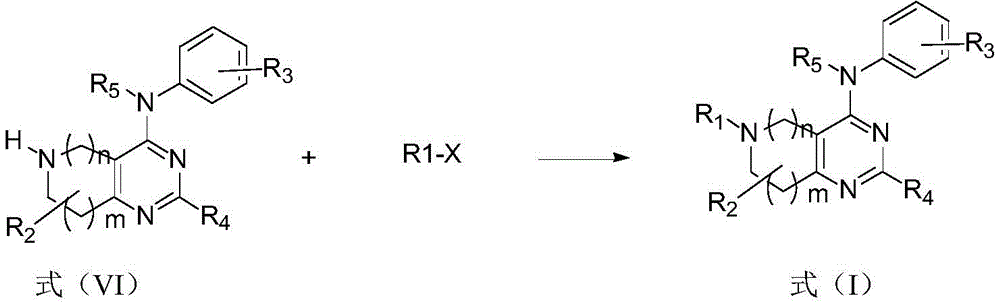CN110256420B - 四氢吡啶并嘧啶衍生物及其制备方法和用途 - Google Patents
四氢吡啶并嘧啶衍生物及其制备方法和用途 Download PDFInfo
- Publication number
- CN110256420B CN110256420B CN201910382406.3A CN201910382406A CN110256420B CN 110256420 B CN110256420 B CN 110256420B CN 201910382406 A CN201910382406 A CN 201910382406A CN 110256420 B CN110256420 B CN 110256420B
- Authority
- CN
- China
- Prior art keywords
- pyrimidin
- amino
- fluoro
- methyl
- dihydropyrido
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- FHLXQXCQSUICIN-UHFFFAOYSA-N 1,2,3,4-tetrahydropyrido[3,2-d]pyrimidine Chemical class C1=CC=C2NCNCC2=N1 FHLXQXCQSUICIN-UHFFFAOYSA-N 0.000 title claims abstract description 20
- 238000002360 preparation method Methods 0.000 title abstract description 48
- 239000003814 drug Substances 0.000 claims abstract description 16
- 206010012601 diabetes mellitus Diseases 0.000 claims abstract description 9
- 208000030159 metabolic disease Diseases 0.000 claims abstract description 9
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 8
- 101000996752 Homo sapiens Glucose-dependent insulinotropic receptor Proteins 0.000 claims abstract description 7
- 208000008589 Obesity Diseases 0.000 claims abstract description 7
- 235000020824 obesity Nutrition 0.000 claims abstract description 7
- 102100033839 Glucose-dependent insulinotropic receptor Human genes 0.000 claims abstract description 6
- 150000001875 compounds Chemical class 0.000 claims description 79
- JFDZBHWFFUWGJE-UHFFFAOYSA-N benzonitrile Chemical compound N#CC1=CC=CC=C1 JFDZBHWFFUWGJE-UHFFFAOYSA-N 0.000 claims description 75
- -1 6- (2- (5-phenyl-1, 3, 4-oxadiazol-2-yl) ethyl) -5,6,7, 8-tetrahydropyrido [4,3-d ] pyrimidine Chemical compound 0.000 claims description 64
- 238000004519 manufacturing process Methods 0.000 claims description 45
- 238000006243 chemical reaction Methods 0.000 claims description 17
- 150000003839 salts Chemical class 0.000 claims description 15
- 229910052736 halogen Inorganic materials 0.000 claims description 14
- 150000002367 halogens Chemical group 0.000 claims description 14
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 14
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical compound NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 claims description 12
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 11
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 10
- 239000003960 organic solvent Substances 0.000 claims description 10
- 125000004527 pyrimidin-4-yl group Chemical group N1=CN=C(C=C1)* 0.000 claims description 8
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 7
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 claims description 6
- 125000004483 piperidin-3-yl group Chemical group N1CC(CCC1)* 0.000 claims description 6
- LYPMLDSUKABGGI-INIZCTEOSA-N (2S)-1-[2-[4-(4-cyano-2-fluoroanilino)-2-methyl-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-7-yl]acetyl]pyrrolidine-2-carbonitrile Chemical compound C(#N)C1=CC(=C(C=C1)NC=1C2=C(N=C(N=1)C)CN(CC2)CC(=O)N1[C@@H](CCC1)C#N)F LYPMLDSUKABGGI-INIZCTEOSA-N 0.000 claims description 5
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 5
- SLVNKDYKIANJLL-UHFFFAOYSA-N 2-[4-(2-fluoro-4-methylsulfonylanilino)-7,8-dihydro-5H-pyrido[4,3-d]pyrimidin-6-yl]-1-(1,3-thiazolidin-3-yl)ethanone Chemical compound FC1=C(C=CC(=C1)S(=O)(=O)C)NC=1C2=C(N=CN=1)CCN(C2)CC(=O)N1CSCC1 SLVNKDYKIANJLL-UHFFFAOYSA-N 0.000 claims description 5
- 208000001145 Metabolic Syndrome Diseases 0.000 claims description 5
- 201000000690 abdominal obesity-metabolic syndrome Diseases 0.000 claims description 5
- 201000010099 disease Diseases 0.000 claims description 5
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 5
- YIKOECYQDIUGKZ-HNNXBMFYSA-N (2S)-1-[2-[4-(2-fluoro-4-methylsulfonylanilino)-2-methyl-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-7-yl]acetyl]pyrrolidine-2-carbonitrile Chemical compound FC1=C(C=CC(=C1)S(=O)(=O)C)NC=1C2=C(N=C(N=1)C)CN(CC2)CC(=O)N1[C@@H](CCC1)C#N YIKOECYQDIUGKZ-HNNXBMFYSA-N 0.000 claims description 4
- GOXKWSYEEILJDU-UHFFFAOYSA-N 3-fluoro-4-[[2-methyl-6-[(5-phenyl-1,3,4-oxadiazol-2-yl)methyl]-7,8-dihydro-5H-pyrido[4,3-d]pyrimidin-4-yl]amino]benzonitrile Chemical compound FC=1C=C(C#N)C=CC=1NC=1C2=C(N=C(N=1)C)CCN(C2)CC=1OC(=NN=1)C1=CC=CC=C1 GOXKWSYEEILJDU-UHFFFAOYSA-N 0.000 claims description 4
- YNQYGMYFHPQTPN-UHFFFAOYSA-N 3-fluoro-4-[[2-methyl-6-[2-(5-phenyl-1,3,4-oxadiazol-2-yl)ethyl]-7,8-dihydro-5H-pyrido[4,3-d]pyrimidin-4-yl]amino]benzonitrile Chemical compound FC=1C=C(C#N)C=CC=1NC=1C2=C(N=C(N=1)C)CCN(C2)CCC=1OC(=NN=1)C1=CC=CC=C1 YNQYGMYFHPQTPN-UHFFFAOYSA-N 0.000 claims description 4
- LUNWBOORMIHNEQ-UHFFFAOYSA-N 3-fluoro-4-[[2-methyl-7-[2-(5-phenyl-1,3,4-oxadiazol-2-yl)ethyl]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-4-yl]amino]benzonitrile Chemical compound FC=1C=C(C#N)C=CC=1NC=1C2=C(N=C(N=1)C)CN(CC2)CCC=1OC(=NN=1)C1=CC=CC=C1 LUNWBOORMIHNEQ-UHFFFAOYSA-N 0.000 claims description 4
- GHLNHJZJGUAHBX-UHFFFAOYSA-N N-(2-fluoro-4-methylsulfonylphenyl)-2-methyl-6-[(5-phenyl-1,3,4-oxadiazol-2-yl)methyl]-7,8-dihydro-5H-pyrido[4,3-d]pyrimidin-4-amine Chemical compound FC1=C(C=CC(=C1)S(=O)(=O)C)NC=1C2=C(N=C(N=1)C)CCN(C2)CC=1OC(=NN=1)C1=CC=CC=C1 GHLNHJZJGUAHBX-UHFFFAOYSA-N 0.000 claims description 4
- RYYGFXRAEXOVSH-UHFFFAOYSA-N N-(2-fluoro-4-methylsulfonylphenyl)-2-methyl-6-[2-(5-phenyl-1,3,4-oxadiazol-2-yl)ethyl]-7,8-dihydro-5H-pyrido[4,3-d]pyrimidin-4-amine Chemical compound FC1=C(C=CC(=C1)S(=O)(=O)C)NC=1C2=C(N=C(N=1)C)CCN(C2)CCC=1OC(=NN=1)C1=CC=CC=C1 RYYGFXRAEXOVSH-UHFFFAOYSA-N 0.000 claims description 4
- BVXWQFYLPYERRD-UHFFFAOYSA-N N-(2-fluoro-4-methylsulfonylphenyl)-2-methyl-7-[(5-phenyl-1,3,4-oxadiazol-2-yl)methyl]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-4-amine Chemical compound FC1=C(C=CC(=C1)S(=O)(=O)C)NC=1C2=C(N=C(N=1)C)CN(CC2)CC=1OC(=NN=1)C1=CC=CC=C1 BVXWQFYLPYERRD-UHFFFAOYSA-N 0.000 claims description 4
- WMTNNZSZKYHYPK-UHFFFAOYSA-N N-(2-fluoro-4-methylsulfonylphenyl)-2-methyl-7-[2-(5-phenyl-1,3,4-oxadiazol-2-yl)ethyl]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-4-amine Chemical compound FC1=C(C=CC(=C1)S(=O)(=O)C)NC=1C2=C(N=C(N=1)C)CN(CC2)CCC=1OC(=NN=1)C1=CC=CC=C1 WMTNNZSZKYHYPK-UHFFFAOYSA-N 0.000 claims description 4
- 239000003937 drug carrier Substances 0.000 claims description 4
- 239000000203 mixture Substances 0.000 claims description 4
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 4
- RFQYDLZSQOUNNW-UHFFFAOYSA-N 3-fluoro-4-[[2-methyl-7-[(5-phenyl-1,3,4-oxadiazol-2-yl)methyl]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-4-yl]amino]benzonitrile Chemical compound FC=1C=C(C#N)C=CC=1NC=1C2=C(N=C(N=1)C)CN(CC2)CC=1OC(=NN=1)C1=CC=CC=C1 RFQYDLZSQOUNNW-UHFFFAOYSA-N 0.000 claims description 3
- BFVGBDFGFOMOBD-UHFFFAOYSA-N 3-fluoro-4-[[2-methyl-7-[2-oxo-2-(1,3-thiazolidin-3-yl)ethyl]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-4-yl]amino]benzonitrile Chemical compound FC=1C=C(C#N)C=CC=1NC=1C2=C(N=C(N=1)C)CN(CC2)CC(N1CSCC1)=O BFVGBDFGFOMOBD-UHFFFAOYSA-N 0.000 claims description 3
- QXWDMFIMQAPVKX-UHFFFAOYSA-N C(#N)C1=CC(=C(C=C1)NC=1C2=C(N=C(N1)C)CCN=C2)F Chemical compound C(#N)C1=CC(=C(C=C1)NC=1C2=C(N=C(N1)C)CCN=C2)F QXWDMFIMQAPVKX-UHFFFAOYSA-N 0.000 claims description 3
- LDBQPFWCZHXELI-UHFFFAOYSA-N C(#N)C1=CC(=C(C=C1)NC=1C2=C(N=CN1)CCN=C2)F Chemical compound C(#N)C1=CC(=C(C=C1)NC=1C2=C(N=CN1)CCN=C2)F LDBQPFWCZHXELI-UHFFFAOYSA-N 0.000 claims description 3
- FHSVWTHLXJWZIJ-UHFFFAOYSA-N N-(2-fluoro-4-methylsulfonylphenyl)-6-[2-(5-phenyl-1,3,4-oxadiazol-2-yl)ethyl]-7,8-dihydro-5H-pyrido[4,3-d]pyrimidin-4-amine Chemical compound FC1=C(C=CC(=C1)S(=O)(=O)C)NC=1C2=C(N=CN=1)CCN(C2)CCC=1OC(=NN=1)C1=CC=CC=C1 FHSVWTHLXJWZIJ-UHFFFAOYSA-N 0.000 claims description 3
- QOPVNWQGBQYBBP-UHFFFAOYSA-N chloroethyl chloroformate Chemical compound CC(Cl)OC(Cl)=O QOPVNWQGBQYBBP-UHFFFAOYSA-N 0.000 claims description 3
- 229910052739 hydrogen Inorganic materials 0.000 claims description 3
- HHVIBTZHLRERCL-UHFFFAOYSA-N sulfonyldimethane Chemical group CS(C)(=O)=O HHVIBTZHLRERCL-UHFFFAOYSA-N 0.000 claims description 3
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 3
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N N-phenyl amine Natural products NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 claims description 2
- 150000001448 anilines Chemical class 0.000 claims description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 2
- 238000010626 work up procedure Methods 0.000 claims description 2
- ALSCEGDXFJIYES-UHFFFAOYSA-N pyrrolidine-2-carbonitrile Chemical compound N#CC1CCCN1 ALSCEGDXFJIYES-UHFFFAOYSA-N 0.000 claims 2
- 229940079593 drug Drugs 0.000 abstract description 9
- 230000002401 inhibitory effect Effects 0.000 abstract description 6
- 230000009977 dual effect Effects 0.000 abstract description 3
- 230000001270 agonistic effect Effects 0.000 abstract description 2
- 230000002265 prevention Effects 0.000 abstract 1
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 94
- 238000005160 1H NMR spectroscopy Methods 0.000 description 49
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 27
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 16
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 12
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 11
- 230000000694 effects Effects 0.000 description 11
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 9
- 125000000217 alkyl group Chemical group 0.000 description 8
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 8
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 6
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 6
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 6
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- 108010004460 Gastric Inhibitory Polypeptide Proteins 0.000 description 6
- 102100039994 Gastric inhibitory polypeptide Human genes 0.000 description 6
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 6
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 6
- 239000007787 solid Substances 0.000 description 6
- 239000000243 solution Substances 0.000 description 6
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 5
- 210000004027 cell Anatomy 0.000 description 5
- 238000001704 evaporation Methods 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- AJJISMLYIMQAKP-OAHLLOKOSA-N 5-[4-[(2r)-4-(3-fluoro-4-methylsulfonylphenoxy)butan-2-yl]piperidin-1-yl]-3-propan-2-yl-1,2,4-oxadiazole Chemical compound CC(C)C1=NOC(N2CCC(CC2)[C@H](C)CCOC=2C=C(F)C(=CC=2)S(C)(=O)=O)=N1 AJJISMLYIMQAKP-OAHLLOKOSA-N 0.000 description 4
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 4
- 101710198884 GATA-type zinc finger protein 1 Proteins 0.000 description 4
- 229940100607 GPR119 agonist Drugs 0.000 description 4
- DTHNMHAUYICORS-KTKZVXAJSA-N Glucagon-like peptide 1 Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(N)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CC=1N=CNC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)C1=CC=CC=C1 DTHNMHAUYICORS-KTKZVXAJSA-N 0.000 description 4
- 102400000322 Glucagon-like peptide 1 Human genes 0.000 description 4
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 4
- 102000004877 Insulin Human genes 0.000 description 4
- 108090001061 Insulin Proteins 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical group C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 4
- 239000004480 active ingredient Substances 0.000 description 4
- 125000003545 alkoxy group Chemical group 0.000 description 4
- 125000003118 aryl group Chemical group 0.000 description 4
- 239000008280 blood Substances 0.000 description 4
- 210000004369 blood Anatomy 0.000 description 4
- 238000001035 drying Methods 0.000 description 4
- 239000008103 glucose Substances 0.000 description 4
- 229940125396 insulin Drugs 0.000 description 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 4
- 238000010898 silica gel chromatography Methods 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- OFOBLEOULBTSOW-UHFFFAOYSA-N Malonic acid Chemical compound OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 3
- 229920002472 Starch Polymers 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 210000000227 basophil cell of anterior lobe of hypophysis Anatomy 0.000 description 3
- 239000000872 buffer Substances 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 239000012043 crude product Substances 0.000 description 3
- 125000001072 heteroaryl group Chemical group 0.000 description 3
- 125000000623 heterocyclic group Chemical group 0.000 description 3
- 230000005764 inhibitory process Effects 0.000 description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 102000005962 receptors Human genes 0.000 description 3
- 108020003175 receptors Proteins 0.000 description 3
- 230000001603 reducing effect Effects 0.000 description 3
- 239000008107 starch Substances 0.000 description 3
- 235000019698 starch Nutrition 0.000 description 3
- 208000001072 type 2 diabetes mellitus Diseases 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- WSLDOOZREJYCGB-UHFFFAOYSA-N 1,2-Dichloroethane Chemical group ClCCCl WSLDOOZREJYCGB-UHFFFAOYSA-N 0.000 description 2
- STXKJIIHKFGUCY-UHFFFAOYSA-N 5,6,7,8-tetrahydropyrido[4,3-d]pyrimidine Chemical compound C1=NC=C2CNCCC2=N1 STXKJIIHKFGUCY-UHFFFAOYSA-N 0.000 description 2
- AYJRTVVIBJSSKN-UHFFFAOYSA-N 5-[4-[[6-(4-methylsulfonylphenyl)pyridin-3-yl]oxymethyl]piperidin-1-yl]-3-propan-2-yl-1,2,4-oxadiazole Chemical compound CC(C)C1=NOC(N2CCC(COC=3C=NC(=CC=3)C=3C=CC(=CC=3)S(C)(=O)=O)CC2)=N1 AYJRTVVIBJSSKN-UHFFFAOYSA-N 0.000 description 2
- 125000000041 C6-C10 aryl group Chemical group 0.000 description 2
- XZMCDFZZKTWFGF-UHFFFAOYSA-N Cyanamide Chemical group NC#N XZMCDFZZKTWFGF-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 208000013016 Hypoglycemia Diseases 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- CEUZLEBLJQSQRZ-UHFFFAOYSA-N N-(2-fluoro-4-methylsulfonylphenyl)-5,6,7,8-tetrahydropyrido[4,3-d]pyrimidin-4-amine Chemical compound FC1=C(C=CC(=C1)S(=O)(=O)C)NC=1C2=C(N=CN=1)CCNC2 CEUZLEBLJQSQRZ-UHFFFAOYSA-N 0.000 description 2
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 239000004698 Polyethylene Substances 0.000 description 2
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 2
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 2
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 2
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 230000008484 agonism Effects 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- 239000001768 carboxy methyl cellulose Substances 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 239000003086 colorant Substances 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- 229940090124 dipeptidyl peptidase 4 (dpp-4) inhibitors for blood glucose lowering Drugs 0.000 description 2
- 239000000796 flavoring agent Substances 0.000 description 2
- 125000005842 heteroatom Chemical group 0.000 description 2
- 230000002218 hypoglycaemic effect Effects 0.000 description 2
- 239000000859 incretin Substances 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 210000003734 kidney Anatomy 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 239000006166 lysate Substances 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 150000007522 mineralic acids Chemical class 0.000 description 2
- 239000013642 negative control Substances 0.000 description 2
- 239000006186 oral dosage form Substances 0.000 description 2
- 150000007524 organic acids Chemical class 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- XNGIFLGASWRNHJ-UHFFFAOYSA-N phthalic acid Chemical compound OC(=O)C1=CC=CC=C1C(O)=O XNGIFLGASWRNHJ-UHFFFAOYSA-N 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 239000013641 positive control Substances 0.000 description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 2
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 2
- 239000012312 sodium hydride Substances 0.000 description 2
- 229910000104 sodium hydride Inorganic materials 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 239000003826 tablet Substances 0.000 description 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 2
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- VBBFIBMVFSUUBP-MERQFXBCSA-N (2S)-1-(2-aminoacetyl)-N-(4-nitrophenyl)pyrrolidine-2-carboxamide hydrochloride Chemical compound Cl.NCC(=O)N1CCC[C@H]1C(=O)NC1=CC=C([N+]([O-])=O)C=C1 VBBFIBMVFSUUBP-MERQFXBCSA-N 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- UDFTYHXUNLWFLI-UHFFFAOYSA-N 2-chloro-1-(1,3-thiazolidin-3-yl)ethanone Chemical compound ClCC(=O)N1CCSC1 UDFTYHXUNLWFLI-UHFFFAOYSA-N 0.000 description 1
- UGUIBNHHDIEZJI-UHFFFAOYSA-N 2-fluoro-4-methylsulfonylaniline Chemical compound CS(=O)(=O)C1=CC=C(N)C(F)=C1 UGUIBNHHDIEZJI-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- CNADQFDROXAKOX-UHFFFAOYSA-N 3-fluoro-4-[(2-methyl-5,6,7,8-tetrahydropyrido[3,4-d]pyrimidin-4-yl)amino]benzonitrile Chemical compound FC=1C=C(C#N)C=CC=1NC=1C2=C(N=C(N=1)C)CNCC2 CNADQFDROXAKOX-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- JHRSWWWDHNIWPJ-UHFFFAOYSA-N 4-[(6-benzyl-2-methyl-7,8-dihydro-5H-pyrido[4,3-d]pyrimidin-4-yl)amino]-3-fluorobenzonitrile Chemical compound C(C1=CC=CC=C1)N1CC2=C(N=C(N=C2NC2=C(C=C(C#N)C=C2)F)C)CC1 JHRSWWWDHNIWPJ-UHFFFAOYSA-N 0.000 description 1
- GIIHVBDGTQUKQM-UHFFFAOYSA-N 4-[(7-benzyl-2-methyl-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-4-yl)amino]-3-fluorobenzonitrile Chemical compound C(C1=CC=CC=C1)N1CC=2N=C(N=C(C=2CC1)NC1=C(C=C(C#N)C=C1)F)C GIIHVBDGTQUKQM-UHFFFAOYSA-N 0.000 description 1
- RLMBRRQWBTWGMB-UHFFFAOYSA-N 4-amino-3-fluorobenzonitrile Chemical compound NC1=CC=C(C#N)C=C1F RLMBRRQWBTWGMB-UHFFFAOYSA-N 0.000 description 1
- WIBCTLGIHZHLRI-UHFFFAOYSA-N 6-benzyl-7,8-dihydro-5h-pyrido[4,3-d]pyrimidine Chemical compound C1CC2=NC=NC=C2CN1CC1=CC=CC=C1 WIBCTLGIHZHLRI-UHFFFAOYSA-N 0.000 description 1
- NFVMTDIDLKAGHC-UHFFFAOYSA-N 6-benzyl-N-(2-fluoro-4-methylsulfonylphenyl)-2-methyl-7,8-dihydro-5H-pyrido[4,3-d]pyrimidin-4-amine Chemical compound C(C1=CC=CC=C1)N1CC2=C(N=C(N=C2NC2=C(C=C(C=C2)S(=O)(=O)C)F)C)CC1 NFVMTDIDLKAGHC-UHFFFAOYSA-N 0.000 description 1
- YCIRNNCJFXJIIF-UHFFFAOYSA-N 6-benzyl-N-(2-fluoro-4-methylsulfonylphenyl)-7,8-dihydro-5H-pyrido[4,3-d]pyrimidin-4-amine Chemical compound C(C1=CC=CC=C1)N1CC2=C(N=CN=C2NC2=C(C=C(C=C2)S(=O)(=O)C)F)CC1 YCIRNNCJFXJIIF-UHFFFAOYSA-N 0.000 description 1
- WRAQQWXIZRXFBG-UHFFFAOYSA-N 7-benzyl-N-(2-fluoro-4-methylsulfonylphenyl)-2-methyl-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-4-amine Chemical compound C(C1=CC=CC=C1)N1CC=2N=C(N=C(C=2CC1)NC1=C(C=C(C=C1)S(=O)(=O)C)F)C WRAQQWXIZRXFBG-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 1
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical compound NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 206010067482 No adverse event Diseases 0.000 description 1
- AHLPHDHHMVZTML-UHFFFAOYSA-N Orn-delta-NH2 Natural products NCCCC(N)C(O)=O AHLPHDHHMVZTML-UHFFFAOYSA-N 0.000 description 1
- UTJLXEIPEHZYQJ-UHFFFAOYSA-N Ornithine Natural products OC(=O)C(C)CCCN UTJLXEIPEHZYQJ-UHFFFAOYSA-N 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 235000011054 acetic acid Nutrition 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 108060000200 adenylate cyclase Proteins 0.000 description 1
- 102000030621 adenylate cyclase Human genes 0.000 description 1
- 239000003463 adsorbent Substances 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 235000010419 agar Nutrition 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 230000000172 allergic effect Effects 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- SNAAJJQQZSMGQD-UHFFFAOYSA-N aluminum magnesium Chemical compound [Mg].[Al] SNAAJJQQZSMGQD-UHFFFAOYSA-N 0.000 description 1
- 235000001014 amino acid Nutrition 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 229940127003 anti-diabetic drug Drugs 0.000 description 1
- 239000003472 antidiabetic agent Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 208000010668 atopic eczema Diseases 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-N benzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-N 0.000 description 1
- 229940092714 benzenesulfonic acid Drugs 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical compound OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 230000009693 chronic damage Effects 0.000 description 1
- 235000015165 citric acid Nutrition 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 208000002173 dizziness Diseases 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 239000006196 drop Substances 0.000 description 1
- 230000000857 drug effect Effects 0.000 description 1
- 230000004064 dysfunction Effects 0.000 description 1
- 230000002526 effect on cardiovascular system Effects 0.000 description 1
- 238000005538 encapsulation Methods 0.000 description 1
- 125000004185 ester group Chemical group 0.000 description 1
- 150000002148 esters Chemical group 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 1
- 210000001508 eye Anatomy 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 239000012634 fragment Substances 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 230000005182 global health Effects 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 210000002216 heart Anatomy 0.000 description 1
- 238000002868 homogeneous time resolved fluorescence Methods 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 229940071870 hydroiodic acid Drugs 0.000 description 1
- 230000003345 hyperglycaemic effect Effects 0.000 description 1
- 239000005457 ice water Substances 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 230000003914 insulin secretion Effects 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 239000007927 intramuscular injection Substances 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 239000007928 intraperitoneal injection Substances 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 210000004153 islets of langerhan Anatomy 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- SFDJOSRHYKHMOK-UHFFFAOYSA-N nitramide Chemical group N[N+]([O-])=O SFDJOSRHYKHMOK-UHFFFAOYSA-N 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 229960003104 ornithine Drugs 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- NTTOTNSKUYCDAV-UHFFFAOYSA-N potassium hydride Chemical compound [KH] NTTOTNSKUYCDAV-UHFFFAOYSA-N 0.000 description 1
- 229910000105 potassium hydride Inorganic materials 0.000 description 1
- BDAWXSQJJCIFIK-UHFFFAOYSA-N potassium methoxide Chemical compound [K+].[O-]C BDAWXSQJJCIFIK-UHFFFAOYSA-N 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 229960004115 sitagliptin phosphate Drugs 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- UIIMBOGNXHQVGW-UHFFFAOYSA-M sodium bicarbonate Substances [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 239000012453 solvate Substances 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 239000008174 sterile solution Substances 0.000 description 1
- 239000008223 sterile water Substances 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 125000001174 sulfone group Chemical group 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- RTZRUVMEWWPNRR-UHFFFAOYSA-N tert-butyl n-(3-iodo-1h-pyrrolo[2,3-b]pyridin-5-yl)carbamate Chemical compound CC(C)(C)OC(=O)NC1=CN=C2NC=C(I)C2=C1 RTZRUVMEWWPNRR-UHFFFAOYSA-N 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 230000004584 weight gain Effects 0.000 description 1
- 235000019786 weight gain Nutrition 0.000 description 1
- UGOMMVLRQDMAQQ-UHFFFAOYSA-N xphos Chemical compound CC(C)C1=CC(C(C)C)=CC(C(C)C)=C1C1=CC=CC=C1P(C1CCCCC1)C1CCCCC1 UGOMMVLRQDMAQQ-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B2200/00—Indexing scheme relating to specific properties of organic compounds
- C07B2200/07—Optical isomers
Landscapes
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Diabetes (AREA)
- General Chemical & Material Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Child & Adolescent Psychology (AREA)
- Emergency Medicine (AREA)
- Endocrinology (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
本发明涉及一种四氢吡啶并嘧啶衍生物及其制备方法和用途,属于医药化学领域,该四氢吡啶并嘧啶衍生物具有式(I)的结构,该衍生物具有良好的GPR119激动活性和DPP‑4抑制活性的双重作用,可以作为抗糖尿病、代谢性疾病和预防代谢失调以及肥胖的药物。本发明还提供该四氢吡啶并嘧啶衍生物的制备方法和包含作为活性组分的该四氢吡啶并嘧啶衍生物的药物组合物。
Description
技术领域
本发明涉及四氢吡啶并嘧啶衍生物及其制备方法和在制备治疗抗糖尿病、代谢性疾病、肥胖症药物中的应用,属于化学药物领域。
背景技术
糖尿病是近年来被认为影响全球主要健康问题的疾病之一,长期处于高血糖的状态,易导致各种组织,尤其是眼、肾、心脏、血管、神经的慢性损害、功能障碍。更严重的会引起心脑血管并发症。因此,严格控制血糖是降低2型糖尿病发病率的重中之重。根据IDF最新报告数据,到2017年全球糖尿病患者约有4.25亿,估计在2045年患病人数将会达到6.29亿。目前临床使用的抗糖尿病药物随着时间的推移逐渐表现出疗效下降,耐受性不好,依从性差等许多副作用。因此需要新的策略来持续控制血糖,避免低血糖和其他的副作用。
GPR119激动剂能够活化腺苷酸环化酶,提升cAMP的水平,这促进了GLP-1、葡萄糖依赖性促胰岛素释放多肽(glucose-dependent insulinotropic peptide,GIP)和胰岛素的释放。同时,GLP-1和GIP可与β细胞的受体结合,进一步诱导胰岛素的释放,因而GPR119激动剂通过两种直接的作用机制提升胰岛素的释放;GLP-1(或是GIP)也能够保持胰岛β细胞活力。因此口服GPR119激动剂不但可以促进胰岛素的分泌,而且可以保护β细胞活力,这就有效的改善了2型糖尿病人体内的葡萄糖平衡。GPR119激动剂具有应用在治疗糖尿病和相关联症状上的价值,尤其是对于2型糖尿病,肥胖,葡萄糖耐受不良,胰岛素抵抗,代谢综合症,高血脂,血胆脂醇过多,以及动脉硬化症。
DPP-4抑制剂通过抑制内源性GLP-1和葡萄糖依赖性促胰岛素释放多肽(glucose-dependent insulinotropic peptide,GIP)这两种肠促胰岛素(incretin)的降解,促进葡萄糖依赖性胰岛素分泌作用。临床结果显示,DPP-4抑制剂具有较好的降糖效果,且未发现有其他降糖药物出现的体重增加或低血糖等不良反应。
尽管目前已经公开了一系列的治疗糖尿病和代谢性疾病等的化合物,但仍需努力开发新的具有更好的药效的化合物,尤其是开发能够通过多个介导途径的药物。本发明公开了一系列具有双重作用的基于GPR119激动作用和DPP-4抑制作用的四氢吡啶并嘧啶类化合物及其制备方法和用途。
发明内容
本发明提供了四氢吡啶并嘧啶衍生物、其制备方法及含有该衍生物的药物组合物以及在药学上可接受的盐作为治疗糖尿病尤其是2型糖尿病和代谢综合症的药物及其预防代谢失调和肥胖的药物,该四氢吡啶并嘧啶衍生物具有双重作用的基于GPR119激动作用和DPP-4抑制作用的效果。
该四氢吡啶并嘧啶衍生物具有式(I)的结构:
其中:R1选自(CH2)q-R6;
R2、R3、R4、R5选自下列基团:氢原子、卤素、羟基、硝基、胺基、羧基、氰基、烷基、烷氧基、酯基、砜基、被卤素或羟基或氰基取代的烷基、被烷基取代的胺基或被卤素或羟基或氰基取代的烷氧基;
n、m选自0-4;
R6选自取代或未取代的芳基、取代或未取代的杂芳基、取代或未取代的杂环基或-CO-R7;
R7选自取代或未取代的芳基、取代或未取代的杂芳基、取代或未取代的杂环基;
q选自1-6;
进一步地,芳基优选C6-C10芳基;杂芳基优选含1-6个N、O或S杂原子的C2-8芳基,杂环基优选1-6个N、O或S杂原子的C2-8环基;
取代基优选卤素、羟基、硝基、胺基、羧基、氰基、烷基、烷氧基、酯基、被卤素或羟基或氰基取代的烷基、被烷基取代的胺基或被卤素或羟基或氰基取代的烷氧基、酰胺基或C6-10芳基;
烷基优选C1-6烷基;
R2、R3、R4、R5优选氢原子、卤素、氰基、烷基、被卤素取代的烷基、甲基砜基(Ms);
n和m优选0-2;
q优选1-3;
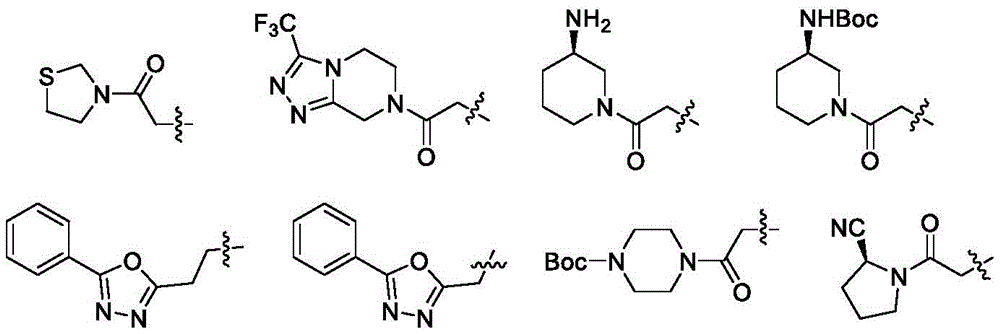
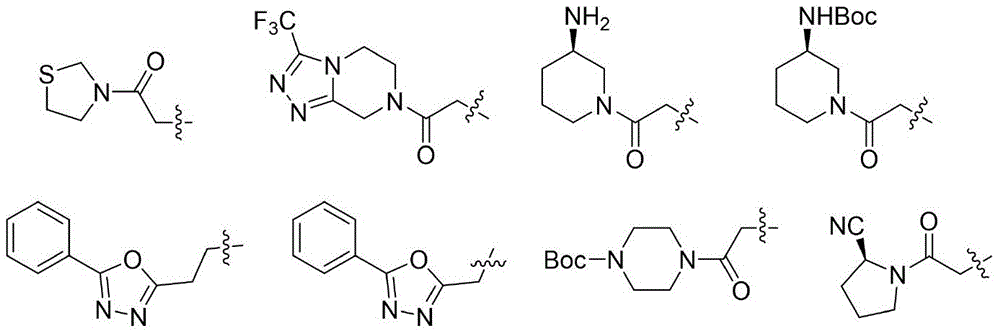
进一步地:R1优选下列基团:
R2为氢原子;
R3为氢原子或卤素或氰基或甲基砜基(Ms);
R4为氢原子或C1-6烷基,优选甲基;
R5为氢原子;
进一步地,式(I)化合物为式(II)结构化合物
式(II)化合物
式(I)化合物为式(III)结构化合物
进一步地,式(I)化合物优选为如下化合物:
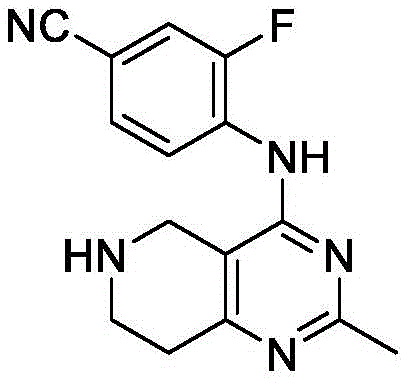
1.3-氟-4-((6-(2-氧代-2-噻唑烷-3-乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈
2.3-氟-4-((6-(2-氧代-2-(3-(三氟甲基)-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈
3.叔丁基(R)-(1-(2-(4-((4-氰基-2-氟苯基)氨基)-7,8-二氢吡啶并[4,3-d]嘧啶)-6(5H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
4.3-氟-4-((6-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈
5.3-氟-4-((6-(2-(5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈
6.化合物2-(4-((2-氟-4-(甲砜基)苯基)氨基)-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-(噻唑烷-3-基)-1-氧代乙烷的制备
7.2-(4-((2-氟-4-(甲砜基)苯基)氨基)-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-(3-三氟甲基)-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷
8.N-(2-氟-4-甲砜基苯基)-6-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺
9.叔丁基-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)哌嗪-1-氨基甲酸酯
10.3-氟-4-((2-甲基-6-(2-氧代-2-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4基)-胺基)苯甲腈
11.3-氟-4-((2-甲基-6-((5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4基)-胺基)苯甲腈
12.叔丁基(R)-(1-(2-(4-((4-氰基-2-氟苯基)氨基)-2甲基-7,8-二氢吡啶并[4,3-d]嘧啶)-6(5H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
13.3-氟-4-((2-甲基-6-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4基)-胺基)苯甲腈
14.2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷
15.N-(2-氟-4-甲砜基苯基)-2-甲基-6-((5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺
16.叔丁基4-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)哌嗪-1-氨基甲酸酯
17.2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-噻唑烷-3-基)-1-氧代乙烷
18.叔丁基(R)-(1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
19.N-(2-氟-4-甲砜基苯基)-2-甲基-6-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺
20.(S)-1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)吡咯烷-2-甲腈
21.叔丁基4-(2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)乙酰基)哌嗪-1-氨基甲酸酯
22.(1S)-2-(2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)乙酰基)环戊基-1-甲腈
23.叔丁基(R)-(1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
24.2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)-1-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷
25.3-氟-4-((2-甲基-7-(2-氧代-2-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)乙基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈
26.3-氟-4-((2-甲基-7-((5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈
27.叔丁基4-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)哌嗪-1-氨基甲酸酯
28.3-氟-4-((2-甲基-7-(2-氧代-2-(噻唑烷-3-基)乙基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈
29.叔丁基(R)-(1-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
30.3-氟-4-((2-甲基-7-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈
31.(S)-1-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)吡咯烷-2-甲腈
32.2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)-1-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷
33.N-(2-氟-4-甲砜基苯基)-2-甲基-7-((5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-胺
34.叔丁基4-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)哌嗪-1-氨基甲酸酯
35.2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)-1-(噻唑烷-3-基)-1-氧代乙烷
36.叔丁基(R)-(1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
37.N-(2-氟-4-甲砜基苯基)-2-甲基-7-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-胺
38.(S)-1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)吡咯烷-2-甲腈
进一步地,式(I)化合物优选为下列化合物:
18.叔丁基(R)-(1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
20.(S)-1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)吡咯烷-2-甲腈
22.(1S)-2-(2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)乙酰基)环戊基-1-甲腈
23.叔丁基(R)-(1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
29.叔丁基(R)-(1-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
31.(S)-1-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)吡咯烷-2-甲腈
36.叔丁基(R)-(1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
38.(S)-1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)吡咯烷-2-甲腈
本发明还提供一种式(I)化合物的制备方法,具体包括如下步骤:
步骤(1):式(IV)化合物与取代或未取代苯胺进行反应,生成式(V)化合物
在有机溶剂中,在强碱或Pd催化剂存在下,在室温至100℃反应,反应完后处理后得到式(V)化合物。
有机溶剂优选四氢呋喃,1,4-二氧六环、N,N-二甲基甲酰胺(DMF),二甲基亚砜(DMSO)或乙腈;
强碱优选氢化钠、氢化钾、甲醇钠或甲醇钾;
反应优选在氮气保护下进行;
Pd催化剂优选Pd2(dba)3。
步骤(2):式(V)化合物脱去苄基生成式(VI)化合物
在有机溶剂中,使用1-氯乙基氯甲酸酯使式(V)脱去苄基,后处理后得到式(VI)化合物。
有机溶剂优选1,2-二氯乙烷、二氯甲烷或氯仿;
步骤(3):式(VI)化合物与R1-X反应生成式(I)化合物
其中X为卤素。
在有机溶剂中,在碱的存在下,式(VI)化合物与R1-X反应生成式(I)化合物。
有机溶剂优选N,N-二甲基甲酰胺(DMF)或二甲基亚砜(DMSO);
碱优选K2CO3,Na2CO3。
另一方面,本发明提供上述化合物的立体异构体、互变异构体、对映异构体、非对映异构体、外消旋异构体、其与酸所形成的医学上可接受的盐。
成盐的酸可以是有机酸或无机酸,例如:与盐酸、氢溴酸、氢碘酸、硫酸、硝酸、磷酸、碳酸等无机酸的盐;与甲酸、乙酸、丙酸、三氟乙酸、邻苯二甲酸、草酸、丙二酸、琥珀酸、富马酸、马来酸、乳酸、苹果酸、酒石酸、柠檬酸、苯甲酸、甲磺酸、乙磺酸、苯磺酸、对甲苯磺酸等有机酸的盐;与赖氨酸、精氨酸、鸟氨酸、谷氨酸、天冬氨酸等氨基酸的盐。
另一方面,本发明化合物或其医学上可接受的盐可以根据需要转换成溶剂化物。作为这样的溶剂,可以举出:水、甲醇、乙醇、1-丙醇、2-丙醇、丁醇、叔丁醇、乙腈、丙酮、甲基乙基酮、氯仿、乙酸乙酯、二乙醚、叔丁基甲基醚、苯、甲苯、N,N-二甲基甲酰胺(DMF)、二甲基亚砜(DMSO)等。特别可以举出:水、甲醇、乙醇、1-丙醇、2-丙醇、乙腈、丙酮、甲基乙基酮、和乙酸乙酯作为优选的溶剂。
本发明提供了一类四氢吡啶并嘧啶衍生物或其可药用的盐在制备作为治疗糖尿病药物中的用途。
本发明提供了一类四氢吡啶并嘧啶衍生物或其可药用的盐在制备代谢综合症的药物及其预防代谢失调和肥胖的药物。
本发明还提供了一种药物组合物,其含有治疗有效剂量的四氢吡啶并嘧啶衍生物或其可药用的盐及其可药用的载体或赋形物。该药物组合物用作治疗糖尿病药物。该药物组合物在制备代谢综合症的药物及其预防代谢失调和肥胖的药物。该药物组合物包含至少一种作为活性成分的本发明所述式(1)化合物和一种或多种无机或有机、固体或液体的药学上可接受的载体或者赋形剂。术语“药学上可接受的”是指当给药至动物例如哺乳动物(例如人类)时生理学上可耐受且通常不会产生过敏或类似的不良反应(例如头晕等)的添加剂或组合物。药物载体和赋形剂可以包括但不限于稀释剂,润滑剂,聚乙二醇;粘合剂,例如硅酸铝镁、淀粉、明胶、甲基纤维素、羧甲基纤维素钠和/或聚乙烯吡咯烷酮;并且,如果需要的话,还包括崩解剂,例如淀粉、琼脂、海藻酸或其盐如海藻酸钠;和/或吸附剂、着色剂、防腐剂、稳定剂、矫味剂和甜味剂。
该组合物可以被制备成药物制剂。可以将所述式(I)化合物作为活性成分混合在任何常用的口服剂型或局部(粉末、软膏或滴剂)的形式给药,包括片剂、胶囊剂和液体制剂(例如酏剂和混悬剂),其中包含着色剂、矫味剂、稳定剂和掩盖味道的物质。对于混合口服剂型来说,所述式(I)化合物作为活性成分可以与各种普通片剂材料混合以助于压片和装入胶囊。可以将所述式(1)化合物在药学上可接受的无菌液体载体例如无菌水、无菌有机溶剂或者两者的混合物中溶解或混悬。液体载体可以是适合注射剂的载体,比如生理盐水、丙二醇或者聚乙二醇水溶液。在其他情况下,还可以将微粉化的活性成分分散在淀粉或羧甲基纤维素钠的水溶液中或分散在适当的油中来制得。液体药物制剂(指无菌溶液或混悬剂)可以用于静脉注射、肌肉注射、腹膜内注射或者皮下注射。
具体实施方式
下面通过实施例对本发明给予进一步的说明,当然,本发明不仅限于下述的实施例。
实施例1:
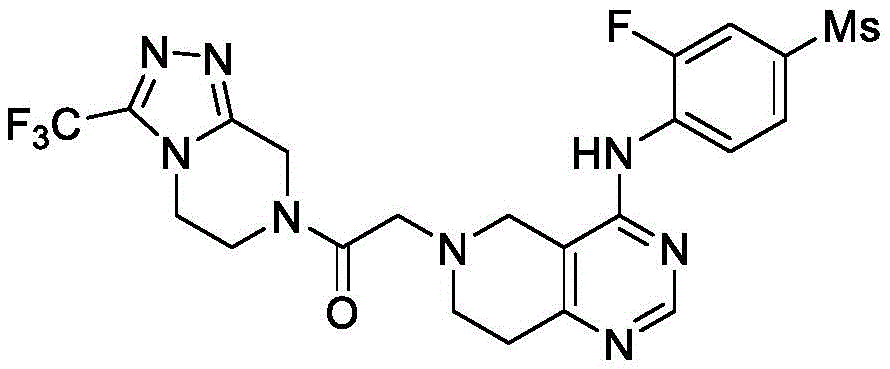
化合物3-氟-4-((6-(2-氧代-2-噻唑烷-3-乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈的制备
步骤(1):4-((6-苄基-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)-3-氟苯甲腈
在100mL两颈烧瓶中依次加入4-氯-7-苄基-5,6,7,8-四氢吡啶并[4,3-d]嘧啶(1.5g,5.8mmol),2-氟-4-氰基苯胺(0.86g,6.3mmol)和20mL四氢呋喃,氮气保护,将反应瓶降温到0℃,缓慢加入60%氢化钠(0.46g,11.5mmol)。之后反应升温至65℃,搅拌过夜反应。将反应液用乙酸乙酯萃取2次,饱和氯化钠洗涤2次,无水硫酸钠干燥,减压蒸干得到粗品。经硅胶柱层析纯化(石油醚(PE):乙酸乙酯(EA)=1:1)得0.7g黄色固体,收率35%。MS-ESI:[M+H]+:360.3。
步骤(2):3-氟-4-((5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈
将4-((6-苄基-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)-3-氟苯甲腈(0.7g)溶于1,2-二氯乙烷(10mL),在冰水浴中缓慢滴加1-氯乙基氯甲酸酯(0.3mL,2.8mmol),反应回流过夜。反应结束后将反应液蒸干,得黄色固体。将其溶于甲醇,回流反应2h。减压蒸馏除去大部分甲醇,用乙酸乙酯和饱和氯化钠萃取,取水层用饱和NaHCO3调pH至8-9,溶液用二氯甲烷萃取,无水硫酸镁干燥,蒸干,硅胶柱层析纯化得黄色固体0.4g,收率76%。1H-NMR(600MHz,CDCl3)δ8.89(t,J=8.4Hz,1H),8.70(s,1H),7.52(d,J=8.7Hz,1H),7.44(dd,J=10.9,1.8Hz,1H),6.59(d,J=4.1Hz,1H),3.96(s,2H),3.25(t,J=5.8Hz,2H),2.90(t,J=5.8Hz,2H)。
步骤(3):3-氟-4-((6-(2-氧代-2-噻唑烷-3-乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈
在10mL反应瓶中分别加入3-氟-4-((5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈(0.1g,0.4mmol),2-氯-1-(噻唑烷-3-基)-1-氧代乙烷(0.52mmol),无水碳酸钾(0.07g,0.52mmol)和N,N-二甲基甲酰胺(2mL)。反应液在70℃搅拌过夜。反应结束后,将反应液用乙酸乙酯萃取2次,饱和氯化钠洗涤2次,无水硫酸镁干燥,蒸干得到粗品。用硅胶柱层析分离纯化得黄色固体0.096g,收率65%。
1H-NMR(600MHz,CDCl3)δ8.81(t,J=8.3Hz,1H),8.69(s,1H),7.51(d,J=8.6Hz,1H),7.49-7.38(m,1H),6.59(s,1H),4.65(d,J=9.6Hz,2H),3.92(t,J=6.4Hz,1H),3.87(t,J=6.2Hz,1H),3.76(d,J=11.5Hz,2H),3.56(d,J=15.1Hz,2H),3.14(t,J=6.2Hz,1H),3.09-2.97(m,5H)。
实施例2:
3-氟-4-((6-(2-氧代-2-(3-(三氟甲基)-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈
参照实施例1的制备方法,收率75%。1H-NMR(600MHz,CDCl3)δ8.47(s,1H),8.28(m,1H),7.42(d,J=7.8Hz,1H),7.37(d,J=9.7Hz,1H),5.00(m,2H),4.15(s,2H),4.04(s,2H),3.58(m,4H),2.85(m,4H).
实施例3:
叔丁基(R)-(1-(2-(4-((4-氰基-2-氟苯基)氨基)-7,8-二氢吡啶并[4,3-d]嘧啶)-6(5H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
参照实施例1的制备方法,收率62%。1H-NMR(600MHz,CDCl3)δ8.77(t,J=8.0Hz,1H),8.67(s,1H),7.50(d,J=8.5Hz,1H),7.43(d,J=10.6Hz,1H),6.72(s,1H),4.77(d,J=5.0Hz,1H),3.86(d,J=12.7Hz,1H),3.75(m,2H),3.59(m,2H),3.59-3.49(m,2H),3.45(s,1H),3.30(m,1H),3.00(s,4H),2.01–1.88(m,2H),1.61(d,J=5.7Hz,2H),1.37(s,9H).
实施例4:
3-氟-4-((6-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈
参照实施例1的制备方法,收率54%。1H-NMR(600MHz,CDCl3)δ8.81(t,J=8.1Hz,1H),8.68(s,1H),8.10–7.98(m,2H),7.62–7.49(m,4H),7.44(dd,J=10.8,1.8Hz,1H),6.67(s,1H),3.70(s,2H),3.31(d,J=6.5Hz,2H),3.26(d,J=6.0Hz,2H),3.00(s,4H).
实施例5:
3-氟-4-((6-(2-(5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈
参照实施例1的制备方法,收率59%。1H-NMR(600MHz,CDCl3)δ8.75(t,J=8.3Hz,1H),8.69(s,1H),8.09(dd,J=8.3,1.3Hz,2H),7.63–7.47(m,4H),7.44(dd,J=10.7,1.8Hz,1H),6.65(s,1H),4.25(s,2H),3.80(s,2H),3.11(t,J=5.7Hz,2H),3.06(d,J=5.3Hz,2H).
实施例6:
化合物2-(4-((2-氟-4-(甲砜基)苯基)氨基)-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-(噻唑烷-3-基)-1-氧代乙烷的制备
步骤(1):6-苄基-N-(2-氟-4-甲砜基苯基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺
在100mL反应瓶中依次加入4-氯-7-苄基-5,6,7,8-四氢吡啶并[4,3-d]嘧啶(1.5g,5.8mmol),2-氟-4-甲砜基苯胺(1.3g,6.9mmol),Pd2(dba)3(1.2mmol),X-Phos(1.2mmol),CS2CO3(2.5mmol)和无水(30mL)。反应液氮气保护,回流过夜反应。反应结束后,用乙酸乙酯萃取反应液2次,饱和氯化钠洗涤2次,无水硫酸钠干燥,蒸干得到粗品。经硅胶柱层析纯化(PE:EA=1:1)得2g黄色固体,收率83%。MS-ESI:[M+H]+:399.3.
步骤(2)N-(2-氟-4-甲砜基苯基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺
参照实施例1步骤(2)的制备方法,收率46%。1H-NMR(600MHz,CDCl3)δ8.93(t,J=8.2Hz,1H),8.70(s,1H),7.78(dd,J=8.7,1.3Hz,1H),7.72(dd,J=10.4,2.0Hz,1H),6.62(d,J=4.4Hz,1H),3.97(s,2H),3.25(t,J=5.8Hz,2H),3.08(s,3H),2.90(t,J=5.8Hz,2H).
步骤(3):2-(4-((2-氟-4-(甲砜基)苯基)氨基)-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-(噻唑烷-3-基)-1-氧代乙烷
参照实施例1步骤(3)的制备方法,收率71%。1H-NMR(600MHz,CDCl3)δ8.85(t,J=8.1Hz,1H),8.70(s,1H),7.78(dd,J=8.7,1.6Hz,1H),7.73(dd,J=10.3,2.0Hz,1H),6.61(s,1H),4.65(d,J=9.2Hz,2H),3.92(t,J=6.4Hz,1H),3.87(t,J=6.2Hz,1H),3.77(d,J=11.7Hz,2H),3.57(d,J=15.0Hz,2H),3.14(t,J=6.2Hz,1H),3.09(s,3H),3.04(dt,J=7.6,5.2Hz,5H).
实施例7:
2-(4-((2-氟-4-(甲砜基)苯基)氨基)-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-(3-三氟甲基)-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷
参照实施例1的制备方法,收率65%。1H-NMR(600MHz,CDCl3)δ8.83(t,J=8.3Hz,1H),8.71(s,1H),7.78(d,J=8.5Hz,1H),7.73(d,J=10.1Hz,1H),6.60(s,1H),5.12(m,2H),4.23(t,J=5.4Hz,2H),4.16(m,2H),3.71(m,2H),3.66(s,2H),3.09(s,3H),3.01(s,4H).
实施例8:
N-(2-氟-4-甲砜基苯基)-6-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺
参照实施例1的制备方法,收率62%。1H-NMR(600MHz,CDCl3)δ8.89(t,J=8.2Hz,1H),8.70(s,1H),8.06(d,J=7.1Hz,2H),7.78(d,J=8.8Hz,1H),7.74(dd,J=10.3,1.8Hz,1H),7.60–7.48(m,3H),6.61(d,J=4.4Hz,1H),3.68(s,2H),3.30(t,J=6.7Hz,2H),3.24(t,J=6.7Hz,2H),3.09(s,3H),2.99(s,4H).
实施例9:
叔丁基-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)哌嗪-1-氨基甲酸酯
步骤(1):4-((6-苄基-2-甲基-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4基)-胺基)-3-氟苯甲腈
参照实施例6步骤(1)的制备方法,收率62%。MS-ESI:[M+H]+:374.5.步骤(2):3-氟-4-((2-甲基-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4基)-胺基)苯甲腈
参照实施例1步骤(2)的制备方法,收率58%。1H-NMR(600MHz,CDCl3)δ8.95(t,J=8.4Hz,1H),7.52(d,J=8.7Hz,1H),7.43(dd,J=11.0,1.7Hz,1H),6.58(d,J=4.1Hz,1H),3.94(s,2H),3.25(t,J=5.9Hz,2H),2.87(t,J=5.8Hz,2H),2.64(s,3H).
步骤(3):叔丁基-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)哌嗪-1-氨基甲酸酯
参照实施例1的制备方法,收率73%。1H-NMR(600MHz,CDCl3)δ8.88(t,J=8.4Hz,1H),7.51(d,J=8.6Hz,1H),7.43(dd,J=10.9,1.7Hz,1H),6.56(d,J=4.3Hz,1H),3.65(d,J=10.1Hz,4H),3.61–3.51(m,4H),3.46(s,4H),2.96(dd,J=10.8,4.4Hz,4H),2.63(s,3H),1.49(d,J=5.6Hz,9H).
实施例10:
化合物3-氟-4-((2-甲基-6-(2-氧代-2-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4基)-胺基)苯甲腈的制备
参照实施例1的制备方法,收率73%。1H-NMR(600MHz,CDCl3)δ8.88(t,J=7.9Hz,1H),7.52(d,J=8.5Hz,1H),7.44(d,J=10.9Hz,1H),6.52(s,1H),5.13(m,2H),4.22(s,2H),4.15(d,J=10.3Hz,2H),3.65(m,4H),2.97(m,4H),2.64(s,3H).
实施例11:
化合物3-氟-4-((2-甲基-6-((5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4基)-胺基)苯甲腈的制备
参照实施例1的制备方法,收率51%。1H-NMR(600MHz,CDCl3)δ8.86(t,J=8.4Hz,1H),8.09(d,J=7.1Hz,2H),7.56(m,4H),7.43(dd,J=10.8,1.8Hz,1H),6.56(d,J=4.3Hz,1H),4.24(s,2H),3.76(s,2H),3.09(t,J=5.8Hz,2H),3.00(t,J=5.7Hz,2H),2.63(s,3H).
实施例12:
化合物叔丁基(R)-(1-(2-(4-((4-氰基-2-氟苯基)氨基)-2甲基-7,8-二氢吡啶并[4,3-d]嘧啶)-6(5H)-基)乙酰基)哌啶-3-基)氨基甲酸酯的制备
参照实施例1的制备方法,收率66%。1H-NMR(600MHz,CDCl3)δ8.88(t,J=8.4Hz,1H),7.50(d,J=8.7Hz,1H),7.42(dd,J=10.9,1.8Hz,1H),6.55(s,1H),4.64(d,J=6.6Hz,2H),3.91(t,J=6.4Hz,1H),3.86(t,J=6.2Hz,1H),3.77–3.73(m,1H),3.73–3.67(m,2H),3.54(d,J=15.0Hz,2H),3.12(t,J=6.2Hz,1H),3.04(d,J=6.4Hz,1H),3.03–2.98(m,2H),2.96(m,2H),2.63(s,3H),1.35(s,1H),1.30(s,2H),1.29–1.22(m,10H).
实施例13:
化合物3-氟-4-((2-甲基-6-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4基)-胺基)苯甲腈的制备
参照实施例1的制备方法,收率64%。1H-NMR(600MHz,CDCl3)δ8.90(t,J=8.4Hz,1H),8.05(d,J=7.7Hz,2H),7.62–7.48(m,4H),7.43(d,J=10.9Hz,1H),6.57(d,J=3.6Hz,1H),3.63(s,2H),3.29(t,J=7.0Hz,2H),3.22(t,J=7.0Hz,2H),2.95(dd,J=8.7,4.1Hz,4H),2.62(s,3H).
实施例14:
化合物2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷的制备
步骤(1):6-苄基-N-(2-氟-4-甲砜基苯基)-2-甲基-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺
参照实施例6步骤(1)的制备方法,收率72%。1H-NMR(600MHz,CDCl3)δ9.00–8.92(m,1H),7.78(dd,J=8.7,1.4Hz,1H),7.71(dd,J=10.4,2.0Hz,1H),7.43–7.37(m,4H),7.34(dd,J=9.4,4.4Hz,1H),6.53(d,J=4.5Hz,1H),3.82(s,2H),3.54(s,2H),3.08(s,3H),2.94(t,J=5.6Hz,2H),2.87(t,J=5.7Hz,2H),2.64(s,3H).
步骤(2):N-(2-氟-4-甲砜基苯基)-2-甲基-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺
参照实施例1步骤(2)的制备方法,收率72%。1H-NMR(400MHz,CDCl3)δ9.00(t,J=8.2Hz,1H),7.78(d,J=8.7Hz,1H),7.71(d,J=10.4Hz,1H),6.59(d,J=3.9Hz,1H),3.93(s,2H),3.23(t,J=5.8Hz,2H),3.08(s,3H),2.85(t,J=5.6Hz,2H),2.63(s,3H).
步骤(3)
2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷
参照实施例1步骤(3)的制备方法,收率47%。1H-NMR(600MHz,CDCl3)δ8.92(d,J=7.1Hz,1H),7.79(d,J=8.3Hz,1H),7.72(d,J=10.1Hz,1H),6.55(s,1H),5.13(m,2H),4.22(s,2H),4.15(d,J=3.0Hz,2H),3.74–3.56(m,4H),3.09(s,3H),2.97(m,4H),2.64(s,3H).
实施例15:
化合物N-(2-氟-4-甲砜基苯基)-2-甲基-6-((5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺的制备
参照实施例1的制备方法,收率53%。1H NMR(600MHz,CDCl3)δ8.90(t,J=8.1Hz,1H),8.09(d,J=7.6Hz,2H),7.78(d,J=8.7Hz,1H),7.72(d,J=10.3Hz,1H),7.56(m,3H),6.60(s,1H),4.24(s,2H),3.78(s,2H),3.10(s,2H),3.09(s,3H),3.01(t,J=5.4Hz,2H),2.63(s,3H).
实施例16:
化合物叔丁基4-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)哌嗪-1-氨基甲酸酯的制备
参照实施例1的制备方法,收率58%。1H-NMR(600MHz,CDCl3)δ8.94(t,J=8.2Hz,1H),7.79(d,J=8.8Hz,1H),7.73(d,J=10.3Hz,1H),6.58(s,1H),3.66(m,4H),3.57(m,4H),3.46(s,4H),3.09(s,3H),2.97(m,4H),2.64(s,3H),1.49(s,9H).
实施例17:
化合物2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-噻唑烷-3-基)-1-氧代乙烷的制备
参照实施例1的制备方法,收率38%。1H-NMR(600MHz,CDCl3)δ8.92(t,J=8.1Hz,1H),7.78(dd,J=8.6,1.5Hz,1H),7.72(dd,J=10.4,2.1Hz,1H),6.61(s,1H),4.70–4.58(m,2H),3.89(m,2H),3.75–3.69(m,2H),3.55(m,2H),3.13(t,J=6.2Hz,1H),3.09(s,3H),3.08–2.95(m,5H),2.63(s,3H).
实施例18:
化合物叔丁基(R)-(1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)哌啶-3-基)氨基甲酸酯的制备
参照实施例1的制备方法,收率57%。1H-NMR(600MHz,CDCl3)δ8.92(t,J=8.1Hz,1H),7.81–7.75(m,1H),7.72(dd,J=10.3,2.0Hz,1H),6.68(s,1H),4.81(d,J=5.4Hz,1H),3.83(d,J=12.5Hz,1H),3.75–3.59(m,4H),3.53(m,3H),3.34(m,1H),3.09(s,3H),3.03–2.94(m,4H),2.63(s,3H),1.91(s,2H),1.61(m,2H),1.39(s,9H).
实施例19:
化合物N-(2-氟-4-甲砜基苯基)-2-甲基-6-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺的制备
参照实施例1的制备方法,收率65%。1H-NMR(600MHz,CDCl3)δ8.96(t,J=8.2Hz,1H),8.10–7.98(m,2H),7.79(dd,J=8.7,1.6Hz,1H),7.73(dd,J=10.4,2.0Hz,1H),7.59–7.49(m,3H),6.59(d,J=4.0Hz,1H),3.65(s,2H),3.30(t,J=6.9Hz,2H),3.22(t,J=7.0Hz,2H),3.09(s,3H),3.01–2.92(m,4H),2.63(s,3H).
实施例20:
化合物(S)-1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)吡咯烷-2-甲腈的制备
参照实施例1的制备方法,收率39%。1H-NMR(600MHz,CDCl3)δ8.90(dd,J=14.9,6.9Hz,2H),7.78(dd,J=8.7,2.0Hz,2H),7.75–7.68(m,2H),6.66(s,1H),4.84–4.76(m,1H),3.82–3.71(m,2H),3.61–3.45(m,2H),3.09(s,5H),3.07–2.93(m,4H),2.63(s,3H),2.39–2.15(m,4H),2.06–1.99(m,2H).
实施例21:
化合物叔丁基4-(2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)乙酰基)哌嗪-1-氨基甲酸酯的制备
步骤(1):7-苄基-N-(2-氟-4-甲砜基苯基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺
参照实施例6步骤(1)的制备方法,收率64%。1H-NMR(600MHz,CDCl3)δ8.97(t,J=8.2Hz,1H),8.67(s,1H),7.79(d,J=8.7Hz,1H),7.73(dd,J=10.3,1.9Hz,1H),7.39(m,4H),7.32(m,1H),6.87(d,J=4.1Hz,1H),3.80(s,2H),3.70(s,2H),3.09(s,3H),2.93(s,2H),2.75(s,2H).
步骤(2):N-(2-氟-4-甲砜基苯基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺
参照实施例1步骤(2)的制备方法,收率57%。1H-NMR(600MHz,CDCl3)δ8.99(s,1H),8.69(s,1H),7.80(d,J=8.1Hz,1H),7.74(d,J=10.0Hz,1H),6.90(s,1H),4.04(s,2H),3.30(s,2H),3.09(s,3H),2.66(s,2H).
步骤(3):叔丁基4-(2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)乙酰基)哌嗪-1-氨基甲酸酯
参照实施例1步骤(3)的制备方法,收率62%。1H-NMR(600MHz,CDCl3)δ8.97(t,J=8.1Hz,1H),8.69(s,1H),7.79(d,J=8.7Hz,1H),7.73(d,J=10.3Hz,1H),6.89(d,J=3.7Hz,1H),3.76(s,2H),3.63(s,2H),3.58(s,2H),3.49(d,J=13.8Hz,2H),3.44(s,4H),3.09(s,3H),3.01(t,J=5.5Hz,2H),2.74(t,J=5.3Hz,2H),1.48(s,9H).
实施例22:
化合物(1S)-2-(2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)乙酰基)环戊基-1-甲腈的制备
参照实施例1的制备方法,收率45%。1H-NMR(600MHz,CDCl3)δ8.96(t,J=8.1Hz,1H),8.69(s,1H),7.79(d,J=8.8Hz,1H),7.74(d,J=10.3Hz,1H),6.90(s,1H),4.81(d,J=6.3Hz,1H),3.83(s,1H),3.77(m,1H),3.75–3.53(m,2H),3.48(m,2H),3.09(s,4H),2.77(t,J=5.6Hz,2H),2.45–2.15(m,4H),2.16–1.96(m,2H).
实施例23:
化合物叔丁基(R)-(1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)乙酰基)哌啶-3-基)氨基甲酸酯的制备
参照实施例1的制备方法,收率51%。1H-NMR(600MHz,CDCl3)δ8.98(d,J=7.9Hz,1H),8.69(s,1H),7.79(d,J=8.5Hz,1H),7.74(d,J=10.3Hz,1H),6.89(s,1H),3.79(m,3H),4.79(s,1H),3.64(m,2H),3.47(m,3H),3.32(m,1H),3.09(s,3H),3.04(m,2H),2.77(m,2H),2.03(m,1H),1.90(s,1H),1.47(s,2H),1.39(s,9H).
实施例24:
化合物2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)-1-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷的制备
参照实施例1的制备方法,收率55%。1H-NMR(600MHz,CDCl3)δ8.96(t,J=8.0Hz,1H),8.69(s,1H),7.80(d,J=8.5Hz,1H),7.74(d,J=10.2Hz,1H),6.87(s,1H),5.26–5.02(m,2H),4.21(t,J=5.4Hz,2H),4.14(t,J=5.3Hz,2H),3.71(d,J=53.6Hz,2H),3.55(d,J=46.1Hz,2H),3.09(s,3H),3.01(t,J=5.5Hz,2H),2.75(t,J=5.6Hz,2H).
实施例25:
化合物3-氟-4-((2-甲基-7-(2-氧代-2-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)乙基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈的制备
步骤(1):4-((7-苄基-2-甲基-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)-3-氟苯甲腈
参照实施例6步骤(1)的制备方法,收率65%。1H NMR(600MHz,CDCl3)δ8.99(t,J=8.4Hz,1H),7.52(d,J=8.7Hz,1H),7.42(dd,J=11.0,1.8Hz,1H),7.40–7.29(m,5H),6.81(d,J=4.7Hz,1H),3.75(s,2H),3.61(s,2H),2.89(t,J=
5.8Hz,2H),2.68(t,J=5.7Hz,2H),2.61(s,3H).
步骤(2):3-氟-4-((2-甲基-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈
参照实施例1步骤(2)的制备方法,收率60%。1H NMR(600MHz,CDCl3)δ9.01(t,J=8.4Hz,1H),7.52(d,J=8.7Hz,1H),7.43(dd,J=11.0,1.8Hz,1H),6.85(d,J=4.6Hz,1H),3.99(s,2H),3.28(t,J=5.8Hz,2H),2.63(s,3H),2.60(t,J=5.7Hz,2H),1.27(s,1H).
步骤(3):3-氟-4-((2-甲基-7-(2-氧代-2-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)乙基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈
参照实施例1步骤(3)的制备方法,收率57%。1H-NMR(400MHz,CDCl3)δ8.97(s,1H),7.51(s,1H),7.43(d,J=10.4Hz,1H),6.81(s,1H),5.13(m,2H),4.16(m,4H),3.69(s,2H),3.56(s,2H),2.98(d,J=5.0Hz,2H),2.69(s,2H),2.62(s,3H).
实施例26:
化合物3-氟-4-((2-甲基-7-((5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈的制备
参照实施例1的制备方法,收率71%。1H-NMR(400MHz,DMSO)δ8.97(t,J=8.4Hz,1H),8.08(d,J=7.3Hz,2H),7.64–7.35(m,5H),6.81(s,1H),4.16(s,2H),3.84(s,2H),3.08(d,J=5.0Hz,2H),2.73(s,2H),2.62(s,3H).
实施例27:
化合物叔丁基4-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)哌嗪-1-氨基甲酸酯的制备
参照实施例1的制备方法,收率71%。1H-NMR(400MHz,DMSO)δ8.99(t,J=8.2Hz,1H),7.53(d,J=7.5Hz,1H),7.43(d,J=10.9Hz,1H),6.83(s,1H),3.70(s,2H),3.59(s,4H),3.44(s,6H),2.98(d,J=5.3Hz,2H),2.69(s,2H),2.63(s,3H),1.48(s,9H).
实施例28:
化合物3-氟-4-((2-甲基-7-(2-氧代-2-(噻唑烷-3-基)乙基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈的制备
参照实施例1的制备方法,收率66%。1H-NMR(600MHz,CDCl3)δ8.99–8.91(m,1H),7.50(d,J=8.7Hz,1H),7.44–7.35(m,1H),6.81(d,J=4.5Hz,1H),4.73–4.53(m,2H),3.94–3.78(m,2H),3.78–3.68(m,2H),3.51–3.36(m,2H),3.02(m,4H),2.72–2.65(m,2H),2.61(d,J=12.9Hz,3H).
实施例29:
化合物叔丁基(R)-(1-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)哌啶-3-基)氨基甲酸酯的制备
参照实施例1的制备方法,收率62%。1H-NMR(600MHz,CDCl3)δ9.07–8.83(m,1H),7.51(d,J=8.5Hz,1H),7.42(d,J=10.9Hz,1H),6.85(s,1H),4.86(d,J=5.5Hz,1H),3.83–3.57(m,4H),3.52–3.37(m,2H),3.33(m,1H),2.98(dd,J=15.9,10.7Hz,2H),2.80–2.66(m,2H),2.61(s,3H),1.86(s,4H),1.66–1.52(m,2H),1.42(s,9H).
实施例30:
化合物3-氟-4-((2-甲基-7-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈的制备
参照实施例1的制备方法,收率65%。1H-NMR(600MHz,CDCl3)δ8.97(t,J=8.4Hz,1H),8.10–7.99(m,2H),7.59–7.48(m,4H),7.42(dd,J=10.9,1.8Hz,1H),6.81(d,J=4.7Hz,1H),3.72(s,2H),3.25(t,J=7.2Hz,2H),3.14(t,J=7.2Hz,2H),2.98(t,J=5.8Hz,2H),2.67(t,J=5.6Hz,2H),2.62(s,3H).
实施例31:
化合物(S)-1-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)吡咯烷-2-甲腈的制备
参照实施例1的制备方法,收率47%。1H-NMR(600MHz,CDCl3)δ8.98(t,J=8.4Hz,1H),7.52(d,J=8.7Hz,1H),7.43(dd,J=10.9,1.8Hz,1H),6.84(d,J=4.7Hz,1H),4.80(dd,J=8.0,2.5Hz,1H),3.75(s,2H),3.69–3.53(m,2H),3.52–3.39(m,2H),3.04(dd,J=6.6,5.3Hz,2H),2.71(t,J=5.7Hz,2H),2.62(s,3H),2.40–2.13(m,4H).
实施例32:
化合物2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)-1-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷的制备
步骤(1):7-苄基-N-(2-氟-4-甲砜基苯基)-2-甲基-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-胺
参照实施例6步骤(1)的制备方法,收率58%。1H-NMR(400MHz,CDCl3)δ9.03(s,1H),7.78(d,J=8.7Hz,1H),7.71(d,J=10.5Hz,1H),7.35(m,5H),6.84(s,1H),3.74(s,2H),3.61(s,2H),3.07(s,3H),2.89(t,J=5.7Hz,2H),2.68(s,2H),2.60(s,3H).
步骤(2):N-(2-氟-4-甲砜基苯基)-2-甲基-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-胺
参照实施例1步骤(2)的制备方法,收率55%。1H-NMR(400MHz,CDCl3)δ9.05(t,J=8.3Hz,1H),7.79(d,J=8.4Hz,1H),7.72(d,J=10.5Hz,1H),6.86(s,1H),3.98(s,2H),3.27(s,2H),3.08(s,3H),2.62(s,3H),2.61(s,2H).
步骤(3):2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)-1-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷
参照实施例1步骤(3)的制备方法,收率64%。1H-NMR(400MHz,CDCl3)δ9.01(t,J=8.0Hz,1H),7.78(d,J=8.6Hz,1H),7.71(d,J=10.2Hz,1H),6.84(s,1H),5.25(m,2H),4.19(s,2H),4.12(s,2H),3.67(d,J=12.2Hz,2H),3.55(s,2H),3.08(s,3H),2.97(s,2H),2.69(s,2H),2.61(s,3H).
实施例33:
化合物N-(2-氟-4-甲砜基苯基)-2-甲基-7-((5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-胺的制备
参照实施例1的制备方法,收率58%。1H-NMR(400MHz,CDCl3)δ9.02(t,J=8.2Hz,1H),8.08(d,J=6.8Hz,2H),7.78(d,J=8.5Hz,1H),7.71(d,J=10.5Hz,1H),7.61–7.46(m,3H),6.84(s,1H),4.15(s,2H),3.83(s,2H),3.07(s,5H),2.73(s,2H),2.61(s,3H).
实施例34:
化合物叔丁基4-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)哌嗪-1-氨基甲酸酯的制备
参照实施例1的制备方法,收率63%。1H-NMR(400MHz,CDCl3)δ8.96(t,J=8.3Hz,1H),7.72(d,J=8.1Hz,1H),7.64(d,J=10.1Hz,1H),6.78(s,1H),3.62(s,2H),3.54(s,4H),3.37(m,6H),3.01(s,3H),2.90(s,2H),2.62(s,2H),2.55(s,3H),1.40(s,9H).
实施例35:
化合物2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)-1-(噻唑烷-3-基)-1-氧代乙烷的制备
参照实施例1的制备方法,收率41%。1H-NMR(400MHz,CDCl3)δ9.25–8.90(m,1H),7.79(d,J=7.8Hz,1H),7.72(d,J=10.3Hz,1H),6.86(s,1H),4.64(d,J=9.0Hz,2H),3.88(d,J=5.7Hz,2H),3.74(s,2H),3.45(d,J=9.9Hz,2H),3.09(s,2H),3.08(s,3H),3.02(s,2H),2.71(s,2H),2.62(s,3H).
实施例36:
化合物叔丁基(R)-(1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)哌啶-3-基)氨基甲酸酯的制备
参照实施例1的制备方法,收率55%。1H-NMR(400MHz,CDCl3)δ9.02(s,1H),7.77(d,J=8.6Hz,1H),7.70(d,J=10.3Hz,1H),6.87(s,1H),4.89(s,1H),3.76(s,1H),3.76–3.67(m,2H),3.60(s,2H),3.41(t,J=34.0Hz,4H),3.07(s,3H),2.98(s,2H),2.73(s,2H),2.60(s,3H),2.02(s,2H),1.87(s,2H),1.37(s,9H).
实施例37:
化合物N-(2-氟-4-甲砜基苯基)-2-甲基-7-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-胺的制备
参照实施例1的制备方法,收率59%。1H NMR(400MHz,CDCl3)δ9.02(t,J=8.1Hz,1H),8.04(d,J=6.6Hz,2H),7.78(d,J=8.7Hz,1H),7.71(d,J=10.3Hz,1H),7.52(d,J=7.7Hz,3H),6.83(s,1H),3.72(s,2H),3.25(t,J=7.0Hz,2H),3.20–3.11(m,2H),3.08(s,3H),2.98(s,2H),2.67(s,2H),2.62(s,3H).
实施例38:
化合物(S)-1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)吡咯烷-2-甲腈的制备
参照实施例1的制备方法,收率39%。1H-NMR(400MHz,CDCl3)δ9.03(t,J=8.3Hz,1H),7.79(d,J=8.6Hz,1H),7.72(d,J=10.3Hz,1H),6.86(s,1H),5.36(s,1H),3.73(m,4H),3.69–3.53(m,2H),3.45(m,2H),3.08(s,3H),3.04(d,J=5.7Hz,2H),2.72(s,2H),2.62(s,3H),2.03(s,2H).
实施例39:
(1):GPR119激动活性测试
首先将含有人GPR119基因的DNA片段转染到HEK293细胞(人胚肾293细胞)中,通过培养得到稳定表达GPR119的细胞株,在96孔板上接种该细胞株。细胞在37℃,5%CO2条件下培养48h后,移去培养液,加入100μL缓冲液,并于室温孵育15min。再给予不同浓度的待测化合物,孵育30min后,移去缓冲液,加入75μL预冷的裂解液,并在冰上孵育20min,适当地振荡。将裂解液转移到1.5mL离心管中,以13000rpm的转速离心10min。取50μL上清液,采用HTRF cAMP试剂盒标准步骤,以GSK-1292263为阳性对照,测定细胞内cAMP的浓度,由剂量依赖的cAMP浓度变化获得化合物EC50值,用于评价化合物对受体的亲和力强弱,计算化合物刺激cAMP作用与GSK-1292263产生最大作用的比值(%max),用于比较化合物对受体的内在活性(IA)。
(2):DPP-4抑制活性
在96孔板上依次加入缓冲液、发光底物Gly-Pro-pNA、受试药物和DPP-4酶,设空白对照组和对照组,考察药物单浓度下的抑制活性(10μM),设复孔。每孔的总体积为100μl,缓冲液pH8.2,温度37℃,反应时间60min。磷酸西他列汀为阳性对照药。抑制率%=[(OD阴性对照组-OD空白对照组)-(OD受试组-OD受试组空白对照组)]/(OD阴性对照组-OD空白对照组)×100%。
(3):测试结果见表1。
表1:hGPR119活性和DPP-4抑制活性测试结果
上表显示了本发明的化合物对hGPR119具有良好的激动活性,对DPP-4也具有良好的抑制活性。因此,可以作为治疗与GPR119和DPP-4相关的疾病的药物中,尤其是代谢综合症或糖尿病,更典型的是代谢失调和肥胖疾病。具有良好的前景。
以上对本发明的实施例进行了说明,但本发明的保护内容不仅仅限定于以上实施例,在本发明的所属技术领域中,只要掌握通常知识,就可以在其技术要旨范围内进行多种多样的变更。
Claims (9)
2.根据权利要求1所述的四氢吡啶并嘧啶衍生物及其医药学上可接受的盐,其特征在于R2为氢原子;R3为氢原子或卤素或氰基或甲基砜基;R4为氢原子或C1-6烷基;R5为氢原子。
4.根据权利要求1所述的四氢吡啶并嘧啶衍生物及其医药学上可接受的盐,其特征在于式(I)化合物为如下化合物:
(1).3-氟-4-((6-(2-氧代-2-噻唑烷-3-乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈
(2).3-氟-4-((6-(2-氧代-2-(3-(三氟甲基)-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈
(3).叔丁基(R)-(1-(2-(4-((4-氰基-2-氟苯基)氨基)-7,8-二氢吡啶并[4,3-d]嘧啶)-6(5H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
(4).3-氟-4-((6-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈
(5).3-氟-4-((6-(2-(5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶)-4-氨基)苯甲腈
(6).化合物2-(4-((2-氟-4-(甲砜基)苯基)氨基)-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-(噻唑烷-3-基)-1-氧代乙烷
(7).2-(4-((2-氟-4-(甲砜基)苯基)氨基)-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-(3-三氟甲基)-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷
(8).N-(2-氟-4-甲砜基苯基)-6-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺
(9).叔丁基-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)哌嗪-1-氨基甲酸酯
(10).3-氟-4-((2-甲基-6-(2-氧代-2-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4基)-胺基)苯甲腈
(11).3-氟-4-((2-甲基-6-((5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4基)-胺基)苯甲腈
(12).叔丁基(R)-(1-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶)-6(5H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
(13).3-氟-4-((2-甲基-6-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4基)-胺基)苯甲腈
(14).2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷
(15).N-(2-氟-4-甲砜基苯基)-2-甲基-6-((5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺
(16).叔丁基4-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)哌嗪-1-氨基甲酸酯
(17).2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)-1-噻唑烷-3-基)-1-氧代乙烷
(18).叔丁基(R)-(1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
(19).N-(2-氟-4-甲砜基苯基)-2-甲基-6-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4-胺
(20).(S)-1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-7,8-二氢吡啶并[4,3-d]嘧啶-6(5H)-基)乙酰基)吡咯烷-2-甲腈
(21).叔丁基4-(2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)乙酰基)哌嗪-1-氨基甲酸酯
(22).(1S)-2-(2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)乙酰基)环戊基-1-甲腈
(23).叔丁基(R)-(1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
(24).2-(4-((2-氟-4-甲砜基苯基)氨基)-5,8-二氢吡啶并[4,3-d]嘧啶-7(6H)-基)-1-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷
(25).3-氟-4-((2-甲基-7-(2-氧代-2-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)乙基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈
(26).3-氟-4-((2-甲基-7-((5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈
(27).叔丁基4-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)哌嗪-1-氨基甲酸酯
(28).3-氟-4-((2-甲基-7-(2-氧代-2-(噻唑烷-3-基)乙基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈
(29).叔丁基(R)-(1-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
(30).3-氟-4-((2-甲基-7-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-基)胺基)苯甲腈
(31).(S)-1-(2-(4-((4-氰基-2-氟苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)吡咯烷-2-甲腈
(32).2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)-1-(3-三氟甲基-5,6-二氢-[1,2,4]三氮唑并[4,3-a]哌嗪-7(8H)-基)-1-氧代乙烷
(33).N-(2-氟-4-甲砜基苯基)-2-甲基-7-((5-苯基-1,3,4-噁二唑-2-基)甲基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-胺
(34).叔丁基4-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)哌嗪-1-氨基甲酸酯
(35).2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)-1-(噻唑烷-3-基)-1-氧代乙烷
(36).叔丁基(R)-(1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)哌啶-3-基)氨基甲酸酯
(37).N-(2-氟-4-甲砜基苯基)-2-甲基-7-(2-(5-苯基-1,3,4-噁二唑-2-基)乙基)-5,6,7,8-四氢吡啶并[3,4-d]嘧啶-4-胺
(38).(S)-1-(2-(4-((2-氟-4-甲砜基苯基)氨基)-2-甲基-5,8-二氢吡啶并[3,4-d]嘧啶-7(6H)-基)乙酰基)吡咯烷-2-甲腈。
5.一种制备权利要求1所述的四氢吡啶并嘧啶衍生物及其医药学上可接受的盐的方法,其特征在于具体包括如下步骤:
步骤(1):式(IV)化合物与取代或未取代苯胺进行反应,生成式(V)化合物
在有机溶剂中,在强碱存在下,在室温至100℃反应,反应完后处理后得到式(V)化合物;
步骤(2):式(V)化合物脱去苄基生成式(VI)化合物
在有机溶剂中,使用1-氯乙基氯甲酸酯使式(V)脱去苄基,后处理后得到式(VI)化合物;
步骤(3):式(VI)化合物与R1-X反应生成式(I)化合物
其中X为卤素;
在有机溶剂中,在碱的存在下,式(VI)化合物与R1-X反应生成式(I)化合物;
R1-R5以及n和m如权利要求1中所定义。
6.一种药物组合物,其特征在于包含权利要求1-3任一项的化合物和医药学上可接受的载体。
7.根据权利要求1-3任一项化合物或其医药学上可接受的盐或权利要求6的组合物在制备作为治疗与GPR119和DPP-4相关的疾病的药物中的用途。
8.根据权利要求7所述的用途,其特征在于所述疾病为代谢综合症或糖尿病。
9.根据权利要求8所述的用途,其特征在于所述疾病为代谢失调和肥胖。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910382406.3A CN110256420B (zh) | 2019-05-07 | 2019-05-07 | 四氢吡啶并嘧啶衍生物及其制备方法和用途 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910382406.3A CN110256420B (zh) | 2019-05-07 | 2019-05-07 | 四氢吡啶并嘧啶衍生物及其制备方法和用途 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN110256420A CN110256420A (zh) | 2019-09-20 |
| CN110256420B true CN110256420B (zh) | 2020-04-28 |
Family
ID=67914507
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201910382406.3A Expired - Fee Related CN110256420B (zh) | 2019-05-07 | 2019-05-07 | 四氢吡啶并嘧啶衍生物及其制备方法和用途 |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN110256420B (zh) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114957246B (zh) * | 2021-02-19 | 2025-11-11 | 苏州恩华生物医药科技有限公司 | 5,6,7,8-四氢吡啶并[4,3-d]嘧啶-4(3H)-酮衍生物及其应用 |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TR199902455T2 (xx) * | 1997-04-16 | 2000-01-21 | Abbott Laboratories | 5,7-Dis�bstit�e 4-Aminopirido (2,3-D) Pirimidin bile�ikleri ve bunlar�n adenosin kinaz inhibit�rleri olarak kullan�m� |
| TW200530235A (en) * | 2003-12-24 | 2005-09-16 | Renovis Inc | Bicycloheteroarylamine compounds as ion channel ligands and uses thereof |
| US20060128710A1 (en) * | 2004-12-09 | 2006-06-15 | Chih-Hung Lee | Antagonists to the vanilloid receptor subtype 1 (VR1) and uses thereof |
| EP1885369B1 (en) * | 2005-05-04 | 2015-09-23 | Evotec AG | Fused heterocyclic compounds, and compositions and uses thereof |
| JP6475866B2 (ja) * | 2015-11-27 | 2019-02-27 | 大鵬薬品工業株式会社 | 縮環ピリミジン化合物又はその塩 |
| IT201600130706A1 (it) * | 2016-12-23 | 2018-06-23 | Fondazione St Italiano Tecnologia | 4 amino-2-pirido-biciclo pirimidine e lo uso come medicamenti |
-
2019
- 2019-05-07 CN CN201910382406.3A patent/CN110256420B/zh not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| CN110256420A (zh) | 2019-09-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10112950B2 (en) | Substituted imidazo[1,2-a]pyrazines as LSD1 inhibitors | |
| EP2358716B1 (en) | Inhibitors of fatty acid binding protein (fabp) | |
| TWI542590B (zh) | 1,2-雙取代雜環化合物 | |
| KR101499308B1 (ko) | 가용성 구아닐레이트 시클라제 활성화제 | |
| TWI829676B (zh) | 噁二唑暫時受體電位通道抑制劑 | |
| EP3810587A1 (en) | Substituted alkoxypyridinyl indolsulfonamides | |
| EP2485735B1 (en) | Inhibitors of fatty acid binding protein (fabp) | |
| CN111499634B (zh) | 一种喹唑啉化合物及其在医药上的应用 | |
| US20210261552A1 (en) | Erk inhibitor and use thereof | |
| CN105732640B (zh) | 醛糖还原酶抑制剂及其用途 | |
| EP2786998B1 (en) | Thienyl [3, 2-d]pyrimidin-4-one compounds, preparation method, pharmaceutical compositions and use thereof | |
| JP2021535164A (ja) | 心筋サルコメア阻害剤 | |
| TW201043632A (en) | Quinoxaline compounds | |
| US12371432B2 (en) | Haloallylamine compounds and application thereof | |
| WO2017147047A1 (en) | Fxr modulators and methods of their use | |
| WO2015058661A1 (zh) | Bcr-abl激酶抑制剂及其应用 | |
| JP7301758B2 (ja) | 縮合二環式化合物、その組成物及び応用 | |
| WO2019085894A1 (zh) | 一类含氮稠环化合物及其制备方法和用途 | |
| WO2024208225A1 (zh) | 二氢噻吩并嘧啶类化合物、其制备方法和应用 | |
| WO2022228515A1 (zh) | 甲硫氨酸腺苷转移酶抑制剂、其制备方法及应用 | |
| US20180155344A1 (en) | Tricyclic piperidine compounds | |
| WO2024012496A1 (zh) | 大麻素受体化合物及其用途 | |
| CN110256420B (zh) | 四氢吡啶并嘧啶衍生物及其制备方法和用途 | |
| CN119874686A (zh) | 氘代苯并咪唑衍生物及其制备方法和用途 | |
| TWI846030B (zh) | Sarm1酶活性抑制劑及其應用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20210604 Address after: 330029 6th floor, medical Valley, no.898, Nanjing East Road, Qingshanhu District, Nanchang City, Jiangxi Province Patentee after: Jiangxi Lecheng Biotechnology Co.,Ltd. Address before: No. 1688 Meiling Avenue, Wanli District, Nanchang City, Jiangxi Province Patentee before: JIANGXI University OF TRADITIONAL CHINESE MEDICINE |
|
| TR01 | Transfer of patent right | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20200428 Termination date: 20210507 |
|
| CF01 | Termination of patent right due to non-payment of annual fee |