CN112538093B - Preparation method of photoinitiator - Google Patents
Preparation method of photoinitiator Download PDFInfo
- Publication number
- CN112538093B CN112538093B CN202011500888.7A CN202011500888A CN112538093B CN 112538093 B CN112538093 B CN 112538093B CN 202011500888 A CN202011500888 A CN 202011500888A CN 112538093 B CN112538093 B CN 112538093B
- Authority
- CN
- China
- Prior art keywords
- phosphine oxide
- trimethylbenzoyl
- butoxide
- photoinitiator
- tert
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/50—Organo-phosphines
- C07F9/53—Organo-phosphine oxides; Organo-phosphine thioxides
- C07F9/5333—Arylalkane phosphine oxides or thioxides
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
Abstract
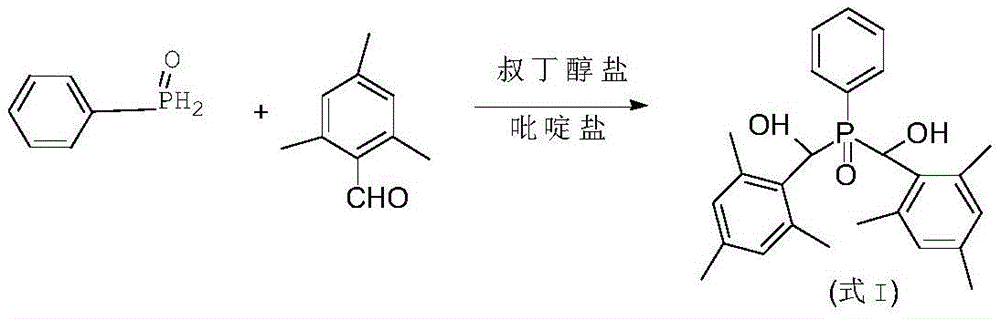
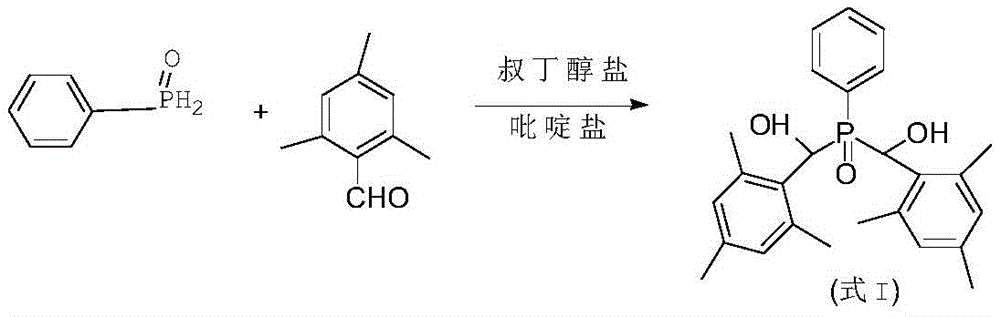
The invention provides a preparation method of a photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, which is characterized by comprising the following steps: (1) taking water as a solvent, phenyl phosphine oxide and 2,4, 6-trimethylbenzaldehyde as raw materials, and tert-butoxide and pyridinium as catalysts, and obtaining an intermediate compound shown as a formula I through reaction, wherein the formula is as follows:(2) oxidizing the formula I under the action of an oxidizing agent to obtain a target product, namely, a photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, wherein the formula is as follows:
Description
Technical Field
The invention belongs to the field of organic synthesis, and particularly relates to a preparation method of a photoinitiator, and more particularly relates to a preparation method of a photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide.
Background
Phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide (trade name photoinitiator 819, also called BAPO) is a novel photoinitiator with good photochemical reaction activity, the maximum absorption wavelength can reach 450nm, the photodecomposition speed is high, after photolysis, two mesitylbenzoyl free radicals and a diphenyl phosphonyl free radical are generated, the free radicals have high activity and high photocuring speed, the bleaching effect is achieved, and thick coating curing is facilitated. The phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide has excellent performance, is often combined with secondary amine and tertiary amine, and is widely applied to a plurality of fields such as coating, printing ink, adhesive, corrosion inhibitor, welding agent and the like. The ultraviolet curing agent is suitable for initiating radiation polymerization reaction of some unsaturated resins under ultraviolet irradiation, and the deep curing effect of the ultraviolet curing agent is very ideal. The ultraviolet curing photoinitiator is suitable for ultraviolet curing varnish and paint systems, such as woodware, paper, metal, plastics, optical fibers, printing ink, preimpregnation systems and the like, is particularly suitable for white formulations, glass fiber reinforced polyester/styrene systems and varnish systems matched with light stabilizers and used outdoors, and is a photoinitiator required by ideal weather-resistant ultraviolet curing coatings. Phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide can be used alone or in combination with other photoinitiators, such as Irgacure 184 or Irgacure 651.
At present, there are several methods reported in the literature for preparing phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide:
(1) the method comprises the following steps of taking phenylphosphonic dichloride as a starting raw material, reacting with sodium or potassium metal in a solvent, and then reacting with 2,4, 6-trimethylbenzoyl chloride to obtain an intermediate product bis (2,4, 6-trimethylbenzoyl) phenylphosphine, and oxidizing the intermediate by hydrogen peroxide to obtain the phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide.
(2) And (2) similarly, taking phenyl phosphine dichloride as a starting material, firstly reacting with sodium or potassium metal in a solvent, then adding tert-butyl alcohol for alcoholysis, then reacting with 2,4, 6-trimethylbenzoyl chloride to obtain an intermediate product bis (2,4, 6-trimethylbenzoyl) phenyl phosphine, and oxidizing the intermediate by hydrogen peroxide to obtain the target product phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide. The method has similar problems with the method (1), and is also not suitable for industrial development.
(3) The method takes 2,4, 6-trimethyl benzoyl chloride as a reaction raw material, then the reaction raw material is subjected to substitution reaction with phenyl phosphine and a phenyl phosphine bimetallic complex, and then oxidation reaction is carried out to prepare the phenyl bis (2,4, 6-trimethyl benzoyl) phosphine oxide, however, the 2,4, 6-trimethyl benzoyl chloride used as a starting raw material for the reaction has high production cost, a large amount of hydrogen chloride and sulfur dioxide are generated in the preparation process, the environmental pollution is serious, and the phenyl phosphine bimetallic complex is not easy to prepare.
(4) 1,3, 5-trimethyl-2- (trichloromethyl) benzene is used as a reaction raw material, firstly reacts with metal, and then reacts with phenyl phosphine dichloride to prepare phenyl bis (2,4, 6-trimethyl benzoyl) phosphine oxide through an oxidation reaction (or reacts with phenyl phosphorus dimallate and is prepared through the oxidation reaction). However, the production of the starting material 1,3, 5-trimethyl-2- (trichloromethyl) benzene requires carbon tetrachloride with high toxicity, and the reaction also generates a large amount of acid gas and a large amount of acid wastewater to pollute the environment, so that the method is not suitable for industrial large-scale production.
(5) Phenyl phosphine oxide is used as a raw material, firstly, the phenyl phosphine oxide reacts with 2,4, 6-trimethylbenzaldehyde under the action of an alkaline catalyst to prepare an intermediate double-hydroxy phosphine oxide, and then, phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide is prepared by using heteropoly acid as a catalyst and peroxide as an oxidant. However, in the preparation process of the method, benzene, ester, chloroalkane and the like with high toxicity are used as solvents, so that the method is expensive and environment-friendly, and the whole reaction route is complicated and the yield is not high.
Disclosure of Invention
The invention aims to overcome a series of defects existing in the prior art for synthesizing phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, and provides a preparation method of phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, which comprises the following steps: (1) taking water as a solvent, phenyl phosphine oxide and 2,4, 6-trimethylbenzaldehyde as raw materials, and tert-butoxide and pyridinium as catalysts, and obtaining an intermediate compound shown as a formula I through reaction, wherein the formula is as follows:
(2) oxidizing the formula I under the action of an oxidizing agent to obtain a target product, namely, a photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, wherein the formula is as follows:
according to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the step (1) specifically comprises the steps of taking water as a solvent, adding phenyl phosphine oxide and 2,4, 6-trimethylbenzaldehyde into the water, then adding tert-butoxide and pyridinium into the water, uniformly stirring the mixture, and then controlling the temperature to be 0-100 ℃ and the reaction time to be 1-10 h.
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the molar ratio of the phenyl phosphine oxide to the 2,4, 6-trimethylbenzaldehyde is 1: 2-4.
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the molar ratio of the phenyl phosphine oxide to the 2,4, 6-trimethylbenzaldehyde is preferably 1: 2-3.
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the molar and mass ratio of the phenyl phosphine oxide to tert-butoxide and pyridinium is 1mol: 1-20 g: 1-100 g.
According to the preparation method of the photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, the tert-butoxide is selected from one or more of potassium tert-butoxide, sodium tert-butoxide or magnesium tert-butoxide.
According to the preparation method of the photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, the pyridinium is one or more selected from cetylpyridinium chloride, 2-chloro-1-methylpyridinium iodide or 1, 2-dimethylpyridinium iodide.
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the step (2) specifically comprises the steps of adding an oxidant into a reaction system containing the compound shown in the formula I obtained in the step (1) at 0-100 ℃, reacting for 1-10 hours, after the reaction is finished, adding an organic solvent for extraction, taking an organic layer, drying, distilling under reduced pressure, and recrystallizing to obtain the target product phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide; the oxidant is selected from one or more of hydrogen peroxide, sodium peroxide, potassium peroxide, magnesium peroxide, calcium peroxide or perdiphosphonic acid.
According to the preparation method of the photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, the reaction temperature in the step (2) is preferably 20-50 ℃, and more preferably 20-30 ℃.
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the reaction time in the step (2) is preferably 1-5 h.
According to the preparation method of the photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, in the step (2), heteropoly acid or heteropoly acid salt is further used as a catalyst.
According to the preparation method of the photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, in the step (2), preferably, the heteropoly acid or heteropoly acid salt is selected from H4SiW12O40、H3PW12O40、H4SiMo12O40Or H3PMo12O40One or more of (a).
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the organic solvent in the step (2) is selected from one or more of ethyl acetate and dichloromethane.
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the molar and mass ratio of the phenyl phosphine oxide to the oxidant to the catalyst is 1mol: 1-50 g: 1-100 g.
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the reaction temperature of the step (1) and the reaction temperature of the step (2) are preferably room temperature.
According to the preparation method of the photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, a solvent selected by recrystallization is n-hexane or petroleum ether.
Furthermore, the invention also provides the application of tert-butoxide and pyridinium in preparing a photoinitiation, in particular to the application of tert-butoxide and pyridinium in preparing phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide.
The main contributions of the present invention with respect to the prior art are the following:
(1) the invention takes water as a solvent, takes phenyl phosphine oxide and 2,4, 6-trimethylbenzaldehyde as raw materials, takes tert-butoxide salt and pyridinium as catalysts, obtains a compound with an intermediate formula I through reaction, and then further oxidizes the compound to obtain phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, and the prior art has no related report of preparing the phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide by using the method, and the invention belongs to an pioneering invention.
(2) In the step (1), tert-butoxide and pyridinium are used as catalysts, and the pyridinium has the function of a phase transfer catalyst, so that water can be directly used as a reaction solvent, and an organic solvent with toxicity and relatively high price is avoided, and the method is more economic and more environment-friendly compared with the prior art.
(3) The method uses a very small amount of catalyst or oxidant in the steps (1) and (2), has mild reaction temperature, obvious reaction time, simple post-treatment, high yield and purity and unexpected technical effect.
Detailed Description
In order to make the objects, technical solutions and advantages of the embodiments of the present disclosure more apparent, the technical solutions of the embodiments of the present disclosure are clearly and completely described. It is to be understood that the described embodiments are only a few embodiments of the present disclosure, and not all embodiments. All other embodiments, which can be derived by a person skilled in the art from the described embodiments of the disclosure without any inventive step, are within the scope of protection of the disclosure.
The invention provides a preparation method of phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, which comprises the following steps: (1) taking water as a solvent, phenyl phosphine oxide and 2,4, 6-trimethylbenzaldehyde as raw materials, and tert-butoxide and pyridinium as catalysts, and obtaining an intermediate compound shown as a formula I through reaction, wherein the formula is as follows:
(2) oxidizing the formula I under the action of an oxidizing agent to obtain a target product, namely, a photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, wherein the formula is as follows:
according to the preparation method of the photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, the step (1) specifically comprises the steps of taking water as a solvent, adding phenylphosphine oxide and 2,4, 6-trimethylbenzaldehyde into the water, then adding tert-butoxide and pyridinium, uniformly stirring, and then controlling the temperature at 0-100 ℃ and the reaction time at 1-10 h.
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the molar ratio of the phenyl phosphine oxide to the 2,4, 6-trimethylbenzaldehyde is 1: 2-4.
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the molar ratio of the phenyl phosphine oxide to the 2,4, 6-trimethylbenzaldehyde is preferably 1: 2-3.
According to the preparation method of the photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, the molar and mass ratio of the phenylphosphine oxide to the tert-butoxide and the pyridinium is 1mol: 1-20 g: 1-100 g.
According to the preparation method of the photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, the tert-butoxide is selected from one or more of potassium tert-butoxide, sodium tert-butoxide or magnesium tert-butoxide.
According to the preparation method of the photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, the pyridinium is one or more selected from cetylpyridinium chloride, 2-chloro-1-methylpyridinium iodide or 1, 2-dimethylpyridinium iodide.
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the step (2) specifically comprises the steps of adding an oxidant into a reaction system containing the compound shown in the formula I obtained in the step (1) at 0-100 ℃, reacting for 1-10 hours, after the reaction is finished, adding an organic solvent for extraction, taking an organic layer, drying, distilling under reduced pressure, and recrystallizing to obtain the target product phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide; the oxidant is selected from one or more of hydrogen peroxide, sodium peroxide, potassium peroxide, magnesium peroxide, calcium peroxide or perdiphosphonic acid.
According to the preparation method of the photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, the reaction temperature in the step (2) is preferably 20-50 ℃, and more preferably 20-30 ℃.
According to the preparation method of the photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, the reaction time in the step (2) is preferably 1-5 h.
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, heteropoly acid or heteropoly acid salt is further used as a catalyst in the step (2).
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the step (2) preferably selects heteropoly acid or heteropoly acid salt from H4SiW12O40、H3PW12O40、H4SiMo12O40Or H3PMo12O40One or more of (a).
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the organic solvent in the step (2) is selected from one or more of ethyl acetate and dichloromethane.
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the molar and mass ratio of the phenyl phosphine oxide to the oxidant to the catalyst is 1mol: 1-50 g: 1-100 g.
According to the preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, the reaction temperature of the step (1) and the reaction temperature of the step (2) are preferably room temperature.
According to the preparation method of the photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, a solvent selected by recrystallization is n-hexane or petroleum ether.
Example 1
Water, 100mmol of phenylphosphine oxide, 200mmol of 2,4, 6-trimethylbenzaldehyde, 2g of potassium tert-butoxide and 10g of cetylpyridinium chloride are added in sequence into a reaction flask, stirred sufficiently until uniform, reacted at room temperature for 5h, and the reaction is completed by TLC to obtain a mixed solution containing the intermediate compound of formula I. 2gH is reacted with4SiW12O40Adding the mixture into a mixed solution, slowly dropwise adding 5g of hydrogen peroxide into the system while stirring, reacting at room temperature for 10 hours after dropwise adding is finished, detecting that the reaction is complete by TLC, adding ethyl acetate for extraction, taking an organic phase, drying by anhydrous sodium sulfate, distilling under reduced pressure, and recrystallizing by using n-hexane to obtain 39.1g of target product phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, wherein the yield is 93.5%, and the purity is 97.1%.
Example 2
Adding water, 100mmol of phenylphosphine oxide, 210mmol of 2,4, 6-trimethyl benzaldehyde, 2g of potassium tert-butoxide and 10g of 2-chloro-1-methylpyridinium iodide into a reaction bottle in sequence, fully stirring the mixture to be uniform, reacting the mixture at room temperature for 4 hours, and detecting the completion of the reaction by TLC to obtain a mixed solution containing the intermediate compound shown in the formula I. 10gH is added4SiW12O40Adding the mixture into a mixed solution, slowly dropwise adding 5g of hydrogen peroxide into the system while stirring, reacting at room temperature for 8 hours after dropwise adding, detecting complete reaction by TLC, adding ethyl acetate for extraction, taking an organic phase, drying by anhydrous sodium sulfate, distilling under reduced pressure, and recrystallizing by using n-hexane to obtain 38.5g of target product phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, wherein the yield is 92.1%, and the purity is 95.6%.
Example 3
Adding water, 100mmol of phenylphosphine oxide, 300mmol of 2,4, 6-trimethyl benzaldehyde, 2g of sodium tert-butoxide and 10g of 1, 2-dimethylpyridinium iodide into a reaction bottle in sequence, fully stirring the mixture to be uniform, reacting the mixture at room temperature for 2 hours, and detecting the completion of the reaction by TLC to obtain a mixed solution containing the intermediate compound shown in the formula I. Will be 10gH4SiMo12O40Adding the mixture into a mixed solution, slowly dropwise adding 5g of hydrogen peroxide into the system while stirring, reacting at room temperature for 10 hours after dropwise adding is finished, detecting that the reaction is complete by TLC, adding dichloromethane for extraction, taking an organic phase, drying by anhydrous sodium sulfate, distilling under reduced pressure, and recrystallizing by petroleum ether to obtain 40g of target product phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, wherein the yield is 95.7%, and the purity is 98.2%.
Example 4
Water, 100mmol of phenylphosphine oxide, 200mmol of 2,4, 6-trimethylbenzaldehyde, 2g of potassium tert-butoxide and 10g of cetylpyridinium chloride are added in sequence into a reaction flask, stirred sufficiently until uniform, reacted at room temperature for 5h, and the reaction is completed by TLC to obtain a mixed solution containing the intermediate compound of formula I. 2gH is reacted with3PW12O40Adding into the mixed solution, then slowly dropping 3g of hydrogen peroxide into the system while stirring, reacting at room temperature for 5h after dropping, detecting complete reaction by TLC, adding dichloromethane for extraction, taking an organic phase, drying by anhydrous sodium sulfate, distilling under reduced pressure, and recrystallizing by petroleum ether to obtain 38.8g of the target product phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, wherein the yield is 92.8%, and the purity is 98.4%.
Finally, it should be noted that: it should be understood that the above examples are only for clearly illustrating the present invention and are not intended to limit the embodiments. Other variations and modifications will be apparent to persons skilled in the art in light of the above description. And are neither required nor exhaustive of all embodiments. And obvious variations or modifications of the invention may be made without departing from the scope of the invention.
Claims (11)
1. A method for preparing a photoinitiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide, which is characterized by comprising the following steps:
(1) taking water as a solvent, phenyl phosphine oxide and 2,4, 6-trimethylbenzaldehyde as raw materials, and tert-butoxide and pyridinium as catalysts, and obtaining an intermediate compound shown as a formula I through reaction, wherein the formula is as follows:
(2) oxidizing formula I under the action of an oxidizing agent to obtain a target product, namely, phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide, wherein the formula is as follows:
the tert-butoxide is selected from one or more of potassium tert-butoxide, sodium tert-butoxide or magnesium tert-butoxide; the pyridinium is one or more selected from cetyl pyridinium chloride, 2-chloro-1-methylpyridinium iodide or 1, 2-dimethylpyridinium iodide.
2. The method for preparing phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide as a photoinitiator according to claim 1, wherein the step (1) specifically comprises the steps of adding phenylphosphine oxide and 2,4, 6-trimethylbenzaldehyde into water by using water as a solvent, then adding tert-butoxide and pyridinium into the mixture, uniformly stirring the mixture, and then controlling the temperature to be 0-100 ℃ and the reaction time to be 1-10 h.
3. The method for preparing the photo-initiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide according to claim 1, wherein the molar ratio of the phenyl phosphine oxide to 2,4, 6-trimethylbenzaldehyde is 1: 2-4; the molar and mass ratio of the phenylphosphine oxide to the tert-butoxide or the pyridinium is 1mol: 1-20 g: 1-100 g.
4. The method for preparing photo-initiator phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide according to claim 3, wherein the molar ratio of the phenylphosphine oxide to 2,4, 6-trimethylbenzaldehyde is 1: 2-3.
5. The preparation method of the photo-initiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide according to claim 1, wherein the step (2) specifically comprises the steps of adding an oxidant into the reaction system containing the compound of formula I obtained in the step (1) at 0-100 ℃, reacting for 1-10 hours, after the reaction is completed, adding an organic solvent for extraction, taking an organic layer, drying, distilling under reduced pressure, and recrystallizing to obtain the target product phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide; the oxidant is selected from one or more of hydrogen peroxide, sodium peroxide, potassium peroxide, magnesium peroxide, calcium peroxide or perdiphosphonic acid.
6. The method of claim 5, wherein the step (2) further comprises using heteropoly acid or heteropoly acid salt as catalyst.
7. The method of claim 6 wherein said heteropoly acid or heteropoly acid salt is selected from H4SiW12O40、H3PW12O40、H4SiMo12O40Or H3PMo12O40One or more of (a).
8. The method of claim 5, wherein the organic solvent is one or more selected from ethyl acetate and dichloromethane.
9. The preparation method of the photoinitiator phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide according to claim 6, wherein the molar and mass ratio of the phenyl phosphine oxide to the oxidant and the catalyst is 1mol: 1-50 g: 1-100 g.
10. The method for preparing phenyl bis (2,4, 6-trimethylbenzoyl) phosphine oxide as a photoinitiator according to claim 2 or 5, wherein the reaction temperature of the step (1) and the reaction temperature of the step (2) are both room temperature.
11. The method for preparing phenylbis (2,4, 6-trimethylbenzoyl) phosphine oxide as a photoinitiator according to claim 5, wherein a solvent selected for recrystallization is n-hexane or petroleum ether.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202011500888.7A CN112538093B (en) | 2020-12-18 | 2020-12-18 | Preparation method of photoinitiator |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202011500888.7A CN112538093B (en) | 2020-12-18 | 2020-12-18 | Preparation method of photoinitiator |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN112538093A CN112538093A (en) | 2021-03-23 |
| CN112538093B true CN112538093B (en) | 2022-07-22 |
Family
ID=75018854
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202011500888.7A Active CN112538093B (en) | 2020-12-18 | 2020-12-18 | Preparation method of photoinitiator |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN112538093B (en) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6492539B1 (en) * | 1997-03-07 | 2002-12-10 | Basell Polypropylen Gmbh | Preparation of preparing substituted indanones |
| CN101830931A (en) * | 2010-04-01 | 2010-09-15 | 天津久日化学工业有限公司 | Preparation method of 2,4,6-trimethylbenzoyl-diphenyl phosphine oxide and derivative thereof |
| CN104910207A (en) * | 2015-02-12 | 2015-09-16 | 天津墨森科技有限公司 | Preparation method of di (2,4,6-trimethylbenzoyl) phenyl phosphine oxide and (2,4,6-trimethylbenzoyl) diphenyl phosphine oxide |
| CN105646578A (en) * | 2016-03-16 | 2016-06-08 | 三峡大学 | Preparation method of photoinitiator phenyl bis(2,4,6-trimethylbenzoyl)-phosphine oxide |
-
2020
- 2020-12-18 CN CN202011500888.7A patent/CN112538093B/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6492539B1 (en) * | 1997-03-07 | 2002-12-10 | Basell Polypropylen Gmbh | Preparation of preparing substituted indanones |
| CN101830931A (en) * | 2010-04-01 | 2010-09-15 | 天津久日化学工业有限公司 | Preparation method of 2,4,6-trimethylbenzoyl-diphenyl phosphine oxide and derivative thereof |
| CN104910207A (en) * | 2015-02-12 | 2015-09-16 | 天津墨森科技有限公司 | Preparation method of di (2,4,6-trimethylbenzoyl) phenyl phosphine oxide and (2,4,6-trimethylbenzoyl) diphenyl phosphine oxide |
| CN105646578A (en) * | 2016-03-16 | 2016-06-08 | 三峡大学 | Preparation method of photoinitiator phenyl bis(2,4,6-trimethylbenzoyl)-phosphine oxide |
Non-Patent Citations (1)
| Title |
|---|
| 赵永涛等.2,2'-亚甲基双(6-环己基-4-甲基苯酚)的水相合成研究.《化学工程师》.2012,(第09期),第60-62页. * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN112538093A (en) | 2021-03-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN103980310B (en) | The preparation method of two (2,4, the 6-trimethylbenzoyl) phosphine oxide of phenyl | |
| CN101830931A (en) | Preparation method of 2,4,6-trimethylbenzoyl-diphenyl phosphine oxide and derivative thereof | |
| CN107814694B (en) | Fluorene initiator, preparation method thereof, photocuring composition with fluorene initiator and application of composition in photocuring field | |
| CN101885739B (en) | Preparation method of 2,4,6-trimethylbenzene formacylphenyl phosphinicacid ethyl ester and derivative thereof | |
| CN104910207A (en) | Preparation method of di (2,4,6-trimethylbenzoyl) phenyl phosphine oxide and (2,4,6-trimethylbenzoyl) diphenyl phosphine oxide | |
| CN113248636A (en) | Thioxanthone visible light initiator, preparation method and application | |
| CN113861010B (en) | Monosubstituted and polysubstituted functional group aromatic ketone compound, preparation method thereof and photopolymerization initiator thereof | |
| CN112538093B (en) | Preparation method of photoinitiator | |
| CN113979834B (en) | Photocuring initiator, preparation method thereof and photocuring composition containing photocuring initiator | |
| CN109369406B (en) | Photocatalytic synthesis method and application of 1-hydroxycyclohexyl phenyl ketone compound | |
| CN112547122A (en) | Use of tert-butoxide and pyridinium salts in the preparation of a photoinitiator | |
| JP6628075B2 (en) | 1,4-bis (substituted oxy) -2-naphthoic acid compound and use thereof | |
| CN105541913B (en) | A kind of preparation method of 2,4,6 trimethyl benzoyl diphenyl base phosphine oxide | |
| CN119219524B (en) | A method for preparing isocyanate catalyzed by photoinitiator | |
| CN110283206A (en) | The continuous preparation method of trimethylbenzoy-dipheny phosphine oxide-type compound | |
| CN103980307A (en) | Preparation method of 2,4,6-trimethyl benzoyl phenyl ethyl phosphinate | |
| CN109896942A (en) | A kind of preparation method of alpha-alcohol ketone photoinitiator | |
| CN112094296A (en) | Preparation method of bis (2,4, 6-trimethylbenzoyl) phenylphosphine oxide | |
| CN116425795A (en) | A kind of acyl phosphine oxide photoinitiator and its preparation method and application | |
| JPH09169737A (en) | Process for producing N-methylimidazoles | |
| DE2511187A1 (en) | PROCESS FOR THE PRODUCTION OF ORGANOSILANES CONTAINING FLUORALKYL | |
| CN113493377B (en) | Continuous synthesis method of diphenyl ketone compound | |
| BE1031909B1 (en) | Synthesis method for the arylboration of olefins by photocatalysis | |
| CN110105471B (en) | Water-soluble photoinitiator and preparation method thereof | |
| CN115819455B (en) | A kind of acylphosphine oxide compound and preparation method thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| TA01 | Transfer of patent application right | ||
| TA01 | Transfer of patent application right |
Effective date of registration: 20220628 Address after: 473000 1st floor, plant 2, north area of photoelectric Incubation Park, the intersection of Xinchen West Road and Yezhuang Road, bailixi street, high tech Zone, Nanyang City, Henan Province Applicant after: Henan Rongfeng Photoelectric Technology Co.,Ltd. Address before: 251600 Shanghe Economic Development Zone, Ji'nan, Shandong Applicant before: SHANDONG ZHUOJUN INDUSTRIAL Co.,Ltd. |
|
| GR01 | Patent grant | ||
| GR01 | Patent grant |