CN110872302A - Pyrimidine-containing spiro compounds and uses thereof - Google Patents
Pyrimidine-containing spiro compounds and uses thereof Download PDFInfo
- Publication number
- CN110872302A CN110872302A CN201811013143.0A CN201811013143A CN110872302A CN 110872302 A CN110872302 A CN 110872302A CN 201811013143 A CN201811013143 A CN 201811013143A CN 110872302 A CN110872302 A CN 110872302A
- Authority
- CN
- China
- Prior art keywords
- radical
- halo
- alkyl
- alkoxy
- alkylthio
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/02—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains two hetero rings
- C07D491/10—Spiro-condensed systems
- C07D491/107—Spiro-condensed systems with only one oxygen atom as ring hetero atom in the oxygen-containing ring
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/90—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having two or more relevant hetero rings, condensed among themselves or with a common carbocyclic ring system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
本发明公开了一种含嘧啶的螺环类化合物,结构如通式I所示:式中各取代基的定义见说明书。本发明的化合物具有广谱杀菌活性,对黄瓜霜霉病、小麦白粉病、玉米锈病、稻瘟病、黄瓜炭疽病等具有优良的防治效果。The invention discloses a pyrimidine-containing spirocyclic compound whose structure is shown in general formula I: The definition of each substituent in the formula is shown in the specification. The compound of the invention has broad-spectrum bactericidal activity, and has excellent control effect on cucumber downy mildew, wheat powdery mildew, corn rust, rice blast, cucumber anthracnose and the like.
Description
技术领域technical field
本发明属农用杀菌领域,具体涉及一种新型的含嘧啶的螺环类化合物及其用途。The invention belongs to the field of agricultural sterilization, and in particular relates to a novel pyrimidine-containing spirocyclic compound and use thereof.
背景技术Background technique
专利WO2007054580(A1)公布了如下通式所示的螺环类化合物通式及具体化合物CK1和CK2,作为医用青光眼、癫痫、偏头痛和癌症等疾病的预防和治疗的应用Patent WO2007054580 (A1) discloses the general formula of the spiro compounds shown in the following general formula and the specific compounds CK1 and CK2, as the application of the prevention and treatment of medical glaucoma, epilepsy, migraine and cancer
但结构如本发明通式I所示的含嘧啶的螺环类化合物作为农用的未见报道。However, there is no report on the pyrimidine-containing spiro compounds having the structure as shown in the general formula I of the present invention as agricultural use.
发明内容SUMMARY OF THE INVENTION
本发明的目的在于提供一种可以控制多种病菌的含嘧啶的螺环类化合物及用于农业或其他领域中制备防治病菌的药物的用途。The purpose of the present invention is to provide a pyrimidine-containing spiro compound capable of controlling various pathogens and the use of a medicament for preventing and treating pathogens in agriculture or other fields.
为实现上述目的,本发明的技术方案如下:For achieving the above object, technical scheme of the present invention is as follows:
一种含嘧啶的螺环类化合物,其特征在于:含嘧啶的螺环类化合物为通式I所示化合物;A pyrimidine-containing spiro compound, characterized in that: the pyrimidine-containing spiro compound is a compound shown in general formula I;
R1选自氢、卤素、氰基、硝基、氨基、羧基、C1-C12烷基、卤代C1-C12烷基、C3-C12环烷基、C1-C12烷氧基、卤代C1-C12烷氧基、C1-C12烷硫基、卤代C1-C12烷硫基、C1-C12烷基亚磺酰基、C1-C12烷基磺酰基、C2-C12烯基、卤代C2-C12烯基、C2-C12炔基、卤代C2-C12炔基、C3-C12烯氧基、卤代C3-C12烯氧基、C3-C12炔氧基、卤代C3-C12炔氧基、C1-C12烷基氨基、二(C1-C12烷基)氨基、C1-C12烷基氨基羰基、卤代C1-C12烷基氨基羰基、C1-C12烷氧基羰基、卤代C1-C12烷氧基羰基、C1-C12烷氧基C1-C12烷基或C1-C2烷硫基C1-C12烷基;R 1 is selected from hydrogen, halogen, cyano, nitro, amino, carboxyl, C 1 -C 12 alkyl, halogenated C 1 -C 12 alkyl, C 3 -C 12 cycloalkyl, C 1 -C 12 Alkoxy, halogenated C 1 -C 12 alkoxy, C 1 -C 12 alkylthio, halogenated C 1 -C 12 alkylthio, C 1 -C 12 alkylsulfinyl, C 1 -C 12 Alkylsulfonyl, C 2 -C 12 alkenyl, halogenated C 2 -C 12 alkenyl, C 2 -C 12 alkynyl, halogenated C 2 -C 12 alkynyl, C 3 -C 12 alkenyloxy , halogenated C 3 -C 12 alkenyloxy, C 3 -C 12 alkynyloxy, halogenated C 3 -C 12 alkynyloxy, C 1 -C 12 alkylamino, di(C 1 -C 12 alkyl ) amino, C 1 -C 12 alkylaminocarbonyl, halogenated C 1 -C 12 alkylaminocarbonyl, C 1 -C 12 alkoxycarbonyl, halogenated C 1 -C 12 alkoxycarbonyl, C 1 - C 12 alkoxy C 1 -C 12 alkyl or C 1 -C 2 alkylthio C 1 -C 12 alkyl;
R2选自氢、卤素、氰基、硝基、氨基、羧基、甲酰基、C1-C12烷基、卤代C1-C12烷基、C1-C12烷氧基、卤代C1-C12烷氧基或二氧戊环基 R 2 is selected from hydrogen, halogen, cyano, nitro, amino, carboxyl, formyl, C 1 -C 12 alkyl, halogenated C 1 -C 12 alkyl, C 1 -C 12 alkoxy, halo C 1 -C 12 alkoxy or dioxolane
R1和R2与相连的嘧啶环组成含C、N、O或S的五元、六元、七元或八元环;R 1 and R 2 and the connected pyrimidine ring form a five-membered, six-membered, seven-membered or eight-membered ring containing C, N, O or S;
W选自氢、卤素、C1-C12烷基、卤代C1-C12烷基、C3-C8环烷基、C1-C12烷氧基、C1-C12烷硫基或C1-C12烷基磺酰基;W is selected from hydrogen, halogen, C 1 -C 12 alkyl, halogenated C 1 -C 12 alkyl, C 3 -C 8 cycloalkyl, C 1 -C 12 alkoxy, C 1 -C 12 alkylthio group or C 1 -C 12 alkylsulfonyl;
X1、X2、X3、X4相同或不同的选自C或N,其中X1、X2、X3、X4与另外两个C(一个与O相连,另一个与CO相连)组成被0-4个R3取代的芳基或杂芳基;X 1 , X 2 , X 3 , X 4 which are the same or different are selected from C or N, wherein X 1 , X 2 , X 3 , X 4 are connected to two other Cs (one is connected to O and the other is connected to CO) The composition is aryl or heteroaryl substituted by 0-4 R 3 ;
R3选自卤素、羟基、氨基、氰基、硝基、C1-C12烷基、卤代C1-C12烷基、C1-C12烷氧基、卤代C1-C12烷氧基、C3-C12环烷基、C1-C12烷基氨基、卤代C1-C12烷基氨基、二(C1-C12烷基)氨基、卤代二(C1-C12烷基)氨基、C1-C12烷硫基、卤代C1-C12烷硫基、C2-C12烯基、C2-C12炔基、C2-C12烯氧基、卤代C2-C12烯氧基、C2-C12炔氧基、卤代C2-C12炔氧基、C1-C12烷基磺酰基、卤代C1-C12烷基磺酰基、C1-C12烷基羰基、卤代C1-C12烷基羰基、C1-C12烷氧基羰基、卤代C1-C12烷氧基羰基、C1-C12烷氧基C1-C12烷基、卤代C1-C12烷氧基C1-C12烷基、C1-C12烷硫基C1-C12烷基、卤代C1-C12烷硫基C1-C12烷基、C1-C12烷氧基羰基C1-C12烷基、卤代C1-C12烷氧基羰基C1-C12烷基、C1-C12烷硫基羰基C1-C12烷基、卤代C1-C12烷硫基羰基C1-C12烷基、C1-C12烷基羰基氧基、卤代C1-C12烷基羰基氧基、C1-C12烷氧基羰基氧基、卤代C1-C12烷氧基羰基氧基、C1-C12烷基磺酰基氧基、卤代C1-C12烷基磺酰基氧基、C1-C12烷氧基C1-C12烷氧基或卤代C1-C12烷氧基C1-C12烷氧基。R 3 is selected from halogen, hydroxyl, amino, cyano, nitro, C 1 -C 12 alkyl, halogenated C 1 -C 12 alkyl, C 1 -C 12 alkoxy, halogenated C 1 -C 12 Alkoxy, C 3 -C 12 cycloalkyl, C 1 -C 12 alkylamino, halogenated C 1 -C 12 alkylamino, bis(C 1 -C 12 alkyl)amino, halogenated bis(C 1 -C 12 alkyl) amino, C 1 -C 12 alkylthio, halogenated C 1 -C 12 alkylthio, C 2 -C 12 alkenyl, C 2 -C 12 alkynyl, C 2 -C 12 Alkenyloxy, halogenated C 2 -C 12 alkenyloxy, C 2 -C 12 alkynyloxy, halogenated C 2 -C 12 alkynyloxy, C 1 -C 12 alkylsulfonyl, halogenated C 1 - C 12 alkylsulfonyl, C 1 -C 12 alkylcarbonyl, halogenated C 1 -C 12 alkylcarbonyl, C 1 -C 12 alkoxycarbonyl, halogenated C 1 -C 12 alkoxycarbonyl, C 1 -C 12 alkoxy C 1 -C 12 alkyl, halogenated C 1 -C 12 alkoxy C 1 -C 12 alkyl, C 1 -C 12 alkylthio C 1 -C 12 alkyl, halogen Substituted C 1 -C 12 alkylthio C 1 -C 12 alkyl, C 1 -C 12 alkoxycarbonyl C 1 -C 12 alkyl, halogenated C 1 -C 12 alkoxycarbonyl C 1 -C 12 Alkyl, C 1 -C 12 alkylthiocarbonyl C 1 -C 12 alkyl, halogenated C 1 -C 12 alkylthiocarbonyl C 1 -C 12 alkyl, C 1 -C 12 alkylcarbonyloxy, Halogenated C 1 -C 12 alkylcarbonyloxy, C 1 -C 12 alkoxycarbonyloxy, halogenated C 1 -C 12 alkoxycarbonyloxy, C 1 -C 12 alkylsulfonyloxy , halogenated C 1 -C 12 alkylsulfonyloxy, C 1 -C 12 alkoxy C 1 -C 12 alkoxy or halogenated C 1 -C 12 alkoxy C 1 -C 12 alkoxy .
本发明含嘧啶的螺环类化合物中,较为优选的化合物包括:所示通式I中:Among the pyrimidine-containing spiro compounds of the present invention, more preferred compounds include: in the general formula I shown:
R1选自氢、卤素、氰基、硝基、氨基、羧基、C1-C6烷基、卤代C1-C6烷基、C3-C6环烷基、C1-C6烷氧基、卤代C1-C6烷氧基、C1-C6烷硫基、C1-C6烷基亚磺酰基、C1-C6烷基磺酰基、卤代C1-C6烷硫基、C2-C6烯基、卤代C2-C6烯基、C2-C6炔基、卤代C2-C6炔基、C3-C6烯氧基、卤代C3-C6烯氧基、C3-C6炔氧基、卤代C3-C6炔氧基、C1-C6烷基氨基、二(C1-C6烷基)氨基、C1-C6烷基氨基羰基、卤代C1-C6烷基氨基羰基、C1-C6烷氧基羰基、卤代C1-C6烷氧基羰基、C1-C6烷氧基C1-C6烷基或C1-C6烷硫基C1-C6烷基;R 1 is selected from hydrogen, halogen, cyano, nitro, amino, carboxyl, C 1 -C 6 alkyl, halogenated C 1 -C 6 alkyl, C 3 -C 6 cycloalkyl, C 1 -C 6 Alkoxy, halogenated C 1 -C 6 alkoxy, C 1 -C 6 alkylthio, C 1 -C 6 alkylsulfinyl, C 1 -C 6 alkylsulfonyl, halogenated C 1 - C 6 alkylthio, C 2 -C 6 alkenyl, halogenated C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, halogenated C 2 -C 6 alkynyl, C 3 -C 6 alkenyloxy , halogenated C 3 -C 6 alkenyloxy, C 3 -C 6 alkynyloxy, halogenated C 3 -C 6 alkynyloxy, C 1 -C 6 alkylamino, di(C 1 -C 6 alkyl ) amino, C 1 -C 6 alkylaminocarbonyl, halogenated C 1 -C 6 alkylaminocarbonyl, C 1 -C 6 alkoxycarbonyl, halogenated C 1 -C 6 alkoxycarbonyl, C 1 - C 6 alkoxy C 1 -C 6 alkyl or C 1 -C 6 alkylthio C 1 -C 6 alkyl;
R2选自氢、卤素、氰基、硝基、氨基、羧基、甲酰基、C1-C6烷基、卤代C1-C6烷基、C1-C6烷氧基、卤代C1-C6烷氧基或二氧戊环基;R 2 is selected from hydrogen, halogen, cyano, nitro, amino, carboxyl, formyl, C 1 -C 6 alkyl, halogenated C 1 -C 6 alkyl, C 1 -C 6 alkoxy, halogenated C 1 -C 6 alkoxy or dioxolane;
R1和R2与相连的嘧啶环组成含C、N、O或S的五元或六元环;R 1 and R 2 and the connected pyrimidine ring form a five- or six-membered ring containing C, N, O or S;
W选自氢、卤素、C1-C6烷基、卤代C1-C6烷基、C3-C6环烷基、C1-C6烷氧基、C1-C6烷硫基或C1-C6烷基磺酰基;W is selected from hydrogen, halogen, C 1 -C 6 alkyl, halogenated C 1 -C 6 alkyl, C 3 -C 6 cycloalkyl, C 1 -C 6 alkoxy, C 1 -C 6 alkylthio group or C 1 -C 6 alkylsulfonyl;
X1、X2、X3、X4选自C,其中X1、X2、X3、X4与另外两个C(一个与O相连,另一个与CO相连)组成被0-4个R3取代的芳基;X 1 , X 2 , X 3 , X 4 are selected from C, wherein X 1 , X 2 , X 3 , X 4 and two other Cs (one is connected to O, the other is connected to CO) are composed of 0-4 R 3 substituted aryl;
R3选自卤素、羟基、氨基、氰基、硝基、C1-C6烷基、卤代C1-C6烷基、C1-C6烷氧基、卤代C1-C6烷氧基、C3-C6环烷基、C1-C6烷基氨基、卤代C1-C6烷基氨基、二(C1-C6烷基)氨基、卤代二(C1-C6烷基)氨基、C1-C6烷硫基、卤代C1-C6烷硫基、C2-C6烯基、C2-C6炔基、C2-C6烯氧基、卤代C2-C6烯氧基、C2-C6炔氧基、卤代C2-C6炔氧基、C1-C6烷基磺酰基、卤代C1-C6烷基磺酰基、C1-C6烷基羰基、卤代C1-C6烷基羰基、C1-C6烷氧基羰基、卤代C1-C6烷氧基羰基、C1-C6烷氧基C1-C6烷基、卤代C1-C6烷氧基C1-C6烷基、C1-C6烷硫基C1-C6烷基、卤代C1-C6烷硫基C1-C6烷基、C1-C6烷氧基羰基C1-C6烷基、卤代C1-C6烷氧基羰基C1-C6烷基、C1-C6烷硫基羰基C1-C6烷基、卤代C1-C6烷硫基羰基C1-C6烷基、C1-C6烷基羰基氧基、卤代C1-C6烷基羰基氧基、C1-C6烷氧基羰基氧基、卤代C1-C6烷氧基羰基氧基、C1-C6烷基磺酰基氧基、卤代C1-C6烷基磺酰基氧基、C1-C6烷氧基C1-C6烷氧基或卤代C1-C6烷氧基C1-C6烷氧基。R 3 is selected from halogen, hydroxy, amino, cyano, nitro, C 1 -C 6 alkyl, halogenated C 1 -C 6 alkyl, C 1 -C 6 alkoxy, halogenated C 1 -C 6 Alkoxy, C 3 -C 6 cycloalkyl, C 1 -C 6 alkylamino, halogenated C 1 -C 6 alkylamino, bis(C 1 -C 6 alkyl)amino, halogenated bis(C 1 -C 6 alkyl) amino, C 1 -C 6 alkylthio, halogenated C 1 -C 6 alkylthio, C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, C 2 -C 6 Alkenyloxy, halogenated C 2 -C 6 alkenyloxy, C 2 -C 6 alkynyloxy, halogenated C 2 -C 6 alkynyloxy, C 1 -C 6 alkylsulfonyl, halogenated C 1 - C 6 alkylsulfonyl, C 1 -C 6 alkylcarbonyl, halogenated C 1 -C 6 alkylcarbonyl, C 1 -C 6 alkoxycarbonyl, halogenated C 1 -C 6 alkoxycarbonyl, C 1 -C 6 alkoxy C 1 -C 6 alkyl, halogenated C 1 -C 6 alkoxy C 1 -C 6 alkyl, C 1 -C 6 alkylthio C 1 -C 6 alkyl, halogenated Substituted C 1 -C 6 alkylthio C 1 -C 6 alkyl, C 1 -C 6 alkoxycarbonyl C 1 -C 6 alkyl, halogenated C 1 -C 6 alkoxycarbonyl C 1 -C 6 Alkyl, C 1 -C 6 alkylthiocarbonyl C 1 -C 6 alkyl, halogenated C 1 -C 6 alkylthiocarbonyl C 1 -C 6 alkyl, C 1 -C 6 alkylcarbonyloxy, Halogenated C 1 -C 6 alkylcarbonyloxy, C 1 -C 6 alkoxycarbonyloxy, halogenated C 1 -C 6 alkoxycarbonyloxy, C 1 -C 6 alkylsulfonyloxy , halogenated C 1 -C 6 alkylsulfonyloxy, C 1 -C 6 alkoxy C 1 -C 6 alkoxy or halogenated C 1 -C 6 alkoxy C 1 -C 6 alkoxy .
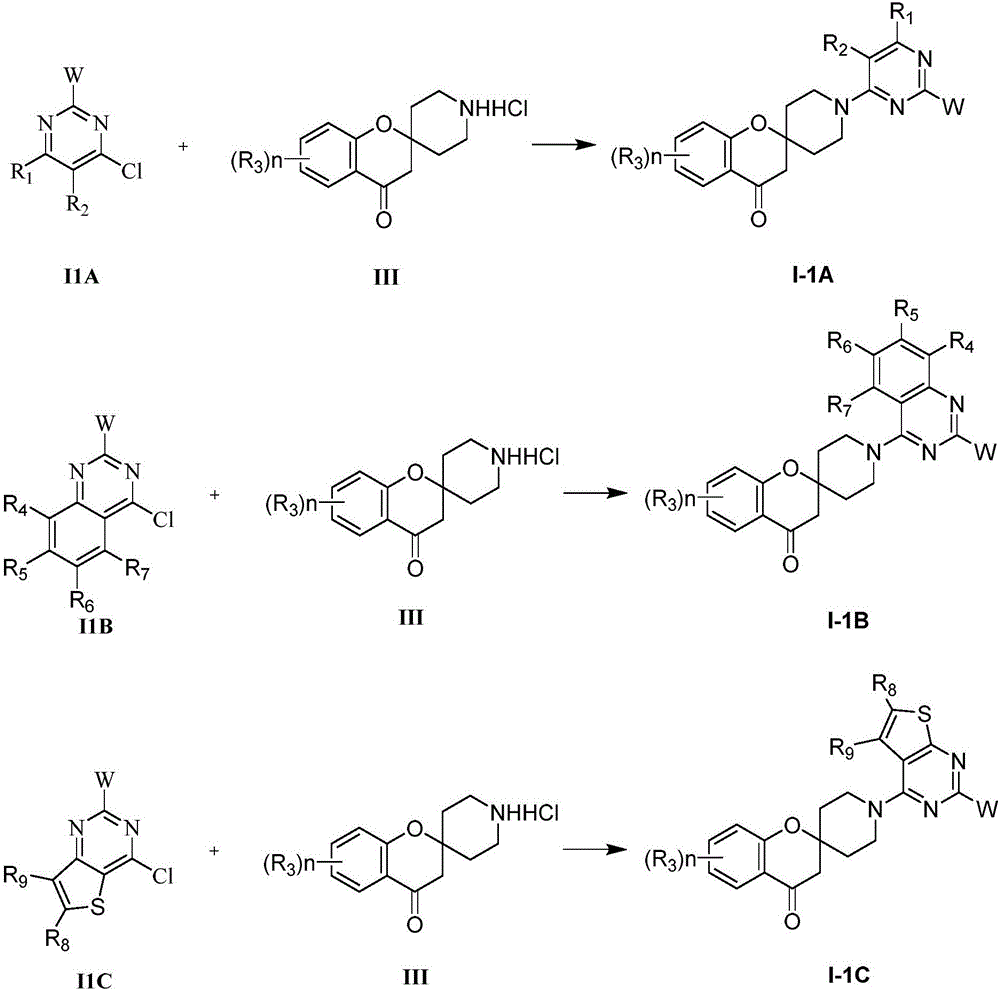
本发明含嘧啶的螺环类化合物中,进一步优选的化合物包括:所示通式I中X1、X2、X3、X4选自C,其中X1、X2、X3、X4与X1和X4分别相连的C(即,与X1相邻的C同时与O相连,与X4相邻的C同时与CO相连)组成被0-4个R3取代的芳基,通式I的结构式如I-1所示;Among the pyrimidine-containing spiro compounds of the present invention, further preferred compounds include: X 1 , X 2 , X 3 , X 4 in the general formula I are selected from C, wherein X 1 , X 2 , X 3 , X 4 The Cs that are connected to X and X, respectively ( that is, the C adjacent to X is connected to O at the same time, and the C adjacent to X is connected to CO at the same time ) constitutes an aryl group substituted by 0-4 Rs, The structural formula of general formula I is shown in I-1;
式中,R1选自氢、卤素、氰基、硝基、氨基、羧基、C1-C4烷基、卤代C1-C4烷基、C3-C4环烷基、C1-C4烷氧基、卤代C1-C4烷氧基、C1-C4烷硫基、C1-C4烷基亚磺酰基、C1-C4烷基磺酰基、卤代C1-C4烷硫基、C2-C4烯基、卤代C2-C4烯基、C2-C4炔基、卤代C2-C4炔基、C3-C4烯氧基、卤代C3-C4烯氧基、C3-C4炔氧基、卤代C3-C4炔氧基、C1-C4烷基氨基、二(C1-C4烷基)氨基、C1-C4烷基氨基羰基、卤代C1-C4烷基氨基羰基、C1-C4烷氧基羰基、卤代C1-C4烷氧基羰基、C1-C4烷氧基C1-C4烷基或C1-C4烷硫基C1-C4烷基;In the formula, R 1 is selected from hydrogen, halogen, cyano, nitro, amino, carboxyl, C 1 -C 4 alkyl, halogenated C 1 -C 4 alkyl, C 3 -C 4 cycloalkyl, C 1 -C 4 alkoxy, halogenated C 1 -C 4 alkoxy, C 1 -C 4 alkylthio, C 1 -C 4 alkylsulfinyl, C 1 -C 4 alkylsulfonyl, halogenated C 1 -C 4 alkylthio, C 2 -C 4 alkenyl, halogenated C 2 -C 4 alkenyl, C 2 -C 4 alkynyl, halogenated C 2 -C 4 alkynyl, C 3 -C 4 Alkenyloxy, halogenated C 3 -C 4 alkenyloxy, C 3 -C 4 alkynyloxy, halogenated C 3 -C 4 alkynyloxy, C 1 -C 4 alkylamino, bis(C 1 -C 4 alkyl) amino, C 1 -C 4 alkylaminocarbonyl, halogenated C 1 -C 4 alkylaminocarbonyl, C 1 -C 4 alkoxycarbonyl, halogenated C 1 -C 4 alkoxycarbonyl, C 1 -C 4 alkoxy C 1 -C 4 alkyl or C 1 -C 4 alkylthio C 1 -C 4 alkyl;
R2选自氢、卤素、氰基、硝基、氨基、羧基、甲酰基、C1-C4烷基、卤代C1-C4烷基、C1-C4烷氧基、卤代C1-C4烷氧基或二氧戊环基;R 2 is selected from hydrogen, halogen, cyano, nitro, amino, carboxyl, formyl, C 1 -C 4 alkyl, halogenated C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogenated C 1 -C 4 alkoxy or dioxolane;
R1和R2与相连的嘧啶环组成含C、N、O或S的五元或六元环;R 1 and R 2 and the connected pyrimidine ring form a five- or six-membered ring containing C, N, O or S;
W选自氢、卤素、C1-C4烷基、卤代C1-C4烷基、C3-C4环烷基、C1-C4烷氧基、C1-C4烷硫基或C1-C4烷基磺酰基;W is selected from hydrogen, halogen, C 1 -C 4 alkyl, halogenated C 1 -C 4 alkyl, C 3 -C 4 cycloalkyl, C 1 -C 4 alkoxy, C 1 -C 4 alkylthio group or C 1 -C 4 alkylsulfonyl;
R3选自卤素、羟基、氨基、氰基、硝基、C1-C4烷基、卤代C1-C4烷基、C1-C4烷氧基、卤代C1-C4烷氧基、C3-C4环烷基、C1-C4烷基氨基、卤代C1-C4烷基氨基、二(C1-C4烷基)氨基、卤代二(C1-C4烷基)氨基、C1-C4烷硫基、卤代C1-C4烷硫基、C2-C4烯基、C2-C4炔基、C2-C4烯氧基、卤代C2-C4烯氧基、C2-C4炔氧基、卤代C2-C4炔氧基、C1-C4烷基磺酰基、卤代C1-C4烷基磺酰基、C1-C4烷基羰基、卤代C1-C4烷基羰基、C1-C4烷氧基羰基、卤代C1-C4烷氧基羰基、C1-C4烷氧基C1-C4烷基、卤代C1-C4烷氧基C1-C4烷基、C1-C4烷硫基C1-C4烷基、卤代C1-C4烷硫基C1-C4烷基、C1-C4烷氧基羰基C1-C4烷基、卤代C1-C4烷氧基羰基C1-C4烷基、C1-C4烷硫基羰基C1-C4烷基、卤代C1-C4烷硫基羰基C1-C4烷基、C1-C4烷基羰基氧基、卤代C1-C4烷基羰基氧基、C1-C4烷氧基羰基氧基、卤代C1-C4烷氧基羰基氧基、C1-C4烷基磺酰基氧基、卤代C1-C4烷基磺酰基氧基、C1-C4烷氧基C1-C4烷氧基或卤代C1-C4烷氧基C1-C4烷氧基;R 3 is selected from halogen, hydroxy, amino, cyano, nitro, C 1 -C 4 alkyl, halogenated C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogenated C 1 -C 4 Alkoxy, C 3 -C 4 cycloalkyl, C 1 -C 4 alkylamino, halogenated C 1 -C 4 alkylamino, bis(C 1 -C 4 alkyl)amino, halogenated bis(C 1 -C 4 alkyl) amino, C 1 -C 4 alkylthio, halogenated C 1 -C 4 alkylthio, C 2 -C 4 alkenyl, C 2 -C 4 alkynyl, C 2 -C 4 Alkenyloxy, halogenated C 2 -C 4 alkenyloxy, C 2 -C 4 alkynyloxy, halogenated C 2 -C 4 alkynyloxy, C 1 -C 4 alkylsulfonyl, halogenated C 1 - C 4 alkylsulfonyl, C 1 -C 4 alkylcarbonyl, halogenated C 1 -C 4 alkylcarbonyl, C 1 -C 4 alkoxycarbonyl, halogenated C 1 -C 4 alkoxycarbonyl, C 1 -C 4 alkoxy C 1 -C 4 alkyl, halogenated C 1 -C 4 alkoxy C 1 -C 4 alkyl, C 1 -C 4 alkylthio C 1 -C 4 alkyl, halogenated Substituted C 1 -C 4 alkylthio C 1 -C 4 alkyl, C 1 -C 4 alkoxycarbonyl C 1 -C 4 alkyl, halogenated C 1 -C 4 alkoxycarbonyl C 1 -C 4 Alkyl, C 1 -C 4 alkylthiocarbonyl C 1 -C 4 alkyl, halogenated C 1 -C 4 alkylthiocarbonyl C 1 -C 4 alkyl, C 1 -C 4 alkylcarbonyloxy, Halogenated C 1 -C 4 alkylcarbonyloxy, C 1 -C 4 alkoxycarbonyloxy, halogenated C 1 -C 4 alkoxycarbonyloxy, C 1 -C 4 alkylsulfonyloxy , halogenated C 1 -C 4 alkylsulfonyloxy, C 1 -C 4 alkoxy C 1 -C 4 alkoxy or halogenated C 1 -C 4 alkoxy C 1 -C 4 alkoxy ;
本发明含嘧啶的螺环类化合物中,再进一步优选的化合物包括:所示通式I中:Among the pyrimidine-containing spiro compounds of the present invention, further preferred compounds include: in the general formula I shown:
通式I-1所示化合物的结构为:I-1A、I-1B、I-1C;The structure of the compound represented by general formula I-1 is: I-1A, I-1B, I-1C;
式中:where:
R1选自氢、卤素、氰基、硝基、氨基、羧基、C1-C4烷基、卤代C1-C4烷基、C3-C4环烷基、C1-C4烷氧基、卤代C1-C4烷氧基、C1-C4烷硫基、卤代C1-C4烷硫基、C1-C4烷基亚磺酰基、C1-C4烷基磺酰基、C2-C4烯基、卤代C2-C4烯基、C2-C4炔基、卤代C2-C4炔基、C3-C4烯氧基、卤代C3-C4烯氧基、C3-C4炔氧基、卤代C3-C4炔氧基、C1-C4烷基氨基、二(C1-C4烷基)氨基、C1-C4烷基氨基羰基、卤代C1-C4烷基氨基羰基、C1-C4烷氧基羰基、卤代C1-C4烷氧基羰基、C1-C4烷氧基C1-C4烷基或C1-C4烷硫基C1-C4烷基;R 1 is selected from hydrogen, halogen, cyano, nitro, amino, carboxyl, C 1 -C 4 alkyl, halogenated C 1 -C 4 alkyl, C 3 -C 4 cycloalkyl, C 1 -C 4 Alkoxy, halogenated C 1 -C 4 alkoxy, C 1 -C 4 alkylthio, halogenated C 1 -C 4 alkylthio, C 1 -C 4 alkylsulfinyl, C 1 -C 4Alkylsulfonyl , C2 - C4alkenyl , halogenated C2 - C4alkenyl , C2 - C4alkynyl , halogenated C2 - C4alkynyl , C3 - C4alkenyloxy , halogenated C 3 -C 4 alkenyloxy, C 3 -C 4 alkynyloxy, halogenated C 3 -C 4 alkynyloxy, C 1 -C 4 alkylamino, di(C 1 -C 4 alkyl ) amino, C 1 -C 4 alkylaminocarbonyl, halogenated C 1 -C 4 alkylaminocarbonyl, C 1 -C 4 alkoxycarbonyl, halogenated C 1 -C 4 alkoxycarbonyl, C 1 - C 4 alkoxy C 1 -C 4 alkyl or C 1 -C 4 alkylthio C 1 -C 4 alkyl;
R2选自氢、卤素、氰基、硝基、氨基、羧基、甲酰基、C1-C4烷基、卤代C1-C4烷基、C1-C4烷氧基或卤代C1-C4烷氧基、二氧戊环基;R 2 is selected from hydrogen, halogen, cyano, nitro, amino, carboxyl, formyl, C 1 -C 4 alkyl, halogenated C 1 -C 4 alkyl, C 1 -C 4 alkoxy or halo C 1 -C 4 alkoxy, dioxolane;
n选自0至4的整数,当n为0时,苯环上无取代基;当n大于1时,R3可相同或不同;n is selected from an integer from 0 to 4, when n is 0, there is no substituent on the benzene ring; when n is greater than 1, R can be the same or different;
R4、R5、R6或R7可相同或不同,分别选自氢、卤素、羟基、氨基、氰基、硝基、C1-C4烷基、卤代C1-C4烷基、C1-C4烷氧基、卤代C1-C4烷氧基或C3-C4环烷基;R 4 , R 5 , R 6 or R 7 , which may be the same or different, are independently selected from hydrogen, halogen, hydroxy, amino, cyano, nitro, C 1 -C 4 alkyl, halogenated C 1 -C 4 alkyl , C 1 -C 4 alkoxy, halogenated C 1 -C 4 alkoxy or C 3 -C 4 cycloalkyl;
R8、R9可相同或不同,分别选自氢、卤素、C1-C4烷基、卤代C1-C4烷基、C1-C4烷氧基、卤代C1-C4烷氧基、C1-C4烷硫基、卤代C1-C4烷硫基、C3-C4环烷基、未取代的或被1-5个R3取代的芳基、芳甲基、芳基羰基、芳甲基羰基、芳氧基羰基、杂芳基、杂芳甲基、杂芳基羰基、杂芳甲基羰基或杂芳氧基羰基;R 8 and R 9 may be the same or different, and are respectively selected from hydrogen, halogen, C 1 -C 4 alkyl, halogenated C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogenated C 1 -C 4 alkoxy, C 1 -C 4 alkylthio, halogenated C 1 -C 4 alkylthio, C 3 -C 4 cycloalkyl, unsubstituted or aryl substituted by 1-5 R 3 , arylmethyl, arylcarbonyl, arylmethylcarbonyl, aryloxycarbonyl, heteroaryl, heteroarylmethyl, heteroarylcarbonyl, heteroarylmethylcarbonyl or heteroaryloxycarbonyl;
W选自氢、卤素、C1-C4烷基、卤代C1-C4烷基、C3-C4环烷基、C1-C4烷氧基、C1-C4烷硫基或C1-C4烷基磺酰基;W is selected from hydrogen, halogen, C 1 -C 4 alkyl, halogenated C 1 -C 4 alkyl, C 3 -C 4 cycloalkyl, C 1 -C 4 alkoxy, C 1 -C 4 alkylthio group or C 1 -C 4 alkylsulfonyl;
R3选自卤素、羟基、氨基、氰基、硝基、C1-C4烷基、卤代C1-C4烷基、C1-C4烷氧基、卤代C1-C4烷氧基、C3-C4环烷基、C1-C4烷基氨基、卤代C1-C4烷基氨基、二(C1-C4烷基)氨基、卤代二(C1-C4烷基)氨基、C1-C4烷硫基、卤代C1-C4烷硫基、C2-C4烯基、C2-C4炔基、C2-C4烯氧基、卤代C2-C4烯氧基、C2-C4炔氧基、卤代C2-C4炔氧基、C1-C4烷基磺酰基、卤代C1-C4烷基磺酰基、C1-C4烷基羰基、卤代C1-C4烷基羰基、C1-C4烷氧基羰基、卤代C1-C4烷氧基羰基、C1-C4烷氧基C1-C4烷基、卤代C1-C4烷氧基C1-C4烷基、C1-C4烷硫基C1-C4烷基、卤代C1-C4烷硫基C1-C4烷基、C1-C4烷氧基羰基C1-C4烷基、卤代C1-C4烷氧基羰基C1-C4烷基、C1-C4烷硫基羰基C1-C4烷基、卤代C1-C4烷硫基羰基C1-C4烷基、C1-C4烷基羰基氧基、卤代C1-C4烷基羰基氧基、C1-C4烷氧基羰基氧基、卤代C1-C4烷氧基羰基氧基、C1-C4烷基磺酰基氧基、卤代C1-C4烷基磺酰基氧基、C1-C4烷氧基C1-C4烷氧基或卤代C1-C4烷氧基C1-C4烷氧基;R 3 is selected from halogen, hydroxy, amino, cyano, nitro, C 1 -C 4 alkyl, halogenated C 1 -C 4 alkyl, C 1 -C 4 alkoxy, halogenated C 1 -C 4 Alkoxy, C 3 -C 4 cycloalkyl, C 1 -C 4 alkylamino, halogenated C 1 -C 4 alkylamino, bis(C 1 -C 4 alkyl)amino, halogenated bis(C 1 -C 4 alkyl) amino, C 1 -C 4 alkylthio, halogenated C 1 -C 4 alkylthio, C 2 -C 4 alkenyl, C 2 -C 4 alkynyl, C 2 -C 4 Alkenyloxy, halogenated C 2 -C 4 alkenyloxy, C 2 -C 4 alkynyloxy, halogenated C 2 -C 4 alkynyloxy, C 1 -C 4 alkylsulfonyl, halogenated C 1 - C 4 alkylsulfonyl, C 1 -C 4 alkylcarbonyl, halogenated C 1 -C 4 alkylcarbonyl, C 1 -C 4 alkoxycarbonyl, halogenated C 1 -C 4 alkoxycarbonyl, C 1 -C 4 alkoxy C 1 -C 4 alkyl, halogenated C 1 -C 4 alkoxy C 1 -C 4 alkyl, C 1 -C 4 alkylthio C 1 -C 4 alkyl, halogenated Substituted C 1 -C 4 alkylthio C 1 -C 4 alkyl, C 1 -C 4 alkoxycarbonyl C 1 -C 4 alkyl, halogenated C 1 -C 4 alkoxycarbonyl C 1 -C 4 Alkyl, C 1 -C 4 alkylthiocarbonyl C 1 -C 4 alkyl, halogenated C 1 -C 4 alkylthiocarbonyl C 1 -C 4 alkyl, C 1 -C 4 alkylcarbonyloxy, Halogenated C 1 -C 4 alkylcarbonyloxy, C 1 -C 4 alkoxycarbonyloxy, halogenated C 1 -C 4 alkoxycarbonyloxy, C 1 -C 4 alkylsulfonyloxy , halogenated C 1 -C 4 alkylsulfonyloxy, C 1 -C 4 alkoxy C 1 -C 4 alkoxy or halogenated C 1 -C 4 alkoxy C 1 -C 4 alkoxy ;
本发明含嘧啶的螺环类化合物中,又进一步优选的化合物包括:通式I-1A、I-1B、I-1C所示化合物,式中:Among the pyrimidine-containing spiro compounds of the present invention, further preferred compounds include: compounds represented by general formulas I-1A, I-1B, and I-1C, in which:
R1选自氢、氟、氯、溴、碘、氰基、硝基、氨基、羧基、甲基、乙基、正丙基、异丙基、正丁基、仲丁基、异丁基、叔丁基、一氟甲基、一氯甲基、二氟甲基、三氟甲基、三氟乙基、甲氧基甲基、乙氧基甲基或三氟乙氧基甲基;R 1 is selected from hydrogen, fluorine, chlorine, bromine, iodine, cyano, nitro, amino, carboxyl, methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, monofluoromethyl, monochloromethyl, difluoromethyl, trifluoromethyl, trifluoroethyl, methoxymethyl, ethoxymethyl or trifluoroethoxymethyl;
R2选自氢、氟、氯、溴、碘、氰基、硝基、氨基、羧基、甲酰基、甲基、乙基、甲氧基、乙氧基或三氟乙氧基、二氧戊环基;R 2 is selected from hydrogen, fluorine, chlorine, bromine, iodine, cyano, nitro, amino, carboxyl, formyl, methyl, ethyl, methoxy, ethoxy or trifluoroethoxy, dioxolane ring base;
n选自0至4的整数,当n为0时,苯环上无取代基;当n大于1时,R3可相同或不同;n is selected from an integer from 0 to 4, when n is 0, there is no substituent on the benzene ring; when n is greater than 1, R can be the same or different;
R4、R5、R6或R7可相同或不同,分别选自氢、氟、氯、溴、碘、氨基、氰基、硝基、甲基、乙基、正丙基、异丙基、正丁基、仲丁基、异丁基、叔丁基、三氟甲基、三氯甲基、二氟一氯甲基、二氯一氟甲基、甲氧基、乙氧基、正丙氧基、异丙氧基、正丁氧基、仲丁氧基、异丁氧基、叔丁氧基、三氟甲氧基或三氟乙氧基;R 4 , R 5 , R 6 or R 7 may be the same or different and are independently selected from hydrogen, fluorine, chlorine, bromine, iodine, amino, cyano, nitro, methyl, ethyl, n-propyl, isopropyl , n-butyl, sec-butyl, isobutyl, tert-butyl, trifluoromethyl, trichloromethyl, difluoro-chloromethyl, dichloro-fluoromethyl, methoxy, ethoxy, n- propoxy, isopropoxy, n-butoxy, sec-butoxy, isobutoxy, tert-butoxy, trifluoromethoxy or trifluoroethoxy;
R8、R9可相同或不同,分别选自氢、氟、氯、溴、碘、甲基、乙基、正丙基、异丙基、正丁基、仲丁基、异丁基、叔丁基、三氟甲基、三氯甲基、二氟一氯甲基、二氯一氟甲基、三氟乙基、甲氧基、乙氧基、正丙氧基、异丙氧基、正丁氧基、仲丁氧基、异丁氧基、叔丁氧基、三氟甲氧基、三氟乙氧基、未取代的或被1-5个R3取代的芳基、芳甲基、芳基羰基、芳甲基羰基、芳氧基羰基、杂芳基、杂芳甲基、杂芳基羰基、杂芳甲基羰基或杂芳氧基羰基;R 8 and R 9 can be the same or different, and are respectively selected from hydrogen, fluorine, chlorine, bromine, iodine, methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl Butyl, trifluoromethyl, trichloromethyl, difluoro-chloromethyl, dichloro-fluoromethyl, trifluoroethyl, methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, sec-butoxy, isobutoxy, tert-butoxy, trifluoromethoxy, trifluoroethoxy, unsubstituted or substituted with 1-5 R 3 aryl, aryl methyl aryl, arylcarbonyl, arylmethylcarbonyl, aryloxycarbonyl, heteroaryl, heteroarylmethyl, heteroarylcarbonyl, heteroarylmethylcarbonyl, or heteroaryloxycarbonyl;
W选自氢、氟、氯、溴、碘、甲基、乙基、正丙基、异丙基、正丁基、仲丁基、异丁基、叔丁基、一氟甲基、一氯甲基、二氟甲基、三氟甲基、三氟乙基、甲氧基、乙氧基、甲硫基、乙硫基、甲基磺酰基或乙基磺酰基;W is selected from hydrogen, fluorine, chlorine, bromine, iodine, methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, monofluoromethyl, monochloro methyl, difluoromethyl, trifluoromethyl, trifluoroethyl, methoxy, ethoxy, methylthio, ethylthio, methylsulfonyl or ethylsulfonyl;
R3选自氟、氯、溴、碘、氰基、氨基、硝基、甲基、乙基、正丙基、异丙基、正丁基、仲丁基、异丁基、叔丁基、三氟甲基、三氯甲基、二氟一氯甲基、二氯一氟甲基、甲氧基、乙氧基、正丙氧基、异丙氧基、正丁氧基、仲丁氧基、异丁氧基、叔丁氧基、三氟甲氧基、三氟乙氧基、甲氧基羰基、乙氧基羰基、氨基羰基、甲基氨基羰基、乙基氨基羰基、二甲氨基羰基或丙炔氧基; R is selected from fluorine, chlorine, bromine, iodine, cyano, amino, nitro, methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, Trifluoromethyl, trichloromethyl, difluoro-chloromethyl, dichloro-fluoromethyl, methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, sec-butoxy group, isobutoxy, tert-butoxy, trifluoromethoxy, trifluoroethoxy, methoxycarbonyl, ethoxycarbonyl, aminocarbonyl, methylaminocarbonyl, ethylaminocarbonyl, dimethylamino carbonyl or propynyloxy;
本发明含嘧啶的螺环类化合物中,更进一步优选的化合物包括:所述通式I-1A、I-1B、I-1C所示化合物,式中Among the pyrimidine-containing spiro compounds of the present invention, further preferred compounds include: the compounds represented by the general formulae I-1A, I-1B, and I-1C, wherein
R1选自氢、氟、氯、溴、碘、甲基、乙基或二氟甲基;R 1 is selected from hydrogen, fluorine, chlorine, bromine, iodine, methyl, ethyl or difluoromethyl;
R2选自氢、氟、氯、溴、碘、硝基、氨基、甲酰基、甲基、乙基、甲氧基、乙氧基或二氧戊环基;R 2 is selected from hydrogen, fluorine, chlorine, bromine, iodine, nitro, amino, formyl, methyl, ethyl, methoxy, ethoxy or dioxolane;
n选自0至4的整数,当n为0时,苯环上无取代基;当n大于1时,R3可相同或不同;n is selected from an integer from 0 to 4, when n is 0, there is no substituent on the benzene ring; when n is greater than 1, R can be the same or different;
R4、R5、R6或R7可相同或不同,分别选自氢、氟、氯、溴、碘或甲基;R 4 , R 5 , R 6 or R 7 may be the same or different and are independently selected from hydrogen, fluorine, chlorine, bromine, iodine or methyl;
R8、R9可相同或不同,分别选自氢、氟、氯、溴或碘;R 8 and R 9 may be the same or different, and are respectively selected from hydrogen, fluorine, chlorine, bromine or iodine;
W选自氢、氟、氯、溴、碘或甲基;W is selected from hydrogen, fluorine, chlorine, bromine, iodine or methyl;
R3选自氟、氯、溴、碘、氰基、硝基、甲基、三氟甲基、甲氧基、三氟甲氧基或丙炔氧基; R is selected from fluorine, chlorine, bromine, iodine, cyano, nitro, methyl, trifluoromethyl, methoxy, trifluoromethoxy or propynyloxy;
本发明含嘧啶的螺环类化合物中,再更进一步优选的化合物包括:Among the pyrimidine-containing spiro compounds of the present invention, further preferred compounds include:
通式I-1A、I-1B、I-1C所示化合物,式中Compounds represented by general formulas I-1A, I-1B and I-1C, in which
R1选自氟、氯、溴、碘、甲基、乙基或二氟甲基;R 1 is selected from fluorine, chlorine, bromine, iodine, methyl, ethyl or difluoromethyl;
R2选自氟、氯、溴、碘、硝基、氨基、甲酰基、甲基或甲氧基、二氧戊环基;R 2 is selected from fluorine, chlorine, bromine, iodine, nitro, amino, formyl, methyl or methoxy, dioxolane;
n选自1至4的整数,当n大于1时,R3可相同或不同;n is selected from an integer from 1 to 4, and when n is greater than 1, R can be the same or different;
R4、R5、R6、R7均选自氢;R 4 , R 5 , R 6 and R 7 are all selected from hydrogen;
R8选自氢;R 8 is selected from hydrogen;
R9选自氯;R 9 is selected from chlorine;
W选自氢、氟、氯、溴或碘;W is selected from hydrogen, fluorine, chlorine, bromine or iodine;
R3选自氟、氯、溴、碘、氰基、硝基、甲基、三氟甲基、甲氧基、三氟甲氧基或丙炔氧基; R is selected from fluorine, chlorine, bromine, iodine, cyano, nitro, methyl, trifluoromethyl, methoxy, trifluoromethoxy or propynyloxy;
一种含嘧啶的螺环类化合物在农业或其他领域中用作制备杀菌剂药物的用途。A pyrimidine-containing spiro compound is used for preparing fungicides in agriculture or other fields.
一种杀菌组合物,含所述的含嘧啶的螺环类化合物作为活性组分;其中,组合物中活性组分的重量百分含量为0.1-99%。A bactericidal composition contains the pyrimidine-containing spiro compound as an active component; wherein, the weight percentage of the active component in the composition is 0.1-99%.
一种组合物在农业或其他领域中防治病菌的用途。Use of a composition for controlling pathogens in agriculture or other fields.
上面给出的通式I化合物的定义中,汇集所用术语一般定义如下:In the definitions of compounds of general formula I given above, the terms used collectively are generally defined as follows:
卤素:指氟、氯、溴或碘。烷基:直链或支链烷基,例如甲基、乙基、丙基、异丙基、正丁基或叔丁基。环烷基:取代或未取代的环状烷基,例如环丙基、环戊基或环己基。取代基如甲基、卤素等。卤代烷基:直链或支链烷基,在这些烷基上的氢原子可部分或全部被卤原子所取代,例如,氯甲基、二氯甲基、三氯甲基、氟甲基、二氟甲基、三氟甲基等。烷基亚磺酰基:直链或支链烷基经亚磺酰基(-SO-)连接到结构上,如甲基亚磺酰基。卤代烷基亚磺酰基:直链或支链烷基亚磺酰基,其烷基上的氢原子可部分或全部被卤原子所取代。卤代烷基磺酰基:直链或支链烷基磺酰基,其烷基上的氢原子可部分或全部被卤原子所取代。烷基氨基硫基:如CH3NHS-、C2H5NHS-。二烷基氨基硫基:如(CH3)2NS-、(C2H5)2NS-。烷基氨基磺酰基:烷基-NH-SO2-。二烷基氨基磺酰基:(烷基)2-N-SO2-。烷基磺酰基氨基羰基:烷基-SO2-NH-CO-。烷基羰基氨基磺酰基:烷基-CO-NH-SO2-。烷基羰基烷基:烷基-CO-烷基-。烷基磺酰基氧基:烷基-S(O)2-O-。卤代烷基磺酰基氧基:烷基磺酰基氧基的烷基上的氢原子可部分或全部被卤原子所取代,例如CF3-SO2-O。环烷基氧基羰基:如环丙氧基羰基、环己氧基羰基等。烷氧基:直链或支链烷基,经氧原子键连接到结构上。卤代烷氧基:直链或支链烷氧基,在这些烷氧基上的氢原子可部分或全部被卤原子所取代。例如,氯甲氧基、二氯甲氧基、三氯甲氧基、氟甲氧基、二氟甲氧基、三氟甲氧基、氯氟甲氧基、三氟乙氧基等。卤代烷氧基羰基:烷氧基羰基的烷基上的氢原子可部分或全部被卤原子所取代,如ClCH2CH2OCO-、CF3CH2OCO-等。烷氧基烷基:烷基-O-烷基-,例如CH3OCH2-。卤代烷氧基烷基:烷氧基烷基的烷基上的氢原子可部分或全部被卤原子所取代,如ClCH2CH2OCH2-、CF3CH2OCH2-等。烷氧基羰基烷基:烷氧基羰基-烷基-,例如CH3OCOCH2-。卤代烷氧基羰基烷基:烷氧基羰基烷基的烷基上的氢原子可部分或全部被卤原子所取代,例如CF3CH2OCOCH2-。烷基羰基氧基:如CH3COO-等。卤代烷基羰基氧基:烷基羰基氧基的氢原子可部分或全部被卤原子所取代,如CF3COO-等。烷氧基羰基氧基:烷氧基羰基-氧基-,例如CH3OCOO-。卤代烷氧基羰基氧基:烷氧基羰基氧基的烷基上的氢原子可部分或全部被卤原子所取代,例如CF3OCOO-。烷硫基羰基烷基:烷硫基羰基-烷基-,例如CH3SCOCH2-。卤代烷硫基羰基烷基:烷硫基羰基烷基的烷基上的氢原子可部分或全部被卤原子所取代,例如CF3CH2SCOCH2-。烷氧基烷氧基:如CH3OCH2O-等。卤代烷氧基烷氧基:烷氧基上的氢原子可部分或全部被卤原子所取代,例如CF3OCH2O-。烷氧基烷氧基羰基:如CH3OCH2CH2OCO-等。烷硫基:直链或支链烷基,经硫原子键连接到结构上。卤代烷硫基:直链或支链烷硫基,在这些烷基上的氢原子可部分或全部被卤原子所取代。例如,氯甲硫基、二氯甲硫基、三氯甲硫基、氟甲硫基、二氟甲硫基、三氟甲硫基、氯氟甲硫基等。烷硫基烷基:烷基-S-烷基-,例如CH3SCH2-。卤代烷硫基烷基:烷硫基烷基的烷基上的氢原子可部分或全部被卤原子所取代,如ClCH2CH2SCH2-、CF3CH2SCH2-等。烷基氨基:直链或支链烷基,经氮原子键连接到结构上。卤代烷基氨基:直链或支链烷基氨基,在这些烷基上的氢原子可部分或全部被卤原子所取代。二烷基氨基:如(CH3)2N-,(CH3CH2)2N-。卤代二烷基氨基:烷基上的氢原子可部分或全部被卤原子所取代,如(CF3)2N-,(CF3CH2)2N-。烯基:直链或支链烯类,例如乙烯基、1-丙烯基、2-丙烯基和不同的丁烯基、戊烯基和己烯基异构体。烯基还包括多烯类,如1,2-丙二烯基和2,4-己二烯基。卤代烯基:直链或支链烯类,在这些烯基上的氢原子可部分或全部被卤原子所取代。烯氧基:直链或支链烯类,经氧原子键连接到结构上。卤代烯氧基:直链或支链烯氧基,在这些烯氧基上的氢原子可部分或全部被卤原子所取代。烯基硫基:直链或支链烯类,经硫原子键连接到结构上。如CH2=CHCH2S-。烯氧基羰基:如CH2=CHCH2OCO-等。炔基:直链或支链炔类,例如乙炔基、1-丙炔基、2-丙炔基和不同的丁炔基、戊炔基和己炔基异构体。炔基还包括由多个三键组成的基团,如2,5-己二炔基。卤代炔基:直链或支链炔类,在这些炔基上的氢原子可部分或全部被卤原子所取代。炔氧基:直链或支链炔类,经氧原子键连接到结构上。卤代炔氧基:直链或支链炔氧基,在这些炔氧基上的氢原子可部分或全部被卤原子所取代。炔氧基羰基:如CH≡CCH2OCO-等。烷基磺酰基:直链或支链烷基经磺酰基(-SO2-)连接到结构上,如甲基磺酰基。卤代烷基磺酰基:直链或支链烷基磺酰基,其烷基上的氢原子可部分或全部被卤原子所取代。烷基羰基:烷基经羰基连接到结构上,如CH3CO-,CH3CH2CO-。卤代烷基羰基:烷基羰基的烷基上的氢原子可部分或全部被卤原子所取代,如CF3CO-。烷氧基羰基:烷氧基经羰基连接到结构上。如CH3OCO-,CH3CH2OCO-。氨基羰基:如NH2CO-。烷基氨基羰基:烷基-NH-CO-,例如CH3NHCO-,CH3CH2NHCO-。二烷基氨基羰基:如(CH3)2NCO-,(CH3CH2)2NCO-。(杂)芳基、(杂)芳基烷基、(杂)芳基羰基、(杂)芳甲基羰基、(杂)芳基羰基烷基、(杂)芳氧基羰基、(杂)芳基烷基氧基羰基中的芳基部分包括苯基或萘基等。杂芳基是含1个或多个N杂原子的五元环或六元环。例如吡啶基、嘧啶基、吡嗪基、哒嗪基、三嗪基等。(杂)芳基:如苯基等。Halogen: refers to fluorine, chlorine, bromine or iodine. Alkyl: straight or branched chain alkyl such as methyl, ethyl, propyl, isopropyl, n-butyl or tert-butyl. Cycloalkyl: A substituted or unsubstituted cyclic alkyl group such as cyclopropyl, cyclopentyl or cyclohexyl. Substituents such as methyl, halogen and the like. Haloalkyl: straight-chain or branched-chain alkyl groups in which hydrogen atoms may be partially or fully substituted by halogen atoms, e.g., chloromethyl, dichloromethyl, trichloromethyl, fluoromethyl, dichloromethyl Fluoromethyl, trifluoromethyl, etc. Alkylsulfinyl: A straight or branched chain alkyl group is attached to the structure via a sulfinyl group (-SO-), such as methylsulfinyl. Halogenated alkylsulfinyl: a straight-chain or branched-chain alkylsulfinyl group in which the hydrogen atoms on the alkyl group may be partially or fully substituted by halogen atoms. Haloalkylsulfonyl: A straight-chain or branched-chain alkylsulfonyl group in which the hydrogen atoms on the alkyl group may be partially or fully substituted by halogen atoms. Alkylaminothio: such as CH 3 NHS-, C 2 H 5 NHS-. Dialkylaminothio: such as (CH 3 ) 2 NS-, (C 2 H 5 ) 2 NS-. Alkylaminosulfonyl: alkyl-NH- SO2- . Dialkylaminosulfonyl: (alkyl) 2 -N- SO2- . Alkylsulfonylaminocarbonyl: alkyl-SO2 - NH-CO-. Alkylcarbonylaminosulfonyl: alkyl-CO-NH- SO2- . Alkylcarbonylalkyl: alkyl-CO-alkyl-. Alkylsulfonyloxy: alkyl-S(O) 2 -O-. Haloalkylsulfonyloxy: The hydrogen atoms on the alkyl group of an alkylsulfonyloxy group may be partially or fully substituted with halogen atoms, eg CF3 -SO2 - O. Cycloalkyloxycarbonyl: such as cyclopropoxycarbonyl, cyclohexyloxycarbonyl, etc. Alkoxy: A straight or branched chain alkyl group attached to the structure through an oxygen atom bond. Halogenated alkoxy: a straight-chain or branched alkoxy group, and the hydrogen atoms on these alkoxy groups may be partially or totally substituted by halogen atoms. For example, chloromethoxy, dichloromethoxy, trichloromethoxy, fluoromethoxy, difluoromethoxy, trifluoromethoxy, chlorofluoromethoxy, trifluoroethoxy and the like. Halogenated alkoxycarbonyl: The hydrogen atoms on the alkyl group of alkoxycarbonyl can be partially or completely substituted by halogen atoms, such as ClCH 2 CH 2 OCO-, CF 3 CH 2 OCO- and the like. Alkoxyalkyl: alkyl-O - alkyl-, eg CH3OCH2- . Halogenated alkoxyalkyl group: The hydrogen atoms on the alkyl group of the alkoxyalkyl group may be partially or completely replaced by halogen atoms, such as ClCH 2 CH 2 OCH 2 -, CF 3 CH 2 OCH 2 - and the like. Alkoxycarbonylalkyl: alkoxycarbonyl - alkyl-, eg CH3OCOCH2- . Halogenated alkoxycarbonylalkyl : The hydrogen atoms on the alkyl group of an alkoxycarbonylalkyl group may be partially or fully substituted by halogen atoms, eg CF3CH2OCOCH2- . Alkylcarbonyloxy: such as CH 3 COO-, etc. Haloalkylcarbonyloxy: The hydrogen atoms of alkylcarbonyloxy may be partially or completely replaced by halogen atoms, such as CF 3 COO- and the like. Alkoxycarbonyloxy: alkoxycarbonyl-oxy-, eg CH3OCOO- . Halogenated alkoxycarbonyloxy: The hydrogen atoms on the alkyl group of an alkoxycarbonyloxy group may be partially or totally substituted by halogen atoms, eg CF 3 OCOO-. Alkylthiocarbonylalkyl: alkylthiocarbonyl - alkyl-, eg CH3SCOCH2- . Haloalkylthiocarbonylalkyl : The hydrogen atoms on the alkyl group of an alkylthiocarbonylalkyl group may be partially or fully substituted by halogen atoms, eg CF3CH2SCOCH2- . Alkoxyalkoxy: such as CH 3 OCH 2 O-, etc. Haloalkoxyalkoxy: The hydrogen atoms on the alkoxy group may be partially or totally substituted by halogen atoms, eg CF3OCH2O- . Alkoxyalkoxycarbonyl: such as CH 3 OCH 2 CH 2 OCO-, etc. Alkylthio: A straight or branched chain alkyl group attached to the structure via a sulfur atom bond. Haloalkylthio: straight-chain or branched-chain alkylthio, the hydrogen atoms on these alkyl groups may be partially or totally substituted by halogen atoms. For example, chloromethylthio, dichloromethylthio, trichloromethylthio, fluoromethylthio, difluoromethylthio, trifluoromethylthio, chlorofluoromethylthio and the like. Alkylthioalkyl: alkyl-S - alkyl-, eg CH3SCH2- . Halogenated alkylthioalkyl: The hydrogen atoms on the alkyl group of the alkylthioalkyl group may be partially or completely substituted by halogen atoms, such as ClCH 2 CH 2 SCH 2 -, CF 3 CH 2 SCH 2 - and the like. Alkylamino: A straight or branched chain alkyl group attached to the structure through a nitrogen atom bond. Haloalkylamino: straight-chain or branched-chain alkylamino, the hydrogen atoms on these alkyl groups may be partially or fully substituted by halogen atoms. Dialkylamino: such as (CH 3 ) 2 N-, (CH 3 CH 2 ) 2 N-. Halogenated dialkylamino: The hydrogen atoms on the alkyl group may be partially or completely replaced by halogen atoms, such as (CF 3 ) 2 N-, (CF 3 CH 2 ) 2 N-. Alkenyl: Linear or branched alkenes such as vinyl, 1-propenyl, 2-propenyl and the different butenyl, pentenyl and hexenyl isomers. Alkenyl also includes polyenes such as 1,2-propadienyl and 2,4-hexadienyl. Halogenated alkenyl: straight or branched chain alkenes, the hydrogen atoms in these alkenyl groups may be partially or completely replaced by halogen atoms. Alkenyloxy: Straight or branched chain alkenes, bonded to the structure through an oxygen atom. Haloalkenyloxy: straight-chain or branched-chain alkenyloxy, the hydrogen atoms on these alkenyloxy groups may be partially or totally substituted by halogen atoms. Alkenylthio: Straight or branched chain alkenes attached to the structure through a sulfur atom bond. Such as CH 2 =CHCH 2 S-. Alkenyloxycarbonyl: such as CH 2 =CHCH 2 OCO- and the like. Alkynyl: straight or branched chain alkynes such as ethynyl, 1-propynyl, 2-propynyl and the different butynyl, pentynyl and hexynyl isomers. Alkynyl also includes groups composed of multiple triple bonds, such as 2,5-hexadiynyl. Halogenated alkynyl: straight or branched chain alkynes in which the hydrogen atoms on these alkynyl groups may be partially or completely replaced by halogen atoms. Alkynoxy: Straight or branched chain alkynes that are bonded to the structure through an oxygen atom. Halogenated alkynyloxy: straight-chain or branched alkynyloxy, the hydrogen atoms on these alkynyloxy groups may be partially or totally substituted by halogen atoms. Alkynyloxycarbonyl: such as CH≡CCH 2 OCO-, etc. Alkylsulfonyl: A straight or branched chain alkyl group is attached to the structure via a sulfonyl group ( -SO2- ), such as methylsulfonyl. Haloalkylsulfonyl: A straight-chain or branched-chain alkylsulfonyl group in which the hydrogen atoms on the alkyl group may be partially or fully substituted by halogen atoms. Alkylcarbonyl : An alkyl group is attached to a structure via a carbonyl group, eg CH3CO- , CH3CH2CO- . Haloalkylcarbonyl: The hydrogen atoms on the alkyl group of an alkylcarbonyl group may be partially or completely replaced by halogen atoms, such as CF 3 CO-. Alkoxycarbonyl: An alkoxy group is attached to the structure through a carbonyl group. Such as CH 3 OCO-, CH 3 CH 2 OCO-. Aminocarbonyl: such as NH 2 CO-. Alkylaminocarbonyl: alkyl-NH - CO-, eg CH3NHCO- , CH3CH2NHCO- . Dialkylaminocarbonyl: such as (CH 3 ) 2 NCO-, (CH 3 CH 2 ) 2 NCO-. (hetero)aryl, (hetero)arylalkyl, (hetero)arylcarbonyl, (hetero)arylmethylcarbonyl, (hetero)arylcarbonylalkyl, (hetero)aryloxycarbonyl, (hetero)aryl The aryl moiety in the alkoxycarbonyl group includes phenyl or naphthyl, and the like. Heteroaryl is a five- or six-membered ring containing one or more N heteroatoms. For example, pyridyl, pyrimidinyl, pyrazinyl, pyridazinyl, triazinyl and the like. (Hetero)aryl: such as phenyl and the like.
表1、表2、表3分别列举了通式I中R1、R2和W的部分具体取代基,但它们并非仅限于这些取代基。Table 1, Table 2 and Table 3 respectively list some specific substituents of R 1 , R 2 and W in the general formula I, but they are not limited to these substituents.
表1 R1取代基Table 1 R 1 Substituents
表2 R2取代基Table 2 R 2 Substituents
表3 W取代基Table 3 W Substituents
本发明的部分化合物可以用表4-表17中列出的具体化合物来说明,但并不限定本发明。表中涉及的通式化合物I-1A、I-1B、I-1C中,W=R4=R5=R6=R7=R8=H。Part of the compounds of the present invention can be illustrated by the specific compounds listed in Table 4 to Table 17, but the present invention is not limited. In the general formula compounds I-1A, I-1B and I-1C mentioned in the table, W=R 4 =R 5 =R 6 =R 7 =R 8 =H.
通式I-1A中,In general formula I-1A,
当R1=Cl、R2=H时,(R3)n取代基见表4,代表化合物编号依次为4-(1)-4-(279)。When R 1 =Cl, R 2 =H, the (R 3 ) n substituents are shown in Table 4, and the compound numbers are 4-(1)-4-(279).
表4Table 4
表5:通式I-1A中,当R1=Cl、R2=CH3时,取代基(R3)n与表4一致,依次对应表4的4-1-4-279,代表化合物编号依次为5-(1)-5-(279)。Table 5: In the general formula I-1A, when R 1 =Cl, R 2 =CH 3 , the substituent (R 3 ) n is the same as that in Table 4, corresponding to 4-1-4-279 of Table 4 in turn, representing the compound The numbers are 5-(1)-5-(279).
表6:通式I-1A中,当R1=Cl、R2=OCH3时,取代基(R3)n与表4一致,依次对应表4的4-1-9-279,代表化合物编号依次为6-(1)-6-(279)。Table 6: In general formula I-1A, when R 1 =Cl, R 2 =OCH 3 , the substituent (R 3 ) n is the same as that in Table 4, corresponding to 4-1-9-279 of Table 4 in turn, representing the compound The numbers are 6-(1)-6-(279).
表7:通式I-1A中,当R1=Cl、R2=CHO时,取代基(R3)n与表4一致,依次对应表4的4-1-4-279,代表化合物编号依次为7-(1)-7-279。Table 7: In the general formula I-1A, when R 1 =Cl, R 2 =CHO, the substituent (R 3 ) n is the same as that in Table 4, corresponding to 4-1-4-279 in Table 4 in turn, representing the compound number The sequence is 7-(1)-7-279.
表8:通式I-1A中,当R1=Cl、R2=Br时,取代基(R3)n与表4一致,依次对应表4的4-1-4-279,代表化合物编号依次为8-(1)-8-(279)。Table 8: In the general formula I-1A, when R 1 =Cl, R 2 =Br, the substituent (R 3 ) n is the same as that in Table 4, corresponding to 4-1-4-279 in Table 4 in turn, representing the compound number The sequence is 8-(1)-8-(279).
表9:通式I-1A中,当R1=Cl、R2=NH2时,取代基(R3)n与表4一致,依次对应表4的4-1-4-279,代表化合物编号依次为9-(1)-9-(279)。Table 9: In the general formula I-1A, when R 1 =Cl, R 2 =NH 2 , the substituent (R 3 ) n is the same as that in Table 4, corresponding to 4-1-4-279 in Table 4 in turn, representing the compound The numbers are 9-(1)-9-(279).
表10:通式I-1A中,当R1=Cl、R2=NO2时,取代基(R3)n与表4一致,依次对应表4的4-1-4-279,代表化合物编号依次为10-(1)-10-(279)。Table 10: In the general formula I-1A, when R 1 =Cl, R 2 =NO 2 , the substituent (R 3 ) n is consistent with Table 4, corresponding to 4-1-4-279 of Table 4 in turn, representing the compound The numbers are 10-(1)-10-(279).
表11:通式I-1A中,当R1=Cl、R2=4-Br-Ph时,取代基(R3)n与表4一致,依次对应表4的4-1-4-279,代表化合物编号依次为11-(1)-11-(279)。Table 11: In general formula I-1A, when R 1 =Cl, R 2 =4-Br-Ph, the substituent (R 3 ) n is consistent with Table 4, corresponding to 4-1-4-279 of Table 4 in turn , and the compound numbers are 11-(1)-11-(279).
表12:通式I-1A中,当R1=Cl、R2=COOH时,取代基(R3)n与表4一致,依次对应表4的4-1-4-279,代表化合物编号依次为12-(1)-12-(279)。Table 12: In general formula I-1A, when R 1 =Cl, R 2 =COOH, the substituent (R 3 ) n is consistent with Table 4, corresponding to 4-1-4-279 of Table 4 in turn, representing the compound number The sequence is 12-(1)-12-(279).
表13:通式I-1A中,当R1=CH3、R2=Cl时,取代基(R3)n与表4一致,依次对应表4的4-1-4-279,代表化合物编号依次为13-(1)-13-(279)。Table 13: In the general formula I-1A, when R 1 =CH 3 and R 2 =Cl, the substituent (R 3 ) n is the same as that in Table 4, corresponding to 4-1-4-279 of Table 4 in turn, representing the compound The serial numbers are 13-(1)-13-(279).
表14:通式I-1A中,当R1=C2H5、R2=Cl时,取代基(R3)n与表4一致,依次对应表4的4-1-4-279,代表化合物编号依次为14-(1)-14-(279)。Table 14: In general formula I-1A, when R 1 =C 2 H 5 , R 2 =Cl, the substituent (R 3 ) n is consistent with Table 4, corresponding to 4-1-4-279 of Table 4 in turn, The representative compound numbers are 14-(1)-14-(279).
表15:通式I-1A中,当R1=CHF2、R2=Cl时,取代基(R3)n与表4一致,依次对应表4的4-1-4-279,代表化合物编号依次为15-(1)-15-(279)。Table 15: In the general formula I-1A, when R 1 =CHF 2 and R 2 =Cl, the substituent (R 3 ) n is the same as that in Table 4, corresponding to 4-1-4-279 of Table 4 in turn, representing the compound The serial numbers are 15-(1)-15-(279).
表16:通式I-1B中,当W=R4=R5=R6=R7=R8=H时,取代基(R3)n与表4一致,依次对应表4的4-1-4-279,代表化合物编号依次为16-(1)-16-(279)。Table 16: In the general formula I-1B, when W=R 4 =R 5 =R 6 =R 7 =R 8 =H, the substituent (R 3 ) n is consistent with Table 4, and corresponds to 4- of Table 4 in turn 1-4-279, the representative compound number is 16-(1)-16-(279).
表17:通式I-1C中,当W=R8=H,R9=Cl时,取代基(R3)n与表4一致,依次对应表4的4-1-4-279,代表化合物编号依次为17-(1)-17-(279)。Table 17: In general formula I-1C, when W=R 8 =H, R 9 =Cl, the substituent (R 3 ) n is consistent with Table 4, corresponding to 4-1-4-279 of Table 4 in turn, representing The compound numbers are 17-(1)-17-(279) in sequence.
本发明化合物按照以下方法制备,反应式如下,式中各基团除另有说明外定义同前:The compound of the present invention is prepared according to the following method, and the reaction formula is as follows, and each group in the formula is defined as before unless otherwise stated:
通式化合物I的制备采用如下方法:The preparation of compound I of general formula adopts the following method:
进一步的说通式化合物I-1A、I-1B和I-1C的制备还可采用如下方法:Further, the preparation of general formula compounds I-1A, I-1B and I-1C can also adopt the following methods:
中间体M和R与I1A、I1B、I1C和III在碱性条件下于适宜的溶剂中反应得到通式I与I-1A、I-1B和I-1C化合物。Intermediates M and R are reacted with I1A, I1B, I1C and III under basic conditions in a suitable solvent to give compounds of general formula I and I-1A, I-1B and I-1C.
适宜的碱可选自如氢氧化钾、氢氧化钠、碳酸钠、碳酸钾、碳酸氢钠、三乙胺、吡啶、甲醇钠、乙醇钠、氢化钠、叔丁醇钾或叔丁醇钠等。Suitable bases can be selected from, for example, potassium hydroxide, sodium hydroxide, sodium carbonate, potassium carbonate, sodium bicarbonate, triethylamine, pyridine, sodium methoxide, sodium ethoxide, sodium hydride, potassium tert-butoxide or sodium tert-butoxide, and the like.
反应在适宜的溶剂中进行,适宜的溶剂可选自如四氢呋喃、1,4-二氧六环、乙腈、甲苯、二甲苯、苯、N,N-二甲基甲酰胺、N-甲基吡咯烷酮、二甲亚砜、丙酮或丁酮等。The reaction is carried out in a suitable solvent, such as tetrahydrofuran, 1,4-dioxane, acetonitrile, toluene, xylene, benzene, N,N-dimethylformamide, N-methylpyrrolidone, Dimethyl sulfoxide, acetone or butanone, etc.
反应温度可在室温至溶剂沸点温度之间,通常为20-100℃。The reaction temperature can be between room temperature and the boiling point of the solvent, usually 20-100°C.
反应时间为30分钟至20小时,通常1-10小时。The reaction time is 30 minutes to 20 hours, usually 1-10 hours.
中间体I1部分有市售,也可以按公知方法制备,例如参照文献JP2000007662、US4977264、US6090815、US20040092402、JP09124613、US5468751、US4985426、US4845097、Journal of the American Chemical Society(1957),79,1455、Journal of ChemicalSociety(1955),p.3478-3481描述的方法制备。Part of intermediate I1 is commercially available, and can also be prepared by known methods, for example, refer to documents JP2000007662, US4977264, US6090815, US20040092402, JP09124613, US5468751, US4985426, US4845097, Journal of the American Chemical Society (1957), 79,1455, Prepared as described in Chemical Society (1955), p. 3478-3481.
中间体Ⅲ是制备本发明通式I-1化合物的关键中间体,按以下方法制备:Intermediate III is the key intermediate for preparing the compound of general formula I-1 of the present invention, and is prepared by the following method:
中间体M1与N-Boc-4-哌啶酮在适宜的温度下,以甲醇作溶剂,以四氢吡咯作缚酸剂反应30分钟至10小时,通常1-4小时,制得中间体M2,此步骤操作方法参照Journal ofCombinatorial Chemistry,10(2),225-229;2008和WO2008065508;M2经脱Boc制得III,此步骤操作方法参照Bioorganic&Medicinal Chemistry Letters,20(2),746-754;2010和WO2012112743。Intermediate M1 is reacted with N-Boc-4-piperidone at suitable temperature, using methanol as solvent and tetrahydropyrrole as acid binding agent for 30 minutes to 10 hours, usually 1-4 hours, to obtain intermediate M2 , the operation method of this step refers to Journal of Combinatorial Chemistry, 10(2), 225-229; 2008 and WO2008065508; M2 is de-Boc to obtain III, the operation method of this step refers to Bioorganic&Medicinal Chemistry Letters, 20(2), 746-754; 2010 and WO2012112743.
虽然本发明的通式I化合物与现有技术中公开的某些化合物也属于含嘧啶的螺环类化合物,但结构特征仍存在显著不同。并且由于这些结构上的差异而使得本发明的化合物具有更好的杀菌活性。Although the compounds of the general formula I of the present invention and some compounds disclosed in the prior art also belong to the pyrimidine-containing spiro compounds, there are still significant differences in the structural characteristics. And because of these structural differences, the compounds of the present invention have better bactericidal activity.
通式I化合物对农业或其他领域中的多种病菌显示出优异的活性。因此,本发明的技术方案还包括通式I化合物在农业或其他领域中用作制备杀菌剂的用途。The compounds of general formula I show excellent activity against various pathogens in agriculture or other fields. Therefore, the technical solution of the present invention also includes the use of the compound of general formula I in agriculture or other fields for preparing fungicides.
下面提及的病害的例子仅用来说明本发明,但绝不限定本发明。The examples of diseases mentioned below are only intended to illustrate the present invention, but in no way limit the present invention.
通式I化合物可用于防治下列病害:卵菌纲病害,如霜霉病(黄瓜霜霉病、油菜霜霉病、大豆霜霉病、甜菜霜霉病、甘蔗霜霉病、烟草霜霉病、豌豆霜霉病、丝瓜霜霉病、冬瓜霜霉病、甜瓜霜霉病、白菜类霜霉病、菠菜霜霉病、萝卜霜霉病、葡萄霜霉病、葱霜霉病),白锈菌(油菜白锈病、白菜类白锈病),猝倒病(油菜猝倒病、烟草猝倒病、番茄猝倒病、辣椒猝倒病、茄子猝倒病、黄瓜猝倒病、棉苗猝倒病),绵腐病(辣椒绵腐病、丝瓜绵腐病、冬瓜绵腐病),疫病(蚕豆疫病、黄瓜疫病、南瓜疫病、冬瓜疫病、西瓜疫病、甜瓜疫病、辣椒疫病、韭菜疫病、大蒜疫病、棉花疫病),晚疫病(马铃薯晚疫病、番茄晚疫病)等;半知菌病害,如枯萎病(甘薯枯萎病、棉花枯萎病、芝麻枯萎病、蓖麻枯萎病、番茄枯萎病、菜豆枯萎病、黄瓜枯萎病、丝瓜枯萎病、南瓜枯萎病、冬瓜枯萎病、西瓜枯萎病、甜瓜枯萎病、辣椒枯萎病、蚕豆枯萎病、油菜枯萎病、大豆枯萎病),根腐病(辣椒根腐病、茄子根腐病、菜豆根腐病、黄瓜根腐病、苦瓜根腐病、棉黑根腐病、蚕豆根腐病),立枯病(棉苗立枯病、芝麻立枯病、辣椒立枯病、黄瓜立枯病、白菜立枯病),炭疽病(高粱炭疽病、棉花炭疽病、红麻炭疽病、黄麻炭疽病、亚麻炭疽病、烟草炭疽病、桑炭疽病、辣椒炭疽病、茄子炭疽病、菜豆炭疽病、黄瓜炭疽病、苦瓜炭疽病、西葫芦炭疽病、冬瓜炭疽病、西瓜炭疽病、甜瓜炭疽病、荔枝炭疽病),黄萎病(棉花黄萎病、向日葵黄萎病、番茄黄萎病、辣椒黄萎病、茄子黄萎病),黑星病(西葫芦黑星病、冬瓜黑星病、甜瓜黑星病),灰霉病(棉铃灰霉病、红麻灰霉病、番茄灰霉病、辣椒灰霉病、菜豆灰霉病、芹菜灰霉病、菠菜灰霉病、猕猴桃灰霉病),褐斑病(棉花褐斑病、黄麻褐斑病、甜菜褐斑病、花生褐斑病、辣椒褐斑病、冬瓜褐斑病、大豆褐斑病、向日葵褐斑病、豌豆褐斑病、蚕豆褐斑病),黑斑病(亚麻假黑斑病、油菜黑斑病、芝麻黑斑病、向日葵黑斑病、蓖麻黑斑病、番茄黑斑病、辣椒黑斑病、茄子黑斑病、菜豆黑斑病、黄瓜黑斑病、芹菜黑斑病、胡萝卜黑腐病、胡萝卜黑斑病、苹果黑斑病、花生黑斑病),斑枯病(番茄斑枯病、辣椒斑枯病、芹菜斑枯病),早疫病(番茄早疫病、辣椒早疫病、茄子早疫病、马铃薯早疫病、芹菜早疫病),轮纹病(大豆轮纹病、芝麻轮纹病、菜豆轮纹病),叶枯病(芝麻叶枯病、向日葵叶枯病、西瓜叶枯病、甜瓜叶枯病),茎基腐病(番茄茎基腐病、菜豆茎基腐病),及其他(玉米圆斑病、红麻腰折病、稻瘟病、栗黑鞘病、甘蔗眼斑病、棉铃曲霉病、花生冠腐病、大豆茎枯病、大豆黑点病、甜瓜大斑病、花生网斑病、茶赤叶斑病、辣椒白星病、冬瓜叶斑病、芹菜黑腐病、菠菜心腐病、红麻叶霉病、红麻斑点病、黄麻茎斑病、大豆紫斑病、芝麻叶斑病、蓖麻灰斑病、茶褐色叶斑病、茄子褐色圆星病、菜豆红斑病、苦瓜白斑病、西瓜斑点病、黄麻枯腐病、向日葵根茎腐病、菜豆炭腐病、大豆靶点病、茄子棒孢叶斑病、黄瓜靶斑病、番茄叶霉病、茄子叶霉病、蚕豆赤斑病等)等;担子菌病害,如锈病(小麦条锈病、小麦杆锈病、小麦叶锈病、花生锈病、向日葵锈病、甘蔗锈病、韭菜锈病、葱锈病、栗锈病、大豆锈病),黑穗病(玉米丝黑穗病、玉米黑粉病、高粱丝黑穗病、高粱散黑穗病、高粱坚黑穗病、高梁柱黑粉病、栗粒黑穗病、甘蔗黑穗病、菜豆锈病)及其他(如小麦纹枯病、水稻纹枯病等)等;子囊菌病害,如白粉病(小麦白粉病、油菜白粉病、芝麻白粉病、向日葵白粉病、甜菜白粉病、茄子白粉病、豌豆白粉病、丝瓜白粉病、南瓜白粉病、西葫芦白粉病、冬瓜白粉病、甜瓜白粉病、葡萄白粉病、蚕豆白粉病),菌核病(亚麻菌核病、油菜菌核病、大豆菌核病、花生菌核病、烟草菌核病、辣椒菌核病、茄子菌核病、菜豆菌核病、豌豆菌核病、黄瓜菌核病、苦瓜菌核病、冬瓜菌核病、西瓜菌核病、芹菜菌核病),黑星病(苹果黑星病、梨黑星病)等。Compounds of general formula I can be used to control the following diseases: Oomycetes diseases, such as downy mildew (cucumber downy mildew, rape downy mildew, soybean downy mildew, sugar beet downy mildew, sugarcane downy mildew, tobacco downy mildew, Pea downy mildew, loofah downy mildew, wax gourd downy mildew, melon downy mildew, cabbage downy mildew, spinach downy mildew, radish downy mildew, grape downy mildew, onion downy mildew), white rust (Rape white rust, Chinese cabbage-like white rust), damping off (rape damping off, tobacco damping off, tomato damping off, pepper damping off, eggplant damping off, cucumber damping off, cotton seedling damping off ), cotton rot (chili cotton rot, loofah cotton rot, wax gourd cotton rot), blight (broad bean blight, cucumber blight, pumpkin blight, wax gourd blight, watermelon blight, muskmelon blight, pepper blight, leek blight, garlic blight , cotton blight), late blight (potato late blight, tomato late blight), etc; disease, cucumber wilt, loofah wilt, pumpkin wilt, wax gourd wilt, watermelon wilt, muskmelon wilt, pepper wilt, broad bean wilt, rapeseed wilt, soybean wilt), root rot (chili root rot) Disease, eggplant root rot, bean root rot, cucumber root rot, bitter gourd root rot, cotton black root rot, broad bean root rot), blight (cotton seedling blight, sesame blight, pepper blight of cucumber, blight of cabbage), anthracnose (sorghum anthracnose, cotton anthracnose, kenaf anthracnose, jute anthracnose, flax anthracnose, tobacco anthracnose, mulberry anthracnose, pepper anthracnose , eggplant anthracnose, kidney bean anthracnose, cucumber anthracnose, bitter gourd anthracnose, zucchini anthracnose, wax gourd anthracnose, watermelon anthracnose, melon anthracnose, lychee anthracnose), verticillium wilt (cotton verticillium wilt, sunflower verticillium wilt) disease, tomato verticillium wilt, pepper verticillium wilt, eggplant verticillium wilt), scab (zucchini scab, wax gourd scab, muskmelon scab), gray mold (boll boll gray mold, kenaf gray Mildew, tomato gray mold, pepper gray mold, bean gray mold, celery gray mold, spinach gray mold, kiwifruit gray mold), brown spot (cotton brown spot, jute brown spot, beet brown spot Leaf spot, peanut brown spot, pepper brown spot, wax gourd brown spot, soybean brown spot, sunflower brown spot, pea brown spot, broad bean brown spot), black spot (flax false black spot, rapeseed Black spot, sesame black spot, sunflower black spot, castor black spot, tomato black spot, pepper black spot, eggplant black spot, kidney bean black spot, cucumber black spot, celery black spot, Carrot black rot, carrot black spot, apple black spot, peanut black spot), leaf blight (tomato leaf blight, pepper blight, celery blight), early blight (tomato early blight, pepper blight) blight, eggplant early blight, potato early blight, celery early blight), ring disease (soybean ring disease, sesame ring disease, kidney bean ring disease), leaf blight (sesame leaf blight, sunflower leaf blight, watermelon Leaf blight, melon leaf blight), stem base rot (tomato stem base rot, bean stem base rot), and others (maize round spot, kenaf Fracture disease, rice blast, chestnut black sheath disease, sugarcane eye spot disease, cotton boll aspergillosis, peanut crown rot, soybean stem blight, soybean black spot disease, melon large leaf spot disease, peanut net spot disease, tea red leaf spot disease , pepper white spot, wax gourd leaf spot, celery black rot, spinach heart rot, kenaf leaf mold, kenaf spot, jute stem spot, soybean purple spot, sesame leaf spot, castor gray spot , tea-brown leaf spot disease, eggplant brown spot disease, kidney bean red spot disease, bitter gourd white spot disease, watermelon spot disease, jute blight rot, sunflower rhizome rot, kidney bean charcoal rot disease, soybean target disease, eggplant corynebacterium leaf spot disease , cucumber target spot, tomato leaf mold, eggplant leaf mold, broad bean red spot, etc.); Basidiomycete diseases, such as rust (wheat stripe rust, wheat stem rust, wheat leaf rust, peanut rust, sunflower rust, sugarcane rust, leek rust, onion rust, chestnut rust, soybean rust), smut (corn smut, corn smut, sorghum head smut, sorghum smut, sorghum smut, sorghum column Smut, chestnut smut, sugarcane smut, bean rust) and others (such as wheat sheath blight, rice sheath blight, etc.), etc.; Ascomycete diseases, such as powdery mildew (wheat powdery mildew, rape powdery mildew) , sesame powdery mildew, sunflower powdery mildew, sugar beet powdery mildew, eggplant powdery mildew, pea powdery mildew, loofah powdery mildew, pumpkin powdery mildew, zucchini powdery mildew, wax gourd powdery mildew, melon powdery mildew, grape powdery mildew, broad bean powdery mildew), Sclerotinia (Flax Sclerotinia, Rape Sclerotinia, Soybean Sclerotinia, Peanut Sclerotinia, Tobacco Sclerotinia, Pepper Sclerotinia, Eggplant Sclerotinia, Bean Sclerotinia, Pea Sclerotinia, Cucumber Sclerotinia, bitter gourd sclerotinia, wax gourd sclerotinia, watermelon sclerotinia, celery sclerotinia), sclerotinia (apple scab, pear scab), etc.
由于其积极的特性,上述化合物可有利地用于保护农业和园艺业重要的作物、家畜和种畜,以及人类常去的环境免于病菌的伤害。Due to their positive properties, the above-mentioned compounds can be advantageously used to protect agriculturally and horticulturally important crops, livestock and breeding stock, as well as the environment frequented by humans, from pathogens.
为获得理想效果,化合物的用量因各种因素而改变,例如所用化合物、预保护的作物、有害生物的类型、感染程度、气候条件、施药方法、采用的剂型。The amount of compound to be used to obtain the desired effect will vary depending on various factors such as the compound used, the crop being pre-protected, the type of pest, the degree of infestation, the climatic conditions, the method of application, the dosage form employed.
每公顷10克-5公斤的化合物剂量能提供充分的防治。Doses of 10 g to 5 kg of compound per hectare provide adequate control.
本发明还包括以通式I所示化合物作为活性组分的杀菌组合物。该杀菌组合物中活性组分的重量百分含量在0.5-99%之间。该杀菌组合物中还包括农业、林业、卫生上可接受的载体。The present invention also includes a bactericidal composition using the compound represented by the general formula I as an active ingredient. The weight percent content of active components in the bactericidal composition is between 0.5-99%. Agricultural, forestry and hygienically acceptable carriers are also included in the fungicidal composition.
本发明的组合物可以制剂的形式施用。通式I所示化合物作为活性组分溶解或分散于载体中或配制成制剂以便作为杀菌使用时更易于分散。例如:这些化学制剂可被制成可湿性粉剂、油悬剂、水悬剂、水乳剂、水剂或乳油等。在这些组合物中,至少加入一种液体或固体载体,并且当需要时可以加入适当的表面活性剂。The compositions of the present invention can be administered in the form of formulations. The compound represented by the general formula I is dissolved or dispersed in a carrier as an active ingredient or formulated into a preparation for easier dispersion when used as a sterilization. For example, these chemical preparations can be made into wettable powders, oil suspensions, water suspensions, water emulsions, water preparations or emulsifiable concentrates. In these compositions, at least one liquid or solid carrier is added, and when desired, suitable surfactants can be added.
本发明的技术方案还包括防治病菌的方法:将本发明的杀菌组合物施于所述的病菌或其生长介质上。通常选择的较为适宜有效量为每公顷10克到1000克,优选有效量为每公顷20克到500克。The technical solution of the present invention also includes a method for preventing and treating germs: applying the bactericidal composition of the present invention to the germs or their growth medium. The more suitable effective amount is usually selected to be 10 grams to 1000 grams per hectare, and the preferred effective amount is 20 grams to 500 grams per hectare.
对于某些应用,例如在农业上可在本发明的杀菌组合物中加入一种或多种其它的杀菌剂、杀虫杀螨剂、除草剂、植物生长调节剂或肥料等,由此可产生附加的优点和效果。For certain applications, such as in agriculture, one or more other fungicides, insecticides, acaricides, herbicides, plant growth regulators or fertilizers can be added to the fungicidal compositions of the present invention, thereby producing Additional benefits and effects.
应明确的是,在本发明的权利要求所限定的范围内,可进行各种变换和改动。It should be understood that various changes and modifications can be made within the scope defined by the claims of the present invention.
具体实施方式Detailed ways
以下具体实施例用来进一步说明本发明,但本发明绝非仅限于这些例子(除另有注明外,所用原料均有市售)。The following specific examples are used to further illustrate the present invention, but the present invention is by no means limited to these examples (unless otherwise noted, the raw materials used are all commercially available).
合成实施例Synthesis Example
实施例1:中间体4,5-二氯-6-甲基嘧啶的制备Example 1: Preparation of intermediate 4,5-dichloro-6-methylpyrimidine
1)4-羟基-5-氯-6-甲基嘧啶的制备1) Preparation of 4-hydroxy-5-chloro-6-methylpyrimidine
室温搅拌下向11.30g(0.11mol)醋酸甲脒的50ml甲醇溶液中缓慢滴加8.80g(0.16mol)甲醇钠的甲醇溶液,滴毕室温继续搅拌2h。然后向上述溶液中滴加11.17g(0.068mol)中间体2-氯乙酰乙酸乙酯,继续室温搅拌反应5-7小时。TLC监测反应完毕后,减压蒸除溶剂,用盐酸调pH=5~6,抽滤得橙黄色固体,水相用(3×50ml)乙酸乙酯萃取,无水硫酸镁干燥、过滤、脱溶。残余物溶于50ml乙酸乙酯中,放置过夜,过滤得橙黄色固体6.48g。收率66%,熔点181~184℃。A methanol solution of 8.80 g (0.16 mol) sodium methoxide was slowly added dropwise to a methanol solution of 11.30 g (0.11 mol) of formamidine acetate in 50 ml of methanol with stirring at room temperature, and stirring was continued at room temperature for 2 h. Then, 11.17 g (0.068 mol) of the intermediate ethyl 2-chloroacetoacetate was added dropwise to the above solution, and the reaction was continued to stir at room temperature for 5-7 hours. After the completion of the reaction monitored by TLC, the solvent was evaporated under reduced pressure, the pH was adjusted to 5-6 with hydrochloric acid, and an orange-yellow solid was obtained by suction filtration. The aqueous phase was extracted with (3×50 ml) ethyl acetate, dried over anhydrous magnesium sulfate, filtered, removed dissolve. The residue was dissolved in 50 ml of ethyl acetate, left overnight, and filtered to obtain 6.48 g of an orange-yellow solid. The yield is 66%, and the melting point is 181-184°C.
2)4,5-二氯-6-甲基嘧啶的制备2) Preparation of 4,5-dichloro-6-methylpyrimidine
将14.5g(0.1mol)4-羟基-5-氯-6-甲基嘧啶溶于50ml甲苯溶液中,搅拌下向反应瓶中滴入50ml三氯氧磷,滴毕升温回流反应5-7小时。TLC监测反应完毕后,减压蒸除甲苯和过量的三氯氧磷,搅拌下将反应物倾入冰水中,水相用(3×50ml)乙酸乙酯萃取,合并有机相,无水硫酸镁干燥、过滤、脱溶。残余物柱层析(洗脱剂为乙酸乙酯与石油醚,体积比为1:5)分离得黄色液体14.43g,收率88.5%。14.5g (0.1mol) of 4-hydroxy-5-chloro-6-methylpyrimidine was dissolved in 50ml of toluene solution, 50ml of phosphorus oxychloride was added dropwise to the reaction flask under stirring, and the temperature was raised and refluxed for 5-7 hours. . After the completion of the reaction monitored by TLC, toluene and excess phosphorus oxychloride were evaporated under reduced pressure, the reactant was poured into ice-water with stirring, the aqueous phase was extracted with (3×50ml) ethyl acetate, the organic phases were combined, anhydrous magnesium sulfate Dry, filter and dissolve. The residue was separated by column chromatography (the eluent was ethyl acetate and petroleum ether, the volume ratio was 1:5) to obtain 14.43 g of a yellow liquid with a yield of 88.5%.
实施例2:4,5-二氯噻吩并[2,3-d]嘧啶的制备Example 2: Preparation of 4,5-dichlorothieno[2,3-d]pyrimidine
取2-氨基-3-氰基-4-氧代-5,5-二氢噻吩和250ml三氯氧磷(POCl3)于反应瓶中,室温下慢慢滴加38ml N,N-二甲基甲酰胺,约30分钟滴加完毕。室温反应1小时,再升温至75℃反应3小时。降至室温后,将反应液倒入碎冰中,过滤得深灰色固体89.1g,收率86.9%,熔点160-161℃。Take 2-amino-3-cyano-4-oxo-5,5-dihydrothiophene and 250ml phosphorus oxychloride (POCl 3 ) in a reaction flask, and slowly add 38ml N,N-dimethylformaldehyde dropwise at room temperature Formamide was added dropwise in about 30 minutes. The reaction was carried out at room temperature for 1 hour, and then the temperature was raised to 75°C for 3 hours. After cooling to room temperature, the reaction solution was poured into crushed ice, and filtered to obtain 89.1 g of a dark gray solid with a yield of 86.9% and a melting point of 160-161°C.
实施例3:中间体4-氯喹唑啉的制备Embodiment 3: the preparation of intermediate 4-chloroquinazoline
1)喹唑啉-4(3H)-酮的制备1) Preparation of quinazolin-4(3H)-one
取13.7g(0.1mol)邻氨基苯甲酸与20ml甲酰胺于250ml三口瓶中,升温至140℃反应5-8小时。TLC监测反应完毕后,将反应液降温至100℃,搅拌下滴加80ml水,之后冷却至室温,过滤并用用无水乙醚洗涤滤饼得红棕色10.96g,收率75.1%。Take 13.7g (0.1mol) of anthranilic acid and 20ml of formamide in a 250ml three-necked flask, heat up to 140°C and react for 5-8 hours. After the completion of the reaction monitored by TLC, the reaction solution was cooled to 100°C, 80ml of water was added dropwise with stirring, then cooled to room temperature, filtered and washed with anhydrous ether to obtain 10.96g of reddish brown, yield 75.1%.
2)4-氯喹唑啉的制备2) Preparation of 4-chloroquinazoline
取14.6g(0.1mol)喹唑啉-4(3H)-酮于250ml单口瓶中,50ml氯化亚砜作溶剂,升温至回流反应4-6小时。TLC监测反应完毕后,冷却后将反应液倒入水中搅拌30min,过滤并用无水乙醚洗涤得红棕色固体10.96g,收率92.7%。Take 14.6 g (0.1 mol) of quinazolin-4(3H)-one in a 250 ml single-necked bottle, use 50 ml of thionyl chloride as a solvent, and heat up to reflux for 4-6 hours. After the completion of the reaction monitored by TLC, after cooling, the reaction solution was poured into water and stirred for 30 min, filtered and washed with anhydrous ether to obtain 10.96 g of a reddish-brown solid with a yield of 92.7%.
实施例4:中间体4-氧代-2-螺(哌啶-4-基)-苯并吡喃盐酸盐的合成Example 4: Synthesis of intermediate 4-oxo-2-spiro(piperidin-4-yl)-benzopyran hydrochloride
1)4-氧代-2-螺[苯并二氢吡喃-2,4-哌啶]-1-羧酸叔丁酯的制备1) Preparation of tert-butyl 4-oxo-2-spiro[chroman-2,4-piperidine]-1-carboxylate
将13.60g(0.10mol)邻羟基苯乙酮置于150ml甲醇中,再加入19.90g(0.10mol)N-Boc-4-哌啶酮,室温搅拌下滴加8.64g(0.12mol)四氢吡咯,滴毕,升温至70℃继续反应1-5小时。反应完毕后,减压蒸除溶剂,加入(3×50ml)乙酸乙酯萃取,有机相用饱和食盐水50ml洗涤,脱溶后得无色油状液体30.39g,收率95.82%。Place 13.60g (0.10mol) o-hydroxyacetophenone in 150ml methanol, then add 19.90g (0.10mol) N-Boc-4-piperidone, and add 8.64g (0.12mol) tetrahydropyrrole dropwise with stirring at room temperature , After dripping, the temperature was raised to 70 °C and the reaction was continued for 1-5 hours. After the reaction was completed, the solvent was evaporated under reduced pressure, and ethyl acetate (3×50ml) was added for extraction. The organic phase was washed with 50ml of saturated brine, and 30.39g of colorless oily liquid was obtained after precipitation, with a yield of 95.82%.
2)4-氧代-2-螺(哌啶-4-基)-苯并吡喃盐酸盐的制备2) Preparation of 4-oxo-2-spiro(piperidin-4-yl)-benzopyran hydrochloride
将3.17g(0.01mol)4-氧代-2-螺[苯并二氢吡喃-2,4-哌啶]-1-羧酸叔丁酯乙胺加入50ml乙酸乙酯中,室温搅拌下滴加6ml浓盐酸,固体溶解,继续搅拌4-5小时,TLC监测反应完毕后,减压蒸除溶剂,加入10ml二氯甲烷搅拌半小时,过滤,并用二氯甲烷洗涤滤饼得2.33g白色固体。3.17g (0.01mol) of 4-oxo-2-spiro[chroman-2,4-piperidine]-1-carboxylate tert-butyl ester ethylamine was added to 50ml of ethyl acetate, stirred at room temperature 6ml of concentrated hydrochloric acid was added dropwise, the solid was dissolved, and the stirring was continued for 4-5 hours. After TLC monitoring was completed, the solvent was evaporated under reduced pressure, 10ml of dichloromethane was added, stirred for half an hour, filtered, and the filter cake was washed with dichloromethane to obtain 2.33g of white solid.
实施例5:化合物14-(1)的制备Example 5: Preparation of compound 14-(1)
将0.25g(0.001mol)4-氧代-2-螺(哌啶-4-基)-苯并吡喃盐酸盐和0.18g(0.001mol)4,5-二氯-6-乙基嘧啶加入20ml DMF中。加入0.15g(0.015mol)三乙胺,加热至90℃反应2-6小时,TLC监测反应完毕后,减压蒸除溶剂,加入(3×50ml)乙酸乙酯萃取,有机相用饱和食盐水50ml洗涤,脱溶后残余物柱层析(洗脱剂为乙酸乙酯与石油醚(沸程60-90℃),体积比为1:4)得白色固体0.28g,收率77.9%。0.25g (0.001mol) 4-oxo-2-spiro(piperidin-4-yl)-benzopyran hydrochloride and 0.18g (0.001mol) 4,5-dichloro-6-ethylpyrimidine Add to 20ml DMF. Add 0.15g (0.015mol) triethylamine, heat to 90°C and react for 2-6 hours, after monitoring the reaction by TLC, evaporate the solvent under reduced pressure, add (3×50ml) ethyl acetate for extraction, and use saturated brine for the organic phase 50ml was washed, and the residue was subjected to column chromatography (eluent: ethyl acetate and petroleum ether (boiling range 60-90°C), volume ratio: 1:4) after desolvation to obtain 0.28g of white solid with a yield of 77.9%.
1H-NMR(300MHz,内标TMS,溶剂CDCl3)δ(ppm):1.28(t,J=7.2Hz,3H,CH3),1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.78(s,2H,COCH2),2.88(q,J=7.2Hz,2H,CH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.53(s,1H,Pyrimidine 2-H). 1 H-NMR (300 MHz, internal standard TMS, solvent CDCl 3 ) δ (ppm): 1.28 (t, J=7.2 Hz, 3H, CH 3 ), 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd , 2H, Piperidine-CH 2 ), 2.78(s, 2H, COCH 2 ), 2.88(q, J=7.2Hz, 2H, CH 2 ), 3.49(td, 2H, Piperidine-CH 2 ), 4.20(d, 2H, Piperidine-CH 2 ), 7.03 (t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.52 (td, J=7.5, 1.5Hz, 1H, Ph-7-H), 7.89 ( dd,J=7.5,1.5Hz,1H,Ph-5-H),8.53(s,1H,Pyrimidine 2-H).
实施例6:化合物16-(1)的制备Example 6: Preparation of compound 16-(1)
将0.16g(0.001mol)4-氯喹唑啉和0.25g(0.001mol)4-氧代-2-螺(哌啶-4-基)-苯并吡喃盐酸盐加入20ml DMF中。加入0.15g(0.015mol)三乙胺,加热至90℃反应2-6小时,TLC监测反应完毕后,减压蒸除溶剂,加入(3×50ml)乙酸乙酯萃取,有机相用饱和食盐水50ml洗涤,脱溶后残余物柱层析(洗脱剂为乙酸乙酯与石油醚(沸程60-90℃),体积比为1:4)得0.25g油状固体,收率72.6%。0.16 g (0.001 mol) of 4-chloroquinazoline and 0.25 g (0.001 mol) of 4-oxo-2-spiro(piperidin-4-yl)-benzopyran hydrochloride were added to 20 ml of DMF. Add 0.15g (0.015mol) triethylamine, heat to 90°C and react for 2-6 hours, after monitoring the reaction by TLC, evaporate the solvent under reduced pressure, add (3×50ml) ethyl acetate for extraction, and use saturated brine for the organic phase 50ml was washed, and the residue was subjected to column chromatography after desolvation (the eluent was ethyl acetate and petroleum ether (boiling range 60-90°C), the volume ratio was 1:4) to obtain 0.25g of oily solid with a yield of 72.6%.
1H-NMR(600MHz,内标TMS,溶剂CDCl3)δ(ppm):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.81(s,2H,COCH2),3.32(td,2H,Piperidine-CH2),3.63(d,2H,Piperidine-CH2),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.44-7.56(m,2H,Ph-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.86-7.91(m,2H,Ph-2H),7.95(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.75(s,1H,Pyrimidine 2-H). 1 H-NMR (600MHz, internal standard TMS, solvent CDCl 3 ) δ (ppm): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.81 (s, 2H, COCH 2 ), 3.32 (td, 2H, Piperidine-CH 2 ), 3.63 (d, 2H, Piperidine-CH 2 ), 7.03 (t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.44- 7.56(m,2H,Ph-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.86-7.91(m,2H,Ph-2H),7.95(dd,J= 7.5,1.5Hz,1H,Ph-5-H),8.75(s,1H,Pyrimidine 2-H).
实施例7:化合物17-(1)的制备Example 7: Preparation of compound 17-(1)
将0.20g(0.001mol)4,5-二氯噻吩并[2,3-d]嘧啶和0.25g(0.001mol)4-氧代-2-螺(哌啶-4-基)-苯并吡喃盐酸盐加入20ml DMF中。加入0.15g(0.015mol)三乙胺,加热至90℃反应2-6小时,TLC监测反应完毕后,减压蒸除溶剂,加入(3×50ml)乙酸乙酯萃取,有机相用饱和食盐水50ml洗涤,脱溶后残余物柱层析(洗脱剂为乙酸乙酯与石油醚(60-90℃),体积比为1:4)得淡黄固体0.29g,收率74.5%,熔点180.6℃。0.20g (0.001mol) 4,5-dichlorothieno[2,3-d]pyrimidine and 0.25g (0.001mol) 4-oxo-2-spiro(piperidin-4-yl)-benzopyridine The pyranyl hydrochloride was added to 20 ml of DMF. Add 0.15g (0.015mol) triethylamine, heat to 90°C and react for 2-6 hours, after monitoring the reaction by TLC, evaporate the solvent under reduced pressure, add (3×50ml) ethyl acetate for extraction, and use saturated brine for the organic phase 50ml was washed, and the residue was subjected to column chromatography after precipitation (the eluent was ethyl acetate and petroleum ether (60-90°C), the volume ratio was 1:4) to obtain 0.29g of pale yellow solid, yield 74.5%, melting point 180.6 °C.
1H-NMR(600MHz,内标TMS,溶剂CDCl3)δ(ppm):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.80(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.23(s,1H,SCH),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.58(s,1H,Pyrimidine2-H). 1 H-NMR (600MHz, internal standard TMS, solvent CDCl 3 ) δ (ppm): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.80 (s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 7.03 (t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.23 ( s,1H,SCH),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.58(s ,1H,Pyrimidine2-H).
本发明的其他化合物可以参照以上实施例制备。Other compounds of the present invention can be prepared with reference to the above examples.
部分化合物的物性数据及核磁数据(1HNMR,600MHz,内标TMS,ppm)如下:The physical property data and nuclear magnetic data ( 1 HNMR, 600MHz, internal standard TMS, ppm) of some compounds are as follows:
化合物13-(1):熔点132.9℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.53(s,3H,CH3),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.48(s,1H,Pyrimidine 2-H).Compound 13-(1): melting point 132.9°C. δ(CDCl 3 ): 1.83(td, 2H, Piperidine-CH 2 ), 2.20(dd, 2H, Piperidine-CH 2 ), 2.53(s, 3H, CH 3 ), 2.78(s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 7.03 (t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.52 (td, J= 7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.48(s,1H,Pyrimidine 2-H).
化合物15-(1):熔点108.4℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.83(t,J=53.1Hz,1H,CHF2),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.64(s,1H,Pyrimidine2-H).Compound 15-(1): melting point 108.4°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.78 (s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ) ), 4.20 (d, 2H, Piperidine-CH 2 ), 6.83 (t, J=53.1 Hz, 1H, CHF 2 ), 7.03 (t, J=7.5 Hz, 2H, Ph-6, 8-2H), 7.52 (td, J=7.5, 1.5Hz, 1H, Ph-7-H), 7.89 (dd, J=7.5, 1.5Hz, 1H, Ph-5-H), 8.64 (s, 1H, Pyrimidine2-H).
化合物13-(17):熔点151.9℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.53(s,3H,CH3),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.98(d,J=8.7Hz,1H,Ph-8-H),7.45(dd,J=8.7,2.4Hz,1H,Ph-7-H),7.84(d,J=2.4Hz,1H,Ph-5-H),8.48(s,1H,Pyrimidine 2-H).Compound 13-(17): melting point 151.9°C. δ(CDCl 3 ): 1.83(td, 2H, Piperidine-CH 2 ), 2.20(dd, 2H, Piperidine-CH 2 ), 2.53(s, 3H, CH 3 ), 2.78(s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 6.98 (d, J=8.7Hz, 1H, Ph-8-H), 7.45 (dd, J=8.7, 2.4Hz, 1H, Ph-7-H), 7.84(d, J=2.4Hz, 1H, Ph-5-H), 8.48(s, 1H, Pyrimidine 2-H).
化合物14-(17):熔点164.8℃。δ(CDCl3):1.28(t,J=7.2Hz,3H,CH3),1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.78(s,2H,COCH2),2.88(q,J=7.2Hz,2H,CH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.98(d,J=8.7Hz,1H,Ph-8-H),7.45(dd,J=8.7,2.4Hz,1H,Ph-7-H),7.84(d,J=2.4Hz,1H,Ph-5-H),8.53(s,1H,Pyrimidine 2-H).Compound 14-(17): melting point 164.8°C. δ(CDCl 3 ): 1.28 (t, J=7.2Hz, 3H, CH 3 ), 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.78 (s, 2H , COCH 2 ), 2.88 (q, J=7.2Hz, 2H, CH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 6.98 (d, J= 8.7Hz, 1H, Ph-8-H), 7.45 (dd, J=8.7, 2.4Hz, 1H, Ph-7-H), 7.84 (d, J=2.4Hz, 1H, Ph-5-H), 8.53(s,1H,Pyrimidine 2-H).
化合物15-(17):熔点176.0℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.83(t,J=53.1Hz,1H,CHF2),6.98(d,J=8.7Hz,1H,Ph-8-H),7.45(dd,J=8.7,2.4Hz,1H,Ph-7-H),7.84(d,J=2.4Hz,1H,Ph-5-H),8.64(s,1H,Pyrimidine 2-H).Compound 15-(17): melting point 176.0°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.78 (s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ) ), 4.20 (d, 2H, Piperidine-CH 2 ), 6.83 (t, J=53.1 Hz, 1H, CHF 2 ), 6.98 (d, J=8.7 Hz, 1H, Ph-8-H), 7.45 (dd , J=8.7, 2.4Hz, 1H, Ph-7-H), 7.84(d, J=2.4Hz, 1H, Ph-5-H), 8.64(s, 1H, Pyrimidine 2-H).
化合物13-(3):熔点158.4℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.53(s,3H,CH3),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.99(dd,J=9.0,4.2Hz,1H,Ph-8-H),7.25(dd,J=9.0,3.0Hz,1H,Ph-7-H),7.53(dd,J=8.1,3.0Hz,1H,Ph-5-H),8.48(s,1H,Pyrimidine 2-H).Compound 13-(3): melting point 158.4°C. δ(CDCl 3 ): 1.83(td, 2H, Piperidine-CH 2 ), 2.20(dd, 2H, Piperidine-CH 2 ), 2.53(s, 3H, CH 3 ), 2.78(s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 6.99 (dd, J=9.0, 4.2Hz, 1H, Ph-8-H), 7.25 (dd, J= 9.0,3.0Hz,1H,Ph-7-H),7.53(dd,J=8.1,3.0Hz,1H,Ph-5-H),8.48(s,1H,Pyrimidine 2-H).
化合物14-(3):熔点129.4℃。δ(CDCl3):1.28(t,J=7.2Hz,3H,CH3),1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.78(s,2H,COCH2),2.88(q,J=7.2Hz,2H,CH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.99(dd,J=9.0,4.2Hz,1H,Ph-8-H),7.25(dd,J=9.0,3.0Hz,1H,Ph-7-H),7.53(dd,J=8.1,3.0Hz,1H,Ph-5-H),8.53(s,1H,Pyrimidine 2-H).Compound 14-(3): melting point 129.4°C. δ(CDCl 3 ): 1.28 (t, J=7.2Hz, 3H, CH 3 ), 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.78 (s, 2H , COCH 2 ), 2.88 (q, J=7.2Hz, 2H, CH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 6.99 (dd, J= 9.0,4.2Hz,1H,Ph-8-H),7.25(dd,J=9.0,3.0Hz,1H,Ph-7-H),7.53(dd,J=8.1,3.0Hz,1H,Ph-5 -H),8.53(s,1H,Pyrimidine 2-H).
化合物15-(3):熔点161.9℃。δ(CDCl3):δ1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.83(t,J=53.1Hz,1H,CHF2),6.99(dd,J=9.0,4.2Hz,1H,Ph-8-H),7.25(dd,J=9.0,3.0Hz,1H,Ph-7-H),7.53(dd,J=8.1,3.0Hz,1H,Ph-5-H),8.64(s,1H,Pyrimidine 2-H).Compound 15-(3): melting point 161.9°C. δ(CDCl 3 ): δ1.83(td,2H,Piperidine- CH2 ),2.20(dd,2H,Piperidine- CH2 ),2.78(s,2H, COCH2 ),3.49(td,2H,Piperidine- CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 6.83 (t, J=53.1 Hz, 1H, CHF 2 ), 6.99 (dd, J=9.0, 4.2 Hz, 1H, Ph-8-H) ,7.25(dd,J=9.0,3.0Hz,1H,Ph-7-H),7.53(dd,J=8.1,3.0Hz,1H,Ph-5-H),8.64(s,1H,Pyrimidine 2- H).
化合物13-(51):熔点180.9.0℃。δ(CDCl3):δ1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.53(s,3H,CH3),2.86(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),7.17(d,J=9.3Hz,1H,Ph-8-H),8.38(dd,J=9.3,2.4Hz,1H,Ph-7-H),8.48(s,1H,Pyrimidine 2-H),8.79(d,J=2.4Hz,1H,Ph-5-H).Compound 13-(51): melting point 180.9.0°C. δ(CDCl 3 ): δ1.83(td, 2H, Piperidine-CH 2 ), 2.20(dd, 2H, Piperidine-CH 2 ), 2.53(s, 3H, CH 3 ), 2.86(s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 7.17 (d, J=9.3Hz, 1H, Ph-8-H), 8.38 (dd, J= 9.3, 2.4Hz, 1H, Ph-7-H), 8.48 (s, 1H, Pyrimidine 2-H), 8.79 (d, J=2.4Hz, 1H, Ph-5-H).
化合物14-(51):油状物.δ(CDCl3):1.28(t,J=7.2Hz,3H,CH3),1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.86(s,2H,COCH2),2.88(q,J=7.2Hz,2H,CH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),7.17(d,J=9.3Hz,1H,Ph-8-H),8.38(dd,J=9.3,2.4Hz,1H,Ph-7-H),8.53(s,1H,Pyrimidine 2-H),8.79(d,J=2.4Hz,1H,Ph-5-H).Compound 14-(51): Oil. δ(CDCl 3 ): 1.28 (t, J=7.2 Hz, 3H, CH 3 ), 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine -CH 2 ), 2.86(s, 2H, COCH 2 ), 2.88(q, J=7.2Hz, 2H, CH 2 ), 3.49(td, 2H, Piperidine-CH 2 ), 4.20(d, 2H, Piperidine- CH 2 ), 7.17 (d, J=9.3Hz, 1H, Ph-8-H), 8.38 (dd, J=9.3, 2.4Hz, 1H, Ph-7-H), 8.53 (s, 1H, Pyrimidine 2 -H), 8.79(d, J=2.4Hz, 1H, Ph-5-H).
化合物15-(51):熔点198.6℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.86(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.83(t,J=53.1Hz,1H,CHF2),7.17(d,J=9.3Hz,1H,Ph-8-H),8.38(dd,J=9.3,2.4Hz,1H,Ph-7-H),8.64(s,1H,Pyrimidine 2-H),8.79(d,J=2.4Hz,1H,Ph-5-H).Compound 15-(51): melting point 198.6°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.86 (s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ) ), 4.20 (d, 2H, Piperidine-CH 2 ), 6.83 (t, J=53.1 Hz, 1H, CHF 2 ), 7.17 (d, J=9.3 Hz, 1H, Ph-8-H), 8.38 (dd , J=9.3, 2.4Hz, 1H, Ph-7-H), 8.64 (s, 1H, Pyrimidine 2-H), 8.79 (d, J=2.4Hz, 1H, Ph-5-H).
化合物13-(274):熔点140.0℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.53(s,3H,CH3),2.58(d,J=2.1Hz,1H,CH),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),4.68(d,J=2.1Hz,2H,OCH2),6.97(d,J=9.0Hz,1H,Ph-8-H),7.19(dd,J=9.0,2.7Hz,1H,Ph-7-H),7.41(d,J=2.7Hz,1H,Ph-5-H),8.47(s,1H,Pyrimidine 2-H).Compound 13-(274): melting point 140.0°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.53 (s, 3H, CH 3 ), 2.58 (d, J=2.1 Hz, 1H , CH), 2.78(s, 2H, COCH 2 ), 3.49(td, 2H, Piperidine-CH 2 ), 4.20(d, 2H, Piperidine-CH 2 ), 4.68(d, J=2.1Hz, 2H, OCH 2 ), 6.97 (d, J=9.0Hz, 1H, Ph-8-H), 7.19 (dd, J=9.0, 2.7Hz, 1H, Ph-7-H), 7.41 (d, J=2.7Hz, 1H,Ph-5-H),8.47(s,1H,Pyrimidine 2-H).
化合物14-(274):熔点155.0℃。δ(CDCl3):1.28(t,J=7.2Hz,3H,CH3),1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.58(d,J=2.1Hz,1H,CH),2.78(s,2H,COCH2),2.88(q,J=7.2Hz,2H,CH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),4.68(d,J=2.1Hz,2H,OCH2),6.97(d,J=9.0Hz,1H,Ph-8-H),7.19(dd,J=9.0,2.7Hz,1H,Ph-7-H),7.41(d,J=2.7Hz,1H,Ph-5-H),8.53(s,1H,Pyrimidine 2-H).Compound 14-(274): melting point 155.0°C. δ(CDCl 3 ): 1.28 (t, J=7.2 Hz, 3H, CH 3 ), 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.58 (d, J = 2.1Hz, 1H, CH), 2.78(s, 2H, COCH 2 ), 2.88(q, J=7.2Hz, 2H, CH 2 ), 3.49(td, 2H, Piperidine-CH 2 ), 4.20(d, 2H, Piperidine-CH 2 ), 4.68 (d, J=2.1 Hz, 2H, OCH 2 ), 6.97 (d, J=9.0 Hz, 1H, Ph-8-H), 7.19 (dd, J=9.0, 2.7 Hz, 1H, Ph-7-H), 7.41(d, J=2.7Hz, 1H, Ph-5-H), 8.53(s, 1H, Pyrimidine 2-H).
化合物15-(274):油状物。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.58(d,J=2.1Hz,1H,CH),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),4.68(d,J=2.1Hz,2H,OCH2),6.83(t,J=53.1Hz,1H,CHF2),6.97(d,J=9.0Hz,1H,Ph-8-H),7.19(dd,J=9.0,2.7Hz,1H,Ph-7-H),7.41(d,J=2.7Hz,1H,Ph-5-H),8.64(s,1H,Pyrimidine 2-H).Compound 15-(274): Oil. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.58 (d, J=2.1 Hz, 1H, CH ), 2.78 (s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 4.68 (d, J=2.1Hz, 2H, OCH 2 ), 6.83 (t, J=53.1 Hz, 1H, CHF 2 ), 6.97 (d, J=9.0 Hz, 1H, Ph-8-H), 7.19 (dd, J=9.0, 2.7 Hz, 1H, Ph-7-H), 7.41 (d, J=2.7Hz, 1H, Ph-5-H), 8.64(s, 1H, Pyrimidine 2-H).
化合物13-(275):油状物。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.53(s,3H,CH3),2.58(d,J=2.1Hz,1H,CH),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),4.74(d,J=2.4Hz,2H,OCH2),6.55(d,J=2.1Hz,1H,Ph-8-H),6.65(dd,J=8.7,2.1Hz,1H,Ph-6-H),7.85(d,J=8.7Hz,1H,Ph-5-H),8.47(s,1H,Pyrimidine 2-H).Compound 13-(275): Oil. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.53 (s, 3H, CH 3 ), 2.58 (d, J=2.1 Hz, 1H , CH), 2.78(s, 2H, COCH 2 ), 3.49(td, 2H, Piperidine-CH 2 ), 4.20(d, 2H, Piperidine-CH 2 ), 4.74(d, J=2.4Hz, 2H, OCH 2 ), 6.55(d, J=2.1Hz, 1H, Ph-8-H), 6.65(dd, J=8.7, 2.1Hz, 1H, Ph-6-H), 7.85(d, J=8.7Hz, 1H,Ph-5-H),8.47(s,1H,Pyrimidine 2-H).
化合物14-(275):油状物。δ(CDCl3):1.28(t,J=7.2Hz,3H,CH3),1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.58(d,J=2.1Hz,1H,CH),2.78(s,2H,COCH2),2.88(q,J=7.2Hz,2H,CH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),4.74(d,J=2.4Hz,2H,OCH2),6.55(d,J=2.1Hz,1H,Ph-8-H),6.65(dd,J=8.7,2.1Hz,1H,Ph-6-H),7.85(d,J=8.7Hz,1H,Ph-5-H),8.53(s,1H,Pyrimidine 2-H).Compound 14-(275): Oil. δ(CDCl 3 ): 1.28 (t, J=7.2 Hz, 3H, CH 3 ), 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.58 (d, J = 2.1Hz, 1H, CH), 2.78(s, 2H, COCH 2 ), 2.88(q, J=7.2Hz, 2H, CH 2 ), 3.49(td, 2H, Piperidine-CH 2 ), 4.20(d, 2H, Piperidine-CH 2 ), 4.74 (d, J=2.4 Hz, 2H, OCH 2 ), 6.55 (d, J=2.1 Hz, 1H, Ph-8-H), 6.65 (dd, J=8.7, 2.1 Hz,1H,Ph-6-H),7.85(d,J=8.7Hz,1H,Ph-5-H),8.53(s,1H,Pyrimidine 2-H).
化合物15-(275):熔点156.7℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.58(d,J=2.1Hz,1H,CH),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),4.73(d,J=2.1Hz,2H,OCH2),6.55(d,J=2.1Hz,1H,Ph-8-H),6.65(dd,J=8.7,2.1Hz,1H,Ph-6-H),6.83(t,J=53.1Hz,1H,CHF2),7.85(d,J=8.7Hz,1H,Ph-5-H),8.64(s,1H,Pyrimidine 2-H).Compound 15-(275): melting point 156.7°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.58 (d, J=2.1 Hz, 1H, CH ), 2.78 (s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 4.73 (d, J=2.1Hz, 2H, OCH 2 ), 6.55 (d, J=2.1 Hz, 1H, Ph-8-H), 6.65 (dd, J=8.7, 2.1 Hz, 1H, Ph-6-H), 6.83 (t, J=53.1 Hz, 1H, CHF 2 ), 7.85 (d, J=8.7Hz, 1H, Ph-5-H), 8.64(s, 1H, Pyrimidine 2-H).
化合物4-(1):熔点172.0℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.75(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.54(s,1H,Pyrimidine 5-H),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.37(s,1H,Pyrimidine2-H).Compound 4-(1): melting point 172.0°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.75 (s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ) ), 4.20(d, 2H, Piperidine-CH 2 ), 6.54(s, 1H, Pyrimidine 5-H), 7.03(t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.52(td, J=7.5, 1.5Hz, 1H, Ph-7-H), 7.89 (dd, J=7.5, 1.5Hz, 1H, Ph-5-H), 8.37 (s, 1H, Pyrimidine2-H).
化合物5-(1):熔点153.5℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.25(s,3H,CH3),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.39(s,1H,Pyrimidine 2-H).Compound 5-(1): melting point 153.5°C. δ(CDCl 3 ): 1.83(td, 2H, Piperidine-CH 2 ), 2.20(dd, 2H, Piperidine-CH 2 ), 2.25(s, 3H, CH 3 ), 2.78(s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 7.03 (t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.52 (td, J= 7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.39(s,1H,Pyrimidine 2-H).
化合物9-(1):熔点157.6℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.78(s,2H,COCH2),3.32(td,2H,Piperidine-CH2),3.63(d,2H,Piperidine-CH2),3.86(s,2H,NH2),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.16(s,1H,Pyrimidine 2-H).Compound 9-(1): melting point 157.6°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.78 (s, 2H, COCH 2 ), 3.32 (td, 2H, Piperidine-CH 2 ) ), 3.63 (d, 2H, Piperidine-CH 2 ), 3.86 (s, 2H, NH 2 ), 7.03 (t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.52 (td, J= 7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.16(s,1H,Pyrimidine 2-H).
化合物11-(1):熔点172.6℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.67(s,2H,COCH2),3.32(td,2H,Piperidine-CH2),3.67(d,2H,Piperidine-CH2),6.98(t,J=7.5Hz,2H,Ph-6,8-2H),7.20(d,J=9.3Hz,2H,4-Br-Ph-3,5-2H),7.47(td,J=7.5,1.5Hz,1H,Ph-7-H),7.20(d,J=9.3Hz,2H,4-Br-Ph-2,6-2H),7.83(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.43(s,1H,Pyrimidine 2-H).Compound 11-(1): melting point 172.6°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.67 (s, 2H, COCH 2 ), 3.32 (td, 2H, Piperidine-CH 2 ) ), 3.67(d, 2H, Piperidine-CH 2 ), 6.98(t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.20(d, J=9.3Hz, 2H, 4-Br-Ph -3,5-2H),7.47(td,J=7.5,1.5Hz,1H,Ph-7-H),7.20(d,J=9.3Hz,2H,4-Br-Ph-2,6-2H ), 7.83(dd, J=7.5, 1.5Hz, 1H, Ph-5-H), 8.43(s, 1H, Pyrimidine 2-H).
化合物8-(1):熔点142.7℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.78(s,2H,COCH2),3.32(td,2H,Piperidine-CH2),3.63(d,2H,Piperidine-CH2),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.37(s,1H,Pyrimidine 2-H).Compound 8-(1): melting point 142.7°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.78 (s, 2H, COCH 2 ), 3.32 (td, 2H, Piperidine-CH 2 ) ), 3.63 (d, 2H, Piperidine-CH 2 ), 7.03 (t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.52 (td, J=7.5, 1.5Hz, 1H, Ph-7 -H), 7.89(dd, J=7.5, 1.5Hz, 1H, Ph-5-H), 8.37(s, 1H, Pyrimidine 2-H).
化合物7-(1):熔点183.5℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.77(s,2H,COCH2),3.32(td,2H,Piperidine-CH2),3.63(d,2H,Piperidine-CH2),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.36(s,1H,Pyrimidine 2-H),10.33(s,1H,CHO).Compound 7-(1): melting point 183.5°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.77 (s, 2H, COCH 2 ), 3.32 (td, 2H, Piperidine-CH 2 ) ), 3.63 (d, 2H, Piperidine-CH 2 ), 7.03 (t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.52 (td, J=7.5, 1.5Hz, 1H, Ph-7 -H), 7.89(dd, J=7.5, 1.5Hz, 1H, Ph-5-H), 8.36(s, 1H, Pyrimidine 2-H), 10.33(s, 1H, CHO).
化合物10-(1):熔点196.0℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.76(s,2H,COCH2),3.32(td,2H,Piperidine-CH2),3.63(d,2H,Piperidine-CH2),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.38(s,1H,Pyrimidine 2-H).Compound 10-(1): melting point 196.0°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.76 (s, 2H, COCH 2 ), 3.32 (td, 2H, Piperidine-CH 2 ) ), 3.63 (d, 2H, Piperidine-CH 2 ), 7.03 (t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.52 (td, J=7.5, 1.5Hz, 1H, Ph-7 -H), 7.89(dd, J=7.5, 1.5Hz, 1H, Ph-5-H), 8.38(s, 1H, Pyrimidine 2-H).
化合物12-(1):熔点168.1℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.78(s,2H,COCH2),3.32(td,2H,Piperidine-CH2),3.63(d,2H,Piperidine-CH2),12.86(s,1H,COOH),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.38(s,1H,Pyrimidine2-H).Compound 12-(1): melting point 168.1°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.78 (s, 2H, COCH 2 ), 3.32 (td, 2H, Piperidine-CH 2 ) ), 3.63 (d, 2H, Piperidine-CH 2 ), 12.86 (s, 1H, COOH), 7.03 (t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.52 (td, J=7.5 ,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.38(s,1H,Pyrimidine2-H).
化合物6-(1):熔点166.3℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.75(s,2H,COCH2),3.32(td,2H,Piperidine-CH2),3.63(d,2H,Piperidine-CH2),3.74(s,3H,OCH3),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.18(s,1H,Pyrimidine2-H).Compound 6-(1): melting point 166.3°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.75 (s, 2H, COCH 2 ), 3.32 (td, 2H, Piperidine-CH 2 ) ), 3.63 (d, 2H, Piperidine-CH 2 ), 3.74 (s, 3H, OCH 3 ), 7.03 (t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.52 (td, J= 7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.18(s,1H,Pyrimidine2-H).
化合物17-274:油状物.δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.58(d,J=2.1Hz,1H,CH),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),4.73(d,J=2.1Hz,2H,OCH2),6.55(d,J=2.1Hz,1H,Ph-8-H),6.65(dd,J=8.7,2.1Hz,1H,Ph-6-H),7.23(s,1H,SCH),7.85(d,J=8.7Hz,1H,Ph-5-H),8.64(s,1H,Pyrimidine 2-H).Compounds 17-274: Oil. δ (CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.58 (d, J=2.1 Hz, 1H, CH ), 2.78(s, 2H, COCH 2 ), 3.49(td, 2H, Piperidine-CH 2 ), 4.20(d, 2H, Piperidine-CH 2 ), 4.73(d, J=2.1Hz, 2H, OCH 2 ) ,6.55(d,J=2.1Hz,1H,Ph-8-H),6.65(dd,J=8.7,2.1Hz,1H,Ph-6-H),7.23(s,1H,SCH),7.85( d, J=8.7Hz, 1H, Ph-5-H), 8.64(s, 1H, Pyrimidine 2-H).
化合物16-(1):油状物.δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.81(s,2H,COCH2),3.32(td,2H,Piperidine-CH2),3.63(d,2H,Piperidine-CH2),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.44-7.56(m,2H,Ph-2H),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.86-7.91(m,2H,Ph-2H),7.95(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.75(s,1H,Pyrimidine 2-H).Compound 16-(1): Oil. δ(CDCl 3 ): 1.83(td, 2H, Piperidine-CH 2 ), 2.20(dd, 2H, Piperidine-CH 2 ), 2.81(s, 2H, COCH 2 ), 3.32 (td, 2H, Piperidine-CH 2 ), 3.63 (d, 2H, Piperidine-CH 2 ), 7.03 (t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.44-7.56 (m, 2H, Ph-2H), 7.52 (td, J=7.5, 1.5Hz, 1H, Ph-7-H), 7.86-7.91 (m, 2H, Ph-2H), 7.95 (dd, J=7.5, 1.5Hz ,1H,Ph-5-H),8.75(s,1H,Pyrimidine 2-H).
化合物17-(1):熔点108.4℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.80(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),7.03(t,J=7.5Hz,2H,Ph-6,8-2H),7.23(s,1H,SCH),7.52(td,J=7.5,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.58(s,1H,Pyrimidine2-H).Compound 17-(1): melting point 108.4°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.80 (s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ) ), 4.20 (d, 2H, Piperidine-CH 2 ), 7.03 (t, J=7.5Hz, 2H, Ph-6, 8-2H), 7.23 (s, 1H, SCH), 7.52 (td, J=7.5 ,1.5Hz,1H,Ph-7-H),7.89(dd,J=7.5,1.5Hz,1H,Ph-5-H),8.58(s,1H,Pyrimidine2-H).
化合物5-(3):油状物.δ(CDCl3):δ1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.25(s,3H,CH3),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.99(dd,J=9.0,4.2Hz,1H,Ph-8-H),7.25(dd,J=9.0,3.0Hz,1H,Ph-7-H),7.53(dd,J=8.1,3.0Hz,1H,Ph-5-H),8.64(s,1H,Pyrimidine 2-H).Compound 5-(3): Oil. δ(CDCl 3 ): δ1.83(td, 2H, Piperidine-CH 2 ), 2.20(dd, 2H, Piperidine-CH 2 ), 2.25(s, 3H, CH 3 ), 2.78(s, 2H, COCH 2 ), 3.49(td, 2H, Piperidine-CH 2 ), 4.20(d, 2H, Piperidine-CH 2 ), 6.99(dd, J=9.0, 4.2Hz, 1H, Ph -8-H),7.25(dd,J=9.0,3.0Hz,1H,Ph-7-H),7.53(dd,J=8.1,3.0Hz,1H,Ph-5-H),8.64(s, 1H, Pyrimidine 2-H).
化合物12-(3):油状物.δ(CDCl3):δ1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.99(dd,J=9.0,4.2Hz,1H,Ph-8-H),7.25(dd,J=9.0,3.0Hz,1H,Ph-7-H),7.53(dd,J=8.1,3.0Hz,1H,Ph-5-H),8.64(s,1H,Pyrimidine 2-H).Compound 12-(3): Oil. δ(CDCl 3 ): δ1.83(td, 2H, Piperidine-CH 2 ), 2.20(dd, 2H, Piperidine-CH 2 ), 2.78(s, 2H, COCH 2 ), 3.49(td, 2H, Piperidine-CH 2 ), 4.20(d, 2H, Piperidine-CH 2 ), 6.99(dd, J=9.0, 4.2Hz, 1H, Ph-8-H), 7.25(dd, J=9.0, 3.0Hz, 1H, Ph-7-H), 7.53 (dd, J=8.1, 3.0Hz, 1H, Ph-5-H), 8.64 (s, 1H, Pyrimidine 2-H).
化合物8-(3):熔点162.6℃。δ(CDCl3):δ1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.99(dd,J=9.0,4.2Hz,1H,Ph-8-H),7.25(dd,J=9.0,3.0Hz,1H,Ph-7-H),7.53(dd,J=8.1,3.0Hz,1H,Ph-5-H),8.64(s,1H,Pyrimidine 2-H).Compound 8-(3): melting point 162.6°C. δ(CDCl 3 ): δ1.83(td,2H,Piperidine- CH2 ),2.20(dd,2H,Piperidine- CH2 ),2.78(s,2H, COCH2 ),3.49(td,2H,Piperidine- CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 6.99 (dd, J=9.0, 4.2Hz, 1H, Ph-8-H), 7.25 (dd, J=9.0, 3.0Hz, 1H, Ph -7-H), 7.53(dd, J=8.1, 3.0Hz, 1H, Ph-5-H), 8.64(s, 1H, Pyrimidine 2-H).
化合物7-(3):熔点133.0℃。δ(CDCl3):δ1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.99(dd,J=9.0,4.2Hz,1H,Ph-8-H),7.25(dd,J=9.0,3.0Hz,1H,Ph-7-H),7.53(dd,J=8.1,3.0Hz,1H,Ph-5-H),8.64(s,1H,Pyrimidine 2-H),10.33(s,1H,CHO).Compound 7-(3): melting point 133.0°C. δ(CDCl 3 ): δ1.83(td,2H,Piperidine- CH2 ),2.20(dd,2H,Piperidine- CH2 ),2.78(s,2H, COCH2 ),3.49(td,2H,Piperidine- CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 6.99 (dd, J=9.0, 4.2Hz, 1H, Ph-8-H), 7.25 (dd, J=9.0, 3.0Hz, 1H, Ph -7-H), 7.53(dd, J=8.1, 3.0Hz, 1H, Ph-5-H), 8.64(s, 1H, Pyrimidine 2-H), 10.33(s, 1H, CHO).
化合物6-(3):油状物.δ(CDCl3):δ1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),3.74(s,3H,OCH3),4.20(d,2H,Piperidine-CH2),6.99(dd,J=9.0,4.2Hz,1H,Ph-8-H),7.25(dd,J=9.0,3.0Hz,1H,Ph-7-H),7.53(dd,J=8.1,3.0Hz,1H,Ph-5-H),8.64(s,1H,Pyrimidine 2-H).Compound 6-(3): Oil. δ(CDCl 3 ): δ1.83(td, 2H, Piperidine-CH 2 ), 2.20(dd, 2H, Piperidine-CH 2 ), 2.78(s, 2H, COCH 2 ), 3.49(td, 2H, Piperidine-CH 2 ), 3.74(s, 3H, OCH 3 ), 4.20(d, 2H, Piperidine-CH 2 ), 6.99(dd, J=9.0, 4.2Hz, 1H, Ph -8-H),7.25(dd,J=9.0,3.0Hz,1H,Ph-7-H),7.53(dd,J=8.1,3.0Hz,1H,Ph-5-H),8.64(s, 1H, Pyrimidine 2-H).
化合物13-(56):熔点166.0℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.32(s,3H,Ph-CH3),2.53(s,3H,CH3),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.92(d,J=8.4Hz,1H,Ph-8-H),7.33(d,J=8.4Hz,1H,Ph-7-H),7.43-7.48(m,2H,Ph-2H),7.68(s,1H,Ph-5-H),7.71-7.77(m,1H,Ph-H),7.77-7.92(m,2H,Ph-2H),8.75(s,1H,Pyrimidine 2-H).Compound 13-(56): melting point 166.0°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.32 (s, 3H, Ph-CH 3 ), 2.53 (s, 3H, CH 3 ) ), 2.78(s, 2H, COCH 2 ), 3.49(td, 2H, Piperidine-CH 2 ), 4.20(d, 2H, Piperidine-CH 2 ), 6.92(d, J=8.4Hz, 1H, Ph-8 -H), 7.33(d, J=8.4Hz, 1H, Ph-7-H), 7.43-7.48(m, 2H, Ph-2H), 7.68(s, 1H, Ph-5-H), 7.71- 7.77(m,1H,Ph-H),7.77-7.92(m,2H,Ph-2H),8.75(s,1H,Pyrimidine 2-H).
化合物15-(56):熔点177.5℃。δ(CDCl3):δ1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.32(s,3H,Ph-CH3),2.78(s,2H,COCH2),3.49(td,2H,Piperidine-CH2),4.20(d,2H,Piperidine-CH2),6.83(t,J=53.1Hz,1H,CHF2),6.92(d,J=8.4Hz,1H,Ph-8-H),7.33(d,J=8.4Hz,1H,Ph-7-H),7.43-7.48(m,2H,Ph-2H),7.68(s,1H,Ph-5-H),7.71-7.77(m,1H,Ph-H),7.77-7.92(m,2H,Ph-2H),8.75(s,1H,Pyrimidine2-H).Compound 15-(56): melting point 177.5°C. δ(CDCl 3 ): δ1.83(td, 2H, Piperidine-CH 2 ), 2.20(dd, 2H, Piperidine-CH 2 ), 2.32(s, 3H, Ph-CH 3 ), 2.78(s, 2H, COCH 2 ), 3.49 (td, 2H, Piperidine-CH 2 ), 4.20 (d, 2H, Piperidine-CH 2 ), 6.83 (t, J=53.1 Hz, 1H, CHF 2 ), 6.92 (d, J=8.4 Hz,1H,Ph-8-H),7.33(d,J=8.4Hz,1H,Ph-7-H),7.43-7.48(m,2H,Ph-2H),7.68(s,1H,Ph- 5-H),7.71-7.77(m,1H,Ph-H),7.77-7.92(m,2H,Ph-2H),8.75(s,1H,Pyrimidine2-H).
化合物16-(56):熔点281.0℃。δ(CDCl3):1.83(td,2H,Piperidine-CH2),2.20(dd,2H,Piperidine-CH2),2.32(s,3H,Ph-CH3),2.81(s,2H,COCH2),3.32(td,2H,Piperidine-CH2),3.63(d,2H,Piperidine-CH2),6.92(d,J=8.4Hz,1H,Ph-8-H),7.33(d,J=8.4Hz,1H,Ph-7-H),7.43-7.48(m,2H,Ph-2H),7.68(s,1H,Ph-5-H),7.71-7.77(m,1H,Ph-H),7.77-7.92(m,2H,Ph-2H),8.75(s,1H,Pyrimidine 2-H).Compound 16-(56): melting point 281.0°C. δ(CDCl 3 ): 1.83 (td, 2H, Piperidine-CH 2 ), 2.20 (dd, 2H, Piperidine-CH 2 ), 2.32 (s, 3H, Ph-CH 3 ), 2.81 (s, 2H, COCH 2 ), 3.32(td, 2H, Piperidine-CH 2 ), 3.63(d, 2H, Piperidine-CH 2 ), 6.92(d, J=8.4Hz, 1H, Ph-8-H), 7.33(d, J= 8.4Hz,1H,Ph-7-H),7.43-7.48(m,2H,Ph-2H),7.68(s,1H,Ph-5-H),7.71-7.77(m,1H,Ph-H) ,7.77-7.92(m,2H,Ph-2H),8.75(s,1H,Pyrimidine 2-H).
生物活性测定实施例Biological Activity Assay Example
本发明化合物对农业领域中的多种病菌表现出较好的活性。The compounds of the present invention show good activity against various pathogens in the agricultural field.
实施例8:杀菌活性测定Example 8: Determination of Bactericidal Activity
用本发明化合物样品对植物的多种真菌病害进行了离体抑菌活性或活体保护效果试验。杀菌活性测定结果见以下各实施例。The in vitro antibacterial activity or in vivo protection effect test was carried out with the compound samples of the present invention against various fungal diseases of plants. The results of the bactericidal activity assay are shown in the following examples.
(1)离体杀菌活性测定(1) Determination of bactericidal activity in vitro
测定方法如下:采用高通量筛选方法,即将待测化合物样品用适合的溶剂(溶剂的种类如丙酮、甲醇、DMF等,并且依据其对样品的溶解能力而选择)溶解,配制成所需浓度待测液。在超净工作环境下,将待测液加入到96孔培养板的微孔中,再将病原菌繁殖体悬浮液加入其中,处理后的培养板放置在恒温培养箱中培养。24小时后进行调查,调查时目测病原菌繁殖体萌发或生长情况,并根据对照处理的萌发或生长情况,评价化合物抑菌活性。The determination method is as follows: using a high-throughput screening method, that is, the compound sample to be tested is dissolved in a suitable solvent (the type of solvent such as acetone, methanol, DMF, etc., and is selected according to its ability to dissolve the sample), and the desired concentration is prepared. liquid to be tested. In an ultra-clean working environment, the liquid to be tested is added to the microwells of a 96-well culture plate, and then the pathogenic bacteria propagule suspension is added to it, and the treated culture plate is placed in a constant temperature incubator for cultivation. An investigation was conducted 24 hours later. During the investigation, the germination or growth of the propagules of the pathogenic bacteria was visually observed, and the bacteriostatic activity of the compounds was evaluated according to the germination or growth of the control treatment.
部分化合物的离体抑菌活性(以抑制率表示)测试结果如下:The test results of in vitro antibacterial activity (expressed as inhibition rate) of some compounds are as follows:
对稻瘟病菌的抑制率:Inhibition rate against rice blast fungus:
在25ppm剂量下,化合物:13-(17)、15-(1)、14-(3)、5-(1)、10-(1)、13-(3)、16-(56)、7-(1)、6-(1)、17-(1)、16-(1)、4-(1)等对稻瘟病的抑制率在80%以上。At 25ppm dose, compounds: 13-(17), 15-(1), 14-(3), 5-(1), 10-(1), 13-(3), 16-(56), 7 -(1), 6-(1), 17-(1), 16-(1), 4-(1), etc., the inhibition rate of rice blast is more than 80%.
(2)活体保护活性测定(2) Determination of in vivo protective activity
测定方法如下:采用活体盆栽测定方法,即将待测化合物样品用少量溶剂(溶剂的种类如丙酮、甲醇、DMF等,并且依据其对样品的溶解能力而选择,溶剂量与喷液量的体积比等于或小于0.05)溶解,用含有0.1%吐温80的水稀释,配制成所需浓度待测液。在作物喷雾机上,将待测液喷施于病害寄主植物上(寄主植物为在温室内培养的标准盆栽苗),24小时后进行病害接种。依据病害特点,将需要控温保湿培养的病害植物接种后放在人工气候室中培养,待病害完成侵染后,移入温室培养,将不需要保湿培养的病害植物直接在温室内接种并培养。待对照充分发病后(通常为一周时间)进行化合物防病效果评估。The determination method is as follows: adopt the in vivo pot assay method, that is, use a small amount of solvent (the type of solvent such as acetone, methanol, DMF, etc.) for the sample of the compound to be tested, and select it according to its dissolving ability to the sample, and the volume ratio of the solvent amount to the spray liquid amount. equal to or less than 0.05) dissolved, diluted with water containing 0.1% Tween 80, and prepared into a liquid to be tested with the required concentration. On a crop sprayer, the liquid to be tested is sprayed on the diseased host plants (the host plants are standard potted seedlings cultivated in a greenhouse), and disease inoculation is carried out 24 hours later. According to the characteristics of the disease, the diseased plants that need temperature control and moisturizing cultivation are inoculated and cultivated in an artificial climate chamber. After the disease is infected, they are transferred to the greenhouse for cultivation, and the diseased plants that do not require moisturizing cultivation are directly inoculated and cultivated in the greenhouse. After the control is fully developed (usually one week), the evaluation of the compound's disease prevention effect is carried out.
部分化合物的活体保护活性测试结果如下:The in vivo protective activity test results of some compounds are as follows:
对黄瓜霜霉病的活体保护活性:In vivo protective activity against cucumber downy mildew:
在400ppm剂量下,本发明通式Ⅰ化合物对黄瓜霜霉病防效在80%以上,其中部分化合物:15-(1)、14-(3)、13-(274)、13-(3)、6-(1)、17-(1)、16-(1)、13-(275)、11-(1)等对黄瓜霜霉病防效为100%,而化合物CK1,CK2对黄瓜霜霉病防效分别为0和50%;At a dose of 400 ppm, the compound of general formula I of the present invention has a control effect of more than 80% on cucumber downy mildew, and some of the compounds are: 15-(1), 14-(3), 13-(274), 13-(3) , 6-(1), 17-(1), 16-(1), 13-(275), 11-(1), etc. were 100% effective against cucumber downy mildew, while compounds CK1 and CK2 were effective against cucumber frost. Mildew control efficacy was 0 and 50%, respectively;
对小麦白粉病的活体保护活性:In vivo protective activity against wheat powdery mildew:
在400ppm剂量下,化合物:15-(1)、12-(1)、14-(3)、13-(3)、8-(1)、7-(1)、6-(1)、17-(1)、16-(1)、14-(274)、11-(1)等对小麦白粉病防效在80%以上;At 400 ppm dose, compounds: 15-(1), 12-(1), 14-(3), 13-(3), 8-(1), 7-(1), 6-(1), 17 -(1), 16-(1), 14-(274), 11-(1), etc., the control effect on wheat powdery mildew is more than 80%;
对玉米锈病的活体保护活性:In vivo protective activity against corn rust:
在400ppm剂量下,化合物:13-(17)、15-(1)、12-(1)、14-(3)、13-(1)、8-(1)、7-(1)、6-(1)、17-(1)、16-(1)、14-(274)、14-(1)等对玉米锈病防效在80%以上;At 400 ppm dose, compounds: 13-(17), 15-(1), 12-(1), 14-(3), 13-(1), 8-(1), 7-(1), 6 -(1), 17-(1), 16-(1), 14-(274), 14-(1), etc., the control effect on corn rust is more than 80%;
对黄瓜炭疽病的活体保护活性:In vivo protective activity against cucumber anthracnose:
在400ppm剂量下,化合物:17-(274)、13-(3)、11-(1)等对黄瓜炭疽病防效在80%以上。At a dose of 400 ppm, the compounds: 17-(274), 13-(3), 11-(1), etc., had more than 80% control effects on cucumber anthracnose.
Claims (10)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811013143.0A CN110872302B (en) | 2018-08-31 | 2018-08-31 | Pyrimidine-containing spiro-compound and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811013143.0A CN110872302B (en) | 2018-08-31 | 2018-08-31 | Pyrimidine-containing spiro-compound and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN110872302A true CN110872302A (en) | 2020-03-10 |
| CN110872302B CN110872302B (en) | 2022-09-27 |
Family
ID=69715269
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201811013143.0A Active CN110872302B (en) | 2018-08-31 | 2018-08-31 | Pyrimidine-containing spiro-compound and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN110872302B (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN116102565A (en) * | 2023-01-16 | 2023-05-12 | 贵州大学 | A class of sirochromone compounds containing isopropanolamine and its preparation method and application |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1351497A (en) * | 1999-04-01 | 2002-05-29 | 辉瑞产品公司 | Aminopyrimidines as sorbitol dehydrogenase inhibitors |
| CN101309923A (en) * | 2005-11-14 | 2008-11-19 | 索尔瓦药物有限公司 | N-sulfamoyl-N -benzopyranpiperidines as inhibitors of carbonic anhydrases |
| WO2011141474A1 (en) * | 2010-05-12 | 2011-11-17 | Boehringer Ingelheim International Gmbh | Novel ccr2 receptor antagonists, method for producing the same, and use thereof as medicaments |
-
2018
- 2018-08-31 CN CN201811013143.0A patent/CN110872302B/en active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1351497A (en) * | 1999-04-01 | 2002-05-29 | 辉瑞产品公司 | Aminopyrimidines as sorbitol dehydrogenase inhibitors |
| CN101309923A (en) * | 2005-11-14 | 2008-11-19 | 索尔瓦药物有限公司 | N-sulfamoyl-N -benzopyranpiperidines as inhibitors of carbonic anhydrases |
| WO2011141474A1 (en) * | 2010-05-12 | 2011-11-17 | Boehringer Ingelheim International Gmbh | Novel ccr2 receptor antagonists, method for producing the same, and use thereof as medicaments |
Non-Patent Citations (1)
| Title |
|---|
| AHMED H. ABDELAZEEM,等: "Novel pyrazolopyrimidine derivatives targeting COXs and iNOS enzymes; design, synthesis and biological evaluation as potential anti-inflammatory agents", 《EUROPEAN JOURNAL OF PHARMACEUTICAL SCIENCES》 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN116102565A (en) * | 2023-01-16 | 2023-05-12 | 贵州大学 | A class of sirochromone compounds containing isopropanolamine and its preparation method and application |
Also Published As
| Publication number | Publication date |
|---|---|
| CN110872302B (en) | 2022-09-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101875633B (en) | Pyrazolyl acrylonitrile compound and application thereof | |
| CN104710436B (en) | A kind of pyrazolyl pyrimidines aminated compounds and purposes | |
| JP2011506465A (en) | Bactericidal bicyclic pyrazole | |
| SK15898A3 (en) | Halobenzimidazoles and their use as microbicides | |
| CN101084224B (en) | Thieno-pyrimidine compounds having fungicidal activity | |
| CN108069984B (en) | Substituted five-membered heterocyclic compound containing pyrimido ring and preparation method and application thereof | |
| CN102993100B (en) | Substituted pyrazole (sulphur) ketone compound and application thereof | |
| CN108059629B (en) | Pyrimidine-containing substituted pyrazole compound and preparation method and application thereof | |
| CN110317200A (en) | Pyrimidine compound and application thereof | |
| CN110452167B (en) | Quinoline derivatives and preparation method and use thereof | |
| CN110872302B (en) | Pyrimidine-containing spiro-compound and application thereof | |
| CN116063285B (en) | Substituted pyrimidine (thio) ester compounds and uses thereof | |
| CN112745312B (en) | Pyridazinone isoxazole methyl ether compound and application thereof | |
| CN103288855A (en) | Isothiazole-o-pyrimidone compound and application thereof | |
| CN102827034B (en) | P-dicyanoaniline-containing compounds and applications thereof | |
| CN102827033B (en) | Compound containing o-dicyanoaniline and application thereof | |
| CN113549053B (en) | Pyrazoloquine (azolyl) ether compound and application thereof | |
| CN110396083B (en) | Pyridazinonyl-contained butenolide compound and application thereof | |
| CN112745260B (en) | Amide compound containing substituted acetophenone structural fragment and preparation method and application thereof | |
| CN110964037B (en) | Pyrimidine-fused ring-containing compound and preparation method and application thereof | |
| CN105777640B (en) | A kind of pyrazole cyclohexanediol ether compound and its application | |
| CN107540664B (en) | Pyrazolyl pyrimidine ether compound and application thereof | |
| CN115232083B (en) | Substituted isoxazole ethylamine compounds and preparation method and use thereof | |
| CN112741095A (en) | Application of pyridazinone isoxazole methyl sulfide compound as bactericide | |
| CN112778232B (en) | Oxime ether compound and application thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |