CN112870448B - Ultrahigh molecular weight polyethylene plate and preparation method and application thereof - Google Patents
Ultrahigh molecular weight polyethylene plate and preparation method and application thereof Download PDFInfo
- Publication number
- CN112870448B CN112870448B CN202110067333.6A CN202110067333A CN112870448B CN 112870448 B CN112870448 B CN 112870448B CN 202110067333 A CN202110067333 A CN 202110067333A CN 112870448 B CN112870448 B CN 112870448B
- Authority
- CN
- China
- Prior art keywords
- molecular weight
- ultra
- antioxidant
- weight polyethylene
- high molecular
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000004699 Ultra-high molecular weight polyethylene Substances 0.000 title claims abstract description 103
- 229920000785 ultra high molecular weight polyethylene Polymers 0.000 title claims abstract description 103
- 238000002360 preparation method Methods 0.000 title claims abstract description 14
- 239000003963 antioxidant agent Substances 0.000 claims abstract description 107
- 230000003078 antioxidant effect Effects 0.000 claims abstract description 88
- 239000011812 mixed powder Substances 0.000 claims abstract description 80
- 238000000748 compression moulding Methods 0.000 claims abstract description 53
- 238000004132 cross linking Methods 0.000 claims abstract description 45
- 239000000463 material Substances 0.000 claims abstract description 21
- 238000000034 method Methods 0.000 claims abstract description 20
- 238000000137 annealing Methods 0.000 claims abstract description 12
- 238000002156 mixing Methods 0.000 claims abstract description 12
- GVJHHUAWPYXKBD-UHFFFAOYSA-N (±)-α-Tocopherol Chemical compound OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 claims description 176
- 239000010410 layer Substances 0.000 claims description 141
- 235000006708 antioxidants Nutrition 0.000 claims description 100
- 235000019165 vitamin E Nutrition 0.000 claims description 89
- 239000011709 vitamin E Substances 0.000 claims description 89
- 229930003427 Vitamin E Natural products 0.000 claims description 88
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 claims description 88
- 229940046009 vitamin E Drugs 0.000 claims description 88
- 239000002344 surface layer Substances 0.000 claims description 15
- 238000003475 lamination Methods 0.000 claims description 14
- 238000000465 moulding Methods 0.000 claims description 13
- LNTHITQWFMADLM-UHFFFAOYSA-N gallic acid Chemical compound OC(=O)C1=CC(O)=C(O)C(O)=C1 LNTHITQWFMADLM-UHFFFAOYSA-N 0.000 claims description 9
- ZTHYODDOHIVTJV-UHFFFAOYSA-N Propyl gallate Chemical compound CCCOC(=O)C1=CC(O)=C(O)C(O)=C1 ZTHYODDOHIVTJV-UHFFFAOYSA-N 0.000 claims description 6
- 229940074391 gallic acid Drugs 0.000 claims description 6
- 235000004515 gallic acid Nutrition 0.000 claims description 6
- -1 phosphite compound Chemical class 0.000 claims description 6
- QAIPRVGONGVQAS-DUXPYHPUSA-N trans-caffeic acid Chemical compound OC(=O)\C=C\C1=CC=C(O)C(O)=C1 QAIPRVGONGVQAS-DUXPYHPUSA-N 0.000 claims description 6
- 238000001816 cooling Methods 0.000 claims description 4
- ACEAELOMUCBPJP-UHFFFAOYSA-N (E)-3,4,5-trihydroxycinnamic acid Natural products OC(=O)C=CC1=CC(O)=C(O)C(O)=C1 ACEAELOMUCBPJP-UHFFFAOYSA-N 0.000 claims description 3
- RPWFJAMTCNSJKK-UHFFFAOYSA-N Dodecyl gallate Chemical compound CCCCCCCCCCCCOC(=O)C1=CC(O)=C(O)C(O)=C1 RPWFJAMTCNSJKK-UHFFFAOYSA-N 0.000 claims description 3
- BGYHLZZASRKEJE-UHFFFAOYSA-N [3-[3-(3,5-ditert-butyl-4-hydroxyphenyl)propanoyloxy]-2,2-bis[3-(3,5-ditert-butyl-4-hydroxyphenyl)propanoyloxymethyl]propyl] 3-(3,5-ditert-butyl-4-hydroxyphenyl)propanoate Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(CCC(=O)OCC(COC(=O)CCC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)(COC(=O)CCC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)COC(=O)CCC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)=C1 BGYHLZZASRKEJE-UHFFFAOYSA-N 0.000 claims description 3
- 150000001412 amines Chemical class 0.000 claims description 3
- 229940074360 caffeic acid Drugs 0.000 claims description 3
- 235000004883 caffeic acid Nutrition 0.000 claims description 3
- ADRVNXBAWSRFAJ-UHFFFAOYSA-N catechin Natural products OC1Cc2cc(O)cc(O)c2OC1c3ccc(O)c(O)c3 ADRVNXBAWSRFAJ-UHFFFAOYSA-N 0.000 claims description 3
- 235000005487 catechin Nutrition 0.000 claims description 3
- QAIPRVGONGVQAS-UHFFFAOYSA-N cis-caffeic acid Natural products OC(=O)C=CC1=CC=C(O)C(O)=C1 QAIPRVGONGVQAS-UHFFFAOYSA-N 0.000 claims description 3
- 235000010386 dodecyl gallate Nutrition 0.000 claims description 3
- SSDSCDGVMJFTEQ-UHFFFAOYSA-N octadecyl 3-(3,5-ditert-butyl-4-hydroxyphenyl)propanoate Chemical compound CCCCCCCCCCCCCCCCCCOC(=O)CCC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 SSDSCDGVMJFTEQ-UHFFFAOYSA-N 0.000 claims description 3
- 235000010388 propyl gallate Nutrition 0.000 claims description 3
- 239000000473 propyl gallate Substances 0.000 claims description 3
- 229940075579 propyl gallate Drugs 0.000 claims description 3
- 239000003381 stabilizer Substances 0.000 claims description 3
- PFTAWBLQPZVEMU-DZGCQCFKSA-N (+)-catechin Chemical compound C1([C@H]2OC3=CC(O)=CC(O)=C3C[C@@H]2O)=CC=C(O)C(O)=C1 PFTAWBLQPZVEMU-DZGCQCFKSA-N 0.000 claims 1
- 229950001002 cianidanol Drugs 0.000 claims 1
- 230000005855 radiation Effects 0.000 abstract description 6
- 239000000843 powder Substances 0.000 abstract description 3
- 239000002861 polymer material Substances 0.000 abstract description 2
- 239000002245 particle Substances 0.000 description 23
- 238000000498 ball milling Methods 0.000 description 12
- 238000010894 electron beam technology Methods 0.000 description 11
- 150000003254 radicals Chemical class 0.000 description 9
- 239000012300 argon atmosphere Substances 0.000 description 8
- 229920003023 plastic Polymers 0.000 description 7
- 239000004033 plastic Substances 0.000 description 7
- 238000009826 distribution Methods 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 4
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- 229920000642 polymer Polymers 0.000 description 4
- 238000006243 chemical reaction Methods 0.000 description 3
- 210000001503 joint Anatomy 0.000 description 3
- 239000003960 organic solvent Substances 0.000 description 3
- 230000001590 oxidative effect Effects 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- 239000004698 Polyethylene Substances 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 230000009286 beneficial effect Effects 0.000 description 2
- 150000001765 catechin Chemical class 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 238000009792 diffusion process Methods 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 229920006351 engineering plastic Polymers 0.000 description 2
- 230000005251 gamma ray Effects 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 210000002540 macrophage Anatomy 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 230000035515 penetration Effects 0.000 description 2
- OJMIONKXNSYLSR-UHFFFAOYSA-N phosphorous acid Chemical class OP(O)O OJMIONKXNSYLSR-UHFFFAOYSA-N 0.000 description 2
- 229920000573 polyethylene Polymers 0.000 description 2
- 230000001681 protective effect Effects 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 208000003076 Osteolysis Diseases 0.000 description 1
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 1
- 238000005299 abrasion Methods 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 239000012620 biological material Substances 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 238000012668 chain scission Methods 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000032798 delamination Effects 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000013467 fragmentation Methods 0.000 description 1
- 238000006062 fragmentation reaction Methods 0.000 description 1
- 210000004394 hip joint Anatomy 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 210000000629 knee joint Anatomy 0.000 description 1
- 239000002648 laminated material Substances 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 238000005461 lubrication Methods 0.000 description 1
- 208000029791 lytic metastatic bone lesion Diseases 0.000 description 1
- 238000003801 milling Methods 0.000 description 1
- 239000012299 nitrogen atmosphere Substances 0.000 description 1
- 239000007800 oxidant agent Substances 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 239000005022 packaging material Substances 0.000 description 1
- 150000002978 peroxides Chemical class 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 238000007348 radical reaction Methods 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 238000005070 sampling Methods 0.000 description 1
- 229910000077 silane Inorganic materials 0.000 description 1
- 229920001169 thermoplastic Polymers 0.000 description 1
- 239000004416 thermosoftening plastic Substances 0.000 description 1
- 238000009827 uniform distribution Methods 0.000 description 1
- 238000009461 vacuum packaging Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/16—Macromolecular materials obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2430/00—Materials or treatment for tissue regeneration
- A61L2430/24—Materials or treatment for tissue regeneration for joint reconstruction
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Dermatology (AREA)
- Medicinal Chemistry (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Transplantation (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Treatments Of Macromolecular Shaped Articles (AREA)
- Casting Or Compression Moulding Of Plastics Or The Like (AREA)
Abstract
本发明属于高分子材料技术领域,具体涉及一种超高分子量聚乙烯板材及其制备方法和应用。本发明提供了一种超高分子量聚乙烯板材制备方法,包括以下步骤:将抗氧剂和超高分子量聚乙烯混合,得到抗氧剂含量不同的多种混合粉料;将所述多种混合粉料按照抗氧剂含量层叠后依次进行模压成型、辐照交联和退火处理,得到所述超高分子量聚乙烯板材;所述层叠后层材料以中心层为中心对称排列;所述层叠后各层材料中抗氧剂的含量以中心层为基准向外递增。本发明将不同抗氧剂浓度的混合粉料按照抗氧剂浓度由内往外递增的方式进行层叠,保证辐照过程中不同样品深度的抗氧化剂含量和辐照剂量的比例保持恒定,得到交联密度均匀的超高分子量聚乙烯板材。
The invention belongs to the technical field of polymer materials, and in particular relates to an ultra-high molecular weight polyethylene sheet and a preparation method and application thereof. The invention provides a method for preparing an ultra-high molecular weight polyethylene sheet, which comprises the following steps: mixing an antioxidant and ultra-high molecular weight polyethylene to obtain various mixed powders with different antioxidant contents; The powders are stacked according to the antioxidant content and then subjected to compression molding, radiation cross-linking and annealing treatment in sequence to obtain the ultra-high molecular weight polyethylene sheet; the layered materials are arranged symmetrically with the center layer as the center; after the stacking The content of antioxidants in the materials of each layer increases outward based on the center layer. In the invention, the mixed powders with different antioxidant concentrations are stacked in a manner that the antioxidant concentration increases from the inside to the outside, so as to ensure that the ratio of the antioxidant content and the irradiation dose of different sample depths in the irradiation process is kept constant, and cross-linking is obtained. UHMWPE sheet with uniform density.
Description
技术领域technical field
本发明属于高分子材料技术领域,具体涉及一种超高分子量聚乙烯板材及其制备方法和应用。The invention belongs to the technical field of polymer materials, and in particular relates to an ultra-high molecular weight polyethylene sheet and a preparation method and application thereof.
背景技术Background technique
超高分子量聚乙烯(分子量100万以上的聚乙烯)由于其优异的综合性能,具有其它工程塑料无可比拟的耐磨性、耐腐蚀性、抗冲击性和自润滑性,是一种新型热塑性工程塑料,被广泛用于人工关节置换。但是超高分子量聚乙烯人工关节在体内长期服役过程中,与其搭配材料摩擦产生磨屑被巨噬细胞吞噬后不能被吸收降解,导致巨噬细胞死亡,同时会产生磨损、动作性骨溶解,无菌性松动、炎症等,常常需进行二次手术或多次翻修置换手术。为了解决上述问题,研究开发出了用硅烷化学交联的超高分子量聚乙烯,但是其产量极低且临床磨损率极高(Kurtz S.UHMWPE Biomaterials Handbook[J].2009,33-43),随后出现的高能电子束和γ射线辐照交联的方法改善了交联超高分子量聚乙烯的耐磨性。Ultra-high molecular weight polyethylene (polyethylene with a molecular weight of more than 1 million) has unparalleled wear resistance, corrosion resistance, impact resistance and self-lubrication properties of other engineering plastics due to its excellent comprehensive properties. It is a new type of thermoplastic. Engineering plastics are widely used in artificial joint replacement. However, during the long-term service of the UHMWPE artificial joint in the body, friction with its matching material produces wear debris that is phagocytosed by macrophages and cannot be absorbed and degraded, resulting in the death of macrophages. At the same time, wear and active osteolysis will occur. Bacterial loosening, inflammation, etc., often require a second operation or multiple revision and replacement operations. In order to solve the above problems, ultra-high molecular weight polyethylene chemically cross-linked with silane has been developed, but its yield is extremely low and the clinical wear rate is extremely high (Kurtz S.UHMWPE Biomaterials Handbook[J].2009,33-43), Subsequent methods of crosslinking with high-energy electron beams and γ-ray irradiation improved the abrasion resistance of crosslinked UHMWPE.
但是辐照交联时存在射线穿透力问题,辐照剂量在厚板材中的分布不均匀,总体趋势是外层剂量大,内层剂量小。实验发现,10MeV直线电子加速器对超高分子量聚乙烯的穿透深度低于50mm,板材内外层辐照剂量悬殊。辐照剂量的差异,直接导致交联密度分布不均匀。交联密度及其分布与超高分子量聚乙烯板材的晶区与非晶区的分布以及交联超高分子量聚乙烯板材制品的强度、韧性磨损性密切相关。虽然辐照交联通过提高其交联度大大提高了耐磨性,但其在交联形成的过程中残留的自由基可与溶解氧发生连锁氧化反应,形成不稳定的过氧化物,过氧化物的降解通过断链和重结晶使聚合物脆化,导致聚合物性能下降,最终表现为交联超高分子量聚乙烯材料出现氧化碎裂、分层等问题。为解决上述问题,在超高分子量聚乙烯板材中添加生物相容性的抗氧化剂(如维生素E)来消除残留自由基,改善超高分子量聚乙烯人工关节的氧化稳定性。However, there is a problem of ray penetration during irradiation cross-linking, and the distribution of the irradiation dose in the thick plate is not uniform. The general trend is that the outer layer dose is large and the inner layer dose is small. The experiment found that the penetration depth of 10MeV linear electron accelerator to ultra-high molecular weight polyethylene is less than 50mm, and the radiation dose of the inner and outer layers of the plate is very different. The difference in irradiation dose directly leads to the uneven distribution of crosslinking density. The crosslinking density and its distribution are closely related to the distribution of the crystal and amorphous regions of the UHMWPE sheet and the strength, toughness and wear resistance of the crosslinked UHMWPE sheet product. Although radiation crosslinking greatly improves wear resistance by increasing its crosslinking degree, the residual free radicals in the process of crosslinking formation can undergo chain oxidation reaction with dissolved oxygen to form unstable peroxides. The degradation of the polymer embrittles the polymer through chain scission and recrystallization, which leads to the decline of the polymer performance, and finally manifests as oxidative fragmentation and delamination of the cross-linked ultra-high molecular weight polyethylene material. In order to solve the above problems, biocompatible antioxidants (such as vitamin E) are added to the ultra-high molecular weight polyethylene sheet to eliminate residual free radicals and improve the oxidative stability of ultra-high molecular weight polyethylene artificial joints.
传统抗氧化剂的添加方式主要分为两大类,一类是在辐照前将抗氧化剂混入超高分子量聚乙烯粉料中,形成预制品,例如公开号为EP2291417B1和CN102089333A的专利公开的方案。然而研究表明辐照交联之前添加抗氧剂将抑制交联,超高分子量聚乙烯中抗氧剂含量较高时(如维生素E浓度高于0.1wt%),抗氧剂会与聚烯烃中辐照产生的自由基反应,从而阻碍聚合物中自由基之间的交联反应;抗氧剂不仅抑制了交联反应,也加速了超高分子量聚乙烯分子链的断裂,降低了分子量,不利于提高交联程度,影响耐磨损性能和综合力学性能;当维生素E的含量过低(如维生素E的含量低于0.01wt%时),维生素E受到高能射线的辐射,会丧失抗氧化活性,失去抗氧化作用。另一添加抗氧剂的方式是将辐照交联后的超高分子量聚乙烯浸入抗氧剂中,在120℃左右使抗氧剂扩散到超高分子量聚乙烯板材表面。将板材取出后继续保持120℃一段时间使抗氧剂向内部扩散。也可将超高分子量聚乙烯假体毛坯辐照交联后浸入抗氧剂溶液中,取出后进一步热处理使抗氧剂向内部扩散。然而采用辐照-浸泡-扩散的方法,抗氧剂扩散困难,主要分布在表层,内部抗氧剂的含量微乎其微,内外浓度不均一。而且热处理过程耗时长,长时间热处理也导致材料性能下降。而内部过低的抗氧剂含量起不到消除自由基的作用,不能有效消除辐照过程中产生的自由基以及发挥后续使用过程中的抗氧化作用。The traditional methods of adding antioxidants are mainly divided into two categories. One is to mix antioxidants into ultra-high molecular weight polyethylene powder before irradiation to form pre-products, such as the solutions disclosed in patents with publication numbers EP2291417B1 and CN102089333A. However, studies have shown that adding antioxidants before irradiation crosslinking will inhibit crosslinking. When the content of antioxidants in ultra-high molecular weight polyethylene is high (for example, the concentration of vitamin E is higher than 0.1 wt%), antioxidants will interact with polyolefins. The free radical reaction generated by irradiation hinders the cross-linking reaction between free radicals in the polymer; the antioxidant not only inhibits the cross-linking reaction, but also accelerates the breakage of the ultra-high molecular weight polyethylene molecular chain, reduces the molecular weight, and prevents the It is beneficial to improve the degree of cross-linking and affect the wear resistance and comprehensive mechanical properties; when the content of vitamin E is too low (for example, when the content of vitamin E is less than 0.01wt%), vitamin E will lose its antioxidant activity when irradiated by high-energy rays , loses its antioxidant effect. Another way to add antioxidant is to immerse the cross-linked ultra-high molecular weight polyethylene in the antioxidant, and make the antioxidant diffuse to the surface of the ultra-high molecular weight polyethylene sheet at about 120°C. After the plate was taken out, it was kept at 120°C for a period of time to diffuse the antioxidant into the interior. The ultra-high molecular weight polyethylene prosthesis blank can also be irradiated and cross-linked and then immersed in an antioxidant solution, taken out, and further heat-treated to diffuse the antioxidant to the inside. However, with the method of irradiation-immersion-diffusion, the diffusion of antioxidants is difficult, mainly distributed in the surface layer, the content of internal antioxidants is very small, and the concentration inside and outside is not uniform. Moreover, the heat treatment process takes a long time, and the long time heat treatment also leads to the deterioration of material properties. However, the internal antioxidant content that is too low cannot eliminate free radicals, and cannot effectively eliminate free radicals generated during the irradiation process and play an antioxidant role in the subsequent use process.
发明内容SUMMARY OF THE INVENTION
有鉴于此,本发明提供了一种超高分子量聚乙烯板材及其制备方法和应用,本发明将抗氧剂浓度由内到外递增设置,以保证辐照过程中,不同样品深度的抗氧化剂含量和辐照剂量的比例保持恒定,从而获得具有均匀交联密度的超高分子量聚乙烯板材。In view of this, the present invention provides an ultra-high molecular weight polyethylene sheet and a preparation method and application thereof. The present invention increases the concentration of antioxidants from the inside to the outside, so as to ensure that during the irradiation process, the antioxidants at different sample depths are The ratio of the content and the irradiation dose was kept constant, so that UHMWPE sheets with uniform crosslink density were obtained.
为了解决上述技术问题,本发明提供了一种超高分子量聚乙烯板材制备方法,包括以下步骤:In order to solve the above-mentioned technical problems, the present invention provides a method for preparing an ultra-high molecular weight polyethylene sheet, comprising the following steps:
将抗氧剂和超高分子量聚乙烯混合,得到抗氧剂含量不同的多种混合粉料;Mixing antioxidant and ultra-high molecular weight polyethylene to obtain various mixed powders with different antioxidant contents;
将所述多种混合粉料按照抗氧剂含量层叠后依次进行模压成型、辐照交联和退火处理,得到所述超高分子量聚乙烯板材;The multiple mixed powders are layered according to the antioxidant content and then subjected to compression molding, irradiation crosslinking and annealing treatment in sequence to obtain the ultra-high molecular weight polyethylene sheet;
所述层叠后层材料以中心层为中心对称排列;The layered materials are arranged symmetrically with the center layer as the center;
所述层叠后各层材料中抗氧剂的含量以中心层为基准向外递增。After the lamination, the content of the antioxidant in the materials of each layer increases outward on the basis of the center layer.
优选的,所述混合粉料中抗氧剂的质量百分含量为0.01~1%中的多种。Preferably, the mass percentage content of antioxidants in the mixed powder is 0.01-1%.
优选的,所述层叠的层数为3~9层。Preferably, the number of the stacked layers is 3 to 9 layers.
优选的,所述中心层中抗氧剂的质量百分含量为0.01~0.3%;所述超高分子量聚乙烯板材的表层中抗氧剂的质量百分含量为0.1~1%。Preferably, the mass percentage content of the antioxidant in the central layer is 0.01-0.3%; the mass percentage content of the antioxidant in the surface layer of the ultra-high molecular weight polyethylene sheet is 0.1-1%.
优选的,所述超高分子量聚乙烯板材的厚度为30mm~100mm。Preferably, the thickness of the ultra-high molecular weight polyethylene sheet is 30mm˜100mm.
优选的,所述模压成型包括:依次进行高温模压成型和低温模压成型;Preferably, the compression molding comprises: sequentially performing high temperature compression molding and low temperature compression molding;
所述高温模压成型的温度为170~240℃,压力为1~50MPa,时间为0.5~10h;The temperature of the high-temperature compression molding is 170-240° C., the pressure is 1-50MPa, and the time is 0.5-10h;
所述低温模压成型的温度为110~130℃,压力为1~50MPa,时间为0.5~72h。The temperature of the low-temperature molding is 110-130° C., the pressure is 1-50 MPa, and the time is 0.5-72 h.
优选的,所述辐照交联的辐照剂量为25~150kGy,剂量率为5~50kGy/pass;Preferably, the irradiation dose of the irradiation crosslinking is 25-150 kGy, and the dose rate is 5-50 kGy/pass;
所述退火处理的温度为110~130℃,保温时间为0.5~72h。The temperature of the annealing treatment is 110-130° C., and the holding time is 0.5-72 h.
优选的,所述抗氧剂包括亚磷酸酯化合物、没食子酸、儿茶素、十二烷基没食子酸、没食子酸丙酯、没食子酸月桂酯、咖啡酸、受阻胺稳定剂TH-944、维生素E、抗氧剂1076和抗氧剂1010中的一种或多种。Preferably, the antioxidants include phosphite compounds, gallic acid, catechins, lauryl gallic acid, propyl gallate, lauryl gallate, caffeic acid, hindered amine stabilizer TH-944, vitamins E. One or more of antioxidant 1076 and antioxidant 1010.
本发明还提供了上述技术方案所述制备方法制备得到的超高分子量聚乙烯板材,所述超高分子量聚乙烯板材中最高交联密度和最低交联密度的差值为0~100mol/m3。The present invention also provides an ultra-high molecular weight polyethylene sheet prepared by the preparation method described in the above technical solution, wherein the difference between the highest cross-linking density and the lowest cross-linking density in the ultra-high molecular weight polyethylene sheet is 0-100 mol/m 3 . .
本发明还提供了上述技术方案所述超高分子量聚乙烯板材在制备人工关节中的应用。The present invention also provides the application of the ultra-high molecular weight polyethylene sheet in the above technical solution in the preparation of artificial joints.
本发明提供了一种超高分子量聚乙烯板材制备方法,包括以下步骤:将抗氧剂和超高分子量聚乙烯混合,得到抗氧剂含量不同的多种混合粉料;将所述多种混合粉料按照抗氧剂含量层叠后依次进行模压成型、辐照交联和退火处理,得到所述超高分子量聚乙烯板材;所述层叠后层材料以中心层为中心对称排列;所述层叠后各层材料中抗氧剂的含量以中心层为基准向外递增。本发明将抗氧剂按照一定浓度顺序和超高分子量聚乙烯混合,将不同抗氧剂浓度的混合粉料按照抗氧剂浓度由内往外递增的方式进行层叠,保证辐照过程中不同样品深度的抗氧化剂含量和辐照剂量的比例保持恒定,得到交联密度均匀的超高分子量聚乙烯板材。同时本发明通过分层设置抗氧剂的浓度调控抗氧剂对辐照交联抑制作用的影响,确保内外层抗氧剂有效浓度足够消除残余自由基,提高了超高分子量聚乙烯板材的拉伸强度、断裂伸长率和冲击强度。The invention provides a method for preparing an ultra-high molecular weight polyethylene sheet, which comprises the following steps: mixing an antioxidant and ultra-high molecular weight polyethylene to obtain various mixed powders with different antioxidant contents; The powders are layered according to the antioxidant content and then subjected to compression molding, irradiation cross-linking and annealing treatment in sequence to obtain the ultra-high molecular weight polyethylene sheet; the layered materials are arranged symmetrically with the center layer as the center; after the layering The content of antioxidants in the materials of each layer increases outward based on the center layer. In the present invention, antioxidants are mixed with ultra-high molecular weight polyethylene according to a certain concentration sequence, and mixed powders with different antioxidant concentrations are stacked in a manner in which the antioxidant concentrations increase from the inside to the outside, so as to ensure different sample depths during the irradiation process. The ratio of the antioxidant content and the irradiation dose is kept constant, and the ultra-high molecular weight polyethylene sheet with uniform crosslinking density is obtained. At the same time, the present invention regulates the effect of the antioxidant on the inhibition of radiation crosslinking by setting the concentration of the antioxidant in layers, so as to ensure that the effective concentration of the antioxidant in the inner and outer layers is sufficient to eliminate residual free radicals, and the tensile strength of the ultra-high molecular weight polyethylene sheet is improved. Tensile strength, elongation at break and impact strength.
本发明还提供了上述技术方案所述制备方法制备得到的超高分子量聚乙烯板材,所述超高分子量聚乙烯板材中最高交联密度和最低交联密度的差值为0~100mol/m3。本发明提供的超高分子量聚乙烯板材具有均匀的交联密度,从而提高了超高分子量聚乙烯板材的拉伸强度、断裂伸长率和冲击强度。The present invention also provides an ultra-high molecular weight polyethylene sheet prepared by the preparation method described in the above technical solution, wherein the difference between the highest cross-linking density and the lowest cross-linking density in the ultra-high molecular weight polyethylene sheet is 0-100 mol/m 3 . . The ultra-high molecular weight polyethylene sheet provided by the invention has uniform cross-linking density, thereby improving the tensile strength, elongation at break and impact strength of the ultra-high molecular weight polyethylene sheet.
附图说明Description of drawings
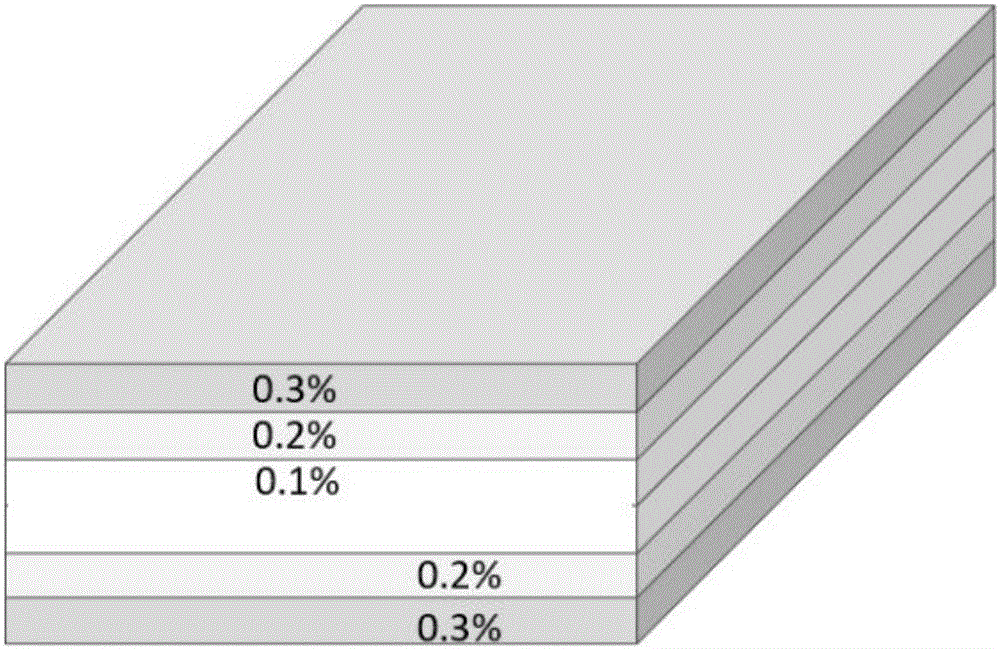
图1为实施例2制备得到的超高分子量聚乙烯板材的结构示意图。1 is a schematic structural diagram of the ultra-high molecular weight polyethylene sheet prepared in Example 2.
具体实施方式Detailed ways
本发明提供了一种超高分子量聚乙烯板材制备方法,包括以下步骤:The invention provides a method for preparing an ultra-high molecular weight polyethylene sheet, comprising the following steps:
将抗氧剂和超高分子量聚乙烯混合,得到抗氧剂含量不同的多种混合粉料;Mixing antioxidant and ultra-high molecular weight polyethylene to obtain various mixed powders with different antioxidant contents;
将所述多种混合粉料按照抗氧剂含量层叠后依次进行模压成型、辐照交联和退火处理,得到所述超高分子量聚乙烯板材;The multiple mixed powders are layered according to the antioxidant content and then subjected to compression molding, irradiation crosslinking and annealing treatment in sequence to obtain the ultra-high molecular weight polyethylene sheet;
所述层叠后层材料以中心层为中心对称排列;The layered materials are arranged symmetrically with the center layer as the center;
所述层叠后各层材料中抗氧剂的含量以中心层为基准向外递增。After the lamination, the content of the antioxidant in the materials of each layer increases outward on the basis of the center layer.
本发明将抗氧剂和超高分子量聚乙烯混合,得到抗氧剂含量不同的多种混合粉料。在本发明中,所述抗氧剂优选包括亚磷酸酯化合物、没食子酸、儿茶素、十二烷基没食子酸、没食子酸丙酯、没食子酸月桂酯、咖啡酸、受阻胺稳定剂TH-944、维生素E、抗氧剂1076和抗氧剂1010中的一种或多种,更优选为维生素E;当所述抗氧剂包括上述两种或两种以上上述具体物质时,本发明对具体物质的配比无特殊限定,采用任意配比即可。在本发明中,所述多种混合粉料中抗氧剂的质量百分含量优选为0.01~1%中的多种,更优选为0.1~0.9%,更进一步优选为0.175~0.35%,本发明得到多种氧化剂含量的混合粉料。在本发明中,所述混合粉料的平均粒径优选为50~200μm,更优选为80~130μm。在本发明中,所述混合优选包括两种混合方式中的任意一种:In the invention, the antioxidant and the ultra-high molecular weight polyethylene are mixed to obtain various mixed powders with different contents of the antioxidant. In the present invention, the antioxidant preferably includes phosphite compounds, gallic acid, catechins, lauryl gallic acid, propyl gallate, lauryl gallate, caffeic acid, hindered amine stabilizer TH- 944. One or more of vitamin E, antioxidant 1076 and antioxidant 1010, more preferably vitamin E; when the antioxidant includes two or more of the above-mentioned specific substances, the The proportion of specific substances is not particularly limited, and any proportion may be used. In the present invention, the mass percentage content of antioxidants in the various mixed powders is preferably a variety of 0.01-1%, more preferably 0.1-0.9%, still more preferably 0.175-0.35%, the present The invention obtains mixed powders with various oxidant contents. In the present invention, the average particle size of the mixed powder is preferably 50-200 μm, more preferably 80-130 μm. In the present invention, the mixing preferably includes any one of two mixing modes:
第一种混合方式优选包括以下步骤:将抗氧剂和超高分子量聚乙烯混合后进行球磨;所述球磨的转速优选为30~800r/min,更优选为100~400r/min;时间优选为5~60min,更优选为10~30min。The first mixing method preferably includes the following steps: mixing the antioxidant and ultra-high molecular weight polyethylene and then performing ball milling; the rotation speed of the ball milling is preferably 30-800 r/min, more preferably 100-400 r/min; the time is preferably 5 to 60 minutes, more preferably 10 to 30 minutes.
第二种混合方式优选包括以下步骤:将抗氧剂溶解于有机溶剂中,得到抗氧剂溶液;The second mixing method preferably includes the following steps: dissolving the antioxidant in an organic solvent to obtain an antioxidant solution;
将所述抗氧剂溶液和超高分子量聚乙烯混合后进行干燥,得到混合粉料。The antioxidant solution and the ultra-high molecular weight polyethylene are mixed and then dried to obtain a mixed powder.
本发明将抗氧剂溶解于有机溶剂中,得到抗氧剂溶液。在本发明中,所述有机溶剂优选包括丙酮、正丁醇、乙醇、石油醚或环己烷,更优选为正丁醇或乙醇;所述抗氧剂溶液的浓度优选为20~300g/L,更优选为80~200g/L。In the present invention, the antioxidant is dissolved in an organic solvent to obtain an antioxidant solution. In the present invention, the organic solvent preferably includes acetone, n-butanol, ethanol, petroleum ether or cyclohexane, more preferably n-butanol or ethanol; the concentration of the antioxidant solution is preferably 20-300 g/L , more preferably 80 to 200 g/L.
得到抗氧剂溶液后本发明将所述抗氧剂溶液和超高分子量聚乙烯混合后进行干燥,得到混合粉料。本发明对所述混合的方式无特殊限定,只要能够混合均匀即可。在本发明中,所述干燥的温度优选为30~90℃,更优选为40~70℃;时间优选为3~15天,更优选为6~10天。本发明对所述干燥的方式无特殊限定,只要能够达到上述温度即可。After the antioxidant solution is obtained, in the present invention, the antioxidant solution is mixed with ultra-high molecular weight polyethylene and then dried to obtain a mixed powder. The present invention does not specifically limit the manner of mixing, as long as the mixing can be uniform. In the present invention, the drying temperature is preferably 30-90°C, more preferably 40-70°C; the time is preferably 3-15 days, more preferably 6-10 days. In the present invention, the drying method is not particularly limited, as long as the above-mentioned temperature can be reached.
得到抗氧剂含量不同的多种混合粉料后,本发明将所述多种混合粉料按照抗氧剂含量层叠后依次进行模压成型、辐照交联和退火处理,得到所述超高分子量聚乙烯板材;所述层叠后层材料以中心层为中心对称排列;所述层叠后各层材料中抗氧剂的含量以中心层为基准向外递增。在本发明中,所述层叠的层数优选为3~9层,更优选为5~7层。在本发明中,若相邻两层中抗氧剂浓度相同,则相邻两层认为是一层。在本发明中,所述层叠后得到的坯料以中心层为中心对称排列,两个对称层中抗氧剂的含量相同,且两个对称层的厚度也相同。本发明对各对称层的厚度无特殊限定,只要能够使抗氧剂含量和辐照剂量的比例随层叠材料的深度变化保持恒定即可。After obtaining multiple mixed powders with different antioxidant contents, the present invention stacks the multiple mixed powders according to the antioxidant content and sequentially performs compression molding, irradiation crosslinking and annealing treatment to obtain the ultra-high molecular weight. Polyethylene sheet; the layer materials after lamination are arranged symmetrically with the center layer as the center; the content of antioxidants in each layer material after the lamination increases outwards based on the center layer. In the present invention, the number of layers to be stacked is preferably 3 to 9 layers, and more preferably 5 to 7 layers. In the present invention, if the antioxidant concentration in the two adjacent layers is the same, the two adjacent layers are regarded as one layer. In the present invention, the blanks obtained after the lamination are symmetrically arranged with the center layer as the center, the content of the antioxidant in the two symmetrical layers is the same, and the thicknesses of the two symmetrical layers are also the same. In the present invention, the thickness of each symmetrical layer is not particularly limited, as long as the ratio of the antioxidant content and the irradiation dose can be kept constant as the depth of the laminated material changes.
在本发明中,所述超高分子量聚乙烯板材的中心层材料中抗氧剂的质量百分含量优选为0.01~0.3%,更优选为0.05~0.1%;所述超高分子量聚乙烯板材的表层材料中抗氧剂的质量百分含量优选为0.1~1%,更优选为0.3~0.9%。在本发明中,所述层叠后各层材料中抗氧剂的含量以中心层为基准向外递增;本发明对所述递增的幅度无特殊限定,只要能够使抗氧化含量和辐照剂量的比例随层叠材料的深度变化保持恒定即可。In the present invention, the mass percentage content of the antioxidant in the center layer material of the UHMWPE sheet is preferably 0.01-0.3%, more preferably 0.05-0.1%; The mass percentage content of the antioxidant in the surface layer material is preferably 0.1-1%, more preferably 0.3-0.9%. In the present invention, after the lamination, the content of antioxidants in the materials of each layer increases outward based on the center layer; the present invention does not limit the range of the increase, as long as the antioxidant content and the radiation dose can be adjusted. It is sufficient that the ratio remains constant as the depth of the laminate changes.
在本发明中,所述超高分子量聚乙烯板材的层叠层数具体为3层,5层,7层或9层。当层数为3层时,中心层中抗氧剂的质量百分含量为0.1%;表层中抗氧剂的质量百分含量为0.3%。当层数为5层时,中心层中抗氧剂的质量百分含量为0.1%;与中心层相邻层中抗氧剂的质量百分含量为0.2%;表层中抗氧剂的质量百分含量为0.3%。当层数为5层时还可以为,中心层中抗氧剂的质量百分含量为0.3%;与中心层相邻层中抗氧剂的质量百分含量为0.6%;表层中抗氧剂的质量百分含量为0.9%。当层数为7层时,中心层中抗氧剂的质量百分含量为0.1%;与中心层相邻层中抗氧剂的质量百分含量为0.15%;与表层相邻层中抗氧剂的质量百分含量为0.25%;表层中抗氧剂的质量百分含量为0.3%。当层数为7层时还可以为,中心层中抗氧剂的质量百分含量为0.05%;与中心层相邻层中抗氧剂的质量百分含量为0.15%;与表层相邻层中抗氧剂的质量百分含量为0.2%;表层中抗氧剂的质量百分含量为0.3%。当层数为9层时,中心层中抗氧剂的质量百分含量为0.05%;与中心层相邻层中抗氧剂的质量百分含量为0.075%;从表层开始第三层中抗氧剂的质量百分含量为0.175%;与表层相邻层中抗氧剂的质量百分含量为0.27%;表层中抗氧剂的质量百分含量为0.35%。In the present invention, the number of laminated layers of the ultra-high molecular weight polyethylene sheet is specifically 3 layers, 5 layers, 7 layers or 9 layers. When the number of layers is 3, the mass percentage content of antioxidant in the central layer is 0.1%; the mass percentage content of antioxidant in the surface layer is 0.3%. When the number of layers is 5, the mass percentage of antioxidant in the central layer is 0.1%; the mass percentage of antioxidant in the layer adjacent to the central layer is 0.2%; the mass percentage of antioxidant in the surface layer is 100% The sub-content is 0.3%. When the number of layers is 5, the mass percentage of antioxidant in the central layer is 0.3%; the mass percentage of antioxidant in the layer adjacent to the central layer is 0.6%; the antioxidant in the surface layer is 0.6% by mass. The mass percentage of 0.9%. When the number of layers is 7, the mass percentage of antioxidant in the central layer is 0.1%; the mass percentage of antioxidant in the layer adjacent to the central layer is 0.15%; the antioxidant in the layer adjacent to the surface layer is 0.15% by mass The mass percentage of the agent is 0.25%; the mass percentage of the antioxidant in the surface layer is 0.3%. When the number of layers is 7, the mass percentage content of antioxidant in the central layer is 0.05%; the mass percentage content of antioxidant in the layer adjacent to the central layer is 0.15%; the mass percentage content of the layer adjacent to the surface layer The mass percentage content of the antioxidant in the middle layer is 0.2%; the mass percentage content of the antioxidant in the surface layer is 0.3%. When the number of layers is 9, the mass percentage of antioxidant in the central layer is 0.05%; the mass percentage of antioxidant in the layer adjacent to the central layer is 0.075%; starting from the surface layer, the antioxidant in the third layer The mass percentage content of the oxygen agent is 0.175%; the mass percentage content of the antioxidant in the layer adjacent to the surface layer is 0.27%; the mass percentage content of the antioxidant in the surface layer is 0.35%.
在本发明中,所述层叠优选在模具中进行,本发明对所述模具的形状无特殊限定,根据超高分子量聚乙烯板材所需形状设定即可。In the present invention, the lamination is preferably performed in a mold, and the shape of the mold is not particularly limited in the present invention, and can be set according to the desired shape of the ultra-high molecular weight polyethylene sheet.
本发明对所述模压成型的装置无特殊限定,在本发明的实施例中,所述模压成型在平板硫化机中进行。本发明在进行模压成型之前还优选包括将层叠后的产物进行初步模压成型,所述初步模压成型的压力优选为5~15MPa,更优选为8~10MPa;时间优选为0.8~1.2h,更优选为1h。本发明进行初步模压成型能够将混合粉料压实,利于后续模压成型的进行。在本发明中,所述模压成型优选包括:依次进行高温模压成型和低温模压成型;所述高温模压成型的温度优选为170~240℃,更优选为200~220℃;压力优选为1~50MPa,更优选为3~20MPa;时间优选为0.5~10h,更优选为3~6h。所述低温模压成型的温度优选为110~130℃,更优选为120~125℃;压力优选为1~50MPa,更优选为10~20MPa;时间优选为0.5~72h,更优选为2.5~10h。在本发明中,所述低温模压成型的温度优选在高温模压成型温度的基础上降温得到,本发明对所述降温速率无特殊要求,只要能够降温至所需温度即可。在本发明中,所述初步模压成型和模压成型优选在真空的条件下进行,本发明对所述真空条件的真空度无特殊限定,只要能够除去模压成型环境中的空气即可。The present invention does not have a special limitation on the press forming device. In the embodiment of the present invention, the press forming is performed in a flat plate vulcanizer. The present invention preferably also includes performing preliminary molding on the laminated product before molding, and the pressure of the preliminary molding is preferably 5-15 MPa, more preferably 8-10 MPa; the time is preferably 0.8-1.2 h, more preferably is 1h. In the present invention, the preliminary compression molding can compact the mixed powder, which is beneficial to the subsequent compression molding. In the present invention, the compression molding preferably includes: performing high temperature compression molding and low temperature compression molding in sequence; the temperature of the high temperature compression molding is preferably 170-240°C, more preferably 200-220°C; the pressure is preferably 1-50MPa , more preferably 3-20MPa; time is preferably 0.5-10h, more preferably 3-6h. The temperature of the low temperature compression molding is preferably 110-130°C, more preferably 120-125°C; the pressure is preferably 1-50 MPa, more preferably 10-20 MPa; the time is preferably 0.5-72 h, more preferably 2.5-10 h. In the present invention, the temperature of the low temperature compression molding is preferably obtained by lowering the temperature on the basis of the high temperature compression molding temperature. The present invention has no special requirements on the cooling rate, as long as the temperature can be lowered to the desired temperature. In the present invention, the preliminary compression molding and compression molding are preferably performed under vacuum conditions, and the vacuum degree of the vacuum conditions is not particularly limited in the present invention, as long as the air in the compression molding environment can be removed.
在本发明中,所述模压成型后还优选包括将模压成型的产物降温后脱模。在本发明中,所述降温后温度优选为室温。本发明对所述降温的方式无特殊限定,采用本领域技术人员熟知的方式即可。In the present invention, after the compression molding, the compression molding product is preferably cooled and then demolded. In the present invention, the temperature after cooling is preferably room temperature. The present invention has no particular limitation on the cooling method, and a method well known to those skilled in the art may be adopted.
本发明在进行辐照交联之前优选将模压成型产物进行真空包装,所述真空包装用材料优选为铝塑袋。在本发明中,所述辐照交联优选包括高能电子束辐照交联、钴源衰变形成的γ射线辐照交联或X射线辐照交联,更优选为高能电子束辐照交联。在本发明中,所述高能电子束辐照交联用的电子束能量优选为1~20MeV,更优选为10~15MeV。在本发明中,所述辐照交联的辐照剂量优选为25~150kGy,更优选为75~100kGy;剂量率为5~50kGy/pass,更优选为20~40kGy/pass,更进一步优选为25~35kGy/pass。In the present invention, the molded product is preferably vacuum-packed before irradiation cross-linking, and the vacuum-packaging material is preferably an aluminum-plastic bag. In the present invention, the irradiation cross-linking preferably includes high-energy electron beam irradiation cross-linking, γ-ray irradiation cross-linking formed by cobalt source decay, or X-ray irradiation cross-linking, more preferably high-energy electron beam irradiation cross-linking . In the present invention, the electron beam energy used for the high-energy electron beam irradiation crosslinking is preferably 1-20 MeV, more preferably 10-15 MeV. In the present invention, the irradiation dose of the irradiation crosslinking is preferably 25-150kGy, more preferably 75-100kGy; the dose rate is 5-50kGy/pass, more preferably 20-40kGy/pass, still more preferably 25~35kGy/pass.
在本发明中,所述退火处理优选在保护气氛下进行,所述保护气氛优选包括氩气气氛或氮气气氛,更优选为氩气气氛。在本发明中,所述退火处理的温度优选为110~130℃,更优选为115~120℃;保温时间优选为0.5~72h,更优选为6~15h。在本发明中,所述退火处理能够使抗氧剂与自由基充分反应,消除多余自由基。In the present invention, the annealing treatment is preferably performed under a protective atmosphere, and the protective atmosphere preferably includes an argon atmosphere or a nitrogen atmosphere, more preferably an argon atmosphere. In the present invention, the temperature of the annealing treatment is preferably 110-130°C, more preferably 115-120°C; the holding time is preferably 0.5-72h, more preferably 6-15h. In the present invention, the annealing treatment can fully react the antioxidant with the free radicals and eliminate the redundant free radicals.
本发明可以根据实际需要涉及超高分子量聚乙烯板材的厚度,在本发明中,所述超高分子量聚乙烯板材的厚度优选为30mm~100mm,更优选为70~90mm。The present invention can relate to the thickness of the ultra-high molecular weight polyethylene sheet according to actual needs. In the present invention, the thickness of the ultra-high molecular weight polyethylene sheet is preferably 30-100 mm, more preferably 70-90 mm.
本发明还提供了上述技术方案所述制备方法制备得到的超高分子量聚乙烯板材,所述超高分子量聚乙烯板材中最高交联密度和最低交联密度的差值为0~100mol/m3。在本发明中,所述超高分子量聚乙烯板材具有均匀的交联密度,从而提高了超高分子量聚乙烯板材的拉伸强度、断裂伸长率和冲击强度。The present invention also provides an ultra-high molecular weight polyethylene sheet prepared by the preparation method described in the above technical solution, wherein the difference between the highest cross-linking density and the lowest cross-linking density in the ultra-high molecular weight polyethylene sheet is 0-100 mol/m 3 . . In the present invention, the ultra-high molecular weight polyethylene sheet has a uniform crosslinking density, thereby improving the tensile strength, elongation at break and impact strength of the ultra-high molecular weight polyethylene sheet.
本发明还提供了上述技术方案所述超高分子量聚乙烯板材在制备人工关节中的应用。在本发明中,所述人工关节优选包括人工髋关节或人工膝关节。The present invention also provides the application of the ultra-high molecular weight polyethylene sheet in the above technical solution in the preparation of artificial joints. In the present invention, the artificial joint preferably includes an artificial hip joint or an artificial knee joint.
为了进一步说明本发明,下面结合实施例对本发明提供的技术方案进行详细地描述,但不能将它们理解为对本发明保护范围的限定。In order to further illustrate the present invention, the technical solutions provided by the present invention are described in detail below with reference to the examples, but they should not be construed as limiting the protection scope of the present invention.
实施例1Example 1
将1.8g维生素E和1800g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在100r/min转速下球磨50min,得到维生素E质量百分含量为0.1%的混合粉料;Ball milling 1.8g vitamin E and 1800g ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120μm for 50min at a rotational speed of 100r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.1%;
将5.4g维生素E和1800g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在100r/min转速下球磨50min,得到维生素E质量百分含量为0.3%的混合粉料;5.4g of vitamin E and 1800g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120 μm are ball-milled for 50 minutes at a speed of 100r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.3%;
将900g、维生素E质量百分含量为0.3%的混合粉料平铺在模具的膜腔(尺寸为250mm*250mm*125mm)中,作为第1层;在第1层表面平铺1800g、维生素E质量百分含量为0.1%的混合粉料,作为第2层;在第2层表面平铺900g、维生素E质量百分含量为0.3%的混合粉料,作为第3层;Spread 900g of mixed powder with a mass percentage of vitamin E of 0.3% in the film cavity of the mold (size is 250mm*250mm*125mm) as the first layer; lay 1800g of vitamin E on the surface of the first layer. The mixed powder with a mass percentage of 0.1% is used as the second layer; 900g of mixed powder with a mass percentage of vitamin E of 0.3% is laid on the surface of the second layer as the third layer;
将层叠后得到产物置于平板硫化机中,抽真空后在80℃、10MPa的条件下进行初步模压成型1h;初步模压成型后升温至200℃,在6MPa的压力下进行高温模压成型6h;将温度降温至125℃,在10MPa的压力下进行低温模压成型2.5h,将低温模压成型的产物冷却室温脱膜;The product obtained after lamination was placed in a flat vulcanizer, vacuumed and then subjected to preliminary compression molding at 80 °C and 10 MPa for 1 hour; after preliminary compression molding, the temperature was raised to 200 °C, and high temperature compression molding was performed at a pressure of 6 MPa for 6 hours; The temperature is lowered to 125°C, and the low-temperature compression molding is carried out under the pressure of 10 MPa for 2.5 hours, and the low-temperature compression molding product is cooled to room temperature for release;
将脱膜后的产物进行铝塑袋真空包装,利用高能电子束(能量为10MeV)进行辐照交联,辐照剂量率为25kGy/pass,总剂量为75kGy;The product after stripping is vacuum-packed in an aluminum-plastic bag, and cross-linked by irradiation with a high-energy electron beam (energy of 10MeV), the irradiation dose rate is 25kGy/pass, and the total dose is 75kGy;
将辐照交联后的产物在氩气气氛中,110℃退火10h,得到超高分子量聚乙烯板材。The irradiated cross-linked product was annealed at 110° C. for 10 h in an argon atmosphere to obtain an ultra-high molecular weight polyethylene sheet.
实施例2Example 2
将1.2g维生素E和1200g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在200r/min转速下球磨40min,得到维生素E质量百分含量为0.1%的混合粉料;1.2g of vitamin E and 1200g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120 μm are ball-milled for 40 minutes at a rotational speed of 200r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.1%;
将2.4g维生素E和1200g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在200r/min转速下球磨40min,得到维生素E质量百分含量为0.2%的混合粉料;2.4g of vitamin E and 1200g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120 μm are ball-milled for 40 minutes at a speed of 200r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.2%;
将3.6g维生素E和1200g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在200r/min转速下球磨40min,得到维生素E质量百分含量为0.3%的混合粉料;3.6g of vitamin E and 1200g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120 μm are ball-milled for 40 minutes at a speed of 200r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.3%;
将600g、维生素E质量百分含量为0.3%的混合粉料平铺在模具的膜腔(尺寸为250mm*250mm*125mm)中,作为第1层;在第1层表面平铺600g、维生素E质量百分含量为0.2%的混合粉料,作为第2层;在第2层表面平铺1200g、维生素E质量百分含量为0.1%的混合粉料,作为第3层;在第3层表面平铺600g、维生素E质量百分含量为0.2%的混合粉料,作为第4层;在第4层表面平铺600g、维生素E质量百分含量为0.3%的混合粉料,作为第5层;Spread 600g of mixed powder with a mass percentage of vitamin E of 0.3% in the film cavity of the mold (size is 250mm*250mm*125mm) as the first layer; lay 600g of vitamin E on the surface of the first layer. The mixed powder with a mass percentage of 0.2% is used as the second layer; 1200g of mixed powder with a mass percentage of vitamin E of 0.1% is spread on the surface of the second layer as the third layer; on the surface of the third layer Spread 600g of mixed powder with a mass percentage of vitamin E of 0.2% as the fourth layer; spread 600g of mixed powder with a mass percentage of vitamin E of 0.3% on the surface of the fourth layer as the fifth layer ;
将层叠后得到产物置于平板硫化机中,抽真空后在80℃、10MPa的条件下进行初步模压成型1h;初步模压成型后升温至210℃,在6MPa的压力下进行高温模压成型4h;将温度降温至125℃,在10MPa的压力下进行低温模压成型2.5h,将低温模压成型的产物冷却室温脱膜;The product obtained after lamination was placed in a flat vulcanizer, vacuumed and then subjected to preliminary compression molding at 80 °C and 10 MPa for 1 hour; after preliminary compression molding, the temperature was raised to 210 °C, and high temperature compression molding was carried out under the pressure of 6 MPa for 4 hours; The temperature is lowered to 125°C, and the low-temperature compression molding is carried out under the pressure of 10 MPa for 2.5 hours, and the low-temperature compression molding product is cooled to room temperature for release;
将脱膜后的产物进行铝塑袋真空包装,利用高能电子束(能量为10MeV)进行辐照交联,辐照剂量率为25kGy/pass,总剂量为75kGy;The product after stripping is vacuum-packed in an aluminum-plastic bag, and cross-linked by irradiation with a high-energy electron beam (energy of 10MeV), the irradiation dose rate is 25kGy/pass, and the total dose is 75kGy;
将辐照交联后的产物在氩气气氛中,110℃退火10h,得到超高分子量聚乙烯板材。The irradiated cross-linked product was annealed at 110° C. for 10 h in an argon atmosphere to obtain an ultra-high molecular weight polyethylene sheet.
实施例2制备得到的超高分子量聚乙烯板材的结构示意图如图1所示。The schematic structural diagram of the ultra-high molecular weight polyethylene sheet prepared in Example 2 is shown in FIG. 1 .
实施例3Example 3
将2.7g维生素E和900g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在300r/min转速下球磨30min,得到维生素E质量百分含量为0.3%的混合粉料;Ball milling 2.7g of vitamin E and 900g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120μm for 30min at a rotational speed of 300r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.3%;
将2.25g维生素E和900g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在300r/min转速下球磨30min,得到维生素E质量百分含量为0.25%的混合粉料;Ball milling 2.25g vitamin E and 900g ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120μm for 30min at a rotational speed of 300r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.25%;
将1.35g维生素E和900g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在300r/min转速下球磨30min,得到维生素E质量百分含量为0.15%的混合粉料;Ball milling 1.35g vitamin E and 900g ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120μm for 30min at a rotational speed of 300r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.15%;
将0.9g维生素E和900g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在300r/min转速下球磨30min,得到维生素E质量百分含量为0.1%的混合粉料;0.9g of vitamin E and 900g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120 μm are ball-milled for 30 minutes at a rotational speed of 300r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.1%;
将450g、维生素E质量百分含量为0.15%的混合粉料平铺在模具的膜腔(尺寸为250mm*250mm*125mm)中,作为第1层;在第1层表面平铺450g、维生素E质量百分含量为0.25%的混合粉料,作为第2层;在第2层表面平铺450g、维生素E质量百分含量为0.15%的混合粉料,作为第3层;在第3层表面平铺900g、维生素E质量百分含量为0.1%的混合粉料,作为第4层;在第4层表面平铺450g、维生素E质量百分含量为0.15%的混合粉料,作为第5层;在第5层表面平铺450g、维生素E质量百分含量为0.25%的混合粉料,作为第6层;在第6层表面平铺450g、维生素E质量百分含量为0.3%的混合粉料,作为第7层;Spread 450g of mixed powder with a mass percentage of vitamin E of 0.15% in the film cavity of the mold (size is 250mm*250mm*125mm) as the first layer; lay 450g of vitamin E on the surface of the first layer. Mixed powder with a mass percentage of 0.25% is used as the second layer; 450g of mixed powder with a mass percentage of vitamin E of 0.15% is spread on the surface of the second layer as the third layer; on the surface of the third layer Spread 900g of mixed powder with a mass percentage of vitamin E of 0.1% as the fourth layer; spread 450g of mixed powder with a mass percentage of vitamin E of 0.15% on the surface of the fourth layer as the fifth layer ; Spread 450g of mixed powder with 0.25% vitamin E mass percentage on the surface of the 5th layer as the 6th layer; Spread 450g of mixed powder with 0.3% vitamin E mass percentage on the surface of the 6th layer material, as layer 7;
将层叠后得到产物置于平板硫化机中,抽真空后在80℃、10MPa的条件下进行初步模压成型1h;初步模压成型后升温至230℃,在6MPa的压力下进行高温模压成型2h;将温度降温至120℃,在15MPa的压力下进行低温模压成型2.5h,将低温模压成型的产物冷却室温脱膜;The product obtained after lamination was placed in a flat vulcanizer, vacuumed and then subjected to preliminary molding at 80 °C and 10 MPa for 1 h; after preliminary molding, the temperature was raised to 230 °C, and high-temperature molding was carried out under a pressure of 6 MPa for 2 h; The temperature was lowered to 120°C, and the low-temperature compression molding was carried out under the pressure of 15MPa for 2.5 hours, and the low-temperature compression molding product was cooled to room temperature for release;
将脱膜后的产物进行铝塑袋真空包装,利用高能电子束(能量为10MeV)进行辐照交联,辐照剂量率为20kGy/pass,总剂量为80kGy;The product after stripping is vacuum-packed in an aluminum-plastic bag, and cross-linked by irradiation with a high-energy electron beam (energy of 10MeV), the irradiation dose rate is 20kGy/pass, and the total dose is 80kGy;
将辐照交联后的产物在氩气气氛中,110℃退火10h,得到超高分子量聚乙烯板材。The irradiated cross-linked product was annealed at 110° C. for 10 h in an argon atmosphere to obtain an ultra-high molecular weight polyethylene sheet.
实施例4Example 4
将2.8g维生素E和800g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在400r/min转速下球磨20min,得到维生素E质量百分含量为0.35%的混合粉料;Ball milling 2.8g vitamin E and 800g ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120μm at a speed of 400r/min for 20min to obtain a mixed powder with a mass percentage of vitamin E of 0.35%;
将2.2g维生素E和800g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在400r/min转速下球磨20min,得到维生素E质量百分含量为0.275%的混合粉料;2.2g of vitamin E and 800g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120 μm are ball-milled for 20 minutes at a speed of 400r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.275%;
将1.4g维生素E和800g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在400r/min转速下球磨20min,得到维生素E质量百分含量为0.175%的混合粉料;1.4g of vitamin E and 800g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120 μm are ball-milled for 20 minutes at a speed of 400r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.175%;
将0.6g维生素E和800g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在400r/min转速下球磨20min,得到维生素E质量百分含量为0.075%的混合粉料;0.6g of vitamin E and 800g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120 μm are ball-milled for 20 minutes at a rotational speed of 400r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.075%;
将0.2g维生素E和400g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在400r/min转速下球磨20min,得到维生素E质量百分含量为0.05%的混合粉料0.2g of vitamin E and 400g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120μm were ball-milled for 20min at a speed of 400r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.05%
将400g、维生素E质量百分含量为0.35%的混合粉料平铺在模具的膜腔(尺寸为250mm*250mm*125mm)中,作为第1层;在第1层表面平铺400g、维生素E质量百分含量为0.275%的混合粉料,作为第2层;在第2层表面平铺400g、维生素E质量百分含量为0.175%的混合粉料,作为第3层;在第3层表面平铺400g、维生素E质量百分含量为0.075%的混合粉料,作为第4层;在第4层表面平铺400g、维生素E质量百分含量为0.05%的混合粉料,作为第5层;在第5层表面平铺400g、维生素E质量百分含量为0.075%的混合粉料,作为第6层;在第6层表面平铺400g、维生素E质量百分含量为0.175%的混合粉料,作为第7层;在第7层表面平铺400g、维生素E质量百分含量为0.275%的混合粉料,作为第8层;在第8层表面平铺400g、维生素E质量百分含量为0.35%的混合粉料,作为第9层;Spread 400g of mixed powder with a mass percentage of vitamin E of 0.35% in the film cavity of the mold (size is 250mm*250mm*125mm) as the first layer; lay 400g of vitamin E on the surface of the first layer. The mixed powder with a mass percentage of 0.275% is used as the second layer; 400g of mixed powder with a mass percentage of vitamin E of 0.175% is spread on the surface of the second layer as the third layer; on the surface of the third layer Spread 400g of mixed powder with a mass percentage of vitamin E of 0.075% as the fourth layer; spread 400g of mixed powder with a mass percentage of vitamin E of 0.05% on the surface of the fourth layer as the fifth layer ; Spread 400g of mixed powder with 0.075% vitamin E mass percentage on the surface of the 5th layer as the 6th layer; Spread 400g of mixed powder with 0.175% vitamin E mass percentage on the surface of the 6th layer material, as the seventh layer; 400g of mixed powder with 0.275% vitamin E mass percentage on the surface of the seventh layer, as the eighth layer; 400g, vitamin E mass percentage on the surface of the eighth layer. It is 0.35% mixed powder as the 9th layer;
将层叠后得到产物置于平板硫化机中,抽真空后在80℃、10MPa的条件下进行初步模压成型1h;初步模压成型后升温至190℃,在10MPa的压力下进行高温模压成型8h;将温度降温至125℃,在10MPa的压力下进行低温模压成型2.5h,将低温模压成型的产物冷却室温脱膜;The product obtained after lamination was placed in a flat vulcanizer, vacuumed and then subjected to preliminary molding at 80 °C and 10 MPa for 1 hour; after preliminary molding, the temperature was raised to 190 °C, and high-temperature molding was carried out at a pressure of 10 MPa for 8 hours; The temperature is lowered to 125°C, and the low-temperature compression molding is carried out under the pressure of 10 MPa for 2.5 hours, and the low-temperature compression molding product is cooled to room temperature for release;
将脱膜后的产物进行铝塑袋真空包装,利用高能电子束(能量为10MeV)进行辐照交联,辐照剂量率为30kGy/pass,总剂量为90kGy;The product after stripping is vacuum-packed in an aluminum-plastic bag, and cross-linked by irradiation with a high-energy electron beam (energy of 10MeV), the irradiation dose rate is 30kGy/pass, and the total dose is 90kGy;
将辐照交联后的产物在氩气气氛中,110℃退火72h,得到超高分子量聚乙烯板材。The irradiated cross-linked product was annealed at 110° C. for 72 hours in an argon atmosphere to obtain an ultra-high molecular weight polyethylene sheet.
实施例5Example 5
将3.6g维生素E和1200g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在500r/min转速下球磨10min,得到维生素E质量百分含量为0.3%的混合粉料;Ball milling 3.6g vitamin E and 1200g ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120μm for 10min at a rotational speed of 500r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.3%;
将7.2g维生素E和1200g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在500r/min转速下球磨10min,得到维生素E质量百分含量为0.6%的混合粉料;7.2g of vitamin E and 1200g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120 μm are ball-milled for 10 minutes at a rotational speed of 500r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.6%;
将10.8g维生素E和1200g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在500r/min转速下球磨10min,得到维生素E质量百分含量为0.9%的混合粉料;Ball milling 10.8g vitamin E and 1200g ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120μm for 10min at a rotational speed of 500r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.9%;
将600g、维生素E质量百分含量为0.9%的混合粉料平铺在模具的膜腔(尺寸为250mm*250mm*125mm)中,作为第1层;在第1层表面平铺600g、维生素E质量百分含量为0.6%的混合粉料,作为第2层;在第2层表面平铺600g、维生素E质量百分含量为0.3%的混合粉料,作为第3层;在第3层表面平铺600g、维生素E质量百分含量为0.6%的混合粉料,作为第4层;在第4层表面平铺600g、维生素E质量百分含量为0.9%的混合粉料,作为第5层;Spread 600g of mixed powder with a mass percentage of vitamin E of 0.9% in the film cavity of the mold (size is 250mm*250mm*125mm) as the first layer; lay 600g of vitamin E on the surface of the first layer. The mixed powder with a mass percentage of 0.6% is used as the second layer; 600g of mixed powder with a mass percentage of vitamin E of 0.3% is spread on the surface of the second layer as the third layer; on the surface of the third layer Spread 600g of mixed powder with 0.6% vitamin E mass percentage as the fourth layer; spread 600g of mixed powder with 0.9% vitamin E mass percentage on the surface of the fourth layer as the fifth layer ;
将层叠后得到产物置于平板硫化机中,抽真空后在80℃、10MPa的条件下进行初步模压成型1h;初步模压成型后升温至200℃,在6MPa的压力下进行高温模压成型6h;将温度降温至115℃,在30MPa的压力下进行低温模压成型4h,将低温模压成型的产物冷却室温脱膜;The product obtained after lamination was placed in a flat vulcanizer, vacuumed and then subjected to preliminary compression molding at 80 °C and 10 MPa for 1 hour; after preliminary compression molding, the temperature was raised to 200 °C, and high temperature compression molding was performed at a pressure of 6 MPa for 6 hours; The temperature was lowered to 115 °C, and the low-temperature compression molding was carried out under the pressure of 30 MPa for 4 hours, and the low-temperature compression molding product was cooled to room temperature for release;
将脱膜后的产物进行铝塑袋真空包装,利用高能电子束(能量为10MeV)进行辐照交联,辐照剂量率为40kGy/pass,总剂量为80kGy;The product after stripping is vacuum-packed in an aluminum-plastic bag, and cross-linked by irradiation with a high-energy electron beam (energy of 10MeV), the irradiation dose rate is 40kGy/pass, and the total dose is 80kGy;
将辐照交联后的产物在氩气气氛中,130℃退火0.5h,得到超高分子量聚乙烯板材。The irradiated cross-linked product was annealed at 130° C. for 0.5 h in an argon atmosphere to obtain an ultra-high molecular weight polyethylene sheet.
实施例6Example 6
将3.6g维生素E和1200g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在50r/min转速下球磨50min,得到维生素E质量百分含量为0.3%的混合粉料;Ball milling 3.6g vitamin E and 1200g ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120μm for 50min at a rotational speed of 50r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.3%;
将2.0g维生素E和1000g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在50r/min转速下球磨50min,得到维生素E质量百分含量为0.2%的混合粉料;Ball-milling 2.0g vitamin E and 1000g ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120μm for 50min at a rotational speed of 50r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.2%;
将1.2g维生素E和800g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在50r/min转速下球磨50min,得到维生素E质量百分含量为0.15%的混合粉料;1.2g of vitamin E and 800g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120 μm are ball-milled for 50 minutes at a speed of 50r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.15%;
将0.4g维生素E和800g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯在50r/min转速下球磨50min,得到维生素E质量百分含量为0.05%的混合粉料;0.4g of vitamin E and 800g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120 μm are ball-milled for 50 minutes at a rotational speed of 50r/min to obtain a mixed powder with a mass percentage of vitamin E of 0.05%;
将600g、维生素E质量百分含量为0.3%的混合粉料平铺在模具的膜腔(尺寸为250mm*250mm*125mm)中,作为第1层;在第1层表面平铺500g、维生素E质量百分含量为0.2%的混合粉料,作为第2层;在第2层表面平铺400g、维生素E质量百分含量为0.15%的混合粉料,作为第3层;在第3层表面平铺800g、维生素E质量百分含量为0.05%的混合粉料,作为第4层;在第4层表面平铺400g、维生素E质量百分含量为0.15%的混合粉料,作为第5层;在第5层表面平铺500g、维生素E质量百分含量为0.2%的混合粉料,作为第6层;在第6层表面平铺600g、维生素E质量百分含量为0.3%的混合粉料,作为第7层;Spread 600g of mixed powder with a mass percentage of vitamin E of 0.3% in the film cavity of the mold (size is 250mm*250mm*125mm) as the first layer; lay 500g of vitamin E on the surface of the first layer. The mixed powder with a mass percentage of 0.2% is used as the second layer; 400g of mixed powder with a mass percentage of vitamin E of 0.15% is spread on the surface of the second layer as the third layer; on the surface of the third layer Lay 800g of mixed powder with 0.05% vitamin E mass percentage as the 4th layer; spread 400g of mixed powder with 0.15% vitamin E mass percentage on the surface of the 4th layer as the 5th layer ; Spread 500g of mixed powder with 0.2% vitamin E mass percentage on the surface of the 5th layer as the 6th layer; Spread 600g of mixed powder with 0.3% vitamin E mass percentage on the surface of the 6th layer material, as layer 7;
将层叠后得到产物置于平板硫化机中,抽真空后在80℃、10MPa的条件下进行初步模压成型1h;初步模压成型后升温至200℃,在6MPa的压力下进行高温模压成型6h;将温度降温至125℃,在10MPa的压力下进行低温模压成型2.5h,将低温模压成型的产物冷却室温脱膜;The product obtained after lamination was placed in a flat vulcanizer, vacuumed and then subjected to preliminary compression molding at 80 °C and 10 MPa for 1 hour; after preliminary compression molding, the temperature was raised to 200 °C, and high temperature compression molding was performed at a pressure of 6 MPa for 6 hours; The temperature is lowered to 125°C, and the low-temperature compression molding is carried out under the pressure of 10 MPa for 2.5 hours, and the low-temperature compression molding product is cooled to room temperature for release;
将脱膜后的产物进行铝塑袋真空包装,利用高能电子束(能量为10MeV)进行辐照交联,辐照剂量率为25kGy/pass,总剂量为75kGy;The product after stripping is vacuum-packed in an aluminum-plastic bag, and cross-linked by irradiation with a high-energy electron beam (energy of 10MeV), the irradiation dose rate is 25kGy/pass, and the total dose is 75kGy;
将辐照交联后的产物在氩气气氛中,110℃退火10h,得到超高分子量聚乙烯板材。The irradiated cross-linked product was annealed at 110° C. for 10 h in an argon atmosphere to obtain an ultra-high molecular weight polyethylene sheet.
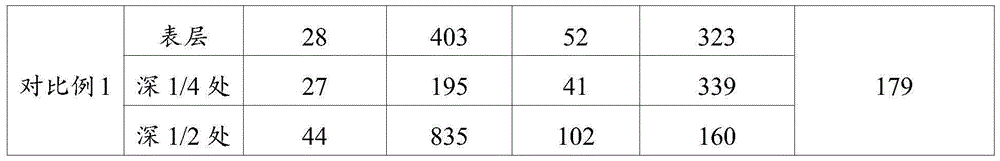
对比例1Comparative Example 1
将7.2g维生素E和3600g分子量为300万平均粒径为90~120μm的超高分子量聚乙烯进行球磨,得到维生素E质量百分含量为0.2%的混合粉料;Ball-milling 7.2 g of vitamin E and 3600 g of ultra-high molecular weight polyethylene with a molecular weight of 3 million and an average particle size of 90-120 μm to obtain a mixed powder with a mass percentage of vitamin E of 0.2%;
将所述混合粉料平铺在膜腔中,按照实施例1的方法将平铺后的产物依次进行初步模压成、模压成型、辐照交联和退火处理,得到超高分子量聚乙烯板材。The mixed powder is flattened in the film cavity, and the flattened product is sequentially subjected to preliminary molding, molding, irradiation crosslinking and annealing according to the method of Example 1 to obtain an ultra-high molecular weight polyethylene sheet.
测试例test case
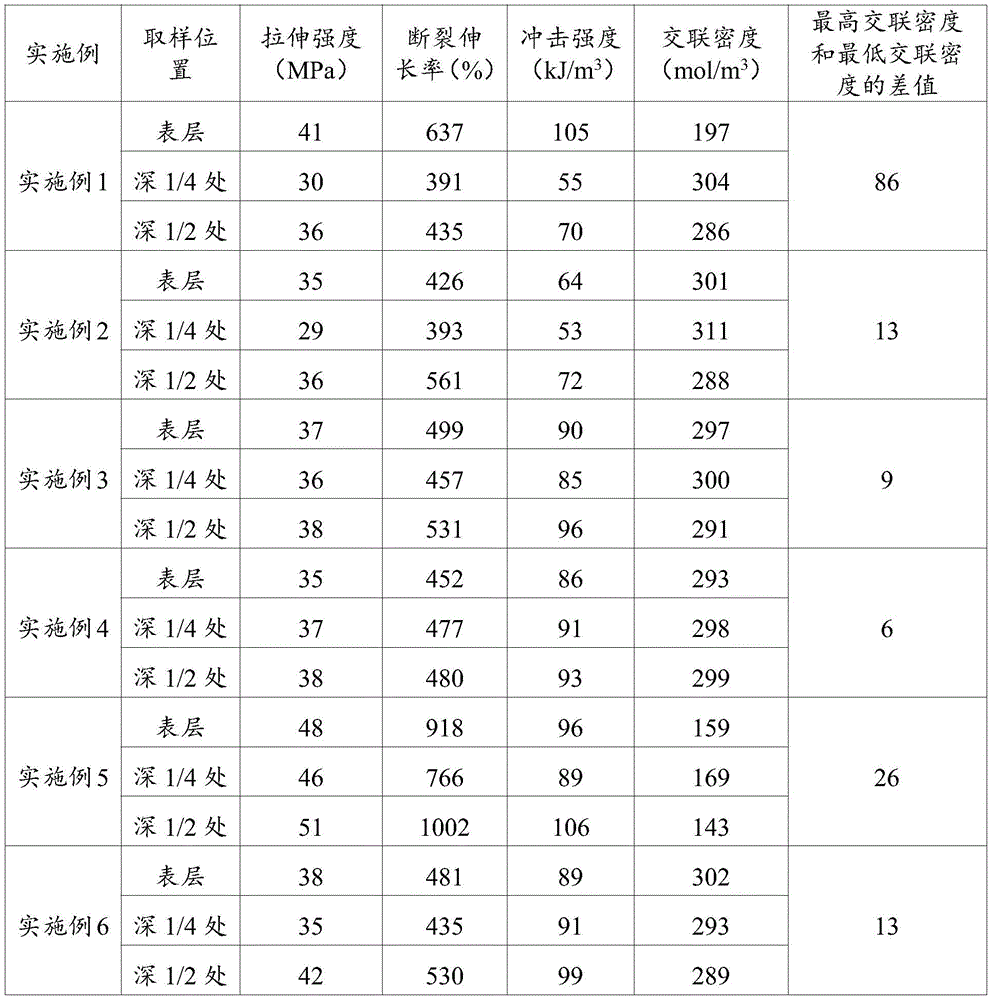
利用高精度雕铣设备对实施例1~6和对比例1制备得到的超高分子量聚乙烯板材进行取样,取样位置为超高分子量聚乙烯板材的表层、中心层(深度1/2处)和深度1/4处三个位置;将得到的取样样品按照ASTM D638标准进行拉伸性能测试,按照ASTM F648标准进行冲击性能测试,按照ASTM F2214测试材料的交联密度,其结果列于表1中。The ultra-high molecular weight polyethylene sheets prepared in Examples 1 to 6 and Comparative Example 1 were sampled by using high-precision engraving and milling equipment, and the sampling positions were the surface layer, central layer (1/2 depth) and Three positions at 1/4 of the depth; the obtained samples were tested for tensile properties according to ASTM D638, impact properties according to ASTM F648, and crosslinking density according to ASTM F2214. The results are listed in Table 1 .
表1实施例1~6和对比例1超高分子量聚乙烯板材的性能。Table 1 Properties of Examples 1 to 6 and Comparative Example 1 UHMWPE sheets.
从表1中数据可知,分层添加抗氧剂之后交联板材不同位置的交联密度分布均匀性提高了,实施例6中从中心到两侧每层混合粉料的总量不同,即每层厚度不同,但是保持抗氧剂浓度从两侧到中心逐层递减的趋势,也达到了交联密度分布均匀的效果。对比例1中制备得到的超高分子量聚乙烯板材中抗氧剂浓度分布均匀,中心出抗氧剂浓度高辐照强度较弱抑制了交联反应,表层辐照强度相对中心层大,交联密度高,整体交联密度不均一。From the data in Table 1, it can be seen that the uniformity of the crosslinking density distribution at different positions of the crosslinked sheet is improved after the antioxidant is added in layers. The layer thickness is different, but the trend of decreasing the antioxidant concentration from both sides to the center is maintained, which also achieves the effect of uniform crosslink density distribution. The ultra-high molecular weight polyethylene sheet prepared in Comparative Example 1 has a uniform distribution of antioxidant concentration, and the high radiation intensity of the antioxidant in the center inhibits the cross-linking reaction weakly. , the overall crosslink density is not uniform.
尽管上述实施例对本发明做出了详尽的描述,但它仅仅是本发明一部分实施例,而不是全部实施例,人们还可以根据本实施例在不经创造性前提下获得其他实施例,这些实施例都属于本发明保护范围。Although the above embodiment has made a detailed description of the present invention, it is only a part of the embodiments of the present invention, rather than all the embodiments. People can also obtain other embodiments according to the present embodiment without creativity. These embodiments All belong to the protection scope of the present invention.
Claims (6)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110067333.6A CN112870448B (en) | 2021-01-19 | 2021-01-19 | Ultrahigh molecular weight polyethylene plate and preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110067333.6A CN112870448B (en) | 2021-01-19 | 2021-01-19 | Ultrahigh molecular weight polyethylene plate and preparation method and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN112870448A CN112870448A (en) | 2021-06-01 |
| CN112870448B true CN112870448B (en) | 2022-08-26 |
Family
ID=76049629
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110067333.6A Active CN112870448B (en) | 2021-01-19 | 2021-01-19 | Ultrahigh molecular weight polyethylene plate and preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN112870448B (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN113995886B (en) * | 2021-10-28 | 2023-05-16 | 苏州微创关节医疗科技有限公司 | Artificial joint member, preparation method thereof and friction pair |
| CN114344568B (en) * | 2021-12-13 | 2023-03-17 | 苏州微创关节医疗科技有限公司 | Joint prosthesis with surface cross-linked layer, preparation method thereof and mould pressing assembly |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8933145B2 (en) * | 2009-02-20 | 2015-01-13 | The General Hospital Corporation | High temperature melting |
| CN102604188B (en) * | 2012-03-02 | 2014-02-05 | 中国科学院宁波材料技术与工程研究所 | Antioxidant cross-linked polymer and preparation method thereof |
-
2021
- 2021-01-19 CN CN202110067333.6A patent/CN112870448B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN112870448A (en) | 2021-06-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN112870448B (en) | Ultrahigh molecular weight polyethylene plate and preparation method and application thereof | |
| CN102276864B (en) | Surface crosslinked polyethylene | |
| JP5314423B2 (en) | Ultra high molecular weight polyethylene article and method of forming ultra high molecular weight polyethylene article | |
| CN102604188B (en) | Antioxidant cross-linked polymer and preparation method thereof | |
| US20140024736A1 (en) | Polymer articles having chemically bonded agents and methods of making the same | |
| JPH08509148A (en) | Non-oxidizing polymer medical implant | |
| US9889224B2 (en) | Spatial control of additives by high temperature | |
| US10220547B2 (en) | Peroxide cross-linking and high temperature melting | |
| CA2619937C (en) | Highly crystalline polyethylene | |
| KR102142720B1 (en) | A adhesion prevention film with excellent adhesion property | |
| JP5735443B2 (en) | Ultra high molecular weight polyethylene article and method of forming ultra high molecular weight polyethylene article | |
| CN107754013A (en) | The high crosslinked ultra-high-molecular-weight polyethylene artificial joint material of high antioxygen and preparation method | |
| JP5969637B2 (en) | Ultra high molecular weight polyethylene article and method of forming ultra high molecular weight polyethylene article | |
| CN111234279A (en) | A kind of water vapor barrier biodegradable polymer film and its preparation and application | |
| CN112757743A (en) | Guided tissue regeneration membrane and preparation method thereof | |
| CN114502208A (en) | Iodine infused ultra high molecular weight polyethylene | |
| CN115195004A (en) | Capacitor polypropylene film high-temperature breakdown performance improving method based on gamma ray irradiation crosslinking modification | |
| CN111073004B (en) | Method for improving irradiation crosslinking density and oxidation stability of ultrahigh molecular weight polyethylene joint material | |
| KR101631313B1 (en) | Multilayered radiation shielding thin-film composite materials made by uniform or multi-sized non-leaded tungsten particles on which polymers were synthesized for uniform dispersion of the particles in the same polymer or compatible polymer | |
| CN114058102B (en) | Wear-resistant and oxidation-resistant ultrahigh molecular weight polyethylene material and preparation method and application thereof | |
| CN101006124B (en) | Polymeric film | |
| CN116619786A (en) | A kind of ultra-high molecular weight polyethylene plate and its preparation method and application | |
| KR101325680B1 (en) | Hydrogels having metronidazole and manufacturing method therof | |
| US20220056222A1 (en) | Multi(meth)acrylate-functionalized resins as co-agents for crosslinking of thermoplastic polymers | |
| CN118852704A (en) | Ultra-high molecular weight polyethylene material and implant |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |