CN108452805B - A kind of NiTiO3/TiO2 catalyst for photolyzing water to produce hydrogen and its preparation method and use - Google Patents
A kind of NiTiO3/TiO2 catalyst for photolyzing water to produce hydrogen and its preparation method and use Download PDFInfo
- Publication number
- CN108452805B CN108452805B CN201710087506.4A CN201710087506A CN108452805B CN 108452805 B CN108452805 B CN 108452805B CN 201710087506 A CN201710087506 A CN 201710087506A CN 108452805 B CN108452805 B CN 108452805B
- Authority
- CN
- China
- Prior art keywords
- tio
- nitio
- catalyst
- solution
- catalyst according
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000003054 catalyst Substances 0.000 title claims abstract description 70
- 229910052739 hydrogen Inorganic materials 0.000 title claims abstract description 33
- 239000001257 hydrogen Substances 0.000 title claims abstract description 33
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 title claims abstract description 32
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 title claims abstract description 31
- 238000002360 preparation method Methods 0.000 title claims abstract description 18
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 title claims abstract description 17
- 238000006243 chemical reaction Methods 0.000 claims abstract description 19
- 239000002071 nanotube Substances 0.000 claims abstract description 18
- 238000000034 method Methods 0.000 claims abstract description 16
- 238000001027 hydrothermal synthesis Methods 0.000 claims abstract description 9
- 238000011065 in-situ storage Methods 0.000 claims abstract description 3
- 238000006303 photolysis reaction Methods 0.000 claims abstract 2
- 230000015843 photosynthesis, light reaction Effects 0.000 claims abstract 2
- 229910010413 TiO 2 Inorganic materials 0.000 claims description 83
- 239000000243 solution Substances 0.000 claims description 50
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical group CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 41
- 238000003756 stirring Methods 0.000 claims description 26
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 24
- 238000001354 calcination Methods 0.000 claims description 17
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical class [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims description 13
- 239000012670 alkaline solution Substances 0.000 claims description 13
- 235000011114 ammonium hydroxide Nutrition 0.000 claims description 13
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 claims description 12
- 239000003960 organic solvent Substances 0.000 claims description 10
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 claims description 9
- 229910017604 nitric acid Inorganic materials 0.000 claims description 9
- 239000002904 solvent Substances 0.000 claims description 8
- 239000007864 aqueous solution Substances 0.000 claims description 7
- 239000012018 catalyst precursor Substances 0.000 claims description 6
- 238000001035 drying Methods 0.000 claims description 6
- 239000012153 distilled water Substances 0.000 claims description 5
- 239000000463 material Substances 0.000 claims description 5
- 239000002253 acid Substances 0.000 claims description 4
- 239000003513 alkali Substances 0.000 claims description 3
- 239000002114 nanocomposite Substances 0.000 claims description 3
- 239000011858 nanopowder Substances 0.000 claims description 3
- 239000005456 alcohol based solvent Substances 0.000 claims description 2
- 238000000137 annealing Methods 0.000 claims description 2
- 150000004945 aromatic hydrocarbons Chemical class 0.000 claims description 2
- 238000004140 cleaning Methods 0.000 claims description 2
- 239000003759 ester based solvent Substances 0.000 claims description 2
- 239000004210 ether based solvent Substances 0.000 claims description 2
- 238000001914 filtration Methods 0.000 claims description 2
- 150000008282 halocarbons Chemical class 0.000 claims description 2
- 238000002156 mixing Methods 0.000 claims description 2
- 150000002825 nitriles Chemical class 0.000 claims description 2
- 239000002243 precursor Substances 0.000 claims description 2
- 150000002431 hydrogen Chemical class 0.000 claims 1
- 238000004519 manufacturing process Methods 0.000 abstract description 20
- 239000011941 photocatalyst Substances 0.000 abstract description 13
- YZCKVEUIGOORGS-NJFSPNSNSA-N Tritium Chemical compound [3H] YZCKVEUIGOORGS-NJFSPNSNSA-N 0.000 abstract 1
- 239000000203 mixture Substances 0.000 description 17
- 230000001699 photocatalysis Effects 0.000 description 15
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonium chloride Substances [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 9
- QGZKDVFQNNGYKY-UHFFFAOYSA-N ammonia Natural products N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 9
- 230000003197 catalytic effect Effects 0.000 description 7
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- 238000005406 washing Methods 0.000 description 6
- 239000007789 gas Substances 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 3
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 238000002441 X-ray diffraction Methods 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 230000007613 environmental effect Effects 0.000 description 3
- 230000031700 light absorption Effects 0.000 description 3
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 239000003426 co-catalyst Substances 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 238000003912 environmental pollution Methods 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- SOQBVABWOPYFQZ-UHFFFAOYSA-N oxygen(2-);titanium(4+) Chemical compound [O-2].[O-2].[Ti+4] SOQBVABWOPYFQZ-UHFFFAOYSA-N 0.000 description 2
- 238000001259 photo etching Methods 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 239000010970 precious metal Substances 0.000 description 2
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 2
- 230000035484 reaction time Effects 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 238000000967 suction filtration Methods 0.000 description 2
- 238000010998 test method Methods 0.000 description 2
- VZGDMQKNWNREIO-UHFFFAOYSA-N tetrachloromethane Chemical compound ClC(Cl)(Cl)Cl VZGDMQKNWNREIO-UHFFFAOYSA-N 0.000 description 2
- 239000004408 titanium dioxide Substances 0.000 description 2
- 229910052724 xenon Inorganic materials 0.000 description 2
- FHNFHKCVQCLJFQ-UHFFFAOYSA-N xenon atom Chemical compound [Xe] FHNFHKCVQCLJFQ-UHFFFAOYSA-N 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 238000003917 TEM image Methods 0.000 description 1
- 230000032900 absorption of visible light Effects 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 230000007812 deficiency Effects 0.000 description 1
- 238000000921 elemental analysis Methods 0.000 description 1
- 238000005265 energy consumption Methods 0.000 description 1
- 238000004817 gas chromatography Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000002073 nanorod Substances 0.000 description 1
- 238000007146 photocatalysis Methods 0.000 description 1
- 238000013032 photocatalytic reaction Methods 0.000 description 1
- 239000002096 quantum dot Substances 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 239000004065 semiconductor Substances 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 238000004627 transmission electron microscopy Methods 0.000 description 1
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J23/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00
- B01J23/70—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of the iron group metals or copper
- B01J23/74—Iron group metals
- B01J23/755—Nickel
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J35/00—Catalysts, in general, characterised by their form or physical properties
- B01J35/30—Catalysts, in general, characterised by their form or physical properties characterised by their physical properties
- B01J35/39—Photocatalytic properties
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B3/00—Hydrogen; Gaseous mixtures containing hydrogen; Separation of hydrogen from mixtures containing it; Purification of hydrogen
- C01B3/02—Production of hydrogen or of gaseous mixtures containing a substantial proportion of hydrogen
- C01B3/04—Production of hydrogen or of gaseous mixtures containing a substantial proportion of hydrogen by decomposition of inorganic compounds, e.g. ammonia
- C01B3/042—Decomposition of water
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B2203/00—Integrated processes for the production of hydrogen or synthesis gas
- C01B2203/10—Catalysts for performing the hydrogen forming reactions
- C01B2203/1041—Composition of the catalyst
- C01B2203/1047—Group VIII metal catalysts
- C01B2203/1052—Nickel or cobalt catalysts
- C01B2203/1058—Nickel catalysts
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B2203/00—Integrated processes for the production of hydrogen or synthesis gas
- C01B2203/10—Catalysts for performing the hydrogen forming reactions
- C01B2203/1041—Composition of the catalyst
- C01B2203/1094—Promotors or activators
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/30—Hydrogen technology
- Y02E60/36—Hydrogen production from non-carbon containing sources, e.g. by water electrolysis
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Combustion & Propulsion (AREA)
- Inorganic Chemistry (AREA)
- Catalysts (AREA)
Abstract
Description
技术领域technical field
本发明属于半导体光催化领域,具体涉及一种用于光解水产氢的 NiTiO3/TiO2纳米复合材料催化剂及其制备方法和用途。The invention belongs to the field of semiconductor photocatalysis, and in particular relates to a NiTiO 3 /TiO 2 nanocomposite material catalyst for photolyzing water to produce hydrogen and a preparation method and application thereof.
背景技术Background technique
不可再生的化石能源的大量消耗使得能源枯竭以及严重的环境污染成为当前全球面临的一大难题。寻求环境友好型可再生能源成为解决能源危机和环境问题的最佳选择。氢气作为最清洁的能源而引起了广泛关注。直接利用太阳光分解水制备氢气被认为是一种解决能源危机和环境污染问题的有效途径。而利用太阳光分解水的过程离不开光催化剂的催化作用。目前发现的催化剂普遍存在稳定性差,光电转化效率低,环境危害较大,合成成本较高,光腐蚀等问题而无法大规模的应用。二氧化钛具有成本低,化学性质稳定,无污染等特点,被认为是较有前景的光催化剂。然而,二氧化钛的价带较宽对光的吸收利用较为有限,一般会添加助催化剂来增加可见光的吸收提高其光的吸收利用效率。The massive consumption of non-renewable fossil energy has made energy depletion and serious environmental pollution a major problem facing the world today. Seeking environmentally friendly renewable energy has become the best choice to solve the energy crisis and environmental problems. Hydrogen has attracted widespread attention as the cleanest energy source. The direct use of sunlight to split water to produce hydrogen is considered to be an effective way to solve the energy crisis and environmental pollution problems. The use of sunlight to split water is inseparable from the catalytic action of photocatalysts. The catalysts found so far generally suffer from problems such as poor stability, low photoelectric conversion efficiency, greater environmental harm, high synthesis cost, and photocorrosion, which cannot be applied on a large scale. Titanium dioxide has the characteristics of low cost, stable chemical properties, and no pollution, and is considered to be a promising photocatalyst. However, the wider valence band of titanium dioxide has limited light absorption and utilization. Generally, co-catalysts are added to increase the absorption of visible light to improve its light absorption and utilization efficiency.
为了提高光催化剂的催化效率,CN103872174A(申请号201210552885.7) 提供了一种具有可见光吸收特性的Au修饰的TiO2纳米棒阵列光阳极材料的制备方法。利用Au量子点对TiO2表面和底部共同修饰,增加TiO2在可见光范围的吸收范围,提高光解水效率。CN102513129A(申请号201110393837.3)提供了一种光催化TiO2/Cu2O复合薄膜制备方法,并通过修饰Pt提高其光催化制氢性能。然而,以上几种催化剂需要用到贵金属,制备成本较高。此外,这几种催化剂大多数不能实现循环利用,无法达到环保经济的目的。In order to improve the catalytic efficiency of the photocatalyst, CN103872174A (application number 201210552885.7) provides a preparation method of Au-modified TiO2 nanorod array photoanode material with visible light absorption characteristics. The surface and bottom of TiO 2 are co-modified with Au quantum dots, which increases the absorption range of TiO 2 in the visible light range and improves the photo-water splitting efficiency. CN102513129A (application number 201110393837.3) provides a method for preparing a photocatalytic TiO 2 /Cu 2 O composite thin film, and the photocatalytic hydrogen production performance is improved by modifying Pt. However, the above catalysts need to use precious metals, and the preparation cost is high. In addition, most of these catalysts cannot be recycled and cannot achieve the purpose of environmental protection and economy.
发明内容SUMMARY OF THE INVENTION
本发明的目的是解决现有技术的不足,提供一种用于光解水产氢的 NiTiO3/TiO2催化剂及其制备方法,所述催化剂具有提高光解水产氢效率、可循环使用、环保经济等优点,所述制备方法反应条件温和,操作步骤简单。The purpose of the present invention is to solve the deficiencies of the prior art, and to provide a NiTiO 3 /TiO 2 catalyst for photo-splitting water to produce hydrogen and a preparation method thereof. and other advantages, the preparation method has mild reaction conditions and simple operation steps.
为了实现上述目的,本发明提供如下技术方案:In order to achieve the above object, the present invention provides the following technical solutions:
一种用于光解水产氢的NiTiO3/TiO2催化剂,为NiTiO3与TiO2的纳米复合材料。A NiTiO 3 /TiO 2 catalyst for photo-splitting water to produce hydrogen is a nano-composite material of NiTiO 3 and TiO 2 .
根据本发明,所述NiTiO3/TiO2催化剂中NiTiO3/TiO2的摩尔比可以为 (0-0.055):1且不为0;优选为(0.005-0.045):1;进一步优选为(0.01-0.030):1;更进一步优选为(0.012-0.025):1。According to the present invention, the molar ratio of NiTiO 3 /TiO 2 in the NiTiO 3 /TiO 2 catalyst may be (0-0.055):1 and not 0; preferably (0.005-0.045):1; more preferably (0.01) -0.030):1; more preferably (0.012-0.025):1.
根据本发明,所述NiTiO3/TiO2催化剂中的TiO2相为锐钛矿相。According to the present invention, the TiO 2 phase in the NiTiO 3 /TiO 2 catalyst is an anatase phase.
根据本发明,所述NiTiO3/TiO2催化剂为一维纳米管。所述纳米管的长可以介于100-800nm之间;内径可以为3-5nm。According to the present invention, the NiTiO 3 /TiO 2 catalyst is a one-dimensional nanotube. The length of the nanotubes may be between 100-800 nm; the inner diameter may be 3-5 nm.
根据本发明,所述NiTiO3/TiO2催化剂有较大的比表面积,比表面积可以为 80-150m2/g。According to the present invention, the NiTiO 3 /TiO 2 catalyst has a larger specific surface area, and the specific surface area can be 80-150 m 2 /g.
本发明的催化剂具有下述诸多优点:The catalyst of the present invention has the following advantages:
(1)所述NiTiO3/TiO2催化剂相对于TiO2有较高的光催化效率。其中,催化剂中所含有的NiTiO3可以有效提高TiO2纳米管的光催化效率;(1) The NiTiO 3 /TiO 2 catalyst has higher photocatalytic efficiency than TiO 2 . Among them, NiTiO 3 contained in the catalyst can effectively improve the photocatalytic efficiency of TiO 2 nanotubes;
(2)所述NiTiO3/TiO2催化剂可将TiO2纳米管的产氢率提高至少15倍,且其产氢率为P25粉体的至少60倍;(2) The NiTiO 3 /TiO 2 catalyst can increase the hydrogen production rate of TiO 2 nanotubes by at least 15 times, and its hydrogen production rate is at least 60 times higher than that of P25 powder;
(3)所述NiTiO3/TiO2催化剂结构稳定,使用过程中未发现光刻蚀现象;(3) The NiTiO 3 /TiO 2 catalyst has a stable structure, and no photoetching phenomenon is found during use;
(4)所述NiTiO3/TiO2催化剂可以循环使用4次以上,例如可以循环使用至少5次,其光解水产氢效果无明显衰减。(4) The NiTiO 3 /TiO 2 catalyst can be recycled for more than 4 times, for example, it can be recycled for at least 5 times, and the effect of photo-splitting water to produce hydrogen has no obvious attenuation.
本发明还提供如上所述NiTiO3/TiO2催化剂的制备方法,所述NiTiO3/TiO2催化剂以质子化钛酸盐纳米管(以下简称“HTNT”)为前驱体,利用水热原位反应的方法制备。The present invention also provides a method for preparing the above-mentioned NiTiO 3 /TiO 2 catalyst, wherein the NiTiO 3 /TiO 2 catalyst uses protonated titanate nanotubes (hereinafter referred to as "HTNT") as precursors, and utilizes hydrothermal in-situ reaction method prepared.
根据本发明,所述方法包括如下制备步骤:According to the present invention, the method comprises the following preparation steps:
1)制备HTNT,其包括下述步骤:1) prepare HTNT, it comprises the following steps:
1a)将TiO2和碱性溶液进行水热反应;反应完成后,分离得到钛酸盐纳米管;1a) hydrothermally react TiO 2 with an alkaline solution; after the reaction is completed, separate titanate nanotubes;
1b)将步骤1a)的钛酸盐纳米管用水洗涤,洗去多余的碱;再用酸液洗涤,得到质子化钛酸盐纳米管;再次使用水洗涤,有机溶剂洗涤,干燥后即获得 HTNT;1b) Washing the titanate nanotubes of step 1a) with water to remove excess alkali; then washing with acid solution to obtain protonated titanate nanotubes; washing with water again, washing with organic solvents, and drying to obtain HTNTs ;
2)制备NiTiO3/TiO2催化剂前体,其包括:将HTNT与Ni(NO3)2溶液混合,搅拌,再加入碱液调节反应液pH,继续搅拌,过滤,再用H2O和有机溶剂清洗,干燥,得到NiTiO3/TiO2催化剂前体;2) Preparation of NiTiO 3 /TiO 2 catalyst precursor, which includes: mixing HTNT and Ni(NO 3 ) 2 solution, stirring, then adding alkaline solution to adjust the pH of the reaction solution, continuing to stir, filtering, and then using H 2 O and organic Solvent cleaning and drying to obtain NiTiO 3 /TiO 2 catalyst precursor;
3)退火处理制备NiTiO3/TiO2催化剂,其包括:将上述NiTiO3/TiO2催化剂前体进行煅烧即可获得NiTiO3/TiO2催化剂。3) Annealing treatment to prepare a NiTiO 3 /TiO 2 catalyst, which includes: calcining the above-mentioned NiTiO 3 /TiO 2 catalyst precursor to obtain a NiTiO 3 /TiO 2 catalyst.
步骤1a)中,优选地,所述碱性溶液可以为NaOH水溶液;所述碱性溶液的浓度可以为5-10mol/L;In step 1a), preferably, the alkaline solution can be an aqueous NaOH solution; the concentration of the alkaline solution can be 5-10 mol/L;
优选地,所述TiO2可以为纳米粉;Preferably, the TiO 2 can be nano powder;
优选地,所述TiO2的质量和碱性溶液的体积之比(g:mL)可以为(2.0~3.5): (40~100),优选为(2.5~3.0):(60~90),例如为2.5:80;Preferably, the ratio (g:mL) of the mass of the TiO 2 to the volume of the alkaline solution may be (2.0-3.5): (40-100), preferably (2.5-3.0): (60-90), For example, 2.5:80;
优选地,所述水热反应的温度为120-300℃,优选为160-240℃,作为示例性的实例,水热反应温度为160℃;Preferably, the temperature of the hydrothermal reaction is 120-300°C, preferably 160-240°C, as an illustrative example, the temperature of the hydrothermal reaction is 160°C;
优选地,所述水热反应的时间可以为1.5-24h,作为示例性的实例,水热反应时间为24h。Preferably, the time of the hydrothermal reaction may be 1.5-24h, and as an illustrative example, the time of the hydrothermal reaction is 24h.
优选地,所述分离可以为使用真空泵进行抽滤;Preferably, the separation can be suction filtration using a vacuum pump;
步骤1b)中,所述水优选为蒸馏水;In step 1b), the water is preferably distilled water;
优选地,所述酸液可以为浓硝酸与水任意配比的硝酸水溶液;所述硝酸水溶液的浓度可以为0.001mol/L-0.1mol/L,优选为0.005mol/L-0.05mol/L,例如为 0.01mol/L;Preferably, the acid solution can be an aqueous nitric acid solution with an arbitrary ratio of concentrated nitric acid and water; the concentration of the aqueous nitric acid solution can be 0.001mol/L-0.1mol/L, preferably 0.005mol/L-0.05mol/L, For example, 0.01mol/L;
优选地,对于所述有机溶剂没有特别限定,只要其不与产物反应即可。优选地,所述溶剂为对反应试剂呈惰性的溶剂。作为示例性的实例,有机溶剂可以选自腈类溶剂(如乙腈)、芳烃类溶剂(如苯、甲苯)、醇类溶剂(如甲醇、乙醇、异丙醇、正丙醇)、醚类溶剂(如乙醚)、酯类溶剂(如乙酸乙酯)、卤代烃类溶剂(如二氯甲烷、四氯化碳)中的一种或多种。进一步优选地,所述有机溶剂为乙醇。Preferably, the organic solvent is not particularly limited as long as it does not react with the product. Preferably, the solvent is a solvent inert to the reaction reagents. As an illustrative example, the organic solvent may be selected from nitrile solvents (such as acetonitrile), aromatic hydrocarbon solvents (such as benzene, toluene), alcohol solvents (such as methanol, ethanol, isopropanol, n-propanol), ether solvents (such as diethyl ether), one or more of ester solvents (such as ethyl acetate), halogenated hydrocarbon solvents (such as dichloromethane, carbon tetrachloride). Further preferably, the organic solvent is ethanol.
步骤2)中,所述Ni(NO3)2溶液的浓度可以为0.0001-0.1mol/L,优选为 0.001-0.015mol/L;In step 2), the concentration of the Ni(NO 3 ) 2 solution may be 0.0001-0.1 mol/L, preferably 0.001-0.015 mol/L;
优选地,所述Ni(NO3)2与HTNT的摩尔比可以为0:1~0.05:1,优选为0.01: 1~0.02:1,更优选为0.018:1;Preferably, the molar ratio of Ni(NO 3 ) 2 to HTNT may be 0:1 to 0.05:1, preferably 0.01:1 to 0.02:1, more preferably 0.018:1;
优选地,所述HTNT与Ni(NO3)2反应时间可以为5-24h,作为示例性的实例,反应时间为24h;Preferably, the reaction time of the HTNT and Ni(NO 3 ) 2 can be 5-24h, as an exemplary example, the reaction time is 24h;
优选地,所述碱液可以为氨水,NaHCO3水溶液,Na2CO3水溶液中的一种或多种,例如可以使用氨水;Preferably, the alkali solution can be one or more of ammonia water, NaHCO 3 aqueous solution, and Na 2 CO 3 aqueous solution, for example, ammonia water can be used;
优选地,加入碱液调节反应液pH为9-11;Preferably, adding lye to adjust the pH of the reaction solution to be 9-11;
优选地,所述反应液调节pH为9-11后还可以继续搅拌1小时以上,例如可继续搅拌2小时,5小时或10小时;Preferably, after the pH of the reaction solution is adjusted to 9-11, stirring can be continued for more than 1 hour, for example, stirring can be continued for 2 hours, 5 hours or 10 hours;
优选地,所述步骤2)中洗涤使用的有机溶剂具有如上步骤1)中所述的定义。Preferably, the organic solvent used for washing in the step 2) has the definition as described in the above step 1).
步骤3)中,优选地,所述煅烧温度可以为300-600℃,优选为350-450℃,作为示例性的实例,煅烧温度为450℃;In step 3), preferably, the calcination temperature can be 300-600°C, preferably 350-450°C, as an exemplary example, the calcination temperature is 450°C;
优选地,所述煅烧时间可以为1小时以上,例如可以为2小时。Preferably, the calcination time may be more than 1 hour, for example, may be 2 hours.
本发明还提供如上所述NiTiO3/TiO2催化剂的用途,所述NiTiO3/TiO2催化剂可用于催化光解水产氢。The present invention also provides the use of the above-mentioned NiTiO 3 / TiO 2 catalyst, which can be used to catalyze the photo-splitting of water to produce hydrogen.
本发明的有益效果:Beneficial effects of the present invention:
1.本发明的NiTiO3/TiO2催化剂,结构稳定,未发现光刻蚀现象。循环使用4次以上,光解水产氢效果无明显衰减。1. The NiTiO 3 /TiO 2 catalyst of the present invention has a stable structure and no photoetching phenomenon is found. After being used for more than 4 times, the effect of photolyzing water to produce hydrogen has no obvious attenuation.
2.NiTiO3/TiO2催化剂表现为一种独特的一维纳米管状结构,该结构可以显著提高催化剂的光利用效率,从而提高TiO2光解水产氢效率。2. The NiTiO 3 /TiO 2 catalyst exhibits a unique one-dimensional nanotube-like structure, which can significantly improve the photo-utilization efficiency of the catalyst, thereby improving the efficiency of TiO 2 photo-splitting water for hydrogen production.
3.NiTiO3/TiO2催化剂的制备在较低温度下进行,同时反应过程中未引入有毒的贵金属作为助催化剂,降低能耗的同时又不会对环境造成严重影响。3. The preparation of NiTiO 3 /TiO 2 catalyst is carried out at a lower temperature, and no toxic precious metals are introduced as co-catalysts during the reaction process, which reduces energy consumption and does not seriously affect the environment.
附图说明Description of drawings
图1是S1-S9样品的XRD图;其中,(a)为S1-S9样品的XRD图,(b), (c)为局部放大图。Figure 1 is the XRD pattern of the S1-S9 sample; wherein, (a) is the XRD pattern of the S1-S9 sample, and (b) and (c) are partial enlarged images.
图2是S9样品的XRD图。Figure 2 is the XRD pattern of the S9 sample.
图3(a)和(b)分别是S0和S5的透射电镜图。Figures 3(a) and (b) are TEM images of S0 and S5, respectively.
图4是实施例11中S0-S9样品的产氢量随时间变化图。FIG. 4 is a graph showing the variation of hydrogen production with time for S0-S9 samples in Example 11. FIG.
图5是实施例11中S0-S9样品的产氢量随催化剂中的NiTiO3含量变化图。FIG. 5 is a graph showing the variation of the hydrogen production amount of the S0-S9 samples in Example 11 with the NiTiO 3 content in the catalyst.
图6是S5催化剂的循环催化产氢量图。Figure 6 is a graph of the cyclic catalytic hydrogen production of the S5 catalyst.
具体实施方式Detailed ways
本发明的用于光解水产氢的NiTiO3/TiO2催化剂采用下述光催化测试方法测定其光催化效率:The photocatalytic efficiency of the NiTiO 3 /TiO 2 catalyst for photo-splitting water to produce hydrogen of the present invention is measured by the following photocatalytic test method:
i)将0.01-1g本发明的NiTiO3/TiO2催化剂加入光催化系统中的光催化反应器中,再向其中加入100mL纯水或体积分数为5%-50%的空穴牺牲剂水溶液,其中的空穴牺牲剂可为甲醇、乙醇、乙酸、乳酸等。i) Add 0.01-1 g of the NiTiO 3 /TiO 2 catalyst of the present invention into the photocatalytic reactor in the photocatalytic system, and then add 100 mL of pure water or an aqueous solution of a hole sacrificial agent with a volume fraction of 5% to 50%, The hole sacrificing agent can be methanol, ethanol, acetic acid, lactic acid and the like.
ii)启动光催化系统连接的真空泵同时开始搅拌,除去系统内的空气,直至压力值达到负的一个大气压且反应器内液面处无气泡冒出。ii) Start the vacuum pump connected to the photocatalytic system and start stirring at the same time to remove the air in the system until the pressure value reaches a negative one atmosphere and no bubbles emerge from the liquid surface in the reactor.
iii)启动磁控玻璃气泵促进系统内的气体流动,使得气体分散均匀,将模拟自然光的氙灯光源置于反应器上方,打开氙灯即开始光催化反应。iii) Start the magnetron glass air pump to promote the gas flow in the system, so that the gas is dispersed evenly, place the xenon lamp light source simulating natural light above the reactor, and turn on the xenon lamp to start the photocatalytic reaction.
iv)利用光催化系统自带的气相色谱对系统内水分解产生气体进行定时取样分析,1h取样一次,每个样品取样分析10次,确定光催化分解水反应的气体产物种类及其含量。iv) Use the gas chromatography that comes with the photocatalytic system to periodically sample and analyze the gas generated by the water splitting in the system, sample once every 1 hour, and sample and analyze each
下面结合具体实施例,进一步阐述本发明。应理解,这些实施例仅用于说明本发明而不用于限制本发明的范围。此外,应理解,在阅读了本发明所记载的内容之后,本领域技术人员可以对本发明作各种改动或修改,这些等价形式同样落于本发明所限定的范围。The present invention will be further described below in conjunction with specific embodiments. It should be understood that these examples are only used to illustrate the present invention and not to limit the scope of the present invention. In addition, it should be understood that after reading the contents described in the present invention, those skilled in the art can make various changes or modifications to the present invention, and these equivalent forms also fall within the limited scope of the present invention.
除非另有说明,实施例中使用的原料和试剂均为市售物质。Unless otherwise specified, the raw materials and reagents used in the examples are all commercially available materials.
实施例样品的XRD是使用miniflex-600粉末衍射仪进行表征。The XRD of the example samples was characterized using a miniflex-600 powder diffractometer.
实施例样品的透射电镜是使用扫描透射电子显微镜Tecnai G2F20进行表征。Transmission electron microscopy of the example samples was characterized using a scanning transmission electron microscope Tecnai G2F20.
实施例样品的Ni、Ti含量分析测试是使用Ultima 2电感耦合全谱等离子体光谱仪进行表征。The Ni and Ti content analysis and test of the samples of the examples were characterized by using an
实施例1(制备HTNT)Example 1 (Preparation of HTNT)
称取320g NaOH,室温搅拌条件下溶解于1000mL蒸馏水中,配制8mol/L 的NaOH溶液。向10个100mL反应釜中分别加入2.5g TiO2纳米粉和80mL上述NaOH溶液,将反应釜移置鼓风干燥箱中160℃水热反应24h。反应后,利用真空泵抽滤后得到钛酸盐纳米管。Weigh 320 g of NaOH and dissolve it in 1000 mL of distilled water under stirring at room temperature to prepare an 8 mol/L NaOH solution. 2.5g of TiO 2 nanopowder and 80mL of the above-mentioned NaOH solution were added to ten 100mL reaction kettles respectively, and the reaction kettles were moved to a blast drying oven for hydrothermal reaction at 160°C for 24h. After the reaction, the titanate nanotubes are obtained by suction filtration with a vacuum pump.
将水热制备的钛酸盐纳米管经蒸馏水洗3遍,洗去多余NaOH。再用0.01 mol/L硝酸洗10遍,使得钛酸盐纳米管中的钠可以被硝酸中的氢充分置换即钛酸盐质子化。经过蒸馏水洗5遍,乙醇洗3遍,干燥后即获得HTNT,命名为 S0。The hydrothermally prepared titanate nanotubes were washed three times with distilled water to remove excess NaOH. Then wash with 0.01 mol/L nitric acid for 10 times, so that the sodium in the titanate nanotubes can be fully replaced by the hydrogen in the nitric acid, that is, the titanate is protonated. After washing 5 times with distilled water and 3 times with ethanol, HTNT was obtained after drying, which was named S0.
实施例2Example 2
将1g实施例1中合成的HTNT与40mL 0.001mol/L的Ni(NO3)2溶液混合,磁力搅拌24h,再向其中逐滴加入氨水溶液调节溶液pH,直至溶液pH为9-11,继续搅拌5h,利用真空泵抽滤,再用H2O和乙醇反复清洗,干燥。将抽滤产物利用管式炉退火。450℃煅烧2h,获得一维管状NiTiO3/TiO2光催化剂,命名S1。Mix 1 g of HTNT synthesized in Example 1 with 40 mL of 0.001 mol/L Ni(NO 3 ) 2 solution, stir magnetically for 24 h, and then add aqueous ammonia solution dropwise to it to adjust the pH of the solution, until the pH of the solution is 9-11, continue After stirring for 5 h, the mixture was filtered by a vacuum pump, washed repeatedly with H 2 O and ethanol, and dried. The suction filtered product was annealed in a tube furnace. After calcination at 450 °C for 2 h, a one-dimensional tubular NiTiO 3 /TiO 2 photocatalyst was obtained, named S1.
实施例3Example 3
将1g HTNT与40mL 0.002mol/L的Ni(NO3)2溶液混合,磁力搅拌24h,再向其中逐滴加入氨水溶液调节溶液pH,直至溶液pH为9-11,继续搅拌5h,利用真空泵抽滤,再用H2O和乙醇反复清洗,干燥。将抽滤产物利用管式炉退火。 450℃煅烧2h,获得一维管状NiTiO3/TiO2光催化剂,命名为S2。Mix 1 g of HTNT with 40 mL of 0.002 mol/L Ni(NO 3 ) 2 solution, stir magnetically for 24 h, and then add aqueous ammonia solution dropwise to adjust the pH of the solution until the pH of the solution is 9-11, continue stirring for 5 h, and use a vacuum pump to pump filtered, washed repeatedly with H 2 O and ethanol, and dried. The suction filtered product was annealed in a tube furnace. After calcination at 450 °C for 2 h, a one-dimensional tubular NiTiO 3 /TiO 2 photocatalyst was obtained, which was named S2.
实施例4Example 4
将1g实施例1中合成的HTNT与40mL 0.003mol/L的Ni(NO3)2溶液混合,磁力搅拌24h,再向其中逐滴加入氨水溶液调节溶液pH,直至溶液pH为9-11,继续搅拌5h,利用真空泵抽滤,再用H2O和乙醇反复清洗,干燥。将抽滤产物利用管式炉退火。450℃煅烧2h,获得一维管状NiTiO3/TiO2光催化剂,命名S3。Mix 1 g of HTNT synthesized in Example 1 with 40 mL of 0.003 mol/L Ni(NO 3 ) 2 solution, stir magnetically for 24 h, and then add aqueous ammonia solution dropwise to it to adjust the pH of the solution, until the pH of the solution is 9-11, continue After stirring for 5 h, the mixture was filtered by a vacuum pump, washed repeatedly with H 2 O and ethanol, and dried. The suction filtered product was annealed in a tube furnace. After calcination at 450 °C for 2 h, a one-dimensional tubular NiTiO 3 /TiO 2 photocatalyst was obtained, named S3.
实施例5Example 5
将1g实施例1中合成的HTNT与40mL 0.004mol/L的Ni(NO3)2溶液混合,磁力搅拌24h,再向其中逐滴加入氨水溶液调节溶液pH,直至溶液pH为9-11,继续搅拌5h,利用真空泵抽滤,再用H2O和乙醇反复清洗,干燥。将抽滤产物利用管式炉退火。450℃煅烧2h,获得一维管状NiTiO3/TiO2光催化剂,命名S4。Mix 1 g of HTNT synthesized in Example 1 with 40 mL of 0.004 mol/L Ni(NO 3 ) 2 solution, stir magnetically for 24 h, and then add aqueous ammonia solution dropwise to it to adjust the pH of the solution, until the pH of the solution is 9-11, continue After stirring for 5 h, the mixture was filtered by a vacuum pump, washed repeatedly with H 2 O and ethanol, and dried. The suction filtered product was annealed in a tube furnace. After calcination at 450 °C for 2 h, a one-dimensional tubular NiTiO 3 /TiO 2 photocatalyst was obtained, named S4.
实施例6Example 6
将1g实施例1中合成的HTNT与40mL 0.005mol/L的Ni(NO3)2溶液混合,磁力搅拌24h,再向其中逐滴加入氨水溶液调节溶液pH,直至溶液pH为9-11,继续搅拌5h,利用真空泵抽滤,再用H2O和乙醇反复清洗,干燥。将抽滤产物利用管式炉退火。450℃煅烧2h,获得一维管状NiTiO3/TiO2光催化剂,命名S5。Mix 1 g of HTNT synthesized in Example 1 with 40 mL of 0.005 mol/L Ni(NO 3 ) 2 solution, stir magnetically for 24 h, and then add aqueous ammonia solution dropwise to adjust the pH of the solution until the pH of the solution is 9-11, and continue After stirring for 5 h, the mixture was filtered by a vacuum pump, washed repeatedly with H 2 O and ethanol, and dried. The suction filtered product was annealed in a tube furnace. After calcination at 450 °C for 2 h, a one-dimensional tubular NiTiO 3 /TiO 2 photocatalyst was obtained, named S5.
实施例7Example 7
将1g实施例1中合成的HTNT与40mL 0.006mol/L的Ni(NO3)2溶液混合,磁力搅拌24h,再向其中逐滴加入氨水溶液调节溶液pH,直至溶液pH为9-11,继续搅拌5h,利用真空泵抽滤,再用H2O和乙醇反复清洗,干燥。将抽滤产物利用管式炉退火。450℃煅烧2h,获得一维管状NiTiO3/TiO2光催化剂,命名S6。Mix 1 g of HTNT synthesized in Example 1 with 40 mL of 0.006 mol/L Ni(NO 3 ) 2 solution, stir magnetically for 24 h, and then add aqueous ammonia solution dropwise to it to adjust the pH of the solution, until the pH of the solution is 9-11, continue After stirring for 5 h, the mixture was filtered by a vacuum pump, washed repeatedly with H 2 O and ethanol, and dried. The suction filtered product was annealed in a tube furnace. After calcination at 450 °C for 2 h, a one-dimensional tubular NiTiO 3 /TiO 2 photocatalyst was obtained, named S6.
实施例8Example 8
将1g实施例1中合成的HTNT与40mL 0.007mol/L的Ni(NO3)2溶液混合,磁力搅拌24h,再向其中逐滴加入氨水溶液调节溶液pH,直至溶液pH为9-11,继续搅拌5h,利用真空泵抽滤,再用H2O和乙醇反复清洗,干燥。将抽滤产物利用管式炉退火。450℃煅烧2h,获得一维管状NiTiO3/TiO2光催化剂,命名S7。Mix 1 g of HTNT synthesized in Example 1 with 40 mL of 0.007 mol/L Ni(NO 3 ) 2 solution, stir magnetically for 24 h, and add aqueous ammonia solution dropwise to it to adjust the pH of the solution until the pH of the solution is 9-11, continue After stirring for 5 h, the mixture was filtered by a vacuum pump, washed repeatedly with H 2 O and ethanol, and dried. The suction filtered product was annealed in a tube furnace. After calcination at 450 °C for 2 h, a one-dimensional tubular NiTiO 3 /TiO 2 photocatalyst was obtained, named S7.
实施例9Example 9
将1g实施例1中合成的HTNT与40mL 0.008mol/L的Ni(NO3)2溶液混合,磁力搅拌24h,再向其中逐滴加入氨水溶液调节溶液pH,直至溶液pH为9-11,继续搅拌5h,利用真空泵抽滤,再用H2O和乙醇反复清洗,干燥。将抽滤产物利用管式炉退火。450℃煅烧2h,获得一维管状NiTiO3/TiO2光催化剂,命名S8。Mix 1 g of HTNT synthesized in Example 1 with 40 mL of 0.008 mol/L Ni(NO 3 ) 2 solution, stir magnetically for 24 h, and then add aqueous ammonia solution dropwise to it to adjust the pH of the solution, until the pH of the solution is 9-11, continue After stirring for 5 h, the mixture was filtered by a vacuum pump, washed repeatedly with H 2 O and ethanol, and dried. The suction filtered product was annealed in a tube furnace. After calcination at 450 °C for 2 h, a one-dimensional tubular NiTiO 3 /TiO 2 photocatalyst was obtained, named S8.
实施例10Example 10
将1g实施例1中合成的HTNT与40mL 0.015mol/L的Ni(NO3)2溶液混合,磁力搅拌24h,再向其中逐滴加入氨水溶液调节溶液pH,直至溶液pH为9-11,继续搅拌5h,利用真空泵抽滤,再用H2O和乙醇反复清洗,干燥。将抽滤产物利用管式炉退火。450℃煅烧2h,获得一维管状NiTiO3/TiO2光催化剂,命名S9。实施例11(元素分析及光催化产氢测试)Mix 1 g of HTNT synthesized in Example 1 with 40 mL of 0.015 mol/L Ni(NO 3 ) 2 solution, stir magnetically for 24 h, and then add aqueous ammonia solution dropwise to adjust the pH of the solution until the pH of the solution is 9-11, and continue After stirring for 5 h, the mixture was filtered by a vacuum pump, washed repeatedly with H 2 O and ethanol, and dried. The suction filtered product was annealed in a tube furnace. After calcination at 450 °C for 2 h, a one-dimensional tubular NiTiO 3 /TiO 2 photocatalyst was obtained, named S9. Example 11 (elemental analysis and photocatalytic hydrogen production test)
称取0.1g如上实施例1-10中制备的催化剂S0-S9采用上述的测试方法测试其光催化产氢效率,测试结果见图4和图5。Weigh 0.1 g of catalysts S0-S9 prepared in Examples 1-10 above to test their photocatalytic hydrogen production efficiency using the above-mentioned test method. The test results are shown in Figures 4 and 5.
从图4可见,所述催化剂的产氢量随着时间的增加而增加,而纯TiO2(S0) 的产氢量随时间的增加几乎没有变化;且随着NiTiO3含量的增加,其产氢量随时间的增加量增大;再有,在一定范围内,如NiTiO3与TiO2的摩尔比(x:1)的x 介于0~0.0177之间时,随着NiTiO3含量的增加,其产氢量也增加。It can be seen from Figure 4 that the hydrogen production of the catalyst increases with time, while the hydrogen production of pure TiO 2 (S0) hardly changes with time; and with the increase of NiTiO 3 content, its production The amount of hydrogen increases with time; moreover, within a certain range, such as the molar ratio of NiTiO 3 to TiO 2 (x:1), when x is between 0 and 0.0177, with the increase of NiTiO 3 content , its hydrogen production also increased.
从图5可见,NiTiO3/TiO2催化剂产氢速率随着NiTiO3含量增加先上升再下降,而在NiTiO3与TiO2的摩尔比为0.0177:1时产氢速率达到最高。It can be seen from Figure 5 that the hydrogen production rate of the NiTiO 3 /TiO 2 catalyst increases first and then decreases with the increase of NiTiO 3 content, and the hydrogen production rate reaches the highest when the molar ratio of NiTiO 3 to TiO 2 is 0.0177:1.
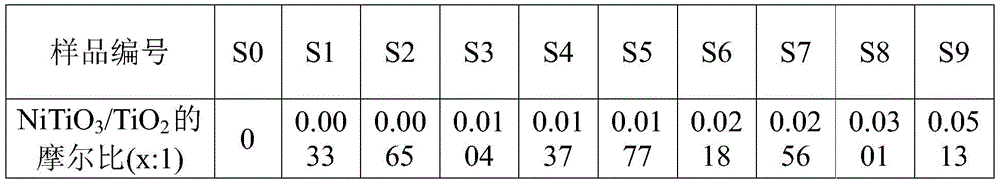
将上述实施例1-10中制备的催化剂S0-S9经ICP含量分析测试,结果见表 1。The catalysts S0-S9 prepared in the above-mentioned examples 1-10 were tested by ICP content analysis, and the results were shown in Table 1.
表1 S1-S9样品中NiTiO3与TiO2的摩尔比Table 1 Molar ratio of NiTiO3 to TiO2 in S1-S9 samples
实施例12(催化剂循环催化产氢测试)Example 12 (catalyst cycle catalytic hydrogen production test)
将S5催化剂催化产氢测试后过滤回收,干燥后直接用于下一次催化产氢测试,测试结果见图6。The S5 catalyst was filtered and recovered after the catalytic hydrogen production test. After drying, it was directly used for the next catalytic hydrogen production test. The test results are shown in Figure 6.
从图6可见催化剂经过五次光催化循环,产氢效果无明显下降,催化剂性能较稳定,可多次循环使用,催化活性几乎没有变化。It can be seen from Figure 6 that the catalyst has undergone five photocatalytic cycles, and the hydrogen production effect does not decrease significantly, the catalyst performance is relatively stable, and it can be recycled for many times, and the catalytic activity has almost no change.
以上,对本发明的实施方式进行了说明。但是,本发明不限定于上述实施方式。凡在本发明的精神和原则之内,所做的任何修改、等同替换、改进等,均应包含在本发明的保护范围之内。The embodiments of the present invention have been described above. However, the present invention is not limited to the above-described embodiments. Any modification, equivalent replacement, improvement, etc. made within the spirit and principle of the present invention shall be included within the protection scope of the present invention.
Claims (17)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201710087506.4A CN108452805B (en) | 2017-02-17 | 2017-02-17 | A kind of NiTiO3/TiO2 catalyst for photolyzing water to produce hydrogen and its preparation method and use |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201710087506.4A CN108452805B (en) | 2017-02-17 | 2017-02-17 | A kind of NiTiO3/TiO2 catalyst for photolyzing water to produce hydrogen and its preparation method and use |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN108452805A CN108452805A (en) | 2018-08-28 |
| CN108452805B true CN108452805B (en) | 2020-06-09 |
Family
ID=63221566
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201710087506.4A Active CN108452805B (en) | 2017-02-17 | 2017-02-17 | A kind of NiTiO3/TiO2 catalyst for photolyzing water to produce hydrogen and its preparation method and use |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN108452805B (en) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110280248B (en) * | 2019-07-18 | 2022-03-01 | 哈尔滨工业大学 | A kind of preparation method of nickel titanate/titanium dioxide nanocomposite material |
| CN112275319B (en) * | 2020-11-03 | 2023-06-06 | 扬州工业职业技术学院 | Organic selenium catalyst loaded with carbon nano tubes and preparation method and application thereof |
| CN112452344B (en) * | 2020-12-14 | 2021-09-14 | 北京科技大学 | In-situ synthesis of monatomic Bi0Bridged TiO2Method for preparing @ BiOCl composite photocatalytic material |
| CN112657516B (en) * | 2021-01-06 | 2023-03-31 | 福州大学 | Direct Z-type photocatalyst and preparation method and application thereof |
| CN114832825B (en) * | 2022-05-18 | 2024-02-09 | 东南大学 | A method for preparing a spherical shell separation double-coated structure catalyst |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3980584A (en) * | 1974-08-21 | 1976-09-14 | Alexei Efimovich Dronov | Carrier and catalyst for chemical processes and method of preparing said catalyst |
| CN102874867A (en) * | 2011-07-15 | 2013-01-16 | 中国科学院理化技术研究所 | Preparation method of monodisperse submicron titanium dioxide microspheres |
| CN103263920B (en) * | 2013-05-16 | 2015-06-17 | 中国科学技术大学 | A kind of TiO2 loaded highly dispersed metal catalyst and preparation method thereof |
| CN105060352B (en) * | 2015-07-22 | 2016-09-21 | 齐鲁工业大学 | A preparation method of nickel oxide nanosheet/titanium dioxide nanorod heterojunction material |
| CN105905940B (en) * | 2016-04-12 | 2017-03-29 | 渤海大学 | A kind of preparation method of nickel titanate/titanium dioxide composite nanomaterial |
-
2017
- 2017-02-17 CN CN201710087506.4A patent/CN108452805B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN108452805A (en) | 2018-08-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN108452805B (en) | A kind of NiTiO3/TiO2 catalyst for photolyzing water to produce hydrogen and its preparation method and use | |
| CN103263920B (en) | A kind of TiO2 loaded highly dispersed metal catalyst and preparation method thereof | |
| CN112675831A (en) | Preparation method of MOF-derived zinc oxide composite titanium dioxide heterojunction and application of heterojunction in photoelectric water decomposition | |
| CN105664929B (en) | A kind of nanometer sheet and preparation method thereof containing noble metal | |
| CN101786005A (en) | Method for preparing cadmium sulfide-titanium dioxide nano-tube composite catalyst | |
| CN109876843B (en) | Copper alloy modified titanium dioxide/carbon nitride heterojunction photocatalyst and preparation method thereof | |
| CN101792117A (en) | Method for preparing tungsten-doped anatase type nano titanium dioxide composite powder | |
| CN108620061B (en) | A preparation method of mesoporous tungsten oxide (WO3) doped bismuth tungstate (Bi2WO6) composite photocatalyst | |
| CN102895963A (en) | Method of loading titanium dioxide nanorod arrays on surface of titanium wire mesh | |
| CN106622291A (en) | Method for preparing zinc oxide/zinc sulfide nano heterojunction photocatalyst | |
| CN108940281A (en) | Novel nano photocatalytic material Ag2MoO4-WO3Method for preparing heterojunction | |
| CN107626297B (en) | A kind of tiny balloon shape bismuth/composite bismuth vanadium photocatalyst and its preparation method and application | |
| CN102744087A (en) | Electrochemistry preparation method for flaky nanometer bismuth oxychloride film photocatalyst | |
| CN106480708A (en) | A kind of Bi2WO6 coating weaving face fabric and preparation method thereof | |
| CN101745430B (en) | Cellulose composite material with photocatalysis activity and preparation method and application thereof | |
| CN113387326B (en) | Application of tin disulfide nano catalyst in piezocatalysis decomposition of water to produce hydrogen | |
| CN102125831B (en) | Preparation Method of Mesoporous Bi2O3/TiO2 Nano Photocatalyst | |
| WO2023272413A1 (en) | Application of tin disulfide nanocatalyst in production of hydrogen by piezoelectric catalytic decomposition of water | |
| CN107952423A (en) | A kind of titanium dioxide high-efficiency photocatalysis material of 2D and preparation method thereof | |
| CN111939949A (en) | Bismuth oxybromide/titanium dioxide nanotube composite material photocatalyst and preparation method thereof | |
| CN106915769A (en) | A kind of ultra-thin mixed crystal titanium dioxide nanoplate and its preparation method and application | |
| CN1868589A (en) | Method for preparing carbon nanometer tube/titanium dioxide composite photocatalyst | |
| CN108187701A (en) | A kind of AgCl/BiOCl photochemical catalyst preparation methods of tubulose AgCl structures | |
| CN113398968A (en) | MOF-derived TiO2Porous g-C3N4Composite photocatalyst and preparation method and application thereof | |
| CN103861631A (en) | Preparation method of nitrogen-doped titanium dioxide nanoribbon visible light photocatalyst |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| TR01 | Transfer of patent right | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20241023 Address after: Room 413A, Building 5, Artificial Intelligence Industrial Park, No. 266 Chuangyan Road, Qilin Science and Technology Innovation Park, Jiangning District, Nanjing City, Jiangsu Province, China 210000 Patentee after: Huaju Holdings (Jiangsu) Co.,Ltd. Country or region after: China Address before: Fuzhou City, Fujian province 350002 Yangqiao Road No. 155 Patentee before: FUJIAN INSTITUTE OF RESEARCH ON THE STRUCTURE OF MATTER, CHINESE ACADEMY OF SCIENCES Country or region before: China |
|
| TR01 | Transfer of patent right | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20241231 Address after: Room 2-697, 1688 Jinshi Road, Baoshan District, Shanghai 200000 Patentee after: Shanghai Mumei Network Technology Co.,Ltd. Country or region after: China Address before: Room 413A, Building 5, Artificial Intelligence Industrial Park, No. 266 Chuangyan Road, Qilin Science and Technology Innovation Park, Jiangning District, Nanjing City, Jiangsu Province, China 210000 Patentee before: Huaju Holdings (Jiangsu) Co.,Ltd. Country or region before: China |