CN111295213A - Injection device with dose limiter - Google Patents
Injection device with dose limiter Download PDFInfo
- Publication number
- CN111295213A CN111295213A CN201880071726.0A CN201880071726A CN111295213A CN 111295213 A CN111295213 A CN 111295213A CN 201880071726 A CN201880071726 A CN 201880071726A CN 111295213 A CN111295213 A CN 111295213A
- Authority
- CN
- China
- Prior art keywords
- dose
- housing
- limiter
- injection device
- feature
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 238000002347 injection Methods 0.000 title claims abstract description 122
- 239000007924 injection Substances 0.000 title claims abstract description 122
- 230000007246 mechanism Effects 0.000 claims abstract description 34
- 239000003814 drug Substances 0.000 claims abstract description 28
- 238000006073 displacement reaction Methods 0.000 claims abstract description 7
- 238000000034 method Methods 0.000 claims description 11
- JUFFVKRROAPVBI-PVOYSMBESA-N chembl1210015 Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(=O)N[C@H]1[C@@H]([C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O)[C@@H](O)[C@@H](CO[C@]3(O[C@@H](C[C@H](O)[C@H](O)CO)[C@H](NC(C)=O)[C@@H](O)C3)C(O)=O)O2)O)[C@@H](CO)O1)NC(C)=O)C(=O)NCC(=O)NCC(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CO)C(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H](N)CC=1NC=NC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)C1=CC=CC=C1 JUFFVKRROAPVBI-PVOYSMBESA-N 0.000 description 54
- 239000002585 base Substances 0.000 description 52
- 108010011459 Exenatide Proteins 0.000 description 50
- 229960001519 exenatide Drugs 0.000 description 50
- 101000976075 Homo sapiens Insulin Proteins 0.000 description 23
- QEFRNWWLZKMPFJ-YGVKFDHGSA-N L-methionine S-oxide Chemical compound CS(=O)CC[C@H](N)C(O)=O QEFRNWWLZKMPFJ-YGVKFDHGSA-N 0.000 description 22
- PBGKTOXHQIOBKM-FHFVDXKLSA-N insulin (human) Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@H]1CSSC[C@H]2C(=O)N[C@H](C(=O)N[C@@H](CO)C(=O)N[C@H](C(=O)N[C@H](C(N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC=3C=CC(O)=CC=3)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=3C=CC(O)=CC=3)C(=O)N[C@@H](CSSC[C@H](NC(=O)[C@H](C(C)C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=3C=CC(O)=CC=3)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=3NC=NC=3)NC(=O)[C@H](CO)NC(=O)CNC1=O)C(=O)NCC(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)O)C(O)=O)C(=O)N[C@@H](CC(N)=O)C(O)=O)=O)CSSC[C@@H](C(N2)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C(C)C)NC(=O)[C@@H](NC(=O)CN)[C@@H](C)CC)[C@@H](C)CC)[C@@H](C)O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@@H](N)CC=1C=CC=CC=1)C(C)C)C1=CN=CN1 PBGKTOXHQIOBKM-FHFVDXKLSA-N 0.000 description 22
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical class N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 12
- 235000001014 amino acid Nutrition 0.000 description 9
- 150000001413 amino acids Chemical class 0.000 description 9
- 239000012634 fragment Substances 0.000 description 8
- 150000003839 salts Chemical class 0.000 description 8
- 150000001875 compounds Chemical class 0.000 description 7
- 239000000427 antigen Substances 0.000 description 6
- 102000036639 antigens Human genes 0.000 description 6
- 108091007433 antigens Proteins 0.000 description 6
- 238000003466 welding Methods 0.000 description 6
- 108090001061 Insulin Proteins 0.000 description 5
- 102000004877 Insulin Human genes 0.000 description 5
- 239000000853 adhesive Substances 0.000 description 5
- 230000001070 adhesive effect Effects 0.000 description 5
- 229940125396 insulin Drugs 0.000 description 5
- 108090000765 processed proteins & peptides Proteins 0.000 description 5
- 108060003951 Immunoglobulin Proteins 0.000 description 4
- 102000018358 immunoglobulin Human genes 0.000 description 4
- 230000003993 interaction Effects 0.000 description 4
- 239000007788 liquid Substances 0.000 description 4
- 108010047041 Complementarity Determining Regions Proteins 0.000 description 3
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 3
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 3
- -1 alkaline earth metal cations Chemical class 0.000 description 3
- 210000003719 b-lymphocyte Anatomy 0.000 description 3
- 230000000295 complement effect Effects 0.000 description 3
- 206010012601 diabetes mellitus Diseases 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 150000004676 glycans Chemical class 0.000 description 3
- 229940088597 hormone Drugs 0.000 description 3
- 239000005556 hormone Substances 0.000 description 3
- 239000003055 low molecular weight heparin Substances 0.000 description 3
- 229940127215 low-molecular weight heparin Drugs 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 229920001282 polysaccharide Polymers 0.000 description 3
- 239000005017 polysaccharide Substances 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 238000013519 translation Methods 0.000 description 3
- 208000004476 Acute Coronary Syndrome Diseases 0.000 description 2
- 208000002249 Diabetes Complications Diseases 0.000 description 2
- 206010012689 Diabetic retinopathy Diseases 0.000 description 2
- 108010088406 Glucagon-Like Peptides Proteins 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 150000001447 alkali salts Chemical class 0.000 description 2
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 2
- 230000000903 blocking effect Effects 0.000 description 2
- 125000000151 cysteine group Chemical group N[C@@H](CS)C(=O)* 0.000 description 2
- 230000000881 depressing effect Effects 0.000 description 2
- LMHMJYMCGJNXRS-IOPUOMRJSA-N exendin-3 Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)NCC(=O)NCC(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CO)C(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC=1N=CNC=1)[C@H](C)O)[C@H](C)O)C(C)C)C1=CC=CC=C1 LMHMJYMCGJNXRS-IOPUOMRJSA-N 0.000 description 2
- 239000011888 foil Substances 0.000 description 2
- 238000003780 insertion Methods 0.000 description 2
- 230000037431 insertion Effects 0.000 description 2
- 239000000203 mixture Substances 0.000 description 2
- 239000000178 monomer Substances 0.000 description 2
- 229940090048 pen injector Drugs 0.000 description 2
- 230000002265 prevention Effects 0.000 description 2
- 102000004196 processed proteins & peptides Human genes 0.000 description 2
- 230000001681 protective effect Effects 0.000 description 2
- 239000012453 solvate Substances 0.000 description 2
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 description 1
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 1
- 125000001831 (C6-C10) heteroaryl group Chemical group 0.000 description 1
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 1
- 208000032529 Accidental overdose Diseases 0.000 description 1
- 208000035285 Allergic Seasonal Rhinitis Diseases 0.000 description 1
- 206010002383 Angina Pectoris Diseases 0.000 description 1
- 201000001320 Atherosclerosis Diseases 0.000 description 1
- 102000004506 Blood Proteins Human genes 0.000 description 1
- 108010017384 Blood Proteins Proteins 0.000 description 1
- 108010037003 Buserelin Proteins 0.000 description 1
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 description 1
- 125000000041 C6-C10 aryl group Chemical group 0.000 description 1
- 208000017667 Chronic Disease Diseases 0.000 description 1
- 108010041986 DNA Vaccines Proteins 0.000 description 1
- 108010000437 Deamino Arginine Vasopressin Proteins 0.000 description 1
- 208000005189 Embolism Diseases 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 102000012673 Follicle Stimulating Hormone Human genes 0.000 description 1
- 108010079345 Follicle Stimulating Hormone Proteins 0.000 description 1
- 102000003886 Glycoproteins Human genes 0.000 description 1
- 108090000288 Glycoproteins Proteins 0.000 description 1
- 229920002683 Glycosaminoglycan Polymers 0.000 description 1
- 108010069236 Goserelin Proteins 0.000 description 1
- BLCLNMBMMGCOAS-URPVMXJPSA-N Goserelin Chemical compound C([C@@H](C(=O)N[C@H](COC(C)(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N1[C@@H](CCC1)C(=O)NNC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H]1NC(=O)CC1)C1=CC=C(O)C=C1 BLCLNMBMMGCOAS-URPVMXJPSA-N 0.000 description 1
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 1
- 102000002265 Human Growth Hormone Human genes 0.000 description 1
- 108010000521 Human Growth Hormone Proteins 0.000 description 1
- 239000000854 Human Growth Hormone Substances 0.000 description 1
- 108010024118 Hypothalamic Hormones Proteins 0.000 description 1
- 102000015611 Hypothalamic Hormones Human genes 0.000 description 1
- 102000006496 Immunoglobulin Heavy Chains Human genes 0.000 description 1
- 108010019476 Immunoglobulin Heavy Chains Proteins 0.000 description 1
- 102000013463 Immunoglobulin Light Chains Human genes 0.000 description 1
- 108010065825 Immunoglobulin Light Chains Proteins 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 101150008417 LIN gene Proteins 0.000 description 1
- 108010000817 Leuprolide Proteins 0.000 description 1
- XVVOERDUTLJJHN-UHFFFAOYSA-N Lixisenatide Chemical compound C=1NC2=CC=CC=C2C=1CC(C(=O)NC(CC(C)C)C(=O)NC(CCCCN)C(=O)NC(CC(N)=O)C(=O)NCC(=O)NCC(=O)N1C(CCC1)C(=O)NC(CO)C(=O)NC(CO)C(=O)NCC(=O)NC(C)C(=O)N1C(CCC1)C(=O)N1C(CCC1)C(=O)NC(CO)C(=O)NC(CCCCN)C(=O)NC(CCCCN)C(=O)NC(CCCCN)C(=O)NC(CCCCN)C(=O)NC(CCCCN)C(=O)NC(CCCCN)C(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)CC)NC(=O)C(NC(=O)C(CC(C)C)NC(=O)C(CCCNC(N)=N)NC(=O)C(NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(CCC(O)=O)NC(=O)C(CCC(O)=O)NC(=O)C(CCSC)NC(=O)C(CCC(N)=O)NC(=O)C(CCCCN)NC(=O)C(CO)NC(=O)C(CC(C)C)NC(=O)C(CC(O)=O)NC(=O)C(CO)NC(=O)C(NC(=O)C(CC=1C=CC=CC=1)NC(=O)C(NC(=O)CNC(=O)C(CCC(O)=O)NC(=O)CNC(=O)C(N)CC=1NC=NC=1)C(C)O)C(C)O)C(C)C)CC1=CC=CC=C1 XVVOERDUTLJJHN-UHFFFAOYSA-N 0.000 description 1
- 102000009151 Luteinizing Hormone Human genes 0.000 description 1
- 108010073521 Luteinizing Hormone Proteins 0.000 description 1
- 108010021717 Nafarelin Proteins 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 108091034117 Oligonucleotide Proteins 0.000 description 1
- 108090000526 Papain Proteins 0.000 description 1
- 102000057297 Pepsin A Human genes 0.000 description 1
- 108090000284 Pepsin A Proteins 0.000 description 1
- 108010047386 Pituitary Hormones Proteins 0.000 description 1
- 102000006877 Pituitary Hormones Human genes 0.000 description 1
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 208000010378 Pulmonary Embolism Diseases 0.000 description 1
- 108010010056 Terlipressin Proteins 0.000 description 1
- 208000001435 Thromboembolism Diseases 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 125000000539 amino acid group Chemical group 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 230000000890 antigenic effect Effects 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- CUWODFFVMXJOKD-UVLQAERKSA-N buserelin Chemical compound CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](COC(C)(C)C)NC(=O)[C@@H](NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H]1NC(=O)CC1)CC1=CC=C(O)C=C1 CUWODFFVMXJOKD-UVLQAERKSA-N 0.000 description 1
- 229960002719 buserelin Drugs 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- POIUWJQBRNEFGX-XAMSXPGMSA-N cathelicidin Chemical compound C([C@@H](C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(O)=O)NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CC(C)C)C1=CC=CC=C1 POIUWJQBRNEFGX-XAMSXPGMSA-N 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- 210000003464 cuspid Anatomy 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 229960004281 desmopressin Drugs 0.000 description 1
- NFLWUMRGJYTJIN-NXBWRCJVSA-N desmopressin Chemical compound C([C@H]1C(=O)N[C@H](C(N[C@@H](CC(N)=O)C(=O)N[C@@H](CSSCCC(=O)N[C@@H](CC=2C=CC(O)=CC=2)C(=O)N1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O)=O)CCC(=O)N)C1=CC=CC=C1 NFLWUMRGJYTJIN-NXBWRCJVSA-N 0.000 description 1
- 230000029087 digestion Effects 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 229960005153 enoxaparin sodium Drugs 0.000 description 1
- 229940088598 enzyme Drugs 0.000 description 1
- 108010015174 exendin 3 Proteins 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 229960002913 goserelin Drugs 0.000 description 1
- 229960002897 heparin Drugs 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- 229920002674 hyaluronan Polymers 0.000 description 1
- 229960003160 hyaluronic acid Drugs 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 125000004435 hydrogen atom Chemical class [H]* 0.000 description 1
- 239000000960 hypophysis hormone Substances 0.000 description 1
- 229940043650 hypothalamic hormone Drugs 0.000 description 1
- 239000000601 hypothalamic hormone Substances 0.000 description 1
- 229940072221 immunoglobulins Drugs 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 239000004026 insulin derivative Substances 0.000 description 1
- GFIJNRVAKGFPGQ-LIJARHBVSA-N leuprolide Chemical compound CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)CC1=CC=C(O)C=C1 GFIJNRVAKGFPGQ-LIJARHBVSA-N 0.000 description 1
- 229960004338 leuprorelin Drugs 0.000 description 1
- XVVOERDUTLJJHN-IAEQDCLQSA-N lixisenatide Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)NCC(=O)NCC(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H](N)CC=1N=CNC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)C1=CC=CC=C1 XVVOERDUTLJJHN-IAEQDCLQSA-N 0.000 description 1
- 108010004367 lixisenatide Proteins 0.000 description 1
- 229960001093 lixisenatide Drugs 0.000 description 1
- 229940040129 luteinizing hormone Drugs 0.000 description 1
- 208000002780 macular degeneration Diseases 0.000 description 1
- 230000035800 maturation Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- RWHUEXWOYVBUCI-ITQXDASVSA-N nafarelin Chemical compound C([C@@H](C(=O)N[C@H](CC=1C=C2C=CC=CC2=CC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N1[C@@H](CCC1)C(=O)NCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H]1NC(=O)CC1)C1=CC=C(O)C=C1 RWHUEXWOYVBUCI-ITQXDASVSA-N 0.000 description 1
- 229960002333 nafarelin Drugs 0.000 description 1
- 229940055729 papain Drugs 0.000 description 1
- 235000019834 papain Nutrition 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 229940111202 pepsin Drugs 0.000 description 1
- 239000008194 pharmaceutical composition Substances 0.000 description 1
- 230000001817 pituitary effect Effects 0.000 description 1
- 229920001184 polypeptide Polymers 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 125000001500 prolyl group Chemical group [H]N1C([H])(C(=O)[*])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 235000018102 proteins Nutrition 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 230000017854 proteolysis Effects 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 229960004532 somatropin Drugs 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 229960003813 terlipressin Drugs 0.000 description 1
- BENFXAYNYRLAIU-QSVFAHTRSA-N terlipressin Chemical compound NCCCC[C@@H](C(=O)NCC(N)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H]1NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CC=2C=CC=CC=2)NC(=O)[C@H](CC=2C=CC(O)=CC=2)NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)CN)CSSC1 BENFXAYNYRLAIU-QSVFAHTRSA-N 0.000 description 1
- CIJQTPFWFXOSEO-NDMITSJXSA-J tetrasodium;(2r,3r,4s)-2-[(2r,3s,4r,5r,6s)-5-acetamido-6-[(1r,2r,3r,4r)-4-[(2r,3s,4r,5r,6r)-5-acetamido-6-[(4r,5r,6r)-2-carboxylato-4,5-dihydroxy-6-[[(1r,3r,4r,5r)-3-hydroxy-4-(sulfonatoamino)-6,8-dioxabicyclo[3.2.1]octan-2-yl]oxy]oxan-3-yl]oxy-2-(hydroxy Chemical compound [Na+].[Na+].[Na+].[Na+].O([C@@H]1[C@@H](COS(O)(=O)=O)O[C@@H]([C@@H]([C@H]1O)NC(C)=O)O[C@@H]1C(C[C@H]([C@@H]([C@H]1O)O)O[C@@H]1[C@@H](CO)O[C@H](OC2C(O[C@@H](OC3[C@@H]([C@@H](NS([O-])(=O)=O)[C@@H]4OC[C@H]3O4)O)[C@H](O)[C@H]2O)C([O-])=O)[C@H](NC(C)=O)[C@H]1C)C([O-])=O)[C@@H]1OC(C([O-])=O)=C[C@H](O)[C@H]1O CIJQTPFWFXOSEO-NDMITSJXSA-J 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M5/00—Devices for bringing media into the body in a subcutaneous, intra-vascular or intramuscular way; Accessories therefor, e.g. filling or cleaning devices, arm-rests
- A61M5/178—Syringes
- A61M5/31—Details
- A61M5/315—Pistons; Piston-rods; Guiding, blocking or restricting the movement of the rod or piston; Appliances on the rod for facilitating dosing ; Dosing mechanisms
- A61M5/31533—Dosing mechanisms, i.e. setting a dose
- A61M5/31535—Means improving security or handling thereof, e.g. blocking means, means preventing insufficient dosing, means allowing correction of overset dose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M5/00—Devices for bringing media into the body in a subcutaneous, intra-vascular or intramuscular way; Accessories therefor, e.g. filling or cleaning devices, arm-rests
- A61M5/178—Syringes
- A61M5/31—Details
- A61M5/315—Pistons; Piston-rods; Guiding, blocking or restricting the movement of the rod or piston; Appliances on the rod for facilitating dosing ; Dosing mechanisms
- A61M5/31533—Dosing mechanisms, i.e. setting a dose
- A61M5/31535—Means improving security or handling thereof, e.g. blocking means, means preventing insufficient dosing, means allowing correction of overset dose
- A61M5/31536—Blocking means to immobilize a selected dose, e.g. to administer equal doses
- A61M2005/3154—Blocking means to immobilize a selected dose, e.g. to administer equal doses limiting maximum permissible dose
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M5/00—Devices for bringing media into the body in a subcutaneous, intra-vascular or intramuscular way; Accessories therefor, e.g. filling or cleaning devices, arm-rests
- A61M5/178—Syringes
- A61M5/31—Details
- A61M5/315—Pistons; Piston-rods; Guiding, blocking or restricting the movement of the rod or piston; Appliances on the rod for facilitating dosing ; Dosing mechanisms
- A61M5/31533—Dosing mechanisms, i.e. setting a dose
- A61M5/31535—Means improving security or handling thereof, e.g. blocking means, means preventing insufficient dosing, means allowing correction of overset dose
- A61M5/31541—Means preventing setting of a dose beyond the amount remaining in the cartridge
Landscapes
- Health & Medical Sciences (AREA)
- Vascular Medicine (AREA)
- Engineering & Computer Science (AREA)
- Anesthesiology (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Hematology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Infusion, Injection, And Reservoir Apparatuses (AREA)
Abstract
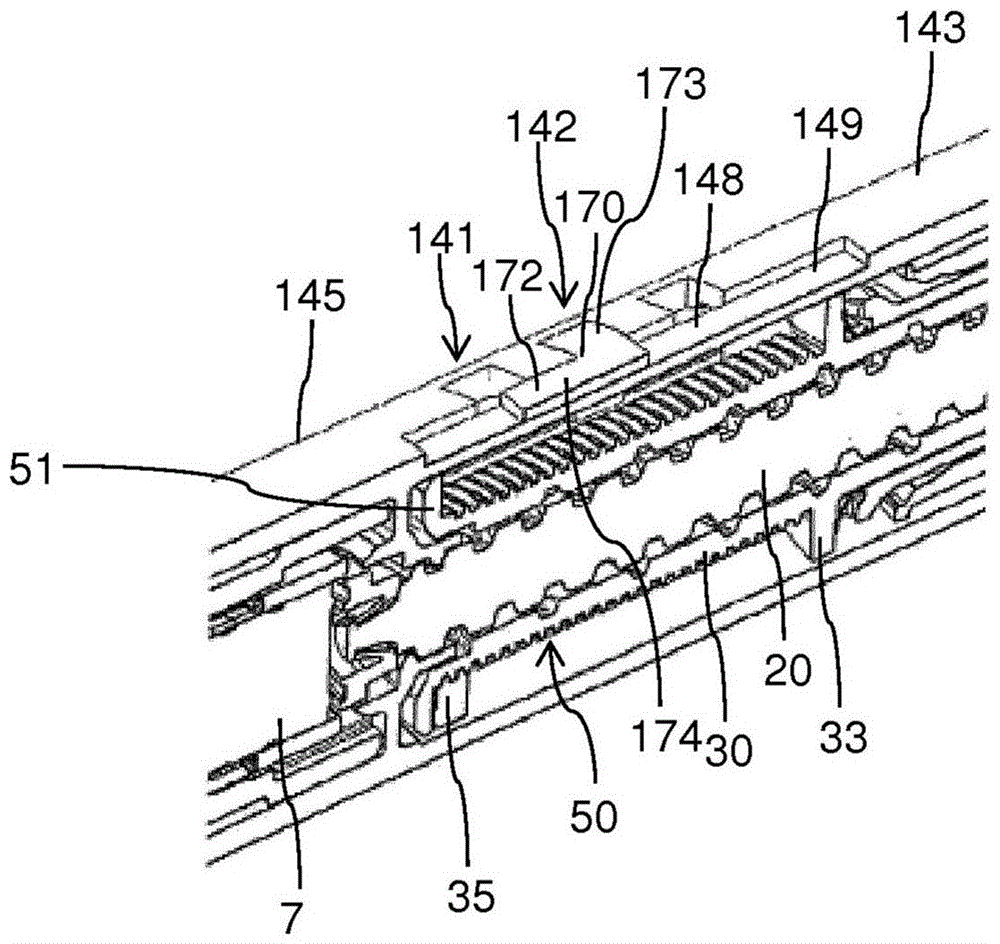
本公开涉及一种用于设定和注射一定剂量的药剂的注射装置,其包括:‑细长外壳(10),其沿轴向方向(z)延伸并被配置为容纳剂量设定机构(9),其中所述外壳(10)包括具有至少第一紧固特征件(41;141)的侧壁(43;143);‑剂量刻度盘(12),其可相对于所述外壳(10)旋转以设定剂量;‑剂量跟踪器(50),其布置在所述外壳(10)内侧并且可操作地可连接到所述剂量刻度盘(12),所述剂量跟踪器(50)包括至少一个跟踪止挡特征件(51),其中所述剂量跟踪器(50)是在设定剂量期间可相对于所述外壳(10)平移地或旋转地移位中的一个,并且其中所述剂量跟踪器(50)相对于所述外壳(10)的位置状态指示所述剂量的规格;和‑限制器(70;170),其可从所述外壳(10)外侧附接到所述外壳(10)的所述侧壁(43;143),并且可与所述至少第一紧固特征件(41,141)接合,其中所述限制器(70;170)包括当所述限制器(70;170)附接到所述侧壁(43)时从所述侧壁(43;143)向内延伸的至少一个限制器止挡特征件(71;171),其中所述限制器止挡特征件(71;171)和所述跟踪止挡特征件(51)被配置为相互接合并阻止所述剂量跟踪器(50)的移位超过预定义的最大剂量位置状态。
The present disclosure relates to an injection device for setting and injecting a dose of a medicament, comprising: an elongated housing (10) extending in an axial direction (z) and configured to accommodate a dose setting mechanism (9) ), wherein the housing (10) comprises a side wall (43; 143) having at least a first fastening feature (41; 141); a dose dial (12), which can be relative to the housing (10) rotates to set a dose; - a dose tracker (50) arranged inside the housing (10) and operably connectable to the dose dial (12), the dose tracker (50) comprising at least a tracking stop feature (51), wherein the dose tracker (50) is one of translationally or rotationally displaceable relative to the housing (10) during setting of a dose, and wherein the dose a positional state of the tracker (50) relative to the housing (10) indicating the size of the dose; and a limiter (70; 170) attachable to the housing (10) from outside the housing (10) 10) of said side wall (43; 143) and engageable with said at least first fastening feature (41, 141), wherein said limiter (70; 170) comprises when said limiter (70) ; 170) at least one limiter stop feature (71; 171) extending inwardly from said side wall (43; 143) when attached to said side wall (43), wherein said limiter stop feature The member (71; 171) and the tracking stop feature (51) are configured to mutually engage and prevent displacement of the dose tracker (50) beyond a predefined maximum dose position state.
Description
说明illustrate
在一方面,本公开涉及用于设定和分配一定剂量的药剂的注射装置,如笔式注射器。特别地,本公开涉及一种提供最大剂量机构的注射装置,即仅可操作用于分配不超过预定义最大阈值的剂量的剂量设定和分配机构。In one aspect, the present disclosure relates to an injection device, such as a pen injector, for setting and dispensing a dose of a medicament. In particular, the present disclosure relates to an injection device that provides a maximum dose mechanism, ie a dose setting and dispensing mechanism that is only operable to dispense a dose that does not exceed a predefined maximum threshold.
背景background
用于设定和分配单剂或多剂液体药剂的注射装置本身在本领域中是众所周知的。通常,此类装置具有与普通注射筒器基本相似的目的。Injection devices for setting and dispensing single or multiple doses of liquid medicaments are themselves well known in the art. Typically, such devices serve a substantially similar purpose to that of a common syringe.
注射装置、特别是笔式注射器必须满足许多使用者特定的要求。例如,在患者患有如糖尿病等慢性疾病的情况下,患者可能身体虚弱并且还可能视力受损。因此,尤其旨在用于家庭用药的适用注射装置需要在构造上具有鲁棒性并且应易于使用。而且,对装置及其部件的操纵和一般处置应当明了且容易理解。此外,剂量设定程序以及剂量分配程序必须易于操作并且必须是明确的。Injection devices, especially pen injectors, have to meet many user-specific requirements. For example, where the patient suffers from a chronic disease such as diabetes, the patient may be frail and may also be visually impaired. Therefore, suitable injection devices especially intended for home use need to be robust in construction and should be easy to use. Moreover, the manipulation and general handling of the device and its components should be clear and easily understood. Furthermore, the dose setting procedure as well as the dose dispensing procedure must be easy to operate and must be unambiguous.
典型地,此类装置包括外壳,所述外壳包括特定的药筒保持器,所述药筒保持器被适配成接收至少部分地填充有待分配药剂的药筒。此类装置还包括驱动机构,所述驱动机构通常具有可移位的活塞杆,所述活塞杆被适配成与药筒的活塞可操作地接合。借助于驱动机构及其活塞杆,药筒的活塞可在远侧方向上或分配方向上移位,并因此可以借助刺穿组装件排出预定义量的药剂,所述穿刺组件与注射装置的外壳的远端区段可释放地联接。Typically, such devices comprise a housing comprising a specific cartridge holder adapted to receive a cartridge at least partially filled with the medicament to be dispensed. Such devices also include a drive mechanism, typically having a displaceable piston rod adapted to operatively engage the piston of the cartridge. By means of the drive mechanism and its piston rod, the piston of the cartridge can be displaced in the distal direction or in the dispensing direction and thus can expel a predefined amount of medicament by means of the piercing assembly, which is in contact with the housing of the injection device The distal segment is releasably coupled.
在多剂量药筒中设置并包含有待由注射装置分配的药剂。此类药筒典型地包括玻璃桶,所述玻璃桶在远侧方向上由可刺穿的密封件密封并且在近侧方向上由活塞进一步密封。对于可重复使用的注射装置,可以将空药筒更换成新的。相反,当药筒中的药剂已经被分配或用完时,将丢弃一次性类型的注射装置。The medicament to be dispensed by the injection device is provided and contained in a multi-dose cartridge. Such cartridges typically include a glass barrel that is sealed in a distal direction by a pierceable seal and further sealed in a proximal direction by a piston. For reusable injection devices, the empty cartridge can be replaced with a new one. Conversely, the disposable type of injection device is discarded when the medicament in the cartridge has been dispensed or used up.
对于一些应用,限制可以从药筒分配或排出的最大剂量规格可能是有利的。然后,可以防止意外用药过量。For some applications, it may be advantageous to limit the maximum dosage strength that can be dispensed or expelled from the cartridge. Then, accidental overdose can be prevented.
目标Target
因此,本公开文本的一个目标是提供一种提供最大剂量功能或最大剂量限制器的注射装置。最大剂量功能或最大剂量限制器应易于适应现有的注射装置设计。最大剂量功能或最大剂量限制器也应该可以按需配置,并且应该提供一种简单直观的方法来改变可通过注射装置排出的最大剂量规格。Accordingly, it is an object of the present disclosure to provide an injection device that provides a maximum dose function or maximum dose limiter. The maximum dose function or maximum dose limiter should be easily adaptable to existing injection device designs. The maximum dose function or maximum dose limiter should also be configurable on demand and should provide an easy and intuitive way to change the maximum dose specification that can be expelled through the injection device.
通过仅修改有限数量的现有装置部件,就应该可以达成最大剂量功能的实现。进一步的目的是通过仅改变装置的单个部件或少数几个部件来单独修改最大剂量值或剂量规格。因此,应该可以通过改变装置或其驱动机构的仅一个或少数几个部件来配置装置或其驱动机构的最大剂量功能。最大剂量功能或最大剂量限制器应普遍适用于各种驱动机构和注射装置。特别地,最大剂量功能或最大剂量限制器应同样适用于一次性注射装置以及可重复使用的注射装置。By modifying only a limited number of existing device components, it should be possible to achieve maximum dose functionality. A further aim is to individually modify the maximum dose value or dose specification by changing only a single component or a few components of the device. Therefore, it should be possible to configure the maximum dose function of the device or its drive mechanism by changing only one or a few components of the device or its drive mechanism. The maximum dose function or maximum dose limiter shall be universally applicable to various drive mechanisms and injection devices. In particular, the maximum dose function or maximum dose limiter should apply equally to single-use injection devices as well as reusable injection devices.
发明内容SUMMARY OF THE INVENTION
在一方面,提供了一种用于设定和分配从而注射一定剂量的药剂的注射装置。注射装置包括沿轴向方向延伸的细长外壳。对外壳进行配置并确定尺寸以容纳剂量设定机构。典型地,也对外壳确定尺寸并配置以容纳填充有药剂的药筒。外壳包括具有至少第一紧固特征件的侧壁。典型地,外壳为圆柱形或管形形状。圆柱体长轴线在轴向方向上延伸,并且侧壁为管形或圆柱形状。外壳的侧壁的几何形状因此可以限定径向方向和周向方向。In one aspect, an injection device for setting and dispensing a dose of a medicament is provided. The injection device includes an elongated housing extending in an axial direction. The housing is configured and dimensioned to accommodate the dose setting mechanism. Typically, the housing is also sized and configured to receive a cartridge filled with a medicament. The housing includes a side wall having at least a first fastening feature. Typically, the housing is cylindrical or tubular in shape. The long axis of the cylinder extends in the axial direction, and the side wall is tubular or cylindrical. The geometry of the side walls of the housing can thus define radial and circumferential directions.
注射装置还包括剂量刻度盘。剂量刻度盘可相对于外壳旋转以设定剂量。剂量刻度盘可以可旋转地支撑在外壳之上或之中。例如,剂量刻度盘可以可旋转地支撑在外壳的近端区段处。剂量刻度盘是使用者可致动的。因此,使用者可以相对于外壳抓握并旋转剂量刻度盘以设定或选择可变规格的剂量。The injection device also includes a dose dial. The dose dial is rotatable relative to the housing to set the dose. The dose dial may be rotatably supported on or in the housing. For example, the dose dial may be rotatably supported at the proximal end section of the housing. The dose dial is user actuatable. Thus, the user can grip and rotate the dose dial relative to the housing to set or select a variable gauge dose.
注射装置还包括布置在外壳内侧的剂量跟踪器。剂量跟踪器可操作地可连接到剂量刻度盘。剂量跟踪器包括至少一个跟踪止挡特征件。剂量跟踪器是在设定剂量期间相对于外壳平移地或旋转地移位中的至少一个。剂量跟踪器相对于外壳的位置状态指示剂量的规格。在当前上下文中,部件(例如,剂量跟踪器)的“位置状态”包括部件的位置和部件相对于另一部件(例如,相对于外壳)的角定向。剂量跟踪器可以在轴向地固定的同时被旋转地支撑在外壳内侧。替代性地,剂量跟踪器可以在转向上固定到外壳上的同时相对于外壳可滑动地支撑在外壳内侧。此外,剂量跟踪器可相对于外壳平移地且旋转地移位。它可以沿螺旋路径相对于外壳移动。它可以与外壳螺纹接合。The injection device also includes a dose tracker disposed inside the housing. The dose tracker is operably connectable to the dose dial. The dose tracker includes at least one tracking stop feature. The dose tracker is at least one of translationally or rotationally displaced relative to the housing during setting of a dose. The positional status of the dose tracker relative to the housing indicates the specification of the dose. In the current context, a "positional state" of a component (eg, a dose tracker) includes the location of the component and the angular orientation of the component relative to another component (eg, relative to the housing). The dose tracker may be rotatably supported inside the housing while being axially fixed. Alternatively, the dose tracker may be slidably supported inside the housing relative to the housing while being rotationally fixed to the housing. Additionally, the dose tracker is translationally and rotationally displaceable relative to the housing. It can move relative to the housing along a helical path. It can be threadedly engaged with the housing.
剂量跟踪器属于剂量设定机构。剂量跟踪器相对于外壳的位置状态明确地与实际设定的剂量规格相关。依据剂量跟踪器的具体实现方式,剂量跟踪器相对于外壳的旋转程度和/或纵向或轴向平移程度指示实际设定的剂量规格。A dose tracker is a dose setting mechanism. The positional state of the dose tracker relative to the housing is unambiguously related to the actual set dose specification. Depending on the specific implementation of the dose tracker, the degree of rotation and/or the degree of longitudinal or axial translation of the dose tracker relative to the housing is indicative of the actual set dose specification.
注射装置还包括可附接到外壳侧壁的限制器。可从外壳外侧附接限制器。它可与至少第一紧固特征件接合。限制器包括至少一个限制器止挡特征件,当限制器附接到侧壁时,所述限制器止挡特征件从侧壁向内延伸。限制器止挡特征件和跟踪止挡特征件被配置为相互接合并阻止剂量跟踪器的移位超过预定义的最大剂量位置状态。The injection device also includes a restrictor attachable to the side wall of the housing. The limiter can be attached from the outside of the housing. It is engageable with at least the first fastening feature. The limiter includes at least one limiter stop feature that extends inwardly from the sidewall when the limiter is attached to the sidewall. The limiter stop feature and the tracking stop feature are configured to interengage and prevent displacement of the dose tracker beyond a predefined maximum dose position state.
限制器及其至少一个限制器止挡特征件可附接且可固定到外壳侧壁的特定部分,所述特定部分对应于剂量跟踪器的跟踪止挡特征件的预定义的最大剂量位置状态。当剂量跟踪器到达最大剂量位置状态时,限制器和/或限制器止挡特征件附接到其上的外壳的特定部分与跟踪止挡特征件的位置状态重合。The limiter and its at least one limiter stop feature are attachable and fixable to a specific portion of the housing sidewall that corresponds to a predefined maximum dose position state of the tracking stop feature of the dose tracker. When the dose tracker reaches the maximum dose position state, the limiter and/or the particular portion of the housing to which the limiter stop feature is attached coincides with the position state of the track stop feature.
换言之,当剂量跟踪器达到最大剂量位置状态时,其跟踪止挡特征件与固定到侧壁的相应部分上的限制器止挡特征件接合(例如,邻接)。限制器止挡特征件和跟踪止挡特征件可以包括相互对应的止挡面,例如在周向和径向方向上延伸以轴向接合。替代性地,限制器止挡特征件和跟踪止挡特征件包括在轴向方向和径向方向上延伸的彼此对应的止挡面以周向接合。当被配置为轴向接合时,限制器止挡特征件和跟踪止挡特征件的相互接合提供轴向止挡,从而阻挡和阻止剂量跟踪器的纵向或轴向平移超过预定义的最大轴向剂量位置状态。In other words, when the dose tracker reaches the maximum dose position state, its tracking stop feature engages (eg, abuts) a limiter stop feature secured to a corresponding portion of the sidewall. The limiter stop feature and the tracking stop feature may include mutually corresponding stop surfaces, eg, extending in circumferential and radial directions for axial engagement. Alternatively, the limiter stop feature and the tracking stop feature include mutually corresponding stop surfaces extending in the axial and radial directions for circumferential engagement. When configured for axial engagement, the interengagement of the limiter stop feature and the track stop feature provides an axial stop, thereby blocking and preventing longitudinal or axial translation of the dose tracker beyond a predefined maximum axial Dose position status.
当被配置为周向或切向接合时,限制器止挡特征件和跟踪止挡特征件的相互接合提供旋转止挡,从而阻挡和阻止剂量跟踪器相对于外壳的旋转超过预定义的最大旋转剂量位置状态。When configured for circumferential or tangential engagement, the interengagement of the limiter stop feature and the track stop feature provides a rotational stop, thereby blocking and preventing rotation of the dose tracker relative to the housing beyond a predefined maximum rotation Dose position status.
预定义的最大剂量位置状态限定了可以由注射装置选择和分配的最大剂量。通过将限制器止挡特征件放置在外壳侧壁上的对应于剂量跟踪器的特别是跟踪止挡特征件的位置状态的预定义位置,可以限定注射装置可分配的最大剂量。The predefined maximum dose position state defines the maximum dose that can be selected and dispensed by the injection device. By placing the limiter stop feature at a predefined position on the housing side wall that corresponds to the positional state of the dose tracker, in particular the tracking stop feature, the maximum dose that can be dispensed by the injection device can be defined.
典型地,限制器可通过至少第一紧固特征件附接到外壳的侧壁。在此,第一紧固特征件可以提供双重功能。首先,第一紧固特征件可以提供将限制器附接到外壳侧壁的相应部分上的紧固结构。此外,第一紧固特征件在侧壁上的位置限定了剂量跟踪器的最大剂量位置状态。Typically, the restraint can be attached to the side wall of the housing by at least a first fastening feature. Here, the first fastening feature may serve a dual function. First, the first fastening feature may provide a fastening structure for attaching the restraint to a corresponding portion of the side wall of the housing. Furthermore, the location of the first securing feature on the sidewall defines the maximum dose position state of the dose tracker.
可将限制器从外壳外侧附接到侧壁。即使在递送给患者或医务人员之后,这也允许修改注射装置的最大剂量规格。将限制器从外壳外侧附接到外壳使得能够随后在无需打开注射装置的外壳的情况下修改注射装置。The restrictor can be attached to the side wall from the outside of the housing. This allows modification of the maximum dose specification of the injection device even after delivery to the patient or medical staff. Attaching the restrictor to the housing from outside the housing enables subsequent modification of the injection device without opening the housing of the injection device.
在另外的实施例中,外壳特别是外壳的侧壁包括至少第二紧固特征件,所述第二紧固特征件相对于外壳的轴向方向和周向方向之一与至少第一紧固特征件分离。在此,限制器选择性地可与至少第一和第二紧固特征件之一接合。第二紧固特征件可以与第一紧固特征件同样地成形和配置。第二紧固特征件仅通过其在外壳侧壁上的位置或定位而区别于第一紧固特征件。特征件和第二紧固特征件。第一和第二紧固特征件相对于外壳的轴向方向(z)和周向或切向方向(w)之一彼此分离。In further embodiments, the housing, in particular the sidewall of the housing, includes at least a second fastening feature that is connected to at least the first fastening with respect to one of an axial direction and a circumferential direction of the housing. Feature separation. Here, the limiter is selectively engageable with at least one of the first and second fastening features. The second fastening feature may be shaped and configured the same as the first fastening feature. The second fastening feature differs from the first fastening feature only by its location or positioning on the side wall of the housing. feature and a second fastening feature. The first and second fastening features are separated from each other with respect to one of an axial direction (z) and a circumferential or tangential direction (w) of the housing.
下文和上文关于第一紧固特征件所描述的所有特征和效果可以同样适用于第二紧固特征件。此外,以下仅相对于第一和第二紧固特征件之一或相对于第一和第二紧固特征件二者所描述的特征和效果同样适用于其中注射装置仅包括外壳侧壁上的单个即第一紧固特征件的实施例。All features and effects described below and above with respect to the first fastening feature may equally apply to the second fastening feature. Furthermore, the features and effects described below with respect to only one of the first and second fastening features or with respect to both the first and second fastening features are equally applicable where the injection device comprises only a Embodiment of a single or first fastening feature.
典型地,限制器原则上可与至少第一和第二紧固特征件二者接合。一次仅可以与第一和第二紧固特征件之一连接或接合。例如,限制器可以与第一紧固特征件接合而第二紧固特征件保持脱离。在该配置中,限制器和限制器在第一紧固特征件处至侧壁的附接限定了注射装置的第一最大剂量规格。根据需要以及如果需要,限制器可以从第一紧固特征件上拆下,并可以附接到第二紧固特征件。然后,与第一紧固特征件相比,限制器及其限制器止挡特征件位于外壳侧壁上的不同位置。在该第二配置中,限制器以及限制器至侧壁和第二紧固特征件的附接限定了注射装置的第二最大剂量规格,由于第一和第二紧固特征件的空间分离,所述第二最大剂量规格不同于第一最大剂量规格。Typically, the limiter is in principle engageable with both at least the first and second fastening features. Only one of the first and second fastening features can be connected or engaged at a time. For example, the limiter may engage with the first fastening feature while the second fastening feature remains disengaged. In this configuration, the restrictor and the attachment of the restrictor to the side wall at the first fastening feature define a first maximum dose specification of the injection device. The limiter can be detached from the first fastening feature and can be attached to the second fastening feature as needed and if desired. The limiter and its limiter stop feature are then located at a different location on the housing sidewall than the first fastening feature. In this second configuration, the restrictor and the attachment of the restrictor to the side wall and the second securing feature define a second maximum dose specification of the injection device, due to the spatial separation of the first and second securing features, The second maximum dosage strength is different from the first maximum dosage strength.
在另一个实施例中,第一和第二紧固特征件中的至少一个包括延伸穿过外壳侧壁的贯通开口。在此,限制器止挡特征件被成形和配置为延伸穿过贯通开口。在外壳的侧壁中提供至少一个贯通开口提供了将限制器附接到外壳的外表面并且因此从外壳的外侧附接的可能性。由于贯通开口和限制器止挡特征件是互补的或对应地成形的,因此限制器止挡特征件可从外侧穿过侧壁中的贯通开口插入,以便限制器止挡特征件的自由端从侧壁的内表面突起。在那里,限制器止挡特征件可以与对应地成形的跟踪止挡特征件可操作地接合,以阻止和阻挡剂量跟踪器的旋转和/或平移移位超过预定义的最大剂量位置状态。In another embodiment, at least one of the first and second fastening features includes a through opening extending through the sidewall of the housing. Here, the limiter stop feature is shaped and configured to extend through the through opening. Providing at least one through opening in the side wall of the housing provides the possibility to attach the restrictor to the outer surface of the housing and thus from the outside of the housing. Since the through opening and the limiter stop feature are complementary or correspondingly shaped, the limiter stop feature can be inserted from the outside through the through opening in the side wall so that the free end of the limiter stop feature is free from The inner surface of the side wall protrudes. There, the limiter stop feature may be operatively engaged with a correspondingly shaped tracking stop feature to prevent and block rotational and/or translational displacement of the dose tracker beyond a predefined maximum dose position state.
典型地并且根据另一个实施例,至少第一和第二紧固特征件中的每一个均包括延伸穿过侧壁的贯通开口。此外,第一和第二紧固特征件可包括相同或同样的几何形状。以这种方式,同一个限制器可以选择性地附接到第一和第二紧固特征件之一。这提供并使得能够重新配置注射装置。当例如附接到第一紧固特征件从而限定第一最大剂量规格时,限制器可以从第一紧固特征件上拆下并且可以附接到第二紧固特征件,以便限定不同于第一最大剂量规格的第二最大剂量规格。Typically and according to another embodiment, each of at least the first and second fastening features includes a through opening extending through the side wall. Furthermore, the first and second fastening features may comprise the same or the same geometry. In this way, the same limiter can be selectively attached to one of the first and second fastening features. This provides and enables reconfiguration of the injection device. When, for example, attached to the first fastening feature to define a first maximum dose size, the limiter is detachable from the first fastening feature and attachable to the second fastening feature to define a different A second maximum dose strength of a maximum dose strength.
在另一个实施例中,限制器包括基底部分。限制器止挡特征件从基底部分突起。当附接到侧壁时,基底部分可以位于侧壁外侧或可以位于侧壁内侧。基底部分可以被配置为提供对限制器的明确定义的抓握的手柄。基底部分还可以改进限制器到侧壁的紧固。基底部分可以永久地附接到侧壁,例如通过粘合剂的方式或通过焊接的方式。In another embodiment, the restrictor includes a base portion. A limiter stop feature protrudes from the base portion. When attached to the sidewall, the base portion may be located outside the sidewall or may be located inside the sidewall. The base portion may be configured as a handle that provides a well-defined grip on the limiter. The base portion may also improve the securing of the restrictor to the side wall. The base portion may be permanently attached to the side wall, eg by means of adhesive or by welding.
当基底部分附接到第一和第二紧固特征件之一时,它可以成形为覆盖第一和第二紧固特征件。因此,基底部分可以包括盖,所述盖的尺寸至少等于或大于外壳侧壁上的第一和第二紧固特征件之间的距离。以这种方式并且当例如借助于第一限制器止挡特征件附接到第一紧固特征件时,基底部分覆盖第一紧固特征件和第二紧固特征件二者。在另一种配置中并且当例如借助于限制器止挡特征件附接到第二紧固特征件时,基底部分也覆盖第一紧固特征件和第二紧固特征件二者。When the base portion is attached to one of the first and second fastening features, it may be shaped to cover the first and second fastening features. Accordingly, the base portion may include a cover having dimensions at least equal to or greater than the distance between the first and second fastening features on the sidewall of the housing. In this manner and when attached to the first fastening feature, eg by means of the first limiter stop feature, the base portion covers both the first and second fastening features. In another configuration and when attached to the second fastening feature, eg by means of a limiter stop feature, the base portion also covers both the first and second fastening features.
对于任一种配置,也可想到,借助于基底部分将限制器附接到外壳的外表面。在另一个实施例中,第一和第二紧固特征件的至少一个贯通开口的截面可以大于限制器的限制器止挡特征件的外截面。这使得能够将限制器止挡特征件相当平滑且容易地插入到第一或第二紧固特征件的贯通开口中并且穿过所述贯通开口。为了重新配置注射装置及其剂量设定机构,也可以很容易地从第一或第二紧固特征件的贯通开口上拆下和抽出限制器止挡特征件。For either configuration, it is also conceivable to attach the limiter to the outer surface of the housing by means of the base portion. In another embodiment, the cross-section of the at least one through opening of the first and second fastening features may be larger than the outer cross-section of the limiter stop feature of the limiter. This enables a fairly smooth and easy insertion of the limiter stop feature into and through the through opening of the first or second fastening feature. The limiter stop feature can also be easily removed and withdrawn from the through opening of the first or second fastening feature in order to reconfigure the injection device and its dose setting mechanism.
在另一个实施例中,基底部分可附接到侧壁的外表面。由于限制器止挡特征件从基底部分突起,所以基底部分可位于侧壁的外侧或外表面,而限制器止挡特征件延伸穿过侧壁并从侧壁的内表面突起以便与剂量跟踪器的跟踪止挡特征件接合。至基底部分的外表面的附接还有利于永久地访问基底部分以及因此限制器。由于基底部分保留在外表面处,因此使用者或医务人员均可访问,从而使得能够将限制器从侧壁上拆下,以便在需要时重新配置注射装置。In another embodiment, the base portion may be attached to the outer surface of the sidewall. Since the limiter stop feature protrudes from the base portion, the base portion may be located on the outside or outer surface of the sidewall, while the limiter stop feature extends through the sidewall and protrudes from the inner surface of the sidewall to interface with the dose tracker of the tracking stop feature engages. The attachment to the outer surface of the base portion also facilitates permanent access to the base portion and thus the limiter. Since the base portion remains at the outer surface, it is accessible to the user or medical personnel, enabling the restrictor to be removed from the side wall in order to reconfigure the injection device if needed.
在另一个实施例中,限制器止挡特征件可插入并穿过第一和第二紧固特征件的至少一个贯通开口,使得限制器止挡特征件的背向基底部分的自由端从侧壁的内表面向内突起。因此,限制器止挡特征件的伸长(典型地,径向伸长)大于侧壁的厚度,并且也大于延伸穿过侧壁的贯通开口的厚度。以这种方式,将基底部分附接和邻接到侧壁的外表面导致一种配置,在这种配置中限制器止挡特征件的自由端从侧壁的内表面向内突起。In another embodiment, the limiter stop feature is insertable through the at least one through opening of the first and second fastening features such that the free end of the limiter stop feature facing away from the base portion is from the side The inner surface of the wall protrudes inwardly. Accordingly, the extension (typically, radial extension) of the limiter stop feature is greater than the thickness of the sidewall, and is also greater than the thickness of the through opening extending through the sidewall. In this manner, attaching and abutting the base portion to the outer surface of the sidewall results in a configuration in which the free end of the limiter stop feature protrudes inwardly from the inner surface of the sidewall.
在另一个实施例中,第一和第二紧固特征件中的至少一个包括在侧壁的外表面上的凹入部分。凹入部分被布置成与第一和/或第二紧固特征件的至少一个贯通开口相邻。典型地,凹入部分被成形和配置为将限制器的基底部分接收在其中。以这种方式,基底部分的面向外部的表面可以与外壳的侧壁的外表面齐平。实际上,当限制器附接到第一和第二紧固特征件之一时,并且当限制器特别是其基底部分覆盖至少第一和第二紧固特征件二者时,外壳的外表面可以包括没有突起或凹部的相当均匀且光滑的形状。In another embodiment, at least one of the first and second fastening features includes a recessed portion on the outer surface of the sidewall. The recessed portion is arranged adjacent to the at least one through opening of the first and/or second fastening feature. Typically, the recessed portion is shaped and configured to receive the base portion of the restrictor therein. In this way, the outwardly facing surface of the base portion may be flush with the outer surface of the side wall of the housing. Indeed, when the limiter is attached to one of the first and second fastening features, and when the limiter, in particular its base portion, covers at least both the first and second fastening features, the outer surface of the housing may Comprising a fairly uniform and smooth shape with no protrusions or recesses.
在另一个实施例中,凹入部分的深度等于或大于基底部分的厚度。以这种方式,基底部分可以完全接收在凹入部分内侧。当附接到并布置在凹入部分中时,基底部分不从侧壁的外表面突起。这提供了外壳的外圆周的相当均匀且光滑的形状。In another embodiment, the depth of the recessed portion is equal to or greater than the thickness of the base portion. In this way, the base portion can be received completely inside the recessed portion. When attached to and disposed in the recessed portion, the base portion does not protrude from the outer surface of the side wall. This provides a fairly uniform and smooth shape of the outer circumference of the casing.
在另一个实施例中,第一紧固特征件和第二紧固特征件各自包括凹入部分。在此,第一紧固特征件的和第二紧固特征件的凹入部分相互合并并在侧壁的外表面上形成细长凹部。典型地,在侧壁的外表面上的细长凹部为限制器的基底部分提供了一种容器。当限制器的基底部分插入到细长凹部中时,其限制器止挡特征件可延伸进入并穿过相应的第一或第二紧固特征件的贯通开口。In another embodiment, the first fastening feature and the second fastening feature each include a recessed portion. Here, the recessed portions of the first fastening feature and the second fastening feature merge with each other and form an elongated recess on the outer surface of the side wall. Typically, the elongated recess on the outer surface of the side wall provides a kind of receptacle for the base portion of the restrictor. When the base portion of the limiter is inserted into the elongated recess, its limiter stop feature can extend into and through the through opening of the corresponding first or second fastening feature.
在另一个实施例中,细长凹部沿轴向方向和/或沿周向方向延伸。第一和第二紧固特征件之一的至少一个贯通开口被切向地或周向地或轴向地布置为与细长凹部相邻。如果细长凹部沿轴向方向延伸,则贯通开口被切向地或周向地布置为与细长凹部相邻。当细长凹部沿周向或切向方向延伸时,第一和第二紧固特征件之一的至少一个贯通开口被轴向地布置为与细长凹部相邻。In another embodiment, the elongated recesses extend in the axial direction and/or in the circumferential direction. At least one through opening of one of the first and second fastening features is disposed tangentially or circumferentially or axially adjacent the elongated recess. If the elongated recesses extend in the axial direction, the through-openings are arranged tangentially or circumferentially adjacent to the elongated recesses. The at least one through opening of one of the first and second fastening features is axially disposed adjacent the elongated recess when the elongated recess extends in a circumferential or tangential direction.
典型地,第一和第二紧固特征件各自包括贯通开口,其中这些贯通开口轴向地彼此分离。两个贯通开口被周向地布置为与细长凹部相邻。典型地,限制器被成形为附接到细长凹部的一部分并仅与第一和第二紧固特征件的贯通开口之一接合。然后,另一个贯通开口和细长凹部的剩余部分可以保持未被覆盖。该残留部分或未占用的贯通开口可以用单独的盖单独覆盖。Typically, the first and second fastening features each include through openings, wherein the through openings are axially separated from each other. Two through-openings are arranged circumferentially adjacent the elongated recess. Typically, the limiter is shaped to attach to a portion of the elongated recess and engage only one of the through openings of the first and second fastening features. The remainder of the other through opening and the elongated recess can then remain uncovered. This residual portion or unoccupied through-opening can be covered separately with a separate cover.
在另一个实施例中,可以设置一组至少两个形状不同的限制器。至少两个限制器可包括被配置为完全填充细长凹部的相等尺寸的基底部分。两个限制器可以通过它们的至少一个限制器止挡特征件相对于基底部分的形状和位置区别于彼此。第一限制器可包括被配置为当限制器附接到外壳的侧壁时与第一紧固特征件接合的限制器止挡特征件。第二限制器可包括限制器止挡特征件,所述限制器止挡特征件被专门配置为当限制器附接到外壳的侧壁时与第二紧固特征件接合。In another embodiment, a set of at least two differently shaped restrictors may be provided. The at least two restraints may include equally sized base portions configured to completely fill the elongated recess. The two restraints may be distinguished from each other by the shape and position of their at least one restraint stop feature relative to the base portion. The first limiter may include a limiter stop feature configured to engage the first securing feature when the limiter is attached to the sidewall of the housing. The second limiter may include a limiter stop feature that is specifically configured to engage the second securing feature when the limiter is attached to the sidewall of the housing.
以这种方式,具有同样地成形的基底部分的第一和第二限制器可以总是完全填充并覆盖侧壁的外表面上的细长凹部。依据第一和第二限制器之一的特定选择,第一和第二紧固特征件中的仅一个以及穿过侧壁的相应的第一和第二贯通开口中的仅一个将被限制器止挡特征件占用。In this way, the first and second restrictors with likewise shaped base portions can always completely fill and cover the elongated recesses on the outer surface of the side wall. Depending on the particular choice of one of the first and second restraints, only one of the first and second fastening features and only one of the corresponding first and second through openings through the sidewall will be restrained Stop feature occupancy.
在另一个实施例中,可以仅设置一个限制器,所述限制器的基底部分关于对称轴线对称。在此,在侧壁的外表面上的凹入部分可以包括相应的对称性,使得限制器在第一配置和第二配置中可附接和可紧固到外壳的外表面,其中第二配置与第一配置相比例如旋转了180°。典型地,对称轴线与第一和第二紧固特征件之间的距离的中心重合。以这种方式,当处于第一配置时限制器的限制器止挡特征件接合第一紧固特征件,并且当处于第二配置时,例如当限制器旋转约180°时限制器止挡特征件接合第二紧固特征件。In another embodiment, only one limiter may be provided, the base portion of which is symmetrical about the axis of symmetry. Here, the recessed portion on the outer surface of the side wall may include corresponding symmetry such that the restrictor is attachable and fastenable to the outer surface of the housing in a first configuration and a second configuration, wherein the second configuration Compared to the first configuration, it is for example rotated by 180°. Typically, the axis of symmetry coincides with the center of the distance between the first and second fastening features. In this manner, the limiter stop feature of the limiter engages the first securing feature when in the first configuration, and the limiter stop feature when in the second configuration, such as when the limiter is rotated about 180° The piece engages the second fastening feature.
在另一个实施例中,基底部分是L形的并且包括第一支腿和第二支腿。第一和第二支腿相对于彼此成90°延伸。当附接到外壳的侧壁时,第一支腿可以沿侧壁的周向方向延伸,并且第二支腿可以沿侧壁的轴向方向延伸。当细长凹部沿轴向方向延伸时,第二支腿可以沿细长凹部延伸,并且第一支腿可以沿侧壁的圆周垂直于其延伸。In another embodiment, the base portion is L-shaped and includes a first leg and a second leg. The first and second legs extend at 90° relative to each other. When attached to the side wall of the housing, the first leg may extend in the circumferential direction of the side wall and the second leg may extend in the axial direction of the side wall. When the elongated recess extends in the axial direction, the second leg may extend along the elongated recess, and the first leg may extend perpendicular to the circumference of the side wall.
L形的基底部分有利于将限制器附接到侧壁。第二支腿可以附接到并且可以接收在侧壁的外表面上的凹入部分或细长凹部中,从而限定限制器相对于外壳的径向位置。第一支腿可以支撑限制器的至少一个限制器止挡特征件,所述限制器止挡特征件径向向内延伸到外壳的内部。在此,具有至少一个限制器止挡特征件附接到其上的第一支腿突起到外壳中并与剂量跟踪器的跟踪止挡特征件接合,而第二支腿则在外壳的明确定义的径向位置处提供了限制器的明确定义的附接。The L-shaped base portion facilitates attachment of the restrictor to the side wall. The second leg can be attached to and can be received in a recessed portion or elongated recess on the outer surface of the side wall, thereby defining the radial position of the limiter relative to the housing. The first leg may support at least one limiter stop feature of the limiter that extends radially inwardly to the interior of the housing. Here, a first leg having at least one limiter stop feature attached thereto protrudes into the housing and engages with the tracking stop feature of the dose tracker, while the second leg is well defined in the housing Provides a well-defined attachment of the limiter at the radial position of the .
在另外的实施例中,限制器止挡特征件从第一支腿的背向第二支腿的端部突起。在此,限制器止挡特征件可以构成第一支腿,而第二支腿可以被视为限制器的沿外壳的侧壁延伸的凸缘部分。In further embodiments, the limiter stop feature protrudes from the end of the first leg that faces away from the second leg. Here, the limiter stop feature may constitute the first leg, while the second leg may be considered a flange portion of the limiter extending along the side wall of the housing.
在另外的实施例中,注射装置配备有可附接到侧壁的外表面上的盖。盖被配置为至少覆盖第一紧固特征件和第二紧固特征件。盖还可被配置为覆盖侧壁的外表面上与第一和/或第二紧固特征件的至少一个贯通开口相邻的凹入部分。当附接到外壳的侧壁时,盖有效地隐藏和掩盖限制器的位置和存在。In further embodiments, the injection device is equipped with a cap attachable to the outer surface of the side wall. The cover is configured to cover at least the first fastening feature and the second fastening feature. The cover may also be configured to cover a recessed portion of the outer surface of the sidewall adjacent the at least one through opening of the first and/or second fastening feature. When attached to the side wall of the housing, the cover effectively hides and conceals the location and presence of the restrictor.
盖可以包括柔性箔,所述柔性箔可粘附地附接到外壳侧壁的外表面。盖可以是透明的。替代性地,盖可以是不透明的。盖可以包括印刷表面。盖可以包括附接到外壳外表面的标签。标签可以包含关于药剂的使用和/或关于注射装置的使用的说明或信息。The cover may include a flexible foil that is adhesively attached to the outer surface of the sidewall of the housing. The cover may be transparent. Alternatively, the cover may be opaque. The cover may include a printed surface. The cover may include a label attached to the outer surface of the housing. The label may contain instructions or information regarding the use of the medicament and/or regarding the use of the injection device.
代替柔性箔,盖可以包括由塑料材料制成或由金属制成的套管或壳体。盖可以与细长外壳积极地接合。盖和外壳可以包括相互对应的紧固特征件,诸如突起和对应地成形的凹部。盖和外壳的侧壁可以包括相互接合的夹持或卡扣特征件,其提供盖到外壳侧壁的可拆卸或不可拆卸的紧固。Instead of a flexible foil, the cover may comprise a sleeve or housing made of plastic material or metal. The cover can be positively engaged with the elongated housing. The cover and housing may include mutually corresponding fastening features, such as protrusions and correspondingly shaped recesses. The lid and the side walls of the housing may include interengaging clip or snap features that provide releasable or non-removable securing of the lid to the side walls of the housing.
例如,盖可以包括呈套管或圆柱体的一部分形式的壳体,例如半壳。盖可以包括在内表面部分上的径向突起或径向凹部,以与设置在注射装置外壳侧壁的外表面上的对应地成形的凹部或突起接合。以这种方式,盖可以被夹持或固定到外壳的侧壁。当盖包括塑料壳体时,盖的外表面可以包括标签。盖也可以借助于粘合剂或借助于胶粘标签紧固到注射装置。还可以想到借助于焊接将盖附接到外壳。For example, the cover may comprise a casing in the form of a sleeve or part of a cylinder, such as a half-shell. The cover may include radial projections or radial recesses on the inner surface portion for engagement with correspondingly shaped recesses or projections provided on the outer surface of the side wall of the injection device housing. In this way, the cover can be clamped or fixed to the side wall of the housing. When the cover includes a plastic housing, the outer surface of the cover may include a label. The cap can also be fastened to the injection device by means of adhesive or by means of an adhesive label. It is also conceivable to attach the cover to the housing by means of welding.
在另一个实施例中,注射装置还包括活塞杆。活塞杆典型地是驱动机构和/或剂量设定机构的部件。活塞杆可以轴向地移位以从药筒分配一定剂量的药剂。当注射装置处于分配模式时,驱动机构和/或其剂量设定机构被配置为在轴向远侧方向上纵向地驱动和移位活塞杆。In another embodiment, the injection device further includes a piston rod. The piston rod is typically part of the drive mechanism and/or the dose setting mechanism. The piston rod is axially displaceable to dispense a dose of medicament from the cartridge. When the injection device is in the dispensing mode, the drive mechanism and/or its dose setting mechanism is configured to drive and displace the piston rod longitudinally in the axial distal direction.
典型地,注射装置设置有填充有药剂(例如,液体药剂)的药筒。药筒典型地借助于塞子在近侧方向上密封。塞子可在药筒内侧轴向地移位,以将液体药剂从其远端排出。药筒的远端典型地由可刺穿密封件密封。可刺穿密封件可借助于双尖注射针刺穿。注射针典型地可以可释放地附接到注射装置的外壳的远端和/或分配端,典型地附接到属于注射装置的外壳的药筒保持器的远端。Typically, an injection device is provided with a cartridge filled with a medicament (eg, a liquid medicament). The cartridge is typically sealed in the proximal direction by means of a stopper. The stopper is axially displaceable inside the cartridge to expel the liquid medicament from its distal end. The distal end of the cartridge is typically sealed by a pierceable seal. The pierceable seal can be pierced by means of a double pointed injection needle. The injection needle is typically releasably attachable to the distal end and/or the dispensing end of the housing of the injection device, typically to the distal end of the cartridge holder belonging to the housing of the injection device.
在另外的实施例中,注射装置包括至少部分地填充有药剂的药筒。药筒包括填充有药剂的桶。药筒以及因此桶在轴向近侧方向上被塞子密封。塞子可借助于活塞杆相对于桶轴向地移位。当在剂量分配期间活塞杆在远侧方向上推进时,它向塞子施加驱动压力。由于药筒固定在外壳内侧,因此塞子开始在远侧方向上移动,由此增加药筒的内部压力,从而导致从药筒中排出药剂。In further embodiments, the injection device includes a cartridge at least partially filled with a medicament. Cartridges include barrels filled with medicament. The cartridge, and thus the barrel, is sealed by the plug in the axial proximal direction. The plug is axially displaceable relative to the barrel by means of the piston rod. When the piston rod is advanced in the distal direction during dose dispensing, it applies driving pressure to the bung. Since the cartridge is secured inside the housing, the stopper begins to move in a distal direction, thereby increasing the internal pressure of the cartridge, resulting in expulsion of the medicament from the cartridge.
在另一方面,提供了一种修改注射装置的最大剂量规格的方法。所述方法包括提供注射装置的步骤。注射装置包括沿轴向方向延伸并被配置为容纳剂量设定机构的细长外壳。典型地,外壳还被配置为接收并容纳填充有药剂的药筒。外壳还包括具有至少第一紧固特征件的侧壁。典型地,侧壁还包括至少第二紧固特征件。注射装置还包括布置在外壳内侧的剂量跟踪器。剂量跟踪器包括至少一个跟踪止挡特征件。剂量跟踪器是在设定剂量期间相对于外壳平移地或旋转地移位中的至少一个。剂量跟踪器相对于外壳的位置状态明确地指示实际设定的剂量规格。In another aspect, a method of modifying a maximum dose specification of an injection device is provided. The method includes the step of providing an injection device. The injection device includes an elongated housing extending in an axial direction and configured to accommodate the dose setting mechanism. Typically, the housing is also configured to receive and house a cartridge filled with a medicament. The housing also includes a sidewall having at least a first fastening feature. Typically, the sidewall also includes at least a second fastening feature. The injection device also includes a dose tracker disposed inside the housing. The dose tracker includes at least one tracking stop feature. The dose tracker is at least one of translationally or rotationally displaced relative to the housing during setting of a dose. The positional state of the dose tracker relative to the housing unambiguously indicates the actual dose specification set.
所述修改最大剂量规格的方法还包括借助于至少一个紧固特征件将限制器附接到外壳侧壁的步骤。限制器包括至少一个限制器止挡特征件,当附接到侧壁时,所述限制器止挡特征件从侧壁向内延伸。限制器止挡特征件和跟踪止挡特征件被配置为相互接合并阻止剂量跟踪器的移位超过预定义的最大剂量位置状态。The method of modifying the maximum dose specification further includes the step of attaching the limiter to the side wall of the housing by means of at least one fastening feature. The limiter includes at least one limiter stop feature that extends inwardly from the sidewall when attached to the sidewall. The limiter stop feature and the tracking stop feature are configured to interengage and prevent displacement of the dose tracker beyond a predefined maximum dose position state.
典型地,所述修改注射装置的最大剂量规格的方法适用于如上所述的注射装置。因此,以上结合注射装置描述的所有特征、益处和效果均同样适用于所述修改注射装置的最大剂量规格的方法。Typically, the method of modifying the maximum dose specification of an injection device is applicable to an injection device as described above. Therefore, all the features, benefits and effects described above in connection with the injection device are equally applicable to the method of modifying the maximum dosage strength of the injection device.
在当前上下文中,术语“远侧”或“远端”涉及注射装置面向人或动物的注射部位的一端。术语“近侧”或“近端”涉及注射装置的相对端,所述相对端距人或动物的注射部位最远。In the present context, the term "distal" or "distal" refers to the end of the injection device facing the injection site of the human or animal. The term "proximal" or "proximal end" refers to the opposite end of the injection device that is furthest from the injection site in the human or animal.
如在本文使用的,术语“药物”或“药剂”意指含有至少一种药用活性化合物的药物配制品,As used herein, the term "drug" or "medicine" means a pharmaceutical formulation containing at least one pharmaceutically active compound,
其中在一个实施方案中,所述药用活性化合物具有高达1500Da的分子量,和/或是肽、蛋白质、多糖、疫苗、DNA、RNA、酶、抗体或抗体片段、激素或寡核苷酸、或上述药用活性化合物的混合物,wherein in one embodiment the pharmaceutically active compound has a molecular weight of up to 1500 Da, and/or is a peptide, protein, polysaccharide, vaccine, DNA, RNA, enzyme, antibody or antibody fragment, hormone or oligonucleotide, or Mixtures of the above-mentioned pharmaceutically active compounds,
其中在另外的实施方案中,所述药用活性化合物可用于治疗和/或预防糖尿病或与糖尿病相关的并发症(如糖尿病视网膜病变)、血栓栓塞症(如深静脉或肺血栓栓塞症)、急性冠状动脉综合征(ACS)、心绞痛、心肌梗死、癌症、黄斑变性、炎症、枯草热、动脉粥样硬化和/或类风湿性关节炎,Wherein in additional embodiments, the pharmaceutically active compound is useful for the treatment and/or prevention of diabetes or complications associated with diabetes (eg, diabetic retinopathy), thromboembolism (eg, deep vein or pulmonary thromboembolism), Acute coronary syndrome (ACS), angina, myocardial infarction, cancer, macular degeneration, inflammation, hay fever, atherosclerosis and/or rheumatoid arthritis,
其中在另外的实施方案中,所述药用活性化合物包括至少一种用于治疗和/或预防糖尿病或与糖尿病相关的并发症(如糖尿病视网膜病变)的肽,wherein, in further embodiments, the pharmaceutically active compound comprises at least one peptide for the treatment and/or prevention of diabetes or diabetes-related complications such as diabetic retinopathy,
其中在另外的实施方案中,所述药用活性化合物包括至少一种人胰岛素或人胰岛素类似物或衍生物、胰高血糖素样肽(GLP-1)或其类似物或衍生物、或毒蜥外泌肽(exendin)-3或毒蜥外泌肽-4、或毒蜥外泌肽-3或毒蜥外泌肽-4的类似物或衍生物。Wherein in further embodiments, the pharmaceutically active compound comprises at least one human insulin or a human insulin analog or derivative, glucagon-like peptide (GLP-1) or an analog or derivative thereof, or a toxic Exendin-3 or Exendin-4, or an analog or derivative of Exendin-3 or Exendin-4.
胰岛素类似物是例如Gly(A21)、Arg(B31)、Arg(B32)人胰岛素;Lys(B3)、Glu(B29)人胰岛素;Lys(B28)、Pro(B29)人胰岛素;Asp(B28)人胰岛素;人胰岛素,其中B28位的脯氨酸被Asp、Lys、Leu、Val或Ala替代,并且其中B29位Lys可以被Pro替代;Ala(B26)人胰岛素;Des(B28-B30)人胰岛素;Des(B27)人胰岛素和Des(B30)人胰岛素。Insulin analogs are eg Gly(A21), Arg(B31), Arg(B32) human insulin; Lys(B3), Glu(B29) human insulin; Lys(B28), Pro(B29) human insulin; Asp(B28) Human insulin; Human insulin in which the proline at position B28 is replaced by Asp, Lys, Leu, Val or Ala, and in which Lys at position B29 can be replaced by Pro; Ala (B26) human insulin; Des (B28-B30) human insulin ; Des(B27) human insulin and Des(B30) human insulin.
胰岛素衍生物是例如B29-N-肉豆蔻酰-des(B30)人胰岛素;B29-N-棕榈酰-des(B30)人胰岛素;B29-N-肉豆蔻酰人胰岛素;B29-N-棕榈酰人胰岛素;B28-N-肉豆蔻酰LysB28ProB29人胰岛素;B28-N-棕榈酰-LysB28ProB29人胰岛素;B30-N-肉豆蔻酰-ThrB29LysB30人胰岛素;B30-N-棕榈酰-ThrB29LysB30人胰岛素;B29-N-(N-棕榈酰-Υ-谷氨酰)-des(B30)人胰岛素;B29-N-(N-石胆酰-Υ-谷氨酰)-des(B30)人胰岛素;B29-N-(ω-羧基十七烷酰)-des(B30)人胰岛素和B29-N-(ω-羧基十七烷酰)人胰岛素。Insulin derivatives are eg B29-N-myristoyl-des(B30) human insulin; B29-N-palmitoyl-des(B30) human insulin; B29-N-myristoyl human insulin; B29-N-palmitoyl Human insulin; B28-N-myristoyl-LysB28ProB29 human insulin; B28-N-palmitoyl-LysB28ProB29 human insulin; B30-N-myristoyl-ThrB29LysB30 human insulin; B30-N-palmitoyl-ThrB29LysB30 human insulin; B29- N-(N-palmitoyl-Y-glutamyl)-des(B30) human insulin; B29-N-(N-lithoyl-Y-glutamyl)-des(B30) human insulin; B29-N -(ω-carboxyheptadecanoyl)-des(B30) human insulin and B29-N-(ω-carboxyheptadecanoyl) human insulin.
毒蜥外泌肽-4例如意指毒蜥外泌肽-4(1-39),一种具有以下序列的肽:H-His-Gly-Glu-Gly-Thr-Phe-Thr-Ser-Asp-Leu-Ser-Lys-Gln-Met-Glu-Glu-Glu-Ala-Val-Arg-Leu-Phe-Ile-Glu-Trp-Leu-Lys-Asn-Gly-Gly-Pro-Ser-Ser-Gly-Ala-Pro-Pro-Pro-Ser-NH2。Exendin-4 means for example Exendin-4(1-39), a peptide having the following sequence: H-His-Gly-Glu-Gly-Thr-Phe-Thr-Ser-Asp -Leu-Ser-Lys-Gln-Met-Glu-Glu-Glu-Ala-Val-Arg-Leu-Phe-Ile-Glu-Trp-Leu-Lys-Asn-Gly-Gly-Pro-Ser-Ser-Gly -Ala-Pro-Pro-Pro-Ser-NH2.
毒蜥外泌肽-4衍生物例如选自以下化合物列表:Exendin-4 derivatives are for example selected from the following list of compounds:
H-(Lys)4-des Pro36,des Pro37毒蜥外泌肽-4(1-39)-NH2、H-(Lys)4-des Pro36, des Pro37 Exendin-4(1-39)-NH2,
H-(Lys)5-des Pro36,des Pro37毒蜥外泌肽-4(1-39)-NH2、H-(Lys)5-des Pro36, des Pro37 Exendin-4(1-39)-NH2,
des Pro36毒蜥外泌肽-4(1-39)、des Pro36 Exendin-4(1-39),
des Pro36[Asp28]毒蜥外泌肽-4(1-39)、des Pro36[Asp28]Exendin-4(1-39),
des Pro36[IsoAsp28]毒蜥外泌肽-4(1-39)、des Pro36[IsoAsp28]Exendin-4(1-39),
des Pro36[Met(O)14,Asp28]毒蜥外泌肽-4(1-39)、des Pro36[Met(O)14,Asp28]Exendin-4(1-39),
des Pro36[Met(O)14,IsoAsp28]毒蜥外泌肽-4(1-39)、des Pro36[Met(O)14,IsoAsp28]Exendin-4(1-39),
des Pro36[Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)、des Pro36[Trp(O2)25,Asp28]Exendin-4(1-39),
des Pro36[Trp(O2)25,IsoAsp28]毒蜥外泌肽-4(1-39)、des Pro36[Trp(O2)25,IsoAsp28]Exendin-4(1-39),
des Pro36[Met(O)14Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)、des Pro36[Met(O)14Trp(O2)25,Asp28]Exendin-4(1-39),
des Pro36[Met(O)14Trp(O2)25,IsoAsp28]毒蜥外泌肽-4(1-39);或des Pro36[Met(O)14Trp(O2)25,IsoAsp28]Exendin-4(1-39); or
des Pro36[Asp28]毒蜥外泌肽-4(1-39)、des Pro36[Asp28]Exendin-4(1-39),
des Pro36[IsoAsp28]毒蜥外泌肽-4(1-39)、des Pro36[IsoAsp28]Exendin-4(1-39),
des Pro36[Met(O)14,Asp28]毒蜥外泌肽-4(1-39)、des Pro36[Met(O)14,Asp28]Exendin-4(1-39),
des Pro36[Met(O)14,IsoAsp28]毒蜥外泌肽-4(1-39)、des Pro36[Met(O)14,IsoAsp28]Exendin-4(1-39),
des Pro36[Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)、des Pro36[Trp(O2)25,Asp28]Exendin-4(1-39),
des Pro36[Trp(O2)25,IsoAsp28]毒蜥外泌肽-4(1-39)、des Pro36[Trp(O2)25,IsoAsp28]Exendin-4(1-39),
des Pro36[Met(O)14Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)、des Pro36[Met(O)14Trp(O2)25,Asp28]Exendin-4(1-39),
des Pro36[Met(O)14Trp(O2)25,IsoAsp28]毒蜥外泌肽-4(1-39)、des Pro36[Met(O)14Trp(O2)25,IsoAsp28]Exendin-4(1-39),
其中基团-Lys6-NH2可以与毒蜥外泌肽-4衍生物的C-端结合;Wherein the group -Lys6-NH2 can be combined with the C-terminus of the exendin-4 derivative;
或具有以下序列的毒蜥外泌肽-4衍生物:or an exendin-4 derivative having the following sequence:
des Pro36毒蜥外泌肽-4(1-39)-Lys6-NH2(AVE0010)、des Pro36 Exendin-4(1-39)-Lys6-NH2(AVE0010),
H-(Lys)6-des Pro36[Asp28]毒蜥外泌肽-4(1-39)-Lys6-NH2、H-(Lys)6-des Pro36[Asp28]Exendin-4(1-39)-Lys6-NH2,
des Asp28 Pro36,Pro37,Pro38毒蜥外泌肽-4(1-39)-NH2、des Asp28 Pro36, Pro37, Pro38 Exendin-4(1-39)-NH2,
H-(Lys)6-des Pro36,Pro38[Asp28]毒蜥外泌肽-4(1-39)-NH2、H-(Lys)6-des Pro36, Pro38[Asp28]Exendin-4(1-39)-NH2,
H-Asn-(Glu)5des Pro36,Pro37,Pro38[Asp28]毒蜥外泌肽-4(1-39)-NH2、H-Asn-(Glu)5des Pro36,Pro37,Pro38[Asp28]Exendin-4(1-39)-NH2,
des Pro36,Pro37,Pro38[Asp28]毒蜥外泌肽-4(1-39)-(Lys)6-NH2、des Pro36,Pro37,Pro38[Asp28]Exendin-4(1-39)-(Lys)6-NH2,
H-(Lys)6-des Pro36,Pro37,Pro38[Asp28]毒蜥外泌肽-4(1-39)-(Lys)6-NH2、H-(Lys)6-des Pro36,Pro37,Pro38[Asp28]Exendin-4(1-39)-(Lys)6-NH2,
H-Asn-(Glu)5-des Pro36,Pro37,Pro38[Asp28]毒蜥外泌肽-4(1-39)-(Lys)6-NH2、H-Asn-(Glu)5-des Pro36,Pro37,Pro38[Asp28]Exendin-4(1-39)-(Lys)6-NH2,
H-(Lys)6-des Pro36[Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)-Lys6-NH2、H-(Lys)6-des Pro36[Trp(O2)25,Asp28]Exendin-4(1-39)-Lys6-NH2,
H-des Asp28 Pro36,Pro37,Pro38[Trp(O2)25]毒蜥外泌肽-4(1-39)-NH2、H-des Asp28 Pro36, Pro37, Pro38[Trp(O2)25]Exendin-4(1-39)-NH2,
H-(Lys)6-des Pro36,Pro37,Pro38[Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)-NH2、H-(Lys)6-des Pro36,Pro37,Pro38[Trp(O2)25,Asp28]Exendin-4(1-39)-NH2,
H-Asn-(Glu)5-des Pro36,Pro37,Pro38[Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)-NH2、H-Asn-(Glu)5-des Pro36,Pro37,Pro38[Trp(O2)25,Asp28]Exendin-4(1-39)-NH2,
des Pro36,Pro37,Pro38[Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)-(Lys)6-NH2、des Pro36,Pro37,Pro38[Trp(O2)25,Asp28]Exendin-4(1-39)-(Lys)6-NH2,
H-(Lys)6-des Pro36,Pro37,Pro38[Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)-(Lys)6-NH2、H-(Lys)6-des Pro36,Pro37,Pro38[Trp(O2)25,Asp28]Exendin-4(1-39)-(Lys)6-NH2,
H-Asn-(Glu)5-des Pro36,Pro37,Pro38[Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)-(Lys)6-NH2、H-Asn-(Glu)5-des Pro36,Pro37,Pro38[Trp(O2)25,Asp28]Exendin-4(1-39)-(Lys)6-NH2,
H-(Lys)6-des Pro36[Met(O)14,Asp28]毒蜥外泌肽-4(1-39)-Lys6-NH2、H-(Lys)6-des Pro36[Met(O)14,Asp28]Exendin-4(1-39)-Lys6-NH2,
des Met(O)14Asp28 Pro36,Pro37,Pro38毒蜥外泌肽-4(1-39)-NH2、des Met(O)14Asp28 Pro36,Pro37,Pro38Exendin-4(1-39)-NH2,
H-(Lys)6-desPro36,Pro37,Pro38[Met(O)14,Asp28]毒蜥外泌肽-4(1-39)-NH2、H-(Lys)6-desPro36,Pro37,Pro38[Met(O)14,Asp28]Exendin-4(1-39)-NH2,
H-Asn-(Glu)5-des Pro36,Pro37,Pro38[Met(O)14,Asp28]毒蜥外泌肽-4(1-39)-NH2、H-Asn-(Glu)5-des Pro36,Pro37,Pro38[Met(O)14,Asp28]Exendin-4(1-39)-NH2,
des Pro36,Pro37,Pro38[Met(O)14,Asp28]毒蜥外泌肽-4(1-39)-(Lys)6-NH2、des Pro36,Pro37,Pro38[Met(O)14,Asp28]Exendin-4(1-39)-(Lys)6-NH2,
H-(Lys)6-des Pro36,Pro37,Pro38[Met(O)14,Asp28]毒蜥外泌肽-4(1-39)-(Lys)6-NH2、H-(Lys)6-des Pro36,Pro37,Pro38[Met(O)14,Asp28]Exendin-4(1-39)-(Lys)6-NH2,
H-Asn-(Glu)5 des Pro36,Pro37,Pro38[Met(O)14,Asp28]毒蜥外泌肽-4(1-39)-(Lys)6-NH2、H-Asn-(Glu)5 des Pro36,Pro37,Pro38[Met(O)14,Asp28]Exendin-4(1-39)-(Lys)6-NH2,
H-Lys6-des Pro36[Met(O)14,Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)-Lys6-NH2、H-Lys6-des Pro36[Met(O)14,Trp(O2)25,Asp28]Exendin-4(1-39)-Lys6-NH2,
H-des Asp28 Pro36,Pro37,Pro38[Met(O)14,Trp(O2)25]毒蜥外泌肽-4(1-39)-NH2、H-des Asp28 Pro36,Pro37,Pro38[Met(O)14,Trp(O2)25]Exendin-4(1-39)-NH2,
H-(Lys)6-des Pro36,Pro37,Pro38[Met(O)14,Asp28]毒蜥外泌肽-4(1-39)-NH2、H-(Lys)6-des Pro36,Pro37,Pro38[Met(O)14,Asp28]Exendin-4(1-39)-NH2,
H-Asn-(Glu)5-des Pro36,Pro37,Pro38[Met(O)14,Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)-NH2、H-Asn-(Glu)5-des Pro36,Pro37,Pro38[Met(O)14,Trp(O2)25,Asp28]Exendin-4(1-39)-NH2,
des Pro36,Pro37,Pro38[Met(O)14,Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)-(Lys)6-NH2、des Pro36,Pro37,Pro38[Met(O)14,Trp(O2)25,Asp28]Exendin-4(1-39)-(Lys)6-NH2,
H-(Lys)6-des Pro36,Pro37,Pro38[Met(O)14,Trp(O2)25,Asp28]毒蜥外泌肽-4(S1-39)-(Lys)6-NH2、H-(Lys)6-des Pro36,Pro37,Pro38[Met(O)14,Trp(O2)25,Asp28]Exendin-4(S1-39)-(Lys)6-NH2,
H-Asn-(Glu)5-des Pro36,Pro37,Pro38[Met(O)14,Trp(O2)25,Asp28]毒蜥外泌肽-4(1-39)-(Lys)6-NH2;H-Asn-(Glu)5-des Pro36,Pro37,Pro38[Met(O)14,Trp(O2)25,Asp28]Exendin-4(1-39)-(Lys)6-NH2;
或上述任一毒蜥外泌肽-4衍生物的药学上可接受的盐或溶剂化物。or a pharmaceutically acceptable salt or solvate of any of the above Exendin-4 derivatives.
激素是例如如列于Rote Liste,2008年版第50章中的脑垂体激素或下丘脑激素或调节活性肽及其拮抗剂,如促性腺激素(Gonadotropine)(促卵泡激素(Follitropin)、促黄体素、绒毛膜促性腺激素(Choriongonadotropin)、促配子成熟激素)、生长激素(Somatropine)(促生长激素(Somatropin))、去氨加压素、特利加压素、戈那瑞林、曲普瑞林、亮丙瑞林、布舍瑞林、那法瑞林、戈舍瑞林。Hormones are, for example, pituitary or hypothalamic hormones as listed in
多糖是例如糖胺聚糖、透明质酸、肝素、低分子量肝素或超低分子量肝素或其衍生物、或上述多糖的硫酸化形式(例如多硫酸化形式)、和/或其药学上可接受的盐。多硫酸化低分子量肝素的药学上可接受的盐的例子是依诺肝素钠。Polysaccharides are, for example, glycosaminoglycans, hyaluronic acid, heparin, low molecular weight heparin or ultra low molecular weight heparin or derivatives thereof, or sulfated forms (eg, polysulfated forms) of the foregoing polysaccharides, and/or pharmaceutically acceptable of salt. An example of a pharmaceutically acceptable salt of polysulfated low molecular weight heparin is enoxaparin sodium.
抗体是球状血浆蛋白(约150kDa),也称为共享基本结构的免疫球蛋白。由于它们在氨基酸残基上添加了糖链,因此是糖蛋白。每种抗体的基本功能单元是免疫球蛋白(Ig)单体(仅含有一个Ig单元);分泌的抗体也可以是具有两个Ig单位的二聚体(如IgA)、具有四个Ig单位的四聚体(如硬骨鱼IgM)、或具有五个Ig单位的五聚体(如哺乳动物IgM)。Antibodies are globular plasma proteins (about 150 kDa), also known as immunoglobulins that share a basic structure. Since they have sugar chains added to amino acid residues, they are glycoproteins. The basic functional unit of each antibody is an immunoglobulin (Ig) monomer (containing only one Ig unit); secreted antibodies can also be dimers with two Ig units (eg IgA), four Ig units Tetramers (eg, teleost IgM), or pentamers (eg, mammalian IgM) with five Ig units.
Ig单体是由四条多肽链组成的“Y”形分子;两条相同的重链和两条相同的轻链通过半胱氨酸残基之间的二硫键连接。每个重链长约440个氨基酸;每条轻链长约220个氨基酸。重链和轻链各自包含稳定其折叠的链内二硫键。每条链由名为Ig结构域的结构域构成。这些结构域包含约70-110个氨基酸,并根据其大小和功能分为不同类别(例如可变区或V区和恒定区或C区)。这些结构域具有独特的免疫球蛋白折叠,其中两个β折叠成“三明治”状,通过保守半胱氨酸和其他带电氨基酸之间的相互作用保持在一起。Ig monomers are "Y" shaped molecules composed of four polypeptide chains; two identical heavy chains and two identical light chains are linked by disulfide bonds between cysteine residues. Each heavy chain is about 440 amino acids long; each light chain is about 220 amino acids long. The heavy and light chains each contain intrachain disulfide bonds that stabilize their folding. Each chain is made up of domains called Ig domains. These domains comprise approximately 70-110 amino acids and are grouped into different classes (eg, variable or V regions and constant or C regions) according to their size and function. These domains have a unique immunoglobulin fold in which two beta folds form a "sandwich" shape, held together by interactions between conserved cysteines and other charged amino acids.
有五种类型的哺乳动物Ig重链,由α、δ、ε、γ和μ表示。存在的重链的类型定义了抗体的同种型;这些链分别在IgA、IgD、IgE、IgG和IgM抗体中发现。There are five types of mammalian Ig heavy chains, represented by alpha, delta, epsilon, gamma, and mu. The type of heavy chain present defines the antibody's isotype; these chains are found in IgA, IgD, IgE, IgG and IgM antibodies, respectively.
不同重链的大小和组成不同;α和γ包含大约450个氨基酸,且δ包含大约500个氨基酸,而μ和ε包含大约550个氨基酸。每个重链具有恒定区(CH)和可变区(VH)两个区域。在一个物种中,恒定区在相同同种型的所有抗体中基本相同,但在不同同种型的抗体中不同。重链γ、α和δ具有由三个串联Ig结构域构成的恒定区,和用于增加柔性的铰链区;重链μ和ε具有由四个免疫球蛋白结构域构成的恒定区。所述重链的可变区在由不同B细胞产生的抗体中不同,但对于由单个B细胞或B细胞克隆产生的所有抗体来说是相同的。每个重链的可变区长大约110个氨基酸,且由单个Ig结构域构成。Different heavy chains differ in size and composition; alpha and gamma contain approximately 450 amino acids, delta approximately 500 amino acids, and mu and epsilon approximately 550 amino acids. Each heavy chain has two regions, a constant region ( CH ) and a variable region (V H ). Within a species, the constant regions are essentially the same in all antibodies of the same isotype, but differ in antibodies of different isotypes. Heavy chains gamma, alpha and delta have a constant region consisting of three tandem Ig domains, and a hinge region for increased flexibility; heavy chains mu and epsilon have a constant region consisting of four immunoglobulin domains. The variable region of the heavy chain differs in antibodies produced by different B cells, but is the same for all antibodies produced by a single B cell or B cell clone. The variable region of each heavy chain is approximately 110 amino acids long and consists of a single Ig domain.
在哺乳动物中,有两种类型的免疫球蛋白轻链,由λ和κ表示。轻链具有两个连续的结构域:一个恒定结构域(CL)和一个可变结构域(VL)。轻链的近似长度为211至217个氨基酸。每种抗体包含两条总是相同的轻链;在哺乳动物中每种抗体仅存在一种类型的轻链κ或λ。In mammals, there are two types of immunoglobulin light chains, denoted by λ and κ. Light chains have two contiguous domains: a constant domain (CL) and a variable domain (VL). The approximate length of the light chain is 211 to 217 amino acids. Each antibody contains two light chains that are always the same; only one type of light chain, kappa or lambda, is present per antibody in mammals.
尽管所有抗体的一般结构非常相似,但给定抗体的独特性质是由如上文详述的可变(V)区确定的。更具体地,可变环(每个轻链(VL)三个,重链(VH)上三个)负责结合抗原,即负责其抗原特异性。这些环称为互补决定区(CDR)。因为来自VH结构域和VL结构域的多个CDR构成了抗原结合位点,所以是重链和轻链的组合(而不是各自单独)决定了最终的抗原特异性组合。Although the general structure of all antibodies is very similar, the unique properties of a given antibody are determined by the variable (V) regions as detailed above. More specifically, the variable loops (three on each light chain (VL) and three on the heavy chain (VH)) are responsible for antigen binding, ie for its antigenic specificity. These loops are called complementarity determining regions (CDRs). Because multiple CDRs from the VH and VL domains constitute the antigen-binding site, it is the combination of heavy and light chains (rather than each alone) that determines the final antigen-specific combination.
“抗体片段”含有至少一个如上文定义的抗原结合片段,并且展现出与衍生所述抗体片段的完全抗体本质上相同的功能和特异性。用木瓜蛋白酶进行的限制性蛋白水解将Ig原型裂解成三个片段。两个相同的氨基末端片段是抗原结合片段(Fab),每个片段包含一条完整的L链和约半条的H链。第三个片段是可结晶片段(Fc),所述片段大小相似但包含两条重链的羧基末端的一半及其链间二硫键。Fc包含碳水化合物、补体结合位点和FcR结合位点。有限的胃蛋白酶消化产生单个F(ab')2片段,所述片段包含Fab段和铰链区二者,包括H-H链间二硫键。F(ab')2对于抗原结合是二价的。F(ab')2的二硫键可以被裂解以获得Fab'。此外,重链和轻链的可变区可以融合在一起以形成单链可变片段(scFv)。An "antibody fragment" contains at least one antigen-binding fragment as defined above, and exhibits substantially the same function and specificity as the complete antibody from which the antibody fragment is derived. Restriction proteolysis with papain cleaved the Ig prototype into three fragments. The two identical amino-terminal fragments are antigen-binding fragments (Fab), each containing a complete L chain and about half an H chain. The third fragment is a crystallizable fragment (Fc), which is similar in size but contains the carboxy-terminal half of the two heavy chains and their interchain disulfide bonds. Fc contains carbohydrate, complement binding site and FcR binding site. Limited pepsin digestion yields a single F(ab')2 fragment comprising both the Fab segment and the hinge region, including the H-H interchain disulfide bonds. F(ab')2 is bivalent for antigen binding. The disulfide bond of F(ab')2 can be cleaved to obtain Fab'. In addition, the variable regions of the heavy and light chains can be fused together to form a single chain variable fragment (scFv).
药学上可接受的盐是例如酸加成盐和碱性盐。酸加成盐是例如HCl或HBr盐。碱性盐是例如具有阳离子的盐,所述阳离子选自碱金属或碱土金属的阳离子,例如Na+或K+或Ca2+,或铵离子N+(R1)(R2)(R3)(R4),其中R1至R4彼此独立地表示:氢、任选地经取代的C1-C6-烷基基团、任选地经取代的C2-C6-烯基基团、任选地经取代的C6-C10-芳基基团、或任选地经取代的C6-C10-杂芳基基团。药学上可接受的盐的其他实施例描述于以下文献中:“Remington's Pharmaceutical Sciences”第17版Alfonso R.Gennaro(编),MarkPublishing Company,Easton,Pa.,U.S.A.,1985以及Encyclopedia of PharmaceuticalTechnology。Pharmaceutically acceptable salts are, for example, acid addition salts and basic salts. Acid addition salts are, for example, HCl or HBr salts. Basic salts are, for example, salts having cations selected from alkali or alkaline earth metal cations, such as Na+ or K+ or Ca2+, or ammonium ions N+(R1)(R2)(R3)(R4), wherein R1 to R4 independently of each other represent: hydrogen, optionally substituted C1-C6-alkyl group, optionally substituted C2-C6-alkenyl group, optionally substituted C6-C10-aryl group group, or an optionally substituted C6-C10-heteroaryl group. Additional examples of pharmaceutically acceptable salts are described in "Remington's Pharmaceutical Sciences" 17th Ed. Alfonso R. Gennaro (ed.), Mark Publishing Company, Easton, Pa., U.S.A., 1985 and the Encyclopedia of Pharmaceutical Technology.
药学上可接受的溶剂化物是例如水合物。Pharmaceutically acceptable solvates are, for example, hydrates.
对于本领域技术人员来说还清楚的是,在不脱离本文所公开的精神和范围的情况下,可以对注射装置进行各种更改和变动。此外,应注意,所附权利要求中使用的任何附图标记不应被解释为对本发明的范围进行限制。It will also be apparent to those skilled in the art that various modifications and variations can be made in the injection device without departing from the spirit and scope disclosed herein. Furthermore, it should be noted that any reference signs used in the appended claims shall not be construed as limiting the scope of the invention.
附图说明Description of drawings
在下文中,将参考附图详细描述驱动机构和注射装置的实施方案,其中:In the following, embodiments of the drive mechanism and the injection device will be described in detail with reference to the accompanying drawings, in which:
图1示意性地示出了注射装置的实施例,Figure 1 schematically shows an embodiment of an injection device,
图2示出了图1的注射装置的部件的分解视图,Figure 2 shows an exploded view of the components of the injection device of Figure 1,
图3是从其上拆下了限制器的注射装置的一个实施例的侧视图,Figure 3 is a side view of one embodiment of an injection device from which the restrictor has been removed,
图4是可附接到注射装置的侧壁上的限制器的侧视图,Figure 4 is a side view of a restrictor attachable to a side wall of an injection device,
图5是根据图4的限制器的仰视图,Figure 5 is a bottom view of the restrictor according to Figure 4,
图6是图3的注射装置的侧视图,其上附接了图4和图5的限制器,Figure 6 is a side view of the injection device of Figure 3 with the restrictor of Figures 4 and 5 attached thereto,
图7是注射装置的另一个实施例的侧视图,Figure 7 is a side view of another embodiment of an injection device,
图8是相对于纵向轴线稍微旋转的图7的注射装置的另外的视图,Figure 8 is a further view of the injection device of Figure 7 rotated slightly relative to the longitudinal axis,
图9是限制器的另一个实施例的独立透视图,Figure 9 is an isolated perspective view of another embodiment of the restrictor,
图10从另一个角度示出了图9的限制器,Figure 10 shows the restrictor of Figure 9 from another angle,
图11指示了图9的处于第一配置的限制器,所述限制器根据图7或图8附接到注射装置的侧壁,Figure 11 indicates the restrictor of Figure 9 in a first configuration attached to the side wall of the injection device according to Figure 7 or Figure 8,
图12示出了图9的处于第二配置的限制器,所述限制器附接到注射装置的侧壁,Figure 12 shows the restrictor of Figure 9 in a second configuration attached to the side wall of the injection device,
图13是穿过根据图7或图8的注射装置的一部分的纵向截面,Figure 13 is a longitudinal section through a part of the injection device according to Figure 7 or Figure 8,
图14是根据图13的其上附接有限制器的截面,Fig. 14 is a section according to Fig. 13 with the restrictor attached thereto,
图15是根据图14的截面的平面图,Fig. 15 is a plan view according to the section of Fig. 14,
图16示出了可附接到注射装置外壳上的壳体,Figure 16 shows a housing attachable to an injection device housing,
图17示出了图16的壳体内侧,Figure 17 shows the inside of the housing of Figure 16,
图18示出了图16和图17的其上附接有标签的壳体,以及Figure 18 shows the housing of Figures 16 and 17 with the label attached thereto, and
图19示出了图7的设置有图16的壳体的注射装置。FIG. 19 shows the injection device of FIG. 7 provided with the housing of FIG. 16 .
具体实施方式Detailed ways
如图1和图2所示的注射装置1是预填充的一次性注射装置,其包括外壳10,注射针15可附装到所述外壳上。注射针15由内针帽16和外针帽17或保护帽18保护,保护帽被配置为包围并保护注射装置1的外壳10的远侧区段。外壳10可包括并形成主外壳部分,所述主外壳部分被配置为容纳如图2所示的驱动机构8。注射装置1还可包括表示为药筒保持器14的远侧外壳部件。药筒保持器14可永久地或可释放地连接到主外壳10。药筒保持器14典型地被配置为容纳填充有液体药剂的药筒6。药筒6包括借助于位于桶25内的塞子7在近侧方向3上密封的圆柱形或管形桶25。塞子7可借助于活塞杆20在远侧方向2上相对于药筒6的桶25移位。药筒6的远端由可刺穿的密封件26密封,可刺穿的密封件被配置为隔板并且可被注射针15的在近侧定向的尖端刺穿。药筒保持器14在其远端包括螺纹插口28,以与注射针15的相应螺纹部分螺纹接合。通过将注射针15附接到药筒保持器14的远端,药筒6的密封件26被穿透,从而建立到药筒6的内部的流体输送通路。The
当注射装置1被配置为给予例如人胰岛素时,注射装置1的近端处的剂量刻度盘12设定的剂量可以所谓的国际单位(IU)来显示,其中1IU是约45.5μg纯结晶胰岛素(1/22mg)的生物学当量。When the
如图1和图2中进一步所示,外壳10包括剂量窗口13,所述剂量窗口可以是外壳10中孔隙的形式。剂量窗口13容许使用者查看数字套管80的有限部分,数字套管被配置为在转动剂量刻度盘12时移动,以提供当前设定剂量的视觉指示。当在剂量设定和/或分配或排出期间被转动时,剂量刻度盘12相对于外壳10在螺旋路径上旋转。As further shown in FIGS. 1 and 2 , the
注射装置1可以被配置为使得转动剂量旋钮12引起机械咔嗒声以向使用者提供声学反馈。数字套管80与胰岛素药筒6中的活塞以机械方式相互作用。在将针15刺入患者的皮肤部分中,并在推动触发器11或注射按钮时,将从注射装置1排出显示窗口13中显示的胰岛素剂量。当在推动触发器11后注射装置1的针15在皮肤部分中保留一定时间时,较高百分比的所述剂量实际上被注射至患者体内。胰岛素剂量的排出还可引起机械咔嗒声,但其与使用剂量刻度盘12时产生的声音不同。The
在此实施方案中,在胰岛素剂量递送期间,将剂量刻度盘12转动至其轴向移动的初始位置,也就是说没有旋转,同时将数字套管80旋转以返回其初始位置,例如显示零单位的剂量。In this embodiment, during insulin dose delivery, the
注射装置1可以用于若干次注射过程,直至药筒6排空或注射装置1中的药剂到达有效期(例如,首次使用后28天)为止。The
另外,在首次使用注射装置1之前,可能需要进行所谓的“待发注射(prime shot)”以从药筒6和针15中移除空气,例如通过选择两个单位的药剂并在保持注射装置1的针15朝上的同时压下触发器11来进行。为便于呈现,在下文中将假定,所排出的量基本上对应于注射剂量,使得例如从注射装置1排出的药剂的量等于使用者接收的剂量。In addition, prior to the first use of the
如图2中更详细地图示的排出或驱动机构8包括许多机械相互作用的部件。外壳10的凸缘状支撑件包括螺纹轴向贯通开口,其与活塞杆20的第一螺纹或远侧螺纹22螺纹接合。活塞杆20的远端包括轴承21,压力脚23在其上自由旋转,以活塞杆20的纵向轴线作为旋转轴线。压力脚23被配置为轴向地邻接抵靠药筒6的塞子7的面向近侧的推力接收面。在分配动作期间,活塞杆20相对于外壳10旋转,从而经历相对于外壳10、30并且因此相对于药筒6的桶25的、远侧定向的推进运动。结果,由于活塞杆20与外壳10的螺纹接合,药筒6的塞子7在远侧方向2上移位了明确定义的距离。The ejection or drive
活塞杆20在其近端还设有第二螺纹24。远侧螺纹22和近侧螺纹24旋向相反。The
还设置了驱动套管30,其具有中空内部以接收活塞杆20。驱动套管30包括与活塞杆20的近侧螺纹24螺纹接合的内螺纹。此外,驱动套管30在其远端包括外螺纹区段31。螺纹区段31轴向限制在远侧凸缘部32与另一凸缘部33之间,所述另一凸缘部位于距远侧凸缘部32的预定轴向距离处。在这两个凸缘部分32、33之间设置有半圆形螺母形式的最后剂量限制器35,所述半圆形螺母具有与驱动套管30的螺纹区段31配合的内螺纹。A
最后剂量限制器35还包括在其外圆周处的径向凹部或径向突起,以与外壳10的侧壁内侧处的互补形状的凹部或突起接合。以这种方式,最后剂量限制器35用花键接合到外壳10。连续剂量设定程序期间驱动套管30在剂量递增方向4或顺时针方向的旋转导致最后剂量限制器35相对于驱动套管30的累积轴向移位。还设置了环形弹簧40,其与凸缘部分33的面向近侧的表面轴向邻接。此外,设置了管形接合器(clutch)60。接合器60在第一端设置有一系列周向定向的锯齿。朝向接合器60的第二相对端定位径向向内定向的凸缘。The
此外,设置了也表示为数字套管80的剂量刻度盘套管。数字套管80设置在弹簧40和接合器60的外侧,并且位于外壳10的径向内侧。绕数字套管80的外表面设置有螺旋凹槽81。外壳10设置有剂量窗口13,通过剂量窗口可以看到数字80的外表面的一部分。外壳10还在插入件62的内侧壁部分处设置有螺旋肋,所述螺旋肋将座接于数字套管80的螺旋凹槽81中。管形插入件62被插入外壳10的近端。它在转向上且轴向地固定到外壳10。在外壳10上设置有第一和第二止挡件,以限制剂量设定过程,在此期间,数字套管80相对于外壳10以螺旋运动旋转。In addition, a dose dial sleeve, also denoted
剂量刻度盘握把形式的剂量刻度盘12绕数字套管80的近端的外表面布置。剂量刻度盘12的外径典型地对应于外壳10的外径并与之匹配。剂量刻度盘12稳固到数字80以防止其之间的相对移动。剂量刻度盘12设置有中央开口。A
触发器11(也表示为剂量按钮)基本上为T形。它设置在注射装置10的近端。触发器11的杆64延伸穿过剂量刻度盘12中的开口、穿过驱动套管30的延伸部的内径并进入活塞杆20的近端处的接收凹部。杆64被保持用于驱动套管30中的有限轴向移动并且抵抗相对于其旋转。触发器11的头部总体上是圆形的。触发器侧壁或裙部从头部的圆周延伸,并且还适配为座接于剂量刻度盘12的近侧可及的环形凹部中。The trigger 11 (also denoted as a dose button) is substantially T-shaped. It is provided at the proximal end of the
使用者旋转剂量刻度盘12以拨选剂量。在弹簧40也用作咔嗒发声器并且接合器60被接合的情况下,驱动套管30、弹簧或咔哒发声器40、接合器60、和数字套管80与剂量刻度盘12一起旋转。通过弹簧40并通过接合器60提供对所拨选剂量的可听见的且有触觉的反馈。通过锯齿在弹簧40与接合器60之间传递扭矩。数字套管80上的螺旋凹槽81和驱动套管30中的螺旋凹槽具有相同的导程。这允许数字套管80从外壳10和驱动套管30延伸,以相同的速率爬上活塞杆20。在行程极限处,数字套管80上的径向止挡件与设置在外壳10上的第一止挡件或第二止挡件接合,以防止进一步移动。由于活塞杆20上整体螺纹和从动螺纹的相反方向,防止了活塞杆20的旋转。The user rotates the
通过驱动套管30的旋转,键合到外壳10的最后剂量限制器35沿螺纹区段31推进。当达到最终剂量分配位置时,在最后剂量限制器35的表面上形成的径向止挡件邻接驱动套管30的凸缘部分33上的径向止挡件,阻止最后剂量限制器35和驱动套管30二者进一步旋转。By driving the rotation of the
如果使用者无意中拨选超过希望的剂量,则被配置为笔式注射器的注射装置1允许在未从药筒6分配药剂的情况下拨小剂量。简单地反向旋转剂量刻度盘12来完成这一点。这导致系统反向起作用。弹簧或点击器40的柔性臂然后用作防止弹簧40旋转的棘轮。通过接合器60传递的扭矩使锯齿相互压制以产生对应于所拨选剂量减少的咔嗒声。典型地,锯齿被布置成使得每个锯齿的周向范围对应于单位剂量。The
当已经拨选了希望的剂量时,使用者可以通过压下触发器11简单地分配设定的剂量。这使接合器60相对于数字套管80轴向地移位,使其犬齿脱离。然而,接合器60保持与驱动套管30的旋转键合。数字套管80和剂量刻度盘12现在可根据螺旋凹槽81自由地旋转。When the desired dose has been dialed, the user can simply dispense the set dose by depressing the
轴向移动使弹簧40的柔性臂变形,以确保在分配期间锯齿不会被翻修。这防止驱动套管30相对于外壳10旋转,尽管它仍然相对于外壳轴向自由移动。所述变形随后用于沿驱动套管30推回弹簧40和接合器60,以在从触发器11移除远侧定向的分配压力时恢复接合器60与数字套管80之间的连接。Axial movement deforms the flexible arms of
驱动套管30的纵向轴向移动使得活塞杆20通过外壳10的支撑件的贯通开口旋转,从而在药筒6中推进塞子7。一旦已经分配了所拨选的剂量,就通过从剂量刻度盘12延伸的至少一个止挡件与外壳10的至少一个对应的止挡件的接触来防止数字套管80进一步旋转。可通过数字套管80的轴向延伸边缘或止挡件中的一个与外壳10的至少一个或若干个对应止挡件的邻接来确定零剂量位置。The longitudinal axial movement of the
如上所述的排出机构或驱动机构8仅是通常可在一次性笔式注射器中实现的多个不同配置的驱动机构之一的示例。如上所述的驱动机构在例如WO 2004/078239 A1、WO2004/078240 A1或WO 2004/078241 A1中更详细地解释,其全部内容通过引用并入本文。The expelling mechanism or drive
如图2所图示的剂量设定机构9至少包括剂量刻度盘12和数字套管80。当在设定剂量期间以及为了设定剂量而旋转剂量刻度盘12时,数字套管80开始相对于外壳沿螺旋路径旋转,所述螺旋路径由数字套管的外螺纹或螺旋凹槽81与外壳的内表面上对应地成形的螺纹区段的螺纹接合限定。The
在剂量设定期间并且当驱动机构8或剂量设定机构9处于剂量设定模式时,驱动套管30与剂量刻度盘12并与数字套管80一致地旋转。驱动套管30以螺纹的方式与活塞杆20接合,所述活塞杆在剂量设定期间相对于外壳10静止。因此,驱动套管30在剂量设定期间经受螺旋或螺旋形运动。当剂量刻度盘在剂量递增方向4(例如,顺时针方向)旋转时,驱动套管30开始在近侧方向上行进。为了调节或校正剂量的规格,剂量刻度盘12可在相反的方向上、因此在剂量递减方向5上(例如,逆时针)旋转。During dose setting and when either the
驱动套管30和数字套管80中的至少一个用作剂量跟踪器50,所述剂量跟踪器包括跟踪止挡特征件51。在例如图13至图15中所图示的实施例中,凸缘32或33中的一个提供并与剂量跟踪器50的跟踪止挡特征件51重合。还设置了作为单独件的限制器70。At least one of the
如图3、图7或图8所图示,外壳10包括管形或圆柱形侧壁43、143。侧壁43、143包括第一紧固特征件41、141和第二紧固特征件42、142。如例如图3所指示,第一紧固特征件41与第二紧固特征件42轴向地分离。在所示的实施例中,第一紧固特征件41和第二紧固特征件42各自包括延伸穿过外壳10的侧壁43的贯通开口44。As illustrated in FIGS. 3 , 7 or 8 , the
限制器70包括平坦形状的基底部分72和从基底部分72突起的限制器止挡特征件71。对限制器止挡特征件71进行配置并确定尺寸以与第一紧固特征件41并与第二紧固特征件42配合。对限制器止挡特征件71进行配置并确定尺寸以一次延伸穿过第一和第二紧固特征件41、42之一的贯通开口44。限制器止挡特征件41可分别插入并穿过第一紧固特征件41的或第二紧固特征件42的贯通开口44。The
如例如图15中所示,限制器170以这样的配置附接到侧壁143的外表面145:限制器止挡特征件171延伸穿过第二紧固特征件142,限制器止挡特征件171或至少其自由端从侧壁的内表面146径向向内突起。由于限制器止挡特征件71、171和整个限制器70、170静止地固定到外壳10的侧壁43、143,因此在设定剂量期间,它为剂量跟踪器50的跟踪止挡特征件51提供轴向止挡。在剂量设定期间,剂量跟踪器50及其跟踪止挡特征件51可在近侧方向上行进的轴向路径受到限制器70、170及其限制器止挡特征件71、171的位置的限制。As shown, for example, in FIG. 15, the
依据限制器70、170和剂量跟踪器50的相互接合的止挡特征件的具体几何形状,还可以想到,限制器止挡特征件71、171阻止剂量跟踪器50的旋转超过预定义的最大旋转位置。以这种方式,限制器止挡特征件71、171可以被布置并附接到外壳10的侧壁43、143的预定义部分,所述预定义部分对应于可旋转地支撑在外壳10内侧的剂量跟踪器50的预定义角位置。例如,限制器止挡特征件71、171可形成针对跟踪止挡特征件51的周向或径向止挡,所述跟踪止挡特征件设置在由数字套管80形成的剂量跟踪器50的外表面上,如图2所示。Depending on the specific geometry of the
即使所有当前图示的实施例都包括第一紧固特征件41、141和第二紧固特征件42、142,也可以想到一些实施例,其中在外壳10的侧壁43、143中仅设置单个即第一紧固特征件41、141。在此,第一紧固特征件41、141还包括贯通开口44、144,以通过其接收对应地成形的限制器止挡特征件71、171。除了限制器止挡特征件71、171和跟踪止挡特征件51之外,驱动机构8或剂量设定机构9还可以包括附加的最大剂量限制器功能。如图2所图示,可以在数字套管80的外表面上设置跟踪止挡特征件51,所述跟踪止挡特征件被配置为与插入件62的止挡特征件63邻接和接合。插入件62被静止地附接和固定到外壳10。由于已设定了例如约80I.U.的最大剂量,跟踪止挡特征件51与插入件62的止挡特征件63邻接,从而阻止数字套管80的进一步旋转和/或平移。在该实施方案中,数字套管80同样用作剂量跟踪器50。Even though all of the presently illustrated embodiments include first and second fastening features 41 , 141 , 42 , 142 , embodiments are conceivable in which only the
除了数字套管80和插入件62的相互作用所提供的这种最大剂量限制器功能之外,外壳100的侧壁43、143仅包括用于限制器70、170的第一紧固特征件41、141可能就足够了。通过将限制器70、172附接到侧壁43、143,实现了附加的最大剂量止挡,并且推翻了最初提供的最大剂量限制器功能。通过将限制器70、172附接到侧壁43、143,从注射装置1外侧修改驱动机构8的或剂量设定机构9的最大剂量限制器功能。In addition to this maximum dose limiter function provided by the interaction of the
可以由剂量跟踪器50和限制器70的相互作用来代替由数字套管80和插入件62的相互作用所提供的剂量限制器功能。对于此类实施方案,外壳10和侧壁43、143包括至少第一和第二紧固特征件41、42、141、142是特别有益的。然后,在第一配置中,限制器70被接合或附接到第一紧固特征件41,限制器止挡特征件71从外壳10的侧壁43的第一明确定义的轴向部分突起。在第二配置中,限制器70被接合或附接到第二紧固特征件41。然后,限制器止挡特征件71从外壳10的侧壁43的第二明确定义的轴向部分突起。在本实施例中并且如图3所示,第二紧固特征件42位于第一紧固特征件41的近侧。在第二配置中并且当限制器止挡特征件71穿过第二紧固特征件42的贯通开口44突起时,与第一配置相比最大剂量规格增加,在第一配置中,限制器止挡特征件71穿过第一紧固特征件41的贯通开口44突起。The dose limiter function provided by the interaction of the
当附接到外壳43的外表面45时,限制器70的平坦形状的基底部分72与外表面45基本齐平,同时限制器止挡特征件71径向向内突起并进入外壳10的内部。在如图4和图5所示的实施例中,限制器止挡特征件71包括从平坦形状的基底部分72垂直延伸的销。限制器止挡特征件71在其自由端包括倾斜区段73,以便于将限制器止挡特征件71插入和组装到贯通开口44中。When attached to the
如图3至图5所图示,限制器70的基底部分72包括轴向延伸部,所述轴向延伸部至少等于或大于第一紧固特征件41与第二紧固特征件42之间的距离。此外,限制器止挡特征件71布置在基底部分72的轴向端附近。以这种方式并且当限制器止挡特征件71附接到第一紧固特征件41时,基底部分72的背向限制器止挡特征件71的部分覆盖第二紧固特征件42。反之亦然,当限制器止挡特征件71附接到第二紧固特征件42时,基底部分72的远端部分覆盖第一紧固特征件41。以这种方式,基底部分72包括允许在第一和第二配置中紧固和附接限制器70的对称形状,其中通过将限制器70旋转180°,第一配置可转变成第二配置。As illustrated in FIGS. 3-5 , the
在如图7至图15所图示的另一个实施例中,第一和第二紧固特征件141、142在外壳10的侧壁143的外表面145上也沿轴向方向(z)彼此分离。紧固特征件141、142中的每一个包括延伸穿过侧壁143的贯通开口144。甚至设置了位于第二紧固特征件142近侧的第三紧固特征件。这些紧固特征件等距地布置在外壳10上。以与上述相同的方式,紧固特征件141、142的贯通开口144被配置为接收如图9和图10所图示的限制器170的限制器止挡特征件171。In another embodiment as illustrated in FIGS. 7-15 , the first and second fastening features 141 , 142 are also in the axial direction (z) of each other on the
如在图11中更详细地示出,第一紧固特征件141包括凹入部分147。以类似的方式,第二紧固特征件也包括凹入部分147。凹入部分周向地分别与第一和第二紧固特征件141、142的贯通开口144相邻。如图11和图12进一步所图示,第一紧固特征件141和第二紧固特征件142的凹入部分147在侧壁143的外表面145上合并并形成共同的细长凹部148。细长凹部148包括沿细长外壳10的纵向或轴向方向(z)延伸的纵向凹入部分。As shown in more detail in FIG. 11 , the
细长凹部148包括底部149,所述底部位于距侧壁143的外表面145的径向距离处。该径向距离至少等于或大于限制器170的基底部分172的厚度。以这种方式,细长凹部148被配置为整体容纳限制器170的基底部分172。当附接到侧壁143时,限制器170及其基底部分172不从侧壁143的外表面145径向突起。The
如图9至图12所图示,限制器170是L形的。限制器170包括第一支腿173和第二支腿174。第一支腿173和第二支腿174以大约90°的角度延伸。当附接到外壳10时,第一支腿173沿外壳10的圆周(w)延伸,并且第二支腿174沿外壳10的轴向伸长(z)延伸。第一支腿174以及因此基底部分172被组装在细长凹部148内侧,而第二支腿143朝向第一或第二紧固特征件141、142之一的贯通开口144延伸并进入其中。在图11中,限制器止挡特征件171以及因此第一支腿173延伸到第一紧固特征件141中。在图12中,限制器止挡特征件171以及因此第一支腿173延伸到第二紧固特征件142中并与之接合。As illustrated in FIGS. 9-12 , the
当组装到外壳10时,限制器170的基底部分172与细长凹部148的底部149邻接。以这种方式,限制器170相对于外壳10的径向位置被明确定义。The
如图11和图12所示,还设置了与细长凹部148周向地相邻的凹入区段150。凹入区段150与第一和第二紧固特征件141、142的贯通开口144周向地相对。凹入区段150与细长凹部148合并。当附接到第一和第二紧固特征件141、142之一时,它们提供对限制器170的基底部分172的侧边缘的访问。以这种方式,并且例如借助于诸如螺丝刀之类的有尖工具,基底部分172以及因此整个限制器170可被径向向外抬升,以将限制器170从外壳10上拆下。限制器170然后可以附接到另一紧固特征件,以便修改注射装置1的最大剂量规格。As shown in FIGS. 11 and 12 , a recessed
限制器70、170可以摩擦地或粘附地附接到外壳10的侧壁43、143。可以借助于单独的部件,例如借助于紧固件将限制器70、170紧固到侧壁43、143。此类紧固件可以包括胶粘标签或盖90,所述盖覆盖限制器70、170并将其固定到侧壁43、143。在图11或图12的实施例中,限制器170可以压配在细长凹部148之一中或在第一或第二紧固特征件141、142之一的贯通开口144之一中。也可以通过粘合剂的方式或通过焊接的方式将它永久地附接和固定到外壳10上。限制器70、170和外壳10可以包括适合于焊接(诸如,激光焊接或超声焊接)的可模制塑料材料。The
为了将限制器70、172临时地或永久地固定到外壳10上,可以设置标签100,所述标签可粘附地附接到侧壁43、143的外表面45、145。标签100可以设置有印刷在其上的可读信息。所述信息可以包括关于药剂和/或关于注射装置1的处理或使用的说明或一般信息。To temporarily or permanently secure the
标签100可以完全覆盖限制器70、170以及第一和第二紧固特征件41、42、141、142。The
此外并且如图16至图19所图示,可以设置单独的盖90。盖90可以包括塑料材料。盖90可以包括沿纵向方向切割的略微管形的形状,以便包括半圆形的截面并且包括半壳的形状。典型地,盖90的圆周大于外壳10的外圆周的一半。以这种方式,盖90可以被夹持在外壳10的外表面45、145上。盖90是可弹性变形的,以便至少暂时地扩大其自由端之间的距离,并允许沿外壳10的径向方向对盖90进行夹持附接。在一个实施例中,限制器70、170和盖90可以一体地形成。在此,限制器70可以从盖90的内表面突起。限制器70、170和盖90可以被成形和配置为使得当盖90被附接和固定到外壳10时,限制器70、170位于贯通开口44、144之一中。Additionally and as illustrated in Figures 16-19, a
为了相对于轴向和/或周向方向稳固和固定盖90,可以在盖90的内表面和外壳10的外表面145上设置相互对应的紧固元件91、151。在如图7、图8、图17和图19所示的实施例中,盖90的紧固元件91包括径向向内突起的销,并且外壳10的紧固元件151包括对应地成形的凹部。当正确地组装时,紧固元件91被卡扣到紧固元件151中。以这种方式,盖90相对于所有三个尺寸都固定到外壳10上。In order to stabilize and fix the
另外或替代地,标签100可用于将盖90固定到外壳10。如图18所指示,标签100可以以这样的方式粘附地附接到盖90:标签100的一部分也覆盖注射装置1的侧壁143的外表面145的未被盖90覆盖的一部分。由于标签100粘附地附接到外壳10的外表面145和盖90的外表面二者,因此可以借助于标签100将盖90固定到外壳10上。Additionally or alternatively, the
上面结合第一和第二紧固特征件41、141、42、142描述的任何特征对于在外壳10的侧壁43、143上仅具有一个紧固特征件41、141的注射装置的实施例或实施方案同样有效。仅作为实施例,紧固特征件41、141可包括凹入部分147,以接收限制器70、170的基底部分72、172。此外,唯一的第一紧固特征件41、141可以被盖90和/或标签100覆盖。Any of the features described above in connection with the first and second fastening features 41 , 141 , 42 , 142 for embodiments of the injection device having only one
附图标记列表List of reference signs
1 注射装置1 Injection device
2 远侧方向2 Distal direction
3 近侧方向3 Proximal orientation
4 剂量递增方向4 Direction of dose escalation
5 剂量递减方向5 Direction of dose decrement
6 药筒6 cartridges
7 塞子7 stoppers
8 驱动机构8 drive mechanism
9 剂量设定机构9 Dose setting mechanism
10 外壳10 shell
11 触发器11 Triggers
12 剂量刻度盘12 dose dial
13 剂量窗口13 Dosage window
14 药筒保持器14 Cartridge holder
15 注射针15 injection needles
16 内针帽16 inner needle cap
17 外针帽17 Outer needle cap
18 保护帽18 Protective caps
20 活塞杆20 Piston Rod
21 轴承21 Bearings
22 第一螺纹22 First thread
23 压脚23 Presser foot
24 第二螺纹24 Second thread
25 桶25 barrels
26 密封件26 Seals
28 螺纹插口28 threaded socket
30 驱动套管30 Drive bushing
31 螺纹区段31 Thread section
32 凸缘32 flange
33 凸缘33 Flange
35 最后剂量限制器35 Last dose limiter
36 肩部36 Shoulder
40 弹簧40 spring
41 紧固特征件41 Fastening Features
42 紧固特征件42 Fastening Features
43 侧壁43 Side walls
44 贯通开口44 Through opening
45 外表面45 outer surface
50 剂量跟踪器50 Dose Tracker
51 跟踪止挡特征件51 Tracking Stop Feature
60 接合器60 adapter
62 插入件62 Inserts
63 止挡特征件63 Stop features
64 杆64 bars
70 限制器70 Limiter
71 限制器止挡特征件71 Limiter Stop Feature
72 基底部分72 Base part
73 倾斜区段73 Inclined section
80 数字套管80 digital casing
81 凹槽81 groove
90 罩90 hood
91 紧固元件91 Fastening elements
100 标签100 tags
141 紧固特征件141 Fastening Features
142 紧固特征件142 Fastening Features
143 侧壁143 Sidewall
144 贯通开口144 Through opening
145 外表面145 outer surface
146 内表面146 inner surface
147 凹入部分147 Recessed part
148 凹部148 Recess
149 按钮149 buttons
150 凹部150 Recess
151 紧固元件151 Fastening elements
170 限制器170 Limiter
171 限制器止挡特征件171 Limiter Stop Feature
172 基底部分172 Base part
173 支腿173 Outriggers
174 支腿174 Outriggers
Claims (17)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP17200311.3 | 2017-11-07 | ||
| EP17200311 | 2017-11-07 | ||
| PCT/EP2018/080076 WO2019091877A1 (en) | 2017-11-07 | 2018-11-05 | Injection device with a dose limiter |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN111295213A true CN111295213A (en) | 2020-06-16 |
Family
ID=60269684
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201880071726.0A Pending CN111295213A (en) | 2017-11-07 | 2018-11-05 | Injection device with dose limiter |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP3706835A1 (en) |
| JP (1) | JP2021501635A (en) |
| CN (1) | CN111295213A (en) |
| WO (1) | WO2019091877A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114681718A (en) * | 2022-03-30 | 2022-07-01 | 宁波睿爱产品设计有限公司 | Multiple quantitative injector |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3990059A1 (en) * | 2019-06-26 | 2022-05-04 | Sanofi | Injection device with a filling level indicator |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1074273A1 (en) * | 1999-08-06 | 2001-02-07 | Becton Dickinson and Company | Improved medication delivery pen |
| CN1374876A (en) * | 1999-09-16 | 2002-10-16 | 诺沃挪第克公司 | Dose setting limiter |
| CN102413854A (en) * | 2009-02-26 | 2012-04-11 | Shl集团有限责任公司 | Dose setting mechanism |
| CN106794319A (en) * | 2014-10-09 | 2017-05-31 | 赛诺菲 | Delivery device |
| US20170259006A1 (en) * | 2014-11-24 | 2017-09-14 | Sanoi | Mechanism for setting a maximum dose of a medicament for a variable dose drug delivery device |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB0304823D0 (en) | 2003-03-03 | 2003-04-09 | Dca Internat Ltd | Improvements in and relating to a pen-type injector |
| GB0304822D0 (en) | 2003-03-03 | 2003-04-09 | Dca Internat Ltd | Improvements in and relating to a pen-type injector |
-
2018
- 2018-11-05 CN CN201880071726.0A patent/CN111295213A/en active Pending
- 2018-11-05 WO PCT/EP2018/080076 patent/WO2019091877A1/en not_active Ceased
- 2018-11-05 EP EP18793681.0A patent/EP3706835A1/en not_active Withdrawn
- 2018-11-05 JP JP2020524221A patent/JP2021501635A/en active Pending
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1074273A1 (en) * | 1999-08-06 | 2001-02-07 | Becton Dickinson and Company | Improved medication delivery pen |
| CN1374876A (en) * | 1999-09-16 | 2002-10-16 | 诺沃挪第克公司 | Dose setting limiter |
| CN102413854A (en) * | 2009-02-26 | 2012-04-11 | Shl集团有限责任公司 | Dose setting mechanism |
| CN106794319A (en) * | 2014-10-09 | 2017-05-31 | 赛诺菲 | Delivery device |
| US20170259006A1 (en) * | 2014-11-24 | 2017-09-14 | Sanoi | Mechanism for setting a maximum dose of a medicament for a variable dose drug delivery device |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114681718A (en) * | 2022-03-30 | 2022-07-01 | 宁波睿爱产品设计有限公司 | Multiple quantitative injector |
| CN114681718B (en) * | 2022-03-30 | 2024-03-19 | 宁波睿爱产品设计有限公司 | Multiple quantitative injector |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2019091877A1 (en) | 2019-05-16 |
| JP2021501635A (en) | 2021-01-21 |
| EP3706835A1 (en) | 2020-09-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101828232B1 (en) | Resettable drug delivery device | |
| US10682464B2 (en) | Drive mechanism of an injection device | |
| CN103648558B (en) | Drive component for delivery device and corresponding delivery device | |
| US11759575B2 (en) | Injection device | |
| US10076610B2 (en) | Drive mechanism for a drug delivery device | |
| US10773027B2 (en) | Injection Device | |
| CN111886040B (en) | Injection device | |
| CN106794306A (en) | Spring assembly and drug delivery device with the spring assembly | |
| CN105102019B (en) | Drive Mechanisms for Drug Delivery Devices | |
| US10576210B2 (en) | Drive mechanism for an injection device | |
| CN106470720A (en) | Display assembly for injection device | |
| TW201618821A (en) | Drive sleeve, drug delivery device and method for assembling a drug delivery device | |
| TW201625324A (en) | Housing and drug delivery device herewith | |
| US9861758B2 (en) | Drive mechanism of a drug delivery device | |
| CN111315431A (en) | Injection device | |
| CN106999658A (en) | Medicament maximum dose set mechanism for variable dose delivery device | |
| US10639428B2 (en) | Insert and drug delivery device herewith | |
| US10420893B2 (en) | Drug delivery device | |
| CN111295213A (en) | Injection device with dose limiter | |
| CN111902178A (en) | Injection device | |
| CN111315430A (en) | Injection device with preselector | |
| CN113164688B (en) | Component for drug delivery device, drug delivery device and method for producing drug delivery device | |
| CN111902171B (en) | Injection device | |
| CN113993565B (en) | Injection device with fill level indicator | |
| US20210187201A1 (en) | Injection device with a preselector |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| WD01 | Invention patent application deemed withdrawn after publication | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20200616 |