CN111560436A - TERT mutation detection kit and application thereof in noninvasive tumor diagnosis - Google Patents
TERT mutation detection kit and application thereof in noninvasive tumor diagnosis Download PDFInfo
- Publication number
- CN111560436A CN111560436A CN202010467657.4A CN202010467657A CN111560436A CN 111560436 A CN111560436 A CN 111560436A CN 202010467657 A CN202010467657 A CN 202010467657A CN 111560436 A CN111560436 A CN 111560436A
- Authority
- CN
- China
- Prior art keywords
- tert
- detecting
- mutation
- detection
- pcr
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
- C12Q1/6883—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material
- C12Q1/6886—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material for cancer
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/118—Prognosis of disease development
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/156—Polymorphic or mutational markers
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Engineering & Computer Science (AREA)
- Immunology (AREA)
- Pathology (AREA)
- Analytical Chemistry (AREA)
- Zoology (AREA)
- Genetics & Genomics (AREA)
- Wood Science & Technology (AREA)
- Physics & Mathematics (AREA)
- Biotechnology (AREA)
- Microbiology (AREA)
- Molecular Biology (AREA)
- Hospice & Palliative Care (AREA)
- Biophysics (AREA)
- Oncology (AREA)
- Biochemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Abstract
The invention particularly relates to a TERT mutation detection kit and application thereof in tumor noninvasive diagnosis. The invention provides a primer combination, a kit and a method for detecting TERT gene promoter mutation based on a ddPCR method and application (ddPCR-TERTpm kit) so as to solve the problems of complex detection and low sensitivity in the prior art. The ddPCR-TERTpm kit is suitable for detecting tumor and urine mutation TERT promoters of patients with bladder and renal pelvis cancer. The ddPCR amplification analysis is carried out after the urine DNA is extracted, and the method has the advantages of rapidness, simplicity, convenience, high sensitivity, specificity and the like. In addition, the detection kit or the technology is also suitable for detecting malignant melanoma, glioma, hepatocellular carcinoma and other tumors and blood samples, and molecular diagnosis of DNA (deoxyribonucleic acid) from thyroid tumors and fine needle puncture samples.
Description
Technical Field
The invention belongs to the technical field of TERT mutation detection, and particularly relates to a ddPCR detection method for telomerase promoter (TERTp) mutation (C228T or C250T) carried by tumor cells of Bladder Cancer (BC) and Renal Pelvis Cancer (RPC).
Background
The information in this background section is only for enhancement of understanding of the general background of the invention and is not necessarily to be construed as an admission or any form of suggestion that this information forms the prior art that is already known to a person of ordinary skill in the art.
Bladder Cancer (BC) is the most common malignant tumor of the urinary system in China and all over the world, and the annual incidence number reaches 350000. Patients require a lifelong follow-up visit due to the recurrence of BC. Cystoscope and urine cast-off cytology are conventional methods, but the former is high in cost and traumatic, causes pain to patients, and the latter is low in sensitivity and easy to miss diagnosis especially for patients with low-grade tumors. The company Israel nucleic has introduced the loader EpiCheck in recent yearsTMThe kit is used for urine DNA methylation detection (comprising 15 genes) of bladder cancer patients to monitor the postoperative condition of the patients, but the sensitivity, specificity and positive predictive value are respectively 68%, 88% and 45% ((the total amount is 15 genes))Eur Urol Oncol.2018; 1: 307-. In view of the above, there is a need to create a specific, accurate, sensitive and non-invasive detection method for diagnosing bladder cancer patients and monitoring disease recurrence.
Renal Pelvis Cancer (RPC), like BC, originates from urinary epithelial cells and belongs to transitional cell carcinoma, but the incidence is relatively low and accounts for 10% of all urinary tumors. However, the incidence of RPC has risen dramatically over the past 20 years, and most patients have seen it not early due to the lack of early symptoms and effective diagnostic tools. Therefore, it is necessary to find reliable RPC markers and to develop accurate and sensitive detection methods.
The activation of specific telomerase of malignant tumor has very important function in canceration and progression process. Telomerase reverse transcriptase (TERT) is the rate-limiting component of Telomerase, which is present in normal cells but is transcriptionally activated in cancer. TERT promoter mutations, i.e., C228T and C250T, have recently been found to occur in a number of malignancies, including BC and RPC. In addition, TERTp mutations are also frequently observed in thyroid cancer, hepatocellular carcinoma, malignant melanoma, malignant glioma, and the like. Since these TERTp mutations are not present in normal tissue cells, the detection of the C228T and C250T mutations is a reliable and specific BC, RPC marker. The present data show that up to 84% BC and 50% RPC patient tumor cells carry the C228T or C250T mutations (most of them are C228T mutations), and the inventors analyzed the patient urine-derived DNA using Sanger sequencing and detected the presence of mutations. It has also been recently reported that mutant TERTp can be detected in the patient's urine even 10 years prior to clinical diagnosis of bladder cancer. Therefore, the TERTp mutation can be used as a urine marker for BC and RPC patients and can also be used as a marker for early screening of tumors of healthy adults.
Disclosure of Invention
Based on the above-mentioned background art, the present invention aims to provide a non-invasive, sensitive and highly specific detection method, which improves the convenience of diagnosing bladder cancer and monitoring the progress of the disease. Based on the technical purpose, the invention provides an optimized ddPCR detection method, urine is used as a detection sample to detect TERTp mutation, and the lower detection limit can reach 0.05%.
According to the technical effects, the invention provides the following technical scheme:
in a first aspect of the present invention, a primer combination is provided, wherein the sequence of the primer combination is as follows:
forward:5’-CCTGCCCCTTCACCTTCCAG-3’(SEQ ID NO.1);
reverse:5'-AGCGCTGCCTGAAACTCG-3’(SEQ ID NO.2)。
in a second aspect of the invention, the primer combination of the first aspect is used for detecting TERT mutant products.
The invention designs a group of primers aiming at the mutation of two sites of C228T and C250T, and can identify the mutation condition of the two sites simultaneously. Based on the properties of the primer, the skilled person can apply the primer to a common PCR amplification method to identify two mutation sites so as to perform nucleic acid amplification operation.
The third aspect of the invention provides a TERT mutation detection kit, which comprises a primer, a probe and an amplification reagent;
the primer sequences are as follows:
forward:5’-CCTGCCCCTTCACCTTCCAG-3’(SEQ ID NO.1);
reverse:5'-AGCGCTGCCTGAAACTCG-3’(SEQ ID NO.2);
the probe sequence is as follows:
TERT mutant probe sequence: 5 '-/56-FAM/CCC + C + T + T + CCGG/3IABk FQ/-3' (SEQ ID NO. 3);
TERT wild-type probe sequence: 5 '-/5 HEX/CCCC + C + T + CCGG/3 IABKFQ/-3' (SEQ ID NO. 4).
In order to realize the non-invasive detection of tumor patients and the pathological samples obtained in a non-invasive manner such as urine and the like as detection objects, the invention essentially needs to provide a detection method with high sensitivity and accuracy. ddPCR is an absolute quantitative detection method, and can detect the change and difference of trace genes. In order to further improve the sensitivity of the detection method, the invention provides a primer and probe combination for efficiently recognizing the mutation site, and also improves the reaction system of ddPCR to further improve the amplification efficiency.
Based on the high sensitivity and accuracy of the detection kit, the noninvasive sampling detection of the patient can be realized by applying the kit to bladder cancer and renal pelvis cancer, and the pain of the patient is reduced. Similarly, the method can be applied to the detection of other TERT mutation related tumors, and can also obviously improve the detection accuracy.
In a fourth aspect of the present invention, there is provided a method for noninvasive diagnosis of bladder cancer or renal pelvis cancer, the method comprising detecting a pathological sample of an individual to be diagnosed using the primer combination of the first aspect or the kit of the third aspect.
In a fifth aspect of the present invention, there is provided a method for determining the prognosis of bladder cancer or renal pelvis cancer, the method comprising detecting a pathological sample of an individual to be diagnosed using the primer combination of the first aspect or the kit of the third aspect.
The beneficial effects of one or more technical schemes are as follows:
the invention provides a method for detecting two mutation sites of C228T and C250T in a TERT promoter based on ddPCR (polymerase chain reaction). the detection condition provided by the invention has good detection sensitivity which is obviously lower than the threshold (1%) of urine detection for bladder cancer patients in the existing report.
In addition, the invention also optimizes a ddPCR reaction system and provides a reagent system suitable for ddPCR.
Drawings
The accompanying drawings, which are incorporated in and constitute a part of this specification, are included to provide a further understanding of the invention, and are incorporated in and constitute a part of this specification, illustrate exemplary embodiments of the invention and together with the description serve to explain the invention and not to limit the invention.
FIG. 1 is a schematic flow chart of a diagnostic or prognostic procedure for bladder cancer or renal pelvis using TERT mutation detection;
FIG. 2 is a graph showing the results of the accuracy of ddPCR detection of TERT promoter mutation in example 1;
wherein, FIG. 2(A) is a graph showing the relationship between the amount of mutant-mutant TERT promoter detected by PCR of C228T;
FIG. 2(B) is a graph showing the relationship between the amount of mutant number-mutant TERT promoter detected by PCR in C250T.
FIG. 3 is a graph showing the results of the consistency of the mutation detection of the tumor tissue and urine mutant TERT promoter in the bladder cancer patients in example 1;
wherein in FIG. 3A both the tumor and urine are negative in the patient; in FIG. 3B, both the patient's tumor and urine are positive for C228T; (C) both the patient tumor and urine were positive for C250T.
Detailed Description
It is to be understood that the following detailed description is exemplary and is intended to provide further explanation of the invention as claimed. Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs.
It is noted that the terminology used herein is for the purpose of describing particular embodiments only and is not intended to be limiting of exemplary embodiments according to the invention. As used herein, the singular forms "a", "an" and "the" are intended to include the plural forms as well, and it should be understood that when the terms "comprises" and/or "comprising" are used in this specification, they specify the presence of stated features, steps, operations, devices, components, and/or combinations thereof, unless the context clearly indicates otherwise.
As described in the background art, in order to provide a non-invasive, sensitive and high-specificity TERT mutation detection method, the invention provides a detection method based on ddPCR detection, provides primer sequences capable of simultaneously detecting two mutation sites of C288T and C250T, and optimizes a reaction system.
In a first aspect of the present invention, a primer combination is provided, wherein the sequence of the primer combination is as follows:
forward:5’-CCTGCCCCTTCACCTTCCAG-3’(SEQ ID NO.1);
reverse:5'-AGCGCTGCCTGAAACTCG-3’(SEQ ID NO.2)。
in a second aspect of the invention, the primer combination of the first aspect is used for detecting TERT mutant products.
Preferably, the TERT mutation detection product is a detection product which is subjected to nucleic acid amplification after being specifically combined by the primer combination, and further is a product which is subjected to detection based on PCR;
in some embodiments, the PCR-based detection product comprises fluorescent quantitative PCR, reverse transcription PCR, RACE-PCR, nested PCR, inverse PCR, touchdown PCR, or digital PCR.
In a more specific embodiment, the Digital PCR is ddPCR (Droplet Digital PCR).
Preferably, the product for detecting TERT mutation is a detection kit.
Preferably, the TERT mutation is two mutation sites of C228T and C250T.
Preferably, the product for detecting TERT mutation is used for detecting bladder cancer, renal pelvis cancer, malignant melanoma, brain glioma and liver tumor samples.
Preferably, the product for detecting TERT mutation detects tumor tissue, urine or blood as sample.
The third aspect of the invention provides a TERT mutation detection kit, which comprises a primer, a probe and an amplification reagent;
the primer sequences are as follows:
forward:5’-CCTGCCCCTTCACCTTCCAG-3’(SEQ ID NO.1);
reverse:5'-AGCGCTGCCTGAAACTCG-3’(SEQ ID NO.2);
the probe sequence is as follows:
TERT mutant probe sequence: 5 '-/56-FAM/CCC + C + T + T + CCGG/3IABk FQ/-3' (SEQ ID NO. 3);
TERT wild-type probe sequence: 5 '-/5 HEX/CCCC + C + T + CCGG/3 IABKFQ/-3' (SEQ ID NO. 4).
Preferably, the amplification reagents include, but are not limited to, restriction enzymes, buffer salts, ddPCR premix, and the balance is supplemented by water.
Because ddPCR is a water-in-oil reaction system, amplification reagents are formed into a water-in-oil reaction system through a droplet reactor, and then the number of droplets for positive reaction is counted based on the flow cytometry detection principle. Therefore, the selection of the amplification reagents is of great significance for the formation of water-in-oil reaction systems.
In order to adapt to urine extracted DNA as a detection sample, the invention is improved to a certain extent aiming at a commercially available ddPCR premixing system, and the detection sensitivity is obviously improved by adding components such as buffer salt, alkaloid and the like.
Further preferably, the amplification reagent further comprises an alkaloid. Related research shows that part of alkaloid has the effect of promoting gene amplification. According to the invention, through screening, Betaine (Betaine) and buffer salt applied to the reaction system have a good nucleic acid coating effect, and the amplification efficiency can be obviously improved.
In a specific embodiment, the restriction enzyme is CviQI enzyme and the buffer salt is Na2EDTA。
In a fourth aspect of the present invention, there is provided a method for noninvasive diagnosis of bladder cancer or renal pelvis cancer, the method comprising detecting a pathological sample of an individual to be diagnosed using the primer combination of the first aspect or the kit of the third aspect.
Preferably, the diagnosis method comprises the steps of detecting the type and content of TERT mutation in a pathological sample of a subject to be diagnosed, and judging the diseased condition of the subject to be diagnosed according to the analysis of a statistical analysis result.
In a fifth aspect of the present invention, there is provided a method for determining the prognosis of bladder cancer or renal pelvis cancer, the method comprising detecting a pathological sample of an individual to be diagnosed using the primer combination of the first aspect or the kit of the third aspect.
Preferably, the prognosis method is as follows: and detecting the type and content of TERT mutation in the pathological sample of the patient, and judging the prognosis condition of the patient according to the analysis of the statistical analysis result.
In order to make the technical scheme of the present invention more clearly understood by those skilled in the art, the technical scheme of the present invention will be described in detail below by combining specific examples and comparative examples, wherein the reagents mentioned in the following examples are all commercially available products.
Example 1
In this example, a ddPCR-based method for detecting TERT promoter in urine samples of tumors from bladder cancer patients is provided.
One, ddPCR reaction conditions
I. Equipment: QX200 ddPCR system (Bio-Rad Laboratories).
ddPCR detection reagent
(a) Primers and probes
Primer:
5’-CCTGCCCCTTCACCTTCCAG-3’(forward)
5'-AGCGCTGCCTGAAACTCG-3’(reverse)
(the PCR product was 160bp in length).
And (3) probe:
TERT mutation probe sequence: 5 '-/56-FAM/CCC + C + T + T + CCGG/3IABk FQ/-3'
TERT wild-type probe sequence: 5 '-/5 HEX/CCCC + C + T + CCGG/3 IABKFQ/-3'
(+: locked nucleic acid modification).
(b) Reaction system
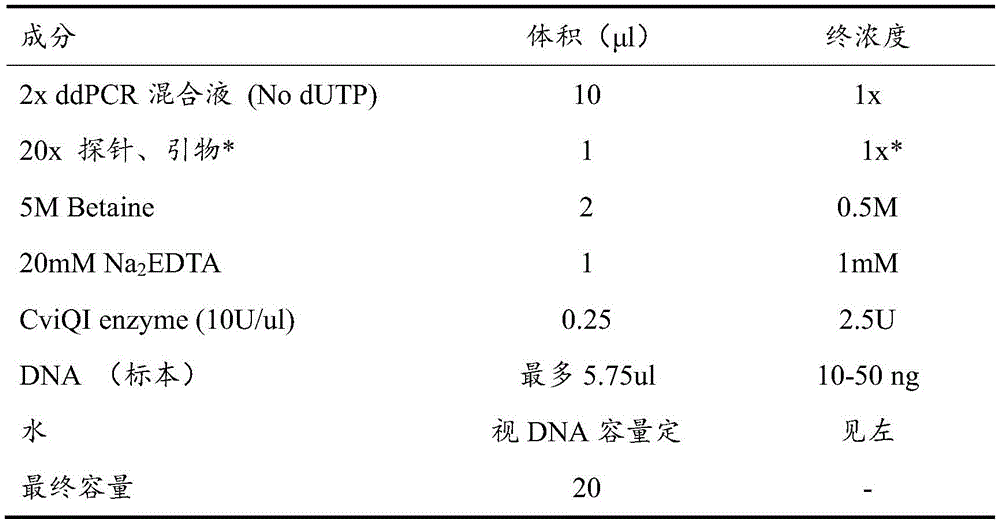
The reaction system used in this example is shown in Table 1.
TABLE 1 ddPCR reaction System
The final concentration of primers was 900nmol/L and the final concentration of probes was 250 nmol/L.
(c) Reaction conditions
The C1000 touch Thermal Cycler with 96-deep well reaction type is recommended. (i) At 95 ℃ for 2 minutes (rate of temperature rise: 2 ℃ C./second). (ii) Immediately following 40PCR cycles: 94 ℃, 30 seconds, 57 ℃, 1 minute. (iii)98 deg.C (heating rate: 2 deg.C/sec) for 10 minutes.
(d) Analysis of results
After PCR, 96-well plates were placed in a QX200 Droplet Reader, QuantaSoft analysis software (Bio-Rad, PCR machine kit) for analysis of results.
Second, result analysis
The detection of tumor tissue and urine samples of patients by the ddPCR detection method is shown in FIG. 2 and FIG. 3, and the sensitivity and accuracy results are shown.
The sensitivity test was carried out by adding 0.01%, 0.5%, 1.0%, 5%, 50% of mutant DNA to DNA derived from non-mutant cells, respectively, and then performing ddPCR assay. The linear relationship between the PCR signal and the amount of the mutant DNA is shown in FIG. 2, and the lowest detection conditions of ddPCR provided in this example can detect 0.05% C228T and 0.1% C250T.
The accuracy detection operation mode is as follows: patients were randomly screened and tested for mutations at C228T and C250T in tumor tissue and urine samples, respectively, under the conditions described above, and the results are shown in FIG. 3. Both the C228T and C250T mutations in the patient tumor samples shown in fig. 3A were shown to be negative, as were the C228T and C250T mutations in the urine samples. The patient's tumor sample in FIG. 3B showed C228T positive (indicated by the arrow), and the urine sample test also showed C228T positive. The patient in fig. 3C showed a positive C250T in the tumor sample (indicated by the arrow), and the urine sample test also showed consistent results.
The accurate detection result shows that the detection results of the detection method applied to the tumor and the urine are consistent, and the detection method can detect 0.05 percent of C228T and 0.1 percent of C250T at least. Based on the detection condition, the urine of the patient can be used as a sample to carry out mutation condition detection, tumor tissues are obtained without an operation, and noninvasive detection is realized.
Example 2
In this embodiment, a method for detecting TERT promoter in tumor and urine samples of renal pelvis cancer patients based on ddPCR is provided.
One, ddPCR reaction conditions
III, equipment: QX200 ddPCR system (Bio-Rad Laboratories).
ddPCR detection reagent
(c) Primers and probes
Primer:
5’-CCTGCCCCTTCACCTTCCAG-3’(forward)
5'-AGCGCTGCCTGAAACTCG-3’(reverse)
(the PCR product was 160bp in length).
And (3) probe:
TERT mutation probe sequence: 5 '-/56-FAM/CCC + C + T + T + CCGG/3IABk FQ/-3'
TERT wild-type probe sequence: 5 '-/5 HEX/CCCC + C + T + CCGG/3 IABKFQ/-3'
(+: locked nucleic acid modification).
(d) Reaction system
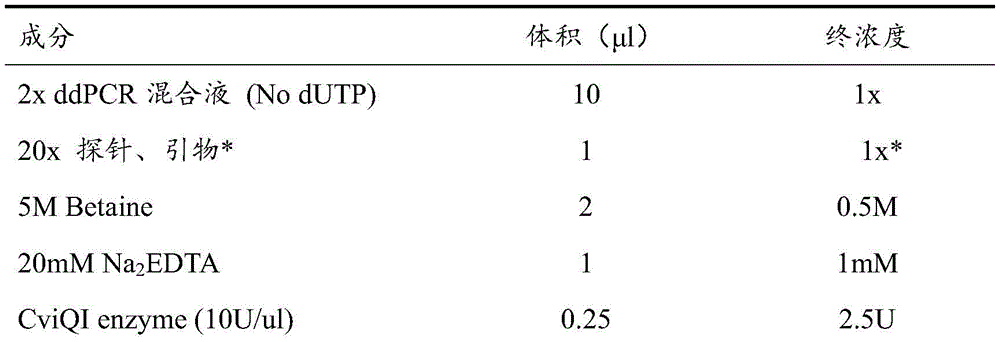
The reaction system used in this example is shown in Table 2.
TABLE 2 ddPCR reaction System
The final concentration of primers was 900nmol/L and the final concentration of probes was 250 nmol/L.
(c) Reaction conditions
The C1000 touch Thermal Cycler with 96-deep well reaction type is recommended. (i) At 95 ℃ for 2 minutes (rate of temperature rise: 2 ℃ C./second). (ii) Immediately following 40PCR cycles: 94 ℃, 30 seconds, 57 ℃, 1 minute. (iii)98 deg.C (heating rate: 2 deg.C/sec) for 10 minutes.
(d) Analysis of results
After PCR, 96-well plates were placed in a QX200 Droplet Reader, QuantaSoft analysis software (Bio-Rad, PCR machine kit) for analysis of results.
Second, result analysis
When a sample of a patient with renal pelvis cancer is evaluated by the same method as in example 1, the sensitivity detection results can reach 0.05% C228T and 0.1% C250T, and the accuracy is good.
Example 3
In this example, a method for detecting TERT promoter in tumor and urine samples of bladder cancer patients based on ddPCR is provided.
One, ddPCR reaction conditions
V. equipment: QX200 ddPCR system (Bio-Rad Laboratories).
ddPCR detection reagent
(e) Primers and probes
Primer:
5’-CCTGCCCCTTCACCTTCCAG-3’(forward)
5'-AGCGCTGCCTGAAACTCG-3’(reverse)
(the PCR product was 160bp in length).
And (3) probe:
TERT mutation probe sequence: 5 '-/56-FAM/CCC + C + T + T + CCGG/3IABk FQ/-3'
TERT wild-type probe sequence: 5 '-/5 HEX/CCCC + C + T + CCGG/3 IABKFQ/-3'
(+: locked nucleic acid modification).
(f) Reaction system
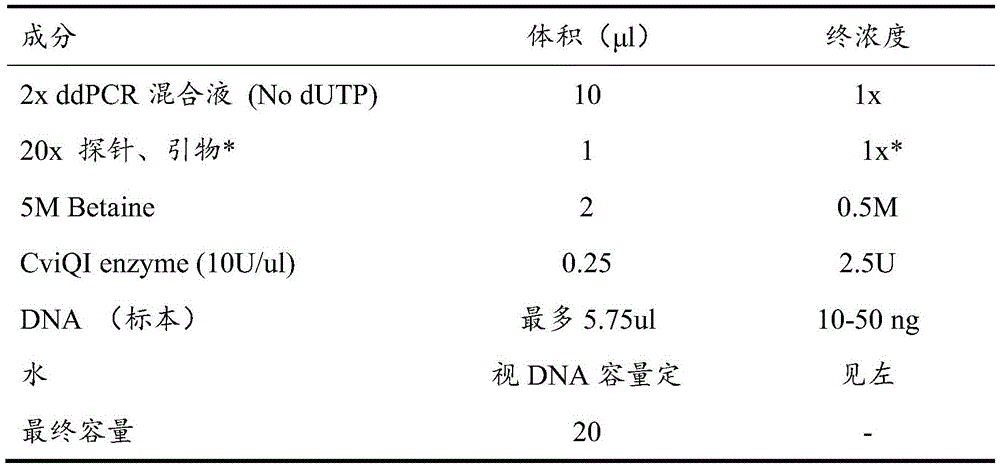
The reaction system used in this example is shown in Table 3.
TABLE 3 ddPCR reaction System
The final concentration of primers was 900nmol/L and the final concentration of probes was 250 nmol/L.
(c) Reaction conditions
The C1000 touch Thermal Cycler with 96-deep well reaction type is recommended. (i) At 95 ℃ for 2 minutes (rate of temperature rise: 2 ℃ C./second). (ii) Immediately following 40PCR cycles: 94 ℃, 30 seconds, 57 ℃, 1 minute. (iii)98 deg.C (heating rate: 2 deg.C/sec) for 10 minutes.
(d) Analysis of results
After PCR, 96-well plates were placed in a QX200 Droplet Reader, QuantaSoft analysis software (Bio-Rad, PCR machine kit) for analysis of results.
Second, result analysis
When a sample of a patient with renal pelvis cancer is evaluated by the same method as in example 1, the sensitivity detection results can reach 0.1% C228T and 0.1% C250T, and the accuracy is good.
Example 4
In this example, a method for detecting TERT promoter in tumor and urine samples of bladder cancer patients based on ddPCR is provided.
One, ddPCR reaction conditions
Vii, apparatus: QX200 ddPCR system (Bio-Rad Laboratories).
ddPCR detection reagent
(g) Primers and probes
Primer:
5’-CCTGCCCCTTCACCTTCCAG-3’(forward)
5'-AGCGCTGCCTGAAACTCG-3’(reverse)
(the PCR product was 160bp in length).
And (3) probe:
TERT mutation probe sequence: 5 '-/56-FAM/CCC + C + T + T + CCGG/3IABk FQ/-3'
TERT wild-type probe sequence: 5 '-/5 HEX/CCCC + C + T + CCGG/3 IABKFQ/-3'
(+: locked nucleic acid modification).
(h) Reaction system
The reaction system used in this example is shown in Table 4.
TABLE 4 ddPCR reaction System
The final concentration of primers was 900nmol/L and the final concentration of probes was 250 nmol/L.
(c) Reaction conditions
The C1000 touch Thermal Cycler with 96-deep well reaction type is recommended. (i) At 95 ℃ for 2 minutes (rate of temperature rise: 2 ℃ C./second). (ii) Immediately following 40PCR cycles: 94 ℃, 30 seconds, 57 ℃, 1 minute. (iii)98 deg.C (heating rate: 2 deg.C/sec) for 10 minutes.
(d) Analysis of results
After PCR, 96-well plates were placed in a QX200 Droplet Reader, QuantaSoft analysis software (Bio-Rad, PCR machine kit) for analysis of results.
Second, result analysis
When a sample of a patient with renal pelvis cancer is evaluated by the same method as in example 1, the sensitivity detection results can reach 1% C228T and 1% C250T, and the accuracy is good.
Example 5
In this example, a ddPCR-based method for detecting TERT promoter in urine samples of tumors from bladder cancer patients is provided.
One, ddPCR reaction conditions
IX. Equipment: QX200 ddPCR system (Bio-Rad Laboratories).
ddPCR detection reagent
(i) Primers and probes
Primer:
5’-CCTGCCCCTTCACCTTCCAG-3’(forward)
5'-AGCGCTGCCTGAAACTCG-3’(reverse)
(the PCR product was 160bp in length).
And (3) probe:
TERT mutation probe sequence: 5 '-/56-FAM/CCC + C + T + T + CCGG/3IABk FQ/-3'
TERT wild-type probe sequence: 5 '-/5 HEX/CCCC + C + T + CCGG/3 IABKFQ/-3'
(+: locked nucleic acid modification).
(j) Reaction system
The reaction system used in this example is shown in Table 5.
TABLE 5 ddPCR reaction System
The final concentration of primers was 900nmol/L and the final concentration of probes was 250 nmol/L.
(c) Reaction conditions
The C1000 touch Thermal Cycler with 96-deep well reaction type is recommended. (i) At 95 ℃ for 2 minutes (rate of temperature rise: 2 ℃ C./second). (ii) Immediately following 40PCR cycles: 94 ℃, 30 seconds, 57 ℃, 1 minute. (iii)98 deg.C (heating rate: 2 deg.C/sec) for 10 minutes.
(d) Analysis of results
After PCR, 96-well plates were placed in a QX200 Droplet Reader, QuantaSoft analysis software (Bio-Rad, PCR machine kit) for analysis of results.
Second, result analysis
When a sample of a patient with renal pelvis cancer is evaluated by the same method as in example 1, the sensitivity detection results can reach 0.8% C228T and 1% C250T, and the accuracy is good.
The above description is only a preferred embodiment of the present invention and is not intended to limit the present invention, and various modifications and changes may be made by those skilled in the art. Any modification, equivalent replacement, or improvement made within the spirit and principle of the present invention should be included in the protection scope of the present invention.
SEQUENCE LISTING
<110> Zheng Chengyun
<120> TERT mutation detection kit and application thereof in tumor noninvasive diagnosis
<130>2010
<160>4
<170>PatentIn version 3.3
<210>1
<211>20
<212>DNA
<213> Artificial sequence
<400>1
cctgcccctt caccttccag 20
<210>2
<211>18
<212>DNA
<213> Artificial sequence
<400>2
agcgctgcct gaaactcg 18
<210>3
<211>10
<212>DNA
<213> Artificial sequence
<400>3
<210>4
<211>10
<212>DNA
<213> Artificial sequence
<400>4
Claims (10)
1. A primer combination, wherein the sequence of the primer combination is as follows:
forward:5’-CCTGCCCCTTCACCTTCCAG-3’;
reverse:5'-AGCGCTGCCTGAAACTCG-3’。
2. use of the primer combination of claim 1 for detecting TERT mutant products.
3. Use of the primer combination according to claim 2 for detecting TERT mutant products, wherein the detection of TERT mutant products is a detection product of nucleic acid amplification after specific binding by the primer combination, preferably a PCR-based detection product.
4. The use of the primer combination of claim 3 for detecting TERT mutant products, wherein the products detected by PCR comprise quantitative fluorescent PCR, reverse transcription PCR, RACE-PCR, nested PCR, reverse PCR, touchdown PCR or digital PCR; preferably, the digital PCR is ddPCR.
5. The use of the primer combination of claim 2 for detecting TERT mutation products, wherein the product for detecting TERT mutation is a detection kit;
or the TERT mutation is two mutation sites of C228T and C250T;
or the product for detecting TERT mutation is used for detecting bladder, renal pelvis cancer, malignant melanoma, brain glioma and liver tumor samples;
or the product for detecting TERT mutation, wherein the detection sample is tumor tissue, urine or blood.
6. A TERT mutation detection kit is characterized in that the detection kit comprises a primer, a probe and an amplification reagent;
the primer sequences are as follows:
forward:5’-CCTGCCCCTTCACCTTCCAG-3’;
reverse:5'-AGCGCTGCCTGAAACTCG-3’;
the probe sequence is as follows:
TERT mutant probe sequence: 5 '-/56-FAM/CCC + C + T + T + CCGG/3IABk FQ/-3';
TERT wild-type probe sequence: 5 '-/5 HEX/CCCC + C + T + CCGG/3 IABKFQ/-3'.
7. The TERT mutation detection kit of claim 6, wherein said amplification reagents include, but are not limited to, restriction enzymes, buffer salts, ddPCR premix, the balance being supplemented with water; preferably, the amplification reagent further comprises an alkaloid.
8. The TERT mutation detection kit of claim 7, wherein the restriction enzyme is CThe viQIenzyme and the buffer salt are Na2EDTA。
9. A method for non-invasive diagnosis of bladder cancer or renal pelvis cancer, which comprises detecting a pathological sample of an individual to be diagnosed using the primer combination of claim 1 or the kit of any one of claims 6 to 8;
preferably, the diagnosis method comprises the steps of detecting the type and content of TERT mutation in a pathological sample of a subject to be diagnosed, and judging the diseased condition of the subject to be diagnosed according to the analysis of a statistical analysis result.
10. A method for determining the prognosis of bladder cancer or renal pelvis cancer, which comprises detecting a pathological sample of an individual to be diagnosed by using the primer combination of claim 1 or the kit of any one of claims 6 to 8;
preferably, the prognosis method is as follows: and detecting the type and content of TERT mutation in the pathological sample of the patient, and judging the prognosis condition of the patient according to the analysis of the statistical analysis result.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010467657.4A CN111560436A (en) | 2020-05-28 | 2020-05-28 | TERT mutation detection kit and application thereof in noninvasive tumor diagnosis |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010467657.4A CN111560436A (en) | 2020-05-28 | 2020-05-28 | TERT mutation detection kit and application thereof in noninvasive tumor diagnosis |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN111560436A true CN111560436A (en) | 2020-08-21 |
Family
ID=72075026
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010467657.4A Withdrawn CN111560436A (en) | 2020-05-28 | 2020-05-28 | TERT mutation detection kit and application thereof in noninvasive tumor diagnosis |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111560436A (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103571953A (en) * | 2013-10-28 | 2014-02-12 | 深圳市第二人民医院 | Detection method of single nucleotide polymorphism sequences of TERT (telomerase reverse transcriptase) promoters |
| CN106148516A (en) * | 2016-06-29 | 2016-11-23 | 苏州捷诺威生物科技有限公司 | A kind of test kit for detecting the sudden change of TERT gene promoter and detection method and application |
| CN108350504A (en) * | 2015-06-24 | 2018-07-31 | Ucl商业有限公司 | The diagnostic method of carcinoma of urinary bladder |
-
2020
- 2020-05-28 CN CN202010467657.4A patent/CN111560436A/en not_active Withdrawn
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103571953A (en) * | 2013-10-28 | 2014-02-12 | 深圳市第二人民医院 | Detection method of single nucleotide polymorphism sequences of TERT (telomerase reverse transcriptase) promoters |
| CN108350504A (en) * | 2015-06-24 | 2018-07-31 | Ucl商业有限公司 | The diagnostic method of carcinoma of urinary bladder |
| CN106148516A (en) * | 2016-06-29 | 2016-11-23 | 苏州捷诺威生物科技有限公司 | A kind of test kit for detecting the sudden change of TERT gene promoter and detection method and application |
Non-Patent Citations (1)
| Title |
|---|
| ASHLEIGH C MCEVOY等: "Sensitive droplet digital PCR method for detection of TERT promoter mutations in cell free DNA from patients with metastatic melanoma", 《ONCOTARGET》 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Horiike et al. | Detection of epidermal growth factor receptor mutation in transbronchial needle aspirates of non-small cell lung cancer | |
| KR101566368B1 (en) | Urine gene expression ratios for detection of cancer | |
| Feng et al. | Elevated serum-circulating RNA in patients with conventional renal cell cancer | |
| US20080206756A1 (en) | Biomarker panel for colorectal cancer | |
| CA3026809A1 (en) | Compositions and methods for diagnosing lung cancers using gene expression profiles | |
| CN111394457B (en) | Application of biomarker, reagent for detecting renal clear cell carcinoma and kit thereof | |
| CN102586401A (en) | Method and kit for detecting mutation of BRAF gene of human colorectal cancer | |
| EP1918710A1 (en) | Characterizing prostate cancer | |
| CN111518908B (en) | Urine prostate cancer marker combination and application thereof in preparation of accurate diagnostic reagent | |
| JP6145083B2 (en) | Compositions and methods for detecting mutations in JAK2 nucleic acids | |
| Wu et al. | Clinical significance of methylation of E-cadherin and p14ARF gene promoters in skin squamous cell carcinoma tissues | |
| CN112662773A (en) | TERT promoter methylation detection primer composition and application thereof | |
| CN107641649B (en) | Primer pair, kit and method for detecting microsatellite NR27 site stability | |
| WO2018211404A1 (en) | Composite epigenetic biomarkers for accurate screening, diagnosis and prognosis of colorectal cancer | |
| KR102036222B1 (en) | Markers for prognosing a patient with chronic kidney disease patients caused by diabetic nephropathy | |
| Hamada et al. | Diagnostic usefulness of PCR profiling of the differentially expressed marker genes in thyroid papillary carcinomas | |
| CN111560436A (en) | TERT mutation detection kit and application thereof in noninvasive tumor diagnosis | |
| US20210079482A1 (en) | Methylation-based biomarkers in breast cancer screening, diagnosis, or prognosis | |
| JP2006521110A (en) | Detection and monitoring of lung cancer | |
| CN114410769A (en) | A kind of SNP marker, kit and application related to hypercholesterolemia based on SOD3 gene | |
| CN115961048B (en) | A gene methylation detection primer combination, reagent and application thereof | |
| KR20210120737A (en) | The composition for detecting mutations of PIK3CA and the kit consisting of the compounds | |
| KR102036221B1 (en) | Development of epigenetic diagnostic kit for prognosing chronic kidney disease | |
| KR102036219B1 (en) | Composition for predicting exacerbation of chronic kidney disease using alteration of CpG methylation in promoter region of gene and uses thereof | |
| KR102036220B1 (en) | Composition for detecting chronic kidney disease specific epigenetic methylation markers and detecting method thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| WW01 | Invention patent application withdrawn after publication | ||
| WW01 | Invention patent application withdrawn after publication |
Application publication date: 20200821 |