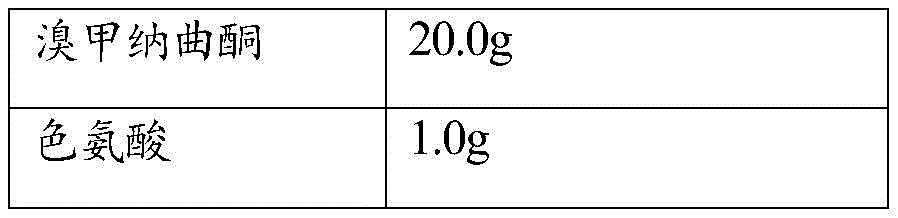CN110960486B - Methylnaltrexone bromide injection composition - Google Patents
Methylnaltrexone bromide injection composition Download PDFInfo
- Publication number
- CN110960486B CN110960486B CN201811143671.8A CN201811143671A CN110960486B CN 110960486 B CN110960486 B CN 110960486B CN 201811143671 A CN201811143671 A CN 201811143671A CN 110960486 B CN110960486 B CN 110960486B
- Authority
- CN
- China
- Prior art keywords
- methylnaltrexone bromide
- injection
- composition
- tryptophan
- methylnaltrexone
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- IFGIYSGOEZJNBE-LHJYHSJWSA-N (3s,4r,4as,7ar,12bs)-3-(cyclopropylmethyl)-4a,9-dihydroxy-3-methyl-2,4,5,6,7a,13-hexahydro-1h-4,12-methanobenzofuro[3,2-e]isoquinoline-3-ium-7-one;bromide Chemical compound [Br-].C([N@@+]1(C)[C@@H]2CC=3C4=C(C(=CC=3)O)O[C@@H]3[C@]4([C@@]2(O)CCC3=O)CC1)C1CC1 IFGIYSGOEZJNBE-LHJYHSJWSA-N 0.000 title claims abstract description 63
- 229960002834 methylnaltrexone bromide Drugs 0.000 title claims abstract description 63
- 238000002347 injection Methods 0.000 title claims abstract description 41
- 239000007924 injection Substances 0.000 title claims abstract description 41
- 239000000203 mixture Substances 0.000 title claims abstract description 30
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 claims abstract description 45
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims abstract description 26
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 claims abstract description 22
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 18
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 claims abstract description 15
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 claims abstract description 15
- 239000008215 water for injection Substances 0.000 claims abstract description 15
- 239000011780 sodium chloride Substances 0.000 claims abstract description 11
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 claims description 9
- VEUACKUBDLVUAC-UHFFFAOYSA-N [Na].[Ca] Chemical compound [Na].[Ca] VEUACKUBDLVUAC-UHFFFAOYSA-N 0.000 claims description 9
- 229960001484 edetic acid Drugs 0.000 claims description 9
- 239000000126 substance Substances 0.000 abstract description 7
- SHWNNYZBHZIQQV-UHFFFAOYSA-J EDTA monocalcium diisodium salt Chemical compound [Na+].[Na+].[Ca+2].[O-]C(=O)CN(CC([O-])=O)CCN(CC([O-])=O)CC([O-])=O SHWNNYZBHZIQQV-UHFFFAOYSA-J 0.000 abstract description 2
- 230000000694 effects Effects 0.000 description 8
- 238000002360 preparation method Methods 0.000 description 7
- 230000000052 comparative effect Effects 0.000 description 6
- 238000003756 stirring Methods 0.000 description 6
- 239000003963 antioxidant agent Substances 0.000 description 5
- 230000003078 antioxidant effect Effects 0.000 description 5
- 238000000034 method Methods 0.000 description 5
- 238000012360 testing method Methods 0.000 description 5
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 4
- 102000003840 Opioid Receptors Human genes 0.000 description 3
- 108090000137 Opioid Receptors Proteins 0.000 description 3
- 239000003814 drug Substances 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 230000007774 longterm Effects 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 206010067484 Adverse reaction Diseases 0.000 description 2
- 206010010774 Constipation Diseases 0.000 description 2
- 239000004902 Softening Agent Substances 0.000 description 2
- 230000006838 adverse reaction Effects 0.000 description 2
- 239000003708 ampul Substances 0.000 description 2
- 230000000202 analgesic effect Effects 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 230000008499 blood brain barrier function Effects 0.000 description 2
- 210000001218 blood-brain barrier Anatomy 0.000 description 2
- 210000003169 central nervous system Anatomy 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 239000000835 fiber Substances 0.000 description 2
- 238000011049 filling Methods 0.000 description 2
- 239000000706 filtrate Substances 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 230000002496 gastric effect Effects 0.000 description 2
- 238000011835 investigation Methods 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 229940005483 opioid analgesics Drugs 0.000 description 2
- 238000004806 packaging method and process Methods 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 238000007789 sealing Methods 0.000 description 2
- 239000011265 semifinished product Substances 0.000 description 2
- 230000001954 sterilising effect Effects 0.000 description 2
- IFGIYSGOEZJNBE-KNLJMPJLSA-N (4r,4as,7ar,12bs)-3-(cyclopropylmethyl)-4a,9-dihydroxy-3-methyl-2,4,5,6,7a,13-hexahydro-1h-4,12-methanobenzofuro[3,2-e]isoquinoline-3-ium-7-one;bromide Chemical compound [Br-].C[N+]1([C@@H]2CC=3C4=C(C(=CC=3)O)O[C@@H]3[C@]4([C@@]2(O)CCC3=O)CC1)CC1CC1 IFGIYSGOEZJNBE-KNLJMPJLSA-N 0.000 description 1
- 208000004998 Abdominal Pain Diseases 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- 206010012735 Diarrhoea Diseases 0.000 description 1
- 229940123257 Opioid receptor antagonist Drugs 0.000 description 1
- 230000008485 antagonism Effects 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 230000002860 competitive effect Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 208000002173 dizziness Diseases 0.000 description 1
- 206010016766 flatulence Diseases 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- 230000008570 general process Effects 0.000 description 1
- 102000051367 mu Opioid Receptors Human genes 0.000 description 1
- 239000002623 mu opiate receptor antagonist Substances 0.000 description 1
- 229940127240 opiate Drugs 0.000 description 1
- 239000003401 opiate antagonist Substances 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 1
- 238000013112 stability test Methods 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 108020001612 μ-opioid receptors Proteins 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/47—Quinolines; Isoquinolines
- A61K31/485—Morphinan derivatives, e.g. morphine, codeine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/02—Inorganic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/12—Carboxylic acids; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/16—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing nitrogen, e.g. nitro-, nitroso-, azo-compounds, nitriles, cyanates
- A61K47/18—Amines; Amides; Ureas; Quaternary ammonium compounds; Amino acids; Oligopeptides having up to five amino acids
- A61K47/183—Amino acids, e.g. glycine, EDTA or aspartame
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/08—Solutions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/10—Laxatives
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Emergency Medicine (AREA)
- Inorganic Chemistry (AREA)
- Dermatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
The invention provides a methylnaltrexone bromide injection composition, wherein each 1000ml of the composition comprises the following components: 20g of methylnaltrexone bromide, 0.01-1.0g of tryptophan and 0.1-2.0g of calcium disodium edetate; 0.01-0.5g of citric acid and 5.5-7.5g of sodium chloride; adjusting pH to 3.0-5.0 with hydrochloric acid, and adding appropriate amount of water for injection. The methylnaltrexone bromide injection has the advantages of low content of related substances, high stability and the like.
Description
Technical Field
The invention relates to the field of pharmacy, and in particular relates to a methylnaltrexone bromide injection composition.
Background
Methylnaltrexone bromide (methylnaltrexone bromide, MNTX), english name: methynaltrexone bromide, a selective mu opioid receptor antagonist. Being a quaternary ammonium salt, limits the ability of methylnaltrexone bromide to cross the blood-brain barrier, allowing methylnaltrexone bromide to act as a peripherally acting opioid receptor antagonist, e.g. in gastrointestinal tissues, and thus, methylnaltrexone bromide reduces the constipation effects of opioids without affecting the analgesic effects of opioids on the central nervous system.
Methylnaltrexone bromide has the following characteristics: 1) act on peripheral mu receptors and do not activate central nervous system opioid receptors, possibly acting as competitive antagonism; 2) safety, no serious toxic or side effect is found in a series of experiments, and common adverse reactions are abdominal pain, diarrhea, flatulence and dizziness; 3) the effect is quick, and the effect can be achieved within minutes; 4) the curative effect of the constipation relieving agent and the softening agent is better than that of the stool cathartic and the softening agent; 5) the acting route is not unique, and the medicine can be directly combined with opioid receptors on gastrointestinal tracts to take effect and can also be combined with the opioid receptors by the whole body through blood distribution. Therefore, the bromomethylnaltrexone can treat not only the gastrointestinal side effects of the opioid, but also the parenteral adverse reactions; 6) does not pass through the blood brain barrier and does not weaken the central analgesic effect of the opiate.
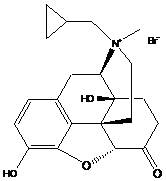
Methylnaltrexone bromide has the following structural formula:
the molecular formula is as follows: c21H26NO4Br
Molecular weight: 436.36
Up to now, the package specification of methylnaltrexone bromide injection is 0.6 ml of aqueous solution containing 12mg of methylnaltrexone bromide per bottle. However, since the aqueous solution of methylnaltrexone bromide is not stable enough and is easily degraded particularly by light, on the other hand, the methylnaltrexone bromide has insufficient solubility in water, which makes it difficult to prepare the preparation.
Therefore, the prior art still needs an injection of methylnaltrexone bromide, which is simple to prepare, stable in quality and qualified after long-term storage.
Disclosure of Invention
In the process of developing the methylnaltrexone bromide injection, the applicant finds that the tryptophan and the citric acid are matched according to a certain proportion, the content of related substances of the methylnaltrexone bromide injection is obviously reduced, the stability of the methylnaltrexone bromide injection is improved, and an unexpected technical effect is achieved. Based on the above findings, the inventors have provided a methylnaltrexone bromide injection having a simple process, a low content of related substances, a high stability, and a high safety of administration, and have completed the present invention.
Specifically, the invention provides a methylnaltrexone bromide injection composition, wherein each 1000ml of the composition comprises the following components:
| methylnaltrexone bromide | 20.0g |
| Tryptophan | 0.01-1.0g |
| Edetic acid calcium sodium salt | 0.1-2.0g |
| Citric acid | 0.01-1.0g |
| Sodium chloride | 5.5-7.5g |
| Hydrochloric acid | Adjusting the pH to 3.0-5.0 |
| Water for injection | 1000ml |
In a preferable embodiment of the invention, the weight ratio of tryptophan to citric acid is 1: 1.
In addition, the invention particularly provides the following three compositions of methylnaltrexone bromide injection liquid:
1) a methylnaltrexone bromide injection composition, wherein each 1000ml of the composition comprises the following components:
| methylnaltrexone bromide | 20.0g |
| Tryptophan | 0.01g |
| Edetic acid calcium sodium salt | 0.1g |
| Citric acid | 0.01g |
| Sodium chloride | 6.0g |
| Hydrochloric acid | Adjusting the pH to 3.5 |
| Water for injection | 1000ml |
2) A methylnaltrexone bromide injection composition, wherein each 1000ml of the composition comprises the following components:
| methylnaltrexone bromide | 20.0g |
| Tryptophan | 0.5g |
| Edetic acid calcium sodium salt | 1.0g |
| Citric acid | 0.5g |
| Sodium chloride | 7.0g |
| Hydrochloric acid | Adjusting the pH to 4.5 |
| Water for injection | 1000ml |
3) A methylnaltrexone bromide injection composition, wherein each 1000ml of the composition comprises the following components:
| methylnaltrexone bromide | 20.0g |
| Tryptophan | 1.0g |
| Edetic acid calcium sodium salt | 2.0g |
| Citric acid | 1.0g |
| Sodium chloride | 75g |
| Hydrochloric acid | Adjusting the pH to 5.0 |
| Water for injection | 1000ml |
On the other hand, the invention also relates to a preparation method of the methylnaltrexone bromide injection, which comprises the following steps:
adding a prescribed amount of methylnaltrexone bromide raw material and auxiliary materials into about 3/4 prescribed amount of water for injection, stirring to dissolve, adjusting the pH value to 3.5 by using 1.0mol/L hydrochloric acid, adding the water for injection to the prescribed amount, adding 0.15% of liquid medicine amount (w/v) of needle activated carbon, stirring to uniformly mix, stirring for 15 minutes, filtering by using a 0.22 mu m fiber mixing water system microporous filter membrane, and inspecting a semi-finished product. Filling the filtrate in ampoule, charging nitrogen, sealing, sterilizing at 121 deg.C for 15 min, testing, and packaging.
Experiments show that the methylnaltrexone bromide injection has low content of related substances, high stability and high safety of patients.
The specific implementation mode is as follows:
the detailed description of the invention is merely intended to further illustrate or describe the invention and should not be construed as limiting the invention in any way.
Example 1: methylnaltrexone bromide injection
The composition and content of methylnaltrexone bromide injection (specification is 12mg of methylnaltrexone bromide in each injection):
| methylnaltrexone bromide | 20.0g |
| Tryptophan | 0.01g |
| Edetic acid calcium sodium salt | 0.1g |
| Citric acid | 0.01g |
| Sodium chloride | 6.0g |
| Hydrochloric acid | Adjusting the pH to 3.0-5.0 |
| Water for injection | 1000ml |
The preparation process comprises the following steps:
adding a prescribed amount of methylnaltrexone bromide raw material and auxiliary materials into about 3/4 prescribed amount of water for injection, stirring to dissolve, adjusting the pH value to 3.0-5.0 by using 1.0mol/L hydrochloric acid, adding water for injection to the prescribed amount, adding 0.15% of liquid medicine amount (w/v) of needle activated carbon, stirring to mix uniformly, stirring for 15 minutes, filtering by using a 0.22 mu m fiber mixing water system microporous filter membrane, and inspecting a semi-finished product. Filling the filtrate in ampoule, charging nitrogen, sealing, sterilizing at 121 deg.C for 15 min, testing, and packaging.
Example 2: methylnaltrexone bromide injection
The composition and content of methylnaltrexone bromide injection (specification is 12mg of methylnaltrexone bromide in each injection):
| methylnaltrexone bromide | 20.0g |
| Tryptophan | 1.0g |
| Edetic acid calcium sodium salt | 2.0g |
| Citric acid | 1.0g |
| Sodium chloride | 75g |
| Hydrochloric acid | Adjusting the pH to 3.0-5.0 |
| Water for injection | 1000ml |
The preparation process comprises the following steps: the same as in example 1.
Example 3: methylnaltrexone bromide injection
The composition and content of methylnaltrexone bromide injection (specification is 12mg of methylnaltrexone bromide in each injection):
| methylnaltrexone bromide | 20.0g |
| Tryptophan | 1.0g |
| Calcium disodium edetate | 2.0g |
| Citric acid | 1.0g |
| Sodium chloride | 75g |
| Hydrochloric acid | Adjusting the pH to 5.0 |
| Water for injection | 1000ml |
The preparation process comprises the following steps: the same as in example 1.
Example 4 (comparative example 1): methylnaltrexone bromide injection
The comparative examples were prepared according to the method in the specification of Chinese patent 200780009723.6, page 27, table 1, column 2, and 200780009723.6.
Example 5 (comparative example 2): methylnaltrexone bromide injection
The comparative example was prepared according to the recipe process of example 1 of chinese patent specification 201410808984.6.
Experimental example 1: the type and amount of antioxidant
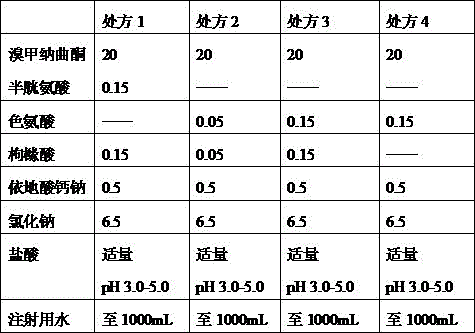
Adding main and auxiliary materials according to a prescription design table, operating according to general process procedures of the preparation, selecting a common antioxidant for investigation, wherein specific prescriptions and results are shown in tables 1-2:
TABLE 1 antioxidant survey prescription Table
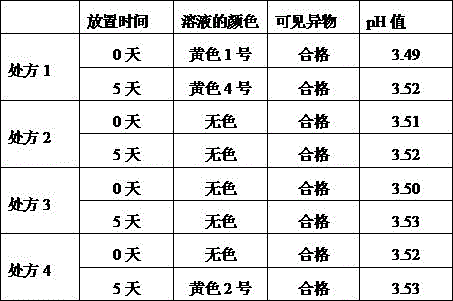
The preparation process comprises the following steps: the same as in example 1. The sterilized injection was placed in an incubator at 80 ℃ for 5 days, and the results were as follows:
TABLE 2 basic performance indexes of antioxidant prescription and investigation results after being placed at 80 ℃ for 5 days
According to the experimental results, the formulas 2 and 3 are preferably screened and determined, namely tryptophan and citric acid are selected as the antioxidant.
Experimental example 2: stability study of methylnaltrexone bromide injection
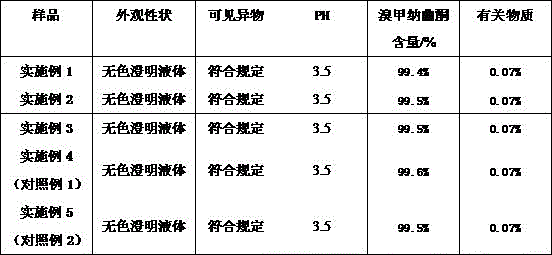
Examining the methylnaltrexone bromide injection prepared in the examples 1 to 5, the examination results of related substances and the like are shown in the following tables 3 to 5:
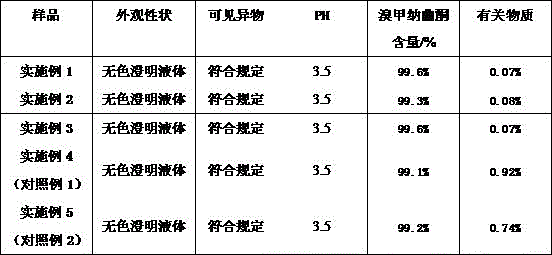
TABLE 3 Methylnaltrexone bromide injection test results (day 0)
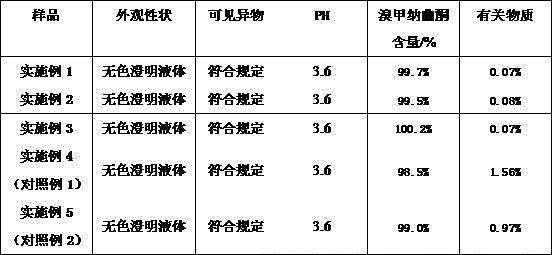
TABLE 4 accelerated test results for methylnaltrexone bromide injection (40 ℃. + -. 2 ℃, RH 75%. + -. 5%, 6 months)
Accelerated tests show that the content and stability of related substances of the methylnaltrexone bromide injection (examples 1-3) are obviously better than those of the comparative example (examples 4-5).
TABLE 5 Long-term stability test results for methylnaltrexone bromide injection (25 deg.C 2 deg.C, RH60% + -10%, 24 months)
Long-term stability studies show that the content of related substances and the stability of the methylnaltrexone bromide injection (examples 1-3) are obviously better than those of the comparative example (examples 4-5).
Claims (4)
1. A methylnaltrexone bromide injection composition, wherein the composition of each 1000mL of the composition is as follows:
The weight ratio of the tryptophan to the citric acid is 1: 1.
2. The methylnaltrexone bromide injection composition of claim 1, having the following composition per 1000mL of said composition:
。
3. The methylnaltrexone bromide injection composition of claim 1, having the following composition per 1000mL of said composition:
。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811143671.8A CN110960486B (en) | 2018-09-29 | 2018-09-29 | Methylnaltrexone bromide injection composition |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811143671.8A CN110960486B (en) | 2018-09-29 | 2018-09-29 | Methylnaltrexone bromide injection composition |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN110960486A CN110960486A (en) | 2020-04-07 |
| CN110960486B true CN110960486B (en) | 2022-05-13 |
Family
ID=70027092
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201811143671.8A Active CN110960486B (en) | 2018-09-29 | 2018-09-29 | Methylnaltrexone bromide injection composition |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN110960486B (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN120241599A (en) * | 2025-05-12 | 2025-07-04 | 北京化药科创医药科技发展有限公司 | Methylnaltrexone bromide injection resistant to terminal sterilization and preparation method thereof |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101357118A (en) * | 2008-09-26 | 2009-02-04 | 郑飞雄 | Medicine composition containing 15 kinds of amino acid and preparation method thereof |
| CN101405031A (en) * | 2006-08-04 | 2009-04-08 | 惠氏公司 | Formulations for parenteral delivery of compounds and uses thereof |
| CN101406474A (en) * | 2008-02-28 | 2009-04-15 | 云南绿野生物医药有限公司 | Nalmefene injection and preparation method thereof |
| CN101732243A (en) * | 2008-11-26 | 2010-06-16 | 重庆医药工业研究院有限责任公司 | Stable methyl naltrexone injection and preparation method thereof |
| CN105582011A (en) * | 2014-10-24 | 2016-05-18 | 江苏国丹生物制药有限公司 | Methylnaltrexone bromide solid composition and preparation method thereof |
| CN105769755A (en) * | 2014-12-23 | 2016-07-20 | 北大方正集团有限公司 | Methyhaaltrexone bromide injection and preparation method thereof |
-
2018
- 2018-09-29 CN CN201811143671.8A patent/CN110960486B/en active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101405031A (en) * | 2006-08-04 | 2009-04-08 | 惠氏公司 | Formulations for parenteral delivery of compounds and uses thereof |
| CN101406474A (en) * | 2008-02-28 | 2009-04-15 | 云南绿野生物医药有限公司 | Nalmefene injection and preparation method thereof |
| CN101357118A (en) * | 2008-09-26 | 2009-02-04 | 郑飞雄 | Medicine composition containing 15 kinds of amino acid and preparation method thereof |
| CN101732243A (en) * | 2008-11-26 | 2010-06-16 | 重庆医药工业研究院有限责任公司 | Stable methyl naltrexone injection and preparation method thereof |
| CN105582011A (en) * | 2014-10-24 | 2016-05-18 | 江苏国丹生物制药有限公司 | Methylnaltrexone bromide solid composition and preparation method thereof |
| CN105769755A (en) * | 2014-12-23 | 2016-07-20 | 北大方正集团有限公司 | Methyhaaltrexone bromide injection and preparation method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| CN110960486A (en) | 2020-04-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2019038584A1 (en) | ORAL PHARMACEUTICAL COMPOSITION COMPRISING ZONISAMIDE AND PREPARATION METHOD THEREOF | |
| CN113332239A (en) | Adrenaline hydrochloride injection and preparation method thereof | |
| CN1332662C (en) | Levo ornidazole vein administration agent and its preparation method | |
| CN100536848C (en) | Stable nalmefene hydrochloride injection and preparation method thereof | |
| CN110960486B (en) | Methylnaltrexone bromide injection composition | |
| CN110269837B (en) | Naloxone hydrochloride injection and preparation method thereof | |
| CA3016020C (en) | Liquid medicinal preparation | |
| CN101485650B (en) | Diclofenac sodium and lidocaine hydrochloride injection and preparation method thereof | |
| CN102688185B (en) | Stable palonosetron injection and preparation method thereof | |
| CN112294756A (en) | Cisatracurium besilate injection and preparation method thereof | |
| CN102166185B (en) | Isotonic naloxone injection and preparation method thereof | |
| CN102274194B (en) | Pharmaceutical composition containing tropisetron compound and preparation method thereof | |
| CN111568860B (en) | Ornidazole injection and preparation method thereof | |
| CN114569545B (en) | Stable mitoxantrone preparation | |
| CN105769755A (en) | Methyhaaltrexone bromide injection and preparation method thereof | |
| CN100367962C (en) | A kind of stable palonosetron injection and preparation method thereof | |
| CN102440951B (en) | Azasetron hydrochloride injection and preparation method thereof | |
| CN113197848A (en) | Metalhydroxylamine bitartrate pharmaceutical composition and preparation method thereof | |
| CN100386078C (en) | Nimodipine emulsion injection and preparation method | |
| CN105816424A (en) | Argatroban composition and preparation method thereof | |
| CN104958255B (en) | A kind of Flumazenil parenteral solution and preparation method thereof | |
| AU2022329224B2 (en) | Pharmaceutical composition, preparation method therefor and application thereof | |
| CN104840465B (en) | Flumazenil injection and preparation method thereof | |
| CN103202805A (en) | Vinpocetine-containing pharmaceutical composition for injection and preparation method thereof | |
| CN103877578B (en) | Pharmaceutical naloxone hydrochloride composition for injection and preparation method of pharmaceutical naloxone hydrochloride composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |