CN108753625B - Polysaccharide-producing space Hericium coralloides ST21-3 and application thereof in improving biological immune activity - Google Patents
Polysaccharide-producing space Hericium coralloides ST21-3 and application thereof in improving biological immune activity Download PDFInfo
- Publication number
- CN108753625B CN108753625B CN201810510343.0A CN201810510343A CN108753625B CN 108753625 B CN108753625 B CN 108753625B CN 201810510343 A CN201810510343 A CN 201810510343A CN 108753625 B CN108753625 B CN 108753625B
- Authority
- CN
- China
- Prior art keywords
- hericium
- polysaccharide
- coralloides
- mycelium
- hericium coralloides
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 150000004676 glycans Chemical class 0.000 title claims abstract description 64
- 229920001282 polysaccharide Polymers 0.000 title claims abstract description 64
- 239000005017 polysaccharide Substances 0.000 title claims abstract description 64
- 244000126211 Hericium coralloides Species 0.000 title claims abstract description 45
- 235000000035 Hericium coralloides Nutrition 0.000 title claims abstract description 45
- 230000005965 immune activity Effects 0.000 title description 7
- 240000000588 Hericium erinaceus Species 0.000 claims abstract description 36
- 235000007328 Hericium erinaceus Nutrition 0.000 claims abstract description 36
- 238000004321 preservation Methods 0.000 claims abstract description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 28
- 238000000855 fermentation Methods 0.000 claims description 25
- 230000004151 fermentation Effects 0.000 claims description 25
- 230000000694 effects Effects 0.000 claims description 16
- 239000002609 medium Substances 0.000 claims description 16
- 239000001963 growth medium Substances 0.000 claims description 15
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 claims description 14
- 239000008103 glucose Substances 0.000 claims description 14
- 238000000034 method Methods 0.000 claims description 14
- 238000002360 preparation method Methods 0.000 claims description 13
- 239000000843 powder Substances 0.000 claims description 11
- 230000001965 increasing effect Effects 0.000 claims description 10
- 210000002865 immune cell Anatomy 0.000 claims description 8
- 238000012258 culturing Methods 0.000 claims description 6
- 230000036039 immunity Effects 0.000 claims description 6
- 229940041514 candida albicans extract Drugs 0.000 claims description 5
- 229910000402 monopotassium phosphate Inorganic materials 0.000 claims description 5
- 239000002904 solvent Substances 0.000 claims description 5
- 239000012138 yeast extract Substances 0.000 claims description 5
- 229920002472 Starch Polymers 0.000 claims description 4
- WRUGWIBCXHJTDG-UHFFFAOYSA-L magnesium sulfate heptahydrate Chemical compound O.O.O.O.O.O.O.[Mg+2].[O-]S([O-])(=O)=O WRUGWIBCXHJTDG-UHFFFAOYSA-L 0.000 claims description 4
- 229940061634 magnesium sulfate heptahydrate Drugs 0.000 claims description 4
- 235000019796 monopotassium phosphate Nutrition 0.000 claims description 4
- 235000019698 starch Nutrition 0.000 claims description 4
- 239000008107 starch Substances 0.000 claims description 4
- 238000009629 microbiological culture Methods 0.000 claims description 3
- PJNZPQUBCPKICU-UHFFFAOYSA-N phosphoric acid;potassium Chemical compound [K].OP(O)(O)=O PJNZPQUBCPKICU-UHFFFAOYSA-N 0.000 claims description 3
- 239000004480 active ingredient Substances 0.000 claims 1
- 244000005700 microbiome Species 0.000 abstract description 6
- 235000013305 food Nutrition 0.000 abstract description 5
- 241000233866 Fungi Species 0.000 abstract description 2
- 230000000813 microbial effect Effects 0.000 abstract description 2
- 231100000350 mutagenesis Toxicity 0.000 abstract description 2
- 238000002703 mutagenesis Methods 0.000 abstract description 2
- 238000012827 research and development Methods 0.000 abstract description 2
- MWUXSHHQAYIFBG-UHFFFAOYSA-N Nitric oxide Chemical compound O=[N] MWUXSHHQAYIFBG-UHFFFAOYSA-N 0.000 description 30
- 239000000243 solution Substances 0.000 description 27
- 239000012153 distilled water Substances 0.000 description 17
- 210000002540 macrophage Anatomy 0.000 description 15
- 210000004027 cell Anatomy 0.000 description 13
- 239000000047 product Substances 0.000 description 12
- 238000012360 testing method Methods 0.000 description 11
- 239000008055 phosphate buffer solution Substances 0.000 description 9
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 8
- 238000001914 filtration Methods 0.000 description 7
- 238000002156 mixing Methods 0.000 description 7
- 239000002244 precipitate Substances 0.000 description 7
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- 238000007865 diluting Methods 0.000 description 6
- 230000035772 mutation Effects 0.000 description 6
- 239000000725 suspension Substances 0.000 description 6
- 238000002835 absorbance Methods 0.000 description 5
- 230000001580 bacterial effect Effects 0.000 description 5
- 238000009835 boiling Methods 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- 239000006228 supernatant Substances 0.000 description 5
- 238000009777 vacuum freeze-drying Methods 0.000 description 5
- 238000005406 washing Methods 0.000 description 5
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 4
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 4
- 238000010009 beating Methods 0.000 description 4
- 210000004369 blood Anatomy 0.000 description 4
- 239000008280 blood Substances 0.000 description 4
- 239000003153 chemical reaction reagent Substances 0.000 description 4
- 238000001035 drying Methods 0.000 description 4
- 239000012091 fetal bovine serum Substances 0.000 description 4
- 230000002068 genetic effect Effects 0.000 description 4
- 239000000203 mixture Substances 0.000 description 4
- 238000011160 research Methods 0.000 description 4
- LPXPTNMVRIOKMN-UHFFFAOYSA-M sodium nitrite Chemical compound [Na+].[O-]N=O LPXPTNMVRIOKMN-UHFFFAOYSA-M 0.000 description 4
- 238000005303 weighing Methods 0.000 description 4
- 108020004463 18S ribosomal RNA Proteins 0.000 description 3
- 239000002028 Biomass Substances 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- 229930182555 Penicillin Natural products 0.000 description 3
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 235000019441 ethanol Nutrition 0.000 description 3
- 239000008188 pellet Substances 0.000 description 3
- 229940049954 penicillin Drugs 0.000 description 3
- 239000000523 sample Substances 0.000 description 3
- 241000894006 Bacteria Species 0.000 description 2
- 241000123222 Hericium Species 0.000 description 2
- QTENRWWVYAAPBI-YZTFXSNBSA-N Streptomycin sulfate Chemical compound OS(O)(=O)=O.OS(O)(=O)=O.OS(O)(=O)=O.CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@H]1[C@H](N=C(N)N)[C@@H](O)[C@H](N=C(N)N)[C@@H](O)[C@@H]1O.CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@H]1[C@H](N=C(N)N)[C@@H](O)[C@H](N=C(N)N)[C@@H](O)[C@@H]1O QTENRWWVYAAPBI-YZTFXSNBSA-N 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- 239000002158 endotoxin Substances 0.000 description 2
- 238000000227 grinding Methods 0.000 description 2
- 230000007365 immunoregulation Effects 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 238000011081 inoculation Methods 0.000 description 2
- 229920006008 lipopolysaccharide Polymers 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- 238000012216 screening Methods 0.000 description 2
- 235000010288 sodium nitrite Nutrition 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N sulfuric acid Substances OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 229920001817 Agar Polymers 0.000 description 1
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 1
- 208000006313 Delayed Hypersensitivity Diseases 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 208000007882 Gastritis Diseases 0.000 description 1
- 206010064571 Gene mutation Diseases 0.000 description 1
- 229920001503 Glucan Polymers 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- 206010021143 Hypoxia Diseases 0.000 description 1
- 239000007836 KH2PO4 Substances 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 208000008469 Peptic Ulcer Diseases 0.000 description 1
- 239000001888 Peptone Substances 0.000 description 1
- 108010080698 Peptones Proteins 0.000 description 1
- 102000004142 Trypsin Human genes 0.000 description 1
- 108090000631 Trypsin Proteins 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 208000030961 allergic reaction Diseases 0.000 description 1
- WQZGKKKJIJFFOK-PHYPRBDBSA-N alpha-D-galactose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-PHYPRBDBSA-N 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 230000003266 anti-allergic effect Effects 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 244000052616 bacterial pathogen Species 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 230000017531 blood circulation Effects 0.000 description 1
- 230000000747 cardiac effect Effects 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 239000006285 cell suspension Substances 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 239000012295 chemical reaction liquid Substances 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000029087 digestion Effects 0.000 description 1
- 239000012470 diluted sample Substances 0.000 description 1
- 238000007598 dipping method Methods 0.000 description 1
- 229910000397 disodium phosphate Inorganic materials 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 230000005714 functional activity Effects 0.000 description 1
- 229930182830 galactose Natural products 0.000 description 1
- 239000006481 glucose medium Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 230000004727 humoral immunity Effects 0.000 description 1
- 230000007954 hypoxia Effects 0.000 description 1
- 230000002519 immonomodulatory effect Effects 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 230000036737 immune function Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000002054 inoculum Substances 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000005486 microgravity Effects 0.000 description 1
- 230000000877 morphologic effect Effects 0.000 description 1
- 239000010413 mother solution Substances 0.000 description 1
- 230000003505 mutagenic effect Effects 0.000 description 1
- 230000036438 mutation frequency Effects 0.000 description 1
- 210000000822 natural killer cell Anatomy 0.000 description 1
- 239000013642 negative control Substances 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 230000000242 pagocytic effect Effects 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 208000011906 peptic ulcer disease Diseases 0.000 description 1
- 235000019319 peptone Nutrition 0.000 description 1
- OQUKIQWCVTZJAF-UHFFFAOYSA-N phenol;sulfuric acid Chemical compound OS(O)(=O)=O.OC1=CC=CC=C1 OQUKIQWCVTZJAF-UHFFFAOYSA-N 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 239000013641 positive control Substances 0.000 description 1
- GNSKLFRGEWLPPA-UHFFFAOYSA-M potassium dihydrogen phosphate Chemical compound [K+].OP(O)([O-])=O GNSKLFRGEWLPPA-UHFFFAOYSA-M 0.000 description 1
- LWIHDJKSTIGBAC-UHFFFAOYSA-K potassium phosphate Substances [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 1
- 230000035755 proliferation Effects 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 238000012113 quantitative test Methods 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 239000012488 sample solution Substances 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 238000012163 sequencing technique Methods 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 238000013112 stability test Methods 0.000 description 1
- 239000012086 standard solution Substances 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 230000001502 supplementing effect Effects 0.000 description 1
- 239000012588 trypsin Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N1/00—Microorganisms, e.g. protozoa; Compositions thereof; Processes of propagating, maintaining or preserving microorganisms or compositions thereof; Processes of preparing or isolating a composition containing a microorganism; Culture media therefor
- C12N1/14—Fungi; Culture media therefor
- C12N1/145—Fungal isolates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12R—INDEXING SCHEME ASSOCIATED WITH SUBCLASSES C12C - C12Q, RELATING TO MICROORGANISMS
- C12R2001/00—Microorganisms ; Processes using microorganisms
- C12R2001/645—Fungi ; Processes using fungi
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L31/00—Edible extracts or preparations of fungi; Preparation or treatment thereof
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/125—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives containing carbohydrate syrups; containing sugars; containing sugar alcohols; containing starch hydrolysates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0634—Cells from the blood or the immune system
- C12N5/0645—Macrophages, e.g. Kuepfer cells in the liver; Monocytes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P19/00—Preparation of compounds containing saccharide radicals
- C12P19/04—Polysaccharides, i.e. compounds containing more than five saccharide radicals attached to each other by glycosidic bonds
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/34—Sugars
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Genetics & Genomics (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Microbiology (AREA)
- Biotechnology (AREA)
- Biomedical Technology (AREA)
- Mycology (AREA)
- General Health & Medical Sciences (AREA)
- General Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- Polymers & Plastics (AREA)
- Nutrition Science (AREA)
- Immunology (AREA)
- Food Science & Technology (AREA)
- Botany (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Tropical Medicine & Parasitology (AREA)
- Virology (AREA)
- Molecular Biology (AREA)
- Gastroenterology & Hepatology (AREA)
- General Chemical & Material Sciences (AREA)
- Hematology (AREA)
- Cell Biology (AREA)
- Medicines Containing Plant Substances (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Coloring Foods And Improving Nutritive Qualities (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Abstract
The invention discloses space coralloid hericium erinaceus ST21-3 for producing polysaccharide and application thereof in improving biological immunocompetence. The preservation number of the coralloid Hericium erinaceus (Hericium coralloides) Fullarton-H-ST21-3 in the common microorganism center of China Committee for culture Collection of microorganisms is CGMCC No. 15379. The invention adopts space spacecraft of Tiangong No. 2 and Shenzhou No. 11 to carry hericium erinaceus which are returned to the ground and subjected to space mutagenesis, takes the original hericium erinaceus on the ground as a contrast, and breeds a strain with higher immunocompetence, thereby providing practical basis for the application of space hericium erinaceus mycelium polysaccharide in improving biological immunocompetence. Meanwhile, the invention widens the application range of space microorganisms to a certain extent and fills the gap of the research and development of the microbial functions of food space fungi.
Description
Technical Field
The invention relates to space coralloid hericium erinaceus ST21-3 for producing polysaccharide and application thereof in improving biological immunocompetence.
Background
Hericium Erinaceus Polysaccharides (HEP) are composed of glucose, galactose and mannose, are glucans formed by main chains connected through beta- (1, 3) glycosidic bonds and branched chains connected through beta- (1, 6) glycosidic bonds, and are important functional active substances produced by fermentation of Hericium erinaceus. The Hericium erinaceus polysaccharide comprises polysaccharide in mycelium cells and polysaccharide in fermentation liquor. The Hericium erinaceus mycelium polysaccharide has the biological activities of reducing blood sugar, reducing blood lipid, resisting tumor, resisting oxidation, etc., in addition to the immunoregulation activity. The Hericium erinaceus polysaccharide immunoregulation activity is shown in the following steps: can improve the activity of natural killer cells, promote the generation of delayed hypersensitivity (anti-allergic reaction), enhance the phagocytic function of macrophages, and has no obvious effect on humoral immunity and proliferation of immune organs. This indicates that the hericium erinaceus mycelium polysaccharide mainly regulates the immune function of the organism by enhancing the activity of immune cells. In addition, many researches prove that the hericium erinaceus polysaccharide has important functions in the aspects of treating gastritis and peptic ulcer, improving the hypoxia tolerance of organisms, increasing the cardiac blood output, accelerating the blood circulation of the organisms and the like, and is a functional bacterium for both food and medicine.
Currently, domestic and foreign scholars mainly focus on optimization of polysaccharide substance extraction conditions and analysis of contained substance structures, research on immunomodulatory activity of polysaccharide substances is still in the beginning stage, research on space microorganisms mostly stays in application to space pathogenic bacteria, saprophytic bacteria and pharmacy, and development of food space microorganisms is still in the sprouting stage.
Disclosure of Invention
The invention aims to provide space coralloid hericium erinaceus ST21-3 capable of producing polysaccharide and application thereof in improving biological immune activity.
The coralloid Hericium erinaceus (Hericium coralloides) Fularton-H-ST 21-3 is preserved in China general microbiological culture Collection center (CGMCC for short; address: No. 3 Hospital No.1 of North West Lu of the Chaoyang district, China academy of sciences, postal code: 100101) in 24 months and 4 in 2018, and the preservation number is CGMCC NO. 15379.
The invention also protects the application of the coralloid Hericium (Hericium coralloides) Fullarton-H-ST21-3 or the mycelium thereof in preparing products; the application of the product is (1) and/or (2) as follows:
(1) increasing the activity of immune cells;
(2) improving immunity of organism.
The invention also provides a preparation method of the hericium erinaceus mycelium polysaccharide, which comprises the following steps: culturing Hericium coralloides (Hericium coralloides) Fullarton-H-ST21-3 to obtain mycelium; extracting polysaccharide from mycelium to obtain the coralloid hericium erinaceus mycelium polysaccharide.
In the method, the Hericium coralloides (Hericium coralloides) Fullarton-H-ST21-3 is cultured by using a seed fermentation medium.
In the method, the culture condition is shaking culture at 28 ℃ and 160 r/min.
The method specifically comprises the following steps:
(a) inoculating Hericium coralloides (Hericium coralloides) Fullarton-H-ST21-3 into seed fermentation medium for culturing;
(b) after the step (a) is finished, transferring the culture into a seed fermentation culture medium according to the inoculation amount of 10% (volume percentage) for culture, and collecting mycelia; extracting polysaccharide from mycelium.
In the step (a), the culture condition is shaking culture at 28 ℃ and 160r/min, and the culture time is 12 days.
In the step (b), the culture condition is shaking culture at 28 ℃ and 160r/min, and the culture time is 5 days.
In the method, the method for collecting the mycelium specifically comprises the following steps: filtering the culture system with three layers of sterile gauze, and collecting mycelium (washing with sterile distilled water for 3-4 times, and vacuum freeze drying).
In the method, the method for extracting the polysaccharide from the mycelium specifically comprises the following steps: freeze-drying mycelium, crushing the mycelium into powder, mixing the powder with distilled water, extracting the mixture for 2 hours in a boiling water bath, filtering the mixture by three layers of gauze, collecting an extracting solution (the extracting solution can be combined after residues are repeatedly extracted), carrying out alcohol precipitation on the extracting solution (the condition can be that the extracting solution is placed at 4 ℃ for 12 hours), collecting precipitate, and drying the precipitate (constant temperature drying at 60 ℃ or vacuum freeze drying can be adopted) to obtain the polysaccharide.
The invention also protects the hericium erinaceus mycelium polysaccharide obtained by any one of the methods.
The invention also protects the application of the hericium erinaceus mycelium polysaccharide in preparing products; the application of the product is (1) and/or (2) as follows:
(1) increasing the activity of immune cells;
(2) improving immunity of organism.
The invention also provides a product, the active component of which is Hericium coralloides (Hericium coralloides) Fullarton-H-ST21-3 or mycelium thereof; the application of the product is (1) and/or (2) as follows:
(1) increasing the activity of immune cells;
(2) improving immunity of organism.
The invention also protects a product, the active component of which is the hericium erinaceus mycelium polysaccharide; the application of the product is (1) and/or (2) as follows:
(1) increasing the activity of immune cells;
(2) improving immunity of organism.
The invention also provides a kit for preparing the Hericium erinaceus mycelium polysaccharide, which comprises Hericium coralloides (Hericium coralloides) Fullarton-H-ST21-3 and a seed fermentation culture medium.
Any of the above products can be specifically food, medicine or health care product and the like.
Any one of the seed fermentation culture media consists of a solute and a solvent; the solutes and their concentrations in the seed fermentation medium were as follows: 15-25g/L of soluble starch, 25-35g/L of glucose, 5-15g/L of yeast extract powder, 0.5-0.7g/L of magnesium sulfate heptahydrate and 2.5-3.5g/L of potassium dihydrogen phosphate; the solvent is water.
The concentrations in the seed fermentation medium and in the seed fermentation medium are as follows: 20g/L of soluble starch, 30g/L of glucose, 10g/L of yeast extract powder, 0.6g/L of magnesium sulfate heptahydrate and 3.0g/L of potassium dihydrogen phosphate.
The solvent in any one of the above seed fermentation media may be distilled water.
Any of the above-described methods for increasing the activity of an immune cell may be specifically characterized by increasing the amount of NO released from macrophages.
The macrophage may specifically be RAW264.7 cells.
The invention adopts space craft No. 2 and 11, which are carried by spacecraft of Tiangong and 2 and returned to ground and mutagenized by space, and the Hericium erinaceus has certain biological properties (such as individual shape, colony characteristics, physiological and biochemical characteristics, immunocompetence and the like) and fermentation production performance (such as biomass, product quantity, enzyme activity, titer, fermentation speed and the like) and changes the gene mutation frequency to a certain extent so as to change the functional activity due to the mutagenic actions of space microgravity effect, high vacuum, extreme temperature difference, weak magnetic field, high-energy particle (electron, proton and heavy ion) radiation and the like.
According to the invention, the original hericium erinaceus on the ground is used as a reference, and the strain with higher immunocompetence is bred, so that a practical basis is provided for the application of the spatial hericium erinaceus mycelium polysaccharide in the improvement of the biological immunocompetence. Meanwhile, the invention widens the application range of space microorganisms to a certain extent and fills the gap of the research and development of the microbial functions of food space fungi.
Detailed Description
The following examples are given to facilitate a better understanding of the invention, but do not limit the invention. The experimental procedures in the following examples are conventional unless otherwise specified. The test materials used in the following examples were purchased from a conventional biochemical reagent store unless otherwise specified. The quantitative tests in the following examples, all set up three replicates and the results averaged.
Ground Hericium erinaceus (Hericium erinaceus) GT 21: china center for industrial microorganism preservation, number 14026, abbreviated as ground Hericium Erinaceus GT 21.
YEPD medium: 10g of yeast extract powder, 20g of peptone, 20g of glucose, 17g of agar and distilled water to a constant volume of 1000mL, and the pH value is 6.0.
Seed fermentation culture medium: 20g of soluble starch, 30g of glucose, 10g of yeast extract powder, 0.6g of magnesium sulfate heptahydrate, 3g of monopotassium phosphate and distilled water to a constant volume of 1000mL, and the pH value is natural.
The macrophages used in the examples were RAW264.7 cells, purchased from ATCC, usa.
Example 1 screening of spatial Hericium Erinaceus
Space mutagenesis of bacterial strains
The ground hericium erinaceus GT21 is carried back by spacecrafts of Tiangong No. 2 and Shenzhou No. 11 to obtain a plurality of space mutation strains.
Secondly, activation of the strains
Taking a space mutation strain frozen in a-80 ℃ ultra-low temperature refrigerator and a ground hericium erinaceus GT21 glycerol tube storage strain, dipping a small amount of bacterial liquid by an inoculating ring, streaking, inoculating on a YEPD culture medium test tube inclined plane, culturing for 6 days at 28 ℃, continuously activating for 3 generations, and then using for subsequent tests.
Third, the enlargement culture of the bacterial strains
Inoculating the space mutation strain activated in the second step for 3 generations and ground hericium GT21 into a YEPD culture medium triangular flask inclined plane, and culturing at 28 ℃ for 6 days.
Fourthly, separation and purification of bacterial strains
And after the third step is finished, taking the space mutation strain and the ground hericium erinaceus GT21 which grow well on the inclined plane of the YEPD culture medium triangular flask, washing surface spores with 100mL of sterile normal saline, beating for 1min by using a sterile homogenizer according to 8 times/sec to fully separate the spores from the mycelia, and filtering by using three layers of sterile gauze to obtain a pure spore suspension. Taking 1mL of spore suspension, diluting with 9mL of sterile physiological saline by 10-fold gradient, and taking 10-2The spores were diluted and plated on YEPD medium plates and incubated at 28 ℃ for 6 days. Observing the colonial morphology of space mutation strain and ground Hericium Erinaceus GT21, and selecting bacteriaThe space mutation strain single colony with the colony characteristic greatly different from the ground strain is purified and cultured on a YEPD culture medium test tube inclined plane and cultured for 4 days at 28 ℃ to obtain the strains with the numbers of ST21-1 to ST 21-10.
Fifthly, re-screening of strains
1. Preparation of mycelia
Inoculating a strain to be tested to a seed fermentation culture medium, carrying out shaking culture at 28 ℃ and 160r/min for 12 days, beating a sterile homogenizer for 8 times/sec for 1min, beating mycelium pellets, transferring the mycelium pellets into the seed fermentation culture medium according to the inoculation amount of 10% (volume percentage), carrying out shaking culture at 28 ℃ and 160r/min for 5 days, taking a fermentation system, filtering by using three layers of sterile gauze, collecting mycelia, washing the obtained mycelia with sterile distilled water for 3-4 times, and carrying out vacuum freeze drying.
2. Preparation of mycelium crude polysaccharide
Grinding the freeze-dried mycelia in the step 1 into powder, weighing 1g of a sample, adding 18mL of distilled water, extracting for 2h in a boiling water bath, filtering with three layers of gauze, repeatedly extracting filter residues for 1 time, combining extracting solutions, fixing the volume of the extracting solution in 20mL of distilled water, adding 300mL of absolute ethyl alcohol, shaking up, standing at 4 ℃ for 12h, centrifuging at 10000r/min for 20min, collecting precipitates, centrifuging and washing with 95% of ethyl alcohol for 2 times (the centrifugation conditions are the same as above), and drying the precipitates at the constant temperature of 60 ℃ until the mass is constant, thereby obtaining the crude mycelia polysaccharide.
3. Reagent preparation
(1) Preparing a double-antibody solution: 3mg of penicillin solid powder was dissolved in 100mL of distilled water to prepare a 30. mu.g/mL penicillin solution. 10mg of streptomycin solid powder was dissolved in 100mL of distilled water to prepare a 100. mu.g/mL streptomycin solution. Mixing the penicillin solution and the streptomycin solution in equal volume to obtain double-antibody solution, and storing at-20 deg.C.
(2) Preparation of PBS (phosphate buffer solution): taking 0.14mol/L NaCl, 0.0027mol/L KCl and 0.004 mol/L Na2HPO4And 0.002mol/L KH2PO4The mixture was mixed in equal volumes, adjusted to pH 7.40 with 1mol/L NaOH, sterile filtered through a 0.22 μm filter and diluted with distilled water to a PBS concentration of 10 μ g/mL for use.
(3) The kit for detecting nitric oxide Griess comprises: biyuntian corporation, stored at 4 ℃.
4. Preparation of culture medium
DMEM complete medium (volume percent): and (3) uniformly mixing 89% (volume percentage content) of DMEM high-sugar medium, 10% (volume percentage content) of FBS (fetal bovine serum) and 1% (volume percentage content) of double-antibody solution to obtain the DMEM complete medium.
DMEM high-glucose medium and FBS (fetal bovine serum) were purchased from Shanghai Biotech engineering, Inc.
5. Preparation of samples
Accurately weighing 5mg of the mycelium crude polysaccharide prepared in the step 2 in a sterile centrifuge tube, preparing a 5mg/mL polysaccharide solution by using PBS (phosphate buffer solution) with pH7.4, centrifuging for 30min at 12000r/min, collecting supernatant, transferring the supernatant into a 1.5mL sterile centrifuge tube in a sterile operation, and diluting a polysaccharide sample to 1mg/mL, 500 mu g/mL and 250 mu g/mL by using PBS with pH 7.4.
6. Preparation of macrophage suspension
Selecting macrophage in logarithmic phase, sucking out culture solution from cell culture flask, adding 1ml PBS (pH7.4) for cleaning, sucking out supernatant, adding 0.25% trypsin, placing the flask in 5% CO2The cells were suspended in an incubator at 37 ℃ for 6min, 1mL of DMEM complete medium was added to terminate the digestion reaction, and the cells were aspirated with a pipette. Transferring the cell suspension into 15mL sterile centrifuge tube, centrifuging at 1000r/min for 5min, removing supernatant, adding 1mL PBS (pH7.4) to wash the precipitate, centrifuging at 1000r/min for 5min, removing supernatant, adding DMEM complete medium to suspend the cells, counting with a blood counting cell plate, diluting the cells to 5 × 10 with DMEM complete medium5cell/mL, namely macrophage suspension.
7. Nitric Oxide (NO) concentration determination
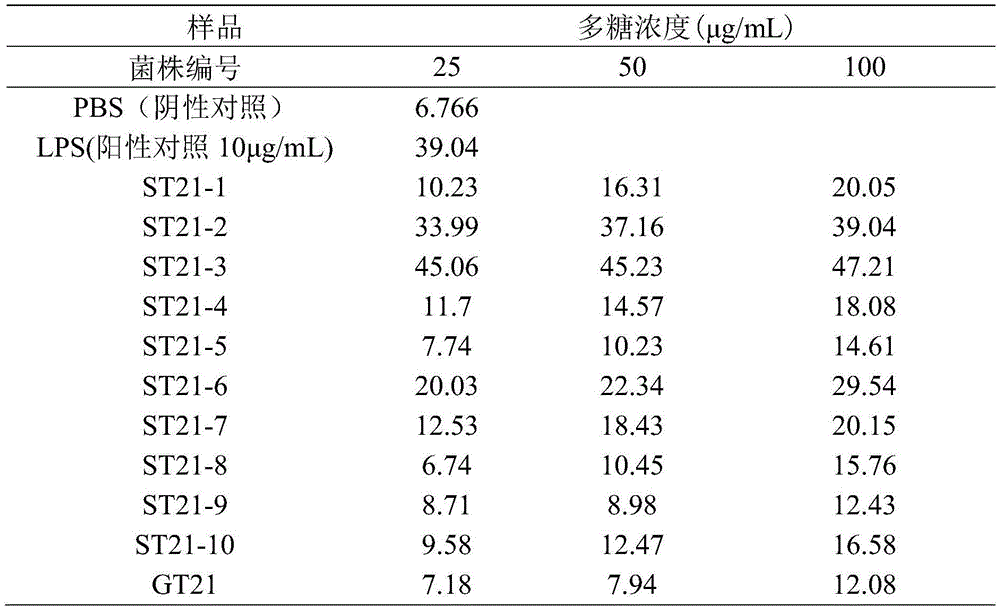
The macrophage suspension prepared in step 6 was added to 48-well plates (purchased from Coster Co., Ltd.) in an amount of 180. mu.L, 20. mu.L of the polysaccharide samples obtained in step 5 at different concentrations were added to each well, PBS (10. mu.g/mL) at pH7.4 was used as a negative control, LPS (10. mu.g/mL bacterial lipopolysaccharide, Wawter) was used as a positive control, and the mixture was washed with water and washed with water at 37 ℃ to obtain a suspension℃、 5%CO2Culturing in an incubator for 48h to obtain macrophage treating liquid. Diluting the sodium nitrite standard substance in the Griess kit into different gradients by using a DMEM complete culture medium, respectively adding the sodium nitrite standard substance and macrophage HEP treatment solution into another 48-hole plate according to the addition amount of 200 mu L/hole, then respectively adding the Griess Reagent I and Griess Reagent II into the kit according to the addition amount of 200 mu L/hole at room temperature, and measuring the absorbance value (A value) at 540 nm. The results are shown in Table 1.
TABLE 1 Effect of mycelium polysaccharide solutions of different concentrations on NO release (μmol/L) from RAW264.7 cells
As can be seen from Table 1, the amounts of NO released from RAW264.7 macrophages stimulated by the crude hyphal polysaccharides of the ST21-1 to ST21-10 strains were significantly different and increased with the increase in the concentration of the hyphal polysaccharide solution, wherein the NO released from RAW264.7 macrophages stimulated by the crude hyphal polysaccharides of the ST21-3 strain was higher than those of the other strains at each concentration. Because the NO release amount of macrophages is in positive correlation with in vitro immune activity, hypha polysaccharide produced by the ST21-3 strain has the strongest stimulation effect on RAW264.7 macrophages, the NO release amount is 47.21 mu mol/L, which is 3.91 times of NO release amount of ground strain GT21 mycelium polysaccharide acting on RAW264.7 cells, and the ST21-3 strain has higher in vitro immune activity and can become the most valuable strain for improving biological immune activity.
Sixthly, morphological identification and molecular biological identification of ST21-3 strain
1. Colonial characteristics of the ST21-3 strain: the colony is white, slightly yellowish, felt-like, and has many irregular spherical bulges on the surface, and the aerial mycelium is wool-like. The back gradually changes from white to yellow and yellow brown from the center of the colony, and the color change gradually enlarges and deepens.
2. The 18S rDNA of the ST21-3 strain is detected, and the sequencing result is shown as the sequence in the sequence table 1. The 18s rDNA identification showed that the ST21-3 strain was more than 99% similar to Hericium coralloides. Through morphology and 18S rDNA identification, the ST21-3 strain can be determined to belong to the coralloid hericium erinaceus.
Collection of seven, ST21-3 strains
The strain ST21-3 is named as Hericium coralloides (Hericium coralloides) Fullarton-H-ST21-3, and is preserved in China general microbiological culture Collection center (CGMCC for short; address: No. 3 of West Lu No.1 of Beijing university Hokko at sunward, China academy of sciences Microbiol research, postal code: 100101) 24 days in 2018, wherein the preservation number is CGMCC NO. 15379. Hericium coralloides (Hericium coralloides)
Fullarton-H-ST21-3 is abbreviated as Hericium coralloides SST 21-3.
Eighth, genetic stability
Subculturing the Hericium coralloides SST21-3 strain, performing a genetic stability test, and determining the influence of mycelium polysaccharide solutions with different concentrations in each generation on NO release amount of macrophage RAW264.7 cells to judge the genetic stability, wherein the results are shown in Table 2.
TABLE 2 influence of the 10 th generation different concentrations of mycelium polysaccharide solutions of ST21-3 on NO release from RAW264.7 cells (. mu. mol/L)
As can be seen from Table 2, after the passage of ST21-3 to 10, the NO release amount is still at a high level, which indicates that the immunocompetence of polysaccharide solutions with different concentrations on RAW264.7 cells is at a high level, and the genetic stability of Hericium coralloides SST21-3 in the aspect of improving the immunobiological activity can be basically judged.
Example 2 measurement of polysaccharide from Hericium erinaceum mycelium ST21-3
1. Preparation of mycelia
Inoculating Hericium coralloides SST21-3 to a seed fermentation culture medium, performing shaking culture at 28 deg.C and 160r/min for 12 days, beating with an aseptic homogenizer for 8 times/sec for 1min to disperse mycelium pellets, transferring into the seed fermentation culture medium according to the inoculum size of 10% (volume percentage), performing shaking culture at 28 deg.C and 160r/min for 5 days, collecting the fermentation system, filtering with three layers of aseptic gauze, collecting mycelia, washing the obtained mycelia with aseptic distilled water for 3-4 times, and vacuum freeze-drying.
2. Preparation of mycelium crude polysaccharide
Grinding the freeze-dried mycelia in the step 1 into powder, weighing 1g of a sample, adding 18mL of distilled water, extracting for 2h in a boiling water bath, filtering with three layers of gauze, repeatedly extracting filter residues for 1 time, combining extracting solutions, fixing the volume of the extracting solution in 20mL of distilled water, adding 300mL of absolute ethyl alcohol, shaking up, standing at 4 ℃ for 12h, centrifuging at 10000r/min for 20min, collecting precipitates, and carrying out vacuum freeze drying on the precipitates to obtain crude mycelia polysaccharide.
3. Drawing of standard curve
And (3) drying standard glucose at 105 ℃ to constant weight, preparing a 10mg/mL glucose standard product mother solution, and diluting 200 times to 50 mug/mL with constant volume by using a volumetric flask, thus obtaining the glucose standard solution. Respectively sucking 0, 0.2, 0.4, 0.6, 0.8 and 1.0mL of standard glucose solution, placing the standard glucose solution in a test tube, supplementing the standard glucose solution to 1mL by distilled water, then adding 0.5mL of 5% (mass percent) phenol aqueous solution, quickly adding 2.5mL of concentrated sulfuric acid (mass fraction of 98%) after uniformly mixing, fully mixing the reaction liquid by using a vortex oscillator, then placing the test tube in a boiling water bath for reacting for 15min, cooling the water bath for 10min to room temperature, measuring the absorbance value (A value) of the test tube at 490nm by using an ultraviolet spectrophotometer, and drawing a standard curve by using the glucose mass concentration as an abscissa and the absorbance value as an ordinate. The regression equation for the standard curve is y-0.0152 x +0.0208 (R)2=0.9995)。
4. Determination of Total sugar content
The total sugar content measured by the phenol-sulfuric acid method is the polysaccharide content. And (3) weighing 10mg of the crude polysaccharide prepared in the step (2) by using a ten-thousandth electronic balance, dissolving the crude polysaccharide in 50mL of distilled water, and then diluting the crude polysaccharide with the distilled water until the polysaccharide concentration is within the range of a standard curve to obtain a sample solution to be detected. Taking 1mL of diluted sample solution to be detected into a test tube, adding 0.5mL of 5% (mass percent) phenol solution, quickly adding 2.5mL of concentrated sulfuric acid (mass fraction of 98%) after uniformly mixing, fully mixing the reaction solution by using a vortex oscillator, then placing the test tube into a boiling water bath for reacting for 15min, cooling the test tube in the water bath for 10min to room temperature, and measuring the absorbance value (A value) of the test tube at 490nm by using an ultraviolet spectrophotometer. And substituting the measured absorbance y value into a glucose standard curve regression equation to obtain the polysaccharide content x value (mg/mL). The results of polysaccharide from Hericium coralloides ST21-3 mycelium are shown in Table 3.
Because the small molecules of the crude polysaccharide are removed by alcohol precipitation during preparation, the content of reducing sugar in the crude polysaccharide is extremely low and can be ignored.
The total content of hypha polysaccharide is equal to fermentation hypha biomass multiplied by hypha polysaccharide content/100.
TABLE 3 Hericium coralloides ST21-3 mycelium polysaccharide results
As can be seen from Table 3, the biomass of mycelium of Hericium coralloides SST21-3 was 3.52g/100mL, the polysaccharide content was 1.08mg/100mL, and the total amount of polysaccharide was 38.17mg/100 mL.
Sequence listing
<110> Fuleton biotechnology and technology (Beijing) Ltd
<120> space coralloid hericium coralloid ST21-3 for producing polysaccharide and application thereof in improving biological immune activity
<160> 1
<170> SIPOSequenceListing 1.0
<210> 1
<211> 648
<212> DNA
<213> Hericium coralloides (Hericium coralloides)
<400> 1
ccttccgtaa gggggcctgc ggaaggatca ttaatgaatt tgaaaggagt tttgttgctg 60
gcttgtcaac ccaggcatgt gcacactccg atctcatcca tcttacacct gtgcaccctt 120
gcgtgggtct gtcggctttg cggtvgttcg ggcttgcgtt ttttctataa acttttatgt 180
agtaacagaa tgtcttaaat gctataaacg catcttatac aactttcaac aacggatctc 240
ttggctctcg catcgatgaa gaacgcagcg aaatgcgata agtaatgtga attgcagaat 300
tcagtgaatc atcgaatctt tgaacgcacc ttgcgcccct tggtattccg agggcacgcc 360
tgttcgagtg tcgtgaaatt ctcaactcaa tcctcttgtt atgagagggc tgggcttgga 420
cttggaggtc ttgccggtgg ttccttcggg accgtcggct cctcttgaat gcatgagtgg 480
atcccttttt gtagggtttg cccttggtgt gataatatct acgccgcggg tagccttgcg 540
cgctggtctg cttctaaccg tccttcggga catgtttttc atctcaactt gacctcgaat 600
caggctgctg ccgctgattt gcccaagcat atcaatacgc gaaggaaa 648
Claims (7)
1. Hericium coralloides (Hericium coralloides) Fullarton-H-ST21-3, wherein the preservation number of the Hericium coralloides (Hericium coralloides) in the China general microbiological culture Collection center is CGMCC No. 15379.
2. Use of the Hericium coralloides (Hericium coralloides) Fullarton-H-ST21-3 or its mycelium for the preparation of a product; the application of the product is (1) and/or (2) as follows:
(1) increasing the activity of immune cells;
(2) improving immunity of organism.
3. A preparation method of hericium erinaceus mycelium polysaccharide comprises the following steps: culturing Hericium coralloides (Hericium coralloides) Fullarton-H-ST21-3 to obtain mycelium, and extracting polysaccharide from the mycelium to obtain the mycelium polysaccharide of Hericium coralloides.
4. A method as claimed in claim 3, comprising the steps of: in the method, the Hericium coralloides (Hericium coralloides) Fullarton-H-ST21-3 is cultured in a seed fermentation medium;
the seed fermentation culture medium consists of solute and solvent; the solutes and their concentrations in the seed fermentation medium were as follows: 15-25g/L of soluble starch, 25-35g/L of glucose, 5-15g/L of yeast extract powder, 0.5-0.7g/L of magnesium sulfate heptahydrate and 2.5-3.5g/L of potassium dihydrogen phosphate; the solvent is water.
5. The method of claim 3 or 4, wherein: in the method, the culture condition is shaking culture at 28 ℃ and 160 r/min.
6. A product comprising as an active ingredient the Hericium coralloides (Hericium coralloides) Fullarton-H-ST21-3 or a mycelium thereof according to claim 1; the application of the product is (1) and/or (2) as follows:
(1) increasing the activity of immune cells;
(2) improving immunity of organism.
7. A kit for preparing a mycelia polysaccharide of Hericium erinaceus comprising Hericium coralloides (Hericium coralloides) Fullarton-H-ST21-3 of claim 1 and the seed fermentation medium of claim 4.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201810510343.0A CN108753625B (en) | 2018-05-24 | 2018-05-24 | Polysaccharide-producing space Hericium coralloides ST21-3 and application thereof in improving biological immune activity |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201810510343.0A CN108753625B (en) | 2018-05-24 | 2018-05-24 | Polysaccharide-producing space Hericium coralloides ST21-3 and application thereof in improving biological immune activity |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN108753625A CN108753625A (en) | 2018-11-06 |
| CN108753625B true CN108753625B (en) | 2021-11-30 |
Family
ID=64005839
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201810510343.0A Active CN108753625B (en) | 2018-05-24 | 2018-05-24 | Polysaccharide-producing space Hericium coralloides ST21-3 and application thereof in improving biological immune activity |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN108753625B (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11920126B2 (en) | 2018-03-28 | 2024-03-05 | Ecovative Design Llc | Bio-manufacturing process |
| US11932584B2 (en) | 2006-12-15 | 2024-03-19 | Ecovative Design Llc | Method of forming a mycological product |
| US12161069B2 (en) | 2015-04-15 | 2024-12-10 | Ecovative Llc | High density rigid molded body of composite mycological material |
| US12503576B2 (en) | 2017-03-31 | 2025-12-23 | Ecovative Llc | Solution based post-processing methods for mycological biopolymer material and mycological product made thereby |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109609391B (en) * | 2019-02-15 | 2023-03-17 | 福州大学 | Quantitative inoculation method of mycelium pellets |
| CN116515641B (en) * | 2023-04-26 | 2023-09-29 | 吉林农业大学 | A coral-like hericium and its applications |
| CN118184805A (en) * | 2024-02-01 | 2024-06-14 | 吉林农业大学 | A coral-like Hericium erinaceus polysaccharide and its preparation method and application |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20040035985A (en) * | 2002-10-14 | 2004-04-30 | (주)에이비아이 | Composition for growth promotion by increasing expression of growth hormone and weight gain in quantity |
| KR101066429B1 (en) * | 2011-01-24 | 2011-09-21 | 장효준 | Method for preparing mycelia and fruiting bodies of Rhizome oleracea with improved antioxidant, immune and anticancer effects |
| CN103725730A (en) * | 2013-12-27 | 2014-04-16 | 内蒙古科技大学 | Refining technology of mycelia polysaccharides of selenium-enriched hericium erinaceus |
-
2018
- 2018-05-24 CN CN201810510343.0A patent/CN108753625B/en active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20040035985A (en) * | 2002-10-14 | 2004-04-30 | (주)에이비아이 | Composition for growth promotion by increasing expression of growth hormone and weight gain in quantity |
| KR101066429B1 (en) * | 2011-01-24 | 2011-09-21 | 장효준 | Method for preparing mycelia and fruiting bodies of Rhizome oleracea with improved antioxidant, immune and anticancer effects |
| CN103725730A (en) * | 2013-12-27 | 2014-04-16 | 内蒙古科技大学 | Refining technology of mycelia polysaccharides of selenium-enriched hericium erinaceus |
Non-Patent Citations (6)
| Title |
|---|
| "不同品种猴头菌子实体粗多糖含量及体外免疫活性比较";李玉等;《食用菌学报》;20140615;第21卷(第2期);54-56 * |
| "猴头菌不同发育阶段产生的多糖结构特征及免疫活性";唐川等;《菌物学报》;20160122;第35卷(第1期);68-76 * |
| "猴头菌多糖提取工艺的研究";秦培鹏;《中国优秀硕士学位论文全文 农业科技辑》;20150515;D048-54 * |
| "猴头菌活性多糖高产菌株的筛选";李巧珍等;《食用菌学报》;20120615;第19卷(第2期);66-68 * |
| "珊瑚状猴头菌营养成分及其多糖对小鼠免疫功能的影响";孟俊龙等;《中国食品学报》;20160229;第16卷(第2期);50-55 * |
| "醇沉条件对猴头菌多糖得率和品质的影响";蒋俊等;《食用菌学报》;20110615;第18卷(第2期);57-61 * |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11932584B2 (en) | 2006-12-15 | 2024-03-19 | Ecovative Design Llc | Method of forming a mycological product |
| US12161069B2 (en) | 2015-04-15 | 2024-12-10 | Ecovative Llc | High density rigid molded body of composite mycological material |
| US12503576B2 (en) | 2017-03-31 | 2025-12-23 | Ecovative Llc | Solution based post-processing methods for mycological biopolymer material and mycological product made thereby |
| US11920126B2 (en) | 2018-03-28 | 2024-03-05 | Ecovative Design Llc | Bio-manufacturing process |
Also Published As
| Publication number | Publication date |
|---|---|
| CN108753625A (en) | 2018-11-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN108753625B (en) | Polysaccharide-producing space Hericium coralloides ST21-3 and application thereof in improving biological immune activity | |
| CN102899276B (en) | Streptococcus thermophilus capable of lowering cholesterol levels and application thereof | |
| CN101215526B (en) | A cell culture method for promoting the synthesis of secondary metabolites of Ganoderma lucidum | |
| CN108676728B (en) | Polysaccharide-producing space coralloid hericium ST21-2 and application thereof in improving biological oxidation resistance | |
| WO2025130836A1 (en) | Xanthomonas campestris strain producing high yield, temperature-resistant and quick-dissolving xanthan gum and use thereof | |
| CN119193415A (en) | Bacillus subtilis strain and its application | |
| CN111607525B (en) | A kind of Ganoderma lucidum mycelium culture medium and culture method thereof | |
| CN113717865B (en) | Ganoderma lucidum mutant strain and application thereof | |
| CN116606848A (en) | High-throughput breeding method of ganoderma lucidum strain | |
| CN120230654B (en) | Saccharomyces cerevisiae for high yield of soluble beta-glucan and application thereof | |
| CN116426431A (en) | Bacillus natto and application thereof | |
| CN100453636C (en) | A kind of Armillaria chrysanthemum and its cultivation method and application | |
| CN117551575A (en) | Fermentation method of bacillus natto and application of bacillus natto | |
| CN110591968A (en) | A kind of method for producing phospholipase D by Streptomyces cinnamonis and method for measuring phospholipase D activity | |
| CN113151071A (en) | Bacillus belgii and application thereof | |
| CN107937318B (en) | Bacillus subtilis MXT-1 for degrading wheat pentosan and application thereof | |
| CN119351236B (en) | Kluyveromyces marxianus with high β-glucan content and use thereof | |
| CN113444646B (en) | Mould for producing beta-caryophyllene and application thereof | |
| CN119410505B (en) | High nucleic acid Kluyveromyces marxianus mutant strain and application thereof | |
| CN111100828B (en) | Leckerella capable of producing high-activity alginate lyase and application thereof | |
| CN118755601B (en) | A strain of Micrococcus sucrose MEP0410 and its application in Dendrobium officinale seedling cultivation | |
| CN109439554B (en) | Space selenium-enriched yeast and selenium-enriched product | |
| CN109161490B (en) | Candida tropicalis HY4-17-21 for producing nucleic acid and preparation method thereof | |
| CN118109304A (en) | A kind of associated fungus of Inonotus obliquus, Acremonium truncatum, fermentation liquid inducer and application thereof | |
| CN111704679B (en) | A kind of Hericium erinaceus fermented mycelium β-glucan and its preparation method and application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |