CN110470768B - Method for measuring residual amounts of pyrazosulfuron-ethyl, triazophos and butachlor in water - Google Patents
Method for measuring residual amounts of pyrazosulfuron-ethyl, triazophos and butachlor in water Download PDFInfo
- Publication number
- CN110470768B CN110470768B CN201910794199.2A CN201910794199A CN110470768B CN 110470768 B CN110470768 B CN 110470768B CN 201910794199 A CN201910794199 A CN 201910794199A CN 110470768 B CN110470768 B CN 110470768B
- Authority
- CN
- China
- Prior art keywords
- standard
- triazophos
- pyrazosulfuron
- butachlor
- ethyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- HKPHPIREJKHECO-UHFFFAOYSA-N butachlor Chemical compound CCCCOCN(C(=O)CCl)C1=C(CC)C=CC=C1CC HKPHPIREJKHECO-UHFFFAOYSA-N 0.000 title claims abstract description 39
- AMFGTOFWMRQMEM-UHFFFAOYSA-N triazophos Chemical compound N1=C(OP(=S)(OCC)OCC)N=CN1C1=CC=CC=C1 AMFGTOFWMRQMEM-UHFFFAOYSA-N 0.000 title claims abstract description 39
- BGNQYGRXEXDAIQ-UHFFFAOYSA-N Pyrazosulfuron-ethyl Chemical group C1=NN(C)C(S(=O)(=O)NC(=O)NC=2N=C(OC)C=C(OC)N=2)=C1C(=O)OCC BGNQYGRXEXDAIQ-UHFFFAOYSA-N 0.000 title claims abstract description 38
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 title claims abstract description 32
- 238000000034 method Methods 0.000 title claims abstract description 28
- 239000012224 working solution Substances 0.000 claims abstract description 11
- 238000001514 detection method Methods 0.000 claims abstract description 10
- 238000010811 Ultra-Performance Liquid Chromatography-Tandem Mass Spectrometry Methods 0.000 claims abstract description 7
- 238000010812 external standard method Methods 0.000 claims abstract description 5
- 238000012360 testing method Methods 0.000 claims abstract description 4
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 36
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 claims description 21
- 150000002500 ions Chemical class 0.000 claims description 19
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 claims description 12
- 239000000243 solution Substances 0.000 claims description 12
- 239000007788 liquid Substances 0.000 claims description 11
- 239000011550 stock solution Substances 0.000 claims description 11
- 238000001035 drying Methods 0.000 claims description 10
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 9
- 239000012086 standard solution Substances 0.000 claims description 9
- 238000012544 monitoring process Methods 0.000 claims description 7
- 239000007789 gas Substances 0.000 claims description 6
- 238000002156 mixing Methods 0.000 claims description 6
- 238000002360 preparation method Methods 0.000 claims description 6
- 239000011780 sodium chloride Substances 0.000 claims description 6
- RQBBFKINEJYDOB-UHFFFAOYSA-N acetic acid;acetonitrile Chemical compound CC#N.CC(O)=O RQBBFKINEJYDOB-UHFFFAOYSA-N 0.000 claims description 5
- 238000010828 elution Methods 0.000 claims description 5
- 238000000605 extraction Methods 0.000 claims description 5
- 238000005259 measurement Methods 0.000 claims description 5
- 239000012528 membrane Substances 0.000 claims description 5
- 239000002352 surface water Substances 0.000 claims description 5
- 238000001819 mass spectrum Methods 0.000 claims description 4
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 claims description 4
- 229910052757 nitrogen Inorganic materials 0.000 claims description 4
- 239000007864 aqueous solution Substances 0.000 claims description 3
- 238000006243 chemical reaction Methods 0.000 claims description 3
- AJEHNBIPLQJTNU-UHFFFAOYSA-N cyanomethyl acetate Chemical compound CC(=O)OCC#N AJEHNBIPLQJTNU-UHFFFAOYSA-N 0.000 claims description 3
- HQVFCQRVQFYGRJ-UHFFFAOYSA-N formic acid;hydrate Chemical compound O.OC=O HQVFCQRVQFYGRJ-UHFFFAOYSA-N 0.000 claims description 3
- 239000012074 organic phase Substances 0.000 claims description 3
- 239000012071 phase Substances 0.000 claims description 3
- 238000005185 salting out Methods 0.000 claims description 3
- 238000001195 ultra high performance liquid chromatography Methods 0.000 claims description 3
- 239000003643 water by type Substances 0.000 claims description 3
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 claims description 2
- 238000001914 filtration Methods 0.000 claims description 2
- 235000019253 formic acid Nutrition 0.000 claims description 2
- GBMDVOWEEQVZKZ-UHFFFAOYSA-N methanol;hydrate Chemical compound O.OC GBMDVOWEEQVZKZ-UHFFFAOYSA-N 0.000 claims description 2
- 238000005303 weighing Methods 0.000 claims description 2
- 238000011002 quantification Methods 0.000 abstract description 3
- 239000000523 sample Substances 0.000 description 26
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 9
- 239000000126 substance Substances 0.000 description 8
- 239000004009 herbicide Substances 0.000 description 5
- 241000196324 Embryophyta Species 0.000 description 4
- 240000007594 Oryza sativa Species 0.000 description 3
- 235000007164 Oryza sativa Nutrition 0.000 description 3
- 238000004949 mass spectrometry Methods 0.000 description 3
- 235000009566 rice Nutrition 0.000 description 3
- 229920000742 Cotton Polymers 0.000 description 2
- 241000607479 Yersinia pestis Species 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 238000007664 blowing Methods 0.000 description 2
- 238000011097 chromatography purification Methods 0.000 description 2
- 238000010899 nucleation Methods 0.000 description 2
- 150000007524 organic acids Chemical group 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 239000000575 pesticide Substances 0.000 description 2
- 230000035945 sensitivity Effects 0.000 description 2
- 239000002689 soil Substances 0.000 description 2
- 241000238876 Acari Species 0.000 description 1
- 235000017060 Arachis glabrata Nutrition 0.000 description 1
- 244000105624 Arachis hypogaea Species 0.000 description 1
- 235000010777 Arachis hypogaea Nutrition 0.000 description 1
- 235000018262 Arachis monticola Nutrition 0.000 description 1
- 235000016068 Berberis vulgaris Nutrition 0.000 description 1
- 241000335053 Beta vulgaris Species 0.000 description 1
- 240000007124 Brassica oleracea Species 0.000 description 1
- 235000003899 Brassica oleracea var acephala Nutrition 0.000 description 1
- 235000011301 Brassica oleracea var capitata Nutrition 0.000 description 1
- 235000001169 Brassica oleracea var oleracea Nutrition 0.000 description 1
- -1 Chinese alias Chemical compound 0.000 description 1
- 241000219146 Gossypium Species 0.000 description 1
- 240000005979 Hordeum vulgare Species 0.000 description 1
- 235000007340 Hordeum vulgare Nutrition 0.000 description 1
- 241000255777 Lepidoptera Species 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 229940100389 Sulfonylurea Drugs 0.000 description 1
- 241000209140 Triticum Species 0.000 description 1
- 235000021307 Triticum Nutrition 0.000 description 1
- 210000001015 abdomen Anatomy 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 230000000895 acaricidal effect Effects 0.000 description 1
- 239000000642 acaricide Substances 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 239000012491 analyte Substances 0.000 description 1
- ZOMSMJKLGFBRBS-UHFFFAOYSA-N bentazone Chemical compound C1=CC=C2NS(=O)(=O)N(C(C)C)C(=O)C2=C1 ZOMSMJKLGFBRBS-UHFFFAOYSA-N 0.000 description 1
- 235000013339 cereals Nutrition 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 229910001873 dinitrogen Inorganic materials 0.000 description 1
- 235000013399 edible fruits Nutrition 0.000 description 1
- 230000035784 germination Effects 0.000 description 1
- 239000005337 ground glass Substances 0.000 description 1
- 230000002363 herbicidal effect Effects 0.000 description 1
- 238000001294 liquid chromatography-tandem mass spectrometry Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 239000005645 nematicide Substances 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 235000020232 peanut Nutrition 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 239000012488 sample solution Substances 0.000 description 1
- YROXIXLRRCOBKF-UHFFFAOYSA-N sulfonylurea Chemical class OC(=N)N=S(=O)=O YROXIXLRRCOBKF-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N30/00—Investigating or analysing materials by separation into components using adsorption, absorption or similar phenomena or using ion-exchange, e.g. chromatography or field flow fractionation
- G01N30/02—Column chromatography
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N30/00—Investigating or analysing materials by separation into components using adsorption, absorption or similar phenomena or using ion-exchange, e.g. chromatography or field flow fractionation
- G01N30/02—Column chromatography
- G01N30/04—Preparation or injection of sample to be analysed
- G01N30/06—Preparation
Landscapes
- Physics & Mathematics (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Analytical Chemistry (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Immunology (AREA)
- Pathology (AREA)
- Other Investigation Or Analysis Of Materials By Electrical Means (AREA)
Abstract
The invention provides a method for measuring residual amounts of pyrazosulfuron-ethyl, triazophos and butachlor in water, belonging to the technical field of water quality detection methods. The method comprises the following steps: preparing a sample, preparing a standard working solution, testing the standard working solution in the second step by adopting ultra performance liquid chromatography-tandem mass spectrometry, drawing a standard curve by taking the peak areas of various targets as vertical coordinates and corresponding concentrations as horizontal coordinates, and quantifying by an external standard method; and (3) performing ultra performance liquid chromatography-tandem mass spectrometry on the sample to be detected in the first step under the same conditions, measuring the chromatographic peak areas of the pyrazosulfuron-ethyl, the triazophos and the butachlor in the sample to be detected, and substituting the chromatographic peak areas into a standard curve to obtain the content of the pyrazosulfuron-ethyl, the triazophos and the butachlor in the sample to be detected. The detection limit of the method is 0.1 mug/L, the quantification limit is 0.3 mug/L, the detection cost is low, and the method is accurate.
Description
Technical Field
The invention belongs to the technical field of water quality detection methods, and particularly relates to a method for measuring residual amounts of pyrazosulfuron-ethyl, triazophos and butachlor in water.
Background
Pyrazosulfuron-ethyl (CAS number: 98389-04-9) belongs to sulfonylurea herbicides, and is suitable for crops: the rice seedling field, direct seeding field and transplanting field are selective internal absorption conduction type herbicides, which are absorbed mainly by root systems and quickly transferred in the body of weed plants to inhibit growth, and weeds gradually die. The rice can decompose the pesticide, and has little influence on the growth of rice, and the effective period is 25-35 days. The stability is that the product can be stabilized for half a year at 50 ℃ and is stable to light. The structural formula of pyrazosulfuron-ethyl is as follows:
triazophos (CAS number: 24017-47-8) is a broad-spectrum organic phosphorus pesticide, acaricide, nematicide, and is mainly used for preventing and treating lepidoptera pests, mites, fly larvae, underground pests and the like on fruit trees, cotton and grain crops. The pure product is light yellow liquid, can be dissolved in most organic solvents, is stable to light, can be hydrolyzed in acid and alkali media, is harmful to the environment at 140 ℃, and is particularly concerned about the pollution to water bodies. The structural formula of the triazophos is as follows:
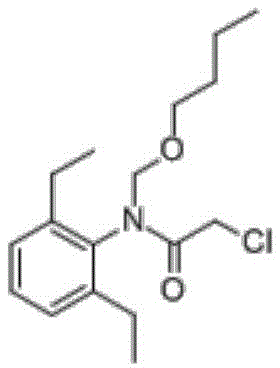
butachlor (CAS number: 23184-66-9), Chinese alias, bentazon, norbenazon, selective preemergent herbicides. Is insoluble in water and soluble in various organic solvents. Stable chemical property under normal temperature and neutral and alkalescent conditions. The decomposition of the organic acid is accelerated under the strong acid condition, and the organic acid can be degraded in soil. Is mainly used for preventing and killing annual gramineous weeds and certain broadleaf weeds in direct seeding or transplanting paddy fields. It is also selective for wheat, barley, beet, cotton, peanut and cabbage crops. The effective dose is 1.0-4.5kg (effective component) per hectare. The herbicide is generally used for treating the surface of soil before germination, can be applied after seedling emergence in paddy fields and is an important variety of herbicides in paddy fields. Butachlor has the following structural formula:
in the prior art, a method for detecting the residual quantity of pyrazosulfuron-ethyl, triazophos and butachlor in water is not available.
Disclosure of Invention
The invention aims to provide a method for measuring the residual amounts of pyrazosulfuron-ethyl, triazophos and butachlor in water, which is simple and convenient, high in sensitivity and good in repeatability.
The invention provides a method for measuring the residual quantity of pyrazosulfuron-ethyl, triazophos and butachlor in water, which comprises the following steps:
the method comprises the following steps: sample preparation
Weighing a water sample, adding acetic acid acetonitrile for extraction, adding sodium chloride for salting out, transferring an upper layer organic phase, repeatedly extracting for two times, combining two extraction solutions, drying by using nitrogen, adding a methanol water solution for constant volume, and filtering by using a membrane to obtain a sample to be detected; the volume ratio of the water sample to the acetic acid acetonitrile is 2: 1; volume mL of water sample: the mass g of sodium chloride is 50: 15; the volume ratio of the water sample to the methanol aqueous solution is 50: 1;
step two: preparation of standard working solution
Standard solution: firstly, preparing pyrazosulfuron-ethyl, triazophos and butachlor standard solutions with the concentration of 100mg/L respectively;
standard stock solutions: 1mL of the pyrazosulfuron-ethyl, triazophos and butachlor standard solutions are respectively put into a volumetric flask, dissolved by methanol and fixed to the volume to be calibrated, and prepared into standard stock solution with the concentration of 10 mg/L;
mixing standard intermediate liquid: 1mL of each standard stock solution is taken to be placed in a volumetric flask, methanol is dissolved and the volume is determined to scale, and a mixed standard intermediate solution with the concentration of 1000ng/mL is prepared;
drawing a standard curve: sucking a certain volume of liquid from the mixed standard intermediate liquid to prepare 5, 10, 20, 50 and 100ng/mL series of standard working liquids;
step three: measurement of
Testing the standard working solution in the second step by adopting ultra performance liquid chromatography-tandem mass spectrometry, drawing a standard curve by taking the peak areas of various target objects as vertical coordinates and the corresponding concentrations as horizontal coordinates, and quantifying by adopting an external standard method;
carrying out ultra performance liquid chromatography-tandem mass spectrometry on the sample to be detected in the first step under the same conditions, measuring the chromatographic peak areas of pyrazosulfuron-ethyl, triazophos and butachlor in the sample to be detected, and bringing the chromatographic peak areas into a standard curve to obtain the content of pyrazosulfuron-ethyl, triazophos and butachlor in the sample to be detected;
the determination conditions are as follows:
ultra-high performance liquid chromatography conditions
A chromatographic column: waters BEH C182.1mm x 50mm,1.7 μm, or equivalent performance columns;
flow rate: 0.25 mL/min;
sample introduction amount: 10 mu L of the solution;
column temperature: 40 ℃;
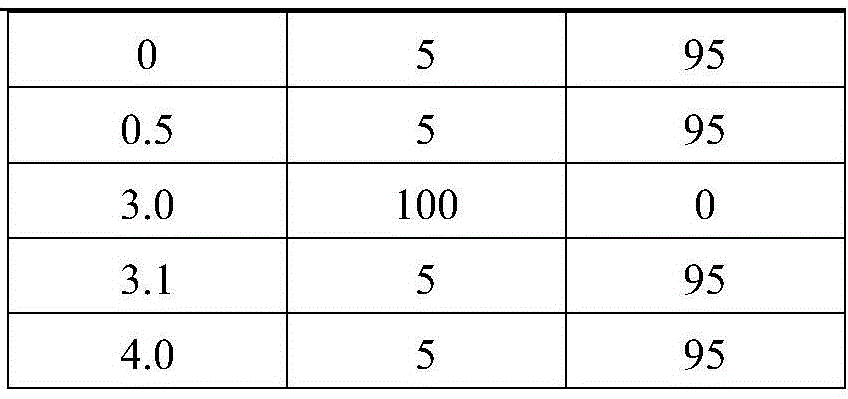
mobile phase: acetonitrile and 0.1% formic acid, gradient elution, as in Table 1
TABLE 1 gradient elution procedure
| Time | Acetonitrile | 0.1% formic acid water |
| 0 | 5 | 95 |
| 0.5 | 5 | 95 |
| 3.0 | 100 | 0 |
| 3.1 | 5 | 95 |
| 4.0 | 5 | 95 |
Conditions of Mass Spectrometry
An ion source: ESI;
the scanning mode is as follows: scanning positive ions;
the detection mode comprises the following steps: monitoring multiple reactions;
atomizer pressure: 50 psi;
flow rate of drying gas: 12L/min;
temperature of the drying gas: 350 ℃;
capillary voltage: 4000V;
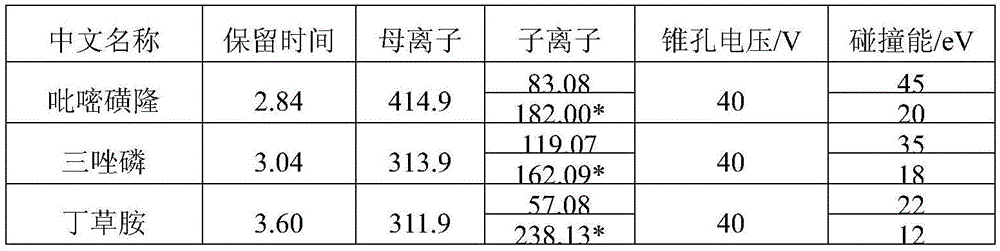
monitoring ion pairs, cone hole voltage and collision energy, see table 2;
TABLE 2 monitoring ion Pair, Cone hole Voltage and Collision energy
Note: is a quantitative ion
Preferably, the water sample is surface water.
Preferably, the volume fraction of acetonitrile acetate in the first step is 1%.
Preferably, the volume fraction of methanol in the first step is 20%.
Preferably, the temperature for blowing the nitrogen gas is 40 ℃.
Preferably, the filter is a 0.22 μm filter.
The invention has the advantages of
The invention provides a method for measuring the residual quantity of pyrazosulfuron-ethyl, triazophos and butachlor in water, which has high sensitivity, the detection limit of the method for measuring pyrazosulfuron-ethyl, triazophos and butachlor in surface water is 0.1 mu g/L, the quantification limit is 0.3 mu g/L, the detection cost is low, and the method is accurate.
Drawings
FIG. 1 is a total ion current chromatogram of pyrazosulfuron-ethyl, triazophos and butachlor standard substances in example 1 of the invention;
FIG. 2 is an ion mass spectrum of a pyrazosulfuron-ethyl standard substance in example 1 of the present invention under MRM mode;
FIG. 3 is an ion mass spectrum of triazophos standard substance in example 1 of the present invention in MRM mode;
FIG. 4 is an ion mass spectrum of the butachlor standard substance in the example 1 of the invention in MRM mode.
Detailed Description
The present invention will be described in further detail with reference to specific examples, it should be understood that the present invention is not limited to the examples, but is capable of modification and variation in various forms without departing from the scope of the present invention.
The reagents used in the examples, unless otherwise indicated, were analytically pure and the water should conform to the secondary water specification of GB/T6682.
Acetonitrile: carrying out chromatographic purification; methanol: carrying out chromatographic purification; sodium chloride: analyzing and purifying; acetic acid: the top grade is pure;
example 1
A method for measuring residual amounts of pyrazosulfuron-ethyl, triazophos and butachlor in water comprises the following steps:
the method comprises the following steps: sample preparation
A ground glass bottle is used for collecting a water sample, the collected sample is stored in a refrigerator at 4 ℃, and the transportation process of the sample needs to be shockproof, stored at low temperature and protected from sunlight irradiation, and the inside of a transport vehicle is prevented from being polluted. All samples were collected in duplicate, with one full procedure blank and one transport blank for each batch.
Measuring 10mL of acetic acid by using a measuring cylinder, pouring the acetic acid into a 1000mL volumetric flask, adding acetonitrile to a constant volume, uniformly mixing, and pouring into a 1% acetonitrile acetate special bottle;
transferring 50.00mL of a uniform sample by using a big belly pipette, adding the uniform sample into a 250mL separating funnel, accurately adding 25.00mL of 1% (volume fraction) acetic acid acetonitrile, carrying out vortex mixing, adding 15g of sodium chloride, carrying out vortex mixing, salting out, accurately transferring an upper layer organic phase, repeatedly extracting for two times, combining two extraction solutions, drying by blowing nitrogen at 40 ℃, adding 1.00mL of 20% (volume fraction) methanol aqueous solution for constant volume, and passing through a 0.22 mu m filter membrane to obtain a sample to be detected;
step two: preparation of standard working solution
Standard solution: firstly, preparing pyrazosulfuron-ethyl, triazophos and butachlor standard solutions with the concentration of 100mg/L respectively;
standard stock solutions: 1mL of the pyrazosulfuron-ethyl, triazophos and butachlor standard solutions are respectively put into a 10mL volumetric flask, dissolved by methanol, and fixed to a certain volume to be scaled to prepare a standard stock solution with the concentration of 10mg/L, and the standard stock solution is stored in a refrigerator at 4 ℃ in a dark place;
mixing standard intermediate liquid: taking 1mL of each standard stock solution, putting the standard stock solutions into a 10mL volumetric flask, dissolving the methanol, fixing the volume to a scale, preparing a mixed standard intermediate solution with the concentration of 1000ng/mL, and storing the mixed standard intermediate solution in a refrigerator at 4 ℃ in a dark place;
drawing a standard curve: sucking a certain volume of liquid from the mixed standard intermediate liquid to prepare 5, 10, 20, 50 and 100ng/mL series of standard working solutions;
step three: measurement of
Testing the standard working solution obtained in the second step by adopting ultra-high performance liquid chromatography-tandem mass spectrometry to obtain a total ion current chromatogram of pyrazosulfuron-ethyl, triazophos and butachlor standard substances in the standard working solution and an ion mass chromatogram of the pyrazosulfuron-ethyl, triazophos and butachlor standard substances in an MRM (multi-resolution mass spectrometry) mode, as shown in figures 1-4, drawing a standard curve by taking the peak areas of various targets as vertical coordinates and the corresponding concentrations as horizontal coordinates, and quantifying by adopting an external standard method, wherein the standard curve equation is shown in table 3;
TABLE 3
| Name (R) | Regression equation | Correlation coefficient |
| Pyrazosulfuron-ethyl | Y=5623.45x-3725.46 | 0.9996 |
| Triazophos | Y=928.46x-6901.74 | 0.9997 |
| Butachlor | Y=579.63x-783.22 | 0.9997 |
The detection limit of the method is 0.1 mug/L, and the quantification limit is 0.3 mug/L;
carrying out ultra performance liquid chromatography-tandem mass spectrometry on the sample to be detected in the first step under the same conditions, measuring the chromatographic peak areas of pyrazosulfuron-ethyl, triazophos and butachlor in the sample to be detected, and substituting the chromatographic peak areas into a linear equation to obtain the content of pyrazosulfuron-ethyl, triazophos and butachlor in the sample to be detected;
quantifying by an external standard method, and calculating the content of the target object in the sample according to the following formula:
in the formula:
x is the content of the substance to be detected in the sample, and the unit is microgram per liter (mu g/L);
c-the concentration of analyte in the sample solution obtained from the standard working curve in nanograms per milliliter (ng/mL);
v-volume volumetric volume in milliliters (mL);
V0-sample volume in milliliters (mL);
f is dilution multiple; (f 1)
The determination conditions are as follows:
ultra-high performance liquid chromatography conditions
A chromatographic column: waters BEH C182.1mm x 50mm,1.7 μm, or equivalent performance columns;
flow rate: 0.25 mL/min;
sample introduction amount: 10 mu L of the solution;
column temperature: 40 ℃;
mobile phase: acetonitrile and 0.1% formic acid water, gradient elution, as in table 2;
TABLE 2 gradient elution procedure
Conditions of Mass Spectrometry
An ion source: ESI;
the scanning mode is as follows: scanning positive ions;
the detection mode is as follows: monitoring multiple reactions;
atomizer pressure: 50 psi;
flow rate of drying gas: 12L/min;
temperature of the drying gas: 350 ℃;
capillary voltage: 4000V;
ion pairs, cone hole voltage and collision energy were monitored, see table 3;
TABLE 3 monitoring ion Pair, Cone hole Voltage and Collision energy
Note: is a quantitative ion
The surface waters from 5 different locations, labeled surface waters No. 1, No. 2, No. 3, No. 4 and No. 5, were measured as described in example 1 above, 5 consecutive measurements were made for each sample, and the average of the 5 measurements (in μ g/L) was taken, with the results shown in table 4:
TABLE 4
| Number 1 | Number 2 | Number 2 | Number 4 | Number 5 | |
| Pyrazosulfuron-ethyl | 20.59 | 20.6 | 20.63 | 20.61 | 20.62 |
| Triazophos | 18.45 | 18.47 | 18.44 | 18.46 | 18.45 |
| Butachlor | 19.20 | 19.22 | 19.23 | 19.21 | 19.23 |
While certain exemplary embodiments of the present invention have been described above by way of illustration only, it will be apparent to those of ordinary skill in the art that the described embodiments may be modified in various different ways without departing from the spirit and scope of the invention. Accordingly, the drawings and description are illustrative in nature and should not be construed as limiting the scope of the invention.
Claims (6)
1. A method for measuring residual amounts of pyrazosulfuron-ethyl, triazophos and butachlor in water is characterized by comprising the following steps:
the method comprises the following steps: sample preparation
Weighing a water sample, adding acetic acid acetonitrile for extraction, adding sodium chloride for salting out, transferring an upper layer organic phase, repeatedly extracting for two times, combining two extraction solutions, drying by using nitrogen, adding a methanol water solution for constant volume, and filtering by using a membrane to obtain a sample to be detected; the volume ratio of the water sample to the acetic acid acetonitrile is 2: 1; volume mL of water sample: the mass g of sodium chloride is 50: 15; the volume ratio of the water sample to the methanol aqueous solution is 50: 1;
step two: preparation of standard working solution
Standard solution: firstly, preparing pyrazosulfuron-ethyl, triazophos and butachlor standard solutions with the concentration of 100mg/L respectively;
standard stock solutions: 1mL of the pyrazosulfuron-ethyl, triazophos and butachlor standard solutions are respectively put into a volumetric flask, dissolved by methanol and fixed to the volume to be calibrated, and prepared into standard stock solution with the concentration of 10 mg/L;
mixing standard intermediate liquid: taking 1mL of each standard stock solution, dissolving in methanol, fixing the volume to a scale, and preparing a mixed standard intermediate solution with the concentration of 1000 ng/mL;
drawing a standard curve: sucking a certain volume of liquid from the mixed standard intermediate liquid to prepare 5, 10, 20, 50 and 100ng/mL series of standard working solutions;
step three: measurement of
Testing the standard working solution in the second step by adopting ultra performance liquid chromatography-tandem mass spectrometry, drawing a standard curve by taking the peak areas of various target objects as vertical coordinates and the corresponding concentrations as horizontal coordinates, and quantifying by adopting an external standard method;
carrying out ultra performance liquid chromatography-tandem mass spectrometry on the sample to be detected in the first step under the same conditions, measuring the chromatographic peak areas of pyrazosulfuron-ethyl, triazophos and butachlor in the sample to be detected, and bringing the chromatographic peak areas into a standard curve to obtain the content of pyrazosulfuron-ethyl, triazophos and butachlor in the sample to be detected;
the determination conditions are as follows:
ultra-high performance liquid chromatography conditions
A chromatographic column: waters BEH C182.1mm x 50mm,1.7 μm, or equivalent performance columns;
flow rate: 0.25 mL/min;
sample introduction amount: 10 mu L of the solution;
column temperature: at 40 ℃;
mobile phase: acetonitrile and 0.1% formic acid, gradient elution, as in Table 1
TABLE 1
Mass spectrum conditions:
an ion source: ESI;
the scanning mode is as follows: scanning positive ions;
the detection mode is as follows: monitoring multiple reactions;
atomizer pressure: 50 psi;
flow rate of drying gas: 12L/min;
temperature of the drying gas: 350 ℃;
capillary voltage: 4000V;
monitoring ion pairs, cone hole voltage and collision energy, see table 2;
TABLE 2
2. The method for determining the residual amounts of pyrazosulfuron-ethyl, triazophos and butachlor in water quality as claimed in claim 1, wherein the water sample is surface water.
3. The method for determining the residual quantity of pyrazosulfuron-ethyl, triazophos and butachlor in water according to claim 1, wherein the volume fraction of acetonitrile acetate in the first step is 1%.
4. The method for determining the residual quantity of pyrazosulfuron-ethyl, triazophos and butachlor in water as claimed in claim 1, wherein the volume fraction of methanol in the first step is 20%.
5. The method for measuring the residual amounts of pyrazosulfuron-ethyl, triazophos and butachlor in water quality as claimed in claim 1, wherein the temperature for drying nitrogen is 40 ℃.
6. The method for measuring the residual quantity of pyrazosulfuron-ethyl, triazophos and butachlor in water quality as claimed in claim 1, wherein the filter membrane is a 0.22 μm filter membrane.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910794199.2A CN110470768B (en) | 2019-08-27 | 2019-08-27 | Method for measuring residual amounts of pyrazosulfuron-ethyl, triazophos and butachlor in water |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910794199.2A CN110470768B (en) | 2019-08-27 | 2019-08-27 | Method for measuring residual amounts of pyrazosulfuron-ethyl, triazophos and butachlor in water |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN110470768A CN110470768A (en) | 2019-11-19 |
| CN110470768B true CN110470768B (en) | 2022-05-24 |
Family
ID=68512284
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201910794199.2A Active CN110470768B (en) | 2019-08-27 | 2019-08-27 | Method for measuring residual amounts of pyrazosulfuron-ethyl, triazophos and butachlor in water |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN110470768B (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN113960196A (en) * | 2021-10-11 | 2022-01-21 | 太湖流域水文水资源监测中心(太湖流域水环境监测中心) | Method for on-line determination of sulfonylurea herbicide in water |
| CN115406987B (en) * | 2022-08-30 | 2023-08-08 | 湖南农业大学 | Establishment method of GC-MS detection and analysis conditions for clopyrazosulfuron-methyl |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DK2155699T5 (en) * | 2007-05-15 | 2017-05-22 | Merial Inc | ARYLAZOL-2-YL-CYANOETHYLAMINO COMPOUNDS, METHOD OF PREPARATION AND PROCEDURE FOR USING THEREOF |
| CN102221589A (en) * | 2011-03-28 | 2011-10-19 | 中国水稻研究所 | Method for detecting residual quantity of pesticides in vegetable sample by utilizing liquid chromatogram-high resolution mass spectrometer |
| KR102085600B1 (en) * | 2012-06-30 | 2020-03-06 | 다우 아그로사이언시즈 엘엘씨 | Insecticidal n-substituted sulfilimine and sulfoximine pyridine n-oxides |
| RU2638043C2 (en) * | 2012-12-19 | 2017-12-11 | ДАУ АГРОСАЙЕНСИЗ ЭлЭлСи | Pesticide compositions and related methods |
| CN108896694B (en) * | 2018-07-05 | 2019-09-27 | 中国农业科学院农业质量标准与检测技术研究所 | A LC-QToF-MS screening analysis method for pesticide residues in animal food |
-
2019
- 2019-08-27 CN CN201910794199.2A patent/CN110470768B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN110470768A (en) | 2019-11-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Whitehead Jr et al. | Method for measurement of the quaternary amine compounds paraquat and diquat in human urine using high-performance liquid chromatography–tandem mass spectrometry | |
| Demoliner et al. | Development and validation of a method using SPE and LC-ESI-MS-MS for the determination of multiple classes of pesticides and metabolites in water samples | |
| CN107037149B (en) | A method for the determination of fipronil and its metabolite residues in eggs | |
| CN111855871B (en) | Method for measuring iprovalicarb and fluopyram in tobacco by combination of filtration type solid-phase extraction and supercritical fluid chromatography-mass spectrometry | |
| CN113376298A (en) | Method for rapidly determining residual quantity of 4 pesticides in rice | |
| CN102854271A (en) | Method for measuring residues of three phenoxy carboxylic acid pesticides in tobacco and tobacco products | |
| CN112461960B (en) | Method for simultaneously measuring various heterocyclic pesticides, degradation products and intermediates in water | |
| CN110470768B (en) | Method for measuring residual amounts of pyrazosulfuron-ethyl, triazophos and butachlor in water | |
| She et al. | Determination of glufosinate-ammonium residue in wheat and soil by ultra-performance liquid chromatography-tandem mass spectrometry | |
| CN108680663B (en) | Detection method of glyphosate and metabolite thereof in sugarcane | |
| Wang et al. | Determination of agrochemical residues in aquatic vegetables by solid-phase extraction combined with HPLC spectrometry analyses | |
| CN108020627B (en) | Method for determining residual quantity of three phenoxy carboxylic acid pesticides in tobacco by ultra-high performance synthetic phase chromatography-tandem mass spectrometry | |
| Zhang et al. | Extraction of acetanilide herbicides in naked oat (Avena nuda L.) by using ionic‐liquid‐based matrix solid‐phase dispersion‐foam flotation solid‐phase extraction | |
| CN112858542B (en) | Liquid chromatography mass spectrometry method for measuring atrazine, imidacloprid and metabolites thereof in leaves | |
| Amin et al. | Sensitive spectrophotometric determination of deltamethrin insecticide in its formulation and environmental samples | |
| CN108802215A (en) | The assay method of Rynaxypyr residual quantity in a kind of vegetables | |
| JP2008528956A (en) | Method for analyzing phosphorous acid, fosetyl-Al or both simultaneously | |
| CN106596442A (en) | Method for rapidly and quantitatively detecting phorate residue | |
| CN110568092A (en) | method for simultaneously detecting phosphine, ethephon, glufosinate-ammonium and glyphosate in water | |
| Suvardhan et al. | Extractive spectrofluorometric determination of quinalphos using fluorescein in environmental samples | |
| CN104198642A (en) | Method for detecting aminopyralid pesticide residues in barley | |
| CN112305113A (en) | Analysis method of metham sodium | |
| CN109142589B (en) | Method for determining tetramycin residual quantity in soil by using high performance liquid chromatograph | |
| CN108693274B (en) | Method for detecting triazole pesticide residues in white wine by combining solidification-floating dispersion liquid microextraction and HPLC | |
| Zhang et al. | Determination of methyl bromide residues in tea by headspace solid-phase microextraction coupled with gas chromatography-mass spectrometry |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |