CN111733129A - A method for the induction of high-efficiency DC-CIK cells by polymyocytes - Google Patents
A method for the induction of high-efficiency DC-CIK cells by polymyocytes Download PDFInfo
- Publication number
- CN111733129A CN111733129A CN202010255218.7A CN202010255218A CN111733129A CN 111733129 A CN111733129 A CN 111733129A CN 202010255218 A CN202010255218 A CN 202010255218A CN 111733129 A CN111733129 A CN 111733129A
- Authority
- CN
- China
- Prior art keywords
- cells
- tumor
- cik
- polymyocytes
- immune
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 238000000034 method Methods 0.000 title claims description 24
- 230000006698 induction Effects 0.000 title description 5
- 210000004027 cell Anatomy 0.000 claims abstract description 92
- 230000035755 proliferation Effects 0.000 claims abstract description 16
- 210000002865 immune cell Anatomy 0.000 claims abstract description 13
- 230000004069 differentiation Effects 0.000 claims abstract description 4
- 229940115272 polyinosinic:polycytidylic acid Drugs 0.000 claims abstract description 4
- 210000005087 mononuclear cell Anatomy 0.000 claims description 10
- 230000035800 maturation Effects 0.000 claims description 5
- 239000000411 inducer Substances 0.000 claims description 3
- 230000001939 inductive effect Effects 0.000 claims description 3
- 230000002195 synergetic effect Effects 0.000 claims description 2
- 206010028980 Neoplasm Diseases 0.000 abstract description 95
- 201000011510 cancer Diseases 0.000 abstract description 25
- 238000011282 treatment Methods 0.000 abstract description 21
- 230000036039 immunity Effects 0.000 abstract description 17
- 210000004881 tumor cell Anatomy 0.000 abstract description 17
- 230000000694 effects Effects 0.000 abstract description 15
- 210000000987 immune system Anatomy 0.000 abstract description 15
- 230000002476 tumorcidal effect Effects 0.000 abstract description 12
- 102000002689 Toll-like receptor Human genes 0.000 abstract description 9
- 108020000411 Toll-like receptor Proteins 0.000 abstract description 9
- 230000036737 immune function Effects 0.000 abstract description 8
- 208000007660 Residual Neoplasm Diseases 0.000 abstract description 7
- 239000002253 acid Substances 0.000 abstract description 7
- 206010027476 Metastases Diseases 0.000 abstract description 5
- 230000028993 immune response Effects 0.000 abstract description 5
- 230000009401 metastasis Effects 0.000 abstract description 5
- 230000004913 activation Effects 0.000 abstract description 4
- 229920001577 copolymer Polymers 0.000 abstract description 4
- 239000003446 ligand Substances 0.000 abstract description 4
- 230000004083 survival effect Effects 0.000 abstract description 4
- 108091036414 Polyinosinic:polycytidylic acid Proteins 0.000 abstract description 3
- 230000003902 lesion Effects 0.000 abstract description 3
- 241001465754 Metazoa Species 0.000 abstract description 2
- 239000012980 RPMI-1640 medium Substances 0.000 description 30
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 28
- 229910002092 carbon dioxide Inorganic materials 0.000 description 14
- 239000001569 carbon dioxide Substances 0.000 description 14
- 239000003636 conditioned culture medium Substances 0.000 description 13
- 239000002609 medium Substances 0.000 description 13
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 12
- 238000001815 biotherapy Methods 0.000 description 11
- 238000005516 engineering process Methods 0.000 description 11
- 238000012546 transfer Methods 0.000 description 11
- 238000004113 cell culture Methods 0.000 description 9
- 238000002360 preparation method Methods 0.000 description 9
- 238000002512 chemotherapy Methods 0.000 description 8
- 239000000203 mixture Substances 0.000 description 8
- 238000001959 radiotherapy Methods 0.000 description 8
- 102000004127 Cytokines Human genes 0.000 description 7
- 108090000695 Cytokines Proteins 0.000 description 7
- GLNADSQYFUSGOU-GPTZEZBUSA-J Trypan blue Chemical compound [Na+].[Na+].[Na+].[Na+].C1=C(S([O-])(=O)=O)C=C2C=C(S([O-])(=O)=O)C(/N=N/C3=CC=C(C=C3C)C=3C=C(C(=CC=3)\N=N\C=3C(=CC4=CC(=CC(N)=C4C=3O)S([O-])(=O)=O)S([O-])(=O)=O)C)=C(O)C2=C1N GLNADSQYFUSGOU-GPTZEZBUSA-J 0.000 description 7
- 230000001093 anti-cancer Effects 0.000 description 7
- 210000004698 lymphocyte Anatomy 0.000 description 7
- 238000001356 surgical procedure Methods 0.000 description 7
- 230000000259 anti-tumor effect Effects 0.000 description 6
- 210000004369 blood Anatomy 0.000 description 6
- 239000008280 blood Substances 0.000 description 6
- 238000000338 in vitro Methods 0.000 description 6
- 210000005259 peripheral blood Anatomy 0.000 description 6
- 239000011886 peripheral blood Substances 0.000 description 6
- 108010074328 Interferon-gamma Proteins 0.000 description 5
- 108010050904 Interferons Proteins 0.000 description 5
- 102000014150 Interferons Human genes 0.000 description 5
- 210000001744 T-lymphocyte Anatomy 0.000 description 5
- 210000004443 dendritic cell Anatomy 0.000 description 5
- 238000001514 detection method Methods 0.000 description 5
- 238000009169 immunotherapy Methods 0.000 description 5
- 238000001727 in vivo Methods 0.000 description 5
- 229940079322 interferon Drugs 0.000 description 5
- 229920006395 saturated elastomer Polymers 0.000 description 5
- 102400000888 Cholecystokinin-8 Human genes 0.000 description 4
- 101800005151 Cholecystokinin-8 Proteins 0.000 description 4
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 4
- 102000008070 Interferon-gamma Human genes 0.000 description 4
- 108010002350 Interleukin-2 Proteins 0.000 description 4
- 230000010100 anticoagulation Effects 0.000 description 4
- 239000000427 antigen Substances 0.000 description 4
- 102000036639 antigens Human genes 0.000 description 4
- 108091007433 antigens Proteins 0.000 description 4
- 210000000601 blood cell Anatomy 0.000 description 4
- 244000309466 calf Species 0.000 description 4
- 239000012530 fluid Substances 0.000 description 4
- 230000006870 function Effects 0.000 description 4
- 229940044627 gamma-interferon Drugs 0.000 description 4
- 229960002897 heparin Drugs 0.000 description 4
- 229920000669 heparin Polymers 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- 230000002093 peripheral effect Effects 0.000 description 4
- 230000028327 secretion Effects 0.000 description 4
- 239000013049 sediment Substances 0.000 description 4
- 210000002966 serum Anatomy 0.000 description 4
- IZTQOLKUZKXIRV-YRVFCXMDSA-N sincalide Chemical compound C([C@@H](C(=O)N[C@@H](CCSC)C(=O)NCC(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(N)=O)NC(=O)[C@@H](N)CC(O)=O)C1=CC=C(OS(O)(=O)=O)C=C1 IZTQOLKUZKXIRV-YRVFCXMDSA-N 0.000 description 4
- 238000010186 staining Methods 0.000 description 4
- 239000006228 supernatant Substances 0.000 description 4
- 238000005406 washing Methods 0.000 description 4
- 102100039620 Granulocyte-macrophage colony-stimulating factor Human genes 0.000 description 3
- 101000746373 Homo sapiens Granulocyte-macrophage colony-stimulating factor Proteins 0.000 description 3
- 108010063738 Interleukins Proteins 0.000 description 3
- 102000015696 Interleukins Human genes 0.000 description 3
- 230000004663 cell proliferation Effects 0.000 description 3
- 238000002659 cell therapy Methods 0.000 description 3
- 239000012636 effector Substances 0.000 description 3
- 230000002147 killing effect Effects 0.000 description 3
- 210000002540 macrophage Anatomy 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 210000001616 monocyte Anatomy 0.000 description 3
- 239000008188 pellet Substances 0.000 description 3
- 230000001737 promoting effect Effects 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 238000002560 therapeutic procedure Methods 0.000 description 3
- 230000005909 tumor killing Effects 0.000 description 3
- 241000894006 Bacteria Species 0.000 description 2
- 206010054094 Tumour necrosis Diseases 0.000 description 2
- 241000700605 Viruses Species 0.000 description 2
- 210000000612 antigen-presenting cell Anatomy 0.000 description 2
- 230000005975 antitumor immune response Effects 0.000 description 2
- 230000001363 autoimmune Effects 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 230000009400 cancer invasion Effects 0.000 description 2
- 238000005119 centrifugation Methods 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 238000000684 flow cytometry Methods 0.000 description 2
- 230000007774 longterm Effects 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 210000000663 muscle cell Anatomy 0.000 description 2
- 238000005192 partition Methods 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 231100000331 toxic Toxicity 0.000 description 2
- 230000002588 toxic effect Effects 0.000 description 2
- -1 37°C Chemical compound 0.000 description 1
- 206010003445 Ascites Diseases 0.000 description 1
- 208000035143 Bacterial infection Diseases 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 208000026310 Breast neoplasm Diseases 0.000 description 1
- 241000699800 Cricetinae Species 0.000 description 1
- 201000008808 Fibrosarcoma Diseases 0.000 description 1
- 208000029433 Herpesviridae infectious disease Diseases 0.000 description 1
- 101000581981 Homo sapiens Neural cell adhesion molecule 1 Proteins 0.000 description 1
- 208000005045 Interdigitating dendritic cell sarcoma Diseases 0.000 description 1
- 102100037850 Interferon gamma Human genes 0.000 description 1
- 108090001005 Interleukin-6 Proteins 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 102100027347 Neural cell adhesion molecule 1 Human genes 0.000 description 1
- 208000015914 Non-Hodgkin lymphomas Diseases 0.000 description 1
- 241001505332 Polyomavirus sp. Species 0.000 description 1
- 206010037075 Protozoal infections Diseases 0.000 description 1
- 239000006146 Roswell Park Memorial Institute medium Substances 0.000 description 1
- 206010039491 Sarcoma Diseases 0.000 description 1
- 241000700584 Simplexvirus Species 0.000 description 1
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 230000003698 anagen phase Effects 0.000 description 1
- 210000004102 animal cell Anatomy 0.000 description 1
- 230000002155 anti-virotic effect Effects 0.000 description 1
- 238000009175 antibody therapy Methods 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- CFQGDIWRTHFZMQ-UHFFFAOYSA-N argon helium Chemical compound [He].[Ar] CFQGDIWRTHFZMQ-UHFFFAOYSA-N 0.000 description 1
- ISQINHMJILFLAQ-UHFFFAOYSA-N argon hydrofluoride Chemical compound F.[Ar] ISQINHMJILFLAQ-UHFFFAOYSA-N 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 208000022362 bacterial infectious disease Diseases 0.000 description 1
- 239000007640 basal medium Substances 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 229940126587 biotherapeutics Drugs 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 238000002619 cancer immunotherapy Methods 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 230000007012 clinical effect Effects 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- 230000001086 cytosolic effect Effects 0.000 description 1
- 230000007123 defense Effects 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000001415 gene therapy Methods 0.000 description 1
- 210000004602 germ cell Anatomy 0.000 description 1
- 230000036449 good health Effects 0.000 description 1
- 208000006454 hepatitis Diseases 0.000 description 1
- 231100000283 hepatitis Toxicity 0.000 description 1
- 230000002519 immonomodulatory effect Effects 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 230000005847 immunogenicity Effects 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 238000002372 labelling Methods 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 210000002751 lymph Anatomy 0.000 description 1
- 210000004962 mammalian cell Anatomy 0.000 description 1
- 201000001441 melanoma Diseases 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 230000001394 metastastic effect Effects 0.000 description 1
- 206010061289 metastatic neoplasm Diseases 0.000 description 1
- 244000000010 microbial pathogen Species 0.000 description 1
- 238000002324 minimally invasive surgery Methods 0.000 description 1
- 238000011242 molecular targeted therapy Methods 0.000 description 1
- 208000025113 myeloid leukemia Diseases 0.000 description 1
- 210000003098 myoblast Anatomy 0.000 description 1
- 210000000822 natural killer cell Anatomy 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 230000000149 penetrating effect Effects 0.000 description 1
- 210000003819 peripheral blood mononuclear cell Anatomy 0.000 description 1
- 230000035790 physiological processes and functions Effects 0.000 description 1
- 230000006461 physiological response Effects 0.000 description 1
- 230000015323 positive regulation of phagocytosis Effects 0.000 description 1
- 238000004393 prognosis Methods 0.000 description 1
- 230000002062 proliferating effect Effects 0.000 description 1
- 230000009993 protective function Effects 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 238000002271 resection Methods 0.000 description 1
- 208000029922 reticulum cell sarcoma Diseases 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 238000013517 stratification Methods 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 102000003390 tumor necrosis factor Human genes 0.000 description 1
- 238000012795 verification Methods 0.000 description 1
- 230000002747 voluntary effect Effects 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0634—Cells from the blood or the immune system
- C12N5/0636—T lymphocytes
- C12N5/0638—Cytotoxic T lymphocytes [CTL] or lymphokine activated killer cells [LAK]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0634—Cells from the blood or the immune system
- C12N5/0636—T lymphocytes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0634—Cells from the blood or the immune system
- C12N5/0639—Dendritic cells, e.g. Langherhans cells in the epidermis
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0634—Cells from the blood or the immune system
- C12N5/0646—Natural killers cells [NK], NKT cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/40—Nucleotides, nucleosides or bases
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/50—Cell markers; Cell surface determinants
Landscapes
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Wood Science & Technology (AREA)
- Biotechnology (AREA)
- Organic Chemistry (AREA)
- Immunology (AREA)
- Genetics & Genomics (AREA)
- Chemical & Material Sciences (AREA)
- Zoology (AREA)
- Hematology (AREA)
- Microbiology (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Cell Biology (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
本发明涉及免疫技术领域,具体涉及利用聚肌苷酸和聚胞苷酸的共聚物‑聚肌胞(Poly I:C),聚肌胞是一种是动物体内3型Toll样受体的配体,激活TLR‑3后可介导机体一系列的免疫反应,对机体的特异性免疫和非特异性免疫均有很好的促进作用。本发明用聚肌胞诱导的DC‑CIK具有较对照组更高增殖、高分化(尤其CD3+CD4‑CD8+,CTL)、高杀瘤活性。利用聚肌胞诱导的自体高效DC‑CIK及其它免疫细胞,应用于临床肿瘤患者的治疗。以期恢复或提高肿瘤患者免疫功能,提高机体自身的免疫系统对肿瘤细胞进行杀灭和抑制其增殖。降低肿瘤细胞负荷,清除微小的残留病灶或明显抑制了残留肿瘤/癌症细胞增殖的方式,消除复发、转移的因素,增大治愈的可能性,延长生存时间,提高生活质量。从而达到治疗肿瘤/癌症的目的。
The invention relates to the technical field of immunity, in particular to the use of a copolymer of polyinosinic acid and polycytidylic acid-polyinosinic acid (Poly I:C), which is a ligand for type 3 Toll-like receptors in animals. After activation of TLR-3, it can mediate a series of immune responses in the body, and has a good promotion effect on the specific and non-specific immunity of the body. Compared with the control group, the DC-CIK induced by polymyocytes in the present invention has higher proliferation, higher differentiation (especially CD3+CD4-CD8+, CTL) and higher tumoricidal activity. Autologous high-efficiency DC-CIK and other immune cells induced by polymyocytes are used in the treatment of clinical tumor patients. In order to restore or improve the immune function of tumor patients, improve the body's own immune system to kill tumor cells and inhibit their proliferation. Reduce tumor cell load, remove tiny residual lesions or significantly inhibit the proliferation of residual tumor/cancer cells, eliminate recurrence and metastasis factors, increase the possibility of cure, prolong survival time, and improve quality of life. So as to achieve the purpose of treating tumor/cancer.
Description
技术领域technical field
本发明涉及免疫技术领域,具体涉及利用聚肌苷酸和聚胞苷酸的共聚物-聚肌胞(polyinosinic:polycytidylic acid copolymer,Poly I:C),聚肌胞是一种是动物体内3型 Toll样受体的配体,激活TLR-3后可介导机体一系列的免疫反应,如诱导干扰素、白介素、肿瘤坏死因子等细胞因子的分泌,促进单核细胞、巨噬细胞、淋巴细胞及树突状细胞的增殖与成熟等,对机体的特异性免疫和非特异性免疫均有很好的促进作用。The invention relates to the technical field of immunity, in particular to the use of a copolymer of polyinosinic acid and polycytidylic acid-polyinosinic acid (polyinosinic: polycytidylic acid copolymer, Poly I:C), and polyinosinic acid is a kind of 3 type in animal body. The ligand of Toll-like receptor, after activation of TLR-3, can mediate a series of immune responses in the body, such as inducing the secretion of cytokines such as interferon, interleukin, tumor necrosis factor, etc., promoting monocytes, macrophages, lymphocytes, etc. And the proliferation and maturation of dendritic cells, etc., to the body's specific immunity and non-specific immunity have a good role in promoting.
使用聚肌胞加入诱导培养基中协同诱导DC-CIK(巨噬细胞-细胞因子活化的肿瘤杀伤细胞)细胞的增殖与分化,使DC-CIK的杀瘤活性得到大幅提升。以期获得一种聚肌胞诱导的自体高效DC-CIK细胞,应用于临床肿瘤患者的治疗。达到更好的杀灭残余的肿瘤细胞,减少肿瘤复发、转移的因素,增大治愈的可能性,延长患者的生存时间,提高患者生存质量。The proliferation and differentiation of DC-CIK (macrophage-cytokine-activated tumor killer cells) cells are synergistically induced by adding polymyocytes to the induction medium, which greatly improves the tumoricidal activity of DC-CIK. In order to obtain a polymyocyte-induced autologous high-efficiency DC-CIK cells, which can be used in the treatment of clinical tumor patients. It can better kill residual tumor cells, reduce the factors of tumor recurrence and metastasis, increase the possibility of cure, prolong the survival time of patients, and improve the quality of life of patients.
背景技术Background technique
自体免疫细胞(DC-CIK)应用于肿瘤生物治疗是一种新兴的、安全的及具有显著疗效的肿瘤治疗模式,是一种治疗肿瘤的新型治疗方法。该方法是运用生物学技术和生物制剂,对从肿瘤患者外周血采集、分离的免疫细胞-单个核细胞(包括树突状细胞、T淋巴细胞、B淋巴细胞)进行体外诱导培养和扩增后,回输到病人体内以激发和增强机体自身免疫功能,从而达到治疗肿瘤的目的。肿瘤生物治疗技术是继手术、放疗和化疗之后的第四大肿瘤治疗的方法。The application of autologous immune cells (DC-CIK) in tumor biological therapy is an emerging, safe and effective tumor treatment mode, and it is a new therapeutic method for tumor treatment. The method uses biological technology and biological preparations to induce in vitro culture and expansion of immune cells-mononuclear cells (including dendritic cells, T lymphocytes, B lymphocytes) collected and isolated from the peripheral blood of tumor patients. , back into the patient's body to stimulate and enhance the body's own immune function, so as to achieve the purpose of tumor treatment. Tumor biotherapeutics is the fourth most important method of tumor treatment after surgery, radiotherapy and chemotherapy.
聚肌胞是聚肌苷酸和聚胞苷酸的共聚物,是一种高效的干扰素(IFN)诱生剂,具有广谱抗病毒、刺激吞噬、调整机体免疫、抗肿瘤、抗某些细菌和原虫感染等功能。目前在人类医学上用于治疗肝炎、疱疹病毒感染及一些肿瘤性疾病等方面,显示了其确实的临床疗效。Polyinosin is a copolymer of polyinosinic acid and polycytidylic acid. It is an efficient interferon (IFN) inducer with broad-spectrum anti-virus, stimulation of phagocytosis, regulation of immunity, anti-tumor, and anti-cerebral properties. Bacterial and protozoal infections, etc. At present, it is used in human medicine to treat hepatitis, herpes virus infection and some tumor diseases, etc., and it has shown its real clinical effect.
同时,聚肌胞是动物细胞3型Toll样受体(TLR-3)的配体,聚肌胞激活TLR-3后可介导机体一系列的免疫反应,如诱导干扰素、白介素、肿瘤坏死因子等细胞因子的分泌,促进免疫细胞(包括:单核细胞、巨噬细胞、淋巴细胞及树突状细胞)的增殖与成熟等,促进体内抗体的生成。聚肌胞对机体的特异性免疫和非特异性免疫均有很好的促进作用。At the same time, polymyocytes are ligands of
聚肌胞的免疫调节作用:免疫是机体的一种生理性保护功能,是机体识别和消灭外来侵入的任何异物(病毒、细菌等),处理衰老、损伤、死亡、变性的自身细胞以及识别和处理体内突变细胞和病毒感染细胞的能力。现代免疫学认为,免疫力是人体识别和排除“异己”的生理反应。机体的免疫能力可分为特异性免疫与非特异性免疫两种,两者是密切联系的。非特异性免疫是生物在种系发展过程中不断与病原微生物斗争中形成的,并可遗传给后代的一种免疫功能,它是与人体的组织特性和生理机能密切相关的。特异性免疫是机体在后天受内外环境因素的刺激而获得的免疫功能。它能识别再次接触的相同抗原,并发生相应的免疫学反应, 它需要在高度分化的组织和细胞的参与下才能完成。Immunomodulatory effect of polymyocytes: Immunity is a physiological protective function of the body. It is the body's ability to recognize and eliminate any foreign intrusion (viruses, bacteria, etc.) Ability to handle mutant cells and virus-infected cells in vivo. Modern immunology believes that immunity is the physiological response of the human body to identify and exclude "others". The immune capacity of the body can be divided into specific immunity and non-specific immunity, and the two are closely related. Non-specific immunity is an immune function formed by organisms in the process of germline development in the continuous struggle with pathogenic microorganisms, and can be inherited to offspring. It is closely related to the tissue characteristics and physiological functions of the human body. Specific immunity is the immune function acquired by the body after being stimulated by internal and external environmental factors. It can recognize the same antigen when it is contacted again, and the corresponding immunological response occurs, which requires the participation of highly differentiated tissues and cells to complete.
(1)聚肌胞增强免疫细胞的活性;(1) Polymyocytes enhance the activity of immune cells;
(2)聚肌胞促进多种细胞因子的分泌;(2) Polymyocytes promote the secretion of various cytokines;
(3)聚肌胞对Mx蛋白的诱生作用;(3) Induction effect of polymyocytes on Mx protein;
(4)聚肌胞促进体内抗体的生成。(4) Polymyocytes promote the production of antibodies in vivo.
聚肌胞的抗肿瘤作用研究已经证明,聚肌胞不仅可以抑制致瘤病毒和致瘤病毒如多瘤病毒、单纯泡疹病毒等引发的肿瘤。对移植肿瘤,如小鼠的多种网状细胞肉瘤、腹水瘤、S91黑色素瘤、乳腺瘤、纤维肉瘤等,以及地鼠的鼠肉瘤,都不同程度的抑制。Studies on the anti-tumor effect of polymyocytes have proved that polymyocytes can not only inhibit tumors caused by oncoviruses and oncoviruses such as polyoma virus and herpes simplex virus. The transplanted tumors, such as a variety of reticulum cell sarcoma, ascites tumor, S91 melanoma, breast tumor, fibrosarcoma, etc. in mice, as well as mouse sarcoma in hamsters, are inhibited to varying degrees.
肿瘤生物治疗是一种新兴的、具有显著疗效的肿瘤治疗模式,是一种自身免疫抗癌的新型治疗方法。它是运用生物技术和生物制剂对从病人体内采集的免疫细胞进行体外培养和扩增后回输到病人体内的方法,来激发、增强机体自身免疫功能,从而达到治疗肿瘤的目的。肿瘤生物治疗是继手术、放疗和化疗之后的第四大肿瘤治疗技术。Tumor biological therapy is an emerging tumor treatment mode with remarkable curative effect, and a new treatment method for autoimmune anti-cancer. It is a method of using biotechnology and biological preparations to culture and expand immune cells collected from patients in vitro and then infuse them into the patient's body to stimulate and enhance the body's autoimmune function, so as to achieve the purpose of treating tumors. Tumor biological therapy is the fourth largest tumor treatment technology after surgery, radiotherapy and chemotherapy.
免疫系统是人体的防御体系,一方面发挥着清除细菌、病毒、外来异物的功能,另一方面消除体内衰老细胞以及发生突变的细胞(部份突变细胞会变成肿瘤细胞)。机体免疫系统和肿瘤细胞相互作用的结果决定了肿瘤的最终演变。对于健康的人来说,其免疫系统的强大足以及时清除突变的癌细胞。但对于肿瘤病人来说,普遍存在免疫系统低下,不能有效地识别、杀灭肿瘤/癌症细胞;另一方面,肿瘤/癌症细胞大量增殖,会进一步抑患者的免疫功能,而且,肿瘤/癌症细胞有多种机制来逃脱免疫细胞的识别与杀伤,肿瘤/癌症的免疫治疗就是借助分子生物学技术和细胞工程技术,提高肿瘤/癌症的免疫原性,给机体补充足够数量的功能正常的免疫细胞和相关分子,激发和增强机体抗瘤免疫应答,提高肿瘤/癌症对机体抗肿瘤/ 癌症免疫效应的敏感性,在体内、外诱导肿瘤/癌症特异性和非特异性效应细胞和分子,达到最终清除肿瘤/癌症的目的。The immune system is the body's defense system. On the one hand, it plays the function of removing bacteria, viruses, and foreign bodies. On the other hand, it eliminates senescent cells and mutated cells in the body (some mutated cells will become tumor cells). The outcome of the interaction between the body's immune system and tumor cells determines the final evolution of the tumor. In a healthy person, the immune system is strong enough to clear mutated cancer cells in time. However, for tumor patients, the immune system is generally low, which cannot effectively identify and kill tumor/cancer cells; on the other hand, the massive proliferation of tumor/cancer cells will further suppress the patient's immune function. There are various mechanisms to escape the recognition and killing of immune cells. The immunotherapy of tumor/cancer is to improve the immunogenicity of tumor/cancer by means of molecular biology technology and cell engineering technology, and to supplement the body with a sufficient number of normal immune cells. and related molecules, stimulate and enhance the body's anti-tumor immune response, improve the sensitivity of tumors/cancers to the body's anti-tumor/cancer immune effects, induce tumor/cancer specific and non-specific effector cells and molecules in vivo and in vitro, and achieve final clearance Tumor/Cancer Purpose.
肿瘤/癌症生物治疗,其作用不是杀死全部肿瘤/癌症细胞,而是由于当肿瘤/癌症细胞负荷明显降低时,机体的免疫功能恢复后,通过清除微小的残留病灶或明显抑制了残留肿瘤/ 癌症细胞增殖的方式来达到治疗肿瘤/癌症的目的。肿瘤/癌症免疫治疗正是通过人为的干预,来调动机体自身的免疫系统对癌细胞进行杀灭和抑制其增殖。Tumor/cancer biological therapy, its effect is not to kill all tumor/cancer cells, but because when the tumor/cancer cell load is significantly reduced, after the body's immune function is restored, by removing tiny residual lesions or significantly inhibiting residual tumor/cancer The way cancer cells proliferate for the purpose of treating tumors/cancers. Tumor/cancer immunotherapy is precisely through human intervention to mobilize the body's own immune system to kill cancer cells and inhibit their proliferation.
实验及临床均提示机体的免疫系统具有清除肿瘤/癌症的作用,在原发性肿瘤/癌症手术切除或经氩氟刀等微创手术消融掉局部肿瘤/癌症后,用免疫疗法能杀灭剩余的肿瘤/癌症细胞,消除复发、转移的因素,增大治愈的可能性,延长生存时间,提高生活质量。Both experiments and clinical studies have shown that the body's immune system has the effect of removing tumors/cancers. After the primary tumor/cancer is surgically removed or the local tumor/cancer is ablated by minimally invasive surgery such as argon fluoride knife, immunotherapy can kill the remaining tumor/cancer. It can eliminate the factors of recurrence and metastasis, increase the possibility of cure, prolong the survival time, and improve the quality of life.
截止2013年,肿瘤/癌症生物治疗已被视为继手术、放疗、化疗之后的第四种治疗方法。生物疗法包括细胞因子治疗、免疫细胞治疗、基因治疗、分子靶向治疗和抗体治疗等。As of 2013, tumor/cancer biological therapy has been regarded as the fourth treatment method after surgery, radiotherapy and chemotherapy. Biological therapy includes cytokine therapy, immune cell therapy, gene therapy, molecular targeted therapy and antibody therapy.
生物细胞免疫疗法是继手术、放疗和化疗之后的第四大肿瘤治疗技术。生物细胞免疫疗法治疗肿瘤,具有以下优势:Biological cellular immunotherapy is the fourth largest tumor treatment technology after surgery, radiotherapy and chemotherapy. Biological cell immunotherapy for the treatment of tumors has the following advantages:
1.效果确切,有效率高。对有些肿瘤/癌症,有效率高达70%。1. The effect is exact and the efficiency is high. For some tumors/cancers, the effective rate is as high as 70%.
2.无放、化疗毒副作用,病人不痛苦,耐受性好,杀瘤特异性强。2. No toxic and side effects of radiotherapy and chemotherapy, no pain for patients, good tolerance, and strong tumor killing specificity.
3.能够激发全身性的抗癌效应,对多发病灶或转移的恶性肿瘤同样有效。3. It can stimulate the systemic anti-cancer effect, and is also effective for multiple foci or metastatic malignant tumors.
4.可以帮助机体快速恢复被放、化疗破坏的抗癌免疫系统,提高远期抗癌能力。4. It can help the body quickly restore the anti-cancer immune system destroyed by radiotherapy and chemotherapy, and improve the long-term anti-cancer ability.
5.对肿瘤/癌症术后防复发效果显著,远期抗癌效果良好。5. It has remarkable anti-recurrence effect on tumor/cancer after operation, and has good long-term anti-cancer effect.
6.可单独使用也可与其它治疗方法联合使用。单次有效,多次使用,效果更佳。6. It can be used alone or in combination with other treatment methods. Single effective, multiple use, the effect is better.
自体DC-CIK细胞是众多免疫细胞疗法的一种。DC-CIK细胞能以不同的机制识别肿瘤细胞,通过直接的细胞质颗粒穿透封闭的肿瘤细胞膜,实现对肿瘤细胞的裂解;通过诱导肿瘤细胞凋亡杀伤肿瘤细胞;DC-CIK细胞能分泌IL-2、IL-6、IFN-γ等多种抗肿瘤的细胞因子; DC-CIK细胞回输后可激活机体自身的免疫系统,提高机体的免疫功能。Autologous DC-CIK cells are one of many immune cell therapies. DC-CIK cells can recognize tumor cells by different mechanisms, lyse tumor cells by penetrating the closed tumor cell membrane through direct cytoplasmic particles; kill tumor cells by inducing tumor cell apoptosis; DC-CIK cells can secrete IL- 2. Various anti-tumor cytokines such as IL-6 and IFN-γ; DC-CIK cells can activate the body's own immune system and improve the body's immune function after infusion.
该疗法既可单独使用,也可以作为手术、化疗和放疗后的有力辅助手段,效果显著。结合手术切除、介入、射频、氩氦刀等治疗,可清除不能用手术切除的极微小瘤灶或是体内散存的瘤细胞,在延缓或阻止肿瘤的转移或复发方面有重要作用;对于部分暂时不适宜做手术、介入或其它治疗的肿瘤患者,也可以先进行DC-CIK细胞治疗,提高身体机能状况,改善生活质量,争取其它治疗机会。The therapy can be used alone or as a powerful adjunct after surgery, chemotherapy and radiotherapy, with remarkable results. Combined with surgical resection, intervention, radiofrequency, argon-helium knife and other treatments, it can remove extremely small tumor foci that cannot be surgically removed or scattered tumor cells in the body, and plays an important role in delaying or preventing tumor metastasis or recurrence; for some Cancer patients who are temporarily unsuitable for surgery, intervention or other treatments can also undergo DC-CIK cell therapy first to improve their physical function, improve their quality of life, and strive for other treatment opportunities.
由于生物免疫治疗技术是利用人体自身的免疫细胞,而不是传统的化学药品来杀伤肿瘤细胞的,因此该技术安全无毒副作用,适用于肿瘤/癌症各阶段的治疗,自DC-CIK在临床开展以来已为无数肿瘤/癌症患者解除了病痛的折磨,为肿瘤患者带来健康的福音!Since the biological immunotherapy technology uses the body's own immune cells instead of traditional chemicals to kill tumor cells, this technology is safe and has no toxic side effects, and is suitable for the treatment of tumors/cancers at all stages. Since then, it has relieved the pain of countless tumor/cancer patients and brought good health to tumor patients!
肿瘤生物治疗是优于手术、放疗和化疗的最新肿瘤治疗技术,是通过生物技术在高标准的实验室内培养出可杀伤肿瘤的自体免疫细胞,回输体内,直接杀伤癌细胞的治疗方法。与传统的治疗方法不同,肿瘤生物治疗在不损伤和机体免疫系统和功能的前提下,直接识别、消灭存在于人体内血液、淋巴中的癌细胞,恢复和增强机体自然抗癌免疫系统和功能。肿瘤生物治疗具有不伤身体、无痛、无需住院等优点,能提高患者自身的免疫与生活质量,并适用于所有的肿瘤疾病,该生物治疗技术已正式运用于临床。Tumor biological therapy is the latest tumor treatment technology superior to surgery, radiotherapy and chemotherapy. It is a treatment method in which autologous immune cells that can kill tumors are cultivated in a high-standard laboratory through biotechnology, and then reinfused into the body to directly kill cancer cells. Different from traditional treatment methods, tumor biotherapy can directly identify and eliminate cancer cells existing in the blood and lymph of the human body without damaging the body's immune system and function, and restore and enhance the body's natural anti-cancer immune system and function. . Tumor biological therapy has the advantages of no harm to the body, no pain, and no need to be hospitalized. It can improve the patient's own immunity and quality of life, and is applicable to all tumor diseases. This biological therapy technology has been formally used in clinical practice.
DC即树突状细胞,发现于1973年。它是机体中功能最强的专职抗原递呈细胞,它能高效地摄取、加工处理和递呈抗原。DC细胞与肿瘤的发生、发展有着密切关系,是激活机体免疫系统、激发抵御肿瘤/癌症侵袭最有效的途径之一。通过大量体外活化培养负载肿瘤抗原的 DC细胞,在细胞数量达到规模化后回输给病人,可诱导机体产生强烈的抗肿瘤免疫反应。临床验证,大部分实体瘤内浸润的DC数量多则患者预后好。DC stands for dendritic cells and was discovered in 1973. It is the most functional professional antigen-presenting cell in the body, which can efficiently ingest, process and present antigens. DC cells are closely related to the occurrence and development of tumors, and are one of the most effective ways to activate the body's immune system and stimulate resistance to tumor/cancer invasion. By activating and culturing DC cells loaded with tumor antigens in a large amount in vitro, and returning to the patient after the number of cells reaches a large scale, a strong anti-tumor immune response can be induced in the body. According to clinical verification, a large number of infiltrating DCs in most solid tumors results in a better prognosis for patients.
在肿瘤免疫中,DC不能直接杀伤肿瘤细胞,但能通过识别肿瘤细胞特异性抗原,将其信号呈递给具杀伤效应的T细胞来达到监测、杀灭肿瘤的功能,因此常与肿瘤杀伤细胞CIK细胞联合使用。In tumor immunity, DCs cannot directly kill tumor cells, but can monitor and kill tumors by recognizing tumor cell-specific antigens and presenting their signals to T cells with killing effect. Cells are used in combination.
DC是迄今为止发现的功能最为强大的抗原提呈细胞(即把肿瘤的相关信息提供给人体内正常存在的具有杀伤肿瘤活性的细胞),在人体的免疫系统里扮演近似“雷达”的角色;CIK 是外周血单个核细胞,在体外经多种细胞因子共同诱导培养后,产生的一类以CD3+CD56+T 细胞为主要效应细胞的异质细胞群,因此CIK同时具有T细胞和NK细胞这两种人体内主要具有抗肿瘤活性细胞的效应,其在体外抗肿瘤活性较以往生物治疗中培养的LAK、CTL、TIL活性强100~1000倍。医学家们对其更形象地描述是:如果T细胞在人体的免疫系统充当“炮弹”的角色,那么DC-CIK则相当于威力更为强大的“导弹”。DCs are the most powerful antigen-presenting cells discovered so far (that is, provide tumor-related information to cells that normally exist in the body with tumor-killing activity), and play a role similar to "radar" in the human immune system; CIK is peripheral blood mononuclear cells. After being co-induced and cultured by a variety of cytokines in vitro, it produces a heterogeneous cell population with CD3+CD56+ T cells as the main effector cells. Therefore, CIK has both T cells and NK cells. These two types of human body mainly have the effect of anti-tumor active cells, and their anti-tumor activity in vitro is 100-1000 times stronger than that of LAK, CTL and TIL cultured in previous biological therapy. Medical scientists describe it more vividly: if T cells act as "cannonballs" in the body's immune system, then DC-CIK is equivalent to a more powerful "missile".
发明内容SUMMARY OF THE INVENTION
聚肌胞是哺乳动物细胞3型Toll样受体(TLR-3)的配体,聚肌胞具有激活TLR-3后可介导机体一系列的免疫反应,如诱导干扰素、白介素、肿瘤坏死因子等细胞因子的分泌,促进免疫细胞(包括:单核细胞、巨噬细胞、淋巴细胞及树突状细胞)的增殖与成熟等,促进体内抗体的生成。对机体的特异性免疫和非特异性免疫均有很好的促进作用。Polymyocytes are ligands of
因此,选择聚肌胞作为DC-CIK协同诱导剂,高效诱导自体单个核细胞的增殖、分化、成熟,回输至病人体内,用于肿瘤患者的治疗。以期恢复或提高肿瘤患者免疫功能,提高机体自身的免疫系统对肿瘤细胞进行杀灭和抑制其增殖。降低肿瘤细胞负荷,清除微小的残留病灶或明显抑制了残留肿瘤/癌症细胞增殖的方式,消除复发、转移的因素,增大治愈的可能性,延长生存时间,提高生活质量。从而达到治疗肿瘤/癌症的目的。Therefore, polymyocytes are selected as DC-CIK synergistic inducers, which can efficiently induce the proliferation, differentiation and maturation of autologous mononuclear cells, and then infuse them into patients for the treatment of tumor patients. In order to restore or improve the immune function of tumor patients, improve the body's own immune system to kill tumor cells and inhibit their proliferation. Reduce tumor cell load, remove tiny residual lesions or significantly inhibit the proliferation of residual tumor/cancer cells, eliminate recurrence and metastasis factors, increase the possibility of cure, prolong survival time, and improve quality of life. So as to achieve the purpose of treating tumor/cancer.
首先选择采集正常健康自愿献血者外周血,经常规分离单个核细胞。用不同浓度的聚肌胞协同诱导DC-CIK细胞,经对细胞的增殖数量多少、流式细胞仪检测其CD标志物及对肿瘤细胞的杀伤活性,确立最适有效浓度。First, the peripheral blood of normal healthy voluntary blood donors was selected, and mononuclear cells were routinely isolated. DC-CIK cells were induced synergistically with different concentrations of polymyocytes, and the optimal effective concentration was established by measuring the number of cells proliferating, detecting their CD markers and killing activity on tumor cells by flow cytometry.
附图说明Description of drawings
图1是据检测的OD450值,用Graphpad Prism5软件,以聚肌胞浓度为横坐标,对应的OD值为纵坐标制作细胞的增殖曲线;Fig. 1 is the OD 450 value according to the detection, with Graphpad Prism5 software, with the polymyocyte concentration as the abscissa, and the corresponding OD value as the ordinate to make the cell proliferation curve;
图2是根据计数结果,用Graphpad Prism5软件,以细胞培养天数为横坐标,以细胞数目为纵坐标,制作的柱状图;Figure 2 is a histogram made according to the counting results, using Graphpad Prism5 software, with the days of cell culture as the abscissa and the number of cells as the ordinate;
图3是选择流式CD3+CD4-CD8+细胞,以CD4为横坐标,CD8为纵坐标制作的分区图;Figure 3 is a partition map made by selecting CD3+CD4-CD8+ cells by flow-through, with CD4 as the abscissa and CD8 as the ordinate;
图4是各组杀瘤活性百分率,用Graphpad Prism5软件制作的杀瘤活性柱状图。Figure 4 is a histogram of the tumoricidal activity of the percentage of each group's tumoricidal activity, which was produced by Graphpad Prism5 software.
具体实施方式Detailed ways
实施例一、聚肌胞最适浓度的选择Embodiment 1. Selection of the optimal concentration of polymyocytes
材料:Material:
1.聚肌胞(产地:上海源叶生物科技有限公司;Cas:24939-03-5)1. Poly muscle cells (Origin: Shanghai Yuanye Biotechnology Co., Ltd.; Cas: 24939-03-5)
2.Hyclone RPMI 16402. Hyclone RPMI 1640
3.健康自愿者外周抗凝血3. Peripheral anticoagulation in healthy volunteers
4.条件培养基:含IL-2 100000IU/ml、抗人CD3人源化嵌合抗体1000μg/ml、IL-1α4. Conditioned medium: IL-2 100000IU/ml, anti-human CD3 humanized chimeric antibody 1000μg/ml, IL-1α
10000ng/ml、GMCSF10000ng/ml、γ干扰素10000ng/ml。10000ng/ml, GMCSF10000ng/ml, gamma interferon 10000ng/ml.
5.CCK8检测试剂盒(北仁化学科技(北京)有限公司,型号:CK04)5. CCK8 detection kit (Beijing Chemical Technology (Beijing) Co., Ltd., model: CK04)
6.其它:板式酶标仪(深圳雷杜生命科学股份有限公司,型号:RT-6100)、二氧化碳孵箱 (Thermo SCIENTIFIC,型号:3111)、洁净工作台(Thermo SCIENTIFIC,型号:1379)等。6. Others: plate microplate reader (Shenzhen Redu Life Science Co., Ltd., model: RT-6100), carbon dioxide incubator (Thermo SCIENTIFIC, model: 3111), clean workbench (Thermo SCIENTIFIC, model: 1379), etc.
准备:Prepare:
1.DC-CIK培养基的制作:分别取“方法步骤2”的含血浆培养基,按DC-CIK条件培养基:基础培养基=1:100比例加入500μl DC-CIK条件培养基,即成。1. Preparation of DC-CIK medium: respectively take the plasma-containing medium of "Method Step 2", and add 500 μl DC-CIK conditioned medium according to the ratio of DC-CIK conditioned medium: basal medium = 1:100. .
方法:method:
1.无菌采集健康自愿者外周血5ml,250IU肝素抗凝,并轻轻充分混匀。转移至15ml离心管,2000rpm离心沉淀10分钟;1. Aseptically collect 5ml of peripheral blood from healthy volunteers, anticoagulate with 250IU heparin, and mix gently and thoroughly. Transfer to a 15ml centrifuge tube and centrifuge at 2000rpm for 10 minutes;
2.收集上层血浆约2.5ml加入50mlRPMI-1640中备用;2. Collect about 2.5ml of upper plasma and add it to 50ml of RPMI-1640 for later use;
3.血球沉淀物,加生理盐水混悬至10ml;3. Blood cell sediment, add normal saline to suspend to 10ml;
4.将“步骤3”样品用吸管沿管壁缓慢地加到已预先加入3ml淋巴细胞分层液的15ml一次性无菌塑料离心管中,使加入的血液重叠于分层液上,形成明显的分界面(注意不要摇动);4. Slowly add the "
5.置水平离心机2000rpm离心20min,小心取出;5. Centrifuge at 2000rpm for 20min in a horizontal centrifuge and take it out carefully;
6.用一次性无菌塑料巴氏吸管轻轻插入离心管中,吸出白色云雾状的单个核细胞层,转移至15ml离心管中,加生理盐水至15ml,充分混匀细胞,2000rpm离心5min,如此反复洗涤2次;苔盼蓝染色计数,再次2000rpm离心5min,弃上清;6. Gently insert a disposable sterile plastic Pasteur pipette into the centrifuge tube, suck out the white cloudy mononuclear cell layer, transfer it to a 15ml centrifuge tube, add normal saline to 15ml, mix the cells thoroughly, and centrifuge at 2000rpm for 5min. Repeat the washing twice in this way; trypan blue staining and counting, centrifuge again at 2000rpm for 5min, and discard the supernatant;
7.根据细胞计数结果,用DC-CIK-Hyclone RPMI 1640培养基悬浮细胞沉淀,使细胞终浓度为2×105个/ml,备用;7. According to the cell count results, suspend the cell pellet in DC-CIK-Hyclone RPMI 1640 medium to make the final cell concentration 2×10 5 cells/ml, for later use;
8.分别在96孔细胞培养板中用DC-CIK-Hyclone RPMI 1640培养基4倍梯度稀释聚肌胞,使聚肌胞终浓度分别为1、0.25、0.0625……直至9.54E-07mg/ml。同时设无聚肌胞的空白对照。每一浓度2复孔、每孔100μl;8. In 96-well cell culture plates, use DC-CIK-Hyclone RPMI 1640 medium to dilute the polymyocytes by 4-fold gradient, so that the final concentrations of polymyocytes are 1, 0.25, 0.0625... until 9.54E-07mg/ml . At the same time, a blank control without polymyocytes was set. 2 duplicate wells for each concentration, 100 μl per well;
9.再分别加入“步骤7”的备用细胞,每孔100μl;9. Add the spare cells from "Step 7" separately, 100 μl per well;
10.置饱和湿度、5%二氧化碳、37℃二氧化碳培养箱培养72小时;10. Incubate in a saturated humidity, 5% carbon dioxide, 37°C carbon dioxide incubator for 72 hours;
11.培养结束前4hr,每孔分别加入CCK8检测试剂10μl;11. 4 hours before the end of the culture, add 10 μl of CCK8 detection reagent to each well;
12.培养结束,用板式酶标仪检测OD450值。12. At the end of the culture, use a plate reader to detect the OD 450 value.
结果:result:
据检测的OD450值,用Graphpad Prism5软件,以聚肌胞浓度为横坐标,对应的OD值为纵坐标制作细胞的增殖曲线;图1是“聚肌胞最适浓度的选择”图。According to the detected OD 450 value, use the Graphpad Prism5 software to make the cell proliferation curve with the polymyocyte concentration as the abscissa and the corresponding OD value as the ordinate.
结论:in conclusion:
据OD450值,用Graphpad Prism5软件制作细胞的增殖曲线(柱状图)。According to the OD 450 value, the cell proliferation curve (bar graph) was made with Graphpad Prism5 software.
聚肌胞终浓度分别为1、0.25、0.0625mg/ml有明显刺激DC-CIK细胞增殖的作用。The final concentrations of polymyocytes were 1, 0.25, and 0.0625 mg/ml, respectively, which significantly stimulated the proliferation of DC-CIK cells.
实施例2聚肌胞对DC-CIK增殖的影响Example 2 The effect of polymyocytes on the proliferation of DC-CIK
材料:Material:
1.聚肌胞(上海源叶生物科技有限公司;Cas:24939-03-5)1. Polymuscular (Shanghai Yuanye Biotechnology Co., Ltd.; Cas: 24939-03-5)
2.Hyclone RPMI 16402. Hyclone RPMI 1640
3.健康自愿者外周抗凝血3. Peripheral anticoagulation in healthy volunteers
4.条件培养基:含IL-2 100000IU/ml、抗人CD3人源化嵌合抗体1000μg/ml、IL-1α10000ng/ml、GMCSF10000ng/ml、γ干扰素10000ng/ml。4. Conditioned medium: 100000 IU/ml IL-2, 1000 μg/ml anti-human CD3 humanized chimeric antibody, 10000 ng/ml IL-1α, 10000 ng/ml GMCSF, 10000 ng/ml gamma interferon.
5.板式酶标仪(深圳雷杜生命科学股份有限公司,型号:RT-6100)、二氧化碳孵箱(Thermo SCIENTIFIC,型号:3111)、洁净工作台(Thermo SCIENTIFIC,型号:1379)等5. Plate microplate reader (Shenzhen Redu Life Science Co., Ltd., model: RT-6100), carbon dioxide incubator (Thermo SCIENTIFIC, model: 3111), clean workbench (Thermo SCIENTIFIC, model: 1379), etc.
准备:Prepare:
1.对照组DC-CIK培养基的制作:分别取“方法步骤2”的含2%自体血浆的HycloneRPMI 1640 各250ml培养基,加入DC-CIK条件培养基2.5ml,即成Control DC-CIK-HycloneRPMI 1640培养基。1. Preparation of control DC-CIK medium: take 250ml of HycloneRPMI 1640 medium containing 2% autologous plasma from "Method Step 2", add 2.5ml of DC-CIK conditioned medium to form Control DC-CIK- HycloneRPMI 1640 medium.
2.实验组DC-CIK培养基的制作:分别取“方法步骤2”的含2%自体血浆的HycloneRPMI 1640 各250ml培养基,加入DC-CIK条件培养基2.5ml。再加入聚肌胞,使终浓度为1mg/ml,即成DC-CIK-Hyclone RPMI 1640培养基。2. Preparation of DC-CIK medium in experimental group: 250ml of HycloneRPMI 1640 medium containing 2% autologous plasma of "method step 2" was taken, and 2.5ml of DC-CIK conditioned medium was added. Then polymyocytes were added to make the final concentration 1 mg/ml to form DC-CIK-Hyclone RPMI 1640 medium.
方法:method:
1.无菌采集健康自愿者外周血20ml,1000IU肝素抗凝,并轻轻充分混匀。转移至50ml离心管,2000rpm离心沉淀10分钟;1. Aseptically collect 20ml of peripheral blood from healthy volunteers, anticoagulate with 1000IU heparin, and mix gently and thoroughly. Transfer to a 50ml centrifuge tube and centrifuge at 2000rpm for 10 minutes;
2.收集上层血浆约10ml血浆加入500ml Hyclone RPMI-1640中,等分为2瓶,即每瓶250ml。备用;2. Collect about 10ml of upper plasma and add it to 500ml of Hyclone RPMI-1640, and divide it into 2 bottles, ie each bottle is 250ml. spare;
3.血球沉淀物,加生理盐水混悬至20ml;3. Blood cell sediment, add normal saline to suspend to 20ml;
4.将“步骤3”样品用吸管沿管壁缓慢地加到2支已预先加入2.5ml淋巴细胞分层液的15ml 一次性无菌塑料离心管中,使加入的血液重叠于分层液上,形成明显的分界面(注意不要摇动);4. Slowly add the "
5.置水平离心机2000rpm离心20min,小心取出;5. Centrifuge at 2000rpm for 20min in a horizontal centrifuge and take it out carefully;
6.用一次性无菌塑料巴氏吸管轻轻插入离心管中,吸出白色云雾状的单个核细胞层,转移至50ml离心管中,加生理盐水至50ml,充分混匀细胞,2000rpm离心5min,如此反复洗涤2次;第三次用生理盐水悬浮细胞,苔盼蓝染色计数、并分为两份,使每份细胞均含2.5×106个单个核细胞,再次2000rpm离心5min,弃上清;6. Gently insert a disposable sterile plastic Pasteur pipette into the centrifuge tube, suck out the white cloudy mononuclear cell layer, transfer it to a 50ml centrifuge tube, add normal saline to 50ml, mix the cells thoroughly, and centrifuge at 2000rpm for 5min. Repeat the washing twice; for the third time, suspend the cells with normal saline, count with trypan blue, and divide them into two parts, so that each part of the cells contains 2.5×10 6 mononuclear cells, centrifuge again at 2000 rpm for 5 min, and discard the supernatant. ;
7.根据细胞计数结果,分别用Control DC-CIK-Hyclone RPMI 1640和DC-CIK-Hyclone RPMI 1640培养基5ml悬浮细胞沉淀;7. According to the cell count results, use 5ml of Control DC-CIK-Hyclone RPMI 1640 and DC-CIK-Hyclone RPMI 1640 medium to suspend the cells respectively;
8.分别转移至25cm2细胞培养瓶中,并作好标记;8. Transfer to 25cm 2 cell culture flasks and mark them;
9.置饱和湿度、5%二氧化碳、37℃二氧化碳培养箱培养0、72、120、168小时;9. Incubate in a saturated humidity, 5% carbon dioxide, 37°C carbon dioxide incubator for 0, 72, 120, 168 hours;
10.培养结束分别取样,苔盼蓝染色计数。10. Samples were taken at the end of the culture and counted by trypan blue staining.
结果:result:
根据计数结果,用Graphpad Prism5软件,以细胞培养天数为横坐标,以细胞数目为纵坐标,制作柱状图;图2是“聚肌胞对DC-CIK增殖的影响”图。According to the counting results, use Graphpad Prism5 software to make a histogram with the days of cell culture as the abscissa and the number of cells as the ordinate; Figure 2 is the graph of "the effect of polymyocytes on the proliferation of DC-CIK".
结论:in conclusion:
根据以上结果,可以看出随着培养时间的增加,聚肌胞组和无聚肌胞组细胞数量均有所增加。两者第三天细胞数量无明显差异,但聚肌胞组细胞数量在第五天和第七天较无聚肌胞组有显著差异,聚肌胞组细胞数量明显高于无聚肌胞组。According to the above results, it can be seen that with the increase of culture time, the number of cells in the polymyocyte group and the non-polymyocyte group both increased. There was no significant difference in the number of cells on the third day between the two groups, but the number of cells in the polymyocytes group was significantly different on the fifth and seventh days compared with the non-aggregate myoblast group. .
实施例3聚肌胞诱导的高效DC-CIK表型鉴定Example 3 Phenotypic identification of high-efficiency DC-CIK induced by polymyocytes
材料:Material:
1.聚肌胞(上海源叶生物科技有限公司;Cas:24939-03-5)1. Polymuscular (Shanghai Yuanye Biotechnology Co., Ltd.; Cas: 24939-03-5)
2.Hyclone RPMI 16402. Hyclone RPMI 1640
3.健康自愿者外周抗凝血3. Peripheral anticoagulation in healthy volunteers
4.条件培养基:含IL-2 100000IU/ml、抗人CD3人源化嵌合抗体1000μg/ml、IL-1α10000ng/ml、GMCSF10000ng/ml、γ干扰素10000ng/ml。4. Conditioned medium: 100000 IU/ml IL-2, 1000 μg/ml anti-human CD3 humanized chimeric antibody, 10000 ng/ml IL-1α, 10000 ng/ml GMCSF, 10000 ng/ml gamma interferon.
5.流式标记抗体CD3:BV510,CD4:BUV496,CD8:BV7865. Flow labeled antibodies CD3:BV510, CD4:BUV496, CD8:BV786
6.其它:流式细胞仪(BD,型号:FACSymphony)、二氧化碳孵箱(ThermoSCIENTIFIC,型号:3111)、洁净工作台(Thermo SCIENTIFIC,型号:1379)等6. Others: flow cytometer (BD, model: FACSymphony), carbon dioxide incubator (Thermo SCIENTIFIC, model: 3111), clean bench (Thermo SCIENTIFIC, model: 1379), etc.
准备:Prepare:
1.对照组DC-CIK培养基的制作:取“方法步骤2”的含2%自体血浆的Hyclone RPMI1640 250ml培养基,加入DC-CIK条件培养基2.5ml即成DC-CIK条件培养基,即成ControlDC-CIK-Hyclone RPMI 1640培养基。1. Preparation of DC-CIK medium for control group: take 250ml of Hyclone RPMI1640 medium containing 2% autologous plasma from "Method Step 2", add 2.5ml of DC-CIK conditioned medium to obtain DC-CIK conditioned medium, namely into ControlDC-CIK-Hyclone RPMI 1640 medium.
2.实验组DC-CIK培养基的制作:取“方法步骤2”的含2%自体血浆的Hyclone RPMI1640 250ml培养基,加入DC-CIK条件培养基2.5ml。再加入聚肌胞,使终浓度为1mg/ml,即成DC-CIK-Hyclone RPMI 1640培养基。2. Preparation of DC-CIK medium for experimental group: take 250ml of Hyclone RPMI1640 medium containing 2% autologous plasma from "Method step 2", and add 2.5ml of DC-CIK conditioned medium. Then polymyocytes were added to make the final concentration 1 mg/ml to form DC-CIK-Hyclone RPMI 1640 medium.
方法:method:
1.无菌采集健康自愿者外周血20ml,1000IU肝素抗凝,并轻轻充分混匀。转移至50ml离心管,2000rpm离心沉淀10min;1. Aseptically collect 20ml of peripheral blood from healthy volunteers, anticoagulate with 1000IU heparin, and mix gently and thoroughly. Transfer to a 50ml centrifuge tube and centrifuge at 2000rpm for 10min;
2.收集上层血浆约10ml,分别取12.5ml血浆加入500ml YQ-1640/RPMI-1640中,等分为2 瓶,即每瓶250ml。备用;2. Collect about 10ml of upper plasma, respectively take 12.5ml of plasma and add it to 500ml of YQ-1640/RPMI-1640, and divide into 2 bottles, ie, each bottle is 250ml. spare;
3.血球沉淀物,加生理盐水混悬至20ml;将“步骤3”样品用吸管沿管壁缓慢地加到2 支已预先加入3ml淋巴细胞分层液的15ml一次性无菌塑料离心管中,使加入的血液重叠于分层液上,形成明显的分界面(注意不要摇动);3. Add the blood cell sediment to 20ml of normal saline; slowly add the "
4.置水平离心机2000rpm离心20min,小心取出;4. Centrifuge at 2000rpm for 20min in a horizontal centrifuge and take it out carefully;
5.用一次性无菌塑料巴氏吸管轻轻插入离心管中,吸出白色云雾状的单个核细胞层,转移至50ml离心管中,加生理盐水至50ml,充分混匀细胞,2000rpm离心5min,如此反复洗涤2次;第三次用生理盐水悬浮细胞,苔盼蓝染色计数、并分为两份,使每份细胞均含2.5×106个单个核细胞,再次2000rpm离心5min,弃上清;5. Gently insert a disposable sterile plastic Pasteur pipette into the centrifuge tube, suck out the white cloudy mononuclear cell layer, transfer it to a 50ml centrifuge tube, add normal saline to 50ml, mix the cells thoroughly, and centrifuge at 2000rpm for 5min. Repeat the washing twice; for the third time, suspend the cells with normal saline, count with trypan blue, and divide them into two parts, so that each part of the cells contains 2.5×10 6 mononuclear cells, centrifuge again at 2000 rpm for 5 min, and discard the supernatant. ;
6.根据细胞计数结果,分别用Control DC-CIK-Hyclone RPMI 1640DC-CIK-Hyclone RPMI 1640培养基5ml悬浮细胞沉淀;6. According to the cell count results, use Control DC-CIK-Hyclone RPMI 1640DC-CIK-Hyclone RPMI 1640 medium to suspend the cell pellet in 5ml;
7.分别转移至25cm2细胞培养瓶中,并作好标记;7. Transfer to 25cm 2 cell culture flasks and mark them;
8.置饱和湿度、5%二氧化碳、37℃、二氧化碳培养箱培养0、72、120、168小时;8. Incubate in a saturated humidity, 5% carbon dioxide, 37°C, carbon dioxide incubator for 0, 72, 120, 168 hours;
9.培养结束分别取样用CD3、CD4、CD8荧光抗体进行常规标记;9. At the end of the culture, samples were taken for routine labeling with CD3, CD4, and CD8 fluorescent antibodies;
10.用流式细胞仪进行检测。10. Detect with flow cytometer.
结果:result:
选择流式CD3+CD4-CD8+细胞,以CD4为横坐标,CD8为纵坐标制作分区图。图3是“聚肌胞诱导的高效DC-CIK表型鉴定”图。Select CD3+CD4-CD8+ cells by flow cytometry, and make a partition map with CD4 as the abscissa and CD8 as the ordinate. Figure 3 is a graph of "Polymyocyte-induced high-efficiency DC-CIK phenotype identification".
结论:in conclusion:
随着细胞培养时间的增加,CD3+CD4-CD8+细胞数量均不同程度增加。聚肌胞组较无聚肌胞 CD3+CD4-CD8+细胞数量明显增加,具有显著差异。With the increase of cell culture time, the number of CD3+CD4-CD8+ cells increased to varying degrees. The number of CD3+CD4-CD8+ cells in the polymyocytes group was significantly higher than that without polymyocytes, and there was a significant difference.
实施例4聚肌胞诱导的高效DC-CIK杀瘤活性Example 4 High-efficiency DC-CIK tumoricidal activity induced by polymyocytes
材料:Material:
1.聚肌胞:(上海源叶生物科技有限公司;Cas:24939-03-5)1. Poly muscle cells: (Shanghai Yuanye Biotechnology Co., Ltd.; Cas: 24939-03-5)
2.Hyclone RPMI 16402. Hyclone RPMI 1640
3.健康自愿者外周抗凝血3. Peripheral anticoagulation in healthy volunteers
4.条件培养基:含IL-2 100000IU/ml、抗人CD3人源化嵌合抗体1000μg/ml、IL-1α10000ng/ml、GMCSF10000ng/ml、γ干扰素10000ng/ml。4. Conditioned medium: 100000 IU/ml IL-2, 1000 μg/ml anti-human CD3 humanized chimeric antibody, 10000 ng/ml IL-1α, 10000 ng/ml GMCSF, 10000 ng/ml gamma interferon.
5.人类髓性白血病细胞株K5625. Human myeloid leukemia cell line K562
6.CCK8检测试剂盒(北仁化学科技(北京)有限公司,型号:CK04)6. CCK8 detection kit (Beijing Chemical Technology (Beijing) Co., Ltd., model: CK04)
7.板式酶标仪(深圳雷杜生命科学股份有限公司,型号:RT-6100)、二氧化碳孵箱(Thermo SCIENTIFIC,型号:3111)、洁净工作台(Thermo SCIENTIFIC,型号:1379)等。7. Plate microplate reader (Shenzhen Redu Life Science Co., Ltd., model: RT-6100), carbon dioxide incubator (Thermo SCIENTIFIC, model: 3111), clean workbench (Thermo SCIENTIFIC, model: 1379), etc.
准备:Prepare:
1.对照组DC-CIK培养基的制作:取“方法步骤2”的含2%自体血浆的Hyclone RPMI1640 250ml培养基,加入DC-CIK条件培养基2.5ml即成Control DC-CIK-Hyclone RPMI1640 培养基。1. Preparation of control DC-CIK medium: Take 250ml of Hyclone RPMI1640 medium containing 2% autologous plasma from "Method step 2" and add 2.5ml of DC-CIK conditioned medium to form Control DC-CIK-Hyclone RPMI1640 culture base.
2.实验组DC-CIK培养基的制作:取“方法步骤2”的含2%自体血浆的Hyclone RPMI1640 250ml培养基,加入DC-CIK条件培养基2.5ml。再加入聚肌胞,使终浓度为1mg/ml,即成DC-CIK-Hyclone RPMI 1640培养基。2. Preparation of DC-CIK medium for experimental group: take 250ml of Hyclone RPMI1640 medium containing 2% autologous plasma from "Method step 2", and add 2.5ml of DC-CIK conditioned medium. Then polymyocytes were added to make the final concentration 1 mg/ml to form DC-CIK-Hyclone RPMI 1640 medium.
3.靶细胞:取处于对数生长期的K562细胞,苔盼蓝染色计数并常规离心洗涤。然后用含 5%新生小牛血清的Hyclone RPMI 1640培养基调细胞浓度为4×104个/ml,备用。3. Target cells: K562 cells in logarithmic growth phase were taken, counted by trypan blue staining, and washed by conventional centrifugation. Then, the cell concentration was adjusted to 4×10 4 cells/ml with Hyclone RPMI 1640 medium containing 5% newborn calf serum, for use.
方法:method:
1.无菌采集健康自愿者外周血20ml,1000IU肝素抗凝,并轻轻充分混匀。转移至50ml离心管,2000rpm离心沉淀10分钟;1. Aseptically collect 20ml of peripheral blood from healthy volunteers, anticoagulate with 1000IU heparin, and mix gently and thoroughly. Transfer to a 50ml centrifuge tube and centrifuge at 2000rpm for 10 minutes;
2.收集上层血浆约10ml,加入500ml RPMI-1640中,等分为2瓶,即每瓶250ml。备用;2. Collect about 10ml of upper plasma, add it to 500ml of RPMI-1640, and divide it into 2 bottles, ie each bottle is 250ml. spare;
3.血球沉淀物,加生理盐水混悬至20ml;3. Blood cell sediment, add normal saline to suspend to 20ml;
4.将“步骤3”样品用吸管沿管壁缓慢地加到2支已预先加入2.5ml淋巴细胞分层液的15ml 一次性无菌塑料离心管中,使加入的血液重叠于分层液上,形成明显的分界面(注意不要摇动);4. Slowly add the "
5.置水平离心机2000rpm离心20min,小心取出;5. Centrifuge at 2000rpm for 20min in a horizontal centrifuge and take it out carefully;
6.用一次性无菌塑料巴氏吸管轻轻插入离心管中,吸出白色云雾状的单个核细胞层,转移至50ml离心管中,加生理盐水至50ml,充分混匀细胞,2000rpm离心5min,如此反复洗涤2次;第三次用生理盐水悬浮细胞,苔盼蓝染色计数、并分为两份,使每份细胞均含2.5×106个单个核细胞,再次2000rpm离心5min,弃上清;6. Gently insert a disposable sterile plastic Pasteur pipette into the centrifuge tube, suck out the white cloudy mononuclear cell layer, transfer it to a 50ml centrifuge tube, add normal saline to 50ml, mix the cells thoroughly, and centrifuge at 2000rpm for 5min. Repeat the washing twice; for the third time, suspend the cells with normal saline, count with trypan blue, and divide them into two parts, so that each part of the cells contains 2.5×10 6 mononuclear cells, centrifuge again at 2000 rpm for 5 min, and discard the supernatant. ;
7.根据细胞计数结果,分别用Control DC-CIK-Hyclone RPMI 1640和DC-CIK-Hyclone RPMI 1640培养基5ml悬浮细胞沉淀;7. According to the cell count results, use 5ml of Control DC-CIK-Hyclone RPMI 1640 and DC-CIK-Hyclone RPMI 1640 medium to suspend the cells respectively;
8.分别转移至25cm2细胞培养瓶中,并作好标记;8. Transfer to 25cm 2 cell culture flasks and mark them;
9.置饱和湿度、5%二氧化碳、37℃二氧化碳培养箱培养0、72、120、168小时;9. Incubate in a saturated humidity, 5% carbon dioxide, 37°C carbon dioxide incubator for 0, 72, 120, 168 hours;
10.培养结束前分别取样用生理盐水洗涤3次,苔盼蓝染色分别计数。分别各取5×106个细胞,均用Hyclone RPMI 1640悬浮细胞。2000rpm、离心沉淀5min。沉淀物再次用含5%新生小牛血清的Hyclone RPMI 1640培养基2.5ml重悬细胞,使终浓度为2×106个/ml;10. Before the end of the culture, samples were taken and washed 3 times with normal saline, and were counted by trypan blue staining. 5×10 6 cells were taken respectively, and the cells were suspended in Hyclone RPMI 1640. 2000rpm, centrifugation for 5min. The pellet was resuspended with 2.5ml of Hyclone RPMI 1640 medium containing 5% newborn calf serum to make the final concentration 2×10 6 cells/ml;
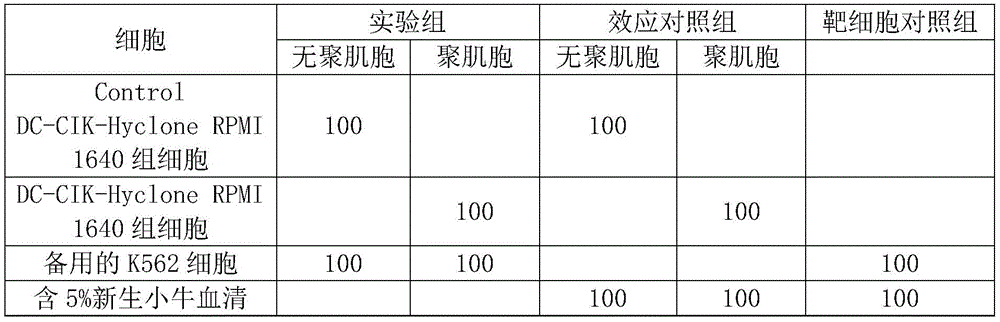
11.取圆底一次性无菌的96孔细胞培养板,如下表1分别加样。即:Control DC-CIK-Hyclone RPMI 1640组细胞杀瘤活性检测加样为:实验组每孔加入浓度为2× 106个/ml的Control DC-CIK-Hyclone RPMI 1640组细胞100μl,同时加入浓度为4× 104个/ml的K562细胞100μl;效应对照组每孔加入浓度为2×106个/ml的Control DC-CIK-HycloneRPMI 1640组细胞100μl,同时加入含5%新生小牛血清的Hyclone RPMI 1640 100μl;靶细胞组则仅每孔加入浓度为4×104个/ml的K562细胞100μl和含5%新生小牛血清的HycloneRPMI 1640 100μl即可。11. Take a round-bottomed disposable sterile 96-well cell culture plate and add samples to it in Table 1 below. That is: for the control DC-CIK-Hyclone RPMI 1640 group cell tumoricidal activity test, add the sample as follows: in the experimental group, add 100 μl of Control DC-CIK-Hyclone RPMI 1640 group cells at a concentration of 2×10 6 cells/ml to each well, and at the same time add the concentration of 2×10 6 cells/ml. 100 μl of K562 cells at 4×10 4 cells/ml; 100 μl of Control DC-CIK-HycloneRPMI 1640 cells at a concentration of 2×10 6 cells/ml were added to each well of the effect control group, and at the same time, 5% newborn calf serum was added. Hyclone RPMI 1640 100 μl; for the target cell group, only 100 μl of K562 cells with a concentration of 4×10 4 cells/ml and 100 μl of HycloneRPMI 1640 containing 5% newborn calf serum can be added to each well.
12.置饱和湿度、5%二氧化碳、37℃二氧化碳培养箱培养16小时;每孔加入CCK8,每孔 10μl。4小时后,用板式酶标仪测定OD450值。12. Incubate in a saturated humidity, 5% carbon dioxide, 37°C carbon dioxide incubator for 16 hours; add CCK8 to each well, 10 μl per well. After 4 hours, the OD 450 value was determined with a plate reader.
表1 Hyclone 1640培养基加样表Table 1 Hyclone 1640 medium loading table
备注:每组均作四复孔;单位:μl。Remarks: Four duplicate wells were made in each group; unit: μl.
结果:result:
1、结果计算:杀瘤百分率=[1-(实验组OD450-效应对照组OD450)/靶细胞对照组OD450]×100%;1. Calculation of results: tumor killing percentage=[1-(OD 450 of experimental group-OD 450 of effector control group)/OD 450 of target cell control group]×100%;
2、将各组检测数据代入以上公式,即得各组的杀瘤活性;2. Substitute the detection data of each group into the above formula to obtain the tumoricidal activity of each group;
3、将各组杀瘤活性百分率,用Graphpad Prism5软件制作杀瘤活性柱状图;图4是“聚肌胞诱导的高效DC-CIK杀瘤活性”图。3. Using the Graphpad Prism5 software to make a histogram of the tumoricidal activity of the percentages of each group's tumoricidal activity; Fig. 4 is a graph of "highly effective DC-CIK tumoricidal activity induced by polymyocytes".
结论:in conclusion:
根据上述结果可以看出,随着细胞培养时间增加,两组细胞杀瘤活性均有不同程度增加,聚肌胞组较无聚肌胞组杀瘤活性增加更为显著,具有显著差异。According to the above results, it can be seen that with the increase of cell culture time, the tumoricidal activity of the two groups of cells increased to different degrees, and the tumoricidal activity of the polymyocytes group increased more significantly than that of the non-polymyocyte group, and there was a significant difference.
Claims (2)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010255218.7A CN111733129A (en) | 2020-04-02 | 2020-04-02 | A method for the induction of high-efficiency DC-CIK cells by polymyocytes |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010255218.7A CN111733129A (en) | 2020-04-02 | 2020-04-02 | A method for the induction of high-efficiency DC-CIK cells by polymyocytes |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN111733129A true CN111733129A (en) | 2020-10-02 |
Family
ID=72646867
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010255218.7A Pending CN111733129A (en) | 2020-04-02 | 2020-04-02 | A method for the induction of high-efficiency DC-CIK cells by polymyocytes |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111733129A (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN113846065A (en) * | 2021-10-14 | 2021-12-28 | 北京创世客生物技术有限公司 | Use of engineered CIK immune cells in the treatment of cancer |
| WO2022199138A1 (en) * | 2021-03-24 | 2022-09-29 | 深圳先进技术研究院 | Nano artificial antigen presenting cell, preparation method therefor and use thereof |
| CN118987032A (en) * | 2024-08-16 | 2024-11-22 | 广州百吉生物制药有限公司 | Anti-aging pharmaceutical composition and application thereof |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103710308A (en) * | 2013-12-10 | 2014-04-09 | 吴炯 | Method for inducing DC-CIK by utilizing muramyl dipeptide |
| CN105132372A (en) * | 2015-08-28 | 2015-12-09 | 淄博金砖生物科技有限公司 | Application of DC-CIK cell inducing compound in tumor cell immunotherapy |
| CN106222140A (en) * | 2016-08-04 | 2016-12-14 | 英普乐孚生物技术(上海)有限公司 | A kind of NK cell non-serum culture medium and compound method thereof |
| CN107574149A (en) * | 2016-07-05 | 2018-01-12 | 上海细胞治疗研究院 | A kind of efficient rush maturation method of BMDC and application thereof |
| CN107693789A (en) * | 2017-07-31 | 2018-02-16 | 华中科技大学同济医学院附属协和医院 | One kind includes cell-targeting antibody and Poly (I:C compound) and its preparation method and application |
-
2020
- 2020-04-02 CN CN202010255218.7A patent/CN111733129A/en active Pending
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103710308A (en) * | 2013-12-10 | 2014-04-09 | 吴炯 | Method for inducing DC-CIK by utilizing muramyl dipeptide |
| CN105132372A (en) * | 2015-08-28 | 2015-12-09 | 淄博金砖生物科技有限公司 | Application of DC-CIK cell inducing compound in tumor cell immunotherapy |
| CN107574149A (en) * | 2016-07-05 | 2018-01-12 | 上海细胞治疗研究院 | A kind of efficient rush maturation method of BMDC and application thereof |
| CN106222140A (en) * | 2016-08-04 | 2016-12-14 | 英普乐孚生物技术(上海)有限公司 | A kind of NK cell non-serum culture medium and compound method thereof |
| CN107693789A (en) * | 2017-07-31 | 2018-02-16 | 华中科技大学同济医学院附属协和医院 | One kind includes cell-targeting antibody and Poly (I:C compound) and its preparation method and application |
Non-Patent Citations (3)
| Title |
|---|
| H NAVABI等: "A clinical grade poly I:C-analogue (Ampligen) promotes optimal DC maturation and Th1-type T cell responses of healthy donors and cancer patients in vitro" * |
| 吴学杰;王贵强;: "树突状细胞疫苗在肝病治疗中的研究进展" * |
| 王静: "OK-432联合聚肌胞诱导小鼠树突状细胞成熟及其抗损伤作用" * |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022199138A1 (en) * | 2021-03-24 | 2022-09-29 | 深圳先进技术研究院 | Nano artificial antigen presenting cell, preparation method therefor and use thereof |
| CN113846065A (en) * | 2021-10-14 | 2021-12-28 | 北京创世客生物技术有限公司 | Use of engineered CIK immune cells in the treatment of cancer |
| CN113846065B (en) * | 2021-10-14 | 2022-09-06 | 冬青南京生物科技有限公司 | Use of engineered CIK immune cells in the treatment of cancer |
| CN118987032A (en) * | 2024-08-16 | 2024-11-22 | 广州百吉生物制药有限公司 | Anti-aging pharmaceutical composition and application thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN105087488A (en) | Preparation method and application of DC-CIK cell induced by tumor antigen | |
| CN108588022B (en) | Method for enriching human CD4+ and CD8+ TCM cells through in vitro culture | |
| CN102816735A (en) | Method for culturing autologous peripheral blood lymphocytes | |
| CN111733129A (en) | A method for the induction of high-efficiency DC-CIK cells by polymyocytes | |
| CN108251365A (en) | Immune cell media system | |
| CN103301449A (en) | Preparation method of large-scale culture dendritic cell vaccine and application thereof | |
| CN105524883A (en) | Capri cell and preparation method thereof | |
| JP2022028818A (en) | Methods for Activated Dendritic Cell Compositions and Immunotherapeutic Treatments for Subjects with Advanced Cancer | |
| CN107502590A (en) | A kind of method of human umbilical cord's blood candidate stem cell efficient amplification NK cells | |
| CN106222141A (en) | NK cell culture fluid and cell culture processes | |
| JP4275680B2 (en) | Culture methods for lymphocyte activity / proliferation | |
| CN103981144B (en) | The preparation method of autoserum antigen sensibilization DC CIK cells | |
| CN105296421B (en) | The T cell and preparation method of a kind of activation of bispecific antibody and application | |
| KR102763373B1 (en) | Optimally activated dendritic cells that induce enhanced or increased anti-tumor immune responses | |
| CN107119015B (en) | Exosome, preparation method thereof and application thereof in preparation of medicine for treating lung cancer | |
| CN106754704A (en) | The method of the external evoked amplification of immunocyte | |
| CN102443567A (en) | In-vitro amplification method of tumor specific gamma T lymphocytes | |
| CN114058580A (en) | Methods for Proliferating Natural Killer Cells and Natural Killer T Cells in Vitro | |
| US20250255904A1 (en) | Identification, in vitro amplification, and application method of memory cd8 t cells specific for tumor antigen | |
| CN106479973A (en) | A kind of external IAK immunocyte cultural method | |
| CN114292813B (en) | Media formulation for activating global anti-tumor immune system and method for preparing agonist-activated global immune effector cells | |
| Hironaka et al. | Essential requirement of toll-like receptor 4 expression on CD11c+ cells for locoregional immunotherapy of malignant ascites using a streptococcal preparation OK-432 | |
| CN104651313A (en) | Cascade-activated immune cell as well as preparation method and application thereof | |
| CN114657124A (en) | Preparation method of compound immune cells with high killing capacity on tumor cells | |
| WO2023125696A2 (en) | Activated lymphocyte expansion method having stable and controllable quality, and anti-tumor use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20201002 |
|
| WD01 | Invention patent application deemed withdrawn after publication |