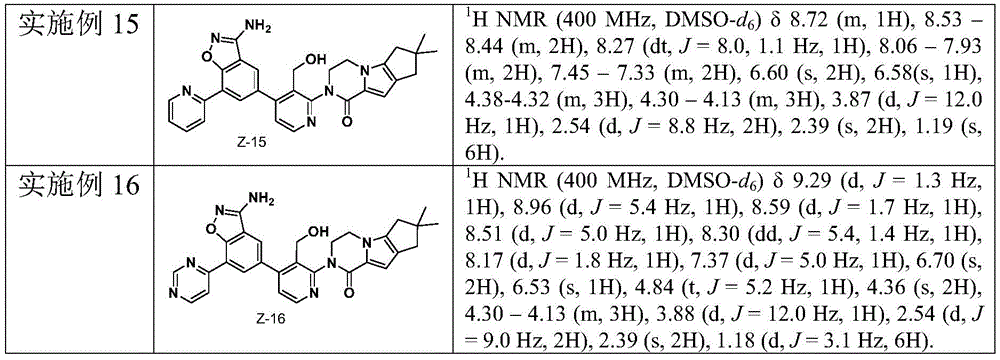
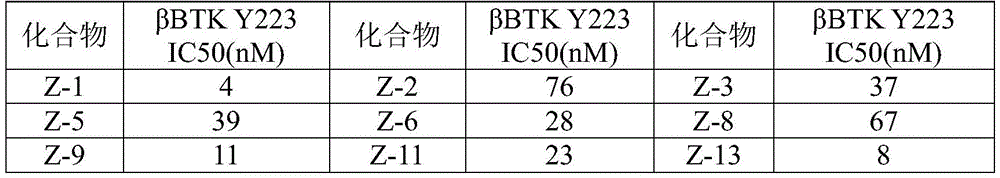
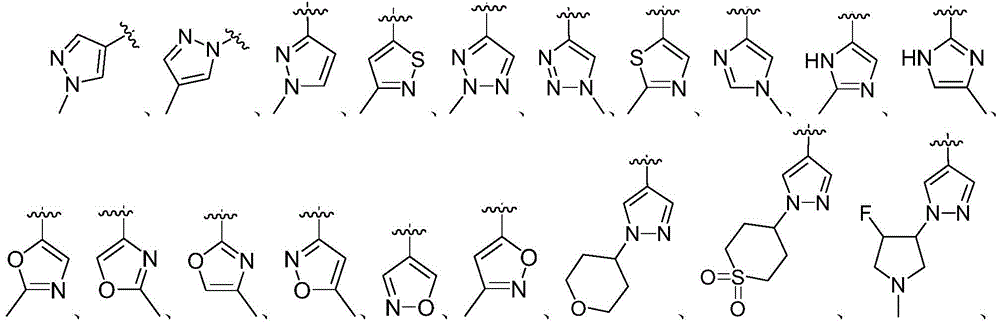
CN110256446B - Benzo-heterocycle substituted cyclopenta [4,5] pyrrolopyrazine-1-one derivatives and application thereof - Google Patents
Benzo-heterocycle substituted cyclopenta [4,5] pyrrolopyrazine-1-one derivatives and application thereof Download PDFInfo
- Publication number
- CN110256446B CN110256446B CN201910689812.4A CN201910689812A CN110256446B CN 110256446 B CN110256446 B CN 110256446B CN 201910689812 A CN201910689812 A CN 201910689812A CN 110256446 B CN110256446 B CN 110256446B
- Authority
- CN
- China
- Prior art keywords
- ring
- compound
- alkyl
- pharmaceutically acceptable
- acceptable salt
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 150000001875 compounds Chemical class 0.000 claims abstract description 94
- 150000003839 salts Chemical class 0.000 claims abstract description 21
- -1 C1-10Alkyl Chemical group 0.000 claims description 68
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 claims description 28
- 229910052736 halogen Inorganic materials 0.000 claims description 19
- 150000002367 halogens Chemical class 0.000 claims description 19
- 239000003814 drug Substances 0.000 claims description 17
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 17
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 16
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 claims description 14
- 125000000217 alkyl group Chemical group 0.000 claims description 14
- 201000010099 disease Diseases 0.000 claims description 14
- 239000001257 hydrogen Substances 0.000 claims description 14
- 229910052739 hydrogen Inorganic materials 0.000 claims description 14
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims description 13
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 claims description 12
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 claims description 12
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical compound C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 claims description 12
- 210000003719 b-lymphocyte Anatomy 0.000 claims description 11
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 claims description 10
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 9
- 229920006395 saturated elastomer Polymers 0.000 claims description 9
- DHXVGJBLRPWPCS-UHFFFAOYSA-N Tetrahydropyran Chemical compound C1CCOCC1 DHXVGJBLRPWPCS-UHFFFAOYSA-N 0.000 claims description 8
- 229940079593 drug Drugs 0.000 claims description 8
- RSEBUVRVKCANEP-UHFFFAOYSA-N 2-pyrroline Chemical compound C1CC=CN1 RSEBUVRVKCANEP-UHFFFAOYSA-N 0.000 claims description 7
- 230000001404 mediated effect Effects 0.000 claims description 7
- 125000002911 monocyclic heterocycle group Chemical group 0.000 claims description 7
- 239000008194 pharmaceutical composition Substances 0.000 claims description 7
- ARGCQEVBJHPOGB-UHFFFAOYSA-N 2,5-dihydrofuran Chemical compound C1OCC=C1 ARGCQEVBJHPOGB-UHFFFAOYSA-N 0.000 claims description 6
- VSWICNJIUPRZIK-UHFFFAOYSA-N 2-piperideine Chemical compound C1CNC=CC1 VSWICNJIUPRZIK-UHFFFAOYSA-N 0.000 claims description 6
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical group C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 claims description 6
- 229940043355 kinase inhibitor Drugs 0.000 claims description 6
- WCPAKWJPBJAGKN-UHFFFAOYSA-N oxadiazole Chemical group C1=CON=N1 WCPAKWJPBJAGKN-UHFFFAOYSA-N 0.000 claims description 6
- 239000003757 phosphotransferase inhibitor Substances 0.000 claims description 6
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 6
- 125000000714 pyrimidinyl group Chemical group 0.000 claims description 6
- FTAHXMZRJCZXDL-UHFFFAOYSA-N 3-piperideine Chemical compound C1CC=CCN1 FTAHXMZRJCZXDL-UHFFFAOYSA-N 0.000 claims description 5
- 125000000168 pyrrolyl group Chemical group 0.000 claims description 5
- UGUHFDPGDQDVGX-UHFFFAOYSA-N 1,2,3-thiadiazole Chemical group C1=CSN=N1 UGUHFDPGDQDVGX-UHFFFAOYSA-N 0.000 claims description 4
- WNXJIVFYUVYPPR-UHFFFAOYSA-N 1,3-dioxolane Chemical compound C1COCO1 WNXJIVFYUVYPPR-UHFFFAOYSA-N 0.000 claims description 4
- WYNCHZVNFNFDNH-UHFFFAOYSA-N Oxazolidine Chemical compound C1COCN1 WYNCHZVNFNFDNH-UHFFFAOYSA-N 0.000 claims description 4
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical group C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 claims description 4
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical group C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 claims description 4
- HONIICLYMWZJFZ-UHFFFAOYSA-N azetidine Chemical compound C1CNC1 HONIICLYMWZJFZ-UHFFFAOYSA-N 0.000 claims description 4
- 125000002883 imidazolyl group Chemical group 0.000 claims description 4
- 125000000842 isoxazolyl group Chemical group 0.000 claims description 4
- 125000002971 oxazolyl group Chemical group 0.000 claims description 4
- AHHWIHXENZJRFG-UHFFFAOYSA-N oxetane Chemical compound C1COC1 AHHWIHXENZJRFG-UHFFFAOYSA-N 0.000 claims description 4
- 125000003373 pyrazinyl group Chemical group 0.000 claims description 4
- PBMFSQRYOILNGV-UHFFFAOYSA-N pyridazine Chemical group C1=CC=NN=C1 PBMFSQRYOILNGV-UHFFFAOYSA-N 0.000 claims description 4
- 238000006467 substitution reaction Methods 0.000 claims description 4
- RAOIDOHSFRTOEL-UHFFFAOYSA-N tetrahydrothiophene Chemical compound C1CCSC1 RAOIDOHSFRTOEL-UHFFFAOYSA-N 0.000 claims description 4
- 125000003831 tetrazolyl group Chemical group 0.000 claims description 4
- MUGSKSNNEORSJG-UHFFFAOYSA-N 3174-74-1 Chemical compound C1CC=CCO1 MUGSKSNNEORSJG-UHFFFAOYSA-N 0.000 claims description 3
- 208000023275 Autoimmune disease Diseases 0.000 claims description 3
- BUDQDWGNQVEFAC-UHFFFAOYSA-N Dihydropyran Chemical compound C1COC=CC1 BUDQDWGNQVEFAC-UHFFFAOYSA-N 0.000 claims description 3
- 239000003937 drug carrier Substances 0.000 claims description 3
- 208000026935 allergic disease Diseases 0.000 claims description 2
- 208000027866 inflammatory disease Diseases 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims description 2
- 230000001613 neoplastic effect Effects 0.000 claims description 2
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 2
- 230000002062 proliferating effect Effects 0.000 claims description 2
- 125000001475 halogen functional group Chemical group 0.000 claims 3
- 125000004356 hydroxy functional group Chemical group O* 0.000 claims 3
- 150000002431 hydrogen Chemical class 0.000 claims 2
- 150000003053 piperidines Chemical class 0.000 claims 2
- 125000001425 triazolyl group Chemical group 0.000 claims 1
- 238000002360 preparation method Methods 0.000 abstract description 16
- 229940002612 prodrug Drugs 0.000 abstract description 7
- 239000000651 prodrug Substances 0.000 abstract description 7
- 239000012453 solvate Substances 0.000 abstract description 7
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 90
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 52
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 44
- 238000006243 chemical reaction Methods 0.000 description 40
- 239000000243 solution Substances 0.000 description 35
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 30
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 26
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 22
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 20
- 125000005843 halogen group Chemical group 0.000 description 17
- 239000012043 crude product Substances 0.000 description 15
- 239000003921 oil Substances 0.000 description 15
- 239000007787 solid Substances 0.000 description 15
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 14
- KZPYGQFFRCFCPP-UHFFFAOYSA-N 1,1'-bis(diphenylphosphino)ferrocene Chemical compound [Fe+2].C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1 KZPYGQFFRCFCPP-UHFFFAOYSA-N 0.000 description 13
- 238000005160 1H NMR spectroscopy Methods 0.000 description 13
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 13
- 230000002401 inhibitory effect Effects 0.000 description 13
- 238000000034 method Methods 0.000 description 12
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 12
- 125000001424 substituent group Chemical group 0.000 description 12
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 11
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 11
- 102100029823 Tyrosine-protein kinase BTK Human genes 0.000 description 10
- 229910052757 nitrogen Inorganic materials 0.000 description 10
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 10
- 238000010898 silica gel chromatography Methods 0.000 description 9
- 238000012360 testing method Methods 0.000 description 9
- 229940124291 BTK inhibitor Drugs 0.000 description 8
- KZBUYRJDOAKODT-UHFFFAOYSA-N Chlorine Chemical compound ClCl KZBUYRJDOAKODT-UHFFFAOYSA-N 0.000 description 8
- 238000004440 column chromatography Methods 0.000 description 8
- 125000000753 cycloalkyl group Chemical group 0.000 description 8
- 238000003818 flash chromatography Methods 0.000 description 8
- 239000000203 mixture Substances 0.000 description 8
- 238000000746 purification Methods 0.000 description 8
- 239000011541 reaction mixture Substances 0.000 description 8
- 125000006273 (C1-C3) alkyl group Chemical group 0.000 description 7
- 206010028980 Neoplasm Diseases 0.000 description 7
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 7
- 125000004432 carbon atom Chemical group C* 0.000 description 7
- 102000052116 epidermal growth factor receptor activity proteins Human genes 0.000 description 7
- 108700015053 epidermal growth factor receptor activity proteins Proteins 0.000 description 7
- 239000012467 final product Substances 0.000 description 7
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 7
- 239000003208 petroleum Substances 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 6
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 6
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 6
- 102000019260 B-Cell Antigen Receptors Human genes 0.000 description 6
- 108010012919 B-Cell Antigen Receptors Proteins 0.000 description 6
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 210000004027 cell Anatomy 0.000 description 6
- 238000001914 filtration Methods 0.000 description 6
- 230000005764 inhibitory process Effects 0.000 description 6
- 239000012044 organic layer Substances 0.000 description 6
- 230000026731 phosphorylation Effects 0.000 description 6
- 238000006366 phosphorylation reaction Methods 0.000 description 6
- 229910000027 potassium carbonate Inorganic materials 0.000 description 6
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 5
- 102000004190 Enzymes Human genes 0.000 description 5
- 108090000790 Enzymes Proteins 0.000 description 5
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 5
- 108091000080 Phosphotransferase Proteins 0.000 description 5
- 125000003545 alkoxy group Chemical group 0.000 description 5
- 239000000460 chlorine Substances 0.000 description 5
- 238000001816 cooling Methods 0.000 description 5
- 239000000706 filtrate Substances 0.000 description 5
- 239000011259 mixed solution Substances 0.000 description 5
- 102000020233 phosphotransferase Human genes 0.000 description 5
- 239000011535 reaction buffer Substances 0.000 description 5
- 238000003756 stirring Methods 0.000 description 5
- 239000000758 substrate Substances 0.000 description 5
- 125000006552 (C3-C8) cycloalkyl group Chemical group 0.000 description 4
- 125000005916 2-methylpentyl group Chemical group 0.000 description 4
- GAWIXWVDTYZWAW-UHFFFAOYSA-N C[CH]O Chemical group C[CH]O GAWIXWVDTYZWAW-UHFFFAOYSA-N 0.000 description 4
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 4
- 241000124008 Mammalia Species 0.000 description 4
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 4
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 4
- JRNVZBWKYDBUCA-UHFFFAOYSA-N N-chlorosuccinimide Chemical compound ClN1C(=O)CCC1=O JRNVZBWKYDBUCA-UHFFFAOYSA-N 0.000 description 4
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 4
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 4
- 239000003153 chemical reaction reagent Substances 0.000 description 4
- ZYGHJZDHTFUPRJ-UHFFFAOYSA-N coumarin Chemical compound C1=CC=C2OC(=O)C=CC2=C1 ZYGHJZDHTFUPRJ-UHFFFAOYSA-N 0.000 description 4
- 125000001072 heteroaryl group Chemical group 0.000 description 4
- NROKBHXJSPEDAR-UHFFFAOYSA-M potassium fluoride Chemical compound [F-].[K+] NROKBHXJSPEDAR-UHFFFAOYSA-M 0.000 description 4
- 125000006413 ring segment Chemical group 0.000 description 4
- DTQVDTLACAAQTR-UHFFFAOYSA-N trifluoroacetic acid Substances OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 4
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 4
- NDOVLWQBFFJETK-UHFFFAOYSA-N 1,4-thiazinane 1,1-dioxide Chemical compound O=S1(=O)CCNCC1 NDOVLWQBFFJETK-UHFFFAOYSA-N 0.000 description 3
- OZAIFHULBGXAKX-UHFFFAOYSA-N 2-(2-cyanopropan-2-yldiazenyl)-2-methylpropanenitrile Chemical compound N#CC(C)(C)N=NC(C)(C)C#N OZAIFHULBGXAKX-UHFFFAOYSA-N 0.000 description 3
- NYXQOIBMMREITL-UHFFFAOYSA-N 5-bromo-2-fluoro-3-iodobenzonitrile Chemical compound BrC=1C=C(C(=C(C#N)C=1)F)I NYXQOIBMMREITL-UHFFFAOYSA-N 0.000 description 3
- JVPYZLHZBATMEP-UHFFFAOYSA-N 5-bromo-7-iodo-1h-indazol-3-amine Chemical compound C1=C(Br)C=C2C(N)=NNC2=C1I JVPYZLHZBATMEP-UHFFFAOYSA-N 0.000 description 3
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 3
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- 208000010839 B-cell chronic lymphocytic leukemia Diseases 0.000 description 3
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 3
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 3
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 3
- 208000031422 Lymphocytic Chronic B-Cell Leukemia Diseases 0.000 description 3
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 229910052786 argon Inorganic materials 0.000 description 3
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 3
- 229910052794 bromium Inorganic materials 0.000 description 3
- 229910052801 chlorine Inorganic materials 0.000 description 3
- 125000004093 cyano group Chemical group *C#N 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- 238000007865 diluting Methods 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 230000005284 excitation Effects 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 239000011737 fluorine Substances 0.000 description 3
- 229910052731 fluorine Inorganic materials 0.000 description 3
- 238000004128 high performance liquid chromatography Methods 0.000 description 3
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 3
- 239000011630 iodine Substances 0.000 description 3
- 229910052740 iodine Inorganic materials 0.000 description 3
- 230000031700 light absorption Effects 0.000 description 3
- PIBWKRNGBLPSSY-UHFFFAOYSA-L palladium(II) chloride Chemical compound Cl[Pd]Cl PIBWKRNGBLPSSY-UHFFFAOYSA-L 0.000 description 3
- 125000003538 pentan-3-yl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])[H] 0.000 description 3
- 239000001632 sodium acetate Substances 0.000 description 3
- 235000017281 sodium acetate Nutrition 0.000 description 3
- 208000024891 symptom Diseases 0.000 description 3
- 150000003852 triazoles Chemical group 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- NWZSZGALRFJKBT-KNIFDHDWSA-N (2s)-2,6-diaminohexanoic acid;(2s)-2-hydroxybutanedioic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O.NCCCC[C@H](N)C(O)=O NWZSZGALRFJKBT-KNIFDHDWSA-N 0.000 description 2
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 description 2
- 125000001399 1,2,3-triazolyl group Chemical group N1N=NC(=C1)* 0.000 description 2
- BBVIDBNAYOIXOE-UHFFFAOYSA-N 1,2,4-oxadiazole Chemical group C=1N=CON=1 BBVIDBNAYOIXOE-UHFFFAOYSA-N 0.000 description 2
- 125000001376 1,2,4-triazolyl group Chemical group N1N=C(N=C1)* 0.000 description 2
- FKASFBLJDCHBNZ-UHFFFAOYSA-N 1,3,4-oxadiazole Chemical group C1=NN=CO1 FKASFBLJDCHBNZ-UHFFFAOYSA-N 0.000 description 2
- UCNGGGYMLHAMJG-UHFFFAOYSA-N 1-methyl-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyrazole Chemical compound C1=NN(C)C=C1B1OC(C)(C)C(C)(C)O1 UCNGGGYMLHAMJG-UHFFFAOYSA-N 0.000 description 2
- QWENRTYMTSOGBR-UHFFFAOYSA-N 1H-1,2,3-Triazole Chemical group C=1C=NNN=1 QWENRTYMTSOGBR-UHFFFAOYSA-N 0.000 description 2
- 125000003660 2,3-dimethylpentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(C([H])([H])[H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- LFASPBCAVCUZES-UHFFFAOYSA-N 2h-pyrrolo[1,2-a]pyrazin-1-one Chemical compound O=C1NC=CN2C=CC=C12 LFASPBCAVCUZES-UHFFFAOYSA-N 0.000 description 2
- 125000003469 3-methylhexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 2
- DXGAGMFBYXLUJG-UHFFFAOYSA-N 4-bromo-2-ethyl-6-iodoaniline Chemical compound CCc1cc(Br)cc(I)c1N DXGAGMFBYXLUJG-UHFFFAOYSA-N 0.000 description 2
- 229960000549 4-dimethylaminophenol Drugs 0.000 description 2
- NSPMIYGKQJPBQR-UHFFFAOYSA-N 4H-1,2,4-triazole Chemical group C=1N=CNN=1 NSPMIYGKQJPBQR-UHFFFAOYSA-N 0.000 description 2
- 208000003950 B-cell lymphoma Diseases 0.000 description 2
- 241000282421 Canidae Species 0.000 description 2
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 2
- 206010025323 Lymphomas Diseases 0.000 description 2
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 2
- 229910002666 PdCl2 Inorganic materials 0.000 description 2
- 108010009978 Tec protein-tyrosine kinase Proteins 0.000 description 2
- RRUDCFGSUDOHDG-UHFFFAOYSA-N acetohydroxamic acid Chemical compound CC(O)=NO RRUDCFGSUDOHDG-UHFFFAOYSA-N 0.000 description 2
- 239000013543 active substance Substances 0.000 description 2
- 239000012491 analyte Substances 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 206010003246 arthritis Diseases 0.000 description 2
- 125000003118 aryl group Chemical group 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 125000005605 benzo group Chemical group 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- 230000004663 cell proliferation Effects 0.000 description 2
- 208000032852 chronic lymphocytic leukemia Diseases 0.000 description 2
- 229960000956 coumarin Drugs 0.000 description 2
- 235000001671 coumarin Nutrition 0.000 description 2
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 2
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 239000013024 dilution buffer Substances 0.000 description 2
- 208000035475 disorder Diseases 0.000 description 2
- GNBHRKFJIUUOQI-UHFFFAOYSA-N fluorescein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 GNBHRKFJIUUOQI-UHFFFAOYSA-N 0.000 description 2
- JKFAIQOWCVVSKC-UHFFFAOYSA-N furazan Chemical group C=1C=NON=1 JKFAIQOWCVVSKC-UHFFFAOYSA-N 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 125000005842 heteroatom Chemical group 0.000 description 2
- IKDUDTNKRLTJSI-UHFFFAOYSA-N hydrazine monohydrate Substances O.NN IKDUDTNKRLTJSI-UHFFFAOYSA-N 0.000 description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- 125000004499 isoxazol-5-yl group Chemical group O1N=CC=C1* 0.000 description 2
- 239000007791 liquid phase Substances 0.000 description 2
- 239000012280 lithium aluminium hydride Substances 0.000 description 2
- YNESATAKKCNGOF-UHFFFAOYSA-N lithium bis(trimethylsilyl)amide Chemical compound [Li+].C[Si](C)(C)[N-][Si](C)(C)C YNESATAKKCNGOF-UHFFFAOYSA-N 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 239000012046 mixed solvent Substances 0.000 description 2
- 125000002950 monocyclic group Chemical group 0.000 description 2
- 239000012074 organic phase Substances 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- IVDFJHOHABJVEH-UHFFFAOYSA-N pinacol Chemical compound CC(C)(O)C(C)(C)O IVDFJHOHABJVEH-UHFFFAOYSA-N 0.000 description 2
- SCVFZCLFOSHCOH-UHFFFAOYSA-M potassium acetate Chemical compound [K+].CC([O-])=O SCVFZCLFOSHCOH-UHFFFAOYSA-M 0.000 description 2
- 239000011698 potassium fluoride Substances 0.000 description 2
- 235000003270 potassium fluoride Nutrition 0.000 description 2
- 229910000160 potassium phosphate Inorganic materials 0.000 description 2
- 235000011009 potassium phosphates Nutrition 0.000 description 2
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- 230000035755 proliferation Effects 0.000 description 2
- 150000003254 radicals Chemical class 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 230000019491 signal transduction Effects 0.000 description 2
- 239000012279 sodium borohydride Substances 0.000 description 2
- 229910000033 sodium borohydride Inorganic materials 0.000 description 2
- 229910000029 sodium carbonate Inorganic materials 0.000 description 2
- LPXPTNMVRIOKMN-UHFFFAOYSA-M sodium nitrite Chemical compound [Na+].[O-]N=O LPXPTNMVRIOKMN-UHFFFAOYSA-M 0.000 description 2
- MFRIHAYPQRLWNB-UHFFFAOYSA-N sodium tert-butoxide Chemical compound [Na+].CC(C)(C)[O-] MFRIHAYPQRLWNB-UHFFFAOYSA-N 0.000 description 2
- BCNZYOJHNLTNEZ-UHFFFAOYSA-N tert-butyldimethylsilyl chloride Chemical compound CC(C)(C)[Si](C)(C)Cl BCNZYOJHNLTNEZ-UHFFFAOYSA-N 0.000 description 2
- 238000002877 time resolved fluorescence resonance energy transfer Methods 0.000 description 2
- CXNIUSPIQKWYAI-UHFFFAOYSA-N xantphos Chemical compound C=12OC3=C(P(C=4C=CC=CC=4)C=4C=CC=CC=4)C=CC=C3C(C)(C)C2=CC=CC=1P(C=1C=CC=CC=1)C1=CC=CC=C1 CXNIUSPIQKWYAI-UHFFFAOYSA-N 0.000 description 2
- CYPYTURSJDMMMP-WVCUSYJESA-N (1e,4e)-1,5-diphenylpenta-1,4-dien-3-one;palladium Chemical compound [Pd].[Pd].C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1 CYPYTURSJDMMMP-WVCUSYJESA-N 0.000 description 1
- 125000005918 1,2-dimethylbutyl group Chemical group 0.000 description 1
- RHFWLPWDOYJEAL-UHFFFAOYSA-N 1,2-oxazol-3-amine Chemical compound NC=1C=CON=1 RHFWLPWDOYJEAL-UHFFFAOYSA-N 0.000 description 1
- VCDWKCPGKQANIF-UHFFFAOYSA-N 1-(6-bromopyridin-3-yl)-4-methylpiperazine Chemical compound C1CN(C)CCN1C1=CC=C(Br)N=C1 VCDWKCPGKQANIF-UHFFFAOYSA-N 0.000 description 1
- LMDZBCPBFSXMTL-UHFFFAOYSA-N 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide Substances CCN=C=NCCCN(C)C LMDZBCPBFSXMTL-UHFFFAOYSA-N 0.000 description 1
- PVOAHINGSUIXLS-UHFFFAOYSA-N 1-Methylpiperazine Chemical compound CN1CCNCC1 PVOAHINGSUIXLS-UHFFFAOYSA-N 0.000 description 1
- SQMVRFXDBRYXFQ-UHFFFAOYSA-N 1-methyl-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-3,6-dihydro-2h-pyridine Chemical compound C1N(C)CCC(B2OC(C)(C)C(C)(C)O2)=C1 SQMVRFXDBRYXFQ-UHFFFAOYSA-N 0.000 description 1
- 125000003764 2,4-dimethylpentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- LLKRSJVPTKFSLS-UHFFFAOYSA-N 2-bromo-5-iodopyridine Chemical compound BrC1=CC=C(I)C=N1 LLKRSJVPTKFSLS-UHFFFAOYSA-N 0.000 description 1
- 125000001731 2-cyanoethyl group Chemical group [H]C([H])(*)C([H])([H])C#N 0.000 description 1
- 125000006176 2-ethylbutyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(C([H])([H])*)C([H])([H])C([H])([H])[H] 0.000 description 1
- YEDUAINPPJYDJZ-UHFFFAOYSA-N 2-hydroxybenzothiazole Chemical compound C1=CC=C2SC(O)=NC2=C1 YEDUAINPPJYDJZ-UHFFFAOYSA-N 0.000 description 1
- 125000004493 2-methylbut-1-yl group Chemical group CC(C*)CC 0.000 description 1
- 125000003229 2-methylhexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- FPQQSJJWHUJYPU-UHFFFAOYSA-N 3-(dimethylamino)propyliminomethylidene-ethylazanium;chloride Chemical compound Cl.CCN=C=NCCCN(C)C FPQQSJJWHUJYPU-UHFFFAOYSA-N 0.000 description 1
- 125000004337 3-ethylpentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(C([H])([H])C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003542 3-methylbutan-2-yl group Chemical group [H]C([H])([H])C([H])(*)C([H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000005917 3-methylpentyl group Chemical group 0.000 description 1
- LGOZNQPHTIGMQJ-UHFFFAOYSA-N 4-bromo-2-ethylaniline Chemical compound CCC1=CC(Br)=CC=C1N LGOZNQPHTIGMQJ-UHFFFAOYSA-N 0.000 description 1
- PEXAZYDITWXYNJ-UHFFFAOYSA-N 5-bromo-2-fluorobenzoic acid Chemical compound OC(=O)C1=CC(Br)=CC=C1F PEXAZYDITWXYNJ-UHFFFAOYSA-N 0.000 description 1
- OZAIFHULBGXAKX-VAWYXSNFSA-N AIBN Substances N#CC(C)(C)\N=N\C(C)(C)C#N OZAIFHULBGXAKX-VAWYXSNFSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 208000024893 Acute lymphoblastic leukemia Diseases 0.000 description 1
- 208000014697 Acute lymphocytic leukaemia Diseases 0.000 description 1
- 208000031261 Acute myeloid leukaemia Diseases 0.000 description 1
- 108010029445 Agammaglobulinaemia Tyrosine Kinase Proteins 0.000 description 1
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical class [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 1
- 206010002556 Ankylosing Spondylitis Diseases 0.000 description 1
- 208000032791 BCR-ABL1 positive chronic myelogenous leukemia Diseases 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 241000282693 Cercopithecidae Species 0.000 description 1
- 241000282994 Cervidae Species 0.000 description 1
- 208000010833 Chronic myeloid leukaemia Diseases 0.000 description 1
- 206010061818 Disease progression Diseases 0.000 description 1
- 208000009386 Experimental Arthritis Diseases 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 201000005569 Gout Diseases 0.000 description 1
- 206010018634 Gouty Arthritis Diseases 0.000 description 1
- 239000007821 HATU Substances 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 102000003777 Interleukin-1 beta Human genes 0.000 description 1
- 108090000193 Interleukin-1 beta Proteins 0.000 description 1
- 108090001005 Interleukin-6 Proteins 0.000 description 1
- 229910010084 LiAlH4 Inorganic materials 0.000 description 1
- 206010052178 Lymphocytic lymphoma Diseases 0.000 description 1
- 208000025205 Mantle-Cell Lymphoma Diseases 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 208000034578 Multiple myelomas Diseases 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 208000033761 Myelogenous Chronic BCR-ABL Positive Leukemia Diseases 0.000 description 1
- 208000033776 Myeloid Acute Leukemia Diseases 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 101150003085 Pdcl gene Proteins 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 208000006664 Precursor Cell Lymphoblastic Leukemia-Lymphoma Diseases 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- 201000004681 Psoriasis Diseases 0.000 description 1
- 201000001263 Psoriatic Arthritis Diseases 0.000 description 1
- 208000036824 Psoriatic arthropathy Diseases 0.000 description 1
- 206010039491 Sarcoma Diseases 0.000 description 1
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical class [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 1
- 208000021712 Soft tissue sarcoma Diseases 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- GCTFWCDSFPMHHS-UHFFFAOYSA-M Tributyltin chloride Chemical compound CCCC[Sn](Cl)(CCCC)CCCC GCTFWCDSFPMHHS-UHFFFAOYSA-M 0.000 description 1
- 241000282458 Ursus sp. Species 0.000 description 1
- QPFDUULIDNELSE-UHFFFAOYSA-N [4-(morpholin-4-ylmethyl)phenyl]boronic acid Chemical compound C1=CC(B(O)O)=CC=C1CN1CCOCC1 QPFDUULIDNELSE-UHFFFAOYSA-N 0.000 description 1
- 229960001171 acetohydroxamic acid Drugs 0.000 description 1
- WETWJCDKMRHUPV-UHFFFAOYSA-N acetyl chloride Chemical compound CC(Cl)=O WETWJCDKMRHUPV-UHFFFAOYSA-N 0.000 description 1
- 239000012346 acetyl chloride Substances 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 239000012300 argon atmosphere Substances 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- 230000035578 autophosphorylation Effects 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- HTZCNXWZYVXIMZ-UHFFFAOYSA-M benzyl(triethyl)azanium;chloride Chemical compound [Cl-].CC[N+](CC)(CC)CC1=CC=CC=C1 HTZCNXWZYVXIMZ-UHFFFAOYSA-M 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- MUALRAIOVNYAIW-UHFFFAOYSA-N binap Chemical compound C1=CC=CC=C1P(C=1C(=C2C=CC=CC2=CC=1)C=1C2=CC=CC=C2C=CC=1P(C=1C=CC=CC=1)C=1C=CC=CC=1)C1=CC=CC=C1 MUALRAIOVNYAIW-UHFFFAOYSA-N 0.000 description 1
- 238000004166 bioassay Methods 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- ZADPBFCGQRWHPN-UHFFFAOYSA-N boronic acid Chemical compound OBO ZADPBFCGQRWHPN-UHFFFAOYSA-N 0.000 description 1
- 239000012267 brine Substances 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 239000013592 cell lysate Substances 0.000 description 1
- 230000011748 cell maturation Effects 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000012230 colorless oil Substances 0.000 description 1
- 230000021615 conjugation Effects 0.000 description 1
- 125000000000 cycloalkoxy group Chemical group 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000002188 cycloheptatrienyl group Chemical group C1(=CC=CC=CC1)* 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000003678 cyclohexadienyl group Chemical group C1(=CC=CCC1)* 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 description 1
- 231100000135 cytotoxicity Toxicity 0.000 description 1
- 230000003013 cytotoxicity Effects 0.000 description 1
- 230000006837 decompression Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 125000004772 dichloromethyl group Chemical group [H]C(Cl)(Cl)* 0.000 description 1
- MXFYYFVVIIWKFE-UHFFFAOYSA-N dicyclohexyl-[2-[2,6-di(propan-2-yloxy)phenyl]phenyl]phosphane Chemical compound CC(C)OC1=CC=CC(OC(C)C)=C1C1=CC=CC=C1P(C1CCCCC1)C1CCCCC1 MXFYYFVVIIWKFE-UHFFFAOYSA-N 0.000 description 1
- 125000006001 difluoroethyl group Chemical group 0.000 description 1
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 230000005750 disease progression Effects 0.000 description 1
- 238000009509 drug development Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- PQVSTLUFSYVLTO-UHFFFAOYSA-N ethyl n-ethoxycarbonylcarbamate Chemical compound CCOC(=O)NC(=O)OCC PQVSTLUFSYVLTO-UHFFFAOYSA-N 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000012065 filter cake Substances 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 125000003784 fluoroethyl group Chemical group [H]C([H])(F)C([H])([H])* 0.000 description 1
- 125000004216 fluoromethyl group Chemical group [H]C([H])(F)* 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 239000001307 helium Substances 0.000 description 1
- 229910052734 helium Inorganic materials 0.000 description 1
- SWQJXJOGLNCZEY-UHFFFAOYSA-N helium atom Chemical compound [He] SWQJXJOGLNCZEY-UHFFFAOYSA-N 0.000 description 1
- 210000003958 hematopoietic stem cell Anatomy 0.000 description 1
- 238000002868 homogeneous time resolved fluorescence Methods 0.000 description 1
- NPZTUJOABDZTLV-UHFFFAOYSA-N hydroxybenzotriazole Substances O=C1C=CC=C2NNN=C12 NPZTUJOABDZTLV-UHFFFAOYSA-N 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 208000018937 joint inflammation Diseases 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 1
- GLXDVVHUTZTUQK-UHFFFAOYSA-M lithium hydroxide monohydrate Substances [Li+].O.[OH-] GLXDVVHUTZTUQK-UHFFFAOYSA-M 0.000 description 1
- 229940040692 lithium hydroxide monohydrate Drugs 0.000 description 1
- 239000012139 lysis buffer Substances 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 230000036210 malignancy Effects 0.000 description 1
- 201000001441 melanoma Diseases 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 210000001616 monocyte Anatomy 0.000 description 1
- YOHYSYJDKVYCJI-UHFFFAOYSA-N n-[3-[[6-[3-(trifluoromethyl)anilino]pyrimidin-4-yl]amino]phenyl]cyclopropanecarboxamide Chemical compound FC(F)(F)C1=CC=CC(NC=2N=CN=C(NC=3C=C(NC(=O)C4CC4)C=CC=3)C=2)=C1 YOHYSYJDKVYCJI-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 239000013642 negative control Substances 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- 239000012299 nitrogen atmosphere Substances 0.000 description 1
- 102000037979 non-receptor tyrosine kinases Human genes 0.000 description 1
- 108091008046 non-receptor tyrosine kinases Proteins 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 201000008482 osteoarthritis Diseases 0.000 description 1
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 1
- 125000003367 polycyclic group Chemical group 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 235000011056 potassium acetate Nutrition 0.000 description 1
- 238000004262 preparative liquid chromatography Methods 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 230000004952 protein activity Effects 0.000 description 1
- ZVJHJDDKYZXRJI-UHFFFAOYSA-N pyrroline Natural products C1CC=NC1 ZVJHJDDKYZXRJI-UHFFFAOYSA-N 0.000 description 1
- 238000010791 quenching Methods 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 238000009738 saturating Methods 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- YPNVIBVEFVRZPJ-UHFFFAOYSA-L silver sulfate Chemical compound [Ag+].[Ag+].[O-]S([O-])(=O)=O YPNVIBVEFVRZPJ-UHFFFAOYSA-L 0.000 description 1
- 229910000367 silver sulfate Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 235000010288 sodium nitrite Nutrition 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N sulfuric acid Substances OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- 201000000596 systemic lupus erythematosus Diseases 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- WROMPOXWARCANT-UHFFFAOYSA-N tfa trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F.OC(=O)C(F)(F)F WROMPOXWARCANT-UHFFFAOYSA-N 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 150000003573 thiols Chemical class 0.000 description 1
- 125000006000 trichloroethyl group Chemical group 0.000 description 1
- 125000003866 trichloromethyl group Chemical group ClC(Cl)(Cl)* 0.000 description 1
- 125000004205 trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 1
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 1
- FTVLMFQEYACZNP-UHFFFAOYSA-N trimethylsilyl trifluoromethanesulfonate Chemical compound C[Si](C)(C)OS(=O)(=O)C(F)(F)F FTVLMFQEYACZNP-UHFFFAOYSA-N 0.000 description 1
- KPZYAGQLBFUTMA-UHFFFAOYSA-K tripotassium;phosphate;trihydrate Chemical compound O.O.O.[K+].[K+].[K+].[O-]P([O-])([O-])=O KPZYAGQLBFUTMA-UHFFFAOYSA-K 0.000 description 1
- UGOMMVLRQDMAQQ-UHFFFAOYSA-N xphos Chemical compound CC(C)C1=CC(C(C)C)=CC(C(C)C)=C1C1=CC=CC=C1P(C1CCCCC1)C1CCCCC1 UGOMMVLRQDMAQQ-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Rheumatology (AREA)
- Physical Education & Sports Medicine (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Dermatology (AREA)
- Pain & Pain Management (AREA)
- Hematology (AREA)
- Oncology (AREA)
- Immunology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The invention relates to benzoheterocyclically substituted cyclopenta [4,5]]Pyrrolopyrazin-1-one derivatives, preparation methods and medical uses thereof. Specifically, the invention discloses a compound of formula (I) or a pharmaceutically acceptable salt, a stereoisomer, a solvate or a prodrug thereof, and a preparation method and application thereof, wherein the definition of each group in the formula is shown in the specification and the claims.
Description
Technical Field
The invention belongs to the technical field of medicines. In particular, the invention relates to benzo-heterocycle substituted cyclopenta [4,5] pyrrolopyrazine-1-one derivatives, a preparation method thereof, application of the derivatives as BTK inhibitors, and a pharmaceutical composition prepared from the derivatives.
Background
The BTK kinase is a non-receptor tyrosine kinase in the TEC kinase family, is a key regulator of a BCR signal pathway, and has important functions on B cell maturation, proliferation and survival. BTK is over-expressed in various B cell lymphomas, and is the only clinically validated target point for effective drug development in the current TEC kinase family. Inhibition of BTK can inhibit the proliferation of a range of B cell lymphomas.
Activation of the B cell antigen receptor (BCR) signaling pathway is important for the induction and maintenance of B cell malignancies and autoimmune diseases. Bruton's tyrosine kinase (Btk) plays a key role in the BCR signal channel of hematopoietic cells and is a very good target point in the research of new therapies for lymphoma. The BTK inhibitor acts on a BCR passage to inhibit the autophosphorylation of Btk, the phosphorylation of physiological substrates PLC gamma of Btk's and the phosphorylation of downstream kinases ERK.
The BTK inhibitor acts on Chronic Lymphocytic Leukemia (CLL) cells, induces cytotoxicity, and inhibits CLL cell proliferation ability. Inhibit proliferation of BCR-activated primary B cells, and inhibit secretion of TNF alpha, IL-1 beta, IL-6, etc. in primary monocytes. The BTK inhibitor acts on a collagen-induced arthritis model, and remarkably reduces clinical arthritis symptoms such as foot swelling and joint inflammation by inhibiting B cell activity.
Only the single BTK inhibitor irrutinib is currently approved for the market, so that there is a need to develop more active, safer and more effective BTK inhibitors.
Disclosure of Invention
The invention aims to provide a compound which has a novel structure and can be used as a Btk inhibitor.
The invention provides a compound shown as a formula (I) in a first aspect, or a pharmaceutically acceptable salt, a stereoisomer, a solvate or a prodrug thereof:
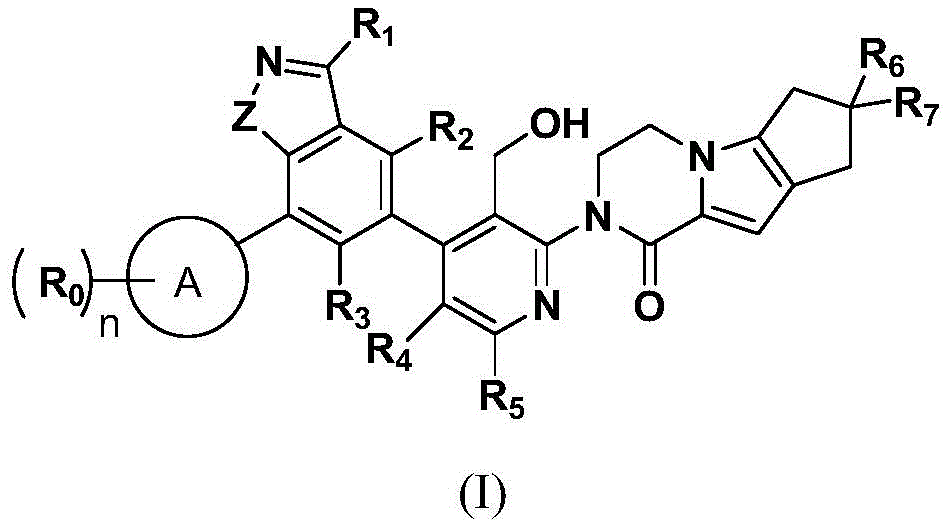
in the formula (I), the compound is shown in the specification,
ring A is C6-10An aryl ring (preferably phenyl), a 5-to 6-membered monocyclic heteroaryl ring or a 4-to 6-membered unsaturated monocyclic heterocycle;
R0is hydrogen, halogen (preferably fluorine, chlorine, bromine), C1-8Alkyl (preferably C)1-6Alkyl, more preferably C1-3Alkyl), halo C1-8Alkyl (preferably halogenated C)1-6Alkyl, more preferably halogenated C1-3Alkyl group), C1-8Alkoxy (preferably C)1-6Alkoxy, more preferably C1-3Alkoxy), halo C1-8Alkoxy (preferably halo C)1-6Alkoxy, more preferably halo C1-3Alkoxy), optionally substituted C3-8Cycloalkyl (preferably C)3-6Cycloalkyl), C3-8Cycloalkoxy (preferably C)3-6Cycloalkoxy), 5-to 6-membered monocyclic heteroaryl, or-Y-L; wherein Y is a bond, - (CH)2)m-、-O-(CH2)m-or c (o); l is hydroxy, cyano-substituted C1-3Alkyl, optionally substituted C6-10Aryl, optionally substituted 5-to 6-membered monocyclic heteroaryl, optionally substituted 4-to 6-membered saturated monocyclic heterocycle, C1-8Alkoxy (preferably C)1-6Alkoxy, more preferably C1-3Alkoxy) or NRa0Rb0(ii) a Said optionally substituted being unsubstituted or substituted by 1,2 or 3 hydroxy, halogen, C1-10Alkyl (preferably C)1-6Alkyl, more preferably C1-3Alkyl), hydroxy-substituted C1-10Alkyl (preferably hydroxy-substituted C)1-6Alkyl, more preferably hydroxy-substituted C1-3Alkyl), halo C1-8Alkyl (preferably halogenated C)1-6Alkyl, more preferably halogenated C1-3Alkyl, most preferably fluoro C1-3Alkyl), 4 to 6 membered saturated mono-heterocyclic ring;
n, m are each independently 1,2 or 3; it is to be noted that n and m refer to the number of substituents on ring A, and it is understood that when R is0By hydrogen is meant that ring a may not contain substituents, in which case the number of hydrogen atoms does not apply to the definition of n and m, for example: when ring A is a benzene ring, R0Hydrogen is understood to mean 5 hydrogens on the phenyl ring.
Z is O or NRa;
R1Is halogen, C1-10Alkyl (preferably C)1-6Alkyl, more preferably C1-3Alkyl) or NRa0Rb0;
Ra、Ra0、Rb0Each independently is hydrogen or C1-8Alkyl (preferably C)1-6Alkyl, more preferably C1-3Alkyl groups);
R2、R3、R4、R5each independently hydrogen, halogen (preferably fluorine, chlorine, bromine), C1-8Alkyl (preferably C)1-6Alkyl, more preferably C1-3Alkyl), halo C1-8Alkyl (preferably halogenated C)1-6Alkyl, more preferably halogenated C1-3Alkyl group), C1-8Alkoxy (preferably C)1-6Alkoxy, more preferably C1-3Alkoxy) or C3-8Cycloalkyl (preferably C)3-6Cycloalkyl groups);
R6、R7each independently is hydrogen or C1-8Alkyl (preferably C)1-6Alkyl, more preferably C1-3Alkyl groups).
In another preferred embodiment, in formula (I), the 4 to 6 membered saturated mono-heterocyclic ring is selected from: azetidine, oxetane, tetrahydrofuran, tetrahydrothiophene, tetrahydropyrrole, piperidine, oxazolidine, piperazine, dioxolane, dioxane, morpholine, thiomorpholine-1, 1-dioxide, or tetrahydropyran.
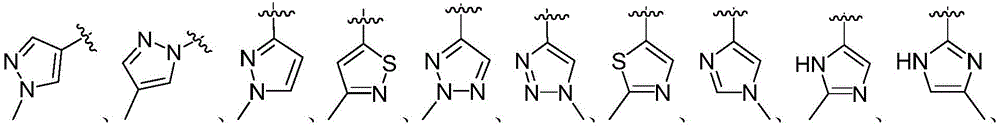
In another preferred embodiment, in formula (I), the 5-to 6-membered monocyclic heteroaryl ring is selected from: thiophene ring, N-alkylcyclopyrrole ring, furan ring, thiazole ring, imidazole ring, oxazole ring, pyrrole ring, pyrazole ring, triazole ring, 1,2, 3-triazole ring, 1,2, 4-triazole ring, 1,2, 5-triazole ring, 1,3, 4-triazole ring, tetrazole ring, isoxazole ring, oxadiazole ring, 1,2, 3-oxadiazole ring, 1,2, 4-oxadiazole ring, 1,2, 5-oxadiazole ring, 1,3, 4-oxadiazole ring, thiadiazole ring, pyridine ring, pyridazine ring, pyrimidine ring, pyrazine ring.
In another preferred embodiment, in formula (I), the 4 to 6 membered unsaturated mono-heterocyclic ring is selected from: 2, 3-dihydro-1H-pyrrole, 2, 5-dihydrofuran, 1,2,3, 6-tetrahydropyridine, 1,2,3, 4-tetrahydropyridine, 3, 6-dihydro-2H-pyran or 3, 4-dihydro-2H-pyran.
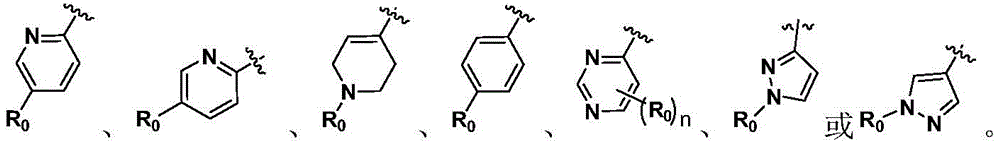
In another preferred embodiment, in formula (I), the A ring is a benzene ring, a pyrazole ring, a pyrimidine ring, a pyridine ring or a 1,2,3, 6-tetrahydropyridine ring.
In another preferred embodiment, in formula (I), n is 1 or 2.
In another preferred embodiment, in formula (I), R0Is hydrogen, C1-8Alkyl (preferably C)1-6Alkyl, more preferably C1-3Alkyl) or-Y-L; wherein Y is a bond or- (CH)2)m-; l is an optionally substituted 4-to 6-membered saturated mono-heterocyclic ring; said optionally substituted is unsubstituted or substituted by 1,2 or 3C1-10Alkyl (preferably C)1-6Alkyl, more preferably C1-3Alkyl) substituted; m is 1.
In another preferred embodiment, in formula (I), the structureIs a structure selected from the group consisting of:
in another preferred embodiment, the 5-to 6-membered monocyclic heteroaryl ring is selected from:
the 5-to 6-membered monocyclic heteroaryl ring is optionally substituted with 1 or 2R0And (4) substitution.
in another preferred embodiment, in formula (I), the compound of formula (I) is a compound selected from the group consisting of:
in a second aspect, the present invention provides a pharmaceutical composition comprising a compound of the first aspect of the present invention, or a pharmaceutically acceptable salt, solvate, stereoisomer or prodrug thereof; and a pharmaceutically acceptable carrier or excipient.
In a third aspect, the present invention provides a compound according to the first aspect, or a pharmaceutically acceptable salt, solvate, stereoisomer or prodrug thereof, or a pharmaceutical composition according to the second aspect, for use in the preparation of a kinase inhibitor medicament.
In another preferred embodiment, the kinase inhibitor is a BTK inhibitor.
In a fourth aspect, the present invention provides the use of a compound according to the first aspect of the present invention, or a pharmaceutically acceptable salt, solvate, stereoisomer or prodrug thereof, or a pharmaceutical composition according to the second aspect of the present invention, in the manufacture of a medicament for the treatment of a B-cell mediated disease.
In a fifth aspect, the present invention provides a method of treating a B-cell mediated disease, comprising administering to a patient in need thereof a therapeutically effective amount of a compound according to the first aspect of the present invention, or a pharmaceutically acceptable salt, solvate, stereoisomer or prodrug thereof, or a pharmaceutical composition according to the second aspect of the present invention.
In a sixth aspect, the present invention provides a method of treating a disease mediated by B cells, comprising administering to a patient in need thereof a therapeutically effective amount of a compound according to the first aspect of the present invention, or a pharmaceutically acceptable salt, solvate, stereoisomer or prodrug thereof, and another therapeutically active agent.
In another preferred embodiment, the disease mediated by B cells is selected from the group consisting of: a neoplastic disease, a proliferative disease, an allergic disease, an autoimmune disease, or an inflammatory disease.
In another preferred embodiment, the disease mediated by B cells is selected from the group consisting of: solid tumors, acute lymphocytic leukemia, chronic lymphocytic leukemia, acute myelogenous leukemia, chronic myelogenous leukemia, rheumatoid arthritis, psoriatic arthritis, osteoarthritis, systemic lupus erythematosus, psoriasis, rheumatoid spondylitis, and gouty arthritis.
In another preferred embodiment, the disease mediated by B cells is a solid tumor.
In another preferred embodiment, the solid tumor is at least one selected from the group consisting of lymphoma, soft tissue sarcoma, lymphocytic lymphoma, mantle cell lymphoma, melanoma, and multiple myeloma.
It is to be understood that within the scope of the present invention, the above-described features of the present invention and those specifically described below (e.g., in the examples) may be combined with each other to form new or preferred embodiments. Not to be reiterated herein, but to the extent of space.
Detailed Description
The inventors of the present invention have conducted extensive and intensive studies and have unexpectedly found that such benzoheterocycle-substituted cyclopenta [4,5] pyrrolopyrazine-1-one derivatives have high inhibitory activities against enzymes such as BTK WT and cells such as pBTKY 223. Therefore, the series of compounds are expected to be developed into medicaments for treating tumors. On this basis, the inventors have completed the present invention.
Definition of terms
As used herein, "alkyl" refers to straight and branched chain saturated aliphatic hydrocarbon groups, C1-8The alkyl group is an alkyl group containing 1 to 8 carbon atoms, and may preferably be C1-6Alkyl or C1-3Alkyl, defined similarly; non-limiting examples of alkyl groups include: methyl group, ethyl group, n-propyl group, isopropyl group, n-butyl group, isobutyl group, tert-butyl group, sec-butyl group, n-pentyl group, 1-dimethylpropyl group, 1, 2-dimethylpropyl group, 2-dimethylpropyl group, 1-ethylpropyl group, 2-methylbutyl group, 3-methylbutyl group, n-hexyl group, 1-ethyl-2-methylpropyl group, 1, 2-trimethylpropyl group, 1-dimethylbutyl group, 1, 2-dimethylbutyl group, 2-dimethylbutyl group, 1, 3-dimethylbutyl group, 2-ethylbutyl group, 2-methylpentyl group, 3-methylpentyl group, 4-methylpentyl group, 2, 3-dimethylbutyl group, n-heptyl group, 2-methylhexyl group, 3-methylhexyl group, 4-methylhexyl group, 2-methylpentyl group, 3-methylhexyl group, 4-methylhexyl group, 2-dimethylpropyl group, 2-pentyl group, 2-methylpropyl group, 2-methyl-pentyl group, 3-pentyl group, 2-methyl-pentyl group, 2-pentyl group, and 3-pentyl group, 5-methylhexyl, 2, 3-dimethylpentyl, 2, 4-dimethylpentyl, 2-dimethylpentyl, 3-dimethylpentyl, 2-ethylpentyl, 3-ethylpentyl, n-octyl, 2, 3-dimethylhexyl, 2, 4-dimethylhexyl, 2, 5-dimethylhexyl, 2-dimethylhexyl, 3-dimethylhexyl, 4-dimethylhexyl, 2-ethylhexyl, 3-ethylhexyl, 4-ethylhexyl, 2-methyl-2-ethylpentyl, 2-methyl-3-ethylpentyl, n-nonyl, 2-methyl-2-ethylhexyl, 2-methyl-3-ethylhexyl, 2-diethylpentyl, 2, 3-dimethylpentyl, 2-dimethylhexyl, 2, 3-dimethylhexyl, 2, 4-ethylhexyl, 2-methyl-2-ethylpentyl, 2-dimethylhexyl, 2-ethylhexyl, 2-ethylpentyl, 2-dimethylpentyl, 2-ethylhexyl, 3-dimethylpentyl, 2-ethylhexyl, 2-dimethylpentyl, 2-hexyl, 2-dimethylhexyl, 2-hexyl, and the like, N-decyl, 3-diethylhexyl, 2-diethylhexyl, and various branched isomers thereof.
As used herein, "cycloalkyl" refers to a saturated or partially unsaturated monocyclic cyclic hydrocarbon group, ")"C3-8Cycloalkyl "refers to a cyclic hydrocarbon group containing 3 to 8 carbon atoms, and may preferably be C3-6Cycloalkyl, defined similarly; non-limiting examples of cycloalkyl groups include cyclopropyl, cyclobutyl, cyclopentyl, cyclopentenyl, cyclohexyl, cyclohexenyl, cyclohexadienyl, cycloheptyl, cycloheptatrienyl, cyclooctyl, and the like, with cyclopropyl, cyclopentyl, cyclohexenyl being preferred.
As used herein, "C" is1-8Alkoxy means-O- (C)1-8Alkyl) wherein alkyl is as defined above. Preferably C1-6Alkoxy, more preferably C1-3An alkoxy group. Non-limiting examples include methoxy, ethoxy, propoxy, isopropoxy, butoxy, tert-butoxy, isobutoxy, pentoxy, and the like.
As used herein, "C" is3-8Cycloalkoxy means-O- (C)3-8Cycloalkyl), wherein cycloalkyl is as defined above. Preferably C3-6A cycloalkoxy group. Non-limiting examples include cyclopropoxy, cyclobutoxy, cyclopentyloxy, cyclohexyloxy, and the like.
As used herein, "C" is6-10Aryl "refers to an all-carbon monocyclic or fused polycyclic (i.e., rings which share adjacent pairs of carbon atoms) group having a conjugated pi-electron system, and refers to aryl groups containing 6 to 10 carbon atoms; phenyl and naphthyl are preferred, with phenyl being most preferred.
As used herein, "a bond" means that the two groups connected by it are linked by a covalent bond.
As used herein, "halogen" refers to fluorine, chlorine, bromine or iodine.
As used herein, "halo" refers to a group in which one or more (e.g., 1,2,3,4, or 5) hydrogens are replaced with a halogen.
For example, "halo C1-8Alkyl "means an alkyl group substituted with one or more (e.g., 1,2,3,4, or 5) halogens, wherein alkyl is as defined above. Is selected from halo C1-6Alkyl, more preferably halogenated C1-3An alkyl group. Halogen substituted C1-8Examples of alkyl groups include, but are not limited to, monochloromethyl, dichloromethyl, trichloromethyl, monochloroethyl, 1, 2-dichloroethyl, trichloroethyl, monobromoethylA group such as a fluoromethyl group, a difluoromethyl group, a trifluoromethyl group, a fluoroethyl group, a difluoroethyl group, and a trifluoroethyl group.
Also for example, "halo C1-8Alkoxy "means an alkoxy group substituted with one or more (e.g., 1,2,3,4, or 5) halogens, wherein the alkoxy group is as defined above. Preferably a halogen atom1-6Alkoxy, more preferably halo C1-3An alkoxy group. Halogen substituted C1-8Examples of alkoxy groups include, but are not limited to, trifluoromethoxy, trifluoroethoxy, monofluoromethoxy, monofluoroethoxy, difluoromethoxy, difluoroethoxy, and the like.
Also for example, "halo C3-8Cycloalkyl "refers to a cycloalkyl group substituted with one or more (e.g., 1,2,3,4, or 5) halogens, wherein cycloalkyl is as defined above. Preferably a halogen atom3-6A cycloalkyl group. Halogen substituted C3-8Examples of cycloalkyl groups include, but are not limited to, trifluorocyclopropyl, monofluorocyclopropyl, monofluorocyclohexyl, difluorocyclopropyl, difluorocyclohexyl, and the like.
As used herein, "amino" refers to NH2"cyano" means CN, "nitro" means NO2"benzyl" means-CH2-phenyl, "oxo" means ═ O, "carboxy" means-c (O) OH, "acetyl" means-c (O) CH3And "hydroxymethyl" means-CH2OH, "hydroxyethyl" means-CH2CH2OH or-CHOHCH3"hydroxy" means-OH, and "thiol" means SH.
As used herein, "heteroaryl ring" is used interchangeably with "heteroaryl" and refers to a monocyclic heteroaryl group having 5 to 10 ring atoms, preferably 5 or 6 membered or a bicyclic heteroaryl group having 8 to 10 membered ring atoms; 6, 10 or 14 pi electrons are shared in the ring array; and a group having 1 to 5 hetero atoms in addition to carbon atoms. "heteroatom" means nitrogen, oxygen or sulfur.
As used herein, "4 to 6 membered saturated monocyclic heterocycle" means that 1,2 or 3 carbon atoms in the 4 to 6 membered monocyclic ring are substituted by a substituent selected from nitrogen, oxygen or S (O)t(wherein t is an integer from 0 to 2) but excludes the ring moiety of-O-O-, -O-S-, or-S-S-, the remaining ring atoms being carbon; preferably 5 to 6 membered. Examples of 4-to 6-membered saturated monoheterocycles include (but are not limited to)) Propylene oxide, azetidine, oxetane, tetrahydrofuran, tetrahydrothiophene, tetrahydropyrrole, piperidine, pyrroline, oxazolidine, piperazine, dioxolane, dioxane, morpholine, thiomorpholine-1, 1-dioxide, tetrahydropyran, and the like.
As used herein, "unsaturated" refers to a pi-electron system that contains one or more unsaturated bonds but does not have a complete conjugation.
As used herein, "4 to 6 membered unsaturated mono-heterocyclic" includes (but is not limited to): 2, 3-dihydro-1H-pyrrole, 2, 5-dihydrofuran, 1,2,3, 6-tetrahydropyridine, 1,2,3, 4-tetrahydropyridine, 3, 6-dihydro-2H-pyran, 3, 4-dihydro-2H-pyran, and the like.
As used herein, "5-to 6-membered monocyclic heteroaryl ring" refers to a monocyclic heteroaryl ring containing 5 to 6 ring atoms, including for example (but not limited to): thiophene ring, N-alkylcyclopyrrole ring, furan ring, thiazole ring, imidazole ring, oxazole ring, pyrrole ring, pyrazole ring, triazole ring, 1,2, 3-triazole ring, 1,2, 4-triazole ring, 1,2, 5-triazole ring, 1,3, 4-triazole ring, tetrazole ring, isoxazole ring, oxadiazole ring, 1,2, 3-oxadiazole ring, 1,2, 4-oxadiazole ring, 1,2, 5-oxadiazole ring, 1,3, 4-oxadiazole ring, thiadiazole ring, pyridine ring, pyridazine ring, pyrimidine ring, pyrazine ring, and the like.
As used herein, "hydroxy-substituted C1-10Alkyl "means that one or more hydrogens on the alkyl group are replaced with a hydroxy group, including C substituted with a hydroxy group1-10Alkyl, dihydroxy-substituted C1-10Alkyl, trihydroxy-substituted C1-10Alkyl, and the like, examples include, but are not limited to, hydroxymethyl, hydroxyethyl, 2-hydroxy-2-methylbutyl, 2-hydroxy-butyl, and the like.
As used herein, "cyano-substituted C1-3Alkyl "means that one or more hydrogens on the alkyl group are replaced with a cyano group, including C substituted with a cyano group1-3Alkyl, dicyano substituted C1-3Alkyl and the like, examples include, but are not limited to, cyanomethyl, cyanoethyl and the like.
As used herein, "substituted" refers to one or more hydrogen atoms in the group, preferably 1 to 5 hydrogen atoms are substituted independently of each other with a corresponding number of substituents, more preferably 1 to 3 hydrogen atoms are substituted independently of each other with a corresponding number of substituents. It goes without saying that the substituents are only in their possible chemical positions, and that the person skilled in the art is able to determine (experimentally or theoretically) possible or impossible substitutions without undue effort. For example, amino or hydroxyl groups having free hydrogen may be unstable in combination with carbon atoms having unsaturated (e.g., olefinic) bonds.
As used herein, any of the above groups may be substituted or unsubstituted. When the above groups are substituted, the substituents are preferably 1 to 5 (more preferably 1 to 3) or less groups independently selected from halogen, -O (CH)2)pOC1-8Alkyl, -O (CH)2)pOH、-(CH2)pOC1-8Alkyl, 4-to 6-membered saturated monocyclic heterocycle, C1-8Alkyl (preferably C)1-6Alkyl, more preferably C1-3Alkyl group), C3-8Cycloalkyl (preferably C)3-6Cycloalkyl), halo C1-8Alkyl (preferably halogenated C)1-6Alkyl, more preferably halogenated C1-3Alkyl), halo C3-8Cycloalkyl (preferably halo C)3-6Cycloalkyl), hydroxy-substituted C1-8Alkyl (preferably hydroxy-substituted C)1-6Alkyl, more preferably hydroxy-substituted C1-3Alkyl), hydroxymethyl, hydroxyethyl, hydroxy, carboxyl, NRa0Rb0、-C(O)OC1-6Alkyl, acetyl, C1-8Alkoxy (preferably C)1-6Alkoxy, more preferably C1-3Alkoxy group), C1-8Alkoxy-substituted C1-8Alkyl (preferably C)1-6Alkoxy-substituted C1-6Alkyl, more preferably C1-3Alkoxy-substituted C1-3Alkyl), halo C1-8Alkoxy (preferably halo C)1-6Alkoxy, more preferably halo C1-3Alkoxy), -SO2C1-8Alkyl (preferably-SO)2C1-6Alkyl, more preferably-SO2C1-3Alkyl group), C6-10Aryl (preferably phenyl), 5-to 6-membered monocyclic heteroaryl, or-Y-L; wherein Y is (CH)2)qOr C (O); l is a 4 to 6 membered saturated monocyclic heterocycle or a 5 to 6 membered monocyclic heteroaryl ring; p, q are each independently 1,2 or 3; ra0、Rb0Each independently of the others is hydrogen, acetyl, C1-8Alkyl (preferably C)1-6Alkyl, more preferably C1-3Alkyl group), C1-8Alkoxy-substituted C1-8Alkyl (preferably C)1-6Alkoxy-substituted C1-6Alkyl, more preferably C1-3Alkoxy-substituted C1-3Alkyl groups).
The various substituent groups described herein above may themselves be substituted with groups described herein.
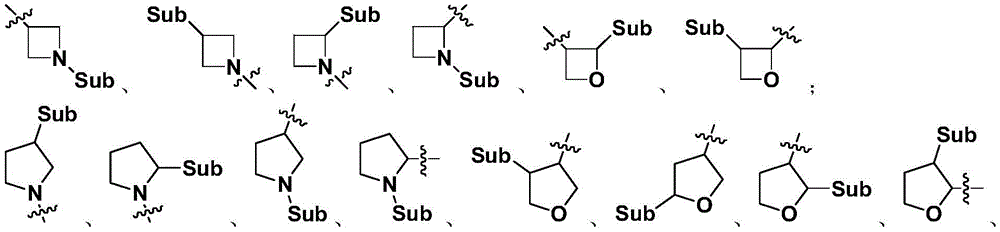
When the 4-to 6-membered saturated mono-heterocyclic ring described herein is substituted, the positions of the substituents may be at their possible chemical positions, and representative substitution of exemplary mono-heterocyclic rings are as follows:
wherein "Sub" represents the various types of substituents described herein;representing a connection to another atom.
When a 4-to 6-membered saturated mono-heterocyclic ring according to the present invention is a substituent, it may itself be substituted or substituted with 1,2 or 3 substituents selected from the group consisting of: halogen, hydroxy, C1-3Alkyl, O ═ NRa0Rb0Hydroxymethyl, hydroxyethyl, carboxy, -C (O) OC1-3Alkyl, acetyl, halo C1-3Alkyl radical, C1-3Alkoxy radical, C3-6Cycloalkyl, azetidine, oxetane, tetrahydrofuran, tetrahydrothiophene, tetrahydropyrrole, piperidine, oxazolidine, piperazine, dioxolane, dioxaneOxahexacyclic, morpholine, thiomorpholine-1, 1-dioxide, tetrahydropyran, thiophene ring, N-alkylpyrrole ring, furan ring, thiazole ring, imidazole ring, oxazole ring, pyrrole ring, pyrazole ring, triazole ring, tetrazole ring, isoxazole ring, oxadiazole ring, thiadiazole ring, pyridine ring, pyridazine ring, pyrimidine ring, pyrazine ring; wherein R isa0、Rb0Each independently is hydrogen or C1-3An alkyl group.
As used herein, "therapeutically effective amount" refers to an amount of a compound of the invention that will elicit the biological or medical response of an individual, e.g., decrease or inhibit enzyme or protein activity or ameliorate symptoms, alleviate a condition, slow or delay disease progression or prevent disease, etc.
As used herein, "pharmaceutically acceptable carrier" refers to a non-toxic, inert, solid, semi-solid substance or liquid filler, diluent, encapsulating material or auxiliary formulation or any type of adjuvant that is compatible with the patient, preferably a mammal, more preferably a human, and that is suitable for delivering an active agent to a target site without terminating the activity of the agent.
As used herein, "patient" refers to an animal, preferably a mammal, more preferably a human. The term "mammal" refers to warm-blooded vertebrate mammals, including, for example, cats, dogs, rabbits, bears, foxes, wolves, monkeys, deer, mice, pigs, and humans.
As used herein, "treating" or "treatment" refers to alleviating, delaying progression, attenuating, preventing, or maintaining an existing disease or disorder (e.g., cancer). Treatment also includes curing, preventing the development of, or alleviating to some extent one or more symptoms of the disease or disorder.
Preparation method
The compounds of formula (I) of the present invention can be readily prepared by various synthetic procedures, which are well known to those skilled in the art, according to the structure of the specific compound, with reference to the exemplary preparation methods in the following examples. Reagents and raw material compounds used in the preparation process are commercially available or can be prepared by a person skilled in the art according to different designed compound structures by a known method.
Compared with the prior art, the invention has the main advantages that:
provides a series of benzo-heterocycle substituted cyclopenta [4,5] pyrrolopyrazine-1-ketone derivatives with novel structures, which have higher inhibitory activity on BTK WT and other enzymes and pBTKY 223 and other cells and can be used as medicaments for treating tumors.
The invention will be further illustrated with reference to the following specific examples. It should be understood that these examples are for illustrative purposes only and are not intended to limit the scope of the present invention. The experimental procedures, in which specific conditions are not noted in the following examples, are generally carried out according to conventional conditions or according to conditions recommended by the manufacturers. Unless otherwise indicated, percentages and parts are by weight. Unless otherwise defined, terms used herein have the same meaning as those familiar to those skilled in the art. In addition, any methods and materials similar or equivalent to those described herein can be used in the present invention.
As used herein, DMB is 2, 4-dimethoxybenzyl, THF is tetrahydrofuran, EA is ethyl acetate, PE is petroleum ether, Ac2O is acetic anhydride, NBS is N-bromosuccinimide, DCM is dichloromethane, AIBN is azobisisobutyronitrile, Pd (dppf) Cl2Is 1,1' -bis (diphenylphosphino) ferrocene]Palladium dichloride, TFA trifluoroacetic acid, TBSCl tert-butyldimethylchlorosilane, NCS N-chlorosuccinimide, DHP dihydrotetrahydropyran, LiAlH4Is lithium aluminum hydride, PMB is p-methoxybenzyl, LiHMDS is lithium bis (trimethylsilyl) amide, Pd2(dba)3Is tris (dibenzylideneacetone) dipalladium, RuPhos is 2-dicyclohexylphosphonium-2 ',6' -diisopropoxy-1, 1' -biphenyl, DMAP is 4-dimethylaminopyridine, THP is tetrahydropyran, N-BuLi is N-butyllithium, TMsOTf is trimethylsilyl trifluoromethanesulfonate, TEABAC is triethylbenzylammonium chloride, HATU is 2- (7-azobenzotriazol) -N, N, N ', N ' -tetramethylurea hexafluorophosphate, DMF is dimethylformamide, DMSO is dimethyl sulfoxide, DIEA is N, N-diisopropylethylamine, BINAP is (2R,3S) -2,2' -bisdiphenylphosphino-1, 1' -binaphthyl.
As used herein, room temperature means about 20-25 ℃.
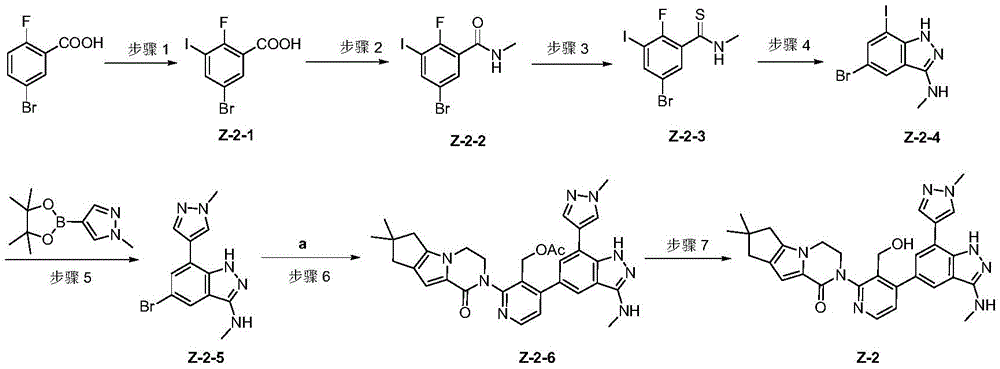
Preparation of intermediate a
Step 1: compound a-1(5 g, 14.6 mmol) was dissolved in 100ml of a mixed solvent of dichloromethane and methanol (volume ratio 1/1), and sodium borohydride (1.11 g, 29.2 mmol) was added in portions in an ice bath, and after the addition was completed, the ice bath was removed and stirred at room temperature for 3 hours. And LCMS detection shows that the raw materials are completely converted. The reaction mixture was concentrated, and the remaining oil was diluted with ethyl acetate, washed twice with water, washed once with saturated brine, then dried over anhydrous sodium sulfate, filtered and concentrated to give a-2(5 g, yield: 100%) as a colorless oil.
Step 2: compound a-2(5 g, 14.5 mmol) was dissolved in 100ml of dichloromethane, triethylamine (4.39 g, 43.5 mmol) was added, then a dichloromethane solution of acetyl chloride (2.26 g, 29 mmol) was added dropwise under cooling in an ice bath, and after completion of the addition, the reaction solution was naturally warmed to room temperature and stirred overnight. The reaction was washed with water directly, dried over anhydrous sodium sulfate, filtered, concentrated, and the remaining oil was purified by column chromatography (dichloromethane/methanol ═ 30/1) to give a-3(5 g, yield 89.3%) as a pale yellow oil.
And step 3: to a 100mL round bottom flask was added compound a-3(1 g, 2.58 mmol), bis pinacol boronate (1.31 g, 5.16 mmol), Pd (dppf) Cl2(94 mg, 0.13 mmol), X-Phos (124 mg,0.26mmol), potassium acetate (0.76 g, 7.74 mmol) and dioxane (35ml) were heated to 100 ℃ under nitrogen, stirred overnight, cooled to room temperature, filtered and concentrated, the remaining oil was diluted with 150ml ethyl acetate, washed with water, washed with saturated brine, dried over anhydrous sodium sulfate, filtered and concentrated to give a black oil a (yield)>100%, crude was used directly in the next step).
Example 12 preparation of 4- (3-amino-7- (5- (4-methylpiperazin-1-yl) pyridin-2-yl) -1H-indazol-5-yl) -3- (hydroxymethyl) pyridin-2-yl) -7, 7-dimethyl-2, 3,4,6,7, 8-hexahydro-1H-cyclopenta [4,5] pyrrolo [1,2-a ] pyrazin-1-one (Z-1)
Step 1: pd is added2(dba)3(229mg,0.25mmol), Xantphos (290mg,0.5mmol) and sodium tert-butoxide (2.88g,30mmol) were added to a solution of 2-bromo-5-iodopyridine (3.41g,12mmol) and 1-methylpiperazine (1.0g,10mmol) in toluene (50ml), heated to 60 ℃ under argon and the reaction stirred for 24 hours. After the reaction solution is cooled to room temperature, the solid is filtered, the filter cake is washed by ethyl acetate, the filtrate is decompressed and concentrated to obtain a crude product, and the crude product is subjected to Combi-flash column chromatography [ DCM: MeOH ═ 100: 0-90: 10]After purification, the compound 1- (6-bromopyridin-3-yl) -4-methylpiperazine (2.68g, 79.3%) was obtained. M+=256.0[M+1]+;
Step 2: a solution of compound B (1.3g,5.08mmol) in tetrahydrofuran (20ml) was cooled to-78 deg.C under argon and n-butyllithium (2.5M,3ml,7.62mmol) was slowly added dropwise. After the addition was complete, stirring was continued for 10 minutes and tributyltin chloride (2.48g,7.62mmol) was added. Slowly heating the reaction solution to room temperature, adding potassium fluoride to quench the reaction, and performing Combi-flash column chromatography on the crude product after decompression and concentration [ DCM: MeOH ═ 100: 0-95: 5]After purification, Compound Z-1-1(1.03g, 45%) was obtained. M+=468.2[M+1]+;
And step 3: compound Z-1-1(348mg,0.75mmol) and 5-bromo-7-iodo-1H-indazol-3-amine (252mg,0.75mmol) were dissolved in 5ml dioxane, tetrakistriphenylphosphine palladium (87mg,0.75mmol) and potassium fluoride (130mg,2.24mmol) were added, and the mixture was heated to 140 ℃ in a microwave reactor under argon atmosphere and reacted for 60 minutes. After cooling to room temperature, the reaction solution was poured into water, extracted with ethyl acetate (3 × 20ml), the organic phases were combined, washed with 30ml of saturated brine, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure to give a crude product, which was purified by Combi-flash column chromatography [ DCM: MeOH ═ 100: 0-90: 10, 0.1% Et3N]After purification, Compound Z-1-2(140mg, 40.6%) was obtained. M+=389.0[M+1]+;
And 4, step 4: the compound Z-1-2(100mg,0.26mmol) and the compound a (10)3mg,0.26mmol) was dissolved in a mixed solvent of 4ml of acetonitrile and 2ml of water, PdCl was added2(dppf) (19mg,0.026mmol), potassium phosphate (110mg,0.52mmol) and sodium acetate (71mg,0.52mmol) were heated to 110 ℃ in a microwave reactor under argon protection for 20 minutes. After the reaction solution was cooled to room temperature, it was diluted with 30ml of ethyl acetate, washed with 10ml of water and 10ml of saturated saline in this order, dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure to obtain a crude product, which was purified by Combi-flash column chromatography [ DCM: MeOH ═ 100: 0-90: 10, 0.1% Et3N]Purification gave Compound Z-1-3(140mg, 58.6%). M+=660.3[M+1]+;
And 5: reacting LiOH2O (30mg,0.71mmol) was added to a mixed solution of the compound Z-1-3(120mg,0.182mmol) in tetrahydrofuran (4ml), water (2ml) and isopropanol (2ml), and stirred at room temperature for two hours. The reaction solution was diluted with 30ml of ethyl acetate, washed with 20ml of water, washed with 20ml of saturated brine, dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure to give a crude product, which was purified by preparative liquid phase to give compound Z-1(35mg, 26.7%). M+=618.2[M+1]+;1H NMR(DMSO-d6,400MHz):=11.65(s,1H),8.47(d,J=4.9Hz,1H),8.41(d,J=2.9Hz,1H),8.06(d,J=1.2Hz,1H),7.94(d,J=9.0Hz,1H),7.85(d,J=1.2Hz,1H),7.45(dd,J=9.0,2.9Hz,1H),7.40(d,J=5.1Hz,1H),6.53(s,1H),5.50(br.s.,2H),4.96-5.03(m,1H),4.31-4.47(m,2H),4.10-4.30(m,3H),3.83(d,J=7.6Hz,1H),3.27(br.s.,4H),2.51-2.60(m,2H),2.49(br.s.,4H),2.39(s,2H),2.22(s,3H),1.18ppm(br.s.,6H).
Example 22 preparation of- (3- (hydroxymethyl) -4- (7- (1-methyl-1H-pyrazol-4-yl) -3- (methylamino) -1H-indazol-5-yl) pyridin-2-yl) -7-, 7-dimethyl-2, 3,4,6,7, 8-hexahydro-1H-cyclopenta [4,5] pyrrolo [1,2-a ] pyrazin-1-one (Z-2)
Step 1: 5-bromo-2-fluorobenzoic acid (8.68 g,40 mmol) was dissolved in 70ml of concentrated sulfuric acid, elemental iodine (10.16 g,40 mmol) was added in portions under cooling in an ice bath, the reaction solution was stirred at room temperature for 5 hours, the reaction solution was slowly poured onto crushed ice, a red solid was gradually precipitated, filtered, and dried under vacuum to give Z-2-1 as a red solid (11 g, yield 79.7%).
Step 2: in a round-bottomed flask, compound Z-2-1(3.45 g,10mmol), 25% methylamine aqueous solution (4ml), EDCI (3.84 g, 20 mmol), HOBT (2.7 g, 20 mmol), DIPEA (3.87 g,30mmol) and DMF (30ml) were added, the reaction solution was stirred overnight at room temperature, diluted with ethyl acetate, washed three times with water, washed with saturated sodium chloride, dried and concentrated, and the remaining oil was purified by column chromatography (petroleum ether/ethyl acetate: 4/1-2/1) to give Z-2-2(2.68 g,7.6 mmol) as a yellow solid.
And step 3: compound Z-2-2(0.5 g, 1.4 mmol) was dissolved in toluene (15ml), lawson's reagent (0.62 g, 1.54 mmol) was added, the reaction solution was warmed to 105 degrees, stirred for 4 hours, cooled to room temperature, concentrated, and the residue was purified by column chromatography (petroleum ether/ethyl acetate: 3/1) to give Z-2-3(0.45 g, yield: 86%) as a yellow solid.
And 4, step 4: compound Z-2-3(0.3 g, 0.8 mmol) was dissolved in DMSO (6ml), hydrazine hydrate (0.5ml) was added, the reaction was warmed to 120 degrees, stirred for 2 hours, cooled to room temperature, diluted with ethyl acetate, washed with water three times, washed with saturated sodium chloride, dried and concentrated, and the remaining oil was purified by column chromatography (petroleum ether/ethyl acetate 4/1-2/1) to give Z-2-4(0.15 g, yield 53.6%) as a yellow solid.
And 5: the compound Z-2-4(0.15 g, 0.43 mmol), 1-methyl-4- (4,4,5, 5-tetramethyl-1, 3, 2-dioxaborolan-2-yl) -1H-pyrazole (104 mg,0.5mmol), Pd (pddf) Cl2(16 mg, 0.0215 mmol), potassium carbonate (115 mg, 0.86 mmol) were mixed in dioxane (15ml), warmed to 70 ℃ overnight under nitrogen, cooled to room temperature, filtered, diluted with ethyl acetate, washed with water, washed with saturated sodium chloride, dried and concentrated, and the remaining oil was purified by column chromatography (petroleum ether/ethyl acetate: 2/1-1/2) to give Z-2-5 as a yellow oil (90 mg, yield: 68.7%).
Step 6: compound Z-2-5(104 mg,0.34mmol), compound a (135mg,0.34mmol), pd (dppf) Cl2(12 mg, 17 mmol), sodium acetate (56 mg,0.68mmol), potassium phosphate trihydrate (181 mg,0.68mmol), and dioxane (15ml) were mixed in a microwave tube, stirred under microwave conditions (100 degrees) for 20 minutes, cooled to room temperature, diluted with ethyl acetate, washed with water, washed with saturated brine, dried, and concentrated, and the remaining oil was purified by column chromatography (dichloromethane/methanol ═ 30/1) to give Z-2-6(146 mg, 60% yield) as a yellow oil.
And 7: compound Z-2-6(197 mg,0.34mmol) was dissolved in iPA/THF/H2O ═ 8ml/4ml/4ml, lithium hydroxide monohydrate (65 mg, 1.7 mmol) was added, stirred at room temperature for 1.5 hours, diluted with ethyl acetate, washed with water, washed with saturated brine, dried and concentrated, and the residue was purified by HPLC to give Z-2(8.5 mg) as a pale yellow solid.1H NMR(400MHz,DMSO-d6)δ11.52(s,1H),8.45(dd,J=5.0,1.2Hz,1H),8.29(d,J=5.5Hz,1H),7.98(d,J=0.9Hz,1H),7.74–7.65(m,2H),7.35(dd,J=5.1,2.5Hz,1H),6.52(s,1H),6.06(d,J=5.4Hz,1H),4.92(dt,J=6.0,3.4Hz,1H),4.38(t,J=5.1Hz,2H),4.19(m,3H),3.91–3.79(m,4H),2.87(d,J=5.0Hz,3H),2.54(d,J=8.2Hz,2H),2.39(s,2H),1.19(s,6H).
EXAMPLE 32 preparation of 4- (3-amino-7- (1-methyl-1H-pyrazol-4-yl) benzo [ d ] isoxazol-5-yl) -3- (hydroxymethyl) pyridin-2-yl) -7, 7-dimethyl-2, 3,4,6,7, 8-hexahydro-1H-cyclopenta [4,5] pyrrolo [1,2-a ] pyrazin-1-one (Z-3)
Step 1: the starting material acetylhydroxylamine (222 mg, 2.95 mmol) was dissolved in DMF, t-BuOK (330 mg, 2.95 mmol) was added, the temperature was raised to 30 degrees, stirring was performed for 1 hour, 5-bromo-2-fluoro-3-iodobenzonitrile (800 mg, 2.46 mmol) was added, the reaction solution was stirred overnight, diluted with ethyl acetate, washed three times with water, washed with saturated sodium chloride, dried and concentrated, and the residue was purified by column chromatography (petroleum ether/ethyl acetate: 2/1-1/1) to give Z-3-1 as a pale yellow solid (390 mg, yield: 47%).
Step 2: for detailed operation reference is made to step 5 of the final product Z-2 operation.
And step 3: for detailed operation reference is made to step 6 of the final product Z-2 operation.
And 4, step 4: for detailed operation reference is made to step 7 of the final product Z-2 operation.1H NMR(400MHz,DMSO-d6)δ8.53(d,J=5.0Hz,1H),8.38(s,1H),8.07(s,1H),7.98(d,J=1.7Hz,1H),7.79(d,J=1.6Hz,1H),7.40(d,J=5.0Hz,1H),6.57-6.52(m,3H),4.94(t,J=5.2Hz,1H),4.39(d,J=5.2Hz,2H),4.30–4.20(m,3H),3.94(s,3H),3.87(d,J=10.4Hz,1H),2.58(d,J=8.3Hz,2H),2.43(s,2H),1.22(s,6H).
Example 42 preparation of (3- (hydroxymethyl) -4- (3-methyl-7- (1-methyl-1H-pyrazol-4-yl) -1H-indazol-5-yl) pyridin-2-yl) -7, 7-dimethyl-2, 3,4,6,7, 8-hexahydro-1H-cyclopenta [4,5] pyrrolo [1,2-a ] pyrazin-1-one (Z-4)
Step 1: iodine (2.54g,10.0mmol) and silver sulfate (3.12g,10.0mmol) were added to a solution of 4-bromo-2-ethylaniline (2.0g,10.0mmol) in ethanol (30ml), and the mixture was stirred at room temperature for 3 hours. The solid was removed by filtration, the filtrate was concentrated and then dissolved in 50ml of dichloromethane, washed with aqueous sodium hydroxide solution (1N, 30ml), saturated brine (30ml), dried over anhydrous sodium sulfate, filtered, the filtrate was concentrated under reduced pressure to obtain a crude product, which was purified by Combi-flash column chromatography [ PE: EA is 100: 0-95: 5]After purification, the compound 4-bromo-2-ethyl-6-iodoaniline (1.97g, 60.4%) was obtained. M+=328.0[M+1]+;1H NMR(400MHz,DMSO-d6)7.53(d,J=2.4Hz,1H),7.08(d,J=2.4Hz,1H),5.02(S,2H),2.45-2.51(m,2H),1.07(t,J=7.6Hz,3H).
Step 2: sodium nitrite (54mg,0.78mmol) was added to a solution of 4-bromo-2-ethyl-6-iodoaniline (230mg,0.71mmol) in acetic acid (5ml), the reaction was stirred at room temperature for 3 hours, and the reaction was concentrated under reduced pressure to give a crude product, which was purified by Combi-flash column chromatography [ DCM: MeOH 99: 1]Purification gave compound Z-4-1(156mg, 65.5%). M+=328.0[M+1]+;1H NMR(400MHz,DMSO-d6)12.98(s,1H),7.96(d,J=1.6Hz,1H),7.80(d,J=1.6Hz,1H),2.42(s,3H)
And step 3: PdCl2(dppf) (92mg,0.125mmol) and potassium carbonate (345mg,2.5mmol) were added to a mixed solution of compound Z-4-1(420mg,1.25mmol) and 1-methyl-4- (4,4,5, 5-tetramethyl-1, 3, 2-dioxaborolan-2-yl) -1H-pyrazole (260mg,1.25mmol) in 8ml dioxane and 2ml water. Stir vigorously at 65 ℃ for 7 hours under nitrogen. After the reaction is finished, filtering, decompressing and concentrating the filtrate to obtain a crude product, and performing Combi-flash column chromatography [ DCM: MeOH ═ 100: 0-95: 5]Purification gave Compound Z-4-2(250mg, 68.7%). M+=291.0[M+1]+;
And 4, step 4: PdCl2(dppf) (50mg,0.069mmol), potassium phosphate (291mg,1.37mmol) and sodium acetate (187mg,1.37mmol) were added to a mixed solution of compound Z-4-2(200mg,0.68mmol) and compound a (273mg,0.68mmol) in 5ml of acetonitrile and 2ml of water. The reaction was carried out in a microwave reactor at 110 ℃ for 15 minutes under a nitrogen atmosphere. After the reaction is finished, filtering, decompressing and concentrating the filtrate to obtain a crude product, and performing Combi-flash column chromatography [ DCM: MeOH ═ 100: 0-95: 5]Purification gave compound Z-4-3(117mg, 30.2%).
And 5: reacting LiOH2O (30mg,0.71mmol) was added to a mixed solution of Z-4-3(100mg,0.177mmol) in tetrahydrofuran (4ml), water (2ml) and isopropanol (2ml), and stirred at room temperature for two hours. The reaction solution was diluted with 30ml of ethyl acetate, washed with 20ml of water, washed with 20ml of saturated brine, dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure to give a crude product, which was purified by preparative liquid phase to give compound Z-4(50mg, 54.3%). M+=522.2[M+1]+;1H NMR(DMSO-d6,400MHz):=12.72(br.s.,1H),8.47(d,J=4.9Hz,1H),8.35(s,1H),8.03(s,1H),7.76(d,J=1.5Hz,1H),7.71(d,J=1.5Hz,1H),7.42(d,J=5.1Hz,1H),6.52(s,1H),4.99(t,J=5.0Hz,1H),4.35(d,J=4.9Hz,2H),4.10-4.29(m,3H),3.90(s,3H),3.79-3.87(m,1H),2.50-2.59(m,5H),2.40(s,2H),1.19ppm(s,6H).
Example 52 preparation of 4- (3-amino-7- (4- (morpholinomethyl) phenyl) -1H-indazol-5-yl) -3- (hydroxymethyl) pyridin-2-yl) -7, 7-dimethyl-2, 1,3,4,6,7, 8-hexahydro-1H-cyclopenta [4,5] pyrrolo [1,2-a ] pyrazin-1-one (Z-5)
Step 1: 5-bromo-2-fluoro-3-iodobenzonitrile (2 g, 6.1 mmol) was dissolved in THF (40ml), 85% hydrazine hydrate (1.5ml) was added, the temperature was raised to 30 ℃, stirred overnight, diluted with ethyl acetate, washed three times with water, washed with saturated sodium chloride, dried and concentrated, and the residue was purified by column chromatography (petroleum ether/ethyl acetate: 2/1-1/1) to give Z-5-1(1.9 g, yield: 91.8%) as a pale yellow solid.
Step 2: for detailed operation reference is made to step 5 of the final product Z-2 operation.
And step 3: for detailed operation reference is made to step 6 of the final product Z-2 operation.
And 4, step 4: for detailed operation reference is made to step 7 of the final product Z-2 operation.1H NMR(400MHz,DMSO-d6)δ11.66(s,1H),8.46(d,J=5.0Hz,1H),7.83(d,J=1.6Hz,1H),7.69-7.65(m,2H),7.57(d,J=1.6Hz,1H),7.44–7.35(m,3H),5.48(s,2H),4.92(t,J=5.1Hz,1H),4.39-4.41(m,2H),4.20-4.16(m,3H),3.85-3.87(m,1H),3.59-3.54(m,4H),3.50(s,2H),2.62–2.47(m,3H),2.38(d,J=8.7Hz,6H),1.19(s,6H).
EXAMPLE 62 preparation of (4- (3-amino-7- (4- (morpholinomethyl) phenyl) benzo [ d ] isoxazol-5-yl) -3- (hydroxymethyl) pyridin-2-yl) -7, 7-dimethyl-3-cyclopenta [4,5] pyrrolo [1,2-a ] pyrazin-1 (6H) -one (Z-6)
Step 1: 5-bromo-2-fluoro-3-iodobenzonitrile (920mg,12.3mmol) was dissolved in DMF (30mL), and potassium tert-butoxide (1.38g,12.3mmol) was added to the reaction solution, and the resulting reaction solution was stirred under protection of helium at room temperature overnight. Then, N-hydroxyacetamide (4g,12.3mmol) was added to the reaction solution, and the temperature was raised to 50 ℃ for reaction for 2 hours. After the reaction solution was extracted with ethyl acetate and water, the organic layer was concentrated to obtain a crude product, which was separated and purified by silica gel column chromatography to obtain a pure product of 5-bromo-7-iodobenzo [ d ] isoxazol-3-amine (2.2g, yield 53%).
Step 2: mixing 5-bromo-7-iodobenzo [ d ]]Isoxazol-3-amine (500mg,1.5mmol), 4- (morpholinomethyl) phenylboronic acid (326mg,1.5mmol), Pd (dppf) Cl2(110mg,0.15mmol) and K2CO3(622mg,4.5mmol) was placed in a microwave tube, dioxane (10mL) and water (5 drops) were added, and the reaction mixture was reacted at 80 ℃ for 40min using a microwave synthesizer. After the reaction mixture was extracted with ethyl acetate and water, the organic layer was concentrated to obtain a crude product, which was separated and purified by silica gel column chromatography to obtain a pure compound Z-6-1(220mg, yield 38.4%).
And step 3: compound Z-6-1(220mg,0.57mmol) was dissolved in dichloromethane (20mL) and (Boc) was added to the reaction solution separately2O (247mg,1.13mmol) and DMAP (7mg,0.057 mmol). The resulting reaction mixture was stirred at room temperature overnight. After the reaction solution was extracted with methylene chloride and water, the organic layer was concentrated to obtain a crude product, which was separated and purified by silica gel column chromatography to obtain a pure compound Z-6-2 (280mg, yield 84%)
And 4, step 4: compound a (135mg,0.34mmol), compound Z-6-2(200mg,0.34mmol), Pd (dppf) Cl2(25mg,0.034mmol) and K2CO3(141mg,1.02mmol) was placed in a microwave tube, dioxane (10mL) and water (5 drops) were added, and the reaction mixture was reacted at 110 ℃ for 30min using a microwave synthesizer. After the reaction mixture was extracted with ethyl acetate and water, the organic layer was concentrated to obtain a crude product, which was separated and purified by silica gel column chromatography to obtain a pure compound Z-6-3 (180mg, yield 61.6%).
And 5: compound Z-6-3(170mg,0.197mmol) was dissolved in TFA (10mL), and the reaction was stirred at room temperature for 30 min. After most of TFA was removed under reduced pressure, the reaction solution was neutralized with saturated sodium bicarbonate solution, and after the reaction solution was extracted with ethyl acetate and water, the organic layer was concentrated to obtain the crude compound Z-6-4, which was used in the next step.
Step 6: compound Z-6-4(30mg,0.045mmol) was dissolved in THF (5mL) and added to LiOH (300 m)g,12.5mmol) in an aqueous solution (5 mL). The reaction mixture was stirred at room temperature for 1 hour. After most of THF was removed under reduced pressure, the reaction solution was extracted with ethyl acetate and water, and the mixture obtained after the organic layer was concentrated under reduced pressure was purified by preparative liquid chromatography to obtain the final product Z-6(8.1mg, yield 29%).1H NMR(400MHz,DMSO-d6)δ8.50(d,J=5.0Hz,1H),7.98-7.96(m,2H),7.88-7.85(m,2H),7.45-7.42(m,3H),6.55-6.52(m,3H),4.96(t,J=5.2Hz,1H),4.36(d,J=5.2Hz,2H),4.20-4.17(m,3H),3.86-3.84(m,1H),3.55(t,J=4.7Hz,4H),3.49(s,2H),2.62–2.51(m,3H),2.39-2.35(m,5H),1.18(s,6H).
Example 72 preparation of (4- (3-amino-7- (1-methyl-1, 2,3, 6-tetrahydropyridin-4-yl) -1H-indazol-5-yl) -3 (hydroxymethyl) pyridin-2-yl) -7, 7-dimethyl-3, 4,7, 8-tetrahydro-2H-cyclopenta [4,5] pyrrolo [1,2-a ] pyrazin-1 (6H) -one (Z-7)
Step 1: to a 100ml round bottom flask was added 1-methyl-4- (4,4,5, 5-tetramethyl-1, 3, 2-dioxaborolan-2-yl) -1,2,3, 6-tetrahydropyridine (446 mg, 2.00 mmol), 5-bromo-7-iodo-1H-indazol-3-amine (676 mg, 2.00 mmol), Pd (dppf) Cl2(73 mg,0.1 mmol), sodium carbonate (630 mg, 6mmol), water (5mL) and dioxane (35mL), warmed to 100 ℃ under nitrogen, stirred overnight, cooled to room temperature, filtered and concentrated, the remaining oil was diluted with 150mL ethyl acetate, washed with water, washed with saturated brine, dried over anhydrous sodium sulfate, filtered and concentrated, and subjected to silica gel column chromatography to give Z-7-1(618 mg, yield: 100%) as a white solid.
Step 2: into a 100ml round-bottomed flask, the above-mentioned compound Z-7-1(618 mg, 2.00 mmol), compound a (800 mg, 2.00 mmol), Pd (dppf) Cl were added2(73 mg,0.1 mmol), potassium carbonate (830 mg, 6mmol), water (5mL) and dioxane (35mL) under nitrogen, stirring at elevated temperature overnight, cooling to room temperature, filtering and concentrating, diluting the remaining oil with 150mL ethyl acetate, washing with water, and saturating with foodWashed with brine, dried over anhydrous sodium sulfate, filtered, concentrated, subjected to silica gel column chromatography and then subjected to HPLC to prepare compound Z-7(200 mg, yield: 19%).1H NMR(400MHz,DMSO-d6)δ8.44(d,J=5.0Hz,1H),8.12(s,1H),7.73(s,1H),7.40(s,1H),7.32(d,J=5.1Hz,1H),6.51(s,1H),6.24(s,2H),5.44(s,2H),4.89(s,1H),4.35(s,2H),4.22–4.08(m,3H),3.81(s,1H),3.12(s,2H),2.81–2.49(m,6H),2.39(s,2H),2.32(s,3H),1.18(s,6H).
Example 82 preparation of 4- (3-amino-7- (1- (1-methylpiperidin-4-yl) -1H-pyrazol-4-yl) -1H-indazol-5-yl ] -3- (hydroxymethyl) pyridin-2-yl) -7, 7-dimethyl-3, 4,7, 8-tetrahydro-2H-cyclopenta [4,5] pyrrolo [1,2-a ] pyrazin-1 (6H) -one (Z-8)
Step 1: to a 100mL round bottom flask, compound Z-8-1(752 mg, 2.00 mmol), trifluoroacetic acid (5mL), and dichloromethane (5mL) were added, stirred overnight at room temperature under nitrogen, and concentrated to dryness to give Z-8-2 as an oil which was taken directly to the next step (yield: 100%).
Step 2: to a 100ml round bottom flask, compound Z-8-2(750 mg, 2.00 mmol), aqueous formaldehyde (1ml), dichloromethane (20ml), tetrahydrofuran (20ml) were added in portions, sodium borohydride acetate (1266 mg, 6.00 mmol) was added in portions, the reaction was stirred overnight, quenched with a saturated ammonium chloride solution in ice bath, extracted with dichloromethane, dried over anhydrous sodium sulfate as an organic phase, filtered, concentrated, and subjected to silica gel column chromatography to obtain Z-8-3(500 mg, yield: 86%) as a white solid.
And step 3: to a 100ml round bottom flask was added compound Z-8-3(500 mg, 1.72 mmol), 5-bromo-7-iodo-1H-indazol-3-amine (580 mg, 1.72 mmol), Pd (dppf) Cl2(73 mg,0.1 mmol), sodium carbonate (630 mg, 6mmol), water (5mL) and dioxane (35mL) under nitrogen, heating to 100 ℃, stirring overnight, cooling to room temperature, filtering and concentrating, diluting the residual oily substance with 150mL ethyl acetate, washing with water, washing with saturated salt water, drying with anhydrous sodium sulfate, filtering, concentrating, and performing silica gel column chromatography to obtain the final productTo a white solid, Z-8-4(375 mg, yield: 58%).
And 4, step 4: to a 100mL round-bottom flask was added the above compound Z-8-4(125 mg, 0.33 mmol), compound a (132 mg, 0.33 mmol), Pd (dppf) Cl2(15 mg,0.2 mmol), potassium carbonate (830 mg, 6mmol), water (5mL) and dioxane (35mL), under nitrogen, stirred at elevated temperature under reflux overnight, cooled to room temperature, filtered and concentrated, the remaining oil was diluted with 150mL ethyl acetate, washed with water, washed with saturated brine, dried over anhydrous sodium sulfate, filtered and concentrated, and then subjected to silica gel column chromatography followed by HPLC to give compound Z-8(100 mg, yield: 50%).1H NMR(400MHz,DMSO-d6)δ11.50(s,1H),8.45(d,J=5.0Hz,1H),8.39(s,1H),8.00(s,1H),7.71(s,2H),7.36(d,J=5.1Hz,1H),6.51(d,J=5.2Hz,1H),5.46(s,2H),4.96–4.85(m,1H),4.37(d,J=6.2Hz,2H),4.27–4.07(m,4H),3.85(d,J=22.2Hz,1H),2.85(d,J=6.0Hz,2H),2.56(t,J=11.9Hz,2H),2.39(s,2H),2.19(s,3H),2.06–1.96(m,5H),1.19(s,6H).
Example 9
Compound Z-9 was prepared according to the method of example 1.1H NMR(400MHz,DMSO-d6)δ11.70(s,1H),8.61–8.38(m,2H),8.11(s,1H),8.00(d,J=9.0Hz,1H),7.89(s,1H),7.47(dd,J=22.2,5.6Hz,2H),6.57(s,1H),5.54(s,2H),5.03(s,1H),4.43(s,2H),4.24(d,J=10.6Hz,2H),3.83(d,J=28.8Hz,4H),3.38(s,1H),3.26(s,4H),2.61(t,J=19.8Hz,2H),2.38(d,J=40.8Hz,2H),1.22(s,6H).
Examples 10 to 14
Compounds Z-10 to Z-14 can be prepared by the method described in example 2.
Examples 15 to 16
Compounds Z-15 to Z-16 can be prepared by the method described in example 3.
Example 17
Compound Z-17 can be prepared by the method described in example 5.1H NMR(400MHz,DMSO-d6)δ11.80(s,1H),8.65(d,J=2.2Hz,1H),8.48(d,J=5.0Hz,1H),8.20(d,J=1.5Hz,1H),8.07(d,J=8.3Hz,1H),7.95(d,J=1.4Hz,1H),7.80(dd,J=8.2,2.2Hz,1H),7.63–7.47(m,2H),7.41(d,J=5.0Hz,1H),6.53(s,1H),5.53(s,2H),5.03–4.95(m,1H),4.39(t,J=4.7Hz,2H),4.31–4.04(m,3H),3.86-3.84(m,1H),3.57-3.53(m,6H),2.55(d,J=8.3Hz,2H),2.39-2.36(m,4H),1.19(d,J=2.7Hz,6H).
Example 18
Compound Z-18 can be prepared by a method described in example 8.1H NMR(400MHz,DMSO-d6)δ11.52(s,1H),8.54–8.32(m,2H),8.01(s,1H),7.71(s,2H),7.36(d,J=5.0Hz,1H),6.52(s,1H),5.49(s,2H),4.99–4.86(m,1H),4.40(d,J=12.6Hz,3H),4.30–4.06(m,3H),3.96(d,J=11.2Hz,2H),3.82(s,1H),3.47(t,J=9.8Hz,2H),2.54(d,J=8.4Hz,2H),2.39(s,2H),2.00(d,J=18.4Hz,4H),1.19(s,6H).
Biological assay
Test example-Lantha screening kinase reaction experiment method
Compounds were pre-dissolved in 100% DMSO. 10mM of drug stock was dissolved at room temperature and diluted in 8 vol% DMSO gradient to a final concentration of 10-0.005. mu.M. The reaction was initiated by adding 2.5. mu.l of the analyte solution and 2.5. mu.l of the kinase (Invitrogen PV3363) diluted in the reaction buffer to each well of a 384-well plate (Corning 3676), and diluting a mixture of Fluososci-PolyGT (Invitrogen PV3610) substrate and ATP (Invitrogen PV3227) with 5. mu.l of the reaction buffer. Where Blank wells (Blank) were used in place of kinase with reaction buffer and kinase wells (Enzyme) were not loaded with any drug. After shaking the reaction mixture at 25 ℃ for 60 minutes in the absence of light. Mu.l of Detection Solution (Invitrogen PV3528 mixed Solution with EDTA diluted with TR-FRET dilution buffer at a working concentration of 5mM EDTA and 0.2nM lanthastening Tb PY20 antibody) was added and the mixture was subjected to shake reaction at room temperature for 30 minutes. The plate was read on a VictorX5 fluorescence microplate reader (PerkinElmer) and the light absorption was measured at an excitation wavelength of 340nm, emission wavelengths of 500nm and 520 nm.
The inhibition was calculated as follows (see Invitrogen, specification PV 3363):
1.Emission rate(ER):Coumarin Emission(520nm)/Fluorescein Emission(500nm)
2. inhibition Ratio (IR): (ER)kinase-ERtest compound)/(ERkinase-ERblank) X 100%. Half inhibitory concentration IC50 was calculated by fitting using XLFIT 5.0 software (IDBS, england). The results are shown in table 1:
inhibitory Activity of the Compounds of Table 1 on BTK WT
Test example two experimental methods for detecting HTRF by intracellular beta BTK Y223 phosphorylation
Compounds were pre-dissolved in 100% DMSO. 10mM of drug stock was dissolved at room temperature and diluted in 5 vol% DMSO gradient to a final concentration of 3-0.0014. mu.M. Ramos cells were plated at 4X 105Per well density into 96-well plate, 45. mu.l each well of 1640 medium containing 10% (V/V) FBS, 5. mu.l each well of diluted analyte solution, 37 ℃ and 5% (V/V) CO2The culture was carried out for 1 hour. Add 10. mu.l of sodium peroxovanadate diluent (serum-free 1640 dilution) to the negative control wells, 10. mu.l of noneSerum medium was incubated with shaker at 25 ℃ for 30 min. Mu.l of lysis buffer (4 Xlysis: blocking stock 25: 1) was added to each well and incubated for 30min at 25 ℃ in a shaker. After shaking at 800rpm for 1 minute, 16. mu.l of the cell lysate was added to a 384-well plate (Greiner784075), 4. mu.l of the premixed antibody solution (Phospho-BTK d2 antibody and Phospho-BTK Cryptate antibody were diluted 20-fold using the detection solution) was added, and the mixture was incubated overnight at 25 ℃ with shaking. The plates were read on a VictorX5 fluorescence microplate reader (PerkinElmer) and the light absorption was measured at 317nm excitation wavelength, 500nm emission wavelength and 520nm emission wavelength (cf. Cisbio, 63ADK017PEH instructions). Half inhibitory concentration IC50 was calculated by fitting using XLFIT 5.0 software (IDBS, england). The results are shown in table 2:
TABLE 2 inhibitory Activity of Compounds on beta BTK Y223 cells
As can be seen from tables 1 and 2, representative compounds of the present invention have high inhibitory activity against enzymes and cells.
Test example three Activity inhibition test for wild-type EGFR kinase
Reagents used in the following z-lyte test methods are all available from Invitrogen.
The inhibitory activity of the test substances against wild-type EGFR kinase (Invitrogen, PV3872) was determined by the z-lyte method.
The working concentrations of the components in the 10uL wild-type EGFR kinase reaction system were: mu.M ATP, 0.8 ng/. mu.L wild type EGFR kinase (Invitrogen, PV3872), 2. mu.M Tyr04 substrate (Invitrogen, PV 3193). The final concentration of DMSO after addition of the test substance was 2%.
A10 mM stock of drug dissolved at room temperature was diluted to a final concentration of 10-0.005. mu.M with a gradient of 4% DMSO in water. 2.5. mu.L of the test substance solution and 5. mu.L of a mixture of wild-type EGFR kinase and Tyr04 substrate diluted in the reaction buffer were added to each wellThen, 2.5. mu.L of ATP was added to initiate the reaction. Wherein the C1 well is used as a reaction buffer to replace ATP, the C2 well is not added with any drug, and the C3 well is added with a phosphorylated substrate as described in the specification. After 60 minutes of reaction at 25 ℃ with shaking and protection from light. 5 μ L of fusion Reagent B (Invitrogen, diluted with TR-FRET dilution buffer) was added and the reaction was shaken for 60 minutes at room temperature. The plates are read on a VictorX5 fluorescence microplate reader (PerkinElmer) and the light absorption at an excitation wavelength of 405nm, emission wavelengths of 450nm and 520nm is determined (e.g., C3)520nmIndicating the reading at 520nm of the C3 pore).
The inhibition was calculated as follows (see Invitrogen, specification PV 3193):
1、ER=Coumarin Emission(450nm)/Fluorescein Emission(520nm)
2. phosphorylation rate (1- ((ER × C3)520nm-C3450nm)/((C1450nm-C3450nm)+ER×(C3520nm-C1520nm))))×100%
3. Inhibition Ratio (IR) ═ 1- (phosphorylation rate of test compound)/(phosphorylation rate of C2)) × 100%
Half maximal inhibitory concentration IC was calculated by fitting XLFIT 5.0 software (IDBS, UK)50。
TABLE 3 inhibitory Activity of Compounds on EGFR WT
As can be seen from table 3, representative compounds of the invention have lower inhibitory activity against wild-type EGFR kinase. Thus, the exemplary compounds of the invention have selective inhibitory activity against BTK WT kinase.
All documents referred to herein are incorporated by reference into this application as if each were individually incorporated by reference. Furthermore, it should be understood that various changes and modifications of the present invention can be made by those skilled in the art after reading the above teachings of the present invention, and these equivalents also fall within the scope of the present invention as defined by the appended claims.
Claims (16)
1. A compound of formula (I), or a pharmaceutically acceptable salt thereof:
in the formula (I), the compound is shown in the specification,
ring a is phenyl, a 5-to 6-membered monocyclic heteroaryl ring, or a 4-to 6-membered unsaturated monocyclic heterocycle;
R0is hydrogen, halogen, C1-8Alkyl, halo C1-8Alkyl radical, C1-8Alkoxy, halo C1-8Alkoxy, optionally substituted C3-8Cycloalkyl radical, C3-8Cycloalkoxy or-Y-L; wherein Y is a bond or- (CH)2)m-; l is hydroxy, cyano-substituted C1-3Alkyl, optionally substituted 4-to 6-membered saturated monocyclic heterocycle, C1-8Alkoxy or NRa0Rb0(ii) a Said optionally substituted being unsubstituted or substituted by 1,2 or 3 hydroxy, halogen, C1-10Alkyl, hydroxy substituted C1-10Alkyl, halo C1-8Alkyl substituted;
n, m are each independently 1,2 or 3;
z is O or NRa;
R1Is halogen, C1-10Alkyl or NRa0Rb0;
Ra、Ra0、Rb0Each independently is hydrogen or C1-8An alkyl group;
R2、R3、R4、R5each independently is hydrogen;
R6、R7each independently is C1-8An alkyl group.
2. The compound of claim 1, or a pharmaceutically acceptable salt thereof, wherein in formula (I), the 4-to 6-membered saturated monoheterocycle is selected from the group consisting of: azetidine, oxetane, tetrahydrofuran, tetrahydrothiophene, tetrahydropyrrole, piperidine, oxazolidine, piperazine, dioxolane, dioxane, morpholine, or tetrahydropyran.
3. The compound of claim 1, or a pharmaceutically acceptable salt thereof, wherein in formula (I), the 5-to 6-membered monocyclic heteroaryl ring is selected from: a thiophene ring, an N-alkyl ring pyrrole ring, a furan ring, a thiazole ring, an imidazole ring, an oxazole ring, a pyrrole ring, a pyrazole ring, a triazole ring, a tetrazole ring, an isoxazole ring, an oxadiazole ring, a thiadiazole ring, a pyridine ring, a pyridazine ring, a pyrimidine ring, or a pyrazine ring.
4. The compound of claim 1, or a pharmaceutically acceptable salt thereof, wherein in formula (I), the 4-to 6-membered unsaturated mono-heterocyclic ring is selected from: 2, 3-dihydro-1H-pyrrole, 2, 5-dihydrofuran, 1,2,3, 6-tetrahydropyridine, 1,2,3, 4-tetrahydropyridine, 3, 6-dihydro-2H-pyran or 3, 4-dihydro-2H-pyran.
5. The compound according to claim 1, or a pharmaceutically acceptable salt thereof, wherein in formula (I), ring a is a benzene ring, a pyrazole ring, a pyrimidine ring, a pyridine ring, or a 1,2,3, 6-tetrahydropyridine ring.
6. The compound of claim 1, or a pharmaceutically acceptable salt thereof, wherein in formula (I), n is 1 or 2.
7. The compound of claim 1, or a pharmaceutically acceptable salt thereof, wherein in formula (I), R is0Is hydrogen, C1-3Alkyl or-Y-L; wherein Y is a bond or- (CH)2)m-; l is optionally substituted piperidine, piperazine, morpholine or tetrahydropyran; said optionally substituted piperidine, piperazine, morpholine or tetrahydropyran is unsubstituted or substituted by 1 or 2C1-3Alkyl substituted; m is 1.
12. a pharmaceutical composition comprising a compound of claim 1, or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable carrier or excipient.
13. Use of a compound according to any one of claims 1 to 11, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition according to claim 12, in the manufacture of a medicament for use as a kinase inhibitor.
14. The use of claim 13, wherein the kinase inhibitor drug is a drug for the treatment of a B cell mediated disease.
15. The use according to claim 13, wherein the kinase inhibitor medicament is a medicament for the treatment of at least one of a proliferative disease, an allergic disease, an autoimmune disease, and an inflammatory disease.
16. The use according to claim 13, wherein the kinase inhibitor medicament is a medicament for the treatment of a neoplastic disease.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201810864321 | 2018-08-01 | ||
| CN2018108643214 | 2018-08-01 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN110256446A CN110256446A (en) | 2019-09-20 |
| CN110256446B true CN110256446B (en) | 2021-12-14 |
Family
ID=67912351
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201910689812.4A Active CN110256446B (en) | 2018-08-01 | 2019-07-29 | Benzo-heterocycle substituted cyclopenta [4,5] pyrrolopyrazine-1-one derivatives and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN110256446B (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN119930630A (en) | 2020-02-20 | 2025-05-06 | 和记黄埔医药(上海)有限公司 | Heteroaryl heterocyclic compounds and uses thereof |
| KR20230104125A (en) * | 2020-09-21 | 2023-07-07 | 허치슨 메디파르마 리미티드 | Heteroaryl Heterocyclic Compounds and Uses Thereof |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2552514T3 (en) * | 2011-11-03 | 2015-11-30 | Hoffmann-La Roche Ag | Bicyclic Piperazine Compounds |
| JP6507234B2 (en) * | 2014-10-02 | 2019-04-24 | エフ.ホフマン−ラ ロシュ アーゲーF. Hoffmann−La Roche Aktiengesellschaft | Pyrazole carboxamide compounds for use in the treatment of disorders mediated by bruton tyrosine kinase (BTK) |
| ES2907622T3 (en) * | 2014-10-06 | 2022-04-25 | Merck Patent Gmbh | Heteroaryl Compounds as BTK Inhibitors and Uses of These |
| WO2016161571A1 (en) * | 2015-04-08 | 2016-10-13 | Merck Sharp & Dohme Corp. | Indazole and azaindazole btk inhibitors |
| UY37559A (en) * | 2017-01-06 | 2018-05-31 | Pharmacyclics Llc | PIRAZOLO [3,4-B] PIRIDINE AND PIRROLO [2,3-B] PIRIDINE AS INHIBITORS OF BRUTON TYPOSINE KINASE |
-
2019
- 2019-07-29 CN CN201910689812.4A patent/CN110256446B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN110256446A (en) | 2019-09-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN113563323B (en) | Benzothiazolyl biaryl compound, preparation method and application | |
| JP7453989B2 (en) | Fused tricyclic compounds useful as anticancer agents | |
| CN111295384B (en) | Bicyclic derivative inhibitor, its preparation method and application | |
| CN110267954B (en) | Aniline-substituted 1,2-dihydropyrrolo[3,4-c]pyridine/pyrimidin-3-one derivatives and their uses | |
| AU2016250976B2 (en) | Imidazo isoindole derivative, preparation method therefor and medical use thereof | |
| KR20220102622A (en) | ROCK inhibitors and methods for their preparation and use | |
| CN111601804B (en) | Nitrogen-containing heteroaromatic derivative regulator, preparation method and application thereof | |
| CN110117284B (en) | Nitrogen-containing heterocyclic compound and preparation method and application thereof | |
| CN120265634A (en) | Triazole compound, preparation method and medical use thereof | |
| TWI685498B (en) | New macrocyclic compounds, a process for their preparation and pharmaceutical compositions containing them | |
| CA2890876A1 (en) | N-pyrrolidinyl, n'-pyrazolyl- urea, thiourea, guanidine and cyanoguanidine compounds as trka kinase inhibitors | |
| JP2010523639A (en) | Pharmaceutical compounds | |
| CN115038707B (en) | Azapine condensed ring compounds and their medical uses | |
| CN118994158A (en) | Nitrogen-containing heterocyclic compound, preparation method and application | |
| JP2021521214A (en) | Selective JAK2 inhibitors and their applications | |
| EP3694860A1 (en) | Pyrrolotriazine compounds and methods of inhibiting tam kinases | |
| KR102149734B1 (en) | Novel 5H-pyrroro[2,3-d]pyrimidin-6(7H)-one derivative | |
| CN110256446B (en) | Benzo-heterocycle substituted cyclopenta [4,5] pyrrolopyrazine-1-one derivatives and application thereof | |
| CN119823149A (en) | Tricyclic derivative inhibitor, preparation method and application thereof | |
| EP3978500B1 (en) | Process for the preparation of tricyclic pi3k inhibitor compounds | |
| TW202404590A (en) | Egfr inhibitors | |
| WO2020233645A1 (en) | Macrolide derivatives, preparation method and application thereof | |
| CN114423763A (en) | Regioselective synthesis of imidazo [1,2-a ] pyrimidines | |
| CN115867557B (en) | Azapine condensed ring compounds and their medical uses | |
| AU2022305807B2 (en) | Pyrimidine compounds for use as map4k1 inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |