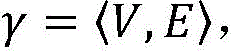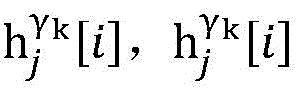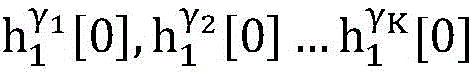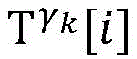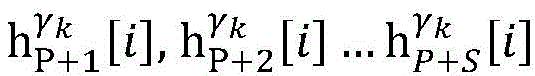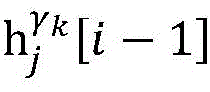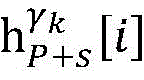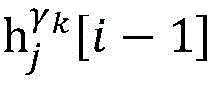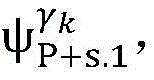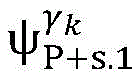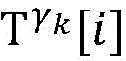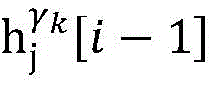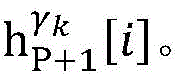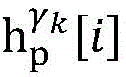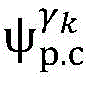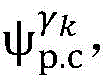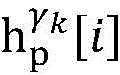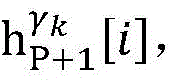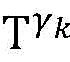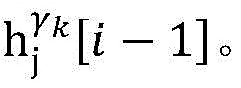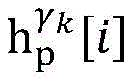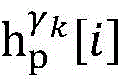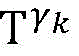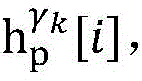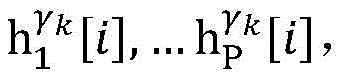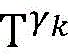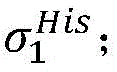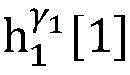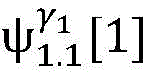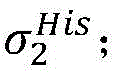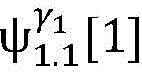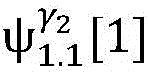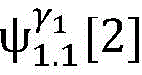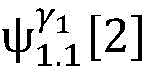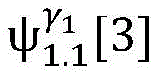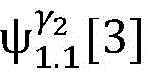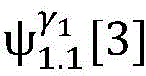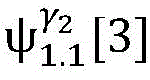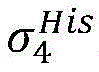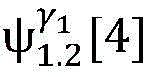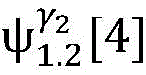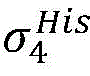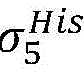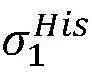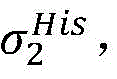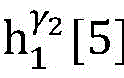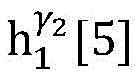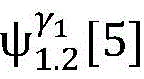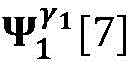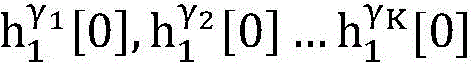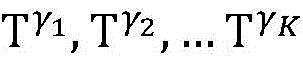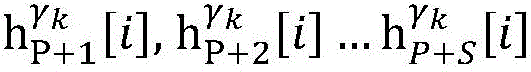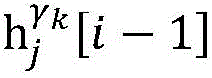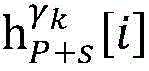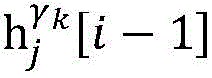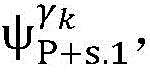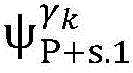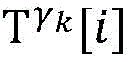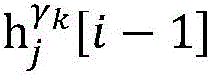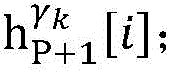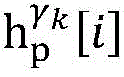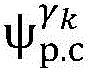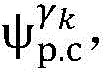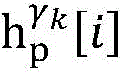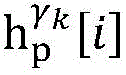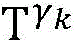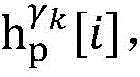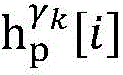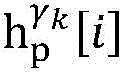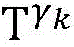CN112451088A - Model-based radio frequency ablation operation auxiliary system - Google Patents
Model-based radio frequency ablation operation auxiliary system Download PDFInfo
- Publication number
- CN112451088A CN112451088A CN202010893585.XA CN202010893585A CN112451088A CN 112451088 A CN112451088 A CN 112451088A CN 202010893585 A CN202010893585 A CN 202010893585A CN 112451088 A CN112451088 A CN 112451088A
- Authority
- CN
- China
- Prior art keywords
- heart
- model
- state machine
- cardiac
- activation signal
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B34/00—Computer-aided surgery; Manipulators or robots specially adapted for use in surgery
- A61B34/10—Computer-aided planning, simulation or modelling of surgical operations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B34/00—Computer-aided surgery; Manipulators or robots specially adapted for use in surgery
- A61B34/10—Computer-aided planning, simulation or modelling of surgical operations
- A61B2034/101—Computer-aided simulation of surgical operations
- A61B2034/105—Modelling of the patient, e.g. for ligaments or bones
Landscapes
- Health & Medical Sciences (AREA)
- Surgery (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medical Informatics (AREA)
- Robotics (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Molecular Biology (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Measurement And Recording Of Electrical Phenomena And Electrical Characteristics Of The Living Body (AREA)
- Surgical Instruments (AREA)
Abstract
The invention relates to a model-based radio frequency ablation surgery auxiliary system. The invention proposes to establish a general heart model for generating and conducting the intracardiac electric signals, combines the electric signal observation in the radio frequency ablation operation center and the related knowledge in the field of the heart electrophysiology, enumerates and models the suspected heart state and identifies the parameters; the intracardiac electric signals are labeled by using the model of the suspected heart state, so that the possible state of the current heart is estimated for a doctor, and the more rapid and accurate diagnosis is achieved.
Description
Technical Field
The invention relates to an auxiliary system which can be used in a radio frequency ablation operation and is used for assisting a doctor in judging the heart state of a patient.
Background
With the accelerated aging of the population in China, arrhythmia, especially tachyarrhythmia (Tachycardia), has become one of the important diseases harming the health of people in China. Cardiac muscle with nodeThe beating of the rhythm is triggered by the intracardiac electric signals, and the electric signal conduction path in the heart can be abstracted into a topological networkWhere V is cardiac tissue and E is the conduction pathway connecting the cardiac tissue. Electrical signals are generated by specific heart tissues and are conducted along a topological network, and arrhythmia can be caused by abnormal generation and conduction of the electrical signals, wherein the rapid arrhythmia can cause the blood pumping efficiency of the heart to be reduced, and myocardial ischemia and even death can be caused.
Radio frequency ablation surgery is one of the main treatment methods for tachyarrhythmia, and doctors can analyze the generation and conduction modes of electric signals in the heart of patients by penetrating a catheter (Catheters) into the heart through femoral veins to observe intracardiac electric signals (EGM), so as to realize diagnosis of disease conditions and locate focuses. The physician can then ablate a portion of the conductive pathway of the reentry loop by releasing a Radio Frequency (RF) signal through the catheter, severing the reentry loop while killing the corresponding heart tissue, and restoring the normal rhythm of the heart.
Radio frequency ablation procedures have shown great effectiveness in the treatment of ventricular tachycardia, atrial tachycardia and junctional tachycardia. Better therapeutic effects than drugs are also achieved in complex tachycardia cases such as Atrial Fibrillation (Atrial Fibrillation). However, rf ablation still faces some difficult challenges, resulting in too high cost, too high threshold, and low prevalence of the procedure. The major challenges facing radiofrequency ablation procedures include:
challenge one: indirection and discontinuity of patient observation
Because the radio frequency ablation operation belongs to the minimally invasive operation, a doctor can not directly observe the specific position of the catheter in the heart in the operation process, can only indirectly infer the position of the catheter in a three-dimensional space through two groups of catheter two-dimensional projections generated by X rays, and infer the position relation between the catheter and each chamber of the heart according to experience. Meanwhile, in order to control the time length of the X-ray irradiation on the patient, the X-ray projection can be only acquired intermittently, so that the position information of the catheter is discontinuous in time.
Due to the limited number of catheters that are present simultaneously inside the heart, the physician can only observe a limited number of positions simultaneously inside the heart. In response to this disadvantage, the current solution is to move an ablation catheter continuously to locate the reentry path and to ablate, using several fixed diagnostic catheters as reference points. The discontinuity in the observation of electrical signals in the heart both temporally and spatially creates a large diagnostic blind spot that makes accurate and rapid diagnosis of a patient's condition difficult.
Challenge two: mapping of current and historical observations to actual cardiac conditions of a patient
Due to the indirect nature of the rf ablation procedure, the physician needs to combine the information contained in the current and historical observations during the procedure to restore the pattern of electrical signal generation and conduction in the patient's heart. The 'brain tonifying' process puts very high requirements on the experience and the space imagination of doctors, and the mental consumption of the doctors in the operation is greatly increased.
Challenge three: the diversity of patient physiological conditions leads to uncertainties in the diagnosis
Because of the incomplete observation of the patient's heart by the physician during the rf ablation procedure, different heart conditions may be indistinguishable from observation, resulting in a historical observation that may be interpreted by a variety of suspected heart conditions. During the operation, doctors need to 1) list all suspected heart conditions which can explain historical observation, 2) extract information from historical intracardiac electric signals by using domain knowledge, eliminate the heart conditions with contradiction through reasoning analysis, and finally obtain accurate diagnosis.
The rapid and accurate diagnosis requires a great deal of medical knowledge and reasoning methods, and most of the knowledge in the field exists in a heart electrophysiology textbook in a case-by-case mode and is not formally summarized. Meanwhile, due to the limitations of short-term memory and reasoning ability of human beings, doctors are easy to miss some heart conditions which can explain historical observation when dealing with some complex heart electrophysiological conditions, or use conflicting assumptions, which leads to wrong understanding of the mechanism of some electrophysiological conditions and even wrong diagnosis and treatment.
In summary, the rf ablation procedure needs to consider various and complex heart conditions of the patient, and needs a doctor to perform complex reasoning and operation, thereby leading to the following problems:
1) the operation takes a long time
The time consumed by a single rf ablation procedure, usually in the order of 2-6 hours, presents a significant challenge to the physician's endurance and concentration, and also poses a safety risk to the patient's health.
2) High degree of dependence on doctor experience and great popularization difficulty
Due to the variety of patient conditions, the diagnosis and treatment of rf ablation procedures requires detailed analysis of specific problems. The success rate and efficiency of the operation depend on the experience of doctors to a great extent, which greatly improves the threshold of the radio frequency ablation operation and limits the popularization of the radio frequency ablation operation.
The domestic demand of the radiofrequency ablation operation tends to rise year by year, and the number of the radiofrequency ablation operation is 54559 cases in 2010 to 133897 cases in 2017, and is 1393 cases higher than that in 2016. The intelligent ablation equipment is developed by utilizing an information technology, the mechanism of complex arrhythmia is explored, the radio frequency ablation operation flow is optimized, the threshold of the radio frequency ablation operation can be reduced by providing assistance for doctors, the operation cost is reduced, and the operation safety is improved.
With the advancement of technology, medical instrument manufacturers have developed various auxiliary systems for reducing the burden on doctors during ablation procedures, aiming at the challenges of radio frequency ablation procedures.
For challenge one: indirection and discontinuity of patient state observation
Medical instrument manufacturers develop catheters capable of simultaneously observing electrocardiosignals at more positions, and spatial continuity of measurement is improved in a specific heart area. The resulting reduction in the number of catheter movements also improves the time continuity of the measurement. However, the amount of original data is increased greatly, and the processing of newly added data by the system matched with the original data is limited to visualization, so that the diagnosis assistance of doctors is limited.
For challenge two: mapping of current and historical observations to actual cardiac conditions of a patient
Medical instrument manufacturers have developed trackable catheters and associated catheter tracking systems using electromagnetic positioning principles. By using the historical catheter position information, the system can realize the following functions: 1) a doctor can perform position sampling on the inner wall of a target cavity by using a catheter in the initial stage of an operation, and generates a three-dimensional view of the inner wall of a heart by using the historical position information of the catheter, so that a reference system is provided for the subsequent operation; 2) the phase difference of signals acquired at different positions at the same phase in the heart cycle is visualized, and a doctor can deduce the conduction mode of the electrocardiosignals in the heart to a certain extent; 3) by visualizing the measured electric signal intensity at different positions in the heart, a doctor can roughly judge the position of scar tissue and has certain help for positioning the reentry ring. The auxiliary system corresponds the historical observation to the actual heart condition of the patient to a certain extent, provides effective data visualization for doctors, reduces the burden of the doctors and improves the efficiency of the radio frequency ablation operation.
There are also some model-based diagnostic attempts in the medical and academic circles, adjusting the heart model parameters using the patient's data, establishing a patient-specific heart model, and performing diagnosis and reentry loop positioning according to the simulation of the model. However, because the complexity of the model is too high, the data of the patient cannot identify all parameters of the model, and the degree of 'exclusive' is limited, so that the model can only play a certain guiding role in diagnosis. Meanwhile, due to the overlarge calculated amount, the model is only suitable for preoperative and postoperative analysis, and the real-time guidance on diagnosis and positioning in the operation process is limited.
For challenge three: diversity of patient conditions
The existing radio frequency ablation operation auxiliary system mainly provides visualization of historical data, and analysis of the historical data is in a relatively preliminary stage. The most time-consuming cardiac state estimation and suspected pathology tracking aspects of rf ablation procedures still rely on the experience of the physician and constant trial and error, and thus are the main entry point for this project.
For challenge four: the theory of the complex tachyarrhythmia mechanism is controversial
The academic and medical community has developed complex heart models based on the electrophysiological knowledge of the heart for the mechanism simulation and exploration of complex arrhythmias such as fibrillation. But also because of the complexity of the model, the model is difficult to specialize for patients, and has no guiding function for diagnosis and positioning in the operation process of a specific patient.
In summary, the existing technology has limited diagnostic and lesion location assisting effects for doctors during the rf ablation operation, and in order to improve the success rate and efficiency of the ablation operation, reduce the burden of doctors, and provide cheap and efficient medical conditions for the masses, we need to develop an rf ablation operation assisting system that provides diagnosis and treatment assistance for doctors during the operation.
Disclosure of Invention
The invention aims to assist a doctor in judging the heart state of a patient and positioning a focus in a radio frequency ablation operation, thereby improving the efficiency of the doctor and reducing the burden of the doctor.
In order to achieve the above object, the present invention provides a model-based rf ablation surgery assisting system, which is characterized in that a cardiac model is used to explain and distinguish possible discrepancies during a surgical diagnosis process, and the cardiac model is updated and excluded by using new information obtained during the surgical process, so as to provide a possible diagnosis and corresponding basis for a doctor. It comprises the following units:
an initial cardiac topology building unit for building K cardiac topologies gamma1,γ2,…γKThe method comprises the following steps:
establishing K cardiac topographies gamma according to known patient information before operation1,γ2,…γKCorresponding to K suspected heart internal electric signal conduction network structures of the patient. Each cardiac topology includes a point state machine and a line state machine: the point state machine models the key tissues which can be observed and can not be observed in the heart, including general tissues such as the sinoatrial node and the like and case-related tissues such as the entrance and the exit of a reentry ring and the like, and mainly simulates the generation and the resistance of the electrical signals of the heart tissuesSeparating; and the line state machine simulates the transmission characteristics of electric signals between key tissues in the heart, including transmission delay and the like. By adjusting the topological structures and parameters of the point state machine and the line state machine, doctors can distinguish different tachycardia mechanisms and explain corresponding pathologies.
An activation signal recognition unit for recognizing an activation signal of cardiac tissue in an intracardiac electrical signal (EGM), comprising the steps of:
during operation, the patient has N catheters in the heart, and each catheter can observe the activation signals of different peripheral tissues, and the activation signals are accumulated in time, namely the intracardiac electric signals seen by the doctor. The activation signal recognition unit recognizes the activation signal from the intracardiac electrical signal and outputs a time when the activation signal occurs and a corresponding catheter name when the activation signal is received.
A topology updating unit for updating observable point state machine positions and connections in the heart topology when the catheter is moved, comprising the steps of:
each catheter can observe the activation signal generated by not less than one heart tissue, in the system by the topology gamma of the heart1,γ2,…γKThe observable point state machine representation in (1). If certain tissue of the heart is activated in the process of electric signal conduction, the state machine of the observable point in the corresponding cardiac topological structure simultaneously sends out an activation signal. As the catheter moves, the observable cardiac tissue also changes, and the position and connectivity of the observable point state machine in the cardiac topology also needs to change. The topology updating unit updates the positions and connections of the observable point state machines in the cardiac topology according to the new position of the catheter.
The heart model screening unit enumerates and screens all heart models which can possibly explain historical activation signals after the activation signals are obtained once, and comprises the state and parameter information of the heart models, and the heart model screening unit comprises the following steps:
step 0, before the operation is started, 0 activation signals are received, and the heart topology gamma is initially determined1,γ2,…γKEstablishing K label treesLabeling each node of the tree as a heart modelRepresenting the j-th interpretable first i activation signal by gammakIs the cardiac state of the topology. Each cardiac model includes a cardiac topology γ, a cardiac model parameter range cp, and a set of electrical signal historical propagation paths Ψ. Heart modelRespectively as label treeOf a root node whose cardiac topology is initially γ1,γ2,…γKThe heart model parameter range cp includes the refractory period length of the point state machine in the topology structure, the transmission delay of the line state machine, and the like. The parameter range cp is initially [0, ∞ ] without prior information]The electrical signal history propagation path set Ψ is used for recording the generation and conduction process of the activation signal in the heart, wherein each propagation path is a tree structure, the root node of the tree is a signal source of the activation signal, a point state machine which can generate an initial activation signal in the corresponding heart topology structure is provided, and the history propagation path set Ψ is initially an empty set.
And 5, initializing p to 1.
Step 6, assuming a heart modelThere are C propagation paths in the historical set Ψ of propagation paths of the electrical signalThen the c-th propagation path to the s-th signal sourceIn the cardiac topology gammakThe corresponding point state machine in the network carries out breadth-first graph search, C is 1, … and C, and the newly searched point state machine is added into the propagation pathUntil all branches satisfy the search stop condition.
Step 8, searching the result and the ith activation signal sigma according to the graphiTemporal extraction of occurrence with respect to cardiac modelsThe linear limits of the midpoint state machine and the line state machine parameter ranges cp.
Step 10, modeling the heartThe set Ψ of historical propagation paths of the electrical signals is presented to the physician as a condition of the heart that accounts for the first i activation signals.
Step 11, according to the heart modelThe appearance sequence of observable point state machines in the electrical signal historical propagation path set psi converts the activation signal sigma of the intracardiac electrical signals1,σ2,…σiLabeled as the name of the corresponding point state machine.
Step 12, update p to p +1, assumeThe ith layer hasGenerating P heart modelsIf P is more than or equal to P, the step 13 is entered, otherwise, the step 5 is returned.
Step 13, updating j to j +1, assuming a label treeJ heart models exist in the i-1 th layer, if J is larger than or equal to J, the step 14 is carried out, and if not, the step 2 is carried out.
Step 14, updating k to k + 1; if K is larger than or equal to K, updating i to i +1, and entering the step 1, otherwise, initializing j to 1, and returning to the step 2.
Preferably, in step 0, the prior information of the parameter range of the heart model is from clinical literature.
Preferably, in the step 0, the signal source of the activation signal includes common pacemaker tissue, premature beat tissue, external electrical stimulation applied through a catheter, and the like.
Preferably, in step 6, the search stop condition includes: the point state machine which can be observed by the unmarked catheter n is searched, or the leaf node of the cardiac topological structure is reached, or the point state machine in the refractory period is searched, or the signals which are simultaneously and relatively conducted appear in the search.
Preferably, in step 7, the condition causing ambiguity includes: the initial source of the activation signal is uncertain, the observable point state machine corresponding to the activation signal is uncertain, whether the catheter is in full contact with the heart tissue is uncertain, whether the activation signal is noise is uncertain, and the like.
Preferably, in step 9, the linear constraint conflict is mainly represented by the fact that there is no intersection between the linear constraint obtained by the new information and the original linear constraint.
Preferably, in step 9, the parameter range cp is updated by linearly programming cp.
Preferably, in step 11, there may be a plurality of situations for labeling the intracardiac electrical signals, and the physician may select the heart model to display the corresponding intracardiac electrical signal labeling through the interface during the operation.
The invention proposes to establish a general heart model for generating and conducting the intracardiac electric signals, combines the electric signal observation in the radio frequency ablation operation center and the related knowledge in the field of the heart electrophysiology, enumerates and models the suspected heart state and identifies the parameters; the intracardiac electric signals are labeled by using the model of the suspected heart state, so that the possible state of the current heart is estimated for a doctor, and the more rapid and accurate diagnosis is achieved.
Drawings
FIG. 1 is a schematic view of a heart model;
FIG. 2 is a schematic view of a catheter arrangement in a cardiac RF ablation procedure;
FIG. 3 is a schematic diagram of intracardiac electrical signals;
fig. 4A to 4C are schematic views of a cardiac topology applied in a cardiac rf ablation procedure;
FIG. 5 is a roadmap for the present invention;
fig. 6 is an exemplary presentation of the use of the present invention.
Detailed Description
The invention will be further illustrated with reference to the following specific examples. It should be understood that these examples are for illustrative purposes only and are not intended to limit the scope of the present invention. Further, it should be understood that various changes or modifications of the present invention may be made by those skilled in the art after reading the teaching of the present invention, and such equivalents may fall within the scope of the present invention as defined in the appended claims.
The invention is based on a heart model that was proposed by the inventors in 2010 to simulate the condition of the patient's heart. The heart model models the transmission pattern and delay of electrical signals within the heart following the understanding of the heart in cardiac electrophysiology clinical practice. The activity of electrical signals in the heart mainly has three properties of generation, obstruction and conduction, and as shown in the figure, the inventor designs two state machines of points and lines in a heart model, which correspond to the points and the lines in fig. 1 respectively. Where the point state machine models the nature of the electrical signals produced and blocked by a particular heart tissue, and the line state machine models the delay in the transmission of electrical signals from point to point. Therefore, different heart states can be modeled by adjusting the number, connection mode and parameters of the point and line state machines, and corresponding electrocardiosignals are generated. By varying the model topology and parameters, the cardiac model proposed by the inventors can simulate different cardiac conditions.
In a cardiac rf ablation procedure, a physician observes the generation and conduction of electrical signals in the heart by moving catheters HRA, HIS, CS, ABL, RVA within the heart, diagnosing the heart condition and locating the lesion, as shown in fig. 2.
As can be seen from fig. 3, in the intracardiac electrical signals, each activation signal represents the electrical activity upon activation of cardiac tissue in the vicinity of a catheter electrode, and by sequencing and delaying the activation signals between different catheter electrodes, the physician can infer the generation and conduction pattern of the electrical signals throughout the heart.
Common fixed catheters in ablation procedures are mainly HRA, His, CS, RVA, the sources and positions of activation signals that each catheter can receive are different, and each catheter can observe activation signals from a plurality of heart tissues and is represented by a observable point state machine in the cardiac topology, as shown by the solid dots in figures 4A to 4C. While there are some known but not directly observable tissue structures in different cardiac states that contribute to the generation and conduction of electrical signals, the present invention also represents these structures as point and line state machines, as indicated by the open dots in fig. 4A-4C. Because of the limited observation points, there may be multiple cardiac conditions that can account for the current observation, by modeling these ambiguities using different cardiac models and using physiological knowledge to rule out some of them. The main steps of the invention are as follows:
step 0, establishing a heart topological structure for all suspected heart conditions by using diagnosis before operation;
and 6, repeating the steps 1 to 5.
A specific implementation of the above steps 1-6 is specifically shown by an example:
assuming that a patient is tested and that the physician suspects that the patient may have two possible cardiac conditions, sinus tachycardia or atrioventricular node reentrant tachycardia, before the operation, that need to be distinguished during the operation, it can be obtained from the above steps, as shown in fig. 6:
0) pre-operative establishment of two cardiac topologies γ from possible cardiac conditions1And gamma2The doctor sends two electrical signal stimulations with an interval of 360ms to the heart of the patient through the HRA _ A electrode;
2) Establishing a cardiac modelAndfinding a propagation path from HRA _ A to His _ A to obtainAnd labeled His _ a in both heart models;
4) By at γ1And gamma2Performing path search to the propagation pathAndis extended to obtainAndwhereinWherein a search stop occurs in one path due to the presence of electrical signals simultaneously conducted in opposite directions, as shown by the double cross in fig. 6;
5) in pairAfter the labeling is carried out,andthe search is stopped because the leaf nodes of the topological structure are reached;
6) due to the fact thatAndthe extension of the wire can not be continued,only using the new transmission path initiated by S2Andexplanation;
7) due to the fact thatAndthe interval is far greater thanAndresult inThere is a conflict of information in the process,is deleted, andthe current observation can still be interpreted due to the presence of another conduction pathway;
so far there has been no topology based γ2The patient was diagnosed with atrioventricular nodal reentry tachycardia. Propagation path set capable of realizing real-time observation and explaining historical observation by doctor in operation processAnd based on topologyStructure gamma1Label tree of
By using the invention, a doctor can obtain N increasing labeled trees in the operation process, wherein N is a possible topological structure, each leaf node at a certain moment represents a heart model capable of explaining the current heart condition, more ambiguities appear along with the operation to cause the branching of the tree, and meanwhile, the tree is cut into more details along with the gradual increase of the information quantity. The medically relevant knowledge is partly assembled in the heart model, partly in the exclusion rules and extracted model parameter constraints, and can be gradually expanded with the progress of the knowledge, so as to provide assistance for the diagnosis of the doctor and give readable explanation to the given suggestions.
Claims (8)
1. A model-based radio frequency ablation surgery auxiliary system is characterized in that a heart model is used for explaining and distinguishing possible ramifications in a surgery diagnosis process, and the heart model is updated and excluded by using new information obtained in the surgery process, so that a doctor is provided with possible diagnosis and corresponding basis, and the model-based radio frequency ablation surgery auxiliary system comprises the following units:
an initial cardiac topology building unit for building K cardiac topologies gamma1,γ2,...γKThe method comprises the following steps:
establishing K cardiac topographies gamma according to known patient information before operation1,γ2,...γKCorresponding to K kinds of suspected heart internal electric signal conduction network structures of the patient, each heart topological structure comprises a point state machine and a line state machine: the point state machine models observable and non-observable key tissues and case-related tissues in the heart and is used for simulating the generation and the blockage of the electrical signals of the heart tissues; the linear state machine is used for simulating the transmission characteristics of electric signals between key tissues in the heart; by adjusting the topological structure and parameters of the point state machine and the line state machine, doctors can distinguish different tachycardia mechanisms and explain corresponding pathologies;
An activation signal recognition unit for recognizing an activation signal of cardiac tissue in an intracardiac electrical signal, comprising the steps of:
during operation, N catheters are arranged in the heart of a patient, and activation signals of different peripheral tissues can be observed by each catheter, and the activation signals are accumulated in time, namely intracardiac electric signals seen by a doctor; the activation signal identification unit identifies an activation signal from the intracardiac electric signals and outputs the moment of occurrence of the activation signal and the corresponding catheter name when receiving the activation signal;
a topology updating unit for updating observable point state machine positions and connections in the heart topology when the catheter is moved, comprising the steps of:
each catheter can observe the activation signal generated by not less than one heart tissue, in the system by the topology gamma of the heart1,γ2,...γKA observable point state machine representation; if a certain tissue of the heart is activated in the process of electric signal conduction, simultaneously sending out an activation signal corresponding to a state machine of a measurable point in the topological structure of the heart; when the catheter moves, observable cardiac tissue also changes, and the position and connection of the observable point state machine in the cardiac topological structure also change; the topological structure updating unit updates the position and the connection of the observable point state machine in the cardiac topological structure according to the new position of the catheter;
the heart model screening unit enumerates and screens all heart models which can possibly explain historical activation signals after the activation signals are obtained once, and comprises the state and parameter information of the heart models, and the heart model screening unit comprises the following steps:
step 0, before the operation is started, 0 activation signals are received, and the heart topology gamma is initially determined1,γ2,...γKEstablishing K label treesLabeling each node of the tree as a heart model Representing the j-th interpretable first i activation signal by gammakA cardiac state that is a topological structure; each heart model comprises a heart topological structure gamma, a heart model parameter range cp and an electric signal historical propagation path set Ψ; heart modelRespectively as label treeOf a root node whose cardiac topology is initially γ1,γ2,...γK(ii) a The parameter range cp of the heart model comprises the refractory period length of a point state machine in the topological structure and the transmission delay of a line state machine, and the parameter range cp is initially [0, ∞ ] without prior information]The electrical signal historical propagation path set psi is initially an empty set;
step 1, waiting for the ith activation signal σiRecording receipt of the activation signal sigmaiInitializing k to 1 and j to 1;
step 2, if the heart model of the (i-1) th roundIf the historical propagation path set psi of the medium electric signal is an empty set, the step 3 is carried out, otherwise, the step 4 is carried out;
step 3, if the topological structure gammakThere are S point state machines that may generate the initial activation signal, assuming the ith layer of the label treeIf P heart models exist in the model, S heart models are establishedAs a label treeInEach heart model, whereinReplicationAll of (1), the historical set of propagation paths of the electrical signal augments the propagation paths A 1 st propagation path representing the S-th signal source is a tree structure taking a point state machine corresponding to the S-th signal source as a root, and initially only includes a root node, wherein S is 1, 2.
Step 4, supposing the ith layer of the label treeP heart models exist in the heart, and the heart models are duplicatedTo build a heart model
Step 5, initializing p to 1;
step 6, assumeThere are C propagation paths in the historical set Ψ of propagation paths of the electrical signalThen the c-th propagation path to the s-th signal sourceIn the cardiac topology gammakCarrying out breadth-first graph search on the corresponding point state machine in the network, wherein C is 1Until all branches meet the search stop condition;
step 7, if in pairIn the process of graph search for propagation paths in (1), ambiguity can be caused, and the ith layer of the label tree is assumed to be labeledHaving P heart models in it, the copy is made for each ambiguous caseAltering heart model content based on ambiguity information and generatingP +1 and added to the label treeIn the ith layer andshared father node
Step 8, rootSearching results according to the graph and the ith activation signal sigmaiTemporal extraction of occurrence with respect to cardiac modelsLinear limits of the midpoint state machine and line state machine parameter ranges cp;
step 9, if the extracted linear limit related to cp is equal toIf there is a conflict with cp of (2), then the label tree is selectedDeletion inOtherwise updating according to linear limitsThe parameter range cp of (1);
step 10, modeling the heartThe historical propagation path set Ψ of the electrical signals is presented to the physician as a cardiac condition that can account for the first i activation signals;
step 11, according to the heart modelThe appearance sequence of observable point state machines in the electrical signal historical propagation path set psi converts the activation signal sigma of the intracardiac electrical signals1,σ2,...σiLabeling as the name of the corresponding point state machine;
step 12, update p to p +1, assumeLayer i has generated P heart modelsIf P is more than or equal to P, entering the step 13, otherwise, returning to the step 5;
step 13, updating j to j +1, assuming a label treeJ heart models exist in the i-1 th layer, if J is larger than or equal to J, the step 14 is carried out, and if not, the step 2 is carried out;
step 14, updating k to k + 1; if K is larger than or equal to K, updating i to i +1, and entering the step 1, otherwise, initializing j to 1, and returning to the step 2.
2. The model-based rf ablation procedure assistance system of claim 1, wherein in step 0, the a priori information on the range of the heart model parameters is from a clinical literature.
3. The model-based rf ablation procedure assistance system of claim 1, wherein in step 0, the signal source of the activation signal comprises a conventional pacemaker tissue, a premature tissue, an external electrical stimulus applied through a catheter.
4. The model-based rf ablation procedure assistance system of claim 1, wherein the search stopping condition in step 6 comprises: the point state machine which can be observed by the unmarked catheter n is searched, or the leaf node of the cardiac topological structure is reached, or the point state machine in the refractory period is searched, or the signals which are simultaneously and relatively conducted appear in the search.
5. The model-based rf ablation procedure assistance system of claim 1, wherein the ambiguity-causing condition of step 7 comprises: the initial source of the activation signal is uncertain, the observable point state machine corresponding to the activation signal is uncertain, whether the catheter is in full contact with the heart tissue is uncertain, and whether the activation signal is noise is uncertain.
6. The model-based rf ablation procedure assistance system of claim 1, wherein in step 9 the conflicting linear limits are manifested by a lack of intersection between the linear limits obtained from the new information and the original linear limits.
7. The model-based rf ablation procedure assistance system of claim 1, wherein in step 9, parameter range cp update is performed by linear programming of cp.
8. The model-based rf ablation procedure assistance system of claim 1, wherein in step 11, there are multiple situations for the labeling of the intracardiac electrical signals, and the physician selects the heart model to display the corresponding intracardiac electrical signal labeling through the interface during the procedure.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2019108323066 | 2019-09-04 | ||
| CN201910832306.6A CN110522512A (en) | 2019-09-04 | 2019-09-04 | Model-Based Radiofrequency Ablation Surgical Assisting System |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN112451088A true CN112451088A (en) | 2021-03-09 |
| CN112451088B CN112451088B (en) | 2021-11-19 |
Family
ID=68666745
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201910832306.6A Pending CN110522512A (en) | 2019-09-04 | 2019-09-04 | Model-Based Radiofrequency Ablation Surgical Assisting System |
| CN202010893585.XA Expired - Fee Related CN112451088B (en) | 2019-09-04 | 2020-08-31 | Model-based radio frequency ablation operation auxiliary system |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201910832306.6A Pending CN110522512A (en) | 2019-09-04 | 2019-09-04 | Model-Based Radiofrequency Ablation Surgical Assisting System |
Country Status (1)
| Country | Link |
|---|---|
| CN (2) | CN110522512A (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN118103924A (en) * | 2021-08-09 | 2024-05-28 | 维克多医疗股份有限公司 | Organization status graphic display system |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111297474B (en) * | 2019-12-19 | 2022-11-08 | 成都迈格因科技有限公司 | Individualized positioning and mapping system for auricular fibrillation focus |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20070073179A1 (en) * | 2005-09-15 | 2007-03-29 | St. Jude Medical, Atrial Fibrillation Division, Inc. | System and Method for Three Dimensional Mapping of Electrophysiology Information |
| US20080221425A1 (en) * | 2007-03-09 | 2008-09-11 | Olson Eric S | System and method for local deformable registration of a catheter navigation system to image data or a model |
| CN101325912A (en) * | 2005-12-15 | 2008-12-17 | 皇家飞利浦电子股份有限公司 | System and method for visualizing heart morphology during electrophysiology mapping and treatment |
| CN105361945A (en) * | 2014-08-06 | 2016-03-02 | 韦伯斯特生物官能(以色列)有限公司 | Wavefront analysis based on ablation parameters |
| CN105960201A (en) * | 2014-02-25 | 2016-09-21 | 圣犹达医疗用品心脏病学部门有限公司 | System and method for local electrophysiological characterization of cardiac substrate using multi-electrode catheters |
| CN106128255A (en) * | 2016-06-14 | 2016-11-16 | 高跃技 | A kind of cardiac electrophysiology signal imitation system and analogy method thereof |
| CN106456035A (en) * | 2014-05-09 | 2017-02-22 | 波士顿科学医学有限公司 | Medical devices for mapping cardiac tissue |
| CN106901718A (en) * | 2015-12-07 | 2017-06-30 | 韦伯斯特生物官能(以色列)有限公司 | Many active regions are shown on electro-anatomical map |
-
2019
- 2019-09-04 CN CN201910832306.6A patent/CN110522512A/en active Pending
-
2020
- 2020-08-31 CN CN202010893585.XA patent/CN112451088B/en not_active Expired - Fee Related
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20070073179A1 (en) * | 2005-09-15 | 2007-03-29 | St. Jude Medical, Atrial Fibrillation Division, Inc. | System and Method for Three Dimensional Mapping of Electrophysiology Information |
| CN101325912A (en) * | 2005-12-15 | 2008-12-17 | 皇家飞利浦电子股份有限公司 | System and method for visualizing heart morphology during electrophysiology mapping and treatment |
| US20080221425A1 (en) * | 2007-03-09 | 2008-09-11 | Olson Eric S | System and method for local deformable registration of a catheter navigation system to image data or a model |
| CN105960201A (en) * | 2014-02-25 | 2016-09-21 | 圣犹达医疗用品心脏病学部门有限公司 | System and method for local electrophysiological characterization of cardiac substrate using multi-electrode catheters |
| CN106456035A (en) * | 2014-05-09 | 2017-02-22 | 波士顿科学医学有限公司 | Medical devices for mapping cardiac tissue |
| CN105361945A (en) * | 2014-08-06 | 2016-03-02 | 韦伯斯特生物官能(以色列)有限公司 | Wavefront analysis based on ablation parameters |
| CN106901718A (en) * | 2015-12-07 | 2017-06-30 | 韦伯斯特生物官能(以色列)有限公司 | Many active regions are shown on electro-anatomical map |
| CN106128255A (en) * | 2016-06-14 | 2016-11-16 | 高跃技 | A kind of cardiac electrophysiology signal imitation system and analogy method thereof |
Non-Patent Citations (2)
| Title |
|---|
| 刘鹏飞等: "基于真实解剖模型的心脏导管射频消融有限元仿真", 《中国生物医学工程学报》 * |
| 杨勇等: "新型心脏三维标测和灌注射频消融系统", 《中国医疗器械信息》 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN118103924A (en) * | 2021-08-09 | 2024-05-28 | 维克多医疗股份有限公司 | Organization status graphic display system |
| US12502158B2 (en) | 2021-08-09 | 2025-12-23 | The Vektor Group, Inc. | Tissue state graphic display system |
Also Published As
| Publication number | Publication date |
|---|---|
| CN112451088B (en) | 2021-11-19 |
| CN110522512A (en) | 2019-12-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3928702A2 (en) | Ecg based arrhythmia location identification and improved mapping | |
| US12514482B2 (en) | Identification of ablation gaps | |
| US20210393187A1 (en) | Ventricular far field estimation using autoencoder | |
| EP3936070A1 (en) | Automatic contiguity estimation of wide area circumferential ablation points | |
| JP7775549B2 (en) | atrial fibrillation | |
| JP7721353B2 (en) | Automated segmentation of anatomical structures at large circumferential ablation points | |
| US20220008126A1 (en) | Optimized ablation for persistent atrial fibrillation | |
| RU2758750C1 (en) | Re-annotation of electroanatomic map | |
| JP2022031248A (en) | System and method to detect and identify cardiac pace-mapping sites and pacing maneuvers | |
| CN112451088B (en) | Model-based radio frequency ablation operation auxiliary system | |
| JP7696776B2 (en) | Automatic Identification of Scar Regions in Organic Tissue Using Multiple Imaging Modalities | |
| JP7801108B2 (en) | Classification of abnormal cardiac activity into different classes | |
| JP2022089795A (en) | Setting an automatic interest window based on learning data analysis | |
| US20230146716A1 (en) | Digital twin of atria for atrial fibrillation patients | |
| JP2023059862A (en) | Point-list linking to three-dimensional anatomy | |
| JP2022091729A (en) | Signal and correction processing for anatomical structure mapping data | |
| JP2022104909A (en) | Post-ablation validation via visual signal | |
| JP7758264B2 (en) | Ventricular Far-Field Estimation Using Autoencoders | |
| US20250255535A1 (en) | Automated tool for vein shaving in anatomical map | |
| US20220238203A1 (en) | Adaptive navigation and registration interface for medical imaging | |
| CN114190885A (en) | Automatic identification of scar regions within organ tissue using multiple imaging modalities |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20211119 |
|
| CF01 | Termination of patent right due to non-payment of annual fee |