CN112522360A - Composition, sampling device, kit and household virus detection method - Google Patents
Composition, sampling device, kit and household virus detection method Download PDFInfo
- Publication number
- CN112522360A CN112522360A CN202011302458.4A CN202011302458A CN112522360A CN 112522360 A CN112522360 A CN 112522360A CN 202011302458 A CN202011302458 A CN 202011302458A CN 112522360 A CN112522360 A CN 112522360A
- Authority
- CN
- China
- Prior art keywords
- composition
- swab
- virus
- sample
- sampling device
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/02—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving viable microorganisms
- C12Q1/24—Methods of sampling, or inoculating or spreading a sample; Methods of physically isolating an intact microorganisms
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12M—APPARATUS FOR ENZYMOLOGY OR MICROBIOLOGY; APPARATUS FOR CULTURING MICROORGANISMS FOR PRODUCING BIOMASS, FOR GROWING CELLS OR FOR OBTAINING FERMENTATION OR METABOLIC PRODUCTS, i.e. BIOREACTORS OR FERMENTERS
- C12M33/00—Means for introduction, transport, positioning, extraction, harvesting, peeling or sampling of biological material in or from the apparatus
- C12M33/02—Means for introduction, transport, positioning, extraction, harvesting, peeling or sampling of biological material in or from the apparatus by impregnation, e.g. using swabs or loops
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6806—Preparing nucleic acids for analysis, e.g. for polymerase chain reaction [PCR] assay
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/70—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving virus or bacteriophage
- C12Q1/701—Specific hybridization probes
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N1/00—Sampling; Preparing specimens for investigation
- G01N1/28—Preparing specimens for investigation including physical details of (bio-)chemical methods covered elsewhere, e.g. G01N33/50, C12Q
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- General Health & Medical Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Analytical Chemistry (AREA)
- Biochemistry (AREA)
- Immunology (AREA)
- Physics & Mathematics (AREA)
- General Engineering & Computer Science (AREA)
- Microbiology (AREA)
- Molecular Biology (AREA)
- Biotechnology (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Pathology (AREA)
- General Physics & Mathematics (AREA)
- Virology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biomedical Technology (AREA)
- Sustainable Development (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Abstract
The present invention discloses a composition for sample preservation, comprising: penicillin, antifungal, guanidinium isothiocyanate and lithium chloride dissolved in Hanks' balanced salt solution. The composition of the invention optimizes the liquid components in the composition to ensure that the sample can be preserved at normal temperature, and can be used for subsequent protein immunoassay and nucleic acid assay on the premise of ensuring virus inactivation. Meanwhile, the normal-temperature storage capacity of the device enables the device to be convenient for home sampling of patients.
Description
Technical Field
The invention belongs to the technical field of detection, and particularly relates to a composition, a sampling device, a kit and a home virus detection method.
Background
A virus is a noncellular organism that is small in size, simple in structure, contains only one nucleic acid (DNA or RNA), and must be parasitic in living cells and proliferated in a replicative manner. The virus is a non-cell life form, which is composed of a long nucleic acid chain and a protein shell, and has no own metabolic mechanism and no enzyme system. Therefore, the virus leaves the host cell and becomes a chemical substance which does not have any vital activity and can not independently propagate. Once inside the host cell, it can use the materials and energy in the cell and the ability to replicate, transcribe and translate to produce a new generation of virus as it does with the genetic information contained in its own nucleic acid. In nature, the virus is basically RNA virus, such as novel coronavirus, AIDS virus, SARS virus, MERS virus, Ebola virus (EBV), Spanish influenza virus, influenza A H1N1 virus, avian influenza virus, etc. Among them, new coronavirus, SARS virus, etc. can infect human body through upper respiratory tract. Therefore, whether the virus is infected or not can be confirmed by carrying out corresponding virus detection through the throat or the nose of the oral swab.
But the virus RNA is easy to degrade and disappear under the unprotected state; the preservation solution in the preservation tube for storing the nasopharyngeal swab in the current market can not preserve the sample at normal temperature. The specimen for virus separation and nucleic acid detection needs to be stored at 4 ℃ and detected within 24 hours; samples that cannot be detected within 24 hours need to be stored at or below-70 ℃ (if the samples are not stored at-70 ℃, the samples are stored in a refrigerator at-20 ℃), and repeated freezing and thawing should be avoided during transportation. The common virus inactivator such as formaldehyde can denature protein through crosslinking, can generate amino action with nucleotide, has good inactivation effect, but cannot be used for storing nasopharyngeal swab samples and subsequent nucleic acid detection.
Disclosure of Invention
Aiming at the defects existing in the virus preservation in the prior art, the invention provides a composition, a sampling device, a kit and a household virus detection method.
Specifically, the present invention relates to the following aspects:
1. a composition for sample preservation comprising: penicillin, antifungal, guanidinium isothiocyanate and lithium chloride dissolved in Hanks' balanced salt solution.
2. Composition according to item 1, wherein the penicillin is present in the composition in an amount of 80-200U/ml, preferably 120-150U/ml.
3. The composition according to item 1, wherein the antifungal agent is a water-soluble triazole antifungal agent, preferably fluconazole.
4. The composition according to item 3, wherein the antifungal agent is contained in the composition in an amount of 0.1 to 10. mu.g/ml, preferably 1 to 5. mu.g/ml.
5. The composition according to item 1, wherein the content of guanidinium isothiocyanate in the composition is 1.5 to 3mol/L, preferably 1.8 to 2.2 mol/L.
6. The composition of item 1, wherein the lithium chloride is in a saturated state in the composition.
7. The composition of any one of claims 1-6, wherein the sample comprises a pharyngeal swab, a nasal swab.
8. A sampling device comprising the composition of any one of items 1-7.
9. The sampling device of claim 8, further comprising a sampling tube and a swab.
10. The sampling device of claim 9, wherein the sampling tube is a foldable circular sheet.
11. The sampling device of claim 9, wherein said swab head material is polypropylene or polyamide fiber.
12. A test kit comprising the composition of any one of items 1 to 7, or the sampling device of any one of items 8 to 11.
13. Use of a composition according to any one of items 1 to 7 in the preservation of a virus.
14. Use according to item 13, wherein the virus is a new coronavirus.
15. A method for detecting a home virus, comprising: the subject self-collects the sample with the swab; hermetically preserving the swab head by immersing the swab head in a preserving fluid of a sampling device, wherein the preserving fluid comprises the composition of any one of items 1 to 7; and sending the sampling device to a detection department for detection.
16. The method of detecting according to claim 15, wherein the sample comprises a pharyngeal swab or a nasal swab.
The invention aims to realize the normal temperature preservation of the sample by optimizing the liquid components in the composition, and can be used for subsequent protein immunoassay and nucleic acid assay on the premise of ensuring virus inactivation. Meanwhile, the normal-temperature storage capacity of the device enables the device to be convenient for home sampling of patients.
Drawings
FIG. 1 is a schematic view of a foldable round sheet-type sampling tube according to the present invention;
reference numerals:
1, a self-adhesive layer; 2 sample preservation composition thickening layer; 3 a swab placement area.
Detailed Description
The present invention is further illustrated by the following examples, which are intended to be purely exemplary of the invention and are not intended to be limiting.
Unless defined otherwise, technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art. Although methods and materials similar or equivalent to those described herein can be used in experimental or practical applications, the materials and methods are described below. In case of conflict, the present specification, including definitions, will control, and the materials, methods, and examples are illustrative only and not intended to be limiting. The present invention is further illustrated by the following examples, which are not intended to limit the scope of the invention.
The present invention provides a composition for sample preservation, the composition comprising: penicillin, antifungal, guanidinium isothiocyanate and lithium chloride, said penicillin, said antifungal, said guanidinium isothiocyanate and said lithium chloride being dissolved in Hanks' balanced salt solution.
Hanks balanced salt solution is the most commonly used phosphate buffer solution called Hanks' BSS or HBSS, and mainly comprises sodium chloride, potassium chloride, sodium dihydrogen phosphate, potassium dihydrogen phosphate, sodium bicarbonate, glucose, etc., contains calcium ions and magnesium ions, and has a pH of 7.2-7.4.
In a particular embodiment, the penicillin is present in the composition in an amount of 80-200U/ml, e.g. 80U/ml, 90U/ml, 100U/ml, 110U/ml, 120U/ml, 130U/ml, 140U/ml, 150U/ml, 160U/ml, 170U/ml, 180U/ml, 190U/ml, 200U/ml, preferably 120U/ml, 150U/ml. Wherein U refers to the active titer unit of penicillin, i.e. the amount of the effective components contained in ml or per mg, and 1mg of penicillin sodium is defined as 1670 units.
In a specific embodiment, the antifungal agent is a water-soluble triazole antifungal drug. The triazole antifungal drug comprises fluconazole, itraconazole, voriconazole, posaconazole, laviconazole and the like, and is preferably fluconazole.
Of these, fluconazole was marketed in 1990 in the united states as a bis-triazole antifungal agent. The fungus is killed by inhibiting the synthesis of ergosterol by inhibiting fungal cytochrome dependent enzymes, and can be used for treating various fungal infections such as cryptococcosis, chronic mucocutaneous candidiasis and disseminated candidiasis. It has high oral bioavailability, less medicine interaction and high curative effect, and is widely used clinically. The fluconazole can treat chronic mucocutaneous candidiasis of patients with leukopenia and AIDS, and has no obvious adverse reaction. The curative effect of fluconazole in treating esophageal candidiasis of AIDS patients is obviously better than that of ketoconazole. In addition, the fluconazole has obvious curative effect on tinea versicolor, onychomycosis, candida vaginitis and candida balanitis, is an effective medicament for treating certain superficial candidiasis, tinea manuum, tinea pedis, tinea capitis, tinea corporis and deep fungal infection, particularly a first-choice medicament for treating deep and superficial generalized mycosis, cryptococcosis neoformans, meningitis and the like infected by various candida, and is safe to apply in a large amount for a long time and has few adverse reactions.
In a particular embodiment, the antifungal agent is present in an amount of 0.1 to 10. mu.g/ml, and may be, for example, 0.1. mu.g/ml, 0.5. mu.g/ml, 1. mu.g/ml, 2. mu.g/ml, 3. mu.g/ml, 4. mu.g/ml, 5. mu.g/ml, 6. mu.g/ml, 7. mu.g/ml, 8. mu.g/ml, 9. mu.g/ml, 10. mu.g/ml, preferably 1 to 5. mu.g/ml.
In a specific embodiment, the amount of guanidinium isothiocyanate in the composition is 1.5 to 3mol/L, and may be, for example, 1.5mol/L, 1.6mol/L, 1.7mol/L, 1.8mol/L, 1.9mol/L, 2mol/L, 2.1mol/L, 2.2mol/L, 2.3mol/L, 2.4mol/L, 2.5mol/L, 2.6mol/L, 2.7mol/L, 2.8mol/L, 2.9mol/L, 3mol/L, and preferably 1.8 to 2.0 mol/L.
In a specific embodiment, the lithium chloride in the composition is in a saturated state, i.e., 0.67 g/ml.
In a specific embodiment, the sample comprises a pharyngeal swab, a nasal swab.
The present invention also provides a sampling device comprising any one or more of the compositions described above. The sampling device also includes a sampling tube and a swab. The sampling tube may be any sampling tube known in the art for collecting viruses or samples, such as a sampling tube of a centrifugal tube type. The swab may be any swab known in the art for virus or sample collection, the swab head material being polypropylene or polyamide fibres.
In a specific embodiment, the sampling tube is a foldable circular sheet. As shown in FIG. 1, the collapsible round sheet sampling tube comprises a self-adhesive layer 1, a thickened layer 2 of sample holding composition, and a swab placement area 3. The foldable round sheet type sampling tube is made of filter paper materials. The non-setting adhesive layer 1 is located at the edge of the foldable round sheet type sampling tube, and is used for sticking the edge of the sampling tube after the foldable round sheet type sampling tube is folded, so as to seal the sampling tube and facilitate the storage of the sampling tube. A swab placement area 3 for placing a swab, and a thickened layer 2 of sample preserving composition surrounding the swab placement area 3 for storing a composition for sample preservation, the material being a polyethylene foam.
The invention also provides a detection kit, which comprises the composition or the sampling device.
The invention also provides the use of the composition for sample preservation in virus preservation. Preferably, the virus is a new coronavirus.
The invention also provides a method for detecting the home virus, which comprises the following steps: the subject self-collects the sample with the swab; immersing the swab head in a preserving fluid of a sampling device for sealed preservation, wherein the preserving fluid comprises the composition of any one of the above parts; and sending the sampling device to a detection department for detection.
Wherein the collected sample comprises a pharyngeal swab or a nasal swab.
Specifically, the following method can be adopted for self-collection of samples by using the swab:
1. throat swab: the two pharyngeal tonsils and the pharyngeal posterior wall at the two sides are simultaneously wiped by using plastic rod swabs with 2 polypropylene fiber heads, the force is based on the obvious protrusion of the cheek, the left side and the right side respectively scrape 20 times back and forth, the swab heads are immersed into a tube containing 3ml of virus preservation solution (isotonic saline solution, tissue culture solution or phosphate buffer solution can also be used), the tail parts are discarded, and the tube covers are screwed.
2. A nasal swab: the plastic rod swabs of 1 polypropylene fiber head are gently inserted into the nasal passage at the nasal palate, and slowly rotated and withdrawn after staying for a moment. Another plastic rod swab with a polypropylene fiber tip was used to collect the other nostril in the same manner. The two swabs were immersed in the same tube containing 3ml of the sample solution, the tail was discarded, and the cap was screwed on.
The collected sample is sent to a detection center within 48 hours at normal temperature. The sealed samples are put into sealing bags for sealing, and one sample is packed in each bag.
The composition disclosed by the invention contains penicillin, antifungal agent, guanidine isothiocyanate and lithium chloride with specific contents, and the components of the penicillin, the antifungal agent, the guanidine isothiocyanate, the lithium chloride and the like can realize normal-temperature preservation of a virus sample, and can be used for subsequent protein immunoassay and nucleic acid assay on the premise of ensuring virus inactivation. Meanwhile, the normal-temperature storage capacity of the device enables the device to be convenient for home sampling of patients.
Examples
(1) Formulation of compositions for sample preservation
Preparing Hanks balanced salt solution: stock solution A, NaCl 160g, MgSO4·7H2O 2g,KCl 8g MgCl2·6H2O 2g CaCl22.8g of the extract was dissolved in 1000ml of double distilled water; stock solution B: (1) na (Na)2HPO4·12H2O 3.04g,KH2PO41.2g, glucose 20.0g dissolved in 800ml double distilled water, (2) 0.4% phenol red solution: 0.4g of phenol red was put in a glass mortar, 0.1N NaOH was added dropwise and ground until completely dissolved, and about 10ml of 0.1N NaOH was added. And (3) sucking the dissolved phenol red into a 100ml measuring flask, washing residual phenol red liquid in a mortar by using double distilled water, merging the phenol red liquid into the measuring flask, and finally adding the double distilled water to 100 ml. (1) Mixing the solution and the solution (2), and adding double distilled water to 1000ml to obtain stock solution B. Application liquid: 1 part of stock solution A, 1 part of stock solution B and 18 parts of double distilled water.
Composition formulated for sample preservation: taking example 1 as an example, 6mg of penicillin (1000U ═ 6mg) was weighed out and completely dissolved in 90ml of prepared Hanks balanced salt solution; then 0.2mg of fluconazole is weighed and dissolved in the solution, and is completely dissolved; weighing 4ml of 5M guanidinium isothiocyanate, uniformly mixing, finally weighing 67g of lithium chloride, adding the lithium chloride into the solution, adding Hanks balanced salt solution to 100ml after complete dissolution, shaking up, and finally adjusting the pH value of the solution to 7.2-7.8 to obtain the composition for sample preservation. Other examples and comparative examples were calculated according to the content parameters in Table 1 and were prepared according to the above procedure.
Wherein the compositions of comparative examples 7-10 were lacking penicillin, fluconazole, guanidinium isothiocyanate, and lithium chloride, respectively.
TABLE 1 component contents of examples and comparative examples
(2) Test examples
Control materials: virus preservative fluid (Hangzhou Mabo biotechnology limited)
Collecting samples: the novel coronavirus was collected using sampling swabs, and each swab was added to the sample-preserving composition prepared in each of examples 1 to 5 and comparative examples 1 to 10, and the preservation solution of the wheat biovirus was used as a control group.
(3) Preservation effect
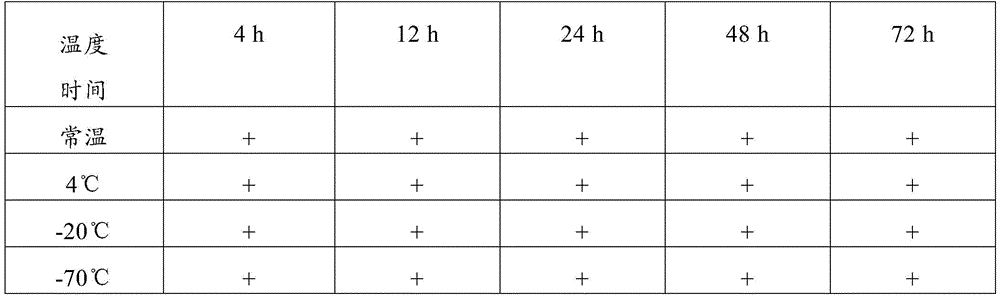
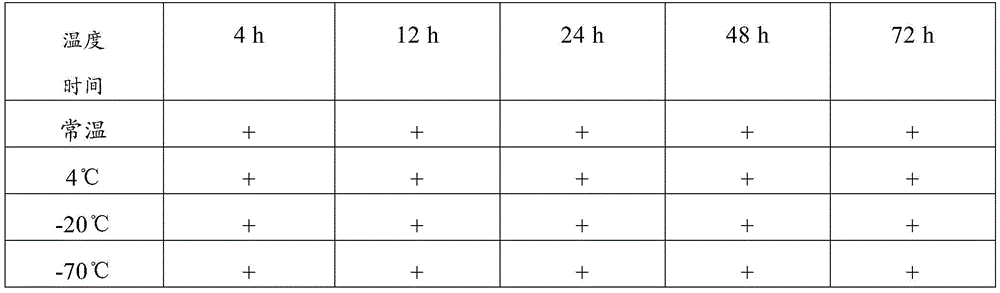
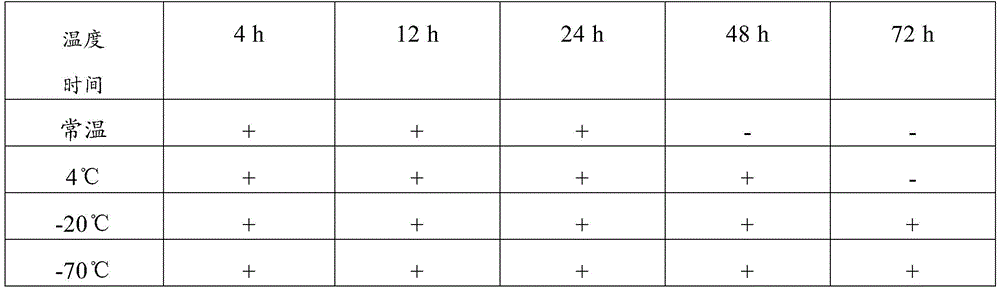
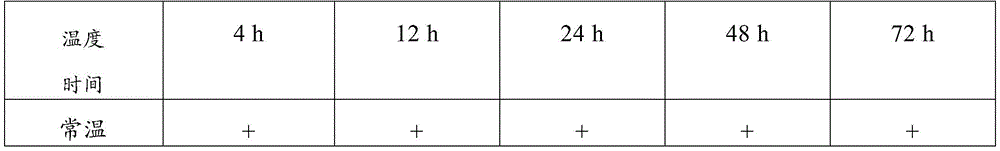
The detection method comprises the following steps: storing the collected novel coronavirus samples under different conditions (the storage temperatures are respectively normal temperature, 4 ℃, 20 ℃ and 70 ℃), performing PCR nucleic acid sensitivity experiments on the samples after the samples are stored for 4h, 12h, 24h, 48h and 72h, measuring CT values and calculating analysis results. Furthermore, the tested CT value is less than or equal to 37.0, the tested CT value is determined as positive (+), the tested CT value is not tested or the tested CT value is 0, the tested CT value is determined as negative (-), the tested CT value is determined as a gray area when the tested CT value is more than 37 and less than 40, and the test and the detection are repeated on the sample with the result in the gray area. If the repeated detection has an obvious S-shaped curve, the result is consistent with the first detection result, and the gene detection is judged to be positive (+). And recording the final detection result to verify the sample preservation effect.
The results are shown in tables 2-17:
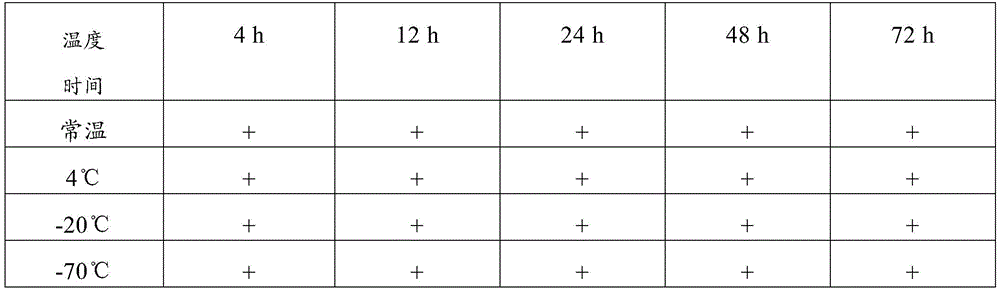
table 2 test results of the composition of example 1
Wherein, + represents positive and-represents negative, and the following tables are the same
Table 3 test results of the composition of example 2
Table 4 test results for the composition of example 3
Table 5 test results for the composition of example 4
Table 6 test results for the compositions of example 5
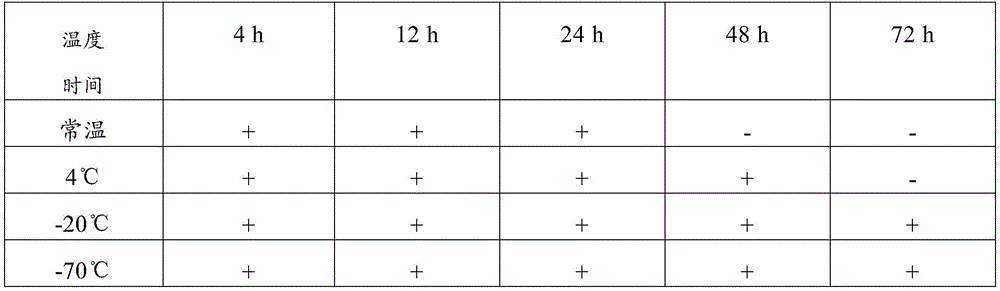
TABLE 7 examination results of the composition of comparative example 1
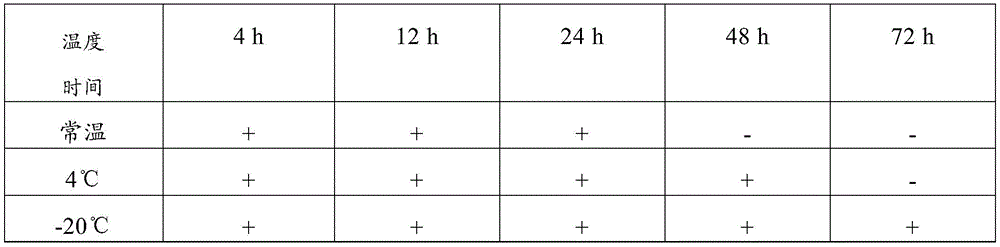
Table 8 test results of the composition of comparative example 2
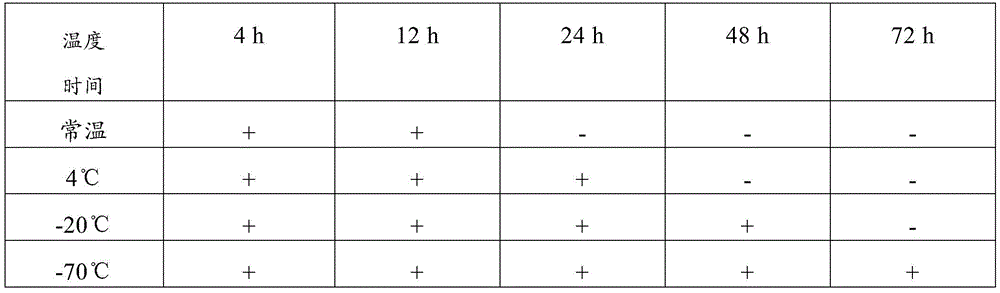
TABLE 9 test results for comparative example 3 composition
TABLE 10 test results for comparative example 4 composition
TABLE 11 test results for comparative example 5 composition
Table 12 test results of the composition of comparative example 6
TABLE 13 test results for comparative example 7 composition
TABLE 14 examination results of the comparative example 8 composition
TABLE 15 test results for comparative example 9 composition
TABLE 16 test results for comparative example 10 composition
TABLE 17 test results of comparison with virus-preserving solutions
Therefore, the sample preservation combination provided by the invention can preserve virus samples for a long time at normal temperature, has good preservation effect, has higher detection positive rate after long-time normal-temperature storage than common virus preservation solution, and has good application value.
It should be noted that the above-mentioned embodiments do not limit the present invention in any way, and all technical solutions obtained by using equivalent alternatives or equivalent variations fall within the protection scope of the present invention.
Claims (10)
1. A composition for sample preservation comprising: penicillin, antifungal, guanidinium isothiocyanate and lithium chloride dissolved in Hanks' balanced salt solution.
2. Composition according to claim 1, wherein the penicillin is present in the composition in an amount of 80-200U/ml, preferably 120-150U/ml.
3. The composition according to claim 1, characterized in that the antifungal is a water-soluble triazole antifungal drug, preferably fluconazole.
4. A composition according to claim 3, characterized in that the content of antifungal agent in the composition is 0.1-10 μ g/ml, preferably 1-5 μ g/ml.
5. The composition according to claim 1, wherein the guanidinium isothiocyanate is present in the composition in an amount of 1.5-3mol/L, preferably 1.8-2.2 mol/L.
6. The composition of claim 1, wherein the lithium chloride is in a saturated state in the composition.
7. The composition of any one of claims 1-6, wherein the sample comprises a pharyngeal swab, a nasal swab.
8. A sampling device comprising the composition of any one of claims 1-7.
9. The sampling device of claim 8, further comprising a sampling tube and a swab.
10. The sampling device of claim 9, wherein the sampling tube is a foldable circular sheet.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2020100813303 | 2020-02-06 | ||
| CN202010081330 | 2020-02-06 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN112522360A true CN112522360A (en) | 2021-03-19 |
Family
ID=74981426
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202011302458.4A Pending CN112522360A (en) | 2020-02-06 | 2020-11-19 | Composition, sampling device, kit and household virus detection method |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN112522360A (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115948502A (en) * | 2022-12-29 | 2023-04-11 | 广州精科生物技术有限公司 | A kind of inactivated virus preservation solution and its application |
Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20020037512A1 (en) * | 1997-12-10 | 2002-03-28 | Sierra Diagnostics, Inc. | Methods and reagents for preservation of DNA in bodily fluids |
| JP2004329176A (en) * | 2003-05-12 | 2004-11-25 | Plant Genome Center Co Ltd | RNA isolation method |
| US20070015165A1 (en) * | 2005-07-13 | 2007-01-18 | Sigma-Aldrich Co. | Method for the isolation of RNA from biological sources |
| US20080064108A1 (en) * | 1997-12-10 | 2008-03-13 | Tony Baker | Urine Preservation System |
| US20110027862A1 (en) * | 2009-06-29 | 2011-02-03 | Life Technologies Corporation | Sample stabilization |
| US20160108463A1 (en) * | 2007-10-01 | 2016-04-21 | Longhorn Vaccines And Diagnostics, Llc | Biological Specimen Collection and Transport System |
| CN107227306A (en) * | 2017-06-26 | 2017-10-03 | 郑州安图生物工程股份有限公司 | A kind of swab eluent with Sample preservation and inactivation function |
| CN109486904A (en) * | 2018-12-29 | 2019-03-19 | 广州阳普医疗科技股份有限公司 | A kind of whole blood RNA saves liquid and its application |
| CN110117589A (en) * | 2019-05-14 | 2019-08-13 | 东莞博奥木华基因科技有限公司 | Saliva preserving fluid and preparation method and application thereof |
| CN111363729A (en) * | 2020-03-12 | 2020-07-03 | 苏州白垩纪生物科技有限公司 | RNA virus inactivation preservation solution and application thereof |
| CN111518775A (en) * | 2020-06-18 | 2020-08-11 | 合肥铼科生物科技有限公司 | Preserving fluid for preserving virus sample at normal temperature for long time and application thereof |
| CN111549101A (en) * | 2020-06-09 | 2020-08-18 | 无锡市申瑞生物制品有限公司 | Preservation solution for biological sample nucleic acid detection and application |
| CN111718908A (en) * | 2020-08-11 | 2020-09-29 | 杭州博日科技有限公司 | Virus sample preserving fluid and preparation method and application thereof |
| CN111893160A (en) * | 2020-08-22 | 2020-11-06 | 南京健邦锦源医疗科技有限公司 | Virus preserving fluid and preparation method thereof |
-
2020
- 2020-11-19 CN CN202011302458.4A patent/CN112522360A/en active Pending
Patent Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20020037512A1 (en) * | 1997-12-10 | 2002-03-28 | Sierra Diagnostics, Inc. | Methods and reagents for preservation of DNA in bodily fluids |
| US20080064108A1 (en) * | 1997-12-10 | 2008-03-13 | Tony Baker | Urine Preservation System |
| JP2004329176A (en) * | 2003-05-12 | 2004-11-25 | Plant Genome Center Co Ltd | RNA isolation method |
| US20070015165A1 (en) * | 2005-07-13 | 2007-01-18 | Sigma-Aldrich Co. | Method for the isolation of RNA from biological sources |
| US20160108463A1 (en) * | 2007-10-01 | 2016-04-21 | Longhorn Vaccines And Diagnostics, Llc | Biological Specimen Collection and Transport System |
| US20110027862A1 (en) * | 2009-06-29 | 2011-02-03 | Life Technologies Corporation | Sample stabilization |
| CN107227306A (en) * | 2017-06-26 | 2017-10-03 | 郑州安图生物工程股份有限公司 | A kind of swab eluent with Sample preservation and inactivation function |
| CN109486904A (en) * | 2018-12-29 | 2019-03-19 | 广州阳普医疗科技股份有限公司 | A kind of whole blood RNA saves liquid and its application |
| CN110117589A (en) * | 2019-05-14 | 2019-08-13 | 东莞博奥木华基因科技有限公司 | Saliva preserving fluid and preparation method and application thereof |
| CN111363729A (en) * | 2020-03-12 | 2020-07-03 | 苏州白垩纪生物科技有限公司 | RNA virus inactivation preservation solution and application thereof |
| CN111549101A (en) * | 2020-06-09 | 2020-08-18 | 无锡市申瑞生物制品有限公司 | Preservation solution for biological sample nucleic acid detection and application |
| CN111518775A (en) * | 2020-06-18 | 2020-08-11 | 合肥铼科生物科技有限公司 | Preserving fluid for preserving virus sample at normal temperature for long time and application thereof |
| CN111718908A (en) * | 2020-08-11 | 2020-09-29 | 杭州博日科技有限公司 | Virus sample preserving fluid and preparation method and application thereof |
| CN111893160A (en) * | 2020-08-22 | 2020-11-06 | 南京健邦锦源医疗科技有限公司 | Virus preserving fluid and preparation method thereof |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115948502A (en) * | 2022-12-29 | 2023-04-11 | 广州精科生物技术有限公司 | A kind of inactivated virus preservation solution and its application |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN111518775B (en) | Preservation solution for preserving virus samples at normal temperature for long time and application thereof | |
| CN101999343B (en) | Cell preserving fluid and preparation method and use thereof | |
| Pritchard et al. | Pulau virus; a new member of the Nelson Bay orthoreovirus species isolated from fruit bats in Malaysia | |
| Intisar et al. | Peste des petits ruminants infection in domestic ruminants in Sudan | |
| CN111718908A (en) | Virus sample preserving fluid and preparation method and application thereof | |
| Keeler et al. | Use of FTA® sampling cards for molecular detection of avian influenza virus in wild birds | |
| US20140038172A1 (en) | Matrix and System for Preserving Biological Specimens for Qualitative and Quantitative Analysis | |
| CN112931487B (en) | Virus preservation solution and application thereof | |
| CN101979516B (en) | Virus sampling liquid composition | |
| US20130072387A1 (en) | Method of Pooling and/or Concentrating Biological Specimens for Analysis | |
| CN112522360A (en) | Composition, sampling device, kit and household virus detection method | |
| JP7740802B2 (en) | Storage container for cell-containing liquid and storage liquid | |
| CN109679946B (en) | Blood virus RNA protective agent and blood collection tube | |
| Nollens et al. | Characterization of a parainfluenza virus isolated from a bottlenose dolphin (Tursiops truncatus) | |
| CN112608978A (en) | Novel nucleic acid hand-free storage solution | |
| KR20240046683A (en) | Preservation solution for cell-containing solution and preservation container for cell-containing solution | |
| CN102000324A (en) | Long-efficiency and stable animal interferon solution preparation and preparation method thereof | |
| Hall et al. | Little brown bats (Myotis lucifugus) are resistant to SARS-CoV-2 infection | |
| Schock et al. | Respiratory disease due to acute Sarcocystis tenella infection in sheep | |
| CN115584363A (en) | Virus preserving fluid capable of being sterilized by irradiation | |
| CN113980954A (en) | A kind of virus RNA protective agent and its preparation method and application | |
| CN111778242A (en) | A kind of oral fluid virus DNA protective agent and preparation method and application thereof | |
| CN117165580B (en) | Composition for stabilizing nucleic acid in sample, preparation method and application thereof | |
| JP7586593B2 (en) | Blood storage composition and blood collection container | |
| WO2022101877A1 (en) | Composition for cryopreservation of biological samples for pcr |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20210319 |
|
| RJ01 | Rejection of invention patent application after publication |