CN111686054B - Plant composition with quick and sustained effects and application thereof - Google Patents
Plant composition with quick and sustained effects and application thereof Download PDFInfo
- Publication number
- CN111686054B CN111686054B CN202010771848.XA CN202010771848A CN111686054B CN 111686054 B CN111686054 B CN 111686054B CN 202010771848 A CN202010771848 A CN 202010771848A CN 111686054 B CN111686054 B CN 111686054B
- Authority
- CN
- China
- Prior art keywords
- extract
- italian
- composition
- ethanol
- personal care
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 37
- 230000002459 sustained effect Effects 0.000 title description 5
- 229940114496 olive leaf extract Drugs 0.000 claims abstract description 23
- 229940119217 chamomile extract Drugs 0.000 claims abstract description 8
- 235000020221 chamomile extract Nutrition 0.000 claims abstract description 8
- 239000000284 extract Substances 0.000 claims description 54
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 37
- 240000007817 Olea europaea Species 0.000 claims description 22
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 22
- 239000000243 solution Substances 0.000 claims description 21
- 239000002537 cosmetic Substances 0.000 claims description 16
- 238000000034 method Methods 0.000 claims description 12
- 238000002360 preparation method Methods 0.000 claims description 12
- 235000002725 Olea europaea Nutrition 0.000 claims description 11
- 238000010438 heat treatment Methods 0.000 claims description 11
- 239000000843 powder Substances 0.000 claims description 10
- 235000007519 Chimonanthus praecox Nutrition 0.000 claims description 7
- 244000308760 Helichrysum petiolatum Species 0.000 claims description 7
- 240000001359 Origanum dictamnus Species 0.000 claims description 7
- 235000015987 Origanum dictamnus Nutrition 0.000 claims description 7
- 235000004383 Origanum vulgare subsp. vulgare Nutrition 0.000 claims description 7
- 239000006071 cream Substances 0.000 claims description 7
- 238000002156 mixing Methods 0.000 claims description 7
- 238000001035 drying Methods 0.000 claims description 6
- IDGUHHHQCWSQLU-UHFFFAOYSA-N ethanol;hydrate Chemical compound O.CCO IDGUHHHQCWSQLU-UHFFFAOYSA-N 0.000 claims description 6
- 239000006210 lotion Substances 0.000 claims description 5
- 230000008569 process Effects 0.000 claims description 5
- 239000002904 solvent Substances 0.000 claims description 5
- 239000002671 adjuvant Substances 0.000 claims description 4
- 238000004140 cleaning Methods 0.000 claims description 3
- 238000001914 filtration Methods 0.000 claims description 3
- -1 serum Substances 0.000 claims description 3
- 210000002966 serum Anatomy 0.000 claims description 3
- 239000008187 granular material Substances 0.000 claims description 2
- 238000000227 grinding Methods 0.000 claims description 2
- 239000011259 mixed solution Substances 0.000 claims description 2
- 230000001376 precipitating effect Effects 0.000 claims description 2
- 238000007873 sieving Methods 0.000 claims description 2
- 238000000605 extraction Methods 0.000 claims 2
- 230000000694 effects Effects 0.000 abstract description 40
- 241000196324 Embryophyta Species 0.000 abstract description 4
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 38
- 210000003491 skin Anatomy 0.000 description 20
- 235000011187 glycerol Nutrition 0.000 description 14
- 238000012360 testing method Methods 0.000 description 14
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 11
- 230000004044 response Effects 0.000 description 11
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 11
- 240000001432 Calendula officinalis Species 0.000 description 10
- 239000012071 phase Substances 0.000 description 10
- 239000000047 product Substances 0.000 description 10
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 9
- 239000000126 substance Substances 0.000 description 9
- 230000000638 stimulation Effects 0.000 description 8
- 241001073507 Callicarpa Species 0.000 description 7
- 239000007864 aqueous solution Substances 0.000 description 7
- 229940075529 glyceryl stearate Drugs 0.000 description 7
- DCAYPVUWAIABOU-UHFFFAOYSA-N hexadecane Chemical class CCCCCCCCCCCCCCCC DCAYPVUWAIABOU-UHFFFAOYSA-N 0.000 description 7
- 239000003921 oil Substances 0.000 description 7
- 239000002994 raw material Substances 0.000 description 7
- AJBZENLMTKDAEK-UHFFFAOYSA-N 3a,5a,5b,8,8,11a-hexamethyl-1-prop-1-en-2-yl-1,2,3,4,5,6,7,7a,9,10,11,11b,12,13,13a,13b-hexadecahydrocyclopenta[a]chrysene-4,9-diol Chemical compound CC12CCC(O)C(C)(C)C1CCC(C1(C)CC3O)(C)C2CCC1C1C3(C)CCC1C(=C)C AJBZENLMTKDAEK-UHFFFAOYSA-N 0.000 description 6
- 235000003880 Calendula Nutrition 0.000 description 6
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- 239000008367 deionised water Substances 0.000 description 6
- 229910021641 deionized water Inorganic materials 0.000 description 6
- 230000007794 irritation Effects 0.000 description 6
- 229940082862 olea europaea leaf extract Drugs 0.000 description 6
- OULAJFUGPPVRBK-UHFFFAOYSA-N tetratriacontyl alcohol Natural products CCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCO OULAJFUGPPVRBK-UHFFFAOYSA-N 0.000 description 6
- KWIUHFFTVRNATP-UHFFFAOYSA-N Betaine Natural products C[N+](C)(C)CC([O-])=O KWIUHFFTVRNATP-UHFFFAOYSA-N 0.000 description 5
- KWIUHFFTVRNATP-UHFFFAOYSA-O N,N,N-trimethylglycinium Chemical compound C[N+](C)(C)CC(O)=O KWIUHFFTVRNATP-UHFFFAOYSA-O 0.000 description 5
- 229960003237 betaine Drugs 0.000 description 5
- 238000001816 cooling Methods 0.000 description 5
- 239000000463 material Substances 0.000 description 5
- 210000003205 muscle Anatomy 0.000 description 5
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 4
- AMUTYVGRCVFCCD-UHFFFAOYSA-N 5,6-diaminopyridine-3-carboxylic acid Chemical compound NC1=CC(C(O)=O)=CN=C1N AMUTYVGRCVFCCD-UHFFFAOYSA-N 0.000 description 4
- NIXOWILDQLNWCW-UHFFFAOYSA-N Acrylic acid Chemical compound OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 4
- 235000005881 Calendula officinalis Nutrition 0.000 description 4
- 241000628997 Flos Species 0.000 description 4
- 206010040880 Skin irritation Diseases 0.000 description 4
- 229920002385 Sodium hyaluronate Polymers 0.000 description 4
- 229920002125 Sokalan® Polymers 0.000 description 4
- 230000003110 anti-inflammatory effect Effects 0.000 description 4
- 229960001631 carbomer Drugs 0.000 description 4
- 229940082500 cetostearyl alcohol Drugs 0.000 description 4
- GLDOVTGHNKAZLK-UHFFFAOYSA-N octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 description 4
- 229940100460 peg-100 stearate Drugs 0.000 description 4
- 239000000419 plant extract Substances 0.000 description 4
- 239000003755 preservative agent Substances 0.000 description 4
- 230000002335 preservative effect Effects 0.000 description 4
- 230000037307 sensitive skin Effects 0.000 description 4
- 230000036556 skin irritation Effects 0.000 description 4
- 231100000475 skin irritation Toxicity 0.000 description 4
- 229940010747 sodium hyaluronate Drugs 0.000 description 4
- YWIVKILSMZOHHF-QJZPQSOGSA-N sodium;(2s,3s,4s,5r,6r)-6-[(2s,3r,4r,5s,6r)-3-acetamido-2-[(2s,3s,4r,5r,6r)-6-[(2r,3r,4r,5s,6r)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2- Chemical compound [Na+].CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 YWIVKILSMZOHHF-QJZPQSOGSA-N 0.000 description 4
- 235000018625 Helichrysum angustifolium Nutrition 0.000 description 3
- 244000292571 Helichrysum italicum Species 0.000 description 3
- 235000013530 Helichrysum italicum Nutrition 0.000 description 3
- 208000002193 Pain Diseases 0.000 description 3
- 208000003251 Pruritus Diseases 0.000 description 3
- 206010040830 Skin discomfort Diseases 0.000 description 3
- 239000000969 carrier Substances 0.000 description 3
- 239000004205 dimethyl polysiloxane Substances 0.000 description 3
- 235000013870 dimethyl polysiloxane Nutrition 0.000 description 3
- 239000002085 irritant Substances 0.000 description 3
- 231100000021 irritant Toxicity 0.000 description 3
- 229920000435 poly(dimethylsiloxane) Polymers 0.000 description 3
- 230000002040 relaxant effect Effects 0.000 description 3
- 230000035945 sensitivity Effects 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 239000000230 xanthan gum Substances 0.000 description 3
- 229920001285 xanthan gum Polymers 0.000 description 3
- 235000010493 xanthan gum Nutrition 0.000 description 3
- 229940082509 xanthan gum Drugs 0.000 description 3
- CCOQPGVQAWPUPE-UHFFFAOYSA-N 4-tert-butylcyclohexan-1-ol Chemical compound CC(C)(C)C1CCC(O)CC1 CCOQPGVQAWPUPE-UHFFFAOYSA-N 0.000 description 2
- 241000006375 Calceolaria Species 0.000 description 2
- 235000007866 Chamaemelum nobile Nutrition 0.000 description 2
- 235000007516 Chrysanthemum Nutrition 0.000 description 2
- 244000189548 Chrysanthemum x morifolium Species 0.000 description 2
- ZGTMUACCHSMWAC-UHFFFAOYSA-L EDTA disodium salt (anhydrous) Chemical compound [Na+].[Na+].OC(=O)CN(CC([O-])=O)CCN(CC(O)=O)CC([O-])=O ZGTMUACCHSMWAC-UHFFFAOYSA-L 0.000 description 2
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 2
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 2
- 244000042664 Matricaria chamomilla Species 0.000 description 2
- 235000007232 Matricaria chamomilla Nutrition 0.000 description 2
- 241000207834 Oleaceae Species 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 239000004480 active ingredient Substances 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- 230000003078 antioxidant effect Effects 0.000 description 2
- 235000006708 antioxidants Nutrition 0.000 description 2
- 239000008346 aqueous phase Substances 0.000 description 2
- 230000004888 barrier function Effects 0.000 description 2
- CDQSJQSWAWPGKG-UHFFFAOYSA-N butane-1,1-diol Chemical compound CCCC(O)O CDQSJQSWAWPGKG-UHFFFAOYSA-N 0.000 description 2
- 238000007596 consolidation process Methods 0.000 description 2
- 229940008099 dimethicone Drugs 0.000 description 2
- 230000001804 emulsifying effect Effects 0.000 description 2
- 230000014509 gene expression Effects 0.000 description 2
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 2
- 230000028709 inflammatory response Effects 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- 230000007803 itching Effects 0.000 description 2
- 230000007774 longterm Effects 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 230000007433 nerve pathway Effects 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 230000035807 sensation Effects 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- PRAKJMSDJKAYCZ-UHFFFAOYSA-N squalane Chemical compound CC(C)CCCC(C)CCCC(C)CCCCC(C)CCCC(C)CCCC(C)C PRAKJMSDJKAYCZ-UHFFFAOYSA-N 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 230000002195 synergetic effect Effects 0.000 description 2
- HDTRYLNUVZCQOY-UHFFFAOYSA-N α-D-glucopyranosyl-α-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(O)C(O)C(CO)O1 HDTRYLNUVZCQOY-UHFFFAOYSA-N 0.000 description 1
- LQIAZOCLNBBZQK-UHFFFAOYSA-N 1-(1,2-Diphosphanylethyl)pyrrolidin-2-one Chemical compound PCC(P)N1CCCC1=O LQIAZOCLNBBZQK-UHFFFAOYSA-N 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- 101800005049 Beta-endorphin Proteins 0.000 description 1
- 241000219357 Cactaceae Species 0.000 description 1
- 201000004624 Dermatitis Diseases 0.000 description 1
- 239000003109 Disodium ethylene diamine tetraacetate Substances 0.000 description 1
- 108010049140 Endorphins Proteins 0.000 description 1
- 102000009025 Endorphins Human genes 0.000 description 1
- 229930194248 Licoflavone Natural products 0.000 description 1
- MEHHCBRCXIDGKZ-UHFFFAOYSA-N Licoflavone C Natural products CC(C)=CCC1=C(O)C=C(O)C(C(C=2)=O)=C1OC=2C1=CC=C(O)C=C1 MEHHCBRCXIDGKZ-UHFFFAOYSA-N 0.000 description 1
- VTAJIXDZFCRWBR-UHFFFAOYSA-N Licoricesaponin B2 Natural products C1C(C2C(C3(CCC4(C)CCC(C)(CC4C3=CC2)C(O)=O)C)(C)CC2)(C)C2C(C)(C)CC1OC1OC(C(O)=O)C(O)C(O)C1OC1OC(C(O)=O)C(O)C(O)C1O VTAJIXDZFCRWBR-UHFFFAOYSA-N 0.000 description 1
- 208000007920 Neurogenic Inflammation Diseases 0.000 description 1
- 241000207836 Olea <angiosperm> Species 0.000 description 1
- 241000795633 Olea <sea slug> Species 0.000 description 1
- OQILCOQZDHPEAZ-UHFFFAOYSA-N Palmitinsaeure-octylester Natural products CCCCCCCCCCCCCCCC(=O)OCCCCCCCC OQILCOQZDHPEAZ-UHFFFAOYSA-N 0.000 description 1
- AVFIYMSJDDGDBQ-UHFFFAOYSA-N Parthenium Chemical compound C1C=C(CCC(C)=O)C(C)CC2OC(=O)C(=C)C21 AVFIYMSJDDGDBQ-UHFFFAOYSA-N 0.000 description 1
- 241001495454 Parthenium Species 0.000 description 1
- 239000004264 Petrolatum Substances 0.000 description 1
- 244000234609 Portulaca oleracea Species 0.000 description 1
- 235000001855 Portulaca oleracea Nutrition 0.000 description 1
- 102100027467 Pro-opiomelanocortin Human genes 0.000 description 1
- 102000003566 TRPV1 Human genes 0.000 description 1
- HDTRYLNUVZCQOY-WSWWMNSNSA-N Trehalose Natural products O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-WSWWMNSNSA-N 0.000 description 1
- 101150016206 Trpv1 gene Proteins 0.000 description 1
- 235000018936 Vitellaria paradoxa Nutrition 0.000 description 1
- 241001135917 Vitellaria paradoxa Species 0.000 description 1
- 229940014843 acetyl dipeptide-1 cetyl ester Drugs 0.000 description 1
- 108010074988 acetyltyrosyl-arginine cetyl ester Proteins 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 229930013930 alkaloid Natural products 0.000 description 1
- HDTRYLNUVZCQOY-LIZSDCNHSA-N alpha,alpha-trehalose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-LIZSDCNHSA-N 0.000 description 1
- 230000008485 antagonism Effects 0.000 description 1
- 230000002421 anti-septic effect Effects 0.000 description 1
- 235000008714 apigenin Nutrition 0.000 description 1
- KZNIFHPLKGYRTM-UHFFFAOYSA-N apigenin Chemical compound C1=CC(O)=CC=C1C1=CC(=O)C2=C(O)C=C(O)C=C2O1 KZNIFHPLKGYRTM-UHFFFAOYSA-N 0.000 description 1
- 229940117893 apigenin Drugs 0.000 description 1
- XADJWCRESPGUTB-UHFFFAOYSA-N apigenin Natural products C1=CC(O)=CC=C1C1=CC(=O)C2=CC(O)=C(O)C=C2O1 XADJWCRESPGUTB-UHFFFAOYSA-N 0.000 description 1
- KIUVQMGRTDPAFR-UHFFFAOYSA-N benzoic acid 2-hydroxy-2-phenylpropanamide Chemical group C(C1=CC=CC=C1)(=O)O.OC(C(=O)N)(C)C1=CC=CC=C1 KIUVQMGRTDPAFR-UHFFFAOYSA-N 0.000 description 1
- WOPZMFQRCBYPJU-NTXHZHDSSA-N beta-endorphin Chemical compound C([C@@H](C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCC(N)=O)C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H]1N(CCC1)C(=O)[C@@H](NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CCSC)NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)CC=1C=CC(O)=CC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)[C@@H](C)O)C1=CC=CC=C1 WOPZMFQRCBYPJU-NTXHZHDSSA-N 0.000 description 1
- 235000019437 butane-1,3-diol Nutrition 0.000 description 1
- 229940002386 calendula officinalis extract Drugs 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 238000002845 discoloration Methods 0.000 description 1
- 235000019301 disodium ethylene diamine tetraacetate Nutrition 0.000 description 1
- 239000003974 emollient agent Substances 0.000 description 1
- 238000004945 emulsification Methods 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 239000000469 ethanolic extract Substances 0.000 description 1
- GJQLBGWSDGMZKM-UHFFFAOYSA-N ethylhexyl palmitate Chemical compound CCCCCCCCCCCCCCCC(=O)OC(CC)CCCCC GJQLBGWSDGMZKM-UHFFFAOYSA-N 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 230000001815 facial effect Effects 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 229960005150 glycerol Drugs 0.000 description 1
- 229940075507 glyceryl monostearate Drugs 0.000 description 1
- LPLVUJXQOOQHMX-UHFFFAOYSA-N glycyrrhetinic acid glycoside Natural products C1CC(C2C(C3(CCC4(C)CCC(C)(CC4C3=CC2=O)C(O)=O)C)(C)CC2)(C)C2C(C)(C)C1OC1OC(C(O)=O)C(O)C(O)C1OC1OC(C(O)=O)C(O)C(O)C1O LPLVUJXQOOQHMX-UHFFFAOYSA-N 0.000 description 1
- 229960004949 glycyrrhizic acid Drugs 0.000 description 1
- UYRUBYNTXSDKQT-UHFFFAOYSA-N glycyrrhizic acid Natural products CC1(C)C(CCC2(C)C1CCC3(C)C2C(=O)C=C4C5CC(C)(CCC5(C)CCC34C)C(=O)O)OC6OC(C(O)C(O)C6OC7OC(O)C(O)C(O)C7C(=O)O)C(=O)O UYRUBYNTXSDKQT-UHFFFAOYSA-N 0.000 description 1
- 239000001685 glycyrrhizic acid Substances 0.000 description 1
- 235000019410 glycyrrhizin Nutrition 0.000 description 1
- LPLVUJXQOOQHMX-QWBHMCJMSA-N glycyrrhizinic acid Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@H](O[C@@H]1O[C@@H]1C([C@H]2[C@]([C@@H]3[C@@]([C@@]4(CC[C@@]5(C)CC[C@@](C)(C[C@H]5C4=CC3=O)C(O)=O)C)(C)CC2)(C)CC1)(C)C)C(O)=O)[C@@H]1O[C@H](C(O)=O)[C@@H](O)[C@H](O)[C@H]1O LPLVUJXQOOQHMX-QWBHMCJMSA-N 0.000 description 1
- JFHZXDZUXGBFAQ-KYJUHHDHSA-N hexadecyl (2s)-2-[[(2s)-2-acetamido-3-(4-hydroxyphenyl)propanoyl]amino]-5-(diaminomethylideneamino)pentanoate Chemical compound CCCCCCCCCCCCCCCCOC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@@H](NC(C)=O)CC1=CC=C(O)C=C1 JFHZXDZUXGBFAQ-KYJUHHDHSA-N 0.000 description 1
- 239000003906 humectant Substances 0.000 description 1
- 229940071826 hydroxyethyl cellulose Drugs 0.000 description 1
- 230000002757 inflammatory effect Effects 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 235000015110 jellies Nutrition 0.000 description 1
- 239000008274 jelly Substances 0.000 description 1
- 210000002510 keratinocyte Anatomy 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 239000001788 mono and diglycerides of fatty acids Substances 0.000 description 1
- JXTPJDDICSTXJX-UHFFFAOYSA-N n-Triacontane Natural products CCCCCCCCCCCCCCCCCCCCCCCCCCCCCC JXTPJDDICSTXJX-UHFFFAOYSA-N 0.000 description 1
- 210000000653 nervous system Anatomy 0.000 description 1
- 229940012843 omega-3 fatty acid Drugs 0.000 description 1
- 235000020660 omega-3 fatty acid Nutrition 0.000 description 1
- 239000006014 omega-3 oil Substances 0.000 description 1
- 230000000149 penetrating effect Effects 0.000 description 1
- 229940066842 petrolatum Drugs 0.000 description 1
- 235000019271 petrolatum Nutrition 0.000 description 1
- 230000008092 positive effect Effects 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 229960004063 propylene glycol Drugs 0.000 description 1
- 238000010298 pulverizing process Methods 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 229940057910 shea butter Drugs 0.000 description 1
- 238000002791 soaking Methods 0.000 description 1
- 229940032094 squalane Drugs 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 230000000475 sunscreen effect Effects 0.000 description 1
- 239000000516 sunscreening agent Substances 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 210000001170 unmyelinated nerve fiber Anatomy 0.000 description 1
- 230000024883 vasodilation Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9783—Angiosperms [Magnoliophyta]
- A61K8/9789—Magnoliopsida [dicotyledons]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/005—Preparations for sensitive skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/74—Biological properties of particular ingredients
- A61K2800/75—Anti-irritant
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Botany (AREA)
- Birds (AREA)
- Epidemiology (AREA)
- Mycology (AREA)
- Microbiology (AREA)
- Biotechnology (AREA)
- Engineering & Computer Science (AREA)
- Dermatology (AREA)
- Cosmetics (AREA)
Abstract
The invention discloses a plant composition with quick, slow and sustainable effects and application thereof. The composition comprises an Italian chamomile extract and an olive leaf extract.
Description
Technical Field
The invention relates to the field of daily chemical industry, in particular to a composition suitable for sensitive skin, and a preparation method and application thereof.
Background
The international forum for pruritus (IFSI)2016 defined sensitive skin as an unpleasant sensation (burning, pain, itching, stinging) generally caused by several non-irritant ingredients, sometimes accompanied by redness of the skin. Modern cosmetics often use plant extracts or chemically synthesized active ingredients with soothing and anti-inflammatory effects to relieve the consumer's unpleasant feeling of skin sensitivity. The chemically synthesized molecule is hydroxyphenylpropionamide benzoic acid4-tert-butylcyclohexanol (1609) Acetyl dipeptide-1 cetyl ester (Calmosensine) TM ) Etc.; the natural plant such as glycyrrhizic acid and licoflavone contained in Glycyrrhrizae radix, and herba Centellae extract containing herba Centellae GlycosidesAsiatic acid and its natural derivatives, alkaloids and omega-3 fatty acids from purslane, and apigenin from chamomile.
In addition to using a single plant extract, it is also common to use compounded plant ingredients to achieve soothing effects, as disclosed in patent application CN200810239323.0, where an extract of cactus is compounded with glycerin and trehalose.
There have been many studies in this area, but they have been primarily directed to relieving skin discomfort and reducing redness by inhibiting skin inflammation or repairing the barrier. However, the irritation and redness caused by cosmetics on the skin of sensitive skin consumers occur in many cases within half an hour, and consumers desire to quickly resolve the discomfort and redness caused by skin sensitivity (internal consumer research reports). The unpleasant feelings of sensitive skin (burning, pain, itching, stabbing) are mediated by the unmyelinated C-fibers in the nervous system, and the current research thinking is lacking of fast soothing by regulating the nervous action and simultaneously sustainable consolidation effect to solve the trouble of sensitive muscle consumers.
Therefore, there is a pressing need in the art to find new ways to address both rapid relief of discomfort and redness in sensitive muscle consumers, and to consolidate and enhance the relief effect.
Disclosure of Invention
The invention aims to provide a composition which can quickly relieve skin and can keep the effect.
In a first aspect of the invention, a composition is provided comprising an extract of Italian Callicarpa and an extract of Olea europaea.
In another embodiment, the composition consists of an extract of italian camomile and an extract of olive leaves.
In another embodiment, the composition comprises 0.01-5 w/w% of Italian chamomile extract and 0.01-5 w/w% of olive leaf extract, based on the total mass of the composition.
In another embodiment, the composition further comprises 50 w/w% glycerin and the balance water.
In another embodiment, the composition comprises an Italian chamomilla extract and an olive leaf extract in a mass ratio of 1: 0.5-2.
In a second aspect of the present invention, there is provided a process for the preparation of a composition provided by the present invention as described above, said process comprising the steps of: the composition provided by the invention as described above is obtained by mixing the extract of Italian Calceolaria with the extract of Olea europaea leaves.
In a third aspect of the invention there is provided the use of a composition as provided herein, as described above, as a cosmetic or personal care product; or for the preparation of cosmetic or personal care products.
In another embodiment, the personal care product comprises a cream, lotion, serum, toner, and gel.
In a fourth aspect of the invention, there is provided a cosmetic or personal care product comprising a composition as provided by the invention as described above and a cosmetically acceptable carrier.
In a fifth aspect of the present invention there is provided a process for the preparation of a cosmetic or personal care product as hereinbefore described provided by the present invention, the process comprising the steps of: mixing an emulsified or non-emulsified cosmetically acceptable carrier with a composition as provided herein as described above to give a cosmetic or personal care product as provided herein as described above.
Therefore, the skin care product can quickly relieve skin irritation and redness of sensitive muscle consumers, and can consolidate and improve the relieving effect.
Drawings
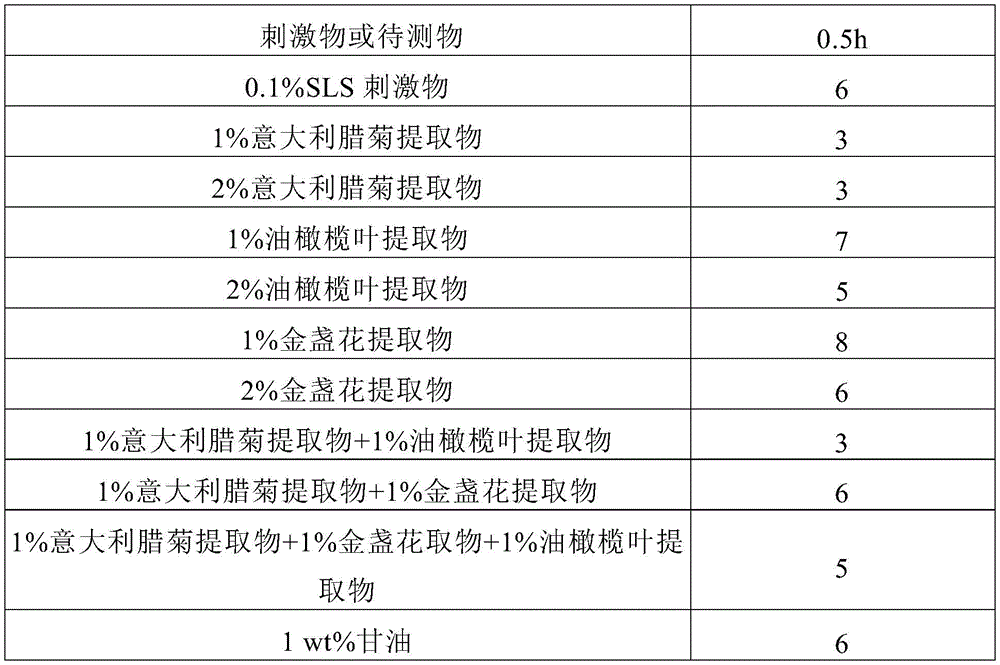
FIG. 1 shows the number of 0.5-hour stimulus response cases.
FIG. 2 shows the number of 48-hour stimulus response cases.
Figure 3 shows a 0.5 hour soothing effect.
Figure 4 shows the 48 hour soothing effect.
Detailed Description
The inventors have made extensive and intensive studies considering the rapid resolution of the sensations of discomfort and redness associated with skin sensitivity by modulating the nervous and inflammatory signals, alleviating the symptoms of sensitive muscles, reducing redness associated with vasodilation, for example by promoting the expression of beta endorphins in the skin to soothe the discomfort of sensitive muscles, and anti-inflammatory to consolidate the soothing effect. Based on such an idea, many substances have been tried and have been met with great obstacles, and it has been unexpectedly found that the use of an extract of Italian Calceolaria in combination with an extract of Olea europaea leaves has a synergistic effect in rapidly soothing the skin and sustaining the soothing effect. On the basis of this, the present invention has been completed.
As used herein, "Helichrysum italicum" is an evergreen subfbust or perennial herb. The extract used in the invention is an ethanol extract. In one embodiment of the invention, Helichrysum italicum powder or granules are used, mixed with a solution having an ethanol concentration of 50-95 v/v% based on the total volume of the aqueous ethanol solution, extracted at a temperature of 50 ℃ to reflux temperature, and the extract is filtered and concentrated to obtain Helichrysum italicum extract.
As used herein, "olives (oleaeuropaea l.)" are oil crops of the olea genus of the family Oleaceae (Oleaceae). The invention uses the alcohol extract of the effective part of the leaf. In one embodiment of the present invention, dry olive leaf powder or small particles are used, mixed with a solution having an ethanol concentration of 50-85 v/v% based on the total volume of an aqueous ethanol solution, extracted at 70-90 deg.C, and the extract is filtered and concentrated to obtain an olive leaf extract.
As used herein, the term "soothing" refers to reducing skin discomfort and increasing the skin's tolerance to external stimuli. Preferably, the effect is a reduction in skin redness or inflammatory response.
The term "effective amount" means: the amount is effective to reduce the redness or inflammatory response of the skin after a suitable period of use for the purpose of soothing the skin.
"composition" refers to a composition that, when applied to an individual (typically a human), is capable of penetrating the skin to induce the desired soothing effect.
As used herein, the term "cosmetically acceptable carrier" refers to a carrier that allows a cosmetic or personal care product to be applied, including various excipients and diluents, which are not themselves essential active ingredients, and which do not have undue toxicity after application. Suitable carriers are well known to those of ordinary skill in the art. A sufficient discussion of cosmetically acceptable excipients can be found in the cosmetic hygiene specifications 2015 edition. Such carriers may include humectants, emulsifiers, thickeners, chelating agents, emollients, and the like in the composition. Such as, but not limited to, butylene glycol, glycerin, betaine, sodium hyaluronate, propylene glycol, glyceryl stearate/PEG-100 stearate, glyceryl stearate, xanthan gum, hydroxyethyl cellulose, carbomer, disodium EDTA, the isomeric hexadecanes, isooctyl palmitate, cetostearyl alcohol, cetyl stearyl alcohol, dimethicone, and the like.
The term "administering" as used herein means directly applying the composition to form a substantial amount of the composition on the skin of the individual to whom it is applied.
The terms "individual" or "individual" are used herein to refer to a person who can receive the compositions and/or methods described for application to the skin.
As used herein, "room temperature" means 15-40 deg.C, preferably 20-30 deg.C.
The inventor forms a composition by using a Italian wintersweet extract and an olive leaf extract, wherein the dosage (mass) ratio of the Italian wintersweet extract to the olive leaf extract is 1: 0.5-2; such as, but not limited to, 1: 0.6-1.8, 1: 0.7-1.6, 1: 0.5-1.5, 1: 0.8-1.2, and the like.
In one embodiment of the present invention, the composition provided by the present invention can be obtained by mixing an extract of Italian Helichrysum and an extract of Olea europaea.
Further, the compositions provided herein can be combined with cosmetically acceptable carriers to provide a variety of cosmetic or personal care products that can be applied to human skin, including, but not limited to, barrier creams, sunscreen creams, facial creams, eye creams, lotions, serums, lotions, and the like.
In one embodiment of the present invention, a cosmetic or personal care product for application to human skin is obtained by adding an aqueous phase formed from some cosmetically acceptable carrier to an oil phase formed from some cosmetically acceptable carrier and adding the composition provided herein after emulsification.
In some embodiments of the invention, the cosmetically acceptable carrier for forming the aqueous phase includes, but is not limited to, one or more of glycerin, butylene glycol, carbomer, EDTA2 Na, sodium hyaluronate, betaine, water.
In some embodiments of the invention, the cosmetically acceptable carrier for forming the oil phase includes, but is not limited to, one or more of glyceryl stearate, glyceryl stearate/PEG-100 stearate complex, isomeric hexadecane, isooctyl palmitate, cetostearyl alcohol, dimethicone, petrolatum, shea butter, squalane, lecithin.
Although numerical ranges and parameters setting forth the broad scope of the invention are approximate, the values set forth in the specific examples are presented as precisely as possible. Any numerical value, however, inherently contains certain errors necessarily resulting from the individual testing measurements. As used herein, "about" generally means that the actual value is within plus or minus 10%, 5%, 1%, or 0.5% of a particular value or range. Alternatively, the term "about" means that the actual value falls within the acceptable standard error of the mean, as considered by those skilled in the art. Except in the experimental examples, or where otherwise expressly indicated, it is to be understood that all ranges, amounts, values and percentages herein used (e.g., to describe amounts of materials, length of time, temperature, operating conditions, quantitative ratios, and the like) are to be modified by the word "about". Accordingly, unless indicated to the contrary, the numerical parameters set forth in the specification and attached claims are approximations that may vary depending upon the desired properties sought to be obtained. At the very least, these numerical parameters are to be understood as meaning the number of significant digits and the number resulting from applying ordinary rounding techniques.
Unless otherwise defined herein, the scientific and technical terms used herein have the same meaning as commonly understood and used by one of ordinary skill in the art. Furthermore, as used herein, a singular noun covers a plural of that noun without conflicting context; the use of plural nouns also covers the singular form of such nouns.
To make the features and effects of the present invention comprehensible to those skilled in the art, general description and definitions are made below with reference to terms and expressions mentioned in the specification and claims. Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs.
The theory or mechanism described and disclosed herein, whether correct or incorrect, should not limit the scope of the present invention in any way, i.e., the present disclosure may be practiced without limitation to any particular theory or mechanism.
All features defined herein as numerical ranges or percentage ranges, such as values, amounts, levels and concentrations, are for brevity and convenience only. Accordingly, the description of numerical ranges or percentage ranges should be considered to cover and specifically disclose all possible subranges and individual numerical values (including integers and fractions) within the range.
The features mentioned above, or those mentioned in the embodiments, may be combined in any combination. All features disclosed in this specification may be combined in any combination, provided that there is no conflict between such features and the combination, and all possible combinations are to be considered within the scope of the present specification. Each feature disclosed in this specification may be replaced by an alternative feature serving the same, equivalent, or similar purpose. Thus, unless expressly stated otherwise, the features disclosed are merely generic examples of equivalent or similar features.
The main advantages of the invention are:
1. the Italian chamomile extract used in the composition provided by the invention can play a role through a nerve pathway, quickly takes effect, and can inhibit TRPV1 to reduce downstream neurogenic inflammation by promoting beta endorphin in the skin, so that the effect can be maintained.
2. The composition provided by the invention combines the Italian chamomile extract and the olive leaf extract, has better stimulation relieving effect than the addition of any one component used alone in a short time, and has multiplication effect; the olive leaf extract can also enhance the sustained relieving effect of the Italian chamomile extract for a long time, and is better than the single use.
The invention will be further illustrated with reference to the following specific examples. It should be understood that these examples are for illustrative purposes only and are not intended to limit the scope of the present invention. The experimental procedures, in which specific conditions are not noted in the following examples, are generally carried out according to conventional conditions or according to conditions recommended by the manufacturers. All percentages, ratios, proportions, or parts are by weight unless otherwise specified. The weight volume percentage units in the present invention are well known to those skilled in the art and refer to, for example, the weight of solute in a 100 ml solution. Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs. In addition, any methods and materials similar or equivalent to those described herein can be used in the methods of the present invention. The preferred embodiments and materials described herein are intended to be exemplary only.
The mass percentages in the test examples which follow are based on the total weight of an aqueous solution formed from 99.9g of water and 0.1g of Sodium Lauryl Sulfate (SLS).
Test examples
Immediate stimulus relaxation test
Materials:
stimulus: 0.1 wt% SLS aqueous solution, i.e., an aqueous solution formed from 99.9g of water and 0.1g of Sodium Lauryl Sulfate (SLS)
Raw materials of a substance to be tested:
italian camomile extract: cleaning fresh flos Helianthi (above root), drying, grinding, pulverizing, and sieving with 150 mesh sieve and 200 mesh sieve to obtain flos Helianthi powder. Adding ethanol aqueous solution (ethanol concentration of 50-95 v/v% based on total volume of ethanol aqueous solution) as extractant into Italian flos Chrysanthemi powder, and extracting with heating at 50-70 deg.C for at least two times to obtain extractive solution. Combining the extractive solutions and collecting the concentrated solution. Distilling under reduced pressure at 40 deg.C, recovering ethanol from extract of above root of Italian Helichrysum L, adding appropriate amount of adjuvants such as glycerol, and storing.
Olive leaf extract: the olive leaves collected in 2 months or 5 months are used, and the storage time of the olive leaves for preparation is not more than 48 hours after the olive leaves are collected from trees. Cleaning fresh olive leaves, drying to remove water, and drying at 70-100 deg.C for 1-3 hr to obtain dry olive leaves. The dried leaves of Olea europaea are crushed to produce a coarse powder or smaller particles. Adding crushed olive leaves into a mixed solution of ethanol and water, wherein the concentration of the ethanol is 50-85 v/v% based on the total volume of the ethanol water solution, and then heating and extracting at 70-90 ℃ for 4-8 hours at least twice. Recovering ethanol solvent from Olea Europaea extractive solution, adding water and glycerol, precipitating with water, filtering, and storing.
Calendula extract: fresh marigold is picked, cleaned, dried in the sun, ground, crushed and sieved with 150-mesh and 200-mesh sieve to prepare the marigold powder. Adding herba Sidae Rhombifoliae powder into ethanol water solution (ethanol concentration is 50-70 v/v% based on total volume of ethanol water solution), soaking for 1-2 hr, extracting with ultrasonic circulation device at 40-60 deg.C for 4 times, each time for 20-30 min, and ultrasonic power of 100W. Mixing extractive solutions, filtering, distilling supernatant under reduced pressure to recover solvent, and collecting concentrated solution. Recovering ethanol solvent from herba Sidae Rhombifoliae extract, adding adjuvants such as glycerol, etc., and storing.
The substance to be tested: in 0.1 wt% SLS aqueous solution, raw materials of the substance to be detected in different concentrations based on the total weight of the 0.1 wt% SLS aqueous solution:
1 wt% of Parthenia italica extract
2 wt% of Parthenium italicum extract
1 wt% olive leaf extract
2 wt% olive leaf extract
1 wt% calendula extract
1 wt% of Italian Callicarpa extract +1 wt% of Olea europaea leaf extract
1 wt% of Italian Callicarpa extract +1 wt% of calendula extract
1 wt% of Italian Callicarpa extract +1 wt% of calendula extract +1 wt% of Olea europaea leaf extract
1 wt% Glycerol
The method comprises the following steps:
the irritant and the substance to be tested were placed in patch chambers, respectively, and applied to the back of the subject, and after 24 hours, the skin was uncovered and observed for 0.5h and 48h of skin irritation response. The sample size was 35 persons. The number of cases (representing the frequency of occurrence of the stimulus response) and the score (representing the severity of the stimulus response) in which the stimulus response occurred were counted, respectively, and the soothing effect (%) of the test material on the stimulus response with respect to SLS was calculated.
The comfort effect calculation formula is as follows: relieving effect (%) - (SLS score-raw material score to be measured)/SLS score 100
The mean irritation score (i.e., the mean irritation score-the sum of the irritation scores of the sample/total number of subjects) was used to evaluate the soothing immediate skin irritation effect of the composition, with higher irritation scores indicating greater immediate skin irritation for the sample
As a result:
see table 1 and fig. 1, table 2 and fig. 2, table 3 and fig. 3, and table 4 and fig. 4, respectively.
Table 1: number of stimulus response cases (0.5 hour)
Table 2: number of stimulus response cases (48 hours)
TABLE 3 soothing Effect (0.5 hours)
Table 4: soothing effect (48 hours)
Discussion: (the test substance "1% Italian Callicarpa extract + 1% Marigold extract" in each of the above tables is referred to as "combination A", the test substance "1% Italian Callicarpa extract + 1% Marigold extract + 1% Olea europaea leaf extract" is referred to as "combination B", and the test substance "1% Italian Callicarpa extract + 1% Olea europaea leaf extract" is referred to as "combination C")
1. Of the 3 tested components, only the italian wintersweet extract significantly reduced the number of irritants at 0.5h and showed a significant soothing effect, probably because the italian wintersweet extract promotes the action of beta-endorphins produced by keratinocytes on the nerve pathways, which has the effect of quickly soothing symptoms; there was no significant difference in the effect of the Italian chamomile extract at concentrations of 1% or 2% over the various time periods. Therefore 1% was used as the test concentration for this component in the subsequent combination test.
2. The olive leaf extract is a common anti-inflammatory and antioxidant raw material, and has a certain relieving effect (25%) at a concentration of 2% without obviously reducing the number of irritation cases within 0.5 h; the soothing effect was still present at 48 hours, but there was no significant difference between 1% and 2%. The olive leaf extract is very easy to be oxidized and discolored, and is difficult to apply in a formula. The test concentration of 1% was used as the component in the subsequent combination test.
3. The calendula extract has anti-inflammatory and antioxidant capabilities, and generates certain stimulation (-25%) within 0.5h, and generates certain relieving effect within 48 h; the concentration of the component is not obviously different at 1% or 2%, and the stability of the formula is damaged due to easy discoloration, and 1% is used as the testing concentration of the component in the subsequent combination test.
4. The number of cases of stimulus response of combination C at 0.5h (3) was superior to that of either Olea europaea leaf extract (7) or calendula officinalis extract (8) alone, but the effect was similar to that of Italian chamomilla extract (3) alone, indicating immediate soothing efficacy;
5. the stimulation response example number (4) of the combination C at 48h is better than that of all the raw material used independently, such as olive leaf extract (6), calendula extract (8) or Italian chamomile (6), and the combination C has long-term soothing effect which is better than that of all the raw material used independently;
6. The results in table 3 show that olive leaf extract did not affect the positive effect of combination C on a reduction of 0.5h stimulation (still reducing stimulation by 50%); in the aspect of the relieving degree, the combination C improves the relieving degree to 50 percent, is better than the addition of any one raw material used independently, and has multiplication effect;
7. the results in tables 2 and 4 show that the olive leaf extract also enhances the sustained relaxing effect of the italian chrysanthemum extract at 48 hours, the number of stimulation cases caused by SLS can be reduced from 6 to 4, and the relaxing effect is improved from 33.3% to 55.6%, which indicates that the combination of the italian chrysanthemum extract and the olive leaf extract has a remarkable effect in long-term relaxing;
8. comparing combinations a and C, combination C is best at 0.5h for relief, while combination a has little effect during this time period; at 48h, both combinations were effective, but combination C was better than a; indicating that combination C works best at a compromise of immediate relief and sustained action.
9. Compared with the combination B and the combination C, the combination of the three components in the combination B does not have synergistic effect, even generates antagonism to some extent, but reduces the effect; combination B is very close to combination C in the number of stimulation instances of 0.5h, while combination C is twice as much as combination B in the soothing effect of this period; the effect of combination C and combination B was also very similar over the number of stimulation cases of 48h, while combination B was only slightly higher in soothing effect than combination C; the invention can quickly relieve the skin discomfort of consumers and play a long-acting consolidation role, and the combination C is the optimal combination in two aspects.
10. Glycerol and deionized water as solvents for the plant extract had little effect when used alone, indicating that the efficacy of combination C came primarily from the plant extract itself.
Use example 1
Preparation of cream
Heating 1.5g of glyceryl stearate, 2g of glyceryl stearate/PEG-100 stearate complex, 5g of isomeric hexadecane, 5g of isooctyl palmitate and 4g of cetostearyl alcohol until the isomeric hexadecane, the isooctyl palmitate and the cetostearyl alcohol are dissolved (oil phase), adding 4g of glycerol and 1g of butanediol into 77g of deionized water, heating to 75 ℃ (water phase), adding the oil phase into the water phase while homogenizing, emulsifying for 5min, cooling to below 30 ℃, and adding 2g of Italian wintersweet extract, 2g of olea europaea leaf extract and a proper amount of preservative and essence.
Use example 2
Preparation of the emulsion
Heating 1.5g glyceryl monostearate, 2g glyceryl stearate/PEG-100 stearate complex, 5g polydimethylsiloxane, 5g ethylhexyl palmitate, 2g cetyl stearyl alcohol to dissolve (oil phase), adding 71g deionized water into 5g of glycerol, 3g1, 3-butanediol, 0.25g carbomer, 0.02g EDTA2 Na, heating to 75 deg.C (water phase), adding oil phase into water phase while homogenizing, emulsifying for 5min, cooling to below 30 deg.C, adding Italian flos Chrysanthemi extract 1g and Olea Europaea Wilfordii leaf extract 1g, 0.1g potassium hydroxide, and appropriate amount of antiseptic and essence.
Use example 3
Preparation of essence
Adding 1g of betaine, 5g of glycerol, 0.05g of sodium hyaluronate, 4g of propylene glycol, 0.05g of xanthan gum and 0.15g of hydroxyethyl cellulose into 85g of deionized water, heating to 75 ℃, homogenizing for a few minutes, cooling to below 30 ℃, adding 3g of Italian wintersweet extract and 3g of olive leaf extract, and a proper amount of preservative and essence, and uniformly stirring.
Use example 4
Preparation of toner
Adding 1g of betaine, 5g of glycerol, 0.05g of sodium hyaluronate, 2g of butanediol and 0.5g of xanthan gum into 81g of deionized water, heating to 75 ℃, homogenizing for a few minutes, cooling to below 30 ℃, adding 1g of Italian helichrysum extract and 1g of olive leaf extract, and appropriate amount of preservative and essence, and uniformly stirring.
Use example 5
Preparation of jelly
Adding 1g of betaine, 7g of glycerol, 3g of 1, 3-butanediol, 0.3g of carbomer and 0.02g of EDTA 2Na into 83g of deionized water, heating to 75 ℃, homogenizing for a few minutes, cooling to 45 ℃, adding 1g of Italian helichrysum extract, 1g of olive leaf extract, 0.1g of potassium hydroxide and a proper amount of preservative and essence, and stirring uniformly.
The foregoing is merely a preferred embodiment of the invention and is not intended to limit the scope of the invention, which is defined by the claims appended hereto, and any other technical entity or method that is encompassed by the claims as broadly defined herein, or equivalent variations thereof, is contemplated as being encompassed by the claims.
Claims (6)
1. A composition consisting of an extract of italian camomile and an extract of olive leaves; the mass ratio of the Italian wintersweet extract to the olive leaf extract in the composition is 1: 0.5-2;
the Italian chamomile extract is obtained by the following steps: drying the parts above the roots of the Italian Helichrysum L, grinding, crushing, and sieving by a sieve of 150 meshes and 200 meshes to obtain Italian Helichrysum L powder; adding ethanol water solution into Italian Helichrysum powder, wherein the ethanol concentration is 50-95 v/v% based on the total volume of the ethanol water solution, and heating and extracting at 50-70 deg.C for at least two times to obtain extractive solution; mixing the extractive solutions and collecting concentrated solution; distilling under reduced pressure at 40 deg.C, recovering ethanol from extract of the part above the root of Parthenia italica, adding adjuvants, and storing;
the olive leaf extract is obtained by the following steps: cleaning olive leaves collected in 2 or 5 months and stored for no more than 48 hours, drying to remove water, and drying at 70-100 deg.C for 1-3 hours to obtain dried olive leaves; crushing dry leaves of Olea europaea to obtain coarse powder or granules; adding crushed olive leaves into a mixed solution of ethanol and water, wherein the concentration of the ethanol is 50-85 v/v% based on the total volume of an ethanol water solution, and then heating and extracting for at least two times, wherein the extraction temperature is 70-90 ℃, and the extraction time is 4-8 hours; recovering ethanol solvent from Olea Europaea extractive solution, adding water and adjuvants, precipitating with water again, filtering, and storing.
2. A method of preparing the composition of claim 1, comprising the steps of: mixing the Italian chamomile extract with the olive leaf extract to obtain the composition as claimed in claim 1.
3. Use of a composition according to claim 1 as a cosmetic or personal care product; or for the preparation of cosmetic or personal care products.
4. The use of claim 3, wherein said personal care product comprises a cream, lotion, serum, lotion, and gel.
5. A cosmetic or personal care product characterized in that it comprises a composition according to claim 1 and a cosmetically acceptable carrier.
6. A process for the preparation of a cosmetic or personal care product according to claim 5, characterized in that it comprises the steps of: mixing an emulsified or non-emulsified cosmetically acceptable carrier with the composition of claim 1 to obtain the cosmetic or personal care product of claim 5.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010771848.XA CN111686054B (en) | 2020-08-04 | 2020-08-04 | Plant composition with quick and sustained effects and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010771848.XA CN111686054B (en) | 2020-08-04 | 2020-08-04 | Plant composition with quick and sustained effects and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111686054A CN111686054A (en) | 2020-09-22 |
| CN111686054B true CN111686054B (en) | 2022-07-29 |
Family
ID=72486499
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010771848.XA Active CN111686054B (en) | 2020-08-04 | 2020-08-04 | Plant composition with quick and sustained effects and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111686054B (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114376931A (en) * | 2021-12-30 | 2022-04-22 | 原田(苏州)技术研发有限公司 | Composition for repairing eyelashes and application thereof |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102004050563A1 (en) * | 2004-10-15 | 2006-04-20 | Henkel Kgaa | Topical cosmetic and dermatological composition containing alkylurea and other active agent, useful for treating e.g. ageing of the skin and pigmentation disorders |
| EP3035946A1 (en) * | 2013-08-20 | 2016-06-29 | SWM Luxembourg s.a.r.l. | Product comprising a plant for medicinal, cosmetic, coloring or dermatologic use |
| CN103432043B (en) * | 2013-08-30 | 2015-04-22 | 广州丹奇日用化工厂有限公司 | Face mask containing plant material residues |
| US9744120B2 (en) * | 2015-05-28 | 2017-08-29 | IndusCo, Ltd. | Durable skin sanitizers containing water stable antimicrobial silanol quaternary ammonium compounds |
| CN107823023A (en) * | 2017-12-21 | 2018-03-23 | 李冬梅 | A kind of natural plant extracts composition and its application in cosmetics |
-
2020
- 2020-08-04 CN CN202010771848.XA patent/CN111686054B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN111686054A (en) | 2020-09-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20220218595A1 (en) | Method of using soothing cream including maca root extract as an active ingredient | |
| KR101236946B1 (en) | Cosmetic Composition Comprising Naturalplant Extract for Alleviating Atopic Dermatitis | |
| KR101562560B1 (en) | Natural extract fermentation-maturity composition for preventing skin aging and improving skin wrinkle, Natural cosmetics containing that and Manufacturing method thereof | |
| KR102576693B1 (en) | Cosmetic composition comprising infused oils of calendular and mixed extracts of plants for moisturizing skin | |
| JP4495406B2 (en) | Sendangusa plant extract-containing composition | |
| CN111686054B (en) | Plant composition with quick and sustained effects and application thereof | |
| KR101732906B1 (en) | Skin care and atopic dermatitis cosmetic compositions containing asiatic pennywort extract, magnolia bark extract, hiba arborvitae branch extract, eucalyptus oil and borage seed oil | |
| CN111789799A (en) | Plant composition with red-removing, anti-inflammatory and relieving effects and application thereof | |
| EP1980241B1 (en) | Wet wipes with vitex agnus castus extract for feminine personal care | |
| KR20110030812A (en) | Cosmetic composition for sensitive skin and scalp | |
| WO2006069072A2 (en) | Method and composition for reducing the appearance of wrinkles | |
| JP5325727B2 (en) | Sendangusa plant extract-containing composition | |
| CN115844784B (en) | Composition for relieving inflammation of dry skin and application and preparation method thereof | |
| JP4105498B2 (en) | A composition effective for prevention and alleviation of symptoms of atopic disease | |
| CN116570546A (en) | Traditional Chinese medicine skin-moistening and whitening body lotion and preparation method thereof | |
| CN111184644B (en) | Application of artemisinin as antiallergic component in cosmetics | |
| KR102085064B1 (en) | Manufacturing method of eco-friendly cleaner composition comprising extract of cianidanol and terpene | |
| KR20230077990A (en) | Cosmetic composition for alleviating skin irritation by vitamin c containing mixed extracts of rumex crispus and cimicifuga dahurica root | |
| EP3482764B1 (en) | Insect and/or spider repellent comprising black cumin oil | |
| KR20040065498A (en) | Allergic dermatitis and skin itching relieving composition containing natural herb extract | |
| KR20220134118A (en) | Cosmetic composition for atopic skin using herbal extracts | |
| KR102874415B1 (en) | Cosmetic composition with skin moisturizing, pruritis improving, and skin barrier enhancing property and skin care product usint the same | |
| KR20220005716A (en) | Natural extractfermentation-maturity composition for preventing skin aging and improving skin wrinkle, Natural cosmetics containing that and Manufacturing method thereof | |
| KR102495027B1 (en) | Agent for mitigating hot flash, cosmetic product, and method of using cosmetic product | |
| KR102921758B1 (en) | Cosmetic composition for sensitive skin containing nano-soluble curcumin |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |