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 1
- 229910000024 caesium carbonate Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000007894 caplet Substances 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical compound OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 201000010881 cervical cancer Diseases 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 150000005829 chemical entities Chemical class 0.000 description 1
- 229940044683 chemotherapy drug Drugs 0.000 description 1
- SMJYMSAPPGLBAR-UHFFFAOYSA-N chloromethyl acetate Chemical compound CC(=O)OCCl SMJYMSAPPGLBAR-UHFFFAOYSA-N 0.000 description 1
- 238000003776 cleavage reaction Methods 0.000 description 1
- 208000029742 colonic neoplasm Diseases 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 230000009137 competitive binding Effects 0.000 description 1
- 229940125904 compound 1 Drugs 0.000 description 1
- 229940126214 compound 3 Drugs 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 125000000062 cyclohexylmethoxy group Chemical group [H]C([H])(O*)C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 231100000433 cytotoxic Toxicity 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 230000006240 deamidation Effects 0.000 description 1
- 238000007257 deesterification reaction Methods 0.000 description 1
- 230000007123 defense Effects 0.000 description 1
- 230000007812 deficiency Effects 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 102000038379 digestive enzymes Human genes 0.000 description 1
- 108091007734 digestive enzymes Proteins 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 208000035475 disorder Diseases 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 239000006196 drop Substances 0.000 description 1
- 230000036267 drug metabolism Effects 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 230000001804 emulsifying effect Effects 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 230000002255 enzymatic effect Effects 0.000 description 1
- 230000032050 esterification Effects 0.000 description 1
- 238000005886 esterification reaction Methods 0.000 description 1
- PQJJJMRNHATNKG-UHFFFAOYSA-N ethyl bromoacetate Chemical compound CCOC(=O)CBr PQJJJMRNHATNKG-UHFFFAOYSA-N 0.000 description 1
- 239000003889 eye drop Substances 0.000 description 1
- 229940012356 eye drops Drugs 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 239000012634 fragment Substances 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 206010017758 gastric cancer Diseases 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 239000003349 gelling agent Substances 0.000 description 1
- 229940097043 glucuronic acid Drugs 0.000 description 1
- 230000023611 glucuronidation Effects 0.000 description 1
- 239000003979 granulating agent Substances 0.000 description 1
- 201000010536 head and neck cancer Diseases 0.000 description 1
- 208000014829 head and neck neoplasm Diseases 0.000 description 1
- 150000002391 heterocyclic compounds Chemical class 0.000 description 1
- 238000002868 homogeneous time resolved fluorescence Methods 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-M hydrogensulfate Chemical compound OS([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-M 0.000 description 1
- 229940071870 hydroiodic acid Drugs 0.000 description 1
- 239000005457 ice water Substances 0.000 description 1
- 210000000987 immune system Anatomy 0.000 description 1
- 230000006054 immunological memory Effects 0.000 description 1
- 239000007943 implant Substances 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 238000001361 intraarterial administration Methods 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 230000009545 invasion Effects 0.000 description 1
- 229940044173 iodine-125 Drugs 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 230000000155 isotopic effect Effects 0.000 description 1
- 201000010982 kidney cancer Diseases 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 208000032839 leukemia Diseases 0.000 description 1
- 125000005647 linker group Chemical group 0.000 description 1
- 239000002502 liposome Substances 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 1
- 239000008297 liquid dosage form Substances 0.000 description 1
- 201000007270 liver cancer Diseases 0.000 description 1
- 208000014018 liver neoplasm Diseases 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 239000007937 lozenge Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 159000000003 magnesium salts Chemical class 0.000 description 1
- NXPHGHWWQRMDIA-UHFFFAOYSA-M magnesium;carbanide;bromide Chemical compound [CH3-].[Mg+2].[Br-] NXPHGHWWQRMDIA-UHFFFAOYSA-M 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 description 1
- 229960002510 mandelic acid Drugs 0.000 description 1
- 230000035800 maturation Effects 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- 239000004005 microsphere Substances 0.000 description 1
- 210000003470 mitochondria Anatomy 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 238000010172 mouse model Methods 0.000 description 1
- 239000007922 nasal spray Substances 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 208000002154 non-small cell lung carcinoma Diseases 0.000 description 1
- 239000012457 nonaqueous media Substances 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 238000006384 oligomerization reaction Methods 0.000 description 1
- 238000005580 one pot reaction Methods 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 201000002528 pancreatic cancer Diseases 0.000 description 1
- 208000008443 pancreatic carcinoma Diseases 0.000 description 1
- 239000006072 paste Substances 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 230000000865 phosphorylative effect Effects 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 239000002798 polar solvent Substances 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 230000035755 proliferation Effects 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 230000002285 radioactive effect Effects 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000006722 reduction reaction Methods 0.000 description 1
- 230000001172 regenerating effect Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 230000007017 scission Effects 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 201000011549 stomach cancer Diseases 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 229940014800 succinic anhydride Drugs 0.000 description 1
- 125000000542 sulfonic acid group Chemical group 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- ISIJQEHRDSCQIU-UHFFFAOYSA-N tert-butyl 2,7-diazaspiro[4.5]decane-7-carboxylate Chemical compound C1N(C(=O)OC(C)(C)C)CCCC11CNCC1 ISIJQEHRDSCQIU-UHFFFAOYSA-N 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 229930192474 thiophene Natural products 0.000 description 1
- 230000000451 tissue damage Effects 0.000 description 1
- 231100000827 tissue damage Toxicity 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 239000012049 topical pharmaceutical composition Substances 0.000 description 1
- 238000000844 transformation Methods 0.000 description 1
- GKASDNZWUGIAMG-UHFFFAOYSA-N triethyl orthoformate Chemical compound CCOC(OCC)OCC GKASDNZWUGIAMG-UHFFFAOYSA-N 0.000 description 1
- UCPYLLCMEDAXFR-UHFFFAOYSA-N triphosgene Chemical compound ClC(Cl)(Cl)OC(=O)OC(Cl)(Cl)Cl UCPYLLCMEDAXFR-UHFFFAOYSA-N 0.000 description 1
- 229910052722 tritium Inorganic materials 0.000 description 1
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 description 1
- 210000002700 urine Anatomy 0.000 description 1
- 230000009385 viral infection Effects 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 239000012224 working solution Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/06—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/50—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom condensed with carbocyclic rings or ring systems
- C07D333/52—Benzo[b]thiophenes; Hydrogenated benzo[b]thiophenes
- C07D333/54—Benzo[b]thiophenes; Hydrogenated benzo[b]thiophenes with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the hetero ring
- C07D333/56—Radicals substituted by oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/06—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D495/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms
- C07D495/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D495/04—Ortho-condensed systems
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Immunology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
技术领域technical field
本发明涉及化学医药领域,特别涉及苯并噻吩类化合物化合物或其可药用的盐、异构体及其制备方法、药物组合物和用途。The present invention relates to the field of chemical medicine, in particular to benzothiophene compounds or their pharmaceutically acceptable salts, isomers and their preparation methods, pharmaceutical compositions and uses.
背景技术Background technique
先天性免疫是防御入侵病原体以及组织损伤的第一道防线。模式识别受体(pattern recognition receptors,PRRs)识别来自病原体的病原体相关分子模式(pathogen-associated molecular patterns,PAMPs)或者来自机体自身的损伤相关分子模式(damage-associated molecular patterns,DAMPs),启动级联反应,诱导干扰素(interferons,IFNs)、趋化因子和炎性因子等基因的表达。这种先天性免疫应答不仅可以在早期抑制病原体的增殖与扩散,并且可以诱导后天性免疫应答最终清除感染。另外,这种先天性免疫应答监测肿瘤的发生与侵袭。目前,已经发现了多种PRRs,比如与膜结合的toll样受体(Toll-like receptors,TLRs)、C型凝集素受体(C-type lectin receptors,CLRs),不结合的细胞内核苷酸结合寡聚结构域样受体(nucleotide-binding oligomerization-domain(NOD)-like receptors,NLRs)、视黄酸样受体(retinoic acid-like receptors,RLRs)和细胞质DNA感受器等。Innate immunity is the first line of defense against invading pathogens and tissue damage. Pattern recognition receptors (PRRs) recognize pathogen-associated molecular patterns (PAMPs) from pathogens or damage-associated molecular patterns (DAMPs) from the body itself, initiating a cascade In response, the expression of genes such as interferons (IFNs), chemokines and inflammatory factors is induced. This innate immune response not only suppresses the proliferation and spread of pathogens at an early stage, but also induces an acquired immune response to eventually clear the infection. Additionally, this innate immune response monitors tumor initiation and invasion. At present, a variety of PRRs have been found, such as membrane-bound toll-like receptors (TLRs), C-type lectin receptors (CLRs), unbound intracellular nucleotides Binding oligomerization domain-like receptors (nucleotide-binding oligomerization-domain (NOD)-like receptors, NLRs), retinoic acid-like receptors (retinoic acid-like receptors, RLRs) and cytoplasmic DNA sensors.
病原体DNA以及机体细胞核或线粒体泄漏到细胞质的DNA是一种免疫刺激分子,而细胞质中存在许多DNA感受器,比如黑色素瘤缺乏因子2(absent in melanoma 2,AIM2),干扰素诱导蛋白16(interferon-inducible protein 16,IFI16),RNA聚合酶III和环化GMP-AMP合成酶(cyclic GMP-AMP synthase,cGAS)等。位于细胞质内质网的干扰素基因刺激蛋白(STimulator of INterferon Genes,STING)是这些DNA感受器的汇聚点,控制DNA这种免疫刺激分子所介导的先天性免疫应答。研亢表明,cGAS识别细胞质内双链DNA,催化GTP和ATP生成2′3′-cGAMP。2′3′-cGAMP作为第二信使与干扰素基因刺激蛋白结合,诱导干扰素基因刺激蛋白发生二聚且从内质网膜转移至细胞核周围囊泡,募集TBK1进而使得自身发生磷酸化,从而使得IRF3靠近干扰素基因刺激蛋白。然后,TBK1使得IRF3磷酸化,后者转移至细胞核从而诱导I型干扰素(interferon,IFN)和其他炎性因子的表达。I型干扰素促进树突细胞(dendritic cell,DC)成熟,选择性刺激抗原的交叉呈递,募集CD8+T细胞,从而激活后天性免疫系统杀死肿瘤细胞或者抵抗病原体感染。因此,干扰素基因刺激蛋白在肿瘤和病原体感染中的先天性和后天性免疫应答中发挥重要作用。Pathogen DNA and DNA leaked from the body's nucleus or mitochondria into the cytoplasm is an immunostimulatory molecule, and there are many DNA sensors in the cytoplasm, such as melanoma deficiency factor 2 (absent in melanoma 2, AIM2), interferon-inducible protein 16 (interferon- inducible protein 16, IFI16), RNA polymerase III and cyclic GMP-AMP synthase (cyclic GMP-AMP synthase, cGAS) and so on. The STimulator of INterferon Genes (STING) located in the cytoplasmic endoplasmic reticulum is the convergence point of these DNA sensors and controls the innate immune response mediated by DNA, an immunostimulatory molecule. Research shows that cGAS recognizes double-stranded DNA in the cytoplasm and catalyzes GTP and ATP to generate 2′3′-cGAMP. As a second messenger, 2′3′-cGAMP binds to interferon gene-stimulating protein, induces interferon gene-stimulating protein to dimerize and transfer from the endoplasmic reticulum membrane to vesicles around the nucleus, recruiting TBK1 and phosphorylating itself, thereby Brings IRF3 close to interferon gene-stimulating protein. TBK1 then phosphorylates IRF3, which translocates to the nucleus to induce the expression of type I interferons (IFNs) and other inflammatory factors. Type I interferons promote the maturation of dendritic cells (DCs), selectively stimulate the cross-presentation of antigens, and recruit CD8 + T cells, thereby activating the acquired immune system to kill tumor cells or resist pathogen infection. Therefore, interferon gene-stimulating proteins play important roles in innate and acquired immune responses in tumors and pathogen infections.
鉴于干扰素基因刺激蛋白在机体免疫应答中发挥的关键作用,干扰素基因刺激蛋白激动剂应用于多种疾病的免疫治疗备受关注。比如,瘤内注射2′3′-cGAMP导致小鼠模型中的肿瘤细胞退化,并且能够抑制远端肿瘤细胞的生长,诱导长期的免疫记忆。干扰素基因刺激蛋白激动剂去免疫检查点抑制剂联用,增加免疫检查点抑制剂的疗效。另外,干扰素基因刺激蛋白激动剂诱导的机体免疫应答可以有效控制甲型流感病毒、乙型肝炎病毒、单纯疱疹病毒和HIV等病毒感染。因此,干扰素基因刺激蛋白激动剂的研亢与发展对于癌症和感染性疾病的免疫治疗具有重要意义。但是,目前报道的干扰素基因刺激蛋白激动剂多为环状二核苷酸类似物,但是体内代谢不稳定性、膜透过性差,只能瘤内给药等缺陷极大地限制了干扰素基因刺激蛋白激动剂的应用,因此干扰素基因刺激蛋白小分子激动剂的研亢与开发具有重要意义。In view of the key role of interferon gene-stimulating protein in the body's immune response, the application of interferon gene-stimulating protein agonists in the immunotherapy of various diseases has attracted much attention. For example, intratumoral injection of 2′3′-cGAMP resulted in tumor cell regression in mouse models and was able to inhibit the growth of distant tumor cells and induce long-term immune memory. Interferon gene-stimulating protein agonists combined with immune checkpoint inhibitors can increase the efficacy of immune checkpoint inhibitors. In addition, the immune response induced by interferon gene-stimulating protein agonists can effectively control viral infections such as influenza A virus, hepatitis B virus, herpes simplex virus and HIV. Therefore, the research and development of interferon gene-stimulating protein agonists are of great significance for the immunotherapy of cancer and infectious diseases. However, most of the reported interferon gene-stimulating protein agonists are cyclic dinucleotide analogs, but their metabolic instability in vivo, poor membrane permeability, and only intratumoral administration are limited by the defects of interferon gene-stimulating protein. Therefore, the research and development of interferon gene-stimulating protein small molecule agonists are of great significance.
发明内容SUMMARY OF THE INVENTION
发明目的:本发明目的是克服现有干扰素基因刺激蛋白小分子激动剂结构单一的缺陷,而提供了一种苯并噻吩类化合物、其制备方法、药物组合物及用途。本发明的苯并噻吩类化合物对干扰素基因刺激蛋白具有很好的激动活性,且对肿瘤具有有良好的治疗作用。Purpose of the invention: The purpose of the present invention is to overcome the defect of single structure of the existing interferon gene-stimulating protein small molecule agonists, and provide a benzothiophene compound, its preparation method, pharmaceutical composition and use. The benzothiophene compound of the present invention has good agonistic activity on interferon gene-stimulating protein, and has good therapeutic effect on tumor.
技术方案:Technical solutions:
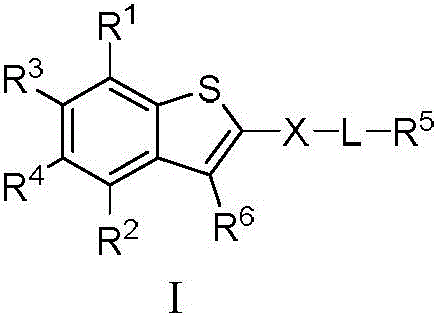
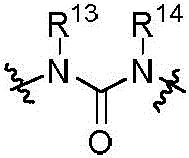
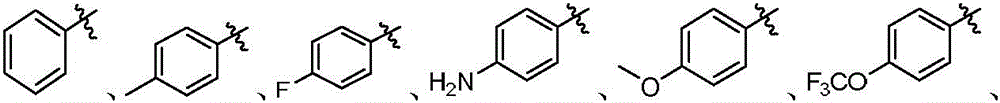
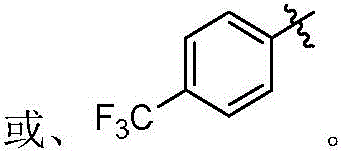
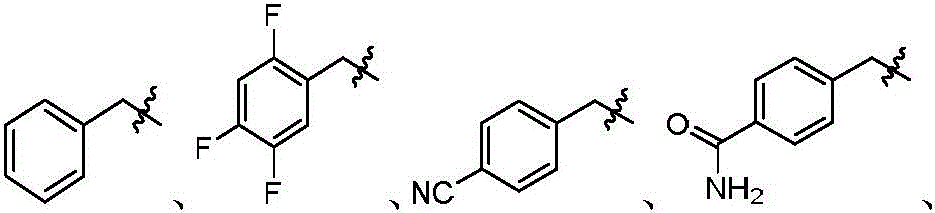
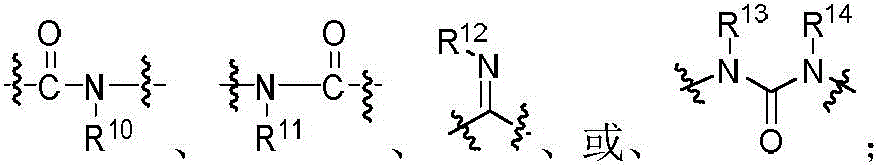
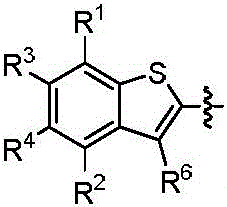
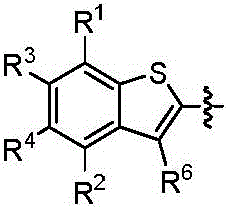
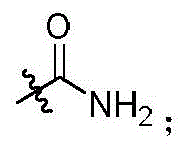
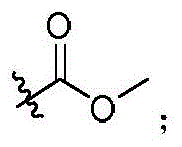
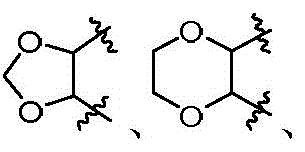
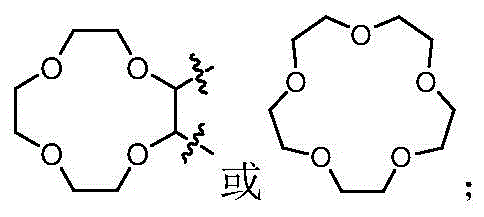
本发明的第一方面,具有通式I所示结构的苯并噻吩类化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物,其结构如下:In the first aspect of the present invention, a benzothiophene compound having a structure represented by general formula I, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or hydrate thereof, has the following structure :
其中,R1和R2独立地选自氢、氘、卤素、未取代或R1-1取代的C1-6烷基、C1-6烷氧基、C1-6卤代烷氧基、-NR1-2R1-3、-(C=O)NR1-4R1-5或-(C=O)OR1-6;wherein, R 1 and R 2 are independently selected from hydrogen, deuterium, halogen, unsubstituted or R 1-1 substituted C 1-6 alkyl, C 1-6 alkoxy, C 1-6 haloalkoxy, - NR 1-2 R 1-3 , -(C=O)NR 1-4 R 1-5 or -(C=O)OR 1-6 ;
R1-1为卤素、C1-4烷基、C1-4卤代烷基、C1-4烷氧基、C1-4卤代烷氧基或-NR1-1-1R1-1-2;R 1-1 is halogen, C 1-4 alkyl, C 1-4 haloalkyl, C 1-4 alkoxy, C 1-4 haloalkoxy or -NR 1-1-1 R 1-1-2 ;
R1-1-1和R1-1-2独立地选自氢或C1-4烷基;R 1-1-1 and R 1-1-2 are independently selected from hydrogen or C 1-4 alkyl;
R1-2~R1-6独立地选自氢或C1-4烷基;R 1-2 to R 1-6 are independently selected from hydrogen or C 1-4 alkyl;
R3和R4独立地选自氢、氘、卤素、羟基、氰基、未取代或R3-1取代的C1-6烷基、C1-6烷氧基、C1-6卤代烷氧基、C2-6炔基、C2-6烯基、C3-10环烷基、(C3-10环烷基)-氧基、(C3-10环烷基)-(C1-6烷氧基)、杂环烷基、杂环烷基-氧基、杂环烷基-(C1-6烷氧基)、C6-10芳基、(C6-10芳基)-氧基、(C6-10芳基)-(C1-6烷氧基)、杂芳基、杂芳基-氧基、杂芳基-(C1-6烷氧基)、-NR3-2R3-3、-(C=O)R3-4、-(C=O)NR3-5R3-6、-(C=O)OR3-7、-S(=O)2NR3-8R3-9、或R3和R4与它们相连的碳原子一起组成4~20元杂环烷氧基;所述的杂环烷基为“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”4~10元杂环烷基;所述的杂芳基为“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基;R 3 and R 4 are independently selected from hydrogen, deuterium, halogen, hydroxy, cyano, unsubstituted or R 3-1 substituted C 1-6 alkyl, C 1-6 alkoxy, C 1-6 haloalkoxy base, C 2-6 alkynyl, C 2-6 alkenyl, C 3-10 cycloalkyl, (C 3-10 cycloalkyl)-oxy, (C 3-10 cycloalkyl)-(C 1 -6 alkoxy), heterocycloalkyl, heterocycloalkyl-oxy, heterocycloalkyl-(C 1-6 alkoxy), C 6-10 aryl, (C 6-10 aryl) -oxy, (C 6-10 aryl)-(C 1-6 alkoxy), heteroaryl, heteroaryl-oxy, heteroaryl-(C 1-6 alkoxy), -NR 3-2 R 3-3 , -(C=O)R 3-4 , -(C=O)NR 3-5 R 3-6 , -(C=O)OR 3-7 , -S(=O ) 2 NR 3-8 R 3-9 , or R 3 and R 4 and the carbon atoms to which they are attached together form a 4-20-membered heterocycloalkoxy group; the heterocycloalkyl group is "heteroatoms selected from N, One or more of O and S, "4- to 10-membered heterocycloalkyl with 1-3 heteroatoms; the heteroaryl group is "heteroatom selected from N, O and S One or more kinds of "5-10-membered heteroaryl groups with 1-3 heteroatoms;
R3-1为卤素、C1-4烷基、C1-4卤代烷基、C1-4烷氧基、C1-4卤代烷氧基、或-NR3-1-1R3-1-2;R 3-1 is halogen, C 1-4 alkyl, C 1-4 haloalkyl, C 1-4 alkoxy, C 1-4 haloalkoxy, or -NR 3-1-1 R 3-1- 2 ;
R3-1-1和R3-1-2独立地选自氢或C1-4烷基;R 3-1-1 and R 3-1-2 are independently selected from hydrogen or C 1-4 alkyl;
R3-2~R3-9独立地选自氢或C1-4烷基;R 3-2 to R 3-9 are independently selected from hydrogen or C 1-4 alkyl;
R5为-(C=O)OR5-1、-(C=O)SR5-2、未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基、-(C=O)NR5-4(S=O)2R5-5或-(C=O)NR7R8;R 5 is -(C=O)OR 5-1 , -(C=O)SR 5-2 , unsubstituted or R 5-3 substituted "heteroatom selected from one or more of N, O and S Species, "5-10-membered heteroaryl with 1-3 heteroatoms, -(C=O)NR 5-4 (S=O) 2 R 5-5 or -(C=O)NR 7 R 8 ;
R5-1和R5-2独立地选自氢、C1-6烷基、(C1-6烷氧基)-(C1-6烷基)-、C3-10环烷基、C6-10芳基、“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基或“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”4~10元杂环烷基;R 5-1 and R 5-2 are independently selected from hydrogen, C 1-6 alkyl, (C 1-6 alkoxy)-(C 1-6 alkyl)-, C 3-10 cycloalkyl, C 6-10 aryl, "hetero atoms are selected from one or more of N, O and S, and the number of hetero atoms is 1-3" 5-10-membered heteroaryl groups or "hetero atoms are selected from N, One or more of O and S, "4- to 10-membered heterocycloalkyl with 1-3 heteroatoms;
R5-3选自卤素、羟基、氨基、氰基、C1-4烷基、C1-4烷氧基、C1-4卤代烷基、或C1-4卤代烷氧基;R 5-3 is selected from halogen, hydroxyl, amino, cyano, C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, or C 1-4 haloalkoxy;
R5-4和R5-5独立地选自氢、C1-6烷基、C1-6卤代烷基或C3-10环烷基;R 5-4 and R 5-5 are independently selected from hydrogen, C 1-6 alkyl, C 1-6 haloalkyl or C 3-10 cycloalkyl;
R7为氢、磺酸基、磷酰基、未取代或R7-1取代的C1-6烷基、C2-6烯基、C3-10环烷基、(C3-10环烷基)-C1-6烷基、杂环烷基、杂环烷基-C1-6烷基、未取代或R7-2取代的C6-10芳基、未取代或R7-3取代的C6-10芳基-C1-6烷基、未取代或R7-4取代的杂芳基-C1-6烷基或未取代或R7-5取代的杂芳基;所述的杂环烷基为“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”4~10元杂环烷基;所述的杂芳基为“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基;R 7 is hydrogen, sulfonic acid group, phosphoryl group, unsubstituted or R 7-1 substituted C 1-6 alkyl, C 2-6 alkenyl, C 3-10 cycloalkyl, (C 3-10 cycloalkane base)-C 1-6 alkyl, heterocycloalkyl, heterocycloalkyl-C 1-6 alkyl, unsubstituted or R 7-2 substituted C 6-10 aryl, unsubstituted or R 7-3 Substituted C 6-10 aryl-C 1-6 alkyl, unsubstituted or R 7-4 substituted heteroaryl-C 1-6 alkyl or unsubstituted or R 7-5 substituted heteroaryl; The heterocycloalkyl group is a 4- to 10-membered heterocycloalkyl group with "heteroatoms selected from one or more of N, O and S, and the number of heteroatoms is 1-3"; the heteroaryl group is a 5- to 10-membered heteroaryl group with "heteroatoms selected from one or more of N, O and S, and the number of heteroatoms is 1-3";
R7-1为卤素、羧基、羟基、-NR7-1-1R7-1-2、氰基或C1-4烷基;R 7-1 is halogen, carboxyl, hydroxyl, -NR 7-1-1 R 7-1-2 , cyano or C 1-4 alkyl;
R7-1-1和R7-1-2独立地选自氢或C1-4烷基;R 7-1-1 and R 7-1-2 are independently selected from hydrogen or C 1-4 alkyl;
R7-2~R7-5独立地选自氘、卤素、羟基、氰基、硝基、未取代或R7-2-1取代的C1-4烷基、未取代或R7-2-2取代的C1-4烷氧基、-NR7-2-3R7-2-4、-(C=O)R7-2-5、-(C=O)NR7-2-6R7-2-7、-(C=O)OR7-2-8或-S(=O)2NR7-2-9R7-2-10;R 7-2 to R 7-5 are independently selected from deuterium, halogen, hydroxyl, cyano, nitro, unsubstituted or R 7-2-1 substituted C 1-4 alkyl, unsubstituted or R 7-2 -2- substituted C 1-4 alkoxy, -NR 7-2-3 R 7-2-4 , -(C=O)R 7-2-5 , -(C=O)NR 7-2- 6 R 7-2-7 , -(C=O)OR 7-2-8 or -S(=O) 2 NR 7-2-9 R 7-2-10 ;
R7-2-1和R7-2-2为卤素、羟基、氰基或-NR7-2-1-1R7-2-1-2;R 7-2-1 and R 7-2-2 are halogen, hydroxyl, cyano or -NR 7-2-1-1 R 7-2-1-2 ;
R7-2-1-1和R7-2-1-2独立地选自氢或C1-4烷基;R 7-2-1-1 and R 7-2-1-2 are independently selected from hydrogen or C 1-4 alkyl;
R7-2-3~R7-2-10独立地选自氢或C1-4烷基;R 7-2-3 to R 7-2-10 are independently selected from hydrogen or C 1-4 alkyl;
R8为氢、氰基、R8-1O-、-NR8-2R8-3、R8-4S-、未取代或R8-5取代的C1-6烷基、C2-6烯基、C3-10环烷基、(C3-10环烷基)-C1-6烷基、杂环烷基、杂环烷基-C1-6烷基、未取代或R8-6取代的C6-10芳基、未取代或R8-7取代的C6-10芳基-C1-6烷基、未取代或R8-8取代的杂芳基、未取代或R8-9取代的杂芳基-C1-6烷基;所述的杂环烷基为“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”4~10元杂环烷基;所述的杂芳基为“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基;R 8 is hydrogen, cyano, R 8-1 O-, -NR 8-2 R 8-3 , R 8-4 S-, unsubstituted or R 8-5 substituted C 1-6 alkyl, C 2 -6 alkenyl, C 3-10 cycloalkyl, (C 3-10 cycloalkyl)-C 1-6 alkyl, heterocycloalkyl, heterocycloalkyl-C 1-6 alkyl, unsubstituted or R 8-6 substituted C 6-10 aryl, unsubstituted or R 8-7 substituted C 6-10 aryl-C 1-6 alkyl, unsubstituted or R 8-8 substituted heteroaryl, unsubstituted Substituted or R 8-9 substituted heteroaryl-C 1-6 alkyl; Said heterocycloalkyl is "one or more heteroatoms selected from N, O and S, and the number of heteroatoms is 1 -3 "4- to 10-membered heterocycloalkyl groups; the heteroaryl group is "one or more heteroatoms selected from N, O and S, and the number of heteroatoms is 1-3"5 ~10-membered heteroaryl;
R8-1~R8-4独立地选自磷酰基、未取代或R8-1-1取代的C1-6烷基、C2-6烯基、C3-10环烷基、(C3-10环烷基)-C1-6烷基、杂环烷基-C1-6烷基、未取代或R8-1-2取代的C6-10芳基、未取代或R8-1-3取代的C6-10芳基-C1-6烷基、未取代或R8-1-4取代的杂芳基-C1-6烷基、未取代或R8-1-5取代的杂芳基、或-(S=O)2OR8-1-6;所述的杂环烷基为“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”4~10元杂环烷基;所述的杂芳基为“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基;R 8-1 to R 8-4 are independently selected from phosphoryl, unsubstituted or R 8-1-1 substituted C 1-6 alkyl, C 2-6 alkenyl, C 3-10 cycloalkyl, ( C 3-10 cycloalkyl)-C 1-6 alkyl, heterocycloalkyl-C 1-6 alkyl, unsubstituted or R 8-1-2 substituted C 6-10 aryl, unsubstituted or R 8-1-3 substituted C 6-10 aryl-C 1-6 alkyl, unsubstituted or R 8-1-4 substituted heteroaryl-C 1-6 alkyl, unsubstituted or R 8-1 -5 -substituted heteroaryl group, or -(S=O) 2 OR 8-1-6 ; the heterocycloalkyl group is "the heteroatom is selected from one or more of N, O and S, the heteroatom is "4-10-membered heterocycloalkyl with 1-3 atoms; the heteroaryl group is "heteroatoms selected from one or more of N, O and S, and the number of heteroatoms is 1-3 "5-10-membered heteroaryl;
R8-1-1为卤素、羟基、氰基、-NR8-1-1-1R8-1-1-2或C1-4烷基;R 8-1-1 is halogen, hydroxyl, cyano, -NR 8-1-1-1 R 8-1-1-2 or C 1-4 alkyl;
R8-1-1-1和R8-1-1-2独立地选自氢或C1-4烷基;R 8-1-1-1 and R 8-1-1-2 are independently selected from hydrogen or C 1-4 alkyl;
R8-1-2~R8-1-5独立地选自卤素、羟基、氰基、C1-4烷基、C1-4烷氧基、C1-4卤代烷基、C1-4卤代烷氧基、-NR8-1-2-1R8-1-2-2、-(C=O)R8-1-2-3、-(C=O)NR8-1-2-4R8-1-2-5、-(C=O)OR8-1-2-6、-S(=O)2NR8-1-2-7R8-1-2-8、或-S(=O)2R8-1-2-9;R 8-1-2 to R 8-1-5 are independently selected from halogen, hydroxyl, cyano, C 1-4 alkyl, C 1-4 alkoxy, C 1-4 haloalkyl, C 1-4 Haloalkoxy, -NR 8-1-2-1 R 8-1-2-2 , -(C=O)R 8-1-2-3 , -(C=O)NR 8-1-2- 4 R 8-1-2-5 , -(C=O)OR 8-1-2-6 , -S(=O) 2 NR 8-1-2-7 R 8-1-2-8 , or -S(=O) 2 R 8-1-2-9 ;
R8-1-6为氢、C1-6烷基、C1-6卤代烷基或C3-10环烷基;R 8-1-6 is hydrogen, C 1-6 alkyl, C 1-6 haloalkyl or C 3-10 cycloalkyl;
R8-1-2-1~R8-1-2-9独立地选自氢或C1-4烷基;R 8-1-2-1 to R 8-1-2-9 are independently selected from hydrogen or C 1-4 alkyl;
R8-5为卤素、羧基、羟基、-NR8-5-1R8-5-2、氰基或C1-4烷基;R 8-5 is halogen, carboxyl, hydroxyl, -NR 8-5-1 R 8-5-2 , cyano or C 1-4 alkyl;
R8-5-1和R8-5-2独立地选自氢或C1-4烷基;R 8-5-1 and R 8-5-2 are independently selected from hydrogen or C 1-4 alkyl;
R8-6~R8-9独立地选自氘、卤素、羟基、氰基、羧基、硝基、未取代或R8-6-1取代的C1-4烷基、未取代或R8-6-2取代的C1-4烷氧基、-NR8-6-3R8-6-4、-(C=O)R8-6-5、-(C=O)NR8-6-6R8-6-7、-(C=O)OR8-6-8、或-S(=O)2NR8-6-9R8-6-10;R 8-6 to R 8-9 are independently selected from deuterium, halogen, hydroxyl, cyano, carboxyl, nitro, unsubstituted or R 8-6-1 substituted C 1-4 alkyl, unsubstituted or R 8 -6-2 substituted C 1-4 alkoxy, -NR 8-6-3 R 8-6-4 , -(C=O)R 8-6-5 , -(C=O)NR 8- 6-6 R 8-6-7 , -(C=O)OR 8-6-8 , or -S(=O) 2 NR 8-6-9 R 8-6-10 ;
R8-6-1和R8-6-2为卤素、羟基、氰基或-NR8-6-1-1R8-6-1-2;R 8-6-1 and R 8-6-2 are halogen, hydroxyl, cyano or -NR 8-6-1-1 R 8-6-1-2 ;
R8-6-1-1和R8-6-1-2独立地选自氢或C1-4烷基;R 8-6-1-1 and R 8-6-1-2 are independently selected from hydrogen or C 1-4 alkyl;
R8-6-3~R8-6-10独立地选自氢或C1-4烷基;R 8-6-3 to R 8-6-10 are independently selected from hydrogen or C 1-4 alkyl;
X为O、C=O、NR9、 X is O, C=O, NR 9 ,
X的左端与相连,右端与L相连;The left end of X with connected, and the right end is connected to L;
R9~R11独立地选自氢或C1-6烷基;R 9 to R 11 are independently selected from hydrogen or C 1-6 alkyl;
R12为羟基、氰基、C1-6烷氧基、-O(C=O)R12-1、-(S=O)2R12-2、-(S=O)R12-3或-NR12- 4R12-5;R 12 is hydroxyl, cyano, C 1-6 alkoxy, -O(C=O)R 12-1 , -(S=O) 2 R 12-2 , -(S=O)R 12-3 or -NR 12-4 R 12-5 ;
R12-1~R12-3独立地选自C1-6烷基;R 12-1 to R 12-3 are independently selected from C 1-6 alkyl;
R12-4和R12-5独立地选自氢、C1-6烷基或-(C=O)R12-4-1;R 12-4 and R 12-5 are independently selected from hydrogen, C 1-6 alkyl or -(C=O)R 12-4-1 ;
R12-4-1为C1-6烷基;R 12-4-1 is C 1-6 alkyl;
R13和R14独立地选自氢或C1-6烷基;R 13 and R 14 are independently selected from hydrogen or C 1-6 alkyl;
X和R5选自以下任一组合:X and R are selected from any combination of the following:
当X为时,R5为-(C=O)NR5-4(S=O)2R5-5或-(C=O)NR7R8;when X is , R 5 is -(C=O)NR 5-4 (S=O) 2 R 5-5 or -(C=O)NR 7 R 8 ;
当X为C=O时,R5为-(C=O)NR5-4(S=O)2R5-5或未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基;When X is C=O, R 5 is -(C=O)NR 5-4 (S=O) 2 R 5-5 or unsubstituted or R 5-3 substituted "heteroatom selected from N, O and One or more of S, "5-10-membered heteroaryl group with 1-3 heteroatoms;
当X为O、NR9、时,R5为-(C=O)OR5-1、-(C=O)SR5-2、未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基、-(C=O)NR5-4(S=O)2R5-5或-(C=O)NR7R8;When X is O, NR 9 , When R 5 is -(C=O)OR 5-1 , -(C=O)SR 5-2 , unsubstituted or R 5-3 substituted "heteroatom is selected from one of N, O and S or more, "5-10-membered heteroaryl with 1-3 heteroatoms, -(C=O)NR 5-4 (S=O) 2 R 5-5 or -(C=O)NR 7R8 ;
R6选自氢、氘、卤素、C1-6烷基、C1-6烷氧基、C1-6卤代烷基、C1-6卤代烷氧基、-(C=O)NR6-1R6-2、-(C=O)OR6-3或-NR6-4R6-5;R 6 is selected from hydrogen, deuterium, halogen, C 1-6 alkyl, C 1-6 alkoxy, C 1-6 haloalkyl, C 1-6 haloalkoxy, -(C=O)NR 6-1 R 6-2 , -(C=O)OR 6-3 or -NR 6-4 R 6-5 ;
R6-1~R6-5独立地选自氢或C1-4烷基;R 6-1 to R 6-5 are independently selected from hydrogen or C 1-4 alkyl;
L为(C(R15R16))n;L is (C(R 15 R 16 )) n ;
R15和R16独立地选自氢、卤素、氰基、C1-6烷基、C1-6卤代烷基、C1-6烷氧基、C1-6卤代烷氧基、C3-6环烷基、-NR15-1R15-2或R15和R16与它们相连的碳原子环合成C3-10环烷基;R 15 and R 16 are independently selected from hydrogen, halogen, cyano, C 1-6 alkyl, C 1-6 haloalkyl, C 1-6 alkoxy, C 1-6 haloalkoxy, C 3-6 Cycloalkyl, -NR 15-1 R 15-2 or R 15 and R 16 ring with the carbon atoms to which they are attached to synthesize C 3-10 cycloalkyl;
R15-1和R15-2独立地选自氢或C1-4烷基;R 15-1 and R 15-2 are independently selected from hydrogen or C 1-4 alkyl;
n为1、2或3的整数。n is an integer of 1, 2 or 3.
在本发明某一实施方案中,某些基团的定义如下,未定义的基团同前任一方案所述,以下简称“在本发明某一实施方案中”,当R1和R2独立地选自卤素时,所述的卤素为氟、氯、溴或碘;In an embodiment of the present invention, certain groups are defined as follows, and undefined groups are as described in any of the previous embodiments, hereinafter referred to as "in an embodiment of the present invention", when R 1 and R 2 are independently When selected from halogen, the halogen is fluorine, chlorine, bromine or iodine;
在本发明某一实施方案中,当R3和R4独立地选自卤素时,所述的卤素为氟、氯、溴或碘。 In a certain embodiment of the present invention, when R3 and R4 are independently selected from halogen, said halogen is fluorine, chlorine, bromine or iodine.
在本发明某一实施方案中,当R3和R4独立地选自C1-6烷氧基时,所述的C1-6烷氧基为C1-4烷氧基。In one embodiment of the present invention, when R 3 and R 4 are independently selected from C 1-6 alkoxy, said C 1-6 alkoxy is C 1-4 alkoxy.
在本发明某一实施方案中,当R3和R4独立地选自C1-6卤代烷氧基时,所述的C1-6卤代烷氧基为C1-4卤代烷氧基。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from C 1-6 haloalkoxy, said C 1-6 haloalkoxy is C 1-4 haloalkoxy.
在本发明某一实施方案中,当R3和R4独立地选自C2-6烯基时,所述的C2-6烯基为C2-4烯基。In a certain embodiment of the present invention, when R3 and R4 are independently selected from C2-6alkenyl , said C2-6alkenyl is C2-4alkenyl .
在本发明某一实施方案中,当R3和R4独立地选自(C3-10环烷基)-氧基时,所述的(C3-10环烷基)-氧基为(C3-6环烷基)-氧基。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from (C 3-10 cycloalkyl)-oxy, said (C 3-10 cycloalkyl)-oxy is ( C 3-6 cycloalkyl)-oxy.
在本发明某一实施方案中,当R3和R4独立地选自(C3-10环烷基)-(C1-6烷氧基)时,所述的(C3-10环烷基)-(C1-6烷氧基)为(C3-6环烷基)-(C1-4烷氧基)。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from (C 3-10 cycloalkyl)-(C 1-6 alkoxy), the (C 3-10 cycloalkane) base)-(C 1-6 alkoxy) is (C 3-6 cycloalkyl)-(C 1-4 alkoxy).
在本发明某一实施方案中,当R3和R4独立地选自“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”4~10元杂环烷基-(C1-6烷氧基)时,所述的4~10元杂环烷基-(C1-6烷氧基)为4~6元杂环烷基-(C1-4烷氧基)。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from "hetero atoms are selected from one or more of N, O and S, and the number of hetero atoms is 1-3" 4-10 In the case of membered heterocycloalkyl-(C 1-6 alkoxy), the 4-10-membered heterocycloalkyl-(C 1-6 alkoxy) is a 4-6 membered heterocycloalkyl-(C 1-4 alkoxy).
在本发明某一实施方案中,当R3和R4独立地选自(C6-10芳基)-氧基时,所述的(C6-10芳基)-氧基为苯氧基。In one embodiment of the present invention, when R 3 and R 4 are independently selected from (C 6-10 aryl)-oxy, said (C 6-10 aryl)-oxy is phenoxy .
在本发明某一实施方案中,当R3和R4独立地选自(C6-10芳基)-(C1-6烷氧基)时,所述的(C6-10芳基)-(C1-6烷氧基)为苯-(C1-4烷氧基)。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from (C 6-10 aryl)-(C 1-6 alkoxy), the (C 6-10 aryl) -(C 1-6 alkoxy) is benzene-(C 1-4 alkoxy).
在本发明某一实施方案中,当R3和R4独立地选自“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-氧基时,所述的5~10元杂芳基-氧基为5-6元杂芳基-氧基。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from "hetero atoms are selected from one or more of N, O and S, and the number of hetero atoms is 1-3" 5-10 In the case of a heteroaryl-oxy group, the 5-10-membered heteroaryl-oxy group is a 5-6-membered heteroaryl-oxy group.
在本发明某一实施方案中,当R3和R4独立地选自“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-(C1-6烷氧基)时,所述的5~10元杂芳基-(C1-6烷氧基)为5-6元杂芳基-(C1-4烷氧基)。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from "hetero atoms are selected from one or more of N, O and S, and the number of hetero atoms is 1-3" 5-10 In the case of heteroaryl-(C 1-6 alkoxy), the 5-10-membered heteroaryl-(C 1-6 alkoxy) is a 5-6 membered heteroaryl-(C 1-4 alkoxy).
在本发明某一实施方案中,当R3-2~R3-9为C1-4烷基时,所述的C1-4烷基为甲基、乙基、丙基、或异丙基。In a certain embodiment of the present invention, when R 3-2 to R 3-9 are C 1-4 alkyl groups, the C 1-4 alkyl groups are methyl, ethyl, propyl, or isopropyl base.
在本发明某一实施方案中,当R3和R4与它们相连的碳原子一起组成“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”4-20元杂环烷氧基时,所述的4-20元杂环烷基为5-12元杂环烷氧基。In a certain embodiment of the present invention, when R 3 and R 4 together with the carbon atoms to which they are attached form "heteroatoms selected from one or more of N, O and S, and the number of heteroatoms is 1-3""4-20-membered heterocycloalkoxy, the 4-20-membered heterocycloalkyl is a 5-12-membered heterocycloalkoxy.
在本发明某一实施方案中,当R5-1为C1-6烷基时,所述的C1-6烷基为C1-4烷基。In a certain embodiment of the present invention, when R 5-1 is a C 1-6 alkyl group, the C 1-6 alkyl group is a C 1-4 alkyl group.
在本发明某一实施方案中,当R5-1为(C1-6烷氧基)-(C1-6烷基)-时,所述的(C1-6烷氧基)-(C1-6烷基)-为(C1-4烷氧基)-(C1-4烷基)-。In a certain embodiment of the present invention, when R 5-1 is (C 1-6 alkoxy)-(C 1-6 alkyl)-, the (C 1-6 alkoxy)-( C 1-6 alkyl)- is (C 1-4 alkoxy)-(C 1-4 alkyl)-.
在本发明某一实施方案中,当R5-1为C3-10环烷基时,所述的C3-10环烷基为C3-6环烷基。In a certain embodiment of the present invention, when R 5-1 is a C 3-10 cycloalkyl group, the C 3-10 cycloalkyl group is a C 3-6 cycloalkyl group.
在本发明某一实施方案中,当R5为未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基时,所述的R5-3个数为一个或多个,当存在多个R5-3时,所述的R5-3可相同或不同。In a certain embodiment of the present invention, when R 5 is unsubstituted or R 5-3 substituted "heteroatoms are selected from one or more of N, O and S, and the number of heteroatoms is 1-3" In the case of a 5- to 10-membered heteroaryl group, the number of the R 5-3 is one or more, and when there are multiple R 5-3 , the R 5-3 can be the same or different.
在本发明某一实施方案中,当R5为未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基时,所述的5~10元杂芳基为5~6元杂芳基。In a certain embodiment of the present invention, when R 5 is unsubstituted or R 5-3 substituted "heteroatoms are selected from one or more of N, O and S, and the number of heteroatoms is 1-3" When a 5- to 10-membered heteroaryl group is used, the 5- to 10-membered heteroaryl group is a 5- to 6-membered heteroaryl group.
在本发明某一实施方案中,当R5-4和R5-5独立地选自C1-6烷基时,所述的C1-6烷基为C1-4烷基。In a certain embodiment of the present invention, when R 5-4 and R 5-5 are independently selected from C 1-6 alkyl, said C 1-6 alkyl is C 1-4 alkyl.
在本发明某一实施方案中,当R7为未取代或R7-1取代的C1-6烷基时,所述的R7-1的个数为一个或多个,当存在多个R7-1时,所述的R7-1可相同或不同。In a certain embodiment of the present invention, when R 7 is an unsubstituted or R 7-1 substituted C 1-6 alkyl group, the number of said R 7-1 is one or more, when there are multiple When R 7-1 , the R 7-1 may be the same or different.
在本发明某一实施方案中,当R7为未取代或R7-1取代的C1-6烷基时,所述的C1-6烷基为C1-4烷基。In a certain embodiment of the present invention, when R 7 is an unsubstituted or R 7-1 substituted C 1-6 alkyl group, the C 1-6 alkyl group is a C 1-4 alkyl group.
在本发明某一实施方案中,当R7为C2-6烯基时,所述的C2-6烯基为C2-4烯基。In one embodiment of the present invention, when R 7 is C 2-6 alkenyl, said C 2-6 alkenyl is C 2-4 alkenyl.
在本发明某一实施方案中,当R7为C3-10环烷基时,所述的C3-10环烷基为C3-6环烷基。In a certain embodiment of the present invention, when R 7 is C 3-10 cycloalkyl, said C 3-10 cycloalkyl is C 3-6 cycloalkyl.
在本发明某一实施方案中,当R7为(C3-10环烷基)-C1-6烷基时,所述的(C3-10环烷基)-C1-6烷基为(C3-6环烷基)-C1-4烷基。In a certain embodiment of the present invention, when R 7 is (C 3-10 cycloalkyl)-C 1-6 alkyl, the (C 3-10 cycloalkyl)-C 1-6 alkyl is (C 3-6 cycloalkyl)-C 1-4 alkyl.
在本发明某一实施方案中,当R7为未取代或R7-2取代的C6-10芳基时,所述的R7-2的个数为一个或多个,当存在多个R7-2时,所述的R7-2可相同或不同。In a certain embodiment of the present invention, when R 7 is an unsubstituted or R 7-2 substituted C 6-10 aryl group, the number of said R 7-2 is one or more, when there are multiple When R 7-2 , the R 7-2 may be the same or different.
在本发明某一实施方案中,当R7为未取代或R7-2取代的C6-10芳基时,所述的C6-10芳基为苯基。In a certain embodiment of the present invention, when R 7 is an unsubstituted or R 7-2 substituted C 6-10 aryl group, the C 6-10 aryl group is phenyl.
在本发明某一实施方案中,当R7为未取代或R7-3取代的C6-10芳基-C1-6烷基时,所述的R7-3的个数为一个或多个,当存在多个R7-3时,所述的R7-3可相同或不同。In a certain embodiment of the present invention, when R 7 is an unsubstituted or R 7-3 substituted C 6-10 aryl-C 1-6 alkyl group, the number of said R 7-3 is one or a Multiple, when multiple R 7-3s are present, the R 7-3s may be the same or different.
在本发明某一实施方案中,当R7为未取代或R7-3取代的C6-10芳基-C1-6烷基时,所述的C6-10芳基-C1-6烷基为苯基-C1-4烷基。In a certain embodiment of the present invention, when R 7 is unsubstituted or R 7-3 substituted C 6-10 aryl-C 1-6 alkyl, the C 6-10 aryl-C 1-6 alkyl 6Alkyl is phenyl- C1-4alkyl .
在本发明某一实施方案中,当R7为未取代或R7-4取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-C1-6烷基时,所述的R7-4的个数为一个或多个,当存在多个R7-4时,所述的R7-4可相同或不同。In a certain embodiment of the present invention, when R 7 is unsubstituted or R 7-4 substituted "heteroatoms are selected from one or more of N, O and S, and the number of heteroatoms is 1-3" In the case of a 5-10-membered heteroaryl-C 1-6 alkyl group, the number of the R 7-4 is one or more, and when there are multiple R 7-4 , the R 7-4 can be same or different.
在本发明某一实施方案中,当R7为未取代或R7-4取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-C1-6烷基时,所述的5~10元杂芳基-C1-6烷基为5~6元杂芳基-C1-4烷基。In a certain embodiment of the present invention, when R 7 is unsubstituted or R 7-4 substituted "heteroatoms are selected from one or more of N, O and S, and the number of heteroatoms is 1-3" In the case of a 5-10-membered heteroaryl-C 1-6 alkyl group, the 5-10-membered heteroaryl-C 1-6 alkyl group is a 5-6-membered heteroaryl-C 1-4 alkyl group.
在本发明某一实施方案中,当R7-1为卤素时,所述的卤素为氟、氯、溴或碘。In one embodiment of the present invention, when R 7-1 is halogen, the halogen is fluorine, chlorine, bromine or iodine.
在本发明某一实施方案中,当R7-1为C1-4烷基时,所述的C1-4烷基为甲基、乙基或丙基。In a certain embodiment of the present invention, when R 7-1 is C 1-4 alkyl, said C 1-4 alkyl is methyl, ethyl or propyl.
在本发明某一实施方案中,当R7-2~R7-4独立地选自卤素时,所述的卤素为氟、氯、溴或碘。In one embodiment of the present invention, when R 7-2 to R 7-4 are independently selected from halogen, the halogen is fluorine, chlorine, bromine or iodine.
在本发明某一实施方案中,当R7-2~R7-4独立地选自未取代或R7-2-1取代的C1-4烷基时,所述的R7-2-1的个数为一个或多个,当存在多个R7-2-1时,所述的R7-2-1可相同或不同。In a certain embodiment of the present invention, when R 7-2 to R 7-4 are independently selected from unsubstituted or R 7-2-1 substituted C 1-4 alkyl, the R 7-2- The number of 1 is one or more, and when there are multiple R 7-2-1s , the R 7-2-1s may be the same or different.
在本发明某一实施方案中,当R7-2~R7-4独立地选自未取代或R7-2-1取代的C1-4烷基时,所述的C1-4烷基为甲基、乙基、丙基或异丙基。In a certain embodiment of the present invention, when R 7-2 to R 7-4 are independently selected from unsubstituted or R 7-2-1 substituted C 1-4 alkyl, the C 1-4 alkane group is methyl, ethyl, propyl or isopropyl.
在本发明某一实施方案中,当R7-2~R7-4独立地选自未取代或R7-2-2取代的C1-4烷氧基时,所述的R7-2-2的个数为一个或多个,当存在多个R7-2-2时,所述的R7-2-2可相同或不同。In a certain embodiment of the present invention, when R 7-2 to R 7-4 are independently selected from unsubstituted or R 7-2-2 substituted C 1-4 alkoxy, the R 7-2 The number of -2 is one or more, and when there are multiple R 7-2-2s , the R 7-2-2s may be the same or different.
在本发明某一实施方案中,当R7-2~R7-4独立地选自未取代或R7-2-2取代的C1-4烷氧基时,所述的C1-4烷氧基为甲氧基、乙氧基、丙氧基或异丙氧基。In a certain embodiment of the present invention, when R 7-2 to R 7-4 are independently selected from unsubstituted or R 7-2-2 substituted C 1-4 alkoxy, the C 1-4 Alkoxy is methoxy, ethoxy, propoxy or isopropoxy.
在本发明某一实施方案中,当R7-2-3~R7-2-10独立地选自C1-4烷基,所述的C1-4烷基为甲基、乙基、丙基或异丙基。In a certain embodiment of the present invention, when R 7-2-3 to R 7-2-10 are independently selected from C 1-4 alkyl, the C 1-4 alkyl is methyl, ethyl, propyl or isopropyl.
在本发明某一实施方案中,当R8-1为未取代或R8-1-1取代的C1-6烷基时,所述的R8-1-1的个数为一个或多个,当存在多个R8-1-1时,所述的R8-1-1可相同或不同。In a certain embodiment of the present invention, when R 8-1 is an unsubstituted or R 8-1-1 substituted C 1-6 alkyl group, the number of said R 8-1-1 is one or more Each, when there are multiple R 8-1-1s , the R 8-1-1s may be the same or different.
在本发明某一实施方案中,当R8-1为未取代或R8-1-1取代的C1-6烷基时,所述的C1-6烷基为C1-4烷基。In a certain embodiment of the present invention, when R 8-1 is unsubstituted or R 8-1-1 substituted C 1-6 alkyl, the C 1-6 alkyl is C 1-4 alkyl .
在本发明某一实施方案中,当R8-1为C2-6烯基时,所述的C2-6烯基为C2-4烯基。In a certain embodiment of the present invention, when R 8-1 is C 2-6 alkenyl, said C 2-6 alkenyl is C 2-4 alkenyl.
在本发明某一实施方案中,当R8-1为C3-10环烷基时,所述的C3-10环烷基为C3-6环烷基。In a certain embodiment of the present invention, when R 8-1 is a C 3-10 cycloalkyl group, the C 3-10 cycloalkyl group is a C 3-6 cycloalkyl group.
在本发明某一实施方案中,当R8-1为(C3-10环烷基)-C1-6烷基时,所述的(C3-10环烷基)-C1-6烷基为(C3-6环烷基)-C1-4烷基。In a certain embodiment of the present invention, when R 8-1 is (C 3-10 cycloalkyl)-C 1-6 alkyl, the (C 3-10 cycloalkyl)-C 1-6 The alkyl group is (C 3-6 cycloalkyl)-C 1-4 alkyl.
在本发明某一实施方案中,当R8-1为未取代或R8-1-3取代的C6-10芳基-C1-6烷基时,所述的R8-1-3的个数为一个或多个,当存在多个R8-1-3时,所述的R8-1-3可相同或不同。In a certain embodiment of the present invention, when R 8-1 is unsubstituted or R 8-1-3 substituted C 6-10 aryl-C 1-6 alkyl, the R 8-1-3 The number of R 8-1-3 is one or more, and when there are multiple R 8-1-3 , the R 8-1-3 can be the same or different.
在本发明某一实施方案中,当R8-1为未取代或R8-1-3取代的C6-10芳基-C1-6烷基时,所述的C6-10芳基-C1-6烷基为苯基-C1-4烷基。In a certain embodiment of the present invention, when R 8-1 is unsubstituted or R 8-1-3 substituted C 6-10 aryl-C 1-6 alkyl, the C 6-10 aryl -C 1-6 alkyl is phenyl-C 1-4 alkyl.
在本发明某一实施方案中,当R8-1为未取代或R8-1-4取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-C1-6烷基时,所述的R8-1-4的个数为一个或多个,当存在多个R8-1-4时,所述的R8-1-4可相同或不同。In a certain embodiment of the present invention, when R 8-1 is unsubstituted or R 8-1-4 substituted "heteroatom is selected from one or more of N, O and S, the number of heteroatoms is 1- When there are 3 "5-10-membered heteroaryl-C 1-6 alkyl groups, the number of said R 8-1-4 is one or more, and when there are multiple R 8-1-4 , Said R 8-1-4 may be the same or different.
在本发明某一实施方案中,当R8-1为未取代或R8-1-4取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-C1-6烷基时,所述的5~10元杂芳基-C1-6烷基为5~6元杂芳基-C1-4烷基。In a certain embodiment of the present invention, when R 8-1 is unsubstituted or R 8-1-4 substituted "heteroatom is selected from one or more of N, O and S, the number of heteroatoms is 1- When there are 3 "5-10-membered heteroaryl-C 1-6 alkyl groups, the 5-10-membered heteroaryl-C 1-6 alkyl groups are 5-6-membered heteroaryl-C 1-4 groups. alkyl.
在本发明某一实施方案中,当R8-1-1为卤素时,所述的卤素为氟、氯、溴或碘。In a certain embodiment of the present invention, when R 8-1-1 is halogen, the halogen is fluorine, chlorine, bromine or iodine.
在本发明某一实施方案中,当R8-1-3为卤素时,所述的卤素为氟、氯、溴或碘。In one embodiment of the present invention, when R 8-1-3 is halogen, said halogen is fluorine, chlorine, bromine or iodine.
在本发明某一实施方案中,当R8-1-4为卤素时,所述的卤素为氟、氯、溴或碘。In one embodiment of the present invention, when R 8-1-4 is halogen, the halogen is fluorine, chlorine, bromine or iodine.
在本发明某一实施方案中,当R8-1-6为C1-6烷基时,所述的C1-6烷基为C1-4烷基。In a certain embodiment of the present invention, when R 8-1-6 is C 1-6 alkyl, said C 1-6 alkyl is C 1-4 alkyl.
在本发明某一实施方案中,当R9~R11独立地选自C1-6烷基时,所述的C1-6烷基为C1-4烷基。In a certain embodiment of the present invention, when R 9 to R 11 are independently selected from C 1-6 alkyl groups, the C 1-6 alkyl groups are C 1-4 alkyl groups.
在本发明某一实施方案中,当R12为C1-6烷氧基时,所述的C1-6烷氧基为C1-4烷氧基。In a certain embodiment of the present invention, when R 12 is C 1-6 alkoxy, said C 1-6 alkoxy is C 1-4 alkoxy.
在本发明某一实施方案中,当R12-1~R12-5独立选自C1-6烷基时,所述的C1-6烷基为C1-4烷基。In a certain embodiment of the present invention, when R 12-1 to R 12-5 are independently selected from C 1-6 alkyl, the C 1-6 alkyl is C 1-4 alkyl.
在本发明某一实施方案中,当X为对,R5为-(C=O)OR5-1、-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8。In one embodiment of the present invention, when X is Yes, R 5 is -(C=O)OR 5-1 , -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 .
在本发明某一实施方案中,当X为时,R5为-(C=O)OR5-1、-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8。In one embodiment of the present invention, when X is , R 5 is -(C=O)OR 5-1 , -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 .
在本发明某一实施方案中,当X为时,R5独立地选自-(C=O)OR5-1。In one embodiment of the present invention, when X is , R 5 is independently selected from -(C=O)OR 5-1 .
在本发明某一实施方案中,R15和R16独立地选自卤素时,所述的卤素为氟、氯、溴或碘。In one embodiment of the present invention, when R 15 and R 16 are independently selected from halogen, said halogen is fluorine, chlorine, bromine or iodine.
在本发明某一实施方案中,当R15和R16独立地选自C1-6烷基时,所述的C1-6烷基为C1-4烷基。In a certain embodiment of the present invention, when R 15 and R 16 are independently selected from C 1-6 alkyl, said C 1-6 alkyl is C 1-4 alkyl.
在本发明某一实施方案中,n为1或2整数。In a certain embodiment of the present invention, n is an integer of 1 or 2.
在本发明某一实施方案中,当R3和R4独立地选自C1-6烷氧基时,所述的C1-6烷氧基为甲氧基、乙氧基、丙氧基、异丙氧基、正丁氧基、仲丁氧基、异丁氧基或叔丁氧基。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from C 1-6 alkoxy, the C 1-6 alkoxy is methoxy, ethoxy, propoxy , isopropoxy, n-butoxy, sec-butoxy, isobutoxy or tert-butoxy.
在本发明某一实施方案中,当R3和R4独立地选自C1-6卤代烷氧基时,所述的C1-6卤代烷氧基为三氟甲氧基。In one embodiment of the present invention, when R 3 and R 4 are independently selected from C 1-6 haloalkoxy, said C 1-6 haloalkoxy is trifluoromethoxy.
在本发明某一实施方案中,当R3和R4独立地选自C2-6烯基时,所述的C2-6烯基为 In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from C 2-6 alkenyl, said C 2-6 alkenyl is
在本发明某一实施方案中,当R3和R4独立地选自(C3-10环烷基)-氧基时,所述的(C3-10环烷基)-氧基为环丙氧基、环丁氧基、环戊氧基或环己氧基。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from (C 3-10 cycloalkyl)-oxy, said (C 3-10 cycloalkyl)-oxy is a ring Propoxy, cyclobutoxy, cyclopentyloxy or cyclohexyloxy.
在本发明某一实施方案中,当R3和R4独立地选自(C3-10环烷基)-(C1-6烷氧基)时,所述的(C3-10环烷基)-(C1-6烷氧基)为环丙甲氧基、环丙乙氧基、环丙丙氧基、环丙丁氧基、环丁甲氧基、环丁乙氧基、环戊甲氧基、环戊乙氧基、环己甲氧基、或、环己乙氧基。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from (C 3-10 cycloalkyl)-(C 1-6 alkoxy), the (C 3-10 cycloalkane) base)-(C 1-6 alkoxy) is cyclopropylmethoxy, cyclopropylethoxy, cyclopropylpropyloxy, cyclopropylbutoxy, cyclobutylmethoxy, cyclobutylethoxy, cyclopentyl Methoxy, cyclopentethoxy, cyclohexylmethoxy, or, cyclohexylethoxy.
在本发明某一实施方案中,当R3和R4独立地选自“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”4~10元杂环烷基-(C1-6烷氧基)时,所述的4~10元杂环烷基-(C1-6烷氧基)为哌啶甲基、哌啶乙基、哌啶丙基、哌啶丁基、吗啉甲基、吗啉乙基、吗啉丙基、吗啉丁基、氮杂环丁烷甲基、氮杂环丁烷乙基、氮杂环丁烷丙基、氮杂环丁烷丁基、哌嗪甲基、哌嗪乙基、哌嗪丙基、哌嗪丁基、吡咯烷基甲基、吡咯烷基乙基、吡咯烷基丙基、或、吡咯烷基丁基。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from "hetero atoms are selected from one or more of N, O and S, and the number of hetero atoms is 1-3" 4-10 In the case of membered heterocycloalkyl-(C 1-6 alkoxy), the 4-10-membered heterocycloalkyl-(C 1-6 alkoxy) is piperidinylmethyl, piperidinylethyl, piperidine pyridylpropyl, piperidinylbutyl, morpholinomethyl, morpholinoethyl, morpholinopropyl, morpholinobutyl, azetidine methyl, azetidine ethyl, azetidine propyl, azetidinebutyl, piperazinemethyl, piperazineethyl, piperazinepropyl, piperazinebutyl, pyrrolidinylmethyl, pyrrolidinylethyl, pyrrolidinylpropyl, or , pyrrolidinyl butyl.
在本发明某一实施方案中,当R3和R4独立地选自(C6-10芳基)-(C1-6烷氧基)时,所述的(C6-10芳基)-(C1-6烷氧基)为苄氧基、苯乙基氧基、苯丙基氧基、或、苯丁基氧基。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from (C 6-10 aryl)-(C 1-6 alkoxy), the (C 6-10 aryl) -(C 1-6 alkoxy) is benzyloxy, phenethyloxy, phenpropyloxy, or, phenbutyloxy.
在本发明某一实施方案中,当R3和R4独立地选自“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-氧基时,所述的5~10元杂芳基-氧基为吡啶氧基、嘧啶氧基、吡嗪氧基、哒嗪氧基、吡咯氧基、咪唑氧基或吡唑氧基。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from "hetero atoms are selected from one or more of N, O and S, and the number of hetero atoms is 1-3" 5-10 In the case of a heteroaryl-oxy group, the 5- to 10-membered heteroaryl-oxy group is a pyridyloxy group, a pyrimidinyloxy group, a pyrazinyloxy group, a pyridazinoxy group, a pyrrolyloxy group, an imidazolyloxy group or a pyridyloxy group oxazolyl.
在本发明某一实施方案中,当R3和R4独立地选自“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-(C1-6烷氧基)时,所述的5~10元杂芳基-(C1-6烷氧基)为吡啶甲氧基、吡唑乙氧基、嘧啶甲氧基、吡嗪甲氧基、哒嗪甲氧基、吡咯甲氧基、咪唑甲氧基或吡唑甲氧基。In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from "hetero atoms are selected from one or more of N, O and S, and the number of hetero atoms is 1-3" 5-10 In the case of heteroaryl-(C 1-6 alkoxy), the 5-10-membered heteroaryl-(C 1-6 alkoxy) is pyridinemethoxy, pyrazoleethoxy, pyrimidine methyl oxy, pyrazinemethoxy, pyridazinemethoxy, pyrrolemethoxy, imidazolemethoxy or pyrazolemethoxy.
在本发明某一实施方案中,当R3和R4与它们相连的碳原子一起组成4-20元杂环烷氧基时,所述的4-20元杂环烷基为二噁戊烷基、二噁己烷基、12-冠醚-4、15-冠醚-5、或、18冠醚-6。In a certain embodiment of the present invention, when R 3 and R 4 together with the carbon atoms to which they are attached form a 4-20-membered heterocycloalkoxy group, the 4-20-membered heterocycloalkyl group is dioxopentane 12-crown-4, 15-crown-5, or 18-crown-6.
在本发明某一实施方案中,当R5-1为C1-6烷基时,所述的C1-6烷基为甲基、乙基、丙基、异丙基、正丁基、异丁基、仲丁基或叔丁基。In a certain embodiment of the present invention, when R 5-1 is C 1-6 alkyl, the C 1-6 alkyl is methyl, ethyl, propyl, isopropyl, n-butyl, isobutyl, sec-butyl or tert-butyl.
在本发明某一实施方案中,当R5-1为(C1-6烷氧基)-(C1-6烷基)-时,所述的(C1-6烷氧基)-(C1-6烷基)-为甲氧基-甲基-、乙氧基-甲基-、丙氧基-甲基-、异丙氧基-甲基-、正丁氧基-甲基-、异丁基氧基-甲基、仲丁氧基-甲基、或、叔丁氧基-甲基-。In a certain embodiment of the present invention, when R 5-1 is (C 1-6 alkoxy)-(C 1-6 alkyl)-, the (C 1-6 alkoxy)-( C 1-6 alkyl)-is methoxy-methyl-, ethoxy-methyl-, propoxy-methyl-, isopropoxy-methyl-, n-butoxy-methyl- , isobutyloxy-methyl, sec-butoxy-methyl, or, tert-butoxy-methyl-.
在本发明某一实施方案中,当R5-1为C3-10环烷基时,所述的C3-10环烷基为环丙基、环丁基、环戊基或环己基。In a certain embodiment of the present invention, when R 5-1 is C 3-10 cycloalkyl, the C 3-10 cycloalkyl is cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl.
在本发明某一实施方案中,当R5为未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基时,所述的R5-3的个数为1个、2个或3个。In a certain embodiment of the present invention, when R 5 is unsubstituted or R 5-3 substituted "heteroatoms are selected from one or more of N, O and S, and the number of heteroatoms is 1-3" In the case of a 5- to 10-membered heteroaryl group, the number of the R 5-3 is 1, 2 or 3.
在本发明某一实施方案中,当R5为未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基时,所述的5~10元杂芳基为吡唑基、1,3,4-噁二唑基、1,2,4-噁二唑基、1,3,4-噻二唑基、或、1,2,4-噻二唑基。In a certain embodiment of the present invention, when R 5 is unsubstituted or R 5-3 substituted "heteroatoms are selected from one or more of N, O and S, and the number of heteroatoms is 1-3" When 5-10-membered heteroaryl, the 5-10-membered heteroaryl is pyrazolyl, 1,3,4-oxadiazolyl, 1,2,4-oxadiazolyl, 1,3, 4-thiadiazolyl, or, 1,2,4-thiadiazolyl.
在本发明某一实施方案中,当R5-4和R5-5独立地选自C1-6烷基时,所述的C1-6烷基为甲基、乙基、丙基、异丙基、正丁基、异丁基、仲丁基或叔丁基。In a certain embodiment of the present invention, when R 5-4 and R 5-5 are independently selected from C 1-6 alkyl, the C 1-6 alkyl is methyl, ethyl, propyl, isopropyl, n-butyl, isobutyl, sec-butyl or tert-butyl.
在本发明某一实施方案中,当R7为未取代或R7-1取代的C1-6烷基时,所述的R7-1的个数为1个、2个或3个。In a certain embodiment of the present invention, when R 7 is an unsubstituted or R 7-1 substituted C 1-6 alkyl group, the number of said R 7-1 is 1, 2 or 3.
在本发明某一实施方案中,当R7为未取代或R7-1取代的C1-6烷基时,所述的C1-6烷基为甲基、乙基、丙基、异丙基、正丁基、异丁基、仲丁基或叔丁基。In a certain embodiment of the present invention, when R 7 is an unsubstituted or R 7-1 substituted C 1-6 alkyl group, the C 1-6 alkyl group is methyl, ethyl, propyl, iso- propyl, n-butyl, isobutyl, sec-butyl or tert-butyl.
在本发明某一实施方案中,当R7为C2-6烯基时,所述的C2-6烯基为 In a certain embodiment of the present invention, when R 7 is C 2-6 alkenyl, said C 2-6 alkenyl is
在本发明某一实施方案中,当R7为C3-10环烷基时,所述的C3-10环烷基为环丙基、环丁基、环戊基或环己基。In a certain embodiment of the present invention, when R 7 is C 3-10 cycloalkyl, said C 3-10 cycloalkyl is cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl.
在本发明某一实施方案中,当R7为(C3-10环烷基)-C1-6烷基时,所述的(C3-10环烷基)-C1-6烷基为环丙甲基、环丁甲基、环戊甲基或环己甲基。In a certain embodiment of the present invention, when R 7 is (C 3-10 cycloalkyl)-C 1-6 alkyl, the (C 3-10 cycloalkyl)-C 1-6 alkyl is cyclopropylmethyl, cyclobutylmethyl, cyclopentylmethyl or cyclohexylmethyl.
在本发明某一实施方案中,当R7为未取代或R7-2取代的C6-10芳基时,所述的R7-2的个数为1个、2个或3个。In a certain embodiment of the present invention, when R 7 is an unsubstituted or R 7-2 substituted C 6-10 aryl group, the number of said R 7-2 is 1, 2 or 3.
在本发明某一实施方案中,当R7为未取代或R7-3取代的C6-10芳基-C1-6烷基时,所述的R7-3的个数为1个、2个或3个。In a certain embodiment of the present invention, when R 7 is an unsubstituted or R 7-3 substituted C 6-10 aryl-C 1-6 alkyl group, the number of said R 7-3 is 1 , 2 or 3.
在本发明某一实施方案中,当R7为未取代或R7-3取代的C6-10芳基-C1-6烷基时,所述的C6-10芳基-C1-6烷基为苄基。In a certain embodiment of the present invention, when R 7 is unsubstituted or R 7-3 substituted C 6-10 aryl-C 1-6 alkyl, the C 6-10 aryl-C 1-6 alkyl 6Alkyl is benzyl.
在本发明某一实施方案中,当R7为未取代或R7-4取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-C1-6烷基时,所述的R7-4的个数为1个、2个或3个。In a certain embodiment of the present invention, when R 7 is unsubstituted or R 7-4 substituted "heteroatoms are selected from one or more of N, O and S, and the number of heteroatoms is 1-3" In the case of a 5- to 10-membered heteroaryl-C 1-6 alkyl group, the number of the R 7-4 is 1, 2 or 3.
在本发明某一实施方案中,当R7为未取代或R7-4取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-C1-6烷基时,所述的5~10元杂芳基-C1-6烷基为吡啶甲基、嘧啶甲基、吡嗪甲基、哒嗪甲基、吡唑甲基、吡咯甲基、噻吩甲基、呋喃甲基、噻唑甲基或咪唑甲基。In a certain embodiment of the present invention, when R 7 is unsubstituted or R 7-4 substituted "heteroatoms are selected from one or more of N, O and S, and the number of heteroatoms is 1-3" When 5-10-membered heteroaryl-C 1-6 alkyl group, the 5-10-membered heteroaryl-C 1-6 alkyl group is pyridine methyl, pyrimidine methyl, pyrazine methyl, pyridazine methyl group, pyrazolemethyl, pyrrolemethyl, thienylmethyl, furylmethyl, thiazolemethyl or imidazolemethyl.
在本发明某一实施方案中,当R7-2~R7-4独立地选自未取代或R7-2-1取代的C1-4烷基时,所述的R7-2-1的个数为1个、2个或3个。In a certain embodiment of the present invention, when R 7-2 to R 7-4 are independently selected from unsubstituted or R 7-2-1 substituted C 1-4 alkyl, the R 7-2- The number of 1s is 1, 2 or 3.
在本发明某一实施方案中,当R7-2~R7-4独立地选自未取代或R7-2-2取代的C1-4烷氧基时,所述的R7-2-2的个数为为1个、2个或3个。In a certain embodiment of the present invention, when R 7-2 to R 7-4 are independently selected from unsubstituted or R 7-2-2 substituted C 1-4 alkoxy, the R 7-2 The number of -2 is 1, 2 or 3.
在本发明某一实施方案中,当R7-2~R7-4独立地选自-NR7-2-3R7-2-4时,所述的-NR7-2- 3R7-2-4为-NH2、或、 In a certain embodiment of the present invention, when R 7-2 to R 7-4 are independently selected from -NR 7-2-3 R 7-2-4 , the -NR 7-2- 3 R 7 -2-4 is -NH 2 , or,
在本发明某一实施方案中,当R7-2~R7-4独立地选自-(C=O)R7-2-5时,所述的-(C=O)R7-2-5为 In a certain embodiment of the present invention, when R 7-2 to R 7-4 are independently selected from -(C=O)R 7-2-5 , the -(C=O)R 7-2 -5 for
在本发明某一实施方案中,当R7-2~R7-4独立地选自-(C=O)NR7-2-6R7-2-7时,所述的-(C=O)NR7-2-6R7-2-7为 In a certain embodiment of the present invention, when R 7-2 to R 7-4 are independently selected from -(C=O)NR 7-2-6 R 7-2-7 , the -(C= O) NR 7-2-6 R 7-2-7 is
在本发明某一实施方案中,当R7-2~R7-4独立地选自-(C=O)OR7-2-8时,所述的-(C=O)OR7-2-8为-COOH、或、 In a certain embodiment of the present invention, when R 7-2 to R 7-4 are independently selected from -(C=O)OR 7-2-8 , the -(C=O)OR 7-2 -8 is -COOH, or,
在本发明某一实施方案中,当R7-2~R7-4独立地选自-S(=O)2NR7-2-9R7-2-10时,所述的-S(=O)2NR7-2-9R7-2-10为 In a certain embodiment of the present invention, when R 7-2 to R 7-4 are independently selected from -S(=O) 2 NR 7-2-9 R 7-2-10 , the -S( =O) 2 NR 7-2-9 R 7-2-10 is
在本发明某一实施方案中,当R8-1为未取代或R8-1-1取代的C1-6烷基时,所述的R8-1-1的个数为1个、2个或3个。In a certain embodiment of the present invention, when R 8-1 is an unsubstituted or R 8-1-1 substituted C 1-6 alkyl group, the number of said R 8-1-1 is 1, 2 or 3.
在本发明某一实施方案中,当R8-1为未取代或R8-1-1取代的C1-6烷基时,所述的C1-6烷基为甲基、乙基、丙基、异丙基、正丁基、异丁基、仲丁基或叔丁基。In a certain embodiment of the present invention, when R 8-1 is unsubstituted or R 8-1-1 substituted C 1-6 alkyl, the C 1-6 alkyl is methyl, ethyl, propyl, isopropyl, n-butyl, isobutyl, sec-butyl or tert-butyl.
在本发明某一实施方案中,当R8-1为C2-6烯基时,所述的C2-6烯基为 In a certain embodiment of the present invention, when R 8-1 is C 2-6 alkenyl, the C 2-6 alkenyl is
在本发明某一实施方案中,当R8-1为C3-10环烷基时,所述的C3-10环烷基为环丙基、环丁基、环戊基或环己基。In a certain embodiment of the present invention, when R 8-1 is C 3-10 cycloalkyl, the C 3-10 cycloalkyl is cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl.
在本发明某一实施方案中,当R8-1为(C3-10环烷基)-C1-6烷基时,所述的(C3-10环烷基)-C1-6烷基为环丙甲基、环丁甲基、环戊甲基、或、环己甲基。In a certain embodiment of the present invention, when R 8-1 is (C 3-10 cycloalkyl)-C 1-6 alkyl, the (C 3-10 cycloalkyl)-C 1-6 Alkyl is cyclopropylmethyl, cyclobutylmethyl, cyclopentylmethyl, or cyclohexylmethyl.
在本发明某一实施方案中,当R8-1为未取代或R8-1-3取代的C6-10芳基-C1-6烷基时,所述的R8-1-3的个数为1个、2个或3个。In a certain embodiment of the present invention, when R 8-1 is unsubstituted or R 8-1-3 substituted C 6-10 aryl-C 1-6 alkyl, the R 8-1-3 The number is 1, 2 or 3.
在本发明某一实施方案中,当R8-1为未取代或R8-1-3取代的C6-10芳基-C1-6烷基时,所述的C6-10芳基-C1-6烷基为苄基或苯乙基。In a certain embodiment of the present invention, when R 8-1 is unsubstituted or R 8-1-3 substituted C 6-10 aryl-C 1-6 alkyl, the C 6-10 aryl -C 1-6 alkyl is benzyl or phenethyl.
在本发明某一实施方案中,当R8-1为未取代或R8-1-4取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-C1-6烷基时,所述的R8-1-4的个数为1个、2个或3个。In a certain embodiment of the present invention, when R 8-1 is unsubstituted or R 8-1-4 substituted "heteroatom is selected from one or more of N, O and S, the number of heteroatoms is 1- When three "5- to 10-membered heteroaryl-C 1-6 alkyl groups are used, the number of said R 8-1-4 is 1, 2 or 3.
在本发明某一实施方案中,当R8-1为未取代或R8-1-4取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-C1-6烷基时,所述的5~10元杂芳基-C1-6烷基为吡啶甲基、嘧啶甲基、吡嗪甲基、哒嗪甲基、吡唑甲基、吡咯甲基、噻吩甲基、呋喃甲基、噻唑甲基或咪唑甲基。In a certain embodiment of the present invention, when R 8-1 is unsubstituted or R 8-1-4 substituted "heteroatom is selected from one or more of N, O and S, the number of heteroatoms is 1- When there are 3 "5-10-membered heteroaryl-C 1-6 alkyl groups, the 5-10-membered heteroaryl-C 1-6 alkyl groups are pyridine methyl, pyrimidine methyl, pyrazine methyl , pyridazine methyl, pyrazole methyl, pyrrole methyl, thienyl methyl, furyl methyl, thiazole methyl or imidazole methyl.
在本发明某一实施方案中,当R8-1-6为C1-6烷基时,所述的C1-6烷基为甲基、乙基、丙基、异丙基、正丁基、异丁基、仲丁基或叔丁基。In a certain embodiment of the present invention, when R 8-1-6 is C 1-6 alkyl, the C 1-6 alkyl is methyl, ethyl, propyl, isopropyl, n-butyl , isobutyl, sec-butyl or tert-butyl.
在本发明某一实施方案中,当R9~R11独立地选自C1-6烷基时,所述的C1-6烷基为甲基、乙基、丙基、异丙基、正丁基、异丁基、仲丁基或叔丁基。In a certain embodiment of the present invention, when R 9 to R 11 are independently selected from C 1-6 alkyl, the C 1-6 alkyl is methyl, ethyl, propyl, isopropyl, n-butyl, isobutyl, sec-butyl or tert-butyl.
在本发明某一实施方案中,当R12为C1-6烷氧基时,所述的C1-6烷氧基为甲氧基、乙氧基、丙氧基、异丙氧基、正丁氧基、异丁氧基、仲丁氧基或叔丁氧基。In a certain embodiment of the present invention, when R 12 is C 1-6 alkoxy, the C 1-6 alkoxy is methoxy, ethoxy, propoxy, isopropoxy, n-butoxy, isobutoxy, sec-butoxy or tert-butoxy.
在本发明某一实施方案中,当R12-1~R12-5独立选自C1-6烷基时,所述的C1-6烷基为甲基、乙基、丙基、异丙基、正丁基、异丁基、仲丁基或叔丁基。In a certain embodiment of the present invention, when R 12-1 to R 12-5 are independently selected from C 1-6 alkyl, the C 1-6 alkyl is methyl, ethyl, propyl, iso- propyl, n-butyl, isobutyl, sec-butyl or tert-butyl.
在本发明某一实施方案中,当R15和R16独立地选自C1-6烷基时,所述的C1-6烷基为甲基、乙基、丙基、异丙基、正丁基、异丁基、仲丁基或叔丁基。In a certain embodiment of the present invention, when R 15 and R 16 are independently selected from C 1-6 alkyl, the C 1-6 alkyl is methyl, ethyl, propyl, isopropyl, n-butyl, isobutyl, sec-butyl or tert-butyl.
在本发明某一实施方案中,当R3和R4独立地选自“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”4~10元杂环烷基-(C1-6烷氧基)时,所述的4~10元杂环烷基-(C1-6烷氧基)为 In a certain embodiment of the present invention, when R 3 and R 4 are independently selected from "hetero atoms are selected from one or more of N, O and S, and the number of hetero atoms is 1-3" 4-10 In the case of membered heterocycloalkyl-(C 1-6 alkoxy), the 4-10-membered heterocycloalkyl-(C 1-6 alkoxy) is
在本发明某一实施方案中,当R3和R4与它们相连的碳原子一起组成“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”4-20元杂环烷基时,所述的4-20元杂环烷基为 In a certain embodiment of the present invention, when R 3 and R 4 together with the carbon atoms to which they are attached form "heteroatoms selected from one or more of N, O and S, the number of heteroatoms is 1-3""4-20-membered heterocycloalkyl, the 4-20-membered heterocycloalkyl is
在本发明某一实施方案中,当R5为未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基时,所述的未取代或R5-3取代的杂芳基为 In a certain embodiment of the present invention, when R 5 is unsubstituted or R 5-3 substituted "hetero atoms are selected from one or more of N, O and S, and the number of hetero atoms is 1-3" In the case of a 5-10-membered heteroaryl group, the unsubstituted or R 5-3 substituted heteroaryl group is
在本发明某一实施方案中,当R7为R7-1取代的C1-6烷基时,所述的R7-1取代的C1-6烷基为三氟甲基、二氟甲基、 In a certain embodiment of the present invention, when R 7 is C 1-6 alkyl substituted by R 7-1 , the C 1-6 alkyl substituted by R 7-1 is trifluoromethyl, difluoro methyl,
在本发明某一实施方案中,当R7为未取代或R7-2取代的C6-10芳基时,所述的未取代或R7-2取代的C6-10芳基为 In a certain embodiment of the present invention, when R 7 is an unsubstituted or R 7-2 substituted C 6-10 aryl group, the unsubstituted or R 7-2 substituted C 6-10 aryl group is
在本发明某一实施方案中,当R7为未取代或R7-3取代的C6-10芳基-C1-6烷基时,所述的未取代或R7-3取代的C6-10芳基-C1-6烷基为 In a certain embodiment of the present invention, when R 7 is unsubstituted or R 7-3 substituted C 6-10 aryl-C 1-6 alkyl, the unsubstituted or R 7-3 substituted C 6-10 aryl-C 1-6 alkyl is
在本发明某一实施方案中,当R7为未取代或R7-4取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-C1-6烷基时,所述的未取代或R7-4取代的杂芳基-C1-6烷基为 In a certain embodiment of the present invention, when R 7 is unsubstituted or R 7-4 substituted "heteroatoms are selected from one or more of N, O and S, and the number of heteroatoms is 1-3" In the case of a 5-10-membered heteroaryl-C 1-6 alkyl group, the unsubstituted or R 7-4 substituted heteroaryl-C 1-6 alkyl group is
在本发明某一实施方案中,当R8-4为R8-1-1取代的C1-6烷基时,所述的R8-1-1取代的C1-6烷基为 In a certain embodiment of the present invention, when R 8-4 is C 1-6 alkyl substituted by R 8-1-1 , the C 1-6 alkyl substituted by R 8-1-1 is
在本发明某一实施方案中,当R8-1为未取代或R8-1-3取代的C6-10芳基-C1-6烷基时,所述的未取代或R8-1-3取代的C6-10芳基-C1-6烷基为 In a certain embodiment of the present invention, when R 8-1 is unsubstituted or R 8-1-3 substituted C 6-10 aryl-C 1-6 alkyl, the unsubstituted or R 8- 1-3 substituted C 6-10 aryl-C 1-6 alkyl is
在本发明某一实施方案中,当R8-1为未取代或R8-1-4取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基-C1-6烷基时,所述的未取代或R8-1-4取代的杂芳基-C1-6烷基为 In a certain embodiment of the present invention, when R 8-1 is unsubstituted or R 8-1-4 substituted "heteroatom is selected from one or more of N, O and S, the number of heteroatoms is 1- 3 "5-10-membered heteroaryl-C 1-6 alkyl, the unsubstituted or R 8-1-4 substituted heteroaryl-C 1-6 alkyl is
在本发明某一实施方案中,R1为氢。In a certain embodiment of the present invention, R1 is hydrogen.
在本发明某一实施方案中,R2为氢、或、卤素。In a certain embodiment of the present invention, R2 is hydrogen , or, halogen.
在本发明某一实施方案中,R3为卤素、C2-6烯基、C1-6烷氧基、或、-NR3-2R3-3。In a certain embodiment of the present invention, R 3 is halogen, C 2-6 alkenyl, C 1-6 alkoxy, or, -NR 3-2 R 3-3 .
在本发明某一实施方案中,R4为卤素、C1-6烷氧基、C2-6烯基、杂环烷基-(C1-6烷氧基)、或、-NR3-2R3-3。In a certain embodiment of the present invention, R 4 is halogen, C 1-6 alkoxy, C 2-6 alkenyl, heterocycloalkyl-(C 1-6 alkoxy), or, -NR 3- 2 R 3-3 .
在本发明某一实施方案中,R3和R4与它们相连的碳原子一起组成4~20元杂环烷氧基。In one embodiment of the present invention, R 3 and R 4 together with the carbon atoms to which they are attached form a 4-20 membered heterocycloalkoxy group.
在本发明某一实施方案中,R3-2为氢或C1-4烷基。In a certain embodiment of the present invention, R 3-2 is hydrogen or C 1-4 alkyl.
在本发明某一实施方案中,R3-3为氢或C1-4烷基。In a certain embodiment of the invention, R 3-3 is hydrogen or C 1-4 alkyl.
在本发明某一实施方案中,R5为-(C=O)OR5-1、未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基、-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8。In a certain embodiment of the present invention, R 5 is -(C=O)OR 5-1 , unsubstituted or R 5-3 substituted "heteroatom selected from one or more of N, O and S, "5-10-membered heteroaryl group with 1-3 heteroatoms, -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 .
在本发明某一实施方案中,R5-1为氢、C1-6烷基、(C1-6烷氧基)-(C1-6烷基)-、或、C3-10环烷基。In a certain embodiment of the present invention, R 5-1 is hydrogen, C 1-6 alkyl, (C 1-6 alkoxy)-(C 1-6 alkyl)-, or, C 3-10 ring alkyl.
在本发明某一实施方案中,R5-4为氢。In a certain embodiment of the present invention, R 5-4 is hydrogen.
在本发明某一实施方案中,R5-5为C1-6烷基。In a certain embodiment of the present invention, R 5-5 is C 1-6 alkyl.
在本发明某一实施方案中,R7为氢、未取代或R7-1取代的C1-6烷基、C2-6烯基、C3-10环烷基、未取代或R7-2取代的C6-10芳基、未取代或R7-3取代的C6-10芳基-C1-6烷基、或、未取代或R7-4取代的杂芳基-C1-6烷基。In a certain embodiment of the present invention, R 7 is hydrogen, unsubstituted or R 7-1 substituted C 1-6 alkyl, C 2-6 alkenyl, C 3-10 cycloalkyl, unsubstituted or R 7 -2- substituted C 6-10 aryl, unsubstituted or R 7-3 substituted C 6-10 aryl-C 1-6 alkyl, or, unsubstituted or R 7-4 substituted heteroaryl-C 1-6 alkyl.
在本发明某一实施方案中,R7-1为卤素、羧基、羟基、或、氰基。In one embodiment of the present invention, R 7-1 is halogen, carboxyl, hydroxyl, or, cyano.
在本发明某一实施方案中,R7-2为卤素、未取代或R7-2-1取代的C1-4烷基、未取代或R7-2-2取代的C1-4烷氧基、或、-NR7-2-3R7-2-4。In a certain embodiment of the present invention, R 7-2 is halogen, unsubstituted or R 7-2-1 substituted C 1-4 alkyl, unsubstituted or R 7-2-2 substituted C 1-4 alkane oxy, or, -NR 7-2-3 R 7-2-4 .
在本发明某一实施方案中,R7-3为卤素、氰基、未取代或R7-2-1取代的C1-4烷基、未取代或R7-2-2取代的C1-4烷氧基、或、-(C=O)NR7-2-6R7-2-7。In a certain embodiment of the present invention, R 7-3 is halogen, cyano, unsubstituted or R 7-2-1 substituted C 1-4 alkyl, unsubstituted or R 7-2-2 substituted C 1 -4alkoxy , or, -(C=O)NR 7-2-6 R 7-2-7 .
在本发明某一实施方案中,R7-4为卤素。In a certain embodiment of the present invention, R 7-4 is halogen.
在本发明某一实施方案中,R7-2-1为卤素。In a certain embodiment of the present invention, R 7-2-1 is halogen.
在本发明某一实施方案中,R7-2-2为卤素。In a certain embodiment of the present invention, R 7-2-2 is halogen.
在本发明某一实施方案中,R7-2-3为氢。In a certain embodiment of the present invention, R 7-2-3 is hydrogen.
在本发明某一实施方案中,R7-2-4为氢。In a certain embodiment of the present invention, R 7-2-4 is hydrogen.
在本发明某一实施方案中,R7-2-6为氢。In a certain embodiment of the present invention, R 7-2-6 is hydrogen.
在本发明某一实施方案中,R7-2-7为氢。In a certain embodiment of the present invention, R 7-2-7 is hydrogen.
在本发明某一实施方案中,R8为氢、氰基、R8-1O-、或、-NR8-2R8-3。In a certain embodiment of the present invention, R 8 is hydrogen, cyano, R 8-1 O-, or, -NR 8-2 R 8-3 .
在本发明某一实施方案中,R8-1为氢、未取代或R8-1-3取代的C6-10芳基-C1-6烷基、未取代或R8-1-4取代的杂芳基-C1-6烷基、或、-(S=O)2OR8-1-6。In a certain embodiment of the invention, R 8-1 is hydrogen, unsubstituted or R 8-1-3 substituted C 6-10 aryl-C 1-6 alkyl, unsubstituted or R 8-1-4 Substituted heteroaryl-C 1-6 alkyl, or, -(S=O) 2 OR 8-1-6 .
在本发明某一实施方案中,R8-1-1为羟基。In a certain embodiment of the present invention, R 8-1-1 is hydroxy.
在本发明某一实施方案中,R8-1-3为卤素。In a certain embodiment of the present invention, R 8-1-3 is halogen.
在本发明某一实施方案中,R8-1-4为卤素。In a certain embodiment of the present invention, R 8-1-4 is halogen.
在本发明某一实施方案中,R8-1-6为氢或C1-6烷基。In a certain embodiment of the present invention, R 8-1-6 is hydrogen or C 1-6 alkyl.
在本发明某一实施方案中,R8-2为氢或C1-6烷基。In a certain embodiment of the present invention, R 8-2 is hydrogen or C 1-6 alkyl.
在本发明某一实施方案中,R8-3为氢或C1-6烷基。In a certain embodiment of the invention, R 8-3 is hydrogen or C 1-6 alkyl.
在本发明某一实施方案中,X为C=O、其中,X的左端与相连,右端与L相连。In an embodiment of the present invention, X is C=O, where the left end of X is the same as connected, and the right end is connected to L.
在本发明某一实施方案中,R10为氢。 In a certain embodiment of the present invention, R10 is hydrogen.
在本发明某一实施方案中,R11为氢。 In a certain embodiment of the present invention, R11 is hydrogen.
在本发明某一实施方案中,R12为羟基、氰基、C1-6烷氧基、-O(C=O)R12-1、-(S=O)2R12-2、或、-NR12-4R12-5。In a certain embodiment of the invention, R 12 is hydroxy, cyano, C 1-6 alkoxy, -O(C=O)R 12-1 , -(S=O) 2 R 12-2 , or , -NR 12-4 R 12-5 .
在本发明某一实施方案中,R12-1为C1-6烷基。In a certain embodiment of the present invention, R 12-1 is C 1-6 alkyl.
在本发明某一实施方案中,R12-2为C1-6烷基。In a certain embodiment of the present invention, R 12-2 is C 1-6 alkyl.
在本发明某一实施方案中,R12-4为氢。In a certain embodiment of the present invention, R 12-4 is hydrogen.
在本发明某一实施方案中,R12-5为氢。In a certain embodiment of the present invention, R 12-5 is hydrogen.
在本发明某一实施方案中,R13为氢。In a certain embodiment of the present invention, R13 is hydrogen.
在本发明某一实施方案中,R14为氢。In a certain embodiment of the present invention, R14 is hydrogen.
在本发明某一实施方案中,当X为对,R5为-(C=O)OR5-1、-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8。In one embodiment of the present invention, when X is Yes, R 5 is -(C=O)OR 5-1 , -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 .
在本发明某一实施方案中,当X为时,R5为-(C=O)OR5-1、-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8。In one embodiment of the present invention, when X is , R 5 is -(C=O)OR 5-1 , -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 .
在本发明某一实施方案中,当X为时,R5独立地选自-(C=O)OR5-1。In one embodiment of the present invention, when X is , R 5 is independently selected from -(C=O)OR 5-1 .
在本发明某一实施方案中,R6为氢。 In a certain embodiment of the present invention, R6 is hydrogen.
在本发明某一实施方案中,R15为氢、卤素、或、C1-6烷基。In a certain embodiment of the present invention, R 15 is hydrogen, halogen, or, C 1-6 alkyl.
在本发明某一实施方案中,R16为氢、卤素、或、C1-6烷基。In a certain embodiment of the present invention, R 16 is hydrogen, halogen, or, C 1-6 alkyl.
在本发明某一实施方案中,In one embodiment of the present invention,
R1为氢;R 1 is hydrogen;
R2为氢、或、卤素;R 2 is hydrogen, or, halogen;
R3为卤素、C2-6烯基、C1-6烷氧基、或、-NR3-2R3-3;R 3 is halogen, C 2-6 alkenyl, C 1-6 alkoxy, or, -NR 3-2 R 3-3 ;
R4为卤素、C1-6烷氧基、C2-6烯基、杂环烷基-(C1-6烷氧基)、-NR3-2R3-3、或、R3和R4与它们相连的碳原子一起组成4~20元杂环烷氧基;R 4 is halogen, C 1-6 alkoxy, C 2-6 alkenyl, heterocycloalkyl-(C 1-6 alkoxy), -NR 3-2 R 3-3 , or, R 3 and R 4 and the carbon atoms to which they are attached together form a 4-20-membered heterocycloalkoxy group;
R3-2为氢或C1-4烷基;R 3-2 is hydrogen or C 1-4 alkyl;
R3-3为氢或C1-4烷基;R 3-3 is hydrogen or C 1-4 alkyl;
R5为-(C=O)OR5-1、未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基、-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8;R 5 is -(C=O)OR 5-1 , unsubstituted or R 5-3 substituted "heteroatom is selected from one or more of N, O and S, and the number of heteroatoms is 1-3. "5-10-membered heteroaryl, -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 ;
R5-1为氢、C1-6烷基、(C1-6烷氧基)-(C1-6烷基)-、或、C3-10环烷基;R 5-1 is hydrogen, C 1-6 alkyl, (C 1-6 alkoxy)-(C 1-6 alkyl)-, or, C 3-10 cycloalkyl;
R5-3为C1-4烷基;R 5-3 is C 1-4 alkyl;
R5-4为氢;R 5-4 is hydrogen;
R5-5为C1-6烷基;R 5-5 is C 1-6 alkyl;
R7为氢、未取代或R7-1取代的C1-6烷基、C2-6烯基、C3-10环烷基、未取代或R7-2取代的C6-10芳基、未取代或R7-3取代的C6-10芳基-C1-6烷基、或、未取代或R7-4取代的杂芳基-C1-6烷基;R 7 is hydrogen, unsubstituted or R 7-1 substituted C 1-6 alkyl, C 2-6 alkenyl, C 3-10 cycloalkyl, unsubstituted or R 7-2 substituted C 6-10 aryl base, unsubstituted or R 7-3 substituted C 6-10 aryl-C 1-6 alkyl, or, unsubstituted or R 7-4 substituted heteroaryl-C 1-6 alkyl;
R7-1为卤素、羧基、羟基、或、氰基;R 7-1 is halogen, carboxyl, hydroxyl, or, cyano;
R7-2为卤素、未取代或R7-2-1取代的C1-4烷基、未取代或R7-2-2取代的C1-4烷氧基、或、-NR7-2-3R7-2-4;R 7-2 is halogen, unsubstituted or R 7-2-1 substituted C 1-4 alkyl, unsubstituted or R 7-2-2 substituted C 1-4 alkoxy, or, -NR 7- 2-3 R 7-2-4 ;
R7-3为卤素、氰基、未取代或R7-2-1取代的C1-4烷基、未取代或R7-2-2取代的C1-4烷氧基、或、-(C=O)NR7-2-6R7-2-7;R 7-3 is halogen, cyano, unsubstituted or R 7-2-1 substituted C 1-4 alkyl, unsubstituted or R 7-2-2 substituted C 1-4 alkoxy, or, - (C=O)NR 7-2-6 R 7-2-7 ;
R7-4为卤素;R 7-4 is halogen;
R7-2-1为卤素;R 7-2-1 is halogen;
R7-2-2为卤素;R 7-2-2 is halogen;
R7-2-3为氢;R 7-2-3 is hydrogen;
R7-2-4为氢;R 7-2-4 is hydrogen;
R7-2-6为氢;R 7-2-6 is hydrogen;
R7-2-7为氢;R 7-2-7 is hydrogen;
R8为氢、氰基、R8-1O-、或、-NR8-2R8-3;R 8 is hydrogen, cyano, R 8-1 O-, or, -NR 8-2 R 8-3 ;
R8-1为氢、未取代或R8-1-3取代的C6-10芳基-C1-6烷基、未取代或R8-1-4取代的杂芳基-C1-6烷基、或、-(S=O)2OR8-1-6;R 8-1 is hydrogen, unsubstituted or R 8-1-3 substituted C 6-10 aryl-C 1-6 alkyl, unsubstituted or R 8-1-4 substituted heteroaryl-C 1- 6 alkyl, or, -(S=O) 2 OR 8-1-6 ;
R8-1-1为羟基;R 8-1-1 is hydroxyl;
R8-1-3为卤素;R 8-1-3 is halogen;
R8-1-4为卤素;R 8-1-4 is halogen;
R8-1-6为氢或C1-6烷基;R 8-1-6 is hydrogen or C 1-6 alkyl;
R8-1-2-3为C1-4烷基;R 8-1-2-3 is C 1-4 alkyl;
R8-1-2-4为氢;R 8-1-2-4 is hydrogen;
R8-1-2-5为氢;R 8-1-2-5 is hydrogen;
R8-2为氢或C1-6烷基;R 8-2 is hydrogen or C 1-6 alkyl;
R8-3为氢或C1-6烷基;R 8-3 is hydrogen or C 1-6 alkyl;
X为C=O、其中,X的左端与相连,右端与L相连;X is C=O, where the left end of X is the same as connected, and the right end is connected to L;
R10为氢;R 10 is hydrogen;
R11为氢;R 11 is hydrogen;
R12为羟基、氰基、C1-6烷氧基、-O(C=O)R12-1、-(S=O)2R12-2、或、-NR12-4R12-5;R 12 is hydroxy, cyano, C 1-6 alkoxy, -O(C=O)R 12-1 , -(S=O) 2 R 12-2 , or, -NR 12-4 R 12- 5 ;
R12-1为C1-6烷基;R 12-1 is C 1-6 alkyl;
R12-2为C1-6烷基;R 12-2 is C 1-6 alkyl;
R12-4为氢;R 12-4 is hydrogen;
R12-5为氢;R 12-5 is hydrogen;
R13为氢;R 13 is hydrogen;
R14为氢;R 14 is hydrogen;
X和R5选自以下任一组合:X and R are selected from any combination of the following:
当X为时,R5为-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8;when X is , R 5 is -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 ;
当X为C=O时,R5为-(C=O)NR5-4(S=O)2R5-5、或、未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基;When X is C=O, R 5 is -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, unsubstituted or R 5-3 substituted "heteroatom is selected from N, One or more of O and S, "5-10-membered heteroaryl group with 1-3 heteroatoms;
当X为时,R5为-(C=O)OR5-1、-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8;when X is , R 5 is -(C=O)OR 5-1 , -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 ;
当X为时,R5为-(C=O)OR5-1、-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8;when X is , R 5 is -(C=O)OR 5-1 , -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 ;
当X为时,R5独立地选自-(C=O)OR5-1;when X is , R 5 is independently selected from -(C=O)OR 5-1 ;
R6为氢;R 6 is hydrogen;
R15为氢、卤素、或、C1-6烷基;R 15 is hydrogen, halogen, or, C 1-6 alkyl;
R16为氢、卤素、或、C1-6烷基。R 16 is hydrogen, halogen, or, C 1-6 alkyl.
在本发明某一实施方案中,In one embodiment of the present invention,
R1为氢;R 1 is hydrogen;
R2为氢、或、卤素;R 2 is hydrogen, or, halogen;
R3为C1-6烷氧基;R 3 is C 1-6 alkoxy;
R4为C1-6烷氧基、杂环烷基-(C1-6烷氧基)、或、R3和R4与它们相连的碳原子一起组成4~20元杂环烷氧基;R 4 is C 1-6 alkoxy, heterocycloalkyl-(C 1-6 alkoxy), or, R 3 and R 4 together with the carbon atoms to which they are attached form a 4-20-membered heterocycloalkoxy group ;
R5为-(C=O)OR5-1、未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基、-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8;R 5 is -(C=O)OR 5-1 , unsubstituted or R 5-3 substituted "heteroatom is selected from one or more of N, O and S, and the number of heteroatoms is 1-3. "5-10-membered heteroaryl, -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 ;
R5-1为氢、C1-6烷基、(C1-6烷氧基)-(C1-6烷基)-、或、C3-10环烷基;R 5-1 is hydrogen, C 1-6 alkyl, (C 1-6 alkoxy)-(C 1-6 alkyl)-, or, C 3-10 cycloalkyl;
R5-3为C1-4烷基;R 5-3 is C 1-4 alkyl;
R5-4为氢;R 5-4 is hydrogen;
R5-5为C1-6烷基;R 5-5 is C 1-6 alkyl;
R7为氢、未取代或R7-1取代的C1-6烷基、C3-10环烷基、未取代或R7-3取代的C6-10芳基-C1-6烷基、或、未取代或R7-4取代的杂芳基-C1-6烷基;R 7 is hydrogen, unsubstituted or R 7-1 substituted C 1-6 alkyl, C 3-10 cycloalkyl, unsubstituted or R 7-3 substituted C 6-10 aryl-C 1-6 alkyl base, or, unsubstituted or R 7-4 substituted heteroaryl-C 1-6 alkyl;
R7-1为卤素、羟基、或、氰基;R 7-1 is halogen, hydroxyl, or, cyano;
R7-3为卤素、氰基、或、未取代或R7-2-1取代的C1-4烷基R 7-3 is halogen, cyano, or, unsubstituted or R 7-2-1 substituted C 1-4 alkyl
R7-4为卤素;R 7-4 is halogen;
R7-2-1为卤素;R 7-2-1 is halogen;
R8为氢、氰基、R8-1O-、或、-NR8-2R8-3;R 8 is hydrogen, cyano, R 8-1 O-, or, -NR 8-2 R 8-3 ;
R8-1为氢、未取代或R8-1-3取代的C6-10芳基-C1-6烷基、或、未取代或R8-1-4取代的杂芳基-C1-6烷基;R 8-1 is hydrogen, unsubstituted or R 8-1-3 substituted C 6-10 aryl-C 1-6 alkyl, or, unsubstituted or R 8-1-4 substituted heteroaryl-C 1-6 alkyl;
R8-1-1为羟基;R 8-1-1 is hydroxyl;
R8-1-3为卤素;R 8-1-3 is halogen;
R8-1-4为卤素;R 8-1-4 is halogen;
X为C=O、其中,X的左端与相连,右端与L相连;X is C=O, where the left end of X is the same as connected, and the right end is connected to L;
R10为氢;R 10 is hydrogen;
R11为氢;R 11 is hydrogen;
R12为羟基、氰基、C1-6烷氧基、-O(C=O)R12-1、-(S=O)2R12-2、或、-NR12-4R12-5;R 12 is hydroxy, cyano, C 1-6 alkoxy, -O(C=O)R 12-1 , -(S=O) 2 R 12-2 , or, -NR 12-4 R 12- 5 ;
R12-1为C1-6烷基;R 12-1 is C 1-6 alkyl;
R12-2为C1-6烷基;R 12-2 is C 1-6 alkyl;
R12-4为氢;R 12-4 is hydrogen;
R12-5为氢;R 12-5 is hydrogen;
R13为氢;R 13 is hydrogen;
R14为氢;R 14 is hydrogen;
X和R5选自以下任一组合:X and R are selected from any combination of the following:
当X为时,R5为-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8;when X is , R 5 is -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 ;
当X为C=O时,R5为-(C=O)NR5-4(S=O)2R5-5、或、未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基;When X is C=O, R 5 is -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, unsubstituted or R 5-3 substituted "heteroatom is selected from N, One or more of O and S, "5-10-membered heteroaryl group with 1-3 heteroatoms;
当X为时,R5为-(C=O)OR5-1、-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8;when X is , R 5 is -(C=O)OR 5-1 , -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 ;
当X为时,R5为-(C=O)OR5-1、-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8;when X is , R 5 is -(C=O)OR 5-1 , -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 ;
当X为时,R5独立地选自-(C=O)OR5-1;when X is , R 5 is independently selected from -(C=O)OR 5-1 ;
R6为氢;R 6 is hydrogen;
R15为氢、卤素、或、C1-6烷基;R 15 is hydrogen, halogen, or, C 1-6 alkyl;
R16为氢、卤素、或、C1-6烷基。R 16 is hydrogen, halogen, or, C 1-6 alkyl.
在本发明某一实施方案中,所述的式I所示化合物可为以下任一结构:In a certain embodiment of the present invention, the compound shown in the described formula I can be any of the following structures:
本发明的第二方面,提供一种本发明第一方面所述化合物的制备方法,其包括以下步骤:The second aspect of the present invention provides a preparation method of the compound described in the first aspect of the present invention, which comprises the following steps:
(1)当X为时,R5为-(C=O)NR7R8、或、-(C=O)NR5-4(S=O)2R5-5时,(1) When X is When R 5 is -(C=O)NR 7 R 8 , or, -(C=O)NR 5-4 (S=O) 2 R 5-5 ,
其中,in,
R17为氢或C1-6烷基;R 17 is hydrogen or C 1-6 alkyl;
R18为R7,R19为R8;或者,R18为R5-4,R19为R5-5;R 18 is R 7 and R 19 is R 8 ; alternatively, R 18 is R 5-4 and R 19 is R 5-5 ;
R1、R2、R3、R4、R5-4、R5-5、R6、R7、R8、R10和L的定义本发明第一方面所限定;Definitions of R 1 , R 2 , R 3 , R 4 , R 5-4 , R 5-5 , R 6 , R 7 , R 8 , R 10 and L as defined in the first aspect of the present invention;
步骤a:在溶剂中,化合物II-1和化合物III-1发生反应生成IV;Step a: in a solvent, compound II-1 and compound III-1 react to generate IV;
步骤b:在溶剂中,化合物IV和化合物V发生反应生成化合物I-1;Step b: in a solvent, compound IV and compound V react to generate compound I-1;
方法二,包括步骤:Method two, including steps:
(2)当X为R5为-(C=O)OR5-1、-(C=O)SR5-2、未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基、-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8时,(2) When X is R 5 is -(C=O)OR 5-1 , -(C=O)SR 5-2 , unsubstituted or R 5-3 substituted "heteroatom selected from one or more of N, O and S Species, "5-10-membered heteroaryl with 1-3 heteroatoms, -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 ,
其中,in,
Y为-NHR11、或、-NHR13;Y is -NHR 11 , or, -NHR 13 ;
R18为R7,R19为R8;或者,R18为R5-4,R19为R5-5;R 18 is R 7 and R 19 is R 8 ; alternatively, R 18 is R 5-4 and R 19 is R 5-5 ;
R20为-COOH、或、-NHR14;R 20 is -COOH, or, -NHR 14 ;
Xa为 X a is
R5a为-(C=O)OR5-1、-(C=O)SR5-2、或、未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基;R 5a is -(C=O)OR 5-1 , -(C=O)SR 5-2 , or, unsubstituted or R 5-3 substituted "heteroatom selected from one of N, O and S" or more, "5-10-membered heteroaryl group with 1-3 heteroatoms;
R1、R2、R3、R4、R6、R5-1、R5-2、R5-3、R5-4、R5-5、R7、R8、R11、R13、R14和L的定义如本发明第一方面所限定;R 1 , R 2 , R 3 , R 4 , R 6 , R 5-1 , R 5-2 , R 5-3 , R 5-4 , R 5-5 , R 7 , R 8 , R 11 , R 13 , R 14 and L are as defined in the first aspect of the present invention;
步骤c:在溶剂中,化合物II-2和化合物III-2发生反应生成化合物I-2;Step c: in a solvent, compound II-2 and compound III-2 react to form compound I-2;
步骤d:在溶剂中,化合物I-2与化合物V反应生成化合物I-3;Step d: in a solvent, compound I-2 reacts with compound V to generate compound I-3;
(3)当X为R5为-(C=O)OR5-1、-(C=O)SR5-2、未取代或R5-3取代的“杂原子选自N、O和S中的一种或多种,杂原子数为1-3个的”5~10元杂芳基、-(C=O)NR5-4(S=O)2R5-5、或、-(C=O)NR7R8时,(3) When X is R 5 is -(C=O)OR 5-1 , -(C=O)SR 5-2 , unsubstituted or R 5-3 substituted "heteroatom selected from one or more of N, O and S Species, "5-10-membered heteroaryl with 1-3 heteroatoms, -(C=O)NR 5-4 (S=O) 2 R 5-5 , or, -(C=O)NR 7 R 8 ,
其中,R1、R2、R3、R4、R5、R6、R12和L的定义如本发明第一方面所限定;Wherein, the definitions of R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 12 and L are as defined in the first aspect of the present invention;
步骤e:在溶剂中,化合物II-3与化合物VI进行反应生成化合物I-4;Step e: in a solvent, compound II-3 is reacted with compound VI to generate compound I-4;
(4)当X为C=O时,R5为-(C=O)NR5-4(S=O)2R5-5时,(4) When X is C=O, and R 5 is -(C=O)NR 5-4 (S=O) 2 R 5-5 ,
其中,R1、R2、R3、R4、R6、R5-4、R5-5和L的定义如本发明第一方面所限定;Wherein, the definitions of R 1 , R 2 , R 3 , R 4 , R 6 , R 5-4 , R 5-5 and L are as defined in the first aspect of the present invention;
步骤f:在溶剂中,化合物II-4与化合物VII进行反应生成化合物I-5。Step f: In a solvent, compound II-4 is reacted with compound VII to generate compound I-5.
本发明的第三方面,提供了一种药物组合物,其含有本发明第一方面所述的化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物,及药用辅料或载体。The third aspect of the present invention provides a pharmaceutical composition, which contains the compound described in the first aspect of the present invention, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or Hydrate, and pharmaceutical excipients or carriers.
进一步地,在上述药物组合物中,本发明第一方面所述的化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物的用量为治疗有效量。Further, in the above pharmaceutical composition, the amount of the compound described in the first aspect of the present invention, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or hydrate thereof is therapeutically effective quantity.
进一步地,在上述药物组合物中,还包含一种或多种第二治疗剂,所述第二治疗剂为预防和/或治疗肿瘤的药物。Further, in the above-mentioned pharmaceutical composition, one or more second therapeutic agents are also included, and the second therapeutic agents are drugs for preventing and/or treating tumors.
进一步地,所述第二治疗剂为传统细胞毒化疗药物、致DNA损伤剂或其他抗肿瘤免疫药物。Further, the second therapeutic agent is a traditional cytotoxic chemotherapeutic drug, a DNA-damaging agent or other anti-tumor immune drug.
本发明的第四方面,提供一种本发明第一方面所述的化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物、或、本发明第三方面所述的药物组合物在制备药物中的用途。The fourth aspect of the present invention provides a compound according to the first aspect of the present invention, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or hydrate thereof, or, the present invention Use of the pharmaceutical composition described in the third aspect in the preparation of a medicament.
进一步地,所述的药物为预防和/或治疗肿瘤或感染性疾病的药物。Further, the medicament is a medicament for preventing and/or treating tumors or infectious diseases.
本发明的第五方面,提供一种本发明第一方面所述的化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物、或、本发明第三方面所述的药物组合物在制备疫苗佐剂中的用途。The fifth aspect of the present invention provides a compound according to the first aspect of the present invention, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or hydrate thereof, or, the present invention Use of the pharmaceutical composition described in the third aspect in the preparation of a vaccine adjuvant.
进一步地,所述的疫苗佐剂为用于治疗肿瘤或感染性疾病的疫苗佐剂。Further, the vaccine adjuvant is a vaccine adjuvant for treating tumors or infectious diseases.
本发明的第六方面,提供一种本发明第一方面所述的化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物、或、本发明第三方面所述的药物组合物在制备干扰素基因刺激蛋白激动剂中的用途。The sixth aspect of the present invention provides a compound according to the first aspect of the present invention, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or hydrate thereof, or, the present invention Use of the pharmaceutical composition described in the third aspect in the preparation of an interferon gene-stimulating protein agonist.
本发明的第七方面,提供了一种药物组合,其包括:本发明第一方面所述的化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物和,免疫检查点抑制剂或致DNA损伤剂。所述的免疫检查点抑制剂为LAG-3抑制剂、TIM3抑制剂、PD-1/PD-L1抑制剂、IDO1抑制剂、CTLA-4抑制剂、TIGIT抑制剂或VISTA抑制剂,优选PD-1/PD-L1抑制剂、IDO1抑制剂、或CTLA-4抑制剂。所述的致DNA损伤剂为PARP抑制剂。本发明第一方面所述的化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物,和免疫检查点抑制剂可同时施用或分别施用;本发明第一方面所述的化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物,和DNA损伤剂可同时施用或分别施用。The seventh aspect of the present invention provides a pharmaceutical combination comprising: the compound described in the first aspect of the present invention, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or Hydrate and, immune checkpoint inhibitors or DNA damaging agents. The immune checkpoint inhibitor is LAG-3 inhibitor, TIM3 inhibitor, PD-1/PD-L1 inhibitor, IDO1 inhibitor, CTLA-4 inhibitor, TIGIT inhibitor or VISTA inhibitor, preferably PD- 1/PD-L1 inhibitor, IDO1 inhibitor, or CTLA-4 inhibitor. The DNA damaging agent is a PARP inhibitor. The compounds described in the first aspect of the present invention, or pharmaceutically acceptable salts, isomers, metabolites, prodrugs, solvates or hydrates thereof, and immune checkpoint inhibitors can be administered simultaneously or separately; The compound of the first aspect of the invention, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or hydrate thereof, and the DNA damaging agent may be administered simultaneously or separately.
本发明的第八方面,提供了一种本发明第六方面所述的药物组合在用于治疗肿瘤的药物中的用途。The eighth aspect of the present invention provides a use of the pharmaceutical combination according to the sixth aspect of the present invention in a medicament for treating tumors.
本发明的第九方面,提供了本发明第一方面所述的化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物在制备治疗肿瘤的药物中的应用,本发明第一方面所述的化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物与免疫检查点抑制剂联用。本发明第一方面所述的化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物和,免疫检查点抑制剂可同时施用或分别施用。The ninth aspect of the present invention provides the compound described in the first aspect of the present invention, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or hydrate thereof in the preparation of a medicament for treating tumors In the application in the first aspect of the present invention, the compound described in the first aspect of the present invention, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or hydrate thereof, is used in combination with an immune checkpoint inhibitor. The compounds described in the first aspect of the present invention, or pharmaceutically acceptable salts, isomers, metabolites, prodrugs, solvates or hydrates thereof, and immune checkpoint inhibitors can be administered simultaneously or separately.
本发明的第十方面,提供了一种本发明第一方面所述的化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物在制备治疗肿瘤的药物中的应用,本发明第一方面所述的化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物与致DNA损伤剂联用。本发明第一方面所述的化合物或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物,和致DNA损伤剂可同时施用或分别施用。The tenth aspect of the present invention provides a compound according to the first aspect of the present invention, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or hydrate thereof, used in the preparation of treating tumors For use in medicine, the compound described in the first aspect of the present invention, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or hydrate thereof, is used in combination with a DNA damage-causing agent. The compounds described in the first aspect of the present invention, or pharmaceutically acceptable salts, isomers, metabolites, prodrugs, solvates or hydrates thereof, and the DNA damaging agent may be administered simultaneously or separately.
本发明中,所述的肿瘤包括但不限于非小细胞肺癌、肝癌、胃癌、白血病、黑色素瘤、结肠癌、神经胶质瘤、肾癌、卵巢癌、胰腺癌、乳腺癌、头颈癌、前列腺癌、多发性骨髓瘤或宫颈癌。In the present invention, the tumors include but are not limited to non-small cell lung cancer, liver cancer, gastric cancer, leukemia, melanoma, colon cancer, glioma, kidney cancer, ovarian cancer, pancreatic cancer, breast cancer, head and neck cancer, prostate cancer cancer, multiple myeloma, or cervical cancer.
术语“免疫检查点抑制剂”是指抑制共抑制受体活性,阻断负向调控信号,使T细胞恢复活性,从而增强免疫应答的物质。The term "immune checkpoint inhibitor" refers to a substance that inhibits the activity of co-inhibitory receptors, blocks negative regulatory signals, and restores the activity of T cells, thereby enhancing the immune response.
术语“LAG-3抑制剂”是指抑制LAG-3活性,使T细胞恢复活性,从而增强免疫应答的物质。The term "LAG-3 inhibitor" refers to a substance that inhibits the activity of LAG-3 and restores the activity of T cells, thereby enhancing the immune response.
术语“TIM3抑制剂”是指抑制TIM3活性,使T细胞恢复活性,从而增强免疫应答的物质。The term "TIM3 inhibitor" refers to a substance that inhibits the activity of TIM3 and restores the activity of T cells, thereby enhancing the immune response.
术语“PD-1/PD-L1抑制剂”是指可以阻断PD-1与PD-L1的结合,阻断负向调控信号,使T细胞恢复活性,从而增强免疫应答的物质。The term "PD-1/PD-L1 inhibitor" refers to a substance that can block the binding of PD-1 to PD-L1, block negative regulatory signals, and restore the activity of T cells, thereby enhancing the immune response.
术语“IDO1抑制剂”是指抑制IDO1活性,使T细胞恢复活性,从而增强免疫应答的物质。The term "IDO1 inhibitor" refers to a substance that inhibits the activity of IDO1 and restores the activity of T cells, thereby enhancing the immune response.
术语“CTLA-4抑制剂”是指阻断CTLA-4所介导的负向调控信号,使T细胞恢复活性,从而增强免疫应答的物质。The term "CTLA-4 inhibitor" refers to a substance that blocks the negative regulatory signals mediated by CTLA-4 and restores the activity of T cells, thereby enhancing the immune response.
术语“VISTA抑制剂”是指抑制VISTA活性,使T细胞恢复活性,从而增强免疫应答的物质。The term "VISTA inhibitor" refers to a substance that inhibits the activity of VISTA and restores the activity of T cells, thereby enhancing the immune response.
术语“TIGIT抑制剂”是指抑制TIGIT活性,使T细胞恢复活性,从而增强免疫应答的物质。The term "TIGIT inhibitor" refers to a substance that inhibits the activity of TIGIT and restores the activity of T cells, thereby enhancing the immune response.
术语“致DNA损伤剂”是指诱导DNA损伤的物质。The term "DNA-damaging agent" refers to a substance that induces DNA damage.
术语“同时施用”是指在同一时间点施用,例如,施用同时包含具有通式I所示结构的苯并噻吩类化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物,和免疫检查点抑制剂的单独的药物组合物;或者,在相同的时间点施用“包含具有通式I所示结构的苯并噻吩类化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物的单独的药物组合物”和“包含免疫检查点抑制剂的单独的药物组合物”。The term "simultaneous administration" refers to administration at the same time point, for example, administration of a benzothiophene compound having the structure shown in general formula I, or a pharmaceutically acceptable salt, isomer, metabolite, or prodrug thereof at the same time , a solvate or a hydrate, and a separate pharmaceutical composition of an immune checkpoint inhibitor; or, at the same time point, administer "a benzothiophene compound having the structure shown in general formula I, or a pharmaceutically acceptable compound thereof. Separate pharmaceutical compositions of accepted salts, isomers, metabolites, prodrugs, solvates or hydrates" and "separate pharmaceutical compositions comprising immune checkpoint inhibitors".
术语“分开施用”是指在不同时间点给药,例如,在不同的时间点施用“包含具有通式I所示结构的苯并噻吩类化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物药物组合物”和“包含免疫检查点抑制剂的单独的药物组合物”;或者,例如,先施用“包含具有通式I所示结构的苯并噻吩类化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物的单独的药物组合物”和“包含免疫检查点抑制剂的单独的药物组合物”中的一种,然后再施用另一种。分开施用可以在时间上接近或间隔很久,但需确保如具有通式I所示结构的苯并噻吩类化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物,和免疫检查点抑制剂可以共同起作用以提供所需的治疗效果。例如,在施用免疫检查点抑制剂之前(例如,5分钟、15分钟、30分钟、45分钟、1小时、2小时、4小时、6小时、12小时、24小时、48小时、72小时、96小时、1星期、2星期、3星期、4星期、5星期、6星期、8星期、或12星期之前)优先施用,或在施用免疫检查点抑制剂之后(例如,5分钟、15分钟、30分钟、45分钟、1小时、2小时、4小时、6小时、12小时、24小时、48小时、72小时、96小时、1星期、2星期、3星期、4星期、5星期、6星期、8星期、或12星期之后)施用具有通式I所示结构的苯并噻吩类化合物、或其药学上可接受的盐、异构体、代谢产物、前药、溶剂合物或水合物。The term "separate administration" refers to administration at different time points, for example, administration at different time points "comprising a benzothiophene compound having the structure shown in general formula I, or a pharmaceutically acceptable salt or isomer thereof. , metabolite, prodrug, solvate or hydrate pharmaceutical composition" and "a separate pharmaceutical composition comprising an immune checkpoint inhibitor"; A single pharmaceutical composition of a thiophene compound, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or hydrate thereof" and "a single pharmaceutical combination comprising an immune checkpoint inhibitor" one of the substances" and then the other. Separate administration can be close in time or at a long interval, but it is necessary to ensure that the benzothiophenes having the structure shown in general formula I, or their pharmaceutically acceptable salts, isomers, metabolites, prodrugs, solvates, etc. Compounds or hydrates, and immune checkpoint inhibitors can work together to provide the desired therapeutic effect. For example, prior to administration of an immune checkpoint inhibitor (eg, 5 minutes, 15 minutes, 30 minutes, 45 minutes, 1 hour, 2 hours, 4 hours, 6 hours, 12 hours, 24 hours, 48 hours, 72 hours, 96 hours, 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 8 weeks, or 12 weeks prior to administration, or after administration of an immune checkpoint inhibitor (eg, 5 minutes, 15 minutes, 30 minutes, 45 minutes, 1 hour, 2 hours, 4 hours, 6 hours, 12 hours, 24 hours, 48 hours, 72 hours, 96 hours, 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 8 weeks, or after 12 weeks) administration of a benzothiophene compound having the structure shown in formula I, or a pharmaceutically acceptable salt, isomer, metabolite, prodrug, solvate or hydrate thereof.
所述的药用辅料可为药物生产领域中广泛采用的那些辅料。辅料主要用于提供一个安全、稳定和功能性的药物组合物,还可以提供方法,使受试者接受给药后活性成分以所期望速率溶出,或促进受试者接受组合物给药后活性成分得到有效吸收。所述的药用辅料可以是惰性填充剂,或者提供某种功能,例如稳定该组合物的整体pH值或防止组合物活性成分的降解。所述的药用辅料可以包括下列辅料中的一种或多种:粘合剂、助悬剂、乳化剂、稀释剂、填充剂、成粒剂、胶粘剂、崩解剂、润滑剂、抗粘着剂、助流剂、润湿剂、胶凝剂、吸收延迟剂、溶解抑制剂、增强剂、吸附剂、缓冲剂、螯合剂、防腐剂、着色剂、矫味剂和甜味剂。The pharmaceutical excipients can be those widely used in the field of pharmaceutical production. Excipients are mainly used to provide a safe, stable and functional pharmaceutical composition, and can also provide a method to enable the subject to dissolve the active ingredient at a desired rate after administration, or to promote the activity of the subject after the composition is administered. The ingredients are effectively absorbed. The pharmaceutical excipients can be inert fillers, or provide some function, such as stabilizing the overall pH of the composition or preventing degradation of the active ingredients of the composition. Described pharmaceutical adjuvants may include one or more of the following adjuvants: binders, suspending agents, emulsifiers, diluents, fillers, granulating agents, adhesives, disintegrating agents, lubricants, anti-sticking agents Agents, glidants, wetting agents, gelling agents, absorption delaying agents, dissolution inhibitors, enhancers, adsorbents, buffers, chelating agents, preservatives, colorants, flavors and sweeteners.
本发明的药物组合物可根据公开的内容使用本领域技术人员已知的任何方法来制备。例如,常规混合、溶解、造粒、乳化、磨细、包封、包埋或冻干工艺。The pharmaceutical compositions of the present invention can be prepared in light of the disclosure using any method known to those skilled in the art. For example, conventional mixing, dissolving, granulating, emulsifying, attenuating, encapsulating, entrapping or lyophilizing processes.
本发明所述的药物组合物可以任何形式给药,包括注射(静脉内)、粘膜、口服(固体和液体制剂)、吸入、眼部、直肠、局部或胃肠外(输注、注射、植入、皮下、静脉内、动脉内、肌内)给药。本发明的药物组合物还可以是控释或延迟释放剂型(例如脂质体或微球)。固体口服制剂的实例包括但不限于粉末、胶囊、囊片、软胶囊剂和片剂。口服或粘膜给药的液体制剂实例包括但不限于悬浮液、乳液、酏剂和溶液。局部用制剂的实例包括但不限于乳剂、凝胶剂、软膏剂、乳膏剂、贴剂、糊剂、泡沫剂、洗剂、滴剂或血清制剂。胃肠外给药的制剂实例包括但不限于注射用溶液、可以溶解或悬浮在药学上可接受载体中的干制剂、注射用悬浮液和注射用乳剂。所述的药物组合物的其它合适制剂的实例包括但不限于滴眼液和其他眼科制剂;气雾剂:如鼻腔喷雾剂或吸入剂;适于胃肠外给药的液体剂型;栓剂以及锭剂。The pharmaceutical compositions of the present invention may be administered in any form, including injection (intravenous), mucosal, oral (solid and liquid formulations), inhalation, ophthalmic, rectal, topical or parenteral (infusion, injection, implant Intradermal, subcutaneous, intravenous, intraarterial, intramuscular) administration. The pharmaceutical compositions of the present invention may also be in controlled release or delayed release dosage forms (eg, liposomes or microspheres). Examples of solid oral formulations include, but are not limited to, powders, capsules, caplets, softgels, and tablets. Examples of liquid formulations for oral or mucosal administration include, but are not limited to, suspensions, emulsions, elixirs, and solutions. Examples of topical formulations include, but are not limited to, creams, gels, ointments, creams, patches, pastes, foams, lotions, drops, or serum formulations. Examples of formulations for parenteral administration include, but are not limited to, solutions for injection, dry formulations that can be dissolved or suspended in a pharmaceutically acceptable carrier, suspensions for injection, and emulsions for injection. Examples of other suitable formulations of the pharmaceutical compositions include, but are not limited to, eye drops and other ophthalmic formulations; aerosols: such as nasal sprays or inhalants; liquid dosage forms suitable for parenteral administration; suppositories and lozenges agent.
术语“药学上可接受的盐”是指本发明化合物的盐,由本发明发现的具有特定取代基的化合物与相对无毒的酸或碱制备。当本发明的化合物中含有相对酸性的功能团时,可以通过在纯的溶液或合适的惰性溶剂中用足够量的碱与这类化合物的游离体形式接触的方式获得碱加成盐。药学上可接受的碱加成盐包括钠、钾、钙、铵、有机氨或镁盐或类似的盐。当本发明的化合物中含有相对碱性的官能团时,可以通过在纯的溶液或合适的惰性溶剂中用足够量的酸与这类化合物的游离体形式接触的方式获得酸加成盐。药学上可接受的酸加成盐的实例包括无机酸盐,所述无机酸包括例如盐酸、氢溴酸、硝酸、碳酸(形成碳酸盐或碳酸氢盐)、磷酸(形成磷酸盐、磷酸一氢盐、磷酸二氢盐、硫酸(形成硫酸盐或硫酸氢盐)、氢碘酸、亚磷酸等;以及有机酸盐,所述有机酸包括如乙酸、丙酸、异丁酸、马来酸、丙二酸、苯甲酸、琥珀酸、辛二酸、反丁烯二酸、乳酸、扁桃酸、邻苯二甲酸、苯磺酸、对甲苯磺酸、柠檬酸、酒石酸和甲磺酸等类似的酸;有机酸盐还包括氨基酸(如精氨酸等)的盐,以及如葡糖醛酸等有机酸的盐。本发明的某些特定的化合物含有碱性和酸性的官能团,从而可以被转换成任一碱或酸加成盐。优选地,以常规方式使盐与碱或酸接触,再分离母体化合物,由此再生化合物的游离体形式。化合物的游离体形式与其各种盐的形式的不同之处在于某些物理性质,例如在极性溶剂中的溶解度不同。The term "pharmaceutically acceptable salts" refers to salts of the compounds of the present invention, prepared from compounds with specific substituents discovered by the present invention and relatively non-toxic acids or bases. When the compounds of the present invention contain relatively acidic functional groups, base addition salts can be obtained by contacting the free form of such compounds with a sufficient amount of base in neat solution or a suitable inert solvent. Pharmaceutically acceptable base addition salts include sodium, potassium, calcium, ammonium, organic ammonia or magnesium salts or similar salts. When the compounds of the present invention contain relatively basic functional groups, acid addition salts can be obtained by contacting the free form of such compounds with a sufficient amount of acid in neat solution or in a suitable inert solvent. Examples of pharmaceutically acceptable acid addition salts include inorganic acid salts including, for example, hydrochloric acid, hydrobromic acid, nitric acid, carbonic acid (forming carbonate or bicarbonate), phosphoric acid (forming phosphate, monophosphate Hydrogen salts, dihydrogen phosphates, sulfuric acid (to form sulfate or hydrogen sulfate), hydroiodic acid, phosphorous acid, etc.; and organic acid salts including, for example, acetic acid, propionic acid, isobutyric acid, maleic acid , malonic acid, benzoic acid, succinic acid, suberic acid, fumaric acid, lactic acid, mandelic acid, phthalic acid, benzenesulfonic acid, p-toluenesulfonic acid, citric acid, tartaric acid and methanesulfonic acid and the like Organic acid salts also include salts of amino acids (such as arginine, etc.), and salts of organic acids such as glucuronic acid. Certain specific compounds of the present invention contain basic and acidic functional groups, which can be used by Converted to any base or acid addition salt. Preferably, the salt is contacted with a base or acid in a conventional manner, and the parent compound is isolated, thereby regenerating the free form of the compound. The free form of the compound and its various salt forms The difference lies in certain physical properties, such as solubility in polar solvents.
本发明的“药学上可接受的盐”可由含有酸根或碱基的母体化合物通过常规化学方法合成。一般情况下,这样的盐的制备方法是:在水或有机溶剂或两者的混合物中,经由游离酸或碱形式的这些化合物与化学计量的适当的碱或酸反应来制备。一般地,优选醚、乙酸乙酯、乙醇、异丙醇或乙腈等非水介质。"Pharmaceutically acceptable salts" of the present invention can be synthesized from parent compounds containing acid groups or bases by conventional chemical methods. Generally, such salts are prepared by reacting the free acid or base form of these compounds with a stoichiometric amount of the appropriate base or acid in water or an organic solvent or a mixture of the two. Generally, non-aqueous media such as ether, ethyl acetate, ethanol, isopropanol or acetonitrile are preferred.
术语“异构体”是指具有相同化学式而有不同的原子排列的化合物。The term "isomer" refers to compounds having the same chemical formula but different arrangements of atoms.
术语“代谢产物”是指式I所示化合物或其盐通过体内代谢产生的药学活性产物。这种产物可以从例如所给药的化合物的氧化、还原、水解、酰胺化、脱酰胺、酯化、脱酯、葡糖醛酸化、酶促裂解等产生。因此,本发明包括本发明的化合物的代谢产物,包括使本发明的化合物与哺乳动物接触足够得到其代谢产物的一段时间的方法而产生的化合物。The term "metabolite" refers to a pharmaceutically active product produced by the in vivo metabolism of a compound of formula I or a salt thereof. Such products may result from, for example, oxidation, reduction, hydrolysis, amidation, deamidation, esterification, deesterification, glucuronidation, enzymatic cleavage, and the like, of the administered compound. Accordingly, the present invention includes metabolites of compounds of the present invention, including compounds produced by methods of contacting a compound of the present invention with a mammal for a period of time sufficient to obtain its metabolites.
代谢产物的鉴定典型地通过制备本发明化合物的放射性标记的同位素、将其以可检测的剂量(例如,大于约0.5mg/kg)非肠道给予动物,例如大鼠、小鼠、豚鼠、猴、或人,允许充分的时间以发生代谢(典型地约30秒到30小时)和从尿、血液或其它生物样本分离其转化产物。这些产物容易分离,因为它们是被标记的(其它通过利用能够结合存在于代谢物中的抗原表位的抗体分离)。以常规的方式确定代谢物结构,例如,通过MS,LC/MS或NMR分析。通常,代谢物的分析是以与本领域技术人员公知的常规药物代谢研亢相同的方法进行的。只要代谢物产物不是以其它方式在体内不能被发现,否则它们可用于本发明化合物的治疗剂量给药的检定测定法。本发明的化合物可以在一个或多个构成该化合物的原子上包含非天然比例的原子同位素。例如,可用放射性同位素标记化合物,比如氚(3H),碘-125(125I)或C-14(14C)。本发明的化合物的所有同位素组成的变换,无论放射性与否,都包括在本发明的范围之内。Metabolites are typically identified by preparing radiolabeled isotopes of the compounds of the invention, and parenterally administering them to animals, eg, rats, mice, guinea pigs, monkeys, at detectable doses (eg, greater than about 0.5 mg/kg) , or humans, allowing sufficient time for metabolism to occur (typically about 30 seconds to 30 hours) and isolation of its transformation products from urine, blood, or other biological samples. These products are easily isolated because they are labeled (others are isolated by the use of antibodies capable of binding epitopes present in the metabolite). Metabolite structures are determined in a conventional manner, eg, by MS, LC/MS or NMR analysis. In general, analysis of metabolites is performed in the same manner as conventional drug metabolism studies known to those skilled in the art. As long as the metabolite products are not otherwise undetectable in vivo, they are useful in assays for therapeutic dosing of the compounds of the present invention. The compounds of the present invention may contain unnatural proportions of atomic isotopes at one or more of the atoms that constitute the compound. For example, compounds can be labeled with radioisotopes, such as tritium ( 3 H), iodine-125 ( 125 I) or C-14 ( 14 C). All transformations of the isotopic composition of the compounds of the present invention, whether radioactive or not, are included within the scope of the present invention.
除了盐的形式,本发明所提供的化合物还存在前药形式。本文所描述的化合物的前药容易地在生理条件下发生化学变化从而转化成本发明的化合物。可在体内转化以提供生物活性物质(即式I所示化合物)的任何化合物是在本发明的范围和主旨内的前药。例如,含有羧基的化合物可形成生理上可水解的酯,其通过在体内水解以得到式I所示化合物本身而充当前药。所述前药优选口服给药,这是因为水解在许多情况下主要在消化酶的影响下发生。当酯本身具有活性或水解发生在血液中时,可使用肠胃外给药。In addition to salt forms, the compounds provided herein also exist in prodrug forms. Prodrugs of the compounds described herein are readily chemically altered under physiological conditions to convert to the compounds of the present invention. Any compound that can be transformed in vivo to provide a biologically active substance (ie, a compound of formula I) is a prodrug within the scope and spirit of the present invention. For example, a compound containing a carboxyl group can form a physiologically hydrolyzable ester, which acts as a prodrug by hydrolysis in vivo to yield the compound of formula I itself. The prodrugs are preferably administered orally, since hydrolysis in many cases occurs primarily under the influence of digestive enzymes. Parenteral administration can be used when the ester itself is active or hydrolysis occurs in the blood.
术语“活性成分”、“治疗剂”、或“活性物质”是指一种化学实体,它可以有效地治疗目标紊乱、疾病或病症。The terms "active ingredient," "therapeutic agent," or "active substance" refer to a chemical entity that is effective in treating the target disorder, disease, or condition.
当所例举的连接基团没有指明其连接方向,其连接方向按与从左往右的读取顺序相同的方向连接。When the exemplified linking group does not indicate its direction of attachment, the direction of attachment is in the same direction as read from left to right.
本领域技术人员可以理解,根据本领域中使用的惯例,本申请描述基团的结构式中所使用的是指,相应的基团通过该位点与式I所示化合物中的其它片段、基团进行连接。It can be understood by those skilled in the art that, according to the conventions used in the art, the formula used in the structural formula of the group described in this application It means that the corresponding group is connected with other fragments and groups in the compound represented by formula I through this site.
在不违背本领域常识的基础上,上述各优选条件,可任意组合,即得本发明各较佳实例。On the basis of not violating common knowledge in the art, the above preferred conditions can be combined arbitrarily to obtain preferred examples of the present invention.
本发明所用试剂和原料均市售可得。The reagents and raw materials used in the present invention are all commercially available.
有益效果:本发明的苯并噻吩类化合物克服现有的干扰素基因刺激蛋白小分子激动剂缺乏、核苷酸类干扰素基因刺激蛋白激动剂成药性差等缺陷。本发明的嘧啶并五元杂环类化合物对干扰素基因刺激蛋白具有很好的激动活性,且对肿瘤和感染性疾病有良好的治疗作用。Beneficial effects: the benzothiophene compound of the present invention overcomes the defects of the existing small molecule agonists of interferon gene-stimulating protein and the poor drugability of nucleotide-type interferon gene-stimulating protein agonists. The pyrimido five-membered heterocyclic compound of the present invention has good agonistic activity on interferon gene-stimulating protein, and has good therapeutic effect on tumors and infectious diseases.
具体实施方式Detailed ways
实施例1:化合物S7的合成Example 1: Synthesis of Compound S7
步骤一:化合物2的合成Step 1: Synthesis of Compound 2
氮气保护下,化合物1(10g,39.2mmol)与Na2S2O3·5H2O(48.6g,196.1mmol),Pd(dppf)Cl2(2.9g,3.92mmol),dppf(3.3g,5.88mmol),Cs2CO3(38.3g,117.6mmol)溶于DMF(300mL)和乙二醇(16mL)中,升温至140℃,过夜反应。待反应完成后,冷却至室温,加入水(20mL)稀释反应液,乙酸乙酯萃取(100ml×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤、浓缩,柱层析纯化得到化合物2(2.9g,35%)。1H NMR(500MHz,Chloroform-d)δ7.07(s,1H),6.93(s,1H),6.22(s,1H),3.90(s,3H),3.89(s,3H),3.88(s,2H).Under nitrogen protection, compound 1 (10 g, 39.2 mmol) was combined with Na 2 S 2 O 3 .5H 2 O (48.6 g, 196.1 mmol), Pd(dppf)Cl 2 (2.9 g, 3.92 mmol), dppf (3.3 g, 5.88 mmol), Cs 2 CO 3 (38.3 g, 117.6 mmol) were dissolved in DMF (300 mL) and ethylene glycol (16 mL), warmed to 140° C., and reacted overnight. After the reaction was completed, it was cooled to room temperature, water (20 mL) was added to dilute the reaction solution, extracted with ethyl acetate (100 mL×3), the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and subjected to column chromatography. Purification gave compound 2 (2.9 g, 35%). 1 H NMR (500MHz, Chloroform-d) δ 7.07(s, 1H), 6.93(s, 1H), 6.22(s, 1H), 3.90(s, 3H), 3.89(s, 3H), 3.88(s , 2H).
步骤二:化合物S7的合成Step 2: Synthesis of Compound S7
将化合物2(100mg,0.48mmol)和丁二酸酐(96mg,0.96mmol)溶于无水THF(3ml)中,加入三乙胺,室温下搅拌反应6h。待反应完成后,减压蒸掉溶剂。向反应残渣中加入水(3ml),乙酸乙酯溶解萃取(10ml×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤、浓缩,柱层析纯化得到化合物S7(118mg,80%)。1H NMR(500MHz,(500MHz,DMSO-d6)δ13.8(br,1H),10.98(s,1H),7.69(d,J=2.1Hz,1H),7.41-7.34(m,2H),3.89(d,J=17.6Hz,6H),2.70-2.62(m,2H),2.59-2.51(m,2H).Compound 2 (100 mg, 0.48 mmol) and succinic anhydride (96 mg, 0.96 mmol) were dissolved in anhydrous THF (3 ml), triethylamine was added, and the reaction was stirred at room temperature for 6 h. After the reaction was completed, the solvent was evaporated under reduced pressure. Water (3 ml) was added to the reaction residue, extracted with ethyl acetate (10 ml×3), the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain compound S7 (118 mg, 80%). 1 H NMR (500 MHz, (500 MHz, DMSO-d 6 ) δ 13.8 (br, 1H), 10.98 (s, 1H), 7.69 (d, J=2.1 Hz, 1H), 7.41-7.34 (m, 2H) , 3.89(d, J=17.6Hz, 6H), 2.70-2.62(m, 2H), 2.59-2.51(m, 2H).
以下实施例2~实施例9中的化合物S8~S15的合成均可参照实施例1的合成方法,只需更换相应的原料即可。For the synthesis of compounds S8 to S15 in the following Examples 2 to 9, reference can be made to the synthesis method of Example 1, and only the corresponding raw materials need to be replaced.
实施例2:化合物S8的合成Example 2: Synthesis of Compound S8
1H NMR(500MHz,DMSO-d6)δ13.8(br,1H),10.98(s,1H),7.69(d,J=2.1Hz,1H),7.37-7.30(m,2H),3.91(s,3H),3.87(s,3H),3.45(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 13.8 (br, 1H), 10.98 (s, 1H), 7.69 (d, J=2.1 Hz, 1H), 7.37-7.30 (m, 2H), 3.91 ( s, 3H), 3.87 (s, 3H), 3.45 (s, 2H).
实施例3:化合物S9的合成Example 3: Synthesis of Compound S9
1H NMR(500MHz,DMSO-d6)δ13.8(br,1H),10.98(s,1H),7.79(d,J=2.2Hz,1H),7.53(d,J=2.2Hz,1H),7.24(s,1H),3.94(s,3H),3.44(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 13.8 (br, 1H), 10.98 (s, 1H), 7.79 (d, J=2.2Hz, 1H), 7.53 (d, J=2.2Hz, 1H) , 7.24(s, 1H), 3.94(s, 3H), 3.44(s, 2H).
实施例4:化合物S10的合成Example 4: Synthesis of Compound S10
1H NMR(500MHz,DMSO-d6)δ13.8(br,1H),10.98(s,1H),7.81(d,J=2.3Hz,1H),7.50(d,J=2.2Hz,1H),7.28(s,1H),6.84(t,J=9.3Hz,1H),5.70(dd,J=9.3,1.8Hz,1H),5.47(dd,J=9.2,1.9Hz,1H),3.87(s,3H),3.45(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 13.8 (br, 1H), 10.98 (s, 1H), 7.81 (d, J=2.3Hz, 1H), 7.50 (d, J=2.2Hz, 1H) , 7.28(s, 1H), 6.84(t, J=9.3Hz, 1H), 5.70(dd, J=9.3, 1.8Hz, 1H), 5.47(dd, J=9.2, 1.9Hz, 1H), 3.87( s, 3H), 3.45(s, 2H).
实施例5:化合物S11的合成Example 5: Synthesis of Compound S11
1H NMR(500MHz,DMSO-d6)δ13.8(br,1H),10.98(s,1H),7.69(d,J=2.2Hz,1H),7.39-7.35(m,2H),4.14(q,J=7.0Hz,2H),3.89(d,J=17.6Hz,5H),3.54(s,2H),1.26(t,J=6.9Hz,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 13.8 (br, 1H), 10.98 (s, 1H), 7.69 (d, J=2.2Hz, 1H), 7.39-7.35 (m, 2H), 4.14 ( q, J=7.0Hz, 2H), 3.89 (d, J=17.6Hz, 5H), 3.54 (s, 2H), 1.26 (t, J=6.9Hz, 3H).
实施例6:化合物S12的合成Example 6: Synthesis of Compound S12
1H NMR(500MHz,DMSO-d6)δ13.8(br,1H),10.98(s,1H),7.69(d,J=2.1Hz,1H),7.37-7.30(m,2H),3.91(s,3H),3.87(s,3H),2.84(s,2H),1.39(s,3H),1.37(s,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 13.8 (br, 1H), 10.98 (s, 1H), 7.69 (d, J=2.1 Hz, 1H), 7.37-7.30 (m, 2H), 3.91 ( s, 3H), 3.87(s, 3H), 2.84(s, 2H), 1.39(s, 3H), 1.37(s, 3H).
实施例7:化合物S13的合成Example 7: Synthesis of Compound S13
1H NMR(500MHz,DMSO-d6)δ13.8(br,1H),10.98(s,1H),7.71(d,J=2.3Hz,1H),7.37(d,J=2.1Hz,1H),7.24(s,1H),4.27(t,J=6.5Hz,2H),3.90(s,3H),3.68-3.57(m,4H),3.39(s,2H),2.73(t,J=6.4Hz,2H),2.58-2.44(m,4H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 13.8 (br, 1H), 10.98 (s, 1H), 7.71 (d, J=2.3Hz, 1H), 7.37 (d, J=2.1Hz, 1H) , 7.24(s, 1H), 4.27(t, J=6.5Hz, 2H), 3.90(s, 3H), 3.68-3.57(m, 4H), 3.39(s, 2H), 2.73(t, J=6.4 Hz, 2H), 2.58-2.44(m, 4H).
实施例8:化合物S14的合成Example 8: Synthesis of Compound S14
1H NMR(500MHz,DMSO-d6)δ13.8(br,1H),10.98(s,1H),7.70(s,1H),7.26-7.20(m,2H),6.00(s,2H),3.45(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 13.8 (br, 1H), 10.98 (s, 1H), 7.70 (s, 1H), 7.26-7.20 (m, 2H), 6.00 (s, 2H), 3.45(s, 2H).
实施例9:化合物S15的合成Example 9: Synthesis of Compound S15
1H NMR(500MHz,DMSO-d6)δ13.8(br,1H),10.98(s,1H),7.79(s,1H),7.27(s,1H),7.23(s,1H),4.30-4.21(m,4H),3.45(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 13.8 (br, 1H), 10.98 (s, 1H), 7.79 (s, 1H), 7.27 (s, 1H), 7.23 (s, 1H), 4.30- 4.21(m, 4H), 3.45(s, 2H).
实施例10:化合物S16的合成Example 10: Synthesis of Compound S16
将化合物S8(100mg,0.34mmol)和肼盐酸盐(24mg,0.34mmol)溶于无水DMF(5mL)中,向上述溶液中加入HATU(193.2mg,0.51mmol)和DIPEA(132mg,1.02mmol),室温下过夜反应。待反应完成后,将反应液倒入冰水中,乙酸乙酯萃取(10mL×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得到化合物S16(86mg,82%)。1H NMR(500MHz,DMSO-d6)δ10.78(s,1H),9.06(t,J=3.7Hz,1H),7.69(d,J=2.1Hz,1H),7.40-7.34(m,2H),4.01(d,J=3.7Hz,2H),3.89(s,3H),3.76(s,3H),3.34(s,2H).Compound S8 (100 mg, 0.34 mmol) and hydrazine hydrochloride (24 mg, 0.34 mmol) were dissolved in dry DMF (5 mL), HATU (193.2 mg, 0.51 mmol) and DIPEA (132 mg, 1.02 mmol) were added to the above solution ) and reacted overnight at room temperature. After the reaction was completed, the reaction solution was poured into ice water, extracted with ethyl acetate (10 mL×3), the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain compound S16 ( 86 mg, 82%). 1 H NMR (500MHz, DMSO-d 6 ) δ 10.78 (s, 1H), 9.06 (t, J=3.7Hz, 1H), 7.69 (d, J=2.1Hz, 1H), 7.40-7.34 (m, 2H), 4.01(d, J=3.7Hz, 2H), 3.89(s, 3H), 3.76(s, 3H), 3.34(s, 2H).
以下实施例11~实施例29中的化合物S17~S35的合成均可参照实施例10的合成方法,只需更换相应的原料即可。For the synthesis of compounds S17 to S35 in the following Examples 11 to 29, reference can be made to the synthesis method of Example 10, and only the corresponding raw materials need to be replaced.
实施例11:化合物S17的合成Example 11: Synthesis of Compound S17
1H NMR(500MHz,DMSO-d6)δ10.78(s,1H),9.86(s,1H),8.73(s,1H),7.69(s,1H),7.39-7.33(m,1H),4.35(m,2H),3.89(s,3H),3.80(s,3H),3.33(s,3H),2.56(m,1H). 1 H NMR (500MHz, DMSO-d 6 ) δ 10.78(s, 1H), 9.86(s, 1H), 8.73(s, 1H), 7.69(s, 1H), 7.39-7.33(m, 1H), 4.35(m, 2H), 3.89(s, 3H), 3.80(s, 3H), 3.33(s, 3H), 2.56(m, 1H).
实施例12:化合物S18的合成Example 12: Synthesis of Compound S18
1H NMR(500MHz,DMSO-d6)δ12.88(s,1H),10.78(s,1H),7.71(d,J=2.1Hz,1H),7.40-7.33(m,2H),3.89(d,J=17.6Hz,6H),3.43(s,2H),3.12(s,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 12.88 (s, 1H), 10.78 (s, 1H), 7.71 (d, J=2.1 Hz, 1H), 7.40-7.33 (m, 2H), 3.89 ( d, J=17.6Hz, 6H), 3.43 (s, 2H), 3.12 (s, 3H).
实施例13:化合物S19的合成Example 13: Synthesis of Compound S19
1H NMR(500MHz,DMSO-d6)δ10.88(s,1H),8.86(s,1H),7.69(d,J=2.1Hz,1H),7.41-7.34(m,2H),3.89(d,J=17.6Hz,6H),3.41(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.88 (s, 1H), 8.86 (s, 1H), 7.69 (d, J=2.1 Hz, 1H), 7.41-7.34 (m, 2H), 3.89 ( d, J=17.6Hz, 6H), 3.41(s, 2H).
实施例14:化合物S20的合成Example 14: Synthesis of Compound S20
1H NMR(500MHz,DMSO-d6)δ10.86(s,1H),10.50(s,1H),7.69(s,1H),7.51(s,1H),7.39(s,1H),7.12(s,1H),3.89(s,3H),3.77(s,3H),3.37(s,2H). 1 H NMR (500MHz, DMSO-d 6 ) δ 10.86(s, 1H), 10.50(s, 1H), 7.69(s, 1H), 7.51(s, 1H), 7.39(s, 1H), 7.12( s, 1H), 3.89 (s, 3H), 3.77 (s, 3H), 3.37 (s, 2H).
实施例15:化合物S21的合成Example 15: Synthesis of Compound S21
1H NMR(500MHz,DMSO-d6)δ10.83(s,1H),7.69(d,J=2.2Hz,1H),7.39-7.33(m,2H),3.91(s,3H),3.87(s,3H),3.63(s,3H),3.36(s,2H),3.18(s,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.83 (s, 1H), 7.69 (d, J=2.2 Hz, 1H), 7.39-7.33 (m, 2H), 3.91 (s, 3H), 3.87 ( s, 3H), 3.63 (s, 3H), 3.36 (s, 2H), 3.18 (s, 3H).
实施例16:化合物S22的合成Example 16: Synthesis of Compound S22
1H NMR(500MHz,DMSO-d6)δ10.83(s,1H),7.71(d,J=2.2Hz,1H),7.37-7.30(m,2H),7.21(s,1H),7.09(s,1H),3.89(d,J=17.6Hz,5H),3.46(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.83 (s, 1H), 7.71 (d, J=2.2 Hz, 1H), 7.37-7.30 (m, 2H), 7.21 (s, 1H), 7.09 ( s, 1H), 3.89 (d, J=17.6Hz, 5H), 3.46 (s, 2H).
实施例17:化合物S23的合成Example 17: Synthesis of Compound S23
1H NMR(500MHz,DMSO-d6)δ10.83(s,1H),7.70(d,J=2.1Hz,1H),7.34(s,1H),7.28(d,J=2.2Hz,1H),6.73(s,1H),4.10(s,2H),3.89(d,J=17.6Hz,6H),3.42(s,2H).1H NMR (500MHz, DMSO-d 6 ) δ 10.83 (s, 1H), 7.70 (d, J=2.1Hz, 1H), 7.34 (s, 1H), 7.28 (d, J=2.2Hz, 1H), 6.73(s, 1H), 4.10(s, 2H), 3.89(d, J=17.6Hz, 6H), 3.42(s, 2H).
实施例18:化合物S24的合成Example 18: Synthesis of Compound S24
1H NMR(500MHz,DMSO-d6)δ10.83(s,1H),7.70(d,J=2.3Hz,1H),7.37-7.33(m,2H),7.09(s,1H),6.33(s,2H),5.89(tt,J=13.8,5.5Hz,1H),5.21(dt,J=13.9,1.0Hz,2H),3.93-3.85(m,6H),3.40(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.83 (s, 1H), 7.70 (d, J=2.3 Hz, 1H), 7.37-7.33 (m, 2H), 7.09 (s, 1H), 6.33 ( s, 2H), 5.89 (tt, J=13.8, 5.5Hz, 1H), 5.21 (dt, J=13.9, 1.0Hz, 2H), 3.93-3.85 (m, 6H), 3.40 (s, 2H).
实施例19:化合物S25的合成Example 19: Synthesis of Compound S25
1H NMR(500MHz,DMSO-d6)δ10.88(s,1H),8.34(s,1H),7.70(d,J=2.1Hz,1H),7.37(s,1H),7.29(d,J=2.0Hz,1H),7.09(s,1H),3.89(d,J=17.6Hz,5H),3.60(t,J=5.5Hz,2H),3.43(s,2H),2.80(t,J=5.5Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.88 (s, 1H), 8.34 (s, 1H), 7.70 (d, J=2.1 Hz, 1H), 7.37 (s, 1H), 7.29 (d, J=2.0Hz, 1H), 7.09(s, 1H), 3.89(d, J=17.6Hz, 5H), 3.60(t, J=5.5Hz, 2H), 3.43(s, 2H), 2.80(t, J=5.5Hz, 2H).
实施例20:化合物S26的合成Example 20: Synthesis of Compound S26
1H NMR(500MHz,DMSO-d6)δ10.88(s,1H),7.70(d,J=2.1Hz,1H),7.40-7.30(m,2H),7.09(s,1H),4.80(d,J=6.8Hz,1H),4.13(t,J=7.0Hz,1H),3.99-3.90(m,1H),3.88(d,J=15.9Hz,6H),3.77-3.67(m,2H),3.51-3.41(m,2H),3.37-3.29(m,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.88 (s, 1H), 7.70 (d, J=2.1 Hz, 1H), 7.40-7.30 (m, 2H), 7.09 (s, 1H), 4.80 ( d, J=6.8Hz, 1H), 4.13 (t, J=7.0Hz, 1H), 3.99-3.90 (m, 1H), 3.88 (d, J=15.9Hz, 6H), 3.77-3.67 (m, 2H) ), 3.51-3.41(m, 2H), 3.37-3.29(m, 2H).
实施例21:化合物S27的合成Example 21: Synthesis of Compound S27
1H NMR(500MHz,DMSO-d6)δ10.88(s,1H),8.16(s,1H),7.72-7.64(m,3H),7.44-7.36(m,4H),7.18(tt,J=7.2,1.5Hz,1H),3.89(d,J=16.7Hz,6H),3.55(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.88 (s, 1H), 8.16 (s, 1H), 7.72-7.64 (m, 3H), 7.44-7.36 (m, 4H), 7.18 (tt, J =7.2, 1.5Hz, 1H), 3.89(d, J=16.7Hz, 6H), 3.55(s, 2H).
实施例22:化合物S28的合成Example 22: Synthesis of Compound S28
1H NMR(500MHz,DMSO-d6)δ10.88(s,1H),8.16(s,1H),7.72(d,J=2.1Hz,1H),7.60-7.54(m,2H),7.44-7.39(m,2H),7.17-7.10(m,2H),3.89(d,J=17.6Hz,6H),3.55(s,2H),2.33(s,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.88 (s, 1H), 8.16 (s, 1H), 7.72 (d, J=2.1 Hz, 1H), 7.60-7.54 (m, 2H), 7.44- 7.39(m, 2H), 7.17-7.10(m, 2H), 3.89(d, J=17.6Hz, 6H), 3.55(s, 2H), 2.33(s, 3H).
实施例23:化合物S29的合成Example 23: Synthesis of Compound S29
1H NMR(500MHz,DMSO-d6)δ10.88(s,1H),8.27(s,1H),7.72(d,J=2.1Hz,1H),7.68-7.61(m,2H),7.44-7.39(m,2H),7.19-7.11(m,2H),3.89(d,J=17.6Hz,6H),3.56(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.88 (s, 1H), 8.27 (s, 1H), 7.72 (d, J=2.1 Hz, 1H), 7.68-7.61 (m, 2H), 7.44- 7.39(m, 2H), 7.19-7.11(m, 2H), 3.89(d, J=17.6Hz, 6H), 3.56(s, 2H).
实施例24:化合物S30的合成Example 24: Synthesis of Compound S30
1H NMR(500MHz,DMSO-d6)δ10.88(s,1H),7.72(d,J=2.1Hz,1H),7.45-7.40(m,2H),7.39-7.29(m,3H),7.33-7.23(m,3H),6.04(s,1H),4.63(t,J=1.0Hz,2H),3.89(d,J=17.6Hz,6H),3.42(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.88 (s, 1H), 7.72 (d, J=2.1 Hz, 1H), 7.45-7.40 (m, 2H), 7.39-7.29 (m, 3H), 7.33-7.23(m, 3H), 6.04(s, 1H), 4.63(t, J=1.0Hz, 2H), 3.89(d, J=17.6Hz, 6H), 3.42(s, 2H).
实施例25:化合物S31的合成Example 25: Synthesis of Compound S31
1H NMR(500MHz,DMSO-d6)δ10.88(s,1H),8.73(t,J=1.7Hz,1H),8.47-8.40(m,1H),7.70(dt,J=9.9,2.1Hz,2H),7.40-7.35(m,2H),7.25(dd,J=7.9,3.5Hz,1H),7.06(s,1H),4.67(s,2H),3.89(d,J=17.6Hz,6H),3.46(s,2H). 1 H NMR (500MHz, DMSO-d 6 ) δ 10.88 (s, 1H), 8.73 (t, J=1.7Hz, 1H), 8.47-8.40 (m, 1H), 7.70 (dt, J=9.9, 2.1 Hz, 2H), 7.40-7.35(m, 2H), 7.25(dd, J=7.9, 3.5Hz, 1H), 7.06(s, 1H), 4.67(s, 2H), 3.89(d, J=17.6Hz , 6H), 3.46(s, 2H).
实施例26:化合物S32的合成Example 26: Synthesis of Compound S32
1H NMR(500MHz,DMSO-d6)δ10.88(s,1H),8.19(s,1H),7.72(d,J=2.1Hz,1H),7.40-7.27(m,7H),4.66(t,J=1.0Hz,2H),3.89(d,J=17.6Hz,6H),3.35(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.88 (s, 1H), 8.19 (s, 1H), 7.72 (d, J=2.1 Hz, 1H), 7.40-7.27 (m, 7H), 4.66 ( t, J=1.0Hz, 2H), 3.89 (d, J=17.6Hz, 6H), 3.35 (s, 2H).
实施例27:化合物S33的合成Example 27: Synthesis of Compound S33
1H NMR(500MHz,DMSO-d6)δ10.88(s,1H),9.33(s,1H),7.74(d,J=2.1Hz,1H),7.35(s,1H),7.21(d,J=2.2Hz,1H),4.08-4.01(m,1H),3.89(d,J=17.6Hz,6H),3.81-3.72(m,4H),3.33(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.88 (s, 1H), 9.33 (s, 1H), 7.74 (d, J=2.1 Hz, 1H), 7.35 (s, 1H), 7.21 (d, J=2.2Hz, 1H), 4.08-4.01 (m, 1H), 3.89 (d, J=17.6Hz, 6H), 3.81-3.72 (m, 4H), 3.33 (s, 2H).
实施例28:化合物S34的合成Example 28: Synthesis of Compound S34
1H NMR(500MHz,DMSO-d6)δ10.88(s,1H),9.52(s,1H),7.70(d,J=2.1Hz,1H),7.39-7.32(m,2H),3.88(d,J=15.4Hz,6H),3.36(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.88 (s, 1H), 9.52 (s, 1H), 7.70 (d, J=2.1 Hz, 1H), 7.39-7.32 (m, 2H), 3.88 ( d, J=15.4Hz, 6H), 3.36(s, 2H).
实施例29:化合物S35的合成Example 29: Synthesis of Compound S35
1H NMR(500MHz,DMSO-d6)δ10.88(s,1H),7.69(d,J=2.1Hz,1H),7.40-7.37(m,1H),7.34(s,1H),7.32(d,J=7.7Hz,1H),7.20(d,J=7.7Hz,1H),3.91(s,3H),3.87(s,3H),3.25(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 10.88 (s, 1H), 7.69 (d, J=2.1 Hz, 1H), 7.40-7.37 (m, 1H), 7.34 (s, 1H), 7.32 ( d, J=7.7Hz, 1H), 7.20 (d, J=7.7Hz, 1H), 3.91 (s, 3H), 3.87 (s, 3H), 3.25 (s, 2H).
实施例30:化合物S36的合成Example 30: Synthesis of Compound S36
步骤一:化合物5的合成Step 1: Synthesis of Compound 5
氮气保护下,化合物3(10g,47.4mmol)溶于无水DMF中,加入无水碳酸钾(13g,94.8mmol),置于0℃下,将化合物4(6.9g,57.8mmol)滴加至上述混悬液中,0℃下搅拌30min。然后,升温至80℃,过夜反应。待反应完全后,冷却至室温,减压蒸掉DMF,加入水,DCM萃取(100mL×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得到化合物5(9.1g,72%)。Under nitrogen protection, compound 3 (10 g, 47.4 mmol) was dissolved in anhydrous DMF, anhydrous potassium carbonate (13 g, 94.8 mmol) was added, and at 0 °C, compound 4 (6.9 g, 57.8 mmol) was added dropwise to The above suspension was stirred at 0 °C for 30 min. Then, the temperature was raised to 80°C, and the reaction was carried out overnight. After the reaction was completed, it was cooled to room temperature, DMF was evaporated under reduced pressure, water was added, extracted with DCM (100 mL×3), the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain Compound 5 (9.1 g, 72%).
步骤二:化合物6的生成Step 2: Generation of Compound 6
将化合物5溶于THF中,0℃下,加入2MNaOH水溶液,转移至室温,搅拌反应2h。待反应完全后,加入2M稀盐酸调节pH至2~3,白色沉淀析出,抽滤,THF洗涤滤饼,收集滤饼、干燥得到化合物6,直接投入下一步反应。Compound 5 was dissolved in THF, 2M NaOH aqueous solution was added at 0° C., the mixture was transferred to room temperature, and the reaction was stirred for 2 h. After the reaction was completed, 2M dilute hydrochloric acid was added to adjust the pH to 2-3, a white precipitate was precipitated, suction filtered, the filter cake was washed with THF, the filter cake was collected and dried to obtain compound 6, which was directly put into the next reaction.
步骤三:化合物8的生成Step 3: Generation of Compound 8
将化合物6(5g,21mmol)溶于无水DMF(20mL)中,依次加入HATU(12g,31.5mmol),DIPEA(8.1g,63mmol)和化合物7(2.8g,21mmol)。室温下过夜反应。待反应完全后,减压蒸掉DMF。向反应残渣中加入水(10mL),乙酸乙酯萃取(20mL×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得到化合物8(6.3g,85%)。1H NMR(500MHz,Chloroform-d)δ8.28-8.18(m,2H),7.51(d,J=2.1Hz,1H),7.46(s,1H),4.09(d,J=5.7Hz,2H),3.89(d,J=14.6Hz,6H),1.42(s,9H).Compound 6 (5 g, 21 mmol) was dissolved in dry DMF (20 mL), HATU (12 g, 31.5 mmol), DIPEA (8.1 g, 63 mmol) and compound 7 (2.8 g, 21 mmol) were added sequentially. React overnight at room temperature. After the reaction was completed, DMF was evaporated under reduced pressure. Water (10 mL) was added to the reaction residue, extracted with ethyl acetate (20 mL×3), the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain compound 8 (6.3 g, 85%). 1 H NMR (500MHz, Chloroform-d) δ 8.28-8.18 (m, 2H), 7.51 (d, J=2.1Hz, 1H), 7.46 (s, 1H), 4.09 (d, J=5.7Hz, 2H) ), 3.89(d, J=14.6Hz, 6H), 1.42(s, 9H).
步骤四:化合物S36的合成Step 4: Synthesis of Compound S36
将化合物8(600mg,1.7mmol)溶于DCM(5mL)中,置于0℃下,加入CF3COOH(1mL)。转移至室温,搅拌反应2h。待反应完全后,减压蒸掉溶剂和多余的CF3COOH。然后将所得到的产物溶于无水DMF(5mL)中,依次加入盐酸羟胺(120mg,1.7mmol),HATU(970mg,2.6mmol)和DIPEA(1.1g,8.5mmol)。室温下搅拌反应6h,待反应完成后,减压蒸掉DMF。向反应残渣中加入水(20mL),乙酸乙酯萃取(50mL×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得到化合物S36(421mg,80%)。1H NMR(500MHz,DMSO-d6)δ9.50(d,J=4.8Hz,1H),8.25(t,J=5.8Hz,1H),8.12(d,J=4.8Hz,1H),7.73(d,J=2.2Hz,1H),7.57(d,J=2.1Hz,1H),7.42(s,1H),3.96-3.86(m,8H).Compound 8 (600 mg, 1.7 mmol) was dissolved in DCM (5 mL), placed at 0 °C, and CF3COOH (1 mL) was added. Transfer to room temperature and stir the reaction for 2h. After the reaction was completed, the solvent and excess CF 3 COOH were evaporated under reduced pressure. The resulting product was then dissolved in dry DMF (5 mL), followed by addition of hydroxylamine hydrochloride (120 mg, 1.7 mmol), HATU (970 mg, 2.6 mmol) and DIPEA (1.1 g, 8.5 mmol). The reaction was stirred at room temperature for 6 h. After the reaction was completed, the DMF was evaporated under reduced pressure. Water (20 mL) was added to the reaction residue, extracted with ethyl acetate (50 mL×3), the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain compound S36 (421 mg, 80 %). 1 H NMR (500 MHz, DMSO-d 6 ) δ 9.50 (d, J=4.8 Hz, 1H), 8.25 (t, J=5.8 Hz, 1H), 8.12 (d, J=4.8 Hz, 1H), 7.73 (d, J=2.2Hz, 1H), 7.57 (d, J=2.1Hz, 1H), 7.42 (s, 1H), 3.96-3.86 (m, 8H).
以下实施例31~实施例64中的化合物S37~S70的合成均可参照实施例30的合成方法,只需更换相应的原料即可。For the synthesis of compounds S37 to S70 in the following Examples 31 to 64, reference can be made to the synthesis method of Example 30, and only the corresponding raw materials need to be replaced.
实施例31:化合物S37的合成Example 31: Synthesis of Compound S37
1H NMR(500MHz,DMSO-d6)δ9.83(d,J=4.8Hz,1H),8.28(d,J=2.1Hz,1H),8.07(t,J=4.4Hz,1H),8.01(d,J=4.9Hz,1H),7.53(d,J=2.0Hz,1H),7.44(s,1H),3.90(d,J=12.8Hz,6H),3.37(td,J=6.2,4.4Hz,2H),2.74(t,J=6.2Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 9.83 (d, J=4.8 Hz, 1H), 8.28 (d, J=2.1 Hz, 1H), 8.07 (t, J=4.4 Hz, 1H), 8.01 (d, J=4.9Hz, 1H), 7.53 (d, J=2.0Hz, 1H), 7.44 (s, 1H), 3.90 (d, J=12.8Hz, 6H), 3.37 (td, J=6.2, 4.4Hz, 2H), 2.74(t, J=6.2Hz, 2H).
实施例32:化合物S38的合成Example 32: Synthesis of Compound S38
1H NMR(500MHz,DMSO-d6)δ8.28(d,J=2.1Hz,1H),8.18-8.10(m,2H),7.65(t,J=5.3Hz,1H),7.54(d,J=2.1Hz,1H),7.44(s,1H),3.89(d,J=14.6Hz,6H),3.59(d,J=5.3Hz,2H),1.20(s,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.28 (d, J=2.1 Hz, 1H), 8.18-8.10 (m, 2H), 7.65 (t, J=5.3 Hz, 1H), 7.54 (d, J=2.1Hz, 1H), 7.44(s, 1H), 3.89(d, J=14.6Hz, 6H), 3.59(d, J=5.3Hz, 2H), 1.20(s, 6H).
实施例33:化合物S39的合成Example 33: Synthesis of Compound S39
1H NMR(500MHz,DMSO-d6)δ8.15(t,J=5.8Hz,1H),7.74(d,J=2.2Hz,1H),7.44(d,J=2.1Hz,1H),7.32(s,1H),4.01(d,J=5.7Hz,2H),3.89(d,J=14.6Hz,6H),3.58(s,3H),3.15(s,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.15 (t, J=5.8 Hz, 1H), 7.74 (d, J=2.2 Hz, 1H), 7.44 (d, J=2.1 Hz, 1H), 7.32 (s, 1H), 4.01 (d, J=5.7Hz, 2H), 3.89 (d, J=14.6Hz, 6H), 3.58 (s, 3H), 3.15 (s, 3H).
实施例34:化合物S40的合成Example 34: Synthesis of Compound S40
1H NMR(500MHz,DMSO-d6)δ8.24(d,J=2.0Hz,1H),8.18(t,J=5.8Hz,1H),7.46-7.39(m,2H),7.16(s,1H),4.06-3.95(m,2H),3.97-3.80(m,7H),1.24(d,J=6.6Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.24 (d, J=2.0 Hz, 1H), 8.18 (t, J=5.8 Hz, 1H), 7.46-7.39 (m, 2H), 7.16 (s, 1H), 4.06-3.95 (m, 2H), 3.97-3.80 (m, 7H), 1.24 (d, J=6.6Hz, 6H).
实施例35:化合物S41的合成Example 35: Synthesis of Compound S41
1H NMR(500MHz,DMSO-d6)δ8.22(d,J=2.2Hz,1H),8.16(t,J=5.8Hz,1H),7.41(d,J=3.6Hz,2H),7.35(d,J=2.2Hz,1H),4.57(t,J=7.3Hz,1H),4.04(d,J=5.9Hz,2H),3.93-3.83(m,8H),3.42(t,J=6.8Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.22 (d, J=2.2 Hz, 1H), 8.16 (t, J=5.8 Hz, 1H), 7.41 (d, J=3.6 Hz, 2H), 7.35 (d, J=2.2Hz, 1H), 4.57 (t, J=7.3Hz, 1H), 4.04 (d, J=5.9Hz, 2H), 3.93-3.83 (m, 8H), 3.42 (t, J= 6.8Hz, 2H).
实施例36:化合物S42的合成Example 36: Synthesis of Compound S42
1H NMR(500MHz,DMSO-d6)δ8.25-8.15(m,1H),7.48-7.41(m,1H),7.38(s,1H),7.20(s,1H),7.09(s,1H),6.09(s,1H),4.09(d,J=5.7Hz,2H),3.89(d,J=13.7Hz,6H). 1 H NMR (500MHz, DMSO-d 6 ) δ 8.25-8.15 (m, 1H), 7.48-7.41 (m, 1H), 7.38 (s, 1H), 7.20 (s, 1H), 7.09 (s, 1H) ), 6.09(s, 1H), 4.09(d, J=5.7Hz, 2H), 3.89(d, J=13.7Hz, 6H).
实施例37:化合物S43的合成Example 37: Synthesis of Compound S43
1H NMR(500MHz,DMSO-d6)δ8.24(d,J=2.0Hz,1H),8.18(t,J=5.8Hz,1H),7.49(d,J=2.1Hz,1H),7.43(s,1H),7.10(s,1H),4.09-3.94(m,2H),3.89(d,J=14.8Hz,6H),2.39(p,J=5.8Hz,1H),1.41(ddd,J=10.4,9.7,5.7Hz,2H),1.04(ddd,J=10.7,9.7,5.6Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.24 (d, J=2.0 Hz, 1H), 8.18 (t, J=5.8 Hz, 1H), 7.49 (d, J=2.1 Hz, 1H), 7.43 (s, 1H), 7.10 (s, 1H), 4.09-3.94 (m, 2H), 3.89 (d, J=14.8Hz, 6H), 2.39 (p, J=5.8Hz, 1H), 1.41 (ddd, J=10.4, 9.7, 5.7Hz, 2H), 1.04 (ddd, J=10.7, 9.7, 5.6Hz, 2H).
实施例38:化合物S44的合成Example 38: Synthesis of Compound S44
1H NMR(500MHz,DMSO-d6)δ8.27(d,J=2.1Hz,1H),8.15(t,J=5.8Hz,1H),7.61-7.54(m,2H),7.47(s,1H),7.41(ddt,J=7.5,2.6,1.1Hz,2H),7.34-7.22(m,3H),4.64(t,J=1.0Hz,2H),4.08(d,J=5.9Hz,2H),3.89(d,J=14.3Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.27 (d, J=2.1 Hz, 1H), 8.15 (t, J=5.8 Hz, 1H), 7.61-7.54 (m, 2H), 7.47 (s, 1H), 7.41 (ddt, J=7.5, 2.6, 1.1Hz, 2H), 7.34-7.22 (m, 3H), 4.64 (t, J=1.0Hz, 2H), 4.08 (d, J=5.9Hz, 2H) ), 3.89(d, J=14.3Hz, 6H).
实施例39:化合物S45的合成Example 39: Synthesis of Compound S45
1H NMR(500MHz,DMSO-d6)δ8.27(d,J=2.2Hz,1H),8.10(t,J=5.8Hz,1H),7.55(d,J=2.1Hz,1H),7.46(s,1H),7.17(dtt,J=8.0,4.9,1.0Hz,1H),7.11(s,1H),6.97(td,J=8.1,5.0Hz,1H),4.69(d,J=1.1Hz,2H),4.10(d,J=5.9Hz,2H),3.88(d,J=15.6Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.27 (d, J=2.2 Hz, 1H), 8.10 (t, J=5.8 Hz, 1H), 7.55 (d, J=2.1 Hz, 1H), 7.46 (s, 1H), 7.17 (dtt, J=8.0, 4.9, 1.0Hz, 1H), 7.11 (s, 1H), 6.97 (td, J=8.1, 5.0Hz, 1H), 4.69 (d, J=1.1 Hz, 2H), 4.10 (d, J=5.9Hz, 2H), 3.88 (d, J=15.6Hz, 6H).
实施例40:化合物S46的合成Example 40: Synthesis of Compound S46
1H NMR(500MHz,DMSO-d6)δ8.27(d,J=2.1Hz,1H),8.14(t,J=5.8Hz,1H),7.69-7.63(m,2H),7.57-7.48(m,4H),7.45(s,1H),4.65(t,J=1.0Hz,2H),4.10(d,J=5.7Hz,2H),3.89(d,J=15.0Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.27 (d, J=2.1 Hz, 1H), 8.14 (t, J=5.8 Hz, 1H), 7.69-7.63 (m, 2H), 7.57-7.48 ( m, 4H), 7.45 (s, 1H), 4.65 (t, J=1.0Hz, 2H), 4.10 (d, J=5.7Hz, 2H), 3.89 (d, J=15.0Hz, 6H).
实施例41:化合物S47的合成Example 41: Synthesis of Compound S47
1H NMR(500MHz,DMSO-d6)δ8.27(d,J=2.3Hz,1H),8.14(t,J=5.8Hz,1H),7.56(d,J=2.1Hz,1H),7.43(s,1H),7.32(dt,J=8.4,1.0Hz,2H),7.15(s,1H),6.85-6.78(m,2H),4.63(t,J=1.0Hz,2H),4.05(d,J=5.9Hz,2H),3.89(d,J=15.2Hz,6H),3.78(s,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.27 (d, J=2.3 Hz, 1H), 8.14 (t, J=5.8 Hz, 1H), 7.56 (d, J=2.1 Hz, 1H), 7.43 (s, 1H), 7.32 (dt, J=8.4, 1.0Hz, 2H), 7.15 (s, 1H), 6.85-6.78 (m, 2H), 4.63 (t, J=1.0Hz, 2H), 4.05 ( d, J=5.9Hz, 2H), 3.89 (d, J=15.2Hz, 6H), 3.78 (s, 3H).
实施例42:化合物S48的合成Example 42: Synthesis of Compound S48
1H NMR(500MHz,DMSO-d6)δ8.27(d,J=2.3Hz,1H),8.10(t,J=5.8Hz,1H),7.66(s,1H),7.62-7.54(m,3H),7.48-7.40(m,3H),4.65(t,J=1.0Hz,2H),4.12(d,J=5.9Hz,2H),3.88(d,J=13.6Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.27 (d, J=2.3 Hz, 1H), 8.10 (t, J=5.8 Hz, 1H), 7.66 (s, 1H), 7.62-7.54 (m, 3H), 7.48-7.40 (m, 3H), 4.65 (t, J=1.0Hz, 2H), 4.12 (d, J=5.9Hz, 2H), 3.88 (d, J=13.6Hz, 6H).
实施例43:化合物S49的合成Example 43: Synthesis of Compound S49
1H NMR(500MHz,DMSO-d6)δ8.26(d,J=2.2Hz,1H),8.10(t,J=5.8Hz,1H),7.55(d,J=2.2Hz,1H),7.46(s,1H),7.40-7.33(m,3H),7.15-7.09(m,2H),4.63(t,J=1.0Hz,2H),4.04(d,J=5.9Hz,2H),3.88(d,J=11.4Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.26 (d, J=2.2 Hz, 1H), 8.10 (t, J=5.8 Hz, 1H), 7.55 (d, J=2.2 Hz, 1H), 7.46 (s, 1H), 7.40-7.33 (m, 3H), 7.15-7.09 (m, 2H), 4.63 (t, J=1.0Hz, 2H), 4.04 (d, J=5.9Hz, 2H), 3.88 ( d, J=11.4Hz, 6H).
实施例44:化合物S50的合成Example 44: Synthesis of Compound S50
1H NMR(500MHz,DMSO-d6)δ8.27(d,J=2.3Hz,1H),8.10(t,J=5.8Hz,1H),7.85-7.79(m,2H),7.59(s,1H),7.53(d,J=2.2Hz,1H),7.47-7.38(m,3H),6.99(s,2H),4.65(t,J=1.0Hz,2H),4.12(d,J=5.9Hz,2H),3.89(d,J=12.6Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.27 (d, J=2.3 Hz, 1H), 8.10 (t, J=5.8 Hz, 1H), 7.85-7.79 (m, 2H), 7.59 (s, 1H), 7.53(d, J=2.2Hz, 1H), 7.47-7.38(m, 3H), 6.99(s, 2H), 4.65(t, J=1.0Hz, 2H), 4.12(d, J=5.9 Hz, 2H), 3.89 (d, J=12.6Hz, 6H).
实施例45:化合物S51的合成Example 45: Synthesis of Compound S51
1H NMR(500MHz,DMSO-d6)δ8.26(d,J=2.3Hz,1H),8.14(t,J=5.8Hz,1H),7.59-7.53(m,2H),7.47(s,1H),7.32(dt,J=8.4,1.1Hz,2H),7.11(dp,J=8.2,0.9Hz,2H),4.65(t,J=1.0Hz,2H),4.09(d,J=5.9Hz,2H),3.89(d,J=15.2Hz,6H),2.33(d,J=0.8Hz,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.26 (d, J=2.3 Hz, 1H), 8.14 (t, J=5.8 Hz, 1H), 7.59-7.53 (m, 2H), 7.47 (s, 1H), 7.32 (dt, J=8.4, 1.1Hz, 2H), 7.11 (dp, J=8.2, 0.9Hz, 2H), 4.65 (t, J=1.0Hz, 2H), 4.09 (d, J=5.9 Hz, 2H), 3.89 (d, J=15.2Hz, 6H), 2.33 (d, J=0.8Hz, 3H).
实施例46:化合物S52的合成Example 46: Synthesis of Compound S52
1H NMR(500MHz,DMSO-d6)δ8.38(s,1H),8.30(d,J=5.0Hz,1H),8.23(t,J=5.8Hz,1H),7.51(ddt,J=8.3,4.9,1.0Hz,2H),7.25(s,1H),7.20-7.12(m,2H),4.67(t,J=1.0Hz,2H),3.94(d,J=5.9Hz,2H),3.89(d,J=10.6Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.38 (s, 1H), 8.30 (d, J=5.0 Hz, 1H), 8.23 (t, J=5.8 Hz, 1H), 7.51 (ddt, J= 8.3, 4.9, 1.0Hz, 2H), 7.25 (s, 1H), 7.20-7.12 (m, 2H), 4.67 (t, J=1.0Hz, 2H), 3.94 (d, J=5.9Hz, 2H), 3.89(d, J=10.6Hz, 6H).
实施例47:化合物S53的合成Example 47: Synthesis of Compound S53
1H NMR(500MHz,DMSO-d6)δ8.40(dd,J=8.0,1.5Hz,1H),8.35(s,1H),8.29(d,J=1.5Hz,1H),8.18-8.10(m,2H),7.57(d,J=2.1Hz,1H),7.49-7.40(m,2H),4.80(s,2H),3.94(d,J=5.7Hz,2H),3.89(d,J=15.2Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.40 (dd, J=8.0, 1.5 Hz, 1H), 8.35 (s, 1H), 8.29 (d, J=1.5 Hz, 1H), 8.18-8.10 ( m, 2H), 7.57 (d, J=2.1Hz, 1H), 7.49-7.40 (m, 2H), 4.80 (s, 2H), 3.94 (d, J=5.7Hz, 2H), 3.89 (d, J =15.2Hz, 6H).
实施例48:化合物S54的合成Example 48: Synthesis of Compound S54
1H NMR(500MHz,DMSO-d6)δ8.30(d,J=2.3Hz,1H),8.15(t,J=5.8Hz,1H),7.98(d,J=2.3Hz,1H),7.37(s,1H),7.12(s,1H),6.91(t,J=9.2Hz,1H),5.71(dd,J=9.3,1.8Hz,1H),5.49(dd,J=9.3,2.0Hz,1H),3.99(d,J=5.9Hz,2H),3.90(s,3H),3.87(p,J=6.5Hz,1H),1.24(d,J=6.6Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.30 (d, J=2.3 Hz, 1H), 8.15 (t, J=5.8 Hz, 1H), 7.98 (d, J=2.3 Hz, 1H), 7.37 (s, 1H), 7.12 (s, 1H), 6.91 (t, J=9.2Hz, 1H), 5.71 (dd, J=9.3, 1.8Hz, 1H), 5.49 (dd, J=9.3, 2.0Hz, 1H), 3.99 (d, J=5.9Hz, 2H), 3.90 (s, 3H), 3.87 (p, J=6.5Hz, 1H), 1.24 (d, J=6.6Hz, 6H).
实施例49:化合物S55的合成Example 49: Synthesis of Compound S55
1H NMR(500MHz,DMSO-d6)δ8.15(t,J=5.8Hz,1H),8.00(s,1H),7.73(d,J=2.2Hz,1H),7.44(d,J=2.3Hz,1H),6.43(s,1H),4.06-3.92(m,2H),3.92-3.80(m,4H),1.23(d,J=6.6Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.15 (t, J=5.8 Hz, 1H), 8.00 (s, 1H), 7.73 (d, J=2.2 Hz, 1H), 7.44 (d, J= 2.3Hz, 1H), 6.43(s, 1H), 4.06-3.92(m, 2H), 3.92-3.80(m, 4H), 1.23(d, J=6.6Hz, 6H).
实施例50:化合物S56的合成Example 50: Synthesis of Compound S56
1H NMR(500MHz,DMSO-d6)δ8.31(d,J=2.1Hz,1H),8.12(t,J=5.8Hz,1H),8.06-8.00(m,2H),7.14(s,1H),3.99(d,J=5.9Hz,2H),3.86(p,J=6.5Hz,1H),1.24(d,J=6.6Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.31 (d, J=2.1 Hz, 1H), 8.12 (t, J=5.8 Hz, 1H), 8.06-8.00 (m, 2H), 7.14 (s, 1H), 3.99 (d, J=5.9Hz, 2H), 3.86 (p, J=6.5Hz, 1H), 1.24 (d, J=6.6Hz, 6H).
实施例51:化合物S57的合成Example 51: Synthesis of Compound S57
1H NMR(500MHz,DMSO-d6)δ8.25(d,J=2.1Hz,1H),8.15(t,J=5.8Hz,1H),7.33(s,1H),7.20(d,J=2.2Hz,1H),7.13(s,1H),6.18(q,J=4.6Hz,1H),3.99(d,J=5.9Hz,2H),3.86(s,3H),3.86(p,J=6.5Hz,1H),2.97(d,J=4.6Hz,3H),1.24(d,J=6.4Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.25 (d, J=2.1 Hz, 1H), 8.15 (t, J=5.8 Hz, 1H), 7.33 (s, 1H), 7.20 (d, J= 2.2Hz, 1H), 7.13 (s, 1H), 6.18 (q, J=4.6Hz, 1H), 3.99 (d, J=5.9Hz, 2H), 3.86 (s, 3H), 3.86 (p, J= 6.5Hz, 1H), 2.97 (d, J=4.6Hz, 3H), 1.24 (d, J=6.4Hz, 6H).
实施例52:化合物S58的合成Example 52: Synthesis of Compound S58
1H NMR(500MHz,DMSO-d6)δ8.15(t,J=5.8Hz,1H),7.97(s,1H),7.78(d,J=2.2Hz,1H),7.29(d,J=2.1Hz,1H),7.12(s,1H),6.87(t,J=13.5Hz,1H),5.69(dd,J=13.5,1.9Hz,1H),5.47(dd,J=13.5,1.9Hz,1H),3.98(d,J=5.7Hz,2H),3.90(s,3H),3.87(p,J=6.5Hz,1H),1.24(d,J=6.4Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.15 (t, J=5.8 Hz, 1H), 7.97 (s, 1H), 7.78 (d, J=2.2 Hz, 1H), 7.29 (d, J= 2.1Hz, 1H), 7.12 (s, 1H), 6.87 (t, J=13.5Hz, 1H), 5.69 (dd, J=13.5, 1.9Hz, 1H), 5.47 (dd, J=13.5, 1.9Hz, 1H), 3.98 (d, J=5.7Hz, 2H), 3.90 (s, 3H), 3.87 (p, J=6.5Hz, 1H), 1.24 (d, J=6.4Hz, 6H).
实施例53:化合物S59的合成Example 53: Synthesis of Compound S59
1H NMR(500MHz,DMSO-d6)δ8.29-8.19(m,2H),7.56-7.49(m,3H),7.44-7.36(m,3H),7.33(s,1H),7.18(tt,J=7.3,1.5Hz,1H),4.14(d,J=5.7Hz,2H),3.89(d,J=12.1Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.29-8.19 (m, 2H), 7.56-7.49 (m, 3H), 7.44-7.36 (m, 3H), 7.33 (s, 1H), 7.18 (tt , J=7.3, 1.5Hz, 1H), 4.14 (d, J=5.7Hz, 2H), 3.89 (d, J=12.1Hz, 6H).
实施例54:化合物S60的合成Example 54: Synthesis of Compound S60
1H NMR(500MHz,DMSO-d6)δ8.23(t,J=5.8Hz,1H),7.78(d,J=2.1Hz,1H),7.60-7.53(m,3H),7.50(s,1H),7.20-7.14(m,2H),5.67(s,1H),4.15(d,J=5.9Hz,2H),3.89(d,J=14.3Hz,6H),2.33(d,J=0.9Hz,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.23 (t, J=5.8 Hz, 1H), 7.78 (d, J=2.1 Hz, 1H), 7.60-7.53 (m, 3H), 7.50 (s, 1H), 7.20-7.14(m, 2H), 5.67(s, 1H), 4.15(d, J=5.9Hz, 2H), 3.89(d, J=14.3Hz, 6H), 2.33(d, J=0.9 Hz, 3H).
实施例55:化合物S61的合成Example 55: Synthesis of Compound S61
1H NMR(500MHz,DMSO-d6)δ8.29(d,J=2.0Hz,1H),8.23(t,J=5.8Hz,1H),8.15(s,1H),7.57(d,J=2.1Hz,1H),7.49-7.42(m,3H),7.21-7.13(m,2H),4.12(d,J=5.9Hz,2H),3.89(d,J=13.6Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.29 (d, J=2.0 Hz, 1H), 8.23 (t, J=5.8 Hz, 1H), 8.15 (s, 1H), 7.57 (d, J= 2.1Hz, 1H), 7.49-7.42 (m, 3H), 7.21-7.13 (m, 2H), 4.12 (d, J=5.9Hz, 2H), 3.89 (d, J=13.6Hz, 6H).
实施例56:化合物S62的合成Example 56: Synthesis of Compound S62
1H NMR(500MHz,DMSO-d6)δ8.31-8.24(m,2H),8.03(s,1H),7.74-7.68(m,2H),7.54-7.47(m,3H),7.38(s,1H),4.16(d,J=5.9Hz,2H),3.88(d,J=15.6Hz,6H). 1 H NMR (500MHz, DMSO-d 6 ) δ 8.31-8.24(m, 2H), 8.03(s, 1H), 7.74-7.68(m, 2H), 7.54-7.47(m, 3H), 7.38(s) , 1H), 4.16 (d, J=5.9Hz, 2H), 3.88 (d, J=15.6Hz, 6H).
实施例57:化合物S63的合成Example 57: Synthesis of Compound S63
1H NMR(500MHz,DMSO-d6)δ8.29-8.20(m,2H),8.11(s,1H),7.54(d,J=2.1Hz,1H),7.43-7.37(m,3H),6.99-6.93(m,2H),4.15(d,J=5.9Hz,2H),3.89(d,J=15.2Hz,6H),3.81(s,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.29-8.20 (m, 2H), 8.11 (s, 1H), 7.54 (d, J=2.1 Hz, 1H), 7.43-7.37 (m, 3H), 6.99-6.93(m, 2H), 4.15(d, J=5.9Hz, 2H), 3.89(d, J=15.2Hz, 6H), 3.81(s, 3H).
实施例58:化合物S64的合成Example 58: Synthesis of Compound S64
1H NMR(500MHz,DMSO-d6)δ8.27-8.19(m,2H),7.72(s,1H),7.52(d,J=2.1Hz,1H),7.38(s,1H),7.25-7.18(m,2H),6.79-6.73(m,2H),5.01(s,2H),4.15(d,J=5.9Hz,2H),3.89(d,J=13.5Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.27-8.19 (m, 2H), 7.72 (s, 1H), 7.52 (d, J=2.1 Hz, 1H), 7.38 (s, 1H), 7.25- 7.18(m, 2H), 6.79-6.73(m, 2H), 5.01(s, 2H), 4.15(d, J=5.9Hz, 2H), 3.89(d, J=13.5Hz, 6H).
实施例59:化合物S65的合成Example 59: Synthesis of Compound S65
1H NMR(500MHz,DMSO-d6)δ8.31-8.22(m,1H),8.12(s,1H),7.46-7.39(m,4H),7.23-7.16(m,3H),4.19(d,J=5.7Hz,2H),3.88(d,J=13.6Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.31-8.22 (m, 1H), 8.12 (s, 1H), 7.46-7.39 (m, 4H), 7.23-7.16 (m, 3H), 4.19 (d , J=5.7Hz, 2H), 3.88(d, J=13.6Hz, 6H).
实施例60:化合物S66的合成Example 60: Synthesis of Compound S66
1H NMR(500MHz,DMSO-d6)δ9.16(s,1H),8.27-8.20(m,2H),7.46(d,J=2.1Hz,1H),7.41(s,1H),3.96(d,J=5.7Hz,2H),3.89(d,J=12.6Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 9.16 (s, 1H), 8.27-8.20 (m, 2H), 7.46 (d, J=2.1 Hz, 1H), 7.41 (s, 1H), 3.96 ( d, J=5.7Hz, 2H), 3.89 (d, J=12.6Hz, 6H).
实施例61:化合物S67的合成Example 61: Synthesis of compound S67
1H NMR(500MHz,DMSO-d6)δ8.69(s,1H),8.27-8.18(m,2H),7.45-7.38(m,2H),4.00-3.94(m,5H),3.89(d,J=9.5Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.69 (s, 1H), 8.27-8.18 (m, 2H), 7.45-7.38 (m, 2H), 4.00-3.94 (m, 5H), 3.89 (d , J=9.5Hz, 6H).
实施例62:化合物S68的合成Example 62: Synthesis of Compound S68
1H NMR(500MHz,DMSO-d6)δ9.59(s,1H),8.28(d,J=2.3Hz,1H),8.20(t,J=5.8Hz,1H),7.43-7.35(m,2H),3.99(d,J=5.7Hz,2H),3.89(d,J=13.7Hz,6H),3.11(s,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 9.59 (s, 1H), 8.28 (d, J=2.3 Hz, 1H), 8.20 (t, J=5.8 Hz, 1H), 7.43-7.35 (m, 2H), 3.99(d, J=5.7Hz, 2H), 3.89(d, J=13.7Hz, 6H), 3.11(s, 3H).
实施例63:化合物S69的合成Example 63: Synthesis of Compound S69
1H NMR(500MHz,DMSO-d6)δ8.31-8.22(m,2H),8.13(s,1H),7.48-7.40(m,2H),3.99(d,J=5.7Hz,2H),3.90(d,J=12.8Hz,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.31-8.22 (m, 2H), 8.13 (s, 1H), 7.48-7.40 (m, 2H), 3.99 (d, J=5.7Hz, 2H), 3.90(d, J=12.8Hz, 6H).
实施例64:化合物S70的合成Example 64: Synthesis of Compound S70
1H NMR(500MHz,DMSO-d6)δ8.43(d,J=5.0Hz,1H),8.26(t,J=5.8Hz,1H),8.06(s,1H),7.21(s,1H),3.99(d,J=5.9Hz,2H),3.93(s,3H),3.88(s,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.43 (d, J=5.0 Hz, 1H), 8.26 (t, J=5.8 Hz, 1H), 8.06 (s, 1H), 7.21 (s, 1H) , 3.99(d, J=5.9Hz, 2H), 3.93(s, 3H), 3.88(s, 3H).
实施例65:化合物S71和S72的合成Example 65: Synthesis of Compounds S71 and S72
步骤一:化合物10的合成Step 1: Synthesis of Compound 10
将化合物6(10g,42mmol)溶于无水DMF(80mL)中,依次加入化合物9(4.9g,50.4mmol)、EDCI(13.7g,71.4mmol)和TEA(29mL,210mmol)。室温下过夜反应,待反应完成后,减压蒸掉溶剂。向反应残渣中加入水(20mL),乙酸乙酯萃取(50mL×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得到化合物10(10g,87%)。Compound 6 (10 g, 42 mmol) was dissolved in dry DMF (80 mL), and compound 9 (4.9 g, 50.4 mmol), EDCI (13.7 g, 71.4 mmol) and TEA (29 mL, 210 mmol) were added sequentially. The reaction was carried out overnight at room temperature. After the reaction was completed, the solvent was evaporated under reduced pressure. Water (20 mL) was added to the reaction residue, extracted with ethyl acetate (50 mL×3), the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain compound 10 (10 g, 87 %).
步骤二:化合物11的合成Step 2: Synthesis of Compound 11
氮气保护下,将化合物10(10g,35.1mmol)溶于无水THF(80mL)中,置于0℃下,将甲基溴化镁的四氢呋喃溶液(1M,36mL,35.1mmol)缓慢滴加至上述溶液中,滴加完毕后,转移至室温,搅拌反应4h。带反应完全后,置于0℃下,缓慢滴加稀盐酸,直至无气泡产生。减压蒸掉溶剂,乙酸乙酯萃取(50mL×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得到化合物11(7.3g,88%)。Under nitrogen protection, compound 10 (10 g, 35.1 mmol) was dissolved in anhydrous THF (80 mL), and at 0 °C, a solution of methylmagnesium bromide in tetrahydrofuran (1 M, 36 mL, 35.1 mmol) was slowly added dropwise to In the above solution, after the dropwise addition was completed, it was transferred to room temperature, and the reaction was stirred for 4h. After the reaction was completed, place it at 0°C and slowly add dilute hydrochloric acid dropwise until no bubbles were generated. The solvent was evaporated under reduced pressure, extracted with ethyl acetate (50 mL×3), the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain compound 11 (7.3 g, 88%).
步骤三:化合物12的合成Step 3: Synthesis of Compound 12
氮气保护下,将化合物11(7g,30mmol)溶于无水THF(100mL)中,加入HMPA(3.2mL,18mmol),置于-78℃下冷却,然后向上述溶液中缓慢滴加LDA的THF溶液(1M,36mL,36mmol),保持此温度反应30min。然后,将溴乙酸乙酯缓慢滴加至上述溶液中,滴加完毕,转移至室温,过夜反应。待反应完全后,置于0℃下,缓慢滴加饱和氯化铵溶液,直至无气泡产生。减压旋掉溶剂,乙酸乙酯萃取(50mL×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得到化合物12(6.8g,70%)。Under nitrogen protection, compound 11 (7 g, 30 mmol) was dissolved in anhydrous THF (100 mL), HMPA (3.2 mL, 18 mmol) was added, cooled at -78 °C, and then LDA in THF was slowly added dropwise to the above solution. The solution (1M, 36 mL, 36 mmol) was kept at this temperature for 30 min. Then, ethyl bromoacetate was slowly added dropwise to the above solution. After the dropwise addition was completed, the solution was transferred to room temperature and reacted overnight. After the reaction was completed, put it at 0 °C, and slowly add saturated ammonium chloride solution dropwise until no bubbles were generated. The solvent was removed under reduced pressure, extracted with ethyl acetate (50 mL×3), the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain compound 12 (6.8 g, 70%).
步骤四:化合物S71的合成Step 4: Synthesis of Compound S71
将盐酸羟胺(13g,186mmol)于乙醇/水的混合溶液(1∶1,60mL),室温下搅拌,待完全溶解后,将原料12(6g,18.6mmol)加入到上述溶液中,加热至回流,过夜反应。待反应完全后,减压蒸掉乙醇和水,柱层析分离纯化得到化合物S71(6.3g,45%)。1H NMR (500MHz,DMSO-d6)δ11.52(s,1H),7.58(s,1H),7.49(s,1H),7.37(s,1H),4.08(q,J=7.1Hz,2H),3.85(s,3H),3.83(s,3H),3.00(t,J=7.9,Hz,2H),2.60(t,J=7.9Hz,2H),1.19(t,J=7.1Hz,3H).A mixed solution (1:1, 60 mL) of hydroxylamine hydrochloride (13 g, 186 mmol) in ethanol/water was stirred at room temperature. After it was completely dissolved, raw material 12 (6 g, 18.6 mmol) was added to the above solution, and heated to reflux. , overnight reaction. After the reaction was completed, ethanol and water were evaporated under reduced pressure, and the compound S71 (6.3 g, 45%) was obtained by column chromatography for separation and purification. 1 H NMR (500 MHz, DMSO-d 6 ) δ 11.52 (s, 1H), 7.58 (s, 1H), 7.49 (s, 1H), 7.37 (s, 1H), 4.08 (q, J=7.1 Hz, 2H), 3.85(s, 3H), 3.83(s, 3H), 3.00(t, J=7.9, Hz, 2H), 2.60(t, J=7.9Hz, 2H), 1.19(t, J=7.1Hz) , 3H).
步骤五:化合物S72的合成Step 5: Synthesis of Compound S72
将化合物S71(100mg,0.3mmol)溶于THF(2mL)中,置于0℃下,缓慢滴加氢氧化钠水溶液(1M,0.6mL,0.6mmol),转移至室温,反应2h。待反应完成后,转移至0℃下,加入稀盐酸(1M),调节pH至中性,冷冻干燥得到化合物S72(93mg,95%)。1H NMR(500MHz,DMSO-d6)δ11.52(s,1H),7.58(s,1H),7.49(s,1H),7.37(s,1H),3.85(s,3H),3.83(s,3H),3.00(t,J=7.9,Hz,2H),2.60(t,J=7.9Hz,2H).Compound S71 (100 mg, 0.3 mmol) was dissolved in THF (2 mL), placed at 0 °C, slowly added dropwise with aqueous sodium hydroxide solution (1 M, 0.6 mL, 0.6 mmol), transferred to room temperature, and reacted for 2 h. After the reaction was completed, it was transferred to 0° C., diluted hydrochloric acid (1 M) was added, the pH was adjusted to neutrality, and the compound S72 (93 mg, 95%) was obtained by lyophilization. 1 H NMR (500 MHz, DMSO-d 6 ) δ 11.52 (s, 1H), 7.58 (s, 1H), 7.49 (s, 1H), 7.37 (s, 1H), 3.85 (s, 3H), 3.83 ( s, 3H), 3.00 (t, J=7.9, Hz, 2H), 2.60 (t, J=7.9 Hz, 2H).
实施例66:化合物S73的合成Example 66: Synthesis of Compound S73
将原料S72(100mg,0.32mmol)溶于THF(5mL)中,向上述反应液中加入乙酸氯甲酯(139mg,1.28mmol)KI(212mg,1.28mmol)和碳酸铯(209mg,0.64mmol),室温下反应5h。待反应完全后,抽滤,滤液浓缩,加入水(2mL),乙酸乙酯萃取(5mL×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得到化合物S73。1H NMR(500MHz,DMSO-d6)δ11.52(s,1H),7.44-7.35(m,1H),7.49(s,1H),7.37(s,1H),5.96(s,2H),3.89(s,3H),3.83(s,3H),3.12(t,J=8.4Hz,2H),2.60(t,J=8.5Hz,2H),2.10(s,3H).The raw material S72 (100 mg, 0.32 mmol) was dissolved in THF (5 mL), chloromethyl acetate (139 mg, 1.28 mmol) KI (212 mg, 1.28 mmol) and cesium carbonate (209 mg, 0.64 mmol) were added to the above reaction solution, The reaction was carried out for 5h at room temperature. After the reaction was completed, suction filtered, concentrated the filtrate, added water (2 mL), extracted with ethyl acetate (5 mL×3), combined the organic phases, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography Compound S73 is obtained. 1 H NMR (500 MHz, DMSO-d 6 ) δ 11.52 (s, 1H), 7.44-7.35 (m, 1H), 7.49 (s, 1H), 7.37 (s, 1H), 5.96 (s, 2H), 3.89(s, 3H), 3.83(s, 3H), 3.12(t, J=8.4Hz, 2H), 2.60(t, J=8.5Hz, 2H), 2.10(s, 3H).
实施例67:化合物S5的合成Example 67: Synthesis of Compound S5
步骤一:化合物13的合成Step 1: Synthesis of Compound 13
将化合物12(200mg,0.62mmol)溶于甲醇(2mL)中,置于0℃下,加入NaOH水溶液(1M,1.2mL,1.2mmol),室温下搅拌反应2h。待反应完全后,置于0℃下,加入稀盐酸调节pH至2~3,沉淀析出,抽滤,甲醇洗涤滤饼,收集滤饼、干燥得到化合物13(164mg,90%),直接投入下一步反应。Compound 12 (200 mg, 0.62 mmol) was dissolved in methanol (2 mL), placed at 0 °C, NaOH aqueous solution (1 M, 1.2 mL, 1.2 mmol) was added, and the reaction was stirred at room temperature for 2 h. After the reaction was completed, it was placed at 0° C., and diluted hydrochloric acid was added to adjust the pH to 2 to 3. The solution was precipitated, filtered with suction, and the filter cake was washed with methanol. The filter cake was collected and dried to obtain compound 13 (164 mg, 90%), which was directly put into one-step reaction.
步骤二:化合物S5的合成Step 2: Synthesis of Compound S5
将化合物13(100mg,0.34mmol)溶于无水DMF(5mL)中,依次加入HATU(194mg,0.51mmol),DIPEA(219mg,1.7mmol)和甲磺酰胺(36mg,0.37mmol)。室温下搅拌,过夜反应。待反应完全后,减压旋掉DMF,加入水(1mL),乙酸乙酯萃取(5mL×3),合并有机相,无水硫酸钠干燥,过滤,浓缩,柱层析分离纯化得到化合物S5(71mg,56%)。1H NMR(500MHz,DMSO-d6)δ8.08(d,J=2.3Hz,1H),7.52(d,J=2.1Hz,1H),7.45(s,1H),4.36(s,1H),3.89(d,J=11.9Hz,6H),3.54(t,J=8.4Hz,2H),3.14(s,3H),2.62(t,J=8.5Hz,2H).Compound 13 (100 mg, 0.34 mmol) was dissolved in dry DMF (5 mL) and HATU (194 mg, 0.51 mmol), DIPEA (219 mg, 1.7 mmol) and methanesulfonamide (36 mg, 0.37 mmol) were added sequentially. Stir at room temperature and react overnight. After the reaction was complete, spin off DMF under reduced pressure, add water (1 mL), extract with ethyl acetate (5 mL×3), combine the organic phases, dry over anhydrous sodium sulfate, filter, concentrate, and separate and purify by column chromatography to obtain compound S5 ( 71 mg, 56%). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.08 (d, J=2.3 Hz, 1H), 7.52 (d, J=2.1 Hz, 1H), 7.45 (s, 1H), 4.36 (s, 1H) , 3.89(d, J=11.9Hz, 6H), 3.54(t, J=8.4Hz, 2H), 3.14(s, 3H), 2.62(t, J=8.5Hz, 2H).
实施例68:化合物S6的合成Example 68: Synthesis of Compound S6
合成参照实施例67的合成方法,只需更换相应的原料即可。The synthesis refers to the synthesis method of Example 67, and only needs to replace the corresponding raw materials.
1H NMR(500MHz,DMSO-d6)δ8.08(d,J=2.0Hz,2H),7.48(d,J=2.2Hz,1H),7.43(s,1H),3.89(d,J=11.0Hz,6H),3.55(t,J=8.4Hz,2H),3.22-3.03(m,2H),2.62(t,J=8.4Hz,2H),1.35(t,J=6.9Hz,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.08 (d, J=2.0 Hz, 2H), 7.48 (d, J=2.2 Hz, 1H), 7.43 (s, 1H), 3.89 (d, J= 11.0Hz, 6H), 3.55 (t, J=8.4Hz, 2H), 3.22-3.03 (m, 2H), 2.62 (t, J=8.4Hz, 2H), 1.35 (t, J=6.9Hz, 3H) .
实施例69:化合物S1的合成Example 69: Synthesis of Compound S1
将化合物13(200mg,0.68mmol)和N-甲基吗啉(0.22mL,2.04mmol)溶于无水THF(5mL)中,置于-78℃下,将氯甲基异丁酯(97μL,0.75mmol)滴加至上述溶液中,保持此温度下搅拌反应15min。然后将甲酰肼(41mg,0.68mmol)加入到上述溶液中,缓慢升温至室温,室温下搅拌反应1h。待反应完全后,加入水淬灭反应,减压蒸掉溶剂,乙酸乙酯萃取(5mL×3),合并有机相,无水硫酸钠干燥,过滤,浓缩得到残渣直接投入下一步反应。Compound 13 (200 mg, 0.68 mmol) and N-methylmorpholine (0.22 mL, 2.04 mmol) were dissolved in dry THF (5 mL), placed at -78 °C, and chloromethyl isobutyl ester (97 μL, 0.75 mmol) was added dropwise to the above solution, and the reaction was stirred at this temperature for 15 min. Then, formic hydrazide (41 mg, 0.68 mmol) was added to the above solution, the temperature was slowly raised to room temperature, and the reaction was stirred at room temperature for 1 h. After the reaction was completed, water was added to quench the reaction, the solvent was evaporated under reduced pressure, and extracted with ethyl acetate (5 mL×3).
将上一步得到的反应残渣溶于DCM中,加入TEA(0.29mL,2.04mmol),向上述溶液中加入对甲苯磺酰氯(195mg,1.02mmol),室温下搅拌反应12h。待反应完全后,加入水淬灭反应,DCM萃取(5mL×3),合并有机相,无水硫酸钠干燥,过滤,浓缩,柱层析分离纯化得到化合物S1(97mg,45%)。1H NMR(500MHz,DMSO-d6)δ8.08(d,J=2.2Hz,2H),7.44-7.36(m,2H),3.90(d,J=12.1Hz,6H),3.54(t,J=7.8Hz,2H),3.19(t,J=7.8Hz,2H).The reaction residue obtained in the previous step was dissolved in DCM, TEA (0.29 mL, 2.04 mmol) was added, p-toluenesulfonyl chloride (195 mg, 1.02 mmol) was added to the above solution, and the reaction was stirred at room temperature for 12 h. After the reaction was complete, water was added to quench the reaction, extracted with DCM (5 mL×3), the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated, and separated and purified by column chromatography to obtain compound S1 (97 mg, 45%). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.08 (d, J=2.2 Hz, 2H), 7.44-7.36 (m, 2H), 3.90 (d, J=12.1 Hz, 6H), 3.54 (t, J=7.8Hz, 2H), 3.19(t, J=7.8Hz, 2H).
实施例70:化合物S2的合成Example 70: Synthesis of Compound S2
将化合物13(200mg,0.68mmol)和N-甲基吗啉(0.22mL,2.04mmol)溶于无水THF(5mL)中,置于-78℃下,将氯甲基异丁酯(97μL,0.75mmol)滴加至上述溶液中,保持此温度下搅拌反应15min。然后将化合物14(51mg,0.68mmol)加入到上述溶液中,缓慢升温至室温,室温下搅拌反应1h。待反应完全后,加入水淬灭反应,减压蒸掉溶剂,乙酸乙酯萃取(5mL×3),合并有机相,无水硫酸钠干燥,过滤,浓缩得到残渣直接投入下一步反应。Compound 13 (200 mg, 0.68 mmol) and N-methylmorpholine (0.22 mL, 2.04 mmol) were dissolved in dry THF (5 mL), placed at -78 °C, and chloromethyl isobutyl ester (97 μL, 0.75 mmol) was added dropwise to the above solution, and the reaction was stirred at this temperature for 15 min. Then compound 14 (51 mg, 0.68 mmol) was added to the above solution, the temperature was slowly raised to room temperature, and the reaction was stirred at room temperature for 1 h. After the reaction was completed, water was added to quench the reaction, the solvent was evaporated under reduced pressure, and extracted with ethyl acetate (5 mL×3).
将上述反应残渣溶于吡啶中,加热回流,过夜反应。待反应完全后,冷却至室温,加入水淬灭反应。减压蒸掉溶剂,乙酸乙酯萃取(5mL×3),合并有机相,无水硫酸钠干燥,过滤,浓缩,柱层析分离纯化制得化合物S2(90mg,40%)。1H NMR(500MHz,DMSO-d6)δ8.09(d,J=2.3Hz,1H),7.52-7.46(m,2H),3.89(d,J=14.1Hz,6H),3.62-3.54(m,3H),3.44-3.37(m,2H),2.39(s,2H).The above reaction residue was dissolved in pyridine, heated to reflux, and reacted overnight. After the reaction was completed, it was cooled to room temperature, and water was added to quench the reaction. The solvent was evaporated under reduced pressure, extracted with ethyl acetate (5 mL×3), the organic phases were combined, dried over anhydrous sodium sulfate, filtered, concentrated, and separated and purified by column chromatography to obtain compound S2 (90 mg, 40%). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.09 (d, J=2.3 Hz, 1H), 7.52-7.46 (m, 2H), 3.89 (d, J=14.1 Hz, 6H), 3.62-3.54 ( m, 3H), 3.44-3.37 (m, 2H), 2.39 (s, 2H).
实施例71:化合物S3的合成Example 71: Synthesis of Compound S3
步骤一:化合物14的合成Step 1: Synthesis of Compound 14
氮气保护下,将化合物11(2g,8.5mmol)溶于无水THF(10mL)中,加入HMPA(0.9mL,5.0mmol),置于-78℃下冷却,然后向上述溶液中缓慢滴加LDA的THF溶液(1M,9.5mL,9.4mmol),保持此温度反应30min。然后,将溴乙腈缓慢滴加至上述溶液中,滴加完毕,转移至室温,过夜反应。待反应完全后,置于0℃下,缓慢滴加饱和氯化铵溶液,直至无气泡产生。减压旋掉溶剂,乙酸乙酯萃取(10mL×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得到化合物14(1.1g,50%)。Under nitrogen protection, compound 11 (2 g, 8.5 mmol) was dissolved in anhydrous THF (10 mL), HMPA (0.9 mL, 5.0 mmol) was added, cooled at -78 °C, and then LDA was slowly added dropwise to the above solution. solution in THF (1 M, 9.5 mL, 9.4 mmol), and the reaction was maintained at this temperature for 30 min. Then, bromoacetonitrile was slowly added dropwise to the above solution, the dropwise addition was completed, and the solution was transferred to room temperature and reacted overnight. After the reaction was completed, put it at 0 °C, and slowly add saturated ammonium chloride solution dropwise until no bubbles were generated. The solvent was removed under reduced pressure, extracted with ethyl acetate (10 mL×3), the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain compound 14 (1.1 g, 50%).
步骤二:化合物15的合成Step 2: Synthesis of Compound 15
将化合物14(1g,3.83mmol)溶于乙醇(10mL)中,加入碳酸钾(1.6g,11.5mmol)和盐酸羟胺(798mg,11.5mmol),加热回流,过夜反应。待反应完全后,减压蒸掉溶剂,加入水,乙酸乙酯萃取(10mL×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得到化合物15(943mg,80%)。Compound 14 (1 g, 3.83 mmol) was dissolved in ethanol (10 mL), potassium carbonate (1.6 g, 11.5 mmol) and hydroxylamine hydrochloride (798 mg, 11.5 mmol) were added, heated to reflux, and reacted overnight. After the reaction was completed, the solvent was evaporated under reduced pressure, water was added, extracted with ethyl acetate (10 mL×3), the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain compound 15 (943 mg, 80%).
步骤三:化合物S3的合成Step 3: Synthesis of Compound S3
将化合物15(200mg,0.65mmol)加入到原甲酸三乙酯(5mL)中,加入催化量的三氟乙酸,室温下搅拌10min,然后升温至60℃,搅拌反应30min。待反应完全后,冷却至室温,减压浓缩,加入水,乙酸乙酯萃取(10mL×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得到化合物S3(103mg,50%)。1H NMR(500MHz,DMSO-d6)δ9.33(s,1H),8.10(d,J=2.1Hz,1H),7.51(s,1H),7.45(d,J=2.2Hz,1H),3.90(d,J=12.1Hz,6H),3.41(t,J=7.8Hz,2H),2.96(t,J=7.8Hz,2H).Compound 15 (200 mg, 0.65 mmol) was added to triethyl orthoformate (5 mL), a catalytic amount of trifluoroacetic acid was added, and the mixture was stirred at room temperature for 10 min, then heated to 60° C. and stirred for 30 min. After the reaction was completed, cooled to room temperature, concentrated under reduced pressure, added water, extracted with ethyl acetate (10 mL×3), combined the organic phases, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain Compound S3 (103 mg, 50%). 1 H NMR (500 MHz, DMSO-d 6 ) δ 9.33 (s, 1H), 8.10 (d, J=2.1 Hz, 1H), 7.51 (s, 1H), 7.45 (d, J=2.2 Hz, 1H) , 3.90(d, J=12.1Hz, 6H), 3.41(t, J=7.8Hz, 2H), 2.96(t, J=7.8Hz, 2H).
实施例72:化合物S4的合成Example 72: Synthesis of Compound S4
将化合物13(200mg,0.68mmol)和N-甲基吗啉(0.22mL,2.04mmol)溶于无水THF(5mL)中,置于-78℃下,将氯甲基异丁酯(97μL,0.75mmol)滴加至上述溶液中,保持此温度下搅拌反应15min。然后将甲酰肼(41mg,0.68mmol)加入到上述溶液中,缓慢升温至室温,室温下搅拌反应1h。待反应完全后,加入水淬灭反应,减压蒸掉溶剂,乙酸乙酯萃取(5mL×3),合并有机相,无水硫酸钠干燥,过滤,浓缩得到残渣直接投入下一步反应。Compound 13 (200 mg, 0.68 mmol) and N-methylmorpholine (0.22 mL, 2.04 mmol) were dissolved in dry THF (5 mL), placed at -78 °C, and chloromethyl isobutyl ester (97 μL, 0.75 mmol) was added dropwise to the above solution, and the reaction was stirred at this temperature for 15 min. Then, formic hydrazide (41 mg, 0.68 mmol) was added to the above solution, the temperature was slowly raised to room temperature, and the reaction was stirred at room temperature for 1 h. After the reaction was completed, water was added to quench the reaction, the solvent was evaporated under reduced pressure, and extracted with ethyl acetate (5 mL×3).
将上述反应残渣溶于甲苯(5mL)中,加入劳森试剂(412mg,1.02mmol),加热回流,过夜反应。带反应完全后,减压蒸掉溶剂,柱层析分离纯化制得化合物S4(73mg,32%)。1HNMR(500MHz,DMSO-d6)δ8.90(s,1H),8.10(d,J=2.3Hz,1H),7.57-7.51(m,2H),3.90(d,J=11.5Hz,6H),3.45(dd,J=8.2,7.5Hz,2H),3.30-3.23(m,2H).The above reaction residue was dissolved in toluene (5 mL), Lawson's reagent (412 mg, 1.02 mmol) was added, and the mixture was heated to reflux and reacted overnight. After the reaction was completed, the solvent was evaporated under reduced pressure, and the compound S4 (73 mg, 32%) was obtained by separation and purification by column chromatography. 1 HNMR (500MHz, DMSO-d 6 ) δ 8.90 (s, 1H), 8.10 (d, J=2.3Hz, 1H), 7.57-7.51 (m, 2H), 3.90 (d, J=11.5Hz, 6H) ), 3.45 (dd, J=8.2, 7.5Hz, 2H), 3.30-3.23 (m, 2H).
以下实施例73~实施例89中的化合物S74~S90的合成参照实施例65的合成方法,只需更换相应的原料即可。The synthesis of compounds S74 to S90 in the following Examples 73 to 89 refers to the synthesis method of Example 65, and only needs to replace the corresponding raw materials.
实施例73:化合物S74的合成Example 73: Synthesis of Compound S74
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),11.52(s,1H),7.58(s,1H),7.51(d,J=4.9Hz,1H),3.94(s,3H),3.87(s,3H),3.11(t,J=8.5Hz,2H),2.56(t,J=8.5Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 11.52 (s, 1H), 7.58 (s, 1H), 7.51 (d, J=4.9 Hz, 1H), 3.94 (s, 3H), 3.87(s, 3H), 3.11(t, J=8.5Hz, 2H), 2.56(t, J=8.5Hz, 2H).
实施例74:化合物S75的合成Example 74: Synthesis of Compound S75
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),11.52(s,1H),7.65(s,1H),7.35(d,J=2.1Hz,1H),7.28(s,1H),3.92(s,3H),3.09(t,J=8.5Hz,2H),2.57(t,J=8.5Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 11.52 (s, 1H), 7.65 (s, 1H), 7.35 (d, J=2.1 Hz, 1H), 7.28 (s, 1H), 3.92(s, 3H), 3.09(t, J=8.5Hz, 2H), 2.57(t, J=8.5Hz, 2H).
实施例75:化合物S76的合成Example 75: Synthesis of Compound S76
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),11.52(s,1H),8.28(s,2H),7.30-7.24(m,1H),6.02(s,2H),3.07(t,J=8.4Hz,2H),2.56(t,J=8.4Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 11.52 (s, 1H), 8.28 (s, 2H), 7.30-7.24 (m, 1H), 6.02 (s, 2H), 3.07(t, J=8.4Hz, 2H), 2.56(t, J=8.4Hz, 2H).
实施例76:化合物S77的合成Example 76: Synthesis of Compound S77
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),11.52(s,1H),7.93(d,J=2.1Hz,1H),7.40(d,J=2.1Hz,1H),7.27(s,1H),6.86(t,J=9.2Hz,1H),5.68(dd,J=9.3,2.0Hz,1H),5.46(dd,J=9.3,1.8Hz,1H),3.87(s,3H),3.17(t,J=8-5Hz,2H),2.57(t,J=8.4Hz,2H). 1 H NMR (500MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 11.52 (s, 1H), 7.93 (d, J=2.1Hz, 1H), 7.40 (d, J=2.1Hz, 1H) , 7.27(s, 1H), 6.86(t, J=9.2Hz, 1H), 5.68(dd, J=9.3, 2.0Hz, 1H), 5.46(dd, J=9.3, 1.8Hz, 1H), 3.87( s, 3H), 3.17 (t, J=8-5Hz, 2H), 2.57 (t, J=8.4Hz, 2H).
实施例77:化合物S78的合成Example 77: Synthesis of Compound S78
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),11.52(s,1H),7.92(d,J=2.1Hz,1H),7.40(d,J=2.1Hz,1H),7.27(s,1H),6.86(t,J=9.2Hz,1H),5.68(dd,J=9.3,2.0Hz,1H),5.46(dd,J=9.3,1.8Hz,1H),3.87(s,3H),3.17(t,J=8.5Hz,2H),2.57(t,J=8.4Hz,2H). 1 H NMR (500MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 11.52 (s, 1H), 7.92 (d, J=2.1Hz, 1H), 7.40 (d, J=2.1Hz, 1H) , 7.27(s, 1H), 6.86(t, J=9.2Hz, 1H), 5.68(dd, J=9.3, 2.0Hz, 1H), 5.46(dd, J=9.3, 1.8Hz, 1H), 3.87( s, 3H), 3.17 (t, J=8.5Hz, 2H), 2.57 (t, J=8.4Hz, 2H).
实施例78:化合物S79的合成Example 78: Synthesis of Compound S79
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),8.26(s,1H),7.34(d,J=2.2Hz,1H),7.18(s,1H),7.09(d,J=2.2Hz,1H),6.11(q,J=4.5Hz,1H),3.86(s,3H),3.16(t,J=8.5Hz,2H),2.97(d,J=4.6Hz,3H),2.57(t,J=8.4Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 8.26 (s, 1H), 7.34 (d, J=2.2 Hz, 1H), 7.18 (s, 1H), 7.09 (d, J=2.2Hz, 1H), 6.11 (q, J=4.5Hz, 1H), 3.86 (s, 3H), 3.16 (t, J=8.5Hz, 2H), 2.97 (d, J=4.6Hz, 3H) , 2.57(t, J=8.4Hz, 2H).
实施例79:化合物S80的合成Example 79: Synthesis of Compound S80
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),11.26(s,1H),7.78(s,1H),7.44-7.37(m,2H),3.89(d,J=7.5Hz,6H),3.15(s,2H),1.31(s,6H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 11.26 (s, 1H), 7.78 (s, 1H), 7.44-7.37 (m, 2H), 3.89 (d, J=7.5 Hz, 6H), 3.15(s, 2H), 1.31(s, 6H).
实施例80:化合物S81的合成Example 80: Synthesis of Compound S81
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),10.65(s,1H),7.46-7.40(m,2H),7.37(d,J=2.2Hz,1H),3.89(d,J=9.7Hz,6H),3.37-3.29(m,1H). 1 H NMR (500MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 10.65 (s, 1H), 7.46-7.40 (m, 2H), 7.37 (d, J=2.2Hz, 1H), 3.89 ( d, J=9.7Hz, 6H), 3.37-3.29 (m, 1H).
实施例81:化合物S82的合成Example 81: Synthesis of Compound S82
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),7.58(s,1H),7.49(s,1H),7.37(s,1H),3.85(s,3H),3.83(s,3H),3.66(s,3H),3.00(t,J=7.9,Hz,2H),2.60(t,J=7.9Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 7.58 (s, 1H), 7.49 (s, 1H), 7.37 (s, 1H), 3.85 (s, 3H), 3.83 ( s, 3H), 3.66 (s, 3H), 3.00 (t, J=7.9, Hz, 2H), 2.60 (t, J=7.9 Hz, 2H).
实施例82:化合物S83的合成Example 82: Synthesis of Compound S83
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),7.46-7.38(m,2H),7.32(d,J=2.2Hz,1H),3.89(d,J=9.9Hz,6H),3.21(t,J=8.4Hz,2H),2.59(t,J=8.4Hz,2H),2.13(s,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 7.46-7.38 (m, 2H), 7.32 (d, J=2.2Hz, 1H), 3.89 (d, J=9.9Hz, 6H), 3.21(t, J=8.4Hz, 2H), 2.59(t, J=8.4Hz, 2H), 2.13(s, 2H).
实施例83:化合物S84的合成Example 83: Synthesis of Compound S84
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),7.46(d,J=2.1Hz,1H),7.38(s,1H),7.28(d,J=2.0Hz,1H),3.89(d,J=5.3Hz,6H),3.04(t,J=8.5Hz,2H),2.57(t,J=8.4Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 7.46 (d, J=2.1 Hz, 1H), 7.38 (s, 1H), 7.28 (d, J=2.0 Hz, 1H) , 3.89(d, J=5.3Hz, 6H), 3.04(t, J=8.5Hz, 2H), 2.57(t, J=8.4Hz, 2H).
实施例84:化合物S85的合成Example 84: Synthesis of Compound S85
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),7.52(d,J=5.0Hz,1H),7.18(s,1H),3.94(s,3H),3.87(s,3H),3.06(t,J=8.4Hz,2H),2.60(t,J=8.5Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 7.52 (d, J=5.0 Hz, 1H), 7.18 (s, 1H), 3.94 (s, 3H), 3.87 (s, 3H), 3.06(t, J=8.4Hz, 2H), 2.60(t, J=8.5Hz, 2H).
实施例85:化合物S86的合成Example 85: Synthesis of Compound S86
1H NMR(500MHz,DMSO-d6)δ7.48(d,J=2.0Hz,1H),7.44-7.38(m,2H),4.10(q,J=7.0Hz,2H),3.89(d,J=9.0Hz,6H),3.04(t,J=8.5Hz,2H),2.61(t,J=8.5Hz,2H),1.26(t,J=7.0Hz,3H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 7.48 (d, J=2.0 Hz, 1H), 7.44-7.38 (m, 2H), 4.10 (q, J=7.0 Hz, 2H), 3.89 (d, J=9.0Hz, 6H), 3.04 (t, J=8.5Hz, 2H), 2.61 (t, J=8.5Hz, 2H), 1.26 (t, J=7.0Hz, 3H).
实施例86:化合物S87的合成Example 86: Synthesis of Compound S87
1H NMR(500MHz,DMSO-d6)δ7.49(d,J=2.1Hz,1H),7.46-7.40(m,2H),3.89(d,J=8.2Hz,6H),3.65(p,J=5.5Hz,1H),3.01(t,J=8.4Hz,2H),2.59(t,J=8.4Hz,2H),1.60(dd,J=5.5,2.3Hz,4H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 7.49 (d, J=2.1 Hz, 1H), 7.46-7.40 (m, 2H), 3.89 (d, J=8.2 Hz, 6H), 3.65 (p, J=5.5Hz, 1H), 3.01 (t, J=8.4Hz, 2H), 2.59 (t, J=8.4Hz, 2H), 1.60 (dd, J=5.5, 2.3Hz, 4H).
实施例87:化合物S88的合成Example 87: Synthesis of Compound S88
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),7.89(d,J=2.3Hz,1H),7.52(d,J=2.1Hz,1H),7.29(s,1H),3.92(s,3H),3.05(t,J=8.5Hz,2H),2.55(t,J=8.4Hz,2H). 1 H NMR (500MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 7.89 (d, J=2.3Hz, 1H), 7.52 (d, J=2.1Hz, 1H), 7.29 (s, 1H) , 3.92(s, 3H), 3.05(t, J=8.5Hz, 2H), 2.55(t, J=8.4Hz, 2H).
实施例88:化合物S89的合成Example 88: Synthesis of Compound S89
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),7.38(d,J=2.1Hz,1H),7.33-7.27(m,2H),6.01(s,2H),3.04(t,J=8.4Hz,2H),2.59(t,J=8.4Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 7.38 (d, J=2.1 Hz, 1H), 7.33-7.27 (m, 2H), 6.01 (s, 2H), 3.04 ( t, J=8.4Hz, 2H), 2.59 (t, J=8.4Hz, 2H).
实施例89:化合物S90的合成Example 89: Synthesis of Compound S90
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),7.42-7.37(m,2H),7.34(d,J=2.2Hz,1H),5.26(s,2H),3.89(d,J=5.3Hz,6H),3.06(t,J=8.5Hz,2H),2.53(t,J=8.4Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 7.42-7.37 (m, 2H), 7.34 (d, J=2.2Hz, 1H), 5.26 (s, 2H), 3.89 ( d, J=5.3Hz, 6H), 3.06 (t, J=8.5Hz, 2H), 2.53 (t, J=8.4Hz, 2H).
实施例90:化合物S91的合成Example 90: Synthesis of Compound S91
将化合物S84(100mg,0.31mmol)溶于无水DMF(5mL)中,依次加入HATU(179mg,0.47mmol)、甲磺酰胺(32mg,0.33mol)和DIPEA(120mg,0.93mmol),室温下搅拌反应5h。待反应完全后,加入水淬灭反应,减压蒸掉DMF,乙酸乙酯萃取(5mL×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得到化合物S91(55mg,45%)。1H NMR(500MHz,DMSO-d6)δ7.58(s,1H),7.49(s,1H),7.37(s,1H),7.10(s,1H),3.89(s,3H),3.74(s,3H),3.14(s,3H),2.95(t,J=8.5Hz,2H),2.57(t,J=8.4Hz,2H).Compound S84 (100 mg, 0.31 mmol) was dissolved in anhydrous DMF (5 mL), HATU (179 mg, 0.47 mmol), methanesulfonamide (32 mg, 0.33 mol) and DIPEA (120 mg, 0.93 mmol) were added in sequence, and stirred at room temperature The reaction was carried out for 5 hours. After the reaction was completed, water was added to quench the reaction, DMF was evaporated under reduced pressure, extracted with ethyl acetate (5 mL×3), the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography Compound S91 was obtained (55 mg, 45%). 1 H NMR (500MHz, DMSO-d 6 ) δ 7.58(s, 1H), 7.49(s, 1H), 7.37(s, 1H), 7.10(s, 1H), 3.89(s, 3H), 3.74( s, 3H), 3.14 (s, 3H), 2.95 (t, J=8.5Hz, 2H), 2.57 (t, J=8.4Hz, 2H).
以下实施例91~实施例93中的S92~S94的合成均可参照实施90中的合成方法,只需更换相应的原料即可。For the synthesis of S92 to S94 in the following Examples 91 to 93, reference can be made to the synthesis method in Example 90, and only the corresponding raw materials need to be replaced.
实施例91:化合物S92的合成Example 91: Synthesis of Compound S92
1H NMR(500MHz,DMSO-d6)δ11.52(s,1H),8.10(s,1H),7.58(s,1H),7.49(s,1H),7.37(s,1H),3.85(s,3H),3.83(s,3H),3.00(t,J=7.9,Hz,2H),2.99(s,3H),2.60(t,J=7.9Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 11.52 (s, 1H), 8.10 (s, 1H), 7.58 (s, 1H), 7.49 (s, 1H), 7.37 (s, 1H), 3.85 ( s, 3H), 3.83 (s, 3H), 3.00 (t, J=7.9, Hz, 2H), 2.99 (s, 3H), 2.60 (t, J=7.9Hz, 2H).
实施例92:化合物S93的合成Example 92: Synthesis of Compound S93
1H NMR(500MHz,DMSO-d6)δ11.52(s,1H),7.96(s,1H),7.79(s,1H),7.39-7.32(m,2H),3.89(d,J=8.2Hz,6H),3.63(s,3H),3.06(t,J=8.5Hz,2H),2.48(t,J=8.5Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 11.52 (s, 1H), 7.96 (s, 1H), 7.79 (s, 1H), 7.39-7.32 (m, 2H), 3.89 (d, J=8.2 Hz, 6H), 3.63 (s, 3H), 3.06 (t, J=8.5Hz, 2H), 2.48 (t, J=8.5Hz, 2H).
实施例93:化合物S94的合成Example 93: Synthesis of Compound S94
1H NMR(500MHz,DMSO-d6)δ8.01(s,1H),7.65(s,1H),7.49(dd,J=17.4,2.0Hz,1H),7.42(s,1H),3.89(d,J=9.0Hz,6H),3.63(s,3H),2.99(t,J=8.5Hz,2H),2.43(t,J=8.5Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 8.01 (s, 1H), 7.65 (s, 1H), 7.49 (dd, J=17.4, 2.0 Hz, 1H), 7.42 (s, 1H), 3.89 ( d, J=9.0Hz, 6H), 3.63 (s, 3H), 2.99 (t, J=8.5Hz, 2H), 2.43 (t, J=8.5Hz, 2H).
实施例94:化合物S95和S96的合成Example 94: Synthesis of Compounds S95 and S96
步骤一:化合物S95的合成Step 1: Synthesis of compound S95
将化合物2(200mg,0.96mmol)溶于无水THF(2mL)中,置于0℃下,将三光气(142mg,0.48mmol)的无水THF(2mL)溶液缓慢滴加至上述溶液中,然后加入三乙胺(0.2mL,1.5mmol),加热回流,过夜反应。待反应完全后,冷却至室温,抽滤,无水THF洗涤滤饼,滤液浓缩,干燥。将干燥得到的残渣重新溶解在2mL无水THF中,置于0℃下,将化合物13的无水THF(2mL)溶液缓慢滴加至上述溶液中,滴加完毕,加热回流反应4h。待反应完成后,冷却至室温,减压蒸掉溶剂,加入水(2mL),乙酸乙酯萃取(5mL×3),合并有机相,饱和食盐水洗涤,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得到化合物S95(169mg,52%)。1H NMR(500MHz,DMSO-d6)δ9.69(s,1H),7.69(d,J=2.1Hz,1H),7.43-7.35(m,2H),6.71(t,J=5.8Hz,1H),4.16(q,J=7.0Hz,2H),3.98-3.89(m,6H),3.87(s,2H),1.24(t,J=6.9Hz,3H).Compound 2 (200 mg, 0.96 mmol) was dissolved in anhydrous THF (2 mL), placed at 0 °C, and a solution of triphosgene (142 mg, 0.48 mmol) in anhydrous THF (2 mL) was slowly added dropwise to the above solution. Then triethylamine (0.2 mL, 1.5 mmol) was added, heated to reflux, and reacted overnight. After the reaction was completed, it was cooled to room temperature, filtered with suction, the filter cake was washed with anhydrous THF, and the filtrate was concentrated and dried. The dried residue was redissolved in 2 mL of anhydrous THF, placed at 0 °C, and the solution of compound 13 in anhydrous THF (2 mL) was slowly added dropwise to the above solution. After the dropwise addition, the reaction was heated under reflux for 4 h. After the reaction was completed, it was cooled to room temperature, the solvent was evaporated under reduced pressure, water (2 mL) was added, extracted with ethyl acetate (5 mL×3), the organic phases were combined, washed with saturated brine, dried over anhydrous sodium sulfate, filtered, and concentrated. Purification by column chromatography gave compound S95 (169 mg, 52%). 1 H NMR (500 MHz, DMSO-d 6 ) δ 9.69 (s, 1H), 7.69 (d, J=2.1 Hz, 1H), 7.43-7.35 (m, 2H), 6.71 (t, J=5.8 Hz, 1H), 4.16 (q, J=7.0Hz, 2H), 3.98-3.89 (m, 6H), 3.87 (s, 2H), 1.24 (t, J=6.9Hz, 3H).
步骤二:化合物S96的合成Step 2: Synthesis of Compound S96
将化合物S95(100mg,0.30mmol)溶于甲醇(2mL)中,将NaOH水溶液(1M,0.6mL,0.6mmol)滴加至上述溶液中,室温下搅拌反应2h。待反应完全后,稀盐酸调节pH至2~3,沉淀析出,抽滤,甲醇洗涤滤饼,收集滤饼、干燥制得化合物S96(84mg,90%)。1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),9.60(s,1H),7.69(d,J=2.1Hz,1H),7.32-7.24(m,2H),7.15(t,J=5.8Hz,1H),4.00(d,J=5.7Hz,2H),3.89(d,J=17.9Hz,6H).Compound S95 (100 mg, 0.30 mmol) was dissolved in methanol (2 mL), NaOH aqueous solution (1 M, 0.6 mL, 0.6 mmol) was added dropwise to the above solution, and the reaction was stirred at room temperature for 2 h. After the reaction was completed, the pH was adjusted to 2-3 with dilute hydrochloric acid, precipitated out, filtered with suction, washed the filter cake with methanol, collected the filter cake, and dried to obtain compound S96 (84 mg, 90%). 1 H NMR (500MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 9.60 (s, 1H), 7.69 (d, J=2.1Hz, 1H), 7.32-7.24 (m, 2H), 7.15 ( t, J=5.8Hz, 1H), 4.00 (d, J=5.7Hz, 2H), 3.89 (d, J=17.9Hz, 6H).
实施例95:化合物S97的合成Example 95: Synthesis of Compound S97
合成参照实施例90中的合成方法,只需更换相应的原料即可。The synthesis refers to the synthesis method in Example 90, and only needs to replace the corresponding raw materials.
1H NMR(500MHz,DMSO-d6)δ12.88(br,1H),9.60(s,1H),7.70(d,J=2.1Hz,1H),7.41-7.36(m,2H),7.08(t,J=4.3Hz,1H),3.89(d,J=17.9Hz,6H),3.47(td,J=6.2,4.4Hz,2H),2.62(t,J=6.2Hz,2H). 1 H NMR (500 MHz, DMSO-d 6 ) δ 12.88 (br, 1H), 9.60 (s, 1H), 7.70 (d, J=2.1 Hz, 1H), 7.41-7.36 (m, 2H), 7.08 ( t, J=4.3Hz, 1H), 3.89 (d, J=17.9Hz, 6H), 3.47 (td, J=6.2, 4.4Hz, 2H), 2.62 (t, J=6.2Hz, 2H).
实施例96:化合物对人源干扰素基因刺激蛋白蛋白结合力的测试Example 96: Test of compound binding ability to human interferon gene-stimulating protein
干扰素基因刺激蛋白竞争性结合测试实验的具体步骤根据CISBIO公司的试剂盒。基本的原理:特定的Anti 6His-Tb3+抗体作为给体,其与6His标记的h干扰素基因刺激蛋白蛋白结合,d2标记的干扰素基因刺激蛋白配体作为受体(CISBIO,catalogueno.64BDSTGPEG),化合物溶于DMSO中设置10个浓度点,与上述的配体和受体共孵育,检测待测化合物与体系中的配体竞争性结合干扰素基因刺激蛋白的IC50。The specific steps of the interferon gene-stimulated protein competitive binding test experiment are based on the kit from CISBIO. Basic principle: a specific Anti 6His-Tb 3+ antibody is used as a donor, which binds to the 6His-labeled h interferon gene-stimulating protein protein, and the d2-labeled interferon gene-stimulating protein ligand is used as a receptor (CISBIO, catalogue no. 64BDSTGPEG ), the compound was dissolved in DMSO to set 10 concentration points, incubated with the above-mentioned ligands and receptors, and the IC 50 of the compound to be tested that competed with the ligand in the system to bind to the interferon gene-stimulating protein was detected.
具体的实验操作步骤如下The specific experimental steps are as follows
①实验设置阴性对照孔、标准品孔、化合物孔,分别向阴性对照孔、标准品孔、化合物孔中加入5μL阴性对照品、不同浓度的标准品cGAMP和不同浓度的待测样品;① Set up negative control wells, standard wells, and compound wells in the experiment, and add 5 μL of negative control, standard cGAMP of different concentrations, and samples to be tested in different concentrations to the negative control wells, standard wells, and compound wells respectively;
②向阴性对照孔中加入5μL 1X detection buffer,向标准品和化合物孔中加入5μL 1X 6His标记的h干扰素基因刺激蛋白蛋白;②Add 5μL of 1X detection buffer to the negative control wells, and add 5μL of 1X 6His-labeled h-interferon gene-stimulating protein to the standard and compound wells;
③向所有的孔中加入10μL 1X干扰素基因刺激蛋白配体-d2和1X Anti 6His-Tb3+混合工作液;③Add 10μL of 1X IFN-gene-stimulating protein ligand-d2 and 1X Anti 6His-Tb 3+ mixed working solution to all wells;
④用膜将板封好,室温孵育4小时;④ Seal the plate with membrane and incubate at room temperature for 4 hours;
⑤撕掉膜,使用HTRF compatible reader分别在616nm和665nm处读取吸光度值。⑤Tear off the film, and use the HTRF compatible reader to read the absorbance values at 616nm and 665nm respectively.
⑥计算受体和供体发射信号的比值:Ratio=Signal 665nm/Signal 616nm X 104 ⑥ Calculate the ratio of the signal emitted by the acceptor and the donor: Ratio=Signal 665nm/Signal 616nm X 10 4
⑦利用Ratio值计算得出IC50值,再通过标准品cGAMP对化合物的IC50值进行换算。⑦ Calculate the IC 50 value using the Ratio value, and then convert the IC 50 value of the compound by the standard cGAMP.
实验结果如下表1所示,C:IC50>100nM;B:IC50=100nM-10nM;A:IC50<10nM;The experimental results are shown in Table 1 below, C: IC 50 >100 nM; B: IC 50 =100 nM-10 nM; A: IC 50 <10 nM;
表1待测化合物对人源干扰素基因刺激蛋白蛋白结合力Table 1 Binding ability of compounds to be tested to human interferon gene-stimulating protein
实施例97:IFNβ释放量的测试Example 97: Measurement of the amount of IFNβ released
实验操作步骤:将THP-1细胞铺板(1×105个/well),次日,将细胞换液后,不同浓度的待测化合物与THP-1细胞共孵育24h,随后收集培养基上清,按如下步骤进行ELISA检测IFN-β含量。Experimental operation steps: THP-1 cells were plated (1×10 5 cells/well), the next day, after the cells were changed, the compounds to be tested with different concentrations were incubated with THP-1 cells for 24 hours, and then the medium supernatant was collected , and the IFN-β content was detected by ELISA according to the following steps.
①标准品的加样:设置标准品孔和样本孔,标准品孔各加不同浓度(0、25、50、100、200、400pg/ml)的标准品50μL。①Standard addition: Set standard wells and sample wells, and add 50 μL of standard with different concentrations (0, 25, 50, 100, 200, 400pg/ml) to each standard well.
②加样:分别设空白孔(空白对照孔不加样品及酶标试剂,其余各步操作相同)、待测样品孔。在酶标包被板上待测样品孔中先加样品稀释液40μL,然后再加待测样品10μL(样品最终稀释度为5倍)。加样将样品加于酶标板孔底部,尽量不触及孔壁,轻轻晃动混匀。② Sample addition: set up blank wells (the blank control wells do not add samples and enzyme labeling reagents, and the operations in other steps are the same) and the wells for the samples to be tested. First add 40 μL of sample diluent to the well of the sample to be tested on the enzyme-labeled coated plate, and then add 10 μL of the sample to be tested (the final dilution of the sample is 5 times). Add the sample to the bottom of the well of the microtiter plate, try not to touch the wall of the well, and shake gently to mix.
③加酶:每孔加入酶标试剂100μL,空白孔除外。③ Add enzyme: Add 100 μL of enzyme labeling reagent to each well, except for blank wells.
④温育:用封板膜封板后置37℃温育60分钟。④ Incubation: Seal the plate with a sealing film and incubate at 37°C for 60 minutes.
⑤配液:将20倍浓缩洗涤液用蒸馏水20倍稀释后备用。⑤ Dosing: Dilute the 20-fold concentrated washing solution with distilled water 20-fold for later use.
⑥洗涤:小心揭掉封板膜,弃去液体,甩干,每孔加满洗涤液,静置30秒后弃去,如此重复5次,拍干。⑥Washing: Carefully peel off the sealing film, discard the liquid, spin dry, fill each well with washing solution, let it stand for 30 seconds, then discard, repeat 5 times, and pat dry.
⑦显色:每孔先加入显色剂A50μl,再加入显色剂B50μl,轻轻震荡混匀,37℃避光显色15分钟.⑦ Color development: first add 50 μl of color developer A to each well, then add 50 μl of color developer B, gently shake and mix, and develop color at 37°C for 15 minutes in the dark.
⑧终止:每孔加终止液50μL,终止反应(此时蓝色立转黄色)。⑧ Stop: Add 50 μL of stop solution to each well to stop the reaction (the blue turns to yellow at this time).
⑨测定:以空白孔调零,450nm波长依序测量各孔的吸光度(OD值)。测定应在加终止液后15分钟以内进行。⑨Determination: Zero the blank well, and measure the absorbance (OD value) of each well in sequence at a wavelength of 450 nm. The measurement should be carried out within 15 minutes after adding the stop solution.
⑩计算:通过标准品得出标准曲线后计算各样品中IFN-β含量,根据不同浓度下待测化合物诱导产生的IFN-β含量计算EC50值。⑩Calculation: Calculate the IFN-β content in each sample after the standard curve is obtained from the standard substance, and calculate the EC 50 value according to the IFN-β content induced by the test compound at different concentrations.
干扰素基因刺激蛋白蛋白激活后,诱导IFNβ的表达从而发挥抗肿瘤或抗感染的效果。IFNβ的生成量是干扰素基因刺激蛋白蛋白激活程度的重要指标。本实验采用ELISA的方法探亢了不同浓度的待测化合物与THP-1细胞共孵育后IFNβ的生成量的情况,结果如下表2所示。C:EC50>10μM;B:EC50=10μM-1μM;A:EC50<1μM。After the activation of the interferon gene-stimulating protein protein, the expression of IFNβ is induced to exert anti-tumor or anti-infection effects. The production of IFNβ is an important indicator of the activation of interferon gene-stimulated protein. In this experiment, the method of ELISA was used to detect the production of IFNβ after co-incubation of different concentrations of the compounds to be tested with THP-1 cells. The results are shown in Table 2 below. C: EC 50 >10 μM; B: EC 50 =10 μM-1 μM; A: EC 50 <1 μM.
表2不同浓度的待测化合物与THP-1细胞共孵育后IFNβ的生成量的情况Table 2 The production of IFNβ after co-incubation of test compounds with different concentrations of THP-1 cells
Claims (13)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110117453.2A CN112898286A (en) | 2021-01-28 | 2021-01-28 | Benzothiophene compound or pharmaceutically acceptable salt and isomer thereof, and preparation method, pharmaceutical composition and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110117453.2A CN112898286A (en) | 2021-01-28 | 2021-01-28 | Benzothiophene compound or pharmaceutically acceptable salt and isomer thereof, and preparation method, pharmaceutical composition and application thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN112898286A true CN112898286A (en) | 2021-06-04 |
Family
ID=76119541
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110117453.2A Pending CN112898286A (en) | 2021-01-28 | 2021-01-28 | Benzothiophene compound or pharmaceutically acceptable salt and isomer thereof, and preparation method, pharmaceutical composition and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN112898286A (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022002077A1 (en) * | 2020-06-30 | 2022-01-06 | 上海海和药物研究开发股份有限公司 | Aryl aromatic heterocyclic derivative and preparation method therefor and use thereof |
| US11939343B2 (en) | 2019-08-02 | 2024-03-26 | Mersana Therapeutics, Inc. | Sting agonist compounds and methods of use |
| US12156870B2 (en) | 2020-04-02 | 2024-12-03 | Mersana Therapeutics, Inc. | Antibody drug conjugates comprising sting agonists |
| CN119143740A (en) * | 2024-08-15 | 2024-12-17 | 杭州科兴生物化工有限公司 | Benzothiophene derivative and preparation method and application thereof |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001070675A2 (en) * | 2000-03-24 | 2001-09-27 | Methylgene, Inc. | Inhibitors of histone deacetylase |
| US20040006011A1 (en) * | 1996-07-12 | 2004-01-08 | Gour Barbara J. | Peptidomimetic modulators of cell adhesion |
| WO2005039548A2 (en) * | 2003-10-15 | 2005-05-06 | Probiodrug Ag | Use of effectors of glutaminyl and glutamate cyclases |
| CN101837127A (en) * | 2003-05-05 | 2010-09-22 | 前体生物药物股份公司 | The application of glutaminyl and glutamate cyclase effector |
| WO2020059660A1 (en) * | 2018-09-20 | 2020-03-26 | 日清食品ホールディングス株式会社 | Compound having enac activation potency |
| CN111393404A (en) * | 2019-01-02 | 2020-07-10 | 中国科学院上海药物研究所 | Benzothiophene compounds, and pharmaceutical composition and application thereof |
-
2021
- 2021-01-28 CN CN202110117453.2A patent/CN112898286A/en active Pending
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040006011A1 (en) * | 1996-07-12 | 2004-01-08 | Gour Barbara J. | Peptidomimetic modulators of cell adhesion |
| WO2001070675A2 (en) * | 2000-03-24 | 2001-09-27 | Methylgene, Inc. | Inhibitors of histone deacetylase |
| CN101837127A (en) * | 2003-05-05 | 2010-09-22 | 前体生物药物股份公司 | The application of glutaminyl and glutamate cyclase effector |
| WO2005039548A2 (en) * | 2003-10-15 | 2005-05-06 | Probiodrug Ag | Use of effectors of glutaminyl and glutamate cyclases |
| WO2020059660A1 (en) * | 2018-09-20 | 2020-03-26 | 日清食品ホールディングス株式会社 | Compound having enac activation potency |
| CN111393404A (en) * | 2019-01-02 | 2020-07-10 | 中国科学院上海药物研究所 | Benzothiophene compounds, and pharmaceutical composition and application thereof |
Non-Patent Citations (3)
| Title |
|---|
| AGNIESZKA TAFELSKA-KACZMAREK 等: "Highly Asymmetric Reduction of New Benzofuryl and Benzothiophenyl α-Amino Ketones", 《INTERNATIONAL ELECTRONIC CONFERENCE ON SYNTHETIC ORGANIC CHEMISTRY》 * |
| DMITRY A. ANDROSOV 等: "A convenient approach towards 2- and 3-aminobenzo[b]thiophenes", 《TETRAHEDRON》 * |
| NOBUYUKI MATSUNAGA 等: "C17,20-Lyase inhibitors I. Structure-based de novo design and SAR study of C17,20-lyase inhibitors", 《BIOORGANIC & MEDICINAL CHEMISTRY》 * |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11939343B2 (en) | 2019-08-02 | 2024-03-26 | Mersana Therapeutics, Inc. | Sting agonist compounds and methods of use |
| US12156870B2 (en) | 2020-04-02 | 2024-12-03 | Mersana Therapeutics, Inc. | Antibody drug conjugates comprising sting agonists |
| US12440576B2 (en) | 2020-04-02 | 2025-10-14 | Mersana Therapeutics, Inc. | Antibody drug conjugates comprising sting agonists |
| WO2022002077A1 (en) * | 2020-06-30 | 2022-01-06 | 上海海和药物研究开发股份有限公司 | Aryl aromatic heterocyclic derivative and preparation method therefor and use thereof |
| CN119143740A (en) * | 2024-08-15 | 2024-12-17 | 杭州科兴生物化工有限公司 | Benzothiophene derivative and preparation method and application thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN112898286A (en) | Benzothiophene compound or pharmaceutically acceptable salt and isomer thereof, and preparation method, pharmaceutical composition and application thereof | |
| CN1251097B (en) | Phthalazines with angiogenesis inhibiting activity | |
| CN109721527B (en) | Novel anti-PD-L1 compound, application thereof and composition containing same | |
| CN112424185B (en) | Compound containing benzene ring, preparation method and application thereof | |
| WO2019007418A1 (en) | Fxr receptor agonist | |
| CN110092745B (en) | Compound containing aromatic ring and application thereof | |
| JPH06507625A (en) | Substituted pyrimidinones with acid functionality as angiotensin II antagonists | |
| WO2011113366A1 (en) | Method for preparing deuterated diphenylurea | |
| CN110092779B (en) | Substituted phenyl compound and application thereof | |
| MX2015002310A (en) | Novel phenyl-pyridine/pyrazine amides for the treatment of cancer. | |
| AU2009217541A1 (en) | Tetrahydrothieno pyridines | |
| MX2007007830A (en) | Pyridine compounds for the treatment of prostaglandin mediated diseases. | |
| CN117881677A (en) | Fused isoxazolyl compounds as KAT6Aw inhibitors | |
| WO2025167663A1 (en) | Pyridazine compound and use thereof | |
| CN108863850A (en) | Biaryl base class compound and its preparation method and application | |
| CN114502532A (en) | Sulfo-substituted biaryl compound or salt thereof, and preparation method and application thereof | |
| WO2023125896A1 (en) | Glp-1 agonist intermediate, preparation method therefor, and use thereof in medicine | |
| WO2024188107A1 (en) | Indoline compound containing thiazole structure, preparation method therefor, and application thereof | |
| CN110092740A (en) | A kind of fused ring compound and its application | |
| CN113024563A (en) | Pyrimido five-membered heterocyclic compound or pharmaceutically acceptable salt and isomer thereof, and preparation method, pharmaceutical composition and application thereof | |
| CN113788827B (en) | Phthalazinone compound, preparation method and medical application thereof | |
| CN111635373B (en) | Polycyclic sulfonamide ROR gamma modulators | |
| CN120247920A (en) | A PARP/CK2α dual-target kinase inhibitor small molecule and its pharmaceutical composition and application | |
| WO2021129841A1 (en) | Compound used as ret kinase inhibitor and application thereof | |
| JP2022516922A (en) | Fluorine-containing substituted benzothiophene compounds and their pharmaceutical compositions and applications |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| TA01 | Transfer of patent application right | ||
| TA01 | Transfer of patent application right |
Effective date of registration: 20210721 Address after: 100036 Beijing city Haidian District Cuiwei Road No. 2, building A, room 1209, static source in Applicant after: Beijing Kexiang Zhongsheng Pharmaceutical Technology Co.,Ltd. Address before: No. 639 Jiangning longmian Road District of Nanjing City, Jiangsu province 211198 Applicant before: CHINA PHARMACEUTICAL University |
|
| TA01 | Transfer of patent application right | ||
| TA01 | Transfer of patent application right |
Effective date of registration: 20210810 Address after: 224500 4th floor, North building, building 2, enterprise accelerator, Xin'an Avenue, Binhai Pharmaceutical Industrial Park, Binhai County, Yancheng City, Jiangsu Province Applicant after: Yaokang Zhongtuo (Jiangsu) Pharmaceutical Technology Co.,Ltd. Address before: 100036 Beijing city Haidian District Cuiwei Road No. 2, building A, room 1209, static source in Applicant before: Beijing Kexiang Zhongsheng Pharmaceutical Technology Co.,Ltd. |
|
| WD01 | Invention patent application deemed withdrawn after publication | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20210604 |