CN112006981A - A kind of long-acting compound anthelmintic drug liquid preparation and preparation method and application thereof - Google Patents
A kind of long-acting compound anthelmintic drug liquid preparation and preparation method and application thereof Download PDFInfo
- Publication number
- CN112006981A CN112006981A CN202010748308.XA CN202010748308A CN112006981A CN 112006981 A CN112006981 A CN 112006981A CN 202010748308 A CN202010748308 A CN 202010748308A CN 112006981 A CN112006981 A CN 112006981A
- Authority
- CN
- China
- Prior art keywords
- long
- preparation
- acting compound
- liquid preparation
- anthelmintic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/216—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acids having aromatic rings, e.g. benactizyne, clofibrate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/22—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin
- A61K31/23—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin of acids having a carboxyl group bound to a chain of seven or more carbon atoms
- A61K31/231—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin of acids having a carboxyl group bound to a chain of seven or more carbon atoms having one or two double bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/365—Lactones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/4164—1,3-Diazoles
- A61K31/4184—1,3-Diazoles condensed with carbocyclic rings, e.g. benzimidazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4402—Non condensed pyridines; Hydrogenated derivatives thereof only substituted in position 2, e.g. pheniramine, bisacodyl
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/50—Pyridazines; Hydrogenated pyridazines
- A61K31/501—Pyridazines; Hydrogenated pyridazines not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/10—Alcohols; Phenols; Salts thereof, e.g. glycerol; Polyethylene glycols [PEG]; Poloxamers; PEG/POE alkyl ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/12—Carboxylic acids; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/14—Esters of carboxylic acids, e.g. fatty acid monoglycerides, medium-chain triglycerides, parabens or PEG fatty acid esters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/16—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing nitrogen, e.g. nitro-, nitroso-, azo-compounds, nitriles, cyanates
- A61K47/18—Amines; Amides; Ureas; Quaternary ammonium compounds; Amino acids; Oligopeptides having up to five amino acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/22—Heterocyclic compounds, e.g. ascorbic acid, tocopherol or pyrrolidones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/24—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing atoms other than carbon, hydrogen, oxygen, halogen, nitrogen or sulfur, e.g. cyclomethicone or phospholipids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/36—Polysaccharides; Derivatives thereof, e.g. gums, starch, alginate, dextrin, hyaluronic acid, chitosan, inulin, agar or pectin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/36—Polysaccharides; Derivatives thereof, e.g. gums, starch, alginate, dextrin, hyaluronic acid, chitosan, inulin, agar or pectin
- A61K47/38—Cellulose; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/42—Proteins; Polypeptides; Degradation products thereof; Derivatives thereof, e.g. albumin, gelatin or zein
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/44—Oils, fats or waxes according to two or more groups of A61K47/02-A61K47/42; Natural or modified natural oils, fats or waxes, e.g. castor oil, polyethoxylated castor oil, montan wax, lignite, shellac, rosin, beeswax or lanolin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
- A61P33/10—Anthelmintics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
- A61P33/14—Ectoparasiticides, e.g. scabicides
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Engineering & Computer Science (AREA)
- Inorganic Chemistry (AREA)
- Molecular Biology (AREA)
- Tropical Medicine & Parasitology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Emergency Medicine (AREA)
- Biophysics (AREA)
- Dermatology (AREA)
- Biochemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
本发明涉及一种长效复方驱虫药物液体制剂及其制备方法与应用,属兽药制剂领域。该复方制剂主要由抗寄生虫活性成分米尔贝肟、苯并咪唑类及保幼激素类成分组成,以质量百分数计:含米尔贝肟0.5~1%、苯并咪唑类5~10%、保幼激素类3~5%、高分子助悬剂0.1~5%、润湿剂0.1~5%、抗氧化剂0.01~1%,余量为分散介质。本发明将抗寄生虫药物联合使用,扩大抗虫谱,起到全面的,高效的体内外同驱的作用。The invention relates to a long-acting compound anthelmintic medicinal liquid preparation, a preparation method and application thereof, and belongs to the field of veterinary medicinal preparations. The compound preparation is mainly composed of anti-parasitic active ingredients mirbeoxime, benzimidazoles and juvenile hormones, and in mass percentage: 0.5-1% of mirbexime, 5-10% of benzimidazoles, Juvenile hormones 3-5%, macromolecular suspending agent 0.1-5%, wetting agent 0.1-5%, antioxidant 0.01-1%, and the balance is dispersion medium. The invention combines the anti-parasitic drugs to expand the anti-parasitic spectrum and plays a comprehensive and efficient role of both internal and external driving.
Description
技术领域technical field
本发明属于兽药技术领域,具体涉及一种长效复方驱虫药物液体制剂及其制备方法与应用。The invention belongs to the technical field of veterinary drugs, and particularly relates to a long-acting compound anthelmintic drug liquid preparation and a preparation method and application thereof.
背景技术Background technique
寄生虫可通过消耗宠物体内的营养物质,给宠物造成机械性损伤,使宠物因缺乏营养而生长缓慢、发育停滞、瘦弱、贫血、消瘦;或流产、死胎、失去繁殖力;或出现皮糙、皮炎、瘙痒、脱毛、体弱、免疫力、抗病力下降,容易发生疾病、传染病等。动物患寄生虫病后,由于经常和人类接触,大部分的寄生虫都可以经过宠物传染给人类,严重威胁着儿童和成人的健康。Parasites can cause mechanical damage to pets by consuming nutrients in pets, causing pets to grow slowly, stagnant, thin, anemia, and weight loss due to lack of nutrients; or miscarriage, stillbirth, loss of fertility; or appear rough skin, Dermatitis, itching, hair loss, frailty, decreased immunity and disease resistance, prone to diseases, infectious diseases, etc. After animals suffer from parasitic diseases, due to frequent contact with humans, most of the parasites can be transmitted to humans through pets, which seriously threatens the health of children and adults.
米尔贝肟是大环内酯类抗体内外寄生虫药物,主要用做犬的驱虫药;能特效防治心丝虫,高效防治钩虫、圆虫、鞭虫、蛔虫等体内寄生虫,以及毛囊虫、疥癣、虱、跳蚤等体外寄生虫,并且其对阿维菌素类药物敏感的犬毒性较小,因此,其应用和研究越来越受到重视。目前,市场上流通的米尔贝肟多为口服片剂;米尔贝肟有较高的脂溶性,易溶于甲醇、乙醇、丙酮等有机溶剂。Milbeoxime is a macrolide antibody internal and external parasite drug, which is mainly used as an anthelmintic for dogs; it can effectively prevent and treat heartworms, effectively control internal parasites such as hookworms, roundworms, whipworms, roundworms, and hair follicles. , scabies, lice, fleas and other ectoparasites, and its toxicity to abamectin-sensitive dogs is less, therefore, its application and research more and more attention. At present, most of the mirbeoxime circulating in the market are oral tablets; mirbeoxime has high fat solubility and is easily soluble in organic solvents such as methanol, ethanol and acetone.
苯并咪唑类驱虫药,具有广谱驱虫作用。线虫对其敏感,其对绦虫、吸虫也有较强作用,但所需剂量较大。苯并咪唑类驱虫药的作用机理主要是与线虫的微管蛋白结合发挥作用。苯并咪唑类与β-微管蛋白结合后,阻止其与α-微管蛋白进行多聚化组装成微管。微管是许多细胞器的基本结构单位,是有丝分裂、蛋白装配及能量代谢等细胞繁殖过程所必需。苯并咪唑类对线虫微管蛋白的亲和力显著高于哺乳动物的微管蛋白,因此对哺乳动物的毒性很小。本品不但对成虫作用强,对未成熟虫体和幼虫也有较强作用,还有杀虫卵作用。Benzimidazole anthelmintic with broad-spectrum anthelmintic effect. Nematodes are sensitive to it, and it also has a strong effect on tapeworms and flukes, but the required dose is large. The mechanism of action of benzimidazole anthelmintics is mainly by binding to the tubulin of nematodes. Binding of benzimidazoles to β-tubulin prevents its multimerization with α-tubulin to assemble into microtubules. Microtubules are the basic structural units of many organelles and are necessary for cellular reproduction processes such as mitosis, protein assembly, and energy metabolism. Benzimidazoles have a significantly higher affinity for nematode tubulin than mammalian tubulin, and therefore have little toxicity to mammals. This product not only has a strong effect on adults, but also has a strong effect on immature worms and larvae, as well as killing eggs.
昆虫的成长、发育和成熟都要受甾族化合物蜕皮激素和倍半萜类化合物保幼激素的调控或影响。保幼激素作为一类新型的几丁质合成抑制剂,其作用机理主要基于以下3个方面的协调:(1)通过在血淋巴中高浓度的存留,加速昆虫前胸腺向性激素的分泌,干扰表皮中蜕皮激素的生物合成;(2)使昆虫几丁质合成酶受到抑制,引起表皮中几丁质合成受阻而不断减少,昆虫在蜕皮时便不能形成新的表皮,因而变态受阻,成为畸形或死亡;(3)使昆虫缺少产卵所需的刺激因素,产生没有生活力的卵或抑制卵的孵化。The growth, development and maturation of insects are all regulated or influenced by the steroid ecdysone and the sesquiterpenoid juvenile hormone. Juvenile hormone is a new type of chitin synthesis inhibitor, and its mechanism of action is mainly based on the coordination of the following three aspects: (1) Through the retention of high concentrations in hemolymph, it accelerates the secretion of insect prothymic sex hormones and interferes with the epidermis. The biosynthesis of ecdysone in ecdysone; (2) the chitin synthase of insects is inhibited, causing the synthesis of chitin in the epidermis to be blocked and continuously reduced. Death; (3) The insect lacks the stimuli needed to lay eggs, resulting in the production of non-viable eggs or inhibiting the hatching of eggs.
本发明解决了单一驱虫药物抗虫谱较窄,用药剂量大,易产生抗药性;且现有驱虫制剂短期内驱虫有效,且对虫卵作用较弱,虫卵在一定的孵育期后,成长为幼虫、成虫,继续感染主体,复发率高,不能满足市场需求的技术问题,从整体到局部、宏观到微观的角度出发,更合理的将抗寄生虫药物复配使用,扩大抗虫谱,起到更好的治疗效果,提供一种长效的复方驱虫药物制剂及其制备方法。The invention solves the problem that a single anthelmintic drug has a narrow anti-insect spectrum, a large dosage, and is easy to produce drug resistance; and the existing anthelmintic preparation is effective for deworming in a short term, and has a weak effect on the eggs, and the eggs are in a certain incubation period. After growing into larvae and adults, continue to infect the main body, the recurrence rate is high, and the technical problems that cannot meet the market demand, from the perspective of the whole to the local, macro and micro, it is more reasonable to compound the use of anti-parasitic drugs to expand the anti-parasitic drugs. The insect spectrum has a better therapeutic effect, and provides a long-acting compound anthelmintic medicinal preparation and a preparation method thereof.
发明内容SUMMARY OF THE INVENTION
本发明的目的在于提供一种长效复方驱虫药物液体制剂及其制备方法与应用,利用米尔贝肟在动物体内持续长效驱杀动物体内线虫,苯并咪唑类药物驱杀动物体内绦虫以及保幼激素类药物的体外杀虫作用相结合增效驱虫。该复方制剂联合用药合理,安全可靠,扩大了抗虫谱,从而全面的防治犬体内外寄生虫。The object of the present invention is to provide a long-acting compound anthelmintic drug liquid preparation and a preparation method and application thereof, utilizing mirbeoxime to continuously and long-actly drive and kill nematodes in animals, benzimidazole drugs to drive and kill tapeworms in animals, and The in vitro insecticidal effect of juvenile hormone drugs is combined with synergistic deworming. The combination of the compound preparation is reasonable, safe and reliable, and the anti-insect spectrum is expanded, thereby comprehensively preventing and treating parasites inside and outside the dog.
本发明所述的长效复方驱虫药物液体制剂中有效成分为米尔贝肟、苯并咪唑类药物、保幼激素类药物。The active ingredients in the long-acting compound anthelmintic drug liquid preparation of the present invention are milbe oxime, benzimidazole drugs, and juvenile hormone drugs.
所述苯并咪唑类药物选自阿苯达唑、甲苯咪唑、芬苯达唑、奥芬达唑、康苯达唑、丁苯咪唑、三氯苯达唑、尼托比明中的一种;优选地,所述苯并咪唑类药物为阿苯达唑、康苯达唑、三氯苯达唑中的一种;更优选地,所述苯并咪唑类药物为阿苯达唑。Described benzimidazole class medicine is selected from a kind of in albendazole, mebendazole, fenbendazole, ofendazole, conbendazole, bubendazole, triclabendazole, nitorbimine ; Preferably, the benzimidazole drug is a kind of albendazole, conbendazole, triclbendazole; more preferably, the benzimidazole drug is albendazole.
所述保幼激素类药物为烯虫酯、吡丙醚、哒幼酮、双氧威中的一种;优选地,所述保幼激素类药物为吡丙醚、烯虫酯、双氧威中的一种;更优选地,所述保幼激素类药物为吡丙醚。The juvenile hormone drug is one of methoprene, pyriproxyfen, pyridoxine, and dioxycarb; preferably, the juvenile hormone drug is pyriproxyfen, methoprene, dioxycarb one of; more preferably, the juvenile hormone drug is pyriproxyfen.
本发明所述的长效复方驱虫药物液体制剂中还含有高分子助悬剂、润湿剂、抗氧化剂、分散介质。所述复方制剂中各组分以质量百分数计:米尔贝肟0.5~1%、苯并咪唑类5~10%、保幼激素类3~5%、高分子助悬剂0.1~5%、润湿剂0.1~5%、抗氧化剂0.01~1%,余量为分散介质。优选地,所述的长效复方驱虫药物液体制剂以质量百分数计为:米尔贝肟0.5~1%、苯并咪唑类5~10%、保幼激素类3~5%、高分子助悬剂2~4%、润湿剂1~2%、抗氧化剂0.1~0.3%,余量为分散介质。The long-acting compound anthelmintic drug liquid preparation of the present invention further contains a macromolecule suspending agent, a wetting agent, an antioxidant, and a dispersion medium. Each component in the compound preparation is calculated in mass percentage: 0.5-1% of milbe oxime, 5-10% of benzimidazoles, 3-5% of juvenile hormones, 0.1-5% of macromolecular suspending agent, moisturizing agent Wet agent 0.1-5%, antioxidant 0.01-1%, and the balance is dispersion medium. Preferably, the long-acting compound anthelmintic drug liquid preparation is calculated in mass percentage as: 0.5-1% of milbe oxime, 5-10% of benzimidazoles, 3-5% of juvenile hormones, and macromolecular suspension 2 to 4% of the agent, 1 to 2% of the wetting agent, 0.1 to 0.3% of the antioxidant, and the balance is the dispersion medium.
本发明所述的高分子助悬剂选自羟丙甲基纤维素、黄原胶、明胶、单硬脂酸铝、双硬脂酸铝、氢化蓖麻油、微晶蜡、黄蜡、硬脂酸甘油酯中的一种或几种。The macromolecular suspending agent of the present invention is selected from hydroxypropyl methylcellulose, xanthan gum, gelatin, aluminum monostearate, aluminum distearate, hydrogenated castor oil, microcrystalline wax, yellow wax, stearic acid One or more of glycerides.
所述的润湿剂选自卵磷脂、大豆磷脂、羟化卵磷脂、吐温80、司盘40、司盘60中的一种或几种。The wetting agent is selected from one or more of lecithin, soybean lecithin, hydroxylated lecithin, Tween 80, Span 40 and Span 60.
所述的抗氧化剂选自α-生育酚、对羟基叔丁基茴香醚(BHA)、二叔丁基对甲苯酚(BHT)中的一种或几种。The antioxidant is selected from one or more of α-tocopherol, p-hydroxy-tert-butyl anisole (BHA), and di-tert-butyl-p-cresol (BHT).
所述的分散介质选自乳酸乙酯、丙二醇、大豆油、油酸乙酯、N,N-二甲基甲酰胺、肉豆蔻酸异丙酯、甲醛缩甘油、单油酸甘油酯中的一种或几种。Described dispersion medium is selected from one in ethyl lactate, propylene glycol, soybean oil, ethyl oleate, N,N-dimethylformamide, isopropyl myristate, acetal formaldehyde, glycerol monooleate. species or several.
所述长效复方驱虫药物液体制剂的制备方法为:The preparation method of the long-acting compound anthelmintic medicinal liquid preparation is:
(1)按组方量称取各原料组分,将分散介质在100~180℃高温处理0.5~2h。(1) Weigh each raw material component according to the formula, and treat the dispersion medium at a high temperature of 100~180℃ for 0.5~2h.
(2)采用低温型方法制备:取占注射液总体积50~70%的分散介质,在60~100℃,往分散介质中加入高分子助悬剂,维持0.5~1h,使高分子助悬剂完全胶化;冷却至40℃以下,加入润湿剂、抗氧化剂、米尔贝肟、苯并咪唑类药物、保幼激素类药物,高速剪切处理。(2) Preparation by low temperature method: take the dispersion medium that accounts for 50-70% of the total volume of the injection, add a macromolecule suspending agent to the dispersion medium at 60-100°C, and maintain it for 0.5-1h to make the macromolecular suspending agent suspend. The agent is completely gelled; cooled to below 40°C, added wetting agent, antioxidant, milbe oxime, benzimidazole drugs, juvenile hormone drugs, high-speed shearing treatment.
或,采用高温型方法制备:取占注射液总体积50~70%的分散介质,在100~140℃,往分散介质中加入高分子助悬剂,维持0.5~1h,使高分子助悬剂完全胶化后;加入抗氧化剂,溶解搅拌均匀后迅速冷却至40℃以下,加入润湿剂、米尔贝肟、苯并咪唑类药物、保幼激素类药物,高压均质处理。Or, prepare by high temperature method: take the dispersion medium that accounts for 50-70% of the total volume of the injection, add a polymer suspending agent to the dispersion medium at 100-140°C, and maintain it for 0.5-1h to make the polymer suspending agent After complete gelation; add antioxidants, dissolve and stir evenly, then quickly cool to below 40°C, add wetting agent, milbe oxime, benzimidazole drugs, juvenile hormone drugs, and high-pressure homogenization treatment.
(3)取步骤(2)中所得混合物,冷却至室温,用少量的分散介质定容至刻度;半成品检测合格后分装,即得。(3) Take the mixture obtained in step (2), cool it to room temperature, and use a small amount of dispersing medium to make up the volume to the mark; after the semi-finished product is tested qualified, it is divided into packaging, and that is obtained.
步骤(2)中所述的高压均质的压力为20000~26000psi,时间为0.5~1h;所述的高速剪切的转速1000~4000rpm,时间为0.5~1h。The pressure of the high-pressure homogenization described in the step (2) is 20000-26000 psi, and the time is 0.5-1 h; the rotating speed of the high-speed shearing is 1000-4000 rpm, and the time is 0.5-1 h.
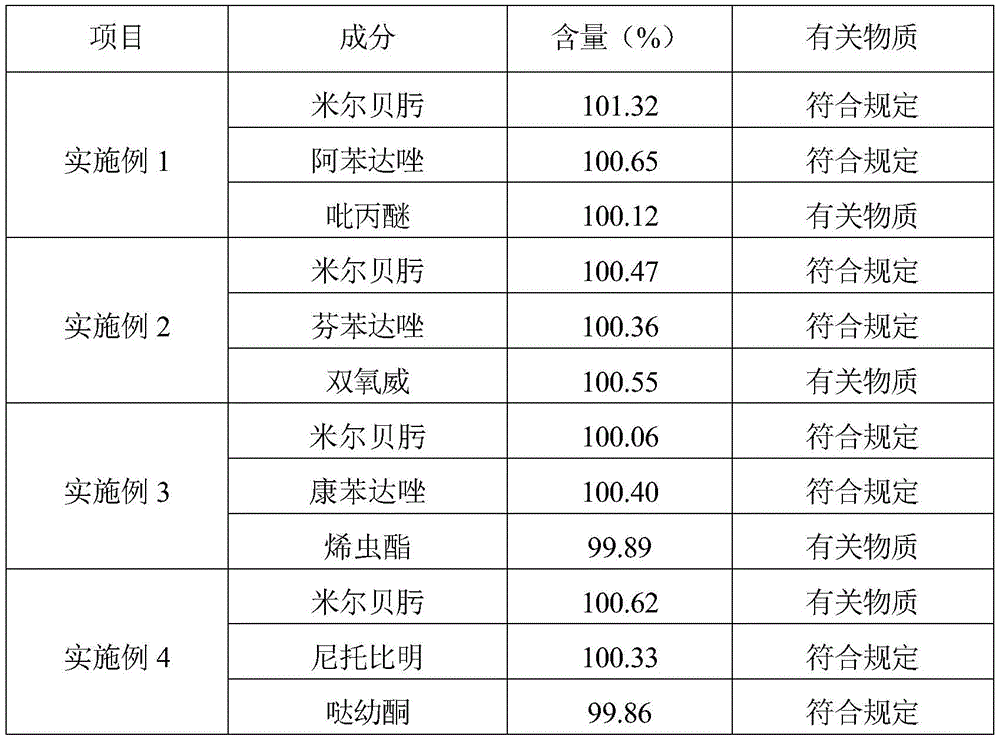
米尔贝肟有关物质和含量测定:《兽药质量标准汇编》(2014版)中米尔贝肟片剂项下方法,对米尔贝肟进行有关物质和含量测定。Determination of related substances and content of mirbeoxime: the method under the item of mirbexime tablet in "Compendium of Quality Standards for Veterinary Drugs" (2014 edition), the related substances and content of mirbexime were determined.
本发明所述的长效复方驱虫药物液体制剂主要用于防治宠物寄生虫病感染,所述宠物包括犬、猫、蜥蜴、豚鼠、羊驼、马、兔、龟等。The long-acting compound anthelmintic drug liquid preparation of the present invention is mainly used for preventing and treating pet parasitic infection, and the pets include dogs, cats, lizards, guinea pigs, alpacas, horses, rabbits, tortoises and the like.
本发明所述的长效复方驱虫药物液体制剂可用于防治体内外寄生虫病,有效的体内寄生虫包括犬心丝虫、胃肠道线虫、蛔虫、钩虫、鞭虫、圆虫、绦虫;有效的体外寄生虫为跳蚤、虱子、螨虫。The long-acting compound anthelmintic medicinal liquid preparation of the present invention can be used for preventing and treating internal and external parasitic diseases, and the effective internal parasites include canine heartworm, gastrointestinal nematode, roundworm, hookworm, whipworm, roundworm and tapeworm; Effective ectoparasites are fleas, lice, and mites.
本发明所述的长效复方驱虫药物液体制剂优选为长效注射液。The long-acting compound anthelmintic medicinal liquid preparation of the present invention is preferably a long-acting injection.
有益效果:Beneficial effects:
1.本发明将抗线虫药物、抗绦虫药物以及昆虫保幼激素药物复配使用,扩大了抗虫谱。米尔贝肟对体外寄生虫作用较弱,增加保幼激素类后,可起到协同增效的作用,使得体外寄生虫虫卵、幼虫的灭杀率提高。1. The present invention combines anti-nematode drugs, anti-taenia drugs and insect juvenile hormone drugs to expand the anti-insect spectrum. Milbe oxime has a weak effect on ectoparasites. After adding juvenile hormones, it can play a synergistic effect, which improves the killing rate of ectoparasite eggs and larvae.
2.本发明产品对体内的线虫、绦虫及体外的跳蚤、虱子、螨虫等都有良好的驱杀作用,达到优异的体内体外同驱的效果,且寄生虫感染不易复发;同时经过试验研究,本发明的长效复方驱虫药物液体制剂稳定且安全,宠物可以放心使用。2. The product of the present invention has a good killing effect on nematodes, tapeworms in vivo and fleas, lice, mites in vitro, etc., achieves excellent in vivo and in vitro effects, and parasitic infection is not easy to recur; Simultaneously through experimental research, The long-acting compound anthelmintic drug liquid preparation of the present invention is stable and safe, and pets can use it with confidence.
3.本发明所制得的复方米尔贝肟长效注射液为注射液剂型,提高了片剂、胶囊剂、口服剂型的生物利用度,可以保证临床使用过程中,达到最优的驱杀体内外寄生虫的效果。3. The compound mirboxime long-acting injection prepared by the present invention is an injection dosage form, which improves the bioavailability of tablets, capsules, and oral dosage forms, and can ensure that in the course of clinical use, the optimal expelling and killing body can be achieved. Effects of internal and external parasites.
4.本发明所制得的复方米尔贝肟长效注射液因为添加了高分子助悬剂,形成凝胶态,对主药形成了一定的包裹,添加了润湿剂,用磷脂双分子形成包裹层,分散介质选用大分子油性物质,综合制剂制备后具有长效作用,在体内皮下,形成的油性凝胶态,机体不会对其产生异体反应,在较长时间内,被机体逐渐分解释药,形成长效机制。4. The compound mirbeoxime long-acting injection prepared by the present invention forms a gel state due to the addition of a macromolecular suspending agent, forms a certain package for the main drug, adds a wetting agent, and forms a phospholipid bimolecule. The coating layer and the dispersion medium are macromolecular oily substances. After the comprehensive preparation is prepared, it has a long-term effect. In the body subcutaneously, the oily gel state is formed, and the body will not have an allogeneic reaction to it. Explain the medicine and form a long-acting mechanism.
5.本发明所制得的复方米尔贝肟长效注射液成本较低,操作简单,生物相容性好,刺激性较小,安全性高,促进和改善疾病的转归和预后。5. The compound mirboxime long-acting injection prepared by the present invention has the advantages of low cost, simple operation, good biocompatibility, less irritation and high safety, and promotes and improves the outcome and prognosis of the disease.
具体实施方式Detailed ways
以下将通过实施例形式的具体实施方式,对上述内容做进一步的详细说明。但不应该将此理解为上述主题的范围仅限于以下实施例。凡基于本发明内容所实现的技术均属于范围内。The above content will be further described in detail below through specific implementations in the form of examples. However, this should not be construed as limiting the scope of the above-mentioned subject matter to the following examples. All technologies implemented based on the content of the present invention belong to the scope.
实施例1一种长效复方驱虫药物液体制剂及其制备方法Embodiment 1 A kind of long-acting compound anthelmintic medicinal liquid preparation and preparation method thereof
长效复方驱虫药物液体制剂的组方及用量:The composition and dosage of the long-acting compound deworming drug liquid preparation:
长效复方驱虫药物液体制剂的制备方法:The preparation method of long-acting compound anthelmintic medicine liquid preparation:
采用高温型方法制备:取700mL的油酸乙酯+N,N-二甲基甲酰胺(1:1),在130℃,往油酸乙酯+N,N-二甲基甲酰胺(1:1)中加入单硬脂酸铝、双硬脂酸铝,维持1h,使单硬脂酸铝、双硬脂酸铝完全胶化后;加入α-生育酚,溶解搅拌均匀后迅速冷却至40℃以下,加入吐温80、卵磷脂、米尔贝肟(粒度D90≤5μm)、阿苯达唑(粒度D90≤5μm)、吡丙醚(粒度D90≤5μm);高压均质(压力为20000psi)处理1h,取冷却至室温的混合物,用少量的油酸乙酯+N,N-二甲基甲酰胺(1:1)定容至刻度;半成品检测合格后分装,即得。Prepared by high temperature method: take 700 mL of ethyl oleate+N,N-dimethylformamide (1:1), and at 130 °C, transfer ethyl oleate+N,N-dimethylformamide (1 : 1) add aluminum monostearate and aluminum distearate, maintain for 1h, and make aluminum monostearate and aluminum distearate gel completely; add α-tocopherol, dissolve and stir evenly and then cool down to Below 40℃, add Tween 80, lecithin, milbe oxime (particle size D90≤5μm), albendazole (particle size D90≤5μm), pyriproxyfen (particle size D90≤5μm); high pressure homogenization (pressure is 20000psi) ) for 1 hour, take the mixture cooled to room temperature, and dilute to the mark with a small amount of ethyl oleate + N,N-dimethylformamide (1:1).
实施例2一种长效复方驱虫药物液体制剂及其制备方法Embodiment 2 A kind of long-acting compound anthelmintic medicinal liquid preparation and preparation method thereof
长效复方驱虫药物液体制剂的组方:The composition of the long-acting compound deworming drug liquid preparation:
长效复方驱虫药物液体制剂的制备方法:The preparation method of long-acting compound anthelmintic medicine liquid preparation:
采用高温型方法制备:取700mL的大豆油+N,N-二甲基甲酰胺(1:1),在130℃,往大豆油+N,N-二甲基甲酰胺(1:1)中加入单硬脂酸铝,维持1h,使单硬脂酸铝完全胶化后;加入对羟基叔丁基茴香醚,溶解搅拌均匀后迅速冷却至40℃以下,加入吐温80、卵磷脂、米尔贝肟(粒度D90≤5μm)、芬苯达唑(粒度D90≤5μm)、双氧威(粒度D90≤5μm);高压均质(压力为20000psi)处理1h,取冷却至室温的混合物,用少量的大豆油+N,N-二甲基甲酰胺(1:1)定容至刻度;半成品检测合格后分装,即得。Prepared by high temperature method: take 700mL of soybean oil+N,N-dimethylformamide (1:1), put soybean oil+N,N-dimethylformamide (1:1) into soybean oil+N,N-dimethylformamide (1:1) at 130°C Add aluminum monostearate and keep it for 1 hour to make the aluminum monostearate gel completely; add p-hydroxytert-butyl anisole, dissolve and stir evenly, quickly cool to below 40°C, add Tween 80, lecithin, mirin Beoxime (particle size D90≤5μm), fenbendazole (particle size D90≤5μm), dioxycarb (particle size D90≤5μm); high pressure homogenization (pressure 20000psi) for 1h, take the mixture cooled to room temperature, add a small amount of Soybean oil + N,N-dimethylformamide (1:1) is adjusted to the mark; the semi-finished product is tested and repackaged after passing the test.
实施例3一种长效复方驱虫药物液体制剂及其制备方法Embodiment 3 A kind of long-acting compound anthelmintic medicinal liquid preparation and preparation method thereof
长效复方驱虫药物液体制剂的组方:The composition of the long-acting compound deworming drug liquid preparation:
长效复方驱虫药物液体制剂的制备方法:The preparation method of long-acting compound anthelmintic medicine liquid preparation:
采用高温型方法制备:取700mL的油酸乙酯,在130℃,往油酸乙酯中加入双硬脂酸铝、硬脂酸甘油酯,维持1h,使双硬脂酸铝、硬脂酸甘油酯完全胶化后;加入α-生育酚+二叔丁基对甲苯酚(BHT)(1:1),溶解搅拌均匀后迅速冷却至40℃以下,加入司盘40、羟化卵磷脂、米尔贝肟(粒度D90≤5μm)、康苯达唑(粒度D90≤5μm)、烯虫酯(粒度D90≤5μm);高压均质(压力为20000psi)处理0.5h,取冷却至室温的混合物,用少量的油酸乙酯定容至刻度;半成品检测合格后分装,即得。Preparation by high temperature method: take 700 mL of ethyl oleate, add aluminum distearate and glyceryl stearate to ethyl oleate at 130°C, and maintain for 1 h to make aluminum distearate, stearic acid After the glycerides are completely gelled; add α-tocopherol + di-tert-butyl-p-cresol (BHT) (1:1), dissolve and stir evenly, cool down quickly to below 40°C, add Span 40, hydroxylated lecithin, Milbeoxime (particle size D90≤5μm), conbendazole (particle size D90≤5μm), methoprene (particle size D90≤5μm); high pressure homogenization (pressure 20000psi) for 0.5h, take the mixture cooled to room temperature, Dilute the volume to the mark with a small amount of ethyl oleate; after the semi-finished product has passed the inspection, it is divided into packages.
实施例4一种长效复方驱虫药物液体制剂及其制备方法Embodiment 4 A kind of long-acting compound anthelmintic medicine liquid preparation and preparation method thereof
长效复方驱虫药物液体制剂的组方:The composition of the long-acting compound deworming drug liquid preparation:
长效复方驱虫药物液体制剂的制备方法:The preparation method of long-acting compound anthelmintic medicine liquid preparation:
采用低温型方法制备:取700mL的丙二醇,在80℃,往丙二醇中加入微晶蜡、羟丙甲基纤维素,维持1h,使微晶蜡、羟丙甲基纤维素完全胶化后,冷却至40℃以下,加入吐温80、卵磷脂、二叔丁基对甲苯酚+对羟基叔丁基茴香醚(1:1)、米尔贝肟(粒度D90≤5μm)、尼托比明(粒度D90≤5μm)、哒幼酮(粒度D90≤5μm);高速剪切(转速为3000rpm)处理1h,取冷却至室温的混合物,用少量的丙二醇定容至刻度;半成品检测合格后分装,即得。Preparation by low-temperature method: take 700 mL of propylene glycol, add microcrystalline wax and hydroxypropyl methylcellulose to propylene glycol at 80 ° C, maintain for 1 hour, make the microcrystalline wax and hydroxypropyl methyl cellulose completely gelatinized, then cool To below 40 ℃, add Tween 80, lecithin, di-tert-butyl-p-cresol + p-hydroxy-tert-butyl anisole (1:1), milbe oxime (particle size D90≤5μm), nitorbiamine (particle size D90≤5μm), pyridone (particle size D90≤5μm); high-speed shearing (rotation speed 3000rpm) for 1h, take the mixture cooled to room temperature, and use a small amount of propylene glycol to make up the volume to the mark; semi-finished products are divided into packaging after passing the inspection, namely have to.
实施例5一种长效复方驱虫药物液体制剂及其制备方法Embodiment 5 A kind of long-acting compound anthelmintic medicinal liquid preparation and preparation method thereof
长效复方驱虫药物液体制剂的组方:The composition of the long-acting compound deworming drug liquid preparation:
长效复方驱虫药物液体制剂的制备方法:The preparation method of long-acting compound anthelmintic medicine liquid preparation:
采用低温型方法制备:取700mL的乳酸乙酯,在80℃,往乳酸乙酯中加入微晶蜡、羟丙甲基纤维素,维持1h,使微晶蜡、羟丙甲基纤维素完全胶化后,冷却至40℃以下,加入吐温80、卵磷脂、α-生育酚、米尔贝肟(粒度D90≤5μm)、尼托比明(粒度D90≤5μm)、哒幼酮(粒度D90≤5μm);高速剪切(转速为3000rpm)处理1h,取冷却至室温的混合物,用少量的乳酸乙酯定容至刻度;半成品检测合格后分装,即得。Preparation by low temperature method: take 700 mL of ethyl lactate, add microcrystalline wax and hydroxypropyl methyl cellulose to ethyl lactate at 80°C, and maintain for 1 h to make the microcrystalline wax and hydroxypropyl methyl cellulose completely glue. After melting, cool to below 40℃, add Tween 80, lecithin, α-tocopherol, milbe oxime (particle size D90≤5μm), nitorbamine (particle size D90≤5μm), pyridone (particle size D90≤5μm) 5 μm); high-speed shearing (rotation speed is 3000 rpm) for 1 h, take the mixture cooled to room temperature, and use a small amount of ethyl lactate to make up to the mark; after the semi-finished product is qualified, it is divided into packaging, that is, it is obtained.
实施例6一种长效复方驱虫药物液体制剂及其制备方法Embodiment 6 A kind of long-acting compound anthelmintic medicinal liquid preparation and preparation method thereof
长效复方驱虫药物液体制剂的组方:The composition of the long-acting compound deworming drug liquid preparation:
长效复方驱虫药物液体制剂的制备方法:The preparation method of long-acting compound anthelmintic medicine liquid preparation:
采用低温型方法制备:取700mL的单油酸甘油酯+肉豆蔻酸异丙酯(1:1),在80℃,往单油酸甘油酯+肉豆蔻酸异丙酯(1:1)中加入明胶、微晶蜡,维持1h,使明胶、微晶蜡完全胶化后,冷却至40℃以下,加入吐温80、大豆磷脂、二叔丁基对甲苯酚、米尔贝肟(粒度D90≤5μm)、奥芬达唑(粒度D90≤5μm)、烯虫酯(粒度D90≤5μm);高速剪切(转速为4000rpm)处理0.5h,取冷却至室温的混合物,用少量的单油酸甘油酯+肉豆蔻酸异丙酯(1:1)定容至刻度;半成品检测合格后分装,即得。Prepared by a low temperature method: take 700 mL of glycerol monooleate + isopropyl myristate (1:1), put glycerol monooleate + isopropyl myristate (1:1) at 80 °C Add gelatin and microcrystalline wax, keep for 1h, after gelatin and microcrystalline wax are completely gelatinized, cool to below 40 ℃, add Tween 80, soybean lecithin, di-tert-butyl-p-cresol, milbe oxime (particle size D90≤ 5μm), orfendazole (particle size D90≤5μm), methoprene (particle size D90≤5μm); high-speed shearing (rotation speed is 4000rpm) for 0.5h, take the mixture cooled to room temperature, use a small amount of glycerol monooleate Ester + isopropyl myristate (1:1) and make up to the mark; the semi-finished product is tested and packaged after passing the test.
实施例7一种长效复方驱虫药物液体制剂及其制备方法Embodiment 7 A kind of long-acting compound anthelmintic medicinal liquid preparation and preparation method thereof
长效复方驱虫药物液体制剂的组方:The composition of the long-acting compound deworming drug liquid preparation:
长效复方驱虫药物液体制剂的制备方法:The preparation method of long-acting compound anthelmintic medicine liquid preparation:
采用低温型方法制备:取700mL的甲醛缩甘油,在80℃,往甲醛缩甘油中加入明胶、微晶蜡,维持1h,使明胶、微晶蜡完全胶化后,冷却至40℃以下,加入氢化蓖麻油、司盘40、α-生育酚+对羟基叔丁基茴香醚(1:1)、米尔贝肟(粒度D90≤5μm)、三氯苯达唑(粒度D90≤5μm)、双氧威(粒度D90≤5μm);高速剪切(转速为4000rpm)处理0.5h,取冷却至室温的混合物,用少量的甲醛缩甘油定容至刻度;半成品检测合格后分装,即得。Preparation by low-temperature method: take 700 mL of formaldehyde acetal, add gelatin and microcrystalline wax to formaldehyde acetal at 80 °C, maintain for 1 h, make gelatin and microcrystalline wax completely gelatinized, cool to below 40 °C, add Hydrogenated Castor Oil, Span 40, α-Tocopherol + p-Hydroxy-tert-butylanisole (1:1), Milbeoxime (Particle Size D90≤5μm), Triclobendazole (Particle Size D90≤5μm), Dioxygen Granules (particle size D90≤5μm); high-speed shearing (rotation speed is 4000rpm) for 0.5h, take the mixture cooled to room temperature, and use a small amount of formaldehyde acetal to make up to the mark; semi-finished products are tested and packaged after passing the test.
实施例8一种长效复方驱虫药物液体制剂及其制备方法Embodiment 8 A kind of long-acting compound anthelmintic medicinal liquid preparation and preparation method thereof
长效复方驱虫药物液体制剂的组方:The composition of the long-acting compound deworming drug liquid preparation:
长效复方驱虫药物液体制剂的制备方法:The preparation method of long-acting compound anthelmintic medicine liquid preparation:
采用低温型方法制备:取700mL的丙二醇+肉豆蔻酸异丙酯(1:1),在80℃,往丙二醇+肉豆蔻酸异丙酯(1:1)中加入黄蜡、羧丙甲基纤维素,维持1h,使黄蜡、羧丙甲基纤维素完全胶化后,冷却至40℃以下,加入司盘40、羟化卵磷脂、二叔丁基对甲苯酚、米尔贝肟(粒度D90≤5μm)、丁苯咪唑(粒度D90≤5μm)、吡丙醚(粒度D90≤5μm);高速剪切(转速为4000rpm)处理0.5h,取冷却至室温的混合物,用少量的丙二醇+肉豆蔻酸异丙酯(1:1)定容至刻度;半成品检测合格后分装,即得。Preparation by low-temperature method: take 700 mL of propylene glycol + isopropyl myristate (1:1), add yellow wax and carboxypropyl methyl fiber to propylene glycol + isopropyl myristate (1:1) at 80 °C After the yellow wax and carboxypropyl methylcellulose are completely gelatinized, they are cooled to below 40°C, and Span 40, hydroxylated lecithin, di-tert-butyl-p-cresol, milbe oxime (particle size D90≤ 5μm), bubendazole (particle size D90≤5μm), pyriproxyfen (particle size D90≤5μm); high-speed shearing (rotation speed is 4000rpm) for 0.5h, take the mixture cooled to room temperature, use a small amount of propylene glycol + myristic acid Dilute the volume of isopropyl ester (1:1) to the mark; after the semi-finished product has passed the inspection, it is divided into packages.
实施例9米尔贝肟复方长效注射液的体外性能评价Example 9 In vitro performance evaluation of mirboxime compound long-acting injection
米尔贝肟复方长效注射液进行体外性能评价,包括性状、粒度、3h沉降体积比,再分散性、通针性、有关物质和含量等测定。测定结果见表1-2。Milbeoxime compound long-acting injection was subjected to in vitro performance evaluation, including the determination of properties, particle size, 3h sedimentation volume ratio, redispersibility, acupuncture, related substances and content. The measurement results are shown in Table 1-2.
表1米尔贝肟复方长效注射液的体外性能评价Table 1 In vitro performance evaluation of mirboxime compound long-acting injection
注:*代表再分散性,*的数量越多,代表再分散性越好,+代表通针性,+越多表明通针性越好。Note: * represents the redispersibility, the more the number of *, the better the redispersibility, the + represents the needle penetration, the more + the better the needle penetration.
表2米尔贝肟复方长效注射液含量及有关物质结果Table 2 Milbeoxime compound long-acting injection content and related substance results
由表1和表2可知,实施例1-4的米尔贝肟复方长效注射液的性状、粒度、沉降体积比、再分散性及通针等性状均良好,有关物质也符合规定。As can be seen from Table 1 and Table 2, the properties such as the Milbeoxime compound long-acting injection of Examples 1-4 are all good in properties, particle size, sedimentation volume ratio, redispersibility and acupuncture, and related substances are also in compliance with the regulations.
实施例10米尔贝肟复方长效注射液治疗寄生虫疗效评价Example 10 Evaluation of the curative effect of Milbeoxime compound long-acting injection in treating parasites
本实验对实施例1中所得的长效复方驱虫药物液体制剂进行动物临床实验以检验其疗效。实验犬经实验室粪便漂浮法和直接涂片法镜检发现感染有大量的肠道寄生虫(有毛圆线虫、钩虫、鞭虫、蛔虫、绦虫等)。In this experiment, animal clinical experiments were carried out on the long-acting compound anthelmintic drug liquid preparation obtained in Example 1 to test its curative effect. The experimental dogs were found to be infected with a large number of intestinal parasites (such as Trichostrongylus, hookworm, whipworm, roundworm, tapeworm, etc.) by laboratory fecal floatation method and direct smear method.
将经诊断严重感染肠道寄生虫病的犬120只,随机分为6组,实验组1为空白对照组,实验组2为市售米尔贝肟片剂(2.5mg/片,海正),实验组3-6为本发明实施例1、实施例3、实施例7、对比例1(不含米尔贝肟,其它组分用量不变的实施例1)产品,按照单次以米尔贝肟计每公斤体重2.5mg给药,对比例1按实施例1剂量给予,给药后第0天、5天、30天、90天及150天分别统计各组药物对犬体内线虫、体内绦虫、体外寄生虫的平均检出率。120 dogs diagnosed with severe intestinal parasitic diseases were randomly divided into 6 groups, the experimental group 1 was the blank control group, and the experimental group 2 was the commercially available Milbeoxime tablet (2.5mg/tablet, Hisun), Experimental groups 3-6 are the products of Example 1, Example 3, Example 7, and Comparative Example 1 (Example 1 with no milbe oxime, and the dosage of other components is unchanged) according to a single dose of milbe oxime. 2.5 mg per kilogram of body weight was administered, and Comparative Example 1 was administered according to the dosage of Example 1, and on the 0th, 5th, 30th, 90th and 150th days after administration, the effects of each group of drugs on nematodes, tapeworms, Average detection rate of ectoparasites.
表3犬体内线虫检出率统计Table 3 Statistics on the detection rate of nematodes in dogs
表4犬体内绦虫检出率统计Table 4 Statistics on the detection rate of tapeworms in dogs
表5犬体外寄生虫检出率统计Table 5 Statistics on the detection rate of ectoparasites in dogs
从表3-5可以看出,本发明实施例1对犬体内线虫、绦虫,犬体外寄生虫均有明显的杀虫效果,检出率均低于空白对照组和市售阳性组,并且作用时间长,在150天时仍有很好的驱虫效果。证实将米尔贝肟、苯并咪唑类药物、保幼激素类杀虫剂结合使用,抗寄生虫效果明显优于单方的市售米尔贝肟制剂。通过对比实验证实,在没有米尔贝肟存在的条件下,对比例1产品的抗体外寄生虫效果相对于实施例1明显较差,说明米尔贝肟对保幼激素类药物具有抗体外寄生虫的增效作用,使得体外寄生虫虫卵、幼虫的灭杀率提高。As can be seen from Table 3-5, the embodiment of the present invention 1 has obvious insecticidal effect to nematodes, tapeworms, and ectoparasites in dogs, and the detection rates are all lower than the blank control group and the commercially available positive group, and the effect For a long time, there is still a good deworming effect at 150 days. It is confirmed that the combined use of mirbexime, benzimidazole drugs and juvenile hormone insecticides has significantly better anti-parasitic effect than the single commercially available mirbexime preparation. It was confirmed by comparative experiments that in the absence of mirbeoxime, the antibody ectoparasite effect of the product of Comparative Example 1 was significantly poorer than that of Example 1, indicating that mirbeoxime has the ability of antibody ectoparasites to juvenile hormone drugs. The synergistic effect increases the killing rate of ectoparasite eggs and larvae.
试验结果表明,本发明实施例制备的长效复方驱虫药物液体制剂对犬体内外寄生虫在150天的时间内,有良好的驱杀寄生虫的作用。The test results show that the long-acting compound anthelmintic medicinal liquid preparation prepared in the embodiment of the present invention has a good effect of repelling and killing parasites in dogs within 150 days.
实施例11米尔贝肟复方长效注射液安全性试验Embodiment 11 Safety test of mirboxime compound long-acting injection
选取20g左右的昆明种小白鼠40只,随机将其分为4组,每组10只,公母各半。第1组为对照组,肌肉注射0.5m1生理盐水;第2-4组为试验组,选用不同剂量(高剂量、中剂量和低剂量)的本发明实施例1的长效复方驱虫药物液体制剂制备,因为市售阳性对照药物制剂米尔贝肟用于犬的使用剂量为2.5mg-1.0mg/10kg,所以本发明安全性试验高剂量组、中剂量组和低剂量组,分别肌肉注射5.0mL、3.0mL和1.0mL,仅用1次,之后连续观察3-5个月,期间记录小鼠的进食、饮水、行为变化及死亡情况,试验结束后对各组小鼠随机挑选3只进行解剖,观察脏器的病变情况,并记录。40 Kunming mice of about 20 g were selected and randomly divided into 4 groups with 10 mice in each group, half male and female. The 1st group is the control group, intramuscular injection of 0.5m1 normal saline; the 2nd-4th group is the test group, the long-acting compound anthelmintic drug liquid of Example 1 of the present invention of different doses (high dose, medium dose and low dose) is selected for use Preparation preparation, because the dosage of commercially available positive control pharmaceutical preparation milbeoxime for dogs is 2.5mg-1.0mg/10kg, so the safety test high-dose group, middle-dose group and low-dose group of the present invention are injected intramuscularly with 5.0 mL, 3.0mL, and 1.0mL were used only once, followed by continuous observation for 3-5 months, during which the eating, drinking, behavioral changes and death of the mice were recorded. After the experiment, 3 mice in each group were randomly selected for Dissect, observe the pathological changes of organs, and record.
结果:对照组和试验2-4组小鼠在试验期间均未出现死亡,进食和饮水正常,行为未发生明显异常,通过剖检,试验2-4组小鼠心肝、脾、肺、肾、胃、肠道、均未有异常病变,与对照组无明显差异。结果表明本发明制备的长效复方驱虫药物液体制剂使用安全。Results: The mice in the control group and experimental groups 2-4 did not die during the test period. They ate and drank normally, and had no obvious abnormal behavior. There were no abnormal lesions in the stomach and intestines, and there was no significant difference with the control group. The results show that the long-acting compound anthelmintic drug liquid preparation prepared by the present invention is safe to use.
Claims (10)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010748308.XA CN112006981B (en) | 2020-07-30 | 2020-07-30 | Long-acting compound anthelmintic liquid preparation and preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010748308.XA CN112006981B (en) | 2020-07-30 | 2020-07-30 | Long-acting compound anthelmintic liquid preparation and preparation method and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN112006981A true CN112006981A (en) | 2020-12-01 |
| CN112006981B CN112006981B (en) | 2023-01-10 |
Family
ID=73500037
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010748308.XA Active CN112006981B (en) | 2020-07-30 | 2020-07-30 | Long-acting compound anthelmintic liquid preparation and preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN112006981B (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112641723A (en) * | 2020-12-30 | 2021-04-13 | 南京朗博特动物药业有限公司 | Pharmaceutical composition for treating otoacarid and preparation method thereof |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060222684A1 (en) * | 2003-07-30 | 2006-10-05 | Ute Isele | Palatable ductile chewable veterinary composition |
| CN1905794A (en) * | 2003-12-17 | 2007-01-31 | 梅瑞尔有限公司 | Topical formulations comprising a 1-N-arylpyrazole derivative and a formamidine |
| US20070042013A1 (en) * | 2005-08-19 | 2007-02-22 | Soll Mark D | Long acting injectable formulations |
| US20070128239A1 (en) * | 2005-12-06 | 2007-06-07 | Wyeth | Benzimidazole non-aqueous compositions |
| CN101773467A (en) * | 2010-01-20 | 2010-07-14 | 北京大北农动物保健科技有限责任公司 | Milbemycin or milbemycin oxime sustained-release injection for animals and preparation method |
| CN102481279A (en) * | 2009-03-27 | 2012-05-30 | 奴布卢克实验室有限公司 | A topical parasiticide composition |
| CN102869253A (en) * | 2010-04-02 | 2013-01-09 | 梅里亚有限公司 | Parasiticidal compositions comprising multiple active agents, methods and uses thereof |
-
2020
- 2020-07-30 CN CN202010748308.XA patent/CN112006981B/en active Active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060222684A1 (en) * | 2003-07-30 | 2006-10-05 | Ute Isele | Palatable ductile chewable veterinary composition |
| CN1905794A (en) * | 2003-12-17 | 2007-01-31 | 梅瑞尔有限公司 | Topical formulations comprising a 1-N-arylpyrazole derivative and a formamidine |
| US20070042013A1 (en) * | 2005-08-19 | 2007-02-22 | Soll Mark D | Long acting injectable formulations |
| US20070128239A1 (en) * | 2005-12-06 | 2007-06-07 | Wyeth | Benzimidazole non-aqueous compositions |
| CN102481279A (en) * | 2009-03-27 | 2012-05-30 | 奴布卢克实验室有限公司 | A topical parasiticide composition |
| CN101773467A (en) * | 2010-01-20 | 2010-07-14 | 北京大北农动物保健科技有限责任公司 | Milbemycin or milbemycin oxime sustained-release injection for animals and preparation method |
| CN102869253A (en) * | 2010-04-02 | 2013-01-09 | 梅里亚有限公司 | Parasiticidal compositions comprising multiple active agents, methods and uses thereof |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112641723A (en) * | 2020-12-30 | 2021-04-13 | 南京朗博特动物药业有限公司 | Pharmaceutical composition for treating otoacarid and preparation method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| CN112006981B (en) | 2023-01-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5728719A (en) | Systemic control of parasites | |
| JP4498667B2 (en) | Internal parasite control gel composition | |
| CN117043161A (en) | Methods and pharmaceutical compositions for treating diseases | |
| CN112006981B (en) | Long-acting compound anthelmintic liquid preparation and preparation method and application thereof | |
| CN115671288A (en) | A kind of anti-parasitic compound soft chewing pharmaceutical preparation and its preparation method and application | |
| JP3170077B2 (en) | Systemic control of parasites | |
| CN114939104B (en) | External transdermal preparation for relieving cat stress, preparation method and application | |
| BR112013002177B1 (en) | COMPOSITION OF A VETERINARY DRUG FOR USE IN THE CONTROL OF HEART INFESTATIONS IN A NON-HUMAN GUINEA PIG, AND KIT FOR TREATMENT AND / OR PREVENTION OF THE HEART IN A NON-HUMAN GUINEA PIG | |
| CN119112798B (en) | A kind of fluorouracil nanostructure lipid carrier and preparation method thereof | |
| JPH02268115A (en) | Continuously releasable large pill | |
| Thomazini et al. | Impact of concerning excipients on animal safety: insights for veterinary pharmacotherapy and regulatory considerations | |
| BRPI0619620B1 (en) | oral veterinary composition and use thereof | |
| Adil et al. | Toxicity and adverse effects of veterinary pharmaceuticals in animals | |
| US10632137B2 (en) | Composition containing ivermectin for exterminating Clavinema mariae infection on Sebastes schlegeli | |
| CN110772509A (en) | Imidacloprid and moxidectin diatomic alcohol plastid and preparation method and application thereof | |
| CN112972437B (en) | Long-acting external preparation containing iodonitrophenol and preparation method and application thereof | |
| CN110721152B (en) | Sustained-release composition for treating animal skin parasite and fungus infection | |
| BR112019018700A2 (en) | pharmaceutical compositions and their uses | |
| CN108403631A (en) | Ten thousand rhzomorph oil mixed suspension injections of tartaric acid Thailand and preparation method thereof | |
| RU2715432C1 (en) | Antiparasitic agent for treatment and prevention of animals by voluntary feeding | |
| CN109394702B (en) | A kind of composition containing albendazole and its preparation method and use | |
| RU2847348C1 (en) | Antiparasitic agent for the treatment and prevention of ectoparasites in dogs. | |
| CN110022862B (en) | Pharmaceutical preparation and process for producing the same | |
| CN111658606A (en) | Ponazuril solution preparation and application | |
| WO2010045700A1 (en) | Method for preparing a veterinary suspension formulation for administering water-insoluble medicaments |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |