CN112871462A - Beneficiation process method for refined quality-based recycling of lead-zinc sulfide ore wastewater - Google Patents
Beneficiation process method for refined quality-based recycling of lead-zinc sulfide ore wastewater Download PDFInfo
- Publication number
- CN112871462A CN112871462A CN202110045194.7A CN202110045194A CN112871462A CN 112871462 A CN112871462 A CN 112871462A CN 202110045194 A CN202110045194 A CN 202110045194A CN 112871462 A CN112871462 A CN 112871462A
- Authority
- CN
- China
- Prior art keywords
- wastewater
- zinc
- lead
- concentrate
- ore
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 239000002351 wastewater Substances 0.000 title claims abstract description 173
- 238000000034 method Methods 0.000 title claims abstract description 66
- 238000004064 recycling Methods 0.000 title claims abstract description 42
- JQJCSZOEVBFDKO-UHFFFAOYSA-N lead zinc Chemical compound [Zn].[Pb] JQJCSZOEVBFDKO-UHFFFAOYSA-N 0.000 title claims abstract description 38
- 239000005083 Zinc sulfide Substances 0.000 title claims abstract description 25
- 229910052984 zinc sulfide Inorganic materials 0.000 title claims abstract description 25
- 239000012141 concentrate Substances 0.000 claims abstract description 87
- 239000011701 zinc Substances 0.000 claims abstract description 87
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 claims abstract description 83
- 229910052725 zinc Inorganic materials 0.000 claims abstract description 83
- 229910052717 sulfur Inorganic materials 0.000 claims abstract description 71
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims abstract description 70
- 239000011593 sulfur Substances 0.000 claims abstract description 70
- 238000000926 separation method Methods 0.000 claims abstract description 55
- 229910052748 manganese Inorganic materials 0.000 claims abstract description 43
- 239000011572 manganese Substances 0.000 claims abstract description 43
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 claims abstract description 42
- 238000000227 grinding Methods 0.000 claims abstract description 23
- 229910052500 inorganic mineral Inorganic materials 0.000 claims abstract description 22
- 239000011707 mineral Substances 0.000 claims abstract description 22
- 238000001556 precipitation Methods 0.000 claims abstract description 9
- 238000005189 flocculation Methods 0.000 claims abstract description 7
- 230000016615 flocculation Effects 0.000 claims abstract description 7
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 24
- 239000003814 drug Substances 0.000 claims description 17
- TUZCOAQWCRRVIP-UHFFFAOYSA-N butoxymethanedithioic acid Chemical compound CCCCOC(S)=S TUZCOAQWCRRVIP-UHFFFAOYSA-N 0.000 claims description 16
- 239000003795 chemical substances by application Substances 0.000 claims description 14
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 claims description 12
- 235000008733 Citrus aurantifolia Nutrition 0.000 claims description 10
- 235000011941 Tilia x europaea Nutrition 0.000 claims description 10
- 239000004571 lime Substances 0.000 claims description 10
- 239000004088 foaming agent Substances 0.000 claims description 9
- 238000001914 filtration Methods 0.000 claims description 8
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 claims description 7
- 229920002401 polyacrylamide Polymers 0.000 claims description 7
- 229910000365 copper sulfate Inorganic materials 0.000 claims description 6
- ARUVKPQLZAKDPS-UHFFFAOYSA-L copper(II) sulfate Chemical compound [Cu+2].[O-][S+2]([O-])([O-])[O-] ARUVKPQLZAKDPS-UHFFFAOYSA-L 0.000 claims description 6
- 238000007670 refining Methods 0.000 claims description 6
- 238000003756 stirring Methods 0.000 claims description 6
- NWONKYPBYAMBJT-UHFFFAOYSA-L zinc sulfate Chemical compound [Zn+2].[O-]S([O-])(=O)=O NWONKYPBYAMBJT-UHFFFAOYSA-L 0.000 claims description 6
- 229910000368 zinc sulfate Inorganic materials 0.000 claims description 6
- 229960001763 zinc sulfate Drugs 0.000 claims description 6
- FWLHAQYOFMQTHQ-UHFFFAOYSA-N 2-N-[8-[[8-(4-aminoanilino)-10-phenylphenazin-10-ium-2-yl]amino]-10-phenylphenazin-10-ium-2-yl]-8-N,10-diphenylphenazin-10-ium-2,8-diamine hydroxy-oxido-dioxochromium Chemical compound O[Cr]([O-])(=O)=O.O[Cr]([O-])(=O)=O.O[Cr]([O-])(=O)=O.Nc1ccc(Nc2ccc3nc4ccc(Nc5ccc6nc7ccc(Nc8ccc9nc%10ccc(Nc%11ccccc%11)cc%10[n+](-c%10ccccc%10)c9c8)cc7[n+](-c7ccccc7)c6c5)cc4[n+](-c4ccccc4)c3c2)cc1 FWLHAQYOFMQTHQ-UHFFFAOYSA-N 0.000 claims description 5
- 238000000498 ball milling Methods 0.000 claims description 5
- 239000013505 freshwater Substances 0.000 claims description 5
- 229920000767 polyaniline Polymers 0.000 claims description 5
- 238000011049 filling Methods 0.000 claims description 3
- 238000007885 magnetic separation Methods 0.000 claims description 3
- 238000005065 mining Methods 0.000 claims description 3
- 229910052683 pyrite Inorganic materials 0.000 claims description 3
- NIFIFKQPDTWWGU-UHFFFAOYSA-N pyrite Chemical compound [Fe+2].[S-][S-] NIFIFKQPDTWWGU-UHFFFAOYSA-N 0.000 claims description 3
- 239000011028 pyrite Substances 0.000 claims description 3
- HQABUPZFAYXKJW-UHFFFAOYSA-O butylazanium Chemical compound CCCC[NH3+] HQABUPZFAYXKJW-UHFFFAOYSA-O 0.000 claims 1
- 229940079593 drug Drugs 0.000 claims 1
- ZOOODBUHSVUZEM-UHFFFAOYSA-N ethoxymethanedithioic acid Chemical compound CCOC(S)=S ZOOODBUHSVUZEM-UHFFFAOYSA-N 0.000 claims 1
- 239000012991 xanthate Substances 0.000 claims 1
- 238000011084 recovery Methods 0.000 abstract description 13
- 229910052709 silver Inorganic materials 0.000 abstract description 9
- 239000004332 silver Substances 0.000 abstract description 9
- 230000018044 dehydration Effects 0.000 abstract description 4
- 238000006297 dehydration reaction Methods 0.000 abstract description 4
- 239000011133 lead Substances 0.000 description 67
- 238000004065 wastewater treatment Methods 0.000 description 13
- 238000005188 flotation Methods 0.000 description 10
- 239000003153 chemical reaction reagent Substances 0.000 description 8
- GEHJYWRUCIMESM-UHFFFAOYSA-L sodium sulfite Chemical compound [Na+].[Na+].[O-]S([O-])=O GEHJYWRUCIMESM-UHFFFAOYSA-L 0.000 description 8
- 231100000989 no adverse effect Toxicity 0.000 description 7
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 6
- 239000010665 pine oil Substances 0.000 description 6
- 238000004519 manufacturing process Methods 0.000 description 5
- 239000000843 powder Substances 0.000 description 5
- 239000010865 sewage Substances 0.000 description 5
- 239000012452 mother liquor Substances 0.000 description 4
- 235000010265 sodium sulphite Nutrition 0.000 description 4
- PTFCDOFLOPIGGS-UHFFFAOYSA-N Zinc dication Chemical compound [Zn+2] PTFCDOFLOPIGGS-UHFFFAOYSA-N 0.000 description 3
- 230000007613 environmental effect Effects 0.000 description 3
- 239000010419 fine particle Substances 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- 238000005456 ore beneficiation Methods 0.000 description 3
- JPVYNHNXODAKFH-UHFFFAOYSA-N Cu2+ Chemical compound [Cu+2] JPVYNHNXODAKFH-UHFFFAOYSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 2
- 229910001431 copper ion Inorganic materials 0.000 description 2
- 230000005611 electricity Effects 0.000 description 2
- 238000003912 environmental pollution Methods 0.000 description 2
- 150000002500 ions Chemical class 0.000 description 2
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- CWYNVVGOOAEACU-UHFFFAOYSA-N Fe2+ Chemical compound [Fe+2] CWYNVVGOOAEACU-UHFFFAOYSA-N 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 239000004568 cement Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 238000005265 energy consumption Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 229910001385 heavy metal Inorganic materials 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 229910052976 metal sulfide Inorganic materials 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 230000001376 precipitating effect Effects 0.000 description 1
- 238000003672 processing method Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B03—SEPARATION OF SOLID MATERIALS USING LIQUIDS OR USING PNEUMATIC TABLES OR JIGS; MAGNETIC OR ELECTROSTATIC SEPARATION OF SOLID MATERIALS FROM SOLID MATERIALS OR FLUIDS; SEPARATION BY HIGH-VOLTAGE ELECTRIC FIELDS
- B03D—FLOTATION; DIFFERENTIAL SEDIMENTATION
- B03D1/00—Flotation
- B03D1/02—Froth-flotation processes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B03—SEPARATION OF SOLID MATERIALS USING LIQUIDS OR USING PNEUMATIC TABLES OR JIGS; MAGNETIC OR ELECTROSTATIC SEPARATION OF SOLID MATERIALS FROM SOLID MATERIALS OR FLUIDS; SEPARATION BY HIGH-VOLTAGE ELECTRIC FIELDS
- B03B—SEPARATING SOLID MATERIALS USING LIQUIDS OR USING PNEUMATIC TABLES OR JIGS
- B03B9/00—General arrangement of separating plant, e.g. flow sheets
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B03—SEPARATION OF SOLID MATERIALS USING LIQUIDS OR USING PNEUMATIC TABLES OR JIGS; MAGNETIC OR ELECTROSTATIC SEPARATION OF SOLID MATERIALS FROM SOLID MATERIALS OR FLUIDS; SEPARATION BY HIGH-VOLTAGE ELECTRIC FIELDS
- B03D—FLOTATION; DIFFERENTIAL SEDIMENTATION
- B03D1/00—Flotation
- B03D1/08—Subsequent treatment of concentrated product
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B03—SEPARATION OF SOLID MATERIALS USING LIQUIDS OR USING PNEUMATIC TABLES OR JIGS; MAGNETIC OR ELECTROSTATIC SEPARATION OF SOLID MATERIALS FROM SOLID MATERIALS OR FLUIDS; SEPARATION BY HIGH-VOLTAGE ELECTRIC FIELDS
- B03D—FLOTATION; DIFFERENTIAL SEDIMENTATION
- B03D1/00—Flotation
- B03D1/12—Agent recovery
Landscapes
- Chemical & Material Sciences (AREA)
- Dispersion Chemistry (AREA)
- Manufacture And Refinement Of Metals (AREA)
Abstract
The invention discloses a mineral processing technique for refined grading and recycling lead-zinc sulfide ore wastewater, wherein lead-zinc sulfide ore is subjected to ore grinding and grading, raw ore concentration and dehydration, lead (silver) and zinc selection, zinc tail concentration and sulfur selection, and finally tailings are precipitated, dehydrated, filled and the like; the beneficiation wastewater generated by beneficiation comprises: raw ore concentrated wastewater, zinc tail concentrated wastewater, lead concentrate wastewater, zinc concentrate wastewater, sulfur concentrate wastewater, manganese concentrate wastewater, tailing wastewater and the like; directly using the raw ore concentrated wastewater and the lead concentrate wastewater for ore grinding classification and lead separation; the zinc tail concentrated wastewater and the zinc concentrate wastewater are directly used for zinc separation; directly using the sulfur concentrate wastewater, the manganese concentrate wastewater and the tailing wastewater for sulfur and manganese separation; and all the waste water which is directly recycled and discharged is recycled for ore grinding classification, lead separation and the like after flocculation precipitation treatment, so that fine ore dressing of recycling waste water according to quality is realized. The process method has high main grade and recovery rate of ore dressing and low ore dressing cost, and realizes the complete reuse and zero discharge of the ore dressing wastewater.
Description
Technical Field
The invention relates to a beneficiation wastewater recycling process method, in particular to a beneficiation process method for refined quality-based recycling of lead-zinc sulfide ore wastewater.
Background
Lead and zinc resources are important mineral resources in China, nearly thousands of existing lead and zinc colored mines mainly comprise lead-zinc-silver polymetallic sulfide ores, and valuable elements such as sulfur, iron, manganese and the like are often associated. The lead-zinc sulfide ore beneficiation process is complex, the types of the wastewater are various, the components are mixed, the influence of the wastewater on beneficiation indexes is large, the treatment difficulty is large, the cost is high, the combination of the wastewater treatment and recycling and the beneficiation process is not tight, and the utilization degree of useful medicament components in the wastewater is low; meanwhile, because the lead-zinc ore beneficiation wastewater belongs to heavy metal-related wastewater and has the risk of environmental pollution, how to tightly combine the lead-zinc beneficiation wastewater treatment and recycling process with the beneficiation process, realize the low-cost and high-efficiency comprehensive utilization of the beneficiation wastewater, eliminate the environmental risk, and is particularly important for enterprises and countries.
In a lead-zinc nonferrous metal mine, the traditional mineral separation process for lead-zinc flotation in China at present comprises ore grinding, classification, lead flotation, zinc flotation, sulfur flotation, manganese flotation and the like, and the waste water generated by lead-zinc sulfide ore separation comprises the following steps: lead concentrate wastewater, zinc concentrate wastewater, sulfur concentrate wastewater, manganese concentrate wastewater, tailing wastewater and the like; the traditional beneficiation process method has the problems that the beneficiation recovery rate of lead-zinc-silver-sulfur-manganese ore and the grade of concentrate are difficult to further improve due to low concentration of the selected ore pulp, the water and electricity consumption is high, and the like; meanwhile, because the traditional beneficiation process and a mixed centralized treatment method are often adopted for wastewater treatment, the problems of large beneficiation wastewater treatment capacity, high treatment cost, large treatment difficulty and large influence of wastewater recycling on lead, zinc and sulfur indexes exist, and useful medicament components in the wastewater are difficult to be well utilized, so that the wastewater is difficult to be fully utilized, and the risk of environmental pollution exists.
Disclosure of Invention
The invention provides a beneficiation process method for finely recycling the wastewater according to the quality, which has the advantages of high beneficiation index, complete recycling of beneficiation wastewater, no adverse effect of the wastewater on the index, low consumption of water and electricity medicaments and small treatment capacity of the tail end wastewater, and solves the problems of low concentration of selected ore pulp, large influence of the beneficiation wastewater on the beneficiation index, high cost and difficulty in centralized treatment of the wastewater and environmental safety risk.
The invention provides the following technical scheme: a mineral processing process method for refining, grading and recycling lead-zinc sulfide ore wastewater comprises the following steps:
after ball milling and grading of lead-zinc raw ore, the mass concentration of the graded overflow of a swirler is 25-30%, the fineness of minus 200 meshes accounts for 75-80%, the raw ore is concentrated and dehydrated to 50-52% of mass concentration by a raw ore concentrator, a stirring barrel is used before lead separation, lead separation agents zinc sulfate 400-; the lead recovery rate is not lower than 91 percent, and the silver recovery rate in lead is not lower than 62 percent; concentrating, filtering and dehydrating the lead concentrate pulp to obtain final lead concentrate ore powder and lead concentrate wastewater; and the raw ore grading overflow wastewater and the lead concentrate wastewater are all returned to the ore grinding and grading and lead dressing for use.
The mass concentration of the lead tailings is 42-45%,
adding 350-450g/t of copper sulfate and 2500g/t of lime into the lead tailings to inhibit pyrite, adding 180-220g/t of butyl xanthate to collect zinc and 10-20g/t of foaming agent to obtain zinc concentrate; the main grade of zinc is not lower than 50 percent, and the recovery rate of zinc is not lower than 91.8 percent; concentrating, filtering and dehydrating the zinc concentrate to obtain final zinc concentrate and generate zinc concentrate wastewater; concentrating and dehydrating the zinc tailings until the mass concentration is 50-52% and before sulfur separation, stirring the zinc tailings in a stirring barrel, returning zinc concentrate wastewater and zinc tailing concentrated wastewater to zinc separation preferentially, and treating the surplus in a sewage treatment station.
Adding sulfuric acid (98%) into a stirring barrel before sulfur separation to adjust the pH value to about 7, and adding 600-800g/t of butyl xanthate for collecting sulfur (pyrite) to obtain sulfur concentrate and tailings; the main grade of sulfur is not lower than 46 percent, and the recovery rate of sulfur is not lower than 82 percent; concentrating, filtering and dehydrating the sulfur concentrate to obtain final sulfur concentrate and generate sulfur concentrate wastewater; performing high-gradient manganese separation on the sulfur separation tailings, and concentrating, filtering and dehydrating the manganese concentrate to obtain final manganese concentrate and generate manganese concentrate wastewater; and precipitating and dehydrating the tailings to obtain final tailings, adding cement into the tailings for filling and generating tailing wastewater. The sulfur concentrate wastewater, the manganese concentrate wastewater and the tailing wastewater are preferentially used for sulfur and manganese separation, and the redundant part is treated by a sewage treatment station.
As a further improvement of the beneficiation process method for refined grading and recycling of the lead-zinc sulfide ore wastewater, the raw ore concentrated wastewater and the lead concentrate wastewater are returned to the ball milling and grading and lead separation for use.
As a further improvement of the beneficiation process method for refined quality-based recycling of the lead-zinc sulfide ore wastewater, the zinc tail concentrated wastewater and the zinc concentrate wastewater are returned to be used for zinc beneficiation.
As a further improvement of the beneficiation process for refined quality-based recycling of the lead-zinc sulfide ore wastewater, the tailing concentrated wastewater, the sulfur concentrate wastewater and the manganese concentrate wastewater are returned together for sulfur and manganese separation.
As a further improvement of the beneficiation process for refined grading and recycling of the lead-zinc sulfide ore wastewater, zinc tail wastewater, zinc concentrate wastewater, sulfur concentrate wastewater, manganese concentrate wastewater, tailing wastewater and the like which are produced by zinc separation, sulfur separation and manganese separation are subjected to flocculation precipitation treatment in a sewage treatment station and then returned to the ore grinding and grading, lead separation and the like for use.
As a further improvement of the beneficiation process for refined quality-based recycling of the lead-zinc sulfide ore wastewater, because the wastewater is recycled according to quality, the respective wastewater returns to respective operation, the beneficiation reagents for each operation of lead selection, zinc selection and sulfur selection can be properly reduced in dosage, and the index can be further improved.
As a further improvement of the beneficiation process method for fine quality-based recycling of the lead-zinc sulfide ore wastewater, the beneficiation wastewater entering the sewage treatment station only needs to add polyaluminium chloride and polyacrylamide to precipitate and remove copper, lead and zinc ions and fine-particle minerals in the wastewater, and has no adverse effect on lead (silver) flotation indexes.
According to the beneficiation process method for refining, grading and recycling the lead-zinc sulfide ore wastewater, the mass concentration of lead roughing flotation is stably controlled to be 50-52%, and the mass concentration of lead tailings is stably controlled to be 42-45%. The mass concentration of lead roughing and lead tail is 15 percent higher than that of the conventional flotation process flow without raw ore concentration.
The raw ore concentrated wastewater and the lead concentrate wastewater are all directly returned to ore grinding classification and lead separation for use as mother liquor, have no adverse effect on lead (silver) separation indexes, and can save lead separation agents.
The zinc tail concentrated wastewater and the zinc concentrate wastewater are used as mother liquor to return to zinc selection preferentially, have no adverse effect on zinc finger selection, and can save zinc selection agents.
The tailing precipitation wastewater, the sulfur concentrate wastewater and the manganese concentrate wastewater are used as mother liquor to return to sulfur separation preferentially and directly, no adverse effect is caused on sulfur separation indexes, and sulfur separation agents can be saved.
According to the beneficiation wastewater entering the sewage treatment station, the polyaluminium chloride and the polyacrylamide are added, so that copper ions, lead ions, zinc ions, fine particle minerals and the like in the wastewater are precipitated and removed, collecting agents, foaming agents and the like in water are reserved, the copper ions, the lead ions, the zinc ions, the fine particle minerals and the like are reused for grinding classification, lead separation and the like, no adverse effect is caused on grinding classification and lead separation indexes, and lead (silver) separation agents can be saved.
The beneficiation process method for refined grading recycling of the lead-zinc sulfide ore wastewater has the advantages of high lead-zinc-sulfur concentration, long flotation time, small reagent dosage, small wastewater treatment capacity, low beneficiation cost, high beneficiation index, complete recycling of beneficiation wastewater and elimination of environmental risks.
According to the beneficiation process method for refined quality-based recycling of the lead-zinc sulfide ore wastewater, the beneficiation reagent and the water treatment reagent are common reagents, most of wastewater is directly recycled as mother liquor, so that the reagents in the water are recycled, fresh water resources and reagent consumption are effectively saved, the beneficiation process and the wastewater treatment and recycling method are effectively combined and reasonably matched, and refined beneficiation is realized.
Drawings
FIG. 1 is a flow chart of a conventional lead-zinc polymetallic ore beneficiation and wastewater centralized treatment recycling process;
FIG. 2 is a flow chart of a process for the concentrated beneficiation of lead-zinc polymetallic ores and the concentrated treatment and recycling of wastewater;
FIG. 3 is a flow chart of a lead-zinc multi-metal high-concentration beneficiation and wastewater centralized treatment recycling process;
FIG. 4 is a flow chart of the mineral processing process for refining, grading and recycling the lead-zinc sulfide ore wastewater.
In the figure, C represents mass concentration.
Detailed Description
The invention is further described by way of example with reference to the accompanying drawings.
Example 1
The properties of the ore of example 1 are: the raw ore grade of the lead-zinc sulfide ore comprises 3.0 percent of Pb3, 5.4 percent of Zns, 125g/t of Ag, 27 percent of S, 3.8 percent of manganese, and the oxidation rate of the ore is as follows: pb5.52% and Zn4.85%. The beneficiation process flow is shown in figure 1, raw ores are ground and classified, then respective beneficiation reagents are added to remove lead, zinc and sulfur, and sulfur tails are subjected to high-gradient magnetic separation of manganese; all the mineral processing wastewater is treated in a centralized way and then is completely reused for each operation.
The technical process conditions of the example 1 are as follows:
selecting lead: the grinding fineness is 75 percent when the granularity is-200 meshes (-0.074 mm); the initial concentration of lead dressing roughing is 35 percent; the lead-selecting medicament conditions are as follows: aniline black powder: butyl xanthate: buthanium nigrum ═ 8: 1: 1, 90g/t, zinc sulfate 1000g/t, sodium sulfite 700g/t, lime 2000g/t (pH of ore pulp 9.0);
selecting zinc: selecting zinc, wherein the initial concentration of the zinc roughing is 27%, the copper sulfate is 500g/t, the lime is 6000g/t, the butyl xanthate is 300g/t, and the foaming agent (pine oil) is 50 g/t;
selecting sulfur: selecting sulfur, wherein the initial concentration of the sulfur coarse selection is 20%, the sulfuric acid is 12000g/t, the butyl xanthate is 1200g/t, and the foaming agent (pine oil) is 20 g/t;
selecting manganese: the magnetic field intensity is 1.0T;
the beneficiation wastewater is centralized and reused, and the water treatment agent is as follows: 1750g/t of polyaluminium chloride and 50g/t of polyacrylamide, and the production test results are shown in Table 1.
Table 1: example 1 test index
Mineral processing wastewater treatment water quality (mg/L)
| Item | PH | COD | Cu | Pb | Zn | SS |
| Before treatment | 7.83 | 360 | 0.13 | 0.66 | 1.19 | 294 |
| After treatment | 7.50 | 335 | 0.01 | 0.05 | 0.30 | 15 |
Example 2
Example 2 the ore properties and mineral composition were the same as in example 1. The mineral processing process flow is shown in figure 2, after grinding and grading raw ores, respective mineral processing agents are added to remove lead and zinc, zinc tailings are concentrated to remove high-alkali water, sulfur is selected again, and sulfur tailings are subjected to high-gradient magnetic separation of manganese; all the mineral processing wastewater is treated in a centralized way and then is completely reused for each operation.
The technical process conditions of the embodiment 2 are as follows: selecting lead: the grinding fineness is 75 percent when the granularity is-200 meshes (-0.074 mm); the initial concentration of lead dressing roughing is 35 percent; the lead-selecting medicament conditions are as follows: aniline black powder: butyl xanthate: buthanium nigrum ═ 8: 1: 1, 90g/t, zinc sulfate 1000g/t, sodium sulfite 700g/t, lime 2000g/t (pH of ore pulp 9.0);
selecting zinc: selecting zinc, wherein the initial concentration of the zinc roughing is 27%, the copper sulfate is 500g/t, the lime is 5000g/t, the butyl xanthate is 300g/t, and the foaming agent (pine oil) is 50 g/t;
selecting sulfur: selecting sulfur, wherein the initial concentration of the sulfur coarse selection is 50%, the sulfuric acid is 6000g/t, the butyl xanthate is 800g/t, and the foaming agent (pine oil) is 10 g/t;
selecting manganese: the magnetic field intensity is 1.0T;
the beneficiation wastewater is centralized and reused, and the water treatment agent is as follows: 1750g/t of polyaluminium chloride, 50g/t of polyacrylamide,
the results of the production tests are shown in Table 2.
Table 2: example 2 test index
Mineral processing wastewater treatment water quality (mg/L)
| Item | PH | COD | Cu | Pb | Zn | SS |
| Before treatment | 7.53 | 350 | 0.12 | 0.68 | 1.20 | 295 |
| After treatment | 6.85 | 330 | 0.01 | 0.05 | 0.30 | 15 |
Example 3:
example 3 the ore properties and mineral composition were the same as in example 1. The beneficiation process flow is shown in figure 3. The technical process conditions of the embodiment 3 are as follows:
carrying out grading overflow concentration on raw ores, and then removing lead and zinc; concentrating the zinc-selecting tailings, then selecting sulfur and manganese, and recycling the beneficiation wastewater after centralized treatment for each operation of beneficiation.
The method specifically comprises the following steps: after ball milling and grading of lead-zinc raw ore, controlling the mass concentration of the grading overflow of the cyclone to be 30% and the fineness of 80% with the fineness of-200 meshes (74 microns), concentrating and dehydrating the grading overflow until the mass concentration is 50% to remove lead, wherein the initial concentration of lead-selecting roughing is 50%; the lead-selecting medicament conditions are as follows: aniline black powder: butyl xanthate: buthanium nigrum ═ 8: 1: 1, 70g/t, zinc sulfate 600g/t, sodium sulfite 400g/t, lime 1200g/t (pH of ore pulp 9.0);
selecting zinc: the initial concentration of the selected zinc roughing is 42 percent, the copper sulfate is 450g/t, the lime is 3000g/t, the butyl xanthate is 250g/t, and the foaming agent (the pine oil) is 30 g/t.
Selecting sulfur: selecting sulfur, roughing and selecting the raw materials with the initial concentration of 50 percent, 6000g/t sulfuric acid and 800g/t butyl xanthate;
selecting manganese: the magnetic field intensity is 1.0T;
the beneficiation wastewater is centralized and reused, and the water treatment agent is as follows: 1750g/t of polyaluminium chloride, 50g/t of polyacrylamide,
the production test indexes are shown in Table 3.
Table 3: example 3 test index
Mineral processing wastewater treatment water quality (mg/L)
| Item | PH | COD | Cu | Pb | Zn | SS |
| Before treatment | 7.15 | 350 | 0.12 | 0.65 | 1.10 | 295 |
| After treatment | 7.10 | 330 | 0.01 | 0.05 | 0.20 | 10 |
Example 4:
the properties and mineral composition of the ore selected for this example 4 were the same as in example 1. The beneficiation process flow is shown in figure 4. The technical process conditions of the example 4 are as follows:
carrying out grading overflow concentration on raw ores, and then removing lead and zinc; concentrating the zinc-selecting tailings, and then removing sulfur and manganese; all 100 percent of the raw ore concentrated wastewater and the lead concentrate wastewater are directly used for grinding classification and lead separation; at least 80% of the zinc tail concentrated wastewater and the zinc concentrate wastewater are directly used for zinc separation; at least 80% of the sulfur concentrate wastewater, the manganese concentrate wastewater and the tailing wastewater are directly used for sulfur and manganese separation; and (3) directly recycling all the excessive wastewater (at most 20% of zinc tail concentrated wastewater, zinc concentrate wastewater, sulfur concentrate wastewater, manganese concentrate wastewater and tailing wastewater) and recycling the wastewater for ore grinding classification, lead separation and the like after flocculation precipitation treatment, so that fine ore dressing of recycling the wastewater according to quality is realized. The dissolved medicine and the insufficient part are supplemented by mining underground water as fresh water, and the usage amount of the fresh water is 5 percent at most.
The method specifically comprises the following steps: after ball milling and grading of lead-zinc raw ore, controlling the mass concentration of grading overflow of a swirler to be 30% and the fineness of 80% with the fineness of-200 meshes (74 microns), concentrating and dehydrating the grading overflow until the mass concentration is 50% to remove lead, wherein the initial concentration of lead-selecting roughing is 50%; the lead-selecting medicament conditions are as follows: aniline black powder: butyl xanthate: buthanium nigrum ═ 8: 1: 1, 65g/t, zinc sulfate 500g/t, sodium sulfite 300g/t, lime 1000g/t (pH of ore pulp 9.0);
selecting zinc: the initial concentration of the selected zinc roughing is 42 percent, the copper sulfate is 400g/t, the lime is 2500g/t, the butyl xanthate is 200g/t, and the foaming agent (the pine oil) is 20 g/t.
Selecting sulfur: selecting sulfur, roughing and selecting the raw materials with the initial concentration of 50 percent, 6000g/t sulfuric acid and 800g/t butyl xanthate;
selecting manganese: the magnetic field intensity is 1.0T;
and (2) performing centralized treatment on redundant beneficiation wastewater (at most 20% of zinc tail concentrated wastewater, zinc concentrate wastewater, sulfur concentrate wastewater, manganese concentrate wastewater and tailing wastewater) and then recycling the redundant beneficiation wastewater for grinding classification and lead separation, wherein the water treatment agent comprises the following components: 350g/t of polyaluminium chloride and 10g/t of polyacrylamide.
The production test indexes are shown in Table 4.
Table 4: example 4 test index
Mineral processing wastewater treatment water quality (mg/L)
| Item | PH | COD | Cu | Pb | Zn | SS |
| Before treatment | 6.95 | 410 | 0.10 | 0.60 | 1.10 | 290 |
| After treatment | 6.95 | 400 | 0.01 | 0.01 | 0.20 | 10 |
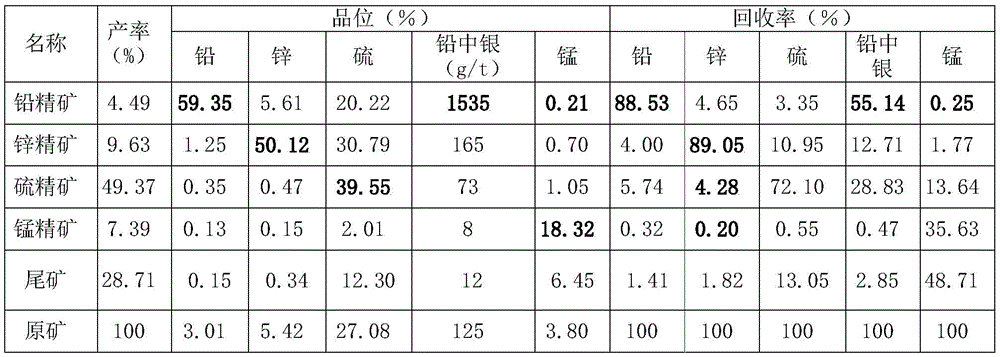
In this example 4, the main grade of the lead concentrate is 60.65%, the lead recovery rate is 91.88%, and the recovery rate of silver in lead is 62.93%; the main grade of the zinc concentrate is 50.67 percent, and the zinc recovery rate is 92.65 percent; the main grade of the sulfur concentrate is 46.66 percent, and the sulfur recovery rate is 82.66 percent; the main grade of the manganese concentrate is 20.63%, the manganese recovery rate is 62.16%, and the mineral separation index is high; more than 80% of the operation wastewater is directly reused for each operation, the recycling rate of the beneficiation wastewater is 100%, the energy consumption is saved by 20-25%, the cost of the medicament is reduced by 20-30%, and the treatment capacity of the tail end wastewater is only below 20%.
Table 4: comparison of examples
Comparison of technological index and Effect
Dose comparison
(g/t: amount of raw ore per ton)
In the beneficiation process of example 4, the raw ore classification overflow concentrated wastewater and the lead concentrate wastewater are all reused for the grinding classification and lead concentration operation, the grinding classification and lead concentration are not adversely affected, wastewater treatment is not required, and the wastewater contains chemical components required for lead concentration.
The zinc tailing concentrated overflow water and the zinc concentrate wastewater are recycled for zinc separation operation, the zinc separation is not affected, wastewater treatment is not needed, and the wastewater contains medicament components required by zinc separation.
The sulfur concentrate wastewater, the manganese concentrate wastewater and the tailing wastewater are recycled for sulfur and manganese separation operation, no adverse effect is caused on sulfur and manganese separation, no wastewater treatment is needed, and the wastewater contains medicament components required by sulfur separation.
The beneficiation wastewater is directly reused for zinc beneficiation, sulfur beneficiation, manganese beneficiation and other wastewater in a beneficiation plant, and is reused for ore grinding classification, lead beneficiation and the like after flocculation precipitation, so that the beneficiation wastewater is completely reused. The water for dissolving medicine and the insufficient water are supplemented by mining underground water as fresh water.
The invention discloses a mineral processing method for refined grading recycling of lead-zinc sulfide ore wastewater, wherein lead-zinc sulfide ore is subjected to ore grinding and grading, raw ore concentration and dehydration, lead (silver) separation, zinc separation, concentration and sulfur separation of zinc tails, final tailing precipitation, dehydration, filling and the like, and concentrate is subjected to concentration, filtration and dehydration to obtain lead concentrate, zinc concentrate, sulfur concentrate and manganese concentrate products; the beneficiation wastewater generated by beneficiation comprises: raw ore concentrated wastewater, zinc tail concentrated wastewater, lead concentrate wastewater, zinc concentrate wastewater, sulfur concentrate wastewater, manganese concentrate wastewater, tailing wastewater and the like; directly using the raw ore concentrated wastewater and the lead concentrate wastewater for ore grinding classification and lead separation; the zinc tail concentrated wastewater and the zinc concentrate wastewater are directly used for zinc separation; directly using the sulfur concentrate wastewater, the manganese concentrate wastewater and the tailing wastewater for sulfur and manganese separation; and all the waste water which is directly recycled and discharged is recycled for ore grinding classification, lead separation and the like after flocculation precipitation treatment, so that fine ore dressing of recycling waste water according to quality is realized. The process method has the advantages of high beneficiation main grade and recovery rate, low beneficiation cost, realization of full recycling and zero discharge of beneficiation wastewater, full utilization of the traditional Chinese medicine in the wastewater, high index, simple treatment, stable flow, convenient control, low consumption of hydroelectric medicine and the like, well solves the problems of long lead-zinc flotation flow, low concentration, insufficient beneficiation index, high consumption of hydroelectric medicine and unstable operation, and has very important significance for improving the recovery rate of lead-zinc-silver and the quality of concentrate, saving energy, reducing consumption and realizing clean production.
The technology is not only suitable for the beneficiation of lead-zinc polymetallic sulphide ores, but also suitable for the beneficiation of other non-ferrous metal sulphide ores.
Claims (4)
1. A mineral processing technique for refined grading recycling of lead-zinc sulfide ore wastewater is characterized by comprising the following steps: the method comprises the following steps:
after ball milling and grading of lead-zinc raw ore, the mass concentration of the graded overflow of the cyclone is 25-30%, the fineness of minus 200 meshes accounts for 75-80%, the raw ore is concentrated and dehydrated to 50-52% of mass concentration by a raw ore concentrator, a stirring barrel is used before lead separation, lead separation agents zinc sulfate 400-; concentrating, filtering and dehydrating the lead concentrate pulp to obtain final lead concentrate;
adding 350-450g/t of copper sulfate and 2500-2000 g/t of lime into the lead tailings to inhibit pyrite, adding 180-220g/t of butyl xanthate to collect zinc and 10-20g/t of foaming agent to obtain zinc concentrate ore pulp and zinc tailings; concentrating, filtering and dehydrating the zinc concentrate pulp to obtain final zinc concentrate;
concentrating and dehydrating the zinc tailings until the mass concentration is 50-52%, adding sulfuric acid into a stirring barrel before sulfur separation, adjusting the pH value to about 7, adding 600-800g/t of butyl xanthate for sulfur collection, and obtaining sulfur concentrate ore pulp and sulfur tailings; concentrating, filtering and dehydrating the sulfur concentrate pulp to obtain final sulfur concentrate;
removing high-gradient magnetic separation manganese from the sulfur tailings to obtain manganese concentrate and final tailings;
the tailings are precipitated and dehydrated for filling;
directly recycling all the raw ore concentrated wastewater and the lead concentrate wastewater for ore grinding grading and lead dressing; the zinc tailing concentrated overflow water and the zinc concentrate wastewater are recycled for zinc separation operation, the sulfur concentrate wastewater, the manganese concentrate wastewater and the tailing wastewater are recycled for sulfur separation and manganese separation operation, and the excessive wastewater is recycled for grinding classification and lead separation after flocculation precipitation, so that the whole recycling of the beneficiation wastewater is realized.
2. The beneficiation process method for refining, grading and recycling the lead-zinc sulfide ore wastewater according to the claim 1, which is characterized by comprising the following steps: the water for dissolving medicine and the insufficient water are supplemented by mining underground water as fresh water.
3. The beneficiation process method for refining, grading and recycling the lead-zinc sulfide ore wastewater according to the claim 1, which is characterized by comprising the following steps: in the combined collecting agent, the mass ratio of the aniline black drug to the butylammonium xanthate is 8: 1: 1.
4. the beneficiation process method for refining, grading and recycling the lead-zinc sulfide ore wastewater according to the claim 1, which is characterized by comprising the following steps: adding the following water treatment agents into the excessive wastewater for flocculation and precipitation: 350g/t of polyaluminium chloride and 10g/t of polyacrylamide.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110045194.7A CN112871462A (en) | 2021-01-13 | 2021-01-13 | Beneficiation process method for refined quality-based recycling of lead-zinc sulfide ore wastewater |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110045194.7A CN112871462A (en) | 2021-01-13 | 2021-01-13 | Beneficiation process method for refined quality-based recycling of lead-zinc sulfide ore wastewater |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN112871462A true CN112871462A (en) | 2021-06-01 |
Family
ID=76045821
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110045194.7A Pending CN112871462A (en) | 2021-01-13 | 2021-01-13 | Beneficiation process method for refined quality-based recycling of lead-zinc sulfide ore wastewater |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN112871462A (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115259459A (en) * | 2022-05-05 | 2022-11-01 | 中国矿业大学(北京) | Method for internal recycling of grading branch wastewater flow of concentrating mill |
| CN115821032A (en) * | 2022-12-07 | 2023-03-21 | 广东省科学院资源利用与稀土开发研究所 | Recycling method of lead-zinc ore beneficiation wastewater |
| CN116924534A (en) * | 2023-07-10 | 2023-10-24 | 云南科力环保股份公司 | Lead zinc ore tailing concentrating sedimentation and wastewater recycling method |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0790960A1 (en) * | 1994-11-18 | 1997-08-27 | Actew Corporation Ltd. | Wastewater treatment method and plant |
| CN1562794A (en) * | 2004-04-06 | 2005-01-12 | 南京栖霞山锌阳矿业有限公司 | Method for cyclic utilizing mineral dressing waste water from sulphur ore of lead-zinc |
| CN101195110A (en) * | 2007-12-31 | 2008-06-11 | 南京银茂铅锌矿业有限公司 | High concentration energy-saving environment protection beneficiation method for plumbum and zincium sulfuration mine |
| CN101856635A (en) * | 2010-04-26 | 2010-10-13 | 青岛黄金铅锌开发有限公司 | Method using mineral dressing backwater to float and to recover gold, silver, lead and zinc in cyanidation tailings of gold mine |
| CN102600979A (en) * | 2012-03-15 | 2012-07-25 | 南京银茂铅锌矿业有限公司 | Lead-zinc sulfide mineral separation method for saving fresh water |
| CN107840424A (en) * | 2017-11-04 | 2018-03-27 | 南京银茂铅锌矿业有限公司 | A kind of method of the efficient separate-recycling of lead-zinc metallogenic belt beneficiation wastewater |
| CN108176515A (en) * | 2017-12-21 | 2018-06-19 | 南京银茂铅锌矿业有限公司 | A kind of Pb-Zn deposits ore-dressing technique method for saving clean water |
| CN111589589A (en) * | 2020-05-28 | 2020-08-28 | 南京银茂铅锌矿业有限公司 | High-concentration efficient lead-zinc ore dressing process |
-
2021
- 2021-01-13 CN CN202110045194.7A patent/CN112871462A/en active Pending
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0790960A1 (en) * | 1994-11-18 | 1997-08-27 | Actew Corporation Ltd. | Wastewater treatment method and plant |
| CN1562794A (en) * | 2004-04-06 | 2005-01-12 | 南京栖霞山锌阳矿业有限公司 | Method for cyclic utilizing mineral dressing waste water from sulphur ore of lead-zinc |
| CN101195110A (en) * | 2007-12-31 | 2008-06-11 | 南京银茂铅锌矿业有限公司 | High concentration energy-saving environment protection beneficiation method for plumbum and zincium sulfuration mine |
| CN101856635A (en) * | 2010-04-26 | 2010-10-13 | 青岛黄金铅锌开发有限公司 | Method using mineral dressing backwater to float and to recover gold, silver, lead and zinc in cyanidation tailings of gold mine |
| CN102600979A (en) * | 2012-03-15 | 2012-07-25 | 南京银茂铅锌矿业有限公司 | Lead-zinc sulfide mineral separation method for saving fresh water |
| CN107840424A (en) * | 2017-11-04 | 2018-03-27 | 南京银茂铅锌矿业有限公司 | A kind of method of the efficient separate-recycling of lead-zinc metallogenic belt beneficiation wastewater |
| CN108176515A (en) * | 2017-12-21 | 2018-06-19 | 南京银茂铅锌矿业有限公司 | A kind of Pb-Zn deposits ore-dressing technique method for saving clean water |
| CN111589589A (en) * | 2020-05-28 | 2020-08-28 | 南京银茂铅锌矿业有限公司 | High-concentration efficient lead-zinc ore dressing process |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115259459A (en) * | 2022-05-05 | 2022-11-01 | 中国矿业大学(北京) | Method for internal recycling of grading branch wastewater flow of concentrating mill |
| CN115259459B (en) * | 2022-05-05 | 2024-02-02 | 中国矿业大学(北京) | Method for recycling in-process wastewater of sectional quality-dividing branch of concentrating mill |
| CN115821032A (en) * | 2022-12-07 | 2023-03-21 | 广东省科学院资源利用与稀土开发研究所 | Recycling method of lead-zinc ore beneficiation wastewater |
| CN116924534A (en) * | 2023-07-10 | 2023-10-24 | 云南科力环保股份公司 | Lead zinc ore tailing concentrating sedimentation and wastewater recycling method |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101884951B (en) | Combined mineral dressing technology of fine grain and micro grain cassiterite | |
| CN100537042C (en) | Flotation method of iron-bearing sphalerite and pyrrhotite type complex lead-zinc-silver sulfide ores | |
| CN111940118B (en) | Recovery method of secondary copper-containing low-grade copper-sulfur ore | |
| CN102029220B (en) | Separating flotation method of low-grade complex lead-antimony-zinc | |
| CN112264197B (en) | Combined inhibitor for high-magnetic pyrite type copper-sulfur ore and beneficiation method thereof | |
| CN104073645B (en) | A kind of method reclaiming valuable metal in ultra-fine cyanidation tailings with high cyanogen backwater to float | |
| CN111589589B (en) | High-concentration efficient lead-zinc ore dressing process | |
| CN100594986C (en) | High-concentration energy-saving and environment-friendly beneficiation method for lead-zinc sulfide ore | |
| CN101585017A (en) | Ore-selecting method of difficultly-selected copper zinc sulphur ore | |
| CN102357406B (en) | Beneficiation method for mud oxidized lead-zinc ores | |
| CN110170381B (en) | Beneficiation method for recovering cassiterite from tin-copper paragenic ore | |
| CN109127115B (en) | Method for recovering lead-zinc mineral from high-sulfur lead-zinc ore tailings | |
| CN111617884B (en) | Flotation separation method for copper, lead, zinc and arsenic in complex multi-metal sulfide ore | |
| CN112221699B (en) | Clean and efficient beneficiation method for complex gold, silver, copper, lead and zinc-containing composite ore | |
| CN112871462A (en) | Beneficiation process method for refined quality-based recycling of lead-zinc sulfide ore wastewater | |
| CN104148163A (en) | Beneficiation method for processing low grade tin-lead-zinc multi-metal oxidized ores | |
| CN107840424A (en) | A kind of method of the efficient separate-recycling of lead-zinc metallogenic belt beneficiation wastewater | |
| CN111495581A (en) | Beneficiation method for recycling lead-zinc ore beneficiation treatment wastewater | |
| CN106000620A (en) | Beneficiation method for recovering high iron and high sulfur concentrate from copper tailings containing sulfur and iron | |
| CN104815746A (en) | Recovery method of high-iron highly-argillaceous alkaline gangue refractory oxide copper ore | |
| CN113731642A (en) | Beneficiation method for high-sulfur lead-zinc ore under natural pH condition | |
| CN117000437A (en) | Flotation method of copper and cobalt ore with high oxidation rate | |
| CN100515576C (en) | Flotation method of ultra-fine lead-poor antimony-zinc flocculation carrier | |
| CN1017686B (en) | Method for floating copper concentrate from copper, lead and zinc-containing multi-metal complex sulfide ore | |
| CN109158216B (en) | High-efficiency flotation process for high-arsenic high-carbon refractory gold ore |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20210601 |
|
| RJ01 | Rejection of invention patent application after publication |