Background
Vinyl compounds having a hydrogen atom bonded to SP2 carbon atom contained in an alkenyl group are represented by trifluoroethylene, and are expected for various uses such as cleaning gas, etching gas, refrigerant, and organic synthetic block.
As a method for producing the vinyl compound, for example, there are known: a method of using a mixed gas obtained by diluting a halogenated vinyl compound having a halogen atom bonded to a SP2 carbon atom of an alkenyl group with nitrogen as a starting material and reacting the resultant with hydrogen in the presence of a palladium catalyst (see, for example, patent document 1).
Documents of the prior art
Patent document
Patent document 1: japanese Kohyo publication No. 2013-534529
Disclosure of Invention
Technical problem to be solved by the invention
An object of the present invention is to provide a method for obtaining a vinyl compound having a hydrogen atom bonded to the SP2 carbon atom of an alkenyl group at a high conversion rate and a high selectivity without diluting a halogenated vinyl compound.
Technical solution for solving technical problem
The present invention includes the following configurations.
Item 1. a method for producing a vinyl compound having a hydrogen atom bonded to a SP2 carbon atom of an alkenyl group, comprising:
in the presence of a nickel-containing catalyst,
a step of reacting a halogenated vinyl compound having a halogen atom bonded to the SP2 carbon atom of an alkenyl group with a hydrogen-containing compound to obtain the vinyl compound.
Item 2. the production method according to item 1, wherein,
the vinyl compound is a vinyl compound represented by the general formula (1),
[ in the formula, R1、R2And R3The same or different, represent a fluorine atom, an alkyl group or a fluoroalkyl group.]。
Item 3 the production method according to item 2, wherein R in the general formula (1)1、R2And R3Is a fluorine atom or a fluoroalkyl group.
The process according to any one of the above 1 to 3, wherein,
the halogenated vinyl compound is a halogenated vinyl compound represented by the general formula (2),
[ in the formula, R1、R2And R3The same or different, represent a fluorine atom, an alkyl group or a fluoroalkyl group. X represents a halogen atom.]。
The process according to item 4, wherein X in the general formula (2) is a halogen atom other than a fluorine atom.
The process according to any one of items 1 to 5, wherein in the reaction, the contact time (W/F) of the halogenated vinyl compound with respect to the nickel-containing catalyst (total amount of the carrier and the nickel-containing catalyst when supported on the carrier) is 4 to 200g sec/cc.
The process according to any one of claims 1 to 6, wherein the reaction temperature in the reaction is 200 to 600 ℃.
The process according to any one of the above 1 to 7, wherein the hydrogen-containing compound and the halogenated vinyl compound are present in the reaction field in a ratio of 0.5 to 1.5 mol based on 1 mol of the halogenated vinyl compound.
The production method according to any one of claims 1 to 8, wherein the content of the inert gas in the raw material gas is 0 to 1% by volume.
Item 10. A composition containing a vinyl compound represented by the general formula (1) and a vinyl compound represented by the general formula (3),
[ in the formula, R1、R2And R3The same or different, represent a fluorine atom, an alkyl group or a fluoroalkyl group.];
[ in the formula, R1And R2The meaning of (a) is the same as above.],
The content of the vinyl compound represented by the general formula (1) is 90.0 to 97.0 mol% based on 100 mol% of the total composition.
The composition of claim 10, which is used as a cleaning gas, an etching gas, a refrigerant, or an organic synthetic block.
ADVANTAGEOUS EFFECTS OF INVENTION
According to the present invention, a vinyl compound having a hydrogen atom bonded to the SP2 carbon atom of an alkenyl group can be obtained at a high conversion rate and a high selectivity without diluting a halogenated vinyl compound.
Detailed Description
In the present specification, "including" is a concept including "," consisting essentially of … … (containing ease of "and" consisting of … … (containing of) ". In the present specification, the numerical range is denoted by "a to B" and means a to B.
In the present invention, "selectivity" means: the ratio (mol%) of the total molar amount of the target compound contained in the effluent gas from the outlet of the reactor to the total molar amount of the compounds other than the raw material compound in the effluent gas.
In the present invention, "conversion" means: the ratio (mol%) of the total molar amount of compounds other than the raw material compound contained in the effluent gas from the reactor outlet to the molar amount of the raw material compound supplied to the reactor.
In the present invention, "yield" means: the ratio (mol%) of the total molar amount of the target compound contained in the effluent gas from the reactor outlet to the molar amount of the raw material compound supplied to the reactor.
1. Process for producing vinyl compound
The method for producing a vinyl compound of the present invention is a method for producing a vinyl compound having a hydrogen atom bonded to a SP2 carbon atom of an alkenyl group, the method comprising: a step of reacting a halogenated vinyl compound having a halogen atom bonded to the SP2 carbon atom of an alkenyl group with a hydrogen-containing compound in the presence of a nickel-containing catalyst to obtain the vinyl compound.
Conventionally, as shown in patent document 1, a mixed gas obtained by diluting a halogenated vinyl compound having a halogen atom bonded to a SP2 carbon atom in an alkenyl group with nitrogen is used as a starting material, and the mixed gas is reacted with hydrogen in the presence of a palladium catalyst. This method is considered to have a conversion of 91% at the maximum, but since dilution of the raw material compound is necessary, it is disadvantageous in the case where yield is required to be increased. In addition, the selectivity cannot be said to be high. Further, the palladium catalyst as a catalyst has a short catalyst life, is not suitable for a long-term reaction, is expensive, and tends to increase the cost.
According to the present invention, as described above, by reacting a halogenated vinyl compound having a halogen atom bonded to the SP2 carbon atom of an alkenyl group with a hydrogen-containing compound in the presence of a nickel-containing catalyst, a vinyl compound having a hydrogen atom bonded to the SP2 carbon atom of an alkenyl group can be obtained at a high conversion rate and a high selectivity without diluting the raw material compound. In addition, the nickel catalyst used has a long catalyst life and can withstand long-term reaction.
(1-1) halogenated vinyl Compound
The halogenated vinyl compound as a substrate that can be used in the production method of the present invention is a halogenated vinyl compound in which a halogen atom is bonded to a SP2 carbon atom of an alkenyl group, and is preferably a halogenated vinyl compound represented by the general formula (2):
[ in the formula, R1、R2And R3The same or different, represent a fluorine atom, an alkyl group or a fluoroalkyl group. X represents a halogen atom.]。
In the general formula (2), as R1、R2And R3Examples of the alkyl group include alkyl groups having 1 to 10 carbon atoms, particularly 1 to 6 carbon atoms such as methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, and tert-butyl groups. The alkyl group may also be1 to 6, particularly 1 to 3 substituents having halogen atom or the like described later are used.
In the general formula (2), as R1、R2And R3Examples of the fluoroalkyl group include fluoroalkyl groups having 1 to 10 carbon atoms, particularly 1 to 6 carbon atoms (particularly perfluoroalkyl groups) such as a trifluoromethyl group and a pentafluoroethyl group.
Wherein, as R1、R2And R3From the viewpoints of conversion of the reaction, selectivity of the target product, catalyst life, and the like, a fluorine atom or fluoroalkyl group is preferable, and a fluorine atom or perfluoroalkyl group is more preferable.
In the general formula (2), examples of the halogen atom represented by X include a fluorine atom, a chlorine atom, a bromine atom and an iodine atom. Among them, from the viewpoints of conversion rate of the reaction, yield and selectivity of the target product, catalyst life and the like, a halogen atom (chlorine atom, bromine atom and iodine atom) other than a fluorine atom is preferable, and a chlorine atom is more preferable.
As the halogenated vinyl compound to be the raw material compound, R is preferable from the viewpoint that the desired vinyl compound can be produced particularly at a high conversion rate, yield and selectivity and that the catalyst life is particularly excellent1、R2And R3Both fluorine atoms and X is preferably a chlorine atom.
R mentioned above1、R2、R3And X may be the same or different.
As the halogenated vinyl compound which becomes the raw material compound satisfying the above-mentioned conditions, specific examples include:
These halogenated vinyl compounds may be used alone or in combination of 2 or more. The halogenated vinyl compound can be a known or commercially available compound.
In the method of the present invention, when the reaction is carried out by a gas-phase flow continuous method described later, the supplied raw material gas may contain an inert gas such as nitrogen or argon in addition to the halogenated vinyl compound to be the raw material compound, but since the method of the present invention can obtain a vinyl compound by reacting with a hydrogen-containing gas without diluting the halogenated vinyl compound to be the raw material compound, the total amount of the raw material gas is 100% by volume, the amount of the inert gas is preferably 0 to 1% by volume, and the raw material gas is most preferably composed of only the halogenated vinyl compound.
(1-2) Hydrogen-containing Compound
As the hydrogen-containing compound, a compound containing hydrogen and another element (for example, oxyhydrogen which is a compound of oxygen and hydrogen) is contained in addition to hydrogen. However, since the reaction of the present invention is a reaction in which a halogen atom of a halogenated vinyl compound is substituted with a hydrogen atom, it is preferable that the hydrogen-containing compound contains no hydrogen halide (hydrogen fluoride, hydrogen chloride) or the like or contains a very small amount (for example, 5% by volume or less based on the total amount of the hydrogen-containing compound). In addition, hydrogen gas is preferably used from the viewpoint of the conversion rate, yield and selectivity of the reaction. These hydrogen-containing compounds may be used alone or in combination of 2 or more.
The hydrogen-containing compound is usually supplied to the reactor in a gaseous phase using a hydrogen-containing gas, preferably together with the halogenated vinyl compound (raw material compound). The amount of the hydrogen-containing compound is preferably 0.5 to 1.5 mol, more preferably 0.6 to 1.4 mol, and still more preferably 0.7 to 1.3 mol, based on 1 mol of the halogenated vinyl compound (raw material compound) in the reaction field. In the case where the reaction is carried out in the gas phase flow continuous mode described later, the amount of the hydrogen-containing compound to be supplied is preferably 0.5 to 1.5 mol, more preferably 0.6 to 1.4 mol, and still more preferably 0.7 to 1.3 mol, based on 1 mol of the amount of the halogenated vinyl compound (raw material compound) to be supplied. By setting the content in this range, the reaction with the hydrogen-containing compound can be further promoted, the production of impurities can be further reduced, the selectivity of the vinyl compound as a product can be high, and the vinyl compound can be recovered at a high yield.
(1-3) reaction
In the step of reacting the halogenated vinyl compound represented by the general formula (2) with the hydrogen-containing compound of the present invention, for example, R is preferably selected as the raw material compound among the halogenated vinyl compounds represented by the general formula (2)1、R2And R3All are fluorine atoms, and X is preferably a chlorine atom.
In other words, the following reaction formula is preferred:
the vinyl compound is obtained by one step.
(1-3) catalyst
The step of reacting a halogenated vinyl compound represented by the general formula (2) with a hydrogen-containing compound in the present invention is a step of reacting a halogenated vinyl compound having a halogen atom bonded to the SP2 carbon atom of an alkenyl group with a hydrogen-containing compound in the presence of a nickel-containing catalyst.
The catalyst containing nickel is not particularly limited, and examples thereof include nickel metal, nickel oxide, nickel nitrate, a ligand-coordinated homogeneous nickel catalyst, and raney nickel. These nickel-containing catalysts may be used alone, or 2 or more of them may be used in combination.
The specific surface area of the nickel-containing catalyst is preferably 500 to 2000m from the viewpoint of being able to produce a vinyl compound particularly at high conversion, yield and selectivity2A concentration of 800 to 1500m2(ii) in terms of/g. In the present invention, the specific surface area of the nickel-containing catalyst is measured by the BET method. When the specific surface area of the nickel-containing catalyst is within such a range, the density of the particles of the nickel-containing catalyst is not excessively low, and therefore the target compound can be obtained with a higher selectivity. In addition, the conversion rate of the raw material compound can be further improved. In addition, inWhen the nickel-containing catalyst is supported on a carrier as described later, the specific surface area of the nickel-containing catalyst supported on the carrier is also preferably in the above range.
Such a nickel-containing catalyst can react a halogenated vinyl compound with a hydrogen-containing compound to obtain a vinyl compound with particularly high conversion, yield and selectivity, has a long catalyst life, and can be used for long-term or repeated reactions.
The nickel-containing catalyst can be synthesized by using a known or commercially available catalyst, and can be also referred to CHEMISTRY LETTERS (1990) P879-880 and the like.
In the present invention, the reaction is carried out in a gas phase by contacting the above-mentioned raw material compound with a hydrogen-containing compound in the presence of a catalyst, but in this case, the catalyst is preferably contacted with the raw material compound in a solid state (solid phase) from the viewpoint of reactivity.
In the present invention, for example, when a gas-phase continuous flow reaction is carried out, the nickel-containing catalyst may be in the form of a powder, but is preferably in the form of pellets from the viewpoint of reactivity. The nickel-containing catalyst may be used as it is or may be supported on a carrier. This increases the specific surface area of the catalyst, improves the reaction efficiency, and enables the production of a vinyl compound with particularly high conversion, yield, and selectivity. The carrier used for the support is not particularly limited, and examples thereof include carbon, alumina, zirconia, silica, titania, silica alumina, and chromium oxide. Examples of carbon include activated carbon, amorphous carbon, graphite, and diamond. These vectors may be used alone, or 2 or more of them may be used in combination. Among these, carbon and silica are preferable, carbon is more preferable, and activated carbon is even more preferable, from the viewpoint of a large specific surface area and easy support of nickel.
When the nickel-containing catalyst is supported on the carrier, the amount of the supported catalyst is not particularly limited, and from the viewpoint of particularly producing a vinyl compound at a high conversion rate, yield and selectivity, the total amount of the nickel-containing catalyst and the carrier is 100% by mass, and the nickel-containing catalyst is preferably contained in an amount of 0.1 to 75% by mass, more preferably 1 to 60% by mass.
The nickel-containing catalyst supported on the carrier preferably has a bulk density of 0.01 to 10g/mL, more preferably 0.1 to 5g/mL, from the viewpoint of enabling the production of a vinyl compound with particularly high conversion, yield and selectivity. In the present invention, the bulk density of the nickel-containing catalyst supported on the carrier is measured by a bulk density measuring instrument. When the bulk density of the nickel-containing catalyst supported on the carrier is in such a range, the density of the particles of the nickel-containing catalyst supported on the carrier is not excessively low, and therefore the target compound can be obtained with a higher selectivity. In addition, the conversion rate of the raw material compound can be further improved.
The pore volume of the nickel-containing catalyst supported on the carrier is preferably 0.1 to 1.5mL/g, more preferably 0.25 to 1.0mL/g, from the viewpoint of enabling the production of a vinyl compound with particularly high conversion, yield and selectivity. In the present invention, the pore volume of the nickel-containing catalyst supported on the carrier can be measured by the BET method. When the pore volume of the nickel-containing catalyst supported on the carrier is in such a range, the density of the particles of the nickel-containing catalyst supported on the carrier is not excessively low, and therefore the target compound can be obtained with a higher selectivity. In addition, the conversion rate of the raw material compound can be further improved.
The average pore diameter of the nickel-containing catalyst supported on the carrier is preferably 5 to 20 μm, more preferably 8 to 15 μm, from the viewpoint of enabling the production of a vinyl compound particularly at a high conversion rate, yield and selectivity. In the present invention, the average pore diameter of the nickel-containing catalyst supported on the carrier is measured by the BET method.
When the catalyst containing nickel is supported on a carrier, the method of supporting the catalyst is not particularly limited, and for example, the catalyst can be supported by adding the carrier to an aqueous solution containing a nickel compound (nickel oxide, nickel nitrate, a homogeneous nickel catalyst in which a ligand is coordinated, raney nickel, or the like) and heating the solution to remove water. Various conditions may be conventionally employed.
(1-4) reaction temperature
In the step of obtaining a vinyl compound by reacting a halogenated vinyl compound with a hydrogen-containing compound of the present invention, the reaction temperature is preferably 200 to 600 ℃, more preferably 250 to 500 ℃, and still more preferably 300 to 400 ℃ in view of particularly enabling production of a vinyl compound with high conversion, yield, and selectivity. From the viewpoint of conversion, yield and selectivity, it is preferable that the reaction temperature is set to be high (for example, 400 to 600 ℃) when the reaction time to be described later is short (for example, when the W/F is 4g sec./cc or more and less than 5g sec./cc). Further, when the reaction time described later is long (for example, when W/F is 5g sec./cc or more), the reaction temperature is low (for example, 200 to 400 ℃ C.) and there is no problem.
(1-5) reaction time
In the present invention, when the reaction is carried out in a gas phase, the reaction time is, for example, the contact time (W/F) of the raw material compound (halogenated vinyl compound) with respect to the catalyst (nickel-containing catalyst; total amount of the carrier and the nickel-containing catalyst when supported on the carrier) in the case of adopting a gas-phase flow-through method [ W: weight (g) of catalyst (nickel-containing catalyst; total amount of carrier and nickel-containing catalyst when supported on carrier), F: the longer the flow rate (cc/sec) ] of the raw material compound (halogenated vinyl compound) is set, the larger the conversion rate is; the shorter the length of the reaction, the higher the selectivity for the vinyl compound, and therefore, from the viewpoint of the conversion rate of the reaction being particularly high and the vinyl compound being obtainable in a higher yield and with a high selectivity, the preferred range is 4 to 200g sec./cc, more preferably 4.5 to 100g sec./cc, and still more preferably 5 to 50g sec./cc. Further, the above contact time means a time for which the raw material compound and the catalyst are contacted.
The above-mentioned contact time represents conditions under which the reaction is carried out in a gas phase, particularly in a gas phase continuous flow system, but can be appropriately adjusted even when the reaction is carried out in a batch system.
(1-6) reaction pressure
In the present invention, the reaction pressure in the reaction of the halogenated vinyl compound with the hydrogen-containing compound to obtain the vinyl compound is preferably-0.05 to 2MPa, more preferably-0.01 to 1MPa, and still more preferably normal pressure to 0.5MPa, from the viewpoint that the vinyl compound can be produced particularly with high conversion, yield, and selectivity. In the present invention, when the pressure is not particularly indicated, the pressure is a gauge pressure.
In the reaction of the present invention, the reactor for reacting the raw material compound (halogenated vinyl compound) with the hydrogen-containing compound in the presence of the nickel-containing catalyst is not particularly limited in shape and structure as long as it can withstand the above temperature and pressure. Examples of the reactor include a vertical reactor, a horizontal reactor, and a multitubular reactor. Examples of the material of the reactor include glass, stainless steel, iron, nickel, and iron-nickel alloy.
(1-7) reaction example
The step of reacting a halogenated vinyl compound with a hydrogen-containing compound to obtain a vinyl compound of the present invention can be carried out by any of a gas-phase continuous flow method and a batch method in which a raw material compound (halogenated vinyl compound) and a hydrogen-containing compound are continuously supplied to a reactor and a target compound (vinyl compound) is continuously discharged from the reactor. When the objective compound is accumulated in the reactor, the hydrogenation reaction proceeds further, and therefore, it is preferable to carry out the hydrogenation reaction in a gas-phase continuous flow type. In the step of reacting a halogenated vinyl compound with a hydrogen-containing compound to obtain a vinyl compound of the present invention, it is preferably carried out in a gas phase, particularly in a gas phase continuous flow system using a fixed bed reactor. When the reaction is carried out in a gas-phase continuous flow system, the apparatus, operation, and the like can be simplified, and the reaction is economically advantageous.
In the present invention, the atmosphere in the step of reacting the halogenated vinyl compound with the hydrogen-containing compound to obtain the vinyl compound is preferably an inert gas atmosphere, a hydrogen gas atmosphere, or the like, from the viewpoint of the efficiency of the reaction. Examples of the inert gas include nitrogen, helium, and argon. Among these inert gases, nitrogen is preferable from the viewpoint of cost reduction.
After the reaction, the vinyl compound represented by the general formula (1) can be obtained by purification treatment according to a conventional method if necessary.
(1-8) target Compound
The compound to be obtained in this way is a vinyl compound having a hydrogen atom bonded to the SP2 carbon atom of an alkenyl group, and is preferably a vinyl compound represented by the general formula (1):
[ in the formula, R1、R2And R3The same or different, represent a fluorine atom, an alkyl group or a fluoroalkyl group.]。
R in the general formula (1)1、R2And R3And R in the above general formula (2)1、R2And R3The same is true. Thus, specific examples of the vinyl compound represented by the general formula (1) to be produced include:
The vinyl compound obtained in this way can be effectively used in various applications such as etching gases, cleaning gases, and organic synthesis blocks for forming the foremost fine structures of semiconductors, liquid crystals, and the like. The organic synthesis block will be described later.
2. Composition comprising a metal oxide and a metal oxide
The vinyl compound can be obtained by the above-described operation, but it can also be obtained in the form of a composition containing the vinyl compound represented by the general formula (1).
For example, the composition may contain a vinyl compound represented by the general formula (3), a vinyl compound represented by the general formula (4), or the like:
[ in the formula, R1And R2The meaning of (a) is the same as above.];
[ in the formula, R1The meaning of (a) is the same as above.]。
In this case, the content of the fluorinated vinyl compound represented by the general formula (1) is preferably 90.0 to 97.2 mol%, more preferably 90.5 to 97.1 mol%, based on 100 mol% of the total amount of the composition of the present invention. The content of the vinyl compound represented by the general formula (3) is preferably 1.0 to 5.0 mol%, more preferably 1.4 to 4.0 mol%, based on 100 mol% of the total amount of the composition of the present invention. The content of the vinyl compound represented by the general formula (4) is preferably 0.1 to 2.5 mol%, more preferably 0.2 to 2.2 mol%, based on 100 mol% of the total amount of the composition of the present invention.
Further, according to the production method of the present invention, even in the case of obtaining the vinyl compound in the form of the above-mentioned composition, since the vinyl compound represented by the general formula (1) can be obtained with a high conversion rate of reaction, and with a high yield and a high selectivity, components other than the vinyl compound represented by the general formula (1) in the composition can be reduced, and therefore, the labor for purification to obtain the vinyl compound represented by the general formula (1) can be reduced.
The composition of the present invention can be effectively used not only for applications of etching gases for forming the foremost fine structures of semiconductors, liquid crystals, and the like, but also for various applications such as building blocks for organic synthesis, cleaning gases, and the like. The organic synthesis block means a substance capable of being a precursor of a compound having a highly reactive skeleton. For example, the composition of the present invention and CF3Si(CH3)3When a fluorine-containing organosilicon compound is reacted, CF can be introduced3Fluoroalkyl groups such as a fluoro group, and can be converted into a cleaning agent to be a fluorine-containing pharmaceutical intermediate.
While the embodiments of the present invention have been described above, various changes in form and details can be made without departing from the spirit and scope of the claimed invention.
[ examples ] A method for producing a compound
The following examples are provided to clarify the features of the present invention. The present invention is not limited to these examples.
Synthesis example 1: Ni-AC
5g of nickel nitrate was dissolved in 25g of pure water, and activated carbon (specific surface area 1200 m) was added thereto2(g) so that the amount of nickel nitrate based on the total amount of carbon and nickel nitrate became 20 mass%, then, the catalyst was heated at 80 ℃ for 1 hour under reduced pressure to remove water, and then dried at 200 ℃ under reduced pressure and fired at 400 ℃ for 3 hours to obtain a catalyst in which nickel was supported on activated carbon.
2Synthesis example 2: Ni-SiO
5g of nickel nitrate was dissolved in 25g of pure water, and silica (specific surface area: 800 m) was added thereto2(g) so that the amount of nickel nitrate based on the total amount of silica and nickel nitrate became 20 mass%, then, the mixture was heated at 80 ℃ for 1 hour under reduced pressure to remove water, and then dried at 200 ℃ under reduced pressure and fired at 400 ℃ for 3 hours to obtain a catalyst in which nickel was supported on silica.
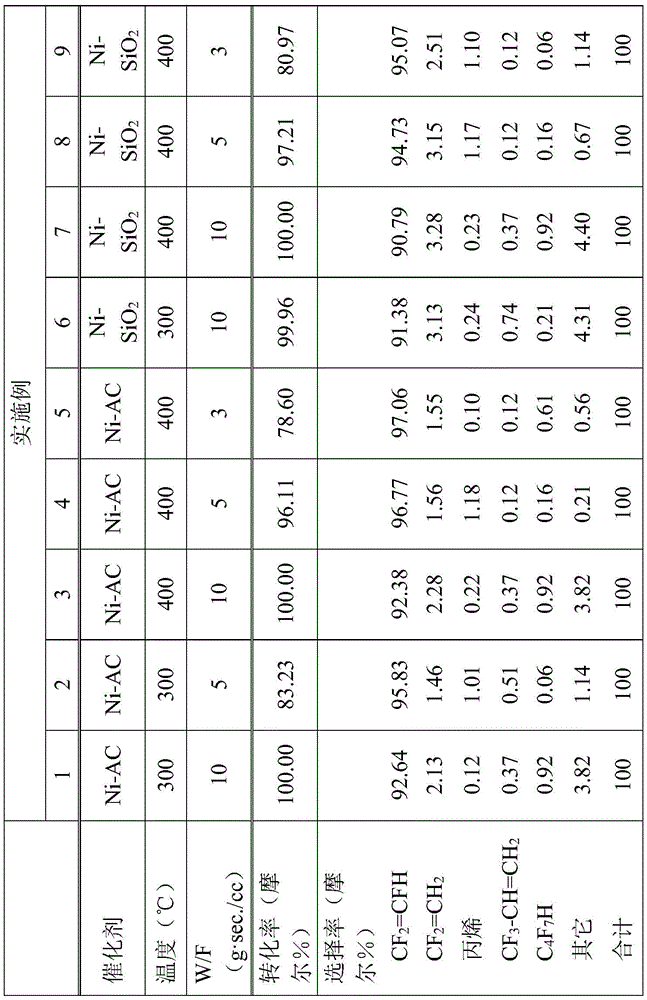
Examples 1 to 9
In the process for producing the vinyl compound of examples 1 to 9, R in the halogenated vinyl compound represented by the general formula (2) as the starting compound1、R2And R3Is a fluorine atom and X is a chlorine atom, according to the following reaction formula:
to obtain the vinyl compound.
10g of the catalyst obtained in Synthesis example 1 or 2 was charged into a SUS pipe (outer diameter: 1/2 ft) as a reaction tube. After drying at 200 ℃ for 2 hours under a nitrogen atmosphere, the pressure was set to normal pressure, and the amount of hydrogen gas supplied was adjusted to CF21 mol of ═ CFCl (raw material compound) and CF (total carbon monoxide gas content) were set to 1 mol2In such a manner that the contact time (W/F) between CFCl (starting compound) and the catalyst is 3g sec/cc, 5g sec/cc or 10g sec/cc, CF is flowed into the reaction tube2CFCl (starting compound) and hydrogen.
The reaction is carried out in a gas phase continuous flow mode.
The reaction tube was heated at 300 ℃ or 400 ℃ to start the reaction.
After 1 hour from the start of the reaction, a fraction passed through the pesticidal column was collected.
Thereafter, mass analysis was performed by gas chromatography/mass spectrometry (GC/MS) using a gas chromatograph (product name "GC-2014" manufactured by shimadzu corporation), and structural analysis by NMR spectroscopy was performed using NMR (product name "400 YH" manufactured by JEOL corporation).
From the results of mass analysis and structure analysis, it was confirmed that CF was produced as the objective compound2CFH. The conditions and results of the catalyst, temperature and contact temperature are shown in table 1.
[ TABLE 1 ]