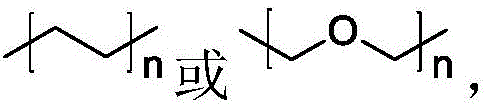CN108690033B - Fluorescent probe containing flavonoid drug active molecules and preparation method and application thereof - Google Patents
Fluorescent probe containing flavonoid drug active molecules and preparation method and application thereof Download PDFInfo
- Publication number
- CN108690033B CN108690033B CN201710217368.7A CN201710217368A CN108690033B CN 108690033 B CN108690033 B CN 108690033B CN 201710217368 A CN201710217368 A CN 201710217368A CN 108690033 B CN108690033 B CN 108690033B
- Authority
- CN
- China
- Prior art keywords
- formula
- compound
- fluorescent probe
- drug active
- preparation
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/12—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains three hetero rings
- C07D491/14—Ortho-condensed systems
- C07D491/147—Ortho-condensed systems the condensed system containing one ring with oxygen as ring hetero atom and two rings with nitrogen as ring hetero atom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N21/00—Investigating or analysing materials by the use of optical means, i.e. using sub-millimetre waves, infrared, visible or ultraviolet light
- G01N21/62—Systems in which the material investigated is excited whereby it emits light or causes a change in wavelength of the incident light
- G01N21/63—Systems in which the material investigated is excited whereby it emits light or causes a change in wavelength of the incident light optically excited
- G01N21/64—Fluorescence; Phosphorescence
- G01N21/6486—Measuring fluorescence of biological material, e.g. DNA, RNA, cells
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Biomedical Technology (AREA)
- Molecular Biology (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Analytical Chemistry (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- General Physics & Mathematics (AREA)
- Immunology (AREA)
- Pathology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
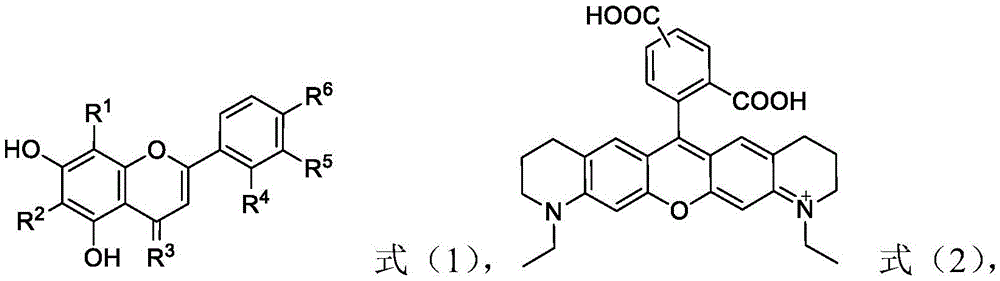
本发明涉及荧光探针领域,公开了一种含黄酮类药物活性分子的荧光探针及其制备方法和应用,本发明的荧光探针由通式X‑L‑Y表示,其中,X是由下述式(1)提供的黄酮类药物活性结构;Y是由下述式(2)提供的荧光基团;偶联结构L为
或,n为1‑6的整数;式(1)中,R1、R4、R5和R6各自独立地选自氢、羟基或碳原子数为1‑6的烷氧基;R2选自氢或羟基;R3为氧。本发明的含黄酮类药物活性分子的荧光探针具有特异靶向性,还具有较高的肿瘤或癌症细胞的增殖抑制活性。The invention relates to the field of fluorescent probes, and discloses a fluorescent probe containing flavonoid drug active molecules and a preparation method and application thereof. The fluorescent probe of the present invention is represented by the general formula X-L-Y, wherein X is represented by The flavonoid drug active structure provided by the following formula (1); Y is a fluorescent group provided by the following formula (2); the coupling structure L is
or , n is an integer of 1-6; in formula (1), R 1 , R 4 , R 5 and R 6 are each independently selected from hydrogen, hydroxyl or an alkoxy group with 1-6 carbon atoms; R 2 is selected from From hydrogen or hydroxyl; R 3 is oxygen. The fluorescent probe containing the flavonoid drug active molecule of the present invention has specific targeting properties, and also has high tumor or cancer cell proliferation inhibitory activity.Description
Technical Field
The invention relates to a fluorescent probe containing flavonoid drug active molecules and a preparation method and application thereof.
Background
Scutellaria baicalensis is a traditional flavonoid traditional Chinese medicine for inflammation and detumescence, and has a plurality of pharmacological actions such as anti-inflammation, detumescence, antivirus and anticancer. The scutellaria baicalensis as a natural product has complex components, so at present, scientists cannot determine a molecular action mechanism and a biological function way for generating the drug effect, and the generated problem is always a bottleneck for restricting the clinical application of natural product medicines.
Fluorescence microscopy is an emerging analytical technique in recent years and is widely used in the fields of environmental science, medicine, pharmacy, biology and the like, particularly in the fields of medicine and biology. The fluorescent probe converts chemical information such as intermolecular interaction and the like into a fluorescent signal and feeds the fluorescent signal back to the outside to enable the whole process to be visualized, and the distribution and action mode of certain substances such as drugs and the like in cells are usually monitored in real time by using the fluorescent probe. Therefore, the design of specifically targeted fluorescent probes is of great importance.
Disclosure of Invention
The invention aims to provide a fluorescent probe containing flavonoid drug active molecules with specific targeting and a preparation method and application thereof.
The inventor of the invention finds that the fluorescent probe containing flavonoid drug active molecules represented by the general formula X-L-Y has specific targeting properties of mitochondria and lysosomes, can act on the mitochondria and the lysosomes in tumor or cancer cells in a targeting manner, also has high proliferation inhibition activity of the tumor or cancer cells, shows that the proliferation inhibition activity of the tumor or cancer cells is basically unchanged, and has good medicinal value and market prospect when being used as an excellent fluorescent probe.
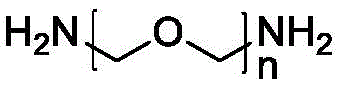
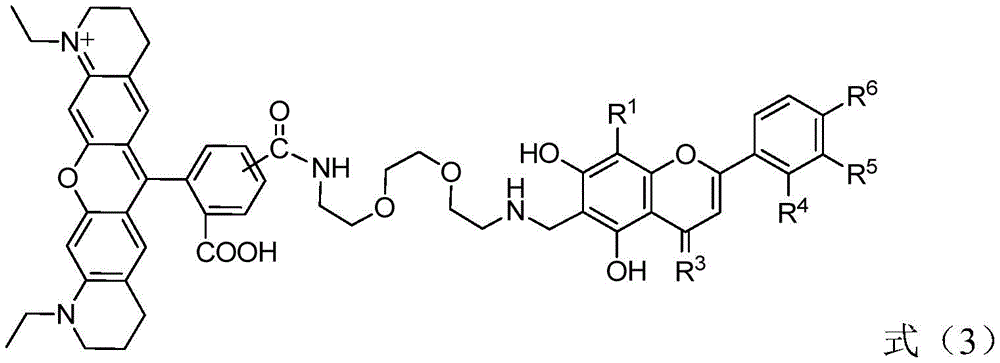
Therefore, the invention provides a fluorescent probe containing flavonoid drug active molecules, wherein the fluorescent probe is represented by a general formula X-L-Y, wherein X is a flavonoid drug active structure provided by the following formula (1); y is a fluorescent group provided by the following formula (2); the coupling structure L is n is an integer of 1 to 6;
in the formula (1), R1、R4、R5And R6Each independently selected from hydrogen, hydroxyl or alkoxy with 1-6 carbon atoms; r2Selected from hydrogen or hydroxy; r3Is oxygen.
The second aspect of the present invention provides a method for preparing the above fluorescent probe containing flavonoid-containing pharmaceutically active molecules, wherein the method comprises the following steps,
1) reacting a compound having a structure represented by the formula (2) withCarrying out amide condensation reaction to obtain an intermediate compound represented by a general formula L-Y, wherein n is an integer of 1-6;
2) and (3) carrying out Mannich reaction on the intermediate compound represented by the general formula L-Y and the compound having the structure represented by the formula (1) to obtain the fluorescent probe containing flavonoid drug active molecules represented by the general formula X-L-Y.
The third aspect of the invention provides the application of the fluorescent probe containing flavonoid drug active molecules or the fluorescent probe containing flavonoid drug active molecules prepared by the preparation method in preparing drugs for treating tumors or cancers.
Through the technical scheme, the fluorescent probe containing flavonoid drug active molecules provided by the invention has good targeting property for mitochondria and lysosomes, and can act on the mitochondria and the lysosomes in tumor or cancer cells in a targeted manner; in addition, the fluorescent probe containing flavonoid drug active molecules represented by the general formula X-L-Y also has good proliferation inhibition activity on tumor or cancer cells.
Drawings
The accompanying drawings, which are included to provide a further understanding of the invention and are incorporated in and constitute a part of this specification, illustrate embodiments of the invention and together with the description serve to explain the principles of the invention and not to limit the invention. In the drawings:
FIG. 1 is a graph showing the confocal fluorescence images of the fluorescent probe of the formula (3-1-2) in A549 (left) and HeLa (right) cells in test example 2.
FIG. 2 shows the fluorescent probe represented by the formula (3-1-2) in test example 2 together with cell membrane dye DIO and nuclear dyeLive 647RedConfocal laser fluorescence imaging of Reagent in a549 (left) and HeLa (right) cells.
FIG. 3 is a graph showing the laser confocal fluorescence images of the fluorescent probe represented by the formula (3-1-2) in test example 2 and the mitochondrial dye Rhodamine123 in A549 (left) and HeLa (right) cells.
FIG. 4 is a graph showing laser confocal fluorescence images of the fluorescent probe shown in the formula (3-1-2) structure in test example 2 and the lysosomal dye DND-26 in A549 (left) and HeLa (right) cells.
FIG. 5 is a graph showing the results of the degree of overlap between the fluorescent probe represented by the formula (3-1-2) in the test example 2 and the mitochondrial dye Rhodamine123 in the same two-dimensional dimension in A549 (left) and HeLa (right) cells.
FIG. 6 is a graph showing the results of the degree of overlap of the fluorescent probe represented by the formula (3-1-2) structure in test example 2 with the lysosomal dye DND-26 in the same two-dimensional dimension in A549 (left) and HeLa (right) cells.
FIG. 7 is a graph showing the results of the test example 2 on the proliferation inhibitory activity of the fluorescent probe represented by the formula (3-1-2) and the flavonoid drug active molecule on HeLa cells.
Detailed Description
The endpoints of the ranges and any values disclosed herein are not limited to the precise range or value, and such ranges or values should be understood to encompass values close to those ranges or values. For ranges of values, between the endpoints of each of the ranges and the individual points, and between the individual points may be combined with each other to give one or more new ranges of values, and these ranges of values should be considered as specifically disclosed herein.
The term "C" as used in the present specification1-6The alkoxy group "may be linear or branched. Examples thereof include methoxy, ethoxy, propoxy, isopropoxy, butoxy, isobutoxy, sec-butoxy, tert-butoxy, pentyloxy, hexyloxy and the like.
In the present invention, e.g.The substituent R on the equivalent structure may be substituted at an arbitrary position on the ring, or may be substituted at a plurality of positions on the ring, and when the substitution is performed at a plurality of positions, the substituent R at each position may be the same or different.
In the present invention, the phrase "the fluorescent probe containing a flavonoid-containing pharmaceutically active molecule is represented by the following general formula X-L-Y" means that the fluorescent probe containing a flavonoid-containing pharmaceutically active molecule may be one of the compounds represented by the following general formula X-L-Y, or two or more of the compounds represented by the following general formula X-L-Y.
The fluorescent probe containing flavonoid drug active molecules is represented by a general formula X-L-Y, wherein X is a flavonoid drug active structure provided by the following formula (1); y is a fluorescent group provided by the following formula (2); the coupling structure L isn is an integer of 1 to 6;
in the formula (1), R1、R4、R5And R6Each independently selected from hydrogen, hydroxyl or alkoxy with 1-6 carbon atoms; r2Selected from hydrogen or hydroxy, R3Is oxygen.
Preferably, R1、R4、R5And R6Each independently selected from hydrogen, hydroxyl or alkoxy with 1-3 carbon atoms; r2Selected from hydrogen or hydroxy; r3Is oxygen; n is 1,2,3 or 4.
More preferably, R1、R4、R5And R6Each independently selected from hydrogen, hydroxyl or alkoxy with 1-3 carbon atoms; r2Selected from hydrogen or hydroxy; r3Is oxygen; n is 2,3 or 4.
Further preferably, R1、R4、R5And R6Each independently selected from hydrogen, hydroxy, methoxy or ethoxy; r2Selected from hydrogen or hydroxy; r3Is oxygen; n is 2,3 or 4.
In a preferred embodiment of the present invention, the general formula X-L-Y is a compound having a structure represented by the following formula (3),
in a preferred embodiment of the present invention, the general formula X-L-Y is a compound having a structure represented by the following formula (3-1),
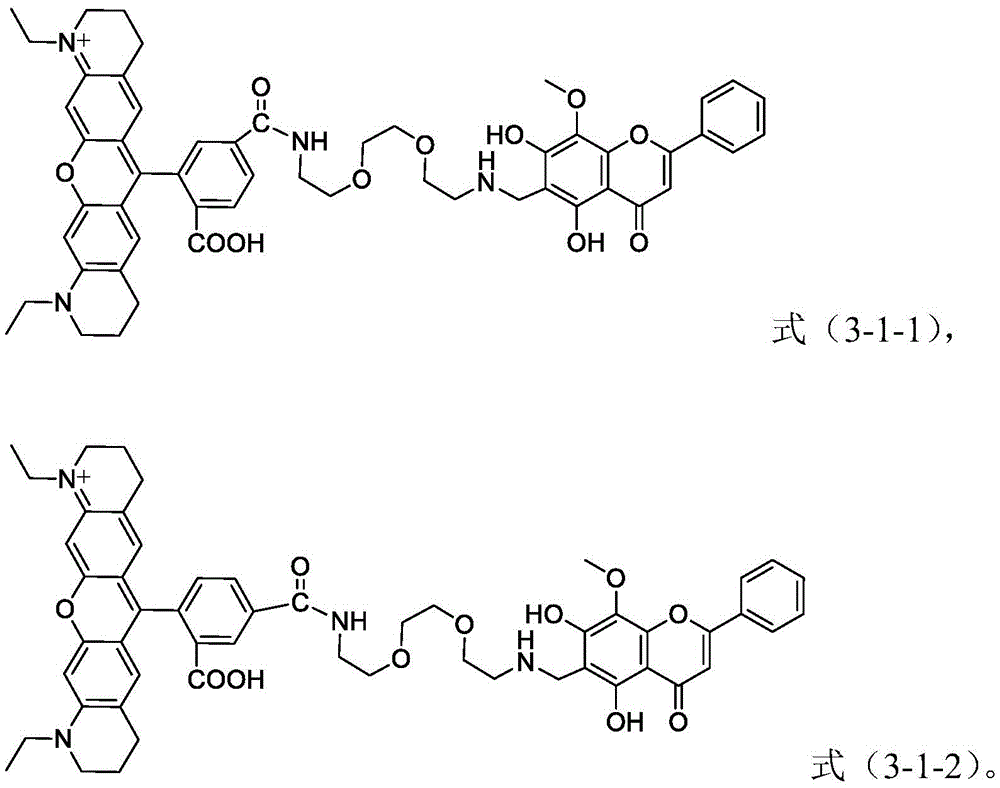
in a preferred embodiment of the present invention, the general formula X-L-Y is a compound having a structure represented by the following formula (3-1-1) or a compound having a structure represented by the following formula (3-1-2),
in a particularly preferred embodiment of the present invention, the fluorescent probe containing a flavonoid-containing pharmaceutically active molecule is a compound having a structure represented by the above formula (3-1-1) and/or a compound having a structure represented by the above formula (3-1-2).
When the fluorescent probe containing flavonoid-based pharmaceutically active molecules is a compound having a structure represented by the formula (3-1-1) or a compound having a structure represented by the formula (3-1-2) (i.e., a mixture of the two), there is no particular requirement on the mixing ratio of the compound having a structure represented by the formula (3-1-1) and the compound having a structure represented by the formula (3-1-2). Preferably, the molar ratio of the compound of the structure represented by the formula (3-1-1) to the compound of the structure represented by the formula (3-1-2) is 1: 0.2 to 5, more preferably 1: 0.3 to 4; more preferably 1: 0.4 to 3; more preferably 1: 0.5 to 2; more preferably 1: 0.8-1.5; more preferably 1: 0.9-1.2; particularly preferably 1: 1.
the compound provided by the invention, especially the compound with the structure shown in the formula (3-1), has good water solubility and stability, and good proliferation inhibition activity of tumor or cancer cells.
The invention also provides a preparation method of the fluorescent probe containing flavonoid drug active molecules, wherein the method comprises the following steps,
1) reacting a compound having a structure represented by the formula (2) withCarrying out amide condensation reaction to obtain an intermediate compound represented by a general formula L-Y, wherein n is an integer of 1-6;
2) and (3) carrying out Mannich reaction on the intermediate compound represented by the general formula L-Y and the compound having the structure represented by the formula (1) to obtain the fluorescent probe containing flavonoid drug active molecules represented by the general formula X-L-Y.
According to the invention, the compound with the structure shown in the formula (2) can be a commercially available product, or can be synthesized after being optimized by a conventional method in the field, and when the compound is obtained by synthesis, for example, the compound can be obtained by using the compound shown in the reference (1) azalone, a.v.; wang, t.y.; chen, z.; cornish, v.w.angelw.chem.int.ed.engl.2013, 52,650; (2) uddin, m.j.; cress, b.c.; ghebeseleshie, k.; marnett, l.j.bioconjugate.chem.2013, 24,712; (3) uddin, m.j.; a method as described in Marnett, l.j.org.lett.2008,10,4799.
The process for producing a compound having a structure represented by formula (2) is described by way of example of production of a compound having a structure represented by formula (2), and the process for producing a compound having a structure represented by formula (2) is preferably carried out by the following reaction scheme:
wherein, LiAlH4Represents lithium aluminum hydride, NaBH4 represents sodium borohydride, 1M represents 1mol/L, and reflux represents reflux.
According to the present invention, the compound of the structure represented by the formula (2) is preferably a compound of the structure represented by the formula (2-1) or a compound of the structure represented by the formula (2-2),
according to the invention, in step 1) a compound of the formula (2) is reacted with Carrying out amide condensation reaction to obtain an intermediate compound represented by a general formula L-Y, wherein n is an integer of 1-6; preferably, n is 1,2,3 or 4; more preferably, n is 2,3 or 4.
In a preferred embodiment of the present invention, in step 1), a compound having a structure represented by formula (2) is reacted withCarrying out amide condensation reaction to obtain an intermediate compound represented by a general formula L-Y, wherein n is 2; the specific preparation method is preferably carried out by adopting the following reaction route:
wherein EDCI represents 1-ethyl- (3-dimethylaminopropyl) carbodiimide hydrochloride, HOBT represents 1-hydroxybenzotriazole, and TEA represents triethylamine.
According to the invention, in step 1), the compound of formula (2) is reacted with The amount of (A) can vary within wide limits. Preferably, compounds of the structure of formula (2) andthe molar ratio of (1): 2-2.5.
The amide condensation reaction may employ a solvent conventionally used in amide reactions, and may be, for example, one or more of dichloromethane, tetrahydrofuran, and Dimethylformamide (DMF); dimethylformamide is preferred. These solvents are preferably anhydrous solvents. In the present invention, the concentration of the compound having the structure represented by the formula (2) in the solvent is not particularly limited as long as the L-Y intermediate compound can be obtained, and may be, for example, 0.01 to 0.2mmol/mL, preferably 0.01 to 0.02 mmol/mL.
In the present invention, preferably, the amide condensation reaction conditions include: the reaction temperature is 15-80 ℃, and the reaction time is 10-72 h; more preferably, the conditions of the amide condensation reaction include: the reaction temperature is 40-50 ℃, and the reaction time is 24-48 h.
According to the invention, in the step 2), the intermediate compound represented by the general formula L-Y and the compound having the structure represented by the formula (1) are subjected to Mannich reaction to obtain the fluorescent probe containing flavonoid drug active molecules represented by the general formula X-L-Y.
Preferably, in the formula (1), R1、R4、R5And R6Each independently selected from hydrogen, hydroxyl or alkoxy with 1-3 carbon atoms; r2Selected from hydrogen or hydroxy; r3Is oxygen; more preferably, R1、R4、R5And R6Each independently selected from hydrogen, hydroxyl or alkoxy with 1-3 carbon atoms; r2Selected from hydrogen or hydroxy; r3Is oxygen; further preferably, R1、R4、R5And R6Each independently selected from hydrogen, hydroxy, methoxy or ethoxy; r2Selected from hydrogen or hydroxy; r3Is oxygen.
In a preferred embodiment of the present invention, the compound of the structure represented by the formula (1) is a compound of the structure represented by the following formula (1-1),
in a particularly preferred embodiment of the present invention, the fluorescent probe having a flavonoid-containing pharmaceutically active structure represented by the above formula X-L-Y is one or more compounds having a structure represented by the following formula (3).
Preferably, the compound of the structure represented by the formula (3) is a compound of the structure represented by the following formula (3-1),
more preferably, the general formula X-L-Y is a compound having a structure represented by the following formula (3-1-1) and a compound having a structure represented by the following formula (3-1-2),
the compound represented by the above formula (3-1) can be synthesized according to the following scheme.
According to the present invention, in step 2), the molar ratio of the intermediate compound to the compound having the structure represented by formula (1) is not particularly limited, as long as the fluorescent probe containing a flavonoid-containing pharmaceutically active molecule represented by the above general formula X-L-Y can be obtained, and for example, the molar ratio of the intermediate compound to the compound having the structure represented by formula (1) is 1: 0.8-1.3.
In addition, the Mannich reaction may employ a solvent such as methanol, ethanol, tetrahydrofuran, and the like. The concentration of the intermediate compound in the solvent is not particularly limited as long as a fluorescent probe having a flavonoid-containing pharmaceutically active structure represented by the general formula X-L-Y can be obtained, and for example, the concentration of the compound having a structure represented by the formula (1) may be 0.001 to 0.002mmol/mL, more preferably 0.0015 to 0.002 mmol/mL.
Preferably, the conditions of the Mannich reaction include: the reaction temperature is 20-110 ℃, and the reaction time is 1-20 h; more preferably, the conditions of the Mannich reaction include: the reaction temperature is 65-80 ℃, and the reaction time is 4-10 h.
In order to obtain a more pure product, the preparation method of the fluorescent probe containing flavonoid drug active molecules can further comprise the step of carrying out recrystallization or chromatographic separation and purification on the obtained fluorescent probe by using ethanol.
The invention also provides the application of the fluorescent probe containing flavonoid drug active molecules or the fluorescent probe containing flavonoid drug active molecules prepared by the preparation method in preparing drugs for treating tumors or cancers.
The present invention is not particularly limited with respect to the type of the tumor or cancer. Preferably, the tumor or cancer is lung cancer, cervical cancer, or breast cancer.
The fluorescent probe containing flavonoid drug active molecules represented by the general formula X-L-Y provided by the invention has good mitochondrial targeting and lysosome targeting. Besides, the fluorescent probe containing flavonoid drug active molecules provided by the invention has good targeting property to mitochondria and lysosomes, and also has high proliferation inhibition activity to tumor or cancer cells, so that the fluorescent probe containing flavonoid drug active molecules can act on various tumor or cancer cells in a targeting way, and further research the action mechanism and the way of related molecules in organisms.
In addition, the fluorescent probe containing flavonoid drug active molecules shows basically unchanged proliferation inhibition activity of tumor or cancer cells, is used as a specific targeting fluorescent probe, and has better drug effect.
The present invention will be described in detail below by way of examples.
Preparation example 1
This preparation example is used to illustrate the preparation of a compound having a structure represented by formula (2).
(1) 3.23g (20mmol) of 7-hydroxy-2-quinolone (Compound 1) is added to 90mL of tetrahydrofuran, and 1.22g (32mmol) of lithium aluminum hydride is slowly added thereto with stirring in an ice bath, and the mixture is refluxed and reacted overnight. After the reaction was stopped, the reaction was cooled, 35.0ml of a saturated ammonium chloride solution was immediately added to quench the reaction, extraction was performed with ethyl acetate, the organic phase was washed with a supersaturated sodium chloride solution, and concentration was performed to obtain a yellow solid 2.83g, with a yield of 95%. ESI-MS (M/z) < M + H]+calcd.for C9H11NO,150.09;found,150.25。
(2) 2.50g (16.8mmol) of 7-hydroxy-1, 2,3, 4-tetrahydroquinoline (Compound 2) was added to 65mL of acetic acid, and 2.53g (67.2mmol) of sodium borohydride was slowly added thereto with stirring at room temperature. When about half of the reaction starting material was consumed at room temperature, acetaldehyde was added in portions until the starting material was completely consumed. Removing most of acetic acid with NaHCO3The pH was adjusted to neutral, filtered and recrystallized to give 2.68g of a pale yellow solid with a yield of 90%. ESI-MS (M/z) < M + H]+calcd.for C11H16NO,178.12;found,178.10.
1.50g (8.47mmol) of 1-ethyl-7-hydroxy-1, 2,3, 4-tetrahydroquinoline (compound 3) obtained in step (2) was added to 60mL of n-butyric acid, 3 drops of concentrated sulfuric acid were added as a catalyst, 0.82g (4.24mmol) of trimellitic anhydride (compound 4) was added, and the mixture was refluxed for 12 hours. Cooled to room temperature, ether was added to precipitate a dark red oil which was collected and separated by silica gel chromatography (CHCl)3:CH3OH ═ 5:1-1:1), yielding a dark red solid (yield: 28%) to give respectively the compound of formula (2)The compounds of the structures shown are 5-isomer of formula (2-1) and 6-isomer of formula (2-2).
The structural formulae of formula (2-1) and formula (2-2) are as follows:
5-isomer:1HNMR(400MHz,DMSO-d6)(ppm):8.16(d,J=7.6Hz,1H),8.01(d,J=8Hz,1H),7.59(s,1H),6.44(s,2H),6.22(s,2H),3.42-3.38(m,4H),3.28-3.25(m,4H),2.47-2.42(m,4H),1.76(m,J=6.4Hz,4H),1.12(t,J=8Hz,6H).MALDI-TOF-MS(m/z):[M]+calcd.for C31H31N2O5 +,511.22;found,511.28.
6-isomer:1HNMR(400MHz,DMSO-d6)(ppm):8.41(s,1H),8.24(dd,J1=8Hz,J2=1.2Hz,1H),7.32(d,J=7.6Hz,1H),6.47(s,2H),6.26(s,2H),3.43-3.38(m,4H),3.30-3.27(m,4H),2.47-2.43(m,4H),1.77(m,J=6.4Hz,4H),1.12(t,J=8Hz,6H).13CNMR(400MHz,DMSO-d6)(ppm):MALDI-TOF-MS(m/z):[M]+calcd.for C31H31N2O5 +,511.22;found,511.29.
preparation example 2
This preparation example is used to illustrate the preparation of a compound having a structure represented by formula (6).
(1) 400mg (0.78mmol) of a mixture of the compound with the structure shown in the formula (2-1) and the compound with the structure shown in the formula (2-2) (the mixing molar ratio is 1:1), 179.5mg (0.94mmol) of 1-ethyl- (3-dimethylaminopropyl) carbodiimide hydrochloride (EDCI), 126.5mg (0.94mmol) of 1-Hydroxybenzotriazole (HOBT) and 325.5 muL of triethylamine are added into 100mL of dry dichloromethane, and the mixture is stirred and reacted for 1-2 h at 50 ℃. Then 285. mu.L of 1, 8-diamino-3, 6-dioxaoctane was added thereto, and the reaction was continued for 24 hours. Water was added to terminate the reaction, and the mixture was extracted and concentrated to give a mixture of the compound represented by the formula (6-1) and the compound represented by the formula (6-2) as a dark red solid (0.20g, yield 40.3%). A part of the mixture was taken out and separated to obtain a compound represented by the formula (6-1) and a compound represented by the formula (6-2), and the data of the formula (6-2) are shown below.
Formula (6-2):1HNMR(400MHz,DMSO-d6)(ppm):8.68(s,1H),8.27(d,J=8Hz,1H),7.52(d,J=8Hz,1H),6.93(d,J=8Hz,2H),6.68(d,J=8Hz,2H),3.61-3.51(m,12H),3.00(t,J1=J2=8Hz,2H),2.89(s,1H),2.74-2.72(m,4H),2.63-2.58(m,4H),2.28-2.26(m,J=4Hz,2H),1.91-1.78(m,6H),1.22(t,J1=J2=8Hz,6H).13CNMR(400MHz,DMSO-d6)(ppm):169.8,158.5,151.9,151.5,147.5,146.1,136.4,130.1,128.0,127.5,127.0,119.8,119.7,119.6,118.9,118.3,104.3,104.1,100.0,96.6,96.2,72.6,70.1,67.6,57.2,48.0,45.6,38.1,37.6,35.6,34.5,28.4,27.3,25.6,23.0,22.0,16.2,10.9.MALDI-TOF-MS(m/z):[M]+calcd.for C37H45N4O6 +,641.33;found,641.30.
formula (6-2): MALDI-TOF-MS (M/z) [ M ]]+calcd.for C37H45N4O6 +,641.33;found,641.30.
Example 1
This example illustrates the fluorescent probe containing flavonoid-containing pharmaceutically active molecules and the preparation method thereof according to the present invention.
(1) 100mg (0.16mmol) of the mixture of the compound of the structure represented by the formula (6-1) and the compound of the structure represented by the formula (6-2) obtained in preparation example 2, 9.60mg (0.32mmol) of paraformaldehyde and 2.18mg (0.016mmol) of ZnCl2After adding 100mL of ethanol and reacting for 30min, 45.3mg (0.16mmol) of the compound having the structure represented by the formula (1-1) was added and reacted at 65 ℃ for 10 hours. Cooled to room temperature, the ethanol is removed and the residue is chromatographed on silica gel (CHCl)3: CH3OH ═ 10:1), the obtained solid was used with high efficiencyLiquid Chromatography (HPLC) separation and purification gave the compound of formula (3-1-1) and the compound of formula (3-1-2), respectively (19.8 mg in total, 13.2% yield).
Formula (3-1-2):1HNMR(400MHz,CD3OD)(ppm):8.26(d,J=8Hz,1H),8.07(d,J=8Hz,1H),7.93(d,J=8Hz,2H),7.87(d,J=8Hz,1H),7.65(s,1H),7.50-7.48(m,5H),6.72(d,J=12Hz,1H),6.60(s,1H),5.24(t,J=4Hz,2H),3.89(m,4H),3.57-3.48(m,13H),3.39(t,J=6Hz,1H),2.91-2.87(m,1H),2.78-2.76(m,1H),2.60(s,1H),2.56(s,1H),2.09(t,J=8Hz,2H),1.93(q,J1=8Hz,J2=4Hz,4H),1.84-1.80(m,4H),1.50(t,J=8Hz,2H),0.80(t,J=8Hz,6H).13CNMR(600MHz,CD3OD)(ppm):182.8,178.0,166.9,164.1,162.4,161.8,161.6,156.9,156.5,155.7,154.4,153.6,153.2,148.4,137.7,134.6,132.1,131.9,131.2,131.1,129.4,129.0,128.3,127.9,127.1,126.7,126.0,125.3,123.7,113.5,112.8,109.0,107.1,104.7,103.7,94.3,69.9,69.0,61.0,41.8,39.7,39.0,37.4,35.1,31.7,27.1,26.7,25.5,22.3,20.7,19.9,16.2,13.1,10.1.MALDI-TOF-MS(m/z):[M]+calcd.for C54H57N4O11 +,937.4;found,937.4.
formula (3-1-1): MALDI-TOF-MS (M/z) [ M ]]+calcd.for C54H57N4O11 +,937.4;found,937.4.
Test example 1: experiment for inhibiting proliferation of various tumor cells
MTT method for detecting probe cell proliferation inhibition activity experiment, cell strains A549 (human lung adenocarcinoma cell strain), HeLa (human cervical cancer cell strain) and MCF-7 (human breast cancer cell strain) are respectively cultured in DMEM medium (Gibco, USA) containing 10 vol% of total bone serum (FBS, Hyclone, USA). The collection was in log phase after 2-3 daysLong-term cells were seeded in 96-well plates (A549: 2000 cells/well/100. mu.L; HeLa: 4000 cells/well/100. mu.L; MCF-7: 4000 cells/well/100. mu.L) and cultured for 24h, and then compounds (200, 100, 50, 25, 10, 5, 2.5,. mu.M) were added at gradient concentrations, respectively. The same volume of culture medium containing 1% DMSO without probe and flavonoid drug active molecule was set as experimental control, culture medium with 1% DMSO only and no cells was set as blank control, and 5 replicates were run at each concentration. After an additional 48h incubation, the probe-containing medium was aspirated, washed once with 200. mu.L/well PBS, and 100. mu.L of medium containing 0.5mg/mL MTT was added to each well for an additional 4h incubation. The MTT containing medium was discarded without blotting adherent crystals, 100. mu.L of DMSO was added to each well and placed on a shaker for 10-15min to dissolve the crystals. Then the cell culture plate is moved to an enzyme labeling instrument, the absorbance value is measured at 492nm, the survival rate of the cells is detected by an MTT method, and the biological activities of the probe and the flavonoid drug active structure are compared. The cell viability calculation formula is as follows: survival (%) (experimental OD-blank OD)/(control OD-blank OD)]X 100%. And the IC of the compound was calculated using Origin 8.0 software using the inhibition and concentration for curve fitting50The value is obtained.
TABLE 1
In table 1, the inhibitory activity of the flavonoid drug active structure with the structure shown in formula (1-1) on the proliferation of three cells, namely a549 cell, HeLa cell and MCF-7 cell, is slightly higher than that of the compound with the structure shown in formula (3-1-2) on the proliferation of three cells, namely a549 cell, HeLa cell and MCF-7 cell, but as shown in fig. 7, the inhibitory activity trend of the compound with the structure shown in formula (3-1-2) obtained after modification and that of the flavonoid drug active structure with the structure shown in formula (1-1) on the proliferation of three cells is consistent, which indicates that the biological activity of the flavonoid drug active molecule with the structure shown in formula (1-1) is retained after the flavonoid drug active molecule is modified and obtained from the compound with the structure shown in formula (3-1-2).
Test example 2: laser confocal fluorescence imaging test
(1) Cell imaging analysis
The confocal dish was seeded with 1mL of A-549 and HeLa cell suspensions at a cell density of about 1X 104cells/mL were incubated at 37 ℃ for 12h to adhere. mu.M (0.2%) of a compound having the structure represented by formula (3-1-2) was incubated with the cells at 37 ℃ for 2h, 6h and 12h, respectively, and no compound having the structure represented by formula (3-1-2) and only cells were used as a control group. The drug-containing medium was removed, washed three times with PBS solution, and colorless basal medium DMEM was added. And moving the confocal dish to a sample stage of a laser scanning confocal microscope for imaging. The excitation wavelength of the laser scanning confocal microscopy imaging is selected to be 559nm, and the emission wavelength is 570-670 nm. The resulting image was opened and processed with FV10-ASW 3.1Viewer software as shown in FIG. 1.
(2) Cell co-localization imaging analysis
The confocal dish was seeded with 1mL of A-549 and HeLa cell suspensions at a cell density of about 1X 104cells/mL were incubated at 37 ℃ for 12h to adhere. mu.M (0.2%) of a compound having the structure represented by formula (3-1-2) was incubated with the cells at 37 ℃ for 2h, 6h and 12h, respectively, and no compound having the structure represented by formula (3-1-2) and only cells were used as a control group. The drug-containing medium was removed and washed three times with PBS solution. Then the selected cell membrane dye DIO (5. mu.L/mL) and nuclear dye were addedLive 647RedReagent (2 drops), incubated at 37 ℃ for 15-20min, washed again three times with PBS solution, and colorless basal medium DMEM was added. And moving the confocal dish to a sample stage of a laser scanning confocal microscope for imaging. The excitation wavelengths of the laser scanning confocal microscopy images are respectively selected to be 559nm, 488nm and 635nm, and the emission wavelengths are respectively 570-615nm, 500-545nm and 650-750 nm. The resulting image was opened and processed with FV10-ASW 3.1Viewer software as shown in FIG. 2. Then, based on the nuclear and membrane localization results, 1mL of A-549 and HeLa cell suspensions were inoculated into the confocal dishCell density of about 1X 104cells/mL were incubated at 37 ℃ for 12h to adhere. mu.M (0.2%) of a compound having the structure represented by formula (3-1-2) was incubated with the cells at 37 ℃ for 2h, 6h and 12h, respectively, and no compound having the structure represented by formula (3-1-2) and only cells were used as a control group. The drug-containing medium was removed and washed three times with PBS solution. Then the chosen mitochondrial dye Rhodamine123(500nM) and the lysosomal dye were addedGreen DND-26(500nM), incubated 15-20min at 37 deg.C, washed three more times with PBS solution, and colorless basal medium DMEM was added. And moving the confocal dish to a sample stage of a laser scanning confocal microscope for imaging. The excitation wavelength of the laser scanning confocal microscopy is respectively selected to be 559nm and 488nm, and the emission wavelength is respectively 570-615nm and 500-545 nm. The resulting image was opened and processed with FV10-ASW 3.1Viewer software, as shown in fig. 3-4; and the degree of overlap of the fluorescent probe with the mitochondrial and lysosomal dyes in the same two-dimensional dimension, as shown in fig. 5-6.
The results of laser confocal fluorescence imaging are shown in fig. 1-4, in which fig. 1 is the laser confocal fluorescence imaging diagrams of the fluorescent probe shown in the structure of formula (3-1-2) in A549 cells (left) and HeLa cells (right), and fig. 2 is the fluorescent probe shown in the structure of formula (3-1-2), DIO and DIOLive 647RedThe confocal fluorescence images of the fluorescence probe co-localized in the A549 cell (left) and the HeLa cell (right) of the Reagent, FIG. 3 is the confocal fluorescence image of the fluorescence probe shown in the formula (3-1-2) and the mitochondrial dye Rhodamine123 co-localized in the A549 cell (left) and the HeLa cell (right), respectively, and FIG. 4 is the confocal fluorescence image of the fluorescence probe shown in the formula (3-1-2) and the lysosomal dye DND-26 co-localized in the A549 cell (left) and the HeLa cell (right), respectively. FIGS. 5-6 show fluorescent probe and mitochondrial staining, respectivelyThe degree of overlap of the lysosome dye in the same two-dimensional dimension of co-localization in A549 cells (left) and HeLa cells (right) is clearly shown by comparing FIGS. 1-4, and the distribution of the fluorescent probe represented by the structure of formula (3-1-2) in the cells is regional, and is almost not overlapped with the cell membrane and nuclear dye, indicating that the fluorescent probe is distributed in cytoplasm; and the result of co-localization of the mitochondrial dye and the lysosomal dye shows that the fluorescent probe is completely overlapped with the mitochondrial dye signal and partially overlapped with the lysosomal dye signal; it is also evident from FIGS. 5-6 that the fluorescent probe completely overlaps with the mitochondrial dye signal and partially overlaps with the lysosomal dye signal, both of which indicate that the flavonoid-containing pharmaceutically active molecule has a target or one of its targets of action as mitochondria and lysosomes.
The preferred embodiments of the present invention have been described above in detail, but the present invention is not limited thereto. Within the scope of the technical idea of the invention, many simple modifications can be made to the technical solution of the invention, including combinations of various technical features in any other suitable way, and these simple modifications and combinations should also be regarded as the disclosure of the invention, and all fall within the scope of the invention.
Claims (9)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201710217368.7A CN108690033B (en) | 2017-04-05 | 2017-04-05 | Fluorescent probe containing flavonoid drug active molecules and preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201710217368.7A CN108690033B (en) | 2017-04-05 | 2017-04-05 | Fluorescent probe containing flavonoid drug active molecules and preparation method and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN108690033A CN108690033A (en) | 2018-10-23 |
| CN108690033B true CN108690033B (en) | 2021-01-01 |
Family
ID=63841928
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201710217368.7A Active CN108690033B (en) | 2017-04-05 | 2017-04-05 | Fluorescent probe containing flavonoid drug active molecules and preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN108690033B (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110256467A (en) * | 2019-07-17 | 2019-09-20 | 大连理工大学 | Fluorescent probe for tracing tedizolid antibiotics and application |
| CN111620918A (en) * | 2020-06-19 | 2020-09-04 | 辽宁中医药大学 | 8-beta-D-glucopyranose-4', 7-dihydroxyisoflavone FAM derivative and synthetic method thereof |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2014354643B2 (en) * | 2013-11-27 | 2020-03-05 | Redwood Bioscience, Inc. | Hydrazinyl-pyrrolo compounds and methods for producing a conjugate |
| CN104212440B (en) * | 2014-09-03 | 2016-03-30 | 无锡艾德美特生物科技有限公司 | One class quinazoline ditosylate salt fluorescent probe and Synthesis and applications thereof |
-
2017
- 2017-04-05 CN CN201710217368.7A patent/CN108690033B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN108690033A (en) | 2018-10-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Lagisetty et al. | CLEFMA—An anti-proliferative curcuminoid from structure–activity relationship studies on 3, 5-bis (benzylidene)-4-piperidones | |
| CN101948488B (en) | Ruthenium-selenium coordination compound and application thereof in preparing fluorescent probe and antineoplastic medicine | |
| Karami et al. | Novel fluorescence palladium-alkoxime complexes: Synthesis, characterization, DNA/BSA spectroscopic and docking studies, evaluation of cytotoxicity and DNA cleavage mechanism | |
| Bian et al. | Rh (III)‐Catalyzed Redox‐Neutral [4+ 2] Annulation for Direct Assembly of 3‐Acyl Isoquinolin‐1 (2H)‐ones as Potent Antitumor Agents | |
| Chen et al. | Design, synthesis, anticancer activity and cytotoxicity of novel 4-piperidone/cyclohexanone derivatives | |
| Ipek et al. | Synthesis of novel hybrid lonidamine-coumarin derivatives and their anticancer activities | |
| CN108690033B (en) | Fluorescent probe containing flavonoid drug active molecules and preparation method and application thereof | |
| CN113683557A (en) | A kind of application of cyclopentyl iridium/rhodium dimer | |
| CN114133390A (en) | A kind of dehydrocamelin derivative and its preparation method and application | |
| Zhai et al. | Discovery and optimization of novel 5-indolyl-7-arylimidazo [1, 2-a] pyridine-8-carbonitrile derivatives as potent antitubulin agents targeting colchicine-binding site | |
| Jin et al. | Synthesis of a novel fluorescent berberine derivative convenient for its subcellular localization study | |
| Bokhtia et al. | Ursolic Acid Conjugates: A New Frontier in Anticancer Drug Development | |
| CN110357899A (en) | A kind of traceable antitumor podophyllotoxin derivative and its preparation and application | |
| Feng et al. | Several Golgi targeting fluorescence markers based on 1, 8-naphthalimide derivatives with amide and long carbon chain | |
| Liang et al. | Fluorescence live cell imaging revealed wogonin targets mitochondria | |
| CN110003291B (en) | A fluoroglycosyl-modified paclitaxel compound and its synthesis method and application | |
| CN104059062B (en) | Condensed Ring Compounds Containing Benzothiazole and Triazole Biheterocycles and Their Applications | |
| Xu et al. | Design, Synthesis, and Biological Evaluation of Novel Amyl Ester Tethered Dihydroartemisinin‐Isatin Hybrids as Potent Anti‐Breast Cancer Agents | |
| Fu et al. | Synthesis and pharmacological characterization of glucopyranosyl-conjugated benzyl derivatives as novel selective cytotoxic agents against colon cancer | |
| CN111675919A (en) | PCP and its application in the preparation of antitumor drugs | |
| Lv et al. | Bis-sulfonyl-chalcone-BODIPY molecular probes for in vivo and in vitro imaging | |
| CN119751438A (en) | A 2-(4-hydroxyphenyl)benzothiophene compound using a methylene blue analog as a protein degrader and its application | |
| CN110437156A (en) | Paeonol dihydropyrimidinone derivatives and preparation method and application thereof | |
| CN109384760A (en) | The flavone derivative of base containing mustargen, preparation method and the application of antitumor direction | |
| CN114380856A (en) | Silicon rhodamine derivatives for the detection of hydrogen sulfide in the brain and preparation method and application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |