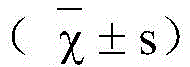CN111714475A - Use of and honokiol in the preparation of medicaments for treating and/or preventing rosacea - Google Patents
Use of and honokiol in the preparation of medicaments for treating and/or preventing rosacea Download PDFInfo
- Publication number
- CN111714475A CN111714475A CN202010832764.2A CN202010832764A CN111714475A CN 111714475 A CN111714475 A CN 111714475A CN 202010832764 A CN202010832764 A CN 202010832764A CN 111714475 A CN111714475 A CN 111714475A
- Authority
- CN
- China
- Prior art keywords
- honokiol
- rosacea
- group
- preparation
- treating
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- FVYXIJYOAGAUQK-UHFFFAOYSA-N honokiol Chemical compound C1=C(CC=C)C(O)=CC=C1C1=CC(CC=C)=CC=C1O FVYXIJYOAGAUQK-UHFFFAOYSA-N 0.000 title claims abstract description 47
- 201000004700 rosacea Diseases 0.000 title claims abstract description 37
- 241001303601 Rosacea Species 0.000 title claims abstract description 36
- BYTORXDZJWWIKR-UHFFFAOYSA-N Hinokiol Natural products CC(C)c1cc2CCC3C(C)(CO)C(O)CCC3(C)c2cc1O BYTORXDZJWWIKR-UHFFFAOYSA-N 0.000 title claims abstract description 28
- VVOAZFWZEDHOOU-UHFFFAOYSA-N honokiol Natural products OC1=CC=C(CC=C)C=C1C1=CC(CC=C)=CC=C1O VVOAZFWZEDHOOU-UHFFFAOYSA-N 0.000 title claims abstract description 28
- 239000003814 drug Substances 0.000 title claims abstract description 20
- 238000002360 preparation method Methods 0.000 title claims abstract description 18
- 210000004969 inflammatory cell Anatomy 0.000 claims abstract description 25
- 206010015150 Erythema Diseases 0.000 claims abstract description 19
- 230000008595 infiltration Effects 0.000 claims abstract description 9
- 238000001764 infiltration Methods 0.000 claims abstract description 9
- 229940079593 drug Drugs 0.000 claims abstract description 8
- 239000006071 cream Substances 0.000 claims description 5
- 239000007788 liquid Substances 0.000 claims description 4
- 239000007787 solid Substances 0.000 claims description 2
- 241000699670 Mus sp. Species 0.000 abstract description 17
- 231100000321 erythema Toxicity 0.000 abstract description 17
- 210000000056 organ Anatomy 0.000 abstract description 7
- 210000004027 cell Anatomy 0.000 abstract description 6
- 206010067482 No adverse event Diseases 0.000 abstract description 5
- 206010040844 Skin exfoliation Diseases 0.000 abstract description 5
- 230000005856 abnormality Effects 0.000 abstract description 5
- 230000035618 desquamation Effects 0.000 abstract description 5
- 230000004580 weight loss Effects 0.000 abstract description 5
- 238000002474 experimental method Methods 0.000 abstract description 4
- BDJRBEYXGGNYIS-UHFFFAOYSA-N nonanedioic acid Chemical compound OC(=O)CCCCCCCC(O)=O BDJRBEYXGGNYIS-UHFFFAOYSA-N 0.000 description 12
- 210000003491 skin Anatomy 0.000 description 9
- 239000011159 matrix material Chemical group 0.000 description 7
- 206010030113 Oedema Diseases 0.000 description 6
- 238000007490 hematoxylin and eosin (H&E) staining Methods 0.000 description 6
- 238000002347 injection Methods 0.000 description 5
- 239000007924 injection Substances 0.000 description 5
- 206010067484 Adverse reaction Diseases 0.000 description 4
- 230000006838 adverse reaction Effects 0.000 description 4
- 238000010171 animal model Methods 0.000 description 4
- CDQSJQSWAWPGKG-UHFFFAOYSA-N butane-1,1-diol Chemical compound CCCC(O)O CDQSJQSWAWPGKG-UHFFFAOYSA-N 0.000 description 4
- 230000001225 therapeutic effect Effects 0.000 description 4
- 210000001519 tissue Anatomy 0.000 description 4
- 241000699666 Mus <mouse, genus> Species 0.000 description 3
- 210000004207 dermis Anatomy 0.000 description 3
- 208000035859 Drug effect increased Diseases 0.000 description 2
- 229940000406 drug candidate Drugs 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 239000003777 experimental drug Substances 0.000 description 2
- 230000001815 facial effect Effects 0.000 description 2
- 238000000034 method Methods 0.000 description 2
- 230000001575 pathological effect Effects 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 229910021642 ultra pure water Inorganic materials 0.000 description 2
- 239000012498 ultrapure water Substances 0.000 description 2
- NHBKXEKEPDILRR-UHFFFAOYSA-N 2,3-bis(butanoylsulfanyl)propyl butanoate Chemical compound CCCC(=O)OCC(SC(=O)CCC)CSC(=O)CCC NHBKXEKEPDILRR-UHFFFAOYSA-N 0.000 description 1
- 208000002874 Acne Vulgaris Diseases 0.000 description 1
- 238000011725 BALB/c mouse Methods 0.000 description 1
- 208000017667 Chronic Disease Diseases 0.000 description 1
- IECPWNUMDGFDKC-UHFFFAOYSA-N Fusicsaeure Natural products C12C(O)CC3C(=C(CCC=C(C)C)C(O)=O)C(OC(C)=O)CC3(C)C1(C)CCC1C2(C)CCC(O)C1C IECPWNUMDGFDKC-UHFFFAOYSA-N 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 206010033733 Papule Diseases 0.000 description 1
- 206010037888 Rash pustular Diseases 0.000 description 1
- QJJXYPPXXYFBGM-LFZNUXCKSA-N Tacrolimus Chemical compound C1C[C@@H](O)[C@H](OC)C[C@@H]1\C=C(/C)[C@@H]1[C@H](C)[C@@H](O)CC(=O)[C@H](CC=C)/C=C(C)/C[C@H](C)C[C@H](OC)[C@H]([C@H](C[C@H]2C)OC)O[C@@]2(O)C(=O)C(=O)N2CCCC[C@H]2C(=O)O1 QJJXYPPXXYFBGM-LFZNUXCKSA-N 0.000 description 1
- 206010043189 Telangiectasia Diseases 0.000 description 1
- 206010000496 acne Diseases 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 238000001574 biopsy Methods 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 230000002498 deadly effect Effects 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 210000002615 epidermis Anatomy 0.000 description 1
- 238000011010 flushing procedure Methods 0.000 description 1
- IECPWNUMDGFDKC-MZJAQBGESA-N fusidic acid Chemical compound O[C@@H]([C@@H]12)C[C@H]3\C(=C(/CCC=C(C)C)C(O)=O)[C@@H](OC(C)=O)C[C@]3(C)[C@@]2(C)CC[C@@H]2[C@]1(C)CC[C@@H](O)[C@H]2C IECPWNUMDGFDKC-MZJAQBGESA-N 0.000 description 1
- 229960004675 fusidic acid Drugs 0.000 description 1
- 230000004047 hyperresponsiveness Effects 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- VAOCPAMSLUNLGC-UHFFFAOYSA-N metronidazole Chemical compound CC1=NC=C([N+]([O-])=O)N1CCO VAOCPAMSLUNLGC-UHFFFAOYSA-N 0.000 description 1
- 229960000282 metronidazole Drugs 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- 239000002547 new drug Substances 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 208000029561 pustule Diseases 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 230000000306 recurrent effect Effects 0.000 description 1
- 239000012192 staining solution Substances 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 229960001967 tacrolimus Drugs 0.000 description 1
- QJJXYPPXXYFBGM-SHYZHZOCSA-N tacrolimus Natural products CO[C@H]1C[C@H](CC[C@@H]1O)C=C(C)[C@H]2OC(=O)[C@H]3CCCCN3C(=O)C(=O)[C@@]4(O)O[C@@H]([C@H](C[C@H]4C)OC)[C@@H](C[C@H](C)CC(=C[C@@H](CC=C)C(=O)C[C@H](O)[C@H]2C)C)OC QJJXYPPXXYFBGM-SHYZHZOCSA-N 0.000 description 1
- 208000009056 telangiectasis Diseases 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/045—Hydroxy compounds, e.g. alcohols; Salts thereof, e.g. alcoholates
- A61K31/05—Phenols
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/10—Anti-acne agents
Landscapes
- Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Dermatology (AREA)
- Epidemiology (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
本发明提供了和厚朴酚在制备治疗和/或预防玫瑰痤疮的药物中的用途。实验证明,和厚朴酚能够显著减轻玫瑰痤疮小鼠的红斑情况,降低炎症细胞浸润,减少炎症细胞数目,有效治疗玫瑰痤疮。同时,玫瑰痤疮小鼠施用和厚朴酚后不会产生脱屑、体重减轻等不良反应,各器官组织细胞的形态也未发现异常。所以,和厚朴酚在制备治疗和/或预防玫瑰痤疮的药物中具有非常好的应用前景。
The present invention provides the use of honokiol in preparing a medicament for treating and/or preventing rosacea. Experiments have shown that Honokiol can significantly reduce the erythema of rosacea mice, reduce the infiltration of inflammatory cells, reduce the number of inflammatory cells, and effectively treat rosacea. At the same time, after administration of honokiol to rosacea mice, there were no adverse reactions such as desquamation and weight loss, and no abnormality was found in the morphology of cells in various organs. Therefore, honokiol has a very good application prospect in the preparation of medicines for treating and/or preventing rosacea.
Description
技术领域technical field
本发明属于药物领域,具体涉及和厚朴酚在制备治疗和/或预防玫瑰痤疮的药物中的用途。The invention belongs to the field of medicine, in particular to the use of honokiol in the preparation of a medicine for treating and/or preventing rosacea.
背景技术Background technique
玫瑰痤疮是一种主要累及面部皮肤,以炎症和血管神经高反应性为特征的慢性疾病玫瑰痤疮的临床特征包括面部红斑、丘疹和/或脓疱,毛细血管扩张和反复潮红,尽管它不是一种致命的疾病,但对玫瑰痤疮患者的生活质量有不利影响。Rosacea is a chronic disease that primarily affects the facial skin and is characterized by inflammation and vascular-nerve hyperresponsiveness. Clinical features of rosacea include facial erythema, papules, and/or pustules, telangiectasia, and recurrent flushing, although it is not a A deadly disease that adversely affects the quality of life of rosacea sufferers.
临床上玫瑰痤疮的治疗方法多为外用药物,比如他克莫司软膏、夫西地酸乳膏,甲硝唑凝胶等,但是这些药物的治疗效果欠佳、易复发,而且使用过程中会有局部刺激的不良反应,对皮肤造成伤害,严重影响患者的生活质量。因此,如何安全有效地治疗玫瑰痤疮仍是皮肤医学领域的难题。Clinically, the treatment methods for rosacea are mostly topical drugs, such as tacrolimus ointment, fusidic acid cream, metronidazole gel, etc., but the treatment effect of these drugs is not good, easy to relapse, and may cause symptoms during use. There are adverse reactions of local irritation, causing damage to the skin and seriously affecting the quality of life of patients. Therefore, how to treat rosacea safely and effectively is still a difficult problem in the field of dermatology.
所以,研制能够有效治疗玫瑰痤疮、并且不会产生不良反应的新药物具有非常重要的意义。Therefore, it is of great significance to develop new drugs that can effectively treat rosacea without adverse reactions.
发明内容SUMMARY OF THE INVENTION
本发明的目的在于提供和厚朴酚在制备治疗和/或预防玫瑰痤疮的药物中的用途。The purpose of the present invention is to provide the use of honokiol in the preparation of a medicament for treating and/or preventing rosacea.
本发明提供了一种和厚朴酚在制备治疗和/或预防玫瑰痤疮的药物中的用途。The present invention provides the use of honokiol in the preparation of a medicament for treating and/or preventing rosacea.
进一步地,所述药物为外用制剂。Further, the medicine is an external preparation.
进一步地,所述药物为外用液体制剂、固体制剂、乳膏或霜剂。Further, the medicine is a liquid preparation, solid preparation, cream or cream for external use.
进一步地,所述外用液体制剂中,和厚朴酚的浓度为5%~10%mg/mL。Further, in the liquid preparation for external use, the concentration of honokiol is 5%-10% mg/mL.
进一步地,所述药物能够改善皮肤红斑,降低炎症细胞浸润,和/或减少炎症细胞数目。Further, the drug can improve skin erythema, reduce inflammatory cell infiltration, and/or reduce the number of inflammatory cells.
进一步地,所述玫瑰痤疮LL-37诱导的玫瑰痤疮。Further, the rosacea LL-37 induces rosacea.
实验结果表明,和厚朴酚能够显著减轻玫瑰痤疮小鼠的红斑情况,降低炎症细胞浸润,减少炎症细胞数目,有效治疗玫瑰痤疮。同时,玫瑰痤疮小鼠施用和厚朴酚后不会产生脱屑、体重减轻等不良反应,各器官组织细胞的形态也未发现异常。和厚朴酚在制备治疗和/或预防玫瑰痤疮的药物中具有非常好的应用前景。The experimental results show that Honokiol can significantly reduce the erythema of rosacea mice, reduce the infiltration of inflammatory cells, reduce the number of inflammatory cells, and effectively treat rosacea. At the same time, after administration of honokiol to rosacea mice, there were no adverse reactions such as desquamation and weight loss, and no abnormality was found in the morphology of cells in various organs. And honokiol has a very good application prospect in the preparation of medicines for treating and/or preventing rosacea.
显然,根据本发明的上述内容,按照本领域的普通技术知识和惯用手段,在不脱离本发明上述基本技术思想前提下,还可以做出其它多种形式的修改、替换或变更。Obviously, according to the above-mentioned content of the present invention, according to the common technical knowledge and conventional means in the field, without departing from the above-mentioned basic technical idea of the present invention, other various forms of modification, replacement or change can also be made.
以下通过实施例形式的具体实施方式,对本发明的上述内容再作进一步的详细说明。但不应将此理解为本发明上述主题的范围仅限于以下的实例。凡基于本发明上述内容所实现的技术均属于本发明的范围。The above content of the present invention will be further described in detail below through the specific implementation in the form of examples. However, this should not be construed as limiting the scope of the above-mentioned subject matter of the present invention to the following examples. All technologies implemented based on the above content of the present invention belong to the scope of the present invention.
附图说明Description of drawings
图1为各组注射部位的皮肤照片,其中NHK组表示和厚朴酚组。Figure 1 is the skin photos of the injection sites of each group, in which the NHK group represents and the honokiol group.
图2为各组小鼠皮肤组织HE染色病理学图片,其中NHK组表示和厚朴酚组。Figure 2 is the HE staining pathological pictures of mouse skin tissue in each group, in which the NHK group and the honokiol group are represented.
具体实施方式Detailed ways
本发明所用原料与设备均为已知产品,通过购买市售产品所得。The raw materials and equipment used in the present invention are all known products, obtained by purchasing commercially available products.
实施例1和厚朴酚对LL-37致玫瑰痤疮的治疗效果Therapeutic effect of Example 1 and honokiol on LL-37-induced rosacea
1、实验材料1. Experimental materials
1)实验药:和厚朴酚用丁二醇溶解为浓度0.5%和1.0%的溶液(mg/mL);壬二酸用丁二醇溶解为浓度15.0%的溶液(mg/mL)。1) Experimental drug: Honokiol was dissolved in butanediol into solutions (mg/mL) with a concentration of 0.5% and 1.0%; azelaic acid was dissolved in butanediol into a solution with a concentration of 15.0% (mg/mL).
2)实验动物:SPF级BALB/c雌性小鼠(6周龄)和小鼠专用饲料均购自成都达硕实验动物有限公司,实验室喂养1周,适应环境。2) Experimental animals: SPF grade BALB/c female mice (6 weeks old) and mouse-specific feed were purchased from Chengdu Dashuo Laboratory Animal Co., Ltd., and were fed in the laboratory for 1 week and adapted to the environment.
3)主要试验试剂:苏木素染液和伊红染液购买自珠海Baso公司;和厚朴酚购买于上海萨恩化学技术有限公司;LL-37由美国Invivogen(San Diego,CA)公司合成,CAS号为597562-32-8;基质丁二醇购买于上海德茂化工有限公司。3) Main test reagents: Hematoxylin and eosin staining solutions were purchased from Zhuhai Baso Company; Honokiol was purchased from Shanghai San Chemical Technology Co., Ltd.; LL-37 was synthesized by Invivogen (San Diego, CA), USA, CAS No. 597562-32-8; base butanediol was purchased from Shanghai Demao Chemical Co., Ltd.
2、实验方法2. Experimental method
1)将BALB/c小鼠分为0.5%和厚朴酚组、1.0%和厚朴酚组、15.0%壬二酸组、基质组(即丁二醇组)、LL-37组(即玫瑰痤疮动物模型组)和空白组(每组n=9),对小鼠进行预处理:分别将实验药涂抹于相对应组的小鼠背部皮肤(约0.1ml/cm2),2次/天,共给药6天;LL-37组和空白组不予以涂抹任何制剂。1) BALB/c mice were divided into 0.5% and honokiol group, 1.0% and honokiol group, 15.0% azelaic acid group, matrix group (ie butanediol group), LL-37 group (ie rosette). Acne animal model group) and blank group (n=9 in each group), mice were pretreated: experimental drugs were applied to the back skin of mice in the corresponding groups (about 0.1 ml/cm 2 ), 2 times/day , a total of 6 days of administration; LL-37 group and blank group did not apply any preparations.
2)在第4天进行造模:在0.5%和厚朴酚组、1.0%和厚朴酚组、15.0%壬二酸组、基质组和LL-37组皮内注射LL-37(320μM),空白组皮内注射超纯水,每12小时注射50μl,共4次。2) Modeling on the 4th day: intradermal injection of LL-37 (320 μM) in 0.5% and honokiol group, 1.0% and honokiol group, 15.0% azelaic acid group, matrix group and LL-37 group , the blank group was injected intradermally with ultrapure water, 50 μl every 12 hours, a total of 4 times.
3)在最后一次注射LL-37/超纯水的12小时后(即涂抹药物第6天)进行红斑评分,随后处死小鼠,取背部注射部位皮肤行组织活检:a.通过HE染色观察小鼠皮肤组织病理学改变并计数炎症细胞;b.在实验过程中观察小鼠一般情况,注意是否有不良反应,并取和厚朴酚组和空白组小鼠的各器官组织制成病理切片及HE染色,观察各器官的细胞形态。3) The erythema was scored 12 hours after the last injection of LL-37/ultrapure water (ie, the sixth day after the drug was applied), and then the mice were sacrificed, and the skin of the back injection site was taken for tissue biopsy: a. Histopathological changes of mouse skin and counted inflammatory cells; b. During the experiment, observe the general condition of the mice, pay attention to whether there are adverse reactions, and take the various organs and tissues of the mice in the honokiol group and the blank group to make pathological sections and HE staining was used to observe the cell morphology of each organ.
3、实验结果3. Experimental results
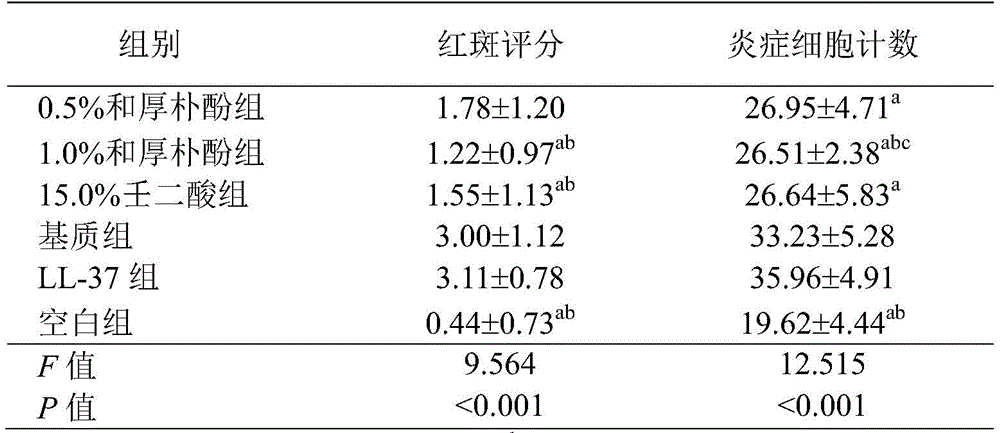
1)红斑评分结果1) Erythema score results
空白组小鼠皮肤无红斑和水肿形成;0.5%和厚朴酚组可见边界模糊的淡红斑,水肿轻微;1.0%和厚朴酚组可见红斑,无明显水肿;15.0%壬二酸组可见边界模糊的淡红斑,无明显水肿;基质组可见边界清楚、颜色鲜艳的红斑,伴有水肿;LL-37组可见边界清楚、颜色较深的红斑,伴有明显水肿(图1)。从表1的红斑评分结果可知,1.0%和厚朴酚组、15.0%壬二酸组红斑评分分别与基质组、LL-37组相比,均具有统计学差异(均P<0.05),1.0%和厚朴酚组和15.0%壬二酸组无统计学差异(均P>0.05)。说明和厚朴酚能够显著降低LL-37诱导的玫瑰痤疮动物模型的红斑评分,对玫瑰痤疮具有显著的治疗效果,且治疗效果随和厚朴酚浓度的增加而增加。There was no erythema and edema in the skin of the mice in the blank group; the 0.5% and honokiol groups had pale erythema with blurred borders and slight edema; the 1.0% and honokiol groups had erythema and no obvious edema; the 15.0% azelaic acid group had borders. Blurred pale erythema without obvious edema; erythema with clear border and bright color with edema in the stroma group; erythema with clear border and darker color with obvious edema in the LL-37 group (Figure 1). From the erythema score results in Table 1, it can be seen that the erythema scores of the 1.0% and honokiol groups and the 15.0% azelaic acid group were significantly different from those of the matrix group and the LL-37 group, respectively (all P<0.05), 1.0 % and honokiol group and 15.0% azelaic acid group had no statistical difference (all P>0.05). It indicated that honokiol could significantly reduce the erythema score of LL-37-induced rosacea animal model, and had a significant therapeutic effect on rosacea, and the therapeutic effect increased with the increase of honokiol concentration.
表1小鼠皮肤注射部位红斑评分和炎症细胞计数比较 Table 1 Comparison of erythema scores and inflammatory cell counts at skin injection sites in mice
注:n=9。a:与LL-37组对比,p<0.05;b:与基质组对比,p<0.05;c:与15.0%壬二酸组对比,p<0.05。Note: n=9. a : compared with the LL-37 group, p<0.05; b : compared with the matrix group, p<0.05; c : compared with the 15.0% azelaic acid group, p<0.05.
2)HE染色显示及炎症细胞计数结果2) HE staining and inflammatory cell count results
HE染色显示:空白组表皮结构完整,真皮层内炎症细胞浸润不明显。0.5%和厚朴酚组、1.0%和厚朴酚组和15.0%壬二酸组表皮结构完整,真皮内可见少量炎症细胞浸润;基质组和LL-37组局部表皮缺如,真皮内可见血管扩张和较多炎症细胞浸润,以淋巴细胞和中性粒细胞为主(图2)。在×400倍光镜下分别计数炎症细胞(结果见表1):0.5%和厚朴酚组和15.0%壬二酸组的炎症细胞计数均显著少于LL-37组(均P<0.05),不过与基质组相比均无统计学差异(均P>0.05)。1.0%和厚朴酚组的炎症细胞计数均显著少于基质组和LL-37组(p<0.05);并且,1.0%和厚朴酚组的炎症细胞计数显著少于15.0%壬二酸组,有统计学差异(p<0.05)。可以看出,0.5%和厚朴酚组的炎症细胞数显著少于LL-37组,具有统计学差异(P<0.05);1.0%和厚朴酚组的炎症细胞数显著少于基质组、LL-37组、15.0%壬二酸组,均具有统计学差异(P<0.05),所以和厚朴酚能够显著降低LL-37诱导的玫瑰痤疮动物模型的炎症细胞数,对玫瑰痤疮具有显著的治疗效果,且治疗效果随和厚朴酚浓度的增加而增加。HE staining showed that the epidermal structure of the blank group was intact, and the infiltration of inflammatory cells in the dermis was not obvious. In the 0.5% and honokiol group, 1.0% and honokiol group and 15.0% azelaic acid group, the epidermal structure was intact, and a small amount of inflammatory cell infiltration was seen in the dermis; the local epidermis was absent in the matrix group and LL-37 group, and blood vessels were seen in the dermis Expansion and more inflammatory cell infiltration, dominated by lymphocytes and neutrophils (Figure 2). Inflammatory cells were counted under a light microscope at ×400 magnification (the results are shown in Table 1): the counts of inflammatory cells in the 0.5% and honokiol groups and the 15.0% azelaic acid group were significantly lower than those in the LL-37 group (all P<0.05) , but there was no statistical difference compared with the matrix group (all P>0.05). The inflammatory cell counts in the 1.0% and honokiol groups were significantly lower than those in the stroma group and LL-37 group (p<0.05); and the inflammatory cell counts in the 1.0% and honokiol groups were significantly lower than those in the 15.0% azelaic acid group , there was a statistical difference (p<0.05). It can be seen that the number of inflammatory cells in the 0.5% and honokiol group was significantly less than that in the LL-37 group, with a statistical difference (P<0.05); the number of inflammatory cells in the 1.0% and honokiol group was significantly less than that in the matrix group, LL-37 group and 15.0% azelaic acid group had statistical difference (P<0.05), so Honokiol can significantly reduce the number of inflammatory cells in LL-37-induced rosacea animal model, and has a significant effect on rosacea. and the therapeutic effect increased with the increase of honokiol concentration.
3)不良反应3) Adverse reactions
各组小鼠一般情况良好,未见脱屑、体重减轻等不良反应;HE染色中和厚朴酚组的各器官组织细胞的形态未发现异常。The mice in each group were generally in good condition, and no adverse reactions such as desquamation and weight loss were found; no abnormality was found in the morphology of the cells of each organ and tissue in the neutralized honokiol group by HE staining.
上述结果表明,和厚朴酚能够显著减轻玫瑰痤疮小鼠的红斑情况,降低炎症细胞浸润,减少炎症细胞数目,有效治疗玫瑰痤疮。同时,玫瑰痤疮小鼠施用和厚朴酚后不会产生脱屑、体重减轻等不良反应,各器官组织细胞的形态也未发现异常。The above results show that Honokiol can significantly reduce the erythema of rosacea mice, reduce the infiltration of inflammatory cells, reduce the number of inflammatory cells, and effectively treat rosacea. At the same time, after administration of honokiol to rosacea mice, there were no adverse reactions such as desquamation and weight loss, and no abnormality was found in the morphology of cells in various organs.
综上,本发明提供了和厚朴酚、厚朴酚或其混合物在制备治疗和/或预防玫瑰痤疮的药物中的用途。实验证明,和厚朴酚能够显著减轻玫瑰痤疮小鼠的红斑情况,降低炎症细胞浸润,减少炎症细胞数目,有效治疗玫瑰痤疮。同时,玫瑰痤疮小鼠施用和厚朴酚后不会产生脱屑、体重减轻等不良反应,各器官组织细胞的形态也未发现异常。所以,和厚朴酚在制备治疗和/或预防玫瑰痤疮的药物中具有非常好的应用前景。To sum up, the present invention provides the use of honokiol, honokiol or a mixture thereof in the preparation of a medicament for treating and/or preventing rosacea. Experiments have shown that Honokiol can significantly reduce the erythema of rosacea mice, reduce the infiltration of inflammatory cells, reduce the number of inflammatory cells, and effectively treat rosacea. At the same time, after administration of honokiol to rosacea mice, there were no adverse reactions such as desquamation and weight loss, and no abnormality was found in the morphology of cells in various organs. Therefore, honokiol has a very good application prospect in the preparation of medicines for treating and/or preventing rosacea.
Claims (6)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2019112862384 | 2019-12-13 | ||
| CN201911286238.4A CN110812345A (en) | 2019-12-13 | 2019-12-13 | Application of honokiol in preparation of medicine for treating and/or preventing rosacea |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN111714475A true CN111714475A (en) | 2020-09-29 |
Family
ID=69545495
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201911286238.4A Pending CN110812345A (en) | 2019-12-13 | 2019-12-13 | Application of honokiol in preparation of medicine for treating and/or preventing rosacea |
| CN202010832764.2A Pending CN111714475A (en) | 2019-12-13 | 2020-08-18 | Use of and honokiol in the preparation of medicaments for treating and/or preventing rosacea |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201911286238.4A Pending CN110812345A (en) | 2019-12-13 | 2019-12-13 | Application of honokiol in preparation of medicine for treating and/or preventing rosacea |
Country Status (1)
| Country | Link |
|---|---|
| CN (2) | CN110812345A (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112168912A (en) * | 2020-09-30 | 2021-01-05 | 云南贝泰妮生物科技集团股份有限公司 | Plant compound capable of inhibiting acne-related pathogenic bacteria and application thereof |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002076393A2 (en) * | 2001-03-23 | 2002-10-03 | Emory University | Antiagionecic, antitumor, chemopreventative agents |
-
2019
- 2019-12-13 CN CN201911286238.4A patent/CN110812345A/en active Pending
-
2020
- 2020-08-18 CN CN202010832764.2A patent/CN111714475A/en active Pending
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002076393A2 (en) * | 2001-03-23 | 2002-10-03 | Emory University | Antiagionecic, antitumor, chemopreventative agents |
Non-Patent Citations (1)
| Title |
|---|
| 陈茵等主编: "《中药药理学》", 30 June 2012, 人民卫生出版社 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN110812345A (en) | 2020-02-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN108553411B (en) | Azelaic acid gel and preparation method and application thereof | |
| Zhan et al. | Ethosomes: a promising drug delivery platform for transdermal application | |
| Al-Mobeeriek | Effects of myrrh on intra-oral mucosal wounds compared with tetracycline-and chlorhexidine-based mouthwashes | |
| BR112015011673B1 (en) | composition and use of it | |
| CN113274342B (en) | Colchicine external composition | |
| CN112587593B (en) | Composition for treating acne and preparation method thereof | |
| Ye et al. | ACNO hydrogel enhances diabetic wound healing by modulating the Bcl-2/Bax/Caspase-3/PARP pathway | |
| CN111714475A (en) | Use of and honokiol in the preparation of medicaments for treating and/or preventing rosacea | |
| JP2025532409A (en) | Use of lithospermic acid in the manufacture of a drug for treating diabetic ulcers | |
| CN107519236A (en) | A kind of topical agent for treating onychomycosis | |
| WO2016090892A1 (en) | Water-soluble gel for treating diabetic foot | |
| AU2016390488A1 (en) | Application of dimethylamino micheliolide | |
| CN119015275A (en) | Application of daphnetin in preparing medicine for treating renal clear cell carcinoma | |
| CN115068407A (en) | Baricitinib gel and preparation method and application thereof | |
| CN102872158A (en) | External drug combination for curing eczema and preparation method thereof | |
| Yang et al. | Dissolving cinnamaldehyde HA/PVA microneedles for the treatment of skin diseases caused by Microsporum canis | |
| CN105796690A (en) | Rose traditional Chinese medicine composition for treating psoriasis and preparation method and application | |
| CN119818464B (en) | Application of rasagiline mesylate in preparation of medicines for treating diabetic ulcers | |
| BERNABE et al. | Efficacy of snail mucin in wound healing: a review | |
| CN119857089B (en) | Application of hypericin in the preparation of medicine for pathological scars | |
| CN115364097B (en) | Application of a pyridone derivative containing a heteroatom cyclobutane substituent | |
| CN105878427A (en) | Peony seed oil compound spray for curing burn and scald and preparation method | |
| RU2814358C1 (en) | Method of treating papulopustular subtype of rosacea by points | |
| CN101953836A (en) | Application of fluoropirfenidone in preparing medicaments for treating psoriasis | |
| RU2710541C1 (en) | Method of treating angular cheilitis caused by deficiency of riboflavin |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20200929 |
|
| RJ01 | Rejection of invention patent application after publication |