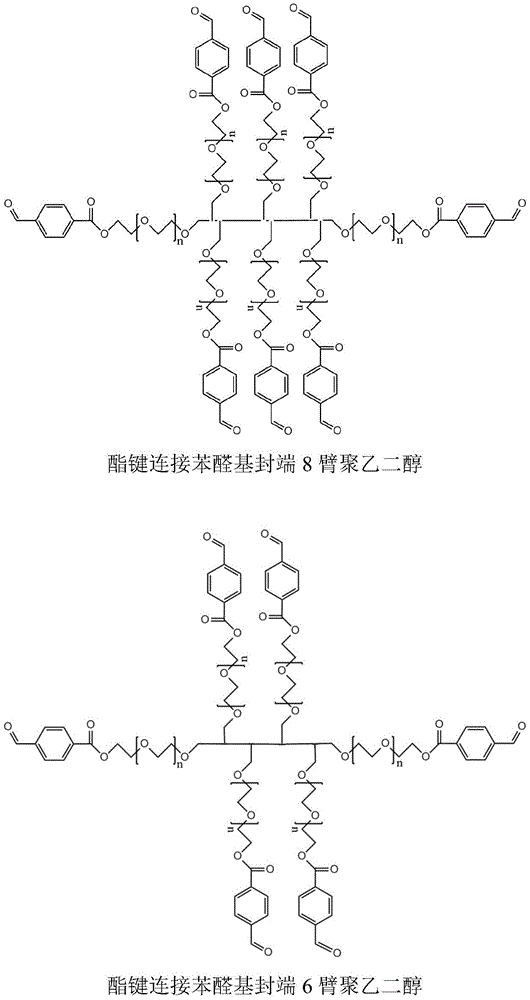
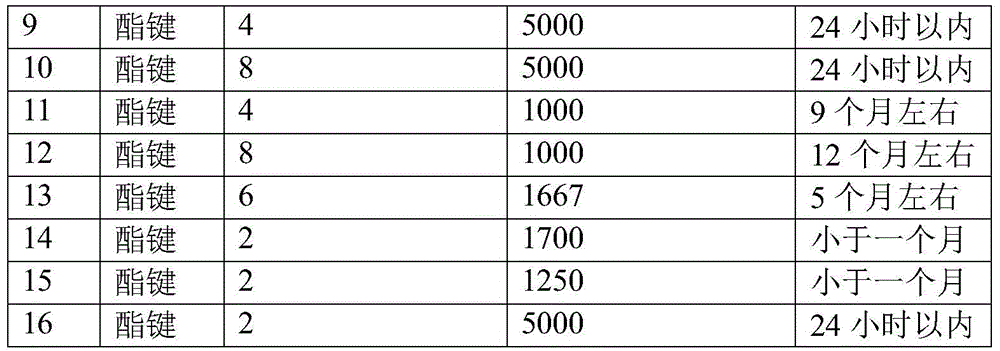
CN112225912B - Degradable medical hydrogel - Google Patents
Degradable medical hydrogel Download PDFInfo
- Publication number
- CN112225912B CN112225912B CN202011119562.XA CN202011119562A CN112225912B CN 112225912 B CN112225912 B CN 112225912B CN 202011119562 A CN202011119562 A CN 202011119562A CN 112225912 B CN112225912 B CN 112225912B
- Authority
- CN
- China
- Prior art keywords
- polyethylene glycol
- shaped multi
- arm polyethylene
- arm
- hydrogel
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000000017 hydrogel Substances 0.000 title claims abstract description 65
- 229920001223 polyethylene glycol Polymers 0.000 claims abstract description 65
- 239000002202 Polyethylene glycol Substances 0.000 claims abstract description 63
- 150000001875 compounds Chemical class 0.000 claims abstract description 19
- 125000003172 aldehyde group Chemical group 0.000 claims abstract description 18
- 238000004132 cross linking Methods 0.000 claims abstract description 7
- 238000011065 in-situ storage Methods 0.000 claims abstract description 5
- 239000000243 solution Substances 0.000 claims description 36
- 108010039918 Polylysine Proteins 0.000 claims description 20
- 229920000656 polylysine Polymers 0.000 claims description 19
- 229920002873 Polyethylenimine Polymers 0.000 claims description 13
- 239000007853 buffer solution Substances 0.000 claims description 10
- 125000003277 amino group Chemical group 0.000 claims description 8
- 238000002156 mixing Methods 0.000 claims description 8
- 238000000034 method Methods 0.000 claims description 6
- 239000000203 mixture Substances 0.000 claims description 6
- 238000002360 preparation method Methods 0.000 claims description 4
- HUMNYLRZRPPJDN-KWCOIAHCSA-N benzaldehyde Chemical group O=[11CH]C1=CC=CC=C1 HUMNYLRZRPPJDN-KWCOIAHCSA-N 0.000 claims description 3
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims 1
- 229910052739 hydrogen Inorganic materials 0.000 claims 1
- 239000001257 hydrogen Substances 0.000 claims 1
- 230000015556 catabolic process Effects 0.000 description 15
- 238000006731 degradation reaction Methods 0.000 description 15
- 239000000499 gel Substances 0.000 description 13
- 239000007864 aqueous solution Substances 0.000 description 10
- 239000008055 phosphate buffer solution Substances 0.000 description 8
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 7
- 239000004698 Polyethylene Substances 0.000 description 6
- 150000001299 aldehydes Chemical class 0.000 description 6
- HUMNYLRZRPPJDN-UHFFFAOYSA-N benzaldehyde Chemical group O=CC1=CC=CC=C1 HUMNYLRZRPPJDN-UHFFFAOYSA-N 0.000 description 6
- 229920000573 polyethylene Polymers 0.000 description 6
- 238000001727 in vivo Methods 0.000 description 5
- 230000002265 prevention Effects 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- 229910021538 borax Inorganic materials 0.000 description 4
- 239000003814 drug Substances 0.000 description 4
- 125000000524 functional group Chemical group 0.000 description 4
- 230000007774 longterm Effects 0.000 description 4
- QVLTXCYWHPZMCA-UHFFFAOYSA-N po4-po4 Chemical compound OP(O)(O)=O.OP(O)(O)=O QVLTXCYWHPZMCA-UHFFFAOYSA-N 0.000 description 4
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 4
- 239000004328 sodium tetraborate Substances 0.000 description 4
- 235000010339 sodium tetraborate Nutrition 0.000 description 4
- -1 succinimide organic acid Chemical class 0.000 description 4
- 230000032683 aging Effects 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- QNGNSVIICDLXHT-UHFFFAOYSA-N para-ethylbenzaldehyde Natural products CCC1=CC=C(C=O)C=C1 QNGNSVIICDLXHT-UHFFFAOYSA-N 0.000 description 3
- 230000002980 postoperative effect Effects 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 238000001959 radiotherapy Methods 0.000 description 3
- 238000007789 sealing Methods 0.000 description 3
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- 241000700159 Rattus Species 0.000 description 2
- 239000002262 Schiff base Substances 0.000 description 2
- 150000004753 Schiff bases Chemical class 0.000 description 2
- 208000031737 Tissue Adhesions Diseases 0.000 description 2
- 210000000683 abdominal cavity Anatomy 0.000 description 2
- 150000003934 aromatic aldehydes Chemical class 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 239000004202 carbamide Substances 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 239000000945 filler Substances 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 150000002433 hydrophilic molecules Chemical class 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 239000008363 phosphate buffer Substances 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 229920001184 polypeptide Polymers 0.000 description 2
- 108090000765 processed proteins & peptides Proteins 0.000 description 2
- 102000004196 processed proteins & peptides Human genes 0.000 description 2
- 230000005855 radiation Effects 0.000 description 2
- 230000008961 swelling Effects 0.000 description 2
- 230000017423 tissue regeneration Effects 0.000 description 2
- 238000011282 treatment Methods 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- 241000282412 Homo Species 0.000 description 1
- 238000006845 Michael addition reaction Methods 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 125000000304 alkynyl group Chemical group 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 150000001540 azides Chemical class 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 230000009172 bursting Effects 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- KZNICNPSHKQLFF-UHFFFAOYSA-N dihydromaleimide Natural products O=C1CCC(=O)N1 KZNICNPSHKQLFF-UHFFFAOYSA-N 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 229920001477 hydrophilic polymer Polymers 0.000 description 1
- 230000028993 immune response Effects 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 230000000269 nucleophilic effect Effects 0.000 description 1
- 239000012434 nucleophilic reagent Substances 0.000 description 1
- 230000006320 pegylation Effects 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 125000002467 phosphate group Chemical group [H]OP(=O)(O[H])O[*] 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 230000017854 proteolysis Effects 0.000 description 1
- 239000000565 sealant Substances 0.000 description 1
- 239000007779 soft material Substances 0.000 description 1
- 230000006641 stabilisation Effects 0.000 description 1
- 238000011105 stabilization Methods 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 229960002317 succinimide Drugs 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 229920001059 synthetic polymer Polymers 0.000 description 1
- 150000003573 thiols Chemical class 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J3/00—Processes of treating or compounding macromolecular substances
- C08J3/02—Making solutions, dispersions, lattices or gels by other methods than by solution, emulsion or suspension polymerisation techniques
- C08J3/03—Making solutions, dispersions, lattices or gels by other methods than by solution, emulsion or suspension polymerisation techniques in aqueous media
- C08J3/075—Macromolecular gels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L26/00—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form
- A61L26/0009—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form containing macromolecular materials
- A61L26/0014—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form containing macromolecular materials obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L26/00—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form
- A61L26/0009—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form containing macromolecular materials
- A61L26/0019—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form containing macromolecular materials obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L26/00—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form
- A61L26/0061—Use of materials characterised by their function or physical properties
- A61L26/008—Hydrogels or hydrocolloids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L26/00—Chemical aspects of, or use of materials for, wound dressings or bandages in liquid, gel or powder form
- A61L26/0061—Use of materials characterised by their function or physical properties
- A61L26/009—Materials resorbable by the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/16—Macromolecular materials obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/14—Macromolecular materials
- A61L27/18—Macromolecular materials obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/52—Hydrogels or hydrocolloids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L27/00—Materials for grafts or prostheses or for coating grafts or prostheses
- A61L27/50—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L27/58—Materials at least partially resorbable by the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/04—Macromolecular materials
- A61L31/048—Macromolecular materials obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/04—Macromolecular materials
- A61L31/06—Macromolecular materials obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L31/145—Hydrogels or hydrocolloids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L31/148—Materials at least partially resorbable by the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2400/00—Materials characterised by their function or physical properties
- A61L2400/06—Flowable or injectable implant compositions
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2371/00—Characterised by the use of polyethers obtained by reactions forming an ether link in the main chain; Derivatives of such polymers
- C08J2371/02—Polyalkylene oxides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2377/00—Characterised by the use of polyamides obtained by reactions forming a carboxylic amide link in the main chain; Derivatives of such polymers
- C08J2377/04—Polyamides derived from alpha-amino carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2379/00—Characterised by the use of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen, or carbon only, not provided for in groups C08J2361/00 - C08J2377/00
- C08J2379/02—Polyamines
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Dispersion Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Transplantation (AREA)
- Heart & Thoracic Surgery (AREA)
- Surgery (AREA)
- Vascular Medicine (AREA)
- Dermatology (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Medicinal Preparation (AREA)
Abstract
The invention discloses a medical hydrogel, which is formed by in-situ crosslinking of aldehyde-terminated star-shaped multi-arm polyethylene glycol and a polyamino compound, wherein the aldehyde group is connected with the star-shaped multi-arm polyethylene glycol through ester bonds, the number of arms of the aldehyde-terminated star-shaped multi-arm polyethylene glycol is 2-8, and the single-arm molecular weight is 1000-5000Da. The hydrogel with short-term degradability is obtained by selecting the aldehyde-terminated star-shaped multi-arm polyethylene glycol connected by ester bonds between aldehyde groups and the star-shaped multi-arm polyethylene glycol, wherein the star-shaped multi-arm polyethylene glycol has a specific arm number and a molecular weight range.
Description
Technical Field
The invention belongs to the technical field of biomedicine, and particularly relates to medical hydrogel which can be used as a radiation protection material for fields such as radiotherapy gaskets, postoperative tissue sealing and leakage prevention, tissue adhesion prevention, tissue filling agents, tissue repair, skin dressing, drug release and the like.
Background
The hydrogel is a soft material containing a large amount of moisture obtained by crosslinking a hydrophilic polymer. The hydrogel has excellent physical and chemical properties and biological characteristics, such as high water content, high elasticity, softness, biocompatibility and the like, and has important application value in the biomedical research fields of drug delivery, tissue engineering and the like. The injectable hydrogel is a hydrogel which has certain fluidity and can be applied by an injection method, presents phase transition between sol and gel for external stimulation (temperature, temperature/pH change and the like), is in a liquid state or a semisolid state with shear thinning property before being injected into a human body, and can form gel in situ after being injected into the human body, so that invasive surgery is not needed, the risk of infection is effectively avoided, and the pain of a patient is reduced. Various injectable PEG hydrogels that have been developed so far include amphiphilic polyester/polypeptide hydrogels with PEG as the hydrophilic segment, PEG hydrogels prepared by supramolecular interactions, and PEG hydrogels prepared by mild chemical reactions.
Polyethylene glycol (PEG) is a class of nonionic polymers, and is a class of synthetic polymers approved by the U.S. Food and Drug Administration (FDA) for clinical applications in humans due to its good biocompatibility and safety. PEG can be used as a pharmaceutic adjuvant, and can also be used for modifying (pegylating) a medicament by using PEG containing a terminal active functional group. The pegylation technology has more advantages, and particularly has the characteristics of prolonging the in vivo circulation time, enhancing the biological activity, avoiding proteolysis and reducing the immune response in the aspect of modifying protein and polypeptide medicaments. Polyethylene glycol conjugates can be prepared by attaching reactive terminal functional groups, such as amino, thiol, azide, alkynyl, and aldehyde groups, to improve the performance of polyethylene glycol.
The PEG hydrogel is one of ideal materials for tumor radiotherapy gaskets and postoperative tissue sealing, leakage prevention and the like. After the treatment effect is completed for a certain time, the hydrogel needs to be degraded in vivo, otherwise, the long-term existence in vivo can cause unnecessary side effects. The cycles of different treatments usually differ widely, and therefore the degradation time of the hydrogel system needs to be adapted to the requirements of different applications.
CN105963792A discloses a medical hydrogel composition comprising a first component and a second component, wherein the first component comprises polylysine and polyethyleneimine; the second component comprises one or more of four-arm-polyethylene glycol-succinimide glutarate, four-arm-polyethylene glycol-succinimide succinate and four-arm-polyethylene glycol-succinimide carbonate. When the gel is used, the nucleophilic reagent (polylysine and polyethyleneimine) of the first component and the electrophilic reagent (one or more of four-arm-polyethylene glycol-succinimide glutarate, four-arm-polyethylene glycol-succinimide succinate and four-arm-polyethylene glycol-succinimide carbonate) of the second component are subjected to Michael addition reaction, so that the gel can be rapidly formed and has the excellent property of low swelling. However, the succinimide organic acid ester-terminated polyethylene glycol material has a short half-life in water, is very easily hydrolyzed, requires a special technique to be stored in a powder form at room temperature for a long period of time, and is used within a short time (generally 1 hour) after dissolution, and is low in convenience.
The inventor's early research (CN 109939065A) disclosed a medical hydrogel, which was formed by in-situ crosslinking of aldehyde-terminated star-shaped multi-arm polyethylene glycol and a polyamino compound, wherein the aldehyde and the star-shaped multi-arm polyethylene glycol are connected by ether bond, amido bond, urethane bond, imine bond or urea bond. The invention utilizes the reaction of aldehyde group at the end of multi-arm polyethylene glycol and amino group of multi-amino compound to generate schiff base to generate cross-linking, thus forming the medical injectable gel. The prepared gel has short gelling time, ideal gel bursting strength and good stability in aqueous solution. The long-term stability of the benzaldehyde-terminated polyethylene glycol connected by ether bond, amide bond and ester bond in aqueous solution was studied in this patent application. 400mg of each of ether bond linkage, amide bond linkage and ester bond linkage benzaldehyde group-terminated 8-arm polyethylene glycol (M.W.10K) was dissolved in 2mL of 0.1M borate buffer solution (pH 9.2) as solution A; preparing a phosphate buffer solution containing 1.48% (w/v) of polyethyleneimine (M.W.1.8K) as a solution B; and mixing the solutions A and B in equal volume to obtain the viscous hydrogel, wherein the initial gelling time is 25 seconds, 2 seconds and 5 seconds respectively. And (3) placing the three solutions A in an oven at 37 ℃ for 1, 2, 4, 16, 24 and 40 hours, and then respectively measuring the change of the gel forming time after mixing with the solution B and the initial gel forming time. The results show that the ester-linked polyethylene glycol loses the gelling ability after 40 hours, while the ether-linked and amide-linked benzoyl-terminated 8-arm polyethylene glycol gelling time remains essentially unchanged.
Further research by the inventors of the present application (CN 109646723A) discloses a medical hydrogel, which, based on the previous research, defines the molar ratio of the aldehyde group in the star-shaped multi-arm polyethylene glycol terminated by the aldehyde group to the amino group in the polyamino compound as 0.4 to 4.4, the polyamino compound is polylysine or a mixture of polylysine and polyethyleneimine, and the molar ratio of polylysine to polyethyleneimine is 2 to 30. The hydrogel has the advantages of quick gelling, long-term stability in aqueous solution, and good swelling performance and stability after multiple irradiations.
However, the chemical bond (ether bond, amido bond, urethane bond, imine bond or urea bond) between the aldehyde group and the star-shaped multi-arm polyethylene glycol in the hydrogel is difficult to degrade, and the hydrogel has a long stabilization period (more than two years) in an aqueous solution, so that the requirement of different occasions on the degradation time of the hydrogel cannot be met. For example, in hydrogel sealants, the degradation time is typically several days to 6 months, and radiation protective hydrogels are typically 3 to 12 months. The hydrogel in the prior art cannot meet the requirement of short-term degradability.
Disclosure of Invention
Aiming at the defects of the prior art, the invention provides a degradable medical hydrogel, and all components of the hydrogel have long-term stability in aqueous solution through the regulation of a specific aqueous solution system.
The specific technical scheme of the invention is as follows:
the medical hydrogel is formed by in-situ crosslinking of aldehyde-terminated star-shaped multi-arm polyethylene glycol and a polyamino compound, wherein the aldehyde group is connected with the star-shaped multi-arm polyethylene glycol through an ester bond chemical bond, the number of arms of the aldehyde-terminated multi-arm polyethylene glycol is 2-8, and the molecular weight of a single arm is 1000-5000Da.
The number of arms of the aldehyde-terminated multi-arm polyethylene glycol is preferably 4 to 8.
The aldehyde group is selected from one or more of aromatic aldehyde and alkyl aldehyde, and is preferably a benzaldehyde group.
The molar ratio of amino groups in the polyamino compound to aldehyde groups in the aldehyde-terminated star-shaped multi-arm polyethylene glycol is 0.4-4.4, the polyamino compound is polylysine or a mixture of polylysine and polyethyleneimine, and the molar ratio of polylysine to polyethyleneimine is 2-30.
The aldehyde-terminated star-shaped multi-arm polyethylene glycol is stored for a long time in a dry powder or solution form, and the pH value of the solution is 4-6.
The invention also aims to provide the medical hydrogel which can be used for preparing radiotherapy gaskets, postoperative tissue sealing and leakage prevention, tissue adhesion prevention, tissue fillers, tissue repair, skin dressings and medicinal preparations.
Dissolving the aldehyde-terminated star-shaped multi-arm polyethylene glycol in a pH4-6 buffer solution to prepare an aldehyde-terminated star-shaped multi-arm polyethylene glycol solution; dissolving a polyamino compound in a buffer solution with pH4-10 to prepare a polyamino compound solution; mixing the two to obtain the medical hydrogel.
The above pH4-10 buffer is preferably a phosphate or borate buffer having a pH of 4-10.
The final concentration of the aldehyde-terminated star-shaped multi-arm polyethylene glycol solution is 2-30% (w/v), preferably 10-20% (w/v); the concentration of the polyamino compound solution is 0.5-20%, preferably 1-5% (w/v).
The two-component hydrogel is prepared from a first component containing nucleophilic functional groups and a second component containing electrophilic functional groups, wherein the first component is an aldehyde-terminated hydrophilic compound, the number of arms is not less than two, the hydrophilic compound is aldehyde-terminated star-shaped multi-arm polyethylene glycol, preferably 8-arm, 6-arm and 4-arm polyethylene glycol (the single-arm molecular weight is 1000-5000 Da), and the aldehyde group is one or more of aromatic aldehyde and alkyl aldehyde, preferably benzaldehyde. The aldehyde group and the polymer may be connected by a chemical bond capable of hydrolysis such as an ester bond.
The second component can be selected from compounds containing polyamino groups, including polylysine (including epsilon-polylysine and polylysine) and one or more of polyethyleneimine.
Both of the above components may be provided in the form of an aqueous solution or powder due to the stability of the aldehyde group and the amino group in the aqueous solution. When in use, the two components are respectively dissolved in the buffer solution, and then the components are mixed to obtain the hydrogel. The two components of the hydrogel can be stored in a double syringe respectively, and the two components are sprayed out through a mixing head or injected to a designated position to form gel when in use.
The invention utilizes the reaction of aldehyde group at the end of multi-arm polyethylene glycol and amino group of multi-amino compound to generate schiff base to generate cross-linking, thus forming the medical injectable gel.
The invention has the advantages that:
the invention provides medical hydrogel with good degradability compared with the prior art.
The invention obtains the hydrogel which can be degraded in a short time by selecting the star-shaped multi-arm polyethylene glycol with the end capped by aldehyde group connected with the star-shaped multi-arm polyethylene glycol through ester bond, wherein the star-shaped multi-arm polyethylene glycol has specific arm number and molecular weight range. In the prior art, the degradation time of aldehyde polyethylene glycol hydrogel connected by amide bonds and other bonds which are not easy to hydrolyze is more than 2 years, and the hydrogel can be degraded within several days to 1 year by adjusting the arm number and the molecular weight of the star-shaped multi-arm polyethylene glycol.
Drawings
FIG. 1 shows the degradation of the abdominal cavity of a rat with aldehyde-terminated polyethylene glycol hydrogel connected by ester bond. Wherein A and D are the forms of the hydrogel after being implanted for one week, B and E are the forms of the hydrogel after being implanted for 3 months, and C and F are the forms of the hydrogel after being implanted for 8 months.
Detailed Description
The following examples are provided to illustrate specific steps of the present invention, but are not intended to limit the scope of the invention.
The terms used in the present invention generally have meanings commonly understood by those of ordinary skill in the art, unless otherwise specified.
The invention is described in further detail below with reference to specific examples and data, it being understood that these examples are intended to illustrate the invention and are not intended to limit the scope of the invention in any way.
In the following examples, various procedures and methods not described in detail are conventional methods well known in the art.
The invention will be further described with reference to specific examples, but the scope of the invention is not limited thereto.
EXAMPLE 1 stability of differently formulated hydrogels in aqueous solution
1. Investigating the degradation time of different types of benzaldehyde-terminated multi-arm polyethylene glycol
400mg of various types of benzaldehyde-terminated multi-arm polyethylene glycols were dissolved in 2mL of phosphate buffer (ph 5.6) as a solution a; preparing a borax buffer solution (ph 9.2) containing polylysine (amino group to benzaldehyde group molar ratio is 1); and mixing the solution A and the solution B in equal volume to obtain the viscous hydrogel.
The result shows that the aldehyde connecting bond of the aldehyde-terminated polyethylene glycol obviously influences the stability of the hydrogel in the aqueous solution, when the connecting bond is a chemical bond which is not easy to hydrolyze, such as ether bond or amido bond, the stability of the hydrogel is more than 2 years, and when the ester bond is used as the connecting bond, the degradation time of the hydrogel is between 1 and 12 months; when ester bonds are connecting bonds, hydrogel degradation is slower when the number of the polyethylene glycol arms is larger, and hydrogel degradation is slower when the molecular weight of the single polyethylene glycol arm is lower.
2. Taking the aldehyde-terminated star-shaped multi-arm polyethylene glycol with 8 arms and a single-arm molecular weight of 1250 as an example, the influence of the molar ratio of the amino in the amino compound to the aldehyde in the aldehyde-terminated star-shaped multi-arm polyethylene glycol and the molar ratio of polylysine to polyethyleneimine on the degradation time of the hydrogel is examined
400mg of 8-arm, single-arm, aldehyde-terminated, star-shaped, multi-arm polyethylene glycol having a molecular weight of 1250Da was dissolved in 2mL of phosphate buffer (pH 5.6) to prepare solution A; preparing borax buffer solution (pH9.2) solutions of polylysine and polyethyleneimine (M.W.1.8K) with different contents as a solution B; and mixing the solution A and the solution B in equal volume to obtain the viscous hydrogel.
The result shows that the molar ratio of the amino group in the polyamino compound to the aldehyde group in the aldehyde group-terminated star-shaped multi-arm polyethylene glycol is 0.4-4.4, and the molar ratio of polylysine to polyethyleneimine is 2-30, so that the degradation time of the hydrogel is not greatly influenced.
Example 2
Referring to formulation 5 in example 1, 400mg of ester-linked benzaldehyde-terminated 8-arm polyethylene glycol 8-PEG-ester-BA (one-arm molecular weight 1250 Da) was dissolved in 2mL of a phosphate buffer solution (pH 7.4), a phosphate buffer solution (pH 6.0), a phosphate-phosphate buffer solution (pH 5.0), a phosphate-phosphate buffer solution (pH 4.0), a phosphate-phosphate buffer solution (pH 3.0), and a phosphate-phosphate buffer solution (pH 2.5), respectively, as solutions A; placing the solution A in a 60-degree oven to test the stability of the solution in an accelerated aging mode, taking out the solution at different time points, and preparing a borax buffer solution (pH 9.2) containing 2.75% (w/v) of polylysine (the molar ratio of amino to aldehyde is 1; the solution A and the solution B are mixed in equal volume to test the gelling property (gelling time).
The experimental results show that when the pH of the solution A is between 4 and 6, the gelling time is kept unchanged after aging for 26 days (equivalent to one year of storage at room temperature), and the solution stability is higher. When the gel forming time is less than or equal to 3 or greater than or equal to 7.4, the gel forming time is obviously influenced by aging, and the solution stability is poor.
Example 3
600mg of carbowax 8-PEG-ester-BA (single-arm molecular weight 1250 Da) with an ester bond connected benzaldehyde end cap and 8 arms is dissolved in 2mL of phosphate buffer solution (pH 5.6) to be used as solution A; preparing a 0.075M borax buffer solution (pH 9.2) of polylysine 4.12% (w/v) (the molar ratio of amino to aldehyde groups is 1; and mixing the solution A and the solution B in equal volume to obtain the viscous hydrogel, wherein the gelling time is 13 seconds, and the in vitro degradation time is about 10 months.
Example 4
The hydrogel of formulation 5 in example 1 was implanted into the abdominal cavity of rats and the degradation of the hydrogel in the animals was observed. The hydrogel remained essentially morphologically unchanged for 1 week (FIGS. 1A, D) and 3 months (FIGS. 1B, E); at 8 months (fig. 1C, F) the volume decreased significantly, with significant degradation; complete absorption at 11 months, no hydrogel could be found in the animals. The experimental result shows that the hydrogel of the formula has the degradation in vivo equivalent to that under in vitro conditions and can be completely absorbed in vivo within 11 months.
Claims (4)
1. The preparation method of the medical hydrogel is characterized in that the hydrogel is formed by in-situ crosslinking of benzaldehyde-terminated star-shaped multi-arm polyethylene glycol and a polyamino compound, and the preparation method comprises the following steps: dissolving the benzaldehyde-terminated star-shaped multi-arm polyethylene glycol in a pH (potential of hydrogen) 4-6 buffer solution to prepare a benzaldehyde-terminated star-shaped multi-arm polyethylene glycol solution; dissolving a polyamino compound in a buffer solution with pH4-10 to prepare a polyamino compound solution; mixing the two to obtain the medical hydrogel, wherein the benzaldehyde groups are connected with the star-shaped multi-arm polyethylene glycol through ester bonds, the number of arms of the benzaldehyde-terminated star-shaped multi-arm polyethylene glycol is 2-8, the molecular weight of a single arm is 1000-5000Da, the molar ratio of amino groups in the polyamino compound to aldehyde groups in the benzaldehyde-terminated star-shaped multi-arm polyethylene glycol is 0.4-4.4, the polyamino compound is polylysine or a mixture of polylysine and polyethyleneimine, and the molar ratio of polylysine to polyethyleneimine is 2-30.
2. The method for preparing medical hydrogel according to claim 1, wherein the number of arms of the benzaldehyde-terminated star-shaped multi-arm polyethylene glycol is 4 to 8.
3. The method for preparing medical hydrogel according to claim 1, wherein the final concentration by volume of the benzaldehyde-terminated star-shaped multi-arm polyethylene glycol solution is 2-30%, and the concentration by volume of the polyamino compound solution is 0.5-20%.
4. The preparation method of the medical hydrogel according to claim 3, wherein the final mass volume percentage concentration of the benzaldehyde-terminated star-shaped multi-arm polyethylene glycol solution is 10 to 20%, and the mass volume percentage concentration of the polyamino compound is 1 to 5%.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202011119562.XA CN112225912B (en) | 2020-10-19 | 2020-10-19 | Degradable medical hydrogel |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202011119562.XA CN112225912B (en) | 2020-10-19 | 2020-10-19 | Degradable medical hydrogel |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN112225912A CN112225912A (en) | 2021-01-15 |
| CN112225912B true CN112225912B (en) | 2022-11-29 |
Family
ID=74119064
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202011119562.XA Active CN112225912B (en) | 2020-10-19 | 2020-10-19 | Degradable medical hydrogel |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN112225912B (en) |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114907580B (en) * | 2021-02-09 | 2024-11-29 | 上海瓴就医疗科技有限公司 | Degradable double-component hydrogel and preparation method and application thereof |
| CN113461973B (en) * | 2021-07-21 | 2023-04-07 | 上海瑞凝生物科技有限公司 | Injectable medical hydrogel |
| CN114369354B (en) * | 2021-08-05 | 2024-06-18 | 上海瑞凝生物科技有限公司 | Injectable hydrogels for vascular embolization |
| CN115725088B (en) * | 2021-09-01 | 2025-06-03 | 上海瑞凝生物科技有限公司 | Hyaluronic acid-polyethylene glycol medical hydrogel |
| CN116693839B (en) * | 2022-02-25 | 2025-12-23 | 上海瑞凝生物科技有限公司 | A self-developing hydrogel |
| CN114989457B (en) * | 2022-07-06 | 2025-08-05 | 上海瑞凝生物科技有限公司 | A sodium alginate-polyethylene glycol composite hydrogel and its preparation method |
| CN115068413B (en) * | 2022-08-04 | 2024-02-23 | 上海瑞凝生物科技有限公司 | Doxorubicin hydrochloride/epirubicin hydrochloride sustained-release gel |
| CN116077744B (en) * | 2022-08-05 | 2024-11-12 | 清华大学 | Absorbable self-radiographic hydrogel and preparation method and application thereof |
| CN116082674B (en) * | 2023-03-02 | 2025-08-12 | 上海瑞凝生物科技有限公司 | Adhesive hydrogel and preparation method and application thereof |
| CN117100908A (en) * | 2023-08-22 | 2023-11-24 | 上海瑞凝生物科技有限公司 | A hydrogel kit that can be quickly dissolved on demand and a method of using the same |
| CN118420916B (en) * | 2024-07-03 | 2024-10-18 | 浙江巴泰医疗科技有限公司 | Preformed blood sealing plug and preparation method thereof |
| CN119119455A (en) * | 2024-09-11 | 2024-12-13 | 中国科学院长春应用化学研究所 | A polyethylene glycol derivative and its preparation method and polyethylene glycol hydrogel |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20100112063A1 (en) * | 2007-06-28 | 2010-05-06 | Figuly Garret D | Method for preparing a hydrogel adhesive having extended gelation time and decreased degradation time |
| CN105963792B (en) * | 2016-04-29 | 2019-03-22 | 深圳迈普再生医学科技有限公司 | Medical aquogel composition, medical aquogel and the preparation method and application thereof |
| CN107233629B (en) * | 2017-06-21 | 2020-02-14 | 深圳市第二人民医院 | Injectable hydrogels and their preparation and use |
| CN107693838A (en) * | 2017-11-09 | 2018-02-16 | 杭州亚慧生物科技有限公司 | A kind of medical injectable gel and preparation method thereof |
| CN109939065B (en) * | 2018-08-10 | 2021-08-03 | 上海瑞凝生物科技有限公司 | Medical hydrogel |
| CN109646723B (en) * | 2019-02-02 | 2020-05-22 | 上海瑞凝生物科技有限公司 | Medical hydrogel with radiation protection effect |
-
2020
- 2020-10-19 CN CN202011119562.XA patent/CN112225912B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN112225912A (en) | 2021-01-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN112225912B (en) | Degradable medical hydrogel | |
| CN109646723B (en) | Medical hydrogel with radiation protection effect | |
| CN109939065B (en) | Medical hydrogel | |
| US10322170B2 (en) | Hemostatic compositions | |
| CN114369354B (en) | Injectable hydrogels for vascular embolization | |
| EP2214731B1 (en) | Oxidized cationic polysaccharide-based polymer tissue adhesive for medical use | |
| JP5053758B2 (en) | Rapid gelling biocompatible polymer composition | |
| EP2766059B1 (en) | Hemostatic compositions | |
| US9433699B2 (en) | Hydrogel comprising catechol group-coupled chitosan or polyamine and poloxamer comprising thiol group coupled to end thereof, preparation method thereof, and hemostat using same | |
| US9259473B2 (en) | Polymer hydrogel adhesives formed with multiple crosslinking mechanisms at physiologic pH | |
| JP2004514778A (en) | Polyalkylene glycol viscosity enhancing polymer formulation | |
| US8889889B2 (en) | Crosslinked polymers with the crosslinker as therapeutic for sustained release | |
| CN113461973B (en) | Injectable medical hydrogel | |
| KR101850424B1 (en) | Posphazene-based polymer for tissue adhesion, a method for preparing the same and use thereof | |
| US20250281667A1 (en) | Medical hydrogel | |
| CN117100908A (en) | A hydrogel kit that can be quickly dissolved on demand and a method of using the same | |
| US20250312512A1 (en) | Medical hydrogel having radiation protection function | |
| WO2018005145A1 (en) | Flowable hemostatic gel composition and its methods of use | |
| KR102508680B1 (en) | Drug loaded in situ forming hydrogel | |
| Keshari et al. | In Situ Gelling Tissue Adhesive Hydrogels for Wound Closure and Tissue Regeneration |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| CB03 | Change of inventor or designer information |
Inventor after: Pan Zhen Inventor after: Chen Liang Inventor before: Pan Zhen Inventor before: Chen Liang Inventor before: Hou Sen |
|
| CB03 | Change of inventor or designer information | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |