CN111925442B - New coronavirus RBD specific monoclonal antibody and application - Google Patents
New coronavirus RBD specific monoclonal antibody and application Download PDFInfo
- Publication number
- CN111925442B CN111925442B CN202010839860.XA CN202010839860A CN111925442B CN 111925442 B CN111925442 B CN 111925442B CN 202010839860 A CN202010839860 A CN 202010839860A CN 111925442 B CN111925442 B CN 111925442B
- Authority
- CN
- China
- Prior art keywords
- seq
- rbd
- amino acid
- acid sequence
- monoclonal antibody
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/08—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from viruses
- C07K16/10—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from viruses from RNA viruses
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/569—Immunoassay; Biospecific binding assay; Materials therefor for microorganisms, e.g. protozoa, bacteria, viruses
- G01N33/56983—Viruses
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/51—Complete heavy chain or Fd fragment, i.e. VH + CH1
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/515—Complete light chain, i.e. VL + CL
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/005—Assays involving biological materials from specific organisms or of a specific nature from viruses
- G01N2333/08—RNA viruses
- G01N2333/165—Coronaviridae, e.g. avian infectious bronchitis virus
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2469/00—Immunoassays for the detection of microorganisms
- G01N2469/10—Detection of antigens from microorganism in sample from host
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Virology (AREA)
- Immunology (AREA)
- Molecular Biology (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Biochemistry (AREA)
- Hematology (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Urology & Nephrology (AREA)
- Organic Chemistry (AREA)
- Food Science & Technology (AREA)
- Microbiology (AREA)
- Physics & Mathematics (AREA)
- Analytical Chemistry (AREA)
- Cell Biology (AREA)
- Biotechnology (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Tropical Medicine & Parasitology (AREA)
- Peptides Or Proteins (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
Abstract
本发明属于单克隆抗体技术领域,具体公开了新冠病毒RBD特异性单克隆抗体,重链氨基酸序列如SEQ ID NO:1所示;轻链氨基酸序列如SEQ ID NO:2所示。本发明还提供了上述新冠病毒RBD特异性单克隆抗体的应用。本发明对于新型冠状病毒SARS‑CoV‑2引起的疾病的预防、临床治疗和诊断试剂的研发均具有重要的科学意义和应用前景。
The invention belongs to the technical field of monoclonal antibodies, and specifically discloses a novel coronavirus RBD-specific monoclonal antibody. The heavy chain amino acid sequence is shown in SEQ ID NO: 1; the light chain amino acid sequence is shown in SEQ ID NO: 2. The present invention also provides the application of the above-mentioned novel coronavirus RBD-specific monoclonal antibody. The invention has important scientific significance and application prospects for the prevention, clinical treatment and research and development of diagnostic reagents for diseases caused by the novel coronavirus SARS-CoV-2.
Description
技术领域technical field
本发明属于单克隆抗体技术领域,尤其涉及新冠病毒RBD特异性单克隆抗体和应用。The invention belongs to the technical field of monoclonal antibodies, and in particular relates to novel coronavirus RBD-specific monoclonal antibodies and applications.
背景技术Background technique
抗体是由四条多肽链组成的免疫球蛋白分子,四条多肽链包括两条重链(H链)和两条轻链(L链)。H链由重链可变区(VH)和重链恒定区组成,重链恒定区由三个区CH1、CH2和CH3组成。L链由L链可变区(VL)和轻链恒定区组成,轻链恒定区由CL区组成。VH和VL可进一步分成称为互补决定区(CDRs)的高变区和称为构架区(FR)交替分布的保守区。Antibodies are immunoglobulin molecules composed of four polypeptide chains, including two heavy chains (H chains) and two light chains (L chains). The H chain consists of a heavy chain variable region (VH) and a heavy chain constant region, and the heavy chain constant region consists of three regions CH1, CH2 and CH3. The L chain is composed of the L chain variable region (VL) and the light chain constant region, and the light chain constant region is composed of the CL region. VH and VL can be further divided into hypervariable regions called complementarity determining regions (CDRs) and alternately conserved regions called framework regions (FRs).
目前研究发现:新冠病毒(SARS-CoV-2)具有四种主要的结构蛋白,分别为刺突蛋白(S蛋白)、核衣壳蛋白(N蛋白)、膜蛋白(M蛋白)和包膜蛋白(E蛋白),其中S蛋白有两个亚基:S1和S2,受体结合位点(RBD)位于S1亚基上,其主要功能是识别宿主细胞表面受体,介导与宿主细胞的融合。Current research has found that the new coronavirus (SARS-CoV-2) has four main structural proteins, namely the spike protein (S protein), the nucleocapsid protein (N protein), the membrane protein (M protein) and the envelope protein. (E protein), in which the S protein has two subunits: S1 and S2, the receptor binding site (RBD) is located on the S1 subunit, and its main function is to recognize the host cell surface receptor and mediate fusion with the host cell .
目前针对新发病原体COVID-19尚无特效药物针对性治疗,疫苗研发尚需时日。近期治愈出院的患者血浆中含有高浓度特异性的抗原中和抗体,输入患者体内后,可以中和新冠病原体,介导有效的免疫反应,因此利用恢复期血浆有望为救治感染新冠病毒的患者提供有效的治疗手段,降低死亡率,保障患者生命安全。At present, there is no specific drug-targeted treatment for the emerging pathogen COVID-19, and vaccine development will take time. The plasma of recently cured and discharged patients contains a high concentration of specific antigen-neutralizing antibodies. After infusion into the patient, it can neutralize the new crown pathogen and mediate an effective immune response. Therefore, the use of convalescent plasma is expected to provide treatment for patients infected with the new coronavirus. Effective treatment methods can reduce mortality and ensure the safety of patients' lives.
申请公开号为CN111303280A的中国发明专利申请公开了一种高中和活性抗SARS-CoV-2全人源单克隆抗体,上述专利提供的是识别区域为S1非RBD区的全人源单克隆抗体,但是由于新冠病毒入侵宿主细胞是通过RBD与宿主细胞的ACE2结合,所以上述专利获得的全人源单克隆抗体对病毒的阻断效果有限,并且上述专利是通过标记浆细胞而获得抗体cDNA,但是与浆细胞相比,是记忆B细胞活化后迅速做出反应,所以记忆B细胞能够引发比初次反应更快,也更强烈的体液免疫反应,而浆细胞所引发的体液免疫反应有限。The Chinese invention patent application with the application publication number CN111303280A discloses a fully human monoclonal antibody against SARS-CoV-2 with high neutralization activity. The above patent provides a fully human monoclonal antibody whose recognition region is the S1 non-RBD region, However, since the new coronavirus invades the host cell by binding to the ACE2 of the host cell through RBD, the fully human monoclonal antibody obtained in the above-mentioned patent has limited blocking effect on the virus, and the above-mentioned patent obtains the antibody cDNA by labeling plasma cells, but Compared with plasma cells, memory B cells respond quickly after activation, so memory B cells can trigger a faster and stronger humoral immune response than the initial response, while plasma cells trigger a limited humoral immune response.
发明内容SUMMARY OF THE INVENTION
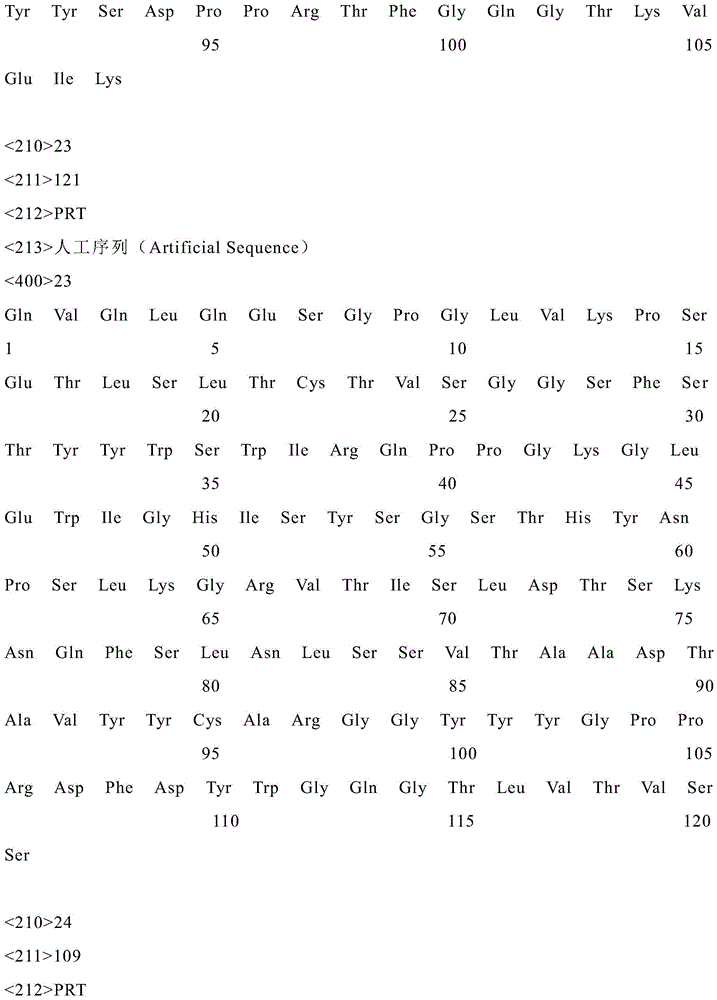
为了达到上述目的,本发明提供了新冠病毒RBD特异性单克隆抗体,具体的,抗体的重链氨基酸序列如SEQ ID NO:1所示;轻链氨基酸序列如SEQ ID NO:2所示(单抗1-CQT149)。重链氨基酸序列还可如SEQ ID NO:3所示;轻链氨基酸序列还可如SEQ ID NO:4所示(单抗2-CQT190)。重链氨基酸序列还可如SEQ ID NO:5所示;轻链氨基酸序列还可如SEQID NO:6所示(单抗3-CQT191)。重链氨基酸序列还可如SEQ ID NO:7所示;轻链氨基酸序列还可如SEQ ID NO:8所示(单抗4-CQT195)。重链氨基酸序列还可如SEQ ID NO:9所示;轻链氨基酸序列还可如SEQ ID NO:10所示(单抗5-CQT197)。重链氨基酸序列还可如SEQ ID NO:11所示;轻链氨基酸序列还可如SEQ ID NO:12所示(单抗6-CQT198)。重链氨基酸序列还可如SEQ ID NO:13所示;轻链氨基酸序列还可如SEQ ID NO:14所示(单抗7-CQT199)。重链氨基酸序列还可如SEQ ID NO:15所示;轻链氨基酸序列还可如SEQ ID NO:16所示(单抗8-CQT200)。重链氨基酸序列还可如SEQ ID NO:17所示;轻链氨基酸序列还可如SEQ ID NO:18所示(单抗9-CQT201)。重链氨基酸序列还可如SEQ ID NO:19所示;轻链氨基酸序列还可如SEQ ID NO:20所示(单抗10-CQT201)。重链氨基酸序列还可如SEQ ID NO:21所示;轻链氨基酸序列还可如SEQ ID NO:22所示(单抗11-CQT202)。重链氨基酸序列还可如SEQ ID NO:23所示;轻链氨基酸序列还可如SEQ ID NO:24所示(单抗12-CQT203)。重链氨基酸序列还可如SEQ ID NO:25所示;轻链氨基酸序列还可如SEQ ID NO:26所示(单抗13-CQT206)。重链氨基酸序列还可如SEQ ID NO:27所示;轻链氨基酸序列还可如SEQ ID NO:28所示(单抗14-CQT209)。重链氨基酸序列还可如SEQ ID NO:29所示;轻链氨基酸序列还可如SEQ ID NO:30所示(单抗15-CQT210)。重链氨基酸序列还可如SEQ ID NO:31所示;轻链氨基酸序列还可如SEQ ID NO:32所示(单抗16-CQT212)。重链氨基酸序列还可如SEQ ID NO:33所示;轻链氨基酸序列还可如SEQ ID NO:34所示(单抗17-CQT215)。重链氨基酸序列还可如SEQ ID NO:35所示;轻链氨基酸序列还可如SEQ ID NO:36所示(单抗18-CQT216)。重链氨基酸序列还可如SEQ ID NO:37所示;轻链氨基酸序列还可如SEQ ID NO:38所示(单抗19-CQT217)。重链氨基酸序列还可如SEQ ID NO:39所示;轻链氨基酸序列还可如SEQ ID NO:40所示(单抗20-CQT218)。In order to achieve the above purpose, the present invention provides a novel coronavirus RBD-specific monoclonal antibody, specifically, the heavy chain amino acid sequence of the antibody is shown in SEQ ID NO: 1; the light chain amino acid sequence is shown in SEQ ID NO: 2 (single anti-1-CQT149). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 3; the light chain amino acid sequence can also be shown in SEQ ID NO: 4 (mab 2-CQT190). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 5; the light chain amino acid sequence can also be shown in SEQ ID NO: 6 (mab 3-CQT191). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 7; the light chain amino acid sequence can also be shown in SEQ ID NO: 8 (mab 4-CQT195). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 9; the light chain amino acid sequence can also be shown in SEQ ID NO: 10 (mab 5-CQT197). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 11; the light chain amino acid sequence can also be shown in SEQ ID NO: 12 (mab 6-CQT198). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 13; the light chain amino acid sequence can also be shown in SEQ ID NO: 14 (mAb 7-CQT199). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 15; the light chain amino acid sequence can also be shown in SEQ ID NO: 16 (mAb 8-CQT200). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 17; the light chain amino acid sequence can also be shown in SEQ ID NO: 18 (mAb 9-CQT201). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 19; the light chain amino acid sequence can also be shown in SEQ ID NO: 20 (mAb 10-CQT201). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 21; the light chain amino acid sequence can also be shown in SEQ ID NO: 22 (mAb 11-CQT202). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 23; the light chain amino acid sequence can also be shown in SEQ ID NO: 24 (mAb 12-CQT203). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 25; the light chain amino acid sequence can also be shown in SEQ ID NO: 26 (mab 13-CQT206). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 27; the light chain amino acid sequence can also be shown in SEQ ID NO: 28 (mab 14-CQT209). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 29; the light chain amino acid sequence can also be shown in SEQ ID NO: 30 (mab 15-CQT210). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 31; the light chain amino acid sequence can also be shown in SEQ ID NO: 32 (mab 16-CQT212). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 33; the light chain amino acid sequence can also be shown in SEQ ID NO: 34 (mAb 17-CQT215). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 35; the light chain amino acid sequence can also be shown in SEQ ID NO: 36 (mAb 18-CQT216). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 37; the light chain amino acid sequence can also be shown in SEQ ID NO: 38 (mAb 19-CQT217). The heavy chain amino acid sequence can also be shown in SEQ ID NO: 39; the light chain amino acid sequence can also be shown in SEQ ID NO: 40 (mAb 20-CQT218).
本发明还提供了上述新冠病毒RBD特异性单克隆抗体,在制备检测或诊断SARS-CoV-2试剂或疫苗或药物中的应用;还提供了编码上述新冠病毒RBD特异性单克隆抗体的核酸分子;还提供了含有上述核酸分子的表达盒、重组载体、重组菌或转基因细胞系;还提供了上述表达盒、重组载体、重组菌或转基因细胞系在制备产品中的应用。The present invention also provides the application of the above-mentioned novel coronavirus RBD-specific monoclonal antibody in the preparation of reagents or vaccines or drugs for detecting or diagnosing SARS-CoV-2; and also provides a nucleic acid molecule encoding the above-mentioned novel coronavirus RBD-specific monoclonal antibody ; also provide an expression cassette, recombinant vector, recombinant bacteria or transgenic cell line containing the above-mentioned nucleic acid molecule; also provide the application of the above-mentioned expression cassette, recombinant vector, recombinant bacteria or transgenic cell line in the preparation of products.
本发明还提供了产品,包括上述新冠病毒RBD特异性单克隆抗体;产品用途如下(b1)-(b4)中的任一种:(b1)结合新型冠状病毒SARS-CoV-2;(b2)检测结合新型冠状病毒SARS-CoV-2;(b3)结合新型冠状病毒SARS-CoV-2的S蛋白;(b4)检测新型冠状病毒SARS-CoV-2的S蛋白The present invention also provides a product, including the above-mentioned new coronavirus RBD-specific monoclonal antibody; the use of the product is any of the following (b1)-(b4): (b1) combined with the new coronavirus SARS-CoV-2; (b2) Detection of binding to the new coronavirus SARS-CoV-2; (b3) binding to the S protein of the new coronavirus SARS-CoV-2; (b4) detection of the S protein of the new coronavirus SARS-CoV-2
本发明还提供了药物组合物,包括上述新冠病毒RBD特异性单克隆抗体和药学上可接受的赋形剂、稀释剂或载体。The present invention also provides a pharmaceutical composition comprising the above-mentioned novel coronavirus RBD-specific monoclonal antibody and a pharmaceutically acceptable excipient, diluent or carrier.
优选的,上述新冠病毒RBD特异性单克隆抗体通过分选特异性记忆B细胞,采用RT-PCR扩增获得抗体可变区cDNA。Preferably, the above-mentioned novel coronavirus RBD-specific monoclonal antibody is obtained by sorting specific memory B cells and amplifying the antibody variable region cDNA by RT-PCR.
本发明的原理和有益效果在于:The principle and beneficial effects of the present invention are:
(1)本发明提供的单克隆抗体具有RBD特异性,与针对S1非RBD区的单可隆抗体相比,本发明提供的单克隆抗体与RBD结合,为抗体药物筛选,诊断、预防和治疗新冠肺炎提供了更加广泛的应用价值。(1) The monoclonal antibody provided by the present invention has RBD specificity. Compared with the monoclonal antibody against the non-RBD region of S1, the monoclonal antibody provided by the present invention is combined with RBD, which is used for antibody drug screening, diagnosis, prevention and treatment. COVID-19 provides wider application value.
(2)本发明提供的单克隆抗体是通过分选RBD特异性记忆B细胞而得到,与通过分选浆细胞的现有技术相比,本发明制备的单克隆抗体能够引发更强烈的体液免疫反应。另外,本发明只针对RBD特异性记忆B细胞进行后续RT-PCR、巢式PCR和抗体功能分析,大大提高了单克隆抗体与RBD特异性结合能力。(2) The monoclonal antibody provided by the present invention is obtained by sorting RBD-specific memory B cells. Compared with the prior art by sorting plasma cells, the monoclonal antibody prepared by the present invention can induce stronger humoral immunity reaction. In addition, the present invention only performs subsequent RT-PCR, nested PCR and antibody function analysis for RBD-specific memory B cells, which greatly improves the specific binding ability of the monoclonal antibody to RBD.
附图说明Description of drawings
图1为采用流式细胞仪分析记忆B细胞的细胞分选图;Figure 1 is a cell sorting diagram of memory B cells analyzed by flow cytometry;
图2为采用流式细胞仪分析RBD特异性记忆B细胞的细胞分选图;Figure 2 is a cell sorting diagram of RBD-specific memory B cells analyzed by flow cytometry;
图3为单细胞抗体基因PCR产物凝胶电泳图;Figure 3 is a gel electrophoresis image of a single-cell antibody gene PCR product;
图4为PCR扩增包含CMV启动子、WPRE-γ或WPRE-κ元件抗体基因表达盒后的琼脂糖凝胶电泳图;Figure 4 is an agarose gel electrophoresis image after PCR amplification of an antibody gene expression cassette comprising CMV promoter, WPRE-γ or WPRE-κ element;
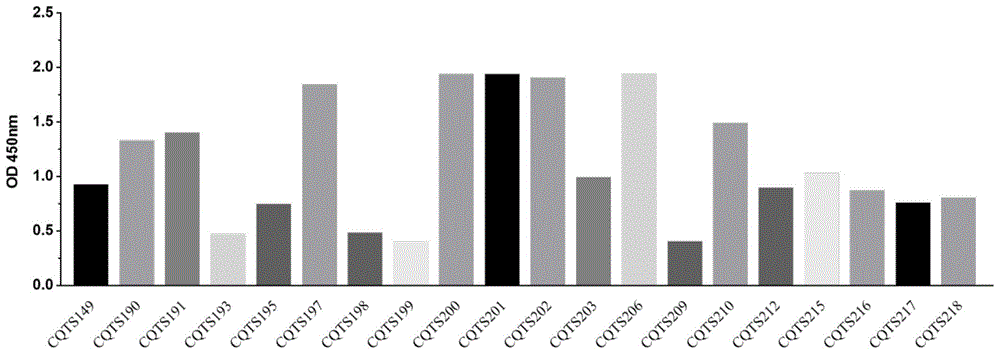
图5为RBD特异性检测结果图。Figure 5 is a graph showing the results of RBD-specific detection.
具体实施方式Detailed ways
下面将结合本发明实施例的附图,对本发明实施例中的技术方案进行清楚、完整地描述,显然,所描述的实施例仅仅是本发明的一部分实施例,而不是全部的实施例。基于本发明中的实施例,本领域普通技术人员在没有作出创造性劳动前提下所获得的所有其他实施例,都属于本发明保护的范围。The technical solutions in the embodiments of the present invention will be clearly and completely described below with reference to the accompanying drawings of the embodiments of the present invention. Obviously, the described embodiments are only a part of the embodiments of the present invention, rather than all the embodiments. Based on the embodiments of the present invention, all other embodiments obtained by those of ordinary skill in the art without creative efforts shall fall within the protection scope of the present invention.
实施例1Example 1
本实施例提供一种新冠病毒RBD特异性单克隆抗体,重链氨基酸序列如SEQ IDNO:1所示;轻链氨基酸序列如SEQ ID NO:2所示。This example provides a novel coronavirus RBD-specific monoclonal antibody, the amino acid sequence of the heavy chain is shown in SEQ ID NO: 1; the amino acid sequence of the light chain is shown in SEQ ID NO: 2.
本实施例还提供了上述新冠病毒RBD特异性单克隆抗体,在制备检测或诊断SARS-CoV-2试剂或药物中的应用。This embodiment also provides the application of the above-mentioned novel coronavirus RBD-specific monoclonal antibody in the preparation of reagents or drugs for detecting or diagnosing SARS-CoV-2.
在实际生产时,可以采用本实施例得到RBD特异性单克隆抗体制备核酸分子,或者制备包含该核酸分子的表达盒、重组载体、重组菌或转基因细胞系,或者制备药物组合物,该药物组合物包括上述新冠病毒RBD特异性单克隆抗体和药学上可接受的赋形剂、稀释剂或载体。In actual production, the RBD-specific monoclonal antibody obtained in this example can be used to prepare a nucleic acid molecule, or an expression cassette, a recombinant vector, a recombinant bacteria or a transgenic cell line containing the nucleic acid molecule can be prepared, or a pharmaceutical composition can be prepared. The substance includes the above-mentioned novel coronavirus RBD-specific monoclonal antibody and a pharmaceutically acceptable excipient, diluent or carrier.
在应用时,本实施例得到RBD特异性单克隆抗体制备的相关产品,可具有如下(b1)-(b4)中的任一种的用途:(b1)结合新型冠状病毒SARS-CoV-2;(b2)检测结合新型冠状病毒SARS-CoV-2;(b3)结合新型冠状病毒SARS-CoV-2的S蛋白;(b4)检测新型冠状病毒SARS-CoV-2的S蛋白。In application, this example obtains related products prepared by RBD-specific monoclonal antibodies, which can be used in any of the following (b1)-(b4): (b1) binding to the new coronavirus SARS-CoV-2; (b2) Detection of binding to the new coronavirus SARS-CoV-2; (b3) detection of the S protein of the new coronavirus SARS-CoV-2; (b4) detection of the S protein of the new coronavirus SARS-CoV-2.
实施例2-20Example 2-20
实施例2-20与实施例1的区别在于:RBD特异性单克隆抗体的氨基酸序列不同,实施例2-20的氨基酸序列如下表所示:The difference between Example 2-20 and Example 1 is that the amino acid sequences of RBD-specific monoclonal antibodies are different, and the amino acid sequences of Example 2-20 are shown in the following table:
以上实施例1-20所提供的RBD特异性单克隆抗体均通过以下方法得到:首先从新冠肺炎康复患者的外周血中分选得到单个RBD特异性记忆B细胞,然后获得RBD特异性记忆B细胞的mRNA,再通过RT-PCR和巢式PCR构建抗体可变区基因表达盒,再将抗体可变区基因表达盒转导入293T细胞表达抗体并收集上清,用ELISA法检测上清的RBD特异性,筛选得到RBD特异性单克隆抗体。The RBD-specific monoclonal antibodies provided in the above Examples 1-20 were all obtained by the following methods: first, a single RBD-specific memory B cell was obtained from the peripheral blood of recovered patients with COVID-19, and then a single RBD-specific memory B cell was obtained. The mRNA was then constructed by RT-PCR and nested PCR to construct the antibody variable region gene expression cassette, and then the antibody variable region gene expression cassette was transfected into 293T cells to express the antibody and the supernatant was collected, and the RBD specificity of the supernatant was detected by ELISA. After screening, RBD-specific monoclonal antibodies were obtained.
具体包括以下步骤:Specifically include the following steps:
S1、采集若干名新冠肺炎康复患者外周血,分离得到PBMC,在-80℃的冰箱中冻存备用。S1. Collect peripheral blood of several recovered patients with new coronary pneumonia, isolate PBMCs, and freeze them in a -80°C refrigerator for future use.
S2、首先采用去死细胞染料(Dead Dye)去除S1得到的PBMC的死细胞,然后采用CD19、mIg-G、mIg-D和S-RBD对PBMC中活的RBD特异性并且结合能力高的记忆B细胞染色标记,筛选出针对RBD特异性记忆B细胞;使用流式细胞分选仪将特异性记忆B细胞分选到96孔板上,每个孔内有一个特异性记忆B细胞,在-80℃的冰箱中冻存备用。S2. First, use dead cell dye (Dead Dye) to remove dead cells of PBMCs obtained by S1, and then use CD19, mIg-G, mIg-D and S-RBD to memory specific and high binding capacity of live RBDs in PBMCs B cells are stained and marked to screen out RBD-specific memory B cells; use flow cytometry to sort specific memory B cells into 96-well plates, each well has one specific memory B cell, and it is in- Store in a freezer at 80°C for later use.
具体的,本实施例优选的Dead Dye染色时的浓度范围为1-2μg/mL,本实施例优选Dead Dye染色时的浓度为1.5μg/mL;CD19为Biolegend生产的B细胞标记物,染色时的浓度范围为1-2μg/mL,本实施例优选CD19染色时的浓度为1.5μg/mL。mIg-G为Biolegend生产的B细胞标表面受体,染色时的浓度范围为1-2μg/mL,本实施例优选mIg-G染色时的浓度为1.5μg/mL;mIg-D为Biolegend生产的B细胞表面受体,染色时的浓度范围为1-2μg/mL,本实施例优选mIg-D染色时的浓度为1.5μg/mL;S-RBD为sinobiological生产的新冠病毒是蛋白受体结构域,染色时的浓度范围为1-2μg/mL,本实施例优选S-RBD染色时的浓度为1.5μg/mL。Specifically, the preferred concentration range of Dead Dye staining in this embodiment is 1-2 μg/mL, and the preferred concentration of Dead Dye staining in this embodiment is 1.5 μg/mL; CD19 is a B cell marker produced by Biolegend. The concentration range of CD19 is 1-2 μg/mL, and the preferred concentration of CD19 in this embodiment is 1.5 μg/mL. mIg-G is a B cell labeled surface receptor produced by Biolegend, and the concentration range during staining is 1-2 μg/mL. In this embodiment, the preferred concentration of mIg-G staining is 1.5 μg/mL; mIg-D is produced by Biolegend. B cell surface receptor, the concentration range of staining is 1-2 μg/mL, in this embodiment, the preferred concentration of mIg-D staining is 1.5 μg/mL; S-RBD is the new coronavirus produced by sinobiological, which is a protein receptor domain , the concentration range during staining is 1-2 μg/mL, and in this embodiment, the preferred concentration of S-RBD during staining is 1.5 μg/mL.
通过流式细胞仪分选RBD特异性记忆B细胞的,通过CD19、mIg-G、mIg-D和S-RBD对PBMC的细胞分选得到对S-RBD具有特异性记忆B细胞的细胞分选图如图1和图2所示,其中图2中的Batch ID 0428、0505、0522、0528是筛选批次。本实施例采用CD19、mIg-G、mIg-D和S-RBD筛选出针对RBD特异性记忆B细胞的原理在于:将PBMC用去死细胞染料(Dead Dye)、B细胞标记物CD19、记忆B细胞标记物mIg-G阳性和mIg-D阴性以及RBD特异性IgG表达的记忆B细胞进行染色,然后使用流式细胞分析仪将细胞群中CD19细胞群划分出来,再通过从CD19阳性细胞群中划分mIg-G+mIg-D-细胞群,再从mIg-G+mIg-D-细胞群划分RBD阳性的记忆B细胞,再通过流式细胞分选仪将RBD阳性的记忆B细胞进行分选。Sorting of RBD-specific memory B cells by flow cytometry, cell sorting of PBMCs by CD19, mIg-G, mIg-D, and S-RBD yields cell sorting of memory B cells specific for S-RBD Figures are shown in Figures 1 and 2, where
S3、分选得到单个RBD特异性记忆B细胞的mRNA,采用RT-PCR扩增获得抗体可变区cDNA。具体的,使用RT-PCR扩增抗体可变区cDNA时,本实施例所设计的引物的引物前段设计通用Leader(参见引物序列表一和引物序列表二),有效提高了抗体基因的扩增率,实验结果如图3所示。S3. The mRNA of a single RBD-specific memory B cell is obtained by sorting, and the variable region cDNA of the antibody is obtained by RT-PCR amplification. Specifically, when RT-PCR is used to amplify the variable region cDNA of the antibody, the primer front section of the primer designed in this example is designed with a general leader (see Primer Sequence Table 1 and Primer Sequence Table 2), which effectively improves the amplification of antibody genes. The experimental results are shown in Figure 3.
S4、采用巢式PCR扩增S1-S3得到的抗体可变区cDNA,构建抗体可变区基因表达盒。S4, using nested PCR to amplify the antibody variable region cDNA obtained from S1-S3 to construct an antibody variable region gene expression cassette.
S3和S4总共通过以下六部分进行:(1)提取RBD特异性记忆B细胞的mRNA;(2)单个细胞mRNA逆转录(RT);(3)加G尾(TDT);(4)第一轮PCR(1st PCR);(5)第二轮PCR(2nd PCR);(6)BCR-ORF PCR扩增构建基因表达盒;(7)CMV、WPRE-γ/κ/l片段扩增及CMV、BCR-Vγ/κ/l((6)的产物)、WPRE-γ/κ/l重叠PCR(Overlap PCR)预连接;(8)BCR-γORF、BCR-κORF、BCR-lPCR扩增。S3 and S4 were performed in a total of six parts: (1) extraction of mRNA from RBD-specific memory B cells; (2) single cell mRNA reverse transcription (RT); (3) G-tailing (TDT); (4) first Round PCR (1st PCR); (5) Second round PCR (2nd PCR); (6) BCR-ORF PCR amplification to construct gene expression cassette; (7) CMV, WPRE-γ/κ/1 fragment amplification and CMV , BCR-Vγ/κ/l (product of (6)), WPRE-γ/κ/l overlap PCR (Overlap PCR) pre-ligation; (8) BCR-γORF, BCR-κORF, BCR-1 PCR amplification.
各部分反应液配制及反应条件如下:The preparation and reaction conditions of each part of the reaction solution are as follows:
(1)采用DynabeadsTM mRNA DIRECTTM Purification Kit(ThermoFisherscientific)进行单细胞mRNA提取,具体包括以下步骤:(1) Use DynabeadsTM mRNA DIRECTTM Purification Kit ( ThermoFisherscientific) to extract single-cell mRNA, which specifically includes the following steps:
①离心:从-80℃冰箱中取出分选有单个RBD特异性记忆B细胞的96孔板后,600×g离心30s,使细胞离心于孔底部;① Centrifugation: After taking out the 96-well plate with single RBD-specific memory B cells sorted from the -80°C refrigerator, centrifuge at 600×g for 30s to centrifuge the cells at the bottom of the well;
②清洗:将Dynabeads oligo(dT)25微球瓶取出后涡旋混匀,按照2μl/孔吸取足量微球,放置于磁铁块上,静置30s,弃上清,用500μl的Lysis Buffer重悬;②Washing: Take out the Dynabeads oligo(dT) 25 microsphere bottle and mix by vortexing, suck up enough microspheres according to 2μl/well, place it on the magnet block, let it stand for 30s, discard the supernatant, resuspend it with 500μl Lysis Buffer hanging;
③配制:按照9μl/孔Lysis Buffer加入到50mL的离心管中,将上述500μl微球悬液加入其中,用枪吹匀;③Preparation: add 9μl/well Lysis Buffer into a 50mL centrifuge tube, add the above 500μl microsphere suspension into it, and blow it evenly with a gun;
④分装:用八连管分装微球,随后采用排枪将其按照9μl/孔加入到细胞板中;④ Dispense: Dispense the microspheres with an eight-connected tube, and then add 9 μl/well of the microspheres to the cell plate using a discharge gun;
⑤润洗:96孔板贴膜,随后润洗管壁四周,共2个循环;⑤Rinse: 96-well plate with film, then rinse around the tube wall, a total of 2 cycles;
⑥孵育:室温静置5min,使RBD特异性记忆B细胞的mRNA充分释放并结合到微球上,孵育结束后,600×g瞬时离心,使微球离心于孔底部。将96孔板放置于DynaMagTM-96sideMagnet磁板上,用排枪吸弃上清;⑥ Incubation: stand at room temperature for 5 minutes to fully release the mRNA of RBD-specific memory B cells and bind to the microspheres. After incubation, centrifuge briefly at 600 × g to make the microspheres centrifuge at the bottom of the well. Place the 96-well plate on the DynaMag TM -96sideMagnet magnetic plate, and discard the supernatant with a discharge gun;
⑦Wash A清洗:按照8μl/孔加入Washing Buffer A,来回走板7-8次,使微球充分洗涤,弃上清;⑦Wash A washing: Add Washing Buffer A according to 8μl/well, and run the plate back and forth 7-8 times to wash the microspheres thoroughly, and discard the supernatant;
⑧Wash B清洗:按照8μl/孔加入Washing Buffer B,来回走板7-8次,使微球充分洗涤,弃上清,随后按照10μl/孔加入预先配制的逆转录(RT)反应液。试剂配制及反应条件如下述(2)描述。⑧ Washing with Wash B: Add Washing Buffer B at 8 μl/well, go back and forth 7-8 times to wash the microspheres thoroughly, discard the supernatant, and then add the pre-prepared reverse transcription (RT) reaction solution at 10 μl/well. Reagent preparation and reaction conditions are described in (2) below.
(2)逆转录(RT)(10μl体系)(2) Reverse transcription (RT) (10 μl system)
所需配制的试剂如下表1所示:The reagents to be prepared are shown in Table 1 below:
反应条件:42℃for 60min(每20min混合一次);Reaction conditions: 42°C for 60min (mix every 20min);
反应结束后,600×g瞬时离心96孔板,然后将96孔板放置于DynaMagTM-96sideMagnet磁板上,用排枪吸弃上清,随后按照10μl/孔加入预先配制的TDT反应液,试剂配制及反应条件如下述(3)描述。After the reaction, the 96-well plate was centrifuged at 600×g for a short time, and then the 96-well plate was placed on the DynaMag TM -96sideMagnet magnetic plate. and reaction conditions are as described in (3) below.
(3)加G尾(TDT)(10μl体系)(3) Add G tail (TDT) (10 μl system)
所需配制的试剂如下表2所示:The reagents to be prepared are shown in Table 2 below:
反应条件:37℃for 40min(每20min混合一次)。Reaction conditions: 37°C for 40 min (mixing every 20 min).
反应结束,600×g瞬时离心96孔板,然后将其放置于DynaMagTM-96side Magnet磁板上,用排枪吸弃上清,随后按照10μl/孔加入预先配制的第一轮PCR(1st PCR)反应液,试剂配制及反应条件如下述(4)描述。After the reaction was completed, the 96-well plate was centrifuged at 600×g for a short time, and then placed on a DynaMag TM -96side Magnet magnetic plate. The supernatant was discarded with a row gun, and then 10 μl/well was added to the pre-prepared first-round PCR (1st PCR) The reaction solution, reagent preparation and reaction conditions are described in (4) below.
(4)1st PCR(10μl体系)(引物序列参见引物序列表)(4) 1st PCR (10 μl system) (see the primer sequence table for primer sequences)
所需配制的试剂如下表3所示:The reagents to be prepared are shown in Table 3 below:
基于PCR原理,1st PCR的实验反应条件为:①95℃预变性3min;②95℃变性15sec,60℃退火5sec,72℃延伸1min,30-35cycles,本实施例优选30cycles;③72℃循环外延伸5min,4℃保存。Based on the PCR principle, the experimental reaction conditions for 1st PCR are: ① 95°C pre-denaturation for 3 min; ② 95°C denaturation for 15 sec, 60°C annealing for 5 sec, 72°C extension for 1 min, 30-35 cycles, 30 cycles are preferred in this example; ③ 72°C external extension for 5 min, Store at 4°C.
(5)第二轮PCR(2nd PCR)(10μl体系)(引物序列参见引物序列表一和引物序列表二)(5) The second round of PCR (2nd PCR) (10 μl system) (see primer sequence table 1 and primer sequence table 2 for primer sequences)
所需配制的试剂如下表4所示:The reagents to be prepared are shown in Table 4 below:
基于PCR原理,2nd PCR的实验反应条件为:①95℃预变性3min;②95℃变性15sec,60℃退火5s,72℃延伸1min,30-35cycles,本实施例优选35cycles;72℃循环外延伸5min,4℃保存。Based on the PCR principle, the experimental reaction conditions of 2nd PCR are as follows: (1) Pre-denaturation at 95°C for 3 min; (2) Denaturation at 95°C for 15sec, annealing at 60°C for 5s, extension at 72°C for 1min, 30-35cycles, in this example, 35cycles is preferred; Store at 4°C.
PCR结束后:每孔取4μl进行1.5%琼脂糖凝胶电泳。将Gamma链与Kappa链或Lamada链配对的细胞孔送测序。After PCR: take 4 μl per well for 1.5% agarose gel electrophoresis. Cell wells with Gamma chains paired with Kappa chains or Lamada chains are sent for sequencing.
(6)抗体表达盒(BCR-ORF)的扩增和构建(6) Amplification and construction of antibody expression cassette (BCR-ORF)
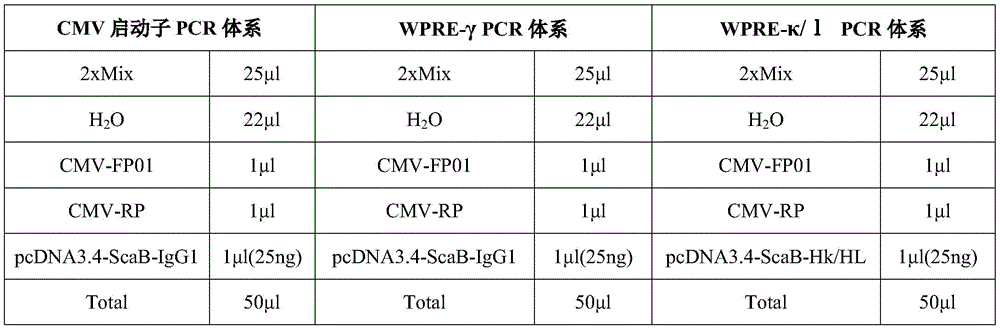
PCR扩增启动子区(CMV启动子)、WPRE-γ(抗体gamma链)和WPRE-κ(抗体kappa链),PCR扩增体系如下表5所示:PCR amplification of the promoter region (CMV promoter), WPRE-γ (antibody gamma chain) and WPRE-κ (antibody kappa chain), the PCR amplification system is shown in Table 5 below:
PCR扩增条件为:①95℃预变性3min;②95℃变性15sec,56℃退火15sec,72℃延伸1min,30cycles;③72℃循环外延伸5min,12℃保存。PCR amplification conditions were: ① pre-denaturation at 95°C for 3 min; ② denaturation at 95°C for 15 sec, annealing at 56°C for 15 sec, extension at 72°C for 1 min, 30 cycles; ③ external extension at 72°C for 5 min, and storage at 12°C.
(7)CMV、WPRE-γ/κ/l片段扩增及CMV、BCR-Vγ/κ/l、WPRE-γ/κ/l重叠PCR(Overlap PCR)预连接(7) CMV, WPRE-γ/κ/1 fragment amplification and CMV, BCR-Vγ/κ/1, WPRE-γ/κ/1 overlap PCR (Overlap PCR) pre-ligation
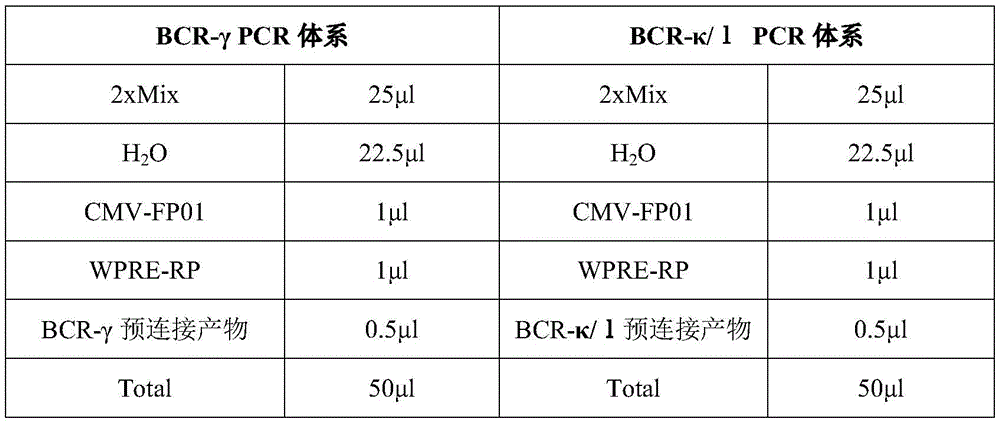
实验体系如下表6所示:The experimental system is shown in Table 6 below:
PCR扩增条件为:95℃预变性3min;95℃变性15sec,50℃退火15sec,72℃延伸1.5min,10cycles;72℃循环外延伸5min,12℃保存。PCR amplification conditions were: pre-denaturation at 95°C for 3 min; denaturation at 95°C for 15 sec, annealing at 50°C for 15 sec, extension at 72°C for 1.5 min, 10 cycles; external extension at 72°C for 5 min, and storage at 12°C.
(8)BCR-γORF、BCR-κORF、BCR-l PCR扩增(8) PCR amplification of BCR-γORF, BCR-κORF, BCR-1
实验体系如下表7所示:The experimental system is shown in Table 7 below:
PCR扩增程序:95℃预变性3min;95℃变性15sec,58℃退火15sec,72℃延伸1.5min,30cycles;72℃循环外延伸5min,12℃保存。PCR amplification program: pre-denaturation at 95°C for 3min; denaturation at 95°C for 15sec, annealing at 58°C for 15sec, extension at 72°C for 1.5min, 30 cycles; external extension at 72°C for 5min, and storage at 12°C.
扩增后,采用琼脂糖凝胶电泳,凝胶成像分析得到的抗体可变区基因大小是否正确,实验结果如图4所示,Marker在中间位置,条带在5000bp处。After amplification, agarose gel electrophoresis was used, and gel imaging was used to analyze whether the size of the antibody variable region gene was correct. The experimental results are shown in Figure 4, the Marker is in the middle position, and the band is at 5000bp.
BCR-γORF和BCR-κ/ORF乙醇沉淀:BCR-γORF和BCR-κORF的PCR产物各取30μl置于8连管中,再加入120μl无水乙醇,6μl醋酸钠溶液,充分混匀,-80℃静置30min;10000rpm,离心20min,弃上清,依次用200μl的70%乙醇和无水乙醇各漂洗一次,于56℃乙醇充分挥发,加入40μl无菌水,振荡,使沉淀充分溶解,检测抗体可变区基因的浓度。BCR-γORF and BCR-κ/ORF ethanol precipitation: Take 30 μl of PCR products of BCR-γORF and BCR-κORF and put them in 8-connected tubes, then add 120 μl absolute ethanol and 6 μl sodium acetate solution, mix well, -80 ℃ stand for 30 min; 10000 rpm, centrifuge for 20 min, discard the supernatant, rinse with 200 μl of 70% ethanol and anhydrous ethanol in turn, fully volatilize the ethanol at 56 ℃, add 40 μl of sterile water, shake to fully dissolve the precipitate, and detect The concentration of antibody variable region genes.
S3和S4所用到的Leader引物参见如下的引物序列表一:The Leader primers used in S3 and S4 are shown in the following primer sequence table 1:
S3和S4所用到的所用到J-region引物参见如下的引物序列表二:The J-region primers used in S3 and S4 are shown in the following primer sequence table 2:
S5、将S4得到的抗体可变区基因表达盒转导入293T细胞48小时内表达抗体并收集上清,用ELISA法检测上清的RBD特异性,筛选RBD特异性全人源单克隆抗体。S5. The antibody variable region gene expression cassette obtained in S4 was transfected into 293T cells to express the antibody within 48 hours, and the supernatant was collected. The RBD specificity of the supernatant was detected by ELISA, and the RBD-specific fully human monoclonal antibody was screened.
(A)使用PBS稀释抗原(终浓度2μg/mL),10μl/孔,包被384孔ELISA板4℃过夜或37℃包被2h(本实施例优选4℃过夜)。NOTE:加完后瞬时离心保证液体在底部。(A) Dilute the antigen with PBS (final concentration 2 μg/mL), 10 μl/well, coat 384-well ELISA plate at 4°C overnight or at 37°C for 2 hours (preferably 4°C overnight in this example). NOTE: Centrifuge briefly after adding to ensure the liquid is at the bottom.
实验体系如下表8所示:The experimental system is shown in Table 8 below:
(B)配制PBST(0.05%Tween 20,Cat#TB220):1L的PBS加入0.5mL的Tween 20;(B) Preparation of PBST (0.05% Tween 20, Cat#TB220): 1L of PBS was added with 0.5mL of Tween 20;
PBST机洗板子(Thermoscientific wellwash versa)或者手洗(机洗完的板子依然要手动拍板/使用微孔板离心机(MPC-P25)离心1min,使板子看不见有水和气泡)。PBST machine-washed plates (Thermoscientific wellwash versa) or hand-washed (the plates after machine washing still need to be tapped manually/centrifuged with a microplate centrifuge (MPC-P25) for 1 min, so that no water and air bubbles can be seen on the plate).
封闭:80μl的5%BSA(BioFroxx,Cat.NO:4240GR100)(PBST配制)加入上述洗好的板子里,放置于37℃的孵育箱孵育1h。PBST机洗板子或者手洗。Blocking: 80 μl of 5% BSA (BioFroxx, Cat. NO: 4240GR100) (prepared with PBST) was added to the above washed plate and placed in a 37° C. incubator for 1 h. Machine wash plates in PBST or hand wash.
(C)加样及标准品。其中,标准品:10μl/well原浓度1μg/mL,梯度稀释为250ng/mL、125ng/mL、62.5ng/mL、31.25ng/mL、15.63ng/mL、7.81ng/mL、3.9ng/mL和1.95ng/mL。(封闭液稀释);样品:转染抗体基因的细胞上清液。阴性对照/空白孔:封闭液10μl/well。(C) Sample addition and standard. Among them, the standard: 10μl/well original concentration 1μg/mL, the gradient dilution is 250ng/mL, 125ng/mL, 62.5ng/mL, 31.25ng/mL, 15.63ng/mL, 7.81ng/mL, 3.9ng/mL and 1.95ng/mL. (dilution in blocking solution); sample: supernatant of cells transfected with antibody gene. Negative control/blank well: 10 μl/well of blocking solution.
在37℃孵育30min。PBST机洗板子或者手洗。Incubate for 30 min at 37°C. Machine wash plates in PBST or hand wash.
(D)加二抗,加入的浓度为10μl/well,然后在37℃下孵育30min。(D) Secondary antibody was added at a concentration of 10 μl/well, and then incubated at 37°C for 30 min.
实验体系如下表9所示:The experimental system is shown in Table 9 below:
PBST机洗板子或者手洗。10μl/well的PNPP(对硝基苯磷酸二钠),使用(Thermoscientific Muttiskan GO)检测5min、10min、15min、20min、25min、30min、35min、40min、45min、50min、55min和60min的OD(450mm)值。50mg的PNPP粉末(Thermo,Prod#34045)+40mL的ddH2O+10mL的Diethanol aminesubstrate Buffer(5X),PNPP避光4℃储存。Machine wash plates in PBST or hand wash. 10μl/well of PNPP (disodium p-nitrophenylphosphate), using (Thermoscientific Muttiskan GO) to detect OD (450mm) for 5min, 10min, 15min, 20min, 25min, 30min, 35min, 40min, 45min, 50min, 55min and 60min value. 50 mg of PNPP powder (Thermo, Prod #34045) + 40 mL of ddH 2 O + 10 mL of Diethanol aminesubstrate Buffer (5X), PNPP was stored at 4°C in the dark.
实验结果如图5所示,OD值大于0.1为阳性。The experimental results are shown in Figure 5, and the OD value greater than 0.1 is positive.
以上所述仅为本发明的优选实施例,对本发明而言仅是说明性的,而非限制性的;本领域普通技术人员理解,在本发明权利要求所限定的精神和范围内可对其进行许多改变,修改,甚至等效变更,但都将落入本发明的保护范围内。The above descriptions are only preferred embodiments of the present invention, which are only illustrative rather than restrictive for the present invention; those of ordinary skill in the art will understand that the invention can be modified within the spirit and scope defined by the claims of the present invention. Numerous changes, modifications, and even equivalent changes can be made, but will fall within the protection scope of the present invention.
Claims (7)
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010839860.XA CN111925442B (en) | 2020-08-19 | 2020-08-19 | New coronavirus RBD specific monoclonal antibody and application |
| CN202210906292.XA CN115925900B (en) | 2020-08-19 | 2020-08-19 | Novel coronavirus RBD specific monoclonal antibody and application |
| CN202210501598.7A CN115340600B (en) | 2020-08-19 | 2020-08-19 | SARS-CoV-2 RBD-specific monoclonal antibodies and their applications |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010839860.XA CN111925442B (en) | 2020-08-19 | 2020-08-19 | New coronavirus RBD specific monoclonal antibody and application |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202210501598.7A Division CN115340600B (en) | 2020-08-19 | 2020-08-19 | SARS-CoV-2 RBD-specific monoclonal antibodies and their applications |
| CN202210906292.XA Division CN115925900B (en) | 2020-08-19 | 2020-08-19 | Novel coronavirus RBD specific monoclonal antibody and application |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111925442A CN111925442A (en) | 2020-11-13 |
| CN111925442B true CN111925442B (en) | 2022-10-11 |
Family
ID=73305615
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010839860.XA Active CN111925442B (en) | 2020-08-19 | 2020-08-19 | New coronavirus RBD specific monoclonal antibody and application |
| CN202210906292.XA Active CN115925900B (en) | 2020-08-19 | 2020-08-19 | Novel coronavirus RBD specific monoclonal antibody and application |
| CN202210501598.7A Active CN115340600B (en) | 2020-08-19 | 2020-08-19 | SARS-CoV-2 RBD-specific monoclonal antibodies and their applications |
Family Applications After (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202210906292.XA Active CN115925900B (en) | 2020-08-19 | 2020-08-19 | Novel coronavirus RBD specific monoclonal antibody and application |
| CN202210501598.7A Active CN115340600B (en) | 2020-08-19 | 2020-08-19 | SARS-CoV-2 RBD-specific monoclonal antibodies and their applications |
Country Status (1)
| Country | Link |
|---|---|
| CN (3) | CN111925442B (en) |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP7116256B1 (en) | 2020-04-02 | 2022-08-09 | リジェネロン・ファーマシューティカルズ・インコーポレイテッド | Anti-SARS-COV-2-Spike Glycoprotein Antibodies and Antigen-Binding Fragments |
| KR20230019166A (en) | 2020-06-03 | 2023-02-07 | 리제너론 파아마슈티컬스, 인크. | Methods for treating or preventing SARS-CoV-2 infection and COVID-19 using anti-SARS-CoV-2 spike glycoprotein antibodies |
| CN112442120A (en) * | 2020-11-25 | 2021-03-05 | 苏州大学 | Neutralizing antibody against SARS-COV-2 of severe acute respiratory syndrome type II coronavirus |
| WO2022112392A1 (en) * | 2020-11-26 | 2022-06-02 | Memo Therapeutics Ag | Anti-sars-cov-2 antibody molecules |
| BR112024000744A2 (en) | 2021-07-14 | 2024-04-30 | Regeneron Pharma | ANTI-SARS-COV-2 SPIKE GLYCOPROTEIN ANTIBODIES AND ANTIGEN-BINDING FRAGMENTS |
| CN116178527B (en) * | 2021-11-26 | 2025-12-09 | 深圳先进技术研究院 | Anti SARS-COV-2 fully human monoclonal antibody 6G18 and its preparation method and application |
| TW202337497A (en) | 2022-02-18 | 2023-10-01 | 中國大陸商重慶明道浩悅生物科技有限公司 | Intranasal formulations and anti-sars-cov-2-spike protein antibodies |
| CN118420752A (en) * | 2022-10-08 | 2024-08-02 | 郴州市第一人民医院 | A fully human neutralizing antibody against the novel coronavirus SARS-CoV-2 and its application |
| CN119331085B (en) * | 2024-10-16 | 2025-03-14 | 中国科学院合肥物质科学研究院 | Sabei coronavirus broad-spectrum neutralizing antibodies and their applications |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111303279A (en) * | 2020-03-17 | 2020-06-19 | 中国医学科学院病原生物学研究所 | A kind of single domain antibody against novel coronavirus and its application |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| PL1915398T3 (en) * | 2005-07-18 | 2016-10-31 | Human anti-b7rp1 neutralizing antibodies | |
| CN101517068B (en) * | 2005-09-07 | 2017-02-08 | 安进弗里蒙特公司 | Human monoclonal antibodies to activin receptor-like kinase-1 |
| PH12022550138A1 (en) * | 2013-03-13 | 2023-03-06 | Amgen Inc | Proteins specific for baff and b7rp1 and uses thereof |
| CA3119836A1 (en) * | 2018-11-15 | 2020-05-22 | Bluewillow Biologics, Inc. | Nanoemulsion compositions having enhanced permeability |
| CN111153991A (en) * | 2020-02-26 | 2020-05-15 | 北京博奥森生物技术有限公司 | Human SARS-CoV-2 monoclonal antibody and its preparation method and use |
| CN111303280B (en) * | 2020-03-22 | 2022-01-07 | 中国人民解放军军事科学院军事医学研究院 | High-neutralization-activity anti-SARS-CoV-2 fully human monoclonal antibody and application |
| RU2723008C9 (en) * | 2020-05-19 | 2021-02-09 | федеральное государственное бюджетное учреждение «Национальный исследовательский центр эпидемиологии и микробиологии имени почетного академика Н.Ф. Гамалеи» Министерства здравоохранения Российской Федерации | Method for producing chinese hamster ovary cell strain, producer of sars-cov-2 virus recombinant rbd protein, chinese hamster ovary cell strain, producer of recombinant rbd protein of sars-cov-2 virus, method of producing recombinant rbd protein of sars-cov-2 virus, a test system for enzyme-linked immunosorbent assay of human blood serum or plasma and its use |
| CN111518202B (en) * | 2020-05-27 | 2021-10-19 | 江苏省疾病预防控制中心(江苏省公共卫生研究院) | Novel coronavirus antibodies and ELISA test kits for novel coronavirus antibodies |
| CN111518204B (en) * | 2020-05-27 | 2021-09-14 | 江苏省疾病预防控制中心(江苏省公共卫生研究院) | Antibodies against novel coronaviruses for immunodetection |
-
2020
- 2020-08-19 CN CN202010839860.XA patent/CN111925442B/en active Active
- 2020-08-19 CN CN202210906292.XA patent/CN115925900B/en active Active
- 2020-08-19 CN CN202210501598.7A patent/CN115340600B/en active Active
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111303279A (en) * | 2020-03-17 | 2020-06-19 | 中国医学科学院病原生物学研究所 | A kind of single domain antibody against novel coronavirus and its application |
Also Published As
| Publication number | Publication date |
|---|---|
| CN115340600B (en) | 2025-01-24 |
| CN115925900A (en) | 2023-04-07 |
| CN115925900B (en) | 2024-06-25 |
| CN111925442A (en) | 2020-11-13 |
| CN115340600A (en) | 2022-11-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN111909260B (en) | Novel coronavirus RBD-specific monoclonal antibodies and applications | |
| CN111925441B (en) | Novel coronavirus RBD-specific monoclonal antibodies and applications | |
| CN111909263B (en) | New coronavirus RBD specific monoclonal antibody and application | |
| CN111925443B (en) | Novel coronavirus RBD-specific monoclonal antibodies and applications | |
| CN111909261B (en) | New coronavirus RBD specific monoclonal antibody and application | |
| CN111925444B (en) | Novel coronavirus RBD-specific monoclonal antibodies and applications | |
| CN111925442B (en) | New coronavirus RBD specific monoclonal antibody and application | |
| CN111925440B (en) | New coronavirus RBD specific monoclonal antibody and application | |
| CN111909262B (en) | New coronavirus RBD specific monoclonal antibody and application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |