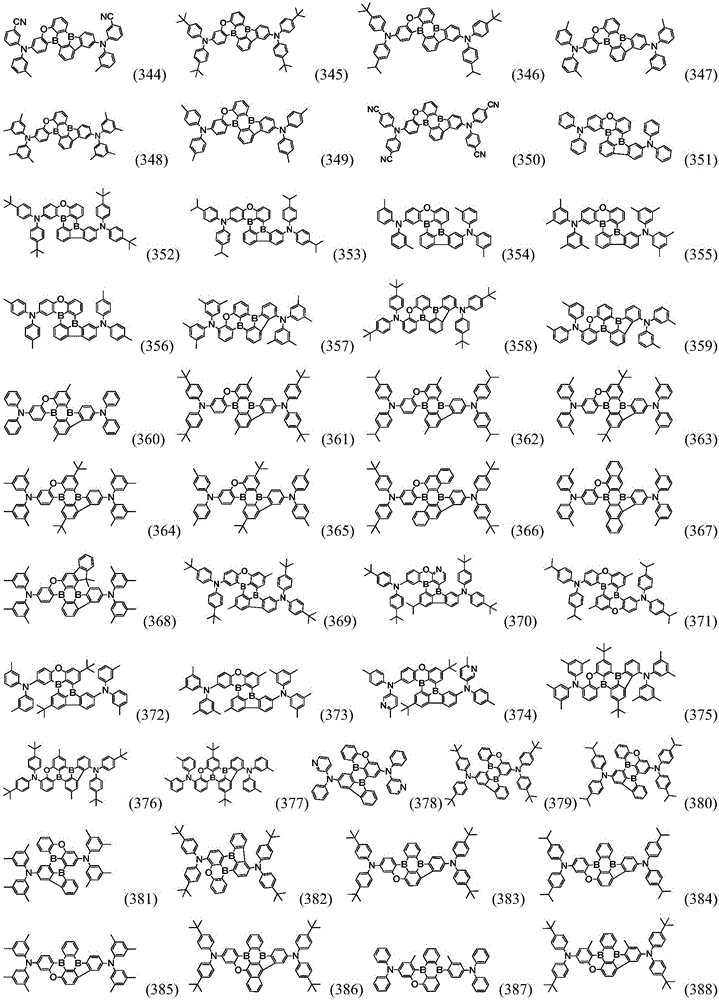CN111440203A - Organic compound with diboron as core and application thereof - Google Patents
Organic compound with diboron as core and application thereof Download PDFInfo
- Publication number
- CN111440203A CN111440203A CN201811635458.9A CN201811635458A CN111440203A CN 111440203 A CN111440203 A CN 111440203A CN 201811635458 A CN201811635458 A CN 201811635458A CN 111440203 A CN111440203 A CN 111440203A
- Authority
- CN
- China
- Prior art keywords
- substituted
- unsubstituted
- group
- atom
- raw material
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 150000002894 organic compounds Chemical class 0.000 title claims abstract description 24
- ZOCHARZZJNPSEU-UHFFFAOYSA-N diboron Chemical compound B#B ZOCHARZZJNPSEU-UHFFFAOYSA-N 0.000 title claims abstract description 8
- 150000001875 compounds Chemical class 0.000 claims abstract description 78
- 239000000463 material Substances 0.000 claims abstract description 71
- 125000001424 substituent group Chemical group 0.000 claims abstract description 7
- 239000002994 raw material Substances 0.000 claims description 94
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 claims description 66
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 claims description 58
- YTZKOQUCBOVLHL-UHFFFAOYSA-N tert-butylbenzene Chemical compound CC(C)(C)C1=CC=CC=C1 YTZKOQUCBOVLHL-UHFFFAOYSA-N 0.000 claims description 58
- 238000006243 chemical reaction Methods 0.000 claims description 40
- 238000003756 stirring Methods 0.000 claims description 30
- ILAHWRKJUDSMFH-UHFFFAOYSA-N boron tribromide Substances BrB(Br)Br ILAHWRKJUDSMFH-UHFFFAOYSA-N 0.000 claims description 26
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 25
- 229910001868 water Inorganic materials 0.000 claims description 25
- 229910015845 BBr3 Inorganic materials 0.000 claims description 21
- UBJFKNSINUCEAL-UHFFFAOYSA-N lithium;2-methylpropane Chemical compound [Li+].C[C-](C)C UBJFKNSINUCEAL-UHFFFAOYSA-N 0.000 claims description 21
- 238000002390 rotary evaporation Methods 0.000 claims description 19
- 125000000217 alkyl group Chemical group 0.000 claims description 18
- 125000003118 aryl group Chemical group 0.000 claims description 18
- 125000005842 heteroatom Chemical group 0.000 claims description 18
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 claims description 17
- -1 biphenylyl group Chemical group 0.000 claims description 17
- 238000001816 cooling Methods 0.000 claims description 17
- 239000000706 filtrate Substances 0.000 claims description 17
- 238000001914 filtration Methods 0.000 claims description 17
- 239000012074 organic phase Substances 0.000 claims description 17
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims description 16
- 125000001072 heteroaryl group Chemical group 0.000 claims description 16
- 239000011259 mixed solution Substances 0.000 claims description 16
- 239000000741 silica gel Substances 0.000 claims description 15
- 229910002027 silica gel Inorganic materials 0.000 claims description 15
- 229910052805 deuterium Inorganic materials 0.000 claims description 14
- 238000000605 extraction Methods 0.000 claims description 14
- 229910052757 nitrogen Inorganic materials 0.000 claims description 14
- 238000000926 separation method Methods 0.000 claims description 14
- 239000007788 liquid Substances 0.000 claims description 13
- 230000007935 neutral effect Effects 0.000 claims description 12
- YZCKVEUIGOORGS-IGMARMGPSA-N Protium Chemical group [1H] YZCKVEUIGOORGS-IGMARMGPSA-N 0.000 claims description 10
- YZCKVEUIGOORGS-NJFSPNSNSA-N Tritium Chemical group [3H] YZCKVEUIGOORGS-NJFSPNSNSA-N 0.000 claims description 10
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 10
- 150000001975 deuterium Chemical group 0.000 claims description 10
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 10
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 10
- 229910052722 tritium Inorganic materials 0.000 claims description 10
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 claims description 8
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 8
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 claims description 8
- 239000011261 inert gas Substances 0.000 claims description 8
- 125000001624 naphthyl group Chemical group 0.000 claims description 8
- 125000004430 oxygen atom Chemical group O* 0.000 claims description 8
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 8
- 125000004076 pyridyl group Chemical group 0.000 claims description 8
- 239000002904 solvent Substances 0.000 claims description 8
- 229910052717 sulfur Inorganic materials 0.000 claims description 8
- 125000004434 sulfur atom Chemical group 0.000 claims description 8
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical group [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 claims description 6
- 125000001931 aliphatic group Chemical group 0.000 claims description 6
- 230000015572 biosynthetic process Effects 0.000 claims description 6
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 claims description 6
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 6
- 229910052731 fluorine Inorganic materials 0.000 claims description 6
- 125000001153 fluoro group Chemical group F* 0.000 claims description 6
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 6
- 238000004519 manufacturing process Methods 0.000 claims description 6
- 125000004593 naphthyridinyl group Chemical group N1=C(C=CC2=CC=CN=C12)* 0.000 claims description 6
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 claims description 6
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 6
- 229910000077 silane Inorganic materials 0.000 claims description 6
- 238000003786 synthesis reaction Methods 0.000 claims description 6
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 6
- YJTKZCDBKVTVBY-UHFFFAOYSA-N 1,3-Diphenylbenzene Chemical group C1=CC=CC=C1C1=CC=CC(C=2C=CC=CC=2)=C1 YJTKZCDBKVTVBY-UHFFFAOYSA-N 0.000 claims description 4
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 claims description 4
- 229910000147 aluminium phosphate Inorganic materials 0.000 claims description 4
- 125000002178 anthracenyl group Chemical group C1(=CC=CC2=CC3=CC=CC=C3C=C12)* 0.000 claims description 4
- 235000010290 biphenyl Nutrition 0.000 claims description 4
- 239000004305 biphenyl Substances 0.000 claims description 4
- 229910052736 halogen Inorganic materials 0.000 claims description 4
- 150000002367 halogens Chemical class 0.000 claims description 4
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 4
- 125000002098 pyridazinyl group Chemical group 0.000 claims description 4
- 125000000714 pyrimidinyl group Chemical group 0.000 claims description 4
- 150000003839 salts Chemical class 0.000 claims description 4
- 125000004306 triazinyl group Chemical group 0.000 claims description 4
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Divinylene sulfide Natural products C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 claims description 2
- 125000004653 anthracenylene group Chemical group 0.000 claims description 2
- 125000000732 arylene group Chemical group 0.000 claims description 2
- 125000006267 biphenyl group Chemical group 0.000 claims description 2
- 125000002529 biphenylenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C12)* 0.000 claims description 2
- 229910052799 carbon Inorganic materials 0.000 claims description 2
- 125000004432 carbon atom Chemical group C* 0.000 claims description 2
- CPPKAGUPTKIMNP-UHFFFAOYSA-N cyanogen fluoride Chemical group FC#N CPPKAGUPTKIMNP-UHFFFAOYSA-N 0.000 claims description 2
- 125000004988 dibenzothienyl group Chemical group C1(=CC=CC=2SC3=C(C21)C=CC=C3)* 0.000 claims description 2
- 125000005509 dibenzothiophenyl group Chemical group 0.000 claims description 2
- 150000002240 furans Chemical class 0.000 claims description 2
- 125000002541 furyl group Chemical group 0.000 claims description 2
- 125000005549 heteroarylene group Chemical group 0.000 claims description 2
- 125000004957 naphthylene group Chemical group 0.000 claims description 2
- 125000001792 phenanthrenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C=CC12)* 0.000 claims description 2
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 claims description 2
- 125000001725 pyrenyl group Chemical group 0.000 claims description 2
- 125000005551 pyridylene group Chemical group 0.000 claims description 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims description 2
- 125000001544 thienyl group Chemical group 0.000 claims description 2
- 229930192474 thiophene Natural products 0.000 claims description 2
- 150000003577 thiophenes Chemical class 0.000 claims description 2
- 125000005730 thiophenylene group Chemical group 0.000 claims description 2
- 238000004770 highest occupied molecular orbital Methods 0.000 abstract description 8
- 230000009477 glass transition Effects 0.000 abstract description 3
- 238000009826 distribution Methods 0.000 abstract description 2
- 239000010410 layer Substances 0.000 description 57
- 238000012360 testing method Methods 0.000 description 40
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 29
- 239000007858 starting material Substances 0.000 description 29
- 238000000921 elemental analysis Methods 0.000 description 28
- 238000002360 preparation method Methods 0.000 description 23
- 238000000034 method Methods 0.000 description 19
- 239000000203 mixture Substances 0.000 description 17
- 238000005859 coupling reaction Methods 0.000 description 16
- 238000002347 injection Methods 0.000 description 16
- 239000007924 injection Substances 0.000 description 16
- 230000031709 bromination Effects 0.000 description 15
- 238000005893 bromination reaction Methods 0.000 description 15
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 13
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 12
- 230000000052 comparative effect Effects 0.000 description 11
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 9
- 230000000903 blocking effect Effects 0.000 description 9
- 238000001704 evaporation Methods 0.000 description 8
- 238000010438 heat treatment Methods 0.000 description 8
- 239000011368 organic material Substances 0.000 description 8
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical group [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 6
- 230000005525 hole transport Effects 0.000 description 6
- MFRIHAYPQRLWNB-UHFFFAOYSA-N sodium tert-butoxide Chemical compound [Na+].CC(C)(C)[O-] MFRIHAYPQRLWNB-UHFFFAOYSA-N 0.000 description 6
- BWHDROKFUHTORW-UHFFFAOYSA-N tritert-butylphosphane Chemical compound CC(C)(C)P(C(C)(C)C)C(C)(C)C BWHDROKFUHTORW-UHFFFAOYSA-N 0.000 description 6
- 229910052796 boron Inorganic materials 0.000 description 5
- 229910001873 dinitrogen Inorganic materials 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 230000005284 excitation Effects 0.000 description 5
- 238000002156 mixing Methods 0.000 description 5
- 239000000758 substrate Substances 0.000 description 5
- QFLWZFQWSBQYPS-AWRAUJHKSA-N (3S)-3-[[(2S)-2-[[(2S)-2-[5-[(3aS,6aR)-2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]pentanoylamino]-3-methylbutanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-4-[1-bis(4-chlorophenoxy)phosphorylbutylamino]-4-oxobutanoic acid Chemical compound CCCC(NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@@H](NC(=O)CCCCC1SC[C@@H]2NC(=O)N[C@H]12)C(C)C)P(=O)(Oc1ccc(Cl)cc1)Oc1ccc(Cl)cc1 QFLWZFQWSBQYPS-AWRAUJHKSA-N 0.000 description 4
- 238000002484 cyclic voltammetry Methods 0.000 description 4
- 230000003111 delayed effect Effects 0.000 description 4
- 239000007772 electrode material Substances 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- 238000007738 vacuum evaporation Methods 0.000 description 4
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- DKHNGUNXLDCATP-UHFFFAOYSA-N dipyrazino[2,3-f:2',3'-h]quinoxaline-2,3,6,7,10,11-hexacarbonitrile Chemical compound C12=NC(C#N)=C(C#N)N=C2C2=NC(C#N)=C(C#N)N=C2C2=C1N=C(C#N)C(C#N)=N2 DKHNGUNXLDCATP-UHFFFAOYSA-N 0.000 description 3
- 230000008020 evaporation Effects 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 230000005855 radiation Effects 0.000 description 3
- 239000011541 reaction mixture Substances 0.000 description 3
- 238000010992 reflux Methods 0.000 description 3
- 238000004809 thin layer chromatography Methods 0.000 description 3
- GLGNXYJARSMNGJ-VKTIVEEGSA-N (1s,2s,3r,4r)-3-[[5-chloro-2-[(1-ethyl-6-methoxy-2-oxo-4,5-dihydro-3h-1-benzazepin-7-yl)amino]pyrimidin-4-yl]amino]bicyclo[2.2.1]hept-5-ene-2-carboxamide Chemical compound CCN1C(=O)CCCC2=C(OC)C(NC=3N=C(C(=CN=3)Cl)N[C@H]3[C@H]([C@@]4([H])C[C@@]3(C=C4)[H])C(N)=O)=CC=C21 GLGNXYJARSMNGJ-VKTIVEEGSA-N 0.000 description 2
- IUSARDYWEPUTPN-OZBXUNDUSA-N (2r)-n-[(2s,3r)-4-[[(4s)-6-(2,2-dimethylpropyl)spiro[3,4-dihydropyrano[2,3-b]pyridine-2,1'-cyclobutane]-4-yl]amino]-3-hydroxy-1-[3-(1,3-thiazol-2-yl)phenyl]butan-2-yl]-2-methoxypropanamide Chemical compound C([C@H](NC(=O)[C@@H](C)OC)[C@H](O)CN[C@@H]1C2=CC(CC(C)(C)C)=CN=C2OC2(CCC2)C1)C(C=1)=CC=CC=1C1=NC=CS1 IUSARDYWEPUTPN-OZBXUNDUSA-N 0.000 description 2
- XFJBGINZIMNZBW-CRAIPNDOSA-N 5-chloro-2-[4-[(1r,2s)-2-[2-(5-methylsulfonylpyridin-2-yl)oxyethyl]cyclopropyl]piperidin-1-yl]pyrimidine Chemical compound N1=CC(S(=O)(=O)C)=CC=C1OCC[C@H]1[C@@H](C2CCN(CC2)C=2N=CC(Cl)=CN=2)C1 XFJBGINZIMNZBW-CRAIPNDOSA-N 0.000 description 2
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 230000005540 biological transmission Effects 0.000 description 2
- 238000004140 cleaning Methods 0.000 description 2
- 238000010549 co-Evaporation Methods 0.000 description 2
- 229940125758 compound 15 Drugs 0.000 description 2
- 229940125807 compound 37 Drugs 0.000 description 2
- 229940125898 compound 5 Drugs 0.000 description 2
- 125000005266 diarylamine group Chemical group 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 230000002035 prolonged effect Effects 0.000 description 2
- 230000000171 quenching effect Effects 0.000 description 2
- 238000001228 spectrum Methods 0.000 description 2
- MPDDTAJMJCESGV-CTUHWIOQSA-M (3r,5r)-7-[2-(4-fluorophenyl)-5-[methyl-[(1r)-1-phenylethyl]carbamoyl]-4-propan-2-ylpyrazol-3-yl]-3,5-dihydroxyheptanoate Chemical compound C1([C@@H](C)N(C)C(=O)C2=NN(C(CC[C@@H](O)C[C@@H](O)CC([O-])=O)=C2C(C)C)C=2C=CC(F)=CC=2)=CC=CC=C1 MPDDTAJMJCESGV-CTUHWIOQSA-M 0.000 description 1
- 101100072645 Arabidopsis thaliana IPS3 gene Proteins 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- VSCWAEJMTAWNJL-UHFFFAOYSA-K aluminium chloride Substances Cl[Al](Cl)Cl VSCWAEJMTAWNJL-UHFFFAOYSA-K 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 239000008367 deionised water Substances 0.000 description 1
- 229910021641 deionized water Inorganic materials 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 238000011982 device technology Methods 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 125000004987 dibenzofuryl group Chemical group C1(=CC=CC=2OC3=C(C21)C=CC=C3)* 0.000 description 1
- 238000000113 differential scanning calorimetry Methods 0.000 description 1
- 239000003792 electrolyte Substances 0.000 description 1
- 238000000295 emission spectrum Methods 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 238000002189 fluorescence spectrum Methods 0.000 description 1
- 230000005283 ground state Effects 0.000 description 1
- 230000003301 hydrolyzing effect Effects 0.000 description 1
- UEXQBEVWFZKHNB-UHFFFAOYSA-N intermediate 29 Natural products C1=CC(N)=CC=C1NC1=NC=CC=N1 UEXQBEVWFZKHNB-UHFFFAOYSA-N 0.000 description 1
- 239000004973 liquid crystal related substance Substances 0.000 description 1
- 238000004020 luminiscence type Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 238000004776 molecular orbital Methods 0.000 description 1
- 239000012299 nitrogen atmosphere Substances 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- 239000012044 organic layer Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 239000010970 precious metal Substances 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 125000005576 pyrimidinylene group Chemical group 0.000 description 1
- 238000010791 quenching Methods 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 239000004065 semiconductor Substances 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 239000011877 solvent mixture Substances 0.000 description 1
- 238000010025 steaming Methods 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 238000002411 thermogravimetry Methods 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000001052 transient effect Effects 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F5/00—Compounds containing elements of Groups 3 or 13 of the Periodic Table
- C07F5/02—Boron compounds
- C07F5/027—Organoboranes and organoborohydrides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0803—Compounds with Si-C or Si-Si linkages
- C07F7/081—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te
- C07F7/0812—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te comprising a heterocyclic ring
- C07F7/0816—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te comprising a heterocyclic ring said ring comprising Si as a ring atom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
- H10K50/12—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers comprising dopants
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1007—Non-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1088—Heterocyclic compounds characterised by ligands containing oxygen as the only heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1096—Heterocyclic compounds characterised by ligands containing other heteroatoms
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
The invention relates to an organic compound taking diboron as a core and application thereof to an O L ED device, wherein the structure of the compound is shown as a general formula (1), the whole molecule of the organic compound taking diboron as the core is a larger rigid structure, the introduction of a substituent group reduces the planarity of the material, so that the steric hindrance of the material is increased, the material is not easy to rotate, and the three-dimensional structure is more stable, so that the compound has higher glass transition temperature and molecular thermal stability, in addition, the HOMO and L UMO distribution positions of the compound are mutually separated, so that the compound has proper HOMO and L UMO energy levels, and therefore, after the compound is applied to an O L ED device, the luminous efficiency and the service life of the device can be effectively improved.
Description
Technical Field
The invention relates to the technical field of semiconductor materials, in particular to an organic compound containing two boron atoms in a structure and application thereof in an organic electroluminescent device.
Background
The Organic electroluminescent (O L ED: Organic L light Emission Diodes) device technology can be used for manufacturing novel display products and novel lighting products, is expected to replace the existing liquid crystal display and fluorescent lamp lighting, and has wide application prospect.
The organic light emitting phenomenon is an example of conversion of current into visible light by internal processing of specific organic molecules. The organic light emission phenomenon is based on the following mechanism: when the organic material layer is interposed between the anode and the cathode, if a voltage is applied between the two electrodes, electrons and holes are injected from the cathode and the anode into the organic material layer; the electrons and holes injected into the organic material layer are recombined to form excitons (exiton), which drop to the ground state to emit light. An organic light emitting device based on the above mechanism generally includes a cathode, an anode, and one or more organic material layers interposed therebetween.
The material of the organic material layer in the organic electroluminescent device may be classified into a hole injection material, a hole transport material, a light emitting material, an electron transport material, or an electron injection material according to its use. In this regard, an organic material having a p-type property, which is easily oxidized and electrochemically stable when it is oxidized, is mainly used as a hole injection material or a hole transport material. Meanwhile, an organic material having an n-type property, which is easily reduced and electrochemically stable when reduced, is mainly used as an electron injection material or an electron transport material. As the light emitting layer material, a material having both p-type and n-type properties, which is stable when it is oxidized and reduced, is preferable, and a material having a higher light emitting efficiency for converting excitons into light when the excitons are formed is also preferable.
However, the conventional organic fluorescent materials can emit light only by using 25% singlet excitons formed by electric excitation, the internal quantum efficiency of the device is low (up to 25%), the external quantum efficiency is generally lower than 5%, and there is a great difference from the efficiency of a phosphorescent device, although the intersystem crossing is enhanced by the strong spin-orbit coupling of heavy atom centers of the phosphorescent material, the singlet excitons and the triplet excitons formed by the electric excitation can be effectively used for emitting light, and the internal quantum efficiency of the device reaches 100%.
The materials generally have small singlet-triplet energy level difference (△ Est), triplet excitons can be converted into singlet excitons through intersystem crossing to emit light, the singlet excitons and the triplet excitons formed under electric excitation can be fully utilized, the internal quantum efficiency of the device can reach 100 percent, meanwhile, the materials have controllable structures, stable properties, low price and no need of precious metals, and have wide application prospects in the field of O L EDs.
Although TADF materials can theoretically achieve 100% exciton utilization, there are actually the following problems: (1) the T1 and S1 states of the designed molecule have strong CT characteristics, and a very small energy gap of S1-T1 state can realize high conversion rate of T1 → S1 state excitons through a TADF process, but simultaneously lead to low radiation transition rate of S1 state, so that the high exciton utilization rate and the high fluorescence radiation efficiency are difficult to realize at the same time; (2) even though doped devices have been employed to mitigate the T1 exciton concentration quenching effect, most TADF material devices suffer from severe efficiency roll-off at high current densities.
In addition, the material used in the organic electroluminescent device preferably also has excellent thermal stability, a suitable band gap (band gap), and a suitable Highest Occupied Molecular Orbital (HOMO) or lowest occupied molecular orbital (L UMO) level, as well as excellent chemical stability, charge mobility, and the like.
Therefore, there is a continuous need to develop new materials for organic electroluminescent devices.
Disclosure of Invention
The compound has high glass transition temperature and molecular thermal stability, has proper HOMO and L UMO energy levels, has a singlet state-triplet state energy level difference (△ Est), and can be used as a host material and a doping material of a light-emitting layer of an organic electroluminescent device, so that the light-emitting efficiency and the service life of the device are improved.
The technical scheme of the invention is as follows:
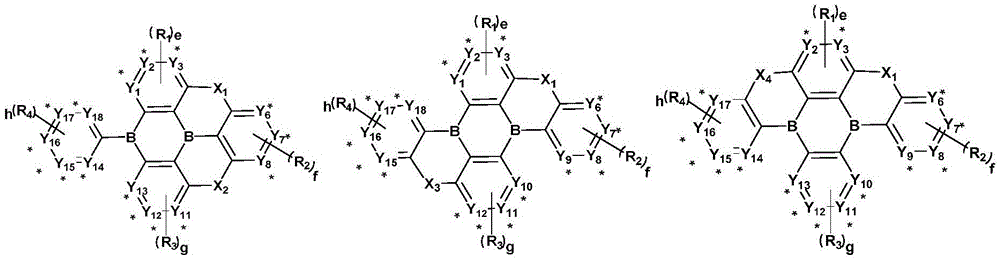
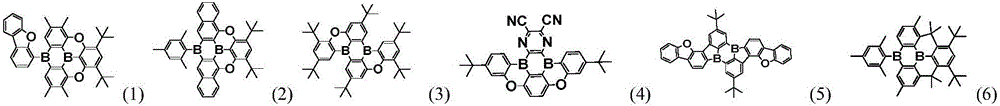
an organic compound with diboron as a core, wherein the structure of the organic compound is shown as a general formula (1):
wherein, X1、X2、X3、X4Each independently represents a single bond, an oxygen atom, a sulfur atom,-CR5=CR6-、-C(R7R8) -or-Si (R)9R10) -one of the above;
a. b, c and d are respectively and independently 0 or 1; and a + b + c + d is 2;
Y1~Y18each independently represents a nitrogen atom or C-R11(ii) a Y at the connection site1~Y18Represented as a carbon atom;
e. f, g and h are respectively and independently 0, 1, 2, 3 or 4;
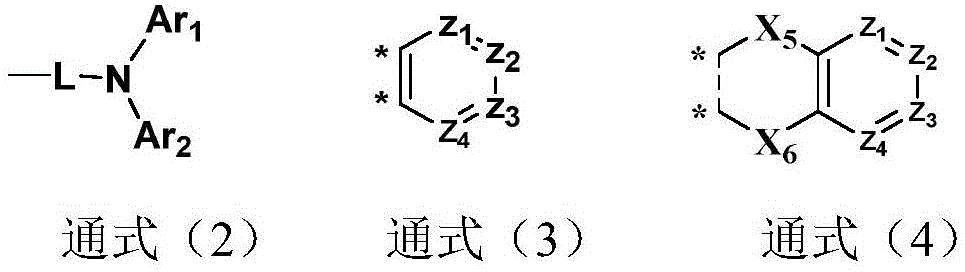
R1~R4each independently represents a hydrogen atom, a structure represented by general formula (2), general formula (3) or general formula (4);
in the general formula (2), L represents a single bond, substituted or unsubstituted C6-30One of arylene, substituted or unsubstituted 5-30 membered heteroarylene containing a heteroatom;
Ar1、Ar2each independently represents substituted or unsubstituted C6-30One of an aryl group, a substituted or unsubstituted 5-30 membered heteroaryl group containing a heteroatom; ar (Ar)1、Ar2Can also be connected with each other to form a ring;
in the general formula (4), X5、X6Each independently represents a single bond, a sulfur atom, an oxygen atom, -N (R)12)-、CR13=CR14、-C(R15R16) -or-Si (R)17R18) -one of (a);
the R is5~R10、R12~R18Are each independently represented by C1-20Straight chain alkyl, C2-20Alkylene radical, C3-20Branched alkyl radical, C1-20Linear alkyl substituted silane radical, C3-20Branched alkyl substituted silane group, substituted or unsubstituted C6-30One of aryl, substituted or unsubstituted 5-to 30-membered heteroaryl containing a heteroatom, and R5、R6、R13、R14May also be represented as a hydrogen atom; r13And R14、R15And R16、R17And R18May be linked to form a 5-to 30-membered aliphatic or aromatic ring, respectively;
in the general formulae (3) and (4), Z1~Z4Each independently represents a nitrogen atom or C-R19;
The R is11、R19Each independently represents a hydrogen atom, a protium atom, a deuterium atom, a tritium atom, a fluorine atom, a cyano group, a phosphoric acid or a salt thereof, or C1-20Straight chain alkyl, C2-20Alkylene radical, C3-20Branched alkyl radical, C1-20Linear alkyl substituted silane radical, C3-20Branched alkyl substituted silane group, substituted or unsubstituted C6-30One of an aryl group, a substituted or unsubstituted 5-to 30-membered heteroaryl group containing a heteroatom; wherein two or more R11、R19The radicals alsoMay be linked to each other to form a 5-to 30-membered aliphatic or aromatic ring;
general formula (3) and general formula (4) are independently through Y1-Y2、Y2-Y3、Y3-Y4、Y5-Y6、Y6-Y7、Y7-Y8、Y8-Y9、Y10-Y11、Y11-Y12、Y12-Y13、Y14-Y15、Y15-Y16、Y16-Y17Or Y17-Y18Is connected with a parallel ring of the general formula (1);
the substituent of the substitutable group is selected from deuterium, cyano, halogen, C1-20Alkyl, protium atom, deuterium atom, tritium atom, C6-30One or more of aryl or 5 to 30 membered heteroaryl containing a heteroatom;
the hetero atom of the heteroaryl is one or more selected from oxygen atom, sulfur atom or nitrogen atom.
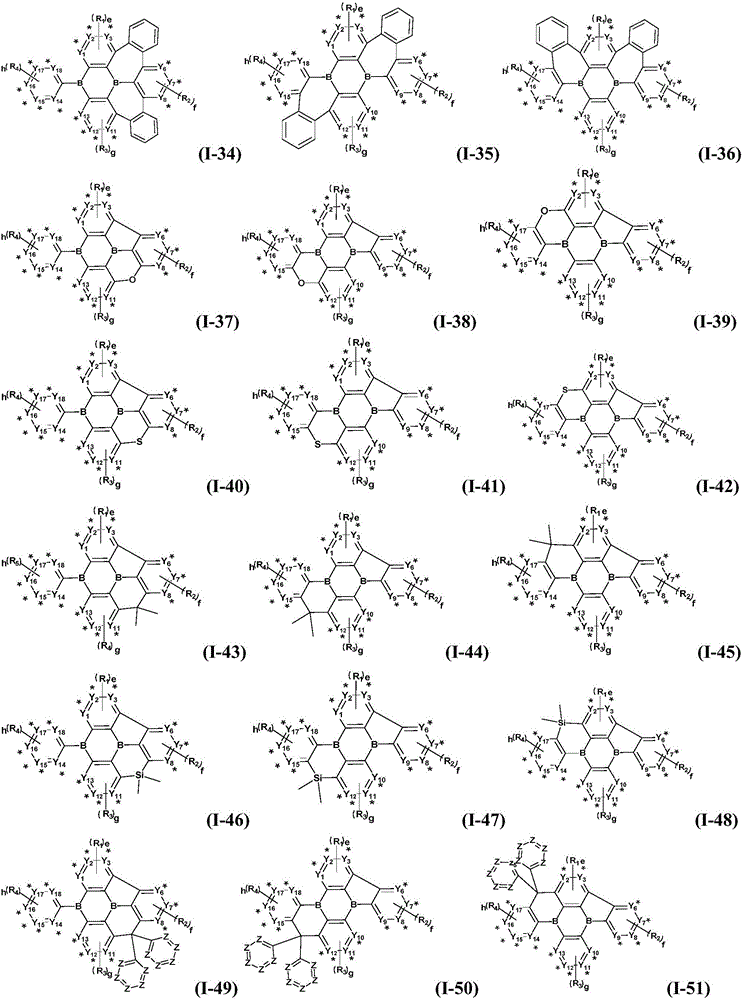
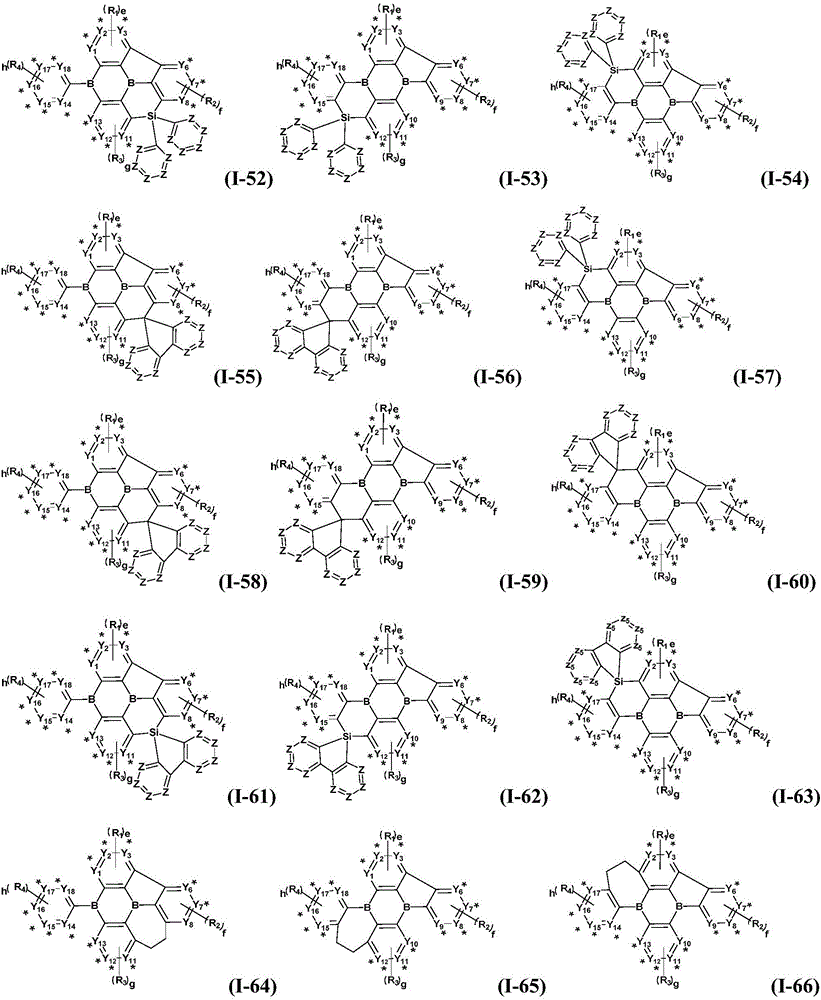
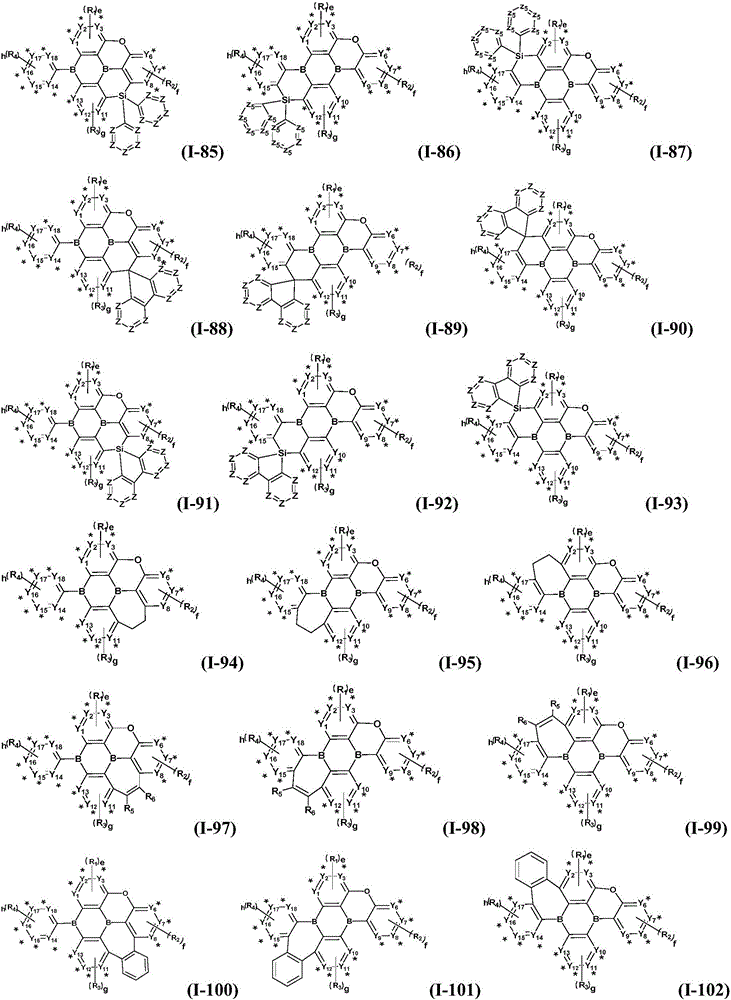
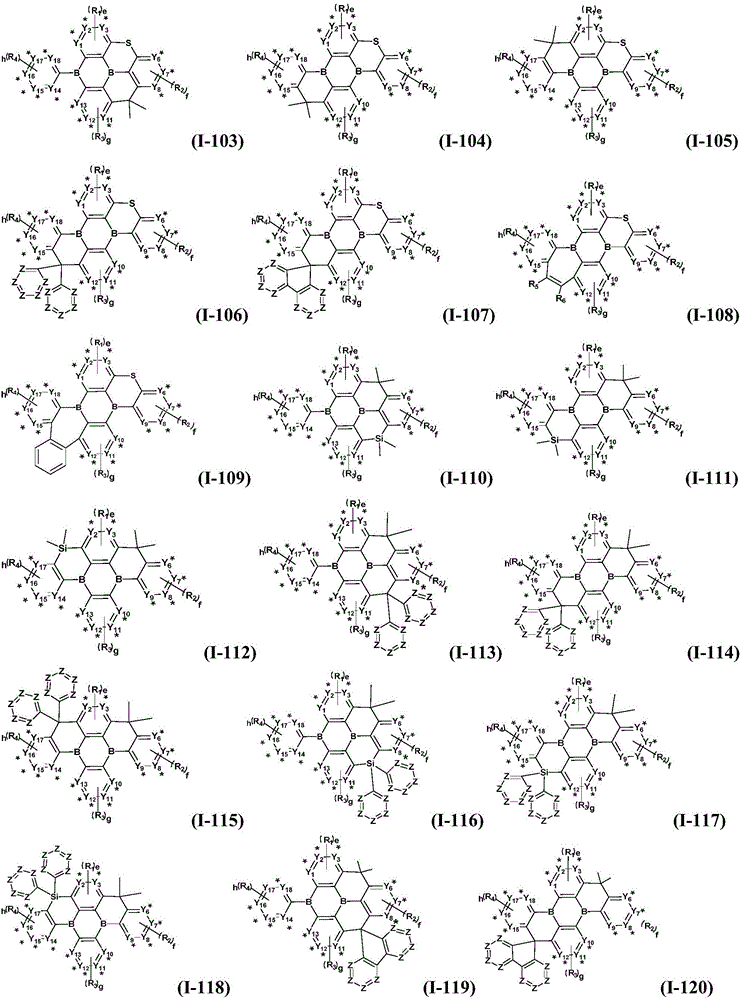
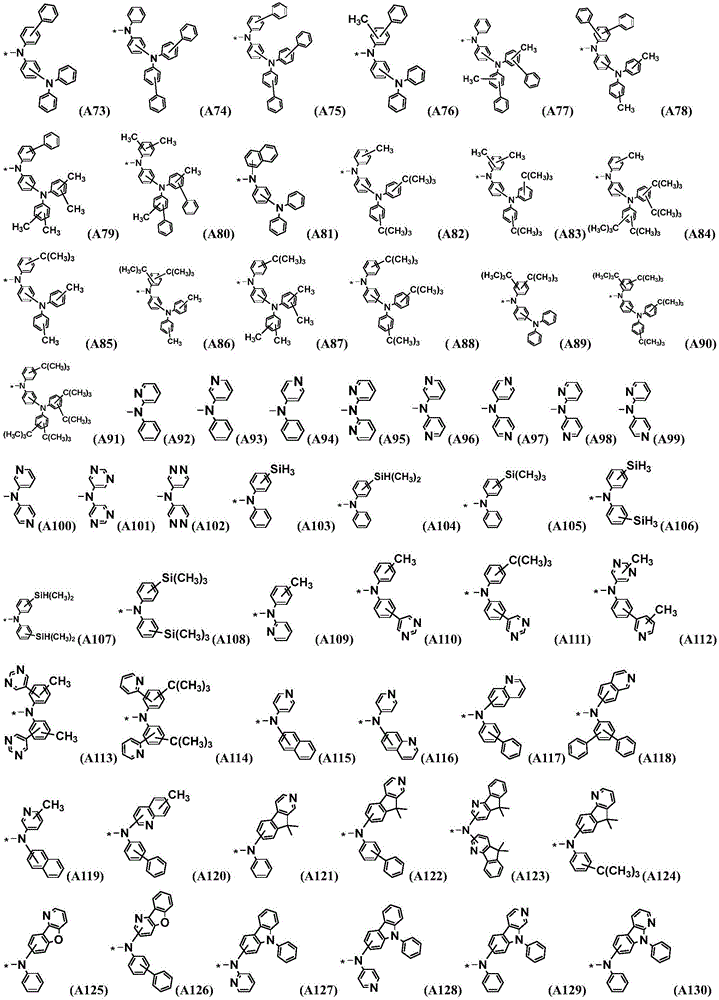
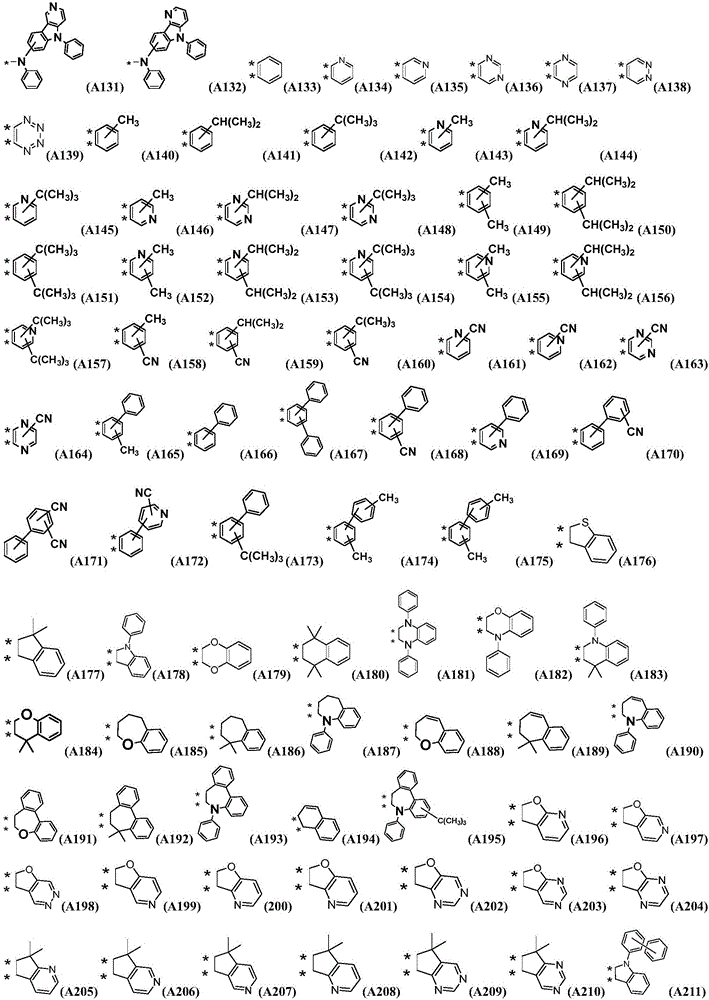
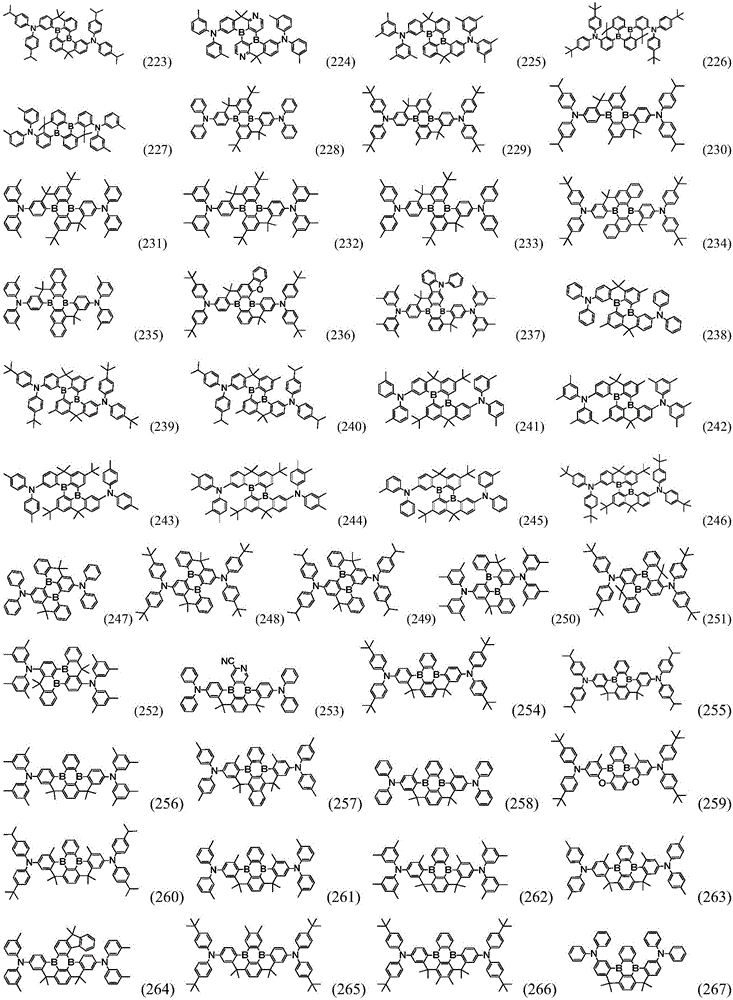
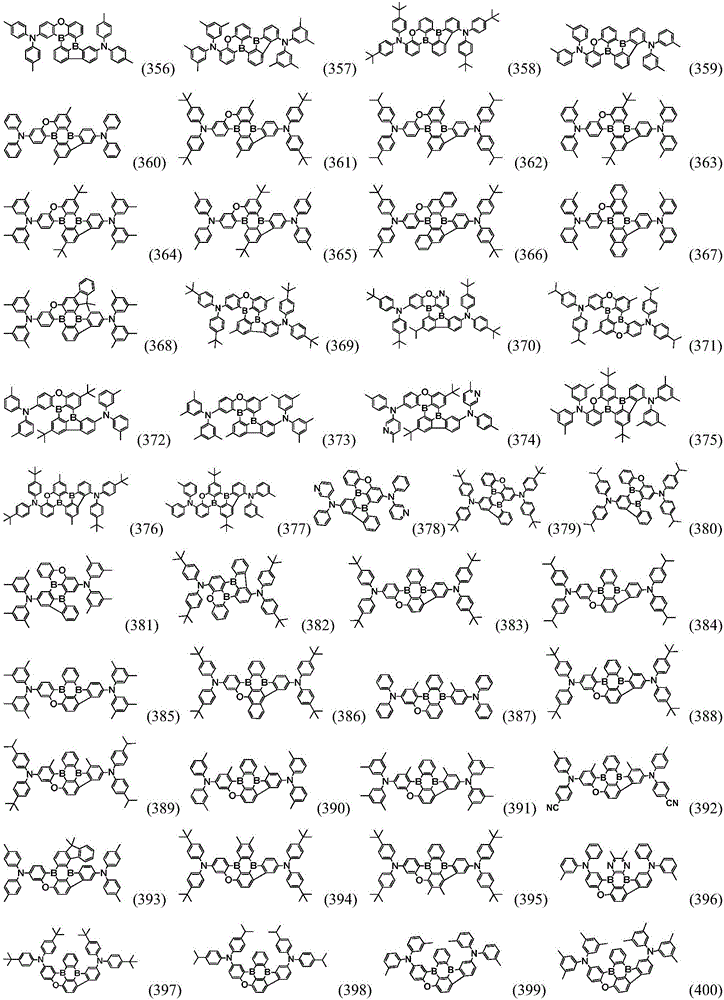
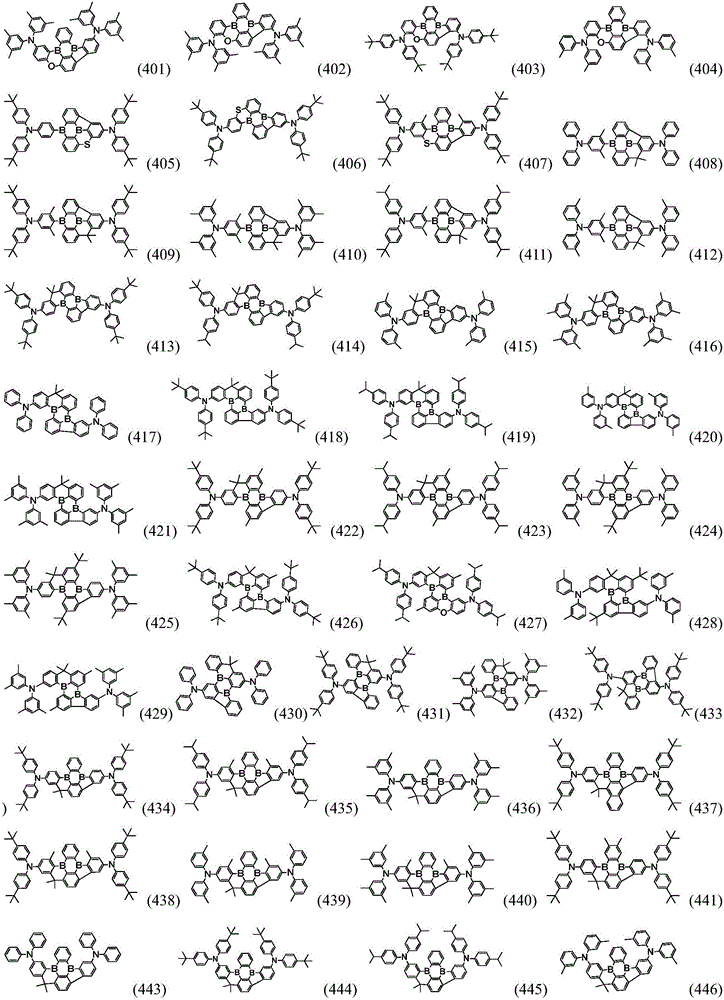
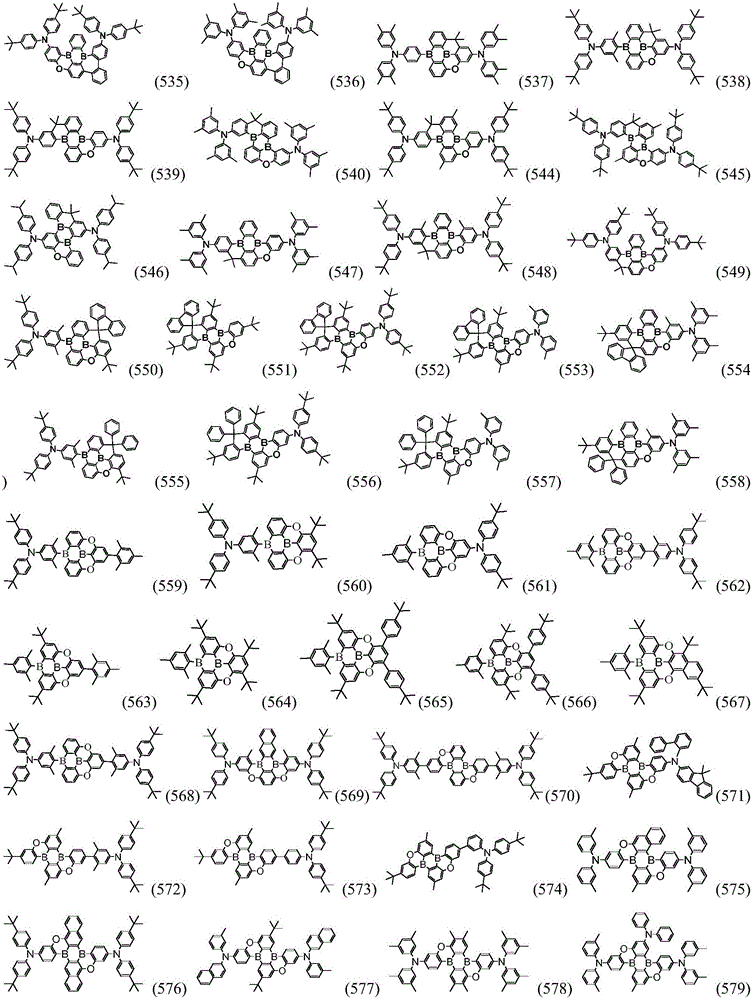
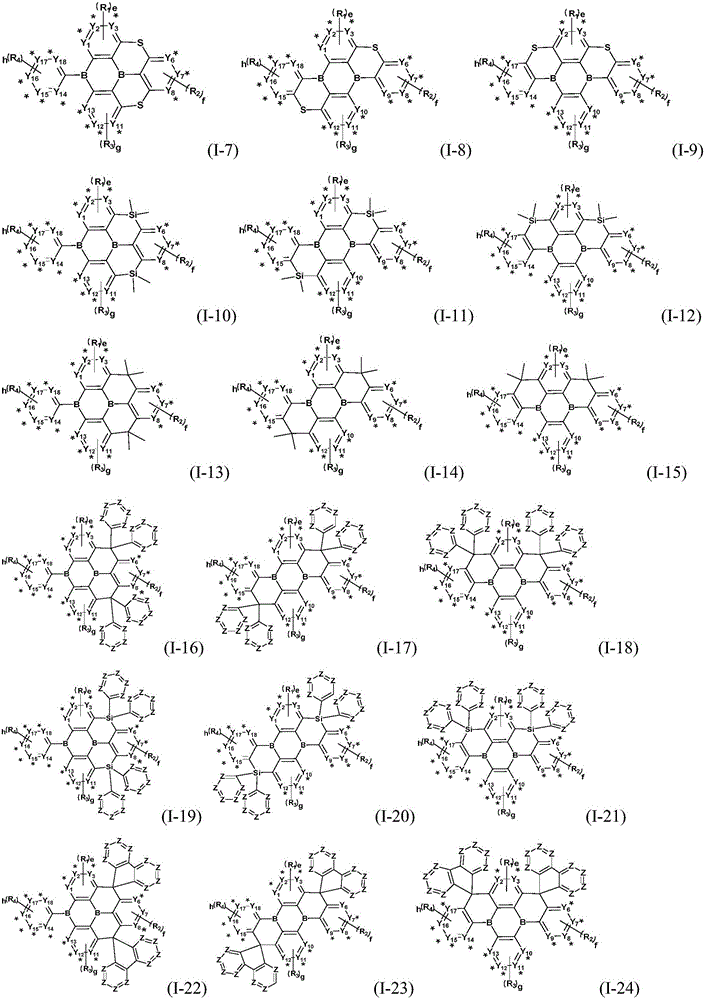
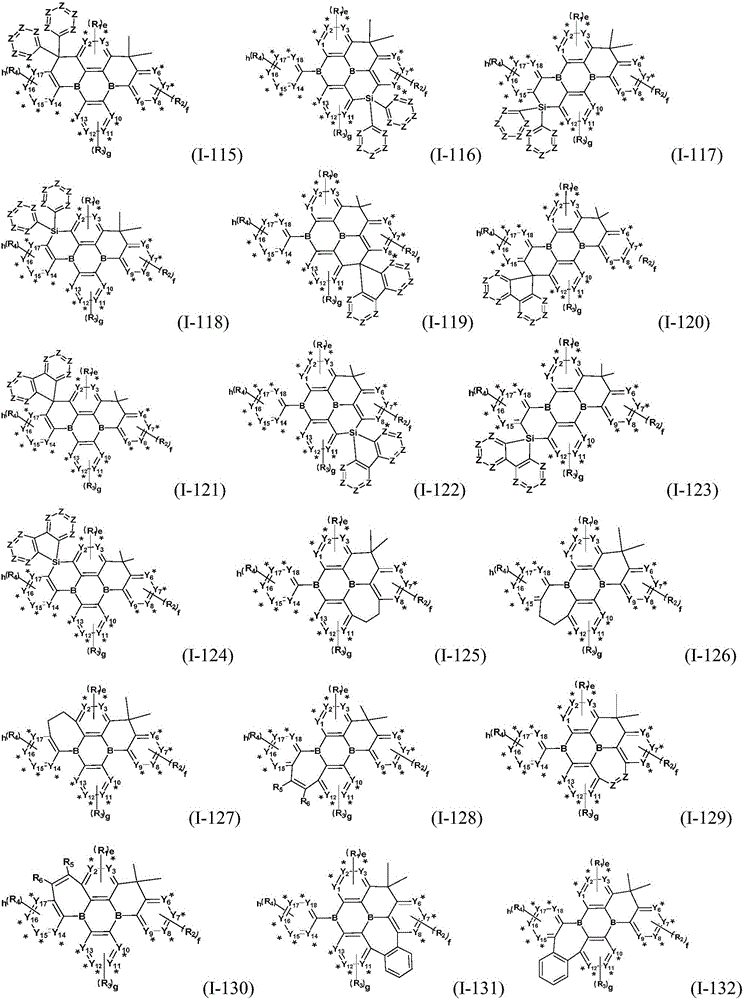
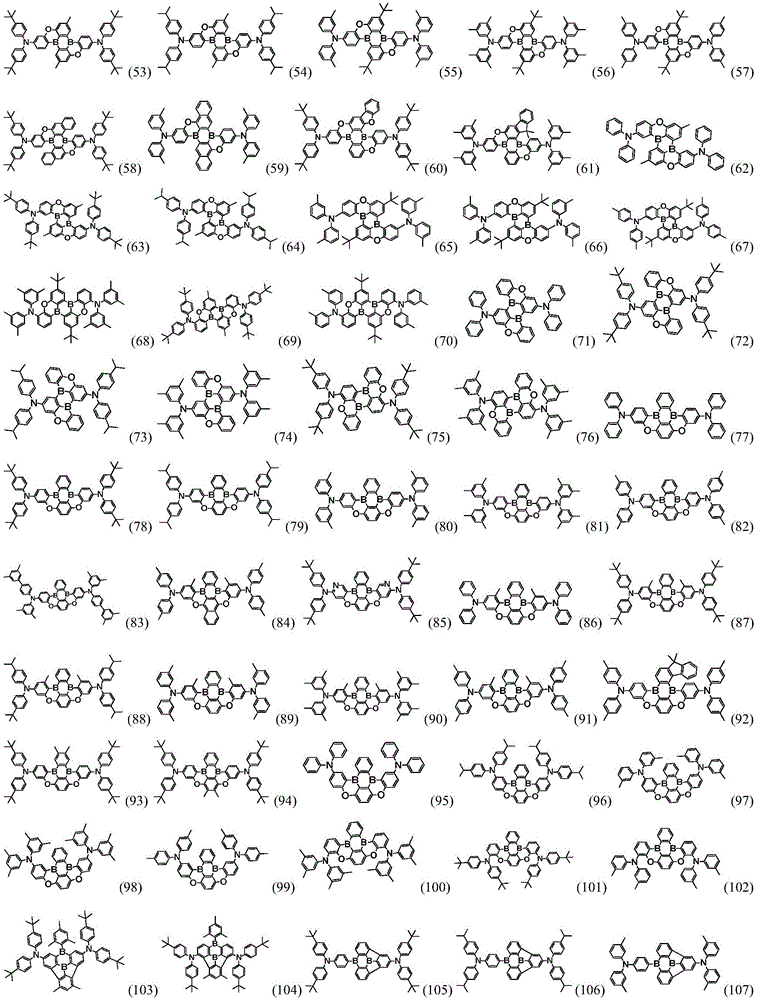
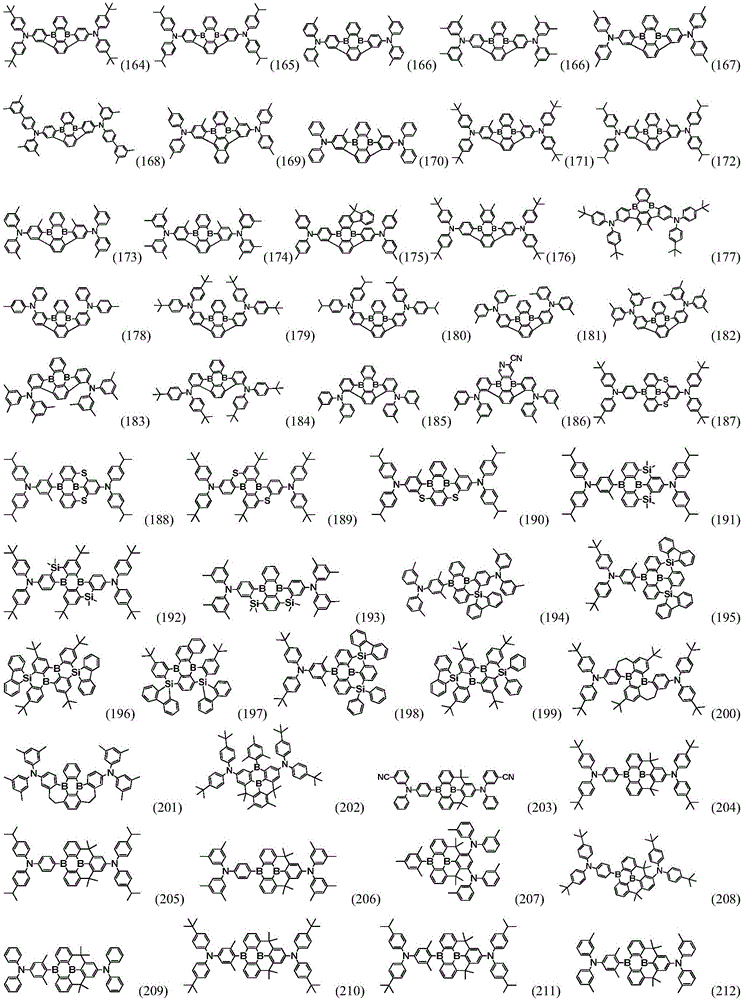
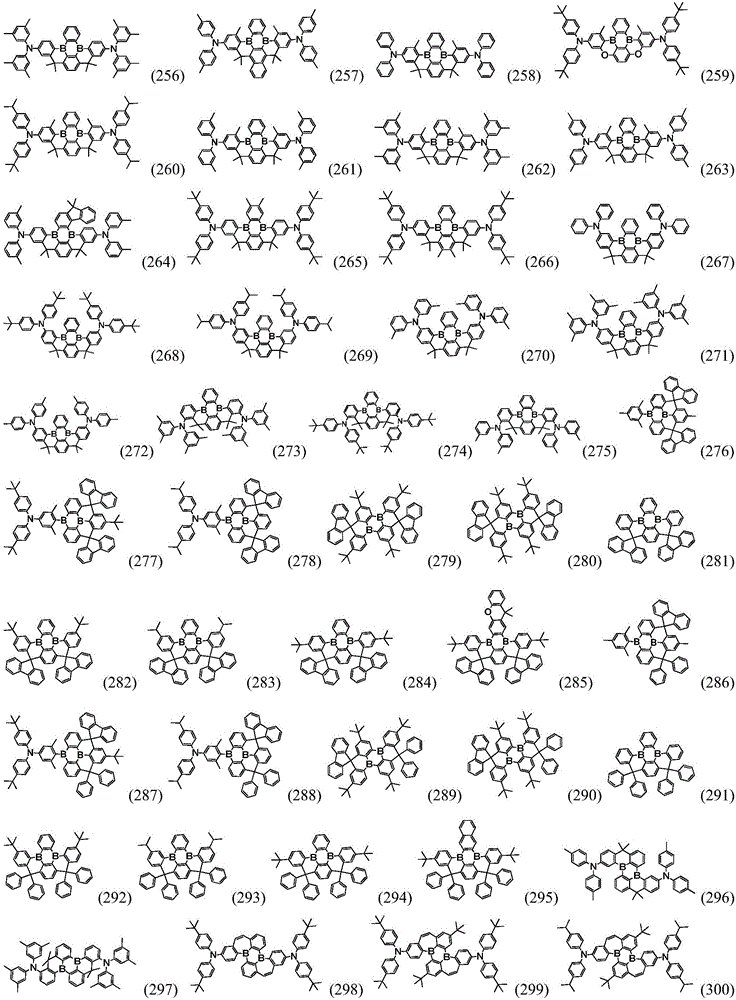
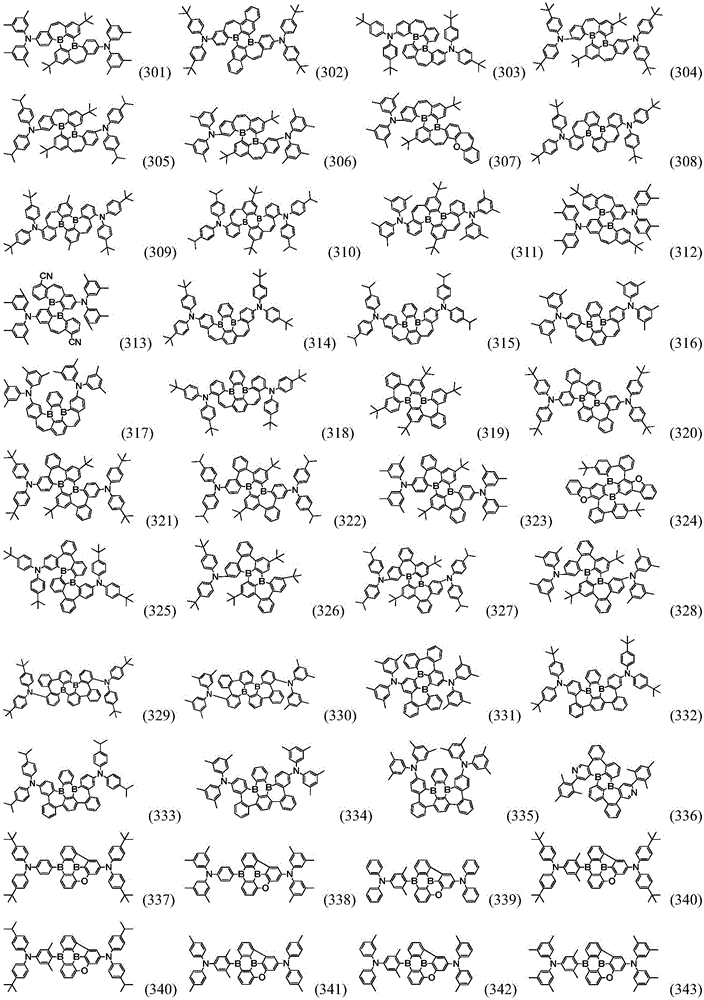
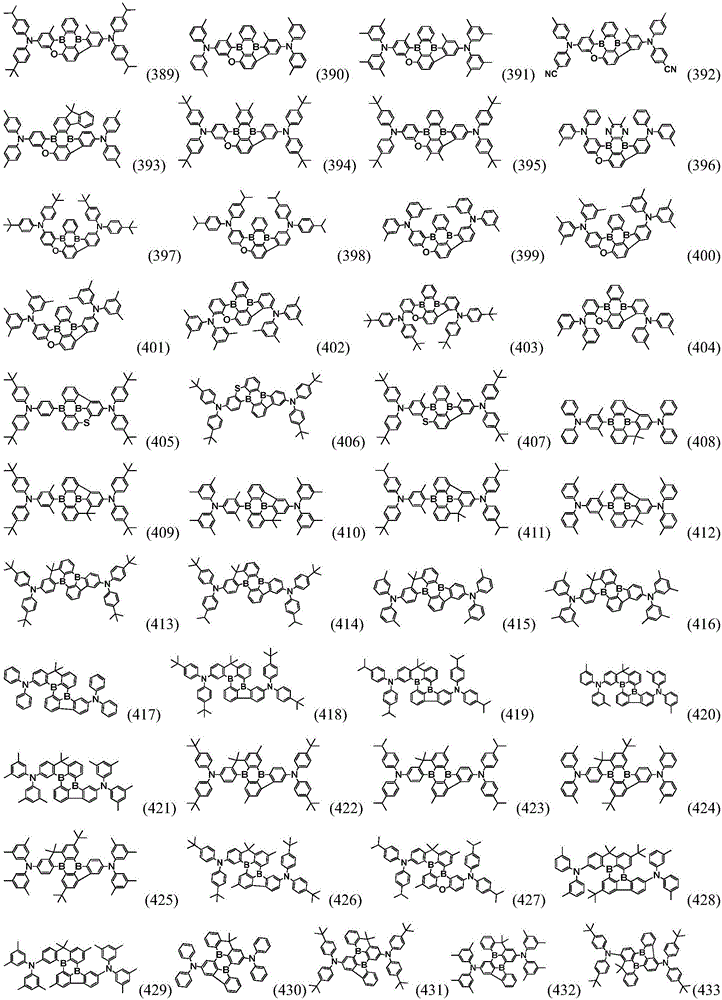
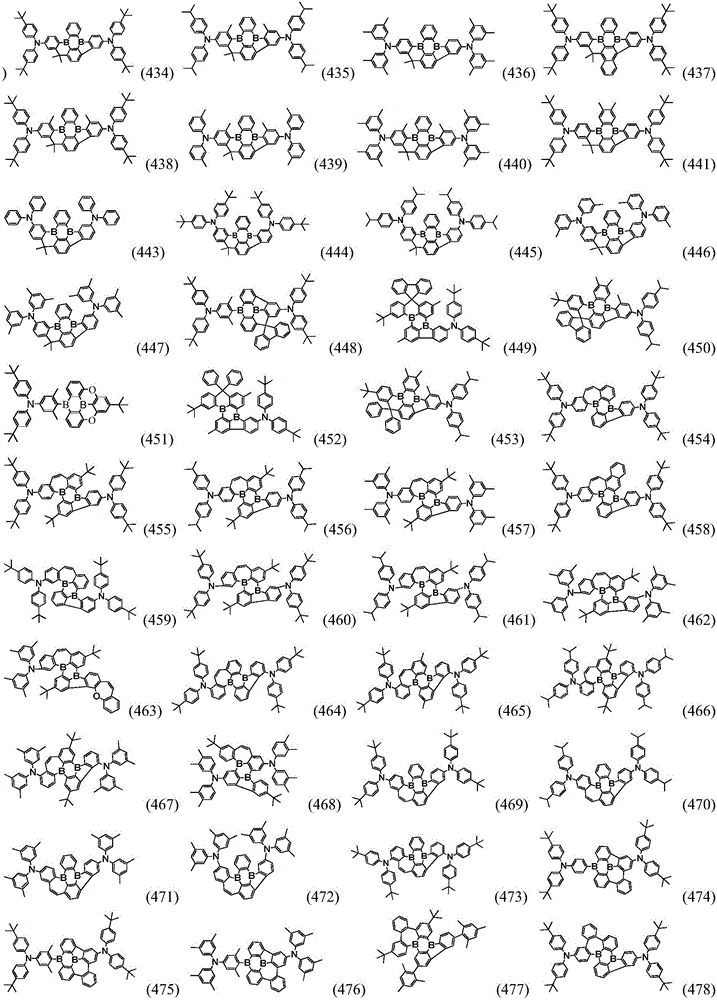
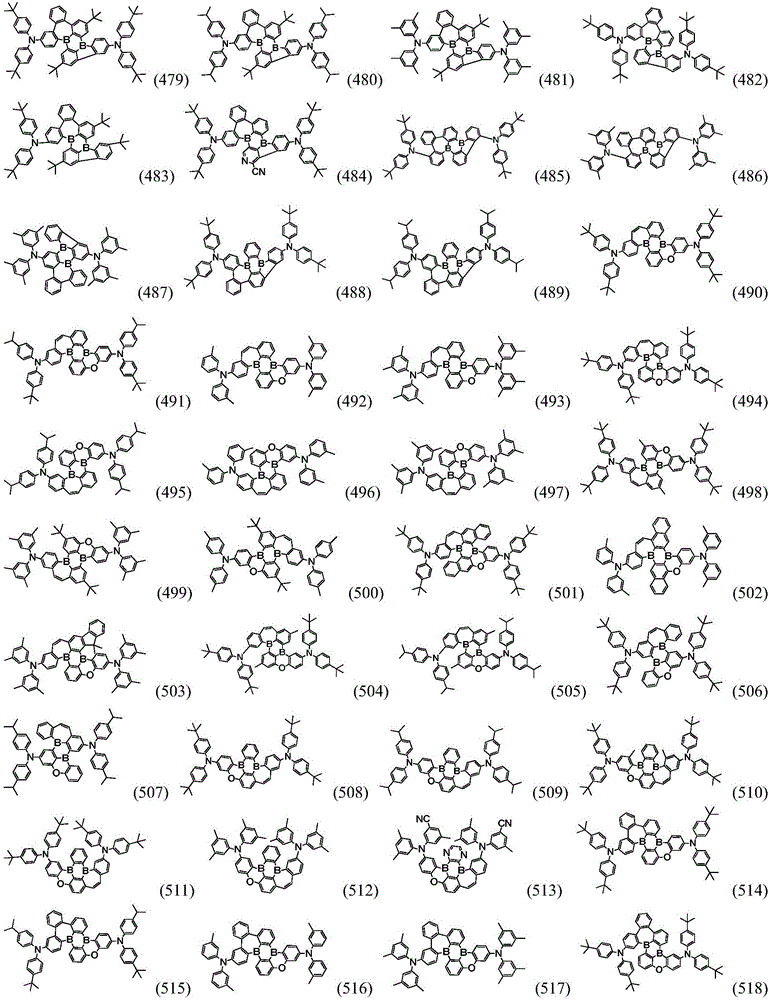
Preferably, the organic compound has a structure represented by any one of general formula (III-1) to general formula (III-3):
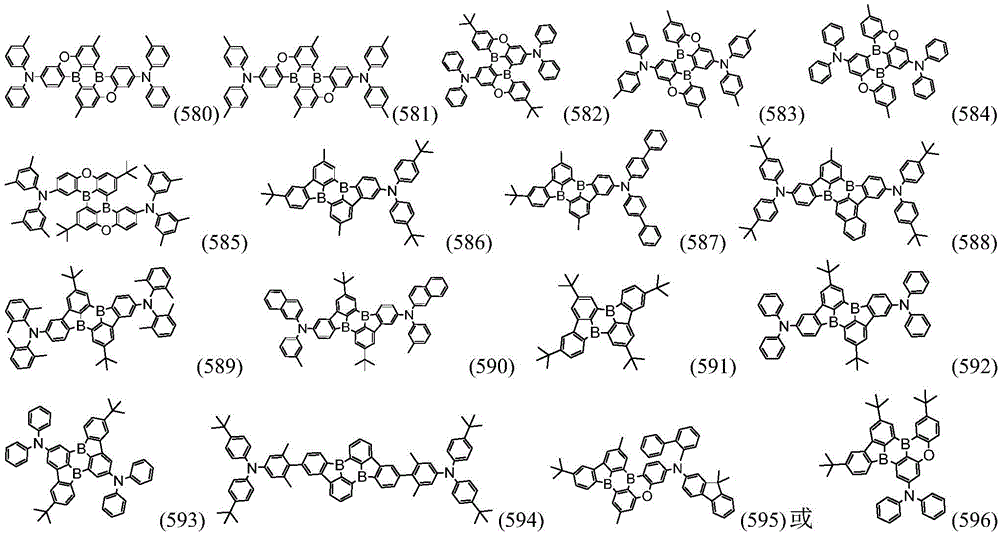
more preferably, the organic compound has a structure represented by any one of general formula (I-1) to general formula (I-133):
z is nitrogen atom or C-R20;R20Represented by hydrogen atom, protium atom, deuterium atom, tritium atom, fluorine atom, cyano group, phosphoric acid or a salt thereof, C1-20Straight chain alkyl, C2-20Alkylene radical, C3-20Branched alkyl radical, C1-20Linear alkyl substituted silane radical, C3-20Branched alkyl substituted silane group, substituted or unsubstituted C6-30One of an aryl group, a substituted or unsubstituted 5-to 30-membered heteroaryl group containing a heteroatom; wherein two or more R20The groups may also be linked to each other to form a 5-to 30-membered aliphatic or aromatic ring;
the substituent of the substitutable group is selected from cyano, halogen and C1-20Alkyl, protium atom, deuterium atom, tritium atom, C6-30One or more of aryl or 5 to 30 membered heteroaryl containing a heteroatom;
the hetero atom of the heteroaryl is one or more selected from oxygen atom, sulfur atom or nitrogen atom.
Preferably, the L represents one of a single bond, a substituted or unsubstituted phenylene group, a substituted or unsubstituted biphenylene group, a substituted or unsubstituted naphthylene group, a substituted or unsubstituted anthracenylene group, a substituted or unsubstituted pyridinylene group, a substituted or unsubstituted pyrimidinylene group, a substituted or unsubstituted pyridazylene group, a substituted or unsubstituted furanylidene group, a substituted or unsubstituted thiophenylene group, a substituted or unsubstituted dibenzofuranylene group, a substituted or unsubstituted dibenzothiophenylene group, and a substituted or unsubstituted carbazolyl group;
Ar1、Ar2each independently represents a substituted or unsubstituted phenyl group, a substituted or unsubstituted biphenylyl group, a substituted or unsubstituted terphenylyl group, a substituted or unsubstituted naphthyl group, a substituted or unsubstituted naphthyridinyl group, a substituted or unsubstituted anthracenyl group, a substituted or unsubstituted phenanthrenyl group, a substituted or unsubstituted pyrenyl group, a substituted or unsubstituted benzophenanthrenyl group, a substituted or unsubstituted azabenzophenanthrenyl group, a substituted or unsubstituted pyridyl group, one of substituted or unsubstituted pyrimidinyl, substituted or unsubstituted pyridazinyl, substituted or substituted triazinyl, substituted or unsubstituted furan, substituted or unsubstituted thiophene, substituted or unsubstituted dibenzofuranyl, substituted or unsubstituted dibenzothiophenyl, substituted or unsubstituted carbazolyl, substituted or unsubstituted naphthocarbazolyl, substituted or unsubstituted naphthofuranyl, substituted or unsubstituted azacarbazolyl;
the R is5~R10、R12~R18Each independently represents methyl, ethyl, propyl, isopropyl, butyl, tert-butyl, pentyl, phenyl, naphthyl, biphenyl, terphenyl, naphthyridinyl or pyridyl;
the R is11、R19、R20Each independently represents a hydrogen atom, a protium atom, a deuterium atom, a tritium atom, a fluorine atom, a cyano group, a methyl group, an ethyl group, a propyl group, an isopropyl group, a butyl group, a tert-butyl group, a pentyl group, a substituted or unsubstituted phenyl group, a substituted or unsubstituted biphenyl group, a substituted or unsubstituted naphthyl group, a substituted or unsubstituted anthracenyl group, a substituted or unsubstituted pyridyl group, a substituted or unsubstituted pyrimidyl group, a substituted or unsubstituted pyridazinyl group, a substituted or substituted triazinyl group, a substituted or unsubstituted furyl group, a substituted or unsubstituted thienyl group, a substituted or unsubstituted dibenzofuryl groupOr unsubstituted dibenzothienyl, substituted or unsubstituted carbazolyl;
the substituent of the substitutable group is one or more selected from deuterium, cyano, methyl, ethyl, propyl, isopropyl, butyl, tertiary butyl, pentyl, cyano, fluorine atom, phenyl, naphthyl, biphenyl, terphenyl, naphthyridinyl or pyridyl.
Further preferably, R1~R4At least one of the structures is represented by a general formula (2), a general formula (3) or a general formula (4).
More preferably, R1 to R4 are each independently represented by:
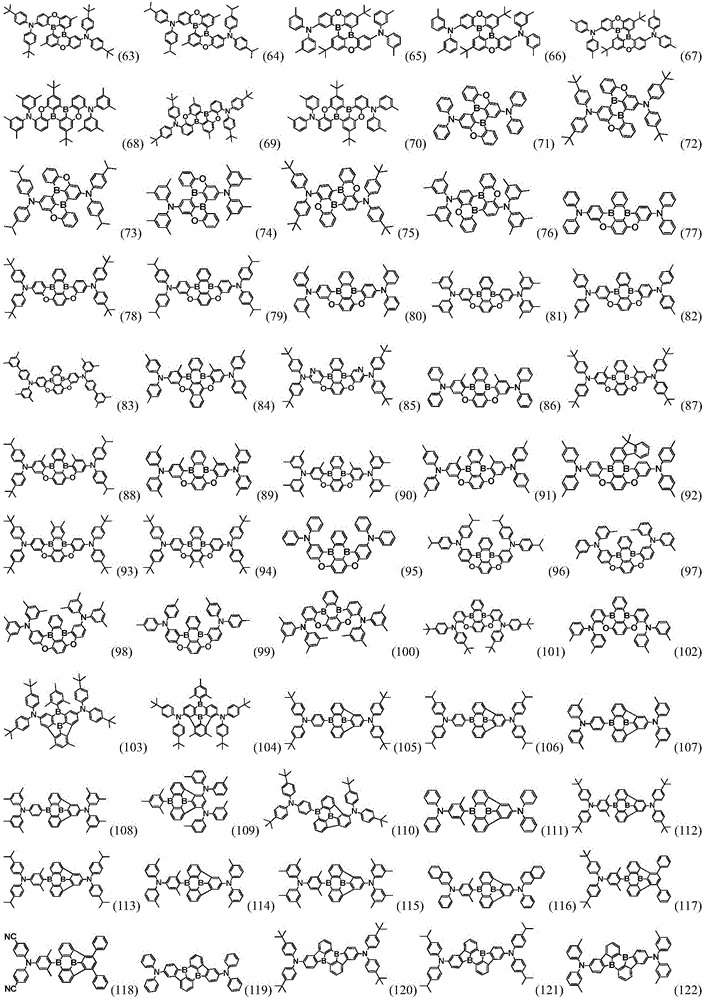
In a preferred embodiment, the specific compound of formula (1) is represented by:
any one of them.
A preparation method of the organic compound comprises the following three conditions:
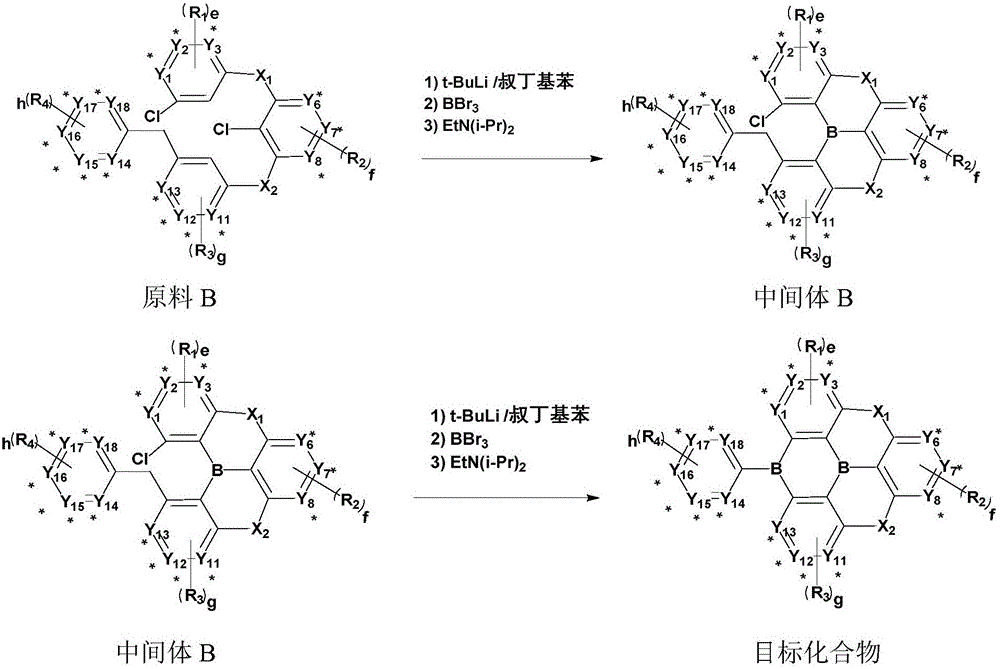
A. in the general formula (1), when a and b are 1, and c and d are 0, the synthesis steps are as follows:
the specific reaction process of the above reaction equation is:
1) under the protection of inert gas, dissolving a raw material B by using tert-butyl benzene, wherein the proportion of the raw material B to the tert-butyl benzene is that every 1g of the raw material B is dissolved by using 50-100 m L tert-butyl benzene;
2) slowly adding tert-butyl lithium into the reaction system in the step 1), and stirring the mixed solution; wherein the molar ratio of the tert-butyl lithium to the raw material B is 1.0-2.0: 1;
3) reacting the mixed solution obtained in the step 2) for 2-4 hours at the temperature of 50-70 ℃, naturally cooling to room temperature, and then slowly dropwise adding BBr3And diisopropylethylamine, and stirring and reacting for 1-4 hours at room temperature; the BBr3The molar ratio of the diisopropylethylamine to the raw material B is 1.0-2.0: 1, and the molar ratio of the diisopropylethylamine to the raw material B is 2.0-3.0: 1;
4) after the reaction is finished, adding water and dichloromethane for extraction and liquid separation, taking an organic phase, adding anhydrous magnesium sulfate for dewatering, filtering, carrying out rotary evaporation on the filtrate until no solvent exists, and passing through a neutral silica gel column to obtain an intermediate B;
5) under the protection of inert gas, dissolving an intermediate B by using tert-butyl benzene, wherein the ratio of the intermediate B to the tert-butyl benzene is that every 1g of the intermediate B is dissolved by using 50-100 m L tert-butyl benzene;
6) slowly adding tert-butyl lithium into the reaction system in the step 5), and stirring the mixed solution; wherein the molar ratio of the tert-butyl lithium to the intermediate B is 1.0-2.0: 1;
7) reacting the mixed solution obtained in the step 6) for 2-4 hours at the temperature of 50-70 ℃, naturally cooling to room temperature, and then slowly dropwise adding BBr3And diisopropylethylamine, stirred at room temperature and reactedThe reaction time is 1-4 hours; the BBr3The molar ratio of the diisopropylethylamine to the intermediate B is 1.0-2.0: 1, and the molar ratio of the diisopropylethylamine to the intermediate A is 2.0-3.0: 1;
8) after the reaction is finished, adding water and dichloromethane for extraction and liquid separation, taking an organic phase, adding anhydrous magnesium sulfate for dewatering, filtering, carrying out rotary evaporation on the filtrate until no solvent exists, and passing through a neutral silica gel column to obtain a target compound;
B. in the general formula (1), when a and c are 1 and b and d are 0, the synthesis steps are as follows:
the specific reaction process of the above reaction equation is:
1) under the protection of inert gas, dissolving a raw material C-I and a raw material C-II by using tert-butyl benzene, wherein the proportion of the raw material C-I to the tert-butyl benzene is that every 1g of the raw material C-I is dissolved by using 30-80 m L m of tert-butyl benzene, and the molar ratio of the raw material C-I to the raw material C-II is 1: 1;
2) slowly adding tert-butyl lithium into the reaction system in the step 1), and stirring the mixed solution; wherein the molar ratio of the tert-butyl lithium to the raw material C-I is 2.0-3.0: 1;
3) reacting the mixed solution obtained in the step 2) for 2-4 hours at the temperature of 50-70 ℃, naturally cooling to room temperature, and then slowly dropwise adding BBr3And diisopropylethylamine, and stirring and reacting for 1-4 hours at room temperature; the BBr3The molar ratio of the diisopropylethylamine to the raw material C-I is 2.0-3.0: 1, and the molar ratio of the diisopropylethylamine to the raw material C-I is 3.0-6.0: 1;
4) after the reaction is finished, adding water and dichloromethane for extraction and liquid separation, taking an organic phase, adding anhydrous magnesium sulfate for dewatering, filtering, carrying out rotary evaporation on the filtrate until no solvent exists, and passing through a neutral silica gel column to obtain a target compound;
C. in the general formula (1), when a and d are 1 and b and c are 0, the synthesis steps are as follows:
the specific reaction process of the above reaction equation is:
1) under the protection of inert gas, dissolving a raw material D-I and a raw material D-II by using tert-butyl benzene, wherein the proportion of the raw material D-I to the tert-butyl benzene is that every 1g of the raw material D-I is dissolved by using 30-80 m L m of tert-butyl benzene, and the molar ratio of the raw material D-I to the raw material D-II is 1: 1;
2) slowly adding tert-butyl lithium into the reaction system in the step 1), and stirring the mixed solution; wherein the molar ratio of the tert-butyl lithium to the raw material D-I is 2.0-3.0: 1;
3) reacting the mixed solution obtained in the step 2) for 2-4 hours at the temperature of 50-70 ℃, naturally cooling to room temperature, and then slowly dropwise adding BBr3And diisopropylethylamine, and stirring and reacting for 1-4 hours at room temperature; the BBr3The molar ratio of the diisopropylethylamine to the raw material D-I is 2.0-3.0: 1, and the molar ratio of the diisopropylethylamine to the raw material D-I is 3.0-6.0: 1;
4) after the reaction is finished, adding water and dichloromethane for extraction and liquid separation, taking an organic phase, adding anhydrous magnesium sulfate for dewatering, filtering, carrying out rotary evaporation on the filtrate until no solvent exists, and passing through a neutral silica gel column to obtain a target compound;
an organic electroluminescent device prepared from organic compounds with double boron as a core.
The organic electroluminescent device has light emitting layer of organic compound with double boron as core.
A lighting or display element comprising said organic electroluminescent device.
The beneficial technical effects of the invention are as follows:
the compound structure molecule of the invention contains the combination of electron donor (donor, D) and electron acceptor (acceptor, A) which can increase the orbit overlap and improve the luminous efficiency, and simultaneously connects diarylamine group to obtain the charge transfer state material with HOMO and L UMO space separation, the double boron system material has strong electron-withdrawing action, so that the overlap of the front line orbit between the electron donors connected with the double boron system material is smaller, the small energy level difference between S1 state and T1 state is realized, thereby realizing the reverse intersystem crossing under the condition of thermal stimulation, the boron-containing compound can destroy the crystallinity of the molecule because D-A forms a certain dihedral angle and simultaneously connects diarylamine group, avoids the aggregation action between molecules, has good film forming property and fluorescence quantum efficiency, and is suitable for being used as the main body material or the doping material of the luminous layer.
The compound disclosed by the invention is simple in preparation method, wide in market prospect and suitable for large-scale popularization and application.
When the compound provided by the invention is applied to an O L ED device, high film stability can be kept through device structure optimization, the photoelectric performance of the O L ED device and the service life of the O L ED device can be effectively improved, and the compound provided by the invention has good application effect and industrialization prospect in an O L ED light-emitting device.
The organic electroluminescent device can be applied to lighting or display elements, so that the current efficiency, the power efficiency and the external quantum efficiency of the device are greatly improved, meanwhile, the service life of the device is obviously prolonged, the organic electroluminescent device has a good application effect in an O L ED light-emitting device, and has a good industrialization prospect.
Drawings
FIG. 1 is a schematic structural diagram of an O L ED device according to the present invention;
in the figure: 1 is a transparent substrate layer, 2 is an ITO anode layer, 3 is a hole injection layer, 4 is a hole transport layer, 5 is an electron blocking layer, 6 is a light-emitting layer, 7 is a hole blocking layer or an electron transport layer, 8 is an electron injection layer, and 9 is a cathode reflection electrode layer.
Fig. 2 is a graph of efficiency measured at different temperatures for a device made according to the present invention and a device of a comparative example.
Detailed Description
The present invention will be described in detail with reference to the accompanying drawings and examples.
The structural formula of the materials referred to herein is as follows:
The detection method used herein is as follows:
Glass transition temperature Tg: measured by differential scanning calorimetry (DSC, DSC204F1 DSC, German Nasicon company), the rate of temperature rise was 10 ℃/min.
Thermogravimetric analysis (TGA-50H thermogravimetric analyzer from Shimadzu corporation, Japan) performed at a temperature at which the weight loss was 0.5% in a nitrogen atmosphere, and the nitrogen flow rate was 20m L/min.
△ Est is the difference between the singlet level and the triplet level of the material, and is obtained by measuring the fluorescence emission spectrum and the phosphorescence emission spectrum of the compound, respectively, and calculating the fluorescence emission peak and the phosphorescence emission peak (measuring equipment: F L S980 fluorescence spectrometer by Edinburgh Instruments, Optistat DN-V2 low-temperature component by Oxford Instruments).
Highest occupied molecular orbital HOMO energy level: is tested by an ionization energy testing system (IPS3) in an atmospheric environment.
Cyclic voltammetry stability was determined by observing the redox characteristics of the material using cyclic voltammetry under conditions in which the test sample was dissolved in a 2:1 volume ratio solvent mixture of dichloromethane and acetonitrile at a concentration of 1mg/M L, the electrolyte was a 0.1M organic solution of tetrabutylammonium tetrafluoroborate, the reference electrode was Ag/Ag+The electrode, the counter electrode is a titanium plate, the working electrode is an ITO electrode, and the cycle time is 20 times.
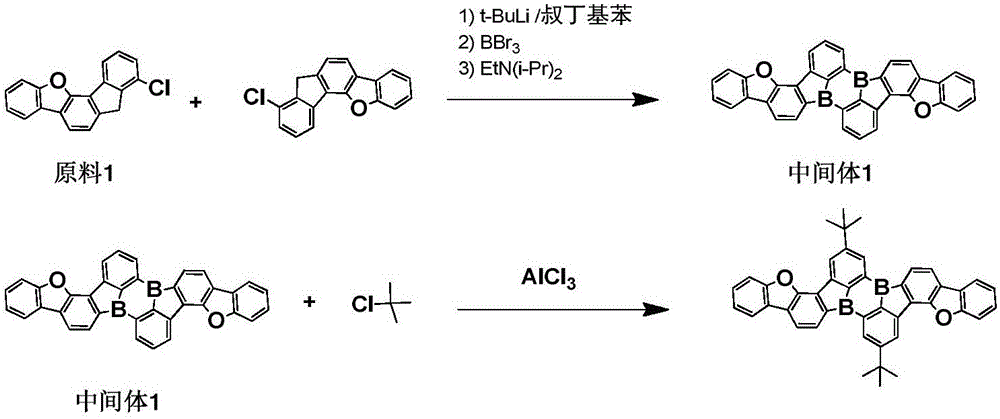
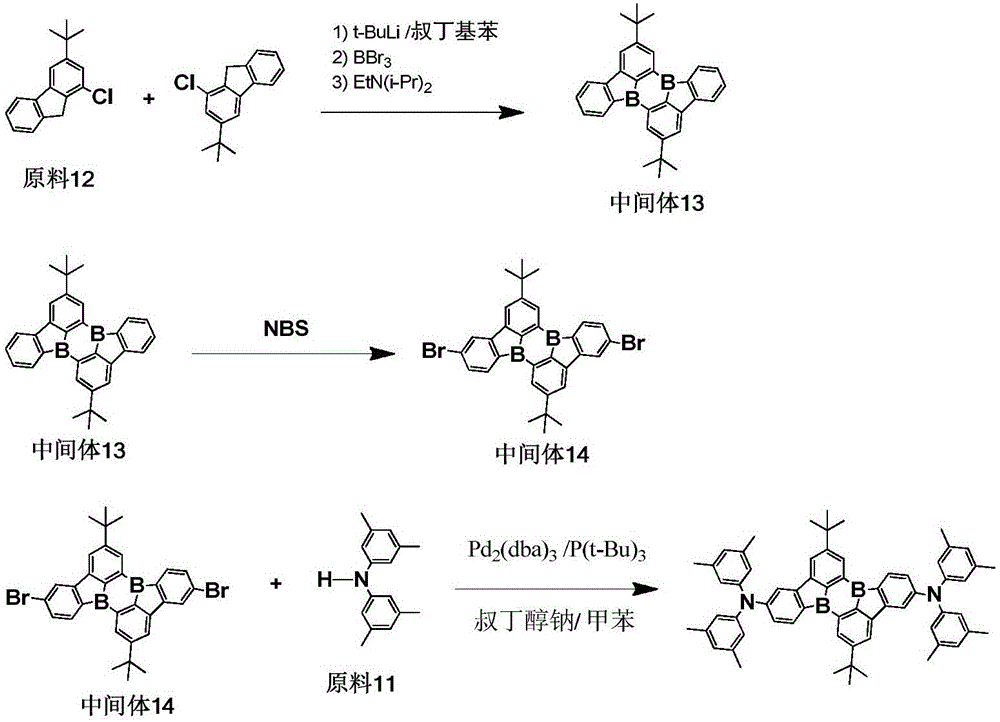
< example 1> preparation of Compound 5
1) Adding 0.01mol of raw material 1, 0.015mol of tert-butyl lithium and 150ml of tert-butyl benzene into a 250ml three-necked bottle under the protection of nitrogen gas, stirring and mixing, heating to 60 ℃, stirring and reacting for 2 hours, then naturally cooling to room temperature, dropwise adding 0.015mol of BBr3 and 0.1mol of diisopropylethylamine, stirring and reacting for 1 hour at room temperature, taking a sample point plate to show that no raw material 1 is left, completely reacting, adding water and dichloromethane for extraction and liquid separation, taking an organic phase, adding anhydrous magnesium sulfate for water removal, filtering, carrying out reduced pressure rotary evaporation on the filtrate at (-0.09MPa, 25 ℃) to obtain an intermediate 1, wherein the purity of HP L C is 98.5%, and the yield is 45.7%;
elemental analysis Structure (molecular formula C)36H18B2O2): theoretical value C, 85.77; h, 3.60; b, 4.29; o, 6.35; test values are: c, 85.77; h, 3.61; b, 4.28; o, 6.34. ESI-MS (M/z) (M +): the theoretical value is 504.15, found 504.64.
2) Adding 0.01mol of intermediate 1, 200ml of dichloromethane and 0.03mol of tert-chlorobutane into a three-neck flask with the thickness of 250m L, cooling to 0-5 ℃, and then adding 0.03mol of anhydrous AlCl3Reacting for 2-5 h, completely reacting, hydrolyzing, extracting with dichloromethane, separating, collecting organic phase, adding anhydrous magnesium sulfate to remove water, filtering, performing reduced pressure rotary evaporation on the filtrate (0.09 MPa, 25 ℃), passing through a neutral silica gel column to obtain the target product compound 5, wherein the purity of HP L C is 99.1%, and the yield is 52.5%.
Elemental analysis Structure (molecular formula C)44H34B2O2): theoretical value C, 85.74; h, 5.56; b, 3.51; o, 5.19; test values are: c, 85.75; h, 5.55; b, 3.50; and O, 5.20. ESI-MS (M/z) (M)+): theoretical value is 616.27, found 616.71.
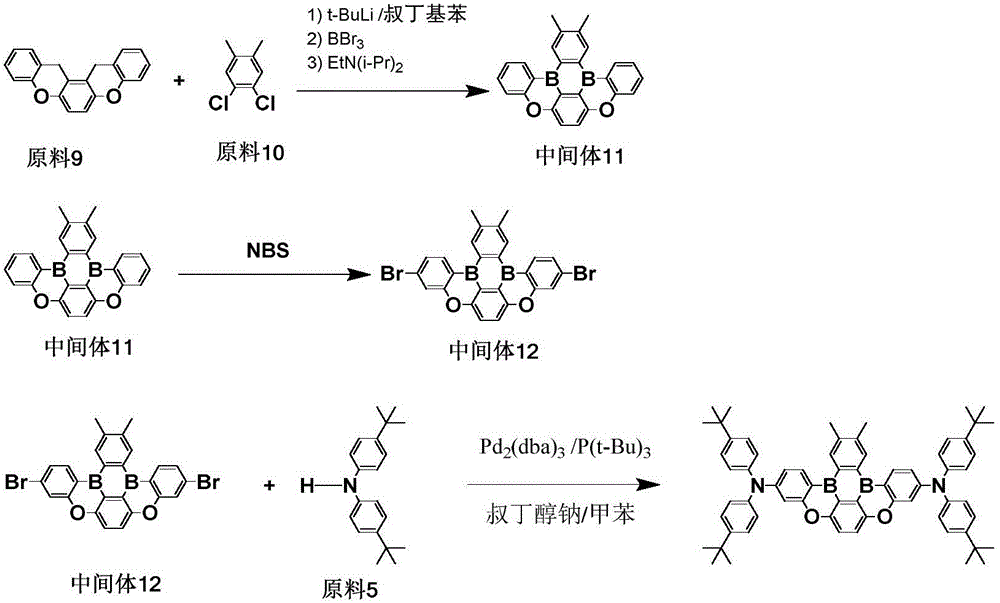
< example 2> preparation of Compound 11
1) Adding 0.01mol of raw material 2, 0.015mol of tert-butyl lithium and 150ml of tert-butyl benzene into a 250ml three-necked bottle under the protection of nitrogen gas, stirring and mixing, heating to 60 ℃, stirring and reacting for 2 hours, then naturally cooling to room temperature, dropwise adding 0.015mol of BBr3 and 0.1mol of diisopropylethylamine, stirring and reacting for 1 hour at room temperature, taking a sample point plate to show that no raw material 2 remains and completely reacting, adding water and dichloromethane for extraction and liquid separation, taking an organic phase, adding anhydrous magnesium sulfate for water removal, filtering, carrying out reduced pressure rotary evaporation on the filtrate at (-0.09MPa, 25 ℃) to obtain an intermediate 2, wherein the purity of HP L C is 98.7%, and the yield is 54.1%;
elemental analysisStructure (molecular formula C)28H22B2O2): theoretical value C, 81.61; h, 5.38; b, 5.25; o, 7.76; test values are: c, 81.62; h, 5.37; b, 5.26; and O, 7.75. ESI-MS (M/z) (M +): theoretical value is 412.18, found 412.57.
2) 0.01mol of intermediate 2 was added to a 250m L three-necked flask, and dissolved in 150m L of dichloromethane, stirred at room temperature (20-25 ℃), 0.03mol of NBS (N-bromosuccinimide) was added in portions, and the reaction was observed by thin layer chromatography (T L C) until completion, the reaction mixture was poured into 200m L of water and stirred for 2 hours, extracted with dichloromethane, separated, the organic phase was taken, anhydrous magnesium sulfate was added to remove water, filtered, and subjected to reduced pressure rotary evaporation (-0.09MPa, 25 ℃) to obtain intermediate 3, which was 98.5% pure at HP L C and 73.1% yield.
Elemental analysis Structure (molecular formula C)28H20B2Br2O2): theoretical value C, 59.01; h, 3.54; b, 3.79; br, 28.04; o, 5.61; test values are: c, 59.02; h, 3.55; b, 3.77; br, 28.03; and O, 5.63. ESI-MS (M/z) (M)+): theoretical value is 568.00, found 568.55.
3) To a 250m L three-necked flask, 0.02mol of the prepared intermediate 3, 0.05mol of the starting material 3, 0.06mol of sodium tert-butoxide, 2.5 × 10 mol were added under a nitrogen atmosphere-5mol Pd2(dba)3And 2.5 × 10-5And (2) adding 150m of L toluene into the mixture to dissolve the tri-tert-butylphosphine, heating the mixture to 100 ℃, refluxing the mixture for 24 hours, observing the reaction by utilizing T L C until the reaction is complete, naturally cooling the mixture to room temperature, filtering the mixture, and rotatably steaming the filtrate until no fraction is produced, wherein the obtained substance is purified by a silica gel column to obtain the compound 11, the purity of the compound is 99.3%, and the yield of the compound is 65.9%.
Elemental analysis Structure (molecular formula C)62H48B2N2O2): theory C, 85.14; h, 5.53; b, 2.47; n, 3.20; o, 3.66; test values are: c, 85.13; h, 5.52; b, 2.46; n, 3.21; and O, 3.68. ESI-MS (M/z) (M)+): theoretical value is 874.39, found 874.79.
< example 3> preparation of Compound 15
1) Adding 0.01mol of 4, 0.015mol of tert-butyl lithium and 150ml of tert-butyl benzene into a 250ml three-necked bottle under the protection of nitrogen gas, stirring and mixing, heating to 60 ℃, stirring and reacting for 2 hours, then naturally cooling to room temperature, dropwise adding 0.015mol of BBr3 and 0.1mol of diisopropylethylamine, stirring and reacting for 1 hour at room temperature, taking a sample point plate to show that no 4 is left, completely reacting, adding water and dichloromethane for extraction and liquid separation, taking an organic phase, adding anhydrous magnesium sulfate for water removal, filtering, carrying out reduced pressure rotary evaporation on the filtrate at (-0.09MPa, 25 ℃) to obtain an intermediate 4, wherein the purity of HP L C is 98.6%, and the yield is 66.5%;
elemental analysis Structure (molecular formula C)25H16BClO2): theoretical value C, 76.08; h, 4.09; b, 2.74; cl, 8.98; o, 8.11; test values are: c, 76.03; h, 4.10; b, 2.75; cl, 8.99; and O, 8.13. ESI-MS (M/z) (M +): theoretical value is 394.09, found 394.65.
2) Adding 0.01mol of intermediate 4, 0.015mol of tert-butyl lithium and 150ml of tert-butyl benzene into a 250ml three-necked bottle under the protection of nitrogen gas, stirring and mixing, heating to 60 ℃, stirring and reacting for 2 hours, then naturally cooling to room temperature, dropwise adding 0.015mol of BBr3 and 0.1mol of diisopropylethylamine, stirring and reacting for 1 hour at room temperature, taking a sample point plate, indicating that no intermediate 4 is left, completely reacting, adding water and dichloromethane for extraction and liquid separation, taking an organic phase, adding anhydrous magnesium sulfate for water removal, filtering, carrying out reduced pressure rotary evaporation on the filtrate at (-0.09MPa, 25 ℃) to obtain an intermediate 5, wherein the purity of HP L C is 98.9%, and the yield is 59.3%;
elemental analysis Structure (molecular formula C)24H14B2O2): theoretical value C, 80.97; h, 3.96; b, 6.07; o, 8.99; test values are: c, 80.99; h, 3.98; b, 6.06; o, 8.97. ESI-MS (M/z) (M +): theoretical value is 356.12, found 356.83.
3) 0.01mol of intermediate 5 was added to a 250m L three-necked flask, and dissolved in 150m L of dichloromethane, stirred at room temperature (20-25 ℃), 0.03mol of NBS (N-bromosuccinimide) was added in portions, and the reaction was observed by thin layer chromatography (T L C) until completion, the reaction mixture was poured into 200m L of water and stirred for 2 hours, extracted with dichloromethane, separated, the organic phase was taken, anhydrous magnesium sulfate was added to remove water, filtered, and subjected to reduced pressure rotary evaporation (-0.09MPa, 25 ℃) to neutral silica gel column to obtain intermediate 6, which was 98.5% pure at HP L C and 73.1% yield.
Elemental analysis Structure (molecular formula C)24H12B2Br2O2): theoretical value C, 56.11; h, 2.35; b, 4.21; br, 31.10; o, 6.23; test values are: c, 56.12; h, 2.33; b, 4.20; br, 31.11; and O, 6.24. ESI-MS (M/z) (M)+): theoretical value is 511.94, found 512.51.
4) To a 250m L three-necked flask, 0.02mol of the prepared intermediate 6, 0.05mol of the starting material 5, 0.06mol of sodium tert-butoxide, 2.5 × 10 mol were added under a nitrogen atmosphere-5mol Pd2(dba)3And 2.5 × 10-5And (2) adding 150m of L toluene into the mixture to dissolve the tri-tert-butylphosphine, heating the mixture to 100 ℃, refluxing the mixture for 24 hours, observing the reaction by utilizing T L C until the reaction is complete, naturally cooling the mixture to room temperature, filtering the mixture, and carrying out rotary evaporation on the filtrate until no fraction is produced, wherein the obtained substance is purified by a silica gel column to obtain the compound 50 with the purity of 99.5 percent and the yield of 59.2 percent.
Elemental analysis Structure (molecular formula C)64H64B2N2O2): theory C, 84.03; h, 7.05; b, 2.36; n, 3.06; o, 3.50; test values are: c, 84.04; h, 7.04; b, 2.38; n, 3.05; and O, 3.49. ESI-MS (M/z) (M)+): theoretical value is 914.52, found 914.94.
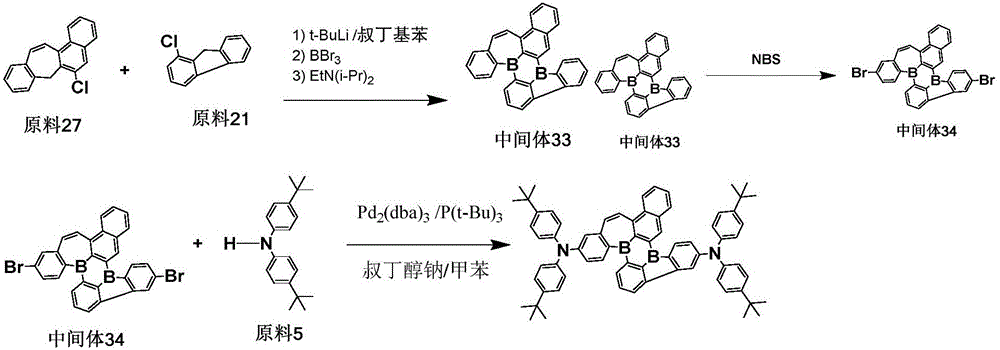
< example 4> preparation of Compound 37
The procedure of example 2 was repeated, except that the starting material 6 was used in place of the starting material 2 to prepare an intermediate 7, bromination was carried out to obtain an intermediate 8, and the intermediate 8 and the starting material 7 were subjected to coupling reaction to obtain the objective compound 37 with a purity of 98.3% and a yield of 68.7%.
Elemental analysis Structure (molecular formula C)60H56B2N2O2): theoretical value C, 83.92; h, 6.57; b, 2.52; n, 3.26; o, 3.73; test values are: c, 83.93; h, 6.55; b, 2.51; n, 3.28; and O, 3.74. ESI-MS (M/z) (M)+): theoretical value is 858.45, found 858.66.
< example 5> preparation of Compound 58
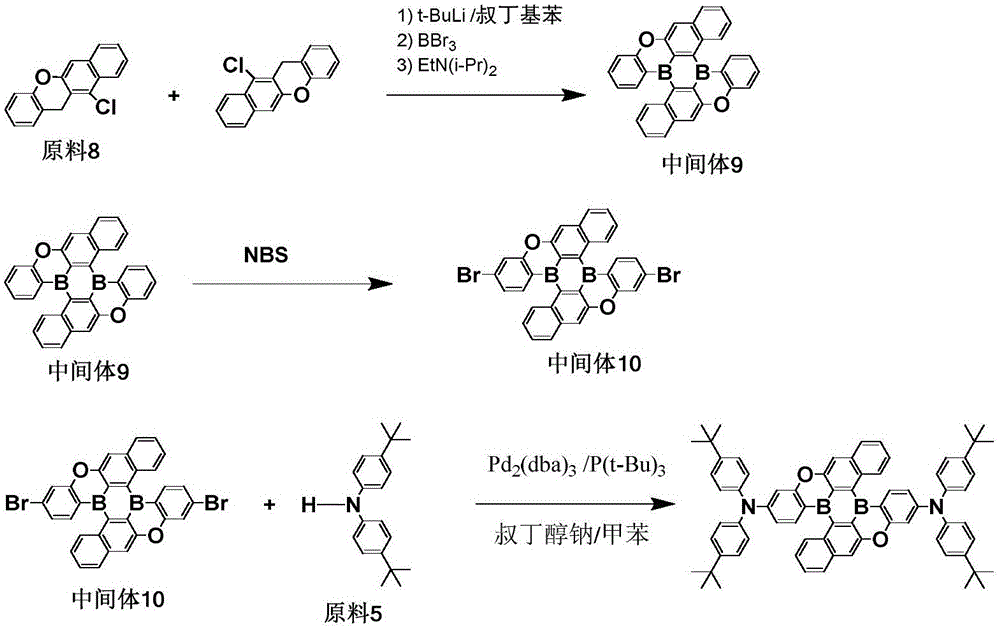
The procedure of example 2 was repeated except that raw material 8 was used instead of raw material 2 to prepare intermediate 9, bromination was carried out to obtain intermediate 10, and coupling reaction was carried out using intermediate 10 and raw material 5 to obtain target compound 58 with a purity of 99.6% and a yield of 68.9%.
Elemental analysis Structure (molecular formula C)72H68B2N2O2): theoretical value C, 85.20; h, 6.75; b, 2.13; n, 2.76; o, 3.15; test values are: c, 85.20; h, 6.76; b, 2.14; n, 2.77; and O, 3.13. ESI-MS (M/z) (M)+): theoretical value is 1014.55, found 1014.96.
< example 6> preparation of Compound 93
1) Adding 0.005mol of raw material 9, 0.005mol of raw material 10, 0.015mol of tert-butyl lithium and 150ml of tert-butyl benzene into a 250ml three-necked bottle under the protection of nitrogen gas, stirring and mixing, heating to 60 ℃, stirring and reacting for 2 hours, then naturally cooling to room temperature, dropwise adding 0.015mol of BBr3 and 0.1mol of diisopropylethylamine, stirring and reacting for 1 hour at room temperature, taking a sample point plate to show that no raw material 7 remains and the reaction is complete, adding water and dichloromethane for extraction and liquid separation, taking an organic phase, adding anhydrous magnesium sulfate for dewatering, filtering, carrying out reduced pressure rotary evaporation on the filtrate (-0.09MPa, 25 ℃), passing through a neutral silica gel column to obtain an intermediate 11, wherein the purity of HP L C is 98.5%, and the yield is 57.9%;
elemental analysis Structure (molecular formula C)26H18B2O2): theoretical value C, 81.31; h, 4.72; b, 5.63; o, 8.33; test values are: c, 81.32; h, 4.73; b, 5.60; and O, 8.35. ESI-MS (M/z) (M +): theoretical value is 384.15, found 384.62.
2) 0.01mol of the intermediate 11 was added to a 250m L three-necked flask, and dissolved in 150m L of dichloromethane, stirred at room temperature (20-25 ℃), 0.03mol of NBS (N-bromosuccinimide) was added in portions, and the reaction was observed by thin layer chromatography (T L C) until completion, the reaction mixture was poured into 200m L of water and stirred for 2 hours, extracted with dichloromethane, separated, and the organic phase was taken, dehydrated with anhydrous magnesium sulfate, filtered, and subjected to reduced pressure rotary evaporation (-0.09MPa, 25 ℃) to a neutral silica gel column to obtain the intermediate 12, which was 99.1% pure at HP L C and 77.5% yield.
Elemental analysis Structure (molecular formula C)26H16B2Br2O2): theoretical value C, 57.63; h, 2.98; b, 3.99; br, 29.49; o, 5.91; test values are: c, 57.61; h, 2.99; b, 4.00; br, 29.50; and O, 5.90. ESI-MS (M/z) (M)+): theoretical value is 539.97, found 540.25.
3) To a 250m L three-necked flask, 0.02mol of the prepared intermediate 12, 0.05mol of the starting material 5, 0.06mol of sodium t-butoxide, 2.5 × 10 mol were added under a nitrogen atmosphere-5mol Pd2(dba)3And 2.5 × 10-5And (2) adding 150m of L toluene into the mixture to dissolve the tri-tert-butylphosphine, heating the mixture to 100 ℃, refluxing the mixture for 24 hours, observing the reaction by utilizing T L C until the reaction is complete, naturally cooling the mixture to room temperature, filtering the mixture, and carrying out rotary evaporation on the filtrate until no fraction is produced, wherein the obtained substance is purified by a silica gel column to obtain the compound 93 with the purity of 99.5 percent and the yield of 72.1 percent.
Elemental analysis Structure (molecular formula C)66H68B2N2O2): theory C, 84.07; h, 7.27; b, 2.29; n, 2.97; o, 3.39; test values are: c, 84.09; h, 7.26; b, 2.30; n, 2.95; and O, 3.40. ESI-MS (M/z) (M)+): theoretical value is 942.55, found 942.89.
< example 7> preparation of Compound 140
The procedure of example 2 was repeated, except that the starting material 12 was used in place of the starting material 2 to prepare an intermediate 13, bromination was carried out to obtain an intermediate 14, and the intermediate 14 and the starting material 11 were subjected to coupling reaction to obtain the objective compound 140 with a purity of 99.7% and a yield of 57.1%.
Elemental analysis Structure (molecular formula C)64H64B2N2): theoretical value C, 87.07; h, 7.31; b, 2.45; n, 3.17; test values are: c, 87.07; h, 7.32; b, 2.46; and N, 3.15. ESI-MS (M/z) (M)+): theoretical value is 882.53, found 882.95.
< example 8> preparation of Compound 171
The procedure of example 6 was repeated except that starting material 13 was used instead of starting material 9 and starting material 14 was used instead of starting material 10 to prepare intermediate 15, bromination was carried out to obtain intermediate 16, and intermediate 16 and starting material 5 were used for coupling reaction to obtain the title compound 171 with a purity of 99.5% and a yield of 67.7%.
Elemental analysis Structure (molecular formula C)66H68B2N2): theoretical value C, 87.03; h, 7.52; b, 2.37; n, 3.08; test values are: c, 87.04; h, 7.51; b, 2.35; and N, 3.10. ESI-MS (M/z) (M)+): theoretical value is 910.56, found 910.99.
< example 9> preparation of Compound 189
The procedure of example 2 was repeated, except that raw material 15 was used instead of raw material 2, intermediate 17 was prepared, intermediate 18 was obtained after bromination, and intermediate 18 and raw material 5 were used for coupling reaction to obtain the objective compound 189 with a purity of 99.5% and a yield of 55.7%.
Elemental analysis Structure (molecular formula C)72H80B2N2S2): theoretical value C, 81.65; h, 7.61; b, 2.04; n, 2.64; s, 6.05; test values are: c, 81.64; h, 7.62; b, 2.05; n, 2.65; and S, 6.03. ESI-MS (M/z) (M)+): theoretical value is 1058.59, found 1058.88.
< example 10> preparation of Compound 217
The procedure of example 3 was repeated, except that the raw material 16 was used in place of the raw material 4, to prepare an intermediate 19, then to prepare a diboron compound intermediate 20, after bromination to obtain an intermediate 21, and the intermediate 21 was subjected to a coupling reaction with the raw material 5 to obtain the desired compound 217 with a purity of 99.0% and a yield of 48.5%.
Elemental analysis Structure (molecular formula C)64H63B2N): theoretical value C, 88.58; h, 7.32; b, 2.49; n, 1.61; test values are: c, 88.59; h, 7.31; b, 2.48; n, 1.62. ESI-MS (M/z) (M)+): the theoretical value is 867.51, and the actual value is 867.92.
< example 11> preparation of Compound 257
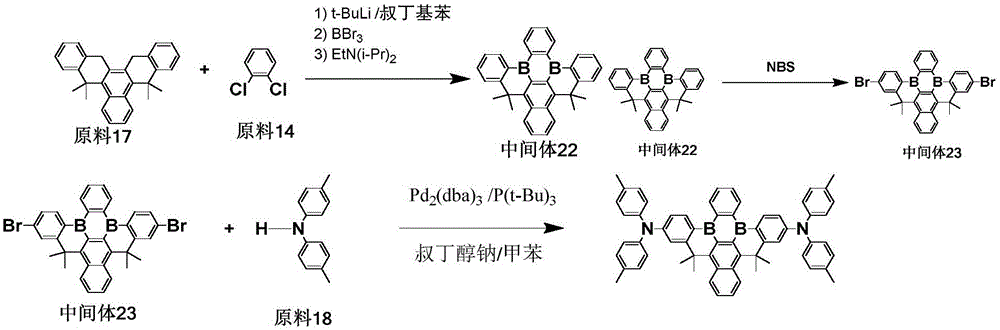
The procedure of example 6 was repeated, except that raw material 17 was used instead of raw material 9, raw material 14 was used instead of raw material 10, intermediate 22 was prepared, bromination was carried out to give intermediate 23, and intermediate 23 and raw material 18 were used for coupling reaction to give the objective compound 257 in a purity of 99.7% and a yield of 63.2%.
Elemental analysis Structure (molecular formula C)62H54B2N2): theoretical value C, 87.74; h, 6.41; b, 2.55; n, 3.30; test values are: c,87.72;H,6.43;B,2.54;N,3.32。ESI-MS(m/z)(M+): theoretical value is 848.45, found 848.76.
< example 12> preparation of Compound 280
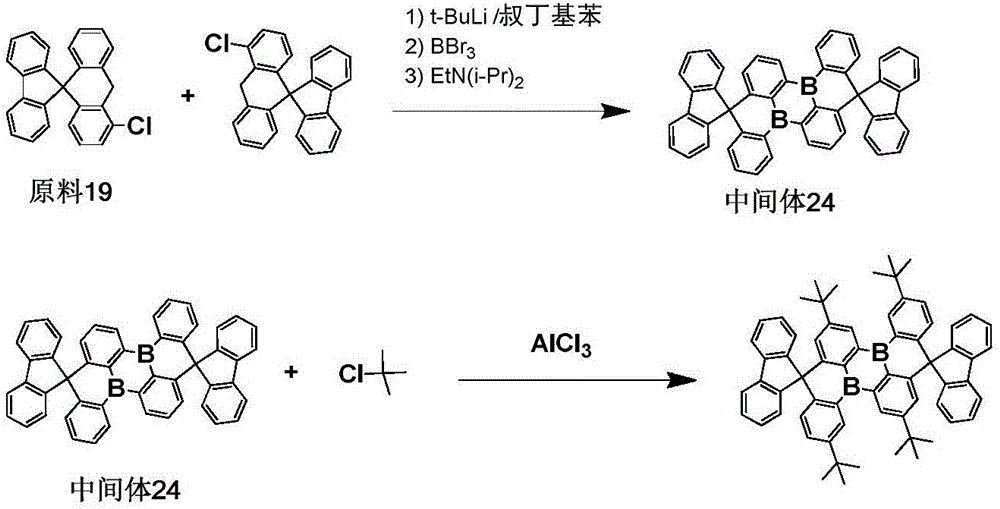
The procedure of example 1 was repeated, except that the starting material 19 was used in place of the starting material 1 to prepare an intermediate 24, which was then substituted with a tert-butyl group, to obtain the objective compound 280 in a purity of 99.6% in a yield of 75.3%.
Elemental analysis Structure (molecular formula C)66H62B2): theoretical value C, 90.41; h, 7.13; b, 2.47; test values are: c, 90.41; h, 7.14; b, 2.45. ESI-MS (M/z) (M)+): the theoretical value is 876.50, and the actual value is 877.02.
< example 13> preparation of Compound 321
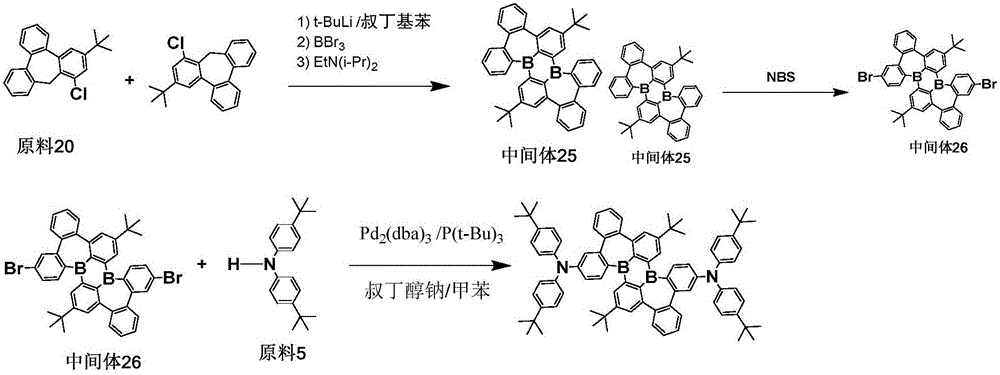
The procedure of example 2 was repeated except that the starting material 20 was used in place of the starting material 2 to prepare an intermediate 25, bromination was carried out to obtain an intermediate 26, and the intermediate 26 was subjected to a coupling reaction with the starting material 5 to obtain the target compound 321; the purity was 99.7% and the yield was 61.1%.
Elemental analysis Structure (molecular formula C)84H88B2N2): theoretical value C, 87.94; h, 7.73; b, 1.88; n, 2.44; test values are: c, 87.93; h, 7.75; b, 1.87; and N, 2.45. ESI-MS (M/z) (M)+): the theoretical value is 1146.71, and the actual value is 1147.14.
< example 14> preparation of Compound 350
The procedure of example 6 was repeated, except that raw material 6 was used in place of raw material 9, raw material 21 was used in place of raw material 10, intermediate 27 was prepared, bromination was carried out to give intermediate 28, and intermediate 28 and raw material 22 were used in a coupling reaction to give the objective compound 350 in a purity of 99.5% and a yield of 58.8%.
Elemental analysis Structure (molecular formula C)52H28B2N6O): theoretical value C, 80.65; h, 3.64; b, 2.79; n, 10.85; o, 2.07; test values are: c, 80.66; h, 3.65; b, 2.80; n, 10.84; o, 2.05. ESI-MS (M/z) (M)+): the theoretical value is 774.25, and the actual value is 774.77.
< example 15> preparation of Compound 396
The procedure of example 6 was repeated, except that raw material 23 was used instead of raw material 9, raw material 24 was used instead of raw material 10, intermediate 29 was prepared, bromination was carried out to give intermediate 30, and intermediate 30 and raw material 25 were used for coupling reaction to give target compound 396 with a purity of 99.5% and a yield of 56.6%.
Elemental analysis Structure (molecular formula C)50H38B2N4O): theoretical value C, 81.99; h, 5.23; b, 2.95; n, 7.65; o, 2.18; test values are: c, 82.00; h, 5.22; b, 2.94; n, 7.65; o, 2.19. ESI-MS (M/z) (M)+): the theoretical value was 732.32, and the actual value was 732.81.
< example 16> preparation of Compound 431
The procedure of example 6 was repeated, except that raw material 21 was used instead of raw material 9, raw material 26 was used instead of raw material 10, intermediate 31 was prepared, bromination was carried out to give intermediate 32, and intermediate 32 and raw material 5 were used for coupling reaction to give the objective compound 431 with a purity of 99.7% and a yield of 53.3%.
Elemental analysis Structure (molecular formula C)67H70B2N2): theoretical value C, 87.01; h, 7.63; b, 2.34; n, 3.03; test values are: c, 87.02; h, 7.62; b, 2.35; and N, 3.01. ESI-MS (M/z) (M)+): the theoretical value is 924.57, and the actual value is 925.01.
< example 17> preparation of Compound 458
The procedure of example 6 was repeated except that raw material 27 was used instead of raw material 9 and raw material 21 was used instead of raw material 10 to prepare intermediate 33, bromination was carried out to obtain intermediate 34, and intermediate 34 and raw material 5 were used for coupling reaction to obtain the objective compound 458 with a purity of 99.8% and a yield of 57.6%.
Elemental analysis Structure (molecular formula C)70H68B2N2): theoretical value C, 87.68; h, 7.15; b, 2.25; n, 2.92; test values are: c, 87.69; h, 7.14; b, 2.24; and N, 2.93. ESI-MS (M/z) (M)+): theoretical value is 958.56, found 958.95.
< example 18> preparation of Compound 499
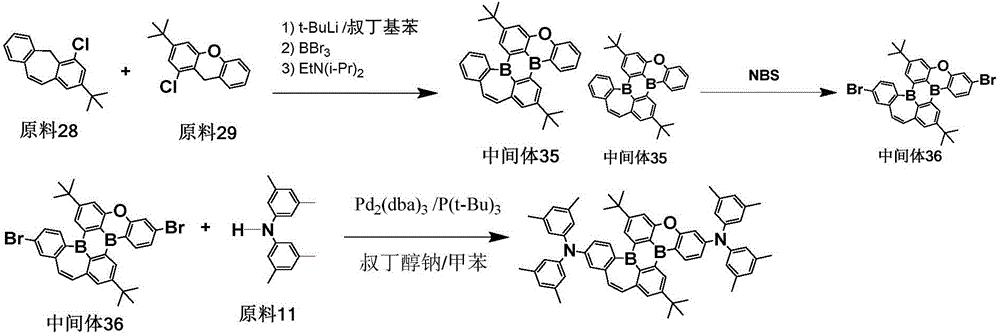
The procedure of example 6 was repeated, except that starting material 28 was used instead of starting material 9, starting material 29 was used instead of starting material 10, intermediate 35 was prepared, bromination was carried out to give intermediate 36, and intermediate 36 and starting material 11 were used for the coupling reaction to give the objective compound 499 in a purity of 99.6% with a yield of 58.1%.
Elemental analysis Structure (molecular formula C)66H66B2N2O): theoretical value C, 85.71; h, 7.19; b, 2.34; n, 3.03; o, 1.73; test values are: c, 85.69; h, 7.20; b, 2.35; n, 3.04; o, 1.72. ESI-MS (M/z) (M)+): theoretical value is 924.54, found 924.93.
< example 19> preparation of Compound 537
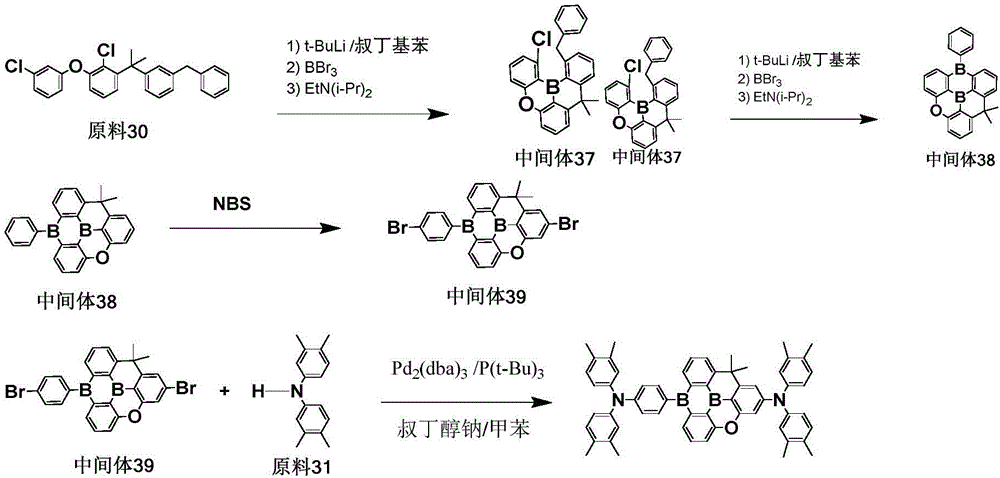
The procedure of example 3 was repeated, except that the starting material 30 was used in place of the starting material 4, to prepare an intermediate 37, then a diboron compound intermediate 38 was prepared, bromination was carried out to obtain an intermediate 39, and the intermediate 39 and the starting material 31 were subjected to coupling reaction to obtain the target compound 537 with a purity of 99.9% and a yield of 53.6%.
Elemental analysis Structure (molecular formula C)59H54B2N2O): theoretical value C, 85.51; h, 6.57; b, 2.61; n, 3.38; o, 1.93; test values are: c, 85.53; h, 6.55; b, 2.63; n, 3.37; o, 1.92. ESI-MS (M/z) (M)+): the theoretical value is 828.44, and the actual value is 828.72.
< example 20> preparation of Compound 551
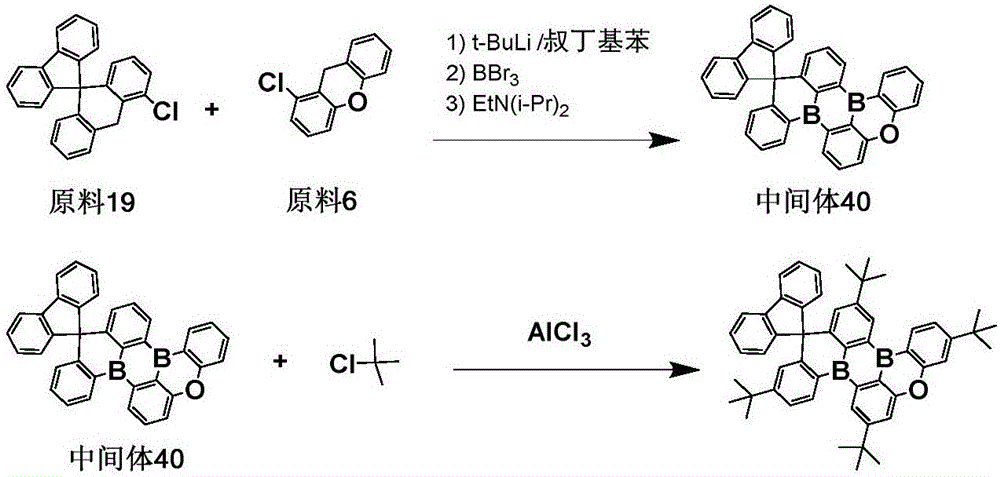
The procedure of example 1 was repeated except that the starting materials 19 and 6 were used instead of the starting material 1 to prepare an intermediate 40, which was then substituted with a tert-butyl group to obtain the objective compound 551.
Elemental analysis Structure (molecular formula C)53H54B2O): theoretical value C, 87.37; h, 7.47; b, 2.97; o, 2.20; test values are: c, 87.35; h, 7.45; b, 2.99; o, 2.21. ESI-MS (M/z) (M)+): theoretical value is 728.44, found 728.88.
< example 21> preparation of Compound 572
The procedure of example 6 was repeated, except that raw material 32 was used in place of raw material 9, raw material 33 was used in place of raw material 10, intermediate 41 was prepared, bromination was carried out to give intermediate 42, and intermediate 12 and raw material 5 were used in the coupling reaction to give the objective compound 572 in 99.1% purity and 69.6% yield.
Elemental analysis Structure (molecular formula C)58H59B2NO2): theory of the inventionThe value C, 84.57; h, 7.22; b, 2.62; n, 1.70; o, 3.88; test values are: c, 84.56; h, 7.21; b, 2.63; n, 1.71; and O, 3.89. ESI-MS (M/z) (M)+): the theoretical value is 823.47, and the actual value is 823.69.
The compound prepared by the invention can be used as a luminescent layer material, and the thermal performance, the luminescent spectrum and the cyclic voltammetry stability of the compound are firstly tested, and the test results are shown in table 1.
TABLE 1
As can be seen from the data in Table 1, the compound of the invention has higher thermal stability and smaller singlet state-triplet state energy level difference, so that the efficiency and the service life of an O L ED device using the compound of the invention as a luminescent layer host material and a doping material are improved, and the compound of the invention simultaneously has excellent cyclic voltammetry stability, which is a necessary condition for a long-life device.
GH-1 and GH-2 are used as double host materials, the compounds of the invention are used as doped luminescent materials, the mass ratio of the compounds GH-1 and GH-2 to the compounds of the invention is 45: 45:10, the organic film is prepared by co-evaporation, 365nm ultraviolet light is used for excitation, the distribution condition of the fluorescence intensity of the organic film along with the test angle is measured, the smaller the anisotropy factor α and α is measured by optical fitting software, the larger the horizontal photon-emitting component of the doped material of the organic film is, the higher the utilization rate of the radiation luminescence of the doped material is, the shorter the life of delayed fluorescence is when the organic film is used as a TADF material (thermal excitation delayed fluorescence material), the triplet state is easy to jump back to the singlet state through the back gap, so that triplet state quenching is avoided, and the efficiency and the life of the device can be improved, and the results are.
TABLE 2
| Organic film (30nm) | α (degree) | Delayed fluorescence lifetime τ |
| GH-1, GH-2, compound 15 ═ 45: 45:10 | 8.2 | 7.4 |
| GH-1, GH-2, compound 93 ═ 45: 45:10 | 10.6 | 9.8 |
| GH-1: GH-2 compound 140 ═ 45: 45:10 | 9.7 | 10.1 |
| GH-1: GH-2 compound 189 ═ 45: 45:10 | 11.3 | 14.7 |
| GH-1: GH-2 compound 321 ═ 45: 45:10 | 12.6 | 15.5 |
| GH-1: GH-2 compound 396 ═ 45: 45:10 | 13.1 | 12.8 |
| GH-1: GH-2 compound 499 ═ 45: 45:10 | 10.8 | 8.3 |
| GH-1: GH-2 compound 572 ═ 45: 45:10 | 9.9 | 8.5 |
The organic film is subjected to double-source co-evaporation through ANS evaporation equipment, the evaporation substrate is made of high-transparency quartz glass, the doping quality concentration of an object is 10%, after evaporation is finished, packaging is carried out in a glove box (the concentration of water and oxygen is less than 1ppm), a sample is placed in a fused silicon semi-cylindrical prism through refractive index matching fluid, the light emitting angle is changed through a rotating table, a Sphere Optics SMS-500 spectrometer is adopted in spectrum testing, and the delayed fluorescence life is obtained through an F L S980 transient life tester of an Edinburgh instrument.
From table 2, it can be seen that the compound of the present invention has a small anisotropy factor of the luminescent molecules, thus improving the light extraction efficiency of the organic layer and increasing the efficiency of the O L ED device.
The effect of the compound synthesized by the present invention as a doping material for a light emitting layer in a device is explained in detail by device examples 1 to 18 and device comparative example 1 below. Device examples 2-18 and device comparative example 1 compared with device example 1, the manufacturing process of the device was completely the same, and the same substrate material and electrode material were used, and the film thickness of the electrode material was also kept the same, except that the material of the light emitting layer in the device was changed. Except that the doping material of the light-emitting layer 6 in the device is changed. The structural composition of the resulting device of each example is shown in table 3. The test results of the resulting devices are shown in table 4.
< device example 1>
As shown in fig. 1, an electroluminescent device is prepared by the steps of:
a) cleaning the ITO anode layer 2 on the transparent substrate layer 1, respectively ultrasonically cleaning the ITO anode layer 2 with deionized water, acetone and ethanol for 15 minutes, and then treating the ITO anode layer 2 in a plasma cleaner for 2 minutes;
b) evaporating a hole injection layer material HAT-CN on the ITO anode layer 2 in a vacuum evaporation mode, wherein the thickness of the hole injection layer material HAT-CN is 10nm, and the hole injection layer material HAT-CN is used as a hole injection layer 3;
c) evaporating a hole transport material HT-1 with the thickness of 60nm on the hole injection layer 3 in a vacuum evaporation mode, wherein the layer is a hole transport layer 4;
d) evaporating an electron blocking material EB-1 on the hole transmission layer 4 in a vacuum evaporation mode, wherein the thickness of the electron blocking material EB-1 is 20nm, and the electron blocking layer 5 is formed on the hole transmission layer;
e) and a light-emitting layer 6 is evaporated on the electron blocking layer 5, the host material is the compounds GH-1 and GH-2 prepared in the embodiment of the invention, the doping material is the compound 11, and the mass ratio of the compounds GH-1 and GH-2 to the compound 11 is 45: 45:10, thickness of 30 nm;
f) evaporating electron transport materials ET-1 and L iq on the light-emitting layer 6 in a vacuum evaporation mode, wherein the mass ratio of the electron transport materials ET-1 to L iq is 1:1, the thickness of the electron transport materials ET-1 to L iq is 40nm, and the organic material of the layer is used as a hole blocking/electron transport layer 7;
g) an electron injection layer L iF is vacuum-evaporated on the hole blocking/electron transporting layer 7, the thickness of the electron injection layer is 1nm, and the electron injection layer is an electron injection layer 8;
h) vacuum evaporating cathode Al (100nm) on the electron injection layer 8, which is a cathode reflection electrode layer 9;
after the O L ED light emitting device was completed as described above, the anode and the cathode were connected by a known driving circuit, and the current efficiency of the device and the lifetime of the device were measured, and the test results of the fabricated O L ED light emitting device are shown in table 4.
TABLE 3
TABLE 4
Note that the life test system is an O L ED device life tester, which is commonly studied by the owner of the present invention and the university of shanghai.
From the results in table 4, it can be seen that the compound of the present invention can be used as a doping material of a light emitting layer in the fabrication of an O L ED light emitting device, and compared with comparative device 1, the compound has a greater improvement in efficiency and lifetime than the known O L ED material, and particularly, the driving lifetime of the device is greatly improved.
The effect of the compound synthesized by the present invention as a host material for a light emitting layer in a device is illustrated by device examples 19 to 23 and comparative example 2 below. Compared with the device example 1, the device examples 19-23 and the device comparative example 2 of the present invention have the same manufacturing process, and adopt the same substrate material and electrode material, the film thickness of the electrode material is also kept consistent, the difference is that the main body material in the light emitting layer 6 is different, the film thickness of the hole transport layer of the device is 100nm, the electron blocking layer is EB-2, the doping material is RD-1, and the doping concentration thereof becomes 10%. The structural composition of each device is shown in table 5. The test results of the resulting devices are shown in table 6.
TABLE 5
TABLE 6
| Numbering | Current efficiency (cd/A) | Color(s) | L T97 Life (Hr) @10mA/cm2 |
| Device example 19 | 23.5 | Red light | 345.3 |
| Device example 20 | 24.3 | Red light | 354.4 |
| Device example 21 | 25.2 | Red light | 348.3 |
| Device example 22 | 24.5 | Red light | 335.8 |
| Device example 23 | 25.1 | Red light | 339.3 |
| Device comparative example 2 | 16.5 | Red light | 301.3 |
Note that the life test system is an O L ED device life tester, which is commonly studied by the owner of the present invention and the university of shanghai.
The results in Table 6 show that the compound of the invention can be used as a doping material of a light-emitting layer for manufacturing an O L ED light-emitting device, and compared with the device in comparative example 2, the compound has the advantages that the efficiency and the service life are greatly improved compared with those of the known O L ED material, and particularly the driving service life of the device is greatly prolonged.
Through further experimental study, the efficiency of the O L ED device prepared by the material of the invention is found to be stable when the device works at low temperature or high temperature, the device examples 6, 13 and 22, the device comparative example 1 and the device comparative example 2 are subjected to efficiency test at the temperature range of-10 to 80 ℃, and the obtained results are shown in Table 7 and FIG. 2.
TABLE 7
As can be seen from the data in table 7, device examples 6, 13, and 22 are device structures in which the material of the present invention and the known material are combined, and compared with device comparative examples 1 and 2, the efficiency is high at low temperature, and the efficiency is steadily increased during the temperature increase process.
The above description is only for the purpose of illustrating the preferred embodiments of the present invention and is not to be construed as limiting the invention, and any modifications, equivalents, improvements and the like that fall within the spirit and principle of the present invention are intended to be included therein.
Claims (10)
1. An organic compound with diboron as a core is characterized in that the structure of the organic compound is shown as a general formula (1):
wherein, X1、X2、X3、X4Each independently represents a single bond, an oxygen atom, a sulfur atom,-CR5=CR6-、-C(R7R8) -or-Si (R)9R10) -one of the above;
a. b, c and d are respectively and independently 0 or 1; and a + b + c + d is 2;
Y1~Y18each independently represents a nitrogen atom or C-R11(ii) a Y at the connection site1~Y18Represented as a carbon atom;
e. f, g and h are respectively and independently 0, 1, 2, 3 or 4;
R1~R4each independently represents a hydrogen atom, a structure represented by general formula (2), general formula (3) or general formula (4);
in the general formula (2), L represents a single bond, substituted or unsubstituted C6-30One of arylene, substituted or unsubstituted 5-30 membered heteroarylene containing a heteroatom;
Ar1、Ar2each independently represents substituted or unsubstituted C6-30One of an aryl group, a substituted or unsubstituted 5-30 membered heteroaryl group containing a heteroatom; ar (Ar)1、Ar2Can also be connected with each other to form a ring;
in the general formula (4), X5、X6Each independently represents a single bond, a sulfur atom, an oxygen atom, -N (R)12)-、CR13=CR14、-C(R15R16) -or-Si (R)17R18) -one of (a); and X5、X6May not be simultaneously represented as a single bond;
the R is5~R10、R12~R18Are each independently represented by C1-20Straight chain alkyl, C3-20Branched alkyl radical, C1-20Linear alkyl substituted silane radical, C3-20Branched alkyl substituted silane group, substituted or unsubstituted C6-30One of aryl, substituted or unsubstituted 5-to 30-membered heteroaryl containing a heteroatom, and R5、R6、R13、R14May also be represented as a hydrogen atom; r13And R14、R15And R16、R17And R18May be linked to form a 5-to 30-membered aliphatic or aromatic ring, respectively;
in the general formulae (3) and (4), Z1~Z4Each independently represents a nitrogen atom or C-R19;
The R is11、R19Each independently represents a hydrogen atom, a protium atom, a deuterium atom, a tritium atom, a fluorine atom, a cyano group, a phosphoric acid or a salt thereof, or C1-20Straight chain alkyl, C2-20Alkylene radical, C3-20Branched alkyl radical, C1-20Linear alkyl substituted silane radical, C3-20Branched alkyl substituted silane group, substituted or unsubstituted C6-30One of an aryl group, a substituted or unsubstituted 5-to 30-membered heteroaryl group containing a heteroatom; wherein two or more R11、R19The groups may also be linked to each other to form a 5-to 30-membered aliphatic or aromatic ring;
general formula (3) and general formula (4) are independently through Y1-Y2、Y2-Y3、Y3-Y4、Y5-Y6、Y6-Y7、Y7-Y8、Y8-Y9、Y10-Y11、Y11-Y12、Y12-Y13、Y14-Y15、Y15-Y16、Y16-Y17Or Y17-Y18Is connected with a parallel ring of the general formula (1);
said substitutable groupThe substituents of the radicals being optionally selected from deuterium, cyano, halogen, C1-20Alkyl, protium atom, deuterium atom, tritium atom, C6-30One or more of aryl or 5 to 30 membered heteroaryl containing a heteroatom;
the hetero atom of the heteroaryl is one or more selected from oxygen atom, sulfur atom or nitrogen atom.
3. the organic compound according to claim 1, wherein the organic compound has a structure represented by any one of general formula (I-1) to general formula (I-133):
z is nitrogen atom or C-R20;R20Represented by hydrogen atom, protium atom, deuterium atom, tritium atom, fluorine atom, cyano group, phosphoric acid or a salt thereof, C1-20Straight chain alkyl, C2-20Alkylene radical, C3-20Branched alkyl radical, C1-20Linear alkyl substituted silane radical, C3-20Branched alkyl substituted silane group, substituted or unsubstituted C6-30One of an aryl group, a substituted or unsubstituted 5-to 30-membered heteroaryl group containing a heteroatom; wherein two or more R20The groups may also be linked to each other to form a 5-to 30-membered aliphatic or aromatic ring;
other symbols have the meanings defined in claim 1;
the substituent of the substitutable group is selected from cyano, halogen and C1-20Alkyl, protium atom, deuterium atom, tritium atom, C6-30One or more of aryl or 5 to 30 membered heteroaryl containing a heteroatom;
the hetero atom of the heteroaryl is one or more selected from oxygen atom, sulfur atom or nitrogen atom.
4. The organic compound according to claim 3, wherein L represents one of a single bond, a substituted or unsubstituted phenylene group, a substituted or unsubstituted biphenylene group, a substituted or unsubstituted naphthylene group, a substituted or unsubstituted anthrylene group, a substituted or unsubstituted pyridylene group, a substituted or unsubstituted pyrimidylene group, a substituted or unsubstituted pyridazylene group, a substituted or unsubstituted furanylene group, a substituted or unsubstituted thiophenylene group, a substituted or unsubstituted dibenzofuranylene group, a substituted or unsubstituted dibenzothiophenylene group, and a substituted or unsubstituted carbazolyl group;
Ar1、Ar2each independently represents a substituted or unsubstituted phenyl group, a substituted or unsubstituted biphenylyl group, a substituted or unsubstituted terphenylyl group, a substituted or unsubstituted naphthyl group, a substituted or unsubstituted naphthyridinyl group, a substituted or unsubstituted anthracenyl group, a substituted or unsubstituted phenanthrenyl group, a substituted or unsubstituted pyrenyl group, a substituted or unsubstituted benzophenanthrenyl group, a substituted or unsubstituted azabenzophenanthrenyl group, a substituted or unsubstituted pyridyl group, one of substituted or unsubstituted pyrimidinyl, substituted or unsubstituted pyridazinyl, substituted or substituted triazinyl, substituted or unsubstituted furan, substituted or unsubstituted thiophene, substituted or unsubstituted dibenzofuranyl, substituted or unsubstituted dibenzothiophenyl, substituted or unsubstituted carbazolyl, substituted or unsubstituted naphthocarbazolyl, substituted or unsubstituted naphthofuranyl, substituted or unsubstituted azacarbazolyl;
the R is5~R10、R12~R18Each independently represents methyl, ethyl, propyl, isopropyl, butyl, tert-butyl, pentyl, phenyl, naphthyl, biphenyl, terphenyl, naphthyridinyl or pyridyl;
the R is11、R19、R20Each independently represents a hydrogen atom, a protium atom, a deuterium atom, a tritium atom, a fluorine atom, a cyano group, a methyl group, an ethyl group, a propyl group, an isopropyl group, a butyl group, a tert-butyl group, a pentyl group, a substituted or unsubstituted phenyl group, a substituted or unsubstituted biphenyl group, a substituted or unsubstituted naphthyl group, a substituted or unsubstituted anthracenyl group, a substituted or unsubstituted pyridyl group, a substituted or unsubstituted pyrimidyl group, a substituted or unsubstituted pyridazinyl group, a substituted or substituted triazinyl group, a substituted or unsubstituted furyl group, a substituted or unsubstituted thienyl group, a substituted or unsubstituted dibenzofuranyl group, a substituted or unsubstituted dibenzothienyl group, a substituted or unsubstituted carbazolyl group;
the substituent of the substitutable group is one or more selected from deuterium, cyano, methyl, ethyl, propyl, isopropyl, butyl, tertiary butyl, pentyl, cyano, fluorine atom, phenyl, naphthyl, biphenyl, terphenyl, naphthyridinyl or pyridyl.
5. The organic compound of claim 1, wherein R is1~R4At least one of the structures is represented by a general formula (2), a general formula (3) or a general formula (4).
7. A method for producing an organic compound according to any one of claims 1 to 6, characterized by comprising the following three cases:
A. in the general formula (1), when a and b are 1, and c and d are 0, the synthesis steps are as follows:
the specific reaction process of the above reaction equation is:
1) under the protection of inert gas, dissolving a raw material B by using tert-butyl benzene, wherein the proportion of the raw material B to the tert-butyl benzene is that every 1g of the raw material B is dissolved by using 50-100 m L tert-butyl benzene;
2) slowly adding tert-butyl lithium into the reaction system in the step 1), and stirring the mixed solution; wherein the molar ratio of the tert-butyl lithium to the raw material B is 1.0-2.0: 1;
3) reacting the mixed solution obtained in the step 2) for 2-4 h at the temperature of 50-70 DEG CNaturally cooling to room temperature, and then slowly dripping BBr3And diisopropylethylamine, and stirring and reacting for 1-4 hours at room temperature; the BBr3The molar ratio of the diisopropylethylamine to the raw material B is 1.0-2.0: 1, and the molar ratio of the diisopropylethylamine to the raw material B is 2.0-3.0: 1;
4) after the reaction is finished, adding water and dichloromethane for extraction and liquid separation, taking an organic phase, adding anhydrous magnesium sulfate for dewatering, filtering, carrying out rotary evaporation on the filtrate until no solvent exists, and passing through a neutral silica gel column to obtain an intermediate B;
5) under the protection of inert gas, dissolving an intermediate B by using tert-butyl benzene, wherein the ratio of the intermediate A to the tert-butyl benzene is that every 1g of the intermediate B is dissolved by using 50-100 m L tert-butyl benzene;
6) slowly adding tert-butyl lithium into the reaction system in the step 5), and stirring the mixed solution; wherein the molar ratio of the tert-butyl lithium to the intermediate B is 1.0-2.0: 1;
7) reacting the mixed solution obtained in the step 6) for 2-4 hours at the temperature of 50-70 ℃, naturally cooling to room temperature, and then slowly dropwise adding BBr3And diisopropylethylamine, and stirring and reacting for 1-4 hours at room temperature; the BBr3The molar ratio of the diisopropylethylamine to the intermediate B is 1.0-2.0: 1, and the molar ratio of the diisopropylethylamine to the intermediate B is 2.0-3.0: 1;
8) after the reaction is finished, adding water and dichloromethane for extraction and liquid separation, taking an organic phase, adding anhydrous magnesium sulfate for dewatering, filtering, carrying out rotary evaporation on the filtrate until no solvent exists, and passing through a neutral silica gel column to obtain a target compound;
B. in the general formula (1), when a and c are 1 and b and d are 0, the synthesis steps are as follows:
the specific reaction process of the above reaction equation is:
1) under the protection of inert gas, dissolving a raw material C-I and a raw material C-II by using tert-butyl benzene, wherein the proportion of the raw material C-I to the tert-butyl benzene is that every 1g of the raw material C-I is dissolved by using 30-80 m L m of tert-butyl benzene, and the molar ratio of the raw material C-I to the raw material C-II is 1: 1;
2) slowly adding tert-butyl lithium into the reaction system in the step 1), and stirring the mixed solution; wherein the molar ratio of the tert-butyl lithium to the raw material C-I is 2.0-3.0: 1;
3) reacting the mixed solution obtained in the step 2) for 2-4 hours at the temperature of 50-70 ℃, naturally cooling to room temperature, and then slowly dropwise adding BBr3And diisopropylethylamine, and stirring and reacting for 1-4 hours at room temperature; the BBr3The molar ratio of the diisopropylethylamine to the raw material C-I is 2.0-3.0: 1, and the molar ratio of the diisopropylethylamine to the raw material C-I is 3.0-6.0: 1;
4) after the reaction is finished, adding water and dichloromethane for extraction and liquid separation, taking an organic phase, adding anhydrous magnesium sulfate for dewatering, filtering, carrying out rotary evaporation on the filtrate until no solvent exists, and passing through a neutral silica gel column to obtain a target compound;
C. in the general formula (1), when a and d are 1 and b and c are 0, the synthesis steps are as follows:
the specific reaction process of the above reaction equation is:
1) under the protection of inert gas, dissolving a raw material D-I and a raw material D-II by using tert-butyl benzene, wherein the proportion of the raw material D-I to the tert-butyl benzene is that every 1g of the raw material D-I is dissolved by using 30-80 m L m of tert-butyl benzene, and the molar ratio of the raw material D-I to the raw material D-II is 1: 1;
2) slowly adding tert-butyl lithium into the reaction system in the step 1), and stirring the mixed solution; wherein the molar ratio of the tert-butyl lithium to the raw material D-I is 2.0-3.0: 1;
3) reacting the mixed solution obtained in the step 2) for 2-4 hours at the temperature of 50-70 ℃, naturally cooling to room temperature, and then slowly dropwise adding BBr3And diisopropylethylamine, and stirring and reacting for 1-4 hours at room temperature; the BBr3The molar ratio of the diisopropylethylamine to the raw material D-I is 2.0-3.0: 1, and the molar ratio of the diisopropylethylamine to the raw material D-I is 3.0-6.0: 1;
4) and after the reaction is finished, adding water and dichloromethane for extraction and liquid separation, taking an organic phase, adding anhydrous magnesium sulfate for dewatering, filtering, performing rotary evaporation on the filtrate until no solvent exists, and passing through a neutral silica gel column to obtain the target compound.
8. An organic electroluminescent device prepared from the organic compound with a diboron core as defined in any one of claims 1 to 6.
9. An organic electroluminescent device, characterized in that the material of the light-emitting layer of the organic electroluminescent device is the organic compound with the core of diboron according to any one of claims 1 to 6.
10. A lighting or display element, characterized in that the element comprises an organic electroluminescent device as claimed in any one of claims 8 to 9.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811635458.9A CN111440203A (en) | 2018-12-29 | 2018-12-29 | Organic compound with diboron as core and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811635458.9A CN111440203A (en) | 2018-12-29 | 2018-12-29 | Organic compound with diboron as core and application thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN111440203A true CN111440203A (en) | 2020-07-24 |
Family
ID=71626523
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201811635458.9A Withdrawn CN111440203A (en) | 2018-12-29 | 2018-12-29 | Organic compound with diboron as core and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111440203A (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111574543A (en) * | 2019-02-18 | 2020-08-25 | 江苏三月科技股份有限公司 | Organic compound containing boron and application thereof in organic electroluminescent device |
| CN111574544A (en) * | 2019-02-18 | 2020-08-25 | 江苏三月科技股份有限公司 | Organic compound containing boron and application thereof in organic electroluminescent device |
| CN114641482A (en) * | 2019-11-04 | 2022-06-17 | 默克专利有限公司 | Material for organic electroluminescent device |
| CN114957306A (en) * | 2022-06-17 | 2022-08-30 | 京东方科技集团股份有限公司 | Thermal activation delayed fluorescent material and electroluminescent device |
-
2018
- 2018-12-29 CN CN201811635458.9A patent/CN111440203A/en not_active Withdrawn
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111574543A (en) * | 2019-02-18 | 2020-08-25 | 江苏三月科技股份有限公司 | Organic compound containing boron and application thereof in organic electroluminescent device |
| CN111574544A (en) * | 2019-02-18 | 2020-08-25 | 江苏三月科技股份有限公司 | Organic compound containing boron and application thereof in organic electroluminescent device |
| CN114641482A (en) * | 2019-11-04 | 2022-06-17 | 默克专利有限公司 | Material for organic electroluminescent device |
| CN114957306A (en) * | 2022-06-17 | 2022-08-30 | 京东方科技集团股份有限公司 | Thermal activation delayed fluorescent material and electroluminescent device |
| WO2023241293A1 (en) * | 2022-06-17 | 2023-12-21 | 京东方科技集团股份有限公司 | Thermally activated delayed fluorescent material and electroluminescent device |
| CN114957306B (en) * | 2022-06-17 | 2024-02-09 | 京东方科技集团股份有限公司 | Thermal activation delay fluorescent material and electroluminescent device |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN111574544A (en) | Organic compound containing boron and application thereof in organic electroluminescent device | |
| CN111574543A (en) | Organic compound containing boron and application thereof in organic electroluminescent device | |
| CN110964021A (en) | A kind of compound with fluorene as the core, preparation method and application thereof | |
| CN110498765B (en) | Compound, organic electroluminescent material and organic electroluminescent device | |
| CN113072568B (en) | Organic electroluminescent material containing diboron and application thereof | |
| CN107586261B (en) | Organic compound containing spiro dibenzosuberene fluorene and application thereof | |
| CN111377956A (en) | Organic compound with double boron as core, preparation method thereof and application thereof in organic electroluminescent device | |
| CN110903294B (en) | Compound with benzo [1,2-b:5, 4-b' ] dibenzofuran as core and application thereof | |
| CN110272427B (en) | Compound with fluorene as core, preparation method thereof and application thereof in organic electroluminescent device | |
| CN110903276A (en) | Organic compound and organic electroluminescent device | |
| CN114075229A (en) | A boron-containing organic compound and its application in organic electroluminescent devices | |
| CN114276337A (en) | TADF material and organic electroluminescent device comprising same | |
| CN111440203A (en) | Organic compound with diboron as core and application thereof | |
| CN111662190A (en) | Organic compound containing pyrene or aza-pyrene and application thereof | |
| CN107868048A (en) | A kind of compound containing fluorenes and its application | |
| CN111440202A (en) | Organic compound with diboron as core and application thereof in O L ED | |
| CN110526905B (en) | Ketone-containing compound and application thereof in organic electroluminescent device | |
| CN110642732B (en) | Organic compound containing spirofluorene anthrone structure and application thereof | |
| CN110655504B (en) | Compound with dibenzosuberene as core and application thereof in organic electroluminescent device | |
| CN109912592B (en) | Compound containing pyridoindole and application of compound in organic electroluminescent device | |
| CN111362810B (en) | Compound containing dibenzosuberene and application thereof in organic electroluminescent device | |
| JP2025515662A (en) | Organic light-emitting compound and organic electroluminescent device containing the same | |
| CN109796450B (en) | Compound with pyridoindole as core and application thereof in electroluminescent device | |
| CN110963989A (en) | Compound with xanthene as core and application thereof | |
| CN109574908B (en) | Compound containing spirodimethyl anthracene fluorene and application thereof in organic electroluminescent device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| WW01 | Invention patent application withdrawn after publication |
Application publication date: 20200724 |
|
| WW01 | Invention patent application withdrawn after publication |