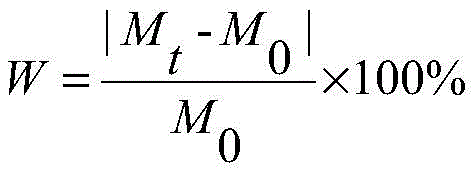CN111388154B - Absorbable implant medical device - Google Patents
Absorbable implant medical device Download PDFInfo
- Publication number
- CN111388154B CN111388154B CN201811625648.2A CN201811625648A CN111388154B CN 111388154 B CN111388154 B CN 111388154B CN 201811625648 A CN201811625648 A CN 201811625648A CN 111388154 B CN111388154 B CN 111388154B
- Authority
- CN
- China
- Prior art keywords
- acid
- anhydride
- gel layer
- medical device
- corrosion
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000007943 implant Substances 0.000 title claims description 37
- 239000000463 material Substances 0.000 claims abstract description 57
- 239000000126 substance Substances 0.000 claims abstract description 52
- 229910052751 metal Inorganic materials 0.000 claims abstract description 33
- 239000002184 metal Substances 0.000 claims abstract description 33
- 239000000758 substrate Substances 0.000 claims abstract description 19
- 239000010410 layer Substances 0.000 claims description 123
- 239000003814 drug Substances 0.000 claims description 87
- 229940079593 drug Drugs 0.000 claims description 75
- 150000008064 anhydrides Chemical class 0.000 claims description 46
- 239000011248 coating agent Substances 0.000 claims description 42
- 238000000576 coating method Methods 0.000 claims description 42
- 229920000642 polymer Polymers 0.000 claims description 22
- 229920000747 poly(lactic acid) Polymers 0.000 claims description 21
- 239000004626 polylactic acid Substances 0.000 claims description 21
- UREBDLICKHMUKA-CXSFZGCWSA-N dexamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-CXSFZGCWSA-N 0.000 claims description 17
- 229960003957 dexamethasone Drugs 0.000 claims description 17
- -1 polybutylene succinate Polymers 0.000 claims description 15
- 229920002732 Polyanhydride Polymers 0.000 claims description 14
- 239000002253 acid Substances 0.000 claims description 14
- 230000001737 promoting effect Effects 0.000 claims description 14
- QFJCIRLUMZQUOT-HPLJOQBZSA-N sirolimus Chemical compound C1C[C@@H](O)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 QFJCIRLUMZQUOT-HPLJOQBZSA-N 0.000 claims description 13
- 229920000728 polyester Polymers 0.000 claims description 11
- 229940124599 anti-inflammatory drug Drugs 0.000 claims description 10
- 229920001577 copolymer Polymers 0.000 claims description 10
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 claims description 9
- 229920000331 Polyhydroxybutyrate Polymers 0.000 claims description 9
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 claims description 9
- 239000011247 coating layer Substances 0.000 claims description 9
- 230000003628 erosive effect Effects 0.000 claims description 9
- 239000000178 monomer Substances 0.000 claims description 9
- ZAHRKKWIAAJSAO-UHFFFAOYSA-N rapamycin Natural products COCC(O)C(=C/C(C)C(=O)CC(OC(=O)C1CCCCN1C(=O)C(=O)C2(O)OC(CC(OC)C(=CC=CC=CC(C)CC(C)C(=O)C)C)CCC2C)C(C)CC3CCC(O)C(C3)OC)C ZAHRKKWIAAJSAO-UHFFFAOYSA-N 0.000 claims description 9
- 229960002930 sirolimus Drugs 0.000 claims description 9
- 230000002792 vascular Effects 0.000 claims description 9
- 239000002260 anti-inflammatory agent Substances 0.000 claims description 8
- 229940127218 antiplatelet drug Drugs 0.000 claims description 8
- 229930012538 Paclitaxel Natural products 0.000 claims description 7
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 claims description 7
- 229940127217 antithrombotic drug Drugs 0.000 claims description 7
- 239000008139 complexing agent Substances 0.000 claims description 7
- 230000002401 inhibitory effect Effects 0.000 claims description 7
- 239000000203 mixture Substances 0.000 claims description 7
- 229960001592 paclitaxel Drugs 0.000 claims description 7
- 239000011975 tartaric acid Substances 0.000 claims description 7
- 235000002906 tartaric acid Nutrition 0.000 claims description 7
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 claims description 7
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 claims description 6
- 229920001397 Poly-beta-hydroxybutyrate Polymers 0.000 claims description 6
- 229920000954 Polyglycolide Polymers 0.000 claims description 6
- BJEPYKJPYRNKOW-UHFFFAOYSA-N malic acid Chemical compound OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 claims description 6
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 claims description 6
- 229920001610 polycaprolactone Polymers 0.000 claims description 6
- 239000004632 polycaprolactone Substances 0.000 claims description 6
- 108010077051 polycysteine Proteins 0.000 claims description 6
- 239000004633 polyglycolic acid Substances 0.000 claims description 6
- 230000035755 proliferation Effects 0.000 claims description 6
- 239000001577 tetrasodium phosphonato phosphate Substances 0.000 claims description 6
- CWERGRDVMFNCDR-UHFFFAOYSA-N thioglycolic acid Chemical compound OC(=O)CS CWERGRDVMFNCDR-UHFFFAOYSA-N 0.000 claims description 6
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 claims description 6
- 239000003146 anticoagulant agent Substances 0.000 claims description 5
- 229920000141 poly(maleic anhydride) Polymers 0.000 claims description 5
- FUFLCEKSBBHCMO-UHFFFAOYSA-N 11-dehydrocorticosterone Natural products O=C1CCC2(C)C3C(=O)CC(C)(C(CC4)C(=O)CO)C4C3CCC2=C1 FUFLCEKSBBHCMO-UHFFFAOYSA-N 0.000 claims description 4
- DBAKFASWICGISY-BTJKTKAUSA-N Chlorpheniramine maleate Chemical compound OC(=O)\C=C/C(O)=O.C=1C=CC=NC=1C(CCN(C)C)C1=CC=C(Cl)C=C1 DBAKFASWICGISY-BTJKTKAUSA-N 0.000 claims description 4
- MFYSYFVPBJMHGN-ZPOLXVRWSA-N Cortisone Chemical compound O=C1CC[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 MFYSYFVPBJMHGN-ZPOLXVRWSA-N 0.000 claims description 4
- MFYSYFVPBJMHGN-UHFFFAOYSA-N Cortisone Natural products O=C1CCC2(C)C3C(=O)CC(C)(C(CC4)(O)C(=O)CO)C4C3CCC2=C1 MFYSYFVPBJMHGN-UHFFFAOYSA-N 0.000 claims description 4
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 claims description 4
- 206010070834 Sensitisation Diseases 0.000 claims description 4
- 239000013543 active substance Substances 0.000 claims description 4
- 239000004227 calcium gluconate Substances 0.000 claims description 4
- 229960004494 calcium gluconate Drugs 0.000 claims description 4
- 235000013927 calcium gluconate Nutrition 0.000 claims description 4
- NEEHYRZPVYRGPP-UHFFFAOYSA-L calcium;2,3,4,5,6-pentahydroxyhexanoate Chemical compound [Ca+2].OCC(O)C(O)C(O)C(O)C([O-])=O.OCC(O)C(O)C(O)C(O)C([O-])=O NEEHYRZPVYRGPP-UHFFFAOYSA-L 0.000 claims description 4
- 229960004588 cilostazol Drugs 0.000 claims description 4
- RRGUKTPIGVIEKM-UHFFFAOYSA-N cilostazol Chemical compound C=1C=C2NC(=O)CCC2=CC=1OCCCCC1=NN=NN1C1CCCCC1 RRGUKTPIGVIEKM-UHFFFAOYSA-N 0.000 claims description 4
- 229960004544 cortisone Drugs 0.000 claims description 4
- 229960002897 heparin Drugs 0.000 claims description 4
- 229920000669 heparin Polymers 0.000 claims description 4
- 229920002961 polybutylene succinate Polymers 0.000 claims description 4
- 239000004631 polybutylene succinate Substances 0.000 claims description 4
- 150000004579 taxol derivatives Chemical class 0.000 claims description 4
- LLPGFORTLHNELX-UHFFFAOYSA-N (2-carbamoylphenyl) hydrogen sulfate Chemical compound NC(=O)C1=CC=CC=C1OS(O)(=O)=O LLPGFORTLHNELX-UHFFFAOYSA-N 0.000 claims description 3
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 claims description 3
- VILCJCGEZXAXTO-UHFFFAOYSA-N 2,2,2-tetramine Chemical compound NCCNCCNCCN VILCJCGEZXAXTO-UHFFFAOYSA-N 0.000 claims description 3
- NBYLBWHHTUWMER-UHFFFAOYSA-N 2-Methylquinolin-8-ol Chemical compound C1=CC=C(O)C2=NC(C)=CC=C21 NBYLBWHHTUWMER-UHFFFAOYSA-N 0.000 claims description 3
- WXHLLJAMBQLULT-UHFFFAOYSA-N 2-[[6-[4-(2-hydroxyethyl)piperazin-1-yl]-2-methylpyrimidin-4-yl]amino]-n-(2-methyl-6-sulfanylphenyl)-1,3-thiazole-5-carboxamide;hydrate Chemical compound O.C=1C(N2CCN(CCO)CC2)=NC(C)=NC=1NC(S1)=NC=C1C(=O)NC1=C(C)C=CC=C1S WXHLLJAMBQLULT-UHFFFAOYSA-N 0.000 claims description 3
- YDQUROLTIDVHRK-UHFFFAOYSA-N 2-hydroxypropane-1,2,3-tricarboxylic acid;propane-1,2,3-triol Chemical compound OCC(O)CO.OC(=O)CC(O)(C(O)=O)CC(O)=O YDQUROLTIDVHRK-UHFFFAOYSA-N 0.000 claims description 3
- BSYNRYMUTXBXSQ-FOQJRBATSA-N 59096-14-9 Chemical compound CC(=O)OC1=CC=CC=C1[14C](O)=O BSYNRYMUTXBXSQ-FOQJRBATSA-N 0.000 claims description 3
- 239000005725 8-Hydroxyquinoline Substances 0.000 claims description 3
- LGDFHDKSYGVKDC-UHFFFAOYSA-N 8-hydroxyquinoline-5-sulfonic acid Chemical compound C1=CN=C2C(O)=CC=C(S(O)(=O)=O)C2=C1 LGDFHDKSYGVKDC-UHFFFAOYSA-N 0.000 claims description 3
- 239000004114 Ammonium polyphosphate Substances 0.000 claims description 3
- ROFVEXUMMXZLPA-UHFFFAOYSA-N Bipyridyl Chemical compound N1=CC=CC=C1C1=CC=CC=N1 ROFVEXUMMXZLPA-UHFFFAOYSA-N 0.000 claims description 3
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 claims description 3
- RPNUMPOLZDHAAY-UHFFFAOYSA-N Diethylenetriamine Chemical compound NCCNCCN RPNUMPOLZDHAAY-UHFFFAOYSA-N 0.000 claims description 3
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 claims description 3
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 claims description 3
- TZXKOCQBRNJULO-UHFFFAOYSA-N Ferriprox Chemical compound CC1=C(O)C(=O)C=CN1C TZXKOCQBRNJULO-UHFFFAOYSA-N 0.000 claims description 3
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 claims description 3
- 102000001554 Hemoglobins Human genes 0.000 claims description 3
- 108010054147 Hemoglobins Proteins 0.000 claims description 3
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical compound ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 claims description 3
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical compound NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 claims description 3
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 claims description 3
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 claims description 3
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 claims description 3
- 239000004472 Lysine Substances 0.000 claims description 3
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 claims description 3
- AHLPHDHHMVZTML-UHFFFAOYSA-N Orn-delta-NH2 Natural products NCCCC(N)C(O)=O AHLPHDHHMVZTML-UHFFFAOYSA-N 0.000 claims description 3
- UTJLXEIPEHZYQJ-UHFFFAOYSA-N Ornithine Natural products OC(=O)C(C)CCCN UTJLXEIPEHZYQJ-UHFFFAOYSA-N 0.000 claims description 3
- NIBALARBNHMNPW-UHFFFAOYSA-N P1(OCCCCCO1)=O.NCCNCCN.[K] Chemical compound P1(OCCCCCO1)=O.NCCNCCN.[K] NIBALARBNHMNPW-UHFFFAOYSA-N 0.000 claims description 3
- BVBIQASDHXEFKW-UHFFFAOYSA-N P1(OCCCCO1)=O.C(CN)N.[Na] Chemical compound P1(OCCCCO1)=O.C(CN)N.[Na] BVBIQASDHXEFKW-UHFFFAOYSA-N 0.000 claims description 3
- 229920000805 Polyaspartic acid Polymers 0.000 claims description 3
- 239000004698 Polyethylene Substances 0.000 claims description 3
- 239000002202 Polyethylene glycol Substances 0.000 claims description 3
- 108010020346 Polyglutamic Acid Proteins 0.000 claims description 3
- 108010039918 Polylysine Proteins 0.000 claims description 3
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 claims description 3
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 claims description 3
- DGEZNRSVGBDHLK-UHFFFAOYSA-N [1,10]phenanthroline Chemical compound C1=CN=C2C3=NC=CC=C3C=CC2=C1 DGEZNRSVGBDHLK-UHFFFAOYSA-N 0.000 claims description 3
- WNLRTRBMVRJNCN-UHFFFAOYSA-L adipate(2-) Chemical compound [O-]C(=O)CCCCC([O-])=O WNLRTRBMVRJNCN-UHFFFAOYSA-L 0.000 claims description 3
- 239000000783 alginic acid Substances 0.000 claims description 3
- 235000010443 alginic acid Nutrition 0.000 claims description 3
- 229920000615 alginic acid Polymers 0.000 claims description 3
- 229960001126 alginic acid Drugs 0.000 claims description 3
- 150000004781 alginic acids Chemical class 0.000 claims description 3
- 235000019826 ammonium polyphosphate Nutrition 0.000 claims description 3
- 229920001276 ammonium polyphosphate Polymers 0.000 claims description 3
- 235000003704 aspartic acid Nutrition 0.000 claims description 3
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 claims description 3
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 claims description 3
- FNAQSUUGMSOBHW-UHFFFAOYSA-H calcium citrate Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O.[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O FNAQSUUGMSOBHW-UHFFFAOYSA-H 0.000 claims description 3
- 239000001354 calcium citrate Substances 0.000 claims description 3
- 239000003795 chemical substances by application Substances 0.000 claims description 3
- 229930002875 chlorophyll Natural products 0.000 claims description 3
- 235000019804 chlorophyll Nutrition 0.000 claims description 3
- ATNHDLDRLWWWCB-AENOIHSZSA-M chlorophyll a Chemical compound C1([C@@H](C(=O)OC)C(=O)C2=C3C)=C2N2C3=CC(C(CC)=C3C)=[N+]4C3=CC3=C(C=C)C(C)=C5N3[Mg-2]42[N+]2=C1[C@@H](CCC(=O)OC\C=C(/C)CCC[C@H](C)CCC[C@H](C)CCCC(C)C)[C@H](C)C2=C5 ATNHDLDRLWWWCB-AENOIHSZSA-M 0.000 claims description 3
- 229960003291 chlorphenamine Drugs 0.000 claims description 3
- 239000001530 fumaric acid Substances 0.000 claims description 3
- 239000004220 glutamic acid Substances 0.000 claims description 3
- 235000013922 glutamic acid Nutrition 0.000 claims description 3
- 235000018977 lysine Nutrition 0.000 claims description 3
- 229940049920 malate Drugs 0.000 claims description 3
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 claims description 3
- 239000011976 maleic acid Substances 0.000 claims description 3
- 239000001630 malic acid Substances 0.000 claims description 3
- 235000011090 malic acid Nutrition 0.000 claims description 3
- KNBBZGZVUYEUGI-UHFFFAOYSA-N methyl 5-methyl-2-sulfanylbenzoate Chemical compound COC(=O)C1=CC(C)=CC=C1S KNBBZGZVUYEUGI-UHFFFAOYSA-N 0.000 claims description 3
- MGFYIUFZLHCRTH-UHFFFAOYSA-N nitrilotriacetic acid Chemical compound OC(=O)CN(CC(O)=O)CC(O)=O MGFYIUFZLHCRTH-UHFFFAOYSA-N 0.000 claims description 3
- 229960003104 ornithine Drugs 0.000 claims description 3
- 230000000399 orthopedic effect Effects 0.000 claims description 3
- 235000006408 oxalic acid Nutrition 0.000 claims description 3
- KHPXUQMNIQBQEV-UHFFFAOYSA-N oxaloacetic acid Chemical compound OC(=O)CC(=O)C(O)=O KHPXUQMNIQBQEV-UHFFFAOYSA-N 0.000 claims description 3
- 229960003540 oxyquinoline Drugs 0.000 claims description 3
- JZRYQZJSTWVBBD-UHFFFAOYSA-N pentaporphyrin i Chemical compound N1C(C=C2NC(=CC3=NC(=C4)C=C3)C=C2)=CC=C1C=C1C=CC4=N1 JZRYQZJSTWVBBD-UHFFFAOYSA-N 0.000 claims description 3
- 239000005015 poly(hydroxybutyrate) Substances 0.000 claims description 3
- 108010064470 polyaspartate Proteins 0.000 claims description 3
- 229920000573 polyethylene Polymers 0.000 claims description 3
- 229920000921 polyethylene adipate Polymers 0.000 claims description 3
- 229920001223 polyethylene glycol Polymers 0.000 claims description 3
- 229920002643 polyglutamic acid Polymers 0.000 claims description 3
- 229920000656 polylysine Polymers 0.000 claims description 3
- 108010055896 polyornithine Proteins 0.000 claims description 3
- 229920000137 polyphosphoric acid Polymers 0.000 claims description 3
- 150000004032 porphyrins Chemical class 0.000 claims description 3
- 239000001508 potassium citrate Substances 0.000 claims description 3
- 229960002635 potassium citrate Drugs 0.000 claims description 3
- QEEAPRPFLLJWCF-UHFFFAOYSA-K potassium citrate (anhydrous) Chemical compound [K+].[K+].[K+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O QEEAPRPFLLJWCF-UHFFFAOYSA-K 0.000 claims description 3
- 235000011082 potassium citrates Nutrition 0.000 claims description 3
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 3
- MCJGNVYPOGVAJF-UHFFFAOYSA-N quinolin-8-ol Chemical compound C1=CN=C2C(O)=CC=CC2=C1 MCJGNVYPOGVAJF-UHFFFAOYSA-N 0.000 claims description 3
- MHTSJSRDFXZFHQ-UHFFFAOYSA-N quinoline-8-thiol Chemical compound C1=CN=C2C(S)=CC=CC2=C1 MHTSJSRDFXZFHQ-UHFFFAOYSA-N 0.000 claims description 3
- 230000000241 respiratory effect Effects 0.000 claims description 3
- FQENQNTWSFEDLI-UHFFFAOYSA-J sodium diphosphate Chemical compound [Na+].[Na+].[Na+].[Na+].[O-]P([O-])(=O)OP([O-])([O-])=O FQENQNTWSFEDLI-UHFFFAOYSA-J 0.000 claims description 3
- GCLGEJMYGQKIIW-UHFFFAOYSA-H sodium hexametaphosphate Chemical compound [Na]OP1(=O)OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])O1 GCLGEJMYGQKIIW-UHFFFAOYSA-H 0.000 claims description 3
- 235000019982 sodium hexametaphosphate Nutrition 0.000 claims description 3
- 229940048086 sodium pyrophosphate Drugs 0.000 claims description 3
- 235000019818 tetrasodium diphosphate Nutrition 0.000 claims description 3
- UEUXEKPTXMALOB-UHFFFAOYSA-J tetrasodium;2-[2-[bis(carboxylatomethyl)amino]ethyl-(carboxylatomethyl)amino]acetate Chemical compound [Na+].[Na+].[Na+].[Na+].[O-]C(=O)CN(CC([O-])=O)CCN(CC([O-])=O)CC([O-])=O UEUXEKPTXMALOB-UHFFFAOYSA-J 0.000 claims description 3
- 235000013337 tricalcium citrate Nutrition 0.000 claims description 3
- UNXRWKVEANCORM-UHFFFAOYSA-N triphosphoric acid Chemical compound OP(O)(=O)OP(O)(=O)OP(O)(O)=O UNXRWKVEANCORM-UHFFFAOYSA-N 0.000 claims description 3
- 229940070710 valerate Drugs 0.000 claims description 3
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 claims description 3
- ISLHTVXEVCZNHS-UHFFFAOYSA-N 4,5-dihydroxybenzene-1,3-disulfonic acid;sodium Chemical compound [Na].OC1=CC(S(O)(=O)=O)=CC(S(O)(=O)=O)=C1O ISLHTVXEVCZNHS-UHFFFAOYSA-N 0.000 claims description 2
- ZDPZWHLPCUZHPA-UHFFFAOYSA-N 6-hydroxy-5-nitrosonaphthalene-2-sulfonic acid Chemical compound C1=C(S(O)(=O)=O)C=CC2=C(N=O)C(O)=CC=C21 ZDPZWHLPCUZHPA-UHFFFAOYSA-N 0.000 claims description 2
- 125000000738 acetamido group Chemical group [H]C([H])([H])C(=O)N([H])[*] 0.000 claims description 2
- 150000001298 alcohols Chemical class 0.000 claims description 2
- 229940121363 anti-inflammatory agent Drugs 0.000 claims description 2
- 230000006698 induction Effects 0.000 claims description 2
- 108010094020 polyglycine Proteins 0.000 claims description 2
- 229960004676 antithrombotic agent Drugs 0.000 claims 1
- 108010087948 polymethionine Proteins 0.000 claims 1
- 230000007797 corrosion Effects 0.000 abstract description 29
- 238000005260 corrosion Methods 0.000 abstract description 29
- 239000000243 solution Substances 0.000 description 38
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 36
- 239000011159 matrix material Substances 0.000 description 32
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 27
- 239000002904 solvent Substances 0.000 description 19
- 238000000034 method Methods 0.000 description 16
- 229920001427 mPEG Polymers 0.000 description 15
- 206010061218 Inflammation Diseases 0.000 description 14
- 241000283973 Oryctolagus cuniculus Species 0.000 description 14
- 230000004054 inflammatory process Effects 0.000 description 14
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 12
- 238000001816 cooling Methods 0.000 description 11
- 238000002513 implantation Methods 0.000 description 9
- 238000002156 mixing Methods 0.000 description 9
- 239000012634 fragment Substances 0.000 description 8
- 230000000052 comparative effect Effects 0.000 description 7
- 239000008279 sol Substances 0.000 description 7
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 6
- 229910052742 iron Inorganic materials 0.000 description 6
- 238000005516 engineering process Methods 0.000 description 4
- FALRKNHUBBKYCC-UHFFFAOYSA-N 2-(chloromethyl)pyridine-3-carbonitrile Chemical compound ClCC1=NC=CC=C1C#N FALRKNHUBBKYCC-UHFFFAOYSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 3
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 3
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 229960001138 acetylsalicylic acid Drugs 0.000 description 3
- WNNNWFKQCKFSDK-UHFFFAOYSA-N allylglycine Chemical compound OC(=O)C(N)CC=C WNNNWFKQCKFSDK-UHFFFAOYSA-N 0.000 description 3
- 239000000043 antiallergic agent Substances 0.000 description 3
- 210000004204 blood vessel Anatomy 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 239000000106 platelet aggregation inhibitor Substances 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 229940014800 succinic anhydride Drugs 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 3
- 238000002604 ultrasonography Methods 0.000 description 3
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 230000003110 anti-inflammatory effect Effects 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 239000007857 degradation product Substances 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 230000001939 inductive effect Effects 0.000 description 2
- 230000008595 infiltration Effects 0.000 description 2
- 238000001764 infiltration Methods 0.000 description 2
- 239000011259 mixed solution Substances 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 230000002093 peripheral effect Effects 0.000 description 2
- 229920002714 polyornithine Polymers 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 238000002791 soaking Methods 0.000 description 2
- 238000009210 therapy by ultrasound Methods 0.000 description 2
- 238000001291 vacuum drying Methods 0.000 description 2
- 238000005303 weighing Methods 0.000 description 2
- GRWFGVWFFZKLTI-UHFFFAOYSA-N α-pinene Chemical compound CC1=CCC2C(C)(C)C1C2 GRWFGVWFFZKLTI-UHFFFAOYSA-N 0.000 description 2
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- YXAOOTNFFAQIPZ-UHFFFAOYSA-N 1-nitrosonaphthalen-2-ol Chemical compound C1=CC=CC2=C(N=O)C(O)=CC=C21 YXAOOTNFFAQIPZ-UHFFFAOYSA-N 0.000 description 1
- GRWFGVWFFZKLTI-IUCAKERBSA-N 1S,5S-(-)-alpha-Pinene Natural products CC1=CC[C@@H]2C(C)(C)[C@H]1C2 GRWFGVWFFZKLTI-IUCAKERBSA-N 0.000 description 1
- JPSKCQCQZUGWNM-UHFFFAOYSA-N 2,7-Oxepanedione Chemical compound O=C1CCCCC(=O)O1 JPSKCQCQZUGWNM-UHFFFAOYSA-N 0.000 description 1
- WQNHWIYLCRZRLR-UHFFFAOYSA-N 2-(3-hydroxy-2,5-dioxooxolan-3-yl)acetic acid Chemical compound OC(=O)CC1(O)CC(=O)OC1=O WQNHWIYLCRZRLR-UHFFFAOYSA-N 0.000 description 1
- BOGVTNYNTGOONP-UHFFFAOYSA-N 3,4-dihydroxyoxolane-2,5-dione Chemical compound OC1C(O)C(=O)OC1=O BOGVTNYNTGOONP-UHFFFAOYSA-N 0.000 description 1
- KPYCVQASEGGKEG-UHFFFAOYSA-N 3-hydroxyoxolane-2,5-dione Chemical compound OC1CC(=O)OC1=O KPYCVQASEGGKEG-UHFFFAOYSA-N 0.000 description 1
- OFNISBHGPNMTMS-UHFFFAOYSA-N 3-methylideneoxolane-2,5-dione Chemical compound C=C1CC(=O)OC1=O OFNISBHGPNMTMS-UHFFFAOYSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 208000031481 Pathologic Constriction Diseases 0.000 description 1
- 229920001244 Poly(D,L-lactide) Polymers 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- ISWQCIVKKSOKNN-UHFFFAOYSA-L Tiron Chemical compound [Na+].[Na+].OC1=CC(S([O-])(=O)=O)=CC(S([O-])(=O)=O)=C1O ISWQCIVKKSOKNN-UHFFFAOYSA-L 0.000 description 1
- 239000000956 alloy Substances 0.000 description 1
- 229910045601 alloy Inorganic materials 0.000 description 1
- MVNCAPSFBDBCGF-UHFFFAOYSA-N alpha-pinene Natural products CC1=CCC23C1CC2C3(C)C MVNCAPSFBDBCGF-UHFFFAOYSA-N 0.000 description 1
- 230000033115 angiogenesis Effects 0.000 description 1
- 210000000702 aorta abdominal Anatomy 0.000 description 1
- 239000012300 argon atmosphere Substances 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 210000001124 body fluid Anatomy 0.000 description 1
- 239000010839 body fluid Substances 0.000 description 1
- 230000001680 brushing effect Effects 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 210000004351 coronary vessel Anatomy 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- 229920006237 degradable polymer Polymers 0.000 description 1
- 238000007598 dipping method Methods 0.000 description 1
- NWADXBLMWHFGGU-UHFFFAOYSA-N dodecanoic anhydride Chemical compound CCCCCCCCCCCC(=O)OC(=O)CCCCCCCCCCC NWADXBLMWHFGGU-UHFFFAOYSA-N 0.000 description 1
- 238000010041 electrostatic spinning Methods 0.000 description 1
- OAYLNYINCPYISS-UHFFFAOYSA-N ethyl acetate;hexane Chemical compound CCCCCC.CCOC(C)=O OAYLNYINCPYISS-UHFFFAOYSA-N 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 238000011010 flushing procedure Methods 0.000 description 1
- 230000035876 healing Effects 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 210000004969 inflammatory cell Anatomy 0.000 description 1
- 150000004698 iron complex Chemical class 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- YOURXVGYNVXQKT-UHFFFAOYSA-N oxacycloundecane-2,11-dione Chemical compound O=C1CCCCCCCCC(=O)O1 YOURXVGYNVXQKT-UHFFFAOYSA-N 0.000 description 1
- 229920000232 polyglycine polymer Polymers 0.000 description 1
- 230000001376 precipitating effect Effects 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 230000036262 stenosis Effects 0.000 description 1
- 208000037804 stenosis Diseases 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- UCPYLLCMEDAXFR-UHFFFAOYSA-N triphosgene Chemical compound ClC(Cl)(Cl)OC(=O)OC(Cl)(Cl)Cl UCPYLLCMEDAXFR-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/82—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/02—Inorganic materials
- A61L31/022—Metals or alloys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/08—Materials for coatings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/08—Materials for coatings
- A61L31/10—Macromolecular materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L31/148—Materials at least partially resorbable by the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L31/00—Materials for other surgical articles, e.g. stents, stent-grafts, shunts, surgical drapes, guide wires, materials for adhesion prevention, occluding devices, surgical gloves, tissue fixation devices
- A61L31/14—Materials characterised by their function or physical properties, e.g. injectable or lubricating compositions, shape-memory materials, surface modified materials
- A61L31/16—Biologically active materials, e.g. therapeutic substances
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2210/00—Particular material properties of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2210/0004—Particular material properties of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof bioabsorbable
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Heart & Thoracic Surgery (AREA)
- Vascular Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Surgery (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Cardiology (AREA)
- Transplantation (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Inorganic Chemistry (AREA)
- Materials For Medical Uses (AREA)
Abstract
The invention relates to an absorbable implanted medical device, which comprises a metal substrate and a gel layer covering the surface of the metal substrate, wherein the gel layer comprises an organic gel material and a corrosion-promoting substance, and the mass ratio of the organic gel material to the corrosion-promoting substance is (1.5-5.5): 1. The organogel material can be transformed from a gel state to a sol state, and the sol state can release corrosion-promoting substances, so that the corrosion of the absorbable implantable medical device can be artificially controlled after the absorbable implantable medical device is implanted.
Description
Technical Field
The invention relates to the field of medical instruments, in particular to an absorbable implanted medical instrument.
Background
The current treatment of luminal stenosis is the intervention, which has progressed from early balloon-only dilatation, metal bare stents to Drug Eluting Stents (DES) and even the latest bioabsorbable stents (BRS) today. From the clinical point of view, an ideal absorbable stent should have the characteristics that the shorter the period the stent gradually erodes or degrades and is absorbed by the body, the better when the absorbable stent has completed its intended use, i.e., the diseased site heals and returns to normal form and function. Depending on the site of clinical stent application, the luminal healing period is typically 3 to 6 months, during which time the stent needs to maintain structural integrity and provide adequate support. After the stent is effectively supported, the biological risk is higher the longer the stent is in the lumen, so the corrosion speed of the stent needs to be increased to shorten the corrosion and absorption period of the stent, thereby reducing the late biological risk of the stent. However, the current BRS has two problems: firstly, the corrosion rate of the existing absorbable stent is controlled by the established objective factors such as the material composition, the coating structure and the like of the existing absorbable stent, but the integrity and the sufficient mechanical property of the stent structure at the early stage of implantation and the rapid corrosion and absorption of the stent at the later stage of implantation cannot be ensured simultaneously due to the existence of the patient with the heterogeneity; secondly, inflammation often occurs after the stent is corroded, and at the moment, the outer-layer medicine is basically completely released and cannot well resist the inflammation.
Disclosure of Invention
In view of this, there is a need for an absorbable implantable medical device that can be artificially controlled for corrosive effects after implantation.
An absorbable implanted medical device comprises a metal substrate and a gel layer covering the surface of the metal substrate, wherein the gel layer comprises an organic gel material and an erosion promoting substance, and the mass ratio of the organic gel material to the erosion promoting substance is (1.5-5.5): 1.
Further, the gel layer also comprises an active drug, and the mass ratio of the organogel material to the active drug to the corrosion-promoting substance is (2-5.5): (0.05-0.5): 1.
further, the gel layer comprises an outer gel layer and an inner gel layer, the outer gel layer covers the outer surface of the metal base, the inner gel layer covers the inner surface of the metal base, and the thickness ratio between the outer gel layer and the inner gel layer is 1.5-4.
Further, at least the outer gel layer of the outer gel layer and the inner gel layer further comprises an active drug.
Further, the absorbable implant medical device further comprises a drug coating, wherein the drug coating comprises an active drug and a polymer, and the mass ratio of the active drug to the corrosion-promoting substance is 1: (2-20), the drug coating layer covers at least the outer surface of the gel layer.
Further, the thickness of the gel layer ranges from 2 to 33 micrometers.
Further, the active drug is at least one selected from drugs for inhibiting vascular proliferation, antiplatelet drugs, antithrombotic drugs, anti-inflammatory drugs and anti-allergic drugs, and the drugs for inhibiting vascular proliferation is at least one selected from paclitaxel, rapamycin, paclitaxel derivatives and rapamycin derivatives; the antiplatelet drug is cilostazol; the antithrombotic drug is heparin; the anti-inflammatory drug is dexamethasone; the anti-sensitization medicine is at least one of calcium gluconate, chlorphenamine and cortisone.
Further, the corrosion-promoting substance is at least one selected from degradable polyester, degradable polyanhydride, degradable polyamino acid and complexing agent.
Further, the degradable polyester is selected from any one or a physical blend of at least two of polylactic acid, polyglycolic acid, polybutylene succinate, poly (beta-hydroxybutyrate), polycaprolactone, polyethylene adipate, polypentanoate, polyhydroxyalkyl alcohol ester and poly (malate), or a copolymer of at least two of monomers forming the degradable polyester-based polymer; the degradable polyanhydride is selected from at least one of polyanhydride, polysuccinic anhydride, polyhexamic anhydride, polysebacic anhydride, polydodecanoic anhydride, polycitric anhydride, polymalic anhydride, polysuccinic anhydride, polytartaric anhydride, polyitaconic anhydride and polymaleic anhydride; or the degradable polyanhydride is a copolymer formed by copolymerizing at least one of monomers forming the polyethylene anhydride, the polysuccinic anhydride, the polyhexamic anhydride, the polysebacic anhydride, the polycitric anhydride, the polymalic anhydride, the polysuccinic anhydride, the polytartaric anhydride, the polyitaconic anhydride and the polymaleic anhydride and at least one of monomers forming the polylactic acid, the polyglycolic acid, the polycaprolactone, the polysuccinate, the poly (beta-hydroxybutyrate), the polyethylene glycol adipate and the polyhydroxybutyrate valerate; the degradable polyamino acid is at least one selected from polylysine, polyaspartic acid, polycysteine, polyminosine, polyornithine, polyglycine and polyglutamic acid; the complexing agent is selected from the group consisting of 8-hydroxyquinoline, 8-hydroxyquinaldine, 4, 5-dihydroxybenzene-1, 3-disulfonic acid sodium, 4- [3, 5-di-hydroxyphenyl-1H-1, 2, 4-triazole ] -benzoic acid, 8-mercaptoquinoline, thioglycolic acid, methyl 5-methyl-2-mercaptobenzoate, ethylenediamine, triethylenetetramine, ethylenediamine tetraacetic acid, tetrasodium ethylenediamine tetraacetate, N' - [5- [ [4- [ [5- (acetylamino) pentyl ] amino ] -1, 4-dioxobutyl ] hydroxylamine ] pentyl ] -N- (5-aminopentyl) -N-hydroxysuccinamide, phenanthroline, bipyridine, porphyrin, porphine, chlorophyll, hemoglobin, 1, 2-dimethyl-3-hydroxy-4-pyridone, 1-nitroso-2-naphthol-6-sulfonic acid, sulfosalicylic acid, 8-hydroxyquinoline-5-sulfonic acid, tripolyphosphoric acid, hexametaphosphoric acid, at least one of polyphosphoric acid, sodium pyrophosphate, sodium hexametaphosphate, ammonium polyphosphate, potassium diethylenetriamine pentamethylene phosphonate, sodium ethylenediamine tetramethylene phosphonate, oxalic acid, tartaric acid, malic acid, succinic acid, oxaloacetic acid, fumaric acid, maleic acid, citric acid, nitrilotriacetic acid, diethylenetriamine pentacarboxylic acid, alginic acid, glutamic acid, aspartic acid, ornithine, lysine, potassium citrate, calcium citrate, glycerol citrate, acetylsalicylic acid, and sulfosalicylamide.
Further, the absorbable implant medical device is a vascular stent, orthopedic implant, gynecological implant, male implant, or respiratory implant.
According to the absorbable implanted medical device, the gel layer is arranged on the surface of the metal substrate and comprises the organic gel and the corrosion promoting substances, the mass ratio of the organic gel material to the corrosion promoting substances is (1.5-5.5): 1, the absorbable medical device can maintain the structural integrity and the sufficient mechanical property in the early period of implantation, and when corrosion degradation is needed in the later period, the gel state of the organic gel is converted into the sol state through artificial external induction to release the corrosion promoting substances, and the corrosion promoting substances can accelerate the corrosion degradation of the medical device. The process can be controlled to start under artificial conditions, and the purpose of controlling the corrosion of medical instruments is achieved.
Drawings
Fig. 1 is a radial cross-sectional view of a first embodiment of a resorbable implantable medical device 100.
Fig. 2 is a radial cross-sectional view of a second embodiment of an implantable resorbable medical device 200.
Fig. 3 is a radial cross-sectional view of a third embodiment of a resorbable implantable medical device 300.
Fig. 4 is a radial cross-sectional view of an absorbable implanted medical device 400 provided in accordance with a fourth embodiment.
Detailed Description
In order to make the aforementioned objects, features and advantages of the present invention comprehensible, embodiments accompanied with figures are described in detail below. In the following description, numerous specific details are set forth in order to provide a thorough understanding of the present invention. This invention may, however, be embodied in many different forms and should not be construed as limited to the embodiments set forth herein, but rather should be construed as broadly as the present invention is capable of modification in various respects, all without departing from the spirit and scope of the present invention.
Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs. The terminology used in the description of the invention herein is for the purpose of describing particular embodiments only and is not intended to be limiting of the invention.
The absorbable implanted medical device mentioned in the embodiments can be a vascular stent, an orthopedic implant, a gynecological implant, a male implant or a respiratory implant.
Referring to fig. 1, an embodiment provides an absorbable implantable medical device 100, the absorbable implantable medical device 100 including a metal substrate 1 and a gel layer 2 disposed on a surface of the metal substrate 1. The metal matrix 1 may be, but not limited to, a degradable metal such as an iron matrix or an alloy.
In the present embodiment, the gel layer 2 comprises an organogel material and an erosion promoting substance. The organic gel material is mPEG 112 -b-PDLAG 12 Or mPEG 45 -b-PDLAG 12 . The preparation method of the organogel material comprises the following steps: dissolving DL-allylglycine in tetrahydrofuran, adding triphosgene to form a mixture, adding alpha-pinene to the mixture, stirring in anhydrous tetrahydrofuran at 50 ℃, continuously flushing the mixture with dry nitrogen, concentrating the crude product, precipitating with hexane, recrystallizing with ethyl acetate-hexane three times, and vacuum drying to obtain DL-allylglycine NCA; mixing mPEG 112 -NH 2 Or mPEG 45 -NH 2 Adding into anhydrous N, N-dimethylformamide solution of DL-allylglycine NCA, stirring in glove box at room temperature under argon atmosphere for 12 hr to form gel, and stirring vigorouslyPrecipitating the gel with diethyl ether, filtering, collecting, and vacuum drying at room temperature to obtain mPEG 112 -b-PDLAG 12 Or mPEG 45 -b-PDLAG 12 。
In one embodiment, the corrosion-promoting substance is selected from at least one of a degradable polyester, a degradable polyanhydride, a degradable polyamino acid, and a complexing agent. The degradable polyester, the degradable polyanhydride, the degradable polyamino acid and the degradation products thereof can ionize hydrogen ions to generate a low pH value environment, so that the corrosion of the metal matrix 1 is promoted. The complexing agent reacts with the metal matrix 1 to form a water-soluble iron complex, which is beneficial to the occurrence of corrosion reaction, thereby promoting the corrosion of the metal matrix 1.
In one embodiment, the degradable polyester is selected from any one or a physical blend of at least two of polylactic acid, polyglycolic acid, polybutylene succinate, poly (beta-hydroxybutyrate), polycaprolactone, polyethylene adipate, polypentanoate, polyhydroxyalkyl alcohol esters, and poly (malate), or a copolymer of at least two of the monomers forming the degradable polyester-based polymer.
In one embodiment, the degradable polyanhydride is selected from at least one of polyanhydride, succinic anhydride, adipic anhydride, sebacic anhydride, dodecanoic anhydride, citric anhydride, malic anhydride, succinic anhydride, tartaric anhydride, itaconic anhydride, and maleic anhydride. Or the degradable polyanhydride is a copolymer formed by copolymerizing at least one of the monomers for forming the polyethylene anhydride, the succinic anhydride, the polyadipic anhydride, the polysebacic anhydride, the polycitric anhydride, the polymalic anhydride, the polysuccinic anhydride, the polytartaric anhydride, the polyitaconic anhydride and the polymaleic anhydride and at least one of the monomers for forming the polylactic acid, the polyglycolic acid, the polycaprolactone, the polybutylene succinate, the poly (beta-hydroxybutyrate), the polyethylene glycol adipate and the polyhydroxybutyrate valerate.
In one embodiment, the degradable polyamino acid is at least one selected from the group consisting of polylysine, polyaspartic acid, polycysteine, polyminosine, polyornithine, and polyglutamic acid.
In one embodiment, the complexing agent is selected from the group consisting of 8-hydroxyquinoline, 8-hydroxyquinaldine, sodium 4, 5-dihydroxybenzene-1, 3-disulfonate, 4- [3, 5-di-hydroxyphenyl-1H-1, 2, 4-triazole ] -benzoic acid, 8-mercaptoquinoline, thioglycolic acid, methyl 5-methyl-2-mercaptobenzoate, ethylenediamine, triethylenetetramine, ethylenediaminetetraacetic acid, tetrasodium ethylenediaminetetraacetate, N' - [5- [ [4- [ [5- (acetylaminohydroxy-amino) pentyl ] amino ] -1, 4-dioxobutyl ] hydroxylamine ] pentyl ] -N- (5-aminopentyl) -N-hydroxysuccinamide, phenanthroline, bipyridine, porphyrin, porphine, chlorophyll, hemoglobin, 1, 2-dimethyl-3-hydroxy-4-pyridone, 1-nitroso-2-naphthol, sodium 1-nitroso-2-naphthol-6-sulfonate, sulfosalicylic acid, 8-hydroxyquinoline-5-sulfonic acid, tripolyphosphoric acid, hexametaphosphoric acid, at least one of polyphosphoric acid, sodium pyrophosphate, sodium hexametaphosphate, ammonium polyphosphate, potassium diethylenetriamine pentamethylene phosphonate, sodium ethylenediamine tetramethylene phosphonate, oxalic acid, tartaric acid, malic acid, succinic acid, oxaloacetic acid, fumaric acid, maleic acid, citric acid, nitrilotriacetic acid, diethylenetriamine pentacarboxylic acid, alginic acid, glutamic acid, aspartic acid, ornithine, lysine, potassium citrate, calcium citrate, glycerol citrate, acetylsalicylic acid, and sulfosalicylamide.
In other embodiments, the corrosion-promoting substance is not limited to the above-listed substances, and any substance that promotes corrosion of the metal substrate 1 and is not toxic or harmful to the human body may be used.
In the natural state (state not induced by the inducing conditions), the gel layer 2 is in a gel state. The organogel material in the gel layer 2 causes the gel layer 2 to transform from a gel state to a sol state under the influence of external conditions, such as ultrasonic conditions (inducing conditions), thereby facilitating the release of the polymer in the gel layer 2. The polymer in the gel layer 2 is released in the sol state, and the released polymer and/or degradation products of the polymer may promote corrosion of the absorbable implantable medical device 100. Specifically, in the later stage of implantation, intervention ultrasound can be performed on the absorbable implanted medical device 100 in the body of a patient at any time, so that the gel layer 2 of the absorbable implanted medical device 100 is changed from a gel state to a sol state, corrosion-promoting substances in the gel layer 2 are released, the corrosion-promoting substances can accelerate corrosion of the metal matrix 1, the structural integrity and sufficient mechanical properties of the metal matrix 1 in the early stage of implantation are ensured, and the corrosion period of the medical device in the later stage of implantation is shortened.
In the embodiment, the mass ratio of the organogel material to the corrosion-promoting substance in the gel layer 2 is (1.5-5.5): 1, so that the release rate of the corrosion-promoting substance is controlled in a reasonable range, and the corrosion-promoting substance is continuously released in vivo after the corrosion is started.
Referring to fig. 2, the second embodiment provides an implantable absorbable medical device 200, where the implantable absorbable medical device 200 adds a drug coating to the implantable absorbable medical device 100. The drug coating comprises a first drug coating 31 and a second drug coating 32, wherein the first drug coating 31 is coated on the surface of the gel layer 2, and the second drug coating 32 is coated on the inner surface of the metal substrate 1.
The drug coating layer comprises an active drug and a polymer, wherein in the drug coating layer (comprising the first drug coating layer 31 and the second drug coating layer 32), the mass ratio between the active drug and the polymer is 1: (2-20) so that the release rate of the drug is controlled within a reasonable range, and the drug is continuously released in the body in the absorbable and implantable medical device 200.
The active drug is at least one selected from drugs for inhibiting vascular proliferation, antiplatelet drugs, antithrombotic drugs, anti-inflammatory drugs and anti-allergy drugs. Wherein the drug for inhibiting the vascular proliferation is at least one of paclitaxel, rapamycin, paclitaxel derivatives and rapamycin derivatives; the antiplatelet drug is cilostazol; the antithrombotic drug is heparin; the anti-inflammatory drug is dexamethasone; the anti-sensitization drug is at least one of calcium gluconate, chlorphenamine and cortisone.
The active agents in the first drug coating 31 and the second drug coating 32 may be the same or different.
In one embodiment, the molecular weight of the polymer in the drug coating is less than 20 ten thousand, allowing the drug release rate to be controlled within a desired range.
In one embodiment, the polymer in the drug coating is selected from at least one of a degradable polyester, a degradable polyanhydride, and a degradable polyamino acid, such that the polymer in the drug coating also acts as a corrosion-promoting agent. The first drug coating 31 and the second drug coating 32 completely cover the metal substrate 1, and a closed system can be formed. Compared with the medicine coating only arranged on the outer surface or the inner surface of the metal matrix 1, the polymer in the medicine coating completely coating the metal matrix 1 has better corrosion-promoting effect on the metal matrix 1, the released polymer is prevented from being washed or diluted by body fluid, and the corrosion-promoting effect is weakened. In other embodiments, the drug coating may be disposed only on the outer surface of the metal substrate 1.
Referring to fig. 3, embodiment three provides an absorbable implantable medical device 300, in which the gel layer 4 is different from the gel layer 2 in embodiment two, compared to the absorbable implantable medical device 200. The gel layer 4 comprises an organic gel material and a corrosion-promoting substance, and also comprises an active medicament, wherein the mass ratio of the organic gel material to the active medicament to the corrosion-promoting substance is (2-5.5): (0.05-0.5): 1, so that the release rate of the active medicament is controlled within a reasonable range, and the active medicament is continuously released in the whole erosion process.
The active drug is at least one selected from drugs for inhibiting angiogenesis, antiplatelet drugs, antithrombotic drugs, anti-inflammatory drugs and anti-allergic drugs. Wherein, the medicine for inhibiting the vascular proliferation is at least one of paclitaxel, rapamycin, paclitaxel derivatives and rapamycin derivatives; the antiplatelet drug is cilostazol; the antithrombotic drug is heparin; the anti-inflammatory drug is dexamethasone; the anti-sensitization drug is at least one of calcium gluconate, chlorphenamine maleate and cortisone.
In this embodiment, the active agent is an anti-inflammatory agent.
The organogel material in the gel layer 4 causes the gel layer 4 to transition from a gel state to a sol state under the influence of external conditions, such as ultrasonic conditions, thereby facilitating the release of erosion promoting substances and anti-inflammatory drugs in the gel layer 4.
Specifically, corrosion of the absorbable implantable medical device 300 is often accompanied by inflammation, and the absorbable implantable medical device 300 in the patient's body can be subjected to interventional ultrasound at any time, and the gel layer 4 is also transformed from a gel state to a sol state, so that the anti-inflammatory drug in the gel layer 4 is released to resist the inflammation. In this embodiment, the anti-inflammatory drug includes dexamethasone, and in other embodiments, without limitation, dexamethasone, any drug that achieves an anti-inflammatory effect may be applied to the gel layer 4.
Referring to fig. 4, in contrast to the implantable absorbable medical device 300, the gel layer of the fourth embodiment includes an outer gel layer 41 and an inner gel layer 42, the outer gel layer 41 covers the outer surface of the metal substrate 1, the inner gel layer 42 covers the inner surface of the metal substrate 1, and the second drug coating layer 32 covers the inner gel layer 42. Wherein, the thickness ratio between the outer gel layer 41 and the inner gel layer 42 is 1.5-4, so the corrosion of the metal matrix 1 can be further accelerated.
When the erosion promoting substances in the outer gel layer 41 and the inner gel layer 42 are degradable polymers, the polymers in the outer gel layer 41, the inner gel layer 42, the first drug coating 31 and the second drug coating 32 may be the same or different.
The outer gel layer 41, the inner gel layer 42, the first drug coating 31 and the second drug coating 32 are all between [2, 33] microns thick, with the total coating thickness being less than 40 microns to accommodate the size of the blood vessel.
In one embodiment, at least the outer gel layer of the outer gel layer and the inner gel layer further comprises an active drug.
In one embodiment, the active agents in the outer and inner gel layers may be the same or different.
The outer gel layer 41 or the inner gel layer 42 is prepared as follows: the corrosion-promoting substance and the active drug are uniformly dissolved in the organogel in a sol state to form a solution, and then the solution is coated on the outer surface or the inner surface of the metal substrate 1, and after standing and cooling for 8 hours or more at room temperature, a gel-state layered structure is formed, thereby obtaining the outer gel layer 41 or the inner gel layer 42.
The first drug coating 31 or the second drug coating 32 is prepared as follows: the polymer and the active drug are dissolved in an organic solvent to form a solution, and then the solution is coated on the metal substrate 1 having the outer gel layer 41 and the inner gel layer 42 and left to stand at room temperature to obtain the first drug coating layer 31 or the second drug coating layer 32. Wherein the organic solvent is at least one selected from chloroform, dichloromethane, ethyl acetate, tetrahydrofuran, acetone, methanol, ethanol, acetonitrile, 1, 4-dioxane, dimethylformamide and isopropanol.
The preparation method adopts the process of spraying, dipping, brushing and electrostatic spinning.
The following examples are further illustrated with respect to the stent.
The scaffolds of examples 1-7 were made of pure iron as the matrix, with a wall thickness of 70 μm and a surface coverage of 12.7%, and the scaffold specifications were(inner diameter after expansion x length after expansion). 4 supports are arranged, and the number of rabbits is 2. The stents were implanted in the abdominal aorta of rabbits, 2 stents each. And (3) taking out 2 stents in one rabbit body at the 3 rd month, and testing the quality loss rate of the stents. Simultaneously in 3 rd month, intervene the supersound to the internal support of another rabbit, in 6 th month, take out test support quality loss rate with 2 internal supports of another rabbit.
The mass of the stent matrix before implantation is M 0 Taking out the implanted stent from the blood vessel at a preset observation time point, soaking the stent in 1mol/L sodium hydroxide solution to digest tissues, then taking out the stent and fragments thereof from the solution, putting the stent and fragments thereof into 3% tartaric acid and/or organic solution for ultrasonic treatment to ensure that corrosion products and polymers on the surface of the stent and a drug layer are completely peeled off or dissolved in the 3% tartaric acid, taking out the rest non-corroded stent matrix or fragments thereof, drying and weighing the rest non-corroded stent matrix or fragments thereof with the mass of M t . The mass loss rate is calculated according to the following formula:
w-mass loss rate
M t Quality of remaining stent matrix after Corrosion
M 0 Initial quality of the stent base
Example 1
Selecting the organogel material to be mPEG 112 -b-PDLAG 12 Dissolving an organogel material in a solvent, controlling the concentration of the organogel material to be 22mg/mL to obtain a sol-state gel solution, then dissolving a corrosion-promoting substance in the gel solution, completely and uniformly coating the sol-state gel solution on the outer surface of a stent matrix, standing and cooling for 8 hours to obtain a gel layer, wherein the thickness of the gel layer is 33 mu m, and thus the absorbable implant stent is formed. Wherein, the solvent adopts dimethylformamide, the corrosion-promoting substance adopts polylactic acid-glycolic acid copolymer with molecular weight of 5000, and the mass ratio of the organogel material to the polylactic acid-glycolic acid copolymer is 5.5. The absorbable implant stent is implanted into a rabbit body, and the mass loss rate is calculated by adopting the method.
The experimental results show that: at month 3, the mass loss rate W was 12.76%; at month 6, the mass loss rate W was 52.12%.
Example 2
Selecting organic gel material as mPEG 45 -b-PDLAG 12 Dissolving an organogel material in a solvent, controlling the concentration of the organogel material to be 22mg/mL to obtain a sol-state gel solution, then dissolving a corrosion-promoting substance in the gel solution, completely and uniformly coating the sol-state gel solution on the outer surface of a stent matrix, standing and cooling for 8 hours to obtain a gel layer, wherein the thickness of the gel layer is 14 mu m, and thus the absorbable implant stent is formed. Wherein, the solvent adopts dimethylformamide, the corrosion-promoting substance adopts polylactic acid-glycolic acid copolymer with molecular weight of 5000, and the mass ratio of the organogel material to the polylactic acid-glycolic acid copolymer is 1.5. Implanting the absorbable implant stent into a rabbit body by the methodThe mass loss rate is calculated.
The experimental results show that: in month 3, the mass loss rate W was 12.16%; at month 6, the mass loss rate W was 55.27%.
Example 3
Selecting the organogel material to be mPEG 112 -b-PDLAG 12 Dissolving an organogel material in a solvent, controlling the concentration of the organogel material to be 22mg/mL to obtain a sol-state gel solution, then dissolving a corrosion-promoting substance in the gel solution, completely and uniformly coating the sol-state gel solution on the outer surface of a stent matrix, standing and cooling for 8 hours to obtain a gel layer, wherein the thickness of the gel layer is 16 mu m. Wherein the solvent adopts dimethylformamide, the corrosion-promoting substance adopts poly-racemic polylactic acid with the molecular weight of 5000, and the mass ratio of the organogel material to the poly-racemic polylactic acid is 2.
Mixing and dissolving 3 ten thousand of poly-racemic polylactic acid with molecular weight and rapamycin in ethyl acetate according to the mass ratio of 2. The absorbable implant stent is implanted into a rabbit body, and the mass loss rate is calculated by adopting the method. The experimental results show that: in month 3, the mass loss rate W was 26.73%; at month 6, the mass loss rate W was 75.33%.
Example 4
Selecting organic gel material as mPEG 45 -b-PDLAG 12 Dissolving an organogel material in a solvent, controlling the concentration of the organogel material to be 22mg/mL to obtain a sol-state gel solution, then dissolving a corrosion-promoting substance and an active drug in the gel solution, completely and uniformly coating the sol-state gel solution on the outer surface of a stent matrix, standing and cooling for 8 hours to obtain a gel layer, wherein the thickness of the gel layer is 14 mu m. Wherein, the solvent adopts dimethylformamide, the corrosion-promoting substance adopts poly-racemic polylactic acid with the molecular weight of 1 ten thousand, the active drug adopts dexamethasone, and the mass ratio of the organogel material, the dexamethasone and the poly-racemic polylactic acid is 2.05.
Mixing and dissolving 3-ten-thousand-molecular-weight poly (racemic polylactic acid) and paclitaxel in ethyl acetate according to the mass ratio of 20. The absorbable implant stent is implanted into a rabbit body, and the mass loss rate is calculated by adopting the method.
The experimental results show that: in month 3, the mass loss rate W is 26.98%; at month 6, the mass loss rate W was 60.37%.
Example 5
Selecting organic gel material as mPEG 112 -b-PDLAG 12 Dissolving an organogel material in a solvent, controlling the concentration of the organogel material to be 22mg/mL to obtain a sol-state gel solution, then dissolving a corrosion-promoting substance and an active drug in the gel solution, coating the sol-state gel solution on the surface of a stent matrix by adopting an asymmetric technology, standing and cooling for 8 hours to obtain an inner gel layer and an outer gel layer, wherein the thickness of the outer gel layer is 18 micrometers, the thickness of the inner gel layer is 12 micrometers, and the thickness ratio between the outer gel layer 41 and the inner gel layer 42 is 1.5. Wherein, the solvent adopts dimethylformamide, the corrosion-promoting substance adopts poly-racemic polylactic acid with the molecular weight of 1 ten thousand, the active drug adopts dexamethasone, and the mass ratio of the organogel material, the dexamethasone and the poly-racemic polylactic acid is 5.5.
Mixing and dissolving 3-ten-thousand molecular weight poly-racemic polylactic acid and rapamycin in ethyl acetate according to the mass ratio of 20. The absorbable implant stent is implanted into a rabbit body, and the mass loss rate is calculated by adopting the method.
The experimental results show that: at month 3, the mass loss rate W was 26.87%; at month 6, the mass loss rate W was 70.28%.
Example 6
Selecting the organogel material to be mPEG 45 -b-PDLAG 12 Dissolving an organogel material in a solvent, controlling the concentration of the organogel material to be 22mg/mL to obtain a sol-state gel solution, then dissolving an erosion promoting substance in the gel solution, completely and uniformly coating the gel solution on the outer surface of a stent matrix, standing and cooling for 8 hours to obtain a gel layer, wherein the thickness of the gel layer is 14 mu m, and thus the absorbable implant stent is formed. Wherein, the solvent adopts dimethylformamide, the corrosion-promoting substance adopts polysuccinic anhydride with the molecular weight of 2 ten thousand, and the mass ratio of the organogel material to the polysuccinic anhydride is 1.5. The absorbable implant stent is implanted into a rabbit body, and the mass loss rate is calculated by adopting the method.
The experimental results show that: at month 3, the mass loss rate W was 12.63%; at month 6, the mass loss rate W was 43.33%.
Example 7
Selecting organic gel material as mPEG 112 -b-PDLAG 12 Dissolving an organogel material in a solvent, controlling the concentration of the organogel material to be 22mg/mL to obtain a sol-state gel solution, then dissolving a corrosion-promoting substance and an active drug in the gel solution, coating the sol-state gel solution on the surface of a stent matrix by adopting an asymmetric technology, standing and cooling for 8 hours to obtain an inner gel layer and an outer gel layer, wherein the thickness of the outer gel layer is 18 microns, the thickness of the inner gel layer is 12 microns, and the thickness ratio between the outer gel layer 41 and the inner gel layer 42 is 1.5. Wherein, the solvent adopts dimethylformamide, the corrosion-promoting substance adopts polycysteine with the molecular weight of 1 ten thousand, the active drug adopts dexamethasone, and the mass ratio of the organogel material, the dexamethasone to the polycysteine is 5.5.
The method comprises the steps of mixing and dissolving 3-ten-thousand molecular weight polycysteine and rapamycin in ethyl acetate according to the mass ratio of 20. The absorbable implant stent is implanted into a rabbit body, and the mass loss rate is calculated by adopting the method.
The experimental results show that: at month 3, the mass loss rate W was 19.56%; at month 6, the mass loss rate W was 57.21%.
Comparative example 1
The absorbable implant stent is a pure iron bare stent, and the mass loss rate is calculated by implanting the absorbable implant stent into a rabbit body by adopting the method.
The experimental results show that: in month 3, the mass loss rate W was 10.23%; at month 6, the mass loss rate W was 19.37%.
The stent mass loss rate W in month 3 was not much different from that in example 1, but was much different from that in month 6, and the stent mass loss rate W in month 6 of example 1 was significantly greater than that in month 6 of comparative example 1, indicating that the gel layer was effective in promoting the corrosion of the stent.
Comparative example 2
The absorbable implant stent is a pure iron matrix. Mixing and dissolving 3 ten thousand of poly-racemic polylactic acid with molecular weight and rapamycin in ethyl acetate according to the mass ratio of 20. The absorbable implant stent is implanted into a rabbit body, and the mass loss rate is calculated by adopting the method.
The experimental results show that: at month 3, the mass loss rate W was 26.15%; at month 6, the mass loss rate W was 33.26%.
The stent mass loss rate W in month 3 was not much different from that in example 3, but the stent mass loss rate W in month 6 was much different, and the stent mass loss rate W in month 6 of example 3 was significantly larger than that in month 6 of comparative example 2, indicating that the gel layer was able to effectively promote the corrosion of the stent.
The scaffolds of examples 8-9 were made of pure iron as the matrix, 70 μm in wall thickness, 12.7% in surface coverage, and the scaffold specifications were4 supports are arranged, and the number of pigs is 2. Each pig was implanted with 2 stents, which were implanted in the Left Circumflex (LCX) or anterior descending (LAD) and Right Coronary Artery (RCA), respectively. At month 3, 2 stents in one pig were removed, and the rate of mass loss of one stent was tested for histopathological observation of the other stent vessel segment. Meanwhile, in the 3 rd month period, interventional ultrasound is carried out on the stent in the other pig body, in the 6 th month period, 2 stents in the other pig body are taken out, one stent tests the quality loss rate of the stent, and histopathological observation is carried out on the other stent and the tissue where the other stent is located.
The mass of the stent matrix before implantation is M 0 Taking out the implanted stent from the blood vessel at a preset observation time point, soaking the stent in 1mol/L sodium hydroxide solution to digest tissues, then taking out the stent and fragments thereof from the solution, putting the stent and fragments thereof into 3 percent tartaric acid for ultrasonic treatment to ensure that corrosion products and polymers on the surface of the stent and drug layers completely fall off or are dissolved in the 3 percent tartaric acid, taking out the residual unetched stent matrix or fragments thereof, drying and weighing the stent matrix or fragments thereof with the mass M t . The mass loss rate is calculated according to the following formula:
w-mass loss rate
M t Quality of remaining stent matrix after Corrosion
M 0 Initial quality of the stent base
In histopathological observation, the inflammation integral calculation mode is as follows:
the inflammation score for each individual stent strut was ranked as follows: 0= no inflammatory cells around the stent rod; 1= mild, non-peripheral lymphocyte infiltration in the support periphery; 2= local, non-peripheral, medium to dense cell aggregates surrounding the stent rod; 3= dense lymphohistiocytic infiltration around the stent rod. Inflammation score per cross section = sum of inflammation scores for individual stent struts divided by number of stent struts, and inflammation score per stent = sum of inflammation scores for individual cross sections divided by number of cross sections. Each stent is taken at least three cross sections, and each cross section is taken at least five stent rods.
Example 8
Selecting organic gel material as mPEG 112 -b-PDLAG 12 Dissolving an organogel material in a solvent, controlling the concentration of the organogel material to be 22mg/mL to obtain a sol-state gel solution, then dissolving a corrosion-promoting substance in the gel solution, completely and uniformly coating the sol-state gel solution on the inner surface and the outer surface of a stent matrix, standing and cooling for 8 hours to obtain an inner gel layer and a first outer gel layer, wherein the thickness of the first outer gel layer and the thickness of the inner gel layer are both 2 micrometers. Wherein, the solvent adopts dimethylformamide, the corrosion-promoting substance adopts poly-racemic polylactic acid with the molecular weight of 1 ten thousand, and the mass ratio of the organogel material to the poly-racemic polylactic acid is 2.
And dissolving a polymer (corrosion-promoting substance) and an active drug in the gel solution, coating the outer surface of the first outer gel layer by adopting a single-side coating technology, standing and cooling for 8 hours to form a second outer gel layer, wherein the first outer gel layer and the second outer gel layer form the outer gel layer, the thickness of the second outer gel layer is 6 microns, and the thickness of the outer gel layer is 8 microns. Wherein, the solvent adopts dimethylformamide, the polymer adopts poly-racemic polylactic acid with the molecular weight of 1 ten thousand, and the active medicament adopts dexamethasone.
The inner gel layer and the outer gel layer are integrated, wherein the mass ratio of the organogel material, dexamethasone and the poly-racemic polylactic acid is 2. The thickness ratio between the outer gel layer 41 and the inner gel layer 42 that is finally formed is 4.
Mixing and dissolving 3-ten-thousand molecular weight poly-racemic polylactic acid and rapamycin in ethyl acetate according to the mass ratio of 20. The absorbable implant stent is implanted into a pig body, and the mass loss rate is calculated and histopathological observation is carried out by adopting the method.
The experimental results show that: in month 3, the mass loss rate W was 27.11%; at month 6, the mass loss rate W was 76.69%, and the inflammation score of the stent was 0.4.
Example 9
Selecting organic gel material as mPEG 112 -b-PDLAG 12 Dissolving an organogel material in a solvent, controlling the concentration of the organogel material to be 22mg/mL to obtain a sol-state gel solution, then dissolving a corrosion-promoting substance and an active drug in the gel solution, coating the sol-state gel solution on the surface of a stent matrix by adopting an asymmetric technology, standing and cooling for 8 hours to obtain an inner gel layer and an outer gel layer, wherein the thickness of the outer gel layer is 8 micrometers, the thickness of the inner gel layer is 2 micrometers, and the thickness ratio between the outer gel layer 41 and the inner gel layer 42 is 4. Wherein, the solvent adopts dimethylformamide, the corrosion-promoting substance adopts acetylsalicylic acid with the molecular weight of 1 ten thousand, the active medicament adopts dexamethasone, and the mass ratio of the organic gel to the dexamethasone to the acetylsalicylic acid is 2.5.
The method comprises the steps of mixing and dissolving acetylsalicylic acid with the molecular weight of 3 million and paclitaxel in ethyl acetate according to the mass ratio of 20. The absorbable implant stent is implanted into a pig body, and the mass loss rate is calculated and histopathological observation is carried out by adopting the method.
The experimental results show that: in month 3, the mass loss rate W is 48.67%; at month 6, the mass loss rate W was 89.21%, and the inflammation score of the stent was 0.6.
Comparative example 3
The absorbable implant stent is a pure iron matrix. Mixing and dissolving 3 ten thousand of poly-dl-lactide with molecular weight and paclitaxel in ethyl acetate according to the mass ratio of 20 to form a mixed solution, and completely and uniformly coating the mixed solution on the inner surface and the outer surface of the stent to form a drug coating, wherein the thickness of the drug coating is 5 mu m, so that the absorbable implant stent is formed. The absorbable implant stent is implanted into a pig body, and the mass loss rate is calculated and histopathological observation is carried out by adopting the method.
The experimental results show that: in month 3, the mass loss rate W was 27.02%; at month 6, the mass loss rate W was 33.16%, and the inflammation score of the stent was 1.2.
The stent mass loss rate W in month 3 was not much different from that in example 8, but the stent mass loss rate W in month 6 was much different, and the stent mass loss rate W in month 6 of example 8 was significantly larger than that in month 6 of comparative example 3, indicating that the gel layer was able to effectively promote the corrosion of the stent. The integral of inflammation for comparative example 3 is significantly greater than that for example 8, indicating that the active drug dexamethasone in the gel layer acts as an anti-inflammatory.
The technical features of the embodiments described above may be arbitrarily combined, and for the sake of brevity, all possible combinations of the technical features in the embodiments described above are not described, but should be considered as being within the scope of the present specification as long as there is no contradiction between the combinations of the technical features.
The above-mentioned embodiments only express several embodiments of the present invention, and the description thereof is specific and detailed, but not to be understood as limiting the scope of the invention. It should be noted that, for a person skilled in the art, several variations and modifications can be made without departing from the inventive concept, which falls within the scope of the present invention. Therefore, the protection scope of the present patent shall be subject to the appended claims.
Claims (10)
1. An absorbable implant medical device comprises a metal substrate, and is characterized in that the absorbable implant medical device also comprises a gel layer covering the surface of the metal substrate, wherein the gel layer comprises an organic gel material and a corrosion-promoting substance, and the mass ratio of the organic gel material to the corrosion-promoting substance is (1.5-5.5): 1; wherein the organogel material satisfies: can be transformed from a gel state to a sol state under external induction; the organogel material is mPEG112-b-PDLAG12 or mPEG45-b-PDLAG12; the molecular weight of the corrosion-promoting substance is 5000-20000.
2. The implantable absorbable medical device of claim 1, wherein said gel layer further comprises an active drug, and the mass ratio of said organogel material, said active drug and said erosion promoting substance is (2-5.5): (0.05-0.5): 1.
3. the resorbable implantable medical device of claim 1, wherein said gel layer comprises an outer gel layer covering an outer surface of said metal substrate and an inner gel layer covering an inner surface of said metal substrate, wherein a thickness ratio between said outer gel layer and said inner gel layer is between 1.5 and 4.
4. The resorbable implantable medical device of claim 3 wherein at least said outer gel layer of said outer gel layer and said inner gel layer further comprises an active drug.
5. The resorbable implantable medical device of claim 1 further comprising a drug coating comprising an active drug and a polymer, wherein a mass ratio between said active drug and said erosion promoting substance is 1: (2-20), the drug coating layer covers at least the outer surface of the gel layer.
6. The resorbable implantable medical device of claim 1 wherein said gel layer has a thickness ranging from 2 to 33 microns.
7. The resorbable implantable medical device of claim 2 wherein said active agent is selected from at least one of a vascular proliferation inhibiting agent selected from at least one of paclitaxel, rapamycin, a paclitaxel derivative, and a rapamycin derivative, an antiplatelet agent, an antithrombotic agent, an anti-inflammatory agent, and an anti-sensitizing agent; the antiplatelet drug is cilostazol; the antithrombotic drug is heparin; the anti-inflammatory drug is dexamethasone; the anti-sensitization medicine is at least one of calcium gluconate, chlorphenamine and cortisone.
8. The resorbable implantable medical device of claim 1 wherein said corrosion-promoting substance is selected from at least one of a degradable polyester, a degradable polyanhydride, a degradable polyamino acid, and a complexing agent.
9. The resorbable implantable medical device of claim 8 wherein said degradable polyester is selected from the group consisting of any one or a physical blend of at least two of polylactic acid, polyglycolic acid, polybutylene succinate, poly (β -hydroxybutyrate), polycaprolactone, polyethylene adipate, polypentanoate, polyhydroxyalkyl alcohol esters, and poly (malate), or a copolymer of at least two of the monomers forming said degradable polyester-based polymer;
the degradable polyanhydride is selected from at least one of polyanhydride, polysuccinic anhydride, polyhexamic anhydride, polysebacic anhydride, polydodecanoic anhydride, polycitric anhydride, polymalic anhydride, polysuccinic anhydride, polytartaric anhydride, polyitaconic anhydride and polymaleic anhydride; or the degradable polyanhydride is a copolymer formed by copolymerizing at least one of monomers forming the polyethylene anhydride, the polysuccinic anhydride, the polyhexamic anhydride, the polysebacic anhydride, the polycitric anhydride, the polymalic anhydride, the polysuccinic anhydride, the polytartaric anhydride, the polyitaconic anhydride and the polymaleic anhydride and at least one of monomers forming the polylactic acid, the polyglycolic acid, the polycaprolactone, the polysuccinate, the poly (beta-hydroxybutyrate), the polyethylene glycol adipate and the polyhydroxybutyrate valerate;
the degradable polyamino acid is at least one selected from polylysine, polyaspartic acid, polycysteine, poly-methionine, poly-ornithine, poly-glycine and poly-glutamic acid;
the complexing agent is selected from the group consisting of 8-hydroxyquinoline, 8-hydroxyquinaldine, 4, 5-dihydroxybenzene-1, 3-disulfonic acid sodium, 4- [3, 5-di-hydroxyphenyl-1H-1, 2, 4-triazole ] -benzoic acid, 8-mercaptoquinoline, thioglycolic acid, methyl 5-methyl-2-mercaptobenzoate, ethylenediamine, triethylenetetramine, ethylenediamine tetraacetic acid, tetrasodium ethylenediamine tetraacetate, N' - [5- [ [4- [ [5- (acetylamino) pentyl ] amino ] -1, 4-dioxobutyl ] hydroxylamine ] pentyl ] -N- (5-aminopentyl) -N-hydroxysuccinamide, phenanthroline, bipyridine, porphyrin, porphine, chlorophyll, hemoglobin, 1, 2-dimethyl-3-hydroxy-4-pyridone, 1-nitroso-2-naphthol-6-sulfonic acid, sulfosalicylic acid, 8-hydroxyquinoline-5-sulfonic acid, tripolyphosphoric acid, hexametaphosphoric acid, at least one of polyphosphoric acid, sodium pyrophosphate, sodium hexametaphosphate, ammonium polyphosphate, potassium diethylenetriamine pentamethylene phosphonate, sodium ethylenediamine tetramethylene phosphonate, oxalic acid, tartaric acid, malic acid, succinic acid, oxaloacetic acid, fumaric acid, maleic acid, citric acid, nitrilotriacetic acid, diethylenetriamine pentacarboxylic acid, alginic acid, glutamic acid, aspartic acid, ornithine, lysine, potassium citrate, calcium citrate, glycerol citrate, acetylsalicylic acid, and sulfosalicylamide.
10. The absorbable implantable medical device of claim 1, wherein the absorbable implantable medical device is a vascular stent, orthopedic implant, gynecological implant, male implant, or respiratory implant.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811625648.2A CN111388154B (en) | 2018-12-28 | 2018-12-28 | Absorbable implant medical device |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811625648.2A CN111388154B (en) | 2018-12-28 | 2018-12-28 | Absorbable implant medical device |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111388154A CN111388154A (en) | 2020-07-10 |
| CN111388154B true CN111388154B (en) | 2023-01-03 |
Family
ID=71410691
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201811625648.2A Active CN111388154B (en) | 2018-12-28 | 2018-12-28 | Absorbable implant medical device |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111388154B (en) |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9114125B2 (en) * | 2008-04-11 | 2015-08-25 | Celonova Biosciences, Inc. | Drug eluting expandable devices |
| JP5984806B2 (en) * | 2010-06-30 | 2016-09-06 | サーモディクス,インコーポレイティド | Lipid coatings for medical devices that deliver bioactive agents |
| EP2407184A3 (en) * | 2010-07-16 | 2014-10-29 | Biotronik AG | Abluminally coated drug-eluting stents having a form-fitting protective layer |
| CN106806938B (en) * | 2015-11-27 | 2020-04-14 | 先健科技(深圳)有限公司 | Absorbable Iron-Based Alloy Implantable Medical Devices |
| CN106902395B (en) * | 2015-12-22 | 2020-04-07 | 先健科技(深圳)有限公司 | Absorbable iron-based alloy implantation medical instrument |
| CN108261570A (en) * | 2016-12-30 | 2018-07-10 | 先健科技(深圳)有限公司 | Absorbable iron-based instrument |
| US11020410B2 (en) * | 2017-02-03 | 2021-06-01 | The Brigham And Women's Hospital, Inc. | Self-assembled gels formed with anti-retroviral drugs, prodrugs thereof, and pharmaceutical uses thereof |
| CN109078195B (en) * | 2018-08-30 | 2021-04-02 | 青岛大学 | Thermosensitive drug-loaded micro-organic gel with shell layer containing graphene quantum dots and core containing magnetic nanoparticles, and preparation method and application thereof |
-
2018
- 2018-12-28 CN CN201811625648.2A patent/CN111388154B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN111388154A (en) | 2020-07-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2009283162B2 (en) | Implantable medical device coatings with biodegradable elastomer and releasable taxane agent | |
| CN104587534A (en) | An absorbable iron-base alloy support | |
| CN103142335B (en) | A kind of thermoplastic degradable fiber woven stent and preparation method thereof | |
| CN106581784B (en) | Absorbable Iron-Based Alloy Implantable Medical Devices | |
| CN102387825A (en) | Implantable medical devices having bioabsorbable primer polymer coatings | |
| US20150004207A1 (en) | Interventional medical device and manufacturing method thereof | |
| CN109939271B (en) | Coating structure of medical biodegradable zinc alloy stent and preparation method thereof | |
| Chen et al. | Assessment of structure integrity, corrosion behavior and microstructure change of AZ31B stent in porcine coronary arteries | |
| Zhang et al. | A self-healing coating with embedding of polyphenols on magnesium: towards enhanced corrosion protection for biodegradable vascular implants | |
| CN111000659B (en) | Biodegradable bile pancreatic duct bracket | |
| EP3563879A1 (en) | Absorbable iron-based instrument | |
| CN111388154B (en) | Absorbable implant medical device | |
| CN109954172A (en) | Absorbable Iron-Based Implantable Devices | |
| TWI794616B (en) | Drug-eluting stent | |
| CN116019985B (en) | Paranasal sinus medicine-carrying bracket and preparation method thereof | |
| CN116966402A (en) | Drug sustained-release medical device and preparation method thereof | |
| CN115569244A (en) | Sandwich type degradable biological material and application thereof | |
| US20220241467A1 (en) | Stent with immediately removeable coating | |
| CN106178139A (en) | A kind of support and preparation method thereof | |
| Li et al. | Preparation and evaluation of PDLLA/sirolimus coatings on fully bioabsorbable drug-eluting stents | |
| CN210301826U (en) | Coating structure of medical biodegradable zinc alloy bracket | |
| CN108261570A (en) | Absorbable iron-based instrument | |
| JPWO2008123108A1 (en) | In-vivo medical device | |
| AU2015218518A1 (en) | Coating II | |
| JP2002085549A (en) | Material for intravascular treatment and appliance for intravascular treatment |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| TA01 | Transfer of patent application right |
Effective date of registration: 20210203 Address after: 518000 4th floor, building 4, Jinxiu Science Park, Wuhe Avenue, Longhua District, Shenzhen City, Guangdong Province Applicant after: Yuanxin Science and Technology (Shenzhen) Co.,Ltd. Address before: 518000 1st-5th Floor of Saiba Research Building, Langshan Second Road, North District of Nanshan High-tech Industrial Park, Shenzhen City, Guangdong Province Applicant before: Lifetech Scientific (Shenzhen) Co.,Ltd. |
|
| TA01 | Transfer of patent application right | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |