CN111362866A - Azabenzene modified organic compound and application thereof - Google Patents
Azabenzene modified organic compound and application thereof Download PDFInfo
- Publication number
- CN111362866A CN111362866A CN201811598707.1A CN201811598707A CN111362866A CN 111362866 A CN111362866 A CN 111362866A CN 201811598707 A CN201811598707 A CN 201811598707A CN 111362866 A CN111362866 A CN 111362866A
- Authority
- CN
- China
- Prior art keywords
- substituted
- unsubstituted
- group
- organic compound
- atom
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/26—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/06—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom containing only hydrogen and carbon atoms in addition to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/06—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom containing only hydrogen and carbon atoms in addition to the ring nitrogen atom
- C07D213/127—Preparation from compounds containing pyridine rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/06—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom containing only hydrogen and carbon atoms in addition to the ring nitrogen atom
- C07D213/16—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom containing only hydrogen and carbon atoms in addition to the ring nitrogen atom containing only one pyridine ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/06—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom containing only hydrogen and carbon atoms in addition to the ring nitrogen atom
- C07D213/22—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom containing only hydrogen and carbon atoms in addition to the ring nitrogen atom containing two or more pyridine rings directly linked together, e.g. bipyridyl
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D251/00—Heterocyclic compounds containing 1,3,5-triazine rings
- C07D251/02—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings
- C07D251/12—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members
- C07D251/14—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hydrogen or carbon atoms directly attached to at least one ring carbon atom
- C07D251/24—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hydrogen or carbon atoms directly attached to at least one ring carbon atom to three ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/10—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/10—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/04—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/10—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/626—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing more than one polycyclic condensed aromatic rings, e.g. bis-anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/654—Aromatic compounds comprising a hetero atom comprising only nitrogen as heteroatom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6574—Polycyclic condensed heteroaromatic hydrocarbons comprising only oxygen in the heteroaromatic polycondensed ring system, e.g. cumarine dyes
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
本发明公开了一种由氮杂苯修饰的有机化合物及其应用,属于半导体技术领域。所述由氮杂苯修饰的有机化合物的结构如通式(I)所示:
本发明还公开了上述有机化合物的应用。本发明的有机化合物具有良好的热稳定性和较高的玻璃化温度,同时具有合适的HOMO能级,采用本发明提供的有机化合物的器件通过结构优化,可有效提升OLED器件的光电性能以及OLED器件的寿命。The invention discloses an organic compound modified by azepine and its application, and belongs to the technical field of semiconductors. The structure of the organic compound modified by the azabenzene is shown in the general formula (I):
The invention also discloses the application of the above organic compound. The organic compound of the present invention has good thermal stability and high glass transition temperature, and at the same time has a suitable HOMO energy level. The device using the organic compound provided by the present invention can effectively improve the optoelectronic performance of the OLED device and the OLED through structural optimization. device lifetime.Description
技术领域technical field
本发明涉及一种由氮杂苯修饰的有机化合物及其应用,属于半导体技术领域。The invention relates to an organic compound modified by azepines and its application, and belongs to the technical field of semiconductors.
背景技术Background technique
有机电致发光器件技术既可以用来制造新型显示产品,也可以用于制作新型照明产品,有望替代现有的液晶显示和荧光灯照明,应用前景十分广泛。OLED发光器件犹如三明治的结构,包括电极材料膜层,以及夹在不同电极膜层之间的有机功能材料,各种不同功能材料根据用途相互叠加在一起共同组成OLED发光器件。作为电流器件,当对OLED发光器件的两端电极施加电压,并通过电场作用有机层功能材料膜层中的正负电荷,正负电荷进一步在发光层中复合,即产生OLED电致发光。Organic electroluminescent device technology can be used not only to manufacture new display products, but also to manufacture new lighting products. The OLED light-emitting device is like a sandwich structure, including electrode material film layers and organic functional materials sandwiched between different electrode film layers. Various functional materials are superimposed on each other according to the purpose to form an OLED light-emitting device. As a current device, when a voltage is applied to the electrodes at both ends of the OLED light-emitting device, the positive and negative charges in the functional material film layer of the organic layer are acted by the electric field, and the positive and negative charges are further combined in the light-emitting layer, that is, OLED electroluminescence is generated.
当前,OLED显示技术已经在智能手机,平板电脑等领域获得应用,进一步还将向电视等大尺寸应用领域扩展,但是,和实际的产品应用要求相比,OLED器件的发光效率,使用寿命等性能还需要进一步提升。对于OLED发光器件提高性能的研究包括:降低器件的驱动电压,提高器件的发光效率,提高器件的使用寿命等。为了实现OLED器件的性能的不断提升,不但需要从OLED器件结构和制作工艺的创新,更需要OLED光电功能材料不断研究和创新,创制出更高性能OLED的功能材料。At present, OLED display technology has been applied in smart phones, tablet computers and other fields, and will be further expanded to large-scale application fields such as TVs. However, compared with the actual product application requirements, the luminous efficiency of OLED devices, service life and other properties Further improvement is needed. Research on improving the performance of OLED light-emitting devices includes: reducing the driving voltage of the device, improving the luminous efficiency of the device, and improving the service life of the device. In order to realize the continuous improvement of the performance of OLED devices, it is not only necessary to innovate the structure and production process of OLED devices, but also to continuously research and innovate OLED optoelectronic functional materials to create functional materials of higher performance OLEDs.
构成OLED器件的OLED光电功能材料膜层至少包括两层以上结构,产业上应用的OLED器件结构,则包括空穴注入层、空穴传输层、电子阻挡层、发光层、空穴阻挡层、电子传输层、电子注入层等多种膜层,也就是说应用于OLED器件的光电功能材料至少包含空穴注入材料,空穴传输材料,发光材料,电子传输材料等,材料类型和搭配形式具有丰富性和多样性的特点。另外,对于不同结构的OLED器件搭配而言,所使用的光电功能材料具有较强的选择性,相同的材料在不同结构器件中的性能表现,也可能完全迥异。The OLED photoelectric functional material film layer constituting the OLED device includes at least two or more layers of structure, and the industrially applied OLED device structure includes a hole injection layer, a hole transport layer, an electron blocking layer, a light emitting layer, a hole blocking layer, an electron Transport layer, electron injection layer and other film layers, that is to say, the optoelectronic functional materials applied to OLED devices include at least hole injection materials, hole transport materials, light-emitting materials, electron transport materials, etc., and the material types and matching forms are rich. characteristics of sexuality and diversity. In addition, for the collocation of OLED devices with different structures, the optoelectronic functional materials used have strong selectivity, and the performance of the same material in devices with different structures may also be completely different.
因此,针对当前OLED器件的产业应用要求,以及OLED器件的不同功能膜层,器件的光电特性需求,必须选择更适合,具有高性能的OLED功能材料或材料组合,才能实现器件的高效率、长寿命和低电压的综合特性。就当前OLED显示照明产业的实际需求而言,目前OLED材料的发展还远远不够,落后于面板制造企业的要求,作为材料企业开发更高性能的有机功能材料显得尤为重要。Therefore, according to the current industrial application requirements of OLED devices, as well as the different functional film layers of OLED devices, the optoelectronic characteristics of the device, it is necessary to choose more suitable, high-performance OLED functional materials or material combinations, in order to achieve high efficiency and long-term performance of the device. Combined characteristics of longevity and low voltage. As far as the actual needs of the current OLED display lighting industry are concerned, the development of OLED materials is far from enough, and it lags behind the requirements of panel manufacturing companies. It is particularly important for material companies to develop higher-performance organic functional materials.
发明内容SUMMARY OF THE INVENTION
本发明的目的之一,是提供一种由氮杂苯修饰的有机化合物。本发明的有机化合物具有良好的热稳定性和较高的玻璃化温度,同时具有合适的HOMO能级,采用本发明提供的有机化合物的器件通过结构优化,可有效提升OLED器件的光电性能以及OLED器件的寿命。One of the objects of the present invention is to provide an organic compound modified by azepines. The organic compound of the present invention has good thermal stability and high glass transition temperature, and at the same time has a suitable HOMO energy level. The device using the organic compound provided by the present invention can effectively improve the optoelectronic performance of the OLED device and the OLED through structural optimization. device lifetime.
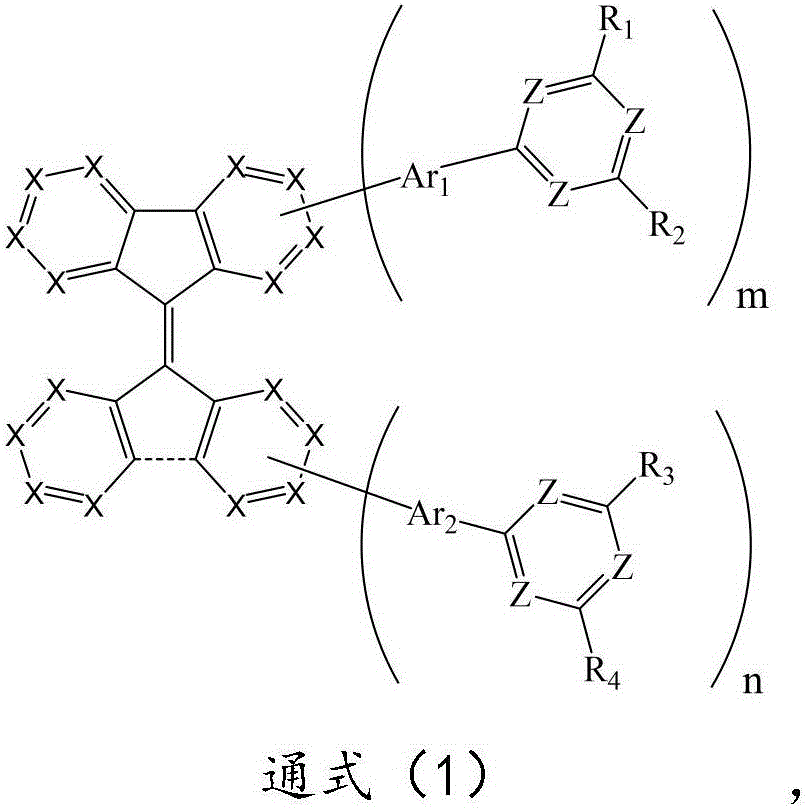
本发明解决上述技术问题的技术方案如下:一种由氮杂苯修饰的有机化合物,该有机化合物的结构如通式(1)所示:The technical scheme that the present invention solves the above-mentioned technical problem is as follows: a kind of organic compound modified by azepine, the structure of this organic compound is shown as general formula (1):
通式(1)中,-----表示为两个基团以单键连接或不连接;In the general formula (1), ----- represents that two groups are connected or not connected by a single bond;
m、n分别独立的表示为数字0或1,且m+n≥1;m and n are independently represented as
Z表示为氮原子或C-H,且至少有一个Z表示为氮原子;Z is represented as nitrogen atom or C-H, and at least one Z is represented as nitrogen atom;
所述X表示为氮原子或C(R0);连接位点处的X表示为碳原子;The X is represented as a nitrogen atom or C(R 0 ); the X at the attachment site is represented as a carbon atom;
Ar1、Ar2分别独立地表示为单键、经取代或未取代的C6-C30亚芳基、含有一个或多个杂原子的经取代或未取代的5至30元亚杂芳基中的一种;Ar 1 and Ar 2 are each independently represented as a single bond, a substituted or unsubstituted C 6 -C 30 arylene group, a substituted or unsubstituted 5- to 30-membered heteroarylene group containing one or more heteroatoms one of the
R1、R2、R3、R4分别独立地表示为经取代或未取代的C6-C30芳基、含有一个或多个杂原子的经取代或未取代的5至30元杂芳基中的一种;R 1 , R 2 , R 3 , R 4 are each independently represented as a substituted or unsubstituted C 6 -C 30 aryl group, a substituted or unsubstituted 5- to 30-membered heteroaryl group containing one or more heteroatoms one of the bases;
R0表示为氢原子、氕、氘、氚、卤素原子、氰基、C1-C20的烷基、经取代或未取代的C6-C30芳基、含有一个或多个杂原子的经取代或未取代的5至30元杂芳基中的一种;R 0 represents hydrogen atom, protium, deuterium, tritium, halogen atom, cyano group, C 1 -C 20 alkyl group, substituted or unsubstituted C 6 -C 30 aryl group, containing one or more heteroatoms One of substituted or unsubstituted 5- to 30-membered heteroaryl groups;
上述可被取代基团的取代基任选自氕、氘、氚、氰基、卤素、C1-C20的烷基、C6-C30芳基、含有一个或多个杂原子的5至30元杂芳基中的一种或多种;The substituents of the above-mentioned substitutable groups are optionally selected from protium, deuterium, tritium, cyano, halogen, C 1 -C 20 alkyl, C 6 -C 30 aryl, 5 to
所述杂芳基中的杂原子任选自氮、氧或硫原子中的一种或多种。The heteroatoms in the heteroaryl group are optionally selected from one or more of nitrogen, oxygen or sulfur atoms.
本发明的化合物中π共轭效应使得其有很强的空穴传输能力,高的空穴传输速率能够降低器件的起始电压,提高有机电致发光器件的效率;且其中氮杂苯的引入增加了分子的不对称性,能够降低分子的结晶性,降低分子的平面性,阻止分子在平面上移动,从而提高分子的热稳定性;同时,本发明提供的化合物的结构使得电子和空穴在发光层的分布更加平衡,在恰当的HOMO能级下,提升了空穴注入和传输性能;在合适的LUMO能级下,又起到了电子阻挡的作用,提升激子在发光层中的复合效率。The π-conjugation effect in the compound of the present invention makes it have strong hole transport ability, and the high hole transport rate can reduce the initial voltage of the device and improve the efficiency of the organic electroluminescence device; and the introduction of azabenzene The asymmetry of the molecule is increased, the crystallinity of the molecule can be reduced, the planarity of the molecule can be reduced, and the movement of the molecule on the plane can be prevented, thereby improving the thermal stability of the molecule; at the same time, the structure of the compound provided by the present invention enables electrons and holes. The distribution of the light-emitting layer is more balanced. Under the appropriate HOMO energy level, the hole injection and transport performance is improved; under the appropriate LUMO energy level, it also plays the role of electron blocking and improves the recombination of excitons in the light-emitting layer. efficiency.
在上述技术方案的基础上,本发明还可以做如下改进。On the basis of the above technical solutions, the present invention can also be improved as follows.
进一步,当m、n同时为1,所述通式(1)中至少有两个Z表示为氮原子,且为氮的Z不都位于同一个氮杂苯上。Further, when m and n are 1 at the same time, at least two Zs in the general formula (1) are represented as nitrogen atoms, and the Zs that are nitrogen are not all located on the same azabenzene.
进一步,m+n=1,所述通式(1)中X表示为氮原子的个数为0、1或2。Further, m+n=1, and X in the general formula (1) represents that the number of nitrogen atoms is 0, 1 or 2.
进一步,m+n=1,所述通式(1)中,Z表示为氮原子的个数为1、2或3。Further, m+n=1, and in the general formula (1), Z represents that the number of nitrogen atoms is 1, 2 or 3.
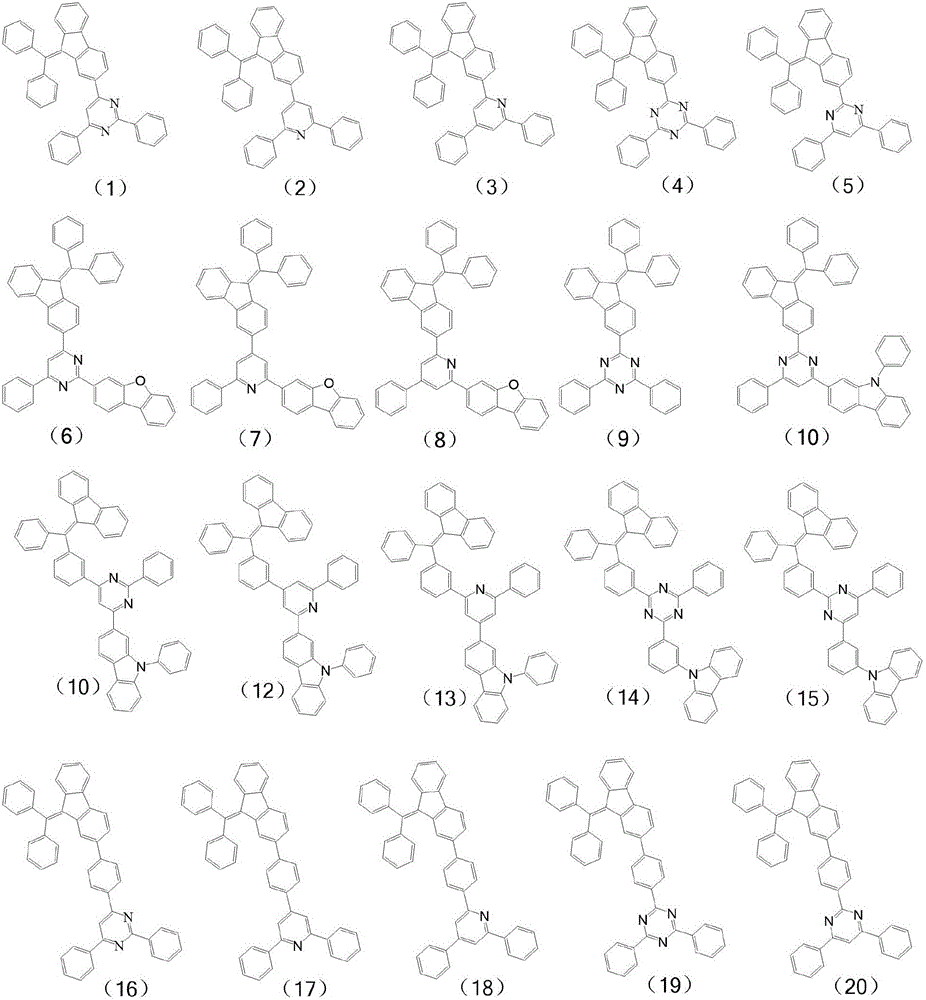
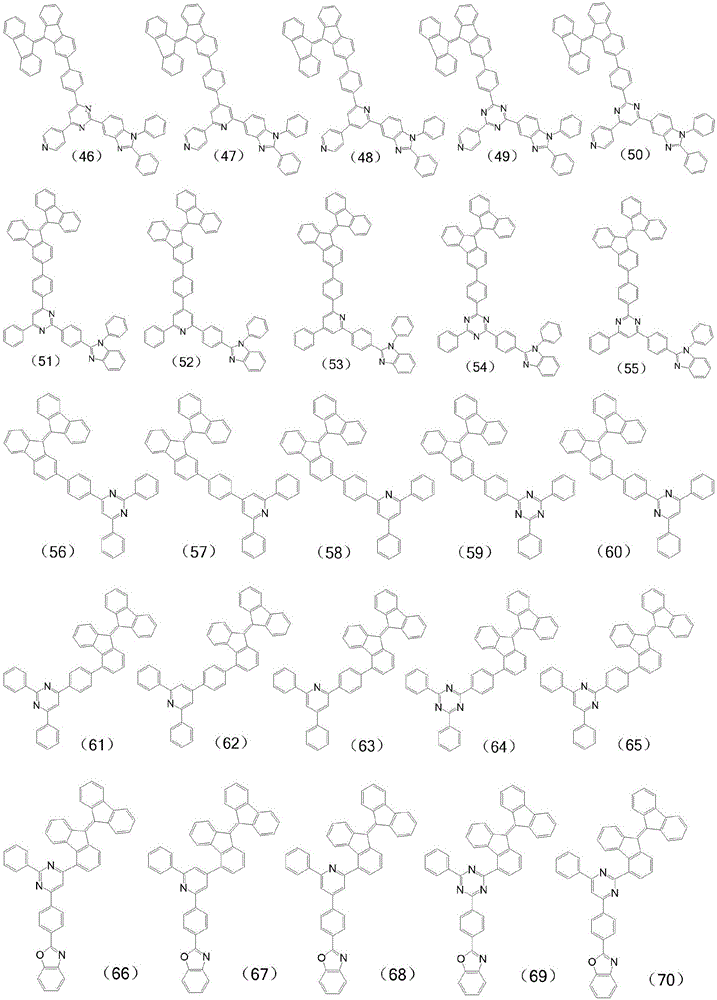
进一步,所述有机化合物可表示为下列通式(2)-通式(5)所示结构中的一种:Further, the organic compound can be represented as one of the structures represented by the following general formula (2)-general formula (5):
其中使用的符号和标记具有上述给出的含义。The symbols and signs used therein have the meanings given above.
进一步,所述Ar1、Ar2分别独立地表示为经取代或未取代的亚苯基、经取代或未取代的亚萘基、经取代或未取代的亚吡啶基、经取代或未取代的亚二联苯基、经取代或未取代的亚三联苯基、经取代或未取代的亚萘啶基、经取代或未取代的亚咔唑基、经取代或未取代的亚二苯并呋喃基、经取代或未取代的亚蒽基、经取代或未取代的亚嘧啶基、经取代或未取代的亚菲基、取代或未取代的亚苯并噁唑、取代或未取代的亚苯并咪唑、取代或未取代的亚苯并噻唑中的一种;Further, the Ar 1 and Ar 2 are respectively independently represented as substituted or unsubstituted phenylene, substituted or unsubstituted naphthylene, substituted or unsubstituted pyridylene, substituted or unsubstituted Diphenylene, substituted or unsubstituted terphenylene, substituted or unsubstituted naphthyridinylene, substituted or unsubstituted carbazolylylene, substituted or unsubstituted dibenzofuran group, substituted or unsubstituted anthracylene, substituted or unsubstituted pyrimidylene, substituted or unsubstituted phenanthrene, substituted or unsubstituted benzoxazole, substituted or unsubstituted phenylene One of imidazole, substituted or unsubstituted benzothiazole;
所述R1、R2、R3、R4分别独立地表示为经取代或未取代的苯基、经取代或未取代的萘基、经取代或未取代的萘啶基、经取代或未取代的二联苯基、经取代或未取代的三联苯基、经取代或未取代的咔唑基、经取代或未取代的二苯并呋喃基、经取代或未取代的蒽基、经取代或未取代的菲基、经取代或未取代的二苯并噻吩基、经取代或未取代的吡啶基、经取代或未取代的嘧啶基、经取代或未取代的苯并咪唑基、经取代或未取代的苯并噁唑基、经取代或未取代的苯并噻唑基、经取代或未取代的喹喔啉基、经取代或未取代的喹啉基;Said R 1 , R 2 , R 3 , R 4 are each independently represented as substituted or unsubstituted phenyl, substituted or unsubstituted naphthyl, substituted or unsubstituted naphthyridinyl, substituted or unsubstituted Substituted biphenyl, substituted or unsubstituted terphenyl, substituted or unsubstituted carbazolyl, substituted or unsubstituted dibenzofuranyl, substituted or unsubstituted anthracenyl, substituted or unsubstituted or unsubstituted phenanthryl, substituted or unsubstituted dibenzothienyl, substituted or unsubstituted pyridyl, substituted or unsubstituted pyrimidinyl, substituted or unsubstituted benzimidazolyl, substituted or unsubstituted or unsubstituted benzoxazolyl, substituted or unsubstituted benzothiazolyl, substituted or unsubstituted quinoxalinyl, substituted or unsubstituted quinolinyl;
所述R0表示为氢原子、氕、氘、氚、氰基、氟原子、甲基、乙基、丙基、异丙基、丁基、叔丁基、戊基、己基、经取代或未取代的苯基、经取代或未取代的萘基、经取代或未取代的萘啶基、经取代或未取代的二联苯基、经取代或未取代的三联苯基、经取代或未取代的咔唑基、经取代或未取代的二苯并呋喃基;The R 0 represents hydrogen atom, protium, deuterium, tritium, cyano, fluorine, methyl, ethyl, propyl, isopropyl, butyl, tert-butyl, pentyl, hexyl, substituted or unsubstituted Substituted phenyl, substituted or unsubstituted naphthyl, substituted or unsubstituted naphthyridinyl, substituted or unsubstituted biphenyl, substituted or unsubstituted terphenyl, substituted or unsubstituted carbazolyl, substituted or unsubstituted dibenzofuranyl;
所述可取代基团的取代基任选自氕、氘、氚、氰基、氟原子、甲基、乙基、丙基、异丙基、丁基、叔丁基、戊基、己基、苯基、萘基、萘啶基、吡啶基、二联苯基、三联苯基、呋喃基、咔唑基或噻吩基。The substituent of the substitutable group is optionally selected from protium, deuterium, tritium, cyano, fluorine, methyl, ethyl, propyl, isopropyl, butyl, tert-butyl, pentyl, hexyl, benzene group, naphthyl, naphthyl, pyridyl, biphenyl, terphenyl, furyl, carbazolyl or thienyl.
进一步,所述有机化合物的具体结构式为:Further, the specific structural formula of the organic compound is:
中的任意一种。any of the .
本发明的目的之二,是提供一种有机电致发光器件。本发明的化合物在OLED发光器件中具有良好的应用效果,具有良好的产业化前景。Another object of the present invention is to provide an organic electroluminescent device. The compounds of the present invention have good application effects in OLED light-emitting devices, and have good industrialization prospects.
本发明解决上述技术问题的技术方案如下:一种有机电致发光器件,所述有机电致发光器件的功能层含有上述的由氮杂苯修饰的有机化合物。The technical solution of the present invention to solve the above technical problem is as follows: an organic electroluminescence device, wherein the functional layer of the organic electroluminescence device contains the above-mentioned organic compound modified by azepine.
在上述技术方案的基础上,本发明还可以做如下改进。On the basis of the above technical solutions, the present invention can also be improved as follows.
进一步,包含发光层和/或电子阻挡层和/或空穴传输层,所述发光层和/或电子阻挡层和/或空穴传输层含有上述的由氮杂苯修饰的有机化合物。Further, it includes a light-emitting layer and/or an electron blocking layer and/or a hole transport layer, and the light-emitting layer and/or the electron blocking layer and/or the hole transport layer contains the above-mentioned organic compound modified with azepine.
本发明的目的之三,是提供一种照明或显示元件。本发明的有机电致发光器件可以应用在显示原件,使器件的电流效率,功率效率和外量子效率均得到很大改善;同时,对于器件寿命提升非常明显,在OLED发光器件中具有良好的应用效果,具有良好的产业化前景。The third object of the present invention is to provide an illumination or display element. The organic electroluminescence device of the invention can be applied to display elements, so that the current efficiency, power efficiency and external quantum efficiency of the device are greatly improved; meanwhile, the life of the device is significantly improved, and it has a good application in OLED light-emitting devices. It has good industrialization prospects.
本发明解决上述技术问题的技术方案如下:一种照明或显示元件,包括上述的有机电致发光器件。The technical solutions of the present invention to solve the above-mentioned technical problems are as follows: an illumination or display element, comprising the above-mentioned organic electroluminescence device.
本发明的有益效果是:The beneficial effects of the present invention are:
1.本发明提供的化合物中π共轭效应使得其有很强的空穴传输能力,高的空穴传输速率能够降低器件的起始电压,提高有机电致发光器件的效率;且其中氮杂苯的引入增加了分子的不对称性,能够降低分子的结晶性,降低分子的平面性,阻止分子在平面上移动,从而提高分子的热稳定性;同时,本发明提供的化合物的结构使得电子和空穴在发光层的分布更加平衡,在恰当的HOMO能级下,提升了空穴注入和传输性能;在合适的LUMO能级下,又起到了电子阻挡的作用,提升激子在发光层中的复合效率。1. The π-conjugation effect in the compound provided by the present invention makes it have strong hole transport ability, and the high hole transport rate can reduce the initial voltage of the device and improve the efficiency of the organic electroluminescence device; The introduction of benzene increases the asymmetry of the molecule, which can reduce the crystallinity of the molecule, reduce the planarity of the molecule, and prevent the molecule from moving on the plane, thereby improving the thermal stability of the molecule; at the same time, the structure of the compound provided by the present invention makes the electron The distribution of holes and holes in the light-emitting layer is more balanced. Under the appropriate HOMO energy level, the hole injection and transport performance is improved; under the appropriate LUMO energy level, it plays the role of electron blocking and improves the excitons in the light-emitting layer. compound efficiency in .
2.氮杂苯上的取代基,使得分子间的距离变大,分子间相互作用力减弱,所以具有了较低的蒸镀温度,从而使得材料的工业加工窗口变宽。2. The substituents on the azabenzene make the intermolecular distance larger and the intermolecular interaction force weakened, so it has a lower evaporation temperature, thereby broadening the industrial processing window of the material.
3.本发明的化合物在OLED器件应用时,通过器件结构优化,可保持高的膜层稳定性,可有效提升OLED器件的光电性能以及OLED器件的寿命。本发明化合物在OLED发光器件中具有良好的应用效果和产业化前景。3. When the compound of the present invention is applied to an OLED device, by optimizing the device structure, it can maintain high film layer stability, and can effectively improve the optoelectronic performance of the OLED device and the lifespan of the OLED device. The compound of the present invention has good application effect and industrialization prospect in the OLED light-emitting device.
附图说明Description of drawings
图1为本发明所列举的材料应用于OLED器件的结构示意图,其中,各标号所代表的部件如下:FIG. 1 is a schematic structural diagram of the materials enumerated in the present invention applied to an OLED device, wherein the components represented by each reference number are as follows:
其中,1、基板层,2、ITO阳极层,3、空穴注入层,4、空穴传输层,5、电子阻挡层,6、发光层,7、电子传输层或空穴阻挡层,8、电子注入层,9、阴极反射电极层。Among them, 1. Substrate layer, 2. ITO anode layer, 3. Hole injection layer, 4. Hole transport layer, 5. Electron blocking layer, 6. Light-emitting layer, 7. Electron transport layer or hole blocking layer, 8 , electron injection layer, 9, cathode reflective electrode layer.
图2为本发明制备的器件和对比器件在不同温度下测量的效率曲线图。FIG. 2 is a graph showing the efficiency curves of the device prepared by the present invention and the comparative device measured at different temperatures.
具体实施方式Detailed ways
以下结合附图对本发明的原理和特征进行描述,所举实例只用于解释本发明,并非用于限定本发明的范围。The principles and features of the present invention will be described below with reference to the accompanying drawings. The examples are only used to explain the present invention, but not to limit the scope of the present invention.
反应原料J-9的合成:Synthesis of reaction raw material J-9:
在300mL冰醋酸中加入15mmol原料M-9,加热回流,加入55mmol锌粉,继续加热回流反应20-30min,将反应液在硅藻土上过滤,加入与滤液等体积的蒸馏水进行沉淀,所得粗产品用正己烷重结晶纯化,即得到中间体S-9,收率:72%。Add 15mmol of raw material M-9 to 300mL of glacial acetic acid, heat under reflux, add 55mmol of zinc powder, continue to heat under reflux for 20-30min, filter the reaction solution on diatomaceous earth, add the same volume of distilled water as the filtrate for precipitation, the obtained crude The product was purified by recrystallization with n-hexane to obtain intermediate S-9, yield: 72%.
取8mmol中间体S-9加入80mmol的三溴化磷,将混合物加热到160℃反应50min,冷却至室温,倒入50倍于反应液体积的水中,加入固态碳酸氢钠调节pH值直至中性,然后用二氯甲烷萃取,旋蒸浓缩萃取液,所得粗产物用甲醇重结晶纯化,即得到中间体K-9,收率:58%。Take 8mmol of intermediate S-9, add 80mmol of phosphorus tribromide, heat the mixture to 160°C for 50min, cool to room temperature, pour into 50 times the volume of the reaction solution, add solid sodium bicarbonate to adjust pH until neutral , and then extracted with dichloromethane, the extract was concentrated by rotary evaporation, and the obtained crude product was purified by recrystallization from methanol to obtain intermediate K-9, yield: 58%.
在三口烧瓶中,通入氮气保护下,将1.2mmol的中间体K-9和1.3mmol的原料N-9溶解到250mL的乙腈中,加热到60℃,加入3mmol的二氮杂二环,反应3h后过滤,用60℃的乙腈冲洗滤饼,合并滤液和洗涤液之后旋蒸浓缩,所得粗产品用甲苯重结晶纯化,得到中间体J-9,收率:56%。In a three-necked flask, under the protection of nitrogen, 1.2 mmol of intermediate K-9 and 1.3 mmol of raw material N-9 were dissolved in 250 mL of acetonitrile, heated to 60 ° C, and 3 mmol of diazabicyclo was added to react. After 3 hours, filter, rinse the filter cake with acetonitrile at 60°C, combine the filtrate and washing liquid, and then concentrate by rotary evaporation. The obtained crude product is purified by recrystallization from toluene to obtain intermediate J-9, yield: 56%.
元素分析结构(分子式C25H16BrN):理论值C,73.18;H,3.93;Br,19.47;N,3.41;测试值:C,73.15;H,3.95;Br,19.48;N,3.43。ESI-MS(m/z)(M+):理论值为409.05,实测值为409.02。Elemental analysis structure (molecular formula C 25 H 16 BrN): theoretical C, 73.18; H, 3.93; Br, 19.47; N, 3.41; found: C, 73.15; H, 3.95; Br, 19.48; N, 3.43. ESI-MS (m/z) (M + ): theoretical value 409.05, found value 409.02.
反应原料J-10的合成:详细反应条件参考原料J-9的合成。Synthesis of raw material J-10: For detailed reaction conditions, refer to the synthesis of raw material J-9.
结构表征:元素分析结构(分子式C24H15BrN2):理论值C,70.09;H,3.68;Br,19.43;N,6.81;测试值:C,70.05;H,3.64;Br,19.40;N,6.83。ESI-MS(m/z)(M+):理论值为410.04,实测值为410.06。Structural Characterization: Elemental Analysis Structure (molecular formula C 24 H 15 BrN 2 ): Theoretical C, 70.09; H, 3.68; Br, 19.43; N, 6.81; Tested: C, 70.05; H, 3.64; Br, 19.40; N , 6.83. ESI-MS (m/z) (M + ): theoretical value 410.04, found value 410.06.
反应原料J-11的合成:详细反应条件参考原料J-9的合成。Synthesis of raw material J-11: For detailed reaction conditions, refer to the synthesis of raw material J-9.
结构表征:元素分析结构(分子式C25H14BrN):理论值C,73.54;H,3.46;Br,19.57;N,3.43;测试值:C,73.55;H,3.45;Br,19.54;N,3.40。ESI-MS(m/z)(M+):理论值为407.03,实测值为407.05。Structural characterization: Elemental analysis Structure (molecular formula C 25 H 14 BrN): theoretical value C, 73.54; H, 3.46; Br, 19.57; N, 3.43; measured value: C, 73.55; H, 3.45; Br, 19.54; N, 3.40. ESI-MS (m/z) (M + ): theoretical value 407.03, found value 407.05.
中间体I的制备:Preparation of Intermediate I:
在250mL三口瓶中,通入氮气,加入0.04mol原料A-1,150mL的THF,0.05mol原料B-1,0.0004mol四(三苯基膦)钯,搅拌均匀,然后加入0.06mol的K2CO3水溶液(2M),加热至80℃,回流反应10小时,取样点板,反应完全,自然冷却,用200mL二氯甲烷萃取,分层,萃取液用无水硫酸钠干燥,过滤,滤液旋蒸,过硅胶柱纯化,得到中间体C-1;HPLC纯度99.6%,收率80.4%。元素分析结构(分子式C9H5Cl2N3):理论值C,47.82;H,2.23;Cl,31.36;N,18.59;测试值:C,47.81;H,2.23;Cl,31.36;N,18.60。ESI-MS(m/z)(M+):理论值为224.99,实测值为225.10。In a 250mL three-necked flask, nitrogen was introduced, 0.04mol of raw material A-1, 150mL of THF, 0.05mol of raw material B- 1 , 0.0004mol of tetrakis(triphenylphosphine) palladium were added, stirred well, and then 0.06mol of K was added. CO 3 aqueous solution (2M), heated to 80°C, refluxed for 10 hours, sampling point plate, the reaction was complete, naturally cooled, extracted with 200 mL of dichloromethane, separated into layers, the extract was dried with anhydrous sodium sulfate, filtered, and the filtrate was spun It was evaporated and purified by silica gel column to obtain intermediate C-1; the HPLC purity was 99.6%, and the yield was 80.4%. Elemental analysis structure (molecular formula C 9 H 5 Cl 2 N 3 ): Theoretical C, 47.82; H, 2.23; Cl, 31.36; N, 18.59; found: C, 47.81; H, 2.23; Cl, 31.36; N, 18.60. ESI-MS (m/z) (M + ): theoretical value 224.99, found value 225.10.
在250mL三口瓶中,通入氮气,加入0.02mol中间体C-1,120mL的THF,0.025mol原料D-1,0.0002mol四(三苯基膦)钯,搅拌均匀,然后加入0.03mol的K2CO3水溶液(2M),加热至80℃,回流反应10小时,取样点板,反应完全,自然冷却,用200mL二氯甲烷萃取,分层,萃取液用无水硫酸钠干燥,过滤,滤液旋蒸,过硅胶柱纯化,得到中间体E-1;HPLC纯度99.1%,收率67.3%。元素分析结构(分子式C15H10ClN3):理论值C,67.30;H,3.77;Cl,13.24;N,15.70;测试值:C,67.31;H,3.75;Cl,13.25;N,15.71。ESI-MS(m/z)(M+):理论值为267.06,实测值为267.05。In a 250 mL three-necked flask, nitrogen was introduced, 0.02 mol of intermediate C-1, 120 mL of THF, 0.025 mol of raw material D-1, 0.0002 mol of tetrakis(triphenylphosphine) palladium were added, stirred well, and then 0.03 mol of K was added. 2 CO 3 aqueous solution (2M), heated to 80 ° C, refluxed for 10 hours, sampling point plate, the reaction was complete, naturally cooled, extracted with 200 mL of dichloromethane, separated into layers, the extract was dried with anhydrous sodium sulfate, filtered, and the filtrate was Rotary-evaporated and purified by silica gel column to obtain intermediate E-1; the HPLC purity is 99.1%, and the yield is 67.3%. Elemental analysis structure (molecular formula C 15 H 10 ClN 3 ): theoretical C, 67.30; H, 3.77; Cl, 13.24; N, 15.70; found: C, 67.31; H, 3.75; Cl, 13.25; N, 15.71. ESI-MS (m/z) (M + ): theoretical value 267.06, found value 267.05.
氮气氛围下,称取0.01mol原料F-1溶解于45ml四氢呋喃中,降温至-78℃,缓慢滴入含有0.02mol正丁基锂的环己烷溶液,滴加完毕后,保温搅拌30分钟;缓慢滴加含有0.035mol硼酸三甲酯的四氢呋喃溶液,滴加完毕后,缓慢升温至室温,保温反应10小时;反应结束后,降温至0℃,缓慢滴入蒸馏水,待无气体产生后,搅拌1小时,然后升至室温;反应液用150ml乙酸乙酯萃取,萃取液用150ml饱和食盐水分三次进行洗涤,最后无水硫酸镁干燥,溶液进行减压蒸馏,将得到的固体用400ml甲苯:乙醇=3:1的混合液进行重结晶,最后得到中间体G-1;HPLC纯度94.7%,收率76.8%。元素分析结构(分子式C7H5BClNO):理论值:C,42.60;H,2.55;B,5.48;N,7.10;O,24.32;测试值:C,42.59;H,2.54;B,5.46;N,7.11;O,24.30;ESI-MS(m/z)(M+):理论值为197.01,实测值为197.03。Under a nitrogen atmosphere, weigh 0.01mol of raw material F-1 and dissolve it in 45ml of tetrahydrofuran, cool to -78°C, slowly drop in a cyclohexane solution containing 0.02mol of n-butyllithium, and keep stirring for 30 minutes after the addition is complete; Slowly add the tetrahydrofuran solution containing 0.035mol trimethyl borate dropwise, after the dropwise addition, slowly heat up to room temperature, and keep the reaction for 10 hours; after the reaction is completed, cool down to 0 °C, slowly add distilled water, and stir after no gas is generated. 1 hour, then warmed to room temperature; the reaction solution was extracted with 150 ml of ethyl acetate, the extract was washed three times with 150 ml of saturated brine, and finally dried over anhydrous magnesium sulfate, the solution was distilled under reduced pressure, and the obtained solid was washed with 400 ml of toluene:ethanol The mixed solution of =3:1 was recrystallized, and finally intermediate G-1 was obtained; the HPLC purity was 94.7%, and the yield was 76.8%. Elemental analysis structure (molecular formula C7H5BClNO): Theoretical value: C, 42.60; H, 2.55; B, 5.48; N, 7.10; O, 24.32; Test value: C, 42.59; H, 2.54; B, 5.46; N, 7.11; O, 24.30; ESI-MS (m/z) (M+): theoretical value 197.01, found value 197.03.
在250mL三口瓶中,通入氮气,加入0.02mol中间体E-1,150ml的THF,0.025mol中间体G-1,0.0002mol四(三苯基膦)钯,搅拌均匀,然后加入0.03mol的K2CO3水溶液(2M),加热至80℃,回流反应10小时,取样点板,反应完全,自然冷却,用200ml二氯甲烷萃取,分层,萃取液用无水硫酸钠干燥,过滤,滤液旋蒸,过硅胶柱纯化,得到中间体H-1;HPLC纯度99.2%,收率67.1%。元素分析结构(分子式C22H13ClN4O):理论值C,68.67;H,3.41;Cl,9.21;N,14.56;O,4.16;测试值:C,68.66;H,3.40;Cl,9.23;N,14.55;O,4.14;ESI-MS(m/z)(M+):理论值为384.08,实测值为384.05。In a 250mL three-necked flask, nitrogen was introduced, 0.02mol of intermediate E-1, 150ml of THF, 0.025mol of intermediate G-1, 0.0002mol of tetrakis(triphenylphosphine) palladium were added, stirred evenly, and then 0.03mol of K2CO3 aqueous solution (2M), heated to 80 ℃, refluxed for 10 hours, sampling point plate, the reaction was complete, naturally cooled, extracted with 200ml of dichloromethane, layered, the extract was dried with anhydrous sodium sulfate, filtered, and the filtrate was rotary evaporated , purified by silica gel column to obtain intermediate H-1; HPLC purity 99.2%, yield 67.1%. Elemental analysis structure (molecular formula C22H13ClN4O): theoretical value C, 68.67; H, 3.41; Cl, 9.21; N, 14.56; O, 4.16; measured value: C, 68.66; H, 3.40; Cl, 9.23; , 4.14; ESI-MS(m/z)(M+): The theoretical value is 384.08, and the measured value is 384.05.
在250mL三口瓶中,通入氮气,加入0.02mol中间体H-1溶解于150ml四氢呋喃中,再将0.024mol双(频哪醇根基)二硼、0.0002mol(1,1’-双(二苯基膦)二茂铁)二氯钯(II)以及0.05mol乙酸钾,搅拌均匀,将上述反应物的混合溶液于反应温度80℃下,加热回流5小时,反应结束后,冷却,将混合物过滤,滤液放入真空烘箱中干燥,然后过硅胶柱分离纯化,得到中间体I-1;HPLC纯度99.6%,收率91.2%。元素分析结构(分子式C28H25BN4O3):理论值C,70.60;H,5.29;B,2.27;N,11.76;O,10.08;测试值:C,70.61;H,5.26;B,2.25;N,11.75;O,10.06;。ESI-MS(m/z)(M+):理论值为476.20,实测值为476.22。In a 250mL three-necked flask, nitrogen was introduced, 0.02mol of intermediate H-1 was added and dissolved in 150ml of tetrahydrofuran, and then 0.024mol of bis(pinacol radical) diboron, 0.0002mol of (1,1'-bis(diphenyl) phosphine) ferrocene) dichloropalladium (II) and 0.05 mol potassium acetate, stir evenly, heat the mixed solution of the above reactants at a reaction temperature of 80 ° C for 5 hours under reflux, after the reaction is completed, cool and filter the mixture , the filtrate was dried in a vacuum oven, and then separated and purified by a silica gel column to obtain intermediate I-1; the HPLC purity was 99.6%, and the yield was 91.2%. Elemental analysis structure (molecular formula C28H25BN4O3): theoretical value C, 70.60; H, 5.29; B, 2.27; N, 11.76; O, 10.08; test value: C, 70.61; H, 5.26; B, 2.25; N, 11.75; O , 10.06;. ESI-MS (m/z) (M+): theoretical value 476.20, found value 476.22.
以中间体I-1的合成方法制备中间体I,涉及到的各种原料的具体结构如表1所示。为了便于,同一种物质在不同的反应步骤中出现用了不同的代号表示。Intermediate I was prepared by the synthesis method of intermediate I-1, and the specific structures of various raw materials involved are shown in Table 1. For convenience, the same substance appears in different reaction steps with different codes.
表1Table 1
实施例1Example 1
在250mL三口瓶中,通入氮气,加入0.01mol原料J-1,150mL的THF,0.015mol中间体I-1,0.0001mol四(三苯基膦)钯,搅拌均匀,然后加入0.02mol的K2CO3水溶液(2M),加热至80℃,回流反应15小时,取样点板,反应完全,自然冷却,用200mL二氯甲烷萃取,分层,萃取液用无水硫酸钠干燥,过滤,滤液旋蒸,用石油醚做淋洗剂过硅胶柱纯化,得到目标化合物1;HPLC纯度99.1%,收率76.8%。元素分析结构(分子式C48H30N4O):理论值C,84.93;H,4.45;N,8.25;O,2.36;测试值:C,84.91;H,4.46;N,8.23;O,2.35。ESI-MS(m/z)(M+):理论值为678.24,实测值为678.25。In a 250 mL three-necked flask, nitrogen was introduced, 0.01 mol of raw material J-1, 150 mL of THF, 0.015 mol of intermediate I-1, 0.0001 mol of tetrakis(triphenylphosphine) palladium were added, stirred well, and then 0.02 mol of K was added. 2 CO 3 aqueous solution (2M), heated to 80 °C, refluxed for 15 hours, sampling point plate, the reaction was complete, cooled naturally, extracted with 200 mL of dichloromethane, separated into layers, the extract was dried with anhydrous sodium sulfate, filtered, and the filtrate was Rotary evaporation, purified by silica gel column using petroleum ether as eluent to obtain the target compound 1; the HPLC purity is 99.1%, and the yield is 76.8%. Elemental analysis structure (molecular formula C 48 H 30 N 4 O): Theoretical C, 84.93; H, 4.45; N, 8.25; O, 2.36; Tested: C, 84.91; H, 4.46; N, 8.23; O, 2.35 . ESI-MS (m/z) (M + ): theoretical value 678.24, found value 678.25.
实施例2Example 2
按化合物93的合成方法制备化合物1,不同之处在于用用中间体I-2代替中间体I-1,所得目标化合物1纯度99.3%,收率78.1%。元素分析结构(分子式C42H28N2):理论值C,89.97;H,5.03;N,5.00;测试值:C,89.96;H,5.05;N,5.01。ESI-MS(m/z)(M+):理论值为560.23,实测值为560.21。Compound 1 was prepared according to the synthesis method of compound 93, except that intermediate I-2 was used instead of intermediate I-1, and the obtained target compound 1 had a purity of 99.3% and a yield of 78.1%. Elemental analysis structure (molecular formula C 42 H 28 N 2 ): Theoretical C, 89.97; H, 5.03; N, 5.00; Tested: C, 89.96; H, 5.05; N, 5.01. ESI-MS (m/z) (M + ): theoretical value 560.23, found value 560.21.
实施例3Example 3
按化合物93的合成方法制备化合物4,不同之处在于用中间体I-3代替中间体I-1,所得目标化合物4纯度99.2%,收率78.5%。元素分析结构(分子式C41H27N3):理论值C,87.67;H,4.85;N,7.48;测试值:C,87.65;H,4.86;N,7.46。ESI-MS(m/z)(M+):理论值为561.22,实测值为561.23。
实施例4Example 4
按化合物93的合成方法制备化合物33,不同之处在于用原料J-2代替原料J-1,用中间体I-4代替中间体I-1,所得目标化合物33纯度99.1%,收率80.3%。元素分析结构(分子式C41H25N3):理论值C,87.99;H,4.50;N,7.51;测试值:C,87.98;H,4.51;N,7.52。ESI-MS(m/z)(M+):理论值为559.20,实测值为559.22。Compound 33 was prepared according to the synthesis method of compound 93, except that starting material J-2 was used instead of starting material J-1, and intermediate I-4 was used instead of intermediate I-1. The obtained target compound 33 had a purity of 99.1% and a yield of 80.3%. . Elemental analysis structure (molecular formula C 41 H 25 N 3 ): Theoretical C, 87.99; H, 4.50; N, 7.51; found: C, 87.98; H, 4.51; N, 7.52. ESI-MS (m/z) (M + ): theoretical value 559.20, found value 559.22.
实施例5Example 5
按化合物93的合成方法制备化合物34,不同之处在于用原料J-8代替原料J-1,用中间体I-5代替中间体I-1,所得目标化合物34纯度99.4%,收率78.4%。元素分析结构(分子式C43H31N5):理论值C,83.60;H,5.06;N,11.34;测试值:C,83.57;H,5.03;N,11.36。ESI-MS(m/z)(M+):理论值为617.26,实测值为617.28。The compound 34 was prepared according to the synthetic method of compound 93, except that the starting material J-8 was used instead of the starting material J-1, and the intermediate I-5 was used instead of the intermediate I-1. The obtained target compound 34 had a purity of 99.4% and a yield of 78.4%. . Elemental analysis structure (molecular formula C 43 H 31 N 5 ): Theoretical C, 83.60; H, 5.06; N, 11.34; found: C, 83.57; H, 5.03; N, 11.36. ESI-MS (m/z) (M + ): theoretical value 617.26, found value 617.28.
实施例6Example 6
按化合物93的合成方法制备化合物58,不同之处在于用原料J-2代替原料J-1,用中间体I-6代替中间体I-1,所得目标化合物58纯度99.3%,收率72.7%。元素分析结构(分子式C49H31N):理论值C,92.86;H,4.93;N,2.21;测试值:C,92.85;H,4.95;N,2.20。ESI-MS(m/z)(M+):理论值为633.25,实测值为633.23。The compound 58 was prepared according to the synthetic method of compound 93, except that the starting material J-2 was used instead of the starting material J-1, and the intermediate I-6 was used instead of the intermediate I-1. The obtained target compound 58 had a purity of 99.3% and a yield of 72.7%. . Elemental analysis structure (molecular formula C 49 H 31 N): Theoretical C, 92.86; H, 4.93; N, 2.21; found: C, 92.85; H, 4.95; N, 2.20. ESI-MS (m/z) (M + ): theoretical value 633.25, found value 633.23.
实施例7Example 7
按化合物93的合成方法制备化合物59,不同之处在于用原料J-2代替原料J-1,用中间体I-7代替中间体I-1,所得目标化合物59纯度98.9%,收率76.8%。元素分析结构(分子式C47H29N3):理论值C,88.79;H,4.60;N,6.61;测试值:C,88.77;H,4.62;N,6.63。ESI-MS(m/z)(M+):理论值为635.24,实测值为635.22。The compound 59 was prepared according to the synthesis method of compound 93, except that the starting material J-2 was used instead of the starting material J-1, and the intermediate I-7 was used instead of the intermediate I-1. The obtained target compound 59 had a purity of 98.9% and a yield of 76.8%. . Elemental analysis structure (molecular formula C 47 H 29 N 3 ): Theoretical C, 88.79; H, 4.60; N, 6.61; found: C, 88.77; H, 4.62; N, 6.63. ESI-MS (m/z) (M + ): theoretical value 635.24, found value 635.22.
实施例8Example 8
按化合物93的合成方法制备化合物75,不同之处在于用原料J-3代替原料J-1,用中间体I-3代替中间体I-1,所得目标化合物75纯度98.7%,收率79.3%。元素分析结构(分子式C56H34N6):理论值C,85.04;H,4.33;N,10.63;测试值:C,85.05;H,4.35;N,10.65。ESI-MS(m/z)(M+):理论值为790.28,实测值为790.26。The compound 75 was prepared according to the synthesis method of compound 93, except that the starting material J-3 was used instead of the starting material J-1, and the intermediate I-3 was used instead of the intermediate I-1. The obtained target compound 75 had a purity of 98.7% and a yield of 79.3%. . Elemental analysis structure (molecular formula C 56 H 34 N 6 ): theoretical C, 85.04; H, 4.33; N, 10.63; found: C, 85.05; H, 4.35; N, 10.65. ESI-MS (m/z) (M + ): theoretical value 790.28, found value 790.26.
实施例9Example 9
按化合物93的合成方法制备化合物79,不同之处在于用原料J-2代替原料J-1,用中间体I-8代替中间体I-1,所得目标化合物79纯度99.2%,收率78.3%。元素分析结构(分子式C50H30N4):理论值C,87.44;H,4.40;N,8.19;测试值:C,87.41;H,4.42;N,8.15。ESI-MS(m/z)(M+):理论值为686.25,实测值为686.27。The compound 79 was prepared according to the synthetic method of compound 93, except that the starting material J-2 was used instead of the starting material J-1, and the intermediate I-8 was used instead of the intermediate I-1. The obtained target compound 79 had a purity of 99.2% and a yield of 78.3%. . Elemental analysis structure (molecular formula C 50 H 30 N 4 ): Theoretical C, 87.44; H, 4.40; N, 8.19; found: C, 87.41; H, 4.42; N, 8.15. ESI-MS (m/z) (M + ): theoretical value 686.25, found value 686.27.
实施例10Example 10
按化合物93的合成方法制备化合物84,不同之处在于用原料J-4代替原料J-1,用中间体I-9代替中间体I-1,所得目标化合物84纯度99.3%,收率83.3%。元素分析结构(分子式C51H37N3):理论值C,88.54;H,5.39;N,6.07;测试值:C,88.55;H,5.39;N,6.08。ESI-MS(m/z)(M+):理论值为691.30,实测值为691.32。Compound 84 was prepared according to the synthesis method of compound 93, except that starting material J-4 was used instead of starting material J-1, and intermediate I-9 was used instead of intermediate I-1. The obtained target compound 84 had a purity of 99.3% and a yield of 83.3%. . Elemental analysis structure (molecular formula C 51 H 37 N 3 ): Theoretical C, 88.54; H, 5.39; N, 6.07; found: C, 88.55; H, 5.39; N, 6.08. ESI-MS (m/z) (M + ): theoretical value 691.30, found value 691.32.
实施例11Example 11
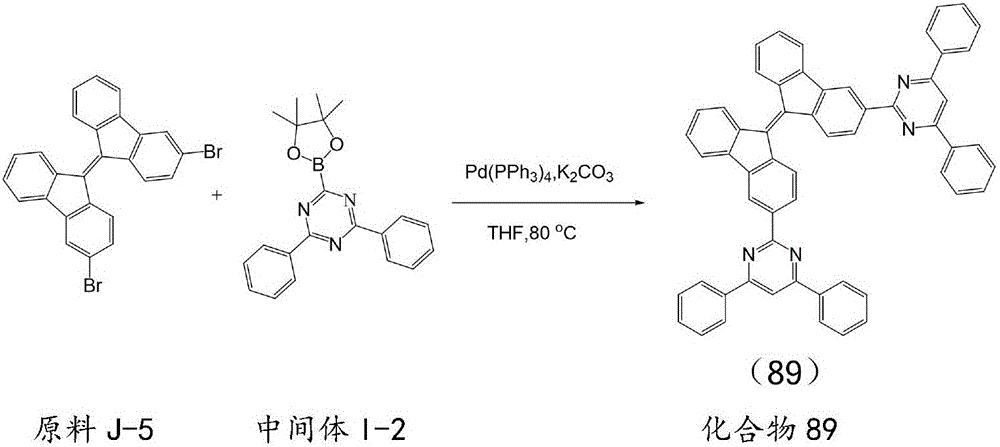
按化合物93的合成方法制备化合物89,不同之处在于用原料J-5代替原料J-1,用中间体I-2代替中间体I-1,所得目标化合物89纯度98.6%,收率81.5%。元素分析结构(分子式C58H36N4):理论值C,88.30;H,4.60;N,7.10;测试值:C,88.28;H,4.63;N,7.12。ESI-MS(m/z)(M+):理论值为788.29,实测值为788.30。The compound 89 was prepared according to the synthetic method of compound 93, except that the starting material J-5 was used instead of the starting material J-1, and the intermediate I-2 was used instead of the intermediate I-1. The obtained target compound 89 had a purity of 98.6% and a yield of 81.5%. . Elemental analysis structure (molecular formula C 58 H 36 N 4 ): Theoretical C, 88.30; H, 4.60; N, 7.10; found: C, 88.28; H, 4.63; N, 7.12. ESI-MS (m/z) (M + ): theoretical value 788.29, found value 788.30.
实施例12Example 12
按化合物93的合成方法制备化合物99,不同之处在于用原料J-9代替原料J-1,用中间体I-10代替中间体I-1,所得目标化合物99纯度99.4%,收率81.3%。元素分析结构(分子式C47H30N6):理论值C,83.16;H,4.45;N,12.38;测试值:C,83.18;H,4.43;N,12.39。ESI-MS(m/z)(M+):理论值为678.25,实测值为678.23。Compound 99 was prepared according to the synthesis method of compound 93, except that the raw material J-1 was replaced by the raw material J-9, and the intermediate I-1 was replaced by the intermediate I-10. The obtained target compound 99 had a purity of 99.4% and a yield of 81.3%. . Elemental analysis structure (molecular formula C 47 H 30 N 6 ): Theoretical C, 83.16; H, 4.45; N, 12.38; found: C, 83.18; H, 4.43; N, 12.39. ESI-MS (m/z) (M + ): theoretical value 678.25, found value 678.23.
实施例13Example 13
按化合物93的合成方法制备化合物144,不同之处在于用中间体I-11代替中间体I-1,所得目标化合物144纯度98.9%,收率78.7%。元素分析结构(分子式C50H32N4):理论值C,87.18;H,4.68;N,8.13;测试值:C,87.16;H,4.66;N,8.15。ESI-MS(m/z)(M+):理论值为688.26,实测值为688.25。Compound 144 was prepared according to the synthetic method of compound 93, except that intermediate I-11 was used instead of intermediate I-1. The obtained target compound 144 had a purity of 98.9% and a yield of 78.7%. Elemental analysis structure (molecular formula C 50 H 32 N 4 ): Theoretical C, 87.18; H, 4.68; N, 8.13; found: C, 87.16; H, 4.66; N, 8.15. ESI-MS (m/z) (M + ): theoretical value 688.26, found value 688.25.
实施例14Example 14
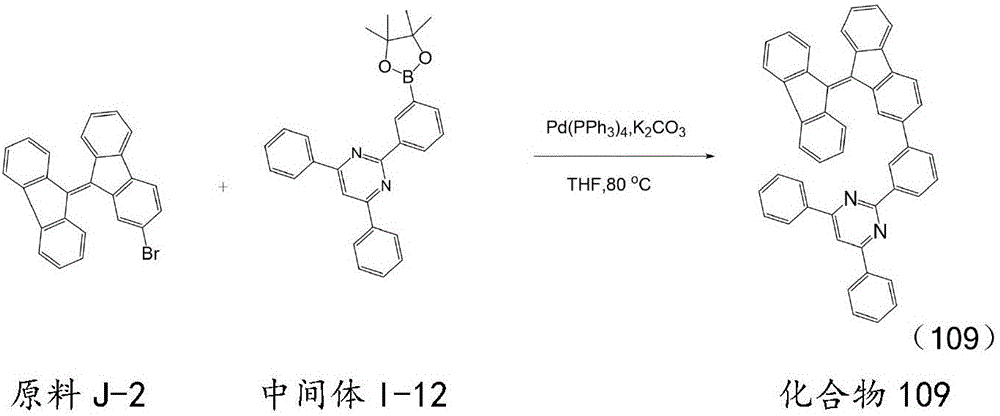
按化合物93的合成方法制备化合物109,不同之处在于用原料J-2代替原料J-1,用中间体I-12代替中间体I-1,所得目标化合物109纯度98.9%,收率80.4%。元素分析结构(分子式C48H30N2):理论值C,90.82;H,4.76;N,4.41;测试值:C,90.83;H,4.77;N,4.43。ESI-MS(m/z)(M+):理论值为634.24,实测值为634.25。Compound 109 was prepared according to the synthesis method of compound 93, except that starting material J-2 was used instead of starting material J-1, and intermediate I-12 was used instead of intermediate I-1. The obtained target compound 109 had a purity of 98.9% and a yield of 80.4%. . Elemental analysis structure (molecular formula C 48 H 30 N 2 ): Theoretical C, 90.82; H, 4.76; N, 4.41; found: C, 90.83; H, 4.77; N, 4.43. ESI-MS (m/z) (M + ): theoretical value 634.24, found value 634.25.
实施例15Example 15
按化合物93的合成方法制备化合物114,不同之处在于用原料J-4代替原料J-1,用中间体I-13代替中间体I-1,所得目标化合物114纯度99.3%,收率80.1%。元素分析结构(分子式C61H36N6O):理论值C,84.31;H,4.18;N,9.67;O,1.84;测试值:C,84.33;H,4.16;N,9.65;O,1.85。ESI-MS(m/z)(M+):理论值为868.30,实测值为868.33。Compound 114 was prepared according to the synthesis method of compound 93, except that starting material J-4 was used instead of starting material J-1, and intermediate I-13 was used instead of intermediate I-1. The obtained target compound 114 had a purity of 99.3% and a yield of 80.1%. . Elemental analysis structure (molecular formula C 61 H 36 N 6 O): Theoretical C, 84.31; H, 4.18; N, 9.67; O, 1.84; Tested: C, 84.33; H, 4.16; N, 9.65; O, 1.85 . ESI-MS (m/z) (M + ): theoretical value 868.30, found value 868.33.
实施例16Example 16
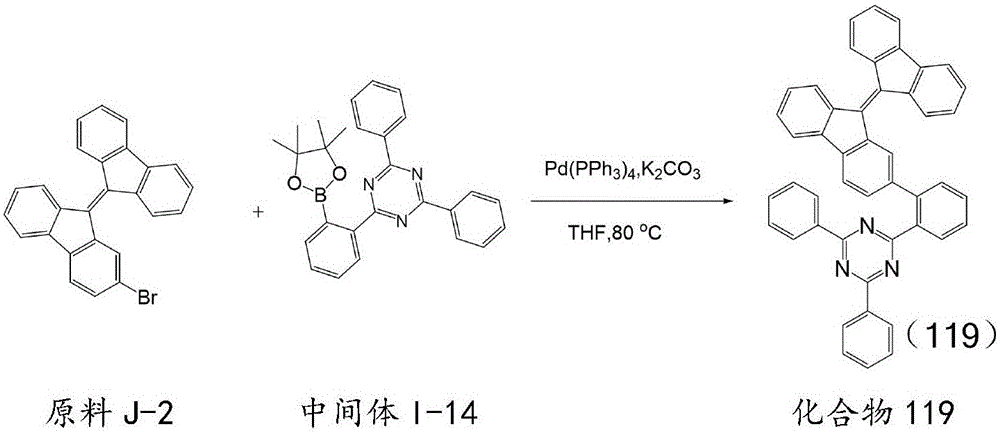
按化合物93的合成方法制备化合物119,不同之处在于用原料J-2代替原料J-1,用中间体I-14代替中间体I-1,所得目标化合物119纯度98.5%,收率74.8%。元素分析结构(分子式C47H29N3):理论值C,88.79;H,4.60;N,6.61;测试值:C,88.81;H,4.61;N,6.60。ESI-MS(m/z)(M+):理论值为635.24,实测值为635.25。The compound 119 was prepared according to the synthetic method of compound 93, except that the starting material J-2 was used instead of the starting material J-1, and the intermediate I-14 was used instead of the intermediate I-1. The obtained target compound 119 had a purity of 98.5% and a yield of 74.8%. . Elemental analysis structure (molecular formula C 47 H 29 N 3 ): Theoretical C, 88.79; H, 4.60; N, 6.61; found: C, 88.81; H, 4.61; N, 6.60. ESI-MS (m/z) (M + ): theoretical value 635.24, found value 635.25.
实施例17Example 17
按化合物93的合成方法制备化合物124,不同之处在于用原料J-7代替原料J-1,用中间体I-11代替中间体I-1,所得目标化合物124纯度98.4%,收率75.9%。元素分析结构(分子式C51H31N3):理论值C,89.32;H,4.56;N,6.13;测试值:C,89.30;H,4.33;N,6.15。ESI-MS(m/z)(M+):理论值为685.25,实测值为685.27。The compound 124 was prepared according to the synthetic method of compound 93, except that the starting material J-7 was used instead of the starting material J-1, and the intermediate I-11 was used instead of the intermediate I-1. The obtained target compound 124 had a purity of 98.4% and a yield of 75.9%. . Elemental analysis structure (molecular formula C 51 H 31 N 3 ): Theoretical C, 89.32; H, 4.56; N, 6.13; found: C, 89.30; H, 4.33; N, 6.15. ESI-MS (m/z) (M + ): theoretical value 685.25, found value 685.27.
实施例18Example 18
按化合物93的合成方法制备化合物154,不同之处在于用原料J-2代替原料J-1,用中间体I-12代替中间体I-1,所得目标化合物154纯度98.6%,收率78.1%。元素分析结构(分子式C45H27N5):理论值C,84.75;H,4.27;N,10.98;测试值:C,84.76;H,4.26;N,10.99。ESI-MS(m/z)(M+):理论值为637.23,实测值为637.25。Compound 154 was prepared according to the synthesis method of compound 93, except that starting material J-2 was used instead of starting material J-1, and intermediate I-12 was used instead of intermediate I-1. The obtained target compound 154 had a purity of 98.6% and a yield of 78.1%. . Elemental analysis structure (molecular formula C 45 H 27 N 5 ): Theoretical C, 84.75; H, 4.27; N, 10.98; found: C, 84.76; H, 4.26; N, 10.99. ESI-MS (m/z) (M + ): theoretical value 637.23, found value 637.25.
实施例19Example 19
按化合物93的合成方法制备化合物169,不同之处在于用原料J-10代替原料J-1,用中间体I-13代替中间体I-1,所得目标化合物169纯度98.6%,收率76.7%。元素分析结构(分子式C55H36N6O):理论值C,82.89;H,4.55;N,10.55;O,2.01;测试值:C,82.87;H,4.52;N,10.53;O,2.03。ESI-MS(m/z)(M+):理论值为796.30,实测值为796.32。The compound 169 was prepared according to the synthesis method of compound 93, except that the starting material J-10 was used instead of the starting material J-1, and the intermediate I-13 was used instead of the intermediate I-1. The obtained target compound 169 had a purity of 98.6% and a yield of 76.7%. . Elemental analysis structure (molecular formula C 55 H 36 N 6 O): Theoretical C, 82.89; H, 4.55; N, 10.55; O, 2.01; Tested: C, 82.87; H, 4.52; N, 10.53; O, 2.03 . ESI-MS (m/z) (M + ): theoretical value 796.30, found value 796.32.
实施例20Example 20
按化合物93的合成方法制备化合物174,不同之处在于用原料J-2代替原料J-1,用中间体I-14代替中间体I-1,所得目标化合物174纯度98.1%,收率78.3%。元素分析结构(分子式C55H36N4O):理论值C,85.91;H,4.72;N,7.29;O,2.08;测试值:C,85.90;H,4.71;N,7.28;O,2.08。ESI-MS(m/z)(M+):理论值为768.29,实测值为768.30。Compound 174 was prepared according to the synthetic method of compound 93, except that starting material J-2 was used instead of starting material J-1, and intermediate I-14 was used instead of intermediate I-1. The obtained target compound 174 had a purity of 98.1% and a yield of 78.3%. . Elemental analysis structure (molecular formula C 55 H 36 N 4 O): Theoretical C, 85.91; H, 4.72; N, 7.29; O, 2.08; Tested: C, 85.90; H, 4.71; N, 7.28; O, 2.08 . ESI-MS (m/z) (M + ): theoretical value 768.29, found value 768.30.
实施例21Example 21
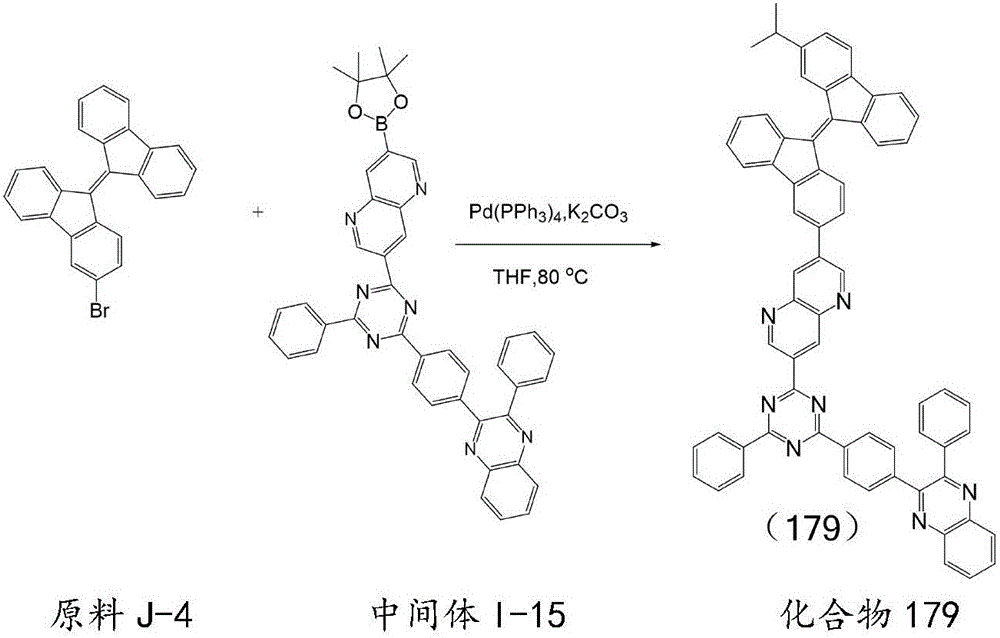
按化合物93的合成方法制备化合物179,不同之处在于用原料J-4代替原料J-1,用中间体I-15代替中间体I-1,所得目标化合物179纯度99.2%,收率77.5%。元素分析结构(分子式C66H43N7):理论值C,84.86;H,4.64;N,10.50;测试值:C,84.85;H,4.65;N,10.52。ESI-MS(m/z)(M+):理论值为933.36,实测值为933.38。Compound 179 was prepared according to the synthesis method of compound 93, except that starting material J-4 was used instead of starting material J-1, and intermediate I-15 was used instead of intermediate I-1. The obtained target compound 179 had a purity of 99.2% and a yield of 77.5%. . Elemental analysis structure (molecular formula C 66 H 43 N 7 ): Theoretical C, 84.86; H, 4.64; N, 10.50; found: C, 84.85; H, 4.65; N, 10.52. ESI-MS (m/z) (M + ): theoretical value 933.36, found value 933.38.
实施例22Example 22
按化合物93的合成方法制备化合物184,不同之处在于用原料J-11代替原料J-1,用中间体I-16代替中间体I-1,所得目标化合物184纯度98.5%,收率76.3%。元素分析结构(分子式C51H28N10O):理论值C,78.45;H,3.61;N,17.94;测试值:C,78.44;H,3.60;N,17.95。ESI-MS(m/z)(M+):理论值为780.25,实测值为780.27。The compound 184 was prepared according to the synthesis method of compound 93, except that the starting material J-1 was replaced by the starting material J-11, and the intermediate I-1 was replaced by the intermediate I-16. The obtained target compound 184 had a purity of 98.5% and a yield of 76.3%. . Elemental analysis structure (molecular formula C 51 H 28 N 10 O): Theoretical C, 78.45; H, 3.61; N, 17.94; found: C, 78.44; H, 3.60; N, 17.95. ESI-MS (m/z) (M + ): theoretical value 780.25, found value 780.27.
本发明化合物在发光器件中使用,可以作为发光层材料使用,也可以作为空穴阻挡/电子传输层材料使用。对本发明上述实施例制备的化合物分别进行热性能、T1能级、HOMO能级的测试,检测结果如表2所示:The compounds of the present invention are used in light-emitting devices, can be used as materials for light-emitting layers, and can also be used as materials for hole-blocking/electron-transporting layers. The compounds prepared in the above-mentioned embodiments of the present invention are respectively tested for thermal properties, T1 energy level, and HOMO energy level, and the detection results are shown in Table 2:
表2Table 2
注:玻璃化温度Tg由示差扫描量热法(DSC,德国耐驰公司DSC204F1示差扫描量热仪)测定,升温速率10℃/min;热失重温度Td是在氮气气氛中失重1%的温度,在日本岛津公司的TGA-50H热重分析仪上进行测定,氮气流量为20mL/min;三线态能级T1是由日立的F4600荧光光谱仪测试,材料的测试条件为2*10-5mol/L的甲苯溶液;最高占据分子轨道HOMO能级是由光电子发射谱仪(AC-2型PESA)测试,测试为大气环境。Note: The glass transition temperature Tg is determined by differential scanning calorimetry (DSC, DSC204F1 differential scanning calorimeter of NETZSCH, Germany), and the heating rate is 10°C/min; the thermal weight loss temperature Td is the temperature at which the weight loses 1% in a nitrogen atmosphere, The measurement was carried out on the TGA-50H thermogravimetric analyzer of Shimadzu Corporation in Japan, and the nitrogen flow rate was 20mL/min; the triplet energy level T1 was tested by Hitachi's F4600 fluorescence spectrometer, and the test condition of the material was 2* 10-5 mol/ The toluene solution of L; the highest occupied molecular orbital HOMO energy level is measured by photoelectron emission spectrometer (AC-2 type PESA), and the test is atmospheric environment.
由上表数据可知,本发明的有机化合物具有高的玻璃转化温度,可提高材料膜相态稳定性,进一步提高器件使用寿命;具有高的T1能级,可以阻挡发光层能量损失,从而提升器件发光效率;合适的HOMO能级可以解决载流子的注入问题,可降低器件电压。因此,本发明的有机化合物用于OLED器件后,可有效提高器件的发光效率及使用寿命。It can be seen from the data in the above table that the organic compound of the present invention has a high glass transition temperature, which can improve the phase stability of the material film and further improve the service life of the device; it has a high T1 energy level, which can block the energy loss of the light-emitting layer, thereby improving the device. Luminous efficiency; suitable HOMO energy level can solve the problem of carrier injection and can reduce the device voltage. Therefore, after the organic compound of the present invention is used in an OLED device, the luminous efficiency and service life of the device can be effectively improved.
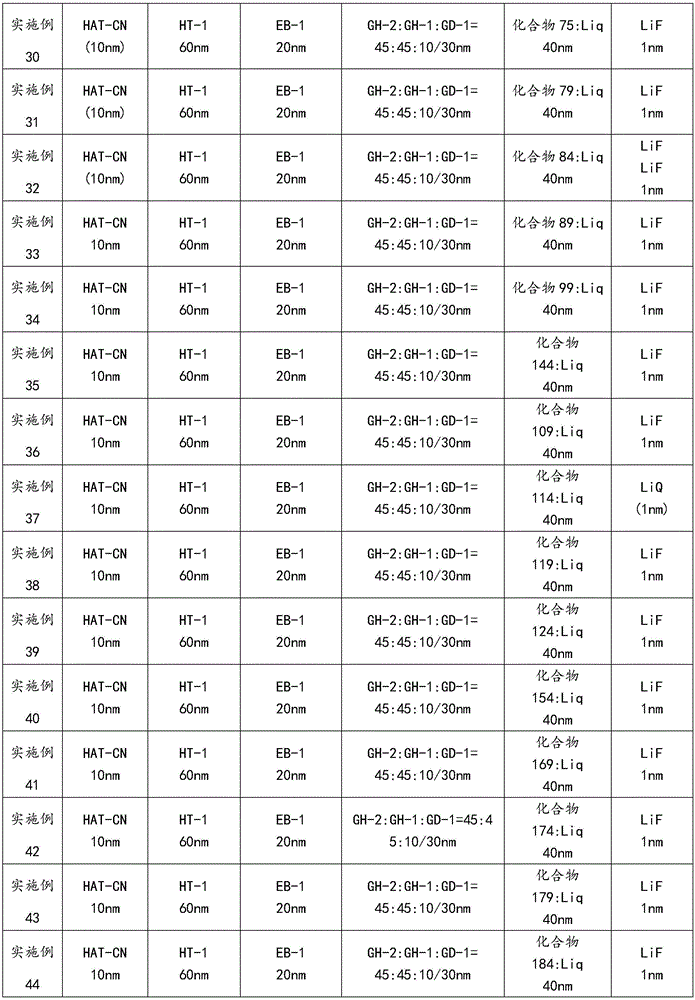
以下通过器件实施例1-22和比较例1详细说明本发明合成的OLED材料在器件中的应用效果。本发明器件实施例2-22和比较例1与器件实施例1相比器件的制作工艺完全相同,并且采用了相同的基板材料和电极材料,电极材料的膜厚也保持一致,所不同的是对器件中的发光层材料、电子阻挡层材料或空穴传输层材料做了更换。各实施例所得器件的性能测试结果如表3所示。The application effects of the OLED materials synthesized by the present invention in devices will be described in detail below through Device Examples 1-22 and Comparative Example 1. The device examples 2-22 and comparative example 1 of the present invention have the same manufacturing process as the device example 1, and the same substrate material and electrode material are used, and the film thickness of the electrode material is also kept the same, the difference is The material of the light-emitting layer, the material of the electron blocking layer or the material of the hole transport layer in the device is replaced. The performance test results of the devices obtained in each embodiment are shown in Table 3.
器件实施例1:一种电致发光器件,其结构如图1所示,具体的制备步骤包括:Device Embodiment 1: An electroluminescent device, the structure of which is shown in Figure 1, and the specific preparation steps include:
a)清洗透明基板层1上的ITO阳极层2,分别用去离子水、丙酮、乙醇超声清洗各15分钟,然后在等离子体清洗器中处理2分钟;a) cleaning the
b)在ITO阳极层2上,通过真空蒸镀方式蒸镀10nm的HAT-CN,该层为空穴注入层3;b) On the
c)在空穴注入层3上,通过真空蒸镀方式蒸镀50nm的HT-1,该层为空穴传输层4;c) On the
d)在空穴传输层4上,通过真空蒸镀方式蒸镀20nm厚的EB-1,作为电子阻挡层5;d) On the
e)在电子阻挡层5之上蒸镀30nm的发光层6,发光层包括主体材料和客体材料,具体材料的选用如表3所示,按照主体材料和客体材料的质量百分比,通过膜厚仪进行速率控制;e) Evaporating a 30 nm light-emitting
f)在发光层6之上,通过真空蒸镀方式蒸镀40nm的ET-1和Liq,ET-1和Liq质量比为1:1,这层材料作为空穴阻挡/电子传输层7;f) On the light-emitting
g)在空穴阻挡/电子传输层7之上,真空蒸镀1nm的LiF,该层为电子注入层8;g) on the hole blocking/
h)在电子注入层8之上,真空蒸镀阴极Al(100nm),该层为阴极电极层9;h) on the electron injection layer 8, vacuum evaporation of cathode Al (100nm), this layer is the
最后对器件进行封装处理。相关现有材料的分子结构式如下所示:Finally, the device is packaged. The molecular structures of the related existing materials are shown below:
按上述步骤制作电致发光器件,测量器件的电流效率和寿命。OLED器件的测试:检测过程使用IVL(电流-电压-亮度)测试系统(日本システム技研株式会社)。测定电致发光光谱,其中电流效率单位为cd/A,根据所得Lambert发射特征的电流/电压/亮度(IVL)特征曲线,计算并确定器件的寿命,该寿命数据为在10mA/cm2的恒电流密度下测得。The electroluminescent device was fabricated according to the above steps, and the current efficiency and life of the device were measured. Testing of OLED devices: The testing process used an IVL (current-voltage-luminance) testing system (Japanese Systeta Giken Co., Ltd.). The electroluminescence spectrum was measured, where the unit of current efficiency was cd/A, and the lifetime of the device was calculated and determined according to the current/voltage/luminance (IVL) characteristic curve of the obtained Lambert emission characteristics, and the lifetime data was constant at 10mA/ cm2 . measured at the current density.
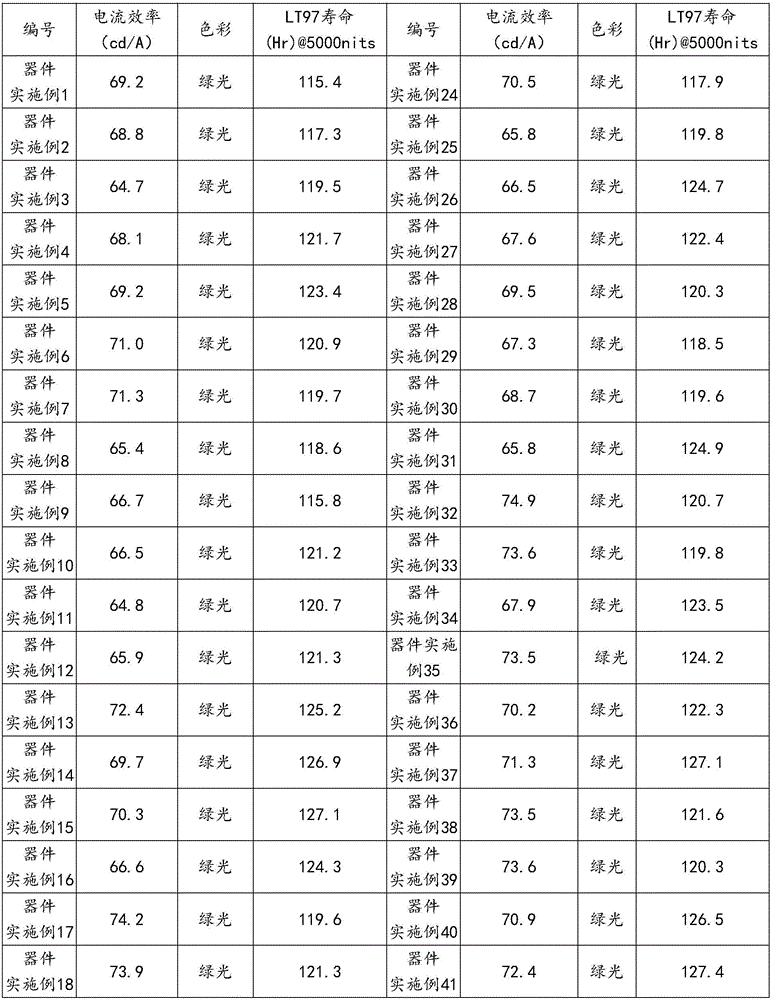
表3table 3
所得电致发光器件的检测数据见表4所示。The detection data of the obtained electroluminescent device are shown in Table 4.
表4Table 4
由表4的结果可以看出本发明由氮杂苯修饰的有机化合物可应用于OLED发光器件制作,并且与器件比较例相比,无论是器件的寿命还是效率方面,均比对比实施例中的现有技术有不同程度的改观,特别是器件的寿命有显著的提升。From the results in Table 4, it can be seen that the organic compounds modified by azepines of the present invention can be applied to the fabrication of OLED light-emitting devices, and compared with the comparative examples of the devices, both in terms of lifespan and efficiency of the devices, they are better than those in the comparative examples. The existing technology has been improved to varying degrees, especially the life of the device has been significantly improved.
通过进一步的实验研究,发现本发明材料制备的OLED器件在低温下工作时效率也比较稳定,将器件实施例5、13、20和器件比较例1在-10~80℃区间进行效率测试,所得结果如表5所示。Through further experimental research, it is found that the efficiency of the OLED device prepared by the material of the present invention is relatively stable when working at low temperature. The results are shown in Table 5.
表5table 5
从表5的数据可知,器件实施例5、13、20为本发明材料和已知材料搭配的器件结构,和器件比较例1相比,不仅低温效率高,而且在温度升高过程中,效率平稳升高。It can be seen from the data in Table 5 that the device examples 5, 13, and 20 are the device structures of the material of the present invention and the known materials. rise steadily.
以上所述仅为本发明的较佳实施例,并不用以限制本发明,凡在本发明的精神和原则之内,所作的任何修改、等同替换、改进等,均应包含在本发明的保护范围之内。The above descriptions are only preferred embodiments of the present invention, and are not intended to limit the present invention. Any modification, equivalent replacement, improvement, etc. made within the spirit and principle of the present invention shall be included in the protection of the present invention. within the range.
Claims (10)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811598707.1A CN111362866A (en) | 2018-12-26 | 2018-12-26 | Azabenzene modified organic compound and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811598707.1A CN111362866A (en) | 2018-12-26 | 2018-12-26 | Azabenzene modified organic compound and application thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN111362866A true CN111362866A (en) | 2020-07-03 |
Family
ID=71202707
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201811598707.1A Pending CN111362866A (en) | 2018-12-26 | 2018-12-26 | Azabenzene modified organic compound and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111362866A (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20210071273A (en) * | 2019-12-06 | 2021-06-16 | 솔루스첨단소재 주식회사 | Organic light-emitting compound and organic electroluminescent device using the same |
| CN114685427A (en) * | 2020-12-28 | 2022-07-01 | 常州强力昱镭光电材料有限公司 | Quinoline bipyridyl compound and application thereof in organic electroluminescent element |
| WO2024258156A1 (en) * | 2023-06-12 | 2024-12-19 | 주식회사 진웅산업 | Novel compound and organic light-emitting device comprising same |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012128509A2 (en) * | 2011-03-21 | 2012-09-27 | 덕산하이메탈(주) | Compound and organic electric element using same, and electronic device thereof |
-
2018
- 2018-12-26 CN CN201811598707.1A patent/CN111362866A/en active Pending
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012128509A2 (en) * | 2011-03-21 | 2012-09-27 | 덕산하이메탈(주) | Compound and organic electric element using same, and electronic device thereof |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20210071273A (en) * | 2019-12-06 | 2021-06-16 | 솔루스첨단소재 주식회사 | Organic light-emitting compound and organic electroluminescent device using the same |
| KR102686342B1 (en) | 2019-12-06 | 2024-07-19 | 솔루스첨단소재 주식회사 | Organic light-emitting compound and organic electroluminescent device using the same |
| CN114685427A (en) * | 2020-12-28 | 2022-07-01 | 常州强力昱镭光电材料有限公司 | Quinoline bipyridyl compound and application thereof in organic electroluminescent element |
| CN114685427B (en) * | 2020-12-28 | 2023-12-22 | 常州强力昱镭光电材料有限公司 | Quinoline bipyridine compound and application thereof in organic electroluminescent element |
| WO2024258156A1 (en) * | 2023-06-12 | 2024-12-19 | 주식회사 진웅산업 | Novel compound and organic light-emitting device comprising same |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN107586261B (en) | Organic compound containing spiro dibenzosuberene fluorene and application thereof | |
| CN110294663A (en) | It is a kind of using anthracene as the compound of core and its application on organic electroluminescence device | |
| WO2020135686A1 (en) | Organic compound with carbazole derivative as core and application thereof to organic electroluminescent device | |
| CN111410952A (en) | Organic electroluminescent material containing boron and application thereof in organic electroluminescent device | |
| CN107868090A (en) | Organic compounds containing 9,9'-spirobifluorene and organic light-emitting electroluminescent devices | |
| CN111233867A (en) | Organic compound with carbazole derivative as core and application thereof in organic electroluminescent device | |
| CN109956962A (en) | A compound with azaspirofluorene structure as parent nucleus and its application in organic electroluminescent devices | |
| CN111662190A (en) | Organic compound containing pyrene or aza-pyrene and application thereof | |
| WO2019085683A1 (en) | Anthrone-containing and nitrogen-containing heterocyclic compound and applications thereof in oleds | |
| CN106467529A (en) | A kind of electroluminescent organic material with 9 Fluorenones as core and its application | |
| CN115304566A (en) | Luminescent auxiliary material and preparation method and application thereof | |
| CN107602574A (en) | A kind of compound and its application using cyano group benzene as core | |
| CN107868049A (en) | Organic compound and organic electroluminescence device using the fluorenes of 9,9 ' spiral shell two as core | |
| CN110845508A (en) | Compound with spirofluorene anthrone as core, preparation method and application thereof | |
| WO2019114769A1 (en) | Compound containing pyridoindole and use thereof in organic electroluminescence device | |
| CN108218853A (en) | It is a kind of using the nitrogenous hexa-member heterocycle of dibenzo as the compound of core and its application on organic electroluminescence device | |
| CN111362866A (en) | Azabenzene modified organic compound and application thereof | |
| CN110577523A (en) | A compound containing triarylamine structure and organic electroluminescent device prepared therefrom | |
| WO2019085684A1 (en) | Cyanobenzene-containing or thiocyanobenzene-containing compound and applications thereof in organic electroluminescent devices | |
| CN110655504A (en) | A compound based on dibenzocycloheptene and its application in organic electroluminescent devices | |
| CN110655486A (en) | A kind of compound with dibenzocycloheptene as core and application thereof | |
| CN110526905A (en) | A kind of compound containing ketone and its application on organic electroluminescence device | |
| WO2023134228A1 (en) | Nitrogen-containing compound and organic electroluminescent device comprising same, and electronic apparatus | |
| WO2020052544A1 (en) | Compound taking benzo[1,2-b:5,4-b']dibenzofuran as core and application thereof | |
| CN110835305A (en) | A kind of organic compound containing dibenzocycloheptene and its preparation method and its application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| CB02 | Change of applicant information |
Address after: 214112 No. 210, Xinzhou Road, Wuxi, Jiangsu Applicant after: Jiangsu March Technology Co.,Ltd. Address before: 214112 No. 210, Xinzhou Road, Wuxi, Jiangsu Applicant before: JIANGSU SUNERA TECHNOLOGY Co.,Ltd. |
|
| CB02 | Change of applicant information | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20200703 |
|
| RJ01 | Rejection of invention patent application after publication |