CN111229119B - Mixed system containing fatty acid type surfactant and preparation method thereof - Google Patents
Mixed system containing fatty acid type surfactant and preparation method thereof Download PDFInfo
- Publication number
- CN111229119B CN111229119B CN201811446089.9A CN201811446089A CN111229119B CN 111229119 B CN111229119 B CN 111229119B CN 201811446089 A CN201811446089 A CN 201811446089A CN 111229119 B CN111229119 B CN 111229119B
- Authority
- CN
- China
- Prior art keywords
- fatty acid
- chain fatty
- acid
- long
- mixed system
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 235000014113 dietary fatty acids Nutrition 0.000 title claims abstract description 44
- 239000000194 fatty acid Substances 0.000 title claims abstract description 44
- 229930195729 fatty acid Natural products 0.000 title claims abstract description 44
- 150000004665 fatty acids Chemical class 0.000 title claims abstract description 44
- 239000004094 surface-active agent Substances 0.000 title claims abstract description 34
- 238000002360 preparation method Methods 0.000 title abstract description 9
- 150000004668 long chain fatty acids Chemical class 0.000 claims abstract description 52
- BXYVQNNEFZOBOZ-UHFFFAOYSA-N n-[3-(dimethylamino)propyl]-n',n'-dimethylpropane-1,3-diamine Chemical compound CN(C)CCCNCCCN(C)C BXYVQNNEFZOBOZ-UHFFFAOYSA-N 0.000 claims abstract description 26
- 238000002156 mixing Methods 0.000 claims abstract description 24
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 18
- 238000000034 method Methods 0.000 claims abstract description 16
- 239000007787 solid Substances 0.000 claims abstract description 14
- 125000004432 carbon atom Chemical group C* 0.000 claims abstract description 10
- POULHZVOKOAJMA-UHFFFAOYSA-N dodecanoic acid Chemical compound CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 claims description 19
- FRPZMMHWLSIFAZ-UHFFFAOYSA-N 10-undecenoic acid Chemical compound OC(=O)CCCCCCCCC=C FRPZMMHWLSIFAZ-UHFFFAOYSA-N 0.000 claims description 10
- KHAVLLBUVKBTBG-UHFFFAOYSA-N caproleic acid Natural products OC(=O)CCCCCCCC=C KHAVLLBUVKBTBG-UHFFFAOYSA-N 0.000 claims description 10
- IPCSVZSSVZVIGE-UHFFFAOYSA-N hexadecanoic acid Chemical compound CCCCCCCCCCCCCCCC(O)=O IPCSVZSSVZVIGE-UHFFFAOYSA-N 0.000 claims description 10
- 125000005480 straight-chain fatty acid group Chemical group 0.000 claims description 8
- SZHOJFHSIKHZHA-UHFFFAOYSA-N tridecanoic acid Chemical compound CCCCCCCCCCCCC(O)=O SZHOJFHSIKHZHA-UHFFFAOYSA-N 0.000 claims description 8
- ZDPHROOEEOARMN-UHFFFAOYSA-N undecanoic acid Chemical compound CCCCCCCCCCC(O)=O ZDPHROOEEOARMN-UHFFFAOYSA-N 0.000 claims description 8
- TUNFSRHWOTWDNC-UHFFFAOYSA-N Myristic acid Natural products CCCCCCCCCCCCCC(O)=O TUNFSRHWOTWDNC-UHFFFAOYSA-N 0.000 claims description 6
- 239000000203 mixture Substances 0.000 claims description 6
- 235000021314 Palmitic acid Nutrition 0.000 claims description 5
- 229960002703 undecylenic acid Drugs 0.000 claims description 5
- YAQXGBBDJYBXKL-UHFFFAOYSA-N iron(2+);1,10-phenanthroline;dicyanide Chemical compound [Fe+2].N#[C-].N#[C-].C1=CN=C2C3=NC=CC=C3C=CC2=C1.C1=CN=C2C3=NC=CC=C3C=CC2=C1 YAQXGBBDJYBXKL-UHFFFAOYSA-N 0.000 claims description 4
- WQEPLUUGTLDZJY-UHFFFAOYSA-N n-Pentadecanoic acid Natural products CCCCCCCCCCCCCCC(O)=O WQEPLUUGTLDZJY-UHFFFAOYSA-N 0.000 claims description 4
- 230000003247 decreasing effect Effects 0.000 claims description 3
- 229920006395 saturated elastomer Polymers 0.000 claims 2
- 238000004519 manufacturing process Methods 0.000 claims 1
- 238000002425 crystallisation Methods 0.000 abstract description 7
- 230000008025 crystallization Effects 0.000 abstract description 7
- 230000000052 comparative effect Effects 0.000 description 11
- 108010052749 N-isobutylidenedipivalamide Proteins 0.000 description 10
- WVJLEJNAJZPAHH-UHFFFAOYSA-N n-[1-(2,2-dimethylpropanoylamino)-2-methylpropyl]-2,2-dimethylpropanamide Chemical compound CC(C)(C)C(=O)NC(C(C)C)NC(=O)C(C)(C)C WVJLEJNAJZPAHH-UHFFFAOYSA-N 0.000 description 10
- 150000001412 amines Chemical class 0.000 description 7
- 239000000243 solution Substances 0.000 description 7
- 230000007704 transition Effects 0.000 description 7
- 238000000113 differential scanning calorimetry Methods 0.000 description 6
- 238000010438 heat treatment Methods 0.000 description 6
- 238000005191 phase separation Methods 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- 150000004985 diamines Chemical class 0.000 description 5
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 4
- 238000001816 cooling Methods 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- 159000000000 sodium salts Chemical class 0.000 description 4
- 239000013078 crystal Substances 0.000 description 3
- KYYWBEYKBLQSFW-UHFFFAOYSA-N hexadecanoic acid Chemical compound CCCCCCCCCCCCCCCC(O)=O.CCCCCCCCCCCCCCCC(O)=O KYYWBEYKBLQSFW-UHFFFAOYSA-N 0.000 description 3
- 239000000693 micelle Substances 0.000 description 3
- 230000002035 prolonged effect Effects 0.000 description 3
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 3
- 230000007928 solubilization Effects 0.000 description 3
- 238000005063 solubilization Methods 0.000 description 3
- ZTUXEFFFLOVXQE-UHFFFAOYSA-N tetradecanoic acid Chemical compound CCCCCCCCCCCCCC(O)=O.CCCCCCCCCCCCCC(O)=O ZTUXEFFFLOVXQE-UHFFFAOYSA-N 0.000 description 3
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- 125000000217 alkyl group Chemical group 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 125000003916 ethylene diamine group Chemical group 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 230000003381 solubilizing effect Effects 0.000 description 2
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 240000007839 Kleinhovia hospita Species 0.000 description 1
- 239000005639 Lauric acid Substances 0.000 description 1
- 208000010195 Onychomycosis Diseases 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- QVYARBLCAHCSFJ-UHFFFAOYSA-N butane-1,1-diamine Chemical compound CCCC(N)N QVYARBLCAHCSFJ-UHFFFAOYSA-N 0.000 description 1
- 238000012824 chemical production Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 239000008367 deionised water Substances 0.000 description 1
- 229910021641 deionized water Inorganic materials 0.000 description 1
- 239000003599 detergent Substances 0.000 description 1
- 238000006073 displacement reaction Methods 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 238000005265 energy consumption Methods 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- SYECJBOWSGTPLU-UHFFFAOYSA-N hexane-1,1-diamine Chemical compound CCCCCC(N)N SYECJBOWSGTPLU-UHFFFAOYSA-N 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 230000010355 oscillation Effects 0.000 description 1
- 238000005504 petroleum refining Methods 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 238000007670 refining Methods 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 238000001338 self-assembly Methods 0.000 description 1
- 239000000344 soap Substances 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 201000005882 tinea unguium Diseases 0.000 description 1
- 235000019871 vegetable fat Nutrition 0.000 description 1
- 238000005303 weighing Methods 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K23/00—Use of substances as emulsifying, wetting, dispersing, or foam-producing agents
- C09K23/017—Mixtures of compounds
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
The invention relates to the field of surfactants, and discloses a mixed system containing a fatty acid type surfactant and a preparation method thereof, wherein the mixed system contains long-chain fatty acid with 11-16 carbon atoms, 3 '-iminobis (N, N-dimethylpropylamine) and water, wherein the molar ratio of the long-chain fatty acid to the 3, 3' -iminobis (N, N-dimethylpropylamine) is 45-305: 100. In another aspect, the disclosed method for preparing a mixed system containing a fatty acid-type surfactant comprises mixing a long-chain fatty acid having 11 to 16 carbon atoms, 3' -iminobis (N, N-dimethylpropylamine) and water until the solid is dissolved. In addition, the invention also discloses a mixed system prepared by the method. The mixed system of the present invention can be stably preserved without crystallization even if the long-chain fatty acid is present at a relatively high concentration. Moreover, the formula and the preparation method of the invention are simple and easy to popularize and apply.
Description
Technical Field
The invention relates to the field of surfactants, in particular to a mixed system containing fatty acid type surfactants and a preparation method thereof.
Background
Fatty acid and sodium salt of fatty acid are a kind of surfactant with wide application range, and relates to civil use, oil exploitation, chemical industry and other aspects. The fatty acid surfactant is healthy and environment-friendly, such as soaps used in daily life and chemical production. And can be prepared by petroleum refining or obtained from animal and vegetable fats in large quantities, and has the advantages of wide sources, low cost and the like. Especially for petrochemical industry, the long-chain fatty acid obtained from oil refining products has more cost advantage than the sodium sulfonate surfactant with long chain alkyl used in large amount at present. However, the fatty acid surfactants have poor solubility and are difficult to exist in a liquid homogeneous form, and have great disadvantages compared with long-chain alkyl sodium sulfonate surfactants. Therefore, obtaining a high concentration solubilization system of fatty acids is the key to achieving its industrial application.
Surfactants exist in a variety of aggregated forms in a stable formulation, including true solutions (required below the critical micelle concentration CMC), micelles, worm-like micelles, and lamellar vesicles. The characters reflected in the formula are dilute solution, viscous solution, gel and solid. Conventional fatty acid surfactants are mostly in the form of their sodium salts. The sodium salt of fatty acid is solid at normal temperature and is easy to phase separate when being miscible with water (the sodium salt of fatty acid is a white solid precipitate, and the upper layer is a clear solution), so that a stable solution system cannot be formed. In recent years, the positive charge quaternary ammonium salt and organic amine are used for solubilizing the fatty acid surfactant, but the obtained solubilizing system can just neutralize the fatty acid to form a stable miscible system only when the molar ratio of the fatty acid to the quaternary ammonium salt (or the organic amine) is 1: 1.
Document Douliez, j.p.; navailes, l.; phase behavior of systems that neutralize Fatty acids with diamines was studied by Nallet, F.Self-Assembly of Fatty Acid-Alkylbodiamine salts.Langmuir 2006,22, 622-. Compared with quaternary ammonium salt and monoamine solubilized fatty acid, diamine solubilized fatty acid has more advantages, a mixed system of fatty acid and diamine in various molar ratios can be obtained, and the composition range of the formula is expanded. However, solubilization of fatty acid-type surfactants with diamines presents an inevitable problem, namely phase-separated crystallization of fatty acids. For example, in this document (Langmuir 2006,22,622-627), when hexadecanoic acid and diamines such as ethylenediamine, butanediamine, hexanediamine, etc. are mixed at a molar ratio of 0.5 to 2, a mixed solution aggregated in lamellar vesicles can be obtained at a fatty acid concentration of 1% (fatty acid/water mass%). However, even at a low concentration of 1% after standing at room temperature for one day, white crystalline solids appeared in the samples, i.e., crystallization occurred, and the performance properties were impaired. Thus, this disadvantage severely limits the large-scale industrial application of diamine-solubilized fatty acid systems.
In addition, the petrochemical industry and the daily chemical industry require higher concentrations of the fatty acid surfactant. The low concentration not only causes the increase of water consumption, but also correspondingly increases the energy consumption of transportation and transportation. In other words, the current fatty acid solubilization system cannot satisfy both the requirements of stability against crystallization and high concentration. In addition, under the condition of high surfactant concentration, the properties and the fluidity of the surfactant can be adjusted by simply adjusting the component ratio, so that the application of the surfactant formula is greatly facilitated.
Disclosure of Invention
The invention aims to overcome the problems of low concentration of fatty acid and easy crystallization in the prior art, and provides a mixed system containing a fatty acid type surfactant and a preparation method thereof.
In order to achieve the above object, the present invention provides, in one aspect, a mixed system containing a fatty acid type surfactant, the mixed system containing a long-chain fatty acid having 11 to 16 carbon atoms, 3 '-iminobis (N, N-dimethylpropylamine), and water, wherein the molar ratio between the long-chain fatty acid and the 3, 3' -iminobis (N, N-dimethylpropylamine) is 45 to 305: 100.
In another aspect, the present invention provides a method for preparing a mixed system containing a fatty acid-type surfactant, which comprises mixing a long-chain fatty acid having 11 to 16 carbon atoms, 3 '-iminobis (N, N-dimethylpropylamine) and water until the solid is dissolved, wherein the molar ratio between the long-chain fatty acid and the 3, 3' -iminobis (N, N-dimethylpropylamine) is 45 to 305: 100.
In addition, the invention also provides a mixed system prepared by the method.
The long-chain fatty acid in the mixed system (miscible system) can exist in higher concentration (up to 15 weight percent), and the proportion of the long-chain fatty acid and other components can be adjusted in a wider range, so that the miscible system is stably stored and does not crystallize. The carbon chain of the 3, 3' -imino-bis (N, N-dimethylpropylamine) is longer, and the double-ended Bola type surfactant can be formed by the action of charges. Therefore, the miscible system of the present invention is more effective as a surfactant. Moreover, the formula and the preparation method of the invention are simple and easy to popularize and apply.
Drawings
FIG. 1 is a graph of Differential Scanning Calorimetry (DSC) measurements of a mixed system according to one embodiment of the invention;
FIG. 2 is a chart showing the results of DSC measurement of the mixed system obtained in comparative example 2.
Detailed Description
The endpoints of the ranges and any values disclosed herein are not limited to the precise range or value, and such ranges or values should be understood to encompass values close to those ranges or values. For ranges of values, between the endpoints of each of the ranges and the individual points, and between the individual points may be combined with each other to give one or more new ranges of values, and these ranges of values should be considered as specifically disclosed herein.
The mixed system containing the fatty acid type surfactant comprises a long-chain fatty acid with 11-16 carbon atoms, 3 '-iminobis (N, N-dimethylpropylamine) and water, wherein the molar ratio of the long-chain fatty acid to the 3, 3' -iminobis (N, N-dimethylpropylamine) is 45-305:100, preferably 50-300: 100.
According to the present invention, since the use concentration of the long-chain fatty acid can be increased by blending a specific long-chain fatty acid and an amine, the content of the long-chain fatty acid may be as high as 15% by weight, and preferably, the content of the long-chain fatty acid is 0.1 to 15% by weight based on the total weight of the mixed system.
According to the present invention, the long-chain fatty acid is a fatty acid having 11 to 16 carbon atoms, preferably a straight-chain fatty acid including a saturated straight-chain fatty acid and/or an unsaturated straight-chain fatty acid, and more preferably, the long-chain fatty acid is at least one selected from the group consisting of undecanoic acid, 10-undecylenic acid (undecylenic acid), dodecanoic acid (lauric acid), tridecanoic acid, tetradecanoic acid (myristic acid), pentadecanoic acid, and hexadecanoic acid (palmitic acid).
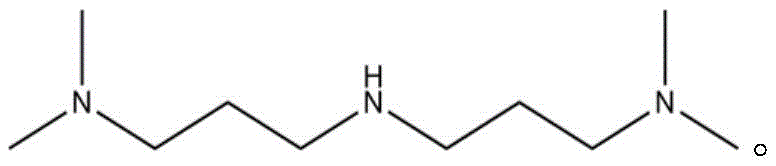
According to the invention, the structural formula of the 3, 3' -imino-bis (N, N-dimethylpropylamine) is shown in the specification
The method for preparing the mixed system containing the fatty acid type surfactant comprises the step of mixing long-chain fatty acid with 11-16 carbon atoms, 3 '-iminobis (N, N-dimethylpropylamine) and water until solid is dissolved, wherein the molar ratio of the long-chain fatty acid to the 3, 3' -iminobis (N, N-dimethylpropylamine) is 45-305:100, and preferably 50-300: 100.
In the present invention, since the use concentration of the long-chain fatty acid can be increased by blending a specific long-chain fatty acid and an amine, the content of the long-chain fatty acid may be as high as 15% by weight, and preferably, the content of the long-chain fatty acid is 0.1 to 15% by weight based on the total weight of the mixed system.
According to the present invention, the long-chain fatty acid is a fatty acid having 11 to 16 carbon atoms, preferably a straight-chain fatty acid including a saturated straight-chain fatty acid and/or an unsaturated straight-chain fatty acid, and more preferably, the long-chain fatty acid is at least one selected from the group consisting of undecanoic acid, 10-undecylenic acid (undecylenic acid), dodecanoic acid (lauric acid), tridecanoic acid, tetradecanoic acid (myristic acid), pentadecanoic acid, and hexadecanoic acid (palmitic acid).
In the present invention, the mixing conditions are not particularly limited as long as the long-chain fatty acid is dissolved to form a miscible system. The solution can be accelerated by adopting a mode of standing for a long time, keeping constant temperature after heating, repeatedly heating and cooling, or a physical mode of vortex oscillation, stirring and the like. Preferably, the mixing is carried out at a temperature of 60-80 ℃.
In the present invention, the mixing may be carried out in various conventional manners, and the order of mixing the components is not particularly limited.
Preferably, the mixing mode is as follows: firstly, long-chain fatty acid is contacted with 3, 3' -imino-bis (N, N-dimethylpropylamine), then water is introduced, and the mixture is placed at a temperature of between 60 and 80 ℃ until the solid is dissolved;
or, the mixing mode is as follows: firstly, long-chain fatty acid is contacted with 3, 3' -imino-bis (N, N-dimethylpropylamine), then water is introduced, the temperature is raised to 60-80 ℃, the temperature is kept for more than 10min, then the temperature is lowered to room temperature or 0 ℃, the temperature is kept for more than 10min, and the temperature is raised and lowered repeatedly until the solid is dissolved. Unless otherwise stated, "room temperature" is generally in the range of 15 to 25 ℃.
In the above preferred mixing mode, there is no special requirement for the duration of the constant temperature, and the duration can be long or short, and the experimenter can adjust and control according to the actual experimental requirements, but considering that the time is shortened as much as possible, the preparation efficiency is improved, the time of the constant temperature is usually below 72h, and each repetition stage can be the same or different.
In addition, the invention also provides a mixed system prepared by the method.
The fatty acid type surfactant is widely applied to the fields of chemical environmental protection, daily necessities such as laundry detergent (emulsifier and/or stabilizer), tertiary oil recovery (oil displacement agent) and the like, and the undecylenic acid can be applied to the fields of medicinal preparations, coating paste for treating onychomycosis and the like.
The present invention will be described in detail below by way of examples.
In the following examples, 10-undecylenic acid was purchased from echiei (shanghai) chemical industry development limited; dodecanoic acid (lauric acid), tetradecanoic acid (myristic acid), hexadecanoic acid (palmitic acid) were purchased from national pharmaceutical group chemicals, ltd; ethylenediamine (EDA) was purchased from alatin reagent; n, N ' -tetramethyl-1, 3-propanediamine (TMPDA), 3 ' -diaminodipropylamine (DADPA) and 3,3 ' -iminobis (N, N-dimethylpropylamine) (IBDPA) were purchased from scheia (shanghai) chemical industry development limited.
Examples 1 to 36
Weighing long-chain fatty acid and IBDPA according to the dosage shown in the table 1, placing the long-chain fatty acid and the IBDPA in a sample bottle, and adding deionized water; the mixing mode after sealing is constant temperature or repeatedly increasing and decreasing temperature, which is shown in the column "constant temperature" or "repeatedly increasing and decreasing temperature" of the mixing mode in table 1. The constant temperature method comprises heating a sample to a certain temperature (see the column of table 1 "temperature") for a certain time (see the column of table 1 "constant temperature time") until the solid is completely dissolved to obtain a mixed system sample; the repeated heating and cooling method comprises heating the sample to a certain temperature (see the column of "temperature" in table 1), keeping the temperature for a certain time (see the column of "constant temperature time" in table 1), cooling to about 0 deg.C, keeping the temperature for 10min, and repeatedly heating and cooling for multiple times (see the column of "constant temperature time" in table 1).
In Table 1, "molar ratio" refers to the molar ratio between long chain fatty acids and IBDPA, and "weight ratio" refers to the weight percentage of long chain fatty acids based on the total weight of the sample.
Comparative example 1
A mixed system containing a fatty acid type surfactant was prepared in the same manner as in example 27, except that IBDPA was replaced with DADPA and the mixing time was prolonged.
Comparative example 2
A mixed system containing a fatty acid type surfactant was prepared in the same manner as in example 28, except that IBDPA was replaced with EDA and the mixing time was extended.
Comparative example 3
A mixed system containing a fatty acid type surfactant was prepared in the same manner as in example 29, except that IBDPA was replaced with EDA and the mixing time was prolonged.
Comparative example 4
A mixed system containing a fatty acid type surfactant was prepared in the same manner as in example 27, except that IBDPA was replaced with TMPDA and the mixing time was prolonged.
TABLE 1
Test example 1
(1) The samples obtained in the above examples and comparative examples were left at room temperature (around 20 ℃ C.) for 24 hours, 72 hours, 1 week, 1 month, and 3 months, and then observed for crystal formation. The observations are shown in table 2 under the column "properties" (showing the properties of each sample at various time points), with visually observed viscosity grades in parentheses:
viscosity is comparable to water: 0
Slightly greater viscosity than water: +
Semi-fluid gel: ++
Oil paste solid: +++
(2) Phase transition signals (fatty acids) of samples prepared in examples and comparative examples after being left for 3 months were measured at a temperature increase rate of 2 ℃/min using a Differential Scanning Calorimeter (DSC) to determine whether phase separation occurred. Wherein, when phase separation does not occur in an IBDPA solubilized aqueous solution system, hexadecanoic acid has an endothermic phase transition peak at 36-39 ℃; the tetradecanoic acid has an endothermic phase transition peak at 14-21 ℃; the dodecanoic acid and 10-undecylenic acid have no phase transition peak at 0 deg.C or higher. In the sample in which the crystallization phase separation did not occur, the peak temperature of the phase transition peak detected by DSC was equal to or lower than the above peak temperature. 10-undecylenic acid and lauric acid system which are separated out by phase separation have phase transition peak at 0 ℃ or above; the tetradecanoic acid and hexadecanoic acid system which is separated out by phase separation will have a phase transition peak with higher temperature. Thus, the presence or absence of fatty acid crystals was judged. The results of the measurements are shown in Table 2 below. The DSC results of the mixed systems prepared in example 12 and comparative example 2 are shown in FIG. 1 and FIG. 2, respectively. FIG. 1 is a mixed system of dodecanoic acid and IBDPA, wherein the dodecanoic acid is not crystallized at 15 wt%; fig. 2 is a comparative dodecanoic acid and EDA mixing system in which crystallization of dodecanoic acid phase separation occurred at 5 wt%. The results of DSC measurements of the other examples and comparative examples are shown in Table 2.
TABLE 2
From the above results, it can be seen that the use of 3, 3' -iminobis (N, N-dimethylpropylamine) allows the mixed system of the present invention to be stored stably without crystal precipitation for 3 months, enabling the use of fatty acids at a higher concentration (15 wt%) than other amines.
In particular, it can be seen from comparative example 4 that the 3,3 '-iminobis (N, N-dimethylpropylamine) used in the present invention enables the molar ratio of long-chain fatty acid to amine in the miscible system to be up to 3:1, i.e., the amine is used in a smaller amount at the same concentration, relative to the use of N, N' -tetramethyldiamine (e.g., TMPDA) to solubilize long-chain fatty acids.
The preferred embodiments of the present invention have been described above in detail, but the present invention is not limited thereto. Within the scope of the technical idea of the invention, many simple modifications can be made to the technical solution of the invention, including combinations of various technical features in any other suitable way, and these simple modifications and combinations should also be regarded as the disclosure of the invention, and all fall within the scope of the invention.
Claims (16)
1. A mixed system containing a fatty acid type surfactant, characterized in that the mixed system contains a long-chain fatty acid having 11 to 16 carbon atoms, 3 '-iminobis (N, N-dimethylpropylamine) and water, wherein the molar ratio of the long-chain fatty acid to the 3, 3' -iminobis (N, N-dimethylpropylamine) is 45 to 305: 100.
2. The hybrid system according to claim 1, wherein the molar ratio between the long chain fatty acid and 3, 3' -iminobis (N, N-dimethylpropylamine) is between 50 and 300: 100.
3. The hybrid system according to claim 1, wherein the long chain fatty acid is present in an amount of 0.1 to 15 wt.%, based on the total weight of the hybrid system.
4. The mixed system according to any of claims 1-3, wherein the long chain fatty acid is a straight chain fatty acid.
5. The mixed system according to any of claims 1-3, wherein the long chain fatty acid is a saturated and/or unsaturated linear fatty acid.
6. The mixing system according to any one of claims 1-3, wherein the long chain fatty acid is selected from at least one of undecanoic acid, 10-undecenoic acid, dodecanoic acid, tridecanoic acid, tetradecanoic acid, pentadecanoic acid, and hexadecanoic acid.
7. A method for producing a mixed system containing a fatty acid type surfactant, which comprises mixing a long-chain fatty acid having 11 to 16 carbon atoms, 3 '-iminobis (N, N-dimethylpropylamine) and water until the solid is dissolved, wherein the molar ratio between the long-chain fatty acid and the 3, 3' -iminobis (N, N-dimethylpropylamine) is 45 to 305: 100.
8. The process of claim 7, wherein the molar ratio between the long chain fatty acid and 3, 3' -iminobis (N, N-dimethylpropylamine) is 50-300: 100.
9. The method of claim 7, wherein the long chain fatty acid is present in an amount of from 0.1 to 15 wt.%, based on the total weight of the mixed system.
10. The method of any one of claims 7-9, wherein the long chain fatty acid is a straight chain fatty acid.
11. The method of any one of claims 7-9, wherein the long chain fatty acid is a saturated and/or unsaturated linear fatty acid.
12. The method of any one of claims 7-9, wherein the long chain fatty acid is selected from at least one of undecanoic acid, 10-undecenoic acid, dodecanoic acid, tridecanoic acid, tetradecanoic acid, pentadecanoic acid, and hexadecanoic acid.
13. The method of claim 7, wherein the mixing is performed at a temperature of 60-80 ℃.
14. The method of claim 7, wherein the mixing is by: the long chain fatty acid is contacted with 3, 3' -iminobis (N, N-dimethylpropylamine), water is introduced and the mixture is kept at a constant temperature of 60 to 80 ℃ until the solid is dissolved.
15. The method of claim 7, wherein the mixing is by: firstly, long-chain fatty acid is contacted with 3, 3' -imino-bis (N, N-dimethylpropylamine), then water is introduced, the mixture is heated to 60-80 ℃ and is kept at the constant temperature for more than 10min, then the mixture is cooled to room temperature or 0 ℃ and is kept at the constant temperature for more than 10min, and the temperature is repeatedly increased and decreased for a plurality of times until the solid is dissolved.
16. A mixed system made by the method of any one of claims 7-15.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811446089.9A CN111229119B (en) | 2018-11-29 | 2018-11-29 | Mixed system containing fatty acid type surfactant and preparation method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811446089.9A CN111229119B (en) | 2018-11-29 | 2018-11-29 | Mixed system containing fatty acid type surfactant and preparation method thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111229119A CN111229119A (en) | 2020-06-05 |
| CN111229119B true CN111229119B (en) | 2021-08-03 |
Family
ID=70862018
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201811446089.9A Active CN111229119B (en) | 2018-11-29 | 2018-11-29 | Mixed system containing fatty acid type surfactant and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111229119B (en) |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2064835C (en) * | 1990-06-15 | 2001-10-02 | Yasunari Nakama | Novel complex and emulsified composition |
| CN103585925A (en) * | 2013-11-22 | 2014-02-19 | 江南大学 | Preparation method of double-end based amide type glycine betaine surfactant and application thereof |
| CN104338484A (en) * | 2013-08-02 | 2015-02-11 | 中国石油天然气股份有限公司 | A kind of surfactant and its preparation and application |
| CN104447380A (en) * | 2014-11-21 | 2015-03-25 | 广州天赐高新材料股份有限公司 | Preparation method of Bola type betaine surfactant |
| CN104437238A (en) * | 2014-12-11 | 2015-03-25 | 江南大学 | Amide-type bis-hydroxypropyl sulphobetaine surfactant as well as preparation method and application thereof |
| WO2015193909A1 (en) * | 2014-06-18 | 2015-12-23 | Ranka, Seema Ajay | Quaternary organosilane-ester/amide compounds and applications thereof. |
-
2018
- 2018-11-29 CN CN201811446089.9A patent/CN111229119B/en active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2064835C (en) * | 1990-06-15 | 2001-10-02 | Yasunari Nakama | Novel complex and emulsified composition |
| CN104338484A (en) * | 2013-08-02 | 2015-02-11 | 中国石油天然气股份有限公司 | A kind of surfactant and its preparation and application |
| CN103585925A (en) * | 2013-11-22 | 2014-02-19 | 江南大学 | Preparation method of double-end based amide type glycine betaine surfactant and application thereof |
| WO2015193909A1 (en) * | 2014-06-18 | 2015-12-23 | Ranka, Seema Ajay | Quaternary organosilane-ester/amide compounds and applications thereof. |
| CN104447380A (en) * | 2014-11-21 | 2015-03-25 | 广州天赐高新材料股份有限公司 | Preparation method of Bola type betaine surfactant |
| CN104437238A (en) * | 2014-12-11 | 2015-03-25 | 江南大学 | Amide-type bis-hydroxypropyl sulphobetaine surfactant as well as preparation method and application thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| CN111229119A (en) | 2020-06-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN107236530B (en) | A kind of water-base viscosity-reducing agent of emulsified superthick oil and preparation method thereof | |
| US20150376490A1 (en) | Non-Aqueous Drilling Additive Useful To Stabilize Viscosity Over Change In Temperature | |
| US4440665A (en) | Concentrated aqueous surfactant compositions | |
| CN104140803B (en) | Thick oil dispersion viscosity reducer and preparation method thereof | |
| US3520820A (en) | Cold water dispersible emulsions of filming amines | |
| EP0340054B1 (en) | Cationactive compositions; application to bituminous emulsions | |
| CN109111906A (en) | A kind of emulsifying and viscosity-reducing agent for condensed oil | |
| CN111229119B (en) | Mixed system containing fatty acid type surfactant and preparation method thereof | |
| CN111229117B (en) | Mixed system containing fatty acid type surfactant and preparation method thereof | |
| CN109985564B (en) | Mixed system containing fatty acid type surfactant and preparation method thereof | |
| CN111229118B (en) | Mixed system containing fatty acid type surfactant and preparation method thereof | |
| CN105482798A (en) | Compound pour point-depressing viscosity reducer for high pour-point crude oil and preparation method thereof | |
| CN111229120B (en) | Mixed system containing fatty acid type surfactant and preparation method thereof | |
| CN111394081A (en) | Self-demulsification type temperature-resistant viscosity reducer for cold recovery of thick oil and preparation method and application thereof | |
| CN115368978B (en) | Concentrated detergent with high-power dilution and thickening performance and preparation method thereof | |
| CN109985566B (en) | Mixed system containing fatty acid type surfactant and preparation method thereof | |
| Sakai et al. | Effects of spacer chain length of amino acid-based gemini surfactants on wormlike micelle formation | |
| GB2284215A (en) | Concentrated surfactant compositions | |
| GB2023637A (en) | Concentrated Aqueous Surfactant Compositions | |
| AU678312B2 (en) | Concentrated surfactant compositions | |
| CN109985565B (en) | Mixed system containing fatty acid type surfactant and preparation method thereof | |
| CN110997886B (en) | Surfactant system | |
| CN105331351B (en) | A kind of degradable water base clean fracturing fluid thickening agent in oil gas field | |
| CN111228126B (en) | Food-grade polyglycerol monolaurate dilutable microemulsion and preparation method thereof | |
| JPS604875B2 (en) | Concentrated aqueous surfactant composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |