CN110437177B - A kind of pleuromutilin derivative and its preparation method and application - Google Patents
A kind of pleuromutilin derivative and its preparation method and application Download PDFInfo
- Publication number
- CN110437177B CN110437177B CN201910601978.6A CN201910601978A CN110437177B CN 110437177 B CN110437177 B CN 110437177B CN 201910601978 A CN201910601978 A CN 201910601978A CN 110437177 B CN110437177 B CN 110437177B
- Authority
- CN
- China
- Prior art keywords
- pharmaceutically acceptable
- compound
- preparation
- acceptable salt
- cdcl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 150000001875 compounds Chemical class 0.000 claims abstract description 23
- 150000003839 salts Chemical group 0.000 claims abstract description 16
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 6
- 239000003814 drug Substances 0.000 claims description 11
- 241000191967 Staphylococcus aureus Species 0.000 claims description 7
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 6
- 229910052739 hydrogen Inorganic materials 0.000 claims description 5
- 208000035473 Communicable disease Diseases 0.000 claims description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 4
- 241000588724 Escherichia coli Species 0.000 claims description 3
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 3
- 239000001257 hydrogen Substances 0.000 claims description 3
- 208000015181 infectious disease Diseases 0.000 claims description 3
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 claims description 2
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 claims description 2
- 229910019142 PO4 Inorganic materials 0.000 claims description 2
- 239000003937 drug carrier Substances 0.000 claims description 2
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 claims description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 2
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 claims description 2
- 150000001558 benzoic acid derivatives Chemical class 0.000 claims 1
- 229940049920 malate Drugs 0.000 claims 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N malic acid Chemical compound OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 claims 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-M methanesulfonate group Chemical class CS(=O)(=O)[O-] AFVFQIVMOAPDHO-UHFFFAOYSA-M 0.000 claims 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims 1
- 235000021317 phosphate Nutrition 0.000 claims 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 claims 1
- 150000003890 succinate salts Chemical class 0.000 claims 1
- 238000002360 preparation method Methods 0.000 abstract description 14
- ZRZNJUXESFHSIO-VYTKZBNOSA-N pleuromutilin Chemical class C([C@H]([C@]1(C)[C@@H](C[C@@](C)(C=C)[C@@H](O)[C@@H]2C)OC(=O)CO)C)C[C@]32[C@H]1C(=O)CC3 ZRZNJUXESFHSIO-VYTKZBNOSA-N 0.000 abstract description 10
- 239000013078 crystal Chemical group 0.000 abstract description 3
- 229940124350 antibacterial drug Drugs 0.000 abstract description 2
- 239000000543 intermediate Substances 0.000 abstract 1
- 230000015572 biosynthetic process Effects 0.000 description 14
- 238000003786 synthesis reaction Methods 0.000 description 13
- 229940079593 drug Drugs 0.000 description 10
- 230000000844 anti-bacterial effect Effects 0.000 description 9
- 238000002844 melting Methods 0.000 description 9
- 230000008018 melting Effects 0.000 description 9
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 8
- 238000006243 chemical reaction Methods 0.000 description 8
- UURAUHCOJAIIRQ-QGLSALSOSA-N tiamulin Chemical compound CCN(CC)CCSCC(=O)O[C@@H]1C[C@@](C)(C=C)[C@@H](O)[C@H](C)[C@@]23CC[C@@H](C)[C@]1(C)[C@@H]2C(=O)CC3 UURAUHCOJAIIRQ-QGLSALSOSA-N 0.000 description 8
- 229960004885 tiamulin Drugs 0.000 description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 8
- 241000894006 Bacteria Species 0.000 description 7
- 239000000243 solution Substances 0.000 description 7
- 230000001580 bacterial effect Effects 0.000 description 6
- 239000013641 positive control Substances 0.000 description 6
- ZRZNJUXESFHSIO-UHFFFAOYSA-N Pleuromutilin Natural products CC1C(O)C(C)(C=C)CC(OC(=O)CO)C2(C)C(C)CCC31C2C(=O)CC3 ZRZNJUXESFHSIO-UHFFFAOYSA-N 0.000 description 5
- 239000003242 anti bacterial agent Substances 0.000 description 5
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 229940088710 antibiotic agent Drugs 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- ITHCSGCUQDMYAI-ZMIZWQJLSA-N 2-carboxy-D-arabinitol 1,5-bisphosphate Chemical compound OP(=O)(O)OC[C@@H](O)[C@@H](O)[C@](O)(COP(O)(O)=O)C(O)=O ITHCSGCUQDMYAI-ZMIZWQJLSA-N 0.000 description 2
- KPVIXBKIJXZQJX-FCEONZPQSA-N 21904a5386 Chemical compound O([C@H]1[C@@]2(C)[C@@H]3C(=O)CC[C@]3([C@H]([C@H](O)[C@](C)(C=C)C1)C)CC[C@H]2C)C(=O)CS[C@@H]1CC[C@@H](N)C[C@H]1O KPVIXBKIJXZQJX-FCEONZPQSA-N 0.000 description 2
- RJQXTJLFIWVMTO-TYNCELHUSA-N Methicillin Chemical compound COC1=CC=CC(OC)=C1C(=O)N[C@@H]1C(=O)N2[C@@H](C(O)=O)C(C)(C)S[C@@H]21 RJQXTJLFIWVMTO-TYNCELHUSA-N 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- 102100021486 Protein S100-G Human genes 0.000 description 2
- 101710122252 Protein S100-G Proteins 0.000 description 2
- 125000000217 alkyl group Chemical group 0.000 description 2
- FMHQJXGMLMSMLC-WBUYAQKGSA-N azamulin Chemical compound O([C@@H]1C[C@@]([C@H]([C@H](C)[C@]23CCC(=O)[C@H]2[C@@]1(C)[C@@H](CC3)C)O)(C)CC)C(=O)CSC1=NNC(N)=N1 FMHQJXGMLMSMLC-WBUYAQKGSA-N 0.000 description 2
- 229950008762 azamulin Drugs 0.000 description 2
- HUMNYLRZRPPJDN-UHFFFAOYSA-N benzaldehyde Chemical compound O=CC1=CC=CC=C1 HUMNYLRZRPPJDN-UHFFFAOYSA-N 0.000 description 2
- 238000004440 column chromatography Methods 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 238000000034 method Methods 0.000 description 2
- 229960003085 meticillin Drugs 0.000 description 2
- 239000010413 mother solution Substances 0.000 description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000008223 sterile water Substances 0.000 description 2
- LLYYNOVSVPBRGV-MVNKZKPCSA-N valnemulin Chemical compound CC(C)[C@@H](N)C(=O)NCC(C)(C)SCC(=O)O[C@@H]1C[C@@](C)(C=C)[C@@H](O)[C@H](C)[C@@]23CC[C@@H](C)[C@]1(C)[C@@H]2C(=O)CC3 LLYYNOVSVPBRGV-MVNKZKPCSA-N 0.000 description 2
- 229950008166 valnemulin Drugs 0.000 description 2
- ZDPAWHACYDRYIW-UHFFFAOYSA-N 1-(4-fluorophenyl)ethanone Chemical compound CC(=O)C1=CC=C(F)C=C1 ZDPAWHACYDRYIW-UHFFFAOYSA-N 0.000 description 1
- YKYWUHHZZRBGMG-JWTNVVGKSA-N 1-methyl-2-[[(1r,5s)-6-[[5-(trifluoromethyl)pyridin-2-yl]methoxymethyl]-3-azabicyclo[3.1.0]hexan-3-yl]methyl]benzimidazole Chemical compound C1([C@@H]2CN(C[C@@H]21)CC=1N(C2=CC=CC=C2N=1)C)COCC1=CC=C(C(F)(F)F)C=N1 YKYWUHHZZRBGMG-JWTNVVGKSA-N 0.000 description 1
- WCDLCPLAAKUJNY-UHFFFAOYSA-N 4-[4-[3-(1h-pyrazol-4-yl)pyrazolo[1,5-a]pyrimidin-6-yl]phenyl]morpholine Chemical compound C1COCCN1C1=CC=C(C2=CN3N=CC(=C3N=C2)C2=CNN=C2)C=C1 WCDLCPLAAKUJNY-UHFFFAOYSA-N 0.000 description 1
- MITGKKFYIJJQGL-UHFFFAOYSA-N 9-(4-chlorobenzoyl)-6-methylsulfonyl-2,3-dihydro-1H-carbazol-4-one Chemical compound ClC1=CC=C(C(=O)N2C3=CC=C(C=C3C=3C(CCCC2=3)=O)S(=O)(=O)C)C=C1 MITGKKFYIJJQGL-UHFFFAOYSA-N 0.000 description 1
- 201000001178 Bacterial Pneumonia Diseases 0.000 description 1
- 108010077805 Bacterial Proteins Proteins 0.000 description 1
- 208000035143 Bacterial infection Diseases 0.000 description 1
- 102000002004 Cytochrome P-450 Enzyme System Human genes 0.000 description 1
- 108010015742 Cytochrome P-450 Enzyme System Proteins 0.000 description 1
- 206010059866 Drug resistance Diseases 0.000 description 1
- 241000233866 Fungi Species 0.000 description 1
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- 241000204031 Mycoplasma Species 0.000 description 1
- 206010034133 Pathogen resistance Diseases 0.000 description 1
- 108090000279 Peptidyltransferases Proteins 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 102000000591 Tight Junction Proteins Human genes 0.000 description 1
- 108010002321 Tight Junction Proteins Proteins 0.000 description 1
- 108020004566 Transfer RNA Proteins 0.000 description 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 230000000845 anti-microbial effect Effects 0.000 description 1
- 208000022362 bacterial infectious disease Diseases 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 230000003115 biocidal effect Effects 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 238000009795 derivation Methods 0.000 description 1
- 238000003113 dilution method Methods 0.000 description 1
- -1 diterpenoid compound Chemical class 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 229950010255 lefamulin Drugs 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 150000004701 malic acid derivatives Chemical class 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 230000010534 mechanism of action Effects 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 239000013642 negative control Substances 0.000 description 1
- 239000002547 new drug Substances 0.000 description 1
- 239000012074 organic phase Substances 0.000 description 1
- QNGNSVIICDLXHT-UHFFFAOYSA-N para-ethylbenzaldehyde Natural products CCC1=CC=C(C=O)C=C1 QNGNSVIICDLXHT-UHFFFAOYSA-N 0.000 description 1
- YZTJYBJCZXZGCT-UHFFFAOYSA-N phenylpiperazine Chemical compound C1CNCCN1C1=CC=CC=C1 YZTJYBJCZXZGCT-UHFFFAOYSA-N 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 206010040872 skin infection Diseases 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 210000001578 tight junction Anatomy 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 239000000273 veterinary drug Substances 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/04—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms
- C07D295/14—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
- C07D295/145—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals with the ring nitrogen atoms and the carbon atoms with three bonds to hetero atoms attached to the same carbon chain, which is not interrupted by carbocyclic rings
- C07D295/15—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals with the ring nitrogen atoms and the carbon atoms with three bonds to hetero atoms attached to the same carbon chain, which is not interrupted by carbocyclic rings to an acyclic saturated chain
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B2200/00—Indexing scheme relating to specific properties of organic compounds
- C07B2200/07—Optical isomers
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Communicable Diseases (AREA)
- Pharmacology & Pharmacy (AREA)
- Oncology (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
技术领域technical field
本发明涉及一种通式(I)所述的截短侧耳素衍生物及其立体异构体、药学上可接受的盐或晶型,或包含它们的药物组合物,以及它们的制备方法和中间体,以及在用于制备抗菌药物中的用途。The present invention relates to a pleuromutilin derivative described in general formula (I) and its stereoisomer, pharmaceutically acceptable salt or crystal form, or a pharmaceutical composition containing them, as well as their preparation method and Intermediate, and its use in the preparation of antibacterial drugs.
背景技术Background technique
抗生素的发现在人类发展史上具有重大的意义,但现在由于临床上广泛长期滥用抗生素导致细菌耐药性日益严重,尤其是“超级细菌”的出现,这对人类生命安全构成了巨大威胁。为了应对有效抗菌药物逐年减少和耐药性细菌日益增加的局面,迫切需要找到一种有效途径来解决这一棘手的全球性问题.The discovery of antibiotics is of great significance in the history of human development, but now due to the widespread and long-term abuse of antibiotics in clinical practice, bacterial drug resistance is becoming more and more serious, especially the emergence of "super bacteria", which poses a huge threat to human life. In order to cope with the decreasing number of effective antimicrobial drugs and the increasing number of drug-resistant bacteria, it is urgent to find an effective way to solve this thorny global problem.
截短侧耳素 (Pleuromutilin,(式1)) 是从高等真菌中分离出来的一种具有良好抗菌活性的三环二萜类的化合物。研究表明,截短侧耳素结合在细菌核糖体50S亚基的23SrRNA上,通过其三环母核定位在核糖体50S亚基的肽基转移酶(PTC)中心,在A位点形成一个紧密的口袋,同时,其侧链部分覆盖了tRNA结合的P位点,由此直接抑制肽键的形成,从而阻止了细菌蛋白质的合成,正是这种特殊的作用机制, 使其不易于其它类抗生素产生交叉耐药性。近年来科研工作者在对其侧链的衍生研究中取得了较好的成果,如泰妙菌素(Tamulin,(式2)) 和沃尼妙林 (Valnemulin,(式3)) 分别在1979和1999年成功被批准为兽用抗生素。此外1980年阿扎莫林 (Azamulin,(式4)) 由于具有优秀抗菌效果而进入临床阶段,但是由于对人细胞色素P450的强烈抑制作用及其低口服生物利用度和短半衰期而宣告终止。2007年葛兰素史克 (GlaxoSmithKline) 公司开发了瑞他妙林 (Ratapamulin,(式5)),这是第一个用于治疗人类皮肤感染的外用截短侧耳素类抗菌剂。Lafemulin (BC-3781,(式6)) 是Nabriva公司开发的一种新型半合成截短侧耳素抗菌剂,用于急性细菌性皮肤和皮肤结构感染 (ABSSSI) 和社区获得性细菌性肺炎 (CABP) 的治疗。该药物在临床阶段表现出良好的安全性和耐受性, FDA于2019年2月接受了Nabriva公司提交的用于治疗CABP的lefamulin口服和静脉注射制剂两种新药申请 (NDAs)。Pleuromutilin (Formula 1) is a tricyclic diterpenoid compound isolated from higher fungi with good antibacterial activity. Studies have shown that pleuromutilin binds to the 23S rRNA of the 50S subunit of the bacterial ribosome, locates at the peptidyl transferase (PTC) center of the 50S subunit of the ribosome through its three-ring mother nucleus, and forms a tight junction at the A site. At the same time, its side chain partially covers the P site of tRNA binding, thereby directly inhibiting the formation of peptide bonds, thus preventing the synthesis of bacterial proteins. It is this special mechanism of action that makes it difficult for other antibiotics produce cross-resistance. In recent years, scientific researchers have achieved good results in the derivation of their side chains, such as tiamulin (Tamulin, (formula 2)) and valnemulin (valnemulin, (formula 3)) respectively in 1979 And in 1999, it was successfully approved as a veterinary antibiotic. In addition, in 1980, Azamulin (Azamulin, (Formula 4)) entered the clinical stage due to its excellent antibacterial effect, but it was terminated due to its strong inhibitory effect on human cytochrome P450 and its low oral bioavailability and short half-life. In 2007, GlaxoSmithKline (GlaxoSmithKline) developed Ratapamulin (Ratapamulin (Formula 5)), which is the first topical pleuromutilin antibacterial agent for the treatment of human skin infections. Lafemulin (BC-3781, (Formula 6)) is a novel semi-synthetic pleuromutilin antibacterial agent developed by Nabriva for acute bacterial skin and skin structure infection (ABSSSI) and community-acquired bacterial pneumonia (CABP ) Treatment. The drug has shown good safety and tolerability in the clinical stage. In February 2019, the FDA accepted two New Drug Applications (NDAs) for lefamulin oral and intravenous formulations submitted by Nabriva for the treatment of CABP.
截短侧耳素在兽用药物发展上比较迅速,但在人类进程中的作用还未能完全体现。 基于未满足的临床需求,开发具有新颖结构、独特作用机制的较高抗菌活性的药物迫在眉睫。The development of pleuromutilin in veterinary drugs is relatively rapid, but its role in the human process has not been fully reflected. Based on unmet clinical needs, it is imminent to develop drugs with novel structures and unique mechanisms of action with high antibacterial activity.
发明内容Contents of the invention
本发明涉及一种结构新颖、并具有良好抗菌活性的截短侧耳素衍生物。The invention relates to a pleuromutilin derivative with novel structure and good antibacterial activity.
本发明涉及一种通式(I)所述的化合物及其立体异构体、药学上可接受的盐,其中The present invention relates to a compound described in general formula (I) and its stereoisomers and pharmaceutically acceptable salts, wherein
A选自苯基,所述的苯基上的氢任选进一步被1-4个选自H、C1-4烷氧基、NH2、NO2或NRARB的取代基所取代;A is selected from phenyl, and the hydrogen on the phenyl is optionally further substituted by 1-4 substituents selected from H, C 1-4 alkoxy, NH 2 , NO 2 or NR A R B ;
RA、RB各自独立的选自H、C1-4烷基或-CO-C1-4烷基。R A and R B are each independently selected from H, C 1-4 alkyl or -CO-C 1-4 alkyl.
本发明一些实施例涉及一种通式(I)所述的化合物及其立体异构体、药学上可接受的盐,其中Some embodiments of the present invention relate to a compound described in general formula (I) and its stereoisomers and pharmaceutically acceptable salts, wherein
A选自苯基,所述的苯基上的氢任选进一步被1-4个H、OCH3、NH2、NO2或NRARB取代;A is selected from phenyl, and the hydrogen on the phenyl is optionally further substituted by 1-4 H, OCH 3 , NH 2 , NO 2 or NR A R B ;
RA、RB各自独立的选自H、甲基、乙基、甲酰基或乙酰基。R A and R B are each independently selected from H, methyl, ethyl, formyl or acetyl.
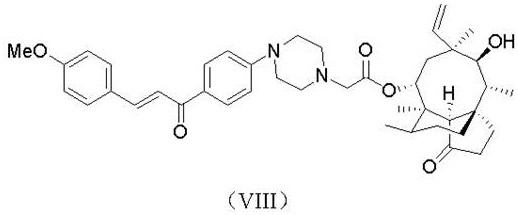
本发明的一些实施例涉及一种通式(I)所述的化合物及其立体异构体、药学上可接受的盐,其中该化合物选自如下结构(式II-式X)之一:Some embodiments of the present invention relate to a compound described in general formula (I) and its stereoisomers and pharmaceutically acceptable salts, wherein the compound is selected from one of the following structures (Formula II-Formula X):
本发明的一些实施例涉及一种通式(I)所述的化合物及其立体异构体、药学上可接受的盐,其中所述的盐选自盐酸盐、富马酸盐、苹果酸盐、氢溴酸盐、琥珀酸盐、磷酸盐、甲磺酸盐或苯甲酸盐。Some embodiments of the present invention relate to a compound described in general formula (I) and its stereoisomers and pharmaceutically acceptable salts, wherein the salts are selected from hydrochloride, fumarate, malic acid salt, hydrobromide, succinate, phosphate, methanesulfonate or benzoate.
本发明的一些实施例涉及一种药物组合物,所述药物组合物含有治疗有效剂量的本发明所述的化合物及其立体异构体或药学上可以接受的盐,以及药学上可接受的载体或者赋形剂。Some embodiments of the present invention relate to a pharmaceutical composition, which contains a therapeutically effective dose of the compound of the present invention and its stereoisomer or pharmaceutically acceptable salt, and a pharmaceutically acceptable carrier or excipients.
本发明的一些实施例涉及本发明所述的化合物及其立体异构体、药学上可接受的盐,或本发明所述的药物组合物,在用于制备感染性疾病药物中的应用。Some embodiments of the present invention relate to the application of the compounds described in the present invention and their stereoisomers, pharmaceutically acceptable salts, or the pharmaceutical compositions described in the present invention in the preparation of drugs for infectious diseases.
本发明的一些实施例涉及的上述的应用,所述的感染性疾病选自由支原体或耐药菌引起的感染性疾病。Some embodiments of the present invention relate to the above application, and the infectious disease is selected from infectious diseases caused by mycoplasma or drug-resistant bacteria.
具体实施例specific embodiment
实施例1:14a的合成Example 1: Synthesis of 14a
第一步:12a的合成The first step: the synthesis of 12a
将4-氟苯乙酮 (500.0 mg, 3.6 mmol) 和氢氧化钠 (144.8 mg, 3.6 mmol) 溶于乙醇中置于冰浴条件下搅拌0.5 h,再将苯甲醛 (345.7 mg, 3.3 mmol) 加入上述混合溶液中继续置于冰浴条件下,TLC监测至反应完全,用1M的盐酸调节反应液pH至酸性,加入适量的水使固体完全析出,抽滤,用水洗涤固体,烘干,用乙醇重结晶,即得到12a (496.3mg,产率67.3%)。4-Fluoroacetophenone (500.0 mg, 3.6 mmol) and sodium hydroxide (144.8 mg, 3.6 mmol) were dissolved in ethanol and stirred in an ice bath for 0.5 h, then benzaldehyde (345.7 mg, 3.3 mmol) Add the above mixed solution and continue to place it in an ice bath, monitor by TLC until the reaction is complete, adjust the pH of the reaction solution to acidity with 1M hydrochloric acid, add an appropriate amount of water to completely precipitate the solid, filter with suction, wash the solid with water, dry, and use Recrystallized from ethanol to obtain 12a (496.3 mg, yield 67.3%).
第二步:13a的合成Step 2: Synthesis of 13a
将化合物12a (300.0 mg, 1.3 mmol),碳酸钾 (366.5 mg, 2.7 mmol) 和哌嗪(228.4 mg, 2.7 mmol) 溶于DMF中置于110oC加热搅拌至反应完全,待TLC检测至反应完全,将其反应液冷却至室温向其加入适量的水,用DCM萃取反应液,再用水反萃取所得有机相,无水硫酸镁干燥,减压蒸馏,经柱层析分离提纯可得13a (252.3 mg, 产率65.1%)。Compound 12a (300.0 mg, 1.3 mmol), potassium carbonate (366.5 mg, 2.7 mmol) and piperazine (228.4 mg, 2.7 mmol) were dissolved in DMF, heated and stirred at 110 o C until the reaction was complete, and the reaction was detected by TLC. Completely, the reaction solution was cooled to room temperature and an appropriate amount of water was added thereto, the reaction solution was extracted with DCM, and the obtained organic phase was back-extracted with water, dried over anhydrous magnesium sulfate, distilled under reduced pressure, separated and purified by column chromatography to obtain 13a ( 252.3 mg, yield 65.1%).
第三步:14a的合成Step 3: Synthesis of 14a
将化合物9 (200.0 mg, 0.4 mmol) 和NaI (67.5 mg, 0.5 mmol) 溶于乙腈中置于75℃搅拌0.5 h,再将化合物13a (131.7 mg, 0.5 mmol) 和碳酸钾 (103.8 mg, 0.8mmol) 加入上述反应液中继续搅拌,TLC检测反应结束后减压蒸馏,粗产品经柱层析分离提纯可得14a (145.6 mg, 产率59.4%, 熔点106.7-108.1 oC)。Compound 9 (200.0 mg, 0.4 mmol) and NaI (67.5 mg, 0.5 mmol) were dissolved in acetonitrile and stirred at 75°C for 0.5 h, then compound 13a (131.7 mg, 0.5 mmol) and potassium carbonate (103.8 mg, 0.8 mmol) was added to the above reaction solution and continued to stir. After the reaction was detected by TLC, it was distilled under reduced pressure. The crude product was separated and purified by column chromatography to obtain 14a (145.6 mg, yield 59.4%, melting point 106.7-108.1 oC ).
1H NMR(400 MHz, CDCl3): δ (ppm) 8.01 (d, J = 8.8 Hz, 2H), 7.80 (d, J =15.6 Hz, 1H), 7.65 (m, 2H), 7.57 (d, J = 15.6 Hz, 1H), 7.46 – 7.37 (m, 3H),6.92 (d, J = 8.8 Hz, 2H), 6.60 – 6.48 (m, 1H), 5.83 (d, J = 8.4, 1H), 5.37(dd, J = 11.2, 1.4 Hz, 1H), 5.22 (dd, J = 17.2, 1.6 Hz, 1H), 3.48 – 3.34 (m,5H), 3.26 (d, J = 17.2 Hz, 1H), 3.13 (d, J = 17.2 Hz, 1H), 2.80 – 2.63 (m,4H), 2.41 – 2.06 (m, 5H), 1.80 (dd, J = 14.4, 2.4 Hz,1H), 1.74 – 1.49 (m,5H), 1.47 (s, 3H), 1.43 – 1.36 (m, 1H), 1.35 – 1.29 (m, 1H), 1.19 (s, 3H),1.17 – 1.10 (m, 1H), 0.90 (d, J = 6.8 Hz, 3H), 0.76 (d, J = 6.8 Hz, 3H). 1 H NMR (400 MHz, CDCl 3 ): δ (ppm) 8.01 (d, J = 8.8 Hz, 2H), 7.80 (d, J =15.6 Hz, 1H), 7.65 (m, 2H), 7.57 (d, J = 15.6 Hz, 1H), 7.46 – 7.37 (m, 3H), 6.92 (d, J = 8.8 Hz, 2H), 6.60 – 6.48 (m, 1H), 5.83 (d, J = 8.4, 1H), 5.37 (dd, J = 11.2, 1.4 Hz, 1H), 5.22 (dd, J = 17.2, 1.6 Hz, 1H), 3.48 – 3.34 (m,5H), 3.26 (d, J = 17.2 Hz, 1H), 3.13 ( d, J = 17.2 Hz, 1H), 2.80 – 2.63 (m,4H), 2.41 – 2.06 (m, 5H), 1.80 (dd, J = 14.4, 2.4 Hz,1H), 1.74 – 1.49 (m,5H) , 1.47 (s, 3H), 1.43 – 1.36 (m, 1H), 1.35 – 1.29 (m, 1H), 1.19 (s, 3H),1.17 – 1.10 (m, 1H), 0.90 (d, J = 6.8 Hz , 3H), 0.76 (d, J = 6.8 Hz, 3H).
13C NMR (100 MHz, CDCl3): δ (ppm) 217.1, 188.1, 169.0, 154.0, 143.2,139.1, 135.3, 130.7, 130.1, 129.3, 128.9, 128.3, 122.0, 117.3, 113.6, 74.6,68.5, 59.9, 58.2, 53.5, 52.6, 47.2, 45.5, 44.0, 41.8, 36.7, 36.1, 34.5, 30.5,26.9, 26.4, 24.9, 16.8, 14.9, 11.5. 13 C NMR (100 MHz, CDCl 3 ): δ (ppm) 217.1, 188.1, 169.0, 154.0, 143.2, 139.1, 135.3, 130.7, 130.1, 129.3, 128.9, 128.3, 125.6, 114.36, 8.7 , 58.2, 53.5, 52.6, 47.2, 45.5, 44.0, 41.8, 36.7, 36.1, 34.5, 30.5, 26.9, 26.4, 24.9, 16.8, 14.9, 11.5.
实施例2:14b的合成Embodiment 2: Synthesis of 14b
制备方法参考实施例1Preparation method reference example 1
产率62.7% 熔点118.2-119.9oCYield 62.7% Melting point 118.2-119.9 o C
1H NMR (400 MHz, CDCl3): δ (ppm) 7.97 (d, J = 8.8 Hz, 2H), 7.75 (d, J= 15.6 Hz, 1H), 7.58 (d, J = 8.8 Hz, 2H), 7.43 (d, J = 15.6 Hz, 1H), 6.93 (d,J = 9.2 Hz, 2H), 6.89 (d, J = 9.6 Hz, 2H), 6.50 (m, 1H), 5.81 (d, J = 8.4 Hz,1H), 5.34 (dd, J = 11.2, 1.2 Hz, 1H), 5.20 (dd, J = 17.2, 1.2 Hz, 1H), 3.84(s, 3H), 3.40 (m, 5H), 3.24 (d, J = 16.8 Hz, 1H), 3.10 (d, J = 16.8 Hz, 1H),2.69 (m, 4H), 2.44 – 2.28 (m, 1H), 2.28 – 2.03 (m, 4H), 1.82 – 1.73 (m, 1H),1.58 (m, 5H), 1.45 (s, 3H), 1.36 (m, 1H), 1.30 (m, 1H), 1.16 (s, 3H), 1.11(m, 1H), 0.88 (d, J = 6.8 Hz, 3H), 0.74 (d, J = 6.8 Hz, 3H). 1 H NMR (400 MHz, CDCl 3 ): δ (ppm) 7.97 (d, J = 8.8 Hz, 2H), 7.75 (d, J = 15.6 Hz, 1H), 7.58 (d, J = 8.8 Hz, 2H) , 7.43 (d, J = 15.6 Hz, 1H), 6.93 (d, J = 9.2 Hz, 2H), 6.89 (d, J = 9.6 Hz, 2H), 6.50 (m, 1H), 5.81 (d, J = 8.4 Hz, 1H), 5.34 (dd, J = 11.2, 1.2 Hz, 1H), 5.20 (dd, J = 17.2, 1.2 Hz, 1H), 3.84(s, 3H), 3.40 (m, 5H), 3.24 ( d, J = 16.8 Hz, 1H), 3.10 (d, J = 16.8 Hz, 1H), 2.69 (m, 4H), 2.44 – 2.28 (m, 1H), 2.28 – 2.03 (m, 4H), 1.82 – 1.73 (m, 1H),1.58 (m, 5H), 1.45 (s, 3H), 1.36 (m, 1H), 1.30 (m, 1H), 1.16 (s, 3H), 1.11(m, 1H), 0.88 ( d, J = 6.8 Hz, 3H), 0.74 (d, J = 6.8 Hz, 3H).
13C NMR (100 MHz, CDCl3): δ (ppm) 217.1, 188.2, 169.0, 161.3, 153.9,143.0, 139.1, 130.6, 130.0, 128.6, 128.1, 119.7, 117.3, 114.4, 113.6, 74.6,68.5, 59.9, 58.2, 55.4, 52.6, 47.2, 45.5, 45.1, 44.0, 41.8, 36.7, 36.1, 34.5,30.5, 26.9, 26.4, 24.9, 16.8, 14.9, 11.5. 13 C NMR (100 MHz, CDCl 3 ): δ (ppm) 217.1, 188.2, 169.0, 161.3, 153.9, 143.0, 139.1, 130.6, 130.0, 128.6, 128.1, 119.7, 117.63, 114.6, 5.7 , 58.2, 55.4, 52.6, 47.2, 45.5, 45.1, 44.0, 41.8, 36.7, 36.1, 34.5, 30.5, 26.9, 26.4, 24.9, 16.8, 14.9, 11.5.
实施例3:14c的合成Embodiment 3: Synthesis of 14c
制备方法参考实施例1Preparation method reference example 1
产率64.5% 熔点134.5-136.3 oC Yield 64.5% Melting point 134.5-136.3 oC
1H NMR(400 MHz, CDCl3): δ (ppm) 7.99 (d, J = 8.8 Hz, 2H), 7.78 (d, J =15.6 Hz, 1H), 7.55 (d, J = 8.8 Hz, 2H), 7.36 (d, J = 15.6 Hz, 1H), 6.91 (d, J= 8.8 Hz, 2H), 6.70 (d, J = 8.8 Hz, 2H), 6.50 (dd, J = 17.2, 10.8 Hz, 1H),5.82 (d, J = 8.4 Hz, 1H), 5.36 (dd, J = 11.2, 1.2 Hz, 1H), 5.21 (dd, J =17.2, 1.2 Hz, 1H), 3.49 (s, 4H), 3.36 (m, 2H), 3.04 (s, 6H), 2.79 (s, 4H),2.33 (m, 1H), 2.29 – 2.05 (m, 4H), 1.82 – 1.74 (m, 1H), 1.71 – 1.63 (m, 2H),1.59 (m, 3H), 1.52 – 1.46 (m, 2H), 1.46 – 1.43 (s, 3H), 1.43 – 1.35 (m, 2H),1.33 (s, 1H), 1.30 – 1.28 (m, 1H), 1.18 (s, 3H), 1.12 (m, 1H), 0.88 (d, J =6.8 Hz, 3H), 0.74 (d, J = 6.8 Hz, 3H). 1 H NMR (400 MHz, CDCl 3 ): δ (ppm) 7.99 (d, J = 8.8 Hz, 2H), 7.78 (d, J = 15.6 Hz, 1H), 7.55 (d, J = 8.8 Hz, 2H) , 7.36 (d, J = 15.6 Hz, 1H), 6.91 (d, J = 8.8 Hz, 2H), 6.70 (d, J = 8.8 Hz, 2H), 6.50 (dd, J = 17.2, 10.8 Hz, 1H) ,5.82 (d, J = 8.4 Hz, 1H), 5.36 (dd, J = 11.2, 1.2 Hz, 1H), 5.21 (dd, J =17.2, 1.2 Hz, 1H), 3.49 (s, 4H), 3.36 ( m, 2H), 3.04 (s, 6H), 2.79 (s, 4H), 2.33 (m, 1H), 2.29 – 2.05 (m, 4H), 1.82 – 1.74 (m, 1H), 1.71 – 1.63 (m, 2H),1.59 (m, 3H), 1.52 – 1.46 (m, 2H), 1.46 – 1.43 (s, 3H), 1.43 – 1.35 (m, 2H),1.33 (s, 1H), 1.30 – 1.28 (m, 1H), 1.18 (s, 3H), 1.12 (m, 1H), 0.88 (d, J =6.8 Hz, 3H), 0.74 (d, J = 6.8 Hz, 3H).
13C NMR(100 MHz, CDCl3): δ (ppm) 217.2, 188.4, 169.0, 153.7, 151.8,144.2, 139.1, 130.4, 130.2, 129.2, 123.1, 117.3, 116.8, 113.7, 111.9, 74.6,68.4, 60.0, 58.2, 52.7, 47.3, 45.5, 45.0, 45.0, 41.8, 40.2, 36.8, 36.1, 34.5,31.6, 30.5, 26.9, 26.4, 24.9, 22.7, 16.8, 14.9, 14.2, 11.6. 13 C NMR (100 MHz, CDCl 3 ): δ (ppm) 217.2, 188.4, 169.0, 153.7, 151.8, 144.2, 139.1, 130.4, 130.2, 129.2, 123.1, 117.3, 116.6, 114.9, 6.7 , 58.2, 52.7, 47.3, 45.5, 45.0, 45.0, 41.8, 40.2, 36.8, 36.1, 34.5, 31.6, 30.5, 26.9, 26.4, 24.9, 22.7, 16.8, 14.9, 14.2, 11.6.
实施例4:14d的合成Example 4: Synthesis of 14d
制备方法参考实施例1Preparation method reference example 1
产率63.3% 熔点159.6-161.3oCYield 63.3% Melting point 159.6-161.3 o C
1H NMR (400 MHz, CDCl3): δ (ppm) 8.10 (s, 1H), 7.98 (d, J = 8.8 Hz,2H), 7.73 (d, J = 15.6 Hz, 1H), 7.59 (q, J = 9.2 Hz, 4H), 7.49 (d, J = 15.6Hz, 1H), 6.90 (d, J = 8.8 Hz, 2H), 6.52 (dd, J = 17.2, 11.2 Hz, 1H), 5.82 (d,J = 8.4 Hz, 1H), 5.35 (d, J = 11.2 Hz, 1H), 5.21 (d, J = 17.6 Hz, 1H), 3.41(m, 5H), 3.25 (d, J = 17.2 Hz, 1H), 3.11 (d, J = 17.2 Hz, 1H), 2.71 (m, 4H),2.35 (m, 2H), 2.25 (m, 2H), 2.18 (d, J = 8.4 Hz, 3H), 2.15 – 2.01 (m, 3H),1.78 (d, J = 14.4 Hz, 1H), 1.72 – 1.48 (m, 5H), 1.45 (s, 4H), 1.39 (m, 1H),1.28 (m, 1H), 1.17 (s, 3H), 1.12 (m, 1H), 0.89 (d, J = 6.8 Hz, 3H), 0.74 (d,J = 6.8 Hz, 3H). 1 H NMR (400 MHz, CDCl 3 ): δ (ppm) 8.10 (s, 1H), 7.98 (d, J = 8.8 Hz, 2H), 7.73 (d, J = 15.6 Hz, 1H), 7.59 (q, J = 9.2 Hz, 4H), 7.49 (d, J = 15.6Hz, 1H), 6.90 (d, J = 8.8 Hz, 2H), 6.52 (dd, J = 17.2, 11.2 Hz, 1H), 5.82 (d, J = 8.4 Hz, 1H), 5.35 (d, J = 11.2 Hz, 1H), 5.21 (d, J = 17.6 Hz, 1H), 3.41(m, 5H), 3.25 (d, J = 17.2 Hz, 1H) , 3.11 (d, J = 17.2 Hz, 1H), 2.71 (m, 4H),2.35 (m, 2H), 2.25 (m, 2H), 2.18 (d, J = 8.4 Hz, 3H), 2.15 – 2.01 ( m, 3H), 1.78 (d, J = 14.4 Hz, 1H), 1.72 – 1.48 (m, 5H), 1.45 (s, 4H), 1.39 (m, 1H), 1.28 (m, 1H), 1.17 (s , 3H), 1.12 (m, 1H), 0.89 (d, J = 6.8 Hz, 3H), 0.74 (d, J = 6.8 Hz, 3H).
13C NMR (100 MHz, CDCl3): δ (ppm) 217.2, 188.2, 169.0, 154.0, 142.8,139.1, 130.7, 129.2, 120.8, 119.8, 117.3, 113.6, 74.6, 68.5, 59.9, 58.2,52.6, 47.1, 45.5, 45.1, 44.0, 41.8, 36.8, 36.1, 34.5, 30.4, 26.9, 26.5, 24.9,24.6, 16.8, 14.9, 11.5. 13 C NMR (100 MHz, CDCl 3 ): δ (ppm) 217.2, 188.2, 169.0, 154.0, 142.8, 139.1, 130.7, 129.2, 120.8, 119.8, 117.3, 113.6, 74.6, 68.5, 119.9, 58.9 , 45.5, 45.1, 44.0, 41.8, 36.8, 36.1, 34.5, 30.4, 26.9, 26.5, 24.9, 24.6, 16.8, 14.9, 11.5.
实施例5:14e的合成Example 5: Synthesis of 14e
制备方法参考实施例1Preparation method reference example 1
产率58.5% 熔点138.6-140.3 oC Yield 58.5% Melting point 138.6-140.3 oC
1H NMR (400 MHz, CDCl3): δ (ppm) 8.25 (d, J = 8.8 Hz, 2H), 7.98 (d, J= 8.8 Hz, 2H), 7.82 – 7.71 (m, 3H), 7.65 (d, J = 15.6 Hz, 1H), 6.90 (d, J =8.8 Hz, 2H), 6.51 (dd, J = 17.2, 11.2 Hz, 1H), 5.80 (d, J = 8.4 Hz, 1H), 5.33(d, J = 11.2 Hz, 1H), 5.20 (d, J = 17.2 Hz, 1H), 3.54 – 3.30 (m, 5H), 3.24(d, J = 17.2 Hz, 1H), 3.10 (d, J = 17.2 Hz, 1H), 2.80 – 2.59 (m, 4H), 2.40 –2.28 (m, 1H), 2.28 – 2.12 (m, 2H), 2.13 – 2.04 (m, 2H), 1.77 (m, 2H), 1.58(m, 5H), 1.44 (s, 3H), 1.36 (m, 1H), 1.29 (m, 1H), 1.16 (s, 3H), 1.13 – 1.06(m, 1H), 0.87 (d, J = 6.8 Hz, 3H), 0.73 (d, J = 6.8 Hz, 3H). 1 H NMR (400 MHz, CDCl 3 ): δ (ppm) 8.25 (d, J = 8.8 Hz, 2H), 7.98 (d, J = 8.8 Hz, 2H), 7.82 – 7.71 (m, 3H), 7.65 ( d, J = 15.6 Hz, 1H), 6.90 (d, J = 8.8 Hz, 2H), 6.51 (dd, J = 17.2, 11.2 Hz, 1H), 5.80 (d, J = 8.4 Hz, 1H), 5.33( d, J = 11.2 Hz, 1H), 5.20 (d, J = 17.2 Hz, 1H), 3.54 – 3.30 (m, 5H), 3.24(d, J = 17.2 Hz, 1H), 3.10 (d, J = 17.2 Hz, 1H), 2.80 – 2.59 (m, 4H), 2.40 –2.28 (m, 1H), 2.28 – 2.12 (m, 2H), 2.13 – 2.04 (m, 2H), 1.77 (m, 2H), 1.58( m, 5H), 1.44 (s, 3H), 1.36 (m, 1H), 1.29 (m, 1H), 1.16 (s, 3H), 1.13 – 1.06(m, 1H), 0.87 (d, J = 6.8 Hz , 3H), 0.73 (d, J = 6.8 Hz, 3H).
13C NMR (100 MHz, CDCl3): δ (ppm) 217.1, 187.0, 169.0, 154.3, 148.3,141.6, 139.9, 139.1, 130.9, 128.8, 127.6, 125.9, 124.2, 117.3, 113.5, 74.6,68.5, 59.8, 58.2, 52.5, 47.0, 45.5, 45.1, 44.0, 41.8, 36.7, 36.1, 34.5, 30.5,26.9, 26.4, 24.9, 16.8, 14.9, 11.5. 13 C NMR (100 MHz, CDCl 3 ): δ (ppm) 217.1, 187.0, 169.0, 154.3, 148.3, 141.6, 139.9, 139.1, 130.9, 128.8, 127.6, 125.9, 124.6, 114.3, 8.8.7 , 58.2, 52.5, 47.0, 45.5, 45.1, 44.0, 41.8, 36.7, 36.1, 34.5, 30.5, 26.9, 26.4, 24.9, 16.8, 14.9, 11.5.
实施例6:14f的合成Embodiment 6: Synthesis of 14f
制备方法参考实施例1Preparation method reference example 1
产率56.3% 熔点102.9-104.2 oC Yield 56.3% Melting point 102.9-104.2 oC
1H NMR (400 MHz, CDCl3): δ (ppm) 8.51 (t, J = 1.6 Hz, 1H), 8.23 (m,1H), 8.02 (d, J = 8.8 Hz, 2H), 7.90 (d, J = 7.6 Hz, 1H), 7.81 (d, J = 15.6Hz, 1H), 7.67 (d, J = 15.6 Hz, 1H), 7.60 (t, J = 8.0 Hz, 1H), 6.92 (d, J =8.8 Hz, 2H), 6.51 (dd, J = 17.2, 11.2 Hz, 1H), 5.82 (d, J = 8.4 Hz, 1H), 5.35(dd, J = 11.2, 1.2 Hz, 1H), 5.21 (dd, J = 17.2, 1.2 Hz, 1H), 3.51 (s, 4H),3.36 (dd, J = 10.4, 6.8 Hz, 2H), 2.76 (s, 4H), 2.34 (m 1H), 2.29 – 2.19 (m,2H), 2.18 – 2.05 (m, 2H), 1.78 (dd, J = 14.4, 2.4 Hz, 1H), 1.74 – 1.63 (m,3H), 1.63 – 1.47 (m, 4H), 1.45 (s, 3H), 1.38 (m, 1H), 1.31 (m, 1H), 1.17 (s,3H), 1.12 (m, 1H), 0.88 (d, J = 6.8 Hz, 3H), 0.74 (d, J = 6.8 Hz, 3H). 1 H NMR (400 MHz, CDCl 3 ): δ (ppm) 8.51 (t, J = 1.6 Hz, 1H), 8.23 (m, 1H), 8.02 (d, J = 8.8 Hz, 2H), 7.90 (d, J = 7.6 Hz, 1H), 7.81 (d, J = 15.6Hz, 1H), 7.67 (d, J = 15.6 Hz, 1H), 7.60 (t, J = 8.0 Hz, 1H), 6.92 (d, J = 8.8 Hz, 2H), 6.51 (dd, J = 17.2, 11.2 Hz, 1H), 5.82 (d, J = 8.4 Hz, 1H), 5.35(dd, J = 11.2, 1.2 Hz, 1H), 5.21 (dd, J = 17.2, 1.2 Hz, 1H), 3.51 (s, 4H), 3.36 (dd, J = 10.4, 6.8 Hz, 2H), 2.76 (s, 4H), 2.34 (m 1H), 2.29 – 2.19 (m, 2H), 2.18 – 2.05 (m, 2H), 1.78 (dd, J = 14.4, 2.4 Hz, 1H), 1.74 – 1.63 (m,3H), 1.63 – 1.47 (m, 4H), 1.45 (s, 3H) , 1.38 (m, 1H), 1.31 (m, 1H), 1.17 (s,3H), 1.12 (m, 1H), 0.88 (d, J = 6.8 Hz, 3H), 0.74 (d, J = 6.8 Hz, 3H).
13C NMR (100 MHz, CDCl3): δ (ppm) 217.1, 187.0, 168.9, 154.2, 148.7,140.0, 139.1, 137.2, 134.3, 130.9, 129.9, 127.6, 124.7, 124.2, 122.1, 117.3,113.5, 74.6, 68.5, 59.8, 58.2, 52.5, 47.0, 45.5, 45.1, 41.8, 36.7, 36.1,34.5, 30.4, 26.9, 26.4, 24.9, 16.8, 14.9, 11.5. 13 C NMR (100 MHz, CDCL 3 ): Δ (PPM) 217.1, 187.0, 168.9, 154.2, 148.7 ,140.0, 137.2, 134.3, 130.9, 127.6, 124.2, 122.3, 117.3,113.5, 74.6.6.6 , 68.5, 59.8, 58.2, 52.5, 47.0, 45.5, 45.1, 41.8, 36.7, 36.1,34.5, 30.4, 26.9, 26.4, 24.9, 16.8, 14.9, 11.5.
实施例7:14g的合成Embodiment 7: the synthesis of 14g
制备方法参考实施例1Preparation method reference example 1
产率57.8% 熔点106.3-107.8oCYield 57.8% Melting point 106.3-107.8 o C
1H NMR (400 MHz, CDCl3): δ (ppm) 8.16 (dd, J = 8.0, 1.2 Hz, 1H), 7.74(m, 1H), 7.67 – 7.59 (m, 1H), 7.50 (dd, J = 7.6, 1.2 Hz, 1H), 7.40 (d, J =8.8 Hz, 2H), 7.17 (d, J = 15.6 Hz, 1H), 6.89 – 6.79 (m, 3H), 6.50 (dd, J =17.6, 11.2 Hz, 1H), 5.82 (d, J = 8.4 Hz, 1H), 5.35 (dd, J = 11.2, 1.6 Hz,1H), 5.21 (dd, J = 17.2, 1.6 Hz, 1H), 3.35 (m, 6H), 2.72 (s, 4H), 2.33 (m,1H), 2.30 – 2.14 (m, 2H), 2.10 (m, 2H), 1.78 (dd, J = 14.4, 2.8 Hz, 1H), 1.71– 1.58 (m, 3H), 1.59 – 1.46 (m, 4H), 1.44 (s, 3H), 1.41 – 1.34 (m, 1H), 1.30(m, 1H), 1.17 (s, 3H), 1.12 (m, 1H), 0.89 (d, J = 6.8 Hz, 3H), 0.73 (d, J =6.8 Hz, 3H). 1 H NMR (400 MHz, CDCl 3 ): δ (ppm) 8.16 (dd, J = 8.0, 1.2 Hz, 1H), 7.74(m, 1H), 7.67 – 7.59 (m, 1H), 7.50 (dd, J = 7.6, 1.2 Hz, 1H), 7.40 (d, J =8.8 Hz, 2H), 7.17 (d, J = 15.6 Hz, 1H), 6.89 – 6.79 (m, 3H), 6.50 (dd, J =17.6, 11.2 Hz, 1H), 5.82 (d, J = 8.4 Hz, 1H), 5.35 (dd, J = 11.2, 1.6 Hz, 1H), 5.21 (dd, J = 17.2, 1.6 Hz, 1H), 3.35 (m, 6H), 2.72 (s, 4H), 2.33 (m,1H), 2.30 – 2.14 (m, 2H), 2.10 (m, 2H), 1.78 (dd, J = 14.4, 2.8 Hz, 1H), 1.71– 1.58 (m, 3H), 1.59 – 1.46 (m, 4H), 1.44 (s, 3H), 1.41 – 1.34 (m, 1H), 1.30(m, 1H), 1.17 (s, 3H), 1.12 (m, 1H ), 0.89 (d, J = 6.8 Hz, 3H), 0.73 (d, J = 6.8 Hz, 3H).
13C NMR (100 MHz, CDCl3): δ (ppm) 217.1, 192.9, 169.0, 152.9, 147.0,139.1, 136.8, 133.8, 130.4, 130.2, 128.9, 124.5, 124.2, 122.5, 117.3, 114.7,74.6, 68.5, 59.9, 58.2, 52.6, 47.4, 45.5, 45.1, 44.0, 41.8, 36.7, 36.1, 34.5,30.5, 26.9, 26.4, 24.9, 16.8, 14.9, 11.5. 13 C NMR (100 MHz, CDCl 3 ): δ (ppm) 217.1, 192.9, 169.0, 152.9, 147.0, 139.1, 136.8, 133.8, 130.4, 130.2, 128.9, 124.5, 124.6, 1214.3, 8, 14 , 59.9, 58.2, 52.6, 47.4, 45.5, 45.1, 44.0, 41.8, 36.7, 36.1, 34.5, 30.5, 26.9, 26.4, 24.9, 16.8, 14.9, 11.5.
实施例8:14h的合成Embodiment 8: the synthesis of 14h
制备方法参考实施例1Preparation method reference example 1
产率89.76% 熔点122.5-124.1oCYield 89.76% Melting point 122.5-124.1 o C
1H NMR (400 MHz, CDCl3): δ (ppm) 7.98 (d, J = 8.8 Hz, 2H), 7.69 (d, J= 15.6 Hz, 1H), 7.49 (d, J = 15.6 Hz, 1H), 7.19 (t, J = 8.0 Hz, 1H), 7.04 (d,J = 7.6 Hz, 1H), 6.93 (s, 1H), 6.90 (d, J = 8.8 Hz, 2H), 6.71 (dd, J = 8.0,1.6 Hz, 1H), 6.51 (dd, J = 17.6, 11.2 Hz, 1H), 5.81 (d, J = 8.4 Hz, 1H), 5.35(dd, J = 11.2, 1.6 Hz, 1H), 5.20 (dd, J = 17.6, 1.6 Hz, 1H), 3.45 (t, J = 4.8Hz, 4H), 3.36 (s, 1H), 3.28 (d, J = 17.2 Hz, 1H), 3.14 (d, J = 17.2 Hz, 1H),2.75 (m, 4H), 2.41 – 2.29 (m, 1H), 2.29 – 2.18 (m, 2H), 2.17 – 2.04 (m, 3H),1.77 (dd, J = 14.4, 2.6 Hz, 1H), 1.71 – 1.46 (m, 5H), 1.45 (s, 3H), 1.40 –1.34 (m, 1H), 1.33 – 1.27 (m, 1H), 1.17 (s, 3H), 1.14 – 1.08 (m, 1H), 0.88(d, J = 6.8 Hz, 3H), 0.74 (d, J = 6.8 Hz, 3H). 1 H NMR (400 MHz, CDCl 3 ): δ (ppm) 7.98 (d, J = 8.8 Hz, 2H), 7.69 (d, J = 15.6 Hz, 1H), 7.49 (d, J = 15.6 Hz, 1H) , 7.19 (t, J = 8.0 Hz, 1H), 7.04 (d, J = 7.6 Hz, 1H), 6.93 (s, 1H), 6.90 (d, J = 8.8 Hz, 2H), 6.71 (dd, J = ( _ _ dd, J = 17.6, 1.6 Hz, 1H), 3.45 (t, J = 4.8Hz, 4H), 3.36 (s, 1H), 3.28 (d, J = 17.2 Hz, 1H), 3.14 (d, J = 17.2 Hz, 1H),2.75 (m, 4H), 2.41 – 2.29 (m, 1H), 2.29 – 2.18 (m, 2H), 2.17 – 2.04 (m, 3H),1.77 (dd, J = 14.4, 2.6 Hz, 1H), 1.71 – 1.46 (m, 5H), 1.45 (s, 3H), 1.40 – 1.34 (m, 1H), 1.33 – 1.27 (m, 1H), 1.17 (s, 3H), 1.14 – 1.08 (m, 1H), 0.88(d, J = 6.8 Hz, 3H), 0.74 (d, J = 6.8 Hz, 3H).
13C NMR (100 MHz, CDCl3): δ (ppm) 217.2, 188.2, 169.0, 154.0, 146.9,143.5, 139.1, 136.4, 130.7, 129.8, 128.4, 121.9, 118.8, 117.3, 117.0, 114.5,113.6, 74.6, 68.5, 59.9, 58.2, 52.6, 47.2, 5.5, 45.0, 44.0, 41.8, 36.7, 36.1,34.5, 30.5, 26.9, 26.4, 24.9, 16.9, 14.9, 11.5. 13 C NMR (100 MHz, CDCL 3 ): Δ (PPM) 217.2, 188.2, 169.0, 154.0, 146.9,143.5, 139.1, 136.4, 130.7, 128.4, 121.9, 117.3, 114.5,113.6, 74.6, 74.6.6 , 68.5, 59.9, 58.2, 52.6, 47.2, 5.5, 45.0, 44.0, 41.8, 36.7, 36.1, 34.5, 30.5, 26.9, 26.4, 24.9, 16.9, 14.9, 11.5.
实施例9:14i的合成Example 9: Synthesis of 14i
制备方法参考实施例1Preparation method reference example 1
产率93.4% 熔点132.5-133.9 oC Yield 93.4% Melting point 132.5-133.9 oC
1H NMR (400 MHz, CDCl3): δ (ppm) 7.98 (d, J = 8.8 Hz, 2H), 7.73 (d, J= 15.6 Hz, 1H), 7.47 (d, J = 8.4 Hz, 2H), 7.38 (d, J = 15.6 Hz, 1H), 6.90 (d,J = 8.8 Hz, 2H), 6.68 (d, J = 8.4 Hz, 2H), 6.53 (dd, J = 17.2, 11.2 Hz, 1H),5.82 (d, J = 8.4 Hz, 1H), 5.35 (d, J = 11.2 Hz, 1H), 5.21 (d, J = 17.2 Hz,1H), 3.42 (t, J = 4.8 Hz, 4H), 3.36 (m, 1H), 3.25 (d, J = 17.2 Hz, 1H), 3.10(d, J = 17.2 Hz, 1H), 2.70 (m, 4H), 2.40 – 2.30 (m, 1H), 2.29 – 2.14 (m, 2H),2.13 – 2.02 (m, 2H), 1.78 (m, 1H), 1.72 – 1.53 (m, 8H), 1.45 (s, 4H), 1.41 –1.31 (m, 3H), 1.17 (s, 3H), 1.12 (m, 1H), 0.88 (d, J = 6.8 Hz,3H), 0.74 (d, J= 6.8 Hz, 3H). 1 H NMR (400 MHz, CDCl 3 ): δ (ppm) 7.98 (d, J = 8.8 Hz, 2H), 7.73 (d, J = 15.6 Hz, 1H), 7.47 (d, J = 8.4 Hz, 2H) , 7.38 (d, J = 15.6 Hz, 1H), 6.90 (d, J = 8.8 Hz, 2H), 6.68 (d, J = 8.4 Hz, 2H), 6.53 (dd, J = 17.2, 11.2 Hz, 1H) ,5.82 (d, J = 8.4 Hz, 1H), 5.35 (d, J = 11.2 Hz, 1H), 5.21 (d, J = 17.2 Hz,1H), 3.42 (t, J = 4.8 Hz, 4H), 3.36 (m, 1H), 3.25 (d, J = 17.2 Hz, 1H), 3.10(d, J = 17.2 Hz, 1H), 2.70 (m, 4H), 2.40 – 2.30 (m, 1H), 2.29 – 2.14 ( m, 2H),2.13 – 2.02 (m, 2H), 1.78 (m, 1H), 1.72 – 1.53 (m, 8H), 1.45 (s, 4H), 1.41 –1.31 (m, 3H), 1.17 (s, 3H), 1.12 (m, 1H), 0.88 (d, J = 6.8 Hz, 3H), 0.74 (d, J = 6.8 Hz, 3H).
13C NMR (100 MHz, CDCl3): δ (ppm) 217.2, 188.4, 169.0, 153.8, 148.8,143.9, 139.1, 130.5, 130.3, 128.9, 125.5, 117.8, 117.32, 114.9, 113.7, 74.6,68.5, 59.9, 58.2, 52.6, 47.3, 45.5, 45.1, 44.0, 41.8, 36.7, 36.1, 34.5, 30.5,26.9, 26.4, 24.9, 16.8, 14.9, 11.6. 13 C NMR (100 MHz, CDCl 3 ): δ (ppm) 217.2, 188.4, 169.0, 153.8, 148.8, 143.9, 139.1, 130.5, 130.3, 128.9, 125.5, 117.8, 117.32, 114.9, 5.9, 6 , 58.2, 52.6, 47.3, 45.5, 45.1, 44.0, 41.8, 36.7, 36.1, 34.5, 30.5, 26.9, 26.4, 24.9, 16.8, 14.9, 11.6.
本发明部分目标物的体外抗菌活性研究In vitro antibacterial activity research of some target objects of the present invention
实验方法experimental method
最低抑菌浓度(MIC)测试方法Minimum Inhibitory Concentration (MIC) Test Method
1、实验菌株:选取耐甲氧西林金黄色葡萄球菌 (ATCC33591) 和耐甲氧西林金黄色葡萄球菌 (ATCC43300) 以及普通菌株大肠杆菌 (ATCC25922) 和金黄色葡萄球菌(ATCC25923) 为MIC值测定菌株。1. Experimental strains: Methicillin-resistant Staphylococcus aureus (ATCC33591) and methicillin-resistant Staphylococcus aureus (ATCC43300) as well as common strains of Escherichia coli (ATCC25922) and Staphylococcus aureus (ATCC25923) were selected as strains for MIC value determination .
2、药物稀释:以乙醇和无菌水为溶剂,将合成化合物和泰妙菌素分别溶解和稀释,配制成浓度为1280 μg·mL-1的母液,置于冰箱避光密封保存备用。2. Dilution of the drug: The synthetic compound and tiamulin were dissolved and diluted respectively with ethanol and sterile water as solvents to prepare a mother solution with a concentration of 1280 μg·mL -1 , and stored in a refrigerator protected from light and sealed for later use.
3、菌液制备:取各受试菌进行活化,挑取单克隆菌落于0.9%生理盐水中,将菌液配置成0.5麦氏浓度 (1.5×108 CFU·mL-1),后用 Mueller-Hinton无菌肉汤培养基 (MHB)稀释10倍备用。3. Bacterial solution preparation: take each test bacteria for activation, pick a single clone colony in 0.9% normal saline, prepare the bacterial solution to a McFarland concentration of 0.5 (1.5×10 8 CFU·mL -1 ), and then use Mueller -Hinton sterile broth (MHB) was diluted 10 times for later use.
4、阳性对照:选取侧链由单苯基哌嗪修饰的已知化合物7和泰妙菌素作为阳性对照。4. Positive control: the known compound 7 whose side chain is modified by monophenylpiperazine and tiamulin were selected as positive controls.
5、MIC 测定:在96孔板中除边缘孔和第二列孔外其余孔分别加入100 μL MHB,向第二孔加入160 μL MHB和40 μL母液。采用二倍稀释法分别对化合物和阳性对照进行稀释,共稀释成128 – 0.25 μg·mL-110个不同浓度梯度的稀释液,再向除边缘孔外每孔加入100μL菌浮液,充分混匀,最后向边缘孔每孔加入无菌水200 μL。37oC恒温培养18 – 24 h,观察受试菌的生长情况,以无生长的药物最低浓度为该药对该受试菌的MIC值;以泰妙菌素为阳性对照,以配制化合物浓度等同的乙醇溶液为阴性对照,每株受试菌进行3个平行实验,实验重复3次。实验结果见表1。5. MIC determination: Add 100 μL of MHB to the wells of the 96-well plate except the edge wells and the second row of wells, and add 160 μL of MHB and 40 μL of mother solution to the second well. The compound and the positive control were respectively diluted by the double dilution method, and were diluted into 10 dilutions of different concentration gradients of 128 – 0.25 μg mL -1 , and then 100 μL of the bacterial suspension was added to each well except the edge wells, and the mixture was fully mixed. Mix well, and finally add 200 μL of sterile water to each edge well. Incubate at a constant temperature of 37 o C for 18-24 hours, observe the growth of the test bacteria, and use the lowest concentration of the drug without growth as the MIC value of the drug to the test bacteria; use tiamulin as a positive control, and prepare the concentration of the compound The same ethanol solution was used as the negative control, and three parallel experiments were carried out for each strain of the tested bacteria, and the experiments were repeated three times. The experimental results are shown in Table 1.
表1:部分目标物MIC测试结果Table 1: MIC test results of some targets
结论:化合物14c – i对金黄色葡萄球菌标准耐药株都表现出优良的抗菌效果,与阳性对照泰妙菌素相比,本发明化合物的抗菌活性都得到了数倍的提高,其中化合物14c、14f、14g、14h和14i对四种菌株的抗菌效果均优于阳性对照泰妙菌素。特别是化合物14h和14i对两种金黄色葡萄球菌的耐药菌株ATCC33591和ATCC43300的MIC均达到0.5 μg·mL-1,比泰妙菌素约高16倍,同时对革兰氏阳性菌大肠杆菌ATCC25922的MIC可达到1 μg·mL-1,比泰妙菌素强32倍。综合以上结果,本发明中的化合物大多表现出了很好的抗菌效果。可望治疗由金黄色葡萄球菌和大肠杆菌引起的细菌感染。Conclusion: Compounds 14c-i have shown excellent antibacterial effects on standard drug-resistant strains of Staphylococcus aureus. Compared with the positive control tiamulin, the antibacterial activities of the compounds of the present invention have been improved several times, and compound 14c , 14f, 14g, 14h and 14i had better antibacterial effects on the four strains than the positive control tiamulin. In particular, the MICs of compounds 14h and 14i against two resistant strains of Staphylococcus aureus, ATCC33591 and ATCC43300, both reached 0.5 μg·mL -1 , about 16 times higher than that of tiamulin. The MIC of ATCC25922 can reach 1 μg·mL -1 , which is 32 times stronger than tiamulin. Based on the above results, most of the compounds of the present invention exhibit good antibacterial effects. It is expected to treat bacterial infections caused by Staphylococcus aureus and Escherichia coli.
Claims (5)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910601978.6A CN110437177B (en) | 2019-07-05 | 2019-07-05 | A kind of pleuromutilin derivative and its preparation method and application |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910601978.6A CN110437177B (en) | 2019-07-05 | 2019-07-05 | A kind of pleuromutilin derivative and its preparation method and application |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN110437177A CN110437177A (en) | 2019-11-12 |
| CN110437177B true CN110437177B (en) | 2022-11-18 |
Family
ID=68428916
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201910601978.6A Active CN110437177B (en) | 2019-07-05 | 2019-07-05 | A kind of pleuromutilin derivative and its preparation method and application |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN110437177B (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114230519B (en) * | 2021-12-06 | 2024-06-04 | 西咸新区沣厚原创医药科技有限公司 | Pleuromutilin cinnamate compounds with drug-resistant bacteria resisting activity, and synthetic method and application thereof |
| CN116396245B (en) * | 2023-04-18 | 2025-08-26 | 西北工业大学宁波研究院 | Pleuromutilin derivatives and preparation methods thereof |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB0027705D0 (en) * | 2000-11-11 | 2000-12-27 | S P A | Novel compounds |
| CN105837530B (en) * | 2016-04-18 | 2018-07-27 | 华南农业大学 | A kind of pleuromutilin derivative and its preparation method and application with piperazine sidechain |
| CN106699690B (en) * | 2016-12-07 | 2019-09-10 | 华南农业大学 | A kind of pleuromutilin derivative and its preparation method and application with acyl piperazine base side chain |
-
2019
- 2019-07-05 CN CN201910601978.6A patent/CN110437177B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN110437177A (en) | 2019-11-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN111793044B (en) | Pleuromutilin derivatives and uses thereof | |
| WO2019042444A1 (en) | Compound for inhibiting and degrading tyrosine protein kinase alk | |
| CN107625766B (en) | Use of a thiazole compound as an antibacterial synergist | |
| CN110437177B (en) | A kind of pleuromutilin derivative and its preparation method and application | |
| CN116768813B (en) | Pleuromutilin derivatives, uses and pharmaceutical compositions thereof | |
| CN110183429A (en) | 4-(N- methyl containing dithiocarbamate) amino piperidine myricetin derivative, preparation method and the usage | |
| CN107226782A (en) | It is a kind of that there is pleuromutilin analog derivative of alkaline parahelium side chain and its preparation method and application | |
| Zhou et al. | Discovery of novel pleuromutilin derivatives as potent antibacterial agents | |
| CN102367239B (en) | 2-aryl-2, 3-dihydro-4H-1, 3-benzothiazin-4-one derivatives and application thereof | |
| CN103242210B (en) | Mullinyl acetate containing substituted squaraine and its application | |
| MX2007009863A (en) | 9-substituted tetracyclines. | |
| CN107311995A (en) | Three ring isoxazole class derivatives and its preparation method and application | |
| JP5886285B2 (en) | Compounds having antibacterial activity against the genus CLOSTRIDIUM | |
| Fan et al. | Nosiheptide analogues as potential antibacterial agents via dehydroalanine region modifications: semi-synthesis, antimicrobial activity and molecular docking study | |
| CN102690332B (en) | Novel glycopeptide antibiotic derivatives and pharmaceutical composition, as well as its preparation method and use | |
| CN110437157B (en) | Aryl pyrimidine pleuromutilin derivative and preparation method and application thereof | |
| CN108484424A (en) | Side chain contains pleuromutilin analog derivative of quaternary ammonium salt group and application thereof | |
| JP2016539983A (en) | Alkylpyrazolylguanidine F1F0-ATPase inhibitor and therapeutic use thereof | |
| CN110372615B (en) | A kind of pleuromutilin derivative with 2-aminobenzenethiol and 1,2,3-triazole side chain and its preparation and application | |
| CN113402531A (en) | Fluoroquinolone derivative and application thereof as antibacterial drug | |
| CN103910664B (en) | A kind of antibacterial activity pleuromutilin-sulphone amide derivative and its preparation method and application | |
| TWI385168B (en) | Quinoline derivative as an antibacterial agent | |
| CN108976222B (en) | Heterocyclic ring-containing biaryl oxazolidinone compound and preparation method thereof | |
| CN107629023A (en) | A kind of new fragrant imido grpup thiazole compound, preparation method and use | |
| CN102453028A (en) | Antifungal agent-2- [ (2, 3-dihydro-4H-benzo [ b ] thiopyran-4-ylidene) hydrazono ] -4-oxotetrahydrothia (oxazole) -5-acetic acid derivative |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |