CN110283266B - A kind of pure organic room temperature phosphorescent polymer material with lactam ring structure and preparation method thereof - Google Patents
A kind of pure organic room temperature phosphorescent polymer material with lactam ring structure and preparation method thereof Download PDFInfo
- Publication number
- CN110283266B CN110283266B CN201910669244.1A CN201910669244A CN110283266B CN 110283266 B CN110283266 B CN 110283266B CN 201910669244 A CN201910669244 A CN 201910669244A CN 110283266 B CN110283266 B CN 110283266B
- Authority
- CN
- China
- Prior art keywords
- polymer material
- room temperature
- lactam ring
- ring structure
- pure organic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 150000003951 lactams Chemical group 0.000 title claims abstract description 34
- 239000002861 polymer material Substances 0.000 title claims abstract description 33
- 238000002360 preparation method Methods 0.000 title claims abstract description 15
- 239000000178 monomer Substances 0.000 claims abstract description 21
- 238000000034 method Methods 0.000 claims abstract description 12
- 239000001257 hydrogen Substances 0.000 claims abstract description 7
- 229910052739 hydrogen Inorganic materials 0.000 claims abstract description 7
- 239000000047 product Substances 0.000 claims description 21
- OZAIFHULBGXAKX-UHFFFAOYSA-N 2-(2-cyanopropan-2-yldiazenyl)-2-methylpropanenitrile Chemical group N#CC(C)(C)N=NC(C)(C)C#N OZAIFHULBGXAKX-UHFFFAOYSA-N 0.000 claims description 16
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical group CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 14
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 14
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 12
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 claims description 8
- 238000001035 drying Methods 0.000 claims description 8
- 229910052757 nitrogen Inorganic materials 0.000 claims description 8
- 238000002390 rotary evaporation Methods 0.000 claims description 8
- 239000007788 liquid Substances 0.000 claims description 7
- JWYVGKFDLWWQJX-UHFFFAOYSA-N 1-ethenylazepan-2-one Chemical compound C=CN1CCCCCC1=O JWYVGKFDLWWQJX-UHFFFAOYSA-N 0.000 claims description 6
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 claims description 6
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical group C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 claims description 6
- 238000006243 chemical reaction Methods 0.000 claims description 6
- DVPHDWQFZRBFND-DMHDVGBCSA-N 1-o-[2-[(3ar,5r,6s,6ar)-2,2-dimethyl-6-prop-2-enoyloxy-3a,5,6,6a-tetrahydrofuro[2,3-d][1,3]dioxol-5-yl]-2-[4-[(2s,3r)-1-butan-2-ylsulfanyl-2-(2-chlorophenyl)-4-oxoazetidin-3-yl]oxy-4-oxobutanoyl]oxyethyl] 4-o-[(2s,3r)-1-butan-2-ylsulfanyl-2-(2-chloropheny Chemical group C1([C@H]2[C@H](C(N2SC(C)CC)=O)OC(=O)CCC(=O)OC(COC(=O)CCC(=O)O[C@@H]2[C@@H](N(C2=O)SC(C)CC)C=2C(=CC=CC=2)Cl)[C@@H]2[C@@H]([C@H]3OC(C)(C)O[C@H]3O2)OC(=O)C=C)=CC=CC=C1Cl DVPHDWQFZRBFND-DMHDVGBCSA-N 0.000 claims description 4
- 239000003999 initiator Substances 0.000 claims description 4
- 239000003960 organic solvent Substances 0.000 claims description 4
- 230000035484 reaction time Effects 0.000 claims description 4
- 239000007795 chemical reaction product Substances 0.000 claims description 2
- 125000000896 monocarboxylic acid group Chemical group 0.000 claims description 2
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 claims description 2
- USHAGKDGDHPEEY-UHFFFAOYSA-L potassium persulfate Chemical compound [K+].[K+].[O-]S(=O)(=O)OOS([O-])(=O)=O USHAGKDGDHPEEY-UHFFFAOYSA-L 0.000 claims description 2
- 125000000217 alkyl group Chemical group 0.000 claims 1
- 239000002002 slurry Substances 0.000 claims 1
- 238000007334 copolymerization reaction Methods 0.000 abstract description 11
- 229920000642 polymer Polymers 0.000 abstract description 11
- 229920001519 homopolymer Polymers 0.000 abstract description 3
- 239000011159 matrix material Substances 0.000 abstract description 3
- 238000006116 polymerization reaction Methods 0.000 abstract description 3
- 239000000843 powder Substances 0.000 abstract description 2
- 230000008569 process Effects 0.000 abstract description 2
- 230000002349 favourable effect Effects 0.000 abstract 1
- 229910000510 noble metal Inorganic materials 0.000 abstract 1
- -1 alkyl chain compound Chemical class 0.000 description 7
- 238000010009 beating Methods 0.000 description 7
- 238000000295 emission spectrum Methods 0.000 description 6
- 238000006862 quantum yield reaction Methods 0.000 description 6
- 238000001228 spectrum Methods 0.000 description 6
- 238000005303 weighing Methods 0.000 description 6
- 239000000463 material Substances 0.000 description 5
- 230000005587 bubbling Effects 0.000 description 3
- 229910001385 heavy metal Inorganic materials 0.000 description 3
- RIMXEJYJXDBLIE-UHFFFAOYSA-N 6-bromohex-1-ene Chemical compound BrCCCCC=C RIMXEJYJXDBLIE-UHFFFAOYSA-N 0.000 description 2
- 150000001491 aromatic compounds Chemical class 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 150000002894 organic compounds Chemical class 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N Benzoic acid Natural products OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical group [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 150000001559 benzoic acids Chemical class 0.000 description 1
- 238000007664 blowing Methods 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 230000009849 deactivation Effects 0.000 description 1
- 230000005281 excited state Effects 0.000 description 1
- 230000008014 freezing Effects 0.000 description 1
- 238000007710 freezing Methods 0.000 description 1
- 229910052741 iridium Inorganic materials 0.000 description 1
- 125000002524 organometallic group Chemical group 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 239000010970 precious metal Substances 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F214/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen
- C08F214/16—Monomers containing bromine or iodine
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F216/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal or ketal radical
- C08F216/02—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal or ketal radical by an alcohol radical
- C08F216/10—Carbocyclic compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/52—Amides or imides
- C08F220/54—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide
- C08F220/60—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide containing nitrogen in addition to the carbonamido nitrogen
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/52—Amides or imides
- C08F220/54—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide
- C08F220/60—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide containing nitrogen in addition to the carbonamido nitrogen
- C08F220/606—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide containing nitrogen in addition to the carbonamido nitrogen and containing other heteroatoms
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Polyamides (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
Abstract
Description
技术领域technical field
本发明属于有机发光材料领域,具体涉及一种具有内酰胺环结构的纯有机室温磷光聚合物材料及其制备方法。The invention belongs to the field of organic light-emitting materials, in particular to a pure organic room temperature phosphorescent polymer material with a lactam ring structure and a preparation method thereof.
背景技术Background technique
室温磷光(RTP)与荧光相比,具有较大的Stokes位移和较长的寿命,在生物成像、分子开关和有机发光二极管(OLED)等领域都有广泛的实际应用前景。传统的无机和有机金属磷光材料常使用Pt、Ir等重金属,然而重金属价格昂贵、毒性高、不够环保。在不含重金属的情况下,单重态和三重态之间通常存在自旋禁阻,因此纯有机化合物通常需要在冷冻低温和惰性环境中才能观察到磷光。Compared with fluorescence, room temperature phosphorescence (RTP) has a larger Stokes shift and longer lifetime, and has a wide range of practical application prospects in bioimaging, molecular switches, and organic light-emitting diodes (OLEDs). Traditional inorganic and organometallic phosphorescent materials often use heavy metals such as Pt and Ir. However, heavy metals are expensive, highly toxic, and not environmentally friendly. In the absence of heavy metals, there is usually a spin-forbidden barrier between the singlet and triplet states, so pure organic compounds usually require freezing temperatures and inert environments to observe phosphorescence.
目前制备纯有机室温磷光材料,通常会使发光体形成晶体或是将发光体引入聚合物,为发光体分子提供一个刚性的分子构型环境,减弱三重激发态分子的非辐射失活,提高磷光发射。不过晶体需要十分严格的生长条件,而无定形材料的制备则更为简便,并且重复性和加工性较好。内酰胺是有机化合物中常见的一种环状结构,其环刚性有助于增强磷光发射。除此以外,聚合物基质和氢键也能为内酰胺类磷光聚合物材料提供刚性环境,使其展现出较好的发磷光性能。但迄今为止,不含氢键的内酰胺类磷光聚合物材料鲜有报道。At present, in the preparation of pure organic room temperature phosphorescent materials, the luminophore is usually formed into a crystal or the luminophore is introduced into a polymer, which provides a rigid molecular configuration environment for the luminophore molecule, reduces the non-radiative deactivation of triplet excited state molecules, and improves phosphorescence. emission. However, crystals require very stringent growth conditions, while amorphous materials are easier to prepare, with better reproducibility and processability. Lactams are cyclic structures commonly found in organic compounds, and their ring rigidity contributes to enhanced phosphorescence emission. In addition, the polymer matrix and hydrogen bonds can also provide a rigid environment for the lactam-based phosphorescent polymer materials to exhibit better phosphorescent properties. But so far, lactam-based phosphorescent polymer materials without hydrogen bonds are rarely reported.
发明内容SUMMARY OF THE INVENTION
本发明的目的就是为了解决上述问题而提供一种具有内酰胺环结构的纯有机室温磷光聚合物材料及其制备方法。The purpose of the present invention is to provide a pure organic room temperature phosphorescent polymer material with a lactam ring structure and a preparation method thereof in order to solve the above problems.
本发明的目的通过以下技术方案实现:The object of the present invention is achieved through the following technical solutions:
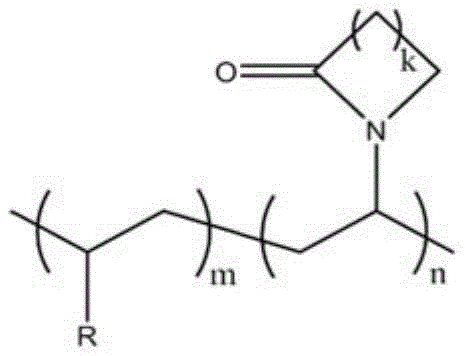
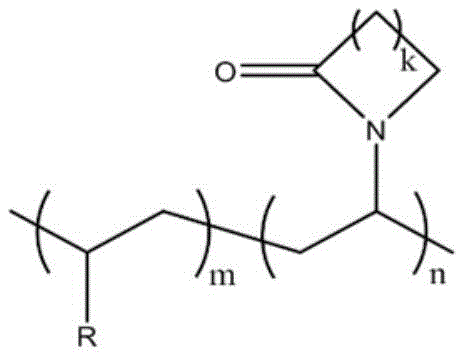
一种具有内酰胺环结构的纯有机室温磷光聚合物材料,该聚合物材料的结构通式如下:A pure organic room temperature phosphorescent polymer material with a lactam ring structure, the general structural formula of the polymer material is as follows:
其中,k=1、2、3、4;m=1,n=1-800;R=磷光单体。Wherein, k=1, 2, 3, 4; m=1, n=1-800; R=phosphorescent monomer.
所述的R为Ar-R1或烷基链化合物,其中,R1=Cl,Br,I,OH,CHO,COCH3,COOH,COOCH3或OCH3。The R is Ar-R 1 or an alkyl chain compound, wherein R 1 =Cl, Br, I, OH, CHO, COCH 3 , COOH, COOCH 3 or OCH 3 .
一种具有内酰胺环结构的纯有机室温磷光聚合物材料的制备方法,将磷光单体和不含氢键的内酰胺类单体溶解在有机溶剂中,并加入引发剂,在惰性气氛下反应,反应产物滴入丙酮或乙醚中打浆,除去清液旋蒸干燥后即得到产品。A method for preparing a pure organic room temperature phosphorescent polymer material with a lactam ring structure. Phosphorescent monomers and lactam monomers without hydrogen bonds are dissolved in an organic solvent, and an initiator is added to react in an inert atmosphere. , the reaction product is dropped into acetone or ether for beating, and the product is obtained after removing the clear liquid by rotary evaporation and drying.
优选地,所述磷光单体为含氧芳香化合物、含重原子芳香化合物或烷基链化合物。Preferably, the phosphorescent monomer is an oxygen-containing aromatic compound, a heavy atom-containing aromatic compound or an alkyl chain compound.
优选地,所述内酰胺类单体为N-乙烯基吡咯烷酮或N-乙烯基己内酰胺。Preferably, the lactam-based monomer is N-vinylpyrrolidone or N-vinylcaprolactam.
优选地,所述有机溶剂为乙醇或N,N-二甲基甲酰胺。Preferably, the organic solvent is ethanol or N,N-dimethylformamide.
优选地,所述磷光单体与内酰胺类单体的摩尔比是1:90-110。Preferably, the molar ratio of the phosphorescent monomer to the lactam monomer is 1:90-110.
优选地,所述引发剂为偶氮二异丁腈或过硫酸钾,用量与磷光单体的质量比为1:3~5。Preferably, the initiator is azobisisobutyronitrile or potassium persulfate, and the mass ratio of the amount to the phosphorescent monomer is 1:3-5.
优选地,所述惰性气氛为氮气,反应温度为60-80℃,反应时间为10-14h,进一步优选地,反应温度为70℃,反应时间为12h。Preferably, the inert atmosphere is nitrogen, the reaction temperature is 60-80°C, and the reaction time is 10-14h, and further preferably, the reaction temperature is 70°C and the reaction time is 12h.
本发明主要利用无氢键、非芳香类内酰胺类聚合物(如聚乙烯基吡咯烷酮或聚乙烯基己内酰胺)以及通过其单体(N-乙烯基吡咯烷酮或N-乙烯基己内酰胺)与微量的磷光单体共聚,得到一系列无定形纯有机室温磷光聚合物材料。随着内酰胺类单体的自聚、与磷光单体分别共聚,可以实现对聚合物波长的调控,从而进一步实现发光颜色的变化,拓展了内酰胺环类发光材料的潜在应用。The present invention mainly utilizes non-hydrogen bond, non-aromatic lactam polymers (such as polyvinylpyrrolidone or polyvinylcaprolactam) and through its monomer (N-vinylpyrrolidone or N-vinylcaprolactam) and trace phosphorescence The monomers were copolymerized to obtain a series of amorphous pure organic room temperature phosphorescent polymer materials. With the self-polymerization of lactam monomers and the separate copolymerization with phosphorescent monomers, the regulation of the polymer wavelength can be realized, thereby further realizing the change of luminescent color, and expanding the potential application of lactam ring luminescent materials.
与现有技术相比,本发明具有如下显著的优点:Compared with the prior art, the present invention has the following significant advantages:
本发明采用不含氢键的内酰胺环类磷光聚合物材料为基质,内酰胺环类均聚物和其与磷光单体的共聚产物皆表现出了较好的磷光特性;同时,,本发明采用了微量特征单体共聚的方法实现波长调节,没有贵金属参与,制备方便、成本合理,调节效率较高;磷光荧光单体的制备以及聚合和后处理过程均比较简便;无定形粉末的产物状态,利于进一步加工,内酰胺环类聚合物生物相容性优异,在生物领域具有应用前景。The present invention adopts lactam ring type phosphorescent polymer material without hydrogen bond as matrix, and both lactam ring type homopolymer and its copolymerization product with phosphorescent monomer show good phosphorescent properties; at the same time, the present invention The wavelength adjustment is realized by the copolymerization of trace characteristic monomers, without the participation of precious metals, the preparation is convenient, the cost is reasonable, and the adjustment efficiency is high; the preparation of phosphorescent fluorescent monomers, as well as the polymerization and post-treatment processes are relatively simple; the product state of the amorphous powder , which is conducive to further processing, and the lactam ring polymer has excellent biocompatibility and has application prospects in the biological field.
附图说明Description of drawings
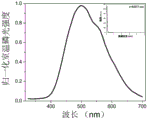
图1为实施例1制备的共聚产物的室温磷光发射谱图及寿命谱图。FIG. 1 is the room temperature phosphorescence emission spectrum and lifetime spectrum of the copolymerization product prepared in Example 1. FIG.
图2为实施例2制备的共聚产物的室温磷光发射谱图及寿命谱图。FIG. 2 is the room temperature phosphorescence emission spectrum and lifetime spectrum of the copolymerization product prepared in Example 2. FIG.
图3为实施例3制备的共聚产物的室温磷光发射谱图及寿命谱图。FIG. 3 is a room temperature phosphorescence emission spectrum and a lifetime spectrum of the copolymerization product prepared in Example 3. FIG.
图4为实施例4制备的共聚产物的温磷光发射谱图及寿命谱图。FIG. 4 is the thermophosphorescence emission spectrum and lifetime spectrum of the copolymerization product prepared in Example 4. FIG.
图5为实施例5制备的共聚产物的温磷光发射谱图及寿命谱图。FIG. 5 is the thermophosphorescence emission spectrum and lifetime spectrum of the copolymerization product prepared in Example 5. FIG.
图6为实施例6制备的共聚产物的温磷光发射谱图及寿命谱图。FIG. 6 is the thermophosphorescence emission spectrum and lifetime spectrum of the copolymerization product prepared in Example 6. FIG.
具体实施方式Detailed ways
下面结合附图和具体实施例对本发明进行详细说明。The present invention will be described in detail below with reference to the accompanying drawings and specific embodiments.
实施例1Example 1
一种具有内酰胺环结构的纯有机室温磷光聚合物材料的制备方法,具体步骤如下:称取33mg苯甲酸类化合物、10mg偶氮二异丁腈溶于1.5mL乙醇,加入1.5mL N-乙烯基吡咯烷酮,溶解,利用针头鼓气,氮气保护下70℃下,反应12h,所得产物滴入丙酮中打浆,除去清液,旋蒸干燥后得到纯有机室温磷光聚合物材料。A method for preparing a pure organic room temperature phosphorescent polymer material with a lactam ring structure, the specific steps are as follows: weighing 33 mg of benzoic acid compounds and 10 mg of azobisisobutyronitrile, dissolved in 1.5 mL of ethanol, adding 1.5 mL of N-vinylpyrrolidone , dissolved, inflated with a needle, and reacted for 12 hours at 70°C under nitrogen protection, the obtained product was dropped into acetone for beating, the supernatant was removed, and the pure organic room temperature phosphorescent polymer material was obtained after rotary evaporation and drying.
所制得的产品聚合物具体结构如下:The specific structure of the prepared product polymer is as follows:
其磷光发射峰在428nm左右(请参阅图1),寿命为0.947ms,量子产率为0.8%。Its phosphorescence emission peak is around 428nm (see Figure 1), the lifetime is 0.947ms, and the quantum yield is 0.8%.
实施例2Example 2
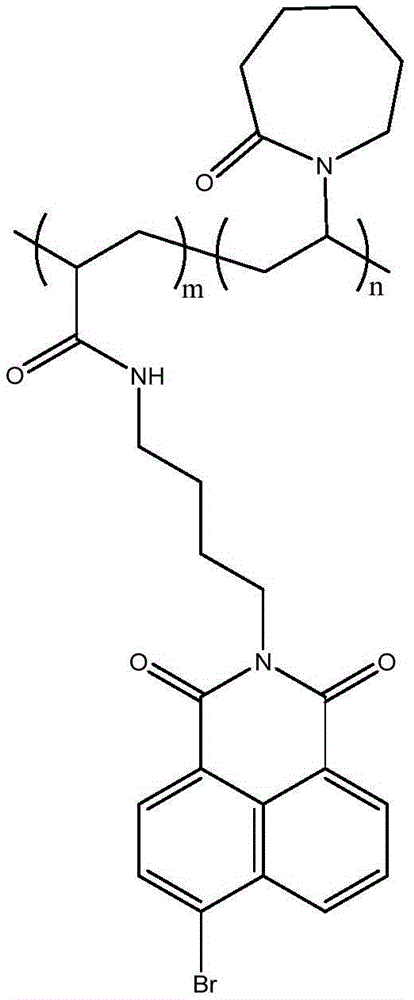
一种具有内酰胺环结构的纯有机室温磷光聚合物材料的制备方法,具体步骤如下:称取38mg溴萘酰亚胺类化合物、8mg偶氮二异丁腈溶于1mL N,N-二甲基甲酰胺,加入1mLN-乙烯基吡咯烷酮,溶解,利用针头鼓气,氮气保护下70℃反应12h。所得产物滴入丙酮中打浆,除去清液,旋蒸干燥后得到纯有机室温磷光聚合物材料。A method for preparing a pure organic room temperature phosphorescent polymer material with a lactam ring structure, the specific steps are as follows: weighing 38 mg of bromonaphthalimide compound, 8 mg of azobisisobutyronitrile and dissolved in 1 mL of N,N-dimethylformaldehyde amide, add 1 mL of N-vinylpyrrolidone, dissolve, use a needle to blow air, and react at 70 °C for 12 h under nitrogen protection. The obtained product is dropped into acetone for beating, the clear liquid is removed, and the pure organic room temperature phosphorescent polymer material is obtained after rotary evaporation and drying.
其磷光发射峰在582nm左右(请参阅图2),且有610nm左右伴峰,寿命为0.463ms,量子产率为1.6%。Its phosphorescence emission peak is around 582nm (please refer to Fig. 2), and there is a secondary peak around 610nm, the lifetime is 0.463ms, and the quantum yield is 1.6%.
实施例3Example 3
一种具有内酰胺环结构的纯有机室温磷光聚合物材料的制备方法,具体步骤如下:称取20mg偶氮二异丁腈溶于2mL乙醇,加入2mL N-乙烯基吡咯烷酮,溶解,利用针头鼓气,鼓气后用针头打入204mg 6-溴-1-己烯单体,氮气保护下70℃反应12h。所得产物滴入乙醚中打浆,除去清液,旋蒸干燥后得到纯有机室温磷光聚合物材料。A method for preparing a pure organic room temperature phosphorescent polymer material with a lactam ring structure, the specific steps are as follows: weighing 20 mg of azobisisobutyronitrile and dissolving it in 2 mL of ethanol, adding 2 mL of N-vinylpyrrolidone, dissolving, and blowing with a needle, After bubbling, 204 mg of 6-bromo-1-hexene monomer was injected with a needle, and the reaction was carried out at 70 °C for 12 h under nitrogen protection. The obtained product is dropped into ether for beating, the clear liquid is removed, and the pure organic room temperature phosphorescent polymer material is obtained after rotary evaporation and drying.
所制得的产品聚合物具体结构如下:The specific structure of the prepared product polymer is as follows:
其磷光发射峰在496nm左右(请参阅图3),寿命为0.037ms,量子产率为0.9%。Its phosphorescence emission peak is around 496nm (see Figure 3), the lifetime is 0.037ms, and the quantum yield is 0.9%.
实施例4Example 4
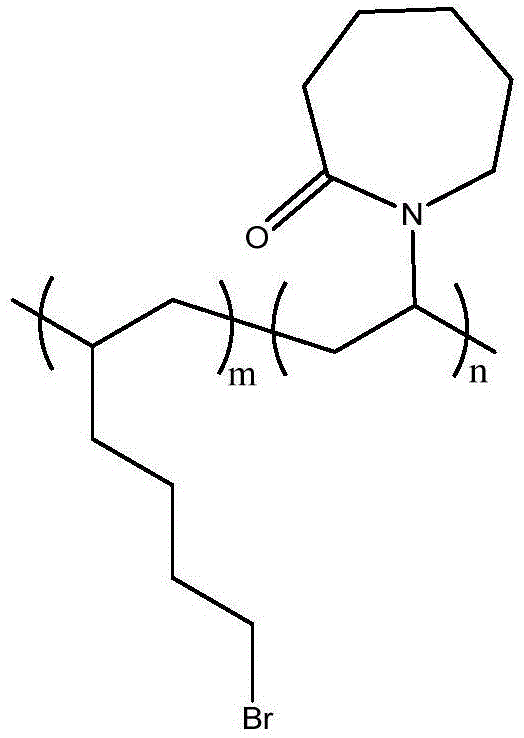
一种具有内酰胺环结构的纯有机室温磷光聚合物材料的制备方法,具体步骤如下:称取7.88mg苯甲酸类化合物、25.4mg偶氮二异丁腈溶于2mL乙醇,加入500mg N-乙烯基己内酰胺,溶解,利用针头鼓气,氮气保护下60℃反应12h。所得产物滴入乙醚中打浆,除去清液,旋蒸干燥后得到纯有机室温磷光聚合物材料。A method for preparing a pure organic room temperature phosphorescent polymer material with a lactam ring structure, the specific steps are as follows: weighing 7.88 mg of benzoic acid compound, 25.4 mg of azobisisobutyronitrile, dissolved in 2 mL of ethanol, adding 500 mg of N-vinyl caprolactam , dissolve, use needle bubbling, and react at 60°C for 12h under nitrogen protection. The obtained product is dropped into ether for beating, the clear liquid is removed, and the pure organic room temperature phosphorescent polymer material is obtained after rotary evaporation and drying.
所制得的产品聚合物具体结构如下:The specific structure of the prepared product polymer is as follows:
其磷光发射峰在425nm左右(请参阅图4),寿命为0.348ms,量子产率为0.8%。Its phosphorescence emission peak is around 425nm (see Figure 4), the lifetime is 0.348ms, and the quantum yield is 0.8%.
实施例5Example 5
一种具有内酰胺环结构的纯有机室温磷光聚合物材料的制备方法,具体步骤如下:称取14.4mg溴萘酰亚胺类化合物、25.7mg偶氮二异丁腈溶于2mL乙醇,加入500mg N-乙烯基己内酰胺,溶解,利用针头鼓气,氮气保护下60℃反应12h。所得产物滴入乙醚中打浆,除去清液,旋蒸干燥后得到纯有机室温磷光聚合物材料。A method for preparing a pure organic room temperature phosphorescent polymer material with a lactam ring structure, the specific steps are as follows: weighing 14.4 mg of bromonaphthalimide compound and 25.7 mg of azobisisobutyronitrile dissolved in 2 mL of ethanol, adding 500 mg of N- Vinyl caprolactam, dissolve, use needle bubbling, react at 60°C for 12h under nitrogen protection. The obtained product is dropped into ether for beating, the clear liquid is removed, and the pure organic room temperature phosphorescent polymer material is obtained after rotary evaporation and drying.
所制得的产品聚合物具体结构如下:The specific structure of the prepared product polymer is as follows:
其磷光发射峰在579nm左右(请参阅图5),寿命为0.439ms,量子产率为0.6%。Its phosphorescence emission peak is around 579nm (see Figure 5), the lifetime is 0.439ms, and the quantum yield is 0.6%.
实施例6Example 6
一种具有内酰胺环结构的纯有机室温磷光聚合物材料的制备方法,具体步骤如下:称取5.87mg 6-溴-1-己烯、25.3mg偶氮二异丁腈溶于2mL乙醇,加入500mg N-乙烯基己内酰胺,溶解,利用针头鼓气,氮气保护下60℃反应12h。所得产物滴入乙醚中打浆,除去清液,旋蒸干燥后得到纯有机室温磷光聚合物材料。A preparation method of a pure organic room temperature phosphorescent polymer material with a lactam ring structure, the specific steps are as follows: weighing 5.87mg 6-bromo-1-hexene, 25.3mg azobisisobutyronitrile dissolved in 2mL ethanol, adding 500mg N -Vinyl caprolactam, dissolved, inflated with a needle, and reacted at 60°C for 12h under nitrogen protection. The obtained product is dropped into ether for beating, the clear liquid is removed, and the pure organic room temperature phosphorescent polymer material is obtained after rotary evaporation and drying.
所制得的产品聚合物具体结构如下:The specific structure of the prepared product polymer is as follows:
其磷光发射峰在568nm左右(请参阅图6),寿命为0.364ms,量子产率为1.4%。Its phosphorescence emission peak is around 568nm (see Figure 6), the lifetime is 0.364ms, and the quantum yield is 1.4%.
上述的对实施例的描述是为便于该技术领域的普通技术人员能理解和使用发明。熟悉本领域技术的人员显然可以容易地对这些实施例做出各种修改,并把在此说明的一般原理应用到其他实施例中而不必经过创造性的劳动。因此,本发明不限于上述实施例,本领域技术人员根据本发明的揭示,不脱离本发明范畴所做出的改进和修改都应该在本发明的保护范围之内。The foregoing description of the embodiments is provided to facilitate understanding and use of the invention by those of ordinary skill in the art. It will be apparent to those skilled in the art that various modifications to these embodiments can be readily made, and the generic principles described herein can be applied to other embodiments without inventive step. Therefore, the present invention is not limited to the above-mentioned embodiments, and improvements and modifications made by those skilled in the art according to the disclosure of the present invention without departing from the scope of the present invention should all fall within the protection scope of the present invention.
Claims (8)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910669244.1A CN110283266B (en) | 2019-07-23 | 2019-07-23 | A kind of pure organic room temperature phosphorescent polymer material with lactam ring structure and preparation method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910669244.1A CN110283266B (en) | 2019-07-23 | 2019-07-23 | A kind of pure organic room temperature phosphorescent polymer material with lactam ring structure and preparation method thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN110283266A CN110283266A (en) | 2019-09-27 |
| CN110283266B true CN110283266B (en) | 2020-12-04 |
Family
ID=68022263
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201910669244.1A Active CN110283266B (en) | 2019-07-23 | 2019-07-23 | A kind of pure organic room temperature phosphorescent polymer material with lactam ring structure and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN110283266B (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN113735772B (en) * | 2021-09-22 | 2024-04-09 | 四川羽玺新材料股份有限公司 | A fluorescent functional monomer, a pressure-sensitive adhesive capable of timely detecting coating defects and a preparation method thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SU374336A1 (en) * | 1971-01-05 | 1973-03-20 | Институт высокомолекул рных соединений СССР | METHOD OF OBTAINING COPOLYMERS ON OS L ^ -VINIL PIRROL IDONA |
| CN108218923A (en) * | 2018-03-06 | 2018-06-29 | 南京邮电大学 | A kind of water-soluble phosphorus photopolymer for detecting pH and its preparation method and application |
-
2019
- 2019-07-23 CN CN201910669244.1A patent/CN110283266B/en active Active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SU374336A1 (en) * | 1971-01-05 | 1973-03-20 | Институт высокомолекул рных соединений СССР | METHOD OF OBTAINING COPOLYMERS ON OS L ^ -VINIL PIRROL IDONA |
| CN108218923A (en) * | 2018-03-06 | 2018-06-29 | 南京邮电大学 | A kind of water-soluble phosphorus photopolymer for detecting pH and its preparation method and application |
Non-Patent Citations (1)
| Title |
|---|
| "Recent Advances in Polymer-Based Metal-Free Room-Temperature Phosphorescent Materials";Nan Gan等;《advanced functional materials》;20181231;全文 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN110283266A (en) | 2019-09-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7910687B2 (en) | Conjugated polymers containing arylamine units, the representation thereof and the use of the same | |
| US7638594B2 (en) | Conjugated polymers containing spirobifluorene units and the use thereof | |
| JP5073187B2 (en) | Blue light emitting polymer and organic electroluminescent device using the same | |
| CN100374526C (en) | Cyclopentaphenanthrene-based compound and organic electroluminescent device using the same | |
| CN111269344B (en) | White light fluorescent high molecular compound based on space charge transfer effect and preparation method and application thereof | |
| KR20030038514A (en) | Light Emitting Polymer Composition, and Organic Electroluminescence Device and Production Process Thereof | |
| TWI705978B (en) | Polymer, coating compositions comprising the same, and organic light emitting device using the same | |
| JP4904002B2 (en) | Spirocyclopentaphenanthrenefluorene compound and organic EL device using the same | |
| JP2003253129A (en) | Luminescent composition | |
| CN110283266B (en) | A kind of pure organic room temperature phosphorescent polymer material with lactam ring structure and preparation method thereof | |
| EP2566932A1 (en) | Polymer-based organic electroluminescent device | |
| CN111234083B (en) | A kind of fluorescent/phosphorescence hybrid white light polymer compound based on space charge transfer effect and its preparation method and application | |
| JP4767547B2 (en) | Thieno [3,2-b] indole polymer and organic electroluminescence device using the same | |
| CA2554940A1 (en) | Organic electroluminescent polymer having 9,9-di(fluorenyl)-2,7-fluorenyl unit and organic electroluminescent device manufactured using the same | |
| JP6983460B2 (en) | New polymer and organic light emitting device containing it | |
| CN110776503A (en) | Organic electroluminescent material based on halogenated aromatic ketone derivatives, and preparation method and application thereof | |
| KR101350744B1 (en) | Electriluminescent polymer containing benzothiadiazole derivatives and organoelectriluminescent device emloying the same | |
| JPH0854833A (en) | Organic electroluminescent device | |
| CN113637106B (en) | High-molecular luminescent material with space charge transfer polymer as sensitizer, preparation method thereof and organic electroluminescent device | |
| CN113698523B (en) | Polymer Compounds and Organic Electroluminescent Devices Containing Space Charge Transfer Polymer Sensitizers and Resonant Condensed Ring Units | |
| CN114805741B (en) | Polymer luminescent material based on polyurethane main chain, and preparation method and application thereof | |
| KR100718793B1 (en) | Organic electroluminescent molecules containing 9,9-di (3,3'-bicarbazyl) -2,7-fluorenyl units and electroluminescent devices using the same | |
| CN118108887A (en) | Money metal complex luminous polymer and preparation method and application thereof | |
| JPWO2004077888A1 (en) | Organic EL element material and organic EL element using the same | |
| KR20080078493A (en) | High color purity blue electroluminescent copolymer and organic electroluminescent device using same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |