CN111286233A - A kind of gel preparation method of fluorescent pigment - Google Patents
A kind of gel preparation method of fluorescent pigment Download PDFInfo
- Publication number
- CN111286233A CN111286233A CN202010048937.1A CN202010048937A CN111286233A CN 111286233 A CN111286233 A CN 111286233A CN 202010048937 A CN202010048937 A CN 202010048937A CN 111286233 A CN111286233 A CN 111286233A
- Authority
- CN
- China
- Prior art keywords
- gel
- fluorescent pigment
- fluorescent
- sodium alginate
- preparing
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D17/00—Pigment pastes, e.g. for mixing in paints
- C09D17/003—Pigment pastes, e.g. for mixing in paints containing an organic pigment
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0001—Post-treatment of organic pigments or dyes
- C09B67/0004—Coated particulate pigments or dyes
- C09B67/0008—Coated particulate pigments or dyes with organic coatings
- C09B67/0013—Coated particulate pigments or dyes with organic coatings with polymeric coatings
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Life Sciences & Earth Sciences (AREA)
- Wood Science & Technology (AREA)
- Pigments, Carbon Blacks, Or Wood Stains (AREA)
Abstract
本发明涉及荧光颜料制备技术领域,公开了一种荧光颜料的凝胶制备方法,包括如下步骤:(1)将无机硅酸盐与荧光染料混合研磨得到混合粉体;(2)将海藻酸钠加入水中,搅拌至完全溶解后加入所述混合粉体,搅拌均匀得到混合浆料;(3)向所述混合浆料中加入无水氯化钙,搅拌均匀得到凝胶中间体;(4)向所述凝胶中间体中加入碳酸钠,搅拌均匀后沉淀,将所得产物洗涤至没有浮色后干燥得到所述荧光颜料。本发明通过海藻酸钠凝胶吸附荧光染料分子,并且将凝胶层转化为无机盐保护层,使得荧光颜料不易渗色,并且具有良好的耐酸碱性,更加稳定。
The invention relates to the technical field of preparation of fluorescent pigments, and discloses a method for preparing a gel of fluorescent pigments, comprising the following steps: (1) mixing and grinding inorganic silicates and fluorescent dyes to obtain mixed powder; (2) mixing sodium alginate Add water, stir until completely dissolved, then add the mixed powder, stir evenly to obtain a mixed slurry; (3) add anhydrous calcium chloride to the mixed slurry, stir evenly to obtain a gel intermediate; (4) Sodium carbonate is added to the gel intermediate, stirred uniformly and then precipitated, the obtained product is washed until there is no floating color, and then dried to obtain the fluorescent pigment. The invention adsorbs fluorescent dye molecules through sodium alginate gel, and converts the gel layer into an inorganic salt protection layer, so that the fluorescent pigment is not easy to bleed, has good acid and alkali resistance, and is more stable.
Description
技术领域technical field
本发明涉及荧光颜料制备技术领域,尤其是涉及一种荧光颜料的凝胶制备方法。The invention relates to the technical field of preparation of fluorescent pigments, in particular to a method for preparing gels of fluorescent pigments.
背景技术Background technique
荧光颜料吸收可见光和紫外线后,能把原来人眼感觉不到的紫外光转变为一定颜色的可见光,与常规反射的光相叠加,其总的反射光强度比一般颜料高,形成非常鲜艳的色彩。而这些荧光颜料本身往往在耐光、耐溶剂等性质方面具有先天不足因此一般将它们通过化学或物理的方法熔合到高分子材料的构架中,再进一步加工成颜料。After the fluorescent pigment absorbs visible light and ultraviolet light, it can convert the original ultraviolet light that the human eye cannot perceive into visible light of a certain color, which is superimposed with the conventional reflected light, and its total reflected light intensity is higher than that of ordinary pigments, forming a very bright color. . These fluorescent pigments themselves often have inherent deficiencies in properties such as light resistance and solvent resistance, so they are generally fused into the framework of polymer materials by chemical or physical methods, and then further processed into pigments.
目前国内环保型荧光液体颜料一般采用乳液聚合法生产,例如,在中国专利文献上公开的“一种水性日光荧光颜料色浆的制备方法”,其公告号CN103555069A,包括分散体系的制备、预乳液制备、反应、加入氧化还原引发体系反应,得到水性日光荧光颜料色浆。At present, domestic environmentally friendly fluorescent liquid pigments are generally produced by emulsion polymerization. For example, "a preparation method of water-based sunlight fluorescent pigment paste" disclosed in Chinese patent documents, its bulletin number CN103555069A, including the preparation of dispersion system, pre-emulsion Preparation, reaction and addition of redox to initiate system reaction to obtain water-based sunlight fluorescent pigment paste.
但乳液聚合法制备的荧光颜料在印染和涂料应用过程中聚合物表面有着强烈的亲水性,本身内部树脂和染料靠有限的范德华力相互吸引,聚合物中的染料很容易溶解到水中或者其他介质中,从而影响产品的性能,如皂洗、耐温、渗色等问题,还会产生大量废水,引起水污染。However, the fluorescent pigment prepared by emulsion polymerization has strong hydrophilicity on the polymer surface during printing and dyeing and coating application. The internal resin and dye are attracted to each other by limited van der Waals force, and the dye in the polymer is easily dissolved in water or other In the medium, which affects the performance of the product, such as soaping, temperature resistance, color bleeding, etc., it will also generate a large amount of waste water, causing water pollution.
发明内容SUMMARY OF THE INVENTION
本发明是为了克服现有技术中通过乳液聚合法制备的荧光颜料在印染和涂料应用过程中聚合物表面有着强烈的亲水性,聚合物中的染料很容易溶解到水中或者其他介质中,从而影响产品的性能,容易产生渗色等问题,提供一种荧光颜料的凝胶制备方法,通过对染料分子具有较强吸附力的海藻酸钠凝胶吸附荧光染料分子,并将凝胶层转化为无机盐保护层,对染料分子进一步包覆和固定,提高保护层的耐酸碱性,制得的荧光颜料不易渗色,耐酸碱性强,使用性能良好。The invention is to overcome the fact that the fluorescent pigment prepared by the emulsion polymerization method in the prior art has strong hydrophilicity on the surface of the polymer in the process of printing, dyeing and coating application, and the dye in the polymer is easily dissolved in water or other media, thereby The performance of the product is affected, and problems such as color bleeding are likely to occur. A method for preparing a fluorescent pigment gel is provided. The fluorescent dye molecules are adsorbed by sodium alginate gel with strong adsorption force on the dye molecules, and the gel layer is converted into The inorganic salt protective layer further coats and fixes the dye molecules, improves the acid and alkali resistance of the protective layer, and the obtained fluorescent pigment is not easy to bleed, has strong acid and alkali resistance, and has good performance.
为了实现上述目的,本发明采用以下技术方案:In order to achieve the above object, the present invention adopts the following technical solutions:
一种荧光颜料的凝胶制备方法,其特征是,包括如下步骤:A kind of gel preparation method of fluorescent pigment is characterized in that, comprises the steps:
(1)将无机硅酸盐与荧光染料混合研磨得到混合粉体;(1) Mixing and grinding inorganic silicate and fluorescent dye to obtain mixed powder;
(2)将海藻酸钠加入水中,搅拌至完全溶解后加入所述混合粉体,搅拌均匀得到混合浆料;(2) adding sodium alginate to the water, stirring until completely dissolved, adding the mixed powder, and stirring to obtain a mixed slurry;
(3)向所述混合浆料中加入无水氯化钙,搅拌均匀得到凝胶中间体;(3) adding anhydrous calcium chloride to the mixed slurry, stirring to obtain a gel intermediate;
(4)向所述凝胶中间体中加入碳酸钠,搅拌均匀后沉淀,将所得产物洗涤至没有浮色后干燥得到所述荧光颜料。(4) adding sodium carbonate to the gel intermediate, stirring evenly and then precipitating, washing the obtained product until there is no floating color, and drying to obtain the fluorescent pigment.
作为优选,步骤(1)中所述的无机硅酸盐为蒙皂石、伊利石、长石中的一种或几种。Preferably, the inorganic silicate described in step (1) is one or more of smectite, illite and feldspar.
作为优选,步骤(1)中所述的荧光染料为商业罗丹明或溶剂型染料。Preferably, the fluorescent dye described in step (1) is commercial rhodamine or solvent-based dye.
作为优选,步骤(1)中荧光染料的添加量为无机硅酸盐的0.1~5wt%。Preferably, the amount of fluorescent dye added in step (1) is 0.1-5 wt % of the inorganic silicate.
作为优选,步骤(2)中海藻酸钠和混合粉体的质量比为1:(2~6.5)。Preferably, in step (2), the mass ratio of sodium alginate to mixed powder is 1:(2-6.5).
作为优选,步骤(2)中海藻酸钠的溶解温度40~80℃,搅拌时间1~5h。Preferably, in step (2), the dissolving temperature of sodium alginate is 40-80° C., and the stirring time is 1-5 h.
作为优选,步骤(3)中加入的无水氯化钙与海藻酸钠的质量比为1:(0.5~2.5)。Preferably, the mass ratio of calcium chloride anhydrous and sodium alginate added in step (3) is 1:(0.5~2.5).
作为优选,步骤(4)中加入的碳酸钠与无水氯化钙质量比为1:(1.1~1.3)。Preferably, the mass ratio of sodium carbonate added in step (4) to anhydrous calcium chloride is 1: (1.1-1.3).
本发明通过环保的海藻酸钠凝胶吸附荧光染料分子,具有更强的吸附力,将染料分子固定在凝胶网络中;然后将凝胶层转化为无机盐保护层,一方面进一步包覆染料分子,另一方面无机盐保护层可以使得荧光颜料具有良好的耐酸碱性,更加稳定。The invention adsorbs fluorescent dye molecules through environmentally friendly sodium alginate gel, has stronger adsorption force, and fixes the dye molecules in the gel network; then the gel layer is converted into an inorganic salt protection layer, and on the one hand, the dye is further coated Molecules, on the other hand, the inorganic salt protective layer can make the fluorescent pigment have good acid and alkali resistance and be more stable.
海藻酸钠是由(1,4)-β-D-甘露糖醛酸(M)和(1,4)-α-L-古洛糖醛酸(G)两种单体通过糖苷键组成的无支链嵌段共聚物,这两种糖醛酸的链式结构非常相似,仅区别于其上羟基位置的不同,但它们成环后进一步聚合成链的空间结构差别非常大,因而决定了它们理化性质的差异。两种单体的连接方式不同时,可以形成三种不同的片段:聚甘露糖醛酸嵌段(MM)、聚古洛糖醛酸嵌段(GG)和甘露糖醛酸-古洛糖醛酸嵌段(MG)。当海藻酸钠与含二价阳离子Ca2+等的溶液接触时,二价阳离子会占据这些亲水空间,海藻酸钠中的GG聚古洛糖醛酸嵌段上的羧基和羟基所带的多个氧原子与这些二价阳离子互相吸引,产生的螯合作用使得其分子链间紧密结合,协同作用加强,二价阳离子与G嵌段形成“egg—box”结构,最终形成三维网络结构的水凝胶,其中氯化钙作为钙源起到交联剂的作用。形成水凝胶后,加入碳酸钠,钙离子会吸引碳酸根离子原位生成纳米无定形碳酸钙,无定形纳米碳酸钙离子由凝胶连接形成无机盐包覆层。Sodium alginate is composed of two monomers (1,4)-β-D-mannuronic acid (M) and (1,4)-α-L-guluronic acid (G) through glycosidic bonds Unbranched block copolymers, the chain structures of these two uronic acids are very similar, only the difference in the hydroxyl position on them, but the spatial structure of their further polymerization into chains after ring formation is very different, which determines differences in their physicochemical properties. When the two monomers are linked differently, three different fragments can be formed: polymannuronic acid block (MM), polyguluronic acid block (GG), and mannuronic acid-guluronic acid Acid block (MG). When sodium alginate is in contact with a solution containing divalent cations such as Ca 2+ , the divalent cations will occupy these hydrophilic spaces, and the carboxyl and hydroxyl groups on the GG polyguluronic acid block in sodium alginate will Multiple oxygen atoms and these divalent cations attract each other, and the resulting chelation makes their molecular chains tightly bond and strengthen the synergy. The divalent cation and the G block form an "egg-box" structure, and finally form a three-dimensional network structure Hydrogels in which calcium chloride acts as a calcium source as a cross-linking agent. After the hydrogel is formed, sodium carbonate is added, and calcium ions attract carbonate ions to form nano-amorphous calcium carbonate in situ, and the amorphous nano-calcium carbonate ions are connected by the gel to form an inorganic salt coating layer.
因此,本发明的有益效果为:通过海藻酸钠凝胶吸附荧光染料分子,并且将凝胶层转化为无机盐保护层,使得荧光颜料不易渗色,并且具有良好的耐酸碱性,更加稳定。Therefore, the beneficial effects of the present invention are: the fluorescent dye molecules are adsorbed by the sodium alginate gel, and the gel layer is converted into an inorganic salt protection layer, so that the fluorescent pigment is not easy to bleed, has good acid and alkali resistance, and is more stable .
附图说明Description of drawings
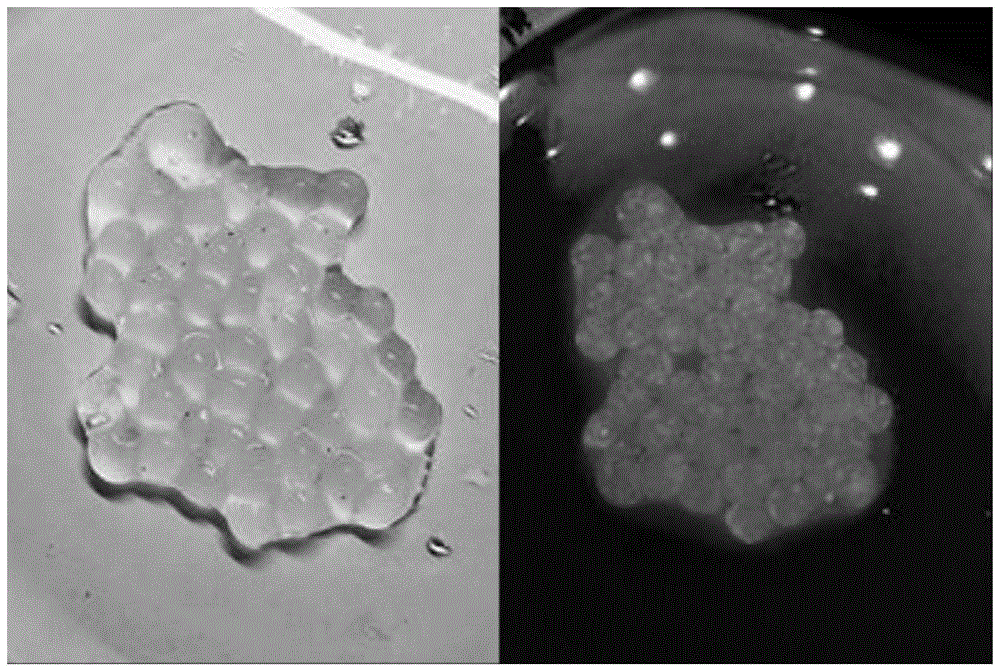
图1是本发明实施例1中制得的荧光颜料在自然光源和紫外光源下的实物图(左为自然光源,右为紫外光源)。Fig. 1 is a real picture of the fluorescent pigment prepared in Example 1 of the present invention under a natural light source and an ultraviolet light source (the left is the natural light source, and the right is the ultraviolet light source).
图2是不同染料初始浓度下的染料吸附率。Figure 2 is the dye adsorption rate at different initial dye concentrations.
图3是实施例1和对比例1在pH=5的条件下处理12h荧光颜料的荧光强度。FIG. 3 is the fluorescence intensity of the fluorescent pigments of Example 1 and Comparative Example 1 treated at pH=5 for 12 hours.
具体实施方式Detailed ways
下面结合附图与具体实施方式对本发明做进一步的描述。The present invention will be further described below with reference to the accompanying drawings and specific embodiments.
实施例1:Example 1:
一种荧光颜料的凝胶制备方法,包括如下步骤:A gel preparation method of a fluorescent pigment, comprising the following steps:
(1)将50g蒙皂石与0.2g罗丹明B混合研磨得到混合粉体;(1) 50g smectite and 0.2g rhodamine B are mixed and ground to obtain mixed powder;
(2)将10g海藻酸钠溶于200mL 50℃温水中,高速搅拌至液体中没有块状聚集物,接着将混合粉体加入浆料中,继续搅拌均匀2h得到混合浆料;(2) Dissolve 10 g of sodium alginate in 200 mL of warm water at 50°C, stir at high speed until there are no lumps in the liquid, then add the mixed powder into the slurry, and continue stirring for 2 hours to obtain a mixed slurry;
(3)边搅拌边向混合浆料中加入5.5g无水氯化钙,浆料中迅速出现凝胶絮状物,继续搅拌直到絮状物消失,形成均匀凝胶,得到凝胶中间体;(3) adding 5.5 g of anhydrous calcium chloride to the mixed slurry while stirring, and gel flocs rapidly appear in the slurry, and continue stirring until the flocs disappear to form a uniform gel to obtain a gel intermediate;
(4)向凝胶中间体中加入5g碳酸钠,溶液颜色发生变化,搅拌至颜色均一不变后沉淀,将所得产物洗涤至没有浮色后离心干燥研磨成粉得到所述荧光颜料。(4) Add 5 g of sodium carbonate to the gel intermediate, the color of the solution changes, stir until the color is uniform and precipitation, wash the obtained product until there is no floating color, centrifugal drying and grinding into powder to obtain the fluorescent pigment.
实施例2:Example 2:
一种荧光颜料的凝胶制备方法,包括如下步骤:A gel preparation method of a fluorescent pigment, comprising the following steps:
(1)将50g高岭石与0.5g溶剂黄172混合研磨得到混合粉体;(1) 50g kaolinite and 0.5g solvent yellow 172 are mixed and ground to obtain mixed powder;
(2)将20g海藻酸钠溶于200mL 50℃温水中,高速搅拌至液体中没有块状聚集物,接着将混合粉体加入浆料中,继续搅拌均匀1h得到混合浆料;(2) Dissolve 20 g of sodium alginate in 200 mL of 50°C warm water, stir at high speed until there is no lumpy aggregate in the liquid, then add the mixed powder into the slurry, and continue stirring for 1 hour to obtain a mixed slurry;
(3)边搅拌边向混合浆料中加入11g无水氯化钙,浆料中迅速出现凝胶絮状物,继续搅拌直到絮状物消失,形成均匀凝胶,得到凝胶中间体;(3) adding 11 g of anhydrous calcium chloride to the mixed slurry while stirring, gel flocs rapidly appear in the slurry, and continue stirring until the flocs disappear to form a uniform gel to obtain a gel intermediate;
(4)向凝胶中间体中加入10g碳酸钠,溶液颜色发生变化,搅拌至颜色均一不变后沉淀,将所得产物洗涤至没有浮色后离心干燥研磨成粉得到所述荧光颜料。(4) Add 10 g of sodium carbonate to the gel intermediate, the color of the solution changes, stir until the color is uniform and precipitation, wash the obtained product until there is no floating color, centrifugal drying and grinding into powder to obtain the fluorescent pigment.
实施例3:Example 3:
一种荧光颜料的凝胶制备方法,包括如下步骤:A gel preparation method of a fluorescent pigment, comprising the following steps:
(1)将15g皂石、35g蒙脱石和0.15g罗丹明6G混合研磨得到混合粉体;(1) 15g saponite, 35g montmorillonite and 0.15g rhodamine 6G are mixed and ground to obtain mixed powder;
(2)将25g海藻酸钠溶于350mL 40℃温水中,高速搅拌至液体中没有块状聚集物,接着将混合粉体加入浆料中,继续搅拌均匀1h得到混合浆料;(2) Dissolve 25 g of sodium alginate in 350 mL of warm water at 40°C, stir at high speed until there is no lumpy aggregate in the liquid, then add the mixed powder into the slurry, and continue stirring for 1 hour to obtain a mixed slurry;
(3)边搅拌边向混合浆料中加入6.5g无水氯化钙,浆料中迅速出现凝胶絮状物,继续搅拌直到絮状物消失,形成均匀凝胶,得到凝胶中间体;(3) 6.5g of anhydrous calcium chloride was added to the mixed slurry while stirring, and gel flocs rapidly appeared in the slurry, and continued stirring until the flocs disappeared to form a uniform gel to obtain a gel intermediate;
(4)向凝胶中间体中加入6g碳酸钠,溶液颜色发生变化,搅拌至颜色均一不变后沉淀,将所得产物洗涤至没有浮色后离心干燥研磨成粉得到所述荧光颜料。(4) Add 6g of sodium carbonate to the gel intermediate, the color of the solution changes, stir until the color is uniform and precipitation, wash the obtained product until there is no floating color, centrifugal drying and grinding into powder to obtain the fluorescent pigment.
实施例4:Example 4:
一种荧光颜料的凝胶制备方法,包括如下步骤:A gel preparation method of a fluorescent pigment, comprising the following steps:
(1)将50g蒙皂石和2.5g罗丹明B混合研磨得到混合粉体;(1) 50g smectite and 2.5g rhodamine B are mixed and ground to obtain mixed powder;
(2)将10g海藻酸钠溶于200mL 80℃温水中,高速搅拌至液体中没有块状聚集物,接着将混合粉体加入浆料中,继续搅拌均匀1h得到混合浆料;(2) Dissolve 10 g of sodium alginate in 200 mL of warm water at 80°C, stir at high speed until there are no lumps in the liquid, then add the mixed powder into the slurry, and continue stirring for 1 hour to obtain a mixed slurry;
(3)边搅拌边向混合浆料中加入5.5g无水氯化钙,浆料中迅速出现凝胶絮状物,继续搅拌直到絮状物消失,形成均匀凝胶,得到凝胶中间体;(3) adding 5.5 g of anhydrous calcium chloride to the mixed slurry while stirring, and gel flocs rapidly appear in the slurry, and continue stirring until the flocs disappear to form a uniform gel to obtain a gel intermediate;
(4)向凝胶中间体中加入5g碳酸钠,溶液颜色发生变化,搅拌至颜色均一不变后沉淀,将所得产物洗涤至没有浮色后离心干燥研磨成粉得到所述荧光颜料。(4) Add 5 g of sodium carbonate to the gel intermediate, the color of the solution changes, stir until the color is uniform and precipitation, wash the obtained product until there is no floating color, centrifugal drying and grinding into powder to obtain the fluorescent pigment.
实施例5:Example 5:
一种荧光颜料的凝胶制备方法,包括如下步骤:A gel preparation method of a fluorescent pigment, comprising the following steps:
(1)将25g皂石、25g蒙脱石和0.15g罗丹明6G混合研磨得到混合粉体;(1) 25g saponite, 25g montmorillonite and 0.15g rhodamine 6G are mixed and ground to obtain mixed powder;
(2)将25g海藻酸钠溶于350mL 50℃温水中,高速搅拌至液体中没有块状聚集物,接着将混合粉体加入浆料中,继续搅拌均匀5h得到混合浆料;(2) Dissolve 25g of sodium alginate in 350mL of 50°C warm water, stir at high speed until there are no lumps in the liquid, then add the mixed powder into the slurry, and continue stirring for 5h to obtain a mixed slurry;
(3)边搅拌边向混合浆料中加入62.5g无水氯化钙,浆料中迅速出现凝胶絮状物,继续搅拌直到絮状物消失,形成均匀凝胶,得到凝胶中间体;(3) adding 62.5g of anhydrous calcium chloride to the mixed slurry while stirring, gel flocs rapidly appear in the slurry, continue to stir until the flocs disappear, form a uniform gel, and obtain a gel intermediate;
(4)向凝胶中间体中加入60g碳酸钠,溶液颜色发生变化,搅拌至颜色均一不变后沉淀,将所得产物洗涤至没有浮色后离心干燥研磨成粉得到所述荧光颜料。(4) Add 60g of sodium carbonate to the gel intermediate, the color of the solution changes, stir until the color is uniform and precipitation, wash the obtained product until there is no floating color, centrifugal drying and grinding into powder to obtain the fluorescent pigment.
实施例6:Example 6:
一种荧光颜料的凝胶制备方法,包括如下步骤:A gel preparation method of a fluorescent pigment, comprising the following steps:
(1)将50g高岭石和0.5g溶剂黄172混合研磨得到混合粉体;(1) 50g kaolinite and 0.5g solvent yellow 172 are mixed and ground to obtain mixed powder;
(2)将0.8g海藻酸钠溶于500mL 50℃温水中,高速搅拌至液体中没有块状聚集物,接着将混合粉体加入浆料中,继续搅拌均匀3h得到混合浆料;(2) Dissolve 0.8 g of sodium alginate in 500 mL of warm water at 50°C, stir at high speed until there are no lumps in the liquid, then add the mixed powder into the slurry, and continue stirring for 3 hours to obtain a mixed slurry;
(3)边搅拌边向混合浆料中加入1.6g无水氯化钙,浆料中迅速出现凝胶絮状物,继续搅拌直到絮状物消失,形成均匀凝胶,得到凝胶中间体;(3) 1.6 g of anhydrous calcium chloride was added to the mixed slurry while stirring, and gel flocs quickly appeared in the slurry, and continued stirring until the flocs disappeared, forming a uniform gel, and obtaining a gel intermediate;
(4)向凝胶中间体中加入1.5g碳酸钠,溶液颜色发生变化,搅拌至颜色均一不变后沉淀,将所得产物洗涤至没有浮色后离心干燥研磨成粉得到所述荧光颜料。(4) Add 1.5g of sodium carbonate to the gel intermediate, the color of the solution changes, stir until the color is uniform and then precipitate, wash the obtained product until there is no floating color, centrifugal drying and grinding into powder to obtain the fluorescent pigment.
实施例7:Example 7:
实施例7与实施例6的区别在于,溶剂黄172的质量为1.5g,海藻酸钠的质量为1.32g,其余均与实施例6中相同。The difference between Example 7 and Example 6 is that the mass of Solvent Yellow 172 is 1.5 g, the mass of sodium alginate is 1.32 g, and the rest are the same as in Example 6.
实施例8:Example 8:
实施例8与实施例6的区别在于,实施例6中荧光染料为2.38g罗丹明B,其余均与实施例6中相同。The difference between Example 8 and Example 6 is that the fluorescent dye in Example 6 is 2.38 g of Rhodamine B, and the rest are the same as those in Example 6.
对比例1:Comparative Example 1:
市售溶剂黄172荧光颜料。Commercially available solvent yellow 172 fluorescent pigment.
对上述实施例中染料的吸附率(吸附时间6h)进行测定,结果如表1和图2所示。The adsorption rate (adsorption time 6h) of the dyes in the above examples was measured, and the results are shown in Table 1 and FIG. 2 .
表1:染料吸附率。Table 1: Dye adsorption rates.
从表1和图2中可以看出,采用本发明中的方法制备的荧光颜料,对染料的吸附性能良好,并且染料的初始浓度越低,吸附率越高。原因是由于染料初始浓度越高,越易发生聚集现象,聚集后聚集体因为空间阻碍且不易离解,所以不易进入层间。It can be seen from Table 1 and Figure 2 that the fluorescent pigment prepared by the method of the present invention has good adsorption performance to dye, and the lower the initial concentration of dye, the higher the adsorption rate. The reason is that the higher the initial concentration of the dye, the more likely the aggregation phenomenon will occur. After aggregation, the aggregates are not easy to enter the interlayer because of space hindrance and not easy to dissociate.
测定实施例1和对比例1中的市售荧光颜料在pH=5的条件下处理12h后的荧光强度变化,结果如图3所示。从图3中可以看出,使用本发明中的方法制备出的荧光颜料耐酸碱性与现有的荧光颜料相比,有了显著提升。The fluorescence intensity changes of the commercially available fluorescent pigments in Example 1 and Comparative Example 1 after being treated for 12 hours under the condition of pH=5 were measured, and the results are shown in FIG. 3 . It can be seen from FIG. 3 that the acid and alkali resistance of the fluorescent pigment prepared by the method of the present invention is significantly improved compared with the existing fluorescent pigment.
Claims (8)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010048937.1A CN111286233A (en) | 2020-01-16 | 2020-01-16 | A kind of gel preparation method of fluorescent pigment |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010048937.1A CN111286233A (en) | 2020-01-16 | 2020-01-16 | A kind of gel preparation method of fluorescent pigment |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN111286233A true CN111286233A (en) | 2020-06-16 |
Family
ID=71021214
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010048937.1A Pending CN111286233A (en) | 2020-01-16 | 2020-01-16 | A kind of gel preparation method of fluorescent pigment |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111286233A (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112159623A (en) * | 2020-09-28 | 2021-01-01 | 成都玉龙化工有限公司 | High-temperature-resistant coating and preparation method thereof |
| CN115468938A (en) * | 2022-08-24 | 2022-12-13 | 中国科学院宁波材料技术与工程研究所 | Fluorescence-based color correction card and application thereof |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104666250A (en) * | 2015-03-06 | 2015-06-03 | 华南农业大学 | Preparation method of chlortetracycline sustained-release microspheres |
| CN107998462A (en) * | 2017-11-10 | 2018-05-08 | 西安交通大学医学院第附属医院 | A magnetic gel for assisting endoscopic mucosal dissection, its preparation and application system |
| CN109894096A (en) * | 2019-04-03 | 2019-06-18 | 深圳市清研环境科技有限公司 | Adsorb the microballoon and preparation method thereof of dyestuff composite hydrogel |
| CN110499665A (en) * | 2019-08-13 | 2019-11-26 | 湖南省南盾防伪技术实业有限公司 | Calcium carbonate wraps up DNA- fluorescent dye composite Nano gel Double anti-counterfeit preparation method for coating |
-
2020
- 2020-01-16 CN CN202010048937.1A patent/CN111286233A/en active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104666250A (en) * | 2015-03-06 | 2015-06-03 | 华南农业大学 | Preparation method of chlortetracycline sustained-release microspheres |
| CN107998462A (en) * | 2017-11-10 | 2018-05-08 | 西安交通大学医学院第附属医院 | A magnetic gel for assisting endoscopic mucosal dissection, its preparation and application system |
| CN109894096A (en) * | 2019-04-03 | 2019-06-18 | 深圳市清研环境科技有限公司 | Adsorb the microballoon and preparation method thereof of dyestuff composite hydrogel |
| CN110499665A (en) * | 2019-08-13 | 2019-11-26 | 湖南省南盾防伪技术实业有限公司 | Calcium carbonate wraps up DNA- fluorescent dye composite Nano gel Double anti-counterfeit preparation method for coating |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112159623A (en) * | 2020-09-28 | 2021-01-01 | 成都玉龙化工有限公司 | High-temperature-resistant coating and preparation method thereof |
| CN115468938A (en) * | 2022-08-24 | 2022-12-13 | 中国科学院宁波材料技术与工程研究所 | Fluorescence-based color correction card and application thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN104530294B (en) | Preparation method of high-strength silicon dioxide and clay dual nano-composite hydrogel | |
| CN1226319C (en) | Viscosity highly increased water-soluble hydrophobic association copolymer and preparation method and use thereof | |
| CN103145920B (en) | Preparation method of temperature, pH and ultraviolet multi-stimuli-responsive semi-interpenetrating network nanocomposite hydrogel | |
| CN115232261B (en) | Hybrid gelling agent for oilfield acidification based on POSS-based cross-linking agent and preparation method thereof | |
| CN104307491B (en) | A kind of modified graphene of efficient absorption methyl orange dye and preparation method thereof | |
| CN108671859B (en) | Method for preparing Janus particles based on lysozyme nano-film | |
| CN105944634A (en) | Preparation method of color composite microspheres with high reactive dye adsorption capacity | |
| CN111286233A (en) | A kind of gel preparation method of fluorescent pigment | |
| CN103193916A (en) | Preparation method of polymeric microspheres for electrophoretic display | |
| CN107814434A (en) | A kind of modified soluble chitosan flocculant and preparation method thereof | |
| CN103556505A (en) | Preparation method of dyeing assistant | |
| CN115323799B (en) | Structural chromogenic cellulose fabric and preparation method thereof | |
| CN103861566B (en) | A kind of preparation method of efficient adsorption modified starch microspheres and application | |
| CN106732423A (en) | A kind of preparation and application of hydroxyethyl cellulose/concave convex rod/sodium humate composite aquogel | |
| CN108033453A (en) | A kind of preparation method of modified precipitated silica | |
| CN101143956A (en) | A low-cost super-absorbent composite water-retaining agent and its preparation method | |
| CN114539444A (en) | A kind of modified carboxymethyl chitosan quaternary ammonium salt and preparation method thereof | |
| CN110354827A (en) | A kind of synthetic method of magnetic hydrogel adsorbent material | |
| CN108341972A (en) | Preparation method of ion coordination crosslinking natural polymer self-repairing hydrogel | |
| CN106905445B (en) | A kind of Polyaminated chitosan decolorising agent of carboxymethyl-and its preparation method and application | |
| CN106008779A (en) | Making method of crosslinked poly(sodium-p-styrenesulfonate) gel microsphere | |
| CN1137991C (en) | Process for immobilizing enzyme/microbe with polyvinyl alcohol | |
| CN106111085B (en) | A kind of material and preparation method thereof of the chitosan-based adsorbent of quick processing waste water from dyestuff | |
| CN109021476B (en) | Preparation method of high-strength anti-aging sodium polyacrylate water-absorbent resin | |
| CN102897826B (en) | Method for preparing nanometer zinc oxide composite material from super concentrated emulsion |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20200616 |