CN109464429B - Inhalation pressure quantitative aerosol pharmaceutical composition and preparation method thereof - Google Patents
Inhalation pressure quantitative aerosol pharmaceutical composition and preparation method thereof Download PDFInfo
- Publication number
- CN109464429B CN109464429B CN201811522217.3A CN201811522217A CN109464429B CN 109464429 B CN109464429 B CN 109464429B CN 201811522217 A CN201811522217 A CN 201811522217A CN 109464429 B CN109464429 B CN 109464429B
- Authority
- CN
- China
- Prior art keywords
- pharmaceutical composition
- lactose
- aerosol
- formoterol
- particle size
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/007—Pulmonary tract; Aromatherapy
- A61K9/0073—Sprays or powders for inhalation; Aerolised or nebulised preparations generated by other means than thermal energy
- A61K9/0078—Sprays or powders for inhalation; Aerolised or nebulised preparations generated by other means than thermal energy for inhalation via a nebulizer such as a jet nebulizer, ultrasonic nebulizer, e.g. in the form of aqueous drug solutions or dispersions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/135—Amines having aromatic rings, e.g. ketamine, nortriptyline
- A61K31/137—Arylalkylamines, e.g. amphetamine, epinephrine, salbutamol, ephedrine or methadone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/10—Alcohols; Phenols; Salts thereof, e.g. glycerol; Polyethylene glycols [PEG]; Poloxamers; PEG/POE alkyl ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/12—Aerosols; Foams
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Pulmonology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Epidemiology (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Emergency Medicine (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Dispersion Chemistry (AREA)
- Otolaryngology (AREA)
- Medicinal Preparation (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
The invention provides an inhalation pressure quantitative aerosol pharmaceutical composition and a preparation method thereof. The pharmaceutical composition of the present invention comprises formoterol or a pharmaceutically acceptable salt thereof as an active ingredient, lactose, a solvent, polyethylene glycol (PEG) and a hydrofluoroalkane propellant. The pharmaceutical composition disclosed by the invention is good in uniformity and physical stability, remarkably improved in administration performance, and has obvious advantages and good market prospects.
Description
Technical Field
The invention belongs to the technical field of inhalation pharmaceutical preparations, and particularly relates to an inhalation pressure quantitative aerosol pharmaceutical composition and a preparation method thereof.
Background
Researches show that the bronchiectasis can obviously relieve dyspnea caused by airway obstructive diseases such as bronchial asthma, chronic tracheitis, asthmatic bronchitis, emphysema and the like. At present, the following medicines are mainly used for treating lung diseases such as asthma and the like: (1) a beta 2 receptor agonist; (2) a glucocorticoid; (3) anti-allergic agents; (4) xanthine drugs; (5) anticholinergic agents. Formoterol as a long-acting selective adrenergic beta 2 receptor agonist has certain bronchodilatory action, can obviously relieve dyspnea caused by the above diseases, and further achieve the therapeutic effect.
Aerosols and dry powder inhalers are common inhaled respiratory tract preparations for the treatment of airway obstructive diseases. The aerosol is a preparation which is packaged in a pressure-resistant container with a specific quantitative valve system together with a drug-containing emulsion or suspension and a proper propellant, and the content is sprayed out in the form of mist by the pressure of the propellant when the aerosol is used, so that the aerosol is inhaled into the lung. The dry powder inhalant is a medicinal preparation which is prepared by filling one or more medicaments into a container such as a capsule, a bubble cap, a storage tank and the like, then feeding the medicaments into respiratory tract in a dry powder form through a special administration device and playing a systemic or local role. The aerosol is mostly in a solution type and a suspension type and uses a propellant, while the dry powder inhalant is a solid containing a carrier, and the two dosage forms have obvious difference from the aspect of formulation, the key point of the aerosol formulation is to research the uniformity and stability of emulsion or suspension, and the key point of the dry powder inhalant is to research the particle science technology among different solid particles.
Aerosol suspensions often suffer from drug heterogeneity upon standing. Airway obstructive diseases such as asthma are potentially fatal diseases, and this heterogeneity is especially critical in the maintenance treatment of these conditions with suspensions. The adverse reaction is caused when the dosage of the medicine is more than the marked amount, and the insufficient administration is caused when the dosage of the medicine is less than the marked amount, so that the possibility of the relapse of the disease is increased.
There are 2 formoterol fumarate inhalation products used clinically: formoterol inhalation aerosol, developed by kexi italy, is marketed only in a few countries in europe, and the product obtained clinical lots in china in 2009, but no product is marketed domestically; formoterol dry powder inhaler, developed by astrazen and marketed in china. The formoterol inhalation aerosol sold in Kaixi is a solution type aerosol, and in order to improve the solubility and ensure the physical stability, more ethanol and cosolvent are added in the prescription, so that the chemical property of the solution type product is unstable, the requirement on the storage condition is severe, and the solution type product can be only stored in a refrigerator for l5 months and at room temperature for 3 months. Formoterol dry powder inhalant developed by astrazen adopts a turbuhaler device, the medicine is stored in a storage, the humidity is sensitive, the dose split charging of the medicine depends on the operation technology of a user, the uniformity is easy to be influenced, in addition, the structure of the turbuhaler device is complex, the product is expensive, and the acceptance degree on the low-end market is not high.
CN1638730 discloses a formoterol pressurized metered dose inhaler (pMDIs), which is a solution type formulation, wherein formoterol is completely dissolved in the system, and strong acid is added to adjust pH, the solution type formulation has the disadvantages of low effective lung deposition and poor stability compared with the suspension type formulation of the present invention, and the introduction of strong acid has a significantly increased requirement for container materials, and there is a safety risk of dissolution of harmful substances.
EP1400239 and US6054488 are directed to a metered dose inhalation aerosol formulation containing formoterol fumarate only, which does not contain polyethylene glycol, and which has low physical stability and low dose uniformity.
Disclosure of Invention
The inventor of the application finds that the addition of the polyethylene glycol and the lactose can obviously improve the dose uniformity of the main drug in the liquid medicine system.
Accordingly, in order to overcome the drawbacks of the prior art, it is an object of the present invention to provide a pharmaceutical composition for an inhalation pressure metered dose aerosol comprising formoterol and a process for the preparation thereof.
The purpose of the invention is realized by the following technical scheme.
In one aspect, the present invention provides an inhalation pressure metered dose aerosol pharmaceutical composition comprising as active ingredients formoterol or a pharmaceutically acceptable salt thereof, lactose, a solvent, polyethylene glycol (PEG) and a hydrofluoroalkane propellant.
Preferably, the pharmaceutically acceptable salt is selected from formoterol fumarate and formoterol fumarate dihydrate.
Preferably, the lactose is selected from one or more of lactose monohydrate and lactose anhydrous.
Preferably, the solvent is selected from one or more of glycerol, propylene glycol and ethanol, preferably ethanol.
Preferably, the polyethylene glycol is selected from one or more of PEG 100-2000, preferably PEG100, PEG300, PEG500, PEG1000, PEG1300, PEG1500 or PEG2000, and more preferably PEG 1000.
Preferably, the hydrofluoroalkane propellant is selected from one or more of 1,1,1, 2-tetrafluoroethane (HFA 134a) and 1,1,1,2,3,3, 3-heptafluoropropane (HFA 227), preferably HFA 227.
Preferably, D of the active ingredient90The particle size is 0.1 to 10 μm, preferably 0.1 to 7 μm, and more preferably 0.1 to 5 μm; d50The particle size is 0.1 to 7 μm, preferably 0.1 to 5 μm, and more preferably 0.1 to 3 μm; d10The particle size is 0.1 to 5 μm, preferably 0.1 to 3 μm, and more preferably 0.1 to 1.5 μm.
Preferably, D of said lactose90The particle size is 0.1 to 30 μm, preferably 0.1 to 10 μm, and more preferably 0.1 to 5 μm; d50The particle size is 0.1 to 15 μm, preferably 0.1 to 5 μm, and more preferably 0.1 to 3 μm; d10The particle size is 0.1 to 5 μm, preferably 0.1 to 3 μm, and more preferably 0.1 to 1.5 μm.
Preferably, the weight percentage of the active ingredient in the pharmaceutical composition is 0.004-0.085%, preferably 0.017-0.085%.
Preferably, the lactose accounts for 0.008-0.085% of the pharmaceutical composition by weight, preferably 0.008-0.043% by weight, and more preferably 0.008-0.014% by weight.
Preferably, the solvent accounts for 0.0-10% of the weight of the pharmaceutical composition, and preferably 0.0-5%; more preferably 0.0% to 2%.
Preferably, the weight percentage of the polyethylene glycol in the pharmaceutical composition is 0.0001-0.1%, preferably 0.001-0.02%.
Preferably, the pharmaceutical composition may also comprise other low volatility components, such as other alcohols, vegetable oils, organic acids and the like, wherein the alcohol may be an alkanol, such as decanol (decyl alcohol), sorbitol, mannitol, lactitol, the sugar alcohols of maltitol, tetrahydrofuryl methanol and dipropylene glycol; the organic acid is saturated carboxylic acid of dodecanoic acid, tetradecanoic acid and stearic acid, such as sorbic acid, etc. The following components can also be added into the prescription, and the components are used as dispersing agents for keeping suspended particles from agglomerating so as to improve the physical stability of the medicine suspension, and are commonly used known pharmaceutical auxiliary materials of the aerosol, such as: saccharin, ascorbic acid, cyclamic acid, amino acids, or aspartame; alkanes such as dodecane and octadecane; terpenes such as menthol, eucalyptol, limonene; sugars such as glucose, sucrose; polysaccharides such as ethyl cellulose, dextran; antioxidants such as butylated hydroxytoluene, ascorbic acid, sodium metabisulfite, butylated hydroxyanisole; polymers such as polyvinyl alcohol, polyvinyl acetate, polyvinyl pyrrolidone; amines such as ethanolamine, diethanolamine, triethanolamine; steroids such as cholesterol, cholesterol esters.
In another aspect, the present invention provides a process for preparing the above inhalation pressure quantitative aerosol pharmaceutical composition, comprising the steps of:
(a) micronizing active ingredient formoterol or pharmaceutically acceptable salt thereof and lactose;
(b) mixing the micronized product of the step (a), a solvent and polyethylene glycol, and closing a valve by a cover;
(c) dosing the mixture of step (b) with a prescribed amount of propellant through a valve.
In the method, ultrasonic bath can be optionally carried out on the aerosol bottle to increase the suspension performance of the main medicine in the system.
The invention also provides another preparation process of the inhalation pressure quantitative aerosol pharmaceutical composition, which comprises the following steps:
(a) micronizing active ingredient formoterol or pharmaceutically acceptable salt thereof and lactose;
(b) dispersing the micronized product, the solvent and the polyethylene glycol in the step (a) into the precooled propellant, uniformly mixing at low temperature, and quantitatively subpackaging the liquid medicine into an aerosol can.
In the above method, in the step (b), the low temperature is-80 ℃ to-60 ℃.
When the formoterol and lactose, especially the lactose with the particle size close to that of the formoterol are prepared into the inhalable suspension type pressure quantitative aerosol medicine composition, the medicine stability and the content uniformity of the formoterol in a suspension type pressure quantitative aerosol system can be effectively improved, which may be that the lactose molecules and the formoterol medicine particles have charge mutual influence in the suspension system, the physical and chemical properties of the formoterol particles are changed, the formoterol particles are more easily and uniformly mixed with a suspension composed of a propellant and other auxiliary material solvents, so that the better suspension stability is kept, the content uniformity of the medicine in use is improved, and the consistency of the medicine dosage in the whole medicine taking process of a patient can be ensured.
The invention has the beneficial effects that:
by adding proper auxiliary materials and processes, the product has better preparation uniformity and physical stability, the administration performance is obviously improved, and the invention has obvious advantages and good market prospect.
Detailed Description
The invention will be further illustrated with reference to the following specific examples. It should be understood that these examples are for illustrative purposes only and are not intended to limit the scope of the present invention. The experimental procedures, in which specific conditions are not noted in the following examples, are generally carried out under conventional conditions or under conditions recommended by the manufacturers.
The micronization of the present invention may be carried out by a known mechanical pulverization method. The mechanical pulverization method is that the active ingredient formoterol and the pharmaceutic adjuvant lactose are respectively pulverized into required particle sizes by a fluid energy mill method.
And (3) a micronization process: the material suction (air inlet) pressure is 8bar, the crushing pressure is 12bar, the feeding speed is 30g/h, all the micro powder in the material collecting bin is collected after the crushing is finished, and then the powder is crushed for a plurality of times until D90Particle diameter, D50Particle size and D10The particle size reaches the required range.
D90Particle size refers to the particle size parameter associated with a sample having a cumulative percent particle size distribution of 90%.
D50Particle size refers to the particle size parameter associated with a sample having a cumulative percent particle size distribution of 50%.
D10Particle size isRefers to the corresponding particle size parameter when the cumulative percentage of particle size distribution of a sample reaches 10%.
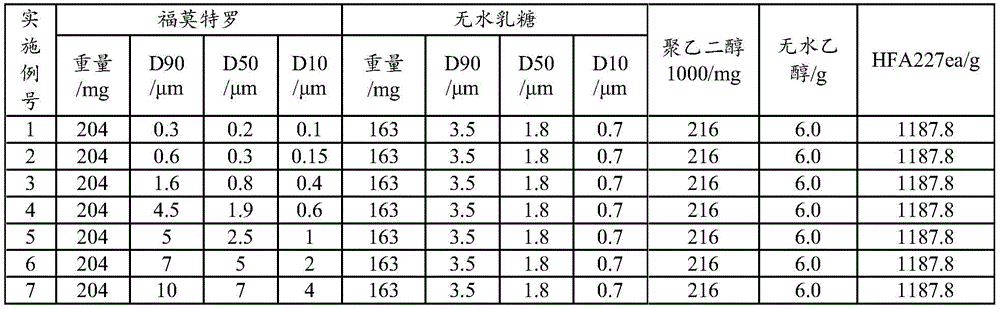
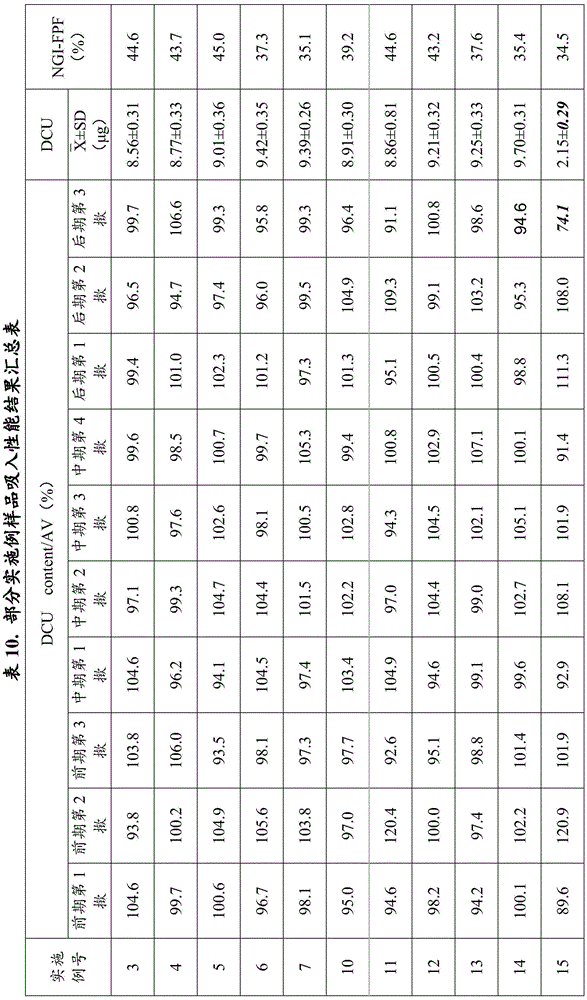
Examples 1 to 7: aerosol formulation (100 bottles)
The anhydrous lactose is micropowder D90Particle diameter of 3.5 μm, D50Particle diameter of 1.8 μm and D10Lactose with particle size of 0.7 μm, active ingredient addition amount of 0.017%, lactose addition amount of 0.014%, solvent including anhydrous ethanol (0.5%) and polyethylene glycol 1000 (0.02%), propellant including HFA227ea, and fine powder of the active ingredient D90Particle size of 0.1 to 10 μm, D50Particle diameter of 0.1 to 7 μm and D10Formoterol fumarate dihydrate having a particle size of 0.1 to 5 μm, as shown in Table 1. The use of metered dose valve systems of different specifications (50 μ l for example) allows the aerosol to be produced in the required dose per actuation.
TABLE 1 Aerosol preparation examples 1 to 7(100 bottles)
The preparation process comprises the following steps: adding formoterol and anhydrous lactose into ethanol and polyethylene glycol, adding liquid HFA227ea, stirring and mixing at low temperature (-80 deg.C to-60 deg.C), canning according to dosage, and sealing quantitative valve system (50 μ l for example). The dose is 12 mug per actuation of formoterol and is 170 actuations in total. Inhalation performance measurements were performed for examples 1-7 and the results show that: in addition to examples 6 and 7, the samples of the other examples have good effective lung deposition, the NGI-FPF is more than 38%, and the DCU determination results also all accord with the pharmacopoeia regulations.
Examples 8 to 14: aerosol formulation (100 bottles)
The lactose monohydrate is in the form of micropowder D90Particle size of 0.1 to 30 μm, D50Particle diameter of 0.1 to 15 μm and D100.1-5 mu m lactose with active component addition of 0.015%, 0.012%, absolute ethyl alcohol (0.5%) and polyethylene glycol 1000 (0.02%), propellant HFA134a, and fine powder of D as active component90Particle diameter of 4.2 μm, D50Particle diameter of 1.6 μm and D10Formoterol fumarate dihydrate with a particle size of 0.5 μm, as shown in table 2. The use of metered dose valve systems of different specifications (63 μ l for example) allows the aerosol to be produced in the required dose per actuation.
TABLE 2 Aerosol preparation examples 8 to 14(100 bottles)
The preparation process comprises the following steps: adding formoterol and lactose monohydrate into ethanol and polyethylene glycol, adding liquid HFA134a, stirring and mixing at low temperature (-80 deg.C to-60 deg.C), canning according to dosage, and sealing quantitative valve system (63 μ l for example). The dose is 12 mug per actuation of formoterol and is 170 actuations in total. Inhalation performance measurements were performed for examples 8-14 and the results show that: in addition to examples 13 and 14, the samples of the other examples have good effective lung deposition, the NGI-FPF is more than 38%, and the DCU determination results also all accord with the pharmacopoeia regulations.
Examples 15 to 20: aerosol formulation (100 bottles)
The lactose monohydrate is in the form of micropowder D90Particle diameter of 3.5 μm, D50Particle diameter of 1.8 μm and D10Lactose with a particle size of 0.7 μm, 0.004-0.085% of active ingredient, 0.008-0.085% of lactose, a weight ratio of the active ingredient to the added lactose of 1: 0.1-1: 20, anhydrous ethanol (0.5%) and polyethylene glycol 1000 (0.02%) as solvents, and HFA227ea as propellant, wherein the active ingredient is formoterol fumarate dihydrate which is micro-powder, the particle size of the D90 is 4.5 μm, the particle size of the D50 is 1.9 μm, and the particle size of the D10 is 0.6 μm, as shown in Table 3. The use of metered dose valve systems of different specifications (50 μ l for example) allows the aerosol to be produced in the required dose per actuation.
TABLE 3 Aerosol formulation examples 15 to 20(100 bottles)
The preparation process comprises the following steps: adding formoterol and lactose monohydrate into ethanol and polyethylene glycol, adding liquid HFA227ea, stirring and mixing at low temperature (-80 deg.C to-60 deg.C), canning according to dosage, and sealing quantitative valve system (50 μ l for example). From examples 15 to 20, the dosages were in the order of 3. mu.g, 60. mu.g, 12. mu.g, 3. mu.g and 60. mu.g of formoterol per push, and 170 strokes were used. Inhalation performance measurements were performed for examples 15-20 and the results show that: except for example 15, the samples of the other examples have good effective lung deposition, the NGI-FPF is more than 38%, and the DCU determination results also all accord with the pharmacopoeia regulations.
Examples 21 to 26: aerosol formulation (100 bottles)
The lactose monohydrate used was as a fine powder to D90 particle size of 3.6 μm, D50 particle size of 1.7 μm and D10 particle size of 0.6 μm, the active ingredient added was 0.015% -0.016%, the lactose added was 0.012% -0.013%, anhydrous ethanol (0.0% -10%) and polyethylene glycol 1000 (0.02%), the propellant was HFA134a, the active ingredient used was as a fine powder to D90 particle size of 4.2 μm, D50 particle size of 1.6 μm and D10 particle size of 0.5 μm formoterol fumarate dihydrate, as shown in table 4. The use of metered dose valve systems of different specifications (63 μ l for example) allows the aerosol to be produced in the required dose per actuation.
TABLE 4 Aerosol preparation examples 21 to 26(100 bottles)
Preparation process of example 21: adding formoterol, lactose monohydrate and polyethylene glycol in the prescription amount into an aerosol can, sealing a quantitative valve system (63 μ l as an example), and pressing HFA134a in the prescription amount into the aerosol can with the valve packaged. The dose is 12 mug per actuation of formoterol and is 170 actuations in total.
The preparation process of the embodiment 22-26: adding formoterol and lactose monohydrate into ethanol and polyethylene glycol, adding liquid HFA134a, stirring and mixing at low temperature (-80 deg.C to-60 deg.C), canning according to dosage, and sealing quantitative valve system (63 μ l for example). The dose is 12 mug per actuation of formoterol and is 170 actuations in total. Inhalation performance measurements were performed for examples 21-26 and the results showed that: the samples of examples 21-26 had good effective lung deposition, with NGI-FPF above 38%; meanwhile, the DCU determination result also completely conforms to the pharmacopoeia regulation.
Examples 27 to 32: aerosol formulation (100 bottles)
The lactose monohydrate is in the form of micropowder D90Particle diameter of 3.5 μm, D50Particle diameter of 1.8 μm and D10Lactose with particle diameter of 0.7 μm, active ingredient addition amount of 0.017%, lactose addition amount of 0.014%, solvent including anhydrous ethanol (1%) and polyethylene glycol 1000 (0.0001% -0.1%), propellant including HFA227ea, and active ingredient in the form of micropowder D90Particle diameter of 4.5 μm, D50Particle diameter of 1.9 μm and D10Formoterol fumarate dihydrate with a particle size of 0.6 μm, as shown in table 5. The use of metered dose valve systems of different specifications (50 μ l for example) allows the aerosol to be produced in the required dose per actuation.
TABLE 5 Aerosol preparation examples 27 to 32(100 bottles)
The preparation process comprises the following steps: adding formoterol and lactose monohydrate into ethanol and polyethylene glycol, adding liquid HFA227ea, stirring and mixing at low temperature (-80 deg.C to-60 deg.C), canning according to dosage, and sealing quantitative valve system (50 μ l for example). The dose is 12 mug per actuation of formoterol and is 170 actuations in total. Inhalation performance measurements were performed for examples 27-32 and the results showed that: the NGI-FPF of the samples of the examples 27-32 is below 38%, but the DCU measurement results all accord with the pharmacopoeia regulations.
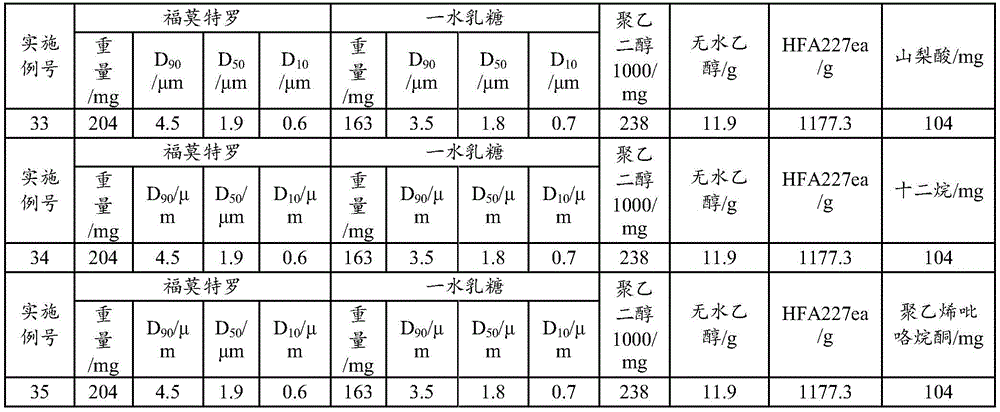
Examples 33 to 35: aerosol formulation (100 bottles)
The lactose monohydrate is in the form of micropowder D90Particle diameter of 3.5 μm, D50The particle size was 1.8 μm with D10Lactose with particle diameter of 0.7 μm, active ingredient addition amount of 0.017%, lactose addition amount of 0.014%, solvent including anhydrous ethanol (1%) and polyethylene glycol 1000 (0.0001% -0.1%), propellant including HFA227ea, and active ingredient in the form of micropowder D90Particle diameter of 4.5 μm, D50Particle diameter of 1.9 μm and D10Formoterol fumarate dihydrate with a particle size of 0.6 μm, to which 0.01% sorbic acid or dodecane or polyvinylpyrrolidone was additionally added, as shown in Table 6. The use of metered dose valve systems of different specifications (50 μ l for example) allows the aerosol to be produced in the required dose per actuation.
TABLE 6 Aerosol preparation examples 33 to 35(100 bottles)
The preparation process comprises the following steps: adding formoterol, lactose monohydrate, sorbic acid (or dodecane or polyvinylpyrrolidone) into ethanol and polyethylene glycol, adding liquid HFA227ea, stirring and mixing at low temperature (-80 to-60 ℃), canning according to the dosage, and sealing a quantitative valve system (50 μ l for example). The dose is 12 mug per actuation of formoterol and is 170 actuations in total. Inhalation performance measurements were performed for examples 33-35 and the results showed that: the samples of examples 33-35 had good effective lung deposition, with NGI-FPF above 38%; meanwhile, the DCU determination result also completely conforms to the pharmacopoeia regulation.
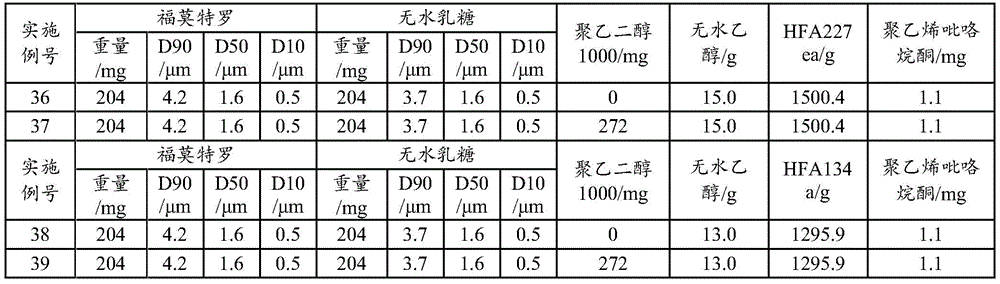
Examples 36 to 39: aerosol formulation (100 bottles)
The anhydrous lactose is lactose with the particle size of D90 being 3.7 mu m, D50 being 1.6 mu m and D10 being 0.5 mu m, the addition amount of the active ingredient is 0.013-0.015%, the addition amount of the anhydrous lactose is 0.010-0.012%, the solvents are absolute ethyl alcohol (1%) and polyethylene glycol 1000 (not added or 0.02%), the propellant is HFA134a or HFA227ea, the active ingredient is formoterol fumarate dihydrate with the particle size of D90 being 4.2 mu m, D50 being 1.6 mu m and D10 being 0.5 mu m, and the prescription ratio is 7 multiplied by 10-7~9×10-7Polyvinylpyrrolidone K25 as shown in table 7. Using different specificationsA metered dose valve system (63 μ l for example) allows the aerosol to be produced in the required dose per stroke.
TABLE 7 Aerosol preparation examples 36 to 39(100 bottles)
The preparation process comprises the following steps: adding formoterol, anhydrous lactose and polyvinylpyrrolidone K25 into ethanol and polyethylene glycol, adding liquid HFA134a or HFA227ea, stirring and mixing at low temperature (-80 to-60 ℃), canning according to the dosage, and sealing a quantitative valve system (63 μ l for example). The dose is 12 mug per actuation of formoterol and is 170 actuations in total. The results show that the samples of the examples 37 and 39 have good effective lung deposition, the NGI-FPF is much higher than that of the examples 36 and 38, meanwhile, the DCU determination results of the examples 36 and 38 do not accord with pharmacopeia regulations, the DCU determination results of the examples 37 and 39 completely accord with the pharmacopeia regulations, and the PEG is proved to be added to effectively improve the uniformity and the physical stability of the preparation.
Example 40: aerosol formulation (100 bottles)
The solvent is absolute ethyl alcohol (1%) and polyethylene glycol 1000 (0.02%), the propellant is HFA134a, the active ingredient is formoterol fumarate dihydrate which is micropowder until the particle size of D90 is 4.2 μm, the particle size of D50 is 1.6 μm and the particle size of D10 is 0.5 μm, the adding amount of the active ingredient is 0.015%, and the prescription proportion is 7 × 10-7~9×10-7Polyvinylpyrrolidone K25 as shown in table 8. The use of metered dose valve systems of different specifications (63 μ l for example) allows the aerosol to be produced in the required dose per actuation.
TABLE 8 Aerosol formulation example 40(100 bottles)
The preparation process comprises the following steps: adding formoterol and polyvinylpyrrolidone K25 into ethanol and polyethylene glycol according to the formula amount, adding liquid HFA134a, stirring and mixing at low temperature (-80 ℃ to-60 ℃), canning according to the dosage, and sealing a quantitative valve system (63 mu l for example) to obtain the compound. The dose is 12 mug per actuation of formoterol and is 170 actuations in total. The results show that the NGI-FPF of the sample of example 40 is below 38%; however, the DCU measurement result conforms to the pharmacopoeia regulation.
Aerosol formulation Process examples 41 and 42(100 bottles)
The preparation process comprises the following steps: adding the formoterol and the anhydrous lactose in the amount corresponding to the prescription in the example 4 into ethanol and polyethylene glycol, adding liquid HFA134a or HFA227ea, stirring and mixing at the low temperature of minus 30 ℃ and minus 100 ℃, canning according to the dose, and sealing and connecting a quantitative valve system (50 mu l for example) to obtain the compound. The dose is 12 mug per actuation of formoterol and is 170 actuations in total. The results show that during the specific operation of example 41, the temperature is already close to the boiling point of the propellant, so that the propellant is seriously volatilized during filling, and the content of the sample is directly abnormally high; in the specific operation of example 42, since the temperature is already close to the melting point of the propellant, the fluidity of the liquid medicine is deteriorated, and good uniformity of the liquid medicine cannot be ensured.
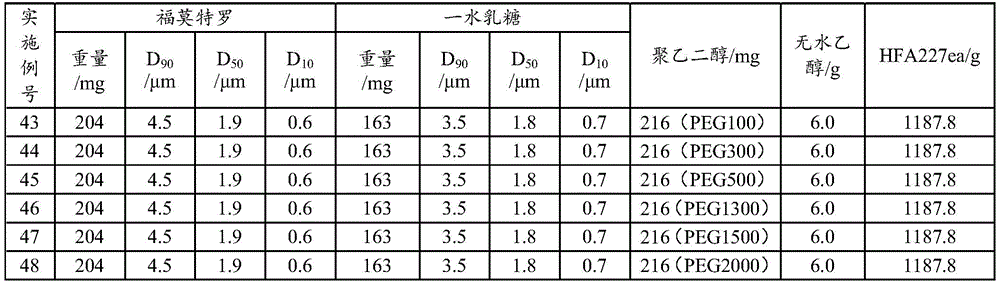
Examples 43 to 48: aerosol formulation (100 bottles)
The lactose monohydrate is in the form of micropowder D90Particle diameter of 3.5 μm, D50Particle diameter of 1.8 μm and D10Lactose with particle diameter of 0.7 μm, active ingredient addition amount of 0.017%, lactose addition amount of 0.014%, solvent including anhydrous ethanol (1%) and polyethylene glycol (0.0001% -0.1%), propellant including HFA227ea, and active ingredient including fine powder D90Particle diameter of 4.5 μm, D50Particle diameter of 1.9 μm and D10Formoterol fumarate dihydrate with a particle size of 0.6 μm, as shown in table 9. The use of metered dose valve systems of different specifications (50 μ l for example) allows the aerosol to be produced in the required dose per actuation.
TABLE 9 Aerosol preparation examples 43 to 48(100 bottles)
The preparation process comprises the following steps: adding formoterol and lactose monohydrate into ethanol and polyethylene glycol, adding liquid HFA134a or HFA227ea, stirring and mixing at low temperature (-80 to-60 ℃), canning according to the dose, and sealing a quantitative valve system (50 mu l for example) to obtain the compound. The dose is 12 mug per actuation of formoterol and is 170 actuations in total. Inhalation performance measurements were performed for examples 43-48 and the results showed that: the NGI-FPF of the samples of the examples 43-48 is below 38%, but the DCU measurement results all accord with the pharmacopoeia regulations.
Example 49
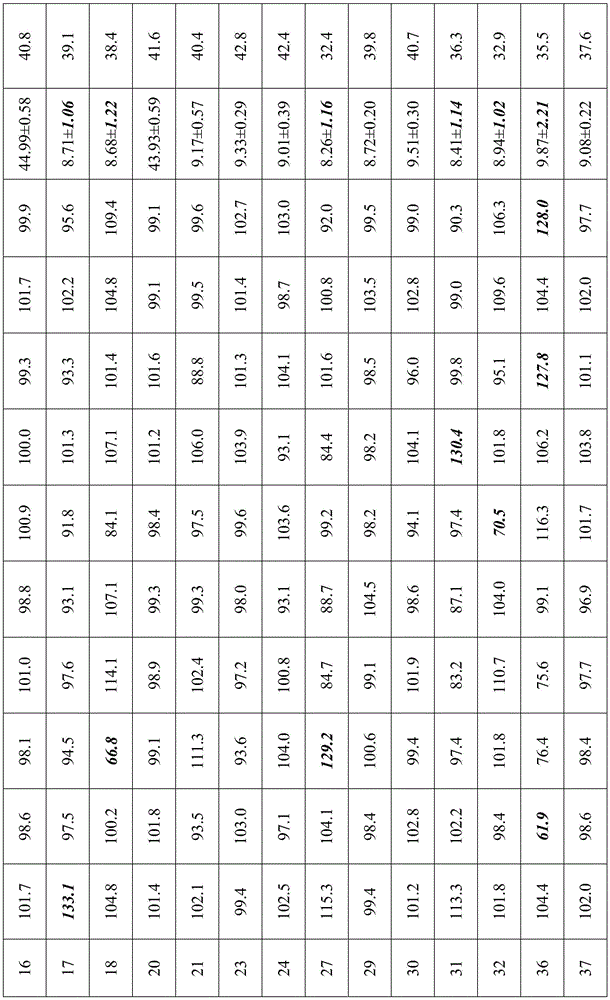
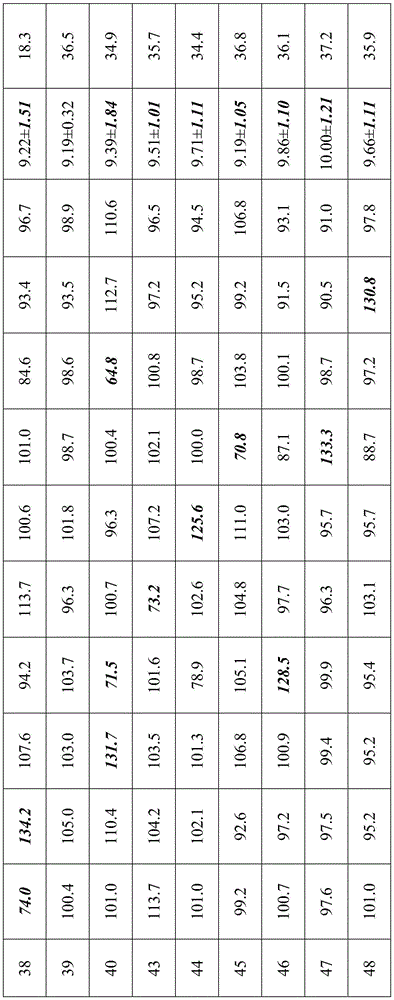
Samples prepared according to examples 3, 4, 5, 6, 7, 10, 11, 12, 13, 14, 15, 16, 17, 18, 20, 21, 23, 24, 27, 29, 30, 31, 32, 36, 37, 38, 39, 40, 43, 44, 45, 46, 47, 48 were taken for inhalation performance measurement, respectively, and included: (1) aerodynamic particle size determination of a sample using a new generation of cascade impactor (NGI) to define the effective lung deposition rate of a drug (NGI-FPF); (2) samples were tested for delivered dose uniformity (DCU) using a single dose sampling device. The results of the experiment are shown in table 10:
and (4) analyzing results:
1. all samples of examples 3, 4, 5, 10, 11, 12, 16, 17, 18, 20, 21, 23, 24, 29, 30 had NGI-FPF above 38%, demonstrating good effective lung deposition for the samples. Meanwhile, the DCU measurement results of the samples in the fifteen examples all accord with the pharmacopoeia regulation (according to the standard of the 2015 edition of Chinese pharmacopoeia, namely, according to one of the following conditions, the DCU measurement results can be judged to accord with the regulation, (1) if 2-3 measurement values in 10 measurement results exceed 75-125% but all exceed 65-135% of the average value, and 2 bottles of test samples are taken for measurement, if 30 doses exceed 75-125% of measurement values and all exceed 65-135% of the average value, the samples prepared according to the invention are proved to have good preparation uniformity and physical stability.
2. The NGI-FPF of the samples of example 15 was less than 38%, demonstrating that effective lung deposition was not ideal for the samples, and the NGI-FPF of the samples of examples 17 and 18 were both above 38%, demonstrating that the samples had good effective lung deposition. Meanwhile, the DCU measurement results of the samples in 3 groups of examples meet the pharmacopoeia regulations (according to the standard of the 2015 edition of Chinese pharmacopoeia, namely, one meeting the following conditions, namely, the DCU measurement results meet the regulations, (1) in 10 measurement results, not less than 9 measurement values are between 75% and 125% of the average value and all measurement values are between 65% and 135% of the average value, and (2) in 10 measurement results, if 2 to 3 measurement values exceed 75% and 125% and all measurement values are between 65% and 135% of the average value, 2 bottles of test articles are taken for measurement, if 30 doses exceed 75% and 125% and all measurement values are between 65% and 135% of the average value, 1 value is still between 65% and 135% of the average value, the SD values of 10 delivered dose data of the samples prepared in examples 15, 17 and 18 are calculated, and the SD values are respectively higher than 0.25 mug, 1.0 mug and 1.0 mug, the preparation uniformity and physical stability of the samples of the three examples are not ideal, and the results of comparing the samples of the other examples show that the effect of the examples when the weight ratio of API/lactose is in the range of 1: 0.1-1: 0.8 is better than that of the examples when the weight ratio of API/lactose is in the range of 1: 1-1: 5, and is better than that of the examples when the weight ratio of API/lactose is in the range of 1: 5-1: 20.
3. The NGI-FPF of the samples of examples 6, 7, 13 and 14 was below 38%, demonstrating that effective lung deposition was not ideal for the samples. Comparing examples 4 and 11, respectively, it is seen that the NGI-FPF gradually decreases with increasing particle size of the API/lactose. The particle size ranges of API in D90 of 0.1-5 μm, D50 of 0.1-3 μm and D10 of 0.1-1.5 μm in example 4 are superior to those in D90 of 0.1-7 μm, D50 of 0.1-5 μm and D10 of 0.1-3 μm in example 6, and are further superior to those in D90 of 0.1-10 μm, D50 of 0.1-7 μm and D10 of 0.1-5 μm in example 7. The effect of example 11 in which the particle size of lactose is 0.1 to 5 μm in D90, 0.1 to 3 μm in D50 and 0.1 to 1.5 μm is better than that of example 13 in which D90 is 0.1 to 10 μm, D50 is 0.1 to 5 μm and D10 is 0.1 to 3 μm, and is further better than that of example 14 in which D90 is 0.1 to 30 μm, D50 is 0.1 to 15 μm and D10 is 0.1 to 5 μm.
4. The NGI-FPF of the samples of examples 27, 31 and 32 was below 38%, demonstrating that effective lung deposition was not ideal for the samples. Meanwhile, the DCU measurement result of the sample of the embodiment accords with the pharmacopoeia regulation (according to the standard of the 2015 edition of Chinese pharmacopoeia, namely, one of the following conditions is met, the DCU measurement result of the sample of the embodiment can be judged to accord with the regulation, wherein in (1)10 measurement results, not less than 9 measurement values are between 75% and 125% of the average value and all measurement values are between 65% and 135% of the average value, in (2)10 measurement results, if 2 to 3 measurement values exceed 75% and 125% and all measurement values are between 65% and 135% of the average value, in addition, 2 bottles of test articles are taken for measurement, in 30 doses, the measurement values exceeding 75% and 125% are not more than 3 and all measurement values are between 65% and 135% of the average value, still 1 value is between 65% and 135% of the average value, the SD value of 10 delivery dose data of the samples prepared in the embodiments 27, 31 and 32 is calculated, the SD value is higher than 1.0 mu g, and the, comparing the results of the samples of the other examples, it is found that the effect of the example is better than that of the example containing 0.02% or more of PEG, and the effect of the example containing no PEG is worse than that of the example containing PEG.
5. The NGI-FPF for the samples of examples 43-48 was less than 38%, demonstrating that effective lung deposition is not ideal for the samples. Meanwhile, the DCU measurement result of the sample in the embodiment accords with the pharmacopoeia regulation (according to the standard of the 2015 edition of Chinese pharmacopoeia, namely, one of the following conditions is met, the DCU measurement result is judged to accord with the regulation, wherein in (1) in 10 measurement results, not less than 9 measurement values are between 75% and 125% of the average value and all measurement values are between 65% and 135% of the average value, and in (2) in 10 measurement results, if 2 to 3 measurement values exceed 75% and 125% but all measurement values are between 65% and 135% of the average value, 2 bottles of test articles are taken for measurement, if 30 doses exceed 75% and 125% measurement values are not more than 3 and all measurement values are between 65% and 135% of the average value, 1 value is still between 65% and 135% of the average value, the SD value of 10 delivered dose data of the prepared samples in the embodiments 43 to 48 is calculated, the SD is higher than 1.0 mu g, and the uniformity and the physical stability of the sample preparation, comparing the results of the samples of example 4, it can be seen that the effect of polyethylene glycol 1000 is superior to that of the other species.
6. The NGI-FPF of the samples of examples 37 and 39 are significantly higher than those of examples 36 and 38, indicating that the formulations of examples 37 and 39 are more homogeneous and stable, which is often a favorable factor in pharmaceutical research, particularly inhalation formulation research. While the delivered dose uniformity (DCU) was measured for each of the samples of examples 36-39, it was found from the results that the samples prepared in examples 36 and 38 did not comply with the pharmacopoeia specifications (according to the standard of the 2015 edition of Chinese pharmacopoeia, i.e. one of the following conditions is met, which was judged to be met by (1) no less than 9 measurements out of 10 measurements between 75% and 125% of the average and all between 65% and 135% of the average, and (2) if 2-3 measurements out of 10 measurements exceed 75% and 125% but all between 65% and 135% of the average, another 2 bottles of test samples were taken for measurement, if 30 doses exceed 75% and 125% and all between 65% and 135% of the average, no more than 3 measurements out of 75% and 125% were calculated for the SD values of 10 delivered dose data of the samples prepared in examples 36 and 38, and it was found that the SD values were all higher than 1.5. mu.g, the amount of the PEG is far larger than that of the samples prepared in examples 37 and 39, the drug is proved to be uneven in a preparation system, and the PEG is further proved to be effective in improving the uniformity and the physical stability of the preparation.
7. In the specific operation of example 41, the temperature is already close to the boiling point of the propellant, so that the propellant is seriously volatilized during filling, and the content of the sample is directly abnormally high; in the specific operation of example 42, since the temperature is already close to the melting point of the propellant, the fluidity of the liquid medicine is deteriorated, and good uniformity of the liquid medicine cannot be ensured. Therefore, the sample preparation temperature is preferably-60 ℃ to-80 ℃.
8. Compared with the prior art, the NGI-FPF of the formoterol dry powder inhalant developed by the Aslicon is between 21% and 37%, and the NGI-FPF of the formoterol solution type inhalant aerosol on the market in Kaixi is about 30%.
Claims (8)
1. An inhalation pressure metered dose aerosol pharmaceutical composition comprising as active ingredients formoterol or a pharmaceutically acceptable salt thereof, lactose, a solvent, polyethylene glycol (PEG) and a hydrofluoroalkane propellant;
wherein the pharmaceutically acceptable salt is selected from formoterol fumarate and formoterol fumarate dihydrate; the lactose is selected from one or two of lactose monohydrate and anhydrous lactose; the solvent is ethanol; the polyethylene glycol is PEG 1000; the hydrofluoroalkane propellant is selected from one or two of 1,1,1, 2-tetrafluoroethane (HFA 134a) and 1,1,1,2,3,3, 3-heptafluoropropane (HFA 227 ea);
d of the active ingredient90Particle size of 0.3 to 5 μm, D50Particle diameter of 0.2 to 2.5 μm, D10The particle size is 0.1-1 μm; d of said lactose90Particle size of 0.3 to 5 μm, D50Particle diameter of 0.2 to 2.5 μm, D10The particle size is 0.1-1 μm; the active ingredient accounts for 0.017-0.085% of the pharmaceutical composition by weight; the lactose accounts for 0.008-0.085% of the pharmaceutical composition by weight; the solvent accounts for 0-10% of the weight of the pharmaceutical composition; the weight percentage of the polyethylene glycol in the pharmaceutical composition is 0.018-0.02%.
2. Aerosol pharmaceutical composition according to claim 1, wherein the hydrofluoroalkane propellant is HFA227 ea.
3. Aerosol pharmaceutical composition according to claim 1 or 2, wherein the lactose represents 0.008 to 0.043% by weight of the pharmaceutical composition.
4. Aerosol pharmaceutical composition according to claim 1 or 2, wherein the lactose represents from 0.008% to 0.014% by weight of the pharmaceutical composition.
5. Aerosol pharmaceutical composition according to claim 1 or 2, wherein the solvent is present in an amount of 0 to 5% by weight of the pharmaceutical composition.
6. Aerosol pharmaceutical composition according to claim 1 or 2, wherein the solvent is present in an amount of 0 to 2% by weight of the pharmaceutical composition.
7. A process for the preparation of a pharmaceutical composition as claimed in any one of claims 1 to 6 for inhalation pressure metered dose aerosol comprising the steps of:
(a) micronizing active ingredient formoterol or pharmaceutically acceptable salt thereof and lactose;
(b) mixing the micronized product of the step (a), a solvent and polyethylene glycol, and closing a valve by a cover;
(c) dosing the mixture of step (b) with a prescribed amount of propellant through a valve.
8. A process for the preparation of a pharmaceutical composition as claimed in any one of claims 1 to 6 for inhalation pressure metered dose aerosol comprising the steps of:
(a) micronizing active ingredient formoterol or pharmaceutically acceptable salt thereof and lactose;
(b) dispersing the micronized product, the solvent and the polyethylene glycol in the step (a) into the precooled propellant, uniformly mixing at low temperature, and quantitatively subpackaging the liquid medicine into an aerosol can;
in the step (b), the low temperature is-80 ℃ to-60 ℃.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811522217.3A CN109464429B (en) | 2018-12-13 | 2018-12-13 | Inhalation pressure quantitative aerosol pharmaceutical composition and preparation method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811522217.3A CN109464429B (en) | 2018-12-13 | 2018-12-13 | Inhalation pressure quantitative aerosol pharmaceutical composition and preparation method thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN109464429A CN109464429A (en) | 2019-03-15 |
| CN109464429B true CN109464429B (en) | 2021-04-27 |
Family
ID=65675021
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201811522217.3A Active CN109464429B (en) | 2018-12-13 | 2018-12-13 | Inhalation pressure quantitative aerosol pharmaceutical composition and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN109464429B (en) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110946319A (en) * | 2019-11-20 | 2020-04-03 | 许达勇 | Nicotine propellant and preparation method thereof |
| CN110840864B (en) * | 2019-12-20 | 2022-02-22 | 广州健康元呼吸药物工程技术有限公司 | Beta 2 receptor agonist inhalation aerosol and product containing same |
| CN119555851B (en) * | 2025-02-06 | 2025-06-03 | 上海新黄河制药有限公司 | Method for measuring in-vitro dissolution curve of suspension for inhalation |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6054488A (en) * | 1996-06-11 | 2000-04-25 | 3M Innovative Properties Company | Medicinal aerosol formulations of formoterol |
| CN1310611A (en) * | 1998-06-19 | 2001-08-29 | 巴克·诺顿药物有限公司 | Pressurized metered dose inhalers and pharmaceutical aerosols |
| CN1455663A (en) * | 2000-07-11 | 2003-11-12 | 阿斯特拉曾尼卡有限公司 | Novel aerosol formulation containing polar fluorinated molecule |
| CN1622802A (en) * | 2002-02-01 | 2005-06-01 | 阿斯利康(瑞典)有限公司 | Composition for inhalation |
| CN1625392A (en) * | 2002-02-01 | 2005-06-08 | 阿斯利康(瑞典)有限公司 | Composition for inhalation |
| CN1635870A (en) * | 2000-10-09 | 2005-07-06 | 3M创新有限公司 | Medicinal Aerosol Preparations |
| CN101652123A (en) * | 2007-02-09 | 2010-02-17 | 先灵公司 | Stable pharmaceutical drug aerosols |
| CN102362860A (en) * | 2011-10-27 | 2012-02-29 | 江阴长风医药科技有限公司 | Budesonide and formoterol aerosol preparation taking hydro-fluoro-alkane as propellant |
| CN102670505A (en) * | 2011-03-17 | 2012-09-19 | 益得生物科技股份有限公司 | Process for preparing quantitative spray inhalant for treating respiratory diseases |
| BR102012027127A2 (en) * | 2012-10-23 | 2014-11-04 | Pablo Cassara Srl Lab | COMPOSITION CONTAINED IN A PHARMACEUTICAL INHALER PRESSURIZED WITH DOSAGE METERS |
| CN106581010A (en) * | 2016-12-28 | 2017-04-26 | 四川普锐特医药科技有限责任公司 | Aerosol preparation and quantitative inhalation aerosol |
| CN107666903A (en) * | 2015-05-01 | 2018-02-06 | 得克萨斯州大学系统董事会 | Multiple medicine fragility base composition |
-
2018
- 2018-12-13 CN CN201811522217.3A patent/CN109464429B/en active Active
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6054488A (en) * | 1996-06-11 | 2000-04-25 | 3M Innovative Properties Company | Medicinal aerosol formulations of formoterol |
| CN1310611A (en) * | 1998-06-19 | 2001-08-29 | 巴克·诺顿药物有限公司 | Pressurized metered dose inhalers and pharmaceutical aerosols |
| CN1455663A (en) * | 2000-07-11 | 2003-11-12 | 阿斯特拉曾尼卡有限公司 | Novel aerosol formulation containing polar fluorinated molecule |
| CN1635870A (en) * | 2000-10-09 | 2005-07-06 | 3M创新有限公司 | Medicinal Aerosol Preparations |
| CN1622802A (en) * | 2002-02-01 | 2005-06-01 | 阿斯利康(瑞典)有限公司 | Composition for inhalation |
| CN1625392A (en) * | 2002-02-01 | 2005-06-08 | 阿斯利康(瑞典)有限公司 | Composition for inhalation |
| CN101652123A (en) * | 2007-02-09 | 2010-02-17 | 先灵公司 | Stable pharmaceutical drug aerosols |
| CN102670505A (en) * | 2011-03-17 | 2012-09-19 | 益得生物科技股份有限公司 | Process for preparing quantitative spray inhalant for treating respiratory diseases |
| CN102362860A (en) * | 2011-10-27 | 2012-02-29 | 江阴长风医药科技有限公司 | Budesonide and formoterol aerosol preparation taking hydro-fluoro-alkane as propellant |
| BR102012027127A2 (en) * | 2012-10-23 | 2014-11-04 | Pablo Cassara Srl Lab | COMPOSITION CONTAINED IN A PHARMACEUTICAL INHALER PRESSURIZED WITH DOSAGE METERS |
| CN107666903A (en) * | 2015-05-01 | 2018-02-06 | 得克萨斯州大学系统董事会 | Multiple medicine fragility base composition |
| CN106581010A (en) * | 2016-12-28 | 2017-04-26 | 四川普锐特医药科技有限责任公司 | Aerosol preparation and quantitative inhalation aerosol |
Also Published As
| Publication number | Publication date |
|---|---|
| CN109464429A (en) | 2019-03-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| SA01220062B1 (en) | Pharmaceutical formulations for dry powder inhalers in the form of solid pellets | |
| EP2983645B1 (en) | Composition comprising at least two dry powders obtained by spray drying to increase the stability of the formulation | |
| CN109464429B (en) | Inhalation pressure quantitative aerosol pharmaceutical composition and preparation method thereof | |
| KR20130140358A (en) | Dry powder for inhalation formulation comprising salmeterol xinafoate, fluticasone propionate and tiotropium bromide, and method for preparing the same | |
| US10517828B2 (en) | Composition comprising at least one dry powder obtained by spray drying to increase the stability of the formulation | |
| CN111358773B (en) | Peramivir dry powder inhalant and preparation method thereof | |
| EP2211863A1 (en) | Combination therapy | |
| KR101728116B1 (en) | Dry powder composition for inhalation comprising tiotropium or pharmaceutically acceptable salt thereof | |
| CN111481550A (en) | Pharmaceutical formulation containing tiotropium bromide and arformoterol | |
| AU2021200396B2 (en) | Pharmaceutical composition containing budesonide and formoterol | |
| CN103520106A (en) | Salbutamol sulphate inhalation aerosol and preparation method thereof | |
| CN103110584A (en) | Tiotropium bromide powder inhalation and preparation method thereof | |
| CN104208688A (en) | Pharmaceutical composition containing individually-packaged mometasone furoate and epinephrine beta2-acceptor stimulant placed in inhalation device for synchronous application | |
| CN112137957B (en) | Medicinal inhalation aerosol and preparation method thereof | |
| CN101744792B (en) | Fluticasone propionate and salmeterol xinafoate compound dry powder inhalation and preparation technology thereof | |
| CN111939143A (en) | Budesonide solution type aerosol and preparation method thereof | |
| CN110840864B (en) | Beta 2 receptor agonist inhalation aerosol and product containing same | |
| CN101756943A (en) | Mometasone furoate dry powder inhalation and preparing process thereof | |
| CN108066329A (en) | A kind of preparation method of the particle of sucking fluticasone or derivatives thereof | |
| CN102379846B (en) | Fluticasone propionate aerosol preparation with hydrofluoroalkane and polyethylene glycol as auxiliary materials | |
| CN104208689A (en) | Pharmaceutical composition containing individually-packaged budesonide and epinephrine beta2-acceptor stimulant placed in inhalation device for synchronous application | |
| CN116139110A (en) | A kind of preparation method of suspension type compound inhalation aerosol | |
| CN104208686A (en) | Pharmaceutical composition containing individually-packaged fluticasone and epinephrine beta2-acceptor stimulant placed in inhalation device for synchronous application | |
| CN116392460A (en) | Salbutamol aerosol | |
| CN112933047A (en) | Preparation method of ultrafine pulverized salmeterol ticasone |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |