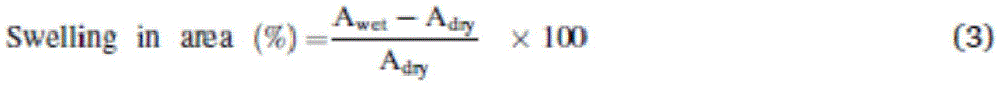CN111682247B - Doped sulfonated polyarylethersulfone composite material and preparation method and application thereof - Google Patents
Doped sulfonated polyarylethersulfone composite material and preparation method and application thereof Download PDFInfo
- Publication number
- CN111682247B CN111682247B CN202010499350.2A CN202010499350A CN111682247B CN 111682247 B CN111682247 B CN 111682247B CN 202010499350 A CN202010499350 A CN 202010499350A CN 111682247 B CN111682247 B CN 111682247B
- Authority
- CN
- China
- Prior art keywords
- spaes
- hcnt
- hours
- mixed liquid
- sulfonated
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000002131 composite material Substances 0.000 title claims abstract description 48
- 229920000110 poly(aryl ether sulfone) Polymers 0.000 title claims abstract description 38
- 238000002360 preparation method Methods 0.000 title claims abstract description 12
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 70
- 239000002041 carbon nanotube Substances 0.000 claims description 62
- 229910021393 carbon nanotube Inorganic materials 0.000 claims description 60
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 54
- 239000007788 liquid Substances 0.000 claims description 40
- 238000010438 heat treatment Methods 0.000 claims description 30
- 238000010992 reflux Methods 0.000 claims description 27
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 claims description 26
- 238000001035 drying Methods 0.000 claims description 25
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 22
- 238000006243 chemical reaction Methods 0.000 claims description 18
- 239000007787 solid Substances 0.000 claims description 16
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 claims description 14
- 239000008367 deionised water Substances 0.000 claims description 11
- 229910021641 deionized water Inorganic materials 0.000 claims description 11
- 239000011521 glass Substances 0.000 claims description 10
- 239000000047 product Substances 0.000 claims description 9
- 239000000706 filtrate Substances 0.000 claims description 8
- 239000012265 solid product Substances 0.000 claims description 8
- 238000002156 mixing Methods 0.000 claims description 6
- 238000001326 spin-polarised Auger electron spectroscopy Methods 0.000 claims description 5
- -1 histidine modified carbon nano tube Chemical class 0.000 claims description 2
- 238000009210 therapy by ultrasound Methods 0.000 claims description 2
- 229910021392 nanocarbon Inorganic materials 0.000 abstract description 53
- 239000003575 carbonaceous material Substances 0.000 abstract description 33
- 239000000463 material Substances 0.000 abstract description 22
- 229920000642 polymer Polymers 0.000 abstract description 15
- 239000003607 modifier Substances 0.000 abstract description 11
- 125000003010 ionic group Chemical group 0.000 abstract description 10
- 150000001413 amino acids Chemical class 0.000 abstract description 8
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 abstract description 7
- 230000001976 improved effect Effects 0.000 abstract description 7
- 229920000090 poly(aryl ether) Polymers 0.000 abstract description 7
- 125000003275 alpha amino acid group Chemical group 0.000 abstract 1
- 239000012528 membrane Substances 0.000 description 79
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 27
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 18
- 230000009102 absorption Effects 0.000 description 17
- 238000010521 absorption reaction Methods 0.000 description 17
- 239000000243 solution Substances 0.000 description 16
- 238000012360 testing method Methods 0.000 description 16
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical group CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 14
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 12
- 239000000523 sample Substances 0.000 description 12
- 239000000945 filler Substances 0.000 description 10
- 230000001965 increasing effect Effects 0.000 description 10
- 230000003993 interaction Effects 0.000 description 10
- 239000011159 matrix material Substances 0.000 description 10
- 238000005259 measurement Methods 0.000 description 10
- 239000000203 mixture Substances 0.000 description 9
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 9
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 8
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 8
- 238000005406 washing Methods 0.000 description 8
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 7
- 230000007423 decrease Effects 0.000 description 7
- 238000000445 field-emission scanning electron microscopy Methods 0.000 description 7
- 238000001914 filtration Methods 0.000 description 7
- 238000001878 scanning electron micrograph Methods 0.000 description 7
- 238000004630 atomic force microscopy Methods 0.000 description 6
- 238000009792 diffusion process Methods 0.000 description 6
- 230000000694 effects Effects 0.000 description 6
- 150000003254 radicals Chemical class 0.000 description 6
- 230000008961 swelling Effects 0.000 description 6
- 230000002378 acidificating effect Effects 0.000 description 5
- 230000008859 change Effects 0.000 description 5
- 238000007306 functionalization reaction Methods 0.000 description 5
- 229910052739 hydrogen Inorganic materials 0.000 description 5
- 239000001257 hydrogen Substances 0.000 description 5
- 239000011256 inorganic filler Substances 0.000 description 5
- 229910003475 inorganic filler Inorganic materials 0.000 description 5
- 239000005518 polymer electrolyte Substances 0.000 description 5
- 230000002829 reductive effect Effects 0.000 description 5
- 238000001179 sorption measurement Methods 0.000 description 5
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical group OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 4
- 230000015556 catabolic process Effects 0.000 description 4
- 238000004140 cleaning Methods 0.000 description 4
- 238000006731 degradation reaction Methods 0.000 description 4
- 238000002149 energy-dispersive X-ray emission spectroscopy Methods 0.000 description 4
- 239000000446 fuel Substances 0.000 description 4
- 150000002500 ions Chemical class 0.000 description 4
- 239000007791 liquid phase Substances 0.000 description 4
- 239000011259 mixed solution Substances 0.000 description 4
- 230000010287 polarization Effects 0.000 description 4
- 238000004626 scanning electron microscopy Methods 0.000 description 4
- VWDWKYIASSYTQR-UHFFFAOYSA-N sodium nitrate Chemical compound [Na+].[O-][N+]([O-])=O VWDWKYIASSYTQR-UHFFFAOYSA-N 0.000 description 4
- 230000000930 thermomechanical effect Effects 0.000 description 4
- 208000016261 weight loss Diseases 0.000 description 4
- 238000005033 Fourier transform infrared spectroscopy Methods 0.000 description 3
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 239000004696 Poly ether ether ketone Substances 0.000 description 3
- 125000003118 aryl group Chemical group 0.000 description 3
- 230000018044 dehydration Effects 0.000 description 3
- 238000006297 dehydration reaction Methods 0.000 description 3
- 229940113088 dimethylacetamide Drugs 0.000 description 3
- 238000000295 emission spectrum Methods 0.000 description 3
- 230000009477 glass transition Effects 0.000 description 3
- 230000010354 integration Effects 0.000 description 3
- 238000000034 method Methods 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 239000012071 phase Substances 0.000 description 3
- 229920002530 polyetherether ketone Polymers 0.000 description 3
- 238000001953 recrystallisation Methods 0.000 description 3
- 238000001228 spectrum Methods 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- WYTZZXDRDKSJID-UHFFFAOYSA-N (3-aminopropyl)triethoxysilane Chemical compound CCO[Si](OCC)(OCC)CCCN WYTZZXDRDKSJID-UHFFFAOYSA-N 0.000 description 2
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 2
- 102000004310 Ion Channels Human genes 0.000 description 2
- 229920000557 Nafion® Polymers 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 230000002776 aggregation Effects 0.000 description 2
- 238000004220 aggregation Methods 0.000 description 2
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- 230000008030 elimination Effects 0.000 description 2
- 238000003379 elimination reaction Methods 0.000 description 2
- 238000000349 field-emission scanning electron micrograph Methods 0.000 description 2
- 125000000487 histidyl group Chemical group [H]N([H])C(C(=O)O*)C([H])([H])C1=C([H])N([H])C([H])=N1 0.000 description 2
- 125000001867 hydroperoxy group Chemical group [*]OO[H] 0.000 description 2
- 125000002883 imidazolyl group Chemical group 0.000 description 2
- 238000005470 impregnation Methods 0.000 description 2
- 238000010348 incorporation Methods 0.000 description 2
- 238000005342 ion exchange Methods 0.000 description 2
- 238000011068 loading method Methods 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 230000000877 morphologic effect Effects 0.000 description 2
- 230000035699 permeability Effects 0.000 description 2
- KJFMBFZCATUALV-UHFFFAOYSA-N phenolphthalein Chemical compound C1=CC(O)=CC=C1C1(C=2C=CC(O)=CC=2)C2=CC=CC=C2C(=O)O1 KJFMBFZCATUALV-UHFFFAOYSA-N 0.000 description 2
- 238000002047 photoemission electron microscopy Methods 0.000 description 2
- 229920001483 poly(ethyl methacrylate) polymer Polymers 0.000 description 2
- 229920002465 poly[5-(4-benzoylphenoxy)-2-hydroxybenzenesulfonic acid] polymer Polymers 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 150000003839 salts Chemical class 0.000 description 2
- 238000000235 small-angle X-ray scattering Methods 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 235000010344 sodium nitrate Nutrition 0.000 description 2
- 238000002336 sorption--desorption measurement Methods 0.000 description 2
- 238000006277 sulfonation reaction Methods 0.000 description 2
- 125000000542 sulfonic acid group Chemical group 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 238000001757 thermogravimetry curve Methods 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- 238000001291 vacuum drying Methods 0.000 description 2
- SXGMVGOVILIERA-UHFFFAOYSA-N (2R,3S)-2,3-diaminobutanoic acid Natural products CC(N)C(N)C(O)=O SXGMVGOVILIERA-UHFFFAOYSA-N 0.000 description 1
- PLVUIVUKKJTSDM-UHFFFAOYSA-N 1-fluoro-4-(4-fluorophenyl)sulfonylbenzene Chemical compound C1=CC(F)=CC=C1S(=O)(=O)C1=CC=C(F)C=C1 PLVUIVUKKJTSDM-UHFFFAOYSA-N 0.000 description 1
- 125000003287 1H-imidazol-4-ylmethyl group Chemical group [H]N1C([H])=NC(C([H])([H])[*])=C1[H] 0.000 description 1
- HEMGYNNCNNODNX-UHFFFAOYSA-N 3,4-diaminobenzoic acid Chemical compound NC1=CC=C(C(O)=O)C=C1N HEMGYNNCNNODNX-UHFFFAOYSA-N 0.000 description 1
- 238000012935 Averaging Methods 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 1
- 239000004215 Carbon black (E152) Substances 0.000 description 1
- 238000001157 Fourier transform infrared spectrum Methods 0.000 description 1
- 239000004693 Polybenzimidazole Substances 0.000 description 1
- 229910006069 SO3H Inorganic materials 0.000 description 1
- 229920004695 VICTREX™ PEEK Polymers 0.000 description 1
- 238000002441 X-ray diffraction Methods 0.000 description 1
- MCMNRKCIXSYSNV-UHFFFAOYSA-N ZrO2 Inorganic materials O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 238000002479 acid--base titration Methods 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 150000001412 amines Chemical group 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- 229920005601 base polymer Polymers 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- VCCBEIPGXKNHFW-UHFFFAOYSA-N biphenyl-4,4'-diol Chemical group C1=CC(O)=CC=C1C1=CC=C(O)C=C1 VCCBEIPGXKNHFW-UHFFFAOYSA-N 0.000 description 1
- 239000002134 carbon nanofiber Substances 0.000 description 1
- 239000002238 carbon nanotube film Substances 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical group 0.000 description 1
- 125000002843 carboxylic acid group Chemical group 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- CETPSERCERDGAM-UHFFFAOYSA-N ceric oxide Chemical compound O=[Ce]=O CETPSERCERDGAM-UHFFFAOYSA-N 0.000 description 1
- 229910000422 cerium(IV) oxide Inorganic materials 0.000 description 1
- 238000002144 chemical decomposition reaction Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 239000012459 cleaning agent Substances 0.000 description 1
- 229910052681 coesite Inorganic materials 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 229910052906 cristobalite Inorganic materials 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000003795 desorption Methods 0.000 description 1
- 239000003599 detergent Substances 0.000 description 1
- YWEUIGNSBFLMFL-UHFFFAOYSA-N diphosphonate Chemical compound O=P(=O)OP(=O)=O YWEUIGNSBFLMFL-UHFFFAOYSA-N 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 239000002019 doping agent Substances 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 239000003344 environmental pollutant Substances 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- 238000007731 hot pressing Methods 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 238000001566 impedance spectroscopy Methods 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 238000009830 intercalation Methods 0.000 description 1
- 230000002687 intercalation Effects 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 230000000873 masking effect Effects 0.000 description 1
- 230000003340 mental effect Effects 0.000 description 1
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical class C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 1
- 239000002808 molecular sieve Substances 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 239000002048 multi walled nanotube Substances 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- DLYUQMMRRRQYAE-UHFFFAOYSA-N phosphorus pentoxide Inorganic materials O1P(O2)(=O)OP3(=O)OP1(=O)OP2(=O)O3 DLYUQMMRRRQYAE-UHFFFAOYSA-N 0.000 description 1
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Substances [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 1
- 238000000717 platinum sputter deposition Methods 0.000 description 1
- 231100000719 pollutant Toxicity 0.000 description 1
- 229920001643 poly(ether ketone) Polymers 0.000 description 1
- 229920002480 polybenzimidazole Polymers 0.000 description 1
- 238000006068 polycondensation reaction Methods 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 239000012286 potassium permanganate Substances 0.000 description 1
- 230000001376 precipitating effect Effects 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 230000006798 recombination Effects 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 238000002791 soaking Methods 0.000 description 1
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000004317 sodium nitrate Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 229910052682 stishovite Inorganic materials 0.000 description 1
- HIFJUMGIHIZEPX-UHFFFAOYSA-N sulfuric acid;sulfur trioxide Chemical compound O=S(=O)=O.OS(O)(=O)=O HIFJUMGIHIZEPX-UHFFFAOYSA-N 0.000 description 1
- 235000011149 sulphuric acid Nutrition 0.000 description 1
- 238000006557 surface reaction Methods 0.000 description 1
- 230000003746 surface roughness Effects 0.000 description 1
- 230000002459 sustained effect Effects 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N titanium dioxide Inorganic materials O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 229910052905 tridymite Inorganic materials 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
- 239000013585 weight reducing agent Substances 0.000 description 1
- 239000010457 zeolite Substances 0.000 description 1
- LEHFSLREWWMLPU-UHFFFAOYSA-B zirconium(4+);tetraphosphate Chemical class [Zr+4].[Zr+4].[Zr+4].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O LEHFSLREWWMLPU-UHFFFAOYSA-B 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/1041—Polymer electrolyte composites, mixtures or blends
- H01M8/1046—Mixtures of at least one polymer and at least one additive
- H01M8/1048—Ion-conducting additives, e.g. ion-conducting particles, heteropolyacids, metal phosphate or polybenzimidazole with phosphoric acid
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/1069—Polymeric electrolyte materials characterised by the manufacturing processes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M2008/1095—Fuel cells with polymeric electrolytes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/30—Hydrogen technology
- Y02E60/50—Fuel cells
Landscapes
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Sustainable Development (AREA)
- Sustainable Energy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Composite Materials (AREA)
- Carbon And Carbon Compounds (AREA)
- Compositions Of Macromolecular Compounds (AREA)
Abstract
The invention discloses a doped sulfonated polyarylethersulfone composite material and a preparation method and application thereof. The sulfonated polyarylethersulfone-doped composite material comprises sulfonated polyarylethersulfone and a modified nano carbon material doped in the sulfonated polyarylethersulfone, wherein the modified nano carbon material is a nano carbon material treated by a modifier, and the modifier is amino acid. The sulfonated polyarylether sulfone-doped composite material comprises sulfonated polyarylether sulfone and a modified nanocarbon material doped in the sulfonated polyarylether sulfone, wherein when amino acid is used for modifying the nanocarbon material, the amino acid can be combined with carboxyl (-COOH) and other groups carried by the nanocarbon material, so that the surface of the obtained modified nanocarbon material is provided with additional ionic groups, the proton conductivity can be improved by improving the density of the ionic groups, and the mechanical integrity is improved by recombining a polymer chain, so that the sulfonated polyarylether sulfone-doped composite material has higher proton conductivity.
Description
Technical Field
The invention relates to the field of polymer composite materials, in particular to a doped sulfonated polyarylethersulfone composite material and a preparation method and application thereof.
Background
Polymer Electrolyte Fuel Cells (PEFCs) have received a great deal of attention as clean and efficient electrochemical energy carriers due to their unique energy density, undetectable pollutant emissions, and system simplicity. Applications of PEFCs range from stationary electrical equipment to automobiles, and although receiving widespread attention, have been limited in ubiquitous commercial applications due to key issues such as poor performance at low Relative Humidity (RH), poor durability, and high cost. Therefore, research has been directed to improving performance at low relative humidity to reduce the use of expensive ancillary components (e.g., humidifiers).
One key component of PEFCs in terms of performance and production cost is their Polymer Electrolyte Membrane (PEM), which functions as both the positive and negative proton transporters and separators. Key characteristics of an ideal PEM include high proton conductivity, good water transport, low electron crossover, thermo-mechanical stability, low fuel permeability, and durability under various operating conditions. To date, the most advanced PEM for PEFCs is Nafion, dupont, which has excellent proton conductivity, low electron conductivity, and good chemical-mechanical stability. However, the widespread use of Nafion in PEFCs is hampered by the high cost, loss of proton conductivity at RHs below 20%, and environmental and mental incompatibility. There have been some attempts to develop alternative PEMs for petrochemical companies.
Hydrocarbon-based aromatic polymers are a material of great interest because of their low cost, flexibility in synthesis and molecular design, film forming tendency, and thermo-mechanical properties, which enable their use in a variety of applications, including rechargeable batteries, fuel cells, and separation science, among others. Sulfonated forms of polyimines, polyether ether ketone (PEEK), polyaryl ether sulfone, polyether ketone, and polybenzimidazole, among other aromatic polymers, have been extensively studied as base backbones for PEM fabrication. Among them, polyarylethersulfone (SPAES) is a polymer having proton conductivity in nature, and the proton conductivity and thermo-mechanical properties thereof can be adjusted by sulfonation (DS) to various degrees. Thus, the utilization of SPAES for PEM applications has been increasing. In the plasma atomic emission spectrum, each proton carries several water molecules, which are transported through the PEM by electroosmotic resistance. Thus, the proton conductivity of the PEM is directly dependent on the presence of water molecules to dissolve the protons of the sulfonic acid groups. Unfortunately, electroosmotic drugs in low RH operation can cause dehydration of the anode, resulting in reduced performance of PEFCs. Furthermore, drying the anode catalyst layer can result in a reduction in current density, especially when using thicker PEMs. Overall, low relative humidity (< 20%) and high temperature (>90C) can hinder the operation of PEFCs with SPAES, thus preserving the properties of bare SPAES.
These low RH problems have been overcome by the integration of hygroscopic inorganic fillers in the PEM, such as zeolites, zirconium phosphates, CeO2、SiO2、ZrO2And TiO2They can improve proton conductivity in low RH. The combination of the inorganic filler with the SPAES matrix results in the recombination of ion channels including intercalation of the inorganic filler and sulfonic acid (-SO)3H) And (4) recombining the groups. Such a minute hygroscopic filler plays a crucial role in the moisture retention and proton transfer of the mixed film. Improved water retention and efficient water diffusion should reduce the ohmic resistance and cathode of the PEMUtilization ratio of the catalyst. However, in different cases, the proton conductivity decreases with the addition of the inorganic filler, due to the lower proton conductivity of the filler itself, and the effect of dilution on the proton exchange primer in the base polymer. Furthermore, the weak interaction between the inorganic filler and the polymer matrix may prevent a homogeneous distribution of the filler in the polymer.
Surface functionalization of acidic or basic fillers is an effective way to solve the above problems. Acidic groups such as sulfonic acid, phosphoric acid, and carboxylic acid or amino groups (-NH) have been reported2) And the functionalization of basic groups such as nitrogen heterocyclic rings can increase the density of PEM ionic groups and promote the proton conductivity of PEM. Although acidic groups have been widely used to modify fillers, acidic groups often reduce proton conductivity under low RH conditions due to high sensitivity to water molecules. In fact, the basic groups, due to their self-ionizing and self-dehydrating properties, have sustained proton conductivity under low RH conditions, and are more suitable for modifying fillers under low RH conditions. The acidic groups in the polymer matrix interact with the basic groups in the filler through hydrogen bonding bridges and electrostatic forces. The resulting acid-base PEM may be expected to have lower water absorption, lower fuel permeability, higher proton conductivity, good thermo-mechanical properties and flexibility.
That is, a polymer composite having high proton conductivity is currently lacking.
Disclosure of Invention
Based on this, there is a need to provide a doped sulfonated polyarylethersulfone composite material, which has higher proton conductivity.
In addition, a preparation method of the doped sulfonated polyarylethersulfone composite material and an application of the doped sulfonated polyarylethersulfone composite material are also necessarily provided.
The sulfonated polyarylethersulfone-doped composite material comprises sulfonated polyarylethersulfone and a modified nano carbon material doped in the sulfonated polyarylethersulfone, wherein the modified nano carbon material is a nano carbon material treated by a modifier, and the modifier is amino acid.
The preparation method of the doped sulfonated polyarylethersulfone composite material comprises the following steps:
pretreating a nano carbon material, and then modifying the pretreated nano carbon material by using an improver solution to obtain a modified nano carbon material; and
and uniformly mixing the modified nano carbon material and the sulfonated polyarylether sulfone under a liquid phase condition, and drying to obtain the doped sulfonated polyarylether sulfone composite material.
The doped sulfonated polyarylethersulfone composite material is applied to the field of preparing polymer electrolyte membranes.
The sulfonated polyarylether sulfone-doped composite material comprises sulfonated polyarylether sulfone and a modified nanocarbon material doped in the sulfonated polyarylether sulfone, wherein when amino acid is used for modifying the nanocarbon material, the amino acid can be combined with carboxyl (-COOH) and other groups carried by the nanocarbon material, so that the surface of the obtained modified nanocarbon material is provided with additional ionic groups, the proton conductivity can be improved by improving the density of the ionic groups, and the mechanical integrity is improved by recombining a polymer chain, so that the sulfonated polyarylether sulfone-doped composite material has higher proton conductivity.
Drawings
In order to more clearly illustrate the embodiments of the present invention or the technical solutions in the prior art, the drawings used in the description of the embodiments or the prior art will be briefly described below, it is obvious that the drawings in the following description are only some embodiments of the present invention, and for those skilled in the art, other drawings can be obtained according to the drawings without creative efforts.
Wherein:
FIG. 1 is a diagram of the mechanism of synthesis of sulfonated polyarylethersulfones.
FIG. 2 is a schematic diagram of the preparation scheme of SPAES/HCNT in example 1.
FIG. 3a is an FE-SEM image of CNTs according to example 1.
FIG. 3b is a FE-SEM picture of HCNT from example 1.
FIG. 3c is a FE-SEM image of CNTs according to example 2.
FIG. 3d is a FE-SEM picture of HCNT from example 2.
Figure 3e is an SEM image of CNTs from example 1.
Figure 3f is an SEM image of HCNT from example 1.
FIG. 4a is an SEM photograph of SPAES in example 3.
FIG. 4b is an SEM picture of SPAES/HCNT 1.5 from example 3.
FIG. 4c is the EDAX spectrum of SPAES from example 3.
FIG. 4d is the EDAX spectrum of SPAES/HCNT 1.5 from example 3.
FIG. 5a is an SEM picture (scale bar 10 μm) of the cross section of SPAES in example 3.
FIG. 5b is an SEM picture (scale bar 10 μm) of the cross-section of SPAES/HCNT 1.5 from example 3.
FIG. 5c is an SEM image of the cross section of SPAES in example 3 (scale bar 2 μm).
FIG. 5d is an SEM picture (scale bar 2 μm) of the cross-section of SPAES/HCNT 1.5 from example 3.
FIG. 5e is an SEM picture (scale bar 100nm) of the cross section of SPAES in example 3.
FIG. 5f is an SEM picture (200 nm on a scale) of the cross-section of SPAES/HCNT 1.5 from example 3.
FIG. 6 is a comparison of AFM of SPAES and SPAES/HCNT 1.5 in example 3, where a and c are AFM maps of SPAES and b and c are AFM maps of SPAES/HCNT 1.5.
FIG. 7 is a FT-IR spectrum of CNT and HCNT of example 1.
FIG. 8a is a TGA plot of CNT, HCNT, and SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2, and SPAES/HCNT 1.5 of example 3.
FIG. 8b is the UTM plot of SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2, and SPAES/HCNT 1.5 of example 3.
FIG. 8c is a DSC scan of SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2, and SPAES/HCNT 1.5 of example 3.
FIG. 8d is a DSC two-pass scan of SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2, and SPAES/HCNT 1.5 of example 3.
FIG. 9 is the water contact angle measurement chart of SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2, and SPAES/HCNT 1.5 of example 3, wherein a is SPAES, b is SPAES/HCNT 0.5, c is SPAES/HCNT1, and d is SPAES/HCNT 1.5.
FIGS. 10a and 10b are graphs showing the change in mass of SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2, and SPAES/HCNT 1.5 of example 3 at different relative humidities.
FIG. 10c is a graph showing the proton conductivity changes at different temperatures for SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2, and SPAES/HCNT 1.5 of example 3 under the condition of 100% relative humidity.
FIG. 10d is a graph showing the proton conductivity changes at different relative humidities of SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2, and SPAES/HCNT 1.5 of example 3 at a temperature of 80 ℃.
FIG. 10e is a graph showing the proton conductivity change of Nafion-115, SPAES/CNT 1.5 of example 3, and SPAES/HCNT 1.5 at different temperatures under the relative humidity of 100%.
FIG. 10f is a graph showing the proton conductivity change of Nafion-115, SPAES/CNT 1.5 of example 3, and SPAES/HCNT 1.5 at different relative humidities at a temperature of 80 ℃.
FIG. 11a is a plot of the polarization and power density of PEMFCs integrating SPAES, SPAES/CNT (1.5 wt%), SPAES/HCNT (1.5 wt%) and Nafion-115 membranes at 60 ℃ and 100% relative humidity.
FIG. 11b is a graph of the polarization and power density of PEMFCs integrating SPAES, SPAES/CNT (1.5 wt%), SPAES/HCNT (1.5 wt%) and Nafion-115 membranes at 60 ℃ and 20% relative humidity.
FIG. 11c is a graph showing the durability test of PEMFCs integrating SPAES, SPAES/CNT (1.5 wt%), SPAES/HCNT (1.5 wt%) and Nafion-115 membranes under the conditions of a temperature of 60 ℃ and a relative humidity of 100%.
FIG. 11d is a graph showing the durability test of PEMFCs integrating SPAES, SPAES/CNT (1.5 wt%), SPAES/HCNT (1.5 wt%) and Nafion-115 membranes under the conditions of temperature of 60 ℃ and relative humidity of 20%.
Figure 12a is an SEM image of SPAES prior to MEA durability testing.
Figure 12b is an SEM image of SPAES after MEA durability testing.
FIG. 12c is an SEM image of SPAES/HCNT (1.5 wt%) prior to MEA durability testing.
FIG. 12d is an SEM image of SPAES/HCNT (1.5 wt%) after MEA durability testing.
Detailed Description
The technical solutions in the embodiments of the present invention will be clearly and completely described below with reference to the drawings in the embodiments of the present invention, and it is obvious that the described embodiments are only a part of the embodiments of the present invention, and not all of the embodiments. All other embodiments, which can be derived by a person skilled in the art from the embodiments given herein without making any creative effort, shall fall within the protection scope of the present invention.
The invention discloses a sulfonated polyarylethersulfone-doped composite material, which comprises Sulfonated Polyarylethersulfone (SPAES) and a modified nano carbon material doped in the sulfonated polyarylethersulfone, wherein the modified nano carbon material is a nano carbon material treated by a modifier, and the modifier is amino acid.
The nano carbon material is a carbon material with at least one dimension of a disperse phase dimension less than 100 nm. The dispersed phase may consist of carbon atoms, may also consist of heterogeneous atoms (non-carbon atoms), and may even be nanoporous.
In the present embodiment, the nanocarbon material includes at least one of Carbon Nanotubes (CNTs), carbon nanofibers, and nanocarbon spheres.
The nano carbon material has higher specific surface area and better mechanical property, and is suitable to be used as a doping material.
The sulfonated polyarylether sulfone-doped composite material comprises sulfonated polyarylether sulfone and a modified nanocarbon material doped in the sulfonated polyarylether sulfone, wherein when amino acid is used for modifying the nanocarbon material, the amino acid can be combined with carboxyl (-COOH) and other groups carried by the nanocarbon material, so that the surface of the obtained modified nanocarbon material is provided with additional ionic groups, the proton conductivity can be improved by improving the density of the ionic groups, and the mechanical integrity is improved by recombining a polymer chain, so that the sulfonated polyarylether sulfone-doped composite material has higher proton conductivity.
Preferably, in the sulfonated polyarylether sulfone-doped composite material, the mass ratio of the modified nano carbon material to the sulfonated polyarylether sulfone is 0.25-6: 100.
specifically, in the sulfonated polyarylethersulfone-doped composite material, the mass ratio of the modified nanocarbon material to the sulfonated polyarylethersulfone may be 0.5: 100. 1: 100. 1.5: 100. 2: 100. 3: 100 or 4: 100.
in this embodiment, the modifier is histidine. The nano carbon material is modified by histidine, and can be combined with carboxyl (-COOH) and other groups carried by the nano carbon material, and the modified nano carbon material can provide additional ionic groups when being soaked in the SPAES matrix, so that sequential acid-base pairs generated along the SPAES/HCNT interface can be used as proton conduction sites, and the SPAES/HCNT composite membrane has enhanced proton conductivity.
Particularly preferably, the modified nanocarbon material is histidine-modified carbon nanotube (HCNT).
The invention also discloses a preparation method of the doped sulfonated polyarylether sulfone composite material, which comprises the following steps:
s10, pretreating the nano carbon material, and modifying the pretreated nano carbon material by using an improver solution to obtain the modified nano carbon material.
Preferably, the pretreatment of the nanocarbon material is performed by: dispersing the nano carbon material into a mixed solution of N, N-Dimethylformamide (DMF) and thionyl chloride, carrying out heat treatment for 8-48 h at the temperature of 60-85 ℃, and then cleaning and drying to obtain the pretreated nano carbon material.
The purpose of the pretreatment is to wash away some impurity groups on the surface of the nano carbon material, and to activate the groups on the surface of the nano carbon material to improve the binding rate of the next surface modification.
Preferably, in the mixed solution of N, N-dimethylformamide and thionyl chloride, the volume ratio of N, N-dimethylformamide to thionyl chloride is 2.5 to 10: 100.
specifically, in the mixed solution of N, N-dimethylformamide and thionyl chloride, the volume ratio of N, N-dimethylformamide to thionyl chloride may be 3: 100. 4: 100. 5: 100. 6: 100. 7: 100. 8: 100 or 9: 100.
specifically, the temperature of the heat treatment may be 70 ℃, the time of the heat treatment may be 24 hours, the detergent for cleaning may be Tetrahydrofuran (THF), and the drying may be vacuum drying.
Preferably, the mass percentage concentration of the modifier solution is 0.5 g/L-4 g/L.
Specifically, the mass percentage concentration of the modifier solution is 1g/L, 1.5g/L, 2g/L, 2.5g/L, 3g/L or 3.5 g/L.
Preferably, the solid-to-liquid ratio of the nanocarbon material to the modifier solution is 0.1g to 4 g: 100 mL.
Specifically, the solid-to-liquid ratio of the nanocarbon material to the modifier solution was 0.2 g: 100mL, 0.3 g: 100mL, 0.4 g: 100mL, 0.5 g: 100mL, 1 g: 100mL, 2 g: 100mL, 2.5 g: 100mL, 3 g: 100mL or 3.5 g: 100 mL.
Modifying the pretreated nano carbon material by using an improver solution to obtain a modified nano carbon material, wherein the operation comprises the following steps: and soaking the pretreated nano carbon material in an improver solution for 10-80 min, heating the whole system to reflux and keeping for 24-96 h, and then cleaning and drying to obtain the modified nano carbon material.
Specifically, the heating reflux can be completed in a reflux condenser, the drying is vacuum drying, and the cleaning agent for cleaning is ethanol.
And S20, uniformly mixing the modified nano-carbon material obtained in the step S10 and the sulfonated polyarylether sulfone under a liquid phase condition, and drying to obtain the doped sulfonated polyarylether sulfone composite material.
Preferably, the solvent is dimethylacetamide (DMAc) in the operation of uniformly mixing the modified nanocarbon material and the sulfonated polyarylethersulfone under the liquid phase condition.
Specifically, the operation of uniformly mixing the modified nanocarbon material and the sulfonated polyarylethersulfone under the liquid phase condition may be: adding the modified nano carbon material into dimethyl acetamide, ultrasonically mixing uniformly, and adding sulfonated polyarylethersulfone to dissolve uniformly.
In the application, the sulfonated polyarylethersulfone can be directly purchased or can be prepared by self.
Referring to FIG. 1, the sulfonated polyarylethersulfone is prepared as follows: after preparing BP, DFDPS and SDFDPS monomers by polycondensation, K is added2CO3And an organic mixed solution (NMP/toluene-2/1 v/v) was added as an entrainer. The reaction mixture was refluxed at 150 ℃ for 5 hours to dehydrate the system, and after toluene removal, the reaction mixture was refluxed at 190 ℃ for polymerization. The viscous solution obtained after 48h of reaction was diluted with NMP and filtered to remove the salt. Precipitating in ethanol to obtain white powder, extracting with deionized water, and removing residual salts and low molecular weight oligomers. The product was dried in a vacuum oven at 80 ℃ for 24h and then 1.0M H was added2SO4(aq) stirring, adding-SO3Conversion of Na to-SO3And H, washing with distilled water for several times until residual sulfuric acid is removed. Finally drying for 24h at 80 ℃ under the vacuum condition to obtain the SPAES polymer.
The doped sulfonated polyarylethersulfone composite material has higher proton conductivity, and can be applied to the field of preparation of polymer electrolyte membranes.
The following are specific examples.
Experimental reagent: polyetheretherketone powder was purchased from Victrex, korea. Sulfuric acid (H2SO4, 95%) was purchased from Daejung Chemicals, korea. Multiwall CNTs (80% MWCNTs) and toluene in anhydrous solution (99.98%) were obtained from Sigma Aldrich, and 3-aminopropyl-triethoxysilane (99% APTES) was obtained from Acros organics. N, N-Dimethylformamide (DMF) was purchased from korea, chemical industry, sanchun.
Purification by recrystallization from toluene gave 4,4' -difluorodiphenyl sulfone (DFDPS, 99.0%, Aldrich). Sulfonation of DFDPS with 65.0% fuming sulfuric acid to produce 3,3 '-disulfonic acid-4, 4' -difluorodiphenyl sulfone (SDFDPS) according to HarrisonPurification was performed by the method of et al (yield 78%). 4,4' -dihydroxybiphenyl (BP, 97.0%, Aldrich) was purified by recrystallization from methanol. 3, 4-diaminobenzoic acid (DABA, 98.0%, TCI) was purified by recrystallization from deionized water. With potassium carbonate (K)2CO399.0%, aldrich), sodium nitrate (NaNO3, 99.0%, aldrich), potassium permanganate (KMnO)499.0%, Aldrich), phosphorus pentoxide (P)2O599.0%, Aldrich), sulfuric acid (H)2SO495.0%, Daejung), hydrochloric acid (HCl, 35.0%, Daejung), hydrogen peroxide (H)2O230.0%, Daejung) and poly (phosphoric acid) (PPA, 116.0%, Junsei) as starting materials. N-methyl-2-pyrrolidone (NMP), dimethylacetamide (DMAc), toluene, and ethanol were stored over molecular sieves prior to use.
Experimental equipment: the surface and cross-sectional properties of the prepared samples were investigated using field emission scanning electron microscopy (FE-SEM, ZEISS, Supra 40VP), x-ray Energy Dispersive Analysis (EDAX) and scanning electron microscopy (SEM, JEOL, JSM-6400). Sample preparation for this analysis was done by platinum sputtering. And (3) carrying out microstructure imaging on the prepared sample by adopting a transmission electron microscope (TEM, JEOL, JEM-2010) with an accelerating voltage of 80-200 kv. An atomic force microscope (AFM, Bruker, multilod-8) was used to capture topographical images and determine the surface roughness of the prepared samples. The functional groups and the crystalline properties of the prepared samples were investigated using Fourier transform infrared spectroscopy (FT-IR, PerkinElmer, Frontier) and X-ray diffraction (XRD, Panalytical, X' pert Pro Power). The ion cluster size of the prepared film was determined using small angle x-ray scattering method (SAXS, Empyrean, Panalyticl). Thermal properties such as thermal stability and glass transition temperature of the prepared film were evaluated by a thermogravimetric analyzer (TGA, TA instruments, Q400) and a differential scanning calorimeter (DSC, TA instruments, Q20). Tensile stress and strain of the film were tested using a universal tester.
Example 1
Referring to FIG. 2, 200mg of Carbon Nanotubes (CNT) were added to a 100mL two-necked round bottom flask of 3mL DMF, followed by 60mL of thionyl chloride. The reaction flask, equipped with reflux condenser, magnetic stirrer and thermometer, was then placed in a pre-heated oil bath. The whole system was heated at 70 ℃ for 24h, then the solid was washed several times with THF until the pH of the filtrate was 7.0.
The product was dried under vacuum for 4h, and then the dried solid, 75mg histidine and 50mL deionized water were mixed for 30 min. And (3) heating the reaction flask with a reflux condenser at 100 ℃ for 48h under reflux, filtering to retain a solid product, washing with ethanol, and drying under vacuum at 60 ℃ for 4h to obtain the histidine-modified carbon nanotube (HCNT).
To 1.133g of DMAc was added 0.001g of HCNT, and the mixture was sonicated for 30min to uniformly disperse the HCNT, followed by dissolving 0.200g of SPAES to obtain a mixed liquid.
And pouring the mixed liquid on a glass plate by using a scraper, heating the mixed liquid in an oven at 80 ℃ for 12 hours, then heating the mixed liquid in an oven at 120 ℃ for 2 hours to obtain a dry film, and drying the obtained dry film in a vacuum oven at 80 ℃ for 24 hours to obtain the needed SPAES/HCNT film.
The SPAES/HCNT membrane produced in example 1 was designated: SPAES/HCNT 0.5, representing a mass ratio of HCNT to SPAES of 0.5: 100.
example 2
300mg of Carbon Nanotubes (CNT) was added to a 100mL two-necked round bottom flask with 3mL of DMF, followed by 60mL of thionyl chloride. The reaction flask, equipped with reflux condenser, magnetic stirrer and thermometer, was then placed in a pre-heated oil bath. The whole system was heated at 70 ℃ for 24h, then the solid was washed several times with THF until the pH of the filtrate was 7.0.
The product was dried under vacuum for 4h, and then the dried solid, 75mg histidine and 50mL deionized water were mixed for 30 min. And (3) heating the reaction flask with a reflux condenser at 100 ℃ for 48h under reflux, filtering to retain a solid product, washing with ethanol, and drying under vacuum at 60 ℃ for 4h to obtain the histidine-modified carbon nanotube (HCNT).
To 1.133g of DMAc was added 0.002g of HCNT, and the mixture was sonicated for 30min to uniformly disperse the HCNT, followed by dissolving 0.200g of SPAES to obtain a mixed liquid.
And pouring the mixed liquid on a glass plate by using a scraper, heating the mixed liquid in an oven at 80 ℃ for 12 hours, then heating the mixed liquid in an oven at 120 ℃ for 2 hours to obtain a dry film, and drying the obtained dry film in a vacuum oven at 80 ℃ for 24 hours to obtain the needed SPAES/HCNT film.
The SPAES/HCNT membrane prepared in example 2 was named: SPAES/HCNT1, representing the mass ratio of HCNT to SPAES of 1: 100.
example 3
200mg of Carbon Nanotubes (CNT) were added to a 100mL two-necked round bottom flask with 3mL of DMF, followed by 60mL of thionyl chloride. The reaction flask, equipped with reflux condenser, magnetic stirrer and thermometer, was then placed in a pre-heated oil bath. The whole system was heated at 70 ℃ for 24h, then the solid was washed several times with THF until the pH of the filtrate was 7.0.
The product was dried under vacuum for 4h, and then the dried solid, 75mg histidine and 50mL deionized water were mixed for 30 min. And (3) heating the reaction flask with a reflux condenser at 100 ℃ for 48h under reflux, filtering to retain a solid product, washing with ethanol, and drying under vacuum at 60 ℃ for 4h to obtain the histidine-modified carbon nanotube (HCNT).
To 1.133g of DMAc was added 0.003g of HCNT, and the mixture was sonicated for 30min to uniformly disperse the HCNT, followed by dissolving 0.200g of SPAES to obtain a mixed liquid.
And pouring the mixed liquid on a glass plate by using a scraper, heating the mixed liquid in an oven at 80 ℃ for 12 hours, then heating the mixed liquid in an oven at 120 ℃ for 2 hours to obtain a dry film, and drying the obtained dry film in a vacuum oven at 80 ℃ for 24 hours to obtain the needed SPAES/HCNT film.
The SPAES/HCNT membrane prepared in example 3 was named: SPAES/HCNT 1.5, representing a mass ratio of HCNT to SPAES of 1.5: 100.
0.200g of SPAES was dissolved in 1.133g of DMAc to obtain a mixed liquid, the mixed liquid was poured onto a glass plate with a spatula, heated in an oven at 80 ℃ for 12 hours, then heated in an oven at 120 ℃ for 2 hours to obtain a dry film, and the obtained dry film was dried in a vacuum oven at 80 ℃ for 24 hours to obtain a SPAES film.
To 1.133g of DMAc was added 0.003g of CNT, and the mixture was sonicated for 30min to uniformly disperse the CNT, followed by dissolving 0.200g of SPAES to obtain a mixed liquid. And pouring the mixed liquid on a glass plate by using a scraper, heating the mixed liquid in an oven at 80 ℃ for 12 hours, then heating the mixed liquid in an oven at 120 ℃ for 2 hours to obtain a dry film, and drying the obtained dry film in a vacuum oven at 80 ℃ for 24 hours to obtain the SPAES/CNT film.
Example 4
200mg of Carbon Nanotubes (CNT) were added to a 100mL two-necked round bottom flask with 3mL of DMF, followed by 60mL of thionyl chloride. The reaction flask, equipped with reflux condenser, magnetic stirrer and thermometer, was then placed in a pre-heated oil bath. The whole system was heated at 70 ℃ for 24h, then the solid was washed several times with THF until the pH of the filtrate was 7.0.
The product was dried under vacuum for 4h, and then the dried solid, 75mg histidine and 50mL deionized water were mixed for 30 min. And (3) heating the reaction flask with a reflux condenser at 100 ℃ for 48h under reflux, filtering to retain a solid product, washing with ethanol, and drying under vacuum at 60 ℃ for 4h to obtain the histidine-modified carbon nanotube (HCNT).
To 1.133g of DMAc was added 0.004g of HCNT, and the mixture was sonicated for 30min to uniformly disperse the HCNT, followed by dissolving 0.200g of SPAES to obtain a mixed liquid.
And pouring the mixed liquid on a glass plate by using a scraper, heating the mixed liquid in an oven at 80 ℃ for 12 hours, then heating the mixed liquid in an oven at 120 ℃ for 2 hours to obtain a dry film, and drying the obtained dry film in a vacuum oven at 80 ℃ for 24 hours to obtain the needed SPAES/HCNT film.
The SPAES/HCNT membrane prepared in example 4 was named: SPAES/HCNT 2, representing the mass ratio of HCNT to SPAES of 2: 100.
example 5
200mg of Carbon Nanotubes (CNT) were added to a 100mL two-necked round bottom flask with 3mL of DMF, followed by 60mL of thionyl chloride. The reaction flask, equipped with reflux condenser, magnetic stirrer and thermometer, was then placed in a pre-heated oil bath. The whole system was heated at 70 ℃ for 24h, then the solid was washed several times with THF until the pH of the filtrate was 7.0.
The product was dried under vacuum for 4h, and then the dried solid, 75mg histidine and 50mL deionized water were mixed for 30 min. And (3) heating the reaction flask with a reflux condenser at 100 ℃ for 48h under reflux, filtering to retain a solid product, washing with ethanol, and drying under vacuum at 60 ℃ for 4h to obtain the histidine-modified carbon nanotube (HCNT).
To 1.133g of DMAc was added 0.008g of HCNT, and the mixture was sonicated for 30min to uniformly disperse the HCNT, followed by dissolving 0.200g of SPAES to obtain a mixed liquid.
And pouring the mixed liquid on a glass plate by using a scraper, heating the mixed liquid in an oven at 80 ℃ for 12 hours, then heating the mixed liquid in an oven at 120 ℃ for 2 hours to obtain a dry film, and drying the obtained dry film in a vacuum oven at 80 ℃ for 24 hours to obtain the needed SPAES/HCNT film.
The SPAES/HCNT membrane prepared in example 5 was named: SPAES/HCNT 4, representing the mass ratio of HCNT to SPAES of 4: 100.
example 6
200mg of Carbon Nanotubes (CNT) were added to a 100mL two-necked round bottom flask with 3mL of DMF, followed by 60mL of thionyl chloride. The reaction flask, equipped with reflux condenser, magnetic stirrer and thermometer, was then placed in a pre-heated oil bath. The whole system was heated at 70 ℃ for 24h, then the solid was washed several times with THF until the pH of the filtrate was 7.0.
The product was dried under vacuum for 4h, and then the dried solid, 75mg histidine and 50mL deionized water were mixed for 30 min. And (3) heating the reaction flask with a reflux condenser at 100 ℃ for 48h under reflux, filtering to retain a solid product, washing with ethanol, and drying under vacuum at 60 ℃ for 4h to obtain the histidine-modified carbon nanotube (HCNT).
To 1.133g of DMAc was added 0.012g of HCNT, and the mixture was sonicated for 30min to uniformly disperse the HCNT, followed by dissolving 0.200g of SPAES to obtain a mixed liquid.
And pouring the mixed liquid on a glass plate by using a scraper, heating the mixed liquid in an oven at 80 ℃ for 12 hours, then heating the mixed liquid in an oven at 120 ℃ for 2 hours to obtain a dry film, and drying the obtained dry film in a vacuum oven at 80 ℃ for 24 hours to obtain the needed SPAES/HCNT film.
The SPAES/HCNT membrane prepared in example 6 was named: SPAES/HCNT 6, representing the mass ratio of HCNT to SPAES of 6: 100.
example 7
200mg of Carbon Nanotubes (CNT) were added to a 100mL two-necked round bottom flask with 3mL of DMF, followed by 60mL of thionyl chloride. The reaction flask, equipped with reflux condenser, magnetic stirrer and thermometer, was then placed in a pre-heated oil bath. The whole system was heated at 70 ℃ for 24h, then the solid was washed several times with THF until the pH of the filtrate was 7.0.
The product was dried under vacuum for 4h, and then the dried solid, 75mg histidine and 50mL deionized water were mixed for 30 min. And (3) heating the reaction flask with a reflux condenser at 100 ℃ for 48h under reflux, filtering to retain a solid product, washing with ethanol, and drying under vacuum at 60 ℃ for 4h to obtain the histidine-modified carbon nanotube (HCNT).
To 1.133g of DMAc was added 0.0005g of HCNT, and the mixture was subjected to ultrasonic treatment for 30min to uniformly disperse the HCNT, followed by dissolving 0.200g of SPAES to obtain a mixed liquid.
And pouring the mixed liquid on a glass plate by using a scraper, heating the mixed liquid in an oven at 80 ℃ for 12 hours, then heating the mixed liquid in an oven at 120 ℃ for 2 hours to obtain a dry film, and drying the obtained dry film in a vacuum oven at 80 ℃ for 24 hours to obtain the needed SPAES/HCNT film.
The SPAES/HCNT membrane prepared in example 7 was named: SPAES/HCNT 0.25, representing a mass ratio of HCNT to SPAES of 0.25: 100.
test example
1) Description of the test methods
Measurement of Water absorption expansion ratio
The water absorption and expansion rates were determined by measuring the length, area and volume of the dry and wet films.
Before measurement, the film samples were oven dried at 60 ℃ for 12h to give dry films. Weight (W) of dry filmdryG), length (L)dryCm), area (A)dry,cm2) Volume (V)dry,cm3) The measurement is performed.
Then, the film sample was soaked in deionized water at 60 ℃ for 24 hours to obtain a wet film. Weight to wet film (W)wetG), length (L)wetCm), area (A)wet,cm2) Volume (V)wet,cm3) Carry out the measurementAmount of the compound (A).
Finally, the water absorption and expansion of the film were calculated using the formulas (1) to (4). Measurements were performed in triplicate to ensure reproducibility of the calculated values.
Ion exchange capacity measurement
The ion exchange capacity of the membrane was calculated using acid-base titration. The weight (W) of the dry film was measureddG), then the sample is immersed in a 2M NaCl solution, using Na in the solution+Ion release of H from membranes+Ions.
Preparing 0.01M NaOH solution, titrating released H by using phenolphthalein as indicator+Ions. The consumption volume (V) of NaOH solution during the titration was subsequently determinedNaOHmL). Repeating the measurement several times, averaging to obtain average result as shown in formula (5), and calculating IEC value
Proton conductivity measurement
The resistance (R, Ω) of the membrane was measured by replacing the current impedance spectroscopy by using a four-probe cell (Bekk-Tech) into which an appropriate partial sample (3cm 0.5cm) of the membrane was loaded longitudinally.
Temperature, resistance and proton conductivity (σ, ms · cm)-1) Calculated according to equation (6), where L is the distance between the four probes, T is the film thickness, and W is the film sample width.
Preparation of Membrane Electrode (MEA) and evaluation of Single cell Performance and durability
The membrane electrode was fabricated by hot pressing the membrane with a commercial gas diffusion electrode (GDE, 0.3 mg/cm Pt loading) on both sides at 70 deg.C and 80bar for 2 minThe hot pressed film of the bell is prepared. The prepared single cell has a length of 5cm2The single cell active area Horizon test system of (a).
The polarization and power density of the membrane electrode were measured at 20% RH (relative humidity) and 100% RH (relative humidity) at 60 ℃ and H2And O2The flow rates of (A) were maintained at 100mL/min and 250mL/min, respectively, without applying any back pressure. The relative humidity of the cell was varied by modifying the preheater and the anode/cathode saturation temperature.
The durability of the membrane electrode was also tested by monitoring the Open Circuit Voltage (OCV) of the membrane electrode at 60 ℃, 20 ℃ and 100% RH for 50 h.
2) Morphological characteristics of CNT and HCNT
The CNTs and HCNT in example 1 and example 2 were observed by field emission scanning electron microscopy (FE-SEM), respectively, to yield fig. 3a, fig. 3b, fig. 3c, and fig. 3d, respectively.
The CNTs and HCNT in example 1 were observed with a Scanning Electron Microscope (SEM) to obtain fig. 3e and fig. 3f, respectively.
FE-SEM and TEM morphologies of CNTs and HCNT showed significant entanglement, linear structure of CNTs, which was retained even after histidine functionalization.
As can be seen from fig. 3a, 3c and 3e, the CNTs are held together due to van der waals interactions between the individual tubes.
As can be seen from FIGS. 3b, 3d and 3f, the functionalization of histidine is due to the presence of-NH-in the histidine2The electrostatic repulsion of the radicals makes the distance between the individual tubes longer.
In addition, referring to fig. 3a to 3f, the dispersion diameter between the HCNT single tubes was slightly higher than that of the CNTs.
3) Morphological characteristics of SPAES and SPAES/HCNT
SPAES and SPAES/HCNT 1.5 from example 3 were observed by Scanning Electron Microscopy (SEM) to give FIGS. 4a and 4b, respectively.
SPAES and SPAES/HCNT 1.5 from example 3 were observed using an x-ray energy dispersive spectrometer to give FIGS. 4c and 4d, respectively.
As can be seen from fig. 4a, the SPAES film surface is relatively dense with no significant cracks or pinholes.
As can be seen from the combination of FIGS. 4a and 4b, the surface of the SPAES/HCNT 1.5 membrane is due to the interaction (-NH)2and-SO3The hydrogen bonds between the H groups promote interfacial compatibility between the filler and the matrix. In this way, proton transport channels are formed with the polymer chains.
Comparing fig. 4c and fig. 4d, it can be seen that the EDAX spectra of SPAES/HCNT has an N peak compared to SPAES (fig. 4 c).
The cross sections of SPAES and SPAES/HCNT 1.5 in example 3 were observed by Scanning Electron Microscopy (SEM) to obtain FIGS. 5a, 5b, 5c, 5d, 5e and 5f, respectively.
As can be seen from fig. 5a, 5b, 5c, 5d, 5e and 5f, the cross-sections of the SPAES and SPAES/HCNT membranes are morphologically similar, and the cross-sectional morphology of both membranes is dense, uniform and defect free. This indicates that the doping of HCNT does not cause any significant change in cross section. This phenomenon is probably due to the low mass proportion of HCNT in the SPAES matrix.
The surfaces of SPAES and SPAES/HCNT 1.5 in example 3 were observed by Atomic Force Microscopy (AFM) to obtain FIG. 6.
As can be seen from fig. 6, with HCNT randomly distributed in SPAES, the presence of protrusions and sinks on the composite surface clearly reflects the incorporation of dopants in the matrix. In addition, the Roughness (RMS) of SPAES/HCNT 1.5 was 21.2nm, which was higher than SPAES (12.6 nm).
4) Structural Properties of CNT and ACNT
The CNTs and HCNT of example 1 were characterized by fourier transform infrared spectroscopy to give figure 7.
As can be seen from FIG. 7, it appears at 1700cm in both spectra-1The peak of (a) refers to the surface carboxylic acid groups in the histidine side chains in Im-CNTs and in CNTs. 1633cm in the imc-CNT spectrum-1The main absorption band indicates that amide bond is formed after the CNT is functionalized with imidazole group. The imidazole groups were successfully grafted onto the CNTs. About 1573cm on the imc-cnt spectrum-1The peak at (A) is due to the absorbance of the N-H group.
5) Thermal performance of CNT, HCNT, SPAES and SPAES/HCNT
Thermodynamic measurements were performed on CNT, HCNT, and SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2, and SPAES/HCNT 1.5 of example 3, respectively, to obtain FIG. 8 a.
Thermodynamic measurements were performed on SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2, and SPAES/HCNT 1.5 of example 3, respectively, to obtain FIGS. 8b, 8c, and 8 d.
As is apparent from fig. 8a, the thermal stability of the CNTs was higher than that of HCNT, and the carbon nanotubes showed only one weight reduction due to the elimination of moisture on the surface of the carbon nanotubes. In contrast, three weight reductions of HCNT occurred: (1) splitting free and bound water molecules below 150 ℃; (2) elimination of unstable carboxyl groups between 150 ℃ and 300 ℃; (3) a third weight loss of HCNT indicated successful binding of histidine to HCNT.
As is evident from the combination of fig. 8a, 8b, 8c and 8d, three weight reductions were observed for the films: (1) loss of bound and free water molecules at 50-160 ℃; (2) SO functionalized on SPAES aromatic chains at 200-3(ii) loss of H groups, and (iii) disintegration of the SPAES backbone above 500 ℃.
In particular, the TGA profile of the SPAES/HCNT composite material exhibits a slight delay drop compared to that of SPAES, probably due to-COOH and-NH of HCNT2Radical with-SO of SPAES3Electrostatic interactions between H groups.
In conjunction with fig. 8c, DSC further analyzed the effect of HCNT incorporation on SPAES chain motion, which results from structural relaxation of SPAES at high temperatures, leading to the generation of endothermic peaks. Endothermic peaks were observed for all films in the temperature range of 93.36-119.6 ℃. The atomic emission spectrum has a glass transition temperature (Td) of about 93.36 ℃, and the atomic emission spectrum has a high thermal stability, so that the proportion of carbon nanotubes injected into the matrix increases. The glass transition temperatures (Td) of SPAES/HCNT (0.5 wt%), SPAES/HCNT (1 wt%) and SPAES/HCNT (1.5 wt%) were 95.24, 119.64 and 119.69C, respectively.
In conjunction with fig. 8d, the SPAES/HCNT composite membrane also showed a higher Td than the SPAES membrane in the case of the DSC second scan. This trend can be attributed to the strong interaction of acid-base pairs at the guest-host interface. Thus, the mobility of the SPAES chains is tightly controlled, so that more heat is required to obtain sufficient chain mobility for the phase change.
Overall, the DSC results show that the addition of HCNT effectively improves the thermal stability of the membrane, which is consistent with the TGA results.
6) Mechanical properties of SPAES and SPAES/HCNT
The limitation of chain motion also affects the mechanical integrity of the membrane, which is reflected in the stress-strain curve.
As can be seen from fig. 8b, all films exhibited characteristic regions of deformation on the stress-strain curve. Before the breaking point, the stress increases rapidly with increasing elongation, and the initial slope of the elastic modulus is larger. The bare SPAES film had acceptable mechanical integrity, tensile strength of 57.1MPa, Young's modulus of 1002MPa, and strain of 4.2%. In comparison, the SPAES/HCNT composite membrane has better mechanical stability. For example, with the addition of 0.5, 1 and 1.5 wt% ACNT, the tensile strength increased to 57.7, 59.0 and 67.5MPa, respectively, and the strain was 4.3, 4.6 and 4.6%, respectively. The higher mechanical integrity may be attributed to strong hydrogen bonding between SPAES and HCNT, indicating that an acid-base pair is formed within the SPAES/HCNT composite membrane.
7) Contact angles of SPAES and SPAES/HCNT, adsorption/desorption of water, water absorption, swelling ratio, IEC and proton conductivity
The water contact angles of SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2, and SPAES/HCNT 1.5 of example 3 were measured at room temperature, respectively, and the surface properties of the film were examined to obtain FIG. 9.
As can be seen from FIG. 9, the SPAES/HCNT (1.5 wt%) membrane is most hydrophilic with a contact angle of 77.6. The contact angles of SPAES/HCNT (1 wt%), SPAES/HCNT (0.5 wt%) and SPAES were 82.1, 84.8 and 87.6, respectively. The decrease in contact angle with increasing HCNT content is due to the addition of more hydrophilic groups to the composite membrane. The adsorption/desorption of water is closely related to the proton conductivity of the membrane. The more ionic groups, the stronger the water adsorption capacity.
The weight of SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2, and SPAES/HCNT 1.5 of example 3 were measured at different Relative Humidities (RH), respectively, and the water absorption of the film was examined to obtain FIGS. 10a and 10 b.
As can be seen from a combination of fig. 10a and 10b, an increase in relative humidity leads to an increase in the amount of water adsorbed and a decrease in relative humidity leads to desorption. Of the membranes studied, SPAES/HCNT (1.5 wt%) exhibited the highest water absorption of 14.5% and also 17.5%.
Table 1 below shows the water absorption, expansion and IEC results obtained from the tests carried out on SPAES, SPAES/HCNT 0.5, SPAES/HCNT-1.0, SPAES/HCNT-1.5 and SPAES/HCNT-2.0, respectively (the methods of testing are as described above).
Table 1: water absorption, expansion and IEC results for SPAES and varying amounts of SPAES/HCNT
| Water absorption rate | Swelling length (S)L) | Swelling area (S)A) | Swelling volume (S)V) | IEC | |
| SPAES | 12.8% | 4.4cm | 9.4cm2 | 12.2cm3 | 1.30 |
| SPAES/HCNT-0.5 | 14.5% | 5.2cm | 11.1cm2 | 14.7cm3 | 1.28 |
| SPAES/HCNT-1.0 | 15.2% | 5.6cm | 12.0cm2 | 16.8cm3 | 1.22 |
| SPAES/HCNT-1.5 | 17.5% | 6.8cm | 14.0cm2 | 22.0cm3 | 1.18 |
| SPAES/HCNT-2.0 | 16.0% | 5.9cm | 12.5cm2 | 18.2cm3 | 1.13 |
As can be seen from a combination of FIG. 10a, FIG. 10b and Table 1, the water adsorption amounts of SPAES, SPAES/HCNT (0.5 wt%) and SPAES/HCNT (1 wt%) were 10.2, 12.1 and 12.7%, respectively, and the water absorptions of SPAES, SPAES/HCNT (0.5 wt%) and SPAES/HCNT (1 wt%) were 12.8, 14.5 and 15.2%, respectively. This tendency is attributed to various interactions (-NH) with water molecules present in the composite membrane2and-SO3H) Further promoting the adsorption of water molecules. In general, a modest degree of swelling will result in more of a successively larger steric proton transfer through the membrane.
As shown in table 1, the expansion ratio of the film showed a tendency similar to the adsorption of water molecules. On this basis, the swelling ratios (S) of SPAES, SPAES/HCNT (0.5 wt%), SPAES/HCNT (1 wt%) and SPAES/HCNT (1.5 wt%) were foundL、SAAnd SV) Respectively (4.4, 9.4 and 12.2), (5.2, 11.1 and 14.7), (5.6, 12.0 and 16.8) and (6.8, 14.0 and 22.0). That is, the density of ionizable hydrophilic groups in the membrane significantly affects proton conductivity through grothuss and the carrier mechanism.
The IEC values of the above membranes are given in table 1. As can be seen from Table 1, the IEC values of SPAES/HCNT membranes were lower than those of SPAES membranes, regardless of the HCNT content. However, the IEC value of HCNT was high after modification of CNTs with histidine groups. However, the obtained data showed the opposite effect. The reason is that impregnation of HCNT in SPAES matrices leads to-SO3Dilution of the H group, thereby reducing the IEC value.
Proton conductivities of SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2, and SPAES/HCNT 1.5 of example 3 were measured at different temperatures, respectively, at RH of 100%, to obtain FIG. 10 c.
The proton conductivities of SPAES/HCNT 0.5 of example 1, SPAES/HCNT1 of example 2 and SPAES/HCNT 1.5 of example 3 were measured at different relative humidities at a temperature of 80 ℃ respectively to obtain FIG. 10 d.
As can be seen from FIG. 10c, the proton conductivity of the SPAES membrane at 90 ℃ was 79.5mS/cm, which is sufficient for PEMFC applications, while the HCNT-filled compositeThe proton conductivity of the composite membrane is 94.1, 109.2, 153.4 and 128.3mS/cm respectively. The smaller channel size and expansion ratio inhibits the diffusion of hydrogen ions over the composite membrane. The increase in proton conductivity of the SPAES/HCNT composite membrane may be due to enhanced transport of Grotthus-type protons. Theoretically, the lower the IEC value of the composite membrane, the lower the proton conductivity. However, in this study, despite the decrease in IEC values, the proton conductivity of the composite material still increased relative to that of the bare membrane. The acid-base pair provides a new way for enhancing the proton conductivity, thereby improving the proton conductivity of the composite film. Hydrogen bonding at the polymer-filler interface can affect the proton conduction pathway of SPAES, thereby inducing ion channels to align along the HCNT surface. However, the sample having the highest HCNT content showed a decrease in proton conductivity as compared with the sample having 1.5 wt%. SO in the Polymer chain in a 2 wt% sample of HCNT3H group masking and HCNT aggregation must occur in 2 wt% of the HCNT sample. Thus, in this work, it was found that an optimum HCNT content of 1.5 wt% could result in peak conductivity.
As can be seen from fig. 10d, the proton conductivity of the membrane was also measured at different relative humidities to examine the applicability of the membrane to low relative humidity PEFC. The proton conductivity of the membrane increases with increasing relative humidity because the increase in water potential causes more external water molecules to attach to the ionic groups of the membrane, resulting in an increased water adsorption capacity of the membrane to the PEM. Research shows that the proton conductivity of the SPAES/HCNT composite membrane is higher than that of a bare membrane under all RH values. The proton conductivity of the composite membrane increases with increasing ACNT content until reaching a peak at 1.5 wt% HCNT loading. As previously described, the decrease in conductivity with increasing HCNT content to 2 wt.% is due to the aggregation of the filler. The proton conductivity of the SPAES membrane was 75.1 and 2.8mS/cm at 100 and 20% RH, respectively. It is notable that the conductivity of SPAES/HCNT (1.5 wt%) is about 2.6 times better than that of SPAES at 20% relative humidity. SO in SPAES/HCNT at 20% relative humidity3Water molecules in the H groups are desorbed, resulting in reduced proton conductivity and reduced channel entrainment rate, due toThis does not allow a regular conductive network and the conductivity values are reduced. In contrast, the SPAES/HCNT composite membrane has higher water-holding capacity due to stronger water absorption of the HCNT surface, and is beneficial to proton conductivity. To determine-NH2The effect of functionalization on HCNT and the degree of fusion of HCNT in the polymer matrix, the proton conductivity (1.5 wt%) of SPAES/HCNT was compared to the properties and RH range.
Proton conductivities of Nafion-115 (purchased) at different temperatures, SPAES/CNT 1.5 of example 3 and SPAES/HCNT 1.5 were measured at RH of 100% respectively to obtain FIG. 10 e.
Proton conductivities of Nafion-115 (commercially available), SPAES/CNT 1.5 of example 3 and SPAES/HCNT 1.5 were measured at 80 ℃ at different relative humidities, respectively, yielding FIG. 10 f.
As can be seen from a combination of FIGS. 10e and 10f, the proton conductivity of the optimized SPAES/HCNT (1.5 wt%) membrane was comparable to that of Nafion-115.
8) Performance and durability testing of PEMFCs
FIG. 11a and FIG. 11b were obtained by studying the polarization and power density curves of PEMFCs integrating SPAES, SPAES/CNT (1.5 wt%), SPAES/HCNT (1.5 wt%) and Nafion-115 membranes.
As can be seen from fig. 11a, since the electronic resistances of the SPAES/CNT (1.5 wt%) and the SPAES are similar, the open circuit voltage of the PEMFC integrated with the SPAES/CNT (1.5 wt%) is almost the same as that of the PEMFC integrated with the SPAES. The peak power output under the load current output is 96.02mW/cm2When the PEMFC is used, the integration of the SPAES can reach 254.3mA cm2The current of (2). Under similar operating conditions, SPAES/CNT integrated, SPAES/HCNT and Nafion-115 provided 83.8, 140.3 and 114.6mW/cm at 109.8, 313.7 and 174mA/cm22Peak power output of. This result is consistent with the higher water absorption, proton conductivity, and similar OCV of the SPAES/HCNT membrane compared to the SPEEK membrane. The high proton conductivity can reduce the ohmic resistance to the maximum extent, and the higher water absorption can reduce the energy loss caused by the ionic resistance in SPAES/HCNT, thereby improving the performance of the PEMFC.
As can be seen from 11b, the PEMFC with integration of SPAES, SPAES/CNT, SPAES/HCNT and Nafion-115 has a load current density of 53.1, 63.8, 101.2 and 40.4mA/cm respectively at 60 ℃ and low RH (20%)2Respectively, to produce peak power densities of 22.9, 19.8, 51.1 and 15.4mW/cm 2. A factor that determines PEMFC performance at low RH (-20%) is back diffusion of water. Because of the low water content of SPAES, SPAES/CNT and Nafion-115, the back diffusion of water from the anode to the cathode must be reduced, which can result in dehydration of the MEA. MEA dehydration leads to an increase in the ohmic resistance of the PEMFC, resulting in a decrease in the performance of the PEMFC. In contrast, SPAES/HCNT (1.5 wt%) was able to accommodate additional water molecules and helped to keep the composite membrane wet enough to promote water diffusion back, improving PEMFC performance even at low RH.
Thus, SPAES/HCNT (1.5%) is an effective membrane that can provide reasonable PEMFC performance.
The above experiments demonstrate that SPAES/HCNT (1.5%) is an effective polymer electrolyte membrane, achieving reasonable PEMFC performance even at low RH.
FIG. 11c and FIG. 11d were obtained by performing durability tests on PEMFCs equipped with SPAES, SPAES/CNT (1.5 wt%) SPAES/HCNT (1.5 wt%) and Nafion-115 membranes.
As can be seen from FIG. 11c, the SPAES, SPAES/CNT and SPAES/HCNT integrated PEMFCs exhibited OCV attenuations of 0.67V, 0.32V and 0.02V at 60 ℃ at 100% RH and decay rates of 13.4, 6 and 0.4mV/h, respectively.
The OCV is accelerated to degrade, and free radicals such as hydroxyl (OH) and Hydroperoxy (HOO) in the membrane have strong effects on SPAES.
As can be seen from fig. 11d, at 20% RH all films showed delayed degradation due to insufficient generation of free radicals.
In both cases, the voltage holding performance of the PEMFC with SPAES/HCNT is better than that of the PEMFC with bare SPAES or SPAES/CNT. The higher stability of SPAES/HCNT is attributed to the compatibilization of HCNT with SPAES domains by-NH of HCNT and SPAES between the backbone and HCNT2Radical and-SO of SPAES3Strong interactions between H groups.
9) Morphology before and after MEAS durability test
The effects of chemical and mechanical degradation during the durability test were quantitatively analyzed by SEM analysis before and after the MEA durability test, resulting in fig. 12a, 12b, 12c, and 12 d.
With reference to fig. 12a, 12b, 12c and 12d, it can be seen that the description of the SPAES film and the SPAES/HCNT after the endurance test appears, and possible reasons include: (1) chemical degradation triggered by HO and HO radicals in the harsh PEMFC environment, (2) mechanical degradation due to physical causes compress both end plates of the cell.
However, the SPAES/HCNT composite membrane has a lower degradation rate and maintains its integrity compared to the SPAES membrane. This is due to the SO of SPAES3There is a strong acid-base interaction between the H group and the amine moiety of HCNT. The presence of the aforementioned interactions enhances the greater sustainability of the SPAES/HCNT composite membranes under mechanical compression and free radical attack in the PEMFC environment.
In summary, the present application addresses carbon nanotubes by containing NH2The functionalization of the organic portion of (a) and its subsequent impregnation into a SPAES matrix to make composite membranes was investigated. The SPAES/HCNT membrane has similar IEC, higher proton conductivity and higher water absorption compared to the SPAES membrane, which makes the SPAES/HCNT membrane a promising PEM for PEMFCs. The high performance of SPAES/HCNT membranes is mainly attributed to their specific, flexible structure consisting of a membrane containing oxidizing groups and-NH2Graphitic plane composition of radicals. In this structure, -NH2and-O3Acid-base interactions between H groups can form a localized proton conducting network in the membrane, thereby increasing water absorption and proton conductivity. The PEMFC with SPAES/HCNT exhibits great performance and durability when used as a PEM in PEMFCs operating at low RH, compared to PEMFCs used on SPEEK or Nafion-115. These results confirm that SPAES/HCNT is a high efficiency PEM that can achieve high performance and durable PEMFCs even at low RH.
The above-mentioned embodiments only express several embodiments of the present invention, and the description thereof is more specific and detailed, but not construed as limiting the claims. It should be noted that, for a person skilled in the art, several variations and modifications can be made without departing from the inventive concept, which falls within the scope of the present invention. Therefore, the protection scope of the present patent shall be subject to the appended claims.
Claims (1)
1. The preparation method of the doped sulfonated polyarylethersulfone composite material is characterized by comprising the following steps:
adding 200mg of carbon nanotubes to a 100mL two-necked round bottom flask with 3mL of DMF, followed by 60mL of thionyl chloride; then a reaction flask provided with a reflux condenser, a magnetic stirrer and a thermometer is arranged in a preheated oil bath; the whole system was heated at 70 ℃ for 24h, then the solid was washed several times with THF until the pH of the filtrate was 7.0;
drying the product for 4h under vacuum, and then mixing the dried solid, 75mg of histidine and 50mL of deionized water for 30 min; the reaction flask is equipped with a reflux condenser and is heated under reflux for 48 hours at 100 ℃, and finally a solid product is filtered and reserved, is cleaned by ethanol and is dried for 4 hours under vacuum of 60 ℃ to obtain the histidine modified carbon nano tube;
adding 0.003g of histidine-modified carbon nanotubes into 1.133g of DMAc, performing ultrasonic treatment for 30min to uniformly disperse the histidine-modified carbon nanotubes, and dissolving 0.200g of SPAES to obtain a mixed liquid;
and pouring the mixed liquid on a glass plate by using a scraper, heating the mixed liquid in an oven at 80 ℃ for 12 hours, then heating the mixed liquid in an oven at 120 ℃ for 2 hours to obtain a dry film, and drying the obtained dry film in a vacuum oven at 80 ℃ for 24 hours to obtain the needed SPAES/HCNT film.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010499350.2A CN111682247B (en) | 2020-06-04 | 2020-06-04 | Doped sulfonated polyarylethersulfone composite material and preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010499350.2A CN111682247B (en) | 2020-06-04 | 2020-06-04 | Doped sulfonated polyarylethersulfone composite material and preparation method and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111682247A CN111682247A (en) | 2020-09-18 |
| CN111682247B true CN111682247B (en) | 2021-03-19 |
Family
ID=72434864
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010499350.2A Active CN111682247B (en) | 2020-06-04 | 2020-06-04 | Doped sulfonated polyarylethersulfone composite material and preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111682247B (en) |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2883292A1 (en) * | 2005-03-16 | 2006-09-22 | Inst Nat Polytech Grenoble | EXTRUSION OF IONIC POLYMERS WITH ACIDIC IONIC GROUPS |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101979354B1 (en) * | 2011-12-01 | 2019-08-29 | 더 보오드 오브 트러스티스 오브 더 유니버시티 오브 일리노이즈 | Transient devices designed to undergo programmable transformations |
| CN105355930B (en) * | 2015-11-30 | 2017-11-10 | 湖北工程学院 | A kind of sulfonated aromatic polymers modified carbon nano-tube compound proton exchange membrane and preparation method thereof |
| US20190301814A1 (en) * | 2018-04-03 | 2019-10-03 | Nanotek Instruments, Inc. | Metallized graphene foam having high through-plane conductivity |
-
2020
- 2020-06-04 CN CN202010499350.2A patent/CN111682247B/en active Active
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2883292A1 (en) * | 2005-03-16 | 2006-09-22 | Inst Nat Polytech Grenoble | EXTRUSION OF IONIC POLYMERS WITH ACIDIC IONIC GROUPS |
Also Published As
| Publication number | Publication date |
|---|---|
| CN111682247A (en) | 2020-09-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Geng et al. | Symmetric sponge-like porous polybenzimidazole membrane for high temperature proton exchange membrane fuel cells | |
| Yusoff et al. | Sulfonated graphene oxide as an inorganic filler in promoting the properties of a polybenzimidazole membrane as a high temperature proton exchange membrane | |
| Sahin | The development of Speek/Pva/Teos blend membrane for proton exchange membrane fuel cells | |
| Gong et al. | A new strategy for designing high-performance sulfonated poly (ether ether ketone) polymer electrolyte membranes using inorganic proton conductor-functionalized carbon nanotubes | |
| Vijayalekshmi et al. | Eco-friendly methanesulfonic acid and sodium salt of dodecylbenzene sulfonic acid doped cross-linked chitosan based green polymer electrolyte membranes for fuel cell applications | |
| Wang et al. | A facile approach of fabricating proton exchange membranes by incorporating polydopamine-functionalized carbon nanotubes into chitosan | |
| Pu et al. | Novel branched sulfonated polyimide/molybdenum disulfide nanosheets composite membrane for vanadium redox flow battery application | |
| Nor et al. | Crosslinked carbon nanodots with highly sulfonated polyphenylsulfone as proton exchange membrane for fuel cell applications | |
| CN105529485B (en) | A kind of preparation method of carbon nanotube loaded heteropoly acid sulfonated poly-ether-ether-ketone (SPEEK) proton exchange membrane (PEM) | |
| Zhang et al. | Branched sulfonated polyimide/functionalized silicon carbide composite membranes with improved chemical stabilities and proton selectivities for vanadium redox flow battery application | |
| JP2013503436A (en) | POLYMER ELECTROLYTE MEMBRANE FOR FUEL CELL AND METHOD FOR PRODUCING THE SAME | |
| Yu et al. | Bifunctional effects of halloysite nanotubes in vanadium flow battery membrane | |
| Neelakandan et al. | Polypyrrole layered SPEES/TPA proton exchange membrane for direct methanol fuel cells | |
| CN105390721B (en) | A kind of preparation method of boron phosphate enveloped carbon nanometer tube compound proton exchange membrane | |
| Qian et al. | Improved proton conductivity and structure stability of an SPEEK/SPPS blend membrane for vanadium redox flow batteries | |
| Wang et al. | Simultaneously enhancing proton conductivity and mechanical stability of the membrane electrolytes by crosslinking of poly (aromatic ether sulfone) with octa-amino polyhedral oligomeric silsesquioxane | |
| Ravi Kumar et al. | Sandwich assembly of sulfonated poly (ether sulfone) with sulfonated multiwalled carbon nanotubes as an efficient architecture for enhanced electrolyte performance in H2/O2 fuel cells | |
| Tripathi et al. | Nanostructured membranes and electrodes with sulfonic acid functionalized carbon nanotubes | |
| Li et al. | Self-healing and highly elastic fluorine-free proton exchange membranes comprised of poly (vinyl alcohol) derivative and phytic acid for durable fuel cells | |
| Ahmed et al. | Tuning the performance of nanofiller reinforced phosphorylated chitosan-based proton exchange membrane | |
| Divya et al. | Highly selective custom‐made chitosan based membranes with reduced fuel permeability for direct methanol fuel cells | |
| CN109888348B (en) | Preparation method of fuel cell proton membrane material solid superacid/nitrogen heterocyclic graphene oxide/2,5-polybenzimidazole | |
| Karuppasamy et al. | Bio‐inspired proton conducting phytagel derived zwitterionic complex membranes for fuel cells | |
| CN111682247B (en) | Doped sulfonated polyarylethersulfone composite material and preparation method and application thereof | |
| He et al. | Hybrid network sulfonated polynorbornene/silica membranes with enhanced proton conductivity by doped phosphotungstic acid |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |