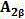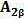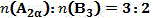CN109400894B - Hyperbranched polytriazole functional polymer material and preparation method thereof - Google Patents
Hyperbranched polytriazole functional polymer material and preparation method thereof Download PDFInfo
- Publication number
- CN109400894B CN109400894B CN201811156670.7A CN201811156670A CN109400894B CN 109400894 B CN109400894 B CN 109400894B CN 201811156670 A CN201811156670 A CN 201811156670A CN 109400894 B CN109400894 B CN 109400894B
- Authority
- CN
- China
- Prior art keywords
- azidoethoxy
- ethane
- hyperbranched
- functional polymer
- ptri1
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G83/00—Macromolecular compounds not provided for in groups C08G2/00 - C08G81/00
- C08G83/002—Dendritic macromolecules
- C08G83/005—Hyperbranched macromolecules
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/0605—Polycondensates containing five-membered rings, not condensed with other rings, with nitrogen atoms as the only ring hetero atoms
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Macromolecular Compounds Obtained By Forming Nitrogen-Containing Linkages In General (AREA)
Abstract
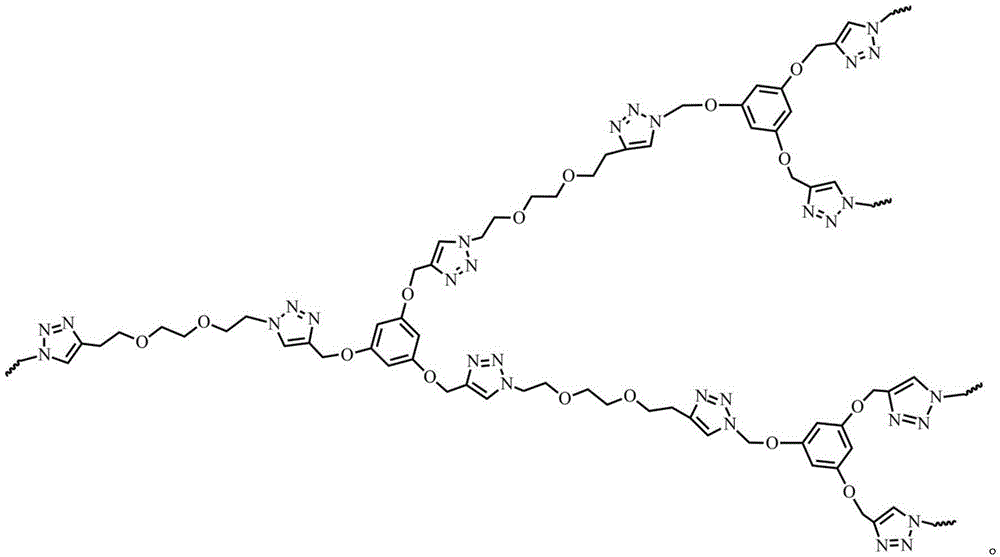
本发明涉及一种超支化聚三唑功能高分子材料及其制备方法。该功能高分子材料的结构式为下列之一:a.
b. 利用双叠氮单体(1‑叠氮基‑2‑(2‑叠氮基乙氧基)乙烷)或(1,2‑双(2‑叠氮基乙氧基)乙烷)和三端基炔单体B3(1,3,5‑三(2‑丙炔基‑1‑氧基)苯)为原料,以“A2+B3”法点击聚合得到两种超支化聚三唑功能高分子材料hb‑PTri1和hb‑PTri2。通过控制单体和单体的物质的量的比例及反应时间,得到了一系列超支化聚三唑功能高分子材料hb‑PTri1,并表征其结构和分子量。本发明原材料皆为工业级产品,价格低廉,制备工艺简单,易于实现大规模生产。The invention relates to a hyperbranched polytriazole functional polymer material and a preparation method thereof. The structural formula of the functional polymer material is one of the following: a.
b. Utilize bisazide monomers (1-azido-2-(2-azidoethoxy)ethane) or (1,2-bis(2-azidoethoxy)ethane) and tri-terminal alkyne monomers B 3 (1,3,5-tris(2-propynyl-1-oxy)benzene) As raw materials, two hyperbranched polytriazole functional polymer materials, hb -PTri1 and hb -PTri2, were obtained by click polymerization by "A 2 +B 3 " method. by controlling Monomer and A series of hyperbranched polytriazole functional polymer materials hb -PTri1 were obtained based on the ratio of the amount of monomers and the reaction time, and their structures and molecular weights were characterized. The raw materials of the invention are all industrial-grade products, the price is low, the preparation process is simple, and large-scale production is easy to be realized.Description
Technical Field
The invention relates to two hyperbranched polytriazole functional polymer materials and a preparation method thereof.
Background
With the development of science and technology and the continuous improvement of the living standard of people, the variety of novel functional materials required by social production and people's life is more and more. As a novel functional material, the hyperbranched polytriazole functional polymer material can be used as a biomedical material, a photoelectric functional material, a surface modification material and the like, and has important research significance and great commercial value.
The hyperbranched polymer is a large class of high molecular substances containing a large number of branching units, and has the structural characteristics of intramolecular topological cavities, multiple branching points, multifunctional end groups and the like. Hyperbranched polymers can also be regarded as special dendrimers, but possess more random branching structures and weaker regularity. The hyperbranched polymer can be prepared by a one-step polymerization method, and has the advantage of easy batch synthesis.
The hyperbranched polymer can be prepared by a single monomer process (By a process comprisingnA functional group A andmmonomers having B functional groupsSynthesis and preparation by the Bimonomer Process (By means of a process comprisingnMonomers having A functional groupsAnd comprisemMonomers having B functional groupsSynthesis). Customary bimonomers "A2+B3The "method" is to use monomers A containing 2A functional groups2And monomers B containing 3B functional groups3An example of the synthesis of hyperbranched polymer molecules.
In recent years, extensive research has been conducted on the azide and terminal alkyne cycloaddition reaction (so-called click reaction) catalyzed by monovalent cu (i) due to the advantages of mild reaction conditions, high conversion rate, environmental friendliness, simple method and the like. If proper polyfunctional azide and terminal alkyne are used as the raw materials of the click reaction, the hyperbranched polytriazole can be obtained. By adjusting the mass proportion and the reaction time of the monomers, a series of isomeric structures of the hyperbranched polytriazole can be obtained.
Disclosure of Invention
The invention aims to provide two hyperbranched polytriazole functional polymer materials.
The second purpose of the invention is to provide a preparation method of the functional polymer material. The process is carried out by2+B3Two novel hyperbranched polytriazole functional polymer materials are synthesized by a 'method click polymerization' one-pot method.
In order to achieve the purpose, the invention adopts the following technical scheme:
a hyperbranched polytriazole functional polymer material is characterized in that the structural formula of the functional polymer material is one of the following:
a hyperbranched prepared by the methodThe method for preparing the polytriazole functional polymer material is characterized by comprising the following specific steps: under the protection of inert atmosphere, 1-azido-2- (2-azidoethoxy) ethane (2)Monomer) or 1, 2-bis (2-azidoethoxy) ethane (b)Monomer) and 1,3, 5-tris (2-propynyl-1-oxy) benzene (A)Monomer) is dissolved in the mixture according to the molar ratio of 1: 1-3: 1N,N-Dimethylformamide (DMF), copper sulphate pentahydrate is added in an amount of0.1 molar amount of monomer and sodium ascorbate in an amount ofReacting for 12-30 hours at room temperature, wherein the molar weight of the monomer is 0.3; dropwise adding the reacted solution into 0.035mol/L disodium ethylene diamine tetraacetate (EDTA-2 Na) aqueous solution to precipitate solid; finally, washing the solid with methanol, filtering and drying to obtain the hyperbranched polytriazole functional polymer material; the 1-azido-2- (2-azidoethoxy) ethane (b)Monomer) is of the formula:
(ii) a The structural formula of the 1, 2-bis (2-azidoethoxy) ethane (monomer) is as follows:(ii) a The 1,3, 5-tri (2-propynyl-1-oxyl) benzene (monomer) junctionThe structure formula is as follows:。
in a word, the invention synthesizes two novel hyperbranched polytriazole functional materials in one step by a' one-pot methodhbPTri1 andhbthe PTri2 has the characteristics of easily obtained raw materials, simple process, convenient operation process and suitability for large-scale industrial production. The hyperbranched polytriazole prepared by the method is a new substance and is not reported in patents or documents at home and abroad.The invention utilizes the azide A with double functional groups2"monomeric and trifunctional terminal alkynes" B3"monomer, by" A2+B3"method for synthesizing two new hyperbranched polytriazoleshbPTri1 andhb-PTri 2) functional polymer material. The polymer containing triazole functional group can be used as a conjugated polymer, and shows wide research space in the aspect of detecting various metal ions by a fluorescence sensor. According to the inventionhbPTri1 andhbPTri2 as fluorescence sensor for Cr3+Detection of (3).
Drawings
FIG. 1 is a drawing ofA monomer,Monomer and hyperbranched polytriazole functional polymer materialhbOf PTri11H NMR spectrum. Wherein the synthesis conditions of sample S1 areReacting for 12 hours; the synthesis conditions for sample S2 wereReacting for 24 hours; the synthesis conditions for sample S3 wereReacting for 30 hours; the synthesis conditions for sample S4 wereReacting for 24 hours; the synthesis conditions for sample S5 wereReaction for 24 hours (solvent is DMSO-d)6);
FIG. 2 isA monomer,Monomer and hyperbranched polytriazole functional polymer materialhbOf PTri21H NMR spectrum under the synthesis conditions ofReaction for 30 hours (solvent is DMSO-d)6)。
Detailed description of the invention
The present invention will be described in detail with reference to examples.
The first embodiment is as follows: this example controls the mass ratio between the monomersChanging the reaction time, and synthesizing by one-pot methodhbPTri1 (i.e. sample S1), the procedure was as follows:
(1) in a 25 mL two-necked flask, 0.1452 g were sequentially addedMonomer (0.93 mmol), 0.1490 gMonomer (0.62 mmol), 0.0232 g CuSO4·5H2O (0.093 mmol), 0.0553 g NaAsc (0.28 mmol) and 4 mL DMF as solvents were stirred at 25 ℃ for 12 hours under nitrogen protection.
(2) The reacted solution was added dropwise to 0.035mol dm-3The aqueous solution of EDTA-2Na was stirred for 30 minutes and filtered to obtain a yellowish green powder. Washing the obtained yellowish green powder with 20 mL anhydrous methanol for 5 times, and vacuum drying at 60 deg.C for 24 hr to obtainhb-PTri1(S1)。
(3) Measurement of hyperbranched polytriazoleshb-molecular weight of PTri1 (S1). Will be provided withhbDissolving PTri1(S1) in DMF to give 2 mg mL-1Solution, determination of weight average molecular weight by gel chromatography (GPC) ((weight average molecular weight))) And polydispersity index (PDI). The sample introduction rate of the measurement is 2.22 muL min-1The eluent was DMF and the measurements are shown in table 1.
Example two: this example controls the mass ratio between the monomersChanging the reaction time, and synthesizing by one-pot methodhbPTri1 (i.e. sample S2), the procedure was as follows:
(1) in a 25 mL two-necked flask, 0.2998 g of the solution was sequentially addedMonomer (1.92 mmol), 0.3075 gMonomer (1.28 mmol), 0.0480 g CuSO4·5H2O (0.192 mmol), 0.1141 g NaAsc (0.58 mmol), 8.5 mL DMF as solvent, and the reaction was stirred at 25 ℃ for 24 hours under nitrogen protection.
(2) The reacted solution was added dropwise to 0.035mol dm-3The aqueous solution of EDTA-2Na was stirred for 30 minutes and filtered to obtain a yellowish green powder. Washing the obtained yellowish green powder with 45 mL anhydrous methanol for 5 times, and vacuum drying at 60 deg.C for 24 hr to obtainhb-PTri1(S2)。
(3) Testing of hyperbranched polytriazoleshbMolecular weight of PTri1(S2), the procedure was as in example one, and the results of the measurements are shown in table 1.
Example three: this example controls the mass ratio between the monomersChanging the reaction time, and synthesizing by one-pot methodhbPTri1 (i.e. sample S3), the procedure was as follows:
(1) in a 50mL two-necked flask, 1.0739 g were sequentially addedMonomer (6.88 mmol), 1.1027 gMonomer (4.59 mmol), 0.1720 g CuSO4·5H2O (0.69 mmol), 0.4081 g NaAsc (2.06 mmol) and 30 mL DMF as solvents were stirred at 25 ℃ under nitrogen for 30 hours.
(2) The reacted solution was added dropwise to 0.035mol dm-3The aqueous solution of EDTA-2Na was stirred for 30 minutes and filtered to obtain a yellowish green powder. Washing the obtained yellowish green powder with 150mL anhydrous methanol for 5 times, and vacuum drying at 60 deg.C for 24 hr to obtainhb-PTri1(S3)。
(3) Testing of hyperbranched polytriazoleshbMolecular weight of PTri1(S3), the procedure was as in example one, and the results of the measurements are shown in table 1.
Example four: this example varied the mass ratio between the monomersSynthesized by one-pot method in one stephbPTri1 (i.e. sample S4), the procedure was as follows:
(1) in a 25 mL two-necked flask, 0.1490 g were sequentially addedMonomer (0.62 mmol), 0.0968 gMonomer (0.62 mmol), 0.0155 g CuSO4·5H2O (0.062 mmol), 0.0368 g NaAsc (0.19 mmol) and 4 mL DMF as solvents were stirred at 25 ℃ under nitrogen for 24 hours.
(2) The reacted solution was added dropwise to 0.035mol dm-3The aqueous solution of EDTA-2Na was stirred for 30 minutes and filtered to obtain a yellowish green powder. Washing the obtained yellowish green powder with 20 mL anhydrous methanol for 5 times, and vacuum drying at 60 deg.C for 24 hr to obtainhb-PTri1(S4)。
(3) Testing of hyperbranched polytriazoleshbMolecular weight of PTri1(S4), the procedure was as in example one, and the results of the measurements are shown in table 1.
Example five: this example varied the mass ratio between the monomersSynthesized by one-pot method in one stephbPTri1 (i.e. sample S5), the procedure was as follows:
(1) in a 25 mL two-necked flask, 0.9744 g were sequentially addedMonomer (6.24 mmol), 0.4997 gMonomer (2.08 mmol), 0.1558 g of CuSO4·5H2O (0.62 mmol), 0.3709 g NaAsc (1.87 mmol) and 14 mL DMF as solvent, and the reaction was stirred at 25 ℃ under nitrogen for 24 hours.
(2) The reacted solution was added dropwise to 0.035mol-3The aqueous solution of EDTA-2Na was stirred for 30 minutes and filtered to obtain a yellowish green powder. Washing the obtained yellowish green powder with 75 mL anhydrous methanol for 5 times, and vacuum drying at 60 deg.C for 24 hr to obtainhb-PTri1(S5)。
(3) To be preparedhbMolecular weight testing of PTri1(S5) was carried out in the same manner as in example one, and the results are shown in Table 1.
Example six: this example varied the monomer species by controlling the mass ratio between the monomersSynthesized by one-pot method in one stephbPTri2, the specific procedure being as follows:
1. in a 50mL two-necked flask, 1.3767 g were sequentially addedMonomer (6.88 mmol), 1.1027 gMonomer (4.59 mmol), 0.1720 g CuSO4·5H2O (0.69 mmol), 0.4081 g NaAsc (2.06 mmol) and 30 mL DMF as solvents were stirred at 25 ℃ under nitrogen for 30 hours.
2. The reacted solution was added dropwise to 0.035mol dm-3The aqueous solution of EDTA-2Na was stirred for 30 minutes and filtered to obtain a yellowish green powder. Washing the obtained yellowish green powder with 150mL anhydrous methanol for 5 times, and vacuum drying at 60 deg.C for 24 hr to obtainhb-PTri2。
TABLE 1 attached, hyperbranched polytriazoleshbMolecular weight of PTri1
| hb-PTri1 | S1 | S2 | S3 | S4 | S5 |
| 3:2 | 3:2 | 3:2 | 1:1 | 3:1 | |
| Reaction time (hours) | 12 | 24 | 30 | 24 | 24 |
| (Da) | 21,000 | 35,000 | 28,000 | 25,000 | 16,000 |
| PDI | 1.74 | 1.40 | 1.82 | 2.15 | 1.82 |
| Yield (%) | 79.6 | 69.0 | 90.2 | 44.2 | 46.2 |
Method for testing fluorescence properties
Preparation ofhbOf PTri1(S3)N,N-dimethylacetamide (DMAc) solution (concentrated)Degree: 1.0X 10-5moldm-3) And then standby. Preparation of acetonitrile (CH) of various metal ions using lithium carbonate and other various metal salts (chlorate)3CN) solution (concentration: 1.0X 10-3moldm-3) And then standby. 3.0 mL of the solution was taken outhbPTri1(S3) DMAc solution and equal amounts of various metal salt solutions were added to the polymer solution at room temperature and sonicated for 2 hours to give various polymer-metal complexes.hbPTri1(S3) and all polymer-metal complexes were subjected to spectrofluorimetry.hbThe fluorescence spectrum test of PTri2 and all its polymer-metal complexes refers to the procedure described above. The results of the measurements are shown in Table 2.
TABLE 2 attached hereto, hyperbranched polytriazoleshbPTri1(S3), hyperbranched polytriazoleshbEfficiency of fluorescence quenching of PTri2 at various metal ions
| Quenching efficiency (%) | Cd | Cr | K | Li | Na | Ni | Pb |
| hb-PTri1(S3) | 44.0 | 91.3 | 45.1 | 42.9 | 45.8 | 68.9 | 46.5 |
| hb-PTri2 | 58.7 | 83.2 | 60.6 | 60.1 | 57.6 | 72.3 | 60.4 |
Claims (2)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2018106255572 | 2018-06-18 | ||
| CN201810625557 | 2018-06-18 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN109400894A CN109400894A (en) | 2019-03-01 |
| CN109400894B true CN109400894B (en) | 2021-01-12 |
Family
ID=65466739
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201811156670.7A Expired - Fee Related CN109400894B (en) | 2018-06-18 | 2018-09-30 | Hyperbranched polytriazole functional polymer material and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN109400894B (en) |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7772358B2 (en) * | 2006-09-06 | 2010-08-10 | The Hong Kong University Of Science And Technology | Polytriazoles constructed by 1,3-dipolar cycloaddition |
| CN102585220B (en) * | 2012-01-16 | 2013-07-24 | 浙江大学 | Hyperbranched polytriazole formate as well as preparation method and application thereof |
| US10420849B2 (en) * | 2014-09-14 | 2019-09-24 | Nanosynthons Llc | Pyrrolidone derivatives, oligomers and polymers |
| CN106519226B (en) * | 2015-09-11 | 2019-03-29 | 华东理工大学 | Polytriazoles resin derived from a kind of trifunctional alkynes and preparation method thereof |
| CN106243352B (en) * | 2016-07-29 | 2018-06-19 | 苏州大学 | A kind of polyphenylene triazole formic acid esters and preparation method and application |
| CN107325284B (en) * | 2017-07-02 | 2020-01-14 | 苏州大学 | Hyperbranched polyphenyl triazole formate as well as preparation method and application thereof |
-
2018
- 2018-09-30 CN CN201811156670.7A patent/CN109400894B/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| CN109400894A (en) | 2019-03-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN102731792B (en) | Preparation method of chitosan grafted polymer with thermosensitivity | |
| CN117700659A (en) | Covalent organic framework adsorbent, preparation method and application | |
| CN106967217A (en) | Polyimidazole class compound and its in-situ preparation method and application | |
| CN109160995A (en) | A kind of column [5] aromatic hydrocarbons self assembly elastomeric material and preparation method thereof | |
| US20250163189A1 (en) | Raft agent for photo-controlled polymerization, preparation method therefor, and applications thereof | |
| CN110330667B (en) | A kind of simple preparation method of water-soluble polyrotaxane crosslinking agent and product thereof | |
| CN101851306B (en) | Synthetic method of polystyrene macromolecular ligand | |
| CN109400894B (en) | Hyperbranched polytriazole functional polymer material and preparation method thereof | |
| CN111574649B (en) | Controllable synthesis method of highly stereoregular polymethyl methacrylate | |
| CN102617770A (en) | Preparation method of linkage polymer based on cucurbituril [6] | |
| CN109535302B (en) | Preparation method of high-temperature-resistant polyaniline modified ion exchange resin | |
| CN110423343B (en) | Polyarylether polymer containing photoelectric functional group, preparation method and application thereof | |
| Chen et al. | Synthesis of selenide-containing polymers by multicomponent polymerization based on γ-butyroselenolactone | |
| CN120040699A (en) | Covalent organic framework material and preparation method and application thereof | |
| CN106279677A (en) | Rich in heteroatomic phosphine amidine base polymer and multicomponent polymerization and the application of preparing this polymer | |
| Yamauchi et al. | Synthesis of acrylonitrile side chain-appended π-conjugated polymers by a Suzuki cross-coupling polycondensation and a Knoevenagel condensation, and their optical properties | |
| CN101811997B (en) | Water-soluble self-doping aniline oligomer and preparation method thereof | |
| CN101417984B (en) | Initiated core containing triazine and use thereof | |
| CN106279673B (en) | A kind of polyfuran compound and preparation method thereof | |
| CN117466823A (en) | Preparation method of one-pot 2- (4-aminophenyl) -5-aminobenzimidazole | |
| CN109851784A (en) | A kind of method for preparing 1,4-stereoregular polytriazole catalyzed by ruthenium complex | |
| CN114133391A (en) | Nuclei-substituted naphthalene diimide electrochemically polymerized monomer and preparation method thereof | |
| CN112940162B (en) | Synthetic method of highly-ordered dendritic heterogeneous sugar-containing polymer containing multiple glycosyl groups | |
| CN111471171A (en) | Aryl sulfonic acid intermediate, preparation method thereof and application thereof in synthesizing low-temperature-sensitivity additive for concrete | |
| CN116041691B (en) | Preparation method of structure-controllable polyion liquid |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20210112 |