CN111848600B - 2,4, 4-trisubstituted dihydrooxazole derivative and application thereof - Google Patents
2,4, 4-trisubstituted dihydrooxazole derivative and application thereof Download PDFInfo
- Publication number
- CN111848600B CN111848600B CN202010749278.4A CN202010749278A CN111848600B CN 111848600 B CN111848600 B CN 111848600B CN 202010749278 A CN202010749278 A CN 202010749278A CN 111848600 B CN111848600 B CN 111848600B
- Authority
- CN
- China
- Prior art keywords
- methyl
- dihydrooxazole
- triazol
- phenyl
- biphenyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- -1 2,4, 4-trisubstituted dihydrooxazole Chemical class 0.000 title claims abstract description 26
- 239000003814 drug Substances 0.000 claims abstract description 32
- 150000003839 salts Chemical class 0.000 claims abstract description 23
- 206010017533 Fungal infection Diseases 0.000 claims abstract description 12
- 208000031888 Mycoses Diseases 0.000 claims abstract description 12
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 3
- 150000001875 compounds Chemical class 0.000 claims description 65
- 241001480037 Microsporum Species 0.000 claims description 18
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 13
- 241000222122 Candida albicans Species 0.000 claims description 12
- 229940095731 candida albicans Drugs 0.000 claims description 10
- 238000006467 substitution reaction Methods 0.000 claims description 9
- 125000001072 heteroaryl group Chemical group 0.000 claims description 8
- 125000000623 heterocyclic group Chemical group 0.000 claims description 8
- 125000003118 aryl group Chemical group 0.000 claims description 7
- 239000008194 pharmaceutical composition Substances 0.000 claims description 7
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 6
- 229910052736 halogen Inorganic materials 0.000 claims description 6
- 150000002367 halogens Chemical group 0.000 claims description 6
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 claims description 5
- 241001225321 Aspergillus fumigatus Species 0.000 claims description 5
- 125000001624 naphthyl group Chemical group 0.000 claims description 5
- 229910052760 oxygen Inorganic materials 0.000 claims description 5
- 229910052717 sulfur Inorganic materials 0.000 claims description 5
- FCMUIKBWXRDRMN-UHFFFAOYSA-N 4-(2,4-difluorophenyl)-2-[4-(3-fluorophenyl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CC(=CC=C1)F)C1=C(C=C(C=C1)F)F FCMUIKBWXRDRMN-UHFFFAOYSA-N 0.000 claims description 4
- 241000221204 Cryptococcus neoformans Species 0.000 claims description 4
- 241000223238 Trichophyton Species 0.000 claims description 4
- 229940091771 aspergillus fumigatus Drugs 0.000 claims description 4
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 claims description 4
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 4
- 125000002541 furyl group Chemical group 0.000 claims description 4
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 4
- 125000001041 indolyl group Chemical group 0.000 claims description 4
- 125000000842 isoxazolyl group Chemical group 0.000 claims description 4
- 229910052757 nitrogen Inorganic materials 0.000 claims description 4
- 125000002971 oxazolyl group Chemical group 0.000 claims description 4
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 4
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 4
- 125000000168 pyrrolyl group Chemical group 0.000 claims description 4
- 125000001544 thienyl group Chemical group 0.000 claims description 4
- GERWUIDIGDBAGO-UHFFFAOYSA-N 1-[4-[4-[4-phenyl-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazol-2-yl]phenyl]piperazin-1-yl]ethanone Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)N1CCN(CC1)C(C)=O)C1=CC=CC=C1 GERWUIDIGDBAGO-UHFFFAOYSA-N 0.000 claims description 3
- CSAMVZSMGPDVPI-UHFFFAOYSA-N 2-(6-bromo-1-benzothiophen-2-yl)-4-(2,4-dichlorophenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)Br)C2=C(C=C(C=C2)Cl)Cl CSAMVZSMGPDVPI-UHFFFAOYSA-N 0.000 claims description 3
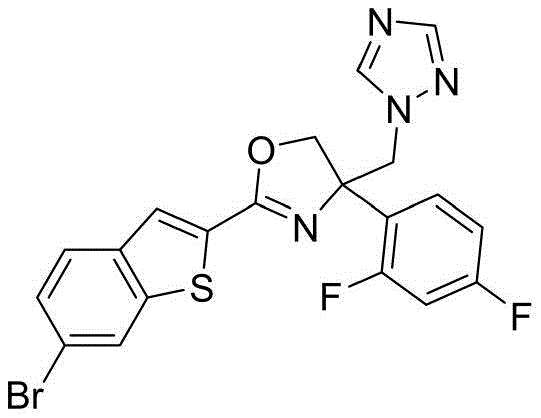
- NSHUZONZAYANBP-UHFFFAOYSA-N 2-(6-bromo-1-benzothiophen-2-yl)-4-(2,4-difluorophenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)Br)C2=C(C=C(C=C2)F)F NSHUZONZAYANBP-UHFFFAOYSA-N 0.000 claims description 3
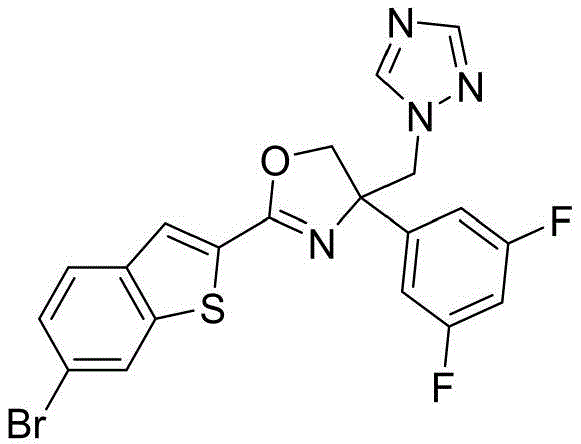
- OQJHOAOJXPNANZ-UHFFFAOYSA-N 2-(6-bromo-1-benzothiophen-2-yl)-4-(3,5-difluorophenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)Br)C2=CC(=CC(=C2)F)F OQJHOAOJXPNANZ-UHFFFAOYSA-N 0.000 claims description 3
- RDYVQGAFJVBGSH-UHFFFAOYSA-N 2-(6-bromo-1-benzothiophen-2-yl)-4-(4-chlorophenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)Br)C2=CC=C(C=C2)Cl RDYVQGAFJVBGSH-UHFFFAOYSA-N 0.000 claims description 3
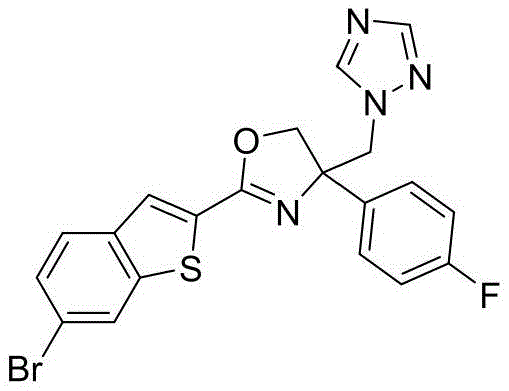
- XQGUANABEGFDSR-UHFFFAOYSA-N 2-(6-bromo-1-benzothiophen-2-yl)-4-(4-fluorophenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)Br)C2=CC=C(C=C2)F XQGUANABEGFDSR-UHFFFAOYSA-N 0.000 claims description 3
- KYZKMSXMEHOMGN-UHFFFAOYSA-N 2-(6-chloro-1-benzothiophen-2-yl)-4-(2,4-dichlorophenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)Cl)C2=C(C=C(C=C2)Cl)Cl KYZKMSXMEHOMGN-UHFFFAOYSA-N 0.000 claims description 3
- DYKQCQVMNLYFGC-UHFFFAOYSA-N 2-(6-chloro-1-benzothiophen-2-yl)-4-(2,4-difluorophenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)Cl)C2=C(C=C(C=C2)F)F DYKQCQVMNLYFGC-UHFFFAOYSA-N 0.000 claims description 3
- KQFPZMUJFKULRN-UHFFFAOYSA-N 2-(6-chloro-1-benzothiophen-2-yl)-4-(3,5-difluorophenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)Cl)C2=CC(=CC(=C2)F)F KQFPZMUJFKULRN-UHFFFAOYSA-N 0.000 claims description 3
- HDRXGGWSEWGELG-UHFFFAOYSA-N 2-(6-chloro-1-benzothiophen-2-yl)-4-(4-chlorophenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)Cl)C2=CC=C(C=C2)Cl HDRXGGWSEWGELG-UHFFFAOYSA-N 0.000 claims description 3
- KTOAMCQQWXLGKV-UHFFFAOYSA-N 2-(6-chloro-1-benzothiophen-2-yl)-4-(4-fluorophenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)Cl)C2=CC=C(C=C2)F KTOAMCQQWXLGKV-UHFFFAOYSA-N 0.000 claims description 3
- SRKWWDJZMJKUBV-UHFFFAOYSA-N 2-(6-chloro-1-benzothiophen-2-yl)-4-(4-fluorophenyl)-4-(tetrazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=NN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)Cl)C2=CC=C(C=C2)F SRKWWDJZMJKUBV-UHFFFAOYSA-N 0.000 claims description 3
- JIBRROSJWTWYEC-UHFFFAOYSA-N 2-(6-fluoro-1-benzothiophen-2-yl)-4-(4-fluorophenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)F)C2=CC=C(C=C2)F JIBRROSJWTWYEC-UHFFFAOYSA-N 0.000 claims description 3
- KDGLBQTTXTVHQZ-UHFFFAOYSA-N 2-phenyl-5-[4-[4-phenyl-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazol-2-yl]phenyl]-1,3,4-oxadiazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C=1OC(=NN1)C1=CC=CC=C1)C1=CC=CC=C1 KDGLBQTTXTVHQZ-UHFFFAOYSA-N 0.000 claims description 3
- NMGKXVFWEOQFJT-UHFFFAOYSA-N 4-(2,4-dichlorophenyl)-2-(6-fluoro-1-benzothiophen-2-yl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)F)C2=C(C=C(C=C2)Cl)Cl NMGKXVFWEOQFJT-UHFFFAOYSA-N 0.000 claims description 3
- PXGSQTSUZKLFDR-UHFFFAOYSA-N 4-(2,4-dichlorophenyl)-2-[4-(2-fluorophenyl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=C(C=CC=C1)F)C1=C(C=C(C=C1)Cl)Cl PXGSQTSUZKLFDR-UHFFFAOYSA-N 0.000 claims description 3
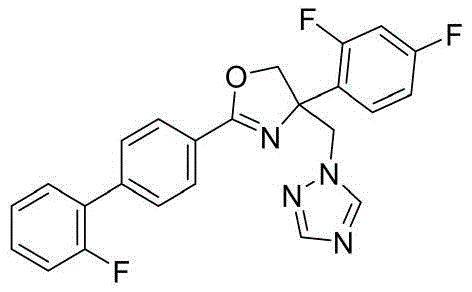
- GCYKPNFVRKLBMD-UHFFFAOYSA-N 4-(2,4-difluorophenyl)-2-[4-(2-fluorophenyl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=C(C=CC=C1)F)C1=C(C=C(C=C1)F)F GCYKPNFVRKLBMD-UHFFFAOYSA-N 0.000 claims description 3
- DYJCMBOEWYSYCV-UHFFFAOYSA-N 4-(3,5-difluorophenyl)-2-(6-fluoro-1-benzothiophen-2-yl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)F)C2=CC(=CC(=C2)F)F DYJCMBOEWYSYCV-UHFFFAOYSA-N 0.000 claims description 3
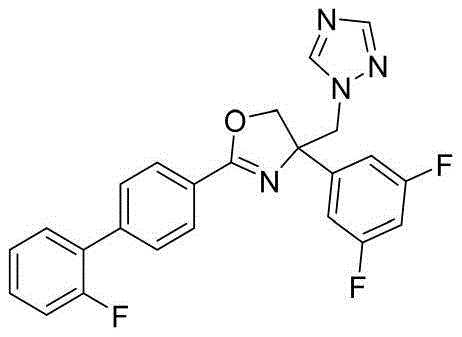
- RFBQORKKYIOIDP-UHFFFAOYSA-N 4-(3,5-difluorophenyl)-2-[4-(2-fluorophenyl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=C(C=CC=C1)F)C1=CC(=CC(=C1)F)F RFBQORKKYIOIDP-UHFFFAOYSA-N 0.000 claims description 3
- JREZIFIQNLDOJK-UHFFFAOYSA-N 4-(3,5-difluorophenyl)-2-[4-(3-fluorophenyl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CC(=CC=C1)F)C1=CC(=CC(=C1)F)F JREZIFIQNLDOJK-UHFFFAOYSA-N 0.000 claims description 3
- LFLUGLJGCZWYDE-UHFFFAOYSA-N 4-(4-chlorophenyl)-2-(4-thiophen-3-ylphenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CSC=C1)C1=CC=C(C=C1)Cl LFLUGLJGCZWYDE-UHFFFAOYSA-N 0.000 claims description 3
- HMNORGYSISZKOU-UHFFFAOYSA-N 4-(4-chlorophenyl)-2-[4-(2-fluorophenyl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=C(C=CC=C1)F)C1=CC=C(C=C1)Cl HMNORGYSISZKOU-UHFFFAOYSA-N 0.000 claims description 3
- NQTIQHNZMXZUFX-UHFFFAOYSA-N 4-(4-chlorophenyl)-2-[4-(3-fluorophenyl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CC(=CC=C1)F)C1=CC=C(C=C1)Cl NQTIQHNZMXZUFX-UHFFFAOYSA-N 0.000 claims description 3
- LIVLINYHTKMLRN-UHFFFAOYSA-N 4-(4-chlorophenyl)-2-[4-(furan-3-yl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=COC=C1)C1=CC=C(C=C1)Cl LIVLINYHTKMLRN-UHFFFAOYSA-N 0.000 claims description 3
- VLNHLPDQPVIMBC-UHFFFAOYSA-N 4-(4-fluorophenyl)-2-(4-thiophen-3-ylphenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CSC=C1)C1=CC=C(C=C1)F VLNHLPDQPVIMBC-UHFFFAOYSA-N 0.000 claims description 3
- WTXFRMCKLJLVDP-UHFFFAOYSA-N 4-(4-fluorophenyl)-2-[4-(2-fluorophenyl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=C(C=CC=C1)F)C1=CC=C(C=C1)F WTXFRMCKLJLVDP-UHFFFAOYSA-N 0.000 claims description 3
- GRRCEUHIAGYRDK-UHFFFAOYSA-N 4-(4-fluorophenyl)-2-[4-(3-fluorophenyl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CC(=CC=C1)F)C1=CC=C(C=C1)F GRRCEUHIAGYRDK-UHFFFAOYSA-N 0.000 claims description 3
- MXLNMGLQUWCDGY-UHFFFAOYSA-N 4-(4-fluorophenyl)-2-[4-(furan-3-yl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=COC=C1)C1=CC=C(C=C1)F MXLNMGLQUWCDGY-UHFFFAOYSA-N 0.000 claims description 3
- YZECXNPQWNHMHU-UHFFFAOYSA-N N-[4-[4-phenyl-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazol-2-yl]phenyl]benzamide Chemical compound C(C1=CC=CC=C1)(=O)NC1=CC=C(C=C1)C=1OCC(N1)(C1=CC=CC=C1)CN1N=CN=C1 YZECXNPQWNHMHU-UHFFFAOYSA-N 0.000 claims description 3
- 241000223229 Trichophyton rubrum Species 0.000 claims description 3
- CIUQDSCDWFSTQR-UHFFFAOYSA-N [C]1=CC=CC=C1 Chemical compound [C]1=CC=CC=C1 CIUQDSCDWFSTQR-UHFFFAOYSA-N 0.000 claims description 3
- 239000004480 active ingredient Substances 0.000 claims description 3
- 125000005257 alkyl acyl group Chemical group 0.000 claims description 3
- 125000005605 benzo group Chemical group 0.000 claims description 3
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 claims description 3
- 125000005842 heteroatom Chemical group 0.000 claims description 3
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 claims description 3
- LUMBSCWASVCSFB-UHFFFAOYSA-N 4-(2,4-dichlorophenyl)-2-(4-thiophen-3-ylphenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CSC=C1)C1=C(C=C(C=C1)Cl)Cl LUMBSCWASVCSFB-UHFFFAOYSA-N 0.000 claims description 2
- PCGOFYSHHFSECI-UHFFFAOYSA-N 4-(2,4-dichlorophenyl)-2-[4-(3-fluorophenyl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CC(=CC=C1)F)C1=C(C=C(C=C1)Cl)Cl PCGOFYSHHFSECI-UHFFFAOYSA-N 0.000 claims description 2
- CGTVOUSDNWQTPD-UHFFFAOYSA-N 4-(2,4-dichlorophenyl)-2-[4-(furan-3-yl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=COC=C1)C1=C(C=C(C=C1)Cl)Cl CGTVOUSDNWQTPD-UHFFFAOYSA-N 0.000 claims description 2
- CJXRFXYYMYXHLL-UHFFFAOYSA-N 4-(2,4-difluorophenyl)-2-(4-thiophen-3-ylphenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CSC=C1)C1=C(C=C(C=C1)F)F CJXRFXYYMYXHLL-UHFFFAOYSA-N 0.000 claims description 2
- IMXXINRINZRAFV-UHFFFAOYSA-N 4-(2,4-difluorophenyl)-2-[4-(furan-3-yl)phenyl]-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=COC=C1)C1=C(C=C(C=C1)F)F IMXXINRINZRAFV-UHFFFAOYSA-N 0.000 claims description 2
- 241000893966 Trichophyton verrucosum Species 0.000 claims description 2
- 241000222126 [Candida] glabrata Species 0.000 claims description 2
- 208000032343 candida glabrata infection Diseases 0.000 claims description 2
- 125000000319 biphenyl-4-yl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])=C1C1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 claims 9
- 241000186063 Arthrobacter Species 0.000 claims 5
- 241000223230 Trichosporon Species 0.000 claims 5
- 241000893451 Arthroderma Species 0.000 claims 4
- 241000219071 Malvaceae Species 0.000 claims 4
- 241001045770 Trichophyton mentagrophytes Species 0.000 claims 3
- 241000235389 Absidia Species 0.000 claims 2
- 241000228197 Aspergillus flavus Species 0.000 claims 2
- 241000228245 Aspergillus niger Species 0.000 claims 2
- 241000079253 Byssochlamys spectabilis Species 0.000 claims 2
- 241000222120 Candida <Saccharomycetales> Species 0.000 claims 2
- 241001480036 Epidermophyton floccosum Species 0.000 claims 2
- 241000248325 Exophiala dermatitidis Species 0.000 claims 2
- 241000427940 Fusarium solani Species 0.000 claims 2
- 244000168141 Geotrichum candidum Species 0.000 claims 2
- 235000017388 Geotrichum candidum Nutrition 0.000 claims 2
- 244000286779 Hansenula anomala Species 0.000 claims 2
- 235000014683 Hansenula anomala Nutrition 0.000 claims 2
- 241000228404 Histoplasma capsulatum Species 0.000 claims 2
- 241001057811 Paracoccus <mealybug> Species 0.000 claims 2
- 241000235648 Pichia Species 0.000 claims 2
- 241000233872 Pneumocystis carinii Species 0.000 claims 2
- 240000005384 Rhizopus oryzae Species 0.000 claims 2
- 235000013752 Rhizopus oryzae Nutrition 0.000 claims 2
- 241000223254 Rhodotorula mucilaginosa Species 0.000 claims 2
- 241000222481 Schizophyllum commune Species 0.000 claims 2
- 241001523006 Talaromyces marneffei Species 0.000 claims 2
- 125000004618 benzofuryl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 claims 2
- 238000004519 manufacturing process Methods 0.000 claims 2
- 125000004215 2,4-difluorophenyl group Chemical group [H]C1=C([H])C(*)=C(F)C([H])=C1F 0.000 claims 1
- 125000001622 2-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C(*)C([H])=C([H])C2=C1[H] 0.000 claims 1
- 241000228405 Blastomyces dermatitidis Species 0.000 claims 1
- 241000282465 Canis Species 0.000 claims 1
- 241000223233 Cutaneotrichosporon cutaneum Species 0.000 claims 1
- 241001634927 Cutaneotrichosporon mucoides Species 0.000 claims 1
- 241000199026 Gyptis <bristle worm> Species 0.000 claims 1
- 241001444195 Madurella Species 0.000 claims 1
- 241000555688 Malassezia furfur Species 0.000 claims 1
- 241000779599 Malpighia Species 0.000 claims 1
- 241000031003 Monascus ruber Species 0.000 claims 1
- 241000306281 Mucor ambiguus Species 0.000 claims 1
- 241000186359 Mycobacterium Species 0.000 claims 1
- 241000893974 Nannizzia fulva Species 0.000 claims 1
- 241000893976 Nannizzia gypsea Species 0.000 claims 1
- 241001226034 Nectria <echinoderm> Species 0.000 claims 1
- 241000222640 Polyporus Species 0.000 claims 1
- 241000589516 Pseudomonas Species 0.000 claims 1
- 241000094595 Sarcoporia Species 0.000 claims 1
- 241000132889 Scedosporium Species 0.000 claims 1
- 241001533598 Septoria Species 0.000 claims 1
- 241001149963 Sporothrix schenckii Species 0.000 claims 1
- 241001480050 Trichophyton violaceum Species 0.000 claims 1
- 241001634961 Trichosporon asahii Species 0.000 claims 1
- 241000601794 Trichothecium Species 0.000 claims 1
- 241001530097 Verbascum Species 0.000 claims 1
- 241000700605 Viruses Species 0.000 claims 1
- 241000235013 Yarrowia Species 0.000 claims 1
- 241000645784 [Candida] auris Species 0.000 claims 1
- 125000004244 benzofuran-2-yl group Chemical group [H]C1=C(*)OC2=C([H])C([H])=C([H])C([H])=C12 0.000 claims 1
- 238000010276 construction Methods 0.000 claims 1
- 244000053095 fungal pathogen Species 0.000 claims 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims 1
- 125000000437 thiazol-2-yl group Chemical group [H]C1=C([H])N=C(*)S1 0.000 claims 1
- 229940055035 trichophyton verrucosum Drugs 0.000 claims 1
- 229940079593 drug Drugs 0.000 abstract description 29
- 238000002360 preparation method Methods 0.000 abstract description 17
- 239000000651 prodrug Substances 0.000 abstract description 13
- 229940002612 prodrug Drugs 0.000 abstract description 13
- 239000012453 solvate Substances 0.000 abstract description 12
- 150000004677 hydrates Chemical class 0.000 abstract description 11
- 230000015572 biosynthetic process Effects 0.000 abstract description 2
- 201000010099 disease Diseases 0.000 abstract description 2
- 238000003786 synthesis reaction Methods 0.000 abstract description 2
- 239000000543 intermediate Substances 0.000 description 47
- 239000011734 sodium Substances 0.000 description 40
- 238000005160 1H NMR spectroscopy Methods 0.000 description 37
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 33
- 238000006243 chemical reaction Methods 0.000 description 32
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 21
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 20
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 19
- 230000000843 anti-fungal effect Effects 0.000 description 16
- 238000000034 method Methods 0.000 description 16
- 239000000243 solution Substances 0.000 description 12
- 229940121375 antifungal agent Drugs 0.000 description 11
- 239000002274 desiccant Substances 0.000 description 11
- 239000012044 organic layer Substances 0.000 description 11
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 10
- 238000004440 column chromatography Methods 0.000 description 10
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 9
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 9
- 239000000203 mixture Substances 0.000 description 9
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 7
- 239000012980 RPMI-1640 medium Substances 0.000 description 7
- 230000001580 bacterial effect Effects 0.000 description 7
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 6
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 6
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 239000002775 capsule Substances 0.000 description 6
- 239000000463 material Substances 0.000 description 6
- 239000003960 organic solvent Substances 0.000 description 6
- 239000002994 raw material Substances 0.000 description 6
- 239000000725 suspension Substances 0.000 description 6
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 description 5
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 5
- 125000000217 alkyl group Chemical group 0.000 description 5
- 239000002552 dosage form Substances 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 125000004435 hydrogen atom Chemical class [H]* 0.000 description 5
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 4
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- 239000000460 chlorine Substances 0.000 description 4
- 239000012153 distilled water Substances 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- 230000002538 fungal effect Effects 0.000 description 4
- 239000007924 injection Substances 0.000 description 4
- 238000002347 injection Methods 0.000 description 4
- 239000002609 medium Substances 0.000 description 4
- 239000006187 pill Substances 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- 239000000829 suppository Substances 0.000 description 4
- 239000003826 tablet Substances 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 4
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- 101100391174 Dictyostelium discoideum forC gene Proteins 0.000 description 3
- 241000233866 Fungi Species 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 239000000443 aerosol Substances 0.000 description 3
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 description 3
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 239000003937 drug carrier Substances 0.000 description 3
- 239000010408 film Substances 0.000 description 3
- 229960004884 fluconazole Drugs 0.000 description 3
- RFHAOTPXVQNOHP-UHFFFAOYSA-N fluconazole Chemical compound C1=NC=NN1CC(C=1C(=CC(F)=CC=1)F)(O)CN1C=NC=N1 RFHAOTPXVQNOHP-UHFFFAOYSA-N 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- 229910052739 hydrogen Inorganic materials 0.000 description 3
- 239000001257 hydrogen Substances 0.000 description 3
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 3
- 239000000865 liniment Substances 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 231100000053 low toxicity Toxicity 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 239000002674 ointment Substances 0.000 description 3
- 125000000962 organic group Chemical group 0.000 description 3
- 239000013641 positive control Substances 0.000 description 3
- 239000007858 starting material Substances 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 229960004740 voriconazole Drugs 0.000 description 3
- BCEHBSKCWLPMDN-MGPLVRAMSA-N voriconazole Chemical compound C1([C@H](C)[C@](O)(CN2N=CN=C2)C=2C(=CC(F)=CC=2)F)=NC=NC=C1F BCEHBSKCWLPMDN-MGPLVRAMSA-N 0.000 description 3
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 description 2
- 125000004738 (C1-C6) alkyl sulfinyl group Chemical group 0.000 description 2
- HEWZVZIVELJPQZ-UHFFFAOYSA-N 2,2-dimethoxypropane Chemical compound COC(C)(C)OC HEWZVZIVELJPQZ-UHFFFAOYSA-N 0.000 description 2
- NVHGSCJXDRTMBB-UHFFFAOYSA-N 2,4-diphenyl-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=CC=C1)C1=CC=CC=C1 NVHGSCJXDRTMBB-UHFFFAOYSA-N 0.000 description 2
- SXJJYMGTZVTKBX-UHFFFAOYSA-N 2-(1,3-benzothiazol-2-yl)-4-phenyl-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound C1C(N=C(O1)C2=NC3=CC=CC=C3S2)(CN4C=NC=N4)C5=CC=CC=C5 SXJJYMGTZVTKBX-UHFFFAOYSA-N 0.000 description 2
- VHVPQPYKVGDNFY-DFMJLFEVSA-N 2-[(2r)-butan-2-yl]-4-[4-[4-[4-[[(2r,4s)-2-(2,4-dichlorophenyl)-2-(1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]piperazin-1-yl]phenyl]-1,2,4-triazol-3-one Chemical compound O=C1N([C@H](C)CC)N=CN1C1=CC=C(N2CCN(CC2)C=2C=CC(OC[C@@H]3O[C@](CN4N=CN=C4)(OC3)C=3C(=CC(Cl)=CC=3)Cl)=CC=2)C=C1 VHVPQPYKVGDNFY-DFMJLFEVSA-N 0.000 description 2
- NHZPQWCZMBAERW-UHFFFAOYSA-N 4-(2,4-difluorophenyl)-2-(6-fluoro-1-benzothiophen-2-yl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)F)C2=C(C=C(C=C2)F)F NHZPQWCZMBAERW-UHFFFAOYSA-N 0.000 description 2
- OFOYOKWGTOFDDC-UHFFFAOYSA-N 4-(4-chlorophenyl)-2-(6-fluoro-1-benzothiophen-2-yl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=C(C=C2)F)C2=CC=C(C=C2)Cl OFOYOKWGTOFDDC-UHFFFAOYSA-N 0.000 description 2
- NLAFMOSZPPMYAX-UHFFFAOYSA-N 4-(imidazol-1-ylmethyl)-2-(4-phenylphenyl)-4-propan-2-yl-5H-1,3-oxazole Chemical compound N1(C=NC=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CC=CC=C1)C(C)C NLAFMOSZPPMYAX-UHFFFAOYSA-N 0.000 description 2
- UXJXWSMWDWIZMU-UHFFFAOYSA-N 4-cyclopropyl-4-(imidazol-1-ylmethyl)-2-(4-phenylphenyl)-5H-1,3-oxazole Chemical compound N1(C=NC=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CC=CC=C1)C1CC1 UXJXWSMWDWIZMU-UHFFFAOYSA-N 0.000 description 2
- XDNJVFGQQWLJJB-UHFFFAOYSA-N 4-phenyl-2-(4-phenylphenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CC=CC=C1)C1=CC=CC=C1 XDNJVFGQQWLJJB-UHFFFAOYSA-N 0.000 description 2
- SLWIFZYRJHUDME-UHFFFAOYSA-N 5-[difluoro(phenyl)methyl]-4-(imidazol-1-ylmethyl)-2-(4-phenylphenyl)-4,5-dihydro-1,3-oxazole Chemical compound N1(C=NC=C1)CC1N=C(OC1C(C1=CC=CC=C1)(F)F)C1=CC=C(C=C1)C1=CC=CC=C1 SLWIFZYRJHUDME-UHFFFAOYSA-N 0.000 description 2
- NRZSZHADDLIIQD-UHFFFAOYSA-N 5-[fluoro(phenyl)methyl]-4-(imidazol-1-ylmethyl)-2-(4-phenylphenyl)-4,5-dihydro-1,3-oxazole Chemical compound N1(C=NC=C1)CC1N=C(OC1C(C1=CC=CC=C1)F)C1=CC=C(C=C1)C1=CC=CC=C1 NRZSZHADDLIIQD-UHFFFAOYSA-N 0.000 description 2
- TWLMPPSSAQUMSE-UHFFFAOYSA-N 5-phenyl-3-[4-phenyl-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazol-2-yl]-1,2-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=NOC(=C1)C1=CC=CC=C1)C1=CC=CC=C1 TWLMPPSSAQUMSE-UHFFFAOYSA-N 0.000 description 2
- 101710134784 Agnoprotein Proteins 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 241000894006 Bacteria Species 0.000 description 2
- FGUUSXIOTUKUDN-IBGZPJMESA-N C1(=CC=CC=C1)N1C2=C(NC([C@H](C1)NC=1OC(=NN=1)C1=CC=CC=C1)=O)C=CC=C2 Chemical compound C1(=CC=CC=C1)N1C2=C(NC([C@H](C1)NC=1OC(=NN=1)C1=CC=CC=C1)=O)C=CC=C2 FGUUSXIOTUKUDN-IBGZPJMESA-N 0.000 description 2
- 125000001313 C5-C10 heteroaryl group Chemical group 0.000 description 2
- 241000222178 Candida tropicalis Species 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 201000007336 Cryptococcosis Diseases 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 102100021695 Lanosterol 14-alpha demethylase Human genes 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 2
- 229960000583 acetic acid Drugs 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 125000002947 alkylene group Chemical group 0.000 description 2
- 125000005530 alkylenedioxy group Chemical group 0.000 description 2
- 150000001408 amides Chemical class 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- 125000004122 cyclic group Chemical group 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- 239000006196 drop Substances 0.000 description 2
- 230000008686 ergosterol biosynthesis Effects 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 125000002883 imidazolyl group Chemical group 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 230000002401 inhibitory effect Effects 0.000 description 2
- 229960004130 itraconazole Drugs 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- 239000012982 microporous membrane Substances 0.000 description 2
- 229960001589 posaconazole Drugs 0.000 description 2
- RAGOYPUPXAKGKH-XAKZXMRKSA-N posaconazole Chemical compound O=C1N([C@H]([C@H](C)O)CC)N=CN1C1=CC=C(N2CCN(CC2)C=2C=CC(OC[C@H]3C[C@@](CN4N=CN=C4)(OC3)C=3C(=CC(F)=CC=3)F)=CC=2)C=C1 RAGOYPUPXAKGKH-XAKZXMRKSA-N 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- 125000004076 pyridyl group Chemical group 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 229920006395 saturated elastomer Polymers 0.000 description 2
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 2
- 238000010998 test method Methods 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- 238000005303 weighing Methods 0.000 description 2
- XMAYWYJOQHXEEK-OZXSUGGESA-N (2R,4S)-ketoconazole Chemical compound C1CN(C(=O)C)CCN1C(C=C1)=CC=C1OC[C@@H]1O[C@@](CN2C=NC=C2)(C=2C(=CC(Cl)=CC=2)Cl)OC1 XMAYWYJOQHXEEK-OZXSUGGESA-N 0.000 description 1
- 125000006727 (C1-C6) alkenyl group Chemical group 0.000 description 1
- 125000006700 (C1-C6) alkylthio group Chemical group 0.000 description 1
- 125000006728 (C1-C6) alkynyl group Chemical group 0.000 description 1
- ZYHTUICRXAAALG-UHFFFAOYSA-N 2-(1-benzofuran-2-yl)-4-phenyl-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C=1OC2=C(C1)C=CC=C2)C2=CC=CC=C2 ZYHTUICRXAAALG-UHFFFAOYSA-N 0.000 description 1
- ZAIIGYZYRVYMNK-UHFFFAOYSA-N 2-(1-benzothiophen-2-yl)-4-phenyl-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=C(S1)C=CC=C2)C2=CC=CC=C2 ZAIIGYZYRVYMNK-UHFFFAOYSA-N 0.000 description 1
- VRYCTTIYXYKRDN-UHFFFAOYSA-N 2-[4-(furan-3-yl)phenyl]-4-phenyl-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=COC=C1)C1=CC=CC=C1 VRYCTTIYXYKRDN-UHFFFAOYSA-N 0.000 description 1
- REBNFRJQCRUNEI-UHFFFAOYSA-N 2-naphthalen-2-yl-4-phenyl-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC2=CC=CC=C2C=C1)C1=CC=CC=C1 REBNFRJQCRUNEI-UHFFFAOYSA-N 0.000 description 1
- GNFTZDOKVXKIBK-UHFFFAOYSA-N 3-(2-methoxyethoxy)benzohydrazide Chemical compound COCCOC1=CC=CC(C(=O)NN)=C1 GNFTZDOKVXKIBK-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- OGLAHQNDFOEYAO-UHFFFAOYSA-N 4-(1,3-dihydrotetrazol-2-ylmethyl)-4-phenyl-2-(4-phenylphenyl)-5H-1,3-oxazole Chemical compound N1N(NN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CC=CC=C1)C1=CC=CC=C1 OGLAHQNDFOEYAO-UHFFFAOYSA-N 0.000 description 1
- WDFZKTFMOYOIIP-UHFFFAOYSA-N 4-(imidazol-1-ylmethyl)-4-methyl-2-(4-phenylphenyl)-5H-1,3-oxazole Chemical compound N1(C=NC=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CC=CC=C1)C WDFZKTFMOYOIIP-UHFFFAOYSA-N 0.000 description 1
- ZOJXXUDGNJOVGD-UHFFFAOYSA-N 4-(imidazol-1-ylmethyl)-4-phenyl-2-(4-phenylphenyl)-5H-1,3-oxazole Chemical compound N1(C=NC=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CC=CC=C1)C1=CC=CC=C1 ZOJXXUDGNJOVGD-UHFFFAOYSA-N 0.000 description 1
- HLYXCEXHCJAMMQ-UHFFFAOYSA-N 4-benzyl-4-(imidazol-1-ylmethyl)-2-(4-phenylphenyl)-5H-1,3-oxazole Chemical compound N1(C=NC=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CC=CC=C1)CC1=CC=CC=C1 HLYXCEXHCJAMMQ-UHFFFAOYSA-N 0.000 description 1
- GUAPGTXZVNZXMT-UHFFFAOYSA-N 4-phenyl-2-(4-phenylmethoxyphenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)OCC1=CC=CC=C1)C1=CC=CC=C1 GUAPGTXZVNZXMT-UHFFFAOYSA-N 0.000 description 1
- ONTQBMCDBMDQSM-UHFFFAOYSA-N 4-phenyl-2-(4-thiophen-3-ylphenyl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C1=CC=C(C=C1)C1=CSC=C1)C1=CC=CC=C1 ONTQBMCDBMDQSM-UHFFFAOYSA-N 0.000 description 1
- DKQNGFWHSZZAPT-UHFFFAOYSA-N 4-phenyl-2-(5-phenylthiophen-2-yl)-4-(1,2,4-triazol-1-ylmethyl)-5H-1,3-oxazole Chemical compound N1(N=CN=C1)CC1(N=C(OC1)C=1SC(=CC1)C1=CC=CC=C1)C1=CC=CC=C1 DKQNGFWHSZZAPT-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 description 1
- 125000003601 C2-C6 alkynyl group Chemical group 0.000 description 1
- 125000000041 C6-C10 aryl group Chemical group 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 241001337994 Cryptococcus <scale insect> Species 0.000 description 1
- 241000195493 Cryptophyta Species 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- 206010061598 Immunodeficiency Diseases 0.000 description 1
- 208000029462 Immunodeficiency disease Diseases 0.000 description 1
- 101710146773 Lanosterol 14-alpha demethylase Proteins 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- BYBLEWFAAKGYCD-UHFFFAOYSA-N Miconazole Chemical compound ClC1=CC(Cl)=CC=C1COC(C=1C(=CC(Cl)=CC=1)Cl)CN1C=NC=C1 BYBLEWFAAKGYCD-UHFFFAOYSA-N 0.000 description 1
- 238000005481 NMR spectroscopy Methods 0.000 description 1
- 229920001213 Polysorbate 20 Polymers 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 108010013803 Sterol 14-Demethylase Proteins 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 1
- 208000002474 Tinea Diseases 0.000 description 1
- JFBZPFYRPYOZCQ-UHFFFAOYSA-N [Li].[Al] Chemical compound [Li].[Al] JFBZPFYRPYOZCQ-UHFFFAOYSA-N 0.000 description 1
- 235000011054 acetic acid Nutrition 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 150000001335 aliphatic alkanes Chemical class 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 150000001350 alkyl halides Chemical class 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 238000002827 antifungal susceptibility testing Methods 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 150000003851 azoles Chemical class 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-N benzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-N 0.000 description 1
- 229940092714 benzenesulfonic acid Drugs 0.000 description 1
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 1
- 125000000499 benzofuranyl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- AGEZXYOZHKGVCM-UHFFFAOYSA-N benzyl bromide Chemical compound BrCC1=CC=CC=C1 AGEZXYOZHKGVCM-UHFFFAOYSA-N 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 238000002815 broth microdilution Methods 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 210000002421 cell wall Anatomy 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 235000015165 citric acid Nutrition 0.000 description 1
- 229960004022 clotrimazole Drugs 0.000 description 1
- VNFPBHJOKIVQEB-UHFFFAOYSA-N clotrimazole Chemical compound ClC1=CC=CC=C1C(N1C=NC=C1)(C=1C=CC=CC=1)C1=CC=CC=C1 VNFPBHJOKIVQEB-UHFFFAOYSA-N 0.000 description 1
- 229940125904 compound 1 Drugs 0.000 description 1
- 238000012790 confirmation Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 239000006184 cosolvent Substances 0.000 description 1
- 230000007812 deficiency Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 238000003113 dilution method Methods 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 239000002662 enteric coated tablet Substances 0.000 description 1
- 125000004185 ester group Chemical group 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 239000008098 formaldehyde solution Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 239000012362 glacial acetic acid Substances 0.000 description 1
- 229910052602 gypsum Inorganic materials 0.000 description 1
- 239000010440 gypsum Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 150000002460 imidazoles Chemical class 0.000 description 1
- 230000007813 immunodeficiency Effects 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 238000011081 inoculation Methods 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 125000005956 isoquinolyl group Chemical group 0.000 description 1
- 229960004125 ketoconazole Drugs 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 229940040145 liniment Drugs 0.000 description 1
- 238000004811 liquid chromatography Methods 0.000 description 1
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 1
- 229940057995 liquid paraffin Drugs 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 238000001819 mass spectrum Methods 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- NDBQJIBNNUJNHA-AENDTGMFSA-N methyl (2r)-2-amino-3-hydroxypropanoate;hydrochloride Chemical compound Cl.COC(=O)[C@H](N)CO NDBQJIBNNUJNHA-AENDTGMFSA-N 0.000 description 1
- 229960002509 miconazole Drugs 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- YZMHQCWXYHARLS-UHFFFAOYSA-N naphthalene-1,2-disulfonic acid Chemical compound C1=CC=CC2=C(S(O)(=O)=O)C(S(=O)(=O)O)=CC=C21 YZMHQCWXYHARLS-UHFFFAOYSA-N 0.000 description 1
- 150000007523 nucleic acids Chemical class 0.000 description 1
- 102000039446 nucleic acids Human genes 0.000 description 1
- 108020004707 nucleic acids Proteins 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 206010033675 panniculitis Diseases 0.000 description 1
- 238000005192 partition Methods 0.000 description 1
- 230000001717 pathogenic effect Effects 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 150000004291 polyenes Chemical class 0.000 description 1
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 1
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 239000012264 purified product Substances 0.000 description 1
- 238000010791 quenching Methods 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 125000005493 quinolyl group Chemical group 0.000 description 1
- 238000006722 reduction reaction Methods 0.000 description 1
- 238000007430 reference method Methods 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 238000003797 solvolysis reaction Methods 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 210000004304 subcutaneous tissue Anatomy 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 238000010189 synthetic method Methods 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 125000003831 tetrazolyl group Chemical group 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- 150000003852 triazoles Chemical class 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- 229940099259 vaseline Drugs 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/06—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/10—Antimycotics
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
技术领域technical field
本发明属于药物合成技术领域,涉及一类2,4,4-三取代二氢噁唑衍生物及其药学上可接受的盐、水合物、溶剂化物或其前药,它们的制备方法以及其作为治疗由真菌感染引起的各类疾病的药物中的用途。The invention belongs to the technical field of drug synthesis, and relates to a class of 2,4,4-trisubstituted dihydrooxazole derivatives and pharmaceutically acceptable salts, hydrates, solvates or prodrugs thereof, their preparation methods and their preparations. Use as a medicament for the treatment of various diseases caused by fungal infections.
背景技术Background technique
真菌感染可分为浅表真菌感染和深部真菌感染,其中浅表真菌感染主要由癣菌引起,如皮肤,毛发和指(趾)甲等真菌感染;深部真菌感染主要由白色念珠菌、新生隐球菌和烟曲霉菌引起,如皮下组织、内膜等深部组织的感染。近三十年,随着免疫缺陷的患者越来越多,真菌感染率急剧飙升,严重威胁人类健康,寻找新结构类型的抗真菌药物迫在眉睫。Fungal infection can be divided into superficial fungal infection and deep fungal infection, among which superficial fungal infection is mainly caused by ringworm, such as skin, hair and nail (toe) fungal infection; deep fungal infection is mainly caused by Candida albicans, Cryptophytes Cocci and Aspergillus fumigatus cause infections of deep tissues such as subcutaneous tissue and intima. In the past three decades, with the increasing number of patients with immunodeficiency, the rate of fungal infection has soared, which seriously threatens human health. It is urgent to find new structural types of antifungal drugs.
目前临床上抗真菌药物根据作用机制的不同,可以分为抑制麦角甾醇合成的唑类药物;破坏细胞壁的棘白霉素类抗真菌药物、造成细胞膜泄漏的多烯类药物和作用于核酸的抗代谢类抗真菌药物。其中,唑类药物通过抑制羊毛甾醇14α-去甲基化酶(CYP51)的活性,阻断麦角甾醇的合成,是抗真菌药物领域研究最活跃,最成熟的靶点。目前临床上的唑类抗真菌药物主要分为两类:咪唑类药物如咪康唑(Miconazole)、克霉唑(Clotrimazole)、酮康唑(Ketoconazole);三氮唑类药物如氟康唑(Fluconazole)、伊曲康唑(Itraconazole、伏立康唑(Voriconazole)和泊沙康唑(Posaconazole)。尽管唑类药物在临床上发挥着不可替代的作用,但是该类药物严重的毒副作用和耐药菌株的产生,督促着药物化学家开发更多结构类型、高效低毒、给药方式多样的抗真菌药物。At present, clinical antifungal drugs can be divided into azole drugs that inhibit ergosterol synthesis according to different mechanisms of action; echinocandicin antifungal drugs that destroy cell walls, polyene drugs that cause cell membrane leakage, and antifungal drugs that act on nucleic acids. Metabolic antifungal drugs. Among them, azoles block the synthesis of ergosterol by inhibiting the activity of lanosterol 14α-demethylase (CYP51), and are the most active and mature targets in the field of antifungal research. Currently clinical azole antifungal drugs are mainly divided into two categories: imidazoles such as Miconazole, Clotrimazole, Ketoconazole; triazoles such as fluconazole ( Fluconazole), Itraconazole (Itraconazole), Voriconazole (Voriconazole) and Posaconazole (Posaconazole). Although azole drugs play an irreplaceable role in clinical practice, these drugs have serious side effects and drug-resistant strains. , urging medicinal chemists to develop antifungal drugs with more structural types, high efficiency and low toxicity, and various modes of administration.
发明内容SUMMARY OF THE INVENTION
本发明的目的是针对现有技术的不足,提供一类2,4,4-三取代二氢噁唑衍生物,以及所述衍生物的药学可接受的盐、水合物、溶剂化合物或前药,并提供所述衍生物的制备方法以及所述衍生物的用途;同时提供含有所述2,4,4-三取代二氢噁唑衍生物的药物组合物。本发明在参考文献的基础上,设计并合成了一系列2,4,4-三取代二氢噁唑衍生物,该类化合物经体外抗真菌活性测试表明,该类化合物具有较强的抗真菌活性,在治疗真菌感染疾病中具有较大研究价值。The object of the present invention is to provide a class of 2,4,4-trisubstituted dihydrooxazole derivatives, and pharmaceutically acceptable salts, hydrates, solvates or prodrugs of the derivatives in view of the deficiencies of the prior art , and provide the preparation method of the derivative and the use of the derivative; and also provide the pharmaceutical composition containing the 2,4,4-trisubstituted dihydrooxazole derivative. The present invention designs and synthesizes a series of 2,4,4-tri-substituted dihydrooxazole derivatives on the basis of the references. The in vitro antifungal activity test shows that these compounds have strong antifungal properties. It has great research value in the treatment of fungal infections.
为实现上述目的,本发明提供通式I所示的2,4,4-三取代二氢噁唑衍生物及其立体异构体或其药学上可接受的盐、水合物、溶剂化物或前药:In order to achieve the above purpose, the present invention provides 2,4,4-trisubstituted dihydrooxazole derivatives represented by general formula I and stereoisomers thereof or pharmaceutically acceptable salts, hydrates, solvates or pro-forms thereof. medicine:
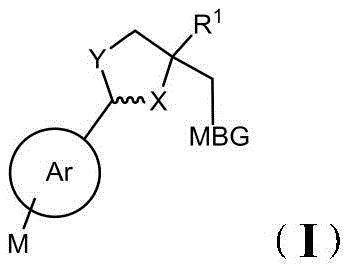
其中:in:
MBG是取代或未取代的四唑基、取代或未取代的三唑基、取代或未取代的咪唑基、或取代或未取代的吡啶基;所述的取代基为:氢、C1-C4烷基、C1-C4烷氧基。MBG is substituted or unsubstituted tetrazolyl, substituted or unsubstituted triazolyl, substituted or unsubstituted imidazolyl, or substituted or unsubstituted pyridyl; the substituents are: hydrogen, C1-C4 alkane base, C1-C4 alkoxy.
Y为O或S;Y is O or S;
X为CH、CH2、N、NH、或O;X is CH, CH2 , N, NH, or O;
为单键或双建; For single-key or double-build;
R1为(C1-C5)烷基、(C3-C6)环烷基、(C1-C4)烷氧基、羧基、-COOR2、-CON(R2)2、5-10元芳基或者5-10元杂芳基,且所述芳基或杂芳基任选0-3个相同或不同的R2取代;R 1 is (C 1 -C 5 ) alkyl, (C 3 -C 6 ) cycloalkyl, (C 1 -C 4 ) alkoxy, carboxyl, -COOR 2 , -CON(R 2 ) 2 , 5 -10-membered aryl or 5-10-membered heteroaryl, and the aryl or heteroaryl is optionally substituted with 0-3 identical or different R 2 ;
Ar环为C3-C6环烷基、5-10元杂环基、C6-C10芳基或C5-C10杂芳基,其中,所述杂环基、杂芳基含有1-3个选自N、O或S的杂原子,并且Ar任选0-3个相同或不同的M取代;The Ar ring is a C3-C6 cycloalkyl group, a 5-10 membered heterocyclic group, a C6 - C10 aryl group or a C5 - C10 heteroaryl group, wherein the heterocyclic group and the heteroaryl group contain 1-3 a heteroatom selected from N, O or S, and Ar is optionally substituted with 0-3 identical or different M;
M为氢、羟基、卤素、硝基、氨基、氰基、(C1-C6)烷基、(C2-C6)烯基、(C2-C6)炔基、(C1-C6)烷氧基、(C1-C6)烷基硫基、任选被羟基、氨基或卤代的(C1-C6)烷基或(C1-C6)烷氧基或(C1-C6)烷基硫基、被单或二(C1-C6烷基)取代的氨基、(C1-C6)烷基酰氨基、游离的、成盐的、酯化的和酰胺化的羧基、(C1-C6)烷基亚磺酰基、磺酰基、(C1-C6)烷氧基、(C1-C6)烷基、(C1-C6)烷基酰基、氨基甲酰基、被单或二(C1-C6烷基)取代的氨基甲酰基、(C1-C3)亚烷基二氧基的取代基;除了M为所连接的供电、吸电基团外,M也可以为5-10元杂环基、C6-C12芳基或C5-C12杂芳基,所述杂环基和杂芳基含有1-3个选自O、N和S的杂原子,且所述的杂环基、芳基或杂芳基任选0-3个相同或不同的R2取代;M is hydrogen, hydroxyl, halogen, nitro, amino, cyano, (C 1 -C 6 ) alkyl, (C 2 -C 6 ) alkenyl, (C 2 -C 6 ) alkynyl, (C 1 - C 6 )alkoxy, (C 1 -C 6 )alkylthio, (C 1 -C 6 )alkyl or (C 1 -C 6 )alkoxy optionally hydroxy, amino or halo (C 1 -C 6 )alkylthio, mono- or di(C 1 -C 6 alkyl) substituted amino, (C 1 -C 6 )alkylamido, free, salified, esterified and amidated carboxyl, (C 1 -C 6 ) alkylsulfinyl, sulfonyl, (C 1 -C 6 ) alkoxy, (C 1 -C 6 ) alkyl, (C 1 -C 6 ) Substituents of alkylacyl, carbamoyl, mono- or di( C1 - C6 alkyl) substituted carbamoyl, ( C1 - C3) alkylenedioxy; except that M is the attached power , in addition to the charge-absorbing group, M can also be a 5-10 membered heterocyclic group, a C 6 -C 12 aryl group or a C 5 -C 12 heteroaryl group, and the heterocyclic group and the heteroaryl group contain 1-3 A heteroatom selected from O, N and S, and the heterocyclic group, aryl group or heteroaryl group is optionally substituted by 0-3 identical or different R 2 ;
R2为氢、羟基、卤素、硝基、氨基、氰基、(C1-C6)烷基、(C1-C6)烯基、(C1-C6)炔基、(C1-C6)烷氧基、任选被羟基、氨基或卤代的(C1-C6)烷基或(C1-C6)烷氧基、被单或二(C1-C6烷基)取代的氨基、(C1-C6)烷基酰氨基、游离的、成盐的、酯化的和酰胺化的羧基、(C1-C6)烷基亚磺酰基、磺酰基、(C1-C6)烷氧基、(C1-C6)烷基、(C1-C6)烷基酰基、氨基甲酰基、被单或二(C1-C6烷基)取代的氨基甲酰基、(C1-C3)亚烷基二氧基。R 2 is hydrogen, hydroxyl, halogen, nitro, amino, cyano, (C 1 -C 6 )alkyl, (C 1 -C 6 )alkenyl, (C 1 -C 6 )alkynyl, (C 1 ) -C 6 )alkoxy, optionally hydroxy, amino or halogenated (C 1 -C 6 )alkyl or (C 1 -C 6 )alkoxy, mono or di(C 1 -C 6 alkyl) ) substituted amino, (C 1 -C 6 ) alkylamido, free, salified, esterified and amidated carboxyl, (C 1 -C 6 ) alkylsulfinyl, sulfonyl, ( C 1 -C 6 )alkoxy, (C 1 -C 6 )alkyl, (C 1 -C 6 )alkylacyl, carbamoyl, amino substituted with mono or di(C 1 -C 6 alkyl) Formyl, (C 1 -C 3 )alkylenedioxy.
本发明优选通式I所示的化合物,及其立体异构体或其药学上可接受的盐、水合物、溶剂化物或前药,The present invention is preferably the compound represented by the general formula I, and its stereoisomers or their pharmaceutically acceptable salts, hydrates, solvates or prodrugs,
其中,in,
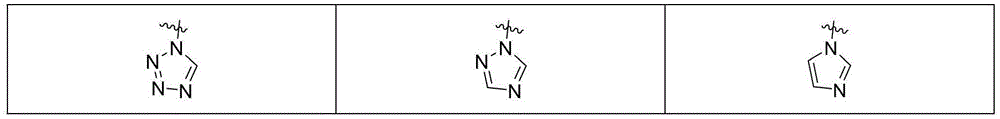
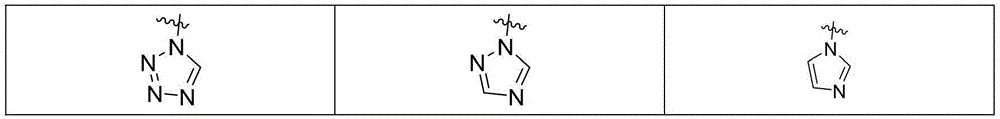
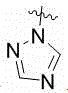
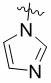
MBG选自以下结构:MBG is selected from the following structures:
本发明优选通式I所示的化合物,及其立体异构体或其药学上可接受的盐、水合物、溶剂化物或前药,The present invention is preferably the compound represented by the general formula I, and its stereoisomers or their pharmaceutically acceptable salts, hydrates, solvates or prodrugs,
其中,in,
Y为O;Y is O;
为双键,X为N。 is a double bond, and X is N.
本发明优选通式I所示的化合物,及其立体异构体或其药学上可接受的盐、水合物、溶剂化物或前药,The present invention is preferably the compound represented by the general formula I, and its stereoisomers or their pharmaceutically acceptable salts, hydrates, solvates or prodrugs,
其中,in,
Y为O;Y is O;
为双键,X为N; is a double bond, and X is N;
MBG选自以下结构:MBG is selected from the following structures:
本发明优选通式I所示的化合物,及其立体异构体或其药学上可接受的盐、水合物、溶剂化物或前药,The present invention is preferably the compound represented by the general formula I, and its stereoisomers or their pharmaceutically acceptable salts, hydrates, solvates or prodrugs,
其中,in,
R1为(C1-C5)烷基、(C3-C6)环烷基、苄基、-(CHF)Ph、-(CF2)Ph或苯基,且苄基、-(CHF)Ph、-(CF2)Ph和苯基的苯环上任选0-3个R2取代。R 1 is (C 1 -C 5 )alkyl, (C 3 -C 6 )cycloalkyl, benzyl, -(CHF)Ph, -(CF 2 )Ph, or phenyl, and benzyl, -(CHF )Ph, -(CF 2 )Ph and the phenyl ring of phenyl are optionally substituted with 0-3 R 2 .
本发明优选通式I所示的化合物,及其立体异构体或其药学上可接受的盐、水合物、溶剂化物或前药,The present invention is preferably the compound represented by the general formula I, and its stereoisomers or their pharmaceutically acceptable salts, hydrates, solvates or prodrugs,
其中,in,
Y为O;Y is O;
为双键,X为N; is a double bond, and X is N;
R1为(C1-C5)烷基、(C3-C6)环烷基、苄基、-(CHF)Ph、-(CF2)Ph或苯基,且苄基、-(CHF)Ph、-(CF2)Ph和苯基的苯环上任选0-3个R2取代;R 1 is (C 1 -C 5 )alkyl, (C 3 -C 6 )cycloalkyl, benzyl, -(CHF)Ph, -(CF 2 )Ph, or phenyl, and benzyl, -(CHF ) Ph, -(CF 2 )Ph and optional 0-3 R 2 substitutions on the benzene ring of phenyl;
MBG为以下结构之一:MBG is one of the following structures:
Ar环为呋喃基、噻吩基、噁唑基、异噁唑基、吡咯基、吡唑基、苯基、萘基、苯并呋喃基、苯并噻唑基、苯并噻吩基、苯并吡唑基或吲哚基,且Ar任选1-3个相同或不同的M取代。Ar ring is furyl, thienyl, oxazolyl, isoxazolyl, pyrrolyl, pyrazolyl, phenyl, naphthyl, benzofuranyl, benzothiazolyl, benzothienyl, benzopyrazole group or indolyl group, and Ar is optionally substituted with 1-3 same or different M.
本发明优选如下化合物及其药学上可接受的盐、水合物、溶剂化物或前药,但这些化合物并不意味着对本发明的任何限制:The present invention preferably the following compounds and their pharmaceutically acceptable salts, hydrates, solvates or prodrugs, but these compounds do not mean any limitation to the present invention:
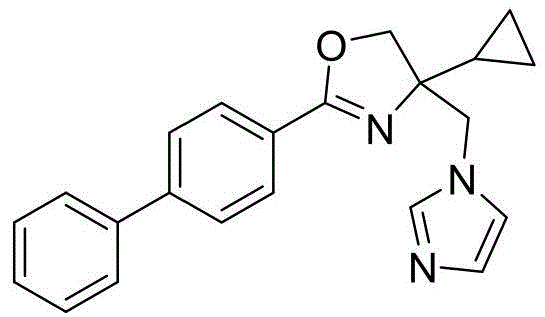
4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-4-甲基-4,5-二氢噁唑4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-methyl-4,5-dihydrooxazole
4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-4-异丙基-4,5-二氢噁唑4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-isopropyl-4,5-dihydrooxazole
4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-4-环丙基-4,5-二氢噁唑4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-cyclopropyl-4,5-dihydrooxazole
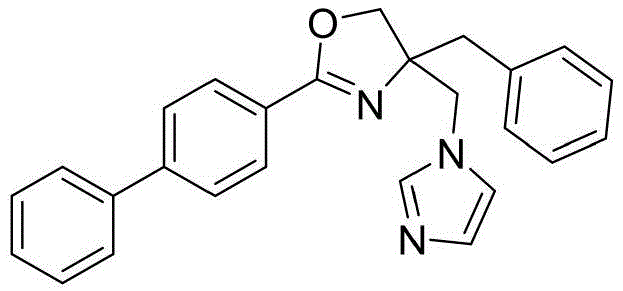
4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-4-苄基-4,5-二氢噁唑4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-benzyl-4,5-dihydrooxazole
4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-5-(氟(苯基)甲基)-4,5-二氢噁唑4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-5-(fluoro(phenyl)methyl)-4,5- Dihydrooxazole
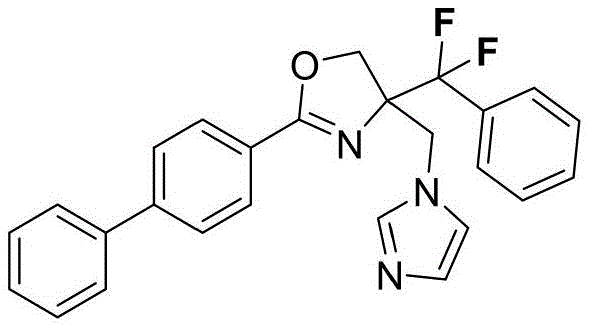
4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-5-(二氟(苯基)甲基)-4,5-二氢噁唑4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-5-(difluoro(phenyl)methyl)-4,5 - Dihydrooxazole
4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-4-苯基-4,5-二氢噁唑4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-phenyl-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-4-苯基-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-phenyl-4,5 - Dihydrooxazole
4-((1H-四氮唑-2-基)甲基)-2-([1,1'-联苯]-4-基)-4-苯基-4,5-二氢噁唑4-((1H-Tetrazol-2-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-phenyl-4,5-dihydrooxazole
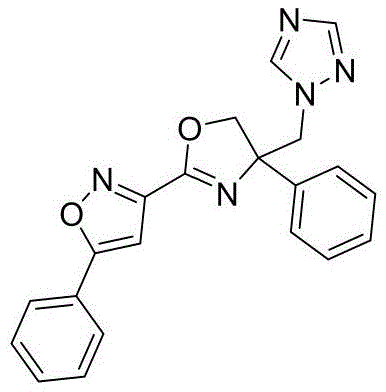
3-(4-((1H-1,2,4-三氮唑-1-基)甲基)-4-苯基-4,5-二氢噁唑-2-基)-5-苯基异噁唑3-(4-((1H-1,2,4-Triazol-1-yl)methyl)-4-phenyl-4,5-dihydrooxazol-2-yl)-5-phenyl Isoxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-4-苯基-2-(5-苯基噻吩-2-基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-4-phenyl-2-(5-phenylthiophen-2-yl)-4,5-dihydrooxane azole
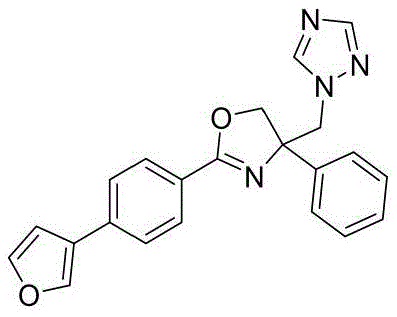
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(4-(呋喃-3-基)苯基)-4-苯基-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(4-(furan-3-yl)phenyl)-4-phenyl-4,5-di Hydroxazole
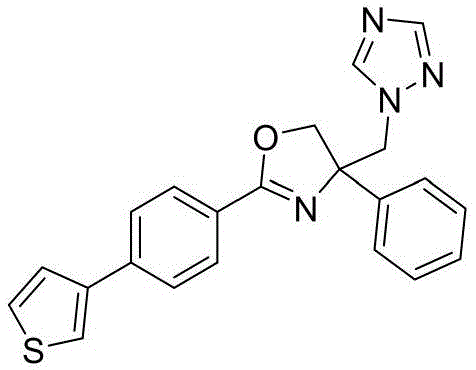
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(4-(噻吩-3-基)苯基)-4-苯基-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(4-(thiophen-3-yl)phenyl)-4-phenyl-4,5-di Hydroxazole
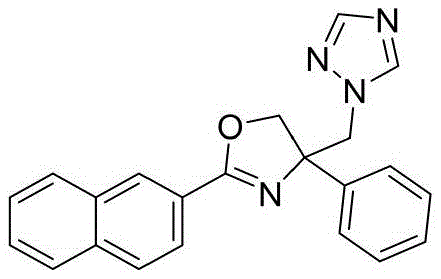
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(萘-2-基)-4-苯基-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(naphthalen-2-yl)-4-phenyl-4,5-dihydrooxazole
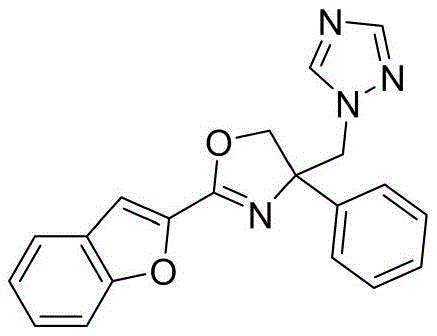
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(苯并呋喃-2-基)-4-苯基-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(benzofuran-2-yl)-4-phenyl-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(苯并[b]噻吩-2-基)-4-苯基-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(benzo[b]thiophen-2-yl)-4-phenyl-4,5-dihydro oxazole
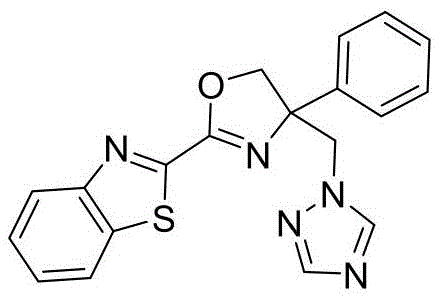
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(苯并[d]噻唑-2-基)-4-苯基-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(benzo[d]thiazol-2-yl)-4-phenyl-4,5-dihydro oxazole
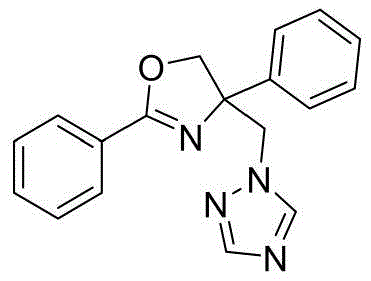
4-((1H-1,2,4-三氮唑-1-基)甲基)-2,4-二苯基-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2,4-diphenyl-4,5-dihydrooxazole
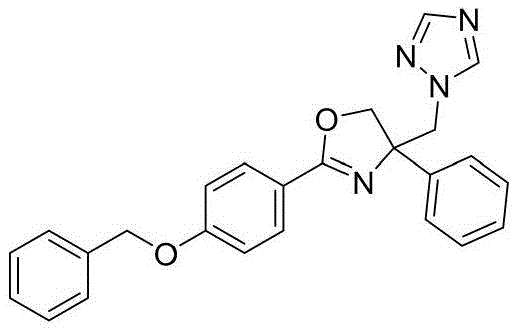
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(4-(苄氧基)苯基)-4-苯基-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(4-(benzyloxy)phenyl)-4-phenyl-4,5-dihydrooxane azole
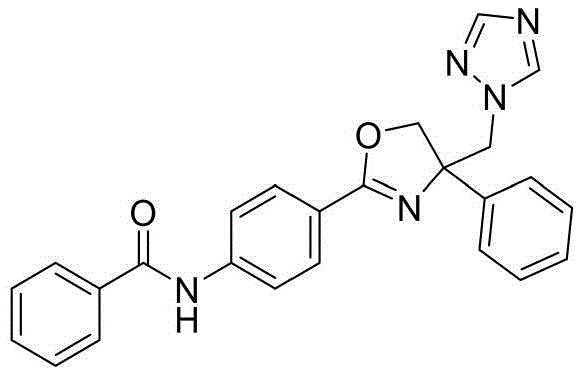
N-(4-(4-((1H-1,2,4-三氮唑-1-基)甲基)-4-苯基-4,5-二氢噁唑-2-基)苯基)苯甲酰胺N-(4-(4-((1H-1,2,4-triazol-1-yl)methyl)-4-phenyl-4,5-dihydrooxazol-2-yl)phenyl ) benzamide
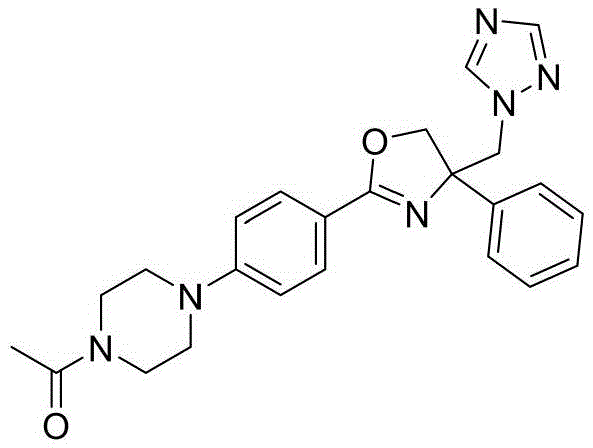
1-(4-(4-(4-((1H-1,2,4-三氮唑-1-基)甲基)-4-苯基-4,5-二氢噁唑-2-基)苯基)哌嗪-1-基)乙酮1-(4-(4-(4-((1H-1,2,4-triazol-1-yl)methyl)-4-phenyl-4,5-dihydrooxazol-2-yl )phenyl)piperazin-1-yl)ethanone
2-(4-(4-((1H-1,2,4-三氮唑-1-基)甲基)-4-苯基-4,5-二氢噁唑-2-基)苯基)-5-苯基-1,3,4-噁二唑2-(4-(4-((1H-1,2,4-triazol-1-yl)methyl)-4-phenyl-4,5-dihydrooxazol-2-yl)phenyl )-5-phenyl-1,3,4-oxadiazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(4-氟苯基)-2-(4-(呋喃-3-基)苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-4-(4-fluorophenyl)-2-(4-(furan-3-yl)phenyl)- 4,5-Dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(4-氯苯基)-2-(4-(呋喃-3-基)苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-4-(4-chlorophenyl)-2-(4-(furan-3-yl)phenyl)- 4,5-Dihydrooxazole
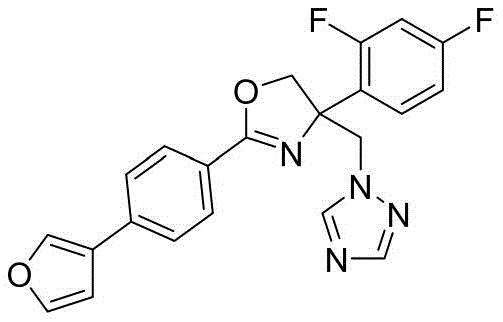
4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(2,4-二氟苯基)-2-(4-(呋喃-3-基)苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-4-(2,4-difluorophenyl)-2-(4-(furan-3-yl)benzene base)-4,5-dihydrooxazole
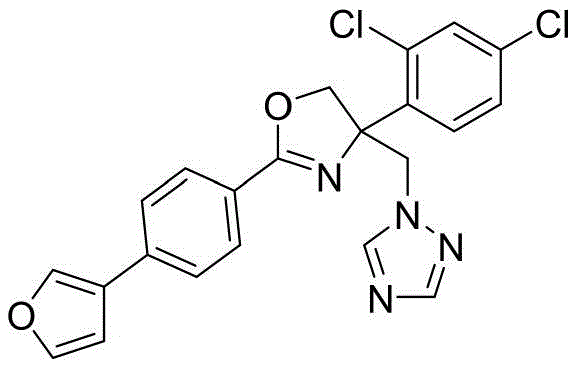
4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(2,4-二氯苯基)-2-(4-(呋喃-3-基)苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-4-(2,4-dichlorophenyl)-2-(4-(furan-3-yl)benzene base)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(4-氟苯基)-2-(4-(噻吩-3-基)苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-4-(4-fluorophenyl)-2-(4-(thiophen-3-yl)phenyl)- 4,5-Dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(4-氯苯基)-2-(4-(噻吩-3-基)苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-4-(4-chlorophenyl)-2-(4-(thiophen-3-yl)phenyl)- 4,5-Dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(2,4-二氟苯基)-2-(4-(噻吩-3-基)苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-4-(2,4-difluorophenyl)-2-(4-(thiophen-3-yl)benzene base)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(2,4-二氯苯基)-2-(4-(噻吩-3-基)苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-4-(2,4-dichlorophenyl)-2-(4-(thiophen-3-yl)benzene base)-4,5-dihydrooxazole
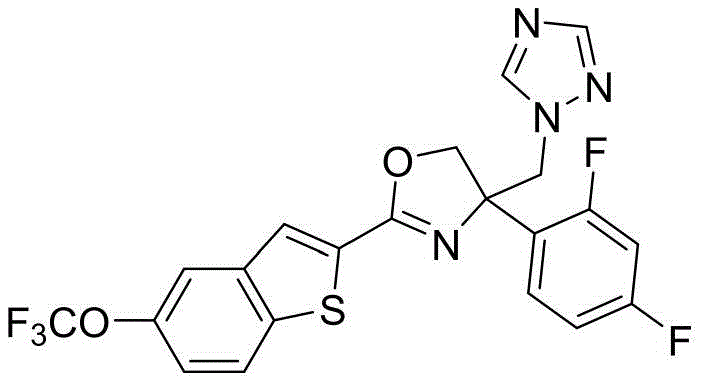
4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(2,4-二氟苯基)-2-(5-(三氟甲氧基)苯并[b]噻吩-2-基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-4-(2,4-difluorophenyl)-2-(5-(trifluoromethoxy)benzene [b]thiophen-2-yl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氟苯并[b]噻吩-2-基)-4-(4-氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-fluorobenzo[b]thiophen-2-yl)-4-(4-fluorophenyl )-4,5-dihydrooxazole
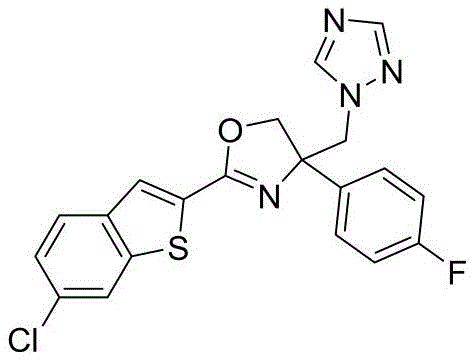
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氟苯并[b]噻吩-2-基)-4-(4-氯苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-fluorobenzo[b]thiophen-2-yl)-4-(4-chlorophenyl )-4,5-dihydrooxazole
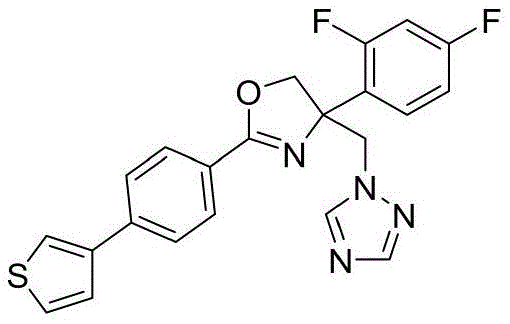
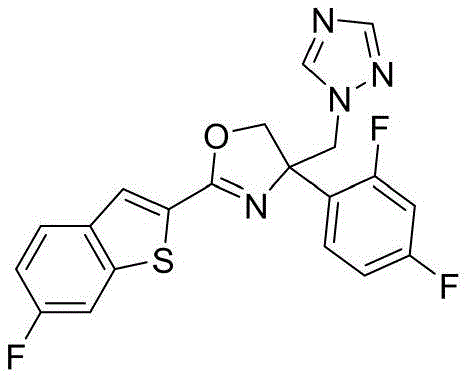
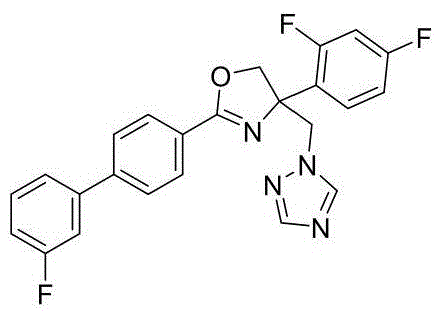
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氟苯并[b]噻吩-2-基)-4-(2,4-二氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-fluorobenzo[b]thiophen-2-yl)-4-(2,4-di Fluorophenyl)-4,5-dihydrooxazole
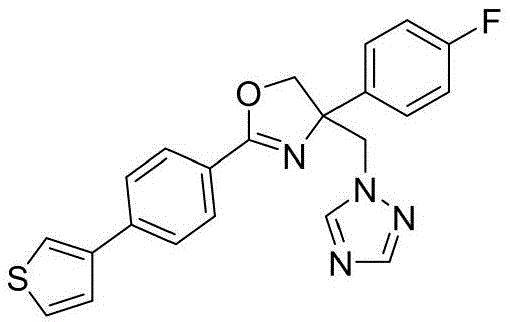
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氟苯并[b]噻吩-2-基)-4-(3,5-二氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-fluorobenzo[b]thiophen-2-yl)-4-(3,5-di Fluorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氟苯并[b]噻吩-2-基)-4-(2,4-二氯苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-fluorobenzo[b]thiophen-2-yl)-4-(2,4-di chlorophenyl)-4,5-dihydrooxazole
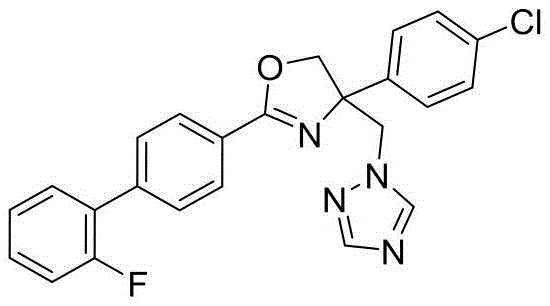
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氯苯并[b]噻吩-2-基)-4-(4-氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-chlorobenzo[b]thiophen-2-yl)-4-(4-fluorophenyl )-4,5-dihydrooxazole
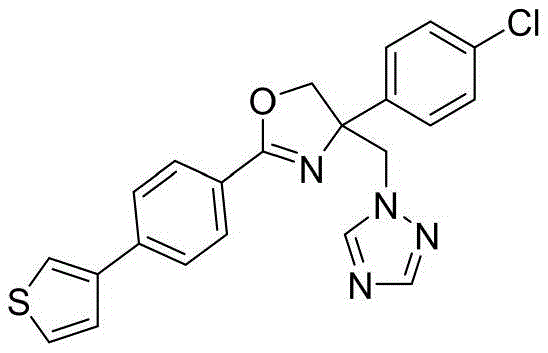
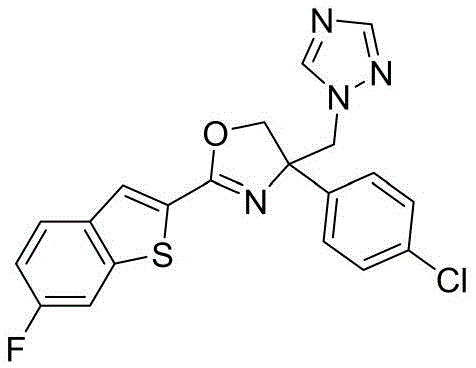
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氯苯并[b]噻吩-2-基)-4-(4-氯苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-chlorobenzo[b]thiophen-2-yl)-4-(4-chlorophenyl )-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氯苯并[b]噻吩-2-基)-4-(2,4-二氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-chlorobenzo[b]thiophen-2-yl)-4-(2,4-di Fluorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氯苯并[b]噻吩-2-基)-4-(3,5-二氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-chlorobenzo[b]thiophen-2-yl)-4-(3,5-di Fluorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氯苯并[b]噻吩-2-基)-4-(2,4-二氯苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-chlorobenzo[b]thiophen-2-yl)-4-(2,4-di chlorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-溴苯并[b]噻吩-2-基)-4-(4-氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-bromobenzo[b]thiophen-2-yl)-4-(4-fluorophenyl )-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-溴苯并[b]噻吩-2-基)-4-(4-氯苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-bromobenzo[b]thiophen-2-yl)-4-(4-chlorophenyl )-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-溴苯并[b]噻吩-2-基)-4-(2,4-二氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-bromobenzo[b]thiophen-2-yl)-4-(2,4-di Fluorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-溴苯并[b]噻吩-2-基)-4-(3,5-二氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-bromobenzo[b]thiophen-2-yl)-4-(3,5-di Fluorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-溴苯并[b]噻吩-2-基)-4-(2,4-二氯苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-bromobenzo[b]thiophen-2-yl)-4-(2,4-di chlorophenyl)-4,5-dihydrooxazole
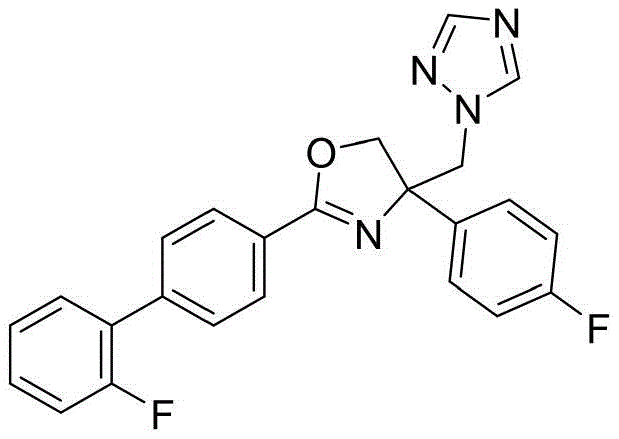
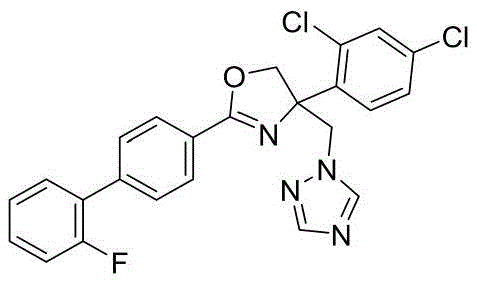
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(2'-氟-[1,1'-联苯]-4-基)-4-(4-氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(2'-fluoro-[1,1'-biphenyl]-4-yl)-4-( 4-Fluorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(2'-氟-[1,1'-联苯]-4-基)-4-(4-氯苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(2'-fluoro-[1,1'-biphenyl]-4-yl)-4-( 4-Chlorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(2'-氟-[1,1'-联苯]-4-基)-4-(2,4-二氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(2'-fluoro-[1,1'-biphenyl]-4-yl)-4-( 2,4-Difluorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(2'-氟-[1,1'-联苯]-4-基)-4-(3,5-二氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(2'-fluoro-[1,1'-biphenyl]-4-yl)-4-( 3,5-Difluorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(2'-氟-[1,1'-联苯]-4-基)-4-(2,4-二氯苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(2'-fluoro-[1,1'-biphenyl]-4-yl)-4-( 2,4-Dichlorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(3'-氟-[1,1'-联苯]-4-基)-4-(4-氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(3'-fluoro-[1,1'-biphenyl]-4-yl)-4-( 4-Fluorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(3'-氟-[1,1'-联苯]-4-基)-4-(4-氯苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(3'-fluoro-[1,1'-biphenyl]-4-yl)-4-( 4-Chlorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(3'-氟-[1,1'-联苯]-4-基)-4-(2,4-二氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(3'-fluoro-[1,1'-biphenyl]-4-yl)-4-( 2,4-Difluorophenyl)-4,5-dihydrooxazole
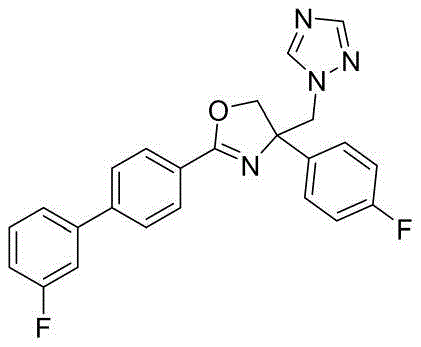
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(3'-氟-[1,1'-联苯]-4-基)-4-(3,5-二氟苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(3'-fluoro-[1,1'-biphenyl]-4-yl)-4-( 3,5-Difluorophenyl)-4,5-dihydrooxazole
4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(3'-氟-[1,1'-联苯]-4-基)-4-(2,4-二氯苯基)-4,5-二氢噁唑4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(3'-fluoro-[1,1'-biphenyl]-4-yl)-4-( 2,4-Dichlorophenyl)-4,5-dihydrooxazole
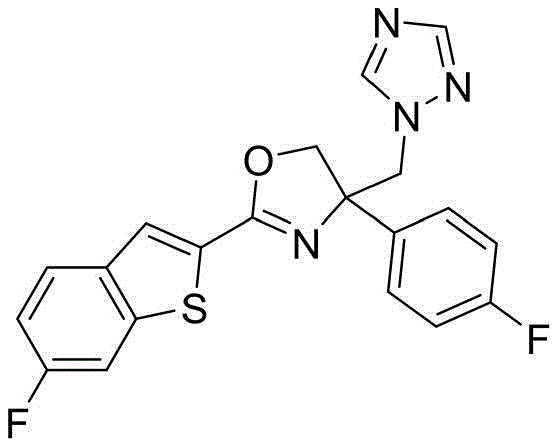
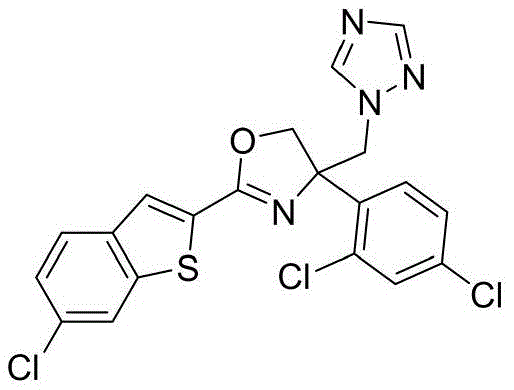
4-((1H-四氮唑-1-基)甲基)-2-(6-氯苯并[b]噻吩-2-基)-4-(4-氟苯基)-4,5-二氢噁唑4-((1H-Tetrazol-1-yl)methyl)-2-(6-chlorobenzo[b]thiophen-2-yl)-4-(4-fluorophenyl)-4,5- Dihydrooxazole
而且,按照本发明所属领域的一些通常方法,本发明中通式I的部分化合物具有碱性基团,可以与酸生成药学上可接受的盐。可药用加成盐包括无机酸和有机酸加成盐,与下列酸加成的盐是特别优选的:盐酸、氢溴酸、硫酸、磷酸、甲磺酸、乙磺酸、对甲苯磺酸、苯磺酸、萘二磺酸、乙酸、丙酸、乳酸、三氟乙酸、马来酸、柠檬酸、富马酸、草酸、酒石酸、苯甲酸等。Moreover, according to some common methods in the field of the present invention, some of the compounds of the general formula I in the present invention have basic groups and can form pharmaceutically acceptable salts with acids. Pharmaceutically acceptable addition salts include inorganic and organic acid addition salts, with addition salts with the following acids being particularly preferred: hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric acid, methanesulfonic acid, ethanesulfonic acid, p-toluenesulfonic acid , benzenesulfonic acid, naphthalenedisulfonic acid, acetic acid, propionic acid, lactic acid, trifluoroacetic acid, maleic acid, citric acid, fumaric acid, oxalic acid, tartaric acid, benzoic acid, etc.
此外,本发明还包括本发明衍生物的前药。本发明衍生物的前药是通式I的衍生物,它们自身可能具有较弱的活性甚至没有活性,但是在给药后,在生理条件下(例如通过代谢、溶剂分解或另外的方式)被转化成相应的生物活性形式。Furthermore, the present invention also includes prodrugs of the derivatives of the present invention. Prodrugs of the derivatives of the present invention are derivatives of general formula I, which themselves may have weak or even no activity, but after administration, are destroyed under physiological conditions (eg by metabolism, solvolysis or otherwise) by into the corresponding biologically active form.
通式I所示的化合物可以以非溶剂化形式和含有药学上可接受的溶剂(如水、乙醇等)的溶剂化形式。通式I所示的化合物可以含有不对称或手性中心,因此可以以不同立体异构形式存在。本发明的所有立体异构形式,包括但不限于非对映异构体、对映异构体和阻转异构体以及它们的混合物(如外消旋混合物),均包括在本发明的范围内。Compounds of formula I can be in unsolvated forms as well as solvated forms containing pharmaceutically acceptable solvents such as water, ethanol, and the like. The compounds represented by the general formula I may contain asymmetric or chiral centers and thus may exist in different stereoisomeric forms. All stereoisomeric forms of the present invention, including but not limited to diastereomers, enantiomers, and atropisomers, and mixtures thereof (eg, racemic mixtures), are included within the scope of the present invention Inside.
通式I所示的化合物可以以不同的互变异构体形式存在,所有这些形式均包括在本发明的范围内。术语“互变异构体”或“互变异构形式”是指经由低能垒互相转化的不同能量的结构异构体。Compounds of general formula I may exist in different tautomeric forms, all of which are included within the scope of the present invention. The term "tautomer" or "tautomeric form" refers to structural isomers of different energies that interconvert via a low energy barrier.
本发明中“卤素”是指氟、氯、溴或碘代;“烷基”是指直链或支链的烷基;“亚烷基”是指直链或支链的亚烷基;“芳基”是指除去芳烃中的一个或不同位置的两个氢原子而得到的有机基团,如苯基、萘基;“杂芳基”是指含有一个或多个选自N、O、S杂原子的单环或多环的环状体系,该环状体系是指具有芳香性的,并且除去环状体系中的一个或不同位置的两个氢原子而得到的有机基团,如噻唑基,咪唑基、吡啶基、吡唑基、(1,2,3)-和(1,2,4)-三唑基、呋喃基、噻吩基、吡咯基,吲哚基,苯并噻唑基,噁唑基,异噁唑基,萘基,喹啉基,异喹啉基,苯并咪唑基,苯并噁唑基等。In the present invention, "halogen" refers to fluorine, chlorine, bromine or iodine; "alkyl" refers to straight-chain or branched alkyl; "alkylene" refers to straight-chain or branched alkylene; " "Aryl" refers to an organic group obtained by removing two hydrogen atoms at one or different positions in an aromatic hydrocarbon, such as phenyl and naphthyl; "heteroaryl" refers to an organic group containing one or more selected from N, O, S heteroatom monocyclic or polycyclic ring system, the ring system refers to an organic group which is aromatic and obtained by removing two hydrogen atoms in one or different positions in the ring system, such as thiazole base, imidazolyl, pyridyl, pyrazolyl, (1,2,3)- and (1,2,4)-triazolyl, furyl, thienyl, pyrrolyl, indolyl, benzothiazolyl , oxazolyl, isoxazolyl, naphthyl, quinolyl, isoquinolyl, benzimidazolyl, benzoxazolyl, etc.
本发明可以含有通式I的衍生物,及其药学上可接受的盐、水合物、溶剂化物或前药作为活性成份,与药学上可接受的载体或赋形剂混合制备成组合物,并制备成临床上可接受的剂型,上述药学上可接受的赋形剂是指任何可用于药学领域的稀释剂、辅助剂和/或载体。本发明的衍生物可以与其他活性成份组合使用,只要它们不产生其他不利的作用,例如过敏反应。The present invention can contain derivatives of general formula I, and pharmaceutically acceptable salts, hydrates, solvates or prodrugs thereof as active ingredients, mixed with pharmaceutically acceptable carriers or excipients to prepare a composition, and Prepared into a clinically acceptable dosage form, the above-mentioned pharmaceutically acceptable excipients refer to any diluents, adjuvants and/or carriers that can be used in the pharmaceutical field. The derivatives of the present invention can be used in combination with other active ingredients as long as they do not produce other adverse effects, such as allergic reactions.
本发明的药用组合物可配制成若干种剂型,其中含有药物领域中一些常用的赋形剂。如上所述的若干种剂型可以采用注射剂、片剂、胶囊剂、气雾剂、栓剂、膜剂、滴丸剂、外用搽剂、软膏剂等剂型药物。The pharmaceutical composition of the present invention can be formulated into several dosage forms, which contain some commonly used excipients in the pharmaceutical field. Several of the above-mentioned dosage forms can be used as injections, tablets, capsules, aerosols, suppositories, films, drop pills, external liniments, ointments and other dosage forms.
用于本发明药物组合物的载体是药物领域中可得到的常见类型,包括:粘合剂、润滑剂、崩解剂、助溶剂、稀释剂、稳定剂、悬浮剂、无色素、矫味剂、防腐剂、加溶剂和基质等。药物制剂可以经口服或胃肠外方式(例如静脉内、皮下、腹膜内或局部)给药,如果某些药物在胃部条件下不稳定的,可将其配制成肠衣片剂。The carriers used in the pharmaceutical compositions of the present invention are of the common types available in the pharmaceutical field, including: binders, lubricants, disintegrants, cosolvents, diluents, stabilizers, suspending agents, colorless, flavoring agents , preservatives, solubilizers and substrates, etc. Pharmaceutical formulations may be administered orally or parenterally (eg, intravenously, subcutaneously, intraperitoneally or topically), and if certain drugs are unstable under gastric conditions, they may be formulated as enteric-coated tablets.
本发明通式I的化合物可以通过包括化学领域众所周知的方法来合成,尤其根据本发明的说明来制备;本发明中的室温指环境温度,为10℃至30℃。The compounds of general formula I of the present invention can be synthesized by methods including well-known methods in the field of chemistry, especially prepared according to the description of the present invention; room temperature in the present invention refers to ambient temperature, which is 10°C to 30°C.
应当理解的是,本文所述的实施例和反应方案仅为例举说明目的,本领域技术人员可以据此进行各种修改和改变,并且这些修饰和改变均包括在本发明的主旨和范围内以及所附权利要求的范围内。It should be understood that the examples and reaction schemes described herein are for illustrative purposes only, and those skilled in the art can make various modifications and changes accordingly, and these modifications and changes are all included within the spirit and scope of the present invention and within the scope of the appended claims.
化合物制备的一般反应方案General reaction scheme for compound preparation
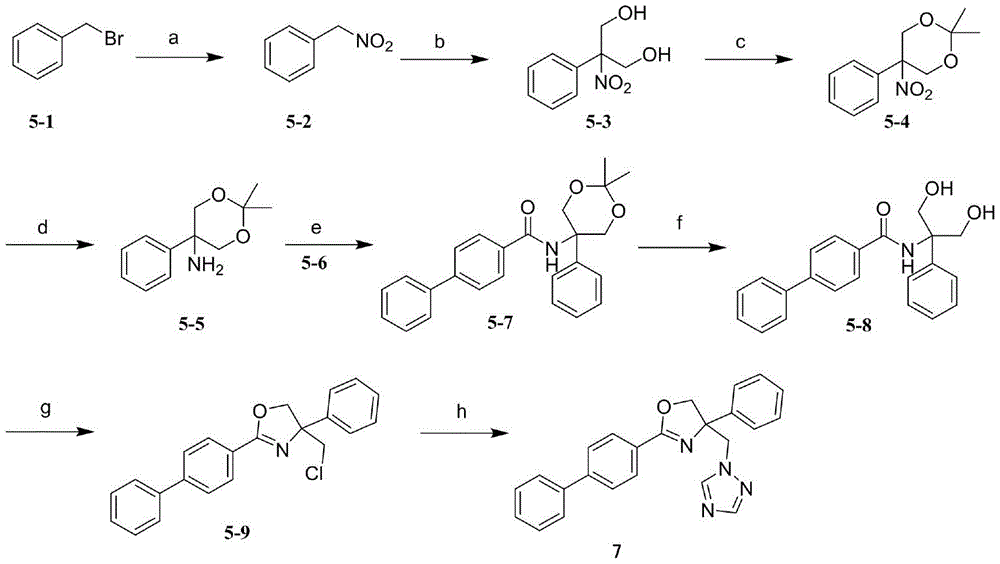
本发明的化合物及其药学上可接受的盐可由如下物质制备:(a)原料一般可以从商业来源获取的或者使用本领域技术人员所熟知的方法来制备,或根据本发明所述的方法制备,(b)可由文献记载的方法制备的已知起始物质,(c)本文的方案和实验过程中所描述的新中间体。本发明中的化合物可以通过以下反应方案及描述合成。The compounds of the present invention and their pharmaceutically acceptable salts can be prepared from the following materials: (a) The starting materials can generally be obtained from commercial sources or prepared using methods well known to those skilled in the art, or prepared according to the methods described in the present invention , (b) known starting materials that can be prepared by methods described in the literature, (c) new intermediates described in the schemes and experimental procedures herein. The compounds of the present invention can be synthesized by the following reaction schemes and descriptions.
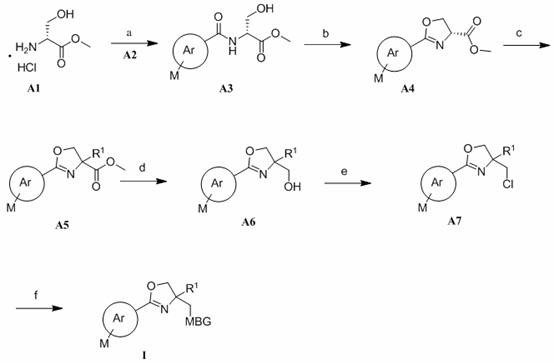
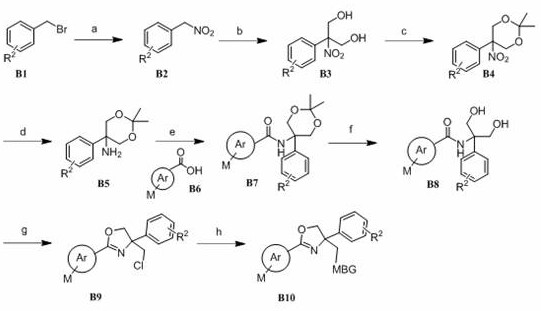
方案IScheme I
上述方案I展示出了制备化合物I的一般合成路线,其中Ar、M、R1和MBG如权利要求书所述。Scheme I above shows a general synthetic route for the preparation of compound I , wherein Ar, M, R1 and MBG are as claimed.
将D-丝氨酸甲酯盐酸盐A1与原料A2发生酰胺反应得到中间体A3,-80℃条件下,中间体A3与SOCl2发生反应得到中间体A4,中间体A4在碱性条件下与R1-X(X为卤素)发生取代反应得到中间体A5,四氢铝锂还原中间体A5的酯基得到中间体A6,A6与SOCl2发生取代反应得到中间体A7,A7与MBG发生取代反应得到目标化合物I。The amide reaction of D-serine methyl ester hydrochloride A1 and raw material A2 is carried out to obtain intermediate A3. Under the condition of -80 °C, intermediate A3 reacts with SOCl 2 to obtain intermediate A4. Intermediate A4 reacts with R under alkaline conditions. 1 -X (X is halogen) undergoes a substitution reaction to obtain intermediate A5, the ester group of intermediate A5 is reduced by lithium aluminum tetrahydrogen to obtain intermediate A6, A6 undergoes a substitution reaction with SOCl 2 to obtain intermediate A7, and A7 undergoes a substitution reaction with MBG The target compound I was obtained.
方案IIScheme II
上述方案II展示出了制备化合物R1为时,化合物的一般合成路线,其中Ar、M、R1、R2和MBG如权利要求书所述。Scheme II above shows the preparation of compound R1 as , the general synthetic route for compounds in which Ar, M, R 1 , R 2 and MBG are as described in the claims.
原料B1与AgNO2发生取代反应得到中间体B2,中间体B2与甲醛发生双羟甲基化反应得到中间体B3,中间体B3与2,2-二甲氧基丙烷发生反应得到中间体B4,中间体B4与Pd/C发生还原反应得到中间体B5,中间体B5与原料B6发生酰胺反应得到中间体B7,中间体B7在酸性条件下脱去保护基得到中间体B8,中间体B8与SOCl2发生取代反应,得到中间体B9,中间体B9与MBG发生取代反应,得到目标化合物B10。Substitution reaction between raw material B1 and AgNO 2 to obtain intermediate B2, intermediate B2 and formaldehyde undergo bismethylolation reaction to obtain intermediate B3, and intermediate B3 reacts with 2,2-dimethoxypropane to obtain intermediate B4, Intermediate B4 undergoes reduction reaction with Pd/C to obtain intermediate B5, intermediate B5 undergoes amide reaction with raw material B6 to obtain intermediate B7, intermediate B7 is deprotected under acidic conditions to obtain intermediate B8, intermediate B8 and SOCl 2. Substitution reaction occurs to obtain intermediate B9, and intermediate B9 undergoes substitution reaction with MBG to obtain target compound B10.
本发明的积极进步效果在于:本发明提供了2,4,4-三取代二氢噁唑衍生物,其制备方法、药物组合物和应用。本发明的2,4,4-三取代二氢噁唑衍生物对各种浅表和深部真菌具有良好的抗真菌活性,与现有临床应用的抗真菌药物相比,具有高效、低毒、抗真菌谱广等优点,可用于制备抗真菌药物。The positive progress effect of the present invention is that the present invention provides 2,4,4-trisubstituted dihydrooxazole derivatives, its preparation method, pharmaceutical composition and application. The 2,4,4-trisubstituted dihydrooxazole derivatives of the present invention have good antifungal activity against various superficial and deep fungi, and have high efficiency, low toxicity, It has the advantages of broad antifungal spectrum, etc., and can be used to prepare antifungal drugs.
本发明提供的实施例和制备例进一步阐明和举例说明本发明化合物及其制备方法。应当理解,下属实例和制备例的范围并不以任何方式限制本发明的范围。本发明的通式I的化合物,可按照方案I和方案II的合成方法制备得来,这些路线中应用的全部可变因数如权利要求中的定义。The examples and preparations provided herein further illustrate and illustrate the compounds of the present invention and methods for their preparation. It should be understood that the scope of the following examples and preparations do not limit the scope of the invention in any way. The compounds of general formula I of the present invention can be prepared according to the synthetic methods of Scheme I and Scheme II, all variables used in these schemes are as defined in the claims.
具体实施方式Detailed ways
不需要进一步详细说明,认为本领域熟练技术人员借助于前面的描述,可以最大程度的利用本发明。因此下面提供的实施例旨在阐述而不是限制本发明的范围。Without further elaboration, it is believed that one skilled in the art can, with the help of the preceding description, utilize the present invention to its fullest extent. The examples provided below are therefore intended to illustrate rather than limit the scope of the present invention.
原料一般可以从商业来源获取的或者使用本领域技术人员所熟知的方法来制备,或根据本发明所述的方法制备。未经特殊说明,所用试剂均为分析纯或化学纯。The starting materials can generally be obtained from commercial sources or prepared using methods well known to those skilled in the art, or prepared according to the methods described herein. Unless otherwise specified, all reagents used are of analytical or chemical grade.
化合物结构确证所用的质谱用Agilent 1100LC/MS测定。柱层析纯化产物使用的是青岛海洋化工厂生产的100-200目或者200-300目硅胶。The mass spectra used for compound structure confirmation were determined with an Agilent 1100 LC/MS. The purified product by column chromatography used 100-200 mesh or 200-300 mesh silica gel produced by Qingdao Ocean Chemical Factory.
实施例1:4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-4-甲基-4,5-二氢噁唑Example 1: 4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-methyl-4,5-dihydrooxane azole
步骤a中:将原料1-1(1.1mmol)、联苯甲酸1-2(1.1mmol)和PyBOP(1H-苯并三唑-1-基氧三吡咯烷基六氟磷酸盐)(1.2mmol)加入到DMF中,室温搅拌反应8h,TLC检测反应完毕,加水,乙酸乙酯萃取,饱和食盐水洗涤有机层,无水硫酸钠干燥过夜。滤除干燥剂,减压浓缩得中间体1-3。In step a: the raw materials 1-1 (1.1 mmol), bibenzoic acid 1-2 (1.1 mmol) and PyBOP (1H-benzotriazol-1-yloxytripyrrolidinyl hexafluorophosphate) (1.2 mmol) were combined ) was added to DMF, stirred at room temperature for 8 h, TLC detected the reaction completion, added water, extracted with ethyl acetate, washed the organic layer with saturated brine, and dried over anhydrous sodium sulfate overnight. The desiccant was filtered off and concentrated under reduced pressure to obtain Intermediate 1-3.
步骤b中:将中间体1-3(5mmol)、SOCl2(12.5mmol)和三乙胺(12.5mmol)加入二氯甲烷中,室温搅拌反应,TLC检测反应完全,蒸除有机溶剂,乙酸乙酯溶解,水洗3次,饱和NaCl洗涤有机层,无水Na2SO4干燥过夜。滤除干燥剂,减压浓缩、柱层析得中间体1-4。In step b: intermediate 1-3 (5 mmol), SOCl 2 (12.5 mmol) and triethylamine (12.5 mmol) were added to dichloromethane, the reaction was stirred at room temperature, TLC detected that the reaction was complete, the organic solvent was evaporated, ethyl acetate The ester was dissolved, washed three times with water, the organic layer was washed with saturated NaCl, and dried over anhydrous Na 2 SO 4 overnight. The desiccant was filtered off, concentrated under reduced pressure and column chromatography to obtain Intermediate 1-4.
步骤c中:将中间体1-4(5mmol)溶于无水THF中,缓慢滴加LiN(Pr-i)2(10mmol)的THF溶液,反应约1h,加入CH3I(10mmol),TLC检测反应完全,升至室温,加饱和NH4Cl溶液淬灭,减压蒸除有机溶剂,乙酸乙酯萃取,饱和NaCl洗涤有机层,无水Na2SO4干燥过夜。滤除干燥剂,减压浓缩得中间体1-5。In step c: the intermediate 1-4 (5 mmol) was dissolved in anhydrous THF, the THF solution of LiN(Pr-i) 2 (10 mmol) was slowly added dropwise, the reaction was carried out for about 1 h, CH 3 I (10 mmol) was added, TLC The reaction was detected to be complete, warmed to room temperature, quenched by adding saturated NH 4 Cl solution, the organic solvent was evaporated under reduced pressure, extracted with ethyl acetate, the organic layer was washed with saturated NaCl, and dried over anhydrous Na 2 SO 4 overnight. The drying agent was filtered off and concentrated under reduced pressure to obtain Intermediate 1-5.
步骤d中:冰浴条件下,将中间体1-5(5mmol)溶于THF中,分批加入四氢铝锂(10mmol),TLC检测反应完全,加饱和NH4Cl溶液淬灭反应,减压蒸除有机溶剂,乙酸乙酯萃取,饱和NaCl洗涤有机层,无水Na2SO4干燥过夜。滤除干燥剂,减压浓缩得中间体1-6。In step d: under ice bath condition, the intermediate 1-5 (5 mmol) was dissolved in THF, and lithium tetrahydroaluminum (10 mmol) was added in batches. TLC detected the reaction to be complete, and saturated NH 4 Cl solution was added to quench the reaction. The organic solvent was evaporated under pressure, extracted with ethyl acetate, the organic layer was washed with saturated NaCl, and dried over anhydrous Na 2 SO 4 overnight. The desiccant was filtered off and concentrated under reduced pressure to obtain Intermediate 1-6.
在步骤e中:将中间体1-6(5mmol)、SOCl2(20mmol)和三乙胺(20mmol)加入二氯甲烷中,室温搅拌反应,TLC检测反应完全,蒸除有机溶剂,乙酸乙酯溶解,水洗3次,饱和NaCl洗涤有机层,无水Na2SO4干燥过夜。滤除干燥剂,减压浓缩、柱层析得中间体1-7。In step e: intermediate 1-6 (5mmol), SOCl 2 (20mmol) and triethylamine (20mmol) were added to dichloromethane, the reaction was stirred at room temperature, TLC detected that the reaction was complete, the organic solvent was evaporated, ethyl acetate Dissolved, washed three times with water, washed the organic layer with saturated NaCl, and dried over anhydrous Na 2 SO 4 overnight. The desiccant was filtered off, concentrated under reduced pressure and column chromatography to obtain Intermediate 1-7.
在步骤f中:冰浴条件下,将咪唑(15mmol)加入DMF中,加入NaH(20mmol),搅拌30min后加入中间体1-7(5mmol),继续反应,TLC检测反应完毕,将反应液倾入水中,乙酸乙酯萃取,饱和NaCl洗涤有机层,无水Na2SO4干燥过夜。滤除干燥剂,减压浓缩、柱层析得目标化合物1。1H-NMR(400MHz,DMSO-d6)δ7.90(d,J=8.4Hz,2H),7.77(d,J=8.4Hz,2H),7.74–7.70(m,2H),7.59(s,1H),7.50(t,J=7.5Hz,2H),7.41(t,J=7.3Hz,1H),7.14(s,1H),6.81(s,1H),4.31(d,J=8.9Hz,1H),4.22–4.14(m,2H),4.12(d,J=8.9Hz,1H),1.31(s,3H).HRMS(ESI,m/z)calcdforC20H19N3O,[M+H]+,318.1601;found 318.1632.In step f: under ice bath conditions, imidazole (15mmol) was added to DMF, NaH (20mmol) was added, intermediate 1-7 (5mmol) was added after stirring for 30min, the reaction was continued, TLC detected the completion of the reaction, and the reaction solution was poured into It was poured into water, extracted with ethyl acetate, the organic layer was washed with saturated NaCl, and dried over anhydrous Na 2 SO 4 overnight. The desiccant was filtered off, concentrated under reduced pressure, and the target compound 1 was obtained by column chromatography. 1 H-NMR (400MHz, DMSO-d 6 )δ7.90(d, J=8.4Hz, 2H), 7.77(d, J=8.4Hz, 2H), 7.74-7.70(m, 2H), 7.59(s ,1H),7.50(t,J=7.5Hz,2H),7.41(t,J=7.3Hz,1H),7.14(s,1H),6.81(s,1H),4.31(d,J=8.9Hz ,1H),4.22–4.14(m,2H),4.12(d,J=8.9Hz,1H),1.31(s,3H).HRMS(ESI,m/z)calcdforC 20 H 19 N 3 O,[M +H] + ,318.1601; found 318.1632.
按照实施例1的方法,分别使用相应的卤代烷,制备得到实施例2-6。According to the method of Example 1, the corresponding haloalkanes were respectively used to prepare Examples 2-6.
实施例2:4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-4-异丙基-4,5-二氢噁唑Example 2: 4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-isopropyl-4,5-dihydro oxazole
ESI-MS[M+H]+(m/z):346.1。ESI-MS [M+H] + (m/z): 346.1.
实施例3:4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-4-环丙基-4,5-二氢噁唑Example 3: 4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-cyclopropyl-4,5-dihydro oxazole
ESI-MS[M+H]+(m/z):344.1。ESI-MS [M+H] + (m/z): 344.1.
实施例4:4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-4-苄基-4,5-二氢噁唑Example 4: 4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-benzyl-4,5-dihydrooxane azole
1H-NMR(400MHz,DMSO-d6)δ7.81(d,J=8.5Hz,2H),7.75–7.68(m,4H),7.61(s,1H),7.49(t,J=7.5Hz,2H),7.41(t,J=7.3Hz,1H),7.28–7.14(m,6H),6.81(s,1H),4.31(s,2H),4.23(d,J=9.2Hz,1H),4.16(d,J=9.2Hz,1H),3.04(d,J=13.5Hz,1H),2.91(d,J=13.5Hz,1H).HRMS(ESI,m/z)calcd for C26H23N3O,[M+H]+,394.1914;found 394.1949. 1 H-NMR (400MHz, DMSO-d 6 )δ7.81(d,J=8.5Hz,2H),7.75-7.68(m,4H),7.61(s,1H),7.49(t,J=7.5Hz ,2H),7.41(t,J=7.3Hz,1H),7.28–7.14(m,6H),6.81(s,1H),4.31(s,2H),4.23(d,J=9.2Hz,1H) ,4.16(d,J=9.2Hz,1H),3.04(d,J=13.5Hz,1H),2.91(d,J=13.5Hz,1H).HRMS(ESI,m/z)calcd for C 26 H 23 N 3 O, [M+H] + , 394.1914; found 394.1949.
实施例5:4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-5-(氟(苯基)甲基)-4,5-二氢噁唑Example 5: 4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-5-(fluoro(phenyl)methyl)- 4,5-Dihydrooxazole
HRMS(ESI,m/z)calcd for C26H22FN3O,[M+H]+,412.1820;found 412.1857.HRMS(ESI,m/z)calcd for C 26 H 22 FN 3 O,[M+H] + , 412.1820; found 412.1857.
实施例6:4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-5-(二氟(苯基)甲基)-4,5-二氢噁唑Example 6: 4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-5-(difluoro(phenyl)methyl) -4,5-Dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ7.84(d,J=8.4Hz,2H),7.76(d,J=8.3Hz,2H),7.71(d,J=7.4Hz,2H),7.69–7.62(m,3H),7.55–7.48(m,5H),7.42(t,J=7.4Hz,1H),7.14(s,1H),6.76(s,1H),4.69(d,J=10.1Hz,1H),4.59(d,J=14.2Hz,1H),4.36–4.29(m,2H).HRMS(ESI,m/z)calcd for C26H21F2N3O,[M+H]+,430.1725;found 430.1727. 1 H-NMR (600MHz, DMSO-d 6 )δ7.84(d,J=8.4Hz,2H),7.76(d,J=8.3Hz,2H),7.71(d,J=7.4Hz,2H), 7.69–7.62(m, 3H), 7.55–7.48(m, 5H), 7.42(t, J=7.4Hz, 1H), 7.14(s, 1H), 6.76(s, 1H), 4.69(d, J= 10.1Hz,1H),4.59(d,J=14.2Hz,1H),4.36–4.29(m,2H).HRMS(ESI,m/z)calcd for C 26 H 21 F 2 N 3 O,[M+ H] + , 430.1725; found 430.1727.
实施例7:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-4-苯基-4,5-二氢噁唑Example 7: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-phenyl -4,5-Dihydrooxazole
步骤a中:将AgNO2(10mmol)加入乙醚(30mL)中,室温搅拌,滴加溴苄(5mmol),避光搅拌反应5h,将反应液倾入水中,用乙醚萃取3次,减压蒸除乙醚得淡黄色油状液体,柱层析得中间体5-2。In step a: AgNO 2 (10 mmol) was added to ether (30 mL), stirred at room temperature, benzyl bromide (5 mmol) was added dropwise, stirred for 5 h in the dark, the reaction solution was poured into water, extracted with ether for 3 times, and evaporated under reduced pressure. Remove ether to obtain light yellow oily liquid, and column chromatography obtains intermediate 5-2.
步骤b中:将中间体5-2(4mmol)溶于乙醇中,加入甲醛溶液(24mmol)、NaOH(4mmol),室温搅拌反应,TLC检测反应完毕,减压蒸除有机溶剂,加水,乙酸乙酯萃取3次,合并有机层,饱和NaCl洗涤有机层,无水Na2SO4干燥过夜。滤除干燥剂,减压浓缩、柱层析得中间体5-3。In step b: the intermediate 5-2 (4 mmol) is dissolved in ethanol, formaldehyde solution (24 mmol) and NaOH (4 mmol) are added, the reaction is stirred at room temperature, the completion of the reaction is detected by TLC, the organic solvent is evaporated under reduced pressure, water is added, ethyl acetate The ester was extracted three times, and the organic layers were combined, washed with saturated NaCl, and dried over anhydrous Na 2 SO 4 overnight. The desiccant was filtered off, concentrated under reduced pressure and subjected to column chromatography to obtain Intermediate 5-3.
步骤c中:将2,2-二甲氧基丙烷(4.5mmol)加入中间体5-3(3mmol)的丙酮溶液中,加入对甲基苯磺酸(0.6mmol),室温反应,TLC检测反应完全,减压蒸除溶剂,柱层析得中间体5-4。In step c: add 2,2-dimethoxypropane (4.5mmol) to the acetone solution of intermediate 5-3 (3mmol), add p-toluenesulfonic acid (0.6mmol), react at room temperature, and detect the reaction by TLC Complete, the solvent was evaporated under reduced pressure, and the intermediate 5-4 was obtained by column chromatography.
在步骤d中:将中间体5-4(2mmol)溶于甲醇(10mL)溶液中,加入Pd/C,通入H2,室温反应,TLC检测反应完全,过滤,减压蒸除溶剂得中间体5-5。In step d: the intermediate 5-4 (2 mmol) was dissolved in methanol (10 mL) solution, Pd/C was added, H 2 was introduced, the reaction was carried out at room temperature, and the reaction was completed by TLC, filtered, and the solvent was evaporated under reduced pressure to obtain the intermediate Body 5-5.
步骤e中:将原料联苯甲酸5-6(1.1mmol)、中间体5-5(1.1mmol)和PyBOP(1.2mmol)加入DMF(10mL)中,室温搅拌反应8h,TLC检测反应完毕,加水,乙酸乙酯萃取,饱和食盐水洗涤有机层,无水硫酸钠干燥过夜。滤除干燥剂,减压浓缩得中间体5-7。In step e: the raw material bibenzoic acid 5-6 (1.1 mmol), intermediate 5-5 (1.1 mmol) and PyBOP (1.2 mmol) were added to DMF (10 mL), and the reaction was stirred at room temperature for 8 h. TLC detected the reaction and added water. , extracted with ethyl acetate, the organic layer was washed with saturated brine, and dried over anhydrous sodium sulfate overnight. The desiccant was filtered off and concentrated under reduced pressure to obtain intermediate 5-7.
步骤f中:将中间体5-7(5mmol)加入到冰乙酸/水(V:V=3:1)中,40℃反应,TLC检测反应完全,倾入水中,乙酸乙酯萃取,饱和NaCl洗涤有机层,无水Na2SO4干燥过夜。滤除干燥剂,减压浓缩、柱层析得中间体5-8。In step f: intermediate 5-7 (5 mmol) was added to glacial acetic acid/water (V:V=3:1), reacted at 40° C., TLC detected that the reaction was complete, poured into water, extracted with ethyl acetate, saturated NaCl The organic layer was washed and dried over anhydrous Na 2 SO 4 overnight. The desiccant was filtered off, concentrated under reduced pressure and column chromatography to obtain Intermediate 5-8.
步骤g中:将中间体5-8(5mmol)、SOCl2(25mmol)和三乙胺(25mmol)加入二氯甲烷中,室温搅拌反应,TLC检测反应完全,蒸除有机溶剂,乙酸乙酯溶解,水洗3次,饱和NaCl洗涤有机层,无水Na2SO4干燥过夜。滤除干燥剂,减压浓缩、柱层析得中间体5-9。In step g: intermediate 5-8 (5mmol), SOCl 2 (25mmol) and triethylamine (25mmol) are added in methylene chloride, the reaction is stirred at room temperature, TLC detects that the reaction is complete, the organic solvent is evaporated, and ethyl acetate is dissolved , washed three times with water, washed the organic layer with saturated NaCl, and dried over anhydrous Na 2 SO 4 overnight. The desiccant was filtered off, concentrated under reduced pressure and column chromatography to obtain Intermediate 5-9.
步骤h中:冰浴条件下,将唑(15mmol)加入DMF(10mL)中,加入NaH(20mmol),搅拌30min后加入中间体5-9(5mmol),继续反应,TLC检测反应完毕,将反应液倾入水中,乙酸乙酯萃取,饱和NaCl洗涤有机层,无水Na2SO4干燥过夜。滤除干燥剂,减压浓缩、柱层析得目标化合物7。1H-NMR(600MHz,DMSO-d6)δ8.32(s,1H),7.96(d,J=8.4Hz,2H),7.90(s,1H),7.79(d,J=8.3Hz,2H),7.73(d,J=7.5Hz,2H),7.53–7.49(m,4H),7.44–7.39(m,3H),7.32(t,J=7.3Hz,1H),5.13(d,J=8.9Hz,1H),4.74(d,J=14.2Hz,1H),4.61(d,J=14.1Hz,1H),4.46(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd for C24H20N4O,[M+Na]+,403.1529;found403.1563.In step h: under ice bath conditions, add azole (15mmol) to DMF (10mL), add NaH (20mmol), add intermediate 5-9 (5mmol) after stirring for 30min, continue the reaction, TLC detects the completion of the reaction, and the reaction The liquid was poured into water, extracted with ethyl acetate, the organic layer was washed with saturated NaCl, and dried over anhydrous Na 2 SO 4 overnight. The desiccant was filtered off, concentrated under reduced pressure, and the target compound 7 was obtained by column chromatography. 1 H-NMR (600MHz, DMSO-d 6 )δ8.32(s, 1H), 7.96(d, J=8.4Hz, 2H), 7.90(s, 1H), 7.79(d, J=8.3Hz, 2H) ), 7.73(d, J=7.5Hz, 2H), 7.53-7.49(m, 4H), 7.44-7.39(m, 3H), 7.32(t, J=7.3Hz, 1H), 5.13(d, J= 8.9Hz,1H),4.74(d,J=14.2Hz,1H),4.61(d,J=14.1Hz,1H),4.46(d,J=8.9Hz,1H).HRMS(ESI,m/z) calcd for C 24 H 20 N 4 O, [M+Na] + , 403.1529; found403.1563.
按照实施例7的方法,分别使用相应的原料制备得到实施例8-56。According to the method of Example 7, the corresponding raw materials were used to prepare Examples 8-56.
实施例8:4-((1H-咪唑-1-基)甲基)-2-([1,1'-联苯]-4-基)-4-苯基-4,5-二氢噁唑Example 8: 4-((1H-imidazol-1-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-phenyl-4,5-dihydrooxane azole
1H-NMR(600MHz,DMSO-d6)δ7.99(d,J=8.4Hz,2H),7.81(d,J=8.4Hz,2H),7.74(d,J=7.2Hz,2H),7.55–7.49(m,5H),7.44–7.39(m,3H),7.32(t,J=7.3Hz,1H),7.09(s,1H),6.75(s,1H),4.82(d,J=9.1Hz,1H),4.52(d,J=14.2Hz,1H),4.43(d,J=9.1Hz,1H),4.35(d,J=14.2Hz,1H).HRMS(ESI,m/z)calcd for C25H21N3O,[M+H]+,380.1757;found380.1796. 1 H-NMR (600MHz, DMSO-d 6 )δ7.99(d,J=8.4Hz,2H),7.81(d,J=8.4Hz,2H),7.74(d,J=7.2Hz,2H), 7.55-7.49(m, 5H), 7.44-7.39(m, 3H), 7.32(t, J=7.3Hz, 1H), 7.09(s, 1H), 6.75(s, 1H), 4.82(d, J= 9.1Hz,1H),4.52(d,J=14.2Hz,1H),4.43(d,J=9.1Hz,1H),4.35(d,J=14.2Hz,1H).HRMS(ESI,m/z) calcd for C 25 H 21 N 3 O, [M+H] + , 380.1757; found380.1796.
实施例9:4-((1H-四氮唑-2-基)甲基)-2-([1,1'-联苯]-4-基)-4-苯基-4,5-二氢噁唑Example 9: 4-((1H-Tetrazol-2-yl)methyl)-2-([1,1'-biphenyl]-4-yl)-4-phenyl-4,5-di Hydroxazole
1H-NMR(600MHz,DMSO-d6)δ9.26(s,1H),7.98(d,J=8.5Hz,2H),7.81(d,J=8.5Hz,2H),7.75–7.73(m,2H),7.54–7.50(m,4H),7.44–7.39(m,3H),7.33(t,J=7.3Hz,1H),5.08(d,J=14.2Hz,1H),5.03(d,J=9.2Hz,1H),4.96(d,J=14.2Hz,1H),4.49(d,J=9.2Hz,1H).HRMS(ESI,m/z)calcd for C23H19N5O,[M+Na]+,404.1482;found 404.1514. 1 H-NMR (600MHz, DMSO-d 6 )δ9.26(s,1H),7.98(d,J=8.5Hz,2H),7.81(d,J=8.5Hz,2H),7.75-7.73(m ,2H),7.54–7.50(m,4H),7.44–7.39(m,3H),7.33(t,J=7.3Hz,1H),5.08(d,J=14.2Hz,1H),5.03(d, J=9.2Hz,1H),4.96(d,J=14.2Hz,1H),4.49(d,J=9.2Hz,1H).HRMS(ESI,m/z)calcd for C 23 H 19 N 5 O, [M+Na] + , 404.1482; found 404.1514.
实施例10:3-(4-((1H-1,2,4-三氮唑-1-基)甲基)-4-苯基-4,5-二氢噁唑-2-基)-5-苯基异噁唑Example 10: 3-(4-((1H-1,2,4-triazol-1-yl)methyl)-4-phenyl-4,5-dihydrooxazol-2-yl)- 5-Phenylisoxazole
1H-NMR(600MHz,DMSO-d6)δ8.34(s,1H),8.00–7.98(m,2H),7.92(s,1H),7.59–7.56(m,4H),7.49(dd,J=8.2,1.0Hz,2H),7.41(t,J=7.6Hz,2H),7.34(t,J=7.3Hz,1H),5.19(d,J=9.1Hz,1H),4.78(d,J=14.2Hz,1H),4.67(d,J=14.2Hz,1H),4.55(d,J=9.1Hz,1H).HRMS(ESI,m/z)calcd for C21H17N5O2,[M+Na]+,394.1274;found 394.1315. 1 H-NMR (600MHz, DMSO-d 6 )δ8.34(s,1H),8.00-7.98(m,2H),7.92(s,1H),7.59-7.56(m,4H),7.49(dd, J=8.2, 1.0Hz, 2H), 7.41(t, J=7.6Hz, 2H), 7.34(t, J=7.3Hz, 1H), 5.19(d, J=9.1Hz, 1H), 4.78(d, J=14.2Hz, 1H), 4.67(d, J=14.2Hz, 1H), 4.55(d, J=9.1Hz, 1H).HRMS(ESI,m/z)calcd for C 21 H 17 N 5 O 2 ,[M+Na] + ,394.1274; found 394.1315.
实施例11:4-((1H-1,2,4-三氮唑-1-基)甲基)-4-苯基-2-(5-苯基噻吩-2-基)-4,5-二氢噁唑Example 11: 4-((1H-1,2,4-triazol-1-yl)methyl)-4-phenyl-2-(5-phenylthiophen-2-yl)-4,5 - Dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.28(s,1H),7.92(s,1H),7.77–7.74(m,2H),7.57(d,J=0.8Hz,2H),7.49–7.46(m,4H),7.40(t,J=7.7Hz,3H),7.33(t,J=7.3Hz,1H),5.12(d,J=8.8Hz,1H),4.73(d,J=14.2Hz,1H),4.60(d,J=14.2Hz,1H),4.46(d,J=8.8Hz,1H).HRMS(ESI,m/z)calcd for C22H18N4OS,[M+Na]+,409.1094;found 409.1134. 1 H-NMR (600MHz, DMSO-d 6 )δ8.28(s,1H),7.92(s,1H),7.77-7.74(m,2H),7.57(d,J=0.8Hz,2H),7.49 –7.46(m, 4H), 7.40(t, J=7.7Hz, 3H), 7.33(t, J=7.3Hz, 1H), 5.12(d, J=8.8Hz, 1H), 4.73(d, J= 14.2Hz,1H),4.60(d,J=14.2Hz,1H),4.46(d,J=8.8Hz,1H).HRMS(ESI,m/z)calcd for C 22 H 18 N 4 OS,[M +Na] + ,409.1094; found 409.1134.
实施例12:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(4-(呋喃-3-基)苯基)-4-苯基-4,5-二氢噁唑Example 12: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(4-(furan-3-yl)phenyl)-4-phenyl-4 ,5-Dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.31(d,J=12.2Hz,2H),7.88(d,J=5.0Hz,2H),7.86(s,1H),7.79(t,J=1.7Hz,1H),7.74(d,J=8.4Hz,2H),7.53–7.49(m,2H),7.39(t,J=7.7Hz,2H),7.32(t,J=7.3Hz,1H),7.06–7.02(m,1H),5.11(d,J=8.9Hz,1H),4.72(d,J=14.2Hz,1H),4.59(d,J=14.1Hz,1H),4.44(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd forC22H18N4O2,[M+Na]+,393.1322;found 393.1356. 1 H-NMR (600MHz, DMSO-d 6 )δ8.31(d,J=12.2Hz,2H),7.88(d,J=5.0Hz,2H),7.86(s,1H),7.79(t,J =1.7Hz,1H),7.74(d,J=8.4Hz,2H),7.53–7.49(m,2H),7.39(t,J=7.7Hz,2H),7.32(t,J=7.3Hz,1H) ), 7.06–7.02(m, 1H), 5.11(d, J=8.9Hz, 1H), 4.72(d, J=14.2Hz, 1H), 4.59(d, J=14.1Hz, 1H), 4.44(d , J=8.9Hz, 1H).HRMS(ESI,m/z)calcd forC 22 H 18 N 4 O 2 , [M+Na] + , 393.1322; found 393.1356.
实施例13:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(4-(噻吩-3-基)苯基)-4-苯基-4,5-二氢噁唑Example 13: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(4-(thiophen-3-yl)phenyl)-4-phenyl-4 ,5-Dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.31(s,1H),8.04(dd,J=2.9,1.3Hz,1H),7.92–7.87(m,3H),7.84(d,J=8.5Hz,2H),7.69(dd,J=5.0,2.9Hz,1H),7.63(dd,J=5.0,1.3Hz,1H),7.53–7.50(m,2H),7.39(t,J=7.7Hz,2H),7.32(t,J=7.3Hz,1H),5.11(d,J=8.9Hz,1H),4.73(d,J=14.2Hz,1H),4.60(d,J=14.1Hz,1H),4.44(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd for C22H18N4OS,[M+Na]+,409.1094;found 409.1140. 1 H-NMR (600MHz, DMSO-d 6 ) δ 8.31 (s, 1H), 8.04 (dd, J=2.9, 1.3 Hz, 1H), 7.92-7.87 (m, 3H), 7.84 (d, J= 8.5Hz, 2H), 7.69 (dd, J=5.0, 2.9Hz, 1H), 7.63 (dd, J=5.0, 1.3Hz, 1H), 7.53–7.50 (m, 2H), 7.39 (t, J=7.7 Hz, 2H), 7.32(t, J=7.3Hz, 1H), 5.11(d, J=8.9Hz, 1H), 4.73(d, J=14.2Hz, 1H), 4.60(d, J=14.1Hz, 1H), 4.44 (d, J=8.9Hz, 1H).HRMS(ESI, m/z) calcd for C 22 H 18 N 4 OS, [M+Na] + , 409.1094; found 409.1140.
实施例14:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(萘-2-基)-4-苯基-4,5-二氢噁唑Example 14: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(naphthalen-2-yl)-4-phenyl-4,5-dihydrooxane azole
1H-NMR(600MHz,DMSO-d6)δ8.43(s,1H),8.33(s,1H),8.08(d,J=7.9Hz,1H),8.03(d,J=0.9Hz,2H),8.00(d,J=8.1Hz,1H),7.88(s,1H),7.66–7.63(m,1H),7.62–7.59(m,1H),7.55(dd,J=8.2,1.1Hz,2H),7.41(t,J=7.7Hz,2H),7.33(t,J=7.3Hz,1H),5.18(d,J=8.8Hz,1H),4.76(d,J=14.2Hz,1H),4.63(d,J=14.2Hz,1H),4.50(d,J=8.8Hz,1H).HRMS(ESI,m/z)calcd for C22H18N4O,[M+Na]+,377.1373;found 377.1403. 1 H-NMR (600MHz, DMSO-d 6 )δ8.43(s, 1H), 8.33(s, 1H), 8.08(d, J=7.9Hz, 1H), 8.03(d, J=0.9Hz, 2H ),8.00(d,J=8.1Hz,1H),7.88(s,1H),7.66-7.63(m,1H),7.62-7.59(m,1H),7.55(dd,J=8.2,1.1Hz, 2H), 7.41(t, J=7.7Hz, 2H), 7.33(t, J=7.3Hz, 1H), 5.18(d, J=8.8Hz, 1H), 4.76(d, J=14.2Hz, 1H) ,4.63(d,J=14.2Hz,1H),4.50(d,J=8.8Hz,1H).HRMS(ESI,m/z)calcd for C 22 H 18 N 4 O,[M+Na] + , 377.1373; found 377.1403.
实施例15:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(苯并呋喃-2-基)-4-苯基-4,5-二氢噁唑Example 15: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(benzofuran-2-yl)-4-phenyl-4,5-di Hydroxazole
1H-NMR(600MHz,DMSO-d6)δ8.30(s,1H),7.91(s,1H),7.78–7.72(m,2H),7.53–7.48(m,4H),7.40(t,J=7.6Hz,2H),7.37–7.32(m,2H),5.17(d,J=8.9Hz,1H),4.74(d,J=14.2Hz,1H),4.63(d,J=14.2Hz,1H),4.50(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd forC20H16N4O2,[M+Na]+,367.1165;found 367.1203. 1 H-NMR (600MHz, DMSO-d 6 )δ8.30(s, 1H), 7.91(s, 1H), 7.78-7.72(m, 2H), 7.53-7.48(m, 4H), 7.40(t, J=7.6Hz, 2H), 7.37–7.32 (m, 2H), 5.17 (d, J=8.9Hz, 1H), 4.74 (d, J=14.2Hz, 1H), 4.63 (d, J=14.2Hz, 1H), 4.50(d, J=8.9Hz, 1H).HRMS(ESI,m/z)calcd forC 20 H 16 N 4 O 2 , [M+Na] + , 367.1165; found 367.1203.
实施例16:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(苯并[d]噻唑-2-基)-4-苯基-4,5-二氢噁唑Example 16: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(benzo[d]thiazol-2-yl)-4-phenyl-4, 5-Dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.33(s,1H),8.26–8.22(m,1H),8.15(dd,J=7.3,1.7Hz,1H),7.91(s,1H),7.63(pd,J=7.1,1.3Hz,2H),7.53–7.48(m,2H),7.43(t,J=7.7Hz,2H),7.35(t,J=7.3Hz,1H),5.29(d,J=9.0Hz,1H),4.81(d,J=14.2Hz,1H),4.69(d,J=14.2Hz,1H),4.62(d,J=9.0Hz,1H).HRMS(ESI,m/z)calcd for C19H15N5OS,[M+Na]+,384.0890;found 384.0923. 1 H-NMR (600MHz, DMSO-d 6 )δ8.33(s,1H),8.26-8.22(m,1H),8.15(dd,J=7.3,1.7Hz,1H),7.91(s,1H) ,7.63(pd,J=7.1,1.3Hz,2H),7.53–7.48(m,2H),7.43(t,J=7.7Hz,2H),7.35(t,J=7.3Hz,1H),5.29( d,J=9.0Hz,1H),4.81(d,J=14.2Hz,1H),4.69(d,J=14.2Hz,1H),4.62(d,J=9.0Hz,1H).HRMS(ESI, m/z) calcd for C 19 H 15 N 5 OS, [M+Na] + , 384.0890; found 384.0923.
实施例17:4-((1H-1,2,4-三氮唑-1-基)甲基)-2,4-二苯基-4,5-二氢噁唑Example 17: 4-((1H-1,2,4-triazol-1-yl)methyl)-2,4-diphenyl-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.30(s,1H),7.90–7.85(m,3H),7.58(t,J=7.4Hz,1H),7.51–7.48(m,4H),7.39(t,J=7.6Hz,2H),7.31(t,J=7.3Hz,1H),5.11(d,J=9.0Hz,1H),4.72(d,J=14.1Hz,1H),4.60(d,J=14.1Hz,1H),4.44(d,J=9.0Hz,1H).HRMS(ESI,m/z)calcd for C18H16N4O,[M+Na]+,327.1216;found 327.1248. 1 H-NMR (600MHz, DMSO-d 6 )δ8.30(s, 1H), 7.90-7.85(m, 3H), 7.58(t, J=7.4Hz, 1H), 7.51-7.48(m, 4H) ,7.39(t,J=7.6Hz,2H),7.31(t,J=7.3Hz,1H),5.11(d,J=9.0Hz,1H),4.72(d,J=14.1Hz,1H),4.60 (d, J=14.1Hz, 1H), 4.44 (d, J=9.0Hz, 1H). HRMS(ESI, m/z) calcd for C 18 H 16 N 4 O, [M+Na] + , 327.1216; found 327.1248.
实施例18:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(4-(苄氧基)苯基)-4-苯基-4,5-二氢噁唑Example 18: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(4-(benzyloxy)phenyl)-4-phenyl-4,5 - Dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.27(s,1H),7.88(s,1H),7.82(d,J=8.9Hz,2H),7.49(d,J=7.2Hz,2H),7.46(d,J=7.2Hz,2H),7.42–7.33(m,5H),7.30(t,J=7.3Hz,1H),7.10(d,J=8.9Hz,2H),5.18(s,2H),5.06(d,J=8.9Hz,1H),4.69(d,J=14.1Hz,1H),4.56(d,J=14.1Hz,1H),4.39(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd for C25H22N4O2,[M+H]+,411.1816;found 411.1856. 1 H-NMR (600MHz, DMSO-d 6 )δ8.27(s,1H),7.88(s,1H),7.82(d,J=8.9Hz,2H),7.49(d,J=7.2Hz,2H) ), 7.46(d, J=7.2Hz, 2H), 7.42–7.33(m, 5H), 7.30(t, J=7.3Hz, 1H), 7.10(d, J=8.9Hz, 2H), 5.18(s ,2H),5.06(d,J=8.9Hz,1H),4.69(d,J=14.1Hz,1H),4.56(d,J=14.1Hz,1H),4.39(d,J=8.9Hz,1H) ).HRMS(ESI,m/z)calcd for C 25 H 22 N 4 O 2 , [M+H] + , 411.1816; found 411.1856.
实施例19:N-(4-(4-((1H-1,2,4-三氮唑-1-基)甲基)-4-苯基-4,5-二氢噁唑-2-基)苯基)苯甲酰胺Example 19: N-(4-(4-((1H-1,2,4-triazol-1-yl)methyl)-4-phenyl-4,5-dihydrooxazole-2- yl)phenyl)benzamide
1H-NMR(600MHz,DMSO-d6)δ10.52(s,1H),8.30(s,1H),7.98(d,J=7.9Hz,2H),7.92(d,J=8.8Hz,2H),7.90–7.86(m,3H),7.62(t,J=7.3Hz,1H),7.56(t,J=7.5Hz,2H),7.51(d,J=7.2Hz,2H),7.39(t,J=7.7Hz,2H),7.32(t,J=7.3Hz,1H),5.09(d,J=8.9Hz,1H),4.71(d,J=14.1Hz,1H),4.59(d,J=14.1Hz,1H),4.43(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd for C25H21N5O2,[M+Na]+,446.1587;found 446.1640. 1 H-NMR (600MHz, DMSO-d 6 )δ10.52(s, 1H), 8.30(s, 1H), 7.98(d, J=7.9Hz, 2H), 7.92(d, J=8.8Hz, 2H) ), 7.90–7.86(m, 3H), 7.62(t, J=7.3Hz, 1H), 7.56(t, J=7.5Hz, 2H), 7.51(d, J=7.2Hz, 2H), 7.39(t , J=7.7Hz, 2H), 7.32(t, J=7.3Hz, 1H), 5.09(d, J=8.9Hz, 1H), 4.71(d, J=14.1Hz, 1H), 4.59(d, J =14.1Hz,1H),4.43(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd for C 25 H 21 N 5 O 2 , [M+Na] + , 446.1587; found 446.1640.
实施例20:1-(4-(4-(4-((1H-1,2,4-三氮唑-1-基)甲基)-4-苯基-4,5-二氢噁唑-2-基)苯基)哌嗪-1-基)乙酮Example 20: 1-(4-(4-(4-((1H-1,2,4-triazol-1-yl)methyl)-4-phenyl-4,5-dihydrooxazole -2-yl)phenyl)piperazin-1-yl)ethanone
1H-NMR(600MHz,DMSO-d6)δ8.25(s,1H),7.87(s,1H),7.71(d,J=9.0Hz,2H),7.50–7.47(m,2H),7.37(t,J=7.7Hz,2H),7.30(t,J=7.3Hz,1H),6.98(d,J=9.0Hz,2H),5.02(d,J=8.9Hz,1H),4.67(d,J=14.1Hz,1H),4.54(d,J=14.1Hz,1H),4.35(d,J=8.9Hz,1H),3.59–3.57(m,4H),3.35–3.31(m,4H),2.04(s,3H).HRMS(ESI,m/z)calcd forC24H26N6O2,[M+Na]+,453.2009;found 453.2050. 1 H-NMR (600MHz, DMSO-d 6 )δ8.25(s,1H),7.87(s,1H),7.71(d,J=9.0Hz,2H),7.50-7.47(m,2H),7.37 (t, J=7.7Hz, 2H), 7.30 (t, J=7.3Hz, 1H), 6.98 (d, J=9.0Hz, 2H), 5.02 (d, J=8.9Hz, 1H), 4.67 (d , J=14.1Hz, 1H), 4.54 (d, J=14.1Hz, 1H), 4.35 (d, J=8.9Hz, 1H), 3.59–3.57 (m, 4H), 3.35–3.31 (m, 4H) ,2.04(s,3H).HRMS(ESI,m/z)calcd forC 24 H 26 N 6 O 2 ,[M+Na] + ,453.2009;found 453.2050.
实施例21:2-(4-(4-((1H-1,2,4-三氮唑-1-基)甲基)-4-苯基-4,5-二氢噁唑-2-基)苯基)-5-苯基-1,3,4-噁二唑Example 21: 2-(4-(4-((1H-1,2,4-triazol-1-yl)methyl)-4-phenyl-4,5-dihydrooxazole-2- yl)phenyl)-5-phenyl-1,3,4-oxadiazole
1H-NMR(600MHz,DMSO-d6)δ8.33(s,1H),8.26(d,J=8.5Hz,2H),8.17(dd,J=8.0,1.5Hz,2H),8.11(d,J=8.5Hz,2H),7.89(s,1H),7.69–7.64(m,3H),7.54–7.51(m,2H),7.41(t,J=7.7Hz,2H),7.33(t,J=7.3Hz,1H),5.18(d,J=8.9Hz,1H),4.76(d,J=14.2Hz,1H),4.63(d,J=14.2Hz,1H),4.51(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd for C26H20N6O2,[M+Na]+,471.1540;found 471.1577. 1 H-NMR (600MHz, DMSO-d 6 )δ8.33(s, 1H), 8.26(d, J=8.5Hz, 2H), 8.17(dd, J=8.0, 1.5Hz, 2H), 8.11(d , J=8.5Hz, 2H), 7.89(s, 1H), 7.69-7.64(m, 3H), 7.54-7.51(m, 2H), 7.41(t, J=7.7Hz, 2H), 7.33(t, J=7.3Hz, 1H), 5.18 (d, J=8.9Hz, 1H), 4.76 (d, J=14.2Hz, 1H), 4.63 (d, J=14.2Hz, 1H), 4.51 (d, J= 8.9Hz, 1H).HRMS(ESI,m/z)calcd for C 26 H 20 N 6 O 2 , [M+Na] + , 471.1540; found 471.1577.
实施例22:4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(4-氟苯基)-2-(4-(呋喃-3-基)苯基)-4,5-二氢噁唑Example 22: 4-((1H-1,2,4-triazol-1-yl)methyl)-4-(4-fluorophenyl)-2-(4-(furan-3-yl) phenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.32(d,J=8.0Hz,2H),7.90–7.84(m,3H),7.79(t,J=1.7Hz,1H),7.74(d,J=8.5Hz,2H),7.55–7.52(m,2H),7.21(t,J=8.9Hz,2H),7.04(dd,J=1.8,0.8Hz,1H),5.09(d,J=9.0Hz,1H),4.72(d,J=14.1Hz,1H),4.59(d,J=14.1Hz,1H),4.43(d,J=9.0Hz,1H).HRMS(ESI,m/z)calcd for C22H17FN4O2,[M+Na]+,411.1228;found411.1264. 1 H-NMR (600MHz, DMSO-d 6 )δ8.32(d, J=8.0Hz, 2H), 7.90-7.84(m, 3H), 7.79(t, J=1.7Hz, 1H), 7.74(d , J=8.5Hz, 2H), 7.55–7.52(m, 2H), 7.21(t, J=8.9Hz, 2H), 7.04(dd, J=1.8, 0.8Hz, 1H), 5.09(d, J= 9.0Hz,1H),4.72(d,J=14.1Hz,1H),4.59(d,J=14.1Hz,1H),4.43(d,J=9.0Hz,1H).HRMS(ESI,m/z) calcd for C 22 H 17 FN 4 O 2 , [M+Na] + , 411.1228; found411.1264.
实施例23:4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(4-氯苯基)-2-(4-(呋喃-3-基)苯基)-4,5-二氢噁唑Example 23: 4-((1H-1,2,4-triazol-1-yl)methyl)-4-(4-chlorophenyl)-2-(4-(furan-3-yl) phenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):405.1。ESI-MS [M+H] + (m/z): 405.1.
实施例24:4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(2,4-二氟苯基)-2-(4-(呋喃-3-基)苯基)-4,5-二氢噁唑Example 24: 4-((1H-1,2,4-triazol-1-yl)methyl)-4-(2,4-difluorophenyl)-2-(4-(furan-3 -yl)phenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):407.1。ESI-MS [M+H] + (m/z): 407.1.
实施例25:4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(2,4-二氯苯基)-2-(4-(呋喃-3-基)苯基)-4,5-二氢噁唑Example 25: 4-((1H-1,2,4-triazol-1-yl)methyl)-4-(2,4-dichlorophenyl)-2-(4-(furan-3 -yl)phenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):439.1。ESI-MS [M+H] + (m/z): 439.1.
实施例26:4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(4-氟苯基)-2-(4-(噻吩-3-基)苯基)-4,5-二氢噁唑Example 26: 4-((1H-1,2,4-triazol-1-yl)methyl)-4-(4-fluorophenyl)-2-(4-(thiophen-3-yl) phenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):405.1。ESI-MS [M+H] + (m/z): 405.1.
实施例27:4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(4-氯苯基)-2-(4-(噻吩-3-基)苯基)-4,5-二氢噁唑Example 27: 4-((1H-1,2,4-triazol-1-yl)methyl)-4-(4-chlorophenyl)-2-(4-(thiophen-3-yl) phenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):421.1。ESI-MS[M+H] + (m/z): 421.1.
实施例28:4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(2,4-二氟苯基)-2-(4-(噻吩-3-基)苯基)-4,5-二氢噁唑Example 28: 4-((1H-1,2,4-triazol-1-yl)methyl)-4-(2,4-difluorophenyl)-2-(4-(thiophene-3 -yl)phenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):423.1。ESI-MS [M+H] + (m/z): 423.1.
实施例29:4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(2,4-二氯苯基)-2-(4-(噻吩-3-基)苯基)-4,5-二氢噁唑Example 29: 4-((1H-1,2,4-triazol-1-yl)methyl)-4-(2,4-dichlorophenyl)-2-(4-(thiophene-3 -yl)phenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):455.1。ESI-MS [M+H] + (m/z): 455.1.
实施例30:4-((1H-1,2,4-三氮唑-1-基)甲基)-4-(2,4-二氟苯基)-2-(5-(三氟甲氧基)苯并[b]噻吩-2-基)-4,5-二氢噁唑Example 30: 4-((1H-1,2,4-triazol-1-yl)methyl)-4-(2,4-difluorophenyl)-2-(5-(trifluoromethyl) Oxy)benzo[b]thiophen-2-yl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):481.1。ESI-MS[M+H] + (m/z): 481.1.
实施例31:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氟苯并[b]噻吩-2-基)-4-(4-氟苯基)-4,5-二氢噁唑Example 31: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(6-fluorobenzo[b]thiophen-2-yl)-4-(4 -Fluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.29(s,1H),8.02–7.96(m,2H),7.91(d,J=16.4Hz,2H),7.55–7.50(m,2H),7.35(td,J=9.0,2.5Hz,1H),7.23(t,J=8.9Hz,2H),5.17(d,J=8.9Hz,1H),4.75(d,J=14.2Hz,1H),4.62(d,J=14.2Hz,1H),4.50(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd for C20H14F2N4OS,[M+Na]+,419.0749;found 419.0778. 1 H-NMR (600MHz, DMSO-d 6 )δ8.29(s, 1H), 8.02-7.96(m, 2H), 7.91(d, J=16.4Hz, 2H), 7.55-7.50(m, 2H) ,7.35(td,J=9.0,2.5Hz,1H),7.23(t,J=8.9Hz,2H),5.17(d,J=8.9Hz,1H),4.75(d,J=14.2Hz,1H) ,4.62(d,J=14.2Hz,1H),4.50(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd for C 20 H 14 F 2 N 4 OS,[M+Na] + ,419.0749; found 419.0778.
实施例32:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氟苯并[b]噻吩-2-基)-4-(4-氯苯基)-4,5-二氢噁唑Example 32: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(6-fluorobenzo[b]thiophen-2-yl)-4-(4 -Chlorophenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):413.1。ESI-MS [M+H] + (m/z): 413.1.
实施例33:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氟苯并[b]噻吩-2-基)-4-(2,4-二氟苯基)-4,5-二氢噁唑Example 33: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-fluorobenzo[b]thiophen-2-yl)-4-(2 ,4-Difluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.34(s,1H),8.04–7.99(m,2H),7.97(s,1H),7.87(s,1H),7.67–7.61(m,1H),7.40–7.34(m,2H),7.13(td,J=8.5,2.5Hz,1H),5.18(dd,J=9.1,2.6Hz,1H),4.65(d,J=14.3Hz,1H),4.59(d,J=14.3Hz,1H),4.52(dd,J=9.0,1.5Hz,1H).HRMS(ESI,m/z)calcd for C20H13F3N4OS,[M+Na]+,437.0654;found 437.0688. 1 H-NMR (600MHz, DMSO-d 6 )δ8.34(s, 1H), 8.04-7.99(m, 2H), 7.97(s, 1H), 7.87(s, 1H), 7.67-7.61(m, 1H), 7.40–7.34 (m, 2H), 7.13 (td, J=8.5, 2.5Hz, 1H), 5.18 (dd, J=9.1, 2.6Hz, 1H), 4.65 (d, J=14.3Hz, 1H) ),4.59(d,J=14.3Hz,1H),4.52(dd,J=9.0,1.5Hz,1H).HRMS(ESI,m/z)calcd for C 20 H 13 F 3 N 4 OS,[M +Na] + ,437.0654; found 437.0688.
实施例34:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氟苯并[b]噻吩-2-基)-4-(3,5-二氟苯基)-4,5-二氢噁唑Example 34: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(6-fluorobenzo[b]thiophen-2-yl)-4-(3 ,5-difluorophenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):415.1。ESI-MS [M+H] + (m/z): 415.1.
实施例35:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氟苯并[b]噻吩-2-基)-4-(2,4-二氯苯基)-4,5-二氢噁唑Example 35: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(6-fluorobenzo[b]thiophen-2-yl)-4-(2 ,4-Dichlorophenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):447.1。ESI-MS [M+H] + (m/z): 447.1.
实施例36:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氯苯并[b]噻吩-2-基)-4-(4-氟苯基)-4,5-二氢噁唑Example 36: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(6-chlorobenzo[b]thiophen-2-yl)-4-(4 -Fluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.29(s,1H),8.24(d,J=1.3Hz,1H),7.97(d,J=8.6Hz,1H),7.94(s,1H),7.90(s,1H),7.54–7.49(m,3H),7.25–7.21(m,2H),5.17(d,J=8.9Hz,1H),4.75(d,J=14.2Hz,1H),4.62(d,J=14.2Hz,1H),4.51(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd for C20H14ClFN4OS,[M+Na]+,435.0453;found 435.0480. 1 H-NMR (600MHz, DMSO-d 6 )δ8.29(s,1H),8.24(d,J=1.3Hz,1H),7.97(d,J=8.6Hz,1H),7.94(s,1H) ), 7.90 (s, 1H), 7.54–7.49 (m, 3H), 7.25–7.21 (m, 2H), 5.17 (d, J=8.9Hz, 1H), 4.75 (d, J=14.2Hz, 1H) ,4.62(d,J=14.2Hz,1H),4.51(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd for C 20 H 14 ClFN 4 OS,[M+Na] + , 435.0453; found 435.0480.
实施例37:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氯苯并[b]噻吩-2-基)-4-(4-氯苯基)-4,5-二氢噁唑Example 37: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-chlorobenzo[b]thiophen-2-yl)-4-(4 -Chlorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.30(s,1H),8.24(d,J=1.9Hz,1H),7.97(d,J=8.5Hz,1H),7.95(d,J=0.4Hz,1H),7.89(s,1H),7.51–7.45(m,5H),5.17(d,J=8.9Hz,1H),4.76(d,J=14.2Hz,1H),4.63(d,J=14.2Hz,1H),4.51(d,J=9.0Hz,1H).HRMS(ESI,m/z)calcdfor C20H14Cl2N4OS,[M+Na]+,451.0158;found 451.0195. 1 H-NMR (600MHz, DMSO-d 6 )δ8.30(s, 1H), 8.24(d, J=1.9Hz, 1H), 7.97(d, J=8.5Hz, 1H), 7.95(d, J =0.4Hz,1H),7.89(s,1H),7.51–7.45(m,5H),5.17(d,J=8.9Hz,1H),4.76(d,J=14.2Hz,1H),4.63(d , J=14.2Hz, 1H), 4.51 (d, J=9.0Hz, 1H).HRMS (ESI, m/z) calcd for C 20 H 14 Cl 2 N 4 OS, [M+Na] + , 451.0158; found 451.0195.
实施例38:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氯苯并[b]噻吩-2-基)-4-(2,4-二氟苯基)-4,5-二氢噁唑Example 38: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-chlorobenzo[b]thiophen-2-yl)-4-(2 ,4-Difluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.34(s,1H),8.26(d,J=1.8Hz,1H),7.99(d,J=7.4Hz,2H),7.86(s,1H),7.66–7.60(m,1H),7.51(dd,J=8.6,1.9Hz,1H),7.40–7.34(m,1H),7.12(td,J=8.5,2.4Hz,1H),5.18(dd,J=9.1,2.5Hz,1H),4.65(d,J=14.3Hz,1H),4.59(d,J=14.3Hz,1H),4.53(dd,J=8.9,1.3Hz,1H).HRMS(ESI,m/z)calcd for C20H13ClF2N4OS,[M+Na]+,453.0359;found 453.0383. 1 H-NMR (600MHz, DMSO-d 6 )δ8.34(s, 1H), 8.26(d, J=1.8Hz, 1H), 7.99(d, J=7.4Hz, 2H), 7.86(s, 1H) ), 7.66–7.60 (m, 1H), 7.51 (dd, J=8.6, 1.9Hz, 1H), 7.40–7.34 (m, 1H), 7.12 (td, J=8.5, 2.4Hz, 1H), 5.18 ( dd, J=9.1, 2.5Hz, 1H), 4.65 (d, J=14.3Hz, 1H), 4.59 (d, J=14.3Hz, 1H), 4.53 (dd, J=8.9, 1.3Hz, 1H). HRMS (ESI, m/z) calcd for C 20 H 13 ClF 2 N 4 OS, [M+Na] + , 453.0359; found 453.0383.
实施例39:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氯苯并[b]噻吩-2-基)-4-(3,5-二氟苯基)-4,5-二氢噁唑Example 39: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(6-chlorobenzo[b]thiophen-2-yl)-4-(3 ,5-difluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.32(s,1H),8.25(d,J=1.8Hz,1H),7.98(d,J=8.0Hz,2H),7.90(s,1H),7.50(dd,J=8.6,2.0Hz,1H),7.24–7.18(m,3H),5.15(d,J=9.1Hz,1H),4.80(d,J=14.2Hz,1H),4.67(d,J=14.1Hz,1H),4.55(d,J=9.1Hz,1H).HRMS(ESI,m/z)calcd for C20H13ClF2N4OS,[M+Na]+,453.0359;found 453.0383. 1 H-NMR (600MHz, DMSO-d 6 )δ8.32(s, 1H), 8.25(d, J=1.8Hz, 1H), 7.98(d, J=8.0Hz, 2H), 7.90(s, 1H) ), 7.50(dd, J=8.6, 2.0Hz, 1H), 7.24–7.18(m, 3H), 5.15(d, J=9.1Hz, 1H), 4.80(d, J=14.2Hz, 1H), 4.67 (d, J=14.1 Hz, 1H), 4.55 (d, J=9.1 Hz, 1H). HRMS(ESI, m/z) calcd for C 20 H 13 ClF 2 N 4 OS, [M+Na] + , 453.0359; found 453.0383.
实施例40:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-氯苯并[b]噻吩-2-基)-4-(2,4-二氯苯基)-4,5-二氢噁唑Example 40: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-chlorobenzo[b]thiophen-2-yl)-4-(2 ,4-Dichlorophenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):463.1。ESI-MS [M+H] + (m/z): 463.1.
实施例41:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-溴苯并[b]噻吩-2-基)-4-(4-氟苯基)-4,5-二氢噁唑Example 41: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-bromobenzo[b]thiophen-2-yl)-4-(4 -Fluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.39(d,J=1.7Hz,1H),8.28(s,1H),7.93(s,1H),7.92–7.88(m,2H),7.62(dd,J=8.5,1.8Hz,1H),7.53–7.50(m,2H),7.23(t,J=8.9Hz,2H),5.17(d,J=8.9Hz,1H),4.75(d,J=14.2Hz,1H),4.62(d,J=14.2Hz,1H),4.51(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd for C20H14BrFN4OS,[M+Na]+,478.9948;found 480.9957. 1 H-NMR (600MHz, DMSO-d 6 )δ8.39(d, J=1.7Hz, 1H), 8.28(s, 1H), 7.93(s, 1H), 7.92-7.88(m, 2H), 7.62 (dd, J=8.5, 1.8Hz, 1H), 7.53–7.50(m, 2H), 7.23(t, J=8.9Hz, 2H), 5.17(d, J=8.9Hz, 1H), 4.75(d, J=14.2Hz,1H),4.62(d,J=14.2Hz,1H),4.51(d,J=8.9Hz,1H).HRMS(ESI,m/z)calcd for C 20 H 14 BrFN 4 OS, [M+Na] + ,478.9948; found 480.9957.
实施例42:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-溴苯并[b]噻吩-2-基)-4-(4-氯苯基)-4,5-二氢噁唑Example 42: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-bromobenzo[b]thiophen-2-yl)-4-(4 -Chlorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.40–8.37(m,1H),8.30(s,1H),7.94(s,1H),7.92–7.89(m,2H),7.62(dd,J=8.5,1.8Hz,1H),7.50(d,J=8.7Hz,2H),7.46(d,J=8.7Hz,2H),5.17(d,J=8.9Hz,1H),4.76(d,J=14.2Hz,1H),4.63(d,J=14.2Hz,1H),4.51(d,J=8.9Hz,1H).ESI-MS[M+H]+(m/z):473.1。 1 H-NMR (600MHz, DMSO-d 6 )δ8.40-8.37(m,1H),8.30(s,1H),7.94(s,1H),7.92-7.89(m,2H),7.62(dd, J=8.5, 1.8Hz, 1H), 7.50(d, J=8.7Hz, 2H), 7.46(d, J=8.7Hz, 2H), 5.17(d, J=8.9Hz, 1H), 4.76(d, J=14.2Hz, 1H), 4.63 (d, J=14.2Hz, 1H), 4.51 (d, J=8.9Hz, 1H). ESI-MS [M+H] + (m/z): 473.1.
实施例43:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-溴苯并[b]噻吩-2-基)-4-(2,4-二氟苯基)-4,5-二氢噁唑Example 43: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-bromobenzo[b]thiophen-2-yl)-4-(2 ,4-Difluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.40(d,J=1.7Hz,1H),8.34(s,1H),7.97(s,1H),7.92(d,J=8.5Hz,1H),7.86(s,1H),7.65–7.61(m,2H),7.39–7.35(m,1H),7.12(td,J=8.5,2.5Hz,1H),5.18(dd,J=9.1,2.6Hz,1H),4.65(d,J=14.3Hz,1H),4.59(d,J=14.3Hz,1H),4.53(dd,J=9.0,1.5Hz,1H).HRMS(ESI,m/z)calcd for C20H13BrF2N4OS,[M+Na]+,496.9854;found 498.9869. 1 H-NMR (600MHz, DMSO-d 6 )δ8.40(d, J=1.7Hz, 1H), 8.34(s, 1H), 7.97(s, 1H), 7.92(d, J=8.5Hz, 1H) ), 7.86 (s, 1H), 7.65–7.61 (m, 2H), 7.39–7.35 (m, 1H), 7.12 (td, J=8.5, 2.5Hz, 1H), 5.18 (dd, J=9.1, 2.6 Hz,1H),4.65(d,J=14.3Hz,1H),4.59(d,J=14.3Hz,1H),4.53(dd,J=9.0,1.5Hz,1H).HRMS(ESI,m/z )calcd for C 20 H 13 BrF 2 N 4 OS, [M+Na] + , 496.9854; found 498.9869.
实施例44:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-溴苯并[b]噻吩-2-基)-4-(3,5-二氟苯基)-4,5-二氢噁唑Example 44: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(6-bromobenzo[b]thiophen-2-yl)-4-(3 ,5-difluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.39(s,1H),8.32(s,1H),7.97(s,1H),7.93–7.88(m,2H),7.64–7.59(m,1H),7.25–7.18(m,3H),5.15(d,J=9.1Hz,1H),4.80(d,J=14.2Hz,1H),4.67(d,J=14.1Hz,1H),4.55(d,J=9.1Hz,1H).HRMS(ESI,m/z)calcd for C20H13BrF2N4OS,[M+Na]+,496.9854;found 498.9869. 1 H-NMR (600MHz, DMSO-d 6 )δ8.39(s,1H),8.32(s,1H),7.97(s,1H),7.93-7.88(m,2H),7.64-7.59(m, 1H), 7.25–7.18(m, 3H), 5.15(d, J=9.1Hz, 1H), 4.80(d, J=14.2Hz, 1H), 4.67(d, J=14.1Hz, 1H), 4.55( d, J=9.1 Hz, 1 H). HRMS (ESI, m/z) calcd for C 20 H 13 BrF 2 N 4 OS, [M+Na] + , 496.9854; found 498.9869.
实施例45:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(6-溴苯并[b]噻吩-2-基)-4-(2,4-二氯苯基)-4,5-二氢噁唑Example 45: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(6-bromobenzo[b]thiophen-2-yl)-4-(2 ,4-Dichlorophenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):507.1。ESI-MS [M+H] + (m/z): 507.1.
实施例46:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(2'-氟-[1,1'-联苯]-4-基)-4-(4-氟苯基)-4,5-二氢噁唑Example 46: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(2'-fluoro-[1,1'-biphenyl]-4-yl) -4-(4-Fluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.34(s,1H),7.99–7.96(m,2H),7.90(s,1H),7.68(dd,J=8.3,1.4Hz,2H),7.59(td,J=7.9,1.6Hz,1H),7.57–7.54(m,2H),7.50–7.46(m,1H),7.37–7.33(m,2H),7.23(t,J=8.9Hz,2H),5.13(d,J=9.0Hz,1H),4.74(d,J=14.2Hz,1H),4.61(d,J=14.1Hz,1H),4.46(d,J=9.0Hz,1H).HRMS(ESI,m/z)calcd for C24H18F2N4O,[M+Na]+,439.1341;found 439.1382. 1 H-NMR (600MHz, DMSO-d 6 )δ8.34(s,1H),7.99-7.96(m,2H),7.90(s,1H),7.68(dd,J=8.3,1.4Hz,2H) ,7.59(td,J=7.9,1.6Hz,1H),7.57-7.54(m,2H),7.50-7.46(m,1H),7.37-7.33(m,2H),7.23(t,J=8.9Hz ,2H),5.13(d,J=9.0Hz,1H),4.74(d,J=14.2Hz,1H),4.61(d,J=14.1Hz,1H),4.46(d,J=9.0Hz,1H) ).HRMS(ESI,m/z)calcd for C 24 H 18 F 2 N 4 O,[M+Na] + , 439.1341; found 439.1382.
实施例47:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(2'-氟-[1,1'-联苯]-4-基)-4-(4-氯苯基)-4,5-二氢噁唑Example 47: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(2'-fluoro-[1,1'-biphenyl]-4-yl) -4-(4-Chlorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.35(s,1H),7.98(d,J=8.4Hz,2H),7.90(s,1H),7.68(dd,J=8.3,1.4Hz,2H),7.61–7.58(m,1H),7.54(d,J=8.6Hz,2H),7.50–7.47(m,1H),7.46(d,J=8.6Hz,2H),7.34(dd,J=10.7,4.4Hz,2H),5.12(d,J=9.0Hz,1H),4.75(d,J=14.2Hz,1H),4.62(d,J=14.1Hz,1H),4.45(d,J=9.0Hz,1H).HRMS(ESI,m/z)calcd forC24H18ClFN4O,[M+Na]+,455.1045;found 455.1093. 1 H-NMR (600MHz, DMSO-d 6 )δ8.35(s,1H),7.98(d,J=8.4Hz,2H),7.90(s,1H),7.68(dd,J=8.3,1.4Hz ,2H),7.61–7.58(m,1H),7.54(d,J=8.6Hz,2H),7.50–7.47(m,1H),7.46(d,J=8.6Hz,2H),7.34(dd, J=10.7, 4.4Hz, 2H), 5.12(d, J=9.0Hz, 1H), 4.75(d, J=14.2Hz, 1H), 4.62(d, J=14.1Hz, 1H), 4.45(d, J=9.0Hz, 1H). HRMS (ESI, m/z) calcd for C 24 H 18 ClFN 4 O, [M+Na] + , 455.1045; found 455.1093.
实施例48:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(2'-氟-[1,1'-联苯]-4-基)-4-(2,4-二氟苯基)-4,5-二氢噁唑Example 48: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(2'-fluoro-[1,1'-biphenyl]-4-yl) -4-(2,4-Difluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.39(s,1H),8.00(d,J=8.5Hz,2H),7.87(s,1H),7.72–7.68(m,3H),7.60(td,J=7.9,1.6Hz,1H),7.50–7.46(m,1H),7.39–7.33(m,3H),7.11(td,J=8.5,2.5Hz,1H),5.15(dd,J=9.2,2.5Hz,1H),4.64(d,J=14.3Hz,1H),4.58(d,J=14.3Hz,1H),4.48(dd,J=9.1,1.6Hz,1H).HRMS(ESI,m/z)calcd for C24H17F3N4O,[M+Na]+,457.1247;found 457.1282. 1 H-NMR (600MHz, DMSO-d 6 )δ8.39(s, 1H), 8.00(d, J=8.5Hz, 2H), 7.87(s, 1H), 7.72-7.68(m, 3H), 7.60 (td, J=7.9, 1.6Hz, 1H), 7.50–7.46 (m, 1H), 7.39–7.33 (m, 3H), 7.11 (td, J=8.5, 2.5Hz, 1H), 5.15 (dd, J =9.2,2.5Hz,1H),4.64(d,J=14.3Hz,1H),4.58(d,J=14.3Hz,1H),4.48(dd,J=9.1,1.6Hz,1H).HRMS(ESI ,m/z)calcd for C 24 H 17 F 3 N 4 O,[M+Na] + ,457.1247; found 457.1282.
实施例49:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(2'-氟-[1,1'-联苯]-4-基)-4-(3,5-二氟苯基)-4,5-二氢噁唑Example 49: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(2'-fluoro-[1,1'-biphenyl]-4-yl) -4-(3,5-Difluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.37(s,1H),7.99(d,J=8.5Hz,2H),7.90(s,1H),7.69(dd,J=8.3,1.4Hz,2H),7.59(td,J=7.9,1.6Hz,1H),7.50–7.46(m,1H),7.38–7.33(m,2H),7.26(dd,J=8.6,2.2Hz,2H),7.21(tt,J=9.1,2.3Hz,1H),5.11(d,J=9.2Hz,1H),4.79(d,J=14.2Hz,1H),4.66(d,J=14.1Hz,1H),4.50(d,J=9.2Hz,1H).HRMS(ESI,m/z)calcd for C24H17F3N4O,[M+Na]+,457.1247;found 457.1281. 1 H-NMR (600MHz, DMSO-d 6 )δ8.37(s,1H),7.99(d,J=8.5Hz,2H),7.90(s,1H),7.69(dd,J=8.3,1.4Hz ,2H),7.59(td,J=7.9,1.6Hz,1H),7.50-7.46(m,1H),7.38-7.33(m,2H),7.26(dd,J=8.6,2.2Hz,2H), 7.21(tt,J=9.1,2.3Hz,1H),5.11(d,J=9.2Hz,1H),4.79(d,J=14.2Hz,1H),4.66(d,J=14.1Hz,1H), 4.50(d, J=9.2Hz, 1H).HRMS(ESI,m/z)calcd for C 24 H 17 F 3 N 4 O, [M+Na] + , 457.1247; found 457.1281.
实施例50:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(2'-氟-[1,1'-联苯]-4-基)-4-(2,4-二氯苯基)-4,5-二氢噁唑Example 50: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(2'-fluoro-[1,1'-biphenyl]-4-yl) -4-(2,4-Dichlorophenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):467.1。ESI-MS [M+H] + (m/z): 467.1.
实施例51:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(3'-氟-[1,1'-联苯]-4-基)-4-(4-氟苯基)-4,5-二氢噁唑Example 51: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(3'-fluoro-[1,1'-biphenyl]-4-yl) -4-(4-Fluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.33(s,1H),7.96(d,J=8.5Hz,2H),7.89(s,1H),7.84(d,J=8.5Hz,2H),7.62–7.59(m,2H),7.57–7.53(m,3H),7.28–7.20(m,3H),5.13(d,J=9.0Hz,1H),4.74(d,J=14.2Hz,1H),4.61(d,J=14.1Hz,1H),4.45(d,J=9.0Hz,1H).HRMS(ESI,m/z)calcd for C24H18F2N4O,[M+Na]+,439.1341;found 439.1390. 1 H-NMR (600MHz, DMSO-d 6 )δ8.33(s, 1H), 7.96(d, J=8.5Hz, 2H), 7.89(s, 1H), 7.84(d, J=8.5Hz, 2H) ), 7.62–7.59 (m, 2H), 7.57–7.53 (m, 3H), 7.28–7.20 (m, 3H), 5.13 (d, J=9.0Hz, 1H), 4.74 (d, J=14.2Hz, 1H),4.61(d,J=14.1Hz,1H),4.45(d,J=9.0Hz,1H).HRMS(ESI,m/z)calcd for C 24 H 18 F 2 N 4 O,[M+ Na] + ,439.1341; found 439.1390.
实施例52:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(3'-氟-[1,1'-联苯]-4-基)-4-(4-氯苯基)-4,5-二氢噁唑Example 52: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(3'-fluoro-[1,1'-biphenyl]-4-yl) -4-(4-Chlorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.34(s,1H),7.96(d,J=8.4Hz,2H),7.89(s,1H),7.84(d,J=8.4Hz,2H),7.61–7.59(m,2H),7.55–7.52(m,3H),7.46(d,J=8.6Hz,2H),7.26(td,J=8.1,1.5Hz,1H),5.12(d,J=9.0Hz,1H),4.74(d,J=14.2Hz,1H),4.62(d,J=14.1Hz,1H),4.45(d,J=9.0Hz,1H).HRMS(ESI,m/z)calcd for C24H18ClFN4O,[M+Na]+,455.1045;found 455.1083. 1 H-NMR (600MHz, DMSO-d 6 )δ8.34(s, 1H), 7.96(d, J=8.4Hz, 2H), 7.89(s, 1H), 7.84(d, J=8.4Hz, 2H) ), 7.61–7.59 (m, 2H), 7.55–7.52 (m, 3H), 7.46 (d, J=8.6Hz, 2H), 7.26 (td, J=8.1, 1.5Hz, 1H), 5.12 (d, J=9.0Hz,1H),4.74(d,J=14.2Hz,1H),4.62(d,J=14.1Hz,1H),4.45(d,J=9.0Hz,1H).HRMS(ESI,m/ z) calcd for C 24 H 18 ClFN 4 O, [M+Na] + , 455.1045; found 455.1083.
实施例53:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(3'-氟-[1,1'-联苯]-4-基)-4-(2,4-二氟苯基)-4,5-二氢噁唑Example 53: 4-((1H-1,2,4-Triazol-1-yl)methyl)-2-(3'-fluoro-[1,1'-biphenyl]-4-yl) -4-(2,4-Difluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.38(s,1H),7.98(d,J=8.5Hz,2H),7.88–7.84(m,3H),7.71(td,J=8.7,6.8Hz,1H),7.63–7.60(m,2H),7.55(td,J=8.1,6.2Hz,1H),7.40–7.35(m,1H),7.29–7.25(m,1H),7.11(td,J=8.5,2.5Hz,1H),5.14(dd,J=9.1,2.5Hz,1H),4.64(d,J=14.3Hz,1H),4.58(d,J=14.3Hz,1H),4.47(dd,J=9.1,1.6Hz,1H).HRMS(ESI,m/z)calcd for C24H17F3N4O,[M+Na]+,457.1247;found 457.1286. 1 H-NMR (600MHz, DMSO-d 6 )δ8.38(s, 1H), 7.98(d, J=8.5Hz, 2H), 7.88-7.84(m, 3H), 7.71(td, J=8.7, 6.8Hz, 1H), 7.63–7.60 (m, 2H), 7.55 (td, J=8.1, 6.2Hz, 1H), 7.40–7.35 (m, 1H), 7.29–7.25 (m, 1H), 7.11 (td , J=8.5, 2.5Hz, 1H), 5.14 (dd, J=9.1, 2.5Hz, 1H), 4.64 (d, J=14.3Hz, 1H), 4.58 (d, J=14.3Hz, 1H), 4.47 (dd, J=9.1, 1.6 Hz, 1H). HRMS(ESI, m/z) calcd for C 24 H 17 F 3 N 4 O, [M+Na] + , 457.1247; found 457.1286.
实施例54:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(3'-氟-[1,1'-联苯]-4-基)-4-(3,5-二氟苯基)-4,5-二氢噁唑Example 54: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(3'-fluoro-[1,1'-biphenyl]-4-yl) -4-(3,5-Difluorophenyl)-4,5-dihydrooxazole
1H-NMR(600MHz,DMSO-d6)δ8.37(s,1H),7.97(d,J=8.5Hz,2H),7.90(s,1H),7.84(d,J=8.5Hz,2H),7.62–7.59(m,2H),7.57–7.53(m,1H),7.29–7.24(m,3H),7.23–7.19(m,1H),5.11(d,J=9.2Hz,1H),4.79(d,J=14.2Hz,1H),4.65(d,J=14.1Hz,1H),4.50(d,J=9.2Hz,1H).HRMS(ESI,m/z)calcd for C24H17F3N4O,[M+Na]+,457.1247;found 457.1279. 1 H-NMR (600MHz, DMSO-d 6 )δ8.37(s, 1H), 7.97(d, J=8.5Hz, 2H), 7.90(s, 1H), 7.84(d, J=8.5Hz, 2H) ), 7.62–7.59 (m, 2H), 7.57–7.53 (m, 1H), 7.29–7.24 (m, 3H), 7.23–7.19 (m, 1H), 5.11 (d, J=9.2Hz, 1H), 4.79(d,J=14.2Hz,1H),4.65(d,J=14.1Hz,1H),4.50(d,J=9.2Hz,1H).HRMS(ESI,m/z)calcd for C 24 H 17 F 3 N 4 O, [M+Na] + , 457.1247; found 457.1279.
实施例55:4-((1H-1,2,4-三氮唑-1-基)甲基)-2-(3'-氟-[1,1'-联苯]-4-基)-4-(2,4-二氟苯基)-4,5-二氢噁唑Example 55: 4-((1H-1,2,4-triazol-1-yl)methyl)-2-(3'-fluoro-[1,1'-biphenyl]-4-yl) -4-(2,4-Difluorophenyl)-4,5-dihydrooxazole
ESI-MS[M+H]+(m/z):467.1。ESI-MS [M+H] + (m/z): 467.1.
实施例56:4-((1H-四氮唑-1-基)甲基)-2-(6-氯苯并[b]噻吩-2-基)-4-(4-氟苯基)-4,5-二氢噁唑Example 56: 4-((1H-Tetrazol-1-yl)methyl)-2-(6-chlorobenzo[b]thiophen-2-yl)-4-(4-fluorophenyl)- 4,5-Dihydrooxazole
1H NMR(600MHz,DMSO)δ9.19(s,1H),8.26(d,J=1.9Hz,1H),7.99(t,J=4.3Hz,2H),7.56–7.47(m,3H),7.24(t,J=8.9Hz,2H),5.12–5.06(m,2H),4.97(d,J=14.2Hz,1H),4.54(d,J=9.2Hz,1H).ESI-MS[M+H]+(m/z):414.1。 1 H NMR(600MHz,DMSO)δ9.19(s,1H),8.26(d,J=1.9Hz,1H),7.99(t,J=4.3Hz,2H),7.56-7.47(m,3H), 7.24(t,J=8.9Hz,2H),5.12-5.06(m,2H),4.97(d,J=14.2Hz,1H),4.54(d,J=9.2Hz,1H).ESI-MS[M +H] + (m/z): 414.1.
本发明部分产物的药理研究。Pharmacological studies of some products of the present invention.
实验方法:参考常规的体外抑菌试验方法(Reference method for brothdilution antifungal susceptibility testing of yeasts and filamentous fungi;Approved Standard M27-A3 and M38-A2)。Experimental method: Refer to the conventional in vitro antibacterial test method (Reference method for brothdilution antifungal susceptibility testing of yeasts and filamentous fungi; Approved Standard M27-A3 and M38-A2).
实验材料和方法:Experimental materials and methods:
(1)实验菌株:(1) Experimental strains:
本实验选用了以下5种常见的人体致病标准真菌菌株作为筛选对象,真菌菌株由沈阳药科大学提供。In this experiment, the following five common human pathogenic standard fungal strains were selected as screening objects, and the fungal strains were provided by Shenyang Pharmaceutical University.
表1实验所用菌种及其编号Table 1 Bacteria used in the experiment and their numbers
(2)试验方法:(2) Test method:
RPMI-1640培养基的配制:RPMI-1640 10g,NaHCO3 2.0g,三氮吗啡琳丙磺酸(sigma)34.5g,加800mL无菌蒸馏水溶解,lmol/L NaOH调整pH至7.0后,定容至1000mL,0.22μm微孔滤膜过滤除菌后放置4℃保存备用。Preparation of RPMI-1640 medium: RPMI-1640 10g, NaHCO 3 2.0g, sigma 34.5g, add 800mL sterile distilled water to dissolve, adjust the pH to 7.0 with 1mol/L NaOH, and make up to volume to 1000mL, sterilized by filtration with a 0.22μm microporous membrane, and stored at 4°C for later use.
丝状真菌菌悬液的制备:丝状真菌(红色毛癣菌、疵状毛癣菌、石膏样小抱子菌和烟曲霉菌)等连续两次传代接种于沙氏培养基平板,在35℃培养箱中培养48h,菌落上加入0.85%生理盐水5mL,制备菌液。用分光光度计调整菌液浓度,A值调整至0.3-0.5;再用培养基稀释50倍作为接种菌悬液。Preparation of filamentous fungal suspension: Filamentous fungi (Trichophyton rubrum, Trichophyton blemishes, Microsporum gypsum and Aspergillus fumigatus) etc. were successively inoculated on Sabouraud medium plates for two consecutive passages, and were incubated at 35 Cultivate in an incubator for 48 hours, and add 5 mL of 0.85% physiological saline to the colony to prepare a bacterial solution. Adjust the concentration of the bacterial solution with a spectrophotometer, and adjust the A value to 0.3-0.5; then dilute it 50 times with the medium as the inoculated bacterial suspension.
球状真菌菌悬液的制备:球状菌(白色念珠菌、热带假丝酵母菌、光滑假丝酵母菌和新生隐球菌)。将活化后的菌株用分区划线法接种于沙氏固体培养基平板上,于32℃恒温培养2-3天,取适量单菌落接入含l0 mL 0.85%无菌生理盐水的三角瓶中,震荡15分钟,用灭菌枪头取少量菌液于血细胞计数板上,显微镜下计数。加RPMI-1640培养基稀释,使最终菌悬液的浓度为1x106个/mL。Preparation of Spherical Fungal Suspension: Spheroids (C. albicans, Candida tropicalis, Candida glabrata and Cryptococcus neoformans). The activated strains were inoculated on Sabouraud's solid medium plate by partition and streak method, cultured at 32°C for 2-3 days, and an appropriate amount of single colony was taken into a conical flask containing 10 mL of 0.85% sterile saline. Shake for 15 minutes, use a sterilized pipette tip to take a small amount of bacterial solution on a hemocytometer, and count under a microscope. Add RPMI-1640 medium to dilute so that the final bacterial suspension concentration is 1×10 6 cells/mL.
药液制备:称取上述化学合成药各6.40mg,依次加入l.0mL二甲基亚砜(DMSO),l.0mL吐温-20和8.0mL灭菌蒸馏水,混匀。配成药液浓度为0.64mg/mL。以相同方法配制阳性对照药氟康唑、伏立康唑。Liquid preparation: Weigh 6.40 mg of each of the above chemically synthesized drugs, add 1.0 mL of dimethyl sulfoxide (DMSO), 1.0 mL of Tween-20 and 8.0 mL of sterile distilled water in sequence, and mix well. The concentration of the drug solution is 0.64mg/mL. The positive control drugs, fluconazole and voriconazole, were prepared in the same way.
接种:第一步,加RPMI-1640培养基:每行的第1孔加入180μL RPMI-1640培养基,2-11孔加入100μL RPMI-1640培养基,12孔加入200μL RPMI-1640培养基。第二步,加药样:向第1孔中加入20μL待测药液,用移液枪混匀后吸取100μL至2孔,依次进行2倍稀释至第10孔后混匀弃去100μL。第三步,加菌悬液:向1-11孔中各加100μL接种菌悬液。第11孔为生长对照,第12孔为空白培养基对照。阳性对照药物不设空白药物对照,即从第1孔开始做倍比梯度稀释直至第10孔,测试浓度(μg/mL)范围32、16、8、4、2、1、0、5、0.25、0.125、0.0625。Inoculation: Step 1, add RPMI-1640 medium: add 180 μL RPMI-1640 medium to the first well of each row, 100 μL RPMI-1640 medium to wells 2-11, and 200 μL RPMI-1640 medium to 12 wells. The second step, adding the drug sample: add 20 μL of the drug to be tested to the first well, mix with a pipette, and draw 100 μL to the second well, and then dilute it 2-fold to the tenth well, mix well and discard 100 μL. The third step, adding bacterial suspension: add 100 μL of inoculating bacterial suspension to each of wells 1-11. Well 11 is a growth control, and well 12 is a blank medium control. There is no blank drug control for the positive control drug, that is, the doubling gradient dilution from the first well to the 10th well, the test concentration (μg/mL) range is 32, 16, 8, 4, 2, 1, 0, 5, 0.25 , 0.125, 0.0625.
培养和检测:以空白对照无菌生长,阳性对照生长良好作为判断试验操作是否合格的标准。每板测试8个样品,每个菌均设置阳性药物对照。待测药物稀释法同上。Cultivation and detection: Aseptic growth of blank control and good growth of positive control are the criteria for judging whether the test operation is qualified. Eight samples were tested per plate, and positive drug controls were set for each bacteria. The dilution method of the drug to be tested is the same as above.
表2实例化合物最低抑菌浓度((MIC,μg/ml)Table 2 Example compound minimum inhibitory concentration ((MIC, μg/ml)
从上述试验结果可以清楚地看出,本发明所要保护的通式I的化合物及其盐类具有良好的抗真菌活性,多个化合物的抗真菌活性强于对照药,与现有的抗真菌药物相比,具有结构新颖、低毒、高效、广谱等优点,因此本发明的化合物具有很好的工业应用前景。It can be clearly seen from the above test results that the compounds of the general formula I to be protected by the present invention and their salts have good antifungal activities, and the antifungal activities of many compounds are stronger than those of the control drugs, which are in line with the existing antifungal drugs. In comparison, it has the advantages of novel structure, low toxicity, high efficiency, broad spectrum, etc. Therefore, the compound of the present invention has a good industrial application prospect.
本发明中通式I的化合物可单独施用,但通常是和药用载体混合物给予,所述药用载体的选择要根据所需用药途径和标准药物实践,下面分别用该类化合物的各种药物剂型,例如片剂、胶囊剂、注射剂、气雾剂、栓剂、膜剂、滴丸剂、外用搽剂和软膏剂的制备方法,说明其在制药领域中的新应用。The compounds of general formula I in the present invention can be administered alone, but are usually administered in admixture with a pharmaceutically acceptable carrier. The choice of the pharmaceutically acceptable carrier depends on the desired route of administration and standard pharmaceutical practice. Various drugs of this type of compound are used below. The preparation methods of dosage forms, such as tablets, capsules, injections, aerosols, suppositories, films, drop pills, external liniments and ointments, illustrate their new applications in the pharmaceutical field.
实施例17:片剂。Example 17: Tablets.
用含有权利要求1中化合物的化合物(以实施例31化合物为例)10g,按照药剂学一般压片法加辅料20g混匀后,压制成100片,每片重300mg。Using 10 g of the compound containing the compound in claim 1 (taking the compound of Example 31 as an example), adding 20 g of auxiliary materials according to the general pharmaceutical tableting method, and mixing, then compressed into 100 tablets, each weighing 300 mg.
实施例18:胶囊剂。Example 18: Capsules.
用含有权利要求1中化合物的化合物(以实施例31化合物为例)10g,按照药剂学胶囊剂的要求将辅料20g混匀后,装入空心胶囊,每个胶囊重300mg。Using 10 g of the compound containing the compound in claim 1 (taking the compound of Example 31 as an example), mix 20 g of the auxiliary materials according to the requirements of pharmaceutical capsules, and then put them into hollow capsules, each capsule weighing 300 mg.
实施例19:注射剂。Example 19: Injection.
用含有权利要求1中化合物的化合物(以实施例31化合物为例)10g,按照药剂学常规方法,进行活性炭吸附,经0.65μm微孔滤膜过滤后,填入氮气罐制成水针制剂,每只装2mL,共灌装100瓶。Using 10 g of the compound containing the compound in claim 1 (taking the compound of Example 31 as an example), according to the conventional method of pharmacy, carry out activated carbon adsorption, filter through a 0.65 μm microporous membrane, fill in a nitrogen tank to prepare a water injection preparation, Each bottle contains 2mL, and a total of 100 bottles are filled.
实施例20:气雾剂。Example 20: Aerosol.
用含有权利要求1中化合物的化合物(以实施例31化合物为例)10g,用适量丙二醇溶解后,加入蒸馏水及其他辐料后,制成500mL的澄清溶液即得。10 g of the compound containing the compound in claim 1 (take the compound of Example 31 as an example) is dissolved in an appropriate amount of propylene glycol, and distilled water and other materials are added to prepare a 500 mL clear solution.
实施例21:栓剂。Example 21: Suppositories.
用含有权利要求1中化合物的化合物(以实施例31化合物为例)10g,将之研细加入甘油适量,研匀后加入已熔化的甘油明胶,研磨均匀,倾入已涂润滑剂的模型中,制得栓剂50颗。Use 10 g of the compound containing the compound in claim 1 (taking the compound of Example 31 as an example), grind it into a fine amount and add an appropriate amount of glycerol, grind it evenly, add the melted glycerin gelatin, grind it evenly, and pour it into a model that has been coated with lubricant , 50 suppositories were prepared.
实施例22:膜剂。Example 22: Film formulation.
用含有权利要求1中化合物的化合物(以实施例31化合物为例)10g,将聚乙烯醇、药用甘油、水等搅拌膨胀后加热溶解,80目筛网过滤,再将实施例18化合物加入到滤液中搅拌溶解,涂膜机制膜100片。With 10 g of the compound containing the compound in claim 1 (taking the compound of Example 31 as an example), polyvinyl alcohol, medicinal glycerin, water, etc. are stirred and expanded, heated and dissolved, filtered through an 80-mesh sieve, and then the compound of Example 18 is added. Stir in the filtrate to dissolve, and coat 100 membranes by machine.
实施例23:滴丸剂。Example 23: Dropping pills.
用含有权利要求1中化合物的化合物(以实施例31化合物为例)10g,与明胶等基质50g加热熔化混匀后,滴入低温液体石蜡中,共制得滴丸1000丸。10 g of the compound containing the compound in claim 1 (taking the compound of Example 31 as an example) was heated, melted and mixed with 50 g of a matrix such as gelatin, and then dropped into low-temperature liquid paraffin to prepare 1000 dripping pills.
实施例24:外用搽剂。Example 24: External liniment.
用含有权利要求1中化合物的化合物(以实施例31化合物为例)10g,按照常规药剂学方法与乳化剂等辅料2.5g混合研磨,再加蒸馏水至200mL制得。Using 10 g of the compound containing the compound in claim 1 (taking the compound of Example 31 as an example), mix and grind 2.5 g of auxiliary materials such as emulsifiers according to conventional pharmaceutical methods, and then add distilled water to 200 mL to prepare.
实施例25:软膏剂。Example 25: Ointment.
用含有权利要求1中化合物的化合物(以实施例31化合物为例)10g,研细后与凡士林等油性基质500g研匀制得。Using 10 g of the compound containing the compound in claim 1 (taking the compound of Example 31 as an example), grind it finely with 500 g of an oily base such as vaseline to make it.
尽管已经通过特定实施方案描述了本发明,但修改和等价变化对于精通此领域的技术人员而言是显见的,且它们都包含在本发明范围。While the invention has been described in terms of specific embodiments, modifications and equivalents will be apparent to those skilled in the art and are intended to be included within the scope of the invention.
Claims (7)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010749278.4A CN111848600B (en) | 2020-07-30 | 2020-07-30 | 2,4, 4-trisubstituted dihydrooxazole derivative and application thereof |
| PCT/CN2021/109144 WO2022022616A1 (en) | 2020-07-30 | 2021-07-29 | 2,4,4-trisubstituted dihydrooxazole derivatives and preparation methods therefor and use thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010749278.4A CN111848600B (en) | 2020-07-30 | 2020-07-30 | 2,4, 4-trisubstituted dihydrooxazole derivative and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111848600A CN111848600A (en) | 2020-10-30 |
| CN111848600B true CN111848600B (en) | 2021-12-07 |
Family
ID=72946418
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010749278.4A Active CN111848600B (en) | 2020-07-30 | 2020-07-30 | 2,4, 4-trisubstituted dihydrooxazole derivative and application thereof |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN111848600B (en) |
| WO (1) | WO2022022616A1 (en) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111848600B (en) * | 2020-07-30 | 2021-12-07 | 沈阳药科大学 | 2,4, 4-trisubstituted dihydrooxazole derivative and application thereof |
| CN114605283B (en) * | 2022-03-22 | 2024-11-19 | 江苏阿尔法集团盛基药业(宿迁)有限公司 | A method for efficiently preparing montelukast sodium side chain intermediate |
| CN114605282B (en) * | 2022-03-22 | 2024-10-08 | 江苏阿尔法集团盛基药业(宿迁)有限公司 | Preparation method of Montelukast sodium side chain intermediate |
Family Cites Families (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4338327A (en) * | 1978-10-06 | 1982-07-06 | Janssen Pharmaceutica, N.V. | Substituted 1-(2-aryl-1,3-dioxolan-2-ylmethyl)-1H-1,2,4-triazoles |
| US4384879A (en) * | 1980-07-15 | 1983-05-24 | Ciba-Geigy Corporation | 4-(1H-Azolylmethyl)-1,3-dioxolan-5-one derivatives, production thereof and use thereof as growth regulators and/or microbicides |
| GB2095236B (en) * | 1981-03-18 | 1985-03-27 | Ici Plc | Heterocyclylmethyl-substituted dioxolanes and their use as fungicides |
| JPS58128383A (en) * | 1982-01-26 | 1983-07-30 | Sumitomo Chem Co Ltd | Triazole-based compound, its preparation, agricultural and gardening fungicide, plant growth regulator, or herbicide containing it as active ingredient |
| EP0094167A3 (en) * | 1982-05-12 | 1984-07-04 | Fbc Limited | Azolyl fungicide and plant growth regulators and compositions containing them |
| US4785117A (en) * | 1987-10-02 | 1988-11-15 | Pennwalt Corporation | 5,5-disubstituted-3-phenyl-3-phenyl-3-[(1H-imidazol-1-ylmethyl) or (1H-1,2,4-triazol-1-ylmethyl)]-2-methylisoxazolidine derivatives (IR 3012) |
| US4835283A (en) * | 1988-03-07 | 1989-05-30 | Pennwalt Corporation | 3,5-diphenyl-3-[(1H-imidazol-1-ylmethyl) or (1H-1,2,4-triazol-1-ylmethyl)]-2 |
| DE4013723A1 (en) * | 1990-04-28 | 1991-10-31 | Basf Ag | 5- (1,2,4-TRIAZOL-1-YLMETHYL) -ISOXAZOLINE |
| ES2038906B1 (en) * | 1991-09-04 | 1994-02-16 | Uriach & Cia Sa J | PROCEDURE FOR THE OBTAINING OF NEW OXAZOILIDINES. |
| JPH06263757A (en) * | 1993-03-10 | 1994-09-20 | Kaken Pharmaceut Co Ltd | Azole compounds |
| JPH0912574A (en) * | 1995-06-28 | 1997-01-14 | Maruho Kk | Antimycotic agent |
| BR0215702A (en) * | 2002-04-12 | 2005-10-18 | Ranbaxy Lab Ltd | 2,2,4-Trisubstituted tetrahydrofuran derivatives as antifungal agents |
| JP2009286773A (en) * | 2008-03-14 | 2009-12-10 | Bayer Cropscience Ag | Insecticidal condensed-ring aryl compounds |
| CN104119322B (en) * | 2014-07-11 | 2016-05-18 | 北京迪尔乐农业高新技术研发中心 | A kind of triazole class compounds for sterilization and its preparation method and application |
| CN111848600B (en) * | 2020-07-30 | 2021-12-07 | 沈阳药科大学 | 2,4, 4-trisubstituted dihydrooxazole derivative and application thereof |
-
2020
- 2020-07-30 CN CN202010749278.4A patent/CN111848600B/en active Active
-
2021
- 2021-07-29 WO PCT/CN2021/109144 patent/WO2022022616A1/en not_active Ceased
Also Published As
| Publication number | Publication date |
|---|---|
| WO2022022616A1 (en) | 2022-02-03 |
| CN111848600A (en) | 2020-10-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101213191B (en) | Antifungal Triazole Derivatives | |
| KR101286499B1 (en) | Neurotherapeutic azole compounds | |
| CN111848600B (en) | 2,4, 4-trisubstituted dihydrooxazole derivative and application thereof | |
| US5900488A (en) | Method for treating mycosis using imidazolylacetonitrile derivatives | |
| CN107311995B (en) | Tricyclic isoxazole class derivative and its preparation method and application | |
| RU2131417C1 (en) | Derivatives of azole, methods of their synthesis and antifungal agent | |
| CN105777664B (en) | Carboxylate of 2 (2 benzyl hydrazono-) thiazole 5 and preparation method thereof and medical usage | |
| CN101519402A (en) | A kind of thiochromone compound and its synthetic method and the application in the preparation of antifungal drugs | |
| CN105541828B (en) | Amide imidazole derivative and application thereof | |
| WO2004018485A1 (en) | Azole derivatives as antifungal agents | |
| CN118496170B (en) | CYP 51-targeted azole compound and application thereof | |
| CN109485607B (en) | Beta-azole-phenyl ketone derivative and application thereof | |
| RU2690161C1 (en) | 3,5-substituted thiazolidine-2,4-dione derivatives, having antimicrobial activity | |
| CN102712596B (en) | Secondary 8-hydroxyquinoline-7-carboxamide derivatives | |
| CN103214457B (en) | Azole antifungal compound and preparation method and application thereof | |
| UA110102C2 (en) | 8-HYDROXYCHINOLINE-7-CARBOXAMIDE SECONDARY DERIVATIVES FOR THEIR APPLICATION AS ANTI-FUNCTIONAL PRODUCTS | |
| RU2662153C1 (en) | Hybrid ethers based on derivatives of thiazolidine-2,4-diona and azoles (1h-1,3-imidazole and 1h-1,3,4-triazole) and application thereof | |
| CN110950845B (en) | Formylacetamide azole derivative and application thereof | |
| CN112142673B (en) | Aryl alkene azole derivatives, preparation method and use thereof | |
| WO2026016515A1 (en) | Azole derivative having nitrogen-containing side chain substituent and use thereof in medicine | |
| CN111320574B (en) | Aryl amide derivative and application thereof | |
| MXPA02009673A (en) | Triazole compounds having amide linkage. | |
| CN114736164B (en) | 1,2,4-triazole benzophenone compounds or pharmaceutically acceptable salts thereof and use thereof | |
| CN118515621A (en) | Azolene alcohol compounds containing ethynyl groups and uses thereof | |
| CN115160250B (en) | 4,6-Biphenyldiphenol derivatives and uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |