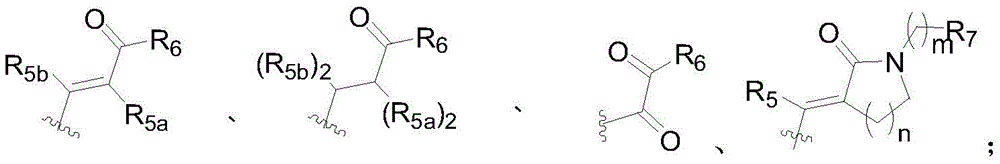CN111606904A - Azaindoles and their applications - Google Patents
Azaindoles and their applications Download PDFInfo
- Publication number
- CN111606904A CN111606904A CN202010265070.5A CN202010265070A CN111606904A CN 111606904 A CN111606904 A CN 111606904A CN 202010265070 A CN202010265070 A CN 202010265070A CN 111606904 A CN111606904 A CN 111606904A
- Authority
- CN
- China
- Prior art keywords
- alkyl
- pharmaceutically acceptable
- compound
- alkoxy
- stereoisomer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 125000005334 azaindolyl group Chemical group N1N=C(C2=CC=CC=C12)* 0.000 title claims abstract description 24
- 150000001875 compounds Chemical class 0.000 claims abstract description 81
- 206010028980 Neoplasm Diseases 0.000 claims abstract description 27
- 239000003814 drug Substances 0.000 claims abstract description 17
- 150000003839 salts Chemical class 0.000 claims description 55
- -1 azaindole compound Chemical class 0.000 claims description 42
- 229910052799 carbon Inorganic materials 0.000 claims description 33
- 229940002612 prodrug Drugs 0.000 claims description 24
- 239000000651 prodrug Substances 0.000 claims description 24
- 229910052739 hydrogen Inorganic materials 0.000 claims description 23
- 229910052736 halogen Inorganic materials 0.000 claims description 17
- 150000002367 halogens Chemical class 0.000 claims description 17
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 15
- 229910052757 nitrogen Inorganic materials 0.000 claims description 13
- 238000002360 preparation method Methods 0.000 claims description 11
- 125000006552 (C3-C8) cycloalkyl group Chemical group 0.000 claims description 10
- 125000003118 aryl group Chemical group 0.000 claims description 10
- 125000001072 heteroaryl group Chemical group 0.000 claims description 8
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims description 6
- 239000004480 active ingredient Substances 0.000 claims description 6
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 6
- 206010058467 Lung neoplasm malignant Diseases 0.000 claims description 4
- 201000005202 lung cancer Diseases 0.000 claims description 4
- 208000020816 lung neoplasm Diseases 0.000 claims description 4
- 201000001441 melanoma Diseases 0.000 claims description 4
- 239000008194 pharmaceutical composition Substances 0.000 claims description 4
- 206010006187 Breast cancer Diseases 0.000 claims description 3
- 208000026310 Breast neoplasm Diseases 0.000 claims description 3
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 claims description 3
- 201000007270 liver cancer Diseases 0.000 claims description 3
- 208000014018 liver neoplasm Diseases 0.000 claims description 3
- 125000004890 (C1-C6) alkylamino group Chemical group 0.000 claims description 2
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 claims description 2
- 125000001831 (C6-C10) heteroaryl group Chemical group 0.000 claims description 2
- 125000003277 amino group Chemical group 0.000 claims description 2
- 239000003937 drug carrier Substances 0.000 claims description 2
- QDKGOMZIPXGDDJ-UHFFFAOYSA-N 2,3-dihydro-1h-indazole Chemical compound C1=CC=C2CNNC2=C1 QDKGOMZIPXGDDJ-UHFFFAOYSA-N 0.000 claims 1
- 229940121957 SMAD3 inhibitor Drugs 0.000 claims 1
- 150000002475 indoles Chemical class 0.000 claims 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 abstract description 18
- 229940079593 drug Drugs 0.000 abstract description 15
- 101100256985 Arabidopsis thaliana SIS3 gene Proteins 0.000 abstract description 8
- CDKIEBFIMCSCBB-CALJPSDSSA-N SIS3 Chemical compound Cl.C1C=2C=C(OC)C(OC)=CC=2CCN1C(=O)\C=C\C(C1=CC=CN=C1N1C)=C1C1=CC=CC=C1 CDKIEBFIMCSCBB-CALJPSDSSA-N 0.000 abstract description 8
- 238000006366 phosphorylation reaction Methods 0.000 abstract description 7
- 230000000259 anti-tumor effect Effects 0.000 abstract description 5
- 238000005481 NMR spectroscopy Methods 0.000 description 62
- 238000006243 chemical reaction Methods 0.000 description 45
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 42
- 230000015572 biosynthetic process Effects 0.000 description 36
- 238000003786 synthesis reaction Methods 0.000 description 36
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 33
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 29
- 239000007787 solid Substances 0.000 description 29
- 239000000243 solution Substances 0.000 description 24
- 238000003756 stirring Methods 0.000 description 20
- VWVKUNOPTJGDOB-BDHVOXNPSA-N Anhydrous tofogliflozin Chemical compound C1=CC(CC)=CC=C1CC1=CC=C(CO[C@@]23[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O3)O)C2=C1 VWVKUNOPTJGDOB-BDHVOXNPSA-N 0.000 description 18
- 102000049939 Smad3 Human genes 0.000 description 17
- 238000005070 sampling Methods 0.000 description 15
- 238000010898 silica gel chromatography Methods 0.000 description 15
- 239000007821 HATU Substances 0.000 description 14
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical class Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 14
- 239000012141 concentrate Substances 0.000 description 14
- 101710143111 Mothers against decapentaplegic homolog 3 Proteins 0.000 description 13
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 12
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 12
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 12
- 239000005457 ice water Substances 0.000 description 12
- 239000000203 mixture Substances 0.000 description 12
- 239000000741 silica gel Substances 0.000 description 12
- 229910002027 silica gel Inorganic materials 0.000 description 12
- CDKIEBFIMCSCBB-UHFFFAOYSA-N 1-(6,7-dimethoxy-3,4-dihydro-1h-isoquinolin-2-yl)-3-(1-methyl-2-phenylpyrrolo[2,3-b]pyridin-3-yl)prop-2-en-1-one;hydrochloride Chemical compound Cl.C1C=2C=C(OC)C(OC)=CC=2CCN1C(=O)C=CC(C1=CC=CN=C1N1C)=C1C1=CC=CC=C1 CDKIEBFIMCSCBB-UHFFFAOYSA-N 0.000 description 11
- QOLHWXNSCZGWHK-BWBORTOCSA-N (6r,7r)-1-[(4s,5r)-4-acetyloxy-5-methyl-3-methylidene-6-phenylhexyl]-4,7-dihydroxy-6-(11-phenoxyundecylcarbamoyloxy)-2,8-dioxabicyclo[3.2.1]octane-3,4,5-tricarboxylic acid Chemical compound C([C@@H](C)[C@H](OC(C)=O)C(=C)CCC12[C@@H]([C@@H](OC(=O)NCCCCCCCCCCCOC=3C=CC=CC=3)C(O1)(C(O)=O)C(O)(C(O2)C(O)=O)C(O)=O)O)C1=CC=CC=C1 QOLHWXNSCZGWHK-BWBORTOCSA-N 0.000 description 10
- 229940126559 Compound 4e Drugs 0.000 description 10
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 10
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 10
- SHOWAGCIRTUYNA-UHFFFAOYSA-N 6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline;hydron;chloride Chemical compound [Cl-].C1C[NH2+]CC2=C1C=C(OC)C(OC)=C2 SHOWAGCIRTUYNA-UHFFFAOYSA-N 0.000 description 9
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 9
- 241000699670 Mus sp. Species 0.000 description 9
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 9
- 238000010790 dilution Methods 0.000 description 9
- 239000012895 dilution Substances 0.000 description 9
- 239000003112 inhibitor Substances 0.000 description 9
- 238000000967 suction filtration Methods 0.000 description 9
- CTSLXHKWHWQRSH-UHFFFAOYSA-N oxalyl chloride Chemical compound ClC(=O)C(Cl)=O CTSLXHKWHWQRSH-UHFFFAOYSA-N 0.000 description 8
- 108010050904 Interferons Proteins 0.000 description 7
- 102000014150 Interferons Human genes 0.000 description 7
- 102000004887 Transforming Growth Factor beta Human genes 0.000 description 7
- 108090001012 Transforming Growth Factor beta Proteins 0.000 description 7
- 210000004027 cell Anatomy 0.000 description 7
- 229940079322 interferon Drugs 0.000 description 7
- 238000000034 method Methods 0.000 description 7
- 210000000822 natural killer cell Anatomy 0.000 description 7
- 239000012074 organic phase Substances 0.000 description 7
- TZYWCYJVHRLUCT-VABKMULXSA-N N-benzyloxycarbonyl-L-leucyl-L-leucyl-L-leucinal Chemical compound CC(C)C[C@@H](C=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(C)C)NC(=O)OCC1=CC=CC=C1 TZYWCYJVHRLUCT-VABKMULXSA-N 0.000 description 6
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 6
- 102000057208 Smad2 Human genes 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- NKANXQFJJICGDU-QPLCGJKRSA-N Tamoxifen Chemical compound C=1C=CC=CC=1C(/CC)=C(C=1C=CC(OCCN(C)C)=CC=1)/C1=CC=CC=C1 NKANXQFJJICGDU-QPLCGJKRSA-N 0.000 description 6
- 239000002253 acid Substances 0.000 description 6
- 125000004432 carbon atom Chemical group C* 0.000 description 6
- 239000000460 chlorine Substances 0.000 description 6
- 230000002401 inhibitory effect Effects 0.000 description 6
- 229940121649 protein inhibitor Drugs 0.000 description 6
- 239000012268 protein inhibitor Substances 0.000 description 6
- 125000001424 substituent group Chemical group 0.000 description 6
- 238000012360 testing method Methods 0.000 description 6
- ZRKFYGHZFMAOKI-QMGMOQQFSA-N tgfbeta Chemical compound C([C@H](NC(=O)[C@H](C(C)C)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CC(C)C)NC(=O)CNC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CCSC)C(C)C)[C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(O)=O)C1=CC=C(O)C=C1 ZRKFYGHZFMAOKI-QMGMOQQFSA-N 0.000 description 6
- TWYYFYNJOJGNFP-CUXYNZQBSA-N (2s,4r,5s,6s)-2-[(4s,5r)-4-acetyloxy-5-methyl-3-methylidene-6-phenylhexyl]-2-carbamoyl-4-[[(e,4s,6s)-4,6-dimethyloct-2-enoyl]oxymethyl]-5-hydroxy-1,3-dioxane-4,5,6-tricarboxylic acid Chemical compound O1[C@H](C(O)=O)[C@](C(O)=O)(O)[C@](COC(=O)/C=C/[C@@H](C)C[C@@H](C)CC)(C(O)=O)O[C@]1(C(N)=O)CCC(=C)[C@@H](OC(C)=O)[C@H](C)CC1=CC=CC=C1 TWYYFYNJOJGNFP-CUXYNZQBSA-N 0.000 description 5
- 101710143123 Mothers against decapentaplegic homolog 2 Proteins 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 239000011259 mixed solution Substances 0.000 description 5
- VOXZDWNPVJITMN-ZBRFXRBCSA-N 17β-estradiol Chemical compound OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 VOXZDWNPVJITMN-ZBRFXRBCSA-N 0.000 description 4
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 4
- AOJJSUZBOXZQNB-TZSSRYMLSA-N Doxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-TZSSRYMLSA-N 0.000 description 4
- 108060001084 Luciferase Proteins 0.000 description 4
- 239000005089 Luciferase Substances 0.000 description 4
- ZNSPHKJFQDEABI-NZQKXSOJSA-N Nc1nc(O[C@H](c2ccc(Cl)cc2-c2ccccc2)C(F)(F)F)cc(n1)N1CCC2(CN[C@@H](C2)C(O)=O)CC1 Chemical compound Nc1nc(O[C@H](c2ccc(Cl)cc2-c2ccccc2)C(F)(F)F)cc(n1)N1CCC2(CN[C@@H](C2)C(O)=O)CC1 ZNSPHKJFQDEABI-NZQKXSOJSA-N 0.000 description 4
- 229930012538 Paclitaxel Natural products 0.000 description 4
- 108700031297 Smad3 Proteins 0.000 description 4
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 4
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 4
- 239000002585 base Substances 0.000 description 4
- 229940125876 compound 15a Drugs 0.000 description 4
- 239000012043 crude product Substances 0.000 description 4
- 238000001514 detection method Methods 0.000 description 4
- 238000001914 filtration Methods 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 4
- 231100000252 nontoxic Toxicity 0.000 description 4
- 230000003000 nontoxic effect Effects 0.000 description 4
- 229960001592 paclitaxel Drugs 0.000 description 4
- 230000026731 phosphorylation Effects 0.000 description 4
- 239000002994 raw material Substances 0.000 description 4
- 239000011734 sodium Substances 0.000 description 4
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 description 4
- ASQOQJYHIYYTEJ-GBESFXJTSA-N (1r,7s,9as)-7-decyl-2,3,4,6,7,8,9,9a-octahydro-1h-quinolizin-1-ol Chemical compound O[C@@H]1CCCN2C[C@@H](CCCCCCCCCC)CC[C@H]21 ASQOQJYHIYYTEJ-GBESFXJTSA-N 0.000 description 3
- FJZNNKJZHQFMCK-LRDDRELGSA-N 1-[(3S,4R)-4-(2,6-difluoro-4-methoxyphenyl)-2-oxopyrrolidin-3-yl]-3-phenylurea Chemical compound C1(=CC(=CC(=C1[C@H]1[C@@H](C(=O)NC1)NC(=O)NC1=CC=CC=C1)F)OC)F FJZNNKJZHQFMCK-LRDDRELGSA-N 0.000 description 3
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 3
- WCDLCPLAAKUJNY-UHFFFAOYSA-N 4-[4-[3-(1h-pyrazol-4-yl)pyrazolo[1,5-a]pyrimidin-6-yl]phenyl]morpholine Chemical compound C1COCCN1C1=CC=C(C2=CN3N=CC(=C3N=C2)C2=CNN=C2)C=C1 WCDLCPLAAKUJNY-UHFFFAOYSA-N 0.000 description 3
- 108010029961 Filgrastim Proteins 0.000 description 3
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 230000002378 acidificating effect Effects 0.000 description 3
- 230000004913 activation Effects 0.000 description 3
- 201000011510 cancer Diseases 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 125000000753 cycloalkyl group Chemical group 0.000 description 3
- 230000002708 enhancing effect Effects 0.000 description 3
- 229960005309 estradiol Drugs 0.000 description 3
- 125000005843 halogen group Chemical group 0.000 description 3
- 230000001965 increasing effect Effects 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 150000007529 inorganic bases Chemical class 0.000 description 3
- GFIJNRVAKGFPGQ-LIJARHBVSA-N leuprolide Chemical compound CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)CC1=CC=C(O)C=C1 GFIJNRVAKGFPGQ-LIJARHBVSA-N 0.000 description 3
- 229960004338 leuprorelin Drugs 0.000 description 3
- 229960003918 levothyroxine sodium Drugs 0.000 description 3
- YDTFRJLNMPSCFM-YDALLXLXSA-M levothyroxine sodium anhydrous Chemical compound [Na+].IC1=CC(C[C@H](N)C([O-])=O)=CC(I)=C1OC1=CC(I)=C(O)C(I)=C1 YDTFRJLNMPSCFM-YDALLXLXSA-M 0.000 description 3
- 150000007522 mineralic acids Chemical class 0.000 description 3
- 150000007524 organic acids Chemical class 0.000 description 3
- 108090000623 proteins and genes Proteins 0.000 description 3
- 239000012265 solid product Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 239000007858 starting material Substances 0.000 description 3
- 238000001308 synthesis method Methods 0.000 description 3
- 238000010189 synthetic method Methods 0.000 description 3
- 239000003826 tablet Substances 0.000 description 3
- 229960001603 tamoxifen Drugs 0.000 description 3
- FRACPXUHUTXLCX-BELIEFIBSA-N tert-butyl N-{1-[(1S)-1-{[(1R,2S)-1-(benzylcarbamoyl)-1-hydroxy-3-[(3S)-2-oxopyrrolidin-3-yl]propan-2-yl]carbamoyl}-2-cyclopropylethyl]-2-oxopyridin-3-yl}carbamate Chemical compound CC(C)(C)OC(=O)NC1=CC=CN(C1=O)[C@@H](CC2CC2)C(=O)N[C@@H](C[C@@H]3CCNC3=O)[C@H](C(=O)NCC4=CC=CC=C4)O FRACPXUHUTXLCX-BELIEFIBSA-N 0.000 description 3
- LLJFNWVJKMVHIL-UHFFFAOYSA-N (2-methoxy-2-oxoethyl)phosphonic acid Chemical compound COC(=O)CP(O)(O)=O LLJFNWVJKMVHIL-UHFFFAOYSA-N 0.000 description 2
- SHAHPWSYJFYMRX-GDLCADMTSA-N (2S)-2-(4-{[(1R,2S)-2-hydroxycyclopentyl]methyl}phenyl)propanoic acid Chemical compound C1=CC([C@@H](C(O)=O)C)=CC=C1C[C@@H]1[C@@H](O)CCC1 SHAHPWSYJFYMRX-GDLCADMTSA-N 0.000 description 2
- GHYOCDFICYLMRF-UTIIJYGPSA-N (2S,3R)-N-[(2S)-3-(cyclopenten-1-yl)-1-[(2R)-2-methyloxiran-2-yl]-1-oxopropan-2-yl]-3-hydroxy-3-(4-methoxyphenyl)-2-[[(2S)-2-[(2-morpholin-4-ylacetyl)amino]propanoyl]amino]propanamide Chemical compound C1(=CCCC1)C[C@@H](C(=O)[C@@]1(OC1)C)NC([C@H]([C@@H](C1=CC=C(C=C1)OC)O)NC([C@H](C)NC(CN1CCOCC1)=O)=O)=O GHYOCDFICYLMRF-UTIIJYGPSA-N 0.000 description 2
- QFLWZFQWSBQYPS-AWRAUJHKSA-N (3S)-3-[[(2S)-2-[[(2S)-2-[5-[(3aS,6aR)-2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]pentanoylamino]-3-methylbutanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-4-[1-bis(4-chlorophenoxy)phosphorylbutylamino]-4-oxobutanoic acid Chemical compound CCCC(NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@@H](NC(=O)CCCCC1SC[C@@H]2NC(=O)N[C@H]12)C(C)C)P(=O)(Oc1ccc(Cl)cc1)Oc1ccc(Cl)cc1 QFLWZFQWSBQYPS-AWRAUJHKSA-N 0.000 description 2
- NUJWKQSEJDYCDB-GNRVTEMESA-N (3s)-1-[(1s,2r,4r)-4-[methyl(propan-2-yl)amino]-2-propylcyclohexyl]-3-[[6-(trifluoromethyl)quinazolin-4-yl]amino]pyrrolidin-2-one Chemical compound CCC[C@@H]1C[C@H](N(C)C(C)C)CC[C@@H]1N1C(=O)[C@@H](NC=2C3=CC(=CC=C3N=CN=2)C(F)(F)F)CC1 NUJWKQSEJDYCDB-GNRVTEMESA-N 0.000 description 2
- DEQANNDTNATYII-OULOTJBUSA-N (4r,7s,10s,13r,16s,19r)-10-(4-aminobutyl)-19-[[(2r)-2-amino-3-phenylpropanoyl]amino]-16-benzyl-n-[(2r,3r)-1,3-dihydroxybutan-2-yl]-7-[(1r)-1-hydroxyethyl]-13-(1h-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentazacycloicosane-4-carboxa Chemical compound C([C@@H](N)C(=O)N[C@H]1CSSC[C@H](NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](CC=2C3=CC=CC=C3NC=2)NC(=O)[C@H](CC=2C=CC=CC=2)NC1=O)C(=O)N[C@H](CO)[C@H](O)C)C1=CC=CC=C1 DEQANNDTNATYII-OULOTJBUSA-N 0.000 description 2
- AZQWKYJCGOJGHM-UHFFFAOYSA-N 1,4-benzoquinone Chemical compound O=C1C=CC(=O)C=C1 AZQWKYJCGOJGHM-UHFFFAOYSA-N 0.000 description 2
- MSWZFWKMSRAUBD-IVMDWMLBSA-N 2-amino-2-deoxy-D-glucopyranose Chemical compound N[C@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O MSWZFWKMSRAUBD-IVMDWMLBSA-N 0.000 description 2
- XDCOYBQVEVSNNB-UHFFFAOYSA-N 4-[(7-naphthalen-2-yl-1-benzothiophen-2-yl)methylamino]butanoic acid Chemical compound OC(=O)CCCNCc1cc2cccc(-c3ccc4ccccc4c3)c2s1 XDCOYBQVEVSNNB-UHFFFAOYSA-N 0.000 description 2
- TXEBWPPWSVMYOA-UHFFFAOYSA-N 4-[3-[(1-amino-2-chloroethyl)amino]propyl]-1-[[3-(2-chlorophenyl)phenyl]methyl]-5-hydroxyimidazolidin-2-one Chemical compound NC(CCl)NCCCC1NC(=O)N(Cc2cccc(c2)-c2ccccc2Cl)C1O TXEBWPPWSVMYOA-UHFFFAOYSA-N 0.000 description 2
- OGWKCGZFUXNPDA-CFWMRBGOSA-N 5j49q6b70f Chemical compound C([C@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C=O)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 OGWKCGZFUXNPDA-CFWMRBGOSA-N 0.000 description 2
- STQGQHZAVUOBTE-UHFFFAOYSA-N 7-Cyan-hept-2t-en-4,6-diinsaeure Natural products C1=2C(O)=C3C(=O)C=4C(OC)=CC=CC=4C(=O)C3=C(O)C=2CC(O)(C(C)=O)CC1OC1CC(N)C(O)C(C)O1 STQGQHZAVUOBTE-UHFFFAOYSA-N 0.000 description 2
- KDCGOANMDULRCW-UHFFFAOYSA-N 7H-purine Chemical compound N1=CNC2=NC=NC2=C1 KDCGOANMDULRCW-UHFFFAOYSA-N 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- QCMHGCDOZLWPOT-FMNCTDSISA-N COC1=C(CC[C@@H]2CCC3=C(C2)C=CC(=C3)[C@H]2CC[C@](N)(CO)C2)C=CC=C1 Chemical compound COC1=C(CC[C@@H]2CCC3=C(C2)C=CC(=C3)[C@H]2CC[C@](N)(CO)C2)C=CC=C1 QCMHGCDOZLWPOT-FMNCTDSISA-N 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- PTOAARAWEBMLNO-KVQBGUIXSA-N Cladribine Chemical compound C1=NC=2C(N)=NC(Cl)=NC=2N1[C@H]1C[C@H](O)[C@@H](CO)O1 PTOAARAWEBMLNO-KVQBGUIXSA-N 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- CYQFCXCEBYINGO-DLBZAZTESA-N Dronabinol Natural products C1=C(C)CC[C@H]2C(C)(C)OC3=CC(CCCCC)=CC(O)=C3[C@H]21 CYQFCXCEBYINGO-DLBZAZTESA-N 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- AEMRFAOFKBGASW-UHFFFAOYSA-N Glycolic acid Chemical compound OCC(O)=O AEMRFAOFKBGASW-UHFFFAOYSA-N 0.000 description 2
- 102100039619 Granulocyte colony-stimulating factor Human genes 0.000 description 2
- 229940121710 HMGCoA reductase inhibitor Drugs 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- 102100040018 Interferon alpha-2 Human genes 0.000 description 2
- 108010047761 Interferon-alpha Proteins 0.000 description 2
- 102000006992 Interferon-alpha Human genes 0.000 description 2
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 2
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 2
- 108010000817 Leuprolide Proteins 0.000 description 2
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- 206010027476 Metastases Diseases 0.000 description 2
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 2
- NWIBSHFKIJFRCO-WUDYKRTCSA-N Mytomycin Chemical compound C1N2C(C(C(C)=C(N)C3=O)=O)=C3[C@@H](COC(N)=O)[C@@]2(OC)[C@@H]2[C@H]1N2 NWIBSHFKIJFRCO-WUDYKRTCSA-N 0.000 description 2
- PWKAYICUBVNJAZ-UHFFFAOYSA-N N-[(1-methylimidazol-4-yl)methyl]-4-(4-methylpiperidin-1-yl)pyrimidin-2-amine Chemical compound CN1C=NC(=C1)CNC1=NC=CC(=N1)N1CCC(CC1)C PWKAYICUBVNJAZ-UHFFFAOYSA-N 0.000 description 2
- ZDZOTLJHXYCWBA-VCVYQWHSSA-N N-debenzoyl-N-(tert-butoxycarbonyl)-10-deacetyltaxol Chemical compound O([C@H]1[C@H]2[C@@](C([C@H](O)C3=C(C)[C@@H](OC(=O)[C@H](O)[C@@H](NC(=O)OC(C)(C)C)C=4C=CC=CC=4)C[C@]1(O)C3(C)C)=O)(C)[C@@H](O)C[C@H]1OC[C@]12OC(=O)C)C(=O)C1=CC=CC=C1 ZDZOTLJHXYCWBA-VCVYQWHSSA-N 0.000 description 2
- TZCCKCLHNUSAMQ-DUGSHLAESA-N NC(=O)C[C@H](NC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](Cc2ccc(F)cc2)NC(=O)[C@H](Cc3c[nH]c4ccccc34)NC(=O)Cc5cccs5)C(=O)N Chemical compound NC(=O)C[C@H](NC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](Cc2ccc(F)cc2)NC(=O)[C@H](Cc3c[nH]c4ccccc34)NC(=O)Cc5cccs5)C(=O)N TZCCKCLHNUSAMQ-DUGSHLAESA-N 0.000 description 2
- AKQOBHZKBDHWQI-BZEFIUHZSA-N O[C@H]1C[C@@H](CCC1)N1C(C2(C3=C1N=C(N=C3)NC1=CC=C(C=C1)S(=O)(=O)NC([2H])([2H])[2H])CC2)=O Chemical compound O[C@H]1C[C@@H](CCC1)N1C(C2(C3=C1N=C(N=C3)NC1=CC=C(C=C1)S(=O)(=O)NC([2H])([2H])[2H])CC2)=O AKQOBHZKBDHWQI-BZEFIUHZSA-N 0.000 description 2
- 108010016076 Octreotide Proteins 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- 108700008625 Reporter Genes Proteins 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- CYQFCXCEBYINGO-UHFFFAOYSA-N THC Natural products C1=C(C)CCC2C(C)(C)OC3=CC(CCCCC)=CC(O)=C3C21 CYQFCXCEBYINGO-UHFFFAOYSA-N 0.000 description 2
- 108010050144 Triptorelin Pamoate Proteins 0.000 description 2
- DRTQHJPVMGBUCF-XVFCMESISA-N Uridine Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C=C1 DRTQHJPVMGBUCF-XVFCMESISA-N 0.000 description 2
- RJURFGZVJUQBHK-UHFFFAOYSA-N actinomycin D Natural products CC1OC(=O)C(C(C)C)N(C)C(=O)CN(C)C(=O)C2CCCN2C(=O)C(C(C)C)NC(=O)C1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)NC4C(=O)NC(C(N5CCCC5C(=O)N(C)CC(=O)N(C)C(C(C)C)C(=O)OC4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-UHFFFAOYSA-N 0.000 description 2
- 125000001931 aliphatic group Chemical group 0.000 description 2
- 125000003545 alkoxy group Chemical group 0.000 description 2
- 125000000217 alkyl group Chemical group 0.000 description 2
- OFCNXPDARWKPPY-UHFFFAOYSA-N allopurinol Chemical compound OC1=NC=NC2=C1C=NN2 OFCNXPDARWKPPY-UHFFFAOYSA-N 0.000 description 2
- 229960003459 allopurinol Drugs 0.000 description 2
- JKOQGQFVAUAYPM-UHFFFAOYSA-N amifostine Chemical compound NCCCNCCSP(O)(O)=O JKOQGQFVAUAYPM-UHFFFAOYSA-N 0.000 description 2
- 229960001097 amifostine Drugs 0.000 description 2
- 150000001412 amines Chemical class 0.000 description 2
- 150000007514 bases Chemical class 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- MSWZFWKMSRAUBD-UHFFFAOYSA-N beta-D-galactosamine Natural products NC1C(O)OC(CO)C(O)C1O MSWZFWKMSRAUBD-UHFFFAOYSA-N 0.000 description 2
- RYYVLZVUVIJVGH-UHFFFAOYSA-N caffeine Chemical compound CN1C(=O)N(C)C(=O)C2=C1N=CN2C RYYVLZVUVIJVGH-UHFFFAOYSA-N 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 229960002436 cladribine Drugs 0.000 description 2
- 229940125797 compound 12 Drugs 0.000 description 2
- 229940127108 compound 5g Drugs 0.000 description 2
- 229940126136 compound 5i Drugs 0.000 description 2
- 125000004122 cyclic group Chemical group 0.000 description 2
- 231100000433 cytotoxic Toxicity 0.000 description 2
- 230000001472 cytotoxic effect Effects 0.000 description 2
- 229960000975 daunorubicin Drugs 0.000 description 2
- STQGQHZAVUOBTE-VGBVRHCVSA-N daunorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(C)=O)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 STQGQHZAVUOBTE-VGBVRHCVSA-N 0.000 description 2
- CYQFCXCEBYINGO-IAGOWNOFSA-N delta1-THC Chemical compound C1=C(C)CC[C@H]2C(C)(C)OC3=CC(CCCCC)=CC(O)=C3[C@@H]21 CYQFCXCEBYINGO-IAGOWNOFSA-N 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- 231100000673 dose–response relationship Toxicity 0.000 description 2
- 229960004679 doxorubicin Drugs 0.000 description 2
- 229960004242 dronabinol Drugs 0.000 description 2
- 229930182833 estradiol Natural products 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- 229960004177 filgrastim Drugs 0.000 description 2
- RFHAOTPXVQNOHP-UHFFFAOYSA-N fluconazole Chemical compound C1=NC=NN1CC(C=1C(=CC(F)=CC=1)F)(O)CN1C=NC=N1 RFHAOTPXVQNOHP-UHFFFAOYSA-N 0.000 description 2
- 238000012395 formulation development Methods 0.000 description 2
- 229960002442 glucosamine Drugs 0.000 description 2
- KWIUHFFTVRNATP-UHFFFAOYSA-N glycine betaine Chemical compound C[N+](C)(C)CC([O-])=O KWIUHFFTVRNATP-UHFFFAOYSA-N 0.000 description 2
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 2
- 229960002885 histidine Drugs 0.000 description 2
- HHXHVIJIIXKSOE-QILQGKCVSA-N histrelin Chemical compound CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)CC(N=C1)=CN1CC1=CC=CC=C1 HHXHVIJIIXKSOE-QILQGKCVSA-N 0.000 description 2
- 108700020746 histrelin Proteins 0.000 description 2
- 229960002193 histrelin Drugs 0.000 description 2
- JYGXADMDTFJGBT-VWUMJDOOSA-N hydrocortisone Chemical compound O=C1CC[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 JYGXADMDTFJGBT-VWUMJDOOSA-N 0.000 description 2
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 2
- YOZNUFWCRFCGIH-BYFNXCQMSA-L hydroxocobalamin Chemical compound O[Co+]N([C@]1([H])[C@H](CC(N)=O)[C@]\2(CCC(=O)NC[C@H](C)OP(O)(=O)OC3[C@H]([C@H](O[C@@H]3CO)N3C4=CC(C)=C(C)C=C4N=C3)O)C)C/2=C(C)\C([C@H](C/2(C)C)CCC(N)=O)=N\C\2=C\C([C@H]([C@@]/2(CC(N)=O)C)CCC(N)=O)=N\C\2=C(C)/C2=N[C@]1(C)[C@@](C)(CC(N)=O)[C@@H]2CCC(N)=O YOZNUFWCRFCGIH-BYFNXCQMSA-L 0.000 description 2
- 238000009169 immunotherapy Methods 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- SUMDYPCJJOFFON-UHFFFAOYSA-N isethionic acid Chemical compound OCCS(O)(=O)=O SUMDYPCJJOFFON-UHFFFAOYSA-N 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 239000002502 liposome Substances 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 229910052749 magnesium Inorganic materials 0.000 description 2
- GLVAUDGFNGKCSF-UHFFFAOYSA-N mercaptopurine Chemical compound S=C1NC=NC2=C1NC=N2 GLVAUDGFNGKCSF-UHFFFAOYSA-N 0.000 description 2
- 230000009401 metastasis Effects 0.000 description 2
- 229960000485 methotrexate Drugs 0.000 description 2
- 238000010172 mouse model Methods 0.000 description 2
- JBLLRCOZJMVOAE-HSQYWUDLSA-N n-[(2s)-1-[[(2s)-1-(1,3-benzothiazol-2-yl)-1-oxo-3-[(3s)-2-oxopyrrolidin-3-yl]propan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]-4-methoxy-1h-indole-2-carboxamide Chemical compound C([C@H](NC(=O)[C@H](CC(C)C)NC(=O)C=1NC=2C=CC=C(C=2C=1)OC)C(=O)C=1SC2=CC=CC=C2N=1)[C@@H]1CCNC1=O JBLLRCOZJMVOAE-HSQYWUDLSA-N 0.000 description 2
- 229960002700 octreotide Drugs 0.000 description 2
- 150000007530 organic bases Chemical class 0.000 description 2
- 229960002139 pilocarpine hydrochloride Drugs 0.000 description 2
- RNAICSBVACLLGM-GNAZCLTHSA-N pilocarpine hydrochloride Chemical compound Cl.C1OC(=O)[C@@H](CC)[C@H]1CC1=CN=CN1C RNAICSBVACLLGM-GNAZCLTHSA-N 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 229960004618 prednisone Drugs 0.000 description 2
- XOFYZVNMUHMLCC-ZPOLXVRWSA-N prednisone Chemical compound O=C1C=C[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 XOFYZVNMUHMLCC-ZPOLXVRWSA-N 0.000 description 2
- 230000035755 proliferation Effects 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 238000010791 quenching Methods 0.000 description 2
- RWWYLEGWBNMMLJ-YSOARWBDSA-N remdesivir Chemical compound NC1=NC=NN2C1=CC=C2[C@]1([C@@H]([C@@H]([C@H](O1)CO[P@](=O)(OC1=CC=CC=C1)N[C@H](C(=O)OCC(CC)CC)C)O)O)C#N RWWYLEGWBNMMLJ-YSOARWBDSA-N 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 2
- 229920006395 saturated elastomer Polymers 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 235000017557 sodium bicarbonate Nutrition 0.000 description 2
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 2
- JHJLBTNAGRQEKS-UHFFFAOYSA-M sodium bromide Chemical compound [Na+].[Br-] JHJLBTNAGRQEKS-UHFFFAOYSA-M 0.000 description 2
- YAPQBXQYLJRXSA-UHFFFAOYSA-N theobromine Chemical compound CN1C(=O)NC(=O)C2=C1N=CN2C YAPQBXQYLJRXSA-UHFFFAOYSA-N 0.000 description 2
- WYWHKKSPHMUBEB-UHFFFAOYSA-N tioguanine Chemical compound N1C(N)=NC(=S)C2=C1N=CN2 WYWHKKSPHMUBEB-UHFFFAOYSA-N 0.000 description 2
- XFCLJVABOIYOMF-QPLCGJKRSA-N toremifene Chemical compound C1=CC(OCCN(C)C)=CC=C1C(\C=1C=CC=CC=1)=C(\CCCl)C1=CC=CC=C1 XFCLJVABOIYOMF-QPLCGJKRSA-N 0.000 description 2
- 229960005026 toremifene Drugs 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 2
- BFDBKMOZYNOTPK-UHFFFAOYSA-N vonoprazan Chemical compound C=1C=CN=CC=1S(=O)(=O)N1C=C(CNC)C=C1C1=CC=CC=C1F BFDBKMOZYNOTPK-UHFFFAOYSA-N 0.000 description 2
- DNXHEGUUPJUMQT-UHFFFAOYSA-N (+)-estrone Natural products OC1=CC=C2C3CCC(C)(C(CC4)=O)C4C3CCC2=C1 DNXHEGUUPJUMQT-UHFFFAOYSA-N 0.000 description 1
- FANCTJAFZSYTIS-IQUVVAJASA-N (1r,3s,5z)-5-[(2e)-2-[(1r,3as,7ar)-7a-methyl-1-[(2r)-4-(phenylsulfonimidoyl)butan-2-yl]-2,3,3a,5,6,7-hexahydro-1h-inden-4-ylidene]ethylidene]-4-methylidenecyclohexane-1,3-diol Chemical compound C([C@@H](C)[C@@H]1[C@]2(CCCC(/[C@@H]2CC1)=C\C=C\1C([C@@H](O)C[C@H](O)C/1)=C)C)CS(=N)(=O)C1=CC=CC=C1 FANCTJAFZSYTIS-IQUVVAJASA-N 0.000 description 1
- GNPHAOQLHRZODS-ZQWQDMLBSA-N (1s,2s,3s,4s)-3,4-bis[butyl-[(4-phenoxyphenyl)methyl]carbamoyl]cyclobutane-1,2-dicarboxylic acid Chemical compound O=C([C@H]1[C@@H]([C@@H]([C@H]1C(O)=O)C(O)=O)C(=O)N(CCCC)CC=1C=CC(OC=2C=CC=CC=2)=CC=1)N(CCCC)CC(C=C1)=CC=C1OC1=CC=CC=C1 GNPHAOQLHRZODS-ZQWQDMLBSA-N 0.000 description 1
- VESKBLGTHHPZJF-QNWVGRARSA-N (2s)-2-amino-5-[[(2r)-2-amino-3-[2-[bis[bis(2-chloroethyl)amino]phosphoryloxy]ethylsulfonyl]propanoyl]-[(r)-carboxy(phenyl)methyl]amino]-5-oxopentanoic acid Chemical compound ClCCN(CCCl)P(=O)(N(CCCl)CCCl)OCCS(=O)(=O)C[C@H](N)C(=O)N(C(=O)CC[C@H](N)C(O)=O)[C@@H](C(O)=O)C1=CC=CC=C1 VESKBLGTHHPZJF-QNWVGRARSA-N 0.000 description 1
- ZBVJFYPGLGEMIN-OYLNGHKZSA-N (2s)-n-[(2s)-1-[[(2s)-1-[[(2s)-1-[[(2s)-1-[[(2r)-1-[[(2s)-1-[[(2s)-1-[(2s)-2-[(2-amino-2-oxoethyl)carbamoyl]pyrrolidin-1-yl]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-3-(1h-indol-3-yl)-1-oxopropan-2-yl]amino]-3-( Chemical compound C1=CC=C2C(CC=3C4=CC=CC=C4C=C(C=3O)C(=O)O)=C(O)C(C(O)=O)=CC2=C1.C([C@@H](C(=O)N[C@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1[C@@H](CCC1)C(=O)NCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)C1=CC=C(O)C=C1 ZBVJFYPGLGEMIN-OYLNGHKZSA-N 0.000 description 1
- DRHZYJAUECRAJM-DWSYSWFDSA-N (2s,3s,4s,5r,6r)-6-[[(3s,4s,4ar,6ar,6bs,8r,8ar,12as,14ar,14br)-8a-[(2s,3r,4s,5r,6r)-3-[(2s,3r,4s,5r,6s)-5-[(2s,3r,4s,5r)-4-[(2s,3r,4r)-3,4-dihydroxy-4-(hydroxymethyl)oxolan-2-yl]oxy-3,5-dihydroxyoxan-2-yl]oxy-3,4-dihydroxy-6-methyloxan-2-yl]oxy-5-[(3s,5s, Chemical compound O([C@H]1[C@H](O)[C@H](O[C@H]([C@@H]1O[C@H]1[C@@H]([C@@H](O)[C@@H](O)[C@@H](CO)O1)O)O[C@H]1CC[C@]2(C)[C@H]3CC=C4[C@@H]5CC(C)(C)CC[C@@]5([C@@H](C[C@@]4(C)[C@]3(C)CC[C@H]2[C@@]1(C=O)C)O)C(=O)O[C@@H]1O[C@H](C)[C@@H]([C@@H]([C@H]1O[C@H]1[C@@H]([C@H](O)[C@@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@](O)(CO)CO3)O)[C@H](O)CO2)O)[C@H](C)O1)O)O)OC(=O)C[C@@H](O)C[C@H](OC(=O)C[C@@H](O)C[C@@H]([C@@H](C)CC)O[C@H]1[C@@H]([C@@H](O)[C@H](CO)O1)O)[C@@H](C)CC)C(O)=O)[C@@H]1OC[C@@H](O)[C@H](O)[C@H]1O DRHZYJAUECRAJM-DWSYSWFDSA-N 0.000 description 1
- HBZBAMXERPYTFS-SECBINFHSA-N (4S)-2-(6,7-dihydro-5H-pyrrolo[3,2-f][1,3]benzothiazol-2-yl)-4,5-dihydro-1,3-thiazole-4-carboxylic acid Chemical compound OC(=O)[C@H]1CSC(=N1)c1nc2cc3CCNc3cc2s1 HBZBAMXERPYTFS-SECBINFHSA-N 0.000 description 1
- PNHBRYIAJCYNDA-VQCQRNETSA-N (4r)-6-[2-[2-ethyl-4-(4-fluorophenyl)-6-phenylpyridin-3-yl]ethyl]-4-hydroxyoxan-2-one Chemical compound C([C@H](O)C1)C(=O)OC1CCC=1C(CC)=NC(C=2C=CC=CC=2)=CC=1C1=CC=C(F)C=C1 PNHBRYIAJCYNDA-VQCQRNETSA-N 0.000 description 1
- PUDHBTGHUJUUFI-SCTWWAJVSA-N (4r,7s,10s,13r,16s,19r)-10-(4-aminobutyl)-n-[(2s,3r)-1-amino-3-hydroxy-1-oxobutan-2-yl]-19-[[(2r)-2-amino-3-naphthalen-2-ylpropanoyl]amino]-16-[(4-hydroxyphenyl)methyl]-13-(1h-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-7-propan-2-yl-1,2-dithia-5,8,11,14,17-p Chemical compound C([C@H]1C(=O)N[C@H](CC=2C3=CC=CC=C3NC=2)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(N[C@@H](CSSC[C@@H](C(=O)N1)NC(=O)[C@H](N)CC=1C=C2C=CC=CC2=CC=1)C(=O)N[C@@H]([C@@H](C)O)C(N)=O)=O)C(C)C)C1=CC=C(O)C=C1 PUDHBTGHUJUUFI-SCTWWAJVSA-N 0.000 description 1
- NWXMGUDVXFXRIG-WESIUVDSSA-N (4s,4as,5as,6s,12ar)-4-(dimethylamino)-1,6,10,11,12a-pentahydroxy-6-methyl-3,12-dioxo-4,4a,5,5a-tetrahydrotetracene-2-carboxamide Chemical compound C1=CC=C2[C@](O)(C)[C@H]3C[C@H]4[C@H](N(C)C)C(=O)C(C(N)=O)=C(O)[C@@]4(O)C(=O)C3=C(O)C2=C1O NWXMGUDVXFXRIG-WESIUVDSSA-N 0.000 description 1
- MWWSFMDVAYGXBV-FGBSZODSSA-N (7s,9s)-7-[(2r,4s,5r,6s)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-9-(2-hydroxyacetyl)-4-methoxy-8,10-dihydro-7h-tetracene-5,12-dione;hydron;chloride Chemical compound Cl.O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@@H](O)[C@H](C)O1 MWWSFMDVAYGXBV-FGBSZODSSA-N 0.000 description 1
- FPVKHBSQESCIEP-UHFFFAOYSA-N (8S)-3-(2-deoxy-beta-D-erythro-pentofuranosyl)-3,6,7,8-tetrahydroimidazo[4,5-d][1,3]diazepin-8-ol Natural products C1C(O)C(CO)OC1N1C(NC=NCC2O)=C2N=C1 FPVKHBSQESCIEP-UHFFFAOYSA-N 0.000 description 1
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 1
- FDKXTQMXEQVLRF-ZHACJKMWSA-N (E)-dacarbazine Chemical compound CN(C)\N=N\c1[nH]cnc1C(N)=O FDKXTQMXEQVLRF-ZHACJKMWSA-N 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- UWYVPFMHMJIBHE-OWOJBTEDSA-N (e)-2-hydroxybut-2-enedioic acid Chemical compound OC(=O)\C=C(\O)C(O)=O UWYVPFMHMJIBHE-OWOJBTEDSA-N 0.000 description 1
- LBDAQUYUIYKBKJ-UHFFFAOYSA-N 1,2,3,4-tetrahydroisoquinoline-6,7-diol;hydrobromide Chemical compound Br.C1CNCC2=C1C=C(O)C(O)=C2 LBDAQUYUIYKBKJ-UHFFFAOYSA-N 0.000 description 1
- HJTAZXHBEBIQQX-UHFFFAOYSA-N 1,5-bis(chloromethyl)naphthalene Chemical compound C1=CC=C2C(CCl)=CC=CC2=C1CCl HJTAZXHBEBIQQX-UHFFFAOYSA-N 0.000 description 1
- MRBFGEHILMYPTF-UHFFFAOYSA-N 1-(2-Pyrimidyl)piperazine Chemical compound C1CNCCN1C1=NC=CC=N1 MRBFGEHILMYPTF-UHFFFAOYSA-N 0.000 description 1
- UBDLJNPJCXUZGB-UHFFFAOYSA-N 1-acetylpiperidin-2-one Chemical compound CC(=O)N1CCCCC1=O UBDLJNPJCXUZGB-UHFFFAOYSA-N 0.000 description 1
- YLHUPYSUKYAIBW-UHFFFAOYSA-N 1-acetylpyrrolidin-2-one Chemical compound CC(=O)N1CCCC1=O YLHUPYSUKYAIBW-UHFFFAOYSA-N 0.000 description 1
- CPKVUHPKYQGHMW-UHFFFAOYSA-N 1-ethenylpyrrolidin-2-one;molecular iodine Chemical compound II.C=CN1CCCC1=O CPKVUHPKYQGHMW-UHFFFAOYSA-N 0.000 description 1
- VSNHCAURESNICA-NJFSPNSNSA-N 1-oxidanylurea Chemical compound N[14C](=O)NO VSNHCAURESNICA-NJFSPNSNSA-N 0.000 description 1
- YHGIBJZESOLKPH-UHFFFAOYSA-N 1-phenyl-2-(2,4,6-trimethylphenyl)ethanone Chemical compound CC1=CC(C)=CC(C)=C1CC(=O)C1=CC=CC=C1 YHGIBJZESOLKPH-UHFFFAOYSA-N 0.000 description 1
- OQZBAQXTXNIPRA-UHFFFAOYSA-N 1-pyridin-4-ylpiperazine Chemical compound C1CNCCN1C1=CC=NC=C1 OQZBAQXTXNIPRA-UHFFFAOYSA-N 0.000 description 1
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 1
- BFPYWIDHMRZLRN-UHFFFAOYSA-N 17alpha-ethynyl estradiol Natural products OC1=CC=C2C3CCC(C)(C(CC4)(O)C#C)C4C3CCC2=C1 BFPYWIDHMRZLRN-UHFFFAOYSA-N 0.000 description 1
- GCKMFJBGXUYNAG-UHFFFAOYSA-N 17alpha-methyltestosterone Natural products C1CC2=CC(=O)CCC2(C)C2C1C1CCC(C)(O)C1(C)CC2 GCKMFJBGXUYNAG-UHFFFAOYSA-N 0.000 description 1
- QMVPQBFHUJZJCS-NTKFZFFISA-N 1v8x590xdp Chemical compound O=C1N(NC(CO)CO)C(=O)C(C2=C3[CH]C=C(O)C=C3NC2=C23)=C1C2=C1C=CC(O)=C[C]1N3[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O QMVPQBFHUJZJCS-NTKFZFFISA-N 0.000 description 1
- ROZCIVXTLACYNY-UHFFFAOYSA-N 2,3,4,5,6-pentafluoro-n-(3-fluoro-4-methoxyphenyl)benzenesulfonamide Chemical compound C1=C(F)C(OC)=CC=C1NS(=O)(=O)C1=C(F)C(F)=C(F)C(F)=C1F ROZCIVXTLACYNY-UHFFFAOYSA-N 0.000 description 1
- SRKVJDYNPSMHJM-UHFFFAOYSA-N 2-(chloromethyl)-4-methoxy-3,5-dimethylpyridine Chemical compound COC1=C(C)C=NC(CCl)=C1C SRKVJDYNPSMHJM-UHFFFAOYSA-N 0.000 description 1
- MOMFXATYAINJML-UHFFFAOYSA-N 2-Acetylthiazole Chemical group CC(=O)C1=NC=CS1 MOMFXATYAINJML-UHFFFAOYSA-N 0.000 description 1
- LBLYYCQCTBFVLH-UHFFFAOYSA-N 2-Methylbenzenesulfonic acid Chemical compound CC1=CC=CC=C1S(O)(=O)=O LBLYYCQCTBFVLH-UHFFFAOYSA-N 0.000 description 1
- QLVGHFBUSGYCCG-UHFFFAOYSA-N 2-amino-n-(1-cyano-2-phenylethyl)acetamide Chemical compound NCC(=O)NC(C#N)CC1=CC=CC=C1 QLVGHFBUSGYCCG-UHFFFAOYSA-N 0.000 description 1
- IVLXQGJVBGMLRR-UHFFFAOYSA-N 2-aminoacetic acid;hydron;chloride Chemical compound Cl.NCC(O)=O IVLXQGJVBGMLRR-UHFFFAOYSA-N 0.000 description 1
- BFSVOASYOCHEOV-UHFFFAOYSA-N 2-diethylaminoethanol Chemical compound CCN(CC)CCO BFSVOASYOCHEOV-UHFFFAOYSA-N 0.000 description 1
- 229940013085 2-diethylaminoethanol Drugs 0.000 description 1
- LGEXGKUJMFHVSY-UHFFFAOYSA-N 2-n,4-n,6-n-trimethyl-1,3,5-triazine-2,4,6-triamine Chemical compound CNC1=NC(NC)=NC(NC)=N1 LGEXGKUJMFHVSY-UHFFFAOYSA-N 0.000 description 1
- WLJVXDMOQOGPHL-PPJXEINESA-N 2-phenylacetic acid Chemical compound O[14C](=O)CC1=CC=CC=C1 WLJVXDMOQOGPHL-PPJXEINESA-N 0.000 description 1
- YLLRPQWLASQXSI-UHFFFAOYSA-N 3-(4b,8a,9,9a-tetrahydro-4aH-pyrido[2,3-b]indol-4-ylamino)phenol Chemical compound Oc1cccc(NC2=CC=NC3NC4C=CC=CC4C23)c1 YLLRPQWLASQXSI-UHFFFAOYSA-N 0.000 description 1
- WEVYNIUIFUYDGI-UHFFFAOYSA-N 3-[6-[4-(trifluoromethoxy)anilino]-4-pyrimidinyl]benzamide Chemical compound NC(=O)C1=CC=CC(C=2N=CN=C(NC=3C=CC(OC(F)(F)F)=CC=3)C=2)=C1 WEVYNIUIFUYDGI-UHFFFAOYSA-N 0.000 description 1
- WUIABRMSWOKTOF-OYALTWQYSA-N 3-[[2-[2-[2-[[(2s,3r)-2-[[(2s,3s,4r)-4-[[(2s,3r)-2-[[6-amino-2-[(1s)-3-amino-1-[[(2s)-2,3-diamino-3-oxopropyl]amino]-3-oxopropyl]-5-methylpyrimidine-4-carbonyl]amino]-3-[(2r,3s,4s,5s,6s)-3-[(2r,3s,4s,5r,6r)-4-carbamoyloxy-3,5-dihydroxy-6-(hydroxymethyl)ox Chemical compound OS([O-])(=O)=O.N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC=C(N=1)C=1SC=C(N=1)C(=O)NCCC[S+](C)C)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1NC=NC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C WUIABRMSWOKTOF-OYALTWQYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- AOJJSUZBOXZQNB-VTZDEGQISA-N 4'-epidoxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-VTZDEGQISA-N 0.000 description 1
- CLPFFLWZZBQMAO-UHFFFAOYSA-N 4-(5,6,7,8-tetrahydroimidazo[1,5-a]pyridin-5-yl)benzonitrile Chemical compound C1=CC(C#N)=CC=C1C1N2C=NC=C2CCC1 CLPFFLWZZBQMAO-UHFFFAOYSA-N 0.000 description 1
- AKPSLMUFDIXDJJ-UHFFFAOYSA-N 4-(bromomethyl)-1,2-dimethoxybenzene Chemical compound COC1=CC=C(CBr)C=C1OC AKPSLMUFDIXDJJ-UHFFFAOYSA-N 0.000 description 1
- AKJHMTWEGVYYSE-AIRMAKDCSA-N 4-HPR Chemical compound C=1C=C(O)C=CC=1NC(=O)/C=C(\C)/C=C/C=C(C)C=CC1=C(C)CCCC1(C)C AKJHMTWEGVYYSE-AIRMAKDCSA-N 0.000 description 1
- HVBSAKJJOYLTQU-UHFFFAOYSA-N 4-aminobenzenesulfonic acid Chemical compound NC1=CC=C(S(O)(=O)=O)C=C1 HVBSAKJJOYLTQU-UHFFFAOYSA-N 0.000 description 1
- TVZGACDUOSZQKY-LBPRGKRZSA-N 4-aminofolic acid Chemical compound C1=NC2=NC(N)=NC(N)=C2N=C1CNC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 TVZGACDUOSZQKY-LBPRGKRZSA-N 0.000 description 1
- CFKMVGJGLGKFKI-UHFFFAOYSA-N 4-chloro-m-cresol Chemical compound CC1=CC(O)=CC=C1Cl CFKMVGJGLGKFKI-UHFFFAOYSA-N 0.000 description 1
- HVCNXQOWACZAFN-UHFFFAOYSA-N 4-ethylmorpholine Chemical compound CCN1CCOCC1 HVCNXQOWACZAFN-UHFFFAOYSA-N 0.000 description 1
- NMUSYJAQQFHJEW-UHFFFAOYSA-N 5-Azacytidine Natural products O=C1N=C(N)N=CN1C1C(O)C(O)C(CO)O1 NMUSYJAQQFHJEW-UHFFFAOYSA-N 0.000 description 1
- XAUDJQYHKZQPEU-KVQBGUIXSA-N 5-aza-2'-deoxycytidine Chemical compound O=C1N=C(N)N=CN1[C@@H]1O[C@H](CO)[C@@H](O)C1 XAUDJQYHKZQPEU-KVQBGUIXSA-N 0.000 description 1
- NMUSYJAQQFHJEW-KVTDHHQDSA-N 5-azacytidine Chemical compound O=C1N=C(N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 NMUSYJAQQFHJEW-KVTDHHQDSA-N 0.000 description 1
- HFEKDTCAMMOLQP-RRKCRQDMSA-N 5-fluorodeoxyuridine monophosphate Chemical compound O1[C@H](COP(O)(O)=O)[C@@H](O)C[C@@H]1N1C(=O)NC(=O)C(F)=C1 HFEKDTCAMMOLQP-RRKCRQDMSA-N 0.000 description 1
- SSQRWSXHOJUZIT-UHFFFAOYSA-N 6,7-bis(2-methoxyethoxy)-1,2,3,4-tetrahydroisoquinoline hydrochloride Chemical compound Cl.COCCOC=1C=C2CCNCC2=CC1OCCOC SSQRWSXHOJUZIT-UHFFFAOYSA-N 0.000 description 1
- VVIAGPKUTFNRDU-UHFFFAOYSA-N 6S-folinic acid Natural products C1NC=2NC(N)=NC(=O)C=2N(C=O)C1CNC1=CC=C(C(=O)NC(CCC(O)=O)C(O)=O)C=C1 VVIAGPKUTFNRDU-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- MKBLHFILKIKSQM-UHFFFAOYSA-N 9-methyl-3-[(2-methyl-1h-imidazol-3-ium-3-yl)methyl]-2,3-dihydro-1h-carbazol-4-one;chloride Chemical compound Cl.CC1=NC=CN1CC1C(=O)C(C=2C(=CC=CC=2)N2C)=C2CC1 MKBLHFILKIKSQM-UHFFFAOYSA-N 0.000 description 1
- OGSPWJRAVKPPFI-UHFFFAOYSA-N Alendronic Acid Chemical compound NCCCC(O)(P(O)(O)=O)P(O)(O)=O OGSPWJRAVKPPFI-UHFFFAOYSA-N 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 229930195573 Amycin Natural products 0.000 description 1
- MLDQJTXFUGDVEO-UHFFFAOYSA-N BAY-43-9006 Chemical compound C1=NC(C(=O)NC)=CC(OC=2C=CC(NC(=O)NC=3C=C(C(Cl)=CC=3)C(F)(F)F)=CC=2)=C1 MLDQJTXFUGDVEO-UHFFFAOYSA-N 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- 206010004593 Bile duct cancer Diseases 0.000 description 1
- 108010006654 Bleomycin Proteins 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- COVZYZSDYWQREU-UHFFFAOYSA-N Busulfan Chemical compound CS(=O)(=O)OCCCCOS(C)(=O)=O COVZYZSDYWQREU-UHFFFAOYSA-N 0.000 description 1
- 238000011740 C57BL/6 mouse Methods 0.000 description 1
- UOFIQIJIFYROJM-UHFFFAOYSA-N CS(=O)(=O)OCC1=NC(=C(N=C1C)C)C Chemical compound CS(=O)(=O)OCC1=NC(=C(N=C1C)C)C UOFIQIJIFYROJM-UHFFFAOYSA-N 0.000 description 1
- FVLVBPDQNARYJU-XAHDHGMMSA-N C[C@H]1CCC(CC1)NC(=O)N(CCCl)N=O Chemical compound C[C@H]1CCC(CC1)NC(=O)N(CCCl)N=O FVLVBPDQNARYJU-XAHDHGMMSA-N 0.000 description 1
- 102000055006 Calcitonin Human genes 0.000 description 1
- 108060001064 Calcitonin Proteins 0.000 description 1
- GAGWJHPBXLXJQN-UORFTKCHSA-N Capecitabine Chemical compound C1=C(F)C(NC(=O)OCCCCC)=NC(=O)N1[C@H]1[C@H](O)[C@H](O)[C@@H](C)O1 GAGWJHPBXLXJQN-UORFTKCHSA-N 0.000 description 1
- GAGWJHPBXLXJQN-UHFFFAOYSA-N Capecitabine Natural products C1=C(F)C(NC(=O)OCCCCC)=NC(=O)N1C1C(O)C(O)C(C)O1 GAGWJHPBXLXJQN-UHFFFAOYSA-N 0.000 description 1
- 206010008342 Cervix carcinoma Diseases 0.000 description 1
- 229920001661 Chitosan Polymers 0.000 description 1
- JWBOIMRXGHLCPP-UHFFFAOYSA-N Chloditan Chemical compound C=1C=CC=C(Cl)C=1C(C(Cl)Cl)C1=CC=C(Cl)C=C1 JWBOIMRXGHLCPP-UHFFFAOYSA-N 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 206010009944 Colon cancer Diseases 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- CMSMOCZEIVJLDB-UHFFFAOYSA-N Cyclophosphamide Chemical compound ClCCN(CCCl)P1(=O)NCCCO1 CMSMOCZEIVJLDB-UHFFFAOYSA-N 0.000 description 1
- UHDGCWIWMRVCDJ-CCXZUQQUSA-N Cytarabine Chemical compound O=C1N=C(N)C=CN1[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O1 UHDGCWIWMRVCDJ-CCXZUQQUSA-N 0.000 description 1
- 230000004568 DNA-binding Effects 0.000 description 1
- 108010092160 Dactinomycin Proteins 0.000 description 1
- 108010019673 Darbepoetin alfa Proteins 0.000 description 1
- GJKXGJCSJWBJEZ-XRSSZCMZSA-N Deslorelin Chemical compound CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H]1NC(=O)CC1)CC1=CNC2=CC=CC=C12 GJKXGJCSJWBJEZ-XRSSZCMZSA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 101100391174 Dictyostelium discoideum forC gene Proteins 0.000 description 1
- MWWSFMDVAYGXBV-RUELKSSGSA-N Doxorubicin hydrochloride Chemical compound Cl.O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 MWWSFMDVAYGXBV-RUELKSSGSA-N 0.000 description 1
- 108050002772 E3 ubiquitin-protein ligase Mdm2 Proteins 0.000 description 1
- 102000012199 E3 ubiquitin-protein ligase Mdm2 Human genes 0.000 description 1
- 206010014733 Endometrial cancer Diseases 0.000 description 1
- 206010014759 Endometrial neoplasm Diseases 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- HTIJFSOGRVMCQR-UHFFFAOYSA-N Epirubicin Natural products COc1cccc2C(=O)c3c(O)c4CC(O)(CC(OC5CC(N)C(=O)C(C)O5)c4c(O)c3C(=O)c12)C(=O)CO HTIJFSOGRVMCQR-UHFFFAOYSA-N 0.000 description 1
- 108010074604 Epoetin Alfa Proteins 0.000 description 1
- 108090000394 Erythropoietin Proteins 0.000 description 1
- 102000003951 Erythropoietin Human genes 0.000 description 1
- 208000000461 Esophageal Neoplasms Diseases 0.000 description 1
- BFPYWIDHMRZLRN-SLHNCBLASA-N Ethinyl estradiol Chemical compound OC1=CC=C2[C@H]3CC[C@](C)([C@](CC4)(O)C#C)[C@@H]4[C@@H]3CCC2=C1 BFPYWIDHMRZLRN-SLHNCBLASA-N 0.000 description 1
- DBVJJBKOTRCVKF-UHFFFAOYSA-N Etidronic acid Chemical compound OP(=O)(O)C(O)(C)P(O)(O)=O DBVJJBKOTRCVKF-UHFFFAOYSA-N 0.000 description 1
- CWYNVVGOOAEACU-UHFFFAOYSA-N Fe2+ Chemical compound [Fe+2] CWYNVVGOOAEACU-UHFFFAOYSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- GHASVSINZRGABV-UHFFFAOYSA-N Fluorouracil Chemical compound FC1=CNC(=O)NC1=O GHASVSINZRGABV-UHFFFAOYSA-N 0.000 description 1
- VWUXBMIQPBEWFH-WCCTWKNTSA-N Fulvestrant Chemical compound OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3[C@H](CCCCCCCCCS(=O)CCCC(F)(F)C(F)(F)F)CC2=C1 VWUXBMIQPBEWFH-WCCTWKNTSA-N 0.000 description 1
- 208000032612 Glial tumor Diseases 0.000 description 1
- 206010018338 Glioma Diseases 0.000 description 1
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 1
- 108010069236 Goserelin Proteins 0.000 description 1
- BLCLNMBMMGCOAS-URPVMXJPSA-N Goserelin Chemical compound C([C@@H](C(=O)N[C@H](COC(C)(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N1[C@@H](CCC1)C(=O)NNC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H]1NC(=O)CC1)C1=CC=C(O)C=C1 BLCLNMBMMGCOAS-URPVMXJPSA-N 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101000987586 Homo sapiens Eosinophil peroxidase Proteins 0.000 description 1
- 101000920686 Homo sapiens Erythropoietin Proteins 0.000 description 1
- 101001033249 Homo sapiens Interleukin-1 beta Proteins 0.000 description 1
- 108091069885 IAP family Proteins 0.000 description 1
- 102000040104 IAP family Human genes 0.000 description 1
- MPBVHIBUJCELCL-UHFFFAOYSA-N Ibandronate Chemical compound CCCCCN(C)CCC(O)(P(O)(O)=O)P(O)(O)=O MPBVHIBUJCELCL-UHFFFAOYSA-N 0.000 description 1
- XDXDZDZNSLXDNA-TZNDIEGXSA-N Idarubicin Chemical compound C1[C@H](N)[C@H](O)[C@H](C)O[C@H]1O[C@@H]1C2=C(O)C(C(=O)C3=CC=CC=C3C3=O)=C3C(O)=C2C[C@@](O)(C(C)=O)C1 XDXDZDZNSLXDNA-TZNDIEGXSA-N 0.000 description 1
- XDXDZDZNSLXDNA-UHFFFAOYSA-N Idarubicin Natural products C1C(N)C(O)C(C)OC1OC1C2=C(O)C(C(=O)C3=CC=CC=C3C3=O)=C3C(O)=C2CC(O)(C(C)=O)C1 XDXDZDZNSLXDNA-UHFFFAOYSA-N 0.000 description 1
- 108010078049 Interferon alpha-2 Proteins 0.000 description 1
- 108010079944 Interferon-alpha2b Proteins 0.000 description 1
- 102000008070 Interferon-gamma Human genes 0.000 description 1
- 108010074328 Interferon-gamma Proteins 0.000 description 1
- 102000013462 Interleukin-12 Human genes 0.000 description 1
- 108010065805 Interleukin-12 Proteins 0.000 description 1
- 108010002350 Interleukin-2 Proteins 0.000 description 1
- 102000000588 Interleukin-2 Human genes 0.000 description 1
- 108010063738 Interleukins Proteins 0.000 description 1
- 102000015696 Interleukins Human genes 0.000 description 1
- LPHGQDQBBGAPDZ-UHFFFAOYSA-N Isocaffeine Natural products CN1C(=O)N(C)C(=O)C2=C1N(C)C=N2 LPHGQDQBBGAPDZ-UHFFFAOYSA-N 0.000 description 1
- SHGAZHPCJJPHSC-NUEINMDLSA-N Isotretinoin Chemical compound OC(=O)C=C(C)/C=C/C=C(C)C=CC1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-NUEINMDLSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 1
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 1
- XUIIKFGFIJCVMT-LBPRGKRZSA-N L-thyroxine Chemical compound IC1=CC(C[C@H]([NH3+])C([O-])=O)=CC(I)=C1OC1=CC(I)=C(O)C(I)=C1 XUIIKFGFIJCVMT-LBPRGKRZSA-N 0.000 description 1
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 1
- 239000005411 L01XE02 - Gefitinib Substances 0.000 description 1
- 239000005551 L01XE03 - Erlotinib Substances 0.000 description 1
- 229920001491 Lentinan Polymers 0.000 description 1
- HLFSDGLLUJUHTE-SNVBAGLBSA-N Levamisole Chemical compound C1([C@H]2CN3CCSC3=N2)=CC=CC=C1 HLFSDGLLUJUHTE-SNVBAGLBSA-N 0.000 description 1
- 241000408529 Libra Species 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- GQYIWUVLTXOXAJ-UHFFFAOYSA-N Lomustine Chemical compound ClCCN(N=O)C(=O)NC1CCCCC1 GQYIWUVLTXOXAJ-UHFFFAOYSA-N 0.000 description 1
- 206010025323 Lymphomas Diseases 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- 229940124761 MMP inhibitor Drugs 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- XOGTZOOQQBDUSI-UHFFFAOYSA-M Mesna Chemical compound [Na+].[O-]S(=O)(=O)CCS XOGTZOOQQBDUSI-UHFFFAOYSA-M 0.000 description 1
- FQISKWAFAHGMGT-SGJOWKDISA-M Methylprednisolone sodium succinate Chemical compound [Na+].C([C@@]12C)=CC(=O)C=C1[C@@H](C)C[C@@H]1[C@@H]2[C@@H](O)C[C@]2(C)[C@@](O)(C(=O)COC(=O)CCC([O-])=O)CC[C@H]21 FQISKWAFAHGMGT-SGJOWKDISA-M 0.000 description 1
- GCKMFJBGXUYNAG-HLXURNFRSA-N Methyltestosterone Chemical compound C1CC2=CC(=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@](C)(O)[C@@]1(C)CC2 GCKMFJBGXUYNAG-HLXURNFRSA-N 0.000 description 1
- 102100025725 Mothers against decapentaplegic homolog 4 Human genes 0.000 description 1
- 101710143112 Mothers against decapentaplegic homolog 4 Proteins 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 101100519207 Mus musculus Pdcd1 gene Proteins 0.000 description 1
- NUGPIZCTELGDOS-QHCPKHFHSA-N N-[(1S)-3-[4-(3-methyl-5-propan-2-yl-1,2,4-triazol-4-yl)piperidin-1-yl]-1-pyridin-3-ylpropyl]cyclopentanecarboxamide Chemical compound C(C)(C)C1=NN=C(N1C1CCN(CC1)CC[C@@H](C=1C=NC=CC=1)NC(=O)C1CCCC1)C NUGPIZCTELGDOS-QHCPKHFHSA-N 0.000 description 1
- LKJPYSCBVHEWIU-UHFFFAOYSA-N N-[4-cyano-3-(trifluoromethyl)phenyl]-3-[(4-fluorophenyl)sulfonyl]-2-hydroxy-2-methylpropanamide Chemical compound C=1C=C(C#N)C(C(F)(F)F)=CC=1NC(=O)C(O)(C)CS(=O)(=O)C1=CC=C(F)C=C1 LKJPYSCBVHEWIU-UHFFFAOYSA-N 0.000 description 1
- UEEJHVSXFDXPFK-UHFFFAOYSA-N N-dimethylaminoethanol Chemical compound CN(C)CCO UEEJHVSXFDXPFK-UHFFFAOYSA-N 0.000 description 1
- HTLZVHNRZJPSMI-UHFFFAOYSA-N N-ethylpiperidine Chemical compound CCN1CCCCC1 HTLZVHNRZJPSMI-UHFFFAOYSA-N 0.000 description 1
- MBBZMMPHUWSWHV-BDVNFPICSA-N N-methylglucamine Chemical compound CNC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO MBBZMMPHUWSWHV-BDVNFPICSA-N 0.000 description 1
- 108010021717 Nafarelin Proteins 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 206010030155 Oesophageal carcinoma Diseases 0.000 description 1
- 206010033128 Ovarian cancer Diseases 0.000 description 1
- 206010061535 Ovarian neoplasm Diseases 0.000 description 1
- 239000012828 PI3K inhibitor Substances 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 206010061902 Pancreatic neoplasm Diseases 0.000 description 1
- 241000282373 Panthera pardus Species 0.000 description 1
- 108010020346 Polyglutamic Acid Proteins 0.000 description 1
- HFVNWDWLWUCIHC-GUPDPFMOSA-N Prednimustine Chemical compound O=C([C@@]1(O)CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)[C@@H](O)C[C@@]21C)COC(=O)CCCC1=CC=C(N(CCCl)CCCl)C=C1 HFVNWDWLWUCIHC-GUPDPFMOSA-N 0.000 description 1
- 102100033237 Pro-epidermal growth factor Human genes 0.000 description 1
- 229940079156 Proteasome inhibitor Drugs 0.000 description 1
- 108010090931 Proto-Oncogene Proteins c-bcl-2 Proteins 0.000 description 1
- 102000013535 Proto-Oncogene Proteins c-bcl-2 Human genes 0.000 description 1
- 102000016971 Proto-Oncogene Proteins c-kit Human genes 0.000 description 1
- 108010014608 Proto-Oncogene Proteins c-kit Proteins 0.000 description 1
- 208000015634 Rectal Neoplasms Diseases 0.000 description 1
- 229940125907 SJ995973 Drugs 0.000 description 1
- 229940124639 Selective inhibitor Drugs 0.000 description 1
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 1
- 208000000453 Skin Neoplasms Diseases 0.000 description 1
- 108700032504 Smad2 Proteins 0.000 description 1
- 206010041067 Small cell lung cancer Diseases 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 208000005718 Stomach Neoplasms Diseases 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 210000001744 T-lymphocyte Anatomy 0.000 description 1
- 108700011582 TER 286 Proteins 0.000 description 1
- DRHKJLXJIQTDTD-OAHLLOKOSA-N Tamsulosine Chemical compound CCOC1=CC=CC=C1OCCN[C@H](C)CC1=CC=C(OC)C(S(N)(=O)=O)=C1 DRHKJLXJIQTDTD-OAHLLOKOSA-N 0.000 description 1
- NAVMQTYZDKMPEU-UHFFFAOYSA-N Targretin Chemical compound CC1=CC(C(CCC2(C)C)(C)C)=C2C=C1C(=C)C1=CC=C(C(O)=O)C=C1 NAVMQTYZDKMPEU-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- BPEGJWRSRHCHSN-UHFFFAOYSA-N Temozolomide Chemical compound O=C1N(C)N=NC2=C(C(N)=O)N=CN21 BPEGJWRSRHCHSN-UHFFFAOYSA-N 0.000 description 1
- CBPNZQVSJQDFBE-FUXHJELOSA-N Temsirolimus Chemical compound C1C[C@@H](OC(=O)C(C)(CO)CO)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 CBPNZQVSJQDFBE-FUXHJELOSA-N 0.000 description 1
- PDMMFKSKQVNJMI-BLQWBTBKSA-N Testosterone propionate Chemical compound C1CC2=CC(=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H](OC(=O)CC)[C@@]1(C)CC2 PDMMFKSKQVNJMI-BLQWBTBKSA-N 0.000 description 1
- FOCVUCIESVLUNU-UHFFFAOYSA-N Thiotepa Chemical compound C1CN1P(N1CC1)(=S)N1CC1 FOCVUCIESVLUNU-UHFFFAOYSA-N 0.000 description 1
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 1
- 239000004473 Threonine Substances 0.000 description 1
- 108010046075 Thymosin Proteins 0.000 description 1
- 102000007501 Thymosin Human genes 0.000 description 1
- 102000011923 Thyrotropin Human genes 0.000 description 1
- 108010061174 Thyrotropin Proteins 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- IVTVGDXNLFLDRM-HNNXBMFYSA-N Tomudex Chemical compound C=1C=C2NC(C)=NC(=O)C2=CC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)S1 IVTVGDXNLFLDRM-HNNXBMFYSA-N 0.000 description 1
- 102000000887 Transcription factor STAT Human genes 0.000 description 1
- 108050007918 Transcription factor STAT Proteins 0.000 description 1
- 208000006593 Urologic Neoplasms Diseases 0.000 description 1
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 description 1
- 108091008605 VEGF receptors Proteins 0.000 description 1
- 102000005789 Vascular Endothelial Growth Factors Human genes 0.000 description 1
- 108010019530 Vascular Endothelial Growth Factors Proteins 0.000 description 1
- 102100033177 Vascular endothelial growth factor receptor 2 Human genes 0.000 description 1
- JXLYSJRDGCGARV-WWYNWVTFSA-N Vinblastine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@](O)(CC)C[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C JXLYSJRDGCGARV-WWYNWVTFSA-N 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- ONSQLDCEJIIUJS-XVFCMESISA-N [(2r,3s,4r,5r)-5-(2-amino-4-oxopyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methyl dihydrogen phosphate Chemical compound NC1=NC(=O)C=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](COP(O)(O)=O)O1 ONSQLDCEJIIUJS-XVFCMESISA-N 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- GLRAHDCHUZLKKC-UHFFFAOYSA-N acetonitrile;2,2,2-trifluoroacetic acid;hydrate Chemical compound O.CC#N.OC(=O)C(F)(F)F GLRAHDCHUZLKKC-UHFFFAOYSA-N 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- DUYNJNWVGIWJRI-LJAQVGFWSA-N acolbifene Chemical compound C1=CC([C@H]2C(=C(C3=CC=C(O)C=C3O2)C)C=2C=CC(O)=CC=2)=CC=C1OCCN1CCCCC1 DUYNJNWVGIWJRI-LJAQVGFWSA-N 0.000 description 1
- 229950002421 acolbifene Drugs 0.000 description 1
- RJURFGZVJUQBHK-IIXSONLDSA-N actinomycin D Chemical compound C[C@H]1OC(=O)[C@H](C(C)C)N(C)C(=O)CN(C)C(=O)[C@@H]2CCCN2C(=O)[C@@H](C(C)C)NC(=O)[C@H]1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)N[C@@H]4C(=O)N[C@@H](C(N5CCC[C@H]5C(=O)N(C)CC(=O)N(C)[C@@H](C(C)C)C(=O)O[C@@H]4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-IIXSONLDSA-N 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 108700025316 aldesleukin Proteins 0.000 description 1
- 229960005310 aldesleukin Drugs 0.000 description 1
- 229940062527 alendronate Drugs 0.000 description 1
- MBMBGCFOFBJSGT-KUBAVDMBSA-N all-cis-docosa-4,7,10,13,16,19-hexaenoic acid Chemical compound CC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CCC(O)=O MBMBGCFOFBJSGT-KUBAVDMBSA-N 0.000 description 1
- SHGAZHPCJJPHSC-YCNIQYBTSA-N all-trans-retinoic acid Chemical compound OC(=O)\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-YCNIQYBTSA-N 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 229960000473 altretamine Drugs 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 229940024606 amino acid Drugs 0.000 description 1
- 235000001014 amino acid Nutrition 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 229960003896 aminopterin Drugs 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-N ammonia Natural products N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 1
- 229960002550 amrubicin Drugs 0.000 description 1
- VJZITPJGSQKZMX-XDPRQOKASA-N amrubicin Chemical compound O([C@H]1C[C@](CC2=C(O)C=3C(=O)C4=CC=CC=C4C(=O)C=3C(O)=C21)(N)C(=O)C)[C@H]1C[C@H](O)[C@H](O)CO1 VJZITPJGSQKZMX-XDPRQOKASA-N 0.000 description 1
- 229960002932 anastrozole Drugs 0.000 description 1
- YBBLVLTVTVSKRW-UHFFFAOYSA-N anastrozole Chemical compound N#CC(C)(C)C1=CC(C(C)(C#N)C)=CC(CN2N=CN=C2)=C1 YBBLVLTVTVSKRW-UHFFFAOYSA-N 0.000 description 1
- 239000004037 angiogenesis inhibitor Substances 0.000 description 1
- 229940121369 angiogenesis inhibitor Drugs 0.000 description 1
- 125000000129 anionic group Chemical group 0.000 description 1
- 230000001093 anti-cancer Effects 0.000 description 1
- 230000001028 anti-proliverative effect Effects 0.000 description 1
- 230000005904 anticancer immunity Effects 0.000 description 1
- 229940045988 antineoplastic drug protein kinase inhibitors Drugs 0.000 description 1
- 230000005975 antitumor immune response Effects 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 229960001372 aprepitant Drugs 0.000 description 1
- ATALOFNDEOCMKK-OITMNORJSA-N aprepitant Chemical compound O([C@@H]([C@@H]1C=2C=CC(F)=CC=2)O[C@H](C)C=2C=C(C=C(C=2)C(F)(F)F)C(F)(F)F)CCN1CC1=NNC(=O)N1 ATALOFNDEOCMKK-OITMNORJSA-N 0.000 description 1
- 229940115115 aranesp Drugs 0.000 description 1
- UVJYAKBJSGRTHA-ZCRGAIPPSA-N arglabin Chemical compound C1C[C@H]2C(=C)C(=O)O[C@@H]2[C@@H]2C(C)=CC[C@]32O[C@]31C UVJYAKBJSGRTHA-ZCRGAIPPSA-N 0.000 description 1
- UVJYAKBJSGRTHA-UHFFFAOYSA-N arglabin Natural products C1CC2C(=C)C(=O)OC2C2C(C)=CCC32OC31C UVJYAKBJSGRTHA-UHFFFAOYSA-N 0.000 description 1
- 229910052785 arsenic Inorganic materials 0.000 description 1
- 229940091658 arsenic Drugs 0.000 description 1
- RQNWIZPPADIBDY-UHFFFAOYSA-N arsenic atom Chemical compound [As] RQNWIZPPADIBDY-UHFFFAOYSA-N 0.000 description 1
- GOLCXWYRSKYTSP-UHFFFAOYSA-N arsenic trioxide Inorganic materials O1[As]2O[As]1O2 GOLCXWYRSKYTSP-UHFFFAOYSA-N 0.000 description 1
- 229960002594 arsenic trioxide Drugs 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- GJMNAFGEUJBOCE-MEQIQULJSA-N asoprisnil Chemical compound C1([C@@H]2C3=C4CCC(=O)C=C4CC[C@H]3[C@@H]3CC[C@]([C@]3(C2)C)(COC)OC)=CC=C(\C=N\O)C=C1 GJMNAFGEUJBOCE-MEQIQULJSA-N 0.000 description 1
- 229950003620 asoprisnil Drugs 0.000 description 1
- 229940120638 avastin Drugs 0.000 description 1
- 229960002756 azacitidine Drugs 0.000 description 1
- KLNFSAOEKUDMFA-UHFFFAOYSA-N azanide;2-hydroxyacetic acid;platinum(2+) Chemical compound [NH2-].[NH2-].[Pt+2].OCC(O)=O KLNFSAOEKUDMFA-UHFFFAOYSA-N 0.000 description 1
- VSRXQHXAPYXROS-UHFFFAOYSA-N azanide;cyclobutane-1,1-dicarboxylic acid;platinum(2+) Chemical compound [NH2-].[NH2-].[Pt+2].OC(=O)C1(C(O)=O)CCC1 VSRXQHXAPYXROS-UHFFFAOYSA-N 0.000 description 1
- 229960002170 azathioprine Drugs 0.000 description 1
- LMEKQMALGUDUQG-UHFFFAOYSA-N azathioprine Chemical compound CN1C=NC([N+]([O-])=O)=C1SC1=NC=NC2=C1NC=N2 LMEKQMALGUDUQG-UHFFFAOYSA-N 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- JUHORIMYRDESRB-UHFFFAOYSA-N benzathine Chemical compound C=1C=CC=CC=1CNCCNCC1=CC=CC=C1 JUHORIMYRDESRB-UHFFFAOYSA-N 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 229960004365 benzoic acid Drugs 0.000 description 1
- DRTQHJPVMGBUCF-PSQAKQOGSA-N beta-L-uridine Natural products O[C@H]1[C@@H](O)[C@H](CO)O[C@@H]1N1C(=O)NC(=O)C=C1 DRTQHJPVMGBUCF-PSQAKQOGSA-N 0.000 description 1
- 229940064804 betadine Drugs 0.000 description 1
- 229960003237 betaine Drugs 0.000 description 1
- 229960004648 betamethasone acetate Drugs 0.000 description 1
- AKUJBENLRBOFTD-QZIXMDIESA-N betamethasone acetate Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)COC(C)=O)(O)[C@@]1(C)C[C@@H]2O AKUJBENLRBOFTD-QZIXMDIESA-N 0.000 description 1
- 229960005354 betamethasone sodium phosphate Drugs 0.000 description 1
- PLCQGRYPOISRTQ-LWCNAHDDSA-L betamethasone sodium phosphate Chemical compound [Na+].[Na+].C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)COP([O-])([O-])=O)(O)[C@@]1(C)C[C@@H]2O PLCQGRYPOISRTQ-LWCNAHDDSA-L 0.000 description 1
- 229960002938 bexarotene Drugs 0.000 description 1
- 208000026900 bile duct neoplasm Diseases 0.000 description 1
- 229960004395 bleomycin sulfate Drugs 0.000 description 1
- RJTANRZEWTUVMA-UHFFFAOYSA-N boron;n-methylmethanamine Chemical compound [B].CNC RJTANRZEWTUVMA-UHFFFAOYSA-N 0.000 description 1
- GXJABQQUPOEUTA-RDJZCZTQSA-N bortezomib Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)B(O)O)NC(=O)C=1N=CC=NC=1)C1=CC=CC=C1 GXJABQQUPOEUTA-RDJZCZTQSA-N 0.000 description 1
- 229960001467 bortezomib Drugs 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 229960002092 busulfan Drugs 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- OSVHLUXLWQLPIY-KBAYOESNSA-N butyl 2-[(6aR,9R,10aR)-1-hydroxy-9-(hydroxymethyl)-6,6-dimethyl-6a,7,8,9,10,10a-hexahydrobenzo[c]chromen-3-yl]-2-methylpropanoate Chemical compound C(CCC)OC(C(C)(C)C1=CC(=C2[C@H]3[C@H](C(OC2=C1)(C)C)CC[C@H](C3)CO)O)=O OSVHLUXLWQLPIY-KBAYOESNSA-N 0.000 description 1
- 229960001948 caffeine Drugs 0.000 description 1
- VJEONQKOZGKCAK-UHFFFAOYSA-N caffeine Natural products CN1C(=O)N(C)C(=O)C2=C1C=CN2C VJEONQKOZGKCAK-UHFFFAOYSA-N 0.000 description 1
- BBBFJLBPOGFECG-VJVYQDLKSA-N calcitonin Chemical compound N([C@H](C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H]([C@@H](C)O)C(=O)N1[C@@H](CCC1)C(N)=O)C(C)C)C(=O)[C@@H]1CSSC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1 BBBFJLBPOGFECG-VJVYQDLKSA-N 0.000 description 1
- 229960004015 calcitonin Drugs 0.000 description 1
- GMRQFYUYWCNGIN-NKMMMXOESA-N calcitriol Chemical compound C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@@H](CCCC(C)(C)O)C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C GMRQFYUYWCNGIN-NKMMMXOESA-N 0.000 description 1
- KVUAALJSMIVURS-ZEDZUCNESA-L calcium folinate Chemical compound [Ca+2].C1NC=2NC(N)=NC(=O)C=2N(C=O)C1CNC1=CC=C(C(=O)N[C@@H](CCC([O-])=O)C([O-])=O)C=C1 KVUAALJSMIVURS-ZEDZUCNESA-L 0.000 description 1
- KVUAALJSMIVURS-QNTKWALQSA-L calcium;(2s)-2-[[4-[[(6s)-2-amino-5-formyl-4-oxo-1,6,7,8-tetrahydropteridin-6-yl]methylamino]benzoyl]amino]pentanedioate Chemical class [Ca+2].C([C@@H]1N(C=O)C=2C(=O)N=C(NC=2NC1)N)NC1=CC=C(C(=O)N[C@@H](CCC([O-])=O)C([O-])=O)C=C1 KVUAALJSMIVURS-QNTKWALQSA-L 0.000 description 1
- 229960004117 capecitabine Drugs 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 229960004562 carboplatin Drugs 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 229940097647 casodex Drugs 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 229940047495 celebrex Drugs 0.000 description 1
- RZEKVGVHFLEQIL-UHFFFAOYSA-N celecoxib Chemical compound C1=CC(C)=CC=C1C1=CC(C(F)(F)F)=NN1C1=CC=C(S(N)(=O)=O)C=C1 RZEKVGVHFLEQIL-UHFFFAOYSA-N 0.000 description 1
- 230000012820 cell cycle checkpoint Effects 0.000 description 1
- 239000013592 cell lysate Substances 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 201000010881 cervical cancer Diseases 0.000 description 1
- 229960005395 cetuximab Drugs 0.000 description 1
- 229960004630 chlorambucil Drugs 0.000 description 1
- JCKYGMPEJWAADB-UHFFFAOYSA-N chlorambucil Chemical compound OC(=O)CCCC1=CC=C(N(CCCl)CCCl)C=C1 JCKYGMPEJWAADB-UHFFFAOYSA-N 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 208000006990 cholangiocarcinoma Diseases 0.000 description 1
- 229960001231 choline Drugs 0.000 description 1
- OEYIOHPDSNJKLS-UHFFFAOYSA-N choline Chemical compound C[N+](C)(C)CCO OEYIOHPDSNJKLS-UHFFFAOYSA-N 0.000 description 1
- 229960004316 cisplatin Drugs 0.000 description 1
- DQLATGHUWYMOKM-UHFFFAOYSA-L cisplatin Chemical compound N[Pt](N)(Cl)Cl DQLATGHUWYMOKM-UHFFFAOYSA-L 0.000 description 1
- 235000015165 citric acid Nutrition 0.000 description 1
- ACSIXWWBWUQEHA-UHFFFAOYSA-N clodronic acid Chemical compound OP(O)(=O)C(Cl)(Cl)P(O)(O)=O ACSIXWWBWUQEHA-UHFFFAOYSA-N 0.000 description 1
- 229960002286 clodronic acid Drugs 0.000 description 1
- 208000029742 colonic neoplasm Diseases 0.000 description 1
- 229940125773 compound 10 Drugs 0.000 description 1
- 229940125796 compound 3d Drugs 0.000 description 1
- 229940125872 compound 4d Drugs 0.000 description 1
- 229940126115 compound 4f Drugs 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 229940111134 coxibs Drugs 0.000 description 1
- 229940043378 cyclin-dependent kinase inhibitor Drugs 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 239000003255 cyclooxygenase 2 inhibitor Substances 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 229960004397 cyclophosphamide Drugs 0.000 description 1
- UWFYSQMTEOIJJG-FDTZYFLXSA-N cyproterone acetate Chemical compound C1=C(Cl)C2=CC(=O)[C@@H]3C[C@@H]3[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@@](C(C)=O)(OC(=O)C)[C@@]1(C)CC2 UWFYSQMTEOIJJG-FDTZYFLXSA-N 0.000 description 1
- 229960000978 cyproterone acetate Drugs 0.000 description 1
- 229960000684 cytarabine Drugs 0.000 description 1
- 239000000824 cytostatic agent Substances 0.000 description 1
- 230000001085 cytostatic effect Effects 0.000 description 1
- 229960003901 dacarbazine Drugs 0.000 description 1
- 229960000640 dactinomycin Drugs 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 229960003603 decitabine Drugs 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 108700025485 deslorelin Proteins 0.000 description 1
- 229960005408 deslorelin Drugs 0.000 description 1
- 229960003957 dexamethasone Drugs 0.000 description 1
- UREBDLICKHMUKA-CXSFZGCWSA-N dexamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-CXSFZGCWSA-N 0.000 description 1
- 229960004833 dexamethasone phosphate Drugs 0.000 description 1
- VQODGRNSFPNSQE-CXSFZGCWSA-N dexamethasone phosphate Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)COP(O)(O)=O)(O)[C@@]1(C)C[C@@H]2O VQODGRNSFPNSQE-CXSFZGCWSA-N 0.000 description 1
- 125000004663 dialkyl amino group Chemical group 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- HPNMFZURTQLUMO-UHFFFAOYSA-N diethylamine Chemical compound CCNCC HPNMFZURTQLUMO-UHFFFAOYSA-N 0.000 description 1
- RGLYKWWBQGJZGM-ISLYRVAYSA-N diethylstilbestrol Chemical compound C=1C=C(O)C=CC=1C(/CC)=C(\CC)C1=CC=C(O)C=C1 RGLYKWWBQGJZGM-ISLYRVAYSA-N 0.000 description 1
- 229960000452 diethylstilbestrol Drugs 0.000 description 1
- 229940063123 diflucan Drugs 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 239000003534 dna topoisomerase inhibitor Substances 0.000 description 1
- 229960003668 docetaxel Drugs 0.000 description 1
- 229940000640 docosahexaenoate Drugs 0.000 description 1
- 238000001647 drug administration Methods 0.000 description 1
- 239000000890 drug combination Substances 0.000 description 1
- 239000002359 drug metabolite Substances 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 229960004199 dutasteride Drugs 0.000 description 1
- JWJOTENAMICLJG-QWBYCMEYSA-N dutasteride Chemical compound O=C([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)N[C@@H]4CC3)C)CC[C@@]21C)NC1=CC(C(F)(F)F)=CC=C1C(F)(F)F JWJOTENAMICLJG-QWBYCMEYSA-N 0.000 description 1
- 229950001287 edotecarin Drugs 0.000 description 1
- 229940121647 egfr inhibitor Drugs 0.000 description 1
- 229940088598 enzyme Drugs 0.000 description 1
- 229960001904 epirubicin Drugs 0.000 description 1
- 229960003265 epirubicin hydrochloride Drugs 0.000 description 1
- 229960003388 epoetin alfa Drugs 0.000 description 1
- AAKJLRGGTJKAMG-UHFFFAOYSA-N erlotinib Chemical compound C=12C=C(OCCOC)C(OCCOC)=CC2=NC=NC=1NC1=CC=CC(C#C)=C1 AAKJLRGGTJKAMG-UHFFFAOYSA-N 0.000 description 1
- 229940105423 erythropoietin Drugs 0.000 description 1
- 201000004101 esophageal cancer Diseases 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- IIUMCNJTGSMNRO-VVSKJQCTSA-L estramustine sodium phosphate Chemical compound [Na+].[Na+].ClCCN(CCCl)C(=O)OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)OP([O-])([O-])=O)[C@@H]4[C@@H]3CCC2=C1 IIUMCNJTGSMNRO-VVSKJQCTSA-L 0.000 description 1
- 229940011871 estrogen Drugs 0.000 description 1
- 239000000262 estrogen Substances 0.000 description 1
- 239000002834 estrogen receptor modulator Substances 0.000 description 1
- VUCAHVBMSFIGAI-ZFINNJDLSA-M estrone sodium sulfate Chemical compound [Na+].[O-]S(=O)(=O)OC1=CC=C2[C@H]3CC[C@](C)(C(CC4)=O)[C@@H]4[C@@H]3CCC2=C1 VUCAHVBMSFIGAI-ZFINNJDLSA-M 0.000 description 1
- AFAXGSQYZLGZPG-UHFFFAOYSA-N ethanedisulfonic acid Chemical compound OS(=O)(=O)CCS(O)(=O)=O AFAXGSQYZLGZPG-UHFFFAOYSA-N 0.000 description 1
- 229960002568 ethinylestradiol Drugs 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- YWKVMGDEOUPQGN-UHFFFAOYSA-N ethyl 2-[4-[2-(3-hydroxy-1-azabicyclo[2.2.2]octan-3-yl)ethynyl]phenyl]acetate Chemical compound C1=CC(CC(=O)OCC)=CC=C1C#CC1(O)C(CC2)CCN2C1 YWKVMGDEOUPQGN-UHFFFAOYSA-N 0.000 description 1
- VCYZVXRKYPKDQB-UHFFFAOYSA-N ethyl 2-fluoroacetate Chemical compound CCOC(=O)CF VCYZVXRKYPKDQB-UHFFFAOYSA-N 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 229940009626 etidronate Drugs 0.000 description 1
- VJJPUSNTGOMMGY-MRVIYFEKSA-N etoposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@H](C)OC[C@H]4O3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 VJJPUSNTGOMMGY-MRVIYFEKSA-N 0.000 description 1
- 229960005420 etoposide Drugs 0.000 description 1
- 229950011548 fadrozole Drugs 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 229950003662 fenretinide Drugs 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 229960004039 finasteride Drugs 0.000 description 1
- DBEPLOCGEIEOCV-WSBQPABSSA-N finasteride Chemical compound N([C@@H]1CC2)C(=O)C=C[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H](C(=O)NC(C)(C)C)[C@@]2(C)CC1 DBEPLOCGEIEOCV-WSBQPABSSA-N 0.000 description 1
- 229960000961 floxuridine Drugs 0.000 description 1
- ODKNJVUHOIMIIZ-RRKCRQDMSA-N floxuridine Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(F)=C1 ODKNJVUHOIMIIZ-RRKCRQDMSA-N 0.000 description 1
- 229960004884 fluconazole Drugs 0.000 description 1
- 229960000390 fludarabine Drugs 0.000 description 1
- GIUYCYHIANZCFB-FJFJXFQQSA-N fludarabine phosphate Chemical compound C1=NC=2C(N)=NC(F)=NC=2N1[C@@H]1O[C@H](COP(O)(O)=O)[C@@H](O)[C@@H]1O GIUYCYHIANZCFB-FJFJXFQQSA-N 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 229960002949 fluorouracil Drugs 0.000 description 1
- 229960001751 fluoxymesterone Drugs 0.000 description 1
- YLRFCQOZQXIBAB-RBZZARIASA-N fluoxymesterone Chemical compound C1CC2=CC(=O)CC[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1CC[C@](C)(O)[C@@]1(C)C[C@@H]2O YLRFCQOZQXIBAB-RBZZARIASA-N 0.000 description 1
- 229960002074 flutamide Drugs 0.000 description 1
- MKXKFYHWDHIYRV-UHFFFAOYSA-N flutamide Chemical compound CC(C)C(=O)NC1=CC=C([N+]([O-])=O)C(C(F)(F)F)=C1 MKXKFYHWDHIYRV-UHFFFAOYSA-N 0.000 description 1
- 235000008191 folinic acid Nutrition 0.000 description 1
- 239000011672 folinic acid Substances 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 229960002258 fulvestrant Drugs 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 108010074605 gamma-Globulins Proteins 0.000 description 1
- 229940083124 ganglion-blocking antiadrenergic secondary and tertiary amines Drugs 0.000 description 1
- 206010017758 gastric cancer Diseases 0.000 description 1
- XGALLCVXEZPNRQ-UHFFFAOYSA-N gefitinib Chemical compound C=12C=C(OCCCN3CCOCC3)C(OC)=CC2=NC=NC=1NC1=CC=C(F)C(Cl)=C1 XGALLCVXEZPNRQ-UHFFFAOYSA-N 0.000 description 1
- 229960005277 gemcitabine Drugs 0.000 description 1
- SDUQYLNIPVEERB-QPPQHZFASA-N gemcitabine Chemical compound O=C1N=C(N)C=CN1[C@H]1C(F)(F)[C@H](O)[C@@H](CO)O1 SDUQYLNIPVEERB-QPPQHZFASA-N 0.000 description 1
- 229960000578 gemtuzumab Drugs 0.000 description 1
- 230000030279 gene silencing Effects 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 229960002989 glutamic acid Drugs 0.000 description 1
- 229960002913 goserelin Drugs 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 229940093915 gynecological organic acid Drugs 0.000 description 1
- 125000005842 heteroatom Chemical group 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- VKYKSIONXSXAKP-UHFFFAOYSA-N hexamethylenetetramine Chemical compound C1N(C2)CN3CN1CN2C3 VKYKSIONXSXAKP-UHFFFAOYSA-N 0.000 description 1
- UUVWYPNAQBNQJQ-UHFFFAOYSA-N hexamethylmelamine Chemical compound CN(C)C1=NC(N(C)C)=NC(N(C)C)=N1 UUVWYPNAQBNQJQ-UHFFFAOYSA-N 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 229960004931 histamine dihydrochloride Drugs 0.000 description 1
- PPZMYIBUHIPZOS-UHFFFAOYSA-N histamine dihydrochloride Chemical compound Cl.Cl.NCCC1=CN=CN1 PPZMYIBUHIPZOS-UHFFFAOYSA-N 0.000 description 1
- 102000044890 human EPO Human genes 0.000 description 1
- 229960000890 hydrocortisone Drugs 0.000 description 1
- 239000000017 hydrogel Substances 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 235000004867 hydroxocobalamin Nutrition 0.000 description 1
- 239000011704 hydroxocobalamin Substances 0.000 description 1
- 229960001103 hydroxocobalamin Drugs 0.000 description 1
- 239000002471 hydroxymethylglutaryl coenzyme A reductase inhibitor Substances 0.000 description 1
- 230000003463 hyperproliferative effect Effects 0.000 description 1
- 229960005236 ibandronic acid Drugs 0.000 description 1
- 229960000908 idarubicin Drugs 0.000 description 1
- 229960001101 ifosfamide Drugs 0.000 description 1
- HOMGKSMUEGBAAB-UHFFFAOYSA-N ifosfamide Chemical compound ClCCNP1(=O)OCCCN1CCCl HOMGKSMUEGBAAB-UHFFFAOYSA-N 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 239000007943 implant Substances 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 239000000411 inducer Substances 0.000 description 1
- 229940090044 injection Drugs 0.000 description 1
- 102000006495 integrins Human genes 0.000 description 1
- 108010044426 integrins Proteins 0.000 description 1
- 229960003130 interferon gamma Drugs 0.000 description 1
- 229940117681 interleukin-12 Drugs 0.000 description 1
- 239000007928 intraperitoneal injection Substances 0.000 description 1
- 229940065638 intron a Drugs 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 238000004255 ion exchange chromatography Methods 0.000 description 1
- 239000003456 ion exchange resin Substances 0.000 description 1
- 229920003303 ion-exchange polymer Polymers 0.000 description 1
- 229940084651 iressa Drugs 0.000 description 1
- UWKQSNNFCGGAFS-XIFFEERXSA-N irinotecan Chemical compound C1=C2C(CC)=C3CN(C(C4=C([C@@](C(=O)OC4)(O)CC)C=4)=O)C=4C3=NC2=CC=C1OC(=O)N(CC1)CCC1N1CCCCC1 UWKQSNNFCGGAFS-XIFFEERXSA-N 0.000 description 1
- 229960004768 irinotecan Drugs 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 229940045996 isethionic acid Drugs 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- JJWLVOIRVHMVIS-UHFFFAOYSA-N isopropylamine Chemical compound CC(C)N JJWLVOIRVHMVIS-UHFFFAOYSA-N 0.000 description 1
- 229960005280 isotretinoin Drugs 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- 229960002014 ixabepilone Drugs 0.000 description 1
- FABUFPQFXZVHFB-CFWQTKTJSA-N ixabepilone Chemical compound C/C([C@@H]1C[C@@H]2O[C@]2(C)CCC[C@@H]([C@@H]([C@H](C)C(=O)C(C)(C)[C@H](O)CC(=O)N1)O)C)=C\C1=CSC(C)=N1 FABUFPQFXZVHFB-CFWQTKTJSA-N 0.000 description 1
- ZLVXBBHTMQJRSX-VMGNSXQWSA-N jdtic Chemical compound C1([C@]2(C)CCN(C[C@@H]2C)C[C@H](C(C)C)NC(=O)[C@@H]2NCC3=CC(O)=CC=C3C2)=CC=CC(O)=C1 ZLVXBBHTMQJRSX-VMGNSXQWSA-N 0.000 description 1
- 108010045069 keyhole-limpet hemocyanin Proteins 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 108010021336 lanreotide Proteins 0.000 description 1
- 229960002437 lanreotide Drugs 0.000 description 1
- GOTYRUGSSMKFNF-UHFFFAOYSA-N lenalidomide Chemical compound C1C=2C(N)=CC=CC=2C(=O)N1C1CCC(=O)NC1=O GOTYRUGSSMKFNF-UHFFFAOYSA-N 0.000 description 1
- 229940115286 lentinan Drugs 0.000 description 1
- 229960003881 letrozole Drugs 0.000 description 1
- HPJKCIUCZWXJDR-UHFFFAOYSA-N letrozole Chemical compound C1=CC(C#N)=CC=C1C(N1N=CN=C1)C1=CC=C(C#N)C=C1 HPJKCIUCZWXJDR-UHFFFAOYSA-N 0.000 description 1
- 229960001691 leucovorin Drugs 0.000 description 1
- 208000032839 leukemia Diseases 0.000 description 1
- 229960001614 levamisole Drugs 0.000 description 1
- 229950008325 levothyroxine Drugs 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 229960002247 lomustine Drugs 0.000 description 1
- 229960003538 lonidamine Drugs 0.000 description 1
- WDRYRZXSPDWGEB-UHFFFAOYSA-N lonidamine Chemical compound C12=CC=CC=C2C(C(=O)O)=NN1CC1=CC=C(Cl)C=C1Cl WDRYRZXSPDWGEB-UHFFFAOYSA-N 0.000 description 1
- 108010078259 luprolide acetate gel depot Proteins 0.000 description 1
- 230000002934 lysing effect Effects 0.000 description 1
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 description 1
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 229960004961 mechlorethamine Drugs 0.000 description 1
- HAWPXGHAZFHHAD-UHFFFAOYSA-N mechlorethamine Chemical class ClCCN(C)CCCl HAWPXGHAZFHHAD-UHFFFAOYSA-N 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- PSGAAPLEWMOORI-PEINSRQWSA-N medroxyprogesterone acetate Chemical compound C([C@@]12C)CC(=O)C=C1[C@@H](C)C[C@@H]1[C@@H]2CC[C@]2(C)[C@@](OC(C)=O)(C(C)=O)CC[C@H]21 PSGAAPLEWMOORI-PEINSRQWSA-N 0.000 description 1
- 229960002985 medroxyprogesterone acetate Drugs 0.000 description 1
- RQZAXGRLVPAYTJ-GQFGMJRRSA-N megestrol acetate Chemical compound C1=C(C)C2=CC(=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@@](C(C)=O)(OC(=O)C)[C@@]1(C)CC2 RQZAXGRLVPAYTJ-GQFGMJRRSA-N 0.000 description 1
- 229960004296 megestrol acetate Drugs 0.000 description 1
- SGDBTWWWUNNDEQ-LBPRGKRZSA-N melphalan Chemical compound OC(=O)[C@@H](N)CC1=CC=C(N(CCCl)CCCl)C=C1 SGDBTWWWUNNDEQ-LBPRGKRZSA-N 0.000 description 1
- 229960001924 melphalan Drugs 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 229960001428 mercaptopurine Drugs 0.000 description 1
- 229960004635 mesna Drugs 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 239000002207 metabolite Substances 0.000 description 1
- 230000001394 metastastic effect Effects 0.000 description 1
- 206010061289 metastatic neoplasm Diseases 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- 229930182817 methionine Natural products 0.000 description 1
- 229960004452 methionine Drugs 0.000 description 1
- GCBCWTWQAFLKJG-SBSPUUFOSA-N methyl (2r)-2-amino-3-(4-chlorophenyl)propanoate;hydrochloride Chemical compound Cl.COC(=O)[C@H](N)CC1=CC=C(Cl)C=C1 GCBCWTWQAFLKJG-SBSPUUFOSA-N 0.000 description 1
- SWVMLNPDTIFDDY-SBSPUUFOSA-N methyl (2r)-2-amino-3-phenylpropanoate;hydrochloride Chemical compound Cl.COC(=O)[C@H](N)CC1=CC=CC=C1 SWVMLNPDTIFDDY-SBSPUUFOSA-N 0.000 description 1
- SWVMLNPDTIFDDY-FVGYRXGTSA-N methyl (2s)-2-amino-3-phenylpropanoate;hydrochloride Chemical compound Cl.COC(=O)[C@@H](N)CC1=CC=CC=C1 SWVMLNPDTIFDDY-FVGYRXGTSA-N 0.000 description 1
- ZXUQEPZWVQIOJE-UHFFFAOYSA-N methyl 2-chloro-2-oxoacetate Chemical compound COC(=O)C(Cl)=O ZXUQEPZWVQIOJE-UHFFFAOYSA-N 0.000 description 1
- YUUAYBAIHCDHHD-UHFFFAOYSA-N methyl 5-aminolevulinate Chemical compound COC(=O)CCC(=O)CN YUUAYBAIHCDHHD-UHFFFAOYSA-N 0.000 description 1
- MWZPENIJLUWBSY-VIFPVBQESA-N methyl L-tyrosinate Chemical compound COC(=O)[C@@H](N)CC1=CC=C(O)C=C1 MWZPENIJLUWBSY-VIFPVBQESA-N 0.000 description 1
- 229960005033 methyl aminolevulinate Drugs 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- JEWJRMKHSMTXPP-BYFNXCQMSA-M methylcobalamin Chemical compound C[Co+]N([C@]1([H])[C@H](CC(N)=O)[C@]\2(CCC(=O)NC[C@H](C)OP(O)(=O)OC3[C@H]([C@H](O[C@@H]3CO)N3C4=CC(C)=C(C)C=C4N=C3)O)C)C/2=C(C)\C([C@H](C/2(C)C)CCC(N)=O)=N\C\2=C\C([C@H]([C@@]/2(CC(N)=O)C)CCC(N)=O)=N\C\2=C(C)/C2=N[C@]1(C)[C@@](C)(CC(N)=O)[C@@H]2CCC(N)=O JEWJRMKHSMTXPP-BYFNXCQMSA-M 0.000 description 1
- 235000007672 methylcobalamin Nutrition 0.000 description 1
- 239000011585 methylcobalamin Substances 0.000 description 1
- 229960004584 methylprednisolone Drugs 0.000 description 1
- 229960001566 methyltestosterone Drugs 0.000 description 1
- 229960005225 mifamurtide Drugs 0.000 description 1
- JMUHBNWAORSSBD-WKYWBUFDSA-N mifamurtide Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@@H](OC(=O)CCCCCCCCCCCCCCC)COP(O)(=O)OCCNC(=O)[C@H](C)NC(=O)CC[C@H](C(N)=O)NC(=O)[C@H](C)NC(=O)[C@@H](C)O[C@H]1[C@H](O)[C@@H](CO)OC(O)[C@@H]1NC(C)=O JMUHBNWAORSSBD-WKYWBUFDSA-N 0.000 description 1
- PQLXHQMOHUQAKB-UHFFFAOYSA-N miltefosine Chemical compound CCCCCCCCCCCCCCCCOP([O-])(=O)OCC[N+](C)(C)C PQLXHQMOHUQAKB-UHFFFAOYSA-N 0.000 description 1
- 229960003775 miltefosine Drugs 0.000 description 1
- 239000002829 mitogen activated protein kinase inhibitor Substances 0.000 description 1
- 229960004857 mitomycin Drugs 0.000 description 1
- 229960000350 mitotane Drugs 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 125000002950 monocyclic group Chemical group 0.000 description 1
- 125000002757 morpholinyl group Chemical group 0.000 description 1
- LNOPIUAQISRISI-UHFFFAOYSA-N n'-hydroxy-2-propan-2-ylsulfonylethanimidamide Chemical compound CC(C)S(=O)(=O)CC(N)=NO LNOPIUAQISRISI-UHFFFAOYSA-N 0.000 description 1
- XZMHJYWMCRQSSI-UHFFFAOYSA-N n-[5-[2-(3-acetylanilino)-1,3-thiazol-4-yl]-4-methyl-1,3-thiazol-2-yl]benzamide Chemical compound CC(=O)C1=CC=CC(NC=2SC=C(N=2)C2=C(N=C(NC(=O)C=3C=CC=CC=3)S2)C)=C1 XZMHJYWMCRQSSI-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- RWHUEXWOYVBUCI-ITQXDASVSA-N nafarelin Chemical compound C([C@@H](C(=O)N[C@H](CC=1C=C2C=CC=CC2=CC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N1[C@@H](CCC1)C(=O)NCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H]1NC(=O)CC1)C1=CC=C(O)C=C1 RWHUEXWOYVBUCI-ITQXDASVSA-N 0.000 description 1
- 229960002333 nafarelin Drugs 0.000 description 1
- 229950007221 nedaplatin Drugs 0.000 description 1
- 229940029345 neupogen Drugs 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- XWXYUMMDTVBTOU-UHFFFAOYSA-N nilutamide Chemical compound O=C1C(C)(C)NC(=O)N1C1=CC=C([N+]([O-])=O)C(C(F)(F)F)=C1 XWXYUMMDTVBTOU-UHFFFAOYSA-N 0.000 description 1
- 229960002653 nilutamide Drugs 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- 208000002154 non-small cell lung carcinoma Diseases 0.000 description 1
- 229960000770 ondansetron hydrochloride Drugs 0.000 description 1
- 229940100688 oral solution Drugs 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 239000012044 organic layer Substances 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- DWAFYCQODLXJNR-BNTLRKBRSA-L oxaliplatin Chemical compound O1C(=O)C(=O)O[Pt]11N[C@@H]2CCCC[C@H]2N1 DWAFYCQODLXJNR-BNTLRKBRSA-L 0.000 description 1
- 229960001756 oxaliplatin Drugs 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- OLDRWYVIKMSFFB-SSPJITILSA-N palonosetron hydrochloride Chemical compound Cl.C1N(CC2)CCC2[C@@H]1N1C(=O)C(C=CC=C2CCC3)=C2[C@H]3C1 OLDRWYVIKMSFFB-SSPJITILSA-N 0.000 description 1
- 229960003359 palonosetron hydrochloride Drugs 0.000 description 1
- WLJNZVDCPSBLRP-UHFFFAOYSA-N pamoic acid Chemical compound C1=CC=C2C(CC=3C4=CC=CC=C4C=C(C=3O)C(=O)O)=C(O)C(C(O)=O)=CC2=C1 WLJNZVDCPSBLRP-UHFFFAOYSA-N 0.000 description 1
- 201000002528 pancreatic cancer Diseases 0.000 description 1
- 208000008443 pancreatic carcinoma Diseases 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 229960002340 pentostatin Drugs 0.000 description 1
- FPVKHBSQESCIEP-JQCXWYLXSA-N pentostatin Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(N=CNC[C@H]2O)=C2N=C1 FPVKHBSQESCIEP-JQCXWYLXSA-N 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 239000008363 phosphate buffer Substances 0.000 description 1
- 229940043441 phosphoinositide 3-kinase inhibitor Drugs 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- 229940096701 plain lipid modifying drug hmg coa reductase inhibitors Drugs 0.000 description 1
- 230000004983 pleiotropic effect Effects 0.000 description 1
- 239000002798 polar solvent Substances 0.000 description 1
- 229920000768 polyamine Polymers 0.000 description 1
- 125000003367 polycyclic group Chemical group 0.000 description 1
- 229920002643 polyglutamic acid Polymers 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- OXCMYAYHXIHQOA-UHFFFAOYSA-N potassium;[2-butyl-5-chloro-3-[[4-[2-(1,2,4-triaza-3-azanidacyclopenta-1,4-dien-5-yl)phenyl]phenyl]methyl]imidazol-4-yl]methanol Chemical compound [K+].CCCCC1=NC(Cl)=C(CO)N1CC1=CC=C(C=2C(=CC=CC=2)C2=N[N-]N=N2)C=C1 OXCMYAYHXIHQOA-UHFFFAOYSA-N 0.000 description 1
- 229960004694 prednimustine Drugs 0.000 description 1
- 229940063238 premarin Drugs 0.000 description 1
- 150000003141 primary amines Chemical class 0.000 description 1
- MFDFERRIHVXMIY-UHFFFAOYSA-N procaine Chemical compound CCN(CC)CCOC(=O)C1=CC=C(N)C=C1 MFDFERRIHVXMIY-UHFFFAOYSA-N 0.000 description 1
- 229960004919 procaine Drugs 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 239000003207 proteasome inhibitor Substances 0.000 description 1
- 239000003197 protein kinase B inhibitor Substances 0.000 description 1
- 239000003909 protein kinase inhibitor Substances 0.000 description 1
- 229940124272 protein stabilizer Drugs 0.000 description 1
- 235000018102 proteins Nutrition 0.000 description 1
- 230000000722 protumoral effect Effects 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 229960004622 raloxifene Drugs 0.000 description 1
- GZUITABIAKMVPG-UHFFFAOYSA-N raloxifene Chemical compound C1=CC(O)=CC=C1C1=C(C(=O)C=2C=CC(OCCN3CCCCC3)=CC=2)C2=CC=C(O)C=C2S1 GZUITABIAKMVPG-UHFFFAOYSA-N 0.000 description 1
- 229960004432 raltitrexed Drugs 0.000 description 1
- 229960000424 rasburicase Drugs 0.000 description 1
- 108010084837 rasburicase Proteins 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 206010038038 rectal cancer Diseases 0.000 description 1
- 201000001275 rectum cancer Diseases 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- WUAPFZMCVAUBPE-IGMARMGPSA-N rhenium-186 Chemical compound [186Re] WUAPFZMCVAUBPE-IGMARMGPSA-N 0.000 description 1
- 125000006413 ring segment Chemical group 0.000 description 1
- 229960004641 rituximab Drugs 0.000 description 1
- 239000003419 rna directed dna polymerase inhibitor Substances 0.000 description 1
- 229960004889 salicylic acid Drugs 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 239000000849 selective androgen receptor modulator Substances 0.000 description 1
- 229960003440 semustine Drugs 0.000 description 1
- 230000011664 signaling Effects 0.000 description 1
- 201000000849 skin cancer Diseases 0.000 description 1
- 208000000587 small cell lung carcinoma Diseases 0.000 description 1
- 229940126586 small molecule drug Drugs 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 229960003787 sorafenib Drugs 0.000 description 1
- 238000009168 stem cell therapy Methods 0.000 description 1
- 238000009580 stem-cell therapy Methods 0.000 description 1
- 201000011549 stomach cancer Diseases 0.000 description 1
- 229960001052 streptozocin Drugs 0.000 description 1
- ZSJLQEPLLKMAKR-GKHCUFPYSA-N streptozocin Chemical compound O=NN(C)C(=O)N[C@H]1[C@@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O ZSJLQEPLLKMAKR-GKHCUFPYSA-N 0.000 description 1
- 229910052712 strontium Inorganic materials 0.000 description 1
- CIOAGBVUUVVLOB-UHFFFAOYSA-N strontium atom Chemical compound [Sr] CIOAGBVUUVVLOB-UHFFFAOYSA-N 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 229950000244 sulfanilic acid Drugs 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 229960002613 tamsulosin Drugs 0.000 description 1
- 229940120982 tarceva Drugs 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 229940063683 taxotere Drugs 0.000 description 1
- 229960004964 temozolomide Drugs 0.000 description 1
- 229960000235 temsirolimus Drugs 0.000 description 1
- NRUKOCRGYNPUPR-QBPJDGROSA-N teniposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@@H](OC[C@H]4O3)C=3SC=CC=3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 NRUKOCRGYNPUPR-QBPJDGROSA-N 0.000 description 1
- 229960001278 teniposide Drugs 0.000 description 1
- BFNYNEMRWHFIMR-UHFFFAOYSA-N tert-butyl 2-cyanoacetate Chemical compound CC(C)(C)OC(=O)CC#N BFNYNEMRWHFIMR-UHFFFAOYSA-N 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229960001712 testosterone propionate Drugs 0.000 description 1
- 229960004559 theobromine Drugs 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 229960001196 thiotepa Drugs 0.000 description 1
- LCJVIYPJPCBWKS-NXPQJCNCSA-N thymosin Chemical compound SC[C@@H](N)C(=O)N[C@H](CO)C(=O)N[C@H](CC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](C)C(=O)N[C@H](C(C)C)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](C(C)C)C(=O)N[C@H](CO)C(=O)N[C@H](CO)C(=O)N[C@H](CCC(O)=O)C(=O)N[C@H]([C@@H](C)CC)C(=O)N[C@H]([C@H](C)O)C(=O)N[C@H](C(C)C)C(=O)N[C@H](CCCCN)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N[C@H](CCC(O)=O)C(=O)N[C@H](CCCCN)C(=O)N[C@H](CCCCN)C(=O)N[C@H](CCC(O)=O)C(=O)N[C@H](C(C)C)C(=O)N[C@H](C(C)C)C(=O)N[C@H](CCC(O)=O)C(=O)N[C@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@H](CCC(O)=O)C(O)=O LCJVIYPJPCBWKS-NXPQJCNCSA-N 0.000 description 1
- 229960000874 thyrotropin Drugs 0.000 description 1
- 230000001748 thyrotropin Effects 0.000 description 1
- XUIIKFGFIJCVMT-UHFFFAOYSA-N thyroxine-binding globulin Natural products IC1=CC(CC([NH3+])C([O-])=O)=CC(I)=C1OC1=CC(I)=C(O)C(I)=C1 XUIIKFGFIJCVMT-UHFFFAOYSA-N 0.000 description 1
- 229940111100 tice bcg Drugs 0.000 description 1
- 229960003087 tioguanine Drugs 0.000 description 1
- PLHJCIYEEKOWNM-HHHXNRCGSA-N tipifarnib Chemical compound CN1C=NC=C1[C@](N)(C=1C=C2C(C=3C=C(Cl)C=CC=3)=CC(=O)N(C)C2=CC=1)C1=CC=C(Cl)C=C1 PLHJCIYEEKOWNM-HHHXNRCGSA-N 0.000 description 1
- 229950009158 tipifarnib Drugs 0.000 description 1
- 229950002376 tirapazamine Drugs 0.000 description 1
- ORYDPOVDJJZGHQ-UHFFFAOYSA-N tirapazamine Chemical compound C1=CC=CC2=[N+]([O-])C(N)=N[N+]([O-])=C21 ORYDPOVDJJZGHQ-UHFFFAOYSA-N 0.000 description 1
- 229940044693 topoisomerase inhibitor Drugs 0.000 description 1
- UCFGDBYHRUNTLO-QHCPKHFHSA-N topotecan Chemical compound C1=C(O)C(CN(C)C)=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 UCFGDBYHRUNTLO-QHCPKHFHSA-N 0.000 description 1
- 229960000303 topotecan Drugs 0.000 description 1
- 238000013518 transcription Methods 0.000 description 1
- 230000035897 transcription Effects 0.000 description 1
- 239000003558 transferase inhibitor Substances 0.000 description 1
- 229960000575 trastuzumab Drugs 0.000 description 1
- 229960001727 tretinoin Drugs 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- KVJXBPDAXMEYOA-CXANFOAXSA-N trilostane Chemical compound OC1=C(C#N)C[C@]2(C)[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CC[C@@]32O[C@@H]31 KVJXBPDAXMEYOA-CXANFOAXSA-N 0.000 description 1
- 229960001670 trilostane Drugs 0.000 description 1
- NOYPYLRCIDNJJB-UHFFFAOYSA-N trimetrexate Chemical compound COC1=C(OC)C(OC)=CC(NCC=2C(=C3C(N)=NC(N)=NC3=CC=2)C)=C1 NOYPYLRCIDNJJB-UHFFFAOYSA-N 0.000 description 1
- 229960001099 trimetrexate Drugs 0.000 description 1
- YFTHZRPMJXBUME-UHFFFAOYSA-N tripropylamine Chemical compound CCCN(CCC)CCC YFTHZRPMJXBUME-UHFFFAOYSA-N 0.000 description 1
- 229960004824 triptorelin Drugs 0.000 description 1
- VXKHXGOKWPXYNA-PGBVPBMZSA-N triptorelin Chemical compound C([C@@H](C(=O)N[C@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1[C@@H](CCC1)C(=O)NCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)C1=CC=C(O)C=C1 VXKHXGOKWPXYNA-PGBVPBMZSA-N 0.000 description 1
- 229960000294 triptorelin pamoate Drugs 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 229960000281 trometamol Drugs 0.000 description 1
- 230000004565 tumor cell growth Effects 0.000 description 1
- 230000004614 tumor growth Effects 0.000 description 1
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 description 1
- 229960004441 tyrosine Drugs 0.000 description 1
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 1
- 241000701161 unidentified adenovirus Species 0.000 description 1
- DRTQHJPVMGBUCF-UHFFFAOYSA-N uracil arabinoside Natural products OC1C(O)C(CO)OC1N1C(=O)NC(=O)C=C1 DRTQHJPVMGBUCF-UHFFFAOYSA-N 0.000 description 1
- 229940045145 uridine Drugs 0.000 description 1
- AUFUWRKPQLGTGF-FMKGYKFTSA-N uridine triacetate Chemical compound CC(=O)O[C@@H]1[C@H](OC(C)=O)[C@@H](COC(=O)C)O[C@H]1N1C(=O)NC(=O)C=C1 AUFUWRKPQLGTGF-FMKGYKFTSA-N 0.000 description 1
- 238000001291 vacuum drying Methods 0.000 description 1
- 229940005605 valeric acid Drugs 0.000 description 1
- 229960000653 valrubicin Drugs 0.000 description 1
- ZOCKGBMQLCSHFP-KQRAQHLDSA-N valrubicin Chemical compound O([C@H]1C[C@](CC2=C(O)C=3C(=O)C4=CC=CC(OC)=C4C(=O)C=3C(O)=C21)(O)C(=O)COC(=O)CCCC)[C@H]1C[C@H](NC(=O)C(F)(F)F)[C@H](O)[C@H](C)O1 ZOCKGBMQLCSHFP-KQRAQHLDSA-N 0.000 description 1
- YCOYDOIWSSHVCK-UHFFFAOYSA-N vatalanib Chemical compound C1=CC(Cl)=CC=C1NC(C1=CC=CC=C11)=NN=C1CC1=CC=NC=C1 YCOYDOIWSSHVCK-UHFFFAOYSA-N 0.000 description 1
- 229950000578 vatalanib Drugs 0.000 description 1
- ZQFGRJWRSLZCSQ-ZSFNYQMMSA-N verteporfin Chemical compound C=1C([C@@]2([C@H](C(=O)OC)C(=CC=C22)C(=O)OC)C)=NC2=CC(C(=C2C=C)C)=NC2=CC(C(=C2CCC(O)=O)C)=NC2=CC2=NC=1C(C)=C2CCC(=O)OC ZQFGRJWRSLZCSQ-ZSFNYQMMSA-N 0.000 description 1
- 229960003895 verteporfin Drugs 0.000 description 1
- 229960003048 vinblastine Drugs 0.000 description 1
- JXLYSJRDGCGARV-XQKSVPLYSA-N vincaleukoblastine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 JXLYSJRDGCGARV-XQKSVPLYSA-N 0.000 description 1
- 229960004528 vincristine Drugs 0.000 description 1
- OGWKCGZFUXNPDA-UHFFFAOYSA-N vincristine Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(OC(C)=O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-UHFFFAOYSA-N 0.000 description 1
- NMDYYWFGPIMTKO-HBVLKOHWSA-N vinflunine Chemical compound C([C@@](C1=C(C2=CC=CC=C2N1)C1)(C2=C(OC)C=C3N(C)[C@@H]4[C@@]5(C3=C2)CCN2CC=C[C@]([C@@H]52)([C@H]([C@]4(O)C(=O)OC)OC(C)=O)CC)C(=O)OC)[C@H]2C[C@@H](C(C)(F)F)CN1C2 NMDYYWFGPIMTKO-HBVLKOHWSA-N 0.000 description 1
- 229960000922 vinflunine Drugs 0.000 description 1
- GBABOYUKABKIAF-GHYRFKGUSA-N vinorelbine Chemical compound C1N(CC=2C3=CC=CC=C3NC=22)CC(CC)=C[C@H]1C[C@]2(C(=O)OC)C1=CC([C@]23[C@H]([C@]([C@H](OC(C)=O)[C@]4(CC)C=CCN([C@H]34)CC2)(O)C(=O)OC)N2C)=C2C=C1OC GBABOYUKABKIAF-GHYRFKGUSA-N 0.000 description 1
- 229960002066 vinorelbine Drugs 0.000 description 1
- 238000001262 western blot Methods 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- ZAFYATHCZYHLPB-UHFFFAOYSA-N zolpidem Chemical compound N1=C2C=CC(C)=CN2C(CC(=O)N(C)C)=C1C1=CC=C(C)C=C1 ZAFYATHCZYHLPB-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/06—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
本发明提供了具有式(Ⅰ)所示结构的氮杂吲哚类化合物及其应用。该氮杂吲哚类化合物可以有效抑制SMAD3‑磷酸化,并且在大鼠LCC诱导的肿瘤模型中具有良好的抗肿瘤活性,并且,与化合物SIS3相比,部分本发明的氮杂吲哚类化合物显着改善了水溶性。本发明的氮杂吲哚类化合物有希望成为防治肿瘤的有效药物。
The present invention provides azaindole compounds with the structure represented by formula (I) and applications thereof. The azaindole compounds can effectively inhibit SMAD3-phosphorylation, and have good antitumor activity in the rat LCC-induced tumor model, and compared with the compound SIS3, some of the azaindole compounds of the present invention Significantly improved water solubility. The azaindole compounds of the present invention are expected to be effective medicines for preventing and treating tumors.
Description
技术领域technical field
本发明涉及药物化学技术领域,具体涉及一种氮杂吲哚类化合物及其应用。The invention relates to the technical field of medicinal chemistry, in particular to an azaindole compound and an application thereof.
背景技术Background technique
肿瘤的生长和转移是癌症患者死亡的主要原因。在肿瘤的侵袭和转移步骤中,转化生长因子β(TGF-β)发挥促癌作用,由于TGF-β的多效性作用和复杂的功能,其抑制作用可能导致副作用,而TGFβ信号的下游介质SMAD3由于含有DNA结合结构域,可直接与靶基因中的启动子结合调节转录,但SMAD2和SMAD4并不具备这些功能。此外,SMAD3具有调节在肿瘤免疫疗法中关键靶标蛋白表达的能力,例如,SMAD3可以上调T细胞上程序性死亡受体1(PD-1)的表达,但SMAD2不能上调PD-1的表达;靶标蛋白SMAD3的沉默可以通过增强肿瘤微环境中NK细胞和嗜中性白细胞的抗肿瘤活性来抑制肿瘤细胞的生长和转移。因此,开发有效低毒的药物来选择性抑制SMAD3蛋白是一种有潜力的抗肿瘤策略。Tumor growth and metastasis are the leading causes of death in cancer patients. During the invasive and metastatic steps of tumors, transforming growth factor beta (TGF-β) exerts tumor-promoting effects, and its inhibitory effect may lead to side effects due to the pleiotropic effects and complex functions of TGF-β, while downstream mediators of TGFβ signaling Because SMAD3 contains a DNA-binding domain, it can directly bind to promoters in target genes to regulate transcription, but SMAD2 and SMAD4 do not have these functions. In addition, SMAD3 has the ability to regulate the expression of key target proteins in tumor immunotherapy, for example, SMAD3 can upregulate the expression of programmed death receptor 1 (PD-1) on T cells, but SMAD2 cannot upregulate the expression of PD-1; the target Silencing of the protein SMAD3 can inhibit tumor cell growth and metastasis by enhancing the antitumor activity of NK cells and neutrophils in the tumor microenvironment. Therefore, developing effective and low-toxic drugs to selectively inhibit SMAD3 protein is a potential anti-tumor strategy.
选择性靶向抑制SMAD3的小分子化合物仍然非常少,因此,发现更多类型的SMAD3蛋白选择性抑制剂具有重要意义。There are still very few small-molecule compounds that selectively target and inhibit SMAD3, so it is of great significance to discover more types of SMAD3 protein-selective inhibitors.
发明内容SUMMARY OF THE INVENTION
基于此,本发明提供了一类新的氮杂吲哚类化合物,该类化合物能够选择性抑制SMAD3-磷酸化,但不抑制SMAD2,并能够通过提高肿瘤微环境中的NK细胞的增殖而提高作用,此类化合物有潜力成为肿瘤免疫治疗的小分子药物。Based on this, the present invention provides a new class of azaindole compounds, which can selectively inhibit SMAD3-phosphorylation, but not SMAD2, and can increase the proliferation of NK cells in the tumor microenvironment by increasing the These compounds have the potential to become small-molecule drugs for tumor immunotherapy.
具技术方案如下:The technical solutions are as follows:
具有式(Ⅰ)所示结构的氮杂吲哚类化合物或者其药学上可接受的盐或者其立体异构体或者其前药分子:Azaindoles having the structure represented by formula (I) or their pharmaceutically acceptable salts or their stereoisomers or their prodrug molecules:
其中:in:
D选自:C、N;并且当D为N时,R2不存在;D is selected from: C, N; and when D is N, R is absent ;
A,B分别独立地选自:N、CR3;A, B are independently selected from: N, CR 3 ;
R1选自:H、C1-C6烷基、C6-C10芳基、C3-C8环烷基、卤素、CN;R 1 is selected from: H, C 1 -C 6 alkyl, C 6 -C 10 aryl, C 3 -C 8 cycloalkyl, halogen, CN;
R2选自:H、C1-C6烷基;R 2 is selected from: H, C 1 -C 6 alkyl;
各R3分别独立地选自:H、C1-C6烷基、卤素、CN;Each R 3 is independently selected from: H, C 1 -C 6 alkyl, halogen, CN;
R4选自: R 4 is selected from:
R5、各R5a和R5b分别独立地选自:H、卤素、CN、C1-C6烷基、C3-C8环烷基;R 5 , each R 5a and R 5b are each independently selected from: H, halogen, CN, C 1 -C 6 alkyl, C 3 -C 8 cycloalkyl;
各R6分别独立地选自: Each R is independently selected from:
R7选自:H、C1-C6烷基、一个或多个R13取代的C6-C10芳基、一个或多个R13取代的6-10元杂芳基;R 7 is selected from: H, C 1 -C 6 alkyl, C 6 -C 10 aryl substituted with one or more R 13 , 6-10-membered heteroaryl substituted with one or more R 13 ;
各R8分别独立地选自:H、卤素、CN、C1-C6烷基、C3-C8环烷基、羟基、C1-C6烷氧基、C1-C6烷氧基取代的C1-C6烷氧基;Each R 8 is independently selected from: H, halogen, CN, C 1 -C 6 alkyl, C 3 -C 8 cycloalkyl, hydroxy, C 1 -C 6 alkoxy, C 1 -C 6 alkoxy substituted C 1 -C 6 alkoxy;
各R9分别独立地选自:H、卤素、CN、C1-C6烷基、C3-C8环烷基、羟基、C1-C6烷氧基、C1-C6烷氧基取代的C1-C6烷氧基;Each R 9 is independently selected from: H, halogen, CN, C 1 -C 6 alkyl, C 3 -C 8 cycloalkyl, hydroxy, C 1 -C 6 alkoxy, C 1 -C 6 alkoxy substituted C 1 -C 6 alkoxy;
R10选自:H、C1-C6烷基;R 10 is selected from: H, C 1 -C 6 alkyl;
R11选自:羟基、C1-C6烷氧基、-N(R14)(R15);R 11 is selected from: hydroxyl, C 1 -C 6 alkoxy, -N(R 14 )(R 15 );
R12选自:H、C1-C6烷基、一个或多个R13取代的C6-C10芳基、一个或多个R13取代的6-10元杂芳基;R 12 is selected from: H, C 1 -C 6 alkyl, one or more R 13 substituted C 6 -C 10 aryl, one or more R 13 substituted 6-10-membered heteroaryl;
各R13分别独立地选自:H、卤素、CN、C1-C6烷基、C3-C8环烷基、羟基、C1-C6烷氧基、C1-C6烷氧基取代的C1-C6烷氧基;Each R 13 is independently selected from: H, halogen, CN, C 1 -C 6 alkyl, C 3 -C 8 cycloalkyl, hydroxy, C 1 -C 6 alkoxy, C 1 -C 6 alkoxy substituted C 1 -C 6 alkoxy;
R14和R15分别独立地选自:H、C1-C6烷基、氨基取代的C1-C6烷基、C1-C6烷胺基取代的C1-C6烷基;R 14 and R 15 are each independently selected from: H, C 1 -C 6 alkyl, amino substituted C 1 -C 6 alkyl, C 1 -C 6 alkylamino substituted C 1 -C 6 alkyl;
n选自:1、2、3、4;n is selected from: 1, 2, 3, 4;
m选自:0、1、2、3、4;m is selected from: 0, 1, 2, 3, 4;
p选自:0-7之间的整数;p is selected from: an integer between 0-7;
q选自:1、2、3、4;q is selected from: 1, 2, 3, 4;
y选自:1、2、3、4、5;y is selected from: 1, 2, 3, 4, 5;
并且,当D为C,A为N,B为CR3,R1为苯基,R2为时,R8不为C1-C6烷氧基。And, when D is C, A is N, B is CR 3 , R 1 is phenyl, and R 2 is , R 8 is not C 1 -C 6 alkoxy.
在其中一些实施例中,D为C,A为N,B为CR4。In some of these embodiments, D is C, A is N, and B is CR4.
在其中一些实施例中,所述氮杂吲哚类化合物具有式(II)所示结构:In some of these embodiments, the azaindoles have the structure shown in formula (II):
在其中一些实施例中,R5为H。 In some of these embodiments, R5 is H.
在其中一些实施例中,n选自:1、2;m选自:1或2。In some of these embodiments, n is selected from: 1, 2; m is selected from: 1 or 2.
在其中一些实施例中,R7选自:一个或多个R13取代的苯基、一个或多个R13取代的六元含氮杂芳基。In some of these embodiments, R 7 is selected from: phenyl substituted with one or more R 13 , six-membered nitrogen-containing heteroaryl substituted with one or more R 13 .
在其中一些实施例中,各R13分别独立地选自:H、C1-C6烷基、C1-C6烷氧基。In some of these embodiments, each R 13 is independently selected from: H, C 1 -C 6 alkyl, C 1 -C 6 alkoxy.
在其中一些实施例中,R7选自: In some of these embodiments, R 7 is selected from:
在其中一些实施例中,所述氮杂吲哚类化合物具有式(III)所示结构:In some embodiments, the azaindoles have the structure shown in formula (III):
在其中一些实施例中,所述氮杂吲哚类化合物具有式(IV)所示结构:In some of these embodiments, the azaindole compound has the structure shown in formula (IV):
在其中一些实施例中,各R8分别独立地选自:H、羟基、C1-C3烷氧基、C1-C3烷氧基取代的C1-C3烷氧基。In some of these embodiments, each R 8 is independently selected from the group consisting of: H, hydroxy, C 1 -C 3 alkoxy, C 1 -C 3 alkoxy substituted C 1 -C 3 alkoxy.
在其中一些实施例中,各R8分别独立地选自:羟基、甲氧基、甲氧基取代的乙氧基。In some of these embodiments, each R 8 is independently selected from the group consisting of: hydroxy, methoxy, methoxy substituted ethoxy.
在其中一些实施例中,R8均为羟基。In some of these embodiments, R 8 is both hydroxyl.
在其中一些实施例中,所述氮杂吲哚类化合物具有式(V)所示结构:In some of these embodiments, the azaindoles have the structure shown in formula (V):
在其中一些实施例中,R9选自:H、卤素、羟基;In some of these embodiments, R 9 is selected from: H, halogen, hydroxyl;
R10选自:H、C1-C3烷基;R 10 is selected from: H, C 1 -C 3 alkyl;
R11选自:C1-C6烷氧基。R 11 is selected from: C 1 -C 6 alkoxy.
在其中一些实施例中,所述氮杂吲哚类化合物具有式(VI)所示结构:In some of these embodiments, the azaindole compounds have the structure shown in formula (VI):
在其中一些实施例中,R12选自:一个或多个R13取代的C6-C10杂芳基;各R13分别独立地选自:H、C1-C6烷基。In some of these embodiments, R 12 is selected from: C 6 -C 10 heteroaryl substituted with one or more R 13 ; each R 13 is independently selected from: H, C 1 -C 6 alkyl.
在其中一些实施例中,R12选自: In some of these embodiments, R 12 is selected from:
在其中一些实施例中,R5a和R5b分别独立地选自:H、卤素。In some of these embodiments, R 5a and R 5b are each independently selected from the group consisting of: H, halogen.
在其中一些实施例中,R5b为H,R5a为F。In some of these embodiments, R 5b is H and R 5a is F.
在其中一些实施例中,R1选自:H、C1-C3烷基、C6-C10芳基、C3-C6环烷基。In some of these embodiments, R 1 is selected from the group consisting of: H, C 1 -C 3 alkyl, C 6 -C 10 aryl, C 3 -C 6 cycloalkyl.
在其中一些实施例中,R1选自:H、苯基、环丙基。In some of these embodiments, R 1 is selected from the group consisting of: H, phenyl, cyclopropyl.
在其中一些实施例中,R2选自:C1-C3烷基。In some of these embodiments, R 2 is selected from: C 1 -C 3 alkyl.
在其中一些实施例中,R3为H。In some of these embodiments, R3 is H.
本发明还提供了上述的氮杂吲哚类化合物或者其药学上可接受的盐或者其立体异构体或者其前药分子的应用。The present invention also provides the use of the above-mentioned azaindole compounds or their pharmaceutically acceptable salts or their stereoisomers or their prodrug molecules.
具体技术方案如下:The specific technical solutions are as follows:
上述的氮杂吲哚类化合物或者其药学上可接受的盐或者其立体异构体或者其前药分子在制备SMAD3抑制剂中的应用。Use of the above-mentioned azaindole compounds or their pharmaceutically acceptable salts or their stereoisomers or their prodrug molecules in the preparation of SMAD3 inhibitors.
上述的氮杂吲哚类化合物或者其药学上可接受的盐或者其立体异构体或者其前药分子在制备预防和/或治疗肿瘤的药物中的应用。Use of the above-mentioned azaindole compounds or their pharmaceutically acceptable salts or their stereoisomers or their prodrug molecules in the preparation of medicaments for preventing and/or treating tumors.
在其中一些实施例中,所述肿瘤为:肺癌、黑色素瘤、乳腺癌、肝癌。In some of these embodiments, the tumor is: lung cancer, melanoma, breast cancer, liver cancer.
本发明还提供了一种预防和/或治疗肿瘤的药用组合物。The present invention also provides a pharmaceutical composition for preventing and/or treating tumors.
具体技术方案如下:The specific technical solutions are as follows:
一种预防和/或治疗肿瘤的药物组合物,包括活性成分以及药学上可接受的载体,所述活性成分包括有上述的氮杂吲哚类化合物或者其药学上可接受的盐或者其立体异构体或者其前药分子。A pharmaceutical composition for preventing and/or treating tumors, comprising an active ingredient and a pharmaceutically acceptable carrier, the active ingredient comprising the above-mentioned azaindole compound or a pharmaceutically acceptable salt thereof or a stereoisomer thereof Construct or its prodrug molecule.
本发明研究发现了一系列新的氮杂吲哚类化合物,其可以有效地选择性地抑制SMAD3-磷酸化,但不抑制SMAD2,并且在大鼠LCC诱导的肿瘤模型中具有良好的抗肿瘤活性,并能够通过提高肿瘤微环境中的NK细胞的增殖而提高抗肿瘤作用。The present study found a series of new azaindole compounds, which can effectively and selectively inhibit SMAD3-phosphorylation, but not SMAD2, and have good antitumor activity in a rat LCC-induced tumor model , and can enhance the anti-tumor effect by increasing the proliferation of NK cells in the tumor microenvironment.
另外,药物化合物的水溶性是影响药物制剂开发的一大原因,如果化合物的水溶性差可能会导致其生物利用度低,代谢不稳定(DrugDiscov.Today 2004,9,1020-1028),体内吸收的分数波动大(J.Med.Chem.2007,50,5858-5862),以及导致后期阶段的制剂开发困难等问题(Adv.DrugDeliv.Rev.1997,23,3-25),这些往往会使药物开发的时间大大延长。本发明的氮杂吲哚类化合物或其盐具有较好的水溶性,部分化合物的HCl盐在水中的溶解度超过20mg/mL,甚至达到94mg/mL或更高,这有利于提高化合物的生物利用度,并且有利于其后期的制剂开发。本发明的氮杂吲哚类化合物有希望成为防治肿瘤的有效药物。In addition, the water solubility of pharmaceutical compounds is a major factor affecting the development of pharmaceutical preparations. Poor water solubility of the compounds may lead to low bioavailability, unstable metabolism (DrugDiscov.Today 2004, 9, 1020-1028), and the absorption of Fractional fluctuations (J.Med.Chem.2007,50,5858-5862), and problems such as difficulty in formulation development at later stages (Adv.DrugDeliv.Rev.1997,23,3-25), tend to make drugs The development time is greatly extended. The azaindole compounds or their salts of the present invention have good water solubility, and the solubility of HCl salts of some compounds in water exceeds 20 mg/mL, and even reaches 94 mg/mL or higher, which is beneficial to improve the bioavailability of the compounds degree, and is conducive to its later formulation development. The azaindole compounds of the present invention are expected to be effective medicines for preventing and treating tumors.
附图说明Description of drawings
图1为化合物16d选择性抑制TGF-β依赖性的SMAD3磷酸化结果图。Figure 1 is a graph showing the results of
图2为化合物16d有效抑制小鼠同源Lewis肺癌(LLC)的生长结果图。Figure 2 is a graph showing the results of
图3为化合物16d通过增强LLC模型小鼠中的NK细胞聚集来抑制癌症进展的结果图。Figure 3 is a graph showing the results of
具体实施方式Detailed ways
本发明所述化合物中,当任何变量(例如R等)在任何组分中出现超过一次,则其每次出现的定义独立于其它每次出现的定义。同样,允许取代基及变量的组合,只要这种组合使化合物稳定。自取代基划入环系统的线表示所指的键可连接到任何能取代的环原子上。如果环系统为多环,其意味着这种键仅连接到邻近环的任何适当的碳原子上。要理解本领域普通技术人员可选择本发明化合物的取代基及取代型式而提供化学上稳定的并可通过本领域技术和下列提出的方法自可容易获得的原料容易的合成的化合物。如果取代基自身被超过一个基团取代,应理解这些基团可在相同碳原子上或不同碳原子上,只要使结构稳定。In the compounds of the present invention, when any variable (eg, R, etc.) occurs more than once in any component, its definition at each occurrence is independent of the definition at each other occurrence. Likewise, combinations of substituents and variables are permissible so long as such combinations stabilize the compound. A line drawn into a ring system from a substituent indicates that the indicated bond may be attached to any substitutable ring atom. If the ring system is polycyclic, it means that such bonds are only attached to any suitable carbon atoms adjacent to the ring. It will be appreciated that one of ordinary skill in the art can select substituents and substitution patterns for the compounds of the present invention to provide compounds that are chemically stable and readily synthesized from readily available starting materials by the skill of the art and the methods set forth below. If a substituent is itself substituted with more than one group, it is understood that these groups may be on the same carbon atom or on different carbon atoms, so long as the structure is stabilized.
本文所用术语“烷基”意指包括具有特定碳原子数目的支链的和直链的饱和脂肪烃基。例如,“C1-C6烷基”中“C1-C6”的定义包括以直链或支链排列的具有1、2、3、4、5或6个碳原子的基团。例如,“C1-C6烷基”具体包括甲基、乙基、正丙基、异丙基、正丁基、叔丁基、异丁基、戊基、己基。The term "alkyl" as used herein is meant to include branched and straight chain saturated aliphatic hydrocarbon groups having the specified number of carbon atoms. For example, the definition of " C1 - C6 " in " C1 - C6 alkyl" includes groups having 1, 2, 3, 4, 5 or 6 carbon atoms in a straight or branched chain arrangement. For example, " C1 - C6 alkyl" specifically includes methyl, ethyl, n-propyl, isopropyl, n-butyl, tert-butyl, isobutyl, pentyl, hexyl.
术语“环烷基”指具有特定碳原子数目的单环饱和脂肪烃基。例如“环烷基”包括环丙基、环丁基、环戊基或环己基等。The term "cycloalkyl" refers to a monocyclic saturated aliphatic hydrocarbon group having the specified number of carbon atoms. For example, "cycloalkyl" includes cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl, and the like.
术语“烷氧基”指具有-O-烷基结构的基团,如-OCH3、-OCH2CH3、-OCH2CH2CH3、-O-CH2CH(CH3)2、-OCH2CH2CH2CH3、-O-CH(CH3)2等。The term "alkoxy" refers to groups having an -O-alkyl structure, such as -OCH 3 , -OCH 2 CH 3 , -OCH 2 CH 2 CH 3 , -O-CH 2 CH(CH 3 ) 2 , - OCH 2 CH 2 CH 2 CH 3 , -O-CH(CH 3 ) 2 and the like.
术语“杂芳基”指含有1个或多个选自O、N或S的杂原子的芳香环,本发明范围内的杂芳基包括但不限于:喹啉基、吡唑基、吡咯基、噻吩基、呋喃基、吡啶基、嘧啶基、吡嗪基、三氮唑基、咪唑基、噁唑基、异噁唑基、哒嗪基;“杂芳基”也理解为包括任何含有氮的杂芳基的N-氧化物衍生物。The term "heteroaryl" refers to an aromatic ring containing one or more heteroatoms selected from O, N or S. Heteroaryl groups within the scope of the present invention include, but are not limited to: quinolinyl, pyrazolyl, pyrrolyl , thienyl, furanyl, pyridyl, pyrimidinyl, pyrazinyl, triazolyl, imidazolyl, oxazolyl, isoxazolyl, pyridazinyl; "heteroaryl" is also understood to include any nitrogen-containing N-oxide derivatives of heteroaryl groups.
术语“取代的”是指用指定取代基的基团置换特定结构中的氢基。The term "substituted" refers to the replacement of a hydrogen group in a particular structure with a group of the designated substituent.
正如本领域技术人员所理解的,本文中所用“卤素”(“halo”)或“卤”意指氯、氟、溴和碘。As understood by those skilled in the art, "halo" ("halo") or "halo" as used herein means chlorine, fluorine, bromine and iodine.
除非另有定义,烷基、环烷基、芳基、杂芳基和杂环烷基取代基可为未被取代的或取代的。例如,C1-C6烷基可被一个、两个或三个选自OH、卤素、烷氧基、二烷基氨基或杂环基例如吗啉基、哌啶基等的取代基取代。Unless otherwise defined, alkyl, cycloalkyl, aryl, heteroaryl and heterocycloalkyl substituents can be unsubstituted or substituted. For example, a C1 - C6 alkyl group can be substituted with one, two, or three substituents selected from OH, halogen, alkoxy, dialkylamino, or heterocyclyl groups such as morpholinyl, piperidinyl, and the like.
本发明包括式Ⅰ-VI化合物的游离形式,也包括其药学上可接受的盐及立体异构体。本文中一些特定的示例性化合物为胺类化合物的质子化了的盐。术语“游离形式”指以非盐形式的胺类化合物。包括在内的药学上可接受盐不仅包括本文所述特定化合物的示例性盐,也包括所有式Ⅰ-VI化合物游离形式的典型的药学上可接受的盐。可使用本领域已知技术分离所述化合物特定盐的游离形式。例如,可通过用适当的碱稀水溶液例如NaOH稀水溶液、碳酸钾稀水溶液、稀氨水及碳酸氢钠稀水溶液处理该盐使游离形式再生。游离形式在某些物理性质例如在极性溶剂中溶解度上与其各自盐形式多少有些区别,但是为发明的目的这种酸盐及碱盐在其它药学方面与其各自游离形式相当。The present invention includes the free forms of the compounds of formulae I-VI, as well as the pharmaceutically acceptable salts and stereoisomers thereof. Some of the specific exemplary compounds herein are protonated salts of amine compounds. The term "free form" refers to the amine compound in non-salt form. Included pharmaceutically acceptable salts include not only exemplary salts of the particular compounds described herein, but also typical pharmaceutically acceptable salts of all compounds of Formulas I-VI in free form. The free forms of specific salts of the compounds can be isolated using techniques known in the art. For example, the free form can be regenerated by treating the salt with a suitable dilute aqueous base, such as dilute aqueous NaOH, dilute aqueous potassium carbonate, dilute aqueous ammonia, and dilute aqueous sodium bicarbonate. The free forms differ somewhat from their respective salt forms in certain physical properties such as solubility in polar solvents, but for the purposes of the invention such acid and base salts are otherwise pharmaceutically equivalent to their respective free forms.
可通过常规化学方法自含有碱性部分或酸性部分的本发明化合物合成本发明的药学上可接受的盐。通常,通过离子交换色谱或通过游离碱和化学计算量或过量的所需盐形式的无机或有机酸在适当溶剂或多种溶剂的组合中反应制备碱性化合物的盐。类似的,通过和适当的无机或有机碱反应形成酸性化合物的盐。The pharmaceutically acceptable salts of the present invention can be synthesized from compounds of the present invention containing a basic or acidic moiety by conventional chemical methods. Generally, salts of basic compounds are prepared by ion exchange chromatography or by reacting the free base and a stoichiometric amount or excess of an inorganic or organic acid in the desired salt form in a suitable solvent or combination of solvents. Similarly, salts of acidic compounds are formed by reaction with a suitable inorganic or organic base.
因此,本发明化合物的药学上可接受的盐包括通过碱性本发明化合物和无机或有机酸反应形成的本发明化合物的常规无毒盐。例如,常规的无毒盐包括得自无机酸例如盐酸、氢溴酸、硫酸、氨基磺酸、磷酸、硝酸等的盐,也包括自有机酸例如乙酸、丙酸、琥珀酸、乙醇酸、硬脂酸、乳酸、苹果酸、酒石酸、柠檬酸、抗坏血酸、扑酸、马来酸、羟基马来酸、苯乙酸、谷氨酸、苯甲酸、水杨酸、对氨基苯磺酸、2一乙酰氧基一苯甲酸、富马酸、甲苯磺酸、甲磺酸、乙烷二磺酸、草酸、羟乙基磺酸、三氟乙酸等制备的盐。Accordingly, pharmaceutically acceptable salts of the compounds of the present invention include conventional non-toxic salts of the compounds of the present invention formed by reacting a basic compound of the present invention with an inorganic or organic acid. For example, conventional non-toxic salts include those derived from inorganic acids such as hydrochloric acid, hydrobromic acid, sulfuric acid, sulfamic acid, phosphoric acid, nitric acid, and the like, as well as those derived from organic acids such as acetic acid, propionic acid, succinic acid, glycolic acid, hard Fatty acid, lactic acid, malic acid, tartaric acid, citric acid, ascorbic acid, pamoic acid, maleic acid, hydroxymaleic acid, phenylacetic acid, glutamic acid, benzoic acid, salicylic acid, p-aminobenzenesulfonic acid, 2-acetyl Salts prepared from oxymonobenzoic acid, fumaric acid, toluenesulfonic acid, methanesulfonic acid, ethanedisulfonic acid, oxalic acid, isethionic acid, trifluoroacetic acid, etc.
如果本发明化合物为酸性的,则适当的“药学上可接受的盐”指通过药学上可接受的无毒碱包括无机碱及有机碱制备的盐。得自无机碱的盐包括铝盐、铵盐、钙盐、铜盐、铁盐、亚铁盐、锂盐、镁盐、锰盐、亚锰盐、钾盐、钠盐、锌盐等。特别优选铵盐、钙盐、镁盐、钾盐和钠盐。得自药学上可接受的有机无毒碱的盐,所述碱包括伯胺、仲胺和叔胺的盐,取代的胺包括天然存在的取代胺、环状胺及碱性离子交换树脂例如精氨酸、甜菜碱、咖啡因、胆碱、N,N'-二苄基乙二胺、二乙胺、2一二乙基氨基乙醇、2一二甲基氨基乙醇、氨基乙醇、乙醇胺、乙二胺、N一乙基吗啉、N一乙基哌啶、葡萄糖胺、氨基葡萄糖、组氨酸、羟钴胺、异丙基胺、赖氨酸、甲基葡萄糖胺、吗啉、哌嗪,哌啶、呱咤、多胺树脂、普鲁卡因、嘌呤、可可碱、三乙胺、三甲胺、三丙胺、氨基丁三醇等。If the compound of the present invention is acidic, appropriate "pharmaceutically acceptable salts" refer to salts prepared from pharmaceutically acceptable non-toxic bases including inorganic and organic bases. Salts derived from inorganic bases include aluminum, ammonium, calcium, copper, iron, ferrous, lithium, magnesium, manganese, manganous, potassium, sodium, zinc, and the like. Ammonium, calcium, magnesium, potassium and sodium salts are particularly preferred. Salts derived from pharmaceutically acceptable organic non-toxic bases including salts of primary, secondary and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines and basic ion exchange resins such as Amino acid, betaine, caffeine, choline, N,N'-dibenzylethylenediamine, diethylamine, 2-diethylaminoethanol, 2-dimethylaminoethanol, aminoethanol, ethanolamine, ethyl Diamine, N-ethylmorpholine, N-ethylpiperidine, Glucosamine, Glucosamine, Histidine, Hydroxocobalamin, Isopropylamine, Lysine, Methylglucamine, Morpholine, Piperazine , Piperidine, quack, polyamine resin, procaine, purine, theobromine, triethylamine, trimethylamine, tripropylamine, tromethamine, etc.
Berg等,“Pharmaceutical Salts,”J.Pharm.Sci.’1977:66:1-19更详细描述了上文所述药学上可接受的盐及其它典型的药学上可接受的盐的制备。Berg et al., "Pharmaceutical Salts," J. Pharm. Sci. '1977:66:1-19 describes the preparation of the pharmaceutically acceptable salts described above and other typical pharmaceutically acceptable salts in more detail.
由于在生理条件下化合物中脱质子化的酸性部分例如羧基可为阴离子的,而这种带有的电荷然后可被内部带有阳离子的质子化了的或烷基化的碱性部分例如四价氮原子平衡抵消,所以应注意本发明化合物是潜在的内盐或两性离子。Since deprotonated acidic moieties such as carboxyl groups in compounds can be anionic under physiological conditions, such charged charges can then be replaced by internally cationic protonated or alkylated basic moieties such as tetravalent The nitrogen atoms balance out, so it should be noted that the compounds of the present invention are potential inner salts or zwitterions.
在一个实施方案中,本申请提供了一种利用具有式Ⅰ-VI的化合物及其药学可接受的盐治疗人或其它哺乳动物肿瘤等过渡增殖性疾病或症状。In one embodiment, the present application provides a method for the treatment of hyperproliferative diseases or conditions such as tumors in humans or other mammals using compounds of formulas I-VI and pharmaceutically acceptable salts thereof.
在一个实施方案中,本申请的化合物及其药学可接受的盐可以用于治疗或控制非小细胞肺癌、小细胞肺癌、淋巴癌、食管癌、卵巢癌、黑色素瘤、子宫颈癌、尿路上皮癌、胰腺癌、乳腺癌、肝癌、胃癌、胆管癌、白血病、黑色素瘤、结肠癌、直肠癌、子宫内膜癌或脑胶质瘤。In one embodiment, the compounds of the present application and pharmaceutically acceptable salts thereof can be used for the treatment or management of non-small cell lung cancer, small cell lung cancer, lymphoma, esophageal cancer, ovarian cancer, melanoma, cervical cancer, urinary tract cancer Skin cancer, pancreatic cancer, breast cancer, liver cancer, stomach cancer, bile duct cancer, leukemia, melanoma, colon cancer, rectal cancer, endometrial cancer or glioma.
药物代谢物及前药:本申请所涉及的化合物及其药学可接受的盐的代谢产物,以及可以在体内转变为本申请所涉及的化合物及其药学可接受的盐的结构的前药,也包含在本申请的权利要求中。Drug metabolites and prodrugs: the metabolites of the compounds involved in the application and their pharmaceutically acceptable salts, as well as prodrugs that can be converted into the structures of the compounds involved in the application and their pharmaceutically acceptable salts in vivo, also Included in the claims of this application.
联合用药:式Ⅰ-VI化合物可以与已知的治疗或改进相似病状的其它药物联用。联合给药时,原来药物的给药方式&剂量保持不变,而同时或随后服用式Ⅰ-VI化合物。当式Ⅰ-VI化合物与其它一种或几种药物同时服用时,优选使用同时含有一种或几种已知药物和式I-VI化合物的药用组合物。药物联用也包括在重叠的时间段服用式Ⅰ-VI化合物与其它一种或几种已知药物。当式Ⅰ-VI化合物与其它一种或几种药物进行药物联用时,式Ⅰ-VI化合物或已知药物的剂量可能比它们单独用药时的剂量较低。Combinations: The compounds of formulae I-VI may be used in combination with other drugs known to treat or ameliorate similar conditions. When combined administration, the original drug administration mode & dosage remain unchanged, while the compound of formula I-VI is administered concurrently or subsequently. When a compound of formulae I-VI is administered concomitantly with one or more other drugs, it is preferred to use a pharmaceutical composition containing one or more known drugs and a compound of formulae I-VI. Drug combinations also include administration of a compound of formulae I-VI with one or more other known drugs at overlapping time periods. When the compounds of formulas I-VI are administered in combination with one or more other drugs, the doses of the compounds of formulas I-VI or known drugs may be lower than when they are administered alone.
可以与式Ⅰ-VI化合物进行药物联用的药物或活性成分包括但不局限为:Drugs or active ingredients that can be used in combination with the compounds of formulae I-VI include, but are not limited to:
雌激素受体调节剂、雄激素受体调节剂、视网膜样受体调节剂、细胞毒素/细胞抑制剂、抗增殖剂、蛋白转移酶抑制剂、HMG-CoA还原酶抑制剂、HIV蛋白激酶抑制剂、逆转录酶抑制剂、血管生成抑制剂、细胞增殖及生存信号抑制剂、干扰细胞周期关卡的药物和细胞凋亡诱导剂,细胞毒类药物、酪氨酸蛋白抑制剂、EGFR抑制剂、VEGFR抑制剂、丝氨酸/苏氨酸蛋白抑制剂、Bcr-Abl抑制剂,c-Kit抑制剂,Met抑制剂,Raf抑制剂,MEK抑制剂,MMP抑制剂,拓扑异构酶抑制剂、组氨酸去乙酰化酶抑制剂、蛋白酶体抑制剂、CDK抑制剂,Bcl-2家族蛋白抑制剂,MDM2家族蛋白抑制剂、IAP家族蛋白抑制剂、STAT家族蛋白抑制剂、PI3K抑制剂、AKT抑制剂、整联蛋白阻滞剂、干扰素-α、白介素-12、COX-2抑制剂、p53、p53激活剂、VEGF抗体、EGF抗体等。Estrogen receptor modulators, androgen receptor modulators, retinal-like receptor modulators, cytotoxic/cytostatics, antiproliferative agents, protein transferase inhibitors, HMG-CoA reductase inhibitors, HIV protein kinase inhibitors agents, reverse transcriptase inhibitors, angiogenesis inhibitors, cell proliferation and survival signal inhibitors, drugs that interfere with cell cycle checkpoints and apoptosis inducers, cytotoxic drugs, tyrosine protein inhibitors, EGFR inhibitors, VEGFR inhibitors, serine/threonine protein inhibitors, Bcr-Abl inhibitors, c-Kit inhibitors, Met inhibitors, Raf inhibitors, MEK inhibitors, MMP inhibitors, topoisomerase inhibitors, histidine Acid deacetylase inhibitors, proteasome inhibitors, CDK inhibitors, Bcl-2 family protein inhibitors, MDM2 family protein inhibitors, IAP family protein inhibitors, STAT family protein inhibitors, PI3K inhibitors, AKT inhibitors , Integrin blocker, interferon-α, interleukin-12, COX-2 inhibitor, p53, p53 activator, VEGF antibody, EGF antibody, etc.
在一个实施方案中,可以与式Ⅰ-VI化合物进行药物联用的药物或活性成分包括但不局限为:阿地白介素、阿仑膦酸、干扰素、阿曲诺英、别嘌醇、别嘌醇钠、帕洛诺司琼盐酸盐、六甲蜜胺、氨基格鲁米特、氨磷汀、氨柔比星、安丫啶、阿纳托唑、多拉司琼、aranesp、arglabin、三氧化二砷、阿诺新、5-氮胞苷、硫唑嘌呤、卡介苗或tice卡介苗、贝他定、醋酸倍他米松、倍他米松磷酸钠制剂、贝沙罗汀、硫酸博来霉素、溴尿甘、bortezomib、白消安、降钙素、阿来佐单抗注射剂、卡培他滨、卡铂、康士得、cefesone、西莫白介素、柔红霉素、苯丁酸氮芥、顺铂、克拉屈滨、克拉屈滨、氯屈磷酸、环磷酰胺、阿糖胞昔、达卡巴嗪、放线菌素D、柔红霉素脂质体、地塞米松、磷酸地塞米松、戊酸雌二醇、地尼白介素2、狄波美、地洛瑞林、地拉佐生、己烯雌酚、大扶康、多西他奇、去氧氟尿苷、阿霉素、屈大麻酚、钦-166-壳聚糖复合物、eligard、拉布立酶、盐酸表柔比星、阿瑞吡坦、表阿霉素、阿法依伯汀、红细胞生成素、依铂、左旋咪唑片、雌二醇制剂、17-β-雌二醇、雌莫司汀磷酸钠、炔雌醇、氨磷汀、羟磷酸、凡毕复、依托泊甙、法倔唑、他莫昔芬制剂、非格司亭、非那司提、非雷司替、氟尿苷、氟康唑、氟达拉滨、5-氟脱氧尿嘧啶核苷一磷酸盐、5-氟尿嘧啶、氟甲睾酮、氟他胺、福麦斯坦、1-β-D-阿糖呋喃糖胞噻啶-5’-硬脂酰磷酸酯、福莫司汀、氟维司群、丙种球蛋白、吉西他滨、吉妥单抗、甲磺酸伊马替尼、卡氮芥糯米纸胶囊剂、戈舍瑞林、盐酸格拉尼西隆、组氨瑞林、和美新、氢化可的松、赤型-羟基壬基腺嘌呤、羟基脲、替坦异贝莫单抗、伊达比星、异环磷酰胺、干扰素α、干扰素-α2、干扰素α-2A、干扰素α-2B、干扰素α-nl、干扰素α-n3、干扰素β、干扰素γ-la、白细胞介素-2、内含子A、易瑞沙、依立替康、凯特瑞、硫酸香菇多糖、来曲唑、甲酰四氢叶酸、亮丙瑞林、亮丙瑞林醋酸盐、左旋四咪唑、左旋亚叶酸钙盐、左甲状腺素钠、左甲状腺素钠制剂、洛莫司汀、氯尼达明、屈大麻酚、氮芥、甲钴胺、甲羟孕酮醋酸酯、醋酸甲地孕酮、美法仑、酯化雌激素、6-琉基嘌呤、美司钠、氨甲蝶呤、氨基乙酰丙酸甲酯、米替福新、美满霉素、丝裂霉素C、米托坦、米托葱醌、曲洛司坦、柠檬酸阿霉素脂质体、奈达铂、聚乙二醇化非格司亭、奥普瑞白介素、neupogen、尼鲁米特、三苯氧胺、NSC-631570、重组人白细胞介素1-β、奥曲肽、盐酸奥丹西隆、去氢氢化可的松口服溶液剂、奥沙利铂、紫杉醇、泼尼松磷酸钠制剂、培门冬酶、派罗欣、喷司他丁、溶链菌制剂、盐酸匹鲁卡品、毗柔比星、普卡霉素、卟吩姆钠、泼尼莫司汀、司替泼尼松龙、泼尼松、倍美力、丙卡巴脐、重组人类红细胞生成素、雷替曲塞、利比、依替膦酸铼-186、美罗华、力度伸-A、罗莫肽、盐酸毛果芸香碱片剂、奥曲肽、沙莫司亭、司莫司汀、西佐喃、索布佐生、唬钠甲强龙、帕福斯酸、干细胞治疗、链佐星、氯化锶-89、左旋甲状腺素钠、他莫昔芬、坦舒洛辛、他索那明、tastolactone、泰索帝、替西硫津、替莫唑胺、替尼泊苷、丙酸睾酮、甲睾酮、硫鸟嘌呤、噻替哌、促甲状腺激素、替鲁膦酸、拓扑替康、托瑞米芬、托西莫单抗、曲妥珠单抗、曲奥舒凡、维A酸、甲氨喋呤片剂、三甲基密胺、三甲曲沙、乙酸曲普瑞林、双羟萘酸曲普瑞林、优福定、尿苷、戊柔比星、维司力农、长春碱、长春新碱、长春酰胺、长春瑞滨、维鲁利秦、右旋丙亚胺、净司他丁斯酯、枢复宁、紫杉醇蛋白质稳定制剂、acolbifene、干扰素r-lb、affinitak、氨基喋呤、阿佐昔芬、asoprisnil、阿他美坦、阿曲生坦、BAY43-9006、阿瓦斯丁、CCI-779、CDC-501、西乐葆、西妥昔单抗、克立那托、环丙孕酮醋酸酯、地西他滨、DN-101、阿霉素-MTC、dSLIM、度他雄胺、edotecarin、依氟鸟氨酸、依喜替康、芬维A胺、组胺二盐酸盐、组氨瑞林水凝胶植入物、钬-166DOTMP、伊班膦酸、干扰素γ、内含子-PEG、ixabepilone、匙孔形血蓝蛋白、L-651582、兰乐肽、拉索昔芬、libra、lonafamib、米泼昔芬、米诺屈酸酯、MS-209、脂质体MTP-PE、MX-6、那法瑞林、奈莫柔比星、新伐司他、诺拉曲特、奥利默森、onco-TCS、osidem、紫杉醇聚谷氨酸酯、帛米酸钠、PN-401、QS-21、夸西洋、R-1549、雷洛昔芬、豹蛙酶、13-顺维A酸、沙铂、西奥骨化醇、T-138067、tarceva、二十二碳六烯酸紫杉醇、胸腺素αl、嘎唑呋林、tipifarnib、替拉扎明、TLK-286、托瑞米芬、反式MID-lo7R、伐司朴达、伐普肽、vatalanib、维替泊芬、长春氟宁、Z-100和唑来麟酸或它们的组合。In one embodiment, the drugs or active ingredients that can be used in combination with the compounds of formulae I-VI include, but are not limited to: aldesleukin, alendronate, interferon, atranoin, allopurinol, allopurinol, Sodium purinol, Palonosetron hydrochloride, Hexamethylmelamine, Aminoglutide, Amifostine, Amrubicin, Amridine, Anastrozole, Dolassetron, aranesp, arglabin, Arsenic trioxide, Arsenic, 5-azacytidine, azathioprine, BCG or tice BCG, betadine, betamethasone acetate, betamethasone sodium phosphate preparation, bexarotene, bleomycin sulfate, bromuria Gan, bortezomib, busulfan, calcitonin, alezolizumab injection, capecitabine, carboplatin, casodex, cefesone, simoleukin, daunorubicin, chlorambucil, cisplatin, Cladribine, Cladribine, Clodronate, Cyclophosphamide, Cytarabine, Dacarbazine, Actinomycin D, Daunorubicin Liposome, Dexamethasone, Dexamethasone Phosphate, Valeric Acid Estradiol, Denisole-2, Dipomet, Deslorelin, Delazoxan, Diethylstilbestrol, Diflucan, Docetaxel, Deoxyfluridine, Doxorubicin, Dronabinol, Chin-166 -Chitosan complex, eligard, rasburicase, epirubicin hydrochloride, aprepitant, epirubicin, epoetin alfa, erythropoietin, eplatin, levamisole tablets, estradiol Preparation, 17-beta-estradiol, estramustine sodium phosphate, ethinyl estradiol, amifostine, hydroxyphosphate, fenbifur, etoposide, fadrozole, tamoxifen preparation, filgrastim , finasteride, feleastide, floxuridine, fluconazole, fludarabine, 5-fluorodeoxyuridine monophosphate, 5-fluorouracil, fluoxymesterone, flutamide, formin Stan, 1-β-D-arabinofuranosyl cytidine-5'-stearoyl phosphate, fomustine, fulvestrant, gamma globulin, gemcitabine, gemtuzumab, imabyl mesylate Matinib, Carzapine Wax Paper Capsules, Goserelin, Granisirone Hydrochloride, Histrelin, and Methionine, Hydrocortisone, Erythro-hydroxynonyladenine, Hydroxyurea, Titan Isobemumab, idarubicin, ifosfamide, interferon alpha, interferon-alpha2, interferon alpha-2A, interferon alpha-2B, interferon alpha-nl, interferon alpha-n3, interferon Interleukin β, Interferon γ-la, Interleukin-2, Intron A, Iressa, Irinotecan, Kaiteri, Lentinan sulfate, Letrozole, Leucovorin, Leuprolide, Leuprolide acetate, levothyroxine, levofolinate calcium salt, levothyroxine sodium, levothyroxine sodium preparations, lomustine, lonidamine, dronabinol, nitrogen mustard, methylcobalamin, Medroxyprogesterone acetate, megestrol acetate, melphalan, esterified estrogens, 6-mercaptopurine, mesna, methotrexate, methyl aminolevulinate, miltefosine, Meiman Amycin, mitomycin C, mitotane, mitosodium quinone, trilostane, doxorubicin citrate liposome, nedaplatin, pegylated filgrastim, oppreleukin, neupogen, nilutamide, tamoxifen, NSC-63 1570. Recombinant human interleukin-1-beta, octreotide, ondansetron hydrochloride, dehydrocortisone oral solution, oxaliplatin, paclitaxel, prednisone sodium phosphate preparation, pegmatase, perol Xin, pentostatin, streptolytic preparations, pilocarpine hydrochloride, pirorubicin, prukamycin, porfim sodium, prednimustine, steprednisolone, prednisone, Premarin, Procarba, Recombinant Human Erythropoietin, Raltitrexed, Ribbi, Etidronate, Rhenium-186, Rituxan, Rituxan-A, Romotide, Pilocarpine Hydrochloride Tablets, Octreotide, Shamo Krastim, Semustine, Sizoran, Sobuzoxan, Methylprednisolone, Paphos Acid, Stem Cell Therapy, Streptozocin, Strontium Chloride-89, Levothyroxine Sodium, Tamoxifen, tamsulosin, tasonamin, tastolactone, taxotere, tesetiazine, temozolomide, teniposide, testosterone propionate, methyltestosterone, thioguanine, thiotepa, thyrotropin, tiludosine acid, topotecan, toremifene, tosilimumab, trastuzumab, triosulfan, tretinoin, methotrexate tablets, trimethylmelamine, trimetrexate, acetic acid Triptorelin, Triptorelin pamoate, Eufodine, Uridine, Valrubicin, Vesrinone, Vinblastine, Vincristine, Vincamide, Vinorelbine, Velulizine, Dextropropimide, net statin, zofenin, paclitaxel protein stabilizer, acolbifene, interferon r-lb, affinitak, aminopterin, azoxifene, asoprisnil, atametan, atraxen Tan, BAY43-9006, Avastin, CCI-779, CDC-501, Celebrex, Cetuximab, Crinator, Cyproterone Acetate, Decitabine, DN-101, Doxorubicin -MTC, dSLIM, dutasteride, edotecarin, eflunomine, ixitecan, fenretinide, histamine dihydrochloride, histrelin hydrogel implant, holmium-166DOTMP, ibandronic acid, interferon gamma, intron-PEG, ixabepilone, keyhole limpet hemocyanin, L-651582, lanreotide, lasoxifene, libra, lonafamib, milepoxifene, minodex Ester, MS-209, liposomal MTP-PE, MX-6, nafarelin, naimrubicin, novarestat, nolatrexide, olimerson, onco-TCS, osidem, paclitaxel Polyglutamic acid ester, sodium bromide, PN-401, QS-21, quasiyang, R-1549, raloxifene, leopard enzyme, 13-cis-retinoic acid, saplatin, theocalcidol , T-138067, tarceva, docosahexaenoate paclitaxel, thymosin alfa, gazofurin, tipifarnib, tirapazamine, TLK-286, toremifene, trans MID-lo7R, valspro Dalta, vapretide, vatalanib, verteporfin, vinflunine, Z-100, and zolelinic acid or a combination thereof.
以下实施例对本发明做进一步的描述,但该实施例并非用于限制本发明的保护范围。The following examples further describe the present invention, but the examples are not intended to limit the protection scope of the present invention.
实施例1:化合物5a-5j的合成Example 1: Synthesis of Compounds 5a-5j
中间体4a的合成:Synthesis of Intermediate 4a:
将NaH(192mg,4.8mmol)放于反应瓶中,置换氮气,加入7mL无水DMF溶解,在冰水浴的条件下加入膦酰基乙酸甲酯二乙酯,搅拌0.5小时,逐滴加入化合物3a(636mg,3.973mmol)的7.5mL DMF溶液,室温搅拌8小时。取样点板,反应基本完毕,加水淬灭反应,搅拌2小时,用乙酸乙酯萃取三次,饱和食盐水洗,干燥过滤,浓缩过硅胶柱纯化得到黄色固体化合物n1(520mg,60.5%);将化合物n1(520mg,2.4mmol)溶解于THF/MeOH/H2O(5mL/8mL/8mL)混合溶液中,加入NaOH(288mg,7.2mmol),室温搅拌4小时。取样点板,反应基本完毕。加HCl(1M)调节pH=5.0,过滤获得中间体4a(413mg,85%)。NaH (192 mg, 4.8 mmol) was placed in the reaction flask, nitrogen was replaced, 7 mL of anhydrous DMF was added to dissolve, methyl phosphonoacetate diethyl ester was added under the condition of ice-water bath, stirred for 0.5 hours, compound 3a ( 636 mg, 3.973 mmol) in 7.5 mL DMF and stirred at room temperature for 8 hours. Sampling plate, the reaction is basically completed, add water to quench the reaction, stir for 2 hours, extract three times with ethyl acetate, wash with saturated brine, dry filter, concentrate and purify through silica gel column to obtain yellow solid compound n1 (520 mg, 60.5%); n1 (520 mg, 2.4 mmol) was dissolved in a mixed solution of THF/MeOH/H 2 O (5 mL/8 mL/8 mL), NaOH (288 mg, 7.2 mmol) was added, and the mixture was stirred at room temperature for 4 hours. Sampling point plate, the reaction is basically completed. HCl (1M) was added to adjust pH=5.0, and intermediate 4a was obtained by filtration (413 mg, 85%).
中间体4b的合成:Synthesis of Intermediate 4b:
向化合物3b(300mg,1.5mmol)的吡啶(10mL)溶液中加入丙二酸n2(312mg,3mmol)和哌啶(1mL)。将混合物在110℃下搅拌6小时。然后将混合物冷却至室温,并用NaHCO3水溶液酸化。用HCl(1M)调pH=5,EA萃取。用饱和食盐水洗涤,无水MgSO4干燥,过滤,浓缩得到粗产物4b,为黄色固体,其无需进一步纯化即可用于下一步(188mg,粗品)。To a solution of compound 3b (300 mg, 1.5 mmol) in pyridine (10 mL) was added malonic acid n2 (312 mg, 3 mmol) and piperidine (1 mL). The mixture was stirred at 110°C for 6 hours. The mixture was then cooled to room temperature and acidified with aqueous NaHCO3 . Adjust pH=5 with HCl (1 M) and extract with EA. Washed with saturated brine, dried over anhydrous MgSO4 , filtered, and concentrated to give crude product 4b as a yellow solid, which was used in the next step without further purification (188 mg, crude).
中间体4c的合成:Synthesis of Intermediate 4c:
将化合物3c(360mg,1.52mmol)和氟乙酸乙酯(177mg,1.67mmol),放于反应瓶中,氮气置换,加无水DCM溶解,逐滴加TiCl4(1M in CH2Cl2,1.82mL,1.82mmol),室温搅拌0.5小时,然后加入TEA(0.42mL,3.02mmol),室温搅拌2小时。取样点板,反应基本完毕,EA萃取三次,合并有机相,干燥,过滤,浓缩,硅胶柱层析纯化得化合物n3(220mg,47%)。Compound 3c (360 mg, 1.52 mmol) and ethyl fluoroacetate (177 mg, 1.67 mmol) were placed in a reaction flask, replaced with nitrogen, dissolved in anhydrous DCM, and added dropwise TiCl 4 (1M in CH 2 Cl 2 , 1.82 mL, 1.82 mmol), stirred at room temperature for 0.5 hours, then added TEA (0.42 mL, 3.02 mmol) and stirred at room temperature for 2 hours. Sampling plate, the reaction is basically completed, EA is extracted three times, the organic phases are combined, dried, filtered, concentrated, and purified by silica gel column chromatography to obtain compound n3 (220 mg, 47%).
1H NMR(400MHz,CDCl3)δ8.46–8.36(m,2H),7.61-7.51(m,3H),7.47–7.38(m,2H),7.22(dd,J=8.0,4.8Hz,1H),6.96(d,J=40.1Hz,1H),4.29(q,J=7.1Hz,2H),3.79(s,3H),1.32(t,J=7.1Hz,3H);13C NMR(100MHz,CDCl3)δ161.9(d,J=33.5Hz),148.7,144.1(d,J=2.2Hz),143.9,143.8(d,J=257.7Hz),130.8(d,J=18.0Hz),130.7,129.6,129.5,128.8,118.4,117.3(d,J=3.5Hz),111.5(d,J=9.3Hz),104.9(d,J=2.5Hz),61.4,30.1,14.3. 1 H NMR(400MHz, CDCl3)δ8.46-8.36(m,2H),7.61-7.51(m,3H),7.47-7.38(m,2H),7.22(dd,J=8.0,4.8Hz,1H) , 6.96 (d, J=40.1 Hz, 1H), 4.29 (q, J=7.1 Hz, 2H), 3.79 (s, 3H), 1.32 (t, J=7.1 Hz, 3H); 13 C NMR (100 MHz, CDCl3)δ161.9(d,J=33.5Hz),148.7,144.1(d,J=2.2Hz),143.9,143.8(d,J=257.7Hz),130.8(d,J=18.0Hz),130.7, 129.6, 129.5, 128.8, 118.4, 117.3 (d, J=3.5Hz), 111.5 (d, J=9.3Hz), 104.9 (d, J=2.5Hz), 61.4, 30.1, 14.3.
将化合物n3(220mg,0.71mmol)和LiOH·H2O(85mg,2.02mmol)放于反应瓶中,加THF和水(10mL和10mL)。室温搅拌3小时,取样点板,反应基本完毕,HCl酸化至pH6,真空浓缩,EA萃取三次,合并有机相,浓缩得灰白色固体中间体4c(150mg,71%)。Compound n3 (220 mg, 0.71 mmol) and LiOH·H 2 O (85 mg, 2.02 mmol) were placed in a reaction flask, THF and water (10 mL and 10 mL) were added. Stir at room temperature for 3 hours, sample and spot plate, the reaction is basically completed, acidified with HCl to pH 6, concentrated in vacuo, extracted three times with EA, combined organic phases, concentrated to give off-white solid Intermediate 4c (150 mg, 71%).
1H NMR(400MHz,DMSO-d6)δ8.36(d,J=4.4Hz,1H),8.21(d,J=7.8Hz,1H),7.66–7.47(m,5H),7.24(dd,J=7.9,4.6Hz,1H),6.76(d,J=40.5Hz,1H),3.66(s,3H);13C NMR(100MHz,DMSO-d6)δ162.8(d,J=33.9Hz),148.6,144.4(d,J=255.7Hz),144.3(d,J=2.0Hz),144.2,131.2,130.3(d,J=18.0Hz),130.1,129.6,129.3,117.9,117.8(d,J=3.3Hz),110.7(d,J=9.8Hz),104.1(d,J=2.4Hz),30.2. 1 H NMR (400MHz, DMSO-d6)δ8.36(d,J=4.4Hz,1H),8.21(d,J=7.8Hz,1H),7.66-7.47(m,5H),7.24(dd,J =7.9,4.6Hz,1H),6.76(d,J=40.5Hz,1H),3.66(s,3H); 13C NMR(100MHz,DMSO-d6)δ162.8(d,J=33.9Hz), 148.6, 144.4 (d, J=255.7Hz), 144.3 (d, J=2.0Hz), 144.2, 131.2, 130.3 (d, J=18.0Hz), 130.1, 129.6, 129.3, 117.9, 117.8 (d, J= 3.3Hz), 110.7 (d, J=9.8Hz), 104.1 (d, J=2.4Hz), 30.2.
中间体4d的合成:Synthesis of intermediate 4d:
将化合物3c(300mg,1.27mmol),2-氰基乙酸叔丁酯(198mg,1.40mmol),哌啶(36mg,0.42mmol),乙酸(174mg,2.90mmol)和甲苯(10ml)溶液放于反应瓶中,搅拌15小时。取样点板反应基本完毕。用EA(80mL)和饱和NaHCO3水溶液(30mL)萃取,干燥,过滤,浓缩,硅胶柱层析纯化得白色固体化合物n4(410mg,90%)。A solution of compound 3c (300 mg, 1.27 mmol), tert-butyl 2-cyanoacetate (198 mg, 1.40 mmol), piperidine (36 mg, 0.42 mmol), acetic acid (174 mg, 2.90 mmol) and toluene (10 ml) was placed in the reaction bottle and stir for 15 hours. The sampling point plate reaction is basically completed. Extracted with EA (80 mL) and saturated aqueous NaHCO 3 (30 mL), dried, filtered, concentrated, and purified by silica gel column chromatography to give compound n4 (410 mg, 90%) as a white solid.
1H NMR(400MHz,CDCl3)δ8.82(d,J=8.0Hz,1H),8.47(d,J=4.3Hz,1H),8.08(s,1H),7.63–7.55(m,3H),7.45–7.38(m,2H),7.34(dd,J=8.1,4.7Hz,1H),3.83(s,3H),1.53(s,9H).13C NMR(100MHz,CDCl3)δ162.9,150.1,149.2,148.8,144.6,131.9,130.8,130.3,128.9,128.5,118.3,117.6,108.1,98.1,82.6,77.4,77.0,76.7,30.5,28.1. 1 H NMR (400 MHz, CDCl 3 ) δ 8.82 (d, J=8.0 Hz, 1H), 8.47 (d, J=4.3 Hz, 1H), 8.08 (s, 1H), 7.63-7.55 (m, 3H) , 7.45–7.38(m, 2H), 7.34(dd, J=8.1, 4.7Hz, 1H), 3.83(s, 3H), 1.53(s, 9H). 13 C NMR(100MHz, CDCl 3 )δ162.9,150.1 ,149.2,148.8,144.6,131.9,130.8,130.3,128.9,128.5,118.3,117.6,108.1,98.1,82.6,77.4,77.0,76.7,30.5,28.1.
将化合物n4(390mg,1.08mmol)放于反应瓶中,加入10mL CH2Cl2溶解,加入3mLTFA,室温搅拌12小时。取样点板反应基本完毕。浓缩得到黄色固体4d(311mg,95%)。Compound n4 (390 mg, 1.08 mmol) was placed in a reaction flask, 10 mL of CH 2 Cl 2 was added to dissolve, 3 mL of TFA was added, and the mixture was stirred at room temperature for 12 hours. The sampling point plate reaction is basically completed. Concentration gave 4d as a yellow solid (311 mg, 95%).
1HNMR(400MHz,DMSO-d6)δ8.70(d,J=7.7Hz,1H),8.51(d,J=4.2Hz,1H),7.98(s,1H),7.71–7.57(m,5H),7.43(dd,J=8.1,4.7Hz,1H),3.78(s,3H).13C NMR(100MHz,DMSO-d6)δ165.1,151.0,149.2,149.0,145.1,131.5,130.9,129.3,128.4,118.8,118.5,117.2,107.4,96.7,30.8. 1 HNMR (400MHz, DMSO-d 6 )δ8.70(d,J=7.7Hz,1H),8.51(d,J=4.2Hz,1H),7.98(s,1H),7.71-7.57(m,5H) The _ 128.4, 118.8, 118.5, 117.2, 107.4, 96.7, 30.8.
中间体4e的合成:Synthesis of Intermediate 4e:
以化合物3c(8.01g,33.90mmol)为原料,参照化合物4a的合成方法制备中间体4e(8.03g,85%)。Using compound 3c (8.01 g, 33.90 mmol) as starting material, the intermediate 4e (8.03 g, 85%) was prepared according to the synthesis method of compound 4a.
1H NMR(400MHz,DMSO-d6)δ8.45–8.31(m,2H),7.68–7.55(m,5H),7.48(d,J=16.0Hz,1H),7.32(dd,J=7.9,4.8Hz,1H),6.40(d,J=16.0Hz,1H),3.69(s,3H).13C NMR(100MHz,DMSO-d6)δ168.8,148.8,145.7,144.4,137.7,131.3,130.2,129.6,129.4,129.3,118.3,117.9,114.6,108.0,30.1.HRMS(ESI)calcd for C17H14N2O2(M+H)+279.1128,found279.1128. 1 H NMR (400 MHz, DMSO-d 6 ) δ 8.45-8.31 (m, 2H), 7.68-7.55 (m, 5H), 7.48 (d, J=16.0 Hz, 1H), 7.32 (dd, J=7.9 , 4.8Hz, 1H), 6.40(d, J=16.0Hz, 1H), 3.69(s, 3H). 13 C NMR (100MHz, DMSO-d 6 )δ168.8, 148.8, 145.7, 144.4, 137.7, 131.3, 130.2 ,129.6,129.4,129.3,118.3,117.9,114.6,108.0,30.1.HRMS(ESI)calcd for C 17 H 14 N 2 O 2 (M+H) + 279.1128,found279.1128.
中间体4f的合成:Synthesis of Intermediate 4f:
以化合物3d(940.4mg,4.0mmoL)为原料,参照化合物4a的合成方法制备中间体4f(747mg,55%)。1H NMR(400MHz,DMSO-d6)δ7.94(d,J=7.8Hz,1H),7.65–7.55(m,4H),7.53–7.43(m,3H),7.31(dt,J=24.9,7.5Hz,2H),6.33(d,J=16.0Hz,1H),3.62(s,3H).Using compound 3d (940.4 mg, 4.0 mmol) as starting material, the intermediate 4f (747 mg, 55%) was prepared according to the synthesis method of compound 4a. 1 H NMR (400 MHz, DMSO-d 6 ) δ 7.94 (d, J=7.8 Hz, 1H), 7.65-7.55 (m, 4H), 7.53-7.43 (m, 3H), 7.31 (dt, J=24.9 ,7.5Hz,2H),6.33(d,J=16.0Hz,1H),3.62(s,3H).
中间体4g的合成:Synthesis of Intermediate 4g:
以化合物3e(300mg,1.27mmol)为原料,参照化合物4b的合成方法制备中间体4g(200mg,粗品)。Using compound 3e (300 mg, 1.27 mmol) as raw material, the intermediate 4g (200 mg, crude product) was prepared according to the synthesis method of compound 4b.
化合物5a的合成Synthesis of compound 5a
向化合物4a(126mg,0.623mmol)和TEA(0.3mL)的DMF(3mL)溶液中加入HATU(237mg,0.52mmol),0℃搅拌0.5小时,加入6,7-二甲氧基-1,2,3,4-四氢异喹啉盐酸盐(120mg,0.623mmol)。室温搅拌5小时,取样点板,反应基本完毕,加冰水稀释,抽滤,乙酸乙酯溶解,无水硫酸钠干燥,过滤浓缩,硅胶柱纯化得到化合物5a,白色固体(117mg,50%)。To a solution of compound 4a (126 mg, 0.623 mmol) and TEA (0.3 mL) in DMF (3 mL) was added HATU (237 mg, 0.52 mmol), stirred at 0°C for 0.5 h, and added 6,7-dimethoxy-1,2 , 3,4-Tetrahydroisoquinoline hydrochloride (120 mg, 0.623 mmol). Stir at room temperature for 5 hours, sample and spot plate, the reaction is basically completed, add ice water for dilution, suction filtration, dissolve in ethyl acetate, dry over anhydrous sodium sulfate, filter and concentrate, and purify with silica gel column to obtain compound 5a as a white solid (117 mg, 50%) .
1H NMR(400MHz,CDCl3)δ8.39(dd,J=4.7,1.4Hz,1H),8.18(d,J=7.4Hz,1H),7.88(d,J=15.3Hz,1H),7.47(s,1H),7.19(dd,J=7.9,4.7Hz,1H),6.89(d,J=15.4Hz,1H),6.66(d,J=7.8Hz,2H),4.79(s,2H),3.89(s,5H),3.86(s,6H),2.87(t,J=28.1Hz,2H).13CNMR(100MHz,CDCl3)δ166.7,148.8,147.9,143.9,135.8,132.3,128.5,127.3,125.8,124.4,118.5,116.8,112.9,111.3,111.1,109.5,56.1,47.2,44.6,43.8,40.2,31.5.HRMS(ESI)calcd for C22H23N3O3(M+H)+378.1812found 378.1804. 1 H NMR (400 MHz, CDCl 3 ) δ 8.39 (dd, J=4.7, 1.4 Hz, 1H), 8.18 (d, J=7.4 Hz, 1H), 7.88 (d, J=15.3 Hz, 1H), 7.47 (s,1H),7.19(dd,J=7.9,4.7Hz,1H),6.89(d,J=15.4Hz,1H),6.66(d,J=7.8Hz,2H),4.79(s,2H) , 3.89(s, 5H), 3.86(s, 6H), 2.87(t, J=28.1Hz, 2H). 13 CNMR(100MHz, CDCl3)δ166.7,148.8,147.9,143.9,135.8,132.3,128.5,127.3, found _ _ _ _ 378.1804.
化合物5b的合成Synthesis of compound 5b
向化合物4b(200mg,0.826mmol)和6,7-二甲氧基-1,2,3,4-四氢异喹啉盐酸盐(140mg,0.60mmol)的DMF(15mL)溶液中加入DIPEA(1mL)和HATU(380mg,1mmol)。室温搅拌16小时。取样点板,反应基本完毕,加水稀释,EA萃取。用饱和食盐水洗涤合并的有机相,再用无水MgSO4干燥,过滤,浓缩,硅胶柱层析纯化得到黄色固体产物化合物5b(90mg,26%)。To a solution of compound 4b (200 mg, 0.826 mmol) and 6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline hydrochloride (140 mg, 0.60 mmol) in DMF (15 mL) was added DIPEA (1 mL) and HATU (380 mg, 1 mmol). Stir at room temperature for 16 hours. Sampling point plate, the reaction is basically completed, diluted with water, EA extraction. The combined organic phases were washed with saturated brine, dried over anhydrous MgSO 4 , filtered, concentrated, and purified by silica gel column chromatography to obtain compound 5b (90 mg, 26%) as a yellow solid product.
1H NMR(400MHz,CDCl3)δ8.36(dd,J=4.7,1.4Hz,1H),8.27(d,J=15.4Hz,1H),8.14(d,J=7.2Hz,1H),7.22-7.14(m,1H),6.88(d,J=15.4Hz,1H),6.75-6.63(m,2H),4.82(s,2H),4.05–3.77(m,11H),3.00-2.80(m,2H),2.01–1.87(m,1H),1.35–1.22(m,2H),0.97–0.85(m,2H).13C NMR(100MHz,CDCl3)δ167.0,148.3,144.6,143.1,135.7,127.8,127.3,126.1,125.9,124.4,118.4,116.8,112.9,112.7,111.5,111.2,109.5,109.2,56.0,47.2,44.5,43.8,40.1,28.3,6.7,6.3.HRMS(ESI)calcd for C25H27N3O3(M+H)+418.2125,found418.2128. 1 H NMR (400 MHz, CDCl 3 ) δ 8.36 (dd, J=4.7, 1.4 Hz, 1H), 8.27 (d, J=15.4 Hz, 1H), 8.14 (d, J=7.2 Hz, 1H), 7.22 -7.14(m, 1H), 6.88(d, J=15.4Hz, 1H), 6.75-6.63(m, 2H), 4.82(s, 2H), 4.05-3.77(m, 11H), 3.00-2.80(m , 2H), 2.01–1.87 (m, 1H), 1.35–1.22 (m, 2H), 0.97–0.85 (m, 2H). 13 C NMR (100MHz, CDCl 3 )δ167.0, 148.3, 144.6, 143.1, 135.7, 127.8,127.3,126.1,125.9,124.4,118.4,116.8,112.9,112.7,111.5,111.2,109.5,109.2,56.0,47.2,44.5,43.8,40.1,28.3,6.7,6.3.HRMS H 27 N 3 O 3 (M+H) + 418.2125, found418.2128.
化合物5c的合成Synthesis of compound 5c
向化合物4c(130mg,0.44mmol)和6,7-二甲氧基-1,2,3,4-四氢异喹啉盐酸盐(111mg,0.48mmol)的DMF(8mL)溶液中加入DIPEA(0.23mL,1.32mmol)和HATU(200mg,0.53mmol)。室温搅拌1小时。取样点板,反应基本完毕,加水稀释,EA萃取。用饱和食盐水洗涤合并的有机相,再用无水MgSO4干燥,过滤,浓缩,硅胶柱层析纯化得到白色固体产物化合物5c(165mg,79%)。To a solution of compound 4c (130 mg, 0.44 mmol) and 6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline hydrochloride (111 mg, 0.48 mmol) in DMF (8 mL) was added DIPEA (0.23 mL, 1.32 mmol) and HATU (200 mg, 0.53 mmol). Stir at room temperature for 1 hour. Sampling point plate, the reaction is basically completed, diluted with water, EA extraction. The combined organic phases were washed with saturated brine, dried over anhydrous MgSO 4 , filtered, concentrated, and purified by silica gel column chromatography to obtain compound 5c (165 mg, 79%) as a white solid product.
1H NMR(400MHz,CDCl3)δ8.43(d,J=4.7Hz,1H),8.37(d,J=7.2Hz,1H),7.58–7.47(m,3H),7.46–7.39(m,2H),7.23(dd,J=7.9,4.8Hz,1H),6.78–6.55(m,3H),4.77–4.61(m,2H),3.91–3.73(m,11H),2.89–2.81(m,2H).13C NMR(100MHz,CDCl3)δ162.9,162.6,149.6,148.6,147.8,147.8,147.0,143.6,143.1,130.7,130.6,130.4,129.9,129.3,128.8,126.3,124.7,118.4,116.9,111.44,109.7,109.6,109.1,104.9,67.1,56.0,30.0.HRMS(ESI)calcd for C28H26FN3O3(M+H)+472.2031,found 472.2034. 1 H NMR (400 MHz, CDCl 3 ) δ 8.43 (d, J=4.7 Hz, 1H), 8.37 (d, J=7.2 Hz, 1H), 7.58-7.47 (m, 3H), 7.46-7.39 (m, 2H), 7.23 (dd, J=7.9, 4.8Hz, 1H), 6.78–6.55 (m, 3H), 4.77–4.61 (m, 2H), 3.91–3.73 (m, 11H), 2.89–2.81 (m, 2H). 13 C NMR (100MHz, CDCl 3 ) δ162.9, 162.6, 149.6, 148.6, 147.8, 147.8, 147.0, 143.6, 143.1, 130.7, 130.6, 130.4, 129.9, 129.3, 128.8, 126.3, 116.9, 7, 1 111.44,109.7,109.6,109.1,104.9,67.1,56.0,30.0.HRMS(ESI)calcd for C 28 H 26 FN 3 O 3 (M+H) + 472.2031,found 472.2034.
化合物5d的合成:Synthesis of compound 5d:
向化合物4d(182mg,0.6mmol)的CH2Cl2(5mL)溶液中加入草酰氯(228mg,1.8mmol),然后滴入DMF,室温搅拌2小时。真空浓缩,将残余物溶解在THF(10mL)中,0℃条件下,加入6,7-二甲氧基-1,2,3,4-四氢异喹啉盐酸盐(138mg,0.6mmol)和TEA(0.4mL,3mmol)的THF(20mL)溶液,在室温搅拌1小时,EA萃取,用饱和食盐水洗涤,干燥,浓缩,硅胶柱色谱纯化得浅黄色固体化合物5d(160mg,56%)。To a solution of compound 4d (182 mg, 0.6 mmol) in CH 2 Cl 2 (5 mL) was added oxalyl chloride (228 mg, 1.8 mmol), then DMF was added dropwise, and the mixture was stirred at room temperature for 2 hours. Concentrate in vacuo, dissolve the residue in THF (10 mL), add 6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline hydrochloride (138 mg, 0.6 mmol) at 0°C ) and TEA (0.4 mL, 3 mmol) in THF (20 mL), stirred at room temperature for 1 hour, extracted with EA, washed with saturated brine, dried, concentrated, and purified by silica gel column chromatography to give compound 5d (160 mg, 56%) as a pale yellow solid ).
1H NMR(400MHz,CDCl3)δ8.77(d,J=7.9Hz,1H),8.49(d,J=3.7Hz,1H),7.87(s,1H),7.64–7.54(m,3H),7.49–7.39(m,2H),7.35(dd,J=8.0,4.7Hz,1H),6.65(d,J=2.5Hz,2H),4.97–4.52(m,2H),3.97–3.79(m,11H),3.02-2.81(m,2H).13C NMR(100MHz,CDCl3)δ163.74,147.86,147.82,147.16,146.87,146.80,143.30,130.55,129.78,129.19,127.94,127.60,125.07,123.30,117.65,116.85,116.52,110.42,108.06,107.10,98.82,76.25,54.98,54.95,29.46,28.66,27.12.HRMS(ESI)calcd for C29H26N4O3(M+H)+479.2078,found479.2079. 1 H NMR (400 MHz, CDCl 3 ) δ 8.77 (d, J=7.9 Hz, 1H), 8.49 (d, J=3.7 Hz, 1H), 7.87 (s, 1H), 7.64-7.54 (m, 3H) ,7.49–7.39(m,2H),7.35(dd,J=8.0,4.7Hz,1H),6.65(d,J=2.5Hz,2H),4.97–4.52(m,2H),3.97–3.79(m , 11H), 3.02-2.81(m, 2H). 13 C NMR (100MHz, CDCl 3 )δ163.74,147.86,147.82,147.16,146.87,146.80,143.30,130.55,129.78,129.19,127.94,123.360,12 117.65,116.85,116.52,110.42,108.06,107.10,98.82,76.25,54.98,54.95,29.46,28.66,27.12.HRMS(ESI)calcd for C 29 H 26 N 4 O 3 (M+H) + 479.2078,found4 2079.
化合物SIS3的合成:Synthesis of compound SIS3:
向化合物4e(556mg,2mmol)的THF(20mL)溶液中加入草酰氯(508mg,4mmol),室温搅拌2小时。真空浓缩,将残余物溶解在THF(10mL)中,0℃条件下,加入6,7-二甲氧基-1,2,3,4-四氢异喹啉盐酸盐(460mg,2mmol)和TEA(606mg,6mmol)的THF(20mL)溶液,在室温搅拌1小时,EA萃取,用饱和食盐水洗涤,干燥,浓缩,硅胶柱色谱纯化得黄色固体化合物SIS3(500mg,55%)。To a solution of compound 4e (556 mg, 2 mmol) in THF (20 mL) was added oxalyl chloride (508 mg, 4 mmol), followed by stirring at room temperature for 2 hours. Concentrate in vacuo, dissolve the residue in THF (10 mL), add 6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline hydrochloride (460 mg, 2 mmol) at 0°C and TEA (606 mg, 6 mmol) in THF (20 mL), stirred at room temperature for 1 hour, extracted with EA, washed with saturated brine, dried, concentrated, and purified by silica gel column chromatography to give yellow solid compound SIS3 (500 mg, 55%).
1H NMR(400MHz,CDCl3)δ8.44(d,J=4.7Hz,1H),8.27(s,1H),7.77(d,J=15.4Hz,1H),7.59–7.49(m,3H),7.45(d,J=7.4Hz,2H),7.28–7.26(m,2H),6.87(d,J=15.4Hz,1H),6.64(s,2H),4.74–4.70(m,2H),3.87–3.78(m,11H),2.84(d,J=20.7Hz,2H).13C NMR(100MHz,CDCl3)δ166.8,148.9,147.8,144.4,143.6,136.0,130.7,129.9,129.4,128.8,128.3,126.1,118.5,117.1,113.6,111.2,109.5,109.1,56.0,47.1,44.4,43.7,40.0,29.9,29.2,28.2.HRMS(ESI)calcd for C28H27N3O3(M+H)+454.2125,found 454.2126. 1 H NMR (400 MHz, CDCl 3 ) δ 8.44 (d, J=4.7 Hz, 1H), 8.27 (s, 1H), 7.77 (d, J=15.4 Hz, 1H), 7.59-7.49 (m, 3H) ,7.45(d,J=7.4Hz,2H),7.28-7.26(m,2H),6.87(d,J=15.4Hz,1H),6.64(s,2H),4.74-4.70(m,2H), 3.87-3.78 (m, 11H), 2.84 (d, J=20.7Hz, 2H). 13 C NMR (100MHz, CDCl 3 ) δ 166.8, 148.9, 147.8, 144.4, 143.6, 136.0, 130.7, 129.9, 129.4, 128.8, 128.3,126.1,118.5,117.1,113.6,111.2,109.5,109.1,56.0,47.1,44.4,43.7,40.0,29.9,29.2,28.2.HRMS(ESI)calcd for C 28 H 27 N 3 O 3 (M+H ) + 454.2125, found 454.2126.
化合物5f的合成:Synthesis of compound 5f:
将化合物SIS3(220mg,0.48mmol)和Pd/C(22mg,10%w/w)放于反应瓶中,氢气置换,加MeOH(20mL)溶解,室温搅拌过夜。过滤所得混合物,蒸发滤液,得到粗产物,通过硅胶柱层析纯化得白色固体化合物5f(120mg,54%).Compound SIS3 (220 mg, 0.48 mmol) and Pd/C (22 mg, 10% w/w) were placed in a reaction flask, replaced with hydrogen, dissolved in MeOH (20 mL), and stirred at room temperature overnight. The resulting mixture was filtered and the filtrate was evaporated to give the crude product, which was purified by silica gel column chromatography to give compound 5f as a white solid (120 mg, 54%).
1H NMR(400MHz,CDCl3)δ8.34(d,J=4.3Hz,1H),7.95(d,J=7.7Hz,1H),7.53–7.40(m,5H),7.11–7.03(m,1H),6.59(d,J=8.5Hz,1H),6.55(s,0.5H),6.33(s,0.5H),4.59(s,1H),4.29(s,1H),3.85–3.82(m,6H),3.75(t,J=5.9Hz,1H),3.70–3.66(m,3H),3.39(t,J=5.8Hz,1H),3.14–3.07(m,2H),2.71–2.53(m,4H).13C NMR(100MHz,CDCl3)δ171.3,171.2,148.1,147.9,147.8,147.6,147.6,142.8,138.2,138.2,131.3,130.4,128.7,128.6,128.5,126.9,126.9,125.7,125.4,124.1,120.2,115.5,115.4,111.5,111.1,110.4,110.3,109.4,108.7,56.0,46.9,43.9,43.2,39.6,35.2,34.9,29.5,28.8,27.9,20.4,20.3.HRMS(ESI)calcd for C28H29N3O3(M+H)+456.2282,found456.2288. 1 H NMR (400 MHz, CDCl 3 ) δ 8.34 (d, J=4.3 Hz, 1H), 7.95 (d, J=7.7 Hz, 1H), 7.53-7.40 (m, 5H), 7.11-7.03 (m, 1H), 6.59(d, J=8.5Hz, 1H), 6.55(s, 0.5H), 6.33(s, 0.5H), 4.59(s, 1H), 4.29(s, 1H), 3.85–3.82(m ,6H),3.75(t,J=5.9Hz,1H),3.70-3.66(m,3H),3.39(t,J=5.8Hz,1H),3.14-3.07(m,2H),2.71-2.53( m, 4H). 13 C NMR (100MHz, CDCl 3 )δ171.3, 171.2, 148.1, 147.9, 147.8, 147.6, 147.6, 142.8, 138.2, 138.2, 131.3, 130.4, 128.7, 128.6, 128.5, 126., 9, 125.7.9, 125.4, 124.1, 120.2, 115.5, 115.4, 111.5, 111.1, 110.4, 110.3, 109.4, 108.7, 56.0, 46.9, 43.9, 43.2, 39.6, 35.2, 34.9, 29.5, 28.8, 27.9, 20.4, 20.3 calcd for C 28 H 29 N 3 O 3 (M+H) + 456.2282, found456.2288.
化合物5g的合成:Synthesis of compound 5g:
将化合物4e(200mg,0.72mmol)和草酰氯(254mg,2.00mmol)放于反应瓶中,加10mLTHF。室温搅拌1小时。减压浓缩得到中间体,加入1,2,3,4-四氢-6,7-异喹啉二醇氢溴酸盐(176mg,0.72mmol)和DIPEA(213mg,1.65mmol),加10mLTHF溶解。室温搅拌1.5小时。取样点板反应基本完毕,EA萃取三次,合并有机相,干燥,过滤,浓缩,硅胶柱层析纯化得化合物5g(130mg,42%)。Compound 4e (200 mg, 0.72 mmol) and oxalyl chloride (254 mg, 2.00 mmol) were placed in a reaction flask, and 10 mL of THF was added. Stir at room temperature for 1 hour. Concentrate under reduced pressure to obtain the intermediate, add 1,2,3,4-tetrahydro-6,7-isoquinolinediol hydrobromide (176 mg, 0.72 mmol) and DIPEA (213 mg, 1.65 mmol), add 10 mL of THF to dissolve . Stir at room temperature for 1.5 hours. The sampling point plate reaction was basically completed, EA was extracted three times, the organic phases were combined, dried, filtered, concentrated, and purified by silica gel column chromatography to obtain compound 5g (130 mg, 42%).
1H NMR(400MHz,DMSO-d6)δ8.79-8.74(m,1H),8.60(dd,J=7.6Hz,1H),8.43(d,J=4.0Hz,1H),7.67–7.59(m,3H),7.56–7.54(m,2H),7.48(d,J=15.4Hz,1H),7.33(s,1H),7.05(d,J=15.4Hz,1H),6.64–6.52(m,2H),4.58(d,J=86.5Hz,2H),3.83–3.69(m,5H),2.64(d,J=48.7Hz,2H).13C NMR(100MHz,DMSO-d6)δ165.94,148.69,144.59,144.34,144.23,144.09,135.14,131.22,129.88,129.53,129.22,125.38,124.59,124.23,117.85,115.62,114.18,114.01,113.70,108.75,55.38,44.25,30.01,28.99.HRMS(ESI)calcd forC26H23N3O3(M+H)+426.1812,found 426.1816. 1 H NMR (400MHz, DMSO-d 6 ) δ 8.79-8.74 (m, 1H), 8.60 (dd, J=7.6Hz, 1H), 8.43 (d, J=4.0Hz, 1H), 7.67-7.59 ( m, 3H), 7.56–7.54 (m, 2H), 7.48 (d, J=15.4Hz, 1H), 7.33 (s, 1H), 7.05 (d, J=15.4Hz, 1H), 6.64–6.52 (m , 2H), 4.58(d, J=86.5Hz, 2H), 3.83-3.69(m, 5H), 2.64(d, J=48.7Hz, 2H). 13 C NMR (100MHz, DMSO-d 6 )δ165. 94,148.69,144.59,144.34,144.23,144.09,135.14,131.22,129.88,129.53,129.22,125.38,124.59,124.23,117.85,115.62,114.18,114.01,113.70,108.75,55.38,44.25,30.01,28.99.HRMS(ESI) calcd for C 26 H 23 N 3 O 3 (M+H) + 426.1812, found 426.1816.
化合物5h的合成:Synthesis of compound 5h:
将化合物4e(200mg,0.72mmol)和草酰氯(254mg,2.0mmol)放于反应瓶中,加10mLTHF。室温搅拌1小时。减压浓缩得到中间体,加入6,7-双(2-甲氧基乙氧基)-1,2,3,4-四氢异喹啉盐酸盐(210mg,0.66mmol)和DIPEA(213mg,1.65mmol),加10mLTHF溶解。室温搅拌1.5小时。取样点板反应基本完毕,EA萃取三次,合并有机相,干燥,过滤,浓缩,硅胶柱层析纯化得黄色固体化合物5h(120mg,29%)。Compound 4e (200 mg, 0.72 mmol) and oxalyl chloride (254 mg, 2.0 mmol) were placed in a reaction flask, and 10 mL of THF was added. Stir at room temperature for 1 hour. Concentrate under reduced pressure to give the intermediate, add 6,7-bis(2-methoxyethoxy)-1,2,3,4-tetrahydroisoquinoline hydrochloride (210 mg, 0.66 mmol) and DIPEA (213 mg) , 1.65mmol), add 10mL THF to dissolve. Stir at room temperature for 1.5 hours. The sampling point plate reaction was basically completed, EA was extracted three times, the organic phases were combined, dried, filtered, concentrated, and purified by silica gel column chromatography to obtain a yellow solid compound 5h (120 mg, 29%).
1H NMR(400MHz,CDCl3)δ8.43(d,J=4.0Hz,1H),8.24(s,1H),7.76(d,J=15.4Hz,1H),7.56–7.50(m,3H),7.45–7.43(m,2H),7.25(s,1H),6.85(d,J=15.4Hz,1H),6.71(d,J=3.4Hz,2H),5.29(s,1H),4.69(d,J=12.4Hz,2H),4.22–4.04(m,4H),3.90–3.71(m,9H),3.45(s,6H),2.81(d,J=19.7Hz,2H).13C NMR(100MHz,CDCl3)δ166.7,148.8,148.1,147.7,144.4,143.5,135.9,130.7,129.9,129.4,128.8,128.3,127.1,118.6,117.1,115.2,113.6,113.2,109.1,77.3,71.1,69.0,59.2,53.5,47.1,44.3,43.6,40.0,29.9,29.2,28.1.HRMS(ESI)calcd for C32H35N3O5(M+H)+542.2649,found 542.2654. 1 H NMR (400 MHz, CDCl 3 ) δ 8.43 (d, J=4.0 Hz, 1H), 8.24 (s, 1H), 7.76 (d, J=15.4 Hz, 1H), 7.56-7.50 (m, 3H) ,7.45–7.43(m,2H),7.25(s,1H),6.85(d,J=15.4Hz,1H),6.71(d,J=3.4Hz,2H),5.29(s,1H),4.69( d, J=12.4Hz, 2H), 4.22–4.04 (m, 4H), 3.90–3.71 (m, 9H), 3.45 (s, 6H), 2.81 (d, J=19.7Hz, 2H). 13 C NMR (100MHz, CDCl 3 )δ166.7,148.8,148.1,147.7,144.4,143.5,135.9,130.7,129.9,129.4,128.8,128.3,127.1,118.6,117.1,115.2,113.6,113.2,109.1 59.2,53.5,47.1,44.3,43.6,40.0,29.9,29.2,28.1.HRMS(ESI)calcd for C 32 H 35 N 3 O 5 (M+H) + 542.2649,found 542.2654.
化合物5i的合成:Synthesis of compound 5i:
向化合物4f(120mg,0.432mmol)和TEA(0.24mL)的DMF(3mL)溶液中加入HATU(197mg,0.518mmol),0℃搅拌0.5小时,加入6,7-二甲氧基-1,2,3,4-四氢异喹啉盐酸盐(99mg,0.432mmol)。室温搅拌5小时,取样点板,反应基本完毕,加冰水稀释,抽滤,乙酸乙酯溶解,无水硫酸钠干燥,过滤浓缩,硅胶柱纯化得到相应的白色固体化合物5i(52mg,27%)。To a solution of compound 4f (120 mg, 0.432 mmol) and TEA (0.24 mL) in DMF (3 mL) was added HATU (197 mg, 0.518 mmol), stirred at 0°C for 0.5 h, and added 6,7-dimethoxy-1,2 , 3,4-Tetrahydroisoquinoline hydrochloride (99 mg, 0.432 mmol). Stir at room temperature for 5 hours, sample the plate, the reaction is basically complete, add ice water for dilution, suction filtration, dissolve in ethyl acetate, dry over anhydrous sodium sulfate, filter and concentrate, and purify with silica gel column to obtain the corresponding white solid compound 5i (52 mg, 27% ).
1H NMR(400MHz,CDCl3)δ7.98(s,1H),7.79(d,J=15.4Hz,1H),7.61–7.27(m,10H),6.91(d,J=15.4Hz,1H),6.63(s,2H),4.72(s,2H),3.85(s,6H),3.62(s,3H),2.85(s,2H).13C NMR(100MHz,CDCl3)δ167.3,147.8,144.5,138.0,136.8,131.0,130.5,129.1,128.8,125.8,122.9,121.4,120.4,113.0,112.7,111.6,111.3,110.9,110.2,109.6,109.1,56.1,56.1,44.5,43.8,31.2,29.3.HRMS(ESI)calcd for C29H28N2O3(M+H)+453.2173,found453.2169. 1 H NMR (400MHz, CDCl 3 ) δ 7.98 (s, 1H), 7.79 (d, J=15.4Hz, 1H), 7.61-7.27 (m, 10H), 6.91 (d, J=15.4Hz, 1H) , 6.63(s, 2H), 4.72(s, 2H), 3.85(s, 6H), 3.62(s, 3H), 2.85(s, 2H). 13 C NMR(100MHz, CDCl 3 )δ167.3,147.8,144.5 ,138.0,136.8,131.0,130.5,129.1,128.8,125.8,122.9,121.4,120.4,113.0,112.7,111.6,111.3,110.9,110.2,109.6,109.1,56.1,56.1,44.5,4 (ESI)calcd for C 29 H 28 N 2 O 3 (M+H) + 453.2173, found453.2169.
化合物5j的合成:Synthesis of compound 5j:
向化合物4g(200mg,0.72mmol)和6,7-二甲氧基-1,2,3,4-四氢异喹啉盐酸盐(165mg,0.72mmol)的无水DMF(3mL)溶液中加入DIPEA(1mL)和HATU(380mg,1mmol),搅拌16小时,加水稀释,用EA萃取,饱和食盐水洗涤,无水MgSO4干燥,过滤,浓缩,硅胶柱层析纯化得到黄色固体产物5j(90mg,27%)。To a solution of compound 4g (200 mg, 0.72 mmol) and 6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline hydrochloride (165 mg, 0.72 mmol) in dry DMF (3 mL) DIPEA (1 mL) and HATU (380 mg, 1 mmol) were added, stirred for 16 hours, diluted with water, extracted with EA, washed with saturated brine, dried over anhydrous MgSO 4 , filtered, concentrated, and purified by silica gel column chromatography to obtain yellow solid product 5j ( 90 mg, 27%).
1H NMR(400MHz,DMSO-d6)δ9.48(s,1H),8.65–8.47(m,2H),7.74–7.67(m,3H),7.64–7.56(m,2H),7.46(d,J=15.6Hz,1H),7.15(d,J=15.6Hz,1H),6.83–6.74(m,2H),4.78(s,1H),4.60(s,1H),3.86(s,3H),3.78–3.67(m,8H),2.81(s,1H),2.70(s,1H).13C NMR(100MHz,CDCl3)δ162.5,148.3,148.0,147.9,147.8,133.4,130.8,130.4,129.4,127.9,126.9,125.3,124.1,122.8,116.0,111.6,111.5,111.2,109.4,109.2,108.8,56.0,47.1,44.6,32.5,29.1.HRMS(ESI)calcd for C28H27N3O3(M+H)+454.2125,found 454.2129. 1 H NMR (400MHz, DMSO-d 6 )δ9.48(s,1H),8.65-8.47(m,2H),7.74-7.67(m,3H),7.64-7.56(m,2H),7.46(d , J=15.6Hz, 1H), 7.15(d, J=15.6Hz, 1H), 6.83–6.74(m, 2H), 4.78(s, 1H), 4.60(s, 1H), 3.86(s, 3H) , 3.78–3.67(m, 8H), 2.81(s, 1H), 2.70(s, 1H). 13 C NMR (100MHz, CDCl 3 )δ162.5, 148.3, 148.0, 147.9, 147.8, 133.4, 130.8, 130.4, 129.4 ( _ _ _ M+H) + 454.2125, found 454.2129.
实施例2:化合物9的合成Example 2: Synthesis of Compound 9
将化合物6(208mg,1mmol)和AlCl3(667mg,5mmol)置于反应瓶中,氮气置换,加20mLDCM溶解,室温搅拌1小时。加入2-氯-2-氧代乙酸甲酯(122mg,1mmol),0℃条件下搅拌0.5小时,然后升温至室温,并在N2保护条件下搅拌过夜。取样点板,反应基本完毕,萃取,DCM萃取三次,合并有机相,干燥,过滤,浓缩,硅胶柱层析纯化得黄色固体化合物7(270mg,92%);1H NMR(400MHz,CDCl3)δ8.71(d,J=7.8Hz,1H),8.49(d,J=4.7Hz,1H),7.63–7.53(m,3H),7.48(d,J=5.7Hz,2H),7.35(dd,J=7.8,4.8Hz,1H),3.74(s,3H),3.24(s,3H).13CNMR(100MHz,CDCl3)δ177.5,162.6,149.1,144.0,137.7,132.1,119.8,119.2,112.7,52.9.Compound 6 (208 mg, 1 mmol) and AlCl 3 (667 mg, 5 mmol) were placed in a reaction flask, replaced with nitrogen, dissolved in 20 mL of DCM, and stirred at room temperature for 1 hour. Methyl 2-chloro-2-oxoacetate (122 mg, 1 mmol) was added, stirred at 0 °C for 0.5 h, then warmed to room temperature and stirred overnight under N2 protection. Sampling plate, the reaction was basically completed, extracted, extracted with DCM three times, the organic phases were combined, dried, filtered, concentrated, and purified by silica gel column chromatography to obtain yellow solid compound 7 (270 mg, 92%); 1 H NMR (400 MHz, CDCl 3 ) δ8.71(d,J=7.8Hz,1H),8.49(d,J=4.7Hz,1H),7.63-7.53(m,3H),7.48(d,J=5.7Hz,2H),7.35(dd , J=7.8, 4.8Hz, 1H), 3.74(s, 3H), 3.24(s, 3H). 13 CNMR (100MHz, CDCl 3 )δ177.5, 162.6, 149.1, 144.0, 137.7, 132.1, 119.8, 119.2, 112.7 , 52.9.
将化合物7(270mg,0.92mmol)和LiOH(66mg,2.76mmol)放于反应瓶中,加MeOH(10mL)和水(5mL)溶解,室温搅拌过夜。取样点板,反应基本完毕,将混合物在真空下浓缩,EA(2×30mL)萃取,合并有机层,干燥,过滤,浓缩,硅胶柱层析纯化得白色固体化合物8(230mg,89%);1H NMR(400MHz,CDCl3)δ8.71(d,J=7.6Hz,1H),8.40(d,J=4.3Hz,1H),7.65–7.48(m,5H),7.39–7.31(m,1H),3.69(s,3H).13C NMR(100MHz,CDCl3)δ182.0,165.8,150.5,147.8,144.7,131.4,130.4,129.1,128.5,119.7,119.5,109.2,30.1.Compound 7 (270 mg, 0.92 mmol) and LiOH (66 mg, 2.76 mmol) were placed in a reaction flask, MeOH (10 mL) and water (5 mL) were added to dissolve, and the mixture was stirred at room temperature overnight. Sampling plate, the reaction was basically completed, the mixture was concentrated under vacuum, extracted with EA (2×30 mL), the organic layers were combined, dried, filtered, concentrated, and purified by silica gel column chromatography to obtain compound 8 (230 mg, 89%) as a white solid; 1 H NMR (400 MHz, CDCl 3 ) δ 8.71 (d, J=7.6 Hz, 1H), 8.40 (d, J=4.3 Hz, 1H), 7.65-7.48 (m, 5H), 7.39-7.31 (m, 1H), 3.69(s, 3H). 13 C NMR (100MHz, CDCl 3 )δ182.0, 165.8, 150.5, 147.8, 144.7, 131.4, 130.4, 129.1, 128.5, 119.7, 119.5, 109.2, 30.1.
将化合物8(230mg,0.82mmol),6,7-二甲氧基-1,2,3,4-四氢异喹啉盐酸盐(228mg,0.98mmol),DIPEA(222mg,1.72mmol)和HATU(373mg,0.98mmol)放于反应瓶中,加DMF(5mL)溶解,室温搅拌8小时。取样点板,反应基本完毕。真空浓缩,EA萃取,干燥,过滤,浓缩,硅胶柱层析纯化得白色固体化合物9(134mg,36%);1H NMR(400MHz,CDCl3)δ8.77(dd,J=18.0,7.7Hz,1H),8.48(d,J=4.4Hz,1H),7.50–7.42(m,3H),7.39–7.32(m,1H),7.30–7.22(m,2H),6.56(d,J=5.0Hz,1H),6.45(s,1H),4.37(s,1H),4.01(s,1H),3.84(dd,J=19.4,15.6Hz,6H),3.65(d,J=26.2Hz,3H),3.45(t,J=5.3Hz,1H),3.30(t,J=6.0Hz,1H),2.73(t,J=5.2Hz,1H),2.39(t,J=5.8Hz,1H).13C NMR(100MHz,CDCl3)δ187.3,165.8,149.3,148.1,147.9,147.8,144.8,131.2,130.2,128.4,126.5,125.5,123.7,123.5,119.4,111.4,110.4,109.2,108.8,56.03,55.98,55.95,46.9,43.5,42.5,38.7,29.7,28.5,27.1.HRMS(ESI)calcd for C27H25N3O4(M+H)+456.1918,found 456.1921.Compound 8 (230 mg, 0.82 mmol), 6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline hydrochloride (228 mg, 0.98 mmol), DIPEA (222 mg, 1.72 mmol) and HATU (373 mg, 0.98 mmol) was placed in a reaction flask, DMF (5 mL) was added to dissolve, and the mixture was stirred at room temperature for 8 hours. Sampling point plate, the reaction is basically completed. Concentrated in vacuo, extracted with EA, dried, filtered, concentrated, and purified by silica gel column chromatography to obtain compound 9 (134 mg, 36%) as a white solid; 1 H NMR (400 MHz, CDCl 3 ) δ 8.77 (dd, J=18.0, 7.7 Hz) ,1H),8.48(d,J=4.4Hz,1H),7.50-7.42(m,3H),7.39-7.32(m,1H),7.30-7.22(m,2H),6.56(d,J=5.0 Hz, 1H), 6.45(s, 1H), 4.37(s, 1H), 4.01(s, 1H), 3.84(dd, J=19.4, 15.6Hz, 6H), 3.65(d, J=26.2Hz, 3H ), 3.45(t, J=5.3Hz, 1H), 3.30(t, J=6.0Hz, 1H), 2.73(t, J=5.2Hz, 1H), 2.39(t, J=5.8Hz, 1H). 13 C NMR (100 MHz, CDCl 3 ) δ 187.3, 165.8, 149.3, 148.1, 147.9, 147.8, 144.8, 131.2, 130.2, 128.4, 126.5, 125.5, 123.7, 123.5, 119.4, 111.4, 110.4, 108.8, 9 ,55.95,46.9,43.5,42.5,38.7,29.7,28.5,27.1.HRMS(ESI)calcd for C 27 H 25 N 3 O 4 (M+H) + 456.1918,found 456.1921.
实施例3:化合物12的合成Example 3: Synthesis of Compound 12
将NaH(112mg,2.8mmol)放于反应瓶中,置换氮气,加入5mL无水DMF溶解,在冰水浴的条件下加入膦酰基乙酸甲酯二乙酯(555mg,2.2mmol),搅拌0.5小时,逐滴加入化合物10(451mg,2.0mmol)的DMF溶液,室温搅拌8小时。取样点板,反应基本完毕,加水淬灭反应,搅拌2小时,乙酸乙酯萃取三次,饱和食盐水洗涤,干燥,过滤,浓缩,硅胶柱纯化得到黄色中间体n7(542mg,84%)。NaH (112 mg, 2.8 mmol) was placed in a reaction flask, nitrogen was replaced, 5 mL of anhydrous DMF was added to dissolve, methyl phosphonoacetate diethyl ester (555 mg, 2.2 mmol) was added in an ice-water bath, and the mixture was stirred for 0.5 hours. A solution of compound 10 (451 mg, 2.0 mmol) in DMF was added dropwise and stirred at room temperature for 8 hours. Sampling and spotting, the reaction was basically completed, water was added to quench the reaction, stirred for 2 hours, extracted with ethyl acetate three times, washed with saturated brine, dried, filtered, concentrated, and purified by silica gel column to obtain yellow intermediate n7 (542 mg, 84%).
将中间体n7(542mg,1.68mmol)溶解在CH2Cl2(9mL)和TFA(2mL)混合溶液中,室温搅拌过夜,取样点板,反应基本完毕,得化合物11(550mg,粗产)。The intermediate n7 (542 mg, 1.68 mmol) was dissolved in a mixed solution of CH 2 Cl 2 (9 mL) and TFA (2 mL), stirred at room temperature overnight, sampled and spotted, and the reaction was basically completed to obtain compound 11 (550 mg, crude product).
向化合物11(150mg,0.565mmol)和TEA(0.5mL,3.6mmol)的DMF(3.5mL)溶液中加入HATU(258mg,0.678mmol),0℃搅拌0.5小时,加入6,7-二甲氧基-1,2,3,4-四氢异喹啉盐酸盐(130mg,0.565mmol)。室温搅拌5小时,点板反应基本完毕,加冰水稀释,抽滤,硅胶柱纯化得到化合物12(234mg,94%);1H NMR(400MHz,MeOH)δ9.39–9.26(m,1H),8.89(d,J=3.5Hz,1H),7.65(t,J=10.6Hz,3H),7.52(t,J=6.0Hz,4H),7.14(dd,J=15.6,9.7Hz,1H),6.64–6.48(m,2H),4.54(d,J=16.8Hz,2H),3.71(dd,J=7.6,3.6Hz,1H),3.65(d,J=7.7Hz,6H),3.62–3.55(m,1H),2.67(d,J=5.1Hz,2H).13C NMR(100MHz,MeOH)δ164.9,164.9,156.8,148.1,148.0,147.8,144.8,138.7,138.5,136.2,136.1,130.79,130.75,129.3,129.2,127.2,127.2,126.7,126.2,124.7,124.5,124.2,122.7,122.6,117.8,113.6,111.6,111.5,109.6,109.4,55.2,55.10,55.05,44.3,43.6,40.6,28.5,27.4.HRMS(ESI)calcdfor C26H24N4O3(M+H)+441.1921,found 441.1921.To a solution of compound 11 (150 mg, 0.565 mmol) and TEA (0.5 mL, 3.6 mmol) in DMF (3.5 mL) was added HATU (258 mg, 0.678 mmol), stirred at 0°C for 0.5 h, and added 6,7-dimethoxy - 1,2,3,4-Tetrahydroisoquinoline hydrochloride (130 mg, 0.565 mmol). After stirring at room temperature for 5 hours, the spotting reaction was basically completed, diluted with ice water, filtered with suction, and purified by silica gel column to obtain compound 12 (234 mg, 94%); 1 H NMR (400 MHz, MeOH) δ9.39–9.26 (m, 1H) ,8.89(d,J=3.5Hz,1H),7.65(t,J=10.6Hz,3H),7.52(t,J=6.0Hz,4H),7.14(dd,J=15.6,9.7Hz,1H) ,6.64–6.48(m,2H),4.54(d,J=16.8Hz,2H),3.71(dd,J=7.6,3.6Hz,1H),3.65(d,J=7.7Hz,6H),3.62– 3.55(m, 1H), 2.67(d, J=5.1Hz, 2H). 13 C NMR (100MHz, MeOH) δ 164.9, 164.9, 156.8, 148.1, 148.0, 147.8, 144.8, 138.7, 138.5, 136.2, 136.1, 130.79 ,130.75,129.3,129.2,127.2,127.2,126.7,126.2,124.7,124.5,124.2,122.7,122.6,117.8,113.6,111.6,111.5,109.6,109.4,55.2,55.10,55.5 , 27.4. HRMS(ESI) calcd for C 26 H 24 N 4 O 3 (M+H) + 441.1921, found 441.1921.
实施例4:化合物13a-13h,化合物14a-14b的合成Example 4: Synthesis of compounds 13a-13h, compounds 14a-14b
化合物13a的合成:Synthesis of compound 13a:
向化合物4e(63mg,0.227mmol)和TEA(0.095mL)的DMF(0.915mL)溶液中加入HATU(104mg,0.273mmol),0℃搅拌0.5小时,加入L-苯丙氨酸甲酯盐酸盐(49mg,0.227mmol)。室温搅拌5小时,取样点板,反应基本完毕,加冰水稀释,抽滤,乙酸乙酯溶解,无水硫酸钠干燥,过滤浓缩,硅胶柱纯化得到白色固体化合物13a(59mg,59%)。To a solution of compound 4e (63 mg, 0.227 mmol) and TEA (0.095 mL) in DMF (0.915 mL) was added HATU (104 mg, 0.273 mmol), stirred at 0°C for 0.5 h, and added L-phenylalanine methyl ester hydrochloride (49 mg, 0.227 mmol). Stir at room temperature for 5 hours, sample and spot plate, the reaction is basically complete, add ice water for dilution, suction filtration, dissolve in ethyl acetate, dry over anhydrous sodium sulfate, filter and concentrate, and purify with silica gel column to obtain compound 13a (59 mg, 59%) as a white solid.
1H NMR(400MHz,CDCl3)δ8.40(dd,J=4.8,1.5Hz,1H),8.19(dd,J=7.9,1.5Hz,1H),7.68(d,J=15.6Hz,1H),7.56–7.49(m,3H),7.44–7.39(m,2H),7.30–7.26(m,2H),7.25–7.17(m,2H),7.11(dd,J=7.9,1.5Hz,2H),6.39(d,J=15.6Hz,1H),6.07(d,J=7.7Hz,1H),5.01(dt,J=7.7,5.7Hz,1H),3.74(s,3H),3.73(s,3H),3.17(t,J=6.1Hz,2H).13C NMR(100MHz,CDCl3)δ172.4,166.5,149.0,145.2,143.7,136.1,135.3,130.8,129.6,129.5,129.4,128.9,128.7,128.6,127.1,118.4,117.3,115.8,108.6,53.4,52.4,38.1,30.0.HRMS(ESI)calcd for C27H25N3O3(M+H)+440.1969,found 440.1965. 1 H NMR (400MHz, CDCl 3 ) δ 8.40 (dd, J=4.8, 1.5Hz, 1H), 8.19 (dd, J=7.9, 1.5Hz, 1H), 7.68 (d, J=15.6Hz, 1H) ,7.56–7.49(m,3H),7.44–7.39(m,2H),7.30–7.26(m,2H),7.25–7.17(m,2H),7.11(dd,J=7.9,1.5Hz,2H) ,6.39(d,J=15.6Hz,1H),6.07(d,J=7.7Hz,1H),5.01(dt,J=7.7,5.7Hz,1H),3.74(s,3H),3.73(s, 3H), 3.17 (t, J=6.1Hz, 2H). 13 C NMR (100MHz, CDCl 3 )δ172.4, 166.5, 149.0, 145.2, 143.7, 136.1, 135.3, 130.8, 129.6, 129.5, 129.4, 128.9, 128.7, 128.6,127.1,118.4,117.3,115.8,108.6,53.4,52.4,38.1,30.0.HRMS(ESI)calcd for C 27 H 25 N 3 O 3 (M+H) + 440.1969,found 440.1965.
化合物13b的合成:Synthesis of compound 13b:
向化合物4e(120mg,0.432mmol)和TEA(0.18mL)的DMF(1.8mL)溶液中加入HATU(164mg,0.432mmol),0℃搅拌0.5小时,加入D-苯丙氨酸甲酯盐酸盐(93mg,0.432mmol)。室温搅拌5小时,取样点板,反应基本完毕,加冰水稀释,抽滤,乙酸乙酯溶解,无水硫酸钠干燥,过滤浓缩,硅胶柱纯化得到白色固体化合物13b(85mg,44%)。To a solution of compound 4e (120 mg, 0.432 mmol) and TEA (0.18 mL) in DMF (1.8 mL) was added HATU (164 mg, 0.432 mmol), stirred at 0°C for 0.5 h, and added D-phenylalanine methyl ester hydrochloride (93 mg, 0.432 mmol). Stir at room temperature for 5 hours, sample and spot plate, the reaction is basically complete, add ice water for dilution, suction filtration, dissolve in ethyl acetate, dry over anhydrous sodium sulfate, filter and concentrate, and purify with silica gel column to obtain compound 13b (85 mg, 44%) as a white solid.
1H NMR(400MHz,CDCl3)δ8.40(dd,J=4.8,1.4Hz,1H),8.20(dd,J=7.9,1.1Hz,1H),7.66(d,J=15.6Hz,1H),7.56–7.46(m,3H),7.40(ddd,J=6.3,4.4,2.7Hz,2H),7.23–7.17(m,2H),7.12–7.07(m,2H),6.37(d,J=15.6Hz,1H),4.99(dt,J=7.7,5.7Hz,1H),3.73(s,3H),3.72(s,3H),3.22–3.10(m,2H).13C NMR(100MHz,CDCl3)δ172.4,166.5,148.9,145.2,143.6,136.1,135.3,130.8,129.6,129.6,129.4,128.9,128.7,128.6,127.1,118.5,117.3,115.9,108.6,53.4,52.4,38.1,30.0.HRMS(ESI)calcd for C27H25N3O3(M+H)+440.1969,found 440.1966. 1 H NMR (400 MHz, CDCl 3 ) δ 8.40 (dd, J=4.8, 1.4 Hz, 1H), 8.20 (dd, J=7.9, 1.1 Hz, 1H), 7.66 (d, J=15.6 Hz, 1H) ,7.56–7.46(m,3H),7.40(ddd,J=6.3,4.4,2.7Hz,2H),7.23–7.17(m,2H),7.12–7.07(m,2H),6.37(d,J= 15.6Hz, 1H), 4.99 (dt, J=7.7, 5.7Hz, 1H), 3.73 (s, 3H), 3.72 (s, 3H), 3.22–3.10 (m, 2H). 13 C NMR (100MHz, CDCl) 3 ) δ172.4, 166.5, 148.9, 145.2, 143.6, 136.1, 135.3, 130.8, 129.6, 129.6, 129.4, 128.9, 128.7, 128.6, 127.1, 118.5, 117.3, 115.9, 108.6, 38.4, 52 RMS( ESI) calcd for C 27 H 25 N 3 O 3 (M+H) + 440.1969, found 440.1966.
化合物13c的合成:Synthesis of compound 13c:
向化合物4e(120mg,0.432mmol)和TEA(0.18mL)的DMF(1.8mL)溶液中加入HATU(164mg,0.432mmol),0℃搅拌0.5小时,加入L-酪氨酸甲酯(84mg,0.432mmol)。室温搅拌5小时,取样点板,反应基本完毕,加冰水稀释,抽滤,乙酸乙酯溶解,无水硫酸钠干燥,过滤浓缩,硅胶柱纯化得到白色固体化合物13c(141mg,72%)。To a solution of compound 4e (120 mg, 0.432 mmol) and TEA (0.18 mL) in DMF (1.8 mL) was added HATU (164 mg, 0.432 mmol), stirred at 0°C for 0.5 h, and added L-tyrosine methyl ester (84 mg, 0.432 mmol). mmol). Stir at room temperature for 5 hours, sample and spot plate, the reaction is basically complete, add ice water for dilution, suction filtration, dissolve in ethyl acetate, dry over anhydrous sodium sulfate, filter and concentrate, and purify with silica gel column to obtain compound 13c (141 mg, 72%) as a white solid.
1H NMR(400MHz,CDCl3)δ8.36(dd,J=4.8,1.4Hz,1H),8.15(dd,J=8.0,1.4Hz,1H),7.65(d,J=15.6Hz,1H),7.49–7.43(m,3H),7.38–7.33(m,2H),7.15(dd,J=7.9,4.8Hz,1H),6.90(d,J=8.5Hz,2H),6.68(d,J=8.5Hz,2H),6.39(d,J=15.6Hz,1H),6.16(d,J=7.8Hz,1H),4.95(dt,J=7.7,5.7Hz,1H),3.70(s,3H),3.69(s,3H),3.06(qd,J=14.0,5.7Hz,2H).13C NMR(100MHz,CDCl3)δ171.5,165.7,154.7,147.7,144.3,142.5,134.4,129.6,129.3,128.5,128.3,127.8,126.0,117.5,116.3,114.7,114.6,107.4,52.5,51.4,36.2,29.0.HRMS(ESI)calcd for C27H25N3O4(M+H)+456.1918,found 456.1914. 1 H NMR (400 MHz, CDCl 3 ) δ 8.36 (dd, J=4.8, 1.4 Hz, 1H), 8.15 (dd, J=8.0, 1.4 Hz, 1H), 7.65 (d, J=15.6 Hz, 1H) ,7.49–7.43(m,3H),7.38–7.33(m,2H),7.15(dd,J=7.9,4.8Hz,1H),6.90(d,J=8.5Hz,2H),6.68(d,J =8.5Hz, 2H), 6.39(d, J=15.6Hz, 1H), 6.16(d, J=7.8Hz, 1H), 4.95(dt, J=7.7, 5.7Hz, 1H), 3.70(s, 3H ), 3.69(s, 3H), 3.06(qd, J=14.0, 5.7Hz, 2H). 13 C NMR (100MHz, CDCl 3 )δ171.5, 165.7, 154.7, 147.7, 144.3, 142.5, 134.4, 129.6, 129.3, 128.5,128.3,127.8,126.0,117.5,116.3,114.7,114.6,107.4,52.5,51.4,36.2,29.0.HRMS(ESI)calcd for C 27 H 25 N 3 O 4 (M+H) + 456.1918,found 456.1914 .
化合物13d的合成:Synthesis of compound 13d:
将化合物13a(55mg,0.125mmol)和LiOH·H2O(78mg,1.859mmol)放于反应瓶中,加H2O/MeOH(2mL/2mL)混合溶液溶解,室温搅拌4小时。用HCl(1M)调节pH=5,过滤收集白色固体化合物13d(49mg,92%)。Compound 13a (55 mg, 0.125 mmol) and LiOH·H 2 O (78 mg, 1.859 mmol) were placed in a reaction flask, and a mixed solution of H 2 O/MeOH (2 mL/2 mL) was added to dissolve, and stirred at room temperature for 4 hours. The pH=5 was adjusted with HCl (1 M) and the white solid compound 13d (49 mg, 92%) was collected by filtration.
HNMR(400MHz,CDCl3)δ8.44–7.79(m,3H),7.50(d,J=15.6Hz,1H),7.38(s,3H),7.09(t,J=29.1Hz,9H),6.43(d,J=6.8Hz,1H),6.27(d,J=15.5Hz,1H),4.89(d,J=4.8Hz,1H),3.56(s,3H),3.11(d,J=32.7Hz,2H).13C NMR(100MHz,CDCl3)δ174.2,167.4,148.6,145.3,143.4,136.2,135.6,130.7,129.6,129.4,129.3,128.9,128.8,128.6,127.0,118.5,117.3,115.5,108.5,53.9,37.4,30.0.HRMS(ESI)calcd for C26H23N3O3(M+H)+426.1812,found 426.1810.HNMR (400MHz, CDCl3 )δ8.44-7.79(m,3H),7.50(d,J=15.6Hz,1H),7.38(s,3H),7.09(t,J=29.1Hz,9H),6.43 (d, J=6.8Hz, 1H), 6.27 (d, J=15.5Hz, 1H), 4.89 (d, J=4.8Hz, 1H), 3.56 (s, 3H), 3.11 (d, J=32.7Hz) , 2H). 13 C NMR (100MHz, CDCl 3 )δ174.2,167.4,148.6,145.3,143.4,136.2,135.6,130.7,129.6,129.4,129.3,128.9,128.8,128.6,127.0,118.5,115.3,118.5,115.3, ,53.9,37.4,30.0.HRMS(ESI)calcd for C 26 H 23 N 3 O 3 (M+H) + 426.1812,found 426.1810.
化合物13e的合成:Synthesis of compound 13e:
将化合物13b(70mg,0.159mmol)和LiOH·H2O(52mg,1.239mmol)放于反应瓶中,加H2O/MeOH(1mL/1mL)混合溶液溶解,室温搅拌4小时。用HCl(1M)调节pH=5,过滤收集白色固体化合物13e(66mg,98%)。Compound 13b (70 mg, 0.159 mmol) and LiOH·H 2 O (52 mg, 1.239 mmol) were placed in a reaction flask, and a mixed solution of H 2 O/MeOH (1 mL/1 mL) was added to dissolve, and stirred at room temperature for 4 hours. The pH=5 was adjusted with HCl (1 M) and the white solid compound 13e (66 mg, 98%) was collected by filtration.
1H NMR(400MHz,CDCl3)δ8.39(dd,J=4.7,1.2Hz,1H),8.15(d,J=7.2Hz,1H),7.65(d,J=15.6Hz,1H),7.57–7.47(m,3H),7.39(dd,J=7.1,2.2Hz,2H),7.26–7.15(m,6H),6.35(d,J=15.6Hz,1H),6.12(d,J=3.4Hz,1H),4.92(d,J=5.7Hz,1H),3.72(s,3H),3.23(ddd,J=20.1,14.0,5.6Hz,2H).13C NMR(100MHz,CDCl3)δ167.3,148.3,145.3,143.2,136.1,135.4,130.7,129.5,129.4,129.2,129.1,128.8,128.5,127.0,118.6,117.2,115.5,108.5,67.9,37.4,30.1.HRMS(ESI)calcd for C26H23N3O3(M+H)+426.1812,found426.1808. 1 H NMR (400 MHz, CDCl 3 ) δ 8.39 (dd, J=4.7, 1.2 Hz, 1H), 8.15 (d, J=7.2 Hz, 1H), 7.65 (d, J=15.6 Hz, 1H), 7.57 –7.47(m,3H),7.39(dd,J=7.1,2.2Hz,2H),7.26–7.15(m,6H),6.35(d,J=15.6Hz,1H),6.12(d,J=3.4 Hz, 1H), 4.92 (d, J=5.7Hz, 1H), 3.72 (s, 3H), 3.23 (ddd, J=20.1, 14.0, 5.6Hz, 2H). 13 C NMR (100MHz, CDCl 3 )δ167 .3,148.3,145.3,143.2,136.1,135.4,130.7,129.5,129.4,129.2,129.1,128.8,128.5,127.0,118.6,117.2,115.5,108.5,67.9,37.4,30.1 6 H.HRMS (ESIcalc) 23 N 3 O 3 (M+H) + 426.1812, found426.1808.
化合物13f的合成:Synthesis of compound 13f:
将化合物13c(42mg,0.092mmol)和LiOH(16mg,0.691mmol)放于反应瓶中,加THF/H2O(2mL/2mL)混合溶液溶解,室温搅拌4小时。用HCl(1M)调节pH=5,过滤收集白色固体化合物13f(40mg,98%)。Compound 13c (42 mg, 0.092 mmol) and LiOH (16 mg, 0.691 mmol) were placed in a reaction flask, THF/H 2 O (2 mL/2 mL) mixed solution was added to dissolve, and the mixture was stirred at room temperature for 4 hours. The pH=5 was adjusted with HCl (1 M) and the white solid compound 13f (40 mg, 98%) was collected by filtration.
1H NMR(400MHz,MeOH)δ8.38(dd,J=8.0,1.4Hz,1H),8.30(d,J=1.5Hz,1H),7.62–7.36(m,7H),7.24(dd,J=7.9,4.8Hz,1H),7.02(d,J=8.4Hz,2H),6.64(m,J=15.1,12.1Hz,3H),4.58–4.50(m,1H),3.66(s,3H),3.02(ddd,J=20.7,13.8,6.0Hz,2H).13C NMR(100MHz,MeOH)δ177.2,167.4,155.5,148.5,144.7,143.0,133.0,130.7,130.2,129.6,129.2,129.1,129.0,128.6,118.6,117.5,117.1,114.6,108.7,56.4,37.3,28.9.HRMS(ESI)calcd for C26H23N3O4(M+H)+442.1761,found 442.1763. 1 H NMR (400MHz, MeOH) δ 8.38 (dd, J=8.0, 1.4Hz, 1H), 8.30 (d, J=1.5Hz, 1H), 7.62-7.36 (m, 7H), 7.24 (dd, J =7.9,4.8Hz,1H),7.02(d,J=8.4Hz,2H),6.64(m,J=15.1,12.1Hz,3H),4.58–4.50(m,1H),3.66(s,3H) , 3.02 (ddd, J=20.7, 13.8, 6.0 Hz, 2H). 13 C NMR (100 MHz, MeOH) δ 177.2, 167.4, 155.5, 148.5, 144.7, 143.0, 133.0, 130.7, 130.2, 129.6, 129.2, 129.1, 129.0 ,128.6,118.6,117.5,117.1,114.6,108.7,56.4,37.3,28.9.HRMS(ESI)calcd for C 26 H 23 N 3 O 4 (M+H) + 442.1761,found 442.1763.
化合物13g的合成:Synthesis of compound 13g:
向化合物4e(100mg,0.360mmol)和TEA(0.15mL)的DMF(1.5mL)溶液中加入HATU(137mg,0.360mmol),0℃搅拌0.5小时,加入4-氯-D-苯丙氨酸甲酯盐酸盐(90mg,0.360mmol)。室温搅拌5小时,取样点板,反应基本完毕,加冰水稀释,抽滤,乙酸乙酯溶解,无水硫酸钠干燥,过滤浓缩,硅胶柱纯化得到白色固体化合物13g(164mg,96%)。To a solution of compound 4e (100 mg, 0.360 mmol) and TEA (0.15 mL) in DMF (1.5 mL) was added HATU (137 mg, 0.360 mmol), stirred at 0°C for 0.5 h, and added 4-chloro-D-phenylalanine methyl Ester hydrochloride (90 mg, 0.360 mmol). Stir at room temperature for 5 hours, sample and spot plate, the reaction is basically complete, add ice water for dilution, suction filtration, dissolve in ethyl acetate, dry over anhydrous sodium sulfate, filter and concentrate, and purify with silica gel column to obtain 13 g (164 mg, 96%) of white solid compound.
1H NMR(400MHz,CDCl3)δ8.40(dd,J=4.8,1.5Hz,1H),8.20(dd,J=7.9,1.5Hz,1H),7.66(d,J=15.6Hz,1H),7.56–7.46(m,3H),7.41(ddd,J=6.3,4.4,2.6Hz,2H),7.24–7.18(m,3H),7.08–6.96(m,2H),6.37(d,J=15.6Hz,1H),6.00(d,J=7.4Hz,1H),4.97(dd,J=13.1,5.6Hz,1H),3.73(s,3H),3.72(s,3H),3.14(qd,J=13.9,5.6Hz,2H).13C NMR(100MHz,CDCl3)δ172.2,166.5,148.9,145.3,143.7,135.5,134.6,133.1,130.8,130.8,129.6,129.6,128.9,128.8,128.7,118.4,117.4,115.6,108.5,53.3,52.5,37.5,30.0.HRMS(ESI)calcd for C27H24ClN3O3(M+H)+474.1579,found 474.1577. 1 H NMR (400MHz, CDCl 3 ) δ 8.40 (dd, J=4.8, 1.5Hz, 1H), 8.20 (dd, J=7.9, 1.5Hz, 1H), 7.66 (d, J=15.6Hz, 1H) ,7.56–7.46(m,3H),7.41(ddd,J=6.3,4.4,2.6Hz,2H),7.24–7.18(m,3H),7.08–6.96(m,2H),6.37(d,J= 15.6Hz, 1H), 6.00(d, J=7.4Hz, 1H), 4.97(dd, J=13.1, 5.6Hz, 1H), 3.73(s, 3H), 3.72(s, 3H), 3.14(qd, J=13.9, 5.6Hz, 2H). 13 C NMR (100MHz, CDCl 3 )δ172.2, 166.5, 148.9, 145.3, 143.7, 135.5, 134.6, 133.1, 130.8, 130.8, 129.6, 129.6, 128.9, 128.8, 128.7, 118 , 117.4, 115.6, 108.5, 53.3, 52.5, 37.5, 30.0. HRMS(ESI) calcd for C 27 H 24 ClN 3 O 3 (M+H) + 474.1579, found 474.1577.
化合物13h的合成:Synthesis of compound 13h:
向化合物4e(93mg,0.334mmol)和TEA(0.23mL)的DMF(2.5mL)溶液中加入HATU(127mg,0.334mmol),0℃搅拌0.5小时,加入n15(78mg,0.334mmol)。室温搅拌5小时,取样点板,反应基本完毕,加冰水稀释,抽滤,乙酸乙酯溶解,无水硫酸钠干燥,过滤浓缩,硅胶柱纯化得到白色固体化合物13h(45mg,27%)。To a solution of compound 4e (93 mg, 0.334 mmol) and TEA (0.23 mL) in DMF (2.5 mL) was added HATU (127 mg, 0.334 mmol), stirred at 0°C for 0.5 h, and added n15 (78 mg, 0.334 mmol). Stir at room temperature for 5 hours, sample and spot plate, the reaction is basically complete, add ice water for dilution, suction filtration, dissolve in ethyl acetate, dry over anhydrous sodium sulfate, filter and concentrate, and purify with silica gel column to obtain white solid compound 13h (45mg, 27%).
1H NMR(400MHz,MeOH)δ8.97(dd,J=8.0,1.2Hz,1H),8.56(dd,J=5.8,1.2Hz,1H),7.71(dd,J=8.0,5.8Hz,1H),7.68–7.61(m,3H),7.54(ddd,J=5.5,2.9,1.6Hz,2H),7.46(d,J=16.0Hz,1H),7.32–7.16(m,6H),6.90(d,J=16.0Hz,1H),4.54(dd,J=8.5,6.7Hz,1H),3.82(s,3H),3.63(ddd,J=15.1,7.1,4.8Hz,1H),3.41–3.32(m,2H),3.27–3.12(m,3H),3.03(dd,J=13.8,8.5Hz,1H),2.87(d,J=0.6Hz,6H).13C NMR(100MHz,MeOH)δ174.9,169.0,148.3,143.5,138.2,137.1,133.4,133.3,132.1,131.7,130.3,129.7,129.2,128.0,123.9,120.6,118.4,111.6,58.4,57.3,44.0,43.9,38.5,35.8,31.9.HRMS(ESI)calcd for C30H33N5O2(M+H)+496.2707,found 496.2706. 1 H NMR (400MHz, MeOH) δ 8.97 (dd, J=8.0, 1.2Hz, 1H), 8.56 (dd, J=5.8, 1.2Hz, 1H), 7.71 (dd, J=8.0, 5.8Hz, 1H) ),7.68–7.61(m,3H),7.54(ddd,J=5.5,2.9,1.6Hz,2H),7.46(d,J=16.0Hz,1H),7.32–7.16(m,6H),6.90( d, J=16.0Hz, 1H), 4.54 (dd, J=8.5, 6.7Hz, 1H), 3.82 (s, 3H), 3.63 (ddd, J=15.1, 7.1, 4.8Hz, 1H), 3.41–3.32 (m, 2H), 3.27–3.12 (m, 3H), 3.03 (dd, J=13.8, 8.5Hz, 1H), 2.87 (d, J=0.6Hz, 6H). 13 C NMR (100MHz, MeOH) δ174 .9, 169.0, 148.3, 143.5, 138.2, 137.1, 133.4, 133.3, 132.1, 131.7, 130.3, 129.7, 129.2, 128.0, 123.9, 120.6, 118.4, 111.6, 58.4, 57.3, 44.0, 3.5. (ESI)calcd for C 30 H 33 N 5 O 2 (M+H) + 496.2707, found 496.2706.
化合物14a的合成:Synthesis of compound 14a:
向化合物4e(102.5mg,0.369mmol)和TEA(0.155mL)的DMF(2mL)溶液中加入HATU(140mg,0.369mmol),0℃搅拌0.5小时,加入2-(1-哌嗪基)嘧啶(60.51mg,0.367mmol)。室温搅拌5小时,取样点板,反应基本完毕,加冰水稀释,抽滤,乙酸乙酯溶解,无水硫酸钠干燥,过滤浓缩,硅胶柱纯化得到白色固体化合物14a(52mg,33%)。To a solution of compound 4e (102.5 mg, 0.369 mmol) and TEA (0.155 mL) in DMF (2 mL) was added HATU (140 mg, 0.369 mmol), stirred at 0°C for 0.5 h, added 2-(1-piperazinyl)pyrimidine ( 60.51 mg, 0.367 mmol). Stir at room temperature for 5 hours, sample and spot plate, the reaction is basically completed, add ice water for dilution, suction filtration, dissolve in ethyl acetate, dry over anhydrous sodium sulfate, filter and concentrate, and purify with silica gel column to obtain compound 14a (52 mg, 33%) as a white solid.
1H NMR(400MHz,CDCl3)δ8.40(dd,J=4.7,1.4Hz,1H),8.31(d,J=4.8Hz,2H),8.19(dd,J=7.9,1.4Hz,1H),7.76(d,J=15.3Hz,1H),7.58–7.34(m,5H),7.22(dd,J=7.8,4.7Hz,1H),6.77(d,J=15.4Hz,1H),6.51(t,J=4.8Hz,1H),3.86(s,4H),3.73(s,7H).13CNMR(100MHz,CDCl3)δ166.7,161.6,157.9,148.9,144.6,143.7,136.5,130.8,129.9,129.5,128.9,128.3,118.6,117.3,112.9,110.5,109.1,43.8,29.9,29.4.HRMS(ES)calcdfor C25H24N6O(M+H)+425.2084,found 425.2078. 1 H NMR (400 MHz, CDCl 3 ) δ 8.40 (dd, J=4.7, 1.4 Hz, 1H), 8.31 (d, J=4.8 Hz, 2H), 8.19 (dd, J=7.9, 1.4 Hz, 1H) ,7.76(d,J=15.3Hz,1H),7.58–7.34(m,5H),7.22(dd,J=7.8,4.7Hz,1H),6.77(d,J=15.4Hz,1H),6.51( t, J=4.8Hz, 1H), 3.86(s, 4H), 3.73(s, 7H). 13 CNMR (100MHz, CDCl 3 ) δ 166.7, 161.6, 157.9, 148.9, 144.6, 143.7, 136.5, 130.8, 129.9, 129.5,128.9,128.3,118.6,117.3,112.9,110.5,109.1,43.8,29.9,29.4.HRMS(ES)calcdfor C 25 H 24 N 6 O(M+H) + 425.2084,found 425.2078.
化合物14b的合成:Synthesis of compound 14b:
向化合物4e(120mg,0.432mmol)和TEA(0.18mL)的DMF(1.8mL)溶液中加入HATU(164mg,0.432mmol),0℃搅拌0.5小时,加入1-(4-吡啶基)哌嗪(70.5mg,0.443mmol)。室温搅拌5小时,取样点板,反应基本完毕,加冰水稀释,抽滤,乙酸乙酯溶解,无水硫酸钠干燥,过滤浓缩,硅胶柱纯化得到白色固体化合物14b(163mg,89%)。To a solution of compound 4e (120 mg, 0.432 mmol) and TEA (0.18 mL) in DMF (1.8 mL) was added HATU (164 mg, 0.432 mmol), stirred at 0°C for 0.5 h, added 1-(4-pyridyl)piperazine ( 70.5 mg, 0.443 mmol). Stir at room temperature for 5 hours, sample and spot plate, the reaction is basically complete, add ice water for dilution, suction filtration, dissolve in ethyl acetate, dry over anhydrous sodium sulfate, filter and concentrate, and purify with silica gel column to obtain compound 14b (163 mg, 89%) as a white solid.
1H NMR(400MHz,DMSO-d6)δ8.58(dd,J=7.9,1.4Hz,1H),8.44(dd,J=4.7,1.4Hz,1H),8.26(d,J=7.2Hz,2H),7.66–7.59(m,3H),7.57–7.54(m,2H),7.51(d,J=15.4Hz,1H),7.33(dd,J=7.9,4.7Hz,1H),7.09(d,J=7.4Hz,2H),7.04(d,J=15.4Hz,1H),3.71(s,3H),3.70(s,8H).13C NMR(100MHz,DMSO-d6)δ166.1,156.4,148.8,145.1,144.3,143.2,135.8,131.3,130.0,129.8,129.6,129.3,117.9,117.9,113.3,108.8,108.2,49.3,30.1,27.4.HRMS(ESI)calcd for C26H25N5O(M+H)+424.2132,found 424.2128. 1 H NMR (400MHz, DMSO-d 6 ) δ 8.58 (dd, J=7.9, 1.4Hz, 1H), 8.44 (dd, J=4.7, 1.4Hz, 1H), 8.26 (d, J=7.2Hz, 2H), 7.66–7.59 (m, 3H), 7.57–7.54 (m, 2H), 7.51 (d, J=15.4Hz, 1H), 7.33 (dd, J=7.9, 4.7Hz, 1H), 7.09 (d , J=7.4Hz, 2H), 7.04(d, J=15.4Hz, 1H), 3.71(s, 3H), 3.70(s, 8H). 13 C NMR(100MHz, DMSO-d 6 )δ166.1,156.4, 148.8,145.1,144.3,143.2,135.8,131.3,130.0,129.8,129.6,129.3,117.9,117.9,113.3,108.8,108.2,49.3,30.1,27.4.HRMS(ESI)calcd for C 26 H 25 N 5 O( M+H) + 424.2132, found 424.2128.
实施例5:化合物16a-16d的合成Example 5: Synthesis of Compounds 16a-16d
中间体15a的合成:Synthesis of Intermediate 15a:
0℃条件下,向3c(4.3g,18.2mmol)的THF(30mL)溶液中逐滴加入LDA(18.2mL,36.4mmol)的THF溶液。升温至室温搅拌1小时,加1-乙酰基吡咯烷-2-酮(9.25g,72.9mmol),45℃下搅拌48小时。取样点板反应基本完毕,加饱和NaHCO3淬灭。加NH4Cl(3mL)和水(50mL)稀释,EA萃取,饱和食盐水(50mL)洗涤,无水Na2SO4干燥,浓缩,硅胶柱色谱纯化得白色固体化合物15a(2.3mg,42%);1H NMR(400MHz,CDCl3)δ8.42(dd,J=4.7,1.4Hz,1H),8.03(dd,J=7.9,1.4Hz,1H),7.61–7.37(m,6H),7.18(dd,J=7.9,4.7Hz,1H),6.86(s,1H),3.82(s,3H),3.32(t,J=6.6Hz,2H),2.47–2.27(m,2H).13C NMR(100MHz,CDCl3)δ172.9,148.4,143.4,141.3,131.0,130.6,128.9,128.8,128.0,123.1,119.7,116.5,108.5,39.7,29.9,26.9.To a solution of 3c (4.3 g, 18.2 mmol) in THF (30 mL) at 0°C was added LDA (18.2 mL, 36.4 mmol) in THF dropwise. The temperature was raised to room temperature and stirred for 1 hour, 1-acetylpyrrolidin-2-one (9.25 g, 72.9 mmol) was added, and the mixture was stirred at 45° C. for 48 hours. The reaction of the sampling point plate was basically completed, and was quenched by adding saturated NaHCO 3 . It was diluted with NH 4 Cl (3 mL) and water (50 mL), extracted with EA, washed with saturated brine (50 mL), dried over anhydrous Na 2 SO 4 , concentrated, and purified by silica gel column chromatography to obtain compound 15a (2.3 mg, 42%) as a white solid. ); 1 H NMR (400 MHz, CDCl 3 ) δ 8.42 (dd, J=4.7, 1.4 Hz, 1H), 8.03 (dd, J=7.9, 1.4 Hz, 1H), 7.61-7.37 (m, 6H), 7.18(dd,J=7.9,4.7Hz,1H),6.86(s,1H),3.82(s,3H),3.32(t,J=6.6Hz,2H),2.47–2.27(m,2H). 13 C NMR (100MHz, CDCl 3 ) δ 172.9, 148.4, 143.4, 141.3, 131.0, 130.6, 128.9, 128.8, 128.0, 123.1, 119.7, 116.5, 108.5, 39.7, 29.9, 26.9.
化合物16a的合成:Synthesis of compound 16a:
向化合物15a(450mg,1.5mmol)的THF(30mL)溶液中分批加入NaH(80mg,2mmoL),0℃下搅拌0.5小时。加入4-(溴甲基)-1,2-二甲氧基苯(416mg,1.8mmol),室温搅拌5小时,取样点板反应基本完毕,EA萃取,用饱和食盐水洗涤,无水Na2SO4干燥,浓缩,硅胶柱色谱纯化得浅黄色固体化合物16a(210mg,31%)。To a solution of compound 15a (450 mg, 1.5 mmol) in THF (30 mL) was added NaH (80 mg, 2 mmol) in portions and stirred at 0°C for 0.5 h. 4-(bromomethyl)-1,2-dimethoxybenzene (416 mg, 1.8 mmol) was added, stirred at room temperature for 5 hours, the sampling and spot plate reaction was basically completed, extracted with EA, washed with saturated brine, anhydrous Na 2 Dry over SO 4 , concentrated, and purified by silica gel column chromatography to give compound 16a (210 mg, 31%) as a pale yellow solid.
1H NMR(400MHz,CDCl3)δ8.40(d,J=4.7Hz,1H),8.02(d,J=7.9Hz,1H),7.55(t,J=2.7Hz,1H),7.53–7.40(m,5H),7.15(dd,J=7.7,4.8Hz,1H),6.82–6.77(m,3H),4.47(s,2H),3.86(s,6H),3.79(s,3H),3.12(t,J=6.6Hz,2H),2.27–2.18(m,2H).13C NMR(100MHz,CDCl3)δ169.3,149.2,148.5,148.4,143.4,141.1,131.1,130.6,129.7,129.2,128.9,128.8,128.0,122.9,120.8,119.7,116.4,111.5,111.0,108.5,56.0,55.9,47.1,43.9,29.9,24.6.HRMS(ESI)calcd for C28H27N3O3(M+H)+454.2125,found 454.2127. 1 H NMR (400 MHz, CDCl 3 ) δ 8.40 (d, J=4.7 Hz, 1H), 8.02 (d, J=7.9 Hz, 1H), 7.55 (t, J=2.7 Hz, 1H), 7.53-7.40 (m, 5H), 7.15(dd, J=7.7, 4.8Hz, 1H), 6.82–6.77(m, 3H), 4.47(s, 2H), 3.86(s, 6H), 3.79(s, 3H), 3.12(t, J=6.6Hz, 2H), 2.27-2.18(m, 2H). 13 C NMR (100 MHz, CDCl 3 ) δ 169.3, 149.2, 148.5, 148.4, 143.4, 141.1, 131.1, 130.6, 129.7, 129.2, 128.9,128.8,128.0,122.9,120.8,119.7,116.4,111.5,111.0,108.5,56.0,55.9,47.1,43.9,29.9,24.6.HRMS(ESI)calcd for C 28 H 27 N 3 O 3 (M+H ) + 454.2125, found 454.2127.
化合物16b的合成:Synthesis of compound 16b:
以化合物3c(300mg,1.27mmol)和1-乙酰基哌啶-2-酮(716mg,5.08mmol)为原料,参照化合物16a的合成方法和步骤,制备得到浅黄色固体化合物16b(60mg,13%)。Using compound 3c (300 mg, 1.27 mmol) and 1-acetylpiperidin-2-one (716 mg, 5.08 mmol) as raw materials, referring to the synthetic method and procedure of compound 16a, a pale yellow solid compound 16b (60 mg, 13%) was prepared ).
1H NMR(400MHz,CDCl3)δ8.36(d,J=0.4Hz,1H),7.94(s,1H),7.57-7.52(m,6H),7.20(s,1H),6.93-6.78(m,3H),4.52(s,2H),3.75(d,J=0.8Hz,9H),3.27(s,2H),2.42(s,2H),1.66(s,2H).13C NMR(100MHz,CDCl3)δ165.3,149.2,148.4,143.3,140.8,128.2,120.6,116.3,111.5,111.0,108.6,56.0,55.9,50.8,47.2,30.0,27.4,23.1.HRMS(ESI)calcd for C29H29N3O3(M+H)+468.2282,found 468.2285. 1 H NMR (400MHz, CDCl 3 ) δ 8.36(d, J=0.4Hz, 1H), 7.94(s, 1H), 7.57-7.52(m, 6H), 7.20(s, 1H), 6.93-6.78( m, 3H), 4.52(s, 2H), 3.75(d, J=0.8Hz, 9H), 3.27(s, 2H), 2.42(s, 2H), 1.66(s, 2H). 13 C NMR(100MHz) , CDCl 3 )δ165.3,149.2,148.4,143.3,140.8,128.2,120.6,116.3,111.5,111.0,108.6,56.0,55.9,50.8,47.2,30.0,27.4,23.1.HRMS(ESI)calcd for C 29 H 29 N 3 O 3 (M+H) + 468.2282, found 468.2285.
化合物16c的合成:Synthesis of compound 16c:
以化合物15a(100mg,0.330mmol)和2-氯甲基-3,5-二甲基-4-甲氧基吡啶(50mg,0.224mmol)为原料,参照化合物16a的合成方法和步骤,制备得到浅黄色固体化合物16c(19mg,12%)。Using compound 15a (100 mg, 0.330 mmol) and 2-chloromethyl-3,5-dimethyl-4-methoxypyridine (50 mg, 0.224 mmol) as raw materials, referring to the synthetic method and procedure of compound 16a, the obtained Light yellow solid compound 16c (19 mg, 12%).
1H NMR(400MHz,MeOH)δ8.62(d,J=7.7Hz,1H),8.51(d,J=5.4Hz,1H),8.43(s,1H),7.65–7.53(m,6H),7.42(s,1H),4.81(s,2H),4.14(s,3H),3.86(s,3H),3.44(t,J=5.4Hz,2H),2.58(s,2H),2.47(s,3H),2.38(s,3H).13C NMR(100MHz,MeOH)δ171.6,170.8,149.0,143.9,142.6,140.6,136.6,134.4,131.1,130.6,129.9,129.2,129.1,129.0,123.1,122.2,116.4,109.7,60.9,45.3,43.6,30.4,24.7,13.2,10.2.HRMS(ESI)calcd forC28H28N4O2(M+H)+453.2285,found 453.2281. 1 H NMR(400MHz,MeOH)δ8.62(d,J=7.7Hz,1H),8.51(d,J=5.4Hz,1H),8.43(s,1H),7.65-7.53(m,6H), 7.42(s, 1H), 4.81(s, 2H), 4.14(s, 3H), 3.86(s, 3H), 3.44(t, J=5.4Hz, 2H), 2.58(s, 2H), 2.47(s ,3H),2.38(s,3H). 13 C NMR(100MHz,MeOH)δ171.6,170.8,149.0,143.9,142.6,140.6,136.6,134.4,131.1,132.6,129.9,129.2,129.1,129.0,123.1,122. ,116.4,109.7,60.9,45.3,43.6,30.4,24.7,13.2,10.2.HRMS(ESI)calcd forC 28 H 28 N 4 O 2 (M+H) + 453.2285,found 453.2281.
化合物16d的合成:Synthesis of
以化合物15a(500mg,1.65mmol)和2-甲磺酰氧甲基-3,5,6-三甲基吡嗪n16(891mg,3.869mmol)为原料,参照化合物16a的合成方法和步骤,制备得到浅黄色固体化合物16d(76mg,11%)。Using compound 15a (500mg, 1.65mmol) and 2-methanesulfonyloxymethyl-3,5,6-trimethylpyrazine n16 (891mg, 3.869mmol) as raw materials, referring to the synthetic method and steps of compound 16a,
1H NMR(400MHz,CDCl3)δ8.41(dd,J=4.7,1.5Hz,1H),8.03(dd,J=7.9,1.5Hz,1H),7.56(t,J=2.7Hz,1H),7.54–7.41(m,5H),7.17(dd,J=7.9,4.7Hz,1H),4.68(s,2H),3.81(s,3H),3.21(t,J=6.6Hz,2H),2.54(s,3H),2.50(s,3H),2.47(s,3H),2.26(td,J=6.6,2.7Hz,2H).;13C NMR(100MHz,CDCl3)δ169.3,150.3,148.9,148.5,145.8,143.4,141.1,131.1,130.6,129.2,128.9,128.8,128.0,123.2,119.7,116.5,108.5,46.5,44.6,29.9,24.8,21.6,21.5,20.7;HRMS(ESI)calcd for C27H27N5O(M+H)+438.2288,found438.2295. 1 H NMR (400MHz, CDCl3) δ 8.41 (dd, J=4.7, 1.5Hz, 1H), 8.03 (dd, J=7.9, 1.5Hz, 1H), 7.56 (t, J=2.7Hz, 1H), 7.54–7.41(m, 5H), 7.17(dd, J=7.9, 4.7Hz, 1H), 4.68(s, 2H), 3.81(s, 3H), 3.21(t, J=6.6Hz, 2H), 2.54 (s, 3H), 2.50 (s, 3H), 2.47 (s, 3H), 2.26 (td, J=6.6, 2.7 Hz, 2H).; 13 C NMR (100 MHz, CDCl3) δ 169.3, 150.3, 148.9, 148.5 , 145.8,143.4,141.1,131.1,130.6,129.2,128.9,128.8,128.0,123.2,119.7,116.5,108.5,46.5,44.6,29.9,24.8,21.6,21.5,20.7 ; 27 N 5 O(M+H) + 438.2288, found438.2295.
实施例6:氮杂吲哚类化合物或其盐的药理作用Example 6: Pharmacological effects of azaindoles or their salts
荧光素酶报告基因筛选实验,实验步骤如下:a)将携带SMAD3报告基因的腺病毒感染A549细胞24h。b)以空白或待测化合物(5μM和10μM)预处理细胞2h,然后用0.1ng/mL TGF-β刺激细胞24h。c)将细胞裂解,将荧光素酶底物加入到细胞裂解物中,测试荧光素酶的活性,通过将待测化合物的荧光素酶活性与空白组的荧光素酶活性相比,得到抑制SMAD3磷酸化的水平。结果如表1所示。Luciferase reporter gene screening experiment, the experimental steps are as follows: a) Infect A549 cells with adenovirus carrying the SMAD3 reporter gene for 24 hours. b) Cells were pretreated with blank or test compound (5 μM and 10 μM) for 2 h, and then stimulated with 0.1 ng/mL TGF-β for 24 h. c) Lysing the cells, adding the luciferase substrate to the cell lysate, testing the luciferase activity, and comparing the luciferase activity of the test compound with the luciferase activity of the blank group to obtain the inhibition of SMAD3 phosphorylation level. The results are shown in Table 1.
表1氮杂吲哚类化合物对SMAD3磷酸化的抑制活性(抑制率%,μM)Table 1 Inhibitory activity of azaindoles on SMAD3 phosphorylation (inhibition rate %, μM)
由上述活性测试结果表明:本发明的化合物对受试细胞显示出抑制活性,能够有效抑制SMAD3蛋白的磷酸化。The above activity test results show that the compounds of the present invention show inhibitory activity on the test cells, and can effectively inhibit the phosphorylation of SMAD3 protein.
实施例7:氮杂吲哚类化合物或其盐的药学性质Example 7: Pharmaceutical properties of azaindoles or their salts
在25℃下,将化合物SIS3、化合物16d、化合物13h、化合物5b或它们的HCl盐(将化合物溶于4当量HCl的甲醇溶液,然后旋干溶剂,真空干燥制备得到)分别添加到纯水或磷酸盐缓冲液(50mM,pH 7.4)中。摇动并离心后,取上清液检测溶液中化合物SIS3、化合物16d、化合物13h、或者化合物5b的浓度,以计算相应的溶解度。检测方法:用HPLC(Agilent 1260Infinity)检测,色谱柱为Shimadzu-GL WondaSil C18-WR,流动相为乙腈-水-TFA(68/32/1‰),检测波长为254nm,进样量为10μL。At 25°C, compound SIS3,
结果如表2所示:本发明的氮杂吲哚类化合物具有较好的水溶性,化合物16d、化合物13h和化合物5b的水溶性远好于现有的SMAD3小分子抑制剂化合物SIS3,化合物16d、化合物13h和化合物5b的盐酸盐在水中的溶解度分别是化合物SIS3·HCl在水中溶解度的667倍、163倍、723倍。The results are shown in Table 2: the azaindole compounds of the present invention have good water solubility, and the water solubility of
表2溶解度测试结果Table 2 Solubility test results
实施例8:化合物16d选择性抑制SMAD3的活化Example 8:
A549细胞不用或用药物(1μM和5μM)预处理2h,然后用1ng/mL TGF-β刺激30分钟,裂解细胞并进行蛋白质印迹分析,与phospho-SMAD3或phospho-SMAD2抗体和相应的二抗(LI-COR IRDye 800标记)一起孵育以进行信号检测后,用剥离缓冲液(25mM甘氨酸-HCl,pH2,1%SDS)洗涤膜30分钟,然后重新用SMAD3或SMAD2一抗检测,结果如图1所示:化合物16d能选择性抑制SMAD3蛋白的活化,但是没有抑制SMAD2蛋白的活化。A549 cells were pretreated without or with drugs (1 μM and 5 μM) for 2 h, then stimulated with 1 ng/mL TGF-β for 30 min, cells were lysed and subjected to Western blot analysis, with phospho-SMAD3 or phospho-SMAD2 antibodies and corresponding secondary antibodies ( After incubation with LI-COR IRDye 800 labeled with LI-COR IRDye 800 for signal detection, the membrane was washed with stripping buffer (25mM Glycine-HCl, pH2, 1% SDS) for 30 minutes, and then re-detected with SMAD3 or SMAD2 primary antibody, the results are shown in Figure 1 Shown:
实施例9:水溶性化合物16d在B1鼠中LLC移植肿瘤模型的抗癌活性Example 9: Anticancer activity of water-
C57BL/6小鼠皮下接种2×106LLC细胞(CRL-1642,ATCC)。当肿瘤大小达到50mm3时,将小鼠随机分为4组,每组6只,并接受化合物16d(0、1.25、2.5或5.0μg/g/天,用生理盐水配制注射液,腹腔注射)治疗20天。每5天测量一次肿瘤体积,并在实验结束时测量肿瘤重量,结果如图2所示,水溶性的化合物16d在小鼠体内治疗以剂量依赖性方式显着降低了LLC肿瘤的体积和重量。C57BL/6 mice were subcutaneously inoculated with 2×10 6 LLC cells (CRL-1642, ATCC). When the tumor size reached 50 mm, the mice were randomly divided into 4 groups of 6 mice and received
接下来,通过小鼠的病理学切片研究化合物16d减弱小鼠肺癌进展的机制。结果如图3所示,化合物16d治疗的小鼠以剂量依赖性方式增加了肿瘤浸润NK细胞的数量,与对照小鼠相比,以2.5或5.0μg/g的剂量进行16天治疗显着增加了NK细胞的积累,这表明用化合物16d治疗肿瘤的小鼠存在增强的抗肿瘤免疫反应。Next, the mechanism by which
这些结果表明,化合物16d可通过增强LLC模型小鼠中的NK细胞介导的抗癌免疫性来显著抑制癌症进展。These results suggest that
以上所述实施例的各技术特征可以进行任意的组合,为使描述简洁,未对以下实施例中的各个技术特征所有可能的组合都进行描述,然而,只要这些技术特征的组合不存在矛盾,都应当认为是本说明书记载的范围。The technical features of the above-described embodiments can be combined arbitrarily. For the sake of brevity, all possible combinations of the technical features in the following embodiments are not described. However, as long as there is no contradiction between the combinations of these technical features, All should be regarded as the scope described in this specification.
以上所述实施例仅表达了本发明的几种实施方式,其描述较为具体和详细,但并不能因此而理解为对本发明专利范围的限制。应当指出的是,对于本领域的普通技术人员来说,在不脱离本发明构思的前提下,还可以做出若干变形和改进,这些都属于本发明的保护范围。因此,本发明专利的保护范围应以所附权利要求为准。The above-mentioned embodiments only represent several embodiments of the present invention, and the descriptions thereof are specific and detailed, but should not be construed as a limitation on the scope of the patent of the present invention. It should be pointed out that for those of ordinary skill in the art, without departing from the concept of the present invention, several modifications and improvements can also be made, which all belong to the protection scope of the present invention. Therefore, the protection scope of the patent of the present invention should be subject to the appended claims.
Claims (18)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010265070.5A CN111606904B (en) | 2020-04-07 | 2020-04-07 | Azaindoles and their applications |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010265070.5A CN111606904B (en) | 2020-04-07 | 2020-04-07 | Azaindoles and their applications |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111606904A true CN111606904A (en) | 2020-09-01 |
| CN111606904B CN111606904B (en) | 2021-10-15 |
Family
ID=72197642
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010265070.5A Active CN111606904B (en) | 2020-04-07 | 2020-04-07 | Azaindoles and their applications |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111606904B (en) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112794851A (en) * | 2021-01-26 | 2021-05-14 | 复旦大学 | 3-(pyridin-3yl)-7-azaindole derivative PI3Kδ inhibitor and preparation method and application thereof |
| CN114280308A (en) * | 2021-11-16 | 2022-04-05 | 中国科学院合肥物质科学研究院 | Application of Smad3 protein as a new target for ovarian cancer resistance to cisplatin |
| US11426412B2 (en) | 2017-10-18 | 2022-08-30 | Jubilant Epipad LLC | Imidazo-pyridine compounds as PAD inhibitors |
| US11459338B2 (en) | 2017-11-24 | 2022-10-04 | Jubilant Episcribe Llc | Heterocyclic compounds as PRMT5 inhibitors |
| US11529341B2 (en) | 2018-03-13 | 2022-12-20 | Jubilant Prodel LLC | Bicyclic compounds as inhibitors of PD1/PD-L1 interaction/activation |
| US11629135B2 (en) | 2017-11-06 | 2023-04-18 | Jubilant Prodell Llc | Pyrimidine derivatives as inhibitors of PD1/PD-L1 activation |
| US11833156B2 (en) | 2017-09-22 | 2023-12-05 | Jubilant Epipad LLC | Heterocyclic compounds as pad inhibitors |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008022467A1 (en) * | 2006-08-25 | 2008-02-28 | Mark Lautens | 2-substituted azain doles and 2 substituted thienopyrroles, their precursors and novel processes for the preparation thereof |
| CN103435612A (en) * | 2013-08-30 | 2013-12-11 | 四川好医生药业集团有限公司 | Compound for treating diabetes mellitus |
-
2020
- 2020-04-07 CN CN202010265070.5A patent/CN111606904B/en active Active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008022467A1 (en) * | 2006-08-25 | 2008-02-28 | Mark Lautens | 2-substituted azain doles and 2 substituted thienopyrroles, their precursors and novel processes for the preparation thereof |
| CN103435612A (en) * | 2013-08-30 | 2013-12-11 | 四川好医生药业集团有限公司 | Compound for treating diabetes mellitus |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11833156B2 (en) | 2017-09-22 | 2023-12-05 | Jubilant Epipad LLC | Heterocyclic compounds as pad inhibitors |
| US12357639B2 (en) | 2017-09-22 | 2025-07-15 | Jubilant Epipad LLC | Heterocyclic compounds as pad inhibitors |
| US11426412B2 (en) | 2017-10-18 | 2022-08-30 | Jubilant Epipad LLC | Imidazo-pyridine compounds as PAD inhibitors |
| US11629135B2 (en) | 2017-11-06 | 2023-04-18 | Jubilant Prodell Llc | Pyrimidine derivatives as inhibitors of PD1/PD-L1 activation |
| US11459338B2 (en) | 2017-11-24 | 2022-10-04 | Jubilant Episcribe Llc | Heterocyclic compounds as PRMT5 inhibitors |
| US11529341B2 (en) | 2018-03-13 | 2022-12-20 | Jubilant Prodel LLC | Bicyclic compounds as inhibitors of PD1/PD-L1 interaction/activation |
| CN112794851A (en) * | 2021-01-26 | 2021-05-14 | 复旦大学 | 3-(pyridin-3yl)-7-azaindole derivative PI3Kδ inhibitor and preparation method and application thereof |
| CN114280308A (en) * | 2021-11-16 | 2022-04-05 | 中国科学院合肥物质科学研究院 | Application of Smad3 protein as a new target for ovarian cancer resistance to cisplatin |
Also Published As
| Publication number | Publication date |
|---|---|
| CN111606904B (en) | 2021-10-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN111606904A (en) | Azaindoles and their applications | |
| CN113166110B (en) | 2-aminopyrimidine compounds and their applications | |
| CN101885722B (en) | Heterocycle alkyne and benzene compound, medicinal composition and application thereof | |
| CN105601573B (en) | 2-Aminopyrimidine compound and its pharmaceutical composition and application | |
| CN103012399B (en) | 7-oxopyridinopyrimidine compound as well as medicinal composition and application thereof | |
| WO2023246903A1 (en) | Selenium-containing heterocyclic compound, pharmaceutical composition thereof and use thereof | |
| CN106467541A (en) | Substituted quinolone analog derivative or its pharmaceutically acceptable salt or stereoisomer and its Pharmaceutical composition and application | |
| CN106866571B (en) | Heterocyclic urea compound and its pharmaceutical composition and application | |
| CN110305161A (en) | 2-aminopyrimidine compounds and their applications | |
| CN101747330A (en) | Pyridyl thiazole amine compound and medicinal composition and application thereof | |
| CN111989329B (en) | Crystal form of mono-mesylate of deuterated 3- (4, 5-substituted aminopyrimidine) phenyl compound and preparation method thereof | |
| CN112313213A (en) | 3-amino pyrazole compound and application thereof | |
| CN112351971B (en) | Quinoline or quinazoline compound and application thereof | |
| CN110437220B (en) | Triazole compound and application thereof | |
| CN112538038B (en) | 2-indolone ERR alpha inverse agonist and pharmaceutical composition and application thereof | |
| CN114539263B (en) | Nitrogen-containing heterocyclic compound, and pharmaceutical composition and application thereof | |
| CN112851667B (en) | Nitrogen-containing heterocyclic compounds and pharmaceutical compositions and applications thereof | |
| CN118084902B (en) | 2-Aminothiazole pyrimidopyridone compound or pharmaceutically acceptable salt or stereoisomer, preparation method and application | |
| CN101298427B (en) | Diaryl urea compound and use thereof | |
| CN111875583A (en) | Triazole derivatives and their preparation methods and uses | |
| CN115703734B (en) | Diarylmethylpyridine compounds and preparation method and application thereof | |
| CN115772167A (en) | Compounds containing urea structure and applications thereof | |
| CN116003400A (en) | 2-formyl tetrahydronaphthyridine compound and medicinal composition and application thereof | |
| HK1234742B (en) | 2-aminopyrimidine compound and pharmaceutical composition and use thereof | |
| HK40003079A (en) | Azabenzimidazole derivatives as pi3k beta inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CB03 | Change of inventor or designer information | ||
| CB03 | Change of inventor or designer information |
Inventor after: Yu Xiyong Inventor after: Hu Wenhui Inventor after: Yang Zhongjin Inventor after: Wu Nannan Inventor before: Yu Xiyong Inventor before: Lan Huiyao Inventor before: Hu Wenhui Inventor before: Yang Zhongjin Inventor before: Wu Nannan |