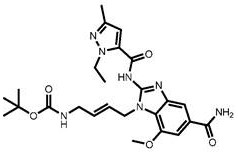
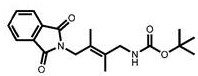
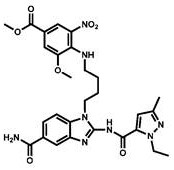
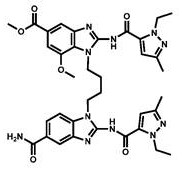
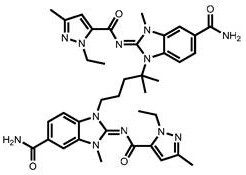
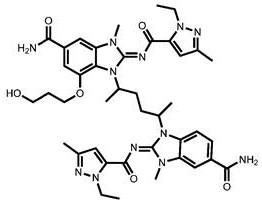
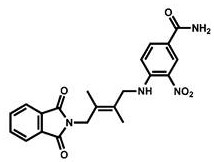
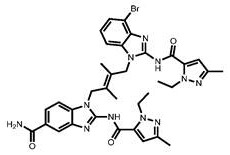
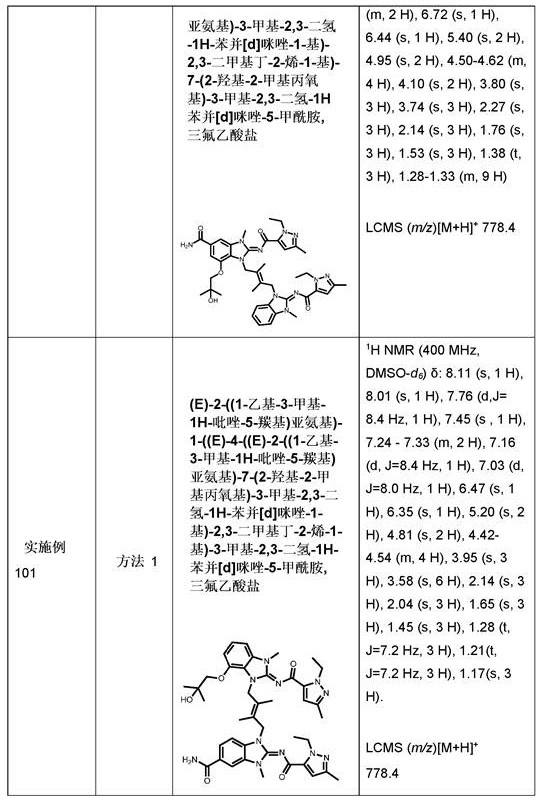
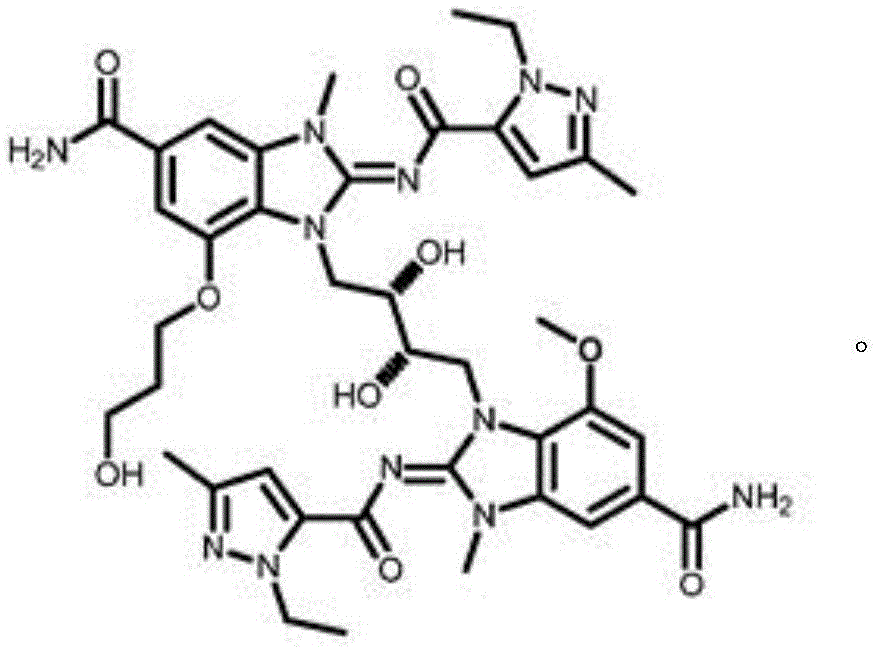
CN111417630B - Modulator of Stimulator of Interferon Genes (STING) - Google Patents
Modulator of Stimulator of Interferon Genes (STING) Download PDFInfo
- Publication number
- CN111417630B CN111417630B CN201880078932.4A CN201880078932A CN111417630B CN 111417630 B CN111417630 B CN 111417630B CN 201880078932 A CN201880078932 A CN 201880078932A CN 111417630 B CN111417630 B CN 111417630B
- Authority
- CN
- China
- Prior art keywords
- alkyl
- optionally substituted
- methyl
- independently
- substituted
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing three or more hetero rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
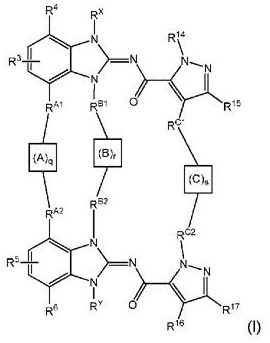
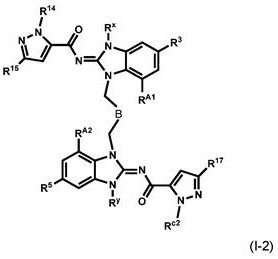
本申请公开具有下式(I)的化合物:其中q、r、s、A、B、C、RA1、RA2、RB1、RB2、RC1、RC2、R3、R4、R5、R6、R14、R15、R16、R17、Rx和Ry如本文中所定义,或其互变异构体、或其盐,特别是药学上可接受的盐。 The present application discloses compounds having the following formula (I): wherein q, r, s, A, B, C, R A1 , R A2 , R B1 , R B2 , R C1 , R C2 , R 3 , R 4 , R 5 , R 6 , R 14 , R 15 , R 16 , R 17 , R x and R y are as defined herein, or tautomers thereof, or salts thereof, especially pharmaceutically acceptable salts thereof.
Description
相关申请Related Applications
本申请要求2017年10月5日提交的美国临时申请No. 62/568,420的优先权,所述申请的完整内容通过引用并入本文。This application claims priority to U.S. Provisional Application No. 62/568,420, filed on October 5, 2017, the entire contents of which are incorporated herein by reference.
发明领域Field of the Invention
本发明涉及可用作跨膜蛋白173((TMEM173),其也称为STING(干扰素基因刺激因子))的调节剂的杂环酰胺类及其制备和使用方法。The present invention relates to heterocyclic amides useful as modulators of transmembrane protein 173 ((TMEM173), also known as STING (stimulator of interferon genes)) and methods of making and using the same.
发明背景Background of the Invention
脊椎动物不断受到微生物入侵的威胁,并已进化了免疫防御机制以消除感染性病原体。在哺乳动物中,该免疫系统包含两个分支;先天性免疫和适应性免疫。先天性免疫系统是由模式识别受体(PRR)引发的第一道防线,其检测来自病原体的配体以及损伤相关分子模式(Takeuchi O.等人,Cell,2010:140,805-820)。已经鉴定了越来越多的这些受体,包括Toll样受体(TLR)、C型凝集素受体、视黄酸诱导基因I(RIG-I)样受体和NOD样受体(NLR)以及双链DNA传感器。PRR的活化导致参与炎性应答的基因(包括抑制病原体复制和促进适应性免疫的1型干扰素、促炎细胞因子和趋化因子)的上调。Vertebrates are constantly threatened by microbial invasion and have evolved immune defense mechanisms to eliminate infectious pathogens. In mammals, the immune system consists of two branches; innate immunity and adaptive immunity. The innate immune system is the first line of defense triggered by pattern recognition receptors (PRRs), which detect ligands from pathogens as well as damage-associated molecular patterns ( Takeuchi O. et al., Cell, 2010: 140, 805-820 ). More and more of these receptors have been identified, including Toll-like receptors (TLRs), C-type lectin receptors, retinoic acid-induced gene I (RIG-I)-like receptors and NOD-like receptors (NLRs) and double-stranded DNA sensors. Activation of PRRs leads to upregulation of genes involved in inflammatory responses, including type 1 interferons, proinflammatory cytokines and chemokines that inhibit pathogen replication and promote adaptive immunity.
衔接蛋白STING(干扰素基因刺激因子),也称为TMEM 173、MPYS、MITA和ERIS,已被鉴定为对胞质核酸的先天性免疫应答中的中心信号转导分子(Ishikawa H和Barber G N, Nature,2008:455,674-678;WO2013/1666000)。STING的活化引起IRF3和NFκB途径的上调,从而导致干扰素-β和其它细胞因子的诱导。STING对病原体或宿主来源的胞质DNA以及称为环状二核苷酸(CDN)的不寻常的核酸的应答是至关重要的。The adaptor protein STING (stimulator of interferon genes), also known as TMEM 173, MPYS, MITA and ERIS, has been identified as a central signal transduction molecule in the innate immune response to cytoplasmic nucleic acids ( Ishikawa H and Barber GN, Nature, 2008: 455, 674-678; WO2013/1666000). Activation of STING causes upregulation of the IRF3 and NFκB pathways, leading to the induction of interferon-β and other cytokines. STING is critical for responding to cytoplasmic DNA of pathogen or host origin and to unusual nucleic acids called cyclic dinucleotides (CDNs).
CDN首先被鉴定为负责控制原核细胞中的许多应答的细菌第二信使。细菌CDN,诸如c-二-GMP是特征在于两个3’,5’磷酸二酯键的对称分子。CDNs were first identified as bacterial second messengers responsible for controlling many responses in prokaryotic cells. Bacterial CDNs, such as c-di-GMP, are symmetrical molecules characterized by two 3',5' phosphodiester bonds.
通过细菌CDN直接活化STING最近已通过X射线晶体学进行了证实(Burdette D L 和Vance R E,Nature Immunology,2013:14,19-26)。因此,细菌CDN及其类似物已作为潜在的疫苗佐剂吸引了兴趣(Libanova R.等人,Microbial Biotechnology 2012:5,168-176;WO2007/054279,WO2005/087238)。Direct activation of STING by bacterial CDN has recently been confirmed by X-ray crystallography ( Burdette DL and Vance RE, Nature Immunology, 2013: 14, 19-26 ). Therefore, bacterial CDN and its analogs have attracted interest as potential vaccine adjuvants ( Libanova R. et al., Microbial Biotechnology 2012: 5, 168-176; WO2007/054279, WO2005/087238).
最近,已经阐明了对胞质DNA的应答,并且显示通过称为环状GMP-AMP合成酶(cGAS,以前称为C6orf150或MB21D1)的酶参与产生新型哺乳动物CDN信号转导分子,其被称为cGAMP,然后其活化STING。与细菌CDN不同,cGAMP是不对称分子,其特征在于它的混合的2’,5’和3’,5’磷酸二酯键。(Gao P等人, Cell, 2013: 153, 1094-1107)。cGAMP(II)与STING的相互作用也已通过X射线晶体学证明(Cai X等人,Molecular Cell,2014:54,289- 296)。Recently, the response to cytoplasmic DNA has been elucidated and shown to be involved in the production of a novel mammalian CDN signaling molecule, called cGAMP, through an enzyme called cyclic GMP-AMP synthase (cGAS, formerly known as C6orf150 or MB21D1), which then activates STING. Unlike bacterial CDNs, cGAMP is an asymmetric molecule characterized by its mixed 2', 5' and 3', 5' phosphodiester bonds. (Gao P et al., Cell, 2013: 153, 1094-1107) . The interaction of cGAMP (II) with STING has also been demonstrated by X-ray crystallography ( Cai X et al., Molecular Cell, 2014: 54, 289-296 ) .
干扰素首先被描述为可以保护细胞免受病毒感染的物质(Isaacs & Lindemann, J. Virus Interference. Proc. R. Soc. Lon. Ser. B. Biol. Sci. 1957:147,258- 267)。在人中,I型干扰素是一种相关蛋白家族,其由在9号染色体上并且编码干扰素α(IFNα)的至少13种同种型和干扰素β(IFNβ)的一种同种型的基因编码。重组IFNα是第一个获批准的生物治疗剂,并且已经成为病毒感染和癌症的重要疗法。除了对细胞的直接抗病毒活性外,已知干扰素是作用于免疫系统细胞的免疫应答的有效调节剂。Interferon was first described as a substance that can protect cells from viral infection ( Isaacs & Lindemann, J. Virus Interference. Proc. R. Soc. Lon. Ser. B. Biol. Sci. 1957: 147, 258-267 ) . In humans, type I interferon is a family of related proteins encoded by genes on chromosome 9 that encode at least 13 isoforms of interferon α (IFNα) and one isoform of interferon β (IFNβ). Recombinant IFNα is the first approved biological therapeutic agent and has become an important therapy for viral infection and cancer. In addition to its direct antiviral activity on cells, interferon is known to be an effective regulator of immune responses acting on cells of the immune system.
施用可调节先天性免疫应答(包括活化或抑制I型干扰素产生和其它细胞因子)的小分子化合物,可成为治疗或预防人疾病(包括病毒感染和自身免疫性疾病)的重要策略。这种类型的免疫调节策略具有鉴定化合物的潜能,所述化合物不仅可用于感染性疾病先天免疫,而且可用于癌症(Zitvogel, L., 等人, Nature Reviews Immunology, 2015 15 (7), p405-414)、过敏性疾病(Moisan J. 等人, Am. J. Physiol. Lung Cell Mol. Physiol., 2006: 290, L987-995)、神经变性疾病诸如肌萎缩侧索硬化症和多发性硬化症(Lemos, H. 等人, J. Immunol., 2014: 192(12), 5571-8; Cirulli, E. 等人, Science, 2015: 347(6229), 1436-41; Freischmidt, A., 等人, Nat. Neurosci., 18 (5), 631-6)、其它炎性病症诸如肠易激疾病(Rakoff-Nahoum S.,Cell.,2004,23,118(2): 229-41),以及作为疫苗佐剂(Persing 等人 Trends Microbiol. 2002: 10(10 Suppl), S32-7和Dubensky 等人, Therapeutic Advances in Vaccines, 于2013年9月5日在线发 表)。Administration of small molecule compounds that can modulate the innate immune response, including activation or inhibition of type I interferon production and other cytokines, could become an important strategy for treating or preventing human diseases, including viral infections and autoimmune diseases. This type of immunomodulatory strategy has the potential to identify compounds that are useful not only for innate immunity to infectious diseases, but also for cancer (Zitvogel, L., et al., Nature Reviews Immunology, 2015 15 (7), p405-414) , allergic diseases (Moisan J. et al., Am. J. Physiol. Lung Cell Mol. Physiol., 2006: 290, L987-995) , and neurodegenerative diseases such as amyotrophic lateral sclerosis and multiple sclerosis ( Lemos, H. et al., J. Immunol., 2014: 192(12), 5571-8; Cirulli, E. et al., Science, 2015: 347(6229), 1436-41; Freischmidt, A., et al., Nat. Neurosci., 18 (5), 631-6). , other inflammatory conditions such as irritable bowel disease ( Rakoff-Nahoum S., Cell., 2004, 23, 118(2): 229-41 ), and as a vaccine adjuvant ( Persing et al. Trends Microbiol. 2002: 10(10 Suppl), S32-7 and Dubensky et al., Therapeutic Advances in Vaccines, published online on September 5, 2013 ).
STING对于抗微生物宿主防御,包括针对一系列DNA和RNA病毒和细菌的保护至关重要(综述于Barber等人. Nat. Rev. Immunol.2015: 15(2): 87-103, Ma和Damania, Cell Host & Microbe, 2016: 19(2) 150-158)。疱疹病毒科、黄病毒科、冠状病毒科、乳头瘤病毒科、腺病毒科、嗜肝病毒科、邻-和副粘病毒科和弹状病毒科已经进化出抑制STING介导的I型干扰素产生和逃避宿主免疫控制的机制(Holm等人, Nat Comm. 2016: 7:10680; Ma等人, PNAS 2015: 112(31) E4306-E4315;Wu等人, Cell Host Microbe 2015: 18(3) 333-44; Liu等人, J Virol 2016: 90(20) 9406-19; Chen等人, Protein Cell 2014: 5 (5) 369-81; Lau等人, Science 2013: 350(6260) 568-71; Ding等人, J Hepatol 2013: 59(1) 52-8; Nitta等人, Hepatology 2013 57(1) 46-58; Sun等人, PloS One 2012: 7(2) e30802;Aguirre等人, PloS Pathog 2012: 8(10) e1002934;Ishikawa等人, Nature 2009: 461(7265) 788-92)。因此,STING的小分子活化可能有益于这些感染性疾病的治疗。STING is essential for antimicrobial host defense, including protection against a range of DNA and RNA viruses and bacteria (reviewed in Barber et al. Nat. Rev. Immunol. 2015: 15(2): 87-103, Ma and Damania, Cell Host & Microbe, 2016: 19(2) 150-158 ). Herpesviridae, Flaviviridae, Coronaviridae, Papillomaviridae, Adenoviridae, Hepadnaviridae, Ortho- and Paramyxoviridae, and Rhabdoviridae have evolved mechanisms to inhibit STING-mediated type I IFN production and evade host immune control ( Holm et al., Nat Comm. 2016: 7:10680; Ma et al. , PNAS 2015: 112(31) E4306-E4315; Wu et al., Cell Host Microbe 2015: 18(3) 333-44; Liu et al., J Virol 2016: 90(20) 9406-19; Chen et al., Protein Cell 2014: 5 (5) 369-81; Lau et al., Science 2013: 350(6260) 568-71; Ding et al., J Hepatol 2013: 59(1) 52-8; Nitta et al., Hepatology 2013 57(1) 46-58; Sun et al., PloS One 2012: 7(2) e30802; Aguirre et al., PloS Pathog 2012: 8(10) e1002934; Ishikawa et al., Nature 2009: 461(7265) 788-92 ). Therefore, small molecule activation of STING may be beneficial for the treatment of these infectious diseases.
相反,增加和延长的I型IFN产生与各种慢性感染相关,所述慢性感染包括分枝杆菌(Collins等人, Cell Host Microbe 2015: 17(6) 820-8); Wassermann等人, Cell Host Microbe 2015: 17(6) 799-810; Watson等人, Cell Host Microbe 2015: 17(6) 811-9)、弗朗西斯氏菌属(Storek等人, J Immunol. 2015: 194(7) 3236-45; Jin等人, J Immunol. 2011: 187(5) 2595-601)、衣原体属(Prantner等人, J Immunol 2010: 184(5) 2551-60)、疟原虫属(Sharma等人, Immunity 2011: 35(2) 194-207) 和HIV (Herzner等 人, Nat Immunol 2015 16(10) 1025-33; Gao等人, Science 2013: 341(6148) 903-6)。类似地,在具有复杂形式的自身免疫性疾病的患者中发现过量的I型干扰素产生。人类中的遗传学证据和来自动物模型研究的支持支持了抑制STING导致驱动自身免疫性疾病的I型干扰素减少的假设(Crow YJ,等人, Nat. Genet. 2006; 38(8) 38917-920, Stetson DB, 等人, Cell 2008; 134 587-598)。因此,STING的抑制剂为具有与感染或复杂自身免疫性疾病相关的慢性I型干扰素和促炎性细胞因子生成的患者提供治疗。过敏性疾病与对过敏原的Th2偏好的免疫应答有关。Th2应答与升高的IgE水平相关,其经由对肥大细胞的作用促进对过敏原的超敏反应,导致例如在过敏性鼻炎和哮喘中见到的症状。在健康个体中,对过敏原的免疫应答与混合的Th2/Th1和调节性T细胞应答更加平衡。已显示1型干扰素的诱导导致局部环境中Th2型细胞因子的减少并促进Th1/Treg应答。在该上下文中,通过例如STING的活化诱导1型干扰素可以在过敏性疾病诸如哮喘和过敏性鼻炎的治疗中提供益处(Huber J.P.等人,J Immunol 2010:185,813-817)。In contrast, increased and prolonged type I IFN production has been associated with a variety of chronic infections, including mycobacteria (Collins et al., Cell Host Microbe 2015: 17(6) 820-8); Wassermann et al., Cell Host Microbe 2015: 17(6) 799-810; Watson et al., Cell Host Microbe 2015: 17(6) 811-9 ), Francisella ( Storek et al., J Immunol. 2015: 194(7) 3236-45; Jin et al., J Immunol. 2011: 187(5) 2595-601 ), Chlamydia ( Prantner et al., J Immunol 2010: 184(5) 2551-60 ), Plasmodium ( Sharma et al., Immunity 2011: 35(2)). 194-207 ) and HIV ( Herzner et al., Nat Immunol 2015 16 (10) 1025-33; Gao et al., Science 2013: 341 (6148) 903-6) . Similarly, excessive type I interferon production has been found in patients with complex forms of autoimmune diseases. Genetic evidence in humans and support from animal model studies support the hypothesis that inhibition of STING leads to a reduction in type I interferon that drives autoimmune diseases ( Crow YJ, et al. , Nat. Genet. 2006; 38 (8) 38917-920 , Stetson DB, et al., Cell 2008; 134 587-598 ). Therefore, inhibitors of STING provide treatment for patients with chronic type I interferon and proinflammatory cytokine production associated with infection or complex autoimmune diseases. Allergic diseases are associated with a Th2-biased immune response to allergens. Th2 responses are associated with elevated IgE levels, which promote hypersensitivity to allergens via actions on mast cells, leading to symptoms such as those seen in allergic rhinitis and asthma. In healthy individuals, the immune response to allergens is more balanced with a mixed Th2/Th1 and regulatory T cell response. Induction of type 1 interferons has been shown to result in a reduction in Th2-type cytokines in the local environment and promote Th1/Treg responses. In this context, induction of type 1 interferons by, for example, activation of STING may provide benefits in the treatment of allergic diseases such as asthma and allergic rhinitis ( Huber JP et al., J Immunol 2010: 185, 813-817).
已显示结合STING且充当激动剂的化合物在与人PBMC温育时诱导1型干扰素和其它细胞因子。诱导人干扰素的化合物可用于治疗各种病症,例如治疗过敏性疾病和其它炎性病症例如过敏性鼻炎和哮喘,治疗感染性疾病、神经变性疾病、癌前综合征和癌症,并且还可用作免疫原性组合物或疫苗佐剂。近期研究表明结合STING的化合物可以充当拮抗剂并能够用于治疗炎症,例如自身免疫性疾病、代谢疾病、神经炎症和导致心脏病(例如心肌梗死)的心脏中的炎症(Ridker等人, N ENG J Med 2017, 377 (12), 1119-1131; King 等人, Nat Med. 2017 Dec;23(12):1481-1487)。Compounds that bind to STING and act as agonists have been shown to induce type 1 interferon and other cytokines when incubated with human PBMCs. Compounds that induce human interferon can be used to treat various conditions, such as allergic diseases and other inflammatory conditions such as allergic rhinitis and asthma, infectious diseases, neurodegenerative diseases, precancerous syndromes and cancer, and can also be used as immunogenic compositions or vaccine adjuvants. Recent studies have shown that compounds that bind to STING can act as antagonists and can be used to treat inflammation, such as autoimmune diseases, metabolic diseases, neuroinflammation, and inflammation in the heart that leads to heart disease (e.g., myocardial infarction) ( Ridker et al., N ENG J Med 2017, 377 (12), 1119-1131 ; King et al., Nat Med. 2017 Dec; 23 (12): 1481-1487 ).
基于近期研究,据信抑制cGas或STING可能用于治疗或预防代谢疾病(诸如胰岛素抗性、非酒精性脂肪肝病 (NAFLD)/ 非酒精性脂肪肝炎 (NASH)、肥胖症、糖尿病、高血压、脂肪肝和心血管疾病 (Qiao等人, Metabolism Clinical and Experimental (2007), 81, 13 – 24; Bai等人, PNAS (2017), 114, no. 46, 12196-12201; Iracheta等人, Journal of Biological Chemistry (2016) 52, 26794-26805; Cruz.等人, Molecular Metabolism (2018) 1-11; Patrasek等人, Proc Natl Acad Sci (2013),110(41): 16544-9; Mao等人, Arterioscler Thromb Vasc Biol. (2017) 37(5): 920-929)。Based on recent studies, it is believed that inhibition of cGas or STING may be useful for treating or preventing metabolic diseases such as insulin resistance, non-alcoholic fatty liver disease (NAFLD)/non-alcoholic steatohepatitis (NASH), obesity, diabetes, hypertension, fatty liver, and cardiovascular disease ( Qiao et al., Metabolism Clinical and Experimental (2007), 81, 13 – 24; Bai et al., PNAS (2017), 114, no. 46, 12196-12201; Iracheta et al., Journal of Biological Chemistry (2016) 52, 26794-26805; Cruz et al., Molecular Metabolism (2018) 1-11; Patrasek et al., Proc Natl Acad Sci (2013), 110(41): 16544-9; Mao et al., Arterioscler Thromb Vasc Biol. (2017) 37(5): 920-929 ).
设想用活化或抑制剂靶向STING可能是治疗其中1型IFN途径的调节是有益的疾病和病症和作为免疫原性或疫苗佐剂的有希望的方法,所述疾病和病症包括炎性、过敏性和自身免疫性疾病、感染性疾病、癌症、癌前综合征、肿瘤转移、代谢疾病、心血管疾病。It is envisioned that targeting STING with activation or inhibitors may be a promising approach for treating diseases and conditions in which modulation of the type 1 IFN pathway is beneficial and as an immunogenic or vaccine adjuvant, including inflammatory, allergic and autoimmune diseases, infectious diseases, cancer, precancerous syndromes, tumor metastasis, metabolic diseases, cardiovascular disease.
皮肤癌和各种皮肤病毒感染涉及免疫豁免环境,并且对病变的局部免疫应答的活化可以是局部治疗方法。STING激动剂可用于治疗病毒性疣、浅表性皮肤癌和恶化前的光化性角化病。通过双重作用机制,STING活化(例如,经由微针贴剂递送或局部制剂)可用于经由抗病毒I型干扰素产生直接控制HPV和通过增强先天免疫活化下游的适应性免疫应答间接控制HPV。STING激动剂可以活化病变中的先天免疫应答并驱动抗-HPV T-细胞应答。Skin cancer and various skin viral infections involve an immunoprivileged environment, and activation of local immune responses to lesions can be a local treatment approach. STING agonists can be used to treat viral warts, superficial skin cancers, and premalignant actinic keratoses. Through a dual mechanism of action, STING activation (e.g., via microneedle patch delivery or topical formulations) can be used to directly control HPV via antiviral type I interferon production and indirectly control HPV by enhancing the adaptive immune response downstream of innate immune activation. STING agonists can activate innate immune responses in lesions and drive anti-HPV T-cell responses.
最近的证据已表明,在肿瘤驻留的树突状细胞内STING途径的自发活化导致I型IFN产生和针对肿瘤的适应性免疫应答。此外,在肿瘤微环境内的抗原呈递细胞(APC)中该途径的活化驱动随后的针对肿瘤相关抗原的T-细胞引发。Corrales和Gajewski, Clin Cancer Res; 21(21); 4774-9, 2015。Recent evidence has shown that spontaneous activation of the STING pathway within tumor-resident dendritic cells leads to type I IFN production and adaptive immune responses against tumors. Furthermore, activation of this pathway in antigen presenting cells (APCs) within the tumor microenvironment drives subsequent T-cell priming against tumor-associated antigens. Corrales and Gajewski, Clin Cancer Res ; 21(21); 4774-9, 2015.
国际专利申请WO2014/093936、WO2014/189805、WO2013/185052、U.S.2014/0341976、WO 2015/077354、WO2015/185565、PCT/IB2017/051945和GB 1501462.4公开了某些环状二核苷酸以及它们经由STING的活化而诱导免疫应答的用途。国际专利申请WO2017/106740描述了可测量地抑制STING信号转导的环状二核苷酸和相关支架的用途和识别STING信号转导的有效抑制剂的方法。国际专利申请WO 2017/175147和WO 2017/175156描述了杂环酰胺类和它们的类似物作为STING调节剂的用途。International patent applications WO2014/093936, WO2014/189805, WO2013/185052, U.S.2014/0341976, WO 2015/077354, WO2015/185565, PCT/IB2017/051945 and GB 1501462.4 disclose certain cyclic dinucleotides and their use in inducing immune responses via activation of STING. International patent application WO2017/106740 describes the use of cyclic dinucleotides and related scaffolds that measurably inhibit STING signaling and methods for identifying effective inhibitors of STING signaling. International patent applications WO 2017/175147 and WO 2017/175156 describe the use of heterocyclic amides and their analogs as STING modulators.
本发明化合物调节STING的活性,且因此,可以在其中STING(干扰素基因刺激因子)的调节是有益的疾病、病症和/或病况的治疗中提供有益的治疗效果,例如用于炎症、过敏性和自身免疫性疾病、代谢疾病、心血管疾病、感染性疾病、癌症、癌前综合症和作为疫苗佐剂。The compounds of the invention modulate the activity of STING and, therefore, may provide beneficial therapeutic effects in the treatment of diseases, disorders and/or conditions in which modulation of STING (stimulator of interferon genes) is beneficial, for example, for inflammatory, allergic and autoimmune diseases, metabolic diseases, cardiovascular diseases, infectious diseases, cancer, precancerous syndromes and as vaccine adjuvants.
发明内容Summary of the invention
本发明涉及根据式(I)的化合物:The present invention relates to compounds according to formula (I):
其中:in:
q为0或1;q is 0 or 1;
r为0或1;r is 0 or 1;
s为0或1;s is 0 or 1;
其中q + r + s = 1或2;where q + r + s = 1 or 2;
当q为0时,RA1和RA2各自独立地为H、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-N(Re)(Rf)、-CO2Rf、-N(Rf)CORb、-N(Rg)SO2(C1-C4烷基)-N(Re)(Rf)、When q is 0, RA1 and RA2 are each independently H, halogen, hydroxy, -OP(O)( OH ) 2 , -OP(O)( RIRII ) 2 , -N( Re )( Rf ), -CO2Rf, -N( Rf ) CORb , -N( Rg ) SO2 ( C1 - C4alkyl )-N( Re )( Rf ),
-N(Rg)CO(C1-C4烷基)-N(Rh)(Rf)、任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-、任选取代的(C1-C6烷基)氨基-和任选取代的(C1-C6烷基)(C1-C4烷基)氨基-,-N(R g )CO(C 1 -C 4 alkyl)-N(R h )(R f ), optionally substituted (C 1 -C 6 alkyl), optionally substituted (C 1 -C 6 alkyl)oxy-, optionally substituted (C 1 -C 6 alkyl)amino-, and optionally substituted (C 1 -C 6 alkyl)(C 1 -C 4 alkyl)amino-,
其中所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-、任选取代的(C1-C6烷基)氨基-和任选取代的(C1-C6烷基)(C1-C4烷基)氨基-的(C1-C6烷基)任选地被1-4个各自独立地选自如下的取代基所取代:羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、C1-C4烷氧基-、-N(Re)(Rf)、-CO2(Rf)、-CON(Re)(Rf)、任选取代的苯基、任选取代的5-6元杂环烷基和任选取代的5-6元杂芳基,其中所述任选取代的苯基、5-6元杂环烷基或5-6元杂芳基任选地被1-4个各自独立地选自如下的取代基所取代:C1-C4烷基、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C6烷基)氨基-、(C1-C6烷基)(C1-C6烷基)氨基-、-(C1-C6烷基)-NH2、卤代(C1-C6烷基)、羟基-(C1-C4烷基)-、-(C1-C4烷基)-O-P(O)(OH)2、-(C1-C4烷基)-O-P(O)(RIRII)2、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2、-C1-C4烷基-(C1-C4烷氧基)和C1-C4烷氧基-(C1-C4烷氧基)-;wherein the optionally substituted (C 1 -C 6 alkyl), optionally substituted (C 1 -C 6 alkyl)oxy-, optionally substituted (C 1 -C 6 alkyl)amino- and optionally substituted (C 1 -C 6 alkyl)(C 1 -C 4 alkyl)amino-(C 1 -C 6 alkyl) are optionally substituted by 1-4 substituents each independently selected from the following substituents: hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , C 1 -C 4 alkoxy- , -N ( Re ) ( R f ) , -CO 2 (R f ), -CON(Re)(R f ), optionally substituted phenyl, optionally substituted 5-6 membered heterocycloalkyl and optionally substituted 5-6 membered heteroaryl, wherein the optionally substituted phenyl, 5-6 membered heterocycloalkyl or 5-6 membered heteroaryl are optionally substituted by 1-4 substituents each independently selected from the following substituents: C 1 -C 6 alkyl, optionally substituted (C 1 -C 6 alkyl)oxy-, optionally substituted ( C 1 -C 6 alkyl)amino-, and optionally substituted (C 1 -C 6 alkyl) are optionally substituted by 1-4 substituents each independently selected from the following substituents: 4 alkyl, halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 6 alkyl)amino-, (C 1 -C 6 alkyl)(C 1 -C 6 alkyl)amino-, -(C 1 -C 6 alkyl)-NH 2 , halo(C 1 -C 6 alkyl), hydroxy-(C 1 -C 4 alkyl)-, -(C 1 -C 4 alkyl)-OP(O)(OH) 2 , -(C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy ) - , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 R II ) 2 , -C 1 -C 4 alkyl-(C 1 -C 4 alkoxy) and C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-;
当r为0时,RB1和RB2各自独立地为H、任选取代的C1-C6烷基、卤代(C1-C6烷基)、任选取代的C2-C6烯基、任选取代的C2-C6炔基、任选取代的C3-C6环烷基、任选取代的4-6元杂环烷基、任选取代的苯基、任选取代的5-6元杂芳基或任选取代的9-10元杂芳基,When r is 0, RB1 and RB2 are each independently H, optionally substituted C1 - C6 alkyl, halo( C1 - C6 alkyl), optionally substituted C2 - C6 alkenyl, optionally substituted C2- C6 alkynyl, optionally substituted C3 - C6 cycloalkyl, optionally substituted 4-6 membered heterocycloalkyl, optionally substituted phenyl, optionally substituted 5-6 membered heteroaryl or optionally substituted 9-10 membered heteroaryl,
其中所述任选取代的C1-C6烷基、任选取代的C2-C6烯基、任选取代的C2-C6炔基、任选取代的C3-C6环烷基、任选取代的4-6元杂环烷基、任选取代的苯基、任选取代的5-6元杂芳基或任选取代的9-10元杂芳基任选地被1-4个各自独立地选自如下的取代基所取代:卤素、硝基、-Rc、-OH、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-ORc、-NH2、-NRcRc、-NRcRd、 -OCORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2、-SO2NRcRd、-OCONH2、-OCONRcRd、-NRdCORc、-NRdSORc、-NRdCO2Rc和-NRdSO2Rc;wherein the optionally substituted C 1 -C 6 alkyl, optionally substituted C 2 -C 6 alkenyl, optionally substituted C 2 -C 6 alkynyl, optionally substituted C 3 -C 6 cycloalkyl, optionally substituted 4-6 membered heterocycloalkyl, optionally substituted phenyl, optionally substituted 5-6 membered heteroaryl or optionally substituted 9-10 membered heteroaryl is optionally substituted with 1-4 substituents each independently selected from the group consisting of halogen, nitro, -R c , -OH, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -OR c , -NH 2 , -NR c R c , -NR c R d , -OCOR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d , -SO 2 NH 2 , -SO 2 NR c R d , -OCONH 2 , -OCONR c R d , -NR d COR c , -NR d SOR c , -NR d CO 2 R c , and -NR d SO 2 R c ;
当s为0时,RC1为H、卤素或C1-C4烷基和RC2 为任选取代的C1-C4烷基,其中所述任选取代的C1-C4烷基任选地被选自如下的取代基所取代: -ORc、-NRcRd、-CO2Rc、-CONRcRd、-SO2NRcRd和-OCONRcRd;When s is 0, R C1 is H, halogen or C 1 -C 4 alkyl and R C2 is optionally substituted C 1 -C 4 alkyl, wherein the optionally substituted C 1 -C 4 alkyl is optionally substituted with a substituent selected from the group consisting of: -OR c , -NR c R d , -CO 2 R c , -CONR c R d , -SO 2 NR c R d and -OCONR c R d ;
当q为1时,RA1和RA2各自独立地为-CH2-、-NRe-或-O-,并且A与RA1和RA2一起形成连接基团,其中A为-卤代(C1-C12烷基)-、任选取代的-C1-C12烷基-、任选取代的-C2-C12烯基-、任选取代的-C2-C12炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-,When q is 1, RA1 and RA2 are each independently -CH2- , -NRe- or -O-, and A together with RA1 and RA2 form a linking group, wherein A is -halo( C1 - C12alkyl )-, optionally substituted- C1 - C12alkyl- , optionally substituted- C2 - C12alkenyl- , optionally substituted- C2 - C12alkynyl- , optionally substituted- C1 - C6alkyl - OC1 - C6alkyl- , optionally substituted- C1 - C6alkyl -NRa - C1 - C6alkyl- , optionally substituted- C1 - C6alkyl- ( C3 - C6cycloalkyl )-C1-C6alkyl-, optionally substituted- C1-C6alkyl-phenyl-C1 - C6alkyl- , optionally substituted- C1 - C6alkyl- (4-6memberedheterocycloalkyl)-C1- C6alkyl- 1 -C 6 alkyl- or optionally substituted -C 1 -C 6 alkyl-(5-6 membered heteroaryl)-C 1 -C 6 alkyl-,
其中所述任选取代的-C1-C12烷基-、任选取代的-C2-C12烯基-、任选取代的-C2-C12炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-的烷基部分任选地被1-4个各自独立地选自如下的取代基所取代:卤素、卤代(C1-C4烷基)、-OH、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-ORc、-NH2、-NRcRd、-OCORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2、-SO2NRcRd、-OCONH2、-OCONRcRd、-NRdCORc、-NRdSORc、-NRdCO2Rc和-NRdSO2Rc,wherein the optionally substituted -C 1 -C 12 alkyl-, optionally substituted -C 2 -C 12 alkenyl-, optionally substituted -C 2 -C 12 alkynyl-, optionally substituted -C 1 -C 6 alkyl-OC 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-NR a -C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-phenyl-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 6 alkyl- or optionally substituted -C 1 -C 6 alkyl-(5-6 membered heteroaryl)-C 1 -C The alkyl portion of the 6 -alkyl- is optionally substituted with 1 to 4 substituents each independently selected from the group consisting of halogen, halo(C 1 -C 4 alkyl), -OH, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -OR c , -NH 2 , -NR c R d , -OCOR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d , -SO 2 NH 2 , -SO 2 NR c R d , -OCONH 2 , -OCONR c R d , -NR d COR c , -NR d SOR c , -NR d CO 2 R c and -NR d SO 2 R c ,
和and
所述任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-的C3-C6环烷基、苯基、4-6元杂环烷基或5-6元杂芳基部分任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、C1-C4烷基、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C1-C4烷氧基)-、-(C1-C4烷氧基)-O-P(O)(OH)2、-(C1-C4烷氧基)-O-P(O)(RIRII)2和C1-C4烷氧基-(C1-C4烷氧基)-;The C 3 -C 6 cycloalkyl, phenyl, 4-6-membered heterocycloalkyl or 5-6-membered heteroaryl portion of the optionally substituted -C 1 -C 6 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 6 alkyl- , optionally substituted -C 1 -C 6 alkyl-phenyl-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(4-6-membered heterocycloalkyl)-C 1 -C 6 alkyl- or optionally substituted -C 1 -C 6 alkyl- ( 5-6 - membered heteroaryl)-C 1 -C 6 alkyl- is optionally substituted with 1 to 4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, C 1 -C 6 4 alkyl, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 1 -C 4 alkoxy)-, -(C 1 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 1 -C 4 alkoxy)-OP(O)(R I R II ) 2 and C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-;
当r为1时, RB1和RB2各自独立地为-CRdRf-,并且B与RB1和RB2一起形成连接基团,其中B为键或B为-卤代(C1-C10烷基)-、任选取代的-C1-C10烷基-、任选取代的-C2-C10烯基-、任选取代的-C2-C10炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的C3-C6环烷基、任选取代的苯基、任选取代的4-6元杂环烷基、任选取代的5-6元杂芳基、任选取代的-C1-C4烷基-(C3-C6环烷基)-C1-C4烷基-、任选取代的-C1-C4烷基-苯基-C1-C4烷基-、任选取代的-C1-C4烷基-(4-6元杂环烷基)-C1-C4烷基-或任选取代的-C1-C4烷基-(5-6元杂芳基)-C1-C4烷基-,When r is 1, RB1 and RB2 are each independently -CRdRf- , and B, together with RB1 and RB2, forms a linking group, wherein B is a bond or B is -halo( C1 - C10 alkyl)-, optionally substituted -C1 - C10 alkyl-, optionally substituted -C2 - C10 alkenyl-, optionally substituted -C2 - C10 alkynyl-, optionally substituted -C1 - C6 alkyl- OC1 - C6 alkyl-, optionally substituted -C1 - C6 alkyl- NRa - C1 - C6 alkyl-, optionally substituted C3 - C6 cycloalkyl, optionally substituted phenyl, optionally substituted 4-6 membered heterocycloalkyl, optionally substituted 5-6 membered heteroaryl, optionally substituted -C1 - C4 alkyl-( C3 - C6 cycloalkyl) -C1 - C4 alkyl-, optionally substituted -C1 -C6 -C 1 -C 4 alkyl-phenyl-C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 4 alkyl-, or optionally substituted -C 1 -C 4 alkyl-(5-6 membered heteroaryl)-C 1 -C 4 alkyl-,
其中所述任选取代的-C1-C10烷基-、任选取代的-C2-C10烯基-、任选取代的-C2-C10炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的-C1-C4烷基-(C3-C6环烷基)-C1-C4烷基-、任选取代的-C1-C4烷基-苯基-C1-C4烷基-、任选取代的-C1-C4烷基-(4-6元杂环烷基)-C1-C4烷基-或任选取代的-C1-C4烷基-(5-6元杂芳基-C1-C4烷基)-的烷基部分任选地被1-4个各自独立地选自如下的取代基所取代:-C1-C4烷基、卤素、卤代(C1-C4烷基)、-OH、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-ORc、-NH2、wherein the alkyl portion of the optionally substituted -C 1 -C 10 alkyl-, optionally substituted -C 2 -C 10 alkenyl-, optionally substituted -C 2 -C 10 alkynyl-, optionally substituted -C 1 -C 6 alkyl-OC 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-NR a -C 1 -C 6 alkyl-, optionally substituted -C 1 -C 4 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-phenyl-C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 4 alkyl- or optionally substituted -C 1 -C 4 alkyl-(5-6 membered heteroaryl-C 1 -C 4 alkyl)- is optionally substituted with 1 to 4 substituents each independently selected from the following substituents: -C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-phenyl-C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 4 alkyl- 1 -C 4 alkyl, halogen, halo(C 1 -C 4 alkyl), -OH, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -OR c , -NH 2 ,
-NRcRd、-OCORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-NR c R d , -OCOR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d ,
-SO2NH2、-SO2NRcRd、-OCONH2、-OCONRcRd、-NRdCORc、-NRdSORc、-SO 2 NH 2 , -SO 2 NR c R d , -OCONH 2 , -OCONR c R d , -NR d COR c , -NR d SOR c ,
-NRdCO2Rc和-NRdSO2Rc,-NR d CO 2 R c and -NR d SO 2 R c ,
和and
所述任选取代的C3-C6环烷基、任选取代的苯基、任选取代的4-6元杂环烷基、任选取代的5-6元杂芳基、任选取代的-C1-C4烷基-(C3-C6环烷基)-C1-C4烷基-、任选取代的-C1-C4烷基-苯基-C1-C4烷基-、任选取代的-C1-C4烷基-(4-6元杂环烷基)-C1-C4烷基-或任选取代的-C1-C4烷基-(5-6元杂芳基)-C1-C4烷基-的C3-C6环烷基、苯基、4-6元杂环烷基或5-6元杂芳基部分任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、C1-C4烷基、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基) O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2和C1-C4烷氧基-(C1-C4烷氧基)-;The C 3 -C 6 cycloalkyl, phenyl, 4-6-membered heterocycloalkyl or 5-6-membered heteroaryl moiety of the optionally substituted C 3 -C 6 cycloalkyl, optionally substituted phenyl, optionally substituted 4-6-membered heterocycloalkyl, optionally substituted 5-6-membered heteroaryl, optionally substituted -C 1 -C 4 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 4 alkyl- , optionally substituted -C 1 -C 4 alkyl-phenyl-C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-(4-6-membered heterocycloalkyl)-C 1 -C 4 alkyl- or optionally substituted -C 1 -C 4 alkyl-(5-6-membered heteroaryl)-C 1 -C 4 alkyl- is optionally substituted with 1 to 4 substituents each independently selected from the following: halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 and C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-;
当s为1时,RC1和RC2各自独立地为-CH2-,并且C与RC1和RC2一起形成连接基团,其中C为-卤代(C1-C12烷基)-、任选取代的-C1-C12烷基-、任选取代的-C2-C12烯基-、任选取代的-C2-C12炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-,When s is 1, R C1 and R C2 are each independently -CH 2 -, and C together with R C1 and R C2 form a linking group, wherein C is -halo(C 1 -C 12 alkyl)-, optionally substituted -C 1 -C 12 alkyl-, optionally substituted -C 2 -C 12 alkenyl-, optionally substituted -C 2 -C 12 alkynyl-, optionally substituted -C 1 -C 6 alkyl-OC 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-NR a -C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-phenyl-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 6 6 alkyl- or optionally substituted -C 1 -C 6 alkyl-(5-6 membered heteroaryl)-C 1 -C 6 alkyl-,
其中所述任选取代的-C1-C12烷基-、任选取代的-C2-C12烯基-、任选取代的-C2-C12炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-的烷基部分任选地被1或2个各自独立地选自如下的取代基所取代:卤素、卤代(C1-C4烷基)、-OH、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-ORc、-NH2、wherein the optionally substituted -C 1 -C 12 alkyl-, optionally substituted -C 2 -C 12 alkenyl-, optionally substituted -C 2 -C 12 alkynyl-, optionally substituted -C 1 -C 6 alkyl-OC 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-NR a -C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-phenyl-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 6 alkyl- or optionally substituted -C 1 -C 6 alkyl-(5-6 membered heteroaryl)-C 1 -C The alkyl portion of 6 -alkyl- is optionally substituted by 1 or 2 substituents independently selected from the group consisting of halogen, halo(C 1 -C 4 alkyl), -OH, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -OR c , -NH 2 ,
-NRcRd、-OCORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-NR c R d , -OCOR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d ,
-SO2NH2、-SO2NRcRd、-OCONH2、-OCONRcRd、-NRdCORc、-SO 2 NH 2 , -SO 2 NR c R d , -OCONH 2 , -OCONR c R d , -NR d COR c ,
-NRdSORc、-NRdCO2Rc和-NRdSO2Rc,-NR d SOR c , -NR d CO 2 R c and -NR d SO 2 R c ,
和and
所述任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-的C3-C6环烷基、苯基、4-6元杂环烷基或5-6元杂芳基部分任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、C1-C4烷基、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2和C1-C4烷氧基-(C1-C4烷氧基)-;The C 3 -C 6 cycloalkyl, phenyl, 4-6-membered heterocycloalkyl or 5-6-membered heteroaryl portion of the optionally substituted -C 1 -C 6 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 6 alkyl- , optionally substituted -C 1 -C 6 alkyl-phenyl-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(4-6-membered heterocycloalkyl)-C 1 -C 6 alkyl- or optionally substituted -C 1 -C 6 alkyl- ( 5-6 - membered heteroaryl)-C 1 -C 6 alkyl- is optionally substituted with 1 to 4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, C 1 -C 6 4 alkyl, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 and C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-;
R3和R5各自独立地为-CON(Rd)(Rf),或R3和R5之一为-CON(Rd)(Rf),且R3和R5中的另一个为H、COOH或-CO2(Rc);R 3 and R 5 are each independently -CON(R d )(R f ), or one of R 3 and R 5 is -CON(R d )(R f ), and the other of R 3 and R 5 is H, COOH or -CO 2 (R c );
R4和R6各自独立地选自H、卤素、卤代(C1-C6烷基)、卤代(C1-C6烷氧基)-、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-NH2、-NRcRc、-NRcRd、-CORc、-CO2Rc、-N(Rd)CORc、-N(Rd)SO2Rc、-N(Rg)SO2(C1-C2烷基)-N(Rh)(Rf)、-N(Rg)CO(C1-C2烷基)-N(Rh)(Rf)、任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-、任选取代的(C1-C6烷基)氨基-和任选取代的(C1-C6烷基)(C1-C4烷基)氨基-, R4 and R6 are each independently selected from H, halogen, halo( C1 - C6 alkyl), halo( C1 - C6 alkoxy)-, hydroxy, -OP(O)(OH) 2 , -OP( O )(R I R II ) 2 , -NH2 , -NRcRc, -NRcRd , -CORc , -CO2Rc , -N( Rd ) CORc , -N (Rd)SO2Rc, -N( Rg ) SO2 ( C1 - C2alkyl )-N( Rh ) ( Rf ), -N ( Rg )CO( C1 - C2alkyl )-N( Rh ) ( Rf ), optionally substituted ( C1 - C6alkyl ), optionally substituted ( C1 - C6alkyl )oxy-, optionally substituted ( C1 -C6alkyl)amino-, and optionally substituted ( C1 - C6alkyl ) -C 6 alkyl)(C 1 -C 4 alkyl)amino-,
其中所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-、任选取代的(C1-C6烷基)氨基-和任选取代的(C1-C6烷基)(C1-C4烷基)氨基-的(C1-C6烷基)任选地被1-4个各自独立地选自如下的取代基所取代:-OH、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-ORc、-NH2、-NRcRc、-NRcRd、-CO2H、-CO2Rc、-OCORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2、-SO2NRcRd、-OCONH2、-OCONRcRd、-NRdCORc、-NRdSORc、-NRdCO2Rc、-NRdSO2Rc、任选取代的苯基、任选取代的5-6元杂环烷基和任选取代的5-6元杂芳基,其中所述任选取代的苯基、5-6元杂环烷基或5-6元杂芳基任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、C1-C4烷基、卤代(C1-C4烷基)、羟基-(C1-C4烷基)-、-(C1-C4烷基)-O-P(O)(OH)2、-(C1-C4烷基)-O-P(O)(RIRII)2、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2、C1-C4烷氧基-(C1-C4烷氧基)-、-CORd、-CON(Rd)(Rf)和-CO2Rd;wherein the optionally substituted (C 1 -C 6 alkyl), optionally substituted (C 1 -C 6 alkyl)oxy-, optionally substituted (C 1 -C 6 alkyl)amino- and optionally substituted (C 1 -C 6 alkyl)(C 1 -C 4 alkyl)amino-(C 1 -C 6 alkyl) are optionally substituted by 1 to 4 substituents each independently selected from the group consisting of -OH, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -OR c , -NH 2 , -NR c R c , -NR c R d , -CO 2 H , -CO 2 R c , -OCOR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d , -SO 2 NH 2 , -SO 2 NR c R d , -OCONH 2 2 , -OCONR c R d , -NR d COR c , -NR d SOR c , -NR d CO 2 R c , -NR d SO 2 R c , optionally substituted phenyl, optionally substituted 5-6 membered heterocycloalkyl and optionally substituted 5-6 membered heteroaryl, wherein the optionally substituted phenyl, 5-6 membered heterocycloalkyl or 5-6 membered heteroaryl is optionally substituted with 1-4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), hydroxy-(C 1 -C 4 alkyl)-, -(C 1 -C 4 alkyl)-OP(O)(OH) 2 , -(C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 , C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-, -COR d , -CON(R d )(R f ) and -CO 2 R d ;
R14 为任选取代的C1-C4烷基,其中所述任选取代的C1-C4烷基任选地被选自如下的取代基所取代: -ORc、-NRcRd、-CO2Rc、-CONRcRd、-SO2NRcRd和-OCONRcRd;R 14 is an optionally substituted C 1 -C 4 alkyl group, wherein the optionally substituted C 1 -C 4 alkyl group is optionally substituted by a substituent selected from the group consisting of: -OR c , -NR c R d , -CO 2 R c , -CONR c R d , -SO 2 NR c R d and -OCONR c R d ;
R16为H、卤素或C1-C4烷基;R 16 is H, halogen or C 1 -C 4 alkyl;
R15和R17各自独立地为H、环丙基或C1-C4烷基;R 15 and R 17 are each independently H, cyclopropyl or C 1 -C 4 alkyl;
Ra为H、-Rc、-CORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2或-SO2NRcRd;R a is H, -R c , -COR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d , -SO 2 NH 2 or -SO 2 NR c R d ;
各Rb独立地为C1-C4烷基、卤代(C1-C4烷基)、-(C1-C4烷基)-OH、-(C1-C4烷基)-O-P(O)(OH)2、-(C1-C4烷基)-O-P(O)(RIRII)2、-(C1-C4烷基)-O-(C1-C4烷基)、-(C1-C4烷基)-N(Re)(Rf)、-(C1-C4烷基)-O-CO(C1-C4烷基)或-(C1-C4烷基)-CO-O-(C1-C4烷基);each R b is independently C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)-OH, -(C 1 -C 4 alkyl)-OP(O)(OH) 2 , -(C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , -(C 1 -C 4 alkyl)-O-(C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)-N(R e )(R f ), -(C 1 -C 4 alkyl)-O-CO(C 1 -C 4 alkyl), or -(C 1 -C 4 alkyl)-CO-O-(C 1 -C 4 alkyl);
各Rc独立地为C1-C4烷基、卤代(C1-C4烷基)、-(C1-C4烷基)-OH、-(C1-C4烷基)-O-P(O)(OH)2、-(C1-C4烷基)-O-P(O)(RIRII)2、-(C1-C4烷基)-O-(C1-C4烷基)、-(C1-C4烷基)-N(Re)(Rf)、-(C1-C4烷基)-O-CO(C1-C4烷基)、-(C1-C4烷基)-CO-O-(C1-C4烷基)、任选取代的C3-C6环烷基、任选取代的苯基、任选取代的4-6元杂环烷基、任选取代的5-6元杂芳基、任选取代的9-10元杂芳基、任选取代的-C1-C4烷基-C3-C6环烷基、任选取代的-C1-C4烷基-苯基、任选取代的-C1-C4烷基-4-6元杂环烷基、任选取代的-C1-C4烷基-5-6元杂芳基或任选取代的-C1-C4烷基-9-10元杂芳基,each R c is independently C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)-OH, -(C 1 -C 4 alkyl)-OP(O)(OH) 2 , -(C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , -(C 1 -C 4 alkyl)-O-(C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)-N(R e )(R f ), -(C 1 -C 4 alkyl)-O-CO(C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)-CO-O-(C 1 -C 4 alkyl), optionally substituted C 3 -C 6 cycloalkyl, optionally substituted phenyl, optionally substituted 4-6 membered heterocycloalkyl, optionally substituted 5-6 membered heteroaryl, optionally substituted 9-10 membered heteroaryl, optionally substituted -C 1 -C 4 alkyl-C 3 -C 6 cycloalkyl, optionally substituted -C 1 -C 4 alkyl-phenyl, optionally substituted -C 1 -C 4 alkyl-4-6 membered heterocycloalkyl, optionally substituted -C 1 -C 4 alkyl-5-6 membered heteroaryl or optionally substituted -C 1 -C 4 alkyl-9-10 membered heteroaryl,
其中所述取代的C3-C6环烷基、任选取代的苯基、任选取代的4-6元杂环烷基、任选取代的5-6元杂芳基、任选取代的9-10元杂芳基、任选取代的-C1-C4烷基-C3-C6环烷基、任选取代的-C1-C4烷基-苯基、任选取代的-C1-C4烷基-4-6元杂环烷基、任选取代的-C1-C4烷基-5-6元杂芳基或任选取代的-C1-C4烷基-9-10元杂芳基的C3-C6环烷基、苯基、4-6元杂环烷基、5-6元杂芳基或任选取代的9-10元杂芳基部分任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、-(C1-C4烷基)NH2、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、-C1-C4烷基、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2、C1-C4烷氧基-(C1-C4烷氧基)-、-CORd、-CON(Rd)(Rf)和-CO2Rd;wherein the substituted C3 - C6 cycloalkyl, optionally substituted phenyl, optionally substituted 4-6 membered heterocycloalkyl, optionally substituted 5-6 membered heteroaryl, optionally substituted 9-10 membered heteroaryl, optionally substituted -C1 - C4 alkyl- C3 - C6 cycloalkyl, optionally substituted -C1 - C4 alkyl-phenyl, optionally substituted -C1 - C4 alkyl-4-6 membered heterocycloalkyl, optionally substituted -C1 - C4 alkyl-5-6 membered heteroaryl or optionally substituted -C1- C4 alkyl-9-10 membered heteroaryl C3 -C6 cycloalkyl , phenyl, 4-6 membered heterocycloalkyl, 5-6 membered heteroaryl or optionally substituted 9-10 membered heteroaryl moiety is optionally substituted with 1-4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, -(C 1 -C 4 alkyl)NH 2 , (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, -C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 , C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-, -COR d , -CON(R d )(R f ) and -CO 2 R d ;
各Rd独立地为H或C1-C4烷基;Each R d is independently H or C 1 -C 4 alkyl;
各Re独立地为H、C1-C4烷基、-CO(C1-C4烷基)、-OCO(C1-C4烷基)、-CO2(C1-C4烷基)、-(C1-C4烷基)NH2、-(C1-C4烷基) C1-C4烷氧基、-CO-(任选取代的5-6元杂环烷基)、-CO(C1-C4烷基)-(任选取代的5-6元杂环烷基)、-CO(任选取代的5-6元杂芳基)或-CO(C1-C4烷基)-(任选取代的5-6元杂芳基),each R e is independently H, C 1 -C 4 alkyl, -CO(C 1 -C 4 alkyl), -OCO(C 1 -C 4 alkyl), -CO 2 (C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)NH 2 , -(C 1 -C 4 alkyl)C 1 -C 4 alkoxy, -CO-(optionally substituted 5-6 membered heterocycloalkyl), -CO(C 1 -C 4 alkyl)-(optionally substituted 5-6 membered heterocycloalkyl), -CO(optionally substituted 5-6 membered heteroaryl), or -CO(C 1 -C 4 alkyl)-(optionally substituted 5-6 membered heteroaryl),
其中所述任选取代的5-6元杂环烷基或任选取代的5-6元杂芳基任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、C1-C4烷基、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基) O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2、C1-C4烷氧基-(C1-C4烷氧基)-、-CORd、-CON(Rd)(Rf)和-CO2Rd;wherein the optionally substituted 5-6 membered heterocycloalkyl or optionally substituted 5-6 membered heteroaryl is optionally substituted by 1-4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 , C 1 -C 4 alkoxy-(C 1 -C 4 4 alkoxy)-, -COR d , -CON(R d )(R f ) and -CO 2 R d ;
各Rf独立地为H或C1-C4烷基;Each R f is independently H or C 1 -C 4 alkyl;
Rg和Rh各自独立地为H或C1-C4烷基,或Rg和Rh与将它们连接的一个或多个原子一起形成5-6元环; Rg and Rh are each independently H or C1 - C4 alkyl, or Rg and Rh together with the one or more atoms to which they are attached form a 5-6 membered ring;
和RI和RII 在每次出现时独立地为(C1-C6烷基)氧基-;和and R I and R II are independently at each occurrence (C 1 -C 6 alkyl)oxy-; and
RX或Ry中的至少一个独立地为C1-C4烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 4 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C4烷基;or R x and R y are both independently C 1 -C 4 alkyl;
或其互变异构体;or a tautomer thereof;
或其盐。or a salt thereof.
应理解,本文提及式(I)化合物及其盐涵盖作为游离碱或其盐、例如作为其药学上可接受的盐的式(I)化合物。因此,在一个实施方案中,本发明涉及作为游离碱的式(I)化合物。在另一个实施方案中,本发明涉及式(I)化合物及其盐。在进一步实施方案中,本发明涉及式(I)化合物及其药学上可接受的盐。It should be understood that references herein to compounds of formula (I) and their salts encompass compounds of formula (I) as free bases or salts thereof, e.g., as pharmaceutically acceptable salts thereof. Thus, in one embodiment, the present invention relates to compounds of formula (I) as free bases. In another embodiment, the present invention relates to compounds of formula (I) and their salts. In a further embodiment, the present invention relates to compounds of formula (I) and their pharmaceutically acceptable salts.
根据式(I)的化合物或其盐、特别是药学上可接受的盐是STING的调节剂。因此,本发明提供了式(I)化合物或其盐、特别是其药学上可接受的盐,其用于疗法中。本发明特别提供了式(I)化合物或其药学上可接受的盐作为活性治疗物质在治疗STING介导的疾病或病症中的用途,特别是用于治疗通过STING的激动或拮抗介导的疾病的用途。本发明还提供了式(I)化合物或其盐、特别是其药学上可接受的盐,其用于制备用于治疗STING介导的疾病或病症的药物。The compound according to formula (I) or a salt thereof, in particular a pharmaceutically acceptable salt thereof, is a modulator of STING. Therefore, the present invention provides a compound of formula (I) or a salt thereof, in particular a pharmaceutically acceptable salt thereof, for use in therapy. The present invention particularly provides the use of a compound of formula (I) or a pharmaceutically acceptable salt thereof as an active therapeutic substance in the treatment of a disease or condition mediated by STING, in particular for the treatment of a disease mediated by the agonist or antagonist of STING. The present invention also provides a compound of formula (I) or a salt thereof, in particular a pharmaceutically acceptable salt thereof, for the preparation of a medicament for the treatment of a disease or condition mediated by STING.
本发明还涉及调节STING的方法,所述方法包括使细胞与根据式(I)的化合物或其盐、特别是药学上可接受的盐接触。本发明进一步涉及治疗STING介导的疾病或病症的方法,所述方法包括将治疗有效量的根据式(I)的化合物或其盐、特别是药学上可接受的盐施用于有需要的患者(人或其它哺乳动物,特别是人)。这样的STING介导的疾病或病症包括炎症、过敏性和自身免疫性疾病、感染性疾病、癌症、癌前综合征、代谢疾病和心血管疾病。此外,STING的调节剂可用作免疫原性组合物或疫苗佐剂。The present invention also relates to a method for regulating STING, the method comprising contacting a cell with a compound according to formula (I) or a salt thereof, in particular a pharmaceutically acceptable salt. The present invention further relates to a method for treating a disease or condition mediated by STING, the method comprising administering a therapeutically effective amount of a compound according to formula (I) or a salt thereof, in particular a pharmaceutically acceptable salt, to a patient (human or other mammal, in particular human) in need thereof. Such STING-mediated diseases or conditions include inflammation, allergic and autoimmune diseases, infectious diseases, cancer, precancerous syndromes, metabolic diseases and cardiovascular diseases. In addition, modulators of STING can be used as immunogenic compositions or vaccine adjuvants.
本发明进一步涉及药物组合物,其包含根据式(I)的化合物或其盐、特别是药学上可接受的盐和药学上可接受的赋形剂。具体地,本发明涉及用于治疗STING介导的疾病或病症的药物组合物,其中所述组合物包含根据式(I)的化合物或其盐、特别是药学上可接受的盐和药学上可接受的赋形剂。The present invention further relates to a pharmaceutical composition comprising a compound according to formula (I) or a salt thereof, in particular a pharmaceutically acceptable salt, and a pharmaceutically acceptable excipient. Specifically, the present invention relates to a pharmaceutical composition for treating a STING-mediated disease or condition, wherein the composition comprises a compound according to formula (I) or a salt thereof, in particular a pharmaceutically acceptable salt, and a pharmaceutically acceptable excipient.
发明详述DETAILED DESCRIPTION OF THE INVENTION
根据本发明的一个方面,本发明涉及式(I)化合物According to one aspect of the present invention, the present invention relates to a compound of formula (I)
其中:in:
q为0或1;q is 0 or 1;
r为0或1;r is 0 or 1;
s为0或1;s is 0 or 1;
其中q + r + s = 1或2;where q + r + s = 1 or 2;
当q为0时,RA1和RA2各自独立地为H、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-N(Re)(Rf)、-CO2Rf、-N(Rf)CORb、-N(Rg)SO2(C1-C4烷基)-N(Re)(Rf)、-N(Rg)CO(C1-C4烷基)-N(Rh)(Rf)、任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-、任选取代的(C1-C6烷基)氨基-和任选取代的(C1-C6烷基)(C1-C4烷基)氨基-,When q is 0, RA1 and RA2 are each independently H, halogen, hydroxy, -OP(O)( OH ) 2 , -OP(O)( RIRII ) 2 , -N( Re )(Rf ) , -CO2Rf, -N( Rf ) CORb , -N( Rg ) SO2 ( C1 - C4 alkyl)-N( Re )( Rf ), -N( Rg )CO( C1 - C4 alkyl)-N( Rh )( Rf ), optionally substituted ( C1 - C6 alkyl), optionally substituted ( C1 - C6 alkyl)oxy-, optionally substituted ( C1 - C6 alkyl)amino-, and optionally substituted ( C1 - C6 alkyl)( C1 - C4 alkyl)amino-,
其中所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-、任选取代的(C1-C6烷基)氨基-和任选取代的(C1-C6烷基)(C1-C4烷基)氨基-的(C1-C6烷基)任选地被1-4个各自独立地选自如下的取代基所取代:羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、C1-C4烷氧基-、-N(Re)(Rf)、-CO2(Rf)、-CON(Re)(Rf)、任选取代的苯基、任选取代的5-6元杂环烷基和任选取代的5-6元杂芳基,其中所述任选取代的苯基、5-6元杂环烷基或5-6元杂芳基任选地被1-4个各自独立地选自如下的取代基所取代:C1-C4烷基、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C6烷基)氨基-、(C1-C6烷基)(C1-C6烷基)氨基-、-(C1-C6烷基)-NH2、卤代(C1-C6烷基)、羟基-(C1-C4烷基)-、-(C1-C4烷基)-O-P(O)(OH)2、-(C1-C4烷基)-O-P(O)(RIRII)2、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2、-C1-C4烷基-(C1-C4烷氧基)和C1-C4烷氧基-(C1-C4烷氧基)-;wherein the optionally substituted (C 1 -C 6 alkyl), optionally substituted (C 1 -C 6 alkyl)oxy-, optionally substituted (C 1 -C 6 alkyl)amino- and optionally substituted (C 1 -C 6 alkyl)(C 1 -C 4 alkyl)amino-(C 1 -C 6 alkyl) are optionally substituted by 1-4 substituents each independently selected from the following substituents: hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , C 1 -C 4 alkoxy- , -N ( Re ) ( R f ) , -CO 2 (R f ), -CON(Re)(R f ), optionally substituted phenyl, optionally substituted 5-6 membered heterocycloalkyl and optionally substituted 5-6 membered heteroaryl, wherein the optionally substituted phenyl, 5-6 membered heterocycloalkyl or 5-6 membered heteroaryl are optionally substituted by 1-4 substituents each independently selected from the following substituents: C 1 -C 6 alkyl, optionally substituted (C 1 -C 6 alkyl)oxy-, optionally substituted ( C 1 -C 6 alkyl)amino-, and optionally substituted (C 1 -C 6 alkyl) are optionally substituted by 1-4 substituents each independently selected from the following substituents: 4 alkyl, halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 6 alkyl)amino-, (C 1 -C 6 alkyl)(C 1 -C 6 alkyl)amino-, -(C 1 -C 6 alkyl)-NH 2 , halo(C 1 -C 6 alkyl), hydroxy-(C 1 -C 4 alkyl)-, -(C 1 -C 4 alkyl)-OP(O)(OH) 2 , -(C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy ) - , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 R II ) 2 , -C 1 -C 4 alkyl-(C 1 -C 4 alkoxy) and C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-;
当r为0时,RB1和RB2各自独立地为H、任选取代的C1-C6烷基、卤代(C1-C6烷基)、任选取代的C2-C6烯基、任选取代的C2-C6炔基、任选取代的C3-C6环烷基、任选取代的4-6元杂环烷基、任选取代的苯基、任选取代的5-6元杂芳基或任选取代的9-10元杂芳基,When r is 0, RB1 and RB2 are each independently H, optionally substituted C1 - C6 alkyl, halo( C1 - C6 alkyl), optionally substituted C2 - C6 alkenyl, optionally substituted C2- C6 alkynyl, optionally substituted C3 - C6 cycloalkyl, optionally substituted 4-6 membered heterocycloalkyl, optionally substituted phenyl, optionally substituted 5-6 membered heteroaryl or optionally substituted 9-10 membered heteroaryl,
其中所述任选取代的C1-C6烷基、任选取代的C2-C6烯基、任选取代的C2-C6炔基、任选取代的C3-C6环烷基、任选取代的4-6元杂环烷基、任选取代的苯基、任选取代的5-6元杂芳基或任选取代的9-10元杂芳基任选地被1-4个各自独立地选自如下的取代基所取代:卤素、硝基、-Rc、-OH、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-ORc、-NH2、-NRcRc、-NRcRd、-OCORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2、-SO2NRcRd、-OCONH2、-OCONRcRd、-NRdCORc、-NRdSORc、-NRdCO2Rc和-NRdSO2Rc;wherein the optionally substituted C 1 -C 6 alkyl, optionally substituted C 2 -C 6 alkenyl, optionally substituted C 2 -C 6 alkynyl, optionally substituted C 3 -C 6 cycloalkyl, optionally substituted 4-6 membered heterocycloalkyl, optionally substituted phenyl, optionally substituted 5-6 membered heteroaryl or optionally substituted 9-10 membered heteroaryl is optionally substituted with 1 to 4 substituents each independently selected from the group consisting of halogen, nitro, -R c , -OH, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -OR c , -NH 2 , -NR c R c , -NR c R d , -OCOR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d , -SO 2 NH 2 , -SO 2 NR c R d , -OCONH 2 , -OCONR c R d , -NR d COR c , -NR d SOR c , -NR d CO 2 R c , and -NR d SO 2 R c ;
当s为0时,RC1为H、卤素或C1-C4烷基,并且RC2 为任选取代的C1-C4烷基,其中所述任选取代的C1-C4烷基任选地被选自如下的取代基所取代: -ORc、-NRcRd、-CO2Rc、-CONRcRd、-SO2NRcRd和-OCONRcRd;When s is 0, R C1 is H, halogen or C 1 -C 4 alkyl, and R C2 is optionally substituted C 1 -C 4 alkyl, wherein the optionally substituted C 1 -C 4 alkyl is optionally substituted with a substituent selected from the group consisting of: -OR c , -NR c R d , -CO 2 R c , -CONR c R d , -SO 2 NR c R d and -OCONR c R d ;
当q为1时,RA1和RA2各自独立地为-CH2-、-NRe-或-O-,并且A与RA1和RA2一起形成连接基团,其中A为-卤代(C1-C12烷基)-、任选取代的-C1-C12烷基-、任选取代的-C2-C12烯基-、任选取代的-C2-C12炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-,When q is 1, RA1 and RA2 are each independently -CH2- , -NRe- or -O-, and A together with RA1 and RA2 form a linking group, wherein A is -halo( C1 - C12alkyl )-, optionally substituted- C1 - C12alkyl- , optionally substituted- C2 - C12alkenyl- , optionally substituted- C2 - C12alkynyl- , optionally substituted- C1 - C6alkyl - OC1 - C6alkyl- , optionally substituted- C1 - C6alkyl -NRa - C1 - C6alkyl- , optionally substituted- C1 - C6alkyl- ( C3 - C6cycloalkyl )-C1-C6alkyl-, optionally substituted- C1-C6alkyl-phenyl-C1 - C6alkyl- , optionally substituted- C1 - C6alkyl- (4-6memberedheterocycloalkyl)-C1- C6alkyl- 1 -C 6 alkyl- or optionally substituted -C 1 -C 6 alkyl-(5-6 membered heteroaryl)-C 1 -C 6 alkyl-,
其中所述任选取代的-C1-C12烷基-、任选取代的-C2-C12烯基-、任选取代的-C2-C12炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-的烷基部分任选地被1-4个各自独立地选自如下的取代基所取代:卤素、卤代(C1-C4烷基)、-OH、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-ORc、-NH2、-NRcRd、-OCORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2、-SO2NRcRd、-OCONH2、-OCONRcRd、-NRdCORc、-NRdSORc、-NRdCO2Rc和-NRdSO2Rc,wherein the optionally substituted -C 1 -C 12 alkyl-, optionally substituted -C 2 -C 12 alkenyl-, optionally substituted -C 2 -C 12 alkynyl-, optionally substituted -C 1 -C 6 alkyl-OC 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-NR a -C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-phenyl-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 6 alkyl- or optionally substituted -C 1 -C 6 alkyl-(5-6 membered heteroaryl)-C 1 -C The alkyl portion of the 6 -alkyl- is optionally substituted with 1 to 4 substituents each independently selected from the group consisting of halogen, halo(C 1 -C 4 alkyl), -OH, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -OR c , -NH 2 , -NR c R d , -OCOR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d , -SO 2 NH 2 , -SO 2 NR c R d , -OCONH 2 , -OCONR c R d , -NR d COR c , -NR d SOR c , -NR d CO 2 R c and -NR d SO 2 R c ,
和and
所述任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-的C3-C6环烷基、苯基、4-6元杂环烷基或5-6元杂芳基部分任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、C1-C4烷基、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C1-C4烷氧基)-、-(C1-C4烷氧基)-O-P(O)(OH)2、-(C1-C4烷氧基)-O-P(O)(RIRII)2和C1-C4烷氧基-(C1-C4烷氧基)-;The C 3 -C 6 cycloalkyl, phenyl, 4-6-membered heterocycloalkyl or 5-6-membered heteroaryl portion of the optionally substituted -C 1 -C 6 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 6 alkyl- , optionally substituted -C 1 -C 6 alkyl-phenyl-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(4-6-membered heterocycloalkyl)-C 1 -C 6 alkyl- or optionally substituted -C 1 -C 6 alkyl- ( 5-6 - membered heteroaryl)-C 1 -C 6 alkyl- is optionally substituted with 1 to 4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, C 1 -C 6 4 alkyl, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 1 -C 4 alkoxy)-, -(C 1 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 1 -C 4 alkoxy)-OP(O)(R I R II ) 2 and C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-;
当r为1时, RB1和RB2各自独立地为-CRdRf-,且B与RB1和RB2一起形成连接基团,其中B为键或B为-卤代(C1-C10烷基)-、任选取代的-C1-C10烷基-、任选取代的-C2-C10烯基-、任选取代的-C2-C10炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的C3-C6环烷基、任选取代的苯基、任选取代的4-6元杂环烷基、任选取代的5-6元杂芳基、任选取代的-C1-C4烷基-(C3-C6环烷基)-C1-C4烷基-、任选取代的-C1-C4烷基-苯基-C1-C4烷基-、任选取代的-C1-C4烷基-(4-6元杂环烷基)-C1-C4烷基-或任选取代的-C1-C4烷基-(5-6元杂芳基)-C1-C4烷基-,When r is 1, RB1 and RB2 are each independently -CRdRf- , and B, together with RB1 and RB2, forms a linking group, wherein B is a bond or B is -halo( C1 - C10 alkyl)-, optionally substituted -C1 - C10 alkyl-, optionally substituted -C2 - C10 alkenyl-, optionally substituted -C2 - C10 alkynyl-, optionally substituted -C1 - C6 alkyl- OC1 - C6 alkyl-, optionally substituted -C1 - C6 alkyl- NRa - C1 - C6 alkyl-, optionally substituted C3 - C6 cycloalkyl, optionally substituted phenyl, optionally substituted 4-6 membered heterocycloalkyl, optionally substituted 5-6 membered heteroaryl, optionally substituted -C1 - C4 alkyl-( C3 - C6 cycloalkyl) -C1 - C4 alkyl-, optionally substituted -C1 -C6 -C 1 -C 4 alkyl-phenyl-C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 4 alkyl-, or optionally substituted -C 1 -C 4 alkyl-(5-6 membered heteroaryl)-C 1 -C 4 alkyl-,
其中所述任选取代的-C1-C10烷基-、任选取代的-C2-C10烯基-、任选取代的-C2-C10炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的-C1-C4烷基-(C3-C6环烷基)-C1-C4烷基-、任选取代的-C1-C4烷基-苯基-C1-C4烷基-、任选取代的-C1-C4烷基-(4-6元杂环烷基)-C1-C4烷基-或任选取代的-C1-C4烷基-(5-6元杂芳基-C1-C4烷基)-的烷基部分任选地被1- 4个各自独立地选自如下的取代基所取代:-C1-C4烷基、卤素、卤代(C1-C4烷基)、-OH、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-ORc、-NH2、-NRcRd、-OCORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2、-SO2NRcRd、-OCONH2、-OCONRcRd、-NRdCORc、-NRdSORc、-NRdCO2Rc和-NRdSO2Rc,wherein the alkyl portion of the optionally substituted -C 1 -C 10 alkyl-, optionally substituted -C 2 -C 10 alkenyl-, optionally substituted -C 2 -C 10 alkynyl-, optionally substituted -C 1 -C 6 alkyl-OC 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-NR a -C 1 -C 6 alkyl-, optionally substituted -C 1 -C 4 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-phenyl-C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 4 alkyl- or optionally substituted -C 1 -C 4 alkyl-(5-6 membered heteroaryl-C 1 -C 4 alkyl)- is optionally substituted with 1-C 4 alkyl- wherein the at least one aryl group is substituted with 4 substituents each independently selected from the group consisting of -C 1 -C 4 alkyl, halogen, halo(C 1 -C 4 alkyl), -OH, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -OR c , -NH 2 , -NR c R d , -OCOR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d , -SO 2 NH 2 , -SO 2 NR c R d , -OCONH 2 , -OCONR c R d , -NR d COR c , -NR d SOR c , -NR d CO 2 R c and -NR d SO 2 R c ,
和and
所述任选取代的C3-C6环烷基、任选取代的苯基、任选取代的4-6元杂环烷基、任选取代的5-6元杂芳基、任选取代的-C1-C4烷基-(C3-C6环烷基)-C1-C4烷基-、任选取代的-C1-C4烷基-苯基-C1-C4烷基-、任选取代的-C1-C4烷基-(4-6元杂环烷基)-C1-C4烷基-或任选取代的-C1-C4烷基-(5-6元杂芳基)-C1-C4烷基-的C3-C6环烷基、苯基、4-6元杂环烷基或5-6元杂芳基部分任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、C1-C4烷基、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基) O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2和C1-C4烷氧基-(C1-C4烷氧基)-;The C 3 -C 6 cycloalkyl, phenyl, 4-6-membered heterocycloalkyl or 5-6-membered heteroaryl moiety of the optionally substituted C 3 -C 6 cycloalkyl, optionally substituted phenyl, optionally substituted 4-6-membered heterocycloalkyl, optionally substituted 5-6-membered heteroaryl, optionally substituted -C 1 -C 4 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 4 alkyl- , optionally substituted -C 1 -C 4 alkyl-phenyl-C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-(4-6-membered heterocycloalkyl)-C 1 -C 4 alkyl- or optionally substituted -C 1 -C 4 alkyl-(5-6-membered heteroaryl)-C 1 -C 4 alkyl- is optionally substituted with 1 to 4 substituents each independently selected from the following: halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 and C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-;
当s为1时,RC1和RC2各自独立地为-CH2-,并且C与RC1和RC2一起形成连接基团,其中C为-卤代(C1-C12烷基)-、任选取代的-C1-C12烷基-、任选取代的-C2-C12烯基-、任选取代的-C2-C12炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-,When s is 1, R C1 and R C2 are each independently -CH 2 -, and C together with R C1 and R C2 form a linking group, wherein C is -halo(C 1 -C 12 alkyl)-, optionally substituted -C 1 -C 12 alkyl-, optionally substituted -C 2 -C 12 alkenyl-, optionally substituted -C 2 -C 12 alkynyl-, optionally substituted -C 1 -C 6 alkyl-OC 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-NR a -C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-phenyl-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 6 6 alkyl- or optionally substituted -C 1 -C 6 alkyl-(5-6 membered heteroaryl)-C 1 -C 6 alkyl-,
其中所述任选取代的-C1-C12烷基-、任选取代的-C2-C12烯基-、任选取代的-C2-C12炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-的烷基部分任选地被1或2个各自独立地选自如下的取代基所取代:卤素、卤代(C1-C4烷基)、-OH、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-ORc、-NH2、-NRcRd、-OCORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2、-SO2NRcRd、-OCONH2、-OCONRcRd、-NRdCORc、-NRdSORc、-NRdCO2Rc、和-NRdSO2Rc,wherein the optionally substituted -C 1 -C 12 alkyl-, optionally substituted -C 2 -C 12 alkenyl-, optionally substituted -C 2 -C 12 alkynyl-, optionally substituted -C 1 -C 6 alkyl-OC 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-NR a -C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-phenyl-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 6 alkyl- or optionally substituted -C 1 -C 6 alkyl-(5-6 membered heteroaryl)-C 1 -C The alkyl portion of the 6 -alkyl- is optionally substituted with 1 or 2 substituents each independently selected from the group consisting of halogen, halo(C 1 -C 4 alkyl), -OH, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -OR c , -NH 2 , -NR c R d , -OCOR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d , -SO 2 NH 2 , -SO 2 NR c R d , -OCONH 2 , -OCONR c R d , -NR d COR c , -NR d SOR c , -NR d CO 2 R c , and -NR d SO 2 R c ,
和and
所述任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-的C3-C6环烷基、苯基、4-6元杂环烷基或5-6元杂芳基部分任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、C1-C4烷基、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2和C1-C4烷氧基-(C1-C4烷氧基)-;The C 3 -C 6 cycloalkyl, phenyl, 4-6-membered heterocycloalkyl or 5-6-membered heteroaryl portion of the optionally substituted -C 1 -C 6 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 6 alkyl- , optionally substituted -C 1 -C 6 alkyl-phenyl-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(4-6-membered heterocycloalkyl)-C 1 -C 6 alkyl- or optionally substituted -C 1 -C 6 alkyl- ( 5-6 - membered heteroaryl)-C 1 -C 6 alkyl- is optionally substituted with 1 to 4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, C 1 -C 6 4 alkyl, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 and C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-;
R3和R5各自独立地为-CON(Rd)(Rf),或R3和R5之一为-CON(Rd)(Rf),且R3和R5中的另一个为H、COOH或-CO2(Rc);R 3 and R 5 are each independently -CON(R d )(R f ), or one of R 3 and R 5 is -CON(R d )(R f ), and the other of R 3 and R 5 is H, COOH or -CO 2 (R c );
R4和R6各自独立地选自H、卤素、卤代(C1-C6烷基)、卤代(C1-C6烷氧基)-、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-NH2、-NRcRc、-NRcRd、-CORc、-CO2Rc、-N(Rd)CORc、-N(Rd)SO2Rc、-N(Rg)SO2(C1-C2烷基)-N(Rh)(Rf)、-N(Rg)CO(C1-C2烷基)-N(Rh)(Rf)、任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-、任选取代的(C1-C6烷基)氨基-和任选取代的(C1-C6烷基)(C1-C4烷基)氨基-, R4 and R6 are each independently selected from H, halogen, halo( C1 - C6 alkyl), halo( C1 - C6 alkoxy)-, hydroxy, -OP(O)(OH) 2 , -OP( O )(R I R II ) 2 , -NH2 , -NRcRc, -NRcRd , -CORc , -CO2Rc , -N( Rd ) CORc , -N (Rd)SO2Rc, -N( Rg ) SO2 ( C1 - C2alkyl )-N( Rh ) ( Rf ), -N ( Rg )CO( C1 - C2alkyl )-N( Rh ) ( Rf ), optionally substituted ( C1 - C6alkyl ), optionally substituted ( C1 - C6alkyl )oxy-, optionally substituted ( C1 -C6alkyl)amino-, and optionally substituted ( C1 - C6alkyl ) -C 6 alkyl)(C 1 -C 4 alkyl)amino-,
其中所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-、任选取代的(C1-C6烷基)氨基-和任选取代的(C1-C6烷基)(C1-C4烷基)氨基-的(C1-C6烷基)任选地被1-4个各自独立地选自如下的取代基所取代:-OH、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-ORc、-NH2、-NRcRc、-NRcRd、-CO2H、-CO2Rc、-OCORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2、-SO2NRcRd、-OCONH2、-OCONRcRd、-NRdCORc、-NRdSORc、-NRdCO2Rc、-NRdSO2Rc、任选取代的苯基、任选取代的5-6元杂环烷基和任选取代的5-6元杂芳基,其中所述任选取代的苯基、5-6元杂环烷基或5-6元杂芳基任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、C1-C4烷基、卤代(C1-C4烷基)、羟基-(C1-C4烷基)-、-(C1-C4烷基)-O-P(O)(OH)2、-(C1-C4烷基)-O-P(O)(RIRII)2、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2、-C1-C4烷氧基-(C1-C4烷氧基)-、-CORd、-CON(Rd)(Rf)和-CO2Rd;wherein the optionally substituted (C 1 -C 6 alkyl), optionally substituted (C 1 -C 6 alkyl)oxy-, optionally substituted (C 1 -C 6 alkyl)amino- and optionally substituted (C 1 -C 6 alkyl)(C 1 -C 4 alkyl)amino-(C 1 -C 6 alkyl) are optionally substituted by 1 to 4 substituents each independently selected from the group consisting of -OH, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -OR c , -NH 2 , -NR c R c , -NR c R d , -CO 2 H , -CO 2 R c , -OCOR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d , -SO 2 NH 2 , -SO 2 NR c R d , -OCONH 2 2 , -OCONR c R d , -NR d COR c , -NR d SOR c , -NR d CO 2 R c , -NR d SO 2 R c , optionally substituted phenyl, optionally substituted 5-6 membered heterocycloalkyl and optionally substituted 5-6 membered heteroaryl, wherein the optionally substituted phenyl, 5-6 membered heterocycloalkyl or 5-6 membered heteroaryl is optionally substituted with 1-4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), hydroxy-(C 1 -C 4 alkyl)-, -(C 1 -C 4 alkyl)-OP(O)(OH) 2 , -(C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 , -C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-, -COR d , -CON(R d )(R f ) and -CO 2 R d ;
R14 为任选取代的C1-C4烷基、其中所述任选取代的C1-C4烷基任选地被选自如下的取代基所取代: -ORc、-NRcRd、-CO2Rc、-CONRcRd、-SO2NRcRd和-OCONRcRd;R 14 is an optionally substituted C 1 -C 4 alkyl group, wherein the optionally substituted C 1 -C 4 alkyl group is optionally substituted by a substituent selected from the group consisting of: -OR c , -NR c R d , -CO 2 R c , -CONR c R d , -SO 2 NR c R d and -OCONR c R d ;
R16为H、卤素或C1-C4烷基;R 16 is H, halogen or C 1 -C 4 alkyl;
R15和R17各自独立地为H、环丙基或C1-C4烷基;R 15 and R 17 are each independently H, cyclopropyl or C 1 -C 4 alkyl;
Ra为H、-Rc、-CORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2或-SO2NRcRd;R a is H, -R c , -COR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d , -SO 2 NH 2 or -SO 2 NR c R d ;
各Rb独立地为C1-C4烷基、卤代(C1-C4烷基)、-(C1-C4烷基)-OH、-(C1-C4烷基)-O-P(O)(OH)2、-(C1-C4烷基)-O-P(O)(RIRII)2、-(C1-C4烷基)-O-(C1-C4烷基)、-(C1-C4烷基)-N(Re)(Rf)、-(C1-C4烷基)-O-CO(C1-C4烷基)或-(C1-C4烷基)-CO-O-(C1-C4烷基);each R b is independently C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)-OH, -(C 1 -C 4 alkyl)-OP(O)(OH) 2 , -(C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , -(C 1 -C 4 alkyl)-O-(C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)-N(R e )(R f ), -(C 1 -C 4 alkyl)-O-CO(C 1 -C 4 alkyl), or -(C 1 -C 4 alkyl)-CO-O-(C 1 -C 4 alkyl);
各Rc独立地为C1-C4烷基、卤代(C1-C4烷基)、-(C1-C4烷基)-OH、-(C1-C4烷基)-O-P(O)(OH)2、-(C1-C4烷基)-O-P(O)(RIRII)2、-(C1-C4烷基)-O-(C1-C4烷基)、-(C1-C4烷基)-N(Re)(Rf)、-(C1-C4烷基)-O-CO(C1-C4烷基)、-(C1-C4烷基)-CO-O-(C1-C4烷基)、任选取代的C3-C6环烷基、任选取代的苯基、任选取代的4-6元杂环烷基、任选取代的5-6元杂芳基、任选取代的9-10元杂芳基、任选取代的-C1-C4烷基-C3-C6环烷基、任选取代的-C1-C4烷基-苯基、任选取代的-C1-C4烷基-4-6元杂环烷基、任选取代的-C1-C4烷基-5-6元杂芳基或任选取代的-C1-C4烷基-9-10元杂芳基,each R c is independently C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)-OH, -(C 1 -C 4 alkyl)-OP(O)(OH) 2 , -(C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , -(C 1 -C 4 alkyl)-O-(C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)-N(R e )(R f ), -(C 1 -C 4 alkyl)-O-CO(C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)-CO-O-(C 1 -C 4 alkyl), optionally substituted C 3 -C 6 cycloalkyl, optionally substituted phenyl, optionally substituted 4-6 membered heterocycloalkyl, optionally substituted 5-6 membered heteroaryl, optionally substituted 9-10 membered heteroaryl, optionally substituted -C 1 -C 4 alkyl-C 3 -C 6 cycloalkyl, optionally substituted -C 1 -C 4 alkyl-phenyl, optionally substituted -C 1 -C 4 alkyl-4-6 membered heterocycloalkyl, optionally substituted -C 1 -C 4 alkyl-5-6 membered heteroaryl or optionally substituted -C 1 -C 4 alkyl-9-10 membered heteroaryl,
其中所述取代的C3-C6环烷基、任选取代的苯基、任选取代的4-6元杂环烷基、任选取代的5-6元杂芳基、任选取代的9-10元杂芳基、任选取代的-C1-C4烷基-C3-C6环烷基、任选取代的-C1-C4烷基-苯基、任选取代的-C1-C4烷基-4-6元杂环烷基、任选取代的-C1-C4烷基-5-6元杂芳基或任选取代的-C1-C4烷基-9-10元杂芳基的C3-C6环烷基、苯基、4-6元杂环烷基、5-6元杂芳基或任选取代的9-10元杂芳基部分任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、-(C1-C4烷基)NH2、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、-C1-C4烷基、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2、C1-C4烷氧基-(C1-C4烷氧基)-、-CORd、-CON(Rd)(Rf)和-CO2Rd;wherein the substituted C3 - C6 cycloalkyl, optionally substituted phenyl, optionally substituted 4-6 membered heterocycloalkyl, optionally substituted 5-6 membered heteroaryl, optionally substituted 9-10 membered heteroaryl, optionally substituted -C1 - C4 alkyl- C3 - C6 cycloalkyl, optionally substituted -C1 - C4 alkyl-phenyl, optionally substituted -C1 - C4 alkyl-4-6 membered heterocycloalkyl, optionally substituted -C1 - C4 alkyl-5-6 membered heteroaryl or optionally substituted -C1- C4 alkyl-9-10 membered heteroaryl C3 -C6 cycloalkyl , phenyl, 4-6 membered heterocycloalkyl, 5-6 membered heteroaryl or optionally substituted 9-10 membered heteroaryl moiety is optionally substituted with 1-4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, -(C 1 -C 4 alkyl)NH 2 , (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, -C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 , C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-, -COR d , -CON(R d )(R f ) and -CO 2 R d ;
各Rd独立地为H或C1-C4烷基;Each R d is independently H or C 1 -C 4 alkyl;
各Re独立地为H、C1-C4烷基、-CO(C1-C4烷基)、-OCO(C1-C4烷基)、-CO2(C1-C4烷基)、-(C1-C4烷基)NH2、-(C1-C4烷基) C1-C4烷氧基、-CO-(任选取代的5-6元杂环烷基)、-CO(C1-C4烷基)-(任选取代的5-6元杂环烷基)、-CO(任选取代的5-6元杂芳基)或-CO(C1-C4烷基)-(任选取代的5-6元杂芳基),each R e is independently H, C 1 -C 4 alkyl, -CO(C 1 -C 4 alkyl), -OCO(C 1 -C 4 alkyl), -CO 2 (C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)NH 2 , -(C 1 -C 4 alkyl)C 1 -C 4 alkoxy, -CO-(optionally substituted 5-6 membered heterocycloalkyl), -CO(C 1 -C 4 alkyl)-(optionally substituted 5-6 membered heterocycloalkyl), -CO(optionally substituted 5-6 membered heteroaryl), or -CO(C 1 -C 4 alkyl)-(optionally substituted 5-6 membered heteroaryl),
其中所述任选取代的5-6元杂环烷基或任选取代的5-6元杂芳基任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、C1-C4烷基、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基) O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2、C1-C4烷氧基-(C1-C4烷氧基)-、-CORd、-CON(Rd)(Rf)和-CO2Rd;wherein the optionally substituted 5-6 membered heterocycloalkyl or optionally substituted 5-6 membered heteroaryl is optionally substituted by 1-4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 , C 1 -C 4 alkoxy-(C 1 -C 4 4 alkoxy)-, -COR d , -CON(R d )(R f ) and -CO 2 R d ;
各Rf独立地为H或C1-C4烷基;Each R f is independently H or C 1 -C 4 alkyl;
Rg和Rh各自独立地为H或C1-C4烷基,或Rg和Rh与将它们连接的一个或多个原子一起形成5-6元环; Rg and Rh are each independently H or C1 - C4 alkyl, or Rg and Rh together with the one or more atoms to which they are attached form a 5-6 membered ring;
和RI和RII 在每次出现时独立地为(C1-C6烷基)氧基-;和and R I and R II are independently at each occurrence (C 1 -C 6 alkyl)oxy-; and
RX或Ry中的至少一个独立地为C1-C4烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 4 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C4烷基;or R x and R y are both independently C 1 -C 4 alkyl;
或其互变异构体;or a tautomer thereof;
或其盐。or a salt thereof.
在整个说明书中提供的式(I)的各种基团和取代基的备选定义旨在特别地个别地描述本文公开的每种化合物种类,以及一种或多种化合物种类的群组。本发明的范围包括这些基团和取代基定义的任何组合。本发明化合物仅是考虑为如本领域技术人员所理解的“化学稳定的”那些。The alternative definitions of the various groups and substituents of formula (I) provided throughout the specification are intended to specifically describe each compound species disclosed herein individually, as well as groups of one or more compound species. The scope of the present invention includes any combination of these group and substituent definitions. The compounds of the present invention are only those considered to be "chemically stable" as understood by those skilled in the art.
本领域技术人员应理解,本发明化合物可以以其它互变异构形式(包括两性离子形式)或异构形式存在。本文所述的化学式和化合物的所有互变异构(包括两性离子形式)和异构形式均旨在涵盖于本发明的范围内。It will be appreciated by those skilled in the art that the compounds of the present invention may exist in other tautomeric forms (including zwitterionic forms) or isomeric forms. All tautomeric (including zwitterionic forms) and isomeric forms of the chemical formulae and compounds described herein are intended to be encompassed within the scope of the present invention.
本领域技术人员还应理解,本发明化合物可以以互变异构(或同分异构)形式(包括但不限于式(A)、式(B)和/或式(C))或两性离子形式(包括但不限于式(D)或式(E))存在。在式(B)、(C)、(D)或(E)中,R每次出现时独立地为H或氮上任何适当的取代基,例如烷基。Those skilled in the art will also appreciate that the compounds of the present invention may exist in tautomeric (or isomeric) forms (including but not limited to Formula (A), Formula (B) and/or Formula (C)) or zwitterionic forms (including but not limited to Formula (D) or Formula (E)). In Formula (B), (C), (D) or (E), each occurrence of R is independently H or any suitable substituent on nitrogen, such as an alkyl group.
为本文所述的本发明的中间体化合物和/或化合物提供的化学名称可以指这样的化合物的任何一种互变异构/同分异构表示(在一些情况下,这样的替代名称随实验提供)。应理解,对所命名的化合物(本发明的中间体化合物或化合物)或在结构上进行描绘的化合物(本发明的中间体化合物或化合物)的任何提及旨在涵盖这样的化合物的所有互变异构/同分异构形式,包括两性离子形式,及其任何混合物。The chemical names provided for the intermediate compounds and/or compounds of the invention described herein may refer to any tautomeric/isomeric representation of such compounds (in some cases, such alternative names are provided by the experiments). It should be understood that any reference to a named compound (an intermediate compound or compound of the invention) or a structurally depicted compound (an intermediate compound or compound of the invention) is intended to encompass all tautomeric/isomeric forms of such compounds, including zwitterionic forms, and any mixtures thereof.
如本文所用,术语“烷基”代表具有指定数目的碳原子的饱和、直链或支链烃基。术语"C1-C4烷基"指含有1至4个碳原子的直链或支链烷基部分。示例性烷基包括但不限于甲基、乙基、正丙基、异丙基、正丁基、异丁基、仲丁基、叔丁基、戊基和己基。As used herein, the term "alkyl" represents a saturated, straight or branched hydrocarbon group having a specified number of carbon atoms. The term " C1 - C4 alkyl" refers to a straight or branched alkyl moiety containing 1 to 4 carbon atoms. Exemplary alkyl groups include, but are not limited to, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, pentyl, and hexyl.
当取代基术语诸如"烷基"与另一取代基术语组合使用时,例如如在“羟基(C1-C4烷基)”中,连接取代基术语(例如,烷基)旨在涵盖二价部分,其中连接点是通过那个连接取代基。“羟基(C1-C4烷基)”基团的实例包括但不限于羟基甲基、羟基乙基和羟基异丙基。When a substituent term such as "alkyl" is used in combination with another substituent term, for example as in "hydroxy(C 1 -C 4 alkyl)", the linking substituent term (e.g., alkyl) is intended to encompass a divalent moiety where the point of attachment is through that linking substituent. Examples of "hydroxy(C 1 -C 4 alkyl)" groups include, but are not limited to, hydroxymethyl, hydroxyethyl, and hydroxyisopropyl.
如本文所用,术语“卤代(烷基)”代表具有指定数目(n)的碳原子和一个或多个(最多达2n + 1个)卤素原子的饱和、直链或支链烃基。例如,术语“卤代(C1-C4)烷基”代表在含有1至4个碳原子的烷基部分的一个或多个碳原子处具有一个或多个可以相同或不同的卤素原子的基团。“卤代(C1-C4烷基)”基团的实例包括但不限于-CF3(三氟甲基)、-CCl3(三氯甲基)、1,1-二氟乙基、2,2,2-三氟乙基和六氟异丙基。As used herein, the term "halo(alkyl)" represents a saturated, straight-chain or branched hydrocarbon radical having a specified number (n) of carbon atoms and one or more (up to 2n + 1) halogen atoms. For example, the term "halo(C 1 -C 4 )alkyl" represents a radical having one or more halogen atoms, which may be the same or different, at one or more carbon atoms of an alkyl moiety containing 1 to 4 carbon atoms. Examples of "halo(C 1 -C 4 alkyl)" radicals include, but are not limited to, -CF 3 (trifluoromethyl), -CCl 3 (trichloromethyl), 1,1-difluoroethyl, 2,2,2-trifluoroethyl and hexafluoroisopropyl.
"烯基"指具有指定数目的碳原子和至少1个和最多达3个碳-碳双键的直链或支链烃基。实例包括乙烯基和丙烯基。"Alkenyl" refers to a straight or branched chain hydrocarbon group having the specified number of carbon atoms and at least 1 and up to 3 carbon-carbon double bonds. Examples include ethenyl and propenyl.
“炔基”指具有指定数目的碳原子和至少1个且最多达3个碳-碳三键的直链或支链烃基。实例包括乙炔基和丙炔基。"Alkynyl" refers to a straight or branched chain hydrocarbon group having the specified number of carbon atoms and at least 1 and up to 3 carbon-carbon triple bonds. Examples include ethynyl and propynyl.
"烷氧基-”或"(烷基)氧基-”指"烷基-氧基-”基团,其含有通过氧连接原子结合的具有指定数目的碳原子的烷基部分。例如,术语"C1-C4烷氧基-”代表通过氧连接原子结合的具有至少1个和最多达4个碳原子的饱和、直链或支链烃部分。示例性"C1-C4烷氧基-"或"(C1-C4烷基)氧基-"基团包括但不限于甲氧基、乙氧基、正丙氧基、异丙氧基、正丁氧基、仲丁氧基和叔丁氧基。"Alkoxy-" or "(alkyl)oxy-" refers to an "alkyl-oxy-" group containing an alkyl moiety having the specified number of carbon atoms bonded through an oxygen linking atom. For example, the term "C 1 -C 4 alkoxy-" represents a saturated, straight-chain or branched hydrocarbon moiety having at least 1 and up to 4 carbon atoms bonded through an oxygen linking atom. Exemplary "C 1 -C 4 alkoxy-" or "(C 1 -C 4 alkyl)oxy-" groups include, but are not limited to, methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, sec-butoxy, and tert-butoxy.
如本文所用,术语“卤代(烷氧基)-”代表通过氧连接原子结合的具有指定数目(n)的碳原子和一个或多个(最多达2n+1个)卤素原子的饱和、直链或支链烃基。例如,术语“卤代(C1-C4烷氧基)-”指“卤代烷基-氧基-”基团,其含有通过氧连接原子结合的“卤代(C1-C4烷基)”部分。示例性“卤代(C1-C4烷氧基)-”基团包括但不限于-OCHF2(二氟甲氧基)、-OCF3(三氟甲氧基)、-OCH2CF3(三氟乙氧基)和-OCH(CF3)2(六氟异丙氧基)。As used herein, the term "halo(alkoxy)-" represents a saturated, straight-chain or branched hydrocarbon radical having a specified number (n) of carbon atoms and one or more (up to 2n+1) halogen atoms bonded through an oxygen linking atom. For example, the term "halo(C 1 -C 4 alkoxy)-" refers to a "haloalkyl-oxy-" radical containing a "halo(C 1 -C 4 alkyl)" moiety bonded through an oxygen linking atom. Exemplary "halo(C 1 -C 4 alkoxy)-" radicals include, but are not limited to, -OCHF 2 (difluoromethoxy), -OCF 3 (trifluoromethoxy), -OCH 2 CF 3 (trifluoroethoxy), and -OCH(CF 3 ) 2 (hexafluoroisopropoxy).
碳环基团或部分是其中环成员是碳原子的环状基团或部分,其可以是饱和的、部分不饱和的(非芳族的)或完全不饱和的(芳族的)。A carbocyclic group or moiety is a cyclic group or moiety in which the ring members are carbon atoms, which may be saturated, partially unsaturated (non-aromatic) or fully unsaturated (aromatic).
“环烷基”指环中含有指定数目的碳原子的非芳族、饱和、烃环基团。例如,术语“C3-C6环烷基”指具有三至六个环碳原子的环状基团。示例性“C3-C6环烷基”基团包括环丙基、环丁基、环戊基和环己基。"Cycloalkyl" refers to a non-aromatic, saturated, hydrocarbon ring group containing the specified number of carbon atoms in the ring. For example, the term "C 3 -C 6 cycloalkyl" refers to a cyclic group having three to six ring carbon atoms. Exemplary "C 3 -C 6 cycloalkyl" groups include cyclopropyl, cyclobutyl, cyclopentyl, and cyclohexyl.
杂环基团或部分是具有至少两种不同元素的原子作为环成员的环状基团或部分,所述环状基团或部分可以是饱和的、部分不饱和的(非芳族的)或完全不饱和的(芳族的)。A heterocyclic group or moiety is a cyclic group or moiety having atoms of at least two different elements as ring members, which cyclic group or moiety may be saturated, partially unsaturated (non-aromatic) or fully unsaturated (aromatic).
“杂原子”指氮、硫或氧原子,例如氮原子或氧原子。"Heteroatom" refers to a nitrogen, sulfur or oxygen atom, for example a nitrogen atom or an oxygen atom.
"杂环烷基"指含有3-10个环原子且含有一个或多个(通常一个或两个)独立地选自氧、硫和氮的杂原子环成员的非芳族、单环或二环基团。杂环烷基基团的连接点可以是任何合适的碳或氮原子。"Heterocycloalkyl" refers to a non-aromatic, monocyclic or bicyclic group containing 3-10 ring atoms and containing one or more (usually one or two) heteroatom ring members independently selected from oxygen, sulfur and nitrogen. The point of attachment of the heterocycloalkyl group can be any suitable carbon or nitrogen atom.
“杂环烷基”基团的实例包括但不限于氮杂环丙烷基、硫杂环丙烷基(thiranyl)、环氧乙烷基、氮杂环丁烷基、氧杂环丁烷基、硫杂环丁烷基、吡咯烷基、四氢呋喃基、四氢噻吩基、1,3-二氧杂环戊烷基、哌啶基、哌嗪基、四氢吡喃基、二氢吡喃基、四氢硫代吡喃基、1,3-二氧杂环己烷基、1,4-二氧杂环己烷基、1,3-氧杂硫杂环戊烷基、1,3-氧杂硫杂环己烷基、1,3-二硫杂环己烷基、1,4-氧杂硫杂环戊烷基、1,4-氧杂硫杂环己烷基、1,4-二硫杂环己烷基、吗啉基、硫代吗啉基和六氢-1H-1,4-二氮杂䓬基。Examples of “heterocycloalkyl” groups include, but are not limited to, aziridine, thiirane, oxirane, azetidinyl, oxetanyl, thietanyl, pyrrolidinyl, tetrahydrofuranyl, tetrahydrothiophenyl, 1,3-dioxolanyl, piperidinyl, piperazinyl, tetrahydropyranyl, dihydropyranyl, tetrahydrothiopyranyl, 1,3-dioxanyl, 1,4-dioxanyl, 1,3-oxathiolanyl, 1,3-dithiolanyl, 1,4-oxathiolanyl, 1,4-dithiolanyl, morpholinyl, thiomorpholinyl, and hexahydro- 1H -1,4-diazepinyl.
“4-元杂环烷基”基团的实例包括氧杂环丁烷基、硫杂环丁烷基和氮杂环丁烷基。Examples of "4-membered heterocycloalkyl" groups include oxetanyl, thietanyl and azetidinyl.
术语"5-6元杂环烷基"代表含有5或6个环原子的饱和、单环基团,其包括一个或两个独立地选自氧、硫和氮的杂原子。5至6元杂环烷基基团的说明性实例包括但不限于吡咯烷基、四氢呋喃基、四氢噻吩基、四氢吡喃基、四氢噻喃基、哌啶基、哌嗪基、吗啉基和硫代吗啉基。The term "5-6 membered heterocycloalkyl" represents a saturated, monocyclic group containing 5 or 6 ring atoms, including one or two heteroatoms independently selected from oxygen, sulfur and nitrogen. Illustrative examples of 5 to 6 membered heterocycloalkyl groups include, but are not limited to, pyrrolidinyl, tetrahydrofuranyl, tetrahydrothienyl, tetrahydropyranyl, tetrahydrothiopyranyl, piperidinyl, piperazinyl, morpholinyl and thiomorpholinyl.
"杂芳基"指含有5至10个环原子的芳族的单环或双环基团,其包括1至4个独立地选自氮、氧和硫的杂原子,其中所述基团的至少一部分是芳族的。例如,该术语涵盖含有与杂环部分稠合的苯基环或与碳环部分稠合的杂芳基环部分的双环杂环-芳基基团。杂芳基基团的结合点可以是任何合适的碳或氮原子。"Heteroaryl" refers to an aromatic monocyclic or bicyclic group containing 5 to 10 ring atoms, including 1 to 4 heteroatoms independently selected from nitrogen, oxygen and sulfur, wherein at least a portion of the group is aromatic. For example, the term encompasses a bicyclic heterocyclic-aryl group containing a phenyl ring fused to a heterocyclic portion or a heteroaryl ring portion fused to a carbocyclic portion. The point of attachment of the heteroaryl group may be any suitable carbon or nitrogen atom.
术语"5-6元杂芳基"代表含有5或6个环原子的芳族的单环基团,其包括至少一个碳原子和1至4个独立地选自氮、氧和硫的杂原子。所选的5-元杂芳基基团含有一个氮、氧或硫环杂原子,并且任选地含有1、2或3个额外氮环原子。所选的6-元杂芳基基团含有1、2或3个氮环杂原子。5-元杂芳基基团的实例包括呋喃基(furyl)(呋喃基(furanyl))、噻吩基、吡咯基、咪唑基、吡唑基、三唑基、四唑基、噻唑基、异噻唑基、噻二唑基、噁唑基、异噁唑基和噁二唑基。所选的6-元杂芳基基团包括吡啶基(pyridinyl)(吡啶基(pyridyl))、吡嗪基、嘧啶基、哒嗪基和三嗪基。The term "5-6 yuan heteroaryl" represents an aromatic monocyclic group containing 5 or 6 ring atoms, which includes at least one carbon atom and 1 to 4 heteroatoms independently selected from nitrogen, oxygen and sulfur. The selected 5-yuan heteroaryl group contains a nitrogen, oxygen or sulfur ring heteroatom, and optionally contains 1,2 or 3 additional nitrogen ring atoms. The selected 6-yuan heteroaryl group contains 1,2 or 3 nitrogen ring heteroatoms. The example of the 5-yuan heteroaryl group includes furyl (furyl) (furanyl (furanyl)), thienyl, pyrrolyl, imidazoles, pyrazolyl, triazolyl, tetrazolyl, thiazolyl, isothiazolyl, thiadiazolyl, oxazolyl, isoxazolyl and oxadiazolyl. The selected 6-yuan heteroaryl group includes pyridyl (pyridinyl) (pyridyl (pyridyl)), pyrazinyl, pyrimidinyl, pyridazinyl and triazinyl.
术语“9-10元杂芳基”指含有9或10个环原子(包括1至4个独立地选自氮、氧和硫的杂原子)的芳族双环基团。9-元杂芳基(6,5-稠合杂芳基)的实例包括苯并噻吩基、苯并呋喃基、吲哚基、吲哚啉基(二氢吲哚基)、异吲哚基、异吲哚啉基、吲唑基、异苯并呋喃基、2,3-二氢苯并呋喃基、苯并噁唑基、苯并异噁唑基、苯并噻唑基、苯并异噻唑基、苯并咪唑基、苯并噁二唑基、苯并噻二唑基、苯并三唑基、嘌呤基、咪唑并吡啶基、吡唑并吡啶基、三唑并吡啶基和1,3-苯并二氧杂环戊烯基。The term "9-10 membered heteroaryl" refers to an aromatic bicyclic group containing 9 or 10 ring atoms, including 1 to 4 heteroatoms independently selected from nitrogen, oxygen and sulfur. Examples of 9-membered heteroaryl (6,5-fused heteroaryl) include benzothiophenyl, benzofuranyl, indolyl, indolinyl (dihydroindolinyl), isoindolyl, isoindolyl, indazolyl, isobenzofuranyl, 2,3-dihydrobenzofuranyl, benzoxazolyl, benzisoxazolyl, benzothiazolyl, benzisothiazolyl, benzimidazolyl, benzoxadiazolyl, benzothiadiazolyl, benzotriazolyl, purinyl, imidazopyridyl, pyrazolopyridyl, triazolopyridyl and 1,3-benzodioxolyl.
10-元杂芳基(6,6-稠合杂芳基)基团的实例包括喹啉基(quinolinyl)(喹啉基(quinolyl))、异喹啉基、酞嗪基、萘啶基(1,5-萘啶基、1,6-萘啶基、1,7-萘啶基、1,8-萘啶基)、喹唑啉基、喹喔啉基、4H-喹嗪基、1,2,3,4-四氢喹啉基(四氢喹啉基)、1,2,3,4-四氢异喹啉基 (四氢异喹啉基)、噌啉基、喋啶基和2,3-二氢苯并[b][1,4]二氧杂环己烯基。Examples of 10-membered heteroaryl (6,6-fused heteroaryl) groups include quinolinyl (quinolyl), isoquinolinyl, phthalazinyl, naphthyridinyl (1,5-naphthyridinyl, 1,6-naphthyridinyl, 1,7-naphthyridinyl, 1,8-naphthyridinyl), quinazolinyl, quinoxalinyl, 4H-quinolizinyl, 1,2,3,4-tetrahydroquinolinyl (tetrahydroquinolinyl), 1,2,3,4-tetrahydroisoquinolinyl (tetrahydroisoquinolinyl), cinnolinyl, pteridinyl, and 2,3-dihydrobenzo[b][1,4]dioxinyl.
术语“卤素”和“卤代”指卤素基团,例如氟、氯、溴或碘取代基。The terms "halogen" and "halo" refer to a halogen radical, such as a fluorine, chlorine, bromine or iodine substituent.
“氧代”代表双键的氧部分;例如,如果直接结合碳原子则形成羰基基团(C=O)。"Oxo" represents the oxygen portion of a double bond; for example, if directly bound to a carbon atom it would form a carbonyl group (C=O).
"羟基"(“hydroxy”或“hydroxyl”)旨在意指基团-OH。"Hydroxy" or "hydroxyl" is intended to mean the group -OH.
如本文所用,术语“氰基”指腈基团,-C≡N。As used herein, the term "cyano" refers to a nitrile group, -C≡N.
如本文所用,术语“任选地取代的”表示基团(诸如烷基、环烷基、烷氧基、杂环烷基、芳基或杂芳基基团)或环或部分可不被取代,或所述基团、环或部分可被如本文提供的取代基定义(A、R3等)中所定义的一个或多个取代基取代。在其中基团可选自许多替代基团的情况下,所选的基团可以是相同或不同的。As used herein, the term "optionally substituted" means that a group (such as an alkyl, cycloalkyl, alkoxy, heterocycloalkyl, aryl or heteroaryl group) or a ring or a moiety may not be substituted, or the group, ring or moiety may be substituted with one or more substituents as defined in the substituent definitions (A, R3, etc.) provided herein. In the case where a group can be selected from a number of alternative groups, the selected groups may be the same or different.
术语“独立地”意味着在多于一个取代基选自许多可能的取代基时,那些取代基可以是相同或不同的。The term "independently" means that when more than one substituent is selected from a number of possible substituents, those substituents may be the same or different.
术语“药学上可接受的”指在合理的医学判断范围内适合用于与人和动物的组织接触而没有过度毒性、刺激性或其它问题或并发症、与合理的收益/风险比相称的那些化合物、材料、组合物和剂型。The term "pharmaceutically acceptable" refers to those compounds, materials, compositions and dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of humans and animals without excessive toxicity, irritation, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
如本文所用,术语“本发明化合物”(“compound(s) of the invention”或“compound(s) of this invention”)意指如本文所定义的式(I)化合物,其呈任何形式,即,任何互变异构/同分异构形式、任何盐或非盐形式(例如作为游离酸或碱形式,或作为盐,特别是其药学上可接受的盐)以及其任何物理形式(例如包括非固体形式(例如,液体或半固体形式),以及固体形式(例如无定形或晶体形式、特定多晶形式、溶剂化物形式(包括水合物形式(例如单水合物、 二水合物和半水合物))),以及各种形式的混合物。As used herein, the term "compound(s) of the invention" or "compound(s) of this invention" means a compound of formula (I) as defined herein, in any form, i.e. any tautomeric/isomerized form, any salt or non-salt form (e.g. as a free acid or base form, or as a salt, in particular a pharmaceutically acceptable salt thereof), and in any physical form thereof (e.g. including non-solid forms (e.g. liquid or semi-solid forms), as well as solid forms (e.g. amorphous or crystalline forms, specific polymorphic forms, solvate forms (including hydrate forms (e.g. monohydrates, dihydrates and hemihydrates))), as well as mixtures of various forms.
因此,包括在本发明内的是如本文所定义的式(I)化合物,其呈任何盐或非盐形式及其任何物理形式,以及各种形式的混合物。尽管这样被包括在本发明内,但应理解,呈其任何盐或非盐形式以及任何物理形式的如本文所定义的式(I)化合物可以出于配制目的而具有不同的活性水平、不同生物利用度和不同的处理特性。Therefore, included in the present invention are compounds of formula (I) as defined herein, in any salt or non-salt form and any physical form thereof, as well as mixtures of various forms. Although so included in the present invention, it is understood that compounds of formula (I) as defined herein, in any salt or non-salt form thereof and in any physical form, may have different activity levels, different bioavailabilities and different handling characteristics for formulation purposes.
在本发明化合物的一个实施方案中,R3和R5各自独立地为-CON(Rd)(Rf),或R3和R5之一为-CON(Rd)(Rf),且R3和R5中的另一个为H或-CO2(Rc)。在一个实施方案中,R3和R5各自独立地为-CON(Rd)(Rf)。在另一个实施方案中,R3和R5之一为-CON(Rd)(Rf)且R3和R5中的另一个为H。在一个特定的实施方案中,R3和R5各自为-CONH2。In one embodiment of the compounds of the invention, R3 and R5 are each independently -CON( Rd )( Rf ), or one of R3 and R5 is -CON( Rd )( Rf ), and the other of R3 and R5 is H or -CO2 ( Rc ). In one embodiment, R3 and R5 are each independently -CON( Rd )( Rf ). In another embodiment, one of R3 and R5 is -CON( Rd )( Rf ) and the other of R3 and R5 is H. In a specific embodiment, R3 and R5 are each -CONH2 .
应理解当q为0时,A不存在且RA1和RA2不连接。类似地,应理解当r为0时,B不存在且RB1和RB2不连接。类似地,应理解当s为0时,C不存在且RC1和RC2不连接。It is understood that when q is 0, A does not exist and RA1 and RA2 are not connected. Similarly, it is understood that when r is 0, B does not exist and RB1 and RB2 are not connected. Similarly, it is understood that when s is 0, C does not exist and RC1 and RC2 are not connected.
在本发明化合物的一个实施方案中,q为1,r为0和s为0 (q+r+s=1),并且该化合物具有式(I-A)或(I-a):In one embodiment of the compound of the present invention, q is 1, r is 0 and s is 0 (q+r+s=1), and the compound has formula (I-A) or (I-a):
。 .
在本发明化合物的一个实施方案中,q为0,r为1和s为0 (q+r+s=1),并且该化合物具有式(I-B)或(I-b):In one embodiment of the compounds of the present invention, q is 0, r is 1 and s is 0 (q+r+s=1), and the compound has formula (I-B) or (I-b):
。 .
在本发明化合物的一个实施方案中,q为0,r为0和s为1 (q+r+s=1),并且该化合物具有式(I-C)或(I-c):In one embodiment of the compound of the present invention, q is 0, r is 0 and s is 1 (q+r+s=1), and the compound has formula (I-C) or (I-c):
。 .
在本发明化合物的一个实施方案中,q为1,r为1和s为0 (q+r+s=2),并且该化合物具有式(I-AB)或(I-ab):In one embodiment of the compounds of the present invention, q is 1, r is 1 and s is 0 (q+r+s=2), and the compound has the formula (I-AB) or (I-ab):
。 .
在本发明化合物的一个实施方案中,q为1,r为0和s为1 (q+r+s=2),并且该化合物具有式(I-AC)或(I-ac):In one embodiment of the compounds of the present invention, q is 1, r is 0 and s is 1 (q+r+s=2), and the compound has the formula (I-AC) or (I-ac):
。 .
在本发明化合物的一个实施方案中,q为0,r为1和s为1 (q+r+s=2),并且该化合物具有式(I-BC)或(I-bc):In one embodiment of the compounds of the present invention, q is 0, r is 1 and s is 1 (q+r+s=2), and the compound has the formula (I-BC) or (I-bc):
。 .
在本发明化合物的一个实施方案中,q为0 ,并且RA1和RA2各自独立地为H、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-N(Re)(Rf)、-CO2Rf、-N(Rf)CORb、-N(Rg)SO2(C1-C4烷基)-N(Re)(Rf)、-N(Rg)CO(C1-C4烷基)-N(Rh)(Rf)、任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-、任选取代的(C1-C6烷基)氨基-和任选取代的(C1-C6烷基)(C1-C4烷基)氨基-,In one embodiment of the compounds of the present invention, q is 0, and RA1 and RA2 are each independently H, halogen, hydroxy, -OP( O )(OH) 2 , -OP ( O)( RIRII ) 2 , -N( Re ) ( Rf ) , -CO2Rf, -N( Rf )CORb, -N( Rg ) SO2 ( C1 -C4alkyl)-N(Re)( Rf ), -N ( Rg )CO( C1 - C4alkyl )-N( Rh )( Rf ), optionally substituted ( C1 - C6alkyl ), optionally substituted ( C1 - C6alkyl )oxy-, optionally substituted ( C1 - C6alkyl )amino-, and optionally substituted ( C1 - C6alkyl )( C1 - C4alkyl )amino-,
其中所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-、任选取代的(C1-C6烷基)氨基-和任选取代的(C1-C6烷基)(C1-C4烷基)氨基-的(C1-C6烷基)任选地被1-4个各自独立地选自如下的取代基所取代:羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、C1-C4烷氧基-、-N(Re)(Rf)、-CO2(Rf)、-CON(Re)(Rf)、任选取代的苯基、任选取代的5-6元杂环烷基和任选取代的5-6元杂芳基,其中所述任选取代的苯基、5-6元杂环烷基或5-6元杂芳基任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C6烷基)氨基-、(C1-C6烷基)(C1-C6烷基)氨基-、-(C1-C6烷基)-NH2、卤代(C1-C6烷基)、羟基-(C1-C4烷基)-、-(C1-C4烷基)-O-P(O)(OH)2、-(C1-C4烷基)-O-P(O)(RIRII)2、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2和C1-C4烷氧基-(C1-C4烷氧基)-。wherein the optionally substituted (C 1 -C 6 alkyl), optionally substituted (C 1 -C 6 alkyl)oxy-, optionally substituted (C 1 -C 6 alkyl)amino- and optionally substituted (C 1 -C 6 alkyl)(C 1 -C 4 alkyl)amino-(C 1 -C 6 alkyl) are optionally substituted by 1 to 4 substituents each independently selected from the group consisting of hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , C 1 -C 4 alkoxy-, -N( Re )(R f ), -CO 2 (R f ), -CON( Re )(R f ), optionally substituted phenyl, optionally substituted 5-6 membered heterocycloalkyl and optionally substituted 5-6 membered heteroaryl, wherein the optionally substituted phenyl, 5-6 membered heterocycloalkyl or 5-6 membered heteroaryl is optionally substituted by 1-4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 6 alkyl)amino-, (C 1 -C 6 alkyl)(C 1 -C 6 alkyl)amino-, -(C 1 -C 6 alkyl)-NH2, halo(C 1 -C 6 alkyl), hydroxy-(C 1 -C 4 alkyl)-, -(C 1 -C 4 alkyl)-OP(O)(OH) 2 , - (C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 and C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-.
在本发明化合物的一个实施方案中,q为0并且RA1和RA2各自独立地为H、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、(C1-C4烷基)、羟基(C1-C4烷基)-、氨基(C1-C4烷基)-、(C1-C4烷基)氨基(C1-C4烷基)-、(C1-C4烷基)(C1-C4烷基)氨基(C1-C4烷基)-、C1-C4烷氧基-、羟基(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2、氨基(C2-C4烷氧基)-、(C1-C4烷基)氨基(C2-C4烷氧基)-、(C1-C4烷基)(C1-C4烷基)氨基(C2-C4烷氧基)-、6-元杂环烷基-(C1-C4烷基)-、苯基(C1-C4烷氧基)-、(C1-C4烷基)OCONH(C1-C4烷基)-、羟基(C1-C4烷基)氨基-、-氨基(C1-C4烷基)-O-P(O)(OH)2、-氨基(C1-C4烷基)-O-P(O)(RIRII)2、(C1-C4烷基)CONH-、(C1-C4烷基)CON(C1-C4烷基)-、-CO2H、-CO2(C1-C4烷基)、氨基(C1-C4烷基)CONH-、(C1-C4烷基)氨基(C1-C4烷基)CONH-、(C1-C4烷基)(C1-C4烷基)氨基(C1-C4烷基)CONH-、氨基(C1-C4烷基)CON(C1-C4烷基)-、(C1-C4烷基)氨基(C1-C4烷基)CON(C1-C4烷基)-、羟基(C1-C4烷基)CONH-、-NHCO(C1-C4烷基)-O-P(O)(OH)2、-NHCO(C1-C4烷基)-O-P(O)(RIRII)2、(C1-C4烷基)(C1-C4烷基)氨基(C1-C4烷基)CON(C1-C4烷基)-、羟基(C1-C4烷基)CON(C1-C4烷基)-、-(C1-C4烷基)NCO(C1-C4烷基)-O-P(O)(OH)2、-(C1-C4烷基)NCO(C1-C4烷基)-O-P(O)(RIRII)2、 HO2C(C1-C4烷氧基)-、(C1-C4烷基)OCO(C1-C4烷氧基)-、H2NCO(C1-C4烷氧基)-、(C1-C4烷基)HNCO(C1-C4烷氧基)-、(C1-C4烷基)(C1-C4烷基)NCO(C1-C4烷氧基)-和-NHSO2(C1-C4烷基)。In one embodiment of the compounds of the present invention, q is 0 and RA1 and RA2 are each independently H, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl), hydroxy(C 1 -C 4 alkyl)-, amino(C 1 -C 4 alkyl)-, (C 1 -C 4 alkyl)amino(C 1 -C 4 alkyl)-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino(C 1 -C 4 alkyl)-, C 1 -C 4 alkoxy-, hydroxy(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II) R II ) 2 , amino(C 2 -C 4 alkoxy)-, (C 1 -C 4 alkyl)amino(C 2 -C 4 alkoxy)-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino(C 2 -C 4 alkoxy)-, 6-membered heterocycloalkyl-(C 1 -C 4 alkyl)-, phenyl(C 1 -C 4 alkoxy)-, (C 1 -C 4 alkyl)OCONH(C 1 -C 4 alkyl)-, hydroxy(C 1 -C 4 alkyl)amino-, -amino(C 1 -C 4 alkyl)-OP(O)(OH) 2 , -amino(C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , (C 1 -C 4 alkyl)CONH-, (C 1 -C 4 alkyl)CON(C 1 -C 4 alkyl)-, -CO 2 H, -CO 2 (C 1 -C 4 alkyl), amino (C 1 -C 4 alkyl) CONH-, (C 1 -C 4 alkyl) amino (C 1 -C 4 alkyl) CONH-, amino (C 1 -C 4 alkyl) CON (C 1 -C 4 alkyl) -, (C 1 -C 4 alkyl) amino (C 1 -C 4 alkyl) CONH-, amino (C 1 -C 4 alkyl) CON (C 1 -C 4 alkyl) -, (C 1 -C 4 alkyl) amino (C 1 -C 4 alkyl) CON (C 1 -C 4 alkyl) -, hydroxy (C 1 -C 4 alkyl) CONH-, -NHCO (C 1 -C 4 alkyl) -OP (O) (OH) 2 , -NHCO (C 1 -C 4 alkyl) -OP (O) (R I R II ) 2 , (C 1 -C 4 alkyl) (C 1 -C 4 alkyl) amino (C 1 -C 4 alkyl) CON (C 1 -C 4 alkyl)-, hydroxy(C 1 -C 4 alkyl)CON(C 1 -C 4 alkyl)-, -(C 1 -C 4 alkyl)NCO(C 1 -C 4 alkyl)-OP(O)(OH) 2 , -(C 1 -C 4 alkyl)NCO(C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , HO 2 C(C 1 -C 4 alkoxy)-, (C 1 -C 4 alkyl)OCO(C 1 -C 4 alkoxy)-, H 2 NCO(C 1 -C 4 alkoxy)-, (C 1 -C 4 alkyl)HNCO(C 1 -C 4 alkoxy)-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)NCO(C 1 -C 4 alkoxy)-, and -NHSO 2 (C 1 -C 4 alkyl).
在一个实施方案中,q为0且RA1和RA2各自独立地为H、(C1-C6烷基)氧基-或羟基(C2-C6烷基)氧基-。在一个实施方案中,q为0且RA1和RA2各自独立地为H、(C1-C6烷基)氧基-、羟基(C2-C6烷基)氧基-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2。在一个实施方案中,q为0且RA1和RA2各自为H。在选定的实施方案中,q为0且RA1和RA2独立地选自 H、-OCH2CH2CH2OH和-OCH3。In one embodiment, q is 0 and RA1 and RA2 are each independently H, (C 1 -C 6 alkyl)oxy- or hydroxy(C 2 -C 6 alkyl)oxy-. In one embodiment, q is 0 and RA1 and RA2 are each independently H, (C 1 -C 6 alkyl)oxy-, hydroxy(C 2 -C 6 alkyl)oxy-, -(C 2 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 . In one embodiment, q is 0 and RA1 and RA2 are each H. In selected embodiments, q is 0 and RA1 and RA2 are independently selected from H, -OCH 2 CH 2 CH 2 OH and -OCH 3 .
在一个实施方案中,q为0且RA2和RA1各自独立地为H、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-,其中所述任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、C1-C4烷氧基、-N(Re)(Rf)、-COOH、任选取代的苯基和任选取代的5-6元杂环烷基,且各Re 独立地选自H、C1-C4烷基、-CO(C1-C4烷基)、-OCO(C1-C4烷基)、-(C1-C4烷基)NH2、-(C1-C4烷基) C1-C4烷氧基或-CO2(C1-C4烷基)。In one embodiment, q is 0 and RA2 and RA1 are each independently H, optionally substituted ( C1 - C6 alkyl) or optionally substituted ( C1 - C6 alkyl)oxy-, wherein the C1- C6 alkyl of the optionally substituted ( C1 - C6 alkyl) or optionally substituted ( C1 - C6 alkyl)oxy- is optionally substituted with 1-4 substituents each independently selected from the group consisting of hydroxy, -OP(O)(OH) 2 , -OP(O)( R1RII ) 2 , C1 - C4 alkoxy, -N( Re )( Rf ), -COOH, optionally substituted phenyl and optionally substituted 5-6 membered heterocycloalkyl, and each Re is independently selected from the group consisting of H, C1 - C4 alkyl, -CO( C1 - C4 alkyl), -OCO( C1 - C4 alkyl), -( C1 - C4 alkyl) NH2 , -( C1 - C4 alkyl) C 1 -C 4 alkoxy or -CO 2 (C 1 -C 4 alkyl).
在一个实施方案中,q为0且RA2和RA1各自独立地为H、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-,和所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧In one embodiment, q is 0 and R A2 and R A1 are each independently H, optionally substituted (C 1 -C 6 alkyl) or optionally substituted (C 1 -C 6 alkyl)oxy-, and the optionally substituted (C 1 -C 6 alkyl), optionally substituted (C 1 -C 6 alkyl)oxy
基-的C1-C6烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-N(Re)(Rf)、C1-C4烷氧基、苯基和含有至少一个氮或氧作为环成员的任选取代的5-6元杂环烷基,和各Re 各自独立地选自H、C1-C4烷基、-(C1-C4烷基)NH2或-(C1-C4烷基)C1-C4烷氧基。The C 1 -C 6 alkyl group is optionally substituted with 1 to 4 substituents each independently selected from the group consisting of hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -N( Re )( Rf ), C 1 -C 4 alkoxy, phenyl, and an optionally substituted 5-6 membered heterocycloalkyl group containing at least one nitrogen or oxygen as a ring member, and each Re is each independently selected from the group consisting of H, C 1 -C 4 alkyl, -(C 1 -C 4 alkyl)NH 2 or -(C 1 -C 4 alkyl)C 1 -C 4 alkoxy.
在一个实施方案中,q为0且RA2或RA1中的至少一个各自独立地为H、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-,和所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1-4个各自独立地选自如下的取代基所取代:-N(Re)(Rf)、四氢吡喃、吡咯烷基、哌嗪基、哌啶基和吗啉基,和各Re 各自独立地选自H、C1-C4烷基、-(C1-C4烷基)NH2或-(C1-C4烷基)C1-C4烷氧基。In one embodiment, q is 0 and at least one of RA2 or RA1 is each independently H, optionally substituted ( C1 - C6 alkyl) or optionally substituted ( C1 - C6 alkyl)oxy-, and the C1-C6 alkyl of the optionally substituted ( C1 - C6 alkyl) or optionally substituted ( C1 - C6 alkyl)oxy- is optionally substituted with 1-4 substituents each independently selected from the group consisting of -N ( Re )( Rf ), tetrahydropyran, pyrrolidinyl, piperazinyl, piperidinyl and morpholinyl, and each Re is each independently selected from H, C1 - C4 alkyl, -( C1 - C4 alkyl) NH2 or -( C1 - C4 alkyl) C1 - C4 alkoxy.
在一个实施方案中,q为0且RA2或RA1中的至少一个各自独立地为H、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-,和所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1-4个各自独立地选自如下的取代基所取代:四氢吡喃、吡咯烷基、哌嗪基、哌啶基和吗啉基,和各Re 各自独立地选自H或C1-C4烷基。In one embodiment, q is 0 and at least one of RA2 or RA1 is each independently H, optionally substituted ( C1 - C6 alkyl) or optionally substituted ( C1 - C6 alkyl)oxy-, and the C1- C6 alkyl of the optionally substituted ( C1 -C6 alkyl) or optionally substituted ( C1 - C6 alkyl)oxy- is optionally substituted with 1-4 substituents each independently selected from the following substituents: tetrahydropyran, pyrrolidinyl, piperazinyl, piperidinyl and morpholinyl, and each Re is each independently selected from H or C1 - C4 alkyl .
在一个实施方案中,q为0且RA2和RA1各自独立地选自H、羟基或任选取代的(C1-C6烷基)氧基-,和所述任选取代的(C1-C6烷基)氧基- 的C1-C6烷基任选地被羟基、苯基或吗啉基取代,其中苯基或吗啉基各自独立地被甲基或甲氧基取代。In one embodiment, q is 0 and RA2 and RA1 are each independently selected from H, hydroxy or optionally substituted ( C1 - C6 alkyl)oxy-, and the C1 - C6 alkyl of the optionally substituted ( C1 - C6 alkyl)oxy- is optionally substituted with hydroxy, phenyl or morpholinyl, wherein phenyl or morpholinyl are each independently substituted with methyl or methoxy.
在一个实施方案中,q为0,RA1和RA2 独立地为H或任选取代的(C1-C6烷基)氧基-,其中所述任选取代的(C1-C6烷基)氧基-的烷基任选地被1个选自如下的取代基所取代:羟基、任选取代的苯基和任选取代的5-6元杂环烷基,其中所述苯基和5-6元杂环烷基任选地被一个选自如下的取代基所取代:C1-C3烷基和C1-C3烷氧基。In one embodiment, q is 0, RA1 and RA2 are independently H or optionally substituted ( C1 - C6 alkyl)oxy-, wherein the alkyl of said optionally substituted ( C1 - C6 alkyl)oxy- is optionally substituted with one substituent selected from the group consisting of hydroxy, optionally substituted phenyl and optionally substituted 5-6 membered heterocycloalkyl, wherein said phenyl and 5-6 membered heterocycloalkyl are optionally substituted with one substituent selected from the group consisting of C1- C3 alkyl and C1 - C3 alkoxy.
在一个实施方案中,q为0,RA1和RA2 独立地为H或任选取代的(C1-C6烷基)氧基-,其中所述任选取代的(C1-C6烷基)氧基-的烷基任选地被1个选自如下的取代基所取代:羟基、任选取代的苯基和任选取代的吗啉基,其中所述苯基和吗啉基任选地被一个选自以下的取代基取代:C1-C3烷基和C1-C3烷氧基。In one embodiment, q is 0, RA1 and RA2 are independently H or optionally substituted ( C1 - C6 alkyl)oxy-, wherein the alkyl of the optionally substituted ( C1 - C6 alkyl)oxy- is optionally substituted with one substituent selected from the group consisting of hydroxy, optionally substituted phenyl and optionally substituted morpholinyl, wherein the phenyl and morpholinyl are optionally substituted with one substituent selected from the group consisting of C1 - C3 alkyl and C1 - C3 alkoxy.
在一个实施方案中,q为0,RA1和RA2 独立地为H、羟基或任选取代的(C1-C12烷基)氧基-,In one embodiment, q is 0, R A1 and R A2 are independently H, hydroxy or optionally substituted (C 1 -C 12 alkyl)oxy-,
其中所述任选取代的(C1-C12烷基)氧基-的烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、COOH和任选取代的苯基,wherein the alkyl group of the optionally substituted (C 1 -C 12 alkyl)oxy-group is optionally substituted by 1 to 4 substituents independently selected from the following: hydroxyl, COOH and optionally substituted phenyl,
其中所述任选取代的苯基任选地被1-4个各自独立地选自如下的取代基所取代:(C1-C4烷基)氧基-。The optionally substituted phenyl group is optionally substituted by 1 to 4 substituents each independently selected from the following: (C 1 -C 4 alkyl)oxy-.
在一个实施方案中,q为0,RA1和RA2之一为H且RA1和RA2中的另一个为羟基或任选取代的(C1-C4烷基)氧基-,In one embodiment, q is 0, one of RA1 and RA2 is H and the other of RA1 and RA2 is hydroxy or optionally substituted (C 1 -C 4 alkyl)oxy-,
其中所述任选取代的(C1-C4烷基)氧基-的烷基任选地被1-2个各自独立地选自如下的取代基所取代:羟基和任选取代的苯基,wherein the alkyl group of the optionally substituted (C 1 -C 4 alkyl)oxy-group is optionally substituted by 1-2 substituents independently selected from the following: hydroxyl and optionally substituted phenyl,
其中所述任选取代的苯基任选地被1-2个各自独立地为(C1-C4烷基)氧基-的取代基取代。The optionally substituted phenyl group is optionally substituted with 1 to 2 substituents each independently being (C 1 -C 4 alkyl)oxy-.
在一个实施方案中,q为0,RA1和RA2二者都为H。In one embodiment, q is 0 and both RA1 and RA2 are H.
在一个实施方案中,r为0且RB1和RB2各自为H。In one embodiment, r is 0 and RB1 and RB2 are each H.
在另一个实施方案中,r为0且RB1和RB2各自独立地为H、任选取代的C1-C6烷基、卤代(C1-C6烷基)、任选取代的C2-C6烯基、任选取代的C2-C6炔基、任选取代的C3-C6环烷基、任选取代的4-6元杂环烷基、任选取代的5-6元杂芳基或任选取代的9元杂芳基。In another embodiment, r is 0 and RB1 and RB2 are each independently H, optionally substituted C1 - C6 alkyl, halo( C1 - C6 alkyl), optionally substituted C2 - C6 alkenyl, optionally substituted C2 - C6 alkynyl, optionally substituted C3 - C6 cycloalkyl, optionally substituted 4-6 membered heterocycloalkyl, optionally substituted 5-6 membered heteroaryl, or optionally substituted 9 membered heteroaryl.
在本发明化合物的一个实施方案中,s为0,和RC1为H、卤素或C1-C4烷基,和RC2为任选取代的C1-C4烷基,其中所述任选取代的C1-C4烷基任选地被选自如下的取代基所取代: -ORc、-NRcRd、-CO2Rc、-CONRcRd、-SO2NRcRd和-OCONRcRd。In one embodiment of the compounds of the invention, s is 0, and R C1 is H, halogen or C 1 -C 4 alkyl, and R C2 is optionally substituted C 1 -C 4 alkyl, wherein said optionally substituted C 1 -C 4 alkyl is optionally substituted with a substituent selected from the group consisting of -OR c , -NR c R d , -CO 2 R c , -CONR c R d , -SO 2 NR c R d and -OCONR c R d .
在本发明化合物的一个实施方案中,当s为0时,RC1为H和RC2为C1-C4烷基。在另一个实施方案中,当s为0时,RC1为C1-C3烷基,特别是甲基。在另一个实施方案中,当s为0时,RC2为C1-C3烷基,特别是甲基或乙基。在一个选定的实施方案中,当s为0时,RC2为乙基。In one embodiment of the compounds of the present invention, when s is 0, R C1 is H and R C2 is C 1 -C 4 alkyl. In another embodiment, when s is 0, R C1 is C 1 -C 3 alkyl, especially methyl. In another embodiment, when s is 0, R C2 is C 1 -C 3 alkyl, especially methyl or ethyl. In a selected embodiment, when s is 0, R C2 is ethyl.
在一个实施方案中,q为1且A与RA1和RA2一起形成4-8元连接基团。在另一个实施方案中,q为1且A与RA1和RA2一起形成4-6元连接基团。在又另一个实施方案中,q为1且A与RA1和RA2一起形成5-元连接基团。In one embodiment, q is 1 and A, together with RA1 and RA2, forms a 4-8 membered linking group. In another embodiment, q is 1 and A, together with RA1 and RA2, forms a 4-6 membered linking group. In yet another embodiment, q is 1 and A, together with RA1 and RA2, forms a 5-membered linking group.
在另一个实施方案中,q为1,RA1和RA2各自独立地为-CH2-、-NRe-或-O-,并且A为取代的-C2-C10烷基-基团或为未取代的-C2-C10烷基-、-C2-C10烯基-、-C2-C10炔基-、-C1-C4烷基-O-C1-C4烷基-或-C1-C4烷基-NRa-C1-C4烷基-基团,所述取代的-C2-C10烷基-基团被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-和C1-C4烷氧基-。In another embodiment, q is 1, RA1 and RA2 are each independently -CH2- , -NRe- or -O-, and A is a substituted -C2 - C10 alkyl- group or an unsubstituted -C2 - C10 alkyl-, -C2-C10 alkenyl-, -C2- C10 alkynyl-, -C1 - C4 alkyl-OC1 - C4 alkyl- or -C1 - C4 alkyl-NRa - C1 - C4 alkyl- group, the substituted -C2 - C10 alkyl- group being substituted with 1-4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP ( O)(R I R II ) 2 , amino, (C1- C4 alkyl)amino-, ( C1 - C4 alkyl)( C1 -C4 alkyl )amino- , halo(C1 - C 4 alkyl), halo(C 1 -C 4 alkoxy)- and C 1 -C 4 alkoxy-.
在另一个实施方案中,q为1、RA1和RA2各自独立地为-CH2-、-NRe-或-O-,并且A为取代的-C2-C8烷基-基团或为未取代的-C2-C8烷基-、-C2-C8烯基-、-C2-C8炔基-、-C1-C2烷基-O-C1-C2烷基-或-C1-C2烷基-NRa-C1-C2烷基-基团,所述取代的-C2-C8烷基-基团被1-2个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-和C1-C4烷氧基-。In another embodiment, q is 1, RA1 and RA2 are each independently -CH2- , -NRe- or -O-, and A is a substituted -C2 - C8 alkyl-group or an unsubstituted -C2- C8 alkyl-, -C2-C8 alkenyl-, -C2- C8 alkynyl-, -C1 - C2 alkyl -OC1 - C2 alkyl- or -C1 -C2 alkyl -NRa - C1 - C2 alkyl-group, the substituted -C2- C8 alkyl-group being substituted with 1-2 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH ) 2 , -OP(O)(R I R II ) 2 , amino, ( C1 - C4 alkyl)amino-, ( C1 - C4 alkyl)( C1 - C4 alkyl )amino-, halo( C1 -C 4 alkyl), halo(C 1 -C 4 alkoxy)- and C 1 -C 4 alkoxy-.
在另一个实施方案中,q为1,RA1和RA2各自独立地为-CH2-、-NRe-或-O-,并且A为取代的-C2-C6烷基-基团或为未取代的-C2-C6烷基-、-C2-C6烯基-、-C2-C6炔基-、-C1-C2烷基-O-C1-C2烷基 -或-C1-C2烷基-NRa-C1-C2烷基-基团,所述取代的-C2-C6烷基-基团被1-2个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-和C1-C4烷氧基-。In another embodiment, q is 1, RA1 and RA2 are each independently -CH2- , -NRe- or -O-, and A is a substituted -C2 - C6 alkyl- group or an unsubstituted -C2 - C6 alkyl-, -C2-C6 alkenyl-, -C2- C6 alkynyl-, -C1- C2 alkyl-OC1 - C2 alkyl- or -C1 - C2 alkyl-NRa - C1 - C2 alkyl- group, the substituted -C2 - C6 alkyl- group being substituted by 1-2 substituents each independently selected from the group consisting of halogen, hydroxy , -OP(O)(OH) 2 , -OP ( O)(R I R II ) 2 , amino, (C1- C4 alkyl)amino-, ( C1 - C4 alkyl)( C1 -C4 alkyl )amino- , halo(C1 - C 4 alkyl), halo(C 1 -C 4 alkoxy)- and C 1 -C 4 alkoxy-.
在另一个实施方案中,q为1,RA1和RA2各自独立地为-CH2-或-O-,并且A为-C2-C4烷基-、-C2-C4烯基-或-C2-C4炔基-基团。In another embodiment, q is 1, RA1 and RA2 are each independently -CH2- or -O-, and A is a -C2 - C4 alkyl-, -C2 - C4 alkenyl- or -C2 - C4 alkynyl- group.
在选定的实施方案中,q为1,RA1和RA2各自为-O-,并且A为-CH2CH2CH2-,其中A与RA1和RA2一起形成-OCH2CH2CH2O-基团。In selected embodiments , q is 1, RA1 and RA2 are each -O-, and A is -CH2CH2CH2- , wherein A together with RA1 and RA2 form a -OCH2CH2CH2O- group.
在另一个实施方案中,q为1,RA1和RA2各自为-O-,并且A为-CH2-苯基-CH2-,其中A与RA1和RA2一起形成-OCH2-苯基-CH2O-基团。在一个特定的实施方案中,q为1,A与RA1和RA2一起形成-OCH2-苯基-CH2O-基团,其中所述-OCH2-基团位于苯环部分上的 1、4位。In another embodiment, q is 1, RA1 and RA2 are each -O-, and A is -CH2 -phenyl- CH2- , wherein A together with RA1 and RA2 form a -OCH2 -phenyl- CH2O- group. In a specific embodiment, q is 1, A together with RA1 and RA2 form a -OCH2 -phenyl- CH2O- group, wherein the -OCH2- group is located at the 1,4 position on the phenyl ring portion.
本文中定义的连接基团的长度代表由-RA1-A-RA2-和/或-RB1-B-RB2-和/或 -RC1-C-RC2-构成的直链(direct chain)中的最低的原子数。例如,当B为任选取代的苯基时,连接基团-RB1-B-RB2-可以表示为-(CH2)-苯基-(CH2)-。这个连接基团表征为当2个-(CH2)-部分位于苯环的相邻碳原子上(1,2取代的苯基)情况下的4-元连接基团。在另一个实施方案中,这个连接基团表征为当2个-(CH2)-部分在苯环上相对位置(1,4取代苯基)取代情况下的6-元连接基团。应理解A、B或C的任何烷基、烯基或炔基基团或部分为直链或支链的-烷基、烯基或炔基基团或部分。例如,-RB1-B-RB2- 连接基团,其中B为-C1-C10烷基-,可含有8-元连接基团,其具有一个(C1-C4烷基)支链基团或2-4个(C1-C3烷基)支链基团,例如4个支链甲基(2个偕-二甲基)或2个支链甲基。The length of the linking group defined herein represents the lowest number of atoms in the direct chain consisting of -RA1 - ARA2- and/or -RB1 - BRB2- and/or -RC1 - CRC2- . For example, when B is an optionally substituted phenyl group, the linking group -RB1 - BRB2- can be represented as -( CH2 )-phenyl-( CH2 )-. This linking group is characterized as a 4-membered linking group when the two -( CH2 )- moieties are located on adjacent carbon atoms of the benzene ring (1,2 substituted phenyl). In another embodiment, this linking group is characterized as a 6-membered linking group when the two -( CH2 )- moieties are substituted at opposite positions on the benzene ring (1,4 substituted phenyl). It should be understood that any alkyl, alkenyl or alkynyl group or moiety of A, B or C is a straight or branched -alkyl, alkenyl or alkynyl group or moiety. For example, the -RB1 - BRB2- linking group, wherein B is -C1 - C10 alkyl-, may contain an 8-membered linking group having one ( C1 - C4 alkyl) branching group or 2-4 ( C1 - C3 alkyl) branching groups, such as 4 branched methyl groups (2 gem-dimethyl groups) or 2 branched methyl groups.
在本发明化合物的一个实施方案中,r为1,和RB1和RB2各自独立地为-CH2-,并且B与RB1和RB2一起形成连接基团,其中B为键或B为-卤代(C1-C10烷基)-、任选取代的-C1-C10烷基-、任选取代的-C2-C10烯基-、任选取代的-C2-C10炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的C3-C6环烷基、任选取代的苯基、任选取代的4-6元杂环烷基、任选取代的5-6元杂芳基、任选取代的-C1-C4烷基-(C3-C6环烷基)-C1-C4烷基-、任选取代的-C1-C4烷基-苯基-C1-C4烷基-、任选取代的-C1-C4烷基-(4-6元杂环烷基)-C1-C4烷基-或任选取代的-C1-C4烷基-(5-6元杂芳基)-C1-C4烷基-,In one embodiment of the compounds of the present invention, r is 1, and RB1 and RB2 are each independently -CH2- , and B together with RB1 and RB2 form a linking group, wherein B is a bond or B is -halo( C1 - C10 alkyl)-, optionally substituted- C1 - C10 alkyl-, optionally substituted- C2 - C10 alkenyl-, optionally substituted- C2 - C10 alkynyl-, optionally substituted- C1 - C6 alkyl- OC1 - C6 alkyl-, optionally substituted- C1 - C6 alkyl- NRa - C1 - C6 alkyl-, optionally substituted C3 - C6 cycloalkyl, optionally substituted phenyl, optionally substituted 4-6 membered heterocycloalkyl, optionally substituted 5-6 membered heteroaryl, optionally substituted- C1 - C4 alkyl-( C3 - C6 cycloalkyl)-C1- C4 alkyl-, optionally substituted- C1 - C6 -C 1 -C 4 alkyl-phenyl-C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 4 alkyl-, or optionally substituted -C 1 -C 4 alkyl-(5-6 membered heteroaryl)-C 1 -C 4 alkyl-,
其中所述任选取代的-C1-C10烷基-、任选取代的-C2-C10烯基-、任选取代的-C2-C10炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的-C1-C4烷基-(C3-C6环烷基)-C1-C4烷基-、任选取代的-C1-C4烷基-苯基-C1-C4烷基-、任选取代的-C1-C4烷基-(4-6元杂环烷基)-C1-C4烷基-或任选取代的-C1-C4烷基-(5-6元杂芳基-C1-C4烷基)-的烷基部分任选地被1- 4个各自独立地选自如下的取代基所取代:-C1-C4烷基、卤素、卤代(C1-C4烷基)、-OH、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-ORc、-NH2、 -NRcRd、-OCORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2、-SO2NRcRd、-OCONH2、-OCONRcRd、-NRdCORc、-NRdSORc、-NRdCO2Rc和-NRdSO2Rc,wherein the alkyl portion of the optionally substituted -C 1 -C 10 alkyl-, optionally substituted -C 2 -C 10 alkenyl-, optionally substituted -C 2 -C 10 alkynyl-, optionally substituted -C 1 -C 6 alkyl-OC 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-NR a -C 1 -C 6 alkyl-, optionally substituted -C 1 -C 4 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-phenyl-C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 4 alkyl- or optionally substituted -C 1 -C 4 alkyl-(5-6 membered heteroaryl-C 1 -C 4 alkyl)- is optionally substituted with 1-C 4 alkyl- wherein the at least one aryl group is substituted with 4 substituents each independently selected from the group consisting of -C 1 -C 4 alkyl, halogen, halo(C 1 -C 4 alkyl), -OH, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -OR c , -NH 2 , -NR c R d , -OCOR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d , -SO 2 NH 2 , -SO 2 NR c R d , -OCONH 2 , -OCONR c R d , -NR d COR c , -NR d SOR c , -NR d CO 2 R c and -NR d SO 2 R c ,
和and
所述任选取代的C3-C6环烷基、任选取代的苯基、任选取代的4-6元杂环烷基、任选取代的5-6元杂芳基、任选取代的-C1-C4烷基-(C3-C6环烷基)-C1-C4烷基-、任选取代的-C1-C4烷基-苯基-C1-C4烷基-、任选取代的-C1-C4烷基-(4-6元杂环烷基)-C1-C4烷基-或任选取代的-C1-C4烷基-(5-6元杂芳基)-C1-C4烷基-的C3-C6环烷基、苯基、4-6元杂环烷基或5-6元杂芳基部分任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、C1-C4烷基、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-和C1-C4烷氧基-(C1-C4烷氧基)-。The C 3 -C 6 cycloalkyl, phenyl, 4-6-membered heterocycloalkyl or 5-6-membered heteroaryl moiety of the optionally substituted C 3 -C 6 cycloalkyl, optionally substituted phenyl, optionally substituted 4-6-membered heterocycloalkyl, optionally substituted 5-6-membered heteroaryl, optionally substituted -C 1 -C 4 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl - phenyl -C 1 -C 4 alkyl-, optionally substituted -C 1 -C 4 alkyl-(4-6-membered heterocycloalkyl)-C 1 -C 4 alkyl- or optionally substituted -C 1 -C 4 alkyl-(5-6-membered heteroaryl)-C 1 -C 4 alkyl- is optionally substituted with 1 to 4 substituents each independently selected from the following: halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)- and C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-.
在本发明化合物的一个实施方案中,r为1,RB1和RB2各自独立地为-CH2-,并且B与RB1和RB2一起形成2-6元连接基团。在另一个实施方案中,r为1,RB1和RB2各自独立地为-CH2-,并且B与RB1和RB2一起形成3-6元连接基团。在又另一个实施方案中,r为1,RB1和RB2各自独立地为-CH2-,并且B与RB1和RB2一起形成4-5元连接基团。In one embodiment of the compounds of the present invention, r is 1, RB1 and RB2 are each independently -CH2- , and B, RB1 and RB2, together form a 2-6 membered linking group. In another embodiment, r is 1, RB1 and RB2 are each independently -CH2- , and B, RB1 and RB2, together form a 3-6 membered linking group. In yet another embodiment, r is 1, RB1 and RB2 are each independently -CH2- , and B, RB1 and RB2, together form a 4-5 membered linking group.
在本发明化合物的一个实施方案中,r为1,RB1和RB2各自独立地为-CRdRf -,并且B与RB1和RB2一起形成2-6元连接基团。在另一个实施方案中,r为1,RB1和RB2各自独立地为-CH2-,并且B与RB1和RB2一起形成3-6元连接基团。在又另一个实施方案中,r为1,RB1和RB2各自独立地为-CH2-,并且B与RB1和RB2一起形成4-5元连接基团。In one embodiment of the compounds of the present invention, r is 1, RB1 and RB2 are each independently -CRdRf- , and B, RB1 and RB2, together form a 2-6 membered linking group. In another embodiment, r is 1, RB1 and RB2 are each independently -CH2- , and B, RB1 and RB2, together form a 3-6 membered linking group. In yet another embodiment, r is 1, RB1 and RB2 are each independently -CH2- , and B, RB1 and RB2, together form a 4-5 membered linking group.
在一个实施方案中,B为键。In one embodiment, B is a bond.
在另一个实施方案中,r为1,RB1和RB2各自独立地为-CH2-,并且B为取代的-C1-C10烷基-基团或为未取代的-C1-C10烷基-、-C2-C10烯基-、-C2-C10炔基-、-C1-C6烷基-O-C1-C6烷基-或-C1-C6烷基-NRa-C1-C6烷基-基团,所述取代的-C1-C10烷基-基团被1-4个各自独立地选自如下的取代基所取代:-C1-C4烷基、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C6烷基)氨基-、(C1-C6烷基)(C1-C6烷基)氨基-、卤代(C1-C6烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、C1-C4烷氧基-(C1-C4烷氧基)-、-NHCO(C1-C4烷基)、任选取代的苯基、任选取代的5-6元杂环烷基和任选取代的5-6元杂芳基,其中所述任选取代的苯基、5-6元杂环烷基或5-6元杂芳基任选地被1-4个各自独立地选自如下的取代基所取代:-C1-C4烷基、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C6烷基)氨基-、(C1-C6烷基)(C1-C6烷基)氨基-、卤代(C1-C6烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-和C1-C4烷氧基-(C1-C4烷氧基)-。In another embodiment, r is 1, RB1 and RB2 are each independently -CH2- , and B is a substituted -C1 - C10 alkyl-group or an unsubstituted -C1 - C10 alkyl-, -C2- C10 alkenyl-, -C2- C10 alkynyl-, -C1 - C6 alkyl- OC1 - C6 alkyl- or -C1 - C6 alkyl- NRa - C1 - C6 alkyl-group, the substituted -C1 - C10 alkyl-group being substituted by 1-4 substituents each independently selected from the group consisting of -C1 - C4 alkyl, halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, ( C1 - C6 alkyl)amino-, (C1- C6 alkyl)( C1 - C6 alkyl)amino-, halo( C1 - C alkyl )-, -C 1 -C 6 alkyl), halo(C 1 -C 6 alkyl), halo(C 1 -C 6 alkyl)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-, -NHCO(C 1 -C 4 alkyl), optionally substituted phenyl, optionally substituted 5-6 membered heterocycloalkyl and optionally substituted 5-6 membered heteroaryl, wherein the optionally substituted phenyl, 5-6 membered heterocycloalkyl or 5-6 membered heteroaryl is optionally substituted with 1 to 4 substituents each independently selected from the group consisting of -C 1 -C 4 alkyl, halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 6 alkyl)amino-, (C 1 -C 6 alkyl)(C 1 -C 6 alkyl)amino-, halo(C 1 -C 6 alkyl), halo(C 1 -C 6 alkyl), In some embodiments , the present invention comprises hydroxy-(C 2 -C 4 alkoxy)-, hydroxy-(C 2 -C 4 alkoxy)-, and hydroxy-(C 1 -C 4 alkoxy)-.
在另一个实施方案中,r为1,RB1和RB2各自独立地为-CH2-,并且B为取代的-C1-C10烷基-基团或为未取代的-C1-C10烷基-、-C2-C10烯基-、-C2-C10炔基-、-C1-C6烷基-O-C1-C6烷基-或-C1-C6烷基-NRa-C1-C6烷基-基团,所述取代的-C1-C10烷基-基团被1-4个各自独立地选自如下的取代基所取代:-C1-C4烷基、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-和C1-C4烷氧基-。In another embodiment, r is 1, RB1 and RB2 are each independently -CH2- , and B is a substituted -C1 - C10 alkyl-group or an unsubstituted -C1 - C10 alkyl-, -C2- C10 alkenyl-, -C2- C10 alkynyl-, -C1 - C6 alkyl- OC1 - C6 alkyl- or -C1 - C6 alkyl- NRa - C1 - C6 alkyl-group, the substituted -C1 - C10 alkyl-group being substituted by 1-4 substituents each independently selected from the group consisting of -C1 - C4 alkyl, halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, ( C1 - C4 alkyl)amino-, (C1- C4 alkyl)( C1 - C4 alkyl)amino-, halo( C1 - C 4 alkyl), halo(C 1 -C 4 alkoxy)- and C 1 -C 4 alkoxy-.
在另一个实施方案中,r为1,RB1和RB2各自独立地为-CH2-,并且B为取代的-C1-C8烷基-基团或为未取代的-C1-C8烷基-、-C2-C8烯基-、-C2-C8炔基-、-C1-C4烷基-O-C1-C4烷基-或-C1-C4烷基-NRa-C1-C4烷基-基团,所述取代的-C1-C8烷基-基团被1-4个各自独立地选自如下的取代基所取代:-C1-C4烷基、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-和C1-C4烷氧基-。In another embodiment, r is 1, RB1 and RB2 are each independently -CH2- , and B is a substituted -C1 - C8 alkyl-group or an unsubstituted -C1 - C8 alkyl-, -C2- C8 alkenyl-, -C2- C8 alkynyl-, -C1 - C4 alkyl- OC1 - C4 alkyl- or -C1- C4 alkyl- NRa - C1 - C4 alkyl-group, the substituted -C1 - C8 alkyl-group being substituted by 1-4 substituents each independently selected from the group consisting of -C1 - C4 alkyl, halogen, hydroxy, -OP (O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, ( C1 - C4 alkyl)amino-, (C1- C4 alkyl)( C1 -C4 alkyl)amino-, halo( C1 - C4 alkyl ) 4 alkyl), halo(C 1 -C 4 alkoxy)- and C 1 -C 4 alkoxy-.
在另一个实施方案中,r为1,RB1和RB2各自独立地为-CH2-,并且B为取代的-C1-C6烷基-基团或为未取代的-C1-C6烷基-、-C2-C6烯基-、-C2-C6炔基-、-C1-C2烷基-O-C1-C2烷基-或-C1-C2烷基-NRa-C1-C2烷基-基团,所述取代的-C1-C6烷基-基团被1-4个各自独立地选自如下的取代基所取代:-C1-C4烷基、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-和C1-C4烷氧基-。In another embodiment, r is 1, RB1 and RB2 are each independently -CH2- , and B is a substituted -C1 - C6 alkyl-group or an unsubstituted -C1 - C6 alkyl-, -C2 -C6 alkenyl-, -C2- C6 alkynyl-, -C1- C2 alkyl- OC1 - C2 alkyl- or -C1 - C2 alkyl- NRa - C1 - C2 alkyl-group, the substituted -C1 - C6 alkyl-group being substituted by 1-4 substituents each independently selected from the group consisting of -C1 - C4 alkyl, halogen , hydroxy, -OP(O)(OH)2 , -OP(O)(R I R II ) 2 , amino, ( C1 - C4 alkyl)amino-, (C1- C4 alkyl)( C1 - C4 alkyl)amino-, halo( C1 - C 4 alkyl), halo(C 1 -C 4 alkoxy)- and C 1 -C 4 alkoxy-.
在另一个实施方案中,r为1、RB1和RB2各自独立地为-CH2-,并且B为取代的-C2-C4烷基-基团或为未取代的-C2-C4烷基-、-C2-C4烯基-、-C2-C4炔基-、-C1烷基-O-C1烷基-或-C1烷基-NRa-C1烷基-基团,所述取代的-C2-C4烷基-基团被1-4个各自独立地选自如下的取代基所取代:-C1-C4烷基、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-和C1-C4烷氧基-。In another embodiment, r is 1, RB1 and RB2 are each independently -CH2- , and B is a substituted -C2 - C4 alkyl-group or an unsubstituted -C2-C4 alkyl-, -C2-C4 alkenyl-, -C2-C4 alkynyl-, -C1 alkyl-OC1 alkyl- or -C1 alkyl-NRa-C1 alkyl-group, wherein the substituted -C2-C4 alkyl - group is substituted with 1-4 substituents each independently selected from the group consisting of -C1 - C4 alkyl, halogen, hydroxy, -OP(O)( OH ) 2 , -OP(O) ( R I R II ) 2 , amino, ( C1 - C4 alkyl)amino-, ( C1 - C4 alkyl)( C1 - C4 alkyl)amino-, halo( C1 - C4 alkyl), halo( C1 - C4 alkoxy)- and C1-C4 alkyl-. 1 -C 4 alkoxy-.
在一个实施方案中,r为1,RB1和RB2各自独立地为-CRdRf,Rd和Rf为H或甲基,和B为-CH=CH-、-CH(CH3)=CH(CH3)-、-CH2CH2-、-CH(OH)CH(OH)-、-CH(CH3)CH(CH3)-、-CF2-CF2-或-CH2CH2CH2-。在一个实施方案中,r为1,RB1和RB2各自独立地为-CRdRf,Rd和Rf为H或甲基,和B为-CH=CH-、-CH2CH2-、-CH(OH)CH(OH)-。在这些实施方案中,r为1,B与RB1和RB2一起形成-CH2CH=CHCH2-、-CH2CH2CH2CH2-、-CH2CH(OH)CH(OH)CH2-或-CH2CH2N(CH3)CH2CH2-基团。在这些实施方案中,r为1、B与RB1和RB2一起形成-CH2CH=CHCH2-。在一个实施方案中,r为1、B与RB1和RB2一起形成-CH2CH2CH2CH2-。In one embodiment, r is 1, RB1 and RB2 are each independently -CRdRf , Rd and Rf are H or methyl, and B is -CH=CH-, -CH( CH3 )=CH( CH3 )-, -CH2CH2- , -CH(OH)CH(OH)-, -CH(CH3)CH(CH3 ) - , -CF2 - CF2- , or -CH2CH2CH2- . In one embodiment, r is 1, RB1 and RB2 are each independently -CRdRf, Rd and Rf are H or methyl , and B is -CH= CH- , -CH2CH2-, -CH( OH )CH(OH ) -. In these embodiments, r is 1, and B, together with RB1 and RB2, forms a -CH2CH = CHCH2- , -CH2CH2CH2CH2- , -CH2CH ( OH )CH(OH) CH2- , or -CH2CH2N ( CH3 ) CH2CH2- group. In these embodiments, r is 1, and B , together with RB1 and RB2, forms -CH2CH = CHCH2- . In one embodiment, r is 1, and B, together with RB1 and RB2, forms -CH2CH2CH2CH2- .
在本发明化合物的一个实施方案中,s为1,且RC1和RC2各自独立地为-CH2-,并且C与RC1和RC2一起形成连接基团,其中C为-卤代(C1-C12烷基)-、任选取代的-C1-C12烷基-、任选取代的-C2-C12烯基-、任选取代的-C2-C12炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-,In one embodiment of the compounds of the present invention, s is 1, and R C1 and R C2 are each independently -CH 2 -, and C together with R C1 and R C2 form a linking group, wherein C is -halo(C 1 -C 12 alkyl)-, optionally substituted -C 1 -C 12 alkyl-, optionally substituted -C 2 -C 12 alkenyl-, optionally substituted -C 2 -C 12 alkynyl-, optionally substituted -C 1 -C 6 alkyl-OC 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-NR a -C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-phenyl-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 6 6 alkyl- or optionally substituted -C 1 -C 6 alkyl-(5-6 membered heteroaryl)-C 1 -C 6 alkyl-,
其中所述任选取代的-C1-C12烷基-、任选取代的-C2-C12烯基-、任选取代的-C2-C12炔基-、任选取代的-C1-C6烷基-O-C1-C6烷基-、任选取代的-C1-C6烷基-NRa-C1-C6烷基-、任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-的烷基部分任选地被1或2个各自独立地选自如下的取代基所取代:卤素、卤代(C1-C4烷基)、-OH、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-ORc、-NH2、-NRcRd、-OCORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2、-SO2NRcRd、-OCONH2、-OCONRcRd、-NRdCORc、-NRdSORc、-NRdCO2Rc和-NRdSO2Rc,wherein the optionally substituted -C 1 -C 12 alkyl-, optionally substituted -C 2 -C 12 alkenyl-, optionally substituted -C 2 -C 12 alkynyl-, optionally substituted -C 1 -C 6 alkyl-OC 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-NR a -C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-phenyl-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(4-6 membered heterocycloalkyl)-C 1 -C 6 alkyl- or optionally substituted -C 1 -C 6 alkyl-(5-6 membered heteroaryl)-C 1 -C The alkyl portion of the 6 -alkyl- is optionally substituted with 1 or 2 substituents each independently selected from the group consisting of halogen, halo(C 1 -C 4 alkyl), -OH, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -OR c , -NH 2 , -NR c R d , -OCOR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d , -SO 2 NH 2 , -SO 2 NR c R d , -OCONH 2 , -OCONR c R d , -NR d COR c , -NR d SOR c , -NR d CO 2 R c and -NR d SO 2 R c ,
和and
所述任选取代的-C1-C6烷基-(C3-C6环烷基)-C1-C6烷基-、任选取代的-C1-C6烷基-苯基-C1-C6烷基-、任选取代的-C1-C6烷基-(4-6元杂环烷基)-C1-C6烷基-或任选取代的-C1-C6烷基-(5-6元杂芳基)-C1-C6烷基-的C3-C6环烷基、苯基、4-6元杂环烷基或5-6元杂芳基部分任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、C1-C4烷基、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2和C1-C4烷氧基-(C1-C4烷氧基)-。The C 3 -C 6 cycloalkyl, phenyl, 4-6-membered heterocycloalkyl or 5-6-membered heteroaryl portion of the optionally substituted -C 1 -C 6 alkyl-(C 3 -C 6 cycloalkyl)-C 1 -C 6 alkyl- , optionally substituted -C 1 -C 6 alkyl-phenyl-C 1 -C 6 alkyl-, optionally substituted -C 1 -C 6 alkyl-(4-6-membered heterocycloalkyl)-C 1 -C 6 alkyl- or optionally substituted -C 1 -C 6 alkyl- ( 5-6 - membered heteroaryl)-C 1 -C 6 alkyl- is optionally substituted with 1 to 4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, C 1 -C 6 R I R II ) 2 and C 1 -C 4 alkoxy- ( C 1 -C 4 alkoxy ) - .
在本发明化合物的一个实施方案中,s为1,RC1和RC2各自独立地为-CH2-,并且C与RC1和RC2一起形成4-8元连接基团。在另一个实施方案中,s为1,且C与RC1和RC2一起形成4-6元连接基团。在又另一个实施方案中,s为1,且C与RC1和RC2一起形成5-元连接基团。In one embodiment of the compounds of the present invention, s is 1, R C1 and R C2 are each independently -CH 2 -, and C together with R C1 and R C2 forms a 4-8 membered linking group. In another embodiment, s is 1, and C together with R C1 and R C2 forms a 4-6 membered linking group. In yet another embodiment, s is 1, and C together with R C1 and R C2 forms a 5-membered linking group.
在另一个实施方案中,s为1,RC1和RC2各自独立地为-CH2-,并且C为取代的-C2-C10烷基-基团或为未取代的-C2-C10烷基-、-C2-C10烯基-、-C2-C10炔基-、-C1-C4烷基-O-C1-C4烷基-或-C1-C4烷基-NRa-C1-C4烷基-基团,所述取代的-C2-C10烷基-基团被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-和C1-C4烷氧基-。In another embodiment, s is 1, R C1 and R C2 are each independently -CH 2 -, and C is a substituted -C 2 -C 10 alkyl- group or an unsubstituted -C 2 -C 10 alkyl-, -C 2 -C 10 alkenyl-, -C 2 -C 10 alkynyl-, -C 1 -C 4 alkyl-OC 1 -C 4 alkyl- or -C 1 -C 4 alkyl-NR a -C 1 -C 4 alkyl- group, the substituted -C 2 -C 10 alkyl- group being substituted by 1-4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkoxy)- and C 1 -C 4 alkoxy-.
在另一个实施方案中,s为1,RC1和RC2各自独立地为-CH2-,并且C为取代的-C2-C8烷基-基团或为未取代的-C2-C8烷基-、-C2-C8烯基-、-C2-C8炔基-、-C1-C2烷基-O-C1-C2烷基-或-C1-C2烷基-NRa-C1-C2烷基-基团,所述取代的-C2-C8烷基-基团被1-2个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-和C1-C4烷氧基-。In another embodiment, s is 1, R C1 and R C2 are each independently -CH 2 -, and C is a substituted -C 2 -C 8 alkyl- group or an unsubstituted -C 2 -C 8 alkyl-, -C 2 -C 8 alkenyl-, -C 2 -C 8 alkynyl-, -C 1 -C 2 alkyl-OC 1 -C 2 alkyl- or -C 1 -C 2 alkyl-NR a -C 1 -C 2 alkyl- group, the substituted -C 2 -C 8 alkyl- group being substituted by 1-2 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl )(C 1 -C 4 alkyl)amino-, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkyl), 4 alkoxy)- and C 1 -C 4 alkoxy-.
在另一个实施方案中,s为1,RC1和RC2各自独立地为-CH2-,并且C为取代的-C2-C6烷基-基团或为未取代的-C2-C6烷基-、-C2-C6烯基-、-C2-C6炔基-、-C1-C2烷基-O-C1-C2烷基-或-C1-C2烷基-NRa-C1-C2烷基-基团,所述取代的-C2-C6烷基-基团被1-2个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、卤代(C1-C4烷基)、卤代(C1-C4烷氧基)-和C1-C4烷氧基-。In another embodiment, s is 1, R C1 and R C2 are each independently -CH 2 -, and C is a substituted -C 2 -C 6 alkyl- group or an unsubstituted -C 2 -C 6 alkyl-, -C 2 -C 6 alkenyl-, -C 2 -C 6 alkynyl-, -C 1 -C 2 alkyl-OC 1 -C 2 alkyl- or -C 1 -C 2 alkyl-NR a -C 1 -C 2 alkyl- group, the substituted -C 2 -C 6 alkyl- group being substituted by 1-2 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl )(C 1 -C 4 alkyl)amino-, halo(C 1 -C 4 alkyl), halo(C 1 -C 4 alkyl), 4 alkoxy)- and C 1 -C 4 alkoxy-.
在另一个实施方案中,s为1,RC1和RC2各自独立地为-CH2-,并且C为-C2-C4烷基-、-C2-C4烯基-或-C2-C4炔基-基团。In another embodiment, s is 1, R C1 and R C2 are each independently -CH 2 -, and C is a -C 2 -C 4 alkyl-, -C 2 -C 4 alkenyl- or -C 2 -C 4 alkynyl- group.
在选定的实施方案中,s为1,RC1和RC2各自独立地为-CH2-,并且C为-CH2CH2CH2-,其中C与RC1和RC2一起形成-CH2CH2CH2CH2CH2-基团。In selected embodiments, s is 1, RC1 and RC2 are each independently -CH2- , and C is -CH2CH2CH2- , wherein C together with RC1 and RC2 form a -CH2CH2CH2CH2CH2- group .
在本发明化合物的一个实施方案中,R4和R6各自独立地选自H、卤素、卤代(C1-C6烷基)、卤代(C1-C6烷氧基)-、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-NH2、-NRcRc、-NRcRd、-CORc、-CO2Rc、-N(Rd)CORc、-N(Rd)SO2Rc、-N(Rg)SO2(C1-C2烷基)-N(Rh)(Rf)、-N(Rg)CO(C1-C2烷基)-N(Rh)(Rf)、任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-、任选取代的(C1-C6烷基)氨基-和任选取代的(C1-C6烷基)(C1-C4烷基)氨基-,In one embodiment of the compounds of the present invention, R4 and R6 are each independently selected from H, halogen, halo( C1 - C6 alkyl), halo( C1 - C6 alkoxy)-, hydroxy, -OP(O)(OH) 2 , -OP(O)( R I R II ) 2 , -NH2 , -NRcRc , -NRcRd , -CORc , -CO2Rc , -N( Rd ) CORc , -N ( Rd ) SO2Rc , -N( Rg ) SO2 (C1-C2 alkyl)-N( Rh )( Rf ), -N( Rg ) CO( C1 - C2 alkyl)-N( Rh )( Rf ), optionally substituted ( C1 - C6 alkyl), optionally substituted ( C1 - C6 alkyl)oxy-, optionally substituted ( C1 - C6 alkyl)oxy-, 6 alkyl)amino- and optionally substituted (C 1 -C 6 alkyl)(C 1 -C 4 alkyl)amino-,
其中所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-、任选取代的(C1-C6烷基)氨基-和任选取代的(C1-C6烷基)(C1-C4烷基)氨基-的(C1-C6烷基)任选地被1-4个各自独立地选自如下的取代基所取代:-OH、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-ORc、-NH2、-NRcRc、-NRcRd、-CO2H、-CO2Rc、OCORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2、-SO2NRcRd、-OCONH2、-OCONRcRd、-NRdCORc、-NRdSORc、-NRdCO2Rc、-NRdSO2Rc、任选取代的苯基、任选取代的5-6元杂环烷基和任选取代的5-6元杂芳基,其中所述任选取代的苯基、5-6元杂环烷基或5-6元杂芳基任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、氨基、(C1-C4烷基)氨基-、(C1-C4烷基)(C1-C4烷基)氨基-、C1-C4烷基、卤代(C1-C4烷基)、羟基-(C1-C4烷基)-、-(C1-C4烷基)-O-P(O)(OH)2、-(C1-C4烷基)-O-P(O)(RIRII)2、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2、C1-C4烷氧基-(C1-C4烷氧基)-、-CORd、-CON(Rd)(Rf)和-CO2Rd。wherein the optionally substituted (C 1 -C 6 alkyl), optionally substituted (C 1 -C 6 alkyl)oxy-, optionally substituted (C 1 -C 6 alkyl)amino- and optionally substituted (C 1 -C 6 alkyl)(C 1 -C 4 alkyl)amino-(C 1 -C 6 alkyl) are optionally substituted by 1 to 4 substituents each independently selected from the group consisting of -OH, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -OR c , -NH 2 , -NR c R c , -NR c R d , -CO 2 H , -CO 2 R c , OCOR c , -CO 2 H , -CO 2 R c , -SOR c , -SO 2 R c , -CONH 2 , -CONR c R d , -SO 2 NH 2 , -SO 2 NR c R d , -OCONH 2 , -OCONR c R d , -NR d COR c , -NR d SOR c , -NR d CO 2 R c , -NR d SO 2 R c , optionally substituted phenyl, optionally substituted 5-6 membered heterocycloalkyl, and optionally substituted 5-6 membered heteroaryl, wherein the optionally substituted phenyl, 5-6 membered heterocycloalkyl, or 5-6 membered heteroaryl is optionally substituted with 1-4 substituents each independently selected from the group consisting of halogen, hydroxy, amino, (C 1 -C 4 alkyl)amino-, (C 1 -C 4 alkyl)(C 1 -C 4 alkyl)amino-, C 1 -C 4 alkyl, halo(C 1 -C 4 alkyl), hydroxy-(C 1 -C 4 alkyl)-, -(C 1 -C 4 alkyl)-OP(O)(OH) 2 , -(C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 , C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-, -COR d , -CON(R d )(R f ) and -CO 2 R d .
在一个实施方案中,R4和R6各自为H。In one embodiment, R4 and R6 are each H.
在一个实施方案中,R3和R5 独立地选自-CO-N(Rd)(Rf),且各Rd和Rf独立地为H或C1-C3烷基。In one embodiment, R 3 and R 5 are independently selected from -CO-N(R d )(R f ), and each R d and R f is independently H or C 1 -C 3 alkyl.
在一个实施方案中,R3和R5为CONH2。In one embodiment, R 3 and R 5 are CONH 2 .
在本发明化合物的一个实施方案中,R14为任选取代的C1-C4烷基,其中所述任选取代的C1-C4烷基任选地被选自如下的取代基所取代: -ORc、-NRcRd、-CO2Rc、-CONRcRd、-SO2NRcRd和-OCONRcRd。In one embodiment of the compounds of the present invention, R14 is optionally substituted C1 - C4 alkyl , wherein said optionally substituted C1 - C4 alkyl is optionally substituted with a substituent selected from the group consisting of -ORc , -NRcRd , -CO2Rc , -CONRcRd , -SO2NRcRd and -OCONRcRd .
在本发明化合物的一个实施方案中,R16为H、卤素或C1-C4烷基。In one embodiment of the compounds of the present invention, R 16 is H, halogen or C 1 -C 4 alkyl.
在本发明化合物的一个实施方案中,R15和R17各自独立地为H、环丙基或C1-C4烷基。In one embodiment of the compounds of the present invention, R 15 and R 17 are each independently H, cyclopropyl or C 1 -C 4 alkyl.
在本发明化合物的一个实施方案中,R14、R15、R16和R17各自独立地为H或C1-C4烷基。In one embodiment of the compounds of the present invention, R 14 , R 15 , R 16 and R 17 are each independently H or C 1 -C 4 alkyl.
在本发明的一个实施方案中,R16为H。In one embodiment of the present invention, R 16 is H.
在另一个实施方案中,R14、R15和R17各自独立地为C1-C4烷基。In another embodiment, R 14 , R 15 and R 17 are each independently C 1 -C 4 alkyl.
在另一个实施方案中,R14、R15和R17各自独立地为C1-C3烷基,具体地,甲基或乙基。在一个选定的实施方案中,R14 为乙基。In another embodiment, R 14 , R 15 and R 17 are each independently C 1 -C 3 alkyl, specifically methyl or ethyl. In a selected embodiment, R 14 is ethyl.
在另一个实施方案中,R15和R17各自为甲基。In another embodiment, R 15 and R 17 are each methyl.
在本发明化合物的一个实施方案中,Ra为H、-Rc、-CORc、-CO2H、-CO2Rc、-SORc、-SO2Rc、-CONH2、-CONRcRd、-SO2NH2或-SO2NRcRd。In one embodiment of the compounds of the present invention, Ra is H, -Rc , -CORc , -CO2H , -CO2Rc , -SORc , -SO2Rc , -CONH2 , -CONRcRd , -SO2NH2 , or -SO2NRcRd .
在另一个实施方案中,Ra为H、C1-C4烷基、-CO(C1-C4烷基)、-CO(C1-C4 In another embodiment, Ra is H, C1 - C4 alkyl, -CO( C1 - C4 alkyl), -CO( C1 - C4
烷基)-OH、-CO(C1-C4烷基)-O-(C1-C4烷基)、-CO(C1-C4烷基)-NH2、-CO(C1-C4烷基)-NH(C1-C4烷基)或-CO(C1-C4烷基)-N(C1-C4烷基)(C1-C4烷基)。-CO(C 1 -C 4 alkyl)-OH, -CO(C 1 -C 4 alkyl)-O-(C 1 -C 4 alkyl), -CO(C 1 -C 4 alkyl)-NH 2 , -CO(C 1 -C 4 alkyl)-NH(C 1 -C 4 alkyl), or -CO(C 1 -C 4 alkyl)-N(C 1 -C 4 alkyl)(C 1 -C 4 alkyl).
在本发明化合物的一个实施方案中,其中Rx和Ry各自独立地为甲基或乙基。在本发明化合物的一个实施方案中,其中Rx和Ry二者都为甲基。 在本发明化合物的一个实施方案中,式(I)化合物,其中Rx和Ry之一为甲基且另一个为H。In one embodiment of the compounds of the present invention, Rx and Ry are each independently methyl or ethyl. In one embodiment of the compounds of the present invention, Rx and Ry are both methyl. In one embodiment of the compounds of the present invention, the compound of formula (I), wherein one of Rx and Ry is methyl and the other is H.
本发明的一个实施方案涉及式(I)化合物,其中:One embodiment of the present invention relates to compounds of formula (I), wherein:
q + r + s = 1或2;q + r + s = 1 or 2;
q为0,并且RA1和RA2独立地选自 H、-OCH2CH2CH2OH和-OCH3;或q is 0, and RA1 and RA2 are independently selected from H , -OCH2CH2CH2OH and -OCH3 ; or
q为1,RA1和RA2各自为-O-,和A为-CH2CH2CH2-;q is 1, RA1 and RA2 are each -O-, and A is -CH 2 CH 2 CH 2 -;
r为0,并且RB1和RB2各自为H;或r is 0, and RB1 and RB2 are each H; or
r为1,RB1和RB2各自独立地为-CH2-,和B为-CH=CH-、-CH2CH2-、-CH(OH)CH(OH)-或-CH2N(CH3)CH2-;r is 1, RB1 and RB2 are each independently -CH2- , and B is -CH=CH- , -CH2CH2- , -CH(OH)CH(OH)-, or -CH2N ( CH3 ) CH2- ;
s为0,RC1为甲基和RC2为乙基;或s is 0, R C1 is methyl and R C2 is ethyl; or
s为1,RC1和RC2各自独立地为-CH2-,和C为-CH2CH2CH2-;s is 1, RC1 and RC2 are each independently -CH 2 -, and C is -CH 2 CH 2 CH 2 -;
R3和R5各自为-CONH2;R 3 and R 5 are each -CONH 2 ;
R4和R6各自为H; R4 and R6 are each H;
R14为乙基;R 14 is ethyl;
R15为甲基;R 15 is methyl;
R16为H; R16 is H;
R17为甲基,R 17 is a methyl group,
或其盐,特别是药学上可接受的盐。or a salt thereof, particularly a pharmaceutically acceptable salt.
在本发明化合物的一个实施方案中,In one embodiment of the compounds of the present invention,
R14、R15、Rc2和R17独立地为甲基或乙基;R 14 , R 15 , R c2 and R 17 are independently methyl or ethyl;
RA1和RA2之一为H且RA1和RA2中的另一个为任选取代的(C1-C4烷基)氧基-,One of R A1 and R A2 is H and the other of R A1 and R A2 is optionally substituted (C 1 -C 4 alkyl)oxy-,
其中任选取代的(C1-C4烷基)氧基-的烷基任选地被1-2个各自独立地选自如下的取代基所取代:羟基和任选取代的苯基,wherein the alkyl group of the optionally substituted (C 1 -C 4 alkyl)oxy-group is optionally substituted by 1 to 2 substituents each independently selected from the following: hydroxyl and optionally substituted phenyl,
其中所述任选取代的苯基任选地被1-2个各自独立地选自(C1-C4烷基)氧基-的取代基所取代;wherein the optionally substituted phenyl is optionally substituted by 1-2 substituents each independently selected from (C 1 -C 4 alkyl)oxy-;
R3和R5二者都为-CO-NH2;和 R3 and R5 are both -CO- NH2 ; and
B为取代的-CH2-CH2-或取代的-CH=CH-,B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个各自独立地选自如下的取代基所取代:氟和C1-2烷基;和wherein the substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 substituents independently selected from the following: fluorine and C 1-2 alkyl; and
RX或Ry中的至少一个独立地为C1-C4烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 4 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C4烷基。Or both RX and Ry are independently C1 - C4 alkyl.
在本发明的一个实施方案中,本发明化合物为式(I-B’)In one embodiment of the present invention, the compound of the present invention is of formula (I-B')
其中in
R3和R5各自独立地为-CON(Rd)(Rf),或R3和R5之一为-CON(Rd)(Rf),且R3和R5中的另一个为H或-CO2(Rc);R 3 and R 5 are each independently -CON(R d )(R f ), or one of R 3 and R 5 is -CON(R d )(R f ), and the other of R 3 and R 5 is H or -CO 2 (R c );
Rc为C1-C4烷基;R c is C 1 -C 4 alkyl;
RB1和RB2各自独立地为-CRdRf -; RB1 and RB2 are each independently -CRdRf- ;
B为-卤代(C1-C5烷基)、未取代的-C1-C5烷基或未取代的-C2-C5烯基-;B is -halo(C 1 -C 5 alkyl), unsubstituted -C 1 -C 5 alkyl or unsubstituted -C 2 -C 5 alkenyl-;
RA2和RA1各自独立地为H、卤素、羟基、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-,R A2 and R A1 are each independently H, halogen, hydroxy, optionally substituted (C 1 -C 6 alkyl) or optionally substituted (C 1 -C 6 alkyl)oxy-,
其中所述任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、C1-C4烷氧基、-N(Re)(Rf)、-CO2(Rf)、任选取代的苯基和任选取代的5-6元杂环烷基; 其中所述任选取代的苯基或5-6元杂环烷基任选地被1-4个各自独立地选自如下的取代基所取代:wherein the C 1 -C 6 alkyl group of the optionally substituted (C 1 -C 6 alkyl) or optionally substituted (C 1 -C 6 alkyl ) oxy- is optionally substituted by 1-4 substituents each independently selected from the following: hydroxyl, C 1 -C 4 alkoxy, -N( Re )( Rf ), -CO2 ( Rf ), optionally substituted phenyl and optionally substituted 5-6 membered heterocycloalkyl; wherein the optionally substituted phenyl or 5-6 membered heterocycloalkyl is optionally substituted by 1-4 substituents each independently selected from the following:
C1-C4烷基、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C6烷基)氨基-、(C1-C6烷基)(C1-C6烷基)氨基-、卤代(C1-C6烷基)、羟基-(C1-C4烷基)-、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-和C1-C4烷氧基-(C1-C4烷氧基)-;C 1 -C 4 alkyl, halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 6 alkyl)amino-, (C 1 -C 6 alkyl)(C 1 -C 6 alkyl)amino-, halo(C 1 -C 6 alkyl), hydroxy-(C 1 -C 4 alkyl)-, halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, and C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-;
各Rd独立地为H或C1-C4烷基;Each R d is independently H or C 1 -C 4 alkyl;
Re选自H、(C1-C4烷基)、-CO(C1-C4烷基)、-OCO(C1-C4烷基)或-CO2(C1-C4烷基);R e is selected from H, (C 1 -C 4 alkyl), -CO(C 1 -C 4 alkyl), -OCO(C 1 -C 4 alkyl) or -CO 2 (C 1 -C 4 alkyl);
各Rf为H或(C1-C4烷基);Each R f is H or (C 1 -C 4 alkyl);
R4和R6为H; R4 and R6 are H;
R14为C1-C4烷基;R 14 is C 1 -C 4 alkyl;
RC1为H或C1-C4烷基;R C1 is H or C 1 -C 4 alkyl;
RC2为C1-C4烷基;R C2 is C 1 -C 4 alkyl;
R15为H或C1-C4烷基;R 15 is H or C 1 - C 4 alkyl;
R16为H或C1-C4烷基;R 16 is H or C 1 - C 4 alkyl;
R17为H或C1-C4烷基;和R 17 is H or C 1 -C 4 alkyl; and
RI和RII 在每次出现时独立地为(C1-C6烷基)氧基-,R I and R II are independently (C 1 -C 6 alkyl)oxy- at each occurrence,
RX或Ry中的至少一个独立地为C1-C4烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 4 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C4烷基;or R x and R y are both independently C 1 -C 4 alkyl;
或其互变异构体,or a tautomer thereof,
或其盐。or a salt thereof.
在本发明的一个实施方案中,本发明化合物为式(I-b’),In one embodiment of the present invention, the compound of the present invention is of formula (I-b'),
其中in
B为-卤代(C1-C5烷基)、未取代的-C1-C5烷基,或未取代的-C2-C5烯基-;B is -halo(C 1 -C 5 alkyl), unsubstituted -C 1 -C 5 alkyl, or unsubstituted -C 2 -C 5 alkenyl-;
RA2和RA1各自独立地为H、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-, RA2 and RA1 are each independently H, halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , optionally substituted (C 1 -C 6 alkyl) or optionally substituted (C 1 -C 6 alkyl)oxy-,
其中所述任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、C1-C4烷氧基、-N(Re)(Rf)、-CO2(Rf)、任选取代的苯基和任选取代的5-6元杂环烷基,和其中所述任选取代的苯基或5-6元杂环烷基任选地被1-4个各自独立地选自如下的取代基所取代:C1-C4烷基、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C6烷基)氨基-、(C1-C6烷基)(C1-C6烷基)氨基-、卤代(C1-C6烷基)、羟基-(C1-C4烷基)-、-(C1-C4烷基)-O-P(O)(OH)2、-(C1-C4烷基)-O-P(O)(RIRII)2、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2、-(C1-C6烷基)-NH2、-C1-C4烷基-(C1-C4烷氧基)和C1-C4烷氧基-(C1-C4烷氧基)-;wherein the C 1 -C 6 alkyl group of the optionally substituted (C 1 -C 6 alkyl) or optionally substituted (C 1 -C 6 alkyl ) oxy- is optionally substituted by 1-4 substituents each independently selected from the group consisting of hydroxy, C 1 -C 4 alkoxy, -N( Re )( Rf ), -CO2 ( Rf ), optionally substituted phenyl, and optionally substituted 5-6 membered heterocycloalkyl; and wherein the optionally substituted phenyl or 5-6 membered heterocycloalkyl group is optionally substituted by 1-4 substituents each independently selected from the group consisting of C 1 -C 4 alkyl, halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 6 alkyl)amino-, (C 1 -C 6 alkyl)(C 1 -C 6 alkyl)amino-, halo(C 1 -C 6 alkyl), hydroxy-(C 1 -C 4 alkyl)-, -(C 1 -(C 1 -C 4 alkyl)-OP(O)(OH) 2 , -(C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 , -(C 1 -C 6 alkyl)-NH 2 , -C 1 -C 4 alkyl-(C 1 -C 4 alkoxy) and C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-;
Re选自H、(C1-C4烷基)、-CO(C1-C4烷基)、-OCO(C1-C4烷基)、-(C1-C4烷基)-NH2、-(C1-C4烷基) C1-C4烷氧基或-CO2(C1-C4烷基),R e is selected from H, (C 1 -C 4 alkyl), -CO(C 1 -C 4 alkyl), -OCO(C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)-NH 2 , -(C 1 -C 4 alkyl)C 1 -C 4 alkoxy or -CO 2 (C 1 -C 4 alkyl),
各Rf为H或(C1-C4烷基);Each R f is H or (C 1 -C 4 alkyl);
R14为C1-C4烷基;R 14 is C 1 -C 4 alkyl;
RC2为C1-C4烷基;R C2 is C 1 -C 4 alkyl;
R15为C1-C4烷基;和R 15 is C 1 -C 4 alkyl; and
R17为C1-C4烷基;R 17 is C 1 - C 4 alkyl;
RI和RII 在每次出现时独立地为(C1-C6烷基)氧基-,R I and R II are independently (C 1 -C 6 alkyl)oxy- at each occurrence,
RX或Ry中的至少一个独立地为C1-C4烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 4 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C4烷基;or R x and R y are both independently C 1 -C 4 alkyl;
或其互变异构体,or a tautomer thereof,
或其盐。or a salt thereof.
在一个实施方案中,式(I-B’)或(I-b’)化合物,其中RA2和RA1各自独立地为H、卤素、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-,和所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-N(Re)(Rf)、C1-C4烷氧基、苯基、含有至少一个氮或氧作为环成员的任选取代的5-6元杂环烷基,各Re 独立地选自H、(C1-C4烷基)、-(C1-C4烷基)-NH2或-(C1-C4烷基) C1-C4烷氧基和各Rf独立地为H或(C1-C4烷基)。In one embodiment, the compound of formula (I-B') or (I-b'), wherein RA2 and RA1 are each independently H, halogen, optionally substituted ( C1 - C6 alkyl) or optionally substituted ( C1 - C6 alkyl)oxy-, and the C1- C6 alkyl of the optionally substituted ( C1 - C6 alkyl) or optionally substituted ( C1 - C6 alkyl)oxy- is optionally substituted with 1-4 substituents each independently selected from the group consisting of hydroxy, -OP(O)(OH) 2 , -OP(O)( RIRII ) 2 , -N( Re )( Rf ), C1 - C4 alkoxy, phenyl, an optionally substituted 5-6 membered heterocycloalkyl containing at least one nitrogen or oxygen as a ring member, and each Re is independently selected from H, ( C1 - C4 alkyl), -( C1 - C4 alkyl) -NH2 or -( C1 - C4 alkyl) C1 -C 4 alkoxy and each R f is independently H or (C 1 -C 4 alkyl).
在一个实施方案中,式(I-B’)或(I-b’)化合物,其中RA2和RA1各自独立地为H、卤素、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-,和所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、-N(Re)(Rf)、C1-C4烷氧基、苯基、含有至少一个氮或氧作为环成员的任选取代的5-6元杂环烷基,并且Re和Rf各自独立地为H或(C1-C4烷基)。In one embodiment, the compound of formula (I-B') or (I-b'), wherein RA2 and RA1 are each independently H, halogen, optionally substituted ( C1 - C6 alkyl) or optionally substituted ( C1 - C6 alkyl)oxy-, and the C1- C6 alkyl of the optionally substituted ( C1 - C6 alkyl), optionally substituted (C1-C6 alkyl ) oxy- is optionally substituted with 1-4 substituents each independently selected from the following substituents: hydroxy, -N( Re )( Rf ), C1 - C4 alkoxy, phenyl, optionally substituted 5-6 membered heterocycloalkyl containing at least one nitrogen or oxygen as a ring member, and Re and Rf are each independently H or ( C1 - C4 alkyl).
在一个实施方案中,式(I-B’)或(I-b’)化合物,其中RA2或RA1中的至少一个独立地为H、卤素、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-,和所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1-4个各自独立地选自如下的取代基所取代:-N(Re)(Rf)、四氢吡喃、吡咯烷基、哌嗪基、哌啶基和吗啉基,各Re 独立地选自H、(C1-C4烷基)、-(C1-C4烷基)-NH2或-(C1-C4烷基)C1-C4烷氧基,和各Rf独立地为H或(C1-C4烷基)。In one embodiment, a compound of formula (I-B') or (I-b'), wherein at least one of RA2 or RA1 is independently H, halogen, optionally substituted ( C1 - C6 alkyl), or optionally substituted ( C1 - C6 alkyl)oxy-, and the C1- C6 alkyl of the optionally substituted (C1-C6 alkyl), optionally substituted (C1-C6 alkyl ) oxy- is optionally substituted with 1-4 substituents each independently selected from the group consisting of -N( Re )( Rf ), tetrahydropyran, pyrrolidinyl, piperazinyl, piperidinyl, and morpholinyl, each Re is independently selected from H, ( C1 - C4 alkyl), -( C1 - C4 alkyl)-NH2 , or -( C1 - C4 alkyl) C1 - C4 alkoxy, and each Rf is independently H or ( C1 - C4 alkyl).
在一个实施方案中,式(I-B’)或(I-b’)化合物,其中RA2或RA1中的至少一个各自独立地为H、卤素、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-,并且所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1-4个各自独立地选自如下的取代基所取代:-N(Re)(Rf)、四氢吡喃、吡咯烷基、哌嗪基、哌啶基或吗啉基,并且Re和Rf各自独立地为H或(C1-C4烷基)。In one embodiment, a compound of formula (I-B') or (I-b'), wherein at least one of RA2 or RA1 is each independently H, halogen, optionally substituted ( C1 - C6 alkyl) or optionally substituted ( C1 - C6 alkyl)oxy-, and the C1- C6 alkyl of the optionally substituted ( C1 - C6 alkyl) or optionally substituted ( C1 - C6 alkyl )oxy- is optionally substituted with 1-4 substituents each independently selected from the following substituents: -N( Re )( Rf ), tetrahydropyran, pyrrolidinyl, piperazinyl, piperidinyl or morpholinyl, and Re and Rf are each independently H or ( C1 - C4 alkyl).
在一个实施方案中,式(I-B’)或(I-b’)化合物,其中Rx和Ry各自独立地为甲基或乙基。 在另一个实施方案中,式(I-B’)或(I-b’)化合物,其中Rx和Ry二者都为甲基。在另一个实施方案中,式(I-B’)或(I-b’)化合物,其中Rx和Ry之一为甲基且另一个为H。In one embodiment, the compound of formula (I-B') or (I-b'), wherein Rx and Ry are each independently methyl or ethyl. In another embodiment, the compound of formula (I-B') or (I-b'), wherein both Rx and Ry are methyl. In another embodiment, the compound of formula (I-B') or (I-b'), wherein one of Rx and Ry is methyl and the other is H.
在一个实施方案中,式(I-B’)或(I-b’)化合物,其中In one embodiment, the compound of formula (I-B') or (I-b'), wherein
B为未取代的-C1-C5烷基或未取代的-C2-C5烯基-;B is unsubstituted -C 1 -C 5 alkyl or unsubstituted -C 2 -C 5 alkenyl-;
RA2和RA1各自独立地为H、卤素、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-,R A2 and R A1 are each independently H, halogen, optionally substituted (C 1 -C 6 alkyl) or optionally substituted (C 1 -C 6 alkyl)oxy-,
其中所述任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1-2个各自独立地选自如下的取代基所取代:羟基、C1-C4烷氧基、-N(Re)(Rf)、-CO2(Rf)、未取代的苯基和未取代的5-6元杂环烷基,wherein the C 1 -C 6 alkyl group of the optionally substituted (C 1 -C 6 alkyl) or optionally substituted (C 1 -C 6 alkyl ) oxy- is optionally substituted by 1-2 substituents each independently selected from the following: hydroxyl, C 1 -C 4 alkoxy, -N( Re )( Rf ), -CO2 ( Rf ), unsubstituted phenyl and unsubstituted 5-6 membered heterocycloalkyl,
Re为H、(C1-C4烷基)、-CO(C1-C4烷基)、-OCO(C1-C4烷基)或-CO2(C1-C4烷基),R e is H, (C 1 -C 4 alkyl), -CO(C 1 -C 4 alkyl), -OCO(C 1 -C 4 alkyl) or -CO 2 (C 1 -C 4 alkyl),
Rf每次出现时为H或(C1-C4烷基);R f at each occurrence is H or (C 1 -C 4 alkyl);
R14为C1-C4烷基;R 14 is C 1 -C 4 alkyl;
RC2为C1-C4烷基;R C2 is C 1 -C 4 alkyl;
R15为C1-C4烷基;和R 15 is C 1 -C 4 alkyl; and
R17为C1-C4烷基;R 17 is C 1 - C 4 alkyl;
RI和RII 在每次出现时独立地为(C1-C6烷基)氧基-,R I and R II are independently (C 1 -C 6 alkyl)oxy- at each occurrence,
RX或Ry中的至少一个独立地为C1-C2烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 2 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C2烷基;or R x and R y are both independently C 1 -C 2 alkyl;
或其互变异构体,or a tautomer thereof,
或其盐。or a salt thereof.
在一个实施方案中,式(I-b’)化合物,其中In one embodiment, the compound of formula (I-b') wherein
B为未取代的-C2-C5烯基-;B is unsubstituted -C 2 -C 5 alkenyl-;
RA2和RA1各自独立地为H、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-,R A2 and R A1 are each independently H, optionally substituted (C 1 -C 6 alkyl) or optionally substituted (C 1 -C 6 alkyl)oxy-,
其中所述任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1个各自独立地选自如下的取代基所取代:羟基、C1-C4烷氧基、未取代的5-6元杂环烷基,wherein the optionally substituted (C 1 -C 6 alkyl) or optionally substituted (C 1 -C 6 alkyl)oxy-C 1 -C 6 alkyl is optionally substituted by 1 substituent independently selected from the following: hydroxyl, C 1 -C 4 alkoxy, unsubstituted 5-6 membered heterocycloalkyl,
R14为C1-C4烷基;R 14 is C 1 -C 4 alkyl;
RC2为C1-C4烷基;R C2 is C 1 -C 4 alkyl;
R15为C1-C4烷基;和R 15 is C 1 -C 4 alkyl; and
R17为C1-C4烷基;R 17 is C 1 - C 4 alkyl;
RX或Ry中的至少一个独立地为C1-C2烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 2 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C2烷基;or R x and R y are both independently C 1 -C 2 alkyl;
或其互变异构体,or a tautomer thereof,
或其盐。or a salt thereof.
在一个实施方案中,式(I-b’)化合物,其中In one embodiment, the compound of formula (I-b') wherein
B为未取代的乙烯基;B is an unsubstituted vinyl group;
RA2和RA1各自独立地为H或任选取代的(C1-C6烷基)氧基-,R A2 and R A1 are each independently H or an optionally substituted (C 1 -C 6 alkyl)oxy-,
其中所述任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被一个选自羟基或未取代的吗啉基的取代基取代;wherein the C 1 -C 6 alkyl group of the optionally substituted (C 1 -C 6 alkyl)oxy-group is optionally substituted with a substituent selected from hydroxyl or unsubstituted morpholinyl;
R14为甲基或乙基;R 14 is methyl or ethyl;
RC2为甲基或乙基;R C2 is methyl or ethyl;
R15为甲基或乙基;和 R15 is methyl or ethyl; and
R17为甲基或乙基;R 17 is methyl or ethyl;
RX或Ry中的至少一个独立地为C1-C2烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 2 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C2烷基;or R x and R y are both independently C 1 -C 2 alkyl;
或其互变异构体,or a tautomer thereof,
或其盐。or a salt thereof.
在一个实施方案中,式(I-1)化合物,In one embodiment, the compound of formula (I-1),
其中in
P为1至6之一的整数;P is an integer from 1 to 6;
RA和RB 独立地为H或C1-C4烷基; RA and RB are independently H or C1 - C4 alkyl;
或N、RA和RB形成任选取代的5或6元杂环,or N, RA and RB form an optionally substituted 5- or 6-membered heterocyclic ring,
其中所述杂环选自吗啉基、哌啶基、哌嗪基和吡咯烷基,和wherein the heterocyclic ring is selected from morpholinyl, piperidinyl, piperazinyl and pyrrolidinyl, and
所述杂环任选地被一个或两个独立地选自如下的取代基取代:羟基和任选地被一个或两个为羟基或C1-C3 烷氧基的取代基取代的C1-C3 烷基;The heterocycle is optionally substituted with one or two substituents independently selected from the group consisting of hydroxy and C 1 -C 3 alkyl optionally substituted with one or two substituents which are hydroxy or C 1 -C 3 alkoxy;
RX或Ry中的至少一个独立地为C1-C2烷基且另一个为H;At least one of RX or Ry is independently C1 - C2 alkyl and the other is H;
或RX和Ry二者都独立地为C1-C2烷基;or R x and R y are both independently C 1 -C 2 alkyl;
或其互变异构体,or a tautomer thereof,
或其盐。or a salt thereof.
在一个实施方案中,式(I-1)化合物,其中Rx和Ry各自独立地为甲基或乙基。 在另一个实施方案中,式(I-1)化合物,其中Rx和Ry二者都为甲基。 在另一个实施方案中,式(I-1)化合物,其中Rx和Ry之一为甲基且另一个为H。In one embodiment, the compound of formula (I-1), wherein Rx and Ry are each independently methyl or ethyl. In another embodiment, the compound of formula (I-1), wherein both Rx and Ry are methyl. In another embodiment, the compound of formula (I-1), wherein one of Rx and Ry is methyl and the other is H.
在一个实施方案中,本发明化合物具有式(I-bc)In one embodiment, the compound of the invention has the formula (I-bc)
其中in
RC1和RC2各自独立地为-CH2-,R C1 and R C2 are each independently -CH 2 -,
C为-卤代(C1-C5烷基)、未取代的-C1-C5烷基或未取代的-C2-C5烯基-;C is -halo(C 1 -C 5 alkyl), unsubstituted -C 1 -C 5 alkyl or unsubstituted -C 2 -C 5 alkenyl-;
RB1和RB2各自独立地为-CRdRf -; RB1 and RB2 are each independently -CRdRf- ;
B为-卤代(C1-C5烷基)、未取代的-C1-C5烷基或未取代的-C2-C5烯基-;B is -halo(C 1 -C 5 alkyl), unsubstituted -C 1 -C 5 alkyl or unsubstituted -C 2 -C 5 alkenyl-;
RA2和RA1各自独立地为H、卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-, RA2 and RA1 are each independently H, halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , optionally substituted (C 1 -C 6 alkyl) or optionally substituted (C 1 -C 6 alkyl)oxy-,
其中所述任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、C1-C4烷氧基、-N(Re)(Rf)、-CO2(Rf)、任选取代的苯基和任选取代的5-6元杂环烷基; 其中所述任选取代的苯基或5-6元杂环烷基任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、氨基、(C1-C6烷基)氨基-、(C1-C6烷基)(C1-C6烷基)氨基-、卤代(C1-C6烷基)、羟基-(C1-C4烷基)-、-(C1-C4烷基)-O-P(O)(OH)2、-(C1-C4烷基)-O-P(O)(RIRII)2、卤代(C1-C4烷氧基)-、C1-C4烷氧基-、羟基-(C2-C4烷氧基)-、-(C2-C4烷氧基)-O-P(O)(OH)2、-(C2-C4烷氧基)-O-P(O)(RIRII)2、-(C1-C6烷基)-NH2和C1-C4烷氧基-(C1-C4烷氧基)-;wherein the C 1 -C 6 alkyl group of the optionally substituted (C 1 -C 6 alkyl) or optionally substituted (C 1 -C 6 alkyl)oxy- is optionally substituted by 1-4 substituents each independently selected from the group consisting of hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , C 1 -C 4 alkoxy, -N( Re )(R f ), -CO 2 (R f ), optionally substituted phenyl, and optionally substituted 5-6 membered heterocycloalkyl; wherein the optionally substituted phenyl or 5-6 membered heterocycloalkyl is optionally substituted by 1-4 substituents each independently selected from the group consisting of halogen, hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , amino, (C 1 -C 6 alkyl)amino-, (C 1 -C 6 alkyl)(C 1 -C 6 alkyl)amino-, halo(C 1 -C 4 alkoxy) 6 alkyl), hydroxy-(C 1 -C 4 alkyl)-, -(C 1 -C 4 alkyl)-OP(O)(OH) 2 , -(C 1 -C 4 alkyl)-OP(O)(R I R II ) 2 , halo(C 1 -C 4 alkoxy)-, C 1 -C 4 alkoxy-, hydroxy-(C 2 -C 4 alkoxy)-, -(C 2 -C 4 alkoxy)-OP(O)(OH) 2 , -(C 2 -C 4 alkoxy)-OP(O)(R I R II ) 2 , -(C 1 -C 6 alkyl)-NH 2 and C 1 -C 4 alkoxy-(C 1 -C 4 alkoxy)-;
各Rd独立地为H或C1-C4烷基;Each R d is independently H or C 1 -C 4 alkyl;
Re选自H、(C1-C4烷基)、-CO(C1-C4烷基)、-OCO(C1-C4烷基)、-(C1-C4烷基)-NH2、-(C1-C4烷基) C1-C4烷氧基或-CO2(C1-C4烷基),R e is selected from H, (C 1 -C 4 alkyl), -CO(C 1 -C 4 alkyl), -OCO(C 1 -C 4 alkyl), -(C 1 -C 4 alkyl)-NH 2 , -(C 1 -C 4 alkyl)C 1 -C 4 alkoxy or -CO 2 (C 1 -C 4 alkyl),
各Rf为H或(C1-C4烷基);Each R f is H or (C 1 -C 4 alkyl);
R6为H; R6 is H;
R14为C1-C4烷基;R 14 is C 1 -C 4 alkyl;
R15为C1-C4烷基;R 15 is C 1 - C 4 alkyl;
R16为C1-C4烷基;R 16 is C 1 - C 4 alkyl;
R17为C1-C4烷基;和R 17 is C 1 -C 4 alkyl; and
RI和RII 在每次出现时独立地为(C1-C6烷基)氧基-,R I and R II are independently (C 1 -C 6 alkyl)oxy- at each occurrence,
RX或Ry中的至少一个独立地为C1-C4烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 4 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C4烷基;or R x and R y are both independently C 1 -C 4 alkyl;
或其互变异构体;or a tautomer thereof;
或其盐,特别是其药学上可接受的盐。or a salt thereof, in particular a pharmaceutically acceptable salt thereof.
在一个实施方案中,式(I-bc)化合物,其中RA2和RA1各自独立地为H、卤素、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-,且所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、-O-P(O)(OH)2、-O-P(O)(RIRII)2、-N(Re)(Rf)、C1-C4烷氧基、苯基、含有至少一个氮或氧作为环成员的任选取代的5-6元杂环烷基,各Re 独立地选自H、C1-C4烷基、-(C1-C4烷基)-NH2或-(C1-C4烷基) C1-C4烷氧基,和各Rf独立地为H或(C1-C4烷基)。In one embodiment, a compound of formula (I-bc) wherein R and R are each independently H, halogen, optionally substituted (C 1 -C 6 alkyl) or optionally substituted (C 1 -C 6 alkyl)oxy-, and the C 1 -C 6 alkyl of the optionally substituted (C 1 -C 6 alkyl)oxy- is optionally substituted with 1-4 substituents each independently selected from the group consisting of hydroxy, -OP(O)(OH) 2 , -OP(O)(R I R II ) 2 , -N( Re )(R f ), C 1 -C 4 alkoxy, phenyl, an optionally substituted 5-6 membered heterocycloalkyl containing at least one nitrogen or oxygen as a ring member, each Re is independently selected from H , C 1 -C 4 alkyl, -(C 1 -C 4 alkyl)-NH 2 or -(C 1 -C 4 alkyl)C 1 -C 4 alkoxy, and each R f is independently H or (C 1 -C 4 alkyl).
在一个实施方案中,式(I-bc)化合物,其中RA2或RA1中的至少一个独立地为H、卤素、任选取代的(C1-C6烷基)或任选取代的(C1-C6烷基)氧基-,且所述任选取代的(C1-C6烷基)、任选取代的(C1-C6烷基)氧基-的C1-C6烷基任选地被1-4个各自独立地选自如下的取代基所取代:-N(Re)(Rf)、四氢吡喃、吡咯烷基、哌嗪基、哌啶基和吗啉基,各Re 独立地选自H、C1-C4烷基、-(C1-C4烷基)-NH2或-(C1-C4烷基) C1-C4烷氧基,和各Rf独立地为H或C1-C4烷基。In one embodiment, a compound of formula (I-bc) wherein at least one of RA2 or RA1 is independently H, halogen, optionally substituted ( C1 - C6 alkyl), or optionally substituted ( C1 - C6 alkyl)oxy-, and the C1- C6 alkyl of the optionally substituted ( C1 - C6 alkyl), optionally substituted ( C1 - C6 alkyl)oxy- is optionally substituted with 1-4 substituents each independently selected from the group consisting of -N( Re )( Rf ), tetrahydropyran, pyrrolidinyl, piperazinyl, piperidinyl, and morpholinyl, each Re is independently selected from H, C1 - C4 alkyl, -( C1 - C4 alkyl) -NH2 , or -( C1 - C4 alkyl) C1 - C4 alkoxy, and each Rf is independently H or C1 - C4 alkyl.
在一个实施方案中,式I-2化合物,In one embodiment, the compound of formula I-2,
其中in
R14、R15、Rc2和R17独立地为C1-C3烷基;R 14 , R 15 , R c2 and R 17 are independently C 1 -C 3 alkyl;
RA1和RA2 独立地为H、羟基、COOH或任选取代的(C1-C6烷基)氧基-,R A1 and R A2 are independently H, hydroxy, COOH or optionally substituted (C 1 -C 6 alkyl)oxy-,
其中任选取代的(C1-C6烷基)氧基-的烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、-CO2(Rf)、-N(Re)(Rf)、任选取代的苯基和任选取代的5-6元杂环烷基,wherein the alkyl group of the optionally substituted (C 1 -C 6 alkyl)oxy-group is optionally substituted by 1 to 4 substituents each independently selected from the group consisting of hydroxy, -CO 2 (R f ), -N(R e )(R f ), optionally substituted phenyl and optionally substituted 5-6 membered heterocycloalkyl,
其中所述任选取代的苯基或5-6元杂环烷基任选地被1-4个各自独立地选自如下的取代基所取代:(C1-C4烷基)氧基-和C1-C4烷基;wherein the optionally substituted phenyl or 5-6 membered heterocycloalkyl is optionally substituted by 1-4 substituents each independently selected from the following: (C 1 -C 4 alkyl)oxy- and C 1 -C 4 alkyl;
R3和R5各自独立地为-CO-N(Rd)(Rf), R3 and R5 are each independently -CO-N( Rd )( Rf ),
各Rd、Re和Rf独立地为H或C1-C3烷基;Each of R d , Re and R f is independently H or C 1 -C 3 alkyl;
B为取代的-C1-C4烷基-或取代的-C2-C4烯基-,B is a substituted -C 1 -C 4 alkyl- or a substituted -C 2 -C 4 alkenyl-,
其中所述取代的-C1-C4烷基-或取代的-C2-C4烯基-的烷基部分被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、(C1-C4烷基)氧基-和C1-4烷基,wherein the alkyl portion of the substituted -C 1 -C 4 alkyl- or substituted -C 2 -C 4 alkenyl- is substituted by 1 to 4 substituents independently selected from the group consisting of halogen, hydroxy, (C 1 -C 4 alkyl)oxy- and C 1-4 alkyl,
RX或Ry中的至少一个独立地为C1-C4烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 4 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C4烷基;or R x and R y are both independently C 1 -C 4 alkyl;
或其互变异构体,or a tautomer thereof,
或其盐(特别是其药学上可接受的盐)。or a salt thereof (especially a pharmaceutically acceptable salt thereof).
在一个实施方案中,式(I-2)化合物,R14、R15、Rc2和R17独立地为甲基或乙基。In one embodiment, in the compound of formula (I-2), R 14 , R 15 , R c2 and R 17 are independently methyl or ethyl.
在一个实施方案中,式(I-2)化合物,R14和Rc2为乙基且R15和R17为甲基。In one embodiment, in the compound of formula (I-2), R 14 and R c2 are ethyl and R 15 and R 17 are methyl.
在一个实施方案中,式(I-2)化合物,R14、R15、Rc2和R17为甲基。In one embodiment, in the compound of formula (I-2), R 14 , R 15 , R c2 and R 17 are methyl.
在一个实施方案中,式(I-2)化合物,RA1和RA2 独立地为H、羟基或任选取代的(C1-C6烷基)氧基-,In one embodiment, in the compound of formula (I-2), R A1 and R A2 are independently H, hydroxy or optionally substituted (C 1 -C 6 alkyl)oxy-,
其中任选取代的(C1-C6烷基)氧基-的烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、COOH和任选取代的苯基,wherein the alkyl group of the optionally substituted (C 1 -C 6 alkyl)oxy-group is optionally substituted by 1 to 4 substituents each independently selected from the group consisting of hydroxyl, COOH and optionally substituted phenyl,
其中所述任选取代的苯基任选地被1-4个各自独立地选自如下的取代基所取代:(C1-C4烷基)氧基-。The optionally substituted phenyl group is optionally substituted by 1 to 4 substituents each independently selected from the following: (C 1 -C 4 alkyl)oxy-.
在一个实施方案中,式(I-2)化合物,RA1和RA2之一为H且RA1和RA2中的另一个为羟基或任选取代的(C1-C6烷基)氧基-,In one embodiment, in the compound of formula (I-2), one of RA1 and RA2 is H and the other of RA1 and RA2 is hydroxy or optionally substituted (C 1 -C 6 alkyl)oxy-,
其中任选取代的(C1-C6烷基)氧基-的烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、COOH和任选取代的苯基,wherein the alkyl group of the optionally substituted (C 1 -C 6 alkyl)oxy-group is optionally substituted by 1 to 4 substituents each independently selected from the group consisting of hydroxyl, COOH and optionally substituted phenyl,
其中所述任选取代的苯基任选地被1-4个各自独立地选自如下的取代基所取代:(C1-C4烷基)氧基-。The optionally substituted phenyl group is optionally substituted by 1 to 4 substituents each independently selected from the following: (C 1 -C 4 alkyl)oxy-.
在一个实施方案中,式(I-2)化合物,RA1和RA2之一为H且RA1和RA2中的另一个为羟基或任选取代的(C1-C4烷基)氧基-,In one embodiment, in the compound of formula (I-2), one of RA1 and RA2 is H and the other of RA1 and RA2 is hydroxy or optionally substituted (C 1 -C 4 alkyl)oxy-,
其中任选取代的(C1-C4烷基)氧基-的烷基任选地被1-2个各自独立地选自如下的取代基所取代:羟基和任选取代的苯基,wherein the alkyl group of the optionally substituted (C 1 -C 4 alkyl)oxy-group is optionally substituted by 1 to 2 substituents each independently selected from the following: hydroxyl and optionally substituted phenyl,
其中所述任选取代的苯基任选地被1-2个各自独立地选自以下的取代基取代:(C1-C4烷基)氧基-。The optionally substituted phenyl group is optionally substituted by 1-2 substituents each independently selected from the following: (C 1 -C 4 alkyl)oxy-.
在一个实施方案中,式(I-2)化合物,RA1和RA2二者都为H。In one embodiment, in the compound of formula (I-2), both RA1 and RA2 are H.
在一个实施方案中,式(I-2)化合物,其中RA1和RA2中的至少一个不是H。In one embodiment, the compound of formula (I-2) wherein at least one of RA1 and RA2 is not H.
在一个实施方案中,式(I-2)化合物,RA1和RA2 各自独立地为任选取代的(C1-C4烷基)氧基-,其中任选取代的(C1-C4烷基)氧基-的烷基任选地被1-2个羟基取代基取代。In one embodiment, in the compound of formula (I-2), RA1 and RA2 are each independently optionally substituted ( C1 - C4 alkyl)oxy-, wherein the alkyl group of the optionally substituted ( C1 - C4 alkyl)oxy- is optionally substituted with 1-2 hydroxy substituents.
在一个实施方案中,式(I-2)化合物,R3和R5 独立地选自-CO-N(Rd)(Rf),且Rd和Rf各自独立地为H或C1-C3烷基。In one embodiment, in the compound of formula (I-2), R 3 and R 5 are independently selected from -CO-N(R d )(R f ), and R d and R f are each independently H or C 1 -C 3 alkyl.
在一个实施方案中,式(I-2)化合物,R3和R5为-CO-NH2。In one embodiment, in the compound of formula (I-2), R 3 and R 5 are -CO-NH 2 .
在一个实施方案中,式(I-2)化合物,B为取代的-C1-C4烷基-或取代的-C2-C4烯基-,In one embodiment, in the compound of formula (I-2), B is substituted -C 1 -C 4 alkyl- or substituted -C 2 -C 4 alkenyl-,
其中所述取代的-C1-C4烷基-或取代的-C2-C4烯基-的烷基部分被1-4个各自独立地选自如下的取代基所取代:卤素和C1-4烷基。The alkyl portion of the substituted -C 1 -C 4 alkyl- or substituted -C 2 -C 4 alkenyl- is substituted with 1 to 4 substituents independently selected from the following: halogen and C 1-4 alkyl.
在一个实施方案中,式(I-2)化合物,B为取代的-CH2-CH2-或取代的-CH=CH-,In one embodiment, in the compound of formula (I-2), B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个各自独立地选自如下的取代基所取代:卤素和C1-4烷基。The substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 substituents independently selected from the following: halogen and C 1-4 alkyl.
在式(I-2)化合物的一个实施方案中,B为取代的-CH2-CH2-或取代的-CH=CH-,In one embodiment of the compound of formula (I-2), B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个各自独立地选自如下的取代基所取代:氟和C1-2烷基。The substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 substituents independently selected from the following: fluorine and C 1-2 alkyl.
在式(I-2)化合物的一个实施方案中,B为取代的-CH2-CH2-或取代的-CH=CH-,In one embodiment of the compound of formula (I-2), B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个氟取代基取代。wherein the substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 fluorine substituents.
在式(I-2)化合物的一个实施方案中,B为取代的-CH2-CH2-或取代的-CH=CH-,In one embodiment of the compound of formula (I-2), B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个各自独立地为C1-2烷基的取代基取代。The substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 substituents each independently being a C 1-2 alkyl group.
在式(I-2)化合物的一个实施方案中,B为取代的-CH2-CH2-或取代的-CH=CH-,In one embodiment of the compound of formula (I-2), B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个羟基取代基取代。wherein the substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 hydroxy substituents.
在式(I-2)化合物的一个实施方案中,B为被1-2个羟基取代基所取代的-CH2-CH2-。In one embodiment of the compounds of formula (I-2), B is -CH 2 -CH 2 - substituted with 1-2 hydroxy substituents.
在一个实施方案中,式(I-2)化合物,其中Rx和Ry各自独立地为甲基或乙基。在另一个实施方案中,式(I-2)化合物,其中Rx和Ry二者都为甲基。在另一个实施方案中,式(I-2)化合物,其中Rx和Ry之一为甲基且另一个为H。In one embodiment, the compound of formula (I-2), wherein Rx and Ry are each independently methyl or ethyl. In another embodiment, the compound of formula (I-2), wherein both Rx and Ry are methyl. In another embodiment, the compound of formula (I-2), wherein one of Rx and Ry is methyl and the other is H.
在一个实施方案中,式(I-2)化合物,其中In one embodiment, the compound of formula (I-2), wherein
R14、R15、Rc2和R17独立地为甲基或乙基;R 14 , R 15 , R c2 and R 17 are independently methyl or ethyl;
RA1和RA2之一为H且RA1和RA2中的另一个为任选取代的(C1-C4烷基)氧基-,One of R A1 and R A2 is H and the other of R A1 and R A2 is optionally substituted (C 1 -C 4 alkyl)oxy-,
其中任选取代的(C1-C4烷基)氧基-的烷基任选地被1-2个各自独立地选自如下的取代基所取代:羟基和任选取代的苯基,wherein the alkyl group of the optionally substituted (C 1 -C 4 alkyl)oxy-group is optionally substituted by 1 to 2 substituents independently selected from the following: hydroxyl and optionally substituted phenyl,
其中所述任选取代的苯基任选地被1-2个各自独立地选自以下的取代基取代:(C1-C4烷基)氧基-;wherein the optionally substituted phenyl is optionally substituted by 1-2 substituents each independently selected from the following: (C 1 -C 4 alkyl)oxy-;
R3和R5二者都为-CO-NH2;和 R3 and R5 are both -CO- NH2 ; and
B为取代的-CH2-CH2-或取代的-CH=CH-,B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个各自独立地选自如下的取代基所取代:氟和C1-2烷基;和wherein the substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 substituents independently selected from the following: fluorine and C 1-2 alkyl; and
RX或Ry中的至少一个独立地为C1-C4烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 4 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C4烷基。Or both RX and Ry are independently C1 - C4 alkyl.
在一个实施方案中,式(I-2)化合物,其中In one embodiment, the compound of formula (I-2), wherein
R14、R15、Rc2和R17独立地为甲基或乙基;R 14 , R 15 , R c2 and R 17 are independently methyl or ethyl;
RA1和RA2之一为H且RA1和RA2中的另一个为任选取代的(C1-C4烷基)氧基-,One of R A1 and R A2 is H and the other of R A1 and R A2 is optionally substituted (C 1 -C 4 alkyl)oxy-,
其中任选取代的(C1-C4烷基)氧基-的烷基任选地被1-2个羟基取代基取代;wherein the alkyl group of the optionally substituted (C 1 -C 4 alkyl)oxy-group is optionally substituted with 1-2 hydroxy substituents;
R3和R5二者都为-CO-NH2;和 R3 and R5 are both -CO- NH2 ; and
B为取代的-CH2-CH2-或取代的-CH=CH-,B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个各自独立地选自如下的取代基所取代:羟基和C1-2烷基;和wherein the substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 substituents independently selected from the following: hydroxyl and C 1-2 alkyl; and
RX或Ry中的至少一个独立地为C1-C4烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 4 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C4烷基。Or both RX and Ry are independently C1 - C4 alkyl.
在一个实施方案中,式I-3化合物,In one embodiment, the compound of formula I-3,
其中in
R14、R15、Rc2和R17独立地为C1-C3烷基;R 14 , R 15 , R c2 and R 17 are independently C 1 -C 3 alkyl;
RA1和RA2 独立地为H、羟基、卤素、COOH或任选取代的(C1-C6烷基)氧基-、R A1 and R A2 are independently H, hydroxy, halogen, COOH or optionally substituted (C 1 -C 6 alkyl)oxy-,
其中任选取代的(C1-C6烷基)氧基-的烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、-CO2(Rf)、-N(Re)(Rf)、(C1-C4烷基)氧基-、任选取代的苯基和任选取代的5-6元杂环烷基,wherein the alkyl group of the optionally substituted (C 1 -C 6 alkyl)oxy-group is optionally substituted by 1 to 4 substituents each independently selected from the group consisting of hydroxy, -CO 2 (R f ), -N( Re )(R f ), (C 1 -C 4 alkyl)oxy-, optionally substituted phenyl and optionally substituted 5-6 membered heterocycloalkyl,
其中所述任选取代的苯基或5-6元杂环烷基任选地被1-4个各自独立地选自如下的取代基所取代:卤素、(C1-C4烷基)氧基-和C1-C4烷基;wherein the optionally substituted phenyl or 5-6 membered heterocycloalkyl is optionally substituted by 1-4 substituents each independently selected from the group consisting of halogen, (C 1 -C 4 alkyl)oxy- and C 1 -C 4 alkyl;
R3和R5各自独立地为-CO-N(Rd)(Rf),或R3和R5之一为-CO-N(Rd)(Rf),且R3和R5中的另一个为H或CO2(RC);R 3 and R 5 are each independently -CO-N(R d )(R f ), or one of R 3 and R 5 is -CO-N(R d )(R f ), and the other of R 3 and R 5 is H or CO 2 (R C );
R4和R6各自独立地为H或卤素, R4 and R6 are each independently H or halogen,
各Rc、Rd、Re和Rf独立地为H或C1-C4烷基;Each of R c , R d , Re and R f is independently H or C 1 -C 4 alkyl;
B为任选取代的-C1-C4烷基-或任选取代的-C2-C4烯基-,B is an optionally substituted -C 1 -C 4 alkyl- or an optionally substituted -C 2 -C 4 alkenyl-,
其中所述任选取代的-C1-C4烷基-或任选取代的-C2-C4烯基-的烷基部分任选地被1-4个各自独立地选自如下的取代基所取代:卤素、羟基、(C1-C4烷基)氧基-和C1-4烷基;和wherein the alkyl portion of the optionally substituted -C 1 -C 4 alkyl- or optionally substituted -C 2 -C 4 alkenyl- is optionally substituted with 1 to 4 substituents each independently selected from the group consisting of halogen, hydroxy, (C 1 -C 4 alkyl)oxy- and C 1-4 alkyl; and
RX或Ry中的至少一个独立地为C1-C4烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 4 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C4烷基;or R x and R y are both independently C 1 -C 4 alkyl;
或其互变异构体,or a tautomer thereof,
或其盐(特别是其药学上可接受的盐)。or a salt thereof (especially a pharmaceutically acceptable salt thereof).
在一个实施方案中,式(I-3)化合物,R14、R15、Rc2和R17独立地为甲基或乙基。In one embodiment, in the compound of formula (I-3), R 14 , R 15 , R c2 and R 17 are independently methyl or ethyl.
在一个实施方案中,式(I-3)化合物,R14和Rc2为乙基,且R15和R17为甲基。In one embodiment, in the compound of formula (I-3), R 14 and R c2 are ethyl, and R 15 and R 17 are methyl.
在一个实施方案中,式(I-3)化合物,RA1和RA2 独立地为H、羟基或任选取代的(C1-C6烷基)氧基-,In one embodiment, in the compound of formula (I-3), R A1 and R A2 are independently H, hydroxy or optionally substituted (C 1 -C 6 alkyl)oxy-,
其中任选取代的(C1-C6烷基)氧基-的烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、COOH和任选取代的苯基,wherein the alkyl group of the optionally substituted (C 1 -C 6 alkyl)oxy-group is optionally substituted by 1 to 4 substituents each independently selected from the group consisting of hydroxyl, COOH and optionally substituted phenyl,
其中所述任选取代的苯基任选地被1-4个各自独立地选自如下的取代基所取代:(C1-C4烷基)氧基-。The optionally substituted phenyl group is optionally substituted by 1 to 4 substituents each independently selected from the following: (C 1 -C 4 alkyl)oxy-.
在一个实施方案中,式(I-3)化合物,RA1和RA2之一为H且RA1和RA2中的另一个为羟基或任选取代的(C1-C6烷基)氧基-,In one embodiment, in the compound of formula (I-3), one of RA1 and RA2 is H and the other of RA1 and RA2 is hydroxy or optionally substituted (C 1 -C 6 alkyl)oxy-,
其中任选取代的(C1-C6烷基)氧基-的烷基任选地被1-4个各自独立地选自如下的取代基所取代:羟基、COOH和任选取代的苯基,wherein the alkyl group of the optionally substituted (C 1 -C 6 alkyl)oxy-group is optionally substituted by 1 to 4 substituents each independently selected from the following: hydroxyl, COOH and optionally substituted phenyl,
其中所述任选取代的苯基任选地被1-4个各自独立地选自如下的取代基所取代:(C1-C4烷基)氧基-。The optionally substituted phenyl group is optionally substituted by 1 to 4 substituents each independently selected from the following: (C 1 -C 4 alkyl)oxy-.
在一个实施方案中,式(I-3)化合物,RA1和RA2之一为H且RA1和RA2中的另一个为羟基或任选取代的(C1-C4烷基)氧基-,In one embodiment, in the compound of formula (I-3), one of RA1 and RA2 is H and the other of RA1 and RA2 is hydroxy or optionally substituted (C 1 -C 4 alkyl)oxy-,
其中任选取代的(C1-C4烷基)氧基-的烷基任选地被1-2个各自独立地选自如下的取代基所取代:羟基和任选取代的苯基,wherein the alkyl group of the optionally substituted (C 1 -C 4 alkyl)oxy-group is optionally substituted by 1 to 2 substituents independently selected from the following: hydroxyl and optionally substituted phenyl,
其中所述任选取代的苯基任选地被1-2个各自独立地选自以下的取代基取代:(C1-C4烷基)氧基-。The optionally substituted phenyl group is optionally substituted by 1-2 substituents each independently selected from the following substituents: (C 1 -C 4 alkyl)oxy-.
在一个实施方案中,式(I-3)化合物,RA1和RA2 各自独立地为任选取代的(C1-C4烷基)氧基-,其中任选取代的(C1-C4烷基)氧基-的烷基任选地被1-2个羟基取代基取代。In one embodiment, in the compound of formula (I-3), RA1 and RA2 are each independently optionally substituted ( C1 - C4 alkyl)oxy-, wherein the alkyl group of the optionally substituted ( C1 - C4 alkyl)oxy- is optionally substituted with 1-2 hydroxy substituents.
在一个实施方案中,式(I-3)化合物,R3和R5为-CO-NH2。In one embodiment, in the compound of formula (I-3), R 3 and R 5 are -CO-NH 2 .
在一个实施方案中,式(I-3)化合物,B为取代的-CH2-CH2-或取代的-CH=CH-,In one embodiment, in the compound of formula (I-3), B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个各自独立地选自如下的取代基所取代:卤素和C1-4烷基。The substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 substituents independently selected from the following: halogen and C 1-4 alkyl.
在式(I-3)化合物的一个实施方案中,B为取代的-CH2-CH2-或取代的-CH=CH-,In one embodiment of the compound of formula (I-3), B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个各自独立地选自如下的取代基所取代:氟和C1-2烷基。The substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 substituents independently selected from the following: fluorine and C 1-2 alkyl.
在式(I-3)化合物的一个实施方案中,B为取代的-CH2-CH2-或取代的-CH=CH-,In one embodiment of the compound of formula (I-3), B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个氟取代基取代。wherein the substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 fluorine substituents.
在式(I-3)化合物的一个实施方案中,B为取代的-CH2-CH2-或取代的-CH=CH-,In one embodiment of the compound of formula (I-3), B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个各自独立地为C1-2烷基的取代基取代。The substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 substituents each independently being a C 1-2 alkyl group.
在式(I-3)化合物的一个实施方案中,B为取代的-CH2-CH2-或取代的-CH=CH-,In one embodiment of the compound of formula (I-3), B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个羟基取代基取代。wherein the substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 hydroxy substituents.
在式(I-3)化合物的一个实施方案中,B为被1-2个羟基取代基所取代的-CH2-CH2-。In one embodiment of the compounds of formula (I-3), B is -CH 2 -CH 2 - substituted with 1-2 hydroxy substituents.
在一个实施方案中,式(I-3)化合物,其中Rx和Ry各自独立地为甲基或乙基。在另一个实施方案中,式(I-3)化合物,其中Rx和Ry二者都为甲基。在另一个实施方案中,式(I-3)化合物,其中Rx和Ry之一为甲基且另一个为H。In one embodiment, the compound of formula (I-3), wherein Rx and Ry are each independently methyl or ethyl. In another embodiment, the compound of formula (I-3), wherein both Rx and Ry are methyl. In another embodiment, the compound of formula (I-3), wherein one of Rx and Ry is methyl and the other is H.
在一个实施方案中,式(I-3)化合物,其中In one embodiment, the compound of formula (I-3), wherein
R14、R15、Rc2和R17独立地为甲基或乙基;R 14 , R 15 , R c2 and R 17 are independently methyl or ethyl;
RA1和RA2之一为H且RA1和RA2中的另一个为任选取代的(C1-C4烷基)氧基-,One of R A1 and R A2 is H and the other of R A1 and R A2 is optionally substituted (C 1 -C 4 alkyl)oxy-,
其中任选取代的(C1-C4烷基)氧基-的烷基任选地被1-2个各自独立地选自如下的取代基所取代:羟基和任选取代的苯基,wherein the alkyl group of the optionally substituted (C 1 -C 4 alkyl)oxy-group is optionally substituted by 1 to 2 substituents each independently selected from the following: hydroxyl and optionally substituted phenyl,
其中所述任选取代的苯基任选地被1-2个各自独立地选自以下的取代基取代:(C1-C4烷基)氧基-;wherein the optionally substituted phenyl is optionally substituted by 1-2 substituents each independently selected from the following: (C 1 -C 4 alkyl)oxy-;
R3和R5二者都为-CO-NH2;和 R3 and R5 are both -CO- NH2 ; and
B为取代的-CH2-CH2-或取代的-CH=CH-,B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个各自独立地选自如下的取代基所取代:氟和C1-2烷基;和wherein the substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 substituents independently selected from the following: fluorine and C 1-2 alkyl; and
RX或Ry中的至少一个独立地为C1-C4烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 4 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C4烷基。Or both RX and Ry are independently C1 - C4 alkyl.
在一个实施方案中,式(I-3)化合物,其中In one embodiment, the compound of formula (I-3), wherein
R14、R15、Rc2和R17独立地为甲基或乙基;R 14 , R 15 , R c2 and R 17 are independently methyl or ethyl;
RA1和RA2之一为H且RA1和RA2中的另一个为任选取代的(C1-C4烷基)氧基-,One of R A1 and R A2 is H and the other of R A1 and R A2 is optionally substituted (C 1 -C 4 alkyl)oxy-,
其中任选取代的(C1-C4烷基)氧基-的烷基任选地被1-2个羟基取代基取代;wherein the alkyl group of the optionally substituted (C 1 -C 4 alkyl)oxy-group is optionally substituted with 1-2 hydroxy substituents;
R3和R5二者都为-CO-NH2;和 R3 and R5 are both -CO- NH2 ; and
B为取代的-CH2-CH2-或取代的-CH=CH-,B is substituted -CH 2 -CH 2 - or substituted -CH=CH-,
其中所述取代的-CH2-CH2-或取代的-CH=CH-被1-4个各自独立地选自如下的取代基所取代:羟基和C1-2烷基;和wherein the substituted -CH 2 -CH 2 - or substituted -CH=CH- is substituted by 1 to 4 substituents independently selected from the following: hydroxyl and C 1-2 alkyl; and
RX或Ry中的至少一个独立地为C1-C4烷基且另一个为H,At least one of RX or Ry is independently C 1 -C 4 alkyl and the other is H,
或RX和Ry二者都独立地为C1-C4烷基。Or both RX and Ry are independently C1 - C4 alkyl.
本发明的代表性化合物包括实施例的化合物。应当理解的是,本发明涵盖作为游离碱和作为其盐(例如作为其药学上可接受的盐)的式(I)化合物。在一个实施方案中,本发明涉及游离碱形式的式(I)化合物。在另一个实施方案中,本发明涉及盐形式、特别是药学上可接受的盐的形式的式(I)化合物。应当进一步理解的是,在一个实施方案中,本发明涉及游离碱形式的实施例的化合物。在另一个实施方案中,本发明涉及盐形式、特别是药学上可接受的盐的形式的实施例的化合物。Representative compounds of the present invention include compounds of the embodiments. It should be understood that the present invention encompasses compounds of formula (I) as free bases and as salts thereof (e.g., as pharmaceutically acceptable salts thereof). In one embodiment, the present invention relates to compounds of formula (I) in the form of free bases. In another embodiment, the present invention relates to compounds of formula (I) in the form of salts, particularly pharmaceutically acceptable salts. It should be further understood that, in one embodiment, the present invention relates to compounds of the embodiments in the form of free bases. In another embodiment, the present invention relates to compounds of the embodiments in the form of salts, particularly pharmaceutically acceptable salts.
本发明化合物的特定实施方案包括:Specific embodiments of the compounds of the present invention include:
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-羟基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-hydroxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide,
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(2E,2'E)-1,1'-(戊烷-1,5-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(2E,2'E)-1,1'-(pentane-1,5-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide,
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide,
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-甲氧基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-methoxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-1-((E)-4-((E)-7-(苄氧基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((E)-4-((E)-7-(benzyloxy)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide,
(E)-1-((E)-4-((E)-4-溴-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((E)-4-((E)-4-bromo-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide,
(E)-1-((2R,3R)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((2R,3R)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-1-((2S,3S)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((2S,3S)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-1-(4-((E)-7-溴-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-(4-((E)-7-bromo-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-1-(((4R,5R)-5-(((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-(((4R,5R)-5-(((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide,
(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-(4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-(4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-羟基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-hydroxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-1-((2S,3S)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二乙氧基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((2S,3S)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-diethoxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
4-(((E)-6-氨基甲酰基-3-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-甲基-2,3-二氢-1H-苯并[d]咪唑-4-基)氧基)丁酸甲酯,4-(((E)-6-carbamoyl-3-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-methyl-2,3-dihydro-1H-benzo[d]imidazol-4-yl)oxy)butanoic acid methyl ester,
(E)-1-((E)-4-((E)-5-氨基甲酰基-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((E)-4-((E)-5-carbamoyl-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-7-(3-(二甲基氨基)丙氧基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-7-(3-(dimethylamino)propoxy)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide,
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((3-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((3-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酸甲酯,(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxylic acid methyl ester,
(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-7-羟基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-7-hydroxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-7-溴-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-7-bromo-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(2E,2'E)-1,1'-(2,3-二羟基丁烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺),(2E,2'E)-1,1'-(2,3-dihydroxybutane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide),
(2E,2'E)-1,1'-((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺),(2E,2'E)-1,1'-((2S,3S)-2,3-diethoxybutane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide),
(E)-1-((2R,3R)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二乙氧基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((2R,3R)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-diethoxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(2E,2'E)-1,1'-((2R,3R)-2,3-二乙氧基丁烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-羟基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺),(2E,2'E)-1,1'-((2R,3R)-2,3-diethoxybutane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-hydroxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide),
(E)-1-((2R,3R)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二乙氧基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((2R,3R)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-diethoxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide,
(E)-1-((2R,3R)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二乙氧基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((2R,3R)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-diethoxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-1-((2R,3R)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二乙氧基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((2R,3R)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-diethoxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-苯乙基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-phenethyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-7-溴-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-7-bromo-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-1-(5-((E)-5-氨基甲酰基-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-1-基)戊基)-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-(5-((E)-5-carbamoyl-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)pentyl)-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide,
(E)-1-(5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-1-基)戊基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-(5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)pentyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide,
(E)-1-(5-((E)-5-氨基甲酰基-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-1-基)戊基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-(5-((E)-5-carbamoyl-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)pentyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide,
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H苯并[d]咪唑-5-甲酸丁酯(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxylic acid butyl ester
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酸,(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxylic acid,
(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-4-(吗啉代甲基)-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-4-(morpholinomethyl)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((2-氟苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((2-fluorobenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-氟苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-fluorobenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((3-氟苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H苯并[d]咪唑-5-甲酰胺,(E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((3-fluorobenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-7-异丁氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-7-isobutoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-7-(3-(二甲基氨基)丙氧基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H苯并[d]咪唑-5-甲酰胺,(E)-7-(3-(dimethylamino)propoxy)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-1-((E)-4-((E)-7-(3-(二甲基氨基)丙氧基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H苯并[d]咪唑-5-甲酰胺,(E)-1-((E)-4-((E)-7-(3-(dimethylamino)propoxy)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-7-(2-羟基-2-甲基丙氧基)-3-甲基-2,3-二氢-1H苯并[d]咪唑-5-甲酰胺,(E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-7-(2-hydroxy-2-methylpropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(2E,2'E)-1,1'-((内消旋)-2,3-二甲氧基丁烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺),(2E,2'E)-1,1'-((meso)-2,3-dimethoxybutane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide),
(E)-1-((E)-4-((E)-7-(3-(4,4-二氟哌啶-1-基)丙氧基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,(E)-1-((E)-4-((E)-7-(3-(4,4-difluoropiperidin-1-yl)propoxy)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide,
(5aE,21E,29E)-8-乙基-26-(3-羟基丙氧基)-5,10,18,22,29,30-六甲基-7,20-二氧代-5,7,8,11,12,13,14,15,20,22,28,31-十二氢苯并[4,5]咪唑并[1,2-a]苯并[4,5]咪唑并[2,1-p]二吡唑并[5,1-e:4',3'-l][1,3,6,15,17]五氮杂环二十一碳十烯(pentaazacyclohenicosine)-3,24-二甲酰胺,(5aE,21E,29E)-8-ethyl-26-(3-hydroxypropoxy)-5,10,18,22,29,30-hexamethyl-7,20-dioxo-5,7,8,11,12,13,14,15,20,22,28,31-dodecahydrobenzo[4,5]imidazo[1,2-a]benzo[4,5]imidazo[2,1-p]dipyrazolo[5,1-e:4',3'-l][1,3,6,15,17]pentaazacyclohenicosine-3,24-dicarboxamide,
或其互变异构体;or a tautomer thereof;
或其盐,特别是其药学上可接受的盐。or a salt thereof, in particular a pharmaceutically acceptable salt thereof.
在一个实施方案中,上述式的化合物,例如式(I)、式(I-2)或式(I-3)的化合物为具有以下结构的(E)-1-((2R,3R)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺In one embodiment, the compound of the above formula, such as the compound of formula (I), formula (I-2) or formula (I-3) is (E)-1-((2R,3R)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide having the following structure
或其互变异构体;or a tautomer thereof;
或其盐,特别是其药学上可接受的盐。or a salt thereof, in particular a pharmaceutically acceptable salt thereof.
在一个实施方案中,上述式的化合物,例如式(I)、式(I-2)或式(I-3)的化合物为具有以下结构的(E)-1-((2S,3S)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺In one embodiment, the compound of the above formula, such as the compound of formula (I), formula (I-2) or formula (I-3) is (E)-1-((2S,3S)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide having the following structure
或其互变异构体;or a tautomer thereof;
或其盐,特别是其药学上可接受的盐。or a salt thereof, in particular a pharmaceutically acceptable salt thereof.
在一个实施方案中,上述式的化合物,例如式(I)、式(I-2)或式(I-3)的化合物不是以下化合物:具有以下结构的(E)-1-((2R,3R)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺In one embodiment, the compound of the above formula, such as the compound of formula (I), formula (I-2) or formula (I-3), is not the following compound: (E)-1-((2R,3R)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide having the following structure
或其互变异构体;or a tautomer thereof;
或其盐,特别是其药学上可接受的盐。or a salt thereof, in particular a pharmaceutically acceptable salt thereof.
在一个实施方案中,上述式的化合物,例如式(I)、式(I-2)或式(I-3)的化合物不是以下化合物:具有以下结构的(E)-1-((2S,3S)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺In one embodiment, the compound of the above formula, such as the compound of formula (I), formula (I-2) or formula (I-3), is not the following compound: (E)-1-((2S,3S)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide having the following structure
或其互变异构体;or a tautomer thereof;
或其盐,特别是其药学上可接受的盐。or a salt thereof, in particular a pharmaceutically acceptable salt thereof.
本发明化合物可以含有一个或多个不对称中心(也称为手性中心),诸如手性碳或手性-SO-部分。含有一个或多个手性中心的本发明化合物可以作为外消旋混合物、非对映异构体混合物、对映异构体富集的混合物、非对映异构体富集的混合物或作为对映异构体纯或非对映异构体纯的单独立体异构体存在。The compounds of the invention may contain one or more asymmetric centers (also referred to as chiral centers), such as chiral carbon or chiral -SO- moieties. The compounds of the invention containing one or more chiral centers may exist as racemic mixtures, diastereomeric mixtures, enantiomerically enriched mixtures, diastereoisomerically enriched mixtures, or as enantiomerically pure or diastereoisomerically pure individual stereoisomers.
存在于本发明化合物中的手性中心的立体化学通常以本文说明的化合物名称和/或化学结构表示。在存在于本发明化合物或本文说明的任何化学结构中的手性中心的立体化学未指定的情况下,该结构旨在涵盖任何立体异构体及其所有混合物。因此,本发明涵盖式(I)化合物的所有异构体及其盐,无论作为分离以便基本上不含其它异构体(即纯的)的单独异构体还是作为混合物(即外消旋体和外消旋混合物)。可以分离经分离以便基本上不含其它异构体(即纯的)的单独异构体,使得存在小于10%,特别是小于约1%,例如小于约0.1%的其它异构体。The stereochemistry of the chiral centers present in the compounds of the present invention is generally indicated in the compound names and/or chemical structures illustrated herein. Where the stereochemistry of the chiral centers present in the compounds of the present invention or in any chemical structure illustrated herein is not specified, the structure is intended to encompass any stereoisomers and all mixtures thereof. Thus, the present invention encompasses all isomers of compounds of formula (I) and their salts, whether as individual isomers separated so as to be substantially free of other isomers (i.e., pure) or as mixtures (i.e., racemates and racemic mixtures). Individual isomers separated so as to be substantially free of other isomers (i.e., pure) may be separated so that there is less than 10%, particularly less than about 1%, for example less than about 0.1% of other isomers.
可以使用本领域技术人员已知的方法拆分本发明化合物的单独立体异构体(或可以富集立体异构体的混合物)。例如,这样的拆分可以如下实施:(1) 通过形成非对映异构体盐、络合物或其它衍生物;(2)通过与立体异构体特异性试剂的选择性反应,例如通过酶促氧化或还原;或(3)通过在手性环境中的气-液色谱或液相色谱,所述手性环境例如在手性载体(诸如具有结合的手性配体的二氧化硅)或在手性溶剂存在的情况下。应理解,在将期望立体异构体通过上述分离方法之一转化成另一化学实体的情况下,需要另一步骤来释放期望形式。或者,特异性立体异构体可以通过使用光学活性试剂、底物、催化剂或溶剂的不对称合成法,或通过不对称转化将一种对映异构体转化成另一种对映异构体来合成。Individual stereoisomers of the compounds of the invention (or mixtures that may be enriched in stereoisomers) may be resolved using methods known to those skilled in the art. For example, such resolution may be performed as follows: (1) by forming diastereomeric salts, complexes, or other derivatives; (2) by selective reaction with stereoisomer-specific reagents, such as by enzymatic oxidation or reduction; or (3) by gas-liquid chromatography or liquid chromatography in a chiral environment, such as in the presence of a chiral support (such as silica with bound chiral ligands) or in a chiral solvent. It should be understood that in the case where the desired stereoisomer is converted into another chemical entity by one of the above separation methods, another step is required to release the desired form. Alternatively, specific stereoisomers may be synthesized by asymmetric synthesis using optically active reagents, substrates, catalysts, or solvents, or by converting one enantiomer into another enantiomer by asymmetric transformation.
本发明还包括本发明化合物的各种氘代形式。结合于碳原子的每个可用氢原子可以独立地被氘原子替代。本领域普通技术人员将知道如何合成本发明化合物的氘代形式。例如,α-氘代α-氨基酸可商购得到或可通过常规技术制备(参见例如: Elemes, Y.和 Ragnarsson, U. J. Chem. Soc., Perkin Trans. 1, 1996, 6, 537-40)。采用这样的化合物可以允许制备其中手性中心处的氢原子被氘原子替代的化合物。其它可商购得到的氘代起始材料可用于制备本发明化合物的氘代类似物(参见例如:可得自Aldrich ChemicalCo., Milwaukee, WI的甲基-d 3 -胺),或者它们可采用氘代试剂使用常规技术合成(例如,通过使用氘化锂铝或硼氘化钠还原或通过金属-卤素交换、随后用D2O或甲醇-d 3淬灭)。The present invention also includes various deuterated forms of the compounds of the present invention. Each available hydrogen atom bound to a carbon atom can be independently replaced by a deuterium atom. One of ordinary skill in the art will know how to synthesize deuterated forms of the compounds of the present invention. For example, α-deuterated α-amino acids are commercially available or can be prepared by conventional techniques ( see, for example: Elemes, Y. and Ragnarsson, UJ Chem. Soc., Perkin Trans. 1, 1996, 6, 537-40 ). Using such compounds can allow the preparation of compounds in which the hydrogen atom at the chiral center is replaced by a deuterium atom. Other commercially available deuterated starting materials can be used to prepare deuterated analogs of compounds of the invention (see, for example, methyl- d3 - amine available from Aldrich Chemical Co., Milwaukee, WI), or they can be synthesized using conventional techniques employing deuterated reagents (e.g., by reduction using lithium aluminum deuteride or sodium borodeuteride or by metal-halogen exchange followed by quenching with D2O or methanol - d3 ).
合适的式(I)化合物的药学上可接受的盐可以包括酸加成盐或碱加成盐。对于合适的药学上可接受的盐的综述,参见Berge等人, J. Pharm. Sci.,66:1-19, (1977)以及P. H. Stahl和C. G. Wermuth, 编辑, Handbook of Pharmaceutical Salts: Properties, Selection and Use, Weinheim/Zürich:Wiley-VCH/VHCA (2002)。Suitable pharmaceutically acceptable salts of compounds of formula (I) may include acid addition salts or base addition salts. For a review of suitable pharmaceutically acceptable salts, see Berge et al. , J. Pharm. Sci., 66: 1-19, (1977) and PH Stahl and CG Wermuth, eds., Handbook of Pharmaceutical Salts: Properties, Selection and Use , Weinheim/Zürich: Wiley-VCH/VHCA (2002).
含有碱性胺或其它碱性官能团的式(I)化合物的盐可以通过本领域已知的任何合适的方法(诸如用合适的无机或有机酸处理游离碱)制备。如此形成的药学上可接受的盐的实例包括乙酸盐、己二酸盐、抗坏血酸盐、天冬氨酸盐、苯磺酸盐、苯甲酸盐、樟脑酸盐、樟脑-磺酸盐(camphor-sulfonate)(樟脑磺酸盐(camsylate))、癸酸盐(caprate)(癸酸盐(decanoate))、己酸盐(caproate)(己酸盐(hexanoate))、辛酸盐(carprylate)(辛酸盐(octanoate))、碳酸盐、碳酸氢盐、肉桂酸盐、柠檬酸盐、环己基氨基磺酸盐、十二烷基硫酸盐(十二烷基硫酸盐(estolate))、乙烷 -1,2-二磺酸盐(乙二磺酸盐)、乙磺酸盐(乙磺酸盐(esylate))、甲酸盐、富马酸盐(半富马酸盐等)、半乳糖酸盐(粘酸盐)、龙胆酸盐(2,5-二羟基苯甲酸盐)、葡庚糖酸盐(葡庚糖酸盐(gluceptate))、葡萄糖酸盐、葡萄糖醛酸盐、谷氨酸盐、戊二酸盐、甘油磷酸盐、乙醇酸盐、马尿酸盐、氢溴酸盐、盐酸盐(二盐酸盐等)、氢碘酸盐、异丁酸盐、乳酸盐、乳糖醛酸盐、月桂酸盐、马来酸盐、苹果酸盐、丙二酸盐、扁桃酸盐、甲磺酸盐(甲磺酸盐(mesylate))、萘-1,5-二磺酸盐(萘二磺酸盐)、萘-磺酸盐(萘磺酸盐(napsylate))、烟酸盐、硝酸盐、油酸盐、草酸盐、棕榈酸盐、双羟萘酸盐、磷酸盐(二磷酸盐等)、丙酸盐、焦谷氨酸盐、水杨酸盐、癸二酸盐、硬脂酸盐、琥珀酸盐、硫酸盐、酒石酸盐、硫氰酸盐、对甲苯磺酸盐(甲苯磺酸盐(tosylate))、十一碳烯酸盐、1-羟基-2-萘甲酸盐、2,2-二氯乙酸盐、2-羟基乙磺酸盐(羟乙基磺酸盐)、2-氧代戊二酸盐、4-乙酰氨基苯甲酸盐和4-氨基水杨酸盐。Salts of compounds of formula (I) containing basic amines or other basic functional groups may be prepared by any suitable method known in the art, such as treating the free base with a suitable inorganic or organic acid. Examples of pharmaceutically acceptable salts so formed include acetates, adipates, ascorbates, aspartates, benzenesulfonates, benzoates, camphorates, camphor-sulfonates (camsylate), caprates (decanoate), caproates (hexanoate), carprylates (octanoate), carbonates, bicarbonates, cinnamates, citrates, cyclohexylsulfamates, dodecyl sulfates (estolate), ethane -1,2-disulfonate (edisylate), ethanesulfonate (esylate), formate, fumarate (hemifumarate, etc.), galactonate (mucate), gentisate (2,5-dihydroxybenzoate), glucoheptonate (gluceptate), gluconate, glucuronate, glutamate, glutarate, glycerophosphate, glycolate, hippurate, hydrobromide, hydrochloride (dihydrochloride, etc.), hydroiodide, isobutyrate, lactate, lactobionate, laurate, maleate, malate, malonate, mandelate, methanesulfonate (mesylat e)), naphthalene-1,5-disulfonate (naphthalenedisulfonate), naphthalene-sulfonate (napsylate), nicotinate, nitrate, oleate, oxalate, palmitate, pamoate, phosphate (diphosphate, etc.), propionate, pyroglutamate, salicylate, sebacate, stearate, succinate, sulfate, tartrate, thiocyanate, p-toluenesulfonate (tosylate), undecylenate, 1-hydroxy-2-naphthoate, 2,2-dichloroacetate, 2-hydroxyethanesulfonate (isethionate), 2-oxoglutarate, 4-acetamidobenzoate and 4-aminosalicylate.
含有羧酸或其它酸性官能团的公开的化合物的盐可以通过与合适的碱反应来制备。这样的药学上可接受的盐可以用提供药学上可接受的阳离子的碱来制备,其包括碱金属盐(特别是钠盐和钾盐)、碱土金属盐(特别是钙盐和镁盐)、铝盐和铵盐,以及由生理学可接受的有机碱制备的盐,所述有机碱诸如三甲胺、三乙胺、吗啉、吡啶、哌啶、甲基吡啶、二环己胺、N,N’-二苄基乙二胺、2-羟基乙胺、双-(2-羟基乙基)胺、三-(2-羟基乙基)胺、普鲁卡因、二苄基哌啶、去氢枞胺、N,N’-双去氢枞胺、葡糖胺、N-甲基葡糖胺、三甲吡啶、胆碱、奎宁、喹啉和碱性氨基酸,诸如赖氨酸和精氨酸。Salts of disclosed compounds containing carboxylic acids or other acidic functional groups can be prepared by reaction with a suitable base. Such pharmaceutically acceptable salts can be prepared with bases that provide pharmaceutically acceptable cations, including alkali metal salts (particularly sodium and potassium salts), alkaline earth metal salts (particularly calcium and magnesium salts), aluminum salts and ammonium salts, and salts prepared from physiologically acceptable organic bases, such as trimethylamine, triethylamine, morpholine, pyridine, piperidine, picoline, dicyclohexylamine, N , N' -dibenzylethylenediamine, 2-hydroxyethylamine, bis-(2-hydroxyethyl)amine, tris-(2-hydroxyethyl)amine, procaine, dibenzylpiperidine, dehydroabietylamine, N , N' -bisdehydroabietylamine, glucosamine, N -methylglucosamine, colidine, choline, quinine, quinoline and basic amino acids, such as lysine and arginine.
本发明在其范围内包括式(I)化合物的盐(例如,氢溴酸盐、二氢溴酸盐、富马酸盐、半富马酸盐等)的所有可能的化学计量和非化学计量形式。The present invention includes within its scope all possible stoichiometric and non-stoichiometric forms of the salts (eg, hydrobromide, dihydrobromide, fumarate, hemifumarate, etc.) of the compounds of formula (I).
当公开的化合物或其盐通过结构命名或描绘时,应理解该化合物或盐,包括其溶剂化物(特别是水合物),可以以结晶形式、非结晶形式或其混合物存在。该化合物或其盐或其溶剂化物(特别是水合物)也可以表现出多晶型现象(即以不同结晶形式存在的能力)。这些不同的结晶形式通常称为“多晶型物”。应理解,本发明包括任何本发明化合物的所有多晶型物,例如通过本文结构命名或描绘的任何化合物的所有多晶型形式,包括其任何盐和/或溶剂化物(特别是水合物)。When a disclosed compound or salt thereof is named or depicted by a structure, it is understood that the compound or salt, including solvates thereof (particularly hydrates), may exist in a crystalline form, a non-crystalline form, or a mixture thereof. The compound or its salt or solvate (particularly hydrate) may also exhibit polymorphism (i.e., the ability to exist in different crystalline forms). These different crystalline forms are generally referred to as "polymorphs". It is understood that the present invention includes all polymorphs of any compound of the present invention, such as all polymorphic forms of any compound named or depicted by a structure herein, including any salt and/or solvate (particularly hydrate) thereof.
多晶型物具有相同的化学组成,但堆积(packing)、几何排列和其它描述性的晶体固态的特性不同。因此,多晶型物可具有不同的物理特性,诸如形状、密度、硬度、可变形性、稳定性和溶解特性。多晶型物典型地表现出不同的熔点、IR光谱和X-射线粉末衍射图,其可用于鉴定。应理解,可产生不同多晶型物,例如,通过改变或调整在结晶/重结晶所述化合物中所用的条件。多晶型形式可以使用多种常规分析技术表征和区分,所述分析技术包括但不限于,X-射线粉末衍射(XRPD)图谱、红外(IR)光谱、拉曼光谱、差示扫描量热(DSC)、热重分析(TGA)和固态核磁共振(SSNMR)。Polymorphs have the same chemical composition, but the characteristics of packing, geometric arrangement and other descriptive crystalline solids are different. Therefore, polymorphs can have different physical properties, such as shape, density, hardness, deformability, stability and solubility characteristics. Polymorphs typically show different melting points, IR spectra and X-ray powder diffraction patterns, which can be used for identification. It should be understood that different polymorphs can be produced, for example, by changing or adjusting the conditions used in the compound described in crystallization/recrystallization. Polymorphic forms can be characterized and distinguished using a variety of conventional analytical techniques, including but not limited to, X-ray powder diffraction (XRPD) spectrum, infrared (IR) spectrum, Raman spectroscopy, differential scanning calorimetry (DSC), thermogravimetric analysis (TGA) and solid-state nuclear magnetic resonance (SSNMR).
技术人员应理解,当结晶期间将溶剂分子并入晶格中时,可以形成式(I)化合物的药学上可接受的溶剂化物(特别是水合物),包括式(I)化合物的药学上可接受的盐的药学上可接受的溶剂化物。溶剂化物可涉及非水溶剂诸如乙醇,或者它们可涉及水作为掺入晶格中的溶剂。其中水为掺入晶格中的溶剂的溶剂化物典型地称为“水合物”。The skilled person will appreciate that pharmaceutically acceptable solvates (particularly hydrates) of compounds of formula (I), including pharmaceutically acceptable solvates of pharmaceutically acceptable salts of compounds of formula (I), may be formed when solvent molecules are incorporated into the crystal lattice during crystallization. Solvates may involve non-aqueous solvents such as ethanol, or they may involve water as a solvent incorporated into the crystal lattice. Solvates in which water is a solvent incorporated into the crystal lattice are typically referred to as "hydrates".
本发明在其范围内包括所有可能的化学计量和非化学计量的盐和/或水合物形式。The present invention includes within its scope all possible stoichiometric and non-stoichiometric salt and/or hydrate forms.
适合于在医药中使用的本发明化合物的盐和溶剂化物(例如水合物和盐的水合物)是其中抗衡离子或相关溶剂是药学上可接受的那些。具有非药学上可接受的抗衡离子的盐也在本发明的范围内,例如,在制备本发明其它化合物的过程中用作中间体。Salts and solvates (e.g., hydrates and hydrates of salts) of the compounds of the invention suitable for use in medicine are those in which the counterion or associated solvent is pharmaceutically acceptable. Salts with non-pharmaceutically acceptable counterions are also within the scope of the invention, for example, used as intermediates in the process of preparing other compounds of the invention.
通常,药学上可接受的盐适当时可以通过使用期望的酸或碱容易地制备。所得盐可以从溶液中结晶或沉淀,或通过研磨形成,并且可以通过过滤或通过蒸发溶剂来回收。In general, pharmaceutically acceptable salts can be readily prepared by using the desired acid or base as appropriate. The resulting salt may be crystallized or precipitated from solution, or formed by trituration, and may be recovered by filtration or by evaporation of the solvent.
由于本发明化合物意图用于药物组合物中,将容易理解它们各自优选以基本上纯的形式,例如至少60%纯,更合适至少75%纯,以及优选至少85%,尤其至少98%纯(%基于重量对重量)提供。所述化合物的不纯制剂可用于制备药物组合物中使用的更纯形式。Since the compounds of the present invention are intended for use in pharmaceutical compositions, it will be readily appreciated that each of them is preferably provided in substantially pure form, e.g., at least 60% pure, more suitably at least 75% pure, and preferably at least 85%, especially at least 98% pure (% on a weight-to-weight basis). Impure preparations of the compounds may be used to prepare purer forms for use in pharmaceutical compositions.
本发明涵盖本发明化合物的所有前药,其在施用于接受者后能够(直接或间接)提供本发明化合物或其活性代谢物或残余物。这样的衍生物对于本领域技术人员来说是可识别的,无需过度的实验。尽管如此,参考Burger’s Medicinal Chemistry and DrugDiscovery, 第5版,第1卷:Principles and Practice(其在教导这样的衍生物的程度上通过引用并入本文)的教导。The present invention encompasses all prodrugs of the compounds of the invention which, upon administration to a recipient, are capable of providing (directly or indirectly) a compound of the invention or its active metabolite or residue. Such derivatives are identifiable to those skilled in the art without undue experimentation. Nevertheless, reference is made to the teachings of Burger's Medicinal Chemistry and Drug Discovery, 5th Edition, Vol. 1: Principles and Practice (which is incorporated herein by reference to the extent that such derivatives are taught).
应当进一步理解,本发明在其范围内包括本发明化合物的任何游离碱形式的所有互变异构形式或异构体形式以及所有可能的化学计量和非化学计量的盐形式。本发明化合物可用于治疗或预防其中调节STING有益的疾病和病症。这样的STING介导的疾病和病症包括炎症、过敏性和自身免疫性疾病、感染性疾病、癌症、癌前综合征、代谢疾病和心血管疾病。本发明化合物也可用作免疫原性组合物或疫苗佐剂。因此,本发明涉及调节STING的方法,其包括使细胞与本发明化合物接触。It should be further understood that the present invention includes within its scope all tautomeric or isomeric forms of any free base form of the compounds of the present invention and all possible stoichiometric and non-stoichiometric salt forms. The compounds of the present invention can be used to treat or prevent diseases and conditions in which modulation of STING is beneficial. Such STING-mediated diseases and conditions include inflammation, allergic and autoimmune diseases, infectious diseases, cancer, precancerous syndromes, metabolic diseases and cardiovascular diseases. The compounds of the present invention can also be used as immunogenic compositions or vaccine adjuvants. Therefore, the present invention relates to a method of modulating STING, which comprises contacting a cell with a compound of the present invention.
本发明的一个方面提供了治疗或预防其中激动STING有益的STING介导的疾病和病症的方法。示例性疾病/病症包括但不限于癌症、感染性疾病(例如,HIV、HBV、HCV、HPV和流感)、疫苗佐剂。One aspect of the invention provides methods for treating or preventing STING-mediated diseases and conditions in which agonizing STING is beneficial. Exemplary diseases/conditions include, but are not limited to, cancer, infectious diseases (e.g., HIV, HBV, HCV, HPV, and influenza), vaccine adjuvants.
本发明的一个方面提供治疗或预防STING介导的疾病或病症的方法,其中抑制STING是有益的。示例性的疾病/病症包括但不限于系统性红斑狼疮 (SLE)、皮肤狼疮、狼疮性肾炎、牛皮癣、糖尿病包括胰岛素依赖型糖尿病 (IDDM)、肥胖症相关性胰岛素抗性和非酒精性脂肪肝病 (NAFLD)、皮肌炎、系统性硬化症(硬皮病)和干燥综合征 (SS)、类风湿性关节炎、牛皮癣关节炎、婴儿期发病的STING相关血管炎 (SAVI)、Aicardi Goutieres综合征 (AGS)、冻疮样狼疮、混合性结缔组织病、与阿尔茨海默氏病相关的神经炎症 、肌萎缩侧索硬化症 (ALS)、帕金森综合征、亨廷顿氏舞蹈病和多发性硬化症以及与心肌梗死相关的心脏炎症。One aspect of the invention provides methods for treating or preventing a STING-mediated disease or condition in which inhibition of STING is beneficial. Exemplary diseases/conditions include, but are not limited to, systemic lupus erythematosus (SLE), cutaneous lupus, lupus nephritis, psoriasis, diabetes including insulin-dependent diabetes mellitus (IDDM), obesity-related insulin resistance and non-alcoholic fatty liver disease (NAFLD), dermatomyositis, systemic sclerosis (scleroderma) and Sjögren's syndrome (SS), rheumatoid arthritis, psoriatic arthritis, STING-associated vasculitis of infancy (SAVI), Aicardi Goutieres syndrome (AGS), pernio lupus, mixed connective tissue disease, neuroinflammation associated with Alzheimer's disease, amyotrophic lateral sclerosis (ALS), Parkinson's disease, Huntington's disease and multiple sclerosis, and cardiac inflammation associated with myocardial infarction.
在一个实施方案中,本发明提供了本发明化合物,其用于疗法中。本发明还提供了式(I)化合物或其药学上可接受的盐,其用于疗法中。本发明尤其提供了式(I)化合物或其药学上可接受的盐,其用于治疗STING介导的疾病或病症。In one embodiment, the present invention provides a compound of the present invention for use in therapy. The present invention also provides a compound of formula (I) or a pharmaceutically acceptable salt thereof for use in therapy. The present invention particularly provides a compound of formula (I) or a pharmaceutically acceptable salt thereof for use in treating a STING-mediated disease or condition.
本发明还提供了式(I)化合物或其药学上可接受的盐,其用作疫苗佐剂。因此,还提供了免疫原性组合物或疫苗佐剂,其包含式(I)化合物或其药学上可接受的盐。The present invention also provides a compound of formula (I) or a pharmaceutically acceptable salt thereof, which is used as a vaccine adjuvant. Therefore, an immunogenic composition or vaccine adjuvant is also provided, which comprises a compound of formula (I) or a pharmaceutically acceptable salt thereof.
在本发明的一个进一步实施方案中,提供了组合物,其包含式(I)化合物或其药学上可接受的盐和一种或多种免疫刺激剂。In a further embodiment of the present invention, there is provided a composition comprising a compound of formula (I) or a pharmaceutically acceptable salt thereof and one or more immunostimulants.
在另一个实施方案中,本发明提供了本发明化合物,其用于治疗STING介导的疾病或病症和/或用作免疫原性组合物或疫苗佐剂。在另一个实施方案中,本发明提供了式(I)化合物或其药学上可接受的盐,其用于改善由于STING介导的疾病或病症而遭受的器官损伤或损害。In another embodiment, the present invention provides a compound of the present invention for use in treating a STING-mediated disease or condition and/or as an immunogenic composition or vaccine adjuvant. In another embodiment, the present invention provides a compound of formula (I) or a pharmaceutically acceptable salt thereof for use in improving organ damage or injury suffered due to a STING-mediated disease or condition.
本发明进一步提供了本发明化合物在制备药物中的用途,所述药物用于治疗STING介导的疾病或病症。本发明进一步提供了式(I)化合物或其盐、特别是其药学上可接受的盐在制备药物中的用途,所述药物用于治疗STING介导的疾病或病症,例如本文所述的疾病和病症。The present invention further provides the use of the compounds of the present invention in the preparation of a medicament for treating a disease or condition mediated by STING. The present invention further provides the use of a compound of formula (I) or a salt thereof, in particular a pharmaceutically acceptable salt thereof, in the preparation of a medicament for treating a disease or condition mediated by STING, such as the diseases and conditions described herein.
本发明进一步提供了式(I)化合物或其盐、特别是其药学上可接受的盐在制备疫苗中的用途。进一步提供了式(I)化合物或其药学上可接受的盐用于制备包含抗原或抗原组合物的免疫原性组合物的用途,所述免疫原性组合物用于治疗或预防疾病。进一步提供了式(I)化合物或其药学上可接受的盐用于制备包含抗原或抗原组合物的疫苗组合物的用途,所述疫苗组合物用于治疗或预防疾病。The present invention further provides the use of a compound of formula (I) or a salt thereof, particularly a pharmaceutically acceptable salt thereof, in the preparation of a vaccine. Further provided is the use of a compound of formula (I) or a pharmaceutically acceptable salt thereof for the preparation of an immunogenic composition comprising an antigen or an antigenic composition, the immunogenic composition being used to treat or prevent a disease. Further provided is the use of a compound of formula (I) or a pharmaceutically acceptable salt thereof for the preparation of a vaccine composition comprising an antigen or an antigenic composition, the vaccine composition being used to treat or prevent a disease.
在另一个实施方案中,本发明涉及治疗STING介导的疾病或病症的方法,其包含将治疗有效量的本发明化合物施用于有需要的人。在另一个实施方案中,本发明涉及治疗STING介导的疾病或病症的方法,其包括将治疗有效量的式(I)化合物或其盐、特别是其药学上可接受的盐施用于有需要的人。In another embodiment, the present invention relates to a method for treating a STING-mediated disease or condition, comprising administering a therapeutically effective amount of a compound of the present invention to a person in need thereof. In another embodiment, the present invention relates to a method for treating a STING-mediated disease or condition, comprising administering a therapeutically effective amount of a compound of formula (I) or a salt thereof, particularly a pharmaceutically acceptable salt thereof, to a person in need thereof.
在另一个实施方案中,本发明涉及治疗或预防疾病的方法,其包含向患有或易患疾病的人对象施用免疫原性组合物,所述免疫原性组合物包含抗原或抗原组合物和式(I)化合物或其药学上可接受的盐。在另一个实施方案中,本发明涉及治疗或预防疾病的方法,其包括向患有或易患疾病的患者人对象施用疫苗组合物,所述疫苗组合物包含抗原或抗原组合物和式(I)化合物或其药学上可接受的盐。In another embodiment, the present invention relates to a method for treating or preventing a disease, comprising administering an immunogenic composition comprising an antigen or antigenic composition and a compound of formula (I) or a pharmaceutically acceptable salt thereof to a human subject suffering from or susceptible to the disease. In another embodiment, the present invention relates to a method for treating or preventing a disease, comprising administering a vaccine composition comprising an antigen or antigenic composition and a compound of formula (I) or a pharmaceutically acceptable salt thereof to a patient human subject suffering from or susceptible to the disease.
在一个实施方案中,本发明涉及式(I)化合物或其药学上可接受的盐,其用于治疗炎症。在一个进一步方面,提供了治疗炎症的方法,其包含向有需要的人施用治疗有效量的式(I)化合物或其药学上可接受的盐。在一个进一步方面,提供了式(I)化合物或其药学上可接受的盐,其用于制备用于治疗炎症的药物。In one embodiment, the present invention relates to a compound of formula (I) or a pharmaceutically acceptable salt thereof for use in treating inflammation. In a further aspect, a method for treating inflammation is provided, comprising administering a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof to a person in need thereof. In a further aspect, a compound of formula (I) or a pharmaceutically acceptable salt thereof is provided for use in preparing a medicament for treating inflammation.
在一个实施方案中,本发明涉及式(I)化合物或其药学上可接受的盐,其用于治疗过敏性疾病。在一个进一步方面,提供了治疗过敏性疾病的方法,其包括向有需要的人施用治疗有效量的式(I)化合物或其药学上可接受的盐。在一个进一步方面,提供了式(I)化合物或其药学上可接受的盐,其用于制备用于治疗过敏性疾病的药物。示例性的过敏性疾病包括过敏性鼻炎、花粉症、特应性皮炎、荨麻疹。In one embodiment, the present invention relates to a compound of formula (I) or a pharmaceutically acceptable salt thereof for use in treating allergic diseases. In a further aspect, a method for treating allergic diseases is provided, comprising administering a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof to a person in need thereof. In a further aspect, a compound of formula (I) or a pharmaceutically acceptable salt thereof is provided for use in preparing a medicament for treating allergic diseases. Exemplary allergic diseases include allergic rhinitis, hay fever, atopic dermatitis, urticaria.
在一个实施方案中,本发明涉及式(I)化合物或其药学上可接受的盐,其用于治疗自身免疫性疾病。在一个进一步方面,提供了治疗自身免疫性疾病的方法,其包括向有需要的人施用治疗有效量的式(I)化合物或其药学上可接受的盐。在一个进一步方面,提供了式(I)化合物或其药学上可接受的盐,其用于制备用于治疗自身免疫性疾病的药物。In one embodiment, the present invention relates to a compound of formula (I) or a pharmaceutically acceptable salt thereof for use in treating an autoimmune disease. In a further aspect, a method for treating an autoimmune disease is provided, comprising administering to a person in need thereof a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof. In a further aspect, a compound of formula (I) or a pharmaceutically acceptable salt thereof is provided for use in preparing a medicament for treating an autoimmune disease.
在一个实施方案中,本发明涉及式(I)化合物或其药学上可接受的盐,其用于治疗感染性疾病。在一个进一步方面,提供了治疗感染性疾病的方法,其包括向有需要的人施用治疗有效量的式(I)化合物或其药学上可接受的盐。在一个进一步方面,提供了式(I)化合物或其药学上可接受的盐,其用于制备用于治疗感染性疾病的药物。In one embodiment, the present invention relates to a compound of formula (I) or a pharmaceutically acceptable salt thereof for use in treating infectious diseases. In a further aspect, a method for treating infectious diseases is provided, comprising administering a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof to a person in need thereof. In a further aspect, a compound of formula (I) or a pharmaceutically acceptable salt thereof is provided for use in preparing a medicament for treating an infectious disease.
在一个实施方案中,本发明涉及通过向人施用治疗有效量的式(I)化合物或其药学上可接受的盐来治疗人中的HIV感染的方法。在一个实施方案中,本发明涉及通过向具有HIV感染或有具有HIV感染的风险的人施用治疗有效量的式(I)化合物或其药学上可接受的盐来治疗人中的HIV感染的方法。在另一个实施方案中,本发明涉及通过向人施用治疗有效量的式(I)化合物或其药学上可接受的盐来治疗具有AIDS感染的人中的AIDS感染的方法。In one embodiment, the invention relates to a method of treating HIV infection in a human by administering to the human a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof. In one embodiment, the invention relates to a method of treating HIV infection in a human by administering to a human having HIV infection or at risk of having HIV infection a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof. In another embodiment, the invention relates to a method of treating AIDS infection in a human having AIDS infection by administering to the human a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof.
在一个实施方案中,本发明涉及通过向人施用治疗有效量的式(I)化合物或其药学上可接受的盐来治疗人中的HBV感染的方法。在一个实施方案中,本发明涉及通过向人施用治疗有效量的式(I)化合物或其药学上可接受的盐来治疗具有HBV感染或有具有HBV感染的风险的人中的HBV感染的方法。In one embodiment, the invention relates to a method of treating HBV infection in a human by administering to the human a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof. In one embodiment, the invention relates to a method of treating HBV infection in a human having HBV infection or at risk of having HBV infection by administering to the human a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof.
在一个实施方案中,治疗HBV感染的方法包括施用第一治疗剂。在一个实施方案中,所述方法包括施用第一治疗剂,所述第一治疗剂为治疗有效量的式(I)化合物或其药学上可接受的盐,并施用一种或多种第二治疗剂。在一个实施方案中,共同施用第一治疗剂和一种或多种第二治疗剂。In one embodiment, the method for treating HBV infection includes administering a first therapeutic agent. In one embodiment, the method includes administering a first therapeutic agent, wherein the first therapeutic agent is a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof, and administering one or more second therapeutic agents. In one embodiment, the first therapeutic agent and one or more second therapeutic agents are co-administered.
在一个实施方案中,依次地或伴随地共同施用第一治疗剂和一种或多种第二治疗剂。在一个实施方案中,一种或多种第二治疗剂也是式(I)化合物。在一个实施方案中,一种或多种第二治疗剂不同于本文中所述的化合物或组合物。一种或多种第二治疗剂的实例包括但不限于抗炎剂、化疗剂或抗感染剂。在其它相关实施方案中,另外的治疗剂可以为HBV药剂、HCV药剂、化疗剂、抗生素、镇痛剂、非甾体抗炎(NSAID)药剂、抗真菌剂、抗寄生虫药、抗恶心药(anti-nausea agent)、止泻剂、免疫调节剂或免疫抑制剂。In one embodiment, the first therapeutic agent and one or more second therapeutic agents are co-administered sequentially or concomitantly. In one embodiment, one or more second therapeutic agents are also compounds of formula (I). In one embodiment, one or more second therapeutic agents are different from compounds or compositions described herein. Examples of one or more second therapeutic agents include, but are not limited to, anti-inflammatory agents, chemotherapeutics, or anti-infective agents. In other related embodiments, additional therapeutic agents may be HBV agents, HCV agents, chemotherapeutics, antibiotics, analgesics, non-steroidal anti-inflammatory (NSAID) agents, antifungal agents, antiparasitic agents, anti-nausea agents, antidiarrheal agents, immunomodulators, or immunosuppressants.
在一个实施方案中,一种或多种第二治疗剂为HBV药剂。在一个实施方案中,HBV药剂可以包括但不限于干扰素α-2b、干扰素5 α-2a和干扰素αcon-1 (聚乙二醇化的和未聚乙二醇化的)、病毒唑;HBV RNA 复制抑制剂;HBV抗原产生抑制剂;HBV 治疗性疫苗;HBV 预防性疫苗;拉米夫定 (3TC);恩替卡韦 (ETV);富马酸替诺福韦酯 (TDF);替比夫定 (LdT);阿德福韦;或HBV抗体疗法(单克隆的或多克隆的)。In one embodiment, the one or more second therapeutic agents are HBV agents. In one embodiment, HBV agents may include, but are not limited to, interferon α-2b, interferon 5 α-2a, and interferon αcon-1 (pegylated and unpegylated), ribavirin; HBV RNA replication inhibitors; HBV antigen production inhibitors; HBV therapeutic vaccines; HBV preventive vaccines; lamivudine (3TC); entecavir (ETV); tenofovir disoproxil fumarate (TDF); telbivudine (LdT); adefovir; or HBV antibody therapy (monoclonal or polyclonal).
在一个实施方案中,一种或多种第二治疗剂为HCV药剂。在一个实施方案中,HCV药剂可以包括但不限于干扰素α-2b、干扰素α-2a和干扰素αcon-1 (聚乙二醇化的和未聚乙二醇化的);病毒唑;HCV RNA 复制抑制剂 (例如ViroPharma的VP50406 系列);HCV反义药剂(antisense agent);HCV 治疗性疫苗;HCV蛋白酶抑制剂;HCV解旋酶抑制剂;或HCV 单克隆或多克隆的抗体疗法。In one embodiment, the one or more second therapeutic agents are HCV agents. In one embodiment, HCV agents may include, but are not limited to, interferon alpha-2b, interferon alpha-2a, and interferon alphacon-1 (pegylated and unpegylated); ribavirin; HCV RNA replication inhibitors (e.g., ViroPharma's VP50406 series); HCV antisense agents; HCV therapeutic vaccines; HCV protease inhibitors; HCV helicase inhibitors; or HCV monoclonal or polyclonal antibody therapies.
在一个实施方案中,一种或多种第二治疗剂为抗炎剂 (即降低炎症的治疗)。在一个实施方案中,降低炎症的治疗可以包括但不限于治疗性生活方式变化(therapeuticlifestyle change)、甾类化合物、NSAID或DMARD。所送甾类化合物可以是皮质类固醇。所述NSAID可以为阿司匹林、对乙酰氨基酚、布洛芬、萘普生、COX抑制剂、吲哚美辛等。所述DMARD可以为TNF抑制剂、嘌呤合成抑制剂、钙调磷酸酶抑制剂、嘧啶合成抑制剂、柳氮磺胺吡啶、甲氨蝶呤等。In one embodiment, one or more second therapeutic agents are anti-inflammatory agents (i.e., treatments that reduce inflammation). In one embodiment, treatments that reduce inflammation may include, but are not limited to, therapeutic lifestyle changes, steroids, NSAIDs, or DMARDs. The steroids delivered may be corticosteroids. The NSAIDs may be aspirin, acetaminophen, ibuprofen, naproxen, COX inhibitors, indomethacin, etc. The DMARDs may be TNF inhibitors, purine synthesis inhibitors, calcineurin inhibitors, pyrimidine synthesis inhibitors, sulfasalazine, methotrexate, etc.
在一个实施方案中,一种或多种第二治疗剂为化疗剂 (即癌症治疗剂)。化疗剂可包括但不限于柔红霉素(daunorubicin)、道诺霉素(daunomycin)、更生霉素、阿霉素、表柔比星、伊达比星、伊索比星(esorubicin)、博来霉素、马磷酰胺、异环磷酰胺、阿糖胞苷、双-氯乙基亚硝基脲(bis-chloroethylnitrosurea)、白消安、丝裂霉素C、放线菌素D、光神霉素、强的松、羟孕酮 、睾酮、他莫昔芬、达卡巴嗪、甲基苄肼、六甲基蜜胺、五甲基蜜胺、米托蒽醌 、安吖啶、苯丁酸氮芥(chlorambucil)、甲基环己基亚硝基脲(methylcyclohexylnitrosurea)、氮芥、美法仑、环磷酰胺、6-巯基嘌呤、6-硫代鸟嘌呤、阿糖胞苷(CA)、5-氮杂胞苷、羟基脲、脱氧助间型霉素(deoxycoformycin)、4-羟基过氧环磷酰胺(4-hydroxyperoxycyclophosphoramide)、5-氟尿嘧啶(5-FU)、5-氟脱氧尿苷(5-FUdR)、甲氨蝶呤(MTX)、秋水仙碱、紫杉醇、长春新碱、长春花碱、依托泊苷、三甲曲沙、替尼泊苷、顺铂、吉西他滨和已烯雌酚(DES)。In one embodiment, the one or more second therapeutic agents are chemotherapeutic agents (i.e., cancer therapeutic agents). Chemotherapeutic agents may include, but are not limited to, daunorubicin, daunomycin, dactinomycin, doxorubicin, epirubicin, idarubicin, esorubicin, bleomycin, mafosfamide, ifosfamide, cytarabine, bis-chloroethylnitrosurea, busulfan, mitomycin C, actinomycin D, mithramycin, prednisone, hydroxyprogesterone, testosterone, tamoxifen, dacarbazine, procarbazine, hexamethylmelamine, pentamethylmelamine, mitoxantrone , amsacrine, chlorambucil, methylcyclohexylnitrosurea, nitrogen mustard, melphalan, cyclophosphamide, 6-mercaptopurine, 6-thioguanine, cytarabine (CA), 5-azacytidine, hydroxyurea, deoxycoformycin, 4-hydroxyperoxycyclophosphoramide, 5-fluorouracil (5-FU), 5-fluorodeoxyuridine (5-FUdR), methotrexate (MTX), colchicine, paclitaxel, vincristine, vinblastine, etoposide, trimetrexate, teniposide, cisplatin, gemcitabine, and diethylstilbestrol (DES).
在一个实施方案中,一种或多种第二治疗剂为免疫调节剂,已知为天然免疫活性剂、检查点抑制剂、T- 细胞刺激剂或恢复针对HBV的适应性免疫应答的其它药剂。免疫调节剂包括但不限于拮抗CTLA-4的抗体或小分子,诸如伊匹单抗(YERVOY)、PD-1,诸如Opdivo/纳武单抗和Keytruda/派姆单抗)、PDL1诸如TECENTRIQ™ (阿特珠单抗)、LAG3、TIM3或IDO。免疫调节剂包括但不限于刺激ICOS、OX-40、TLRs、IL7R或IL12R的抗体或小分子。In one embodiment, one or more second therapeutic agents are immunomodulators, known as natural immune active agents, checkpoint inhibitors, T-cell stimulators or other agents that restore adaptive immune responses to HBV. Immunomodulators include, but are not limited to, antibodies or small molecules that antagonize CTLA-4, such as Ipilimumab (YERVOY), PD-1, such as Opdivo/nivolumab and Keytruda/pembrolizumab), PDL1 such as TECENTRIQ™ (atezumab), LAG3, TIM3 or IDO. Immunomodulators include, but are not limited to, antibodies or small molecules that stimulate ICOS, OX-40, TLRs, IL7R or IL12R.
在一个实施方案中,一种或多种第二治疗剂为抗感染剂。抗感染剂的实例包括但不限于抗生素、抗真菌药物和抗病毒药物。In one embodiment, the one or more second therapeutic agents are anti-infective agents. Examples of anti-infective agents include, but are not limited to, antibiotics, antifungal drugs, and antiviral drugs.
在一个实施方案中,本发明涉及通过向人施用治疗有效量的式(I)化合物或其药学上可接受的盐来治疗人中的HCV感染的方法。在一个实施方案中,本发明涉及通过向人施用治疗有效量的式(I)化合物或其药学上可接受的盐来治疗具有HCV感染或有具有HCV感染风险的人中的HCV感染的方法。In one embodiment, the invention relates to a method of treating HCV infection in a human by administering to the human a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof. In one embodiment, the invention relates to a method of treating HCV infection in a human having HCV infection or at risk of having HCV infection by administering to the human a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof.
在一个实施方案中,本发明涉及通过向人施用治疗有效量的式(I)化合物或其药学上可接受的盐来治疗人中的流感的方法。在一个实施方案中,本发明涉及通过向人施用治疗有效量的式(I)化合物或其药学上可接受的盐来治疗具有感染或有具有感染风险的人中的流感的方法。In one embodiment, the invention relates to a method of treating influenza in a human by administering to the human a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof. In one embodiment, the invention relates to a method of treating influenza in a human having an infection or at risk of having an infection by administering to the human a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof.
在一个实施方案中,本发明涉及通过向人施用治疗有效量的式(I)化合物或其药学上可接受的盐来治疗人中的人乳头瘤病毒(HPV)感染的方法。在一个实施方案中,本发明涉及通过向人施用治疗有效量的式(I)化合物或其药学上可接受的盐来治疗具有HPV感染或有具有HPV感染风险的人中的HPV感染的方法。In one embodiment, the invention relates to a method for treating human papillomavirus (HPV) infection in a human by administering to the human a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof. In one embodiment, the invention relates to a method for treating HPV infection in a human having HPV infection or at risk of having HPV infection by administering to the human a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof.
如本文所用,术语“癌症(cancer)”、“新生物(neoplasm)”和“肿瘤(tumor)”可互换使用,并且以单数或复数形式,指经历恶性转化的细胞,所述恶性转化使得它们对宿主生物体是病理性的。通过良好建立的技术、特别是组织学检查,可以容易地区分原发性癌细胞与非癌细胞。如本文所用,癌细胞的定义不仅包括原发性癌细胞,而且包括源自癌细胞祖先的任何细胞。这包括转移的癌细胞,以及源自癌细胞的体外培养物和细胞系。当提及通常显现为实体肿瘤的一种类型的癌症时,“临床上可检测的”肿瘤是基于肿瘤质量可检测的肿瘤;例如,通过体检时的程序诸如计算机断层扫描(CT)扫描、磁共振成像(MRI)、X-射线、超声或触诊,和/或由于可获得自患者的样品中的一种或多种癌症特异性抗原的表达而可检测的肿瘤。肿瘤可以是造血系统(或血液或血液学或血液相关)癌症,例如,源自血细胞或免疫细胞的癌症,其可被称为“液体肿瘤”。基于血液肿瘤的临床病况的具体实例包括白血病,诸如慢性髓细胞性白血病、急性髓细胞性白血病、慢性淋巴细胞性白血病和急性淋巴细胞性白血病;浆细胞恶性肿瘤诸如多发性骨髓瘤、MGUS和瓦尔登斯特伦氏巨球蛋白血症(Waldenstrom's macroglobulinemia);淋巴瘤诸如非霍奇金氏淋巴瘤、霍奇金氏淋巴瘤等。As used herein, the terms "cancer", "neoplasm" and "tumor" are used interchangeably and, in the singular or plural, refer to cells that undergo malignant transformation, which makes them pathological to the host organism. Primary cancer cells can be easily distinguished from non-cancerous cells by well-established techniques, particularly histological examination. As used herein, the definition of cancer cells includes not only primary cancer cells, but also any cells derived from cancer cell ancestors. This includes metastatic cancer cells, as well as in vitro cultures and cell lines derived from cancer cells. When referring to a type of cancer that usually appears as a solid tumor, a "clinically detectable" tumor is a tumor that is detectable based on tumor mass; for example, by procedures such as computed tomography (CT) scans, magnetic resonance imaging (MRI), X-rays, ultrasound or palpation during a physical examination, and/or a tumor that is detectable due to the expression of one or more cancer-specific antigens in a sample that can be obtained from a patient. The tumor can be a hematopoietic (or blood or hematological or blood-related) cancer, for example, a cancer derived from blood cells or immune cells, which can be referred to as a "liquid tumor". Specific examples of hematological neoplasm-based clinical conditions include leukemias such as chronic myeloid leukemia, acute myeloid leukemia, chronic lymphocytic leukemia, and acute lymphocytic leukemia; plasma cell malignancies such as multiple myeloma, MGUS, and Waldenstrom's macroglobulinemia; lymphomas such as non-Hodgkin's lymphoma, Hodgkin's lymphoma, and the like.
癌症可以是其中存在异常数量的母细胞或不需要的细胞增殖或被诊断为血液癌症(包括淋巴样和髓样恶性肿瘤)的任何癌症。髓样恶性肿瘤包括,但不限于,急性髓样(或髓细胞或髓性或成髓细胞)白血病(未分化或分化的)、急性早幼粒细胞(或早幼粒细胞(promyelocytic)或早幼粒细胞(promyelogenous)或早幼粒细胞(promyeloblastic))白血病、急性髓单核细胞(或髓单核细胞性(myelomonoblastic))白血病、急性单核细胞(或单核细胞性(monoblastic))白血病、红白血病和巨核细胞(或成巨核细胞)白血病。这些白血病可以一起称为急性髓样(或髓细胞性(myelocytic)或骨髓性(myelogenous))白血病(AML)。髓样恶性肿瘤还包括骨髓增生性病症(MPD),其包括,但不限于,慢性髓性(或髓样)白血病(CML)、慢性髓单核细胞性白血病(CMML)、自发性血小板增多症(或血小板增多症)和真性红细胞增多症(PCV)。髓样恶性肿瘤还包括骨髓增生异常(或骨髓增生异常综合征或MDS),其可称为难治性贫血(RA),伴有母细胞过多的难治性贫血(RAEB),以及伴有转化中的母细胞过多的难治性贫血(RAEBT);以及伴有或不伴有原因不明性髓样化生的骨髓纤维化(MFS)。Cancer can be any cancer in which there is an abnormal number of blasts or unwanted cell proliferation or diagnosed as a blood cancer, including lymphoid and myeloid malignancies. Myeloid malignancies include, but are not limited to, acute myeloid (or myeloid cell or myeloid or myeloblastic) leukemia (undifferentiated or differentiated), acute promyelocytic (or promyelocytic or promyelogenous or promyeloblastic) leukemia, acute myelomonocytic (or myelomonocytic (myelomonoblastic)) leukemia, acute monocytic (or monoblastic (monoblastic)) leukemia, erythroleukemia, and megakaryoblastic (or megakaryoblastic) leukemia. These leukemias can be collectively referred to as acute myeloid (or myelocytic (myelocytic) or myeloid (myelogenous)) leukemia (AML). Myeloid malignancies also include myeloproliferative disorders (MPDs), including, but not limited to, chronic myeloid (or myeloid) leukemia (CML), chronic myelomonocytic leukemia (CMML), idiopathic thrombocythemia (or thrombocythemia), and polycythemia vera (PCV). Myeloid malignancies also include myelodysplasia (or myelodysplastic syndrome or MDS), which may be referred to as refractory anemia (RA), refractory anemia with excess blasts (RAEB), and refractory anemia with excess blasts in transformation (RAEBT); and myelofibrosis (MFS) with or without unexplained myeloid metaplasia.
造血系统癌症还包括淋巴样恶性肿瘤,其可以影响淋巴结、脾脏、骨髓、外周血和/或结外部位。淋巴样癌症包括B细胞恶性肿瘤,其包括,但不限于,B细胞非霍奇金氏淋巴瘤(B-NHL)。B-NHL可以是惰性(或低级)、中级(或侵袭性)或高级(非常侵袭性)的。惰性B细胞淋巴瘤包括滤泡性淋巴瘤(FL);小淋巴细胞性淋巴瘤(SLL);边缘区淋巴瘤(MZL),包括淋巴结MZL、结外MZL、脾MZL和具有绒毛淋巴细胞的脾MZL;淋巴浆细胞性淋巴瘤(LPL);和粘膜相关淋巴样组织(MALT或结外边缘区)淋巴瘤。中级B-NHL包括伴有或不伴有白血病参与的套细胞淋巴瘤(MCL)、弥漫性大细胞淋巴瘤(DLBCL)、滤泡性大细胞(或3级或3B级)淋巴瘤和原发性纵隔淋巴瘤(PML)。高级B-NHL包括伯基特淋巴瘤(BL)、伯基特样淋巴瘤(Burkitt-likelymphoma)、小无裂细胞淋巴瘤(SNCCL)和淋巴母细胞淋巴瘤。其它B-NHL包括免疫母细胞淋巴瘤(或免疫细胞瘤)、原发性渗出性淋巴瘤、HIV相关(或AIDS相关)淋巴瘤以及移植后淋巴增生性病症(PTLD)或淋巴瘤。B-细胞恶性肿瘤还包括,但不限于,慢性淋巴细胞性白血病(CLL)、幼淋巴细胞性白血病(PLL)、瓦尔登斯特伦氏巨球蛋白血症(WM)、毛细胞白血病(HCL)、大颗粒淋巴细胞(LGL)白血病、急性淋巴样(或淋巴细胞或淋巴母细胞)白血病和Castleman病。NHL还可以包括T细胞非霍奇金氏淋巴瘤(T-NHL),其包括但不限于非特指型T细胞非霍奇金氏淋巴瘤(NOS)、外周T细胞淋巴瘤(PTCL)、间变性大细胞淋巴瘤(ALCL)、血管免疫母细胞性淋巴样病症(AILD)、鼻自然杀伤(NK)细胞/T细胞淋巴瘤、γ/δ淋巴瘤、皮肤T细胞淋巴瘤、蕈样真菌病和塞扎里综合征(Sezary syndrome)。Hematopoietic cancers also include lymphoid malignancies, which can affect the lymph nodes, spleen, bone marrow, peripheral blood, and/or extranodal sites. Lymphoid cancers include B-cell malignancies, which include, but are not limited to, B-cell non-Hodgkin's lymphoma (B-NHL). B-NHL can be indolent (or low grade), intermediate (or aggressive), or high grade (very aggressive). Indolent B-cell lymphomas include follicular lymphoma (FL); small lymphocytic lymphoma (SLL); marginal zone lymphoma (MZL), including lymph node MZL, extranodal MZL, splenic MZL, and splenic MZL with villous lymphocytes; lymphoplasmacytic lymphoma (LPL); and mucosa-associated lymphoid tissue (MALT or extranodal marginal zone) lymphoma. Intermediate-grade B-NHL includes mantle cell lymphoma (MCL) with or without leukemic involvement, diffuse large cell lymphoma (DLBCL), follicular large cell (or grade 3 or 3B) lymphoma, and primary mediastinal lymphoma (PML). High-grade B-NHL includes Burkitt's lymphoma (BL), Burkitt-like lymphoma, small non-cleaved cell lymphoma (SNCCL), and lymphoblastic lymphoma. Other B-NHLs include immunoblastic lymphoma (or immunocytoma), primary effusion lymphoma, HIV-related (or AIDS-related) lymphoma, and post-transplant lymphoproliferative disorder (PTLD) or lymphoma. B-cell malignancies also include, but are not limited to, chronic lymphocytic leukemia (CLL), prolymphocytic leukemia (PLL), Waldenstrom's macroglobulinemia (WM), hairy cell leukemia (HCL), large granular lymphocytic (LGL) leukemia, acute lymphoid (or lymphocytic or lymphoblastic) leukemia, and Castleman's disease. NHL can also include T-cell non-Hodgkin's lymphoma (T-NHL), which includes but is not limited to non-Hodgkin's lymphoma not otherwise specified (NOS), peripheral T-cell lymphoma (PTCL), anaplastic large cell lymphoma (ALCL), angioimmunoblastic lymphoid disorder (AILD), nasal natural killer (NK) cell/T-cell lymphoma, gamma/delta lymphoma, cutaneous T-cell lymphoma, mycosis fungoides, and Sezary syndrome.
造血系统癌症(Hematopoietic cancer)还包括霍奇金氏淋巴瘤(或疾病),包括经典霍奇金氏淋巴瘤、结节性硬化性霍奇金氏淋巴瘤、混合细胞型霍奇金氏淋巴瘤、淋巴细胞优势性(LP)霍奇金氏淋巴瘤、结节性LP霍奇金氏淋巴瘤和淋巴细胞耗竭的霍奇金氏淋巴瘤。造血系统癌症还包括浆细胞疾病或癌症,诸如多发性骨髓瘤(MM),包括冒烟型(smoldering)MM、意义未明(或未知或不清楚)的单克隆丙种球蛋白病(MGUS)、浆细胞瘤(骨、髓外)、淋巴浆细胞性淋巴瘤(LPL)、瓦尔登斯特伦氏巨球蛋白血症、浆细胞白血病和原发性淀粉样变性(AL)。造血系统癌症还可以包括另外的造血细胞的其它癌症,包括多形核白细胞(或中性粒细胞)、嗜碱性粒细胞、嗜酸性粒细胞、树突状细胞、血小板、红细胞和自然杀伤细胞。在本文中称为“造血细胞组织”的包括造血细胞的组织包括骨髓;外周血;胸腺;和外周淋巴组织,诸如脾脏、淋巴结、与粘膜相关的淋巴组织(诸如肠相关淋巴组织)、扁桃体、淋巴集结(Peyer's patches)和阑尾,以及与其它粘膜(例如支气管衬里)相关的淋巴组织,。Hematopoietic cancer also includes Hodgkin's lymphoma (or disease), including classical Hodgkin's lymphoma, nodular sclerosing Hodgkin's lymphoma, mixed cell type Hodgkin's lymphoma, lymphocyte dominant (LP) Hodgkin's lymphoma, nodular LP Hodgkin's lymphoma and lymphocyte depleted Hodgkin's lymphoma. Hematopoietic cancer also includes plasma cell diseases or cancers, such as multiple myeloma (MM), including smoldering MM, monoclonal gammopathy of undetermined significance (or unknown or unclear) (MGUS), plasmacytoma (bone, extramedullary), lymphoplasmacytic lymphoma (LPL), Waldenstrom's macroglobulinemia, plasma cell leukemia and primary amyloidosis (AL). Hematopoietic cancers may also include other cancers of additional hematopoietic cells, including polymorphonuclear leukocytes (or neutrophils), basophils, eosinophils, dendritic cells, platelets, erythrocytes, and natural killer cells. Tissues including hematopoietic cells referred to herein as "hematopoietic tissues" include bone marrow; peripheral blood; thymus; and peripheral lymphoid tissues, such as spleen, lymph nodes, lymphoid tissue associated with mucosa (such as gut-associated lymphoid tissue), tonsils, Peyer's patches, and appendix, as well as lymphoid tissue associated with other mucosa (e.g., bronchial lining).
在一个实施方案中,本发明涉及式(I)化合物或其药学上可接受的盐,其用于治疗癌症和癌前综合征。在一个进一步方面,提供了治疗癌症和癌前综合征的方法,其包括向有需要的人施用治疗有效量的式(I)化合物或其药学上可接受的盐。在一个进一步方面,提供了式(I)化合物或其药学上可接受的盐,其用于制备用于治疗癌症和癌前综合征的药物。In one embodiment, the present invention relates to a compound of formula (I) or a pharmaceutically acceptable salt thereof for use in treating cancer and precancerous syndromes. In a further aspect, a method for treating cancer and precancerous syndromes is provided, comprising administering to a person in need thereof a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof. In a further aspect, a compound of formula (I) or a pharmaceutically acceptable salt thereof is provided for use in preparing a medicament for treating cancer and precancerous syndromes.
相关的自身免疫性疾病包括但不限于婴儿期发病的STING相关血管炎(SAVI)、Aicardi Goutieres综合征(AGS)、冻疮样狼疮、共济失调性毛细血管扩张(也称为Louis-Bar综合征)、伴有脑的脑白质营养不良的视网膜血管病变(RCVL)、系统性红斑狼疮(SLE)、皮肤狼疮、狼疮性肾炎、牛皮癣、糖尿病(包括胰岛素依赖型糖尿病(IDDM))、皮肌炎、人免疫缺陷病毒(HIV)、AIDS、多发性肌炎、系统性硬化症(硬皮病)和干燥综合征(SS)、类风湿性关节炎、牛皮癣关节炎、多关节炎、骨关节炎、重症肌无力、结节性多动脉炎、血管炎、皮肤血管炎、抗中性粒细胞胞浆抗体(ANCA)相关性血管炎、过敏性紫癜、自身免疫性肝炎、原发性硬化性胆管炎、韦格纳氏肉芽肿病、显微镜下多血管炎、白塞氏病、脊椎炎、巨细胞动脉炎、风湿性多肌痛、雷诺氏现象、原发性胆汁性肝硬化、中枢神经系统的原发性血管炎、显微镜下多血管炎、视神经脊髓炎和混合型结缔组织病。Related autoimmune diseases include, but are not limited to, STING-associated vasculitis of infancy (SAVI), Aicardi Goutieres syndrome (AGS), pernio lupus, ataxia telangiectasia (also known as Louis-Bar syndrome), retinal vasculopathy with cerebral leukodystrophy (RCVL), systemic lupus erythematosus (SLE), cutaneous lupus, lupus nephritis, psoriasis, diabetes (including insulin-dependent diabetes mellitus (IDDM)), dermatomyositis, human immunodeficiency virus (HIV), AIDS, polymyositis, systemic sclerosis (scleroderma) and Sjögren's syndrome (SS), rheumatoid arthritis Arthritis, psoriatic arthritis, polyarthritis, osteoarthritis, myasthenia gravis, polyarteritis nodosa, vasculitis, cutaneous vasculitis, antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis, Henoch-Schönlein purpura, autoimmune hepatitis, primary sclerosing cholangitis, Wegener's granulomatosis, microscopic polyangiitis, Behcet's disease, spondylitis, giant cell arteritis, polymyalgia rheumatica, Raynaud's phenomenon, primary biliary cirrhosis, primary vasculitis of the central nervous system, microscopic polyangiitis, neuromyelitis optica, and mixed connective tissue disease.
炎症代表一组对创伤的血管、细胞和神经应答。炎症可以表征为炎症细胞诸如单核细胞、 嗜中性粒细胞和粒细胞进入组织的运动。这通常与减少的内皮屏障功能和水肿进入组织有关。炎症可以分为急性或慢性。急性炎症是身体对有害刺激的初始应答,并且通过血浆和白细胞从血液到损伤组织的运动增加来实现。生化事件的级联传播炎性应答并使其成熟,其涉及受损伤组织内的局部血管系统、免疫系统和各种细胞。称为慢性炎症的持久炎症导致存在于炎症部位的细胞类型的进行性改变,并且其特征在于来自炎性过程的组织的同时破坏和愈合。Inflammation represents a group of vascular, cellular and neural responses to trauma. Inflammation can be characterized by the movement of inflammatory cells such as monocytes, neutrophils and granulocytes into tissues. This is usually associated with reduced endothelial barrier function and edema into tissues. Inflammation can be divided into acute or chronic. Acute inflammation is the body's initial response to harmful stimuli and is achieved by increased movement of plasma and leukocytes from the blood to damaged tissues. A cascade of biochemical events propagates the inflammatory response and matures it, which involves the local vascular system, immune system and various cells in the damaged tissue. Persistent inflammation, known as chronic inflammation, causes progressive changes in the cell types present at the site of inflammation and is characterized by simultaneous destruction and healing of tissues from the inflammatory process.
当作为对感染的免疫应答的一部分或作为对创伤的急性应答发生时,炎症可能是有益的并且通常是自限制性的。然而,炎症在多种条件下可能是有害的。这包括应答传染因子产生过度炎症,这可导致显著的器官损伤和死亡(例如,在败血症的环境中)。此外,慢性炎症通常是有害的,并且是许多慢性疾病的根源,其对组织造成严重和不可逆的损伤。在这样的情况下,免疫应答通常针对自身组织(自身免疫),尽管对外来实体的慢性应答也可能导致对自身组织的旁观者损伤(bystander damage)。Inflammation can be beneficial and is usually self-limiting when it occurs as part of an immune response to infection or as an acute response to trauma. However, inflammation can be harmful under a variety of conditions. This includes excessive inflammation in response to infectious agents, which can lead to significant organ damage and death (e.g., in the setting of sepsis). In addition, chronic inflammation is usually harmful and is the root cause of many chronic diseases, causing severe and irreversible damage to tissues. In such cases, the immune response is usually directed against one's own tissues (autoimmunity), although chronic responses to foreign entities may also result in bystander damage to one's own tissues.
因此,抗炎治疗的目的是减少这种炎症,以当存在时抑制自身免疫,并允许生理过程或愈合和组织修复进展。Therefore, the goal of anti-inflammatory therapy is to reduce this inflammation to suppress autoimmunity when present and allow physiological processes or healing and tissue repair to proceed.
本发明化合物可用于治疗身体的任何组织和器官的炎症,包括肌肉骨骼炎症、血管炎症、神经炎症、消化系统炎症、眼部炎症、心脏炎症、脂肪组织炎症、生殖系统炎症和其它炎症,如下所示例的。The compounds of the invention can be used to treat inflammation of any tissue or organ of the body, including musculoskeletal inflammation, vascular inflammation, neurological inflammation, digestive system inflammation, ocular inflammation, cardiac inflammation, adipose tissue inflammation, reproductive system inflammation, and other inflammations, as exemplified below.
肌肉骨骼炎症指肌肉骨骼系统的任何炎性病症,特别是影响骨骼关节,包括手、腕、肘、肩、颚、脊柱、颈、髋部、膝盖、踝和脚的关节的那些病症,以及影响连接肌肉到骨骼的组织诸如腱的病症。可以用本发明化合物治疗的肌肉骨骼炎症的实例包括关节炎(包括,例如,骨关节炎、类风湿性关节炎、牛皮癣性关节炎、强直性脊柱炎、急性和慢性感染性关节炎、与痛风和假痛风有关的关节炎和幼年特发性关节炎)、腱炎、滑膜炎、腱鞘炎、滑囊炎、纤维组织炎(纤维肌痛)、上髁炎、肌炎和骨炎(包括,例如,佩吉特氏病、耻骨炎和囊性纤维性骨炎)。Musculoskeletal inflammation refers to any inflammatory condition of the musculoskeletal system, particularly those affecting skeletal joints, including those of the hands, wrists, elbows, shoulders, jaws, spine, neck, hips, knees, ankles and feet, and conditions affecting tissues connecting muscle to bone, such as tendons. Examples of musculoskeletal inflammation that can be treated with the compounds of the invention include arthritis (including, for example, osteoarthritis, rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, acute and chronic infectious arthritis, arthritis associated with gout and pseudogout, and juvenile idiopathic arthritis), tendinitis, synovitis, tenosynovitis, bursitis, fibrositis (fibromyalgia), epicondylitis, myositis, and osteitis (including, for example, Paget's disease, osteitis pubis, and osteitis fibrosa cystica).
眼部炎症指眼睛的任何结构、包括眼睑的炎症。可以用本发明化合物治疗的眼部炎症的实例包括睑炎、睑皮松垂症、结膜炎、泪腺炎、角膜炎、干燥性角结膜炎(干眼病)、巩膜炎、倒睫症和葡萄膜炎。Ocular inflammation refers to inflammation of any structure of the eye, including the eyelids. Examples of ocular inflammation that can be treated with the compounds of the invention include blepharitis, blepharoptosis, conjunctivitis, dacryoadenitis, keratitis, keratoconjunctivitis sicca (dry eyes), scleritis, trichiasis, and uveitis.
可以用本发明化合物治疗的神经系统的炎症的实例包括脑炎、吉兰-巴雷综合征、脑膜炎、神经性肌强直、发作性睡病、多发性硬化症、脊髓炎、CNS血管炎和精神分裂症。Examples of inflammatory diseases of the nervous system that may be treated with the compounds of the invention include encephalitis, Guillain-Barré syndrome, meningitis, neuromyotonia, narcolepsy, multiple sclerosis, myelitis, CNS vasculitis, and schizophrenia.
可以采用本发明化合物治疗的与神经退行性疾病相关联的炎症的实例包括阿尔茨海默氏病和相关的痴呆、肌萎缩侧索硬化症(ALS)和额颞叶变性(FTD)、帕金森氏病和亨廷顿氏舞蹈病。Examples of inflammation associated with neurodegenerative diseases that may be treated with the compounds of the invention include Alzheimer's disease and related dementias, amyotrophic lateral sclerosis (ALS) and frontotemporal lobar degeneration (FTD), Parkinson's disease and Huntington's disease.
可以用本发明化合物治疗的脉管系统或淋巴系统的炎症的实例包括关节硬化、关节炎、静脉炎、血管炎和淋巴管炎。Examples of inflammation of the vasculature or lymphatic system that may be treated with the compounds of the invention include arthrosclerosis, arthritis, phlebitis, vasculitis and lymphangitis.
可以采用本发明化合物治疗的心血管系统的炎症的实例包括但不限于心肌梗死、心力衰竭、先天性心脏缺损、冠状动脉疾病、高血压、心肌病和其它相关的心血管病况。Examples of inflammation of the cardiovascular system that may be treated with the compounds of the invention include, but are not limited to, myocardial infarction, heart failure, congenital heart defects, coronary artery disease, hypertension, cardiomyopathy, and other related cardiovascular conditions.
可以采用本发明化合物治疗的肝脏炎症的实例包括但不限于肝纤维化、酒精性肝病(ALD)、非酒精性脂肪肝病 (NAFLD)和非酒精性脂肪肝炎、和胆汁性肝病(biliary liverdisease)。Examples of liver inflammation that can be treated with the compounds of the invention include, but are not limited to, liver fibrosis, alcoholic liver disease (ALD), non-alcoholic fatty liver disease (NAFLD) and non-alcoholic steatohepatitis, and biliary liver disease.
可以采用本发明化合物治疗的脂肪组织炎症的实例包括但不限于肥胖症和肥胖症诱发的胰岛素抗性。Examples of adipose tissue inflammation that may be treated using the compounds of the invention include, but are not limited to, obesity and obesity-induced insulin resistance.
可以采用本发明化合物治疗的肝脏炎症的实例包括但不限于肝纤维化和纤维瘤酒精性肝病(ALD)、非酒精性脂肪肝病 (NAFLD)和非酒精性脂肪肝炎 (NASH)、和胆汁性肝病。Examples of liver inflammation that may be treated with the compounds of the invention include, but are not limited to, liver fibrosis and fibromas, alcoholic liver disease (ALD), nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH), and biliary liver disease.
可以采用本发明化合物治疗的胰腺炎症的实例包括但不限于胰腺炎和代谢综合征诱发的胰腺β细胞功能障碍。Examples of pancreatic inflammation that may be treated using the compounds of the invention include, but are not limited to, pancreatitis and metabolic syndrome-induced pancreatic beta cell dysfunction.
可以采用本发明化合物治疗的肾脏炎症的实例包括但不限于肾脏肾炎。Examples of renal inflammation that may be treated using the compounds of the invention include, but are not limited to, renal nephritis.
可以采用本发明化合物治疗的肺的炎症的实例包括肺纤维化、COPD和哮喘。Examples of inflammatory diseases of the lung that may be treated using the compounds of the invention include pulmonary fibrosis, COPD and asthma.
可以采用本发明化合物治疗的眼的炎症的实例包括干眼综合征和年龄相关性黄斑变性。Examples of inflammatory conditions of the eye that may be treated using the compounds of the invention include dry eye syndrome and age-related macular degeneration.
可以用本发明化合物治疗的消化系统的炎性病症的实例包括胆管炎、胆囊炎、肠炎、小肠结肠炎、胃炎、胃肠炎、炎性肠病(诸如克罗恩氏病和溃疡性结肠炎)、回肠炎和直肠炎。Examples of inflammatory disorders of the digestive system that may be treated with the compounds of the invention include cholangitis, cholecystitis, enteritis, enterocolitis, gastritis, gastroenteritis, inflammatory bowel disease (such as Crohn's disease and ulcerative colitis), ileitis, and proctitis.
可用本发明化合物治疗的生殖系统的炎性病症的实例包括宫颈炎、绒毛膜羊膜炎、子宫内膜炎、附睾炎、脐炎、卵巢炎、睾丸炎、输卵管炎、输卵管卵巢脓肿、尿道炎、阴道炎、外阴炎和外阴痛。Examples of inflammatory disorders of the reproductive system that may be treated with the compounds of the invention include cervicitis, chorioamnionitis, endometritis, epididymitis, omphalitis, oophoritis, orchitis, salpingitis, tubo-ovarian abscess, urethritis, vaginitis, vulvitis, and vulvodynia.
在一个实施方案中,本发明化合物可以用于治疗系统性红斑狼疮、类风湿性关节炎、骨关节炎、炎症性肠病(IBD) (例如克罗恩氏病、溃疡性结肠炎)。In one embodiment, the compounds of the invention may be used to treat systemic lupus erythematosus, rheumatoid arthritis, osteoarthritis, inflammatory bowel disease (IBD) (eg, Crohn's disease, ulcerative colitis).
本发明化合物可用于治疗具有炎性组分的自身免疫病症。这样的病症包括性急性播散性普遍脱发(Acute disseminated alopecia universalise)、白塞氏病、查加斯病、婴儿期发病的STING相关血管炎(SAVI)、Aicardi Goutieres综合征(AGS)、冻疮样狼疮、共济失调性毛细血管扩张(也称为Louis-Bar综合征)、伴有脑的脑白质营养不良的视网膜血管病变(RCVL)、ANCA)-相关性血管炎、慢性疲劳综合征、家族性自主神经异常(dysautonomia)、脑脊髓炎、强直性脊柱炎、再生障碍性贫血、化脓性汗腺炎、自身免疫性肝炎、自身免疫性卵巢炎、乳糜泻、克罗恩氏病、1型糖尿病、巨细胞动脉炎、古德帕斯彻综合征、格雷夫斯病、吉兰-巴雷综合征、桥本氏病、过敏性紫癜、川崎病、红斑狼疮、显微镜下结肠炎、显微镜下多动脉炎、混合性结缔组织病、多发性硬化症、重症肌无力、眼阵挛-肌阵挛综合征(opsoclonus myoclonus syndrome)、视神经炎、ord's甲状腺炎、天疱疮、结节性多动脉炎、多肌痛、类风湿性关节炎、莱特尔氏综合征、干燥综合征、颞动脉炎、韦格纳肉芽肿病、温抗体型自身免疫性溶血性贫血(warm autoimmune hemolytic anemia)、间质性膀胱炎、莱姆病、硬斑病、牛皮癣、结节病、硬皮病、溃疡性结肠炎、慢性阻塞性肺病、成人呼吸窘迫综合征和白癜风。The compounds of the invention are useful for treating autoimmune disorders with an inflammatory component. Such disorders include acute disseminated alopecia universalise, Behcet's disease, Chagas disease, STING-associated vasculitis of infancy (SAVI), Aicardi Goutieres syndrome (AGS), lupus pernio, ataxia telangiectasia (also known as Louis-Bar syndrome), retinal vasculopathy with cerebral leukodystrophy (RCVL), ANCA-associated vasculitis, chronic fatigue syndrome, dysautonomia, encephalomyelitis, ankylosing spondylitis, aplastic anemia, hidradenitis suppurativa, autoimmune hepatitis, autoimmune oophoritis, celiac disease, Crohn's disease, type 1 diabetes mellitus, giant cell arteritis, Goodpasture's syndrome, Graves' disease, Guillain-Barré syndrome, Hashimoto's disease, Henoch-Schonlein purpura, Kawasaki disease, lupus erythematosus, microscopic colitis, microscopic polyarteritis, mixed connective tissue disease, multiple sclerosis, myasthenia gravis, opsoclonus myoclonus syndrome syndrome, optic neuritis, Ord's thyroiditis, pemphigus, polyarteritis nodosa, polymyalgia, rheumatoid arthritis, Reiter's syndrome, Sjögren's syndrome, temporal arteritis, Wegener's granulomatosis, warm autoimmune hemolytic anemia, interstitial cystitis, Lyme disease, morphea, psoriasis, sarcoidosis, scleroderma, ulcerative colitis, chronic obstructive pulmonary disease, adult respiratory distress syndrome, and vitiligo.
本发明化合物可用于治疗具有炎性组分的T细胞介导的超敏性疾病。这样的病症包括接触性超敏反应、接触性皮炎(包括由毒葛所致)、荨麻疹、皮肤过敏、呼吸道过敏(花粉症、过敏性鼻炎)和谷蛋白敏感性肠病(乳糜泻)。The compounds of the invention are useful for treating T cell mediated hypersensitivity diseases with an inflammatory component. Such conditions include contact hypersensitivity, contact dermatitis (including that caused by poison ivy), urticaria, skin allergies, respiratory allergies (hay fever, allergic rhinitis) and gluten sensitive enteropathy (celiac disease).
可用本发明化合物治疗的其它炎性病症包括例如阑尾炎、皮炎、皮肌炎、心内膜炎、纤维组织炎、牙龈炎、舌炎、肝炎、化脓性汗腺炎、虹膜炎、喉炎、乳腺炎、心肌炎、肾炎、耳炎、胰腺炎、腮腺炎、心外膜炎、腹膜炎、咽炎、胸膜炎、肺炎、前列腺炎、肾盂肾炎和口腔炎、移植排斥(涉及器官诸如肾、肝、心、肺、胰腺(例如,胰岛细胞)、骨髓、角膜、小肠、皮肤同种异体移植物、皮肤同种移植物和心脏瓣膜异种移植物、血清病和移植物抗宿主病)、急性胰腺炎、慢性胰腺炎、急性呼吸窘迫综合征、塞扎里综合征、先天性肾上腺增生、非化脓性甲状腺炎、与癌症相关的高钙血症、天疱疮、大疱性疱疹样皮炎(bullous dermatitisherpetiformis)、严重的多形性红斑、剥脱性皮炎、脂溢性皮炎、季节性或常年性过敏性鼻炎、支气管哮喘、接触性皮炎、异位性皮炎、药物超敏反应、过敏性结膜炎、角膜炎、眼带状疱疹、虹膜炎和虹膜睫状体炎、脉络膜视网膜炎、视神经炎、症状性结节病、爆发性或播撒性肺结核化疗、成人的特发性血小板减少性紫癜、成人的继发性血小板减少症、获得性(自身免疫性)溶血性贫血、成人白血病和淋巴瘤、儿童急性白血病、局限性肠炎、自身免疫性血管炎、多发性硬化症、慢性阻塞性肺病、实体器官移植排斥、败血症。优选的治疗包括移植排斥、类风湿性关节炎、牛皮癣性关节炎、多发性硬化症、1型糖尿病、哮喘、炎性肠病、系统性红斑狼疮、牛皮癣、慢性肺部疾病和伴感染病症(例如败血症)的炎症的治疗。在一个实施方案中,本发明化合物可用于治疗哮喘。Other inflammatory conditions that may be treated with the compounds of the invention include, for example, appendicitis, dermatitis, dermatomyositis, endocarditis, fibrositis, gingivitis, glossitis, hepatitis, hidradenitis suppurativa, iritis, laryngitis, mastitis, myocarditis, nephritis, otitis, pancreatitis, parotitis, pericarditis, peritonitis, pharyngitis, pleurisy, pneumonia, prostatitis, pyelonephritis, and stomatitis, transplant rejection (involving organs such as the kidney, liver, heart, lung, pancreas (e.g., islet cells), bone marrow, cornea, small intestine, skin allografts, skin allografts, and heart valve xenografts, serum sickness, and graft-versus-host disease), acute pancreatitis, chronic pancreatitis, acute respiratory distress syndrome, Sezary syndrome, congenital adrenal hyperplasia, non-suppurative thyroiditis, hypercalcemia associated with cancer, pemphigus, bullous dermatitis herpetiformis, and stomatitis. dermatitisherpetiformis), severe erythema multiforme, exfoliative dermatitis, seborrheic dermatitis, seasonal or perennial allergic rhinitis, bronchial asthma, contact dermatitis, atopic dermatitis, drug hypersensitivity, allergic conjunctivitis, keratitis, herpes zoster ophthalmicus, iritis and iridocyclitis, chorioretinitis, optic neuritis, symptomatic sarcoidosis, fulminant or disseminated pulmonary tuberculosis chemotherapy, idiopathic thrombocytopenic purpura in adults, secondary thrombocytopenia in adults, acquired (autoimmune) hemolytic anemia, adult leukemia and lymphoma, acute leukemia in children, regional enteritis, autoimmune vasculitis, multiple sclerosis, chronic obstructive pulmonary disease, solid organ transplant rejection, sepsis. Preferred treatments include treatment of transplant rejection, rheumatoid arthritis, psoriatic arthritis, multiple sclerosis, type 1 diabetes, asthma, inflammatory bowel disease, systemic lupus erythematosus, psoriasis, chronic lung disease and inflammation associated with infectious conditions (e.g., sepsis). In one embodiment, the compounds of the invention are useful in the treatment of asthma.
其中本发明化合物可具有潜在有益的抗肿瘤作用的癌症疾病和病症的实例包括但不限于肺、骨、胰腺、皮肤、头、颈、子宫、卵巢、胃、结肠、乳腺、食道、小肠、肠、内分泌系统、甲状腺、甲状旁腺、肾上腺、尿道、前列腺、阴茎、睾丸、输尿管、膀胱、肾或肝的癌症;直肠癌;肛门区癌;输卵管、子宫内膜、宫颈、阴道、外阴、肾盂、肾细胞的癌;软组织肉瘤;粘液瘤;横纹肌瘤;纤维瘤;脂肪瘤;畸胎瘤;胆管上皮癌;肝母细胞瘤;血管肉瘤;血管瘤;肝细胞瘤;纤维肉瘤;软骨肉瘤;骨髓瘤;慢性或急性白血病;淋巴细胞性淋巴瘤;原发性CNS淋巴瘤;CNS的肿瘤;脊髓(spinal axis)肿瘤;鳞状细胞癌;滑膜肉瘤;恶性胸膜间皮瘤;脑干胶质瘤;垂体腺瘤;支气管腺瘤;软骨瘤型错构瘤;间皮瘤;霍奇金氏病或一种或多种前述癌症的组合。Examples of cancer diseases and conditions in which the compounds of the invention may have a potentially beneficial anti-tumor effect include, but are not limited to, cancer of the lung, bone, pancreas, skin, head, neck, uterus, ovary, stomach, colon, breast, esophagus, small intestine, intestine, endocrine system, thyroid, parathyroid, adrenal glands, urethra, prostate, penis, testicles, ureter, bladder, kidney or liver; rectal cancer; anal region cancer; fallopian tube, endometrial, cervical, vagina, vulva, renal pelvis, renal cell; soft tissue sarcoma; myxoma; rhabdomyomas; fibromas; lipomas; teratomas; bile duct epithelial carcinomas; hepatoblastomas; angiosarcomas; hemangiomas; hepatomas; fibrosarcomas; chondrosarcomas; myeloma; chronic or acute leukemias; lymphocytic lymphomas; primary CNS lymphomas; tumors of the CNS; spinal cord (spinal cord) axis tumor; squamous cell carcinoma; synovial sarcoma; malignant pleural mesothelioma; brain stem glioma; pituitary adenoma; bronchial adenoma; enchondromatous hamartoma; mesothelioma; Hodgkin's disease, or a combination of one or more of the foregoing cancers.
合适地,本发明涉及用于治疗或减轻选自以下的癌症的严重程度的方法:脑癌(胶质瘤)、胶质母细胞瘤、星形细胞瘤、多形性胶质母细胞瘤、Bannayan-Zonana综合征、考登病、Lhermitte-Duclos病、威尔曼瘤、尤因氏肉瘤、横纹肌肉瘤、室管膜瘤、髓母细胞瘤、头颈癌、肾癌、肝癌、黑色素瘤、卵巢癌、胰腺癌、腺癌、导管腺癌、腺鳞癌、腺泡细胞癌、胰高血糖素瘤、胰岛素瘤、前列腺癌、肉瘤、骨肉瘤、骨巨细胞瘤、甲状腺癌、成淋巴细胞性T细胞白血病、慢性髓性白血病、慢性淋巴细胞性白血病、毛细胞白血病、急性成淋巴细胞性白血病、急性髓性白血病、慢性嗜中性白细胞性白血病、急性成淋巴细胞性T细胞白血病、浆细胞瘤、成免疫细胞性大细胞白血病、套细胞白血病、多发性骨髓瘤、成巨核细胞性白血病、多发性骨髓瘤、急性巨核细胞白血病、前髓细胞性白血病、红白血病、恶性淋巴瘤、霍奇金氏淋巴瘤、非霍奇金氏淋巴瘤、成淋巴细胞性T细胞淋巴瘤、伯基特淋巴瘤、滤泡淋巴瘤、神经母细胞瘤、膀胱癌、尿路上皮癌、外阴癌、宫颈癌、子宫内膜癌、肾癌、间皮瘤、食管癌、唾液腺癌、肝细胞癌、胃癌、鼻咽癌、颊癌、口腔癌、GIST(胃肠道间质瘤)和睾丸癌。在一些实施方案中,本发明化合物可用于治疗实体或液体肿瘤。在一些实施方案中,本发明化合物可用于治疗肉瘤、乳腺癌、结肠直肠癌、胃食管癌、黑色素瘤、非小细胞肺癌(NSCLC)、透明细胞肾细胞癌(RCC)、淋巴瘤、头颈部的鳞状细胞癌(SCCHN)、肝细胞癌(HCC)和/或非霍奇金氏淋巴瘤(NHL)。合适地,本发明涉及治疗或减轻哺乳动物(包括人)中的癌前综合征的严重程度的方法,其中所述癌前综合征选自:宫颈上皮内瘤样病变、意义未明的单克隆丙种球蛋白病(MGUS)、骨髓增生异常综合征、再生障碍性贫血、宫颈病变、皮肤痣(前黑色素瘤)、前列腺上皮内(导管内)瘤样病变(PIN)、导管原位癌(DCIS)、结肠息肉和重型肝炎或肝硬化。Suitably, the present invention relates to a method for treating or lessening the severity of a cancer selected from the group consisting of brain cancer (glioma), glioblastoma, astrocytoma, glioblastoma multiforme, Bannayan-Zonana syndrome, Cowden disease, Lhermitte-Duclos disease, Wilman tumor, Ewing's sarcoma, rhabdomyosarcoma, ependymoma, medulloblastoma, head and neck cancer, kidney cancer, liver cancer, melanoma, ovarian cancer, pancreatic cancer, adenocarcinoma, ductal adenocarcinoma, adenosquamous carcinoma, acinar cell carcinoma, glucagonoma, insulinoma, prostate cancer, sarcoma, osteosarcoma, giant cell tumor of bone, thyroid cancer, lymphoblastic T-cell leukemia, chronic myeloid leukemia, chronic lymphocytic leukemia, hairy cell leukemia, acute myeloid leukemia, Lymphocytic leukemia, acute myeloid leukemia, chronic neutrophilic leukemia, acute lymphoblastic T-cell leukemia, plasmacytoma, immunoblastic large cell leukemia, mantle cell leukemia, multiple myeloma, megakaryoblastic leukemia, multiple myeloma, acute megakaryocytic leukemia, promyelocytic leukemia, erythroleukemia, malignant lymphoma, Hodgkin's lymphoma, non-Hodgkin's lymphoma, lymphoblastic T-cell lymphoma, Burkitt's lymphoma, follicular lymphoma, neuroblastoma, bladder cancer, urothelial carcinoma, vulvar cancer, cervical cancer, endometrial cancer, renal cancer, mesothelioma, esophageal cancer, salivary gland cancer, hepatocellular carcinoma, gastric cancer, nasopharyngeal cancer, cheek cancer, oral cancer, GIST (gastrointestinal stromal tumor) and testicular cancer. In some embodiments, the compounds of the present invention can be used to treat solid or liquid tumors. In some embodiments, the compounds of the invention are useful for treating sarcomas, breast cancer, colorectal cancer, gastroesophageal cancer, melanoma, non-small cell lung cancer (NSCLC), clear cell renal cell carcinoma (RCC), lymphoma, squamous cell carcinoma of the head and neck (SCCHN), hepatocellular carcinoma (HCC) and/or non-Hodgkin's lymphoma (NHL). Suitably, the present invention relates to a method of treating or lessening the severity of a precancerous syndrome in a mammal, including a human, wherein the precancerous syndrome is selected from the group consisting of cervical intraepithelial neoplasia, monoclonal gammopathy of undetermined significance (MGUS), myelodysplastic syndrome, aplastic anemia, cervical lesions, skin nevus (premelanoma), prostatic intraepithelial (intraductal) neoplasia (PIN), ductal carcinoma in situ (DCIS), colon polyps, and severe hepatitis or cirrhosis.
在一个方面,人具有实体瘤。在一个方面,肿瘤选自头颈癌、胃癌、黑色素瘤、肾细胞癌(RCC)、食道癌、非小细胞肺癌、前列腺癌、结肠直肠癌、卵巢癌和胰腺癌。在一个方面,人具有以下中的一种或多种:结肠直肠癌(CRC)、食道癌、宫颈癌、膀胱癌、乳腺癌、头颈癌、卵巢癌、黑色素瘤、肾细胞癌(RCC)、EC鳞状细胞、非小细胞肺癌、间皮瘤和前列腺癌。在另一个方面,人具有液体肿瘤,诸如弥漫性大B细胞淋巴瘤(DLBCL)、多发性骨髓瘤、慢性成淋巴细胞性白血病(CLL)、滤泡性淋巴瘤、急性髓样白血病和慢性髓性白血病。In one aspect, people have solid tumors. In one aspect, tumors are selected from head and neck cancer, gastric cancer, melanoma, renal cell carcinoma (RCC), esophageal cancer, non-small cell lung cancer, prostate cancer, colorectal cancer, ovarian cancer and pancreatic cancer. In one aspect, people have one or more of the following: colorectal cancer (CRC), esophageal cancer, cervical cancer, bladder cancer, breast cancer, head and neck cancer, ovarian cancer, melanoma, renal cell carcinoma (RCC), EC squamous cells, non-small cell lung cancer, mesothelioma and prostate cancer. In another aspect, people have liquid tumors, such as diffuse large B cell lymphoma (DLBCL), multiple myeloma, chronic lymphoblastic leukemia (CLL), follicular lymphoma, acute myeloid leukemia and chronic myeloid leukemia.
在一个实施方案中,本发明化合物可用于治疗皮肤癌(例如,非黑色素瘤皮肤癌、鳞状细胞癌、基底细胞癌)或光化性角化病。除了用于清除浅表皮肤癌的场效应之外,本发明化合物还可以预防被治疗的患者中随后的皮肤癌和恶变前的光化性角化病的发展。In one embodiment, the compounds of the invention are useful for treating skin cancer (e.g., non-melanoma skin cancer, squamous cell carcinoma, basal cell carcinoma) or actinic keratosis. In addition to the field effect for clearing superficial skin cancers, the compounds of the invention may also prevent the development of subsequent skin cancers and premalignant actinic keratoses in the treated patient.
本发明化合物还可用于治疗一种或多种困扰哺乳动物的疾病,所述疾病的特征在于与新血管形成和/或血管通透性相关的病症领域中的细胞增殖,包括血管增殖性病症,其包括关节炎(类风湿性关节炎)和再狭窄;纤维化病症,其包括肝硬化和动脉粥样硬化;系膜细胞增殖性病症,包括肾小球肾炎、糖尿病性肾病、恶性肾硬化、血栓性微血管病综合征、增殖性视网膜病、器官移植排斥和肾小球病;和代谢病症,包括牛皮癣、糖尿病、慢性伤口愈合、炎症和神经变性疾病。The compounds of the invention may also be used to treat one or more diseases afflicting mammals characterized by cell proliferation in the field of disorders associated with neovascularization and/or vascular permeability, including vascular proliferative disorders, including arthritis (rheumatoid arthritis) and restenosis; fibrotic disorders, including cirrhosis and atherosclerosis; mesangial cell proliferative disorders, including glomerulonephritis, diabetic nephropathy, malignant nephrosclerosis, thrombotic microangiopathy syndrome, proliferative retinopathy, organ transplant rejection and glomerulopathy; and metabolic disorders, including psoriasis, diabetes, chronic wound healing, inflammation and neurodegenerative diseases.
本发明化合物可用于治疗神经变性疾病。示例性神经变性疾病包括但不限于多发性硬化症、亨廷顿氏舞蹈病、阿尔茨海默氏病、帕金森氏病、肌萎缩侧索硬化症(ALS)和额颞叶变性(FTD)。The compounds of the invention can be used to treat neurodegenerative diseases. Exemplary neurodegenerative diseases include, but are not limited to, multiple sclerosis, Huntington's disease, Alzheimer's disease, Parkinson's disease, amyotrophic lateral sclerosis (ALS), and frontotemporal lobar degeneration (FTD).
本发明化合物可以用于治疗或预防代谢疾病 (诸如胰岛素抗性、非酒精性脂肪肝病 (NAFLD)/ 非酒精性脂肪肝炎 (NASH)、肥胖症、糖尿病、高血压、脂肪肝和心血管疾病。The compounds of the present invention can be used to treat or prevent metabolic diseases (such as insulin resistance, non-alcoholic fatty liver disease (NAFLD)/non-alcoholic steatohepatitis (NASH), obesity, diabetes, hypertension, fatty liver and cardiovascular disease.
本发明化合物可用于治疗感染性疾病,其为由病原体感染引起或与病原体感染同时发生的任何疾病。病原体被广义地定义为对人体组织环境而言是外来的任何生物物种。常见的引起疾病的病原体包括细菌(许多,如TB)、病毒(许多,如HBV、HIV、流感病毒)和寄生原生动物(如引起疟疾的恶性疟原虫)。本发明化合物可用于治疗源自细菌的感染性疾病,诸如TB感染(结核分枝杆菌)、衣原体感染、土拉菌感染(土拉弗朗西斯菌)、疟原虫感染或来自DNA或RNA病毒的感染。本发明化合物可用于治疗源自以下DNA病毒家族的感染性疾病:疱疹病毒科(单纯疱疹病毒-1、卡波西氏肉瘤相关病毒和爱泼斯坦-巴尔病毒)、乳头瘤病毒科(人乳头瘤病毒)、腺病毒和嗜肝DNA病毒科(乙型肝炎病毒)。RNA病毒科的实例包括逆转录病毒科(人免疫缺陷病毒)黄病毒科(登革病毒、丙型肝炎病毒)、正粘病毒科(流感)和冠状病毒科(人冠状病毒和SARS冠状病毒)。The compounds of the present invention can be used to treat infectious diseases, which are any diseases caused by pathogen infection or occurring simultaneously with pathogen infection. Pathogens are broadly defined as any biological species that are foreign to the human tissue environment. Common pathogens that cause diseases include bacteria (many, such as TB), viruses (many, such as HBV, HIV, influenza viruses) and parasitic protozoa (such as Plasmodium falciparum that causes malaria). The compounds of the present invention can be used to treat infectious diseases derived from bacteria, such as TB infection (Mycobacterium tuberculosis), chlamydia infection, tularemia infection (Francisco tularemia), Plasmodium infection or infection from DNA or RNA viruses. The compounds of the present invention can be used to treat infectious diseases derived from the following DNA virus families: Herpesviridae (herpes simplex virus-1, Kaposi's sarcoma-associated virus and Epstein-Barr virus), Papillomaviridae (human papillomavirus), adenovirus and Hepadnaviridae (hepatitis B virus). Examples of RNA virus families include Retroviridae (human immunodeficiency virus), Flaviviridae (dengue virus, hepatitis C virus), Orthomyxoviridae (influenza), and Coronaviridae (human coronavirus and SARS coronavirus).
本发明化合物可以单独使用或与其它治疗剂组合使用。作为免疫应答的调节剂,本发明化合物还可以用于单一疗法中或与另一种治疗剂组合用于治疗其中STING的调节有益的疾病和病况。因此,根据本发明的组合疗法包括施用式(I)化合物或其药学上可接受的盐和至少一种其它治疗活性剂。在一个实施方案中,根据本发明的组合疗法包括施用至少一种式(I)化合物或其药学上可接受的盐和至少一种其它治疗剂。式(I)化合物及其药学上可接受的盐和其它治疗剂可以在单一药物组合物中一起施用或分开施用,并且当分开施用时,这可以同时或以任何顺序依次进行。选择式(I)化合物及其药学上可接受的盐和其它治疗剂的量和施用的相对时机以实现期望的组合治疗效果。因此,在一个进一步方面,提供了组合产品,其包含式(I)化合物或其药学上可接受的盐以及一种或多种其它治疗剂。The compounds of the present invention can be used alone or in combination with other therapeutic agents. As modulators of immune responses, the compounds of the present invention can also be used in monotherapy or in combination with another therapeutic agent for the treatment of diseases and conditions in which the regulation of STING is beneficial. Therefore, the combination therapy according to the present invention comprises administering a compound of formula (I) or a pharmaceutically acceptable salt thereof and at least one other therapeutically active agent. In one embodiment, the combination therapy according to the present invention comprises administering at least one compound of formula (I) or a pharmaceutically acceptable salt thereof and at least one other therapeutic agent. The compound of formula (I) and its pharmaceutically acceptable salt and the other therapeutic agent can be administered together or separately in a single pharmaceutical composition, and when administered separately, this can be performed simultaneously or sequentially in any order. The amount of the compound of formula (I) and its pharmaceutically acceptable salt and the other therapeutic agent and the relative timing of administration are selected to achieve the desired combined therapeutic effect. Therefore, in a further aspect, a combination product is provided, which comprises a compound of formula (I) or a pharmaceutically acceptable salt thereof and one or more other therapeutic agents.
式(I)化合物及其药学上可接受的盐可以与一种或多种可用于预防或治疗过敏性疾病、炎性疾病或自身免疫性疾病的其它治疗剂组合使用,所述治疗剂例如:抗原免疫疗法、抗组胺剂、甾类化合物、NSAID、支气管扩张剂(例如β2激动剂、肾上腺素能激动剂、抗胆碱能剂、茶碱)、甲氨蝶呤、白细胞三烯调节剂和类似药剂;单克隆抗体疗法诸如抗IgE、抗TNF、抗IL-5、抗IL-6、抗IL-12、抗IL-1和类似药剂;受体疗法例如依那西普和类似药剂;抗原非特异性免疫疗法(例如干扰素或其它细胞因子/趋化因子、细胞因子/趋化因子受体调节剂、细胞因子激动剂或拮抗剂、TLR激动剂和类似药剂)。The compounds of formula (I) and their pharmaceutically acceptable salts can be used in combination with one or more other therapeutic agents useful for preventing or treating allergic diseases, inflammatory diseases or autoimmune diseases, such as: antigen immunotherapy, antihistamines, steroids, NSAIDs, bronchodilators (e.g., β2 agonists, adrenergic agonists, anticholinergics, theophylline), methotrexate, leukotriene modifiers and similar agents; monoclonal antibody therapy such as anti-IgE, anti-TNF, anti-IL-5, anti-IL-6, anti-IL-12, anti-IL-1 and similar agents; receptor therapy such as etanercept and similar agents; antigen nonspecific immunotherapy (e.g., interferon or other cytokines/chemokines, cytokine/chemokine receptor modulators, cytokine agonists or antagonists, TLR agonists and similar agents).
式(I)化合物及其药学上可接受的盐可以与放射疗法和/或手术和/或至少一种可用于治疗癌症和癌前综合征的其它治疗剂组合使用。任何对于所治疗的易受影响的肿瘤具有活性的抗肿瘤剂可以用于组合中。可用的典型抗肿瘤剂包括但不限于抗微管剂,诸如二萜类化合物和长春花生物碱;铂配位络合物;烷基化试剂,诸如氮芥类、氧氮磷环类(oxazaphosphorines)、烷基磺酸酯类、亚硝基脲类和三氮烯类;抗生素药剂,诸如蒽环类、放线菌素类和博来霉素类;拓扑异构酶II抑制剂,诸如表鬼臼毒素类;抗代谢物,诸如嘌呤和嘧啶类似物和抗叶酸化合物;拓扑异构酶I抑制剂,诸如喜树碱类;激素和激素类似物;信号转导途径抑制剂;非受体酪氨酸血管生成抑制剂;免疫治疗剂;促凋亡剂;细胞周期信号转导抑制剂;免疫-肿瘤学剂和免疫刺激剂。Formula (I) compound and its pharmaceutically acceptable salt can be used in combination with radiotherapy and/or surgery and/or at least one other therapeutic agent that can be used to treat cancer and precancerous syndrome. Any antitumor agent that is active for the susceptible tumor to be treated can be used in combination. Available typical antitumor agents include but are not limited to anti-microtubule agents, such as diterpenoids and vinca alkaloids; platinum coordination complexes; alkylating agents, such as nitrogen mustards, oxazaphosphorines, alkyl sulfonates, nitrosoureas and triazenes; antibiotic agents, such as anthracyclines, actinomycins and bleomycins; topoisomerase II inhibitors, such as epipodophyllotoxins; antimetabolites, such as purine and pyrimidine analogs and antifolate compounds; topoisomerase I inhibitors, such as camptothecins; hormones and hormone analogs; signal transduction pathway inhibitors; non-receptor tyrosine angiogenesis inhibitors; immunotherapeutics; pro-apoptotic agents; cell cycle signal transduction inhibitors; immuno-oncology agents and immunostimulants.
抗微管或抗有丝分裂剂是在细胞周期的M期或有丝分裂期对肿瘤细胞的微管具有活性的阶段特异性药剂。抗微管剂的实例包括但不限于二萜类化合物和长春花生物碱。Anti-microtubule or anti-mitotic agents are phase-specific agents that are active against the microtubules of tumor cells during the M phase or mitosis of the cell cycle. Examples of anti-microtubule agents include, but are not limited to, diterpenoids and vinca alkaloids.
源自天然来源的二萜类化合物是在细胞周期的G2/M期起作用的阶段特异性抗癌剂。据信,二萜类化合物通过与该蛋白质结合来稳定微管的β-微管蛋白亚单位。然后蛋白质的分解似乎受到抑制,同时阻止有丝分裂,随后细胞死亡。二萜类化合物的实例包括但不限于紫杉醇及其类似物多西他赛。Diterpenoids derived from natural sources are phase-specific anticancer agents that act during the G2 /M phase of the cell cycle. It is believed that diterpenoids stabilize the β-tubulin subunit of microtubules by binding to this protein. The breakdown of the protein then appears to be inhibited, with a simultaneous arrest of mitosis followed by cell death. Examples of diterpenoids include, but are not limited to, paclitaxel and its analog docetaxel.
紫杉醇, 即5β,20-环氧-1,2α,4,7β,10β,13α-六羟基紫杉烷-11-烯-9-酮4,10-二乙酸酯2-苯甲酸酯-13-[(2R,3S)-N-苯甲酰基-3-苯基异丝氨酸酯],是从太平洋紫杉树短叶红豆杉(Taxus brevifolia)中分离的天然二萜产物,并且可作为注射液TAXOL®商购得到。它是萜烯的紫杉烷家族的成员。紫杉醇已在美国被批准临床用于治疗难治性卵巢癌(Markman等人,Yale Journal of Biology and Medicine,64:583,1991;McGuire等人,Ann. lntem,Med.,111:273,1989)和用于治疗乳腺癌(Holmes等人,J. Nat. CancerInst.,83:1797,1991)。其是治疗皮肤肿瘤(Einzig等人,Proc. Am. Soc. Clin. Oncol.,20:46)和头颈癌(Forastire等人,Sem. Oncol.,20:56,1990)的潜在候选物。该化合物还显示了治疗多囊性肾病(Woo等人,Nature,368:750. 1994)、肺癌和疟疾的潜力。用紫杉醇治疗患者导致与高于阈浓度(50nM)的给药的持续时间有关的(Kearns,C.M.等人,Seminarsin Oncology,3(6) p.16-23,1995)骨髓抑制(多细胞谱系,Ignoff,R.J.等人,CancerChemotherapy Pocket Guide,1998)。Paclitaxel, 5β,20-epoxy-1,2α,4,7β,10β,13α-hexahydroxytaxane-11-ene-9-one 4,10-diacetate 2-benzoate-13-[(2R,3S)-N-benzoyl-3-phenylisoserine ester], is a natural diterpene product isolated from the Pacific yew tree Taxus brevifolia and is commercially available as an injection solution TAXOL®. It is a member of the taxane family of terpenes. Paclitaxel has been approved for clinical use in the United States for the treatment of refractory ovarian cancer (Markman et al., Yale Journal of Biology and Medicine, 64:583, 1991; McGuire et al., Ann. Intem, Med., 111:273, 1989) and for the treatment of breast cancer (Holmes et al., J. Nat. Cancer Inst., 83:1797, 1991). It is a potential candidate for the treatment of skin tumors (Einzig et al., Proc. Am. Soc. Clin. Oncol., 20:46) and head and neck cancer (Forastire et al., Sem. Oncol., 20:56, 1990). The compound also shows potential for the treatment of polycystic kidney disease (Woo et al., Nature, 368:750. 1994), lung cancer and malaria. Treatment of patients with paclitaxel results in bone marrow suppression (multiple cell lineages, Ignoff, RJ et al., Cancer Chemotherapy Pocket Guide , 1998) associated with the duration of administration above the threshold concentration (50 nM) (Kearns, CM et al., Seminars in Oncology, 3 (6) p.16-23, 1995).
多西他赛,即(2R,3S)-N-羧基-3-苯基异丝氨酸,N-叔丁基酯,13-[5β-20-环氧-1,2α,4,7β,10β,13α-六羟基紫杉烷-11-烯-9-酮4-乙酸酯2-苯甲酸酯]三水合物;可作为注射液以TAXOTERE®商购得到。多西他赛指示用于治疗乳腺癌。多西他赛是使用从欧洲紫杉树的针叶提取的天然前体10-去乙酰基-浆果赤霉素III制备的紫杉醇q.v.的半合成衍生物。Docetaxel, (2R,3S)-N-carboxy-3-phenylisoserine, N-tert-butyl ester, 13-[5β-20-epoxy-1,2α,4,7β,10β,13α-hexahydroxytaxane-11-en-9-one 4-acetate 2-benzoate] trihydrate; commercially available as TAXOTERE® as an injection. Docetaxel is indicated for the treatment of breast cancer. Docetaxel is a semisynthetic derivative of paclitaxel qv prepared using the natural precursor 10-deacetyl-baccatin III extracted from the needles of the European yew tree.
长春花生物碱是源自长春花属的植物的阶段特异性抗肿瘤剂。长春花生物碱通过与微管蛋白特异性结合而作用于细胞周期的M期(有丝分裂)。因此,结合的微管蛋白分子不能聚合成微管。有丝分裂被认为在中期停止,随后细胞死亡。长春花生物碱的实例包括但不限于长春碱、长春新碱和长春瑞滨。Vinca alkaloids are stage-specific antitumor agents derived from plants of the genus Vinca. Vinca alkaloids act on the M phase (mitosis) of the cell cycle by specifically binding to tubulin. Therefore, the combined tubulin molecules cannot be polymerized into microtubules. Mitosis is considered to stop in the metaphase, followed by cell death. The example of vinca alkaloids includes but is not limited to vinblastine, vincristine and vinorelbine.
长春碱,即长春花碱硫酸盐,可作为注射液以VELBAN®商购得到。尽管其可能指示作为各种实体瘤的二线治疗,但其主要指示用于治疗睾丸癌和各种淋巴瘤,包括霍奇金氏病;以及淋巴细胞性和组织细胞性淋巴瘤。骨髓抑制是长春碱的剂量限制性副作用。Vinblastine, vinblastine sulfate, is commercially available as an injectable solution as VELBAN® . Although it may be indicated as a second-line treatment for various solid tumors, its primary indications are for the treatment of testicular cancer and various lymphomas, including Hodgkin's disease; and lymphocytic and histiocytic lymphomas. Myelosuppression is a dose-limiting side effect of vinblastine.
长春新碱,即22-氧代-长春花碱硫酸盐,可以作为注射液以ONCOVIN®商购得到。长春新碱指示用于治疗急性白血病,并且还发现可用于霍奇金氏和非霍奇金氏恶性淋巴瘤的治疗方案。脱发和神经学作用是长春新碱的最常见的副作用,并且在较小程度上发生骨髓抑制和胃肠道粘膜炎作用。Vincristine, 22-oxo-vinblastine sulfate, is commercially available as an injection solution as ONCOVIN® . Vincristine is indicated for the treatment of acute leukemias and has also found use in treatment regimens for Hodgkin's and non-Hodgkin's malignant lymphomas. Alopecia and neurological effects are the most common side effects of vincristine, and myelosuppression and gastrointestinal mucositis effects occur to a lesser extent.
长春瑞滨,即3’,4’-二去氢-4’-去氧-C’-去甲长春碱(norvincaleukoblastine)[R-(R*,R*)-2,3-二羟基丁二酸盐(1:2)(盐)],可作为酒石酸长春瑞滨注射液(NAVELBINE®)商购得到,其是一种半合成长春花生物碱。长春瑞滨指示为作为单一药剂或与其它化疗剂诸如顺铂组合用于治疗各种实体瘤,特别是非小细胞肺癌、晚期乳腺癌和激素难治性前列腺癌。骨髓抑制是长春瑞滨的最常见的剂量限制性副作用。Vinorelbine, 3',4'-didehydro-4'-deoxy-C'-norvincaleukoblastine [R-(R*,R*)-2,3-dihydroxysuccinate (1:2) (salt)], is commercially available as vinorelbine tartrate injection ( NAVELBINE® ), a semisynthetic vinca alkaloid. Vinorelbine is indicated for the treatment of various solid tumors, particularly non-small cell lung cancer, advanced breast cancer, and hormone-refractory prostate cancer, as a single agent or in combination with other chemotherapeutic agents such as cisplatin. Myelosuppression is the most common dose-limiting side effect of vinorelbine.
铂配位络合物是非阶段特异性抗癌剂,其与DNA相互作用。铂络合物进入肿瘤细胞,经历水合作用并与DNA形成链内和链间交联,引起对肿瘤不利的生物效应。铂配位络合物的实例包括但不限于奥沙利铂、顺铂和卡铂。Platinum coordination complexes are non-stage specific anticancer agents that interact with DNA. Platinum complexes enter tumor cells, undergo hydration and form intra- and inter-strand crosslinks with DNA, causing biological effects that are unfavorable to tumors. Examples of platinum coordination complexes include, but are not limited to, oxaliplatin, cisplatin, and carboplatin.
顺铂,即顺式-二氯二胺合铂,可作为注射液以PLATINOL®商购得到。顺铂主要指示用于治疗转移性睾丸癌和卵巢癌以及晚期膀胱癌。Cisplatin, cis-dichlorodiamine platinum, is commercially available as an injectable solution as PLATINOL® . Cisplatin is primarily indicated for the treatment of metastatic testicular and ovarian cancer and advanced bladder cancer.
卡铂,即二胺[1,1-环丁烷-二羧酸根(2-)-O,O’]合铂,可以作为注射液以PARAPLATIN®商购得到。卡铂主要指示用于晚期卵巢癌的一线和二线治疗。Carboplatin, diamine[1,1-cyclobutane-dicarboxylate (2-)-O,O']platinum, is commercially available as an injection as PARAPLATIN® . Carboplatin is primarily indicated for first-line and second-line treatment of advanced ovarian cancer.
烷基化试剂是非阶段抗癌特异性试剂和强亲电试剂。典型地,烷基化试剂由烷基化通过DNA分子的亲核部分诸如磷酸基(phosphate)、氨基、巯基、羟基、羧基和咪唑基与DNA形成共价键。这样的烷基化破坏了核酸功能,导致细胞死亡。烷基化试剂的实例包括但不限于氮芥类,诸如环磷酰胺、美法仑和苯丁酸氮芥;烷基磺酸酯类,诸如白消安;亚硝基脲类,诸如卡莫司汀;和三氮烯类,诸如达卡巴嗪。Alkylating agents are non-phasic anticancer specific agents and strong electrophiles. Typically, alkylating agents form covalent bonds with DNA through nucleophilic moieties such as phosphate, amino, sulfhydryl, hydroxyl, carboxyl and imidazole groups of DNA molecules by alkylation. Such alkylation destroys nucleic acid function and causes cell death. Examples of alkylating agents include, but are not limited to, nitrogen mustards such as cyclophosphamide, melphalan and chlorambucil; alkyl sulfonates such as busulfan; nitrosoureas such as carmustine; and triazenes such as dacarbazine.
环磷酰胺,即2-[双(2-氯乙基)氨基]四氢-2H-1,3,2-氧氮磷杂环己烷2-氧化物一水合物,可作为注射液或片剂以CYTOXAN®商购得到。环磷酰胺指示用作单一药剂或与其它化疗剂组合用于治疗恶性淋巴瘤、多发性骨髓瘤和白血病。Cyclophosphamide, 2-[bis(2-chloroethyl)amino]tetrahydro-2H-1,3,2-oxazaphosphorinane 2-oxide monohydrate, is commercially available as an injectable solution or tablet as CYTOXAN® . Cyclophosphamide is indicated for use as a single agent or in combination with other chemotherapeutic agents in the treatment of malignant lymphomas, multiple myeloma, and leukemias.
美法仑,即4-[双(2-氯乙基)氨基]-L-苯丙氨酸,可作为注射液或片剂以ALKERAN®商购得到。美法仑指示用于姑息性治疗多发性骨髓瘤和不可切除的卵巢上皮癌。骨髓抑制是美法仑的最常见的剂量限制性副作用。Melphalan, 4-[bis(2-chloroethyl)amino]-L-phenylalanine, is commercially available as an injection or tablet as ALKERAN® . Melphalan is indicated for the palliative treatment of multiple myeloma and unresectable epithelial ovarian cancer. Myelosuppression is the most common dose-limiting side effect of melphalan.
苯丁酸氮芥,即4-[双(2-氯乙基)氨基]苯丁酸,可作为LEUKERAN®片剂商购得到。苯丁酸氮芥指示用于姑息性治疗慢性淋巴性白血病和恶性淋巴瘤诸如淋巴肉瘤、巨滤泡型淋巴瘤和霍奇金氏病。Chlorambucil, 4-[bis(2-chloroethyl)amino]phenylbutyric acid, is commercially available as LEUKERAN® tablets. Chlorambucil is indicated for the palliative treatment of chronic lymphatic leukemia and malignant lymphomas such as lymphosarcoma, giant follicular lymphoma, and Hodgkin's disease.
白消安,即二甲磺酸-1,4-丁二醇酯,可作为MYLERAN®片剂(MYLERAN® TABLETS)商购得到。白消安指示用于姑息性治疗慢性骨髓性白血病。Busulfan, 1,4-butylene glycol dimethanesulfonate, is commercially available as MYLERAN ® TABLETS . Busulfan is indicated for the palliative treatment of chronic myeloid leukemia.
卡莫司汀,即1,3-[双(2-氯乙基)-1-亚硝基脲,可作为单瓶的冻干材料以BiCNU®商购获得。卡莫司汀指示作为单一药剂或与其它药剂组合用于姑息性治疗脑肿瘤、多发性骨髓瘤、霍奇金氏病和非霍奇金氏淋巴瘤。Carmustine, 1,3-bis(2-chloroethyl)-1-nitrosourea, is commercially available as a single vial of lyophilized material as BiCNU® . Carmustine is indicated as a single agent or in combination with other agents for the palliative treatment of brain tumors, multiple myeloma, Hodgkin's disease, and non-Hodgkin's lymphoma.
达卡巴嗪,即5-(3,3-二甲基-1-三氮烯基)-咪唑-4-甲酰胺,可作为单瓶的物质以DTIC-Dome®商购获得。达卡巴嗪指示用于治疗转移性恶性黑色素瘤和用于与其它药剂组合用于霍奇金氏病的二线治疗。Dacarbazine, 5-(3,3-dimethyl-1-triazene)-imidazole-4-carboxamide, is commercially available as a single vial of the substance as DTIC- Dome® . Dacarbazine is indicated for the treatment of metastatic malignant melanoma and for second-line treatment of Hodgkin's disease in combination with other agents.
抗生素抗肿瘤剂是非阶段特异性药剂,其结合或插入DNA。典型地,这样的作用导致稳定的DNA络合物或链断裂,其破坏核酸的正常功能,导致细胞死亡。抗生素抗肿瘤剂的实例包括但不限于放线菌素类诸如更生霉素、蒽环类诸如柔红霉素和多柔比星;和博来霉素类。Antibiotic antineoplastic agents are non-stage specific agents that bind or intercalate into DNA. Typically, such action results in stable DNA complexes or strand breaks that disrupt the normal function of nucleic acids, leading to cell death. Examples of antibiotic antineoplastic agents include, but are not limited to, actinomycins such as dactinomycin, anthracyclines such as daunorubicin and doxorubicin; and bleomycins.
更生霉素,也称为放线菌素D,可以注射形式以COSMEGEN®商购得到。更生霉素指示用于治疗威尔曼瘤和横纹肌肉瘤。Dactinomycin, also known as actinomycin D, is commercially available in injectable form as COSMEGEN® . Dactinomycin is indicated for the treatment of Wilman tumor and rhabdomyosarcoma.
柔红霉素,即(8S-顺式)-8-乙酰基-10-[(3-氨基-2,3,6-三去氧-α-L-来苏-己吡喃糖基)氧基]-7,8,9,10-四氢-6,8,11-三羟基-1-甲氧基-5,12并四苯二酮盐酸盐,可作为脂质体注射形式以DAUNOXOME®或作为注射剂以CERUBIDINE®商购得到。柔红霉素指示用于在急性非淋巴细胞性白血病和晚期HIV相关的卡波西肉瘤的治疗中诱导缓解。Daunorubicin, (8S-cis)-8-acetyl-10-[(3-amino-2,3,6-trideoxy-α-L-lyxo-hexopyranosyl)oxy]-7,8,9,10-tetrahydro-6,8,11-trihydroxy-1-methoxy-5,12-naphthacene dione hydrochloride, is commercially available as a liposomal injectable form as DAUNOXOME® or as an injectable as CERUBIDINE® . Daunorubicin is indicated for inducing remission in the treatment of acute non-lymphocytic leukemia and advanced HIV-associated Kaposi's sarcoma.
多柔比星,即(8S,10S)-10-[(3-氨基-2,3,6-三去氧-α-L-来苏-己吡喃糖基)氧基]-8-乙醇酰基,7,8,9,10-四氢-6,8,11-三羟基-1-甲氧基-5,12并四苯二酮盐酸盐,可作为注射形式以RUBEX®或ADRIAMYCIN RDF®商购得到。多柔比星主要指示用于治疗急性成淋巴细胞性白血病和急性髓母细胞白血病,但也是治疗一些实体瘤和淋巴瘤的有用成分。Doxorubicin, (8S,10S)-10-[(3-amino-2,3,6-trideoxy-α-L-lyso-hexopyranosyl)oxy]-8-glycolyl, 7,8,9,10-tetrahydro-6,8,11-trihydroxy-1-methoxy-5,12-naphthacene dione hydrochloride, is commercially available as an injectable form as RUBEX® or ADRIAMYCIN RDF® . Doxorubicin is primarily indicated for the treatment of acute lymphoblastic leukemia and acute myeloblastic leukemia, but is also a useful agent in the treatment of some solid tumors and lymphomas.
博来霉素,即从轮枝链霉菌(Streptomyces verticillus)菌株中分离的细胞毒性糖肽类抗生素的混合物,可以BLENOXANE®商购得到。博来霉素指示作为单一药剂或与其它药剂组合用于姑息性治疗鳞状细胞癌、淋巴瘤和睾丸癌。Bleomycin, a mixture of cytotoxic glycopeptide antibiotics isolated from strains of Streptomyces verticillus , is commercially available as BLENOXANE® . Bleomycin is indicated as a single agent or in combination with other agents for the palliative treatment of squamous cell carcinoma, lymphoma, and testicular cancer.
拓扑异构酶II抑制剂包括但不限于表鬼臼毒素类。表鬼臼毒素类是源自盾叶鬼臼(mandrake)植物的阶段特异性抗肿瘤剂。表鬼臼毒素类通常通过与拓扑异构酶II和DNA形成三元复合物引起DNA链断裂而影响细胞周期的S和G2期中的细胞。链断裂积累,接着细胞死亡。表鬼臼毒素类的实例包括但不限于依托泊苷和替尼泊苷。Topoisomerase II inhibitors include, but are not limited to, epipodophyllotoxins. Epipodophyllotoxins are stage-specific antitumor agents derived from the mandrake plant. Epipodophyllotoxins typically affect cells in the S and G2 phases of the cell cycle by forming a ternary complex with topoisomerase II and DNA, causing DNA strand breaks. Strand breaks accumulate, followed by cell death. Examples of epipodophyllotoxins include, but are not limited to, etoposide and teniposide.
依托泊苷,即4’-去甲基-表鬼臼毒素9[4,6-0-(R)-亚乙基-β-D-吡喃葡萄糖苷],可作为注射液或胶囊以VePESID®商购获得,并且通常称为VP-16。依托泊苷指示作为单一药剂或与其它化疗剂组合用于治疗睾丸癌和非小细胞肺癌。Etoposide, 4'-demethyl-epipodophyllotoxin 9[4,6-0-(R)-ethylidene-β-D-pyranoglucopyranoside], is commercially available as an injectable solution or capsule as VePESID® and is commonly known as VP- 16. Etoposide is indicated as a single agent or in combination with other chemotherapeutic agents for the treatment of testicular cancer and non-small cell lung cancer.
替尼泊苷,即4’-去甲基-表鬼臼毒素9[4,6-0-(R)-噻吩亚甲基-β-D-吡喃葡萄糖苷],可作为注射液以VUMON®商购获得,并且通常称为VM-26。替尼泊苷指示作为单一药剂或与其它化疗剂组合用于治疗儿童的急性白血病。Teniposide, 4'-demethyl-epipodophyllotoxin 9[4,6-0-(R)-thenylidene-β-D-pyranoglucopyranoside], is commercially available as an injection as VUMON® and is commonly known as VM-26. Teniposide is indicated as a single agent or in combination with other chemotherapeutic agents for the treatment of acute leukemia in children.
抗代谢物肿瘤剂是通过抑制DNA合成或通过抑制嘌呤或嘧啶碱基合成从而限制DNA合成而作用于细胞周期的S期(DNA合成)的阶段特异性抗肿瘤剂。因此,S期不继续进行,接着细胞死亡。抗代谢物抗肿瘤剂的实例包括但不限于氟尿嘧啶、甲氨蝶呤、阿糖胞苷、巯基嘌呤、硫鸟嘌呤和吉西他滨。Antimetabolite tumor agents are stage-specific antitumor agents that act on the S phase (DNA synthesis) of the cell cycle by inhibiting DNA synthesis or by inhibiting purine or pyrimidine base synthesis to limit DNA synthesis. Therefore, the S phase does not proceed, and then the cell dies. Examples of antimetabolite antitumor agents include, but are not limited to, fluorouracil, methotrexate, cytarabine, mercaptopurine, thioguanine, and gemcitabine.
5-氟尿嘧啶,即5-氟-2,4-(1H,3H)嘧啶二酮,可作为氟尿嘧啶商购获得。施用5-氟尿嘧啶导致抑制胸苷酸合成,并且也掺入到RNA和DNA二者中。结果通常是细胞死亡。5-氟尿嘧啶指示作为单一药剂或与其它化疗剂组合用于治疗乳腺癌、结肠癌、直肠癌、胃癌和胰腺癌。其它氟嘧啶类似物包括5-氟脱氧尿苷(氟尿苷)和5-氟脱氧尿苷单磷酸盐。5-fluorouracil, 5-fluoro-2,4-(1H,3H)pyrimidinedione, is commercially available as fluorouracil. Administration of 5-fluorouracil results in inhibition of thymidylate synthesis and is also incorporated into both RNA and DNA. The result is usually cell death. 5-fluorouracil is indicated for the treatment of breast cancer, colon cancer, rectal cancer, gastric cancer, and pancreatic cancer as a single agent or in combination with other chemotherapeutic agents. Other fluoropyrimidine analogs include 5-fluorodeoxyuridine (floxuridine) and 5-fluorodeoxyuridine monophosphate.
阿糖胞苷,即4-氨基-1-β-D-阿拉伯呋喃糖基-2(1H)-嘧啶酮,可作为CYTOSAR-U®商购获得,并且通常称为Ara-C。据信阿糖胞苷通过将阿糖胞苷末端掺入到生长的DNA链中来抑制DNA链延长,而在S期显示细胞阶段特异性。阿糖胞苷指示作为单一药剂或与其它化疗剂组合用于治疗急性白血病。其它胞苷类似物包括5-氮杂胞苷和2’,2’-二氟脱氧胞苷(吉西他滨)。Cytarabine, 4-amino-1-β-D-arabinofuranosyl-2(1H)-pyrimidinone, is commercially available as CYTOSAR- U® and is commonly referred to as Ara-C. It is believed that cytarabine inhibits DNA chain elongation by terminally incorporating cytarabine into growing DNA chains, while showing cell stage specificity in the S phase. Cytarabine is indicated for the treatment of acute leukemia as a single agent or in combination with other chemotherapeutic agents. Other cytidine analogs include 5-azacytidine and 2',2'-difluorodeoxycytidine (gemcitabine).
巯基嘌呤,即1,7-二氢-6H-嘌呤-6-硫酮一水合物,可作为PURINETHOL®商购得到。巯基嘌呤通过至今尚未确定的机制抑制DNA合成,而在S期显示细胞阶段特异性。巯基嘌呤指示作为单一药剂或与其它化疗剂组合用于治疗急性白血病。有用的巯基嘌呤类似物是硫唑嘌呤。Mercaptopurine, 1,7-dihydro-6H-purine-6-thione monohydrate, is commercially available as PURINETHOL® . Mercaptopurine inhibits DNA synthesis by a mechanism that has not yet been determined, but shows cell phase specificity in the S phase. Mercaptopurine is indicated for the treatment of acute leukemias as a single agent or in combination with other chemotherapeutic agents. A useful mercaptopurine analog is azathioprine.
硫鸟嘌呤,即2-氨基-1,7-二氢-6H-嘌呤-6-硫酮,可作为TABLOID®商购得到。硫鸟嘌呤通过至今尚未确定的机制抑制DNA合成,而在S期显示细胞阶段特异性。硫鸟嘌呤指示作为单一药剂或与其它化疗剂组合用于治疗急性白血病。其它嘌呤类似物包括喷司他丁、红羟基壬基腺嘌呤(erythrohydroxynonyladenine)(EHNA)、磷酸氟达拉滨和克拉屈滨。Thioguanine, 2-amino-1,7-dihydro-6H-purine-6-thione, is commercially available as TABLOID® . Thioguanine inhibits DNA synthesis by a mechanism that has not yet been determined, but shows cell phase specificity in the S phase. Thioguanine is indicated for the treatment of acute leukemias as a single agent or in combination with other chemotherapeutic agents. Other purine analogs include pentostatin, erythrohydroxynonyladenine (EHNA), fludarabine phosphate, and cladribine.
吉西他滨,即2’-脱氧-2’,2’-二氟胞苷一盐酸盐(β-异构体),可作为GEMZAR®商购得到。吉西他滨通过阻断细胞通过G1/S边界的进展而在S期显示细胞阶段特异性。吉西他滨指示与顺铂组合用于治疗局部晚期非小细胞肺癌和单独用于治疗局部晚期胰腺癌。Gemcitabine, 2'-deoxy-2',2'-difluorocytidine monohydrochloride (β-isomer), is commercially available as GEMZAR® . Gemcitabine exhibits cell phase specificity in the S phase by blocking the progression of cells through the G1/S boundary. Gemcitabine is indicated in combination with cisplatin for the treatment of locally advanced non-small cell lung cancer and alone for the treatment of locally advanced pancreatic cancer.
甲氨蝶呤,即N-[4[[(2,4-二氨基-6-蝶啶基)甲基]甲基氨基]苯甲酰基]-L-谷氨酸,可作为甲氨蝶呤钠商购得到。甲氨蝶呤通过抑制合成嘌呤核苷酸和胸苷酸所需的二氢叶酸还原酶抑制DNA合成、修复和/或复制,而特异性地在S期显示细胞阶段效应。甲氨蝶呤指示作为单一药剂或与其它化疗剂组合用于治疗绒毛膜癌、脑膜白血病、非霍奇金氏淋巴瘤,以及乳腺癌、头癌、颈癌、卵巢癌和膀胱癌。Methotrexate, N-[4[[(2,4-diamino-6-pteridinyl)methyl]methylamino]benzoyl]-L-glutamic acid, is commercially available as methotrexate sodium. Methotrexate exhibits cell stage effects specifically in the S phase by inhibiting DNA synthesis, repair and/or replication by inhibiting dihydrofolate reductase required for the synthesis of purine nucleotides and thymidylate. Methotrexate is indicated as a single agent or in combination with other chemotherapeutic agents for the treatment of choriocarcinoma, meningeal leukemia, non-Hodgkin's lymphoma, and breast, head, neck, ovarian, and bladder cancers.
喜树碱类,包括喜树碱和喜树碱衍生物,可作为拓扑异构酶I抑制剂得到或在开发中。喜树碱类的细胞毒活性据信与其拓扑异构酶I抑制活性有关。喜树碱类的实例包括但不限于下文所述的伊立替康、拓扑替康和7-(4-甲基哌嗪子基-亚甲基)-10,11-亚乙基二氧基-20-喜树碱的各种光学形式。Camptothecins, including camptothecin and camptothecin derivatives, are available or under development as inhibitors of topoisomerase I. The cytotoxic activity of camptothecins is believed to be related to their topoisomerase I inhibitory activity. Examples of camptothecins include, but are not limited to, irinotecan, topotecan, and various optical forms of 7-(4-methylpiperazino-methylene)-10,11-ethylenedioxy-20-camptothecin described below.
盐酸伊立替康,即(4S)-4,11-二乙基-4-羟基-9-[(4-哌啶子基哌啶子基)羰基氧基]-1H-吡喃并[3’,4’,6,7]吲嗪并[1,2-b]喹啉-3,14(4H,12H)-二酮盐酸盐,可作为注射液CAMPTOSAR®商购得到。伊立替康是喜树碱的衍生物,其与其活性代谢物SN-38一起结合到拓扑异构酶I-DNA复合物。据信细胞毒性作为由拓扑异构酶I:DNA:伊立替康或SN-38三元复合物与复制酶的相互作用引起的不可修复的双链断裂的结果而发生。伊立替康指示用于治疗结肠或直肠的转移性癌症。Irinotecan hydrochloride, (4S)-4,11-diethyl-4-hydroxy-9-[(4-piperidinopiperidino)carbonyloxy]-1H-pyrano[3',4',6,7]indolizino[1,2-b]quinoline-3,14(4H,12H)-dione hydrochloride, is commercially available as an injection solution CAMPTOSAR® . Irinotecan is a derivative of camptothecin that binds to the topoisomerase I-DNA complex along with its active metabolite SN-38. Cytotoxicity is believed to occur as a result of irreparable double-strand breaks caused by the interaction of the topoisomerase I:DNA:irinotecan or SN-38 ternary complex with the replicase. Irinotecan is indicated for the treatment of metastatic cancer of the colon or rectum.
盐酸拓扑替康,即(S)-10-[(二甲基氨基)甲基]-4-乙基-4,9-二羟基-1H-吡喃并[3’,4’,6,7]吲嗪并[1,2-b]喹啉-3,14-(4H,12H)-二酮单盐酸盐,可作为注射液HYCAMTIN®商购获得。拓扑替康是喜树碱的衍生物,其结合拓扑异构酶I-DNA复合物并防止由拓扑异构酶I应答于DNA分子的扭转张力而引起的单链断裂的再连接(relegation)。拓扑替康指示用于转移性卵巢癌和小细胞肺癌的二线治疗。Topotecan hydrochloride, (S)-10-[(dimethylamino)methyl]-4-ethyl-4,9-dihydroxy-1H-pyrano[3',4',6,7]indolizino[1,2-b]quinoline-3,14-(4H,12H)-dione monohydrochloride, is commercially available as an injection solution HYCAMTIN® . Topotecan is a derivative of camptothecin that binds to the topoisomerase I-DNA complex and prevents the relegation of single-strand breaks caused by topoisomerase I in response to torsional tension in the DNA molecule. Topotecan is indicated for second-line treatment of metastatic ovarian cancer and small cell lung cancer.
激素和激素类似物是用于治疗癌症的有用化合物,其中在激素和癌症的生长和/或生长缺乏之间存在关系。可用于癌症治疗的激素和激素类似物的实例包括但不限于肾上腺皮质类固醇类,诸如泼尼松和泼尼松龙,其可用于治疗儿童的恶性淋巴瘤和急性白血病;氨鲁米特和其它芳香酶抑制剂,诸如阿那曲唑、来曲唑、伏氯唑和依西美坦,其可用于治疗肾上腺皮质癌和含有雌激素受体的激素依赖性乳腺癌;孕激素,诸如醋酸甲地孕酮,其可用于治疗激素依赖性乳腺癌和子宫内膜癌;雌激素和抗雌激素,诸如氟维司群、氟他胺、尼鲁米特、比卡鲁胺、醋酸环丙孕酮和5α-还原酶诸如非那雄胺和度他雄胺,其用于治疗前列腺癌和良性前列腺肥大;抗雌激素,诸如他莫昔芬、托瑞米芬、雷洛昔芬、屈洛昔芬、吲哚昔芬(iodoxyfene),以及选择性雌激素受体调节剂(SERMS),如美国专利No. 5,681,835、5,877,219和6,207,716中描述的那些,其可用于治疗激素依赖性乳腺癌和其它易受影响的癌症;和促性腺激素释放激素(GnRH)及其类似物,其刺激促黄体生成素(LH)和/或促卵泡激素(FSH)的释放,可用于治疗前列腺癌,例如LHRH激动剂和拮抗剂诸如醋酸戈舍瑞林和亮脯利特。Hormones and hormone analogs are useful compounds for the treatment of cancer where a relationship exists between the hormone and the growth and/or lack of growth of the cancer. Examples of hormones and hormone analogs that can be used for cancer treatment include, but are not limited to, adrenocortical steroids, such as prednisone and prednisolone, which are useful for treating malignant lymphomas and acute leukemias in children; aminoglutethimide and other aromatase inhibitors, such as anastrozole, letrozole, vorozole, and exemestane, which are useful for treating adrenocortical carcinoma and hormone-dependent breast cancer containing estrogen receptors; progestins, such as megestrol acetate, which are useful for treating hormone-dependent breast cancer and endometrial cancer; estrogens and antiestrogens, such as fulvestrant, flutamide, nilutamide, bicalutamide, cyproterone acetate, and 5α-reductase inhibitors such as finasteride and dutasteride, which are useful for treating prostate cancer and benign prostatic hypertrophy; antiestrogens, such as tamoxifen, toremifene, raloxifene, droloxifene, iodoxyfene, and selective estrogen receptor modulators (SERMS), such as those described in U.S. Pat. No. 5,681,835, 5,877,219 and 6,207,716, which are useful for treating hormone-dependent breast cancer and other susceptible cancers; and gonadotropin-releasing hormone (GnRH) and its analogs, which stimulate the release of luteinizing hormone (LH) and/or follicle-stimulating hormone (FSH), which are useful for treating prostate cancer, for example, LHRH agonists and antagonists such as goserelin acetate and leuprolide.
信号转导途径抑制剂是阻断或抑制引起细胞内变化的化学过程的那些抑制剂。如本文所用,这种变化是细胞增殖或分化。可用于本发明的信号转导抑制剂包括下列的抑制剂:受体酪氨酸激酶、非受体酪氨酸激酶、SH2/SH3结构域阻断剂、丝氨酸/苏氨酸激酶、磷脂酰肌醇-3激酶、肌醇信号转导和Ras癌基因。Signal transduction pathway inhibitors are those inhibitors that block or inhibit the chemical processes that cause changes in cells. As used herein, this change is cell proliferation or differentiation. Signal transduction inhibitors that can be used in the present invention include the following inhibitors: receptor tyrosine kinases, non-receptor tyrosine kinases, SH2/SH3 domain blockers, serine/threonine kinases, phosphatidylinositol-3 kinases, inositol signal transduction and Ras oncogenes.
几种蛋白酪氨酸激酶催化参与调节细胞生长的各种蛋白质中特定酪氨酰残基的磷酸化。这样的蛋白酪氨酸激酶可以广义地分类为受体或非受体激酶。Several protein tyrosine kinases catalyze the phosphorylation of specific tyrosyl residues in various proteins involved in regulating cell growth. Such protein tyrosine kinases can be broadly classified as either receptor or non-receptor kinases.
受体酪氨酸激酶是具有胞外配体结合结构域、跨膜结构域和酪氨酸激酶结构域的跨膜蛋白。受体酪氨酸激酶参与细胞生长的调节,并通常称为生长因子受体。许多这些激酶的不适当或不受控制的活化,即异常激酶生长因子受体活性,例如通过过表达或突变,已显示导致不受控制的细胞生长。因此,这样的激酶的异常活性已经与恶性组织生长相联系。因此,这样的激酶的抑制剂可以提供癌症治疗方法。生长因子受体包括例如表皮生长因子受体(EGFr)、血小板衍生生长因子受体(PDGFr)、erbB2、erbB4、ret、血管内皮生长因子受体(VEGFr)、具有免疫球蛋白样和表皮生长因子同源结构域的酪氨酸激酶(TIE-2)、胰岛素生长因子-I(IGFI)受体、巨噬细胞集落刺激因子(cfms)、BTK、ckit、cmet、成纤维细胞生长因子(FGF)受体、Trk受体(TrkA、TrkB和TrkC)、肝配蛋白(eph)受体和RET原癌基因。几种生长受体抑制剂正在开发中,且包括配体拮抗剂、抗体、酪氨酸激酶抑制剂和反义寡核苷酸。生长因子受体和抑制生长因子受体功能的药剂描述于例如Kath,John C.,Exp. Opin. Ther.Patents (2000) 10(6):803-818;Shawver等人,DDT Vol 2,No. 2 1997年2月;和Lofts,F.J.等人,“Growth factor receptors as targets”,New Molecular Targets for CancerChemotherapy,ed. Workman,Paul和Kerr,David,CRC press 1994,London中。Receptor tyrosine kinases are transmembrane proteins with an extracellular ligand binding domain, a transmembrane domain and a tyrosine kinase domain. Receptor tyrosine kinases are involved in the regulation of cell growth and are commonly referred to as growth factor receptors. Inappropriate or uncontrolled activation of many of these kinases, i.e., abnormal kinase growth factor receptor activity, e.g., by overexpression or mutation, has been shown to lead to uncontrolled cell growth. Therefore, abnormal activity of such kinases has been associated with malignant tissue growth. Therefore, inhibitors of such kinases can provide a method for treating cancer. Growth factor receptors include, for example, epidermal growth factor receptor (EGFr), platelet-derived growth factor receptor (PDGFr), erbB2, erbB4, ret, vascular endothelial growth factor receptor (VEGFr), tyrosine kinase with immunoglobulin-like and epidermal growth factor homology domains (TIE-2), insulin growth factor-I (IGFI) receptor, macrophage colony stimulating factor (cfms), BTK, ckit, cmet, fibroblast growth factor (FGF) receptor, Trk receptor (TrkA, TrkB and TrkC), ephrin (eph) receptor and RET proto-oncogene. Several growth receptor inhibitors are under development and include ligand antagonists, antibodies, tyrosine kinase inhibitors and antisense oligonucleotides. Growth factor receptors and agents that inhibit growth factor receptor function are described, for example, in Kath, John C., Exp. Opin. Ther. Patents (2000) 10(6):803-818; Shawver et al., DDT Vol 2, No. 2 February 1997; and Lofts, F.J. et al., "Growth factor receptors as targets", New Molecular Targets for Cancer Chemotherapy, ed. Workman, Paul and Kerr, David, CRC press 1994, London.
不是生长因子受体激酶的酪氨酸激酶被称为非受体酪氨酸激酶。可用于本发明的作为抗癌药物靶标或潜在靶标的非受体酪氨酸激酶包括cSrc、Lck、Fyn、Yes、Jak、cAb1、FAK(粘着斑激酶)、布鲁顿酪氨酸激酶和Bcr-Abl。这样的非受体激酶和抑制非受体酪氨酸激酶功能的药剂描述于Sinh,S.和Corey,S.J.,(1999) Journal of Hematotherapy and StemCell Research 8 (5):465 - 80;和Bolen,J.B.,Brugge,J.S.,(1997) Annual review ofImmunology. 15:371-404中。Tyrosine kinases that are not growth factor receptor kinases are referred to as non-receptor tyrosine kinases. Non-receptor tyrosine kinases that can be used as targets or potential targets of anticancer drugs of the present invention include cSrc, Lck, Fyn, Yes, Jak, cAb1, FAK (focal adhesion kinase), Bruton's tyrosine kinase, and Bcr-Abl. Such non-receptor kinases and agents that inhibit the function of non-receptor tyrosine kinases are described in Sinh, S. and Corey, S.J., (1999) Journal of Hematotherapy and Stem Cell Research 8 (5): 465-80; and Bolen, J.B., Brugge, J.S., (1997) Annual review of Immunology. 15: 371-404.
SH2/SH3结构域阻断剂是破坏各种酶或衔接蛋白中的SH2或SH3结构域结合的药剂,所述酶或衔接蛋白包括PI3-K p85亚单元、Src家族激酶、衔接分子(Shc、Crk、Nck、Grb2)和Ras-GAP。作为抗癌药物靶标的SH2/SH3结构域讨论于Smithgall,T.E. (1995),Journalof Pharmacological and Toxicological Methods. 34(3) 125-32中。SH2/SH3 domain blockers are agents that disrupt SH2 or SH3 domain binding in various enzymes or adaptor proteins, including the PI3-K p85 subunit, Src family kinases, adaptor molecules (Shc, Crk, Nck, Grb2), and Ras-GAP. SH2/SH3 domains as anticancer drug targets are discussed in Smithgall, T.E. (1995), Journal of Pharmacological and Toxicological Methods. 34(3) 125-32.
丝氨酸/苏氨酸激酶抑制剂,包括MAP激酶级联阻断剂,其包括Raf激酶(rafk)、丝裂原或细胞外调节激酶(MEK)和细胞外调节激酶(ERK)的阻断剂;和蛋白激酶C家族成员阻断剂,包括PKC(α、β、γ、ε、μ、λ、ι、ζ)、IkB激酶家族(IKKa、IKKb)、PKB家族激酶、akt激酶家族成员和TGFβ受体激酶的阻断剂。这样的丝氨酸/苏氨酸激酶及其抑制剂描述于Yamamoto,T.,Taya,S.,Kaibuchi,K.,(1999),Journal of Biochemistry, 126 (5) 799-803;Brodt,P,Samani,A.,和Navab,R. (2000),Biochemical Pharmacology,60. 1101-1107;Massague,J.,Weis-Garcia,F. (1996) Cancer Surveys. 27:41-64;Philip,P.A.,和Harris,A.L. (1995),Cancer Treatment and Research. 78:3-27,Lackey,K.等人,Bioorganic and Medicinal Chemistry Letters,(10),2000,223-226;美国专利No. 6,268,391;和Martinez-Iacaci,L.等人,Int. J. Cancer (2000),88(1),44-52中。Serine/threonine kinase inhibitors, including MAP kinase cascade blockers, including blockers of Raf kinase (rafk), mitogen- or extracellular-regulated kinase (MEK) and extracellular-regulated kinase (ERK); and protein kinase C family member blockers, including blockers of PKC (α, β, γ, ε, μ, λ, ι, ζ), IkB kinase family (IKKa, IKKb), PKB family kinases, akt kinase family members and TGFβ receptor kinase. Such serine/threonine kinases and their inhibitors are described in Yamamoto, T., Taya, S., Kaibuchi, K., (1999), Journal of Biochemistry, 126 (5) 799-803; Brodt, P, Samani, A., and Navab, R. (2000), Biochemical Pharmacology, 60. 1101-1107; Massague, J., Weis-Garcia, F. (1996) Cancer Surveys. 27:41-64; Philip, P. A., and Harris, A. L. (1995), Cancer Treatment and Research . 78:3-27, Lackey, K. et al., Bioorganic and Medicinal Chemistry Letters, (10), 2000, 223-226; U.S. Pat. No. 6,268,391; and Martinez-Iacaci, L. et al., Int. J. Cancer (2000), 88(1), 44-52.
磷脂酰肌醇-3激酶家族成员的抑制剂,包括PI3-激酶、ATM、DNA-PK和Ku的阻断剂也可用于本发明。这样的激酶讨论于Abraham,R.T. (1996),Current Opinion inImmunology. 8 (3) 412-8;Canman,C.E.,Lim,D.S. (1998),Oncogene 17 (25) 3301-3308;Jackson,S.P. (1997),International Journal of Biochemistry and CellBiology. 29 (7):935-8;和Zhong,H.等人,Cancer res,(2000) 60(6),1541-1545中。Inhibitors of members of the phosphatidylinositol-3 kinase family, including blockers of PI3-kinase, ATM, DNA-PK and Ku, can also be used in the present invention. Such kinases are discussed in Abraham, RT (1996), Current Opinion in Immunology . 8 (3) 412-8; Canman, CE, Lim, DS (1998), Oncogene 17 (25) 3301-3308; Jackson, SP (1997), International Journal of Biochemistry and Cell Biology. 29 (7): 935-8; and Zhong, H. et al., Cancer res, (2000) 60 (6), 1541-1545.
还可用于本发明的是肌醇信号转导抑制剂,诸如磷脂酶C阻断剂和肌醇类似物。这样的信号抑制剂描述于Powis,G.,和Kozikowski A.,(1994) New Molecular Targets forCancer Chemotherapy ed.,Paul Workman和David Kerr,CRC press 1994,London中。Also useful in the present invention are inositol signaling inhibitors, such as phospholipase C blockers and inositol analogs. Such signaling inhibitors are described in Powis, G., and Kozikowski A., (1994) New Molecular Targets for Cancer Chemotherapy ed., Paul Workman and David Kerr, CRC press 1994, London.
另一组信号转导途径抑制剂是Ras癌基因的抑制剂。这样的抑制剂包括法尼基转移酶、牻牛儿基-牻牛儿基转移酶和CAAX蛋白酶的抑制剂以及反义寡核苷酸、核酶和免疫疗法。这样的抑制剂已经显示阻断含有野生型突变ras的细胞中的ras活化,从而起到抗增殖剂的作用。Ras癌基因抑制讨论于Scharovsky,O.G.,Rozados,V.R.,Gervasoni,S.I.Matar,P. (2000),Journal of Biomedical Science. 7(4) 292-8;Ashby,M.N. (1998),Current Opinion in Lipidology. 9 (2) 99 - 102;和BioChim. Biophys. Acta,(19899) 1423(3):19-30中。Another group of signal transduction pathway inhibitors are inhibitors of Ras oncogenes. Such inhibitors include inhibitors of farnesyltransferase, geranyl-geranyltransferase, and CAAX proteases, as well as antisense oligonucleotides, ribozymes, and immunotherapy. Such inhibitors have been shown to block ras activation in cells containing wild-type mutant ras, thereby acting as antiproliferative agents. Ras oncogene inhibition is discussed in Scharovsky, OG, Rozados, VR, Gervasoni, SI Matar, P. (2000), Journal of Biomedical Science . 7(4) 292-8; Ashby, MN (1998), Current Opinion in Lipidology. 9(2) 99 - 102; and BioChim. Biophys. Acta, (19899) 1423(3): 19-30.
如上所述,受体激酶配体结合的抗体拮抗剂也可以用作信号转导抑制剂。这组信号转导途径抑制剂包括将人源化抗体用于受体酪氨酸激酶的细胞外配体结合结构域。例如Imclone C225 EGFR特异性抗体(参见Green,M.C.等人,Monoclonal Antibody Therapyfor Solid Tumors,Cancer Treat. Rev.,(2000),26(4),269-286);Herceptin® erbB2抗体(参见Tyrosine Kinase Signalling in Breast cancer:erbB Family ReceptorTyrosine Kinases,Breast cancer Res.,2000,2(3),176-183);和2CB VEGFR2特异性抗体(参见Brekken,R.A.等人,Selective Inhibition of VEGFR2 Activity by a monoclonalAnti-VEGF antibody blocks tumor growth in mice,Cancer Res. (2000) 60,5117-5124)。As described above, antibody antagonists of receptor kinase ligand binding can also be used as signal transduction inhibitors. This group of signal transduction pathway inhibitors includes the use of humanized antibodies to the extracellular ligand binding domain of receptor tyrosine kinases. For example, Imclone C225 EGFR-specific antibody (see Green, MC et al., Monoclonal Antibody Therapy for Solid Tumors, Cancer Treat. Rev., (2000), 26(4), 269-286); Herceptin® erbB2 antibody (see Tyrosine Kinase Signalling in Breast cancer:erbB Family Receptor Tyrosine Kinases, Breast cancer Res., 2000, 2(3), 176-183); and 2CB VEGFR2-specific antibody (see Brekken, RA et al., Selective Inhibition of VEGFR2 Activity by a monoclonal Anti-VEGF antibody blocks tumor growth in mice, Cancer Res. (2000) 60, 5117-5124).
包括非受体MEK血管生成抑制剂的抗血管生成治疗剂也可能是有用的。抗血管生成剂诸如抑制血管内皮生长因子的作用的那些(例如抗血管内皮细胞生长因子抗体贝伐单抗[Avastin™],以及通过其它机制起作用的化合物(例如利诺胺、整联蛋白αvβ3功能抑制剂、内皮他丁和血管他丁)。Anti-angiogenic therapeutics including non-receptor MEK angiogenesis inhibitors may also be useful. Anti-angiogenic agents such as those that inhibit the action of vascular endothelial growth factor (e.g., the anti-vascular endothelial cell growth factor antibody bevacizumab [Avastin™], and compounds that act through other mechanisms (e.g., linomide, inhibitors of integrin αvβ3 function, endostatin and angiostatin).
用于免疫治疗方案的药剂也可以与式(I)化合物组合使用。免疫治疗方法,包括例如增加患者肿瘤细胞的免疫原性的离体和体内方法,诸如用细胞因子诸如白介素2、白介素4或粒细胞-巨噬细胞集落刺激因子转染,降低T细胞能量的方法,使用转染的免疫细胞诸如细胞因子转染的树突状细胞的方法,使用细胞因子转染的肿瘤细胞系的方法和使用抗独特型抗体的方法。Agents for immunotherapy regimens may also be used in combination with compounds of formula (I). Immunotherapeutic methods include, for example, ex vivo and in vivo methods for increasing the immunogenicity of patient tumor cells, such as transfection with cytokines such as interleukin 2, interleukin 4, or granulocyte-macrophage colony stimulating factor, methods for reducing T cell energy, methods using transfected immune cells such as cytokine-transfected dendritic cells, methods using cytokine-transfected tumor cell lines, and methods using anti-idiotypic antibodies.
用于促凋亡方案的治疗剂(例如,bcl-2反义寡核苷酸)也可用于本发明的组合中。Therapeutic agents used in pro-apoptotic regimens (eg, bcl-2 antisense oligonucleotides) may also be used in the combinations of the invention.
细胞周期信号转导抑制剂抑制参与细胞周期控制的分子。称为细胞周期蛋白依赖性激酶(CDK)的蛋白激酶家族及其与称为细胞周期蛋白的蛋白质家族的相互作用控制通过真核细胞周期的进展。不同细胞周期蛋白/CDK复合物的配位活化和失活对于通过细胞周期的正常进展是必需的。几种细胞周期信号转导的抑制剂正在开发中。例如,细胞周期蛋白依赖性激酶,包括CDK2、CDK4和CDK6及其抑制剂的实例描述于例如Rosania等人,Exp. Opin.Ther. Patents (2000) 10(2):215-230中。Cell cycle signal transduction inhibitors inhibit molecules involved in cell cycle control. A family of protein kinases called cyclin-dependent kinases (CDKs) and their interaction with a family of proteins called cyclins control the progression through the eukaryotic cell cycle. Coordinated activation and inactivation of different cyclin/CDK complexes are essential for normal progression through the cell cycle. Several inhibitors of cell cycle signal transduction are under development. For example, examples of cyclin-dependent kinases, including CDK2, CDK4 and CDK6 and their inhibitors are described in, for example, Rosania et al., Exp. Opin. Ther. Patents (2000) 10 (2): 215-230.
在一个实施方案中,本发明的组合产品(combination)包含式(I)化合物或其盐(特别是其药学上可接受的盐)和至少一种抗肿瘤剂,所述抗肿瘤剂选自抗微管剂、铂配位络合物、烷基化试剂、抗菌剂、拓扑异构酶II抑制剂、抗代谢物、拓扑异构酶I抑制剂、激素和激素类似物、信号转导途径抑制剂、非受体酪氨酸MEK血管生成抑制剂、免疫治疗剂、促凋亡剂和细胞周期信号转导抑制剂。In one embodiment, the combination of the present invention comprises a compound of formula (I) or a salt thereof (particularly a pharmaceutically acceptable salt thereof) and at least one anti-tumor agent selected from anti-microtubule agents, platinum coordination complexes, alkylating agents, antibacterial agents, topoisomerase II inhibitors, antimetabolites, topoisomerase I inhibitors, hormones and hormone analogs, signal transduction pathway inhibitors, non-receptor tyrosine MEK angiogenesis inhibitors, immunotherapeutic agents, pro-apoptotic agents and cell cycle signal transduction inhibitors.
在一个实施方案中,本发明的组合产品包含式(I)化合物或其盐(特别是其药学上可接受的盐)和至少一种抗肿瘤剂,所述抗肿瘤剂是选自二萜类化合物和长春花生物碱的抗微管剂。In one embodiment, the combination product of the present invention comprises a compound of formula (I) or a salt thereof (particularly a pharmaceutically acceptable salt thereof) and at least one anti-tumor agent which is an anti-microtubule agent selected from diterpenoids and vinca alkaloids.
在一个进一步实施方案中,至少一种抗肿瘤剂是二萜类化合物。在一个进一步实施方案中,至少一种抗肿瘤剂是长春花生物碱。In a further embodiment, at least one anti-neoplastic agent is a diterpenoid.In a further embodiment, at least one anti-neoplastic agent is a vinca alkaloid.
在一个实施方案中,本发明的组合产品包含式(I)化合物或其盐(特别是其药学上可接受的盐)和至少一种抗肿瘤剂,所述抗肿瘤剂是铂配位络合物。In one embodiment, the combination product of the present invention comprises a compound of formula (I) or a salt thereof (particularly a pharmaceutically acceptable salt thereof) and at least one anti-tumor agent which is a platinum coordination complex.
在一个进一步实施方案中,至少一种抗肿瘤剂是紫杉醇、卡铂或长春瑞滨。在一个进一步实施方案中,至少一种抗肿瘤剂是卡铂。在一个进一步实施方案中,至少一种抗肿瘤剂是长春瑞滨。在一个进一步实施方案中,至少一种抗肿瘤剂是紫杉醇。在一个实施方案中,本发明的组合产品包含式(I)化合物或盐,特别是其药学上可接受的盐和至少一种抗肿瘤剂,所述抗肿瘤剂是信号转导途径抑制剂。In a further embodiment, at least one antineoplastic agent is paclitaxel, carboplatin or vinorelbine. In a further embodiment, at least one antineoplastic agent is carboplatin. In a further embodiment, at least one antineoplastic agent is vinorelbine. In a further embodiment, at least one antineoplastic agent is paclitaxel. In one embodiment, the combination product of the present invention comprises a compound of formula (I) or a salt, in particular a pharmaceutically acceptable salt thereof, and at least one antineoplastic agent, which is a signal transduction pathway inhibitor.
在一个进一步实施方案中,信号转导途径抑制剂是生长因子受体激酶VEGFR2、TIE2、PDGFR、BTK、erbB2、EGFr、IGFR-1、TrkA、TrkB、TrkC或c-fms的抑制剂。在一个进一步实施方案中,信号转导途径抑制剂是丝氨酸/苏氨酸激酶rafk、akt或PKC-ζ的抑制剂。在一个进一步实施方案中,信号转导途径抑制剂是选自src家族的激酶的非受体酪氨酸激酶的抑制剂。在一个进一步实施方案中,信号转导途径抑制剂是c-src的抑制剂。在一个进一步实施方案中,信号转导途径抑制剂是Ras癌基因的抑制剂,其选自法尼基转移酶和牻牛儿基牻牛儿基转移酶的抑制剂。在一个进一步实施方案中,信号转导途径抑制剂是选自PI3K的丝氨酸/苏氨酸激酶的抑制剂。In a further embodiment, the signal transduction pathway inhibitor is an inhibitor of growth factor receptor kinases VEGFR2, TIE2, PDGFR, BTK, erbB2, EGFr, IGFR-1, TrkA, TrkB, TrkC or c-fms. In a further embodiment, the signal transduction pathway inhibitor is an inhibitor of serine/threonine kinases rafk, akt or PKC-ζ. In a further embodiment, the signal transduction pathway inhibitor is an inhibitor of non-receptor tyrosine kinases selected from kinases of the src family. In a further embodiment, the signal transduction pathway inhibitor is an inhibitor of c-src. In a further embodiment, the signal transduction pathway inhibitor is an inhibitor of Ras oncogenes selected from inhibitors of farnesyl transferase and geranylgeranyl transferase. In a further embodiment, the signal transduction pathway inhibitor is an inhibitor of serine/threonine kinases selected from PI3K.
在一个进一步实施方案中,信号转导途径抑制剂是双重EGFr/erbB2抑制剂,例如N-{3-氯-4-[(3-氟苄基)氧基]苯基}-6-[5-({[2-甲磺酰基)乙基]氨基}甲基)-2-呋喃基]-4-喹唑啉胺。In a further embodiment, the signal transduction pathway inhibitor is a dual EGFr/erbB2 inhibitor, such as N-{3-chloro-4-[(3-fluorobenzyl)oxy]phenyl}-6-[5-({[2-methylsulfonyl)ethyl]amino}methyl)-2-furyl]-4-quinazolinamine.
在一个实施方案中,本发明的组合产品包含式(I)化合物或其盐(特别是其药学上可接受的盐)和至少一种抗肿瘤剂,所述抗肿瘤剂是细胞周期信号转导抑制剂。在其它实施方案中,细胞周期信号转导抑制剂是CDK2、CDK4或CDK6的抑制剂。In one embodiment, the combination product of the present invention comprises a compound of formula (I) or a salt thereof (particularly a pharmaceutically acceptable salt thereof) and at least one anti-tumor agent, wherein the anti-tumor agent is a cell cycle signal transduction inhibitor. In other embodiments, the cell cycle signal transduction inhibitor is an inhibitor of CDK2, CDK4 or CDK6.
用于与式(I)化合物组合或共同施用的其它治疗剂(例如,抗肿瘤剂)的额外实例是免疫调节剂。Additional examples of other therapeutic agents (eg, anti-neoplastic agents) for combination or co-administration with the compounds of Formula (I) are immunomodulators.
如本文所用,“免疫调节剂”指影响免疫系统的任何物质,包括单克隆抗体。免疫调节剂可用作抗肿瘤剂用于治疗癌症。例如,免疫调节剂包括但不限于抗CTLA-4抗体,诸如伊匹木单抗(YERVOY)和抗PD-1抗体(Opdivo/纳武单抗和Keytruda/派姆单抗)。其它免疫调节剂包括,但不限于,ICOS抗体、OX-40抗体、PD-L1抗体、LAG3抗体、TIM-3抗体、41BB抗体和GITR抗体。As used herein, "immunomodulator" refers to any substance that affects the immune system, including monoclonal antibodies. Immunomodulators can be used as anti-tumor agents for the treatment of cancer. For example, immunomodulators include, but are not limited to, anti-CTLA-4 antibodies, such as ipilimumab (YERVOY) and anti-PD-1 antibodies (Opdivo/nivolumab and Keytruda/pembrolizumab). Other immunomodulators include, but are not limited to, ICOS antibodies, OX-40 antibodies, PD-L1 antibodies, LAG3 antibodies, TIM-3 antibodies, 41BB antibodies, and GITR antibodies.
用于与本发明化合物组合或共同施用的其它治疗剂(抗肿瘤剂)的额外实例是抗-PD-L1剂。抗-PD-L1抗体及其制备方法是本领域已知的。这样的针对PD-L1的抗体可以是多克隆或单克隆的、和/或重组和/或人源化的。示例性PD-L1抗体公开于美国专利No. 8,217,149、8,383,796、8,552,154、9,212,224和8,779,108和美国专利申请公开No.20110280877、 2014/0341902和20130045201中。额外示例性的针对PD-L1(也称为CD274或B7-H1)的抗体和使用方法公开于美国专利No. 7,943,743、8,168,179;和7,595,048,WO2014055897、WO2016007235和美国专利申请公开No. 20130034559、20130034559和20150274835中。正在开发作为治疗癌症的免疫调节药剂或免疫调节剂的PD-L1抗体。Additional examples of other therapeutic agents (anti-tumor agents) for combination or co-administration with the compounds of the present invention are anti-PD-L1 agents. Anti-PD-L1 antibodies and methods for their preparation are known in the art. Such antibodies against PD-L1 may be polyclonal or monoclonal, and/or recombinant and/or humanized. Exemplary PD-L1 antibodies are disclosed in U.S. Patent Nos. 8,217,149, 8,383,796, 8,552,154, 9,212,224, and 8,779,108 and U.S. Patent Application Publication Nos. 20110280877, 2014/0341902, and 20130045201. Additional exemplary antibodies to PD-L1 (also known as CD274 or B7-H1) and methods of use are disclosed in U.S. Patent Nos. 7,943,743, 8,168,179; and 7,595,048, WO2014055897, WO2016007235, and U.S. Patent Application Publication Nos. 20130034559, 20130034559, and 20150274835. PD-L1 antibodies are being developed as immunomodulatory agents or immunomodulators for the treatment of cancer.
在一个实施方案中,针对PD-L1的抗体是美国专利No. 8,217,149中公开的抗体。在另一个实施方案中,抗-PD-L1抗体包含美国专利No. 8,217,149中公开的抗体的CDR。在另一个实施方案中,针对PD-L1的抗体是美国专利No. 8,779,108中公开的抗体。在另一个实施方案中,抗-PD-L1抗体包含美国申请No. 8,779,108中公开的抗体的CDR。在另一个实施方案中,针对PD-L1的抗体是美国专利申请公开No. 20130045201中公开的抗体。在另一个实施方案中,抗-PD-L1抗体包含美国专利申请公开No. 20130045201中公开的抗体的CDR。在一个实施方案中,抗-PD-L1抗体是WO 2007/005874中描述的BMS-936559 (MDX-1105)。在另一个实施方案中,抗-PD-L1抗体是MPDL3280A (RG7446)。在另一个实施方案中,抗-PD-L1抗体是MEDI4736,其为WO 2011/066389和US 2013/034559中描述的抗-PD-L1单克隆抗体。在另一个实施方案中,抗-PD-L1抗体是TECENTRIQ™ (阿特珠单抗),其为在2016年5月在美国被批准用于特定类型的膀胱癌的抗-PDL1癌症免疫疗法。在另一个实施方案中,抗-PD-L1抗体是YW243.55.S70,其为WO 2010/077634和美国专利No. 8,217,149中描述的抗PD-L1。可用于本发明的方法的抗-PD-L1抗体的实例及其制备方法描述于PCT专利申请WO2010/077634、WO 2007/005874、WO 2011/066389、美国专利No. 8,217,149和US 2013/034559。In one embodiment, the antibody to PD-L1 is an antibody disclosed in U.S. Patent No. 8,217,149. In another embodiment, the anti-PD-L1 antibody comprises the CDRs of the antibody disclosed in U.S. Patent No. 8,217,149. In another embodiment, the antibody to PD-L1 is an antibody disclosed in U.S. Patent No. 8,779,108. In another embodiment, the anti-PD-L1 antibody comprises the CDRs of the antibody disclosed in U.S. Application No. 8,779,108. In another embodiment, the antibody to PD-L1 is an antibody disclosed in U.S. Patent Application Publication No. 20130045201. In another embodiment, the anti-PD-L1 antibody comprises the CDRs of the antibody disclosed in U.S. Patent Application Publication No. 20130045201. In one embodiment, the anti-PD-L1 antibody is BMS-936559 (MDX-1105) described in WO 2007/005874. In another embodiment, the anti-PD-L1 antibody is MPDL3280A (RG7446). In another embodiment, the anti-PD-L1 antibody is MEDI4736, which is an anti-PD-L1 monoclonal antibody described in WO 2011/066389 and US 2013/034559. In another embodiment, the anti-PD-L1 antibody is TECENTRIQ™ (atezolizumab), which is an anti-PDL1 cancer immunotherapy approved in the United States in May 2016 for a specific type of bladder cancer. In another embodiment, the anti-PD-L1 antibody is YW243.55.S70, which is an anti-PD-L1 described in WO 2010/077634 and U.S. Patent No. 8,217,149. Examples of anti-PD-L1 antibodies useful in the methods of the invention and methods for their preparation are described in PCT patent applications WO 2010/077634, WO 2007/005874, WO 2011/066389, U.S. Pat. No. 8,217,149, and US 2013/034559.
结合人PD-L1并且可用于本发明的治疗方法、药物和用途中的mAb的其它实例描述于WO2013/019906、W02010/077634A1和US8383796中。在本发明的治疗方法、药物和用途中可用作PD-1拮抗剂的具体抗-人PD-L1 mAb包括MPDL3280A、BMS-936559、MEDI4736、MSB0010718C。Other examples of mAbs that bind to human PD-L1 and can be used in the methods of treatment, medicines and uses of the present invention are described in WO2013/019906, WO2010/077634A1 and US8383796. Specific anti-human PD-L1 mAbs that can be used as PD-1 antagonists in the methods of treatment, medicines and uses of the present invention include MPDL3280A, BMS-936559, MEDI4736, MSB0010718C.
用于与本发明化合物组合或共同施用的其它治疗剂(抗肿瘤剂)的额外实例是PD-1拮抗剂。An additional example of other therapeutic agents (anti-tumor agents) for combination or co-administration with the compounds of the invention is a PD-1 antagonist.
“PD-1拮抗剂”意指阻断在癌细胞上表达的PD-L1结合在免疫细胞(T细胞、B细胞或NKT细胞)上表达的PD-1并且优选还阻断在癌细胞上表达的PD-L2结合免疫细胞表达的PD-1的任何化学化合物或生物分子。PD-1及其配体的替代名称或同义词包括:用于PD-1的PDCD1、PD1、CD279和SLEB2;用于PD-L1的PDCD1L1、PDL1、B7H1、B7-4、CD274和B7-H;和用于PD-L2的PDCD1L2、PDL2、B7-DC、Btdc和CD273。在其中待治疗人个体的本发明的方面或实施方案的任何实施方案中,PD-1拮抗剂阻断人PD-L1与人PD-1的结合,并且优选阻断人PD-L1和PD-L2两者与人PD-1的结合。人PD-1氨基酸序列可以在NCBI Locus No.:NP_005009中找到。人PD-L1和PD-L2氨基酸序列分别可以在NCBI Locus No.:NP_054862和NP_079515中找到。"PD-1 antagonist" means any chemical compound or biological molecule that blocks PD-L1 expressed on cancer cells from binding to PD-1 expressed on immune cells (T cells, B cells or NKT cells) and preferably also blocks PD-L2 expressed on cancer cells from binding to PD-1 expressed by immune cells. Alternative names or synonyms for PD-1 and its ligands include: PDCD1, PD1, CD279 and SLEB2 for PD-1; PDCD1L1, PDL1, B7H1, B7-4, CD274 and B7-H for PD-L1; and PDCD1L2, PDL2, B7-DC, Btdc and CD273 for PD-L2. In any embodiment of the aspects or embodiments of the present invention in which a human individual is to be treated, the PD-1 antagonist blocks the binding of human PD-L1 to human PD-1, and preferably blocks the binding of both human PD-L1 and PD-L2 to human PD-1. The human PD-1 amino acid sequence can be found in NCBI Locus No.: NP_005009. The human PD-L1 and PD-L2 amino acid sequences can be found in NCBI Locus No.: NP_054862 and NP_079515, respectively.
可用于本发明的任何方面的PD-1拮抗剂包括特异性结合PD-1或PD-L1并且优选特异性结合人PD-1或人PD-L1的单克隆抗体(mAb)或其抗原结合片段。所述mAb可以是人抗体、人源化抗体或嵌合抗体,并且可以包括人恒定区。在一些实施方案中,人恒定区选自IgG1、IgG2、IgG3和IgG4恒定区,并且在优选实施方案中,人恒定区是IgG1或IgG4恒定区。在一些实施方案中,抗原结合片段选自Fab、Fab'-SH、F(ab')2、scFv和Fv片段。PD-1 antagonists that can be used in any aspect of the present invention include monoclonal antibodies (mAbs) or antigen-binding fragments thereof that specifically bind to PD-1 or PD-L1 and preferably specifically bind to human PD-1 or human PD-L1. The mAb can be a human antibody, a humanized antibody, or a chimeric antibody, and can include a human constant region. In some embodiments, the human constant region is selected from IgG1, IgG2, IgG3, and IgG4 constant regions, and in preferred embodiments, the human constant region is an IgG1 or IgG4 constant region. In some embodiments, the antigen-binding fragment is selected from Fab, Fab'-SH, F(ab')2, scFv, and Fv fragments.
结合人PD-1并且可用于本发明的各个方面和实施方案中的mAb的实例描述于US7488802、US7521051、US8008449、US8354509、US8168757、WO2004/004771、WO2004/072286、WO2004/056875和US2011/0271358中。Examples of mAbs that bind to human PD-1 and can be used in various aspects and embodiments of the invention are described in US7488802, US7521051, US8008449, US8354509, US8168757, WO2004/004771, WO2004/072286, WO2004/056875 and US2011/0271358.
在本发明的任何方面和实施方案中可用作PD-1拮抗剂的具体抗人PD-1mAb包括:MK-3475,一种人源化IgG4mAb,具有WHO Drug Information, Vol. 27, No. 2, 第161-162页 (2013)中描述的结构,并且其包含图6中所示的重链和轻链氨基酸序列;纳武单抗,一种人IgG4 mAb,具有WHO Drug Information, Vol. 27, No. 1, 第68-69页 (2013)中描述的结构,并且其包含图7中所示的重链和轻链氨基酸序列;人源化抗体h409A11、h409A16和h409A17(其描述于WO2008/156712中)以及AMP-514(其正在由Medimmune开发)。Specific anti-human PD-1 mAbs that can be used as PD-1 antagonists in any aspects and embodiments of the invention include: MK-3475, a humanized IgG4 mAb having a structure described in WHO Drug Information , Vol. 27, No. 2, pages 161-162 (2013), and which comprises the heavy chain and light chain amino acid sequences shown in Figure 6; nivolumab, a human IgG4 mAb having a structure described in WHO Drug Information , Vol. 27, No. 1, pages 68-69 (2013), and which comprises the heavy chain and light chain amino acid sequences shown in Figure 7; humanized antibodies h409A11, h409A16 and h409A17 (which are described in WO2008/156712) and AMP-514 (which is being developed by Medimmune).
可用于本发明的任何方面和实施方案中的其它PD-1拮抗剂包括特异性结合PD-1并且优选特异性结合人PD-1的免疫粘附素,例如含有融合至免疫球蛋白分子的恒定区(诸如Fc区)的PD-L1或PD-L2的胞外或PD-1结合部分的融合蛋白。特异性结合PD-1的免疫粘附分子的实例描述于WO2010/027827和WO2011/066342中。在本发明的治疗方法、药物和用途中可用作PD-1拮抗剂的具体融合蛋白包括AMP-224(也称为B7-DCIg),其为PD-L2-FC融合蛋白并结合人PD-1。Other PD-1 antagonists that can be used in any aspect and embodiment of the present invention include immunoadhesins that specifically bind to PD-1 and preferably specifically bind to human PD-1, such as fusion proteins containing an extracellular or PD-1 binding portion of PD-L1 or PD-L2 fused to a constant region (such as an Fc region) of an immunoglobulin molecule. Examples of immunoadhesion molecules that specifically bind to PD-1 are described in WO2010/027827 and WO2011/066342. Specific fusion proteins that can be used as PD-1 antagonists in the treatment methods, drugs and uses of the present invention include AMP-224 (also known as B7-DCIg), which is a PD-L2-FC fusion protein and binds to human PD-1.
KEYTRUDA/派姆单抗是由Merck销售用于治疗肺癌的抗PD-1抗体。派姆单抗的氨基酸序列和使用方法公开在美国专利No. 8,168,757中。KEYTRUDA/pembrolizumab is an anti-PD-1 antibody marketed by Merck for the treatment of lung cancer. The amino acid sequence and methods of use of pembrolizumab are disclosed in U.S. Patent No. 8,168,757.
Opdivo/纳武单抗是由Bristol Myers Squibb销售的针对负免疫调节性人细胞表面受体PD-1(程序性死亡-1或程序性细胞死亡-1/PCD-1)的完全人单克隆抗体,其具有免疫增强活性。纳武单抗通过其配体PD-L1和PD-L2结合并阻断PD-1(Ig超家族跨膜蛋白)的活化,导致T细胞的活化和针对肿瘤细胞或病原体的细胞介导的免疫应答。活化的PD-1通过抑制P13k/Akt途径活化来负调节T细胞活化和效应子功能。纳武单抗的其它名称包括:BMS-936558、MDX-1106和ONO-4538。纳武单抗的氨基酸序列以及使用和制备方法公开在美国专利No. US 8,008,449中。Opdivo/Nivolumab is a fully human monoclonal antibody against the negative immunoregulatory human cell surface receptor PD-1 (programmed death-1 or programmed cell death-1/PCD-1) sold by Bristol Myers Squibb, which has immunoenhancing activity. Nivolumab binds to and blocks the activation of PD-1 (Ig superfamily transmembrane protein) through its ligands PD-L1 and PD-L2, leading to the activation of T cells and cell-mediated immune responses against tumor cells or pathogens. Activated PD-1 negatively regulates T cell activation and effector function by inhibiting P13k/Akt pathway activation. Other names for nivolumab include: BMS-936558, MDX-1106 and ONO-4538. The amino acid sequence of nivolumab and methods of use and preparation are disclosed in U.S. Patent No. US 8,008,449.
用于与式(I)化合物组合或共同施用的其它治疗剂(抗肿瘤剂)的额外实例是针对ICOS的抗体。An additional example of other therapeutic agents (anti-tumor agents) for combination or co-administration with the compounds of formula (I) is an antibody directed against ICOS.
ICOS是一种共刺激性T细胞受体,其与CD28/CTLA-4-Ig超家族具有结构和功能关系(Hutloff, 等人, "ICOS is an inducible T-cell co-stimulator structurally andfunctionally related to CD28", Nature, 397: 263-266 (1999))。ICOS的活化通过ICOS-L (B7RP-1/B7-H2)的结合发生。B7-1和B7-2(CD28和CTLA4的配体)均不结合或活化ICOS。然而,已显示ICOS-L微弱结合CD28和CTLA-4两者(Yao S等人, “B7-H2 is acostimulatory ligand for CD28 in human”, Immunity, 34(5); 729-40 (2011))。ICOS的表达似乎限于T细胞。ICOS表达水平在不同T细胞亚群和T细胞活化状态之间变化。ICOS表达已显示在静息TH17、T滤泡辅助细胞(TFH)和调节性T(Treg)细胞上;然而,与CD28不同;其在初始TH1和TH2效应T细胞群上没有高表达(Paulos CM等人, “The induciblecostimulator (ICOS) is critical for the development of human Th17 cells”, SciTransl Med, 2(55); 55ra78 (2010))。通过TCR参与活化后,在CD4+和CD8+效应T细胞上高度诱导ICOS表达(Wakamatsu E,等人, “Convergent and divergent effects ofcostimulatory molecules in conventional and regulatory CD4+ T cells”, ProcNatal Acad Sci USA, 110(3); 1023-8 (2013))。ICOS is a co-stimulatory T cell receptor that has a structural and functional relationship with the CD28/CTLA-4-Ig superfamily (Hutloff, et al., "ICOS is an inducible T-cell co-stimulator structurally and functionally related to CD28", Nature, 397: 263-266 (1999)). ICOS activation occurs through the binding of ICOS-L (B7RP-1/B7-H2). Neither B7-1 nor B7-2 (ligands for CD28 and CTLA4) binds or activates ICOS. However, ICOS-L has been shown to weakly bind both CD28 and CTLA-4 (Yao S et al., "B7-H2 is a costimulatory ligand for CD28 in human", Immunity, 34(5); 729-40 (2011)). The expression of ICOS appears to be limited to T cells. ICOS expression levels vary between different T cell subsets and T cell activation states. ICOS expression has been shown on resting TH17, T follicular helper cells (TFH) and regulatory T (Treg) cells; however, unlike CD28; it is not highly expressed on naive TH1 and TH2 effector T cell populations (Paulos CM et al., "The inducible costimulator (ICOS) is critical for the development of human Th17 cells", Sci Transl Med, 2(55); 55ra78 (2010)). After activation by TCR engagement, ICOS expression is highly induced on CD4+ and CD8+ effector T cells (Wakamatsu E, et al., "Convergent and divergent effects of costimulatory molecules in conventional and regulatory CD4+ T cells", Proc Natal Acad Sci USA, 110(3); 1023-8 (2013)).
具有激动剂活性的针对人ICOS的鼠抗体的CDR显示于PCT/EP2012/055735 (WO2012/131004)中。针对ICOS的抗体也公开于WO 2008/137915、WO 2010/056804、EP1374902、EP 1374901和EP 1125585中。The CDRs of murine antibodies against human ICOS with agonist activity are shown in PCT/EP2012/055735 (WO2012/131004). Antibodies against ICOS are also disclosed in WO 2008/137915, WO 2010/056804, EP1374902, EP 1374901 and EP 1125585.
针对ICOS的激动剂抗体或ICOS结合蛋白公开于WO2012/13004、WO 2014/033327、WO2016/120789、US20160215059和US20160304610中。在一个实施方案中,针对ICOS的激动剂抗体包括ICOS结合蛋白或其抗原结合部分,其包含以下中的一种或多种:如SEQ ID NO:1中所示的CDRH1;如SEQ ID NO:2中所示的CDRH2;如SEQ ID NO:3中所示的CDRH3;如SEQ IDNO:4中所示的CDRL1;如SEQ ID NO:5中所示的CDRL2和/或如SEQ ID NO:6中所示的CDRL3或每个CDR的直接等同物,其中直接等同物在所述CDR中具有不超过两个氨基酸取代,如WO2016/120789 (其以其整体通过引用并入本文)中所公开。在一个实施方案中,ICOS结合蛋白或其抗原结合部分是针对ICOS的激动剂抗体,其含有VH结构域(包含与SEQ ID NO:7中所示的氨基酸序列具有至少90%同一性的氨基酸序列)和/或VL结构域(包含与如SEQ IDNO:8中所示的氨基酸序列具有至少90%同一性的氨基酸序列),如WO2016/120789中所述,其中所述ICOS结合蛋白特异性结合人ICOS。在一个实施方案中, ICOS结合蛋白是针对ICOS的激动剂抗体,其含有包含SEQ ID NO:7中所示的氨基酸序列的VH结构域和包含SEQ IDNO:8中所示的氨基酸序列的VL结构域,如WO2016/120789中所述。Agonist antibodies or ICOS binding proteins to ICOS are disclosed in WO2012/13004, WO 2014/033327, WO2016/120789, US20160215059 and US20160304610. In one embodiment, an agonist antibody to ICOS comprises an ICOS binding protein, or an antigen binding portion thereof, comprising one or more of: a CDRH1 as shown in SEQ ID NO: 1; a CDRH2 as shown in SEQ ID NO: 2; a CDRH3 as shown in SEQ ID NO: 3; a CDRL1 as shown in SEQ ID NO: 4; a CDRL2 as shown in SEQ ID NO: 5 and/or a CDRL3 as shown in SEQ ID NO: 6, or a direct equivalent of each CDR, wherein the direct equivalent has no more than two amino acid substitutions in the CDRs, as disclosed in WO2016/120789 (which is incorporated herein by reference in its entirety). In one embodiment, the ICOS binding protein, or an antigen binding portion thereof, is an agonist antibody to ICOS, comprising a VH domain (comprising an amino acid sequence having at least 90% identity to the amino acid sequence shown in SEQ ID NO:7) and/or a VL domain (comprising an amino acid sequence having at least 90% identity to the amino acid sequence shown in SEQ ID NO:8), as described in WO2016/120789, wherein the ICOS binding protein specifically binds to human ICOS. In one embodiment, the ICOS binding protein is an agonist antibody to ICOS, comprising a VH domain comprising the amino acid sequence shown in SEQ ID NO:7 and a VL domain comprising the amino acid sequence shown in SEQ ID NO:8, as described in WO2016/120789.
Yervoy(伊匹木单抗)是由Bristol Myers Squibb销售的全人CTLA-4抗体。伊匹木单抗的蛋白结构和使用的方法描述于美国专利No. 6,984,720和7,605,238中。Yervoy (ipilimumab) is a fully human CTLA-4 antibody marketed by Bristol Myers Squibb. The protein structure and methods of use of ipilimumab are described in U.S. Patent Nos. 6,984,720 and 7,605,238.
CD134,也称为OX40,与CD28不同,其是不在静息的初始T细胞上组成性表达的受体的TNFR-超家族的成员。OX40是二级共刺激分子,其在活化后24至72小时后表达;其配体OX40L也不在静息的抗原呈递细胞上表达,但在其活化之后表达。OX40的表达依赖于T细胞的完全活化;没有CD28,OX40的表达被延迟并且水平降至1/4。OX-40抗体、OX-40融合蛋白及其使用方法公开于美国专利No.:US 7,504,101;US 7,758,852;US 7,858,765;US 7,550,140;US 7,960,515;WO2012027328;WO2013028231。CD134, also known as OX40, is a member of the TNFR-superfamily of receptors that are not constitutively expressed on resting naive T cells, unlike CD28. OX40 is a secondary co-stimulatory molecule that is expressed 24 to 72 hours after activation; its ligand OX40L is also not expressed on resting antigen-presenting cells, but is expressed after their activation. The expression of OX40 depends on the full activation of T cells; without CD28, the expression of OX40 is delayed and the level is reduced to 1/4. OX-40 antibodies, OX-40 fusion proteins and methods of use thereof are disclosed in U.S. Patent Nos.: US 7,504,101; US 7,758,852; US 7,858,765; US 7,550,140; US 7,960,515; WO2012027328; WO2013028231.
在一个实施方案中,OX40抗原结合蛋白是WO2012/027328 (PCT/US2011/048752)(国际申请日2011年8月23日)中公开的一种。在另一个实施方案中,抗原结合蛋白包含WO2012/027328 (PCT/US2011/048752)(国际申请日2011年8月23日)中公开的抗体的CDR,或与公开的CDR序列具有90%同一性的CDR。在一个进一步实施方案中,抗原结合蛋白包含WO2012/027328 (PCT/US2011/048752)(国际申请日2011年8月23日)中公开的抗体的VH、VL或两者,或与公开的VH或VL序列具有90%同一性的VH或VL。In one embodiment, the OX40 antigen binding protein is one disclosed in WO2012/027328 (PCT/US2011/048752) (International Application Date August 23, 2011). In another embodiment, the antigen binding protein comprises the CDRs of an antibody disclosed in WO2012/027328 (PCT/US2011/048752) (International Application Date August 23, 2011), or CDRs with 90% identity to the disclosed CDR sequences. In a further embodiment, the antigen binding protein comprises VH, VL, or both of an antibody disclosed in WO2012/027328 (PCT/US2011/048752) (International Application Date August 23, 2011), or VH or VL with 90% identity to the disclosed VH or VL sequences.
在另一个实施方案中,OX40抗原结合蛋白公开于WO2013/028231 (PCT/US2012/024570)(国际申请日2012年2月9日,其以其整体通过引用并入本文)。在另一个实施方案中,抗原结合蛋白包含WO2013/028231 (PCT/US2012/024570)(国际申请日2012年2月9日)中公开的抗体的CDR,或与公开的CDR序列具有90%同一性的CDR。在一个进一步实施方案中,抗原结合蛋白包含WO2013/028231 (PCT/US2012/024570)(国际申请日2012年2月9日)中公开的抗体的VH、VL或两者,或与公开的VH或VL序列具有90%同一性的VH或VL。在一个实施方案中,OX40抗原结合蛋白是分离的针对OX40的激动剂抗体,其包含具有与如WO2013/028231中所示的SEQ ID NO:10的氨基酸序列具有至少90%同一性的序列的轻链可变区和具有与如WO2013/028231中所示的SEQ ID NO:4的氨基酸序列具有至少90%同一性的序列的重链可变区。在一个实施方案中,OX40抗原结合蛋白是分离的抗体,其含有包含如WO2013/028231中所示的SEQ ID NO:10的氨基酸序列的轻链可变区和包含如WO2013/028231中所示的SEQ ID NO:4的氨基酸序列的重链可变区。In another embodiment, the OX40 antigen binding protein is disclosed in WO2013/028231 (PCT/US2012/024570) (International Application Date February 9, 2012, which is incorporated herein by reference in its entirety). In another embodiment, the antigen binding protein comprises the CDRs of the antibodies disclosed in WO2013/028231 (PCT/US2012/024570) (International Application Date February 9, 2012), or CDRs with 90% identity to the disclosed CDR sequences. In a further embodiment, the antigen binding protein comprises VH, VL, or both of the antibodies disclosed in WO2013/028231 (PCT/US2012/024570) (International Application Date February 9, 2012), or VH or VL with 90% identity to the disclosed VH or VL sequences. In one embodiment, the OX40 antigen binding protein is an isolated agonist antibody against OX40 comprising a light chain variable region having a sequence having at least 90% identity to the amino acid sequence of SEQ ID NO: 10 as shown in WO2013/028231 and a heavy chain variable region having a sequence having at least 90% identity to the amino acid sequence of SEQ ID NO: 4 as shown in WO2013/028231. In one embodiment, the OX40 antigen binding protein is an isolated antibody comprising a light chain variable region comprising the amino acid sequence of SEQ ID NO: 10 as shown in WO2013/028231 and a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 4 as shown in WO2013/028231.
因此,在一个实施方案中,提供了治疗有需要的人的方法,其包括施用式(I)化合物或其盐和至少一种免疫调节剂。在一个实施方案中,所述免疫调节剂选自ICOS激动剂抗体、OX-40抗体或PD-1抗体。在一个实施方案中,所述人具有癌症。本文还提供了式(I)化合物或其盐与至少一种免疫调节剂组合用于治疗有需要的人的用途。Therefore, in one embodiment, a method of treating a person in need is provided, comprising administering a compound of formula (I) or a salt thereof and at least one immunomodulator. In one embodiment, the immunomodulator is selected from an ICOS agonist antibody, an OX-40 antibody, or a PD-1 antibody. In one embodiment, the person has cancer. Also provided herein is the use of a compound of formula (I) or a salt thereof in combination with at least one immunomodulator for treating a person in need.
用于与式(I)化合物或其盐组合使用或共同施用的其它治疗剂的额外实例是免疫刺激剂。An additional example of other therapeutic agents for use or co-administration in combination with the compounds of formula (I) or salts thereof are immunostimulants.
如本文所用,“免疫刺激剂”指可以刺激免疫系统的任何药剂。如本文所用,免疫刺激剂包括,但不限于,疫苗佐剂,诸如Toll样受体激动剂、T-细胞检查点阻断剂,诸如针对PD-1和CTL4的mAbs,和T-细胞检查点激动剂,诸如针对OX-40和ICOS的激动剂mAbs。As used herein, "immunostimulator" refers to any agent that can stimulate the immune system. As used herein, immunostimulatory agents include, but are not limited to, vaccine adjuvants such as Toll-like receptor agonists, T-cell checkpoint blockers such as mAbs to PD-1 and CTL4, and T-cell checkpoint agonists such as agonist mAbs to OX-40 and ICOS.
如本文所用的术语“Toll样受体”(或“TLR”)指感测微生物产物和/或启动适应性免疫应答的Toll样受体蛋白家族的成员或其片段。在一个实施方案中,TLR活化树突状细胞(DC)。Toll样受体(TLR)是模式识别受体家族,其最初被鉴定为识别微生物病原体的先天性免疫系统的传感器。TLR识别微生物中的不同结构,经常称为“PAMP”(病原体相关的分子模式)。结合TLR的配体引发细胞内信号转导途径的级联,其诱导参与炎症和免疫的因子的产生。在人类中,已经鉴定了十种TLR。在细胞表面表达的TLR包括TLR-1、-2、-4、-5和-6,而TLR-3、-7/8和-9表达有ER区(ER compartment)。人DC亚群可以根据不同的TLR表达模式进行鉴定。作为实例,DC的髓样或“常规”亚群(mDC)当被刺激时表达TLR 1-8,并产生活化标志物 (例如,CD80、CD86、I类和II类MHC、CCR7)、促炎细胞因子和趋化因子的级联。这种刺激和所得表达的结果是抗原特异性CD4+和CD8+T细胞引发(priming)。这些DC获取增强的摄取抗原的能力,并将其以适当的形式呈递给T细胞。相比之下,DC的浆细胞样亚群(pDC)在活化后仅表达TLR7和TLR9,导致活化NK细胞以及T细胞。因为死亡中的肿瘤细胞可能不利地影响DC功能,已经建议用TLR激动剂活化DC可能有益于在用于治疗癌症的免疫治疗方法中引发抗肿瘤免疫。还已经建议,使用放射和化学疗法成功治疗乳腺癌需要TLR4活化。As used herein, the term "Toll-like receptor" (or "TLR") refers to a member or fragment of the Toll-like receptor protein family that senses microbial products and/or initiates an adaptive immune response. In one embodiment, TLR activates dendritic cells (DCs). Toll-like receptors (TLRs) are a family of pattern recognition receptors that were originally identified as sensors of the innate immune system that recognize microbial pathogens. TLRs recognize different structures in microorganisms, often referred to as "PAMPs" (pathogen-associated molecular patterns). Ligands that bind to TLRs trigger a cascade of intracellular signal transduction pathways that induce the production of factors involved in inflammation and immunity. In humans, ten TLRs have been identified. TLRs expressed on the cell surface include TLR-1, -2, -4, -5, and -6, while TLR-3, -7/8, and -9 express ER compartments. Human DC subsets can be identified based on different TLR expression patterns. As an example, the myeloid or "conventional" subpopulation (mDC) of DC expresses TLR 1-8 when stimulated, and produces a cascade of activation markers (e.g., CD80, CD86, class I and class II MHC, CCR7), proinflammatory cytokines and chemokines. The result of this stimulation and the resulting expression is antigen-specific CD4+ and CD8+T cell priming. These DCs acquire the enhanced ability to ingest antigens and present them to T cells in an appropriate form. In contrast, the plasmacytoid subpopulation (pDC) of DC only expresses TLR7 and TLR9 after activation, resulting in activated NK cells and T cells. Because the tumor cells in death may adversely affect DC function, it has been suggested that activating DC with TLR agonists may be beneficial in eliciting anti-tumor immunity in immunotherapy methods for treating cancer. It has also been suggested that the successful treatment of breast cancer using radiation and chemotherapy requires TLR4 activation.
本领域已知且在本发明中有用的TLR激动剂包括,但不限于,以下:Pam3Cys,一种TLR1/2激动剂;CFA,一种TLR2激动剂;MALP2,一种TLR2激动剂;Pam2Cys,一种TLR2激动剂;FSL-I,一种TLR-2激动剂;Hib-OMPC,一种TLR-2激动剂;聚肌苷酸:聚胞苷酸(聚I:C),一种TLR3激动剂;聚腺苷-聚尿苷酸(聚AU),一种TLR3激动剂;用聚-L-赖氨酸和羧甲基纤维素稳定的聚肌苷酸-聚胞苷酸(Hiltonol),一种TLR3激动剂;细菌鞭毛蛋白,一种TLR5激动剂;咪喹莫特,一种TLR7激动剂;瑞喹莫德,一种TLR7/8激动剂;洛索立宾,一种TLR7/8激动剂;和未甲基化的CpG二核苷酸(CpG-ODN),一种TLR9激动剂。TLR agonists known in the art and useful in the present invention include, but are not limited to, the following: Pam3Cys, a TLR1/2 agonist; CFA, a TLR2 agonist; MALP2, a TLR2 agonist; Pam2Cys, a TLR2 agonist; FSL-I, a TLR-2 agonist; Hib-OMPC, a TLR-2 agonist; polyinosinic:polycytidylic acid (poly I:C), a TLR3 agonist; polyadenosine- Polyuridylic acid (poly-AU), a TLR3 agonist; polyinosinic-polycytidylic acid (HIltonol) stabilized with poly-L-lysine and carboxymethylcellulose, a TLR3 agonist; bacterial flagellin, a TLR5 agonist; imiquimod, a TLR7 agonist; resiquimod, a TLR7/8 agonist; loxoribine, a TLR7/8 agonist; and unmethylated CpG dinucleotide (CpG-ODN), a TLR9 agonist.
本领域已知的且在本发明中有用的额外TLR激动剂还包括,但不限于,氨基烷基氨基葡糖苷磷酸酯(AGP),其结合TLR4受体,已知可用作疫苗佐剂和免疫刺激剂用于在免疫的动物中刺激细胞因子产生,活化巨噬细胞,促进先天性免疫应答,以及增强抗体产生。天然存在的TLR4激动剂的实例是细菌脂多糖。适当地,TLR4激动剂为脂质A的无毒衍生物。脂质ATLR4激动剂的半合成无毒衍生物的实例为单磷酰脂质A,和特别是3-脱-O-酰化的单磷酰脂质A (3D-MPL)。3D-MPL由GlaxoSmithKline Biologicals S.A以名称MPL销售。AGP及其经由TLR4的免疫调节作用公开于专利公开文献,诸如WO2006/016997、WO 2001/090129和/或美国专利No. 6,113,918,并已报道于文献中。额外AGP衍生物公开于美国专利No. 7,129,219、美国专利No. 6,525,028和美国专利No. 6,911,434中。某些AGP充当TLR4的激动剂,而其它被认为是TLR4拮抗剂。Additional TLR agonists known in the art and useful in the present invention also include, but are not limited to, aminoalkyl aminoglucosyl phosphates (AGPs), which bind to the TLR4 receptor and are known to be used as vaccine adjuvants and immunostimulants for stimulating cytokine production in immunized animals, activating macrophages, promoting innate immune responses, and enhancing antibody production. An example of a naturally occurring TLR4 agonist is bacterial lipopolysaccharide. Suitably, the TLR4 agonist is a non-toxic derivative of lipid A. An example of a semi-synthetic non-toxic derivative of a lipid A TLR4 agonist is monophosphoryl lipid A, and particularly 3-de-O-acylated monophosphoryl lipid A (3D-MPL). 3D-MPL is sold by GlaxoSmithKline Biologicals S.A under the name MPL. AGP and its immunomodulatory effect via TLR4 are disclosed in patent publications, such as WO2006/016997, WO 2001/090129 and/or U.S. Patent No. 6,113,918, and have been reported in the literature. Additional AGP derivatives are disclosed in U.S. Patent No. 7,129,219, U.S. Patent No. 6,525,028 and U.S. Patent No. 6,911,434. Some AGPs serve as agonists of TLR4, while others are considered to be TLR4 antagonists.
在一个实施方案中,用于与本发明化合物组合使用的免疫刺激剂是TLR4激动剂。在一个实施方案中,TLR4激动剂被称为CRX-601和CRX-527。它们的结构如下所示:In one embodiment, the immunostimulant used in combination with the compounds of the invention is a TLR4 agonist. In one embodiment, the TLR4 agonists are referred to as CRX-601 and CRX-527. Their structures are shown below:
。 .
另外,另一个优选实施方案采用具有所示结构的TLR4激动剂CRX 547。In addition, another preferred embodiment employs the TLR4 agonist CRX 547 having the structure shown.
还有其它实施方案包括AGP,诸如CRX 602或CRX 526,其为具有较短仲酰基或烷基链的AGP提供增加的稳定性。Still other embodiments include AGPs such as CRX 602 or CRX 526, which provide increased stability for AGPs having shorter secondary acyl or alkyl chains.
因此,在一个实施方案中,提供了治疗有需要的人的方法,其包括施用式(I)化合物或其盐和至少一种免疫刺激剂。在一个实施方案中,所述免疫刺激剂是TLR4激动剂。在一个实施方案中,所述免疫刺激剂是AGP。在又另一个实施方案中,所述TLR4激动剂选自具有式CRX-601、CRX-527、CRX-547、CRX-602或CRX-526的化合物。在一个实施方案中,所述人具有癌症。本文还提供了式(I)化合物或其盐与至少一种免疫刺激剂的组合用于治疗有需要的人的用途。Therefore, in one embodiment, a method for treating a person in need is provided, comprising administering a compound of formula (I) or a salt thereof and at least one immunostimulant. In one embodiment, the immunostimulant is a TLR4 agonist. In one embodiment, the immunostimulant is AGP. In yet another embodiment, the TLR4 agonist is selected from a compound of formula CRX-601, CRX-527, CRX-547, CRX-602 or CRX-526. In one embodiment, the person has cancer. Also provided herein is the use of a combination of a compound of formula (I) or a salt thereof and at least one immunostimulant for treating a person in need.
除了上文所述的免疫刺激剂以外,本发明的组合物可以进一步包含其它治疗剂,由于它们的佐剂性质,其可以起作用以刺激免疫系统对存在于灭活肿瘤细胞上的癌抗原应答。这样的佐剂包括,但不限于,脂质、脂质体、诱导先天性免疫的灭活的细菌(例如,灭活或减毒的单核细胞增多性李斯特氏菌)、经由(NOD)样受体(NLDs)、基于视黄酸诱导基因(RIG)-I样受体(RLR)和/或C型凝集素受体(CLR)介导先天性免疫活化的组合物。PAMP的实例包括脂蛋白、脂多肽、肽聚糖、酵母聚糖、脂多糖、奈瑟菌孔蛋白、鞭毛蛋白、profillin、半乳糖神经酰胺(galactocerimide)、胞壁酰二肽。肽聚糖、脂蛋白和脂磷壁酸是革兰氏阳性的细胞壁组分。脂多糖由大多数细菌表达,其中MPL是一个实例。鞭毛蛋白指由致病菌和共生细菌分泌的细菌鞭毛的结构组分。rt.-半乳糖苷神经酰胺(rt.-GalCer)是自然杀伤T(NKT)细胞的活化剂。胞壁酰二肽是所有细菌共有的生物活性肽聚糖基序。In addition to the immunostimulants described above, the compositions of the present invention may further comprise other therapeutic agents that, due to their adjuvant properties, may act to stimulate the immune system to respond to cancer antigens present on inactivated tumor cells. Such adjuvants include, but are not limited to, lipids, liposomes, inactivated bacteria (e.g., inactivated or attenuated Listeria monocytogenes) that induce innate immunity, compositions that mediate innate immune activation via (NOD)-like receptors (NLDs), retinoic acid-induced gene (RIG)-I-like receptors (RLRs) and/or C-type lectin receptors (CLRs). Examples of PAMPs include lipoproteins, lipopolypeptides, peptidoglycans, yeast polysaccharides, neisseria porins, flagellins, profillin, galactocerimide, muramyl dipeptides. Peptidoglycans, lipoproteins, and lipoteichoic acid are Gram-positive cell wall components. Lipopolysaccharides are expressed by most bacteria, of which MPL is an example. Flagellin refers to the structural component of bacterial flagella secreted by pathogenic and commensal bacteria. rt.-Galactosylceramide (rt.-GalCer) is an activator of natural killer T (NKT) cells. Muramyl dipeptide is a biologically active peptidoglycan motif common to all bacteria.
由于它们的佐剂特质,优选TLR激动剂与其它疫苗、佐剂和/或免疫调节剂组合使用,并且可以在各种组合产品中进行组合。因此,在一些实施方案中,本文所述的结合STING并诱导STING依赖性TBKI活化的式(I)化合物和本文所述的表达并分泌刺激DC诱导、募集和/或成熟的一种或多种细胞因子的灭活肿瘤细胞(如本文所述)可以与一种或多种TLR激动剂一起施用以用于治疗目的。Due to their adjuvant properties, TLR agonists are preferably used in combination with other vaccines, adjuvants and/or immunomodulators, and can be combined in various combination products. Therefore, in some embodiments, the compounds of formula (I) described herein that bind to STING and induce STING-dependent TBKI activation and the inactivated tumor cells described herein that express and secrete one or more cytokines that stimulate DC induction, recruitment and/or maturation (as described herein) can be administered together with one or more TLR agonists for therapeutic purposes.
吲哚胺2,3-双加氧酶1 (IDO1)是调节抗肿瘤免疫应答的关键免疫抑制酶,其通过促进调节性T细胞生成和阻断效应T细胞活化,由此通过允许癌细胞避免免疫监视而促进肿瘤生长。(Lemos H等人,Cancer Res. 2016年4月15日;76(8):2076-81),(Munn DH等人,Trends Immunol. 2016年3月;37(3):193-207)。用于与本发明的式(I)化合物组合使用或共同施用的其它活性成分(抗肿瘤剂)是IDO抑制剂。Epacadostat,即((Z)-N-(3-溴-4-氟苯基)-N'-羟基-4-[2-(氨磺酰氨基)乙基氨基]-1,2,5-噁二唑-3-甲脒)是高度有效且选择性的IDO1酶的口服抑制剂,其逆转肿瘤相关的免疫抑制并恢复有效的抗肿瘤免疫应答。Epacadostat公开于美国专利No. 8,034,953中。Indoleamine 2,3-dioxygenase 1 (IDO1) is a key immunosuppressive enzyme that regulates anti-tumor immune responses by promoting the generation of regulatory T cells and blocking effector T cell activation, thereby promoting tumor growth by allowing cancer cells to avoid immune surveillance. (Lemos H et al., Cancer Res. 2016 Apr 15; 76(8): 2076-81), (Munn DH et al., Trends Immunol. 2016 Mar; 37(3): 193-207). The other active ingredient (antitumor agent) used in combination or co-administered with the compound of formula (I) of the present invention is an IDO inhibitor. Epacadostat, ((Z)-N-(3-bromo-4-fluorophenyl)-N'-hydroxy-4-[2-(sulfamoylamino)ethylamino]-1,2,5-oxadiazole-3-carboxamidine) is a highly potent and selective oral inhibitor of the IDO1 enzyme that reverses tumor-associated immunosuppression and restores effective anti-tumor immune responses. Epacadostat is disclosed in U.S. Pat. No. 8,034,953.
用于与式(I)化合物组合或共同施用的其它治疗剂(抗肿瘤剂)的额外实例是CD73抑制剂和A2a和A2b腺苷拮抗剂。Additional examples of other therapeutic agents (anti-tumor agents) for combination or co-administration with the compounds of formula (I) are CD73 inhibitors and A2a and A2b adenosine antagonists.
在一个实施方案中,本发明化合物可以与治疗感染性疾病的其它治疗方法一起使用。特别地,设想抗病毒剂和抗菌剂。In one embodiment, the compounds of the invention may be used in conjunction with other therapeutic approaches for treating infectious diseases. In particular, antiviral agents and antibacterial agents are contemplated.
式(I)化合物及其药学上可接受的盐可以与至少一种可用于预防或治疗细菌和病毒感染的其它治疗剂组合使用。这样的药剂的实例包括但不限于:聚合酶抑制剂,诸如WO2004/037818-A1中公开的那些,以及WO 2004/037818和WO 2006/045613中公开的那些;JTK-003、JTK-019、NM-283、HCV-796、R-803、R1728、R1626以及WO 2006/018725、WO 2004/074270、WO 2003/095441、US2005/0176701、WO 2006/020082、WO 2005/080388、WO 2004/064925、WO 2004/065367、WO 2003/007945、WO 02/04425、WO 2005/014543、WO 2003/000254、EP 1065213、WO 01/47883、WO 2002/057287、WO 2002/057245中公开的那些和类似药剂;复制抑制剂,诸如阿昔洛韦、泛昔洛韦、更昔洛韦、西多福韦、拉米夫定和类似药剂;蛋白酶抑制剂,诸如HIV蛋白酶抑制剂沙奎那韦、利托那韦、茚地那韦、奈非那韦、安普那韦、夫沙那韦、布瑞那韦、阿扎那韦、替拉那韦、帕利那韦、拉西那韦和HCV蛋白酶抑制剂BILN2061、VX-950、SCH503034;和类似药剂;核苷和核苷酸逆转录酶抑制剂,诸如齐多夫定、地达诺新、拉米夫定、扎西他滨、阿巴卡韦、司他夫定、阿德福韦、阿德福韦酯、福齐夫定、todoxil、恩曲他滨、阿洛夫定、氨多索韦、艾夫他滨、富马酸替诺福韦酯、富马酸/半富马酸替诺福韦艾拉酚胺和类似药剂;非核苷逆转录酶抑制剂(包括具有抗氧化活性的药剂,诸如怡妙康(immunocal)、奥替普拉等),诸如奈韦拉平、地拉韦啶、依法韦仑、洛韦胺、怡妙康、奥替普拉、卡普韦林(capravirine)、TMC-278、TMC-125、依曲韦林、利匹韦林和类似药剂;进入抑制剂,诸如恩夫韦地(T-20)、T-1249、PRO-542、PRO-140、TNX-355、BMS-806、5-Helix和类似药剂;整合酶抑制剂,诸如度鲁特韦、艾维雷韦、拉替拉韦 L-870,180和类似药剂;出芽抑制剂,诸如PA-344和PA-457和类似药剂;趋化因子受体抑制剂,诸如维立韦罗(Sch-C)、Sch-D、TAK779、马拉韦罗(UK-427,857)、TAK449,以及WO 02/74769、WO 2004/054974、WO 2004/055012、WO 2004/055010、WO 2004/055016、WO 2004/055011和WO 2004/054581中公开的那些和类似药剂;药代动力学增强剂诸如可比司他;神经氨酸酶抑制剂,诸如CS-8958、扎那米韦、奥司他韦、培拉米韦和类似药剂;离子通道阻断剂,诸如金刚烷胺或金刚乙胺和类似药剂;和干扰RNA和反义寡核苷酸和诸如ISIS-14803和类似药剂;未确定作用机制的抗病毒剂,例如WO 2005/105761、WO 2003/085375、WO 2006/122011中公开的那些、利巴韦林和类似药剂。The compounds of formula (I) and their pharmaceutically acceptable salts may be used in combination with at least one other therapeutic agent useful for preventing or treating bacterial and viral infections. Examples of such agents include, but are not limited to, polymerase inhibitors such as those disclosed in WO 2004/037818-A1, and those disclosed in WO 2004/037818 and WO 2006/045613; JTK-003, JTK-019, NM-283, HCV-796, R-803, R1728, R1626, and WO 2006/018725, WO 2004/074270, WO 2003/095441, US 2005/0176701, WO 2006/020082, WO 2005/080388, WO 2004/064925, WO 2004/065367, WO 2003/007945, WO 02/04425、WO 2005/014543、WO 2003/000254、EP 1065213、WO 01/47883、WO 2002/057287、WO Those disclosed in 2002/057245 and similar agents; replication inhibitors such as acyclovir, famciclovir, ganciclovir, cidofovir, lamivudine and similar agents; protease inhibitors such as the HIV protease inhibitors saquinavir, ritonavir, indinavir, nelfinavir, amprenavir, fosamprenavir, brenavir, atazanavir, tipranavir, palinavir, lasinavir and HCV protease inhibitors BILN2061, VX-950, SCH503034; and similar agents; nucleoside and nucleotide reverse transcriptase inhibitors such as zidovudine, didanosine, lamivudine, zalcitabine, abacavir, stavudine, adefovir, adefovir dipivoxil, fozivudine, todoxil, emtricitabine, alovudine, aminodosulf , eficitabine, tenofovir disoproxil fumarate, tenofovir alafenamide fumarate/hemifumarate, and similar agents; non-nucleoside reverse transcriptase inhibitors (including agents with antioxidant activity, such as immunocal, oltipraz, etc.), such as nevirapine, delavirdine, efavirenz, loviramide, immunocal, oltipraz, capravirine, TMC-278, TMC-125, etravirine, rilpivirine, and similar agents; entry inhibitors, such as enfuvirtide (T-20), T-1249, PRO-542, PRO-140, TNX-355, BMS-806, 5-Helix, and similar agents; integrase inhibitors, such as dolutegravir, elvitegravir, raltegravir L-870, 180 and similar agents; budding inhibitors such as PA-344 and PA-457 and similar agents; chemokine receptor inhibitors such as viribviroc (Sch-C), Sch-D, TAK779, maraviroc (UK-427,857), TAK449, and WO 02/74769, WO 2004/054974, WO 2004/055012, WO 2004/055010, WO 2004/055016, WO 2004/055011 and WO 2004/054581 and similar agents; pharmacokinetic enhancers such as cobicistat; neuraminidase inhibitors such as CS-8958, zanamivir, oseltamivir, peramivir and similar agents; ion channel blockers such as amantadine or rimantadine and similar agents; and interfering RNA and antisense oligonucleotides and such as ISIS-14803 and similar agents; antiviral agents of undetermined mechanism of action, such as those disclosed in WO 2005/105761, WO 2003/085375, WO 2006/122011, ribavirin and similar agents.
式(I)化合物及其药学上可接受的盐也可以与其它治疗剂组合使用,所述其它治疗剂可用于治疗卡波西肉瘤相关疱疹病毒感染(KSHV和KSHV相关),包括但不限于化学治疗剂,诸如博来霉素、长春碱、长春新碱、环磷酰胺、泼尼松、阿利维A酸和脂质体蒽环霉素类诸如多柔比星、柔红霉素、免疫治疗剂诸如利妥昔单抗、托珠单抗、司妥昔单抗和其它诸如紫杉醇和雷帕霉素。The compounds of formula (I) and their pharmaceutically acceptable salts may also be used in combination with other therapeutic agents useful for treating Kaposi's sarcoma-associated herpesvirus infection (KSHV and KSHV-associated), including but not limited to chemotherapeutic agents such as bleomycin, vinblastine, vincristine, cyclophosphamide, prednisone, alitretinoin and liposomal anthracyclines such as doxorubicin, daunorubicin, immunotherapeutic agents such as rituximab, tocilizumab, siltuximab and others such as paclitaxel and rapamycin.
在本发明的一个实施方案中,至少一种其它治疗剂是抗分枝杆菌剂或杀细菌抗生素。式(I)化合物及其药学上可接受的盐也可以与至少一种其它治疗剂组合使用,所述其它治疗剂可用于治疗TB感染(结核分枝杆菌)和兔热病(土拉弗朗西斯菌),包括但不限于一线口服药剂异烟肼、利福平、吡嗪酰胺、乙胺丁醇、链霉素、利福布汀;可注射剂,包括卡那霉素、阿米卡星、卷曲霉素、链霉素;氟喹诺酮类,包括左氧氟沙星莫西沙星氧氟沙星;口服抑菌剂对氨基水杨酸环丝氨酸三嗪酮硫代酰胺丙硫异烟胺;SQ-109 PNU-100480、利福喷汀利奈唑胺、PA-824 AZD5847、加替沙星莫西沙星、Sirturo(贝达喹啉)德拉马尼(OPC-67683)和在治疗耐药性结核病中具有不确定作用机制的药物,包括氯法齐明、利奈唑胺、阿莫西林/克拉维酸氨硫脲亚胺培南/西司他丁高剂量异烟肼克拉霉素、环丙沙星。式(I)化合物及其药学上可接受的盐还可以与以下组合:抗分枝杆菌剂(诸如异烟肼(INH)、乙胺丁醇(Myambutol®)、利福平(Rifadin®)和吡嗪酰胺(PZA));杀细菌抗生素(诸如利福布汀(Mycobutin®)或利福喷汀(Priftin®))、氨基糖苷(Capreomycin®)、氟喹诺酮(左氧氟沙星、莫西沙星、氧氟沙星)、硫代酰胺(乙硫异烟胺)、环孢菌素(Sandimmune®)、对氨基水杨酸(Paser®)、环丝氨酸(Seromycin®)、卡那霉素(Kantrex®)、链霉素、紫霉素、卷曲霉素(Capastat®))、富马酸贝达喹啉(Sirturo®)、噁唑烷酮(Sutezolid®)、PNU-100480或德拉马尼(OPC-67683)。In one embodiment of the invention, at least one other therapeutic agent is an antimycobacterial agent or a bactericidal antibiotic. The compounds of formula (I) and their pharmaceutically acceptable salts may also be used in combination with at least one other therapeutic agent that can be used to treat TB infection (Mycobacterium tuberculosis) and tularemia (Francisco tularensis), including but not limited to first-line oral agents isoniazid, rifampicin, pyrazinamide, ethambutol, streptomycin, rifabutin; injectable agents, including kanamycin, amikacin, capreomycin, streptomycin; fluoroquinolones, including levofloxacin, moxifloxacin, ofloxacin; oral antibacterial agents para-aminosalicylic acid, cycloserine, triazone, thioamide, prothionamide; SQ-109, PNU-100480, rifapentine, linezolid, PA-824 AZD5847, gatifloxacin-moxifloxacin, Sirturo (bedaquiline) delamanid (OPC-67683), and drugs with uncertain mechanisms of action in the treatment of drug-resistant TB, including clofazimine, linezolid, amoxicillin/clavulanate thiosemicarbazide imipenem/cilastatin high-dose isoniazid clarithromycin, and ciprofloxacin. The compounds of formula (I) and their pharmaceutically acceptable salts may also be combined with antimycobacterial agents (such as isoniazid (INH), ethambutol ( Myambutol® ), rifampicin ( Rifadin® ) and pyrazinamide (PZA)); bactericidal antibiotics (such as rifabutin ( Mycobutin® ) or rifapentine ( Priftin® )), aminoglycosides ( Capreomycin® ), fluoroquinolones (levofloxacin, moxifloxacin, ofloxacin), thioamides (ethionamide), cyclosporine ( Sandimmune® ), para-aminosalicylic acid ( Paser® ), cycloserine ( Seromycin® ), kanamycin ( Kantrex® ), streptomycin, puromycin, capreomycin ( Capastat® ), bedaquiline fumarate ( Sirturo® ), oxazolidinone (Sutezolid ... ), PNU-100480 or delamanid (OPC-67683).
式(I)化合物及其药学上可接受的盐也可以与至少一种其它治疗剂组合使用,所述其它治疗剂可用于治疗衣原体,包括但不限于阿奇霉素、多西环素、红霉素、左氧氟沙星、氧氟沙星。The compound of formula (I) and its pharmaceutically acceptable salts can also be used in combination with at least one other therapeutic agent that can be used to treat Chlamydia, including but not limited to azithromycin, doxycycline, erythromycin, levofloxacin, ofloxacin.
本发明化合物也可以与至少一种其它治疗剂组合使用,所述其它治疗剂可用于治疗疟原虫感染,包括但不限于氯喹、阿托伐醌-氯胍(proguanil)、蒿甲醚-本芴醇、甲氟喹、奎宁、奎尼丁、多西环素、克林霉素、青蒿琥酯、伯氨喹。The compounds of the invention may also be used in combination with at least one other therapeutic agent useful for treating Plasmodium infection, including but not limited to chloroquine, atovaquone-proguanil, artemether-lumefantrine, mefloquine, quinine, quinidine, doxycycline, clindamycin, artesunate, primaquine.
在肌萎缩侧索硬化症(ALS)的治疗中,式(I)化合物或其药学上可接受的盐可以与以下组合使用:谷氨酸阻断剂(利鲁唑(Rilutek®))、奎尼丁(Nuedexta ®)、抗胆碱能药(阿米替林®、Artane ®、东莨菪碱贴剂(Transderm Scop ®))、拟交感神经药(伪麻黄碱)、粘液溶解剂(愈创甘油醚)或镇痛药(曲马多(Ultram ®));酮咯酸(Toradol®);吗啡;芬太尼贴剂(Duragesic®))。In the treatment of amyotrophic lateral sclerosis (ALS), a compound of formula (I) or a pharmaceutically acceptable salt thereof may be used in combination with: a glutamate blocker (riluzole (Rilutek®)), quinidine (Nuedexta®), an anticholinergic (amitriptyline®, Artane®, scopolamine patch (Transderm Scop®)), a sympathomimetic (pseudoephedrine), a mucolytic (guaifenesin), or an analgesic (tramadol (Ultram®)); ketorolac (Toradol®); morphine; fentanyl patch (Duragesic®)).
在多发性硬化症的治疗中,式(I)化合物或其药学上可接受的盐可以与以下组合使用:皮质类固醇(泼尼松、甲基泼尼松龙)、干扰素β1-A (Avonex®、Extavia®、Rebif®、Betaseron®)、聚乙二醇干扰素β-1A (Plegridy®)、醋酸格拉替雷(Copaxone®);醋酸格拉替雷(Glatopa®-Copaxone的通用等同物);富马酸二甲酯(Tecfidera®);芬戈莫德(Gilenya®));特立氟胺(Aubagio®);达伐吡啶(Ampyra®);达克珠单抗(Zinbryta);阿仑单抗(Lemtrada®);那他珠单抗(Tysabri®);或米托蒽醌盐酸盐(Novantrone®)。In the treatment of multiple sclerosis, the compound of formula (I) or a pharmaceutically acceptable salt thereof can be used in combination with: corticosteroids (prednisone, methylprednisolone), interferon beta 1-A (Avonex®, Extavia®, Rebif®, Betaseron®), peginterferon beta-1A (Plegridy®), glatiramer acetate (Copaxone®); glatiramer acetate (Glatopa®-generic equivalent of Copaxone); dimethyl fumarate (Tecfidera®); fingolimod (Gilenya®); teriflunomide (Aubagio®); dalfampridine (Ampyra®); daclizumab (Zinbryta); alemtuzumab (Lemtrada®); natalizumab (Tysabri®); or mitoxantrone hydrochloride (Novantrone®).
本发明化合物还可以用作佐剂以在有需要的患者、特别是人中改善对任何给定抗原产生的免疫应答和/或降低反应原性/毒性。因此,本发明化合物可以与疫苗组合物组合使用,例如通过增加保护的水平或持续时间和/或允许减少抗原剂量来修饰,尤其是增强免疫应答。The compounds of the invention can also be used as adjuvants to improve the immune response and/or reduce the reactogenicity/toxicity to any given antigen in patients, particularly humans, in need thereof. Thus, the compounds of the invention can be used in combination with vaccine compositions to modify, in particular enhance, the immune response, for example by increasing the level or duration of protection and/or allowing a reduction in the antigen dose.
式(I)化合物及其药学上可接受的盐可以与一种或多种用于预防或治疗病毒感染的疫苗或免疫原性抗原组合使用。这样的疫苗或免疫原性抗原包括,但不限于病原体衍生的蛋白质或颗粒诸如通常用作免疫原性物质的减毒病毒、病毒颗粒和病毒蛋白。病毒和病毒抗原来源的实例包括,但不限于脊髓灰质炎病毒、冠状病毒科和冠状病毒、鼻病毒(所有亚型)、腺病毒(所有亚型)、甲型肝炎、乙型肝炎、丙型肝炎、丁型肝炎、人乳头状瘤病毒(包括所有亚型)、狂犬病病毒、人T-淋巴细胞病毒(所有亚型)、风疹病毒、腮腺炎病毒、柯萨奇病毒A(所有亚型)、柯萨奇病毒B(所有亚型)、人肠病毒、疱疹病毒(包括巨细胞病毒)、Epstein-Barr病毒、人疱疹病毒(所有亚型)、单纯疱疹病毒、水痘带状疱疹病毒、人免疫缺陷病毒(HIV)(所有亚型)、Epstein-Barr病毒、呼肠孤病毒(所有亚型)、丝状病毒(Filoviruses)(包括马尔堡病毒和埃博拉病毒(所有病毒株))、沙粒病毒(包括淋巴细胞性脉络丛脑膜炎病毒、拉沙病毒、胡宁病毒和Machupo病毒)、虫媒病毒(包括西尼罗河病毒)、登革病毒(所有血清型)、寨卡病毒、科罗拉多蜱热病毒、辛德比斯病毒、披膜病毒科(Togaviraidae)、黄病毒科、布尼亚病毒科、呼肠孤病毒科、弹状病毒科、正粘病毒科、痘病毒(包括正痘病毒(天花病毒、猴痘病毒、痘苗病毒、牛痘病毒)、亚塔痘病毒(特纳河痘病毒、雅巴猴肿瘤病毒)、副痘病毒、软疣痘病毒)、黄热病、汉坦病毒(包括Hantaan、Seoul、Dobrava、Sin Nombre、Puumala和Dobrava-样Saaremaa)、人副流感病毒和流感病毒(所有类型)、H1N1流感和猪流感病毒、呼吸道合胞体病毒(所有亚群)、轮状病毒(包括人轮状病毒A-E、牛轮状病毒、猕猴轮状病毒)、多瘤病毒(包括猿猴病毒40、JC病毒、BK病毒、科罗拉多壁虱热病毒(Coltiviruses)、依埃契病毒、杯状病毒)、和细小病毒科(包括依赖病毒、细小病毒和红病毒(Erythrovirus))。The compound of formula (I) and its pharmaceutically acceptable salt can be used in combination with one or more vaccines or immunogenic antigens for preventing or treating viral infection. Such vaccines or immunogenic antigens include, but are not limited to pathogen-derived proteins or particles such as attenuated viruses, viral particles and viral proteins commonly used as immunogenic substances. Examples of viruses and sources of viral antigens include, but are not limited to, poliovirus, Coronaviridae and Coronavirus, Rhinovirus (all subtypes), Adenovirus (all subtypes), Hepatitis A, Hepatitis B, Hepatitis C, Hepatitis D, Human Papillomavirus (including all subtypes), Rabies virus, Human T-lymphotropic virus (all subtypes), Rubella virus, Mumps virus, Coxsackievirus A (all subtypes), Coxsackievirus B (all subtypes), Human enterovirus, Herpesvirus (including cytomegalovirus), Epstein-Barr virus, Human herpesvirus (all subtypes), Herpes simplex virus, Varicella-zoster virus, Human immunodeficiency virus (HIV) (all subtypes), Epstein-Barr virus, Reovirus (all subtypes), Filovirus (Filovivirus), ruses) (including Marburg virus and Ebola virus (all strains)), Arenaviruses (including Lymphocytic Choriomeningitis virus, Lassa virus, Junin virus and Machupo virus), Arboviruses (including West Nile virus), Dengue virus (all serotypes), Zika virus, Colorado tick fever virus, Sindbis virus, Togaviridae, Flaviviridae, Bunyaviridae, Reoviridae, Rhabdoviridae, Orthomyxoviridae, Poxviruses (including Orthopoxviruses (Variola virus, Monkeypox virus, Vaccinia virus, Vaccinia virus), Yatapoxviruses (Tenapox virus, Yaba monkey tumor virus), Parapoxviruses, Molluscumpox virus), Yellow fever, Hantaviruses (including Hantaan, Seoul, Dobrava, Sin Nombre, Puumala and Dobrava-like Saaremaa), human parainfluenza and influenza viruses (all types), H1N1 influenza and swine influenza viruses, respiratory syncytial virus (all subgroups), rotaviruses (including human rotavirus A-E, bovine rotavirus, macaque rotavirus), polyomaviruses (including simian virus 40, JC virus, BK virus, Colorado tick fever virus (Coltiviruses), Eichhornia virus, Calicivirus), and Parvoviridae (including Dependovirus, Parvovirus and Erythrovirus).
另一方面,本发明提供治愈HIV的方法,包括向对象施用本发明化合物。患者“治愈”(“cure”或“curing”)用于表示人免疫缺陷病毒或症状的消灭、停止、停下或终止,或所述症状或病毒的进展消灭、停止、停下或终止一定时间。作为一个实例,在一个实施方案中,“治愈”指治疗性施用或组合施用,其单独或与一种或多种药剂组合以在最短例如一年或两年后不进行任何其它治疗性干预下诱导并保持人免疫缺陷病毒的持续的病毒控制(通过例如聚合酶链式反应(PCR)测试、bDNA(支链DNA)测试或NASBA(基于核酸序列的放大)测试获得的不可检测水平的等离子体病毒血症)。使用本领域技术人员已知和熟悉的技术进行上述PCR、bDNA和NASBA测试。作为一个实例,人免疫缺陷病毒或症状的消灭、停止、停下或终止,或所述症状或病毒的进展消灭、停止、停下或终止可能持续最少两年。On the other hand, the present invention provides a method for curing HIV, comprising administering the compounds of the present invention to a subject. Patient "cure" ("cure" or "curing") is used to indicate the elimination, cessation, stop or termination of human immunodeficiency virus or symptoms, or the elimination, cessation, stop or termination of the progression of the symptoms or virus for a certain period of time. As an example, in one embodiment, "cure" refers to therapeutic administration or combined administration, which alone or in combination with one or more agents to induce and maintain sustained viral control of human immunodeficiency virus (undetectable levels of plasma viremia obtained by, for example, polymerase chain reaction (PCR) test, bDNA (branched DNA) test or NASBA (nucleic acid sequence-based amplification) test) without any other therapeutic intervention after a minimum of, for example, one or two years. The above-mentioned PCR, bDNA and NASBA tests are performed using techniques known and familiar to those skilled in the art. As an example, the elimination, cessation, stop or termination of human immunodeficiency virus or symptoms, or the elimination, cessation, stop or termination of the progression of the symptoms or virus may last for at least two years.
在本发明的另一实施方案中,提供用于治愈HIV感染的本发明化合物。In another embodiment of the present invention, there is provided a compound of the present invention for use in curing HIV infection.
在本发明的另一实施方案中,提供本发明化合物在制备药物中的用途,所述药物用于治愈HIV感染。In another embodiment of the present invention, there is provided the use of a compound of the present invention in the preparation of a medicament for curing HIV infection.
另一方面,提供一种组合产品,其包含本发明化合物和一种或多种针对HIV的另外的药物活性剂。这样的化合物和药剂可以存在于药物制剂或组合物中。因此,本发明还包括治疗、治愈和/或预防对象的HIV感染的方法,即向对象施用包含本发明化合物和针对HIV的一种或多种另外的药物活性剂的组合产品(或其药物制剂或组合物)。In another aspect, a combination product is provided, which comprises a compound of the invention and one or more additional pharmaceutically active agents for HIV. Such compounds and agents may be present in a pharmaceutical preparation or composition. Therefore, the present invention also includes a method for treating, curing and/or preventing HIV infection in a subject, i.e., administering to the subject a combination product (or a pharmaceutical preparation or composition thereof) comprising a compound of the invention and one or more additional pharmaceutically active agents for HIV.
在这样的实施方案中,所述针对HIV的一种或多种另外的活性剂选自齐多夫定、地达诺新、拉米夫定、扎西他滨、阿巴卡韦、司他夫定、阿德福韦、阿德福韦酯、福齐夫定替酯(fozivudine, todoxil)、恩曲他滨、阿洛夫定、氨多索韦、艾夫他滨、奈韦拉平、地拉夫定、依法韦仑、洛韦胺、怡妙康(immunocal)、奥替普拉、卡普韦林、lersivirine、GSK2248761、TMC-278、TMC-125、依曲韦林、沙奎那韦、利托那韦、茚地那韦、奈非那韦、安普那韦、夫沙那韦(fosamprenavir)、贝卡那韦、地瑞纳韦、阿扎那韦、替拉那韦、帕利那韦、拉西那韦(lasinavir)、恩夫韦地、T-20、T-1249、PRO-542、PRO-140、TNX-355、BMS-806、BMS-663068和BMS-626529、5-Helix、雷特格韦、埃替格韦 、度鲁特韦、卡博替韦(cabotegrair)、vicriviroc (Sch-C)、Sch-D、TAK779、马拉维诺、TAK449、地达诺新、替诺福韦、洛匹那韦和地瑞纳韦。In such an embodiment, the one or more additional active agents against HIV are selected from zidovudine, didanosine, lamivudine, zalcitabine, abacavir, stavudine, adefovir, adefovir dipivoxil, fozivudine, todoxil), emtricitabine, alovudine, amdosovir, efcitabine, nevirapine, delavirdine, efavirenz, loviride, immunocal, oltipraz, caprovirine, lersivirine, GSK2248761, TMC-278, TMC-125, etravirine, saquinavir, ritonavir, indinavir, nelfinavir, amprenavir, fosamprenavir, becanavir, darunavir, atazanavir, tipranavir, palinavir, lasinavir, enfuvirtide, T-20, T-1249, PRO-542, PRO-140, TNX-355, BMS-806, BMS-663068 and BMS-626529, 5-Helix, raltegravir, elvitegravir , dolutegravir, cabotegrair, vicriviroc (Sch-C), Sch-D, TAK779, maraviroc, TAK449, didanosine, tenofovir, lopinavir, and darunavir.
因此,本发明化合物和一种或多种任何其它药物活性剂可以一起地或单独地施用,并且当单独施用时,可以同时或以任何顺序依次施用。可以选择本发明化合物和所述一种或多种其它药物活性剂的量以及施用的相对时间从而实现所需的组合治疗效果。本发明化合物与其它治疗剂的组合施用可以通过以下方式伴随施用来组合:(1)包括两种化合物的单一药物组合物;或(2) 各自包括化合物之一的单独的药物组合物。或者,可以依次的方式单独地施用所述组合产品,其中首先施用一种治疗剂,然后另外的第二种,反之亦然。这样的依次施用可以在时间上接近或在时间上远离。选择本发明化合物和针对HIV的一种或多种其它药物活性剂的量和施用的相对时间从而实现所需的组合治疗效果。Therefore, the compounds of the present invention and one or more other pharmaceutically active agents can be administered together or separately, and when administered separately, they can be administered simultaneously or sequentially in any order. The amount of the compounds of the present invention and the one or more other pharmaceutically active agents and the relative time of administration can be selected to achieve the desired combined therapeutic effect. The combined administration of the compounds of the present invention and other therapeutic agents can be combined by concomitant administration in the following manner: (1) a single pharmaceutical composition comprising two compounds; or (2) a separate pharmaceutical composition each comprising one of the compounds. Alternatively, the combination product can be administered separately in a sequential manner, wherein one therapeutic agent is administered first, then the other second, and vice versa. Such sequential administration can be close in time or distant in time. The amount of the compounds of the present invention and one or more other pharmaceutically active agents for HIV and the relative time of administration are selected to achieve the desired combined therapeutic effect.
另外,本发明化合物可以与可用于预防、治疗或治愈HIV的一种或多种其它药剂组合使用。这样的药剂的实例包括:核苷酸逆转录酶抑制剂诸如齐多夫定、地达诺新、拉米夫定、扎西他滨、阿巴卡韦、司他夫定、阿德福韦、阿德福韦酯、福齐夫定替酯(fozivudine,todoxil)、恩曲他滨、阿洛夫定、氨多索韦、艾夫他滨、TDF、TAF和类似药剂;非核苷酸逆转录酶抑制剂 (包括具有抗氧化活性的药剂,诸如怡妙康、奥替普拉等) 诸如奈韦拉平、地拉夫定、依法韦仑、洛韦胺、怡妙康、奥替普拉、卡普韦林、lersivirine、GSK2248761、TMC-278、TMC-125、依曲韦林、和类似药剂;蛋白酶抑制剂诸如沙奎那韦、利托那韦、茚地那韦、奈非那韦、安普那韦、夫沙那韦、贝卡那韦、地瑞纳韦、阿扎那韦、替拉那韦、帕利那韦、拉西那韦、和类似药剂;整合酶抑制剂诸如雷特格韦、埃替格韦 、比特雷韦、度鲁特韦、卡博替韦 和类似药剂;成熟抑制剂诸如PA-344和PA-457和类似药剂;和GSK2838232.CXCR4 和/或 CCR5抑制剂诸如vicriviroc (Sch-C)、Sch-D、TAK779、马拉维诺 (UK 427,857)、TAK449以及在WO02/74769、PCT/US03/39644、PCT/US03/39975、PCT/US03/39619、PCT/US03/39618、PCT/US03/39740和PCT/US03/39732中公开的那些和类似药剂。在表A中列出本发明化合物可以与可用于预防或治疗HIV的一种或多种药剂组合使用的其它实例。In addition, the compounds of the present invention can be used in combination with one or more other agents that can be used to prevent, treat or cure HIV. Examples of such agents include: nucleotide reverse transcriptase inhibitors such as zidovudine, didanosine, lamivudine, zalcitabine, abacavir, stavudine, adefovir, adefovir dipivoxil, fozivudine, todoxil, emtricitabine, alovudine, amide, efavirenz, TDF, TAF and similar agents; non-nucleotide reverse transcriptase inhibitors (including agents with antioxidant activity, such as imipenem, oltipraz, etc.) such as nevirapine, delavirdine, efavirenz, loviride, imipenem, oltipraz, caprovirine, lersivirine, GSK2248761, TMC-278, TMC-125, etravirine, and similar agents; protease inhibitors such as saquinavir, ritonavir, indinavir, nelfinavir, amprenavir, fosamprenavir, becanavir, darunavir, atazanavir, tipranavir, palinavir, lasinavir, and similar agents; integrase inhibitors such as raltegravir, elvitegravir, bitravir, dolutegravir, cabotegravir and similar agents; maturation inhibitors such as PA-344 and PA-457 and similar agents; and GSK2838232. CXCR4 and/or CCR5 inhibitors such as vicriviroc (Sch-C), Sch-D, TAK779, maraviroc (UK 427,857), TAK449, and those disclosed in WO02/74769, PCT/US03/39644, PCT/US03/39975, PCT/US03/39619, PCT/US03/39618, PCT/US03/39740, and PCT/US03/39732, and similar agents. Other examples of the compounds of the present invention that can be used in combination with one or more agents useful for preventing or treating HIV are listed in Table A.
表ATable A
。 .
本发明化合物与HIV药剂的组合产品的范围不限于上文所述的那些,原则上还包括与可用于治愈、治疗和/或预防HIV的任何药物组合物的任何组合产品。如所述地,在这样的组合产品中,本发明化合物和其它HIV药剂可以单独地或结合地施用。另外,一种药剂可以在施用一种或多种其它药剂之前、同时或之后施用。The scope of the combination products of the compounds of the present invention and HIV agents is not limited to those described above, and in principle also includes any combination products with any pharmaceutical composition that can be used to cure, treat and/or prevent HIV. As described, in such a combination product, the compounds of the present invention and other HIV agents can be administered individually or in combination. In addition, one agent can be administered before, simultaneously with or after the administration of one or more other agents.
本发明可以在存在或不存在预防或治疗HIV的另外的化合物的情况下与可用作药物增强剂(pharmacological enhancer)的一种或多种药剂组合使用。这样的药物增强剂(或药代动力学增效剂)的实例包括但不限于利托那韦、GS-9350和SPI-452。The present invention can be used in combination with one or more agents that can be used as pharmacological enhancers, in the presence or absence of additional compounds for preventing or treating HIV. Examples of such pharmacological enhancers (or pharmacokinetic enhancers) include, but are not limited to, ritonavir, GS-9350, and SPI-452.
利托那韦为10-羟基-2-甲基-5-(1-甲基乙基)-1-1[2-(1-甲基乙基)-4-噻唑基]-3,6-二氧代-8,11-双(苯基甲基)-2,4,7,12-四氮杂十三烷-13-酸, 5-噻唑基甲基酯[5S-(5S*,8R*,10R*,11R*)]并可获自Abbott park, Illinois的Abbott Laboratories,作为Norvir。利托那韦为HIV蛋白酶抑制剂,表明与其它抗逆转录病毒药剂一起用于治疗HIV感染。利托那韦还抑制P450介导的药物代谢以及P-糖蛋白(Pgp)细胞传输系统,从而导致机体内活性化合物的浓度增加。Ritonavir is 10-hydroxy-2-methyl-5-(1-methylethyl)-1-1[2-(1-methylethyl)-4-thiazolyl]-3,6-dioxo-8,11-bis(phenylmethyl)-2,4,7,12-tetraazatridecane-13-oic acid, 5-thiazolylmethyl ester [5S-(5S*,8R*,10R*,11R*)] and is available from Abbott Laboratories of Abbott Park, Illinois as Norvir. Ritonavir is an HIV protease inhibitor indicated for use with other antiretroviral agents in the treatment of HIV infection. Ritonavir also inhibits P450-mediated drug metabolism and the P-glycoprotein (Pgp) cellular transport system, resulting in increased concentrations of the active compound in the body.
GS-9350为由Gilead Sciences of Foster City California开发的作为药物增强剂的化合物 。GS-9350 is a compound being developed by Gilead Sciences of Foster City California as a drug enhancer.
SPI-452为由Sequoia Pharmaceuticals of Gaithersburg, Maryland开发的作为药物增强剂的化合物。SPI-452 is a compound being developed by Sequoia Pharmaceuticals of Gaithersburg, Maryland as a drug enhancer.
上述其它治疗剂在与本发明化合物组合使用时可以例如以在Physicians' DeskReference (PDR)中指明的那些量或由本领域普通技术人员确定的另外的量使用。The aforementioned additional therapeutic agents, when used in combination with the compounds of the invention, may be used, for example, in those amounts specified in the Physicians' Desk Reference (PDR) or in other amounts as determined by one of ordinary skill in the art.
在本发明的另一实施方案中,提供了一种治疗对象的HIV感染的方法,包括向该对象施用如本文中所述的组合产品。In another embodiment of the invention, there is provided a method of treating HIV infection in a subject comprising administering to the subject a combination product as described herein.
在本发明的另一实施方案中,提供了一种治愈对象的HIV感染的方法,包括向该对象施用如本文中所述的组合产品。In another embodiment of the invention, there is provided a method of curing an HIV infection in a subject comprising administering to the subject a combination product as described herein.
在本发明的另一实施方案中,提供了一种预防对象的HIV感染的方法,包括向该对象施用如本文中所述的组合产品。In another embodiment of the invention, there is provided a method of preventing HIV infection in a subject comprising administering to the subject a combination product as described herein.
在本发明的另一实施方案中,提供了如本文中所述的组合产品,其用作治疗HIV的药物。In another embodiment of the invention there is provided a combination product as described herein for use as a medicament for the treatment of HIV.
在本发明的另一实施方案中,提供了如本文中所述的组合产品,其用作预防HIV的药物。In another embodiment of the present invention there is provided a combination product as described herein for use as a medicament for the prevention of HIV.
在本发明的另一实施方案中,提供了如本文中所述的组合产品,其用作治愈HIV的药物。In another embodiment of the present invention there is provided a combination product as described herein for use as a medicament for curing HIV.
在本发明的另一实施方案中,提供了如本文中所述的组合产品,其用于治疗HIV感染。In another embodiment of the present invention there is provided a combination product as described herein for use in the treatment of HIV infection.
在本发明的另一实施方案中,提供了如本文中所述的组合产品,其用于预防HIV感染。In another embodiment of the present invention there is provided a combination product as described herein for use in the prevention of HIV infection.
在本发明的另一实施方案中,提供了如本文中所述的组合产品,其用于治愈HIV感染。In another embodiment of the present invention there is provided a combination product as described herein for use in curing HIV infection.
在本发明的另一实施方案中,提供了如本文中所述的组合产品在制备药物中的用途,所述药物用于治疗HIV感染。In another embodiment of the present invention there is provided the use of a combination product as described herein in the preparation of a medicament for the treatment of HIV infection.
在本发明的另一实施方案中,提供了如本文中所述的组合产品在制备药物中的用途,所述药物用于预防HIV感染。In another embodiment of the present invention there is provided the use of a combination product as described herein in the preparation of a medicament for the prevention of HIV infection.
在本发明的另一实施方案中,提供了如本文中所述的组合产品在制备药物中的用途,所述药物用于治愈HIV感染。In another embodiment of the present invention there is provided the use of a combination product as described herein in the preparation of a medicament for curing HIV infection.
因此,本发明提供了免疫原性组合物,其包含抗原或抗原组合物和式(I)化合物或其药学上可接受的盐。进一步提供了疫苗组合物,其包含抗原或抗原组合物和式(I)化合物或其药学上可接受的盐。Therefore, the present invention provides an immunogenic composition comprising an antigen or antigen composition and a compound of formula (I) or a pharmaceutically acceptable salt thereof. Further provided is a vaccine composition comprising an antigen or antigen composition and a compound of formula (I) or a pharmaceutically acceptable salt thereof.
式(I)化合物及其药学上可接受的盐还可以与至少一种其它治疗剂组合使用,所述治疗剂可用于预防或治疗病毒感染,例如免疫疗法(例如干扰素或其它细胞因子/趋化因子、细胞因子/趋化因子受体调节剂、细胞因子激动剂或拮抗剂和类似药剂);和治疗疫苗、抗纤维化剂、抗炎剂诸如皮质类固醇或NSAID(非甾体抗炎药剂)和类似药剂。The compounds of formula (I) and their pharmaceutically acceptable salts may also be used in combination with at least one other therapeutic agent useful for preventing or treating viral infections, such as immunotherapy (e.g., interferons or other cytokines/chemokines, cytokine/chemokine receptor modulators, cytokine agonists or antagonists, and similar agents); and therapeutic vaccines, anti-fibrotic agents, anti-inflammatory agents such as corticosteroids or NSAIDs (non-steroidal anti-inflammatory agents), and similar agents.
调节STING的化合物、特别是式(I)化合物或其药学上可接受的盐可以与其它抗炎剂组合施用,所述抗炎剂包括口服或局部皮质类固醇、抗-TNF剂、5-氨基水杨酸和美沙拉秦制剂、羟氯喹、硫代嘌呤、甲氨蝶呤、环磷酰胺、环孢菌素、钙神经素抑制剂、霉酚酸、mTOR抑制剂、JAK抑制剂、Syk抑制剂、RIPK1和RIPK2抑制剂、抗炎生物剂,包括抗-IL6生物制剂、抗-IL1剂、抗-IL17生物制剂、抗-CD22、抗-整联蛋白剂、抗-IFNa、抗CD20或CD4生物制剂和针对T细胞或B细胞受体或白介素的其它细胞因子抑制剂或生物制剂。Compounds that modulate STING, particularly compounds of formula (I) or pharmaceutically acceptable salts thereof, can be administered in combination with other anti-inflammatory agents, including oral or topical corticosteroids, anti-TNF agents, 5-aminosalicylic acid and mesalamine preparations, hydroxychloroquine, thiopurines, methotrexate, cyclophosphamide, cyclosporin, calcineurin inhibitors, mycophenolic acid, mTOR inhibitors, JAK inhibitors, Syk inhibitors, RIPK1 and RIPK2 inhibitors, anti-inflammatory biologics, including anti-IL6 biologics, anti-IL1 agents, anti-IL17 biologics, anti-CD22, anti-integrin agents, anti-IFNa, anti-CD20 or CD4 biologics, and other cytokine inhibitors or biologics directed against T cell or B cell receptors or interleukins.
例如,在系统性红斑狼疮和相关的狼疮病症的治疗中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐,可以与至少一种其它治疗剂组合施用,所述其它治疗剂包括皮质类固醇(诸如泼尼松龙(Delatsone®、Orapred、Millipred、Omnipred、Econopred、Flo-Pred)、免疫抑制剂(诸如甲氨蝶呤(Rhuematrex®、Trexall®))、地塞米松(Decadron®、Solurex®)、吗替麦考酚酯(Cellcept®)、他克莫司®、Sirolimus®)、B细胞疗法(贝利木单抗(Benlysta®)、B细胞抑制剂(Atacicept®、Apratuzumab®(抗-CD22)、SBI-087(抗-CD20)、抗-BAFF抗体(LY2127399、A623)、Velcade®)、硫唑嘌呤(Azasan®、Imuran®)、曲安西龙(Clinacort®、Kenalog-10®)、羟氯喹(Plaquenil®)、沙利度胺(Immunoprin®、Contergan®)、免疫球蛋白疗法(HyQiva®、Flebogamma®、Gamunex®、Privigen®、Gammagard®)、抗-干扰素-α疗法(Rontalizumab®、西法木单抗®、AGS-009®、IFN Kinoid)、抗-干扰素受体(IFNR) (Anifrolumab®)、TLR7和TLR9阻滞剂(IMO-3100)、抗-细胞因子疗法(抗IL6(CNTO-136)、抗-干扰素-γ(AMG811)、免疫调节疗法(Lupuzor™、阿巴西普、Orencia®、AMG557、拉喹莫德、帕喹莫德、来氟米特、抗ICOS(Medi-570)、抗-CD40配体抗体(CDP7657))和/或血小板聚集抑制剂(阿司匹林)。For example, in the treatment of systemic lupus erythematosus and related lupus disorders, compounds that modulate STING, particularly compounds of formula (I) or pharmaceutically acceptable salts thereof, can be administered in combination with at least one other therapeutic agent, including corticosteroids (such as prednisolone ( Delatsone® , Orapred, Millipred, Omnipred, Econopred, Flo-Pred), immunosuppressants (such as methotrexate ( Rhuematrex® , Trexall® )), dexamethasone ( Decadron® , Solurex® ), mycophenolate mofetil ( Cellcept® ), tacrolimus® , Sirolimus® ), B cell therapy (belimumab (Benlysta® ) , B cell inhibitors ( Atacicept® , Apratuzumab® ), (anti-CD22), SBI-087 (anti-CD20), anti-BAFF antibodies (LY2127399, A623), Velcade ® ), azathioprine (Azasan ® , Imuran ® ), triamcinolone (Clinacort ® , Kenalog-10 ® ), hydroxychloroquine (Plaquenil ® ), thalidomide (Immunoprin ® , Contergan ® ), immunoglobulin therapy (HyQiva ® , Flebogamma ® , Gamunex ® , Privigen ® , Gammagard ® ), anti-interferon-α therapy (Rontalizumab ® , sifalimumab ® , AGS-009 ® , IFN Kinoid), anti-interferon receptor (IFNR) (Anifrolumab®), TLR7 and TLR9 blockers (IMO-3100), anti-cytokine therapies (anti-IL6 (CNTO-136), anti-interferon-γ (AMG811), immunomodulatory therapies (Lupuzor™, abatacept, Orencia ® , AMG557, laquinimod, paquinimod, leflunomide, anti-ICOS (Medi-570), anti-CD40 ligand antibody (CDP7657)) and/or platelet aggregation inhibitors (aspirin).
在干燥综合征的治疗中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐,可以与以下组合施用:抗风湿剂(羟氯喹和Plaquenil®、Ridaura®、Kineret®)、胆碱能激动剂(Salagen®、Evoxac®)、JAK抑制剂(Xelijanz®和抗-TNFα治疗剂(Remicade®、Humira®、Enbrel®、Cimzia®、Simponi®)。In the treatment of Sjögren's syndrome, compounds that modulate STING, in particular compounds of formula (I) or pharmaceutically acceptable salts thereof, can be administered in combination with anti-rheumatic agents (hydroxychloroquine and Plaquenil® , Ridaura® , Kineret® ), cholinergic agonists ( Salagen® , Evoxac® ), JAK inhibitors ( Xelijanz® and anti-TNFα therapeutics (Remicade® , Humira® , Enbrel® , Cimzia® , Simponi® ).
在血管炎和具有小或中尺寸血管炎症的疾病的治疗中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐,可以与以下组合施用:烷基化试剂(环磷酰胺、Cytoxan®)、抗风湿的抗-CD20抗体(Rituxan®、利妥昔单抗®)和抗-TNFα抑制剂(Etanrcept®)。In the treatment of vasculitis and diseases with inflammation of small or medium-sized vessels, compounds that modulate STING, in particular compounds of formula (I) or pharmaceutically acceptable salts thereof, can be administered in combination with alkylating agents (cyclophosphamide, Cytoxan® ), anti-rheumatic anti-CD20 antibodies ( Rituxan® , Rituximab® ) and anti-TNFα inhibitors (Etanrcept® ) .
在牛皮癣的治疗中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐,可以与以下组合施用:伊西贝单抗、tildrakizumab、苏金单抗、阿法赛特、钙泊三醇和二丙酸倍他米松、强的松、他扎罗汀局部凝胶、甲氨蝶呤、环孢菌素、富马酸、阿维A酸、光线疗法 (UVA、UVB)、补骨脂素、煤焦油、TNF抑制剂(依那西普、英夫利昔、阿达木单抗、聚乙二醇结合赛妥珠单抗)、PDE-4抑制剂(阿普斯特)、JAK抑制剂(托法替尼)、IL 12/23 (优特克单抗)、IL17 (苏金单抗、伊西贝单抗、布罗达单抗 (AMG-827))、IL23 (tildrakizumab(MK-3222)、古塞库单抗、伊曲珠单抗、英夫利昔的生物仿制药 (Remsima (Inflectra®)、Sandoz GP 11111)、利妥昔单抗的生物仿制药 (CT-P10 (Mabthera®)、PF-05280586(MabThera®))、依那西普的生物仿制药 (CHS-2014)、阿达木单抗的生物仿制药 (GP-2017)、M-518101局部维生素D;Maruho GK-664或CT-327 (局部原肌球蛋白受体激酶A)、CF-101、苏金单抗 (AIN457)或富马酸二甲酯 LAS-41008。In the treatment of psoriasis, compounds that modulate STING, particularly compounds of formula (I) or pharmaceutically acceptable salts thereof, can be administered in combination with isibetamab, tildrakizumab, secukinumab, alefacept, calcipotriol and betamethasone dipropionate, prednisone, tazarotene topical gel, methotrexate, cyclosporine, fumaric acid, acitretin, phototherapy (UVA, UVB), psoralen, coal tar, TNF inhibitors (etanercept, infliximab, adalimumab, certolizumab pegol), PDE-4 inhibitors (apremilast), JAK inhibitors (tofacitinib), IL 12/23 (ustekinumab), IL17 (secukinumab, isibetamab, brodalumab (AMG-827)), IL23 (tildrakizumab (MK-3222), guselkumab, itrazumab, biosimilar infliximab (Remsima (Inflectra®), Sandoz GP 11111), biosimilar rituximab (CT-P10 (Mabthera®), PF-05280586 (MabThera®)), biosimilar etanercept (CHS-2014), biosimilar adalimumab (GP-2017), M-518101 topical vitamin D; Maruho GK-664 or CT-327 (topical tropomyosin receptor kinase A), CF-101, secukinumab (AIN457), or dimethyl fumarate LAS-41008.
在类风湿性关节炎的治疗中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐可以与以下组合施用:托珠单抗、DMARD(甲氨蝶呤、羟氯喹、柳氮磺胺吡啶、来氟米特)、柳氮磺胺吡啶延迟释放、聚乙二醇结合赛妥珠单抗、布洛芬、萘普生钠、阿达木单抗、阿那白滞素(Kineret);依托度酸、萘普生钠、阿巴西普、强的松、英利昔单抗、戈利木单抗、罗非考昔、托法替尼、甲氨蝶呤、选择性JAK1 & JAK2抑制剂(baracitinib)、反义寡核苷酸 (alicafosen)、英夫利昔的生物仿制药(Remsima (Inflectra®))、GS-071 英夫利昔(Aprogen)、SB2 英夫利昔、PF-06438179 英夫利昔、GP11111、 利妥昔单抗的生物仿制药(CT-P10 利妥昔单抗 Celltrion)、BI-695500、GP-2013、PF-05280586、依那西普的生物仿制药 (依那西普 SB4 (Brenzys™)、Benepali®;CHS-0214 依那西普、GP-2015、 阿达木单抗的生物仿制药 (ABP-501 阿达木单抗、BI-695501、Samsung SB5、GP-2017、 PF-06410293、Momenta M923或阿巴西普的生物仿制药 (M834)。In the treatment of rheumatoid arthritis, compounds that modulate STING, particularly compounds of formula (I) or pharmaceutically acceptable salts thereof, can be administered in combination with: tocilizumab, DMARDs (methotrexate, hydroxychloroquine, sulfasalazine, leflunomide), sulfasalazine delayed release, certolizumab pegol, ibuprofen, naproxen sodium, adalimumab, anakinra (Kineret); etodolac, naproxen sodium, abatacept, prednisone, infliximab, golimumab, rofecoxib, tofacitinib, methotrexate, selective JAK1 & JAK2 inhibitors (baracitinib), antisense oligonucleotides (alicafosen), biosimilars of infliximab (Remsima (Inflectra®)), GS-071 infliximab (Aprogen), SB2 infliximab, PF-06438179 infliximab, GP11111, Biosimilars of rituximab (CT-P10 Rituximab Celltrion), BI-695500, GP-2013, PF-05280586, biosimilars of etanercept (etanercept SB4 (Brenzys™), Benepali®; CHS-0214 etanercept, GP-2015, biosimilars of adalimumab (ABP-501 adalimumab, BI-695501, Samsung SB5, GP-2017, PF-06410293, Momenta M923), or biosimilars of abatacept (M834).
在另一个实施方案中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐可以与被认为是护理标准的至少一种其它疗法和/或至少一种其它活性治疗剂组合施用于需要其的患者(美国卫生与人类服务部,国家卫生研究和质量局,国家指南信息中心(U.S. Department of Health and Human Services, Agency for Healthcare Researchand Quality, National Guideline Clearinghouse), https://www.guideline.gov/ 和世界卫生组织(World Health Organization), http://www.who.int/management/quality/standards/en/)用于本文中所述的任何疾病和/或病症。In another embodiment, compounds that modulate STING, particularly compounds of formula (I) or pharmaceutically acceptable salts thereof, can be administered to a patient in need thereof in combination with at least one other therapy and/or at least one other active therapeutic agent that is considered standard of care (U.S. Department of Health and Human Services, Agency for Healthcare Research and Quality, National Guideline Clearinghouse, https://www.guideline.gov/ and World Health Organization, http://www.who.int/management/quality/standards/en/) for any of the diseases and/or conditions described herein.
在另一个实施方案中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐可以与至少一种其它疗法组合,例如与表明治疗牛皮癣的UVA 和/或 UVB 光线疗法组合施用于需要其的患者。In another embodiment, a compound that modulates STING, particularly a compound of formula (I) or a pharmaceutically acceptable salt thereof, can be administered to a patient in need thereof in combination with at least one other therapy, for example, in combination with UVA and/or UVB phototherapy indicated for the treatment of psoriasis.
在另一个实施方案中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐可以与用于本文中所述的适应症的至少一种其它活性治疗剂组合施用于需要其的患者,其中所述至少一种其它活性治疗剂为:皮质类固醇 [口服、局部、通过注射或作为栓剂施用;强的松、甲基强的松龙、泼尼松龙、布地奈德、倍他米松、地塞米松、氢化可的松、曲安奈德、氟替卡松 (糠酸氟替卡松、丙酸氟替卡松)、氟氢缩松 (丙酮缩氟氢羟龙、氟缩酮氢可松)、氟轻松醋酸酯、氯倍他索 (丙酸氯倍他索)]、抗-TNF生物药剂(依那西普、阿达木单抗、英夫利昔、赛妥珠单抗或戈利木单抗)、PDE-4抑制剂(阿普斯特)、5-氨基水杨酸(美沙拉秦/美沙拉明;柳氮磺胺吡啶、巴柳氮)、DMARD (调节疾病的抗风湿药:甲氨蝶呤、羟氯喹、柳氮磺胺吡啶、来氟米特)、硫嘌呤 (咪唑硫嘌呤、巯嘌呤)、JAK抑制剂(托法替尼)、NSAID (阿司匹林、对乙酰氨基酚、布洛芬、萘普生 (萘普生钠)、依托度酸、塞来昔布、双氯芬酸、美洛昔康)、抗-IL6 生物药剂 (托珠单抗)、抗-IL1生物药剂(阿那白滞素、康纳单抗、利纳西普)、抗-IL12或IL23 生物药剂 (优特克单抗、risankizumab、古塞库单抗、tildrakizumab)、抗-CD6 生物药剂 (伊曲珠单抗)、抗-整合素剂(那他珠单抗 (Tysabri®)、etrolizumab)、抗-IL17 生物药剂 (苏金单抗、伊西贝单抗、布罗达单抗)、抗-CD22 生物药剂 (依帕珠单抗)、抗-CD20 生物药剂 (利妥昔单抗、奥法木单抗)、抗-CD20或CD4 生物药剂和其它细胞因子抑制剂或生物T-细胞或B-细胞受体或白介素、钙调磷酸酶抑制剂(环孢菌素、吡美莫司、他克莫司)、阿维A酸、富马酸、富马酸二甲酯、环磷酰胺、环孢菌素(或环孢素)、甲氨蝶呤、霉酚酸 (或吗替麦考酚酯)、局部维生素D (卡泊三醇或钙泊三醇)、mTOR抑制剂(替西罗莫司、依维莫司)、Syk抑制剂(福坦替尼)、抗-IFNa 生物药剂 (西法木单抗)或类维生素A(他扎罗汀)。其它合适的生物药剂的实例包括阿巴西普、贝利木单抗和alicafosen。In another embodiment, compounds that modulate STING, particularly compounds of formula (I) or pharmaceutically acceptable salts thereof, can be administered to a patient in need thereof in combination with at least one other active therapeutic agent for an indication described herein, wherein the at least one other active therapeutic agent is: corticosteroids [orally, topically, by injection or as a suppository; prednisone, methylprednisolone, prednisolone, budesonide, betamethasone, dexamethasone, hydrocortisone, triamcinolone acetonide, fluticasone (fluticasone furoate, fluticasone propionate), flurandrenolide (flurane acetonide, flurandrenolide), fluocinolone acetonide, clobetasol (clobetasol propionate)], anti-TNF biologics (etanercept, adalimumab, infliximab, certolizumab or golimumab), PDE-4 inhibitors (apremilast), 5-aminosalicylic acids (mesalamine/mesalamine; sulfasalazine, balsalazide), DMARDs (Disease-modifying antirheumatic drugs: methotrexate, hydroxychloroquine, sulfasalazine, leflunomide), thiopurines (azathioprine, mercaptopurine), JAK inhibitors (tofacitinib), NSAIDs (aspirin, acetaminophen, ibuprofen, naproxen (naproxen sodium), etodolac, celecoxib, diclofenac, meloxicam), anti-IL6 biologics (tocilizumab), anti-IL1 biologics (anakinra, canakinumab, rilonacept), anti-IL12 or IL23 biologics (ustekinumab, risankizumab, guselkumab, tildrakizumab), anti-CD6 biologics (itrazumab), anti-integrin agents (natalizumab (Tysabri®), etrolizumab), anti-IL17 biologics (secukinumab, isibetamab, brodalumab), anti-CD22 biologics (epazumab), anti-CD20 biologics (rituximab, ofatumumab), anti-CD20 or CD4 biologics and other cytokine inhibitors or biologics T-cell or B-cell receptors or interleukins, calcineurin inhibitors (cyclosporine, pimecrolimus, tacrolimus), acitretin, fumaric acid, dimethyl fumarate, cyclophosphamide, cyclosporine (or cyclosporine), methotrexate, mycophenolic acid (or mycophenolate mofetil), topical vitamin D (calcipotriol or calcipotriol), mTOR inhibitors (temsirolimus, everolimus), Syk inhibitors (fotantinib), anti-IFNa biologics (sifalimumab) or retinoids (tazarotene). Other examples of suitable biologics include abatacept, belimumab and alicafosen.
在本发明的一个实施方案中,至少一种其它治疗剂选自吸入性皮质类固醇、长效β激动剂、吸入性皮质类固醇和长效β激动剂的组合、短效β激动剂、白三烯调节剂、抗-IgE、甲基黄嘌呤支气管扩张剂、肥大细胞抑制剂和长效毒蕈碱拮抗剂。例如,在哮喘的治疗中,抑制STING的化合物,特别是式(I)化合物或其药学上可接受的盐,可以与以下组合施用:吸入性皮质类固醇(ICS)诸如丙酸氟替卡松(Flovent®)、丙酸倍氯米松(QVAR®)、布地奈德(Pulmicort)、醋酸曲安奈德(trimcinolone acetonide)(Azmacort®)、氟尼缩松(Aerobid®)、糠酸莫米松(Asmanex® Twisthaler®)或环索奈德(Alvesco®))、长效β激动剂(LABA)诸如富马酸福莫特罗(Foradil®)、昔萘酸沙美特罗(Serevent®))、ICS和LABA的组合(诸如糠酸氟替卡松和维兰特罗((Breo Ellipta®))、福莫特罗/布地奈德吸入剂(Symbicort®)、丙酸倍氯米松/福莫特罗(Inuvair®)和丙酸氟替卡松/沙美特罗(Advair®)、短效β激动剂((SABA)诸如硫酸沙丁胺醇(ProAir®、Proventil HFA®、Ventolin HFA®、AccuNeb®吸入溶液)、酒石酸左旋沙丁胺醇(Xopenex® HFA)、异丙托溴铵/沙丁胺醇(Combivent® Respimat®)、异丙托溴铵(Atrovent® HFA)、白三烯调节剂(诸如孟鲁司特钠(Singulair®)、扎鲁司特(Accolate®)或齐留通(Zyflo®)以及抗-IgE(诸如奥马珠单抗(Xolair®))、甲基黄嘌呤支气管扩张药(诸如茶碱(Accurbron®、Aerolate®、Aquaphyllin®、Asbron®、Bronkodyl®、Duraphyl®、Elixicon®、Elixomin®、Elixophyllin®、Labid®、Lanophyllin®、Quibron-T®、Slo-Bid®、Slo-Phyllin®、Somophyllin®、Sustaire®、Synophylate®、T-Phyll®、Theo-24®、Theo-Dur®、Theobid®、Theochron®、Theoclear®、Theolair®、Theolixir®、Theophyl®、Theovent®、Uni-dur®、Uniphyl®)、肥大细胞抑制剂(诸如cromulyn sodium (Nasalcrom®)和奈多罗米钠(Tilade®))、长效毒蕈碱拮抗剂((LAMA)诸如糠酸莫米他松/富马酸福莫特罗二水合物(Dulera®))。In one embodiment of the invention, the at least one additional therapeutic agent is selected from an inhaled corticosteroid, a long-acting beta agonist, a combination of an inhaled corticosteroid and a long-acting beta agonist, a short-acting beta agonist, a leukotriene modifier, an anti-IgE, a methylxanthine bronchodilator, a mast cell inhibitor, and a long-acting muscarinic antagonist. For example, in the treatment of asthma, compounds that inhibit STING, particularly compounds of formula (I) or pharmaceutically acceptable salts thereof, may be administered in combination with an inhaled corticosteroid (ICS) such as fluticasone propionate ( Flovent® ), beclomethasone propionate ( QVAR® ), budesonide ( Pulmicort ), trimcinolone acetonide (Azmacort®), flunisolide ( Aerobid® ), mometasone furoate ( Asmanex® Twisthaler® ) or ciclesonide ( Alvesco® ), a long-acting beta agonist (LABA) such as formoterol fumarate ( Foradil® ), salmeterol xinafoate ( Serevent® ), a combination of ICS and a LABA such as fluticasone furoate and vilanterol (Breo Ellipta® ), a formoterol/budesonide inhaler (Symbicort®), or a combination of an inhaled corticosteroid ( ICS) . ), beclomethasone propionate/formoterol (Inuvair ® ) and fluticasone propionate/salmeterol (Advair ® ), short-acting beta agonists (SABAs) such as albuterol sulfate (ProAir ® , Proventil HFA ® , Ventolin HFA ® , AccuNeb ® inhalation solution), levosalbutamol tartrate (Xopenex ® HFA), ipratropium bromide/albuterol (Combivent ® Respimat ® ), ipratropium bromide (Atrovent ® HFA), leukotriene modifiers such as montelukast sodium (Singulair ® ), zafirlukast (Accolate ® ), or zileuton (Zyflo ® ) and anti-IgE such as omalizumab (Xolair ® )), methylxanthine bronchodilators such as theophylline (Accurbron ® , Aerolate ® , Aquaphyllin ® , Asbron ® , Bronkodyl ® , Duraphyl ® , Elixicon ® , Elixomin ® , Elixophyllin ® , Labid ® , Lanophyllin ® , Quibron-T ® , Slo-Bid ® , Slo-Phyllin ® , Somophyllin ® , Sustaire ® , Synophyllin late ® , T-Phyll ® , Theo-24 ® , Theo-Dur ® , Theobid ® , Theochron ® , Theoclear ® , Theolair ® , Theolixir ® , Theophyl ® , Theovent ® , Uni-dur ® , Uniphyl ® ), mast cell inhibitors such as cromulyn sodium (Nasalcrom ® ) and nedocromil sodium (Ti lade ® )), long-acting muscarinic antagonists (LAMA) such as mometasone furoate/formoterol fumarate dihydrate (Dulera ® )).
可能适用于治疗哮喘的组合疗法中的其它药剂包括蛋白酪氨酸激酶抑制剂(马赛替尼)、CRTH2/D-前列腺素类受体拮抗剂(AMG 853)、茚达特罗(Arcapta®Neohaler®)、肾上腺素吸入气雾剂(E004)、糠酸氟替卡松/丙酸氟替卡松、vinanterol吸入/糠酸氟替卡松粉剂(Relovair™)、丙酸氟替卡松/富马酸福莫特罗脱水物(Flutiform®)、瑞利珠单抗、沙丁胺醇干粉吸入剂、噻托溴铵(Spiriva®HandiHaler®)、福莫特罗/布地奈德(Symbicort®SMART®)、糠酸氟替卡松(Veramyst®)、Vectura的VR506、来金珠单抗(RG3637)、磷酸二酯酶(PDE)-3和(PDE)-4抑制剂的组合(RPL554)。Other agents that may be useful in combination therapies for the treatment of asthma include protein tyrosine kinase inhibitors (masitinib), CRTH2/D-prostanoid receptor antagonists (AMG 853), indacaterol (Arcapta ® Neohaler ® ), epinephrine inhalation aerosol (E004), fluticasone furoate/fluticasone propionate, vinanterol inhalation/fluticasone furoate powder (Relovair™), fluticasone propionate/formoterol fumarate dehydrate (Flutiform ® ), reslizumab, albuterol dry powder inhaler, tiotropium (Spiriva ® HandiHaler ® ), formoterol/budesonide (Symbicort ® SMART ® ), fluticasone furoate (Veramyst ® ), Vectura's VR506, levitra (RG3637), and a combination of phosphodiesterase (PDE)-3 and (PDE)-4 inhibitors (RPL554).
在本发明的一个实施方案中,至少一种其它治疗剂选自长效β激动剂,长效吸入抗胆碱能药或毒蕈碱拮抗剂,磷酸二酯酶抑制剂,吸入皮质类固醇长效β激动剂、短效β激动剂和吸入皮质类固醇的组合。例如,在COPD的治疗中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐,可以与以下组合施用:LABA(诸如昔萘酸沙美特罗(Serevent)、芜地溴铵/维兰特罗(Anoro Ellipta®)、芜地溴铵(Incruse Ellipta®)、酒石酸福莫特罗(Brovana®)、富马酸福莫特罗吸入粉剂(Foradil®)、马来酸茚达特罗(Arcapta® Neohaler®)或丙酸氟替卡松/富马酸福莫特罗脱水物(Flutiform®))、长效吸入抗胆碱能药(或毒蕈碱拮抗剂,诸如噻托溴铵(Spiriva®)和阿地溴铵(Tudorza® Pressair®)、磷酸二酯酶(PDE-r)抑制剂(诸如罗氟司特、Daliresp®)、组合ICS/LABA(诸如糠酸氟替卡松和维兰特罗(BreoEllipta®)、丙酸氟替卡松/沙美特罗(Advair®)、布地奈德/福莫特罗(Symbicort®)、莫米松/福莫特罗(Dulera®)、异丙托溴铵/硫酸沙丁胺醇(Duoneb®、Atrovent®)、沙丁胺醇/异丙托铵(Combivent Respimat®))、SABA(诸如异丙托溴铵(Atrovent®)和硫酸沙丁胺醇(ProAir®、Proventil®))和ICS (诸如布地奈德(Pulmicort®)和丙酸氟替卡松(Flovent®)、二丙酸倍氯米松(QVAR®)。In one embodiment of the invention, the at least one additional therapeutic agent is selected from a long-acting beta agonist, a long-acting inhaled anticholinergic or muscarinic antagonist, a phosphodiesterase inhibitor, an inhaled corticosteroid, a combination of a long-acting beta agonist, a short-acting beta agonist, and an inhaled corticosteroid. For example, in the treatment of COPD, compounds that modulate STING, particularly compounds of formula (I) or pharmaceutically acceptable salts thereof, can be administered in combination with a LABA (such as salmeterol xinafoate (Serevent), umeclidinium/vilanterol (Anoro Ellipta® ), umeclidinium (Incruse Ellipta® ), formoterol tartrate (Brovana®), formoterol fumarate inhalation powder ( Foradil® ), indacaterol maleate ( Arcapta® Neohaler® ), or fluticasone propionate/formoterol fumarate dehydrate (Flutiform®)), a long-acting inhaled anticholinergic (or muscarinic antagonist, such as tiotropium ( Spiriva® ) and aclidinium ( Tudorza® Pressair® ), a phosphodiesterase (PDE-r) inhibitor (such as roflumilast, Daliresp® ) , or a combination thereof. ), combination ICS/LABAs (such as fluticasone furoate and vilanterol (BreoEllipta ® ), fluticasone propionate/salmeterol (Advair ® ), budesonide/formoterol (Symbicort ® ), mometasone/formoterol (Dulera ® ), ipratropium/albuterol sulfate (Duoneb ® , Atrovent ® ), albuterol/ipratropium (Combivent Respimat ® )), SABAs (such as ipratropium (Atrovent ® ) and albuterol sulfate (ProAir ® , Proventil ® )), and ICS (such as budesonide (Pulmicort ® ) and fluticasone propionate (Flovent ® ), beclomethasone dipropionate (QVAR ® ).
可能适用于治疗COPD的组合疗法中的其它药剂包括SCH527123(CXCR2拮抗剂)、格隆溴铵((NVA237) Seebri® Breezhaler®)、格隆溴铵和马来酸茚达特罗((QVA149)Ultibro® Breezhaler®)、格隆溴铵和富马酸福莫特罗(PT003)、马来酸茚达特罗(QVA149)、奥达特罗(Striverdi® Respimat®)、噻托溴铵(Spiriva®)/奥达特罗(Striverdi®Respimat®)和阿地铵/福莫特罗吸入剂。Other agents that may be useful in combination therapies for treating COPD include SCH527123 (a CXCR2 antagonist), glycopyrrolate ((NVA237) Seebri ® Breezhaler ® ), glycopyrrolate and indacaterol maleate ((QVA149) Ultibro ® Breezhaler ® ), glycopyrrolate and formoterol fumarate (PT003), indacaterol maleate (QVA149), olodaterol (Striverdi ® Respimat ® ), tiotropium (Spiriva ® )/olodaterol (Striverdi ® Respimat ® ), and aclidinium/formoterol inhaler.
在本发明的一个实施方案中,至少一种其它治疗剂选自口服皮质类固醇、抗胸腺细胞球蛋白、沙利度胺、苯丁酸氮芥、钙通道阻滞剂、局部润肤剂、ACE抑制剂、5-羟色胺再摄取抑制剂、内皮素-1受体抑制剂、抗纤维化剂、质子泵抑制剂或伊马替尼、ARG201和托珠单抗。例如,在系统性硬皮病的治疗中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐,可以与以下组合施用:口服皮质类固醇(诸如泼尼松龙(Delatsone®、Orapred、Millipred、Omnipred、Econopred、Flo-Pred)、免疫抑制剂(诸如甲氨蝶呤(Rhuematrex®、Trexall®)、环孢菌素(Sandimmune®)、抗胸腺细胞球蛋白(Atgam®)、霉酚酸酯(CellCept®)、环磷酰胺(Cytoxan®)、FK506(他克莫司)、沙利度胺(Thalomid®)、苯丁酸氮芥(Leukeran®)、硫唑嘌呤(Imuran®、Azasan®))、钙通道阻滞剂(诸如硝苯地平(Procardia®、Adalat®)或尼卡地平(Cardene®)、局部润肤剂(硝酸甘油软膏剂)、ACE抑制剂(诸如赖诺普利(Zestril®、Prinivil®)、地尔硫卓(Cardizem®、Cardizem SR®、Cardizem CD®、Cardia®、Dilacor®、Tiazac®))、5-羟色胺再摄取抑制剂(诸如氟西汀(Prozac®))、内皮素-1受体抑制剂(诸如波生坦(Tracleer®)或依前列醇(Flolan®、Veletri®、Prostacyclin®))抗纤维化剂(诸如秋水仙碱(Colcrys®)、对氨基苯甲酸(PABA)、二甲基亚砜(DMSO)和D-青霉胺(Cuprimine®、Depen®)、干扰素α和干扰素γ(INF-g))、质子泵抑制剂(诸如奥美拉唑(Prilosec®)、甲氧氯普胺(Reglan®)、兰索拉唑(Prevacid®)、艾美拉唑(Nexium®)、泮托拉唑(Protonix®)、雷贝拉唑(Aciphex®))或伊马替尼(Gleevec®) ARG201 (arGentis Pharmaceutical)、贝利木单抗(Benlysta®)、托珠单抗(Actema®)。In one embodiment of the invention, the at least one additional therapeutic agent is selected from oral corticosteroids, antithymocyte globulin, thalidomide, chlorambucil, calcium channel blockers, topical emollients, ACE inhibitors, serotonin reuptake inhibitors, endothelin-1 receptor inhibitors, antifibrotic agents, proton pump inhibitors or imatinib, ARG201 and tocilizumab. For example, in the treatment of systemic scleroderma, compounds that modulate STING, in particular compounds of formula (I) or pharmaceutically acceptable salts thereof, can be administered in combination with oral corticosteroids (such as prednisolone ( Delatsone® , Orapred, Millipred, Omnipred, Econopred, Flo-Pred), immunosuppressants (such as methotrexate ( Rhuematrex® , Trexall® ), cyclosporine ( Sandimmune® ), antithymocyte globulin ( Atgam® ), mycophenolate mofetil (CellCept®), cyclophosphamide ( Cytoxan® ), FK506 (tacrolimus), thalidomide ( Thalomid® ), chlorambucil (Leukeran®), azathioprine ( Imuran® , Azasan® )), calcium channel blockers (such as nifedipine (Procardia®, Adalat®), tacrolimus (Tacrolimus® ), thalidomide ( Thalomid® ), chlorambucil (Leukeran®), azathioprine (Imuran®, Azasan®), calcium channel blockers (such as nifedipine ( Procardia® , Adalat® ), tacrolimus (Tacrolimus ... ) or nicardipine (Cardene ® ), topical emollients (nitroglycerin ointment), ACE inhibitors (such as lisinopril (Zestril ® , Prinivil ® ), diltiazem (Cardizem ® , Cardizem SR ® , Cardizem CD ® , Cardia ® , Dilacor ® , Tiazac ® )), serotonin reuptake inhibitors (such as fluoxetine (Prozac ® )), endothelin-1 receptor inhibitors (such as bosentan (Tracleer ® ) or epoprostenol (Flolan ® , Veletri ® , Prostacyclin ® )), antifibrotic agents (such as colchicine (Colcrys ® ), para-aminobenzoic acid (PABA), dimethyl sulfoxide (DMSO), and D-penicillamine (Cuprimine ® , Depen ® ), interferon alpha and interferon gamma (INF-g), proton pump inhibitors (such as omeprazole (Prilosec ® ), metoclopramide (Reglan ® ), lansoprazole (Prevacid ® ), esomeprazole (Nexium ® ), pantoprazole (Protonix ® ), rabeprazole (Aciphex ® )) or imatinib (Gleevec ® ) ARG201 (arGentis Pharmaceutical), belimumab (Benlysta ® ), tocilizumab (Actema ® ).
在本发明的一个实施方案中,至少一种其它治疗剂是睫状神经营养生长因子或基因转移剂。例如,在色素性视网膜炎的治疗中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐,可以与以下组合施用:睫状神经营养生长因子(NT-501-CNTF)或基因转移剂UshStat®。In one embodiment of the invention, at least one other therapeutic agent is ciliary neurotrophic growth factor or a gene transfer agent. For example, in the treatment of retinitis pigmentosa, a compound that modulates STING, particularly a compound of formula (I) or a pharmaceutically acceptable salt thereof, can be administered in combination with ciliary neurotrophic growth factor (NT-501-CNTF) or a gene transfer agent UshStat® .
在本发明的一个实施方案中,至少一种其它治疗剂选自三价(IIV3)灭活流感疫苗、四价(IIV4)灭活流感疫苗、三价重组流感疫苗、四价活减毒流感疫苗、抗病毒剂或灭活流感疫苗。例如,在流感的治疗中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐,可以与以下组合施用:三价(IIV3)灭活流感疫苗(诸如Afluria®、Fluarix®、Flucelvax®、FluLaval®、Fluvirin®、Fluzone®)、四价(IIV4)灭活流感疫苗(诸如Fluarix®四价疫苗、Flulaval®四价疫苗、Fluzone®四价疫苗)、三价重组流感疫苗(诸如FluBlok®)、四价活减毒流感疫苗(诸如FluMist®四价疫苗)、抗病毒剂(诸如奥司他韦(Tamiflu®)、扎那米韦(Relenza®)、金刚乙胺(Flumadine®)或金刚烷胺(Symmetrel®))或Fluad®、Fludase、FluNhance®、Preflucel或VaxiGrip®。In one embodiment of the invention, the at least one additional therapeutic agent is selected from a trivalent (IIV3) inactivated influenza vaccine, a quadrivalent (IIV4) inactivated influenza vaccine, a trivalent recombinant influenza vaccine, a quadrivalent live attenuated influenza vaccine, an antiviral agent or an inactivated influenza vaccine. For example, in the treatment of influenza, a compound that modulates STING, in particular a compound of formula (I ) or a pharmaceutically acceptable salt thereof, can be administered in combination with a trivalent (IIV3) inactivated influenza vaccine (such as Afluria® , Fluarix® , Flucelvax® , FluLaval® , Fluvirin® , Fluzone® ), a quadrivalent (IIV4) inactivated influenza vaccine (such as Fluarix®, Flulaval® , Fluzone® ), a trivalent recombinant influenza vaccine (such as FluBlok® ), a quadrivalent live attenuated influenza vaccine (such as FluMist® ), an antiviral agent (such as oseltamivir ( Tamiflu® ), zanamivir ( Relenza® ), rimantadine ( Flumadine® ) or amantadine (Symmetrel® ) ) or Fluad® , Fludase®, FluNhance® . , Preflucel or VaxiGrip ® .
在葡萄球菌感染的治疗中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐,可以与以下组合施用:抗生素(诸如β-内酰胺头孢菌素(Duricef®、Kefzol®、Ancef®、Biocef®等)、萘夫西林(Unipen®)、磺胺(磺胺甲基异噁唑和甲氧苄啶(Bacrim®、Septra®,)、柳氮磺吡啶(Azulfidine®)、乙酰磺胺异噁唑(Gantrisin®)等)或万古霉素(Vancocin®))。In the treatment of staphylococcal infections, compounds that modulate STING, in particular compounds of formula (I) or pharmaceutically acceptable salts thereof, can be administered in combination with antibiotics (such as β-lactam cephalosporins ( Duricef® , Kefzol® , Ancef®, Biocef® , etc.), nafcillin ( Unipen® ), sulfonamides (sulfamethoxazole and trimethoprim (Bacrim®, Septra® , ) , sulfasalazine ( Azulfidine® ), acetylsulfisoxazole (Gantrisin®), etc.) or vancomycin ( Vancocin® ) ).
在本发明的一个实施方案中,至少一种其它治疗剂选自局部免疫调节剂或钙调磷酸酶抑制剂、局部皮质类固醇、口服皮质类固醇、干扰素γ、抗组胺药或抗生素。例如,在特应性皮炎的治疗中,调节STING的化合物,特别是式(I)化合物或其药学上可接受的盐,可以与以下组合施用:局部免疫调节剂或钙调磷酸酶抑制剂(诸如吡美莫司(Elidel®)或他克莫司软膏剂(Protopic®))、局部皮质类固醇(诸如氢化可的松(Synacort®、Westcort®)、倍他米松(Diprolene®)、丙酮缩氟氢羟龙(Cordan®)、氟替卡松(Cutivate®)、曲安西龙(Kenalog®)、醋酸氟轻松(Lidex®)和氯倍他索(Temovate®))、口服皮质类固醇(诸如氢化可的松(Cortef®)、甲基强的松龙(Medrol®)或泼尼松龙(Pediapred®、Prelone®)、免疫抑制剂(诸如环孢菌素(Neoral®)或干扰素γ(Alferon N®、Infergen®、Intron A、Roferon-A®))、抗组胺药(用于瘙痒,诸如Atarax®、Vistaril®、Benadryl®)、抗生素(诸如青霉素衍生物氟氯西林(Floxapen®)或双氯西林(Dynapen®)、红霉素(Eryc®、T-Stat®、Erythra-Derm®等))、非甾体免疫抑制剂(诸如硫唑嘌呤(Imuran®、Azasan®)、甲氨蝶呤(Rhuematrex®、Trexall®)、环孢菌素(Sandimmune®)或霉酚酸酯(CellCept®))。In one embodiment of the invention, the at least one additional therapeutic agent is selected from a topical immunomodulator or calcineurin inhibitor, a topical corticosteroid, an oral corticosteroid, interferon gamma, an antihistamine, or an antibiotic. For example, in the treatment of atopic dermatitis, compounds that modulate STING, particularly compounds of formula (I) or pharmaceutically acceptable salts thereof, may be administered in combination with a topical immunomodulator or calcineurin inhibitor such as pimecrolimus ( Elidel® ) or tacrolimus ointment ( Protopic® ), a topical corticosteroid such as hydrocortisone ( Synacort® , Westcort® ), betamethasone ( Diprolene® ), flurandrenolide (Cordan®), fluticasone (Cutivate®), triamcinolone (Kenalog® ) , fluocinolone acetonide ( Lidex® ) and clobetasol ( Temovate® ), an oral corticosteroid such as hydrocortisone ( Cortef® ), methylprednisolone ( Medrol® ) or prednisolone ( Pediapred® , Prelone® ). ), immunosuppressants (such as cyclosporine (Neoral ® ) or interferon gamma (Alferon N ® , Infergen ® , Intron A, Roferon-A ® )), antihistamines (for itching, such as Atarax ® , Vistaril ® , Benadryl ® ), antibiotics (such as the penicillin derivatives flucloxacillin (Floxapen ® ) or dicloxacillin (Dynapen ® ), erythromycin (Eryc ® , T-Stat ® , Erythra-Derm ® , etc.)), nonsteroidal immunosuppressants (such as azathioprine (Imuran ® , Azasan ® ), methotrexate (Rhuematrex ® , Trexall ® ), cyclosporine (Sandimmune ® ) or mycophenolate mofetil (CellCept ® )).
在帕金森氏病的治疗中,本发明化合物可以与基于L-多巴胺的疗法(卡比多巴(Lodosyn)-左旋多巴)、多巴胺激动剂如普拉克索 (Mrapex)、罗匹尼罗 (Requip)、罗替戈汀 (Neupro)和阿扑吗啡 (Apokyn)、单胺氧化酶(MAO) B抑制剂如司来吉兰(Eldepryl、Zelapar)、雷沙吉兰(Azilect)和沙芬酰胺(Xadago))、儿茶酚O-甲基转移酶(COMT)抑制剂恩他卡朋(Comtan)和托卡朋(Tasmar)、抗胆碱能药物如苯托品(Cogentin)或三己酚油和金刚烷胺组合施用。式(I)化合物或其药学上可接受的盐可以与将电脉冲递送至脑并降低称为深部脑刺激(DBS)的帕金森氏病症状的植入患者中的设备组合施用。In the treatment of Parkinson's disease, the compounds of the invention may be administered in combination with L-dopamine based therapies (carbidopa (Lodosyn) - levodopa), dopamine agonists such as pramipexole (Mrapex), ropinirole (Requip), rotigotine (Neupro) and apomorphine (Apokyn), monoamine oxidase (MAO) B inhibitors such as selegiline (Eldepryl, Zelapar), rasagiline (Azilect) and safinamide (Xadago)), catechol O-methyltransferase (COMT) inhibitors entacapone (Comtan) and tolcapone (Tasmar), anticholinergics such as benztropine (Cogentin) or trihexylphenol oil and amantadine. The compounds of formula (I) or pharmaceutically acceptable salts thereof may be administered in combination with a device implanted in a patient that delivers electrical pulses to the brain and reduces Parkinson's disease symptoms known as deep brain stimulation (DBS).
在心肌梗死的治疗中,本发明化合物可以与抗-IL1β抗体疗法(例如康纳单抗)组合施用。In the treatment of myocardial infarction, compounds of the invention may be administered in combination with anti-IL1β antibody therapy (eg, canakinumab).
本发明化合物也可以作为佐剂与疫苗一起配制以调节其活性。这样的组合物可以含有抗体(诸抗体)或抗体片段(诸抗体片段)或抗原组分,包括但不限于蛋白、DNA、活或死细菌和/或病毒或病毒样颗粒,以及一种或多种具有佐剂活性的组分,包括但不限于铝盐、油和水乳剂、热休克蛋白、脂质A制剂和衍生物、糖脂、其它TLR激动剂诸如CpG DNA或类似药剂、细胞因子诸如GM-CSF或IL-12或类似药剂。The compounds of the invention may also be formulated as adjuvants with vaccines to modulate their activity. Such compositions may contain antibody (antibodies) or antibody fragment (antibody fragments) or antigenic components, including but not limited to proteins, DNA, live or dead bacteria and/or viruses or virus-like particles, and one or more components with adjuvant activity, including but not limited to aluminum salts, oil and water emulsions, heat shock proteins, lipid A preparations and derivatives, glycolipids, other TLR agonists such as CpG DNA or similar agents, cytokines such as GM-CSF or IL-12 or similar agents.
在本发明的一个进一步方面,提供了疫苗佐剂,其包含式(I)化合物或其药学上可接受的盐。进一步提供了疫苗组合物,其包含(I)化合物或其药学上可接受的盐和抗原或抗原组合物。In a further aspect of the present invention, a vaccine adjuvant is provided, which comprises a compound of formula (I) or a pharmaceutically acceptable salt thereof. A vaccine composition is further provided, which comprises a compound of formula (I) or a pharmaceutically acceptable salt thereof and an antigen or an antigen composition.
治疗上“有效量”旨在意指当施用于需要这样的治疗的患者时足以有效治疗或预防的化合物的量,如本文所定义。因此,例如,式(I)化合物或其药学上可接受的盐的治疗有效量是:当施用于有需要的人时足以调节STING的活性、使得该活性介导的疾病病况被降低、减缓或预防的本发明药剂的量。对应于这样的量的给定化合物的量将根据诸如以下的因素而改变:具体化合物(例如,具体化合物的功效(pIC50)、效力(EC50)和生物半衰期)、疾病病况及其严重程度、需要治疗的患者的身份(例如,年龄、体型(size)和体重),但本领域技术人员仍可以按常规决定。同样地,所述化合物的治疗持续时间和施用的时间段(剂量之间的时间段和剂量的时机,例如,餐前/随餐/餐后)将根据需要治疗的哺乳动物的身份(例如,体重)、具体化合物及其特性(例如,药物代谢动力学特性)、疾病或病症及其严重程度和所使用的特定组合物及方法而变化,但本领域技术人员仍可以决定。A therapeutically "effective amount" is intended to mean an amount of a compound sufficient to effectively treat or prevent, as defined herein, when administered to a patient in need of such treatment. Thus, for example, a therapeutically effective amount of a compound of formula (I) or a pharmaceutically acceptable salt thereof is an amount of an agent of the invention sufficient to modulate the activity of STING such that a disease condition mediated by that activity is reduced, mitigated, or prevented when administered to a human in need thereof. The amount of a given compound corresponding to such an amount will vary depending on factors such as the specific compound (e.g., the efficacy ( pIC50 ), potency ( EC50 ), and biological half-life of the specific compound), the disease condition and its severity, the identity of the patient in need of treatment (e.g., age, size, and weight), but can still be determined routinely by one skilled in the art. Likewise, the duration of treatment with the compound and the time period of administration (time period between doses and timing of doses, e.g., before/with/after meals) will vary depending on the identity of the mammal to be treated (e.g., body weight), the specific compound and its properties (e.g., pharmacokinetic properties), the disease or condition and its severity, and the particular composition and method used, but can be determined by one skilled in the art.
“治疗”(“treat”或“treatment”)旨在意指患者中的疾病或病症的至少减轻。减轻疾病或病症的治疗方法包括以任何常规可接受方式使用本发明中的化合物,例如用于阻滞、治疗或治愈如上文所述的STING-介导的疾病或病症。在一个实施方案中,关于癌症的“治疗” (“treat”,“treating”或“treatment”)指在先前患病或诊断的患者或对象中减轻癌症,消除或减少癌症的一种或多种症状,减缓或消除癌症的进展,和延迟病况的再次发生。"Treat" or "treatment" is intended to mean at least amelioration of a disease or condition in a patient. Methods of treatment for amelioration of a disease or condition include use of the compounds of the invention in any conventionally acceptable manner, e.g., for blocking, treating, or curing a STING-mediated disease or condition as described above. In one embodiment, "treat," "treating," or "treatment" with respect to cancer refers to amelioration of cancer, elimination or reduction of one or more symptoms of cancer, slowing or eliminating progression of cancer, and delaying recurrence of the condition in a patient or subject previously ill or diagnosed with the disease.
“预防” (“prevent”,“prventing”或“prventment”)指预防性施用药物以减少疾病或其生物表现的发作的可能性或延迟疾病或其生物表现的发作。预防性治疗是适当的,例如,当对象被认为处于发展癌症的高风险中时,诸如当对象具有强的癌症家族史时或当对象已经暴露于致癌物时。"Prevent," "prventing," or "preventment" refers to the prophylactic administration of a drug to reduce the likelihood of onset of a disease or its biological manifestation or to delay the onset of a disease or its biological manifestation. Prophylactic treatment is appropriate, for example, when a subject is considered to be at high risk for developing cancer, such as when the subject has a strong family history of cancer or when the subject has been exposed to a carcinogen.
本发明化合物可以通过任何合适的施用途径施用,包括全身施用和局部施用。全身施用包括口服施用、肠胃外施用、透皮施用、直肠施用和通过吸入施用。肠胃外施用指除了肠内、透皮或通过吸入以外的施用途径,并通常通过注射或输注。肠胃外施用包括静脉内、肌肉内和皮下注射或输注。吸入指施用至患者的肺部,无论是通过嘴或是通过鼻道吸入。局部施用包括应用至皮肤。The compounds of the present invention can be administered by any suitable route of administration, including systemic administration and topical administration. Systemic administration includes oral administration, parenteral administration, transdermal administration, rectal administration, and administration by inhalation. Parenteral administration refers to routes of administration other than enteral, transdermal, or by inhalation, and is typically by injection or infusion. Parenteral administration includes intravenous, intramuscular, and subcutaneous injection or infusion. Inhalation refers to administration to the patient's lungs, whether by mouth or by inhalation through the nasal passages. Topical administration includes application to the skin.
除了上述适合于治疗肿瘤的施用途径以外,药物组合物可以适于通过肿瘤内或肿瘤旁注射施用。预期将本发明化合物肿瘤内或肿瘤旁直接注射到单个实体瘤中或其附近引发可在整个身体中攻击和破坏癌细胞的免疫应答,实质上减少并在一些情况下永久地消除来自患有疾病的患者的肿瘤。以该方式活化免疫系统以杀死远端部位的肿瘤通常被称为伴随远隔效应,并且已经用多种治疗方式在动物中得到证实(van der Jeught,等人, Oncotarget, 2015, 6(3), 1359-1381)。局部或肿瘤内或肿瘤旁施用的一个进一步优点是以低得多的剂量实现相同的效力的能力,因此最小化或消除在高得多的全身剂量下可观察到的不良事件(Marabelle, A.,等人, Clinical Cancer Research, 2014, 20(7),p1747-1756)。In addition to the above-mentioned route of administration suitable for treating tumors, the pharmaceutical composition may be suitable for administration by intratumoral or paratumoral injection. It is expected that the compound of the present invention will be directly injected into a single solid tumor or near a tumor to trigger an immune response that can attack and destroy cancer cells in the whole body, substantially reducing and permanently eliminating tumors from patients with diseases in some cases. Activating the immune system in this way to kill tumors at distal sites is generally referred to as accompanying abscopal effects, and has been confirmed in animals with a variety of treatments ( van der Jeught, et al., Oncotarget, 2015, 6 (3), 1359-1381 ). A further advantage of local or intratumoral or paratumoral administration is the ability to achieve the same effect at a much lower dose, thereby minimizing or eliminating adverse events observable at much higher systemic doses (Marabelle, A., et al., Clinical Cancer Research, 2014, 20 (7), p1747-1756).
本发明化合物可以经由滴眼剂施用以治疗干燥综合征。The compounds of the present invention may be administered via eye drops for the treatment of Sjögren's syndrome.
本发明化合物可以施用一次或根据其中持续给定时间段以不同的时间间隔施用多个剂量的给药方案施用。例如,剂量可以每天施用一、二、三或四次。可以施用剂量,直到实现期望治疗效果,或无限期施用以维持期望治疗效果。本发明化合物的合适给药方案取决于该化合物的药代动力学特性,诸如吸收、分布和半衰期,其可由技术人员确定。此外,本发明化合物的合适的给药方案 (包括施用这样的方案的持续时间)取决于所治疗的疾病或病症、所治疗的疾病或病症的严重程度、所治疗的患者的年龄和身体状况、待治疗的患者的病史、 并行疗法的性质、期望的治疗效果和技术人员知识和专业内的类似因素。这样的技术人员应进一步理解,合适的给药方案可根据个别患者对于给药方案的应答或由于个别患者需要随时间变化而需要调整。总的每日剂量范围为1 mg至2000 mg。The compounds of the present invention can be administered once or according to a dosage regimen in which multiple doses are administered at different time intervals for a given period of time. For example, the dosage can be administered one, two, three or four times a day. The dosage can be administered until the desired therapeutic effect is achieved, or administered indefinitely to maintain the desired therapeutic effect. The appropriate dosage regimen of the compounds of the present invention depends on the pharmacokinetic properties of the compound, such as absorption, distribution and half-life, which can be determined by a technician. In addition, the appropriate dosage regimen of the compounds of the present invention (including the duration of administering such a regimen) depends on the severity of the disease or condition treated, the disease or condition treated, the age and physical condition of the patient treated, the medical history of the patient to be treated, the nature of concurrent therapy, the desired therapeutic effect and similar factors within the technician's knowledge and profession. Such a technician should further understand that the appropriate dosage regimen can be adjusted according to the response of individual patients to the dosage regimen or because individual patients need to change over time. The total daily dose range is 1 mg to 2000 mg.
对于疗法中的用途,本发明化合物将通常(但不必需)在施用于患者前配制成药物组合物。因此,本发明也涉及药物组合物,其包含本发明化合物以及至少一种药学上可接受的赋形剂。For use in therapy, the compounds of the invention will generally (but not necessarily) be formulated into a pharmaceutical composition prior to administration to a patient. Accordingly, the present invention also relates to a pharmaceutical composition comprising a compound of the invention and at least one pharmaceutically acceptable excipient.
本发明的药物组合物可被制备和包装成散装形式,其中可以提取有效量的本发明化合物,且然后给予患者,诸如用粉末、糖浆和注射用溶液。或者,本发明的药物组合物可被制备和包装成单位剂型。对于口服应用,例如,可以施用一个或多个片剂或胶囊。所述药物组合物的剂量至少含有治疗有效量的本发明化合物(即式(I)化合物或其盐,特别是药学上可接受的盐)。The pharmaceutical composition of the present invention can be prepared and packaged in bulk form, wherein an effective amount of the compound of the present invention can be extracted and then given to the patient, such as with powder, syrup and solution for injection. Alternatively, the pharmaceutical composition of the present invention can be prepared and packaged into unit dosage form. For oral use, for example, one or more tablets or capsules can be administered. The dosage of the pharmaceutical composition contains at least a therapeutically effective amount of the compound of the present invention (i.e., a compound of formula (I) or a salt thereof, particularly a pharmaceutically acceptable salt).
如本文所提供,含有1mg至1000mg的本发明化合物的单位剂型(药物组合物)可以每天施用一、二、三或四次以有效地治疗STING介导的疾病或病症。As provided herein, unit dosage forms (pharmaceutical compositions) containing 1 mg to 1000 mg of a compound of the invention can be administered one, two, three, or four times daily to effectively treat a STING-mediated disease or condition.
本发明的药物组合物通常含有一种本发明化合物。然而,在某些实施方案中,本发明的药物组合物含有多于一种本发明化合物。此外,本发明的药物组合物可以任选地进一步包含一种或多种额外治疗剂(例如,药学活性化合物)。The pharmaceutical compositions of the present invention generally contain one compound of the present invention. However, in certain embodiments, the pharmaceutical compositions of the present invention contain more than one compound of the present invention. In addition, the pharmaceutical compositions of the present invention may optionally further comprise one or more additional therapeutic agents (e.g., pharmaceutically active compounds).
如本文所用,“药学上可接受的赋形剂”指在给予形式中涉及的或与药物组合物相容的药学上可接受的物质、组合物或媒介物。当混合时,每种赋形剂必须与药物组合物的其它成分相容,使得当施用于患者时将实质上降低本发明化合物的效力的相互作用和将导致药物组合物成为药学上不可接受的相互作用得以避免。此外,每种赋形剂的纯度当然必须足够高,使其为药学上可接受的。As used herein, "pharmaceutically acceptable excipient" refers to a pharmaceutically acceptable substance, composition or vehicle that is involved in the form of administration or is compatible with the pharmaceutical composition. When mixed, each excipient must be compatible with the other ingredients of the pharmaceutical composition so that interactions that would substantially reduce the efficacy of the compounds of the invention when administered to a patient and interactions that would cause the pharmaceutical composition to become pharmaceutically unacceptable are avoided. In addition, the purity of each excipient must of course be high enough to make it pharmaceutically acceptable.
本发明化合物和一种或多种药学上可接受的赋形剂通常将被配制成适于通过期望施用途径施用于患者的剂型。常规的剂型包括以下那些:(1)适于口服施用的剂型,诸如片剂、胶囊剂、囊片、丸剂、锭剂、粉剂、糖浆、酏剂、混悬剂、溶液、乳剂、囊剂和扁囊剂;(2)适于肠胃外施用的剂型,诸如无菌溶液、混悬剂和用于重构的粉剂;(3)适于透皮施用的剂型,诸如透皮贴剂;(4)适于直肠施用的剂型,诸如栓剂;(5)适于吸入的剂型,诸如气雾剂和溶液;和(6)适于局部施用的剂型,诸如乳膏、软膏、洗剂、溶液、糊剂、喷雾剂、泡沫剂和凝胶剂。The compounds of the invention and one or more pharmaceutically acceptable excipients will generally be formulated into dosage forms suitable for administration to a patient via the desired route of administration. Conventional dosage forms include those that are: (1) suitable for oral administration, such as tablets, capsules, caplets, pills, lozenges, powders, syrups, elixirs, suspensions, solutions, emulsions, sachets, and cachets; (2) suitable for parenteral administration, such as sterile solutions, suspensions, and powders for reconstitution; (3) suitable for transdermal administration, such as transdermal patches; (4) suitable for rectal administration, such as suppositories; (5) suitable for inhalation, such as aerosols and solutions; and (6) suitable for topical administration, such as creams, ointments, lotions, solutions, pastes, sprays, foams, and gels.
合适的药学上可接受的赋形剂将根据所选具体的剂型而变化。此外,可以根据它们在可以组合物中发挥的特定功能来选择合适的药学上可接受的赋形剂。例如,可以根据它们促进产生均一剂型的能力来选择某些药学上可接受的赋形剂。可以根据它们促进产生稳定的剂型的能力来选择某些药学上可接受的赋形剂。可以根据它们的一旦施用于患者就促进一种或多种本发明化合物从一个器官或身体部分携带或运输至另一器官或身体部分的能力来选择某些药学上可接受的赋形剂。可以根据它们增强患者顺应性的能力来选择某些药学上可接受的赋形剂。Suitable pharmaceutically acceptable excipients will vary according to the selected specific dosage form. In addition, suitable pharmaceutically acceptable excipients can be selected according to the specific functions they play in the composition. For example, certain pharmaceutically acceptable excipients can be selected according to their ability to promote the production of uniform dosage forms. Certain pharmaceutically acceptable excipients can be selected according to their ability to promote the production of stable dosage forms. Certain pharmaceutically acceptable excipients can be selected according to their ability to promote one or more compounds of the present invention to carry or transport to another organ or body part from one organ or body part once they are applied to the patient. Certain pharmaceutically acceptable excipients can be selected according to their ability to enhance patient compliance.
合适的药学上可接受的赋形剂包括下以下类型的赋形剂:稀释剂、填充剂、粘合剂、崩解剂、润滑剂、助流剂、成粒剂、包衣剂、润湿剂、溶剂、共溶剂、助悬剂、乳化剂、增甜剂、调味剂、掩味剂、着色剂、防结块剂、保湿剂、螯合剂、增塑剂、增粘剂、抗氧化剂、防腐剂、稳定剂、表面活性剂和缓冲剂。技术人员应理解,某些药学上可接受的赋形剂可以发挥多于一种功能且可以发挥替代功能,其取决于所述赋形剂在制剂中存在多少和在制剂中存在何种其它成分。Suitable pharmaceutically acceptable excipients include excipients of the following types: diluents, fillers, binders, disintegrants, lubricants, glidants, granulating agents, coating agents, wetting agents, solvents, cosolvents, suspending agents, emulsifiers, sweeteners, flavoring agents, taste masking agents, coloring agents, anti-caking agents, humectants, chelating agents, plasticizers, viscosity increasing agents, antioxidants, preservatives, stabilizers, surfactants and buffers. It will be appreciated by the skilled person that some pharmaceutically acceptable excipients may perform more than one function and may perform alternative functions, depending on how much the excipient is present in the formulation and what other ingredients are present in the formulation.
技术人员具有本领域的知识和技能以使得他们能够选择以适当量用于本发明中的合适的药学上可接受的赋形剂。此外,存在许多技术人员可用的资源,其描述了药学上可接受的赋形剂且可用于选择合适的药学上可接受的赋形剂。实例包括Remington's Pharmaceutical Sciences (Mack Publishing Company)、The Handbook of Pharmaceutical Additives (Gower Publishing Limited)和The Handbook of Pharmaceutical Excipients (the American Pharmaceutical Association and thePharmaceutical Press)。The skilled person has the knowledge and skill in the art to enable them to select suitable pharmaceutically acceptable excipients for use in the present invention in appropriate amounts. In addition, there are many resources available to the skilled person that describe pharmaceutically acceptable excipients and can be used to select suitable pharmaceutically acceptable excipients. Examples include Remington's Pharmaceutical Sciences (Mack Publishing Company), The Handbook of Pharmaceutical Additives (Gower Publishing Limited), and The Handbook of Pharmaceutical Excipients (the American Pharmaceutical Association and the Pharmaceutical Press).
本发明的药物组合物使用本领域技术人员已知的技术和方法制备。通常用于本领域中的一些方法描述于Remington’s Pharmaceutical Sciences (Mack PublishingCompany)中。The pharmaceutical compositions of the present invention are prepared using techniques and methods known to those skilled in the art. Some methods commonly used in the art are described in Remington's Pharmaceutical Sciences (Mack Publishing Company).
在一方面,本发明涉及固体口服剂型,诸如片剂或胶囊剂,其包含有效量的本发明化合物以及稀释剂或填充剂。合适的稀释剂和填充剂包括乳糖、蔗糖、右旋糖、甘露醇、山梨醇、淀粉(例如玉米淀粉、马铃薯淀粉和预胶化淀粉)、纤维素及其衍生物(例如微晶纤维素)、硫酸钙和磷酸氢钙。口服固体剂型可以进一步包含粘合剂。合适的粘合剂包括淀粉(例如玉米淀粉、马铃薯淀粉和预胶化淀粉)、明胶、阿拉伯胶、海藻酸钠、海藻酸、西黄蓍胶、瓜尔胶、聚维酮和纤维素及其衍生物(例如微晶纤维素)。口服固体剂型可以进一步包含崩解剂。合适的崩解剂包括交聚维酮、淀粉羟乙酸钠、交联羧甲基纤维素、海藻酸和羧甲基纤维素钠。口服固体剂型可以进一步包含润滑剂。合适的润滑剂包括硬脂酸、硬脂酸镁、硬脂酸钙和滑石。On the one hand, the present invention relates to solid oral dosage forms, such as tablets or capsules, which contain an effective amount of the compounds of the present invention and a diluent or filler. Suitable diluents and fillers include lactose, sucrose, dextrose, mannitol, sorbitol, starch (e.g., corn starch, potato starch, and pregelatinized starch), cellulose and its derivatives (e.g., microcrystalline cellulose), calcium sulfate, and calcium hydrogen phosphate. Oral solid dosage forms may further include adhesives. Suitable adhesives include starch (e.g., corn starch, potato starch, and pregelatinized starch), gelatin, gum arabic, sodium alginate, alginic acid, tragacanth gum, guar gum, polyvidone, and cellulose and its derivatives (e.g., microcrystalline cellulose). Oral solid dosage forms may further include disintegrants. Suitable disintegrants include crospovidone, sodium starch glycolate, cross-linked carboxymethyl cellulose, alginic acid, and sodium carboxymethyl cellulose. Oral solid dosage forms may further include lubricants. Suitable lubricants include stearic acid, magnesium stearate, calcium stearate, and talc.
应当理解,本发明化合物也可以作为佐剂与疫苗一起配制以调节它们的活性。这样的组合物可以含有抗体(诸抗体)或抗体片段(诸抗体片段)或抗原组分,包括,但不限于,蛋白、DNA、活或死的细菌和/或完整、灭活或裂解的病毒或病毒样颗粒、重组蛋白或其抗原片段,任选地与一种或多种其它具有佐剂活性的组分一起,所述组分包括,但不限于,铝盐、油和水乳剂、热休克蛋白、皂苷、脂质A制剂和衍生物、糖脂、脂质体、TLR激动剂诸如CpG DNA或类似药剂、细胞因子诸如GM-CSF或IL-12或类似药剂。It will be appreciated that the compounds of the invention may also be formulated as adjuvants with vaccines to modulate their activity. Such compositions may contain antibody(ies) or antibody fragment(s) or antigenic components, including, but not limited to, proteins, DNA, live or dead bacteria and/or intact, inactivated or lysed viruses or virus-like particles, recombinant proteins or antigenic fragments thereof, optionally together with one or more other components having adjuvant activity, including, but not limited to, aluminum salts, oil and water emulsions, heat shock proteins, saponins, lipid A preparations and derivatives, glycolipids, liposomes, TLR agonists such as CpG DNA or similar agents, cytokines such as GM-CSF or IL-12 or similar agents.
本发明的某些化合物可以是有效的免疫调节剂,且因此,在其处理时应当小心。Certain compounds of the invention may be potent immunomodulators and, therefore, should be handled with caution.
实施例Example
以下实施例说明本发明。这些实施例不旨在限制本发明的范围,而是为技术人员提供关于制备和使用本发明的化合物、组合物和方法的指导。尽管描述了本发明的具体实施方案,技术人员应理解,在不脱离本发明的精神和范围的情况下,可以进行各种变化和修改。The following examples illustrate the present invention. These examples are not intended to limit the scope of the present invention, but provide guidance for the technical staff on the preparation and use of the compounds, compositions and methods of the present invention. Although specific embodiments of the present invention have been described, the technical staff will appreciate that various changes and modifications may be made without departing from the spirit and scope of the present invention.
应理解,本发明的某些化合物可以是有效的免疫调节剂,且因此,在其处理时应当小心。It will be appreciated that certain compounds of the invention may be potent immunomodulators and, therefore, should be handled with caution.
本文所述的反应适用于产生如本文所定义的具有多种不同取代基基团(例如,R1、R2等)的本发明化合物。技术人员应理解,如果特定取代基与本文所述的合成方法不相容,则可以用对反应条件稳定的合适保护基团保护取代基。合适的保护基团和使用这样的合适的保护基团对不同取代基的保护和脱保护的方法对于本领域技术人员是众所周知的;其实例可见于T. W. Greene ‘Protective Groups in Organic Synthesis’ (第4版, J.Wiley and Sons, 2006)。除非另有说明,否则所有起始材料均得自商业供应商并且不经进一步纯化而使用。The reactions described herein are suitable for producing compounds of the invention having a variety of different substituent groups (e.g., R 1 , R 2 , etc.) as defined herein. It will be appreciated by the skilled artisan that if a particular substituent is incompatible with the synthetic methods described herein, the substituent may be protected with a suitable protecting group that is stable to the reaction conditions. Suitable protecting groups and methods for protecting and deprotecting different substituents using such suitable protecting groups are well known to those skilled in the art; examples thereof can be found in TW Greene 'Protective Groups in Organic Synthesis' (4th edition, J. Wiley and Sons, 2006). Unless otherwise noted, all starting materials were obtained from commercial suppliers and used without further purification.
本文描述的某些中间体化合物形成本发明的又进一步方面。Certain intermediate compounds described herein form yet further aspects of the invention.
一般合成方法General Synthesis Methods
本发明化合物可以使用下面反应方案中说明的合成程序制备,所述合成程序可以通过借鉴熟练有机化学家的知识容易地调整以制备本发明的其它化合物。这些方案中提供的合成适用于采用适当前体产生具有各种不同R基团的本发明化合物,如果需要,所述适当前体可以合适地保护,以实现与本文概述的反应的相容性。如果需要,随后的脱保护提供了通常公开的性质的化合物。尽管仅用式(I)的化合物显示所述方案,但它们是可用于制备本发明化合物的说明性的方法。中间体(用于制备本发明化合物的化合物)也可以作为盐存在。The compounds of the present invention can be prepared using the synthetic procedures described in the following reaction schemes, which can be easily adjusted to prepare other compounds of the present invention by drawing on the knowledge of skilled organic chemists. The synthesis provided in these schemes is suitable for producing compounds of the present invention with various R groups using appropriate precursors, which, if necessary, can be suitably protected to achieve compatibility with the reactions outlined herein. If necessary, subsequent deprotection provides compounds of generally disclosed properties. Although the schemes are shown only with compounds of formula (I), they are illustrative methods that can be used to prepare the compounds of the present invention. Intermediates (compounds for preparing the compounds of the present invention) can also exist as salts.
方法1Method 1
全部变量都如式(I)定义。适当取代的卤代-硝基苯基化合物(1A)与单保护的二胺如(1B)反应以提供N保护的硝基苯胺。除去氨保护基获得胺(1D)。或者,可以通过卤代-硝基苯基化合物(1A)与对称二胺 (1C)反应直接获得胺(1D)。胺(1D)可以与卤代-硝基苯基化合物(1E)反应以获得双-硝基化合物(1F)。在其中(1A)与(1E)相同的情况下,能够通过二胺(1C)与过量卤代-硝基苯基化合物反应直接获得双-硝基化合物(1F)。两个硝基都还原将提供四苯胺(1G)。四苯胺(1G)能够经由以下的两种方法之一转化成(1H),酰胺基苯并咪唑二聚体或大环:1) 用溴化氰处理以获得双-氨基苯并咪唑,然后酰胺与吡唑酸如(1K)或连接的-吡唑二酸(1L)偶联 ;或2) 用异硫氰酸酯(1M)处理直到完全形成二硫脲,然后加入EDC(或其它脱硫试剂)和三乙胺 (或其它合适的碱)并搅拌直到完全脱硫环化(cyclodesulfurization)。采用烷基化试剂和合适的碱烷基化(1H)的苯并咪唑基团提供结构(1I)的化合物。烷基化(即单-、二-、三-烷基化)的位点和程度可通常受条件选择的控制。当在(1I)上存在合适官能团时,这些基团的脱保护或进一步官能化将可能提供另外的化合物(1J)。All variables are defined as in formula (I). Appropriately substituted halo-nitrophenyl compounds (1A) react with mono-protected diamines such as (1B) to provide N-protected nitroanilines. Removal of the amino protecting group yields amine (1D). Alternatively, amine (1D) can be directly obtained by reacting halo-nitrophenyl compounds (1A) with symmetrical diamines (1C). Amine (1D) can be reacted with halo-nitrophenyl compounds (1E) to obtain bis-nitro compounds (1F). In the case where (1A) is the same as (1E), bis-nitro compounds (1F) can be directly obtained by reacting diamine (1C) with excess halo-nitrophenyl compounds. Reduction of both nitro groups will provide tetraphenylamine (1G). Tetraphenylamine (1G) can be converted to (1H), an amidobenzimidazole dimer or macrocycle, by one of two methods: 1) treatment with cyanogen bromide to obtain a bis-aminobenzimidazole followed by coupling of the amide with a pyrazole acid such as (1K) or a linked-pyrazole diacid (1L); or 2) treatment with an isothiocyanate (1M) until dithiourea is completely formed, followed by addition of EDC (or other desulfurization reagent) and triethylamine (or other suitable base) and stirring until cyclodesulfurization is complete. Alkylation of the benzimidazole group of (1H) with an alkylating agent and a suitable base provides compounds of structure (1I). The site and extent of alkylation (i.e., mono-, di-, tri-alkylation) can generally be controlled by the choice of conditions. When suitable functional groups are present on (1I), deprotection or further functionalization of these groups will likely provide additional compounds (1J).
方案1Solution 1
方法2Method 2
全部变量都如式(I)定义。适当取代的卤代-硝基苯基化合物(2A)与单保护的二胺如(2B)反应以提供硝基-苯胺 (2C)。硝基在合适条件下的还原获得二苯胺(dianiline)(2D)。二苯胺(2D)可以经由以下两种方法之一转化成酰胺基苯并咪唑(2E):1) 采用溴化氰处理然后酰胺与吡唑酸如(2M)偶联;或2) 采用异硫氰酸酯(2N)处理直到完全形成硫脲,然后加入EDC (或其它合适的脱硫试剂)和三乙胺 (或其它合适的碱)并搅拌直到完全脱硫环化。除去胺保护基获得胺(2F),其能够与卤代-硝基苯基化合物(2G)反应以获得硝基苯胺(2H)。硝基的还原将提供二苯胺(2I)。二苯胺(2I)能够经由以下两种方法之一转化成酰胺基苯并咪唑 (2J):1) 采用溴化氰处理然后酰胺与吡唑酸如(2O)偶联;或2) 采用异硫氰酸酯(2P)处理直到完全形成硫脲,然后加入EDC (或其它合适的脱硫试剂)和三乙胺 (或其它合适的碱)并搅拌直到完全脱硫环化。采用烷基化试剂和合适的碱烷基化(2J)的苯并咪唑基团提供了结构(2K)的化合物。烷基化(即单-、二-、三-烷基化)的位点和程度可通常受条件选择的控制。当在(2K)上存在合适官能团时,这些基团的脱保护或进一步官能化将可能提供另外的化合物(2L)。All variables are defined as in formula (I). Appropriately substituted halo-nitrophenyl compounds (2A) react with mono-protected diamines such as (2B) to provide nitro-aniline (2C). Reduction of the nitro group under appropriate conditions yields diphenylamine (2D). Diphenylamine (2D) can be converted to amidobenzimidazole (2E) via one of two methods: 1) treatment with cyanogen bromide followed by coupling of the amide with a pyrazole acid such as (2M); or 2) treatment with an isothiocyanate (2N) until thiourea is fully formed, followed by addition of EDC (or other suitable desulfurization reagent) and triethylamine (or other suitable base) and stirring until desulfurization cyclization is complete. Removal of the amine protecting group yields amine (2F), which can react with halo-nitrophenyl compounds (2G) to yield nitroaniline (2H). Reduction of the nitro group will provide diphenylamine (2I). Diphenylamine (2I) can be converted to amidobenzimidazole (2J) via one of two methods: 1) treatment with cyanogen bromide followed by coupling of the amide with a pyrazole acid such as (2O); or 2) treatment with an isothiocyanate (2P) until thiourea is fully formed, followed by addition of EDC (or other suitable desulfurization reagent) and triethylamine (or other suitable base) and stirring until desulfurization and cyclization are complete. Alkylation of the benzimidazole group of (2J) with an alkylating agent and a suitable base provides compounds of structure (2K). The site and extent of alkylation (i.e., mono-, di-, tri-alkylation) can generally be controlled by the choice of conditions. When suitable functional groups are present on (2K), deprotection or further functionalization of these groups will likely provide additional compounds (2L).
方案2Solution 2
方法3Method 3
全部变量都如式(I)定义。适当取代的卤代-硝基苯基化合物(3A)与合适的二亲电试剂(dielectrophile)如二溴化物(3B)反应以提供硝基-苯胺单溴化物(3C)。采用另一适当取代的卤代-硝基苯基化合物(3D)处理(3C)提供连接的双卤代-硝基苯基化合物(3E)。双卤代-硝基苯基化合物(3E)与含连接基团的二胺(3F)的反应提供二硝基大环(3G)。两个硝基都还原将提供四苯胺(3H)。四苯胺(3H)能够经由以下的两种方法之一转化成大环(3I):1) 采用溴化氰处理以提供双-氨基苯并咪唑,然后酰胺与吡唑酸如(3L)或连接的-吡唑二酸(3M)偶联;或2)采用异硫氰酸酯(3N)处理直到完全形成二硫脲,然后加入EDC (或其它脱硫试剂)和三乙胺 (或其它合适的碱)并搅拌直到完全脱硫环化。采用烷基化试剂和合适的碱烷基化(3I)的苯并咪唑基团提供了结构(3J)的化合物。烷基化(即单-、二-、三-烷基化)的位点和程度可通常受条件选择的控制。当在(3J)上存在合适官能团时,这些基团的脱保护或进一步官能化将可能提供另外的化合物(3K)。All variables are defined as in formula (I). Appropriately substituted halo-nitrophenyl compounds (3A) react with suitable dielectrophiles such as dibromide (3B) to provide nitro-aniline monobromide (3C). Treatment of (3C) with another appropriately substituted halo-nitrophenyl compound (3D) provides a linked dihalo-nitrophenyl compound (3E). Reaction of dihalo-nitrophenyl compounds (3E) with diamines (3F) containing linking groups provides dinitro macrocycles (3G). Reduction of both nitro groups will provide tetraphenylamine (3H). Tetraphenylamine (3H) can be converted to the macrocycle (3I) via one of two methods: 1) treatment with cyanogen bromide to provide a bis-aminobenzimidazole followed by coupling of the amide with a pyrazole acid such as (3L) or a linked-pyrazole diacid (3M); or 2) treatment with an isothiocyanate (3N) until dithiourea formation is complete, followed by addition of EDC (or other desulfurization reagent) and triethylamine (or other suitable base) and stirring until desulfurization and cyclization are complete. Alkylation of the benzimidazole group of (3I) with an alkylating agent and a suitable base provides compounds of structure (3J). The site and extent of alkylation (i.e., mono-, di-, tri-alkylation) can generally be controlled by the choice of conditions. When suitable functional groups are present on (3J), deprotection or further functionalization of these groups will likely provide additional compounds (3K).
方案3Solution 3
方法4Method 4
全部变量都如式(I)定义。适当取代的卤代-硝基苯基化合物(4A)与单保护的二胺如(4B)反应以提供硝基-苯胺(4C)。硝基在合适条件下的还原将提供二苯胺(4D)。二苯胺(4D)可以经由以下两种方法之一转化成酰胺基苯并咪唑(4E):1) 采用溴化氰处理,然后是酰胺与吡唑酸如(4N)偶联;或2) 采用异硫氰酸酯(4O)处理直到完全形成硫脲,然后加入EDC (或其它合适的脱硫试剂)和三乙胺 (或其它合适的碱)并搅拌直到完全脱硫环化。采用烷基化试剂和合适的碱烷基化(4E)的苯并咪唑基团提供结构(4F)的化合物。若需要,脱保护/官能化R基团。除去酰胺保护基团提供胺(4G),其能够与卤代-硝基苯基化合物(4H)反应以提供硝基苯胺(4I)。硝基的还原将提供二苯胺(4J)。二苯胺(4J)可经由以下的两种方法之一转化成酰胺基苯并咪唑 (4K):1) 采用溴化氰处理,然后是酰胺与吡唑酸如(4P)偶联;或2) 采用异硫氰酸酯(4Q)处理直到完全形成硫脲,然后加入EDC (或其它合适的脱硫试剂)和三乙胺 (或其它合适的碱)并搅拌直到完全脱硫环化。采用烷基化试剂和适当的碱烷基化新形成的(4K)的苯并咪唑基团提供结构(4L)的化合物。按需要,可脱保护或官能化(即含烯烃部分的连接基团的二羟基化)R基团和/或连接基团以提供另外的化合物(4M)。All variables are defined as in formula (I). Appropriately substituted halo-nitrophenyl compounds (4A) are reacted with mono-protected diamines such as (4B) to provide nitro-anilines (4C). Reduction of the nitro group under appropriate conditions will provide diphenylamines (4D). Diphenylamines (4D) can be converted to amide benzimidazoles (4E) via one of two methods: 1) treatment with cyanogen bromide followed by coupling of the amide with a pyrazole acid such as (4N); or 2) treatment with an isothiocyanate (4O) until thiourea is fully formed, followed by addition of EDC (or other suitable desulfurization reagent) and triethylamine (or other suitable base) and stirring until desulfurization cyclization is complete. Alkylation of the benzimidazole group of (4E) with an alkylating agent and a suitable base provides compounds of structure (4F). Deprotection/functionalization of the R group is performed if desired. Removal of the amide protecting group provides an amine (4G) which can react with a halo-nitrophenyl compound (4H) to provide a nitroaniline (4I). Reduction of the nitro group will provide the diphenylamine (4J). The diphenylamine (4J) can be converted to the amidobenzimidazole (4K) by one of two methods: 1) treatment with cyanogen bromide followed by coupling of the amide with a pyrazole acid such as (4P); or 2) treatment with an isothiocyanate (4Q) until the thiourea is fully formed, followed by addition of EDC (or other suitable desulfurization reagent) and triethylamine (or other suitable base) and stirring until desulfurization and cyclization are complete. Alkylation of the newly formed benzimidazole group of (4K) with an alkylating agent and an appropriate base provides compounds of structure (4L). The R group and/or linking group can be deprotected or functionalized (i.e., dihydroxylation of the linking group containing the olefin moiety) as desired to provide additional compounds (4M).
方案4Solution 4
方法5Method 5
全部变量都如式(I)定义。适当取代的卤代-硝基苯基化合物(5A)与单保护的二胺如(5B)反应以提供N-保护的硝基-苯胺。胺保护基的去除提供胺(5D)。或者,胺(5D)可通过卤代-硝基苯基化合物(5A)与对称二胺 (5C)反应直接获得。胺(5D)可与卤代-硝基苯基化合物(5E)反应以提供双-硝基化合物(5F)。在其中(5A)与(5E)相同的情况下,双-硝基化合物(5F)可通过二胺 (5C)与过量的卤代-硝基苯基化合物反应直接获得。如果需要,脱保护/官能化R基团(即当RA1基团为卤化物时,形成CC键)。硝基的还原将提供四苯胺(5H)。如果存在并取决于所使用的条件,在(5F)中存在的其它基团也可能被还原 (即烯烃、芳基卤化物)。四苯胺(5H)可经由以下两种方法之一转化成(5I),酰胺基苯并咪唑二聚物或大环:1)采用溴化氰处理以提供双-氨基苯并咪唑,然后是酰胺与吡唑酸如(5L)或连接的-吡唑二酸(5M)偶联;或2) 采用异硫氰酸酯(5N)处理直到完全形成二硫脲,然后加入EDC (或其它脱硫试剂)和三乙胺 (或其它合适的碱)并搅拌直到完全脱硫环化。采用烷基化试剂和合适的碱烷基化(5I)的苯并咪唑基团提供了结构(5J)的化合物。烷基化(即单-、二-、三-烷基化)的位点和程度可通常受条件选择的控制。按需要,可脱保护或官能化(即含烯烃部分的连接基团的二羟基化)R基团和/或连接基团以提供另外的化合物(5K)。All variables are defined as in formula (I). Appropriately substituted halo-nitrophenyl compounds (5A) react with mono-protected diamines such as (5B) to provide N-protected nitro-aniline. Removal of the amine protecting group provides amine (5D). Alternatively, amine (5D) can be directly obtained by reacting halo-nitrophenyl compounds (5A) with symmetrical diamines (5C). Amine (5D) can react with halo-nitrophenyl compounds (5E) to provide bis-nitro compounds (5F). In the case where (5A) is identical to (5E), bis-nitro compounds (5F) can be directly obtained by reacting diamines (5C) with excess halo-nitrophenyl compounds. If necessary, deprotection/functionalization of the R group (i.e., when the R A1 group is a halide, a C-C bond is formed). Reduction of the nitro group will provide tetraphenylamine (5H). If present and depending on the conditions used, other groups present in (5F) may also be reduced (i.e., olefins, aryl halides). Tetraphenylamine (5H) can be converted to (5I), an amidobenzimidazole dimer or macrocycle, by one of two methods: 1) treatment with cyanogen bromide to provide a bis-aminobenzimidazole followed by coupling of the amide with a pyrazole acid such as (5L) or a linked-pyrazole diacid (5M); or 2) treatment with an isothiocyanate (5N) until dithiourea formation is complete, followed by addition of EDC (or other desulfurization reagent) and triethylamine (or other suitable base) and stirring until desulfurization and cyclization are complete. Alkylation of the benzimidazole group of (5I) with an alkylating agent and a suitable base provides compounds of structure (5J). The site and extent of alkylation (i.e., mono-, di-, tri-alkylation) can generally be controlled by the choice of conditions. The R group and/or linking group can be deprotected or functionalized (i.e., dihydroxylation of the linking group containing the olefin moiety) as desired to provide additional compounds (5K).
方案5Solution 5
使用Perkin Elmer E-Notebook或MarvinSketch 5.11.4_b82 (Chemaxon)内部的软件命名程序ChemDraw Pro 12.0.2.1076 Plug-In生成本文所述的中间体和最终化合物的名称。The names of the intermediates and final compounds described herein were generated using the software naming program ChemDraw Pro 12.0.2.1076 Plug-In within Perkin Elmer E-Notebook or MarvinSketch 5.11.4_b82 (Chemaxon).
本领域技术人员应了解,在某些情况下,这些程序可能将结构描绘的化合物命名为该化合物的互变异构体或同分异构体。应理解,任何对命名化合物或结构描绘的化合物的提及旨在涵盖这样的化合物的所有互变异构体或同分异构体及其互变异构体和/或同分异构体的任何混合物。Those skilled in the art will appreciate that in some cases, these programs may name a structurally depicted compound as a tautomer or isomer of the compound. It will be understood that any reference to a named compound or a structurally depicted compound is intended to encompass all tautomers or isomers of such a compound and any mixtures of tautomers and/or isomers thereof.
以下缩写可用于本说明书中:The following abbreviations may be used in this manual:
中间体1Intermediate 1
(3-溴丙氧基)(叔丁基)二甲基硅烷(3-Bromopropoxy)(tert-butyl)dimethylsilane
向在DCM (100 mL)中的1H-咪唑 (13.4 g, 197 mmol)加入3-溴丙烷-1-醇 (13.7g, 99 mmol),然后缓慢加入在DCM (20 ml)中的叔丁基氯二甲基硅烷 (17.8 g, 118mmol)。在室温3h之后,浓缩该反应至~100 mL并倾倒在EtOAc (800 mL)中,用5%柠檬酸水溶液(2 x 200 mL)和盐水洗涤。经MgSO4干燥有机层,过滤并浓缩以得到标题化合物(10.0 g,39.5 mmol, 40%收率)。1H NMR (400 MHz, 氯仿-d) δ ppm 3.78 (t, J=5.70 Hz, 2 H),3.56 (t, J=6.46 Hz, 2 H), 2.07 (t, J=5.83 Hz, 2 H), 0.94 (s, 9 H), 0.11 (s, 6H)。3-bromopropane-1-ol (13.7 g, 99 mmol) was added to 1H-imidazole (13.4 g, 197 mmol) in DCM (100 mL), and then tert-butylchlorodimethylsilane (17.8 g, 118 mmol) was slowly added to DCM (20 ml). After 3 h at room temperature, the reaction was concentrated to ~100 mL and poured into EtOAc (800 mL), washed with 5% citric acid aqueous solution (2 x 200 mL) and brine. The organic layer was dried over MgSO 4 , filtered and concentrated to obtain the title compound (10.0 g, 39.5 mmol, 40% yield). 1 H NMR (400 MHz, chloroform- d ) δ ppm 3.78 (t, J =5.70 Hz, 2 H), 3.56 (t, J =6.46 Hz, 2 H), 2.07 (t, J =5.83 Hz, 2 H), 0.94 (s, 9 H), 0.11 (s, 6H).
中间体2Intermediate 2
4-氯-3-甲氧基-5-硝基苯甲酰胺4-Chloro-3-methoxy-5-nitrobenzamide
在室温在NH4OH (10 mL, 77 mmol)中搅拌4-氯-3-甲氧基-5-硝基苯甲酸甲酯(1000 mg, 4.07 mmol)持续24 h。然后,使该反应温度升高至50 oC持续2 h。将另外的2 mL(~3.7 eq)的NH4OH加入容器中。在50℃再搅拌2h (总共4 h)之后,冷却该反应至室温。过滤固体并用冷水漂洗。在低真空(house vacuum)下干燥固体并冻干以提供作为褐色固体的4-氯-3-甲氧基-5-硝基苯甲酰胺 (710 mg, 2.99 mmol, 73%收率)。1H NMR (400 MHz,DMSO-d 6) δ ppm 8.31 (br. s., 1 H), 8.06 (d, J=1.77 Hz, 1 H), 7.88 (d, J=1.77Hz, 1 H), 7.81 (br. s., 1 H), 4.02 (s, 3 H)。LCMS [M+H]+ = 230.9。Methyl 4-chloro-3-methoxy-5-nitrobenzoate (1000 mg, 4.07 mmol) was stirred in NH 4 OH (10 mL, 77 mmol) at room temperature for 24 h. Then, the reaction temperature was raised to 50 ° C for 2 h. An additional 2 mL (~3.7 eq) of NH 4 OH was added to the vessel. After stirring at 50 ° C for another 2 h (4 h total), the reaction was cooled to room temperature. The solid was filtered and rinsed with cold water. The solid was dried under house vacuum and lyophilized to provide 4-chloro-3-methoxy-5-nitrobenzamide (710 mg, 2.99 mmol, 73% yield) as a brown solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.31 (br. s., 1 H), 8.06 (d, J =1.77 Hz, 1 H), 7.88 (d, J =1.77Hz, 1 H), 7.81 (br. s., 1 H), 4.02 (s, 3 H). LCMS [M+H] + = 230.9.
中间体3Intermediate 3
4-氯-3-羟基-5-硝基苯甲酰胺4-Chloro-3-hydroxy-5-nitrobenzamide
将4-氯-3-甲氧基-5-硝基苯甲酰胺 (1 g, 4.34 mmol)悬浮于干燥DCM (15 mL)中并在室温搅拌。向该反应逐滴加入BBr3 (17.4 mL, 1 M在DCM中)。快速形成浆料,将其在室温在氮气下搅拌过夜。将该反应倾倒至冰水(300 mL)中并剧烈搅拌30 min。过滤所得悬浮液并干燥固体以提供标题化合物(610 mg, 2.82 mmol, 65%收率)。1H NMR (400 MHz,DMSO-d 6) δ ppm 11.53 (br. s., 1 H), 8.17 (br. s., 1 H), 7.92 (s, 1 H), 7.72(s, 1 H), 7.66 (br. s., 1 H)。LCMS [M + H]+ = 217。4-Chloro-3-methoxy-5-nitrobenzamide (1 g, 4.34 mmol) was suspended in dry DCM (15 mL) and stirred at room temperature. BBr 3 (17.4 mL, 1 M in DCM) was added dropwise to the reaction. A slurry was quickly formed, which was stirred overnight at room temperature under nitrogen. The reaction was poured into ice water (300 mL) and stirred vigorously for 30 min. The resulting suspension was filtered and the solid was dried to provide the title compound (610 mg, 2.82 mmol, 65% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 11.53 (br. s., 1 H), 8.17 (br. s., 1 H), 7.92 (s, 1 H), 7.72 (s, 1 H), 7.66 (br. s., 1 H). LCMS [M+H] + = 217.
中间体4Intermediate 4
3-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氯-5-硝基苯甲酰胺3-(3-((tert-Butyldimethylsilyl)oxy)propoxy)-4-chloro-5-nitrobenzamide
将(3-溴丙氧基)(叔丁基)二甲基硅烷 (7.3 g, 28.8 mmol)溶解在干燥DMF (75mL)中,加入4-氯-3-羟基-5-硝基苯甲酰胺 (4.8 g, 22.16 mmol),然后加入K2CO3 (6.13g, 44.3 mmol)并在100℃在氮气下搅拌2h。将该反应冷却至室温,倾倒至EtOAc (600 mL)中,用水(600 mL)、盐水洗涤,用MgSO4干燥,过滤,并在真空下浓缩。通过硅胶层析法采用20-80% 己烷/EtOAc洗脱来纯化残余物以得到标题化合物(7.43 g, 19.1 mmol, 86%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.29 (br. s., 1 H), 8.05 (d, J=1.71 Hz, 1H), 7.89 (d, J=1.71 Hz, 1 H), 7.77 (br. s., 1 H), 4.30 (t, J=5.99 Hz, 2 H),3.80 (t, J=5.99 Hz, 2 H), 1.98 (五重峰, J=5.99 Hz, 2 H), 0.80 - 0.90 (m, 9H), 0.02 (s, 6 H)。LCMS [M + H]+ = 389。(3-Bromopropoxy)(tert-butyl)dimethylsilane (7.3 g, 28.8 mmol) was dissolved in dry DMF (75 mL), 4-chloro-3-hydroxy-5-nitrobenzamide (4.8 g, 22.16 mmol) was added, followed by K 2 CO 3 (6.13 g, 44.3 mmol) and stirred at 100 °C under nitrogen for 2 h. The reaction was cooled to room temperature, poured into EtOAc (600 mL), washed with water (600 mL), brine, dried over MgSO 4 , filtered, and concentrated under vacuum. The residue was purified by silica gel chromatography eluting with 20-80% hexanes/EtOAc to give the title compound (7.43 g, 19.1 mmol, 86% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.29 (br. s., 1 H), 8.05 (d, J =1.71 Hz, 1H), 7.89 (d, J =1.71 Hz, 1 H), 7.77 (br. s., 1 H), 4.30 (t, J =5.99 Hz, 2 H) ,3.80 (t, J =5.99 Hz, 2 H), 1.98 (quint, J =5.99 Hz, 2 H), 0.80 - 0.90 (m, 9H), 0.02 (s, 6 H). LCMS [M + H] + = 389.
中间体5Intermediate 5
4-氯-3-(3-吗啉代丙氧基)-5-硝基苯甲酰胺4-Chloro-3-(3-morpholinopropoxy)-5-nitrobenzamide
在70℃搅拌4-氯-3-羟基-5-硝基苯甲酰胺 (5000 mg, 23.09 mmol), 4-(3-氯丙基)吗啉 (4534 mg, 27.7 mmol)、K2CO3 (4148 mg, 30.0 mmol)在DMF (30 mL)中的混合物过夜。在真空下除去溶剂以提供粗固体产物,将其通过硅胶层析法 (12 g柱, MeOH:DCM=1:10)纯化。合并纯级分并在真空中除去溶剂以提供作为黄色固体的4-氯-3-(3-吗啉代丙氧基)-5-硝基苯甲酰胺 (3200 mg, 9.31 mmol, 40.3%收率)。1H NMR (400 MHz, DMSO-d 6)δ ppm 8.29 (s, 1 H), 8.04 (s, 1 H), 7.88 (d, J=1.2 Hz, 1 H), 7.77 (s, 1 H),4.28 (t, J=6.2 Hz, 2 H), 3.62 - 3.52 (m, 4 H), 2.46 - 2.44 (m, 2 H), 2.37(br. s., 4 H), 2.02 - 1.90 (m, 2 H)。LCMS (m/z): 343.8 [M + H]+。A mixture of 4-chloro-3-hydroxy-5-nitrobenzamide (5000 mg, 23.09 mmol), 4-(3-chloropropyl)morpholine (4534 mg, 27.7 mmol), K2CO3 (4148 mg, 30.0 mmol) in DMF (30 mL) was stirred at 70°C overnight. The solvent was removed under vacuum to provide a crude solid product, which was purified by silica gel chromatography (12 g column, MeOH:DCM=1:10). The pure fractions were combined and the solvent was removed in vacuum to provide 4-chloro-3-(3-morpholinopropoxy)-5-nitrobenzamide (3200 mg, 9.31 mmol, 40.3% yield) as a yellow solid. 1 H NMR (400 MHz, DMSO- d 6 )δ ppm 8.29 (s, 1 H), 8.04 (s, 1 H), 7.88 (d, J =1.2 Hz, 1 H), 7.77 (s, 1 H), 4.28 (t, J =6.2 Hz, 2 H), 3.62 - 3.52 (m, 4 H), 2.46 - 2.44 (m, 2 H), 2.37 (br. s., 4 H), 2.02 - 1.90 (m, 2 H). LCMS ( m/z ): 343.8 [M + H] + .
中间体6Intermediate 6
(E)-1-(4-氨基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 盐酸盐( E )-1-(4-aminobut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide hydrochloride
步骤1: (E)-(4-((4-氨基甲酰基-2-硝基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯Step 1: ( E )-tert-butyl (4-((4-carbamoyl-2-nitrophenyl)amino)but-2-en-1-yl)carbamate
在室温搅拌4-氟-3-硝基苯甲酰胺 (10.0 g, 54.3 mmol)、(E)-(4-氨基丁-2-烯-1-基)氨基甲酸叔丁酯 (10.62 g, 57.0 mmol)和K2CO3 (15.01 g, 109 mmol)在DMSO (200mL)中的混合物过夜。将该反应倾倒至水(2000 mL)中并搅拌30 min。通过过滤收集所得固体以得到标题化合物(18.3 g, 52.2 mmol, 96%收率)。LCMS [2M+H]+ = 700.5。A mixture of 4-fluoro-3-nitrobenzamide (10.0 g, 54.3 mmol), ( E )-tert-butyl (4-aminobut-2-en-1-yl) carbamate (10.62 g, 57.0 mmol) and K2CO3 (15.01 g, 109 mmol) in DMSO (200 mL) was stirred overnight at room temperature. The reaction was poured into water (2000 mL) and stirred for 30 min. The resulting solid was collected by filtration to give the title compound (18.3 g, 52.2 mmol, 96% yield). LCMS [2M+H] + = 700.5.
步骤2: (E)-(4-((2-氨基-4-氨基甲酰基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯Step 2: ( E )-tert-butyl (4-((2-amino-4-carbamoylphenyl)amino)but-2-en-1-yl)carbamate
向在DMF (300 mL)中的(E)-(4-((4-氨基甲酰基-2-硝基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯 (18.3 g, 52.2 mmol)加入氯化亚锡二水合物(58.9 g, 261 mmol)。在室温搅拌过夜之后,将该反应逐滴加入饱和NaHCO3水溶液(2000 mL)中并用EtOAc (5 X500 mL)萃取。用盐水(200 mL)洗涤合并的有机层,经Na2SO4干燥,过滤并浓缩以得到作为黄色油的标题化合物(16.5 g, 51.5 mmol, 99%收率)。LCMS [M-BOC+H]+ = 221.1。To tert-butyl ( E )-(4-((4-carbamoyl-2-nitrophenyl)amino)but-2-ene-1-yl)carbamate (18.3 g, 52.2 mmol) in DMF (300 mL) was added stannous chloride dihydrate (58.9 g, 261 mmol). After stirring overnight at room temperature, the reaction was added dropwise to saturated NaHCO aqueous solution (2000 mL) and extracted with EtOAc (5 x 500 mL). The combined organic layers were washed with brine (200 mL ), dried over Na2SO4 , filtered and concentrated to give the title compound (16.5 g, 51.5 mmol, 99% yield) as a yellow oil. LCMS [M-BOC+H] + = 221.1.
步骤3: (E)-(4-(2-氨基-5-氨基甲酰基-1H苯并[d]咪唑-1-基)丁-2-烯-1-基)氨基甲酸叔丁酯Step 3: ( E )-tert-butyl (4-(2-amino-5-carbamoyl-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)carbamate
将(E)-(4-((2-氨基-4-氨基甲酰基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯(16.5 g, 51.5 mmol)和溴化氰(8.18 g, 77 mmol)在THF (200 mL)中的混合物加热至回流过夜。 将该反应冷却至室温,用饱和NaHCO3水溶液(500 mL)稀释,并用EtOAc (5 X 300mL)萃取。用盐水洗涤合并的有机层,经Na2SO4干燥,过滤并浓缩。经硅胶采用50:1至20:1 在MeOH中的DCM(+ 3% NH4OH)洗脱来纯化残余物以得到作为灰白色固体的标题化合物(13.7g, 39.7 mmol, 77%收率)。LCMS [M + H]+ = 346.1。A mixture of ( E )-tert-butyl(4-((2-amino-4-carbamoylphenyl)amino)but-2-en-1-yl)carbamate (16.5 g, 51.5 mmol) and cyanogen bromide (8.18 g, 77 mmol) in THF (200 mL) was heated to reflux overnight. The reaction was cooled to room temperature, diluted with saturated aqueous NaHCO3 solution (500 mL), and extracted with EtOAc (5 x 300 mL). The combined organic layers were washed with brine, dried over Na2SO4 , filtered and concentrated. The residue was purified over silica gel eluting with 50: 1 to 20:1 DCM in MeOH (+ 3% NH4OH ) to give the title compound (13.7 g, 39.7 mmol, 77% yield) as an off-white solid. LCMS [M+H] + = 346.1.
步骤4: (E)-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)氨基甲酸叔丁酯Step 4: ( E )-tert-butyl (4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)carbamate
在0℃向在DCM (500 mL)中的1-乙基-3-甲基-1H-吡唑-5-甲酸 (9.17 g, 59.5mmol)加入EDC (20.53 g, 107 mmol)和HOBt (18.22 g, 119 mmol)。在15 min后,加入(E)-(4-(2-氨基-5-氨基甲酰基-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)氨基甲酸叔丁酯(13.7 g, 39.7 mmol)在DMF (50 mL)中的混合物,然后加入TEA (27.6 mL, 198 mmol)。将反应温热至室温,搅拌过夜并浓缩。用水(500 mL)稀释残余物并用EtOAc (3 X 300 mL)萃取,并用盐水洗涤合并的有机相,经Na2SO4干燥,过滤并浓缩。经硅胶采用50:1至20:1 DCM:MeOH洗脱来纯化残余物以提供粗产物,其用DCM (300 mL)洗涤并通过过滤收集以得到作为灰白色固体的标题化合物(14.0 g, 29.1 mmol, 73%收率)。1H NMR (400 MHz, DMSO-d 6 ) δppm 12.84 (s, 1 H), 8.00 - 7.97 (m, 2 H), 7.80 - 7.78 (m, 1 H), 7.49 (d, J=8.4 Hz, 1 H), 7.34 (s, 1 H), 6.95 (t, J=5.5 Hz, 1 H), 6.66 (s, 1 H), 5.73 -5.65 (m, 2 H), 4.83 (d, J=4.3 Hz, 2 H), 4.62 (q, J=7.0 Hz, 2 H), 3.52 (s, 2H), 2.18 (s, 3 H), 1.38 - 1.33 (m, 12 H); LCMS [M + H]+ = 482.0。To 1-ethyl-3-methyl-1H-pyrazole-5-carboxylic acid (9.17 g, 59.5 mmol) in DCM (500 mL) was added EDC (20.53 g, 107 mmol) and HOBt (18.22 g, 119 mmol) at 0°C. After 15 min, a mixture of ( E )-tert-butyl(4-(2-amino-5-carbamoyl-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)carbamate (13.7 g, 39.7 mmol) in DMF (50 mL) was added, followed by TEA (27.6 mL, 198 mmol). The reaction was warmed to room temperature, stirred overnight and concentrated. The residue was diluted with water (500 mL) and extracted with EtOAc (3 X 300 mL), and the combined organic phases were washed with brine, dried over Na2SO4 , filtered and concentrated. The residue was purified on silica gel eluting with 50:1 to 20:1 DCM:MeOH to provide the crude product, which was washed with DCM (300 mL) and collected by filtration to give the title compound as an off-white solid (14.0 g, 29.1 mmol, 73% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δppm 12.84 (s, 1 H), 8.00 - 7.97 (m, 2 H), 7.80 - 7.78 (m, 1 H), 7.49 (d, J =8.4 Hz, 1 H), 7.34 (s, 1 H), 6.95 (t, J =5.5 Hz, 1 H), 6.66 (s, 1 H), 5.73 -5.65 (m, 2 H), 4.83 (d, J =4.3 Hz, 2 H), 4.62 (q, J =7.0 Hz, 2 H), 3.52 (s, 2H), 2.18 (s, 3 H), 1.38 - 1.33 (m, 12 H); LCMS [M + H] + = 482.0.
步骤5: (E)-1-(4-氨基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺盐酸盐Step 5: ( E )-1-(4-aminobut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide hydrochloride
向(E)-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)氨基甲酸叔丁酯 (3.00 g, 6.23 mmol) 在二氧杂环己烷(60 mL)中的悬浮液加入在二氧杂环己烷中的4 N HCl (15.6 mL, 62.3 mmol),然后加入MeOH (15 mL)以溶解一些剩余的固体。在室温30 min后,该反应混合物变得浑浊并将其搅拌大致3天。通过过滤收集所得固体并用DCM洗涤以获得作为白色固体的标题化合物(2.0g, 4.8 mmol, 77%收率)。1H NMR (400 MHz, DMSO-d 6 ) δ ppm 7.97 - 8.09 (br. s., 1H), 7.82 (d, J=8.11 Hz, 1 H), 7.50 (d, J=8.11 Hz, 1 H), 7.38 (br. s., 1 H),6.70 (s, 1 H), 5.97 - 6.08 (m, 1 H), 5.68 - 5.80 (m, 1 H), 4.91 (d, J=4.31Hz, 2 H), 4.60 (q, J=6.67 Hz, 2 H), 3.42 (br. s., 2 H), 2.18 (s, 3 H), 1.36(t, J=6.97 Hz, 3 H); LCMS [M + H]+ = 382.2。To a suspension of tert-butyl ( E )-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)carbamate (3.00 g, 6.23 mmol) in dioxane (60 mL) was added 4 N HCl in dioxane (15.6 mL, 62.3 mmol) followed by MeOH (15 mL) to dissolve some of the remaining solid. After 30 min at room temperature, the reaction mixture became cloudy and was stirred for approximately 3 days. The resulting solid was collected by filtration and washed with DCM to obtain the title compound as a white solid (2.0 g, 4.8 mmol, 77% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 7.97 - 8.09 (br. s., 1H), 7.82 (d, J =8.11 Hz, 1 H), 7.50 (d, J =8.11 Hz, 1 H), 7.38 (br. s., 1 H), 6.70 (s, 1 H), 5. 97 - 6.08 (m, 1 H), 5.68 - 5.80 (m, 1 H), 4.91 (d, J =4.31Hz, 2 H), 4.60 (q, J =6.67 Hz, 2 H), 3.42 (br. s., 2 H), 2.18 (s, 3 H), 1.36(t, J =6.97 Hz , 3 H); LCMS [M+H] + = 382.2.
中间体7Intermediate 7
(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺( E )-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-5-carboxamide
步骤1: (E)-1-(4-((2-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基-6-硝基苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 1: ( E )-1-(4-((2-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoyl-6-nitrophenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide
用TEA (0.28 mL, 2.0 mmol),然后用K2CO3 (274 mg, 1.98 mmol)和3-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氯-5-硝基苯甲酰胺 (385 mg, 0.990 mmol)处理在DMSO (10 mL)中含(E)-1-(4-氨基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺盐酸盐 (517 mg, 1.24 mmol)的微波管。将该反应加热至75℃。在7 h后,浓缩该混合物,并经硅胶采用10 - 90% EtOAc洗脱 (以除去杂质),然后用0-10% 在DCM中的MeOH洗脱来纯化残余物以得到作为橙色固体的标题化合物(200 mg,0.273 mmol, 28%收率)。1H NMR (400 MHz, DMSO-d 6 ) δ ppm 8.16 (d, J=1.52 Hz, 1 H),7.94 - 8.08 (m, 3 H), 7.74 (d, J=8.11 Hz, 2 H), 7.50 (s, 1 H), 7.31 - 7.43(m, 3 H), 6.62 (s, 1 H), 5.74 - 5.81 (m, 2 H), 4.80 (br. s., 2 H), 4.59 (d, J=6.84 Hz, 2 H), 4.13 (br. s., 2 H), 4.01 (t, J=6.08 Hz, 2 H), 3.63 (t, J=5.96Hz, 2 H), 2.16 (s, 3 H), 1.76 - 1.88 (m, 2 H), 1.33 (t, J=7.10 Hz, 3 H), 0.74- 0.82 (m, 9 H), -0.06 (s, 6 H); LCMS [M + H]+ = 734.6。A microwave tube containing ( E )-1-(4- aminobut -2-en-1- yl )-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide hydrochloride (517 mg, 1.24 mmol) in DMSO (10 mL) was treated with TEA (0.28 mL, 2.0 mmol) followed by K 2 CO 3 (274 mg, 1.98 mmol) and 3-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-chloro-5-nitrobenzamide (385 mg, 0.990 mmol). The reaction was heated to 75° C. After 7 h, the mixture was concentrated and the residue was purified over silica gel eluting with 10-90% EtOAc (to remove impurities) and then 0-10% MeOH in DCM to give the title compound as an orange solid (200 mg, 0.273 mmol, 28% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.16 (d, J =1.52 Hz, 1 H), 7.94 - 8.08 (m, 3 H), 7.74 (d, J =8.11 Hz, 2 H), 7.50 (s, 1 H), 7.31 - 7.43 (m, 3 H), 6.62 (s, 1 H), 5.74 - 5.81 (m, 2 H), 4.80 (br. s., 2 H), 4.59 (d, J =6.84 Hz, 2 H), 4.13 (br. s., 2 H), 4.01 (t, J =6.08 Hz, 2 H), 3.63 (t, J =5.96Hz, 2 H ), 2.16 (s, 3 H), 1.76 - 1.88 (m, 2 H), 1.33 (t, J =7.10 Hz, 3 H), 0.74- 0.82 (m, 9 H), -0.06 (s, 6 H); LCMS [M + H] + = 734.6.
步骤2: (E)-1-(4-((2-氨基-6-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 2: ( E )-1-(4-((2-amino-6-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoylphenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide
将(E)-1-(4-((2-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基-6-硝基苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (1 g, 1.363 mmol)悬浮在MeOH (20 mL)中,加入氢氧化铵 (4.62 mL, 34.1mmol)并在室温搅拌5 min。然后加入在水(5 mL)中的次硫酸钠 (1.675 g, 8.18 mmol)。在60 min后,加入EtOAc (300 ml)并用水(50ml x 3)萃取该混合物。分离有机层,用Na2SO4干燥,并在真空下浓缩以获得作为浅黄色固体的标题化合物(710 mg, 1.009 mmol, 74.0%收率),其不经进一步纯化而使用。1H NMR (400 MHz, DMSO-d 6) δ ppm 12.80 (br. s., 1H), 8.00 (s, 1 H), 7.97 (br. s., 1 H), 7.75 (dd, J=8.49, 1.14 Hz, 1 H), 7.63(br. s., 1 H), 7.28 - 7.41 (m, 2 H), 7.00 (br. s., 1 H), 6.84 (d, J=1.52 Hz,1 H), 6.74 (d, J=1.52 Hz, 1 H), 6.65 (s, 1 H), 5.79 - 5.96 (m, 1 H), 5.64 -5.78 (m, 1 H), 4.81 (d, J=4.82 Hz, 2 H), 4.68 (br. s., 2 H), 4.61 (d, J=7.10Hz, 2 H), 3.92 (t, J=5.83 Hz, 2 H), 3.84 (br. s., 1 H), 3.63 (t, J=6.08 Hz, 2H), 3.57 (br. s., 2 H), 2.17 (s, 3 H), 1.70 - 1.82 (m, 2 H), 1.34 (t, J=7.10Hz, 3 H), 0.68 - 0.83 (m, 9 H), -0.06 (s, 6 H); LCMS [M + H]+ = 704.3。(E)-1-(4-((2-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoyl-6-nitrophenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (1 g, 1.363 mmol) was suspended in MeOH (20 mL), ammonium hydroxide (4.62 mL, 34.1 mmol) was added and stirred at room temperature for 5 min. Then sodium sulfoxylate (1.675 g, 8.18 mmol) in water (5 mL) was added. After 60 min, EtOAc (300 ml) was added and the mixture was extracted with water (50 ml x 3). The organic layer was separated, dried over Na 2 SO 4 , and concentrated under vacuum to afford the title compound (710 mg, 1.009 mmol, 74.0% yield) as a light yellow solid, which was used without further purification. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 12.80 (br. s., 1H), 8.00 (s, 1 H), 7.97 (br. s., 1 H), 7.75 (dd, J =8.49, 1.14 Hz, 1 H), 7.63(br. s., 1 H), 7.28 - 7.41 (m, 2 H), 7.00 (br. s., 1 H), 6.84 (d, J =1.52 Hz, 1 H), 6.74 (d, J =1.52 Hz, 1 H), 6.65 (s, 1 H), 5.79 - 5.96 (m, 1 H), 5.64 -5.78 (m, 1 H), 4.81 (d, J =4.82 Hz, 2 H), 4.68 (br. s., 2 H), 4.61 (d, J =7.10Hz, 2 H), 3.92 (t, J =5.83 Hz, 2 H), 3.84 (br. s., 1 H), 3.63 (t, J =6.08 Hz, 2H), 3.57 (br. s. , 2 H), 2.17 (s, 3 H), 1.70 - 1.82 (m, 2 H), 1.34 (t, J =7.10Hz, 3 H), 0.68 - 0.83 (m, 9 H), -0.06 (s, 6 H); LCMS [M + H] + = 704.3.
步骤3: (E)-2-氨基-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-1H-苯并[d]咪唑-5-甲酰胺Step 3: ( E )-2-amino-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-1H-benzo[d]imidazole-5-carboxamide
在室温向(E)-1-(4-((2-氨基-6-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨At room temperature, ( E )-1-(4-((2-amino-6-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-amino
基甲酰基苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (120 mg, 0.170 mmol)在MeOH (5 mL)中的溶液中加入溴化氰(36 mg, 0.34 mmol)。在2 h后,浓缩该反应,并加入EtOAc(10 mL)。在搅拌30 min后,通过过滤分离固体,并用EtOAc洗涤以获得作为浅棕色固体的标题化合物(120 mg, 0.165mmol, 97%收率),其不经进一步纯化而使用。1H NMR (400 MHz, MeOH-d 4) δ ppm 8.00 (d,J=1.27 Hz, 1 H), 7.81 (dd, J=8.36, 1.77 Hz, 1 H), 7.49 (d, J=1.27 Hz, 1 H),7.39 - 7.45 (m, 1 H), 7.36 (d, J=1.27 Hz, 1 H), 6.61 (s, 1 H), 5.82 - 5.99(m, 2 H), 4.96 - 5.01 (m, 2 H), 4.56 - 4.65 (m, 2 H), 4.12 (t, J=6.21 Hz, 2H), 3.62 - 3.75 (m, 2 H), 2.18 - 2.29 (m, 3 H), 1.79 (t, J=6.21 Hz, 2 H),1.24 - 1.54 (m, 5 H), 0.84 - 0.98 (m, 9 H), -0.01 - 0.11 (m, 6 H); LCMS [M +H]+ = 729.5。To a solution of 2-(1-ethyl-3-methyl-1H-pyrazole-5-formamido)-1H-benzo[d]imidazole-5-carboxamide (120 mg, 0.170 mmol) in MeOH (5 mL) was added cyanogen bromide (36 mg, 0.34 mmol). After 2 h, the reaction was concentrated and EtOAc (10 mL) was added. After stirring for 30 min, the solid was isolated by filtration and washed with EtOAc to obtain the title compound as a light brown solid (120 mg, 0.165 mmol, 97% yield), which was used without further purification. 1 H NMR (400 MHz, MeOH- d 4 ) δ ppm 8.00 (d, J =1.27 Hz, 1 H), 7.81 (dd, J =8.36, 1.77 Hz, 1 H), 7.49 (d, J =1.27 Hz, 1 H), 7.39 - 7.45 (m, 1 H), 7.36 (d, J =1.27 Hz, 1 H), 6.61 (s, 1 H), 5.82 - 5.99 (m, 2 H), 4.96 - 5.01 (m, 2 H), 4.56 - 4.65 (m, 2 H), 4.12 (t, J =6.21 Hz, 2H), 3.62 - 3.75 (m, 2 H), 2.18 - 2.29 (m, 3 H), 1.79 (t, J =6.21 Hz, 2 H), 1.24 - 1.54 (m, 5 H), 0.84 - 0.98 (m, 9 H), -0.01 - 0.11 (m, 6 H); LCMS [M +H] + = 729.5.
步骤4: (E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 4: ( E )-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide
向1-乙基-3-甲基-1H-吡唑-5-甲酸 (33 mg, 0.21 mmol)在DMF (3 mL)中的溶液加入HATU (75 mg, 0.20 mmol)和HOBt (12.6 mg, 0.082 mmol)。在室温搅拌10 min后,加入三乙胺 (0.09 mL, 0.66 mmol),然后加入(E)-2-氨基-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-1H-苯并[d]咪唑-5-甲酰胺 (120 mg, 0.165 mmol)并在室温继续该反应。在3天后,通过逐滴加入水,固体从该反应中沉淀出来。通过过滤分离固体并用水洗涤。然后,经硅胶(12g HP Gold柱)用0-20% 在DCM中的MeOH洗脱来纯化该固体。合并所需级分并浓缩以得到作为灰白色固体的标题化合物(29 mg, 0.034 mmol, 20%收率)。1HNMR (400 MHz, THF-d 4) δ ppm 12.53 (br. s., 2 H), 8.00 (d, J=1.01 Hz, 1 H),7.61 (d, J=1.01 Hz, 1 H), 7.53 (dd, J=8.36, 1.52 Hz, 1 H), 7.36 (d, J=6.84Hz, 2 H), 7.29 (d, J=1.01 Hz, 1 H), 7.12 (d, J=8.36 Hz, 1 H), 6.83 (br. s., 2H), 6.66 (d, J=2.28 Hz, 2 H), 6.06 (dt, J=15.46, 5.58 Hz, 1 H), 5.87 (dt, J=15.46, 5.83 Hz, 1 H), 5.09 (d, J=5.32 Hz, 2 H), 4.89 (d, J=5.58 Hz, 2 H),4.59 - 4.72 (m, 4 H), 3.97 (t, J=6.21 Hz, 2 H), 3.69 (t, J=5.96 Hz, 2 H),2.20 (s, 6 H), 1.73 - 1.78 (m, 2 H), 1.40 (td, J=7.03, 1.14 Hz, 6 H), 0.82 -0.94 (m, 9 H), -0.03 - 0.09 (m, 6 H); LCMS [M/2+H]+ = 433.6。To a solution of 1-ethyl-3-methyl-1H-pyrazole-5-carboxylic acid (33 mg, 0.21 mmol) in DMF (3 mL) was added HATU (75 mg, 0.20 mmol) and HOBt (12.6 mg, 0.082 mmol). After stirring at room temperature for 10 min, triethylamine (0.09 mL, 0.66 mmol) was added, followed by ( E )-2-amino-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-1H-benzo[d]imidazole-5-carboxamide (120 mg, 0.165 mmol) and the reaction was continued at room temperature. After 3 days, by dropwise addition of water, solid precipitated out from the reaction. Solid was separated by filtration and washed with water. Then, the solid was purified by eluting with 0-20% MeOH in DCM through silica gel (12g HP Gold column). Combine required fractions and concentrate to obtain the title compound (29 mg, 0.034 mmol, 20% yield) as off-white solid. 1 HNMR (400 MHz, THF- d 4 ) δ ppm 12.53 (br. s., 2 H), 8.00 (d, J =1.01 Hz, 1 H), 7.61 (d, J =1.01 Hz, 1 H), 7.53 (dd, J =8.36, 1.52 Hz, 1 H), 7.36 (d, J = 6.84Hz, 2 H), 7.29 (d, J =1.01 Hz, 1 H), 7.12 (d, J =8.36 Hz, 1 H), 6.83 (br. s., 2H), 6.66 (d, J =2.28 Hz, 2 H), 6.06 (dt, J =15.46, 5.58 Hz, 1 H), 5.87 (dt, J =15.46, 5.83 Hz, 1 H), 5.09 (d, J =5.32 Hz, 2 H), 4.89 (d, J =5.58 Hz, 2 H),4.59 - 4.72 (m, 4 H), 3.97 (t, J =6.21 Hz, 2 H), 3.69 (t, J =5.96 Hz, 2 H),2.20 (s, 6 H), 1.73 - 1.78 (m, 2 H), 1.40 (td, J =7.03, 1.14 Hz, 6 H), 0.82 -0.94 (m, 9 H), -0.03 - 0.09 (m, 6 H); LCMS [M/2+H] + = 433.6。
中间体8Intermediate 8
1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯1-Ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate
向1L圆底烧瓶加入1-乙基-3-甲基-1H-吡唑-5-甲酸 (25 g, 162 mmol)和DCM(500 mL)。向这个非均相溶液加入DMF (0.1 mL, 1.291 mmol),然后缓慢加入草酰氯(15.61 mL, 178 mmol)。在室温搅拌1 h后,在真空下除去挥发物并将该粗物质与二氯甲烷(每次100 mL)共蒸发两次。假定100%收率并直接使用粗品(1-乙基-3-甲基-1H-吡唑-5-羰基氯 (28.0 g, 162 mmol, 100%收率))。To a 1 L round bottom flask was added 1-ethyl-3-methyl-1H-pyrazole-5-carboxylic acid (25 g, 162 mmol) and DCM (500 mL). To this heterogeneous solution was added DMF (0.1 mL, 1.291 mmol) followed by the slow addition of oxalyl chloride (15.61 mL, 178 mmol). After stirring at room temperature for 1 h, the volatiles were removed under vacuum and the crude material was co-evaporated twice with dichloromethane (100 mL each). Assume 100% yield and use the crude product directly (1-ethyl-3-methyl-1H-pyrazole-5-carbonyl chloride (28.0 g, 162 mmol, 100% yield)).
向干燥的1L 圆底烧瓶加入KSCN (18.92 g, 195 mmol)和丙酮(463 mL)。将这个透明均相溶液冷却至0℃。在0℃搅拌5 min后,作为在丙酮(25 mL)中的溶液加入1-乙基-3-甲基-1H-吡唑-5-羰基氯 (28 g, 162 mmol)。一旦完成加料,使该反应在0℃搅拌。在1 min后,加入另外的KSCN(~2 g)并另外搅拌该反应20 min。此时,将己烷 (200 mL)加入该反应混合物并在真空中将该粗非均相溶液浓缩至三分之一体积。重复两次己烷添加和浓缩的过程(每次300 mL己烷)。在最后一次浓缩后,加入己烷 (200 mL)并通过过滤除去固体,采用己烷 (100 mL)漂洗。将所得透明浅黄色滤液浓缩并通过层析法(330g Gold硅胶柱; 采用0-20% EtOAc / 己烷洗脱)纯化。在~7% EtOAc / 己烷下洗脱所需产物。合并所需级分并浓缩,得到作为透明无色液体的1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (27.5 g, 139mmol, 86%收率)。1H NMR (400 MHz, 氯仿-d) δ ppm 6.77 (s, 1 H), 4.54 (q, J=7.10Hz, 2 H), 2.34 (s, 3 H), 1.44 (t, J=7.22 Hz, 3 H); LCMS [M + H]+ = 196.1。酰基异硫氰酸酯产物随时间降解,因此制备~0.4 M 1,4-二氧杂环己烷溶液并冷冻以避免/减慢分解。将这个溶液解冻并直接用于随后的反应中。To a dry 1L round bottom flask was added KSCN (18.92 g, 195 mmol) and acetone (463 mL). This clear homogeneous solution was cooled to 0°C. After stirring at 0°C for 5 min, 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl chloride (28 g, 162 mmol) was added as a solution in acetone (25 mL). Once the addition was complete, the reaction was allowed to stir at 0°C. After 1 min, additional KSCN (~2 g) was added and the reaction was stirred for an additional 20 min. At this point, hexanes (200 mL) were added to the reaction mixture and the crude heterogeneous solution was concentrated to one third of the volume in vacuo. The process of hexanes addition and concentration was repeated twice (300 mL of hexanes each time). After the last concentration, hexanes (200 mL) were added and the solids were removed by filtration, rinsing with hexanes (100 mL). The resulting clear light yellow filtrate was concentrated and purified by chromatography (330 g Gold silica column; eluted with 0-20% EtOAc/hexanes). The desired product eluted at ~7% EtOAc/hexanes. The desired fractions were combined and concentrated to give 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (27.5 g, 139 mmol, 86% yield) as a clear colorless liquid. 1 H NMR (400 MHz, CHLOROFORM- d ) δ ppm 6.77 (s, 1 H), 4.54 (q, J =7.10 Hz, 2 H), 2.34 (s, 3 H), 1.44 (t, J =7.22 Hz, 3 H); LCMS [M + H] + = 196.1. The acyl isothiocyanate product degraded over time, so a ~0.4 M 1,4-dioxane solution was prepared and frozen to avoid/slow decomposition. This solution was thawed and used directly in subsequent reactions.
中间体9Intermediate 9
(E)-(4-((4-氨基甲酰基-2-甲氧基-6-硝基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯tert-Butyl (E)-(4-((4-Carbamoyl-2-methoxy-6-nitrophenyl)amino)but-2-en-1-yl)carbamate
向4-氯-3-甲氧基-5-硝基苯甲酰胺 (1.50 g, 6.50 mmol)在EtOH (25 mL)中的悬浮液加入(E)-(4-氨基丁-2-烯-1-基)氨基甲酸叔丁酯 (1.454 g, 7.81 mmol)和DIEA(3.4 mL, 20 mmol)。在120℃在密封管中搅拌该反应并使其冷却至室温。通过过滤收集所得橙色固体并用EtOH洗涤以得到标题化合物(2.10 g, 5.52 mmol, 85%收率)。1H NMR(400 MHz, DMSO-d 6 ) δ ppm 8.19 (d, J=1.77 Hz, 1 H) 8.03 (br. s., 1 H) 7.76 (t,J=6.08 Hz, 1 H) 7.55 (d, J=1.52 Hz, 1 H) 7.34 (br. s., 1 H) 6.95 (t, J=5.45Hz, 1 H) 5.53 (br. s., 2 H) 4.09 (br. s., 2 H) 3.88 (s, 3 H) 3.48 (br. s., 2H) 1.35 (s, 9 H)。LCMS (m/z): 325.1 [M-t-Bu + H]+。To a suspension of 4-chloro-3-methoxy-5-nitrobenzamide (1.50 g, 6.50 mmol) in EtOH (25 mL) was added ( E )-tert-butyl (4-aminobut-2-en-1-yl)carbamate (1.454 g, 7.81 mmol) and DIEA (3.4 mL, 20 mmol). The reaction was stirred at 120 °C in a sealed tube and allowed to cool to room temperature. The resulting orange solid was collected by filtration and washed with EtOH to give the title compound (2.10 g, 5.52 mmol, 85% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.19 (d, J=1.77 Hz, 1 H) 8.03 (br. s., 1 H) 7.76 (t,J=6.08 Hz, 1 H) 7.55 (d, J=1.52 Hz, 1 H) 7.34 (br. s., 1 H) 6.9 5 (t, J=5.45Hz, 1 H) 5.53 (br. s., 2 H) 4.09 (br. s., 2 H) 3.88 (s, 3 H) 3.48 (br. s., 2H) 1.35 (s, 9 H). LCMS ( m/z ): 325.1 [M- t -Bu + H] + .
制备本文中所述化合物的替代路径:向3-颈 5-升烧瓶加入4-氯-3-甲氧基-5-硝基苯甲酰胺 (451.10 g, 1956 mmol)、正丁醇 (2174 mL)、N-乙基-N-异丙基丙-2-胺(854mL, 4890 mmol)和(E)-(4-氨基丁-2-烯-1-基)氨基甲酸叔丁酯 (403.1 g, 2164 mmol)。向该烧瓶添附冷凝器和含温度探头的隔板(septum)。采用顶部搅拌器(300 rpm)搅拌该反应烧瓶并使用添附至温度调节器的加热套加热至110℃。在6h后,非均相混合物变成深红色和均相。在110℃搅拌该反应24 h。将该混合物冷却至室温。加入异丙醇 (1200 mL)。在布氏漏斗上过滤固体。用异丙醇 (每次1200 mL)漂洗有机滤饼两次。将固体空气干燥过夜(~14h)。获得作为橙色固体的(E)-(4-((4-氨基甲酰基-2-甲氧基-6-硝基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯 (532 g, 1343 mmol, 68.6 %收率)。通过1H NMR、HPLC追踪和LCMS,估计这个固体的最小纯度为96%。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.19 (d, J=1.77Hz, 1 H) 8.03 (br. s., 1 H) 7.76 (t, J=5.96 Hz, 1 H) 7.56 (d, J=1.77 Hz, 1 H)7.35 (br. s., 1 H) 6.96 (t, J=5.58 Hz, 1 H) 5.53 (br. s., 2 H) 4.09 (br. s.,2 H) 3.88 (s, 3 H) 3.48 (br. s., 2 H) 1.09 - 1.54 (m, 9 H)。LCMS (m/z): 381.2[M + H]+。[00136] Alternative route to prepare compounds described herein: To a 3-neck 5-liter flask was added 4-chloro-3-methoxy-5-nitrobenzamide (451.10 g, 1956 mmol), n-butanol (2174 mL), N-ethyl-N-isopropylpropan-2-amine (854 mL, 4890 mmol), and (E)-tert-butyl (4-aminobut-2-en-1-yl)carbamate (403.1 g, 2164 mmol). A condenser and a septum containing a temperature probe were attached to the flask. The reaction flask was stirred with an overhead stirrer (300 rpm) and heated to 110°C using a heating mantle attached to a thermostat. After 6 h, the heterogeneous mixture turned dark red and homogeneous. The reaction was stirred at 110°C for 24 h. The mixture was cooled to room temperature. Isopropanol (1200 mL) was added. The solid was filtered on a Buchner funnel. The organic filter cake was rinsed twice with isopropanol (1200 mL each time). The solid was air dried overnight (~14 h). (E)-(4-((4-carbamoyl-2-methoxy-6-nitrophenyl)amino)but-2-ene-1-yl)carbamic acid tert-butyl ester (532 g, 1343 mmol, 68.6% yield) was obtained as an orange solid. By 1H NMR, HPLC tracking and LCMS, the minimum purity of this solid was estimated to be 96%. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.19 (d, J =1.77Hz, 1 H) 8.03 (br. s., 1 H) 7.76 (t, J =5.96 Hz, 1 H) 7.56 (d, J =1.77 Hz, 1 H)7.35 (br. s., 1 H) 6.96 ( t, J =5.58 Hz, 1 H) 5.53 (br. s., 2 H) 4.09 (br. s., 2 H) 3.88 (s, 3 H) 3.48 (br. s., 2 H) 1.09 - 1.54 (m, 9 H). LCMS ( m/z ): 381.2[M + H] + .
中间体10Intermediate 10
(E)-4-((4-氨基丁-2-烯-1-基)氨基)-3-甲氧基-5-硝基苯甲酰胺, 盐酸盐( E )-4-((4-aminobut-2-en-1-yl)amino)-3-methoxy-5-nitrobenzamide, hydrochloride
向(E)-(4-((4-氨基甲酰基-2-甲氧基-6-硝基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯 (20 g, 47.3 mmol)在MeOH (50 mL)中的悬浮液缓慢加入在二氧杂环己烷中的4M HCl (100 mL, 400 mmol)。在室温搅拌该反应混合物1 h,然后通过过滤分离所得固体,用二乙醚(3 x 100mL)洗涤,并在高真空下干燥以提供标题化合物(13.90 g, 43.9 mmol,93%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.22 (d, J=2.03 Hz, 1 H), 7.76 - 8.16(br. m., 5 H), 7.60 (d, J=2.03 Hz, 1 H), 7.37 (br. s., 1 H), 5.87 (dt, J=15.52, 5.80 Hz, 1 H), 5.62 (dt, J=15.65, 6.37 Hz, 1 H), 4.18 (d, J=5.32 Hz, 2H), 3.90 (s, 3 H), 3.40 (t, J=5.70 Hz, 2 H)。LCMS (m/z): 281.1 [M + H]+。To a suspension of tert-butyl (E)-(4-((4-carbamoyl-2-methoxy-6-nitrophenyl)amino)but-2-en-1-yl)carbamate (20 g, 47.3 mmol) in MeOH (50 mL) was slowly added 4M HCl in dioxane (100 mL, 400 mmol). The reaction mixture was stirred at room temperature for 1 h, then the resulting solid was isolated by filtration, washed with diethyl ether (3 x 100 mL), and dried under high vacuum to afford the title compound (13.90 g, 43.9 mmol, 93% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.22 (d, J =2.03 Hz, 1 H), 7.76 - 8.16 (br. m., 5 H), 7.60 (d, J =2.03 Hz, 1 H), 7.37 (br. s., 1 H), 5.87 (dt, J =15.52 , 5.80 Hz, 1 H), 5.62 (dt, J =15.65, 6.37 Hz, 1 H), 4.18 (d, J =5.32 Hz, 2H), 3.90 (s, 3 H), 3.40 (t, J =5.70 Hz, 2 H). LCMS ( m/z ): 281.1 [M + H] + .
中间体11Intermediate 11
(E)-1-((E)-4-氨基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 3盐酸盐(E)-1-((E)-4-aminobut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 3 hydrochloride
步骤1: ((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)氨基甲酸叔丁酯Step 1: tert-Butyl ((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)carbamate
在室温向在DMF (5 mL)中的(E)-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-1-基) 丁-2-烯-1-基) 氨基甲酸叔丁酯(530 mg, 1.036 mmol)加入碳酸铯 (675 mg, 2.072 mmol)和碘甲烷(0.097 mL, 1.554mmol)。在室温搅拌该反应。在2 h后,用100 mL EtOAc稀释该反应,用2 x 100 mL水和3 x100 mL盐水洗涤。收集有机层并在真空下浓缩以得到作为黄色固体的标题化合物(630 mg,1.04 mmol, 100%收率)。LCMS m/z = 526 [M + H]+。To tert-butyl (E)-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazol-1-yl)but-2-ene-1-yl)carbamate (530 mg, 1.036 mmol) in DMF (5 mL) was added cesium carbonate (675 mg, 2.072 mmol) and iodomethane (0.097 mL, 1.554 mmol) at room temperature. The reaction was stirred at room temperature. After 2 h, the reaction was diluted with 100 mL EtOAc, washed with 2 x 100 mL water and 3 x 100 mL brine. The organic layer was collected and concentrated under vacuum to give the title compound (630 mg, 1.04 mmol, 100% yield) as a yellow solid. LCMS m/z = 526 [M + H] + .
步骤2: (E)-1-((E)-4-氨基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 3盐酸盐Step 2: (E)-1-((E)-4-aminobut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 3-hydrochloride
向((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)氨基甲酸叔丁酯 (600mg, 1.142 mmol)在MeOH (5 mL)中的溶液加入在二氧杂环己烷中的4 M盐酸 (2.85 mL,11.42 mmol)并在室温搅拌该反应。 在3 h后,在真空下除去挥发物以得到作为橙色固体的标题化合物(650 mg, 1.04 mmol, 92%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 1.33(t, J=7.10 Hz, 3 H) 2.20 (s, 3 H) 3.32 - 3.42 (m, 3 H) 3.66 (br. s., 3 H)4.03 (s, 3 H) 4.54 (q, J=7.10 Hz, 2 H) 5.03 (br. s., 2 H) 5.60 - 5.71 (m, 1H) 5.97 (dt, J=15.59, 5.89 Hz, 1 H) 6.79 (br. s., 1 H) 7.52 - 7.61 (m, 2 H)7.90 (br. s., 1 H) 8.05 (br. s., 3 H) 8.22 (br. s., 1 H)。LCMS m/z = 426 [M +H]+。To a solution of tert-butyl ((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)carbamate (600 mg, 1.142 mmol) in MeOH (5 mL) was added 4 M hydrochloric acid in dioxane (2.85 mL, 11.42 mmol) and the reaction was stirred at room temperature. After 3 h, the volatiles were removed under vacuum to give the title compound as an orange solid (650 mg, 1.04 mmol, 92% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 1.33(t, J =7.10 Hz, 3 H) 2.20 (s, 3 H) 3.32 - 3.42 (m, 3 H) 3.66 (br. s., 3 H)4.03 (s, 3 H) 4.54 (q, J =7.10 Hz, 2 H) 5.03 (br. s., 2 H) 5.60 - 5.71 (m, 1H) 5.97 (dt, J =15.59, 5.89 Hz, 1 H) 6.79 (br. s., 1 H) 7.52 - 7.61 (m, 2 H)7.90 (br. s., 1 H) 8.05 (br. s., 3 H) 8.22 (br. s., 1 H). LCMS m/z = 426 [M +H] + .
中间体12Intermediate 12
(E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-吗啉代丙氧基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺( E )-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-morpholinopropoxy)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazol-5-carboxamide
步骤1: (E)-4-((4-((4-氨基甲酰基-2-(3-吗啉代丙氧基)-6-硝基苯基)氨基)丁-2-烯-1-基)氨基)-3-甲氧基-5-硝基苯甲酰胺Step 1: ( E )-4-((4-((4-carbamoyl-2-(3-morpholinopropoxy)-6-nitrophenyl)amino)but-2-en-1-yl)amino)-3-methoxy-5-nitrobenzamide
将(E)-4-((4-氨基丁-2-烯-1-基)氨基)-3-甲氧基-5-硝基苯甲酰胺, 盐酸盐(1.7 g, 5.37 mmol)、4-氯-3-(3-吗啉代丙氧基)-5-硝基苯甲酰胺 (1.65 g, 4.81mmol)、异丙醇 (15 mL)和DIPEA (2.94 mL, 16.85 mmol)分开到两个24-mL小瓶中。将该小瓶盖上盖子并在120℃加热42 h。通过过滤分离所得固体,采用异丙醇 (2 x 3 mL)漂洗以获得作为砖红色固体的(E)- 4-((4-((4-氨基甲酰基-2-(3-吗啉代丙氧基)-6-硝基苯基)氨基)丁-2-烯-1-基)氨基)-3-甲氧基-5-硝基苯甲酰胺 (1.95 g, 2.79 mmol, 51.9%收率)。LCMS (m/z): 588.2 [M + H]+。(E)-4-((4-aminobut-2-en-1-yl)amino)-3-methoxy-5-nitrobenzamide, hydrochloride (1.7 g, 5.37 mmol), 4-chloro-3-(3-morpholinopropoxy)-5-nitrobenzamide (1.65 g, 4.81 mmol), isopropanol (15 mL), and DIPEA (2.94 mL, 16.85 mmol) were separated into two 24-mL vials. The vials were capped and heated at 120 °C for 42 h. The resulting solid was isolated by filtration, rinsing with isopropanol (2 x 3 mL) to afford ( E )-4-((4-((4-carbamoyl-2-(3-morpholinopropoxy)-6-nitrophenyl)amino)but-2-en-1-yl)amino)-3-methoxy-5-nitrobenzamide (1.95 g, 2.79 mmol, 51.9% yield) as a brick red solid. LCMS ( m/z ): 588.2 [M+H] + .
步骤2: (E)-3-氨基-4-((4-((2-氨基-4-氨基甲酰基-6-(3-吗啉代丙氧基)苯基)氨基)丁-2-烯-1-基)氨基)-5-甲氧基苯甲酰胺Step 2: ( E )-3-amino-4-((4-((2-amino-4-carbamoyl-6-(3-morpholinopropoxy)phenyl)amino)but-2-en-1-yl)amino)-5-methoxybenzamide
在室温向在MeOH (83.0 mL)中的(E)-4-((4-((4-氨基甲酰基-2-(3-吗啉代丙氧基)-6-硝基苯基)氨基)丁-2-烯-1-基)氨基)-3-甲氧基-5-硝基苯甲酰胺 (4.6 g, 6.65mmol)加入在水(70 mL)中的次硫酸钠 (19.08 g, 93.0 mmol)。在15 min后,加入固体碳酸氢钠(24 g)。在10 min后,过滤该反应,并用MeOH (4 x 20 mL)漂洗该固体。将合并的滤液浓缩至硅藻土上,并通过干装载(dry-loading)至硅胶(80 g Gold柱)上,采用在DCM中的2- 40% (10:1 MeOH: 含水NH4OH)洗脱来纯化以得到作为深黄色膜的标题化合物(1.81 g,3.26 mmol, 49%收率)。1H NMR (400 MHz, DMSO-d 6 ) δ ppm 7.64 (br. s., 2 H), 6.99(br. s., 2 H), 6.85 (dd, J=5.07, 1.77 Hz, 2 H), 6.78 (dd, J=4.31, 1.77 Hz, 2H), 5.63 - 5.72 (m, 2 H), 4.66 (d, J=8.11 Hz, 4 H), 3.96 (t, J=6.21 Hz, 2 H),3.74 (s, 3 H), 3.51 - 3.60 (m, 6 H), 3.17 (br. s., 4 H), 2.43 (t, J=7.10 Hz,2 H), 2.35 (br. s., 4 H), 1.87 (t, J=6.72 Hz, 2 H); LCMS (m/z): 528.4 [M + H]+。To ( E )-4-((4-((4-carbamoyl-2-(3-morpholinopropoxy)-6-nitrophenyl)amino)but-2-en-1-yl)amino)-3-methoxy-5-nitrobenzamide (4.6 g, 6.65 mmol) in MeOH (83.0 mL) was added sodium sulfoxylate (19.08 g, 93.0 mmol) in water (70 mL) at room temperature. After 15 min, solid sodium bicarbonate (24 g) was added. After 10 min, the reaction was filtered and the solid was rinsed with MeOH (4 x 20 mL). The combined filtrates were concentrated onto celite and purified by dry-loading onto silica gel (80 g Gold column), eluting with 2-40% (10:1 MeOH:aq. NH 4 OH) in DCM to give the title compound (1.81 g, 3.26 mmol, 49% yield) as a dark yellow film. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 7.64 (br. s., 2 H), 6.99 (br. s., 2 H), 6.85 (dd, J =5.07, 1.77 Hz, 2 H), 6.78 (dd, J =4.31, 1.77 Hz, 2H), 5.63 - 5. 72 (m, 2 H), 4.66 (d, J =8.11 Hz, 4 H), 3.96 (t, J =6.21 Hz, 2 H), 3.74 (s, 3 H), 3.51 - 3.60 (m, 6 H), 3.17 (br. s., 4 H), 2.43 (t, J =7.10 Hz, 2 H), 2.35 (br. s., 4 H), 1.87 (t, J =6.72 Hz, 2 H); LCMS ( m/z ): 528.4 [M + H] + .
步骤3: (E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-吗啉代丙氧基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺Step 3: ( E )-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-morpholinopropoxy)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazol-5-carboxamide
在0℃向在DMF (6.97 mL)中的(E)-3-氨基-4-((4-((2-氨基-4-氨基甲酰基-6-(3-吗啉代丙氧基)苯基)氨基)-丁-2-烯-1-基)氨基)-5-甲氧基苯甲酰胺 (368 mg, 0.697mmol)加入在二氧杂环己烷中的0.4 M1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (2.0mL, 0.80 mmol)。在~10 min后,加入另一部分在二氧杂环己烷中的0.4 M 1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯(0.5 mL, 0.20 mmol),~15 min后加入最后一部分(0.5 mL,0.20 mmol)。在35 min总反应时间后,加入EDC (334 mg, 1.74 mmol),然后加入三乙胺(0.486 mL, 3.49 mmol)。使该混合物温热至室温并搅拌过夜(~14小时)。用3:1水: 饱和NH4Cl水溶液(40 mL)淬灭该反应并用3:1 氯仿: EtOH (2x40 mL)萃取。用水(20 mL)洗涤合并的有机相,经硫酸镁干燥并浓缩。通过硅胶层析法 (40 g柱, 2-40%梯度的[10:1MeOH: 含水NH4OH]/DCM)纯化所得残余物以提供作为桃色固体的标题化合物(361 mg,0.425 mmol, 60.9%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 1.20-1.35 (m, 6 H),1.55 - 1.73 (m, 2 H), 2.02 - 2.31 (m, 12 H), 3.46 (t, J=4.44 Hz, 4 H), 3.70(s, 3 H), 3.93 (t, J=5.96 Hz, 2 H), 4.40 - 4.68 (m, 4 H), 4.80 - 5.00 (m, 4H), 5.69 - 6.00 (m, 2 H), 6.41 - 6.74 (m, 2 H), 7.13 - 7.51 (m, 4 H), 7.56 -7.76 (m, 2 H), 7.99 (d, J=3.55 Hz, 2 H), 12.85 (br. s., 2 H)。LCMS (m/z):851.5 [M + H]+。To ( E )-3-amino-4-((4-((2-amino-4-carbamoyl-6-(3-morpholinopropoxy)phenyl)amino)-but-2-en-1-yl)amino)-5-methoxybenzamide (368 mg, 0.697 mmol) in DMF (6.97 mL) at 0° C. was added 0.4 M 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate in dioxane (2.0 mL, 0.80 mmol). After ~10 min, another portion of 0.4 M 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate in dioxane (0.5 mL, 0.20 mmol) was added and after ~15 min, the last portion (0.5 mL, 0.20 mmol) was added. After 35 min total reaction time, EDC (334 mg, 1.74 mmol) was added followed by triethylamine (0.486 mL, 3.49 mmol). The mixture was allowed to warm to room temperature and stirred overnight (~14 hours). The reaction was quenched with 3:1 water: saturated aqueous NH 4 Cl solution (40 mL) and extracted with 3:1 chloroform: EtOH (2x40 mL). The combined organic phases were washed with water (20 mL), dried over magnesium sulfate and concentrated. The resulting residue was purified by silica gel chromatography (40 g column, 2-40% gradient of [10:1 MeOH: aqueous NH 4 OH]/DCM) to provide the title compound (361 mg, 0.425 mmol, 60.9% yield) as a peach solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 1.20-1.35 (m, 6 H), 1.55 - 1.73 (m, 2 H), 2.02 - 2.31 (m, 12 H), 3.46 (t, J =4.44 Hz, 4 H), 3.70 (s, 3 H), 3.93 (t, J =5.96 Hz, 2 H), 4.40 - 4.68 (m, 4 H), 4.80 - 5.00 (m, 4H), 5.69 - 6.00 (m, 2 H), 6.41 - 6.74 (m, 2 H), 7.13 - 7.51 (m, 4 H), 7.56 -7.76 (m, 2 H), 7.99 (d, J =3.55 Hz, 2 H), 12.85 (br. s., 2 H). LCMS ( m/z ):851.5 [M + H] + .
中间体13Intermediate 13
2,2,3,3-四氟丁烷-1,4-二胺2,2,3,3-Tetrafluorobutane-1,4-diamine
步骤1: 双(4-甲基苯磺酸) 2,2,3,3-四氟丁烷-1,4-二基酯Step 1: 2,2,3,3-tetrafluorobutane-1,4-diyl bis(4-methylbenzenesulfonate)
在0℃经5 min向在吡啶(150 mL)中的2,2,3,3-四氟丁烷-1,4-二醇(10.0 g,61.7 mmol)加入4-甲基苯-1-磺酰氯(29.4 g, 154 mmol),然后将该反应加热至 55℃。在1天后,用冰水淬灭该反应,并通过过滤收集所得固体,溶解在DCM (200 mL)中并用5%H2SO4水溶液(100 mL X 3)洗涤。经Na2SO4干燥有机层并浓缩以得到作为白色固体的标题化合物(27.3 g, 58.0 mmol, 94%收率)。LCMS [M + H]+ = 470.9。4-Methylbenzene-1-sulfonyl chloride (29.4 g, 154 mmol) was added to 2,2,3,3-tetrafluorobutane-1,4-diol (10.0 g, 61.7 mmol) in pyridine (150 mL) at 0°C over 5 min, and the reaction was then heated to 55°C. After 1 day, the reaction was quenched with ice water, and the resulting solid was collected by filtration, dissolved in DCM (200 mL) and washed with 5% H 2 SO 4 aqueous solution (100 mL x 3). The organic layer was dried over Na 2 SO 4 and concentrated to give the title compound (27.3 g, 58.0 mmol, 94% yield) as a white solid. LCMS [M + H] + = 470.9.
步骤2: 1,4-二叠氮基-2,2,3,3-四氟丁烷Step 2: 1,4-Diazido-2,2,3,3-tetrafluorobutane
在110℃搅拌双(4-甲基苯磺酸) 2,2,3,3-四氟丁烷-1,4-二基酯 (10.0 g, 21.3mmol)和在DMF (40 mL)中的叠氮化钠 (5.53 g, 85.0 mmol)过夜。用NaClO (aq)淬灭该反应并用 DCM (5 mL X 3)萃取。用水(10 mL)洗涤合并的有机层,经Na2SO4干燥并浓缩以得到标题化合物(3.5 g, 16.5 mmol, 78%收率)。LCMS [M + H]+ = 213.1。Bis(4-methylbenzenesulfonic acid) 2,2,3,3-tetrafluorobutane-1,4-diyl ester (10.0 g, 21.3 mmol) and sodium azide (5.53 g, 85.0 mmol) in DMF (40 mL) were stirred at 110 ° C overnight. The reaction was quenched with NaClO (aq) and extracted with DCM (5 mL x 3). The combined organic layer was washed with water (10 mL), dried over Na 2 SO 4 and concentrated to give the title compound (3.5 g, 16.5 mmol, 78% yield). LCMS [M + H] + = 213.1.
步骤3: 2,2,3,3-四氟丁烷-1,4-二胺Step 3: 2,2,3,3-Tetrafluorobutane-1,4-diamine
向1,4-二叠氮基-2,2,3,3-四氟丁烷 (36.0 g, 170 mmol)在MeOH (350 mL)中的溶液加入10% 碳载钯 (18.1 g, 17.0 mmol)。在40℃在氢气(4 atm)下搅拌该反应混合物16 h。将该混合物过滤通过硅藻土垫,用MeOH洗涤并在真空中浓缩滤液以得到标题化合物(22.0 g, 124 mmol, 73%收率)。1H NMR (400 MHz, 氯仿-d) δ ppm 3.12 - 3.37 (m, 4H), 1.43 (br. s., 4 H)。To a solution of 1,4-diazide-2,2,3,3-tetrafluorobutane (36.0 g, 170 mmol) in MeOH (350 mL) was added 10% palladium on carbon (18.1 g, 17.0 mmol). The reaction mixture was stirred at 40 °C under hydrogen (4 atm) for 16 h. The mixture was filtered through a pad of celite, washed with MeOH and the filtrate was concentrated in vacuo to give the title compound (22.0 g, 124 mmol, 73% yield). 1 H NMR (400 MHz, CHLOROFORM- d ) δ ppm 3.12 - 3.37 (m, 4H), 1.43 (br. s., 4 H).
中间体14A和14BIntermediates 14A and 14B
7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(5-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺7-(3-((tert-Butyldimethylsilyl)oxy)propoxy)-1-(5-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-5-carboxamide
步骤1: 2,5-二叠氮基己烷Step 1: 2,5-Diazidohexane
向500-mL 圆底烧瓶加入2,5-二溴己烷 (10 g, 41.0 mmol)和DMF (100 mL)。向这个均相溶液加入叠氮化钠 (10.66 g, 164 mmol)。在80℃搅拌该非均相反应混合物1 h。将该混合物冷却至室温并加入水(100 mL)。用二乙醚(3 x 100 mL)萃取水相。用盐水(100mL)洗涤合并的有机相,经硫酸钠干燥,过滤并在真空下浓缩。获得作为黄色油的2,5-二叠氮基己烷 (8.54 g, 33.5 mmol, 83%收率, 66%纯度)。1H NMR (400 MHz, 氯仿-d) δ ppm1.30 (d, J=6.59 Hz, 6 H), 1.40 - 1.76 (m, 4 H), 3.35 - 3.68 (m, 2 H)。To a 500-mL round-bottom flask was added 2,5-dibromohexane (10 g, 41.0 mmol) and DMF (100 mL). To this homogeneous solution was added sodium azide (10.66 g, 164 mmol). The heterogeneous reaction mixture was stirred at 80 °C for 1 h. The mixture was cooled to room temperature and water (100 mL) was added. The aqueous phase was extracted with diethyl ether (3 x 100 mL). The combined organic phases were washed with brine (100 mL), dried over sodium sulfate, filtered and concentrated under vacuum. 2,5-diazidehexane (8.54 g, 33.5 mmol, 83% yield, 66% purity) was obtained as a yellow oil. 1H NMR (400 MHz, CHLOROFORM-d) δ ppm1.30 (d, J=6.59 Hz, 6 H), 1.40 - 1.76 (m, 4 H), 3.35 - 3.68 (m, 2 H).
步骤2: 己烷-2,5-二胺Step 2: Hexane-2,5-diamine
将2,5-二叠氮基己烷 (8.54 g, 50.8 mmol)溶解在MeOH (300 mL)中。将该溶液在单程经过ThalesNano H-Cube®系统(35℃, 25 bar氢气压力, 2 mL/min流速)中氢化。然后浓缩该溶液,并将粗产物用于接下来的反应中。获得作为无色油的己烷-2,5-二胺 (6.04g, 49.4 mmol, 97%收率)。1H NMR (400 MHz, 氯仿-d) δ ppm 1.09 (dd, J=6.21, 1.65Hz, 6 H), 1.21 - 1.62 (m, 8 H), 2.78 - 3.02 (m, 2 H)。2,5-diazidohexane (8.54 g, 50.8 mmol) was dissolved in MeOH (300 mL). The solution was hydrogenated in a single pass through a ThalesNano H- Cube® system (35°C, 25 bar hydrogen pressure, 2 mL/min flow rate). The solution was then concentrated and the crude product was used in the next reaction. Hexane-2,5-diamine (6.04 g, 49.4 mmol, 97% yield) was obtained as a colorless oil. 1 H NMR (400 MHz, chloroform- d ) δ ppm 1.09 (dd, J =6.21, 1.65Hz, 6 H), 1.21 - 1.62 (m, 8 H), 2.78 - 3.02 (m, 2 H).
步骤3: 3-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-((5-((4-氨基甲酰基-2-硝基苯基)氨基)己烷-2-基)氨基)-5-硝基苯甲酰胺Step 3: 3-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-((5-((4-carbamoyl-2-nitrophenyl)amino)hexan-2-yl)amino)-5-nitrobenzamide
向40-mL小瓶中放置 3-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氯-5-硝基苯甲酰胺 (中间体化合物4) (1.255 g, 3.23 mmol)、异丙醇 (8 mL)和DIPEA (1.879mL, 10.76 mmol)。向这个非均相混合物加入作为在异丙醇 (2 mL)中的溶液的己烷-2,5-二胺 (500 mg, 4.30 mmol)。将该小瓶盖上盖子并加热至110℃过夜(~14 h)。将该溶液冷却至室温。加入4-氟-3-硝基苯甲酰胺 (0.594 g, 3.23 mmol),然后加入DIPEA (1.879mL, 10.76 mmol)。将该反应再次加热至110℃持续2 h。在冷却至室温后形成固体。在过滤器上收集固体并用异丙醇 (每次2 mL)漂洗两次。通过硅胶层析法 (ISCO设备, 80 g SiO2筒, 2-20%梯度的MeOH/DCM)纯化这个粗固体。合并相应的级分并浓缩。获得作为橙色玻璃膜(非对映异构体混合物)的3-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-((5-((4-氨基甲酰基-2-硝基苯基)氨基)己烷-2-基)氨基)-5-硝基苯甲酰胺 (300 mg, 0.450 mmol,10.47%收率)。LCMS (m/z): 633.5 [M + H]+。Into a 40-mL vial was placed 3-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-chloro-5-nitrobenzamide (Intermediate Compound 4) (1.255 g, 3.23 mmol), isopropanol (8 mL), and DIPEA (1.879 mL, 10.76 mmol). To this heterogeneous mixture was added hexane-2,5-diamine (500 mg, 4.30 mmol) as a solution in isopropanol (2 mL). The vial was capped and heated to 110 °C overnight (~14 h). The solution was cooled to room temperature. 4-Fluoro-3-nitrobenzamide (0.594 g, 3.23 mmol) was added followed by DIPEA (1.879 mL, 10.76 mmol). The reaction was heated to 110 °C again for 2 h. A solid formed after cooling to room temperature. The solid was collected on the filter and rinsed twice with isopropanol (2 mL each time). The crude solid was purified by silica gel chromatography (ISCO equipment, 80 g SiO2 cartridge, 2-20% gradient of MeOH/DCM). The corresponding fractions were combined and concentrated. 3-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-((5-((4-carbamoyl-2-nitrophenyl)amino)hexane-2-yl)amino)-5-nitrobenzamide (300 mg, 0.450 mmol, 10.47% yield) was obtained as an orange glass film (mixture of diastereomers). LCMS ( m/z ): 633.5 [M + H] + .
步骤4: 3-氨基-4-((5-((2-氨基-4-氨基甲酰基苯基)氨基)己烷-2-基)氨基)-5-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)苯甲酰胺Step 4: 3-amino-4-((5-((2-amino-4-carbamoylphenyl)amino)hexan-2-yl)amino)-5-(3-((tert-butyldimethylsilyl)oxy)propoxy)benzamide
向125-mL Erlenmeyer烧瓶加入3-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-((5-((4-氨基甲酰基-2-硝基苯基)氨基)己烷-2-基)氨基)-5-硝基苯甲酰胺 (386 mg,0.610 mmol)和MeOH (40 mL)。使用ThalesNano H-Cube®系统(5% Pd/C筒, 30℃, 10 bar氢气压力, 1.5 mL/min流速)氢化该溶液。在两次循环后,完成还原。浓缩该溶液以获得3-氨基-4-((5-((2-氨基-4-氨基甲酰基苯基)氨基)己烷-2-基)氨基)-5-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)苯甲酰胺 (352 mg, 0.602 mmol, 99%收率)。LCMS (m/z): 573.5[M + H]+。To a 125-mL Erlenmeyer flask was added 3-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-((5-((4-carbamoyl-2-nitrophenyl)amino)hexan-2-yl)amino)-5-nitrobenzamide (386 mg, 0.610 mmol) and MeOH (40 mL). The solution was hydrogenated using a ThalesNano H- Cube® system (5% Pd/C cartridge, 30° C., 10 bar hydrogen pressure, 1.5 mL/min flow rate). After two cycles, the reduction was complete. The solution was concentrated to afford 3-amino-4-((5-((2-amino-4-carbamoylphenyl)amino)hexan-2-yl)amino)-5-(3-((tert-butyldimethylsilyl)oxy)propoxy)benzamide (352 mg, 0.602 mmol, 99% yield). LCMS ( m/z ): 573.5[M + H] + .
步骤5: 7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(5-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 5: 7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(5-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide
向100-mL圆底烧瓶加入3-氨基-4-((5-((2-氨基-4-氨基甲酰基苯基)氨基)己烷-2-基)氨基)-5-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)苯甲酰胺 (352 mg, 0.614mmol)和DMF (6.1 mL)。在0℃加入1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (中间体化合物8)的溶液 (~0.4 M 在二氧杂环己烷中, 2.75 mL, 1.100 mmol)并搅拌该混合物15min。然后,加入EDC (295 mg, 1.536 mmol),之后加入三乙胺 (0.428 mL, 3.07 mmol)。在室温搅拌该反应过夜(~14 h)。在50 mL 乙酸乙酯和50 mL饱和氯化铵水溶液和水的1:1混合物之间分配该反应。分离各层。采用乙酸乙酯 (2 x 25 mL)萃取水层。采用盐水洗涤合并的有机层,经硫酸钠干燥并在真空下浓缩。通过反相制备型层析法(双相ISCO系统, GeminiC18, 5 um, 50 x 30 mm柱; 40-70%梯度的MeCN/水(含NH4OH改性剂))进行的纯化能够实现第一个洗脱出的非对映异构体和第二个洗脱出的非对映异构体的分离和表征。预期各非对映异构体为外消旋体(即对映异构体对)。分别合并含第一个洗脱出的非对映异构体和第二个洗脱出的非对映异构体的级分并干燥以分别提供作为白色固体的中间体XA和XB。To a 100-mL round-bottom flask was added 3-amino-4-((5-((2-amino-4-carbamoylphenyl)amino)hexan-2-yl)amino)-5-(3-((tert-butyldimethylsilyl)oxy)propoxy)benzamide (352 mg, 0.614 mmol) and DMF (6.1 mL). A solution of 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (Intermediate Compound 8) (~0.4 M in dioxane, 2.75 mL, 1.100 mmol) was added at 0 °C and the mixture was stirred for 15 min. Then, EDC (295 mg, 1.536 mmol) was added followed by triethylamine (0.428 mL, 3.07 mmol). The reaction was stirred at room temperature overnight (~14 h). The reaction was distributed between 50 mL of ethyl acetate and 50 mL of a 1:1 mixture of saturated aqueous ammonium chloride and water. The layers were separated. The aqueous layer was extracted with ethyl acetate (2 x 25 mL). The combined organic layers were washed with brine, dried over sodium sulfate and concentrated under vacuum. Purification by reverse phase preparative chromatography (biphasic ISCO system, GeminiC18, 5 um, 50 x 30 mm column; 40-70% gradient of MeCN/water (containing NH 4 OH modifier)) enabled separation and characterization of the first eluting diastereomer and the second eluting diastereomer. Each diastereomer was expected to be a racemate (i.e., an enantiomeric pair). The fractions containing the first eluting diastereomer and the second eluting diastereomer were combined and dried to provide intermediates XA and XB as white solids, respectively.
中间体14A (第一个洗脱出的非对映异构体)Intermediate 14A (first eluting diastereomer)
外消旋7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(5-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (200 mg, 0.223 mmol, 36.4%收率)。Racemic 7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(5-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (200 mg, 0.223 mmol, 36.4% yield).
LCMS (m/z): 895.6 [M + H]+; 1.37 min保留时间(Acquity UPLC CSH C18,1.7 um, 50 mm x 2.1 mm柱; 40℃; 经1.5 min的3-95%梯度, MeCN/10 mM碳酸氢铵在水中的溶液,其用25% 氢氧化铵溶液调节至pH 10)。LCMS ( m/z ): 895.6 [M + H] + ; 1.37 min retention time (Acquity UPLC CSH C18, 1.7 um, 50 mm x 2.1 mm column; 40°C; 3-95% gradient over 1.5 min, MeCN/10 mM ammonium bicarbonate in water adjusted to pH 10 with 25% ammonium hydroxide solution).
1H NMR (400 MHz, 甲醇-d 4 ) δ ppm 7.98 (br. s., 0.54), 7.91 (s, 0.51),7.80 (t, J=7.22 Hz, 1.06), 7.45 - 7.65 (m, 2.01), 7.40 (s, 0.52), 7.35 (s,0.54), 6.34 - 6.79 (m, 2.09), 5.44 (br. s., 1.20), 4.65 (m, 4.10), 4.28 (m,1.56), 3.99 (br. s., 0.63), 3.79 (m, 1.06), 3.70 (br. s., 0.65), 3.61 (br.s., 0.58), 2.98 (br. s., 2.98), 2.35 - 2.65 (m, 1.40), 2.28 (s, 1.49), 2.23(m, 3.16), 2.17 (br. s., 1.47), 1.99 (br. s., 0.98), 1.81 (br. s., 2.70),1.62 (d, J=6.84 Hz, 1.87), 1.55 (d, J=6.84 Hz, 3.08), 1.50 (d, J=6.59 Hz,1.46), 1.29 - 1.47 (m, 6.68), 0.87 (s, 9.20), 0.02 (s, 6.00)。 1 H NMR (400 MHz, methanol- d 4 ) δ ppm 7.98 (br. s., 0.54), 7.91 (s, 0.51), 7.80 (t, J =7.22 Hz, 1.06), 7.45 - 7.65 (m, 2.01 ), 7.40 (s, 0.52), 7.35 (s,0.54), 6.34 - 6.79 (m, 2.09), 5.44 (br. s., 1.20), 4.65 (m, 4.10), 4.28 (m,1.56), 3.99 (br. s., 0.63), 3.79 (m, 1.06), 3.70 (br. s., 0.65), 3.61 (br.s., 0.58), 2.98 (br.s., 2.98), 2.35 - 2.65 (m, 1.40), 2.28 (s, 1.49), 2.23(m, 3.16), 2.17 (br.s., 1.47 ), 1.99 (br. s., 0.98), 1.81 (br. s., 2.70), 1.62 (d, J =6.84 Hz, 1.87), 1.55 (d, J =6.84 Hz, 3.08), 1.50 (d, J =6.59 Hz,1.46), 1.29 - 1.47 (m, 6.68), 0.87 (s, 9.20), 0.02 (s, 6.00).
中间体14B (第二个洗脱出的非对映异构体)Intermediate 14B (second eluting diastereomer)
外消旋7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(5-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (210 mg, 0.235 mmol, 38.2%收率),作为白色固体。Racemic 7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(5-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (210 mg, 0.235 mmol, 38.2% yield) as a white solid.
LCMS (m/z): 895.6 [M + H]+; 1.42 min保留时间(Acquity UPLC CSH C18,1.7 um, 50 mm x 2.1 mm柱; 40℃; 经1.5 min的3-95%梯度, MeCN/10 mM碳酸氢铵在水中的溶液,其用25% 氢氧化铵溶液调节至pH 10)。LCMS ( m/z ): 895.6 [M + H] + ; 1.42 min retention time (Acquity UPLC CSH C18, 1.7 um, 50 mm x 2.1 mm column; 40°C; 3-95% gradient over 1.5 min, MeCN/10 mM ammonium bicarbonate in water adjusted to pH 10 with 25% ammonium hydroxide solution).
1H NMR (400 MHz, 甲醇-d 4) δ ppm 7.80 (br. s., 2.29), 7.60 (br. s.,3.77), 7.43 (br. s., 3.64), 7.24 (br. s., 3.24), 7.10 (s, 3.41), 6.81 (br.s., 3.41), 5.62 - 6.31 (br. s., 2.02), 5.35 - 5.61 (m, 3.60), 5.26 (br. s.,2.06), 4.57 - 4.84 (m, 11.14), 4.25 - 4.52 (m, 4.04), 4.11 (br. s., 1.46),3.96 (t, J=5.96 Hz, 2.56), 3.84 (br. s., 0.87), 3.59 (br. s., 4.64), 2.85 (q,J=12.17 Hz, 0.97), 2.11 - 2.40 (m, 20.85), 2.06 (s, 5.95), 1.69 - 1.99 (m,8.53), 1.20 - 1.68 (m, 44.08), 0.96 (s, 10.71), 0.86 (s, 21.28), 0.14 (d, J=5.07 Hz, 7.03), 0.01 (d, J=3.80 Hz, 14.00)。 1 H NMR (400 MHz, methanol- d 4 ) δ ppm 7.80 (br. s., 2.29), 7.60 (br. s., 3.77), 7.43 (br. s., 3.64), 7.24 (br. s. , 3.24), 7.10 (s, 3.41), 6.81 (br.s., 3.41), 5.62 - 6.31 (br. s., 2.02), 5.35 - 5.61 (m, 3.60), 5.26 (br. s., 2.06 ), 4.57 - 4.84 (m, 11.14), 4.25 - 4.52 (m, 4.04), 4.11 (br. s., 1.46), 3.96 (t, J =5.96 Hz, 2.56), 3.84 (br. s., 0.87), 3.59 (br. s., 4.64), 2.85 (q, J =12.17 Hz, 0.97), 2.11 - 2.40 (m, 20.85), 2.06 (s, 5.95), 1.69 - 1.99 (m,8.53), 1.20 - 1.68 (m, 44.08), 0.96 (s, 10.71), 0.86 (s, 21.28), 0.14 (d, J =5.07 Hz, 7.03), 0.01 (d, J =3.80 Hz, 14.00).
中间体15Intermediate 15
4-氯-3-((4-甲氧基苄基)氧基)-5-硝基苯甲酰胺4-Chloro-3-((4-methoxybenzyl)oxy)-5-nitrobenzamide
向溶解在DMF (7 mL)中的4-氯-3-羟基-5-硝基苯甲酰胺 (942 mg, 4.35 mmol)加入Cs2CO3 (1.559 g, 4.78 mmol),然后加入4-甲氧基苄基氯(0.622 mL, 4.57 mmol)。在室温搅拌该反应混合物24 h。在剧烈搅拌下,逐滴加入水(15 mL)并搅拌所得固体5min,通过过滤收集并用水漂洗以获得作为浅橙色固体的标题化合物(1.26 g, 3.74 mmol, 82%收率)。1H NMR (400 MHz, CDCl3) δ ppm 7.80 (d, J=1.8 Hz, 1 H), 7.76 (d, J=1.8 Hz,1 H), 7.43 (d, J=8.6 Hz, 2 H), 6.98 (d, J=8.6 Hz, 2 H), 6.13 (br. s., 1 H),5.82 (br. s., 1 H), 5.25 (s, 2 H), 3.87 (s, 3 H); LCMS (m/z): 337.1 [M + H]+。To 4-chloro-3-hydroxy-5-nitrobenzamide (942 mg, 4.35 mmol) dissolved in DMF (7 mL) was added Cs 2 CO 3 (1.559 g, 4.78 mmol) followed by 4-methoxybenzyl chloride (0.622 mL, 4.57 mmol). The reaction mixture was stirred at room temperature for 24 h. Under vigorous stirring, water (15 mL) was added dropwise and the resulting solid was stirred for 5 min, collected by filtration and rinsed with water to obtain the title compound (1.26 g, 3.74 mmol, 82% yield) as a light orange solid. 1 H NMR (400 MHz, CDCl 3 ) δ ppm 7.80 (d, J=1.8 Hz, 1 H), 7.76 (d, J=1.8 Hz, 1 H), 7.43 (d, J=8.6 Hz, 2 H), 6.98 (d, J=8.6 Hz, 2 H), 6.13 (br. s., 1 H),5. 82 (br. s., 1 H), 5.25 (s, 2 H), 3.87 (s, 3 H); LCMS ( m/z ): 337.1 [M + H] + .
中间体16Intermediate 16
(E)-2,3-二甲基丁-2-烯-1,4-二胺, 2盐酸盐(E)-2,3-Dimethylbut-2-ene-1,4-diamine, 2-hydrochloride
步骤1: (E)-2,2'-(2,3-二甲基丁-2-烯-1,4-二基)双(异吲哚啉-1,3-二酮)Step 1: (E)-2,2'-(2,3-dimethylbut-2-ene-1,4-diyl)bis(isoindoline-1,3-dione)
向(E)-1,4-二溴-2,3-二甲基丁-2-烯 (29.5 g, 122 mmol)在DMF (244 mL)中的溶液加入邻苯二甲酰亚胺钾 (45.2 g, 244 mmol)。在室温搅拌白色悬浮液过夜。将该反应倾倒至水(2 L)中并过滤所得白色悬浮液。空气干燥滤饼(48 h)以得到作为白色固体的(E)-2,2'-(2,3-二甲基丁-2-烯-1,4-二基)双(异吲哚啉-1,3-二酮) (37 g, 99 mmol,81%收率)。该固体不经进一步纯化而使用。1H NMR (400 MHz, 氯仿-d) δ ppm 7.84 -7.93 (m, 4 H), 7.72 - 7.81 (m, 4 H), 4.39 (s, 4 H), 1.95 (s, 6 H)。To a solution of (E)-1,4-dibromo-2,3-dimethylbut-2-ene (29.5 g, 122 mmol) in DMF (244 mL) was added potassium phthalimide (45.2 g, 244 mmol). The white suspension was stirred at room temperature overnight. The reaction was poured into water (2 L) and the resulting white suspension was filtered. The filter cake was air dried (48 h) to give (E)-2,2'-(2,3-dimethylbut-2-ene-1,4-diyl)bis(isoindoline-1,3-dione) (37 g, 99 mmol, 81% yield) as a white solid. The solid was used without further purification. 1 H NMR (400 MHz, chloroform- d ) δ ppm 7.84 -7.93 (m, 4 H), 7.72 - 7.81 (m, 4 H), 4.39 (s, 4 H), 1.95 (s, 6 H).
步骤2: (E)-2,3-二甲基丁-2-烯-1,4-二胺, 2盐酸盐Step 2: (E)-2,3-Dimethylbut-2-ene-1,4-diamine, 2-hydrochloride
向(E)-2,2'-(2,3-二甲基丁-2-烯-1,4-二基)双(异吲哚啉-1,3-二酮) (15.3 g,40.9 mmol)在EtOH (332 mL)中的混合物加入肼一水合物(6.01 mL, 123 mmol)。在80℃加热该反应。在3 h后,将该反应冷却至室温。过滤稠白色混合物,用乙醇洗涤滤饼,并浓缩滤液至干。在水(150 mL)和EtOAc (150 mL)之间分配来自滤液的所得白色固体。将水层浓缩至干以获得粘性黄色色调的油。用1 N HCl (250 mL)和EtOAc (250 mL)处理粘性油。通过过滤除去出现的白色沉淀物(副产物)。将滤液转移至分液漏斗。分离水层;过滤以除去剩余的白色固体并浓缩至干以获得作为带灰色的粉色(greyish-pink)固体的(E)-2,3-二甲基丁-2-烯-1,4-二胺, 2盐酸盐 (4.2 g, 22.45 mmol, 54.9%收率)。该物质不经进一步纯化而使用。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.23 (br. s., 6 H), 3.45 (q, J=5.75 Hz,4 H), 1.83 (s, 6 H)。To a mixture of (E)-2,2'-(2,3-dimethylbut-2-ene-1,4-diyl)bis(isoindoline-1,3-dione) (15.3 g, 40.9 mmol) in EtOH (332 mL) was added hydrazine monohydrate (6.01 mL, 123 mmol). The reaction was heated at 80°C. After 3 h, the reaction was cooled to room temperature. The thick white mixture was filtered, the filter cake was washed with ethanol, and the filtrate was concentrated to dryness. The resulting white solid from the filtrate was distributed between water (150 mL) and EtOAc (150 mL). The aqueous layer was concentrated to dryness to obtain a viscous yellow hued oil. The viscous oil was treated with 1 N HCl (250 mL) and EtOAc (250 mL). The white precipitate (by-product) that appeared was removed by filtration. The filtrate was transferred to a separatory funnel. The aqueous layer was separated; filtered to remove the remaining white solid and concentrated to dryness to obtain (E)-2,3-dimethylbut-2-ene-1,4-diamine, 2 hydrochloride (4.2 g, 22.45 mmol, 54.9% yield) as a greyish-pink solid. This material was used without further purification. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.23 (br. s., 6 H), 3.45 (q, J =5.75 Hz, 4 H), 1.83 (s, 6 H).
中间体17Intermediate 17
(E)-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)氨基甲酸叔丁酯tert-Butyl (E)-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)carbamate
步骤1: (E)-(4-((2-氨基-4-氨基甲酰基-6-甲氧基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯Step 1: tert-Butyl (E)-(4-((2-amino-4-carbamoyl-6-methoxyphenyl)amino)but-2-en-1-yl)carbamate
向2-L 圆底烧瓶加入(E)-(4-((4-氨基甲酰基-2-甲氧基-6-硝基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯 (25.8 g, 67.8 mmol)和甲醇(484 mL)。将这个橙色非均相溶液冷却至0℃。在0℃搅拌20 min后,加入氢氧化铵溶液(29% wt, 91 mL, 678 mmol),然后加入作为在水(194 mL)中的溶液的次硫酸钠 (85% wt, 70.0 g, 342 mmol)。从冰浴中除去烧瓶并在室温搅拌。该非均相混合物缓慢变色-从橙色变为灰白色。在室温搅拌3 h后,加入水(~800 mL)直到获得透明溶液。使用减压蒸发甲醇。过滤掉在蒸发期间形成的白色固体并用水洗涤两次(每次300 mL)。空气干燥该固体16 h,然后在50 ℃在真空炉中5 h。获得作为灰白色固体的(E)-(4-((2-氨基-4-氨基甲酰基-6-甲氧基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯 (19.34 g, 54.1 mmol, 80%收率)。通过HPLC、LCMS和1H NMR判断这个固体的纯度为98%。1H NMR (400 MHz, DMSO-d 6) δ ppm 7.62 (br. s., 1 H) 6.98 (br. s., 1H) 6.92 (t, J=5.45 Hz, 1 H) 6.87 (d, J=1.77 Hz, 1 H) 6.79 (d, J=1.77 Hz, 1 H)5.57 (qt, J=15.27, 5.23 Hz, 2 H) 4.67 (br. s., 2 H) 3.82 (br. s., 1 H) 3.76(s, 3 H) 3.51 (dd, J=12.29, 5.70 Hz, 4 H) 1.37 (s, 9 H)。LCMS (m/z): 351.1 (M+ H)+。To a 2-L round bottom flask was added tert-butyl (E)-(4-((4-carbamoyl-2-methoxy-6-nitrophenyl)amino)but-2-en-1-yl)carbamate (25.8 g, 67.8 mmol) and methanol (484 mL). The orange heterogeneous solution was cooled to 0°C. After stirring at 0°C for 20 min, ammonium hydroxide solution (29% wt, 91 mL, 678 mmol) was added, followed by sodium sulfoxylate (85% wt, 70.0 g, 342 mmol) as a solution in water (194 mL). The flask was removed from the ice bath and stirred at room temperature. The heterogeneous mixture slowly changed color - from orange to off-white. After stirring at room temperature for 3 h, water (~800 mL) was added until a clear solution was obtained. The methanol was evaporated using reduced pressure. The white solid formed during evaporation was filtered off and washed twice with water (300 mL each). The solid was air dried for 16 h and then in a vacuum oven at 50 °C for 5 h. (E)-tert-butyl(4-((2-amino-4-carbamoyl-6-methoxyphenyl)amino)but-2-en-1-yl)carbamate (19.34 g, 54.1 mmol, 80% yield) was obtained as an off-white solid. The purity of this solid was 98% as judged by HPLC, LCMS and 1H NMR. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 7.62 (br. s., 1 H) 6.98 (br. s., 1H) 6.92 (t, J =5.45 Hz, 1 H) 6.87 (d, J =1.77 Hz, 1 H) 6.79 (d, J =1.77 Hz, 1 H)5.57 (qt, J =15.27, 5.23 Hz, 2 H) 4.67 (br. s., 2 H) 3.82 (br. s., 1 H) 3.76 (s, 3 H) 3.51 (dd, J =12.29, 5.70 Hz, 4 H) 1.37 (s, 9 H). LCMS ( m/z ): 351.1 (M+ H) + .
步骤2: (E)-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)氨基甲酸叔丁酯Step 2: tert-Butyl (E)-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)carbamate
向2-L圆底烧瓶放入(E)-(4-((2-氨基-4-氨基甲酰基-6-甲氧基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯 (19.34 g, 55.2 mmol)和DMF (184 mL)。将这个溶液冷却至0℃。在0℃搅拌20 min后,加入作为约1.0 M 在二氧杂环己烷中的溶液的1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (44.2 mL, 44.2 mmol)。在0℃搅拌10 min后,完全形成中间体硫脲。加入EDC (15.87 g, 83 mmol)和DIEA (28.9 mL, 166 mmol)。使该反应温热至室温并搅拌过夜(~14 h)。向非均相反应混合物加入250 mL 饱和氯化铵水溶液和750 mL水的混合物。在室温搅拌这个非均相混合物1 h。将固体过滤掉并用水(每次200 mL)漂洗两次。在50℃在真空炉中干燥该灰白色固体3天。获得作为白色固体的(E)-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)氨基甲酸叔丁酯 (21.23 g, 41.5 mmol, 75%收率),通过LCMS、HPLC和1H NMR判断其纯度为~100%。1H NMR (400 MHz, DMSO-d 6) δ ppm 12.84 (br. s., 1 H) 8.00 (br. s., 1 H)7.67 (s, 1 H) 7.30 - 7.45 (m, 2 H) 6.86 - 7.00 (m, 1 H) 6.64 (s, 1 H) 5.54 -5.80 (m, 2 H) 4.92 (d, J=4.82 Hz, 2 H) 4.61 (q, J=7.01 Hz, 2 H) 3.97 (s, 3 H)3.50 (br. s., 2 H) 2.18 (s, 3 H) 1.11 - 1.41 (m, 12 H)。LCMS (m/z): 512.5 (M +H)+。Into a 2-L round bottom flask was placed tert-butyl (E)-(4-((2-amino-4-carbamoyl-6-methoxyphenyl)amino)but-2-en-1-yl)carbamate (19.34 g, 55.2 mmol) and DMF (184 mL). This solution was cooled to 0°C. After stirring at 0°C for 20 min, 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (44.2 mL, 44.2 mmol) was added as a ca. 1.0 M solution in dioxane. After stirring at 0°C for 10 min, the intermediate thiourea was completely formed. EDC (15.87 g, 83 mmol) and DIEA (28.9 mL, 166 mmol) were added. The reaction was allowed to warm to room temperature and stirred overnight (~14 h). To the heterogeneous reaction mixture was added a mixture of 250 mL saturated aqueous ammonium chloride and 750 mL water. This heterogeneous mixture was stirred at room temperature for 1 h. The solid was filtered off and rinsed twice with water (200 mL each time). The off-white solid was dried in a vacuum oven at 50 °C for 3 days. Tert-butyl (E)-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)carbamate (21.23 g, 41.5 mmol, 75% yield) was obtained as a white solid with a purity of ~100% as judged by LCMS, HPLC and 1 H NMR. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 12.84 (br. s., 1 H) 8.00 (br. s., 1 H)7.67 (s, 1 H) 7.30 - 7.45 (m, 2 H) 6.86 - 7.00 (m, 1 H) 6.64 (s, 1 H) 5.54 -5 .80 (m, 2 H) 4.92 (d, J =4.82 Hz, 2 H) 4.61 (q, J =7.01 Hz, 2 H) 3.97 (s, 3 H)3.50 (br. s., 2 H) 2.18 (s, 3 H) 1.11 - 1.41 (m, 12 H). LCMS ( m/z ): 512.5 (M +H) + .
中间体18Intermediate 18
(E)-1-(4-氨基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐(E)-1-(4-aminobut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide, 2-hydrochloride
向1-L圆底烧瓶加入(E)-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)氨基甲酸叔丁酯 (21.23 g, 41.5mmol)、乙醇(234 mL)和叔丁基甲基醚 (96 mL)。向这个非均相溶液加入作为在二氧杂环己烷中的4M 溶液的HCl (114 mL, 456 mmol)。在添加HCl期间,溶液从非均相变成具有透明黄色的均相。在室温搅拌该反应过夜。到了第二天早上,已经沉淀出白色固体。加入更多4MHCl 溶液(15.56 mL, 62.2 mmol)并再搅拌该混合物9 h直到反应完全。将白色固体过滤掉并用1:4 乙醇(200 mL)/TBME (800 mL)混合物漂洗。在真空炉中干燥所得固体过夜(50℃)。获得作为白色固体的(E)-1-(4-氨基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐 (22.56 g, 44.2 mmol, 107%收率),通过LCMS、HPLC和1H NMR判断其纯度为95%。1H NMR (400 MHz, DMSO-d 6) δ ppm12.88 (br. s., 1 H) 7.77 - 8.18 (m, 4 H) 7.68 (d, J=1.27 Hz, 1 H) 7.25 - 7.51(m, 2 H) 6.68 (s, 1 H) 6.04 (dt, J=15.52, 5.80 Hz, 1 H) 5.53 - 5.78 (m, 1 H)4.99 (d, J=5.32 Hz, 2 H) 4.61 (q, J=7.10 Hz, 2 H) 3.99 (s, 3 H) 3.27 - 3.57(m, 2 H) 2.19 (s, 3 H) 1.36 (t, J=7.10 Hz, 3 H)。LCMS (m/z): 412.3 (M + H)+。To a 1-L round bottom flask was added tert-butyl (E)-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)carbamate (21.23 g, 41.5 mmol), ethanol (234 mL) and tert-butyl methyl ether (96 mL). To this heterogeneous solution was added HCl (114 mL, 456 mmol) as a 4 M solution in dioxane. During the addition of HCl, the solution changed from heterogeneous to homogeneous with a clear yellow color. The reaction was stirred at room temperature overnight. By the next morning, a white solid had precipitated. More 4M HCl solution (15.56 mL, 62.2 mmol) was added and the mixture was stirred for an additional 9 h until the reaction was complete. The white solid was filtered off and rinsed with a 1:4 ethanol (200 mL)/TBME (800 mL) mixture. The resulting solid was dried in a vacuum oven overnight (50°C). (E)-1-(4-aminobut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide, 2 hydrochloride (22.56 g, 44.2 mmol, 107% yield) was obtained as a white solid with a purity of 95% as judged by LCMS, HPLC and 1 H NMR. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm12.88 (br. s., 1 H) 7.77 - 8.18 (m, 4 H) 7.68 (d, J =1.27 Hz, 1 H) 7.25 - 7.51 (m, 2 H) 6.68 (s, 1 H) 6.04 (dt, J =15. 52, 5.80 Hz, 1 H) 5.53 - 5.78 (m, 1 H)4.99 (d, J =5.32 Hz, 2 H) 4.61 (q, J =7.10 Hz, 2 H) 3.99 (s, 3 H) 3.27 - 3.57(m, 2 H) 2.19 (s, 3 H) 1.36 (t, J =7.10 Hz, 3 H). LCMS ( m/z ): 412.3 (M + H) + .
中间体19Intermediate 19
(E)-2-(4-氨基-2,3-二甲基丁-2-烯-1-基)异吲哚啉-1,3-二酮(E)-2-(4-amino-2,3-dimethylbut-2-en-1-yl)isoindoline-1,3-dione
步骤1: (3r,5r,7r)-1-((E)-4-溴-2,3-二甲基丁-2-烯-1-基)-1,3,5,7-四氮杂金刚烷-1-氮鎓, 溴化物Step 1: (3r,5r,7r)-1-((E)-4-Bromo-2,3-dimethylbut-2-en-1-yl)-1,3,5,7-tetraazaadamantan-1-ium bromide
经2 min向(E)-1,4-二溴-2,3-二甲基丁-2-烯 (13.59 g, 50.6 mmol)在DCM(200 mL)中的溶液逐份加入1,3,5,7-四氮杂金刚烷 (7.09 g, 50.6 mmol)。搅拌该反应25min并过滤所得固体,采用DCM漂洗并干燥以获得作为白色固体的(3r,5r,7r)-1-((E)-4-溴-2,3-二甲基丁-2-烯-1-基)-1,3,5,7-四氮杂金刚烷-1-氮鎓, 溴化物(16.7 g, 43.7mmol, 86%收率)。LCMS (m/z): 301.1 [M]+。To a solution of (E)-1,4-dibromo-2,3-dimethylbut-2-ene (13.59 g, 50.6 mmol) in DCM (200 mL) was added 1,3,5,7-tetraazaadamantane (7.09 g, 50.6 mmol) portionwise over 2 min. The reaction was stirred for 25 min and the resulting solid was filtered, rinsed with DCM and dried to obtain (3r,5r,7r)-1-((E)-4-bromo-2,3-dimethylbut-2-ene-1-yl)-1,3,5,7-tetraazaadamantane-1-azonia, bromide (16.7 g, 43.7 mmol, 86% yield) as a white solid. LCMS ( m/z ): 301.1 [M] + .
步骤2: (3r,5r,7r)-1-((E)-4-(1,3-二氧代异吲哚啉-2-基)-2,3-二甲基丁-2-烯-1-基)-1,3,5,7-四氮杂金刚烷-1-氮鎓, 溴化物Step 2: (3r,5r,7r)-1-((E)-4-(1,3-dioxoisoindolin-2-yl)-2,3-dimethylbut-2-en-1-yl)-1,3,5,7-tetraazaadamantan-1-ium bromide
向(3r,5r,7r)-1-((E)-4-溴-2,3-二甲基丁-2-烯-1-基)-1,3,5,7-四氮杂金刚烷-1-氮鎓, 溴化物(16.7 g, 43.7 mmol)在丙酮(200 mL)中的悬浮液加入邻苯二甲酰亚胺钾(Potassium 1,3-dioxoisoindolin-2-ide)(8.09 g, 43.7 mmol)。在55℃加热该反应混合物1.5 h。经过接下来的2.5 h,用另外的邻苯二甲酰亚胺钾处理该反应直到起始原料耗尽。将该反应混合物移除热源,搅拌10 min,然后在仍然温热的同时过滤。用丙酮漂洗该固体并干燥以得到 20.4 g粗产物。在冷水(冰浴)中搅拌该粗产物5 min。在过滤器上收集该固体,用冷水漂洗,并干燥以提供作为浅黄色固体的(3r,5r,7r)-1-((E)-4-(1,3-二氧代异吲哚啉-2-基)-2,3-二甲基丁-2-烯-1-基)-1,3,5,7-四氮杂金刚烷-1-氮鎓, 溴化物(10.3 g, 22.9 mmol, 52.6%收率)。LCMS (m/z): 368.2 [M]+。To a suspension of (3r,5r,7r)-1-((E)-4-bromo-2,3-dimethylbut-2-en-1-yl)-1,3,5,7-tetraazaadamantane-1-azonia, bromide (16.7 g, 43.7 mmol) in acetone (200 mL) was added Potassium 1,3-dioxoisoindolin-2-ide (8.09 g, 43.7 mmol). The reaction mixture was heated at 55 °C for 1.5 h. Over the next 2.5 h, the reaction was treated with additional potassium phthalimide until the starting material was consumed. The reaction mixture was removed from the heat source, stirred for 10 min, and then filtered while still warm. The solid was rinsed with acetone and dried to give 20.4 g of crude product. The crude product was stirred in cold water (ice bath) for 5 min. The solid was collected on a filter, rinsed with cold water, and dried to provide (3r,5r,7r)-1-((E)-4-(1,3-dioxoisoindolin-2-yl)-2,3-dimethylbut-2-en-1-yl)-1,3,5,7-tetraazaadamantane-1-azonia, bromide (10.3 g, 22.9 mmol, 52.6% yield) as a light yellow solid. LCMS ( m/z ): 368.2 [M] + .
步骤3: (E)-2-(4-氨基-2,3-二甲基丁-2-烯-1-基)异吲哚啉-1,3-二酮Step 3: (E)-2-(4-amino-2,3-dimethylbut-2-en-1-yl)isoindoline-1,3-dione
在室温向(3r,5r,7r)-1-((E)-4-(1,3-二氧代异吲哚啉-2-基)-2,3-二甲基丁-2-烯-1-基)-1,3,5,7-四氮杂金刚烷-1-氮鎓, 溴化物(10.3 g, 22.97 mmol)在EtOH (100mL)中的悬浮液加入浓盐酸 (7.55 mL, 92 mmol)。该反应混合物的颜色从浅黄色变成浅橙色。在80℃加热该反应55 min。颜色随时间变成更深的橙色。将该反应混合物冷却至室温并加入饱和NaHCO3溶液以提高溶液(约20 mL)的pH。搅拌该混合物5 min,用20 mL水稀释并用3:1 氯仿:EtOH (3x75 mL)萃取。经硫酸钠干燥有机萃取物,浓缩并干燥以提供作为浅棕色固体的(E)-2-(4-氨基-2,3-二甲基丁-2-烯-1-基)异吲哚啉-1,3-二酮 (5.77 g, 22.4mmol, 98%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 7.71 - 8.09 (m, 6 H), 4.25 (s,2 H), 3.45 (s, 2 H), 1.95 (d, J=1.27 Hz, 3 H), 1.65 (d, J=1.27 Hz, 3 H)。LCMS(m/z): 245.2 [M + H]+。To a suspension of (3r,5r,7r)-1-((E)-4-(1,3-dioxoisoindolin-2-yl)-2,3-dimethylbut-2-en-1-yl)-1,3,5,7-tetraazaadamantane-1-azonia, bromide (10.3 g, 22.97 mmol) in EtOH (100 mL) at room temperature was added concentrated hydrochloric acid (7.55 mL, 92 mmol). The color of the reaction mixture changed from light yellow to light orange. The reaction was heated at 80 °C for 55 min. The color changed to a darker orange over time. The reaction mixture was cooled to room temperature and saturated NaHCO 3 solution was added to increase the pH of the solution (approximately 20 mL). The mixture was stirred for 5 min, diluted with 20 mL of water and extracted with 3:1 chloroform:EtOH (3x75 mL). The organic extract was dried over sodium sulfate, concentrated and dried to provide (E)-2-(4-amino-2,3-dimethylbut-2-en-1-yl)isoindoline-1,3-dione (5.77 g, 22.4 mmol, 98% yield) as a light brown solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 7.71 - 8.09 (m, 6 H), 4.25 (s, 2 H), 3.45 (s, 2 H), 1.95 (d, J =1.27 Hz, 3 H), 1.65 (d, J =1.27 Hz, 3 H). LCMS ( m/z ): 245.2 [M + H] + .
中间体20Intermediate 20
(E)-(4-氨基-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯(E)-tert-Butyl (4-amino-2,3-dimethylbut-2-en-1-yl)carbamate
步骤1: 二氯氨基甲酸叔丁酯Step 1: tert-Butyl dichlorocarbamate
在0℃向氨基甲酸叔丁酯 (20.1 g, 172 mmol)在DCM (400 mL)中的溶液加入次氯酸钙(技术等级, 可用氯 65%) (75 g, 343 mmol),然后经35 min逐滴加入6 M盐酸(143mL, 858 mmol)(在加料期间,内部温度5-10℃)。然后,搅拌所得黄色悬浮液20 min。分离各层,用水和盐水洗涤有机层,并经硫酸钠干燥。在减压下(23℃, 80 mbar)小心地浓缩该溶液以提供作为浅黄色液体的二氯氨基甲酸叔丁酯 (33.4 g, 172 mmol, 100%)。1H NMR(400 MHz, CDCl3) δ ppm 1.56 (s, 9 H)。To a solution of tert-butyl carbamate (20.1 g, 172 mmol) in DCM (400 mL) was added calcium hypochlorite (technical grade, available chlorine 65%) (75 g, 343 mmol) at 0°C, followed by the addition of 6 M hydrochloric acid (143 mL, 858 mmol) dropwise over 35 min (internal temperature 5-10°C during addition). The resulting yellow suspension was then stirred for 20 min. The layers were separated, the organic layer was washed with water and brine, and dried over sodium sulfate. The solution was carefully concentrated under reduced pressure (23°C, 80 mbar) to provide tert-butyl dichlorocarbamate (33.4 g, 172 mmol, 100%) as a light yellow liquid. 1 H NMR (400 MHz, CDCl 3 ) δ ppm 1.56 (s, 9 H).
步骤2: (E)-(4-氯-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯Step 2: tert-Butyl (E)-(4-chloro-2,3-dimethylbut-2-en-1-yl)carbamate
将氮气鼓泡通过300 mL 氯仿持续10 min。然后,加入2,3-二甲基丁-1,3-二烯(24.79 mL, 219 mmol),并在氮气气氛下将该溶液冷却至0℃。经80 min加入二氯氨基甲酸叔丁酯 (41 g, 220 mmol)在氯仿 (150 mL)中的溶液以生成(E)-氯(4-氯-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯和(E)-(4-氯-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯的混合物。在冰浴中另外加搅拌15后,以保持内部温度低于室温的速度快速逐滴加入新鲜制备的亚硫酸钠水溶液(3M, 219 mL, 657 mmol)(警示:伴随气体逸出的放热反应)。除去冰浴并另外搅拌该反应15 min。分离各层。用水和盐水洗涤有机层,经硫酸钠干燥,并浓缩以提供作为白色固体的(E)-(4-氯-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯 (45.4 g, 184mmol, 84%收率)。1H NMR (400 MHz, 氯仿-d) δ ppm 4.49 (br. s., 1 H), 4.10 (s, 2H), 3.80 (br. s., 2 H), 1.86 (s, 3 H), 1.82 (d, J=1.25 Hz, 3 H), 1.47 (s, 9H)。Nitrogen was bubbled through 300 mL of chloroform for 10 min. Then, 2,3-dimethylbut-1,3-diene (24.79 mL, 219 mmol) was added and the solution was cooled to 0 °C under a nitrogen atmosphere. A solution of tert-butyl dichlorocarbamate (41 g, 220 mmol) in chloroform (150 mL) was added over 80 min to produce a mixture of tert-butyl (E)-chloro(4-chloro-2,3-dimethylbut-2-en-1-yl)carbamate and tert-butyl (E)-(4-chloro-2,3-dimethylbut-2-en-1-yl)carbamate. After stirring in the ice bath for an additional 15 min, a freshly prepared aqueous sodium sulfite solution (3M, 219 mL, 657 mmol) was added dropwise rapidly at a rate to keep the internal temperature below room temperature (Caution: exothermic reaction with gas evolution). The ice bath was removed and the reaction was stirred for an additional 15 min. The layers were separated. The organic layer was washed with water and brine, dried over sodium sulfate, and concentrated to provide tert-butyl (E)-(4-chloro-2,3-dimethylbut-2-en-1-yl)carbamate (45.4 g, 184 mmol, 84% yield) as a white solid. 1 H NMR (400 MHz, CHLOROFORM- d ) δ ppm 4.49 (br. s., 1 H), 4.10 (s, 2H), 3.80 (br. s., 2 H), 1.86 (s, 3 H), 1.82 (d, J =1.25 Hz, 3 H), 1.47 (s, 9H).
步骤3: (E)-(4-(1,3-二氧代异吲哚啉-2-基)-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯Step 3: tert-Butyl (E)-(4-(1,3-dioxoisoindolin-2-yl)-2,3-dimethylbut-2-en-1-yl)carbamate
向(E)-(4-氯-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯 (40.4 g, 164 mmol)在DMF (300 mL)中的溶液加入邻苯二甲酰亚胺钾(30.4 g, 164 mmol)并在室温搅拌该反应混合物3 h。将该混合物在冰/水浴中冷却并加入水(450 mL)以提供稠沉淀物。在室温搅拌10 min后,过滤固体,用水漂洗,并干燥以提供作为白色固体的(E)-(4-(1,3-二氧代异吲哚啉-2-基)-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯 (50.08 g, 137 mmol, 84%收率)。1H NMR (400 MHz, 氯仿-d) δ ppm 7.87 (dd, J=5.40, 3.14 Hz, 2 H), 7.74 (dd,J=5.27, 3.01 Hz, 2 H), 4.41 - 4.48 (m, 1 H), 4.35 (s, 2 H), 3.78 (br. s., 2H), 1.97 (s, 3 H), 1.70 (d, J=1.25 Hz, 3 H), 1.47 (s, 9 H)。To a solution of (E)-(4-chloro-2,3-dimethylbut-2-ene-1-yl)carbamic acid tert-butyl ester (40.4 g, 164 mmol) in DMF (300 mL) was added potassium phthalimide (30.4 g, 164 mmol) and the reaction mixture was stirred at room temperature for 3 h. The mixture was cooled in an ice/water bath and water (450 mL) was added to provide a thick precipitate. After stirring at room temperature for 10 min, the solid was filtered, rinsed with water, and dried to provide (E)-(4-(1,3-dioxoisoindoline-2-yl)-2,3-dimethylbut-2-ene-1-yl)carbamic acid tert-butyl ester (50.08 g, 137 mmol, 84% yield) as a white solid. 1 H NMR (400 MHz, chloroform- d ) δ ppm 7.87 (dd, J =5.40, 3.14 Hz, 2 H), 7.74 (dd, J =5.27, 3.01 Hz, 2 H), 4.41 - 4.48 (m, 1 H), 4.35 (s, 2 H), 3.78 (br. s., 2H), 1.97 (s, 3 H), 1.70 (d, J =1.25 Hz, 3 H), 1.47 (s, 9 H).
步骤4: (E)-(4-氨基-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯Step 4: tert-Butyl (E)-(4-amino-2,3-dimethylbut-2-en-1-yl)carbamate
平行地设定两个相同的反应。向(E)-(4-(1,3-二氧代异吲哚啉-2-基)-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯 (25 g, 69.0 mmol)在乙醇(400 mL)中的混合物加入肼一水合物 (6.69 mL, 138 mmol)。在80℃搅拌该混合物4.5 h。在加热30 min后,稠沉淀物开始形成并且搅拌变得困难。合并这两个反应并浓缩以除去乙醇并提供白色固体。在水(450 mL)中搅拌这种物质。加入1M HCl (50 mL)和6M HCl (14 mL)溶液以将pH调节至~5并搅拌该悬浮液10 min。过滤掉固体并用水漂洗。用DCM (100 mL)萃取水性滤液以除去任何杂质/颜色。然后用1M氢氧化钠将水相调节至pH 13并用3:1 CHCl3:EtOH (3 x 300 mL)萃取。经硫酸钠干燥合并的有机层,过滤,并浓缩以提供浅橙色油,其快速固化。用5%二乙醚/庚烷 (200 mL)研磨该固体5 min,然后过滤并用庚烷漂洗(第1批产物)。浓缩滤液并在5 mL二乙醚中搅拌。用最少的二乙醚漂洗固体并过滤以提供第二批产物。合并并在真空中干燥以提供作为灰白色固体的(E)-(4-氨基-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯 (23.9g, 111 mmol, 80%收率)。1H NMR (400 MHz, 甲醇-d 4) δ ppm 3.70 (s, 2 H), 3.24 (s,2 H), 1.81 (d, J=1.00 Hz, 3 H), 1.73 (s, 3 H), 1.46 (s, 9 H)。LCMS (m/z):215.3 [M + H]+。Two identical reactions were set up in parallel. To a mixture of (E)-(4-(1,3-dioxoisoindolin-2-yl)-2,3-dimethylbut-2-ene-1-yl)carbamic acid tert-butyl ester (25 g, 69.0 mmol) in ethanol (400 mL) was added hydrazine monohydrate (6.69 mL, 138 mmol). The mixture was stirred at 80 ° C for 4.5 h. After heating for 30 min, a thick precipitate began to form and stirring became difficult. The two reactions were combined and concentrated to remove ethanol and provide a white solid. This material was stirred in water (450 mL). 1M HCl (50 mL) and 6M HCl (14 mL) solutions were added to adjust the pH to ~5 and the suspension was stirred for 10 min. The solid was filtered off and rinsed with water. The aqueous filtrate was extracted with DCM (100 mL) to remove any impurities/color. The 1-hydroxy-2-(4-amino-2,3-dimethylbut-2-ene-1-yl) t-butyl carbamate (23.9g, 111 mmol, 80% yield) of (E)-(4-amino-2,3-dimethylbut-2-ene-1-yl) t-butyl carbamate (23.9g, 111 mmol, 80% yield) was added to the 4-hydroxy-2-(4-amino-2,3-dimethylbut-2-ene-1-yl) carbamate (23.9g, 111 mmol, 80% yield) solution in 5 mL diethyl ether.Then the aqueous phase was adjusted to pH 13 with 1M sodium hydroxide and extracted with 3:1 CHCl 3:EtOH (3 x 300 mL).The organic layer merged through sodium sulfate drying, filtered, and concentrated to provide a light orange oil, which quickly solidifies.Grind this solid 5 min with 5% diethyl ether/heptane (200 mL), then filter and rinse with heptane (the 1st batch product).Concentrate the filtrate and stir in 5 mL diethyl ether.Rinse solid with minimum diethyl ether and filter to provide second batch product.Merge and dry in vacuum to provide (E)-(4-amino-2,3-dimethylbut-2-ene-1-yl) t-butyl carbamate (23.9g, 111 mmol, 80% yield) as off-white solid. 1 H NMR (400 MHz, methanol- d 4 ) δ ppm 3.70 (s, 2 H), 3.24 (s, 2 H), 1.81 (d, J =1.00 Hz, 3 H), 1.73 (s, 3 H), 1.46 (s, 9 H). LCMS ( m/z ): 215.3 [M + H] + .
中间体21Intermediate 21
(E)-4-((4-氨基-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺, 盐酸盐(E)-4-((4-Amino-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide, hydrochloride
步骤1: (E)-(4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯Step 1: tert-Butyl (E)-(4-((4-carbamoyl-2-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)carbamate
向(E)-(4-氨基-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯 (1.92 g, 8.96mmol)和4-氟-3-硝基苯甲酰胺 (1.650 g, 8.96 mmol)在DMSO (25 mL)中的溶液加入碳酸钾(1.486 g, 10.75 mmol)。在室温搅拌亮橙色混合物2 h。将该混合物逐滴加入快速搅拌的冰水(200 mL)中并搅拌1 h。过滤所得沉淀物,用水漂洗,并干燥以提供作为亮黄色固体的(E)-(4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯 (2.9 g, 7.5 mmol, 84%收率)。 1H NMR (400 MHz, DMSO-d 6) δ ppm 8.66 (d, J=2.28Hz, 1 H), 8.36 (t, J=5.32 Hz, 1 H), 7.99 (dd, J=8.87, 2.03 Hz, 2 H), 7.31(br. s., 1 H), 7.02 (t, J=5.70 Hz, 1 H), 6.92 (d, J=9.12 Hz, 1 H), 4.02 (d, J=5.07 Hz, 2 H), 3.60 (d, J=5.58 Hz, 2 H), 1.75 (s, 3 H), 1.68 (s, 3 H), 1.38(s, 9 H)。To a solution of (E)-(4-amino-2,3-dimethylbut-2-ene-1-yl)carbamic acid tert-butyl ester (1.92 g, 8.96 mmol) and 4-fluoro-3-nitrobenzamide (1.650 g, 8.96 mmol) in DMSO (25 mL) was added potassium carbonate (1.486 g, 10.75 mmol). The bright orange mixture was stirred at room temperature for 2 h. The mixture was added dropwise to rapidly stirred ice water (200 mL) and stirred for 1 h. The resulting precipitate was filtered, rinsed with water, and dried to provide (E)-(4-((4-carbamoyl-2-nitrophenyl)amino)-2,3-dimethylbut-2-ene-1-yl)carbamic acid tert-butyl ester (2.9 g, 7.5 mmol, 84% yield) as a bright yellow solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.66 (d, J =2.28Hz, 1 H), 8.36 (t, J =5.32 Hz, 1 H), 7.99 (dd, J =8.87, 2.03 Hz, 2 H), 7.31 (br. s., 1 H), 7.02 (t, J =5 .70 Hz, 1 H), 6.92 (d, J =9.12 Hz, 1 H), 4.02 (d, J =5.07 Hz, 2 H), 3.60 (d, J =5.58 Hz, 2 H), 1.75 (s, 3 H), 1.68 (s, 3 H), 1.38 (s, 9 H).
步骤2: (E)-4-((4-氨基-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺, 盐酸盐Step 2: (E)-4-((4-amino-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide, hydrochloride
向(E)-(4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯 (2.9 g, 7.66 mmol)在DCM (LARA更新)中的悬浮液加入 在二氧杂环己烷中的4MHCl (9.58 mL, 38.3 mmol)。在室温搅拌该反应2 h。过滤所得固体,用DCM漂洗,并干燥以提供作为亮黄色固体的(E)-4-((4-氨基-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺, 盐酸盐 (2.4 g, 7.28 mmol, 95%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm8.68 (d, J=2.28 Hz, 1 H), 8.44 (t, J=5.45 Hz, 1 H), 8.01 (dd, J=9.00, 2.15Hz, 5 H), 7.33 (br. s., 1 H), 6.90 (d, J=9.12 Hz, 1 H), 4.10 (d, J=5.07 Hz, 2H), 3.48 (d, J=5.58 Hz, 2 H), 1.90 (s, 3 H), 1.72 (s, 3 H)。LCMS (m/z): 无显著的[M + H]+。To a suspension of tert-butyl (E)-(4-((4-carbamoyl-2-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)carbamate (2.9 g, 7.66 mmol) in DCM (LARA updated) was added 4M HCl in dioxane (9.58 mL, 38.3 mmol). The reaction was stirred at room temperature for 2 h. The resulting solid was filtered, rinsed with DCM, and dried to afford (E)-4-((4-amino-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide, hydrochloride (2.4 g, 7.28 mmol, 95% yield) as a bright yellow solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm8.68 (d, J =2.28 Hz, 1 H), 8.44 (t, J =5.45 Hz, 1 H), 8.01 (dd, J =9.00, 2.15Hz, 5 H), 7.33 (br. s., 1 H), 6.90 (d, J =9. 12 Hz, 1 H), 4.10 (d, J =5.07 Hz, 2H), 3.48 (d, J =5.58 Hz, 2 H), 1.90 (s, 3 H), 1.72 (s, 3 H). LCMS ( m/z ): No significant [M + H] + .
中间体22Intermediate 22
(2S,3S)-2,3-二乙氧基丁烷-1,4-二胺(2S,3S)-2,3-diethoxybutane-1,4-diamine
步骤1: (4S,5S)-4,5-双(叠氮基甲基)-2,2-二甲基-1,3-二氧杂环戊烷Step 1: (4S,5S)-4,5-bis(azidomethyl)-2,2-dimethyl-1,3-dioxolane
向((4S,5S)-2,2-二甲基-1,3-二氧杂环戊烷-4,5-二基)双(亚甲基)双(4-甲基苯磺酸酯) (5.23 g, 11.11 mmol)在DMF (20 mL)中的溶液加入叠氮化钠 (2.89 g, 44.5mmol)。在80℃搅拌该混合物18 h。用水(100 mL)稀释该混合物并用EtOAc (2 x 100mL)萃取。用水(2 x 100 mL)、盐水(100 mL)洗涤有机相,用硫酸镁干燥并浓缩以提供作为透明油的(4S,5S)-4,5-双(叠氮基甲基)-2,2-二甲基-1,3-二氧杂环戊烷 (2.3 g, 10.8 mmol,98%收率)。1H NMR (400 MHz, 氯仿-d) δ ppm 1.51 (s, 6 H), 3.30 - 3.43 (m, 2 H),3.54 - 3.66 (m, 2 H), 4.10 (td, J=2.8, 1.3 Hz, 2 H)。LCMS (m/z): 无显著的[M +H]+。To a solution of ((4S,5S)-2,2-dimethyl-1,3-dioxolane-4,5-diyl)bis(methylene)bis(4-methylbenzenesulfonate) (5.23 g, 11.11 mmol) in DMF (20 mL) was added sodium azide (2.89 g, 44.5 mmol). The mixture was stirred at 80 °C for 18 h. The mixture was diluted with water (100 mL) and extracted with EtOAc (2 x 100 mL). The organic phase was washed with water (2 x 100 mL), brine (100 mL), dried over magnesium sulfate and concentrated to provide (4S,5S)-4,5-bis(azidomethyl)-2,2-dimethyl-1,3-dioxolane (2.3 g, 10.8 mmol, 98% yield) as a clear oil. 1 H NMR (400 MHz, chloroform- d ) δ ppm 1.51 (s, 6 H), 3.30 - 3.43 (m, 2 H), 3.54 - 3.66 (m, 2 H), 4.10 (td, J =2.8, 1.3 Hz, 2 H). LCMS ( m/z ): No significant [M +H] + .
步骤2: (2S,3S)-1,4-二叠氮基丁烷-2,3-二醇Step 2: (2S,3S)-1,4-diazidebutane-2,3-diol
向(4S,5S)-4,5-双(叠氮基甲基)-2,2-二甲基-1,3-二氧杂环戊烷 (2.3 g,10.84 mmol)在THF (50 mL)中的溶液加入对甲苯磺酸 (0.103 g, 0.542 mmol)。在60℃加热该反应混合物18 h。将该反应混合物冷却至室温并在EtOAc (50 mL)和水(30 mL)之间分配。用EtOAc (2 x 50 mL)萃取水相。用盐水(30 mL)洗涤合并的有机相,经硫酸镁干燥,并浓缩。NMR分析表明没有反应发生。向混合物加入在甲醇中的1.25 M HCl (34.7 mL, 43.4mmol)。在60℃加热该反应18 h。将反应混合物浓缩以提供作为浅黄色透明油的(2S,3S)-1,4-二叠氮基丁烷-2,3-二醇(2.01 g, 10.5 mmol, 97%收率)。1H NMR (400 MHz, 氯仿-d)δ ppm 3.35 - 3.59 (m, 4 H), 3.71 - 3.90 (m, 2 H)。To a solution of (4S,5S)-4,5-bis(azidomethyl)-2,2-dimethyl-1,3-dioxolane (2.3 g, 10.84 mmol) in THF (50 mL) was added p-toluenesulfonic acid (0.103 g, 0.542 mmol). The reaction mixture was heated at 60 °C for 18 h. The reaction mixture was cooled to room temperature and partitioned between EtOAc (50 mL) and water (30 mL). The aqueous phase was extracted with EtOAc (2 x 50 mL). The combined organic phases were washed with brine (30 mL), dried over magnesium sulfate, and concentrated. NMR analysis indicated that no reaction occurred. 1.25 M HCl in methanol (34.7 mL, 43.4 mmol) was added to the mixture. The reaction was heated at 60 °C for 18 h. The reaction mixture was concentrated to afford (2S,3S)-1,4-diazidobutane-2,3-diol (2.01 g, 10.5 mmol, 97% yield) as a light yellow clear oil. 1 H NMR (400 MHz, CHLOROFORM- d ) δ ppm 3.35 - 3.59 (m, 4 H), 3.71 - 3.90 (m, 2 H).
步骤3: (2S,3S)-2,3-二乙氧基丁烷-1,4-二胺Step 3: (2S,3S)-2,3-diethoxybutane-1,4-diamine
在0℃向(2S,3S)-1,4-二叠氮基丁烷-2,3-二醇(2.01 g, 11.68 mmol)在DMF (50mL)中的混合物加入氢化钠 (1.167 g, 29.2 mmol)。在室温搅拌该混合物5 min,然后加入碘乙烷(2.36 mL, 29.2 mmol)。在室温搅拌该混合物18 h。在EtOAc (100 mL)和水(100mL)之间分配该混合物。用盐水(3 x 30 mL)洗涤有机相,用硫酸镁干燥并浓缩以提供作为透明油的粗品(2S,3S)-1,4-二叠氮基-2,3-二乙氧基丁烷(2.47 g)。1H NMR (400 MHz, 氯仿-d) δ ppm 1.27 (t, J=7.0 Hz, 6 H), 3.31 - 3.47 (m, 4 H), 3.56 - 3.82 (m, 6H)。用氮气吹扫粗品(2S,3S)-1,4-二叠氮基-2,3-二乙氧基丁烷(2.47 g)和碳载钯(0.3 g,2.8 mmol)在甲醇 (30 mL)中的混合物并交换为氢气气氛(气球)。在室温搅拌该混合物18h。氢气与氮气交换,将该混合物过滤通过硅藻土并浓缩以提供作为透明油的(2S,3S)-2,3-二乙氧基丁烷-1,4-二胺 (1.86 g, 90%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 1.07 -1.13 (m, 6 H), 2.31 - 2.49 (m, 2 H), 2.57 - 2.68 (m, 2 H), 3.20 - 3.27 (m, 2H), 3.50 - 3.60 (m, 4 H)。To a mixture of (2S,3S)-1,4-diazide butane-2,3-diol (2.01 g, 11.68 mmol) in DMF (50 mL) was added sodium hydride (1.167 g, 29.2 mmol) at 0 ° C. The mixture was stirred at room temperature for 5 min, and then iodoethane (2.36 mL, 29.2 mmol) was added. The mixture was stirred at room temperature for 18 h. The mixture was distributed between EtOAc (100 mL) and water (100 mL). The organic phase was washed with brine (3 x 30 mL), dried over magnesium sulfate and concentrated to provide a crude product (2S,3S)-1,4-diazide-2,3-diethoxybutane (2.47 g) as a clear oil. 1H NMR (400 MHz, chloroform- d ) δ ppm 1.27 (t, J =7.0 Hz, 6 H), 3.31 - 3.47 (m, 4 H), 3.56 - 3.82 (m, 6H). A mixture of crude (2S, 3S)-1,4-diazide-2,3-diethoxybutane (2.47 g) and palladium on carbon (0.3 g, 2.8 mmol) in methanol (30 mL) was purged with nitrogen and exchanged with a hydrogen atmosphere (balloon). The mixture was stirred at room temperature for 18 h. Hydrogen was exchanged with nitrogen, the mixture was filtered through diatomaceous earth and concentrated to provide (2S, 3S)-2,3-diethoxybutane-1,4-diamine (1.86 g, 90% yield) as a clear oil. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 1.07 -1.13 (m, 6 H), 2.31 - 2.49 (m, 2 H), 2.57 - 2.68 (m, 2 H), 3.20 - 3.27 (m, 2H), 3.50 - 3.60 (m, 4 H).
中间体23Intermediate 23
4-氯-3-硝基-5-(三氟甲基)苯甲酰胺4-Chloro-3-nitro-5-(trifluoromethyl)benzamide
在室温向4-氯-3-硝基-5-(三氟甲基)苯甲酸 (3.94 g, 14.62 mmol)在DCM (97mL)中的溶液加入草酰氯 (2.047 mL, 23.39 mmol)和4滴DMF。在搅拌1 h后,加入30% 氢氧化铵溶液(9.49 mL, 73.1 mmol)并搅拌18 h。过滤所得白色沉淀物,首先用水,然后用DCM洗涤并干燥以提供作为白色固体的4-氯-3-硝基-5-(三氟甲基)苯甲酰胺 (3.32 g, 12.36mmol, 85%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.77 (d, J=2.0 Hz, 1 H), 8.57(d, J=1.8 Hz, 1 H), 7.97 (br. s., 2 H)。LCMS (m/z): 269.1 [M + H]+。To a solution of 4-chloro-3-nitro-5-(trifluoromethyl)benzoic acid (3.94 g, 14.62 mmol) in DCM (97 mL) was added oxalyl chloride (2.047 mL, 23.39 mmol) and 4 drops of DMF at room temperature. After stirring for 1 h, 30% ammonium hydroxide solution (9.49 mL, 73.1 mmol) was added and stirred for 18 h. The resulting white precipitate was filtered, first washed with water, then with DCM and dried to provide 4-chloro-3-nitro-5-(trifluoromethyl)benzamide (3.32 g, 12.36 mmol, 85% yield) as a white solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.77 (d, J =2.0 Hz, 1 H), 8.57 (d, J =1.8 Hz, 1 H), 7.97 (br. s., 2 H). LCMS ( m/z ): 269.1 [M + H] + .
中间体24Intermediate 24
3-(5-氨基甲酰基-2-氟-3-硝基苯基)丙酸乙酯Ethyl 3-(5-carbamoyl-2-fluoro-3-nitrophenyl)propionate
向在DMF (60.8 mL)中的3-溴-4-氟-5-硝基苯甲酰胺 (5 g, 18.25 mmol)加入四正丁基氯化铵 (5.18 g, 18.25 mmol)和Pd(OAc)2 (0.418 g, 1.825 mmol)。将氮气鼓泡通过该混合物持续2 min,然后加入3,3-二乙氧基丙-1-烯(8.69 mL, 54.7 mmol)和三丁胺(8.83 mL, 36.5 mmol)。密封容器并在125℃加热该混合物16 h。在乙酸乙酯 (200 mL)和饱和氯化铵水溶液(200 mL)之间分配该混合物。用乙酸乙酯再次萃取水相。用盐水(2x150mL)洗涤合并的有机层,用硫酸镁干燥,过滤,并在真空中浓缩。通过硅胶层析法 (120 g硅胶, 30-100%梯度的EtOAc/己烷)纯化残余物以提供3-(5-氨基甲酰基-2-氟-3-硝基苯基)丙酸乙酯(1.77 g, 5.42 mmol, 29.7%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.48(dd, J=6.7, 2.2 Hz, 1 H), 8.18 - 8.33 (m, 2 H), 7.71 (br. s., 1 H), 4.05 (q,J=7.3 Hz, 2 H), 2.94 - 3.06 (m, 2 H), 2.73 (t, J=7.5 Hz, 2 H), 1.15 (t, J=7.3Hz, 3 H)。LCMS (m/z): 285.1 [M + H]+。To 3-bromo-4-fluoro-5-nitrobenzamide (5 g, 18.25 mmol) in DMF (60.8 mL) was added tetra-n-butylammonium chloride (5.18 g, 18.25 mmol) and Pd(OAc) 2 (0.418 g, 1.825 mmol). Nitrogen was bubbled through the mixture for 2 min, then 3,3-diethoxyprop-1-ene (8.69 mL, 54.7 mmol) and tributylamine (8.83 mL, 36.5 mmol) were added. The vessel was sealed and the mixture was heated at 125 °C for 16 h. The mixture was distributed between ethyl acetate (200 mL) and saturated aqueous ammonium chloride solution (200 mL). The aqueous phase was extracted again with ethyl acetate. The combined organic layers were washed with brine (2x150 mL), dried over magnesium sulfate, filtered, and concentrated in vacuo. The residue was purified by silica gel chromatography (120 g silica gel, 30-100% gradient of EtOAc/hexanes) to provide ethyl 3-(5-carbamoyl-2-fluoro-3-nitrophenyl)propanoate (1.77 g, 5.42 mmol, 29.7% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.48 (dd, J =6.7, 2.2 Hz, 1 H), 8.18 - 8.33 (m, 2 H), 7.71 (br. s., 1 H), 4.05 (q, J =7.3 Hz, 2 H), 2.94 - 3.06 (m, 2 H), 2.73 (t, J =7.5 Hz, 2 H), 1.15 (t, J =7.3 Hz, 3 H). LCMS ( m/z ): 285.1 [M + H] + .
中间体25Intermediate 25
2-氟-1-((4-甲氧基苄基)氧基)-3-硝基苯2-Fluoro-1-((4-methoxybenzyl)oxy)-3-nitrobenzene
向2-氟-3-硝基苯酚 (4.75 g, 30.2 mmol)在DMF (40 mL)中的棕色溶液加入碳酸铯 (10.84 g, 33.3 mmol)和4-甲氧基苄基氯 (4.32 mL, 31.7 mmol)。在室温搅拌该混合物16 h。将水(150 mL)加入剧烈搅拌的反应混合物并搅拌10 min以产生沉淀物。过滤固体,用水漂洗,并干燥以提供2-氟-1-((4-甲氧基苄基)氧基)-3-硝基苯 (8.1 g, 28.1mmol, 93%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 7.63 - 7.75 (m, 2 H), 7.42 (d,J=8.62 Hz, 2 H), 7.36 (d, J=1.77 Hz, 1 H), 6.92 - 7.02 (m, 2 H), 5.21 (s, 2H), 3.77 (s, 3 H)。LCMS (m/z):未观察到[M + H]+。To the brown solution of 2-fluoro-3-nitrophenol (4.75 g, 30.2 mmol) in DMF (40 mL), cesium carbonate (10.84 g, 33.3 mmol) and 4-methoxybenzyl chloride (4.32 mL, 31.7 mmol) were added. The mixture was stirred at room temperature for 16 h. Water (150 mL) was added to the vigorously stirred reaction mixture and stirred for 10 min to produce a precipitate. The solid was filtered, rinsed with water, and dried to provide 2-fluoro-1-((4-methoxybenzyl)oxy)-3-nitrobenzene (8.1 g, 28.1 mmol, 93% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 7.63 - 7.75 (m, 2 H), 7.42 (d, J =8.62 Hz, 2 H), 7.36 (d, J =1.77 Hz, 1 H), 6.92 - 7.02 (m, 2 H), 5.21 (s, 2H), 3.77 (s, 3 H). LCMS ( m / z ): [M + H] + not observed.
中间体26Intermediate 26
(2R,3S)-2,3-二甲氧基丁烷-1,4-二胺(2R,3S)-2,3-Dimethoxybutane-1,4-diamine
步骤1: (2R,3S)-2,3-二甲氧基琥珀酸二甲酯Step 1: (2R,3S)-2,3-dimethoxysuccinic acid dimethyl ester
向(2R,3S)-2,3-二羟基琥珀酸二甲酯 (5.86 g, 32.9 mmol)和氧化银(22.87 g,99 mmol)的混合物加入碘甲烷(41.1 mL, 658 mmol)。在45℃加热该混合物6 h并在室温保持18 h。过滤该混合物,用DCM洗涤并浓缩以提供作为透明油的标题化合物(5.8 g, 28.3mmol, 86%收率),其在存放后固化。1H NMR (400 MHz, 甲醇-d 4 ) δ ppm 4.26 (s, 2 H)3.76 (s, 6 H) 3.46 (s, 6 H)。To a mixture of (2R,3S)-2,3-dihydroxysuccinic acid dimethyl ester (5.86 g, 32.9 mmol) and silver oxide (22.87 g, 99 mmol) was added iodomethane (41.1 mL, 658 mmol). The mixture was heated at 45 ° C for 6 h and kept at room temperature for 18 h. The mixture was filtered, washed with DCM and concentrated to provide the title compound as a clear oil (5.8 g, 28.3 mmol, 86% yield), which solidified after storage. 1 H NMR (400 MHz, methanol- d 4 ) δ ppm 4.26 (s, 2 H) 3.76 (s, 6 H) 3.46 (s, 6 H).
步骤2: (2R,3S)-2,3-二甲氧基丁烷-1,4-二醇Step 2: (2R,3S)-2,3-dimethoxybutane-1,4-diol
在0℃将(2R,3S)-2,3-二甲氧基琥珀酸二甲酯 (5.1 g, 24.73 mmol)在THF (30mL)中的溶液加入LAH (2.065 g, 54.4 mmol)在THF (150 mL)中的混合物。将该混合物温热至室温保持2 h。用饱和硫酸钠溶液(9.1 mL)淬灭反应。过滤混合物,用硫酸镁干燥并浓缩以提供作为透明油的标题化合物(3.6 g, 24.0 mmol, 97%收率)。1H NMR (400 MHz,DMSO-d 6 ) δ ppm 4.51 (t, J=5.58 Hz, 2 H) 3.51 - 3.59 (m, 2 H) 3.38 - 3.45 (m,2 H) 3.32 (s, 6 H) 3.19 - 3.26 (m, 2 H)。A solution of (2R,3S)-2,3-dimethoxysuccinic acid dimethyl ester (5.1 g, 24.73 mmol) in THF (30 mL) was added to a mixture of LAH (2.065 g, 54.4 mmol) in THF (150 mL) at 0 ° C. The mixture was warmed to room temperature for 2 h. The reaction was quenched with saturated sodium sulfate solution (9.1 mL). The mixture was filtered, dried over magnesium sulfate and concentrated to provide the title compound (3.6 g, 24.0 mmol, 97% yield) as a clear oil. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 4.51 (t, J =5.58 Hz, 2 H) 3.51 - 3.59 (m, 2 H) 3.38 - 3.45 (m, 2 H) 3.32 (s, 6 H) 3.19 - 3.26 (m, 2 H).
步骤3: 双(4-甲基苯磺酸)(2R,3S)-2,3-二甲氧基丁烷-1,4-二基酯Step 3: (2R,3S)-2,3-dimethoxybutane-1,4-diyl bis(4-methylbenzenesulfonate)
在-78℃向(2R,3S)-2,3-二甲氧基丁烷-1,4-二醇 (3.3 g, 21.97 mmol)在吡啶(40 mL)中的溶液加入TsCl (12.57 g, 65.9 mmol)。使该混合物温热至室温并搅拌18 h。加入水(150 mL)并将混合物冷却至0℃保持2 h。过滤所得沉淀物,用水漂洗并干燥以提供作为白色固体的标题化合物(7.52 g, 16.4 mmol, 74.6%收率)。1H NMR (400 MHz, 氯仿-d) δ ppm 7.69 - 7.86 (m, 4 H) 7.35 - 7.44 (m, 4 H) 4.08 - 4.33 (m, 4 H) 3.33- 3.45 (m, 2 H) 3.25 (s, 6 H) 2.49 (s, 6 H)。To a solution of (2R,3S)-2,3-dimethoxybutane-1,4-diol (3.3 g, 21.97 mmol) in pyridine (40 mL) was added TsCl (12.57 g, 65.9 mmol) at -78 °C. The mixture was allowed to warm to room temperature and stirred for 18 h. Water (150 mL) was added and the mixture was cooled to 0 °C for 2 h. The resulting precipitate was filtered, rinsed with water and dried to provide the title compound (7.52 g, 16.4 mmol, 74.6% yield) as a white solid. 1 H NMR (400 MHz, chloroform- d ) δ ppm 7.69 - 7.86 (m, 4 H) 7.35 - 7.44 (m, 4 H) 4.08 - 4.33 (m, 4 H) 3.33- 3.45 (m, 2 H) 3.25 (s, 6 H) 2.49 (s, 6 H).
步骤4: (2R,3S)-1,4-二叠氮基-2,3-二甲氧基丁烷Step 4: (2R,3S)-1,4-diazide-2,3-dimethoxybutane
向双(4-甲基苯磺酸)(2R,3S)-2,3-二甲氧基丁烷-1,4-二基酯 (7.52 g, 16.40mmol)在DMF (40 mL)中的溶液加入叠氮化钠 (4.26 g, 65.6 mmol)。在80℃搅拌该混合物18 h。用水(200 mL)稀释该混合物并用EtOAc (2 x 200 mL)萃取。用水(2 x 200 mL)和盐水(100 mL)洗涤有机相,用硫酸镁干燥并浓缩以提供作为透明油的标题化合物(3.16 g,15.8 mmol, 96%收率)。1H NMR (400 MHz, 氯仿-d) δ ppm 3.60 - 3.68 (m, 2 H) 3.48(s, 6 H) 3.37 - 3.45 (m, 4 H)。To a solution of bis(4-methylbenzenesulfonic acid) (2R,3S)-2,3-dimethoxybutane-1,4-diyl ester (7.52 g, 16.40 mmol) in DMF (40 mL) was added sodium azide (4.26 g, 65.6 mmol). The mixture was stirred at 80 °C for 18 h. The mixture was diluted with water (200 mL) and extracted with EtOAc (2 x 200 mL). The organic phase was washed with water (2 x 200 mL) and brine (100 mL), dried over magnesium sulfate and concentrated to provide the title compound (3.16 g, 15.8 mmol, 96% yield) as a clear oil. 1 H NMR (400 MHz, chloroform- d ) δ ppm 3.60 - 3.68 (m, 2 H) 3.48 (s, 6 H) 3.37 - 3.45 (m, 4 H).
步骤5: (2R,3S)-2,3-二甲氧基丁烷-1,4-二胺Step 5: (2R,3S)-2,3-dimethoxybutane-1,4-diamine
向(2R,3S)-1,4-二叠氮基-2,3-二甲氧基丁烷(3.16 g, 15.8 mmol)和碳载钯(0.672 g, 6.31 mmol)在甲醇 (30 mL)中的混合物加入氢气(气球)。在室温搅拌该混合物60 h。在除去氢气之后,将该混合物过滤通过硅藻土并浓缩以提供作为透明油的标题化合物(2.33 g, 15.7 mmol, 100%收率)。1H NMR (400 MHz, 氯仿-d) δ ppm 3.49 (s, 6 H)3.25 - 3.35 (m, 2 H) 2.83 - 2.96 (m, 4 H)。To a mixture of (2R, 3S)-1,4-diazide-2,3-dimethoxybutane (3.16 g, 15.8 mmol) and palladium on carbon (0.672 g, 6.31 mmol) in methanol (30 mL) was added hydrogen (balloon). The mixture was stirred at room temperature for 60 h. After removing the hydrogen, the mixture was filtered through diatomaceous earth and concentrated to provide the title compound (2.33 g, 15.7 mmol, 100% yield) as a clear oil. 1 H NMR (400 MHz, chloroform- d ) δ ppm 3.49 (s, 6 H) 3.25 - 3.35 (m, 2 H) 2.83 - 2.96 (m, 4 H).
中间体27Intermediate 27
((1S,2S)-环丙烷-1,2-二基)二甲胺, 2盐酸盐((1S,2S)-Cyclopropane-1,2-diyl)dimethylamine, 2-hydrochloride
步骤1: (1S,2S)-环丙烷-1,2-二甲酰胺Step 1: (1S,2S)-cyclopropane-1,2-dicarboxamide
向250-mL圆底烧瓶中加入(1S,2S)-环丙烷-1,2-二甲酸二乙酯(38 g, 204 mmol)和氢氧化铵溶液(28% wt水溶液; 380 mL, 3035 mmol)。在25℃搅拌该混合物 48 h。过滤该混合物,并随后用EtOAc (100 mL)洗涤滤饼。在真空下干燥固体以获得作为白色固体的(1S,2S)-环丙烷-1,2-二甲酰胺 (14.5 g, 108 mmol, 53%收率)。 1H NMR (300 MHz,DMSO-d 6 ) δ ppm 7.65 (s, 2 H), 6.90 (s, 2 H), 1.86 (m, 2 H), 0.97 (m, 2 H)。To a 250-mL round-bottom flask was added (1S,2S)-cyclopropane-1,2-dicarboxylic acid diethyl ester (38 g, 204 mmol) and ammonium hydroxide solution (28% wt aqueous solution; 380 mL, 3035 mmol). The mixture was stirred at 25 °C for 48 h. The mixture was filtered and the filter cake was subsequently washed with EtOAc (100 mL). The solid was dried under vacuum to obtain (1S,2S)-cyclopropane-1,2-dicarboxamide (14.5 g, 108 mmol, 53% yield) as a white solid. 1 H NMR (300 MHz, DMSO- d 6 ) δ ppm 7.65 (s, 2 H), 6.90 (s, 2 H), 1.86 (m, 2 H), 0.97 (m, 2 H).
步骤2: (((1S,2S)-环丙烷-1,2-二基)双(亚甲基))二氨基甲酸二叔丁酯Step 2: Di-tert-butyl (((1S,2S)-cyclopropane-1,2-diyl)bis(methylene))dicarbamate
在0℃向(1S,2S)-环丙烷-1,2-二甲酰胺 (14.5 g, 113 mmol)和THF (300 mL)的混合物分批加入LiAlH4 (17.18 g, 453 mmol)。然后,在25℃搅拌该混合物48 h。在0℃通过加入碎冰(200 g)淬灭该混合物。过滤该混合物,并将滤液直接用于接下来的步骤。向滤液加入LiOH (10.52 g, 4.39 mmol)和水(200 mL)。加入Boc酐(56.1 mL, 242 mmol)并在室温搅拌混合物过夜。然后,用DCM (100 mL x 3)萃取反应混合物三次。用硫酸钠干燥合并的有机层,过滤,并在减压下浓缩。通过正相硅胶层析法 (80 g硅胶, 1:4 EtOAc/石油醚)纯化残余物以提供作为无色油的(((1S,2S)-环丙烷-1,2-二基)双(亚甲基))二氨基甲酸二叔丁酯 (10 g, 31.6 mmol, 经两步29%收率)。1H NMR (300 MHz, 甲醇-d 4 ,) δ ppm 3.03(m, 2 H), 2.85 (m, 2 H), 0.83 (m, 2 H), 0.41 (m, 2 H)。To a mixture of (1S,2S)-cyclopropane-1,2-dicarboxamide (14.5 g, 113 mmol) and THF (300 mL) was added LiAlH 4 (17.18 g, 453 mmol) in portions at 0°C. Then, the mixture was stirred at 25°C for 48 h. The mixture was quenched by adding crushed ice (200 g) at 0°C. The mixture was filtered and the filtrate was used directly in the next step. LiOH (10.52 g, 4.39 mmol) and water (200 mL) were added to the filtrate. Boc anhydride (56.1 mL, 242 mmol) was added and the mixture was stirred at room temperature overnight. Then, the reaction mixture was extracted three times with DCM (100 mL x 3). The combined organic layers were dried over sodium sulfate, filtered, and concentrated under reduced pressure. The residue was purified by normal phase silica gel chromatography (80 g silica gel, 1:4 EtOAc/petroleum ether) to provide di-tert-butyl (((1S,2S)-cyclopropane-1,2-diyl)bis(methylene))dicarbamate (10 g, 31.6 mmol, 29% yield over two steps) as a colorless oil. 1 H NMR (300 MHz, methanol- d 4 ,) δ ppm 3.03 (m, 2 H), 2.85 (m, 2 H), 0.83 (m, 2 H), 0.41 (m, 2 H).
步骤3: ((1S,2S)-环丙烷-1,2-二基)二甲胺, 2盐酸盐Step 3: ((1S,2S)-cyclopropane-1,2-diyl)dimethylamine, 2-hydrochloride
向500-mL圆底烧瓶中加入(((1S,2S)-环丙烷-1,2-二基)双(亚甲基))二氨基甲酸二叔丁酯 (10 g, 33.3 mmol)和HCl (4 M在1,4-二氧杂环己烷中, 100 mL, 400 mmol)。在25℃搅拌30 min后,在减压下浓缩该混合物。然后,将残余物溶解于水(100 mL)中并冻干。获得作为灰白色固体的((1S,2S)-环丙烷-1,2-二基)二甲胺, 2盐酸盐 (5.3 g, 29.1mmol, 87%收率)。1H NMR (400 MHz, 甲醇-d 4 ,) δ ppm 3.05 (m, 2 H), 2.81 (m, 2 H),1.27 - 1.18 (m, 2 H), 0.87 - 0.79 (m, 2 H)。LCMS (m/z): 101.2 [M + H]+, 未观察到UV峰。To a 500-mL round-bottom flask was added di-tert-butyl (((1S,2S)-cyclopropane-1,2-diyl)bis(methylene))dicarbamate (10 g, 33.3 mmol) and HCl (4 M in 1,4-dioxane, 100 mL, 400 mmol). After stirring at 25 °C for 30 min, the mixture was concentrated under reduced pressure. The residue was then dissolved in water (100 mL) and lyophilized. ((1S,2S)-cyclopropane-1,2-diyl)dimethylamine, 2-hydrochloride (5.3 g, 29.1 mmol, 87% yield) was obtained as an off-white solid. 1 H NMR (400 MHz, methanol- d 4 ,) δ ppm 3.05 (m, 2 H), 2.81 (m, 2 H), 1.27 - 1.18 (m, 2 H), 0.87 - 0.79 (m, 2 H). LCMS ( m / z ): 101.2 [M + H] + , UV peak not observed.
实施例1Example 1
(E)-1-((E)-4-((Z)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-((E)-4-((Z)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
步骤1: (E)-1-(4-((4-氨基甲酰基-2-(3-吗啉代丙氧基)-6-硝基苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 1: (E)-1-(4-((4-Carbamoyl-2-(3-morpholinopropoxy)-6-nitrophenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide
向(E)-1-(4-氨基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺盐酸盐 (535 mg, 1.280 mmol)在EtOH (5 mL)中的悬浮液加入三乙胺 (471 mg, 4.65 mmol)和4-氯-3-(3-吗啉代丙氧基)-5-硝基苯甲酰胺 (400 mg,1.164 mmol)。密封该反应容器并在120℃加热20 h。在冷却之后,橙色固体从深色溶液中沉淀出来。用EtOAc洗涤该固体并干燥以提供(E)-1-(4-((4-氨基甲酰基-2-(3-吗啉代丙氧基)-6-硝基苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (457 mg, 0.664 mmol, 57.0%收率)。重复三次该反应以提供1.37 g标题化合物。LCMS m/z = 689 [M+H]+。To a suspension of (E)-1-(4-aminobut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide hydrochloride (535 mg, 1.280 mmol) in EtOH (5 mL) was added triethylamine (471 mg, 4.65 mmol) and 4-chloro-3-(3-morpholinopropoxy)-5-nitrobenzamide (400 mg, 1.164 mmol). The reaction vessel was sealed and heated at 120° C. for 20 h. After cooling, an orange solid precipitated from the dark solution. The solid was washed with EtOAc and dried to provide (E)-1-(4-((4-carbamoyl-2-(3-morpholinopropoxy)-6-nitrophenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (457 mg, 0.664 mmol, 57.0% yield). The reaction was repeated three times to provide 1.37 g of the title compound. LCMS m/z = 689 [M+H] + .
步骤2: (E)-1-(4-((2-氨基-4-氨基甲酰基-6-(3-吗啉代丙氧基)苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 2: (E)-1-(4-((2-amino-4-carbamoyl-6-(3-morpholinopropoxy)phenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide
将(E)-1-(4-((4-氨基甲酰基-2-(3-吗啉代丙氧基)-6-硝基苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (1.05 g,1.525 mmol)悬浮在MeOH (16 mL)和28% 氢氧化铵 (5.17 mL, 38.1 mmol)中。在5 min搅拌后,加入次硫酸钠 (1.593 g, 9.15 mmol)在水(4.00 mL)中的溶液,并且随后在室温搅拌2 h。(E)-1-(4-((4-Carbamoyl-2-(3-morpholinopropoxy)-6-nitrophenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (1.05 g, 1.525 mmol) was suspended in MeOH (16 mL) and 28% ammonium hydroxide (5.17 mL, 38.1 mmol). After 5 min stirring, a solution of sodium sulfoxylate (1.593 g, 9.15 mmol) in water (4.00 mL) was added and then stirred at room temperature for 2 h.
加入EtOAc并用水和盐水洗涤有机层。然后,干燥有机相并浓缩以获得作为灰白色固体的粗品(E)-1-(4-((2-氨基-4-氨基甲酰基-6-(3-吗啉代丙氧基)苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (330 mg,0.501 mmol, 32.9%收率)。不经进一步纯化而使用该粗物质。LCMS m/z = 659 [M + H]+。EtOAc was added and the organic layer was washed with water and brine. Then, the organic phase was dried and concentrated to obtain a crude product (E) -1- (4- ((2- amino -4- formamyl -6- (3- morpholino propoxy) phenyl) amino) but-2-ene-1-yl) -2- (1- ethyl -3- methyl -1H- pyrazole -5- formamido) -1H- benzo [d] imidazole -5- carboxamide (330 mg, 0.501 mmol, 32.9% yield) as an off-white solid. The crude material was used without further purification. LCMS m/z = 659 [M + H] + .
步骤3: (E)-2-氨基-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-7-(3-吗啉代丙氧基)-1H-苯并[d]咪唑-5-甲酰胺Step 3: (E)-2-amino-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-7-(3-morpholinopropoxy)-1H-benzo[d]imidazole-5-carboxamide
向(E)-1-(4-((2-氨基-4-氨基甲酰基-6-(3-吗啉代丙氧基)苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (330 mg,0.501 mmol)在MeOH (15 mL)中的溶液加入溴化氰 (159 mg, 1.503 mmol),并在室温搅拌该反应混合物3 h。通过加入EtOAc实现产物的沉淀并随后搅拌1 h。过滤该固体,用EtOAc洗涤并干燥以提供作为浅棕色固体的(E)-2-氨基-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-7-(3-吗啉代丙氧基)-1H-苯并[d]咪唑-5-甲酰胺 (284 mg, 0.416 mmol, 83%收率)。不经进一步纯化而使用该物质。 LCMS m/z = 684 [M + H]+。To a solution of (E)-1-(4-((2-amino-4-carbamoyl-6-(3-morpholinopropoxy)phenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (330 mg, 0.501 mmol) in MeOH (15 mL) was added cyanogen bromide (159 mg, 1.503 mmol) and the reaction mixture was stirred at room temperature for 3 h. Precipitation of the product was achieved by addition of EtOAc and subsequent stirring for 1 h. The solid was filtered, washed with EtOAc and dried to afford (E)-2-amino-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-7-(3-morpholinopropoxy)-1H-benzo[d]imidazole-5-carboxamide (284 mg, 0.416 mmol, 83% yield) as a light brown solid. The material was used without further purification. LCMS m/z = 684 [M + H] + .
步骤4: (E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-吗啉代丙氧基)-1H-苯并[d]咪唑-5-甲酰胺Step 4: (E)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-morpholinopropoxy)-1H-benzo[d]imidazole-5-carboxamide
在室温向(E)-2-氨基-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-7-(3-吗啉代丙氧基)-1H-苯并[d]咪唑-5-甲酰胺 (260 mg, 0.380 mmol)在DMF (4 mL)中的悬浮液加入1-乙基-3-甲基-1H-吡唑-5-甲酸 (117 mg, 0.760 mmol)和1H-苯并[d][1,2,3]三唑-1-醇 水合物(58.2 mg, 0.380mmol)和2-(3H-[1,2,3]三唑并[4,5-b]吡啶-3-基)-1,1,3,3-四甲基异脲鎓六氟磷酸盐(V)(289 mg, 0.760 mmol)和三乙胺 (0.212 mL, 1.521 mmol)在DMF (4 mL)中的溶液。在室温搅拌该混合物过夜。然后,加入水(10 mL)并将所得浑浊溶液放入冰箱中3 h。过滤所得沉淀物(180 mg)并与来自较早反应的80 mg另外的粗固体合并。通过硅胶层析法 (ISCO 24g柱, 0-35%梯度的在DCM中的MeOH)进一步纯化粗产物以在除去溶剂后提供(E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-吗啉代丙氧基)-1H-苯并[d]咪唑-5-甲酰胺 (140 mg, 0.171 mmol)。1H NMR (甲醇-d 4 , 600 MHz): δ ppm 7.96 (s, 1 H),7.71 (dd, J=8.3, 1.6 Hz, 1 H), 7.56 (s, 1 H), 7.35 (d, J=8.4 Hz, 1 H), 7.27(s, 1 H), 6.62 (s, 1 H), 6.55 (s, 1 H), 5.95 (dt, J=15.5, 5.1 Hz, 1 H), 5.76-5.83 (m, 1 H), 5.06 (br d, J=4.6 Hz, 2 H), 4.86 (br d, J=5.3 Hz, 2 H), 4.63(s, 2 H), 4.56 (q, J=7.0 Hz, 2 H), 3.99 (t, J=6.1 Hz, 2 H), 3.64 (br t, J=4.2Hz, 4 H), 2.43-2.48 (m, 2 H), 2.40 (br s, 4 H), 2.21 (s, 3 H), 2.19 (s, 3 H),1.75-1.81 (m, 2 H), 1.37 (t, J=7.1 Hz, 3 H), 1.32 (t, J=7.2 Hz, 3 H)。LCMS m/z= 820.9 [M + H]+。To a suspension of (E)-2-amino-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-7-(3-morpholinopropoxy)-1H-benzo[d]imidazole-5-carboxamide (260 mg, 0.380 mmol) in DMF (4 mL) was added 1-ethyl-3-methyl-1H-pyrazole-5-carboxylic acid (117 mg, 0.760 mmol) and 1H-benzo[d][1,2,3]triazol-1-ol hydrate (58.2 mg, 0.760 mmol) at room temperature. 3-[(2 ... The crude product was further purified by silica gel chromatography (ISCO 24 g column, 0-35% gradient of MeOH in DCM) to afford after removal of solvent (E)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-morpholinopropoxy)-1H-benzo[d]imidazole-5-carboxamide (140 mg, 0.171 mmol). 1 H NMR (Methanol - d 4 , 600 MHz): δ ppm 7.96 (s, 1 H), 7.71 (dd, J =8.3, 1.6 Hz, 1 H), 7.56 (s, 1 H), 7.35 (d, J =8.4 Hz, 1 H), 7.27 (s, 1 H), 6.62 (s, 1 H ), 6.55 (s, 1 H), 5.95 (dt, J =15.5, 5.1 Hz, 1 H), 5.76-5.83 (m, 1 H), 5.06 (br d, J =4.6 Hz, 2 H), 4.86 (br d, J =5.3 Hz, 2 H), 4.63(s, 2 H), 4.5 6 (q, J =7.0 Hz, 2 H), 3.99 (t, J =6.1 Hz, 2 H), 3.64 (br t, J =4.2Hz, 4 H), 2.43-2.48 (m, 2 H), 2.40 (br s, 4 H), 2.21 (s, 3 H), 2.19 (s, 3 H),1.75-1.81 (m, 2 H) , 1.37 (t, J =7.1 Hz, 3 H), 1.32 (t, J =7.2 Hz, 3 H). LCMS m/z = 820.9 [M + H] + .
步骤5: (E)-1-((E)-4-((Z)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 5: (E)-1-((E)-4-((Z)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向(E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-吗啉代丙氧基)-1H-苯并[d]咪唑-5-甲酰胺 (52 mg, 0.063 mmol)在DMF (2 mL)中的悬浮液加入碳酸铯 (62.0 mg, 0.190 mmol)和碘甲烷(9.91 µl, 0.159 mmol)。在室温搅拌该反应混合物12 h。蒸发溶剂并通过硅胶层析法 (0-25%梯度的MeOH / DCM, 硅胶柱 12g)纯化残余物以获得清洁产物(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (44 mg, 0.052 mmol, 82%收率)。1H NMR (DMSO-d 6, 600 MHz): δ ppm 8.07(br s, 1 H), 7.80 (br d, J=8.3 Hz, 1 H), 7.74 (br s, 1 H), 7.48 (br d, J=8.4Hz, 1 H), 7.41 (s, 1 H), 6.29-6.44 (m, 2 H), 5.83-5.99 (m, 1 H), 5.60-5.76(m, 1 H), 4.81-4.94 (m, 2 H), 4.75 (br d, J=5.1 Hz, 2 H), 4.38-4.55 (m, 4 H),4.06 (br s, 2 H), 3.54 (br s, 3 H), 3.45-3.59 (m, 7 H), 2.25-2.30 (m, 2 H),2.15-2.37 (m, 4 H), 2.11 (br d, J=7.0 Hz, 6 H), 1.72 (br s, 2 H),1.19-1.24(m, 3 H), 1.14-1.26 (m, 3 H)。LCMS m/z = 848 [M + H]+。To a suspension of (E)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-morpholinopropoxy)-1H-benzo[d]imidazole-5-carboxamide (52 mg, 0.063 mmol) in DMF (2 mL) was added cesium carbonate (62.0 mg, 0.190 mmol) and iodomethane (9.91 µl, 0.159 mmol). The reaction mixture was stirred at room temperature for 12 h. The solvent was evaporated and the residue was purified by silica gel chromatography (0-25% gradient of MeOH/DCM, silica gel column 12 g) to obtain the clean product (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (44 mg, 0.052 mmol, 82% yield). 1 H NMR (DMSO- d 6 , 600 MHz): δ ppm 8.07 (br s, 1 H), 7.80 (br d, J =8.3 Hz, 1 H), 7.74 (br s, 1 H), 7.48 (br d, J =8.4Hz, 1 H), 7.41 (s, 1 H), 6.29-6. 44 (m, 2 H), 5.83-5.99 (m, 1 H), 5.60-5.76 (m, 1 H), 4.81-4.94 (m, 2 H), 4.75 (br d, J =5.1 Hz, 2 H), 4.38-4.55 (m, 4 H), 4.06 (br s, 2 H), 3.54 (br s, 3 H), 3.45-3.59 (m, 7 H), 2.25-2.30 (m, 2 H), 2.15-2.37 (m, 4 H), 2.11 (br d, J =7.0 Hz, 6 H), 1.72 (br s, 2 H), 1.19-1.24 (m, 3 H), 1.14-1.26 (m, 3 H). LCMS m/z = 848 [M + H] + .
通过上述方法制得的化合物可以互变异构体/同分异构体的形式存在,例如(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺The compounds prepared by the above method may exist in the form of tautomers/isomers, such as (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
实施例2Example 2
(E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)butyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
步骤1: (4-((4-氨基甲酰基-2-硝基苯基)氨基)丁基)氨基甲酸叔丁酯Step 1: tert-Butyl (4-((4-carbamoyl-2-nitrophenyl)amino)butyl)carbamate
在70℃搅拌(4-氨基丁基)氨基甲酸叔丁酯 (5.00 g, 26.6 mmol)、4-氟-3-硝基苯甲酰胺 (4.89 g, 26.6 mmol)和K2CO3 (4.04 g, 29.2 mmol)在DMSO (25mL)中的混合物2 h。将该反应冷却至室温并通过加料漏斗用125 mL水缓慢稀释。通过过滤分离所得固体,干燥并放置在真空炉中在56℃3天以提供作为黄色固体的标题化合物(9.2 g, 26.1mmol,98%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.67 (d, J=2.02 Hz, 1 H) 8.40 (t, J=5.43 Hz, 1 H) 8.01 (d, J=6.82 Hz, 2 H) 7.30 (br. s., 1 H) 7.12 (d, J=9.09 Hz,1 H) 6.87 (br. s., 1 H) 3.42 (q, J=6.57 Hz, 2 H) 2.91 - 3.01 (m, 2 H) 1.60(d, J=6.57 Hz, 2 H) 1.43 - 1.54 (m, 2 H) 1.38 (s, 9 H)。LCMS [M + H]+ = 353。A mixture of tert-butyl (4-aminobutyl)carbamate (5.00 g, 26.6 mmol), 4-fluoro-3-nitrobenzamide (4.89 g, 26.6 mmol) and K2CO3 (4.04 g, 29.2 mmol) in DMSO (25 mL) was stirred at 70°C for 2 h . The reaction was cooled to room temperature and slowly diluted with 125 mL of water via an addition funnel. The resulting solid was isolated by filtration, dried and placed in a vacuum oven at 56°C for 3 days to provide the title compound (9.2 g, 26.1 mmol, 98% yield) as a yellow solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.67 (d, J =2.02 Hz, 1 H) 8.40 (t, J =5.43 Hz, 1 H) 8.01 (d, J =6.82 Hz, 2 H) 7.30 (br. s., 1 H) 7.12 (d, J =9.09 Hz, 1 H) 6.87 (br. s., 1 H) 3.42 (q, J =6.57 Hz, 2 H) 2.91 - 3.01 (m, 2 H) 1.60 (d, J =6.57 Hz, 2 H) 1.43 - 1.54 (m, 2 H) 1.38 (s, 9 H). LCMS [M+H] + = 353.
步骤2: (4-((2-氨基-4-氨基甲酰基苯基)氨基)丁基)氨基甲酸叔丁酯Step 2: tert-Butyl (4-((2-amino-4-carbamoylphenyl)amino)butyl)carbamate
向500 mL圆底烧瓶装入(4-((4-氨基甲酰基-2-硝基苯基)氨基)丁基)氨基甲酸叔丁酯 (9.2 g, 26.1 mmol)、10% Pd/C (0.920 g, 8.64 mmol) (Degussa湿型)、EtOH (100mL)和MeOH (100 mL)。将该烧瓶排空并伴随搅拌放置在氢气球下。将冷凝器放置在烧瓶上方并将氢气球放置在冷凝器上。将该混合物在室温搅拌20 h,然后排空该烧瓶,并使用EtOH助洗将该悬浮液过滤通过硅藻土床。在真空下浓缩该滤液并放置在高真空下以提供作为黑色固体的标题化合物(8.4 g, 26.1 mmol, 100%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm7.44 (br. s., 1 H) 7.04 - 7.15 (m, 2 H) 6.85 (t, J=5.43 Hz, 1 H) 6.74 (br.s., 1 H) 6.37 (d, J=8.08 Hz, 1 H) 4.89 (t, J=5.18 Hz, 1 H) 4.60 (br. s., 2 H)3.07 (q, J=6.48 Hz, 2 H) 2.97 (q, J=6.40 Hz, 2 H) 1.45 - 1.64 (m, 4 H) 1.39(s, 9 H)。LCMS [M + H]+ = 323.1。A 500 mL round bottom flask was charged with tert-butyl (4-((4-carbamoyl-2-nitrophenyl)amino)butyl)carbamate (9.2 g, 26.1 mmol), 10% Pd/C (0.920 g, 8.64 mmol) (Degussa wet type), EtOH (100 mL) and MeOH (100 mL). The flask was evacuated and placed under a hydrogen balloon with stirring. A condenser was placed above the flask and a hydrogen balloon was placed on the condenser. The mixture was stirred at room temperature for 20 h, then the flask was evacuated and the suspension was filtered through a diatomaceous earth bed using EtOH as a washing aid. The filtrate was concentrated under vacuum and placed under high vacuum to provide the title compound (8.4 g, 26.1 mmol, 100% yield) as a black solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm7.44 (br. s., 1 H) 7.04 - 7.15 (m, 2 H) 6.85 (t, J =5.43 Hz, 1 H) 6.74 (br.s., 1 H) 6.37 (d, J =8.08 Hz, 1 H) 4.89 (t, J =5.18 Hz, 1 H) 4.60 (br. s., 2 H)3.07 (q, J =6.48 Hz, 2 H) 2.97 (q, J =6.40 Hz, 2 H) 1.45 - 1.64 (m, 4 H) 1.39 (s, 9 H). LCMS [M + H] + = 323.1.
步骤3: (4-(2-氨基-5-氨基甲酰基-1H-苯并[d]咪唑-1-基)丁基)氨基甲酸叔丁酯, 氢溴酸盐Step 3: tert-Butyl (4-(2-amino-5-carbamoyl-1H-benzo[d]imidazol-1-yl)butyl)carbamate, hydrobromide
将(4-((2-氨基-4-氨基甲酰基苯基)氨基)丁基)氨基甲酸叔丁酯 (8.40 g, 26.1mmol)溶解在MeOH (110 mL)中,并经由注射器加入在CH3CN中的5 M溴化氰溶 液 (5.73mL, 28.7 mmol)。为该深色反应加盖(capped)并在室温搅拌15 h。在真空中浓缩该反应并放置在高真空下以提供作为深色固体的标题化合物(11.17 g, 26.1 mmol, 100%收率)。1HNMR (400 MHz, DMSO-d 6) δ ppm 12.85 (br. s., 1 H) 8.74 (br. s., 2 H) 8.08 (br.s., 1 H) 7.80 - 7.90 (m, 2 H) 7.64 (d, J=8.34 Hz, 1 H) 7.44 (br. s., 1 H)6.89 (t, J=5.56 Hz, 1 H) 4.15 (t, J=7.20 Hz, 2 H) 2.96 (q, J=6.32 Hz, 2 H)1.66 (d, J=7.07 Hz, 2 H) 1.42 - 1.50 (m, 2 H) 1.38 (s, 9 H)。LCMS [M + H]+ =348.1。Tert-butyl (4-((2-amino-4-carbamoylphenyl)amino)butyl)carbamate (8.40 g, 26.1 mmol) was dissolved in MeOH (110 mL) and a 5 M solution of cyanogen bromide in CH3CN (5.73 mL, 28.7 mmol) was added via syringe. The dark reaction was capped and stirred at room temperature for 15 h. The reaction was concentrated in vacuo and placed under high vacuum to provide the title compound as a dark solid (11.17 g, 26.1 mmol, 100% yield). 1 HNMR (400 MHz, DMSO- d 6 ) δ ppm 12.85 (br. s., 1 H) 8.74 (br. s., 2 H) 8.08 (br.s., 1 H) 7.80 - 7.90 (m, 2 H) 7.64 (d, J =8.34 Hz, 1 H) 7.44 (br. s., 1 H)6.89 (t, J =5.56 Hz, 1 H) 4.15 (t, J =7.20 Hz, 2 H) 2.96 (q, J =6.32 Hz, 2 H)1.66 (d, J =7.07 Hz, 2 H) 1.42 - 1.50 (m, 2 H) 1.38 (s, 9 H). LCMS [M+H] + = 348.1.
步骤4: (4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)氨基甲酸叔丁酯Step 4: tert-Butyl (4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)carbamate
在室温搅拌(4-(2-氨基-5-氨基甲酰基-1H-苯并[d]咪唑-1-基)丁基)氨基甲酸叔丁酯, 氢溴酸盐 (11.17 g, 26.1 mmol)、1-乙基-3-甲基-1H-吡唑-5-甲酸 (4.82 g,31.3 mmol)、HATU (11.90 g, 31.3 mmol)、DIPEA (18.22 mL, 104 mmol)和HOBt (1.997g, 13.04 mmol)在DMF (100 mL)中的混合物 21 h。用300 mL水和300 mL的EtOAc稀释该反应,转移到分液漏斗中,并分离各层,并用EtOAc (2 x 150 mL)萃取水层。用饱和NH4Cl (2x 200 mL)、水(1 x 200 mL)和盐水(2 x 200 mL)洗涤合并的EtOAc层。经Na2SO4干燥有机层、过滤、在真空中浓缩,并置于高真空下。经由层析法在硅胶(ISCO® Combiflash, 0-20%MeOH: DCM, 330 g柱, 装载在50 mL DCM中)上纯化该固体。合并所需级分,在真空中浓缩,并置于高真空下以提供作为紫色固体的标题化合物(9.53 g, 19.71 mmol, 76%收率)。1HNMR (400 MHz, DMSO-d 6) δ ppm 12.85 (s, 1 H) 8.01 (br. s., 2 H) 7.81 (d, J=8.34 Hz, 1 H) 7.59 (d, J=8.34 Hz, 1 H) 7.36 (br. s., 1 H) 6.80 - 6.86 (m, 1H) 6.68 (s, 1 H) 4.64 (q, J=6.82 Hz, 2 H) 4.23 (t, J=6.44 Hz, 2 H) 2.98 (d, J=5.81 Hz, 2 H) 2.19 (s, 3 H) 1.76 (d, J=6.57 Hz, 2 H) 1.40 - 1.48 (m, 2 H)1.30 - 1.40 (m, 13 H)。LCMS [M + H]+ = 484.3。A mixture of tert-butyl (4-(2-amino-5-carbamoyl-1H-benzo[d]imidazol-1-yl)butyl)carbamate, hydrobromide (11.17 g, 26.1 mmol), 1-ethyl-3-methyl-1H-pyrazole-5-carboxylic acid (4.82 g, 31.3 mmol), HATU (11.90 g, 31.3 mmol), DIPEA (18.22 mL, 104 mmol), and HOBt (1.997 g, 13.04 mmol) in DMF (100 mL) was stirred at room temperature for 21 h. The reaction was diluted with 300 mL of water and 300 mL of EtOAc, transferred to a separatory funnel, the layers separated, and the aqueous layer extracted with EtOAc (2 x 150 mL). The combined EtOAc layers were washed with saturated NH 4 Cl (2x 200 mL), water (1 x 200 mL), and brine (2 x 200 mL). The organic layer was dried over Na 2 SO 4 , filtered, concentrated in vacuo, and placed under high vacuum. The solid was purified via chromatography on silica gel (ISCO ® Combiflash, 0-20% MeOH: DCM, 330 g column, loaded in 50 mL DCM). The desired fractions were combined, concentrated in vacuo, and placed under high vacuum to provide the title compound (9.53 g, 19.71 mmol, 76% yield) as a purple solid. 1 HNMR (400 MHz, DMSO- d 6 ) δ ppm 12.85 (s, 1 H) 8.01 (br. s., 2 H) 7.81 (d, J =8.34 Hz, 1 H) 7.59 (d, J =8.34 Hz, 1 H) 7.36 (br. s., 1 H) 6.80 - 6.86 ( m, 1H) 6.68 (s, 1 H) 4.64 (q, J =6.82 Hz, 2 H) 4.23 (t, J =6.44 Hz, 2 H) 2.98 (d, J =5.81 Hz, 2 H) 2.19 (s, 3 H) 1.76 (d, J =6.57 Hz, 2 H) 1.40 - 1.48 (m, 2 H)1.30 - 1.40 (m, 13 H). LCMS [M + H] + = 484.3.
步骤5: 1-(4-氨基丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐Step 5: 1-(4-aminobutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide, 2-hydrochloride
用在1,4-二氧杂环己烷中的4 M HCl (42.0 mL, 168 mmol)处理容纳(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)氨基甲酸叔丁酯 (9.53 g, 19.71 mmol)的冰凉的500 mL圆底烧瓶。除去冰浴并在室温搅拌紫色浆料2.5 h。然后,在真空中浓缩该反应,置于高真空下,并将所得固体置于50 ℃的真空炉中15 h并在高真空下冷却以提供作为灰色固体的还含有1,4-二氧杂环己烷的不纯的标题化合物(11.89 g, 假定19.7 mmol, 100%收率)。不经进一步纯化原样使用物质。1H NMR (400MHz, DMSO-d 6) δ ppm 12.91 (br. s, 1 H) 8.03 (d, J=1.26 Hz, 2 H) 7.77 - 7.87(m, 4 H) 7.62 (d, J=8.34 Hz, 1 H) 7.38 (br. s., 1 H) 6.70 (s, 1 H) 6-5 (br.s, 1 H), 4.63 (q, J=7.07 Hz, 2 H) 4.28 (t, J=6.57 Hz, 2 H) 2.77 - 2.87 (m, 2H) 2.20 (s, 3 H) 1.81 - 1.91 (m, 2 H) 1.52 - 1.60 (m, 2 H) 1.38 (t, J=7.07Hz, 3 H)。LCMS [M + H]+ = 384.2。An ice-cooled 500 mL round bottom flask containing tert-butyl (4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)carbamate (9.53 g, 19.71 mmol) was treated with 4 M HCl in 1,4-dioxane (42.0 mL, 168 mmol). The ice bath was removed and the purple slurry was stirred at room temperature for 2.5 h. The reaction was then concentrated in vacuo, placed under high vacuum, and the resulting solid was placed in a vacuum oven at 50 °C for 15 h and cooled under high vacuum to provide the impure title compound (11.89 g, assumed 19.7 mmol, 100% yield) as a gray solid also containing 1,4-dioxane. The material was used as is without further purification. 1 H NMR (400MHz, DMSO- d 6 ) δ ppm 12.91 (br. s, 1 H) 8.03 (d, J =1.26 Hz, 2 H) 7.77 - 7.87(m, 4 H) 7.62 (d, J =8.34 Hz, 1 H) 7.38 (br. s., 1 H) 6.70 (s, 1 H) 6-5 (br.s, 1 H), 4.63 (q, J =7.07 Hz, 2 H) 4.28 (t, J =6.57 Hz, 2 H) 2.77 - 2.87 (m, 2H) 2.20 (s, 3 H) 1.81 - 1.91 (m, 2 H) 1.52 - 1.60 (m, 2 H) 1.38 (t, J =7.07Hz, 3 H). LCMS [M + H] + = 384.2.
步骤6: 4-((4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)氨基)-3-甲氧基-5-硝基苯甲酸甲酯Step 6: 4-((4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)amino)-3-methoxy-5-nitrobenzoic acid methyl ester
向配备有冷凝器、大搅拌棒和内部温度计的250 mL 3-颈圆底烧瓶装入1-(4-氨基丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐(9.38 g, 20.55 mmol)和4-氯-3-甲氧基-5-硝基苯甲酸甲酯 (5.048 g, 20.55 mmol)。加入DMSO (50 mL),然后加入DIPEA (17.95 mL, 103 mmol)并在100 ℃加热深色悬浮液大致24 h,冷却,并逐滴加入至500 mL搅拌水中。在完成加料后,搅拌所得橙色悬浮液20 min并过滤。用水和己烷洗涤分离的橙红色糊料,在布氏漏斗中干燥,然后在56 ℃的真空炉中干燥20 h。然后,用Et2O (60mL)研磨微红色固体,并通过过滤分离。重复该研磨和过滤。将所得固体置于56 ℃的真空炉中3天以提供作为微红色固体的标题化合物(11.17 g, 18.85mmol, 92%收率) 。1H NMR (400 MHz, DMSO-d 6) δ ppm 12.78 (br. s., 1 H) 8.12 (s, 1H) 7.99 (s, 1 H) 7.93 (d, J=7.53 Hz, 2 H) 7.79 (d, J=8.28 Hz, 1 H) 7.53 (d, J=7.78 Hz, 1 H) 7.36 (s, 1 H) 7.31 (br. s., 1 H) 6.60 (s, 1 H) 4.60 (d, J=7.03Hz, 2 H) 4.23 (br. s., 2 H) 3.84 (s, 3 H) 3.80 (s, 3 H) 3.53 (d, J=5.77 Hz, 2H) 2.15 (s, 3 H) 1.82 (br. s., 2 H) 1.62 (br. s., 2 H) 1.35 (t, J=7.03 Hz, 3H)。LCMS [M + H]+ = 711.6。A 250 mL 3-neck round bottom flask equipped with a condenser, large stirring bar and internal thermometer was charged with 1-(4-aminobutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide, 2 hydrochloride (9.38 g, 20.55 mmol) and methyl 4-chloro-3-methoxy-5-nitrobenzoate (5.048 g, 20.55 mmol). DMSO (50 mL) was added followed by DIPEA (17.95 mL, 103 mmol) and the dark suspension was heated at 100 °C for approximately 24 h, cooled, and added dropwise to 500 mL of stirring water. After the addition was complete, the resulting orange suspension was stirred for 20 min and filtered. The separated orange-red paste was washed with water and hexanes, dried in a Buchner funnel, and then dried in a vacuum oven at 56 °C for 20 h. The reddish solid was then triturated with Et20 (60 mL) and isolated by filtration. The trituration and filtration were repeated. The resulting solid was placed in a vacuum oven at 56°C for 3 days to provide the title compound (11.17 g, 18.85 mmol, 92% yield) as a reddish solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 12.78 (br. s., 1 H) 8.12 (s, 1H) 7.99 (s, 1 H) 7.93 (d, J =7.53 Hz, 2 H) 7.79 (d, J =8.28 Hz, 1 H) 7.53 (d, J =7.78 Hz , 1 H) 7.36 (s, 1 H) 7.31 (br. s., 1 H) 6.60 (s, 1 H) 4.60 (d, J =7.03Hz, 2 H) 4.23 (br. s., 2 H) 3.84 (s, 3 H) 3.80 (s, 3 H) 3.53 (d, J =5.77 Hz, 2H) 2.15 (s, 3 H) 1.82 (br. s., 2 H) 1.62 (br. s., 2 H) 1.35 (t, J =7.03 Hz, 3H). LCMS [M + H] + = 711.6.
步骤7: 3-氨基-4-((4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)氨基)-5-甲氧基苯甲酸甲酯Step 7: 3-amino-4-((4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)amino)-5-methoxybenzoic acid methyl ester
在室温在250 mL 圆底烧瓶中伴随着搅拌将4-((4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d] 咪唑-1-基)丁基)氨基)-3-甲氧基-5-硝基苯甲酸甲酯 (5.0 g, 8.44 mmol)大多溶解于DMF (50 mL)。加入雷尼镍(在水中的Raney 2800镍, 大约10 mL浆料, Aldrich)并将冷凝器放在烧瓶顶上。将配有氢气球的3通旋塞阀接合器放置在冷凝器上方并排空该装置,填充氢气,排空,并最后填充氢气。在70 ℃加热该反应7 h。加入另外的8 mL 雷尼镍并在70℃加热该反应14 h。将该反应冷却并在用DMF洗涤的同时过滤通过硅藻土。将含有所需产物的滤液—来自雷尼镍浆料的大约100 mL DMF和20 mL水的溶液作为溶液直接用于接下来的反应中。假定定量收率(Assumed quantitativeyield)。LCMS [M + H]+ = 563.4。Methyl 4-((4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)amino)-3-methoxy-5-nitrobenzoate (5.0 g, 8.44 mmol) was mostly dissolved in DMF (50 mL) in a 250 mL round bottom flask at room temperature with stirring. Raney nickel (Raney 2800 nickel in water, approximately 10 mL slurry, Aldrich) was added and a condenser was placed on top of the flask. A 3-way stopcock adapter with a hydrogen balloon was placed over the condenser and the apparatus was evacuated, filled with hydrogen, evacuated, and finally filled with hydrogen. The reaction was heated at 70 °C for 7 h. An additional 8 mL of Raney nickel was added and the reaction was heated at 70 °C for 14 h. The reaction was cooled and filtered through celite while washing with DMF. The filtrate containing the desired product, a solution of approximately 100 mL DMF and 20 mL water from the Raney nickel slurry, was used directly as a solution in the next reaction. Assumed quantitative yield. LCMS [M + H] + = 563.4.
步骤8: 2-氨基-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酸甲酯, 氢溴酸盐Step 8: 2-amino-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-7-methoxy-1H-benzo[d]imidazole-5-carboxylic acid methyl ester, hydrobromide
采用在CH3CN中的5 M 溴化氰 (1.875 mL, 9.37 mmol)处理3-氨基-4-((4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)氨基)-5-甲氧基苯甲酸甲酯 (来自先前步骤的在DMF/水中的溶液)并在室温搅拌所得溶液22 h。在真空中浓缩该反应并置于高真空下以提供棕色半固体。采用EtOAc研磨该半固体,剧烈搅拌30 min,并通过过滤分离所得固体并在布氏漏斗中干燥以提供作为褐色固体的不纯的标题产物(5.08 g)。不经纯化即使用这个不纯的物质。LCMS [M + H]+ = 588.5。Methyl 3-amino-4-((4-(5-carbamoyl-2-(1-ethyl-3-methyl- 1H -pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)amino)-5-methoxybenzoate (solution in DMF/water from the previous step) was treated with 5 M cyanogen bromide in CH 3 CN (1.875 mL, 9.37 mmol) and the resulting solution was stirred at room temperature for 22 h. The reaction was concentrated in vacuo and placed under high vacuum to afford a brown semisolid. The semisolid was triturated with EtOAc, stirred vigorously for 30 min, and the resulting solid was isolated by filtration and dried in a Buchner funnel to afford the impure title product as a brown solid (5.08 g). This impure material was used without purification. LCMS [M + H] + = 588.5.
步骤9: 1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酸甲酯Step 9: 1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazol-5-carboxylic acid methyl ester
在室温搅拌2-氨基-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酸甲酯, 氢溴酸盐(5.073 g, 7.59 mmol)、1-乙基-3-甲基-1H-吡唑-5-甲酸 (1.287 g, 8.35 mmol)、HATU(3.46 g, 9.11 mmol)和DIPEA (3.98 mL, 22.76 mmol)在DMF (30 mL)中的混合物17 h。在真空中浓缩该反应,然后用水(100 mL)研磨所得残余物并搅拌30 min。过滤所得悬浮液并在布氏漏斗中部分干燥以提供深褐色固体。将固体大多溶解于150 mL的10% IPA: 氯仿中,用水稀释并过滤。然后,分离滤液层,并经Na2SO4干燥有机层,过滤,浓缩并置于高真空下以提供褐色固体。采用温10% IPA: 氯仿 (100 mL)研磨固体并过滤。分离滤液层,经Na2SO4干燥有机层,过滤,加入初始的褐色固体中,在真空中浓缩并置于高真空下。经由层析法在硅胶(Biotage® Isolera, 120 gm Gold柱, 经30 min的0-10% MeOH: DCM, 作为在DCM/MeOH中的溶液装载)上纯化该固体。合并所需产物级分,浓缩,并置于高真空下以提供浅褐色固体。采用DCM (50 mL)研磨该固体并通过过滤分离,并置于56 ℃的真空炉中30 h以提供作为白色固体的1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酸甲酯(1.0 g, 1.4 mmol, 18%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm12.89 (s, 1 H) 12.82 (s, 1 H) 7.90 - 8.01 (m, 2 H) 7.70 - 7.81 (m, 2 H) 7.53(d, J=8.28 Hz, 1 H) 7.30 - 7.40 (m, 2 H) 6.59 (d, J=5.02 Hz, 2 H) 4.50 - 4.64(m, 4 H) 4.38 (br. s., 2 H) 4.27 (br. s., 2 H) 3.87 (d, J=3.76 Hz, 6 H) 2.10(s, 6 H) 1.86 (br. s., 4 H) 1.23 - 1.39 (m, 6 H)。LCMS [M + H]+ = 724.5。A mixture of methyl 2-amino-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-7-methoxy-1H-benzo[d]imidazole-5-carboxylate, hydrobromide (5.073 g, 7.59 mmol), 1-ethyl-3-methyl-1H-pyrazole-5-carboxylic acid (1.287 g, 8.35 mmol), HATU (3.46 g, 9.11 mmol) and DIPEA (3.98 mL, 22.76 mmol) in DMF (30 mL) was stirred at room temperature for 17 h. The reaction was concentrated in vacuo and the resulting residue was triturated with water (100 mL) and stirred for 30 min. The resulting suspension was filtered and partially dried in a Buchner funnel to provide a dark brown solid. Solid is dissolved in 150 mL of 10% IPA: chloroform mostly, diluted with water and filtered.Then, separate filtrate layer, and through Na 2 SO 4 dry organic layer, filter, concentrate and be placed under high vacuum to provide brown solid.Adopt warm 10% IPA: chloroform (100 mL) grinding solid and filter.Separate filtrate layer, through Na 2 SO 4 dry organic layer, filter, add in initial brown solid, concentrate and be placed under high vacuum in vacuum.Via chromatography on silica gel (Biotage ® Isolera, 120 gm Gold post, through 30 min 0-10% MeOH: DCM, as the solution in DCM/MeOH load) upper purification of this solid.Merge required product fraction, concentrate, and be placed under high vacuum to provide light brown solid. The solid was triturated with DCM (50 mL) and isolated by filtration and placed in a vacuum oven at 56 °C for 30 h to afford methyl 1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxylate (1.0 g, 1.4 mmol, 18% yield) as a white solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm12.89 (s, 1 H) 12.82 (s, 1 H) 7.90 - 8.01 (m, 2 H) 7.70 - 7.81 (m, 2 H) 7.53 (d, J =8.28 Hz, 1 H) 7.30 - 7.40 (m, 2 H ) 6.59 (d, J =5.02 Hz, 2 H) 4.50 - 4.64(m, 4 H) 4.38 (br. s., 2 H) 4.27 (br. s., 2 H) 3.87 (d, J =3.76 Hz, 6 H) 2.10(s, 6 H) 1.86 (br. s., 4 H) 1.23 - 1.39 (m, 6H). LCMS [M + H] + = 724.5.
步骤10: 1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酸Step 10: 1-(4-(5-Carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxylic acid
向1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酸甲酯 (550 mg, 0.760 mmol)在MeOH (11 mL)和水(11 mL)中的悬浮液加入NaOH(304 mg, 7.60 mmol)。在室温搅拌该反应过夜。在真空中除去MeOH并采用1 N HCl处理所得溶液直到pH~3。过滤所得浆料并在真空炉中干燥滤饼以提供作为白色固体的1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酸 (650 mg, 0.687mmol, 90%收率)。不经进一步纯化即将该化合物用于接下来的步骤。LCMS m/z = 710 [M +H]+。To a suspension of methyl 1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxylate (550 mg, 0.760 mmol) in MeOH (11 mL) and water (11 mL) was added NaOH (304 mg, 7.60 mmol). The reaction was stirred at room temperature overnight. The MeOH was removed in vacuo and the resulting solution was treated with 1 N HCl until pH ~3. The resulting slurry was filtered and the filter cake was dried in a vacuum oven to provide 1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxylic acid (650 mg, 0.687 mmol, 90% yield) as a white solid. The compound was used in the next step without further purification. LCMS m/z = 710 [M +H] + .
步骤11: 1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺Step 11: 1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide
向1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酸 (320 mg, 0.338 mmol)和HATU (154 mg, 0.406 mmol)在DMF (3.381 mL)中的溶液加入DIEA (295 µL, 1.691 mmol)。在20 min后,将氯化铵 (54.3 mg, 1.014 mmol)加入该反应混合物并在室温搅拌该反应3天。加入另外的HATU (50 mg, 0.132 mmol)和DIEA(58.9 µl, 0.338 mmol)。在室温搅拌该反应10 min并加入氯化铵 (18.26 mg, 0.338mmol)。为使反应完全,加入HOBt水合物(51.7 mg, 0.338 mmol)并在室温搅拌该反应90min。将该反应干装载至硅胶上并通过硅胶层析法 (ISCO-Rf 12g柱, 0%-30%梯度的MeOH/DCM)纯化以提供作为白色固体的1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺 (175 mg, 0.244 mmol, 72.3%收率)。LCMS m/z = 709 [M+ H]+。To a solution of 1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxylic acid (320 mg, 0.338 mmol) and HATU (154 mg, 0.406 mmol) in DMF (3.381 mL) was added DIEA (295 µL, 1.691 mmol). After 20 min, ammonium chloride (54.3 mg, 1.014 mmol) was added to the reaction mixture and the reaction was stirred at room temperature for 3 days. Additional HATU (50 mg, 0.132 mmol) and DIEA (58.9 µl, 0.338 mmol) were added. The reaction was stirred at room temperature for 10 min and ammonium chloride (18.26 mg, 0.338 mmol) was added. For complete reaction, HOBt hydrate (51.7 mg, 0.338 mmol) was added and the reaction was stirred at room temperature for 90 min. The reaction was dry loaded onto silica gel and purified by silica gel chromatography (ISCO-Rf 12g post, 0%-30% gradient of MeOH/DCM) to provide 1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-formamido)-1H-benzo [d] imidazoles-1-yl) butyl)-2-(1-ethyl-3-methyl-1H-pyrazoles-5-formamido)-7-methoxyl-1H-benzo [d] imidazoles-5-carboxamide (175 mg, 0.244 mmol, 72.3% yield) as white solid. LCMS m/z = 709 [M+ H] + .
步骤12: (E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 12: (E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)butyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺 (100 mg, 0.141 mmol)在DMF (4 mL)中的悬浮液加入碳酸铯 (138 mg, 0.423mmol)和碘甲烷(50.1 mg, 0.353 mmol, 100 uL原液(在780 uL DMF中的220 uL 碘甲烷)。将该反应在室温搅拌2 h,干装载至硅胶上并通过硅胶层析法 (ISCO-Rf, 12g柱, 0%-30%梯度的MeOH/DCM)纯化以获得100 mg固体(通过LCMS,~93%纯)。将所得残余物(100 mg)溶解于MeOH中,干装载至硅胶上并再次纯化(ISCO-Rf, 12g柱, 0%-20%梯度的 MeOH/DCM)。合并纯级分并浓缩至干以提供(E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (33 mg,0.044 mmol, 31.4%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 7.98 - 8.13 (m, 3 H)7.84 (dd, J=8.36, 1.52 Hz, 1 H) 7.69 (d, J=1.01 Hz, 1 H) 7.58 (d, J=8.62 Hz,1 H) 7.47 (d, J=14.19 Hz, 2 H) 7.42 (s, 1 H) 6.46 (d, J=6.84 Hz, 2 H) 4.42 -4.55 (m, 4 H) 4.11 - 4.28 (m, 4 H) 3.83 (s, 3 H) 3.51 (s, 3 H) 3.47 (s, 3 H)2.12 (s, 6 H) 1.74 (br. s., 4 H) 1.24 (td, J=7.10, 1.52 Hz, 6 H)。LCMS m/z =737 [M + H]+。To a suspension of 1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide (100 mg, 0.141 mmol) in DMF (4 mL) was added cesium carbonate (138 mg, 0.423 mmol) and iodomethane (50.1 mg, 0.353 mmol, 100 uL stock (220 uL iodomethane in 780 uL DMF). The reaction was stirred at room temperature for 2 h, dry loaded onto silica gel and purified by silica gel chromatography (ISCO-Rf, 12 g column, 0%-30% gradient of MeOH/DCM) to afford 100 mg solid (by LCMS, ~93% pure). The resulting residue (100 mg) was dissolved in MeOH, dry loaded onto silica gel and purified again (ISCO-Rf, 12 g column, 0%-20% gradient of MeOH/DCM). Pure fractions were combined and concentrated to dryness to provide (E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo [d] imidazole-1-yl)butyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo [d] imidazole-5-carboxamide (33 mg, 0.044 mmol, 31.4% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 7.98 - 8.13 (m, 3 H)7.84 (dd, J =8.36, 1.52 Hz, 1 H) 7.69 (d, J =1.01 Hz, 1 H) 7.58 (d, J =8.62 Hz, 1 H) 7.47 (d, J =14.19 Hz, 2 H) 7.4 2 (s, 1 H) 6.46 (d, J =6.84 Hz, 2 H) 4.42 -4.55 (m, 4 H) 4.11 - 4.28 (m, 4 H) 3.83 (s, 3 H) 3.51 (s, 3 H) 3.47 (s, 3 H)2.12 (s, 6 H) 1.74 (br. s., 4 H) 1.24 (td, J =7.10, 1.52 Hz, 6 H). LCMS m/z =737 [M + H] + .
实施例3Example 3
(5aE,21E)-8-乙基-5,10,18,22-四甲基-7,20-二氧代-5,7,8,11,12,13,14,15,20,22,28,29,30,31-十四氢苯并[4,5]咪唑并[1,2-a]苯并[4,5]咪唑并[2,1-p]二吡唑并[5,1-e:4',3'-l][1,3,6,15,17]五氮杂环二十一碳十烯(pentaazacyclohenicosine)-3,24-二甲酰胺(5aE,21E)-8-ethyl-5,10,18,22-tetramethyl-7,20-dioxo-5,7,8,11,12,13,14,15,20,22,28,29,30,31-tetradecahydrobenzo[4,5]imidazo[1,2-a]benzo[4,5]imidazo[2,1-p]dipyrazolo[5,1-e:4',3'-l][1,3,6,15,17]pentaazacyclohenicosine-3,24-dicarboxamide
步骤1: 3-甲基-1-(5-(三甲基甲硅烷基)戊-4-炔-1-基)-1H-吡唑-5-甲酸乙酯Step 1: 3-Methyl-1-(5-(trimethylsilyl)pent-4-yn-1-yl) -1H -pyrazole-5-carboxylic acid ethyl ester
在60℃在氮气气氛下搅拌3-甲基-1H-吡唑-5-甲酸乙酯(22 g, 143 mmol)、(5-氯戊-1-炔-1-基)三甲基硅烷 (24.94 g, 143 mmol)、K2CO3 (39.4 g, 285 mmol)和DMF (4mL)的混合物过夜。然后,将该混合物溶解在DCM中并用水洗涤。将有机相经无水Na2SO4干燥,过滤,在减压下浓缩,并通过在硅胶上的柱层析法(石油醚/EtOAc = 10:1)纯化以提供作为无色油的3-甲基-1-(5-(三甲基甲硅烷基)戊-4-炔-1-基)-1H-吡唑-5-甲酸乙酯(12.5 g,42.7 mmol, 30%收率)。LCMS [M + H]+ = 293。A mixture of ethyl 3-methyl- 1H -pyrazole-5-carboxylate (22 g, 143 mmol), (5-chloropent-1-yn-1-yl)trimethylsilane (24.94 g, 143 mmol), K 2 CO 3 (39.4 g, 285 mmol) and DMF (4 mL) was stirred at 60° C. under a nitrogen atmosphere overnight. The mixture was then dissolved in DCM and washed with water. The organic phase was dried over anhydrous Na 2 SO 4 , filtered, concentrated under reduced pressure, and purified by column chromatography on silica gel (petroleum ether/EtOAc=10:1) to provide ethyl 3-methyl-1-(5-(trimethylsilyl)pent-4-yn-1-yl) -1H -pyrazole-5-carboxylate (12.5 g, 42.7 mmol, 30% yield) as a colorless oil. LCMS [M + H] + = 293.
步骤2: 3-甲基-1-(戊-4-炔-1-基)-1H-吡唑-5-甲酸乙酯Step 2: 3-Methyl-1-(pent-4-yn-1-yl) -1H -pyrazole-5-carboxylic acid ethyl ester
在室温搅拌3-甲基-1-(5-(三甲基甲硅烷基)戊-4-炔-1-基)-1H-吡唑-5-甲酸乙酯(37.7 g, 129 mmol)、K2CO3 (44.5 g, 322 mmol)和EtOH (800 mL)的混合物过夜。然后,过滤该混合物,并在减压下浓缩滤液。将残余物溶解在DCM中,用水洗涤,经无水Na2SO4干燥,过滤,并在减压下浓缩以提供作为无色油的3-甲基-1-(戊-4-炔-1-基)-1H-吡唑-5-甲酸乙酯(20 g, 91 mmol, 70.4%收率)。LCMS [M + H]+ = 221。A mixture of ethyl 3-methyl-1-(5-(trimethylsilyl)pent-4-yn-1-yl) -1H -pyrazole-5-carboxylate (37.7 g, 129 mmol), K 2 CO 3 (44.5 g, 322 mmol) and EtOH (800 mL) was stirred at room temperature overnight. Then, the mixture was filtered and the filtrate was concentrated under reduced pressure. The residue was dissolved in DCM, washed with water, dried over anhydrous Na 2 SO 4 , filtered, and concentrated under reduced pressure to provide ethyl 3-methyl-1-(pent-4-yn-1-yl) -1H -pyrazole-5-carboxylate (20 g, 91 mmol, 70.4% yield) as a colorless oil. LCMS [M + H] + = 221.
步骤3: 1-乙基-3-甲基-1H-吡唑-5-甲酸苄酯Step 3: 1-ethyl-3-methyl- 1H -pyrazole-5-carboxylic acid benzyl ester
在60℃搅拌1-乙基-3-甲基-1H-吡唑-5-甲酸 (20 g, 130 mmol), (溴甲基)苯(22.2 g, 130 mmol)、K2CO3 (26.9 g, 195 mmol)和DMF (200 mL)的混合物过夜。然后,将该混合物溶解在DCM中,用水洗涤,经无水Na2SO4干燥,过滤,在减压下浓缩,并通过柱层析法在硅胶上纯化(石油醚/EtOAc = 10:1)以提供作为无色油的1-乙基-3-甲基-吡唑-5-甲酸苄酯(31.4 g, 129 mmol, 99%收率)。LCMS [M + H]+ = 245。A mixture of 1-ethyl-3-methyl- 1H -pyrazole-5-carboxylic acid (20 g, 130 mmol), (bromomethyl)benzene (22.2 g, 130 mmol), K2CO3 (26.9 g, 195 mmol) and DMF (200 mL ) was stirred at 60°C overnight. The mixture was then dissolved in DCM, washed with water, dried over anhydrous Na2SO4 , filtered, concentrated under reduced pressure, and purified by column chromatography on silica gel (petroleum ether/EtOAc = 10:1) to provide 1-ethyl-3-methyl-pyrazole-5-carboxylic acid benzyl ester (31.4 g, 129 mmol, 99% yield) as a colorless oil. LCMS [M+H] + = 245.
步骤4: 1-乙基-4-碘代-3-甲基-1H-吡唑-5-甲酸苄酯Step 4: 1-ethyl-4-iodo-3-methyl- 1H -pyrazole-5-carboxylic acid benzyl ester
在90℃搅拌1-乙基-3-甲基-1H-吡唑-5-甲酸苄酯 (31.6 g, 129 mmol), 1-碘代吡咯烷-2,5-二酮(34.9 g, 155 mmol)和DMF (400 mL)的混合物持续2天。然后,将该混合物冷却至室温,溶解在DCM中,并用饱和硫代硫酸钠水溶液洗涤。经无水Na2SO4干燥有机层,过滤,在减压下浓缩,并通过柱层析法纯化(石油醚/EtOAc = 10:1)以提供1-乙基-4-碘代-3-甲基-1H-吡唑-5-甲酸苄酯 (42.6 g, 115 mmol, 89%收率)。LCMS [M + H]+ = 371。A mixture of 1-ethyl-3-methyl- 1H -pyrazole-5-carboxylic acid benzyl ester (31.6 g, 129 mmol), 1-iodopyrrolidine-2,5-dione (34.9 g, 155 mmol) and DMF (400 mL) was stirred at 90 ° C for 2 days. Then, the mixture was cooled to room temperature, dissolved in DCM, and washed with saturated sodium thiosulfate aqueous solution. The organic layer was dried over anhydrous Na2SO4 , filtered, concentrated under reduced pressure, and purified by column chromatography (petroleum ether/EtOAc = 10: 1) to provide 1-ethyl-4-iodo-3-methyl- 1H -pyrazole-5-carboxylic acid benzyl ester (42.6 g, 115 mmol, 89% yield). LCMS [M + H] + = 371.
步骤5: 4-(5-(5-(乙氧基羰基)-3-甲基-1H-吡唑-1-基)戊-1-炔-1-基)-1-乙基-3-甲基-1H-吡唑-5-甲酸苄酯Step 5: Benzyl 4-(5-(5-(ethoxycarbonyl)-3-methyl- 1H -pyrazol-1-yl)pent-1-yn-1-yl)-1-ethyl-3-methyl- 1H -pyrazole-5-carboxylate
在60℃在氮气气氛下搅拌3-甲基-1-(戊-4-炔-1-基)-1H-吡唑-5-甲酸乙酯(10.0 g, 45.4 mmol)、1-乙基-4-碘代-3-甲基-1H-吡唑-5-甲酸苄酯 (16.8 g, 45.4mmol)、碘化亚铜(I)(0.864 g, 4.54 mmol)、双(三苯基膦)氯化钯(II)(0.319 g, 0.454mmol)和Et3N (200 mL)的混合物过夜。然后,将该混合物溶解在DCM中并用水洗涤。经无水Na2SO4干燥有机相,过滤,在减压下浓缩,并通过柱层析法在硅胶上纯化(石油醚/EtOAc =5:1)以提供作为黄色固体的4-(5-(5-(乙氧基羰基)-3-甲基-1H-吡唑-1-基)戊-1-炔-1-基)-1-乙基-3-甲基-1H-吡唑-5-甲酸苄酯 (9.5 g, 20.5 mmol, 45.3%收率)。LCMS [M +H]+ = 463。A mixture of ethyl 3-methyl-1-(pent-4-yn-1-yl) -1H -pyrazole-5-carboxylate (10.0 g, 45.4 mmol), benzyl 1-ethyl-4-iodo-3-methyl- 1H -pyrazole-5-carboxylate (16.8 g, 45.4 mmol), copper (I) iodide (0.864 g, 4.54 mmol), bis (triphenylphosphine) palladium (II) chloride (0.319 g, 0.454 mmol) and Et3N (200 mL) was stirred under nitrogen atmosphere overnight at 60° C. The mixture was then dissolved in DCM and washed with water. The organic phase was dried over anhydrous Na2SO4 , filtered, concentrated under reduced pressure, and purified by column chromatography on silica gel (petroleum ether/EtOAc = 5:1) to provide benzyl 4-(5-(5-(ethoxycarbonyl)-3-methyl- 1H -pyrazol-1-yl)pent-1-yn-1-yl)-1-ethyl-3-methyl- 1H -pyrazole-5-carboxylate (9.5 g, 20.5 mmol, 45.3% yield) as a yellow solid. LCMS [M+H] + = 463.
步骤6: 4-(5-(5-(乙氧基羰基)-3-甲基-1H-吡唑-1-基)戊基)-1-乙基-3-甲基-1H-吡唑-5-甲酸Step 6: 4-(5-(5-(Ethoxycarbonyl)-3-methyl- 1H -pyrazol-1-yl)pentyl)-1-ethyl-3-methyl- 1H -pyrazole-5-carboxylic acid
在室温在氢气气氛(4 atm)下搅拌4-(5-(5-(乙氧基羰基)-3-甲基-1H-吡唑-1-基)戊-1-炔-1-基)-1-乙基-3-甲基-1H-吡唑-5-甲酸苄酯 (19.0 g, 41.10 mmol)、10%Pd/C (0.22 g, 2.05 mmol)和THF (500 mL)的混合物2天。然后,过滤该反应混合物并在减压下浓缩。从EtOAc/石油醚 (1:5, v/v)中重结晶所获得的残余物以提供4-(5-(5-(乙氧基羰基)-3-甲基-吡唑-1-基)戊基)-1-乙基-3-甲基-吡唑-5-甲酸 (10.5 g, 27.90 mmol,67.9%收率)。1H-NMR (400 MHz, CDCl3) δ ppm 6.63 (s, 1 H), 4.57-4.48 (m, 4 H),4.38-4.32 (m, 2 H), 2.74-2.62 (m, 2 H), 2.32 (s, 3 H), 2.23 (s, 3 H), 1.91-1.86 (m, 2 H), 1.59-1.54 (m, 2 H), 1.45-1.37 (m, 8 H)。LCMS [M + H]+ = 377。A mixture of 4-(5-(5-(ethoxycarbonyl)-3-methyl- 1H -pyrazol-1-yl)pent-1-yn-1-yl)-1-ethyl-3-methyl- 1H -pyrazole-5-carboxylic acid benzyl ester (19.0 g, 41.10 mmol), 10%Pd/C (0.22 g, 2.05 mmol) and THF (500 mL) was stirred at room temperature under a hydrogen atmosphere (4 atm) for 2 days. Then, the reaction mixture was filtered and concentrated under reduced pressure. The obtained residue was recrystallized from EtOAc/petroleum ether (1:5, v/v) to provide 4-(5-(5-(ethoxycarbonyl)-3-methyl-pyrazol-1-yl)pentyl)-1-ethyl-3-methyl-pyrazole-5-carboxylic acid (10.5 g, 27.90 mmol, 67.9% yield). 1 H-NMR (400 MHz, CDCl 3 ) δ ppm 6.63 (s, 1 H), 4.57-4.48 (m, 4 H), 4.38-4.32 (m, 2 H), 2.74-2.62 (m, 2 H), 2.32 (s, 3 H), 2.23 (s, 3 H), 1.91-1.8 6 (m, 2 H), 1.59-1.54 (m, 2 H), 1.45-1.37 (m, 8 H). LCMS [M + H] + = 377.
步骤7: 4-4-(7-(5-羧基-3-甲基-1H-吡唑-1-基)庚基)-1-乙基-3-甲基-1H-吡唑-5-甲酸Step 7: 4-4-(7-(5-Carboxy-3-methyl- 1H -pyrazol-1-yl)heptyl)-1-ethyl-3-methyl- 1H -pyrazole-5-carboxylic acid
向在室温搅拌的4-(5-(5-(乙氧基羰基)-3-甲基-1H-吡唑-1-基)戊基)-1-乙基-3-甲基-1H-吡唑-5-甲酸 (9.0 g, 23.9 mmol)在MeOH (120 mL)和水(120 mL)中的悬浮液加入2 M NaOH水溶液(60 mL, 119.5 mmol)。在室温搅拌反应混合物30 min。然后,通过加入6 M HCl溶液将该混合物酸化至pH 4,之后从该反应混合物中沉淀出固体。通过过滤收集固体并在减压下干燥以提供作为白色固体的4-(5-(5-羧基-3-甲基-1H-吡唑-1-基)戊基)-1-乙基-3-甲基-1H-吡唑-5-甲酸 (6.5 g, 18.7 mmol, 78.1%收率)。1H-NMR (400 MHz,DMSO-d 6) δ ppm 6.57 (s, 1 H), 4.40-4.34 (m, 4 H), 2.53 (t, J = 7.6 Hz, 2 H),2.16 (s, 3 H), 2.09 (s, 3 H), 1.74-1.67 (m, 2 H), 1.44-1.37 (m, 2 H), 1.27-1.16 (m, 5 H)。LCMS [M + H]+ = 349。2 M NaOH aqueous solution (60 mL, 119.5 mmol) was added to a suspension of 4-(5-(5-(ethoxycarbonyl)-3-methyl- 1H -pyrazol-1-yl)pentyl)-1-ethyl-3-methyl- 1H -pyrazole-5-carboxylic acid (9.0 g, 23.9 mmol) in MeOH (120 mL) and water (120 mL) stirred at room temperature. The reaction mixture was stirred at room temperature for 30 min. Then, the mixture was acidified to pH 4 by adding 6 M HCl solution, and a solid was precipitated from the reaction mixture afterwards. The solid was collected by filtration and dried under reduced pressure to provide 4-(5-(5-carboxyl-3-methyl- 1H -pyrazol-1-yl)pentyl)-1-ethyl-3-methyl- 1H -pyrazole-5-carboxylic acid (6.5 g, 18.7 mmol, 78.1% yield) as a white solid. 1 H-NMR (400 MHz, DMSO- d 6 ) δ ppm 6.57 (s, 1 H), 4.40-4.34 (m, 4 H), 2.53 (t, J = 7.6 Hz, 2 H), 2.16 (s, 3 H), 2.09 (s, 3 H), 1.74-1.67 (m, 2 H), 1 .44-1.37 (m, 2 H), 1.27-1.16 (m, 5 H). LCMS [M + H] + = 349.
步骤8: 1-烯丙基-2-氨基-1H-苯并[d]咪唑-5-甲酰胺, 氢溴酸盐Step 8: 1-Allyl-2-amino-1H-benzo[d]imidazole-5-carboxamide, hydrobromide
在室温向4-氟-3-硝基苯甲酰胺 (10.0 g, 54.3 mmol)在DMF (60 mL)中的溶液逐滴加入烯丙胺(36.6 mL, 489 mmol)并搅拌该混合物5 min。在这段时间后,一次性加入K2CO3 (15.01 g, 109 mmol),并在室温搅拌该混合物30 min。然后,在真空中除去DMF。将残余物悬浮于500 mL水中,过滤掉所得橙色沉淀物,用水洗涤,并在真空中干燥。To a solution of 4-fluoro-3-nitrobenzamide (10.0 g, 54.3 mmol) in DMF (60 mL) was added allylamine (36.6 mL, 489 mmol) dropwise at room temperature and the mixture was stirred for 5 min. After this time, K 2 CO 3 (15.01 g, 109 mmol) was added in one portion and the mixture was stirred at room temperature for 30 min. Then, the DMF was removed in vacuo. The residue was suspended in 500 mL of water and the resulting orange precipitate was filtered off, washed with water and dried in vacuo.
将以上沉淀物溶解于AcOH (600 mL)中并将烧瓶置于20℃水浴中。按小份小心地加入锌(10.65 g, 163 mmol)。通过LCMS监控反应并根据需要按小份加入另外的锌(大致3eq)直到还原完全。在通过LCMS测得反应完全之后,过滤掉固体并在真空中浓缩滤液。将蒸发残余物吸纳在DCM (500 mL)和EtOH (150 mL)中并用15% K2CO3水溶液(100 mL)洗涤。分离有机层,经Na2SO4干燥,过滤,并在真空中浓缩 。The above precipitate was dissolved in AcOH (600 mL) and the flask was placed in a 20°C water bath. Zinc (10.65 g, 163 mmol) was carefully added in small portions. The reaction was monitored by LCMS and additional zinc (approximately 3eq) was added in small portions as needed until the reduction was complete. After the reaction was complete as measured by LCMS, the solid was filtered off and the filtrate was concentrated in vacuo. The evaporation residue was taken up in DCM (500 mL) and EtOH (150 mL) and washed with 15% K 2 CO 3 aqueous solution (100 mL). The organic layer was separated, dried over Na 2 SO 4 , filtered, and concentrated in vacuo.
将以上的蒸发残余物溶解在MeOH (200 mL)中,一次性快速加入在CH3CN中的5.0M 溴化氰 (11.95 mL, 59.7 mmol),并在室温搅拌混合物18 h。在这段时间之后,在真空中浓缩该反应混合物,然后再次溶解在MeOH (200 mL)中。加入甲苯(100 mL)和CH3CN (100mL)的混合物,并在40℃ (0-1 mbar)将所得混合物浓缩至干并在真空中干燥16 h以提供作为深紫色粉末的1-烯丙基-2-氨基-1H-苯并[d]咪唑-5-甲酰胺, 氢溴酸盐 (11.3 g, 38.0mmol, 70.0%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.83 (s, 2 H), 8.07 (br. s.,1 H), 7.88 (d, J=1.00 Hz, 1 H), 7.82 (dd, J=8.41, 1.38 Hz, 1 H), 7.52 (d, J=8.53 Hz, 1 H), 7.43 (br. s., 1 H), 5.87 - 6.02 (m, 1 H), 5.25 (dd, J=10.42,0.88 Hz, 1 H), 5.17 (dd, J=17.32, 1.00 Hz, 1 H), 4.84 (d, J=5.02 Hz, 2 H);LCMS [M + H]+ = 216.9。The above evaporation residue was dissolved in MeOH (200 mL), 5.0M cyanogen bromide (11.95 mL, 59.7 mmol) in CH 3 CN was added quickly in one portion, and the mixture was stirred at room temperature for 18 h. After this time, the reaction mixture was concentrated in vacuo and then dissolved in MeOH (200 mL) again. A mixture of toluene (100 mL) and CH 3 CN (100 mL) was added, and the resulting mixture was concentrated to dryness at 40° C. (0-1 mbar) and dried in vacuo for 16 h to provide 1-allyl-2-amino-1H-benzo[d]imidazole-5-carboxamide, hydrobromide (11.3 g, 38.0 mmol, 70.0% yield) as a dark purple powder. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.83 (s, 2 H), 8.07 (br. s.,1 H), 7.88 (d , J =1.00 Hz, 1 H), 7.82 (dd, J =8.41, 1.38 Hz, 1 H), 7.52 (d, J = 8.53 Hz, 1 H LCMS [M + H ] + = 216.9.
步骤9: 1-烯丙基-2-(1-(5-(5-((1-烯丙基-5-氨基甲酰基-1H-苯并[d]咪唑-2-基)氨基甲酰基)-1-乙基-3-甲基-1H-吡唑-4-基)戊基)-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 9: 1-allyl-2-(1-(5-(5-((1-allyl-5-carbamoyl-1H-benzo[d]imidazol-2-yl)carbamoyl)-1-ethyl-3-methyl-1H-pyrazol-4-yl)pentyl)-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide
向5.0 mL Biotage® 密封管装入4-(5-(5-羧基-3-甲基-1H-吡唑-1-基)戊基)-1-乙基-3-甲基-1H-吡唑-5-甲酸 (634 mg, 1.820 mmol)、1-烯丙基-2-氨基-1H-苯并[d]咪唑-5-甲酰胺, 氢溴酸盐 (1352 mg, 4.55 mmol)、HATU (1730 mg, 4.55 mmol)和NMP (13mL)。在室温搅拌1分钟之后,加入DIPEA (3.17 mL, 18.20 mmol)并在室温搅拌混合物5min,然后在140℃在微波反应器中加热1 h。在这段时间之后,加入5.0 mL水并在室温搅拌该混合物5 min。然后,将其倾倒至250 mL的冰冷水中,并剧烈搅拌1 h。过滤掉所得固体,用水洗涤,使用MeOH/DCM由过滤器溶解,在真空中浓缩,并施以硅胶层析法 (Biotage® UltraSNAP 100 g SiO2柱: 0-40% MeOH/EtOAc)以获得作为粉色固体的1-烯丙基-2-(1-(5-(5-((1-烯丙基-5-氨基甲酰基-1H-苯并[d]咪唑-2-基)氨基甲酰基)-1-乙基-3-甲基-1H-吡唑-4-基)戊基)-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (840 mg,1.128 mmol, 62%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 12.88 (s, 1 H), 12.81 (s,1 H), 7.99 - 8.02 (m, 2 H), 7.97 (br. s., 2 H), 7.77 (ddd, J=8.34, 3.66, 1.39Hz, 2 H), 7.41 (dd, J=16.93, 8.34 Hz, 2 H), 7.34 (br. s., 2 H), 6.65 (s, 1H), 5.87 - 6.02 (m, 2 H), 4.99 - 5.22 (m, 4 H), 4.82 (dd, J=11.62, 4.80 Hz, 4H), 4.50 - 4.61 (m, 4 H), 2.73 (t, J=7.45 Hz, 2 H), 2.15 (s, 3 H), 2.08 (s, 3H), 1.71 - 1.85 (m, 2 H), 1.45 - 1.55 (m, 2 H), 1.27 - 1.34 (m, 5 H); LCMS [M+ H]+ = 745.7。A 5.0 mL Biotage® sealed tube was charged with 4-(5-(5-carboxy-3-methyl-1H-pyrazol-1-yl)pentyl)-1-ethyl-3-methyl-1H-pyrazole-5-carboxylic acid (634 mg, 1.820 mmol), 1-allyl-2-amino-1H-benzo[d]imidazole-5-carboxamide, hydrobromide (1352 mg, 4.55 mmol), HATU (1730 mg, 4.55 mmol) and NMP (13 mL). After stirring at room temperature for 1 minute, DIPEA (3.17 mL, 18.20 mmol) was added and the mixture was stirred at room temperature for 5 min and then heated at 140 °C in a microwave reactor for 1 h. After this time, 5.0 mL of water was added and the mixture was stirred at room temperature for 5 min. It was then poured into 250 mL of ice-cold water and stirred vigorously for 1 h. The resulting solid was filtered off, washed with water, dissolved from the filter using MeOH/DCM, concentrated in vacuo, and subjected to silica gel chromatography ( Biotage® UltraSNAP 100 g SiO column : 0-40% MeOH/EtOAc) to afford 1-allyl-2-(1-(5-(5-((1-allyl-5-carbamoyl-1H-benzo[d]imidazol-2-yl)carbamoyl)-1-ethyl-3-methyl-1H-pyrazol-4-yl)pentyl)-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (840 mg, 1.128 mmol, 62% yield) as a pink solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 12.88 (s, 1 H), 12.81 (s, 1 H), 7.99 - 8.02 (m, 2 H), 7.97 (br. s., 2 H), 7.77 (ddd, J =8.34, 3.66, 1.39Hz, 2 H), 7 , 4.50 - 4.61 (m, 4 H), 2.73 (t, J =7.45 Hz, 2 H), 2.15 (s, 3 H), 2.08 (s, 3H), 1.71 - 1.85 (m, 2 H), 1.45 - 1.55 (m, 2 H), 1.27 - 1.34 (m, 5 H); LCMS [M + H] + = 745.7.
步骤10: 8-乙基-10,18-二甲基-7,20-二氧代-6,7,8,11,12,13,14,15,20,21,28,31-十二氢苯并[4,5]咪唑并[1,2-a]苯并[4,5]咪唑并[2,1-p]二吡唑并[5,1-e:4',3'-1][1,3,6,15,17]五氮杂环二十一碳十烯-3,24-二甲酰胺Step 10: 8-ethyl-10,18-dimethyl-7,20-dioxo-6,7,8,11,12,13,14,15,20,21,28,31-dodecahydrobenzo[4,5]imidazo[1,2-a]benzo[4,5]imidazo[2,1-p]dipyrazolo[5,1-e:4',3'-1][1,3,6,15,17]pentaazacyclohexene-3,24-dicarboxamide
向四个20 mL Biotage® 微波密封管中装入总共1-烯丙基-2-(1-(5-(5-((1-烯丙基-5-氨基甲酰基-1H-苯并[d]咪唑-2-基)氨基甲酰基)-1-乙基-3-甲基-1H-吡唑-4-基)戊基)-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (160 mg, 0.215 mmol)、Hoveyda-Grubbs II催化剂(26.9 mg, 0.043 mmol)和新鲜脱气的1,2-二氯乙烷(DCE) (80mL)。在100℃在微波反应器中加热密封管4 h。在混合物冷却至室温之后,将MeOH (1.0 mL)加入各管并在室温搅拌所得透明溶液 5 min。将2-异氰酸酸根合乙酸钾溶液(15 mg在1.5mL MeOH中)加入各管并在室温搅拌所得混合物 5 min。合并各个管,在真空中浓缩,然后将蒸发残余物吸纳在最小体积的DCM/MeOH中,并通过硅胶层析法纯化 (Biotage® UltraSNAP 100 g SiO2柱; 0-40% MeOH/EtOAc)以获得作为浅绿色固体的含烯烃异构体混合物的所需产物(61 mg)。将该产物进一步纯化(Biotage® Ultra SNAP 25 g SiO2柱; 0-20%MeOH/DCM梯度)以获得作为7:1 反式:顺式混合物的8-乙基-10,18-二甲基-7,20-二氧代-6,7,8,11,12,13,14,15,20,21,28,31-十二氢苯并[4,5]咪唑并[1,2-a]苯并[4,5]咪唑并[2,1-p]二吡唑并[5,1-e:4',3'-1][1,3,6,15,17]五氮杂环二十一碳十烯-3,24-二甲酰胺(54 mg, 0.075 mmol, 35%收率)。反式异构体的表征: 1H NMR (400 MHz, DMSO-d 6) δ ppm12.87 (s, 1 H), 12.84 (s, 1 H), 7.98 (br. s., 4 H), 7.77 (dd, J=7.71, 3.16Hz, 2 H), 7.33 - 7.48 (m, 4 H), 6.55 (s, 1 H), 5.89 - 5.98 (m, 1 H), 5.66 -5.75 (m, 1 H), 4.90 (d, J=7.83 Hz, 4 H), 4.73 (t, J=6.95 Hz, 2 H), 4.47 (q, J=6.99 Hz, 2 H), 2.72 - 2.80 (m, 2 H), 2.17 (s, 3 H), 2.10 (s, 3 H), 1.72 (br.s., 2 H), 1.44 (br. s., 2 H), 1.30 (t, J=7.07 Hz, 5 H); LCMS [M + H]+ =717.6。Four 20 mL Biotage® microwave sealed tubes were charged with a total of 1-allyl-2-(1-(5-(5-((1-allyl-5-carbamoyl-1H-benzo[d]imidazol-2-yl)carbamoyl)-1-ethyl-3-methyl-1H-pyrazol-4-yl)pentyl)-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (160 mg, 0.215 mmol), Hoveyda-Grubbs II catalyst (26.9 mg, 0.043 mmol) and freshly degassed 1,2-dichloroethane (DCE) (80 mL). The sealed tubes were heated at 100 °C in a microwave reactor for 4 h. After the mixture was cooled to room temperature, MeOH (1.0 mL) was added to each tube and the resulting clear solution was stirred at room temperature for 5 min. Potassium 2-isocyanatoacetate solution (15 mg in 1.5 mL MeOH) was added to each tube and the resulting mixture was stirred at room temperature for 5 min. The tubes were combined, concentrated in vacuo, and the evaporation residue was taken up in a minimum volume of DCM/MeOH and purified by silica gel chromatography ( Biotage® UltraSNAP 100 g SiO2 column; 0-40% MeOH/EtOAc) to obtain the desired product (61 mg) as a light green solid containing a mixture of olefin isomers. The product was further purified ( Biotage® Ultra SNAP 25 g SiO2 column ; 0-20% MeOH/DCM gradient) to afford 8-ethyl-10,18-dimethyl-7,20-dioxo-6,7,8,11,12,13,14,15,20,21,28,31-dodecahydrobenzo[4,5]imidazo[1,2-a]benzo[4,5]imidazo[2,1-p]dipyrazolo[5,1-e:4',3'-1][1,3,6,15,17]pentaazacyclohexenedecene-3,24-dicarboxamide (54 mg, 0.075 mmol, 35% yield) as a 7:1 trans:cis mixture. Characterization of trans isomers: 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm12.87 (s, 1 H), 12.84 (s, 1 H), 7.98 (br. s., 4 H), 7.77 (dd, J =7.71, 3.16Hz, 2 H), 7.33 - 7.48 (m, 4 H), 6.55 (s, 1 H), 5.89 - 5.98 (m, 1 H), 5.66 -5.75 (m, 1 H), 4.90 (d, J =7.83 Hz, 4 H), 4.73 (t, J =6.95 Hz, 2 H), 4.47 (q, J =6.99 Hz, 2 H), 2.72 - 2.80 (m, 2 LCMS [M + H ] + =717.6.
步骤11: 8-乙基-10,18-二甲基-7,20-二氧代-6,7,8,11,12,13,14,15,20,21,28,29,30,31-十四氢苯并[4,5]咪唑并[1,2-a]苯并[4,5]咪唑并[2,1-p]二吡唑并[5,1-e:4',3'-1][1,3,6,15,17]五氮杂环二十一碳十烯-3,24-二甲酰胺Step 11: 8-ethyl-10,18-dimethyl-7,20-dioxo-6,7,8,11,12,13,14,15,20,21,28,29,30,31-tetradecahydrobenzo[4,5]imidazo[1,2-a]benzo[4,5]imidazo[2,1-p]dipyrazolo[5,1-e:4',3'-1][1,3,6,15,17]pentaazacyclohexenedecene-3,24-dicarboxamide
向圆底烧瓶装入10% Pd/C (200 mg, 0.188 mmol)并用氮气吹扫。加入8-乙基-10,18-二甲基-7,20-二氧代-6,7,8,11,12,13,14,15,20,21,28,31-十二氢苯并[4,5]咪唑并[1,2-a]苯并[4,5]咪唑并[2,1-p]二吡唑并[5,1-e:4',3'-1][1,3,6,15,17]五氮杂环二十一碳十烯-3,24-二甲酰胺 (100 mg, 0.140 mmol, 7:1 反式:顺式混合物)在MeOH(20.0 mL)和THF (20.0 mL)的混合物中的溶液,用氢气吹扫该烧瓶,并在氢气气氛(1 atm)下搅拌该反应混合物23 h。然后,该烧瓶向空气敞开,剧烈搅拌15 min并过滤,用 MeOH/THF洗涤Pd/C,在真空中浓缩滤液,并施以硅胶层析法 (Biotage® Ultra SNAP 25 g SiO2柱;0-20% MeOH/DCM)以获得作为浅粉色固体的8-乙基-10,18-二甲基-7,20-二氧代-6,7,8,11,12,13,14,15,20,21,28,29,30,31-十四氢苯并[4,5]咪唑并[1,2-a]苯并[4,5]咪唑并[2,1-p]二吡唑并[5,1-e:4',3'-1][1,3,6,15,17]五氮杂环二十一碳十烯-3,24-二甲酰胺(56 mg, 0.078 mmol, 55.8%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 12.88 (br. s.,2 H), 8.02 (s, 4 H), 7.79 - 7.87 (m, 2 H), 7.67 (d, J=8.34 Hz, 1 H), 7.63 (d,J=8.34 Hz, 1 H), 7.37 (br. s., 2 H), 6.57 (s, 1 H), 4.74 (t, J=6.57 Hz, 2 H),4.48 (q, J=6.99 Hz, 2 H), 4.19 - 4.31 (m, 4 H), 2.78 - 2.86 (m, 2 H), 2.16(s, 3 H), 2.08 (s, 3 H), 1.91 (br. s., 4 H), 1.77 - 1.86 (m, 2 H), 1.44 -1.54 (m, 2 H), 1.35 - 1.42 (m, 2 H), 1.29 (t, J=7.07 Hz, 3 H); LCMS (m/z):719.7 [M + H]+。A round bottom flask was charged with 10% Pd/C (200 mg, 0.188 mmol) and purged with nitrogen. A solution of 8-ethyl-10,18-dimethyl-7,20-dioxo-6,7,8,11,12,13,14,15,20,21,28,31-dodecahydrobenzo[4,5]imidazo[1,2-a]benzo[4,5]imidazo[2,1-p]dipyrazolo[5,1-e:4',3'-1][1,3,6,15,17]pentaazacyclohexene-3,24-dicarboxamide (100 mg, 0.140 mmol, 7:1 trans:cis mixture) in a mixture of MeOH (20.0 mL) and THF (20.0 mL) was added, the flask was purged with hydrogen, and the reaction mixture was stirred under hydrogen atmosphere (1 atm) for 23 h. The flask was then opened to air, stirred vigorously for 15 min and filtered, the Pd/C was washed with MeOH/THF, the filtrate was concentrated in vacuo and subjected to silica gel chromatography ( Biotage® Ultra SNAP 25 g SiO2 column; 0-20% MeOH/DCM) to afford 8-ethyl-10,18-dimethyl-7,20-dioxo-6,7,8,11,12,13,14,15,20,21,28,29,30,31-tetradecahydrobenzo[4,5]imidazo[1,2-a]benzo[4,5]imidazo[2,1-p]dipyrazolo[5,1-e:4',3'-1][1,3,6,15,17]pentaazacyclohexenedecene-3,24-dicarboxamide (56 mg, 0.078 mmol, 4% by weight) as a light pink solid. 55.8% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 12.88 (br. s.,2 H), 8.02 (s, 4 H), 7.79 - 7.87 (m, 2 H), 7.67 (d, J =8.34 Hz, 1 H), 7.63 (d, J =8.34 Hz, 1 H), 7.37 ( br. s., 2 H), 6.57 (s, 1 H), 4.74 (t, J =6.57 Hz, 2 H), 4.48 (q, J =6.99 Hz, 2 H), 4.19 - 4.31 (m, 4 H), 2.78 - 2.86 (m, 2 H), 2.16 (s, 3 H), 2.08 ( s, 3 H), 1.91 (br. s., 4 H), 1.77 - 1.86 (m, 2 H), 1.44 -1.54 (m, 2 H), 1.35 - 1.42 (m, 2 H), 1.29 (t, J =7.07 Hz, 3 H); LCMS (m/z): 719.7 [M + H] + .
步骤12: (5aE,21E)-8-乙基-5,10,18,22-四甲基-7,20-二氧代-5,7,8,11,12,13,14,15,20,22,28,29,30,31-十四氢苯并[4,5]咪唑并[1,2-a]苯并[4,5]咪唑并[2,1-p]二吡唑并[5,1-e:4',3'-l][1,3,6,15,17]五氮杂环二十一碳十烯-3,24-二甲酰胺Step 12: (5aE,21E)-8-ethyl-5,10,18,22-tetramethyl-7,20-dioxo-5,7,8,11,12,13,14,15,20,22,28,29,30,31-tetradecahydrobenzo[4,5]imidazo[1,2-a]benzo[4,5]imidazo[2,1-p]dipyrazolo[5,1-e:4',3'-l][1,3,6,15,17]pentaazacyclohexene-3,24-dicarboxamide
向8-乙基-10,18-二甲基-7,20-二氧代-6,7,8,11,12,13,14,15,20,21,28,29,30,31-十四氢苯并[4,5]咪唑并[1,2-a]苯并[4,5]咪唑并[2,1-p]二吡唑并[5,1-e:4',3'-l][1,3,6,15,17]五氮杂环二十一碳十烯-3,24-二甲酰胺 (85 mg, 0.118 mmol)在DMF (3mL)中的溶液加入碘甲烷(0.015 mL 碘甲烷, 0.236 mmol, 83 uL原液,其为180 uL碘甲烷在820 uL DMF中的原液)。在室温搅拌该反应3 h。将该反应干装载至硅胶上并通过硅胶层析法(ISCO-Rf, 12g柱, 0%-30%梯度的MeOH/DCM ) 纯化以提供浅黄色固体。将该物质悬浮在MeCN中并经周末在空气流下浓缩至干以提供作为白色固体的(5aE,21E)-8-乙基-5,10,18,22-四甲基-7,20-二氧代-5,7,8,11,12,13,14,15,20,22,28,29,30,31-十四氢苯并[4,5]咪唑并[1,2-a]苯并[4,5]咪唑并[2,1-p]二吡唑并[5,1-e:4',3'-l][1,3,6,15,17]五氮杂环二十一碳十烯-3,24-二甲酰胺 (58 mg, 0.077 mmol, 65.0%收率)。1H NMR (400MHz, 甲醇-d 4) δ ppm 8.08 (dd, J=6.97, 1.39 Hz, 2 H), 7.87 - 7.99 (m, 2 H),7.63 (d, J=8.62 Hz, 1 H), 7.51 (d, J=8.62 Hz, 1 H), 6.53 (s, 1 H), 4.63 (t, J=6.97 Hz, 2 H), 4.52 (q, J=7.18 Hz, 2 H), 4.20 - 4.32 (m, 4 H), 3.61 (s, 6H), 2.73 - 2.84 (m, 2 H),2.26 (s, 3 H), 2.21 (s, 3 H), 1.87 (br. s., 6 H),1.53 - 1.63 (m, 2 H), 1.30 - 1.40 (m, 5 H)。LCMS m/z = 747 [M + H]+。To a solution of 8-ethyl-10,18-dimethyl-7,20-dioxo-6,7,8,11,12,13,14,15,20,21,28,29,30,31-tetradecahydrobenzo[4,5]imidazo[1,2-a]benzo[4,5]imidazo[2,1-p]dipyrazolo[5,1-e:4',3'-1][1,3,6,15,17]pentaazacyclohexene-3,24-dicarboxamide (85 mg, 0.118 mmol) in DMF (3 mL) was added iodomethane (0.015 mL iodomethane, 0.236 mmol, 83 uL stock solution of 180 uL iodomethane in 820 uL DMF). The reaction was stirred at room temperature for 3 h. The reaction was dry loaded onto silica gel and purified by silica gel chromatography (ISCO-Rf, 12 g column, 0%-30% gradient of MeOH/DCM) to afford a light yellow solid. The material was suspended in MeCN and concentrated to dryness under a stream of air over the weekend to afford (5aE,21E)-8-ethyl-5,10,18,22-tetramethyl-7,20-dioxo-5,7,8,11,12,13,14,15,20,22,28,29,30,31-tetrahydrobenzo[4,5]imidazo[1,2-a]benzo[4,5]imidazo[2,1-p]dipyrazolo[5,1-e:4′,3′-1][1,3,6,15,17]pentaazacyclohexeneicosadecadecene-3,24-dicarboxamide (58 mg, 0.077 mmol, 65.0% yield) as a white solid. 1 H NMR (400MHz, methanol- d 4 ) δ ppm 8.08 (dd, J =6.97, 1.39 Hz, 2 H), 7.87 - 7.99 (m, 2 H), 7.63 (d, J =8.62 Hz, 1 H), 7.51 (d, J =8.62 Hz, 1 H), 6.53 (s, 1 H), 4.63 (t, J =6.97 Hz, 2 H), 4.52 (q, J =7.18 Hz, 2 H), 4.20 - 4.32 (m, 4 H), 3.61 (s, 6H), 2.73 - 2.84 (m, 2 H), 2.26 (s, 3 H), 2.21 (s, 3 H), 1.87 (br. s., 6 H), 1.53 - 1.63 (m, 2 H), 1.30 - 1.40 (m, 5 H). LCMS m/z = 747 [M + H] + .
实施例4Example 4
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐(E)-1-((E)-4-((E)-5-Carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 2-hydrochloride
步骤1: (E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 1: (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (78 mg, 0.068 mmol)在DMF (3 mL)中的悬浮液加入碳酸铯 (66.1 mg, 0.203 mmol)和2.5 eq碘甲烷(50uL原液,由在780 uL DMF中的220 uL碘甲烷制成)。在室温搅拌该反应4h。在黄色反应混合物中形成白色沉淀物。加入另外的碘甲烷 (2.5 eq),并且反应立即失去其黄色。在室温搅拌该反应过周末。需要另外的碳酸铯 (66 mg, 0.20 mmol)和碘甲烷溶液(2.5 eq)驱使反应完全。加入水并采用DCM (3x)然后采用~15% EtOH/DCM (2x)萃取水层。采用盐水洗涤合并的有机萃取物,经硫酸钠干燥并浓缩至干。将所得残余物干装载至12 g硅胶柱上并采用0-20%梯度的MeOH/DCM洗脱以提供作为白色固体的(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (20 mg, 0.022 mmol, 33.1%收率)。LCMS m/z = 893 [M + H]+。To a suspension of (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (78 mg, 0.068 mmol) in DMF (3 mL) was added cesium carbonate (66.1 mg, 0.203 mmol) and 2.5 eq iodomethane (50 uL stock solution made from 220 uL iodomethane in 780 uL DMF). The reaction was stirred at room temperature for 4 h. A white precipitate formed in the yellow reaction mixture. Add other iodomethane (2.5 eq), and the reaction loses its yellow color immediately. Stir the reaction at room temperature over the weekend. Need other cesium carbonate (66 mg, 0.20 mmol) and iodomethane solution (2.5 eq) to drive the reaction to be complete. Add water and adopt DCM (3x) then adopt ~15% EtOH/DCM (2x) aqueous layer extraction. Adopt salt water washing to merge organic extract, dried over sodium sulfate and be concentrated to dryness. The resulting residue was dry loaded onto a 12 g silica gel column and eluted with a 0-20% gradient of MeOH/DCM to afford (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (20 mg, 0.022 mmol, 33.1% yield) as a white solid. LCMS m/z = 893 [M + H] + .
步骤2: (E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐Step 2: (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 2-hydrochloride
向(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (20 mg, 0.022 mmol)在1,4-二氧杂环己烷 (0.5 mL)中的溶液加入在二氧杂环己烷中的4 M HCl (0.011 mL, 0.045 mmol)。按需要加入另外的在二氧杂环己烷中的4 M HCl以驱使脱保护。当反应完全时,过滤该反应并用二氧杂环己烷洗涤滤饼,并在45℃的真空炉中干燥过夜。所得浅黄色固体被认为是(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐 (17 mg, 0.020 mmol, 87%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.01 - 8.13 (m, 3 H) 7.78 - 7.84 (m, 1H) 7.75 (s, 1 H) 7.35 - 7.52 (m, 4 H) 6.42 (s, 2 H), 5.88 - 5.97 (m, 1 H),5.60 - 5.68 (m, 1 H), 4.88 (d, J=5.07 Hz, 2 H) 4.76 (d, J=5.58 Hz, 2 H) 4.36- 4.48 (m, 4 H) 4.11 (t, J=6.34 Hz, 2 H) 3.56 (s, 3 H) 3.53 (s, 3 H) 3.46 (s,2 H) 2.12 (s, 6 H) 1.75 (d, J=6.08 Hz, 2 H) 1.21 (t, J=7.10 Hz, 6 H)。LCMS m/z= 779 [M + H]+。To a solution of (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (20 mg, 0.022 mmol) in 1,4-dioxane (0.5 mL) was added 4 M HCl in dioxane (0.011 mL, 0.045 mmol). Additional 4 M HCl in dioxane was added as needed to drive deprotection. When the reaction was complete, the reaction was filtered and the filter cake washed with dioxane and dried in a vacuum oven at 45°C overnight. The resulting light yellow solid was believed to be (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 2 hydrochloride (17 mg, 0.020 mmol, 87% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.01 - 8.13 (m, 3 H) 7.78 - 7.84 (m, 1H) 7.75 (s, 1 H) 7.35 - 7.52 (m, 4 H) 6.42 (s, 2 H), 5.88 - 5.97 (m, 1 H),5 .60 - 5.68 (m, 1 H), 4.88 (d, J =5.07 Hz, 2 H) 4.76 (d, J =5.58 Hz, 2 H) 4.36- 4.48 (m, 4 H) 4.11 (t, J =6.34 Hz, 2 H) 3.56 (s, 3 H) 3.53 (s, 3 H) 3.46 (s, 2 H) 2.12 (s, 6 H) 1.75 (d, J =6.08 Hz, 2 H) 1.21 (t, J =7.10 Hz, 6 H). LCMS m/z = 779 [M + H] + .
实施例5Example 5
(E)-1-((E)-4-((Z)-5-氨基甲酰基-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,2盐酸盐(E)-1-((E)-4-((Z)-5-Carbamoyl-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 2 hydrochloride
步骤1: (E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-((E)-4-((Z)-5-氨基甲酰基-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 1: (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-((E)-4-((Z)-5-carbamoyl-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (50 mg, 0.058 mmol)在DMF (2 mL)中的悬浮液加入碳酸铯 (75 mg, 0.231 mmol)和碘乙烷(27.0 mg, 0.173mmol)。在3 h后,加入另外的碘乙烷(15uL)并搅拌该反应15 min。在DCM和水之间分配该反应。用DCM/EtOH (3x)萃取水层。用盐水洗涤合并的有机物,经硫酸钠干燥,干装载至硅胶上并通过硅胶层析法 (12 g柱, 0-20% MeOH/DCM梯度) 纯化以提供(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-((E)-4-((Z)-5-氨基甲酰基-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (18mg, 0.020 mmol, 33.8%收率)。LCMS m/z = 921 [M+H]+。To a suspension of (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (50 mg, 0.058 mmol) in DMF (2 mL) was added cesium carbonate (75 mg, 0.231 mmol) and iodoethane (27.0 mg, 0.173 mmol). After 3 h, additional iodoethane (15 uL) was added and the reaction was stirred for 15 min. The reaction was partitioned between DCM and water. The aqueous layer was extracted with DCM/EtOH (3x). The combined organics were washed with brine, dried over sodium sulfate, dry loaded onto silica gel and purified by silica gel chromatography (12 g column, 0-20% MeOH/DCM gradient) to afford (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-((E)-4-((Z)-5-carbamoyl-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (18 mg, 0.020 mmol, 33.8% yield). LCMS m/z = 921 [M+H] + .
步骤2: (E)-1-((E)-4-((Z)-5-氨基甲酰基-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐Step 2: (E)-1-((E)-4-((Z)-5-carbamoyl-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 2-hydrochloride
向(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-((E)-4-((Z)-5-氨基甲酰基-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (17 mg, 0.018 mmol)在1,4-二氧杂环己烷 (923 µL)中的溶液加入在二氧杂环己烷中的HCl (27.7 µL, 0.111 mmol)。在室温1h后,过滤该反应并用二乙醚洗涤滤饼并在55℃的真空炉中干燥过夜以提供作为白色固体的(E)-1-((E)-4-((Z)-5-氨基甲酰基-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐 (14 mg, 0.015 mmol,81%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.03 - 8.14 (m, 3 H), 7.68 - 7.87 (m,2 H), 7.42 - 7.52 (m, 4 H), 6.38 (s, 2 H), 5.80 - 5.97 (m, 1 H), 5.41 - 5.68(m, 1 H), 4.81 - 4.94 (m, 2 H), 4.67 - 4.78 (m, 2 H), 4.34 - 4.45 (m, 4 H),4.01 - 4.22 (m,7 H), 3.32 - 3.51 (m, 2 H), 2.12 (s, 3 H), 2.10 (s, 3 H),1.68- 1.76 (m, 2 H), 1.11 - 1.32 (m, 12 H)。LCMS m/z = 807 [M+H]+。To a solution of (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-((E)-4-((Z)-5-carbamoyl-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (17 mg, 0.018 mmol) in 1,4-dioxane (923 µL) was added HCl in dioxane (27.7 µL, 0.111 mmol). After 1 h at room temperature, the reaction was filtered and the filter cake was washed with diethyl ether and dried in a vacuum oven at 55 °C overnight to provide (E)-1-((E)-4-((Z)-5-carbamoyl-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 2 hydrochloride (14 mg, 0.015 mmol, 81% yield) as a white solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.03 - 8.14 (m, 3 H), 7.68 - 7.87 (m, 2 H), 7.42 - 7.52 (m, 4 H), 6.38 (s, 2 H), 5.80 - 5.97 (m, 1 H), 5.41 - 5.68 (m, 1 H), 4.81 - 4.94 (m, 2 H), 4.67 - 4.78 (m, 2 H), 4.34 - 4.45 (m, 4 H), 4.01 - 4.22 (m, 7 H), 3.32 - 3.51 (m, 2 H), 2.12 (s, 3 H), 2.10 (s, 3 H),1.68- 1.76 (m, 2 H), 1.11 - 1.32 (m, 12 H). LCMS m/z = 807 [M+H] + .
通过以上方法制备的化合物可以互变异构体/同分异构体形式存在,例如(E)-1-((E)-4-((E)-5-氨基甲酰基-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-3-乙基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐The compounds prepared by the above methods may exist as tautomers/isomers, for example (E)-1-((E)-4-((E)-5-carbamoyl-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-3-ethyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 2 hydrochloride
。 .
实施例6Example 6
(2E,2'E)-1,1'-(2,2,3,3-四氟丁烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺), 三氟乙酸盐(2E,2'E)-1,1'-(2,2,3,3-tetrafluorobutane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide), trifluoroacetate
步骤1: 4,4'-((2,2,3,3-四氟丁烷-1,4-二基)双(氮烷二基))双(3-硝基苯甲酰胺)Step 1: 4,4'-((2,2,3,3-tetrafluorobutane-1,4-diyl)bis(azanediyl))bis(3-nitrobenzamide)
在室温经5 min向在DMF (50 mL)中的2,2,3,3-四氟丁烷-1,4-二胺 (1.25 g,7.81 mmol)和碳酸钾(3.24 g, 23.4 mmol)加入4-氟-3-硝基苯甲酰胺 (3.59 g, 19.5mmol),并搅拌反应过夜。用水淬灭混合物,并通过过滤收集所得固体并用MeOH研磨以提供作为黄色固体的标题化合物(600 mg, 1.23 mmol, 16%收率)。LCMS [M+H]+ = 489。4-Fluoro-3-nitrobenzamide (3.59 g, 19.5 mmol) was added to 2,2,3,3-tetrafluorobutane-1,4-diamine (1.25 g, 7.81 mmol) and potassium carbonate (3.24 g, 23.4 mmol) in DMF (50 mL) at room temperature over 5 min, and the reaction was stirred overnight. The mixture was quenched with water, and the resulting solid was collected by filtration and ground with MeOH to provide the title compound (600 mg, 1.23 mmol, 16% yield) as a yellow solid. LCMS [M+H] + = 489.
步骤2: 4,4'-((2,2,3,3-四氟丁烷-1,4-二基)双(氮烷二基))双(3-氨基苯甲酰胺)Step 2: 4,4'-((2,2,3,3-tetrafluorobutane-1,4-diyl)bis(azanediyl))bis(3-aminobenzamide)
在H2下在30℃将在MeOH (100 mL)中的4,4'-((2,2,3,3-四氟丁烷-1,4-二基)双(氮烷二基))双(3-硝基苯甲酰胺) (1.15 g, 2.36 mmol)和碳载钯(0.251 g, 2.36 mmol)搅拌过夜。过滤该反应,并浓缩滤液以得到标题化合物(250 mg, 0.584 mmol, 25%收率)。LCMS [M+H]+ = 429.1。4,4'-((2,2,3,3-tetrafluorobutane-1,4-diyl)bis(azanediyl))bis(3-nitrobenzamide) (1.15 g, 2.36 mmol) and palladium on carbon (0.251 g, 2.36 mmol) in MeOH (100 mL) were stirred under H2 at 30 °C overnight. The reaction was filtered and the filtrate was concentrated to give the title compound (250 mg, 0.584 mmol, 25% yield). LCMS [M+H] + = 429.1.
步骤3: 1,1'-(2,2,3,3-四氟丁烷-1,4-二基)双(2-氨基-1H-苯并[d]咪唑-5-甲酰胺)Step 3: 1,1'-(2,2,3,3-tetrafluorobutane-1,4-diyl)bis(2-amino-1H-benzo[d]imidazole-5-carboxamide)
向在MeOH (1 mL)和水(2 mL)中的4,4'-((2,2,3,3-四氟丁烷-1,4-二基)双(氮烷二基))双(3-氨基苯甲酰胺) (20 mg, 0.047 mmol)加入溴化氰 (29.7 mg, 0.280 mmol),并在30℃搅拌该反应过夜。在真空中除去MeOH并通过过滤收集所得固体以得到标题化合物(15 mg, 0.031 mmol, 67%收率)。LCMS [M+H]+ = 479.0。To 4,4'-((2,2,3,3-tetrafluorobutane-1,4-diyl)bis(azanediyl))bis(3-aminobenzamide) (20 mg, 0.047 mmol) in MeOH (1 mL) and water (2 mL) was added cyanogen bromide (29.7 mg, 0.280 mmol) and the reaction was stirred at 30°C overnight. The MeOH was removed in vacuo and the resulting solid was collected by filtration to give the title compound (15 mg, 0.031 mmol, 67% yield). LCMS [M+H] + = 479.0.
步骤4: 1,1'-(2,2,3,3-四氟丁烷-1,4-二基)双(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺)Step 4: 1,1'-(2,2,3,3-tetrafluorobutane-1,4-diyl)bis(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide)
在室温向在DMF (20 mL)中的HATU (763 mg, 2.01 mmol)和1-乙基-3-甲基-1H-吡唑-5-甲酸 (227 mg, 1.47 mmol)以一批加入EDC (385 mg, 2.01mmol)、1,1'-(2,2,3,3-四氟丁烷-1,4-二基)双(2-氨基-1H-苯并[d]咪唑-5-甲酰胺) (320 mg, 0.667 mmol)和DIEA (0.467 mL, 2.68 mmol)。将该反应加热至70℃持续12 h,浓缩并纯化以得到标题化合物(8 mg, 0.01 mmol, 2%收率)。1H NMR (400 MHz, DMSO-d 6 ) δ ppm 13.05 (s, 2 H),8.01 (d, J=8.6 Hz, 4 H), 7.81 (d, J=8.2 Hz, 2 H), 7.53 (d, J=8.3 Hz, 2 H),7.38 (s, 2 H), 6.73 (s, 2 H), 5.32 (t, J=16.0 Hz, 4 H), 4.59 (dd, J=14.0, 6.9Hz,4 H), 2.06 (s, 6 H), 1.33 (t, J=7.1 Hz, 6 H); LCMS [M+H]+ = 751.1。To HATU (763 mg, 2.01 mmol) and 1-ethyl-3-methyl-1H-pyrazole-5-carboxylic acid (227 mg, 1.47 mmol) in DMF (20 mL) was added EDC (385 mg, 2.01 mmol), 1,1'-(2,2,3,3-tetrafluorobutane-1,4-diyl)bis(2-amino-1H-benzo[d]imidazole-5-carboxamide) (320 mg, 0.667 mmol) and DIEA (0.467 mL, 2.68 mmol) in one portion at room temperature. The reaction was heated to 70 °C for 12 h, concentrated and purified to give the title compound (8 mg, 0.01 mmol, 2% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 13.05 (s, 2 H), 8.01 (d, J =8.6 Hz, 4 H), 7.81 (d, J =8.2 Hz, 2 H), 7.53 (d, J =8.3 Hz, 2 H), 7.38 (s, 2 H), 6.73 (s, 2 H ), 5.32 (t, J =16.0 Hz, 4 H), 4.59 (dd, J =14.0, 6.9Hz, 4 H), 2.06 (s, 6 H), 1.33 (t, J =7.1 Hz, 6 H); LCMS [M+H] + = 751.1.
步骤5: (2E,2'E)-1,1'-(2,2,3,3-四氟丁烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺), 三氟乙酸盐Step 5: (2E,2'E)-1,1'-(2,2,3,3-tetrafluorobutane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide), trifluoroacetate
向100 mL 圆底烧瓶加入1,1'-(2,2,3,3-四氟丁烷-1,4-二基)双(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺) (49 mg, 0.065 mmol)和DMF(0.653 mL)。向溶液加入碳酸铯 (63.8 mg, 0.196 mmol),然后加入碘甲烷 (10.20 µL,0.163 mmol)。在室温搅拌混合物。在30分钟后,加入更多的碘甲烷(10 uL; 0.16 mmol)并在室温搅拌混合物过夜(~14小时)。将这个透明粗混合物直接注射至反相制备型HPLC系统中并纯化(双相ISCO, 20-50% CH3CN / H2O梯度, TFA改性剂)。合并纯级分并浓缩以获得作为灰白色半固体的(2E,2'E)-1,1'-(2,2,3,3-四氟丁烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺), 三氟乙酸盐 (3 mg, 3.19 µmol, 4.89%收率)。1H NMR (400 MHz, 甲醇-d4) δ ppm 1.34 (t, J=7.10 Hz, 6 H) 2.25 (s, 6 H) 3.70 (s, 6 H) 4.60 (q, J=7.10 Hz, 4 H) 5.19 (t, J=15.33 Hz, 4 H) 6.66 (s, 2 H) 7.60 (d, J=8.36 Hz, 2 H) 7.96 (dd, J=8.36, 1.52Hz, 2 H) 8.11 (d, J=1.27 Hz, 2 H)。LCMS m/z = 779 [M+H]+。To a 100 mL round bottom flask was added 1,1'-(2,2,3,3-tetrafluorobutane-1,4-diyl)bis(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide) (49 mg, 0.065 mmol) and DMF (0.653 mL). To the solution was added cesium carbonate (63.8 mg, 0.196 mmol) followed by iodomethane (10.20 µL, 0.163 mmol). The mixture was stirred at room temperature. After 30 minutes, more iodomethane (10 uL; 0.16 mmol) was added and the mixture was stirred at room temperature overnight (~14 hours). This clear crude mixture was directly injected into a reverse phase preparative HPLC system and purified (biphasic ISCO, 20-50% CH 3 CN / H 2 O gradient, TFA modifier). Pure fractions were combined and concentrated to afford (2E,2'E)-1,1'-(2,2,3,3-tetrafluorobutane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide), trifluoroacetate salt (3 mg, 3.19 µmol, 4.89% yield) as an off-white semisolid. 1 H NMR (400 MHz, methanol- d 4) δ ppm 1.34 (t, J =7.10 Hz, 6 H) 2.25 (s, 6 H) 3.70 (s, 6 H) 4.60 (q, J =7.10 Hz, 4 H) 5.19 (t, J =15.33 Hz, 4 H) 6.66 (s, 2 H) 7.60 (d, J =8.36 Hz, 2 H) 7.96 (dd, J =8.36, 1.52Hz, 2 H) 8.11 (d, J =1.27 Hz, 2 H). LCMS m/z = 779 [M+H] + .
实施例7Example 7
1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺, 2 三氟乙酸盐1-((E)-4-((E)-5-Carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide, 2-trifluoroacetate
步骤1: (E)-1-((E)-4-((4-氨基甲酰基-2-硝基苯基)氨基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 1: (E)-1-((E)-4-((4-Carbamoyl-2-nitrophenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
将4-氟-3-硝基苯甲酰胺 (86 mg, 0.467 mmol), (E)-1-((E)-4-氨基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 3盐酸盐 (在PU66420P中使用的名称: (Z)-1-((E)-4-氨基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 3 盐酸盐 (250 mg, 0.467 mmol)和DIPEA (0.245 mL,1.402 mmol)悬浮在异丙醇 (2 mL)中并在120℃在密封小瓶中加热。在18 h后,用25 mLEtOAc稀释该反应并用2 x 25 mL水、25 mL饱和碳酸氢钠溶液和25 mL盐水洗涤。用25 mLEtOAc反萃取水层。收集有机层,在真空下浓缩以提供粗固体产物。将粗产物溶解于6 mLDMSO中,过滤,并通过质量导向的(mass-directed)制备型 -HPLC纯化。合并纯级分,在真空下除去有机物,并采用2 x 50 mL DCM由水性溶剂萃取该化合物。在真空下除去有机物以提供作为黄色固体的标题化合物(102 mg, 0.173 mmol, 37%收率)。LCMS m/z = 590 [M +H]+。4-Fluoro-3-nitrobenzamide (86 mg, 0.467 mmol), (E)-1-((E)-4-aminobut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 3 hydrochloride (name used in PU66420P: (Z)-1-((E)-4-aminobut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 3 hydrochloride (250 mg, 0.467 mmol) and DIPEA (0.245 4-[4-[4-[4-[4-(4-[4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4- ( 4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-(4-
步骤2: (E)-1-((E)-4-((2-氨基-4-氨基甲酰基苯基)氨基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 2: (E)-1-((E)-4-((2-amino-4-carbamoylphenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向(E)-1-((E)-4-((4-氨基甲酰基-2-硝基苯基)氨基) 丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基) 亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (105 mg, 0.178 mmol)在乙酸(0.500 mL)和MeOH (0.5 mL)中的悬浮液加入在活性炭上的1 wt % Pt和2 wt %钒—50-70%湿化粉末(34.7 mg, 1.781 µmol, Strem, 78-1536)。排空该烧瓶并用氮气吹扫,并将其再重复两次。排空该烧瓶并用氢气球冲洗,并在室温在氢气气氛下搅拌。由于在5 h后未完全转化,使用MeOH将该反应混合物过滤通过小型硅藻土塞。在真空下浓缩该反应混合物并存放在冰箱中。将粗物质再次溶解于乙酸(0.500mL)和MeOH (0.5 mL)中,并加入活性炭上的1 wt % Pt和2 wt % 钒 V—50-70%湿化粉末(34.7 mg, 1.781 µmol)。排空该烧瓶并用氮气吹扫,将其再重复两次。排空该烧瓶并用氢气球冲洗,并在室温在氢气气氛下搅拌。在2 h后,使用MeOH将该反应混合物过滤通过小型硅藻土塞,然后在真空下浓缩以获得作为红色油的标题化合物(163 mg, 0.148 mmol, 82%收率)。LCMS m/z = 280 [M+2H/2]+。To a suspension of (E)-1-((E)-4-((4-carbamoyl-2-nitrophenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (105 mg, 0.178 mmol) in acetic acid (0.500 mL) and MeOH (0.5 mL) was added 1 wt % Pt and 2 wt % Vanadium on activated carbon - 50-70% wet powder (34.7 mg, 1.781 µmol, Strem, 78-1536). The flask was evacuated and purged with nitrogen, and this was repeated two more times. The flask was evacuated and flushed with a hydrogen balloon, and stirred at room temperature under a hydrogen atmosphere. Due to incomplete conversion after 5 h, MeOH is used to filter the reaction mixture through a small diatomite plug. The reaction mixture is concentrated under vacuum and stored in a refrigerator. The crude material is dissolved in acetic acid (0.500mL) and MeOH (0.5 mL) again, and 1 wt% Pt and 2 wt% vanadium V-50-70% wetted powder (34.7 mg, 1.781 μmol) on activated carbon are added. The flask is emptied and purged with nitrogen, which is repeated twice more. The flask is emptied and rinsed with a hydrogen balloon, and stirred at room temperature under a hydrogen atmosphere. After 2 h, MeOH is used to filter the reaction mixture through a small diatomite plug, then concentrated under vacuum to obtain the title compound (163 mg, 0.148 mmol, 82% yield) as a red oil. LCMS m/z=280 [M+2H/2] + .
步骤3: 1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺, 2三氟乙酸盐Step 3: 1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide, 2-trifluoroacetate
在0℃向在DMF (1 mL)中的(E)-1-((E)-4-((2-氨基-4-氨基甲酰基苯基)氨基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基) 亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (100 mg, 0.179 mmol)加入1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (0.491 mL, 0.197 mmol, 0.4 M 在二氧杂环己烷中)并在0℃搅拌1 h。在1 h后,加入EDC (51.4 mg, 0.268 mmol)和三乙胺 (0.075 mL, 0.536 mmol)并在40℃搅拌该反应3 h并在室温过夜。用1.5 mL DMSO 稀释该反应并通过质量导向的制备型HPLC(碱性改性剂)纯化标题化合物。收集纯级分并在真空下除去有机物。然后,用2 x 25 mLDCM萃取该化合物,并用10 mL盐水洗涤有机层。在真空下除去挥发物以提供标题化合物(通过LCMS的~80%纯度)。用2.0 mL DMSO稀释该化合物并通过质量导向的制备型 HPLC (TFA改性剂)再次纯化。收集纯级分并在真空下除去溶剂以提供作为黄色油的标题化合物(15 mg,0.016 mmol, 8.8%收率)。1H NMR (400 MHz, 甲醇-d 4) δ ppm 1.31 - 1.38 (m, 6 H),2.24 - 2.26 (m, 6 H), 3.71 (s, 3 H), 3.88 (s, 3 H), 4.50 (q, J=7.10 Hz, 2 H),4.60 (q, J=7.10 Hz, 2 H), 4.88 (d, J=5.83 Hz, 2 H), 5.11 (d, J=5.07 Hz, 2 H),5.78 (dt, J=15.40, 5.73 Hz, 1 H), 5.95 - 6.03 (m, 1 H), 6.63 (s, 1 H), 6.65(s, 1 H), 7.36 (d, J=8.36 Hz, 1 H), 7.51 (d, J=1.27 Hz, 1 H), 7.74 (dd, J=8.36, 1.52 Hz, 1 H), 7.79 (d, J=1.01 Hz, 1 H), 8.00 (d, J=1.52 Hz, 1 H)。LCMSm/z = 721 [M + H]+。To (E)-1-((E)-4-((2-amino-4-carbamoylphenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (100 mg, 0.179 mmol) in DMF (1 mL) was added 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (0.491 mL, 0.197 mmol, 0.4 M in dioxane) at 0°C and stirred at 0°C for 1 h. After 1 h, EDC (51.4 mg, 0.268 mmol) and triethylamine (0.075 mL, 0.536 mmol) were added and the reaction was stirred at 40°C for 3 h and at room temperature overnight. The reaction is diluted with 1.5 mL DMSO and the title compound is purified by mass-guided preparative HPLC (alkaline modifier). Pure fractions are collected and organic matter is removed under vacuum. Then, the compound is extracted with 2 x 25 mL DCM, and the organic layer is washed with 10 mL salt water. Volatiles are removed under vacuum to provide the title compound (by LCMS~80% purity). The compound is diluted with 2.0 mL DMSO and purified again by mass-guided preparative HPLC (TFA modifier). Pure fractions are collected and solvent is removed under vacuum to provide the title compound (15 mg, 0.016 mmol, 8.8% yield) as a yellow oil. 1 H NMR (400 MHz, methanol- d 4 ) δ ppm 1.31 - 1.38 (m, 6 H), 2.24 - 2.26 (m, 6 H), 3.71 (s, 3 H), 3.88 (s, 3 H), 4.50 (q, J =7.10 Hz, 2 H), 4.60 (q, J =7.10 Hz, 2 H), 4.88 (d, J =5.83 Hz, 2 H), 5.11 (d, J =5.07 Hz, 2 H), 5.78 (dt, J =15.40, 5.73 Hz, 1 H), 5.95 - 6.03 (m, 1 H), 6.63 (s, 1 H), 6.65(s, 1 H ), 7.36 (d, J =8.36 Hz, 1 H), 7.51 (d, J =1.27 Hz, 1 H), 7.74 (dd, J =8.36, 1.52 Hz, 1 H), 7.79 (d, J =1.01 Hz, 1 H), 8.00 (d, J =1.52 Hz, 1 H). LCMSm/z = 721 [M + H] + .
实施例8Example 8
1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺, 2三氟乙酸盐1-((E)-4-((E)-5-Carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide, 2-trifluoroacetate
步骤1: (E)-1-((E)-4-((4-氨基甲酰基-2-甲氧基-6-硝基苯基)氨基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 1: (E)-1-((E)-4-((4-Carbamoyl-2-methoxy-6-nitrophenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
将4-氯-3-甲氧基-5-硝基苯甲酰胺 (108 mg, 0.467 mmol), (E)-1-((E)-4-氨基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 3盐酸盐 (250 mg, 0.467 mmol)和DIPEA (0.245 mL,1.402 mmol)悬浮在异丙醇 (2 mL)中并在120℃在密封小瓶中加热。在22 h后,用25 mLEtOAc稀释该反应并用2 x 25 mL水、25 mL 饱和碳酸氢钠溶液和25 mL盐水洗涤。用25 mLEtOAc萃取水层。收集有机层,并在真空下浓缩以提供作为橙色固体的粗产物。将粗产物溶解于12 mL DMSO中,过滤并通过质量导向的制备型HPLC (高pH改性剂, 多次注射)直接纯化。合并纯级分,在真空下除去有机物,并采用2 x 50 mL DCM由水性溶剂萃取标题化合物。在真空下除去挥发物以提供作为橙色固体的标题化合物(53 mg, 0.086 mmol, 18.3%收率)。LCMS m/z = 620 [M + H]+。4-Chloro-3-methoxy-5-nitrobenzamide (108 mg, 0.467 mmol), (E)-1-((E)-4-aminobut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 3 hydrochloride (250 mg, 0.467 mmol) and DIPEA (0.245 mL, 1.402 mmol) were suspended in isopropanol (2 mL) and heated at 120 °C in a sealed vial. After 22 h, the reaction was diluted with 25 mL EtOAc and washed with 2 x 25 mL water, 25 mL saturated sodium bicarbonate solution and 25 mL brine. The aqueous layer was extracted with 25 mL EtOAc. The organic layer is collected and concentrated under vacuum to provide a crude product as an orange solid. The crude product is dissolved in 12 mL DMSO, filtered and directly purified by mass-guided preparative HPLC (high pH modifier, multiple injections). Pure fractions are merged, organic matter is removed under vacuum, and the title compound is extracted by aqueous solvent using 2 x 50 mL DCM. Volatiles are removed under vacuum to provide the title compound (53 mg, 0.086 mmol, 18.3% yield) as an orange solid. LCMS m/z = 620 [M + H] + .
步骤2: (E)-1-((E)-4-((2-氨基-4-氨基甲酰基-6-甲氧基苯基)氨基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 2: (E)-1-((E)-4-((2-amino-4-carbamoyl-6-methoxyphenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向(E)-1-((E)-4-((4-氨基甲酰基-2-甲氧基-6-硝基苯基)氨基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (在PU66420P中使用的名称: (Z)-1-((E)-4-((4-氨基甲酰基-2-甲氧基-6-硝基苯基)氨基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (50 mg, 0.081 mmol))在乙酸(0.500mL)和MeOH (0.5 mL)中的悬浮液加入在活性炭上的 1 wt % Pt和2 wt % 钒—50-70%湿化粉末(15.74 mg, 0.807 µmol, Strem, 78-1536)。排空该小瓶并用氮气吹扫,并将其再重复两次。排空该小瓶并用氢气球冲洗,然后在室温在氢气气氛下搅拌。在4 h后,使用MeOH将该反应混合物过滤通过小型硅藻土塞,然后在真空下浓缩以提供作为浅红色固体的粗产物混合物(52 mg)。LCMS m/z = 590 [M + H]+。To (E)-1-((E)-4-((4-carbamoyl-2-methoxy-6-nitrophenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (name used in PU66420P: (Z)-1-((E)-4-((4-carbamoyl-2-methoxy-6-nitrophenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (50 mg, 0.081 mmol)) in acetic acid (0.500mL) and MeOH (0.5 mL) suspension was added to 1 wt% Pt and 2 wt% vanadium-50-70% wet powder on activated carbon (15.74 mg, 0.807 μmol, Strem, 78-1536). The vial was evacuated and purged with nitrogen, and it was repeated twice more. The vial was evacuated and rinsed with a hydrogen balloon, and then stirred at room temperature under a hydrogen atmosphere. After 4 h, the reaction mixture was filtered through a small diatomaceous earth plug using MeOH, and then concentrated under vacuum to provide a crude product mixture (52 mg) as a light red solid. LCMS m/z = 590 [M + H] + .
步骤3: 1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺, 2三氟乙酸盐Step 3: 1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide, 2-trifluoroacetate
在0℃向在DMF (1 mL)中的(E)-1-((E)-4-((2-氨基-4-氨基甲酰基-6-甲氧基苯基)氨基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (60 mg, 0.102 mmol)加入作为在二氧杂环己烷中的0.4 M溶液的1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (0.280 mL, 0.112 mmol)并在0℃搅拌1 h。在1 h后,加入EDC (29.3 mg, 0.153 mmol)和三乙胺 (0.043 mL, 0.305mmol),并在40℃搅拌该反应2 h,然后在室温搅拌18 h。用1.5 mL DMSO稀释该反应混合物并在质量导向的制备型HPLC (高pH改性剂)上纯化。需要第二制备型HPLC纯化步骤(使用TFA-改性剂)以提供作为灰白色固体的纯标题化合物(5.0 mg, 5.11 mmol, 5.0%收率)。1HNMR (400 MHz, DMSO-d 6) δ ppm 1.21 (t, J=7.10 Hz, 3 H) 1.26 (t, J=7.10 Hz, 3H) 2.10 (s, 3 H) 2.12 (s, 3 H) 3.50 (s, 3 H) 3.75 (s, 3 H) 3.80 (s, 3 H) 4.43(q, J=7.18 Hz, 2 H) 4.53 (q, J=7.01 Hz, 2 H) 4.82 - 4.91 (m, 4 H) 5.76 - 5.80(m, 2 H) 6.36 (s, 1 H) 6.52 (s, 1 H) 7.33 (d, J=1.01 Hz, 1 H) 7.38 (br. s., 1H) 7.43 (d, J=0.76 Hz, 1 H) 7.48 (br. s, 1 H) 7.65 (d, J=1.27 Hz, 1 H) 7.72(d, J=1.01 Hz, 1 H) 7.99 (br. s., 1 H) 8.05 (br. s, 1 H) 12.85 (s, 1 H)。LCMSm/z = 751 [M + H]+。To (E)-1-((E)-4-((2-amino-4-carbamoyl-6-methoxyphenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (60 mg, 0.102 mmol) in DMF (1 mL) at 0 °C was added 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (0.280 mL, 0.112 mmol) as a 0.4 M solution in dioxane and stirred at 0 °C for 1 h. After 1 h, EDC (29.3 mg, 0.153 mmol) and triethylamine (0.043 mL, 0.305 mmol) were added and the reaction was stirred at 40 °C for 2 h, then at room temperature for 18 h. The reaction mixture was diluted with 1.5 mL DMSO and purified on mass-directed preparative HPLC (high pH modifier). A second preparative HPLC purification step (using TFA-modifier) was required to provide the pure title compound (5.0 mg, 5.11 mmol, 5.0% yield) as an off-white solid. 1 HNMR (400 MHz, DMSO- d 6 ) δ ppm 1.21 (t, J =7.10 Hz, 3 H) 1.26 (t, J =7.10 Hz, 3H) 2.10 (s, 3 H) 2.12 (s, 3 H) 3.50 (s, 3 H) 3.75 (s, 3 H) 3.80 (s, 3 H) 4.43(q, J =7.18 Hz, 2 H) 4.53 (q, J =7.01 Hz, 2 H) 4.82 - 4.91 (m, 4 H) 5.76 - 5.80(m, 2 H) 6.36 (s, 1 H) 6.52 (s, 1 H) 7.33 (d, J =1.01 Hz, 1 H) 7.38 (br. s., 1H) 7.43 (d, J =0.76 Hz, 1 H) 7.48 (br. s, 1 H) 7.65 (d, J =1.27 Hz, 1 H) 7.72 (d, J =1.01 Hz, 1 H) 7.99 (br. s., 1 H) 8.05 ( br. s, 1 H) 12.85 (s, 1 H). LCMSm/z = 751 [M + H] + .
实施例9Example 9
1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-羟基丙氧基)-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐1-((E)-4-((E)-5-Carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-hydroxypropoxy)-1H-benzo[d]imidazole-5-carboxamide, 2-hydrochloride
步骤1: ((E)-1-((E)-4-((2-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基-6-硝基苯基)氨基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 1: ((E)-1-((E)-4-((2-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoyl-6-nitrophenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
将3-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氯-5-硝基苯甲酰胺 (182mg, 0.467 mmol), (E)-1-((E)-4-氨基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 3盐酸盐 (250mg, 0.467 mmol)和DIPEA (0.245 mL, 1.402 mmol)悬浮于异丙醇 (2 mL)中并在120℃在密封小瓶中加热。在22 h后,用25 mL EtOAc稀释该反应并用2 x 25 mL水、25 mL 饱和碳酸氢钠溶液和25 mL盐水洗涤。用25 mL EtOAc反萃取水层。收集有机层,并在真空下浓缩。将粗产物溶解在6 mL DMSO中,过滤并通过质量导向的制备型HPLC (高pH 改性剂)纯化。合并纯级分,在真空下除去有机物,并采用2 x 50 mL DCM由水性溶剂萃取该化合物。溶剂蒸发提供作为橙色固体的标题化合物(80 mg, 0.103 mmol, 22%收率)。LCMS m/z = 778 [M +H]+。3-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-chloro-5-nitrobenzamide (182 mg, 0.467 mmol), (E)-1-((E)-4-aminobut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 3 hydrochloride (250 mg, 0.467 mmol) and DIPEA (0.245 mL, 1.402 mmol) were suspended in isopropanol (2 mL) and heated in a sealed vial at 120° C. After 22 h, the reaction was diluted with 25 mL EtOAc and washed with 2 x 25 mL water, 25 mL saturated sodium bicarbonate solution and 25 mL brine. The aqueous layer was back extracted with 25 mL EtOAc. The organic layer was collected and concentrated under vacuum. The crude product was dissolved in 6 mL DMSO, filtered and purified by mass-oriented preparative HPLC (high pH modifier). Pure fractions were combined, organic matter was removed under vacuum, and 2 x 50 mL DCM was used to extract the compound by aqueous solvent. Solvent evaporation provided the title compound (80 mg, 0.103 mmol, 22% yield) as an orange solid. LCMS m/z = 778 [M +H] + .
步骤2: (E)-1-((E)-4-((2-氨基-6-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基苯基)氨基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 2: (E)-1-((E)-4-((2-amino-6-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoylphenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向(E)-1-((E)-4-((2-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基-6-硝基苯基)氨基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (在PU66420P中使用的名称: (Z)-1-((E)-4-((2-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基-6-硝基苯基)氨基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺) (80 mg, 0.103 mmol)在乙酸(0.500 mL)和MeOH(0.5 mL)中的悬浮液加入在活性炭上的 1 wt % Pt和2 wt % 钒—50-70%湿化粉末(20.06mg, 1.028 µmol, Strem 78-1536)。排空该小瓶并用氮气吹扫,并使其再重复两次。排空该小瓶并用氢气球冲洗,并在室温在氢气气氛下搅拌。在4 h后,使用MeOH将该反应混合物过滤通过小型硅藻土塞,然后浓缩并在真空下干燥以提供标题化合物(77 mg, 0.072 mmol,70%收率)。LCMS m/z = 748 [M + H]+。(E)-1-((E)-4-((2-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoyl-6-nitrophenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (name used in PU66420P: A suspension of (Z)-1-((E)-4-((2-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoyl-6-nitrophenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide) (80 mg, 0.103 mmol) in acetic acid (0.500 mL) and MeOH (0.5 mL) was added 1 wt % Pt and 2 wt % Vanadium on activated carbon - 50-70% wet powder (20.06 mg, 1.028 µmol, Strem 78-1536). The vial was evacuated and purged with nitrogen and this was repeated two more times. The vial was evacuated and flushed with a hydrogen balloon and stirred at room temperature under a hydrogen atmosphere. After 4 h, the reaction mixture was filtered through a small celite plug using MeOH, then concentrated and dried under vacuum to provide the title compound (77 mg, 0.072 mmol, 70% yield). LCMS m/z = 748 [M + H] + .
步骤3: 7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 3: 7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide
在0℃向在DMF (1 mL)中的(E)-1-((E)-4-((2-氨基-6-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基苯基)氨基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (80 mg,0.107 mmol)加入在二氧杂环己烷中的1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯(0.294 mL, 0.118 mmol, 0.4 M)并在0℃搅拌1 h。加入EDC (30.8 mg, 0.160 mmol)和三乙胺 (0.045 mL, 0.321 mmol),并在40℃搅拌该反应2 h并在室温保持18 h。将该反应用1.5 mL DMSO稀释并使用质量导向的制备型HPLC (高pH 改性剂)纯化。收集纯级分并在真空下除去有机物。然后,用2 x 25 mL DCM萃取化合物,并用10 mL盐水洗涤有机层。在真空下除去挥发物以提供作为灰白色固体的标题化合物(25 mg, 0.027 mmol, 26%收率)。LCMSm/z = 909 [M+H]+。To (E)-1-((E)-4-((2-amino-6-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoylphenyl)amino)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (80 mg, 0.107 mmol) in DMF (1 mL) at 0 °C was added 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (0.294 mL, 0.118 mmol, 0.4 M) in dioxane and stirred at 0 °C for 1 h. EDC (30.8 mg, 0.160 mmol) and triethylamine (0.045 mL, 0.321 mmol) were added, and the reaction was stirred at 40 ° C for 2 h and kept at room temperature for 18 h. The reaction was diluted with 1.5 mL DMSO and purified using mass-oriented preparative HPLC (high pH modifier). Pure fractions were collected and organic matter was removed under vacuum. Then, the compound was extracted with 2 x 25 mL DCM, and the organic layer was washed with 10 mL brine. Volatiles were removed under vacuum to provide the title compound (25 mg, 0.027 mmol, 26% yield) as an off-white solid. LCMSm/z = 909 [M+H] + .
步骤4: 1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-羟基丙氧基)-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐Step 4: 1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-hydroxypropoxy)-1H-benzo[d]imidazole-5-carboxamide, 2-hydrochloride
向在MeOH (1 mL)中的7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (25 mg, 0.027 mmol)加入在二氧杂环己烷中的盐酸(0.069 mL, 0.275 mmol, 4 M)并在室温搅拌该反应。在10 min后,在真空下除去挥发物以提供作为白色固体的标题化合物(22 mg, 0.025 mmol, 92%收率)。1H NMR (400 MHz, 甲醇-d 4) δ ppm 1.29 - 1.36 (m, 3 H), 1.45 (t, J=6.72 Hz, 3 H), 1.85 - 1.91 (m,2 H), 2.29 (s, 3 H), 2.36 (s, 3 H), 3.64 (t, J=5.45 Hz, 2 H), 3.89 (s, 3 H),3.91 (s, 3 H), 4.18 (t, J=5.32 Hz, 2 H), 4.38 (q, J=6.51 Hz, 2 H), 4.69 (q, J=6.80 Hz, 2 H), 5.19 (br. s., 2 H), 5.30 (br. s., 2 H), 5.85 - 6.06 (m, 2 H),6.80 (s, 1 H), 6.99 (s, 1 H), 7.43 (s, 1 H), 7.61 (s, 1 H), 7.74 (s, 1 H),8.00 (s, 1 H)。LCMS m/z = 795 [M + H]+。To 7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (25 mg, 0.027 mmol) in MeOH (1 mL) was added hydrochloric acid in dioxane (0.069 mL, 0.275 mmol, 4 M) and the reaction was stirred at room temperature. After 10 min, the volatiles were removed under vacuum to afford the title compound as a white solid (22 mg, 0.025 mmol, 92% yield). 1 H NMR (400 MHz, methanol- d 4 ) δ ppm 1.29 - 1.36 (m, 3 H), 1.45 (t, J =6.72 Hz, 3 H), 1.85 - 1.91 (m, 2 H), 2.29 (s, 3 H), 2.36 (s, 3 H), 3.64 (t, J =5.4 5 Hz, 2 H), 3.89 (s, 3 H), 3.91 (s, 3 H), 4.18 (t, J =5.32 Hz, 2 H), 4.38 (q, J =6.51 Hz, 2 H), 4.69 (q, J =6.80 Hz, 2 H), 5.19 (br. s., 2 H), 5.30 (br . s., 2 H), 5.85 - 6.06 (m, 2 H), 6.80 (s, 1 H), 6.99 (s, 1 H), 7.43 (s, 1 H), 7.61 (s, 1 H), 7.74 (s, 1 H), 8.00 (s, 1 H). LCMS m/z = 795 [M + H] + .
实施例10Example 10
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 2三氟乙酸盐(E)-1-((E)-4-((E)-5-Carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 2-trifluoroacetate
向20-mL小瓶放入(E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-吗啉代丙氧基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺 (200 mg, 0.235 mmol)和DMF (2.35 mL)。向这个溶液中加入碳酸铯 (230 mg, 0.706 mmol)和碘甲烷 (37 µL,0.588 mmol)。在室温搅拌该溶液15 min。将DMF (2 mL)和水(2 mL)直接加入该小瓶。使用质量导向的制备型HPLC (15-55%梯度的MeCN/水,含NH4OH作为改性剂)直接纯化这种混合物。将相应级分合并并浓缩。通过质量导向的制备型HPLC (5-35%梯度的MeCN/水,含TFA作为改性剂)进一步纯化经浓缩的混合物。将相应的级分合并并浓缩以提供作为透明油的(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 2三氟乙酸盐 (3 mg, 2.69 umol, 1.14%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm9.94 (br. s., 2 H), 8.09 (br. s., 2 H), 7.81 (s, 1 H), 7.78 (s, 1 H), 7.53(br. s., 2 H), 7.42 (d, J=6.08 Hz, 2 H), 6.42 (m, 2 H), 5.51 - 5.85 (m, 2 H),4.72 - 4.99 (m, 4 H), 4.42 (q, J=6.84 Hz, 4 H), 4.05 (t, J=5.58 Hz, 2 H),3.95 (d, J=11.66 Hz, 2 H), 3.69 (s, 3 H), 3.62 (t, J=11.91 Hz, 2 H), 3.56 (s,3 H), 3.54 (s, 3 H), 3.34 (d, J=11.91 Hz, 2 H), 3.19 (d, J=7.10 Hz, 2 H),3.02 (br. s., 2 H), 2.12 (s, 6 H), 1.94 (m, 2 H), 1.21 (m, 6 H)。LCMS (m/z):878.7 [M + H]+。Into a 20-mL vial was placed (E)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-morpholinopropoxy)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide (200 mg, 0.235 mmol) and DMF (2.35 mL). To this solution was added cesium carbonate (230 mg, 0.706 mmol) and iodomethane (37 µL, 0.588 mmol). The solution was stirred at room temperature for 15 min. DMF (2 mL) and water (2 mL) were added directly to the vial. This mixture was directly purified using mass-directed preparative HPLC (15-55% gradient of MeCN/water with NH4OH as modifier). The corresponding fractions were combined and concentrated. The concentrated mixture was further purified by mass-directed preparative HPLC (5-35% gradient of MeCN/water with TFA as modifier). The corresponding fractions were combined and concentrated to afford (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 2 trifluoroacetate salt (3 mg, 2.69 umol, 1.14% yield) as a clear oil. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm9.94 (br. s., 2 H), 8.09 (br. s., 2 H), 7.81 (s, 1 H), 7.78 (s, 1 H), 7.53 (br. s., 2 H), 7.42 (d, J =6.08 Hz, 2 H), 6.42 (m, 2 H), 5.51 - 5.85 (m, 2 H), 4.72 - 4.99 (m, 4 H), 4.42 (q, J =6.84 Hz, 4 H), 4.05 (t, J =5.58 Hz, 2 H), 3.95 (d, J =11.66 Hz, 2 H), 3.69 (s, 3H), 3.62 (t, J =11.91 Hz, 2 H), 3.56 (s, 3 H), 3.54 (s, 3 H), 3.34 (d, J =11.91 Hz, 2 H), 3.19 (d, J =7.10 Hz, 2 H), 3.02 (br. s., 2 H), 2.12 (s, 6 H), 1.94 ( m, 2 H), 1.21 (m, 6 H). LCMS ( m/z ):878.7 [M + H] + .
实施例11Embodiment 11
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-(4-甲基-4l4-吗啉代)丙氧基)-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 2 三氟乙酸盐, 三氟乙酸盐(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-(4-methyl-4-morpholino)propoxy)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 2-trifluoroacetate, trifluoroacetate
向20-mL小瓶放入(E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-吗啉代丙氧基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺 (200 mg, 0.235 mmol)和DMF (2.35 mL)。向这个溶液加入碳酸铯 (230 mg, 0.706 mmol)和碘甲烷 (37 µL, 0.588mmol)。在室温搅拌该溶液15 min。将DMF (2 mL)和水(2 mL)直接加入该小瓶。使用质量导向的制备型HPLC (15-55%梯度的MeCN/水,含NH4OH作为改性剂)直接纯化这种混合物。将相应的级分合并并浓缩。通过反相制备型HPLC (5-35%梯度的MeCN/水,含TFA 作为改性剂)使用酸性改性剂纯化经浓缩的混合物。将相应的级分合并并浓缩以提供作为透明油的标题化合物(6 mg, 4.81 umol, 2.04%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.08 (br. s.,2 H), 7.81 (s, 2 H), 7.77 (s, 1 H), 7.53 (br. s., 3 H), 7.39-7.40 (m, 3 H),6.41 (s, 1 H), 6.40 (s, 1 H), 5.60-5.75 (m, 2 H), 4.74 - 4.97 (m, 4 H), 4.35-4.51 (m, 4 H), 4.02 (t, J=5.32 Hz, 2 H), 3.82-3.98 (m, 4 H), 3.67 (s, 3 H),3.50-3.54 (m, 8 H), 3.08 (s, 3 H), 2.12 (s, 6 H), 1.95-2.05 (m., 2 H), 1.24-1.20 (m, 6 H)。LCMS (m/z): 892.7 [M]+。Into a 20-mL vial was placed (E)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-morpholinopropoxy)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide (200 mg, 0.235 mmol) and DMF (2.35 mL). To this solution was added cesium carbonate (230 mg, 0.706 mmol) and iodomethane (37 µL, 0.588 mmol). The solution was stirred at room temperature for 15 min. DMF (2 mL) and water (2 mL) were added directly to the vial. This mixture was directly purified using mass-directed preparative HPLC (15-55% gradient of MeCN/water with NH4OH as modifier). The corresponding fractions were combined and concentrated. The concentrated mixture was purified by reverse phase preparative HPLC (5-35% gradient of MeCN/water with TFA as modifier) using an acidic modifier. The corresponding fractions were combined and concentrated to provide the title compound (6 mg, 4.81 umol, 2.04% yield) as a clear oil. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.08 (br. s.,2 H), 7.81 (s, 2 H), 7.77 (s, 1 H), 7.53 (br. s., 3 H), 7.39-7.40 (m, 3 H), 6.41 (s, 1 H), 6.40 (s, 1 H), 5.60-5.75 (m, 2 H), 4.74 - 4.97 (m, 4 H), 4.35-4.51 (m, 4 H), 4.02 (t, J =5.32 Hz, 2 H), 3.82-3.98 (m, 4 H), 3.67 (s, 3 H), 3.50-3.54 (m, 8H), 3.08 (s, 3 H), 2.12 (s, 6 H), 1.95-2.05 (m., 2 H), 1.24-1.20 (m, 6 H). LCMS ( m/z ): 892.7 [M] + .
实施例12Example 12
(2E,2'E)-1,1'-(戊烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺)(2E,2'E)-1,1'-(Pentane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide)
步骤1: 4,4'-(戊烷-1,4-二基双(氮烷二基))双(3-硝基苯甲酰胺)Step 1: 4,4'-(pentane-1,4-diylbis(azanediyl))bis(3-nitrobenzamide)
向反应烧瓶中放入戊烷-1,4-二胺, 2盐酸盐 (1 g, 5.71 mmol)和异丙醇 (9.52mL)。向这个溶液加入4-氟-3-硝基苯甲酰胺 (1.052 g, 5.71 mmol),然后加入DIPEA(4.49 mL, 25.7 mmol)。将该烧瓶盖上盖子并将该反应加热至105℃。在4小时后,加入更多的4-氟-3-硝基苯甲酰胺 (1.052 g, 5.71 mmol)和异丙醇 (10 mL)。在105℃搅拌该混合物过夜(~14 h)。过滤掉形成的沉淀物并用异丙醇漂洗两次(每次5 mL)。获得作为黄色固体的4,4'-(戊烷-1,4-二基双(氮烷二基))双(3-硝基苯甲酰胺) (2.6 g, 5.80 mmol, 100%收率)。LCMS (m/z): 431.3 [M + H]+。Into a reaction flask was placed pentane-1,4-diamine, 2-hydrochloride (1 g, 5.71 mmol) and isopropanol (9.52 mL). To this solution was added 4-fluoro-3-nitrobenzamide (1.052 g, 5.71 mmol) followed by DIPEA (4.49 mL, 25.7 mmol). The flask was capped and the reaction was heated to 105 °C. After 4 hours, more 4-fluoro-3-nitrobenzamide (1.052 g, 5.71 mmol) and isopropanol (10 mL) were added. The mixture was stirred at 105 °C overnight (~14 h). The formed precipitate was filtered off and rinsed twice with isopropanol (5 mL each). 4,4'-(Pentane-1,4-diylbis(azanediyl))bis(3-nitrobenzamide) (2.6 g, 5.80 mmol, 100% yield) was obtained as a yellow solid. LCMS ( m/z ): 431.3 [M + H] + .
步骤2: 4,4'-(戊烷-1,4-二基双(氮烷二基))双(3-氨基苯甲酰胺)Step 2: 4,4'-(pentane-1,4-diylbis(azanediyl))bis(3-aminobenzamide)
向100-mL 圆底烧瓶加入4,4'-(戊烷-1,4-二基双(氮烷二基))双(3-硝基苯甲酰胺) (500 mg, 1.162 mmol)和MeOH (11.6 mL)。向这个溶液加入氯化铵 (249 mg, 4.65mmol)和5.5 mL饱和氯化铵水溶液。向这个溶液加入锌(759 mg, 11.62 mmol)。在室温搅拌该非均相混合物15 min。过滤该混合物并采用MeOH (10 mL)漂洗所收集的固体。向合并的滤液加入硅藻土,并通过快速层析法 (干装载技术, 12 g SiO2 筒, 2-40% MeOH/DCM作为洗脱液,含 NH4OH作为改性剂)纯化粗产物。将相应的级分合并并浓缩。获得作为无色油的4,4'-(戊烷-1,4-二基双(氮烷二基))双(3-氨基苯甲酰胺) (368 mg, 0.944 mmol, 81%收率)。LCMS (m/z): 371.2 [M + H]+。To a 100-mL round bottom flask was added 4,4'-(pentane-1,4-diylbis(azanediyl))bis(3-nitrobenzamide) (500 mg, 1.162 mmol) and MeOH (11.6 mL). To this solution was added ammonium chloride (249 mg, 4.65 mmol) and 5.5 mL of saturated aqueous ammonium chloride solution. To this solution was added zinc (759 mg, 11.62 mmol). The heterogeneous mixture was stirred at room temperature for 15 min. The mixture was filtered and the collected solids were rinsed with MeOH (10 mL). Celite was added to the combined filtrates and the crude product was purified by flash chromatography (dry loading technique, 12 g SiO2 cartridge , 2-40% MeOH/DCM as eluent, with NH4OH as modifier). The corresponding fractions were combined and concentrated. 4,4'-(pentane-1,4-diylbis(azanediyl))bis(3-aminobenzamide) (368 mg, 0.944 mmol, 81% yield) was obtained as a colorless oil. LCMS ( m/z ): 371.2 [M + H] + .
步骤3: 1,1'-(戊烷-1,4-二基)双(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺)Step 3: 1,1'-(Pentane-1,4-diyl)bis(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide)
向100-mL 圆底烧瓶加入4,4'-(戊烷-1,4-二基双(氮烷二基))双(3-氨基苯甲酰胺) (368 mg, 0.993 mmol)和DMF (9.9 mL)。将该溶液冷却至0℃。在0℃搅拌5 min后,加入1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (3 mL 的~0.4 M 二氧杂环己烷溶液, ~1.2 mmol)。在15 min后,加入更多的1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (1 mL的~0.4 M 二氧杂环己烷溶液, ~0.4 mmol)。使该反应在0℃再搅拌15 min。然后,加入EDC(476 mg, 2.483 mmol)和三乙胺 (0.692 mL, 4.97 mmol)。使该反应混合物温热至室温并使其搅拌过夜(~14 h)。将该反应混合物倾倒至4:1 水/饱和氯化铵水溶液 (25 mL)中。用乙酸乙酯 (3 x15 mL)萃取产物。用水(20 mL)、盐水(20 mL)洗涤合并的有机相并经硫酸镁干燥。将粗物质浓缩并通过快速层析法 (24 g SiO2 筒, 2-40% MeOH/DCM作为洗脱液,含NH4OH作为改性剂)纯化。将相应的级分合并并浓缩。获得作为白色粉末的1,1'-(戊烷-1,4-二基)双(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺) (303mg, 0.429 mmol, 43.1%收率)。1H NMR (400 MHz, DMSO-d 6 ) δ ppm 12.88 (br. s, 1 H),12.80 (br. s, 1 H), 7.95 - 7.99 (m, 4 H), 7.70 - 7.73 (m, 2 H), 7.61 (d, J=8.62 Hz, 1 H), 7.34 - 7.42 (m, 3 H), 6.61 (m, 2 H), 5.20 (br. s., 1 H), 4.53- 4.60 (m, 4 H), 4.32 (br. s, 1 H), 4.11 - 4.16 (m, 1 H), 2.34 (br. s, 1 H),2.10 (s, 3 H), 2.09 (s, 3 H), 1.92 (m, 1 H), 1.70 (m, 1 H), 1.61 (m, 1 H),1.52 (d, J=6.84 Hz, 3 H), 1.28 - 1.34 (m, 6 H); LCMS (m/z): 693.6 [M + H]+。To a 100-mL round bottom flask was added 4,4'-(pentane-1,4-diylbis(azanediyl))bis(3-aminobenzamide) (368 mg, 0.993 mmol) and DMF (9.9 mL). The solution was cooled to 0°C. After stirring at 0°C for 5 min, 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (3 mL of a ~0.4 M solution in dioxane, ~1.2 mmol) was added. After 15 min, more 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (1 mL of a ~0.4 M solution in dioxane, ~0.4 mmol) was added. The reaction was stirred at 0°C for an additional 15 min. Then, EDC (476 mg, 2.483 mmol) and triethylamine (0.692 mL, 4.97 mmol) were added. The reaction mixture was allowed to warm to room temperature and stirred overnight (~14 h). The reaction mixture was poured into 4:1 water/saturated aqueous ammonium chloride solution (25 mL). The product was extracted with ethyl acetate (3 x15 mL). The combined organic phase was washed with water (20 mL), brine (20 mL) and dried over magnesium sulfate. The crude material was concentrated and purified by flash chromatography (24 g SiO 2 cartridges, 2-40% MeOH/DCM as eluent, containing NH 4 OH as a modifier). The corresponding fractions were combined and concentrated. 1,1'-(pentane-1,4-diyl)bis(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide) (303 mg, 0.429 mmol, 43.1% yield) was obtained as a white powder. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 12.88 (br. s, 1 H), 12.80 (br. s, 1 H), 7.95 - 7.99 (m, 4 H), 7.70 - 7.73 (m, 2 H), 7.61 (d, J =8.62 Hz, 1 H), 7.34 - 7.42 (m, 3 H), 6.61 (m, 2 H), 5.20 (br. s., 1 H), 4.53- 4.60 (m, 4 H), 4.32 (br. s, 1 H), 4.11 - 4.16 (m, 1 H), 2.34 (br. s, 1 H), 2.10 (s, 3 H) , 2.09 (s, 3 H), 1.92 (m, 1 H), 1.70 (m, 1 H), 1.61 (m, 1 H), 1.52 (d, J =6.84 Hz, 3 H), 1.28 - 1.34 (m, 6 H); LCMS (m/z): 693.6 [M + H] + .
步骤4: (2E,2'E)-1,1'-(戊烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺)Step 4: (2E,2'E)-1,1'-(Pentane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide)
向10-mL小瓶放入1,1'-(戊烷-1,4-二基)双(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺) (50 mg, 0.072 mmol)和DMF (1.4 mL)。向非均相溶液加入碳酸铯 (70.5 mg, 0.217 mmol),然后加入碘甲烷 (0.011 mL, 0.180 mmol)。将该小瓶盖上盖子并在室温搅拌该混合物过夜(~14 h)。用DMSO (1 mL)和(1 mL)稀释该混合物以形成透明均相溶液。通过反相制备型HPLC (双相 ISCO系统, 5-35%梯度的MeCN/水,含0.1%NH4OH 改性剂)直接纯化这个溶液。将相应的级分合并并浓缩。采用MeCN和水(~30 mL)冻干产物。获得作为白色蓬松固体的(2E,2'E)-1,1'-(戊烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺) (25 mg, 0.035mmol, 48.1%收率)。1H NMR (400 MHz, 甲醇-d 4 ) δ ppm 7.87 (d, J=2.28 Hz, 2 H),7.82 (dd, J=8.36, 1.52 Hz, 1 H), 7.73 - 7.80 (m, 1 H), 7.54 (d, J=8.36 Hz, 1H), 7.41 (d, J=8.62 Hz, 1 H), 6.63 (s, 2 H), 4.85 (m, 1 H), 4.63 (q, J=7.18Hz, 4 H), 4.25 - 4.41 (m, 1 H), 4.06 - 4.20 (m, 1 H), 3.57 (s, 3 H), 3.55 (s,3 H), 2.25 (s, 6 H), 2.23 (m, 1 H), 1.81 - 2.03 (m, 2 H), 1.68 (m., 1 H),1.58 (d, J=6.84 Hz, 3 H), 1.39 (t, J=7.10 Hz, 6 H)。LCMS (m/z): 721.6 [M + H]+。Into a 10-mL vial was placed 1,1'-(pentane-1,4-diyl)bis(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide) (50 mg, 0.072 mmol) and DMF (1.4 mL). To the heterogeneous solution was added cesium carbonate (70.5 mg, 0.217 mmol) followed by iodomethane (0.011 mL, 0.180 mmol). The vial was capped and the mixture was stirred at room temperature overnight (~14 h). The mixture was diluted with DMSO (1 mL) and (1 mL) to form a clear homogeneous solution. This solution was directly purified by reverse phase preparative HPLC (biphasic ISCO system, 5-35% gradient of MeCN/water with 0.1% NH 4 OH modifier). The corresponding fractions were combined and concentrated. The product was lyophilized with MeCN and water (~30 mL). (2E,2'E)-1,1'-(pentane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide) was obtained as a white fluffy solid (25 mg, 0.035 mmol, 48.1% yield). 1 H NMR (400 MHz, methanol- d 4 ) δ ppm 7.87 (d, J =2.28 Hz, 2 H), 7.82 (dd, J =8.36, 1.52 Hz, 1 H), 7.73 - 7.80 (m, 1 H), 7.54 (d, J =8.36 Hz, 1H), 7.41 (d, J =8.62 Hz, 1 H), 6.63 (s, 2 H), 4.85 (m, 1 H), 4.63 (q, J =7.18Hz, 4 H), 4.25 - 4.41 (m, 1 H), 4.06 - 4.20 (m, 1 H), 3.57 (s, 3 H), 3.55 (s, 3 H), 2.25 (s, 6 H), 2.23 (m, 1 H), 1.81 - 2.03 (m, 2 H), 1.68 (m., 1 H), 1.58 (d, J =6.84 Hz, 3 H), 1.39 (t, J =7.10 Hz, 6 H). LCMS (m/z): 721.6 [M + H] + .
实施例13Example 13
(2E,2'E)-1,1'-(4-甲基戊烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺)(2E,2'E)-1,1'-(4-methylpentane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide)
步骤1: 4,4'-((4-甲基戊烷-1,4-二基)双(氮烷二基))双(3-硝基苯甲酰胺)Step 1: 4,4'-((4-methylpentane-1,4-diyl)bis(azanediyl))bis(3-nitrobenzamide)
向24-mL小瓶放入4-甲基戊烷-1,4-二胺 (0.49 g, 4.22 mmol)和异丙醇 (14.0mL)。向这个溶液加入4-氟-3-硝基苯甲酰胺 (1.63 g, 8.85 mmol),然后加入DIPEA (2.58ml, 14.76 mmol)。将该小瓶盖上盖子并在105℃搅拌非均相溶液过夜(~14 h)。过滤掉沉淀物并用异丙醇 (2 x 5 mL)漂洗。获得作为橙色固体的4,4'-((4-甲基戊烷-1,4-二基)双(氮烷二基))双(3-硝基苯甲酰胺) (1.46 g, 3.02 mmol, 71.7%收率, 92%纯度)。LCMS(m/z): 445.3 [M + H]+。Into a 24-mL vial was placed 4-methylpentane-1,4-diamine (0.49 g, 4.22 mmol) and isopropanol (14.0 mL). To this solution was added 4-fluoro-3-nitrobenzamide (1.63 g, 8.85 mmol) followed by DIPEA (2.58 ml, 14.76 mmol). The vial was capped and the heterogeneous solution was stirred at 105 °C overnight (~14 h). The precipitate was filtered off and rinsed with isopropanol (2 x 5 mL). 4,4'-((4-methylpentane-1,4-diyl)bis(azanediyl))bis(3-nitrobenzamide) (1.46 g, 3.02 mmol, 71.7% yield, 92% purity) was obtained as an orange solid. LCMS ( m/z ): 445.3 [M + H] + .
步骤2: 4,4'-((4-甲基戊烷-1,4-二基)双(氮烷二基))双(3-氨基苯甲酰胺)Step 2: 4,4'-((4-methylpentane-1,4-diyl)bis(azanediyl))bis(3-aminobenzamide)
向100-mL 圆底烧瓶加入4,4'-((4-甲基戊烷-1,4-二基)双(氮烷二基))双(3-硝基苯甲酰胺) (500 mg, 1.035 mmol)和MeOH (15 mL)。向这个溶液加入氯化铵 (1107 mg,20.70 mmol)、10 mL 饱和氯化铵水溶液和锌(677 mg, 10.35 mmol)。在室温搅拌非均相混合物。在20 min后,加入更多的锌(350 mg, 5.35 mmol)和氯化铵 (600 mg, 11.22 mmol)。在室温总共搅拌90 min后,过滤该混合物。用MeOH (20 mL)漂洗剩余的固体。向合并的滤液加入硅藻土,并通过 硅胶层析法 (干装载技术, 12 g SiO2 筒, 2-40%梯度的MeOH/DCM,含NH4OH作为改性剂)纯化粗产物。将相应的级分合并并浓缩。获得作为白色膜的4,4'-((4-甲基戊烷-1,4-二基)双(氮烷二基))双(3-氨基苯甲酰胺) (247 mg, 0.610 mmol, 59.0%收率)。LCMS (m/z): 385.4 [M + H]+。To a 100-mL round bottom flask was added 4,4'-((4-methylpentane-1,4-diyl)bis(azanediyl))bis(3-nitrobenzamide) (500 mg, 1.035 mmol) and MeOH (15 mL). To this solution was added ammonium chloride (1107 mg, 20.70 mmol), 10 mL of saturated aqueous ammonium chloride, and zinc (677 mg, 10.35 mmol). The heterogeneous mixture was stirred at room temperature. After 20 min, more zinc (350 mg, 5.35 mmol) and ammonium chloride (600 mg, 11.22 mmol) were added. After stirring for a total of 90 min at room temperature, the mixture was filtered. The remaining solid was rinsed with MeOH (20 mL). Celite was added to the combined filtrate and the crude product was purified by silica gel chromatography (dry loading technique, 12 g SiO2 cartridge , 2-40% gradient of MeOH/DCM with NH4OH as modifier). The corresponding fractions were combined and concentrated. 4,4'-((4-methylpentane-1,4-diyl)bis(azanediyl))bis(3-aminobenzamide) (247 mg, 0.610 mmol, 59.0% yield) was obtained as a white film. LCMS ( m/z ): 385.4 [M+H] + .
步骤3: 1,1'-(4-甲基戊烷-1,4-二基)双(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺)Step 3: 1,1'-(4-methylpentane-1,4-diyl)bis(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide)
向100-mL 圆底烧瓶加入4,4'-((4-甲基戊烷-1,4-二基)双(氮烷二基))双(3-氨基苯甲酰胺) (247 mg, 0.642 mmol)和DMF (6.42 mL)。将这个溶液冷却至0℃。在0℃搅拌5 min后,加入作为溶液的1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 ( 在二氧杂环己烷中~0.4 M, 2.5 mL; ~1.0 mmol)。在20 min后,加入更多的1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (在二氧杂环己烷中~0.4 M, 0.45 mL; 0.18 mmol)以确保硫脲形成完全。在0℃搅拌另外的20 min后,加入EDC (308 mg, 1.606 mmol),然后加入三乙胺 (0.448mL, 3.21 mmol)。使反应混合物升温至室温并搅拌过夜(~14 h)。将该反应倾倒至容纳25mL的 3:1水/饱和氯化铵水溶液的烧杯中并搅拌10 min。过滤掉所得白色沉淀物并用水(3x 5 mL)漂洗。在50℃在真空炉中干燥该固体6 h。获得作为灰白色固体的1,1'-(4-甲基戊烷-1,4-二基)双(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺)(341 mg, 0.473 mmol, 73.6%收率)。1H NMR (400 MHz, DMSO-d 6 ) δ ppm 7.93 (m, 3 H),7.84 (s, 1 H), 7.73 (d, J=8.87 Hz, 1 H), 7.67 (d, J=8.36 Hz, 1 H), 7.60 (d, J=8.62 Hz, 1 H), 7.24 - 7.44 (m, 2 H), 6.40 (s, 1 H), 6.25 (br. s., 1 H), 4.39- 4.64 (m, 4 H), 4.11 (t, J=6.46 Hz, 2 H), 2.44 (br. s., 2 H), 2.10 (s, 3 H),2.07 (s, 3 H), 1.90 (br. s., 6 H), 1.68 (br. s., 2 H), 1.31 (t, J=7.10 Hz, 3H), 1.24 (t, J=7.10 Hz, 3 H)。LCMS (m/z): 707.6 [M + H]+。To a 100-mL round-bottom flask was added 4,4'-((4-methylpentane-1,4-diyl)bis(azanediyl))bis(3-aminobenzamide) (247 mg, 0.642 mmol) and DMF (6.42 mL). This solution was cooled to 0°C. After stirring at 0°C for 5 min, 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (~0.4 M in dioxane, 2.5 mL; ~1.0 mmol) was added as a solution. After 20 min, more 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (~0.4 M in dioxane, 0.45 mL; 0.18 mmol) was added to ensure complete thiourea formation. After stirring at 0 °C for an additional 20 min, EDC (308 mg, 1.606 mmol) was added followed by triethylamine (0.448 mL, 3.21 mmol). The reaction mixture was allowed to warm to room temperature and stirred overnight (~14 h). The reaction was poured into a beaker containing 25 mL of 3:1 water/saturated aqueous ammonium chloride solution and stirred for 10 min. The resulting white precipitate was filtered off and rinsed with water (3x 5 mL). The solid was dried in a vacuum oven at 50 °C for 6 h. 1,1'-(4-methylpentane-1,4-diyl)bis(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide) (341 mg, 0.473 mmol, 73.6% yield) was obtained as an off-white solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 7.93 (m, 3 H), 7.84 (s, 1 H), 7.73 (d, J =8.87 Hz, 1 H), 7.67 (d, J =8.36 Hz, 1 H), 7.60 (d, J =8.62 Hz, 1 H), 7.24 - 7 .44 (m, 2 H), 6.40 (s, 1 H), 6.25 (br. s., 1 H), 4.39- 4.64 (m, 4 H), 4.11 (t, J =6.46 Hz, 2 H), 2.44 (br. s., 2 H), 2.10 (s, 3 H), 2.07 (s, 3 H), 1.90 (br. s., 6 H), 1.68 (br. s., 2 H), 1.31 (t, J =7.10 Hz, 3H), 1.24 (t, J =7.10 Hz, 3 H). LCMS ( m/z ): 707.6 [M + H] + .
步骤4: (2E,2'E)-1,1'-(4-甲基戊烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺)Step 4: (2E,2'E)-1,1'-(4-methylpentane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide)
向10-mL小瓶放入1,1'-(4-甲基戊烷-1,4-二基)双(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺) (49 mg, 0.069 mmol)和DMF (0.693 mL)。向这个溶液加入碳酸铯 (67.8 mg, 0.208 mmol)然后加入碘甲烷 (10.84 µL, 0.173 mmol)。将该小瓶盖上盖子并在室温搅拌该混合物过夜(~14 h)。用DMSO (1 mL)和水(1 mL)稀释该混合物以形成透明均相溶液。通过反相制备型HPLC (双相 ISCO系统, 5-35%梯度的MeCN/水,含0.1% NH4OH 改性剂)将这个溶液直接注射并纯化。将相应的级分合并并浓缩。获得作为透明油的(2E,2'E)-1,1'-(4-甲基戊烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺) (15.8 mg, 0.022 mmol,31.0%收率)。1H NMR (400 MHz, 甲醇-d 4 ) δ ppm 7.64 - 7.85 (m, 5 H), 7.34 (d, J=8.36 Hz, 1 H), 6.65 (s, 1 H), 6.57 (s, 1 H), 4.64 (m, 4 H), 4.20 (t, J=5.96Hz, 2 H), 3.54 (s, 3 H), 3.40 (s, 3 H), 2.24 - 2.31 (m, 5 H), 2.23 (s, 3 H),1.82 - 1.95 (m, 8 H), 1.41 (m, 6 H)。LCMS (m/z): 735.4 [M + H]+。Into a 10-mL vial was placed 1,1'-(4-methylpentane-1,4-diyl)bis(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide) (49 mg, 0.069 mmol) and DMF (0.693 mL). To this solution was added cesium carbonate (67.8 mg, 0.208 mmol) followed by iodomethane (10.84 µL, 0.173 mmol). The vial was capped and the mixture was stirred at room temperature overnight (~14 h). The mixture was diluted with DMSO (1 mL) and water (1 mL) to form a clear homogeneous solution. This solution was injected directly and purified by reverse phase preparative HPLC (biphasic ISCO system, 5-35% gradient of MeCN/water with 0.1% NH 4 OH modifier). The corresponding fractions were combined and concentrated. (2E,2'E)-1,1'-(4-methylpentane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide) was obtained as a clear oil (15.8 mg, 0.022 mmol, 31.0% yield). 1 H NMR (400 MHz, methanol- d 4 ) δ ppm 7.64 - 7.85 (m, 5 H), 7.34 (d, J =8.36 Hz, 1 H), 6.65 (s, 1 H), 6.57 (s, 1 H), 4.64 (m, 4 H), 4.20 (t, J =5.96Hz, 2 H), 3.54 (s, 3 H), 3.40 (s, 3 H), 2.24 - 2.31 (m, 5 H), 2.23 (s, 3 H), 1.82 - 1.95 (m, 8 H), 1.41 (m, 6 H). LCMS ( m/z ): 735.4 [M + H] + .
实施例14Embodiment 14
(E)-1-(5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2-甲基戊烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-N,3-二甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-(5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2-methylpentan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-N,3-dimethyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向10-mL小瓶加入1,1'-(4-甲基戊烷-1,4-二基)双(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺) (实施例13的合成中间体) (100 mg, 0.141mmol)和DMF (1.415 mL)。向这个溶液加入碳酸铯 (138 mg, 0.424 mmol)然后加入碘甲烷(0.044 mL, 0.707 mmol)。将该小瓶盖上盖子并在室温搅拌该混合物过夜(~14 h)。通过反相制备型HPLC (双相 ISCO系统, MeCN/水的梯度,含0.1% NH4OH 改性剂)直接纯化这个混合物。将相应的级分合并并浓缩。获得作为白色膜的(E)-1-(5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2-甲基戊烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-N,3-二甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (7.1 mg, 0.0087 mmol, 6.16%收率)。1H NMR (400 MHz, 甲醇-d 4) δ ppm 7.56 - 7.85 (m, 5 H), 7.25 - 7.37 (m, 1 H), 6.66 (s, 1 H), 6.58 (s,1 H), 4.54 - 4.76 (m, 4 H), 4.20 (t, J=5.96 Hz, 2 H), 3.55 (d, J=5.58 Hz, 3H), 3.40 (s, 3 H), 3.02 (s, 3 H), 2.26 (s, 3 H), 2.26 (m, 1 H), 2.23 (s, 3H), 2.23 (m, 1 H), 1.87 (s, 6 H), 1.87 (m, 2 H), 1.42 (m, 6 H)。LCMS (m/z):749.4 [M + H]+。To a 10-mL vial was added 1,1'-(4-methylpentane-1,4-diyl)bis(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide) (synthetic intermediate of Example 13) (100 mg, 0.141 mmol) and DMF (1.415 mL). To this solution was added cesium carbonate (138 mg, 0.424 mmol) followed by iodomethane (0.044 mL, 0.707 mmol). The vial was capped and the mixture was stirred at room temperature overnight (~14 h). This mixture was directly purified by reverse phase preparative HPLC (biphasic ISCO system, gradient of MeCN/water with 0.1% NH4OH modifier). The corresponding fractions were combined and concentrated. (E)-1-(5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2-methylpentan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-N,3-dimethyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (7.1 mg, 0.0087 mmol, 6.16% yield) was obtained as a white film. 1 H NMR (400 MHz, methanol- d 4 ) δ ppm 7.56 - 7.85 (m, 5 H), 7.25 - 7.37 (m, 1 H), 6.66 (s, 1 H), 6.58 (s, 1 H), 4.54 - 4.76 (m, 4 H), 4.20 (t, J =5.96 Hz , 2 H), 3.55 (d, J =5.58 Hz, 3H), 3.40 (s, 3 H), 3.02 (s, 3 H), 2.26 (s, 3 H), 2.26 (m, 1 H), 2.23 (s, 3H), 2.23 (m, 1 H), 1.87 (s, 6 H), 1.87 (m, 2 H), 1.42 (m, 6 H). LCMS ( m/z ):749.4 [M + H] + .
实施例15Embodiment 15
(E)-1-(5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (非对映异构体1)(E)-1-(5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (Diastereomer 1)
步骤1: (E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(-5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 1: (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(-5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
向10-mL小瓶放入7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(5-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (25 mg, 0.028mmol, 例如中间体14B, 第二个洗脱出的非对映异构体)和DMF (559 µL)。向这个非均相溶液加入碳酸铯 (27.3 mg, 0.084 mmol),然后加入碘甲烷 (4.37 µL, 0.070 mmol)。将该小瓶盖上盖子并在室温搅拌该混合物过夜(~14 h)。用DMSO (1 mL)和水(1 mL)稀释该混合物以形成透明均相溶液。通过反相制备型HPLC (双相 ISCO系统, 3-35%梯度的MeCN/水,含0.1% NH4OH 改性剂)将这个溶液直接注射并纯化,将相应的级分合并并浓缩以提供作为白色固体的(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(-5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (18 mg, 0.019 mmol, 66.3%收率)。LCMS: 923.4 [M + H]+, 1.30min保留时间(Acquity UPLC CSH C18, 1.7 um, 50 mm x 2.1 mm柱; 经1.5 min的3-95%梯度, MeCN/10 mM碳酸氢铵在水中的溶液,其采用25% 氢氧化铵溶液调节至pH 10)。Into a 10-mL vial was placed 7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(5-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (25 mg, 0.028 mmol, e.g. Intermediate 14B, second eluting diastereomer) and DMF (559 µL). To this heterogeneous solution was added cesium carbonate (27.3 mg, 0.084 mmol) followed by iodomethane (4.37 µL, 0.070 mmol). The vial was capped and the mixture was stirred at room temperature overnight (~14 h). The mixture was diluted with DMSO (1 mL) and water (1 mL) to form a clear homogeneous solution. This solution was injected directly and purified by reverse phase preparative HPLC (biphasic ISCO system, 3-35% gradient of MeCN/water with 0.1% NH4OH modifier) and the corresponding fractions were combined and concentrated to afford (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(-5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (18 mg, 0.019 mmol, 66.3% yield) as a white solid. LCMS: 923.4 [M + H] + , 1.30 min retention time (Acquity UPLC CSH C18, 1.7 um, 50 mm x 2.1 mm column; 3-95% gradient over 1.5 min, MeCN/10 mM ammonium bicarbonate in water adjusted to pH 10 with 25% ammonium hydroxide solution).
步骤2: (E)-1-(5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 2: (E)-1-(5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向20-mL小瓶中放入(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(-5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (25 mg, 0.027 mmol)和MeOH (2 mL)。向这个溶液加入HCl (3 M CPME溶液, 300 µL, 0.900 mmol)。在室温搅拌该反应混合物过夜。在N2排污单位(N2 blow down unit)下除去挥发物。通过反相制备型HPLC (双相 ISCO系统,20-50%梯度的MeCN/水,含0.1% NH4OH 改性剂)纯化粗产物。将相应的级分合并并浓缩以提供作为玻璃状白色固体的(E)-1-(5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (5.5 mg, 6.46 µmol, 23.85%收率)。1H NMR (400 MHz, 甲醇-d 4) δ ppm 8.01(s, 1.17) 7.88 (br. s., 3.21) 7.49 - 7.73 (m, 8.82) 7.42 (br. s., 2.89) 7.29(br. s., 0.86) 7.20 (s, 2.93) 6.70 (s, 3.36) 6.64 (br. s., 2.95) 4.98 - 5.11(m, 4.53) 4.81 (br. s., 3.36) 4.55 - 4.75 (m, 14.25) 4.29 - 4.55 (m, 3.26)4.08 (m, 2.84) 3.82 (m, 2.33) 3.41 - 3.75 (m, 28.77) 3.23 (br. s., 4.09) 2.19- 2.36 (m, 21.72) 2.00 - 2.18 (m, 11.47) 1.76 - 2.00 (m, 13.31) 1.54 (m,23.37) 1.35 - 1.50 (m, 24.00); LCMS: 809.5 [M + H]+, 0.74 min保留时间(AcquityUPLC CSH C18 50 mm x 2.1 mm柱, 1.7 um; 经1.5 min的5-95%梯度; MeCN/水,含0.1%TFA 改性剂)。Into a 20-mL vial was placed (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(-5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (25 mg, 0.027 mmol) and MeOH (2 mL). To this solution was added HCl (3 M in CPME, 300 µL, 0.900 mmol). The reaction mixture was stirred at room temperature overnight. The volatiles were removed under a N 2 blow down unit.The crude product was purified by reverse phase preparative HPLC (biphasic ISCO system, 20-50% gradient of MeCN/water with 0.1% NH 4 OH modifier). The corresponding fractions were combined and concentrated to afford (E)-1-(5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (5.5 mg, 6.46 µmol, 23.85% yield) as a glassy white solid. 1 H NMR (400 MHz, methanol- d 4 ) δ ppm 8.01 (s, 1.17) 7.88 (br. s., 3.21) 7.49 - 7.73 (m, 8.82) 7.42 (br. s., 2.89) 7.29 (br. s., 0.86) 7.20 (s, 2 .93) 6.70 (s, 3.36) 6.64 (br. s., 2.95) 4.98 - 5.11(m, 4.53) 4.81 (br. s., 3.36) 4.55 - 4.75 (m, 14.25) 4.29 - 4.55 (m, 3.26)4.08 (m, s., 4.09) 2.19 - 2.36 (m, 21.72) 2.00 - 2.18 (m, 11.47) 1.76 - 2.00 (m, 13.31) 1.54 (m,23.37) 1.35 - 1.50 (m, 24.00); LCMS: 809.5 [M + H] + , 0.74 min retention time (AcquityUPLC CSH C18 50 mm x 2.1 mm column, 1.7 um; 5-95% gradient over 1.5 min; MeCN/water with 0.1% TFA modifier).
实施例16Example 16
(E)-1-(5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (非对映异构体2)(E)-1-(5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (Diastereomer 2)
步骤1: (E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 1: (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
向10-mL小瓶放入7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(5-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 ( 25 mg, 0.028mmol, 例如中间体14A, 第一个洗脱出的非对映异构体)和DMF (559 µL)。向这个非均相溶液加入碳酸铯 (27.3 mg, 0.084 mmol)和碘甲烷 (4.37 µl, 0.070 mmol)。将该小瓶盖上盖子并在室温搅拌该混合物过夜(~14 h)。用DMSO (1 mL)和水(1 mL)稀释该混合物以形成透明均相溶液。通过反相制备型HPLC (双相 ISCO系统, 5-35%梯度的MeCN/水,含0.1%NH4OH 改性剂) 将这个溶液直接注射并纯化。将相应的级分合并并浓缩以提供作为白色固体的(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (15 mg, 0.014 mmol, 49.5 %收率, 85%纯度)。LCMS: 923.5 [M + H]+,1.24 min保留时间(Acquity UPLC CSH C18, 1.7 um, 50 mm x 2.1 mm柱; 经1.5 min的3-95%梯度, MeCN/10 mM在水中的碳酸氢铵,采用25% 氢氧化铵溶液调节至pH 10)。Into a 10-mL vial was placed 7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(5-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (25 mg, 0.028 mmol, e.g. Intermediate 14A, first eluting diastereomer) and DMF (559 µL). To this heterogeneous solution was added cesium carbonate (27.3 mg, 0.084 mmol) and iodomethane (4.37 µl, 0.070 mmol). The vial was capped and the mixture was stirred at room temperature overnight (~14 h). The mixture was diluted with DMSO (1 mL) and water (1 mL) to form a clear homogeneous solution. This solution was directly injected and purified by reverse phase preparative HPLC (biphasic ISCO system, 5-35% gradient of MeCN/water with 0.1% NH 4 OH modifier). The corresponding fractions were combined and concentrated to afford (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (15 mg, 0.014 mmol, 49.5 % yield, 85 % purity) as a white solid. LCMS: 923.5 [M + H] + ,1.24 min retention time (Acquity UPLC CSH C18, 1.7 um, 50 mm x 2.1 mm column; 3-95% gradient over 1.5 min, MeCN/10 mM ammonium bicarbonate in water, adjusted to pH 10 with 25% ammonium hydroxide solution).
步骤2: (E)-1-(5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 2: (E)-1-(5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向20-mL小瓶中放入(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(-5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (15 mg, 0.016 mmol)和MeOH (2 mL)。向这个溶液加入HCl (3 M CPME溶液, 300 µL, 0.900 mmol)。将该反应混合物在室温搅拌过夜。使用氮气流除去挥发物。通过反相制备型HPLC (双相 ISCO系统; 20-50%梯度的MeCN/水,含0.1% NH4OH 改性剂)纯化残余物。将相应的级分合并并浓缩以提供作为玻璃状白色固体的外消旋(E)-1-(5-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)己烷-2-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (5 mg,5.87 µmol, 36.1 %收率)。1H NMR (400 MHz, 甲醇-d 4) δ ppm 8.07 (m, 2.61), 7.86(m, 2.78), 7.58 - 7.81 (m, 4.92), 7.52 (s, 2.38), 6.48 - 6.73 (m, 4.98), 5.47(br. s., 1.43), 4.75 (br. s., 2.43), 4.49 - 4.69 (m, 10.33), 4.26 - 4.40 (m,4.06), 4.21 (br. s., 1.08), 3.50 - 3.76 (m, 20.52), 2.68 (s, 1.17), 2.49 (br.s., 0.84), 2.15 - 2.41 (m, 18.94), 1.94 - 2.07 (m, 2.86), 1.78 - 1.94 (m,3.63), 1.70 (m., 3.14), 1.46 - 1.63 (m, 16.31), 1.26 - 1.42 (m, 17.00)。LCMS:809.5 [M + H]+, 0.71 min保留时间(Acquity UPLC CSH C18, 1.7 um, 50 mm x 2.1 mm柱; 经1.5 min的5-95%梯度; MeCN/水,含0.1% TFA 改性剂)。Into a 20-mL vial was placed (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(-5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (15 mg, 0.016 mmol) and MeOH (2 mL). To this solution was added HCl (3 M in CPME, 300 µL, 0.900 mmol). The reaction mixture was stirred at room temperature overnight. Volatiles were removed using a stream of nitrogen. The residue was purified by reverse phase preparative HPLC (biphasic ISCO system; 20-50% gradient of MeCN/water with 0.1% NH4OH modifier). The corresponding fractions were combined and concentrated to afford racemic (E)-1-(5-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)hexan-2-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (5 mg, 5.87 μmol, 36.1% yield) as a glassy white solid. 1 H NMR (400 MHz, methanol- d 4 ) δ ppm 8.07 (m, 2.61), 7.86 (m, 2.78), 7.58 - 7.81 (m, 4.92), 7.52 (s, 2.38), 6.48 - 6.73 (m, 4.98), 5.47 (br. s. , 1.43), 4.75 (br. s., 2.43), 4.49 - 4.69 (m, 10.33), 4.26 - 4.40 (m, 4.06), 4.21 (br. s., 1.08), 3.50 - 3.76 (m, 20.52), 2.68 (s, 1.17) , 2.49 (br.s., 0.84), 2.15 - 2.41 (m, 18.94), 1.94 - 2.07 (m, 2.86), 1.78 - 1.94 (m,3.63), 1.70 (m., 3.14), 1.46 - 1.63 (m, 16.31), 1.26 - 1.42 (m, 17.00). LCMS: 809.5 [M + H] + , 0.71 min retention time (Acquity UPLC CSH C18, 1.7 um, 50 mm x 2.1 mm column; 5-95% gradient over 1.5 min; MeCN/water with 0.1% TFA modifier).
实施例17Embodiment 17
(E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺,2 三氟乙酸盐(E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide, 2-trifluoroacetate
步骤1: 3-(3-(苄氧基)丙氧基)-4-氯-5-硝基苯甲酰胺Step 1: 3-(3-(Benzyloxy)propoxy)-4-chloro-5-nitrobenzamide
在室温在氮气下在1 min期间向4-氯-3-羟基-5-硝基苯甲酰胺 (30 g, 139mmol)和碳酸钾(57.4 g, 416 mmol)在DMF (200 mL)中的搅拌悬浮液逐滴加入((3-溴丙氧基)甲基)苯(47.6 g, 208 mmol)的溶液。在80℃搅拌该反应混合物过夜。将该反应混合物冷却至室温并通过加入200 mL水淬灭。然后,用DCM (3x100 mL)萃取水相。用水(4x200 mL)和盐水(200 mL)洗涤合并的有机层,干燥,并在真空下浓缩。通过硅胶层析法 (100 g柱,1:2 石油醚/EtOAc)纯化粗产物。合并适当的级分并浓缩以提供作为黄色固体的3-(3-(苄氧基)丙氧基)-4-氯-5-硝基苯甲酰胺 (33 g, 90 mmol, 65.3%收率)。LCMS (m/z): 365[M + H]+。To a stirred suspension of 4-chloro-3-hydroxy-5-nitrobenzamide (30 g, 139 mmol) and potassium carbonate (57.4 g, 416 mmol) in DMF (200 mL) was added a solution of ((3-bromopropoxy)methyl)benzene (47.6 g, 208 mmol) dropwise at room temperature under nitrogen over 1 min. The reaction mixture was stirred at 80 °C overnight. The reaction mixture was cooled to room temperature and quenched by adding 200 mL of water. Then, the aqueous phase was extracted with DCM (3x100 mL). The combined organic layers were washed with water (4x200 mL) and brine (200 mL), dried, and concentrated under vacuum. The crude product was purified by silica gel chromatography (100 g column, 1:2 petroleum ether/EtOAc). Appropriate fractions were combined and concentrated to provide 3-(3-(benzyloxy)propoxy)-4-chloro-5-nitrobenzamide (33 g, 90 mmol, 65.3% yield) as a yellow solid. LCMS ( m/z ): 365 [M + H] + .
步骤2: 4-((4-氨基-2,2,3,3-四氟丁基)氨基)-3-(3-(苄氧基)丙氧基)-5-硝基苯甲酰胺Step 2: 4-((4-amino-2,2,3,3-tetrafluorobutyl)amino)-3-(3-(benzyloxy)propoxy)-5-nitrobenzamide
在135℃在密封管中搅拌2,2,3,3-四氟丁烷-1,4-二胺 (4 g, 24.98 mmol)、3-(3-(苄氧基)丙氧基)-4-氯-5-硝基苯甲酰胺 (4.56 g, 12.49 mmol)和DIPEA (6 mL,34.4 mmol)在异丙醇 (18 mL)中的悬浮液过夜。当冷却时,在真空中除去挥发物。通过硅胶层析法 (20 g 柱, 60-100%梯度的EtOAc/石油醚)纯化粗产物。合并适当的级分并浓缩以提供作为微红棕色油的4-((4-氨基-2,2,3,3-四氟丁基)氨基)-3-(3-(苄氧基)丙氧基)-5-硝基苯甲酰胺 (3 g, 6.14 mmol, 24.6%收率)。LCMS (m/z): 489 [M + H]+。A suspension of 2,2,3,3-tetrafluorobutane-1,4-diamine (4 g, 24.98 mmol), 3-(3-(benzyloxy)propoxy)-4-chloro-5-nitrobenzamide (4.56 g, 12.49 mmol) and DIPEA (6 mL, 34.4 mmol) in isopropanol (18 mL) was stirred overnight at 135 °C in a sealed tube. When cooled, the volatiles were removed in vacuo. The crude product was purified by silica gel chromatography (20 g column, 60-100% gradient of EtOAc/petroleum ether). The appropriate fractions were combined and concentrated to provide 4-((4-amino-2,2,3,3-tetrafluorobutyl)amino)-3-(3-(benzyloxy)propoxy)-5-nitrobenzamide (3 g, 6.14 mmol, 24.6% yield) as a reddish brown oil. LCMS ( m/z ): 489 [M + H] + .
步骤3: 3-(3-(苄氧基)丙氧基)-4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-硝基苯甲酰胺Step 3: 3-(3-(Benzyloxy)propoxy)-4-((4-((4-carbamoyl-2-nitrophenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-nitrobenzamide
在60℃在氮气下搅拌4-((4-氨基-2,2,3,3-四氟丁基)氨基)-3-(3-(苄氧基)丙氧基)-5-硝基苯甲酰胺 (3 g, 6.14 mmol), 4-氟-3-硝基苯甲酰胺 (1.696 g, 9.21 mmol)和碳酸钾 (1.698 g, 12.28 mmol)在DMF (30 mL)中的悬浮液过夜。当冷却时,加入水(50mL)并用EtOAc (2x100 mL)萃取水相。用水(3x200 mL)和盐水(200 mL)洗涤合并的有机层,干燥并在真空下浓缩。通过硅胶层析法 (10 g柱, 30-100%梯度的EtOAc/石油醚)纯化粗产物。合并适当的级分并浓缩以获得作为黄色固体的3-(3-(苄氧基)丙氧基)-4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-硝基苯甲酰胺 (2 g, 3.06mmol, 49.9%收率)。LCMS (m/z): 653 [M + H]+。A suspension of 4-((4-amino-2,2,3,3-tetrafluorobutyl)amino)-3-(3-(benzyloxy)propoxy)-5-nitrobenzamide (3 g, 6.14 mmol), 4-fluoro-3-nitrobenzamide (1.696 g, 9.21 mmol) and potassium carbonate (1.698 g, 12.28 mmol) in DMF (30 mL) was stirred overnight at 60 °C under nitrogen. When cooled, water (50 mL) was added and the aqueous phase was extracted with EtOAc (2x100 mL). The combined organic layers were washed with water (3x200 mL) and brine (200 mL), dried and concentrated under vacuum. The crude product was purified by silica gel chromatography (10 g column, 30-100% gradient of EtOAc/petroleum ether). Appropriate fractions were combined and concentrated to afford 3-(3-(benzyloxy)propoxy)-4-((4-((4-carbamoyl-2-nitrophenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-nitrobenzamide (2 g, 3.06 mmol, 49.9% yield) as a yellow solid. LCMS ( m/z ): 653 [M+H] + .
步骤4: 3-氨基-4-((4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-(3-(苄氧基)丙氧基)苯甲酰胺Step 4: 3-amino-4-((4-((2-amino-4-carbamoylphenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-(3-(benzyloxy)propoxy)benzamide
在氮气下向3-(3-(苄氧基)丙氧基)-4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-硝基苯甲酰胺 (1.9 g, 2.91 mmol)在乙酸(20 mL)中的搅拌悬浮液中一次性加入固体锌(1.904 g, 29.1 mmol)。在25℃搅拌该反应混合物2 h。然后,过滤该反应溶液,并在真空下浓缩滤液以提供作为棕色固体的3-氨基-4-((4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-(3-(苄氧基)丙氧基)苯甲酰胺 (1.5g, 2.53 mmol, 87%收率)。LCMS (m/z): 593 [M + H]+。To a stirred suspension of 3-(3-(benzyloxy)propoxy)-4-((4-((4-carbamoyl-2-nitrophenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-nitrobenzamide (1.9 g, 2.91 mmol) in acetic acid (20 mL) under nitrogen was added solid zinc (1.904 g, 29.1 mmol) in one portion. The reaction mixture was stirred at 25 °C for 2 h. The reaction solution was then filtered and the filtrate was concentrated under vacuum to provide 3-amino-4-((4-((2-amino-4-carbamoylphenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-(3-(benzyloxy)propoxy)benzamide (1.5 g, 2.53 mmol, 87% yield) as a brown solid. LCMS ( m/z ): 593 [M+H] + .
步骤5: 2-氨基-1-(4-(2-氨基-5-氨基甲酰基-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-7-(3-(苄氧基)丙氧基)-1H-苯并[d]咪唑-5-甲酰胺Step 5: 2-amino-1-(4-(2-amino-5-carbamoyl-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-7-(3-(benzyloxy)propoxy)-1H-benzo[d]imidazole-5-carboxamide
向3-氨基-4-((4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-(3-(苄氧基)丙氧基)苯甲酰胺 (2.1 g, 3.54 mmol)在MeOH (20 mL)中的溶液加入溴化氰 (1.126 g, 10.63 mmol)。在25℃搅拌该反应混合物16 h。采用二乙醚 (30 mL)稀释该混合物。过滤该混合物并用二乙醚洗涤滤饼。在减压下浓缩滤液以提供作为灰色固体的2-氨基-1-(4-(2-氨基-5-氨基甲酰基-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-7-(3-(苄氧基)丙氧基)-1H-苯并[d]咪唑-5-甲酰胺 (1.8 g, 2.381 mmol, 67.2%收率, ~85%纯度)。不经进一步纯化而直接使用该产物。LCMS (m/z): 643 [M + H]+。To a solution of 3-amino-4-((4-((2-amino-4-carbamoylphenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-(3-(benzyloxy)propoxy)benzamide (2.1 g, 3.54 mmol) in MeOH (20 mL) was added cyanogen bromide (1.126 g, 10.63 mmol). The reaction mixture was stirred at 25 °C for 16 h. The mixture was diluted with diethyl ether (30 mL). The mixture was filtered and the filter cake was washed with diethyl ether. The filtrate was concentrated under reduced pressure to afford 2-amino-1-(4-(2-amino-5-carbamoyl-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-7-(3-(benzyloxy)propoxy)-1H-benzo[d]imidazole-5-carboxamide (1.8 g, 2.381 mmol, 67.2% yield, -85% purity) as a grey solid. The product was used directly without further purification. LCMS ( m/z ): 643 [M+H] + .
步骤6: 7-(3-(苄氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 6: 7-(3-(Benzyloxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-5-carboxamide
向1-乙基-3-甲基-1H-吡唑-5-甲酸 (0.864 g, 5.60 mmol), 2-氨基-1-(4-(2-氨基-5-氨基甲酰基-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-7-(3-(苄氧基)丙氧基)-1H-苯并[d]咪唑-5-甲酰胺 (1.8 g, 2.80 mmol)和DIPEA (1.957 mL, 11.20 mmol)在DMF (20 mL)中的溶液加入HATU (2.66 g, 7.00 mmol)。在60℃搅拌该反应混合物16 h。将该混合物倾倒至水中。通过过滤收集沉淀物,用水、MeCN和二乙醚洗涤,并然后在真空下干燥以提供作为浅棕色固体的7-(3-(苄氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (1.1 g, 1.05 mmol, 37.3%收率, ~87%纯度)。不经进一步纯化而使用该产物。LCMS (m/z): 915 [M + H]+。To a solution of 1-ethyl-3-methyl-1H-pyrazole-5-carboxylic acid (0.864 g, 5.60 mmol), 2-amino-1-(4-(2-amino-5-carbamoyl-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-7-(3-(benzyloxy)propoxy)-1H-benzo[d]imidazole-5-carboxamide (1.8 g, 2.80 mmol) and DIPEA (1.957 mL, 11.20 mmol) in DMF (20 mL) was added HATU (2.66 g, 7.00 mmol). The reaction mixture was stirred at 60 °C for 16 h. The mixture was poured into water. The precipitate was collected by filtration, washed with water, MeCN, and diethyl ether, and then dried under vacuum to afford 7-(3-(benzyloxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (1.1 g, 1.05 mmol, 37.3% yield, -87% purity) as a light brown solid. The product was used without further purification. LCMS ( m/z ): 915 [M+H] + .
步骤7: 1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-羟基丙氧基)-1H-苯并[d]咪唑-5-甲酰胺Step 7: 1-(4-(5-Carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-hydroxypropoxy)-1H-benzo[d]imidazol-5-carboxamide
向7-(3-(苄氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (1.1 g, 1.2 mmol)在MeOH (30 mL)和NMP (10.0mL)中的溶液加入碳载钯 (1.279 g)。在60℃使用H-cube系统(4 atm 氢气)氢化该反应72h。用DMF (20 mL)稀释该混合物。然后,通过过滤除去催化剂并在减压下浓缩滤液。通过制备型HPLC (Gemini-C18, 5µ硅胶, 21x150 mm柱, 10-60%梯度的MeCN/水,含0.1% TFA 改性剂)纯化粗产物。合并纯级分并蒸发至干以提供作为粉色固体的1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-羟基丙氧基)-1H-苯并[d]咪唑-5-甲酰胺(75 mg, 0.086 mmol, 7.19%收率)。1H NMR (400 MHz, DMSO) δ 13.06-13.04 (m, 2 H),8.06-8.03 (m, 3 H), 7.84 (d, J = 8.4 Hz, 1 H), 7.70 (s, 1 H), 7.53 (d, J =8.4 Hz, 1 H), 7.46 (s, 1 H), 7.40 (s, 2 H), 6.66-6.65 (m, 2 H), 5.27-2.20 (m,4 H), 4.62-4.50 (m, 5 H), 4.26 (t, J = 6.1 Hz, 2 H), 3.55 (t, J = 5.7 Hz, 2H), 2.06 (s, 6 H), 1.98-1.82 (m, 2 H), 1.33 (t, J = 7.0 Hz, 6 H)。LCMS (m/z):824.4 [M + H]+。To a solution of 7-(3-(benzyloxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (1.1 g, 1.2 mmol) in MeOH (30 mL) and NMP (10.0 mL) was added palladium on carbon (1.279 g). The reaction was hydrogenated at 60 °C using an H-cube system (4 atm hydrogen) for 72 h. The mixture was diluted with DMF (20 mL). The catalyst was then removed by filtration and the filtrate was concentrated under reduced pressure. The crude product was purified by preparative HPLC (Gemini-C18, 5µ silica gel, 21x150 mm column, 10-60% gradient of MeCN/water with 0.1% TFA modifier). Pure fractions were combined and evaporated to dryness to afford 1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-hydroxypropoxy)-1H-benzo[d]imidazole-5-carboxamide (75 mg, 0.086 mmol, 7.19% yield) as a pink solid. 1 H NMR (400 MHz, DMSO) δ 13.06-13.04 (m, 2 H), 8.06-8.03 (m, 3 H), 7.84 (d, J = 8.4 Hz, 1 H), 7.70 (s, 1 H), 7.53 (d, J =8.4 Hz, 1 H), 7.46 (s, 1 H), 7.40 (s, 2 H), 6.66-6.65 (m, 2 H), 5.27-2.20 (m,4 H), 4.62-4.50 (m, 5 H), 4.26 (t, J = 6.1 Hz, 2 H), 3.55 (t, J = 5.7 Hz, 2H), 2.06 (s, 6 H), 1.98-1.82 (m, 2 H), 1.33 (t, J = 7.0 Hz, 6 H). LCMS ( m/z ):824.4 [M + H] + .
步骤8: (E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 2三氟乙酸盐Step 8: (E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 2-trifluoroacetate
向8-mL小瓶放入1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-羟基丙氧基)-1H-苯并[d]咪唑-5-甲酰胺 (25 mg, 0.030 mmol)、 碳酸铯(49.4 mg, 0.152 mmol)和DMF (1 mL)。向这个溶液加入碘甲烷 (4.26 µL, 0.068 mmol)。将小瓶盖上盖子并在室温搅拌该混合物过夜(~14 小时)。用更多的DMF稀释该样品并通过质量导向的制备型HPLC (XSELECT CSH C18, 5 um填充物, 150x30 mm柱, 15-55%梯度的MeCN/水,含0.1% TFA 改性剂)直接纯化。合并相应的级分并在真空中浓缩以提供作为透明固体的(E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 2三氟乙酸盐 (2 mg, 1.702 µmol, 5.62%收率)。1H NMR (DMSO-d 6, 600 MHz): δ ppm 8.09(s, 2 H), 8.05 (br. s., 1 H), 7.89 (dd, J=8.4, 1.2 Hz, 1 H), 7.75 (s, 1 H),7.57 (d, J=8.3 Hz, 1 H), 7.51 (s, 1 H), 7.48 (br. s., 1 H), 7.46 (br. s., 1H), 6.52 (s, 1 H), 6.51 (s, 1 H), 5.15-5.30 (m, 4 H), 4.51-4.48 (m, 4 H),4.22 (br. t., J=6.5 Hz, 2 H), 3.60 (s, 3 H), 3.57 (s, 3 H), 3.51 (br. t., J=6.0 Hz, 2 H), 2.15 (s, 3 H), 2.12 (s, 3 H), 1.84 (br. t., J=6.3 Hz, 2 H),1.24-1.26 (m, 6 H)。LCMS (m/z): 853.4 [M + H]+。Into an 8-mL vial was placed 1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-hydroxypropoxy)-1H-benzo[d]imidazole-5-carboxamide (25 mg, 0.030 mmol), cesium carbonate (49.4 mg, 0.152 mmol), and DMF (1 mL). To this solution was added iodomethane (4.26 µL, 0.068 mmol). The vial was capped and the mixture was stirred at room temperature overnight (~14 h). The sample was diluted with more DMF and directly purified by mass-directed preparative HPLC (XSELECT CSH C18, 5 um packing, 150x30 mm column, 15-55% gradient of MeCN/water with 0.1% TFA modifier). The corresponding fractions were combined and concentrated in vacuo to afford (E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, 2-trifluoroacetate salt (2 mg, 1.702 µmol, 5.62% yield) as a clear solid. 1 H NMR (DMSO- d 6 , 600 MHz): δ ppm 8.09 (s, 2 H), 8.05 (br. s., 1 H), 7.89 (dd, J =8.4, 1.2 Hz, 1 H), 7.75 (s, 1 H), 7.57 (d, J =8.3 Hz, 1 H), 7.51 (s , 1 H), 7.48 (br. s., 1 H), 7.46 (br. s., 1H), 6.52 (s, 1 H), 6.51 (s, 1 H), 5.15-5.30 (m, 4 H), 4.51-4.48 (m, 4 H), 4.22 (br. t., J =6.5 Hz, 2 H ), 3.60 (s, 3 H), 3.57 (s, 3 H), 3.51 (br. t., J =6.0 Hz, 2 H), 2.15 (s, 3 H), 2.12 (s, 3 H), 1.84 (br. t., J =6.3 Hz, 2 H), 1.24-1.26 (m, 6 H). LCMS (m/z): 853.4 [M + H] + .
实施例18Embodiment 18
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
步骤1: (E)-4-((4-(1,3-二氧代异吲哚啉-2-基)-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺Step 1: (E)-4-((4-(1,3-dioxoisoindolin-2-yl)-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide
向(E)-2-(4-氨基-2,3-二甲基丁-2-烯-1-基)异吲哚啉-1,3-二酮 (5.7 g,23.33 mmol)和4-氟-3-硝基苯甲酰胺 (3.9 g, 21.18 mmol)在DMSO (65 mL)中的溶液加入碳酸钾(6.44 g, 46.6 mmol)。将该反应在室温搅拌3 h。将该反应混合物倾倒至装有300mL快速搅拌水的烧瓶中。将该混合物搅拌5 min,过滤并用水、二乙醚 (2x)和乙酸乙酯(2x)顺序地漂洗经过滤的固体。收集固体,在己烷 (30 mL)中搅拌,过滤并干燥以提供作为亮黄色固体的(E)-4-((4-(1,3-二氧代异吲哚啉-2-基)-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺 (6.77 g, 16.6 mmol, 78%收率)。LCMS (m/z): 409.2 [M + H]+。To a solution of (E)-2-(4-amino-2,3-dimethylbut-2-en-1-yl)isoindoline-1,3-dione (5.7 g, 23.33 mmol) and 4-fluoro-3-nitrobenzamide (3.9 g, 21.18 mmol) in DMSO (65 mL) was added potassium carbonate (6.44 g, 46.6 mmol). The reaction was stirred at room temperature for 3 h. The reaction mixture was poured into a flask containing 300 mL of rapidly stirring water. The mixture was stirred for 5 min, filtered and the filtered solid was rinsed sequentially with water, diethyl ether (2x) and ethyl acetate (2x). The solid was collected, stirred in hexanes (30 mL), filtered and dried to provide (E)-4-((4-(1,3-dioxoisoindolin-2-yl)-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide (6.77 g, 16.6 mmol, 78% yield) as a bright yellow solid. LCMS ( m/z ): 409.2 [M+H] + .
步骤2: (E)-4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-3-((4-甲氧基苄基)氧基)-5-硝基苯甲酰胺Step 2: (E)-4-((4-((4-carbamoyl-2-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-3-((4-methoxybenzyl)oxy)-5-nitrobenzamide
向(E)-4-((4-(1,3-二氧代异吲哚啉-2-基)-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺 (6.4 g, 15.67 mmol)在EtOH (100 mL)中的悬浮液加入肼一水合物 (0.84mL, 17.2 mmol)。在10 min后,加入EtOH (50 ml)以促进搅拌。将该反应混合物在80℃加热20 h。在仍然温热时将该反应过滤,并在固体和滤液二者中都发现所需产物。合并固体和滤液,浓缩至干并在接下来的反应中以粗物质形式使用。To a suspension of (E)-4-((4-(1,3-dioxoisoindolin-2-yl)-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide (6.4 g, 15.67 mmol) in EtOH (100 mL) was added hydrazine monohydrate (0.84 mL, 17.2 mmol). After 10 min, EtOH (50 ml) was added to facilitate stirring. The reaction mixture was heated at 80 °C for 20 h. The reaction was filtered while still warm and the desired product was found in both the solid and the filtrate. The solid and filtrate were combined, concentrated to dryness and used crude in the next reaction.
向(E)-4-((4-氨基-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺 (4.65 g,10.53 mmol)和4-氯-3-((4-甲氧基苄基)氧基)-5-硝基苯甲酰胺 (3.30 g, 9.80 mmol)在1-丁醇 (100 mL)中的亮黄色悬浮液加入碳酸氢钠(2.47 g, 29.4 mmol)。在120℃加热该混合物。在18 h后,将该反应混合物冷却至室温,搅拌15 min并将固体过滤和用正丁醇(2x25 mL)漂洗。用一份水稀释一份饱和NaHCO3溶液。用经稀释的NaHCO3 溶液(2x25 mL)、水(1x25 mL)、经稀释的NaHCO3溶液(50 mL)和水(30 mL)洗涤固体。将固体收集在圆底烧瓶中并在室温在经稀释的bicarb溶液(100 mL)中搅拌2 h。过滤固体,用水洗涤并在真空炉中干燥以获得作为橙色固体的(E)-4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-3-((4-甲氧基苄基)氧基)-5-硝基苯甲酰胺 (3.62 g, 6.3 mmol,63.8%收率)。LCMS (m/z): 579.3 [M + H]+。To a bright yellow suspension of (E)-4-((4-amino-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide (4.65 g, 10.53 mmol) and 4-chloro-3-((4-methoxybenzyl)oxy)-5-nitrobenzamide (3.30 g, 9.80 mmol) in 1-butanol (100 mL) was added sodium bicarbonate (2.47 g, 29.4 mmol). The mixture was heated at 120 °C. After 18 h, the reaction mixture was cooled to room temperature, stirred for 15 min and the solid was filtered and rinsed with n-butanol (2x25 mL). One portion of saturated NaHCO 3 solution was diluted with one portion of water. The solid was washed with diluted NaHCO 3 solution (2x25 mL), water (1x25 mL), diluted NaHCO 3 solution (50 mL) and water (30 mL). The solid was collected in a round bottom flask and stirred in a diluted bicarb solution (100 mL) at room temperature for 2 h. The solid was filtered, washed with water and dried in a vacuum oven to obtain (E)-4-((4-((4-carbamoyl-2-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-3-((4-methoxybenzyl)oxy)-5-nitrobenzamide (3.62 g, 6.3 mmol, 63.8% yield) as an orange solid. LCMS ( m/z ): 579.3 [M + H] + .
步骤3: (E)-3-氨基-4-((4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-5-((4-甲氧基苄基)氧基)苯甲酰胺Step 3: (E)-3-amino-4-((4-((2-amino-4-carbamoylphenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-5-((4-methoxybenzyl)oxy)benzamide
向(E)-4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-3-((4-甲氧基苄基)氧基)-5-硝基苯甲酰胺 (3.46 g, 5.98 mmol)在MeOH (25 mL)和乙酸(20 mL)中的悬浮液加入活性炭上的1% Pt和2% V (50-70%湿化粉末, 1.167 g,0.060 mmol)。在室温在氢气气氛(氢气球)下搅拌该烧瓶6 h。将MeOH (10 mL)用于将固体从烧瓶的侧壁漂洗掉并再继续搅拌16 h。在除去氢气之后,将该反应过滤通过硅藻土,用MeOH漂洗并浓缩以提供稠橙色油。加入DCM (15 mL),并伴随搅拌,用饱和NaHCO3溶液处理所得混合物(以1 mL的份,直到停止冒泡并且水层为碱性)。将液体滗析掉,并在3:1 CHCl3:EtOH和盐水之间分配剩余固体。经硫酸钠干燥有机层并浓缩以提供浅棕色泡沫。用MeOH研磨泡沫以提供(E)-3-氨基-4-((4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-5-((4甲氧基苄基)氧基)苯甲酰胺 (1.4 g, 2.70 mmol, 45.1%收率)。LCMS (m/z): 519.4 [M + H]+。To a suspension of (E)-4-((4-((4-carbamoyl-2-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-3-((4-methoxybenzyl)oxy)-5-nitrobenzamide (3.46 g, 5.98 mmol) in MeOH (25 mL) and acetic acid (20 mL) was added 1% Pt and 2% V on activated carbon (50-70% wet powder, 1.167 g, 0.060 mmol). The flask was stirred at room temperature under a hydrogen atmosphere (hydrogen balloon) for 6 h. MeOH (10 mL) was used to rinse the solids off the sides of the flask and stirring was continued for an additional 16 h. After removal of the hydrogen, the reaction was filtered through celite, rinsed with MeOH and concentrated to provide a thick orange oil. DCM (15 mL) was added, and with stirring, the resulting mixture was treated with saturated NaHCO solution ( 1 mL portion until bubbling stopped and the aqueous layer was alkaline). The liquid was decanted and the remaining solid was distributed between 3:1 CHCl 3 :EtOH and saline. The organic layer was dried over sodium sulfate and concentrated to provide a light brown foam. The foam was ground with MeOH to provide (E)-3-amino-4-((4-((2-amino-4-carbamoylphenyl)amino)-2,3-dimethylbut-2-ene-1-yl)amino)-5-((4-methoxybenzyl)oxy)benzamide (1.4 g, 2.70 mmol, 45.1% yield). LCMS ( m/z ): 519.4 [M+H] + .
步骤4: (E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-((4-甲氧基苄基)氧基)-1H-苯并[d]咪唑-5-甲酰胺Step 4: (E)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-((4-methoxybenzyl)oxy)-1H-benzo[d]imidazol-5-carboxamide
经2 min向(E)-3-氨基-4-((4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-5-((4-甲氧基苄基)氧基)苯甲酰胺 (1.40 g, 2.70 mmol)在DMF(18 mL)中的冰冷溶液逐滴加入1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (~1 M 在二氧杂环己烷中, 5.40 mL, 5.40 mmol)。在15 min后,加入EDC (1.29 g, 6.75 mmol)和TEA(1.881 mL, 13.50 mmol)。将该反应温热至室温并在40℃加热22 h。将该反应冷却至室温并倾倒至快速搅拌的3:1水:饱和NH4Cl水溶液(100 mL)中。立即形成细固体并再搅拌该混合物10 min。过滤该固体,用水(50 mL)洗涤两次并在50℃的真空炉中干燥过夜以提供作为浅褐色固体的(E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-((4-甲氧基苄基)氧基)-1H-苯并[d]咪唑-5-甲酰胺 (2.20 g, 2.48 mmol, 92%收率)。1H NMR (DMSO-d6) δ: 12.97 (d, J=3.3 Hz, 2 H), 8.08 (s, 1 H), 8.03 (br. s., 1H), 7.96 (br. s., 1 H), 7.68-7.75 (m, 2 H), 7.50 (s, 1 H), 7.40 (d, J=9.6 Hz,2 H), 7.16 (d, J=8.4 Hz, 1 H), 7.06-7.12 (m, 2 H), 6.72 (s, 1 H), 6.70 (s, 1H), 6.56 (s, 1 H), 6.50 (s, 1 H), 5.04 (br. s., 2 H), 4.96 (s, 2 H), 4.88(br. s., 2 H), 4.53-4.62 (m, 4 H), 3.59 (s, 3 H), 2.10 (s, 3 H), 2.05 (s, 3H), 1.66 (s, 3 H), 1.32 (t, J=7.0 Hz, 6 H), 1.17 (s, 3 H)。LCMS (m/z): 841.5[M + H]+。To an ice-cold solution of (E)-3-amino-4-((4-((2-amino-4-carbamoylphenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-5-((4-methoxybenzyl)oxy)benzamide (1.40 g, 2.70 mmol) in DMF (18 mL) was added 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (~1 M in dioxane, 5.40 mL, 5.40 mmol) dropwise over 2 min. After 15 min, EDC (1.29 g, 6.75 mmol) and TEA (1.881 mL, 13.50 mmol) were added. The reaction was allowed to warm to room temperature and heated at 40 °C for 22 h. The reaction was cooled to room temperature and poured into a rapidly stirring 3:1 water:saturated aqueous NH 4 Cl solution (100 mL). A fine solid formed immediately and the mixture was stirred for an additional 10 min. The solid was filtered, washed twice with water (50 mL) and dried in a vacuum oven at 50 °C overnight to provide (E)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-((4-methoxybenzyl)oxy)-1H-benzo[d]imidazole-5-carboxamide (2.20 g, 2.48 mmol, 92% yield) as a light brown solid. 1 H NMR (DMSO-d 6 ) δ: 12.97 (d, J=3.3 Hz, 2 H), 8.08 (s, 1 H), 8.03 (br. s., 1H), 7.96 (br. s., 1 H), 7.68-7.75 (m, 2 H), 7.50 (s, 1 H), 7.40 (d . 96 (s, 2 H), 4.88(br. s., 2 H), 4.53-4.62 (m, 4 H), 3.59 (s, 3 H), 2.10 (s, 3 H), 2.05 (s, 3H), 1.66 (s, 3 H), 1.32 (t, J=7.0 Hz, 6 H), 1.17 (s, 3 H). LCMS ( m/z ): 841.5[M + H] + .
步骤5: (E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 5: (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
向(E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-((4-甲氧基苄基)氧基)-1H-苯并[d]咪唑-5-甲酰胺 (1.30 g, 1.546 mmol)在DMF (12mL)中的溶液加入碳酸铯 (1.511 g, 4.64 mmol)和碘甲烷 (0.242 mL, 3.86 mmol)。将该反应混合物在室温搅拌1 h。将水(120 mL)缓慢加入该反应混合物。将该混合物剧烈搅拌30min。在过滤器上收集所得固体并干燥。通过硅胶层析法 (80 g柱, 30-80%梯度的(3:1 EA:EtOH,含1% 氢氧化铵)/己烷)纯化粗产物以提供作为浅黄色固体的标题化合物(1.01 g,1.16 mmol, 74%收率)。1H NMR (DMSO-d6) δ: 8.15 (s, 1 H), 8.10 (br. s., 1 H),8.05 (br. s., 1 H), 7.77-7.82 (m, 2 H), 7.62 (s, 1 H), 7.52 (br. s., 1 H),7.49 (br. s., 1 H), 7.22 (d, J=8.6 Hz, 1 H), 7.17 (d, J=8.6 Hz, 2 H), 6.68(d, J=8.9 Hz, 2 H), 6.41 (s, 1 H), 6.38 (s, 1 H), 5.09 (s, 2 H), 4.96 (s, 2H), 4.77 (s, 2 H), 4.48 (q, J=7.0 Hz, 4 H), 3.58-3.63 (m, 6 H), 3.56 (s, 3H), 2.07-2.11 (m, 6 H), 1.55 (s, 3 H), 1.22-1.28 (m, 6 H), 1.16 (s, 3 H)。LCMS(m/z): 869.6 [M + H]+。To a solution of (E)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-((4-methoxybenzyl)oxy)-1H-benzo[d]imidazole-5-carboxamide (1.30 g, 1.546 mmol) in DMF (12 mL) was added cesium carbonate (1.511 g, 4.64 mmol) and iodomethane (0.242 mL, 3.86 mmol). The reaction mixture was stirred at room temperature for 1 h. Water (120 mL) was slowly added to the reaction mixture. The mixture was stirred vigorously for 30 min. The resulting solid was collected on a filter and dried. The crude product was purified by silica gel chromatography (80 g column, 30-80% gradient of (3:1 EA:EtOH with 1% ammonium hydroxide)/hexanes) to provide the title compound (1.01 g, 1.16 mmol, 74% yield) as a light yellow solid. 1 H NMR (DMSO-d 6 ) δ: 8.15 (s, 1 H), 8.10 (br. s., 1 H), 8.05 (br. s., 1 H), 7.77-7.82 (m, 2 H), 7.62 (s, 1 H), 7.52 (br. s., 1 H), 7.49 (br. s., 1 H), 7.22 (d, J=8.6 Hz, 1 H), 7.17 (d, J=8.6 Hz, 2 H), 6.68 (d, J=8.9 Hz, 2 H), 6.41 (s, 1 H), 6.38 (s, 1 H), 5.09 (s, 2 H), 4.96 (s, 2H), 4.77 ( s, 2 H), 4.48 (q, J=7.0 Hz, 4 H), 3.58-3.63 (m, 6 H), 3.56 (s, 3H), 2.07-2.11 (m, 6 H), 1.55 (s, 3 H), 1.22-1.28 (m, 6 H), 1.16 (s, 3 H). LCMS ( m/z ): 869.6 [M + H] + .
实施例19Embodiment 19
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-羟基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-hydroxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
向(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (930 mg, 1.070 mmol)在DCM (15 mL)中的悬浮液加入HCl (4 M 在二氧杂环己烷中, 1.34 mL, 5.35 mmol)。反应混合物变稠。加入DCM (10 mL)和MeOH (2mL)以获得均相溶液。在室温搅拌该反应16 h。由于反应未完全,浓缩该反应混合物并将固体残余物悬浮在DCM (15 mL)和TFA (0.412 mL, 5.35 mmol)中。在30 min中,完全脱保护。浓缩该反应混合物并在3:1 CHCl3:EtOH和饱和NaHCO3溶液之间分配残余物。过滤存在的固体并干燥以提供标题化合物(427 mg, 0.57 mmol)。浓缩剩余的有机相并通过硅胶层析法(12 g硅胶柱, 1-8%梯度的MeOH/DCM) 纯化残余物以提供作为灰白色固体的另外量的标题化合物(130 mg, 0.17 mmol)。1H NMR (DMSO-d6) δ ppm 10.68 (s, 1 H), 8.10 (s, 1H), 8.01 (br. s., 1 H), 7.96 (br. s., 1 H), 7.80 (d, J=8.4 Hz, 1 H), 7.62 (s,1 H), 7.46 (br. s., 1 H), 7.38 (br. s., 1 H), 7.32 (s, 1 H), 7.22 (d, J=8.4Hz, 1 H), 6.47 (s, 1 H), 6.33 (s, 1 H), 5.07 (s, 2 H), 4.83 (s, 2 H), 4.44-4.56 (m, 4 H), 3.58 (s, 3 H), 3.53 (s, 3 H), 2.14 (s, 3 H), 2.07 (s, 3 H),1.65 (s, 3 H), 1.48 (s, 3 H), 1.31-1.24 (m, 6 H)。LCMS (m/z): 749.5 [M + H]+。To a suspension of (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (930 mg, 1.070 mmol) in DCM (15 mL) was added HCl (4 M in dioxane, 1.34 mL, 5.35 mmol). The reaction mixture thickened. DCM (10 mL) and MeOH (2 mL) were added to obtain a homogeneous solution. The reaction was stirred at room temperature for 16 h. Because the reaction was not complete, the reaction mixture was concentrated and the solid residue was suspended in DCM (15 mL) and TFA (0.412 mL, 5.35 mmol). In 30 min, deprotection was complete. The reaction mixture was concentrated and the residue was distributed between 3:1 CHCl 3 :EtOH and saturated NaHCO 3 solution. The solid present was filtered and dried to provide the title compound (427 mg, 0.57 mmol). The remaining organic phase was concentrated and the residue was purified by silica gel chromatography (12 g silica gel column, 1-8% gradient of MeOH/DCM) to provide the title compound (130 mg, 0.17 mmol) as an additional amount of off-white solid. 1 H NMR (DMSO-d 6 ) δ ppm 10.68 (s, 1 H), 8.10 (s, 1H), 8.01 (br. s., 1 H), 7.96 (br. s., 1 H), 7.80 (d, J=8.4 Hz, 1 H), 7.62 (s,1 H), 7.46 (br. s., 1 H), 7.38 (br. s., 1 H), 7.32 (s, 1 H), 7.22 (d, J=8.4Hz, 1 H), 6.47 (s, 1 H), 6.33 (s, 1 H), 5.07 (s, 2 H), 4.83 (s, 2 H), 4.44-4.56 (m, 4 H) , 3.58 (s, 3 H), 3.53 (s, 3 H), 2.14 (s, 3 H), 2.07 (s, 3 H), 1.65 (s, 3 H), 1.48 (s, 3 H), 1.31-1.24 (m, 6 H). LCMS ( m/z ): 749.5 [M + H] + .
实施例20Embodiment 20
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
步骤1: (E)-4-((4-氨基-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺Step 1: (E)-4-((4-amino-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide
向(E)-2,3-二甲基丁-2-烯-1,4-二胺, 2盐酸盐 (4.4 g, 23.52 mmol)和乙醇(81 ml)中加入碳酸钾(6.76 g, 48.9 mmol)在水(81 ml)中的溶液。一旦所有固体溶解,在室温将4-氟-3-硝基苯甲酰胺 (3.0 g, 16.29 mmol)一次性加入该紫棕色溶液。在室温搅拌该反应混合物105 min,然后在50℃加热90 min并过滤。用6N HCl酸化滤液。用DCM (2x)洗涤水层。用水(1x)萃取合并的有机物。用6N NaOH和饱和碳酸氢钠碱化合并的水层并用3:1 氯仿/乙醇混合物(3x)萃取。经硫酸钠干燥合并的有机萃取物,过滤并浓缩至干以提供作为带棕色的黄色残余物的(E)-4-((4-氨基-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺 (1.8 g, 5.30 mmol, 32.5 %收率)。LCMS (m/z): 279.1 [M + H]+。To (E)-2,3-dimethylbut-2-ene-1,4-diamine, 2-hydrochloride (4.4 g, 23.52 mmol) and ethanol (81 ml) was added a solution of potassium carbonate (6.76 g, 48.9 mmol) in water (81 ml). Once all solids were dissolved, 4-fluoro-3-nitrobenzamide (3.0 g, 16.29 mmol) was added to the purple-brown solution in one portion at room temperature. The reaction mixture was stirred at room temperature for 105 min, then heated at 50 °C for 90 min and filtered. The filtrate was acidified with 6N HCl. The aqueous layer was washed with DCM (2x). The combined organics were extracted with water (1x). The combined aqueous layers were basified with 6N NaOH and saturated sodium bicarbonate and extracted with a 3:1 chloroform/ethanol mixture (3x). The combined organic extracts were dried over sodium sulfate, filtered and concentrated to dryness to afford (E)-4-((4-amino-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide (1.8 g, 5.30 mmol, 32.5 % yield) as a brownish yellow residue. LCMS ( m/z ): 279.1 [M+H] + .
步骤2: (E)-3-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-((4-((4-氨基甲酰基-2-Step 2: (E)-3-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-((4-((4-carbamoyl-2-
硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-5-硝基苯甲酰胺(nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-5-nitrobenzamide
向粗品(E)-4-((4-氨基-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺 (715mg, 2.312 mmol)在1-丁醇 (10.9 mL)中的悬浮液加入DIE (1.14 mL, 6.56 mmol)。搅拌该混合物10 min,然后加入3-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氯-5-硝基苯甲酰胺 (850 mg, 2.186 mmol)。在120℃加热该反应混合物过夜。将该反应冷却至室温并沉淀出橙色固体。过滤该混合物并用乙酸乙酯洗涤滤饼以提供作为亮橙色固体的粗品(E)-3-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-5-硝基苯甲酰胺 (786 mg, 1.022 mmol, 46.8 %收率),其仍含有残留n-BuOH,但原样承载在其上。LCMS (m/z): 631.3 [M + H]+。To a suspension of crude (E)-4-((4-amino-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide (715 mg, 2.312 mmol) in 1-butanol (10.9 mL) was added DIE (1.14 mL, 6.56 mmol). The mixture was stirred for 10 min and then 3-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-chloro-5-nitrobenzamide (850 mg, 2.186 mmol) was added. The reaction mixture was heated at 120 °C overnight. The reaction was cooled to room temperature and an orange solid precipitated. The mixture was filtered and the filter cake was washed with ethyl acetate to afford crude (E)-3-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-((4-((4-carbamoyl-2-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-5-nitrobenzamide (786 mg, 1.022 mmol, 46.8 % yield) as a bright orange solid, which still contained residual n-BuOH but was supported on it as is. LCMS ( m/z ): 631.3 [M+H] + .
步骤3: (E)-3-氨基-4-((4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-5-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)苯甲酰胺Step 3: (E)-3-amino-4-((4-((2-amino-4-carbamoylphenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-5-(3-((tert-butyldimethylsilyl)oxy)propoxy)benzamide
向(E)-3-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-5-硝基苯甲酰胺 (1.82 g, 2.453mmol)在甲醇 (53.3 ml)中的混合物加入氯化铵 (2.62 g, 49.1 mmol)然后加入锌(3.21g, 49.1 mmol)。在室温搅拌非均相混合物10 min。过滤该反应混合物并用甲醇洗涤滤饼。将滤液干装载至硅胶上并通过硅胶层析法 (80 g柱, 0%-20% 甲醇, DCM,含0.1% NH4OH改性剂)纯化。将所需级分浓缩至干以提供作为白色固体的(E)-3-氨基-4-((4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-5-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)苯甲酰胺 (503 mg, 0.881 mmol, 35.9 %收率)。LCMS (m/z): 571.5 [M+ H]+。To a mixture of (E)-3-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-((4-((4-carbamoyl-2-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-5-nitrobenzamide (1.82 g, 2.453 mmol) in methanol (53.3 ml) was added ammonium chloride (2.62 g, 49.1 mmol) followed by zinc (3.21 g, 49.1 mmol). The heterogeneous mixture was stirred at room temperature for 10 min. The reaction mixture was filtered and the filter cake was washed with methanol. The filtrate was dry loaded onto silica gel and purified by silica gel chromatography (80 g column, 0%-20% methanol, DCM with 0.1% NH 4 OH modifier). The desired fractions were concentrated to dryness to afford (E)-3-amino-4-((4-((2-amino-4-carbamoylphenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-5-(3-((tert-butyldimethylsilyl)oxy)propoxy)benzamide (503 mg, 0.881 mmol, 35.9% yield) as a white solid. LCMS ( m/z ): 571.5 [M+H] + .
步骤4: (E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 4: (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-5-carboxamide
在0℃向(E)-3-氨基-4-((4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-5-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)苯甲酰胺 (500 mg,0.876 mmol)在DMF (8.8 mL)中的溶液逐滴和逐份加入在二氧杂环己烷中的1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯(~0.4 M, 4.82 mL, 1.93 mmol)。在15 min后,加入另外的异硫氰酸酯(~0.4 M, 400 µL)并搅拌该反应混合物10 min。用EDC (420 mg, 2.190 mmol)和TEA (610 µl, 4.38 mmol)处理该反应并在室温搅拌过周末。将该反应混合物倾倒至4:1水/饱和氯化铵 (200 mL)中并过滤所得悬浮液。在氮气流下干燥滤饼以提供(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (614 mg, 0.687 mmol, 78 %收率)。1HNMR (400 MHz, DMSO-d 6) δ ppm 12.92 (d, J=11.66 Hz, 2 H), 7.99 - 8.08 (m, 2H), 7.90 (br. s., 1 H), 7.70 (s, 1 H), 7.65 (d, J=8.11 Hz, 1 H), 7.31 - 7.43(m, 3 H), 7.16 (d, J=8.36 Hz, 1 H), 6.60 (s, 1 H), 6.42 (s, 1 H), 5.14 (br.s., 2 H), 4.98 (br. s., 2 H), 4.48 - 4.66 (m, 4 H), 4.14 (t, J=5.58 Hz, 2 H),3.65 (t, J=5.96 Hz, 2 H), 2.02 - 2.14 (m, 6 H), 1.61 - 1.74 (m, 6 H), 1.33(t, 7.13 Hz, 3 H), 1.32 (t, 7.13 Hz, 3 H), 0.80 (s, 11 H), -0.03 (s, 6 H)。LCMS (m/z): 893.4 [M + H]+。To a solution of (E)-3-amino-4-((4-((2-amino-4-carbamoylphenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-5-(3-((tert-butyldimethylsilyl)oxy)propoxy)benzamide (500 mg, 0.876 mmol) in DMF (8.8 mL) at 0°C was added 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (~0.4 M, 4.82 mL, 1.93 mmol) in dioxane dropwise and portionwise. After 15 min, additional isothiocyanate (~0.4 M, 400 µL) was added and the reaction mixture was stirred for 10 min. The reaction was treated with EDC (420 mg, 2.190 mmol) and TEA (610 µl, 4.38 mmol) and stirred at room temperature over the weekend. The reaction mixture was poured into 4:1 water/saturated ammonium chloride (200 mL) and the resulting suspension was filtered. The filter cake was dried under a stream of nitrogen to provide (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (614 mg, 0.687 mmol, 78 % yield). 1 HNMR (400 MHz, DMSO- d 6 ) δ ppm 12.92 (d, J =11.66 Hz, 2 H), 7.99 - 8.08 (m, 2H), 7.90 (br. s., 1 H), 7.70 (s, 1 H), 7.65 (d, J =8.11 Hz, 1 H), 7.3 1 - 7.43(m, 3 H), 7.16 (d, J =8.36 Hz, 1 H), 6.60 (s, 1 H), 6.42 (s, 1 H), 5.14 (br.s., 2 H), 4.98 (br. s., 2 H), 4.48 - 4.66 (m, 4 H), 4.14 (t, J =5.58 Hz, 2 H), 3.65 (t, J =5.96 Hz, 2 H), 2.02 - 2.14 (m, 6 H), 1.61 - 1.74 (m, 6 H), 1.33 (t, 7.13 Hz, 3 H), 1.32 (t, 7.13 Hz, 3 H), 0.80 (s, 11 H), -0. 03 (s, 6 H). LCMS ( m/z ): 893.4 [M + H] + .
步骤5: (E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 5: (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
在室温搅拌(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺(0.245 g, 0.274 mmol)、碳酸铯 (0.268 g, 0.823 mmol)和碘甲烷 (0.043 mL, 0.686mmol)在DMF (2 mL)中的混合物18 h。在EtOAc (20 mL)和盐水(20 mL)之间分配该反应混合物。用EtOAc (2x10 mL)萃取水层。经硫酸镁干燥合并的EtOAc层,过滤并在真空中浓缩。通过硅胶层析法 (RediSepRf High Performance Gold 40 g HP硅胶柱, 40-80%梯度的(3:1乙醇/乙酸乙酯)/己烷,含2% NH4OH 改性剂)纯化粗产物以获得(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (0.040 g, 0.033 mmol, 11.9%收率)。LCMS (m/z): 921.7 [M + H]+。A mixture of (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (0.245 g, 0.274 mmol), cesium carbonate (0.268 g, 0.823 mmol) and iodomethane (0.043 mL, 0.686 mmol) in DMF (2 mL) was stirred at room temperature for 18 h. The reaction mixture was partitioned between EtOAc (20 mL) and brine (20 mL). The aqueous layer was extracted with EtOAc (2 x 10 mL).The combined EtOAc layers were dried over magnesium sulfate, filtered and concentrated in vacuo. The crude product was purified by silica gel chromatography (RediSepRf High Performance Gold 40 g HP silica column, 40-80% gradient of (3:1 ethanol/ethyl acetate)/hexanes with 2% NH 4 OH modifier) to afford (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (0.040 g, 0.033 mmol, 11.9% yield). LCMS ( m/z ): 921.7 [M + H] + .
步骤6: (E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 6: (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (0.030 g, 0.033 mmol)在MeOH (0.5 mL)中的混合物加入在二氧杂环己烷中的HCl (4 M, 0.163 mL, 0.651 mmol),在室温搅拌16 h。在真空中浓缩该反应混合物。通过硅胶层析法 (RediSepRf High Performance Gold 40 gHP硅胶柱, 40-90%梯度的(3:1乙醇/乙酸乙酯)/己烷,含2% NH4OH 改性剂)纯化粗产物以获得(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (0.007 g, 7.72 µmol, 23.7%收率)。1H NMR (600 MHz, DMSO-d6) δ 8.12 (s, 2H), 8.03 (br s, 1 H), 7.81 (d, J=8.66 Hz, 1 H), 7.78 (s, 1 H), 7.50 (s, 1 H),7.47 (s, 1 H), 7.45 (br s, 1 H), 7.24 (d, J=8.85 Hz, 1 H), 6.68 (br s, 1 H),6.45 (s, 1 H), 6.34 (s, 1 H), 5.06 (br s, 2 H), 4.85 (s, 2 H), 4.49-4.53 (m,2 H), 4.49 (br s, 2 H), 4.19 (br t, J=6.36 Hz, 2 H), 3.58 (s, 3 H), 3.56 (s,3 H), 3.44 (br t, J=5.99 Hz, 2 H), 2.10 (s, 3 H), 2.07 (s, 3 H), 1.72 (五重峰, J=6.27 Hz, 2 H), 1.61 (s, 3 H), 1.48 (s, 3 H), 1.27 (t, J=7.10 Hz, 3 H),1.24 (t, J=7.09 Hz, 3 H)。LCMS (m/z): 807.6 [M + H]+。To a mixture of (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (0.030 g, 0.033 mmol) in MeOH (0.5 mL) was added HCl in dioxane (4 M, 0.163 mL, 0.651 mmol) and stirred at room temperature for 16 h. The reaction mixture was concentrated in vacuo. The crude product was purified by silica gel chromatography (RediSepRf High Performance Gold 40 gHP silica column, 40-90% gradient of (3:1 ethanol/ethyl acetate)/hexanes with 2% NH4OH modifier) to afford (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (0.007 g, 7.72 µmol, 23.7% yield). 1 H NMR (600 MHz, DMSO-d 6 ) δ 8.12 (s, 2H), 8.03 (br s, 1 H), 7.81 (d, J =8.66 Hz, 1 H), 7.78 (s, 1 H), 7.50 (s, 1 H), 7.47 (s, 1 H), 7.45 (br s, 1 H) , 7.24 (d, J =8.85 Hz, 1 H), 6.68 (br s, 1 H), 6.45 (s, 1 H), 6.34 (s, 1 H), 5.06 (br s, 2 H), 4.85 (s, 2 H), 4.49-4.53 (m, 2 H), 4.49 (br s, 2 H), 4.19 (br t, J =6.36 Hz, 2 H), 3.58 (s, 3 H), 3.56 (s,3 H), 3.44 (br t, J =5.99 Hz, 2 H), 2.10 (s, 3 H), 2.07 (s, 3 H), 1.72 (quint, J =6.27 Hz, 2 H), 1.61 (s, 3 H ), 1.48 (s, 3 H), 1.27 (t, J =7.10 Hz, 3 H), 1.24 (t, J =7.09 Hz, 3 H). LCMS (m/z): 807.6 [M + H] + .
表1示出实施例21-28,其可以根据如下示例的方法制备:Table 1 shows Examples 21-28, which can be prepared according to the following exemplified method:
实施例29Embodiment 29
(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
步骤1: (E)-4-((4-((2-((4-甲氧基苄基)氧基)-6-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺Step 1: (E)-4-((4-((2-((4-methoxybenzyl)oxy)-6-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide
向(E)-4-((4-氨基-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺, 盐酸盐(2.4 g, 7.62 mmol)和2-氟-1-((4-甲氧基苄基)氧基)-3-硝基苯 (2.114 g, 7.62 mmol)在DMF (20 mL)中的亮黄色/橙色悬浮液加入TEA (3.19 mL, 22.87 mmol)。将稠混合物在室温搅拌2 h,然后在50℃加热18 h。将该反应混合物缓慢加入快速搅拌的水(350 mL)中。固体沉淀成块。声处理(30 min)并搅拌1 h提供自由流动的固体。过滤固体,用水和二乙醚(3x 30 mL)漂洗,并干燥以提供作为亮橙色固体的(E)-4-((4-((2-((4-甲氧基苄基)氧基)-6-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺 (3.52, 6.25mmol, 82%收率)。LCMS (m/z): 536.2 [M + H]+。To a bright yellow/orange suspension of (E)-4-((4-amino-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide, hydrochloride (2.4 g, 7.62 mmol) and 2-fluoro-1-((4-methoxybenzyl)oxy)-3-nitrobenzene (2.114 g, 7.62 mmol) in DMF (20 mL) was added TEA (3.19 mL, 22.87 mmol). The thick mixture was stirred at room temperature for 2 h and then heated at 50 °C for 18 h. The reaction mixture was slowly added to rapidly stirring water (350 mL). The solid precipitated as a clumping mass. Sonication (30 min) and stirring for 1 h afforded a free flowing solid. The solid was filtered, rinsed with water and diethyl ether (3 x 30 mL), and dried to afford (E)-4-((4-((2-((4-methoxybenzyl)oxy)-6-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide (3.52, 6.25 mmol, 82% yield) as a bright orange solid. LCMS ( m/z ): 536.2 [M+H] + .
步骤2: (E)-3-氨基-4-((4-((2-氨基-6-((4-甲氧基苄基)氧基)苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)苯甲酰胺Step 2: (E)-3-Amino-4-((4-((2-amino-6-((4-methoxybenzyl)oxy)phenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)benzamide
向(E)-4-((4-((2-((4-甲氧基苄基)氧基)-6-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺 (3.52 g, 6.57 mmol)在甲醇 (15 mL)和乙酸(10 mL)中的悬浮液加入活性炭上的1% Pt和2% V—50-70%湿化粉末(1.282 g, 0.066 mmol)。将烧瓶的气氛与氮气然后与氢气(气球)交换。在室温搅拌20 h后,将该气氛与氮气交换。由于反应未完全,在氢气气氛下搅拌该反应混合物另外的5 h,然后重新引入氮气。采用10% 甲醇/DCM使该反应混合物通过硅藻土并漂洗。在真空中的浓度提供稠橙色油。油吸纳在DCM (100mL)中并加入饱和碳酸氢钠水溶液直到停止冒泡。有机层的分离和浓缩提供棕色泡沫。由含少量DCM和MeOH的乙酸乙酯获得可过滤的固体。过滤所得固体并干燥以提供作为浅棕色固体的(E)-3-氨基-4-((4-((2-氨基-6-((4-甲氧基苄基)氧基)苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)苯甲酰胺 (1.95 g, 3.90 mmol, 59.3%收率)。LCMS (m/z): 476.3 [M +H]+。To a suspension of (E)-4-((4-((2-((4-methoxybenzyl)oxy)-6-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide (3.52 g, 6.57 mmol) in methanol (15 mL) and acetic acid (10 mL) was added 1% Pt and 2% V on activated carbon - 50-70% wet powder (1.282 g, 0.066 mmol). The atmosphere of the flask was exchanged with nitrogen and then with hydrogen (balloon). After stirring at room temperature for 20 h, the atmosphere was exchanged with nitrogen. As the reaction was not complete, the reaction mixture was stirred under hydrogen atmosphere for an additional 5 h and then nitrogen was reintroduced. The reaction mixture was passed through celite and rinsed with 10% methanol/DCM. Concentration in vacuo afforded a thick orange oil. The oil was taken up in DCM (100 mL) and saturated aqueous sodium bicarbonate solution was added until bubbling ceased. The separation and concentration of the organic layer provide brown foam.Filterable solid is obtained by ethyl acetate containing a small amount of DCM and MeOH.The resulting solid is filtered and dried to provide (E)-3-amino-4-((4-((2-amino-6-((4-methoxybenzyl)oxy)phenyl)amino)-2,3-dimethylbut-2-ene-1-yl)amino)benzamide (1.95 g, 3.90 mmol, 59.3% yield) as a light brown solid.LCMS ( m/z ): 476.3 [M+H] + .
步骤3: (E)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1-(4-(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-((4-甲氧基苄基)氧基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-1H-苯并[d]咪唑-5-甲酰胺Step 3: (E)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1-(4-(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-((4-methoxybenzyl)oxy)-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-1H-benzo[d]imidazole-5-carboxamide
向(E)-3-氨基-4-((4-((2-氨基-6-((4-甲氧基苄基)氧基)苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)苯甲酰胺 (1.93 g, 3.86 mmol)在冷却在冰/水浴中的DMF (20mL)中的浅棕色溶液快速滴加 (经~1 min) 1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯(~1M 在二氧杂环己烷中, 7.71 mL, 7.71 mmol)。搅拌该反应混合物25 min。加入EDC(1.848 g, 9.64 mmol)和TEA (2.69 mL, 19.28 mmol)并将该反应温热至室温并搅拌18h。将该反应混合物倾倒至快速搅拌的1:1饱和NH4Cl水溶液:水(100 mL)中以提供细沉淀物。采用水(2 x 15 mL)洗涤沉淀物,用乙酸乙酯 (20 mL)研磨两次,并干燥以提供作为褐色固体的(E)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1-(4-(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-((4-甲氧基苄基)氧基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-1H-苯并[d]咪唑-5-甲酰胺 (2.32 g, 2.83 mmol, 73%收率)。LCMS (m/z): 798.4[M + H]+。To a light brown solution of (E)-3-amino-4-((4-((2-amino-6-((4-methoxybenzyl)oxy)phenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)benzamide (1.93 g, 3.86 mmol) in DMF (20 mL) cooled in an ice/water bath was added 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (~1 M in dioxane, 7.71 mL, 7.71 mmol) dropwise rapidly (over ~1 min). The reaction mixture was stirred for 25 min. EDC (1.848 g, 9.64 mmol) and TEA (2.69 mL, 19.28 mmol) were added and the reaction was warmed to room temperature and stirred for 18 h. The reaction mixture was poured into a rapidly stirred 1:1 saturated NH4Cl aqueous solution:water (100 mL) to provide a fine precipitate. The precipitate was washed with water (2 x 15 mL), triturated twice with ethyl acetate (20 mL), and dried to provide (E)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1-(4-(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-((4-methoxybenzyl)oxy)-1H-benzo[d]imidazole-1-yl)-2,3-dimethylbut-2-ene-1-yl)-1H-benzo[d]imidazole-5-carboxamide (2.32 g, 2.83 mmol, 73% yield) as a brown solid. LCMS ( m/z ): 798.4[M+H] + .
步骤4: (E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 4: (E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
向(E)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1-(4-(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-((4-甲氧基苄基)氧基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-1H-苯并[d]咪唑-5-甲酰胺 (1.09 g, 1.366 mmol)在DMF (15 mL)中的溶液加入碳酸铯 (1.335 g, 4.10 mmol)和碘甲烷 (0.214 mL, 3.42 mmol)。将该反应混合物在室温搅拌5 h。加入另外的碘甲烷 (0.060 mL, 0.956 mmol)并再搅拌该混合物18 h。加入另外的碘甲烷 (0.060 mL, 0.956 mmol)和碳酸铯 (1.335 g, 4.10 mmol)并在50℃加热该混合物4 h。用水(30 mL)稀释该混合物并沉淀出粘性固体。剧烈搅拌提供可过滤的固体,其随后收集在过滤器上并用水漂洗。硅胶层析法 (40 g硅胶, 10-90%梯度的[3:1 EtOAc:EtOH]/庚烷)提供作为褐色泡沫的(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (430 mg, 0.509 mmol, 37%收率)。1H NMR (400 MHz, DMSO-d 6 ) δ ppm 8.14 (s, 1 H), 8.01 (br. s., 1 H), 7.78 (dd, J=8.4, 1.4 Hz, 1 H),7.45 (br. s., 1 H), 7.25-7.32 (m, 1 H), 7.13-7.23 (m, 4 H), 7.08 (d, J=8.0Hz, 1 H), 6.71 (d, J=8.5 Hz, 2 H), 6.43 (s, 1 H), 6.37 (s, 1 H), 5.03 (s, 2H), 4.97 (s, 2 H), 4.78 (s, 2 H), 4.45-4.53 (m, 4 H), 3.63 (s, 3 H), 3.60 (s,3 H), 3.54 (s, 3 H), 2.10 (s, 3 H), 2.09 (s, 3 H), 1.56 (s, 3 H), 1.19-1.31(m, 6 H), 1.18 (s, 3 H)。LCMS (m/z): 826.5 [M + H]+。To a solution of (E)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1-(4-(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-((4-methoxybenzyl)oxy)-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-1H-benzo[d]imidazole-5-carboxamide (1.09 g, 1.366 mmol) in DMF (15 mL) was added cesium carbonate (1.335 g, 4.10 mmol) and iodomethane (0.214 mL, 3.42 mmol). The reaction mixture was stirred at room temperature for 5 h. Additional iodomethane (0.060 mL, 0.956 mmol) was added and the mixture was stirred for an additional 18 h. Additional iodomethane (0.060 mL, 0.956 mmol) and cesium carbonate (1.335 g, 4.10 mmol) were added and the mixture was heated at 50°C for 4 h. The mixture was diluted with water (30 mL) and a sticky solid precipitated out. Vigorous stirring provided a filterable solid which was then collected on a filter and rinsed with water. Silica gel chromatography (40 g silica gel, 10-90% gradient of [3:1 EtOAc:EtOH]/heptane) afforded (E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (430 mg, 0.509 mmol, 37% yield) as a brown foam. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.14 (s, 1 H), 8.01 (br. s., 1 H), 7.78 (dd, J =8.4, 1.4 Hz, 1 H), 7.45 (br. s., 1 H), 7.25-7.32 (m, 1 H), 7.13-7.2 3 (m, 4 H), 7.08 (d, J =8.0Hz, 1 H), 6.71 (d, J =8.5 Hz, 2 H), 6.43 (s, 1 H), 6.37 (s, 1 H), 5.03 (s, 2H), 4.97 (s, 2 H), 4.78 (s, 2 H), 4.45-4.5 3 (m, 4 H), 3.63 (s, 3 H), 3.60 (s, 3 H), 3.54 (s, 3 H), 2.10 (s, 3 H), 2.09 (s, 3 H), 1.56 (s, 3 H), 1.19-1.31 (m, 6 H), 1.18 (s, 3 H). LCMS ( m/z ): 826.5 [M + H] + .
实施例30Embodiment 30
(E)-1-((E)-4-((E)-7-(苄氧基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-((E)-4-((E)-7-(Benzyloxy)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
向(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-羟基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (45 mg, 0.055mmol)在DMF (1 mL)中的悬浮液加入(溴甲基)苯(7.26 µL, 0.061 mmol),然后加入碳酸钾(9.97 mg, 0.072 mmol)。在50℃初始搅拌该反应混合物3 h,然后在室温搅拌16 h。用水稀释该混合物。用3:1 CHCl3:EtOH萃取水层几次。在真空中蒸发溶剂并通过质量引导的HPLC(XSELECT CSH C18, 5 um填充物, 150x30 mm柱, 30-85%梯度的MeCN/水,含0.1% TFA 改性剂)纯化残余物。将几滴饱和碳酸氢钠溶液加入各清洁级分。使用氮气流除去ACN。过滤悬浮固体,用水漂洗,并干燥以提供作为白色固体的纯(E)-1-((E)-4-((E)-7-(苄氧基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (30 mg, 0.037 mmol, 67%)。1H NMR (400 MHz, DMSO-d 6 ) δ ppm 8.12 (s, 1 H), 8.01 (br. s., 1 H), 7.76 (dd, J=8.4, 1.4 Hz, 1 H),7.44 (br. s., 1 H), 7.12-7.32 (m, 8 H), 7.07 (d, J=7.8 Hz, 1 H), 6.47 (s, 1H), 6.38 (s, 1 H), 5.13 (s, 2 H), 4.99 (s, 2 H), 4.78 (s, 2 H), 4.45-4.55 (m,4 H), 3.59 (s, 3 H), 3.56 (s, 3 H), 2.13 (s, 3 H), 2.10 (s, 3 H), 1.59 (s, 3H), 1.19-1.31 (m, 6 H), 1.19 (s, 3 H)。LCMS (m/z): 796.6 [M + H]+。To a suspension of (E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-hydroxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (45 mg, 0.055 mmol) in DMF (1 mL) was added (bromomethyl)benzene (7.26 µL, 0.061 mmol) followed by potassium carbonate (9.97 mg, 0.072 mmol). The reaction mixture was initially stirred at 50 °C for 3 h and then at room temperature for 16 h. The mixture was diluted with water. The aqueous layer was extracted several times with 3:1 CHCl 3 :EtOH. The solvent was evaporated in vacuo and the residue was purified by mass-guided HPLC (XSELECT CSH C18, 5 um packing, 150x30 mm column, 30-85% gradient of MeCN/water with 0.1% TFA modifier). A few drops of saturated sodium bicarbonate solution were added to each clean fraction. ACN was removed using a stream of nitrogen. The suspended solid was filtered, rinsed with water, and dried to provide pure (E)-1-((E)-4-((E)-7-(benzyloxy)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (30 mg, 0.037 mmol, 67%) as a white solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.12 (s, 1 H), 8.01 (br. s., 1 H), 7.76 (dd, J=8.4, 1.4 Hz, 1 H), 7.44 (br. s., 1 H), 7.12-7.32 (m, 8 H), 7.07 (d, J =7.8 Hz, 1 H), 6.47 (s, 1H), 6.38 (s, 1 H), 5.13 (s, 2 H), 4.99 (s, 2 H), 4.78 (s, 2 H), 4.45-4.55 (m, 4 H), 3.59 (s, 3 H), 3.56 (s, 3 H), 2.1 3 (s, 3 H), 2.10 (s, 3H), 1.59 (s, 3H), 1.19-1.31 (m, 6H), 1.19 (s, 3H). LCMS ( m/z ): 796.6 [M + H] + .
实施例31Embodiment 31
(E)-1-((E)-4-((E)-4-溴-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-((E)-4-((E)-4-bromo-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
步骤1: (E)-(4-((3-溴-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯Step 1: tert-Butyl (E)-(4-((3-bromo-2-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)carbamate
向在乙醇(11.67 mL)中的(E)-(4-氨基-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯 (0.5 g, 2.33 mmol)加入1-溴-3-氟-2-硝基苯 (0.529 g, 2.33 mmol)和DIEA (1.22mL, 7.00 mmol)。在80℃搅拌该混合物18 h。在乙酸乙酯 (50 mL)和盐水(20 mL)之间分配该混合物。用EtOAc (2 x 10 mL)萃取水层。经硫酸镁干燥合并的EtOAc层,过滤并浓缩。通过硅胶层析法 (40 g硅胶,10-20% 乙酸乙酯/己烷的梯度)纯化残余物以获得(E)-(4-((3-溴-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯 (0.740 g, 1.78mmol, 77%收率)。LCMS (m/z): 414.1 [M + H]+。To tert-butyl (E)-(4-amino-2,3-dimethylbut-2-en-1-yl)carbamate (0.5 g, 2.33 mmol) in ethanol (11.67 mL) was added 1-bromo-3-fluoro-2-nitrobenzene (0.529 g, 2.33 mmol) and DIEA (1.22 mL, 7.00 mmol). The mixture was stirred at 80 °C for 18 h. The mixture was partitioned between ethyl acetate (50 mL) and brine (20 mL). The aqueous layer was extracted with EtOAc (2 x 10 mL). The combined EtOAc layers were dried over magnesium sulfate, filtered and concentrated. The residue was purified by silica gel chromatography (40 g silica gel, gradient of 10-20% ethyl acetate/hexanes) to afford tert-butyl (E)-(4-((3-bromo-2-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)carbamate (0.740 g, 1.78 mmol, 77% yield). LCMS ( m/z ): 414.1 [M + H] + .
步骤2: (E)-4-((4-((3-溴-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺Step 2: (E)-4-((4-((3-bromo-2-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide
向在甲醇 (5 mL)中的(E)-(4-((3-溴-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基甲酸叔丁酯 (0.44 g, 1.06 mmol)加入在二氧杂环己烷中的4 M HCl(1.06 mL,4.25 mmol)。在室温搅拌该混合物3 h。加入另外的在二氧杂环己烷中的4 M HCl (1.0 mL,4.0 mmol)并再搅拌3小时。在真空中浓缩该反应混合物以除去HCl和溶剂。向这个残余物加入4-氟-3-硝基苯甲酰胺 (0.214 g, 1.16 mmol)、DIEA (0.927 mL, 5.31 mmol)和1-丁醇(15 ml)并在110℃搅拌该混合物16 h。在乙酸乙酯 (50 mL)和盐水(20 mL)之间分配该混合物。用EtOAc (2 x 10 mL)萃取水层。经硫酸镁干燥合并的 EtOAc层,过滤并浓缩。浓缩提供沉淀物,将其过滤并干燥以提供作为固体的(E)-4-((4-((3-溴-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺 (0.40 g, 0.836 mmol, 79%收率)。LCMS(m/z): 478.1 [M + H]+。To tert-butyl (E)-(4-((3-bromo-2-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)carbamate (0.44 g, 1.06 mmol) in methanol (5 mL) was added 4 M HCl in dioxane (1.06 mL, 4.25 mmol). The mixture was stirred at room temperature for 3 h. Additional 4 M HCl in dioxane (1.0 mL, 4.0 mmol) was added and stirred for another 3 hours. The reaction mixture was concentrated in vacuo to remove HCl and solvent. To this residue was added 4-fluoro-3-nitrobenzamide (0.214 g, 1.16 mmol), DIEA (0.927 mL, 5.31 mmol) and 1-butanol (15 ml) and the mixture was stirred at 110°C for 16 h. The mixture was partitioned between ethyl acetate (50 mL) and brine (20 mL). The aqueous layer was extracted with EtOAc (2 x 10 mL). The combined EtOAc layers were dried over magnesium sulfate, filtered and concentrated. Concentration provided a precipitate, which was filtered and dried to provide (E)-4-((4-((3-bromo-2-nitrophenyl)amino)-2,3-dimethylbut-2-ene-1-yl)amino)-3-nitrobenzamide (0.40 g, 0.836 mmol, 79% yield) as a solid. LCMS ( m/z ): 478.1 [M + H] + .
步骤3: (E)-3-氨基-4-((4-((2-氨基-3-溴苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)苯甲酰胺Step 3: (E)-3-Amino-4-((4-((2-amino-3-bromophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)benzamide
向在甲醇 (1.6 mL)中的(E)-4-((4-((3-溴-2-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺 ( 0.050 g, 0.105 mmol)加入28-30% 氢氧化铵溶液(0.102 mL, 2.61 mmol)和连二亚硫酸钠溶液(0.214 g, 1.045 mmol, 在0.8 mL水中)。在室温搅拌该混合物2 h。在乙酸乙酯 (20 mL)和盐水(20 mL)之间分配该反应混合物。用EtOAc (2 x 10 mL)萃取水层。经硫酸钠干燥合并的EtOAc层,过滤并浓缩。通过硅胶层析法(12 g硅胶,10-40%梯度的 (3:1乙醇/乙酸乙酯)/庚烷,其使用2% NH4OH 改性剂)的纯化提供(E)-3-氨基-4-((4-((2-氨基-3-溴苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)苯甲酰胺 (0.030 g, 0.070 mmol, 67.2%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 7.42 (br.s., 1 H), 7.01-7.15 (m, 2 H), 6.70 (d, J=7.10 Hz, 2 H), 6.38-6.47 (m, 1 H),6.32 (dd, J=7.86, 14.95 Hz, 2 H), 5.01 (t, J=5.07 Hz, 1 H), 4.93 (t, J=5.07Hz, 1 H), 4.74 (s, 2 H), 4.63 (s, 2 H), 3.76 (d, J=4.82 Hz, 2 H), 3.71 (d, J=5.07 Hz, 2 H), 1.78 (br. s., 3 H), 1.77 (br. s., 3 H)。To (E)-4-((4-((3-bromo-2-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide (0.050 g, 0.105 mmol) in methanol (1.6 mL) was added 28-30% ammonium hydroxide solution (0.102 mL, 2.61 mmol) and sodium dithionite solution (0.214 g, 1.045 mmol, in 0.8 mL water). The mixture was stirred at room temperature for 2 h. The reaction mixture was partitioned between ethyl acetate (20 mL) and brine (20 mL). The aqueous layer was extracted with EtOAc (2 x 10 mL). The combined EtOAc layers were dried over sodium sulfate, filtered and concentrated. Purification by silica gel chromatography (12 g silica gel, 10-40% gradient of (3:1 ethanol/ethyl acetate)/heptane using 2% NH4OH modifier) provided (E)-3-amino-4-((4-((2-amino-3-bromophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)benzamide (0.030 g, 0.070 mmol, 67.2% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 7.42 (br.s., 1 H), 7.01-7.15 (m, 2 H), 6.70 (d, J =7.10 Hz, 2 H), 6.38-6.47 (m, 1 H), 6.32 (dd, J =7.86, 14.95 Hz, 2 H), 5.01 (t, J =5.07 Hz, 1 H), 4.93 (t, J =5.07Hz, 1 H), 4.74 (s, 2 H), 4.63 (s, 2 H), 3.76 (d, J =4.82 Hz, 2 H), 3.71 (d, J =5.07 Hz, 2 H), 1.78 (br. s., 3 H), 1.77 (br. s., 3 H).
步骤4: (E)-1-(4-(4-溴-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 4: (E)-1-(4-(4-bromo-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-5-carboxamide
在0℃向在DMF (0.72 mL)中的(E)-3-氨基-4-((4-((2-氨基-3-溴苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)苯甲酰胺 (0.030 g, 0.072 mmol)加入1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯的溶液(1 M 在二氧杂环己烷中, 0.158 mL, 0.158 mmol)。在0℃搅拌该反应混合物1 h。加入EDC (0.043 g, 0.215 mmol)和三乙胺 (0.060 mL, 0.430mmol)并在室温搅拌该混合物16 h。将该反应倾倒至水(10 mL)中并搅拌。将所得固体过滤并在50℃在真空炉中干燥过夜以获得(E)-1-(4-(4-溴-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (0.043 g, 0.056 mmol, 79%收率)。LCMS(m/z): 740.5 [M + H]+。To (E)-3-amino-4-((4-((2-amino-3-bromophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)benzamide (0.030 g, 0.072 mmol) in DMF (0.72 mL) at 0°C was added a solution of 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (1 M in dioxane, 0.158 mL, 0.158 mmol). The reaction mixture was stirred at 0°C for 1 h. EDC (0.043 g, 0.215 mmol) and triethylamine (0.060 mL, 0.430 mmol) were added and the mixture was stirred at room temperature for 16 h. The reaction was poured into water (10 mL) and stirred. The resulting solid was filtered and dried in a vacuum oven at 50 °C overnight to afford (E)-1-(4-(4-bromo-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-5-carboxamide (0.043 g, 0.056 mmol, 79% yield). LCMS ( m/z ): 740.5 [M + H] + .
步骤5: (E)-1-((E)-4-((E)-4-溴-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 5: (E)-1-((E)-4-((E)-4-bromo-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向在DMF (0.54 mL)中的(E)-1-(4-(4-溴-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (0.040 g, 0.054 mmol)加入碳酸铯 (0.053 g,0.162 mmol)和碘甲烷(7.4 µL, 0.12 mmol)。在室温搅拌该混合物16 h。在乙酸乙酯 (10mL)和盐水(5 mL)之间分配该混合物。用EtOAc (10 mL)萃取水层。经硫酸镁干燥合并的EtOAc层,过滤并浓缩。通过质量导向的制备型HPLC (XSELECT CSH C18, 5 um填充物,150x30 mm柱, 30-85%梯度的MeCN/水,含0.075% NH4OH, 10 mM碳酸氢铵, pH 10)纯化该残余物以获得(E)-1-((E)-4-((E)-4-溴-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (0.013 g,0.016 mmol, 30%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.11 (d, J=1.27 Hz, 1 H),8.04 (br. s., 1 H), 7.80 (dd, J=1.39, 8.49 Hz, 1 H), 7.49-7.53 (m, 1 H), 7.46(br. s., 1 H), 7.28 (t, J=8.49 Hz, 2 H), 7.11-7.18 (m, 1 H), 6.45 (s, 1 H),6.42 (s, 1 H), 4.85 (br. s., 4 H), 4.51 (dq, J=3.68, 6.97 Hz, 4 H), 3.78 (s,3 H), 3.59 (s, 3 H), 2.128 (s, 3 H), 2.125 (s, 3 H), 1.59 (s, 6 H), 1.25-1.35(m, 6 H)。LCMS (m/z): 768.4 [M + H]+。To (E)-1-(4-(4-bromo-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-5-carboxamide (0.040 g, 0.054 mmol) in DMF (0.54 mL) was added cesium carbonate (0.053 g, 0.162 mmol) and iodomethane (7.4 µL, 0.12 mmol). The mixture was stirred at room temperature for 16 h. The mixture was partitioned between ethyl acetate (10 mL) and brine (5 mL). The aqueous layer was extracted with EtOAc (10 mL). The combined EtOAc layers were dried over magnesium sulfate, filtered and concentrated. The residue was purified by mass-directed preparative HPLC (XSELECT CSH C18, 5 um packing, 150x30 mm column, 30-85% gradient of MeCN/water with 0.075% NH4OH , 10 mM ammonium bicarbonate, pH 10) to afford (E)-1-((E)-4-((E)-4-bromo-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (0.013 g, 0.016 mmol, 30% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.11 (d, J =1.27 Hz, 1 H), 8.04 (br. s., 1 H), 7.80 (dd, J =1.39, 8.49 Hz, 1 H), 7.49-7.53 (m, 1 H), 7.46 (br. s., 1 H) ), 7.28 (t, J =8.49 Hz, 2 H), 7.11-7.18 (m, 1 H), 6.45 (s, 1 H), 6.42 (s, 1 H), 4.85 (br. s., 4 H), 4.51 (dq, J =3.68, 6.97 Hz, 4 H), 3.78 (s, 3 H) , 3.59 (s, 3 H), 2.128 (s, 3 H), 2.125 (s, 3 H), 1.59 (s, 6 H), 1.25-1.35 (m, 6 H). LCMS ( m/z ): 768.4 [M + H] + .
实施例32和33Examples 32 and 33
实施例32: (E)-1-((2R,3R)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (较早洗脱出的对映异构体)Example 32: (E)-1-((2R,3R)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropyloxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (earlier eluting enantiomer)
实施例33: (E)-1-((2S,3S)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (较晚洗脱出的对映异构体)Example 33: (E)-1-((2S,3S)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropyloxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (later eluting enantiomer)
步骤1: (E)-1-(4-((2-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基-6-硝基苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺Step 1: (E)-1-(4-((2-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoyl-6-nitrophenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide
在室温向(E)-1-(4-氨基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐 (10.4 g, 21.47 mmol)在1-丁醇(150 mL)中的悬浮液加入DIEA (7.50 mL, 42.9 mmol)。然后,在室温搅拌该反应混合物2h。加入3-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氯-5-硝基苯甲酰胺 (10.9 g,28.0 mmol),然后加入碳酸氢钠(5.41 g, 64.4 mmol)。然后在100℃搅拌该反应混合物4天。将该反应混合物冷却至室温并浓缩。将所得橙色泥状物悬浮在乙腈中,然后过滤。用乙腈和水洗涤固体。然后,干燥该固体以获得作为橙红色固体的(E)-1-(4-((2-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基-6-硝基苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺 (10.94 g,14.32 mmol, 67%收率)。1H NMR (400MHz, DMSO-d 6) δ ppm 13.27 - 12.18 (m, 1 H),8.14 (d, J=1.8 Hz, 1 H), 7.97 (br. s., 2 H), 7.78 - 7.62 (m, 2 H), 7.49 (d, J=1.8 Hz, 1 H), 7.38 - 7.24 (m, 3 H), 6.60 (s, 1 H), 5.92 - 5.63 (m, 2 H),4.90 (d, J=5.3 Hz, 2 H), 4.58 (q, J=7.1 Hz, 2 H), 4.13 (t, J=5.4 Hz, 2 H),4.00 (t, J=6.0 Hz, 2 H), 3.87 (s, 3 H), 3.64 (t, J=6.1 Hz, 2 H), 2.16 (s, 3H), 1.82 (五重峰, J=6.0 Hz, 2 H), 1.33 (t, J=7.0 Hz, 3 H), 0.79 (s, 9 H), -0.05 (s, 6 H)。LCMS (m/z): 764.7 [M + H]+。To a suspension of (E)-1-(4-aminobut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide, 2 hydrochloride (10.4 g, 21.47 mmol) in 1-butanol (150 mL) at room temperature was added DIEA (7.50 mL, 42.9 mmol). The reaction mixture was then stirred at room temperature for 2 h. 3-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-chloro-5-nitrobenzamide (10.9 g, 28.0 mmol) was added, followed by sodium bicarbonate (5.41 g, 64.4 mmol). The reaction mixture was then stirred at 100 °C for 4 days. The reaction mixture was cooled to room temperature and concentrated. The resulting orange sludge was suspended in acetonitrile and then filtered. The solid was washed with acetonitrile and water. Then, the solid was dried to obtain (E)-1-(4-((2-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoyl-6-nitrophenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide (10.94 g, 14.32 mmol, 67% yield) as an orange-red solid. 1 H NMR (400MHz, DMSO- d 6 ) δ ppm 13.27 - 12.18 (m, 1 H), 8.14 (d, J =1.8 Hz, 1 H), 7.97 (br. s., 2 H), 7.78 - 7.62 (m, 2 H), 7.49 (d, J =1.8 Hz, 1 H), 7 .38 - 7.24 (m, 3 H), 6.60 (s, 1 H), 5.92 - 5.63 (m, 2 H), 4.90 (d, J =5.3 Hz, 2 H), 4.58 (q, J =7.1 Hz, 2 H), 4.13 (t, J =5.4 Hz, 2 H), 4.00 (t, J =6.0 Hz, 2H), 3.87 (s, 3 H), 3.64 (t, J =6.1 Hz, 2 H), 2.16 (s, 3H), 1.82 (quint, J =6.0 Hz, 2 H), 1.33 (t, J =7.0 Hz, 3 H), 0.79 (s, 9 H), -0.05 (s, 6 H). LCMS ( m/z ): 764.7 [M + H] + .
步骤2: (E)-1-(4-((2-氨基-6-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺Step 2: (E)-1-(4-((2-amino-6-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoylphenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide
向(E)-1-(4-((2-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基-6-硝基苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺 (10.1 g, 13.22 mmol)在60℃搅拌的甲醇 (200 mL)中的溶液加入连二亚硫酸钠 (25.0 g, 121 mmol)在水(200 mL)中的溶液。然后,在同样的温度下搅拌该反应混合物1 h。然后,将该反应混合物冷却至室温并用 500 mL水淬灭。过滤所得混合物。用水(500 mL x 3)洗涤收集的固体,然后用二乙醚 (300 mL)漂洗。然后,在真空炉中干燥该固体以获得作为浅棕色固体的部分纯的(E)-1-(4-((2-氨基-6-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺 (7.36 g, ~10 mmol, ~76%收率)。通过LCMS测得的标题化合物的大致纯度为63% (UV, m/z = 734.6 [M + H]+)以及甲硅烷基-脱保护副产物的20% (UV, m/z = 620.5 [M + H]+)。不经进一步纯化即将该混合物用于接下来的反应。To a solution of (E)-1-(4-((2-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoyl-6-nitrophenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide (10.1 g, 13.22 mmol) in methanol (200 mL) stirred at 60 °C was added a solution of sodium dithionite (25.0 g, 121 mmol) in water (200 mL). The reaction mixture was then stirred at the same temperature for 1 h. The reaction mixture was then cooled to room temperature and quenched with 500 mL of water. The resulting mixture was filtered. The collected solid was washed with water (500 mL x 3) and then rinsed with diethyl ether (300 mL). The solid was then dried in a vacuum oven to afford partially pure (E)-1-(4-((2-amino-6-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoylphenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide (7.36 g, ~10 mmol, ~76% yield) as a light brown solid. The approximate purity of the title compound by LCMS was 63% (UV, m/z = 734.6 [M + H] + ) and 20% of the silyl-deprotected byproduct (UV, m/z = 620.5 [M + H] + ). The mixture was used in the next reaction without further purification.
步骤3: (E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 3: (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide
在室温经由加料漏斗向(E)-1-(4-((2-氨基-6-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-4-氨基甲酰基苯基)氨基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺 (7.36 g, 10.03 mmol, ~76%纯度)在DMF(60 mL)中的溶液逐滴加入1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (15.1 mL, 15.10mmol)在1,4 二氧杂环己烷中的1M 溶液。然后,在室温搅拌该反应混合物1 h。然后,在室温向该反应混合物加入EDC (3.84 g, 20.06 mmol)和TEA (5.6 mL, 40.2 mmol)。在室温搅拌该反应混合物48 h。用EtOAc稀释该反应混合物并用水洗涤。通过过滤除去固体并被识别为甲硅烷基-脱保护的醇衍生物。然后,第二次洗涤有机层。用EtOAc (1x)反萃取合并的水层。用盐水洗涤合并的有机层,用硫酸镁干燥和浓缩。将在处理中较早分离的粗产物和固体悬浮在EtOH中并过滤。用EtOH洗涤该固体,然后干燥以获得灰白色固体(6.0 g)。还获得第二批固体(824 mg)。通过LCMS和NMR表征合并的混合物(6.824 g, ~7.6 mmol, ~76%)的组成为大致3:1下列的混合物: (E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺(LCMS (m/z): 895.8 [M + H]+)和醇衍生物(E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-羟基丙氧基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺 (LCMS (m/ z): 781.7 [M + H]+)。不经进一步纯化即将该混合物用于接下来的反应中。To a solution of (E)-1-(4-((2-amino-6-(3-((tert-butyldimethylsilyl)oxy)propoxy)-4-carbamoylphenyl)amino)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide (7.36 g, 10.03 mmol, ~76% purity) in DMF (60 mL) was added a 1 M solution of 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (15.1 mL, 15.10 mmol) in 1,4-dioxane dropwise via an addition funnel at room temperature. The reaction mixture was then stirred at room temperature for 1 h. Then, EDC (3.84 g, 20.06 mmol) and TEA (5.6 mL, 40.2 mmol) were added to the reaction mixture at room temperature. The reaction mixture was stirred at room temperature for 48 h. The reaction mixture was diluted with EtOAc and washed with water. Solids were removed by filtration and identified as alcohol derivatives of silyl-deprotection. Then, the organic layer was washed for the second time. The water layer merged was extracted with EtOAc (1x). The organic layer merged was washed with salt water, dried over magnesium sulfate and concentrated. The crude product and solid separated earlier in the process were suspended in EtOH and filtered. The solid was washed with EtOH, then dried to obtain a pale solid (6.0 g). A second batch of solids (824 mg) was also obtained. The combined mixture (6.824 g, ~7.6 mmol, ~76%) was characterized by LCMS and NMR as a roughly 3:1 mixture of (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (LCMS ( m/z ): 895.8 [M + H] + ) and the alcohol derivative (E)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-hydroxypropoxy)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazol-5-carboxamide (LCMS ( m/ z ): 781.7 [M+H] + ). The mixture was used in the next reaction without further purification.
步骤4: (E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-羟基丙氧基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐Step 4: (E)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-hydroxypropoxy)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide, 2-hydrochloride
在室温向(E)-7-(3-((叔丁基二甲基甲硅烷基)氧基)丙氧基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (6.0 g,6.70 mmol, 作为3:1 甲硅烷基醚/醇的混合物)在THF (50 mL)中的溶液加入在二氧杂环己烷中的4M HCl 溶液 (8.4 mL, 33.6 mmol)。然后,在室温搅拌该反应混合物5 h。加入另外的HCl溶液(4.2 mL, 16.80 mmol)并搅拌该反应混合物过夜。加入另外的HCl溶液(4.2mL, 16.80 mmol)并搅拌该反应混合物24 h。加入另外的HCl溶液(8.4 mL, 33.6 mmol)。在室温搅拌该混合物1 h,然后在40℃搅拌5 h。加入另外的HCl溶液(8.4 mL, 33.6 mmol)并在40℃搅拌该反应混合物过周末。将该反应混合物冷却至室温,然后过滤。用THF洗涤固体并干燥以获得作为白色固体的(E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-羟基丙氧基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐 (5.6 g, 6.56mmol, 98%收率)。1H NMR (400MHz, DMSO- d 6) δ ppm 7.98 (br. s., 2 H), 7.65 (dd, J=1.1, 3.6 Hz, 2 H), 7.33 (s, 4 H), 6.53 (d, J=1.5 Hz, 2 H), 5.86 - 5.81 (m, 2H), 4.93 (dd, J=3.8, 6.8 Hz, 4 H), 4.57 - 4.47 (m, 4 H), 4.07 (t, J=6.4 Hz, 2H), 3.75 (s, 3 H), 3.49 - 3.42 (m, 2 H), 2.11 (two s, 6 H), 1.80 - 1.64 (m, 2H), 1.27 (two t, J=7.2 Hz, 6 H)。LCMS (m/z): 781.7 [M + H]+。To a solution of (E)-7-(3-((tert-butyldimethylsilyl)oxy)propoxy)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (6.0 g, 6.70 mmol, as a 3:1 mixture of silyl ethers/alcohols) in THF (50 mL) was added 4M HCl solution in dioxane (8.4 mL, 33.6 mmol) at room temperature. The reaction mixture was then stirred at room temperature for 5 h. Additional HCl solution (4.2 mL, 16.80 mmol) was added and the reaction mixture was stirred overnight. Add additional HCl solution (4.2 mL, 16.80 mmol) and stir the reaction mixture for 24 h. Add additional HCl solution (8.4 mL, 33.6 mmol). Stir the mixture at room temperature for 1 h and then at 40 °C for 5 h. Add additional HCl solution (8.4 mL, 33.6 mmol) and stir the reaction mixture at 40 °C over the weekend. Cool the reaction mixture to room temperature and then filter. The solid was washed with THF and dried to afford (E)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-hydroxypropoxy)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide, 2 hydrochloride (5.6 g, 6.56 mmol, 98% yield) as a white solid. 1 H NMR (400MHz, DMSO- d 6 ) δ ppm 7.98 (br. s., 2 H), 7.65 (dd, J =1.1, 3.6 Hz, 2 H), 7.33 (s, 4 H), 6.53 (d, J =1.5 Hz, 2 H), 5.86 - 5.81 (m, 2H), 4.9 3 (dd, J =3.8, 6.8 Hz, 4 H), 4.57 - 4.47 (m, 4 H), 4.07 (t, J =6.4 Hz, 2H), 3.75 (s, 3 H), 3.49 - 3.42 (m, 2 H), 2.11 (two s, 6 H), 1.80 - 1.64 (m, 2H), 1.27 (two t, J =7.2 Hz, 6 H). LCMS ( m/z ): 781.7 [M + H] + .
步骤5: (E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 5: (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
通过使用冰浴在0℃向(E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-羟基丙氧基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺, 2盐酸盐 (5.45 g,6.38 mmol)和碳酸铯 (10.40 g, 31.9 mmol)在DMF (35 mL)中的悬浮液加入碘甲烷(0.918 mL, 14.68 mmol)。移去冰浴并在室温搅拌该混合物16 h。在水和3:1 氯仿/乙醇之间分配该混合物。分离各层。 采用3:1 氯仿/乙醇(6X)萃取水层。用水洗涤有机层。再次用3:1 氯仿/乙醇萃取这个水性洗涤溶液。用硫酸钠干燥合并的有机层,过滤,并浓缩以提供标题化合物(6 g)。通过LCMS测得的标题化合物的大致纯度为76% (UV210-350nm, m/z =809.3 [M + H]+)。不经进一步纯化即将这个化合物用于接下来的反应中。To a suspension of (E)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-hydroxypropoxy)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide, 2 hydrochloride (5.45 g, 6.38 mmol) and cesium carbonate (10.40 g, 31.9 mmol) in DMF (35 mL) was added iodomethane (0.918 mL, 14.68 mmol) by using an ice bath at 0°C. The ice bath was removed and the mixture was stirred at room temperature for 16 h. The mixture was partitioned between water and 3:1 chloroform/ethanol. The layers were separated. The aqueous layer was extracted with 3:1 chloroform/ethanol (6X). The organic layer was washed with water. The aqueous wash solution was extracted again with 3:1 chloroform/ethanol. The combined organic layers were dried over sodium sulfate, filtered, and concentrated to provide the title compound (6 g). The approximate purity of the title compound measured by LCMS was 76% (UV210-350nm, m/z =809.3 [M + H] + ). This compound was used in the next reaction without further purification.
步骤6: (E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 6: (E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropyloxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (5.2 g, 6.43 mmol)在叔丁醇 (52 mL)和水(13 mLl)中的悬浮液加入NMO(1.130 g, 9.64 mmol)。在室温搅拌5 min后,加入叔丁醇中的2.5% 四氧化锇 (4.04 mL,0.321 mmol)并搅拌该混合物18 h。在浓缩后,将该残余物悬浮在水中并过滤。用水漂洗收集的固体并在真空中干燥以提供作为立体异构体混合物的(E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (4.0 g, 73.8%收率)。LCMS (通过UV210-350nm测得~90%纯度; m/z): 843.2 [M + H]+。To a suspension of (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (5.2 g, 6.43 mmol) in tert-butanol (52 mL) and water (13 mLl) was added NMO (1.130 g, 9.64 mmol). After stirring at room temperature for 5 min, 2.5% osmium tetroxide (4.04 mL, 0.321 mmol) in the tert-butyl alcohol is added and the mixture is stirred for 18 h. After concentration, the residue is suspended in water and filtered. The collected solid is rinsed with water and dried in a vacuum to provide (E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl) imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo [d] imidazoles-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazoles-5-carbonyl) imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo [d] imidazoles-5-carboxamide (4.0 g, 73.8% yield) as a mixture of stereoisomers. LCMS (~90% purity by UV 210-350 nm; m/z ): 843.2 [M + H] + .
步骤7: 较早洗脱出的对映异构体 实施例33 (E)-1-((2R,3R)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺和较晚洗脱出的对映异构体实施例34 (E)-1-((2S,3S)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺的纯化Step 7: Earlier eluting enantiomer Example 33 (E)-1-((2R,3R)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropyloxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide and later eluting enantiomer Example 34 Purification of (E)-1-((2S,3S)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
为了登记和筛选,通过具有两个制备型HPLC纯化步骤的序列将1500 mg份的粗产物分成独立的对映异构体。For registration and screening, 1500 mg portions of the crude product were separated into the individual enantiomers by a sequence with two preparative HPLC purification steps.
第一纯化步骤First purification step
这个步骤的目的是将所需的对映异构体的外消旋混合物与几种次要副产物(在对映异构体的共洗脱对附近被洗脱)分离。使用以下方法:The purpose of this step is to separate the racemic mixture of the desired enantiomer from several minor byproducts (eluting near the co-eluting pair of enantiomers). Use the following method:
。 .
结果:将最纯的级分合并并浓缩至10 mL体积。将所得沉淀物过滤并在35℃干燥以提供对映异构体的外消旋混合物(600 mg, 0.71 mmol)。C18 HPLC方法(方法2)还用于表明纯化效果(纯度98.95%)。LCMS (m/z): 843.3 [M + H]+。具有稍微较低纯度的级分(即前级分和尾级分)的类似处理提供另外的量的对映异构体的外消旋混合物(310 mg)。Results: The purest fractions were combined and concentrated to a volume of 10 mL. The resulting precipitate was filtered and dried at 35 °C to provide a racemic mixture of enantiomers (600 mg, 0.71 mmol). The C18 HPLC method (Method 2) was also used to demonstrate the purification effect (purity 98.95%). LCMS ( m/z ): 843.3 [M+H] + . Similar treatment of fractions with slightly lower purity (i.e., front fraction and tail fraction) provided an additional amount of a racemic mixture of enantiomers (310 mg).
第二纯化步骤:这个步骤的目的是分离和分开各个对映异构体。使用以下方法:Second purification step: The purpose of this step is to separate and isolate the individual enantiomers. The following method was used:
。 .
结果:将各个对映异构体的纯级分浓缩并在高真空下在40℃干燥以提供以下固体。通过随后由手性构建模块(chiral building blocks)再次合成能够获得绝对立体化学的分配。Results: The pure fractions of each enantiomer were concentrated and dried under high vacuum at 40°C to afford the following solid. The assignment of absolute stereochemistry can be obtained by subsequent re-synthesis from chiral building blocks.
实施例32 (第一个洗脱出的异构体):Example 32 (first eluting isomer):
(E)-1-((2R,3R)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (247 mg, 0.293 mmol),其为白色固体。1H NMR (400 MHz, DMSO-d 6) δppm 1.26 (t, J=7.1 Hz, 3H) 1.27 (t, J=7.1 Hz, 3H) 1.83 (五重峰, J=6.15 Hz, 2H) 2.10 (s, 3 H) 2.11 (s, 3 H) 3.50 (s, 3 H) 3.50 - 3.55 (m, 5 H) 3.80 (s, 3H) 3.81 - 3.91 (m, 2 H) 4.18 (t, J=6.46 Hz, 2 H) 4.24 - 4.33(m, 2 H) 4.44 -4.53 (m, 5 H) 4.53 - 4.60 (m, 2 H) 4.97 (d, J=6.84 Hz, 1 H) 5.06 (d, J=6.34Hz, 1 H) 6.41(s, 1 H) 6.44 (s, 1 H) 7.41 - 7.50 (m, 4 H) 7.71 (dd, J=2.66,1.14 Hz, 2 H) 8.06 (br. s., 2 H)。LCMS (通过UV210-350nm测得的100%纯度; m/z):843.3 [M + H]+。通过手性分析型HPLC测得的对映异构体过量> 98 % ee。αD20 - 24 (c0.1, MeOH)。(E)-1-((2R,3R)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (247 mg, 0.293 mmol) as a white solid. 1 H NMR (400 MHz, DMSO- d 6 ) δppm 1.26 (t, J =7.1 Hz, 3H) 1.27 (t, J =7.1 Hz, 3H) 1.83 (quint, J =6.15 Hz, 2H) 2.10 (s, 3 H) 2.11 (s, 3 H) 3.50 (s, 3 H) 3.50 - 3.55 (m, 5 H) 3.80 (s, 3H) 3.81 - 3.91 (m, 2 H) 4.18 (t, J =6.46 Hz, 2 H) 4.24 - 4.33(m, 2 H) 4.44 -4.53 (m, 5 H) 4.53 - 4.60 (m, 2 H) 4.97 (d, J =6.84 Hz, 1 H) 5.06 (d, J =6.34 Hz, 1 H) 6.41 (s, 1 H) 6.44 (s, 1 H) 7.41 - 7.50 (m, 4 H) 7.71 (dd, J =2.66,1.14 Hz, 2 H) 8.06 (br. s., 2 H). LCMS (100% purity by UV210-350 nm; m/z ): 843.3 [M + H] + . Enantiomeric excess > 98 % ee by chiral analytical HPLC. αD 20 - 24 ( c 0.1, MeOH).
实施例33 (第二个洗脱出的异构体):Example 33 (second eluting isomer):
(E)-1-((2S,3S)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (227 mg, 0.269 mmol),其为白色固体。1H NMR (400 MHz, DMSO-d 6) δppm 1.26 (t, J=7.1 Hz, 3 H) 1.27 (t, J=7.1 Hz, 3 H) 1.83 (五重峰, J=6.21 Hz,2 H) 2.10 (s, 3 H) 2.11 (s, 3 H) 3.50 (s, 3 H) 3.50 - 3.56 (m, 5 H) 3.80 (s,3 H) 3.81 - 3.92 (m, 2 H) 4.18 (t, J=6.34 Hz, 2 H) 4.24 - 4.33 (m, 2 H) 4.44- 4.52 (m, 5 H) 4.53 - 4.61 (m, 2 H) 4.97 (d, J=6.84 Hz, 1 H) 5.06 (d, J=6.34Hz, 1 H) 6.41 (s, 1 H) 6.44 (s, 1 H) 7.41 - 7.50 (m, 4 H) 7.71 (dd, J=2.53,1.01 Hz, 2 H) 8.06 (br. s., 2 H)。LCMS (通过UV210-350nm测得的100%纯度; m/z):843.3 [M + H]+。通过手性分析型HPLC测得的对映异构体过量98 % ee。αD20 + 23 (c 0.1,MeOH)。(E)-1-((2S,3S)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (227 mg, 0.269 mmol) as a white solid. 1 H NMR (400 MHz, DMSO- d 6 ) δppm 1.26 (t, J =7.1 Hz, 3 H) 1.27 (t, J =7.1 Hz, 3 H) 1.83 (quint, J =6.21 Hz, 2 H) 2.10 (s, 3 H) 2.11 (s, 3 H) 3.50 (s, 3 H) 3.50 - 3.56 (m, 5 H) 3.80 (s, 3 H) 3.81 - 3.92 (m, 2 H) 4.18 (t, J =6.34 Hz, 2 H) 4.24 - 4.33 (m, 2 H) 4.44- 4.52 (m, 5 H) 4.53 - 4.61 (m, 2 H) 4.97 (d, J =6.84 Hz, 1 H) 5.06 (d, J =6.34 Hz, 1 H) 6.41 (s, 1 H) 6.44 (s, 1 H) 7.41 - 7.50 (m, 4 H) 7.71 (dd, J =2.53,1.01 Hz, 2 H) 8.06 (br. s., 2 H). LCMS (100% purity by UV210-350 nm; m/z ): 843.3 [M + H] + . Enantiomeric excess 98 % ee by chiral analytical HPLC. αD 20 + 23 ( c 0.1, MeOH).
实施例34Embodiment 34
(E)-1-(4-((E)-7-溴-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-(4-((E)-7-bromo-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
步骤1: (E)-(4-((2-溴-6-硝基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯Step 1: tert-Butyl (E)-(4-((2-bromo-6-nitrophenyl)amino)but-2-en-1-yl)carbamate
在25℃向(E)-(4-氨基丁-2-烯-1-基)氨基甲酸叔丁酯 (2.2 g, 11.81 mmol)和DIEA (4.37 mL, 25.00 mmol)在异丙醇 (30 mL)中的溶液加入1-溴-2-氟-3-硝基苯 (2.5g, 11.36 mmol)。然后,在25℃搅拌该反应混合物4天。浓缩该反应混合物。在水和EtOAc之间分配所得物质。分离水层并用EtOAc (1x)萃取。然后,用盐水洗涤合并的有机层,用硫酸镁干燥,并浓缩以获得作为黄色固体的(E)-(4-((2-溴-6-硝基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯 (4.6 g, 12 mmol, 100%收率)。不经任何进一步纯化即使用经分离的物质。LCMS (m/z): 332.0 ([M + H]+ - 叔丁基)。To a solution of (E)-(4-aminobutan-2-ene-1-yl)t-butylcarbamate (2.2 g, 11.81 mmol) and DIEA (4.37 mL, 25.00 mmol) in isopropanol (30 mL) was added 1-bromo-2-fluoro-3-nitrobenzene (2.5 g, 11.36 mmol) at 25 ° C. Then, the reaction mixture was stirred at 25 ° C for 4 days. The reaction mixture was concentrated. The resulting material was distributed between water and EtOAc. The aqueous layer was separated and extracted with EtOAc (1x). Then, the combined organic layer was washed with brine, dried over magnesium sulfate, and concentrated to obtain (E)-(4-((2-bromo-6-nitrophenyl)amino)butan-2-ene-1-yl)t-butylcarbamate (4.6 g, 12 mmol, 100% yield) as a yellow solid. The isolated material was used without any further purification. LCMS ( m/z ): 332.0 ([M+H] + -tert-butyl).
步骤2: (E)-(4-((2-氨基-6-溴苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯Step 2: tert-Butyl (E)-(4-((2-amino-6-bromophenyl)amino)but-2-en-1-yl)carbamate
向(E)-(4-((2-溴-6-硝基苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯 (4.4 g,11.39 mmol)和氯化铵 (6.09 g, 114 mmol)在甲醇 (50 mL)中的混合物加入锌(7.45 g,114 mmol)。然后,在室温搅拌该反应混合物30 min。将该反应混合物过滤并浓缩。在EtOAc和水之间分配经分离的残余物。分离水层并用水洗涤有机层第二次。用EtOAc (1x)萃取合并的水层。然后,用盐水洗涤合并的有机层,用硫酸镁干燥,过滤并浓缩以获得作为浅棕色油的标题化合物(4.0 g, ~11.23 mmol)。LCMS (通过UV210-350nm测得的77%纯度; m/z):356.1 [M + H]+。不经任何进一步纯化即使用该产物。To a mixture of tert-butyl (E)-(4-((2-bromo-6-nitrophenyl)amino)but-2-en-1-yl)carbamate (4.4 g, 11.39 mmol) and ammonium chloride (6.09 g, 114 mmol) in methanol (50 mL) was added zinc (7.45 g, 114 mmol). Then, the reaction mixture was stirred at room temperature for 30 min. The reaction mixture was filtered and concentrated. The separated residue was distributed between EtOAc and water. The aqueous layer was separated and the organic layer was washed with water for a second time. The combined aqueous layer was extracted with EtOAc (1x). Then, the combined organic layer was washed with brine, dried over magnesium sulfate, filtered and concentrated to obtain the title compound (4.0 g, ~11.23 mmol) as a light brown oil. LCMS (77% purity measured by UV210-350nm; m/z ): 356.1 [M + H] + . The product was used without any further purification.
步骤3: (E)-(4-(7-溴-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)氨基甲酸叔丁酯Step 3: tert-Butyl (E)-(4-(7-bromo-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)carbamate
在室温向(E)-(4-((2-氨基-6-溴苯基)氨基)丁-2-烯-1-基)氨基甲酸叔丁酯(4.0 g, 11.23 mmol)在DMF (40 mL)中的溶液加入1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯溶液(1 M 在1,4-二氧杂环己烷中, 12.4 mL, 12.40 mmol)。然后搅拌该反应混合物45 min。然后,在室温向该反应混合物加入EDC (3.23 g, 16.84 mmol)和TEA (4.7 mL,33.7 mmol)。在搅拌2 h后,用EtOAc稀释该混合物并用水(2x)洗涤。用EtOAc (1x)再次萃取合并的水层。用盐水洗涤合并的有机层,用硫酸镁干燥,过滤,并浓缩。通过正相层析法(ISCO CombiFlash, 120g Gold柱, DCM/MeOH)纯化残余物以在溶剂蒸发后获得作为灰白色固体的标题化合物(4.5 g, 8.70 mmol, 77%收率)。LCMS (m/z): 517.2 [M + H]+。To a solution of tert-butyl (E)-(4-((2-amino-6-bromophenyl)amino)but-2-en-1-yl)carbamate (4.0 g, 11.23 mmol) in DMF (40 mL) was added 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate solution (1 M in 1,4-dioxane, 12.4 mL, 12.40 mmol) at room temperature. The reaction mixture was then stirred for 45 min. EDC (3.23 g, 16.84 mmol) and TEA (4.7 mL, 33.7 mmol) were then added to the reaction mixture at room temperature. After stirring for 2 h, the mixture was diluted with EtOAc and washed with water (2x). The combined aqueous layer was extracted again with EtOAc (1x). The combined organic layer was washed with brine, dried over magnesium sulfate, filtered, and concentrated. The residue was purified by normal phase chromatography (ISCO CombiFlash, 120 g Gold column, DCM/MeOH) to afford the title compound as an off-white solid after solvent evaporation (4.5 g, 8.70 mmol, 77% yield). LCMS ( m/z ): 517.2 [M + H] + .
步骤4: (E)-N-(1-(4-氨基丁-2-烯-1-基)-7-溴-1H-苯并[d]咪唑-2-基)-1-乙基-3-甲基-1H-吡唑-5-甲酰胺, 盐酸盐Step 4: (E)-N-(1-(4-aminobut-2-en-1-yl)-7-bromo-1H-benzo[d]imidazol-2-yl)-1-ethyl-3-methyl-1H-pyrazole-5-carboxamide, hydrochloride
在室温向(E)-(4-(7-溴-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)氨基甲酸叔丁酯 (1.1 g, 2.126 mmol)在甲醇 (10 mL)中的溶液加入HCl (4 M 在二氧杂环己烷中, 5.00 mL, 20 mmol)。然后,在室温搅拌该反应混合物过夜。在真空中浓缩该反应混合物并悬浮在二乙醚中。过滤所得固体,用二乙醚洗涤,然后干燥以获得作为白色固体的(E)-N-(1-(4-氨基丁-2-烯-1-基)-7-溴-1H-苯并[d]咪唑-2-基)-1-乙基-3-甲基-1H-吡唑-5-甲酰胺, 盐酸盐 (1.06 g)。不经任何进一步纯化即使用经分离的物质。 LCMS (m/z): 417.1 [M + H]+。To a solution of tert-butyl (E)-(4-(7-bromo-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)carbamate (1.1 g, 2.126 mmol) in methanol (10 mL) was added HCl (4 M in dioxane, 5.00 mL, 20 mmol) at room temperature. The reaction mixture was then stirred at room temperature overnight. The reaction mixture was concentrated in vacuo and suspended in diethyl ether. The resulting solid was filtered, washed with diethyl ether, and then dried to afford (E)-N-(1-(4-aminobut-2-en-1-yl)-7-bromo-1H-benzo[d]imidazol-2-yl)-1-ethyl-3-methyl-1H-pyrazole-5-carboxamide, hydrochloride (1.06 g) as a white solid. The isolated material was used without any further purification. LCMS ( m/z ): 417.1 [M+H] + .
步骤5: (E)-N-(7-溴-1-(4-((4-氨基甲酰基-2-硝基苯基)氨基)丁-2-烯-1-基)-1H-苯并[d]咪唑-2-基)-1-乙基-3-甲基-1H-吡唑-5-甲酰胺Step 5: (E)-N-(7-Bromo-1-(4-((4-carbamoyl-2-nitrophenyl)amino)but-2-en-1-yl)-1H-benzo[d]imidazol-2-yl)-1-ethyl-3-methyl-1H-pyrazole-5-carboxamide
在室温向(E)-N-(1-(4-氨基丁-2-烯-1-基)-7-溴-1H-苯并[d]咪唑-2-基)-1-乙基-3-甲基-1H-吡唑-5-甲酰胺, 盐酸盐 (0.965 g, 2.126 mmol)和4-氟-3-硝基苯甲酰胺(0.431 g, 2.339 mmol)在异丙醇 (10 mL)中的悬浮液加入DIEA (0.780 mL, 4.46mmol)。在70℃搅拌该反应混合物过夜。然后,将该反应混合物冷却至室温并在过滤器上收集固体。用异丙醇洗涤固体并干燥以获得作为黄色固体的(E)-N-(7-溴-1-(4-((4-氨基甲酰基-2-硝基苯基)氨基)丁-2-烯-1-基)-1H-苯并[d]咪唑-2-基)-1-乙基-3-甲基-1H-吡唑-5-甲酰胺 (1.2 g, 2.06 mmol, 97%收率)。不经任何进一步纯化即使用经分离的物质。LCMS (m/z): 581.1 [M + H]+。To a suspension of (E)-N-(1-(4-aminobut-2-en-1-yl)-7-bromo-1H-benzo[d]imidazol-2-yl)-1-ethyl-3-methyl-1H-pyrazole-5-carboxamide, hydrochloride (0.965 g, 2.126 mmol) and 4-fluoro-3-nitrobenzamide (0.431 g, 2.339 mmol) in isopropanol (10 mL) was added DIEA (0.780 mL, 4.46 mmol) at room temperature. The reaction mixture was stirred at 70°C overnight. Then, the reaction mixture was cooled to room temperature and the solid was collected on a filter. The solid was washed with isopropanol and dried to afford (E)-N-(7-bromo-1-(4-((4-carbamoyl-2-nitrophenyl)amino)but-2-en-1-yl)-1H-benzo[d]imidazol-2-yl)-1-ethyl-3-methyl-1H-pyrazole-5-carboxamide (1.2 g, 2.06 mmol, 97% yield) as a yellow solid. The isolated material was used without any further purification. LCMS ( m/z ): 581.1 [M+H] + .
步骤6: (E)-N-(1-(4-((2-氨基-4-氨基甲酰基苯基)氨基)丁-2-烯-1-基)-7-溴-1H-苯并[d]咪唑-2-基)-1-乙基-3-甲基-1H-吡唑-5-甲酰胺Step 6: (E)-N-(1-(4-((2-amino-4-carbamoylphenyl)amino)but-2-en-1-yl)-7-bromo-1H-benzo[d]imidazol-2-yl)-1-ethyl-3-methyl-1H-pyrazole-5-carboxamide
向(E)-N-(7-溴-1-(4-((4-氨基甲酰基-2-硝基苯基)氨基)丁-2-烯-1-基)-1H-苯并[d]咪唑-2-基)-1-乙基-3-甲基-1H-吡唑-5-甲酰胺 (1.2 g, 2.064 mmol)和氯化铵(1.1 g, 20.56 mmol)在甲醇 (15 mL)中的溶液加入锌(1.3 g, 19.88 mmol)。然后,在室温搅拌该反应混合物8 h。加入另外的10 eq 氯化铵 (1.1 g, 20.56 mmol)和锌(1.3 g,19.88 mmol)并在室温搅拌该混合物过夜。由于还原仍然未完全,加入乙酸(1.5 mL),然后在室温搅拌30 min。To a solution of (E)-N-(7-bromo-1-(4-((4-carbamoyl-2-nitrophenyl)amino)but-2-en-1-yl)-1H-benzo[d]imidazol-2-yl)-1-ethyl-3-methyl-1H-pyrazole-5-carboxamide (1.2 g, 2.064 mmol) and ammonium chloride (1.1 g, 20.56 mmol) in methanol (15 mL) was added zinc (1.3 g, 19.88 mmol). The reaction mixture was then stirred at room temperature for 8 h. Another 10 eq of ammonium chloride (1.1 g, 20.56 mmol) and zinc (1.3 g, 19.88 mmol) were added and the mixture was stirred at room temperature overnight. As the reduction was still not complete, acetic acid (1.5 mL) was added and then stirred at room temperature for 30 min.
过滤该反应混合物并浓缩滤液。在EtOAc和水之间分配分离的物质。在过滤器上收集出现的固体,用EtOAc和水洗涤。然后,将滤液置于分液漏斗中。用EtOAc (1x)萃取水层。然后,用盐水洗涤经合并的有机层,干燥(MgSO4)并浓缩。将以上分离的残余物和固体溶解于MeOH中并蒸发至硅藻土上。 硅胶层析法 (干装载, 40 g柱,0-10%梯度的MeOH/DCM)提供灰白色固体 (911 mg, ~1.6 mmol),为~3:1的(E)-N-(1-(4-((2-氨基-4-氨基甲酰基苯基)氨基)丁-2-烯-1-基)-7-溴-1H-苯并[d]咪唑-2-基)-1-乙基-3-甲基-1H-吡唑-5-甲酰胺(LCMS (m/z): 551.2 [M + H]+)和脱溴化副产物的混合物。将经分离的物质直接用于接下来的反应。The reaction mixture is filtered and the filtrate is concentrated. The separated material is distributed between EtOAc and water. The solid that appears is collected on the filter and washed with EtOAc and water. Then, the filtrate is placed in a separatory funnel. The aqueous layer is extracted with EtOAc (1x). Then, the organic layer combined is washed with salt water, dried (MgSO ) and concentrated. The above separated residue and solid are dissolved in MeOH and evaporated onto diatomaceous earth. Silica gel chromatography (dry loading, 40 g column, 0-10% gradient of MeOH/DCM) afforded an off-white solid (911 mg, ~1.6 mmol) as a ~3:1 mixture of (E)-N-(1-(4-((2-amino-4-carbamoylphenyl)amino)but-2-en-1-yl)-7-bromo-1H-benzo[d]imidazol-2-yl)-1-ethyl-3-methyl-1H-pyrazole-5-carboxamide (LCMS ( m/z ): 551.2 [M+H] + ) and debrominated byproducts. The isolated material was used directly in the next reaction.
步骤7: (E)-1-(4-(7-溴-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 7: (E)-1-(4-(7-bromo-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide
在室温向(E)-N-(1-(4-((2-氨基-4-氨基甲酰基苯基)氨基)丁-2-烯-1-基)-7-溴-1H-苯并[d]咪唑-2-基)-1-乙基-3-甲基-1H-吡唑-5-甲酰胺 (911 mg, ~1.6 mmol, 其中~25%缺溴原子)在DMF (10 mL)中的溶液加入1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (1.82 mL, 1.82 mmol)在1,4 二氧杂环己烷中的1 M溶液。然后,在室温搅拌该反应混合物1.5 h。然后,在室温加入EDC (633 mg, 3.30 mmol)和TEA (0.921 mL, 6.61 mmol)。在室温搅拌过夜之后,用EtOAc和水稀释该反应混合物。过滤悬浮固体并用EtOAc、水、EtOAc、然后二乙醚洗涤。干燥灰白色固体提供85:15的(E)-1-(4-(7-溴-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (883 mg, ~1.2 mmol, 75%收率, LCMS(m/z): 712.2 [M + H]+)和相应的脱溴类似物(des-bromide analog)的混合物。不经进一步纯化而使用该物质。To a solution of (E)-N-(1-(4-((2-amino-4-carbamoylphenyl)amino)but-2-en-1-yl)-7-bromo-1H-benzo[d]imidazol-2-yl)-1-ethyl-3-methyl-1H-pyrazole-5-carboxamide (911 mg, ~1.6 mmol, with ~25% deficient bromine atoms) in DMF (10 mL) was added a 1 M solution of 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (1.82 mL, 1.82 mmol) in 1,4 dioxane at room temperature. The reaction mixture was then stirred at room temperature for 1.5 h. EDC (633 mg, 3.30 mmol) and TEA (0.921 mL, 6.61 mmol) were then added at room temperature. After stirring at room temperature overnight, the reaction mixture was diluted with EtOAc and water. The suspended solid was filtered and washed with EtOAc, water, EtOAc, and then diethyl ether. The off-white solid was dried to provide a 85:15 mixture of (E)-1-(4-(7-bromo-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (883 mg, ~1.2 mmol, 75% yield, LCMS ( m/z ): 712.2 [M+H] + ) and the corresponding des-bromide analog. The material was used without further purification.
步骤8: (E)-1-((E)-4-((E)-7-溴-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 8: (E)-1-((E)-4-((E)-7-bromo-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
在室温向(E)-1-(4-(7-溴-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (816 mg, 1.145 mmol, ~15%脱溴杂质)和碳酸铯 (1.5 g, 4.60 mmol)在DMF (10 mL)中的混合物加入碘甲烷(0.143 mL, 2.29 mmol)。然后在室温搅拌该反应物4h。加入另外的碘甲烷(0.143 mL, 2.290 mmol)并在室温搅拌该反应混合物45 h。加入水和EtOAc以产生固体 (345 mg, 未反应的起始原料)。分离滤液并用EtOAc萃取水相。用盐水洗涤合并的有机层,经硫酸镁干燥,并浓缩。通过制备型HPLC的纯化 (30 mm x 50 mm GeminiC18,梯度的ACN/水,含0.1% TFA 改性剂)提供作为白色固体的(E)-1-((E)-4-((E)-7-溴-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (107 mg, 0.144 mmol, 12.6%收率)。1H NMR (400MHz, DMSO-d 6) δppm 8.09 (d, J=1.3 Hz, 1 H), 8.03 (br. s., 1 H), 7.81 (dd, J=1.5, 8.3 Hz, 1H), 7.60 (dd, J=0.8, 8.0 Hz, 1 H), 7.50 (d, J=8.3 Hz, 1 H), 7.45 (dd, J=0.8,8.0 Hz, 2 H), 7.23 (t, J=8.2 Hz, 1 H), 6.45 (s, 1 H), 6.39 (s, 1 H), 5.95(td, J=4.8, 15.7 Hz, 1 H), 5.56 (td, J=5.8, 15.7 Hz, 1 H), 5.01 (d, J=3.8 Hz,2 H), 4.78 (d, J=5.5 Hz, 2 H), 4.49 - 4.38 (m, 4 H), 3.97 (s, 2 H), 3.51 (s,3 H), 2.13 (s, 3 H), 2.11 (s, 3 H), 1.15-1.28 (m, 6 H)。LCMS (m/z): 740.3 [M +H]+。To a mixture of (E)-1-(4-(7-bromo-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (816 mg, 1.145 mmol, ~15% debrominated impurity) and cesium carbonate (1.5 g, 4.60 mmol) in DMF (10 mL) was added iodomethane (0.143 mL, 2.29 mmol) at room temperature. The reaction was then stirred at room temperature for 4 h. Additional iodomethane (0.143 mL, 2.290 mmol) was added and the reaction mixture was stirred at room temperature for 45 h. Water and EtOAc were added to produce a solid (345 mg, unreacted starting material). The filtrate is separated and aqueous phase is extracted with EtOAc.The organic layer merged is washed with salt water, dried over magnesium sulfate, and concentrated.The (E)-1-((E)-4-((E)-7-bromo-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl) imino)-3-methyl-2,3-dihydro-1H-benzo [d] imidazoles-1-yl) but-2-ene-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazoles-5-carbonyl) imino)-3-methyl-2,3-dihydro-1H-benzo [d] imidazoles-5-carboxamide (107 mg, 0.144 mmol, 12.6% yield) as white solid is provided by the purification (30 mm x 50 mm GeminiC18, gradient ACN/ water, containing 0.1% TFA modifier) of preparative HPLC. 1 H NMR (400MHz, DMSO- d 6 ) δppm 8.09 (d, J =1.3 Hz, 1 H), 8.03 (br. s., 1 H), 7.81 (dd, J =1.5, 8.3 Hz, 1H), 7.60 (dd, J =0.8, 8.0 Hz, 1 H), 7.50 (d, J = 8.3 Hz, 1 H), 7.45 (dd, J =0.8,8.0 Hz, 2 H), 7.23 (t, J =8.2 Hz, 1 H), 6.45 (s, 1 H), 6.39 (s, 1 H), 5.95 (td, J =4.8, 15.7 Hz, 1 H), 5.56 (td, J =5. 8, 15.7 Hz, 1 H), 5.01 (d, J =3.8 Hz,2 H), 4.78 (d, J =5.5 Hz, 2 H), 4.49 - 4.38 (m, 4 H), 3.97 (s, 2 H), 3.51 (s, 3 H), 2.13 (s, 3 H), 2.11 (s, 3 H), 1.1 5-1.28 (m, 6 H). LCMS ( m/z ): 740.3 [M +H] + .
步骤9: (E)-1-(4-((E)-7-溴-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 9: (E)-1-(4-((E)-7-bromo-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向(E)-1-((E)-4-((E)-7-溴-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (54 mg, 0.073 mmol)在叔丁醇 (0.8 mL)和水(0.2 mL)中的悬浮液加入NMO (26 mg, 0.22 mmol)。在室温搅拌5 min后,加入叔丁醇中的2.5%四氧化锇 (0.183 mL, 0.015 mmol)并在室温继续搅拌2 h。过滤该反应混合物并通过反相HPLC (30 mm x 50 mm Gemini C18,梯度的ACN/水,含0.1% 氢氧化铵改性剂)直接纯化滤液。获得作为白色固体的(E)-1-(4-((E)-7-溴-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (37 mg, 0.48 mmol, 65%收率)。1H NMR (400MHz, DMSO-d 6) δ ppm 8.09 -8.00 (m, 2 H), 7.85 (dd, J=1.5, 8.4 Hz, 1 H), 7.61 - 7.56 (m, 2 H), 7.48 -7.39 (m, 2 H), 7.23 (t, J=8.0 Hz, 1 H), 6.47 (d, J=7.1 Hz, 2 H), 5.36 (d, J=6.6 Hz, 1 H), 5.11 (d, J=6.6 Hz, 1 H), 4.71 (dd, J=9.1, 14.4 Hz, 1 H), 4.50(五重峰, J=6.7 Hz, 4 H), 4.38 (dd, J=3.8, 14.4 Hz, 1 H), 4.30 - 4.15 (m, 2H), 4.04 - 3.89 (m, 2 H), 3.57 (s, 3 H), 3.51 (s, 3 H), 2.11 (s, 3 H), 2.15(s, 3 H), 1.34 - 1.21 (m, 6 H)。LCMS (m/z): 774.3 [M + H]+)。To a suspension of (E)-1-((E)-4-((E)-7-bromo-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (54 mg, 0.073 mmol) in tert-butyl alcohol (0.8 mL) and water (0.2 mL) was added NMO (26 mg, 0.22 mmol). After stirring at room temperature for 5 min, 2.5% osmium tetroxide in tert-butyl alcohol (0.183 mL, 0.015 mmol) was added and stirring was continued at room temperature for 2 h. The reaction mixture was filtered and the filtrate was directly purified by reverse phase HPLC (30 mm x 50 mm Gemini C18, gradient of ACN/water with 0.1% ammonium hydroxide modifier). (E)-1-(4-((E)-7-bromo-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (37 mg, 0.48 mmol, 65% yield) was obtained as a white solid. 1 H NMR (400MHz, DMSO- d 6 ) δ ppm 8.09 -8.00 (m, 2 H), 7.85 (dd, J =1.5, 8.4 Hz, 1 H), 7.61 - 7.56 (m, 2 H), 7.48 -7.39 (m, 2 H), 7.23 (t, J =8.0 Hz, 1 H ), 6.47 (d, J =7.1 Hz, 2 H), 5.36 (d, J =6.6 Hz, 1 H), 5.11 (d, J =6.6 Hz, 1 H), 4.71 (dd, J =9.1, 14.4 Hz, 1 H), 4.50 (quint, J =6.7 Hz, 4 H), 4.38 (dd, J =3.8, 14.4 Hz, 1 H), 4.30 - 4.15 (m, 2H), 4.04 - 3.89 (m, 2 H), 3.57 (s, 3 H), 3.51 (s, 3 H), 2.11 (s, 3 H), 2.15 (s, 3 H), 1.34 - 1.21 (m, 6 H). LCMS ( m/z ): 774.3 [M + H] + ).
实施例35Embodiment 35
(2E,2'E)-1,1'-((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺)(2E,2'E)-1,1'-((2S,3S)-2,3-diethoxybutane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide)
步骤1: 4,4'-(((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(氮烷二基))双(3-硝基苯甲酰胺)Step 1: 4,4'-(((2S,3S)-2,3-diethoxybutane-1,4-diyl)bis(azanediyl))bis(3-nitrobenzamide)
向4-氟-3-硝基苯甲酰胺 (0.985 g, 5.35 mmol)在1-丁醇 (10 mL)中的混合物加入(2S,3S)-2,3-二乙氧基丁烷-1,4-二胺 (0.46 g, 2.61 mmol)和DIEA (1.82 mL,10.4 mmol)。在110℃搅拌该混合物2 h。当冷却至室温时,在过滤器上收集固体,用二乙醚和2-丙醇(1:1)的混合物洗涤并干燥以提供作为橙色固体的4,4'-(((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(氮烷二基))双(3-硝基苯甲酰胺) (0.63 g, 1.25 mmol, 48%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 1.13 (t, J=7.0 Hz, 6 H), 3.45 - 3.74 (m, 8H), 3.79 - 3.89 (m, 2 H), 7.17 (d, J=9.1 Hz, 2 H), 7.31 (br. s., 2 H), 8.02(dd, J=8.9, 2.0 Hz, 4 H), 8.56 (t, J=5.2 Hz, 2 H), 8.66 (d, J=2.0 Hz, 2 H)。LCMS (m/z): 505.1 [M + H]+。To a mixture of 4-fluoro-3-nitrobenzamide (0.985 g, 5.35 mmol) in 1-butanol (10 mL) was added (2S,3S)-2,3-diethoxybutane-1,4-diamine (0.46 g, 2.61 mmol) and DIEA (1.82 mL, 10.4 mmol). The mixture was stirred at 110 °C for 2 h. When cooled to room temperature, the solid was collected on a filter, washed with a mixture of diethyl ether and 2-propanol (1:1) and dried to provide 4,4'-(((2S,3S)-2,3-diethoxybutane-1,4-diyl)bis(azanediyl))bis(3-nitrobenzamide) (0.63 g, 1.25 mmol, 48% yield) as an orange solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 1.13 (t, J =7.0 Hz, 6 H), 3.45 - 3.74 (m, 8H), 3.79 - 3.89 (m, 2 H), 7.17 (d, J =9.1 Hz, 2 H), 7.31 (br. s., 2 H), 8 .02(dd, J =8.9, 2.0 Hz, 4 H), 8.56 (t, J =5.2 Hz, 2 H), 8.66 (d, J =2.0 Hz, 2 H). LCMS ( m/z ): 505.1 [M + H] + .
步骤2: 4,4'-(((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(氮烷二基))双(3-氨基苯甲酰胺)Step 2: 4,4'-(((2S,3S)-2,3-diethoxybutane-1,4-diyl)bis(azanediyl))bis(3-aminobenzamide)
向100 mL 圆底烧瓶加入4,4'-(((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(氮烷二基))双(3-硝基苯甲酰胺) (0.63 g, 1.25 mmol)和甲醇 (20 mL)。向这个混合物加入10mL饱和氯化铵水溶液。向这个混合物加入锌(0.812 g, 12.5 mmol)并在室温搅拌非均相混合物15 min。使用MeOH将该混合物通过过滤器并漂洗,然后浓缩。硅胶层析法 (24 g柱,6-20%梯度的 MeOH/DCM,含1% 氢氧化铵作为改性剂)提供作为浅黄色固体的4,4'-(((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(氮烷二基))双(3-氨基苯甲酰胺) (0.446 g, 1.00mmol, 80%收率)。LCMS (m/z): 445.4 [M + H]+。To a 100 mL round bottom flask was added 4,4'-(((2S,3S)-2,3-diethoxybutane-1,4-diyl)bis(azanediyl))bis(3-nitrobenzamide) (0.63 g, 1.25 mmol) and methanol (20 mL). To this mixture was added 10 mL of saturated aqueous ammonium chloride solution. To this mixture was added zinc (0.812 g, 12.5 mmol) and the heterogeneous mixture was stirred at room temperature for 15 min. The mixture was passed through a filter and rinsed with MeOH and then concentrated. Silica gel chromatography (24 g column, 6-20% gradient of MeOH/DCM with 1% ammonium hydroxide as modifier) provided 4,4'-(((2S,3S)-2,3-diethoxybutane-1,4-diyl)bis(azanediyl))bis(3-aminobenzamide) (0.446 g, 1.00 mmol, 80% yield) as a light yellow solid. LCMS ( m/z ): 445.4 [M+H] + .
步骤3: 1,1'-((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺)Step 3: 1,1'-((2S,3S)-2,3-diethoxybutane-1,4-diyl)bis(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide)
向4,4'-(((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(氮烷二基))双(3-氨基苯甲酰胺) (0.446 g, 1.00 mmol)在DMF (20 mL)中的溶液加入1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (~0.4 M 在二氧杂环己烷中,5.02 mL, 2.00 mmol)。搅拌该混合物15 min。加入EDC (0.481 g, 2.51 mmol)和TEA (0.699 mL, 5.02 mmol)并在室温搅拌该反应混合物18 h。将该混合物倾倒至3:1水:饱和氯化铵水溶液(100 mL)中。立即形成细固体并再继续搅拌10 min。过滤所得固体,用水洗涤,并干燥以提供作为白色固体的1,1'-((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺) (0.539 mg, 0.701 mmol, 70%收率)。LCMS (m/z): 767.5 [M + H]+。To a solution of 4,4'-(((2S,3S)-2,3-diethoxybutane-1,4-diyl)bis(azanediyl))bis(3-aminobenzamide) (0.446 g, 1.00 mmol) in DMF (20 mL) was added 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (~0.4 M in dioxane, 5.02 mL, 2.00 mmol). The mixture was stirred for 15 min. EDC (0.481 g, 2.51 mmol) and TEA (0.699 mL, 5.02 mmol) were added and the reaction mixture was stirred at room temperature for 18 h. The mixture was poured into 3:1 water:saturated aqueous ammonium chloride solution (100 mL). A fine solid was formed immediately and stirring was continued for another 10 min. The resulting solid was filtered, washed with water, and dried to provide 1,1′-((2S,3S)-2,3-diethoxybutane-1,4-diyl)bis(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide) (0.539 mg, 0.701 mmol, 70% yield) as a white solid. LCMS ( m/z ): 767.5 [M+H] + .
步骤4: (2E,2'E)-1,1'-((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺)Step 4: (2E,2'E)-1,1'-((2S,3S)-2,3-diethoxybutane-1,4-diyl)bis(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide)
向1,1'-((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺) (0.09 g, 0.117 mmol)在DMF (5 mL)中的溶液加入碳酸铯 (0.103 g, 0.317 mmol)和碘甲烷 (0.018 mL, 0.282 mmol)。在室温搅拌该反应混合物18 h。加入更多的碳酸铯 (0.019 g, 0.059 mmol)和碘甲烷 (0.015 mL,0.235 mmol)。在50 ℃搅拌该混合物1 h。用水稀释该反应并用EtOAc (3 x 50 mL)萃取。将有机相用盐水(10 mL)洗涤,用硫酸镁干燥并浓缩。使用质量导向的HPLC (XSelect CSHPrep C18, 5 um,15-55%梯度的 ACN/水,含0.1% TFA作为改性剂)纯化产物。合并各级分并除去ACN。用饱和碳酸氢铵溶液碱化水相。过滤所得固体并在冻干机上干燥以提供作为白色固体的(2E,2'E)-1,1'-((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺) (13 mg, 0.016mmol, 14%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.01 - 8.15 (m, 4 H) 7.80 -7.93 (m, 2 H) 7.61 (d, J=8.36 Hz, 2 H) 7.46 (br. s., 2 H) 6.51 (s, 2 H) 4.46- 4.62 (m, 6 H) 4.38 (dd, J=14.45, 8.87 Hz, 2 H) 3.78 - 3.93 (m, 2 H) 3.60(s, 6 H) 3.25 - 3.33 (m, 2 H) 3.02 (dd, J=9.38, 7.10 Hz, 2 H) 2.12 (s, 6 H)1.31 (t, J=7.10 Hz, 6 H) 0.59 (t, J=6.97 Hz, 6 H)。LCMS (m/z): 795.3 [M + H]+。To a solution of 1,1'-((2S,3S)-2,3-diethoxybutane-1,4-diyl)bis(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide) (0.09 g, 0.117 mmol) in DMF (5 mL) was added cesium carbonate (0.103 g, 0.317 mmol) and iodomethane (0.018 mL, 0.282 mmol). The reaction mixture was stirred at room temperature for 18 h. More cesium carbonate (0.019 g, 0.059 mmol) and iodomethane (0.015 mL, 0.235 mmol) were added. The mixture was stirred at 50 °C for 1 h. The reaction was diluted with water and extracted with EtOAc (3 x 50 mL). The organic phase was washed with brine (10 mL), dried over magnesium sulfate and concentrated. Use mass-guided HPLC (XSelect CSHPrep C18, 5 um, 15-55% gradient ACN/ water, containing 0.1% TFA as modifier) purified product. Combine each fraction and remove ACN. Use saturated ammonium bicarbonate solution to alkalize aqueous phase. Filter gained solid and dry on freeze dryer to provide (2E, 2'E)-1,1'-((2S, 3S)-2,3-diethoxybutane-1,4-diyl)bis-(2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl) imino)-3-methyl-2,3-dihydro-1H-benzo [d] imidazole-5-carboxamide) (13 mg, 0.016mmol, 14% yield) as white solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.01 - 8.15 (m, 4 H) 7.80 -7.93 (m, 2 H) 7.61 (d, J =8.36 Hz, 2 H) 7.46 (br. s., 2 H) 6.51 (s, 2 H) 4.46- 4.62 (m, 6 H) 4.38 (dd, J =14.45, 8.87 Hz, 2 H) 3.78 - 3.93 (m, 2 H) 3.60 (s, 6 H) 3.25 - 3.33 (m, 2 H) 3.02 (dd, J =9.38, 7.10 Hz, 2 H) 2.12 (s, 6 H)1.31 (t, J =7.10 Hz, 6 H) 0.59 (t, J =6.97 Hz, 6 H). LCMS ( m/z ): 795.3 [M + H] + .
实施例36Embodiment 36
(E)-1-(((4R,5R)-5-(((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-(((4R,5R)-5-(((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
步骤1: 4-((((4R,5R)-5-(氨基甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)氨基)-3-(3-(苄氧基)丙氧基)-5-硝基苯甲酰胺Step 1: 4-((((4R,5R)-5-(aminomethyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)amino)-3-(3-(benzyloxy)propoxy)-5-nitrobenzamide
向((4R,5R)-2,2-二甲基-1,3-二氧杂环戊烷-4,5-二基)二甲胺 (1 g, 6.24mmol)和3-(3-(苄氧基)丙氧基)-4-氯-5-硝基苯甲酰胺 (2.049 g, 5.62 mmol)在1-丁醇(20 mL)中的溶液加入DIEA (3.27 mL, 18.72 mmol)。在120℃搅拌该反应混合物16 h。在真空下浓缩该混合物以提供粗产物。通过硅胶层析法 (洗脱梯度:在DCM中的0至30% MeOH)纯化粗产物。将纯级分蒸发至干以获得作为红色胶状物的4-((((4R,5R)-5-(氨基甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)氨基)-3-(3-(苄氧基)丙氧基)-5-硝基苯甲酰胺(1.5 g, 2.76 mmol, 44.3%收率)。LCMS (m/z): 489 [M + H]+。To a solution of ((4R,5R)-2,2-dimethyl-1,3-dioxolane-4,5-diyl)dimethylamine (1 g, 6.24 mmol) and 3-(3-(benzyloxy)propoxy)-4-chloro-5-nitrobenzamide (2.049 g, 5.62 mmol) in 1-butanol (20 mL) was added DIEA (3.27 mL, 18.72 mmol). The reaction mixture was stirred at 120 °C for 16 h. The mixture was concentrated under vacuum to provide the crude product. The crude product was purified by silica gel chromatography (elution gradient: 0 to 30% MeOH in DCM). Pure fractions were evaporated to dryness to afford 4-((((4R,5R)-5-(aminomethyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)amino)-3-(3-(benzyloxy)propoxy)-5-nitrobenzamide (1.5 g, 2.76 mmol, 44.3% yield) as a red gum. LCMS ( m/z ): 489 [M+H] + .
步骤2: 3-(3-(苄氧基)丙氧基)-4-((((4R,5R)-5-(((4-氨基甲酰基-2-硝基苯基)氨基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)氨基)-5-硝基苯甲酰胺Step 2: 3-(3-(Benzyloxy)propoxy)-4-((((4R,5R)-5-(((4-carbamoyl-2-nitrophenyl)amino)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)amino)-5-nitrobenzamide
向4-((((4R,5R)-5-(氨基甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)氨基)-3-(3-(苄氧基)丙氧基)-5-硝基苯甲酰胺 (1.4 g, 2.87 mmol)和4-氟-3-硝基苯甲酰胺 (0.580 g, 3.15 mmol)在DMSO (15 mL)中的溶液加入K2CO3 (0.792 g, 5.73 mmol)。在25℃搅拌该反应混合物16 h。将该混合物倾倒至水 (100 mL)中。通过过滤收集沉淀物,用水洗涤并在真空下干燥以获得作为橙色固体的3-(3-(苄氧基)丙氧基)-4-((((4R,5R)-5-(((4-氨基甲酰基-2-硝基苯基)氨基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)氨基)-5-硝基苯甲酰胺 (1.4 g, 1.931 mmol, 67.4%收率)。LCMS (m/z): 653 [M +H]+。To a solution of 4-((((4R,5R)-5-(aminomethyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)amino)-3-(3-(benzyloxy)propoxy)-5-nitrobenzamide (1.4 g, 2.87 mmol) and 4-fluoro-3-nitrobenzamide (0.580 g, 3.15 mmol) in DMSO (15 mL) was added K2CO3 (0.792 g, 5.73 mmol). The reaction mixture was stirred at 25 °C for 16 h. The mixture was poured into water (100 mL). The precipitate was collected by filtration, washed with water and dried under vacuum to afford 3-(3-(benzyloxy)propoxy)-4-((((4R,5R)-5-(((4-carbamoyl-2-nitrophenyl)amino)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)amino)-5-nitrobenzamide (1.4 g, 1.931 mmol, 67.4% yield) as an orange solid. LCMS (m/z): 653 [M +H] + .
步骤3: 3-氨基-4-((((4R,5R)-5-(((2-氨基-4-氨基甲酰基苯基)氨基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)氨基)-5-(3-(苄氧基)丙氧基)苯甲酰胺Step 3: 3-amino-4-((((4R,5R)-5-(((2-amino-4-carbamoylphenyl)amino)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)amino)-5-(3-(benzyloxy)propoxy)benzamide
向3-(3-(苄氧基)丙氧基)-4-((((4R,5R)-5-(((4-氨基甲酰基-2-硝基苯基)氨基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)氨基)-5-硝基苯甲酰胺 (1.35 g,2.068 mmol)在乙酸(20 mL)中的溶液加入锌(1.352 g, 20.68 mmol)。在25℃搅拌该反应混合物3 h。采用DCM (50 mL)稀释该混合物并过滤。在减压下浓缩滤液以提供作为灰色固体的粗品3-氨基-4-((((4R,5R)-5-(((2-氨基-4-氨基甲酰基苯基)氨基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)氨基)-5-(3-(苄氧基)丙氧基)苯甲酰胺 (1.3 g,1.755 mmol, 85%收率)。LCMS (m/z): 593 [M + H]+。To a solution of 3-(3-(benzyloxy)propoxy)-4-((((4R,5R)-5-(((4-carbamoyl-2-nitrophenyl)amino)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)amino)-5-nitrobenzamide (1.35 g, 2.068 mmol) in acetic acid (20 mL) was added zinc (1.352 g, 20.68 mmol). The reaction mixture was stirred at 25 °C for 3 h. The mixture was diluted with DCM (50 mL) and filtered. The filtrate was concentrated under reduced pressure to afford crude 3-amino-4-((((4R,5R)-5-(((2-amino-4-carbamoylphenyl)amino)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)amino)-5-(3-(benzyloxy)propoxy)benzamide (1.3 g, 1.755 mmol, 85% yield) as a grey solid. LCMS (m/z): 593 [M + H] + .
步骤4: 2-氨基-1-(((4R,5R)-5-((2-氨基-5-氨基甲酰基-1H-苯并[d]咪唑-1-基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)-7-(3-(苄氧基)丙氧基)-1H-苯并[d]咪唑-5-甲酰胺Step 4: 2-amino-1-(((4R,5R)-5-((2-amino-5-carbamoyl-1H-benzo[d]imidazol-1-yl)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-7-(3-(benzyloxy)propoxy)-1H-benzo[d]imidazole-5-carboxamide
向3-氨基-4-((((4R,5R)-5-(((2-氨基-4-氨基甲酰基苯基)氨基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)氨基)-5-(3-(苄氧基)丙氧基)苯甲酰胺 (1.35 g,2.278 mmol)在甲醇 (20 mL)中的溶液加入溴化氰 (0.724 g, 6.83 mmol)。在25℃搅拌该反应混合物16 h。用二乙醚 (30 mL)稀释该混合物。将该混合物过滤并用二乙醚洗涤。在减压下浓缩滤液以提供作为灰色固体的2-氨基-1-(((4R,5R)-5-((2-氨基-5-氨基甲酰基-1H-苯并[d]咪唑-1-基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)-7-(3-(苄氧基)丙氧基)-1H-苯并[d]咪唑-5-甲酰胺 (800 mg, 1.12 mmol, 49.2%收率)。LCMS (m/z):643 [M + H]+。To a solution of 3-amino-4-((((4R,5R)-5-(((2-amino-4-carbamoylphenyl)amino)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)amino)-5-(3-(benzyloxy)propoxy)benzamide (1.35 g, 2.278 mmol) in methanol (20 mL) was added cyanogen bromide (0.724 g, 6.83 mmol). The reaction mixture was stirred at 25 °C for 16 h. The mixture was diluted with diethyl ether (30 mL). The mixture was filtered and washed with diethyl ether. The filtrate was concentrated under reduced pressure to afford 2-amino-1-(((4R,5R)-5-((2-amino-5-carbamoyl-1H-benzo[d]imidazol-1-yl)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-7-(3-(benzyloxy)propoxy)-1H-benzo[d]imidazole-5-carboxamide (800 mg, 1.12 mmol, 49.2% yield) as a grey solid. LCMS (m/z): 643 [M + H] + .
步骤5: 7-(3-(苄氧基)丙氧基)-1-(((4R,5R)-5-((5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 5: 7-(3-(Benzyloxy)propoxy)-1-(((4R,5R)-5-((5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide
向1-乙基-3-甲基-1H-吡唑-5-甲酸 (336 mg, 2.178 mmol)、 2-氨基-1-(((4R,5R)-5-((2-氨基-5-氨基甲酰基-1H-苯并[d]咪唑-1-基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)-7-(3-(苄氧基)丙氧基)-1H-苯并[d]咪唑-5-甲酰胺 (700 mg, 1.089mmol)和DIPEA (0.951 mL, 5.45 mmol)在DMF (10 mL)中的混合物加入HATU (1035 mg,2.72 mmol)。在 60℃搅拌该反应混合物16 h。将该混合物倾倒至水中。通过过滤收集沉淀物,用水和二乙醚洗涤,并然后在真空下干燥以提供作为棕色固体的7-(3-(苄氧基)丙氧基)-1-(((4R,5R)-5-((5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (800 mg, 0.743 mmol, 68.2%收率)。LCMS (通过UV测得的~85%纯度, m/z): 915 [M + H]+。To a mixture of 1-ethyl-3-methyl-1H-pyrazole-5-carboxylic acid (336 mg, 2.178 mmol), 2-amino-1-(((4R,5R)-5-((2-amino-5-carbamoyl-1H-benzo[d]imidazol-1-yl)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-7-(3-(benzyloxy)propoxy)-1H-benzo[d]imidazole-5-carboxamide (700 mg, 1.089 mmol) and DIPEA (0.951 mL, 5.45 mmol) in DMF (10 mL) was added HATU (1035 mg, 2.72 mmol). The reaction mixture was stirred at 60° C. for 16 h. The mixture was poured into water. The precipitate was collected by filtration, washed with water and diethyl ether, and then dried under vacuum to afford 7-(3-(benzyloxy)propoxy)-1-(((4R,5R)-5-((5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (800 mg, 0.743 mmol, 68.2% yield) as a brown solid. LCMS (~85% purity by UV, m/z ): 915 [M + H] + .
步骤6: 1-(((4R,5R)-5-((5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-羟基丙氧基)-1H-苯并[d]咪唑-5-甲酰胺Step 6: 1-(((4R,5R)-5-((5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-hydroxypropoxy)-1H-benzo[d]imidazol-5-carboxamide
向7-(3-(苄氧基)丙氧基)-1-(((4R,5R)-5-((5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (650mg, 0.710 mmol)在甲醇 (20 mL)中的溶液加入Pd-C (756 mg, 7.10 mmol)。在60℃使用H-cube (4 atm)氢化该反应72 h。用DMF (20 mL)稀释该混合物。过滤该混合物并在减压下浓缩滤液以提供粗产物。通过制备型HPLC (Gemini-C18柱, 5µ 硅胶, 21 x 150 mm; 30-40%梯度的ACN/水,含0.1% TFA 改性剂)纯化粗产物以提供作为灰白色固体的1-(((4R,5R)-5-((5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-羟基丙氧基)-1H-苯并[d]咪唑-5-甲酰胺 (30 mg, 0.035 mmol, 4.9%收率)。1H NMR (400 MHz, MeOH-d4) δ 7.58 (s, 1 H), 7.48 (d, J = 8.4 Hz, 1 H),7.33 (s, 1 H), 7.17 (d, J = 8.4 Hz, 1 H), 6.89 (s, 1 H), 6.58 (s, 2 H), 5.11- 4.99 (m, 1 H), 4.93 (s, 1 H), 4.61 (dqd, J = 26.0, 13.2, 6.9 Hz, 5 H), 4.46- 4.36 (m, 1 H), 4.31 (dd, J = 13.4, 3.2 Hz, 1 H), 4.19 (dd, J = 14.0, 3.4Hz, 1 H), 4.03 (dd, J = 15.1, 6.4 Hz, 1 H), 3.88 (dd, J = 14.9, 6.4 Hz, 1 H),3.83 - 3.72 (m, 1 H), 2.23 (s, 3 H), 2.18 (s, 3 H), 2.05 (dd, J = 11.5, 5.9Hz, 2 H), 1.64 (d, J = 10.3 Hz, 6 H), 1.50 - 1.28 (m, 6 H)。LCMS (m/z): 825 [M+ H]+。To a solution of 7-(3-(benzyloxy)propoxy)-1-(((4R,5R)-5-((5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (650 mg, 0.710 mmol) in methanol (20 mL) was added Pd—C (756 mg, 7.10 mmol). The reaction was hydrogenated at 60° C. using H-cube (4 atm) for 72 h. The mixture was diluted with DMF (20 mL). The mixture was filtered and the filtrate was concentrated under reduced pressure to provide the crude product. The crude product was purified by preparative HPLC (Gemini-C18 column, 5µ silica, 21 x 150 mm; 30-40% gradient of ACN/water with 0.1% TFA modifier) to afford 1-(((4R,5R)-5-((5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-hydroxypropoxy)-1H-benzo[d]imidazole-5-carboxamide (30 mg, 0.035 mmol, 4.9% yield) as an off-white solid. 1 H NMR (400 MHz, MeOH- d4 ) δ 7.58 (s, 1 H), 7.48 (d, J = 8.4 Hz, 1 H), 7.33 (s, 1 H), 7.17 (d, J = 8.4 Hz, 1 H), 6.89 (s, 1 H), 6.58 (s, 2 H), 5.11- 4.99 (m, 1 H), 4.93 (s, 1 H), 4.61 (dqd, J = 26.0, 13.2, 6.9 Hz, 5 H), 4.46- 4.36 (m, 1 H), 4.31 (dd, J = 13.4, 3.2 Hz, 1 H), 4.19 (dd, J = 14.0, 3.4Hz, 1 H), 4.03 (dd, J = 15.1, 6.4 Hz, 1 H), 3.88 (dd, J = 14.9, 6.4 Hz, 1 H), 3.83 - 3.72 (m, 1 H), 2.23 (s, 3 H), 2.18 (s, 3 H), 2.05 (dd, J = 11.5, 5.9 Hz, 2 H), 1.64 (d, J = 10.3 Hz, 6 H), 1.50 - 1.28 (m, 6 H). LCMS ( m/z ): 825 [M+ H] + .
步骤7: (E)-1-(((4R,5R)-5-(((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-(3-羟基丙氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 7: (E)-1-(((4R,5R)-5-(((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-(3-hydroxypropoxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向1-(((4R,5R)-5-((5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)甲基)-2,2-二甲基-1,3-二氧杂环戊烷-4-基)甲基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-羟基丙氧基)-1H-苯并[d]咪唑-5-甲酰胺 (21 mg,0.025 mmol)和碳酸铯 (24.88 mg, 0.076 mmol)在DMF (0.5 mL)中的冰浴冷却的混合物加入碘甲烷 (4 µL, 0.064 mmol)。移去冰浴并在室温搅拌该混合物16 h。在过滤后,通过质量导向的制备型HPLC (XSELECT CSH C18, 5 um填充物, 150x30 mm柱, 30-85%梯度的MeCN/10 mM碳酸氢铵,用氨将其调节成pH10)直接纯化滤液。纯级分的浓缩提供标题化合物(12 mg, 0.014 mmol, 55%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 1.28 - 1.35 (m,12 H) 1.89 - 1.94 (m, 2 H) 2.08 (s, 3 H) 2.13 (s, 3 H) 3.28 (s, 3 H) 3.30 (s,3 H) 3.53 - 3.57 (m, 2 H) 4.01 - 4.07 (m, 1 H) 4.11 - 4.16 (m, 1 H) 4.27 -4.31 (m, 2 H) 4.37 (dd, J=12.67, 4.31 Hz, 1 H) 4.49 - 4.56 (m, 6 H) 4.60 -4.66 (m, 2 H) 6.59 (d, J=7.10 Hz, 2 H) 7.27 (s, 1 H) 7.42 - 7.50 (m, 4 H)7.72 (dd, J=8.49, 1.39 Hz, 1 H) 7.79 (s, 1 H) 8.05 (d, J=16.48 Hz, 2 H)。LCMS(m/z): 853.4 [M + H]+。To an ice-bath cooled mixture of 1-(((4R,5R)-5-((5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)methyl)-2,2-dimethyl-1,3-dioxolan-4-yl)methyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-hydroxypropoxy)-1H-benzo[d]imidazole-5-carboxamide (21 mg, 0.025 mmol) and cesium carbonate (24.88 mg, 0.076 mmol) in DMF (0.5 mL) was added iodomethane (4 µL, 0.064 mmol). The ice bath was removed and the mixture was stirred at room temperature for 16 h. After filtration, the filtrate was directly purified by mass-directed preparative HPLC (XSELECT CSH C18, 5 um packing, 150x30 mm column, 30-85% gradient of MeCN/10 mM ammonium bicarbonate, adjusted to pH 10 with ammonia). Concentration of pure fractions afforded the title compound (12 mg, 0.014 mmol, 55% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 1.28 - 1.35 (m,12 H) 1.89 - 1.94 (m, 2 H) 2.08 (s, 3 H) 2.13 (s, 3 H) 3.28 (s, 3 H) 3.30 (s,3 H) 3.53 - 3.57 (m, 2 H) 4.01 - 4.07 (m, 1 H) 4.11 - 4.16 (m, 1 H) 4.27 -4.31 (m, 2 H) 4.37 (dd, J =12.67, 4.31 Hz, 1 H) 4.49 - 4.56 (m, 6 H) 4.60 -4.66 (m, 2 H) 6.59 (d, J =7.10 Hz, 2 H) 7.27 (s, 1 H) 7.42 - 7.50 (m, 4 H)7.72 (dd, J =8.49, 1.39 Hz, 1 H) 7.79 (s, 1 H) 8.05 (d, J =16.48 Hz, 2 H). LCMS ( m/z ): 853.4 [M + H] + .
实施例37Embodiment 37
(E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-异丙基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-isopropyl-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
步骤1: 4-((4-氨基-2,2,3,3-四氟丁基)氨基)-3-溴-5-硝基苯甲酰胺Step 1: 4-((4-amino-2,2,3,3-tetrafluorobutyl)amino)-3-bromo-5-nitrobenzamide
在室温向3-溴-4-氟-5-硝基苯甲酰胺 (1.4 g, 5.32 mmol)和2,2,3,3-四氟丁烷-1,4-二胺, 2盐酸盐 (1.3 g, 5.58 mmol)在乙醇(30 mL)中的溶液加入DIEA (3.53mL, 20.23 mmol)。然后,将该反应混合物温热至70 ℃并搅拌4 h。将该反应混合物冷却至室温并然后浓缩。在水和EtOAc之间分配所得物质。将水层分离并采用EtOAc (1x)萃取。然后,用盐水洗涤合并的有机层,采用硫酸镁干燥并浓缩以获得作为黄色固体的4-((4-氨基-2,2,3,3-四氟丁基)氨基)-3-溴-5-硝基苯甲酰胺 (2.15 g, 5.3 mmol, 100%收率)。不经进一步纯化而使用该固体。LCMS (m/z): 403.0 [M + H]+。To a solution of 3-bromo-4-fluoro-5-nitrobenzamide (1.4 g, 5.32 mmol) and 2,2,3,3-tetrafluorobutane-1,4-diamine, 2 hydrochloride (1.3 g, 5.58 mmol) in ethanol (30 mL) was added DIEA (3.53 mL, 20.23 mmol) at room temperature. Then, the reaction mixture was warmed to 70 ° C and stirred for 4 h. The reaction mixture was cooled to room temperature and then concentrated. The resulting material was distributed between water and EtOAc. The aqueous layer was separated and extracted with EtOAc (1x). Then, the combined organic layer was washed with brine, dried with magnesium sulfate and concentrated to obtain 4-((4-amino-2,2,3,3-tetrafluorobutyl)amino)-3-bromo-5-nitrobenzamide (2.15 g, 5.3 mmol, 100% yield) as a yellow solid. The solid was used without further purification. LCMS ( m/z ): 403.0 [M + H] + .
步骤2: 3-溴-4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-硝基苯甲酰胺Step 2: 3-Bromo-4-((4-((4-carbamoyl-2-nitrophenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-nitrobenzamide
在室温向4-氟-3-硝基苯甲酰胺 (1.5 g, 8.15 mmol)和4-((4-氨基-2,2,3,3-四氟丁基)氨基)-3-溴-5-硝基苯甲酰胺 (2.15 g, 5.3 mmol)在乙醇(25 mL)中的悬浮液加入DIEA (2.8 mL, 16.03 mmol)。然后,将该反应混合物温热至80 ℃并搅拌48 h。然后,将该反应混合物冷却至室温并过滤。采用EtOH洗涤固体,然后干燥以获得作为黄色固体的3-溴-4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-硝基苯甲酰胺 (2.6 g, 4.58 mmol, 86%收率)。LCMS (m/z): 567.0 [M + H]+。To a suspension of 4-fluoro-3-nitrobenzamide (1.5 g, 8.15 mmol) and 4-((4-amino-2,2,3,3-tetrafluorobutyl)amino)-3-bromo-5-nitrobenzamide (2.15 g, 5.3 mmol) in ethanol (25 mL) was added DIEA (2.8 mL, 16.03 mmol) at room temperature. The reaction mixture was then warmed to 80 °C and stirred for 48 h. The reaction mixture was then cooled to room temperature and filtered. The solid was washed with EtOH and then dried to obtain 3-bromo-4-((4-((4-carbamoyl-2-nitrophenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-nitrobenzamide (2.6 g, 4.58 mmol, 86% yield) as a yellow solid. LCMS ( m/z ): 567.0 [M + H] + .
步骤3: 4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-3-硝基-5-(丙-1-烯-2-基)苯甲酰胺Step 3: 4-((4-((4-Carbamoyl-2-nitrophenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-3-nitro-5-(prop-1-en-2-yl)benzamide
在室温向装有在DMF (2.5 mL)和水(0.25 mL)中的3-溴-4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-硝基苯甲酰胺 (300 mg, 0.529 mmol)、三氟(丙-1-烯-2-基)-l4-硼烷, 钾盐 (196 mg, 1.322 mmol)和K3PO4 (393 mg, 1.851mmol)的40-mL闪烁小瓶加入PdCl2(dppf)-CH2Cl加合物(44 mg, 0.054 mmol)。然后,排空该反应容器并反填充氮气。然后,将该反应混合物温热至80 ℃并搅拌过夜。当冷却至室温时,用EtOAc和水稀释该混合物。将该双相混合物过滤通过硅藻土垫。然后,分离水层并用水洗涤有机层(再两次)。然后用EtOAc (1x)反萃取合并的水层。用饱和盐水洗涤合并的有机层,用硫酸镁干燥并浓缩。通过正相硅胶层析法 (0-20%梯度的MeOH/DCM)纯化残余物以提供作为橙色固体的4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-3-硝基-5-(丙-1-烯-2-基)苯甲酰胺 (111 mg, 0.211 mmol, 40%收率)。不经进一步纯化而使用该固体。LCMS (m/z): 529.2 [M + H]+。To a 40-mL scintillation vial containing 3-bromo-4-((4-((4-carbamoyl-2-nitrophenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-nitrobenzamide (300 mg, 0.529 mmol), trifluoro(prop-1-en-2-yl)-14-borane, potassium salt (196 mg, 1.322 mmol) and K 3 PO 4 (393 mg, 1.851 mmol) in DMF (2.5 mL) and water (0.25 mL) was added PdCl 2 (dppf)-CH 2 Cl adduct (44 mg, 0.054 mmol) at room temperature. The reaction vessel was then evacuated and backfilled with nitrogen. The reaction mixture was then warmed to 80 °C and stirred overnight. When cooled to room temperature, the mixture was diluted with EtOAc and water. The two-phase mixture is filtered through a diatomaceous earth pad. Then, the aqueous layer is separated and the organic layer is washed with water (twice again). The aqueous layer merged is then extracted with EtOAc (1x). The organic layer merged is washed with saturated brine, dried over magnesium sulfate and concentrated. The residue is purified by normal phase silica gel chromatography (0-20% gradient of MeOH/DCM) to provide 4-((4-((4-carbamoyl-2-nitrophenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-3-nitro-5-(prop-1-ene-2-yl)benzamide (111 mg, 0.211 mmol, 40% yield) as an orange solid. The solid is used without further purification. LCMS ( m/z ): 529.2 [M+H] + .
步骤4: 3-氨基-4-((4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-异丙基苯甲酰胺Step 4: 3-amino-4-((4-((2-amino-4-carbamoylphenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-isopropylbenzamide
在氮气下向4-((4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-3-硝基-5-(丙-1-烯-2-基)苯甲酰胺 (121 mg, 0.229 mmol)在甲醇 (20 mL)中的溶液加入10% Pd/C (26 mg, 0.024 mmol)。将容器的气氛交换为氢气(气球)并搅拌该混合物过夜。在除去氢气后,LCMS分析显示烯烃未完全还原。通过过滤除去Pd催化剂并浓缩该混合物。然后,将残余物溶解于甲醇 (20 mL)并进一步还原(H-Cube, 50 psi氢气, 30 ℃, 1h, Pd/C筒)。在溶剂蒸发后,获得低纯度的浅棕色固体(通过UV测得~40%) (72 mg)并不经进一步纯化而使用。LCMS (m/z): 471.1 [M + H]+。To a solution of 4-((4-((4-carbamoyl-2-nitrophenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-3-nitro-5-(prop-1-en-2-yl)benzamide (121 mg, 0.229 mmol) in methanol (20 mL) under nitrogen was added 10% Pd/C (26 mg, 0.024 mmol). The atmosphere of the vessel was exchanged to hydrogen (balloon) and the mixture was stirred overnight. After removal of hydrogen, LCMS analysis showed that the olefin was not completely reduced. The Pd catalyst was removed by filtration and the mixture was concentrated. The residue was then dissolved in methanol (20 mL) and further reduced (H-Cube, 50 psi hydrogen, 30 °C, 1 h, Pd/C cartridge). After evaporation of the solvent, a light brown solid of low purity (~40% by UV) (72 mg) was obtained and used without further purification. LCMS ( m/z ): 471.1 [M + H] + .
步骤5: 1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-异丙基-1H-苯并[d]咪唑-5-甲酰胺Step 5: 1-(4-(5-Carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-isopropyl-1H-benzo[d]imidazol-5-carboxamide
在室温向3-氨基-4-((4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-异丙基苯甲酰胺 (72 mg, ~40%纯度)在DMF (1.5 mL)中的溶液加入1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯溶液 (1 M 在1,4-二氧杂环己烷中, 0.306 mL,0.306 mmol)。然后,在室温搅拌该反应混合物1 h。然后,在室温向该混合物加入EDC (110mg, 0.574 mmol)和TEA (0.160 mL, 1.148 mmol)。在搅拌过夜之后,过滤该混合物并通过质量导向的反相HPLC (XSELECT CSH C18, 5 um填充物, 150x30 mm 柱, 30-85%梯度的MeCN/水,含0.1% TFA 改性剂)直接纯化滤液以提供1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-异丙基-1H-苯并[d]咪唑-5-甲酰胺 (31 mg, 0.039 mmol,26%收率)。LCMS (m/z): 793.4 [M + H]+。To a solution of 3-amino-4-((4-((2-amino-4-carbamoylphenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-isopropylbenzamide (72 mg, ~40% purity) in DMF (1.5 mL) was added 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate solution (1 M in 1,4-dioxane, 0.306 mL, 0.306 mmol) at room temperature. The reaction mixture was then stirred at room temperature for 1 h. EDC (110 mg, 0.574 mmol) and TEA (0.160 mL, 1.148 mmol) were then added to the mixture at room temperature. After stirring overnight, the mixture was filtered and the filtrate was directly purified by mass-directed reverse phase HPLC (XSELECT CSH C18, 5 um packing, 150x30 mm column, 30-85% gradient of MeCN/water with 0.1% TFA modifier) to provide 1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-isopropyl-1H-benzo[d]imidazole-5-carboxamide (31 mg, 0.039 mmol, 26% yield). LCMS ( m/z ): 793.4 [M+H] + .
步骤6: (E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-异丙基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 6: (E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-isopropyl-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
在室温向1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-异丙基-1H-苯并[d]咪唑-5-甲酰胺 (22 mg, 0.028 mmol)和碳酸铯 (46 mg, 0.141 mmol)在DMF (1 mL)中的混合物加入碘甲烷(5 µL, 0.080 mmol)。然后,搅拌该反应混合物5 h。过滤该混合物并通过质量导向的反相HPLC (XSELECT CSH C18, 5 um填充物, 150x30 mm柱,15-55%梯度的MeCN/水,含0.075% NH4OH, 10 mM碳酸氢铵, pH 10)直接纯化滤液以获得作为白色固体的(E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-异丙基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (6mg, 7.3 umol, 26%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.14 - 8.05 (m, 3 H),7.97 - 7.85 (m, 3 H), 7.66 - 7.57 (m, 1 H), 7.54 - 7.43 (m, 2 H), 6.52 (s, 2H), 5.33 - 5.12 (m, 4 H), 4.55 - 4.45 (m, 4 H), 3.61 (s, 3 H), 3.56 (s, 3 H),2.13 (s, 3 H), 2.12 (s, 3 H), 1.30 - 1.19 (m, 13 H)。LCMS (m/z): 821.4 [M + H]+。To a mixture of 1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-isopropyl-1H-benzo[d]imidazole-5-carboxamide (22 mg, 0.028 mmol) and cesium carbonate (46 mg, 0.141 mmol) in DMF (1 mL) was added iodomethane (5 µL, 0.080 mmol) at room temperature. The reaction mixture was then stirred for 5 h. The mixture was filtered and the filtrate was directly purified by mass-directed reverse phase HPLC (XSELECT CSH C18, 5 um packing, 150x30 mm column, 15-55% gradient of MeCN/water with 0.075% NH4OH , 10 mM ammonium bicarbonate, pH 10) to afford (E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-isopropyl-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (6 mg, 7.3 g) as a white solid. umol, 26% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.14 - 8.05 (m, 3 H), 7.97 - 7.85 (m, 3 H), 7.66 - 7.57 (m, 1 H), 7.54 - 7.43 (m, 2 H), 6.52 (s, 2H), 5.33 - 5.12 (m, 4 H), 4.55 - 4.45 (m, 4 H), 3.61 (s, 3 H), 3.56 (s, 3 H), 2.13 (s, 3 H), 2.12 (s, 3 H), 1.30 - 1.19 (m, 13 H). LCMS ( m/z ): 821.4 [M + H] + .
实施例38Embodiment 38
(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-(4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-(4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
步骤1: 4-((4-氨基-2,2,3,3-四氟丁基)氨基)-3-((4-甲氧基苄基)氧基)-5-硝基苯甲酰胺Step 1: 4-((4-amino-2,2,3,3-tetrafluorobutyl)amino)-3-((4-methoxybenzyl)oxy)-5-nitrobenzamide
在室温向4-氯-3-((4-甲氧基苄基)氧基)-5-硝基苯甲酰胺 (1.2 g, 3.56 mmol)和2,2,3,3-四氟丁烷-1,4-二胺, 2盐酸盐 (1 g, 4.29 mmol)在1-丁醇 (40 mL)中的悬浮液加入碳酸氢钠(1.078 g, 12.83 mmol)。然后,将该反应混合物温热至120 ℃并搅拌5天。将该混合物冷却至室温并用水淬灭。用EtOAc (3x)萃取水相。将所形成的的乳液过滤通过硅藻土饼(a cake of Celite)。用盐水洗涤合并的有机层,干燥并浓缩至硅藻土上。正相层析法(40 g柱,0-8% 梯度的MeOH/DCM)提供作为橙色固体的4-((4-氨基-2,2,3,3-四氟丁基)氨基)-3-((4-甲氧基苄基)氧基)-5-硝基苯甲酰胺 (349 mg, 0.758 mmol, 21.3%收率)。LCMS (m/z): 461.2 [M + H]+。To a suspension of 4-chloro-3-((4-methoxybenzyl)oxy)-5-nitrobenzamide (1.2 g, 3.56 mmol) and 2,2,3,3-tetrafluorobutane-1,4-diamine, 2 hydrochloride (1 g, 4.29 mmol) in 1-butanol (40 mL) was added sodium bicarbonate (1.078 g, 12.83 mmol) at room temperature. The reaction mixture was then warmed to 120 °C and stirred for 5 days. The mixture was cooled to room temperature and quenched with water. The aqueous phase was extracted with EtOAc (3x). The resulting emulsion was filtered through a cake of Celite. The combined organic layers were washed with brine, dried and concentrated onto Celite. Normal phase chromatography (40 g column, 0-8% gradient of MeOH/DCM) provided 4-((4-amino-2,2,3,3-tetrafluorobutyl)amino)-3-((4-methoxybenzyl)oxy)-5-nitrobenzamide (349 mg, 0.758 mmol, 21.3% yield) as an orange solid. LCMS ( m/z ): 461.2 [M+H] + .
步骤2: 3-((4-甲氧基苄基)氧基)-5-硝基-4-((2,2,3,3-四氟-4-((2-硝基苯基)氨基)丁基)氨基)苯甲酰胺Step 2: 3-((4-methoxybenzyl)oxy)-5-nitro-4-((2,2,3,3-tetrafluoro-4-((2-nitrophenyl)amino)butyl)amino)benzamide
在室温向1-氟-2-硝基苯 (0.16 mL, 1.52 mmol)和1-氟-2-硝基苯 (0.16 mL,1.52 mmol)在1-丁醇 (4 mL)中的悬浮液加入碳酸氢钠(191 mg, 2.27 mmol)。然后,将该反应混合物温热至80 ℃并搅拌12天。将该混合物冷却至室温并用水淬灭。用EtOAc (3x)萃取水层。用盐水洗涤合并的有机层,干燥并浓缩。将残余物悬浮在DCM中。然后,将固体过滤,用DCM洗涤并干燥以获得作为橙色固体的 3-((4-甲氧基苄基)氧基)-5-硝基-4-((2,2,3,3-四氟-4-((2-硝基苯基)氨基)丁基)氨基)苯甲酰胺 (330 mg, 0.569 mmol, 75%收率)。LCMS (m/z): 582.2 [M + H]+。Sodium bicarbonate (191 mg, 2.27 mmol) was added to a suspension of 1-fluoro-2-nitrobenzene (0.16 mL, 1.52 mmol) and 1-fluoro-2-nitrobenzene (0.16 mL, 1.52 mmol) in 1-butanol (4 mL) at room temperature. Then, the reaction mixture was warmed to 80 ° C and stirred for 12 days. The mixture was cooled to room temperature and quenched with water. The aqueous layer was extracted with EtOAc (3x). The combined organic layer was washed with brine, dried and concentrated. The residue was suspended in DCM. Then, the solid was filtered, washed with DCM and dried to obtain 3-((4-methoxybenzyl)oxy)-5-nitro-4-((2,2,3,3-tetrafluoro-4-((2-nitrophenyl)amino)butyl)amino)benzamide (330 mg, 0.569 mmol, 75% yield) as an orange solid. LCMS ( m/z ): 582.2 [M + H] + .
步骤3: 3-氨基-4-((4-((2-氨基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-((4-甲氧基苄基)氧基)苯甲酰胺Step 3: 3-amino-4-((4-((2-aminophenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-((4-methoxybenzyl)oxy)benzamide
在室温向3-((4-甲氧基苄基)氧基)-5-硝基-4-((2,2,3,3-四氟-4-((2-硝基苯基)氨基)丁基)氨基)苯甲酰胺 (328 mg, 0.564 mmol)和氯化铵 (302 mg, 5.64 mmol)在甲醇 (5 mL)中的溶液加入锌(369 mg, 5.64 mmol)。然后,在室温搅拌该反应混合物过夜。将该混合物过滤通过硅藻土,浓缩,并在水和EtOAc之间分配。分离水层并用EtOAc (1x)萃取。然后,用盐水洗涤合并的有机层,经硫酸镁干燥并浓缩。将残余物悬浮在DCM中。将固体过滤、用DCM洗涤并干燥以提供不纯的标题化合物(70 mg, ~24%收率)。浓缩滤液并通过质量导向的制备型HPLC (XSELECT CSH C18, 5 um填充物, 150x30 mm柱, 15-55%梯度的MeCN/水,含 0.075% NH4OH, 10 mM碳酸氢铵, pH 10)纯化残余物以提供作为浅棕色固体的纯的3-氨基-4-((4-((2-氨基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-((4-甲氧基苄基)氧基)苯甲酰胺 (92 mg, 0.18 mmol, 31%收率)。LCMS (m/z): 522.3 [M + H]+。To a solution of 3-((4-methoxybenzyl)oxy)-5-nitro-4-((2,2,3,3-tetrafluoro-4-((2-nitrophenyl)amino)butyl)amino)benzamide (328 mg, 0.564 mmol) and ammonium chloride (302 mg, 5.64 mmol) in methanol (5 mL) was added zinc (369 mg, 5.64 mmol) at room temperature. The reaction mixture was then stirred overnight at room temperature. The mixture was filtered through diatomaceous earth, concentrated, and partitioned between water and EtOAc. The aqueous layer was separated and extracted with EtOAc (1x). The combined organic layers were then washed with brine, dried over magnesium sulfate and concentrated. The residue was suspended in DCM. The solid was filtered, washed with DCM and dried to provide impure title compound (70 mg, ~24% yield). The filtrate was concentrated and the residue was purified by mass-directed preparative HPLC (XSELECT CSH C18, 5 um packing, 150x30 mm column, 15-55% gradient of MeCN/water with 0.075% NH4OH, 10 mM ammonium bicarbonate, pH 10) to afford pure 3-amino-4-((4-((2-aminophenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-((4-methoxybenzyl)oxy)benzamide (92 mg, 0.18 mmol, 31% yield) as a light brown solid. LCMS ( m/z ): 522.3 [M+H] + .
步骤4: 2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1-(4-(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-7-((4-甲氧基苄基)氧基)-1H-苯并[d]咪唑-5-甲酰胺Step 4: 2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1-(4-(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-7-((4-methoxybenzyl)oxy)-1H-benzo[d]imidazol-5-carboxamide
在0 ℃向3-氨基-4-((4-((2-氨基苯基)氨基)-2,2,3,3-四氟丁基)氨基)-5-((4-甲氧基苄基)氧基)苯甲酰胺 (92 mg, 0.176 mmol)在DMF (1 mL)中的溶液加入1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯溶液 (1 M 在1,4-二氧杂环己烷中, 0.370 mL, 0.370mmol)。然后,在室温搅拌该反应混合物1 h。然后,在室温向该反应混合物加入EDC (127mg, 0.662 mmol)和TEA (0.184 mL, 1.323 mmol)。然后,搅拌该反应混合物2 h。用水稀释该混合物。将固体过滤,用DCM和水洗涤并干燥以获得作为灰白色固体的2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1-(4-(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-7-((4-甲氧基苄基)氧基)-1H-苯并[d]咪唑-5-甲酰胺(85 mg, 0.10 mmol, 57%收率)。LCMS (m/z): 844.4 [M + H]+。To a solution of 3-amino-4-((4-((2-aminophenyl)amino)-2,2,3,3-tetrafluorobutyl)amino)-5-((4-methoxybenzyl)oxy)benzamide (92 mg, 0.176 mmol) in DMF (1 mL) was added 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate solution (1 M in 1,4-dioxane, 0.370 mL, 0.370 mmol) at 0 °C. Then, the reaction mixture was stirred at room temperature for 1 h. Then, EDC (127 mg, 0.662 mmol) and TEA (0.184 mL, 1.323 mmol) were added to the reaction mixture at room temperature. Then, the reaction mixture was stirred for 2 h. The mixture was diluted with water. The solid was filtered, washed with DCM and water and dried to afford 2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1-(4-(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-7-((4-methoxybenzyl)oxy)-1H-benzo[d]imidazole-5-carboxamide (85 mg, 0.10 mmol, 57% yield) as an off-white solid. LCMS ( m/z ): 844.4 [M+H] + .
步骤5: (E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-(4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 5: (E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-(4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
在室温向2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1-(4-(2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-7-((4-甲氧基苄基)氧基)-1H-苯并[d]咪唑-5-甲酰胺 (65 mg, 0.077 mmol)和碳酸铯 (125 mg, 0.385mmol)在DMF (1 mL)中的混合物加入碘甲烷(0.012 mL, 0.19 mmol)。然后,将该反应混合物搅拌过夜。过滤该反应混合物并通过质量导向的反相HPLC (XSELECT CSH C18, 5 um填充物, 150x30 mm柱, 50-99%梯度的MeCN/水,含0.075% NH4OH, 10 mM碳酸氢铵, pH 10)直接纯化滤液以提供作为灰白色固体的(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-(4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,2,3,3-四氟丁基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (33 mg, 0.038 mmol, 49%收率)。1H NMR (400 MHz, DMSO-d 6) δppm 8.10 (s, 1 H), 7.78 (d, J=1.0 Hz, 1 H), 7.68 - 7.65 (m, 1 H), 7.61 (dd, J=1.9, 7.0 Hz, 1 H), 7.52 (s, 1 H), 7.48 - 7.33 (m, 5 H), 6.77 (d, J=8.6 Hz, 2H), 6.49 (d, J=0.8 Hz, 2 H), 5.25 - 5.09 (m, 4 H), 4.72 (t, J=16.6 Hz, 2 H),4.49 (q, J=7.0 Hz, 4 H), 3.58 (s, 6 H), 3.49 (s, 3 H), 2.12 (s, 3 H), 2.11(s, 3 H), 1.23 (t, J=7.1 Hz, 3 H), 1.24 (t, J=7.1 Hz, 3 H)。LCMS (m/z): 872.4[M + H]+。To a mixture of 2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1-(4-(2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-7-((4-methoxybenzyl)oxy)-1H-benzo[d]imidazole-5-carboxamide (65 mg, 0.077 mmol) and cesium carbonate (125 mg, 0.385 mmol) in DMF (1 mL) was added iodomethane (0.012 mL, 0.19 mmol) at room temperature. The reaction mixture was then stirred overnight. The reaction mixture was filtered and the filtrate was directly purified by mass directed reverse phase HPLC (XSELECT CSH C18, 5 um packing, 150x30 mm column, 50-99% gradient of MeCN/water with 0.075% NH4OH , 10 mM ammonium bicarbonate, pH 10) to afford (E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-(4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,2,3,3-tetrafluorobutyl)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (33 mg, 4% by weight) as an off-white solid. 0.038 mmol, 49% yield). 1 H NMR (400 MHz, DMSO- d 6 ) δppm 8.10 (s, 1 H), 7.78 (d, J =1.0 Hz, 1 H), 7.68 - 7.65 (m, 1 H), 7.61 (dd, J =1.9, 7.0 Hz, 1 H), 7.52 (s, 1 H), 7.48 - 7.33 (m, 5 H), 6.77 (d, J =8.6 Hz, 2H), 6.49 (d, J =0.8 Hz, 2 H), 5.25 - 5.09 (m, 4 H), 4.72 (t, J =16.6 Hz, 2 H), 4.49 (q, J =7.0 Hz, 4 H), 3.58 (s, 6 H), 3.49 (s, 3 H), 2.12 (s, 3 H), 2.11 (s, 3 H), 1.23 (t, J =7.1 Hz, 3 H), 1.24 (t, J =7.1 Hz, 3 H). LCMS ( m/z ): 872.4[M + H] + .
实施例39Embodiment 39
(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-((四氢呋喃-3-基)甲氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-((tetrahydrofuran-3-yl)methoxy)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-羟基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (49 mg, 0.063 mmol)在DMF (1 mL)中的溶液加入3-(溴甲基)四氢呋喃(20.95 mg,0.127 mmol),然后加入碳酸钾 (11.40 mg, 0.083 mmol)。在90℃搅拌该反应混合物24 h。通过制备型HPLC (Phenomenex Eclipse, 5 um填充物, 50x30 mm柱, 25-55%梯度的MeCN/水,含0.1% TFA 改性剂)直接纯化该混合物。合并相应的级分并在真空中浓缩。在EtOAc和碳酸氢钠水溶液之间分配残余物。分离有机层、经硫酸钠干燥并在真空中蒸发以提供作为白色固体的(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-((四氢呋喃-3-基)甲氧基)-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (22.5 mg, 0.027 mmol, 42.6%收率)。1H NMR (400 MHz,DMSO-d 6) δ ppm 8.11 - 8.15 (m, 1 H), 8.05 - 8.11 (m, 1 H), 7.99 - 8.05 (m, 1H), 7.79 (s, 2 H), 7.50 (br. s., 3 H), 7.18 - 7.28 (m, 1 H), 6.46 (s, 1 H),6.37 (s, 1 H), 5.06 (br. s., 2 H), 4.86 (br. s., 2 H), 4.42 - 4.56 (m, 4 H),3.97 - 4.12 (m, 2 H), 3.65 - 3.73 (m, 1 H), 3.59 (s, 3 H), 3.57 (s, 3 H),3.45 - 3.53 (m, 2 H), 2.11 (s, 3 H), 2.08 (s, 3 H), 1.62 (br. s., 4 H), 1.48(br. S., 4 H), 1.15 - 1.36 (m, 8 H)。LCMS (m/z): 833.5 [M + H]+。To a solution of (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-hydroxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (49 mg, 0.063 mmol) in DMF (1 mL) was added 3-(bromomethyl)tetrahydrofuran (20.95 mg, 0.127 mmol) followed by potassium carbonate (11.40 mg, 0.083 mmol). The reaction mixture was stirred at 90 °C for 24 h. The mixture was directly purified by preparative HPLC (Phenomenex Eclipse, 5 um packing, 50x30 mm column, 25-55% gradient of MeCN/water with 0.1% TFA modifier). The corresponding fractions were combined and concentrated in vacuo. The residue was partitioned between EtOAc and aqueous sodium bicarbonate solution. The organic layer was separated, dried over sodium sulfate and evaporated in vacuo to afford (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-((tetrahydrofuran-3-yl)methoxy)-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (22.5 mg, 0.027 mmol, 42.6% yield) as a white solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.11 - 8.15 (m, 1 H), 8.05 - 8.11 (m, 1 H), 7.99 - 8.05 (m, 1H), 7.79 (s, 2 H), 7.50 (br. s., 3 H), 7.18 - 7.28 ( 3 .59 (s, 3 H), 3.57 (s, 3 H), 3.45 - 3.53 (m, 2 H), 2.11 (s, 3 H), 2.08 (s, 3 H), 1.62 (br. s., 4 H), 1.48 (br. S., 4 H), 1.15 - 1.36 (m, 8 H). LCMS ( m/z ): 833.5 [M + H] + .
实施例40Embodiment 40
(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-羟基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-hydroxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
向(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (400 mg, 0.484 mmol)在DCM (8 mL)中的溶液逐滴加入HCl溶液 (4M 在二氧杂环己烷中, 0.726 mL, 2.91 mmol)。在真空中除去大部分DCM。加入1,4-二氧杂环己烷 (8 mL)和更多的HCl溶液(4M 在二氧杂环己烷中, 0.726 mL, 2.91 mmol)。声处理该容器并剧烈搅拌2.5 h (仍然存在胶状物并且反应未完全)。再次在真空中浓缩该混合物并再次悬浮在THF (6 mL)和水(1 mL)中。加入更多的HCl溶液(4M 在二氧杂环己烷中, 0.726 mL, 2.91mmol)并搅拌该混合物3 h。将反应混合物浓缩并溶解在20% 甲醇/DCM (6 mL)中。向这个均相溶液加入HCl溶液(4M 在二氧杂环己烷中, 0.726 mL, 2.91 mmol)并搅拌该混合物 30min。由于形成副产物,再次蒸发溶剂并用3:1 DCM:乙醇混合物(8 mL)和更多的HCl溶液(4M在二氧杂环己烷中, 0.726 mL, 2.91 mmol)替代。在室温搅拌该反应混合物48 h。将混合物浓缩并在10% 甲醇/DCM和饱和碳酸氢钠水溶液之间分配。用3:1 CHCl3:EtOH萃取水层两次。浓缩合并的有机相。通过硅胶层析法 (12 g 硅胶; 10-90% [3:1 EA:EtOH]/庚烷)部分纯化残余物。将粗固体悬浮于几毫升DMSO中。将未溶解的固体过滤、用DCM洗涤、干燥并发现对于合成另外的类似物而言足够纯(~90%, 149 mg)。随后,通过质量导向的HPLC (XSELECTCSH C18, 5 um填充物, 150x30 mm柱, 15-55%梯度的MeCN/水,含0.1% TFA 改性剂)纯化溶解的物质。将几滴饱和碳酸氢钠溶液加入各清洁级分。使用氮气流除去ACN。过滤悬浮固体、用水漂洗并干燥以提供作为白色固体的纯(E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-1-((E)-4-((E)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-羟基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (11.2 mg, 0.016 mmol, 3.2%)。1H NMR (DMSO-d6) δ ppm 8.10 (s, 1H), 7.99 (br. s., 1 H), 7.78 (dd, J=8.4, 1.4 Hz, 1 H), 7.42 (br. s., 1 H),7.20 (d, J=8.5 Hz, 1 H), 7.12 (t, J=8.0 Hz, 1 H), 7.01 (d, J=7.8 Hz, 1 H),6.75 (d, J=8.0 Hz, 1 H), 6.48 (s, 1 H 6.32 (s, 1 H), 5.08 (s, 2 H), 4.82 (s,2 H), 4.44-4.57 (m, 4 H), 3.58 (s, 3 H), 3.51 (s, 3 H), 2.14 (s, 3 H), 2.07(s, 3 H), 1.65 (s, 3 H), 1.48 (s, 3 H), 1.30 (t, J=7.2 Hz, 3 H), 1.25 (t, J=7.2 Hz, 3 H)。LCMS (m/z): 706.4 [M + H]+。To a solution of (E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (400 mg, 0.484 mmol) in DCM (8 mL) was added HCl solution (4M in dioxane, 0.726 mL, 2.91 mmol) dropwise. Most of the DCM was removed in vacuo. 1,4-Dioxane (8 mL) and more HCl solution (4M in dioxane, 0.726 mL, 2.91 mmol) were added. The vessel was sonicated and stirred vigorously for 2.5 h (gum was still present and the reaction was not complete). The mixture was concentrated again in vacuo and resuspended in THF (6 mL) and water (1 mL). More HCl solution (4M in dioxane, 0.726 mL, 2.91 mmol) was added and the mixture was stirred for 3 h. The reaction mixture was concentrated and dissolved in 20% methanol/DCM (6 mL). To this homogeneous solution was added HCl solution (4M in dioxane, 0.726 mL, 2.91 mmol) and the mixture was stirred for 30 min. Due to the formation of by-products, the solvent was evaporated again and replaced with a 3:1 DCM:ethanol mixture (8 mL) and more HCl solution (4M in dioxane, 0.726 mL, 2.91 mmol). The reaction mixture was stirred at room temperature for 48 h. The mixture was concentrated and distributed between 10% methanol/DCM and saturated sodium bicarbonate aqueous solution. The aqueous layer was extracted twice with 3:1 CHCl 3 :EtOH. The combined organic phase was concentrated. The residue was partially purified by silica gel chromatography (12 g silica gel; 10-90% [3:1 EA:EtOH]/heptane). The crude solid was suspended in several milliliters of DMSO. The undissolved solid was filtered, washed with DCM, dried and found to be pure enough (~90%, 149 mg) for the synthesis of other analogs. The dissolved material was then purified by mass-directed HPLC (XSELECTCSH C18, 5 um packing, 150x30 mm column, 15-55% gradient of MeCN/water with 0.1% TFA modifier). A few drops of saturated sodium bicarbonate solution were added to each clean fraction. ACN was removed using a stream of nitrogen. The suspended solid was filtered, rinsed with water and dried to provide pure (E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-1-((E)-4-((E)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-hydroxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (11.2 mg, 0.016 mmol, 3.2%) as a white solid. 1 H NMR (DMSO-d 6 ) δ ppm 8.10 (s, 1H), 7.99 (br. s., 1 H), 7.78 (dd, J =8.4, 1.4 Hz, 1 H), 7.42 (br. s., 1 H), 7.20 (d, J =8.5 Hz, 1 H), 7.12 (t, J =8.0 Hz, 1 H), 7.01 (d, J =7.8 Hz, 1 H), 6.75 (d, J =8.0 Hz, 1 H), 6.48 (s, 1 H 6.32 (s, 1 H), 5.08 (s, 2 H), 4.82 (s, 2 H), 4.44-4.57 (m, 4 H), 3.58 (s, 3H), 3.51 (s, 3 H), 2.14 (s, 3 H), 2.07 (s, 3 H), 1.65 (s, 3 H), 1.48 (s, 3 H), 1.30 (t, J =7.2 Hz, 3 H), 1.25 (t, J =7.2 Hz, 3 H). LCMS ( m/z ): 706.4 [M + H] + .
实施例41Embodiment 41
(E)-1-((2S,3S)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二乙氧基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-((2S,3S)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-diethoxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
步骤1: 4-(((2S,3S)-4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,3-二乙氧基丁基)氨基)-3-((4-甲氧基苄基)氧基)-5-硝基苯甲酰胺Step 1: 4-(((2S,3S)-4-((4-carbamoyl-2-nitrophenyl)amino)-2,3-diethoxybutyl)amino)-3-((4-methoxybenzyl)oxy)-5-nitrobenzamide
向4-氯-3-((4-甲氧基苄基)氧基)-5-硝基苯甲酰胺 (1.55 g, 4.60 mmol)在1-丁醇 (15 mL)中的混合物加入(2S,3S)-2,3-二乙氧基丁烷-1,4-二胺 (1.01 g, 5.75mmol)和DIEA (2.41 mL, 13.8 mmol)。在120℃搅拌该混合物2 h。然后,加入4-氟-3-硝基苯甲酰胺 (0.848 g, 4.60 mmol)。在120℃搅拌该混合物18 h。将该混合物冷却并过滤以除去悬浮固体。在除去溶剂后,硅胶层析法 (40 g硅胶,梯度的5-20% MeOH/DCM)提供作为橙色固体的4-(((2S,3S)-4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,3-二乙氧基丁基)氨基)-3-((4-甲氧基苄基)氧基)-5-硝基苯甲酰胺 (0.86 g, ~25%收率, 被~20%对称双-PMB-保护的副产物污染)。LCMS (m/z): 641.2 [M + H]+。To a mixture of 4-chloro-3-((4-methoxybenzyl)oxy)-5-nitrobenzamide (1.55 g, 4.60 mmol) in 1-butanol (15 mL) was added (2S,3S)-2,3-diethoxybutane-1,4-diamine (1.01 g, 5.75 mmol) and DIEA (2.41 mL, 13.8 mmol). The mixture was stirred at 120°C for 2 h. Then, 4-fluoro-3-nitrobenzamide (0.848 g, 4.60 mmol) was added. The mixture was stirred at 120°C for 18 h. The mixture was cooled and filtered to remove suspended solids. After removal of solvent, silica gel chromatography (40 g silica gel, gradient 5-20% MeOH/DCM) afforded 4-(((2S,3S)-4-((4-carbamoyl-2-nitrophenyl)amino)-2,3-diethoxybutyl)amino)-3-((4-methoxybenzyl)oxy)-5-nitrobenzamide (0.86 g, -25% yield, contaminated with -20% symmetric bis-PMB-protected byproduct) as an orange solid. LCMS ( m/z ): 641.2 [M+H] + .
所收集的沉淀物提供可以用于制备其它实施例的4,4'-(((2S,3S)-2,3-二乙氧基丁烷-1,4-二基)双(氮烷二基))双(3-((4-甲氧基苄基)氧基)-5-硝基苯甲酰胺) (423 mg,0.545 mmol, 12%收率, LCMS (m/z): 777.5 [M + H]+)。The collected precipitate provided 4,4'-(((2S,3S)-2,3-diethoxybutane-1,4-diyl)bis(azanediyl))bis(3-((4-methoxybenzyl)oxy)-5-nitrobenzamide) (423 mg, 0.545 mmol, 12% yield, LCMS ( m/z ): 777.5 [M+H] + ), which was used to prepare other examples.
步骤2: 3-氨基-4-(((2S,3S)-4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,3-二乙氧基丁基)氨基)-5-((4-甲氧基苄基)氧基)苯甲酰胺Step 2: 3-amino-4-(((2S,3S)-4-((2-amino-4-carbamoylphenyl)amino)-2,3-diethoxybutyl)amino)-5-((4-methoxybenzyl)oxy)benzamide
向100 mL 圆底烧瓶加入4-(((2S,3S)-4-((4-氨基甲酰基-2-硝基苯基)氨基)-2,3-二乙氧基丁基)氨基)-3-((4-甲氧基苄基)氧基)-5-硝基苯甲酰胺 (0.86 g, 1.342mmol)和甲醇 (20 mL)。向这个混合物加入10 mL饱和氯化铵水溶液。向这个混合物加入锌(0.878 g, 13.42 mmol)并在室温搅拌非均相混合物15 min。过滤该混合物并用MeOH漂洗滤饼。浓缩滤液。To a 100 mL round bottom flask was added 4-(((2S,3S)-4-((4-carbamoyl-2-nitrophenyl)amino)-2,3-diethoxybutyl)amino)-3-((4-methoxybenzyl)oxy)-5-nitrobenzamide (0.86 g, 1.342 mmol) and methanol (20 mL). To this mixture was added 10 mL of saturated aqueous ammonium chloride solution. To this mixture was added zinc (0.878 g, 13.42 mmol) and the heterogeneous mixture was stirred at room temperature for 15 min. The mixture was filtered and the filter cake was rinsed with MeOH. The filtrate was concentrated.
通过硅胶层析法 (24 g硅胶,6-20%梯度的 MeOH/DCM,含1% NH4OH作为改性剂)纯化粗产物以提供作为浅黄色固体的3-氨基-4-(((2S,3S)-4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,3-二乙氧基丁基)氨基)-5-((4-甲氧基苄基)氧基)苯甲酰胺 (0.127 g, 16%)。LCMS (m/z): 581.3 [M + H]+。The crude product was purified by silica gel chromatography (24 g silica gel, 6-20% gradient of MeOH/DCM with 1% NH4OH as modifier) to afford 3-amino-4-(((2S,3S)-4-((2-amino-4-carbamoylphenyl)amino)-2,3-diethoxybutyl)amino)-5-((4-methoxybenzyl)oxy)benzamide (0.127 g, 16%) as a light yellow solid. LCMS ( m/z ): 581.3 [M+H] + .
步骤3: 1-((2S,3S)-4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二乙氧基丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-((4-甲氧基苄基)氧基)-1H-苯并[d]咪唑-5-甲酰胺Step 3: 1-((2S,3S)-4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,3-diethoxybutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-((4-methoxybenzyl)oxy)-1H-benzo[d]imidazol-5-carboxamide
向3-氨基-4-(((2S,3S)-4-((2-氨基-4-氨基甲酰基苯基)氨基)-2,3-二乙氧基丁基)氨基)-5-((4-甲氧基苄基)氧基)苯甲酰胺 (0.127 g, 0.219 mmol)在DMF (6 mL)中的溶液加入1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (~0.4 在二氧杂环己烷中,1.094mL, 0.437 mmol)。搅拌该混合物15 min。加入EDC (0.105 g, 0.547 mmol)和TEA (0.152mL, 1.094 mmol)并在50℃搅拌该反应18 h。将反应混合物倾倒至3:1水:饱和氯化铵水溶液(20 mL)中。将所得固体过滤、用水洗涤,并干燥以提供作为固体的1-((2S,3S)-4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二乙氧基丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-((4-甲氧基苄基)氧基)-1H-苯并[d]咪唑-5-甲酰胺 (0.142 g, 0.157 mmol, 72%收率)。LCMS (m/z): 903.3 [M + H]+。To a solution of 3-amino-4-(((2S,3S)-4-((2-amino-4-carbamoylphenyl)amino)-2,3-diethoxybutyl)amino)-5-((4-methoxybenzyl)oxy)benzamide (0.127 g, 0.219 mmol) in DMF (6 mL) was added 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (~0.4 in dioxane, 1.094 mL, 0.437 mmol). The mixture was stirred for 15 min. EDC (0.105 g, 0.547 mmol) and TEA (0.152 mL, 1.094 mmol) were added and the reaction was stirred at 50 °C for 18 h. The reaction mixture was poured into 3:1 water:saturated aqueous ammonium chloride solution (20 mL). The resulting solid was filtered, washed with water, and dried to provide 1-((2S,3S)-4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,3-diethoxybutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-((4-methoxybenzyl)oxy)-1H-benzo[d]imidazole-5-carboxamide (0.142 g, 0.157 mmol, 72% yield) as a solid. LCMS ( m/z ): 903.3 [M+H] + .
步骤4: (E)-1-((2S,3S)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二乙氧基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 4: (E)-1-((2S,3S)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-diethoxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
向1-((2S,3S)-4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二乙氧基丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-((4-甲氧基苄基)氧基)-1H-苯并[d]咪唑-5-甲酰胺 (0.112 g, 0.124 mmol)在DMF (5mL)中的溶液加入碳酸铯 (0.121 g, 0.372 mmol)和碘甲烷 (0.031 mL, 0.496 mmol)。在室温搅拌该反应混合物18 h。用水稀释该反应混合物并用EtOAc (3 x 50 mL)萃取。用盐水(10 mL)洗涤有机相、用硫酸镁干燥、过滤并浓缩。使用质量导向的反相HPLC (XSELECT CSHC18, 5 um填充物, 150x30 mm柱, 15-55%梯度的MeCN/水,含0.075% NH4OH, 10 mM碳酸氢铵, pH 10)纯化粗产物。合并纯级分并浓缩以提供作为白色固体的(E)-1-((2S,3S)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二乙氧基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-((4-甲氧基苄基)氧基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (34 mg,0.36 mmol, 29%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 8.00 - 8.25 (m, 3 H) 7.86(dd, J=8.49, 1.39 Hz, 1 H) 7.77 (d, J=1.01 Hz, 1 H) 7.62 (s, 1 H) 7.36 - 7.53(m, 5 H) 6.75 (d, J=8.62 Hz, 2 H) 6.49 (d, J=6.84 Hz, 2 H) 5.22 (d, J=10.39Hz, 1 H) 5.11 (d, J=10.65 Hz, 1 H) 4.44 - 4.69 (m, 5 H) 4.29 - 4.42 (m, 1 H)3.93 - 4.06 (m, 1 H) 3.63 - 3.84 (m, 3 H) 3.58 (s, 3 H) 3.58 (s, 3 H) 3.40(s, 3 H) 3.09 - 3.28 (m, 2 H) 2.80 - 2.94 (m, 2 H) 2.12 (s, 3 H) 2.10 (s, 3H) 1.30 (t, J=7.10 Hz, 6 H) 0.58 (t, J=6.97 Hz, 3 H) 0.47 (t, J=6.97 Hz, 3H)。LCMS (m/z): 931.4 [M + H]+。To a solution of 1-((2S,3S)-4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,3-diethoxybutyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-((4-methoxybenzyl)oxy)-1H-benzo[d]imidazole-5-carboxamide (0.112 g, 0.124 mmol) in DMF (5 mL) was added cesium carbonate (0.121 g, 0.372 mmol) and iodomethane (0.031 mL, 0.496 mmol). The reaction mixture was stirred at room temperature for 18 h. The reaction mixture was diluted with water and extracted with EtOAc (3 x 50 mL). The organic phase was washed with brine (10 mL), dried over magnesium sulfate, filtered and concentrated. The crude product was purified using mass-directed reverse phase HPLC (XSELECT CSHC18, 5 um packing, 150x30 mm column, 15-55% gradient of MeCN/water with 0.075% NH4OH, 10 mM ammonium bicarbonate, pH 10). Pure fractions were combined and concentrated to afford (E)-1-((2S,3S)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-diethoxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-((4-methoxybenzyl)oxy)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (34 mg, 0.36 mmol, 29% yield) as a white solid. 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 8.00 - 8.25 (m, 3 H) 7.86 (dd, J =8.49, 1.39 Hz, 1 H) 7.77 (d, J =1.01 Hz, 1 H) 7.62 (s, 1 H) 7.36 - 7.53 (m, 5 H) 6. 75 (d, J =8.62 Hz, 2 H) 6.49 (d, J =6.84 Hz, 2 H) 5.22 (d, J =10.39Hz, 1 H) 5.11 (d, J =10.65 Hz, 1 H) 4.44 - 4.69 (m, 5 H) 4.29 - 4.42 (m, 1 H)3.93 - 4.06 (m, 1 H) 3.63 - 3.84 (m, 3 H) 3.58 (s, 3 H) 3.58 (s, 3 H) 3.40 (s, 3 H) 3.09 - 3.28 (m, 2 H) 2.80 - 2.94 (m, 2 H) 2.12 (s, 3 H) 2 .10 (s, 3H) 1.30 (t, J =7.10 Hz, 6 H) 0.58 (t, J =6.97 Hz, 3 H) 0.47 (t, J =6.97 Hz, 3H). LCMS ( m/z ): 931.4 [M + H] + .
表2示出实施例43-92,其可以根据如下示例的方法制备:Table 2 shows Examples 43-92, which can be prepared according to the method exemplified below:
。 .
实施例90Embodiment 90
(E)-1-((E)-4-((E)-7-溴-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-((E)-4-((E)-7-Bromo-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
步骤1: (E)-3-((4-((2-溴-6-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-4-硝基苯甲酰胺Step 1: (E)-3-((4-((2-bromo-6-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-4-nitrobenzamide
向(E)-4-((4-氨基-2,3-二甲基丁-2-烯-1-基)氨基)-3-硝基苯甲酰胺, 盐酸盐(1.05 g, 3.34 mmol)和1-溴-2-氟-3-硝基苯 (0.734 g, 3.34 mmol)在1-丁醇 (25 mL)中的亮黄色悬浮液加入DIEA (1.75 mL, 10.01 mmol)。在80℃搅拌该反应5 h,然后在室温搅拌16 h。将固体过滤并用丁醇 (20 mL)和水(3 x 20 mL)漂洗。干燥固体以提供作为橙色固体的标题化合物(1.16 g, 2.40 mmol, 72%收率)。1H NMR (DMSO-d6, 400 MHz) δ 8.66(d, J=2.0 Hz, 1 H), 8.3-8.4 (m, 1 H), 7.99 (br s, 1 H), 7.9-8.0 (m, 1 H),7.87 (dd, J=1.5, 7.8 Hz, 1 H), 7.81 (dd, J=1.5, 8.3 Hz, 1 H), 7.31 (br s, 1H), 6.8-6.9 (m, 1 H), 6.77 (d, J=9.0 Hz, 1 H), 6.06 (t, J=6.0 Hz, 1 H), 3.98(br d, J=5.8 Hz, 2 H), 3.85 (d, J=6.0 Hz, 2 H), 1.64 (s, 3 H), 1.59 (d, J=1.3Hz, 3 H)。LCMS (m/z): 478.2 [M + H]+。To a bright yellow suspension of (E)-4-((4-amino-2,3-dimethylbut-2-en-1-yl)amino)-3-nitrobenzamide, hydrochloride (1.05 g, 3.34 mmol) and 1-bromo-2-fluoro-3-nitrobenzene (0.734 g, 3.34 mmol) in 1-butanol (25 mL) was added DIEA (1.75 mL, 10.01 mmol). The reaction was stirred at 80 °C for 5 h and then at room temperature for 16 h. The solid was filtered and rinsed with butanol (20 mL) and water (3 x 20 mL). The solid was dried to provide the title compound (1.16 g, 2.40 mmol, 72% yield) as an orange solid. 1 H NMR (DMSO-d 6 , 400 MHz) δ 8.66 (d, J =2.0 Hz, 1 H), 8.3-8.4 (m, 1 H), 7.99 (br s, 1 H), 7.9-8.0 (m, 1 H), 7.87 (dd, J =1.5, 7.8 Hz, 1 H), 7.81 (dd , J =1.5, 8.3 Hz, 1 H), 7.31 (br s, 1H), 6.8-6.9 (m, 1 H), 6.77 (d, J =9.0 Hz, 1 H), 6.06 (t, J =6.0 Hz, 1 H), 3.98 (br d, J =5.8 Hz, 2 H), 3.85 (d, J =6.0 Hz, 2 H), 1.64 (s, 3 H), 1.59 (d, J =1.3Hz, 3 H). LCMS ( m/z ): 478.2 [M + H] + .
步骤2: (E)-4-氨基-3-((4-((2-氨基-6-溴苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)苯甲酰胺Step 2: (E)-4-amino-3-((4-((2-amino-6-bromophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)benzamide
向(E)-3-((4-((2-溴-6-硝基苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)-4-硝基苯甲酰胺 (1.06 g, 2.216 mmol)和氯化铵 (1.778 g, 33.2 mmol)在冷却在冰/水浴中的MeOH (50 mL)中的混合物加入锌(1.449 g, 22.16 mmol)并在室温搅拌该反应混合物40min。将该混合物过滤通过硅藻土并采用甲醇漂洗。浓缩滤液并通过硅胶层析法 (24 g硅胶柱;10-60% [3:1 EA:EtOH]/庚烷, 加1% NH4OH溶液, 12 min.;60% 梯度的[3:1 EA:EtOH]/庚烷, 5 min)纯化。将最纯的级分合并并浓缩。将混合的级分浓缩并再次纯化(12 g硅胶柱; 10-55% [3:1 EA:EtOH]/庚烷, 无NH4OH 改性剂, 10 min.; 55%梯度的 [3:1EA:EtOH]/庚烷, 5 min)。将各级分合并并干燥以提供作为褐色泡沫的标题化合物(571mg, 1.09 mmol, 49.3%收率)。LCMS (m/z): 418.3 [M + H]+, 通过UV210-350 nm测得~80%纯度)。To a mixture of (E)-3-((4-((2-bromo-6-nitrophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)-4-nitrobenzamide (1.06 g, 2.216 mmol) and ammonium chloride (1.778 g, 33.2 mmol) in MeOH (50 mL) cooled in an ice/water bath was added zinc (1.449 g, 22.16 mmol) and the reaction mixture was stirred at room temperature for 40 min. The mixture was filtered through celite and rinsed with methanol. The filtrate was concentrated and purified by silica gel chromatography (24 g silica gel column; 10-60% [3:1 EA:EtOH]/heptane, plus 1% NH 4 OH solution, 12 min.; 60% gradient of [3:1 EA:EtOH]/heptane, 5 min). The purest fractions were combined and concentrated. The combined fractions were concentrated and purified again (12 g silica gel column; 10-55% [3:1 EA:EtOH]/heptane, no NH 4 OH modifier, 10 min.; 55% gradient of [3:1 EA:EtOH]/heptane, 5 min.). The fractions were combined and dried to afford the title compound (571 mg, 1.09 mmol, 49.3% yield) as a brown foam. LCMS ( m/z ): 418.3 [M+H] + , ~80% purity by UV 210-350 nm).
步骤3: (E)-1-(4-(7-溴-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺Step 3: (E)-1-(4-(7-bromo-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-5-carboxamide
向(E)-4-氨基-3-((4-((2-氨基-6-溴苯基)氨基)-2,3-二甲基丁-2-烯-1-基)氨基)苯甲酰胺 (570 mg, 1.090 mmol)在冷却在冰/水浴中的DMF (10 mL)中的亮棕色溶液快速滴加1-乙基-3-甲基-1H-吡唑-5-羰基异硫氰酸酯 (~1 M 在二氧杂环己烷中, 2.18mL, 2.18 mmol)。在搅拌15 min后,加入EDC (522 mg, 2.73 mmol)和TEA (0.760 mL,5.45 mmol)并将该反应混合物温热至室温并搅拌16 h。向该搅拌的反应混合物中快速加入5:1 水:饱和NH4Cl水溶液(120 mL)的溶液。快速搅拌所得悬浮液15 min。将固体过滤并用水(3 x 20 mL)漂洗。在二乙醚 (15 mL)中搅拌该固体30 min,然后过滤并用二乙醚漂洗。在干燥之后,获得作为浅黄色固体的标题化合物(772 mg, 0.94 mmol, 86%收率)。1H NMR(DMSO-d 6 ) δ: 13.02 (br. s., 1 H), 12.90 (s, 1 H), 8.02 (s, 1 H), 7.94 (br.s., 1 H), 7.71 (d, J=8.4 Hz, 1 H), 7.61 (d, J=7.9 Hz, 1 H), 7.44 (d, J=7.9Hz, 1 H), 7.35 (br. s., 1 H), 7.26 (d, J=8.4 Hz, 1 H), 7.19 (t, J=8.0 Hz, 1H), 6.64 (s, 1 H), 6.52 (s, 1 H), 5.26 (br. s., 2 H), 5.00 (br. s., 2 H),4.53-4.63 (m, 4 H), 2.12 (s, 3 H), 2.10 (s, 3 H), 1.69 (br. s., 3 H), 1.63(br. s., 3 H), 1.35 (t, J=7.1 Hz, 3 H), 1.32 (t, J=7.1 Hz, 3 H)。LCMS (m/z):740.2/742.4 [M + H]+, 通过UV210-350 nm测得~90%纯度)。To a light brown solution of (E)-4-amino-3-((4-((2-amino-6-bromophenyl)amino)-2,3-dimethylbut-2-en-1-yl)amino)benzamide (570 mg, 1.090 mmol) in DMF (10 mL) cooled in an ice/water bath was added 1-ethyl-3-methyl-1H-pyrazole-5-carbonyl isothiocyanate (~1 M in dioxane, 2.18 mL, 2.18 mmol) dropwise rapidly. After stirring for 15 min, EDC (522 mg, 2.73 mmol) and TEA (0.760 mL, 5.45 mmol) were added and the reaction mixture was warmed to room temperature and stirred for 16 h. To the stirred reaction mixture was added a solution of 5:1 water:saturated aqueous NH 4 Cl (120 mL). The resulting suspension was stirred rapidly for 15 min. The solid was filtered and rinsed with water (3 x 20 mL). The solid was stirred in diethyl ether (15 mL) for 30 min, then filtered and rinsed with diethyl ether. After drying, the title compound (772 mg, 0.94 mmol, 86% yield) was obtained as a light yellow solid. 1 H NMR(DMSO- d 6 ) δ: 13.02 (br. s., 1 H), 12.90 (s, 1 H), 8.02 (s, 1 H), 7.94 (br.s., 1 H), 7.71 (d, J =8.4 Hz, 1 H), 7.61 (d, J =7.9 Hz, 1 H), 7. 44 (d, J =7.9Hz, 1 H), 7.35 (br. s., 1 H), 7.26 (d, J =8.4 Hz, 1 H), 7.19 (t, J =8.0 Hz, 1H), 6.64 (s, 1 H), 6.52 (s, 1 H), 5.26 (br. s., 2 H), 5.00 (br. s., 2 H), 4.53-4.63 (m, 4 H), 2.12 (s, 3 H), 2.10 (s, 3 H), 1.69 (br. s., 3 H), 1.63 (br. s., 3 H), 1.35 (t, J =7.1 Hz, 3 H), 1.32 (t, J =7.1 Hz, 3 H). LCMS ( m/z ): 740.2/742.4 [M+H] + , ~90% purity by UV210-350 nm).
步骤4: (E)-1-((E)-4-((E)-7-溴-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 4: (E)-1-((E)-4-((E)-7-bromo-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向(E)-1-(4-(7-溴-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 (760 mg, 0.923 mmol)在DMF (15 mL)中的溶液加入碳酸钾 (319 mg,2.309 mmol)和碘甲烷 (0.13 mL, 2.12 mmol)。在室温搅拌该反应混合物3 h。用水(30mL)稀释该反应混合物并形成结块固体。然后,在DCM和水之间分配该混合物。然后,用盐水洗涤有机相并浓缩。通过硅胶层析法的纯化(24 g硅胶柱; 10-60% 梯度的[3:1 EA:EtOH]/庚烷, 加1% NH4OH, 15 min.; 60% [3:1 EA:EtOH]/庚烷, 加1% NH4OH, 10 min.)提供在溶剂蒸发后作为浅橙色泡沫的标题化合物(386 mg, 0.477 mmol, 51.7%收率)。1H NMR(DMSO-d 6 ) δ: 8.08-8.12 (m, 1 H), 8.01 (br. s., 1 H), 7.80 (dd, J=8.4, 1.4 Hz,1 H), 7.62-7.67 (m, 1 H), 7.49-7.53 (m, 1 H), 7.43 (br. s., 1 H), 7.25-7.34(m, 2 H), 6.49 (s, 1 H), 6.44 (s, 1 H), 5.16 (s, 2 H), 4.89 (s, 2 H), 4.47-4.57 (m, 4 H), 3.59 (s, 3 H), 3.56 (s, 3 H), 2.13 (s, 3 H ), 2.12 (s, 3 H),1.59 (s, 3 H), 1.50 (s, 3 H), 1.24-1.33 (m, 6 H)。LCMS (m/z): 768.5/770.5 [M +H]+。To a solution of (E)-1-(4-(7-bromo-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-1-yl)-2,3-dimethylbut-2-ene-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxamide (760 mg, 0.923 mmol) in DMF (15 mL) was added potassium carbonate (319 mg, 2.309 mmol) and iodomethane (0.13 mL, 2.12 mmol). The reaction mixture was stirred at room temperature for 3 h. The reaction mixture was diluted with water (30 mL) and a clumping solid was formed. The mixture was then partitioned between DCM and water. The organic phase was then washed with brine and concentrated. Purification by silica gel chromatography (24 g silica gel column; 10-60% gradient of [3:1 EA:EtOH]/heptane, plus 1% NH4OH , 15 min.; 60% [3:1 EA:EtOH]/heptane, plus 1% NH4OH , 10 min.) provided the title compound (386 mg, 0.477 mmol, 51.7% yield) as a light orange foam after evaporation of the solvent. 1 H NMR(DMSO- d 6 ) δ: 8.08-8.12 (m, 1 H), 8.01 (br. s., 1 H), 7.80 (dd, J =8.4, 1.4 Hz, 1 H), 7.62-7.67 (m, 1 H), 7.49-7.53 (m, 1 H), 7.43 (br. s ., 1 H), 7.25-7.34(m, 2 H), 6.49 (s, 1 H), 6.44 (s, 1 H), 5.16 (s, 2 H), 4.89 (s, 2 H), 4.47-4.57 (m, 4 H), 3.59 (s, 3 H), 3.56 (s, 3 H), 2. 13(s,3H), 2.12 (s, 3 H), 1.59 (s, 3 H), 1.50 (s, 3 H), 1.24-1.33 (m, 6 H). LCMS ( m/z ): 768.5/770.5 [M +H] + .
实施例91Embodiment 91
(E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺(E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-5-carboxamide
步骤1: (E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 1: (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向(E)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-(3-吗啉代丙氧基)-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-7-甲氧基-1H-苯并[d]咪唑-5-甲酰胺 (125 mg, 0.147 mmol, 其能够根据PCT 国际申请(Int. Appl.)WO 2017175147的实施例14所述的制备例制备)和碳酸钾 (44.7 mg,0.324 mmol)在DMF (1.4 mL)中的悬浮液加入碘甲烷 (0.019 mL, 0.31 mmol)在DMF (0.4mL)中的溶液。在室温搅拌该混合物18 h,并然后用水稀释。用二氯甲烷(3X)萃取该混合物。将合并的有机层用水洗涤、用硫酸钠干燥、过滤并浓缩。通过反相质量导向的制备型HPLC(XSELECT CSH C18, 5 um填充物, 150x30 mm柱, 15-55%梯度的ACN/水,含0.1% TFA 改性剂)纯化残余物。使含所需产物的级分通过碱性PL-HCO3 MP SPE 筒。浓缩洗脱液以提供(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (45 mg, 0.051 mmol, 34.8%收率)。1H NMR (DMSO-d6) δ ppm 8.04 (br. s., 2H),7.71-7.75 (m, 2H), 7.38-7.48 (m, 4H), 6.40 (s, 1H), 6.35 (s, 1H), 5.66-5.80(m, 2H), 4.79-4.89 (m, 4H), 4.38-4.49 (m, 4H), 4.02 (br. t., J=6.3 Hz, 2H),3.74 (s, 3H), 3.47-3.54 (m, 10H), 2.18-2.29 (m, 6H), 2.12 (s, 3H), 2.10 (s,3H), 1.64-1.72 (m, 2H), 1.18-1.25 (m, 6H)。LCMS (m/z): 878.3 [M + H]+。To a suspension of (E)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-(3-morpholinopropoxy)-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-7-methoxy-1H-benzo[d]imidazole-5-carboxamide (125 mg, 0.147 mmol, which can be prepared according to the preparation described in Example 14 of PCT International Application (Int. Appl.) WO 2017175147) and potassium carbonate (44.7 mg, 0.324 mmol) in DMF (1.4 mL) was added a solution of iodomethane (0.019 mL, 0.31 mmol) in DMF (0.4 mL). The mixture was stirred at room temperature for 18 h, and then diluted with water. The mixture was extracted with dichloromethane (3X). The combined organic layer was washed with water, dried over sodium sulfate, filtered and concentrated. The residue was purified by reverse phase mass-guided preparative HPLC (XSELECT CSH C18, 5 um filler, 150x30 mm column, 15-55% gradient of ACN/ water, containing 0.1% TFA modifier). The fraction containing the desired product was passed through alkaline PL-HCO3 MP SPE cartridge. The eluent was concentrated to afford (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (45 mg, 0.051 mmol, 34.8% yield). 1 H NMR (DMSO-d 6 ) δ ppm 8.04 (br. s., 2H), 7.71-7.75 (m, 2H), 7.38-7.48 (m, 4H), 6.40 (s, 1H), 6.35 (s, 1H), 5.66-5.80 (m, 2H), 4.79-4.89 (m, 4H), 4.38-4.49 (m, 4H), 4.02 (br. t., J=6.3 Hz, 2H), 3.74 (s, 3H), 3.47-3.54 (m, 10H), 2.18-2.29 (m, 6H), 2.12 (s, 3H), 2.10 (s, 3H) , 1.64-1.72 (m, 2H), 1.18-1.25 (m, 6H). LCMS ( m/z ): 878.3 [M + H] + .
步骤2: (E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺Step 2: (E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide
向(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-1-基)丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (26 mg, 0.030 mmol)和NMO (5.20 mg, 0.044 mmol)在叔丁醇 (1.2 mL)和水(0.3 mL)中的混合物加入在叔丁醇中的2.5%四氧化锇 (0.019 mL, 1.5 µmol)。在室温搅拌该混合物 64 h ,并然后过滤。通过质量导向的制备型HPLC (XSELECT CSH C18, 5 um填充物, 150x30 mm柱, 15-55%梯度的ACN/水,含0.1% TFA 改性剂)直接纯化滤液。使含标题化合物的级分通过PL-HCO3 MP SPE 筒。浓缩洗脱液以提供(E)-1-(4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-7-(3-吗啉代丙氧基)-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二羟基丁基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-甲氧基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (11 mg, 0.012 mmol,40.7%收率)。1H NMR (400 MHz, DMSO-d 6) δ ppm 1.24 - 1.29 (m, 6 H) 1.81 - 1.86(m, 2 H) 2.09 - 2.12 (m, 6 H) 2.27 (br. s., 4 H) 2.35 - 2.39 (m, 2 H) 3.47 -3.53 (m, 10 H) 3.81 (s, 3 H) 3.82 - 3.93 (m, 2 H) 4.14 (t, J=6.40 Hz, 2 H)4.26 - 4.33 (m, 2 H) 4.44 - 4.58 (m, 6 H) 4.98 (d, J=6.53 Hz, 1 H) 5.07 (d, J=6.02 Hz, 1 H) 6.39 (s, 1 H) 6.44 (s, 1 H) 7.41 - 7.48 (m, 4 H) 7.72 (dd, J=4.52, 1.00 Hz, 2 H) 8.05 (br. s., 2 H)。LCMS (m/z): 912.2 [M + H]+。To a mixture of (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)but-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (26 mg, 0.030 mmol) and NMO (5.20 mg, 0.044 mmol) in tert-butanol (1.2 mL) and water (0.3 mL) was added 2.5% osmium tetroxide in tert-butanol (0.019 mL, 1.5 µmol). The mixture was stirred at room temperature for 64 h and then filtered. The filtrate was directly purified by mass-directed preparative HPLC (XSELECT CSH C18, 5 um packing, 150x30 mm column, 15-55% gradient of ACN/water with 0.1% TFA modifier). The fractions containing the title compound were passed through a PL-HCO3 MP SPE cartridge. The eluent was concentrated to afford (E)-1-(4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-7-(3-morpholinopropoxy)-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dihydroxybutyl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-methoxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (11 mg, 0.012 mmol, 40.7% yield). 1H NMR (400 MHz, DMSO- d 6 ) δ ppm 1.24 - 1.29 (m, 6 H) 1.81 - 1.86 (m, 2 H) 2.09 - 2.12 (m, 6 H) 2.27 (br. s., 4 H) 2.35 - 2.39 (m, 2 H) 3.47 -3.53 ( m, 10 H) 3.81 (s, 3 H) 3.82 - 3.93 (m, 2 H) 4.14 (t, J =6.40 Hz, 2 H)4.26 - 4.33 (m, 2 H) 4.44 - 4.58 (m, 6 H) 4.98 (d, J =6.53 Hz, 1 H) 5.07 (d, J =6.02 Hz, 1 H) 6.39 (s, 1 H) 6.44 (s, 1 H) 7.41 - 7.48 (m, 4 H) 7.72 (dd, J =4.52, 1.00 Hz, 2 H) 8.05 (br. s., 2 H). LCMS ( m/z ): 912.2 [M + H] + .
实施例92Embodiment 92
(E)-7-(3-氨基丙氧基)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 甲酸盐(E)-7-(3-aminopropoxy)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, formate
在90℃加热(E)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-7-羟基-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺 (384 mg, 0.513 mmol)、(3-溴丙基)氨基甲酸叔丁酯 (733 mg, 3.08 mmol)和K2CO3 (425 mg, 3.08 mmol)在DMF (10 mL)中的混合物9天。LCMS分析表明向O-烷基化中间体的部分转化随时间增加以在9天达到约50%转化率。将该反应混合物过滤并浓缩。通过质量导向的制备型HPLC (XSELECT CSH C18, 5 um填充物, 150x30 mm柱, 15-55%梯度的ACN/水,含0.1% TFA 改性剂)纯化残余物以在溶剂蒸发后提供100 mg棕色残余物。该残余物为N-B℃ 中间体和标题化合物(在溶剂蒸发期间发生B℃基团的去除)的混合物。使残余物溶解在甲醇 (1 mL)和1,4-二氧杂环己烷 (2 mL)中并声处理成棕色溶液,然后加入HCl(4 M 在二氧杂环己烷中, 1.282 mL, 5.13 mmol)。再次声处理该混合物并在室温搅拌该混合物3 h。将该反应混合物浓缩、溶解在DMSO中,然后通过质量导向的制备型HPLC(XSELECT CSH C18, 5 um填充物, 150x30 mm柱, 15-55%梯度的ACN/水,含0.1%甲酸改性剂)纯化以提供作为白色固体的(E)-7-(3-氨基丙氧基)-1-((E)-4-((E)-5-氨基甲酰基-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-1-基)-2,3-二甲基丁-2-烯-1-基)-2-((1-乙基-3-甲基-1H-吡唑-5-羰基)亚氨基)-3-甲基-2,3-二氢-1H-苯并[d]咪唑-5-甲酰胺, 甲酸盐 (8.0 mg, 9.4 µmol, 1.8%收率)。1H NMR (400MHz, DMSO-d 6 ) δ ppm 8.36 (s, 1 H), 8.12 (s, 2 H), 8.05 (br. s., 1 H), 7.76-7.84 (m, 2 H), 7.53 (s, 1 H), 7.47 (s, 1 H), 7.44 (s, 1 H), 7.25 (d, J =8.5Hz, 1 H), 6.47 (s, 1 H), 6.32 (s, 1 H), 5.06 (s, 2 H), 4.85 (s, 2 H), 4.41-4.57 (m, 4 H), 4.22 (t, J =6.4 Hz, 2 H), 3.59 (s, 3 H), 3.56 (s, 3 H), 2.77(t, J =6.9 Hz, 2 H), 2.12 (s, 3 H), 2.06 (s, 3 H), 1.84 (五重峰, J =6.5 Hz, 2H), 1.63 (s, 3 H), 1.49 (s, 3 H), 1.29 (t, J =7.1 Hz, 3 H), 1.24 (t, J =7.1Hz, 3 H)。LCMS (m/z): 806.3 [M + H]+。A mixture of (E)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-7-hydroxy-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide (384 mg, 0.513 mmol), tert-butyl (3-bromopropyl)carbamate (733 mg, 3.08 mmol) and K2CO3 (425 mg, 3.08 mmol) in DMF (10 mL ) was heated at 90 °C for 9 days. LCMS analysis showed that partial conversion to the O-alkylated intermediate increased over time to reach about 50% conversion in 9 days. The reaction mixture was filtered and concentrated. The residue was purified by mass-directed preparative HPLC (XSELECT CSH C18, 5 um packing, 150x30 mm column, 15-55% gradient of ACN/water, containing 0.1% TFA modifier) to provide 100 mg of brown residue after solvent evaporation. The residue was a mixture of NB°C intermediate and the title compound (removal of the B°C group occurred during solvent evaporation). The residue was dissolved in methanol (1 mL) and 1,4-dioxane (2 mL) and sonicated to a brown solution, and then HCl (4 M in dioxane, 1.282 mL, 5.13 mmol) was added. The mixture was sonicated again and stirred at room temperature for 3 h. The reaction mixture was concentrated, dissolved in DMSO, and then purified by mass-directed preparative HPLC (XSELECT CSH C18, 5 um packing, 150x30 mm column, 15-55% gradient of ACN/water with 0.1% formic acid modifier) to afford (E)-7-(3-aminopropoxy)-1-((E)-4-((E)-5-carbamoyl-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazol-1-yl)-2,3-dimethylbut-2-en-1-yl)-2-((1-ethyl-3-methyl-1H-pyrazole-5-carbonyl)imino)-3-methyl-2,3-dihydro-1H-benzo[d]imidazole-5-carboxamide, formate salt as a white solid (8.0 mg, 9.4 µmol, 1.8% yield). 1 H NMR (400MHz, DMSO- d 6 ) δ ppm 8.36 (s, 1 H), 8.12 (s, 2 H), 8.05 (br. s., 1 H), 7.76-7.84 (m, 2 H), 7.53 (s, 1 H), 7.47 (s, 1 H), 7.44 (s, 1 H), 7.25 (d, J =8.5Hz, 1 H), 6.47 (s, 1 H), 6.32 (s, 1 H), 5.06 (s, 2 H), 4.85 (s, 2 H), 4.41-4.57 (m, 4 H), 4.22 (t, J =6.4 Hz, 2 H), 3.59 (s, 3 H), 3.56 (s, 3 H), 2.77(t, J =6.9 Hz, 2 H), 2.12 (s, 3 H), 2.06 (s, 3 H), 1.84 (quint, J =6.5 Hz, 2H), 1.63 (s, 3 H), 1.49 (s, 3 H), 1.29 (t, J =7.1 Hz, 3 H), 1.24 ( t, J =7.1Hz, 3H). LCMS ( m/z ): 806.3 [M + H] + .
表3示出实施例93-109,其可以根据如下示例的方法制备:Table 3 shows Examples 93-109, which can be prepared according to the following exemplified method:
。 .
AlexaFluor-488 FRET测定配体AlexaFluor-488 FRET Assay Ligand
3',6'-二氨基-5-((2-(1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺基)乙基)氨基甲酰基)-3-氧代-3H-螺[异苯并呋喃-1,9'-呫吨]-4',5'-二磺酸3',6'-Diamino-5-((2-(1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-5-carboxamido)ethyl)carbamoyl)-3-oxo-3H-spiro[isobenzofuran-1,9'-xanthene]-4',5'-disulfonic acid
1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酸 二盐酸盐1-(4-(5-Carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxylic acid dihydrochloride
在室温向在THF (3.47 mL)、MeOH (3.47 mL)和水(1.74 mL)中的1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酸甲酯 双三氟乙酸盐 (400 mg,0.434 mmol, 在PCT公开No. WO 2017175147中所述的实施例23)加入8 M 氢氧化钾 (1.09mL, 8.68 mmol)。在搅拌过夜后,浓缩该反应并加入水。用7 N aq HCl将该混合物酸化至pH4-5,并通过过滤收集所得灰色固体以得到标题化合物(335 mg, 0.423 mmol, 97 %收率)。1H NMR (400 MHz, DMSO-d 6 ) δ ppm 12.82 - 12.95 (m, 3 H), 8.08 (s, 1 H), 7.99(br. s., 2 H), 7.83 (d, J=8.34 Hz, 1 H), 7.78 (d, J=8.34 Hz, 1 H), 7.58 (t, J=7.33 Hz, 2 H), 7.36 (br. s., 1 H), 6.60 (d, J=4.80 Hz, 2 H), 4.58 (d, J=6.57Hz, 4 H), 4.29 (br. s., 4 H,) 2.10 (s, 6 H), 1.88 (br. s., 4 H), 1.31 (t, J=6.95 Hz, 6 H); LCMS: Rt = 0.83 min, [M+H]+ = 680.5。To 1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxylic acid methyl ester bis trifluoroacetate (400 mg, 0.434 mmol, Example 23 described in PCT Publication No. WO 2017175147) in THF (3.47 mL), MeOH (3.47 mL), and water (1.74 mL) was added 8 M potassium hydroxide (1.09 mL, 8.68 mmol) at room temperature. After stirring overnight, the reaction was concentrated and water was added. The mixture was acidified to pH 4-5 with 7 N aq HCl, and the resulting grey solid was collected by filtration to give the title compound (335 mg, 0.423 mmol, 97 % yield). 1 H NMR (400 MHz, DMSO- d 6 ) δ ppm 12.82 - 12.95 (m, 3 H), 8.08 (s, 1 H), 7.99 (br. s., 2 H), 7.83 (d, J =8.34 Hz, 1 H), 7.78 (d, J =8.34 Hz, 1 H), 7.5 8 (t, J =7.33 Hz, 2 H), 7.36 (br. s., 1 H), 6.60 (d, J =4.80 Hz, 2 H), 4.58 (d, J =6.57Hz, 4 H), 4.29 (br. s., 4 H,) 2.10 (s, 6 H), 1.88 (br. s., 4 H) , 1.31 (t, J =6.95 Hz, 6 H); LCMS: Rt = 0.83 min, [M+H] + = 680.5.
步骤1: N-(2-氨基乙基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 三氟乙酸盐Step 1: N- (2-aminoethyl)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-5-carboxamide trifluoroacetate
在37℃将1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酸 (10 mg, 0.015 mmol)溶解(采用声处理)在DMSO (300 μL)中。向其中加入氨基甲酸(9H-芴-9-基)甲酯 (2-氨基乙基)酯 盐酸盐 (6.9 mg, 0.022 mmol)和HATU (7.6 mg,0.020 mmol)在DMSO (100 μL)中的溶液,然后加入DIEA (10 μL, 0.057 mmol)。在搅拌过夜后,用DMF (600 µL)稀释该反应,加入4-甲基哌啶(400 µL),和在室温搅拌该反应1 h。浓缩该混合物,并用1:1 DMSO: MeOH (<1 mL)稀释所得残余物并通过反相层析法(JupiterC18制备型柱, 10 mL/min)采用30 - 100 %在水(0.1 % TFA添加剂)中的 (9:1 ACN: 水)洗脱来纯化以得到标题化合物(8.45 mg, 10.1 µmol, 69 %收率)。LCMS: Rt = 0.62 min,[M+H]+ = 722.4。1-(4-(5-Carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazole-5-carboxylic acid (10 mg, 0.015 mmol) was dissolved (with sonication) in DMSO (300 μL) at 37° C. To this was added a solution of (9H-fluoren-9-yl)methyl (2-aminoethyl) carbamate hydrochloride (6.9 mg, 0.022 mmol) and HATU (7.6 mg, 0.020 mmol) in DMSO (100 μL), followed by DIEA (10 μL, 0.057 mmol). After stirring overnight, the reaction was diluted with DMF (600 µL), 4-methylpiperidine (400 µL) was added, and the reaction was stirred at room temperature for 1 h. The mixture was concentrated, and the residue was diluted with 1:1 DMSO: MeOH (<1 mL) and purified by reverse phase chromatography (Jupiter C18 preparative column, 10 mL/min) eluting with 30 - 100 % (9:1 ACN: water) in water (0.1 % TFA additive) to give the title compound (8.45 mg, 10.1 µmol, 69 % yield). LCMS: Rt = 0.62 min, [M+H] + = 722.4.
步骤2: 3',6'-二氨基-5-((2-(1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺基)乙基)氨基甲酰基)-3-氧代-3H-螺[异苯并呋喃-1,9'-呫吨]-4',5'-二磺酸Step 2: 3',6'-diamino-5-((2-(1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-5-carboxamido)ethyl)carbamoyl)-3-oxo-3H-spiro[isobenzofuran-1,9'-xanthene]-4',5'-disulfonic acid
将N-(2-氨基乙基)-1-(4-(5-氨基甲酰基-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-1-基)丁基)-2-(1-乙基-3-甲基-1H-吡唑-5-甲酰胺基)-1H-苯并[d]咪唑-5-甲酰胺 三氟乙酸盐 (8.45mg, 10.1 μmol)溶解在DMF (200 μl)中并加入固体(5,6-) Alexa Fluor 488-ONSu (5.00 mg, 7.92 μmol)中。商业Alexa Fluor 488-ONSu试剂为 5-和6-位异构体的混合物。 N- (2-aminoethyl)-1-(4-(5-carbamoyl-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-1-yl)butyl)-2-(1-ethyl-3-methyl-1H-pyrazole-5-carboxamido)-1H-benzo[d]imidazol-5-carboxamide trifluoroacetate (8.45 mg, 10.1 μmol) was dissolved in DMF (200 μl) and added to solid (5,6-) Alexa Fluor 488-ONSu (5.00 mg, 7.92 μmol). The commercial Alexa Fluor 488-ONSu reagent is a mixture of 5- and 6-isomers.
当产生溶液时,加入DIPEA (2 μL, 0.01 mmol),并在无光下(通过涡旋运动)搅动该混合物过夜。LCMS表明形成具有预期分子量([M+H] 1238.6)的较早和较晚洗脱出的产物峰。浓缩该反应,并将残余物溶解于1:1 DMSO: MeOH (<1 mL)中并通过反相层析法(Jupiter C18制备型柱, 10 mL/min)采用在水(0.1 % TFA添加剂)中的15 - 100 % (9:1ACN: 水)洗脱来纯化。以高纯度获得较早洗脱出的位置异构体。相比之下,较晚洗脱出的异构体的级分还含有未反应的起始原料。将这些含有不纯的较晚洗脱出的异构体的级分合并并浓缩。将这个残余物溶解在1:1 DMSO: MeOH (<1 mL)中并通过反相层析法(WatersSymmetryPrep制备型柱, 10 mL/min)采用在水(0.1 % TFA添加剂)中的15 - 100 % (9:1ACN: 水)洗脱来纯化以得到标题化合物(较晚洗脱出的异构体, 1.94 mg, 1.49 µmol, 19%收率)。LCMS: Rt = 0.69 min, [M+H]+ = 1238.6。注意到标题化合物(5-异构体)的推定结构不是基于严格的结构测定而是基于先前的观察,即通过反相HPLC方法认为5-位异构体通常为较晚洗脱出的异构体。When a solution was produced, DIPEA (2 μL, 0.01 mmol) was added and the mixture was stirred overnight in the absence of light (by vortexing). LCMS showed the formation of earlier and later eluting product peaks with the expected molecular weight ([M+H] 1238.6). The reaction was concentrated, and the residue was dissolved in 1:1 DMSO: MeOH (<1 mL) and purified by reverse phase chromatography (Jupiter C18 preparative column, 10 mL/min) using 15 - 100% (9:1 ACN: water) elution in water (0.1% TFA additive). The earlier eluting positional isomers were obtained in high purity. In contrast, the fractions of the later eluting isomers also contained unreacted starting materials. The fractions containing the impure later eluting isomers were combined and concentrated. This residue was dissolved in 1:1 DMSO: MeOH (<1 mL) and purified by reverse phase chromatography (Waters Symmetry Prep column, 10 mL/min) eluting with 15 - 100 % (9:1 ACN: water) in water (0.1 % TFA additive) to give the title compound (late eluting isomer, 1.94 mg, 1.49 µmol, 19 % yield). LCMS: Rt = 0.69 min, [M+H] + = 1238.6. Note that the proposed structure of the title compound (5-isomer) was not based on rigorous structural determination but on previous observations that the 5-isomer was generally the later eluting isomer by reverse phase HPLC methods.
生物测定和数据:Bioassays and Data:
如上所述,本发明化合物是STING的调节剂,且可用于治疗由STING介导的疾病。本发明化合物的生物活性可以使用用于确定作为STING的调节剂的化合物的活性的任何合适的测定法以及组织和体内模型来确定。As described above, the compounds of the invention are modulators of STING and can be used to treat diseases mediated by STING. The biological activity of the compounds of the invention can be determined using any suitable assays and tissue and in vivo models for determining the activity of compounds as modulators of STING.
每种化合物的pIC50值在至少一次实验中报告或报告为多次实验的平均值。应当理解,本文描述的数据可以具有合理的变化,这取决于进行实验的人使用的具体条件和程序。The pIC50 value for each compound is reported in at least one experiment or as an average of multiple experiments. It is understood that the data described herein can have reasonable variations depending on the specific conditions and procedures used by the person performing the experiments.
结合测定Binding assay
(1) SPA(1) SPA
放射性配体结合测定法被开发以通过与3H-cGAMP(氚标记的单磷酸环状鸟嘌呤(2’,5’) -单磷酸腺嘌呤(3’,5’))竞争来测量式(I)化合物和STING的羧基末端结构域(CTD)的相互作用。还参见Li等人 (Nature Chemical Biology,10, 1043-1048, (2014))。编码跨越残基149至379的人STING的序列(基因ID 340061)的蛋白质在细菌中表达,其具有与用于生物素化的AviTag™融合的羧基末端Flag®肽和用于亲和纯化的六组氨酸标记。将纯化的STING-Flag-AviTag-6Xhis蛋白质使用酶BirA来生物素化至完成(Beckett D. 等 人, Protein Science, 1999, 8:921-929)。在用净DMSO中的100-250 nL化合物预润洗(pre-stamped)的Greiner白色384-孔板(目录号784075)中,通过在含有0.02% (w/v)普郎尼克F127和0.02% (w/v)牛血清白蛋白的磷酸盐缓冲盐水缓冲液中在含有50 nM生物素化-STING、50 nM 3H-cGAMP和1.25 mg/mL链霉亲和素包被的闪烁迫近测定珠粒(PerkinElmer)的平衡结合反应中的竞争来确定式(I)化合物的相对效力。将结合反应在室温温育60分钟。测量发光(ViewLux™)并使用下式以将原始读数表达为抑制%:,其中U为未知的值,C1为被10 μM cGAMP完全抑制的平均应答和C2最大应答平均值。使用等式进行曲线拟合,其中A为最小应答,B为最大应答,C为log10 *XC50,D为斜率,和x为ABASE XE中的 log10化合物浓度[M]。在这样的条件下,阳性对照化合物cGAMP的表观抑制常数为250 nM,为其4-5nM的实际亲和力的约50倍(Zhang X等人, Molecular Cell, 2013, 51:1-10)。 A radioligand binding assay was developed to measure the interaction of the compound of formula (I) and the carboxyl terminal domain (CTD) of STING by competing with 3H-cGAMP (tritium-labeled cyclic guanine monophosphate (2', 5') -adenine monophosphate (3', 5')). See also Li et al. (Nature Chemical Biology, 10, 1043-1048, (2014) ). A protein encoding the sequence of human STING spanning residues 149 to 379 (gene ID 340061) was expressed in bacteria with a carboxyl terminal Flag® peptide fused to AviTag™ for biotinylation and a six-histidine tag for affinity purification. The purified STING-Flag-AviTag-6Xhis protein was biotinylated to completion using the enzyme BirA (Beckett D. et al., Protein Science, 1999, 8:921-929 ). The relative potency of compounds of formula (I) was determined by competition in an equilibrium binding reaction with 50 nM biotinylated-STING, 50 nM 3H-cGAMP and 1.25 mg/mL streptavidin-coated scintillation proximity assay beads (PerkinElmer) in phosphate-buffered saline buffer containing 0.02% (w/v) Pluronic F127 and 0.02% (w/v) bovine serum albumin in Greiner white 384-well plates (Cat. No. 784075) pre-stamped with 100-250 nL of compound in neat DMSO. The binding reaction was incubated at room temperature for 60 minutes. Luminescence was measured (ViewLux™) and the following formula was used to express the raw reading as % inhibition: , where U is the unknown value, C1 is the average response completely inhibited by 10 μM cGAMP and C2 is the average of the maximum responses. Using the equation Curve fitting was performed, where A is the minimum response, B is the maximum response, C is log 10 *XC50, D is the slope, and x is the log 10 compound concentration [M] in ABASE XE. Under such conditions, the apparent inhibition constant of the positive control compound cGAMP is 250 nM, which is about 50 times its actual affinity of 4-5 nM ( Zhang X et al., Molecular Cell, 2013, 51: 1-10).
使用上文所述的SPA测定,测试实施例1-5和7-9、11-14、16和19的化合物并表现出7至超出7.4的上限测定值的范围的pIC50值。Compounds of Examples 1-5 and 7-9, 11-14, 16 and 19 were tested using the SPA assay described above and exhibited pIC50 values ranging from 7 to above the upper assay value of 7.4.
(2) FRET测定(2) FRET assay
使用竞争结合测定法确定分子与人STING的C-末端结构域(CTD)的结合效力。在该测定中,采用具有C-末端生物素化的Avi-tag的STING (149-379)重组蛋白。当与STING结合时,Alexa488标记的正构位点探针(orthosteric site probe) (参见针对FRET测定配体的合成的第347-350页)接受来自Tb-链霉亲和素-Avi-STING的490 nm发射,并且在520 nm处测量荧光的增加。竞争探针结合位点的分子将导致低520nm信号。该测定在含有纯DMSO中的100nL化合物的Greiner黑色384孔板(目录号784076)中运行。将500pM STING、500pM链霉亲和素-Lumi4-Tb和100nM Alexa488探针于含有0.02% (w/v)普郎尼克F127和0.02% (w/v)牛血清白蛋白的磷酸盐缓冲盐水中的溶液使用Combi液体处理器(ThermoFisher)添加至板中。将板以500rpm离心1分钟,在室温温育15分钟,且然后测量Envision读板器(Perkin-Elmer)上337nm激光激发后520nm处的荧光发射。使用上文所述ABASE XE中的标准四参数曲线拟合确定pIC50值。The binding efficacy of the molecule to the C-terminal domain (CTD) of human STING was determined using a competitive binding assay. In this assay, a STING (149-379) recombinant protein with a C-terminal biotinylated Avi-tag was used. When bound to STING, an Alexa488-labeled orthosteric site probe (see pages 347-350 for the synthesis of FRET assay ligands) receives 490 nm emission from Tb-streptavidin-Avi-STING and measures the increase in fluorescence at 520 nm. Molecules competing for the probe binding site will result in a low 520 nm signal. The assay was run in a Greiner black 384-well plate (catalog number 784076) containing 100 nL of compound in pure DMSO. A solution of 500 pM STING, 500 pM Streptavidin-Lumi4-Tb, and 100 nM Alexa488 probe in phosphate buffered saline containing 0.02% (w/v) Pluronic F127 and 0.02% (w/v) bovine serum albumin was added to the plate using a Combi liquid handler (ThermoFisher). The plate was centrifuged at 500 rpm for 1 minute, incubated at room temperature for 15 minutes, and then the fluorescence emission at 520 nm after 337 nm laser excitation was measured on an Envision plate reader (Perkin-Elmer). pIC 50 values were determined using a standard four-parameter curve fit in ABASE XE as described above.
使用上文所述的FRET测定来测试实施例1-42、44-89和91-108并表现出5.0至超出9.9的上限测定值的范围的pIC50值。Examples 1-42, 44-89, and 91-108 were tested using the FRET assay described above and exhibited pIC50 values ranging from 5.0 to above the upper assay value of 9.9.
例如,以下实施例的FRET测定的pIC50为:For example, the pIC50 of the FRET assay in the following example is:
。 .
细胞功能测定Cell function assay
式(I)化合物的功能可以在检测STING特异性活化和/或IFNβ蛋白分泌的抑制的细胞测定法中确定。The function of the compounds of formula (I) can be determined in a cellular assay detecting specific activation of STING and/or inhibition of IFNβ protein secretion.
(1) 功能测定I (PBMC拮抗剂测定):式(I)化合物对STING的抑制可以通过测量由采用EC80浓度的STING激动剂(PCT公开No. WO 2017175147中描述的实施例167)或77 nMBacmam病毒(一种双链DNA病毒)刺激然后采用不同剂量的式(I)化合物处理的外周血液单核细胞(PBMC)分泌的干扰素β的损失来确定。将冷冻的PBMC细胞解冻并在培养基(RPMI-1640,其含有1.5 g/L NaHCO3、4.5 g/L葡萄糖、10 mM Hepes和1 mM丙酮酸钠、10% FBS)中稀释至6X105个细胞/mL的最终浓度。将PBMC-细胞悬浮液以每孔15,000个细胞的密度分配至含有250 nL稀释于DMSO中的化合物的384孔组织培养板(Griener 781073)中。在加入STING激动剂之前温育该PBMC板30分钟。分泌至生长培养基中的IFNβ蛋白的水平在37℃温育4小时之后使用人IFNβ电化学发光试剂盒(Meso Scale Diagnostics)遵循制造商的说明书采用STING激动剂来测量。抑制百分比相对于缺乏化合物处理或EC80的STING激动剂的对照来确定,并绘制为化合物浓度的函数,以使用受体-配体抑制的标准双态模型确定pIC50。(1) Functional Assay I (PBMC Antagonist Assay): Inhibition of STING by the compound of formula (I) can be determined by measuring the loss of interferon β secreted by peripheral blood mononuclear cells (PBMCs) stimulated with an EC80 concentration of a STING agonist (Example 167 described in PCT Publication No. WO 2017175147) or 77 nM Bacmam virus (a double-stranded DNA virus) and then treated with different doses of the compound of formula (I). Frozen PBMC cells were thawed and diluted to a final concentration of 6×10 5 cells/mL in culture medium (RPMI-1640 containing 1.5 g/L NaHCO 3 , 4.5 g/L glucose, 10 mM Hepes and 1 mM sodium pyruvate, 10% FBS). PBMC-cell suspensions were distributed at a density of 15,000 cells per well into 384-well tissue culture plates (Griener 781073) containing 250 nL of compound diluted in DMSO. The PBMC plates were incubated for 30 minutes before the addition of STING agonists. The levels of IFNβ protein secreted into the growth medium were measured using a human IFNβ electrochemiluminescence kit (Meso Scale Diagnostics) with STING agonists following the manufacturer's instructions after incubation for 4 hours at 37°C. The percentage of inhibition was determined relative to a control lacking compound treatment or EC80 of STING agonist and plotted as a function of compound concentration to determine pIC50 using the standard two-state model of receptor-ligand inhibition.
使用上文所述的功能测定I (PBMC拮抗剂测定),测试实施例1-4、12、13、18-20、23-25、27、29-34、36、38、40、41、45、47、48、50、53、54、56-60、63、65-67、69-72、74、76-89、91和93-105。实施例2、3、13、18- 20、23-25、27、29-34、38、40、41、45、47、48、50、53、54、56-60、63、65-67、69-72、74、76-89、91和93-105表现出4.3至超出8.1的上限测定值的范围的pIC50值。Examples 1-4, 12, 13, 18-20, 23-25, 27, 29-34, 36, 38, 40, 41, 45, 47, 48, 50, 53, 54, 56-60, 63, 65-67, 69-72, 74, 76-89, 91, and 93-105 were tested using Functional Assay I (PBMC Antagonist Assay) described above. Examples 2, 3, 13, 18-20, 23-25, 27, 29-34, 38, 40, 41, 45, 47, 48, 50, 53, 54, 56-60, 63, 65-67, 69-72, 74, 76-89, 91, and 93-105 exhibited pIC50 values ranging from 4.3 to above the upper assay value of 8.1 .
例如,以下实施例的PBMC拮抗剂测定的pIC50为:For example, the pIC50 of the PBMC antagonist assay in the following example is:
。 .
(2) 功能测定II (PBMC激动剂测定):式I化合物对STING的活化通过测量用不同剂量的式(I)化合物处理的人外周血单核细胞(PBMC)分泌的IFNβ的水平来确定。将冷冻的PBMC细胞解冻,重悬浮于培养基(RPMI-1640,其含有1.5 g/L NaHCO3、4.5 g/L葡萄糖、10mM Hepes和1 mM丙酮酸钠、10% FBS、10 ng/mL脂多糖)中至3X105个细胞/mL的最终浓度,并以每孔15,000个细胞的密度分配至含有250 nL稀释于DMSO中的化合物的384孔组织培养板(Griener 781073)中。分泌至生长培养基中的IFNβ蛋白的水平在37℃温育4小时之后使用人IFNβ电化学发光试剂盒(Meso Scale Diagnostics)遵循制造商的说明书来测量。活化百分比相对于对照DMSO处理来确定,并绘制为化合物浓度的函数,以使用受体活化的标准模型确定pEC50。(2) Functional assay II (PBMC agonist assay): The activation of STING by the compounds of formula I was determined by measuring the level of IFNβ secreted by human peripheral blood mononuclear cells (PBMCs) treated with different doses of the compounds of formula (I). Frozen PBMC cells were thawed, resuspended in culture medium (RPMI-1640 containing 1.5 g/L NaHCO 3 , 4.5 g/L glucose, 10 mM Hepes and 1 mM sodium pyruvate, 10% FBS, 10 ng/mL lipopolysaccharide) to a final concentration of 3×10 5 cells/mL, and plated at a density of 15,000 cells per well into 384-well tissue culture plates (Griener 781073) containing 250 nL of the compound diluted in DMSO. The level of IFNβ protein secreted into the growth medium was measured after incubation at 37° C. for 4 hours using a human IFNβ electrochemiluminescence kit (Meso Scale Diagnostics) following the manufacturer's instructions. Percent activation was determined relative to control DMSO treatment and plotted as a function of compound concentration to determine the pEC50 using standard models of receptor activation.
使用上文所述的功能测定II (PBMC激动剂测定),测试实施例1-4、12、13、18-20、23-25、27、29-34、36、38、40、41、45、47、48、50、53、54、56-60、63、65- 67、69-72、74、76-89、91、93和94 -105。实施例1-4、12、13、81和88表现出4.3至7.3的范围的pEC50值。所有其它被测试化合物都表现出低于4.3的pEC50。Examples 1-4, 12, 13, 18-20, 23-25, 27, 29-34, 36, 38, 40, 41, 45, 47, 48, 50, 53, 54, 56-60, 63, 65-67, 69-72, 74, 76-89, 91, 93 and 94-105 were tested using the Functional Assay II (PBMC Agonist Assay) described above. Examples 1-4, 12, 13, 81 and 88 exhibited pEC50 values ranging from 4.3 to 7.3. All other tested compounds exhibited pEC50s below 4.3.
(3) 功能测定III (HEK WT激动剂测定):细胞中STING的活化可以使用荧光素酶报告基因测定法在人胚肾细胞(HEK293T)中确定,所述人胚肾细胞(HEK293T)用表达STING和由干扰素刺激的反应元件启动子(pISRE-Luc)驱动的酶萤火虫荧光素酶的质粒(AgilentTechnologies)共转染。将全长人STING(基因ID 340061)和全长人环状鸟嘌呤腺嘌呤合成酶(cGAS)(参考序列NM_138441.2)克隆至含有巨细胞病毒启动子的哺乳动物细胞表达载体中。使用细胞悬浮液用Fugene® 6遵循制造商的说明(3:1 Fugene®:DNA)来制备转染。将五十微升的转染悬浮液分配至含有250 nL式(I)化合物的384孔板的孔中。最终孔组合物含有20,000个细胞/孔、1 ng STING、20 ng pISRE-Luc和空载体pcDNA3.1(Invitrogen)以使总DNA浓度达到125 ng。预期生成STING的最大活化的对照孔用cGAS表达质粒共转染。将板密封并在37℃温育24小时。萤火虫荧光素酶的表达使用Steady-Glo®荧光素酶测定系统(Promega)处理,并使用标准实验室发光读板器进行分析。将数据针对在cGAS存在的情况下的发光应答归一化,绘制为化合物浓度的函数,并使用受体活化的标准模型进行拟合以推导出pEC50。(3) Functional assay III (HEK WT agonist assay): Activation of STING in cells can be determined using a luciferase reporter gene assay in human embryonic kidney cells (HEK293T) co-transfected with a plasmid (Agilent Technologies) expressing STING and the enzyme firefly luciferase driven by the interferon-stimulated response element promoter (pISRE-Luc). Full-length human STING (gene ID 340061) and full-length human cyclic guanine adenine synthetase (cGAS) (reference sequence NM_138441.2) were cloned into a mammalian cell expression vector containing a cytomegalovirus promoter. Transfection was prepared using a cell suspension with Fugene® 6 following the manufacturer's instructions (3:1 Fugene® : DNA). Fifty microliters of the transfection suspension were dispensed into the wells of a 384-well plate containing 250 nL of the compound of formula (I). The final well composition contained 20,000 cells/well, 1 ng STING, 20 ng pISRE-Luc and empty vector pcDNA3.1 (Invitrogen) to bring the total DNA concentration to 125 ng. Control wells expected to generate maximum activation of STING were co-transfected with cGAS expression plasmid. The plates were sealed and incubated at 37°C for 24 hours. Expression of firefly luciferase was processed using the Steady- Glo® luciferase assay system (Promega) and analyzed using a standard laboratory luminescence plate reader. The data were normalized for the luminescence response in the presence of cGAS, plotted as a function of compound concentration, and fitted using a standard model of receptor activation to derive pEC 50 .
使用上文所述的功能测定III (HEK WT激动剂测定),测试实施例1-9、11-22、24-28、35、36、39、45-52和72-74。实施例1-9和11-16、24、26-28、35、39、48、49和73表现出 5.1至超出8.1的上限测定值的范围的pEC50值。最大应答从对照孔的5至139%而变化。Examples 1-9, 11-22, 24-28, 35, 36, 39, 45-52, and 72-74 were tested using the functional assay III (HEK WT agonist assay) described above. Examples 1-9 and 11-16, 24, 26-28, 35, 39, 48, 49, and 73 exhibited pEC 50 values ranging from 5.1 to above the upper assay value of 8.1. The maximum response varied from 5 to 139% of the control wells.
(4) 功能测定IV (THP-1拮抗剂测定)(4) Functional assay IV (THP-1 antagonist assay)
通过测量由采用含dsDNA的杆状病毒(Bacmam病毒) 刺激的人PBMC或不灭的THP-1细胞分泌的干扰素β的损失来确定式(I)化合物对STING的抑制。将以1X105个细胞/孔的密度铺板在圆底96-孔板中的在培养基(RPMI-1640,其含有1.5 g/L NaHCO3、4.5 g/L葡萄糖、10 mM Hepes和1 mM丙酮酸钠、10 % FBS、1% PSF、50 uM β-MeOH)中的THP-1细胞与不同浓度的STING拮抗剂一起温育60分钟,然后加入Bacmam病毒(40 pfu/细胞的最终MOI)。分泌至生长培养基中的IFNβ蛋白的水平在37℃温育6和20小时之后使用人IFNβ电化学发光试剂盒(Meso Scale Diagnostics)遵循制造商的说明书来测量。将IFNβ (pg/ml)水平转化成相对于缺乏化合物处理(对照1)或Bacmam病毒感染(对照2)的对照的百分比抑制,并使用S型四参数最小二乘法拟合模型进行拟合以将所报告的化合物效力定义为pIC50。100 x (1-(样品孔-(对照2)/ (对照1-对照2)。The inhibition of STING by the compounds of formula (I) was determined by measuring the loss of interferon β secreted by human PBMCs or immortalized THP-1 cells stimulated with dsDNA-containing baculovirus (Bacmam virus). THP-1 cells plated at a density of 1×10 5 cells/well in a round-bottom 96-well plate in culture medium (RPMI-1640, which contains 1.5 g/L NaHCO 3 , 4.5 g/L glucose, 10 mM Hepes and 1 mM sodium pyruvate, 10% FBS, 1% PSF, 50 uM β-MeOH) were incubated with different concentrations of STING antagonists for 60 minutes before adding Bacmam virus (final MOI of 40 pfu/cell). The level of IFNβ protein secreted into the growth medium was measured after 6 and 20 hours of incubation at 37°C using a human IFNβ electrochemiluminescence kit (Meso Scale Diagnostics) following the manufacturer's instructions. IFNβ (pg/ml) levels were converted to percent inhibition relative to controls lacking compound treatment (Control 1) or infected with Bacmam virus (Control 2) and fitted using a sigmoid four-parameter least squares fitting model to define the reported compound potency as pIC50. 100 x (1-(sample well-(Control 2)/(Control 1-Control 2).
使用上文所述的功能测定IV (THP-1拮抗剂测定),测试实施例1、3-5、7-9、12-14、16-20、22、24-26、27、29、30、35-41、44、45-56、59、60和62-77。实施例1、3、4、7-9、12、13、16-20、22、24-27、29、30、35、37-39、41、44-56、60、62-75和77表现出4.3至超出9.1的上限测定值的pIC50值。Examples 1, 3-5, 7-9, 12-14, 16-20, 22, 24-26, 27, 29, 30, 35-41, 44, 45-56, 59, 60, and 62-77 were tested using the Functional Assay IV (THP-1 Antagonist Assay) described above. Examples 1, 3, 4, 7-9, 12, 13, 16-20, 22, 24-27, 29, 30, 35, 37-39, 41, 44-56, 60, 62-75, and 77 exhibited pIC50 values ranging from 4.3 to above the upper assay value of 9.1.
例如,以下实施例的THP-1拮抗剂测定的pIC50为:For example, the pIC50 of the THP-1 antagonist of the following example is determined to be:
。 .
Claims (15)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201762568420P | 2017-10-05 | 2017-10-05 | |
| US62/568420 | 2017-10-05 | ||
| PCT/IB2018/057726 WO2019069270A1 (en) | 2017-10-05 | 2018-10-04 | Modulators of stimulator of interferon genes (sting) |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111417630A CN111417630A (en) | 2020-07-14 |
| CN111417630B true CN111417630B (en) | 2023-06-06 |
Family
ID=64049471
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201880078932.4A Expired - Fee Related CN111417630B (en) | 2017-10-05 | 2018-10-04 | Modulator of Stimulator of Interferon Genes (STING) |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US11377440B2 (en) |
| EP (1) | EP3692034A1 (en) |
| JP (1) | JP7291130B2 (en) |
| CN (1) | CN111417630B (en) |
| BR (1) | BR112020006780A2 (en) |
| CA (1) | CA3077337A1 (en) |
| WO (1) | WO2019069270A1 (en) |
Families Citing this family (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB201807924D0 (en) | 2018-05-16 | 2018-06-27 | Ctxt Pty Ltd | Compounds |
| US10947227B2 (en) | 2018-05-25 | 2021-03-16 | Incyte Corporation | Tricyclic heterocyclic compounds as sting activators |
| WO2020010451A1 (en) * | 2018-07-10 | 2020-01-16 | Trillium Therapeutics Inc. | Heteroaromatic-fused imidazolyl amides, compositions and uses thereof as sting agonists |
| WO2020028566A1 (en) | 2018-07-31 | 2020-02-06 | Incyte Corporation | Heteroaryl amide compounds as sting activators |
| WO2020028565A1 (en) | 2018-07-31 | 2020-02-06 | Incyte Corporation | Tricyclic heteraryl compounds as sting activators |
| EP3848366A4 (en) * | 2018-08-29 | 2022-03-16 | Adlai Nortye Biopharma Co., Ltd. | Highly active sting protein agonist compound |
| US11596692B1 (en) | 2018-11-21 | 2023-03-07 | Incyte Corporation | PD-L1/STING conjugates and methods of use |
| WO2020146237A1 (en) | 2019-01-07 | 2020-07-16 | Incyte Corporation | Heteroaryl amide compounds as sting activators |
| JP7655854B2 (en) * | 2019-01-31 | 2025-04-02 | ヒットジェン・インコーポレーテッド | Immunomodulators |
| TW202104214A (en) * | 2019-04-05 | 2021-02-01 | 英商葛蘭素史密斯克藍智慧財產發展有限公司 | Chemical compounds |
| GB201910305D0 (en) | 2019-07-18 | 2019-09-04 | Ctxt Pty Ltd | Compounds |
| GB201910304D0 (en) | 2019-07-18 | 2019-09-04 | Ctxt Pty Ltd | Compounds |
| RS66126B1 (en) * | 2019-07-22 | 2024-11-29 | Lupin Ltd | Macrocyclic compounds as sting agonists and methods and uses thereof |
| JP2022543086A (en) | 2019-08-02 | 2022-10-07 | メルサナ セラピューティクス インコーポレイテッド | Bis-[N-((5-carbamoyl)-1H-benzo[d]imidazol-2-yl)-pyrazole-5-carboxamide] derivatives as STING (interferon gene stimulator) agonists for the treatment of cancer and related Compound |
| CN112521371B (en) * | 2019-09-19 | 2022-11-25 | 中国药科大学 | Heterocyclic amide compounds, their pharmaceutically acceptable salts and their preparation methods and uses |
| CN112940004B (en) * | 2019-12-11 | 2022-07-12 | 中国科学院上海药物研究所 | A kind of heterocyclic compound and use thereof |
| CA3164751A1 (en) * | 2019-12-18 | 2021-06-24 | Benjamin Joseph MORROW | Compounds |
| CN116196435B (en) | 2020-04-02 | 2025-12-02 | 梅尔莎纳医疗公司 | Antibody drug conjugates containing STING agonists |
| US20230141284A1 (en) | 2020-04-10 | 2023-05-11 | Ono Pharmaceutical Co., Ltd. | Cancer therapeutic method |
| WO2021216572A1 (en) | 2020-04-20 | 2021-10-28 | Massachusetts Institute Of Technology | Lipid compositions for delivery of sting agonist compounds and uses thereof |
| US12390537B2 (en) | 2020-05-13 | 2025-08-19 | Massachusetts Institute Of Technology | Compositions of polymeric microdevices and methods of use thereof in cancer immunotherapy |
| CN112920172B (en) * | 2021-02-01 | 2022-03-22 | 厦门大学 | Interferon-stimulated protein targeted compound, radioactive marker thereof, and preparation methods and applications of interferon-stimulated protein targeted compound and radioactive marker |
| JP2024514707A (en) | 2021-04-20 | 2024-04-02 | アンスティテュ・クリー | Compositions and methods for use in immunotherapy |
| WO2022246597A1 (en) * | 2021-05-24 | 2022-12-01 | Forever Millets Limited | Imidazopyridine derivatives as sting agonists |
| AU2022300383A1 (en) * | 2021-06-25 | 2024-01-18 | Bolt Biotherapeutics, Inc. | Bis-benzimidazole sting agonist immunoconjugates, and uses thereof |
| WO2023025256A1 (en) * | 2021-08-26 | 2023-03-02 | 成都先导药物开发股份有限公司 | Sting agonist suitable for use as effector molecule of antibody drug conjugate |
| CN114163420B (en) * | 2021-10-26 | 2023-01-24 | 中山大学附属第一医院 | Endoplasmic reticulum Golgi matrix targeting small molecule, conjugate and application thereof |
| IL319947A (en) | 2022-10-28 | 2025-05-01 | Boehringer Ingelheim Int | Heterocyclic compounds as sting antagonists |
| EP4673445A1 (en) | 2023-02-27 | 2026-01-07 | BioNTech SE | Sting agonists containing benzylic alcohol and benzylic amine functional groups |
| EP4673439A1 (en) | 2023-02-27 | 2026-01-07 | BioNTech SE | Sting agonists containing hydrazide, hydrazine, and hydroxamic acid functional groups |
| EP4687796A1 (en) | 2023-04-05 | 2026-02-11 | Massachusetts Institute of Technology | Hybrid polymeric systems and methods of use thereof |
| US20250108048A1 (en) | 2023-07-31 | 2025-04-03 | Sutro Biopharma, Inc. | Sting agonist compounds and conjugates |
| EP4534147A1 (en) | 2023-10-05 | 2025-04-09 | Sulis Therapeutics ApS | Sting antagonist compounds |
| WO2025228889A1 (en) | 2024-04-30 | 2025-11-06 | Boehringer Ingelheim International Gmbh | Monoarylic heterocyclic compounds as sting antagonists and the use thereof as medicament |
| WO2025228899A1 (en) | 2024-04-30 | 2025-11-06 | Boehringer Ingelheim International Gmbh | Further heterocyclic compounds as sting antagonists and the use thereof as medicament |
| WO2025228895A1 (en) | 2024-04-30 | 2025-11-06 | Boehringer Ingelheim International Gmbh | Hetaryl substituted indazoles and benzimidazoles as sting antagonists and the use thereof as medicament |
| WO2025228902A1 (en) | 2024-04-30 | 2025-11-06 | Boehringer Ingelheim International Gmbh | Heterocyclic acids as sting antagonists and the use thereof as medicament |
| WO2025228900A1 (en) | 2024-04-30 | 2025-11-06 | Boehringer Ingelheim International Gmbh | Arylamide substituted indazoles and the use thereof as medicament |
| US20250333398A1 (en) | 2024-04-30 | 2025-10-30 | Boehringer Ingelheim International Gmbh | Monoaryl and hetaryl substituted indazoles and benzimidazoles as sting antagonists and the use thereof as medicament |
| WO2025250831A1 (en) | 2024-05-30 | 2025-12-04 | Astellas Pharma Inc. | Sting agonist immunostimulatory antibody drug conjugates |
| EP4663204A1 (en) | 2024-06-13 | 2025-12-17 | Sulis Therapeutics ApS | Compounds useful for the modulation of the activity of sting |
| EP4663632A1 (en) | 2024-06-13 | 2025-12-17 | Sulis Therapeutics ApS | Compounds useful for the modulation of the activity of sting |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2674530A1 (en) * | 2007-01-12 | 2008-07-24 | Banyu Pharmaceutical Co., Ltd. | Spirochromanon derivatives |
| WO2017175147A1 (en) * | 2016-04-07 | 2017-10-12 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides useful as protein modulators |
Family Cites Families (83)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5681835A (en) | 1994-04-25 | 1997-10-28 | Glaxo Wellcome Inc. | Non-steroidal ligands for the estrogen receptor |
| US6113918A (en) | 1997-05-08 | 2000-09-05 | Ribi Immunochem Research, Inc. | Aminoalkyl glucosamine phosphate compounds and their use as adjuvants and immunoeffectors |
| GB9716557D0 (en) | 1997-08-06 | 1997-10-08 | Glaxo Group Ltd | Benzylidene-1,3-dihydro-indol-2-one derivatives having anti-cancer activity |
| US6312700B1 (en) | 1998-02-24 | 2001-11-06 | Andrew D. Weinberg | Method for enhancing an antigen specific immune response with OX-40L |
| CA2310896A1 (en) | 1999-07-02 | 2001-01-02 | Japan Tobacco Inc. | Hcv polymerase suitable for crystal structure analysis and method for using the enzyme |
| US7605238B2 (en) | 1999-08-24 | 2009-10-20 | Medarex, Inc. | Human CTLA-4 antibodies and their uses |
| ATE354655T1 (en) | 1999-08-24 | 2007-03-15 | Medarex Inc | HUMAN ANTIBODIES TO CTLA-4 AND THEIR USES |
| JP3871503B2 (en) | 1999-08-30 | 2007-01-24 | 日本たばこ産業株式会社 | Immune disease treatment |
| JP4210454B2 (en) | 2001-03-27 | 2009-01-21 | 日本たばこ産業株式会社 | Inflammatory bowel disease treatment |
| SK13752001A3 (en) | 1999-12-27 | 2002-07-02 | Japan Tobacco, Inc. | Fused-ring compounds and use thereof as drugs |
| ATE395923T1 (en) | 2000-05-19 | 2008-06-15 | Corixa Corp | PROPHYLACTIC AND THERAPEUTIC TREATMENT OF INFECTIOUS, AUTOIMMUNE AND ALLERGIC DISEASES WITH COMPOUNDS BASED ON MONOSACHARIDES |
| US6448281B1 (en) | 2000-07-06 | 2002-09-10 | Boehringer Ingelheim (Canada) Ltd. | Viral polymerase inhibitors |
| US7129219B2 (en) | 2000-08-04 | 2006-10-31 | Corixa Corporation | Immunoeffector compounds |
| US6310224B1 (en) | 2001-01-19 | 2001-10-30 | Arco Chemical Technology, L.P. | Epoxidation catalyst and process |
| RS50236B (en) | 2001-01-22 | 2009-07-15 | Merck & Co.Inc., | NUCLEOSIDE DERIVATIVES AS INVESTORS OF RNA-DEPENDENT RNA VIRAL POLYMERASES |
| JP4212278B2 (en) | 2001-03-01 | 2009-01-21 | 日本たばこ産業株式会社 | Graft rejection inhibitor |
| CN1533390A (en) | 2001-03-19 | 2004-09-29 | СҰҩƷ��ҵ��ʽ���� | Pharmaceutical composition comprising triazaspiro [5.5] undecane derivative as active ingredient |
| AR035543A1 (en) | 2001-06-26 | 2004-06-16 | Japan Tobacco Inc | THERAPEUTIC AGENT FOR HEPATITIS C THAT INCLUDES A CONDENSED RING COMPOUND, CONDENSED RING COMPOUND, PHARMACEUTICAL COMPOSITION THAT UNDERSTANDS, BENZIMIDAZOL, THIAZOL AND BIFENYL COMPOUNDS USED AS INTERMEDIARY COMPARTMENTS OF COMPARTMENTS |
| EP1411928A1 (en) | 2001-07-20 | 2004-04-28 | Boehringer Ingelheim (Canada) Ltd. | Viral polymerase inhibitors |
| US6911434B2 (en) | 2002-02-04 | 2005-06-28 | Corixa Corporation | Prophylactic and therapeutic treatment of infectious and other diseases with immunoeffector compounds |
| US6525028B1 (en) | 2002-02-04 | 2003-02-25 | Corixa Corporation | Immunoeffector compounds |
| IL164376A0 (en) | 2002-04-03 | 2005-12-18 | Applied Research Systems | Ox4or binding agents, their preparation and pharmaceutical compositions containing them |
| EP1497469A4 (en) | 2002-04-04 | 2005-06-08 | Achillion Pharmaceuticals Inc | Assay for evaluation of activity of compounds against hcv using a novel detection system in the hcv replicon |
| DOP2003000641A (en) | 2002-05-10 | 2003-11-15 | Pfizer | INHIBITORS OF RNA-DEPENDENT RNA POLYMERASE OF HEPATITIS C VIRUSES AND COMPOSITIONS AND TREATMENT USED |
| US7550140B2 (en) | 2002-06-13 | 2009-06-23 | Crucell Holland B.V. | Antibody to the human OX40 receptor |
| EP2206517B1 (en) | 2002-07-03 | 2023-08-02 | Ono Pharmaceutical Co., Ltd. | Immunopotentiating compositions comprising anti-PD-L1 antibodies |
| US7304087B2 (en) | 2002-10-24 | 2007-12-04 | Glaxo Group Limited | 1-acyl-pyrrolidine derivatives for the treatment of viral infections |
| ATE402176T1 (en) | 2002-12-13 | 2008-08-15 | Smithkline Beecham Corp | PYRROLIDINE AND AZETIDINE COMPOUNDS ALSCCR5 ANTAGONISTS |
| EP1569934B1 (en) | 2002-12-13 | 2008-01-23 | Smithkline Beecham Corporation | Cyclopropyl compounds as ccr5 antagonists |
| AU2003300911A1 (en) | 2002-12-13 | 2004-07-09 | Smithkline Beecham Corporation | Heterocyclic compounds as ccr5 antagonists |
| AU2003300952A1 (en) | 2002-12-13 | 2004-07-09 | Smithkline Beecham Corporation | Indane compounds as ccr5 antagonists |
| US7645771B2 (en) | 2002-12-13 | 2010-01-12 | Smithkline Beecham Corp. | CCR5 antagonists as therapeutic agents |
| JP2006514646A (en) | 2002-12-13 | 2006-05-11 | スミスクライン ビーチャム コーポレーション | Cyclohexyl compounds as CCR5 antagonists |
| JP4511943B2 (en) | 2002-12-23 | 2010-07-28 | ワイス エルエルシー | Antibody against PD-1 and use thereof |
| US7960522B2 (en) | 2003-01-06 | 2011-06-14 | Corixa Corporation | Certain aminoalkyl glucosaminide phosphate compounds and their use |
| US7223785B2 (en) | 2003-01-22 | 2007-05-29 | Boehringer Ingelheim International Gmbh | Viral polymerase inhibitors |
| US7098231B2 (en) | 2003-01-22 | 2006-08-29 | Boehringer Ingelheim International Gmbh | Viral polymerase inhibitors |
| EP2270051B1 (en) | 2003-01-23 | 2019-05-15 | Ono Pharmaceutical Co., Ltd. | Antibody specific for human PD-1 and CD3 |
| US7148226B2 (en) | 2003-02-21 | 2006-12-12 | Agouron Pharmaceuticals, Inc. | Inhibitors of hepatitis C virus RNA-dependent RNA polymerase, and compositions and treatments using the same |
| WO2005014543A1 (en) | 2003-08-06 | 2005-02-17 | Japan Tobacco Inc. | Condensed ring compound and use thereof as hcv polymerase inhibitor |
| UA86962C2 (en) | 2004-02-20 | 2009-06-10 | Бёрингер Ингельхайм Интернациональ Гмбх | Viral polymerase inhibitors |
| JP4887284B2 (en) | 2004-03-15 | 2012-02-29 | デイビッド・ケイ・アール・カラオリス | Methods for inhibiting the growth of cancer cells or increasing apoptosis of cancer cells |
| EP1748991A1 (en) | 2004-04-28 | 2007-02-07 | Arrow Therapeutics Limited | Morpholinylanilinoquinazo- line derivatives for use as antiviral agents |
| US7153848B2 (en) | 2004-08-09 | 2006-12-26 | Bristol-Myers Squibb Company | Inhibitors of HCV replication |
| UA88909C2 (en) | 2004-08-18 | 2009-12-10 | Пфайзер Инк. | Inhibitors of hepatitis c virus rna-dependent rna polymerase, pharmaceutical composition based thereon and using the same |
| GB0423673D0 (en) | 2004-10-25 | 2004-11-24 | Glaxo Group Ltd | Compounds |
| PL2161336T5 (en) | 2005-05-09 | 2017-10-31 | Ono Pharmaceutical Co | Human monoclonal antibodies to programmed death 1(PD-1) and methods for treating cancer using anti-PD-1 antibodies alone or in combination with other immunotherapeutics |
| CN101247807A (en) | 2005-05-09 | 2008-08-20 | 艾其林医药公司 | Thiazole compounds and methods of use |
| BR122020016659B8 (en) | 2005-05-10 | 2021-07-27 | Incyte Holdings Corp | indoleamine 2,3-dioxygenase modulators and methods of modulating inhibition and immunosuppression activity |
| CN104356236B (en) | 2005-07-01 | 2020-07-03 | E.R.施贵宝&圣斯有限责任公司 | Human monoclonal antibody against programmed death ligand 1 (PD-L1) |
| EP1782826A1 (en) | 2005-11-08 | 2007-05-09 | GBF Gesellschaft für Biotechnologische Forschung mbH | PQS and c-diGMP and its conjugates as adjuvants and their uses in pharmaceutical compositions |
| US20080279851A1 (en) | 2007-05-07 | 2008-11-13 | Medlmmune, Llc | Anti-icos antibodies and their use in treatment of oncology, transplantation and autoimmune disease |
| CA2691357C (en) | 2007-06-18 | 2014-09-23 | N.V. Organon | Antibodies to human programmed death receptor pd-1 |
| PT2594590E (en) | 2007-12-14 | 2015-01-14 | Bristol Myers Squibb Co | Method of producing binding molecules for the human ox40 receptor |
| US8168757B2 (en) | 2008-03-12 | 2012-05-01 | Merck Sharp & Dohme Corp. | PD-1 binding proteins |
| EP2328919A2 (en) | 2008-08-25 | 2011-06-08 | Amplimmune, Inc. | Pd-i antagonists and methods for treating infectious disease |
| CA2998281C (en) | 2008-09-26 | 2022-08-16 | Dana-Farber Cancer Institute, Inc. | Human anti-pd-1 antobodies and uses therefor |
| CN102438652B (en) | 2008-11-12 | 2014-08-13 | 米迪缪尼有限公司 | Antibody formulation |
| EP4331604B9 (en) | 2008-12-09 | 2025-07-23 | F. Hoffmann-La Roche AG | Anti-pd-l1 antibodies and their use to enhance t-cell function |
| CA2778714C (en) | 2009-11-24 | 2018-02-27 | Medimmune Limited | Targeted binding agents against b7-h1 |
| US20130017199A1 (en) | 2009-11-24 | 2013-01-17 | AMPLIMMUNE ,Inc. a corporation | Simultaneous inhibition of pd-l1/pd-l2 |
| US20110280877A1 (en) | 2010-05-11 | 2011-11-17 | Koji Tamada | Inhibition of B7-H1/CD80 interaction and uses thereof |
| CN101898945B (en) | 2010-07-27 | 2013-05-08 | 大连理工大学 | Method for extracting acetone and butyl alcohol in fermentation liquor by salting out |
| NZ716369A (en) | 2010-08-23 | 2017-05-26 | Univ Texas | Anti-ox40 antibodies and methods of using the same |
| ES2612914T3 (en) | 2011-03-31 | 2017-05-19 | Inserm - Institut National De La Santé Et De La Recherche Médicale | Antibodies directed against Icos and their uses |
| KR20190133790A (en) | 2011-08-01 | 2019-12-03 | 제넨테크, 인크. | Methods of treating cancer using pd-1 axis binding antagonists and mek inhibitors |
| CA2845810C (en) | 2011-08-23 | 2017-03-28 | Board Of Regents, The University Of Texas System | Anti-ox40 antibodies and methods of using the same |
| JP2015516989A (en) | 2012-04-30 | 2015-06-18 | バーバー, グレン, エヌ.BARBER, Glen, N. | Modulation of immune response |
| SG10201700698WA (en) | 2012-05-15 | 2017-02-27 | Bristol Myers Squibb Co | Cancer immunotherapy by disrupting pd-1/pd-l1 signaling |
| CN104507538B (en) | 2012-06-08 | 2018-04-06 | 艾杜罗生物科技公司 | Compositions and methods for cancer immunotherapy |
| ES2684552T3 (en) | 2012-09-03 | 2018-10-03 | Inserm - Institut National De La Santé Et De La Recherche Médicale | Antibodies directed against ICOS to treat graft versus host disease |
| CN114507282B (en) | 2012-10-04 | 2025-05-16 | 达纳-法伯癌症研究所公司 | Human monoclonal anti-PD-L1 antibodies and methods of use |
| KR20150095668A (en) | 2012-12-13 | 2015-08-21 | 아두로 바이오테크, 인코포레이티드 | Compositions comprising cyclic purine dinucleotides having defined stereochemistries and methods for their preparation and use |
| US9549944B2 (en) | 2013-05-18 | 2017-01-24 | Aduro Biotech, Inc. | Compositions and methods for inhibiting “stimulator of interferon gene”—dependent signalling |
| US9724408B2 (en) | 2013-05-18 | 2017-08-08 | Aduro Biotech, Inc. | Compositions and methods for activating stimulator of interferon gene-dependent signalling |
| WO2015077354A1 (en) | 2013-11-19 | 2015-05-28 | The University Of Chicago | Use of sting agonist as cancer treatment |
| MX2016015928A (en) | 2014-06-04 | 2017-03-20 | Glaxosmithkline Ip Dev Ltd | Cyclic di-nucleotides as modulators of sting. |
| RU2715038C2 (en) | 2014-07-11 | 2020-02-21 | Дженентек, Инк. | Anti-pd-l1 antibodies and methods for their diagnostic use |
| MA41414A (en) | 2015-01-28 | 2017-12-05 | Centre Nat Rech Scient | ICOS AGONIST BINDING PROTEINS |
| EP3735986A1 (en) | 2015-03-23 | 2020-11-11 | Jounce Therapeutics, Inc. | Antibodies to icos |
| CA2906137A1 (en) * | 2015-09-25 | 2017-03-25 | Pharmascience Inc. | Novel protein kinase inhibitors |
| WO2017106740A1 (en) | 2015-12-16 | 2017-06-22 | Aduro Biotech, Inc. | Methods for identifying inhibitors of "stimulator of interferon gene"-dependent interferon production |
| WO2017175156A1 (en) | 2016-04-07 | 2017-10-12 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides useful as protein modulators |
-
2018
- 2018-10-04 CN CN201880078932.4A patent/CN111417630B/en not_active Expired - Fee Related
- 2018-10-04 US US16/652,221 patent/US11377440B2/en active Active
- 2018-10-04 EP EP18795803.8A patent/EP3692034A1/en not_active Withdrawn
- 2018-10-04 BR BR112020006780-6A patent/BR112020006780A2/en not_active IP Right Cessation
- 2018-10-04 CA CA3077337A patent/CA3077337A1/en active Pending
- 2018-10-04 JP JP2020519371A patent/JP7291130B2/en active Active
- 2018-10-04 WO PCT/IB2018/057726 patent/WO2019069270A1/en not_active Ceased
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2674530A1 (en) * | 2007-01-12 | 2008-07-24 | Banyu Pharmaceutical Co., Ltd. | Spirochromanon derivatives |
| WO2017175147A1 (en) * | 2016-04-07 | 2017-10-12 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides useful as protein modulators |
Also Published As
| Publication number | Publication date |
|---|---|
| CN111417630A (en) | 2020-07-14 |
| US20200291001A1 (en) | 2020-09-17 |
| CA3077337A1 (en) | 2019-04-11 |
| EP3692034A1 (en) | 2020-08-12 |
| BR112020006780A2 (en) | 2020-10-06 |
| JP7291130B2 (en) | 2023-06-14 |
| US11377440B2 (en) | 2022-07-05 |
| JP2020536103A (en) | 2020-12-10 |
| WO2019069270A1 (en) | 2019-04-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN111417630B (en) | Modulator of Stimulator of Interferon Genes (STING) | |
| JP7119158B2 (en) | Heterocyclic Amides Useful as Protein Modulators | |
| EP3692033A1 (en) | Modulators of stimulator of interferon genes (sting) useful in treating hiv | |
| WO2017175156A1 (en) | Heterocyclic amides useful as protein modulators | |
| EA037671B1 (en) | Heterocyclic amides useful as protein modulators | |
| HK40069191A (en) | Heterocyclic amides useful as protein modulators | |
| HK1259737B (en) | Heterocyclic amides useful as protein modulators | |
| HK1259737A1 (en) | Heterocyclic amides useful as protein modulators |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20230606 |