CN112823667B - Application of the composition in preventing or alleviating colic in infants and young children - Google Patents
Application of the composition in preventing or alleviating colic in infants and young children Download PDFInfo
- Publication number
- CN112823667B CN112823667B CN202010135666.3A CN202010135666A CN112823667B CN 112823667 B CN112823667 B CN 112823667B CN 202010135666 A CN202010135666 A CN 202010135666A CN 112823667 B CN112823667 B CN 112823667B
- Authority
- CN
- China
- Prior art keywords
- infants
- bifidobacterium
- composition
- lactalbumin
- casein
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/115—Fatty acids or derivatives thereof; Fats or oils
- A23L33/12—Fatty acids or derivatives thereof
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23C—DAIRY PRODUCTS, e.g. MILK, BUTTER OR CHEESE; MILK OR CHEESE SUBSTITUTES; MAKING OR TREATMENT THEREOF
- A23C9/00—Milk preparations; Milk powder or milk powder preparations
- A23C9/152—Milk preparations; Milk powder or milk powder preparations containing additives
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23C—DAIRY PRODUCTS, e.g. MILK, BUTTER OR CHEESE; MILK OR CHEESE SUBSTITUTES; MAKING OR TREATMENT THEREOF
- A23C9/00—Milk preparations; Milk powder or milk powder preparations
- A23C9/152—Milk preparations; Milk powder or milk powder preparations containing additives
- A23C9/1526—Amino acids; Peptides; Protein hydrolysates; Nucleic acids; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23C—DAIRY PRODUCTS, e.g. MILK, BUTTER OR CHEESE; MILK OR CHEESE SUBSTITUTES; MAKING OR TREATMENT THEREOF
- A23C9/00—Milk preparations; Milk powder or milk powder preparations
- A23C9/152—Milk preparations; Milk powder or milk powder preparations containing additives
- A23C9/1528—Fatty acids; Mono- or diglycerides; Petroleum jelly; Paraffine; Phospholipids; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/135—Bacteria or derivatives thereof, e.g. probiotics
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/17—Amino acids, peptides or proteins
- A23L33/19—Dairy proteins
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/40—Complete food formulations for specific consumer groups or specific purposes, e.g. infant formula
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/22—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin
- A61K31/23—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin of acids having a carboxyl group bound to a chain of seven or more carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/66—Microorganisms or materials therefrom
- A61K35/74—Bacteria
- A61K35/741—Probiotics
- A61K35/744—Lactic acid bacteria, e.g. enterococci, pediococci, lactococci, streptococci or leuconostocs
- A61K35/745—Bifidobacteria
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/1703—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- A61K38/1709—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Polymers & Plastics (AREA)
- Food Science & Technology (AREA)
- Mycology (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Nutrition Science (AREA)
- General Chemical & Material Sciences (AREA)
- Epidemiology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Microbiology (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Molecular Biology (AREA)
- Biophysics (AREA)
- Gastroenterology & Hepatology (AREA)
- Biochemistry (AREA)
- Pain & Pain Management (AREA)
- Rheumatology (AREA)
- Zoology (AREA)
- Marine Sciences & Fisheries (AREA)
- Immunology (AREA)
- Emergency Medicine (AREA)
- Pediatric Medicine (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Dairy Products (AREA)
- Coloring Foods And Improving Nutritive Qualities (AREA)
Abstract
本发明属于食品或医药领域,具体公开了组合物在制备预防、减少或缓解婴幼儿肠绞痛的食品、预防或治疗婴幼儿肠绞痛的药物或者改善婴幼儿上火症状和/或婴幼儿上火行为表现的食品或药物中的用途;其中,所述组合物包含棕榈酸甘油酯、a‑乳清蛋白、β‑酪蛋白以及选自乳双歧杆菌、动物双歧杆菌、长双岐杆菌、短双歧杆菌、青春双歧杆菌、两歧双歧杆菌和婴儿双歧杆菌中至少一种双歧杆菌;其中,按照棕榈酸计算,棕榈酸甘油酯中含有15%以上重量的Sn‑2棕榈酸甘油酯。
The invention belongs to the field of food or medicine, and specifically discloses that the composition can be used in the preparation of food for preventing, reducing or alleviating intestinal colic in infants, medicine for preventing or treating intestinal colic in infants, or improving the symptoms of getting angry in infants and/or infants. The use in foods or medicines of lit behavior; wherein, the composition comprises palmitic acid glycerides, α-lactalbumin, β-casein and selected from Bifidobacterium lactis, Bifidobacterium animalis, Bifidobacterium longum Bacillus, Bifidobacterium breve, Bifidobacterium adolescentis, Bifidobacterium bifidum, and Bifidobacterium infantis; wherein, calculated as palmitic acid, glyceryl palmitate contains more than 15% by weight of Sn‑ 2 glyceryl palmitate.
Description
技术领域technical field
本发明属于食品或医药领域,具体涉及组合物在预防或缓解婴幼儿肠绞痛中的应用。The invention belongs to the field of food or medicine, and specifically relates to the application of the composition in preventing or alleviating intestinal colic in infants and young children.
背景技术Background technique
据统计,哭闹占新生儿儿科问诊咨询的20%,其中很大比例的婴儿在出生后3个月内会出现肠绞痛等腹部不适症状,且在不同性别、不同喂养方式的婴儿中肠绞痛的发生率相近。肠绞痛通常是良性的且从4个月以后伴随婴儿长大逐渐消失。虽然婴儿肠绞痛及其所引发的婴儿夜间哭闹是自我限制和良性病因,但这经常导致父母和护理人员筋疲力尽甚至罹患抑郁疾病。因此,亟需寻找一种有效预防或缓解婴幼儿肠绞痛的方法或产品。According to statistics, crying accounts for 20% of newborn pediatric consultations, and a large proportion of babies will experience abdominal discomfort symptoms such as colic within 3 months after birth, and among babies of different sexes and different feeding methods Colic occurred at a similar rate. Colic is usually benign and gradually disappears as the baby grows from 4 months onwards. Although infantile colic and its resulting night-time crying are self-limiting and benign, it often leads to exhaustion and even depressive illness for parents and caregivers. Therefore, there is an urgent need to find a method or product for effectively preventing or alleviating colic in infants.
生命早期,肠道和大脑都经历快速变化,这也是肠道微生物定植和形成的关键时期。已有研究发现,一系列因素影响生命早期的肠道微生物群组成,包括出生胎龄、生产方式、喂养方式、遗传背景、地理环境、乳母饮食和抗生素使用。足月生产、自然顺产以及出生后6个月坚持纯母乳喂养是早期肠道微生物群建立和成熟的有利因素,可降低一些急慢性疾病的风险,如肥胖、1型糖尿病、过敏和炎性肠病等。目前已有一些研究指出肠绞痛婴儿的肠道菌群与正常婴儿不同。Early in life, when both the gut and the brain undergo rapid changes, this is also a critical period for the colonization and formation of gut microbes. A range of factors have been found to influence gut microbiota composition early in life, including gestational age at birth, mode of delivery, feeding regime, genetic background, geographic environment, nursing mother's diet, and antibiotic use. Full-term delivery, natural vaginal delivery, and exclusive breastfeeding for 6 months after birth are favorable factors for the establishment and maturation of early gut microbiota, which can reduce the risk of some acute and chronic diseases, such as obesity,
母乳脂肪为婴儿早期生长提供45%~60%的能量,母乳脂肪中98%以上是甘油三酯。母乳中的不同脂肪酸与甘油酯化的位置存在差异;其中,母乳中的不饱和脂肪酸如亚油酸、α-亚麻酸多位于甘油三酯的1位和3位;母乳中的长链饱和脂肪酸如棕榈酸则主要位于2位,这样形成的棕榈酸甘油三酯称为Sn-2棕榈酸甘油三酯。在消化道,婴儿胃部的脂肪水解酶主要作用于甘油三酯的1位和3位酯键,因此,不饱和脂肪酸首先游离,继而在十二指肠中与Sn-2棕榈酸甘油单酯一同被降解吸收。但是,普通婴幼儿配方粉中含有棕榈油,其长链饱和脂肪酸大多酯化在甘油三酯1位和3位酯键,水解后易与钙离子结合形成钙皂,降低了脂肪和矿物质的吸收,同时难吸收的钙皂还可能使粪便发硬导致排便困难。国标GB14880容许在婴幼儿配方粉中添加OPO结构酯,这是一种富含Sn-2棕榈酸甘油三酯的食品原料,添加OPO结构酯可提高产品中的Sn-2棕榈酸甘油三酯占比。Breast milk fat provides 45% to 60% of the energy for early growth of infants, and more than 98% of breast milk fat is triglyceride. There are differences between different fatty acids in breast milk and glycerol esterification positions; among them, unsaturated fatty acids in breast milk such as linoleic acid and α-linolenic acid are mostly located in the 1st and 3rd positions of triglycerides; long-chain saturated fatty acids in breast milk For example, palmitic acid is mainly located at the 2 position, and the palmitic triglyceride formed in this way is called Sn-2 palmitic triglyceride. In the digestive tract, the lipolytic enzymes in the baby's stomach mainly act on the 1- and 3-position ester bonds of triglycerides. Therefore, unsaturated fatty acids are first freed, and then combined with Sn-2 monoglyceride palmitate in the duodenum be degraded and absorbed together. However, ordinary infant formula powder contains palm oil, and most of its long-chain saturated fatty acids are esterified in the 1- and 3-position ester bonds of triglycerides. After hydrolysis, it is easy to combine with calcium ions to form calcium soap, which reduces the fat and mineral content. At the same time, calcium soaps that are difficult to absorb may also make the stool hard and cause difficulty in defecation. The national standard GB14880 allows the addition of OPO structural esters in infant formula powder, which is a food raw material rich in Sn-2 palmitic acid triglycerides. Adding OPO structural esters can increase the proportion of Sn-2 palmitic triglycerides in the product. Compare.
母乳富含a-乳清蛋白,它是一种富含必需氨基酸的蛋白质,经消化生成多种生物活性多肽。有研究发现,a-乳清蛋白消化产物可在体外抑制大肠杆菌、肺炎球菌、金黄色葡萄球菌及念珠菌等致病菌。Breast milk is rich in a-lactalbumin, a protein rich in essential amino acids, which can be digested to produce a variety of biologically active peptides. Studies have found that a-lactalbumin digestion products can inhibit pathogenic bacteria such as Escherichia coli, pneumococcus, Staphylococcus aureus and Candida in vitro.
β-酪蛋白是母乳中含量最高的酪蛋白分子,经消化产生的多肽片段具有抑制有害菌生长的作用。β-casein is the most abundant casein molecule in breast milk, and the polypeptide fragments produced by digestion can inhibit the growth of harmful bacteria.
益生菌是肠道微生物群中的重要组成。世界粮农组织(FAO)和世界卫生组织(WHO)对于益生菌的定义是:通过摄取适当数量、能够对于食用者的身体健康发挥有效作用的活菌。双歧杆菌属是益生菌的一种,目前常见的双歧杆菌菌株包括乳双歧杆菌、动物双歧杆菌、长双岐杆菌、短双歧杆菌、青春双歧杆菌、两歧双歧杆菌和婴儿双歧杆菌等,以及一些新型菌株。Probiotics are an important component of the gut microbiota. The World Food and Agriculture Organization (FAO) and the World Health Organization (WHO) define probiotics as: live bacteria that can exert an effective effect on the health of the eater through ingestion of an appropriate amount. Bifidobacterium genus is a kind of probiotics. Currently, common bifidobacterium strains include Bifidobacterium lactis, Bifidobacterium animalis, Bifidobacterium longum, Bifidobacterium breve, Bifidobacterium adolescentis, Bifidobacterium bifidum and Bifidobacterium infantis, etc., and some new strains.
发明内容Contents of the invention
本发明提供了组合物在预防或缓解婴幼儿肠绞痛,或者改善婴幼儿上火症状和/或上火行为表现中的应用。The invention provides the application of the composition in preventing or relieving colic in infants, or improving the symptoms and/or angry behaviors of infants.
本发明涉及组合物在制备预防、减少或缓解婴幼儿肠绞痛的食品、预防或治疗婴幼儿肠绞痛的药物或者改善婴幼儿上火症状和/或婴幼儿上火行为表现的食品或药物中的用途;其中,所述组合物包含棕榈酸甘油酯、a-乳清蛋白、β-酪蛋白以及选自乳双歧杆菌、动物双歧杆菌、长双岐杆菌、短双歧杆菌、青春双歧杆菌、两歧双歧杆菌和婴儿双歧杆菌中至少一种双歧杆菌;其中,按照棕榈酸计算,棕榈酸甘油酯中含有15%以上(例如18%以上、20%以上、15%~98%、15%~90%、20%~80%、20%~95%、30%、40%、50%、60%、70%)重量的Sn-2棕榈酸甘油酯。The present invention relates to the composition in the preparation of food for preventing, reducing or alleviating intestinal colic in infants, the medicine for preventing or treating intestinal colic in infants, or the food or medicine for improving the symptoms and/or behaviors of infants getting angry wherein, the composition comprises glyceryl palmitate, α-lactalbumin, β-casein and selected from Bifidobacterium lactis, Bifidobacterium animalis, Bifidobacterium longum, Bifidobacterium breve, youth At least one kind of Bifidobacterium bifidobacterium, Bifidobacterium bifidum and Bifidobacterium infantis; wherein, calculated according to palmitic acid, glyceryl palmitate contains more than 15% (such as more than 18%, more than 20%, 15% ~98%, 15%~90%, 20%~80%, 20%~95%, 30%, 40%, 50%, 60%, 70%) weight of Sn-2 glyceryl palmitate.
本发明的一些实施方式中,棕榈酸甘油酯包括Sn-2棕榈酸甘油酯以及可选的除Sn-2棕榈酸甘油酯以外的其它棕榈酸甘油酯。In some embodiments of the present invention, glyceryl palmitate includes glyceryl palmitate Sn-2 and optionally glyceryl palmitate other than Sn-2 glyceryl palmitate.
本发明的一些实施方式中,a-乳清蛋白、β-酪蛋白以及Sn-2棕榈酸甘油酯之间的重量比为(1-10):(1-10):(1-10),例如1:(1-2):(1-2)、1:(1-5):(1-5)、1:(1-7):(1-7)、1:(1-6):(1-6)、1:1.32:1.2、1:1:1、1:3:3、1:5:5、1:8:8。In some embodiments of the present invention, the weight ratio between α-lactalbumin, β-casein and Sn-2 glyceryl palmitate is (1-10):(1-10):(1-10), For example 1:(1-2):(1-2), 1:(1-5):(1-5), 1:(1-7):(1-7), 1:(1-6) :(1-6), 1:1.32:1.2, 1:1:1, 1:3:3, 1:5:5, 1:8:8.
本发明的一些实施方式中,双歧杆菌的活菌数按照每克a-乳清蛋白计为107~1013CFU,例如108CFU、109CFU、1010CFU、1011CFU、1012CFU。In some embodiments of the present invention, the number of viable bifidobacteria is 10 7 to 10 13 CFU per gram of a-lactalbumin, such as 10 8 CFU, 10 9 CFU, 10 10 CFU, 10 11 CFU, 10 12 CFU.
本发明的一些实施方式中,双歧杆菌选自双歧杆菌BB12和双歧杆菌HN019。In some embodiments of the present invention, the Bifidobacterium is selected from Bifidobacterium BB12 and Bifidobacterium HN019.
本发明的一些实施方式中,双歧杆菌BB12与双歧杆菌HN019的活菌数比例为(1:100)~(100:1),例如100:1、90:1、80:1、70:1、60:1、50:1、40:1、30:1、20:1、10:1、5:1、1:1、1:5、1:8、1:10、1:14、1:17、1:20、1:30、1:40、1:50、1:60、1:70、1:80、1:90。In some embodiments of the present invention, the ratio of the viable count of Bifidobacterium BB12 to Bifidobacterium HN019 is (1:100)~(100:1), such as 100:1, 90:1, 80:1, 70: 1, 60:1, 50:1, 40:1, 30:1, 20:1, 10:1, 5:1, 1:1, 1:5, 1:8, 1:10, 1:14, 1:17, 1:20, 1:30, 1:40, 1:50, 1:60, 1:70, 1:80, 1:90.
本发明的一些实施方式中,所述组合物还包含除棕榈酸甘油酯以外的脂肪。In some embodiments of the present invention, the composition further comprises fats other than glyceryl palmitate.
本发明的一些实施方式中,除棕榈酸甘油酯以外的脂肪为本领域常用脂肪。In some embodiments of the present invention, fats other than glyceryl palmitate are commonly used fats in the art.
本发明的一些实施方式中,除棕榈酸甘油酯以外的脂肪选自亚油酸甘油酯、亚麻酸甘油酯、二十二碳六烯酸甘油酯和花生四烯酸甘油酯。In some embodiments of the present invention, the fat other than glyceryl palmitate is selected from glyceryl linoleate, glyceryl linolenate, glyceryl docosahexaenoate and glyceryl arachidonic acid.
本发明的一些实施方式中,组合物中,按照脂肪酸计算,棕榈酸甘油酯重量占脂肪总重量的1%~96%,例如10%~90%、20%~80%、30%~70%、20%~90%、10%~50%、5%~40%、5%~60%。其中,脂肪是由脂肪酸和甘油结合形成。In some embodiments of the present invention, in the composition, based on fatty acid calculation, the weight of glyceryl palmitate accounts for 1% to 96% of the total weight of fat, such as 10% to 90%, 20% to 80%, and 30% to 70%. , 20% to 90%, 10% to 50%, 5% to 40%, 5% to 60%. Among them, fat is formed by combining fatty acids and glycerol.
本发明的一些实施方式中,所述组合物还包含除a-乳清蛋白和β-酪蛋白以外的蛋白质。In some embodiments of the present invention, the composition further comprises proteins other than α-lactalbumin and β-casein.
本发明的一些实施方式中,除a-乳清蛋白和β-酪蛋白以外的蛋白质为本领域常用的蛋白质。In some embodiments of the present invention, proteins other than α-lactalbumin and β-casein are proteins commonly used in the art.
本发明的一些实施方式中,除a-乳清蛋白和β-酪蛋白以外的蛋白质选自卵白蛋白、卵磷蛋白、白蛋白、肌蛋白、大豆蛋白、麦谷蛋白、谷蛋白、麦胶蛋白、玉米胶蛋白、胶质蛋白、胶原蛋白、豆球蛋白以及除a-乳清蛋白和β-酪蛋白以外的乳蛋白。In some embodiments of the present invention, the protein other than α-lactalbumin and β-casein is selected from the group consisting of ovalbumin, lecithin, albumin, myosin, soybean protein, glutenin, glutenin, gliadin, Zein, glial, collagen, legumin, and milk proteins other than a-lactalbumin and beta-casein.
本发明的一些实施方式中,所述组合物还包含碳水化合物。In some embodiments of the invention, the composition further comprises carbohydrates.
本发明的一些实施方式中,所述碳水化合物为本领域常用碳水化合物。In some embodiments of the present invention, the carbohydrates are commonly used carbohydrates in the field.
本发明的一些实施方式中,所述碳水化合物选自葡萄糖、果糖、半乳糖、蔗糖、乳糖、麦芽糖、淀粉、纤维素、半纤维素和果胶。In some embodiments of the present invention, the carbohydrate is selected from glucose, fructose, galactose, sucrose, lactose, maltose, starch, cellulose, hemicellulose and pectin.
本发明的一些实施方式中,所述组合物还包含药用辅料及任选的用于预防或缓解婴幼儿肠绞痛、或者改善婴幼儿上火症状和/或婴幼儿上火行为表现的药物活性成分,或者所述组合物还包含食品添加剂。In some embodiments of the present invention, the composition also includes pharmaceutical excipients and optional medicines for preventing or alleviating colic in infants, or improving the symptoms and/or behaviors of infants getting angry active ingredient, or the composition also contains food additives.
本发明的一些实施方式中,药用辅料为本领域常用药用辅料。In some embodiments of the present invention, the pharmaceutical excipients are commonly used pharmaceutical excipients in this field.
本发明的一些实施方式中,药用辅料可选自溶剂、抛射剂、增溶剂、助溶剂、乳化剂、着色剂、黏合剂、崩解剂、填充剂、润滑剂、润湿剂、渗透压调节剂、稳定剂、助流剂、矫味剂、防腐剂、助悬剂、包衣材料、芳香剂、抗黏合剂、整合剂、渗透促进剂、pH值调节剂、缓冲剂、增塑剂、表面活性剂、发泡剂、消泡剂、增稠剂、包合剂、保湿剂、吸收剂、稀释剂、絮凝剂与反絮凝剂、助滤剂和释放阻滞剂。In some embodiments of the present invention, pharmaceutical excipients can be selected from solvents, propellants, solubilizers, cosolvents, emulsifiers, colorants, binders, disintegrants, fillers, lubricants, wetting agents, osmotic pressure Regulators, stabilizers, glidants, flavoring agents, preservatives, suspending agents, coating materials, fragrances, anti-adhesive agents, integrating agents, penetration enhancers, pH regulators, buffers, plasticizers , surfactants, foaming agents, defoamers, thickeners, inclusion agents, humectants, absorbents, diluents, flocculants and deflocculants, filter aids and release retardants.
本发明的一些实施方式中,食品添加剂可选自营养强化剂、抗氧化剂、增味剂、甜味剂、增稠剂、防腐剂、抗结剂和酸度调节剂。In some embodiments of the present invention, food additives can be selected from nutritional fortifiers, antioxidants, flavor enhancers, sweeteners, thickeners, preservatives, anticaking agents and acidity regulators.
本发明的一些实施方式中,营养强化剂可选自维生素(例如维生素A、B1、B6、B12、C、D、E)、矿物质(例如镁、磷、钙、铁、锌、硒或它们的衍生物等)、膳食纤维、牛磺酸和胆碱。In some embodiments of the present invention, nutritional supplements can be selected from vitamins (such as vitamin A, B 1 , B 6 , B 12 , C, D, E), minerals (such as magnesium, phosphorus, calcium, iron, zinc, Selenium or their derivatives, etc.), dietary fiber, taurine and choline.
本发明的一些实施方式中,食品为乳制品。In some embodiments of the invention, the food product is a dairy product.
本发明的一些实施方式中,每百克乳制品中含0.5~16g a-乳清蛋白,例如1g、2g、3g、4g、5g、6g、7g、8g、9g、10g、11g、12g、13g、14g、15g。In some embodiments of the present invention, every 100 grams of dairy products contains 0.5-16 g of a-lactalbumin, such as 1 g, 2 g, 3 g, 4 g, 5 g, 6 g, 7 g, 8 g, 9 g, 10 g, 11 g, 12 g, 13 g , 14g, 15g.
本发明的一些实施方式中,每百克乳制品中含0.8~20gβ-酪蛋白,例如1g、2g、3g、4g、5g、6g、7g、8g、9g、10g、11g、12g、13g、14g、15g、16g、17g、18g、19g。In some embodiments of the present invention, every 100 grams of dairy products contains 0.8-20 g of β-casein, such as 1 g, 2 g, 3 g, 4 g, 5 g, 6 g, 7 g, 8 g, 9 g, 10 g, 11 g, 12 g, 13 g, 14 g , 15g, 16g, 17g, 18g, 19g.
本发明的一些实施方式中,每百克乳制品中双歧杆菌的活菌数为106~1015CFU,例如107CFU、108CFU、109CFU、1010CFU、1011CFU、1012CFU、1013CFU、1014CFU。In some embodiments of the present invention, the number of viable bifidobacteria per 100 grams of dairy products is 10 6 to 10 15 CFU, such as 10 7 CFU, 10 8 CFU, 10 9 CFU, 10 10 CFU, 10 11 CFU, 10 12 CFU, 10 13 CFU, 10 14 CFU.
本发明的一些实施方式中,婴幼儿上火症状选自面色赤红、眼睛红肿、眼屎多、口唇干燥、口臭、有鼻涕渣或黄鼻涕、手脚心发热、尿黄、舌苔增厚和大便发硬。In some embodiments of the present invention, the symptoms of getting angry in infants and young children are selected from the group consisting of red complexion, red and swollen eyes, excess eye mucus, dry lips, bad breath, mucus or yellow nose, hot hands and feet, yellow urine, thickened tongue coating, and hard stools .
本发明的一些实施方式中,婴幼儿上火行为表现选自夜间醒来哭闹、脾气急躁、爱生气、白天哭闹、比平时容易饿、比平时吃得少、爱蹬被和不爱穿衣物。In some embodiments of the present invention, the behavior of infants and young children getting angry is selected from the group consisting of waking up at night and crying, short-tempered, irritable, crying during the day, hungry more than usual, eating less than usual, fondling of quilts and not fond of wearing clothes. clothing.
本发明中,乳制品选自杀菌乳、灭菌乳、复原乳、酸奶、优酪乳、乳粉、配方乳粉、炼乳、干酪、干酪素、乳清粉、乳脂肪和含乳饮料中至少一种,优选为(婴幼儿)配方乳粉。In the present invention, dairy products are selected from sterilized milk, sterilized milk, reconstituted milk, yogurt, yogurt, milk powder, formula milk powder, condensed milk, cheese, casein, whey powder, milk fat and milk-containing beverages, at least One, preferably (infant) formula milk powder.
本发明中,如无特别说明,其中:Among the present invention, if no special instructions, wherein:
术语“棕榈酸甘油酯”是指甘油基上连接至少一个棕榈酸的脂肪酸甘油酯,可选自单酯、二酯和三酯,其中,二酯、三酯的甘油基上也可连接其它脂肪酸基团。The term "glyceryl palmitate" refers to fatty acid glycerides with at least one palmitic acid attached to the glyceryl group, which can be selected from monoesters, diesters, and triesters, wherein other fatty acids can also be attached to the glyceryl groups of the diesters and triesters group.
术语“Sn-2棕榈酸”是指与脂肪的甘油基上的Sn-2位连接的棕榈酸。The term "Sn-2 palmitic acid" refers to palmitic acid attached to the Sn-2 position on a glyceryl group of a fat.
术语“Sn-2棕榈酸甘油酯”是指甘油基上的Sn-2位连接棕榈酸的脂肪酸甘油酯,可以选自Sn-2棕榈酸甘油单酯、Sn-2棕榈酸甘油二酯和Sn-2棕榈酸甘油三酯;其中,Sn-2棕榈酸甘油二酯和Sn-2棕榈酸甘油三酯中甘油基上的Sn-1位和/或Sn-3位可连接任意脂肪酸,包括但不限于棕榈酸、丁酸、己酸、辛酸、癸酸、十八酸、十二酸、十四酸、二十酸、豆蔻油酸、棕榈油酸、菜籽油酸、亚油酸、亚麻酸等。The term "Sn-2 palmitic acid glyceride" refers to the fatty acid glyceride of palmitic acid connected to the Sn-2 position on the glyceryl group, which can be selected from Sn-2 palmitic acid monoglyceride, Sn-2 palmitic acid diglyceride and Sn -2 palmitic acid triglyceride; Wherein, the Sn-1 position and/or Sn-3 position on the glyceryl group in Sn-2 palmitic acid triglyceride and Sn-2 palmitic acid triglyceride can be connected to any fatty acid, including but Not limited to palmitic acid, butyric acid, caproic acid, caprylic acid, capric acid, octadecanoic acid, dodecanoic acid, myristic acid, eicosic acid, myristic acid, palmitoleic acid, rapeseed acid, linoleic acid, linoleic acid Acid etc.
术语“a-乳清蛋白”是从牛奶中提取的一种蛋白质,有营养价值高、易消化吸收、含有多种活性成分等特点,是人体优质蛋白质补充剂之一。The term "a-whey protein" is a protein extracted from milk. It has the characteristics of high nutritional value, easy digestion and absorption, and contains a variety of active ingredients. It is one of the high-quality protein supplements for human body.
术语“β-酪蛋白”是由乳腺腺泡上皮细胞合成的磷酸化蛋白质,广泛存在于哺乳动物(牛、牦牛、山羊、马、兔等)和人的乳汁中。The term "β-casein" is a phosphorylated protein synthesized by mammary acinar epithelial cells, widely present in the milk of mammals (cattle, yak, goat, horse, rabbit, etc.) and human.
本发明取得的有益效果:The beneficial effect that the present invention obtains:
本发明提供了组合物在预防、减少或缓解婴幼儿肠绞痛,或者在改善婴幼儿上火症状和/或婴幼儿上火行为表现中的应用。The present invention provides the application of the composition in preventing, reducing or alleviating intestinal colic in infants, or in improving the symptoms and/or behaviors of infants getting angry.
附图说明Description of drawings
为了使本发明的内容更容易被清楚的理解,下面根据本发明的具体实施例并结合附图,对本发明作进一步详细的说明,其中In order to make the content of the present invention more easily understood, the present invention will be described in further detail below according to specific embodiments of the present invention in conjunction with the accompanying drawings, wherein
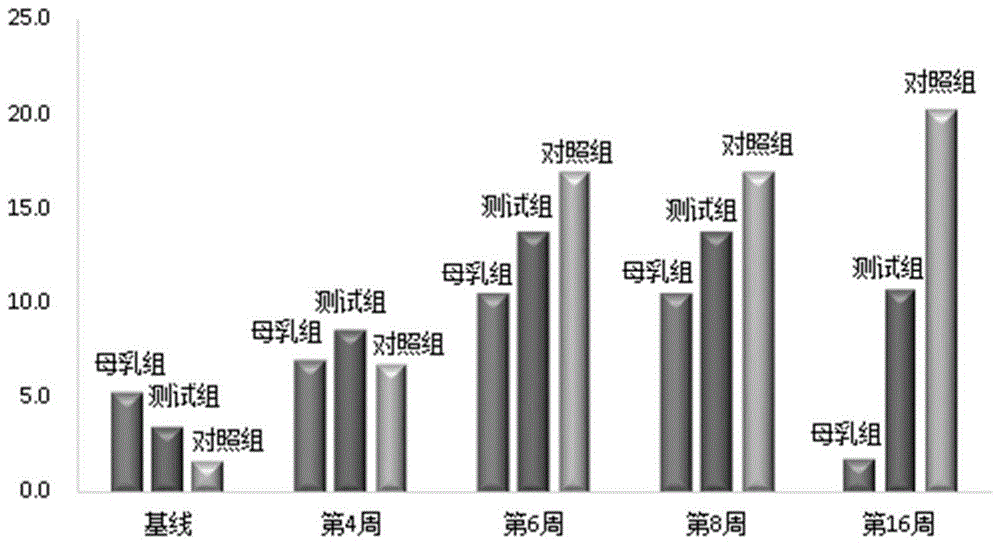
图1为测试组、对照组和母乳组婴儿不同时间点的肠绞痛发生率示意图;Figure 1 is a schematic diagram of the incidence of colic among infants in the test group, control group and breast milk group at different time points;
图2为测试组、对照组和母乳组婴儿在各时间点上火症状及行为表现总分与在基线上火症状及行为表现总分之差的平均分的示意图;Figure 2 is a schematic diagram of the average score of the difference between the total score of the symptoms and behavioral performance of the test group, the control group and the breast milk group at each time point and the total score of the symptoms and behavioral performance at the baseline;
图3为测试组、对照组和母乳组婴儿在各时间点上火行为表现得分与在基线上火行为表现得分之差的平均分的示意图;Figure 3 is a schematic diagram of the average score of the difference between the performance scores of angry behaviors and the scores of angry behaviors at baseline for infants in the test group, the control group and the breast milk group at each time point;
图4为测试组、对照组和母乳组婴儿在各时间点夜间醒来哭闹得分与在基线夜间醒来哭闹得分之差的平均分的示意图。Figure 4 is a schematic diagram of the mean score of the difference between the scores of infants in the test group, the control group and the breast milk group at each time point when they wake up crying at night and at the baseline.
具体实施方式Detailed ways
下面将结合实施例对本发明的实施方案进行清楚、完整地描述,显然,所描述的实施例仅仅是本发明一部分实施例,而不是全部的实施例。以下对至少一个示例性实施例的描述实际上仅仅是说明性的,决不作为对本发明及其应用或使用的任何限制。基于本发明中的实施例,本领域普通技术人员在没有作出创造性劳动前提下所获得的所有其他实施例,都属于本发明保护的范围。The embodiments of the present invention will be clearly and completely described below in conjunction with the examples. Apparently, the described examples are only some of the examples of the present invention, but not all of them. The following description of at least one exemplary embodiment is merely illustrative in nature and in no way taken as limiting the invention, its application or uses. Based on the embodiments of the present invention, all other embodiments obtained by persons of ordinary skill in the art without creative efforts fall within the protection scope of the present invention.
实施例Example
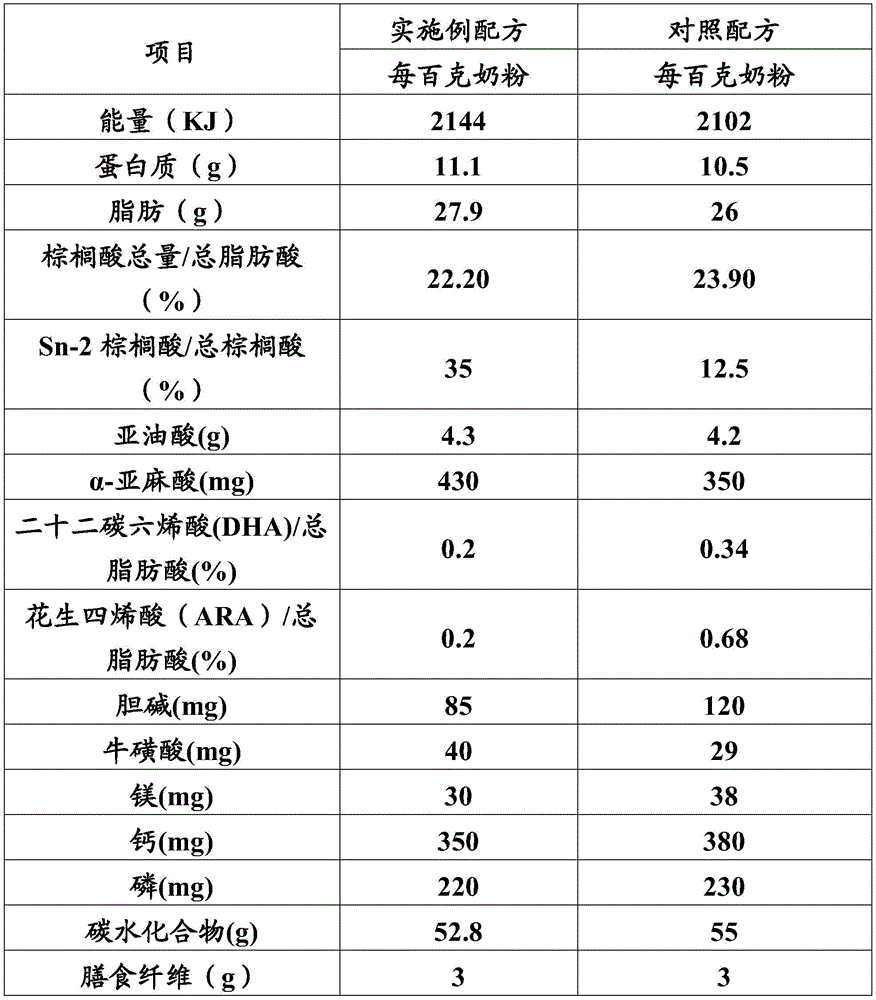
按照表1中的实施例配方制备得到配方奶粉。每百克奶粉中约含Sn-2棕榈酸甘油酯2.17g。Formula milk powder was prepared according to the example formula in Table 1. About 2.17g of Sn-2 glyceryl palmitate is contained in every 100g of milk powder.
表1Table 1
对比例comparative example
按照表1中的对照配方制备得到配方奶粉。每百克奶粉中约含Sn-2棕榈酸甘油酯0.7g。Formula milk powder was prepared according to the control formula in Table 1. About 0.7g of Sn-2 glyceryl palmitate is contained in every 100g of milk powder.
临床实验方法及实验结果Clinical Experimental Methods and Experimental Results
随机对照设计,比较实施例配方与对照配方奶粉的喂养效果。Randomized control design, compare the feeding effect of embodiment formula and contrast formula milk powder.
1.实验对象分组:1. Grouping of experimental subjects:
由儿科医师或经训练的研究人员通过招募筛选问卷筛选需要纳入的婴儿。进入研究前需获得母亲书面的知情同意书。Infants for inclusion were screened by a pediatrician or trained researcher using a recruitment screening questionnaire. Written informed consent from the mother was required before entering the study.
1.1纳入标准1.1 Inclusion criteria
足月儿:孕周≥37周;Full-term infants: gestational weeks ≥ 37 weeks;
出生体重:2.5kg-4kg;Birth weight: 2.5kg-4kg;
正常妊娠、分娩的婴儿(包括剖宫产);Babies with normal pregnancy and delivery (including caesarean section);
身体健康,出生5-10分钟后的Apgar评分>7分;Good health, Apgar score > 7 points 5-10 minutes after birth;
年龄:<15天。Age: <15 days.
1.2排除标准1.2 Exclusion criteria
具有以下任一特征的婴儿被排除:Infants with any of the following characteristics were excluded:
出生时检测到先天性畸形或染色体病且具有临床意义者;Congenital malformations or chromosomal diseases detected at birth with clinical significance;
出生后一周内患有疾病需要机械换气或药物治疗者(不包括蓝光治疗的婴儿期黄疸患者);Those who suffer from diseases requiring mechanical ventilation or drug treatment within one week after birth (excluding infantile jaundice patients treated with blue light);
因可疑或未知的代谢性因素或因机体缺陷影响喂养或代谢者;Those who affect feeding or metabolism due to suspicious or unknown metabolic factors or body defects;
双生子或多胞胎。Twins or multiple births.
1.3实验分组1.3 Experimental grouping
选取0-6月龄足月儿为研究对象,婴儿出生后母乳充足,母亲愿意基本母乳喂养到满6月龄的,作为母乳组;无法实施母乳喂养,决定采用婴儿配方奶粉喂养,1-15日龄婴儿配方奶粉喂养量≥250ml/d,开始干预后奶粉喂养大于80%以上的婴儿,随机分为测试组(喂养实施例配方奶粉)和对照组(喂养对照配方奶粉)。每组的入组人数不少于5人。Full-term infants aged 0-6 months were selected as the research object, and the infants had sufficient breast milk after birth, and the mothers were willing to breastfeed until the age of 6 months, as the breast milk group; breastfeeding was not possible, and infant formula feeding was decided, 1-15 Day-old infant formula feeding amount ≥ 250ml/d, milk powder feeding greater than 80% of infants after starting intervention, are randomly divided into test group (feeding embodiment formula milk powder) and control group (feeding contrast formula milk powder). The number of participants in each group is not less than 5 people.
2.干预研究方法2. Intervention Research Methods
对入组的婴儿开展基线(出生后第15天)的调查与样本的收集,之后连续喂养6个月。在此期间,项目研究人员于开始喂养后的4周、6周、8周、16周、24周分别对实验对象进行随访。调查婴儿的基本人口社会学情况、肠绞痛发生情况及上火情况。Baseline (15th day after birth) survey and sample collection were carried out on the enrolled infants, and then they were fed continuously for 6 months. During this period, the project researchers followed up the subjects at 4 weeks, 6 weeks, 8 weeks, 16 weeks and 24 weeks after the start of feeding. Investigate the infant's basic demographic and sociological conditions, the incidence of colic and the situation of getting angry.
3.临床实验结果3. Results of clinical trials
3.1不同实验组婴儿的基本人口社会学情况3.1 Basic demographic and sociological conditions of infants in different experimental groups
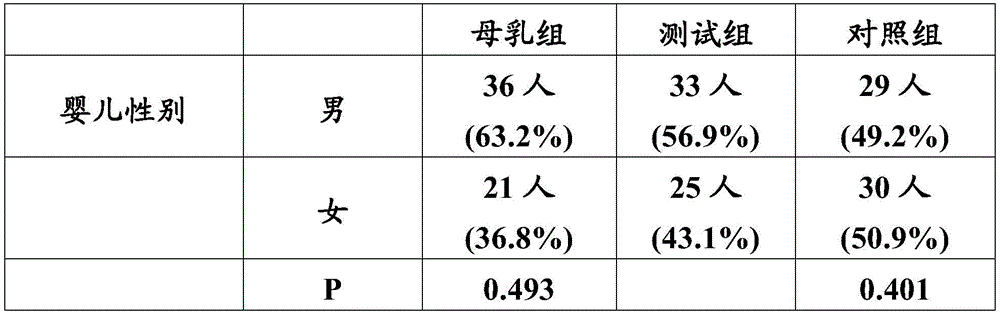
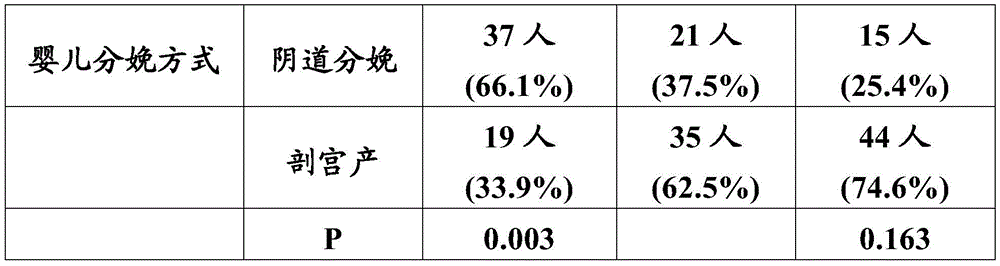
比较三个实验组的婴儿发现:测试组、母乳组和对照组的社会人口学分布相近,只是测试组与母乳组婴儿的父亲工作状况、父母最高学历和家庭收入稍有差异,但对比研究婴儿母乳喂养与婴儿配方奶粉喂养的现有文献、报告通常也体现出这些差异,故这些差异不会妨碍或影响本实验研究结果的对比性。另外,如表3.1所示:三组婴儿的性别比例无显著差异;与测试组和对照组相比,母乳组的阴道分娩率更高,并且,测试组与对照组的阴道分娩率无差异;母乳组或对照组与测试组数据的T检验p值也提供在表3.1中,p值<0.05说明有统计学差异,p值<0.01说明有显著性统计学差异。Comparing the infants of the three experimental groups found that the sociodemographic distributions of the test group, the breast milk group and the control group were similar, but there were slight differences in the father's work status, parents' highest education level and family income between the test group and the breast milk group. The existing literature and reports on breastfeeding and infant formula feeding usually also reflect these differences, so these differences will not hinder or affect the comparison of the results of this experiment. In addition, as shown in Table 3.1: there was no significant difference in the sex ratio of the infants in the three groups; compared with the test group and the control group, the vaginal delivery rate in the breast milk group was higher, and there was no difference in the vaginal delivery rate between the test group and the control group; The T-test p-values of the breast milk group or control group and the test group data are also provided in Table 3.1. A p-value < 0.05 indicates a statistical difference, and a p-value < 0.01 indicates a significant statistical difference.
表3.1婴儿性别及分娩方式构成Table 3.1 Composition of Infant Sex and Mode of Delivery
3.2不同实验组婴儿的肠绞痛发生情况3.2 Incidence of colic among infants in different experimental groups
观察三个实验组婴儿的肠绞痛发生率(%),结果如图1所示。The incidence rate (%) of intestinal colic among infants in the three experimental groups was observed, and the results are shown in FIG. 1 .
由图1可知,尽管在基线时测试组婴儿的肠绞痛发生率高于对照组,但随着喂养时间延长,第6周之后测试组婴儿肠绞痛的发生率均明显低于对照组;而且,测试组婴儿的肠绞痛发生率最高值出现在第6-8周,而后逐渐下降,接近于母乳组的变化趋势,而对照组婴儿的肠绞痛发生率最高值出现在16周,始终未见减少,带给婴儿和家人更大的压力。这说明本发明配方奶粉能有效缓解或改善婴幼儿的肠绞痛症状。It can be seen from Figure 1 that although the incidence of colic in the test group was higher than that in the control group at the baseline, as the feeding time was prolonged, the incidence of colic in the test group was significantly lower than that in the control group after the 6th week; Moreover, the highest incidence of colic in the test group appeared at 6-8 weeks, and then gradually decreased, which was close to the change trend of the breast milk group, while the highest incidence of colic in the control group appeared at 16 weeks, There has been no decrease, and it has brought greater pressure to the baby and the family. This shows that the formula milk powder of the present invention can effectively alleviate or improve the colic symptoms of infants and young children.
3.3不同实验组婴儿的上火情况3.3 Inflammation of infants in different experimental groups
根据中医学理论,婴幼儿上火后一般会出现面色赤红、眼睛红肿、眼屎多、口唇干燥、口臭(除奶味之外的异味)、有鼻涕渣或黄鼻涕、手脚心发热、尿黄、舌苔增厚、大便发硬的症状,还会表现出夜间醒来哭闹、脾气急躁、爱生气、白天哭闹、比平时容易饿、比平时吃得少、爱蹬被、不爱穿衣物的行为。观察三个实验组婴儿在基线(出生后第15天)、第4周、6周、16周、24周的前述上火症状及行为表现的发生情况并进行评分。评分标准:除大便情况外,每出现一项症状或行为表现加一分,不出现则不加分;若大便为水样的,加一分;若大便为细软的,加两分;若大便略成型,加三分;若大便为干硬的,加四分;若大便非常干硬,加五分。各组婴儿在各时间点上火症状及行为表现总分与在基线上火症状及行为表现总分的差值的平均分如图2所示。各组婴儿在各时间点上火行为表现得分与在基线上火行为表现得分的差值的平均分如图3所示。各组婴儿在各时间点夜间醒来哭闹一项得分与在基线夜间醒来哭闹得分的差值的平均分如图4所示。According to the theory of traditional Chinese medicine, after infants and young children get angry, they usually have red complexion, red and swollen eyes, excessive eye mucus, dry lips, bad breath (except for the smell of milk), mucus or yellow nose, hot hands and feet, yellow urine, Thickened tongue coating, hard stools, waking up at night crying, irritable, angry, crying during the day, hungry more than usual, eating less than usual, love to push the quilt, don't like to wear clothes the behavior of. Observe and score the above-mentioned symptoms and behaviors of infants in the three experimental groups at baseline (15th day after birth), 4th week, 6th week, 16th week, and 24th week. Scoring criteria: Except for stool, add one point for each symptom or behavior, and no point for absence; if the stool is watery, add one point; if the stool is soft, add two points; if the stool is soft, add one point; Slightly formed, add three points; if the stool is dry and hard, add four points; if the stool is very dry and hard, add five points. The average score of the difference between the total scores of the symptoms and behavioral performance of infants in each group at each time point and the total score of the symptoms and behavioral performance at the baseline is shown in Figure 2. The average score of the difference between the scores of angry behavior performance scores of infants in each group at each time point and the scores of baseline angry behavior performance is shown in Figure 3. The average score of the difference between the score of crying at night and the score of waking up and crying at baseline in each group of infants at each time point is shown in Figure 4.
由图2可知,测试组与对照组婴儿第4周相对于基线的上火程度改善状况近似,之后随着喂养时间延长,测试组婴儿的上火情况逐渐得到缓解,第24周上火程度明显低于第4周;而对照组婴儿的上火程度在第4-6周大幅加重,而后在第8周恢复至第4周水平,然后又在8-16周有所加重,直至第24周仅比第4周稍有缓解,但仍比测试组婴儿第24周的上火程度严重许多。由图3可知,测试组婴儿第4周相对于基线的上火行为表现改善情况好于对比组婴儿;之后,对照组婴儿上火行为表现程度加重,至第16-24周上火行为表现才得到缓解,但仍比测试组婴儿严重许多;而测试组婴儿第4至24周上火行为表现情况逐渐好转,至第24周与母乳组婴儿的上火行为表现情况近似。由图4可知,对照组婴儿的夜间哭闹情况始终比测试组婴儿更严重。这说明本发明配方奶粉有助于改善婴幼儿的上火症状、上火行为表现。It can be seen from Figure 2 that the infants in the test group and the control group showed similar improvements in the degree of anger compared with the baseline at the 4th week. Afterwards, with the prolongation of the feeding time, the infants in the test group gradually alleviated the degree of irritation, and the degree of irritation in the 24th week was obvious lower than the 4th week; while the infants in the control group showed a sharp increase in the degree of fire in the 4th to 6th week, and then returned to the level of the 4th week in the 8th week, and then increased in the 8th to 16th week until the 24th week It was only slightly relieved from the 4th week, but still much more severe than the 24th week of the test group infants. It can be seen from Figure 3 that the performance of the infants in the test group improved better than that of the infants in the control group in the 4th week compared with the baseline; after that, the infants in the control group showed aggravated behaviors, and the performance of the infants in the control group did not improve until the 16th to 24th week. It was relieved, but it was still much more serious than that of the infants in the test group; while the behavior of the infants in the test group gradually improved from the 4th to the 24th week, and was similar to that of the infants in the breast milk group by the 24th week. It can be seen from Figure 4 that the nighttime crying of the infants in the control group was always more serious than that of the infants in the test group. This shows that the formula milk powder of the present invention is helpful to improve the symptoms of getting angry and the performance of getting angry in infants and young children.
显然,上述实施例仅仅是为清楚地说明所作的举例,而并非对实施方式的限定。对于所属领域的普通技术人员来说,在上述说明的基础上还可以做出其它不同形式的变化或变动。这里无需也无法对所有的实施方式予以穷举。而由此所引伸出的显而易见的变化或变动仍处于本发明创造的保护范围之中。Apparently, the above-mentioned embodiments are only examples for clear description, rather than limiting the implementation. For those of ordinary skill in the art, other changes or changes in different forms can be made on the basis of the above description. It is not necessary and impossible to exhaustively list all the implementation manners here. And the obvious changes or changes derived therefrom are still within the scope of protection of the present invention.
Claims (8)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202310545216.5A CN116584653A (en) | 2019-11-20 | 2020-03-02 | Application of composition in preventing or relieving infant colic |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2019111427130 | 2019-11-20 | ||
| CN201911142713 | 2019-11-20 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202310545216.5A Division CN116584653A (en) | 2019-11-20 | 2020-03-02 | Application of composition in preventing or relieving infant colic |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN112823667A CN112823667A (en) | 2021-05-21 |
| CN112823667B true CN112823667B (en) | 2023-06-02 |
Family
ID=70862730
Family Applications (8)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201911345399.6A Pending CN112823646A (en) | 2019-11-20 | 2019-12-24 | Composition, food or drug and use thereof |
| CN202010104302.9A Active CN111248287B (en) | 2019-11-20 | 2020-02-20 | Application of dairy product in improving neurodevelopment |
| CN202010104301.4A Active CN111227044B (en) | 2019-11-20 | 2020-02-20 | Dairy product, preparation method and medical application thereof |
| CN202010104314.1A Active CN111227045B (en) | 2019-11-20 | 2020-02-20 | Application of dairy product in promoting growth of length of mammal |
| CN202010135335.XA Active CN112823647B (en) | 2019-11-20 | 2020-03-02 | Application of composition in improving or promoting growth of infant |
| CN202010135666.3A Active CN112823667B (en) | 2019-11-20 | 2020-03-02 | Application of the composition in preventing or alleviating colic in infants and young children |
| CN202310545216.5A Withdrawn CN116584653A (en) | 2019-11-20 | 2020-03-02 | Application of composition in preventing or relieving infant colic |
| CN202010135640.9A Active CN112823648B (en) | 2019-11-20 | 2020-03-02 | Composition, food or medicine and application thereof |
Family Applications Before (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201911345399.6A Pending CN112823646A (en) | 2019-11-20 | 2019-12-24 | Composition, food or drug and use thereof |
| CN202010104302.9A Active CN111248287B (en) | 2019-11-20 | 2020-02-20 | Application of dairy product in improving neurodevelopment |
| CN202010104301.4A Active CN111227044B (en) | 2019-11-20 | 2020-02-20 | Dairy product, preparation method and medical application thereof |
| CN202010104314.1A Active CN111227045B (en) | 2019-11-20 | 2020-02-20 | Application of dairy product in promoting growth of length of mammal |
| CN202010135335.XA Active CN112823647B (en) | 2019-11-20 | 2020-03-02 | Application of composition in improving or promoting growth of infant |
Family Applications After (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202310545216.5A Withdrawn CN116584653A (en) | 2019-11-20 | 2020-03-02 | Application of composition in preventing or relieving infant colic |
| CN202010135640.9A Active CN112823648B (en) | 2019-11-20 | 2020-03-02 | Composition, food or medicine and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (8) | CN112823646A (en) |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112823646A (en) * | 2019-11-20 | 2021-05-21 | 内蒙古伊利实业集团股份有限公司 | Composition, food or drug and use thereof |
| CN114223721A (en) * | 2020-09-09 | 2022-03-25 | 内蒙古伊利实业集团股份有限公司 | Application of palmitic acid glyceride in improving or promoting protein absorption of infants |
| CN116998725B (en) * | 2020-11-30 | 2025-09-16 | 内蒙古伊利实业集团股份有限公司 | Composition containing fructo-oligosaccharide and probiotics and application thereof |
| CN114287478A (en) * | 2021-11-23 | 2022-04-08 | 临夏燎原乳业产业研究院有限公司 | Formula yak milk powder for promoting height increase and brain development of children and preparation method thereof |
| CN114145458A (en) * | 2021-11-29 | 2022-03-08 | 内蒙古伊利实业集团股份有限公司 | Application of palmitic acid glyceride in preparing composition for improving or promoting magnesium absorption of human body |
| CN114145346A (en) * | 2021-11-29 | 2022-03-08 | 内蒙古伊利实业集团股份有限公司 | Food composition, application of food composition in improving or promoting magnesium absorption and infant formula milk powder |
| CN115669945B (en) * | 2022-10-24 | 2024-03-08 | 上海艾斯顿医疗科技有限公司 | Liposome-coated polypeptide with neurotrophic effect, and preparation method and application thereof |
| CN115825294B (en) * | 2023-02-07 | 2023-05-26 | 北京三元食品股份有限公司 | Marker group for evaluating infant communication capacity and development condition and application thereof |
| CN116008438B (en) * | 2023-02-07 | 2023-06-06 | 北京三元食品股份有限公司 | Marker group for evaluating fine action development condition of infants and application |
| CN116369527B (en) * | 2023-03-09 | 2024-07-30 | 江南大学 | Bifidobacterium breve capable of regulating bone development and application thereof |
| CN118717809A (en) * | 2023-03-24 | 2024-10-01 | 丰华生物科技股份有限公司 | Use of culture of Bifidobacterium longum subsp. infantis BLI-02 in improving calcium absorption |
Family Cites Families (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2012335224B2 (en) * | 2011-11-10 | 2016-10-13 | Société des Produits Nestlé S.A. | Infant formula with high sn-2 palmitate and oligofructose |
| CN102524421B (en) * | 2011-12-31 | 2013-05-22 | 黑龙江省完达山乳业股份有限公司 | Novel acid-base balance infant formula milk powder and preparation method thereof |
| MX2012010488A (en) * | 2012-09-11 | 2014-03-26 | Nucitec Sa De Cv | Infant formula for optimal growth, gastrointestinal protection and immunological protection of infants. |
| MX2012010487A (en) * | 2012-09-11 | 2014-03-26 | Nucitec Sa De Cv | Composition of nutrients and bioactive substances which promote growth and reduce intolerance and side effects in premature infants. |
| CN103053694A (en) * | 2013-02-18 | 2013-04-24 | 杨凌圣妃乳业有限公司 | Infant goat milk powder simulating components of breast milk and preparation method thereof |
| FR3005392B1 (en) * | 2013-05-07 | 2015-09-18 | United Pharmaceuticals | ANTI-REGURGITATION COMPOSITION PRESERVING INTESTINAL TRANSIT |
| CN104351356A (en) * | 2014-11-25 | 2015-02-18 | 澳优乳业(中国)有限公司 | Novel fourth-generation infant formula and preparation method thereof |
| CN105767213A (en) * | 2014-12-26 | 2016-07-20 | 丰益(上海)生物技术研发中心有限公司 | Grease composition and preparation method thereof |
| CN106259953B (en) * | 2015-06-01 | 2019-01-29 | 内蒙古伊利实业集团股份有限公司 | Breast milk 0-12 month babies ' formula milk powder containing nucleotide and preparation method thereof |
| CN106259952B (en) * | 2015-06-01 | 2019-01-15 | 内蒙古伊利实业集团股份有限公司 | 1-3 years old baby formulas milk powder and preparation method thereof containing nucleotide and dietary fiber |
| CN105053217A (en) * | 2015-09-14 | 2015-11-18 | 海普诺凯营养品有限公司 | Infant formula ewe milk powder and production method thereof |
| CN107439686A (en) * | 2016-05-31 | 2017-12-08 | 内蒙古伊利实业集团股份有限公司 | A kind of full-cream chocolate milk and preparation method thereof |
| CN107668211B (en) * | 2017-11-13 | 2021-08-10 | 海普诺凯营养品有限公司 | Infant formula goat milk powder for enhancing immune function and preparation method thereof |
| WO2019104390A1 (en) * | 2017-12-01 | 2019-06-06 | Freedom Foods Group IP Pty Limited | Milk substitute compositions |
| CN110063372A (en) * | 2018-01-23 | 2019-07-30 | 天津澳斯乳业有限公司 | A kind of infant formula goat milk powder and its production method |
| CN108013143A (en) * | 2018-02-05 | 2018-05-11 | 高培(广州)乳业有限公司 | A formula milk powder for strengthening the gastrointestinal function of infants aged 12 to 36 months |
| CN108157512A (en) * | 2018-02-05 | 2018-06-15 | 高培(广州)乳业有限公司 | A formula milk powder for strengthening the gastrointestinal function of infants aged 6-12 months |
| CN112823646A (en) * | 2019-11-20 | 2021-05-21 | 内蒙古伊利实业集团股份有限公司 | Composition, food or drug and use thereof |
-
2019
- 2019-12-24 CN CN201911345399.6A patent/CN112823646A/en active Pending
-
2020
- 2020-02-20 CN CN202010104302.9A patent/CN111248287B/en active Active
- 2020-02-20 CN CN202010104301.4A patent/CN111227044B/en active Active
- 2020-02-20 CN CN202010104314.1A patent/CN111227045B/en active Active
- 2020-03-02 CN CN202010135335.XA patent/CN112823647B/en active Active
- 2020-03-02 CN CN202010135666.3A patent/CN112823667B/en active Active
- 2020-03-02 CN CN202310545216.5A patent/CN116584653A/en not_active Withdrawn
- 2020-03-02 CN CN202010135640.9A patent/CN112823648B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN112823647B (en) | 2023-04-07 |
| CN116584653A (en) | 2023-08-15 |
| CN112823648B (en) | 2023-03-31 |
| CN111227044A (en) | 2020-06-05 |
| CN111248287A (en) | 2020-06-09 |
| CN111227045A (en) | 2020-06-05 |
| CN112823647A (en) | 2021-05-21 |
| CN112823667A (en) | 2021-05-21 |
| CN111227044B (en) | 2023-05-16 |
| CN112823646A (en) | 2021-05-21 |
| CN111227045B (en) | 2023-04-14 |
| CN111248287B (en) | 2023-04-14 |
| CN112823648A (en) | 2021-05-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN112823667B (en) | Application of the composition in preventing or alleviating colic in infants and young children | |
| RU2552039C2 (en) | Methods and lipid compositions aiding in intestinal flora development | |
| EP2475764B1 (en) | Probiotic derived non-viable material for allergy prevention and treatment | |
| EP1976397B1 (en) | Infant immunological formula | |
| WO2015091789A2 (en) | Nutritional composition to reduce metabolic stress in infants | |
| TW201302094A (en) | Nutritional composition with fortified milk as a substrate | |
| US20240138463A1 (en) | Infant formulas containing human breast milk proteins | |
| MX2013005943A (en) | Use of nutritional compositions including lactoferrin in supporting resistance to diseases and conditions. | |
| US12533368B2 (en) | Compositions for use in the reduction of pain and/or perception of pain in infants and young children | |
| US12527342B2 (en) | Supplementary composition containing 2′-fucosyllactose (2′-fl) and fructo-oligosaccharide (FOS) with synergistic effects on the digestive and immune systems for children, and process of manufacturing a children formula containing said supplementary composition | |
| RU2826606C1 (en) | Compositions for use in reducing pain and/or pain perception in infants and young children | |
| CN111227046B (en) | Application of dairy product in improving infant intestinal SIgA level | |
| US20220000164A1 (en) | Nutritional compositions, their use in reducing metabolic stress and method of reducing metabolic stress | |
| HK1261683A1 (en) | Fortified milk-based nutritional compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |