CN111116582B - mGluR2 antagonist - Google Patents
mGluR2 antagonist Download PDFInfo
- Publication number
- CN111116582B CN111116582B CN201911310474.5A CN201911310474A CN111116582B CN 111116582 B CN111116582 B CN 111116582B CN 201911310474 A CN201911310474 A CN 201911310474A CN 111116582 B CN111116582 B CN 111116582B
- Authority
- CN
- China
- Prior art keywords
- triazolo
- cyclopropylmethyl
- trifluoromethyl
- present
- acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 102100036837 Metabotropic glutamate receptor 2 Human genes 0.000 title abstract description 13
- 108010038421 metabotropic glutamate receptor 2 Proteins 0.000 title abstract description 13
- 239000005557 antagonist Substances 0.000 title abstract description 5
- 150000001875 compounds Chemical class 0.000 claims abstract description 32
- 150000003839 salts Chemical class 0.000 claims abstract description 14
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 8
- 208000019901 Anxiety disease Diseases 0.000 claims abstract description 5
- 208000019022 Mood disease Diseases 0.000 claims abstract description 5
- 208000010877 cognitive disease Diseases 0.000 claims abstract description 5
- 108010010914 Metabotropic glutamate receptors Proteins 0.000 claims description 5
- 102000016193 Metabotropic glutamate receptors Human genes 0.000 claims description 5
- 239000003814 drug Substances 0.000 claims description 5
- 230000002265 prevention Effects 0.000 claims description 4
- YJLFIPVYOCXNNY-UHFFFAOYSA-N 5-[3-(cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl]-N,N-diethyl-1,2-oxazole-3-carboxamide Chemical compound CCN(CC)C(=O)C1=NOC(=C1)C2=C(C3=NN=C(N3C=C2)CC4CC4)C(F)(F)F YJLFIPVYOCXNNY-UHFFFAOYSA-N 0.000 claims description 2
- OOBVAJHGPSHEHY-UHFFFAOYSA-N 5-[3-(cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl]-N,N-dimethyl-1,2-oxazole-3-carboxamide Chemical compound CN(C)C(=O)C1=NOC(=C1)C2=C(C3=NN=C(N3C=C2)CC4CC4)C(F)(F)F OOBVAJHGPSHEHY-UHFFFAOYSA-N 0.000 claims description 2
- VQRNPUXLALBVCF-UHFFFAOYSA-N [5-[3-(cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl]-1,2-oxazol-3-yl]-piperidin-1-ylmethanone Chemical compound C1CCN(CC1)C(=O)C2=NOC(=C2)C3=C(C4=NN=C(N4C=C3)CC5CC5)C(F)(F)F VQRNPUXLALBVCF-UHFFFAOYSA-N 0.000 claims description 2
- LTHRHNPAGONLQR-UHFFFAOYSA-N [5-[3-(cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl]-1,2-oxazol-3-yl]-pyrrolidin-1-ylmethanone Chemical compound C1CCN(C1)C(=O)C2=NOC(=C2)C3=C(C4=NN=C(N4C=C3)CC5CC5)C(F)(F)F LTHRHNPAGONLQR-UHFFFAOYSA-N 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims 2
- DMVNOTPWCPBXEI-UHFFFAOYSA-N 5-[3-(cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl]-N-ethyl-1,2-oxazole-3-carboxamide Chemical compound CCNC(=O)C1=NOC(=C1)C2=C(C3=NN=C(N3C=C2)CC4CC4)C(F)(F)F DMVNOTPWCPBXEI-UHFFFAOYSA-N 0.000 claims 1
- AZIMCEXVIJGRDW-UHFFFAOYSA-N 5-[3-(cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl]-N-methyl-1,2-oxazole-3-carboxamide Chemical compound CNC(=O)C1=NOC(=C1)C2=C(C3=NN=C(N3C=C2)CC4CC4)C(F)(F)F AZIMCEXVIJGRDW-UHFFFAOYSA-N 0.000 claims 1
- 229940002612 prodrug Drugs 0.000 abstract description 8
- 239000000651 prodrug Substances 0.000 abstract description 8
- 239000012453 solvate Substances 0.000 abstract description 8
- 239000008194 pharmaceutical composition Substances 0.000 abstract description 7
- 201000010099 disease Diseases 0.000 abstract description 4
- AYSYSOQSKKDJJY-UHFFFAOYSA-N [1,2,4]triazolo[4,3-a]pyridine Chemical class C1=CC=CN2C=NN=C21 AYSYSOQSKKDJJY-UHFFFAOYSA-N 0.000 abstract description 3
- 230000003042 antagnostic effect Effects 0.000 abstract description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 24
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 15
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 15
- 125000000217 alkyl group Chemical group 0.000 description 15
- 239000000203 mixture Substances 0.000 description 9
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 8
- 238000002360 preparation method Methods 0.000 description 8
- 239000000243 solution Substances 0.000 description 7
- 239000002904 solvent Substances 0.000 description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical group [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 6
- 125000003545 alkoxy group Chemical group 0.000 description 6
- 125000004432 carbon atom Chemical group C* 0.000 description 6
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 5
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Natural products CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 5
- 239000002253 acid Substances 0.000 description 5
- 238000006243 chemical reaction Methods 0.000 description 5
- 229930195712 glutamate Natural products 0.000 description 5
- 238000000034 method Methods 0.000 description 5
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 4
- 239000001257 hydrogen Substances 0.000 description 4
- 229910052739 hydrogen Inorganic materials 0.000 description 4
- 125000004435 hydrogen atom Chemical class [H]* 0.000 description 4
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 4
- 125000004433 nitrogen atom Chemical group N* 0.000 description 4
- 239000003208 petroleum Substances 0.000 description 4
- 239000000741 silica gel Substances 0.000 description 4
- 229910002027 silica gel Inorganic materials 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- OFOBLEOULBTSOW-UHFFFAOYSA-N Malonic acid Chemical compound OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- 150000007513 acids Chemical class 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 3
- 230000005764 inhibitory process Effects 0.000 description 3
- -1 methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, isobutoxy, tert-butoxy, pentoxy Chemical group 0.000 description 3
- 239000012074 organic phase Substances 0.000 description 3
- 239000002245 particle Substances 0.000 description 3
- 230000009870 specific binding Effects 0.000 description 3
- NUPPUNVPCLNIKS-UHFFFAOYSA-N 2-[3-(cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl]ethynyl-trimethylsilane Chemical compound C[Si](C)(C)C#CC1=C(C2=NN=C(N2C=C1)CC3CC3)C(F)(F)F NUPPUNVPCLNIKS-UHFFFAOYSA-N 0.000 description 2
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 2
- QDXKFLHIFXNKGW-UHFFFAOYSA-N 3-(cyclopropylmethyl)-7-ethynyl-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridine Chemical compound C#CC1=C(C2=NN=C(N2C=C1)CC3CC3)C(F)(F)F QDXKFLHIFXNKGW-UHFFFAOYSA-N 0.000 description 2
- MYFWXXPPPYPLOB-UHFFFAOYSA-N 7-chloro-3-(cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridine Chemical compound N=1N=C2C(C(F)(F)F)=C(Cl)C=CN2C=1CC1CC1 MYFWXXPPPYPLOB-UHFFFAOYSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 2
- ROSDSFDQCJNGOL-UHFFFAOYSA-N Dimethylamine Chemical compound CNC ROSDSFDQCJNGOL-UHFFFAOYSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- AEMRFAOFKBGASW-UHFFFAOYSA-N Glycolic acid Chemical compound OCC(O)=O AEMRFAOFKBGASW-UHFFFAOYSA-N 0.000 description 2
- 239000007995 HEPES buffer Substances 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- LCTONWCANYUPML-UHFFFAOYSA-N Pyruvic acid Chemical compound CC(=O)C(O)=O LCTONWCANYUPML-UHFFFAOYSA-N 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- 239000002585 base Substances 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 2
- 238000005119 centrifugation Methods 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 239000000460 chlorine Substances 0.000 description 2
- 125000000753 cycloalkyl group Chemical group 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 239000003480 eluent Substances 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- QGSZBXNCEKAJIH-DJWKRKHSSA-N methyl (2z)-2-chloro-2-hydroxyiminoacetate Chemical compound COC(=O)C(\Cl)=N\O QGSZBXNCEKAJIH-DJWKRKHSSA-N 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 238000000159 protein binding assay Methods 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- 125000006413 ring segment Chemical group 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 2
- 229910000029 sodium carbonate Inorganic materials 0.000 description 2
- 235000017550 sodium carbonate Nutrition 0.000 description 2
- DAEPDZWVDSPTHF-UHFFFAOYSA-M sodium pyruvate Chemical compound [Na+].CC(=O)C([O-])=O DAEPDZWVDSPTHF-UHFFFAOYSA-M 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 2
- XOFLBQFBSOEHOG-UUOKFMHZSA-N γS-GTP Chemical compound C1=2NC(N)=NC(=O)C=2N=CN1[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=S)[C@@H](O)[C@H]1O XOFLBQFBSOEHOG-UUOKFMHZSA-N 0.000 description 2
- 125000006583 (C1-C3) haloalkyl group Chemical group 0.000 description 1
- 125000004765 (C1-C4) haloalkyl group Chemical group 0.000 description 1
- 125000000171 (C1-C6) haloalkyl group Chemical group 0.000 description 1
- 125000006621 (C3-C8) cycloalkyl-(C1-C6) alkyl group Chemical group 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 1
- WUBBRNOQWQTFEX-UHFFFAOYSA-N 4-aminosalicylic acid Chemical compound NC1=CC=C(C(O)=O)C(O)=C1 WUBBRNOQWQTFEX-UHFFFAOYSA-N 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- 208000028698 Cognitive impairment Diseases 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 108010027915 Glutamate Receptors Proteins 0.000 description 1
- 102000018899 Glutamate Receptors Human genes 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 1
- GRRNUXAQVGOGFE-UHFFFAOYSA-N Hygromycin-B Natural products OC1C(NC)CC(N)C(O)C1OC1C2OC3(C(C(O)C(O)C(C(N)CO)O3)O)OC2C(O)C(CO)O1 GRRNUXAQVGOGFE-UHFFFAOYSA-N 0.000 description 1
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 1
- 229930182816 L-glutamine Natural products 0.000 description 1
- 102000018697 Membrane Proteins Human genes 0.000 description 1
- 108010052285 Membrane Proteins Proteins 0.000 description 1
- 101100030361 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) pph-3 gene Proteins 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 229930182555 Penicillin Natural products 0.000 description 1
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 1
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 150000001350 alkyl halides Chemical class 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 229960004909 aminosalicylic acid Drugs 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 230000000049 anti-anxiety effect Effects 0.000 description 1
- 230000001430 anti-depressive effect Effects 0.000 description 1
- 239000000935 antidepressant agent Substances 0.000 description 1
- 229940005513 antidepressants Drugs 0.000 description 1
- 239000002249 anxiolytic agent Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 125000004069 aziridinyl group Chemical group 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-N benzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-N 0.000 description 1
- 229940092714 benzenesulfonic acid Drugs 0.000 description 1
- 230000027455 binding Effects 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 210000004978 chinese hamster ovary cell Anatomy 0.000 description 1
- 235000015165 citric acid Nutrition 0.000 description 1
- 230000003920 cognitive function Effects 0.000 description 1
- 210000004748 cultured cell Anatomy 0.000 description 1
- 239000000625 cyclamic acid and its Na and Ca salt Substances 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- HCAJEUSONLESMK-UHFFFAOYSA-N cyclohexylsulfamic acid Chemical compound OS(=O)(=O)NC1CCCCC1 HCAJEUSONLESMK-UHFFFAOYSA-N 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 1
- 125000004494 ethyl ester group Chemical group 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 230000002964 excitative effect Effects 0.000 description 1
- 239000012091 fetal bovine serum Substances 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003707 hexyloxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 1
- 125000001183 hydrocarbyl group Chemical group 0.000 description 1
- GRRNUXAQVGOGFE-NZSRVPFOSA-N hygromycin B Chemical compound O[C@@H]1[C@@H](NC)C[C@@H](N)[C@H](O)[C@H]1O[C@H]1[C@H]2O[C@@]3([C@@H]([C@@H](O)[C@@H](O)[C@@H](C(N)CO)O3)O)O[C@H]2[C@@H](O)[C@@H](CO)O1 GRRNUXAQVGOGFE-NZSRVPFOSA-N 0.000 description 1
- 229940097277 hygromycin b Drugs 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- 150000004702 methyl esters Chemical class 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- IVZUXVKZLNNSLW-UHFFFAOYSA-N n,n-dimethyl-1,2-oxazole-3-carboxamide Chemical compound CN(C)C(=O)C=1C=CON=1 IVZUXVKZLNNSLW-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000002858 neurotransmitter agent Substances 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 230000009871 nonspecific binding Effects 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- UQPUONNXJVWHRM-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 UQPUONNXJVWHRM-UHFFFAOYSA-N 0.000 description 1
- WLJNZVDCPSBLRP-UHFFFAOYSA-N pamoic acid Chemical compound C1=CC=C2C(CC=3C4=CC=CC=C4C=C(C=3O)C(=O)O)=C(O)C(C(O)=O)=CC2=C1 WLJNZVDCPSBLRP-UHFFFAOYSA-N 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- 229940049954 penicillin Drugs 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- 235000015320 potassium carbonate Nutrition 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- 229940107700 pyruvic acid Drugs 0.000 description 1
- 239000001397 quillaja saponaria molina bark Substances 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 229960004889 salicylic acid Drugs 0.000 description 1
- 229930182490 saponin Natural products 0.000 description 1
- 150000007949 saponins Chemical class 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 229930195734 saturated hydrocarbon Natural products 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000012064 sodium phosphate buffer Substances 0.000 description 1
- 229940054269 sodium pyruvate Drugs 0.000 description 1
- ZDQYSKICYIVCPN-UHFFFAOYSA-L sodium succinate (anhydrous) Chemical compound [Na+].[Na+].[O-]C(=O)CCC([O-])=O ZDQYSKICYIVCPN-UHFFFAOYSA-L 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 230000000946 synaptic effect Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000003944 tolyl group Chemical group 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- CWMFRHBXRUITQE-UHFFFAOYSA-N trimethylsilylacetylene Chemical group C[Si](C)(C)C#C CWMFRHBXRUITQE-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/22—Anxiolytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Chemical & Material Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Psychiatry (AREA)
- Hospice & Palliative Care (AREA)
- Pain & Pain Management (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
本发明涉及一种作为mGluR2拮抗剂的[1,2,4]三唑并[4,3‑a]吡啶衍生物或其药学上可接受的盐、溶剂合物、前药,以及包括它们的药物组合物和用途。本发明化合物对mGluR2具有良好的拮抗作用,可以用于治疗mGluR2相关的疾病,如情绪障碍、焦虑症、认知障碍等。The present invention relates to a [1,2,4]triazolo[4,3-a]pyridine derivative or a pharmaceutically acceptable salt, solvate, prodrug thereof as a mGluR2 antagonist, and a compound comprising them Pharmaceutical compositions and uses. The compounds of the present invention have a good antagonistic effect on mGluR2, and can be used for the treatment of mGluR2-related diseases, such as mood disorders, anxiety disorders, cognitive disorders and the like.
Description
技术领域technical field
本发明涉及对mGluR2具有拮抗作用的[1,2,4]三唑并[4,3-a]吡啶衍生物或其药学上可接受的盐,以及包括该化合物作为活性成分的药用组合物。The present invention relates to a [1,2,4]triazolo[4,3-a]pyridine derivative or a pharmaceutically acceptable salt thereof having an antagonistic effect on mGluR2, and a pharmaceutical composition comprising the compound as an active ingredient .
背景技术Background technique
谷氨酸已知是作为用于在哺乳动物的中枢神经系统中调节记忆、学习以及等高级功能而工作的主要刺激性神经递质。其中,谷氨酸激活代谢型谷氨酸受体(mGluR)具有更高的有助于突触效能的微调的调节作用。在这些谷氨酸受体中,针对mGluR2的拮抗剂显示出改进认知功能的作用,并且还显示了诸如抗抑郁作用和抗焦虑作用,可以用于治疗诸如情绪障碍、焦虑症、认知障碍等疾病。Glutamate is known to work as the primary excitatory neurotransmitter for regulating higher functions such as memory, learning, and the like in the mammalian central nervous system. Among them, glutamate-activated metabotropic glutamate receptors (mGluRs) have a higher regulatory effect that contributes to fine-tuning of synaptic efficacy. Among these glutamate receptors, antagonists against mGluR2 have been shown to improve cognitive function, and have also shown effects such as antidepressant and anti-anxiety, and can be used to treat disorders such as mood disorders, anxiety disorders, cognitive disorders and other diseases.
因此,仍然存在对研发新型的作为mGluR2拮抗剂的需求。Therefore, there remains a need to develop novel mGluR2 antagonists.
发明内容SUMMARY OF THE INVENTION
本发明旨在提供一种作为mGluR2拮抗剂的[1,2,4]三唑并[4,3-a]吡啶衍生物或其药学上可接受的盐、溶剂合物、前药,以及包括它们的药物组合物和用途。The present invention aims to provide a [1,2,4]triazolo[4,3-a]pyridine derivative or a pharmaceutically acceptable salt, solvate, prodrug thereof as an mGluR2 antagonist, and a compound including Their pharmaceutical compositions and uses.
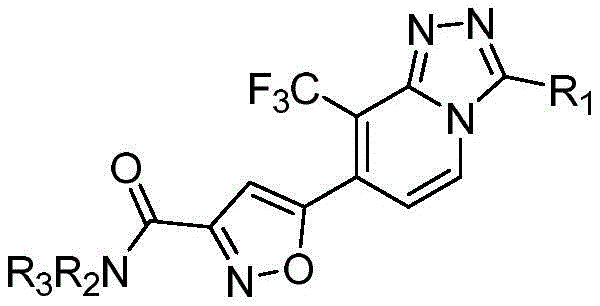
本发明一方面提供一种式I所示化合物或其药学上可接受的盐、溶剂合物、前药:One aspect of the present invention provides a compound of formula I or a pharmaceutically acceptable salt, solvate and prodrug thereof:
其中,R1选自C3-8环烷基-C1-6烷基-,C1-6烷氧基C1-6烷基-,C1-6烷基-,C1-6卤代烷基-;Wherein, R 1 is selected from C 3-8 cycloalkyl-C 1-6 alkyl-, C 1-6 alkoxy C 1-6 alkyl-, C 1-6 alkyl-, C 1-6 haloalkane base-;
R2和R3独立地选自氢、C1-6烷基,C1-6卤代烷基-,或R2和R3与结合的N原子一起组成3-8元含氮杂环。R 2 and R 3 are independently selected from hydrogen, C 1-6 alkyl, C 1-6 haloalkyl-, or R 2 and R 3 together with the bound N atom form a 3-8 membered nitrogen-containing heterocycle.
在一些实施例中,R1选自C3-8环烷基-C1-4烷基-,C1-4烷氧基C1-4烷基-,C1-4烷基-,C1-4卤代烷基-;In some embodiments, R 1 is selected from C 3-8 cycloalkyl-C 1-4 alkyl-, C 1-4 alkoxy C 1-4 alkyl-, C 1-4 alkyl-, C 1-4 haloalkyl-;
R2和R3独立地选自氢、C1-4烷基,C1-4卤代烷基-,或R2和R3与结合的N原子一起组成3-6元含氮杂环。R 2 and R 3 are independently selected from hydrogen, C 1-4 alkyl, C 1-4 haloalkyl-, or R 2 and R 3 together with the bound N atom form a 3-6 membered nitrogen-containing heterocycle.
在一些实施例中,R1选自C3-6环烷基-C1-2烷基-,C1-2烷氧基C1-2烷基-,C1-2烷基-,C1-2卤代烷基-;In some embodiments, R 1 is selected from C 3-6 cycloalkyl-C 1-2 alkyl-, C 1-2 alkoxy C 1-2 alkyl-, C 1-2 alkyl-, C 1-2 haloalkyl-;
R2和R3独立地选自氢、C1-3烷基,C1-3卤代烷基-,或R2和R3与结合的N原子一起组成4-6元含氮杂环。R 2 and R 3 are independently selected from hydrogen, C 1-3 alkyl, C 1-3 haloalkyl-, or R 2 and R 3 together with the bound N atom form a 4-6 membered nitrogen-containing heterocycle.
在一些实施例中,R1选自C3-4环烷基-C1-2烷基-,C1-2烷氧基C1-2烷基-;In some embodiments, R 1 is selected from C 3-4 cycloalkyl-C 1-2 alkyl-, C 1-2 alkoxy C 1-2 alkyl-;
R2和R3独立地选自氢、C1-3烷基,或R2和R3与结合的N原子一起组成4-6元含氮杂环。R 2 and R 3 are independently selected from hydrogen, C 1-3 alkyl, or R 2 and R 3 together with the bound N atom form a 4-6 membered nitrogen-containing heterocycle.
特别地,本发明提供以下具体化合物:In particular, the present invention provides the following specific compounds:
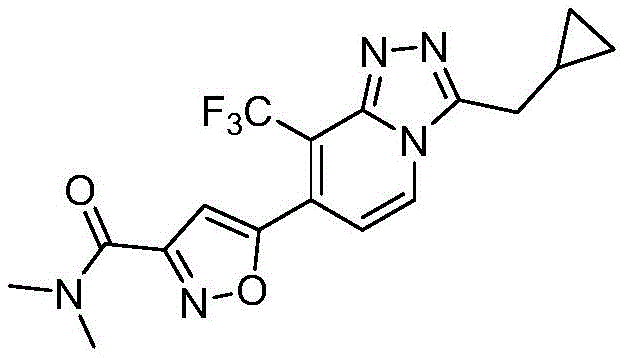
5-(3-(环丙基甲基)-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶-7-基)-N,N-二甲基异噁唑-3-甲酰胺5-(3-(Cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl)-N,N- Dimethylisoxazole-3-carboxamide
5-(3-(环丙基甲基)-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶-7-基)-N,N-二乙基异噁唑-3-甲酰胺5-(3-(Cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl)-N,N- Diethylisoxazole-3-carboxamide
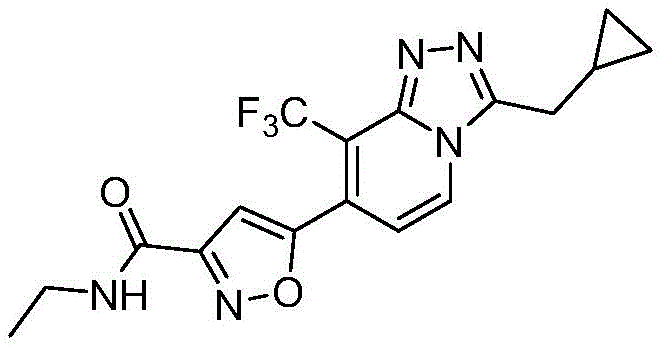
5-(3-(环丙基甲基)-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶-7-基)-N-乙基异噁唑-3-甲酰胺5-(3-(Cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl)-N-ethyl Isoxazole-3-carboxamide
5-(3-(环丙基甲基)-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶-7-基)-N-甲基异噁唑-3-甲酰胺5-(3-(Cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl)-N-methyl Isoxazole-3-carboxamide
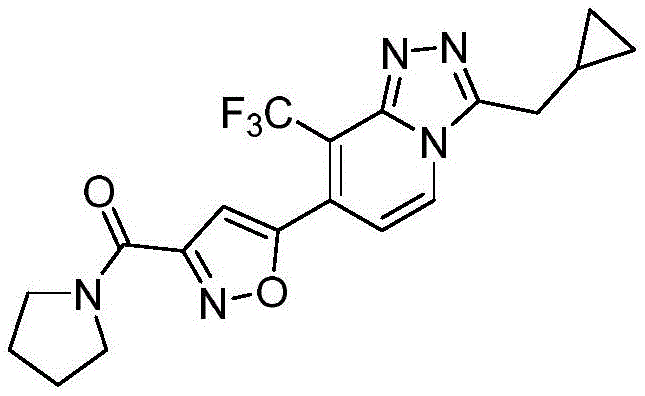
(5-(3-(环丙基甲基)-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶-7-基)异噁唑-3-基)(吡咯烷-1-基)甲酮(5-(3-(Cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl)isoxazole- 3-yl)(pyrrolidin-1-yl)methanone
(5-(3-(环丙基甲基)-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶-7-基)异噁唑-3-基)(哌啶-1-基)甲酮(5-(3-(Cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl)isoxazole- 3-yl)(piperidin-1-yl)methanone
进一步地,本发明还涉及一种药物组合物,其包括治疗有效量的具有化学式(I)的化合物或其药学上可接受的盐、溶剂合物、前药以及药学上可接受的辅料。Further, the present invention also relates to a pharmaceutical composition comprising a therapeutically effective amount of the compound of formula (I) or a pharmaceutically acceptable salt, solvate, prodrug and pharmaceutically acceptable excipients thereof.
本发明还涉及一种具有化学式(I)的化合物或其药学上可接受的盐、溶剂合物、前药,作为用于治疗或预防与mGluR2相关的疾病(如情绪障碍、焦虑症、认知障碍等)的药物。The present invention also relates to a compound of formula (I) or a pharmaceutically acceptable salt, solvate, prodrug thereof, for use in the treatment or prevention of mGluR2-related diseases (such as mood disorders, anxiety disorders, cognitive disorders, etc.) drugs.
定义definition
除非另行说明,本发明“烷基”定义了含有1-6个碳原子(优选1-4,1-3或1-2个碳原子)的直链或支链饱和烃基。烷基的实例包括甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、戊基或己基等。Unless otherwise specified, "alkyl" herein defines a straight or branched chain saturated hydrocarbon group containing 1-6 carbon atoms, preferably 1-4, 1-3 or 1-2 carbon atoms. Examples of alkyl groups include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, pentyl or hexyl, and the like.
“烷氧”定义了含有1-6个碳原子(优选1-4,1-3或1-2个碳原子)的直链或支链饱和烃基氧基。烷氧的实例包括甲氧基、乙氧基、正丙氧基、异丙氧基、正丁氧基、异丁氧基、叔丁氧基、戊氧基或己氧基等"Alkoxy" defines a straight or branched chain saturated hydrocarbyloxy group containing 1-6 carbon atoms, preferably 1-4, 1-3 or 1-2 carbon atoms. Examples of alkoxy groups include methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, isobutoxy, tert-butoxy, pentoxy, or hexyloxy, and the like
“环烷基”定义了含有3-8个碳原子(优选3-6,或3-4个碳原子)的饱和环状烃基,如环丙基、环丁基、环戊基、环己基、环庚基和环辛基。"Cycloalkyl" defines a saturated cyclic hydrocarbon group containing 3-8 carbon atoms (preferably 3-6, or 3-4 carbon atoms), such as cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl and cyclooctyl.
“含氮杂环”定义了含有3-8个环原子(优选3-6,或4-6个环原子)的,且其中一个原子为氮的饱和含氮杂环,如吖丙啶基、吡咯烷基、哌啶基。"Nitrogen-containing heterocycle" defines a saturated nitrogen-containing heterocycle containing 3-8 ring atoms (preferably 3-6, or 4-6 ring atoms), one of which is nitrogen, such as aziridinyl, Pyrrolidinyl, piperidinyl.
“卤素”表示氟、氯、溴或碘,其中优选的是氟或氯。"Halogen" means fluorine, chlorine, bromine or iodine, with fluorine or chlorine being preferred.
在本发明中,本发明化合物的药学上可接受的盐包括其游离碱性化合物与常规酸形成的酸加成盐,所述酸例如是:无机酸,例如氢卤酸(例如盐酸或氢溴酸)、硫酸、硝酸、磷酸等;或有机酸,例如乙酸、丙酸、羟基乙酸、乳酸、丙酮酸、草酸、丙二酸、琥珀酸、马来酸、富马酸、苹果酸、酒石酸、柠檬酸、甲磺酸、乙磺酸、苯磺酸、对甲苯磺酸、环己氨基磺酸、水杨酸、对氨基水杨酸、双羟萘酸以及类似酸。In the present invention, the pharmaceutically acceptable salts of the compounds of the present invention include acid addition salts of the free base compounds thereof with conventional acids such as: inorganic acids such as hydrohalic acids (such as hydrochloric acid or hydrobromide) acid), sulfuric acid, nitric acid, phosphoric acid, etc.; or organic acids such as acetic acid, propionic acid, glycolic acid, lactic acid, pyruvic acid, oxalic acid, malonic acid, succinic acid, maleic acid, fumaric acid, malic acid, tartaric acid, Citric acid, methanesulfonic acid, ethanesulfonic acid, benzenesulfonic acid, p-toluenesulfonic acid, cyclamic acid, salicylic acid, p-aminosalicylic acid, pamoic acid, and similar acids.
在本发明中,本发明的溶剂合物是指一个或多个溶剂分子与本发明的化合物所形成的缔合物。形成溶剂化物的溶剂包括,但并不限于,水、甲醇、乙醇、异丙醇、二甲亚砜、乙酸乙酯、乙酸、氨基乙醇。In the present invention, the solvate of the present invention refers to an association formed by one or more solvent molecules and the compound of the present invention. Solvate-forming solvents include, but are not limited to, water, methanol, ethanol, isopropanol, dimethyl sulfoxide, ethyl acetate, acetic acid, aminoethanol.
在本发明中,对本发明化合物的前药没有具体限制,只要其在生物体内能够代谢成本发明化合物即可,非限制性地包括酯等,例如甲酯、乙酯等。In the present invention, the prodrugs of the compounds of the present invention are not particularly limited as long as they can be metabolized into the compounds of the present invention in vivo, and include esters and the like, such as methyl esters, ethyl esters, and the like without limitation.
制备方法Preparation
本发明另一方面提供一种制备式I所示化合物的方法,其包括以下步骤:Another aspect of the present invention provides a method for preparing the compound shown in formula I, which comprises the following steps:
其中,上述式中R1,R2和R3定义如前文所述;Wherein, in the above formula, R 1 , R 2 and R 3 are defined as described above;
步骤(I):7-氯-3-(环丙基甲基)-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶与TMS保护的乙炔在Sonogashira条件下反应,优选Pd(PPh3)2Cl2,CuI,Et3N,溶剂为甲苯。Step (I): 7-Chloro-3-(cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridine with TMS protected Acetylene is reacted under Sonogashira conditions, preferably Pd(PPh3)2Cl2, CuI, Et3N, and the solvent is toluene.
步骤(II):碱性条件下脱除乙炔基保护基,所述碱为NaOH、KOH;溶剂为甲醇。Step (II): removing the ethynyl protecting group under alkaline conditions, the base is NaOH, KOH; the solvent is methanol.
步骤(III):碱性条件下,2-氯-2-(羟基亚氨基)乙酸甲酯与乙炔基反应生成异噁唑环,所述碱为Na2CO3、K2CO3,溶剂为乙醇。Step (III): under alkaline conditions, methyl 2-chloro-2-(hydroxyimino)acetate reacts with an ethynyl group to generate an isoxazole ring, the bases are Na2CO3 , K2CO3, and the solvent is ethanol.
步骤(IV):制备的异噁唑化合物与R3R2NH反应制备酰胺化合物,其中溶剂为甲醇、乙醇或THF。Step (IV): the prepared isoxazole compound is reacted with R 3 R 2 NH to prepare an amide compound, wherein the solvent is methanol, ethanol or THF.
药物组合物pharmaceutical composition
本发明另一方面提供了一种药物组合物,其包含至少一种式I所示化合物或其药学上可接受的盐、溶剂合物、前药以及一种或更多种药学上可接受的辅剂。药物组合物可被配制成液体、粉末、可注射溶液、悬浮剂、栓剂等。并且考虑所有的给药方式,例如口服、直肠、肠胃外、局部、或通过静脉内、肌内、胸骨内或皮下注射、或以适于吸入的形式。Another aspect of the present invention provides a pharmaceutical composition comprising at least one compound represented by formula I or a pharmaceutically acceptable salt, solvate, prodrug thereof and one or more pharmaceutically acceptable compounds adjuvant. Pharmaceutical compositions can be formulated as liquids, powders, injectable solutions, suspensions, suppositories and the like. And all modes of administration are contemplated, such as oral, rectal, parenteral, topical, or by intravenous, intramuscular, intrasternal or subcutaneous injection, or in a form suitable for inhalation.
应用application
本发明另一方面提供了式I所示化合物或其药学上可接受的盐、溶剂合物、前药在制备药物中的应用,所述药物用于治疗或预防与mGluR2相关的疾病(如情绪障碍、焦虑症、认知障碍等)。Another aspect of the present invention provides the use of a compound represented by formula I or a pharmaceutically acceptable salt, solvate, or prodrug thereof in the preparation of a medicament for the treatment or prevention of mGluR2-related diseases (such as mood disorders, anxiety disorders, cognitive impairment, etc.).
具体实施方式Detailed ways
在下文中更详细地描述了本发明以有助于对本发明的理解。The present invention is described in more detail below to facilitate understanding of the present invention.
所属领域的技术人员将认识到:本发明所描述的化学反应可以用来合适地制备许多本发明的其他化合物,且用于制备本发明的化合物的其它方法都被认为是在本发明的范围之内。例如,根据本发明那些非例证的化合物的合成可以成功地被所属领域的技术人员通过修饰方法完成,如适当的保护干扰基团,通过利用其他已知的试剂除了本发明所描述的,或将反应条件做一些常规的修改。另外,本发明所公开的反应或已知的反应条件也公认地适用于本发明其他化合物的制备。Those skilled in the art will recognize that the chemical reactions described herein can be used to suitably prepare many other compounds of the present invention, and that other methods for preparing the compounds of the present invention are considered to be within the scope of the present invention Inside. For example, the synthesis of those non-exemplified compounds according to the present invention can be successfully accomplished by those skilled in the art by modifying methods, such as appropriate protection of interfering groups, by using other known reagents in addition to those described herein, or by combining The reaction conditions were modified routinely. In addition, the reactions disclosed herein or known reaction conditions are also acknowledged to be applicable to the preparation of other compounds of the present invention.
实施例1:5-(3-(环丙基甲基)-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶-7-基)-N,N-二甲基异噁唑-3-甲酰胺制备Example 1: 5-(3-(Cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl)- Preparation of N,N-dimethylisoxazole-3-carboxamide
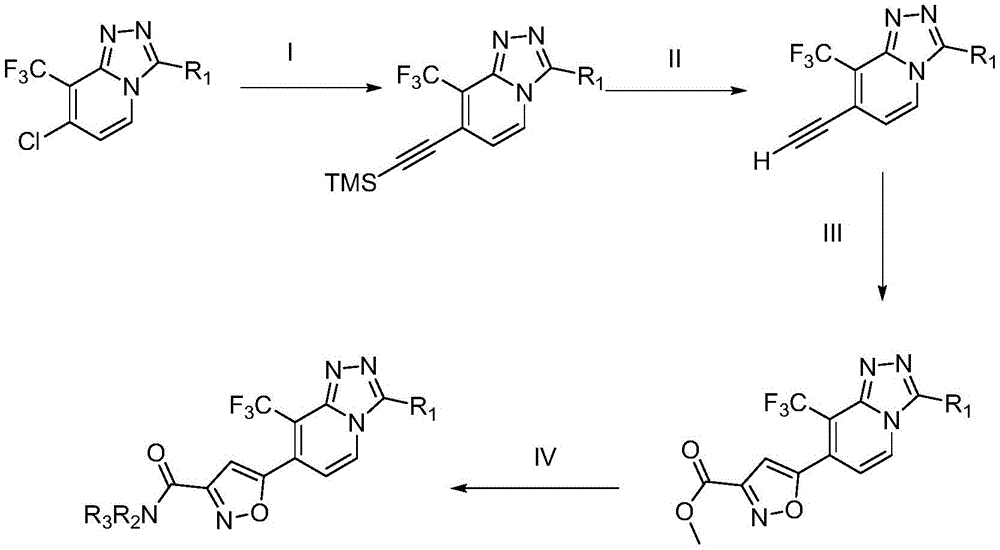
(I)3-(环丙基甲基)-8-(三氟甲基)-7-((三甲基硅基)乙炔基)-[1,2,4]三唑并[4,3-a]吡啶制备(I) 3-(Cyclopropylmethyl)-8-(trifluoromethyl)-7-((trimethylsilyl)ethynyl)-[1,2,4]triazolo[4,3 -a]Pyridine preparation
与N2保护下,向7-氯-3-(环丙基甲基)-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶(11g,40mmol)甲苯(150mL)溶液中加入Pd(PPh3)2Cl2(1.4g,2mmol),CuI(0.38g,2mmol)和Et3N(30mL)。与80℃下,滴入三甲基硅基乙炔(11.2mL,80mmol),并将混合物搅拌6h。过滤所得混合物,滤液中加入250mL乙酸乙酯,使用水洗涤上述溶液,有机相干燥,浓缩。所得残余物经硅胶柱纯化,洗脱液为25%乙酸乙酯/石油醚,得到浅褐色固体(11.59g,86%),LC-MS(ESI)m/z:338.1226(M+H)+。Under the protection of N2, 7-chloro-3-(cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridine (11 g, 40 mmol) in toluene (150 mL) was added Pd( PPh3 )2Cl2 (1.4 g, 2 mmol), CuI (0.38 g, 2 mmol) and Et3N (30 mL). At 80°C, trimethylsilylacetylene (11.2 mL, 80 mmol) was added dropwise, and the mixture was stirred for 6 h. The resulting mixture was filtered, 250 mL of ethyl acetate was added to the filtrate, the above solution was washed with water, the organic phase was dried, and concentrated. The resulting residue was purified by silica gel column eluting with 25% ethyl acetate/petroleum ether to give a light brown solid (11.59 g, 86%), LC-MS (ESI) m/z: 338.1226 (M+H) + .
(II)3-(环丙基甲基)-7-乙炔基-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶制备(II) Preparation of 3-(cyclopropylmethyl)-7-ethynyl-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridine
将3-(环丙基甲基)-8-(三氟甲基)-7-((三甲基硅基)乙炔基)-[1,2,4]三唑并[4,3-a]吡啶(10.11g,30mmol)溶于80mL甲醇中。在室温,于搅拌条件下,加入NaOH(0.12g,3mmol)的水溶液(15mL),混合物搅拌4h。将混合物于减压下浓缩,并向残留物中加入乙酸乙酯(250mL)和水(40mL),分液,有机相经干燥,浓缩,所得残余物经硅胶柱纯化,洗脱液为30%乙酸乙酯/石油醚,得到黄色固体(6.2g,78%)。3-(Cyclopropylmethyl)-8-(trifluoromethyl)-7-((trimethylsilyl)ethynyl)-[1,2,4]triazolo[4,3-a ] Pyridine (10.11 g, 30 mmol) was dissolved in 80 mL of methanol. At room temperature, with stirring, an aqueous solution (15 mL) of NaOH (0.12 g, 3 mmol) was added, and the mixture was stirred for 4 h. The mixture was concentrated under reduced pressure, and to the residue were added ethyl acetate (250 mL) and water (40 mL), the layers were separated, the organic phase was dried and concentrated, and the resulting residue was purified by silica gel column with 30% eluent Ethyl acetate/petroleum ether gave a yellow solid (6.2 g, 78%).
1H NMR(400MHz,CDCl3)δppm 0.21-0.38(m,2H),0.44-0.60(m,2H),1.02-1.15(m,1H),3.01(d,2H),3.28(s,1H),7.35(d,1H),8.22(d,1H);LC-MS(ESI)m/z:266.0831(M+H)+。 1 H NMR (400 MHz, CDCl 3 ) δppm 0.21-0.38 (m, 2H), 0.44-0.60 (m, 2H), 1.02-1.15 (m, 1H), 3.01 (d, 2H), 3.28 (s, 1H) , 7.35 (d, 1H), 8.22 (d, 1H); LC-MS (ESI) m/z: 266.0831 (M+H) + .
(III)5-(3-(环丙基甲基)-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶-7-基)异噁唑-3-羧基甲酯制备:(III) 5-(3-(Cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl)isoxoxane Preparation of oxazole-3-carboxymethyl ester:
将3-(环丙基甲基)-7-乙炔基-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶(5.83g,22mmol)和碳酸钠(14.0g,132mmol)溶于乙醇(100mL)溶液中。在室温,于搅拌条件下,将2-氯-2-(羟基亚氨基)乙酸甲酯(15.1g,110mmol)的乙醇(50mL)溶液滴入上述溶液中。所得反应混合物室温搅拌24h。将混合物于减压下浓缩,并向残留物中加入乙酸乙酯(400mL)和水(100mL),分液,有机相经干燥,浓缩,所得残余物经硅胶柱纯化,洗脱液为30%乙酸乙酯/石油醚,得到白色固体(4.73g,58.7%),LC-MS(ESI)m/z:366.0938(M+H)+。3-(Cyclopropylmethyl)-7-ethynyl-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridine (5.83 g, 22 mmol) and Sodium carbonate (14.0 g, 132 mmol) was dissolved in a solution of ethanol (100 mL). A solution of methyl 2-chloro-2-(hydroxyimino)acetate (15.1 g, 110 mmol) in ethanol (50 mL) was added dropwise to the above solution at room temperature with stirring. The resulting reaction mixture was stirred at room temperature for 24 h. The mixture was concentrated under reduced pressure, and ethyl acetate (400 mL) and water (100 mL) were added to the residue, the layers were separated, the organic phase was dried and concentrated, and the obtained residue was purified by silica gel column with 30% eluent Ethyl acetate/petroleum ether gave a white solid (4.73 g, 58.7%), LC-MS (ESI) m/z: 366.0938 (M+H) + .
(IV)5-(3-(环丙基甲基)-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶-7-基)-N,N-二甲基异噁唑-3-甲酰胺制备(IV) 5-(3-(Cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4,3-a]pyridin-7-yl)-N , Preparation of N-dimethylisoxazole-3-carboxamide
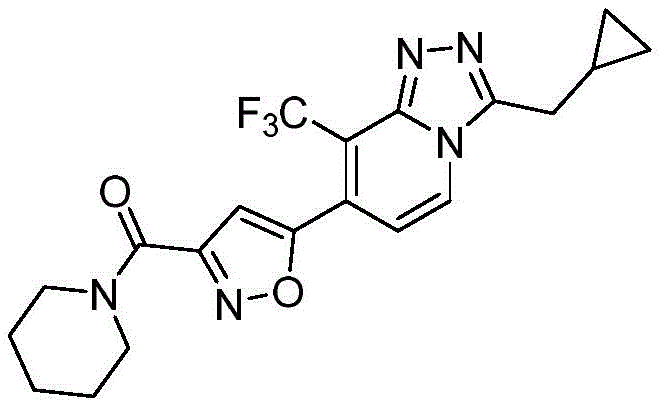
将40%二甲胺水溶液(18.5mL,150mmol)滴加入到5-(3-(环丙基甲基)-8-(三氟甲基)-[1,2,4]三唑并[4,3-a]吡啶-7-基)异噁唑-3-羧基甲酯(3.66g,10mmol)的甲醇(20mL)溶液中,反应混合物在60℃搅拌4h。与减压下除去溶剂,并将残留物使用硅胶柱纯化,洗脱液为45%乙酸乙酯/石油醚,获得白色固体(3.45g,91%)。A 40% aqueous dimethylamine solution (18.5 mL, 150 mmol) was added dropwise to 5-(3-(cyclopropylmethyl)-8-(trifluoromethyl)-[1,2,4]triazolo[4] ,3-a]pyridin-7-yl)isoxazole-3-carboxymethyl ester (3.66 g, 10 mmol) in methanol (20 mL), the reaction mixture was stirred at 60 °C for 4 h. The solvent was removed under reduced pressure and the residue was purified using a silica gel column eluting with 45% ethyl acetate/petroleum ether to give a white solid (3.45 g, 91%).
1H NMR(400MHz,CDCl3)δppm 0.23-0.40(m,2H),0.47-0.62(m,2H),1.07-1.19(m,1H),2.89(s,3H),2.97(s,3H),3.03(d,2H),7.28(s,1H),7.60(d,1H),8.46(d,1H);LC-MS(ESI)m/z:380.1259(M+H)+;熔点:126.4℃。 1 H NMR (400 MHz, CDCl 3 ) δppm 0.23-0.40 (m, 2H), 0.47-0.62 (m, 2H), 1.07-1.19 (m, 1H), 2.89 (s, 3H), 2.97 (s, 3H) , 3.03(d, 2H), 7.28(s, 1H), 7.60(d, 1H), 8.46(d, 1H); LC-MS (ESI) m/z: 380.1259 (M+H) + ; melting point: 126.4 °C.
根据本申请上述方法,使用不同的有机胺底物制备实施例2-6的化合物,所得化合物结构及其表征数据参见表1:According to the above-mentioned method of the present application, different organic amine substrates were used to prepare the compounds of Examples 2-6, and the structures of the obtained compounds and their characterization data are shown in Table 1:
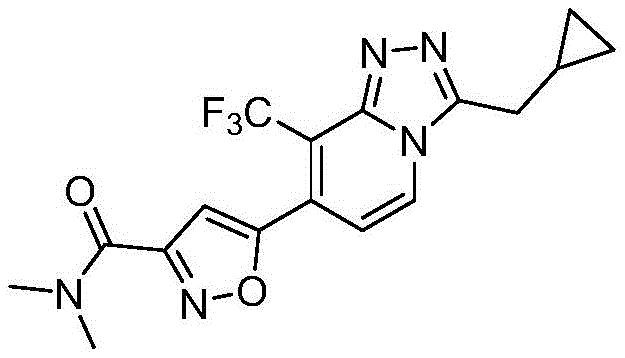
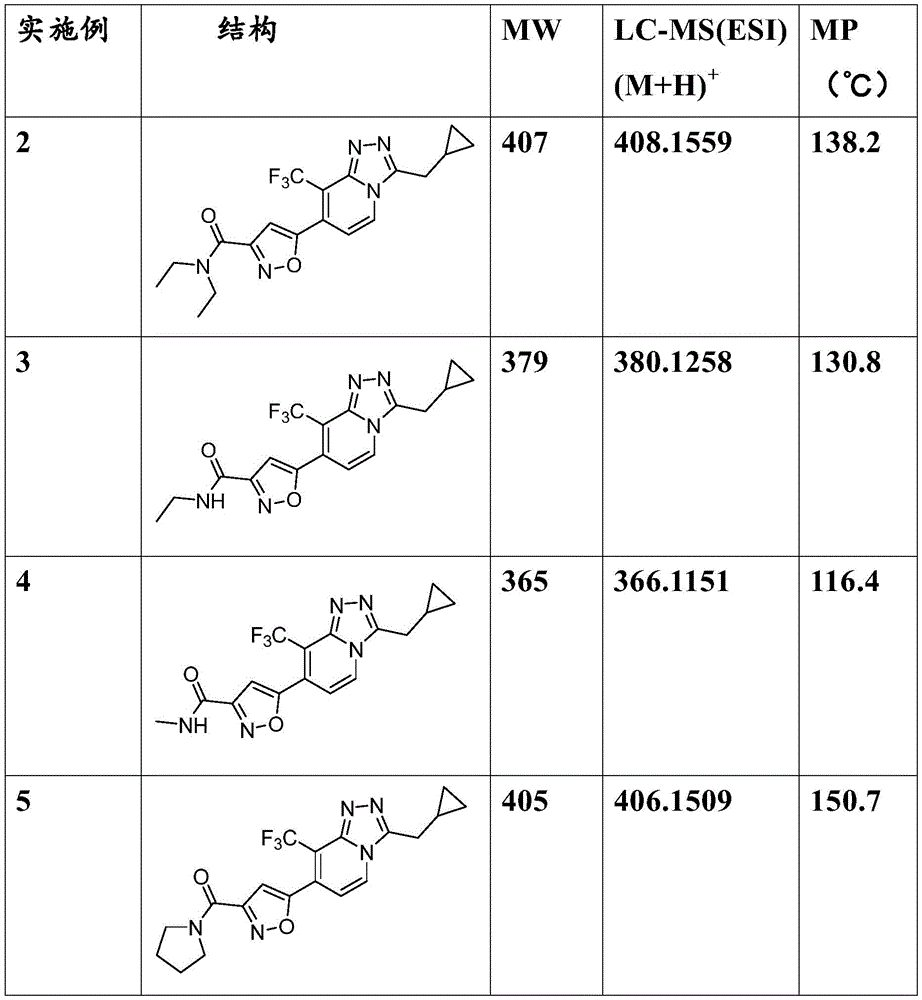
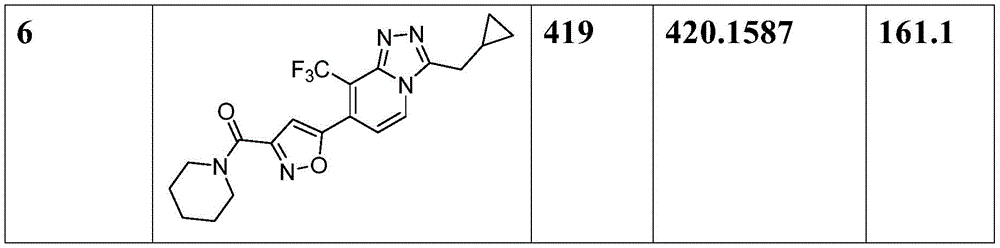
[表1][Table 1]
生物学活性测定:[35S]GTPγS结合测定Biological activity assay: [ 35 S]GTPγS binding assay
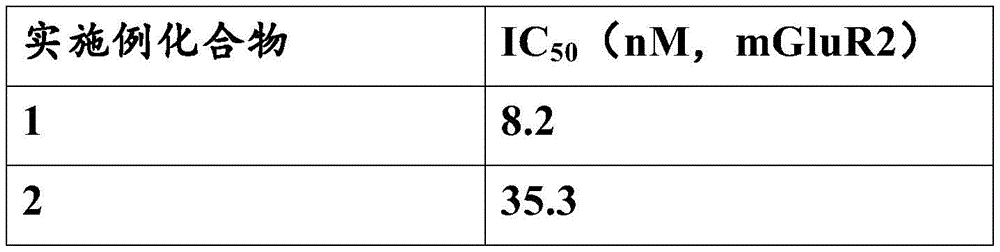
将稳定表达mGluR2的CHO细胞在35℃、5%CO2下使用含有10%胎牛血清的培养基[1%脯氨酸、1mM丙酮酸钠、1mM琥珀酸、1mM琥珀酸二钠、80单位/mL青霉素、80μg/mL链霉素、200μg/mL潮霉素B、3mM L-谷氨酰胺]培养。然后,使用PBS(-)洗涤上述培养的细胞,进行解离后在5℃下以1000rpm转速离心10min,以回收细胞。将所得的物质悬浮在20mM HEPES缓冲液(pH7.4)中,并在均化器中均化,然后在5℃下再进行离心15min,以再次获得颗粒,所得颗粒再进行2-3次均化和离心分离操作。所得颗粒于上述缓冲液匀浆。用缓冲液稀释上述均桨,以用于结合测试(最终浓度:10mM HEPES、800mM NaCl、10mM MgCl2、10MGDP、10μg/mL皂素(Sigma-Aldrich,CAS No:8047-15-2)、0.1%BSA)。向含有10μg膜蛋白/上述混合液中,分别添加化合物1-6,将所得混合物在30℃下培育30min。然后,向上述混合物中添加谷氨酸盐(最终浓度:20(mGluR2)和[35S]GTPγS(最终浓度:0.15nM);并将该混合物在30℃下培育30min。将所得培育溶液抽滤到沃特曼GF/C过滤器上以终止反应,并用3×500μL的冰冷的10mM磷酸钠缓冲液(pH 7.4)洗涤。将过滤器干燥,并向其中添加闪烁液,用液体闪烁仪测量膜结合放射活性。将不存在谷氨酸条件下残余放射性定义为非特异性结合,并且将存在谷氨酸条件下的残余放射性的差异定义为特异性结合。以不同浓度的化合物下的特异性结合的抑制百分率,使用非线性分析获得抑制曲线。以该抑制曲线,计算化合物抑制50%特异性结合的浓度(IC50)。所得结果参见表2。CHO cells stably expressing mGluR2 were incubated at 35°C in 5% CO using medium containing 10% fetal bovine serum [1% proline, 1 mM sodium pyruvate, 1 mM succinic acid, 1 mM disodium succinate, 80 units /mL penicillin, 80 μg/mL streptomycin, 200 μg/mL hygromycin B, 3 mM L-glutamine] culture. Then, the cultured cells were washed with PBS(-), dissociated, and centrifuged at 1000 rpm for 10 min at 5°C to recover the cells. The resulting material was suspended in 20 mM HEPES buffer (pH 7.4) and homogenized in a homogenizer, followed by centrifugation at 5°C for another 15 min to obtain particles again, and the resulting particles were further homogenized 2-3 times. and centrifugation operations. The resulting particles were homogenized in the above buffer. The above homogenate was diluted with buffer for binding assay (final concentrations: 10 mM HEPES, 800 mM NaCl, 10 mM MgCl 2 , 10 MGDP, 10 μg/mL Saponin (Sigma-Aldrich, CAS No: 8047-15-2), 0.1 % BSA). Compounds 1-6 were added to the mixture containing 10 μg of membrane protein/above, respectively, and the resulting mixture was incubated at 30° C. for 30 min. Then, glutamate (final concentration: 20(mGluR2) and [ 35 S]GTPγS (final concentration: 0.15 nM) were added to the above mixture; and the mixture was incubated at 30° C. for 30 min. The resulting incubation solution was suction filtered onto a Waterman GF/C filter to stop the reaction and wash with 3 x 500 μL of ice-cold 10 mM sodium phosphate buffer (pH 7.4). Dry the filter and add scintillation fluid to it and measure the membrane with a liquid scintillation meter Binding radioactivity. The residual radioactivity in the absence of glutamate was defined as non-specific binding, and the difference in residual radioactivity in the presence of glutamate was defined as specific binding. Specific binding at different concentrations of compound For percent inhibition, a non-linear analysis was used to obtain an inhibition curve. From this inhibition curve, the concentration of the compound that inhibited 50% of specific binding ( IC50 ) was calculated. See Table 2 for the results obtained.
[表2][Table 2]
上述活性结果表明,本发明的化合物对于mGluR2具有很好的拮抗作用。The above activity results show that the compounds of the present invention have a good antagonistic effect on mGluR2.
以上描述了本发明优选实施方式,然其并非用以限定本发明。本领域技术人员对在此公开的实施方案可进行并不偏离本发明范畴和精神的改进和变化。The preferred embodiments of the present invention have been described above, but are not intended to limit the present invention. Modifications and variations of the embodiments disclosed herein may be made by those skilled in the art without departing from the scope and spirit of the invention.
Claims (4)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201911310474.5A CN111116582B (en) | 2019-12-18 | 2019-12-18 | mGluR2 antagonist |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201911310474.5A CN111116582B (en) | 2019-12-18 | 2019-12-18 | mGluR2 antagonist |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111116582A CN111116582A (en) | 2020-05-08 |
| CN111116582B true CN111116582B (en) | 2022-07-29 |
Family
ID=70499650
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201911310474.5A Active CN111116582B (en) | 2019-12-18 | 2019-12-18 | mGluR2 antagonist |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111116582B (en) |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080161303A1 (en) * | 2006-10-11 | 2008-07-03 | Amgen Inc. | Imidazo- and triazolo-pyridine compounds and methods of use thereof |
| CN101679413A (en) * | 2007-03-02 | 2010-03-24 | 奥米罗有限公司 | New 3-([1,2,4]triazolo[4,3-a]pyridin-7-yl)benzamide derivatives |
| CN101679408A (en) * | 2006-12-22 | 2010-03-24 | Astex治疗学有限公司 | Bicyclic heterocyclic compounds as FGFR inhibitors |
| CN101827844A (en) * | 2007-10-17 | 2010-09-08 | 诺瓦提斯公司 | Imidazo [1,2-a] pyridine derivatives useful as ALK inhibitors |
| CN102439009A (en) * | 2009-05-12 | 2012-05-02 | 杨森制药有限公司 | 7-aryl-1, 2, 4-triazolo [4,3-a ] pyridine derivatives and their use as positive allosteric modulators of MGLUR2 receptors |
| CN102439015A (en) * | 2009-05-12 | 2012-05-02 | 杨森制药有限公司 | 1,2, 4-triazolo [4,3-a ] pyridine derivatives and their use as positive allosteric modulators of mGluR2 receptors |
| CN103298809A (en) * | 2010-11-08 | 2013-09-11 | 杨森制药公司 | 1,2,4-triazolo[4,3-a]pyridine derivatives and their use as positive allosteric modulators of MGLUR2 receptors |
| CN103298810A (en) * | 2010-11-08 | 2013-09-11 | 杨森制药公司 | 1,2,4-triazolo[4,3-a]pyridine derivatives and their use as positive allosteric modulators of mglur2 receptors |
-
2019
- 2019-12-18 CN CN201911310474.5A patent/CN111116582B/en active Active
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080161303A1 (en) * | 2006-10-11 | 2008-07-03 | Amgen Inc. | Imidazo- and triazolo-pyridine compounds and methods of use thereof |
| CN101679408A (en) * | 2006-12-22 | 2010-03-24 | Astex治疗学有限公司 | Bicyclic heterocyclic compounds as FGFR inhibitors |
| CN101679413A (en) * | 2007-03-02 | 2010-03-24 | 奥米罗有限公司 | New 3-([1,2,4]triazolo[4,3-a]pyridin-7-yl)benzamide derivatives |
| CN101827844A (en) * | 2007-10-17 | 2010-09-08 | 诺瓦提斯公司 | Imidazo [1,2-a] pyridine derivatives useful as ALK inhibitors |
| CN102439009A (en) * | 2009-05-12 | 2012-05-02 | 杨森制药有限公司 | 7-aryl-1, 2, 4-triazolo [4,3-a ] pyridine derivatives and their use as positive allosteric modulators of MGLUR2 receptors |
| CN102439015A (en) * | 2009-05-12 | 2012-05-02 | 杨森制药有限公司 | 1,2, 4-triazolo [4,3-a ] pyridine derivatives and their use as positive allosteric modulators of mGluR2 receptors |
| CN103298809A (en) * | 2010-11-08 | 2013-09-11 | 杨森制药公司 | 1,2,4-triazolo[4,3-a]pyridine derivatives and their use as positive allosteric modulators of MGLUR2 receptors |
| CN103298810A (en) * | 2010-11-08 | 2013-09-11 | 杨森制药公司 | 1,2,4-triazolo[4,3-a]pyridine derivatives and their use as positive allosteric modulators of mglur2 receptors |
Also Published As
| Publication number | Publication date |
|---|---|
| CN111116582A (en) | 2020-05-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8193192B2 (en) | Heterocyclic FXR binding compounds | |
| WO2022088551A1 (en) | Indazole derivative, and preparation method therefor and use thereof | |
| AU2005315881C1 (en) | Pyrrole derivatives having CRTh2 receptor antagonist activity | |
| WO2014134774A1 (en) | Compounds inhibiting leucine-rich repeat kinase enzyme activity | |
| JP2011525927A (en) | Synthesis and use of heterocyclic antibacterial agents | |
| CN101228145A (en) | Organic Compounds for the Treatment of Inflammatory or Allergic Conditions | |
| JPH06501030A (en) | Indole derivatives, pharmaceutical compositions and methods for treating nervous and psychiatric disorders | |
| CN107428752A (en) | Home chemokine inhibiting compounds | |
| US3845050A (en) | Dihydropyridazone compound substituted by paraimidazolidonylphenyl | |
| KR101495260B1 (en) | Gonadotropin releasing hormone receptor antagonists, method for the preparation thereof and pharmaceutical composition comprising the same | |
| TW202146403A (en) | Nitrogen-containing heterocyclic compound, preparation method, pharmaceutical composition and use thereof | |
| WO2015106717A1 (en) | Pyridazinone derivatives, preparation method and use therefor | |
| CN111116582B (en) | mGluR2 antagonist | |
| KR20130032848A (en) | Imide-containing bicyclic azole derivative or its salt and pharmaceutical composition comprising the same | |
| CN114641466A (en) | Sulfonylurea derivatives and use thereof | |
| WO2026021475A1 (en) | Fused tricyclic compound, and preparation method therefor and use thereof | |
| KR101132599B1 (en) | Indole derivatives with apoptosis-inducing effect | |
| CZ112699A3 (en) | N- (2-benzothiazolyl) -1-piperidineethanamine derivatives, process for their preparation and pharmaceutical compositions containing them | |
| KR19990022265A (en) | Pyridineiminyl-1,2-benzisoxazole and -benzisothiazole as antipsychotics | |
| KR20160034359A (en) | Imidazolecarboxamides and their use as faah inhibitors | |
| US7781460B2 (en) | Substituted indazoles as inhibitors of phosphodiesterase type-IV | |
| JP2023504795A (en) | Benzylamide derivatives as inhibitors of transforming growth factor-beta receptor I/alk5 - Patents.com | |
| CN113227080B (en) | 2,4-Diaminopyrimidine Derivatives | |
| KR20090089880A (en) | 2-alkyl-indazole compounds for treating certain CNS-related diseases | |
| JP2000038386A (en) | Novel naphthylidine derivative or its salt |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |